CN113527552B - 一种修饰二烷基次膦酸官能团的氯球改性方法 - Google Patents
一种修饰二烷基次膦酸官能团的氯球改性方法 Download PDFInfo
- Publication number
- CN113527552B CN113527552B CN202110774839.0A CN202110774839A CN113527552B CN 113527552 B CN113527552 B CN 113527552B CN 202110774839 A CN202110774839 A CN 202110774839A CN 113527552 B CN113527552 B CN 113527552B
- Authority
- CN
- China
- Prior art keywords
- chlorine
- modified
- acid
- functional group
- carbon
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000460 chlorine Substances 0.000 title claims abstract description 62
- 229910052801 chlorine Inorganic materials 0.000 title claims abstract description 62
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 title claims abstract description 60
- ACVYVLVWPXVTIT-UHFFFAOYSA-N phosphinic acid Chemical group O[PH2]=O ACVYVLVWPXVTIT-UHFFFAOYSA-N 0.000 title claims abstract description 16
- 238000002715 modification method Methods 0.000 title claims abstract description 15
- 239000002253 acid Substances 0.000 claims abstract description 32
- 150000001804 chlorine Chemical class 0.000 claims abstract description 21
- 125000000524 functional group Chemical group 0.000 claims abstract description 17
- 239000000126 substance Substances 0.000 claims abstract description 17
- 239000011203 carbon fibre reinforced carbon Substances 0.000 claims abstract description 16
- 238000000926 separation method Methods 0.000 claims abstract description 12
- 150000001875 compounds Chemical class 0.000 claims abstract description 11
- 238000006243 chemical reaction Methods 0.000 claims abstract description 10
- 238000002360 preparation method Methods 0.000 claims abstract description 7
- -1 amino modified chlorine Chemical class 0.000 claims abstract description 4
- 150000001263 acyl chlorides Chemical class 0.000 claims abstract description 3
- 125000003368 amide group Chemical group 0.000 claims abstract description 3
- 125000001309 chloro group Chemical group Cl* 0.000 claims abstract description 3
- 238000007342 radical addition reaction Methods 0.000 claims abstract description 3
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims abstract 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 21
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 claims description 19
- 239000003960 organic solvent Substances 0.000 claims description 18
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 17
- TVMXDCGIABBOFY-UHFFFAOYSA-N n-Octanol Natural products CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 claims description 14
- 150000003141 primary amines Chemical group 0.000 claims description 14
- NGNBDVOYPDDBFK-UHFFFAOYSA-N 2-[2,4-di(pentan-2-yl)phenoxy]acetyl chloride Chemical compound CCCC(C)C1=CC=C(OCC(Cl)=O)C(C(C)CCC)=C1 NGNBDVOYPDDBFK-UHFFFAOYSA-N 0.000 claims description 12
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 12
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical group CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims description 12
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 claims description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 10
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 9
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 8
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 claims description 8
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 claims description 8
- 235000019400 benzoyl peroxide Nutrition 0.000 claims description 8
- KBPLFHHGFOOTCA-UHFFFAOYSA-N caprylic alcohol Natural products CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 claims description 8
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 6
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims description 6
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 6
- MWKFXSUHUHTGQN-UHFFFAOYSA-N decan-1-ol Chemical compound CCCCCCCCCCO MWKFXSUHUHTGQN-UHFFFAOYSA-N 0.000 claims description 6
- LSXWFXONGKSEMY-UHFFFAOYSA-N di-tert-butyl peroxide Chemical compound CC(C)(C)OOC(C)(C)C LSXWFXONGKSEMY-UHFFFAOYSA-N 0.000 claims description 6
- 239000003999 initiator Substances 0.000 claims description 6
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 6
- 239000008096 xylene Substances 0.000 claims description 6
- BWDBEAQIHAEVLV-UHFFFAOYSA-N 6-methylheptan-1-ol Chemical compound CC(C)CCCCCO BWDBEAQIHAEVLV-UHFFFAOYSA-N 0.000 claims description 5
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 claims description 5
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 claims description 5
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 5
- BWJUFXUULUEGMA-UHFFFAOYSA-N propan-2-yl propan-2-yloxycarbonyloxy carbonate Chemical compound CC(C)OC(=O)OOC(=O)OC(C)C BWJUFXUULUEGMA-UHFFFAOYSA-N 0.000 claims description 5
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 claims description 4
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 claims description 4
- QDHHCQZDFGDHMP-UHFFFAOYSA-N Chloramine Chemical compound ClN QDHHCQZDFGDHMP-UHFFFAOYSA-N 0.000 claims description 4
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 claims description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 4
- 125000003277 amino group Chemical group 0.000 claims description 4
- WGQKYBSKWIADBV-UHFFFAOYSA-N benzylamine Chemical compound NCC1=CC=CC=C1 WGQKYBSKWIADBV-UHFFFAOYSA-N 0.000 claims description 4
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 claims description 4
- 125000004432 carbon atom Chemical group C* 0.000 claims description 4
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 4
- 239000003350 kerosene Substances 0.000 claims description 4
- 239000000463 material Substances 0.000 claims description 4
- 239000003208 petroleum Substances 0.000 claims description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 claims description 4
- WGYKZJWCGVVSQN-UHFFFAOYSA-N propylamine Chemical compound CCCN WGYKZJWCGVVSQN-UHFFFAOYSA-N 0.000 claims description 4
- 150000003335 secondary amines Chemical class 0.000 claims description 4
- CIHOLLKRGTVIJN-UHFFFAOYSA-N tert‐butyl hydroperoxide Chemical compound CC(C)(C)OO CIHOLLKRGTVIJN-UHFFFAOYSA-N 0.000 claims description 4
- BMVXCPBXGZKUPN-UHFFFAOYSA-N 1-hexanamine Chemical compound CCCCCCN BMVXCPBXGZKUPN-UHFFFAOYSA-N 0.000 claims description 3
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 claims description 3
- 229920001577 copolymer Polymers 0.000 claims description 3
- NAQMVNRVTILPCV-UHFFFAOYSA-N hexane-1,6-diamine Chemical compound NCCCCCCN NAQMVNRVTILPCV-UHFFFAOYSA-N 0.000 claims description 3
- DPBLXKKOBLCELK-UHFFFAOYSA-N pentan-1-amine Chemical compound CCCCCN DPBLXKKOBLCELK-UHFFFAOYSA-N 0.000 claims description 3
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 claims description 3
- BJFPYGGTDAYECS-UHFFFAOYSA-N (3-chlorophenyl)methanamine Chemical compound NCC1=CC=CC(Cl)=C1 BJFPYGGTDAYECS-UHFFFAOYSA-N 0.000 claims description 2
- BEQKKZICTDFVMG-UHFFFAOYSA-N 1,2,3,4,6-pentaoxepane-5,7-dione Chemical compound O=C1OOOOC(=O)O1 BEQKKZICTDFVMG-UHFFFAOYSA-N 0.000 claims description 2
- JPZYXGPCHFZBHO-UHFFFAOYSA-N 1-aminopentadecane Chemical compound CCCCCCCCCCCCCCCN JPZYXGPCHFZBHO-UHFFFAOYSA-N 0.000 claims description 2
- XMNIXWIUMCBBBL-UHFFFAOYSA-N 2-(2-phenylpropan-2-ylperoxy)propan-2-ylbenzene Chemical compound C=1C=CC=CC=1C(C)(C)OOC(C)(C)C1=CC=CC=C1 XMNIXWIUMCBBBL-UHFFFAOYSA-N 0.000 claims description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 claims description 2
- WGTASENVNYJZBK-UHFFFAOYSA-N 3,4,5-trimethoxyamphetamine Chemical compound COC1=CC(CC(C)N)=CC(OC)=C1OC WGTASENVNYJZBK-UHFFFAOYSA-N 0.000 claims description 2
- LPULCTXGGDJCTO-UHFFFAOYSA-N 6-methylheptan-1-amine Chemical compound CC(C)CCCCCN LPULCTXGGDJCTO-UHFFFAOYSA-N 0.000 claims description 2
- MHZGKXUYDGKKIU-UHFFFAOYSA-N Decylamine Chemical compound CCCCCCCCCCN MHZGKXUYDGKKIU-UHFFFAOYSA-N 0.000 claims description 2
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 claims description 2
- WJYIASZWHGOTOU-UHFFFAOYSA-N Heptylamine Chemical compound CCCCCCCN WJYIASZWHGOTOU-UHFFFAOYSA-N 0.000 claims description 2
- YIVJZNGAASQVEM-UHFFFAOYSA-N Lauroyl peroxide Chemical compound CCCCCCCCCCCC(=O)OOC(=O)CCCCCCCCCCC YIVJZNGAASQVEM-UHFFFAOYSA-N 0.000 claims description 2
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 claims description 2
- 125000001931 aliphatic group Chemical group 0.000 claims description 2
- JLSIEHRMXULWOA-UHFFFAOYSA-N carboxyoxy 3-ethyloctan-3-yl carbonate Chemical compound CCCCCC(CC)(CC)OC(=O)OOC(O)=O JLSIEHRMXULWOA-UHFFFAOYSA-N 0.000 claims description 2
- BLCKNMAZFRMCJJ-UHFFFAOYSA-N cyclohexyl cyclohexyloxycarbonyloxy carbonate Chemical compound C1CCCCC1OC(=O)OOC(=O)OC1CCCCC1 BLCKNMAZFRMCJJ-UHFFFAOYSA-N 0.000 claims description 2
- 239000007788 liquid Substances 0.000 claims description 2
- YAHRDLICUYEDAU-UHFFFAOYSA-N methylhexaneamine Chemical compound CCC(C)CC(C)N YAHRDLICUYEDAU-UHFFFAOYSA-N 0.000 claims description 2
- 230000004048 modification Effects 0.000 claims description 2
- 238000012986 modification Methods 0.000 claims description 2
- ITZPOSYADVYECJ-UHFFFAOYSA-N n'-cyclohexylpropane-1,3-diamine Chemical compound NCCCNC1CCCCC1 ITZPOSYADVYECJ-UHFFFAOYSA-N 0.000 claims description 2
- FJDUDHYHRVPMJZ-UHFFFAOYSA-N nonan-1-amine Chemical compound CCCCCCCCCN FJDUDHYHRVPMJZ-UHFFFAOYSA-N 0.000 claims description 2
- IOQPZZOEVPZRBK-UHFFFAOYSA-N octan-1-amine Chemical compound CCCCCCCCN IOQPZZOEVPZRBK-UHFFFAOYSA-N 0.000 claims description 2
- HBXNJMZWGSCKPW-UHFFFAOYSA-N octan-2-amine Chemical compound CCCCCCC(C)N HBXNJMZWGSCKPW-UHFFFAOYSA-N 0.000 claims description 2
- 150000002978 peroxides Chemical class 0.000 claims description 2
- 239000011736 potassium bicarbonate Substances 0.000 claims description 2
- 229910000028 potassium bicarbonate Inorganic materials 0.000 claims description 2
- 235000015497 potassium bicarbonate Nutrition 0.000 claims description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 claims description 2
- 235000011181 potassium carbonates Nutrition 0.000 claims description 2
- 235000011118 potassium hydroxide Nutrition 0.000 claims description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 claims description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 claims description 2
- 235000011121 sodium hydroxide Nutrition 0.000 claims description 2
- 238000006467 substitution reaction Methods 0.000 claims description 2
- GJBRNHKUVLOCEB-UHFFFAOYSA-N tert-butyl benzenecarboperoxoate Chemical compound CC(C)(C)OOC(=O)C1=CC=CC=C1 GJBRNHKUVLOCEB-UHFFFAOYSA-N 0.000 claims description 2
- FAGUFWYHJQFNRV-UHFFFAOYSA-N tetraethylenepentamine Chemical compound NCCNCCNCCNCCN FAGUFWYHJQFNRV-UHFFFAOYSA-N 0.000 claims description 2
- VSRBKQFNFZQRBM-UHFFFAOYSA-N tuaminoheptane Chemical compound CCCCCC(C)N VSRBKQFNFZQRBM-UHFFFAOYSA-N 0.000 claims description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims 2
- 229910052799 carbon Inorganic materials 0.000 claims 2
- VILCJCGEZXAXTO-UHFFFAOYSA-N 2,2,2-tetramine Chemical compound NCCNCCNCCN VILCJCGEZXAXTO-UHFFFAOYSA-N 0.000 claims 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 claims 1
