EP0878537A1 - Pruefvorrichtung - Google Patents
Pruefvorrichtung Download PDFInfo
- Publication number
- EP0878537A1 EP0878537A1 EP97911448A EP97911448A EP0878537A1 EP 0878537 A1 EP0878537 A1 EP 0878537A1 EP 97911448 A EP97911448 A EP 97911448A EP 97911448 A EP97911448 A EP 97911448A EP 0878537 A1 EP0878537 A1 EP 0878537A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- liquid
- absorptive
- sheet
- test
- test device
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01L—CHEMICAL OR PHYSICAL LABORATORY APPARATUS FOR GENERAL USE
- B01L3/00—Containers or dishes for laboratory use, e.g. laboratory glassware; Droppers
- B01L3/50—Containers for the purpose of retaining a material to be analysed, e.g. test tubes
- B01L3/502—Containers for the purpose of retaining a material to be analysed, e.g. test tubes with fluid transport, e.g. in multi-compartment structures
- B01L3/5023—Containers for the purpose of retaining a material to be analysed, e.g. test tubes with fluid transport, e.g. in multi-compartment structures with a sample being transported to, and subsequently stored in an absorbent for analysis
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01L—CHEMICAL OR PHYSICAL LABORATORY APPARATUS FOR GENERAL USE
- B01L3/00—Containers or dishes for laboratory use, e.g. laboratory glassware; Droppers
- B01L3/50—Containers for the purpose of retaining a material to be analysed, e.g. test tubes
- B01L3/508—Containers for the purpose of retaining a material to be analysed, e.g. test tubes rigid containers not provided for above
- B01L3/5085—Containers for the purpose of retaining a material to be analysed, e.g. test tubes rigid containers not provided for above for multiple samples, e.g. microtitration plates
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01L—CHEMICAL OR PHYSICAL LABORATORY APPARATUS FOR GENERAL USE
- B01L2300/00—Additional constructional details
- B01L2300/06—Auxiliary integrated devices, integrated components
- B01L2300/069—Absorbents; Gels to retain a fluid
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01L—CHEMICAL OR PHYSICAL LABORATORY APPARATUS FOR GENERAL USE
- B01L2300/00—Additional constructional details
- B01L2300/08—Geometry, shape and general structure
- B01L2300/0861—Configuration of multiple channels and/or chambers in a single devices
- B01L2300/0864—Configuration of multiple channels and/or chambers in a single devices comprising only one inlet and multiple receiving wells, e.g. for separation, splitting
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01L—CHEMICAL OR PHYSICAL LABORATORY APPARATUS FOR GENERAL USE
- B01L2400/00—Moving or stopping fluids
- B01L2400/04—Moving fluids with specific forces or mechanical means
- B01L2400/0403—Moving fluids with specific forces or mechanical means specific forces
- B01L2400/0406—Moving fluids with specific forces or mechanical means specific forces capillary forces
Definitions
- the present invention relates to a test device which, as compared to a conventional microplate, makes chemical or microbiological tests that require multiple reaction items simpler and more expeditious, and which can be mass-produced in an industrially advantageous manner.
- a test employing a microplate of this type reactions or incubations of a microorganism are independently performed in each well of the plate.
- antimicrobial chemicals having different concentrations are added to a plurality of wells, in which the growth of the microorganism of interest is observed so as to thereby determine the Minimum inhibitory concentration.
- test devices usable for such a test there are commercially sold microplates of a type prepared, for example, by dispensing a predetermined amount of an antimicrobial chemical to each well of the microplate and subsequently drying or freezing for storage.
- these test devices unavoidably come to be expensive, since the cost of a microplate itself is considerably high. Therefore, there exists an increased demand for low-price devices.
- Another problem is that, when numerous tests are conducted through use of a single sample, a predetermined amount of sample must be placed dropwise in each well, making operations intricate.
- an object of the present invention is to provide a test device which can be mass-produced at reduced cost, and, inter alia , which permits simple and expeditious chemical or microbiological tests for multiple reaction items.
- the present inventors conducted careful studies and found that, through use of a liquid-absorptive sheet in which the portions other than the portions corresponding to wells (liquid-absorptive sections) of a microplate have undergone hydrophobicity treatment, leakage of test liquid from the liquid-absorptive sections is prevented, the amount of water absorbed by the water absorptive sections is made constant, and such microplates can be mass-produced at reduced cost because hydrophobicity treatment such as printing is employed for production. They also found that when the thus-prepared liquid-absorptive sections are used in combination with a diffusion section for supplying a test liquid, dropwise addition of a test liquid to a single point of the diffusion section enables testing of multiple items. The present invention was achieved based on these findings.
- the present invention provides a sheet-like test device characterized in that the portions other than the portions that function as liquid-absorptive sections of a liquid-absorptive sheet are subjected to a hydrophobicity treatment so that a plurality of water-absorptive portions which are caused to react with a test liquid are formed separately from one another (hereinafter referred to as a "sheet-like test device of the present invention").
- the present invention also provides a test device characterized by comprising the aforementioned sheet-like test device of the present invention and a diffusion section which maintains contact with at least a portion of each liquid-absorptive section and supplies a test liquid to the liquid-absorptive section (hereinafter referred to as a "test device of the present invention").
- liquid-absorptive sheet of the sheet-like test device of the present invention examples include rayon fibers, polyester fibers, acrylic fibers, Vinylon fibers, cotton fibers, glass fibers, and other fibers made of a synthetic or natural material. Of these, acrylic non-woven fabric is preferred in view of its high liquid-absorptive characteristics.
- the liquid-absorptive sheet preferably has a liquid absorption value of 0.020 g/cm 2 or more, preferably 0.08-0.2 g/cm 2 .
- the liquid absorption value of the present invention is obtained in accordance with the following procedure, which is a modification of a method of Crow et al . (INDA-TEC, 207 (1991)).
- the liquid-absorptive sheet used in the present invention preferably has a thickness of 300-3000 ⁇ m, particularly preferably 500-2000 ⁇ m.
- a test liquid is not sufficiently retained in liquid-absorptive sections, and therefore, accurate measurement cannot be performed due to insufficient reaction.
- the thickness is in excess of 3000 ⁇ m, simplicity of the below-mentioned hydrophobicity treatment will be lost.
- the sheet-like test device of the present invention is obtained through hydrophobicity treatment of the portions other than the portions that function as liquid-absorptive sections so that a plurality of liquid-absorptive sections are formed separately from one another.
- the hydrophobicity treatment is preferably conducted by use of a print method; i.e., by allowing permeation of a liquid containing as a predominant component a hydrophobic resin into predetermined portions (other than liquid-absorptive sections) of a liquid-absorptive sheet and drying.
- a print method i.e., by allowing permeation of a liquid containing as a predominant component a hydrophobic resin into predetermined portions (other than liquid-absorptive sections) of a liquid-absorptive sheet and drying.
- the hydrophobic resins used in the present invention include polyurethane resins, polyacrylic resins, polyester resins, polyamide resins, polyvinyl chloride resins, polystyrene resins, fluororesins, and other rubber-modified products. Any of known resins that are conventionally used, such as those employed as ink binders, may be used so long as their water-solubility and moisture-absorptivity are not excessively high
- Solvents which may be used to dissolve or disperse these hydrophobic resins are selected from among a variety of organic solvents in accordance with the hydrophobic resin employed, and examples thereof include toluene or cyclohexanone.
- additives such as a colorant; an inorganic or organic filler; a water repellent such as a fluororesin or silicone; a surfactant; or a viscosity-modifier.
- a hydrophobic ink is preferably applied onto the predetermined portions of a liquid-absorptive sheet for permeation and is subsequently subjected to drying.
- the hydrophobic ink may be applied once, or preferably several times, in order to ensure hydrophobicity treatment.
- any the following methods is preferably performed: (1) a method in which a pressure is applied to the ink-application surface of the sheet, (2) a method in which a suction force is applied to the surface opposite the ink-application surface of the sheet, (3) a method in which air is blown on the side of the ink-application surface of the sheet, or (4) a suitable combination of two or more methods selected from among (1), (2), and (3).
- At least two rolls e.g., a metal roll having a mirror-polished surface and a rubber roll, are used in combination and disposed one on the other.
- a hydrophobic-ink-coated, liquid-absorptive sheet is inserted therebetween so that the ink-application surface contacts the metal roll.
- a pressure is continuously applied between the two rolls while they are rotated.
- the mirror-polished metal roll may be heated.
- application of pressure alone may result in permeation of the hydrophobic ink to the backside of the liquid-absorptive sheet without additional heating.
- a film which does not absorb a hydrophobic ink is placed on the ink-application surface of the liquid-absorptive sheet and an absorptive material such as paper is preferably placed on the surface opposite the ink-application surface of the sheet.
- a pressure onto the ink-application surface of the liquid-absorptive sheet there may be employed a planar pressing method, wherein an unheated or heated sheet which is flat in its entirety or a sheet patterned in accordance with the ink-application pattern of the hydrophobic ink is used.
- the pressurizing plate may be at ambient temperature as in the case of the above-described roll-pressing method. However, when pressure is applied after the ink has dried, the pressurizing plate must be heated so as to soften or melt the hydrophobic ink.
- a film which does not absorb a hydrophobic ink is placed on the ink-application surface of the liquid-absorptive sheet and an absorptive material such as paper is placed on the surface opposite the ink-application surface of the sheet.
- the pressurizing planar plate and the pressure-receiving surface are both subjected to mold-releasing treatment of heat resistance so that the softened or molten hydrophobic ink does not adhere to the pressurizing planar plate and the pressure-receiving surface.
- a vacuum-suction device provided for securing the sheet for printing to the printer itself may be used as is, and if necessary, through adjustment of the diameters of the suction holes or their arrangement pattern, suction force may be applied to ink simultaneously with printing to thereby cause the ink to permeate the sheet.
- a separate but similar suction device may be provided, with which suction force is applied to ink which is not yet dry after printing to thereby permeate the sheet with ink.
- ink may be caused to permeate into the sheet by placing the ink-applied, liquid-absorptive sheet on a wire-net-like receiving base and by applying air pressure onto the ink-application surface of the sheet by use of a blower equipped with a hood.
- chambers of existing air-pressure/vacuum molding machines for forming plastic sheets may also be utilized.
- a separately provided receiving base in the form of a wire net instead of a female die for molding, is attached to the inside of the chamber, and a liquid-absorptive sheet to which ink has been applied is placed on the receiving base. Subsequently, the chamber is closed and air is discharged without the application of vacuum. Pressure is applied onto the ink-application surface by the feeding of compressed air, to thereby achieve permeation of the ink.
- a heating apparatus for heating plastic sheets and a molding apparatus are disposed in line.
- a pressurized-air or vacuum apparatus alone which is manufactured independently, may be connected downstream of a silk screen printer which may be followed by an optional drying apparatus if necessary, to thereby sequentially perform application of ink, permeation of the ink, and drying of the ink.
- liquid-absorptive sections remaining after hydrophobicity treatment is performed, and they may be selected according to the amount of a test liquid and the degree of liquid absorption.
- preferable liquid-absorptive sections are circular or square having, e.g., a diameter or side length of approximately 0.2-1 cm.
- the amount of the test liquid which is caused to be absorbed by a single liquid-absorptive section is approximately 0.025-0.2 ml in general.
- a material fixed with a water-absorptive polymer may be used.
- the water-absorptive polymers include soluble starch; mannan; agar; sodium alginate; plant-derived polysaccharide (such as Arabic gum); microbial polysaccharide (such as dextran, xanthan gum, or Gellan gum); cellulose substances (such as methylcellulose, carboxymethylcellulose, or other species of cellulose); polyvinyl alcohol; a polyacrylic acid sodium salt; and graft copolymers thereof.
- Examples of techniques for fixation of these water-absorptive polymers onto the liquid-absorptive sections include dipping a printed liquid-absorptive sheet in a liquid obtained by dissolving a water-absorptive polymer in water, an organic solvent, etc. and drying; and adding the above water-absorptive polymer liquid dropwise to liquid-absorptive sections and drying.
- the test device of the present invention includes the aforementioned sheet-like test device and a diffusion section which maintains contact with at least a portion of each liquid-absorptive section and supplies a test liquid to the liquid-absorptive section.
- the ratio of the liquid absorption value in the liquid-absorptive sections to that of the diffusion section is 2 or more, preferably 5-15. When the ratio is less than 2, diffusion of the test liquid into the diffusion section for causing the absorptive sections to absorb the test liquid may become difficult.
- Example materials of the diffusion section are the same as the materials listed for the liquid-absorptive sheet, and cotton non-woven fabric is preferred in view of enhanced diffusion characteristics and low liquid absorption.
- preferred ones are those having a liquid absorption value of 0.005-0.05 g/cm 2 , particularly 0.008-0.02 g/cm 2 , and a liquid diffusion rate, as determined by time necessary for diffusion of water for an entirety of a rectangular sheet (1 cm ⁇ 10 cm) when its shorter side is dipped in purified water, of 15 minutes of less, particularly preferably 30 seconds to 3 minutes.
- the diffusion section used in the present invention is provided to supply a test liquid and diffuse it to the liquid-absorptive sections.
- No particular limitation is imposed on the shape, size, and thickness of the diffusion section, and they may be selected according to the liquid absorption value of the test liquid applied to the liquid-absorptive sections, the number of the liquid-absorptive sections, etc.
- the shape of the diffusion section is preferably sheet-like, and those having a thickness of approximately 0.05-0.6 mm are preferably used.
- liquid-absorptive sections of the above-described sheet-like test device are arranged so as to contact the diffusion section at least partially.
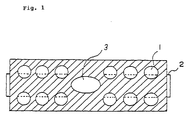
- a more preferable embodiment of the test device of the present invention is a sheet-like test device having a plurality of liquid-absorptive sections, and in addition, at the central location of a plurality of the liquid-absorptive sections, a test-liquid supply hole, and also having a diffusion section made of a liquid-diffusive sheet, wherein the sheet-like test device is arranged such that the test-liquid supply hole and a plurality of liquid-absorptive sections are brought into communication with one another on the diffusion section (See Fig. 1).
- the test device of the present invention is preferably distributed as accommodated in a waterproof casing.
- a sheet-like test device having a waterproof bottom part and a cover having holes corresponding to a plurality of liquid-absorptive sections and a test liquid-supply hole to possess a plurality of liquid-absorptive sections and a test liquid-supply hole, wherein the diffusion section is a liquid-diffusive sheet, and the sheet-like test device arranged such that the test-liquid supply hole and a plurality of liquid-absorptive sections are brought into communication with one another on the diffusion section is secured to the casing (See Fig. 2).
- the cover preferably has an additional outer cover for distribution.
- water-resistant materials used as the bottom part and the cover include plastic sheets made of polyethylene, polypropylene, polystyrene, etc.; and glass.
- test liquid is added dropwise in a specific amount to each liquid-absorptive section.
- the amount of the test liquid is determined according to the area and liquid absorption value of the liquid-absorptive sections.
- test liquid may be poured in a specific amount to only one spot of the diffusion section, and the test liquid thereby diffuses throughout the diffusion section and is absorbed by a plurality of liquid-absorptive sections.
- test liquid may be added dropwise to the liquid-supply holes provided in the liquid-absorptive sections. The amount of the test liquid is determined in accordance with the area, liquid absorption value, etc. of the diffusion section and the liquid-absorptive sections.
- test liquid absorbed by each of the liquid-absorptive sections reacts with a variety of reactive reagents.
- the reactive reagents may be contained in a test device in advance or added after absorption of the liquid. They are preferably contained in advance so that tests may be performed quickly, so long as they do not raise problems in respect to, for example, stability.
- No particular limitation is imposed on the reactive reagents used in the present invention, and examples include reagents for measurement of enzyme activity such as coloring substrates and fluorescent substrates; reagents for identification of microorganisms, such as sugar, amino acids, and fatty acids; reagents for measurement of drug susceptibility of microorganisms against antibiotics, etc.; and test reagents for antibacterial agents.
- reactive reagents may be fixed through addition of a solution of the reagent to pre-sterilized liquid-absorptive sections, and drying under reduced pressure.
- the above-described water-absorptive polymer may be added to the reactive reagent.
- the reactive reagent e.g., L-alanine-p-nitroanilide or L-proline- ⁇ -naphthylamide, which is a coloring substrate used for measurement of enzyme activity in a test liquid (such as suspension of bacteria, body fluid, cerebrospinal fluid, or emulsified foodstuff), is preferably added in an amount of 0.01-0.5 wt.% based on the amount of the test liquid absorbed in the liquid-absorptive sections.
- a test liquid such as suspension of bacteria, body fluid, cerebrospinal fluid, or emulsified foodstuff
- a preferable amount of the reagent is 0.001-0.02 wt.% based on the amount of the test liquid absorbed in the liquid-absorptive sections.
- sugar, amino acids, fatty acids, or their intermediate metabolites are used as reagents for identification of microorganisms, their amounts are preferably 0.1-5 wt.% based on the amount of the test liquid absorbed in liquid-absorptive sections.
- antibiotics are used as reagents for measurement of drug susceptibility of microorganisms, or when antibacterial agents are used for an antibacterial activity test, their concentrations may be arbitrarily determined in accordance with the target drugs and microorganisms of interest.
- gelling agents may also be added to liquid-absorptive sections in order to increase the viscosity of an absorbed test liquid to thereby obtain sufficient gel strength at the liquid-absorptive sections.
- the gelling agents include natural polymers such as xanthan gum, carageenan, Gelan gum, Arabic gum, locust beam gum, guar gum, tragacanth gum, or crystalline cellulose; and synthetic polymers such as polyvinylpyrrolidone, polyvinyl alcohol, polyethylene glycol, polypropylene glycol, polyacrylamide, polyacrylic acid, hydroxyethyl cellulose, or hydroxypropyl cellulose.
- These gelling agents may be fixed onto the liquid-absorptive sections in a manner similar to that employed for fixation of the above-described reagents. In this case, they are preferably added in an amount of 0.1-1 wt.% (natural polymer) or 0.05-0.5 wt.% (synthetic polymer) based on the amount of the test liquid absorbed by the liquid-absorptive sections.
- nutrients such as peptone may be added with an aim of culturing bacteria contained in the test liquid
- cations such as alkali metal salts and alkaline earth metal salts may be added to solutions of reagents so as to increase the gel strength and reaction sensitivity; thus, these components may be incorporated in the liquid-absorptive sections.
- the outer periphery (12 mm x 12 mm) of each of the non-printed portions was cut to make samples for measuring the liquid-absorption value.
- control sample a piece (10 mm x 10 mm) was cut out from a non-printed liquid absorptive piece, and the liquid-absorption value was measured.
- F2B1 Printing was performed consecutively twice from the front face, and subsequent to drying, once from the back face.
- F2 to F4 Consecutively twice to four times from the front face.
- F1B1 Once from the front face, drying, and once from the back face.
- each liquid-absorptive piece After printing, each liquid-absorptive piece, with its printed face upward, was sandwiched between an OPP film placed on the upper side and a wiper placed on the lower side and roll-pressed, to thereby cause the ink to penetrate.
- Liquid-absorptive pieces used in the test Withe the exception of twelve 7.5 mm ⁇ non-printed portions, the entirety of the surface of an acrylic nonwoven fabric (A-200 sheet) having a liquid absorption value of 0.114 g/cm 2 was subjected to uniform printing (print method No. 11). Subsequently, the printed piece was punched to provide a 106 mm x 38 mm rectangular piece, and a 13 mm x 18 mm elliptic hole was formed in its central portion by punching (Fig. 1).
- a PET film (thickness; 75 ⁇ m) was fixed to one side of a cotton nonwoven fabric (PS-140, Asahi Chemical Industry Co., Ltd.) having a liquid absorption value of 0.008 g/cm 2 , and the fabric was then processed into a 15 mm x 110 mm piece by punching.
- Liquid-impermeable base plate and cover The base plate has dimensions that permit accommodation of the above-mentioned liquid-absorptive piece for testing and development layer.
- the cover has openings corresponding to the positions of the non-printed portions and the punched central portion.
- Manufacture method On the base plate were placed the development layer, the control liquid-absorptive piece, and the cover, in this order. Then, the control liquid-absorptive piece and the development layer were secured in place by the base plate placed above and the cover placed below. In order to ensure fixture, the base plate and the cover were fixed to each other with adhesive.
- the development layer of the thus-obtained test device is partially exposed at punched-through portions of the liquid-absorptive piece. Therefore, when the test liquid is added dropwise to such exposed portion(s), the test liquid will reach the liquid-absorptive sections through the development layer (Fig. 2).
- test device obtained in Example 2 tests were performed according to the following test method. Also, comparative tests were performed by the microplate method. The results are shown in Tables 2 and 3.
- Bacterial strain provided for the test:
- a solution was prepared to have the final substrate concentration shown in corresponding parenthesis, and contain 0.5% pepteose peptone (manufactured by DIFCO), 1% casein peptone (manufactured by MARCOR Development Corp.), and 0.1% xanthan gum (manufactured by San-Eigen F.F.I. Inc.).
- Each substrate solution was sterilized and dispensed directly to a test device in an amount of 50 ⁇ l x number of 7.5 mm ⁇ non-printed circular portions of the test device, and dried under reduced pressure.
- each substrate solution was added dropwise to the wells of a microplate in an amount of 100 ⁇ l per well, and immobilized likewise.
- Fresh bacteria were cultured in a sheep blood agar culture media-N "Nissui” (Nissui Pharmaceutical Co., Ltd.) for one day (18 to 24 hours), and suspended in a 0.1% gellan gum (manufactured by MERCK) solution to make the turbidity equivalent to that of a standard nephelometry liquid, McFarland #0.5.
- the bacterium suspension (1 ml) was supplied into each test device through the supply hole open at the central portion thereof. In order to maintain the sealing, a cover was put on the test device.
- the bacterium suspension immediately diffused and was absorbed in the circular non-printed sections within 6 minutes. The amount of absorbed test liquid was 50 ⁇ l ⁇ 10 ⁇ l.
- detection reagents 3, 6: a cinnamaldehyde reagent (bioMerieux Vitek Inc.), 7: Kovac's reagent (Nissui Pharmaceutical Co., Ltd.), 10: nitrite detection reagent I and nitrite detection reagent II (Nissui Pharmaceutical Co., Ltd.)) were added dropwise in an amount of 25 to 50 ⁇ l, and positive(+)/negative(-) was determined after a lapse of 5 to 10 minutes.
- the method using the test device of the present invention showed results identical to those shown by the microplate method. Therefore, through use of the test device of the present invention in which a predetermined amount of test liquid is poured into a single supply hole, can be performed simply and expeditiously a test involving multiple reaction items. Also, when the test device is overturned, the test liquid is not spattered since the test liquid is sealed up in the liquid-absorptive sections, thereby more effectively securing safety in terms of biohazard as compared to microplate-type devices.
- the test device of the present invention can be mass-produced in an industrial advantageous manner, since it can be manufactured through hydrophobicity treatment such as printing using a hydrophobic ink. Further, if the test device is additionally provided with a diffusion section, there is no need to dispense a test liquid to each well as in the case of conventional microplates. This is because the test liquid is poured into a single site of the diffusion section to be absorbed by each liquid-absorptive section , so that a test is conducted simply and expeditiously. Moreover, a test is conducted more expeditiously because a reagent or the like needed for the test can be contained in the absorptive sections in advance.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Hematology (AREA)
- Clinical Laboratory Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Investigating Or Analyzing Non-Biological Materials By The Use Of Chemical Means (AREA)
- Sampling And Sample Adjustment (AREA)
- Investigating Or Analysing Biological Materials (AREA)
- Apparatus Associated With Microorganisms And Enzymes (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP8298474A JPH10136972A (ja) | 1996-11-11 | 1996-11-11 | 試験用具 |
| JP298474/96 | 1996-11-11 | ||
| PCT/JP1997/004020 WO1998021310A1 (fr) | 1996-11-11 | 1997-11-05 | Dispositif de test |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0878537A1 true EP0878537A1 (de) | 1998-11-18 |
| EP0878537A4 EP0878537A4 (de) | 2002-07-31 |
Family
ID=17860179
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP97911448A Withdrawn EP0878537A4 (de) | 1996-11-11 | 1997-11-05 | Pruefvorrichtung |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP0878537A4 (de) |
| JP (1) | JPH10136972A (de) |
| WO (1) | WO1998021310A1 (de) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001090729A2 (en) * | 2000-05-19 | 2001-11-29 | Verification Technologies, Inc. | Method and apparatus for portable product authentication |
| US6512580B1 (en) | 1999-10-27 | 2003-01-28 | Verification Technologies, Inc. | Method and apparatus for portable product authentication |
| WO2011157895A1 (en) * | 2010-06-15 | 2011-12-22 | Åbo Akademi University | Method for manufacturing a sheet-like reaction plate, reaction plate and its use |
| US8163560B2 (en) | 2003-12-04 | 2012-04-24 | Roche Diagnostics Operations, Inc. | Coated test elements |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000116399A (ja) * | 1998-10-14 | 2000-04-25 | Eiken Chem Co Ltd | 薬剤感受性菌・薬剤耐性菌鑑別用分画培地および鑑別方法 |
| US6490030B1 (en) | 1999-01-18 | 2002-12-03 | Verification Technologies, Inc. | Portable product authentication device |
| EP2145683A1 (de) * | 2008-07-14 | 2010-01-20 | F.Hoffmann-La Roche Ag | Analytisches Testelement mit hydrophil modifizierter Oberfläche |
| JP5402721B2 (ja) * | 2010-03-01 | 2014-01-29 | 日立化成株式会社 | 検出装置及び検出方法 |
| KR101656181B1 (ko) * | 2015-03-30 | 2016-09-08 | 경희대학교 산학협력단 | 마이크로 패드 플랫폼 |
| US9914116B2 (en) * | 2015-09-10 | 2018-03-13 | Panasonic Intellectual Property Management Co., Ltd. | Microelement |
| CN116754767A (zh) * | 2023-08-16 | 2023-09-15 | 深圳市第二人民医院(深圳市转化医学研究院) | 自限性自扩散凝集反应卡及其制备方法 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6442455U (de) * | 1987-09-09 | 1989-03-14 | ||
| JPH0528520Y2 (de) * | 1988-12-01 | 1993-07-22 | ||
| JPH04148699A (ja) * | 1990-10-11 | 1992-05-21 | Hitachi Ltd | 生体成分測定用担体 |
| JP3378390B2 (ja) * | 1994-12-14 | 2003-02-17 | 日水製薬株式会社 | 試験用具 |
-
1996
- 1996-11-11 JP JP8298474A patent/JPH10136972A/ja active Pending
-
1997
- 1997-11-05 WO PCT/JP1997/004020 patent/WO1998021310A1/ja not_active Application Discontinuation
- 1997-11-05 EP EP97911448A patent/EP0878537A4/de not_active Withdrawn
Non-Patent Citations (2)
| Title |
|---|
| No further relevant documents disclosed * |
| See also references of WO9821310A1 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6512580B1 (en) | 1999-10-27 | 2003-01-28 | Verification Technologies, Inc. | Method and apparatus for portable product authentication |
| WO2001090729A2 (en) * | 2000-05-19 | 2001-11-29 | Verification Technologies, Inc. | Method and apparatus for portable product authentication |
| WO2001090729A3 (en) * | 2000-05-19 | 2002-04-04 | Verification Technologies Inc | Method and apparatus for portable product authentication |
| US8163560B2 (en) | 2003-12-04 | 2012-04-24 | Roche Diagnostics Operations, Inc. | Coated test elements |
| WO2011157895A1 (en) * | 2010-06-15 | 2011-12-22 | Åbo Akademi University | Method for manufacturing a sheet-like reaction plate, reaction plate and its use |
Also Published As
| Publication number | Publication date |
|---|---|
| JPH10136972A (ja) | 1998-05-26 |
| WO1998021310A1 (fr) | 1998-05-22 |
| EP0878537A4 (de) | 2002-07-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6460171B2 (ja) | 微生物培養シート及びその製造方法 | |
| US20200231930A1 (en) | Microorganism culture sheet | |
| EP0878537A1 (de) | Pruefvorrichtung | |
| CA2142868C (en) | Chemical and microbiological test kit | |
| US5411893A (en) | Dry slide for diagnostic tests | |
| EP1044070B1 (de) | Vorrichtung zur bestimmung von analyten in lösungen | |
| JP3113665B2 (ja) | 微生物乾燥培地 | |
| JP4396078B2 (ja) | 微生物培養器、及びこれを用いた微生物培地 | |
| EP1000169B1 (de) | Verfahren und gerät zum nachweis und zur zählung von mikroorganismen | |
| US5494823A (en) | Apparatus for culturing microorganism | |
| JPH08280377A (ja) | 培養装置 | |
| EP0795600A1 (de) | Erfindung für chemische und mikrobiologische tests | |
| JP6065073B2 (ja) | 微生物培養シート | |
| US3922203A (en) | Culture media in film format for conducting microbial analysis and method of producing the media and loading it into cassettes | |
| JP5724220B2 (ja) | 微生物培養シート、微生物培養シートの製造方法及び培地液注入装置 | |
| JP2009153500A (ja) | 微生物培養シート | |
| JP3378390B2 (ja) | 試験用具 | |
| JPH06181745A (ja) | 化学的及び微生物学的試験用用具 | |
| JP2012143210A (ja) | 微生物培養シート | |
| JPH11299495A (ja) | 試験用具 | |
| JPH08266268A (ja) | 微生物培養具 | |
| JPH11299494A (ja) | 試験用具 | |
| JPS5860999A (ja) | 液体試料中の微生物抑制物質を検出する部材 | |
| JPH01185442A (ja) | 一体型多層分析要素 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19980630 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| A4 | Supplementary search report drawn up and despatched |
Effective date: 20020618 |
|
| AK | Designated contracting states |
Kind code of ref document: A4 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| RIC1 | Information provided on ipc code assigned before grant |
Free format text: 7C 12M 1/00 A, 7G 01N 1/28 B, 7G 01N 21/78 B, 7G 01N 31/22 B, 7G 01N 33/52 B, 7C 12M 1/20 B |
|
| 17Q | First examination report despatched |
Effective date: 20021209 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20030420 |