WO2015056975A1 - 혼성 담지 메탈로센 촉매 - Google Patents
혼성 담지 메탈로센 촉매 Download PDFInfo
- Publication number
- WO2015056975A1 WO2015056975A1 PCT/KR2014/009681 KR2014009681W WO2015056975A1 WO 2015056975 A1 WO2015056975 A1 WO 2015056975A1 KR 2014009681 W KR2014009681 W KR 2014009681W WO 2015056975 A1 WO2015056975 A1 WO 2015056975A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- formula
- substituted
- aryl
- alkyl
- Prior art date
Links
- 239000012968 metallocene catalyst Substances 0.000 title claims abstract description 75
- 150000001875 compounds Chemical class 0.000 claims abstract description 109
- -1 4,5,6,7-tetrahydro-1-indenyl Chemical group 0.000 claims description 51
- 229910052739 hydrogen Inorganic materials 0.000 claims description 32
- 239000001257 hydrogen Substances 0.000 claims description 32
- 239000000126 substance Substances 0.000 claims description 26
- 150000002431 hydrogen Chemical class 0.000 claims description 21
- 125000002877 alkyl aryl group Chemical group 0.000 claims description 18
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 17
- 125000003342 alkenyl group Chemical group 0.000 claims description 15
- 229910052723 transition metal Inorganic materials 0.000 claims description 15
- 150000003624 transition metals Chemical class 0.000 claims description 15
- 125000005843 halogen group Chemical group 0.000 claims description 14
- 238000000034 method Methods 0.000 claims description 13
- 125000004432 carbon atom Chemical group C* 0.000 claims description 12
- 229910052736 halogen Inorganic materials 0.000 claims description 12
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 12
- 150000002367 halogens Chemical class 0.000 claims description 11
- 125000003545 alkoxy group Chemical group 0.000 claims description 10
- 229930195733 hydrocarbon Natural products 0.000 claims description 10
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 claims description 9
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 9
- 125000003277 amino group Chemical group 0.000 claims description 9
- 125000002102 aryl alkyloxo group Chemical group 0.000 claims description 9
- 125000004104 aryloxy group Chemical group 0.000 claims description 9
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 claims description 9
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 claims description 9
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 claims description 9
- 229910052710 silicon Inorganic materials 0.000 claims description 9
- 239000010703 silicon Chemical group 0.000 claims description 9
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 8
- 125000005018 aryl alkenyl group Chemical group 0.000 claims description 8
- 125000003118 aryl group Chemical group 0.000 claims description 8
- 229910052799 carbon Inorganic materials 0.000 claims description 8
- 229910052732 germanium Chemical group 0.000 claims description 8
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 8
- 125000000304 alkynyl group Chemical group 0.000 claims description 7
- 125000001183 hydrocarbyl group Chemical group 0.000 claims description 7
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims description 6
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 6
- 229910052698 phosphorus Inorganic materials 0.000 claims description 6
- 239000011574 phosphorus Substances 0.000 claims description 6
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 5
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 5
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 5
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 5
- 125000002947 alkylene group Chemical group 0.000 claims description 4
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical group [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 claims description 4
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 3
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 3
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 claims description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 3
- 125000000547 substituted alkyl group Chemical group 0.000 claims description 3
- 125000003107 substituted aryl group Chemical group 0.000 claims description 3
- 229910052717 sulfur Inorganic materials 0.000 claims description 3
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 claims description 3
- 125000001412 tetrahydropyranyl group Chemical group 0.000 claims description 3
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims description 2
- 125000001931 aliphatic group Chemical group 0.000 claims description 2
- 150000004703 alkoxides Chemical group 0.000 claims description 2
- 229910052796 boron Inorganic materials 0.000 claims description 2
- AIXAANGOTKPUOY-UHFFFAOYSA-N carbachol Chemical group [Cl-].C[N+](C)(C)CCOC(N)=O AIXAANGOTKPUOY-UHFFFAOYSA-N 0.000 claims description 2
- 150000001793 charged compounds Polymers 0.000 claims description 2
- ZZUFCTLCJUWOSV-UHFFFAOYSA-N furosemide Chemical compound C1=C(Cl)C(S(=O)(=O)N)=CC(C(O)=O)=C1NCC1=CC=CO1 ZZUFCTLCJUWOSV-UHFFFAOYSA-N 0.000 claims description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 2
- 230000003647 oxidation Effects 0.000 claims description 2
- 238000007254 oxidation reaction Methods 0.000 claims description 2
- 229910052760 oxygen Inorganic materials 0.000 claims description 2
- 125000002306 tributylsilyl group Chemical group C(CCC)[Si](CCCC)(CCCC)* 0.000 claims description 2
- 125000000025 triisopropylsilyl group Chemical group C(C)(C)[Si](C(C)C)(C(C)C)* 0.000 claims description 2
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 2
- 125000006832 (C1-C10) alkylene group Chemical group 0.000 claims 1
- 125000004183 alkoxy alkyl group Chemical group 0.000 claims 1
- 239000003426 co-catalyst Substances 0.000 claims 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 claims 1
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 claims 1
- 229920000642 polymer Polymers 0.000 abstract description 26
- 230000000694 effects Effects 0.000 abstract description 16
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 abstract description 6
- 150000001336 alkenes Chemical class 0.000 abstract description 4
- 230000001747 exhibiting effect Effects 0.000 abstract description 2
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 60
- 238000006243 chemical reaction Methods 0.000 description 39
- 239000003054 catalyst Substances 0.000 description 38
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 30
- 238000002360 preparation method Methods 0.000 description 28
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 25
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 20
- 239000003446 ligand Substances 0.000 description 16
- 238000003756 stirring Methods 0.000 description 16
- 238000009826 distribution Methods 0.000 description 15
- LWNGJAHMBMVCJR-UHFFFAOYSA-N (2,3,4,5,6-pentafluorophenoxy)boronic acid Chemical compound OB(O)OC1=C(F)C(F)=C(F)C(F)=C1F LWNGJAHMBMVCJR-UHFFFAOYSA-N 0.000 description 13
- 230000000052 comparative effect Effects 0.000 description 13
- 238000006116 polymerization reaction Methods 0.000 description 12
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 10
- DLEDOFVPSDKWEF-UHFFFAOYSA-N lithium butane Chemical compound [Li+].CCC[CH2-] DLEDOFVPSDKWEF-UHFFFAOYSA-N 0.000 description 10
- 150000007523 nucleic acids Chemical class 0.000 description 10
- 102000039446 nucleic acids Human genes 0.000 description 10
- 108020004707 nucleic acids Proteins 0.000 description 10
- 229920000098 polyolefin Polymers 0.000 description 10
- 239000002002 slurry Substances 0.000 description 10
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 9
- 235000011089 carbon dioxide Nutrition 0.000 description 9
- 239000000178 monomer Substances 0.000 description 9
- MZRVEZGGRBJDDB-UHFFFAOYSA-N n-Butyllithium Substances [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 9
- 238000005160 1H NMR spectroscopy Methods 0.000 description 8
- 238000005481 NMR spectroscopy Methods 0.000 description 8
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 8
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 8
- RXXXUIOZOITBII-UHFFFAOYSA-N indeno[1,2-g]indole Chemical class C1=C2C=CC=CC2=C2C1=C1N=CC=C1C=C2 RXXXUIOZOITBII-UHFFFAOYSA-N 0.000 description 8
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 7
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 7
- 150000002430 hydrocarbons Chemical class 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- 125000000217 alkyl group Chemical group 0.000 description 6
- 238000001035 drying Methods 0.000 description 6
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 description 6
- 125000003983 fluorenyl group Chemical class C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 6
- CPOFMOWDMVWCLF-UHFFFAOYSA-N methyl(oxo)alumane Chemical compound C[Al]=O CPOFMOWDMVWCLF-UHFFFAOYSA-N 0.000 description 6
- 230000037048 polymerization activity Effects 0.000 description 6
- 239000004215 Carbon black (E152) Substances 0.000 description 5
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 5
- 239000005977 Ethylene Substances 0.000 description 5
- 229910007926 ZrCl Inorganic materials 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 5
- 239000002184 metal Substances 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- 230000000704 physical effect Effects 0.000 description 5
- 150000003254 radicals Chemical class 0.000 description 5
- RURFJXKOXIWFJX-UHFFFAOYSA-N (2,3,4,6-tetrafluorophenoxy)boronic acid Chemical compound OB(O)OC1=C(F)C=C(F)C(F)=C1F RURFJXKOXIWFJX-UHFFFAOYSA-N 0.000 description 4
- KWKAKUADMBZCLK-UHFFFAOYSA-N 1-octene Chemical compound CCCCCCC=C KWKAKUADMBZCLK-UHFFFAOYSA-N 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 4
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 4
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 4
- 229910052782 aluminium Inorganic materials 0.000 description 4
- VGRFVJMYCCLWPQ-UHFFFAOYSA-N germanium Chemical compound [Ge].[Ge] VGRFVJMYCCLWPQ-UHFFFAOYSA-N 0.000 description 4
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 239000012044 organic layer Substances 0.000 description 4
- YWAKXRMUMFPDSH-UHFFFAOYSA-N pentene Chemical compound CCCC=C YWAKXRMUMFPDSH-UHFFFAOYSA-N 0.000 description 4
- 239000000377 silicon dioxide Substances 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- VUGMARFZKDASCX-UHFFFAOYSA-N 2-methyl-N-silylpropan-2-amine Chemical compound CC(C)(C)N[SiH3] VUGMARFZKDASCX-UHFFFAOYSA-N 0.000 description 3
- 101150041968 CDC13 gene Proteins 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 3
- 239000011954 Ziegler–Natta catalyst Substances 0.000 description 3
- 229920001577 copolymer Polymers 0.000 description 3
- MROCJMGDEKINLD-UHFFFAOYSA-N dichlorosilane Chemical compound Cl[SiH2]Cl MROCJMGDEKINLD-UHFFFAOYSA-N 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 229910052709 silver Inorganic materials 0.000 description 3
- 239000004332 silver Substances 0.000 description 3
- 239000010936 titanium Substances 0.000 description 3
- AEYIEUDRIHOMGO-UHFFFAOYSA-N (1,4,5,5,6,6-hexafluorocyclohex-2-en-1-yl)oxyboronic acid Chemical compound B(O)(O)OC1(C=CC(C(C1(F)F)(F)F)F)F AEYIEUDRIHOMGO-UHFFFAOYSA-N 0.000 description 2
- MYRTYDVEIRVNKP-UHFFFAOYSA-N 1,2-Divinylbenzene Chemical compound C=CC1=CC=CC=C1C=C MYRTYDVEIRVNKP-UHFFFAOYSA-N 0.000 description 2
- ZGEGCLOFRBLKSE-UHFFFAOYSA-N 1-Heptene Chemical compound CCCCCC=C ZGEGCLOFRBLKSE-UHFFFAOYSA-N 0.000 description 2
- AFFLGGQVNFXPEV-UHFFFAOYSA-N 1-decene Chemical compound CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 2
- CRSBERNSMYQZNG-UHFFFAOYSA-N 1-dodecene Chemical compound CCCCCCCCCCC=C CRSBERNSMYQZNG-UHFFFAOYSA-N 0.000 description 2
- HFDVRLIODXPAHB-UHFFFAOYSA-N 1-tetradecene Chemical compound CCCCCCCCCCCCC=C HFDVRLIODXPAHB-UHFFFAOYSA-N 0.000 description 2
- DCTOHCCUXLBQMS-UHFFFAOYSA-N 1-undecene Chemical compound CCCCCCCCCC=C DCTOHCCUXLBQMS-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- 125000005865 C2-C10alkynyl group Chemical group 0.000 description 2
- 239000002841 Lewis acid Substances 0.000 description 2
- 239000002879 Lewis base Substances 0.000 description 2
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- NIPNSKYNPDTRPC-UHFFFAOYSA-N N-[2-oxo-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 NIPNSKYNPDTRPC-UHFFFAOYSA-N 0.000 description 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 150000003863 ammonium salts Chemical class 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 125000005131 dialkylammonium group Chemical group 0.000 description 2
- WFZSIVNWLIYDJZ-UHFFFAOYSA-N dichloro-methyl-[6-[(2-methylpropan-2-yl)oxy]hexyl]silane Chemical compound CC(C)(C)OCCCCCC[Si](C)(Cl)Cl WFZSIVNWLIYDJZ-UHFFFAOYSA-N 0.000 description 2
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 150000004820 halides Chemical group 0.000 description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 2
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 2
- 150000007517 lewis acids Chemical class 0.000 description 2
- 150000007527 lewis bases Chemical class 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910001629 magnesium chloride Inorganic materials 0.000 description 2
- 125000002950 monocyclic group Chemical group 0.000 description 2
- TVMXDCGIABBOFY-UHFFFAOYSA-N n-Octanol Natural products CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 2
- 150000002902 organometallic compounds Chemical class 0.000 description 2
- 150000004714 phosphonium salts Chemical class 0.000 description 2
- 239000002685 polymerization catalyst Substances 0.000 description 2
- 230000000379 polymerizing effect Effects 0.000 description 2
- 229920005672 polyolefin resin Polymers 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 125000005373 siloxane group Chemical group [SiH2](O*)* 0.000 description 2
- 125000003698 tetramethyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 150000003623 transition metal compounds Chemical class 0.000 description 2
- ZMANZCXQSJIPKH-UHFFFAOYSA-O triethylammonium ion Chemical compound CC[NH+](CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-O 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- YFTHZRPMJXBUME-UHFFFAOYSA-N tripropylamine Chemical compound CCCN(CCC)CCC YFTHZRPMJXBUME-UHFFFAOYSA-N 0.000 description 2
- 238000001291 vacuum drying Methods 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- WCFQIFDACWBNJT-UHFFFAOYSA-N $l^{1}-alumanyloxy(2-methylpropyl)aluminum Chemical compound CC(C)C[Al]O[Al] WCFQIFDACWBNJT-UHFFFAOYSA-N 0.000 description 1
- OJOWICOBYCXEKR-KRXBUXKQSA-N (5e)-5-ethylidenebicyclo[2.2.1]hept-2-ene Chemical compound C1C2C(=C/C)/CC1C=C2 OJOWICOBYCXEKR-KRXBUXKQSA-N 0.000 description 1
- VNPQQEYMXYCAEZ-UHFFFAOYSA-N 1,2,3,4-tetramethylcyclopenta-1,3-diene Chemical compound CC1=C(C)C(C)=C(C)C1 VNPQQEYMXYCAEZ-UHFFFAOYSA-N 0.000 description 1
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical group C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 1
- OHVLMTFVQDZYHP-UHFFFAOYSA-N 1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)-2-[4-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidin-5-yl]piperazin-1-yl]ethanone Chemical compound N1N=NC=2CN(CCC=21)C(CN1CCN(CC1)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)=O OHVLMTFVQDZYHP-UHFFFAOYSA-N 0.000 description 1
- HMDQPBSDHHTRNI-UHFFFAOYSA-N 1-(chloromethyl)-3-ethenylbenzene Chemical compound ClCC1=CC=CC(C=C)=C1 HMDQPBSDHHTRNI-UHFFFAOYSA-N 0.000 description 1
- HMUNWXXNJPVALC-UHFFFAOYSA-N 1-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)C(CN1CC2=C(CC1)NN=N2)=O HMUNWXXNJPVALC-UHFFFAOYSA-N 0.000 description 1
- HECLRDQVFMWTQS-RGOKHQFPSA-N 1755-01-7 Chemical compound C1[C@H]2[C@@H]3CC=C[C@@H]3[C@@H]1C=C2 HECLRDQVFMWTQS-RGOKHQFPSA-N 0.000 description 1
- VZSRBBMJRBPUNF-UHFFFAOYSA-N 2-(2,3-dihydro-1H-inden-2-ylamino)-N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]pyrimidine-5-carboxamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C(=O)NCCC(N1CC2=C(CC1)NN=N2)=O VZSRBBMJRBPUNF-UHFFFAOYSA-N 0.000 description 1
- JQMFQLVAJGZSQS-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]-N-(2-oxo-3H-1,3-benzoxazol-6-yl)acetamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)CC(=O)NC1=CC2=C(NC(O2)=O)C=C1 JQMFQLVAJGZSQS-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- YLZOPXRUQYQQID-UHFFFAOYSA-N 3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)-1-[4-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidin-5-yl]piperazin-1-yl]propan-1-one Chemical compound N1N=NC=2CN(CCC=21)CCC(=O)N1CCN(CC1)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F YLZOPXRUQYQQID-UHFFFAOYSA-N 0.000 description 1
- UDMMZSJNHAWYKX-UHFFFAOYSA-N 4-phenylbicyclo[2.2.1]hept-2-ene Chemical compound C1C(C=C2)CCC21C1=CC=CC=C1 UDMMZSJNHAWYKX-UHFFFAOYSA-N 0.000 description 1
- INYHZQLKOKTDAI-UHFFFAOYSA-N 5-ethenylbicyclo[2.2.1]hept-2-ene Chemical compound C1C2C(C=C)CC1C=C2 INYHZQLKOKTDAI-UHFFFAOYSA-N 0.000 description 1
- CONKBQPVFMXDOV-QHCPKHFHSA-N 6-[(5S)-5-[[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]methyl]-2-oxo-1,3-oxazolidin-3-yl]-3H-1,3-benzoxazol-2-one Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)C[C@H]1CN(C(O1)=O)C1=CC2=C(NC(O2)=O)C=C1 CONKBQPVFMXDOV-QHCPKHFHSA-N 0.000 description 1
- JNTPTNNCGDAGEJ-UHFFFAOYSA-N 6-chlorohexan-1-ol Chemical compound OCCCCCCCl JNTPTNNCGDAGEJ-UHFFFAOYSA-N 0.000 description 1
- 229910000897 Babbitt (metal) Inorganic materials 0.000 description 1
- 208000033242 Blomstrand lethal chondrodysplasia Diseases 0.000 description 1
- 229920000089 Cyclic olefin copolymer Polymers 0.000 description 1
- 239000007818 Grignard reagent Substances 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 102100030326 Serpin B4 Human genes 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical group 0.000 description 1
- 125000005234 alkyl aluminium group Chemical group 0.000 description 1
- XYLMUPLGERFSHI-UHFFFAOYSA-N alpha-Methylstyrene Chemical compound CC(=C)C1=CC=CC=C1 XYLMUPLGERFSHI-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000002902 bimodal effect Effects 0.000 description 1
- 125000006267 biphenyl group Chemical group 0.000 description 1
- GHXRKGHKMRZBJH-UHFFFAOYSA-N boric acid Chemical class OB(O)O.OB(O)O GHXRKGHKMRZBJH-UHFFFAOYSA-N 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 1
- 229960004424 carbon dioxide Drugs 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 201000003766 chondrodysplasia Blomstrand type Diseases 0.000 description 1
- 238000010960 commercial process Methods 0.000 description 1
- 238000007334 copolymerization reaction Methods 0.000 description 1
- DIOQZVSQGTUSAI-UHFFFAOYSA-N decane Chemical compound CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 125000003963 dichloro group Chemical group Cl* 0.000 description 1
- 229960004132 diethyl ether Drugs 0.000 description 1
- SMBQBQBNOXIFSF-UHFFFAOYSA-N dilithium Chemical class [Li][Li] SMBQBQBNOXIFSF-UHFFFAOYSA-N 0.000 description 1
- JLTDJTHDQAWBAV-UHFFFAOYSA-O dimethyl(phenyl)azanium Chemical compound C[NH+](C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-O 0.000 description 1
- HKUFIYBZNQSHQS-UHFFFAOYSA-O dioctadecylazanium Chemical compound CCCCCCCCCCCCCCCCCC[NH2+]CCCCCCCCCCCCCCCCCC HKUFIYBZNQSHQS-UHFFFAOYSA-O 0.000 description 1
- 229940069096 dodecene Drugs 0.000 description 1
- 230000003028 elevating effect Effects 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 150000004795 grignard reagents Chemical class 0.000 description 1
- 229910021482 group 13 metal Inorganic materials 0.000 description 1
- 229910052735 hafnium Inorganic materials 0.000 description 1
- VBJZVLUMGGDVMO-UHFFFAOYSA-N hafnium atom Chemical compound [Hf] VBJZVLUMGGDVMO-UHFFFAOYSA-N 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- 229920006158 high molecular weight polymer Polymers 0.000 description 1
- 150000004678 hydrides Chemical group 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- 108010013554 leupin Proteins 0.000 description 1
- 239000011344 liquid material Substances 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- 229910001092 metal group alloy Inorganic materials 0.000 description 1
- 238000006263 metalation reaction Methods 0.000 description 1
- CAKOOJUEUVRZLR-UHFFFAOYSA-N methyl(dioctadecyl)phosphane Chemical compound CCCCCCCCCCCCCCCCCCP(C)CCCCCCCCCCCCCCCCCC CAKOOJUEUVRZLR-UHFFFAOYSA-N 0.000 description 1
- OXQMIXBVXHWDPX-UHFFFAOYSA-N n,n,2-trimethylpropan-2-amine Chemical compound CN(C)C(C)(C)C OXQMIXBVXHWDPX-UHFFFAOYSA-N 0.000 description 1
- SRLHDBRENZFCIN-UHFFFAOYSA-N n,n-di(butan-2-yl)butan-2-amine Chemical compound CCC(C)N(C(C)CC)C(C)CC SRLHDBRENZFCIN-UHFFFAOYSA-N 0.000 description 1
- VFLWKHBYVIUAMP-UHFFFAOYSA-N n-methyl-n-octadecyloctadecan-1-amine Chemical compound CCCCCCCCCCCCCCCCCCN(C)CCCCCCCCCCCCCCCCCC VFLWKHBYVIUAMP-UHFFFAOYSA-N 0.000 description 1
- KUFYUMSBZMUWAN-UHFFFAOYSA-N n-methyl-n-tetradecyltetradecan-1-amine Chemical compound CCCCCCCCCCCCCCN(C)CCCCCCCCCCCCCC KUFYUMSBZMUWAN-UHFFFAOYSA-N 0.000 description 1
- HSUGDXPUFCVGES-UHFFFAOYSA-N n-tetradecyltetradecan-1-amine Chemical compound CCCCCCCCCCCCCCNCCCCCCCCCCCCCC HSUGDXPUFCVGES-UHFFFAOYSA-N 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- ZCYXXKJEDCHMGH-UHFFFAOYSA-N nonane Chemical compound CCCC[CH]CCCC ZCYXXKJEDCHMGH-UHFFFAOYSA-N 0.000 description 1
- JFNLZVQOOSMTJK-KNVOCYPGSA-N norbornene Chemical compound C1[C@@H]2CC[C@H]1C=C2 JFNLZVQOOSMTJK-KNVOCYPGSA-N 0.000 description 1
- BKIMMITUMNQMOS-UHFFFAOYSA-N normal nonane Natural products CCCCCCCCC BKIMMITUMNQMOS-UHFFFAOYSA-N 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 125000001181 organosilyl group Chemical group [SiH3]* 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 125000000538 pentafluorophenyl group Chemical group FC1=C(F)C(F)=C(*)C(F)=C1F 0.000 description 1
- JQQSUOJIMKJQHS-UHFFFAOYSA-N pentaphenyl group Chemical group C1=CC=CC2=CC3=CC=C4C=C5C=CC=CC5=CC4=C3C=C12 JQQSUOJIMKJQHS-UHFFFAOYSA-N 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 description 1
- PAYRUJLWNCNPSJ-UHFFFAOYSA-O phenylazanium Chemical compound [NH3+]C1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-O 0.000 description 1
- XYFCBTPGUUZFHI-UHFFFAOYSA-O phosphonium Chemical compound [PH4+] XYFCBTPGUUZFHI-UHFFFAOYSA-O 0.000 description 1
- 239000002574 poison Substances 0.000 description 1
- 231100000614 poison Toxicity 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- VSZWPYCFIRKVQL-UHFFFAOYSA-N selanylidenegallium;selenium Chemical compound [Se].[Se]=[Ga].[Se]=[Ga] VSZWPYCFIRKVQL-UHFFFAOYSA-N 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 238000000527 sonication Methods 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- VOITXYVAKOUIBA-UHFFFAOYSA-N triethylaluminium Chemical compound CC[Al](CC)CC VOITXYVAKOUIBA-UHFFFAOYSA-N 0.000 description 1
- MDCWDBMBZLORER-UHFFFAOYSA-N triphenyl borate Chemical compound C=1C=CC=CC=1OB(OC=1C=CC=CC=1)OC1=CC=CC=C1 MDCWDBMBZLORER-UHFFFAOYSA-N 0.000 description 1
- RIOQSEWOXXDEQQ-UHFFFAOYSA-O triphenylphosphanium Chemical compound C1=CC=CC=C1[PH+](C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-O 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 239000003039 volatile agent Substances 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
- 239000004711 α-olefin Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/6592—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring
- C08F4/65922—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not
- C08F4/65927—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not two cyclopentadienyl rings being mutually bridged
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2282—Unsaturated compounds used as ligands
- B01J31/2295—Cyclic compounds, e.g. cyclopentadienyls
Definitions
- the present invention relates to a common supported metallocene catalyst. More specifically, the present invention relates to a common supported metallocene catalyst that can be used for preparing an olepin-based polymer.
- the present application is directed to Korean Patent Application Nos. 10-2013-0124518 and 2014, filed with the Korean Patent Office on October 18, 2013. Claims the benefit of the filing date of Korean Patent Application No. 10-2014-0138348 filed with the Korea Intellectual Property Office on October 14, the entire contents of which are incorporated herein.
- the metallocene catalyst is composed of a combination of a main catalyst composed mainly of transition metal compounds and a cocatalyst composed of organometallic compounds composed mainly of aluminum.
- a catalyst is a homogeneous complex catalyst and is a single site catalyst.
- the polymer has a narrow molecular weight distribution according to the characteristics of a single active site and a homogeneous composition of the comonomer, and the stereoregularity, copolymerization characteristics, molecular weight, It has the property to change the crystallinity.
- U.S. Patent 5,0 2; 562 describes a process for preparing a polymerization catalyst by supporting two different transition metal catalysts on one supported catalyst. It is a method of producing a bimodal distribution polymer by supporting a titanium (Ti) -based Ziegler-Natta catalyst generating high molecular weight and a zirconium (Zr) -based metallocene catalyst generating low molecular weight on one support. As a result, the supporting process is complicated and the morphology of the polymer is degraded by the promoter.
- Ti titanium
- Zr zirconium
- U.S. Patent 5,525,678 discloses metallocene compounds and nonmetallocene compounds. It describes a method of using a catalyst system for olefinic polymerization in which a high molecular weight polymer and a low molecular weight polymer can be simultaneously polymerized by simultaneously supporting on a carrier. This has the disadvantage that the metallocene compound and the non-metallocene compound must be separately supported, and the carrier must be pretreated with various compounds for supporting reaction.
- Patent No. 5,914,289 describes a method for controlling the molecular weight and molecular weight distribution of a polymer by using a metallocene catalyst supported on each carrier, but the amount of solvent used and the time required for preparing the supported catalyst are high. The hassle of having to support each of the metallocene catalysts to be used on a carrier was followed.
- Korean Patent Application No. 2003-12308 discloses a method of controlling the molecular weight distribution by loading a binuclear metallocene catalyst and a mononuclear metallocene catalyst on a carrier together with an activator to change the combination of the catalysts in the reactor and integrating them. have.
- this method is limited in realizing the characteristics of each catalyst at the same time, and also has a disadvantage in that the metallocene catalyst portion is liberated in the carrier component of the finished catalyst, causing fouling.
- the present 'invention is to provide a supported metallocene catalyst heunseong metal capable of producing an olefin polymer having an increased activity is excellent and the high molecular weight and desired physical properties.
- the present invention includes at least one first metallocene compound represented by Formula 1, at least one second metallocene compound selected from compounds represented by Formulas 3 to 5, a promoter compound and a carrier Provided is a commonly supported metallocene catalyst.
- the common supported metallocene catalyst according to the present invention comprises two different kinds of metallocene compounds of ⁇ " , in particular, one metallocene compound is an indeno indole derivative and / or fluorene (The fluorene) derivative exhibits high polymerization activity even when supported by using a ligand compound having a crosslinked structure formed by a bridge, and has excellent activity and can be used for polymerization of an ultrahigh molecular weight olefin polymer.
- the common .supported metallocene catalyst according to the present invention is at least one ' first metallocene compound represented by formula 1, and a second metallocene compound selected from compounds represented by formulas 3 to 5 As mentioned above, it contains a promoter compound and a support
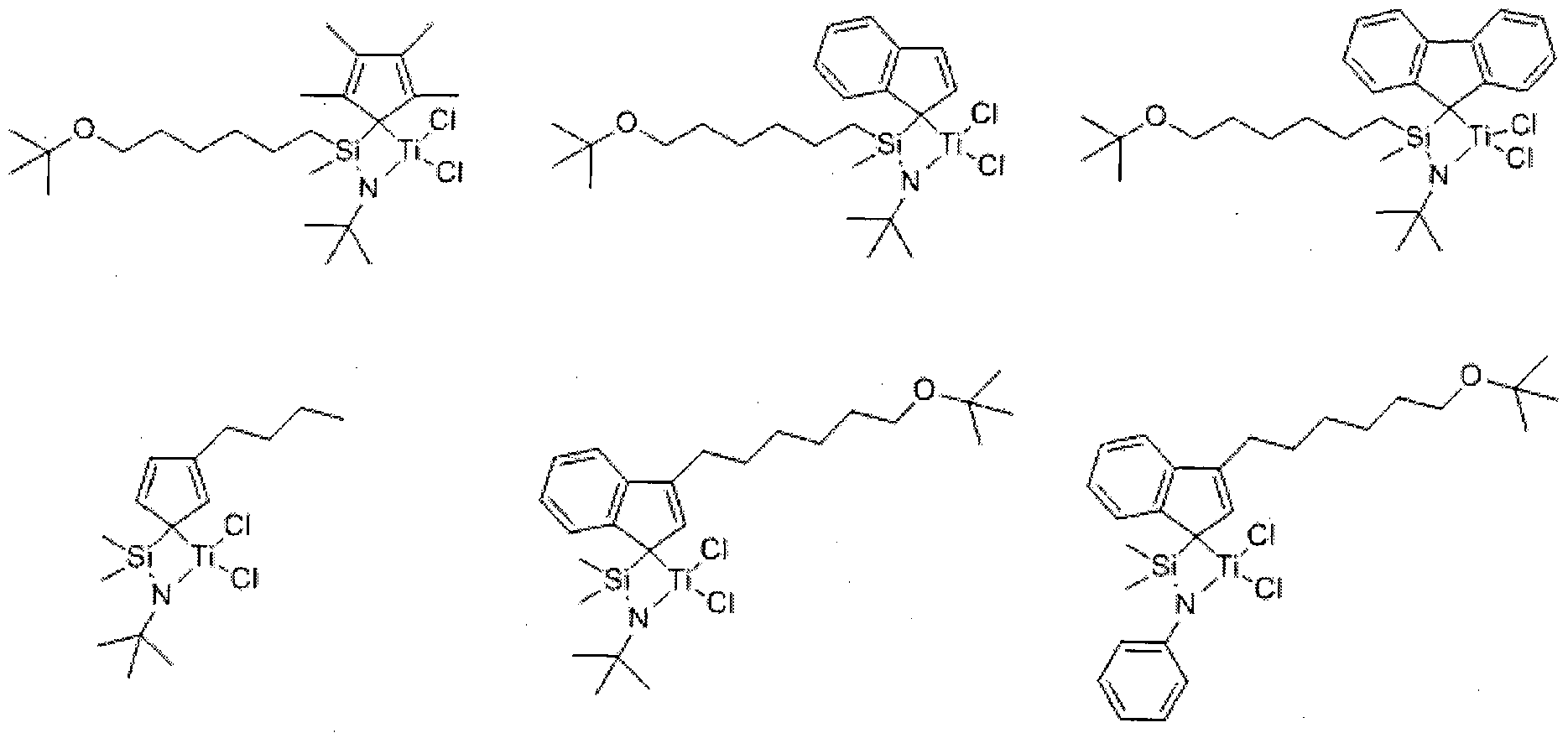
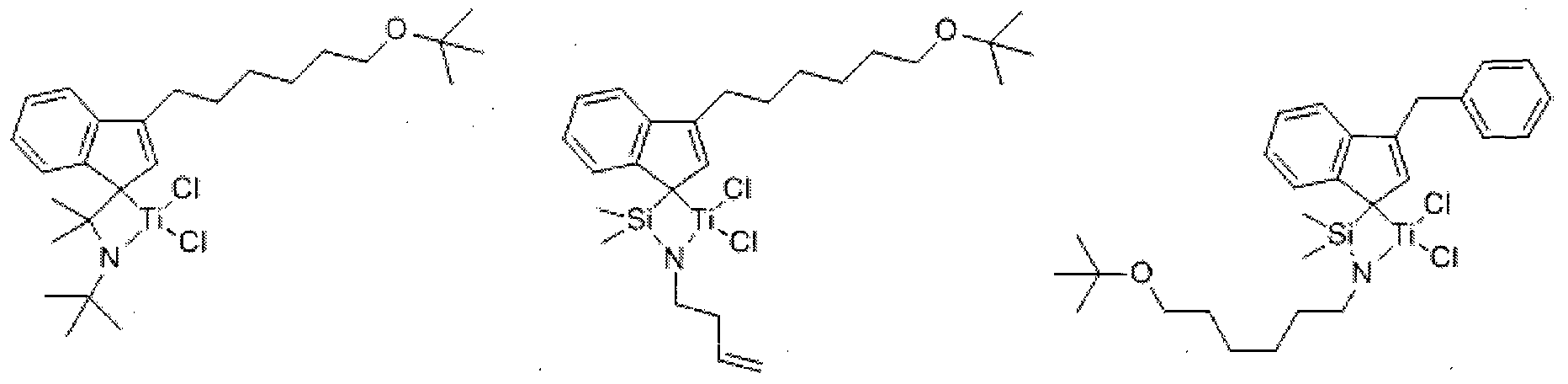
- Chemical Formula 1 A is hydrogen, halogen, C1 to C20 alkyl group, C2 to C20 alkenyl group, C6 to C20 aryl group, C7 to C20 alkylaryl group, C7 to C20 arylalkyl group, C1 to C20 alkoxy group, C2 to C20 A C20 alkoxyalkyl group, a C3 to C20 heterocycloalkyl group, or a C5 to C20 heteroaryl group;
- D is-, -S-, -N (R)-or -Si (R) (R ')-, wherein R and R' are the same or different from each other, and are each independently hydrogen, halogen, C1 to C20 An alkyl group of C2, an alkenyl group of C2 to C20, or an aryl group of C6 to C20;
- L is a C1 to C10 straight or branched chain alkylene group
- B is carbon, silicon or germanium
- Q is hydrogen, halogen, C1 to C20 alkyl group, C2 to C20 alkenyl group, C6 to C20 aryl group, C7 to C20 al3 ⁇ 4aryl group, or C7 to C20 arylalkyl group;
- M is a Group 4 transition metal
- X 1 and X 2 are the same as or different from each other, and each independently halogen, C1 to C20 alkyl group, C2 to C20 alkenyl group, C6 to C20 aryl group, nitro group, amido group, C1 to C20 alkylsilyl group , A C1 to C20 alkoxy group, or a C1 to C20 sulfonate group;
- C 1 and C 2 are the same as or different from each other, and are each independently represented by one of the following Chemical Formula 2a, Chemical Formula 2b, or Chemical Formula 2c, except that C 1 and C 2 are both Chemical Formula 2c;
- R1 to R17 and R1 'to R9' are the same as or different from each other, and each independently hydrogen, halogen, C1 to C20 alkyl group, C2 to C20 alkenyl group, C1 to C20 alkyl Silyl group, C1 to C20 silylalkyl group, C1 to C20 alkoxysilyl group, C1 to C20 alkoxy group, C6 to C20 aryl group, C7 to C20 alkylaryl group, or C7 to C20 arylalkyl group, Two or more adjacent to each other of R10 to R17 may be linked to each other to form a substituted or unsubstituted aliphatic or aromatic ring;
- M 1 is a Group 4 transition metal
- Cp 1 and Cp 2 are the same as or different from each other, and are each independently selected from the group consisting of cyclopentadienyl, indenyl, 4,5,6,7-tetrahydro-1-indenyl, and fluorenyl radicals One, they may be substituted with a hydrocarbon of 1 to 20 carbon atoms;
- R a and R b are the same as or different from each other, and each independently hydrogen, C1 to C20 alkyl, C1 to C10 alkoxy, C2 to C20 alkoxyalkyl, C6 to C20 aryl, C6 to C10 aryloxy, C2 Alkenyl from C20, alkylaryl from C7 to C40, C7-C40 arylalkyl, C8-C40 arylalkenyl, or C2-C10 alkynyl;
- ⁇ is a halogen atom, ci to C20 alkyl, C2 to C10 alkenyl, C7 to C40 alkylaryl, C7 to C40 arylalkyl, C6 to C20 aryl, substituted or unsubstituted C1 to C20 alkylidene, Substituted or unsubstituted amino group, C2 to C20 alkylalkoxy, or C7 to C40 arylalkoxy;
- n 1 or 0;
- M 2 is a Group 4 transition metal
- Cp 3 and Cp 4 are the same as or different from each other, and are each independently selected from the group consisting of cyclopentadienyl, indenyl, 4,5,6,7-tetrahydro-1-indenyl and polorerenyl radicals They may be substituted with a hydrocarbon having 1 to 20 carbon atoms;
- R c and R d are the same as or different from each other, and each independently hydrogen, C1 to C20 alkyl, C1 to C10 alkoxy, C2 to C20 alkoxyalkyl, C6 to C20 aryl, C6 to C10 aryloxy, C2 Alkenyl to C20, alkylaryl of C7 to C40, arylalkyl of C7 to C40, arylalkenyl of C8 to C40, or alkynyl of C2 to C10;
- Z 2 is a halogen atom, C1 to C20 alkyl, C2 to C10 alkenyl, C7 to C40 alkylaryl, C7 to C40 arylalkyl, C6 to C20 aryl, substituted or unsubstituted C1 to C20 alkylidene , Substituted or unsubstituted amino group, C2 to C20 alkylalkoxy, or C7 to C40 arylalkoxy;
- B 1 cross-links the Cp 3 R c ring and the Cp 4 R d ring, or one Cp 4 R d ring
- n 1 or 0;
- M 3 is a Group 4 transition metal
- Cp 5 is any one selected from the group consisting of cyclopentadienyl, indenyl, 4,5,6,7-tetrahydro-1-indenyl fluorenyl radicals, which are to be substituted with hydrocarbons having 1 to 20 carbon atoms Can be;
- R e is hydrogen, C1 to C20 alkyl, C1 to C10 alkoxy, C2 to C20 alkoxyalkyl, C6 to C20 aryl, C6 to C10 aryloxy, C2 to C20 alkenyl, C7 to C40 alkylaryl C7 to C40 arylalkyl, C8 to C40 arylalkenyl, or C2 to C10 alkynyl;
- Z 3 is a halogen atom, C 1 to C 20 alkyl, C 2 to C 10 alkenyl, C 7 to
- C40 alkylaryl C7 to C40 arylalkyl, C6 to C20 aryl, substituted or unsubstituted C1 to C20 alkylidene, substituted or unsubstituted amino group, C2 to C20 alkylalkoxy, or C7 to C40 aryl Alkoxy;
- B 2 is at least one or a combination of carbon, germanium, silicon, phosphorus or nitrogen atom containing radicals which crosslink the Cp 5 R e ring and J;
- J is any one selected from the group consisting of NR f , 0, PR f and S, wherein R f is C1 to C20 alkyl, aryl, substituted alkyl or substituted aryl.
- the alkyl group of C1 to C20 includes a linear or branched alkyl group, and specifically, methyl group, ethyl group, propyl group, isopropyl group, n-butyl group, tert-butyl group, pentyl group, nuclear group, heptyl group, Octyl group etc. can be mentioned, It is not limited to this.
- the alkenyl group of C2 to C20 includes a straight or branched alkenyl group, and specifically, may include an allyl group, ethenyl group, propenyl group, butenyl group, pentenyl group, and the like, but is not limited thereto.
- the C6 to C20 aryl groups include monocyclic or condensed aryl groups, and specifically include phenyl groups, biphenyl groups, naphthyl groups, phenanthrenyl groups, and fluorenyl groups, but are not limited thereto.
- the C5 to C20 heteroaryl group includes a monocyclic or condensed heteroaryl group, carbazolyl group, pyridyl group, quinoline group, isoquinoline group, Thiophenyl group, furanyl group, imidazole group, oxazolyl group, thiazolyl group, triazine group, tetrahydropyranyl group, tetrahydrofuranyl group, etc. are mentioned, It is not limited to this.
- alkoxy group for C1 to C20 examples include a methoxy group, an ethoxy group, a phenyloxy group, a cyclonuxyloxy group, and the like, but are not limited thereto.
- Group 4 transition metal examples include titanium, zirconium, hafnium, and the like, but are not limited thereto.
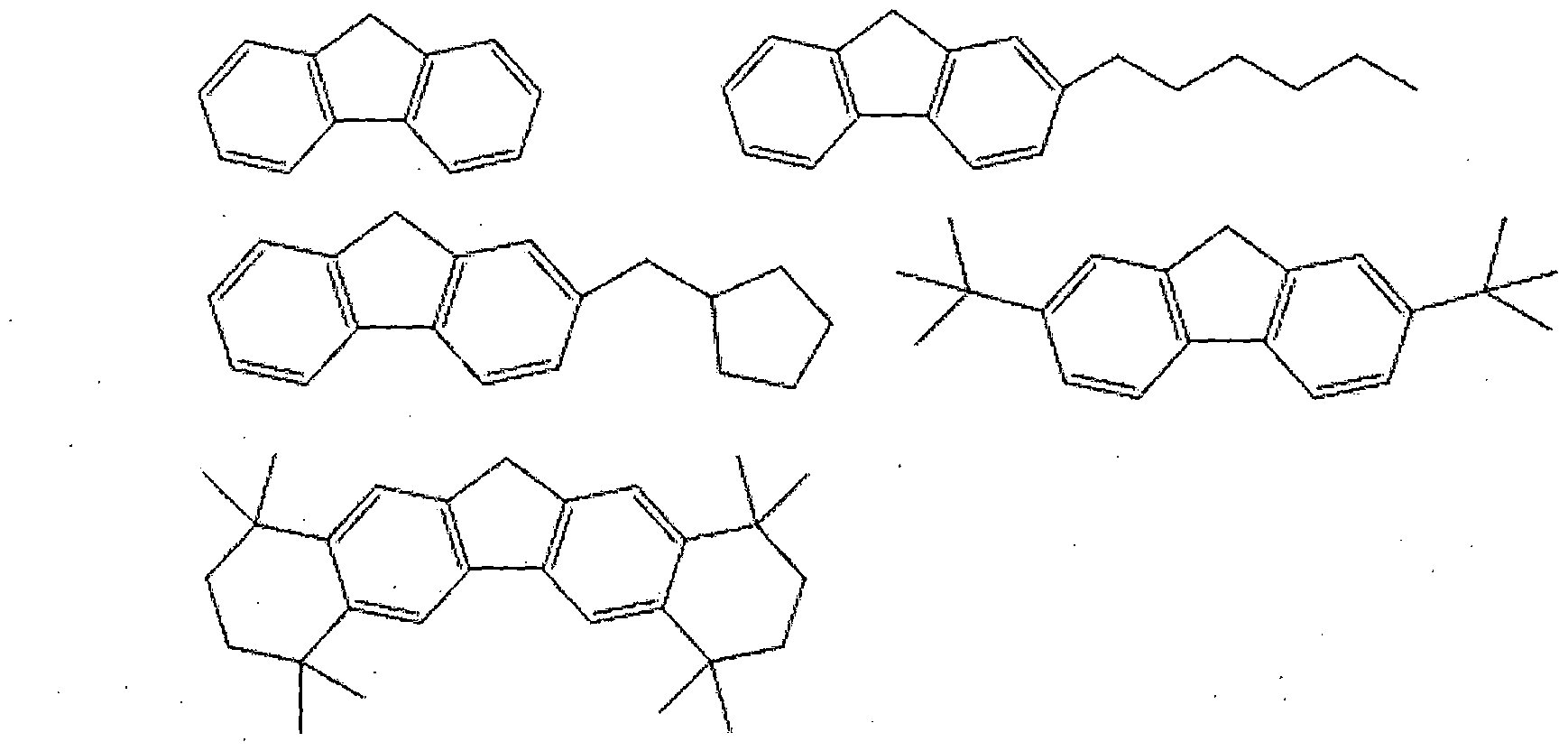
- R1 to R17 and R1 'of the formulas 2a, 2b and 2c are each independently hydrogen, methyl group, ethyl group, propyl group, isopropyl group, n-butyl group, tert-butyl group, pentyl group, nuclear group, heptyl group, octyl group, phenyl group, halogen group, trimethylsilyl group , Triethylsilyl group, tripropylsilyl group, tributylsilyl group, triisopropylsilyl group, trimethylsilylmethyl group, meso groups, or ethoxy groups are more preferred, but are not limited thereto.
- L of Formula 1 is more preferably a straight or branched chain alkylene group of C4 to C8, but is not limited thereto.
- the alkylene group may be substituted or unsubstituted with an alkyl group of C1 to C20, an alkenyl group of C2 to C20, or an aryl group of C6 to C20.
- a in Formula 1 is hydrogen, methyl group, ethyl group, propyl group, isopropyl group, n-butyl group, tert- butyl group, mesoxymethyl group, tert-butoxy methyl group, 1-ethoxyethyl group, 1-methyl It is preferable that it is a 1-methoxyethyl group, a tetrahydropyranyl group, or a tetrahydrofuranyl group, but it is not limited to this.
- B of Formula 1 is preferably silicon, but is not limited thereto.
- the first metallocene compound of Chemical Formula 1 is a non-covalent electron pair which forms a structure in which an indeno indole derivative and / or a fluorene derivative are crosslinked by a bridge, and may act as a Lewis base on the ligand structure. By having it, it is supported on the surface having the Lewis acid characteristic of the carrier and shows high polymerization activeol even when it is supported. It is also highly active as it contains an electronically rich indeno indole group and / or a pluorene group, and has low hydrogen reaction properties due to proper steric hindrance and the electronic effect of the ligand, and high hydrogen even in the presence of hydrogen. Activity is maintained.
- the beta-hydrogen of the polymer chain in which the nitrogen atom of the indeno indole derivative is grown is stabilized by hydrogen bonding, thereby inhibiting beta-hydrogen elimination, thereby polymerizing an ultra high molecular weight olefin polymer.
- specific examples of the compound represented by Chemical Formula 2a may include a compound represented by one of the following structural formulas, but
- the compound of Formula 2c Specific examples include compounds represented by one of the following structural formulae, but
- the first metallocene compound of Chemical Formula 1 is excellent in active activity and may polymerize a high molecular weight olefin polymer. In particular, even when used on a carrier, By exhibiting polymerization activity, an ultrahigh molecular weight polyolefin polymer can be produced.
- the i-metallocene compound of Formula 1 according to the present invention exhibits low hydrogen reaction properties. Still high activity allows the addition of ultra high molecular weight olefinic polymers. Therefore, an olefin polymer that satisfies high molecular weight properties without deterioration of activity can be produced even when used in combination with a catalyst having different properties, thereby including an olefinic polymer having a wide molecular weight distribution while containing an olefinic polymer of the polymer. It can be manufactured easily.
- the first metallocene compound of Chemical Formula 1 may be prepared by connecting an indenoindole derivative and / or fluorene derivative with a bridge compound to prepare a ligand compound, and then performing metallation by introducing a metal precursor compound. Can be.
- the manufacturing method of the said 1st metallocene compound is concretely demonstrated to the Example mentioned later.
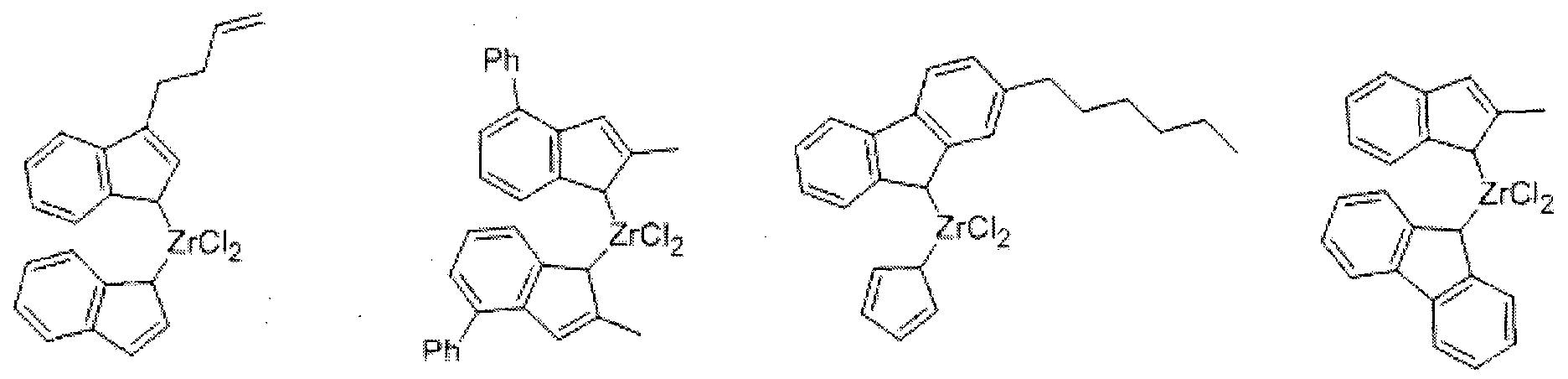
- the second metallocene compound is represented by the following formula
- M 1 is a Group 4 transition metal
- Cp 1 and Cp 2 are the same as or different from each other, and are each independently selected from the group consisting of cyclopentadienyl, indenyl, 4,5,6,7-tetrahydro-1-indenyl, and fluorenyl radicals One, they may be substituted with a hydrocarbon of 1 to 20 carbon atoms;
- R a and R b are the same as or different from each other, and each independently hydrogen, C1 to
- C20 alkyl C1 to C10 alkoxy, C2 to C20 alkoxyalkyl, C6 to C20 aryl, C6 to C10 aryloxy, C2 to C20 alkenyl, C7 to C40 alkylaryl, C7 to C40 arylalkyl Arylalkenyl of C8 to C40, or alkynyl of C2 to C10;
- Z 1 is a halogen atom, C 1 to C 20 alkyl, C 2 to C 10 alkenyl, C 7 to C40 alkylaryl, C7 to C40 arylalkyl, C6 to C20 aryl, substituted or unsubstituted C1 to C20 alkylidene, substituted or unsubstituted amino group, C2 to C20 alkylalkoxy, or C7 to C40 aryl Alkoxy;
- n 1 or 0;
- M 2 is a Group 4 transition metal
- Cp 3 and Cp 4 are the same as or different from each other, and are each independently selected from the group consisting of cyclopentadienyl, indenyl, 4,5,6,7-tetrahydro-1-indenyl and fluorenyl radicals They may be substituted with a hydrocarbon having 1 to 20 carbon atoms;
- 'R c and R d are the same or different and are each independently hydrogen, C1 to C20 alkyl, a C1 to C10 alkoxy, C2 to C20 alkoxy alkyl, C6 to C20 aryl, C6 to C10 aryl-oxy, C2 to C20 alkenyl, C7 to C40 alkylaryl, C7 to C40 arylalkyl, C8 to C40 arylalkenyl, or C2 to C10 alkynyl;
- Z 2 is a halogen atom, C1 to C20 alkyl, C2 to C10 alkenyl, C7 to C40.
- B 1 is one or more of a carbon, germanium, silicon, phosphorus or nitrogen atom containing radical which crosslinks the Cp 3 R c ring and the Cp 4 R d ring or crosslinks one Cp 4 R d ring to M 2 Or a combination thereof;
- n 1 or 0;
- M 3 is a Group 4 transition metal
- Cp 5 is cyclopentadienyl, indenyl, 4,5,6,7-tetrahydro ⁇ 1-indenyl and Any one selected from the group consisting of fluorenyl radicals, which may be substituted with hydrocarbons having 1 to 20 carbon atoms; ,
- R e is hydrogen, C1 to C20 alkyl, C1 to C10 alkoxy, C2 to C20 alkoxyalkyl, C6 to C20 aryl, C6 to C10 aryloxy, C2 to C20 alkenyl, C7 to C40 alkylaryl C7-C40 arylalkyl, C8-C40 arylalkenyl, or C2-C10 alkynyl;
- Z 3 is a halogen atom, C1 to C20 alkyl, C2 to C10 alkenyl, C7 to C40 alkylaryl, C7 to C40 arylalkyl, C6 to C20 aryl, substituted or unsubstituted C1 to C20 alkylidene , Substituted or unsubstituted amino group, C2 to C20 alkylalkoxy, or C7 to C40 arylalkoxy;
- B 2 is one or more or a combination of carbon, germanium, silicon, phosphorus or nitrogen atom containing radicals which crosslink the Cp 5 R e ring and J;
- J is any one selected from the group consisting of NR f , O, PR f and S, wherein R f is ⁇ C1 to C20 alkyl, aryl, substituted alkyl or substituted aryl.
- the compound represented by Formula ' 5 may be, for example, a compound represented, but is not limited thereto.
- the common supported metallocene catalyst according to the present invention may include at least one of the first metallocene compound represented by Chemical Formula 1 and the second metallocene compound selected from the compounds represented by Chemical Formulas 3 to 5. At least one species is commonly supported on a carrier together with a promoter compound. '
- the first metallocene compound represented by the formula (1) of the common supported metallocene catalyst mainly contributes to making a high molecular weight copolymer having a high SCB (short chain branch) content, and a second metal represented by the formula (3).
- Sen compounds may contribute to the production of low molecular weight co-polymers with predominantly low SCB content.
- the second metallocene compound represented by Formula 4 or 5 may contribute to making a low molecular weight copolymer having a moderate SCB content.
- the hybrid supported metallocene catalyst is represented by the chemical formula
- At least one first metallocene compound of Formula 1 and at least one second metallocene compound of Formula 3 may be included.
- the common supported metallocene catalyst may include at least one first metallocene compound of Formula 1 and at least one second metallocene compound of Formula 3, Or one or more second metallocene compounds of Formula 5;
- the first metallocene compound may form a ligand structure in which an indeno indole derivative and a fluorene derivative are crosslinked by a bridge compound, and may act as a Lewis base to the ligand structure.
- a non-covalent electron pair present, it is supported on the surface having the Lewis acid characteristics of the carrier and shows high polymerization activity even when supported.
- the high activity and / or low hydrogen response due to the proper steric hindrance and the electronic effect of the ligand, High activity is maintained even in situations.
- the common supported metallocene catalyst of the present invention includes a first metallocene compound represented by Chemical Formula 1 and a second metallocene compound selected from compounds represented by Chemical Formulas 3 to 5,
- a first metallocene compound represented by Chemical Formula 1 and a second metallocene compound selected from compounds represented by Chemical Formulas 3 to 5
- an olefin polymer having a high SCB content and a high molecular weight olefin copolymer and at the same time a wide molecular weight distribution having excellent physical properties and excellent processability can be prepared.
- the cocatalyst supported on the carrier for activating the metallocene compound is an organometallic compound containing a Group 13 metal. It will not be specifically limited if it can be used at the time of superposition
- the cocatalyst compound may include at least one of an aluminum-containing first cocatalyst of Formula 6 and a borate-based second cocatalyst of Formula 7 below. '
- each R 1 is a halogen, halogen substituted or unsubstituted hydrocarbyl group having 1 to 20 carbon atoms, k is an integer of 2 or more,
- T + is a + monovalent polyatomic ion
- ⁇ is boron in the +3 oxidation state
- G is independently a hydride group, a dialkylamido group, a halide group, an alkoxide group, an aryloxide group, hydro Selected from the group consisting of carbyl groups, halocarbyl groups and halo-substituted hydrocarbyl groups, wherein G has up to 20 carbons, but at up to one position G is a halide group.
- the first cocatalyst of Chemical Formula 6 may be an alkylaluminoxane compound having a repeating unit bonded in a linear, circular or reticular form.
- Examples of the first cocatalyst include methylaluminoxane (MAO) and ethyl. Aluminoxane, isobutyl aluminoxane, butyl aluminoxane, etc. are mentioned.
- the second cocatalyst of Formula 7 may be a borate-based compound in the form of a trisubstituted ammonium salt, or a dialkyl ammonium salt, a trisubstituted phosphonium salt.
- Such a second cocatalyst include trimetalammonium tetraphenylborate, methyldioctadecylammonium tetraphenylborate triethylammonium tetraphenylborate tripropylammonium tetraphenylborate, tri (n-butyl) ammonium tetraphenylborate, methyl Tetratecyclooctadecylammonium tetraphenylborate, ⁇ , ⁇ -dimethylaniline tetraphenylborate, ⁇ , ⁇ -diethylaninynium tetraphenylborate, ⁇ , ⁇ -dimethyl (2,4,6-trimethylaninynium) tetra Phenylborate, trimethylammonium tetrakis (pentafluorophenyl) borate, methylditetradecylammonium tetrakis (
- the mass ratio of the total transition metal to the carrier contained in the first metallocene compound represented by Formula 1 and the second metallocene compound represented by Formulas 3 to 5 is 1 : 10 to 1: 1,000.
- the carrier and the metallocene compound are included in the mass ratio, the optimum shape can be exhibited.
- the mass ratio of the promoter compound to the carrier may be 1: 1 to 1: 100.
- the mass ratio of the first metallocene compound represented by Formula 1 to the second metallocene compound represented by Formulas 3 to 5 is 10: 1 to 1:10, preferably 5: 1 to 1: 5. have.
- the support to the can be used a carrier containing hydroxyl groups on the surface, preferably the drying is to remove the moisture on the surface ", half male large-hydroxy group Carriers having siloxane groups can be used.
- silica, silica-alumina, silica-magnesia, etc., dried at a high temperature may be used, and these are usually oxides, carbonates, sulfates, such as N 0, K 2 C0 3 , BaS0 4 , and Mg (N0 3 ) 2 . , And nitrate components.
- Drying degree of the carrier is preferably 200 to 800 ° C., more preferably 300 to 600 ° C., most preferably 300 to 400 ° C. If the drying temperature of the carrier is less than 200 ° C, there is too much moisture . Moisture and promoter on the surface will react, and if it exceeds 800 ° C, the pores on the surface of the carrier will coalesce and the surface area will decrease, and there will be no hydroxy groups on the surface and only siloxane groups will remain. It is undesirable because the reaction site decreases.
- the amount of hydroxy groups on the surface of the carrier is preferably 0.1 to 10 mmol / g, more preferably 0.5 to 5 mmol / g.
- the amount of hydroxyl groups on the surface of the carrier can be controlled by the method and conditions for preparing the carrier or by drying conditions such as temperature, time, vacuum or spray drying.
- the amount of the hydroxyl group is less than 1 mmol / g, the reaction space with the cocatalyst is small. If the amount of the hydroxyl group is more than 10 mmol / g, the hydroxyl group may be due to moisture other than the hydroxyl group present on the surface of the carrier particle. Not.
- the common supported metallocene catalyst according to the present invention can be used by itself for the polymerization of olepin-based monomers.
- the common supported metallocene catalyst according to the present invention may be prepared by using a pre-polymerized catalyst in contact with an olefinic monomer.
- the catalyst may be separately used for ethylene, propylene, 1-butene, 1-nuxene, and 1-octene. It may be prepared and used as a prepolymerized catalyst by contacting with an olefinic monomer such as the like.
- the common supported metallocene catalyst according to the present invention may include, for example, supporting a cocatalyst compound on a carrier, supporting a first metallocene compound represented by Formula 1 on the carrier, and on the carrier.
- the compound represented by Formula 3 to 5 may be prepared by supporting the second metallocene compound selected.
- the order of the step of supporting the first metallocene compound and the step of supporting the second metallocene compound may be changed as necessary. That is, the first metallocene compound is first supported on the carrier, and then the second metallocene compound is further supported to prepare a common supported metallocene catalyst, or the second metallocene compound is supported on the carrier. After supporting first, the common metallocene catalyst may be prepared by further supporting the first metallocene compound.
- the temperature may be performed at a temperature of about 0 to about 100 ° C. and a pressure of normal pressure, but is not limited thereto.
- the olefin resin can be prepared by polymerizing the olefin resin in the presence of the common supported metallocene catalyst of the present invention as described above.
- the olefinic monomers are ethylene, alpha-olefins, cyclic olefins, double It may be a diene olefin or a triene olefin having two or more bonds.
- olefin monomers include ethylene, propylene, 1-butene, 1-pentene, 4-methyl ⁇ 1-pentene, 1-nuxene, 1-heptene, 1-octene, 1-decene, 1-undecene, 1-dodecene, 1-tetradecene, 1-nucledecene, 1-atose, norbornene, norbonadiene, ethylidenenorbornene, phenylnorbornene, vinylnorbornene, dicyclopentadiene, 1,4-butadiene And 1,5-pentadiene, 1,6-nuxadiene, styrene, alpha-methylstyrene, divinylbenzene, 3-chloromethylstyrene, and the like, and these monomers may be mixed and copolymerized.
- the polymerization reaction may be carried out by homopolymerizing one olefin monomer or copolymerizing two or more monomers using one continuous slurry polymerization reactor, a loop slurry reactor, a gas phase reactor or a solution reactor.
- the common supported metallocene catalyst is an aliphatic hydrocarbon solvent having 5 to 12 carbon atoms, for example, pentane, nucleic acid, heptane, nonane, decan, and isomers thereof and aromatic hydrocarbon solvents such as toluene and benzene, dichloromethane and chlorobenzene. It may be dissolved or liquefied and injected into a hydrocarbon solvent substituted with a chlorine atom such as.
- the solvent used herein is preferably used by removing a small amount of water or air that acts as a catalyst poison by treating a small amount of alkyl aluminum, and may be carried out by further using a promoter.
- an ellepin-based polymer having a wide molecular weight distribution of a BOCD structure having a low SCB content of a low molecular weight ⁇ portion and a high SCB content of a high molecular weight portion is prepared. can do.
- the olefin polymer is not only excellent in physical properties, but also excellent in workability.
- the leupin-based polymers prepared using the common supported metallocene catalyst of the present invention may exhibit a high weight average molecular weight of about 300,000 or more or about 350,000 or more.
- the common supported metallocene catalyst of the present invention exhibits excellent activity
- the olefinic polymer prepared using the common supported metallocene catalyst of the present invention is, for example, about 3.0 to about 8.0, preferably about 4.0 to about
- a broad molecular weight distribution (PDI) of about 8.0, more preferably 5.0 to about 8.0 can be seen to indicate excellent processability.
- PDI broad molecular weight distribution
- the solution was changed to violet color at room temperature overnight.
- the reaction solution was filtered to remove LiCl.
- the toluene of the filtrate was removed by vacuum drying, and the nucleic acid was added and sonicated for 1 hour.
- the quality of the solid (filtered solid) was filtered to filter the slurry is purple (dark violet) the metallocene compound 6 g (Mw 758.02, 7.92 mmol , yield 66mol 0/0) to give the a.
- Two isomers were observed on 1 H-NMR.
- a ligand compound 3.82 g (6 mmol) of toluene and 100 mL MTBE dissolved in 5 mL 'after a 2.5M n-BuLi hexane solution 5.6 mL (l 4 mmol) prepared in 2-1 and writer in dryice / acetone bath Stir overnight at room temperature.
- 2.26 g (6 mmol) of ZrCl 4 (THF) 2 was prepared, and 100 ml of toluene was added to prepare a slurry.
- Toluene slurry of ZrCl 4 (THF) 2 was transferred to litiated ligand in a dry ice / acetone bath. It stirred at room temperature overnight and it changed into violet color.
- the reaction solution was filtered to remove LiCl, and the filtrate was dried in vacuo, and hexane was added to sonication.
- the slurry was filtered to obtain 3.40 g (yield 71.1 mol%) of a metal violet compound of dark violet as a filtered solid.
- t-Butyl-0- (C3 ⁇ 4) 6 -Cl was prepared by the method shown in Tetrahedron Lett. 2951 (.1988) and reacted with NaCp.
- t-Butyl-0- (CH 2 ) 6 -C 5 H 5 was obtained (yield 60%, bp 80 ° C / 0.1 mmHg).
- t-Butyl-0- (CH 2 ) 6 -C 5 H 5 was dissolved in THF at -781 :, and normal butyllithium (n-BuLi) was slowly added thereto, and the temperature was raised to room temperature, followed by reaction for 8 hours. .
- the solution was again synthesized lithium salt in a suspension solution of ZrCl 4 (THF) 2 (1 .7 ( ) g, 4.50 mmol) / THF ( 3 0m £) at -78 ° C. ) was added slowly and further reacted at room temperature for 6 hours.
- 6-t-butoxynucleic acid (6-t-buthoxyhexane) was confirmed by 1 H-NMR. From the 6-t-butoxynucleic acid, it was found that the Gringanrd reaction proceeded well. Thus, 6-t-buthoxyhexyl magnesium chloride was synthesized.
- TiCl 3 (THF) 3 (10 mmol) was rapidly added to the dilithium salt of -78 ligand synthesized from (Dimethyl (tetramethylCpH) t-Butylaminosilane) in THF solution.
- the reaction solution was slowly stirred for 12 hours at room temperature at -78 ° C.
- an equivalent amount of PbCl 2 (10 mmol) was added to the reaction solution at room temperature, followed by stirring for 12 hours. After stirring for 12 hours, a dark black solution was obtained.
- nucleic acid was added to filter the product.
- Silica (SYLOPOL 948, manufactured by Grace Davison) was dehydrated under vacuum at a temperature of 400 ° C. for 15 hours.
- the supported catalyst was prepared in the same manner as in Example 1, except that 0.25 mm of the metallocene catalyst of Preparation Example 2 was used instead of 0.25 mmol of the metallocene catalyst of Preparation Example 1 in Example 1.
- Example 4 After 1 hour of 0.25 mmol reaction of the metallocene catalyst of Preparation Example 1 was finished in Example 1, 0.25 mmol reaction of the metallocene catalyst of Preparation Example 4 was further performed. Thereafter, the supported catalyst was prepared in the same manner except that 0.25 mmol reaction of the metallocene catalyst of Preparation Example 3 was performed.
- Example 4
- the supported catalyst was prepared in the same manner as in Example 3, except that 0.25 mmol of the metallocene catalyst of Preparation Example 2 was used instead of 0.25 mmol of the metallocene catalyst of Preparation Example 1, which was reacted initially in Example 3.
- Example 5
- a supported catalyst was prepared in the same manner as in Comparative Example 3, except that 1.0 mmol of anilinium borate ( ⁇ , ⁇ -dimethylanilinium tetrakis (pentafluorophenyl) borate, AB) was added to the last step.
- anilinium borate ⁇ , ⁇ -dimethylanilinium tetrakis (pentafluorophenyl) borate, AB
- the polymer obtained therefrom was filtered to remove most of the polymerization solvent and then dried in an 80 ° C. vacuum oven for 4 hours.
- Examples 1 to 5 for the common supported catalyst of the present invention include two or more metallocene compounds, but have a much higher activity than the comparative example including only a single catalyst or a second metallocene compound. It can be seen that a polymer having a high molecular weight and a wide molecular weight distribution can be produced.
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016518095A JP6282341B2 (ja) | 2013-10-18 | 2014-10-15 | 混成担持メタロセン触媒 |
| EP14854760.7A EP3037167B1 (en) | 2013-10-18 | 2014-10-15 | Hybrid-supported metallocene catalyst |
| US15/026,544 US9926395B2 (en) | 2013-10-18 | 2014-10-15 | Hybrid supported metallocene catalyst |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2013-0124518 | 2013-10-18 | ||
| KR20130124518 | 2013-10-18 | ||
| KR1020140138348A KR101644113B1 (ko) | 2013-10-18 | 2014-10-14 | 혼성 담지 메탈로센 촉매 |
| KR10-2014-0138348 | 2014-10-14 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015056975A1 true WO2015056975A1 (ko) | 2015-04-23 |
Family
ID=52828353
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2014/009681 WO2015056975A1 (ko) | 2013-10-18 | 2014-10-15 | 혼성 담지 메탈로센 촉매 |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2015056975A1 (ko) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015186970A1 (ko) * | 2014-06-03 | 2015-12-10 | 주식회사 엘지화학 | 폴리올레핀의 제조 방법 및 이로부터 제조된 폴리올레핀 |
| WO2016093580A1 (ko) * | 2014-12-08 | 2016-06-16 | 주식회사 엘지화학 | 가공성이 우수한 에틸렌/알파-올레핀 공중합체 |
| US9988469B2 (en) | 2014-06-03 | 2018-06-05 | Lg Chem, Ltd. | Method for preparing polyolefin and polyolefin prepared thereby |
| JP2018529826A (ja) * | 2016-02-24 | 2018-10-11 | エルジー・ケム・リミテッド | 混成担持メタロセン触媒およびこれを利用したポリオレフィンの製造方法 |
| US10155830B2 (en) | 2014-12-08 | 2018-12-18 | Lg Chem, Ltd. | Ethylene/alpha-olefin copolymers having excellent processability |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5032562A (en) | 1989-12-27 | 1991-07-16 | Mobil Oil Corporation | Catalyst composition and process for polymerizing polymers having multimodal molecular weight distribution |
| US5525678A (en) | 1994-09-22 | 1996-06-11 | Mobil Oil Corporation | Process for controlling the MWD of a broad/bimodal resin produced in a single reactor |
| US5914289A (en) | 1996-02-19 | 1999-06-22 | Fina Research, S.A. | Supported metallocene-alumoxane catalysts for the preparation of polyethylene having a broad monomodal molecular weight distribution |
| KR20030012308A (ko) | 2001-07-31 | 2003-02-12 | 주식회사 예스아이비 | 배팅형 복권 시스템 및 배팅 방법 |
| KR20110035968A (ko) * | 2009-09-29 | 2011-04-06 | 주식회사 엘지화학 | 3종 혼성 메탈로센 담지 촉매 및 그의 제조방법 |
| US20120059135A1 (en) * | 2009-03-30 | 2012-03-08 | Mitsui Chemicals, Inc. | Copolymer of olefin and conjugated diene, and process for producing the same |
| KR20120076156A (ko) * | 2010-12-29 | 2012-07-09 | 주식회사 엘지화학 | 혼성 담지 메탈로센 촉매의 제조방법 및 이를 이용하여 제조된 혼성 담지 메탈로센 촉매 |
| KR20120087706A (ko) * | 2011-01-28 | 2012-08-07 | 주식회사 엘지화학 | 메탈로센 화합물 및 이를 이용하여 제조되는 올레핀계 중합체 |
| WO2013133595A1 (en) * | 2012-03-06 | 2013-09-12 | Sk Innovation Co., Ltd. | Hybrid supported metallocene catalyst, method for preparing the same, and process for preparing polyolefin using the same |
-
2014
- 2014-10-15 WO PCT/KR2014/009681 patent/WO2015056975A1/ko active Application Filing
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5032562A (en) | 1989-12-27 | 1991-07-16 | Mobil Oil Corporation | Catalyst composition and process for polymerizing polymers having multimodal molecular weight distribution |
| US5525678A (en) | 1994-09-22 | 1996-06-11 | Mobil Oil Corporation | Process for controlling the MWD of a broad/bimodal resin produced in a single reactor |
| US5914289A (en) | 1996-02-19 | 1999-06-22 | Fina Research, S.A. | Supported metallocene-alumoxane catalysts for the preparation of polyethylene having a broad monomodal molecular weight distribution |
| KR20030012308A (ko) | 2001-07-31 | 2003-02-12 | 주식회사 예스아이비 | 배팅형 복권 시스템 및 배팅 방법 |
| US20120059135A1 (en) * | 2009-03-30 | 2012-03-08 | Mitsui Chemicals, Inc. | Copolymer of olefin and conjugated diene, and process for producing the same |
| KR20110035968A (ko) * | 2009-09-29 | 2011-04-06 | 주식회사 엘지화학 | 3종 혼성 메탈로센 담지 촉매 및 그의 제조방법 |
| KR20120076156A (ko) * | 2010-12-29 | 2012-07-09 | 주식회사 엘지화학 | 혼성 담지 메탈로센 촉매의 제조방법 및 이를 이용하여 제조된 혼성 담지 메탈로센 촉매 |
| KR20120087706A (ko) * | 2011-01-28 | 2012-08-07 | 주식회사 엘지화학 | 메탈로센 화합물 및 이를 이용하여 제조되는 올레핀계 중합체 |
| WO2013133595A1 (en) * | 2012-03-06 | 2013-09-12 | Sk Innovation Co., Ltd. | Hybrid supported metallocene catalyst, method for preparing the same, and process for preparing polyolefin using the same |
Non-Patent Citations (1)
| Title |
|---|
| TETRAHEDRON LETT., 1988, pages 2951 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015186970A1 (ko) * | 2014-06-03 | 2015-12-10 | 주식회사 엘지화학 | 폴리올레핀의 제조 방법 및 이로부터 제조된 폴리올레핀 |
| US9988469B2 (en) | 2014-06-03 | 2018-06-05 | Lg Chem, Ltd. | Method for preparing polyolefin and polyolefin prepared thereby |
| WO2016093580A1 (ko) * | 2014-12-08 | 2016-06-16 | 주식회사 엘지화학 | 가공성이 우수한 에틸렌/알파-올레핀 공중합체 |
| US10155830B2 (en) | 2014-12-08 | 2018-12-18 | Lg Chem, Ltd. | Ethylene/alpha-olefin copolymers having excellent processability |
| JP2018529826A (ja) * | 2016-02-24 | 2018-10-11 | エルジー・ケム・リミテッド | 混成担持メタロセン触媒およびこれを利用したポリオレフィンの製造方法 |
| US11091568B2 (en) | 2016-02-24 | 2021-08-17 | Lg Chem, Ltd. | Hybrid supported metallocene catalyst and polyolefin preparation method using same |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6282341B2 (ja) | 混成担持メタロセン触媒 | |
| JP6247751B2 (ja) | 混成担持メタロセン触媒の製造方法 | |
| EP3106474B1 (en) | Method of preparing a polyolefin | |
| JP6488002B2 (ja) | 加工性に優れたオレフィン系重合体 | |
| JP2017518423A (ja) | 耐環境応力亀裂性に優れたポリオレフィン | |
| KR102073252B1 (ko) | 올레핀 공중합체 합성용 촉매 조성물 및 올레핀 공중합체의 제조 방법 | |
| WO2016036204A1 (ko) | 가공성이 우수한 올레핀계 중합체 | |
| EP3348585A1 (en) | Supported hybrid metallocene catalyst and polyolefin preparation method using same | |
| JP6440832B2 (ja) | メタロセン化合物、メタロセン担持触媒およびこれを用いるポリオレフィンの製造方法 | |
| KR102064990B1 (ko) | 에틸렌 슬러리 중합용 혼성 담지 촉매 시스템 및 이를 이용한 에틸렌 중합체의 제조 방법 | |
| KR20150037520A (ko) | 폴리올레핀의 제조 방법 및 이로부터 제조된 폴리올레핀 | |
| KR102028736B1 (ko) | 혼성 담지 메탈로센 촉매의 제조방법 및 이를 이용하여 제조된 혼성 담지 메탈로센 촉매 | |
| EP3255066A1 (en) | Method for producing metallocene-supported catalyst | |
| KR101606825B1 (ko) | 혼성 담지 메탈로센 촉매의 제조방법 | |
| WO2015056975A1 (ko) | 혼성 담지 메탈로센 촉매 | |
| EP3330296B1 (en) | Method for preparing supported metallocene catalyst | |
| WO2015186970A1 (ko) | 폴리올레핀의 제조 방법 및 이로부터 제조된 폴리올레핀 | |
| WO2015056974A1 (ko) | 혼성 담지 메탈로센 촉매의 제조방법 | |
| KR102215024B1 (ko) | 폴리올레핀의 제조 방법 | |
| KR20200090041A (ko) | 혼성 담지 메탈로센 촉매의 제조 방법 및 이를 이용한 올레핀 중합체의 제조 방법 | |
| WO2017155211A1 (ko) | 에틸렌 슬러리 중합용 혼성 담지 촉매 시스템 및 이를 이용한 에틸렌 중합체의 제조 방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14854760 Country of ref document: EP Kind code of ref document: A1 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2014854760 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014854760 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2016518095 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15026544 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |