WO2023282346A1 - 析出抑制剤 - Google Patents
析出抑制剤 Download PDFInfo
- Publication number
- WO2023282346A1 WO2023282346A1 PCT/JP2022/027105 JP2022027105W WO2023282346A1 WO 2023282346 A1 WO2023282346 A1 WO 2023282346A1 JP 2022027105 W JP2022027105 W JP 2022027105W WO 2023282346 A1 WO2023282346 A1 WO 2023282346A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sugar
- antisense oligomer
- pharmaceutical composition
- precipitation
- antisense
- Prior art date
Links
- 238000001556 precipitation Methods 0.000 title claims abstract description 93
- 239000003112 inhibitor Substances 0.000 title claims abstract description 76
- 230000000692 anti-sense effect Effects 0.000 claims abstract description 173
- 235000000346 sugar Nutrition 0.000 claims abstract description 82
- 210000002700 urine Anatomy 0.000 claims abstract description 56
- 238000000034 method Methods 0.000 claims abstract description 28
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 claims abstract description 19
- 239000008103 glucose Substances 0.000 claims abstract description 19
- 239000008194 pharmaceutical composition Substances 0.000 claims description 87
- 230000008021 deposition Effects 0.000 claims description 49
- 229930006000 Sucrose Natural products 0.000 claims description 39
- 239000005720 sucrose Substances 0.000 claims description 39
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical compound N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 claims description 18
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 claims description 13
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 claims description 13
- 239000003795 chemical substances by application Substances 0.000 claims description 13
- 125000004573 morpholin-4-yl group Chemical group N1(CCOCC1)* 0.000 claims description 11
- UYTPUPDQBNUYGX-UHFFFAOYSA-N guanine Chemical class O=C1NC(N)=NC2=C1N=CN2 UYTPUPDQBNUYGX-UHFFFAOYSA-N 0.000 claims description 7
- 239000000126 substance Substances 0.000 claims description 7
- 125000000600 disaccharide group Chemical group 0.000 claims description 3
- 125000000185 sucrose group Chemical group 0.000 claims description 3
- 125000000647 trehalose group Chemical group 0.000 claims description 3
- 239000000203 mixture Substances 0.000 abstract description 17
- 230000002401 inhibitory effect Effects 0.000 abstract description 9
- 239000003814 drug Substances 0.000 abstract description 8
- 229940079593 drug Drugs 0.000 abstract description 5
- 125000002791 glucosyl group Chemical group C1([C@H](O)[C@@H](O)[C@H](O)[C@H](O1)CO)* 0.000 abstract 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 36
- 239000000243 solution Substances 0.000 description 31
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 27
- 239000002585 base Substances 0.000 description 27
- -1 infusions Substances 0.000 description 27
- 238000002835 absorbance Methods 0.000 description 22
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 18
- 238000011481 absorbance measurement Methods 0.000 description 15
- 235000010355 mannitol Nutrition 0.000 description 15
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 14
- 210000003734 kidney Anatomy 0.000 description 14
- 230000037396 body weight Effects 0.000 description 13
- 230000002485 urinary effect Effects 0.000 description 13
- 108010051109 Cell-Penetrating Peptides Proteins 0.000 description 12
- 102000020313 Cell-Penetrating Peptides Human genes 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- 241000282567 Macaca fascicularis Species 0.000 description 11
- 239000002904 solvent Substances 0.000 description 11
- 239000008215 water for injection Substances 0.000 description 11
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 10
- 238000011156 evaluation Methods 0.000 description 10
- 108090000765 processed proteins & peptides Proteins 0.000 description 10
- 241000699670 Mus sp. Species 0.000 description 9
- 238000009472 formulation Methods 0.000 description 9
- 239000007788 liquid Substances 0.000 description 9
- 239000011780 sodium chloride Substances 0.000 description 9
- 125000000217 alkyl group Chemical group 0.000 description 8
- 239000007864 aqueous solution Substances 0.000 description 8
- 150000001720 carbohydrates Chemical class 0.000 description 8
- 125000003729 nucleotide group Chemical group 0.000 description 8
- 102000004196 processed proteins & peptides Human genes 0.000 description 8
- 238000012986 modification Methods 0.000 description 7
- 230000004048 modification Effects 0.000 description 7
- 108020004707 nucleic acids Proteins 0.000 description 7
- 150000007523 nucleic acids Chemical class 0.000 description 7
- 102000039446 nucleic acids Human genes 0.000 description 7
- 239000002504 physiological saline solution Substances 0.000 description 7
- 239000001103 potassium chloride Substances 0.000 description 7
- 235000011164 potassium chloride Nutrition 0.000 description 7
- 108091034117 Oligonucleotide Proteins 0.000 description 6
- 239000000654 additive Substances 0.000 description 6
- 125000004432 carbon atom Chemical group C* 0.000 description 6
- 239000002773 nucleotide Substances 0.000 description 6
- 230000036470 plasma concentration Effects 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 150000003839 salts Chemical group 0.000 description 6
- 108091093037 Peptide nucleic acid Proteins 0.000 description 5
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 5
- 125000003545 alkoxy group Chemical group 0.000 description 5
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 5
- 125000003118 aryl group Chemical group 0.000 description 5
- 238000002869 basic local alignment search tool Methods 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 230000005764 inhibitory process Effects 0.000 description 5
- 125000005647 linker group Chemical group 0.000 description 5
- 230000001629 suppression Effects 0.000 description 5
- WZUVPPKBWHMQCE-UHFFFAOYSA-N Haematoxylin Chemical compound C12=CC(O)=C(O)C=C2CC2(O)C1C1=CC=C(O)C(O)=C1OC2 WZUVPPKBWHMQCE-UHFFFAOYSA-N 0.000 description 4
- 241001465754 Metazoa Species 0.000 description 4
- 206010029155 Nephropathy toxic Diseases 0.000 description 4
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 4
- 230000000996 additive effect Effects 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 238000007490 hematoxylin and eosin (H&E) staining Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 108020004999 messenger RNA Proteins 0.000 description 4
- 230000007694 nephrotoxicity Effects 0.000 description 4
- 231100000417 nephrotoxicity Toxicity 0.000 description 4
- 125000000548 ribosyl group Chemical group C1([C@H](O)[C@H](O)[C@H](O1)CO)* 0.000 description 4
- 150000008163 sugars Chemical class 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 210000005239 tubule Anatomy 0.000 description 4
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- 229930195725 Mannitol Natural products 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 101710087566 Ribitol-5-phosphate transferase FKTN Proteins 0.000 description 3
- 125000002947 alkylene group Chemical group 0.000 description 3
- 125000003435 aroyl group Chemical group 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 125000004093 cyano group Chemical group *C#N 0.000 description 3
- 125000000753 cycloalkyl group Chemical group 0.000 description 3
- 230000003111 delayed effect Effects 0.000 description 3
- 150000002016 disaccharides Chemical class 0.000 description 3
- 239000003937 drug carrier Substances 0.000 description 3
- 229910052736 halogen Inorganic materials 0.000 description 3
- 150000002367 halogens Chemical class 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 239000000594 mannitol Substances 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 239000011259 mixed solution Substances 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- MCYTYTUNNNZWOK-LCLOTLQISA-N penetratin Chemical compound C([C@H](NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)CCCNC(N)=N)[C@@H](C)CC)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(N)=O)C1=CC=CC=C1 MCYTYTUNNNZWOK-LCLOTLQISA-N 0.000 description 3
- 108010043655 penetratin Proteins 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 229910052717 sulfur Inorganic materials 0.000 description 3
- 230000009885 systemic effect Effects 0.000 description 3
- 238000010998 test method Methods 0.000 description 3
- HPZMWTNATZPBIH-UHFFFAOYSA-N 1-methyladenine Chemical compound CN1C=NC2=NC=NC2=C1N HPZMWTNATZPBIH-UHFFFAOYSA-N 0.000 description 2
- FZWGECJQACGGTI-UHFFFAOYSA-N 2-amino-7-methyl-1,7-dihydro-6H-purin-6-one Chemical compound NC1=NC(O)=C2N(C)C=NC2=N1 FZWGECJQACGGTI-UHFFFAOYSA-N 0.000 description 2
- OVONXEQGWXGFJD-UHFFFAOYSA-N 4-sulfanylidene-1h-pyrimidin-2-one Chemical compound SC=1C=CNC(=O)N=1 OVONXEQGWXGFJD-UHFFFAOYSA-N 0.000 description 2
- OIVLITBTBDPEFK-UHFFFAOYSA-N 5,6-dihydrouracil Chemical compound O=C1CCNC(=O)N1 OIVLITBTBDPEFK-UHFFFAOYSA-N 0.000 description 2
- LRFVTYWOQMYALW-UHFFFAOYSA-N 9H-xanthine Chemical compound O=C1NC(=O)NC2=C1NC=N2 LRFVTYWOQMYALW-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 2
- SRBFZHDQGSBBOR-IOVATXLUSA-N D-xylopyranose Chemical compound O[C@@H]1COC(O)[C@H](O)[C@H]1O SRBFZHDQGSBBOR-IOVATXLUSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- 108010058514 Phosphate-Binding Proteins Proteins 0.000 description 2
- 102000006335 Phosphate-Binding Proteins Human genes 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- 108091028664 Ribonucleotide Proteins 0.000 description 2
- ISAKRJDGNUQOIC-UHFFFAOYSA-N Uracil Chemical compound O=C1C=CNC(=O)N1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 0.000 description 2
- 238000009825 accumulation Methods 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 230000002776 aggregation Effects 0.000 description 2
- 238000004220 aggregation Methods 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 239000000074 antisense oligonucleotide Substances 0.000 description 2
- 238000012230 antisense oligonucleotides Methods 0.000 description 2
- PYMYPHUHKUWMLA-UHFFFAOYSA-N arabinose Natural products OCC(O)C(O)C(O)C=O PYMYPHUHKUWMLA-UHFFFAOYSA-N 0.000 description 2
- SESFRYSPDFLNCH-UHFFFAOYSA-N benzyl benzoate Chemical compound C=1C=CC=CC=1C(=O)OCC1=CC=CC=C1 SESFRYSPDFLNCH-UHFFFAOYSA-N 0.000 description 2
- SRBFZHDQGSBBOR-UHFFFAOYSA-N beta-D-Pyranose-Lyxose Natural products OC1COC(O)C(O)C1O SRBFZHDQGSBBOR-UHFFFAOYSA-N 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 210000001124 body fluid Anatomy 0.000 description 2
- 239000010839 body fluid Substances 0.000 description 2
- 125000001589 carboacyl group Chemical group 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 229940099112 cornstarch Drugs 0.000 description 2
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical compound NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 239000005547 deoxyribonucleotide Substances 0.000 description 2
- 125000002637 deoxyribonucleotide group Chemical group 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 229940014259 gelatin Drugs 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- FDGQSTZJBFJUBT-UHFFFAOYSA-N hypoxanthine Chemical compound O=C1NC=NC2=C1NC=N2 FDGQSTZJBFJUBT-UHFFFAOYSA-N 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 238000003780 insertion Methods 0.000 description 2
- 230000037431 insertion Effects 0.000 description 2
- DRAVOWXCEBXPTN-UHFFFAOYSA-N isoguanine Chemical compound NC1=NC(=O)NC2=C1NC=N2 DRAVOWXCEBXPTN-UHFFFAOYSA-N 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 150000002772 monosaccharides Chemical class 0.000 description 2
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 235000019198 oils Nutrition 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 239000002336 ribonucleotide Substances 0.000 description 2
- 125000002652 ribonucleotide group Chemical group 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 150000005846 sugar alcohols Chemical class 0.000 description 2
- 230000008685 targeting Effects 0.000 description 2
- RWQNBRDOKXIBIV-UHFFFAOYSA-N thymine Chemical compound CC1=CNC(=O)NC1=O RWQNBRDOKXIBIV-UHFFFAOYSA-N 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- FYADHXFMURLYQI-UHFFFAOYSA-N 1,2,4-triazine Chemical compound C1=CN=NC=N1 FYADHXFMURLYQI-UHFFFAOYSA-N 0.000 description 1
- SATCOUWSAZBIJO-UHFFFAOYSA-N 1-methyladenine Natural products N=C1N(C)C=NC2=C1NC=N2 SATCOUWSAZBIJO-UHFFFAOYSA-N 0.000 description 1
- KIQMCGMHGVXDFW-UHFFFAOYSA-N 1-methylhypoxanthine Chemical compound O=C1N(C)C=NC2=C1NC=N2 KIQMCGMHGVXDFW-UHFFFAOYSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- QIJIUJYANDSEKG-UHFFFAOYSA-N 2,4,4-trimethylpentan-2-amine Chemical class CC(C)(C)CC(C)(C)N QIJIUJYANDSEKG-UHFFFAOYSA-N 0.000 description 1
- HLYBTPMYFWWNJN-UHFFFAOYSA-N 2-(2,4-dioxo-1h-pyrimidin-5-yl)-2-hydroxyacetic acid Chemical compound OC(=O)C(O)C1=CNC(=O)NC1=O HLYBTPMYFWWNJN-UHFFFAOYSA-N 0.000 description 1
- SGAKLDIYNFXTCK-UHFFFAOYSA-N 2-[(2,4-dioxo-1h-pyrimidin-5-yl)methylamino]acetic acid Chemical compound OC(=O)CNCC1=CNC(=O)NC1=O SGAKLDIYNFXTCK-UHFFFAOYSA-N 0.000 description 1
- YSAJFXWTVFGPAX-UHFFFAOYSA-N 2-[(2,4-dioxo-1h-pyrimidin-5-yl)oxy]acetic acid Chemical compound OC(=O)COC1=CNC(=O)NC1=O YSAJFXWTVFGPAX-UHFFFAOYSA-N 0.000 description 1
- MWBWWFOAEOYUST-UHFFFAOYSA-N 2-aminopurine Chemical compound NC1=NC=C2N=CNC2=N1 MWBWWFOAEOYUST-UHFFFAOYSA-N 0.000 description 1
- ASJSAQIRZKANQN-CRCLSJGQSA-N 2-deoxy-D-ribose Chemical group OC[C@@H](O)[C@@H](O)CC=O ASJSAQIRZKANQN-CRCLSJGQSA-N 0.000 description 1
- XMSMHKMPBNTBOD-UHFFFAOYSA-N 2-dimethylamino-6-hydroxypurine Chemical compound N1C(N(C)C)=NC(=O)C2=C1N=CN2 XMSMHKMPBNTBOD-UHFFFAOYSA-N 0.000 description 1
- SMADWRYCYBUIKH-UHFFFAOYSA-N 2-methyl-7h-purin-6-amine Chemical compound CC1=NC(N)=C2NC=NC2=N1 SMADWRYCYBUIKH-UHFFFAOYSA-N 0.000 description 1
- KOLPWZCZXAMXKS-UHFFFAOYSA-N 3-methylcytosine Chemical compound CN1C(N)=CC=NC1=O KOLPWZCZXAMXKS-UHFFFAOYSA-N 0.000 description 1
- VPLZGVOSFFCKFC-UHFFFAOYSA-N 3-methyluracil Chemical compound CN1C(=O)C=CNC1=O VPLZGVOSFFCKFC-UHFFFAOYSA-N 0.000 description 1
- DBTMGCOVALSLOR-UHFFFAOYSA-N 32-alpha-galactosyl-3-alpha-galactosyl-galactose Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(OC2C(C(CO)OC(O)C2O)O)OC(CO)C1O DBTMGCOVALSLOR-UHFFFAOYSA-N 0.000 description 1
- NCPQROHLJFARLL-UHFFFAOYSA-N 4-(2,5-dioxopyrrol-1-yl)butanoic acid Chemical compound OC(=O)CCCN1C(=O)C=CC1=O NCPQROHLJFARLL-UHFFFAOYSA-N 0.000 description 1
- GJAKJCICANKRFD-UHFFFAOYSA-N 4-acetyl-4-amino-1,3-dihydropyrimidin-2-one Chemical compound CC(=O)C1(N)NC(=O)NC=C1 GJAKJCICANKRFD-UHFFFAOYSA-N 0.000 description 1
- AOZBINSIAHDCQU-UHFFFAOYSA-N 5-(2-oxopropyl)-1h-pyrimidine-2,4-dione Chemical compound CC(=O)CC1=CNC(=O)NC1=O AOZBINSIAHDCQU-UHFFFAOYSA-N 0.000 description 1
- MQJSSLBGAQJNER-UHFFFAOYSA-N 5-(methylaminomethyl)-1h-pyrimidine-2,4-dione Chemical compound CNCC1=CNC(=O)NC1=O MQJSSLBGAQJNER-UHFFFAOYSA-N 0.000 description 1
- WPYRHVXCOQLYLY-UHFFFAOYSA-N 5-[(methoxyamino)methyl]-2-sulfanylidene-1h-pyrimidin-4-one Chemical compound CONCC1=CNC(=S)NC1=O WPYRHVXCOQLYLY-UHFFFAOYSA-N 0.000 description 1
- LQLQRFGHAALLLE-UHFFFAOYSA-N 5-bromouracil Chemical compound BrC1=CNC(=O)NC1=O LQLQRFGHAALLLE-UHFFFAOYSA-N 0.000 description 1
- RHIULBJJKFDJPR-UHFFFAOYSA-N 5-ethyl-1h-pyrimidine-2,4-dione Chemical compound CCC1=CNC(=O)NC1=O RHIULBJJKFDJPR-UHFFFAOYSA-N 0.000 description 1
- KELXHQACBIUYSE-UHFFFAOYSA-N 5-methoxy-1h-pyrimidine-2,4-dione Chemical compound COC1=CNC(=O)NC1=O KELXHQACBIUYSE-UHFFFAOYSA-N 0.000 description 1
- ZLAQATDNGLKIEV-UHFFFAOYSA-N 5-methyl-2-sulfanylidene-1h-pyrimidin-4-one Chemical compound CC1=CNC(=S)NC1=O ZLAQATDNGLKIEV-UHFFFAOYSA-N 0.000 description 1
- LRSASMSXMSNRBT-UHFFFAOYSA-N 5-methylcytosine Chemical compound CC1=CNC(=O)N=C1N LRSASMSXMSNRBT-UHFFFAOYSA-N 0.000 description 1
- HDRIVHFYDVZHBY-UHFFFAOYSA-N 6-(methylaminomethyl)-4-oxo-2-sulfanylidene-1H-pyrimidine-5-carboxylic acid Chemical compound C(=O)(O)C=1C(NC(NC=1CNC)=S)=O HDRIVHFYDVZHBY-UHFFFAOYSA-N 0.000 description 1
- DCPSTSVLRXOYGS-UHFFFAOYSA-N 6-amino-1h-pyrimidine-2-thione Chemical compound NC1=CC=NC(S)=N1 DCPSTSVLRXOYGS-UHFFFAOYSA-N 0.000 description 1
- CKOMXBHMKXXTNW-UHFFFAOYSA-N 6-methyladenine Chemical compound CNC1=NC=NC2=C1N=CN2 CKOMXBHMKXXTNW-UHFFFAOYSA-N 0.000 description 1
- SHVCSCWHWMSGTE-UHFFFAOYSA-N 6-methyluracil Chemical compound CC1=CC(=O)NC(=O)N1 SHVCSCWHWMSGTE-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- MSSXOMSJDRHRMC-UHFFFAOYSA-N 9H-purine-2,6-diamine Chemical compound NC1=NC(N)=C2NC=NC2=N1 MSSXOMSJDRHRMC-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 240000005020 Acaciella glauca Species 0.000 description 1
- 229930024421 Adenine Natural products 0.000 description 1
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 1
- 108020000948 Antisense Oligonucleotides Proteins 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- 238000012935 Averaging Methods 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 241000167854 Bourreria succulenta Species 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- RXVWSYJTUUKTEA-UHFFFAOYSA-N D-maltotriose Natural products OC1C(O)C(OC(C(O)CO)C(O)C(O)C=O)OC(CO)C1OC1C(O)C(O)C(O)C(CO)O1 RXVWSYJTUUKTEA-UHFFFAOYSA-N 0.000 description 1
- QWIZNVHXZXRPDR-UHFFFAOYSA-N D-melezitose Natural products O1C(CO)C(O)C(O)C(O)C1OC1C(O)C(CO)OC1(CO)OC1OC(CO)C(O)C(O)C1O QWIZNVHXZXRPDR-UHFFFAOYSA-N 0.000 description 1
- HMFHBZSHGGEWLO-SOOFDHNKSA-N D-ribofuranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H]1O HMFHBZSHGGEWLO-SOOFDHNKSA-N 0.000 description 1
- BWLUMTFWVZZZND-UHFFFAOYSA-N Dibenzylamine Chemical class C=1C=CC=CC=1CNCC1=CC=CC=C1 BWLUMTFWVZZZND-UHFFFAOYSA-N 0.000 description 1
- XBPCUCUWBYBCDP-UHFFFAOYSA-N Dicyclohexylamine Chemical class C1CCCCC1NC1CCCCC1 XBPCUCUWBYBCDP-UHFFFAOYSA-N 0.000 description 1
- 239000004131 EU approved raising agent Substances 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical class NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- 102100028417 Fibroblast growth factor 12 Human genes 0.000 description 1
- 102100028072 Fibroblast growth factor 4 Human genes 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical class F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 101000917234 Homo sapiens Fibroblast growth factor 12 Proteins 0.000 description 1
- 101001060274 Homo sapiens Fibroblast growth factor 4 Proteins 0.000 description 1
- 102000008100 Human Serum Albumin Human genes 0.000 description 1
- 108091006905 Human Serum Albumin Proteins 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical class Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- UGQMRVRMYYASKQ-UHFFFAOYSA-N Hypoxanthine nucleoside Natural products OC1C(O)C(CO)OC1N1C(NC=NC2=O)=C2N=C1 UGQMRVRMYYASKQ-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- AYRXSINWFIIFAE-SCLMCMATSA-N Isomaltose Natural products OC[C@H]1O[C@H](OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O)[C@@H](O)[C@@H](O)[C@@H]1O AYRXSINWFIIFAE-SCLMCMATSA-N 0.000 description 1
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical class NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical class NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 244000246386 Mentha pulegium Species 0.000 description 1
- 235000016257 Mentha pulegium Nutrition 0.000 description 1
- 235000004357 Mentha x piperita Nutrition 0.000 description 1
- SGSSKEDGVONRGC-UHFFFAOYSA-N N(2)-methylguanine Chemical compound O=C1NC(NC)=NC2=C1N=CN2 SGSSKEDGVONRGC-UHFFFAOYSA-N 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical class CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- 108091028043 Nucleic acid sequence Proteins 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 101710126211 POU domain, class 5, transcription factor 1 Proteins 0.000 description 1
- 101710112083 Para-Rep C1 Proteins 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 241000288906 Primates Species 0.000 description 1
- HCBIBCJNVBAKAB-UHFFFAOYSA-N Procaine hydrochloride Chemical compound Cl.CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 HCBIBCJNVBAKAB-UHFFFAOYSA-N 0.000 description 1
- MUPFEKGTMRGPLJ-RMMQSMQOSA-N Raffinose Natural products O(C[C@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@@H](O[C@@]2(CO)[C@H](O)[C@@H](O)[C@@H](CO)O2)O1)[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 MUPFEKGTMRGPLJ-RMMQSMQOSA-N 0.000 description 1
- 102100025639 Sortilin-related receptor Human genes 0.000 description 1
- 101710126735 Sortilin-related receptor Proteins 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 101710192266 Tegument protein VP22 Proteins 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical class CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 1
- MUPFEKGTMRGPLJ-UHFFFAOYSA-N UNPD196149 Natural products OC1C(O)C(CO)OC1(CO)OC1C(O)C(O)C(O)C(COC2C(C(O)C(O)C(CO)O2)O)O1 MUPFEKGTMRGPLJ-UHFFFAOYSA-N 0.000 description 1
- 201000006793 Walker-Warburg syndrome Diseases 0.000 description 1
- 230000001594 aberrant effect Effects 0.000 description 1
- 230000009102 absorption Effects 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 150000001242 acetic acid derivatives Chemical class 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 125000002252 acyl group Chemical group 0.000 description 1
- 229960000643 adenine Drugs 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 125000005600 alkyl phosphonate group Chemical group 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- SRBFZHDQGSBBOR-STGXQOJASA-N alpha-D-lyxopyranose Chemical compound O[C@@H]1CO[C@H](O)[C@@H](O)[C@H]1O SRBFZHDQGSBBOR-STGXQOJASA-N 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- PYMYPHUHKUWMLA-WDCZJNDASA-N arabinose Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)C=O PYMYPHUHKUWMLA-WDCZJNDASA-N 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 150000001483 arginine derivatives Chemical class 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-L aspartate group Chemical class N[C@@H](CC(=O)[O-])C(=O)[O-] CKLJMWTZIZZHCS-REOHCLBHSA-L 0.000 description 1
- 210000003651 basophil Anatomy 0.000 description 1
- UPABQMWFWCMOFV-UHFFFAOYSA-N benethamine Chemical class C=1C=CC=CC=1CNCCC1=CC=CC=C1 UPABQMWFWCMOFV-UHFFFAOYSA-N 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- JUHORIMYRDESRB-UHFFFAOYSA-N benzathine Chemical class C=1C=CC=CC=1CNCCNCC1=CC=CC=C1 JUHORIMYRDESRB-UHFFFAOYSA-N 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical class OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 229960002903 benzyl benzoate Drugs 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000013626 chemical specie Substances 0.000 description 1
- 235000019693 cherries Nutrition 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- VDANGULDQQJODZ-UHFFFAOYSA-N chloroprocaine Chemical class CCN(CC)CCOC(=O)C1=CC=C(N)C=C1Cl VDANGULDQQJODZ-UHFFFAOYSA-N 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 150000001868 cobalt Chemical class 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 150000001879 copper Chemical class 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 229940104302 cytosine Drugs 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical class OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 150000005332 diethylamines Chemical class 0.000 description 1
- NAGJZTKCGNOGPW-UHFFFAOYSA-K dioxido-sulfanylidene-sulfido-$l^{5}-phosphane Chemical compound [O-]P([O-])([S-])=S NAGJZTKCGNOGPW-UHFFFAOYSA-K 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 238000001425 electrospray ionisation time-of-flight mass spectrometry Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-N ethanesulfonic acid Chemical class CCS(O)(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000010855 food raising agent Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-L fumarate(2-) Chemical class [O-]C(=O)\C=C\C([O-])=O VZCYOOQTPOCHFL-OWOJBTEDSA-L 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 150000002301 glucosamine derivatives Chemical class 0.000 description 1
- 150000002306 glutamic acid derivatives Chemical class 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 150000002357 guanidines Chemical class 0.000 description 1
- 125000003104 hexanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 231100000171 higher toxicity Toxicity 0.000 description 1
- 235000001050 hortel pimenta Nutrition 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 1
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 159000000014 iron salts Chemical class 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- DLRVVLDZNNYCBX-RTPHMHGBSA-N isomaltose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)C(O)O1 DLRVVLDZNNYCBX-RTPHMHGBSA-N 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000000644 isotonic solution Substances 0.000 description 1
- 230000003907 kidney function Effects 0.000 description 1
- JCQLYHFGKNRPGE-FCVZTGTOSA-N lactulose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 JCQLYHFGKNRPGE-FCVZTGTOSA-N 0.000 description 1
- 229960000511 lactulose Drugs 0.000 description 1
- PFCRQPBOOFTZGQ-UHFFFAOYSA-N lactulose keto form Natural products OCC(=O)C(O)C(C(O)CO)OC1OC(CO)C(O)C(O)C1O PFCRQPBOOFTZGQ-UHFFFAOYSA-N 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 159000000003 magnesium salts Chemical class 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229940049920 malate Drugs 0.000 description 1
- 150000002688 maleic acid derivatives Chemical class 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 210000004962 mammalian cell Anatomy 0.000 description 1