- MRIZMKJLUDDMHF-UHFFFAOYSA-N cumene;hydrogen peroxide Chemical compound OO.CC(C)C1=CC=CC=C1 MRIZMKJLUDDMHF-UHFFFAOYSA-N 0.000 claims 1
- 229950000752 methylhexaneamine Drugs 0.000 claims 1
- 229910052761 rare earth metal Inorganic materials 0.000 abstract description 8
- 238000000034 method Methods 0.000 abstract description 7
- 150000002910 rare earth metals Chemical class 0.000 abstract description 7
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 abstract description 5
- 229910052735 hafnium Inorganic materials 0.000 abstract description 5
- VBJZVLUMGGDVMO-UHFFFAOYSA-N hafnium atom Chemical compound [Hf] VBJZVLUMGGDVMO-UHFFFAOYSA-N 0.000 abstract description 5
- 229910052726 zirconium Inorganic materials 0.000 abstract description 5
- 229910021645 metal ion Inorganic materials 0.000 abstract description 4
- 238000001179 sorption measurement Methods 0.000 abstract description 4
- 239000012535 impurity Substances 0.000 abstract description 2
- 238000002414 normal-phase solid-phase extraction Methods 0.000 abstract description 2
- 150000003254 radicals Chemical class 0.000 abstract 1
- 229920006395 saturated elastomer Polymers 0.000 abstract 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 24
- 239000011347 resin Substances 0.000 description 9
- 229920005989 resin Polymers 0.000 description 9
- QUXFOKCUIZCKGS-UHFFFAOYSA-N bis(2,4,4-trimethylpentyl)phosphinic acid Chemical compound CC(C)(C)CC(C)CP(O)(=O)CC(C)CC(C)(C)C QUXFOKCUIZCKGS-UHFFFAOYSA-N 0.000 description 8
- 238000010586 diagram Methods 0.000 description 7
- 239000000706 filtrate Substances 0.000 description 6
- 230000007935 neutral effect Effects 0.000 description 6
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 6
- 238000000605 extraction Methods 0.000 description 5
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 4
- 239000008367 deionised water Substances 0.000 description 3
- 229910021641 deionized water Inorganic materials 0.000 description 3
- 239000012071 phase Substances 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 238000000638 solvent extraction Methods 0.000 description 3
- JUKPJGZUFHCZQI-UHFFFAOYSA-N undecanoyl chloride Chemical compound CCCCCCCCCCC(Cl)=O JUKPJGZUFHCZQI-UHFFFAOYSA-N 0.000 description 3
- UNGJJQCAOKZFCP-UHFFFAOYSA-N CC(CP(O)=O)C(C)C Chemical compound CC(CP(O)=O)C(C)C UNGJJQCAOKZFCP-UHFFFAOYSA-N 0.000 description 2
- HZEFDBCGAGWRPF-UHFFFAOYSA-N OP(=O)CC(C)CC(C)(C)C Chemical compound OP(=O)CC(C)CC(C)(C)C HZEFDBCGAGWRPF-UHFFFAOYSA-N 0.000 description 2
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical compound OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 2
- 229960000583 acetic acid Drugs 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000004587 chromatography analysis Methods 0.000 description 2
- 229910017052 cobalt Inorganic materials 0.000 description 2
- 239000010941 cobalt Substances 0.000 description 2
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- SEGLCEQVOFDUPX-UHFFFAOYSA-N di-(2-ethylhexyl)phosphoric acid Chemical compound CCCCC(CC)COP(O)(=O)OCC(CC)CCCC SEGLCEQVOFDUPX-UHFFFAOYSA-N 0.000 description 2
- 238000003912 environmental pollution Methods 0.000 description 2
- PKNLWLCURZNADF-UHFFFAOYSA-N hex-5-enoyl chloride Chemical compound ClC(=O)CCCC=C PKNLWLCURZNADF-UHFFFAOYSA-N 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- FRPZMMHWLSIFAZ-UHFFFAOYSA-N 10-undecenoic acid Chemical compound OC(=O)CCCCCCCCC=C FRPZMMHWLSIFAZ-UHFFFAOYSA-N 0.000 description 1
- FRIBMENBGGCKPD-UHFFFAOYSA-N 3-(2,3-dimethoxyphenyl)prop-2-enal Chemical compound COC1=CC=CC(C=CC=O)=C1OC FRIBMENBGGCKPD-UHFFFAOYSA-N 0.000 description 1
- ODGHZTUDIMAKNX-UHFFFAOYSA-N CCCCCCP(O)=O Chemical compound CCCCCCP(O)=O ODGHZTUDIMAKNX-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- QOSMNYMQXIVWKY-UHFFFAOYSA-N Propyl levulinate Chemical compound CCCOC(=O)CCC(C)=O QOSMNYMQXIVWKY-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 1
- 238000005411 Van der Waals force Methods 0.000 description 1
- QXZUUHYBWMWJHK-UHFFFAOYSA-N [Co].[Ni] Chemical compound [Co].[Ni] QXZUUHYBWMWJHK-UHFFFAOYSA-N 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- KHAVLLBUVKBTBG-UHFFFAOYSA-N caproleic acid Natural products OC(=O)CCCCCCCC=C KHAVLLBUVKBTBG-UHFFFAOYSA-N 0.000 description 1
- NVIVJPRCKQTWLY-UHFFFAOYSA-N cobalt nickel Chemical compound [Co][Ni][Co] NVIVJPRCKQTWLY-UHFFFAOYSA-N 0.000 description 1
- XUDOZULIAWNMIU-UHFFFAOYSA-N delta-hexenoic acid Chemical compound OC(=O)CCCC=C XUDOZULIAWNMIU-UHFFFAOYSA-N 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 125000001033 ether group Chemical group 0.000 description 1
- 239000002360 explosive Substances 0.000 description 1
- 239000012362 glacial acetic acid Substances 0.000 description 1
- INIGCWGJTZDVRY-UHFFFAOYSA-N hafnium zirconium Chemical compound [Zr].[Hf] INIGCWGJTZDVRY-UHFFFAOYSA-N 0.000 description 1
- GJWAEWLHSDGBGG-UHFFFAOYSA-N hexylphosphonic acid Chemical compound CCCCCCP(O)(O)=O GJWAEWLHSDGBGG-UHFFFAOYSA-N 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000010815 organic waste Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 229940100684 pentylamine Drugs 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 238000004064 recycling Methods 0.000 description 1
- 239000002210 silicon-based material Substances 0.000 description 1
- 235000017550 sodium carbonate Nutrition 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- SCSLUABEVMLYEA-UHFFFAOYSA-N tert-butyl pentanoate Chemical compound CCCCC(=O)OC(C)(C)C SCSLUABEVMLYEA-UHFFFAOYSA-N 0.000 description 1
- MZFGYVZYLMNXGL-UHFFFAOYSA-N undec-10-enoyl chloride Chemical compound ClC(=O)CCCCCCCCC=C MZFGYVZYLMNXGL-UHFFFAOYSA-N 0.000 description 1
- 229960002703 undecylenic acid Drugs 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/40—Introducing phosphorus atoms or phosphorus-containing groups
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/10—Selective adsorption, e.g. chromatography characterised by constructional or operational features
- B01D15/20—Selective adsorption, e.g. chromatography characterised by constructional or operational features relating to the conditioning of the sorbent material
- B01D15/206—Packing or coating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/265—Synthetic macromolecular compounds modified or post-treated polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28002—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their physical properties
- B01J20/28011—Other properties, e.g. density, crush strength
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/28—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties
- B01J20/28014—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof characterised by their form or physical properties characterised by their form
- B01J20/28016—Particle form
- B01J20/28021—Hollow particles, e.g. hollow spheres, microspheres or cenospheres
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/10—Acylation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/30—Introducing nitrogen atoms or nitrogen-containing groups
- C08F8/32—Introducing nitrogen atoms or nitrogen-containing groups by reaction with amines
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Analytical Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
Abstract
本发明公开了一种修饰二烷基次膦酸官能团的氯球改性方法,属于固相萃取分离技术领域。其特征在于首先通过氯球上的氯原子与含有伯胺官能团的化合物进行反应,得到氨基改性的氯球;然后利用氨基与酰氯反应生成的酰胺基团将碳碳双键接枝到氯球表面;再利用碳碳双键与单烷基次膦酸的自由基加成反应得到修饰二烷基次膦酸官能团的改性氯球。本发明公布的修饰二烷基次膦酸官能团的氯球改性方法简单、易于产业化,得到的接枝二烷基次膦酸官能团的改性氯球选择性好、化学稳定性高、饱和吸附量大、不存在萃取剂流失的问题,可广泛用于低浓度稀土富集、稀土深度分离、核级锆铪制备、金属离子除杂等领域。
Description
技术领域
本发明属于固相萃取分离技术领域,尤其是涉及一种氯球的改性及其在低浓度稀土富集、稀土分离、锆铪分离、金属离子除杂等方面的应用。
背景技术
有机磷/膦酸类萃取剂被广泛应用于稀土、镍钴、锆铪等相似元素的溶剂萃取分离,已经商业化的有机磷/膦酸类萃取剂有P204(二(2-乙基己基)磷酸酯),P507(2-乙基己基磷酸单2-乙基己基酯)和Cyanex 272(二(2,4,4-三甲基戊基)次膦酸)。随着烷氧基被烷基取代,其酸性按P204>P507>Cyanex 272顺序递减。酸性越强,萃取稀土离子、钴镍、锆铪的能力越强(萃取性能:P204>P507>Cyanex 272),但相似元素的分离性能降低(分离性能:P204<P507<Cyanex 272)。
虽然Cyanex 272分离稀土、镍钴、锆铪的性能比P507高,但Cyanex 272的饱和负载量低,易乳化,常产生第三相,给生产带来困难。溶剂萃取法还因萃取剂溶于水相造成萃取剂流失,大量有机废水会造成环境污染,且有机溶剂易燃易爆,造成安全隐患。萃取色层法同时具有溶剂萃取法的高选择性和离子交换法的设备简单、高效性的双重优点,试剂投入量、消耗量和萃取剂的流失都大为减少,不存在萃取过程产生的乳化难题,不会有第三相生成,也避免了有机溶剂的大量使用给环境造成的污染,降低了安全隐患。
用于萃取色层法的树脂是关键。很多学者关注于浸渍树脂,即将萃取剂(如P204、P507、Cyanex 272等)通过范德华力以物理方式吸附于大孔树脂或硅基材料的表面。这种树脂使用过程中萃取剂易于流失,影响了萃淋树脂的稳定性和循环性能。以干法制备的浸渍树脂,吸附在基底材料表面的萃取剂往往不止一层,实际应用过程中往往只有表面的萃取剂起到作用,大量萃取剂无法与金属离子接触而起不到吸附的作用。以湿法制备的浸渍树脂,萃取剂吸附量有限。鲜有将二烷基次膦酸类萃取剂通过化学键连接到基底表面的报道。
发明内容
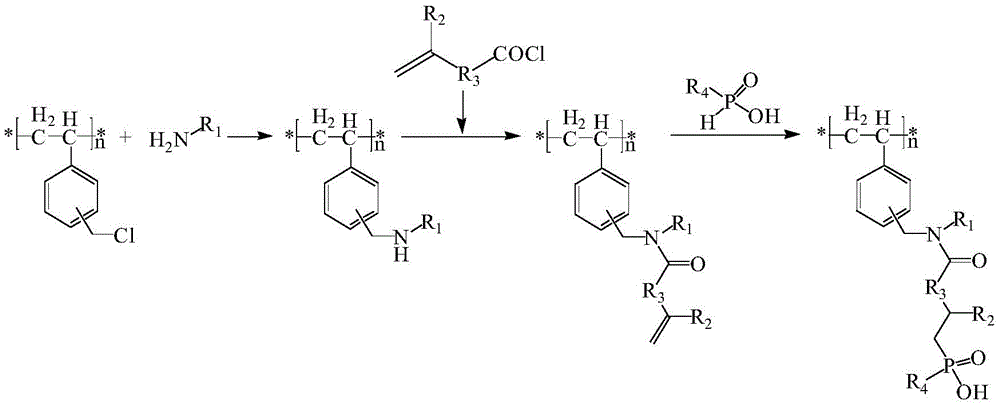
本发明提供一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,首先通过氯球上的氯原子与含有伯胺官能团的化合物进行反应,得到氨基改性的氯球;然后利用氨基与酰氯反应生成的酰胺基团将碳碳双键接枝到氯球表面;再利用碳碳双键与单烷基次膦酸的自由基加成反应得到修饰二烷基次膦酸官能团的改性氯球。
其中氯球为引入了-CH2Cl活性官能团的苯乙烯-二乙烯苯共聚物;R1-NH2为含伯胺的化合物;R2为H或碳原子数为1~8的脂肪族取代基;R3为碳原子数为0~12的脂肪族烷基链;R4为碳原子数为4~12的脂肪族取代基。当R1基团中含有伯胺、仲胺、羟基官能团时,酰氯也可以与这些伯胺、仲胺、羟基官能团反应,从而在氯球表面引入更多的碳碳双键。
所述的含有伯胺官能团的化合物包括但不限于乙胺、丙胺、丁胺、戊胺、己胺、庚胺、辛胺、异辛胺、2-氨基辛烷、壬胺、癸胺、1-氨基十五烷、1,3-二甲基戊胺、2-氨基庚烷、3-氯苄胺、苯胺、苯甲胺、苯乙胺、乙二胺、二乙烯三胺、三乙烯四胺、四乙烯五胺、2-羟基乙胺、1,6-己二胺、N-(3-氨丙基)环己胺。
所述的一种修饰二烷基次膦酸官能团的氯球改性方法,制备步骤如下:
(1)将氯球、含有伯胺官能团的化合物和碱性物质在有机溶剂A中于40~180℃下反应4~24h,经固液分离后得到氨基改性的氯球。其中氯球与含有伯胺官能团的化合物质量比为10:1~1:10,氯球与碱性物质的质量比为20:1~1:10;
(2)向步骤(1)中所得的氨基改性的氯球中加入其体积0.5~20倍的水和/或有机溶剂B,并加入碱性物质,碱性物质与表面接枝氨基的氯球材料的质量比为0.05~2:1;
(3)将具有碳碳双键的酰氯用有机溶剂B溶解,酰氯与有机溶剂B的体积比为1:0.5~10,于0~50℃缓慢滴加到步骤(2)所得的含有氨基改性的氯球和碱性物质的混合体系中,酰氯与表面接枝氨基的氯球的质量比为0.05~3:1,滴加完毕后再反应0~10h,得到表面接枝碳碳双键的氯球。其中酰氯与碱性物质的摩尔比为1:0.5~2;
(4)将步骤(3)所得的表面接枝碳碳双键的氯球与单烷基次膦酸、酸、引发剂和有机溶剂C于80~150℃下反应8~48h,经固液分离得到表面修饰二烷基次膦酸官能团的氯球。
所述的碱性物质为三乙胺、吡啶、碳酸钠、碳酸钾、碳酸氢钠、碳酸氢钾、氢氧化钠或氢氧化钾中的一种或一种以上。
所述的有机溶剂A为1,4-二氧六环、正丁醇、正辛醇、异辛醇、N,N-二甲基甲酰胺、甲苯或二甲苯中的一种或一种以上。
所述的有机溶剂B为乙醚、石油醚、四氢呋喃、1,4-二氧六环、正辛烷、环己烷、磺化煤油、N,N-二甲基甲酰胺、甲苯或二甲苯其中的一种或一种以上。
所述的有机溶剂C为正辛烷、环己烷、石油醚、磺化煤油、四氢呋喃、1,4-二氧六环、正丁醇、正辛醇、异辛醇、癸醇、N,N-二甲基甲酰胺、甲苯或二甲苯其中的一种或一种以上。
所述的酸为醋酸、硫酸、盐酸中的一种或一种以上。
所述的引发剂为叔丁基过氧化氢、异丙苯过氧化氢、过氧化二叔丁基、过氧化二异丙苯、过氧化二苯甲酰、过氧化十二酰、过氧化叔戊酸叔丁酯、过氧化苯甲酸叔丁酯、过氧化二碳酸二异丙酯、过氧化二碳酸二乙基己酯、过氧化二碳酸二环己酯、过氧化二碳酸二苯氧乙酯、偶氮二异丁腈中的一种或一种以上。
所述的表面接枝碳碳双键的氯球与单烷基次膦酸的质量比为5:1~1:5,单烷基次膦酸与酸的摩尔比为5:1~1:2,引发剂质量为单烷基次膦酸质量的0.1~25%。
本发明提供一种修饰二烷基次膦酸官能团的氯球改性方法。苯乙烯-二乙烯苯共聚物机械强度高,化学稳定性好,耐酸碱,是理想的吸附树脂基底材料。氯球比表面积大、具有一定的粒度,用氯球改性的树脂可直接填充在工业级别的离子交换色谱柱中,用于金属离子分离。二烷基次膦酸官能团对稀土、锆铪、镍钴等相似元素具有良好的分离性能,将二烷基次膦酸官能团通过化学键接枝到氯球的表面,不存在萃取剂流失的问题,稳定性好,循环使用性能好,选择性好,产业化应用前景广阔。
附图说明
构成本申请的一部分的附图用来提供对本发明的进一步理解,本发明的示意性实施例及其说明用于解释本发明,并不构成对本发明的不当限定。在附图中:
图1为本发明制备路线示意图,
图2修饰二烷基次膦酸官能团的氯球结构示意图(实施例1),
图3修饰二烷基次膦酸官能团的氯球结构示意图(实施例2),
图4修饰二烷基次膦酸官能团的氯球结构示意图(实施例3)。
具体实施方式
以下结合实施例进一步说明本发明的方法。本发明的实施例用于解释本发明,并不构成对本发明的不当限定。
实施例1
(1)氨基改性氯球:称取10.2g干燥的氯球,加入150mL甲苯溶胀18h,加入12.9g正己胺和10.6g三乙胺,于80℃反应18h。降至室温,过滤,用乙醇洗涤2次、水洗至滤液呈中性,乙醇再洗涤1次后真空干燥,得到氨基改性氯球。
(2)含有C=C双键的酰氯的制备:向18.1g十一烯酸中加入15.2g氯化亚砜,于80℃反应2h,降至室温,通过旋转蒸发仪除去过量氯化亚砜,加入40mL无水乙醚,得到十一烯酰氯的乙醚溶液。
(3)氯球接枝C=C双键:取12.0g氨基改性的氯球,加入50mL无水乙醚和8.2g三乙胺,于常温下通过恒压漏斗缓慢加入十一酰氯的乙醚溶液,滴加完毕后反应4h。过滤,用去离子水洗涤至滤液呈中性,乙醇洗涤,室温晾干。
(4)接枝C=C双键的氯球与单(2,3-二甲基丁基)次膦酸反应:取15.0g接枝C=C双键的氯球、1.2g冰醋酸、5.68g单(2,3-二甲基丁基)次膦酸、0.55g过氧化二叔丁基和20mL正辛烷,回流反应8h,补加过氧化二叔丁基0.35g,再反应8h,再补加过氧化二叔丁基0.35g,再反应12h。降至室温,用乙醇洗涤4次后,于室温晾干,得到修饰二烷基次膦酸官能团的改性氯球(结构示意图如图2所示)。
实施例2
(1)氨基改性氯球:称取20.8g干燥的氯球,加入200mL 1,4-二氧六环溶胀12h,加入18.2g二乙烯三胺和17.6g无水碳酸钠,回流反应12h。降至室温,过滤,用乙醇洗涤2次、水洗至滤液呈中性,乙醇再洗涤1次后室温晾干,得到氨基改性氯球。
(2)氯球接枝C=C双键:取25.0g氨基改性的氯球,加入50mL四氢呋喃和20.2g饱和碳酸氢钠溶液,于常温下通过恒压漏斗缓慢加入含有42.2g十一酰氯的四氢呋喃溶液,滴加完毕后反应6h。过滤,用去离子水洗涤至滤液呈中性,乙醇洗涤后,室温晾干。
(3)接枝C=C双键的氯球与单己基次膦酸反应:取25.0g接枝C=C双键的氯球、20mL异辛醇、1.2mL浓硫酸、18.2g单己基次膦酸和0.80g过氧化二苯甲酰,120℃反应8h,补加过氧化二苯甲酰0.42g,再反应8h,再补加过氧化二苯甲酰0.42g,再反应8h。降至室温,用乙醇洗涤4次后,室温晾干,得到修饰二烷基次膦酸官能团的改性氯球(结构示意图如图3所示)。
实施例3
(1)氨基改性氯球:称取5.2g干燥的氯球,加入80mL N,N-二甲基甲酰胺溶胀10h,加入8.4g 1,6-己二胺和6.6g氢氧化钠,回流反应8h。降至室温,过滤,用乙醇洗涤3次、水洗至滤液呈中性,乙醇再洗涤1次后室温晾干,得到氨基改性氯球。
(2)含有C=C双键的酰氯的制备:向8.1g 5-己烯酸中加入9.2g氯化亚砜,于80℃反应2.5h,降至室温,通过旋转蒸发仪除去过量氯化亚砜,加入20mL干燥的1,4-二氧六环,得到5-己烯酰氯的1,4-二氧六环溶液。
(3)氯球接枝C=C双键:取5.6g氨基改性的氯球,加入20mL环己烷和6.2g碳酸钾溶液,于常温下通过恒压漏斗缓慢加入5-己烯酰氯的1,4-二氧六环溶液,滴加完毕后反应8h。过滤,用去离子水洗涤至滤液呈中性,乙醇洗涤2次后,室温晾干。
(4)接枝C=C双键的氯球与单(2,4,4-三甲基戊基)次膦酸反应:取6.8g接枝C=C双键的氯球、15mL癸醇、0.5mL浓盐酸、12.0g单(2,4,4-三甲基戊基)次膦酸和0.80g过氧化二碳酸二异丙酯,90℃反应6h,补加过氧化二碳酸二异丙酯0.64g,再反应6h,再补加过氧化二碳酸二异丙酯0.64g,再反应8h。降至室温,用乙醇洗涤4次后,室温晾干,得到修饰二烷基次膦酸官能团的改性氯球(结构示意图如图4所示)。
Claims (10)
1.一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,首先通过氯球上的氯原子与含有伯胺官能团的化合物进行反应,得到氨基改性的氯球;然后利用氨基与酰氯反应生成的酰胺基团将碳碳双键接枝到氯球表面;再利用碳碳双键与单烷基次膦酸的自由基加成反应得到修饰二烷基次膦酸官能团的改性氯球;制备路线为:
其中氯球为引入了-CH2Cl活性官能团的苯乙烯-二乙烯苯共聚物;R1-NH2为含伯胺的化合物;R2为H或碳原子数为1~8的脂肪族取代基;R3为碳原子数为0~12的脂肪族烷基链;R4为碳原子数为4~12的脂肪族取代基;当R1基团中含有伯胺、仲胺、羟基官能团时,酰氯也可以与这些伯胺、仲胺、羟基官能团反应,从而在氯球表面引入更多的碳碳双键。
2.如权利要求1所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于所述的含有伯胺官能团的化合物包括但不限于乙胺、丙胺、丁胺、戊胺、己胺、庚胺、辛胺、异辛胺、2-氨基辛烷、壬胺、癸胺、1-氨基十五烷、1,3-二甲基戊胺、2-氨基庚烷、3-氯苄胺、苯胺、苯甲胺、苯乙胺、乙二胺、二乙烯三胺、三乙烯四胺、四乙烯五胺、2-羟基乙胺、1,6-己二胺、N-(3-氨丙基)环己胺。
3.如权利要求1所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,具体制备步骤如下:
(1)将氯球、含有伯胺官能团的化合物和碱性物质在有机溶剂A中于40~180℃下反应4~24h,经固液分离后得到氨基改性的氯球;其中氯球与含有伯胺官能团的化合物质量比为10:1~1:10,氯球与碱性物质的质量比为20:1~1:10;
(2)向步骤(1)中所得的氨基改性的氯球中加入其体积0.5~20倍的水和/或有机溶剂B,并加入碱性物质,碱性物质与表面接枝氨基的氯球材料的质量比为0.05~2:1;
(3)将具有碳碳双键的酰氯用有机溶剂B溶解,酰氯与有机溶剂B的体积比为1:0.5~10,于0~50℃缓慢滴加到步骤(2)所得的含有氨基改性的氯球和碱性物质的混合体系中,酰氯与表面接枝氨基的氯球的质量比为0.05~3:1,滴加完毕后再反应0~10h,得到表面接枝碳碳双键的氯球; 其中酰氯与碱性物质的摩尔比为1:0.5~2;
(4)将步骤(3)所得的表面接枝碳碳双键的氯球与单烷基次膦酸、酸、引发剂和有机溶剂C于80~150℃下反应8~48h,经固液分离得到表面修饰二烷基次膦酸官能团的氯球。
4.根据权利要求3所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,所述的碱性物质为三乙胺、吡啶、碳酸钠、碳酸钾、碳酸氢钠、碳酸氢钾、氢氧化钠或氢氧化钾中的一种或一种以上。
5.根据权利要求3所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,步骤(1)中所述的有机溶剂A为1,4-二氧六环、正丁醇、正辛醇、异辛醇、N,N-二甲基甲酰胺、甲苯或二甲苯中的一种或一种以上。
6.根据权利要求3所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,步骤(2)~(3)中所述的有机溶剂B为乙醚、石油醚、四氢呋喃、1,4-二氧六环、正辛烷、环己烷、磺化煤油、N,N-二甲基甲酰胺、甲苯或二甲苯其中的一种或一种以上。
7.根据权利要求3所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,步骤(4)中所述的有机溶剂C为正辛烷、环己烷、石油醚、磺化煤油、四氢呋喃、1,4-二氧六环、正丁醇、正辛醇、异辛醇、癸醇、N,N-二甲基甲酰胺、甲苯或二甲苯其中的一种或一种以上。
8.根据权利要求3所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,步骤(4)中所述的酸为醋酸、硫酸、盐酸中的一种或一种以上。
9.根据权利要求3所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,步骤(4)中所述的引发剂为叔丁基过氧化氢、异丙苯过氧化氢、过氧化二叔丁基、过氧化二异丙苯、过氧化二苯甲酰、过氧化十二酰、过氧化叔戊酸叔丁酯、过氧化苯甲酸叔丁酯、过氧化二碳酸二异丙酯、过氧化二碳酸二乙基己酯、过氧化二碳酸二环己酯、过氧化二碳酸二苯氧乙酯、偶氮二异丁腈中的一种或一种以上。
10.根据权利要求3所述的一种修饰二烷基次膦酸官能团的氯球改性方法,其特征在于,步骤(4)中所述的表面接枝碳碳双键的氯球与单烷基次膦酸的质量比为5:1~1:5,单烷基次膦酸与酸的摩尔比为5:1~1:2,引发剂质量为单烷基次膦酸质量的0.1~25%。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110774839.0A CN113527552B (zh) | 2021-07-08 | 2021-07-08 | 一种修饰二烷基次膦酸官能团的氯球改性方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110774839.0A CN113527552B (zh) | 2021-07-08 | 2021-07-08 | 一种修饰二烷基次膦酸官能团的氯球改性方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113527552A CN113527552A (zh) | 2021-10-22 |
| CN113527552B true CN113527552B (zh) | 2022-04-05 |
Family
ID=78098091
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110774839.0A Active CN113527552B (zh) | 2021-07-08 | 2021-07-08 | 一种修饰二烷基次膦酸官能团的氯球改性方法 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113527552B (zh) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115160457B (zh) * | 2022-06-01 | 2023-12-12 | 北京科技大学 | 一种二烷基次膦酸官能团改性的萃淋树脂的制备方法 |
| CN118126218B (zh) * | 2024-03-11 | 2024-08-20 | 江苏海洋大学 | 一种多功能键合型萃淋树脂及其制备方法 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0461822A2 (en) * | 1990-06-07 | 1991-12-18 | Korea Research Institute Of Chemical Technology | Phosphoric acid-type chelate resin as uranyl ion adsorbent |
| JPH04239006A (ja) * | 1990-06-07 | 1992-08-26 | Korea Res Inst Of Chem Technol | ウランイオン吸着用燐酸系キレート樹脂の製造方法 |
| JPH05323097A (ja) * | 1991-04-18 | 1993-12-07 | Shiro Matsumoto | 超ウラン元素除去用吸着剤およびその製法 |
| US5457163A (en) * | 1990-06-07 | 1995-10-10 | Korea Research Institute Of Chemical Technology | Phosphonic acid chelating resins for uranyl ion adsorption |
| RU2011144755A (ru) * | 2011-11-03 | 2013-05-10 | Учреждение Российской академии наук Институт химии твердого тела Уральского отделения РАН | Твердый экстрагент для извлечения скандия и способ его получения |
| CN110741102A (zh) * | 2017-07-07 | 2020-01-31 | 轴子稀土和贵金属股份有限公司 | 用于萃取钪的固体萃取剂及其生产方法 |
-
2021
- 2021-07-08 CN CN202110774839.0A patent/CN113527552B/zh active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0461822A2 (en) * | 1990-06-07 | 1991-12-18 | Korea Research Institute Of Chemical Technology | Phosphoric acid-type chelate resin as uranyl ion adsorbent |
| JPH04239006A (ja) * | 1990-06-07 | 1992-08-26 | Korea Res Inst Of Chem Technol | ウランイオン吸着用燐酸系キレート樹脂の製造方法 |
| US5457163A (en) * | 1990-06-07 | 1995-10-10 | Korea Research Institute Of Chemical Technology | Phosphonic acid chelating resins for uranyl ion adsorption |
| JPH05323097A (ja) * | 1991-04-18 | 1993-12-07 | Shiro Matsumoto | 超ウラン元素除去用吸着剤およびその製法 |
| RU2011144755A (ru) * | 2011-11-03 | 2013-05-10 | Учреждение Российской академии наук Институт химии твердого тела Уральского отделения РАН | Твердый экстрагент для извлечения скандия и способ его получения |
| CN110741102A (zh) * | 2017-07-07 | 2020-01-31 | 轴子稀土和贵金属股份有限公司 | 用于萃取钪的固体萃取剂及其生产方法 |
Non-Patent Citations (2)
| Title |
|---|
| METAL ION SELECTIVITY OF MACRORETICULAR CHELATING CATION EXCHANGE RESINS WITH PHOSPHONIC ACID GROUPS ATTACHED TO PHENYL GROUPS OF A STYRENE-DIVINYLBENZENE COPOLYMER MATRIX;Kazunori Yamabe等;《Separation Science and Technology》;20070215;第3511–3528页 * |
| Thermogravimetric and Spectrometric Characterizations of Poly(Styrene-co-Divinylbenzene)Containing Phosphinic and Phosphonic Acid Groups;Luiz Claudio de Santa Maria等;《POLYMER ENGINEERING AND SCIENCE》;20081231;第1897-1900页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113527552A (zh) | 2021-10-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113527552B (zh) | 一种修饰二烷基次膦酸官能团的氯球改性方法 | |
| KR102855811B1 (ko) | 카르복실산계 화합물, 그의 제조방법 및 응용 | |
| CN103789547B (zh) | 一种离子液体萃淋树脂及其制备和应用方法 | |
| CN106498184A (zh) | 一种锂的萃取体系 | |
| CN101708475A (zh) | 磁性苯乙烯系强碱阴离子交换微球树脂及其制备方法 | |
| CN113214480A (zh) | 一种阳离子型框架材料的合成方法及吸附应用 | |
| CN111068630A (zh) | 一种用于去除强酸性废水中重金属阳离子的吡啶胺类螯合树脂及其制备方法 | |
| Sharaf et al. | A novel binary-extractant-impregnated resin for selective recovery of scandium | |
| CN113736995A (zh) | 一种羧酸类化合物作为萃取剂的应用和金属离子萃取方法 | |
| WO2016090809A1 (zh) | 含氨基中性膦萃取剂用于萃取分离四价铈的用途和方法 | |
| CN114672650A (zh) | 一种利用双膦酸萃取剂从钛白废酸中提取钪的方法 | |
| CN112457188A (zh) | 一种羧酸类化合物及其制备方法与应用 | |
| CN109174060B (zh) | 一种含膦酸基螯合树脂及在高酸度下分离与富集钍的方法 | |
| CN113546610B (zh) | 一种在硅基材料表面接枝二烷基次膦酸官能团的方法 | |
| CN110813255B (zh) | 一种双功能化聚合物螯合树脂制备及分离与富集铀的方法 | |
| CN113522243B (zh) | 一种有机次膦酸官能团修饰的硅基吸附材料的制备方法 | |
| KR101586275B1 (ko) | 금속이온의 선택적 흡착을 위한 덴드리머형 하이퍼 브랜치 고분자, 이를 포함하는 흡착제 및 그 제조 방법 | |
| CN101402033A (zh) | 一种螯合型吸附材料及其制备方法 | |
| CN106582569A (zh) | 一种离子印迹化浸渍树脂及其制备方法 | |
| CN111254296A (zh) | 一种具有苯乙烯基膦酸双酯结构的铀萃取剂及其应用 | |
| CN115554988A (zh) | 一种有机链修饰锆基mof吸附剂及其制备方法与应用 | |
| CN111589420A (zh) | 一种毫米级大孔-介孔碳球/杯[4]-双冠-6互穿复合材料及其制备方法和应用 | |
| CN115160457B (zh) | 一种二烷基次膦酸官能团改性的萃淋树脂的制备方法 | |
| JP2015181993A (ja) | 金属吸着剤、水処理用タンク及び水処理システム | |
| JP7558513B2 (ja) | スカンジウムの抽出剤及び分離抽出方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |