WO2016175240A1 - 多汗症治療用外用剤 - Google Patents
多汗症治療用外用剤 Download PDFInfo
- Publication number
- WO2016175240A1 WO2016175240A1 PCT/JP2016/063188 JP2016063188W WO2016175240A1 WO 2016175240 A1 WO2016175240 A1 WO 2016175240A1 JP 2016063188 W JP2016063188 W JP 2016063188W WO 2016175240 A1 WO2016175240 A1 WO 2016175240A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- agent
- external preparation
- oxybutynin
- lotion
- anticholinergic
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/216—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acids having aromatic rings, e.g. benactizyne, clofibrate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0014—Skin, i.e. galenical aspects of topical compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/06—Ointments; Bases therefor; Other semi-solid forms, e.g. creams, sticks, gels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- the present invention relates to an external preparation for treating hyperhidrosis.
- Patent Documents 1 and 2 As a method for treating hyperhidrosis, a method of administering an external composition containing an anticholinergic agent such as oxybutynin has been proposed (Patent Documents 1 and 2).
- An anticholinergic agent is a drug that inhibits binding of acetylcholine to a muscarinic acetylcholine receptor and suppresses parasympathetic nerves. Therefore, administration of anticholinergics may cause side effects such as dry mouth.
- the present inventors In order to treat hyperhidrosis while suppressing the above-mentioned side effects, the present inventors consider that it is important to increase the accumulation of anticholinergic drugs on sweat glands, which are the appendages of the skin, and have conducted intensive studies. It was. As a result, the present inventors have found that a predetermined salt enhances the accumulation of an anticholinergic drug on the appendages of the skin, and have completed the present invention.
- the present invention provides an external preparation for treating hyperhidrosis comprising water, an anticholinergic agent, and one or more salts selected from the group consisting of lactate, tartrate, acetate and phosphate.
- the anticholinergic agent may be oxybutynin or a pharmaceutically acceptable salt thereof.
- the salt may be sodium lactate.
- the external preparation may be a liquid agent.
- the solution may be a lotion dosage form.
- the external preparation for treating hyperhidrosis according to the present invention contains one or more salts selected from the group consisting of lactate, tartrate, acetate and phosphate. Therefore, the accumulation of anticholinergic agents on the skin appendages is high.
- One embodiment of the present invention is an external preparation for treating hyperhidrosis comprising water, an anticholinergic agent and one or more salts selected from the group consisting of lactate, tartrate, acetate and phosphate. .
- the anticholinergic agent is not particularly limited as long as it is an arbitrary drug having an anticholinergic effect, for example, oxybutynin, imidafenacin, tropium, tolterodine, glycopyrrolate, propantheline, benztropine, atropine, homatropine, tropicamide, benactidine, Biberidene, scopolamine, butyl scopolamine bromide, cyclopentrate, darifenacin, dexetimide, dicyclomine, emepronium, hexahydrosiladiphenidol, octyronium, orphenadrine, oxyphenonium, pyrenzepine, procyclidine, darotropium, ipratropium, tiotropium, oxytropium, oxitropium Quinidine, trihexyphenidyl, mivacurium, atracurium, doxaclium, cis atracurium, vector Bro
- the anticholinergic agent is preferably oxybutynin or a pharmaceutically acceptable salt thereof.
- the pharmaceutically acceptable salt of oxybutynin include oxybutynin hydrochloride.
- the content of the anticholinergic agent may be, for example, 0.5 to 35% by mass or 0.5 to 15% by mass based on the total mass of the external preparation.
- the external preparation contains one or more salts selected from the group consisting of lactate, tartrate, acetate and phosphate
- the salt may be anhydrous or hydrated.
- Lactic acid may be either L-form or D-form, or any mixture thereof.
- Tartaric acid may be any of L-form, D-form and meso-form, or any mixture thereof.
- Salts include, for example, salts with monovalent metals such as sodium, potassium and lithium, salts with divalent metals such as calcium and magnesium, salts with trivalent metals such as aluminum, and ammonia, ethylenediamine, triethylamine, And salts with amine compounds such as diethanolamine, triethanolamine and meglumine.
- the salt is preferably lactate, and more preferably sodium lactate.
- the content of the salt may be, for example, 0.1 to 10% by mass based on the total mass of the external preparation.
- the molar ratio of the anticholinergic agent and the salt in the external preparation may be, for example, in the range of 1: 0.5 to 1: 2.
- the external preparation for treating hyperhidrosis may be, for example, a liquid, a cream, a gel, or an aqueous ointment.
- the external preparation for treating hyperhidrosis is a liquid
- water in the liquid becomes a medium for dissolving or dispersing the anticholinergic agent, the salt, and other components.
- the water content may be, for example, 10 to 99% by mass based on the total mass of the liquid agent.
- the liquid preparation includes lower alcohols, surfactants, storage stabilizers, fats and oils, solubilizers, fillers, moisturizers, pH regulators, osmotic pressure regulators, thickeners, cooling agents, astringents and A vasoconstrictor or the like may be included.
- Lower alcohol enhances the solubility and dispersibility of anticholinergic drugs and enhances the distribution of anticholinergic drugs to the skin.
- Specific examples of the lower alcohol include methanol, ethanol and isopropanol.
- the content of the lower alcohol may be, for example, 0 to 90% by mass based on the total mass of the liquid agent.
- the surfactant is useful for emulsifying the anticholinergic agent in a medium such as water.
- the surfactant include nonionic surfactants (polysorbate 20, polysorbate 80, polysorbate 60, polyoxyethylene hydrogenated castor oil 20, polyoxyethylene hydrogenated castor oil 40, polyoxyethylene hydrogenated castor oil 60, and the like).
- ionic surfactants and amphoteric surfactants may be, for example, 0 to 10% by mass based on the total mass of the liquid agent.
- the storage stabilizer examples include paraben, isopropylmethylphenol, phenoxyethanol and thymol.
- fats and oils and solubilizers include fatty acids, fatty acid esters and aliphatic alcohols.
- the filler examples include inorganic powder (such as talc, montmorillonite, smectite and kaolin) and organic powder.
- humectant examples include polyhydric alcohols, sugars, urea, petrolatum, and paraffin.
- the pH of the solution can be in the range of 4.5 to 7.5.
- the measurement of pH is performed using a composite glass electrode in accordance with “2.54 pH measurement method” of the general test method of the 16th revision Japanese Pharmacopoeia.
- Liquids may be in the form of lotions and liniments and are contained in suitable containers (eg, spray containers for spraying liquids, containers for applying liquids and aerosol containers). It may be in the form of a coating agent or a spray.
- suitable containers eg, spray containers for spraying liquids, containers for applying liquids and aerosol containers. It may be in the form of a coating agent or a spray.
- the external preparation for treating hyperhidrosis is a cream
- water, an anticholinergic agent and the salt may be blended in the cream base.
- the cream base is not particularly limited, and may be selected from those usually used such as petrolatum or higher alcohol.
- the cream base may be added with additives usually added to the cream, such as emulsifiers, preservatives, absorption accelerators, and anti-rash agents.
- a gel-like cream agent can also be obtained by adding a gelling agent and a neutralizing agent to the cream agent and adjusting the pH to 4-8.
- Such a gel cream has properties intermediate between the cream and the gel.
- the water content in the cream may be, for example, 0.5 to 70% by weight based on the total weight of the cream.
- the external preparation for treating hyperhidrosis is a gel
- water, an anticholinergic agent and the salt may be blended in the gel base.
- the gel base is not particularly limited, and may be selected from those usually used such as higher alcohols.
- the gel base may be added with additives usually added to the gel, such as a gelling agent, a neutralizing agent, a surfactant, an absorption accelerator, a solubilizing agent, and an anti-rash agent.
- the water content in the gel may be, for example, 0.5 to 70% by mass based on the total mass of the gel.
- water, the anticholinergic agent and the salt may be dissolved or dispersed in a water-soluble base.
- the water-soluble base is not particularly limited, and may be selected from commonly used ones such as solid polyethylene glycol.
- additives usually added to an aqueous ointment such as an absorption accelerator, a humectant, and a rash prevention agent, may be added.
- the water content in the aqueous ointment may be, for example, 0.5 to 30% by mass based on the total mass of the aqueous ointment.
- the external preparation for treating hyperhidrosis can be produced by mixing and mixing the above components.
- the external preparation is shaken, if necessary, and the components are mixed homogeneously. Then, the external preparation is applied, sprayed or sprayed on the skin site where sweating is to be suppressed, and spread as necessary.
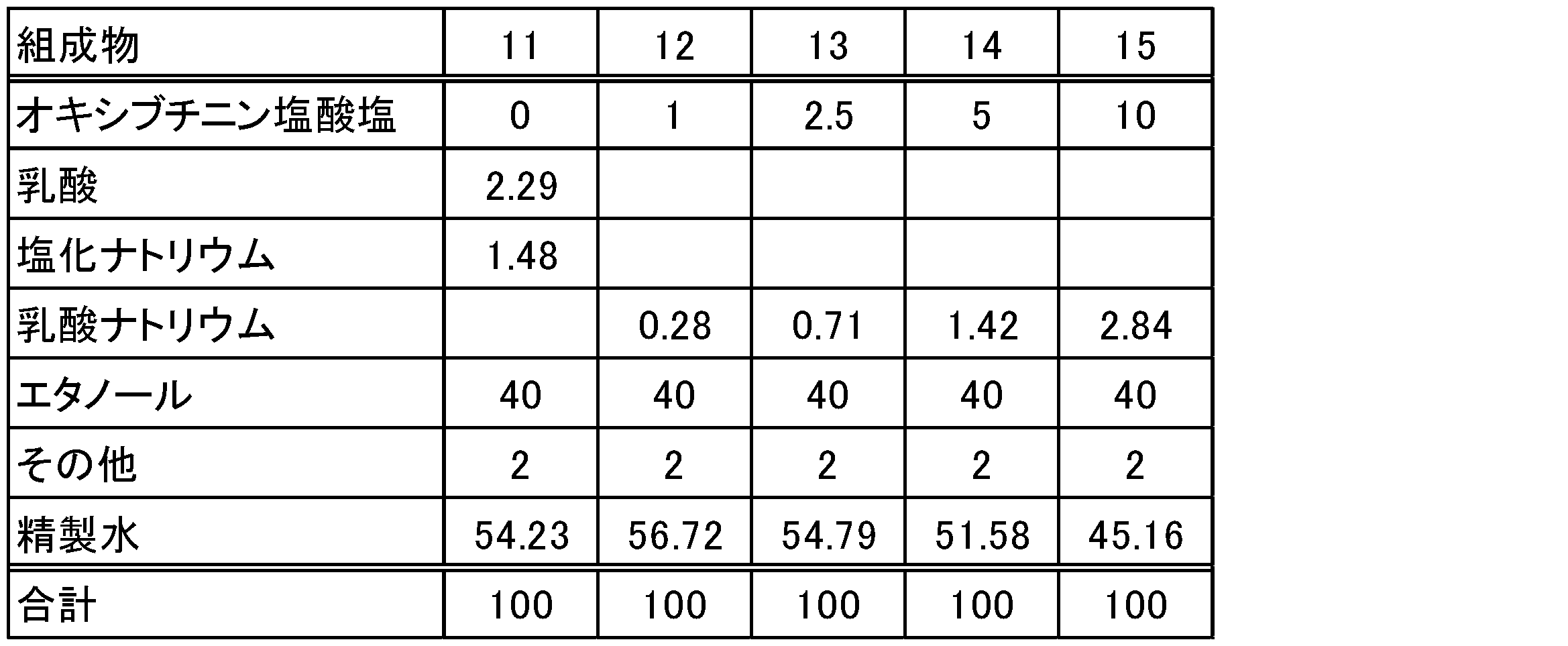
- Test example 2 Lotions were prepared according to the composition in Table 3. The antiperspirant action of the lotion was measured by the pilocarpine-induced sweat test. Further, by the same method as in Test Example 1, a lotion was applied to the skin of pigs, and the concentration of oxybutynin was measured.
- FIGS. FIG. 2 shows the result of the pilocarpine-induced sweat test when the lotion application amount is 10 ⁇ L
- FIG. 3 shows the result of the pilocarpine-induced sweat test when the lotion application amount is 15 ⁇ L. It was confirmed that the antiperspirant action of the lotion was dependent on the oxybutynin concentration. It was also confirmed that the amount of oxybutynin accumulated in the hair follicle was dependent on the oxybutynin concentration.
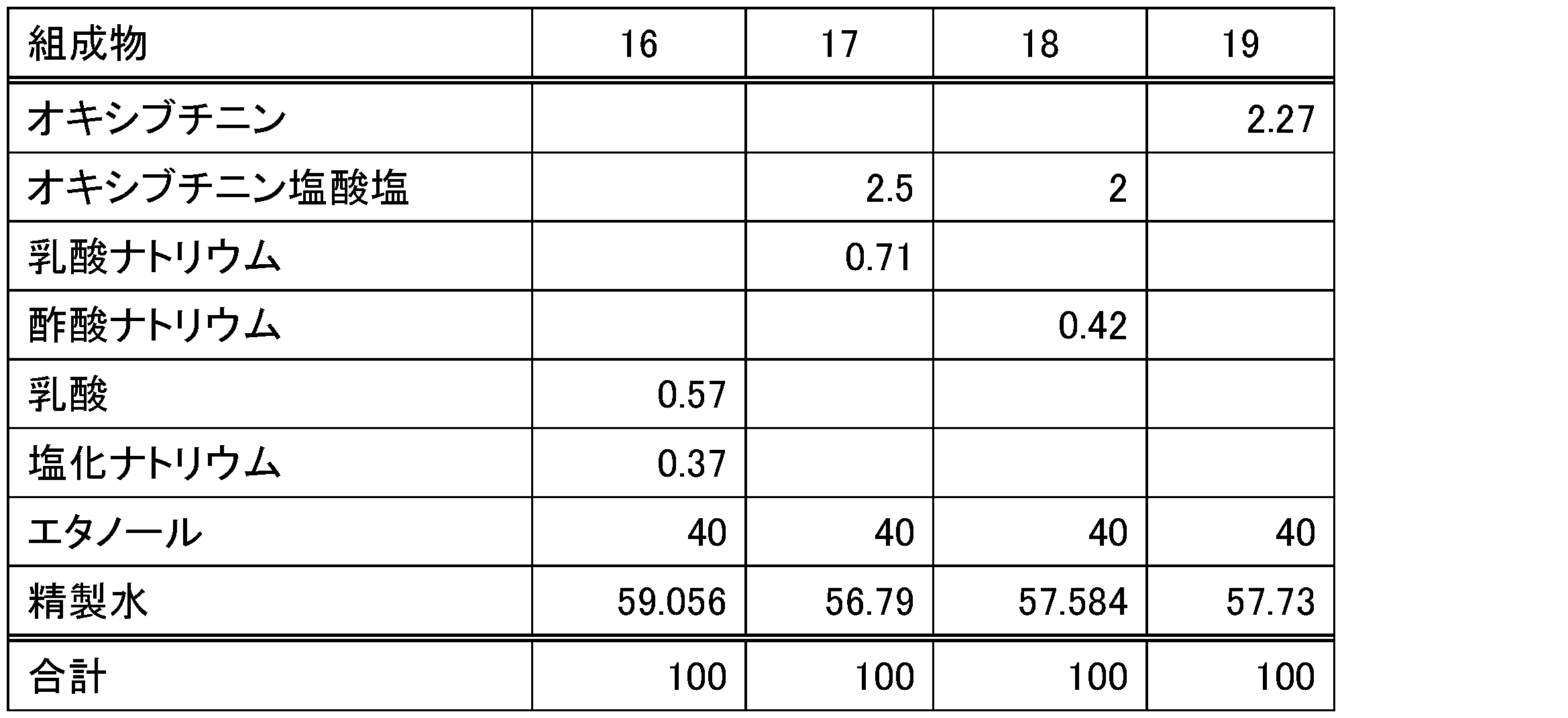
- Test example 3 Lotions were prepared according to the composition in Table 4. The antiperspirant action of the lotion was measured by the pilocarpine-induced sweat test.
- the results are shown in Table 5.
- the lotion containing lactate or acetate showed a stronger antiperspirant action than the lotion containing no salt.
Abstract
Description
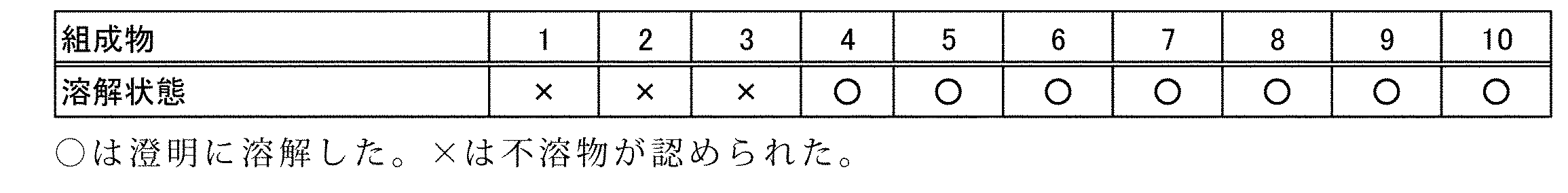
表1の組成に従ってローション剤を調製し、溶解状態を目視で確認した。さらに、ローション剤をブタの皮膚に塗布し、オキシブチニンの毛嚢への集積量を以下の方法で測定した。
1)軽く毛刈りしたブタの皮膚5cm2にローション剤20μLを塗布した。個体数n=3。
2)6時間後、消毒用エタノールで皮膚表面を清拭し、リン酸緩衝液で流水洗浄し、皮膚表面に付着するオキシブチニンを除去した。
3)皮膚から毛20本分の毛嚢部分を採取した。
4)抽出液1mLを用いて毛嚢からオキシブチニンを抽出した。抽出液として、以下の移動相を用いた。
5)高速液体クロマトグラフによりオキシブチニンの濃度を測定した。高速液体クロマトグラフの条件は以下の通りである。
移動相:0.1w/w%リン酸水溶液(ドデシル硫酸ナトリウムを0.5w/v%含む):アセトニトリル=45:55(v/v)
流速:1.5mL/分
カラム:TSKgel ODS-80Ts(東ソー株式会社)
保持時間:10分
表3の組成に従ってローション剤を調製した。ピロカルピン誘発発汗試験により、ローション剤の発汗抑制作用を測定した。また、試験例1と同様の方法により、ローション剤をブタの皮膚に塗布し、オキシブチニンの濃度を測定した。
1)ローション剤を40質量%エタノール水溶液で12倍に希釈した。
2)マウスの足蹠の約0.5cm2にローション剤10μL又は15μLを塗布した。個体数n=5~6。
3)4時間後に麻酔下でヨウ素及びデンプン溶液を足蹠に塗布した。
4)ピロカルピン5μg/足を皮内投与した。
5)5分後にヨウ素デンプン反応により生じる黒点の個数を計数した。
表4の組成に従ってローション剤を調製した。ピロカルピン誘発発汗試験により、ローション剤の発汗抑制作用を測定した。
1)ローション剤を40質量%エタノール水溶液で12倍に希釈した。
2)マウスの足蹠の約0.5cm2にローション剤10μLを塗布した。個体数n=6。
3)3時間後に足蹠を洗浄し、マウスを1時間放置した後、麻酔下でヨウ素及びデンプン溶液を足蹠に塗布した。
4)ピロカルピン15μg/足を皮内投与した。
5)5分後にヨウ素デンプン反応により生じる黒点の個数を計数した。
Claims (5)
- 水、抗コリン作用薬並びに乳酸塩、酒石酸塩、酢酸塩及びリン酸塩からなる群から選択される1以上の塩を含む、多汗症治療用外用剤。
- 抗コリン作用薬が、オキシブチニン又はその薬学的に許容される塩である、請求項1に記載の外用剤。
- 塩が、乳酸ナトリウムである、請求項1又は2に記載の外用剤。
- 液剤である、請求項1~3のいずれか一項に記載の外用剤。
- ローションの剤形である、請求項4に記載の外用剤。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020177027272A KR101937072B1 (ko) | 2015-04-30 | 2016-04-27 | 다한증 치료용 외용제 |
| EP16786516.1A EP3290053B1 (en) | 2015-04-30 | 2016-04-27 | Topical composition comprising oxybutynin for the treatment of hyperhidrosis |
| US15/568,432 US10525132B2 (en) | 2015-04-30 | 2016-04-27 | External agent for treating hyperhidrosis |
| JP2017515577A JP6387458B2 (ja) | 2015-04-30 | 2016-04-27 | 多汗症治療用外用剤 |
| CN201680019209.XA CN107427582B (zh) | 2015-04-30 | 2016-04-27 | 多汗症治疗用外用剂 |
| ES16786516T ES2861052T3 (es) | 2015-04-30 | 2016-04-27 | Composición tópica que comprende oxibutinina para el tratamiento de la hiperhidrosis |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015-092946 | 2015-04-30 | ||
| JP2015092946 | 2015-04-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016175240A1 true WO2016175240A1 (ja) | 2016-11-03 |
Family
ID=57198447
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/063188 WO2016175240A1 (ja) | 2015-04-30 | 2016-04-27 | 多汗症治療用外用剤 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US10525132B2 (ja) |
| EP (1) | EP3290053B1 (ja) |
| JP (1) | JP6387458B2 (ja) |
| KR (1) | KR101937072B1 (ja) |
| CN (1) | CN107427582B (ja) |
| ES (1) | ES2861052T3 (ja) |
| TW (1) | TWI713521B (ja) |
| WO (1) | WO2016175240A1 (ja) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106137955B (zh) * | 2016-08-17 | 2021-10-29 | 杭州百诚医药科技股份有限公司 | 一种外用噻托溴铵制剂的止汗用途 |
| NL2023160B1 (en) * | 2018-05-17 | 2020-05-20 | Notoxins Ip B V | Aqueous formulations comprising oxybutynin for topical treatment of skin diseases |
| WO2019236429A1 (en) * | 2018-06-04 | 2019-12-12 | Nm Therapeutics, Llc | Method and apparatus for treatment of hyperhidrosis |
| EP4140476A1 (en) * | 2021-08-25 | 2023-03-01 | Health Innovation Technology Transfer, S.L. | An oil-in-water emulsion gel comprising tiotropium bromide |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995028914A1 (fr) * | 1994-04-21 | 1995-11-02 | Hisamitsu Pharmaceutical Co., Inc. | Composition de base administree par voie percutanee et composition medicamenteuse administree par voie percutanee obtenue a partir de cette derniere |
| WO2010062930A1 (en) * | 2008-11-26 | 2010-06-03 | Dry Pharma, Llc | Compositions and methods for hyperhidrosis |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4643018B2 (ja) | 1999-04-13 | 2011-03-02 | 久光製薬株式会社 | 経皮吸収型製剤 |
| US7029694B2 (en) * | 2000-04-26 | 2006-04-18 | Watson Laboratories, Inc. | Compositions and methods for transdermal oxybutynin therapy |
| AU2012216593B2 (en) | 2002-11-01 | 2014-09-25 | Allergan Sales, Llc | Compositions and methods for transdermal oxybutynin therapy |
| WO2007046102A2 (en) * | 2005-10-19 | 2007-04-26 | Menni Menashe Zinger | Methods for the treatment of hyperhidrosis |
| KR101317924B1 (ko) | 2011-05-17 | 2013-10-16 | 김동진 | 글리코피롤레이트의 합성방법 및 이 활성성분을 함유하는 약학적 조성물의 제조방법 |
| CN104053962B (zh) * | 2011-12-23 | 2016-06-22 | 菲弗斯有限公司 | 以吹冷空气的方式冷却回转炉开口的装置 |
| US20140037713A1 (en) | 2012-08-03 | 2014-02-06 | Antares Pharma Ipl, Ag | Transdermal compositions for anti-cholinergic agents |
| KR102241660B1 (ko) * | 2013-03-15 | 2021-04-19 | 보도르 라보래토리즈, 인크. | 다한증 치료용 항콜린성 글리코피롤레이트 에스테르 |
-
2016
- 2016-04-27 ES ES16786516T patent/ES2861052T3/es active Active
- 2016-04-27 EP EP16786516.1A patent/EP3290053B1/en active Active
- 2016-04-27 CN CN201680019209.XA patent/CN107427582B/zh active Active
- 2016-04-27 US US15/568,432 patent/US10525132B2/en active Active
- 2016-04-27 WO PCT/JP2016/063188 patent/WO2016175240A1/ja active Application Filing
- 2016-04-27 JP JP2017515577A patent/JP6387458B2/ja active Active
- 2016-04-27 KR KR1020177027272A patent/KR101937072B1/ko active IP Right Grant
- 2016-04-29 TW TW105113554A patent/TWI713521B/zh active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995028914A1 (fr) * | 1994-04-21 | 1995-11-02 | Hisamitsu Pharmaceutical Co., Inc. | Composition de base administree par voie percutanee et composition medicamenteuse administree par voie percutanee obtenue a partir de cette derniere |
| WO2010062930A1 (en) * | 2008-11-26 | 2010-06-03 | Dry Pharma, Llc | Compositions and methods for hyperhidrosis |
Non-Patent Citations (1)
| Title |
|---|
| DEL BOZ, J: "Systemic treatment ot hypernidrosis", ACTAS DERMO-SIFILIOGRAFICAS, vol. 106, no. 4, 6 April 2015 (2015-04-06), pages 271 - 277, XP029220272, ISSN: 0001-7310 * |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2016175240A1 (ja) | 2017-11-16 |
| KR20170120168A (ko) | 2017-10-30 |
| TW201703767A (zh) | 2017-02-01 |
| CN107427582A (zh) | 2017-12-01 |
| JP6387458B2 (ja) | 2018-09-05 |
| US10525132B2 (en) | 2020-01-07 |
| KR101937072B1 (ko) | 2019-01-09 |
| EP3290053A1 (en) | 2018-03-07 |
| US20180140704A1 (en) | 2018-05-24 |
| ES2861052T3 (es) | 2021-10-05 |
| TWI713521B (zh) | 2020-12-21 |
| CN107427582B (zh) | 2021-03-05 |
| EP3290053A4 (en) | 2019-01-09 |
| EP3290053B1 (en) | 2021-02-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6387458B2 (ja) | 多汗症治療用外用剤 | |
| JP6794420B2 (ja) | 外用医薬組成物 | |
| TW201517927A (zh) | 經改良的止汗組成物 | |
| JP2009542779A (ja) | 座瘡病変の治療のためのアダパレンと過酸化ベンゾイルとの組合せ | |
| WO2014123916A1 (en) | A liquid non-ionic salt-free skin and hair treatment composition | |
| KR20140055689A (ko) | 피부 자극이 저감된 고마쥬 타입 각질 제거용 화장료 조성물 | |
| JP2010184903A (ja) | 外用医薬組成物 | |
| JP6564927B2 (ja) | 外用液剤 | |
| JP2021004184A (ja) | アラントイン及び/又はその誘導体の安定化方法 | |
| CN112107526A (zh) | 一种修复皮肤屏障的婴童护肤品组合物及其制备方法 | |
| JP6512599B2 (ja) | 外用医薬組成物 | |
| JP3479048B2 (ja) | 化粧料及び不織布含浸化粧料 | |
| CA2998895A1 (en) | Topical antifungal compositions | |
| JP7356826B2 (ja) | 外用組成物 | |
| JP7467037B2 (ja) | 外用組成物 | |
| WO2022131081A1 (ja) | 外用乳化組成物 | |
| US20230078553A1 (en) | Ointments for treating dry skin | |
| JP2021187770A (ja) | 外用剤 | |
| EP0417245A1 (en) | Topical analgesic composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16786516 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017515577 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20177027272 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15568432 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2016786516 Country of ref document: EP |