- FYGDTMLNYKFZSV-UHFFFAOYSA-N mannotriose Natural products OC1C(O)C(O)C(CO)OC1OC1C(CO)OC(OC2C(OC(O)C(O)C2O)CO)C(O)C1O FYGDTMLNYKFZSV-UHFFFAOYSA-N 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- QWIZNVHXZXRPDR-WSCXOGSTSA-N melezitose Chemical compound O([C@@]1(O[C@@H]([C@H]([C@@H]1O[C@@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)O)CO)CO)[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O QWIZNVHXZXRPDR-WSCXOGSTSA-N 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-M methanesulfonate group Chemical class CS(=O)(=O)[O-] AFVFQIVMOAPDHO-UHFFFAOYSA-M 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 150000002780 morpholines Chemical class 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- XJVXMWNLQRTRGH-UHFFFAOYSA-N n-(3-methylbut-3-enyl)-2-methylsulfanyl-7h-purin-6-amine Chemical compound CSC1=NC(NCCC(C)=C)=C2NC=NC2=N1 XJVXMWNLQRTRGH-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001038 naphthoyl group Chemical group C1(=CC=CC2=CC=CC=C12)C(=O)* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 150000002815 nickel Chemical class 0.000 description 1
- 150000002823 nitrates Chemical class 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 150000003891 oxalate salts Chemical class 0.000 description 1
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical class OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 229940021222 peritoneal dialysis isotonic solution Drugs 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 150000004713 phosphodiesters Chemical class 0.000 description 1
- PTMHPRAIXMAOOB-UHFFFAOYSA-L phosphoramidate Chemical compound NP([O-])([O-])=O PTMHPRAIXMAOOB-UHFFFAOYSA-L 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 150000004885 piperazines Chemical class 0.000 description 1
- 229920000724 poly(L-arginine) polymer Polymers 0.000 description 1
- 108010011110 polyarginine Proteins 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical class CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960001309 procaine hydrochloride Drugs 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- MUPFEKGTMRGPLJ-ZQSKZDJDSA-N raffinose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO[C@@H]2[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO)O2)O)O1 MUPFEKGTMRGPLJ-ZQSKZDJDSA-N 0.000 description 1
- 230000009103 reabsorption Effects 0.000 description 1
- 235000003499 redwood Nutrition 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000007974 sodium acetate buffer Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 150000003890 succinate salts Chemical class 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 150000003892 tartrate salts Chemical class 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical class C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- RYYWUUFWQRZTIU-UHFFFAOYSA-K thiophosphate Chemical compound [O-]P([O-])([O-])=S RYYWUUFWQRZTIU-UHFFFAOYSA-K 0.000 description 1
- 229940113082 thymine Drugs 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-M toluenesulfonate group Chemical group C=1(C(=CC=CC1)S(=O)(=O)[O-])C LBLYYCQCTBFVLH-UHFFFAOYSA-M 0.000 description 1
- 125000005425 toluyl group Chemical group 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000041 toxicology testing Toxicity 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- PBKWZFANFUTEPS-CWUSWOHSSA-N transportan Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(C)C)C(N)=O)[C@@H](C)CC)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)CN)[C@@H](C)O)C1=CC=C(O)C=C1 PBKWZFANFUTEPS-CWUSWOHSSA-N 0.000 description 1
- 108010062760 transportan Proteins 0.000 description 1
- 125000002827 triflate group Chemical class FC(S(=O)(=O)O*)(F)F 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical class OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 150000004043 trisaccharides Chemical class 0.000 description 1
- 229940035893 uracil Drugs 0.000 description 1
- 125000003774 valeryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 229940075420 xanthine Drugs 0.000 description 1
- 150000003751 zinc Chemical class 0.000 description 1
- FYGDTMLNYKFZSV-BYLHFPJWSA-N β-1,4-galactotrioside Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@H](CO)O[C@@H](O[C@@H]2[C@@H](O[C@@H](O)[C@H](O)[C@H]2O)CO)[C@H](O)[C@H]1O FYGDTMLNYKFZSV-BYLHFPJWSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/047—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates having two or more hydroxy groups, e.g. sorbitol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7016—Disaccharides, e.g. lactose, lactulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/7105—Natural ribonucleic acids, i.e. containing only riboses attached to adenine, guanine, cytosine or uracil and having 3'-5' phosphodiester links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/712—Nucleic acids or oligonucleotides having modified sugars, i.e. other than ribose or 2'-deoxyribose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/7125—Nucleic acids or oligonucleotides having modified internucleoside linkage, i.e. other than 3'-5' phosphodiesters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/02—Drugs for disorders of the urinary system of urine or of the urinary tract, e.g. urine acidifiers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/111—General methods applicable to biologically active non-coding nucleic acids
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/11—Antisense
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/314—Phosphoramidates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/323—Chemical structure of the sugar modified ring structure
- C12N2310/3233—Morpholino-type ring
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/50—Methods for regulating/modulating their activity
- C12N2320/53—Methods for regulating/modulating their activity reducing unwanted side-effects
Definitions
- the present invention provides an agent for suppressing precipitation of antisense oligomers in the urine of a subject to whom a pharmaceutical composition containing an antisense oligomer containing sugar is administered, and an antisense oligomer in the urine of a subject to whom the pharmaceutical composition is administered. and a pharmaceutical composition containing the precipitation inhibitor.
- Antisense oligomers are nucleic acids that sequence-specifically hybridize to target mRNAs and pre-mRNAs. Antisense oligomers exert their effects through degradation of mRNA and pre-mRNA, exon skipping, exon inclusion, translation inhibition, etc., and are used as therapeutic agents for several diseases.
- Patent Document 1 discloses an antisense nucleic acid capable of treating Fukuyama muscular dystrophy or the like by normalizing aberrant splicing of fukutin gene having an insertion mutation of SVA-type retrotransposon.
- Non-Patent Document 1 morpholino oligomers accumulate in the kidney (Non-Patent Document 1, Figure 2)
- nephrotoxicity occurs due to administration of morpholino oligomers
- Non-Patent Document 2 Figure 3, Table 5
- the present invention provides an agent for suppressing deposition of antisense oligomers in urine in a subject to whom a pharmaceutical composition containing an antisense oligomer containing a sugar other than glucose described below was administered, and the pharmaceutical composition to which the pharmaceutical composition was administered.
- the precipitation inhibitor according to (1) which is used in an amount such that the sugar concentration in the pharmaceutical composition is 2.6 mg/mL to 1694.1 mg/mL.
- the deposition inhibitor according to any one of (1) to (6), wherein the sugar is trehalose.
- (15-1) A method for suppressing precipitation of an antisense oligomer in the urine of a subject to whom a pharmaceutical composition containing an antisense oligomer is administered, comprising adding a sugar other than glucose to the pharmaceutical composition.
- a method comprising adding sugar in an amount that results in a concentration of 0.5 mg/mL to 3000 mg/mL.
- (15-2) The method according to (15-1), wherein the sugar is added in an amount such that the sugar concentration in the pharmaceutical composition is 2.6 mg/mL to 1694.1 mg/mL.
- (15-3) The method according to (15-1) or (15-2), wherein the concentration of the antisense oligomer in the pharmaceutical composition is 0.05 mg/mL to 2000 mg/mL.
- (16-1) A method for suppressing precipitation of an antisense oligomer in urine in a subject to whom an antisense oligomer has been administered, comprising administering a sugar other than glucose to the subject, is administered in an amount such that the weight ratio of sugar to 1 is 0.1 to 100.
- (16-2) The method according to (16-1), wherein the antisense oligomer and the saccharide are administered separately.
- (16-3) The method according to (16-1) or (16-2), wherein the sugar is used in such an amount that the weight ratio of the sugar to the weight of the antisense oligomer is 1-53.
- (17-1) A pharmaceutical composition containing an antisense oligomer, wherein the pharmaceutical composition contains a sugar other than glucose, and an antisense oligomer precipitation inhibitor in the urine of a subject to whom the pharmaceutical composition is administered is 0.5 mg/mL to A pharmaceutical composition comprising a concentration of 3000 mg/mL.
- an agent for suppressing the precipitation of antisense oligomers in urine in a subject to whom a pharmaceutical composition containing an antisense oligomer is administered, and the precipitation of antisense oligomers in urine in a subject to which the pharmaceutical composition is administered is provided.
- Absorbance measurements at 620 nm under the conditions shown in Table 4 are shown. Absorbance measurements at 620 nm under the conditions shown in Table 5 are shown. Absorbance measurements at 620 nm under the conditions shown in Table 6 are shown. Absorbance measurements at 620 nm under the conditions shown in Table 7 are shown. Absorbance measurements at 620 nm under the conditions shown in Table 8 are shown. Absorbance measurements at 620 nm under the conditions shown in Table 9 are shown. Absorbance measurements at 620 nm under the conditions shown in Table 10 are shown. Absorbance measurements at 620 nm under the conditions shown in Table 11 are shown. PMO No.
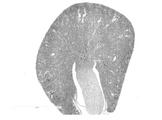
- PMO No. 8 shows hematoxylin and eosin staining images of kidneys when 10 mL/kg of 8 administration solution (50 mg/mL PMO No. 8) was administered.
- PMO No. 8 shows hematoxylin and eosin staining images of kidneys when 10 mL/kg of 8 administration solution (50 mg/mL PMO No. 8, 20 mg/mL sucrose) was administered.
- PMO No. 8 shows hematoxylin and eosin staining images of kidneys when 10 mL/kg of 8 administration solution (50 mg/mL PMO No. 8, 50 mg/mL sucrose) was administered.
- the present invention relates to an antisense oligomer deposition inhibitor in the urine of a subject to whom a pharmaceutical composition containing an antisense oligomer has been administered.
- the term “precipitation inhibitor” means an agent for inhibiting the precipitation of antisense oligomers in the urine of a subject to whom a pharmaceutical composition containing antisense oligomers has been administered.
- the inhibition of deposition reduces the accumulation of antisense oligomers in the kidney, it is expected to lead to a reduction in nephrotoxicity.
- Nephrotoxicity means tissue damage or decreased renal function due to increased accumulation of the administered antisense oligomer in the kidney.
- precipitation in body fluids such as blood is suppressed in vivo, it is predicted that the concentration of antisense oligomers in body fluids will become uniform, leading to stabilization of efficacy and/or reduction of toxicity. be.
- the presence or absence of the precipitation inhibitory effect was determined by adding an antisense oligomer and a precipitation inhibitor to a solvent simulating urine, and determining whether the precipitation inhibitor reduced the increase in absorbance due to the precipitation of the antisense oligomer. can be determined by examining the For example, after adding the antisense oligomer and the precipitation inhibitor to the solvent (e.g., after 5 minutes, 10 minutes, 15 minutes, 20 minutes, 25 minutes, 30 minutes, 40 minutes, 50 minutes, After 60 minutes, 70 minutes, 80 minutes, or 90 minutes), the absorbance can be measured at 37°C.
- the time until precipitation is observed is, for example, 5% or more. , 10% or more, 20% or more, 30% or more, 40% or more, or 50% or more, it can be determined that there is a precipitation suppressing effect.
- Solvents simulating urine include, for example, KCl at 20 nmol/mL to 9000 nmol/mL, 50 nmol/mL to 3600 nmol/mL, or 100 nmol/mL to 1800 nmol/mL, and NaCl at 15 nmol/mL to 2400 nmol/mL and 40 nmol/mL.
- Aqueous solutions containing ⁇ 900 nmol/mL, or 77 nmol/mL to 462 nmol/mL can be used.
- the dose of sugar to the subject and the concentration in urine may have linearity, so the concentration of sugar in a solvent that simulates urine (hereinafter also referred to as "urinary concentration").
- the concentration of sugar in a solvent that simulates urine (hereinafter also referred to as "urinary concentration").
- the urine concentration can be converted to a glucose dosage using the following formula, and the glucose dosage can be calculated from body weight and administration Based on the liquid volume, it can be converted to the concentration in the formulation at the time of administration (at the time of use).
- Glucose dose urine concentration (mg/mL)/estimated urine concentration in cynomolgus monkeys (127.5 (mg/mL)) x dose to cynomolgus monkeys (500 (mg/kg))
- D-mannitol concentration in formulation D-mannitol dose (mg/kg) ⁇ body weight (kg) ⁇ administration liquid volume (mL)
- a body weight of, for example, 3 to 80 kg, and an administration volume of, for example, 50 to 200 mL can be assumed.
- the concentration of antisense oligomers in a solvent that simulates urine can be converted to the concentration of antisense oligomers in the formulation as described in the Examples.
- the deposition inhibitor of the present invention may consist of sugars (excluding glucose) or may contain sugars (excluding glucose).
- the sugar may be, for example, a disaccharide or higher sugar, or a disaccharide sugar.
- sugars include disaccharides such as sucrose, lactose, lactulose, trehalose, maltose, isomaltose; trisaccharides such as raffinose, melezitose, maltotriose; galactose, mannose, fructose, ribose; Monosaccharides such as xylose, arabinose, and lyxose are included.
- the sugar is or comprises sucrose.
- the sugar is or comprises trehalose.
- the deposition inhibitor of the present invention contains a component other than sugar
- the deposition inhibitor is formulated by appropriately blending sugar with a pharmaceutically acceptable carrier or additive (and optionally the deposition inhibitor of the present invention).
- a pharmaceutically acceptable carrier or additive may be changed.
- oral agents such as tablets, coated tablets, pills, powders, granules, capsules, solutions, suspensions, and emulsions
- parenteral agents such as injections, infusions, suppositories, ointments, and patches
- Parenteral agents are preferred.
- the injection may be a freeze-dried preparation.
- the blending ratio of the carrier or additive may be appropriately set based on the range normally employed in the pharmaceutical field.
- the carriers or additives that can be incorporated are not particularly limited, but for example, water, physiological saline, other aqueous solvents, various carriers such as aqueous or oily bases, excipients, binders, pH adjusters, disintegrants, absorption Various additives such as accelerators, lubricants, coloring agents, corrigents, and perfumes are included.
- excipients examples include binders such as gelatin, cornstarch, tragacanth and gum arabic, excipients such as crystalline cellulose, cornstarch, gelatin, alginic acid and the like. Raising agents, lubricants such as magnesium stearate, sweetening agents such as sucrose, lactose or saccharin, flavoring agents such as peppermint, redwood oil or cherries, and the like may be used.
- the dosage unit form is a capsule, the above type of material may further contain a liquid carrier such as oil.
- Sterile compositions for injection can be prepared according to routine pharmaceutical practice (eg, dissolving or suspending the active ingredient in a solvent such as water for injection, natural vegetable oil, etc.).
- Aqueous solutions for injection include, for example, physiological saline, isotonic solutions containing glucose and other adjuvants (e.g., sodium chloride), and the like. ), polyalcohols (eg, propylene glycol, polyethylene glycol), nonionic surfactants (eg, polysorbate 80 TM , HCO-50), and the like.
- the oily liquid for example, sesame oil, soybean oil and the like are used, and they may be used in combination with dissolution aids such as benzyl benzoate and benzyl alcohol.
- buffers e.g., phosphate buffer, sodium acetate buffer
- soothing agents e.g., benzalkonium chloride, procaine hydrochloride, etc.
- stabilizers e.g., human serum albumin, polyethylene glycol, etc.
- preservatives agents eg, benzyl alcohol, phenol, etc.
- antioxidants and the like.
- it may be a freeze-dried preparation.
- antisense oligomers may be either oligonucleotides, morpholino oligomers, or peptide nucleic acid (PNA) oligomers (hereinafter each referred to as “antisense oligos described herein. (also referred to as “nucleotides”, “antisense morpholino oligomers as described herein”, or “antisense peptide nucleic acid oligomers as described herein”).
- PNA peptide nucleic acid
- An antisense oligonucleotide is an antisense oligomer whose constituent units are nucleotides, and such nucleotides may be ribonucleotides, deoxyribonucleotides, or modified nucleotides.
- Modified nucleotides refer to those in which all or part of the nucleobases, sugar moieties, and phosphate-binding moieties that make up ribonucleotides or deoxyribonucleotides have been modified.
- Nucleic acid bases include, for example, adenine, guanine, hypoxanthine, cytosine, thymine, uracil, or modified bases thereof.
- modified bases include pseudouracil, 3-methyluracil, dihydrouracil, 5-alkylcytosine (eg, 5-methylcytosine), 5-alkyluracil (eg, 5-ethyluracil), 5-halouracil (eg, , 5-bromouracil), 6-azapyrimidine, 6-alkylpyrimidine (e.g., 6-methyluracil), 2-thiouracil, 4-thiouracil, 4-acetylcytosine, 5-(carboxyhydroxymethyl)uracil, 5-carboxy methylaminomethyl-2-thiouracil, 5-carboxymethylaminomethyluracil, 1-methyladenine, 1-methylhypoxanthine, 2,2-dimethylguanine, 3-methylcytosine, 2-methyladenine, 2-methylguanine, N
- Modifications of the sugar moiety can include, for example, modifications at the 2' position of ribose and modifications at other parts of the sugar.
- Modifications at the 2'-position of ribose include, for example, -OH group at the 2'-position of ribose with -OR, -R, -R'OR, -SH, -SR, -NH 2 , -NHR, -NR 2 , Modifications substituted for -N 3 , -CN, -F, -Cl, -Br, -I can be mentioned.
- R represents alkyl or aryl.
- R' represents alkylene.
- Modifications of other parts of the sugar include, for example, those in which the 4'-position of ribose or deoxyribose is replaced with S, those in which the 2'-position and 4'-position of the sugar are crosslinked, for example, LNA (Locked Nucleic Acid ) or ENA (2′-O,4′-C-Ethylene-bridged Nucleic Acids), but are not limited thereto.
- LNA Locked Nucleic Acid
- ENA (2′-O,4′-C-Ethylene-bridged Nucleic Acids
- Modifications of the phosphate binding moiety include, for example, phosphodiester linkages, phosphorothioate linkages, phosphorodithioate linkages, alkylphosphonate linkages, phosphoramidate linkages, boranophosphate linkages (e.g., Enya et al: Bioorganic & Medicinal Chemistry , 2008, 18, 9154-9160) (see, eg, Republished Patent Publication No. 2006/129594 and Republished Patent Publication No. 2006/038608).
- alkyl is preferably linear or branched alkyl having 1 to 6 carbon atoms. Specific examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, neopentyl, tert-pentyl, n-hexyl and isohexyl. be done.
- the alkyl may be substituted, and examples of such substituents include halogen, alkoxy, cyano, and nitro, and may be substituted with 1 to 3 of these.
- cycloalkyl having 3 to 12 carbon atoms is preferred as cycloalkyl.
- Specific examples include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclodecyl and cyclododecyl.
- halogen includes fluorine, chlorine, bromine, and iodine.
- alkoxy refers to linear or branched alkoxy having 1 to 6 carbon atoms, such as methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, sec-butoxy, tert. -butoxy, n-pentyloxy, isopentyloxy, n-hexyloxy, isohexyloxy and the like.
- Alkoxy having 1 to 3 carbon atoms is particularly preferred.
- aryl having 6 to 10 carbon atoms is preferred as aryl.
- Specific examples include phenyl, ⁇ -naphthyl, and ⁇ -naphthyl. Phenyl is particularly preferred.
- the aryl may be substituted, and examples of such substituents include alkyl, halogen, alkoxy, cyano, and nitro, which may be substituted 1-3 times.
- alkylene is preferably linear or branched alkylene having 1 to 6 carbon atoms.
- Specific examples include methylene, ethylene, trimethylene, tetramethylene, pentamethylene, hexamethylene, 2-(ethyl)trimethylene, and 1-(methyl)tetramethylene.
- acyl includes linear or branched alkanoyl or aroyl.
- Alkanoyl includes, for example, formyl, acetyl, 2-methylacetyl, 2,2-dimethylacetyl, propionyl, butyryl, isobutyryl, pentanoyl, 2,2-dimethylpropionyl, hexanoyl and the like.
- Aroyl includes, for example, benzoyl, toluoyl and naphthoyl. Such aroyl may be optionally substituted at any substitutable position or may be substituted with alkyl.
- the antisense oligonucleotides described herein can be easily synthesized using various automated synthesizers (e.g., AKTA oligopilot plus 10/100 (GE Healthcare)), or can be synthesized by a third party ( For example, it can be produced by entrusting to Promega or Takara) or the like.
- automated synthesizers e.g., AKTA oligopilot plus 10/100 (GE Healthcare)
- a third party For example, it can be produced by entrusting to Promega or Takara
- the antisense morpholino oligomer described in this specification is an antisense oligomer having a group represented by the following general formula as a structural unit.
- Base represents a nucleobase
- W represents a group represented by any one of the following formulas.
- the morpholino oligomer is preferably an oligomer (phosphorodiamidate morpholino oligomer (hereinafter referred to as "PMO")) having a group represented by the following formula as a constituent unit.
- PMO phosphorodiamidate morpholino oligomer
- Morpholino oligomers can be produced, for example, according to the method described in WO 1991/009033 or WO 2009/064471.
- PMO can be produced according to the method described in WO2009/064471 or WO2013/100190.
- the antisense peptide nucleic acid oligomer described in this specification is an antisense oligomer having a group represented by the following general formula as a structural unit.
- Peptide nucleic acid oligomers can be produced, for example, according to the following literature. 1) P. E. Nielsen, M. Egholm, R. H. Berg, O. Buchardt, Science, 254, 1497 (1991) 2) M. Egholm, O. Buchardt, P. E. Nielsen, R. H. Berg, JACS, 114, 1895 (1992) 3) K. L. Dueholm, M. Egholm, C. Behrens, L. Christensen, H. F. Hansen, T. Vulpius, K. H. Petersen, R. H. Berg, P. E. Nielsen, O. Buchardt, J. Org.
- the antisense oligomers described herein may be in a pharmaceutically acceptable salt form, a hydrate form, or a hydrate form of a pharmaceutically acceptable salt thereof.
- Examples of pharmaceutically acceptable salts of the antisense oligomers described herein include alkali metal salts such as sodium, potassium and lithium salts, alkaline earth metal salts such as calcium and magnesium salts. metal salts such as aluminum salts, iron salts, zinc salts, copper salts, nickel salts, cobalt salts; ammonium salts; t-octylamine salts, dibenzylamine salts, morpholine salts, glucosamine salts, phenylglycine alkyl ester salts, ethylenediamine salt, N-methylglucamine salt, guanidine salt, diethylamine salt, triethylamine salt, dicyclohexylamine salt, N,N'-dibenzylethylenediamine salt, chloroprocaine salt, procaine salt, diethanolamine salt, N-benzyl-phenethylamine salt, piperazine salts, organic amine salts such as tetramethylammonium salts
- inorganic acid salts such as nitrates, perchlorates, sulfates and phosphates; lower alkanesulfonates such as methanesulfonates, trifluoromethanesulfonates and ethanesulfonates; benzenesulfonates, p - arylsulfonates such as toluenesulfonates; organic acid salts such as acetates, malate, fumarates, succinates, citrates, tartrates, oxalates, maleates; glycinates. , lysine salts, arginine salts, ornithine salts, glutamates, and amino acid salts such as aspartates.
- these salts can be produced by known methods.
- the antisense oligomers described herein may be in their hydrate form.
- the 5' end of the antisense oligomer described herein may be any group represented by the following chemical formulas (1) to (3). Preferably, it is the group (1) or (2).
- group (1) groups represented by (1), (2), and (3) above are referred to as “group (1),” “group (2),” and “group (3),” respectively.
- the base sequence of the antisense oligomer described herein is not limited, it may contain, for example, four consecutive purine bases in the base sequence. Also, at least two of the four consecutive purine bases may be guanine.
- the antisense oligomers described herein have (i) a base sequence selected from the group consisting of SEQ ID NOs: 1-12, (ii) a base selected from the group consisting of SEQ ID NOs: 1-12 80% or more, 85% or more, 86% or more, 87% or more, 88% or more, 89% or more, 90% or more, 91% or more, 92% or more, 93% or more, 94% or more, 95% or more, 96% or more, 97% or more, 98% or more, or 99% or more sequence identity, or (iii) one or several nucleotide sequences selected from the group consisting of SEQ ID NOS: 1 to 12 contains or consists of a base sequence in which the bases of are added, deleted, or substituted.
- the antisense oligomer described herein is the genome of an insertion mutant fukutin gene in which an SVA-type retrotransposon sequence (GenBank ACCESSION: AB185332) is inserted into the genome sequence of the fukutin gene (GenBank ACCESSION: AB038490).
- the target sequence is the range from position 115937 to position 115981 of the sequence (SEQ ID NO: 13), or this range is not the target sequence.
- Examples of antisense oligomers targeting the range of positions 115937 to 115981 of SEQ ID NO: 13 include antisense oligomers containing a nucleotide sequence selected from the group consisting of SEQ ID NOS: 1-7.
- Examples of antisense oligomers that do not target the range of positions 115937 to 115981 of SEQ ID NO: 13 include antisense oligomers containing a base sequence selected from the group consisting of SEQ ID NOS: 8-12.
- the antisense oligomers described herein are conjugates with attached functional peptides, such as cell penetrating peptides (CPPs).
- functional peptides such as cell penetrating peptides (CPPs).
- Known functional peptides or commercially available functional peptides can be used herein.
- Functional peptides that can be used herein include, for example, arginine-rich peptides disclosed in WO2008/036127; or organs disclosed in WO2009/005793.
- Targeting peptides such as RXR, RBR, etc.; or peptides comprising amino acid subunits disclosed in WO2012/150960 are included.
- CPPs Cell-penetrating peptides
- Known CPPs or commercially available CPPs can be used herein.
- CPPs that can be used herein are, for example, Pharmacology & Therapeutics 154 (2015) 78-86, Table 1 on page 80, for example TAT(48-60), penetratin, polyarginine, Oct4, WT1-pTj, DPV3, transportan, MAP , VP22, Rep1, KW, KFGF, FGF12, Intefrin ⁇ 3 peptide, C105Y, TP2; Paragraph [0085] of Japanese Patent Publication No. 2017-500856 (International Publication No.
- CPPs listed in Table 1 Examples include DPV10/6, DPV15b, YM-3, Tat, LR11, C45D18, Lyp-1, Lyp-2, BMV GAG, hLF1-22, C45D18, LR20 and the like.
- CPPs are available from Funakoshi, Co., for example. , Ltd. It is commercially available from Commercially available CPPs such as TAT (Funakoshi, Co., Ltd.), penetratin (Funakoshi, Co., Ltd.), or known CPPs, such as R8, can be used herein.
- Preferred CPPs that can be used herein include, for example, hLIMK, TAT, penetratin, R8, etc.
- the CPP can be directly attached to the antisense oligomers herein or can be attached via a linker that can attach the CPP to the antisense oligomer.
- linkers can be used herein. Such linkers include, for example, Japanese Patent Publication No. 2017-500856 (International Publication No. 2015/089487), International Publication No. 2015/089487, International Publication No. 2009/073809, International Publication No. 2013/ 075035 pamphlet, International Publication No. 2015/105083 pamphlet, International Publication No.
- linkers that can be used herein include, for example, 4-maleimidobutyric acid, linkers that can bind to the functional peptides or antisense oligomers herein via disulfide bonds, and the like.
- the conjugates herein can be prepared by methods known to those skilled in the art.
- the pharmaceutical composition described herein may be formulated by appropriately blending a pharmaceutically acceptable carrier or additive (and optionally the sugar of the present invention).
- the pharmaceutical composition is the same as described for the deposition inhibitor of the present invention except that the pharmaceutically acceptable carrier or additive and formulation are the antisense nucleic acid as an active ingredient.
- the precipitation inhibitor of the present invention is used in an amount such that the sugar concentration in the pharmaceutical composition described herein is between 0.5 mg/mL and 3000 mg/mL, such as between 2.6 mg/mL and 1694 mg/mL. used and/or added to pharmaceutical compositions.
- the precipitation inhibitor of the present invention has a sugar concentration in the pharmaceutical composition of 0.5 mg/mL or higher, 1 mg/mL or higher, 1.5 mg/mL or higher, 2 mg/mL or higher, 2.5 mg/mL or higher.
- the deposition inhibitor of the present invention can be used, for example, when the sugar concentration in the pharmaceutical composition is 3000 mg/mL or less, 2500 mg/mL or less, 2500 mg/mL or less, 2000 mg/mL or less, 1500 mg/mL or less, or 1000 mg/mL. The following amounts are used and/or added to the pharmaceutical composition:
- the concentration of antisense oligomer in the pharmaceutical composition described herein is 0.05 mg/mL to 2000 mg/mL, such as 0.3 mg/mL to 350 mg/mL. In one embodiment, the concentration of antisense oligomer in the pharmaceutical composition described herein is 0.05 mg/mL or greater, 0.1 mg/mL or greater, 0.2 mg/mL or greater, 0.3 mg/mL or greater, 0.4 mg/mL or greater.
- the present invention provides a deposition inhibitor for a pharmaceutical composition containing an antisense oligomer, which contains a sugar, and the weight ratio of the sugar to the weight of the antisense oligomer is 0.1 to 100, such as 1 to 100. It relates to a deposition inhibitor, used in an amount of 53. Pharmaceutical compositions and saccharides containing antisense oligomers are described above.
- the deposition inhibitor of the present invention has a sugar weight ratio of 0.05 or more, 0.1 or more, 0.15 or more, 0.2 or more, 0.3 or more, 0.4 or more, 0.5 or more, 0.6 or more when the antisense oligomer is 1.
- the deposition inhibitor of the present invention has a sugar weight ratio of 100 or less, 53 or less, 40 or less, 30 or less, 25 or less, 20 or less, 15 or less, or 10 or less, relative to the weight of the antisense oligomer. The following amounts are used.
- the deposition inhibitor of the present invention is contained in a pharmaceutical composition described herein and administered together with the pharmaceutical composition described herein.
- a pharmaceutical composition in which the antisense oligomer described herein is lyophilized optionally together with a carrier such as glucose and dissolved in a solvent such as water for injection is mixed with the precipitation inhibitor described herein. , and then optionally adjusted with a solvent and administered to the subject.
- the antisense oligomer described herein is freeze-dried together with the precipitation inhibitor of the present invention, dissolved in a solvent such as water for injection to form a pharmaceutical composition, and then the amount is arbitrarily adjusted with the solvent. may be administered to a subject.
- the deposition inhibitors of the present invention are not included in the pharmaceutical compositions described herein and are administered separately (simultaneously or sequentially) from the pharmaceutical compositions described herein.
- a pharmaceutical composition prepared by dissolving a lyophilized antisense oligomer described herein in a solvent such as water for injection and the deposition inhibitor described herein may be separately administered to a subject.
- administering the deposition inhibitor and the pharmaceutical composition "simultaneously” means administering the deposition inhibitor and the pharmaceutical composition at the same time.
- administering the deposition inhibitor and the pharmaceutical composition "sequentially” means administering at different times. Specifically, it can be administered before or after the deposition inhibitor, and the interval between administration of the deposition inhibitor and the pharmaceutical composition in this case is not limited, but for example, several minutes, several hours, or up to about a day. It's okay.
- the subject to which the pharmaceutical composition and/or deposition inhibitor is administered is not limited, but examples include mammals such as primates such as humans, laboratory animals such as rats, mice, and rats, pigs, and cows. , horses, and domestic animals such as sheep, preferably humans.
- the dosage for administering the pharmaceutical composition and/or the deposition inhibitor includes the type of sugar contained in the antisense oligomer and the deposition inhibitor contained in the pharmaceutical composition, the dosage form of the pharmaceutical composition and the precipitation inhibitor, and age. and weight of the subject, route of administration, nature and symptoms of the disease.
- the amount of sugar can be, for example, 0.1 mg to 200 g/human, such as 1 mg to 100 g/human, such as in the range of 10 mg to 50 g/person, such as in the range of 100 mg to 50 g/person, such as in the range of 100 mg to 40 g/person, such as in the range of 100 mg to 30 g/person, such as in the range of 100 mg to 20 g/person, such as It may be in the range of 1 g to 20 g/person, such as in the range of 2 g to 20 g/person, such as in the range of 1 g to 10 g/person, such as in the range of 100 mg to 1 g/person.
- the number of administrations and the administration frequency are not limited, but for example, the administration can be performed once a day or two or three times at an interval of one day to two or three days. Also, for example, it can be administered only once, and can be administered again several days later for a total of two administrations.
- the present invention provides a method or a pharmaceutical composition for inhibiting precipitation of an antisense oligomer in urine in a subject administered an antisense oligomer or a pharmaceutical composition comprising an antisense oligomer. comprising adding sugar in an amount such that the concentration of sugar in said pharmaceutical composition is between 0.5 mg/mL and 3000 mg/mL.
- the antisense oligomer, the pharmaceutical composition, the saccharide, the concentration of the saccharide in the pharmaceutical composition, etc. are as described herein.
- the present invention provides a method for suppressing precipitation of an antisense oligomer in urine in a subject administered an antisense oligomer or a pharmaceutical composition comprising an antisense oligomer, comprising: to the subject, in an amount such that the weight ratio of saccharide to 1 of the antisense oligomer is 0.1-100.
- the weight ratio of the antisense oligomer, the pharmaceutical composition, the sugar, the precipitation inhibitor, the sugar to 1 for the antisense oligomer, and the like are as described in this specification.
- the present invention relates to a pharmaceutical composition
- a pharmaceutical composition comprising an antisense oligomer, the pharmaceutical composition comprising a sugar-containing deposition inhibitor at a concentration of 0.5 mg/mL to 3000 mg/mL.
- the antisense oligomer, the pharmaceutical composition, the saccharide, the concentration of the saccharide in the pharmaceutical composition, etc. are as described herein.
- Antisense oligomers (PMO Nos. 1 to 12 (SEQ ID NOS: 1 to 12)) shown in Table 1 were synthesized according to the method described in International Publication WO2013/100190. The theoretical and ESI-TOF-MS molecular weights of each antisense oligomer are also shown.
- Example 2 Preliminary examination of evaluation of precipitation inhibitory action by sugar
- D-mannitol which is equivalent to the reduced form of D-mannose, which is a monosaccharide
- antisense oligomers were administered to cynomolgus monkeys in order to establish test conditions such as concentrations of sugar and antisense oligomers assuming urine, and changes in plasma concentrations were observed. and the urinary concentration in the short time after administration was estimated from the systemic clearance.
- the urinary concentration in a short time after administration of 500 mg/kg of D-mannitol was estimated to be 127.5 mg/mL. Since D-mannitol is hardly distributed in the body and is rapidly excreted in the urine without being metabolized, it can be assumed that the dosage and urinary concentration are linear.
- the urinary concentration was converted to D-mannitol dosage, and the dosage was converted to the concentration in the formulation at the time of administration (at the time of use) based on the body weight and the volume of the administered solution.
- D-mannitol dose urine concentration (mg/mL)/127.5 (mg/mL) x 500 (mg/kg)
- D-mannitol concentration in formulation D-mannitol dose (mg/kg) ⁇ body weight (kg) ⁇ administration liquid volume (mL)
- body weight was 3-80 kg, and the volume of the administered liquid was 50-200 mL.
- an aqueous sugar solution with an assumed urine concentration of 135 mg/mL can be converted to a urine concentration when 529.4 mg/kg of sugar is administered, and a preparation with a sugar concentration of 7.9 to 847.1 mg/mL is administered ( use).
- PMO No. The antisense oligomer of 12 (SEQ ID NO: 12) was dissolved in physiological saline at 1 and 6 mg/mL, and administered at 10 mL/kg to three 1-year-old male cynomolgus monkeys. Plasma concentrations were measured at 0.25 hours, 0.5 hours, 1 hour, 2 hours, 6 hours and 24 hours (Table 3, values a, b, c, d, e, f, g, each value is average of 3 animals). From these values, the area under the plasma concentration-time curve (AUC 0-24 hr ) and systemic clearance (CL tot ) up to 24 hours after administration were calculated (Table 3, values h, i).
- Antisense oligomer dose urinary concentration (mg/mL)/estimated urinary concentration in cynomolgus monkeys (mg/mL) x dose to cynomolgus monkeys (mg/kg)
- Antisense oligomer concentration in preparation antisense oligomer dose (mg/kg) x body weight (kg) ⁇ administered liquid volume (mL)
- the value obtained when 60 mg/kg of antisense oligomer was administered was used as the estimated urinary concentration in cynomolgus monkeys.
- the body weight was 3 to 80 kg, and the volume of the administered liquid was 50 to 200 mL.
- an antisense oligomer aqueous solution with an assumed urinary concentration of 24.75 mg/mL can be converted to a urine concentration when an antisense oligomer of 43.41 mg/kg is administered, and the antisense oligomer concentration is 0.7-69. It is envisioned that a 5 mg/mL formulation will be administered (used).
- Example 3 Evaluation of precipitation suppression effect of sucrose
- Each antisense oligomer was dissolved in water for injection or in an aqueous sucrose solution and mixed with an aqueous solution containing potassium chloride and sodium chloride assuming urine.
- the evaluated conditions are shown in Tables 4 to 11.
- the absorbance at 620 nm of the mixed solution was measured under heating conditions of 37° C. using a plate reader Infinite F200 Pro (manufactured by TECAN).
- the concentrations of antisense oligomers, potassium chloride, and sodium chloride will vary depending on the dose of antisense oligomers and urine volume. was used to set the conditions under which precipitation was observed.
- Example 5 Evaluation of the inhibitory effect of sucrose on precipitation in the kidney
- PMO No. 11 SEQ ID NO: 11
- the 11 antisense oligomers were dissolved in physiological saline to prepare a control dosing solution containing the antisense oligomers at the concentrations shown in Table 14 and no sucrose.
- mice were necropsied and formalin-fixed, paraffin-embedded specimens of kidneys were prepared. Sections obtained by slicing the specimens were stained with hematoxylin and eosin and subjected to histopathological examination. Precipitation was determined to be suppressed when improvement was observed in renal histopathological examination findings in mice administered an administration solution containing sucrose compared to mice administered an administration solution containing no sucrose. .
- Evaluation results The tested antisense oligomers showed improvement in renal histopathological examination findings in mice administered with a dosing solution containing sucrose, confirming that sucrose suppresses precipitation. The results are shown in Table 14.
- Example 6 Evaluation of precipitation suppression effect of sucrose
- PMO No. for 1, 2, and 7, in order to further verify the usefulness for medical applications, the amount of precipitation in the presence of ions assuming urine and the inhibition of precipitation by sucrose were evaluated.
- Example 7 Evaluation of precipitation suppression effect of trehalose
- Each antisense oligomer was dissolved in water for injection or an aqueous trehalose solution and mixed with an aqueous solution containing potassium chloride and sodium chloride assuming urine.
- the evaluated conditions are shown in Tables 18-21.
- the absorbance at 620 nm of the mixed solution was measured under heating conditions of 37° C. using a plate reader Infinite F200 Pro (manufactured by TECAN).
- the concentrations of antisense oligomers, potassium chloride, and sodium chloride will vary depending on the dose of antisense oligomers and urine volume. was used to set the conditions under which precipitation was observed.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Genetics & Genomics (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Biomedical Technology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- General Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Urology & Nephrology (AREA)
- Plant Pathology (AREA)
- Physics & Mathematics (AREA)
- Biophysics (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Microbiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
(1)アンチセンスオリゴマーを含む医薬組成物が投与された対象における尿中のアンチセンスオリゴマーの析出抑制剤であって、グルコースではない糖を含み、前記医薬組成物中の糖の濃度が0.5mg/mL~3000mg/mLになる量で使用される、析出抑制剤。
(2)前記医薬組成物中の糖の濃度が2.6mg/mL~1694.1mg/mLになる量で使用される、(1)に記載の析出抑制剤。
(3)前記医薬組成物中のアンチセンスオリゴマーの濃度が、0.05mg/mL~2000mg/mLである、(1)又は(2)に記載の析出抑制剤。
(4-1)アンチセンスオリゴマーを含む医薬組成物が投与された対象における尿中のアンチセンスオリゴマーの析出抑制剤であって、グルコースではない糖を含み、アンチセンスオリゴマーを1としたときの糖の重量比が0.1~100となる量で使用される、析出抑制剤。
(4-2)前記医薬組成物と析出抑制剤が別々に投与される、(4-1)に記載の析出抑制剤。
(5)アンチセンスオリゴマーを1としたときの糖の重量比が1~53となる量で使用される、(4-1)又は(4-2)に記載の析出抑制剤。
(6)糖が、二糖類の糖である、(1)~(5)のいずれかに記載の析出抑制剤。
(7)糖が、スクロースである、(1)~(6)のいずれかに記載の析出抑制剤。
(8)糖が、トレハロースである、(1)~(6)のいずれかに記載の析出抑制剤。
(9)アンチセンスオリゴマーがモルホリノオリゴマーである、(1)~(8)のいずれかに記載の析出抑制剤。
(10)アンチセンスオリゴマーがホスホロジアミデートモルホリノオリゴマーである、(1)~(9)のいずれかに記載の析出抑制剤。
(11)アンチセンスオリゴマーの5'末端が、下記化学式(1)~(2):
(12)前記アンチセンスオリゴマーが、その塩基配列中に4つの連続するプリン塩基を含む、(1)~(11)のいずれかに記載の析出抑制剤。
(13)前記4つの連続するプリン塩基のうちの少なくとも2つがグアニンである、(12)に記載の析出抑制剤。
(14)前記アンチセンスオリゴマーが、配列番号1~12からなる群から選択される塩基配列を含む、(1)~(13)のいずれかに記載の析出抑制剤。
(15-1)アンチセンスオリゴマーを含む医薬組成物が投与された対象において、尿中のアンチセンスオリゴマーの析出を抑制するための方法であって、グルコースではない糖を、前記医薬組成物中の糖の濃度が0.5mg/mL~3000mg/mLになる量で添加することを含む、方法。
(15-2)糖を、前記医薬組成物中の糖の濃度が2.6mg/mL~1694.1mg/mLになる量で添加する、(15-1)に記載の方法。
(15-3)前記医薬組成物中のアンチセンスオリゴマーの濃度が、0.05mg/mL~2000mg/mLである、(15-1)又は(15-2)に記載の方法。
(16-1)アンチセンスオリゴマーが投与された対象において、尿中のアンチセンスオリゴマーの析出を抑制するための方法であって、グルコースではない糖を前記対象に投与することを含み、アンチセンスオリゴマーを1としたときの糖の重量比が0.1~100となる量で投与される、方法。
(16-2)前記アンチセンスオリゴマーと前記糖が別々に投与される、(16-1)に記載の方法。
(16-3)糖が、アンチセンスオリゴマーを1としたときの糖の重量比が1~53となる量で使用される、(16-1)又は(16-2)に記載の方法。
(17-1)アンチセンスオリゴマーを含む医薬組成物であって、グルコースではない糖を含む、前記医薬組成物が投与された対象における尿中のアンチセンスオリゴマーの析出抑制剤を0.5mg/mL~3000mg/mLの濃度で含む、医薬組成物。
(17-2)糖を含む析出抑制剤を2.6mg/mL~1694.1mg/mLの濃度で含む、(17-1)に記載の医薬組成物。
(17-3)前記医薬組成物中のアンチセンスオリゴマーの濃度が、0.05mg/mL~2000mg/mLである、(17-1)又は(17-2)に記載の医薬組成物。
(18)糖が、二糖類の糖である、(15-1)~(17-3)のいずれかに記載の方法又は医薬組成物。
(19)糖が、スクロースである、(15-1)~(18)のいずれかに記載の方法又は医薬組成物。
(20)糖が、トレハロースである、(15-1)~(18)のいずれかに記載の方法又は医薬組成物。
(21)アンチセンスオリゴマーがモルホリノオリゴマーである、(15-1)~(20)のいずれかに記載の方法又は医薬組成物。
(22)アンチセンスオリゴマーがホスホロジアミデートモルホリノオリゴマーである、(15-1)~(21)のいずれかに記載の方法又は医薬組成物。
(23)アンチセンスオリゴマーの5'末端が、下記化学式(1)~(2):
(24)前記アンチセンスオリゴマーが、その塩基配列中に4つの連続するプリン塩基を含む、(15-1)~(23)のいずれかに記載の方法又は医薬組成物。
(25)前記4つの連続するプリン塩基のうちの少なくとも2つがグアニンである、(24)に記載の方法又は医薬組成物。
(26)前記アンチセンスオリゴマーが、配列番号1~12からなる群から選択される塩基配列を含む、(15-1)~(25)のいずれかに記載の方法又は医薬組成物。
D-マンニトール製剤中濃度(mg/mL)=D-マンニトール投与量(mg/kg)×体重(kg)÷投与液量(mL)
体重は、例えば3~80kg、投与液量は、例えば50~200mLを想定することができる。
Wは、以下のいずれかの式で表わされる基を表す。
R1は、H、アルキルを表し;
R2及びR3は、同一又は異なって、H、アルキル、シクロアルキル、又はアリールを表し;
Y1は、O、S、CH2、又はNR1を表し;
Y2は、O、S、又はNR1を表し;
Zは、O又はSを表す。))
本明細書において、モルホリノオリゴマーは、好ましくは、以下の式で表わされる基を構成単位とするオリゴマー(ホスホロジアミデートモルホリノオリゴマー(以下、「PMO」という))である。
モルホリノオリゴマーは、例えば、国際公開公報第1991/009033号、又は国際公開公報第2009/064471号に記載の方法に従って製造することができる。特に、PMOは、国際公開公報第2009/064471号、又は国際公開公報第2013/100190号に記載の方法に従って製造することができる。
ペプチド核酸オリゴマーは、例えば、以下の文献に従って製造することができる。
1)P. E. Nielsen, M. Egholm, R. H. Berg, O. Buchardt,Science, 254, 1497 (1991)2)M. Egholm, O. Buchardt, P. E. Nielsen, R. H. Berg,JACS, 114, 1895 (1992)
3)K. L. Dueholm, M. Egholm, C. Behrens, L. Christensen, H. F. Hansen, T. Vulpius, K. H. Petersen, R. H. Berg, P. E. Nielsen, O. Buchardt,J. Org. Chem., 59, 5767 (1994)
4)L. Christensen, R. Fitzpatrick, B. Gildea, K. H. Petersen, H. F. Hansen, T. Koch, M. Egholm,O. Buchardt, P. E. Nielsen, J. Coull, R. H. Berg, J. Pept. Sci., 1, 175 (1995)5)T. Koch, H. F. Hansen, P. Andersen, T. Larsen, H. G. Batz, K. Otteson, H. Orum, J. Pept. Res., 49, 80 (1997)
本明細書に記載のアンチセンスオリゴマーは、その医薬的に許容可能な塩の形態、水和物の形態、又は医薬的に許容可能な塩の水和物の形態であってもよい。
& Therapeutics 154 (2015) 78-86の80頁の表1に列挙されたCPPが含まれ、例えば、TAT(48-60)、ペネトラチン、ポリアルギニン、Oct4、WT1-pTj、DPV3、トランスポータン、MAP、VP22、Rep1、KW、KFGF、FGF12、インテフリンβ3ペプチド、C105Y、TP2;特表2017-500856明細書(国際公開第2015/089487号パンフレット)の段落[0085]、表1に列挙されるCPP、例えばDPV10/6、DPV15b、YM-3、Tat、LR11、C45D18、Lyp-1、Lyp-2、BMV GAG、hLF1-22、C45D18、LR20などが挙げられる。CPPは、例えば、Funakoshi,Co.,Ltd.から市販されている。TAT(Funakoshi,Co.,Ltd.)、ペネトラチン(Funakoshi,Co.,Ltd.)などの市販のCPP、又は公知のCPP、例えばR8などを本明細書において使用することができる。本明細書において用いることができる好ましいCPPは、例えば、hLIMK、TAT、ペネトラチン、R8などを含む(国際公開第2016/187425号パンフレット、国際公開第2018/118662号パンフレット、国際公開第2018/118599号パンフレット、国際公開第2018/118627号パンフレット、EBioMedicine 45 (2019) 630-645などを参照されたい)。CPPは、本明細書のアンチセンスオリゴマーに直接結合することができ、又はCPPをアンチセンスオリゴマーに結合することができるリンカーを介して結合することができる。公知のリンカーを本明細書において使用することができる。このようなリンカーには、例えば、特表2017-500856明細書(国際公開第2015/089487号パンフレット)、国際公開第2015/089487号パンフレット、国際公開第2009/073809号パンフレット、国際公開第2013/075035号パンフレット、国際公開第2015/105083号パンフレット、国際公開第2014/179620号パンフレット、国際公開第2015/006740号パンフレット、国際公開第2017/010575号パンフレットなどに記載されるものが含まれる。本明細書において使用することができる好ましいリンカーは、例えば、4-マレイミド酪酸、ジスルフィド結合を介して本明細書の機能性ペプチド又はアンチセンスオリゴマーに結合することができるリンカーなどを含む。本明細書のコンジュゲートは、当業者に公知の方法によって調製することができる。
国際公開公報WO2013/100190に記載の方法に従い、表1に示すアンチセンスオリゴマー(PMO No.1~12(配列番号1~12))を合成した。各アンチセンスオリゴマーの分子量の理論値及びESI-TOF-MSによる実測値も示す。
尿中を想定した糖及びアンチセンスオリゴマーの濃度等の試験条件設定のため、単糖であるD-マンノースの還元体に相当するD-マンニトール及びアンチセンスオリゴマーをカニクイザルに投与し、血漿中濃度推移と全身クリアランスから投与後短時間における尿中濃度を推定した。
Smith Pharmacokinetics and toxicity testing. Crit Rev Toxicol. 1984;12(4):343-85.)から3.91mLとし、投与後0.25時間の尿中マンニトール濃度を推定した(表2、値k)。
D-マンニトール製剤中濃度(mg/mL)=D-マンニトール投与量(mg/kg)×体重(kg)÷投与液量(mL)
この時、体重を3~80kg、投与液量を50~200mLとした。
アンチセンスオリゴマー製剤中濃度(mg/mL)=アンチセンスオリゴマー投与量(mg/kg)×体重(kg)÷投与液量(mL)
この時、カニクイザルにおける推定尿中濃度は、アンチセンスオリゴマー60mg/kgを投与した際の値を使用した。また、体重を3~80kg、投与液量を50~200mLとした。例えば、尿中濃度24.75mg/mLを想定したアンチセンスオリゴマー水溶液は、アンチセンスオリゴマー43.41mg/kgを投与した場合の尿中濃度に換算でき、アンチセンスオリゴマー濃度が0.7~69.5mg/mLの製剤を投与(使用)することを想定している。
[実施例3:スクロースによる析出抑制作用の評価]
表1に示すアンチセンスオリゴマーのうち、PMO No.8、9、10、11について、医薬用途への有用性をさらに検証するため、尿中を想定したイオン存在下での析出量及びスクロースによる析出抑制作用を評価した。
試験方法
各アンチセンスオリゴマーを注射用水又はスクロース水溶液に溶解し、尿を想定した塩化カリウム及び塩化ナトリウムを含む水溶液と混和した。評価した条件(溶液の組成)を、表4~11に示す。混和した溶液をプレートリーダーInfinite F200 Pro(TECAN社製)を用いて37℃加温条件下、620nmでの吸光度を測定した。尿中でのアンチセンスオリゴマーの析出を想定した場合、アンチセンスオリゴマーの濃度、塩化カリウム濃度及び塩化ナトリウム濃度は、アンチセンスオリゴマーの投与量や尿量によって変動するため、配列ごとに注射用水を媒体に用いて析出がみられる条件を設定した。得られた条件から媒体のみをスクロース水溶液に変更した条件で測定し、両条件において得られた吸光度の比較を行った。吸光度が上昇した場合に、析出がみられたと判断し、析出がみられるまでの時間及び吸光度上昇値を評価した。スクロース水溶液を媒体とした場合に、注射用水を媒体とした場合よりも析出がみられるまでの時間が遅い、又は吸光度上昇値が小さい場合に、析出抑制作用があると判定した。
試験した各アンチセンスオリゴマーは、全てスクロース水溶液を媒体とした場合に、析出がみられるまでの時間が遅延し、吸光度の上昇を抑制したことから、スクロースはアンチセンスオリゴマーの析出を抑制することが示された。それぞれの結果について、図1~8に示す。
[実施例4:スクロースによる腎臓における析出抑制作用の評価]
表1に示すアンチセンスオリゴマーのうち、PMO No.8(配列番号8)について、医薬用途へのさらなる有用性を検証するため、腎臓における尿からの析出及びスクロースによる析出抑制について評価した。
(1)評価方法
PMO No.8のアンチセンスオリゴマーを0.9%の塩化ナトリウムを含む注射用水に溶解し、さらにスクロース(富士フィルム和光純薬製)を生理食塩水で溶解した溶液を加えて、各アンチセンスオリゴマーとスクロースを表12、13に記載の濃度で含む投与液を作製した。
病理組織学的検査により、スクロースを含まない対照の投与液を投与したマウスの腎臓において尿細管の管腔内に好塩基性物質が認められた。この変化は核酸の析出を反映したものと考えられた。スクロース濃度に応じて析出物は減少し、析出抑制作用が認められた。結果を、表13及び図9、10、11、12に示す。
表1に示すアンチセンスオリゴマーのうち、PMO No.11(配列番号11)について、医薬用途へのさらなる有用性を検証するため、腎臓における尿からの析出及びスクロースによる析出抑制について評価した。
(1)評価方法
PMO No.11のアンチセンスオリゴマーを生理食塩水に溶解し、アンチセンスオリゴマーを表14に記載の濃度で含みスクロースを含まない対照の投与液を作製した。
(2)評価結果
試験したアンチセンスオリゴマーは、スクロースを含む投与液を投与したマウスにおいて、腎臓の病理組織学的検査における所見で改善が認められたため、スクロースが析出を抑制することを確認した。結果を、表14に示す。
表1に示すアンチセンスオリゴマーのうち、PMO No.1、2、7について、医薬用途への有用性をさらに検証するため、尿中を想定したイオン存在下での析出量及びスクロースによる析出抑制作用を評価した。
各アンチセンスオリゴマーを注射用水又はスクロース水溶液に溶解し、尿を想定した塩化カリウム及び塩化ナトリウムを含む水溶液と混和した。評価した条件(溶液の組成)を、表4~11、15~17に示す。混和した溶液をプレートリーダーInfinite F200 Pro(TECAN社製)を用いて37℃加温条件下、620nmでの吸光度を測定した。尿中でのアンチセンスオリゴマーの析出を想定した場合、アンチセンスオリゴマーの濃度、塩化カリウム濃度及び塩化ナトリウム濃度は、アンチセンスオリゴマーの投与量や尿量によって変動するため、配列ごとに注射用水を媒体に用いて析出がみられる条件を設定した。得られた条件から媒体のみをスクロース水溶液に変更した条件で測定し、両条件において得られた吸光度の比較を行った。吸光度が上昇した場合に、析出がみられたと判断し、析出がみられるまでの時間及び吸光度上昇値を評価した。スクロース水溶液を媒体とした場合に、注射用水を媒体とした場合よりも析出がみられるまでの時間が遅い、又は吸光度上昇値が小さい場合に、析出抑制作用があると判定した。
試験した各アンチセンスオリゴマーは、全てスクロース水溶液を媒体とした場合に、析出がみられるまでの時間が遅延し、吸光度の上昇を抑制したことから、スクロースはアンチセンスオリゴマーの析出を抑制することが示された。それぞれの結果について、図13~15に示す。
以上の結果より、スクロースはアンチセンスオリゴマーの尿中での析出を抑制することが示された。
表1に示すアンチセンスオリゴマーのうち、PMO No.1、2、7、8について、医薬用途への有用性をさらに検証するため、尿中を想定したイオン存在下での析出量及びトレハロースによる析出抑制作用を評価した。
各アンチセンスオリゴマーを注射用水又はトレハロース水溶液に溶解し、尿を想定した塩化カリウム及び塩化ナトリウムを含む水溶液と混和した。評価した条件(溶液の組成)を、表18~21に示す。混和した溶液をプレートリーダーInfinite F200 Pro(TECAN社製)を用いて37℃加温条件下、620nmでの吸光度を測定した。尿中でのアンチセンスオリゴマーの析出を想定した場合、アンチセンスオリゴマーの濃度、塩化カリウム濃度及び塩化ナトリウム濃度は、アンチセンスオリゴマーの投与量や尿量によって変動するため、配列ごとに注射用水を媒体に用いて析出がみられる条件を設定した。得られた条件から媒体のみをトレハロース水溶液に変更した条件で測定し、両条件において得られた吸光度の比較を行った。吸光度が上昇した場合に、析出がみられたと判断し、析出がみられるまでの時間及び吸光度上昇値を評価した。トレハロース水溶液を媒体とした場合に、注射用水を媒体とした場合よりも析出がみられるまでの時間が遅い、又は吸光度上昇値が小さい場合に、析出抑制作用があると判定した。
試験した各アンチセンスオリゴマーは、全てトレハロース水溶液を媒体とした場合に、析出がみられるまでの時間が遅延し、吸光度の上昇を抑制したことから、トレハロースはアンチセンスオリゴマーの析出を抑制することが示された。それぞれの結果について、図16~19に示す。
Claims (17)
- アンチセンスオリゴマーを含む医薬組成物が投与された対象における尿中のアンチセンスオリゴマーの析出抑制剤であって、グルコースではない糖を含み、前記医薬組成物中の糖の濃度が0.5mg/mL~3000mg/mLになる量で使用される、析出抑制剤。
- 前記医薬組成物中の糖の濃度が2.6mg/mL~1694.1mg/mLになる量で使用される、請求項1に記載の析出抑制剤。
- 前記医薬組成物中のアンチセンスオリゴマーの濃度が、0.05mg/mL~2000mg/mLである、請求項1又は2に記載の析出抑制剤。
- アンチセンスオリゴマーを含む医薬組成物が投与された対象における尿中のアンチセンスオリゴマーの析出抑制剤であって、グルコースではない糖を含み、アンチセンスオリゴマーを1としたときの糖の重量比が0.1~100となる量で使用される、析出抑制剤。
- アンチセンスオリゴマーを1としたときの糖の重量比が1~53となる量で使用される、請求項4に記載の析出抑制剤。
- 糖が、二糖類の糖である、請求項1~5のいずれか一項に記載の析出抑制剤。
- 糖が、スクロースである、請求項1~6のいずれか一項に記載の析出抑制剤。
- 糖が、トレハロースである、請求項1~6のいずれか一項に記載の析出抑制剤。
- アンチセンスオリゴマーがモルホリノオリゴマーである、請求項1~8のいずれか一項に記載の析出抑制剤。
- アンチセンスオリゴマーがホスホロジアミデートモルホリノオリゴマーである、請求項1~9のいずれか一項に記載の析出抑制剤。
- 前記アンチセンスオリゴマーが、その塩基配列中に4つの連続するプリン塩基を含む、請求項1~11のいずれか一項に記載の析出抑制剤。
- 前記4つの連続するプリン塩基のうちの少なくとも2つがグアニンである、請求項12に記載の析出抑制剤。
- 前記アンチセンスオリゴマーが、配列番号1~12からなる群から選択される塩基配列を含む、請求項1~13のいずれか一項に記載の析出抑制剤。
- アンチセンスオリゴマーを含む医薬組成物が投与された対象において、尿中のアンチセンスオリゴマーの析出を抑制するための方法であって、グルコースではない糖を、前記医薬組成物中の糖の濃度が0.5mg/mL~3000mg/mLになる量で添加することを含む、方法。
- アンチセンスオリゴマーが投与された対象において、尿中のアンチセンスオリゴマーの析出を抑制するための方法であって、グルコースではない糖を前記対象に投与することを含み、アンチセンスオリゴマーを1としたときの糖の重量比が0.1~100となる量で投与される、方法。
- アンチセンスオリゴマーを含む医薬組成物であって、グルコースではない糖を含む、前記医薬組成物が投与された対象における尿中のアンチセンスオリゴマーの析出抑制剤を0.5mg/mL~3000mg/mLの濃度で含む、医薬組成物。
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202280047891.9A CN118434424A (zh) | 2021-07-08 | 2022-07-08 | 析出抑制剂 |
| US18/577,142 US20240285770A1 (en) | 2021-07-08 | 2022-07-08 | Precipitation suppressing agent |
| KR1020247003689A KR20240035503A (ko) | 2021-07-08 | 2022-07-08 | 석출 억제제 |
| IL310001A IL310001A (en) | 2021-07-08 | 2022-07-08 | sediment suppressant |
| JP2022555691A JPWO2023282346A1 (ja) | 2021-07-08 | 2022-07-08 | |
| AU2022306542A AU2022306542A1 (en) | 2021-07-08 | 2022-07-08 | Precipitation suppressing agent |
| CA3225573A CA3225573A1 (en) | 2021-07-08 | 2022-07-08 | Precipitation suppressing agent |
| EP22837760.2A EP4368187A1 (en) | 2021-07-08 | 2022-07-08 | Precipitation suppressing agent |
| JP2022178017A JP7466609B2 (ja) | 2021-07-08 | 2022-11-07 | 析出抑制剤 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021-113562 | 2021-07-08 | ||
| JP2021113562 | 2021-07-08 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2023282346A1 true WO2023282346A1 (ja) | 2023-01-12 |
| WO2023282346A8 WO2023282346A8 (ja) | 2024-02-15 |
Family
ID=84800729
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/027105 WO2023282346A1 (ja) | 2021-07-08 | 2022-07-08 | 析出抑制剤 |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US20240285770A1 (ja) |
| EP (1) | EP4368187A1 (ja) |
| JP (2) | JPWO2023282346A1 (ja) |
| KR (1) | KR20240035503A (ja) |
| CN (1) | CN118434424A (ja) |
| AU (1) | AU2022306542A1 (ja) |
| CA (1) | CA3225573A1 (ja) |
| IL (1) | IL310001A (ja) |
| TW (1) | TW202317147A (ja) |
| WO (1) | WO2023282346A1 (ja) |
Citations (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1991009033A1 (en) | 1989-12-20 | 1991-06-27 | Anti-Gene Development Group | Uncharged morpholino-based polymers having phosphorous-containing chiral intersubunit linkages |
| WO2002036767A2 (en) | 2000-11-02 | 2002-05-10 | Inex Pharmaceuticals Corporation | Therapeutic oligonucleotides of reduced toxicity |
| WO2006038608A1 (ja) | 2004-10-05 | 2006-04-13 | Nippon Shinyaku Co., Ltd. | オリゴ二本鎖rna及び医薬組成物 |
| CN1868456A (zh) * | 2006-06-25 | 2006-11-29 | 沈阳药科大学 | 反义脱氧寡核苷酸isis23665脂质体制剂及制备方法 |
| WO2006129594A1 (ja) | 2005-05-30 | 2006-12-07 | Nippon Shinyaku Co., Ltd. | 核酸含有複合体製剤の製造方法 |
| WO2008036127A2 (en) | 2006-05-10 | 2008-03-27 | Avi Biopharma, Inc. | Oligonucleotide analogs having cationic intersubunit linkages |
| JP2008520600A (ja) * | 2004-11-19 | 2008-06-19 | ノヴォソム アクチェンゲゼルシャフト | 局所投与のための医薬組成物におけるまたはそれに関する改善 |
| WO2009005793A2 (en) | 2007-06-29 | 2009-01-08 | Avi Biopharma, Inc. | Tissue specific peptide conjugates and methods |
| WO2009064471A1 (en) | 2007-11-15 | 2009-05-22 | Avi Biopharma, Inc. | Method of synthesis of morpholino oligomers |
| WO2009073809A2 (en) | 2007-12-04 | 2009-06-11 | Alnylam Pharmaceuticals, Inc. | Carbohydrate conjugates as delivery agents for oligonucleotides |
| WO2010009038A2 (en) | 2008-07-18 | 2010-01-21 | Oncogenex Technologies Inc. | Antisense formulation |
| WO2012150960A1 (en) | 2011-05-05 | 2012-11-08 | Avi Biopharma, Inc. | Peptide oligonucleotide conjugates |
| CN102989007A (zh) * | 2011-06-29 | 2013-03-27 | 湖州市中心医院 | 大规模制备注射用中期因子反义寡核苷酸纳米脂质体的方法 |
| WO2013075035A1 (en) | 2011-11-18 | 2013-05-23 | Alnylam Pharmaceuticals | Rnai agents, compositions and methods of use thereof for treating transthyretin (ttr) associated diseases |
| WO2013100190A1 (ja) | 2011-12-28 | 2013-07-04 | 日本新薬株式会社 | アンチセンス核酸 |
| WO2014179620A1 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| WO2015006740A2 (en) | 2013-07-11 | 2015-01-15 | Alnylam Pharmaceuticals, Inc. | Oligonucleotide-ligand conjugates and process for their preparation |
| JP2015091229A (ja) | 2013-10-04 | 2015-05-14 | 国立大学法人神戸大学 | 福山型筋ジストロフィー治療用アンチセンス核酸 |
| WO2015089487A1 (en) | 2013-12-12 | 2015-06-18 | Life Technologies Corporation | Membrane-penetrating peptides to enhance transfection and compositions and methods for using same |
| WO2015105083A1 (ja) | 2014-01-07 | 2015-07-16 | 塩野義製薬株式会社 | アンチセンスオリゴヌクレオチド及び糖誘導体を含む二本鎖オリゴヌクレオチド |
| WO2016187425A1 (en) | 2015-05-19 | 2016-11-24 | Sarepta Therapeutics, Inc. | Peptide oligonucleotide conjugates |
| WO2017010575A1 (ja) | 2015-07-16 | 2017-01-19 | 協和発酵キリン株式会社 | β2GPI遺伝子発現抑制核酸複合体 |
| WO2018118599A1 (en) | 2016-12-19 | 2018-06-28 | Sarepta Therapeutics, Inc. | Exon skipping oligomer conjugates for muscular dystrophy |
| WO2018118627A1 (en) | 2016-12-19 | 2018-06-28 | Sarepta Therapeutics, Inc. | Exon skipping oligomer conjugates for muscular dystrophy |
| WO2018118662A1 (en) | 2016-12-19 | 2018-06-28 | Sarepta Therapeutics, Inc. | Exon skipping oligomer conjugates for muscular dystrophy |
-
2022
- 2022-07-08 CN CN202280047891.9A patent/CN118434424A/zh active Pending
- 2022-07-08 CA CA3225573A patent/CA3225573A1/en active Pending
- 2022-07-08 EP EP22837760.2A patent/EP4368187A1/en active Pending
- 2022-07-08 IL IL310001A patent/IL310001A/en unknown
- 2022-07-08 KR KR1020247003689A patent/KR20240035503A/ko unknown
- 2022-07-08 US US18/577,142 patent/US20240285770A1/en active Pending
- 2022-07-08 AU AU2022306542A patent/AU2022306542A1/en active Pending
- 2022-07-08 WO PCT/JP2022/027105 patent/WO2023282346A1/ja active Application Filing
- 2022-07-08 JP JP2022555691A patent/JPWO2023282346A1/ja active Pending
- 2022-07-08 TW TW111125775A patent/TW202317147A/zh unknown
- 2022-11-07 JP JP2022178017A patent/JP7466609B2/ja active Active
Patent Citations (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1991009033A1 (en) | 1989-12-20 | 1991-06-27 | Anti-Gene Development Group | Uncharged morpholino-based polymers having phosphorous-containing chiral intersubunit linkages |
| WO2002036767A2 (en) | 2000-11-02 | 2002-05-10 | Inex Pharmaceuticals Corporation | Therapeutic oligonucleotides of reduced toxicity |
| JP2004512383A (ja) * | 2000-11-02 | 2004-04-22 | アイネックス ファーマシューティカルズ コーポレイション | 毒性の減少した治療用オリゴヌクレオチド |
| WO2006038608A1 (ja) | 2004-10-05 | 2006-04-13 | Nippon Shinyaku Co., Ltd. | オリゴ二本鎖rna及び医薬組成物 |
| JP2008520600A (ja) * | 2004-11-19 | 2008-06-19 | ノヴォソム アクチェンゲゼルシャフト | 局所投与のための医薬組成物におけるまたはそれに関する改善 |
| WO2006129594A1 (ja) | 2005-05-30 | 2006-12-07 | Nippon Shinyaku Co., Ltd. | 核酸含有複合体製剤の製造方法 |
| WO2008036127A2 (en) | 2006-05-10 | 2008-03-27 | Avi Biopharma, Inc. | Oligonucleotide analogs having cationic intersubunit linkages |
| CN1868456A (zh) * | 2006-06-25 | 2006-11-29 | 沈阳药科大学 | 反义脱氧寡核苷酸isis23665脂质体制剂及制备方法 |
| WO2009005793A2 (en) | 2007-06-29 | 2009-01-08 | Avi Biopharma, Inc. | Tissue specific peptide conjugates and methods |
| WO2009064471A1 (en) | 2007-11-15 | 2009-05-22 | Avi Biopharma, Inc. | Method of synthesis of morpholino oligomers |
| WO2009073809A2 (en) | 2007-12-04 | 2009-06-11 | Alnylam Pharmaceuticals, Inc. | Carbohydrate conjugates as delivery agents for oligonucleotides |
| JP2011528657A (ja) * | 2008-07-18 | 2011-11-24 | オンコジェネックス・テクノロジーズ・インコーポレーテッド | アンチセンス製剤 |
| WO2010009038A2 (en) | 2008-07-18 | 2010-01-21 | Oncogenex Technologies Inc. | Antisense formulation |
| WO2012150960A1 (en) | 2011-05-05 | 2012-11-08 | Avi Biopharma, Inc. | Peptide oligonucleotide conjugates |
| CN102989007A (zh) * | 2011-06-29 | 2013-03-27 | 湖州市中心医院 | 大规模制备注射用中期因子反义寡核苷酸纳米脂质体的方法 |
| WO2013075035A1 (en) | 2011-11-18 | 2013-05-23 | Alnylam Pharmaceuticals | Rnai agents, compositions and methods of use thereof for treating transthyretin (ttr) associated diseases |
| WO2013100190A1 (ja) | 2011-12-28 | 2013-07-04 | 日本新薬株式会社 | アンチセンス核酸 |
| WO2014179620A1 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| WO2015006740A2 (en) | 2013-07-11 | 2015-01-15 | Alnylam Pharmaceuticals, Inc. | Oligonucleotide-ligand conjugates and process for their preparation |
| JP2015091229A (ja) | 2013-10-04 | 2015-05-14 | 国立大学法人神戸大学 | 福山型筋ジストロフィー治療用アンチセンス核酸 |
| WO2015089487A1 (en) | 2013-12-12 | 2015-06-18 | Life Technologies Corporation | Membrane-penetrating peptides to enhance transfection and compositions and methods for using same |
| WO2015105083A1 (ja) | 2014-01-07 | 2015-07-16 | 塩野義製薬株式会社 | アンチセンスオリゴヌクレオチド及び糖誘導体を含む二本鎖オリゴヌクレオチド |
| WO2016187425A1 (en) | 2015-05-19 | 2016-11-24 | Sarepta Therapeutics, Inc. | Peptide oligonucleotide conjugates |
| WO2017010575A1 (ja) | 2015-07-16 | 2017-01-19 | 協和発酵キリン株式会社 | β2GPI遺伝子発現抑制核酸複合体 |
| WO2018118599A1 (en) | 2016-12-19 | 2018-06-28 | Sarepta Therapeutics, Inc. | Exon skipping oligomer conjugates for muscular dystrophy |
| WO2018118627A1 (en) | 2016-12-19 | 2018-06-28 | Sarepta Therapeutics, Inc. | Exon skipping oligomer conjugates for muscular dystrophy |
| WO2018118662A1 (en) | 2016-12-19 | 2018-06-28 | Sarepta Therapeutics, Inc. | Exon skipping oligomer conjugates for muscular dystrophy |
Non-Patent Citations (19)
| Title |
|---|
| "GenBank", Database accession no. AB038490 |
| A W WINKLERJ PARRA, J CLIN INVEST, vol. 16, no. 6, November 1937 (1937-11-01), pages 859 - 67 |
| ALTSCHUL SF ET AL., J MOL BIOL, vol. 215, 1990, pages 403 |
| B CLARKD A SMITH: "Pharmacokinetics and toxicity testing", CRIT REV TOXICOL, vol. 12, no. 4, 1984, pages 343 - 85 |
| CARVER MPCHARLESTON JSSHANKS CZHANG JMENSE MSHARMA AKKAUR HSAZANI P: "Toxicological Characterization of Exon Skipping Phosphorodiamidate Morpholino Oligomers (PMOs) in Non- human Primates", J NEUROMUSCUL DIS, vol. 3, no. 3, 30 August 2016 (2016-08-30), XP009510433, DOI: 10.3233/JND-160157 |
| EBIOMEDICINE, vol. 45, 2019, pages 630 - 645 |
| ENYA ET AL., BIOORGANIC & MEDICINAL CHEMISTRY, vol. 18, 2008, pages 9154 - 9160 |
| HEEMSKERK HADE WINTER CLDE KIMPE SJVAN KUIK-ROMEIJN PHEUVELMANS NPLATENBURG GJVAN OMMEN GJVAN DEUTEKOM JCAARTSMA-RUS A: "In vivo comparison of 2'-O-methyl phosphorothioate and morpholino antisense oligonucleotides for Duchenne muscular dystrophy exon skipping", J GENE MED, vol. 11, no. 3, March 2009 (2009-03-01), pages 257 - 66 |
| HUM MOL GENET, vol. 20, no. 16, 15 August 2011 (2011-08-15), pages 3151 - 3160 |
| K. L. DUEHOLM, M. EGHOLM, C. BEHRENS, L. CHRISTENSEN, H. F. HANSEN, T. VULPIUS, K. H. PETERSEN, R. H. BERG, P. E. NIELSEN, O. BUCH, J. ORG. CHEM., vol. 59, 1994, pages 5767 |
| L. CHRISTENSEN, R. FITZPATRICK, B. GILDEA, K. H. PETERSEN, H. F. HANSEN, T. KOCH, M. EGHOLM, O. BUCHARDT, P. E. NIELSEN, J. COULL,, J. PEPT. SCI., vol. 1, 1995, pages 175 |
| M. EGHOLMO. BUCHARDTP. E. NIELSENR. H. BERG, JACS, vol. 114, 1992, pages 1895 |
| P. E. NIELSENM. EGHOLMR. H. BERGO. BUCHARDT, SCIENCE, vol. 254, 1991, pages 1497 |
| PHARMACOLOGY & THERAPEUTICS, vol. 154, 2015, pages 78 - 86 |
| PROC NATL ACAD SCI USA, vol. 90, 1993, pages 5873 |
| PROC. NATL. ACAD. SCI. USA, 1990, pages 872264 - 2268 |
| SAZANI PNESS KPWELLER DLPOAGE DNELSON KSHREWSBURY AS: "Chemical and mechanistic toxicology evaluation of exon skipping phosphorodiamidate morpholino oligomers in mdx mice", INT J TOXICOL, vol. 30, no. 3, 3 May 2011 (2011-05-03), pages 322 - 33, XP009510407, DOI: 10.1177/1091581811403504 |
| SCIENCE, vol. 227, no. 4693, 1985, pages 1435 - 1441 |
| T. KOCHH. F. HANSENP. ANDERSENT. LARSENH. G. BATZK. OTTESONH. ORUM, J. PEPT. RES., vol. 49, 1997, pages 80 |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202317147A (zh) | 2023-05-01 |
| US20240285770A1 (en) | 2024-08-29 |
| KR20240035503A (ko) | 2024-03-15 |
| JP2023133099A (ja) | 2023-09-22 |
| JPWO2023282346A1 (ja) | 2023-01-12 |
| CN118434424A (zh) | 2024-08-02 |
| IL310001A (en) | 2024-03-01 |
| EP4368187A1 (en) | 2024-05-15 |
| AU2022306542A1 (en) | 2024-01-18 |
| JP7466609B2 (ja) | 2024-04-12 |
| CA3225573A1 (en) | 2023-01-12 |
| WO2023282346A8 (ja) | 2024-02-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102473431B1 (ko) | 안티센스 핵산 | |
| EA031110B1 (ru) | Конъюгаты бороновых кислот олигонуклеотидных аналогов | |
| JP7292636B2 (ja) | エクソン51のスキッピングを誘導するアンチセンス核酸 | |
| EP4083208A1 (en) | Antisense nucleic acid that induces skipping of exon 50 | |
| WO2023282346A1 (ja) | 析出抑制剤 | |
| WO2023282345A1 (ja) | 腎毒性軽減剤 | |
| WO2023282344A1 (ja) | 腎毒性軽減剤 | |
| US20240316093A1 (en) | Nephrotoxicity reducing agent | |
| EP3740500A1 (en) | Compositions and methods for increasing expression of scn2a | |
| WO2020072883A1 (en) | Chirally enriched oligomeric compounds | |
| US12042509B2 (en) | Prekallikrein-modulating compositions and methods of use thereof | |
| JP2021518361A (ja) | アンチセンス送達用キメラペプチド | |
| US20240043836A1 (en) | Complement factor b-modulating compositions and methods of use thereof | |
| TW202346589A (zh) | 抗病毒反義寡聚物 | |
| US20240041913A1 (en) | Angiotensinogen-modulating compositions and methods of use thereof | |
| EP4408525A1 (en) | Angiotensinogen-modulating compositions and methods of use thereof | |
| EP3788065A1 (en) | Methods of improving anemias by combining agents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2022555691 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22837760 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022306542 Country of ref document: AU Ref document number: AU2022306542 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 3225573 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18577142 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 310001 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: P6000050/2024 Country of ref document: AE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112024000295 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 2022306542 Country of ref document: AU Date of ref document: 20220708 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20247003689 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202417007188 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024103044 Country of ref document: RU Ref document number: 2022837760 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11202400104Y Country of ref document: SG |
|
| ENP | Entry into the national phase |
Ref document number: 112024000295 Country of ref document: BR Kind code of ref document: A2 Effective date: 20240108 |