EP0470640B1 - Apparatus and process for the flame-retardant treatment of textiles - Google Patents
Apparatus and process for the flame-retardant treatment of textiles Download PDFInfo
- Publication number
- EP0470640B1 EP0470640B1 EP91113434A EP91113434A EP0470640B1 EP 0470640 B1 EP0470640 B1 EP 0470640B1 EP 91113434 A EP91113434 A EP 91113434A EP 91113434 A EP91113434 A EP 91113434A EP 0470640 B1 EP0470640 B1 EP 0470640B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- fabric
- chamber
- ammonia
- thp
- gas
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/667—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing phosphorus in the main chain
- D06M15/673—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing phosphorus in the main chain containing phosphorus and nitrogen in the main chain
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/244—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing sulfur or phosphorus
- D06M13/282—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing sulfur or phosphorus with compounds containing phosphorus
- D06M13/285—Phosphines; Phosphine oxides; Phosphine sulfides; Phosphinic or phosphinous acids or derivatives thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06B—TREATING TEXTILE MATERIALS USING LIQUIDS, GASES OR VAPOURS
- D06B19/00—Treatment of textile materials by liquids, gases or vapours, not provided for in groups D06B1/00 - D06B17/00
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06B—TREATING TEXTILE MATERIALS USING LIQUIDS, GASES OR VAPOURS
- D06B23/00—Component parts, details, or accessories of apparatus or machines, specially adapted for the treating of textile materials, not restricted to a particular kind of apparatus, provided for in groups D06B1/00 - D06B21/00
- D06B23/14—Containers, e.g. vats
- D06B23/16—Containers, e.g. vats with means for introducing or removing textile materials without modifying container pressure
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06B—TREATING TEXTILE MATERIALS USING LIQUIDS, GASES OR VAPOURS
- D06B23/00—Component parts, details, or accessories of apparatus or machines, specially adapted for the treating of textile materials, not restricted to a particular kind of apparatus, provided for in groups D06B1/00 - D06B21/00
- D06B23/24—Means for regulating the amount of treating material picked up by the textile material during its treatment
- D06B23/28—Means for regulating the amount of treating material picked up by the textile material during its treatment in response to a test conducted on the treating material
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06B—TREATING TEXTILE MATERIALS USING LIQUIDS, GASES OR VAPOURS
- D06B5/00—Forcing liquids, gases or vapours through textile materials to effect treatment, e.g. washing, dyeing, bleaching, sizing impregnating
- D06B5/02—Forcing liquids, gases or vapours through textile materials to effect treatment, e.g. washing, dyeing, bleaching, sizing impregnating through moving materials of indefinite length
- D06B5/08—Forcing liquids, gases or vapours through textile materials to effect treatment, e.g. washing, dyeing, bleaching, sizing impregnating through moving materials of indefinite length through fabrics
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M11/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with inorganic substances or complexes thereof; Such treatment combined with mechanical treatment, e.g. mercerising
- D06M11/58—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with inorganic substances or complexes thereof; Such treatment combined with mechanical treatment, e.g. mercerising with nitrogen or compounds thereof, e.g. with nitrides
- D06M11/59—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with inorganic substances or complexes thereof; Such treatment combined with mechanical treatment, e.g. mercerising with nitrogen or compounds thereof, e.g. with nitrides with ammonia; with complexes of organic amines with inorganic substances
- D06M11/60—Ammonia as a gas or in solution
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/322—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing nitrogen
- D06M13/44—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing nitrogen containing nitrogen and phosphorus
- D06M13/447—Phosphonates or phosphinates containing nitrogen atoms
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/39—Aldehyde resins; Ketone resins; Polyacetals
- D06M15/423—Amino-aldehyde resins
- D06M15/43—Amino-aldehyde resins modified by phosphorus compounds
- D06M15/431—Amino-aldehyde resins modified by phosphorus compounds by phosphines or phosphine oxides; by oxides or salts of the phosphonium radical
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06B—TREATING TEXTILE MATERIALS USING LIQUIDS, GASES OR VAPOURS
- D06B23/00—Component parts, details, or accessories of apparatus or machines, specially adapted for the treating of textile materials, not restricted to a particular kind of apparatus, provided for in groups D06B1/00 - D06B21/00
- D06B23/14—Containers, e.g. vats
- D06B23/18—Sealing arrangements
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06B—TREATING TEXTILE MATERIALS USING LIQUIDS, GASES OR VAPOURS
- D06B9/00—Solvent-treatment of textile materials
- D06B9/06—Solvent-treatment of textile materials with recovery of the solvent
Definitions
- This invention relates to an apparatus for use in the flame-retardant treatment of textiles, in particular cellulosic textiles, and to a process employing said apparatus.
- Cellulosic textiles are generally rendered flame-retardant by impregnation with an aqueous solution of a tetrakis (hydroxyorgano) phosphonium (THP) composition.
- THP composition may be a THP salt, a partly-neutralized derivative thereof or a condensate with a nitrogen- containing compound such as urea. Impregnation with the THP composition is followed by drying and curing with ammonia gas to produce a cured, water- insoluble polymer in association with the textile.
- the ammonia gas may be passed directly into a chamber through which the textile fabric passes, or, preferably, the ammonia gas is forced through the fabric inside the chamber.
- the apparatus has several disadvantages, notably it is not capable of giving a satisfactory cure at processing speeds in excess of about 20 metres/minute; the level of curing can sometimes be less than ideal so that retreatment of the fabric may be necessary; and the relatively large amounts of ammonia which are used result in the loss of ammonia to the atmosphere, with resultant atmospheric pollution.
- US-A-3846155 describes an apparatus and process for the flame-retardant treatment of fabrics in which an attempt is made to minimise atmospheric pollution by the reduction of the level of ammonia used to cure the fabric. This process has the disadvantage that adequate levels of curing may not be achieved.
- the present invention provides an improved apparatus which is capable of operating at higher processing speeds; which gives a consistently high level of curing efficiency which is not susceptible to variation with wide variations of processing parameters such as fabric speed, fabric moisture content, ammonia concentration, and curing temperature; and which utilizes an optimal amount of ammonia for achieving the cure so that the quantities of ammonia employed are considerably reduced and environmental pollution is minimised.
- the present invention provides a process for the flame-retardant treatment of a cellulosic fabric, said process comprising the following stages:
- the present invention also provides apparatus for carrying out the process described in the immediately-preceding paragraph, said apparatus comprising a chamber (1), entry (12) and exit (13) points, sealed relative to the chamber in a subtantially gas-tight manner, for the fabric respectively into and out of the chamber (1), at least one duct (3) located within the chamber (1) and having at least one orifice which (or the totality of which) extends over the width of said fabric, means to close any portion of said orifice not covered by said fabric, means to cause the fabric to travel in contact with the duct (3) and over the orifice, characterised by recycling means (6; 7; 8) to remove at least some of said gas from said chamber (1) and to return said gas thereto, sampling means (15) for analysing the ammonia content of the gas, means for measuring the rate of ammonia fed into the chamber (1), and means for measuring the temperature within the chamber (1).
- the present invention also provides a fabric which has been treated in the apparatus, and a fabric which has been rendered flame-retardant according to the process, both as hereinabove described.
- the essential features of the process are that the ammonia feed rate is precisely controlled relative to the amount of fabric being processed and that the curing chamber is pre-filled with ammonia to provide a reservoir of ammonia to allow for any slight variation in the ammonia usage relative to the ammonia input. Under these conditions the temperature in the chamber gradually increases and the concentration of ammonia in the chamber gradually decreases until they both reach a steady-state condition. When this steady state is reached the ammonia usage is in balance with the ammonia input, which has been adjusted to ensure complete polymerisation of the THP compound with minimal carry-over of unreacted ammonia.
- Each of the exit and entry points for the fabric is preferably sealed, in a substantially gas-tight manner, by means of flexible (e.g. rubber) seals or flaps.
- the chamber there is at least one duct for conveying ammonia gas into the chamber, the duct having at least one orifice or perforation through which the ammonia passes into the atmosphere of the chamber, by way of the fabric moving over the orifice or perforation.
- Each perforation may be of round, square, rectangular or elliptical shape, and may be, for example, in the form of a series of slots or a single slot.
- the orifices extend across substantially the whole width of the fabric, whereby substantially all the ammonia passes through the fabric.
- the chamber may contain from one to four ducts, each with one or more orifices emitting ammonia through the fabric.
- means are provided to maintain the contact-angle between fabric and orifice at less than 60°.
- the duct or ducts may be located anywhere in the chamber, they are preferably at a location such that the ammonia is forced through the fabric shortly after the fabric enters the chamber.
- the fabric also preferably passes through the atmosphere of ammonia gas and water vapour which will be present in the chamber.
- the chamber is preferably fitted with a plurality of rollers, which may be powered or idling, so that the fabric can pass in a serpentine configuration over the rollers. After passage through the atmosphere in the chamber the cured fabric leaves the chamber via the exit point.
- the fabric entering the chamber generally contains some moisture and the curing reaction also produces water as a by-product; this water, together with that entering in the fabric, volatilize in the chamber as steam, owing to the heat of the curing reaction.
- the steam concentration in the chamber is preferably kept to a minimum, and this may be achieved by allowing condensation of the steam on the internal walls of the chamber, preferably associated with means to prevent condensed water from dripping on to the fabric.
- This prevention means may comprise a sloping roof to the chamber, together with means for collecting the condensed water at the bottom of the chamber without contact with the fabric, such as a circumferential gully or drain.
- the condensed water may be retained within the chamber but is preferably continuously removed from the chamber.
- the gases in the chamber comprising ammonia and steam, may be passed outside the chamber by way of an exhaust pipe and subsequently returned to the chamber at a position remote from the exhaust pipe, preferably in the lower portion of the chamber.
- the return may be by way of a perforate duct over which the fabric passes, but preferably by way of a perforate pipe emitting the gases into the atmosphere of the chamber.
- the exhaust pipe is located in the chamber near one or more of the ducts carrying the orifices through which fresh ammonia is forced through the fabric, as this is the location of the maximum heat and steam emission.
- the ammonia duct and exhaust pipe are preferably both located in the upper portion of the chamber, while the return pipe is preferably located in the lower portion of the chamber.
- the ammonia duct or the exhaust pipe may be located in the lower portion of the chamber.
- the exhaust and return of gases may be in association with means to condense water in the chamber, but in addition (or alternatively) the exhaust gases may be passed through a cooler to reduce their water content prior to return of the gas to the chamber.
- the gas returning to the chamber may be at about the same temperature as that leaving via the exhaust pipe but is preferably cooler.
- the chamber When the gases are cooled externally, the chamber may be fitted with a heated jacket to reduce condensation in the chamber.
- a cooling jacket may also be used to increase condensation on the chamber walls, thereby reducing or eliminating the need for external condensation means.
- the recycle exhaust and return line for the ammonia would still be present in order to render more uniform the ammonia concentration within the chamber.
- the apparatus of the present invention also contains means for sampling the atmosphere within the chamber, in order that appropriate adjustments may be made to the input feed rate for ammonia, to ensure that a steady ammonia concentration is maintained.
- the chamber is preferably mounted on a base which defines a lower compartment, with which the chamber fabric entry and exit points are in substantially gas-tight contact.
- This lower compartment is also provided with fabric entry and exit points, together with a compartment exit vent, which preferably is under the reduced pressure of an external suction fan.
- the cellulosic fabric is first impregnated with a tetrakis (hydroxyorgano) phosphonium composition.
- the cellulosic fabric is preferably substantially all cellulosic but may comprise cellulose fibres and other fibres coblendable or coweaveable therewith.
- the non- cellulosic fibres are preferably polyester or polyamide fibres but may also be acrylic (especially modacrylic fibres).
- the polyamide fibres may be aliphatic, such as copolymers of alkylene diamines and alkylene dicarboxylic acids (eg. nylon 66) or polylactams (such as nylon 6), or may be aromatic, such as aramid, based on aromatic dicarboxylic acids and phenylene diamines.
- the fabric can contain at least 30% of cellulosic fibres and up to 70% of coblendable fibres (e.g. 10-70% and especially 25-60% of coblendable fibres), such as polyamide fibres.
- Particularly important blended fabrics are those containing cellulosic fibres and polyester fibres.
- the blended fabric suitably contains up to 70% (e.g. up to 60%) of polyester fibres and from 30%, e.g. from 40% upwards, of cellulosic fibres (e.g. 1-70% or 1-60%, such as 15-60%, particularly 22-38% or 38-60% polyester fibres and 30-99% or 40-99% such as 40-85%, particularly 62-78% or 40-62% cellulosic fibres).
- Blended fabrics comprising 40-78% cellulosic fibres and 22-60% polyester fibres or 30-62% cellulosic fibres and 38-70% polyester fibres are preferred.
- the cellulosic fibres are preferably natural cotton, but may be ramie, flax or regenerated fibres e.g. viscose or cuprammonium fibres.
- the polyester may be a condensation product containing structural units from an aliphatic alcohol (e.g. a dihydric alcohol such as ethylene glycol) and an aromatic dicarboxylic acid (e.g. terephthalic acid).
- the fabric may be non-woven, but is preferably woven.
- the cellulosic and other fibres may be an intimate or non-intimate mixture, but the fibres are preferably in the form of a blend of cellulosic fibres and other fibres (e.g. polyester fibres), as in a cospun blend such as cotton/ polyester staple fibre.
- the fibres may be in the form of core spun yarn, with a core of, for example, polyester sheathed in cotton fibres.
- the warp and weft fibres are preferably the same, but may be different e.g. one may be of cotton fibres and the other of polyester/ cotton fibres.
- the term "blend” also includes unions and union/blends as well as core sheath fibres.
- the fabric is preferably one with a weight of 100-1000g/m 2 e.g. 150-400g/m 2 , such as cotton polyester shirting or sheeting or curtain fabric.
- each hydroxyorgano group is preferably an alpha-hydroxyorgano group having from 1 to 9 carbon atoms, especially one of formula HOC(R 1 R 2 ) - wherein each of R 1 and R 2 (which may be the same or different) represents hydrogen or an alkyl group having from 1 to 4 carbon atoms, e.g. methyl or ethyl.
- R' is hydrogen and especially both R 1 and R 2 are hydrogen, as in tetrakis (hydroxymethyl) phosphonium compounds.
- tetrakis (hydroxyorgano) phosphonium compounds in general will hereafter be illustrated with respect to tetrakis (hydroxymethyl) phosphonium (THP) compounds, but corresponding molar amounts of other tetrakis (hydroxyorgano) phosphonium compounds may be used.
- the THP composition is preferably a THP salt mixed with a nitrogen- containing compound condensable therewith, such as melamine, methylolated melamine or urea, or a precondensate of said THP salt and said nitrogen- containing compound, or a THP salt or at least partly neutralized THP salt, e.g. THP hydroxide, with or without said nitrogen-containing compound.
- a nitrogen- containing compound condensable therewith such as melamine, methylolated melamine or urea
- a precondensate of said THP salt and said nitrogen- containing compound or a THP salt or at least partly neutralized THP salt, e.g. THP hydroxide, with or without said nitrogen-containing compound.
- the THP composition is preferably a precondensate of a THP salt, (e.g. THP chloride or THP sulphate and urea, in a molar ratio of urea to THP of 0.05 to 0.8:1, e.g. 0.05 to 0.6:1, such as 0.05 to 0.35:1 or 0.35 to 0.6:1, and is in aqueous solution, usually with a pH of 4 to 6.5 e.g. 4 to 5.
- a THP salt e.g. THP chloride or THP sulphate and urea
- a molar ratio of urea to THP of 0.05 to 0.8:1, e.g. 0.05 to 0.6:1, such as 0.05 to 0.35:1 or 0.35 to 0.6:1, and is in aqueous solution, usually with a pH of 4 to 6.5 e.g. 4 to 5.
- the concentration of organophosphorus compound in the aqueous impregnating solution may be 5 to 50% (expressed by weight as THP + ion), e.g. 15 to 35%, especially 15 to 25%.
- the solution may contain a wetting agent, e.g. a nonionic or anionic wetting agent.
- the fabric is impregnated with the THP solution and the wet fabric squeezed to a wet-pick up of 30 to 130%, e.g. 60 to 100% (based on the original weight of the fabric).
- the fabric after impregnation usually has an organophosphorus pick-up of less than 40% e.g. 10 to 40% such as 10 to 30% (e.g. 15 to 30% and especially 15 to 25% as THP + ion), based on the original weight of the fabric.
- the impregnated fabric is then dried e.g. to a moisture content of 0 to 20%, e.g. 5 to 15%, such as about 10%, the percentage being calculated from the increase in weight of the fabric and the weight of chemicals impregnated.
- the drying may be in a stenter oven or over heated cans e.g. steam cans and may involve heating at 80 to 120°C for 10 minutes to 1 minute.
- the dried fabric then enters the apparatus of the present invention where it is cured with ammonia.
- the fabric passes through the lower compartment fabric entry point, then through the lower compartment and finally enters the chamber via the chamber entry point.
- the fabric is then passed over the orifices and ammonia is forced through it.
- the fabric travels through the ammonia/water vapour atmosphere in the chamber, prior to leaving via the fabric chamber exit point, and (if present) through the lower compartment and lower compartment fabric exit point.
- the chamber When the operation is started, the chamber is filled with ammonia which displaces the air through the fabric entry and exit points and also though additional vents in the base of the chamber which may be provided for this purpose, until the ammonia concentration is at least 40% by volume (as determined be means of the sampler and analyzer).
- the initial ammonia concentration is preferably set at a high level (70% to 90%), in order to provide a reservoir of ammonia which is used to compensate for the higher ammonia usage during the initial warm-up period.
- the fabric is then passed through the chamber using an ammonia feed-rate calculated to provide sufficient ammonia to ensure maximum chemical reaction plus a small margin to allow for the loss of unreacted ammonia.
- the molar relationship between the ammonia input and the phosphorus in the THP composition on the fabric is at least 1.2:1 and preferably between 1.7:1 and 2.2:1 (when the THP composition is a condensate of THP salt and nitrogen-containing compound such as urea) or at least 1.7:1 and preferably between 2.2:1 and 2.7:1 (when the THP composition is a THP salt per se or a partially neutralized derivative thereof).
- the ammonia usage slightly exceeds the ammonia feed rate and the cured fabric takes out from the chamber an excess of unreacted ammonia.
- the ammonia concentration in the chamber falls during this initial stage, but, provided that the exposure time and the concentration remain sufficiently high, this does not reduce the ammonia uptake by the fabric.
- Heat and steam are produced in the reaction and the temperature within the chamber rises.
- the ammonia uptake by the fabric drops as the temperature rises and the chamber eventually reaches a steady-state condition.
- ammonia concentration and the chamber temperature will remain substantially constant and the ammonia usage will balance the ammonia feed-rate.
- the selected ammonia feed rate determines the ammonia usage, which is independent of the ammonia concentration.
- the cure chamber usually contains 1 to 20m (e.g. 10 to 20m) of fabric, moving at a speed of 5 to 100m/min (e.g. 30 to 100m/min and especially 50 to 80m/min).
- the cure chamber contains sufficient fabric to give an exposure time of at least 4 seconds and preferably 10 seconds at the required fabric speed.
- the volume of the cure chamber should be at least 1.5 times the maximum volume of ammonia to be fed per minute.
- the fresh ammonia gas is preferably undiluted, but may be diluted with up to 30% by volume of steam or air.
- the ammonia gas issuing from the duct into the chamber may be at 10 to 120°C but is preferably at a temperature below 100°C, eg 10 to 30°C.
- the fabric is preferably treated with an oxidising agent in order to convert at least some of the trivalent phosphorus in the THP composition to pentavalent phosphorus in the cured resin, followed preferably by neutralization with an aqueous solution of a base and washing with water.
- the oxidizing agent is preferably a peroxy compound, such as aqueous hydrogen peroxide solution.
- the oxidation may be performed with a gas containing molecular oxygen, (preferably air) and particularly by the gas being drawn or blown through the fabric.
- the fabric can be passed over a vacuum slot or perforate tube through which the oxidising gas is blown or sucked.
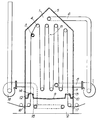
- the apparatus has an upper chamber 1 and a lower compartment 2. Inside chamber 1 is a gassing tube 3 having perforations (not shown) and rollers, exemplified at 4. Chamber 1 has a sloping roof 5, at the apex of which is an exhaust pipe 6 leading from the chamber 1 via a condenser (not shown) and an exhaust fan 7, through a gas return line 8 which leads back into the chamber.
- the chamber 1 has a dished floor 9 with an annular or circumferential gully 10 against the wall 11 of the chamber 1. On either side of floor 9 are fabric entry 12 and fabric exit 13 slots, substantially sealed with rubber flaps 14. Ammonia sampling point 15 is located in the upper part of chamber 1.
- Compartment 2 also contains rollers 4.
- Chamber 1 also preferably has one or more resealable panels (not shown) which can be removed to allow access to the gassing tube and rollers.
- the chamber may be made in two parts, one of which may be removed to allow access.
- impregnated fabric enters the apparatus via slot 16, compartment 2, entry slot 12 and thence into chamber 1.
- the fabric passes over gassing tube 3 through whose perforations ammonia issues through the fabric.
- the fabric then passes over the rollers 4 through the atmosphere of ammonia and steam and thence out of chamber 1 via exit slot 13 compartment 2 and exit slot 17.
- Ammonia steam is if desired sucked by a fan 7 out of chamber 1 via exhaust pipe 6 and if desired water is condensed by a condenser (not shown) before the gas is returned by line 8 and is emitted into chamber 1; any condensate leaves via a condensate line (not shown).
- a precondensate of tetrakis (hydroxymethyl) phosphonium chloride (THPC) and urea was prepared, having a molar ratio of urea to THPC of 0.5:1.0.
- the precondensate was diluted with water to give a solution containing the equivalent of 25% THP + ion.
- a bleached white cotton drill, weighing 280 g/m 2 was padded through this solution to give approximately 80% wet pick-up.
- the fabric was dried for 1 minute at 100°C. It was then cured in a scaled-down version of the apparatus holding 1.2 metres of fabric 0.45 metres wide, using an ammonia input rate of 1.7:1 ammonia to phosphorus and an exposure time of 14.4sec.
- the ammonia concentration in the apparatus was adjusted to different levels before curing commenced.
- the cured fabric was jig-washed in a cold solution of 55g/l hydrogen peroxide, then in 2g/1 sodium carbonate solution at 60°C, and finally in cold water before drying.
- a precondensate as described in Example 1 was diluted to 27.7% THP + ion content. 1000 metres of dyed cotton drill weighing 266/gm 2 and 1.52 metres wide was padded through this solution to give a wet pick-up of 70.3%. The fabric was dried on a four-bay stenter to about 12% moisture content. It was then cured in the cure apparatus (holding 13 metres of fabric). Initially the ammonia concentration was 76%, the speed was set at 32 metres/min and the ammonia flow-rate at 2.2 NH 3 /P. During this stage the temperature rose to 60°C and the ammonia concentration fell to 67% before rising to 69%.
- the processing speed was then increased to 39 metres/min and the relative ammonia flowrate reduced to 1.8 NH 3 /P.
- the fabric was then washed with hydrogen peroxide, soda ash and water in a continuous wash range and dried.
- Example 4 500 metres of white cotton sheeting weighing 178g/m 2 and 1.52 metres wide were treated as described in Example 4 but with a wet pick-up of 92.9%. Curing was carried out immediately after the process of Example 4, so that the starting conditions were ammonia concentration 53%, temperature 73°C, speed 40 metres/min and ammonia flowrate 2.16 NH 3 /P. The temperature remained steady at 73 to 74°C but the ammonia concentration rose to 82%.
- Curing was carried out in three batches with a curing speed of 53 metres/min and an ammonia flowrate of 2.1 NH 3 /P. For all three batches the temperature rose to an equilibrium value of 58 to 60°C and the ammonia concentration fell to within the range 45 to 50%.
Landscapes
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Inorganic Chemistry (AREA)
- Materials Engineering (AREA)
- Chemical Or Physical Treatment Of Fibers (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Treatment Of Fiber Materials (AREA)
- Woven Fabrics (AREA)
- Building Environments (AREA)
- Joints Allowing Movement (AREA)
- Liquid Developers In Electrophotography (AREA)
- Apparatus For Radiation Diagnosis (AREA)
- Encapsulation Of And Coatings For Semiconductor Or Solid State Devices (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB9017537 | 1990-08-10 | ||
| GB909017537A GB9017537D0 (en) | 1990-08-10 | 1990-08-10 | Cure unit |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0470640A1 EP0470640A1 (en) | 1992-02-12 |
| EP0470640B1 true EP0470640B1 (en) | 1996-10-16 |
Family
ID=10680445
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP91113434A Expired - Lifetime EP0470640B1 (en) | 1990-08-10 | 1991-08-09 | Apparatus and process for the flame-retardant treatment of textiles |
Country Status (27)
| Country | Link |
|---|---|
| EP (1) | EP0470640B1 (cs) |
| JP (1) | JP2963559B2 (cs) |
| KR (1) | KR0137645B1 (cs) |
| CN (1) | CN1037162C (cs) |
| AT (1) | ATE144295T1 (cs) |
| AU (1) | AU637925B2 (cs) |
| BR (1) | BR9103412A (cs) |
| CA (1) | CA2048402C (cs) |
| CZ (1) | CZ283768B6 (cs) |
| DE (1) | DE69122697T2 (cs) |
| DK (1) | DK0470640T3 (cs) |
| ES (1) | ES2095275T3 (cs) |
| FI (1) | FI107054B (cs) |
| GB (2) | GB9017537D0 (cs) |
| GR (1) | GR3022191T3 (cs) |
| HU (1) | HU216944B (cs) |
| IE (1) | IE71670B1 (cs) |
| IN (1) | IN183432B (cs) |
| MY (1) | MY107187A (cs) |
| NO (1) | NO177605C (cs) |
| PL (1) | PL168722B1 (cs) |
| PT (1) | PT98631B (cs) |
| RU (1) | RU2032781C1 (cs) |
| SK (1) | SK280774B6 (cs) |
| TR (1) | TR25351A (cs) |
| TW (1) | TW227024B (cs) |
| ZA (1) | ZA916160B (cs) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9412484D0 (en) * | 1994-06-22 | 1994-08-10 | Albright & Wilson | Flame-retardant treatment of fabrics |
| GB2465819A (en) | 2008-12-03 | 2010-06-09 | Rhodia Operations | Flame-retardant treatment of textile materials |
| CN101775721A (zh) * | 2010-03-08 | 2010-07-14 | 江苏华瑞国际实业集团有限公司 | 一种纯棉针织物氨熏方法及阻燃整理方法 |
| US20110275263A1 (en) * | 2010-05-10 | 2011-11-10 | Shulong Li | Flame resistant textile materials |
| AT510909B1 (de) * | 2010-12-20 | 2013-04-15 | Chemiefaser Lenzing Ag | Flammgehemmte cellulosische man-made-fasern |
| RU2572970C2 (ru) | 2011-09-16 | 2016-01-20 | Милликен Энд Компани | Огнеупорная композиция и содержащие ее текстильные материалы |
| GB2497974A (en) | 2011-12-23 | 2013-07-03 | Rhodia Operations | Applying acetoacetamide to textiles, to remove formaldehyde by-product of fire retardant treatment |
| US9453112B2 (en) * | 2013-06-04 | 2016-09-27 | Milliken & Company | Phosphorus-containing polymer, article, and processes for producing the same |
| CN105586745A (zh) * | 2014-10-23 | 2016-05-18 | 山东芦氏特种面料科技有限公司 | 一种阻燃织物氨熏水洗系统 |
| CN105780349B (zh) * | 2014-12-13 | 2018-07-06 | 王深义 | 耐洗阻燃布料氨熏机及其工作方法 |
| CN105128439A (zh) * | 2015-05-26 | 2015-12-09 | 江南大学 | 一种车用阻燃涤麻复合面料 |
| ITUB20155480A1 (it) * | 2015-11-11 | 2017-05-11 | Coramtex Srl | Macchina asciugatrice di tessuto e metodo di asciugatura di tessuto |
| EP3528963B1 (en) | 2016-10-19 | 2022-09-14 | Baldwin Jimek AB | Arrangement at spray nozzle chamber |
| CN121588977A (zh) | 2016-10-19 | 2026-03-03 | Bw转换解决方案公司 | 喷雾喷嘴装置 |
| CN107178615A (zh) * | 2017-07-26 | 2017-09-19 | 芜湖云新材料科技有限公司 | 一种新型气体密封装置 |
| KR101985106B1 (ko) * | 2017-11-10 | 2019-06-03 | 다이텍연구원 | 아라미드섬유계 혼방 제품의 방염가공법 및 이의 방법으로 방염가공된 혼방 제품 |
| CN108221227A (zh) * | 2018-01-30 | 2018-06-29 | 海宁市力天袜业有限公司 | 一种带检测装置的染制袜除味装置 |
| SE543963C2 (en) | 2020-02-28 | 2021-10-12 | Baldwin Jimek Ab | Spray applicator and spray unit comprising two groups of spray nozzles |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1571617A (en) * | 1972-04-17 | 1980-07-16 | Hooker Chemicals Plastics Corp | Apparatus for flame retarding textile materials |
| GB1432602A (en) * | 1972-04-17 | 1976-04-22 | Hooker Chemicals Plastics Corp | Apparatus for treating textile materials |
| GB1439609A (en) * | 1972-08-21 | 1976-06-16 | Albright & Wilson | Flameproofing of textiles |
| GB1439608A (en) * | 1972-08-21 | 1976-06-16 | Albright & Wilson | Flameproofing of textiles |
| US4228497A (en) * | 1977-11-17 | 1980-10-14 | Burroughs Corporation | Template micromemory structure for a pipelined microprogrammable data processing system |
| JPS55112364A (en) * | 1979-02-15 | 1980-08-29 | Santo Tekkosho Kk | Continuous steam treating apparatus of fabric by high pressure steamer |
| ZA826861B (en) * | 1981-09-28 | 1983-07-27 | Albright & Wilson | Flameproofing textiles |
-
1990
- 1990-08-10 GB GB909017537A patent/GB9017537D0/en active Pending
-
1991
- 1991-08-02 CA CA002048402A patent/CA2048402C/en not_active Expired - Lifetime
- 1991-08-05 ZA ZA916160A patent/ZA916160B/xx unknown
- 1991-08-05 IN IN710DE1991 patent/IN183432B/en unknown
- 1991-08-06 TW TW080106186A patent/TW227024B/zh not_active IP Right Cessation
- 1991-08-08 MY MYPI91001437A patent/MY107187A/en unknown
- 1991-08-08 CZ CS912460A patent/CZ283768B6/cs not_active IP Right Cessation
- 1991-08-08 SK SK2460-91A patent/SK280774B6/sk not_active IP Right Cessation
- 1991-08-08 BR BR919103412A patent/BR9103412A/pt not_active IP Right Cessation
- 1991-08-08 GB GB9117090A patent/GB2252570B/en not_active Expired - Lifetime
- 1991-08-08 NO NO913092A patent/NO177605C/no not_active IP Right Cessation
- 1991-08-09 EP EP91113434A patent/EP0470640B1/en not_active Expired - Lifetime
- 1991-08-09 DE DE69122697T patent/DE69122697T2/de not_active Expired - Lifetime
- 1991-08-09 RU SU915001422A patent/RU2032781C1/ru active
- 1991-08-09 HU HU912661A patent/HU216944B/hu unknown
- 1991-08-09 PT PT98631A patent/PT98631B/pt not_active IP Right Cessation
- 1991-08-09 IE IE282491A patent/IE71670B1/en not_active IP Right Cessation
- 1991-08-09 PL PL91291385A patent/PL168722B1/pl unknown
- 1991-08-09 AT AT91113434T patent/ATE144295T1/de not_active IP Right Cessation
- 1991-08-09 DK DK91113434.4T patent/DK0470640T3/da active
- 1991-08-09 ES ES91113434T patent/ES2095275T3/es not_active Expired - Lifetime
- 1991-08-09 AU AU82558/91A patent/AU637925B2/en not_active Expired
- 1991-08-09 FI FI913794A patent/FI107054B/fi not_active IP Right Cessation
- 1991-08-10 CN CN91108556A patent/CN1037162C/zh not_active Expired - Lifetime
- 1991-08-10 KR KR1019910013811A patent/KR0137645B1/ko not_active Expired - Lifetime
- 1991-08-12 TR TR91/0806A patent/TR25351A/xx unknown
- 1991-08-12 JP JP3201931A patent/JP2963559B2/ja not_active Expired - Lifetime
-
1996
- 1996-12-30 GR GR960403668T patent/GR3022191T3/el unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0470640B1 (en) | Apparatus and process for the flame-retardant treatment of textiles | |
| EP0294234B1 (en) | Textile treatment | |
| EP0076138B1 (en) | Flameproofing textiles | |
| US5942006A (en) | Process for the flame-retardant treatment of textiles | |
| US4068026A (en) | Process for flame retarding cellulosics | |
| US4166897A (en) | Phosphorus-containing condensation products, their production and their use as flame retardants | |
| KR920005742A (ko) | 직물의 내염처리(flame retardant treatment)방법 | |
| US4154890A (en) | Process for imparting flame retardant property to cellulosic containing materials | |
| US3919439A (en) | Method of application of THPOH-NH{HD 3 {B fire retardant finish to textiles | |
| Tesoro | The Cross-Linking of Cotton Cellulose with Quaternary Ammonium Derivatives of Bis Chloromethyl Ethers | |
| US4154878A (en) | No-dry process of applying phosphonium salt precondensates to textiles | |
| US3918903A (en) | Dehydration process to impart wrinkle resistance to cellulose-containing fibrous materials | |
| US4137346A (en) | Flame retarding process for proteinaceous material | |
| US3859124A (en) | Durable fire retardant textile materials by anhydrous solvent finishing process | |
| US4058466A (en) | Brominated carbamoyl derivatives | |
| US3932123A (en) | Flameproofing of cotton cellulose with hexamethyl phosphorous triamide | |
| TH18995B (th) | เครื่องสำเร็จและกรรมวิธีการบำบัดผืนผ้าให้หน่วงไฟ | |
| TH20197A (th) | เครื่องสำเร็จและกรรมวิธีการบำบัดผืนผ้าให้หน่วงไฟ | |
| CA1082054A (en) | Apparatus and process for flame retarding cellulosics |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19910809 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IT LI LU NL SE |
|
| 17Q | First examination report despatched |
Effective date: 19940804 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: ALBRIGHT & WILSON UK LIMITED |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AT BE CH DE DK ES FR GR IT LI LU NL SE |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE DK ES FR GR IT LI LU NL SE |
|
| REF | Corresponds to: |
Ref document number: 144295 Country of ref document: AT Date of ref document: 19961115 Kind code of ref document: T |
|
| ITF | It: translation for a ep patent filed | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: E. BLUM & CO. PATENTANWAELTE |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 |
|
| REF | Corresponds to: |
Ref document number: 69122697 Country of ref document: DE Date of ref document: 19961121 |
|
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2095275 Country of ref document: ES Kind code of ref document: T3 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: FG4A Free format text: 3022191 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PFA Owner name: ALBRIGHT & WILSON UK LIMITED Free format text: ALBRIGHT & WILSON UK LIMITED#P.O. BOX 3, 210-222 HAGLEY ROAD WEST#OLDBURY, WARLEY, WEST MIDLANDS B68 0NN (GB) -TRANSFER TO- ALBRIGHT & WILSON UK LIMITED#P.O. BOX 3, 210-222 HAGLEY ROAD WEST#OLDBURY, WARLEY, WEST MIDLANDS B68 0NN (GB) |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20100812 Year of fee payment: 20 Ref country code: ES Payment date: 20100817 Year of fee payment: 20 Ref country code: NL Payment date: 20100810 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20100817 Year of fee payment: 20 Ref country code: LU Payment date: 20100915 Year of fee payment: 20 Ref country code: SE Payment date: 20100812 Year of fee payment: 20 Ref country code: DE Payment date: 20100812 Year of fee payment: 20 Ref country code: FR Payment date: 20100824 Year of fee payment: 20 Ref country code: AT Payment date: 20100812 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GR Payment date: 20100719 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DK Payment date: 20100812 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20100818 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R071 Ref document number: 69122697 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R071 Ref document number: 69122697 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V4 Effective date: 20110809 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EUP |
|
| BE20 | Be: patent expired |
Owner name: *ALBRIGHT & WILSON UK LTD Effective date: 20110809 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: EUG |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20110809 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20120511 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20110810 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20110810 |