WO2013113787A1 - Fungicidal pyrimidine compounds - Google Patents
Fungicidal pyrimidine compounds Download PDFInfo
- Publication number
- WO2013113787A1 WO2013113787A1 PCT/EP2013/051854 EP2013051854W WO2013113787A1 WO 2013113787 A1 WO2013113787 A1 WO 2013113787A1 EP 2013051854 W EP2013051854 W EP 2013051854W WO 2013113787 A1 WO2013113787 A1 WO 2013113787A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- alkoxy
- individualized compound
- compound
- compounds
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/32—One oxygen, sulfur or nitrogen atom
- C07D239/42—One nitrogen atom
Abstract
The present invention relates to fungicidal pyrimidine compounds of formula I as defined herein, to their use and to methods for combating phytopathogenic fungi. The present invention also relates to seeds treated with at least one such compound. Furthermore the invention relates to processes for preparing compounds of formula (I).
Description
Fungicidal pyrimidine compounds
Description The present invention relates to fungicidal pyrimidine compounds, to their use and to methods for combating phytopathogenic fungi. The present invention also relates to seeds treated with at least one such compound. Furthermore the invention relates to processes for preparing compounds of formula I and intermediates as well as processes for the preparation of such intermediates.
WO 1998/003272 describes a process for the preparation of 4-amino-5-chloro-6-(1 -fluoro- ethyl)pyrimidine compounds, which are pest controlling agents for agricultural and horticultural use. WO 1997/028133 discloses acylated 4-amino and 4-hydrazinopyrimidines and their use as pesticides. WO 1995/18795 relates to N-(4-pyrimidinyl)amide pesticides and EP 665225 A1 relates to 4-phenethylamino pyrimidine derivatives useful for chemical control of noxious organisms.
The compounds according to the present invention differ from those described in the abovementioned publications by the specific substituent Ra5 of the pyrimidine group in position 5 as defined herein and in that the phenyl ring is substituted with a heteroaryloxy group.
EP 264217 A2 relates to N-(4-pyrimidinyl)arylalkylamine pesticides, inter alia disclosing N- (4-pyrimidinyl)phenylethylamines. EP 264217 A2 does not mention the specific combination of the specific substituent Ra5 on the pyrimidine ring and the specific ethylene linker between the aminopyrimidine moiety and the phenyl ring together with a heteroaryloxy substitution of the phenyl ring. In many cases, in particular at low application rates, the fungicidal activity of known fungicidal compounds is unsatisfactory. Based on this, it was an object of the present invention to provide compounds having improved activity and/or a broader activity spectrum against phytopathogenic fungi. This objective is achieved by the use of substituted pyrimidine compounds of formula I having good fungicidal activity against phytopathogenic harmful fungi.
Accordingly, the present in the formula I
wherein:
Ra5 is methyl or halomethyl; Ra2, Ra6 independently of each other are hydrogen, halogen, CN , NO2, OH , SH , Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci-C4-alkylthio, Ci-C4-haloalkylthio, Ci- C4-alkylsulfinyl, Ci-C4-haloalkylsulfinyl, Ci-C4-alkylsulfonyl,
Ci-C4-haloalkylsulfonyl, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkoxy,
C2-C4-alkenyl, C2-C4-alkynyl, C2-C4-haloalkenyl, C2-C4-haloalkynyl, Cs-Cs-cycloalkyl, C3-
Cs-cycloalkyloxy, C3-C8-cycloalkyl-Ci-C4-alkyl, NRARB, C(=0)R', C(=NOR")R'" or C(=NH)- 0-R'";
RA,RB independently of one another are hydrogen, Ci-C4-alkyl, C2-C4-alkenyl, C2-C4- alkynyl, phenyl, benzyl, Cs-Cs-cycloalkyl, Cs-Cs-cycloalkenyl or C(=0)-R';
R' is hydrogen, OH, NH2, Ci-C4-alkyl, Ci-C4-haloalkyl, C2-C4-alkenyl, C2-C4-alkynyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci-C4-alkylamino or di(Ci-C4-alkyl)amino;
R" is hydrogen, Ci-C4-alkyl, Ci-C4-haloalkyl, C2-C4-alkenyl, C2-C4-alkynyl or
C1 -C4-a I koxy-C1 -C4-a I ky I ,
R'" is hydrogen or Ci-C4-alkyl; is hydrogen, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy,
Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-haloalkoxy-Ci-C4-alkyl, C2-C4-alkenyl,
C2-C4-haloalkenyl, C2-C4-alkynyl, C2-C4-haloalkynyl, CN, CH2CN, NRARB or
CH2-0-C(=0)R';
, R2 independently of each other are hydrogen, halogen, OH, CN, Ci-C4-alkyl,
Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-halo- alkoxy-Ci-C4-alkyl, C2-C4-alkenyl, C2-C4-haloalkenyl, C2-C4-alkynyl,
C2-C4-haloalkynyl, Cs-Cs-cycloalkyl, C3-Cs-cycloalkyloxy, Cs-Cs-cycloalkyl-
Ci-C4-alkyl, NRARB, C(=0)R', C(=NOR")R"', C(=NH)-0-R"' or benzyl,
wherein the phenyl moiety of benzyl is unsubstituted or carries 1 , 2 , 3, 4 or 5 substituents selected from the group consisting of CN, halogen, Ci-C4-alkyl,
Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, (Ci-C4-alkoxy)carbonyl and di(Ci-C4- alkyl)aminocarbonyl, or two radicals R1 and R2 that are bound to the same carbon atom form together with said carbon atom a saturated or partially unsaturated
3-, 4-, 5-, 6- or 7-membered carbocycle or a saturated or partially unsaturated 3-, 4-, 5-, 6- or 7-membered heterocycle, wherein the ring member atoms of the abovementioned heterocycle include beside carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S, and wherein the abovementioned cycle is unsubstituted or carries 1 , 2, 3 or 4 substituents selected from halogen, CN, OH, SH, Ci-C4-alkyl, Ci-C4-alkoxy or Ci-C4- alkylthio; and
one or two CH2 groups of the abovementioned cycles may be replaced by one or two C(=0) or C(=S) groups respectively; indicates the number of substituents Rb on the phenyl ring and n is 0, 1 , 2, 3 or 4; is halogen, CN, NO2, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, C2-C4-alkenyl, C2-C4-haloalkenyl, C2-C4-alkynyl, C2-C4-haloalkynyl, NRARB, C(=NOR")R"'
or C(=NH)-0-R"\
it being possible for n = 2, 3 or 4 that Rb are identical or different;
Het is a 5- or 6-membered heteroaryl, wherein the ring member atoms of the heteroaryl
include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S and wherein the heteroaryl is unsubstituted or carries 1 , 2, 3 or 4 identical or different groups Rc: is halogen, CN, NO2, Ci-C6-alkyl, Ci-C6-haloalkyl, Ci-C6-alkoxy, Ci-C6-haloalkoxy, Ci-C6-alkylthio, Ci-C6-haloalkylthio, Ci-C6-alkylsulfinyl, Ci-C6-haloalkylsulfinyl,
Ci-C6-alkylsulfonyl, Ci-C6-haloalkylsulfonyl, Ci-C6-alkoxy-Ci-C4-alkyl,
Ci-C6-haloalkoxy-Ci-C4-alkyl, C2-C6-alkenyl, C2-C6-alkynyl, NRARB, C(=0)R', C(=NOR")R'", C(=NH)-0-R'", C3-C8-cycloalkyl, C3-C8-cycloalkyl-Ci-C4-alkyl, phenyl, phenyl-Ci-C4-alkyl, phenoxy, phenoxy-Ci-C4-alkyl or a 5- or 6-membered heteroaryl, wherein the ring member atoms of the heteroaryl include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S, and wherein the aforementioned cyclic radicals are unsubstituted or carry 1 , 2, 3 or 4 identical or different substituents Rd:
Rd is halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy or Ci-C4-haloalkoxy; or two radicals Rc that are bound to adjacent ring member atoms of the Het group form together with said ring member atoms a fused
5-, 6- or 7-membered saturated, partially unsaturated or aromatic carbocycle or heterocycle, wherein the ring member atoms of the fused heterocycle include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S, and wherein the fused carbocycle or heterocycle is unsubstituted or carries 1 , 2, 3 or 4 identical or different radicals groups Re:
Re is halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy or Ci-C4-haloalkoxy; and the N-oxides and the agriculturally acceptable salts thereof.
The present invention furthermore relates to processes for preparing the compounds I. The present invention furthermore relates to intermediates such as compounds of formulae II and III.
The compounds of the present invention are useful for combating harmful fungi. Therefore the present invention furthermore relates to a method for combating harmful fungi, which process comprises treating the fungi or the materials, plants, the soil or seeds to be protected against fungal attack, with an effective amount of at least one compound of formula I or of an N- oxide or an agriculturally acceptable salt thereof.
Furthermore, the present invention also relates to seed comprising a compound of formula I, or an N-oxide or an agriculturally acceptable salt thereof, in an amount of from 0.1 g to 10 kg per 100 kg of seed.
Depending on the substitution pattern, the compounds of formula I and their N-oxides may
have one or more centers of chirality, in which case they are present as pure enantiomers or pure diastereomers or as enantiomer or diastereomer mixtures. Both, the pure enantiomers or diastereomers and their mixtures are subject matter of the present invention.
Agriculturally useful salts of the compounds I encompass especially the salts of those cations or the acid addition salts of those acids whose cations and anions, respectively, have no adverse effect on the fungicidal action of the compounds I. Suitable cations are thus in particular the ions of the alkali metals, preferably sodium and potassium, of the alkaline earth metals, preferably calcium, magnesium and barium, of the transition metals, preferably manganese, copper, zinc and iron, and also the ammonium ion which, if desired, may carry one to four Ci-C4-alkyl substituents and/or one phenyl or benzyl substituent, preferably
diisopropylammonium, tetramethylammonium, tetrabutylammonium, tnmethylbenzylammonium, furthermore phosphonium ions, sulfonium ions, preferably tri(Ci-C4-alkyl)sulfonium, and sulfoxonium ions, preferably tri(Ci-C4-alkyl)sulfoxonium.
Anions of useful acid addition salts are primarily chloride, bromide, fluoride, hydrogensulfate, sulfate, dihydrogenphosphate, hydrogenphosphate, phosphate, nitrate, bicarbonate, carbonate, hexafluorosilicate, hexafluorophosphate, benzoate, and the anions of Ci-C4-alkanoic acids, preferably formate, acetate, propionate and butyrate. They can be formed by reacting a compound I with an acid of the corresponding anion, preferably of hydrochloric acid,
hydrobromic acid, sulfuric acid, phosphoric acid or nitric acid.
The compounds of formula I can be present in atropisomers arising from restricted rotation about a single bond of asymmetric groups. They also form part of the subject matter of the present invention.
In repect of the variables, the embodiments of the intermediates correspond to the embodiments of the compounds of formula I.
The term "compounds I" refers to compounds of formula I. Likewise, this terminology applies to all sub-formulae, e. g. "compounds I .A" refers to compounds of the formula I .A or
"compounds II" refers to compounds of the formula II, etc..
The inventive compounds I can be prepared by various routes in analogy to prior art processes and, advantageously, by the synthesis shown in the following schemes and in the experimental part of this application.
A 4-halopyrimidine compound II, wherein Hal is halogen, preferably CI or F, can be reacted with a suitable phenethyl amine compound III to obtain a compound I according to the present invention as shown in Scheme 1 : Scheme 1
II III
Generally, this reaction is carried out at temperatures of from 0 to 200°C, preferably from 50 to 170°C, preferably in an inert organic solvent and preferably in presence of a base or a catalyst or a combination of a base and a catalyst (e.g. NaF, KF, LiF, NaBr, KBr, LiBr, Nal, Kl,
Lil and ionic liquids, such as imidazolium catalysts).
Suitable solvents are aromatic hydrocarbons such as toluene, o-, m- and p-xylene;
halogenated hydro-carbons such as chlorobenzene, dichlorobenzene; ethers such as dioxane, anisole and tetra-hydrofuran (THF); nitriles such as acetonitrile and propionitrile; ketones, such as acetone, methyl ethyl ketone, diethyl ketone and tert. -butyl methyl ketone; alcohols such as ethanol, n-propanol, isopropanol, n-butanol and tert.-butanol; and also dimethyl sulfoxide (DMSO), dimethylformamide (DMF), dimethyl acetamide, N-methyl-2-pyrrolidone (NMP), N- ethyl-2-pyrrolidone (NEP) and acetic acid ethyl ester, preferably DMSO, DMF, dimethyl acetamide, NMP, or NEP. Particular preference is given to NMP. It is also possible to use mixtures of the solvents mentioned.
Suitable bases are, in general, inorganic compounds, such as alkali metal and alkaline earth metal hydroxides such as lithium hydroxide, sodium hydroxide, potassium hydroxide and calcium hydroxide; alkali metal and alkaline earth metal oxides such as lithium oxide, sodium oxide, potassium oxide and calcium oxide; alkali metal and alkaline earth metal phosphates such as lithium phosphate, sodium phosphate, potassium phosphate and calcium phosphate; alkali metal amides such as lithium amide, sodium amide and potassium amide; alkali metal and alkaline earth metal hydrides lithium hydride, sodium hydride, potassium hydride and calcium hydride; alkali metal and alkaline earth metal carbonates such as lithium carbonate, potassium carbonate and calcium carbonate, caesium carbonate; moreover organic bases, for example tertiary amines such as trimethyl-amine (TMA), triethylamine (TEA), tributylamine (TBA), diisopropylethylamine (DIPEA) and N-methyl-2-pyrrolidone (NMP), pyridine, substituted pyridines such as collidine, lutidine and 4 dimethylaminopyridine (DMAP), and also bicyclic amines. Preference is given to sodium hydride, potassium hydride, lithium carbonate, potassium carbonate, caesium carbonate, TEA, TBA and DIPEA, in particular DIPEA. The bases are generally employed in equimolar amounts, in excess or, if appropriate, as solvent. The amount of base is typically 1.1 to 5.0 molar equivalents relative to 1 mole of compounds II.
The starting materials, are generally reacted with one another in equimolar amounts. In terms of yields, it may be advantageous to employ an excess of compounds III, based on 1.1 to 2.5 equivalents, preferred 1 .1 to 1.5 equivalents of compounds II.
The compounds II are known from the literature or are commercially available or they can be prepared for example in analogy to methods described in: Heterocycles (2009) 78(7), 1627- 1665; New J. Chem. (1994) 18(6), 701 -8; WO 2005/095357; Science of Synthesis (2004) 16, 379-572; WO 2008/156726; WO 2006/072831 ; Organic Reactions (Hoboken, NJ, United States) (2000), 56; or Targets in Heterocyclic Systems (2008) 12, 59-84.
The phenethyl amine compounds III are known from the literature or are commercially available or they can be prepared for example in analogy to methods described in:
WO 2007/046809; WO 201 1/025505; or WO 2010/025451 .
Alternatively, compounds III in which R2 is H can be prepared according to the general reaction Scheme 2:
Scheme 2
Compound AD-1 is reacted with a heterocycle Het-LG, in which LG is a leaving group such as, e.g. F or CI, in the presence of a base and/or a catalyst to provide compound Het-AD.
Alternatively, compound AD-2, in which LG is a leaving group in para-position such as, e.g. F or CI, is reacted with a hydroxyl-heterocycle HO-Het in the presence of a base and/or a catalyst to yield compound Het-AD according to procedures described in WO 201 1/032277;
WO 2008/065393; WO 2007/096647; WO 2007/06714, AU 2006201959 A1 , DE 19518073 A1 , Org. Lett. (2001 ), 3(26), 4315-4317; J. Am. Chem. Soc. (1999), 121 (18), 4369-4378.
Subsequent Henry-reaction with a nitroalkyl compound followed by a reduction leads to compounds III, wherein R2 is hydrogen, as described in Tetrahedron Lett. (2003), 44(12), 2557- 2560; J. Mass Spectrometry (2008), 43(4), 528-534; WO 2008/039882; Bioorg. Med. Chem. Lett. (2007), 17(4), 974-977; Chem. Biodiversity (2005), 2(9), 1217-1231 ; J. Org. Chem. (2005), 70(14), 5519-5527; Bioorg. Med. Chem. (2004), 12(15), 4055-4066; or Pestic. Sci. (1995), 44(4), 341 -355.
Otherwise compounds III can be synthesized in analogy to WO 201 1/053835; GB 2059955; WO 2007/020227; WO 2008/046598; WO 201 1/053835; Eur. J. Med. Chem. (2009), 44(5), 2246-2251 ; or J. Med. Chem. (2007), 50(20), 5003-501 1. Phenethylamines III are commercially available or can be prepared as outlined in Scheme 3:
cheme 3
By addition of cyanide, a carbonyl compound AD-1 can be transformed into its cyano hydrine (see e.g. Chemistry A Eur. J. (201 1 ), 17(44), 12276-12279; Eur. J. Org. Chem. (2002), (19), 3243-3249; Synlett (2003), (3), 353-356; Chirality (2009), 21 (9), 836-842; or J. Org. Chem. (2008), 73(18), 7373-7375). Reduction of the nitrile group provides compounds AD-3.
Depending on the reaction conditions, the nitrile in AD-3 can be reduced and the hydroxyl group be removed in one step to furnish compounds III, wherein R2 is hydrogen (see e.g. Justus Liebigs Annalen der Chemie (1949), 564, 49-54, Justus Liebigs Annalen der Chemie (1957), 605, 200-1 1 , J. Chem. Soc. (1959), 1780-2; or WO 2007/020381 ).
Other conditions allow to preserve the hydroxyl group while selectively reducing the nitrile (see e.g. Archiv der Pharmazie (Weinheim, Germany, 201 1 ), 344(6), 372-385; J. Am. Chem. Soc. (1948), 70, 3738-40, J. Am. Chem. Soc. (1933), 55, 2593-7; Tetrahedron (2001 ), 57(40), 8573-8580, Chem. Pharm. Bull. (2003), 51 (6), 702-709). This hydroxyl group can be
transformed in a further step to provide halogen, cyano (Tetrahedron Lett. (2007), 48(38), 6779- 6784), amino or alkoxy groups by nucleophilic substitution in compounds III, wherein R2 is halogen, CN or Ci-C4-alkoxy.
It may be advantageous to couple AD-1 with a heterocycle Het-LG prior to the
abovementioned reactions as shown in Scheme 2.
Phenethylamine compounds III can also be prepared by reductive amination of aldehyde compounds AD-4 as described in Scheme 4.
Scheme 4
AD-4 III
This conversion can be achieved using ammonia and a reducing agent or a metal organic compound or a cyanide source (see e.g. J. Am. Chem. Soc. (201 1 ), 133(33), 12914-12917; Acta Pharmaceutica Suecica (1976), 13(1 ), 65-74; J. Med. Chem. (1976), 19(6), 763-6; or J. Am. Chem. Soc. (1952), 74, 461 1 -15).
Compounds III can also be prepared by transformation of AD-1 to a nitro alkene AD-6 by first reacting it with nitromethane AD-5 preferably in the presence of a base as described in
Scheme 5 (see e.g. J. Org. Chem (2002), 67(14), 4875-4881 ; Eur. J. Med. Chem. (201 1 ), 46(9), 3986-3995; or J. Am. Chem. Soc. (1985), 107(12), 3601 -6):
Scheme 5
Consecutive reduction, e.g. with UAIH4 or hydrogen together with a suitable catalyst, leads to compounds III (Org. Biomol. Chem. (201 1 ), 9(23), 8171 -8177; J. Am. Chem. Soc. (201 1 ), 133(31 ), 12197-12219; or Eur. J. Med. Chem. (2010), 45(1 ), 1 1 -18).
Alternatively, the nitro alkenes AD-6 can be reacted with an alkoxide to give the
corresponding alkoxy compounds (Eur. J. Med. Chem. (201 1 ), 46(9), 3986-3995, Org. Lett. (2006), 8(20), 4481 -4484; Tetrahedron (1999), 55(43), 12493-12514; J. Org. Chem. (1995), 60(13), 4204-12)
Alternatively, the nitro alkenes can be reacted with metal cyanides to give the corresponding cyano compounds (Synlett (2008), (12), 1857-1861 , J. Org. Chem. (1985), 50(20), 3878-81 ). The terminal nitro group can be reduced selectively in the presence of the nitrile to yield compounds III, wherein R2 is Ci-C4-alkoxy or CN, respectively.
It may be advantageous to couple AD-1 with a heterocycle Het-LG prior to the
abovementioned reactions as shown in Scheme 2.
Alternatively, compounds of the formula III can also be prepared by a process as described in Scheme 6:
AD-7
Substituted benzyl nitrile or phenyl acetaldehyde compounds AD-7 can be alkylated
(Tetrahedron (1988), 44(15), 4737-46; US 20080171761 A1 ; Jingxi Huagong Zhongjianti (2010), 40(3), 26-28; or Adv. Synth. Catal. (201 1 ), 353(2+3), 501 -507) once or twice using alkylation agents which carry a suitable leaving group Y and can then be transformed to the
corresponding amine by reduction of the nitrile or imine intermediate respectively (WO
2010/081692; Tetrahedron Lett. (1985), 26(36), 4299-300; J. Org. Chem. (1981 ), 46(4), 783-8; or Tetrahedron (2002), 58(8), 1513-1518). It may be advantageous to couple AD-7 with a heterocycle Het-LG prior to the abovementioned reactions as shown in Scheme 2.
Furthermore, substituted benzyl nitriles AD-9, which are available from benzyl halides AD-8, can be used as intermediates for the preparation of compounds III, wherein R2 is hydrogen, according to scheme 7 by way of reduction with an appropriate reducing agent (e.g. LiAlhU, PhSiHs or H2 and a catalyst) as shown in Scheme 7 (Tetrahedron (201 1 ), 67(42), 8183-8186; WO 201 1/088181 ; Eur. J. Inorg. Chem. (201 1 ), 201 1 (22), 3381 -3386; or WO 2008/124757):
Scheme 7
AD-8 AD-9
Compounds III can also be synthesized by way of hydroboration of substituted alkenes AD- 12 followed by Suzuki coupling using palladium catalysis (J. Org. Chem. (2007), 72(22), 8422- 8426; Org. Lett. (2007), 9(2), 203-206; or J. Am Chem. Soc. (2005), 127(29), 10186-10187) as show in Scheme 8:
Scheme 8
wherein R2 is H
If individual compounds I cannot be obtained by the routes described above, they can be prepared by derivatization of other compounds I.
The N-oxides may be prepared from the compounds I according to conventional oxidation methods, e. g. by treating compounds I with an organic peracid such as metachloroperbenzoic acid (cf. WO 03/64572 or J. Med. Chem. 1995, 38(1 1 ), 1892-1903,); or with inorganic oxidizing agents such as hydrogen peroxide (cf. J. Heterocyc. Chem. 1981 , 18 (7), 1305-1308) or oxone (cf. J. Am. Chem. Soc. 2001 , 123 (25), 5962-5973). The oxidation may lead to pure mono-N- oxides or to a mixture of different N-oxides, which can be separated by conventional methods such as chromatography.
If the synthesis yields mixtures of isomers, a separation is generally not necessarily required since in some cases the individual isomers can be interconverted during work-up for use or during application (e. g. under the action of light, acids or bases). Such conversions may also take place after use, e. g. in the treatment of plants in the treated plant, or in the harmful fungus to be controlled.
In the definitions of the variables given above, collective terms are used which are generally representative for the substituents in question. The term "Cn-Cm" indicates the number of carbon atoms possible in each case in the substituent or substituent moiety in question.
The term "halogen" refers to fluorine, chlorine, bromine and iodine.
The term "Ci-C4-alkyl" refers to a straight-chained or branched saturated hydrocarbon group having 1 to 4 carbon atoms, for example methyl, ethyl, propyl, 1 -methylethyl, butyl, 1 - methylpropyl, 2-methylpropyl, and 1 ,1 -dimethylethyl. Likewise, the term "Ci-C6-alkyl" refers to a straight-chained or branched saturated hydrocarbon group having 1 to 6 carbon atoms.
The term "Ci-C4-haloalkyl" refers to a straight-chained or branched alkyl group having 1 to 4 carbon atoms (as defined above), wherein some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as mentioned above, for example chloromethyl,
bromomethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, dichlorofluoromethyl, chlorodifluoromethyl, 1 -chloroethyl, 1 -bromoethyl, 1 - fluoroethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2-fluoroethyl, 2-chloro- 2,2-difluoroethyl, 2,2-dichloro-2-fluoroethyl, 2,2,2-trichloroethyl and pentafluoroethyl, 2- fluoropropyl, 3-fluoropropyl, 2,2-difluoropropyl, 2,3-difluoropropyl, 2-chloropropyl, 3- chloropropyl, 2,3-dichloropropyl, 2-bromopropyl, 3-bromopropyl, 3,3,3-trifluoropropyl, 3,3,3- trichloropropyl, CH2-C2F5, CF2-C2F5, CF(CF3)2, 1 -(fluoromethyl)-2-fluoroethyl, l -(chloromethyl)- 2-chloroethyl, 1 -(bromomethyl)-2-bromoethyl, 4-fluorobutyl, 4-chlorobutyl, 4-bromobutyl or nonafluorobutyl. Likewise, the term "Ci-C6-haloalkyl" refers to a straight-chained or branched alkyl group having 1 to 6 carbon atoms.
The term "Ci-C4-hydroxyalkyl" refers to a straight-chained or branched alkyl group having 2 to 4 carbon atoms (as defined above), wherein one hydrogen atom in these groups may be replaced by one hydroxy group, for example hydroxym ethyl, 2-hydroxyethyl, 3-hydroxy-n-propyl, or 4-hydroxy-n-butyl.
The term "Ci-C4-alkoxy" refers to a straight-chain or branched alkyl group having 1 to 4 carbon atoms (as defined above) which is bonded via an oxygen, at any position in the alkyl group, for example methoxy, ethoxy, n-propoxy, 1-methylethoxy, butoxy, 1 -methyhpropoxy, 2- methylpropoxy or 1 ,1 -dimethylethoxy. Likewise, the term "Ci-C6-alkoxy" refers to a straight- chain or branched alkyl group having 1 to 6 carbon atoms.
The term "Ci-C4-haloalkoxy" refers to a Ci-C4-alkoxy group as defined above, wherein some or all of the hydrogen atoms may be replaced by halogen atoms as mentioned above, for example, OCH2F, OCHF2, OCF3, OCH2CI, OCHCI2, OCCI3, chlorofluoromethoxy,
dichlorofluoromethoxy, chlorodifluoromethoxy, 2-fluoroethoxy, 2-chloroethoxy, 2-bromoethoxy, 2-iodoethoxy, 2,2-difluoroethoxy, 2,2,2-trifluoroethoxy, 2-chloro-2-fluoroethoxy, 2-chloro-2,2- difluoroethoxy, 2,2-dichloro-2-fluoroethoxy, 2,2,2-trichloroethoxy, OC2F5, 2-fluoropropoxy, 3- fluoropropoxy, 2,2-difluoropropoxy, 2,3-difluoropropoxy, 2-chloropropoxy, 3-chloropropoxy, 2,3- dichloropropoxy, 2-bromopropoxy, 3-bromopropoxy, 3,3,3-trifluoropropoxy, 3,3,3- trichloropropoxy, OCH2-C2F5, OCF2-C2F5, 1 -(CH2F)-2-fluoroethoxy, 1 -(CH2CI)-2-chloroethoxy, 1 -(CH2Br)-2-bromo-,ethoxy, 4-fluorobutoxy, 4-chlorobutoxy, 4-bromobutoxy or
nonafluorobutoxy. Likewise, the term "Ci-C6-haloalkoxy" refers to a Ci-C6-alkoxy group as defined above, wherein some or all of the hydrogen atoms may be replaced by halogen atoms as mentioned above.
The term "Ci-C4-alkoxy-Ci-C4-alkyl" refers to alkyl having 1 to 4 carbon atoms (as defined
above), wherein one hydrogen atom of the alkyl radical is replaced by a Ci-C4-alkoxy group (as defined above). Likewise, the term "Ci-C6-alkoxy-Ci-C4-alkyl" refers to alkyl having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a Ci-C6-alkoxy group (as defined above).
The term "Ci-C4-alkoxy-Ci-C4-alkoxy" refers to an alkoxy radical having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of this alkoxy radical is replaced by a
Ci-C4-alkoxy group (as defined above). Likewise, the term "Ci-C6-alkoxy-Ci-C4-alkoxy" refers to alkoxy having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a Ci-C6-alkoxy group (as defined above).
The term "Ci-C4-haloalkoxy-Ci-C4-alkyl" refers to alkyl having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a
Ci-C4-haloalkoxy group (as defined above). Likewise, the term "Ci-C6-haloalkoxy-Ci-C4-alkyl" refers to alkyl having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a Ci-C6-alkoxy group (as defined above).
The term "Ci-C4-alkylthio" as used herein refers to straight-chain or branched alkyl groups having 1 to 4 carbon atoms (as defined above) bonded via a sulfur atom, at any position in the alkyl group, for example methylthio, ethylthio, propylthio, isopropylthio, and n butylthio.
Likewise, the term "Ci-C6-alkylthio" as used herein refers to straight-chain or branched alkyl groups having 1 to 6 carbon atoms (as defined above) bonded via a sulfur atom. Accordingly, the terms "Ci-C4-haloalkylthio" and "Ci-C6-haloalkylthio" as used herein refer to straight-chain or branched haloalkyi groups having 1 to 4 or 1 to 6 carbon atoms (as defined above) bonded through a sulfur atom, at any position in the haloalkyi group.
The terms "Ci-C4-alkylsulfinyl" or "Ci-C6-alkylsulfinyl" refer to straight-chain or branched alkyl groups having 1 to 4 or 1 to 6 carbon atoms (as defined above) bonded through a -S(=0)- moiety, at any position in the alkyl group, for example m ethyl su If inyl and ethylsulfinyl, and the like. Accordingly, the terms "Ci-C4-haloalkylsulfinyl" and "Ci-C6-haloalkylsulfinyl", respectively, refer to straight-chain or branched haloalkyi groups having 1 to 4 and 1 to 6 carbon atoms (as defined above), respectively, bonded through a -S(=0)- moiety, at any position in the haloalkyi group.
The terms "Ci-C4-alkylsulfonyl" and "Ci-C6-alkylsulfonyl", respectively, refer to straight-chain or branched alkyl groups having 1 to 4 and 1 to 6 carbon atoms (as defined above),
respectively, bonded through a -S(=0)2- moiety, at any position in the alkyl group, for example methylsulfonyl. Accordingly, the terms "Ci-C4-haloalkylsulfonyl" and "Ci-C6-haloalkylsulfonyl", respectively, refer to straight-chain or branched haloalkyi groups having 1 to 4 and 1 to 6 carbon atoms (as defined above), respectively, bonded through a -S(=0)2- moiety, at any position in the haloalkyi group.
The term "Ci-C4-alkylamino" refers to an amino radical carrying one Ci-C4-alkyl group (as defined above) as substituent, for example methylamino, ethylamino, propylamino, 1 - methylethylamino, butylamino, 1 -methylpropylamino, 2-methylpropylamino, 1 ,1 -di- methylethylamino and the like. Likewise, the term "Ci-C6-alkylamino" refers to an amino radical carrying one Ci-C6-alkyl group (as defined above) as substituent.
The term "di(Ci-C4-alkyl)amino" refers to an amino radical carrying two identical or different Ci-C4-alkyl groups (as defined above) as substituents, for example dimethylamino,
diethylamino, di-n-propylamino, diisopropylamino, N-ethyl-N-methylamino, N-(n-propyl)-N-
methylamino, N-(isopropyl)-N methylamino, N-(n-butyl)-N-methylamino, N-(n-pentyl)-N- methylamino, N-(2-butyl)-N methylamino, N-(isobutyl)-N-methylamino, and the like. Likewise, the term "di(Ci-C6-alkyl)amino" refers to an amino radical carrying two identical or different Ci- C6-alkyl groups (as defined above) as substituents.
The term "(Ci-C4-alkoxy)carbonyl" refers to a Ci-C4-alkoxy radical (as defined above) which is attached via a carbonyl group.
The term "di(Ci-C4-alkyl)aminocarbonyl" refers to a di(Ci-C4)alkylamino radical as defined above which is attached via a carbonyl group.
The term "phenoxy" and refers to a phenyl radical which is attached via an oxygen atom. Likewise, the term "phenoxy-Ci-C4-alkyl" and refers to a phenoxy radical which is attached via a Ci-C4-alkyl group (as defined above).
The term "C2-C4-alkenyl" refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 4 carbon atoms and a double bond in any position, such as ethenyl, 1 - propenyl, 2-propenyl (allyl), 1 -methylethenyl, 1 -butenyl, 2-butenyl, 3-butenyl, 1 -methyl-1 - propenyl, 2-methyl-1 -propenyl, 1 -methyl-2-propenyl, 2-methyl-2-propenyl. Likewise, the term "C2-C6-alkenyl" refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 6 carbon atoms and a double bond in any position.
The term "C2-C4-alkynyl" refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 4 carbon atoms and containing at least one triple bond, such as ethynyl, 1 - propynyl, 2-propynyl, 1 -butynyl, 2-butynyl, 3-butynyl, 1 -methyl-2-propynyl. Likewise, the term "C2-C6-alkynyl" refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 6 carbon atoms and at least one triple bond.
The term "Cs-Cs-cycloalkyl" refers to monocyclic saturated hydrocarbon radicals having 3 to 8 carbon ring members, such as cyclopropyl (C3H5), cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl or cyclooctyl.
The term "C3-C8-cycloalkyl-Ci-C4-alkyl" refers to an alkyl radical having 1 to 4 carbon atoms (as defined above), wherein one hydrogen atom of the alkyl radical is replaced by a Cs-Cs- cycloalkyl group (as defined above).
The term "Cs-Cs-cycloalkyloxy" refers to a cycloalkyl radical having 3 to 8 carbon atoms (as defined above), which is bonded via an oxygen.
The term "saturated or partially unsaturated 3-, 4- 5-, 6- or 7-membered carbocycle" is to be understood as meaning both saturated or partially unsaturated carbocycles having 3, 4, 5, 6 or 7 ring members. Examples include cyclopropyl, cyclopentyl, cyclopentenyl, cyclopentadienyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, cycloheptyl, cycloheptenyl, cycloheptadienyl, and the like.
The term " saturated or partially unsaturated 3-, 4-, 5-, 6-, or 7-membered heterocycle, wherein the ring member atoms of the heterocycle include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S", is to be understood as meaning both saturated and partially unsaturated heterocycles, e.g.:
- a 3- or 4-membered saturated heterocycle which contains 1 or 2 heteroatoms from the group consisting of N, O and S as ring members such as oxirane, aziridine, thiirane, oxetane, azetidine, thiethane, [1 ,2]dioxetane, [1 ,2]dithietane, [1 ,2]diazetidine; or
- a 5- or 6-membered saturated or partially unsaturated heterocycle which contains 1 , 2 or 3 heteroatoms from the group consisting of N, O and S as ring members such as 2-
tetrahydrofuranyl, 3-tetrahydrofuranyl, 2-tetrahydrothienyl, 3-tetrahydrothienyl, 2-pyrrolidinyl,
3- pyrrolidinyl, 3-isoxazolidinyl, 4-isoxazolidinyl, 5-isoxazolidinyl, 3-isothiazolidinyl,
4- isothiazolidinyl, 5-isothiazolidinyl, 3-pyrazolidinyl, 4-pyrazolidinyl, 5-pyrazolidinyl, 2- oxazolidinyl, 4-oxazolidinyl, 5-oxazolidinyl, 2-thiazolidinyl, 4-thiazolidinyl, 5-thiazolidinyl, 2-imidazolidinyl, 4-imidazolidinyl, 1 ,2,4-oxadiazolidin-3-yl, 1 ,2,4-oxadiazolidin-5-yl, 1 ,2,4- thiadiazolidin-3-yl, 1 ,2,4-thiadiazolidin-5-yl, 1 ,2,4-triazolidin-3-yl, 1 ,3,4-oxadiazolidin-2-yl, 1 ,3,4-thiadiazolidin-2-yl, 1 ,3,4-triazolidin-2-yl, 2,3-dihydrofur-2-yl, 2,3-dihydrofur-3-yl, 2,4- dihydrofur-2-yl, 2,4-dihydrofur-3-yl, 2,3-dihydrothien-2-yl, 2,3-dihydrothien-3-yl, 2,4-dihydro- thien-2-yl, 2,4-dihydrothien-3-yl, 2-pyrrolin-2-yl, 2-pyrrolin-3-yl, 3-pyrrolin-2-yl, 3-pyrrolin-3-yl, 2-isoxazolin-3-yl, 3-isoxazolin-3-yl, 4-isoxazolin-3-yl, 2-isoxazolin-4-yl, 3-isoxazolin-4-yl, 4- isoxazolin-4-yl, 2-isoxazolin-5-yl, 3-isoxazolin-5-yl, 4-isoxazolin-5-yl, 2-isothiazolin-3-yl,
3- isothiazolin-3-yl, 4-isothiazolin-3-yl, 2-isothiazolin-4-yl, 3-isothiazolin-4-yl, 4-isothiazolin-4- yl, 2-isothiazolin-5-yl, 3-isothiazolin-5-yl, 4-isothiazolin-5-yl, 2,3-dihydropyrazol-1 -yl,
2.3- dihydropyrazol-2-yl, 2,3-dihydropyrazol-3-yl, 2,3-dihydropyrazol-4-yl, 2,3-dihydropyrazol- 5-yl, 3,4-dihydropyrazol-1 -yl, 3,4-dihydropyrazol-3-yl, 3,4-dihydropyrazol-4-yl, 3,4- dihydropyrazol-5-yl, 4,5-dihydropyrazol-1 -yl, 4,5-dihydropyrazol-3-yl, 4,5-dihydropyrazol-4-yl, 4,5-dihydropyrazol-5-yl, 2,3-dihydrooxazol-2-yl, 2,3-dihydrooxazol-3-yl, 2,3-dihydrooxazol-4- yl, 2,3-dihydrooxazol-5-yl, 3,4-dihydrooxazol-2-yl, 3,4-dihydrooxazol-3-yl, 3,4-dihydrooxazol-
4- yl, 3,4-dihydrooxazol-5-yl, 3,4-dihydrooxazol-2-yl, 3,4-dihydrooxazol-3-yl, 3,4-di- hydrooxazol-4-yl, 2-piperidinyl, 3-piperidinyl, 4-piperidinyl, 1 ,3-dioxan-5-yl, 2-tetrahydro- pyranyl, 4-tetrahydropyranyl, 2-tetrahydrothienyl, 3-hexahydropyridazinyl, 4-hexa- hydropyridazinyl, 2-hexahydropyrimidinyl, 4-hexahydropyrimidinyl, 5-hexahydropyrimidinyl, 2-piperazinyl, 1 ,3,5-hexahydrotriazin-2-yl and 1 ,2,4-hexahydrotriazin-3-yl and also the corresponding -ylidene radicals; and
- a 7-membered saturated or partially unsaturated heterocycle such as tetra- and hexahydroazepinyl, such as 2,3,4,5-tetrahydro[1 H]azepin-1 -, -2-, -3-, -4-, -5-,
-6- or -7-yl, 3,4,5,6-tetrahydro[2H]azepin-2-, -3-, -4-, -5-, -6- or -7-yl, 2,3,4,7-tetrahydro- [1 H]azepin-1 -, -2-, -3-, -4-, -5-, -6- or -7-yl, 2,3,6,7-tetrahydro[1 H]azepin-1 -, -2-,
-3-, -4-, -5-, -6- or -7-yl, hexahydroazepin-1 -, -2-, -3- or -4-yl, tetra- and hexahydrooxepinyl such as 2,3,4,5-tetrahydro[1 H]oxepin-2-, -3-, -4-, -5-, -6- or -7-yl, 2,3,4,7-te- trahydro[1 H]oxepin-2-, -3-, -4-, -5-, -6- or -7-yl, 2,3,6,7-tetrahydro[1 H]oxepin-2-, -3-, -4-, -5-, - 6- or -7-yl, hexahydroazepin-1 -, -2-, -3- or -4-yl, tetra- and hexahydro-1 ,3-diazepinyl, tetra- and hexahydro-1 ,4-diazepinyl, tetra- and hexahydro-1 ,3-oxazepinyl, tetra- and hexahydro-
1 .4- oxazepinyl, tetra- and hexahydro-1 ,3-dioxepinyl, tetra- and hexahydro-1 ,4-dioxepinyl and the corresponding -ylidene radicals; and
The term "5-or 6-membered heteroaryl, wherein the ring member atoms of the heteroaryl include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S", refers to, for example,
- a 5-membered heteroaryl such as pyrrol-1 -yl, pyrrol-2-yl, pyrrol-3-yl, thien-2-yl, thien-3-yl, furan-2-yl, furan-3-yl, pyrazol-1 -yl, pyrazol-3-yl, pyrazol-4-yl, pyrazol-5-yl, imidazol-1 -yl, imidazol-2-yl, imidazol-4-yl, imidazol-5-yl, oxazol-2-yl, oxazol-4-yl, oxazol-5-yl, isoxazol-3-yl, isoxazol-4-yl, isoxazol-5-yl, thiazol-2-yl, thiazol-4-yl, thiazol-5-yl, isothiazol-3-yl, isothiazol-4-yl, isothiazol-5-yl, 1 ,2,4-triazolyl-1 -yl, 1 ,2,4-triazol-3-yl 1 ,2,4-triazol-5-yl, 1 ,2,4-oxadiazol-3-yl, 1 ,2,4-oxadiazol-5-yl and 1 ,2,4-thiadiazol-3-yl, 1 ,2,4-thiadiazol-5-yl; or
- a 6-membered heteroaryl, such as pyridin-2-yl, pyridin-3-yl, pyridin-4-yl, pyridazin-3-yl, pyridazin-4-yl, pyrimidin-2-yl, pyrimidin-4-yl, pyrimidin-5-yl, pyrazin-2-yl and 1 ,3,5-triazin-2-yl and 1 ,2,4-triazin-3-yl.
The term "two radicals Rc that are bound to adjacent ring member atoms form together with said ring member atoms a fused cycle" refers to a condensed bicyclic ring system, wherein 5- or 6-membered heteroaryl carries a fused-on 5-, 6- or 7-membered carbocyclic or heterocyclic ring it being possible that these rings are saturated or partially saturated or aromatic.
The term "one or two Chb groups of the abovementioned cycles may be respectively replaced by one or two C(=0) or C(=S) groups" refers to an exchange of carbon atoms from a saturated or partially unsaturated 3-, 4-, 5-, 6- or 7-membered carbocycle or a saturated or partially unsaturated 3-, 4-, 5-, 6- or 7-membered heterocycle, resulting in cycles such as cyclopropanone, cyclopentanone, cyclopropanethione, cyclopentanethione, 5-oxazolone, cyclohexane-1 ,4-dione, cyclohexane-1 ,4-dithione, cyclohex-2-ene-1 ,4-dione or cyclohex-2-ene- 1 ,4-dithione.
In this application, CH3 shall also be understood as Me, CH2CH3 shall also be understood as Et, OCH3 shall also be understood as OMe, OCH2OCH3 shall also be understood as OMeOMe, CH2OCH3 shall also be understood as MeOMe, COOCH3 shall also be understood as COOMe and COOC2H5 shall also be understood as COOEt. As regards the fungicidal activity of the compounds I, preference is given to those
compounds I and where applicable also to compounds of all sub-formulae provided herein, for example formulae 1.1 , 1.2, 1.3, I.A1 and I.A2 and to the intermediates such as compounds II and III wherein the substituents and variables (e.g. Ra2, Ra5, Ra6, R, R1, R2, RA, RB, Het, n, Ra, Rb, Rc, Rd, Re, R', R" and R'") have independently of each other or more preferably in combination the following meanings including also each combination of meanings for any subset of substituents and variables:
According to a one embodiment for compounds I, Ra5 is CH3 or halomethyl; preferably CH3, CH2F, CHF2, CF3, CH2CI, CHC , CCI3, chlorofluoromethyl, dichlorofluoromethyl,
chlorodifluoromethyl or trichloromethyl; in particular CH3 or CF3. In a most preferred embodiment Ra5 is CH3.
Preferably, RA, RB in radicals Ra2 and Ra6 independently of one another preferably are hydrogen or Ci-C4-alkyl.
Preferably, R' in radicals Ra2 and Ra6, which may the same or different, are hydrogen, NH2, Ci-C4-alkyl or Ci-C4-alkoxy.
Preferably, R" in radicals Ra2 and Ra6, which may the same or different, are hydrogen, Ci-C4-alkyl.
Preferably, R'" in radicals Ra2 and Ra6, which may the same or different, are hydrogen or Ci- C4-alkyl.
According to a further embodiment, Ra2 and Ra6 independently of each other are hydrogen, halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy,
Ci-C4-alkylthio, Ci-C4-haloalkylthio, C2-C4-alkenyl, C2-C4-alkynyl, Cs-Cs-cycloalkyl,
C3-C8-cycloalkyloxy or (Ci-C4-alkoxy)carbonyl, more preferably hydrogen, halogen, CN, C1-C2- alkyl, Ci-C2-alkoxy, Ci-C2-alkoxy-Ci-C2-alkyl, Ci-C2-alkoxy-Ci-C2-alkoxy or (C1-C2-
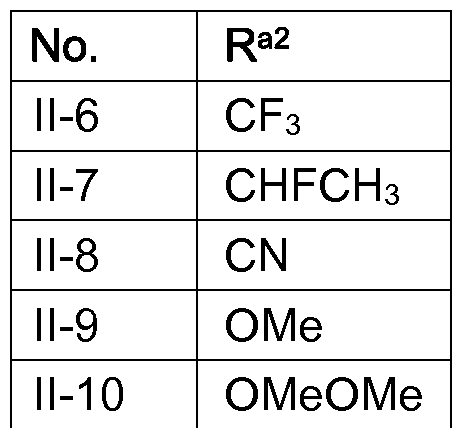
alkoxy)carbonyl; even more preferably hydrogen, F, CI, CH3, CH2CH3, CF3, CHFCH3, CN, OCH3, OCH2OCH3, CH2OCH3, COOCH3 and COOC2H5.
According to a further embodiment, Ra2 and Ra6 independently of each other are halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy,
Ci-C4-alkylthio, Ci-C4-haloalkylthio, C2-C4-alkenyl, C2-C4-alkynyl, Cs-Cs-cycloalkyl,
C3-C8-cycloalkyloxy or (Ci-C4-alkoxy)carbonyl, more preferably halogen, CN,
Ci-C2-alkyl, Ci-C2-alkoxy, Ci-C2-alkoxy-Ci-C2-alkyl, Ci-C2-alkoxy-Ci-C2-alkoxy or (C1-C2- alkoxy)carbonyl, it being possible that one of both, Ra2 or Ra6, can in addition be hydrogen; even more preferably F, CI, CH3, CH2CH3, CF3, CHFCH3, CN, OCH3, OCH2OCH3, CH2OCH3,
COOCH3 and COOC2H5, it being possible that one of both, Ra2 or Ra6, can in addition be hydrogen.
According to a further embodiment, Ra2 and Ra6 independently of each other are halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkoxy or (Ci-C4-alkoxy)carbonyl, and it being possible that one of both, Ra2 or Ra6, can in addition be hydrogen.
According to a preferred embodiment, Ra6 is chlorine, CN, COOEt or COOMe and Ra5 is as defined or preferably defined above. According to a further preferred embodiment, Ra6 is chlorine or COOMe and Ra5 is as defined or preferably defined above. According to another preferred embodiment, Ra6 is chlorine and Ra5 is as defined or preferably defined above.
According to still another preferred embodiment, Ra6 is COOMe and Ra5 is as defined or preferably defined above.
Further preferred embodiments relate to compounds I wherein Ra2, Ra5 and Ra6 independently of each other are in each case one of the following groups 1-1 to I-9 in Table I, groups 11-1 to 11-13 in Table II and groups III-1 to 111-13 in Table III, respectively:
111-10 OMeOMe 111-12 COOMe
111-1 1 MeOMe 111-13 COOEt
Preferably, RA, RB in radical R independently of one another preferably are hydrogen or Ci- C4-alkyl.
Preferably, R' in radical R is hydrogen, OH, NH2, Ci-C4-alkyl or Ci-C4-alkoxy.
In the compounds I according to the invention, R is preferably hydrogen, Ci-C4-alkyl, Ci-C4- alkoxy, Ci-C4-alkoxy-Ci-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl, CN, CH2CN or CH2-0-C(=0)R', wherein R' is hydrogen, Ci-C4-alkyl or Ci-C4-alkoxy; more preferably R is hydrogen, Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkyl, C2-C4-alkenyl or C2-C4-alkynyl; most preferably R is hydrogen or Ci- C4-alkyl, in particular R is hydrogen.
Another preferred embodiment relates to compounds I wherein R is CH3.
According to a further embodiment, the moiety O-Het is bound in para-position to the phenylring, which compounds are of formula 1.1 :
According to a further embodiment, the moiety O-Het is bound in meta-position to the phenylring. According to a further embodiment, the moiety O-Het is bound in ortho-position to the phenylring.
According to a further embodiment, the moiety O-Het is bound in para-position to the phenylring and the substituents R, R1 and R2 are hydrogen, which compounds are of formula I.2:
According to a further embodiment, the moiety O-Het is bound in para-position to the phenylring, R is CH3 and the substituents R1 and R2 are hydrogen, which compounds are of formula 1.3:
According to a further embodiment n is 0, R is hydrogen, the moiety O-Het is bound in para- position to the phenylring and R1 and R2 are both hydrogen, which compounds are of formula I.A1 :
According to a further embodiment n is 0, R is CH3, the moiety O-Het is bound in para- position to the phenylring and R1 and R2 are both hydrogen, which compounds are of formula I.A2:
In the compounds I according to the invention, R1 and R2 independently of each other are preferably hydrogen, halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy or C3-C8-cycloalkyl.
According to a further embodiment, R1 and R2 independently of each other are preferably hydrogen, CN, F, CH3, CH2CH3 or OCH3, more preferably R1 and R2 independently of each other are hydrogen or CH3, even more preferably, R1 and R2 are both hydrogen.
Preferably, RA, RB in radicals R1, R2, Rb and/or Rc, independently of one another preferably are hydrogen or Ci-C4-alkyl.
Preferably, R' in radicals R1, R2 and/or Rc, which may the same or different, are hydrogen, NH2, Ci-C4-alkyl or Ci-C4-alkoxy.
Preferably, R" in radicals R1, R2, Rb and/or Rc, which may the same or different, are hydrogen, Ci-C4-alkyl.
Preferably, R'" in radicals R1, R2, Rb and/or Rc, which may the same or different, are hydrogen or Ci-C4-alkyl.
Further preferred embodiments relate to compounds I wherein the combination of R1 and R2 is in each case one of the following combinations IV-1 to IV-7 in Table IV:
In the compounds I according to the invention, Rb is preferably selected from the group consisting of halogen, CN, NO2, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy and Ci-C4-alkoxy-Ci-C4-alkyl; more preferably Rb is halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl or Ci-C4-alkoxy; most preferably Rb is halogen, CN , CH3, CF3 or OCH3.
According to a further embodiment, n is 1 , 2, 3 or 4 and Rb is selected from the group of halogen, CN, NO2, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy and C1-C4- alkoxy-Ci-C4-alkyl.
In the compounds I according to the invention, n is preferably 0. According to a further embodiment, n is preferably 1. According to a further embodiment, n is preferably 2. According to a further embodiment, n is preferably 3.
One embodiment of the invention relates to compounds I, in which Het is a 6-membered heteroaryl, wherein the ring member atoms of the heteroaryl include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S, and wherin the 6-membered heteroaryl is unsubstituted or carries 1 , 2, 3 or 4 identical or different groups Rc.
If Het is a 6-membered heteroaryl, in one embodiment, Het carries at least one nitrogen as ring member atom. Preference is given to compounds I, in which Het is a pyridyl radical that is selected from pyridin-2-yl, pyridin-3-yl and pyridin-4-yl, and wherein the aforementioned pyridyl radicals are unsubstituted or carry 1 , 2, 3 or 4 identical or different substituents Rc. More preferably, Het is pyridin-2-yl, which is unsubstituted or carries one or two radicals Rc. More preferably, Het is pyridin-4-yl, which is unsubstituted or carries one or two radicals Rc.
Preference is given to compounds I, in which Het is a pyrimidinyl radical that is selected from pyrimidin-2-yl, pyrimidin-4-yl, pyrimidin-5-yl and pyrimidin-6-yl, and wherein the aformentioned pyrimidinyl radicals are unsubstituted or carry 1 , 2 or 3 identical or different substituents Rc.
If Het is a 5-membered heteroaryl, in another embodiment of the invention, Het carries two heteroatoms as ring member atoms. Preference is given to compounds I, in which Het is a pyrazolyl radical that is selected from pyrazol-3-yl, pyrazol-4-yl and pyrazol-5-yl, and wherein the aforementioned pyrazolyl radicals are unsubstituted or carry 1 , 2 or 3 identical or different substituents Rc. Preference is given to compounds I, in which Het is an isoxazolyl radical that is selected from isoxazol-3-yl, isoxazol-4-yl and isoxazol-5-yl, and wherein the aforementioned isoxazolyl radicals are unsubstituted or carry 1 or 2 identical or different substituents Rc.
Preference is given to compounds I, in which Het is an isothiazolyl radical that is selected from isothiazol-3-yl, isothiazol-4-yl and isothiazol-5-yl, and wherein the aforementioned isothiazolyl radicals are unsubstituted or carry 1 or 2 identical or different substituents Rc. Preference is given to compounds I, in which Het is an imidazolyl radical that is selected from imidazol-2-yl, imidazol-4-yl and imidazol-5-yl, and wherein the aforementioned imidazolyl radicals are unsubstituted or carry 1 , 2 or 3 identical or different substituents Rc. Preference is given to compounds I, in which Het is an oxazolyl radical that is selected from oxazol-2-yl, oxazol-4-yl and oxazol-5-yl, and wherein the aforementioned oxazolyl radicals are unsubstituted or carry 1 or 2 identical or different substituents Rc. Preference is given to compounds I, in which Het is a thiazolyl radical that is selected from thiazol-2-yl, thiazol-4-yl and thiazol-5-yl, and wherein the aforementioned thiazolyl radicals are unsubstituted or carry 1 or 2 identical or different substituents Rc.
Preferred embodiments of the invention relate to compounds I, in which the group Het is one of the following radicals H-1 to H-38:
One embodiment of the invention relates to compounds I, wherein Het carries 1 , 2 or 3 radicals Rc. Another embodiment relates to compounds I, wherein Het carries 1 or 2 radicals Rc. A further embodiment relates to compounds I, wherein Het carries one radical Rc. A further embodiment relates to compounds I, wherein Het carries two radicals Rc. A further embodiment relates to compounds I, wherein Het is unsubstituted.
In a further embodiment, two radicals Rc that are bound to adjacent ring member atoms of the Het group do not form together with said ring member atoms any fused cycle.
Preferably, Rc is halogen, CN, d-d-alkyl, d-d-haloalkyl, d-d-alkoxy, d-d-haloalkoxy, d-Ce-alkoxy-d-d-alkyl, C(=0)R', C(=NOR")R"', d-d-cycloalkyl, d-C4-alkyl-C3-C8-cycloalkyl, phenyl, phenoxy, phenoxy-d-C4-alkyl or a 5- or 6-membered heteroaryl, wherein the ring member atoms of the heteroaryl include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S, and wherein the aforementioned cyclic radicals are unsubstituted or carry 1 , 2, 3 or 4 identical or different substituents Rd.
In one embodiment, Rc is halogen and selected from fluorine, chlorine, bromine and iodine and selected from fluorine and chlorine and in particular, Rc is chlorine.
In another embodiment, Rc is CN.
In a further embodiment, Rc is d-C4-alkyl and selected from methyl, ethyl, n-propyl, i-propyl, n-butyl, 1 -methyl-propyl, 2-methyl-propyl and 1 ,1 -dimethylethyl, and selected from methyl, ethyl, n-propyl and i-propyl, and in particular, Rc is methyl.
In a further embodiment, Rc is d-C4-haloalkyl and selected from d-haloalkyl, d-haloalkyl, d-haloalkyl and d-haloalkyl. More preferably, Rc is d-haloalkyl and selected from fluormethyl, difluormethyl, trifluormethyl, chlormethyl, dichlormethyl and trichlormethyl, and in particular, Rc is trifluormethyl.
In a further embodiment, Rc is d-C4-alkoxy and selected from methoxy, ethoxy, n-propyloxy, i-propyloxy, n-butyloxy, 1 -methyl-propyloxy, 2-methyl-propyloxy and 1 ,1 -dimethylethyloxy and in particular from methoxy and ethoxy.
In a further embodiment, Rc is d-C4-haloalkoxy and specifically halomethoxy, such as difluormethoxy, trifluormethoxy, dichlormethoxy and trichlormethoxy, and haloethoxy, such as 2,2-difluorethoxy, 2,2,2-trifluorethoxy, 2,2-dichlorethoxy and 2,2,2-trichlorethoxy, and halo-n- propoxy, halo-i-propoxy, halo-n-butoxy, halo-1 -methyl-propoxy, halo-2-methyl-propoxy or halo- 1 ,1 -dimethylethoxy.
In a further embodiment, Rc is d-d-cycloalkyl, and in particular, Rc is cyclopropyl.
In a further embodiment, Rc is phenyl.
In a further embodiment, Rc is phenoxy.
In a further embodiment, Rc is phenoxy-d-C4-alkyl and selected from phenoxymethyl, 1 - phenoxy-ethyl and 2-phenoxyethyl.
If Rc is present, one embodiment relates to compounds I, wherein Rc carries 1 , 2, 3 or 4 radicals Rd, preferably 1 , 2 or 3 radicals Rd, and more preferably 1 or 2 radicals Rd. In a paricularly preffered embodiment, Rc carries one radical Rd. In another paricularly preferred embodiment, Rc carries two radicals Rd. In a further particularly preferred embodiment the group Rc carries 3 radicals Rd.
In one embodiment, Rd is halogen and selected from fluorine, chlorine, bromine and iodine
and specifically from fluorine and chlorine and in particular, Rc is chlorine.
In another embodiment, Rd is CN.
In a further embodiment, Rd is Ci-C4-alkyl and selected from methyl, ethyl, n-propyl, i-propyl, n-butyl, 1 -methyl-propyl, 2-methyl-propyl and 1 ,1 -dimethylethyl, and preferably selected from methyl, ethyl, n-propyl and i-propyl and in particular, Rd is methyl.
In a further embodiment, Rd is Ci-C4-haloalkyl and selected from Ci-haloalkyl, C2-haloalkyl, C3-haloalkyl and C4-haloalkyl. More preferably, Rc is Ci-haloalkyl and selected from fluormethyl, difluormethyl, trifluormethyl, chlormethyl, dichlormethyl and trichlormethyl, and in particular, Rd is trifluormethyl.
In a further embodiment, Rd is Ci-C4-alkoxy and selected from methoxy, ethoxy, n-propyloxy, i-propyloxy, n-butyloxy, 1 -methyl-propyloxy, 2-methyl-propyloxy and 1 ,1 -dimethylethyloxy and in particular from methoxy and ethoxy.
According to a further embodiment, the present invention relates to compounds of the formula 1.2 wherein:
Ra2 is hydrogen, Ci-C6-alkyl or Ci-C6-alkoxy;
Ra5 is d-Ce-alkyl;
Ra6 is hydrogen, halogen, CN or (Ci-C4-alkoxy)carbonyl ;
n indicates the number of substituents Rb on the phenyl ring and n is 0 or 1 ;
Rb is halogen, Ci-C6-alkyl or Ci-C6-alkoxy;
Het is a pyridinyl or pyrimidinyl wherein the pyridinyl or pyrimidinyl is unsubstituted or carries 1 or 2 groups Rc:
Rc is halogen, Ci-C6-alkyl or Ci-C6-haloalkyl; and
the N-oxides and the agriculturally acceptable salts of the compounds of formula 1.2.
A skilled person will readily understand that the preferences given in connection with compounds of formula I also apply for formulae 1.1 , 1.2, 1.3, I.A1 and I.A2 as defined herein.
With respect to their use, particular preference is given to the compounds of formulae I.A1 and I.A2 compiled in the Tables 1 to 76 below, wherein the definitions for the substituents Ra2, Ra5 and Ra6 are selected from lines 1 to 208 in Table A and wherein the definitions for R are hydrogen or CH3 as described above and wherein the defintions for group Het are selected from H-1 to H-38 in Table H as described above. Here, the groups mentioned in the Tables for a substituent are furthermore, independently of the combination in which they are mentioned, a particularly preferred embodiment of the substituent in question.
Table 1 : Compounds of formula I.A1 , wherein Het is H-1 , and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 2: Compounds of formula I.A1 , wherein Het is H-2, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 3: Compounds of formula I.A1 , wherein Het is H-3, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 4: Compounds of formula I.A1 , wherein Het is H-4, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 5: Compounds of formula I.A1 , wherein Het is H-5, and the meaning of Ra2, Ra5 and
Ra6 for each compound corresponds to one line of Table A.
Table 6: Compounds of formula I.A1 , wherein Het is H-6, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 7: Compounds of formula I.A1 , wherein Het is H-7, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 8: Compounds of formula I.A1 , wherein Het is H-8, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 9: Compounds of formula I.A1 , wherein Het is H-9, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 10: Compounds of formula I .A1 , wherein Het is H-10, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 1 1 : Compounds of formula I .A1 , wherein Het is H-1 1 , and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 12: Compounds of formula I.A1 , wherein Het is H-12, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 13: Compounds of formula I.A1 , wherein Het is H-13, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 14: Compounds of formula I.A1 , wherein Het is H-14, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 15: Compounds of formula I.A1 , wherein Het is H-15, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 16: Compounds of formula I.A1 , wherein Het is H-16, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 17: Compounds of formula I.A1 , wherein Het is H-17, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 18: Compounds of formula I.A1 , wherein Het is H-18, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 19: Compounds of formula I.A1 , wherein Het is H-19, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 20: Compounds of formula I.A1 , wherein Het is H-20, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 21 : Compounds of formula I.A1 , wherein Het is H-21 , and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 22: Compounds of formula I.A1 , wherein Het is H-22, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 23: Compounds of formula I.A1 , wherein Het is H-23, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 24: Compounds of formula I.A1 , wherein Het is H-24, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 25: Compounds of formula I.A1 , wherein Het is H-25, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 26: Compounds of formula I.A1 , wherein Het is H-26, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 27: Compounds of formula I.A1 , wherein Het is H-27, and the meaning of Ra2, Ra5 and
Ra6 for each compound corresponds to one line of Table A.
Table 28: Compounds of formula I.A1 , wherein Het is H-28, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 29: Compounds of formula I.A1 , wherein Het is H-29, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 30: Compounds of formula I.A1 , wherein Het is H-30, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 31 : Compounds of formula I.A1 , wherein Het is H-31 , and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 32: Compounds of formula I.A1 , wherein Het is H-32, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 33: Compounds of formula I.A1 , wherein Het is H-33, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 34: Compounds of formula I.A1 , wherein Het is H-34, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 35: Compounds of formula I.A1 , wherein Het is H-35, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 36: Compounds of formula I.A1 , wherein Het is H-36, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 37: Compounds of formula I.A1 , wherein Het is H-37, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table 38: Compounds of formula I.A1 , wherein Het is H-38, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Tables 39 to 76: Compounds of formula I.A2, wherein Het is defined as in Tables 1 to 38, and the meaning of Ra2, Ra5 and Ra6 for each compound corresponds to one line of Table A.
Table A
line Ra2 Ra5 Ra6
1 Me Me H
2 Et Me H
3 CN Me H
4 OMe Me H
5 OMeOMe Me H
6 MeOMe Me H
7 COOMe Me H
8 COOEt Me H
9 Me Me F
10 Et Me F
1 1 CN Me F
12 OMe Me F
13 OMeOMe Me F
MeOMe Me F
COOMe Me F
COOEt Me F
Me Me CI
Et Me CI
CN Me CI
OMe Me CI
OMeOMe Me CI
MeOMe Me CI
COOMe Me CI
COOEt Me CI
Me Me Me
Et Me Me
CN Me Me
OMe Me Me
OMeOMe Me Me
MeOMe Me Me
COOMe Me Me
COOEt Me Me
Me Me Et
Et Me Et
CN Me Et
OMe Me Et
OMeOMe Me Et
MeOMe Me Et
COOMe Me Et
COOEt Me Et
Me Me CFs
Et Me CFs
CN Me CFs
OMe Me CFs
OMeOMe Me CFs
MeOMe Me CFs
COOMe Me CFs
COOEt Me CFs
Me Me CHFCHs
Et Me CHFCHs
CN Me CHFCHs
OMe Me CHFCHs
OMeOMe Me CHFCHs
MeOMe Me CHFCHs
COOMe Me CHFCHs
COOEt Me CHFCHs
Me Me CN
Et Me CN
CN Me CN
OMe Me CN
OMeOMe Me CN
MeOMe Me CN
COOMe Me CN
COOEt Me CN
Me Me OMe
Et Me OMe
CN Me OMe
OMe Me OMe
OMeOMe Me OMe
MeOMe Me OMe
COOMe Me OMe
COOEt Me OMe
Me Me OMeOMe
Et Me OMeOMe
CN Me OMeOMe
OMe Me OMeOMe
OMeOMe Me OMeOMe
MeOMe Me OMeOMe
COOMe Me OMeOMe
COOEt Me OMeOMe
Me Me MeOMe
Et Me MeOMe
CN Me MeOMe
OMe Me MeOMe
OMeOMe Me MeOMe
86 MeOMe Me MeOMe
87 COOMe Me MeOMe
88 COOEt Me MeOMe
89 Me Me COOMe
90 Et Me COOMe
91 CN Me COOMe
92 OMe Me COOMe
93 OMeOMe Me COOMe
94 MeOMe Me COOMe
95 COOMe Me COOMe
96 COOEt Me COOMe
97 Me Me COOEt
98 Et Me COOEt
99 CN Me COOEt
100 OMe Me COOEt
101 OMeOMe Me COOEt
102 MeOMe Me COOEt
103 COOMe Me COOEt
104 COOEt Me COOEt
105 Me CFs H
106 Et CFs H
107 CN CFs H
108 OMe CFs H
109 OMeOMe CFs H
110 MeOMe CFs H
111 COOMe CFs H
112 COOEt CFs H
113 Me CFs F
114 Et CFs F
115 CN CFs F
116 OMe CFs F
117 OMeOMe CFs F
118 MeOMe CFs F
119 COOMe CFs F
120 COOEt CFs F
121 Me CFs CI
122 Et CFs CI
123 CN CFs CI
124 OMe CFs CI
125 OMeOMe CFs CI
126 MeOMe CFs CI
127 COOMe CFs CI
128 COOEt CFs CI
129 Me CFs Me
130 Et CFs Me
131 CN CFs Me
132 OMe CFs Me
133 OMeOMe CFs Me
134 MeOMe CFs Me
135 COOMe CFs Me
136 COOEt CFs Me
137 Me CFs Et
138 Et CFs Et
139 CN CFs Et
140 OMe CFs Et
141 OMeOMe CFs Et
142 MeOMe CFs Et
143 COOMe CFs Et
144 COOEt CFs Et
145 Me CFs CFs
146 Et CFs CFs
147 CN CFs CFs
148 OMe CFs CFs
149 OMeOMe CFs CFs
150 MeOMe CFs CFs
151 COOMe CFs CFs
152 COOEt CFs CFs
153 Me CFs CHFCHs
154 Et CFs CHFCHs
155 CN CFs CHFCHs
156 OMe CFs CHFCHs
157 OMeOMe CFs CHFCHs
158 MeOMe CFs CHFCHs
159 COOMe CFs CHFCHs
160 COOEt CFs CHFCHs
161 Me CFs CN
162 Et CFs CN
163 CN CFs CN
164 OMe CFs CN
165 OMeOMe CFs CN
166 MeOMe CFs CN
167 COOMe CFs CN
168 COOEt CFs CN
169 Me CFs OMe
170 Et CFs OMe
171 CN CFs OMe
172 OMe CFs OMe
173 OMeOMe CFs OMe
174 MeOMe CFs OMe
175 COOMe CFs OMe
176 COOEt CFs OMe
177 Me CFs OMeOMe
178 Et CFs OMeOMe
179 CN CFs OMeOMe
180 OMe CFs OMeOMe
181 OMeOMe CFs OMeOMe
182 MeOMe CFs OMeOMe
183 COOMe CFs OMeOMe
184 COOEt CFs OMeOMe
185 Me CFs MeOMe
186 Et CFs MeOMe
187 CN CFs MeOMe
188 OMe CFs MeOMe
189 OMeOMe CFs MeOMe
190 MeOMe CFs MeOMe
191 COOMe CFs MeOMe
192 COOEt CFs MeOMe
193 Me CFs COOMe
194 Et CFs COOMe
195 CN CFs COOMe
196 OMe CFs COOMe
197 OMeOMe CFs COOMe
198 MeOMe CFs COOMe
199 COOMe CFs COOMe
200 COOEt CFs COOMe
201 Me CFs COOEt
202 Et CFs COOEt
203 CN CFs COOEt
204 OMe CFs COOEt
205 OMeOMe CFs COOEt
206 MeOMe CFs COOEt
207 COOMe CFs COOEt
208 COOEt CFs COOEt
The compounds I and the compositions according to the invention, respectively, are suitable as fungicides. They are distinguished by an outstanding effectiveness against a broad spectrum of phytopathogenic fungi, including soil-borne fungi, which derive especially from the classes of the Plasmodiophoromycetes, Peronosporomycetes (syn. Oomycetes), Chytridiomycetes, Zygomycetes, Ascomycetes, Basidiomycetes and Deuteromycetes (syn. Fungi imperfecti). Some are systemically effective and they can be used in crop protection as foliar fungicides, fungicides for seed dressing and soil fungicides. Moreover, they are suitable for controlling harmful fungi, which inter alia occur in wood or roots of plants.
The compounds I and the compositions according to the invention are particularly important in the control of a multitude of phytopathogenic fungi on various cultivated plants, such as cereals, e. g. wheat, rye, barley, triticale, oats or rice; beet, e. g. sugar beet or fodder beet; fruits, such as pomes, stone fruits or soft fruits, e. g. apples, pears, plums, peaches, almonds, cherries, strawberries, raspberries, blackberries or gooseberries; leguminous plants, such as lentils, peas, alfalfa or soybeans; oil plants, such as rape, mustard, olives, sunflowers, coconut, cocoa beans, castor oil plants, oil palms, ground nuts or soybeans; cucurbits, such as squashes, cucumber or melons; fiber plants, such as cotton, flax, hemp or jute; citrus fruit, such as oranges, lemons, grapefruits or mandarins; vegetables, such as spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes, cucurbits or paprika; lauraceous plants, such as avocados, cinnamon or camphor; energy and raw material plants, such as corn, soybean, rape, sugar cane or oil palm; corn; tobacco; nuts; coffee; tea; bananas; vines (table grapes and grape juice grape vines); hop; turf; sweet leaf (also called Stevia); natural rubber plants or ornamental and forestry plants, such as flowers, shrubs, broad-leaved trees or evergreens, e. g. conifers; and on the plant propagation material, such as seeds, and the crop material of these plants.
Preferably, compounds I and compositions thereof, respectively are used for controlling a
multitude of fungi on field crops, such as potatoes sugar beets, tobacco, wheat, rye, barley, oats, rice, corn, cotton, soybeans, rape, legumes, sunflowers, coffee or sugar cane; fruits; vines; ornamentals; or vegetables, such as cucumbers, tomatoes, beans or squashes. The term "plant propagation material" is to be understood to denote all the generative parts of the plant such as seeds and vegetative plant material such as cuttings and tubers (e. g.
potatoes), which can be used for the multiplication of the plant. This includes seeds, roots, fruits, tubers, bulbs, rhizomes, shoots, sprouts and other parts of plants, including seedlings and young plants, which are to be transplanted after germination or after emergence from soil.
These young plants may also be protected before transplantation by a total or partial treatment by immersion or pouring.
Preferably, treatment of plant propagation materials with compounds I and compositions thereof, respectively, is used for controlling a multitude of fungi on cereals, such as wheat, rye, barley and oats; rice, corn, cotton and soybeans.
The term "cultivated plants" is to be understood as including plants which have been modified by breeding, mutagenesis or genetic engineering including but not limiting to agricultural biotech products on the market or in development (cf.
http://www.bio.org/speeches/pubs/er/agri_products.asp). Genetically modified plants are plants, which genetic material has been so modified by the use of recombinant DNA techniques that under natural circumstances cannot readily be obtained by cross breeding, mutations or natural recombination. Typically, one or more genes have been integrated into the genetic material of a genetically modified plant in order to improve certain properties of the plant. Such genetic modifications also include but are not limited to targeted post-translational modification of protein(s), oligo- or polypeptides e. g. by glycosylation or polymer additions such as prenylated, acetylated or farnesylated moieties or PEG moieties.
Plants that have been modified by breeding, mutagenesis or genetic engineering, e. g. have been rendered tolerant to applications of specific classes of herbicides, such as auxin herbicides such as dicamba or 2,4-D; bleacher herbicides such as hydroxylphenylpyruvate dioxygenase (HPPD) inhibitors or phytoene desaturase (PDS) inhibittors; acetolactate synthase (ALS) inhibitors such as sulfonyl ureas or imidazolinones; enolpyruvylshikimate-3-phosphate synthase (EPSPS) inhibitors, such as glyphosate; glutamine synthetase (GS) inhibitors such as glufosinate; protoporphyrinogen-IX oxidase inhibitors; lipid biosynthesis inhibitors such as acetyl CoA carboxylase (ACCase) inhibitors; or oxynil (i. e. bromoxynil or ioxynil) herbicides as a result of conventional methods of breeding or genetic engineering. Furthermore, plants have been made resistant to multiple classes of herbicides through multiple genetic modifications, such as resistance to both glyphosate and glufosinate or to both glyphosate and a herbicide from another class such as ALS inhibitors, HPPD inhibitors, auxin herbicides, or ACCase inhibitors. These herbicide resistance technologies are e. g. described in Pest Managem. Sci. 61 , 2005, 246; 61 , 2005, 258; 61 , 2005, 277; 61 , 2005, 269; 61 , 2005, 286; 64, 2008, 326; 64, 2008, 332; Weed Sci. 57, 2009, 108; Austral. J. Agricult. Res. 58, 2007, 708; Science 316, 2007, 1 185; and references quoted therein. Several cultivated plants have been rendered tolerant to herbicides by conventional methods of breeding (mutagenesis), e. g. Clearfield® summer rape (Canola, BASF SE, Germany) being tolerant to imidazolinones, e. g. imazamox, or ExpressSun®
sunflowers (DuPont, USA) being tolerant to sulfonyl ureas, e. g. tribenuron. Genetic engineering methods have been used to render cultivated plants such as soybean, cotton, corn, beets and rape, tolerant to herbicides such as glyphosate and glufosinate, some of which are
commercially available under the trade names RoundupReady® (glyphosate-tolerant,
Monsanto, U.S.A.), Cultivance® (imidazolinone tolerant, BASF SE, Germany) and LibertyLink® (glufosinate-tolerant, Bayer CropScience, Germany).
Furthermore, plants are also covered that are by the use of recombinant DNA techniques capable to synthesize one or more insecticidal proteins, especially those known from the bacterial genus Bacillus, particularly from Bacillus thuringiensis, such as δ-endotoxins, e. g. CrylA(b), CrylA(c), CrylF, CrylF(a2), CryllA(b), CrylllA, CrylllB(bl ) or Cry9c; vegetative insecticidal proteins (VIP), e. g. VIP1 , VIP2, VIP3 or VIP3A; insecticidal proteins of bacteria colonizing nematodes, e. g. Photorhabdus spp. or Xenorhabdus spp.; toxins produced by animals, such as scorpion toxins, arachnid toxins, wasp toxins, or other insect-specific neurotoxins; toxins produced by fungi, such Streptomycetes toxins, plant lectins, such as pea or barley lectins; agglutinins; proteinase inhibitors, such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin or papain inhibitors; ribosome-inactivating proteins (RIP), such as ricin, maize-RIP, abrin, luffin, saporin or bryodin; steroid metabolism enzymes, such as 3- hydroxysteroid oxidase, ecdysteroid-IDP-glycosyl-transferase, cholesterol oxidases, ecdysone inhibitors or HMG-CoA-reductase; ion channel blockers, such as blockers of sodium or calcium channels; juvenile hormone esterase; diuretic hormone receptors (helicokinin receptors); stilben synthase, bibenzyl synthase, chitinases or glucanases. In the context of the present invention these insecticidal proteins or toxins are to be understood expressly also as pre-toxins, hybrid proteins, truncated or otherwise modified proteins. Hybrid proteins are characterized by a new combination of protein domains, (see, e. g. WO 02/015701 ). Further examples of such toxins or genetically modified plants capable of synthesizing such toxins are disclosed, e. g., in
EP-A 374 753, WO 93/007278, WO 95/34656, EP-A 427 529, EP-A 451 878, WO 03/18810 und WO 03/52073. The methods for producing such genetically modified plants are generally known to the person skilled in the art and are described, e. g. in the publications mentioned above. These insecticidal proteins contained in the genetically modified plants impart to the plants producing these proteins tolerance to harmful pests from all taxonomic groups of athropods, especially to beetles (Coeloptera), two-winged insects (Diptera), and moths (Lepidoptera) and to nematodes (Nematoda). Genetically modified plants capable to synthesize one or more insecticidal proteins are, e. g., described in the publications mentioned above, and some of which are commercially available such as YieldGard® (corn cultivars producing the CrylAb toxin), YieldGard® Plus (corn cultivars producing CrylAb and Cry3Bb1 toxins), Starlink® (corn cultivars producing the Cry9c toxin), Herculex® RW (corn cultivars producing Cry34Ab1 , Cry35Ab1 and the enzyme Phosphinothricin-N-Acetyltransferase [PAT]); NuCOTN® 33B (cotton cultivars producing the CrylAc toxin), Bollgard® I (cotton cultivars producing the CrylAc toxin), Bollgard® II (cotton cultivars producing CrylAc and Cry2Ab2 toxins); VIPCOT® (cotton cultivars producing a VIP-toxin); NewLeaf® (potato cultivars producing the Cry3A toxin); Bt- Xtra®, NatureGard®, KnockOut®, BiteGard®, Protecta®, Bt1 1 (e. g. Agrisure® CB) and Bt176 from Syngenta Seeds SAS, France, (corn cultivars producing the CrylAb toxin and PAT enyzme), MIR604 from Syngenta Seeds SAS, France (corn cultivars producing a modified version of the Cry3A toxin, c.f. WO 03/018810), MON 863 from Monsanto Europe S.A., Belgium
(corn cultivars producing the Cry3Bb1 toxin), IPC 531 from Monsanto Europe S.A., Belgium (cotton cultivars producing a modified version of the CrylAc toxin) and 1507 from Pioneer Overseas Corporation, Belgium (corn cultivars producing the Cry1 F toxin and PAT enzyme).
Furthermore, plants are also covered that are by the use of recombinant DNA techniques capable to synthesize one or more proteins to increase the resistance or tolerance of those plants to bacterial, viral or fungal pathogens. Examples of such proteins are the so-called "pathogenesis-related proteins" (PR proteins, see, e. g. EP-A 392 225), plant disease resistance genes (e. g. potato cultivars, which express resistance genes acting against Phytophthora infestans derived from the mexican wild potato Solanum bulbocastanum) or T4-lysozym (e. g. potato cultivars capable of synthesizing these proteins with increased resistance against bacteria such as Erwinia amylvora). The methods for producing such genetically modified plants are generally known to the person skilled in the art and are described, e. g. in the publications mentioned above.
Furthermore, plants are also covered that are by the use of recombinant DNA techniques capable to synthesize one or more proteins to increase the productivity (e. g. bio mass production, grain yield, starch content, oil content or protein content), tolerance to drought, salinity or other growth-limiting environmental factors or tolerance to pests and fungal, bacterial or viral pathogens of those plants.
Furthermore, plants are also covered that contain by the use of recombinant DNA
techniques a modified amount of substances of content or new substances of content, specifically to improve human or animal nutrition, e. g. oil crops that produce health-promoting long-chain omega-3 fatty acids or unsaturated omega-9 fatty acids (e. g. Nexera® rape, DOW Agro Sciences, Canada).
Furthermore, plants are also covered that contain by the use of recombinant DNA
techniques a modified amount of substances of content or new substances of content, specifically to improve raw material production, e. g. potatoes that produce increased amounts of amylopectin (e. g. Amflora® potato, BASF SE, Germany).
The compounds I and compositions thereof, respectively, are particularly suitable for controlling the following plant diseases:
Albugo spp. (white rust) on ornamentals, vegetables (e. g. A. Candida) and sunflowers (e. g. A. tragopogonis); Alternaria spp. (Alternaria leaf spot) on vegetables, rape (A. brassicola or brassicae), sugar beets (A. tenuis), fruits, rice, soybeans, potatoes (e. g. A. solani or A.
alternata), tomatoes (e. g. A. solani or A. alternata) and wheat; Aphanomyces spp. on sugar beets and vegetables; Ascochyta spp. on cereals and vegetables, e. g. A. tritici (anthracnose) on wheat and A. hordei on barley; Bipolaris and Drechslera spp. (teleomorph: Cochliobolus spp.), e. g. Southern leaf blight (D. maydis) or Northern leaf blight (B. zeicola) on corn, e. g. spot blotch (B. sorokiniana) on cereals and e.g. B. oryzae on rice and turfs; Blumeria (formerly Erysiphe) graminis (powdery mildew) on cereals (e. g. on wheat or barley); Botrytis cinerea (teleomorph: Botryotinia fuckeliana: grey mold) on fruits and berries (e. g. strawberries), vegetables (e. g. lettuce, carrots, celery and cabbages), rape, flowers, vines, forestry plants and wheat; Bremia lactucae (downy mildew) on lettuce; Ceratocystis (syn. Ophiostoma) spp. (rot or wilt) on broad-leaved trees and evergreens, e. g. C. ulmi (Dutch elm disease) on elms;
Cercospora spp. (Cercospora leaf spots) on corn (e.g. Gray leaf spot: C. zeae-maydis), rice, sugar beets (e. g. C. beticola), sugar cane, vegetables, coffee, soybeans (e. g. C. sojina or C.
kikuchii) and rice; Cladosporium spp. on tomatoes (e. g. C. fulvum: leaf mold) and cereals, e. g. C. herbarum (black ear) on wheat; Claviceps purpurea (ergot) on cereals; Cochliobolus
(anamorph: Helminthosporium of Bipolaris) spp. (leaf spots) on corn (C. carbonum), cereals (e. g. C. sativus, anamorph: B. sorokiniana) and rice (e. g. C. miyabeanus, anamorph: H.
oryzae); Colletotrichum (teleomorph: Glomerella) spp. (anthracnose) on cotton (e. g. C.
gossypii), corn (e. g. C. graminicola: Anthracnose stalk rot), soft fruits, potatoes (e. g. C.
coccodes: black dot), beans (e. g. C. lindemuthianum) and soybeans (e. g. C. truncatum or C. gloeosporioides); Corticium spp., e. g. C. sasakii (sheath blight) on rice; Corynespora cassiicola (leaf spots) on soybeans and ornamentals; Cycloconium spp., e. g. C. oleaginum on olive trees; Cylindrocarpon spp. (e. g. fruit tree canker or young vine decline, teleomorph: Nectria or Neonectria spp.) on fruit trees, vines (e. g. C. liriodendri, teleomorph: Neonectria liriodendri: Black Foot Disease) and ornamentals; Dematophora (teleomorph: Rosellinia) necatrix (root and stem rot) on soybeans; Diaporthe spp., e. g. D. phaseolorum (damping off) on soybeans;
Drechslera (syn. Helminthosporium, teleomorph: Pyrenophora) spp. on corn, cereals, such as barley (e. g. D. teres, net blotch) and wheat (e. g. D. tritici-repentis: tan spot), rice and turf; Esca (dieback, apoplexy) on vines, caused by Formitiporia (syn. Phellinus) punctata, F. mediterranea, Phaeomoniella chlamydospora (earlier Phaeoacremonium chlamydosporum),
Phaeoacremonium aleophilum and/or Botryosphaeria obtusa; Elsinoe spp. on pome fruits (E. pyri), soft fruits (E. veneta: anthracnose) and vines (E. ampelina: anthracnose); Entyloma oryzae (leaf smut) on rice; Epicoccum spp. (black mold) on wheat; Erysiphe spp. (powdery mildew) on sugar beets (E. betae), vegetables (e. g. E. pisi), such as cucurbits (e. g. E.
cichoracearum), cabbages, rape (e. g. E. cruciferarum); Eutypa lata (Eutypa canker or dieback, anamorph: Cytosporina lata, syn. Libertella blepharis) on fruit trees, vines and ornamental woods; Exserohilum (syn. Helminthosporium) spp. on corn (e. g. E. turcicum); Fusarium
(teleomorph: Gibberella) spp. (wilt, root or stem rot) on various plants, such as F. graminearum or F. culmorum (root rot, scab or head blight) on cereals (e. g. wheat or barley), F. oxysporum on tomatoes, F. solani on soybeans and F. verticillioides on corn; Gaeumannomyces graminis (take-all) on cereals (e. g. wheat or barley) and corn; Gibberella spp. on cereals (e. g. G. zeae) and rice (e. g. G. fujikuroi: Bakanae disease); Glomerella cingulata on vines, pome fruits and other plants and G. gossypii on cotton; Grainstaining complex on rice; Guignardia bidwellii
(black rot) on vines; Gymnosporangium spp. on rosaceous plants and junipers, e. g. G. sabinae (rust) on pears; Helminthosporium spp. (syn. Drechslera, teleomorph: Cochliobolus) on corn, cereals and rice; Hemileia spp., e. g. H. vastatrix (coffee leaf rust) on coffee; Isariopsis clavispora (syn. Cladosporium vitis) on vines; Macrophomina phaseolina (syn. phaseoli) (root and stem rot) on soybeans and cotton; Microdochium (syn. Fusarium) nivale (pink snow mold) on cereals (e. g. wheat or barley); Microsphaera diffusa (powdery mildew) on soybeans;
Monilinia spp., e. g. M. laxa, M. fructicola and M. fructigena (bloom and twig blight, brown rot) on stone fruits and other rosaceous plants; Mycosphaerella spp. on cereals, bananas, soft fruits and ground nuts, such as e. g. M. graminicola (anamorph: Septoria tritici, Septoria blotch) on wheat or M. fijiensis (black Sigatoka disease) on bananas; Peronospora spp. (downy mildew) on cabbage (e. g. P. brassicae), rape (e. g. P. parasitica), onions (e. g. P. destructor), tobacco (P. tabacina) and soybeans (e. g. P. manshurica); Phakopsora pachyrhizi and P. meibomiae (soybean rust) on soybeans; Phialophora spp. e. g. on vines (e. g. P. tracheiphila and P.
tetraspora) and soybeans (e. g. P. gregata: stem rot); Phoma lingam (root and stem rot) on rape
and cabbage and P. betae (root rot, leaf spot and damping-off) on sugar beets; Phomopsis spp. on sunflowers, vines (e. g. P. viticola: can and leaf spot) and soybeans (e. g. stem rot: P.
phaseoli, teleomorph: Diaporthe phaseolorum); Physoderma maydis (brown spots) on corn; Phytophthora spp. (wilt, root, leaf, fruit and stem root) on various plants, such as paprika and cucurbits (e. g. P. capsici), soybeans (e. g. P. megasperma, syn. P. sojae), potatoes and tomatoes (e. g. P. infestans: late blight) and broad-leaved trees (e. g. P. ramorum: sudden oak death); Plasmodiophora brassicae (club root) on cabbage, rape, radish and other plants;
Plasmopara spp., e. g. P. viticola (grapevine downy mildew) on vines and P. halstedii on sunflowers; Podosphaera spp. (powdery mildew) on rosaceous plants, hop, pome and soft fruits, e. g. P. leucotricha on apples; Polymyxa spp., e. g. on cereals, such as barley and wheat (P. graminis) and sugar beets (P. betae) and thereby transmitted viral diseases;
Pseudocercosporella herpotrichoides (eyespot, teleomorph: Tapesia yallundae) on cereals, e. g. wheat or barley; Pseudoperonospora (downy mildew) on various plants, e. g. P. cubensis on cucurbits or P. humili on hop; Pseudopezicula tracheiphila (red fire disease or .rotbrenner', anamorph: Phialophora) on vines; Puccinia spp. (rusts) on various plants, e. g. P. triticina
(brown or leaf rust), P. striiformis (stripe or yellow rust), P. hordei (dwarf rust), P. graminis (stem or black rust) or P. recondita (brown or leaf rust) on cereals, such as e. g. wheat, barley or rye, P. kuehnii (orange rust) on sugar cane and P. asparagi on asparagus; Pyrenophora (anamorph: Drechslera) tritici-repentis (tan spot) on wheat or P. teres (net blotch) on barley; Pyricularia spp., e. g. P. oryzae (teleomorph: Magnaporthe grisea, rice blast) on rice and P. grisea on turf and cereals; Pythium spp. (damping-off) on turf, rice, corn, wheat, cotton, rape, sunflowers, soybeans, sugar beets, vegetables and various other plants (e. g. P. ultimum or P. aphani- dermatum); Ramularia spp., e. g. R. collo-cygni (Ramularia leaf spots, Physiological leaf spots) on barley and R. beticola on sugar beets; Rhizoctonia spp. on cotton, rice, potatoes, turf, corn, rape, potatoes, sugar beets, vegetables and various other plants, e. g. R. solani (root and stem rot) on soybeans, R. solani (sheath blight) on rice or R. cerealis (Rhizoctonia spring blight) on wheat or barley; Rhizopus stolonifer (black mold, soft rot) on strawberries, carrots, cabbage, vines and tomatoes; Rhynchosporium secalis (scald) on barley, rye and triticale; Sarocladium oryzae and S. attenuatum (sheath rot) on rice; Sclerotinia spp. (stem rot or white mold) on vegetables and field crops, such as rape, sunflowers (e. g. S. sclerotiorum) and soybeans (e. g. S. rolfsii or S. sclerotiorum); Septoria spp. on various plants, e. g. S. glycines (brown spot) on soybeans, S. tritici (Septoria blotch) on wheat and S. (syn. Stagonospora) nodorum
(Stagonospora blotch) on cereals; Uncinula (syn. Erysiphe) necator (powdery mildew, anamorph: Oidium tuckeri) on vines; Setospaeria spp. (leaf blight) on corn (e. g. S. turcicum, syn. Helminthosporium turcicum) and turf; Sphacelotheca spp. (smut) on corn, (e. g. S. reiliana: head smut), sorghum und sugar cane; Sphaerotheca fuliginea (powdery mildew) on cucurbits; Spongospora subterranea (powdery scab) on potatoes and thereby transmitted viral diseases; Stagonospora spp. on cereals, e. g. S. nodorum (Stagonospora blotch, teleomorph:
Leptosphaeria [syn. Phaeosphaeria] nodorum) on wheat; Synchytrium endobioticum on potatoes (potato wart disease); Taphrina spp., e. g. T. deformans (leaf curl disease) on peaches and T. pruni (plum pocket) on plums; Thielaviopsis spp. (black root rot) on tobacco, pome fruits, vegetables, soybeans and cotton, e. g. T. basicola (syn. Chalara elegans); Tilletia spp.
(common bunt or stinking smut) on cereals, such as e. g. T. tritici (syn. T. caries, wheat bunt) and T. controversa (dwarf bunt) on wheat; Typhula incarnata (grey snow mold) on barley or
wheat; Urocystis spp., e. g. U. occulta (stem smut) on rye; Uromyces spp. (rust) on vegetables, such as beans (e. g. U. appendiculatus, syn. U. phaseoli) and sugar beets (e. g. U. betae); Ustilago spp. (loose smut) on cereals (e. g. U. nuda and U. avaenae), corn (e. g. U. maydis: corn smut) and sugar cane; Venturia spp. (scab) on apples (e. g. V. inaequalis) and pears; and Verticillium spp. (wilt) on various plants, such as fruits and ornamentals, vines, soft fruits, vegetables and field crops, e. g. V. dahliae on strawberries, rape, potatoes and tomatoes.
The compounds I and compositions thereof, respectively, are also suitable for controlling harmful fungi in the protection of stored products or harvest and in the protection of materials. The term "protection of materials" is to be understood to denote the protection of technical and non-living materials, such as adhesives, glues, wood, paper and paperboard, textiles, leather, paint dispersions, plastics, colling lubricants, fiber or fabrics, against the infestation and destruction by harmful microorganisms, such as fungi and bacteria. As to the protection of wood and other materials, the particular attention is paid to the following harmful fungi: Ascomycetes such as Ophiostoma spp., Ceratocystis spp., Aureobasidium pullulans, Sclerophoma spp., Chaetomium spp., Humicola spp., Petriella spp., Trichurus spp.; Basidiomycetes such as Coniophora spp., Coriolus spp., Gloeophyllum spp., Lentinus spp., Pleurotus spp., Poria spp., Serpula spp. and Tyromyces spp., Deuteromycetes such as Aspergillus spp., Cladosporium spp., Penicillium spp., Trichorma spp., Alternaria spp., Paecilomyces spp. and Zygomycetes such as Mucor spp., and in addition in the protection of stored products and harvest the following yeast fungi are worthy of note: Candida spp. and Saccharomyces cerevisae.
The compounds I and compositions thereof, resepectively, may be used for improving the health of a plant. The invention also relates to a method for improving plant health by treating a plant, its propagation material and/or the locus where the plant is growing or is to grow with an effective amount of compounds I and compositions thereof, respectively.
The term "plant health" is to be understood to denote a condition of the plant and/or its products which is determined by several indicators alone or in combination with each other such as yield (e. g. increased biomass and/or increased content of valuable ingredients), plant vigor (e. g. improved plant growth and/or greener leaves ("greening effect")), quality (e. g. improved content or composition of certain ingredients) and tolerance to abiotic and/or biotic stress. The above identified indicators for the health condition of a plant may be interdependent or may result from each other. The compounds of the formula (I) can be present in different crystal modifications whose biological activity may differ. They are likewise subject matter of the present invention.
The compounds I are employed as such or in form of compositions by treating the fungi or the plants, plant propagation materials, such as seeds, soil, surfaces, materials or rooms to be protected from fungal attack with a fungicidally effective amount of the active substances. The application can be carried out both before and after the infection of the plants, plant propagation materials, such as seeds, soil, surfaces, materials or rooms by the fungi.
Plant propagation materials may be treated with compounds I as such or a composition comprising at least one compound I prophylactically either at or before planting or transplanting.
The invention also relates to agrochemical compositions comprising an auxiliary and at least one compound I according to the invention.
An agrochemical composition comprises a fungicidally effective amount of a compound I. The term "effective amount" denotes an amount of the composition or of the compounds I, which is sufficient for controlling harmful fungi on cultivated plants or in the protection of materials and which does not result in a substantial damage to the treated plants. Such an amount can vary in a broad range and is dependent on various factors, such as the fungal species to be controlled, the treated cultivated plant or material, the climatic conditions and the specific compound I used.
The compounds I, their N-oxides and salts can be converted into customary types of agrochemical compositions, e. g. solutions, emulsions, suspensions, dusts, powders, pastes, granules, pressings, capsules, and mixtures thereof. Examples for composition types are suspensions (e.g. SC, OD, FS), emulsifiable concentrates (e.g. EC), emulsions (e.g. EW, EO, ES, ME), capsules (e.g. CS, ZC), pastes, pastilles, wettable powders or dusts (e.g. WP, SP, WS, DP, DS), pressings (e.g. BR, TB, DT), granules (e.g. WG, SG, GR, FG, GG, MG), insecticidal articles (e.g. LN), as well as gel formulations for the treatment of plant propagation materials such as seeds (e.g. GF). These and further compositions types are defined in the "Catalogue of pesticide formulation types and international coding system", Technical
Monograph No. 2, 6th Ed. May 2008, CropLife International.
The compositions are prepared in a known manner, such as described by Mollet and Grubemann, Formulation technology, Wiley VCH, Weinheim, 2001 ; or Knowles, New
developments in crop protection product formulation, Agrow Reports DS243, T&F Informa, London, 2005.
Examples for suitable auxiliaries are solvents, liquid carriers, solid carriers or fillers, surfactants, dispersants, emulsifiers, wetters, adjuvants, solubilizers, penetration enhancers, protective colloids, adhesion agents, thickeners, humectants, repellents, attractants, feeding stimulants, compatibilizers, bactericides, anti-freezing agents, anti-foaming agents, colorants, tackifiers and binders.
Suitable solvents and liquid carriers are water and organic solvents, such as mineral oil fractions of medium to high boiling point, e.g. kerosene, diesel oil; oils of vegetable or animal origin; aliphatic, cyclic and aromatic hydrocarbons, e. g. toluene, paraffin,
tetrahydronaphthalene, alkylated naphthalenes; alcohols, e.g. ethanol, propanol, butanol, benzylalcohol, cyclohexanol; glycols; dimethyl sulfoxide (DMSO); ketones, e.g. cyclohexanone; esters, e.g. lactates, carbonates, fatty acid esters, gamma-butyrolactone; fatty acids;
phosphonates; amines; amides, e.g. N-methylpyrrolidone, fatty acid dimethylamides; and mixtures thereof.
Suitable solid carriers or fillers are mineral earths, e.g. silicates, silica gels, talc, kaolins, limestone, lime, chalk, clays, dolomite, diatomaceous earth, bentonite, calcium sulfate, magnesium sulfate, magnesium oxide; polysaccharide powders, e.g. cellulose, starch;
fertilizers, e.g. ammonium sulfate, ammonium phosphate, ammonium nitrate, ureas; products of vegetable origin, e.g. cereal meal, tree bark meal, wood meal, nutshell meal, and mixtures thereof.
Suitable surfactants are surface-active compounds, such as anionic, cationic, nonionic and amphoteric surfactants, block polymers, polyelectrolytes, and mixtures thereof. Such surfactants
can be used as emusifier, dispersant, solubilizer, wetter, penetration enhancer, protective colloid, or adjuvant. Examples of surfactants are listed in McCutcheon's, Vol.1 : Emulsifiers & Detergents, McCutcheon's Directories, Glen Rock, USA, 2008 (International Ed. or North American Ed.).
Suitable anionic surfactants are alkali, alkaline earth or ammonium salts of sulfonates, sulfates, phosphates, carboxylates, and mixtures thereof. Examples of sulfonates are alkylarylsulfonates, diphenylsulfonates, alpha-olefin sulfonates, lignine sulfonates, sulfonates of fatty acids and oils, sulfonates of ethoxylated alkylphenols, sulfonates of alkoxylated
arylphenols, sulfonates of condensed naphthalenes, sulfonates of dodecyl- and
tridecylbenzenes, sulfonates of naphthalenes and alkylnaphthalenes, sulfosuccinates or sulfosuccinamates. Examples of sulfates are sulfates of fatty acids and oils, of ethoxylated alkylphenols, of alcohols, of ethoxylated alcohols, or of fatty acid esters. Examples of phosphates are phosphate esters. Examples of carboxylates are alkyl carboxylates, and carboxylated alcohol or alkylphenol ethoxylates.
Suitable nonionic surfactants are alkoxylates, N-subsituted fatty acid amides, amine oxides, esters, sugar-based surfactants, polymeric surfactants, and mixtures thereof. Examples of alkoxylates are compounds such as alcohols, alkylphenols, amines, amides, arylphenols, fatty acids or fatty acid esters which have been alkoxylated with 1 to 50 equivalents. Ethylene oxide and/or propylene oxide may be employed for the alkoxylation, preferably ethylene oxide.
Examples of N-subsititued fatty acid amides are fatty acid glucamides or fatty acid
alkanolamides. Examples of esters are fatty acid esters, glycerol esters or monoglycerides. Examples of sugar-based surfactants are sorbitans, ethoxylated sorbitans, sucrose and glucose esters or alkylpolyglucosides. Examples of polymeric surfactants are home- or copolymers of vinylpyrrolidone, vinylalcohols, or vinylacetate.
Suitable cationic surfactants are quaternary surfactants, for example quaternary ammonium compounds with one or two hydrophobic groups, or salts of long-chain primary amines. Suitable amphoteric surfactants are alkylbetains and imidazolines. Suitable block polymers are block polymers of the A-B or A-B-A type comprising blocks of polyethylene oxide and polypropylene oxide, or of the A-B-C type comprising alkanol, polyethylene oxide and polypropylene oxide. Suitable polyelectrolytes are polyacids or polybases. Examples of polyacids are alkali salts of polyacrylic acid or polyacid comb polymers. Examples of polybases are polyvinylamines or polyethyleneamines.
Suitable adjuvants are compounds, which have a neglectable or even no pesticidal activity themselves, and which improve the biological performance of the compound I on the target. Examples are surfactants, mineral or vegetable oils, and other auxilaries. Further examples are listed by Knowles, Adjuvants and additives, Agrow Reports DS256, T&F Informa UK, 2006, chapter 5.
Suitable thickeners are polysaccharides (e.g. xanthan gum, carboxymethylcellulose), anorganic clays (organically modified or unmodified), polycarboxylates, and silicates.
Suitable bactericides are bronopol and isothiazolinone derivatives such as alkyliso- thiazolinones and benzisothiazolinones.
Suitable anti-freezing agents are ethylene glycol, propylene glycol, urea and glycerin.
Suitable anti-foaming agents are silicones, long chain alcohols, and salts of fatty acids. Suitable colorants (e.g. in red, blue, or green) are pigments of low water solubility and water-
soluble dyes. Examples are inorganic colorants (e.g. iron oxide, titan oxide, iron hexacyanoferrate) and organic colorants (e.g. alizarin-, azo- and phthalocyanine colorants).
Suitable tackifiers or binders are polyvinylpyrrolidons, polyvinylacetates, polyvinyl alcohols, polyacrylates, biological or synthetic waxes, and cellulose ethers.
Examples for composition types and their preparation are:
i) Water-soluble concentrates (SL, LS)
10-60 wt% of a compound I and 5-15 wt% wetting agent (e.g. alcohol alkoxylates) are dissolved in water and/or in a water-soluble solvent (e.g. alcohols) up to 100 wt%. The active substance dissolves upon dilution with water.
ii) Dispersible concentrates (DC)
5-25 wt% of a compound I and 1 -10 wt% dispersant (e. g. polyvinylpyrrolidone) are dissolved in up to 100 wt% organic solvent (e.g. cyclohexanone). Dilution with water gives a dispersion.
iii) Emulsifiable concentrates (EC)
15-70 wt% of a compound I and 5-10 wt% emulsifiers (e.g. calcium dodecylben- zenesulfonate and castor oil ethoxylate) are dissolved in up to 100 wt% water-insoluble organic solvent (e.g. aromatic hydrocarbon). Dilution with water gives an emulsion.
iv)Emulsions (EW, EO, ES)
5-40 wt% of a compound I and 1 -10 wt% emulsifiers (e.g. calcium dodecylbenzenesulfonate and castor oil ethoxylate) are dissolved in 20-40 wt% water-insoluble organic solvent (e.g. aromatic hydrocarbon). This mixture is introduced into up to 100 wt% water by means of an emulsifying machine and made into a homogeneous emulsion. Dilution with water gives an emulsion.
v) Suspensions (SC, OD, FS)
In an agitated ball mill, 20-60 wt% of a compound I are comminuted with addition of 2-10 wt% dispersants and wetting agents (e.g. sodium lignosulfonate and alcohol ethoxylate), 0.1 -2 wt% thickener (e.g. xanthan gum) and up to 100 wt% water to give a fine active substance suspension. Dilution with water gives a stable suspension of the active substance. For FS type composition up to 40 wt% binder (e.g. polyvinylalcohol) is added.
vi) Water-dispersible granules and water-soluble granules (WG, SG)
50-80 wt% of a compound I are ground finely with addition of up to 100 wt% dispersants and wetting agents (e.g. sodium lignosulfonate and alcohol ethoxylate) and prepared as water- dispersible or water-soluble granules by means of technical appliances (e. g. extrusion, spray tower, fluidized bed). Dilution with water gives a stable dispersion or solution of the active substance.
vii) Water-dispersible powders and water-soluble powders (WP, SP, WS)
50-80 wt% of a compound I are ground in a rotor-stator mill with addition of 1 -5 wt% dispersants (e.g. sodium lignosulfonate), 1 -3 wt% wetting agents (e.g. alcohol ethoxylate) and up to 100 wt% solid carrier, e.g. silica gel. Dilution with water gives a stable dispersion or solution of the active substance.
viii) Gel (GW, GF)
In an agitated ball mill, 5-25 wt% of a compound I are comminuted with addition of 3-10 wt% dispersants (e.g. sodium lignosulfonate), 1 -5 wt% thickener (e.g. carboxymethylcellulose) and
up to 100 wt% water to give a fine suspension of the active substance. Dilution with water gives a stable suspension of the active substance.
ix) Microemulsion (ME)
5-20 wt% of a compound I are added to 5-30 wt% organic solvent blend (e.g. fatty acid dimethylamide and cyclohexanone), 10-25 wt% surfactant blend (e.g. alcohol ethoxylate and arylphenol ethoxylate), and water up to 100 %. This mixture is stirred for 1 h to produce spontaneously a thermodynamically stable microemulsion.
x) Microcapsules (CS)
An oil phase comprising 5-50 wt% of a compound I, 0-40 wt% water insoluble organic solvent (e.g. aromatic hydrocarbon), 2-15 wt% acrylic monomers (e.g. methylmethacrylate, methacrylic acid and a di- or triacrylate) are dispersed into an aqueous solution of a protective colloid (e.g. polyvinyl alcohol). Radical polymerization initiated by a radical initiator results in the formation of poly(meth)acrylate microcapsules. Alternatively, an oil phase comprising 5-50 wt% of a compound I according to the invention, 0-40 wt% water insoluble organic solvent (e.g. aromatic hydrocarbon), and an isocyanate monomer (e.g. diphenylmethene-4,4'-diisocyanatae) are dispersed into an aqueous solution of a protective colloid (e.g. polyvinyl alcohol). The addition of a polyamine (e.g. hexamethylenediamine) results in the formation of polyurea microcapsules. The monomers amount to 1 -10 wt%. The wt% relate to the total CS
composition.
xi)Dustable powders (DP, DS)
1 -10 wt% of a compound I are ground finely and mixed intimately with up to 100 wt% solid carrier, e.g. finely divided kaolin.
xii) Granules (GR, FG)
0.5-30 wt% of a compound I is ground finely and associated with up to 100 wt% solid carrier (e.g. silicate). Granulation is achieved by extrusion, spray-drying or the fluidized bed.
xiii) Ultra-low volume liquids (UL)
1 -50 wt% of a compound I are dissolved in up to 100 wt% organic solvent, e.g. aromatic hydrocarbon.
The compositions types i) to xiii) may optionally comprise further auxiliaries, such as 0,1 -1 wt% bactericides, 5-15 wt% anti-freezing agents, 0,1 -1 wt% anti-foaming agents, and 0,1 -1 wt% colorants.
The agrochemical compositions generally comprise between 0.01 and 95%, preferably between 0.1 and 90%, and most preferably between 0.5 and 75%, by weight of active substance. The active substances are employed in a purity of from 90% to 100%, preferably from 95% to 100% (according to NMR spectrum).
Water-soluble concentrates (LS), Suspoemulsions (SE), flowable concentrates (FS), powders for dry treatment (DS), water-dispersible powders for slurry treatment (WS), water- soluble powders (SS), emulsions (ES), emulsifiable concentrates (EC) and gels (GF) are usually employed for the purposes of treatment of plant propagation materials, particularly seeds. The compositions in question give, after two-to-tenfold dilution, active substance concentrations of from 0.01 to 60% by weight, preferably from 0.1 to 40%, in the ready-to-use preparations.
Application can be carried out before or during sowing. Methods for applying or treating compound I and compositions thereof, respectively, on to plant propagation material, especially
seeds include dressing, coating, pelleting, dusting, soaking and in-furrow application methods of the propagation material. Preferably, compound I or the compositions thereof, respectively, are applied on to the plant propagation material by a method such that germination is not induced, e. g. by seed dressing, pelleting, coating and dusting.
When employed in plant protection, the amounts of active substances applied are, depending on the kind of effect desired, from 0.001 to 2 kg per ha, preferably from 0.005 to 2 kg per ha, more preferably from 0.05 to 0.9 kg per ha, in particular from 0.1 to 0.75 kg per ha.
In treatment of plant propagation materials such as seeds, e. g. by dusting, coating or drenching seed, amounts of active substance of from 0.1 to 1000 g, preferably from 1 to 1000 g, more preferably from 1 to 100 g and most preferably from 5 to 100 g, per 100 kilogram of plant propagation material (preferably seed) are generally required.
When used in the protection of materials or stored products, the amount of active substance applied depends on the kind of application area and on the desired effect. Amounts customarily applied in the protection of materials are 0.001 g to 2 kg, preferably 0.005 g to 1 kg, of active substance per cubic meter of treated material.
Various types of oils, wetters, adjuvants, fertilizer, or micronutrients, and other pesticides (e.g. herbicides, insecticides, fungicides, growth regulators, safeners) may be added to the active substances or the compositions comprising them as premix or, if appropriate not until immediately prior to use (tank mix). These agents can be admixed with the compositions according to the invention in a weight ratio of 1 :100 to 100:1 , preferably 1 :10 to 10:1.
The user applies the composition according to the invention usually from a predosage device, a knapsack sprayer, a spray tank, a spray plane, or an irrigation system. Usually, the agrochemical composition is made up with water, buffer, and/or further auxiliaries to the desired application concentration and the ready-to-use spray liquor or the agrochemical composition according to the invention is thus obtained. Usually, 20 to 2000 liters, preferably 50 to 400 liters, of the ready-to-use spray liquor are applied per hectare of agricultural useful area.
According to one embodiment, individual components of the composition according to the invention such as parts of a kit or parts of a binary or ternary mixture may be mixed by the user himself in a spray tank and further auxiliaries may be added, if appropriate.
Mixing the compounds I or the compositions comprising them in the use form as fungicides with other fungicides results in many cases in an expansion of the fungicidal spectrum of activity being obtained or in a prevention of fungicide resistance development. Furthermore, in many cases, synergistic effects are obtained.
The following list of active substances, in conjunction with which the compounds I can be used, is intended to illustrate the possible combinations but does not limit them:
A) Respiration inhibitors
- Inhibitors of complex III at Qo site (e.g. strobilurins): azoxystrobin, coumethoxy- strobin, coumoxystrobin, dimoxystrobin, enestroburin, fenaminstrobin, fenoxy- strobin/flufenoxystrobin, fluoxastrobin, kresoxim-methyl, metominostrobin, orysastrobin, picoxystrobin, pyraclostrobin, pyrametostrobin, pyraoxystrobin, trifloxystrobin, 2-[2-(2,5- dimethyl-phenoxymethyl)-phenyl]-3-methoxy-acrylic acid methyl ester and 2-(2-(3-(2,6-di- chlorophenyl)-1 -methyl-allylideneaminooxymethyl)-phenyl)-2-methoxyimino-N-methyl-
acetamide, pyribencarb, triclopyricarb/chlorodincarb, famoxadone, fenamidone;
- inhibitors of complex III at Qi site: cyazofamid, amisulbrom, [(3S,6S,7R,8R)-8-benz- yl-3-[(3-acetoxy-4-methoxy-pyridine-2-carbonyl)amino]-6-methyl-4,9-dioxo-1 ,5-dioxonan 7-yl] 2-methylpropanoate, [(3S,6S,7R,8R)-8-benzyl-3-[[3-(acetoxymethoxy)-4-methoxy- pyridine-2-carbonyl]amino]-6-methyl-4,9-dioxo-1 ,5-dioxonan-7-yl] 2-methylpropanoate,
[(3S,6S,7R,8R)-8-benzyl-3-[(3-isobutoxycarbonyloxy-4-methoxy-pyridine- 2-carbonyl)amino]-6-methyl-4,9-dioxo-1 ,5-dioxonan-7-yl] 2-methylpropanoate,
[(3S,6S,7R,8R)-8-benzyl-3-[[3-(1 ,3-benzodioxol-5-ylmethoxy)-4-methoxy-pyridine-2-car- bonyl]amino]-6-methyl-4,9-dioxo-1 ,5-dioxonan-7-yl] 2-methylpropanoate;
- inhibitors of complex II (e. g. carboxamides): benodanil, bixafen, boscalid, carboxin, fenfuram, fluopyram, flutolanil, fluxapyroxad, furametpyr, isopyrazam, mepronil, oxycarboxin, penflufen, penthiopyrad, sedaxane, tecloftalam, thifluzamide, N-(4'- trifluoromethylthiobiphenyl-2-yl)-3-difluoromethyl-1-methyl-1 H-pyrazole-4-carboxamide, N- (2-(1 ,3,3-trimethyl-butyl)-phenyl)-1 ,3-dimethyl-5-fluoro-1 H-pyrazole-4-carboxamide, N-[9- (dichloromethylene)-l ,2,3,4-tetrahydro-1 ,4-methanonaphthalen-5-yl]-3-(difluoromethyl)-1 - methyl-1 H-pyrazole-4-carboxamide;
- other respiration inhibitors (e.g. complex I, uncouplers): diflumetorim, (5,8-difluoro- quinazolin-4-yl)-{2-[2-fluoro-4-(4-trifluoromethylpyridin-2-yloxy)-phenyl]-ethyl}-amine; nitrophenyl derivates: binapacryl, dinobuton, dinocap, fluazinam; ferimzone; organometal compounds: fentin salts, such as fentin-acetate, fentin chloride or fentin hydroxide;
ametoctradin; and silthiofam;
B) Sterol biosynthesis inhibitors (SBI fungicides)
- C14 demethylase inhibitors (DMI fungicides): triazoles: azaconazole, bitertanol, bromuconazole, cyproconazole, difenoconazole, diniconazole, diniconazole-M, epoxiconazole, fenbuconazole, fluquinconazole, flusilazole, flutriafol, hexaconazole, imibenconazole, ipconazole, metconazole, myclobutanil, oxpoconazole, paclobutrazole, penconazole, propiconazole, prothioconazole, simeconazole, tebuconazole,
tetraconazole, triadimefon, triadimenol, triticonazole, uniconazole; imidazoles: imazalil, pefurazoate, prochloraz, triflumizol; pyrimidines, pyridines and piperazines: fenarimol, nuarimol, pyrifenox, triforine;
- Delta14-reductase inhibitors: aldimorph, dodemorph, dodemorph-acetate, fenpropimorph, tridemorph, fenpropidin, piperalin, spiroxamine;
- Inhibitors of 3-keto reductase: fenhexamid;
C) Nucleic acid synthesis inhibitors
- phenylamides or acyl amino acid fungicides: benalaxyl, benalaxyl-M, kiralaxyl, metalaxyl, metalaxyl-M (mefenoxam), ofurace, oxadixyl;
- others: hymexazole, octhilinone, oxolinic acid, bupirimate, 5-fluorocytosine, 5-fluoro- 2-(p-tolylmethoxy)pyrimidin-4-amine, 5-fluoro-2-(4-fluorophenylmethoxy)pyrimidin- 4-amine;
D)lnhibitors of cell division and cytoskeleton
- tubulin inhibitors, such as benzimidazoles, thiophanates: benomyl, carbendazim, fuberidazole, thiabendazole, thiophanate-methyl; triazolopyrimidines: 5-chloro- 7-(4-methylpiperidin-1 -yl)-6-(2,4,6-trifluorophenyl)-[1 ,2,4]triazolo[1 ,5-a]pyrimidine
- other cell division inhibitors: diethofencarb, ethaboxam, pencycuron, fluopicolide,
zoxamide, metrafenone, pyriofenone;
E) Inhibitors of amino acid and protein synthesis
- methionine synthesis inhibitors (anilino-pyrimidines): cyprodinil, mepanipyrim, pyrimethanil;
- protein synthesis inhibitors: blasticidin-S, kasugamycin, kasugamycin hydrochloride- hydrate, mildiomycin, streptomycin, oxytetracyclin, polyoxine, validamycin A;
F) Signal transduction inhibitors
- MAP / histidine kinase inhibitors: fluoroimid, iprodione, procymidone, vinclozolin, fenpiclonil, fludioxonil;
- G protein inhibitors: quinoxyfen;
G) Lipid and membrane synthesis inhibitors
- Phospholipid biosynthesis inhibitors: edifenphos, iprobenfos, pyrazophos, isoprothiolane;
- lipid peroxidation: dicloran, quintozene, tecnazene, tolclofos-methyl, biphenyl, chloroneb, etridiazole;
- phospholipid biosynthesis and cell wall deposition: dimethomorph, flumorph, mandipropamid, pyrimorph, benthiavalicarb, iprovalicarb, valifenalate and N-(1 -(1 -(4- cyano-phenyl)ethanesulfonyl)-but-2-yl) carbamic acid-(4-fluorophenyl) ester;
- compounds affecting cell membrane permeability and fatty acides: propamocarb, propamocarb-hydrochlorid
H) lnhibitors with Multi Site Action
- inorganic active substances: Bordeaux mixture, copper acetate, copper hydroxide, copper oxychloride, basic copper sulfate, sulfur;
- thio- and dithiocarbamates: ferbam, mancozeb, maneb, metam, metiram, propineb, thiram, zineb, ziram;
- organochlorine compounds (e.g. phthalimides, sulfamides, chloronitriles): anilazine, chlorothalonil, captafol, captan, folpet, dichlofluanid, dichlorophen, flusulfamide, hexachlorobenzene, pentachlorphenole and its salts, phthalide, tolylfluanid, N-(4-chloro-2- nitro-phenyl)-N-ethyl-4-methyl-benzenesulfonamide;
- guanidines and others: guanidine, dodine, dodine free base, guazatine, guazatine- acetate, iminoctadine, iminoctadine-triacetate, iminoctadine-tris(albesilate), dithianon;
I) Cell wall synthesis inhibitors
- inhibitors of glucan synthesis: validamycin, polyoxin B; melanin synthesis inhibitors: pyroquilon, tricyclazole, carpropamid, dicyclomet, fenoxanil;
J) Plant defence inducers
- acibenzolar-S-methyl, probenazole, isotianil, tiadinil, prohexadione-calcium;
phosphonates: fosetyl, fosetyl-aluminum, phosphorous acid and its salts;
K) Unknown mode of action
- bronopol, chinomethionat, cyflufenamid, cymoxanil, dazomet, debacarb, diclo- mezine, difenzoquat, difenzoquat-methylsulfate, diphenylamin, fenpyrazamine, flumetover, flusulfamide, flutianil, methasulfocarb, nitrapyrin, nitrothal-isopropyl, oxin- copper, proquinazid, tebufloquin, tecloftalam, triazoxide, 2-butoxy-6-iodo- 3-propylchromen-4-one, N-(cyclopropylmethoxyimino-(6-difluoro-methoxy-2,3-difluoro- phenyl)-methyl)-2-phenyl acetamide, N'-(4-(4-chloro-3-trifluoromethyl-phenoxy)-2,5-
dimethyl-phenyl)-N-ethyl-N-methyl formamidine, N'-(4-(4-fluoro-3-trifluoromethyl- phenoxy)-2,5-dimethyl-phenyl)-N-ethyl-N-methyl formamidine, N'-(2-methyl-5- trifluoromethyl-4-(3-trimethylsilanyl-propoxy)-phenyl)-N-ethyl-N-methyl formamidine, N'-(5- difluoromethyl-2-methyl-4-(3-trimethylsilanyl-propoxy)-phenyl)-N-ethyl-N-methyl formamidine, 2-{1 -[2-(5-methyl-3-trifluoromethyl-pyrazole-1 -yl)-acetyl]-piperidin-4-yl}- thiazole-4-carboxylic acid methyl-(1 ,2,3,4-tetrahydro-naphthalen-1 -yl)-amide, 2-{1 -[2-(5- methyl-3-trifluoromethyl-pyrazole-1 -yl)-acetyl]-piperidin-4-yl}-thiazole-4-carboxylic acid methyl-(R)-1 ,2,3,4-tetrahydro-naphthalen-1 -yl-amide,
1 - [4-[4-[5-(2,6-difluorophenyl)-4,5-dihydro-3-isoxazolyl]-2-thiazolyl]-1 -piperidinyl]- 2-[5-methyl-3-(trifluoromethyl)-1 H-pyrazol-1 -yl]ethanone, methoxy-acetic acid
6-tert-butyl-8-fluoro-2,3-dimethyl-quinolin-4-yl ester, N-Methyl-2-{1 -[(5-methyl-3-trifluoro- methyl-1 H-pyrazol-1 -yl)-acetyl]-piperidin-4-yl}-N-[(1 R)-1 ,2,3,4-tetrahydronaphthalen-1 -yl]- 4-thiazolecarboxamide, 3-[5-(4-methylphenyl)-2,3-dimethyl-isoxazolidin-3-yl]-pyridine, 3- [5-(4-chloro-phenyl)-2,3-dimethyl-isoxazolidin-3-yl]-pyridine (pyrisoxazole), N-(6-methoxy- pyridin-3-yl) cyclopropanecarboxylic acid amide, 5-chloro-1 -(4,6-dimethoxy-pyrimidin-2- yl)-2-methyl-1 H-benzoimidazole,
2- (4-chloro-phenyl)-N-[4-(3,4-dimethoxy-phenyl)-isoxazol-5-yl]-2-prop-2-ynyloxy- acetamide;
L) Antifungal biocontrol agents, plant bioactivators: Ampelomyces quisqualis (e.g. AQ 10® from Intrachem Bio GmbH & Co. KG, Germany), Aspergillus flavus (e.g.
AFLAGUARD® from Syngenta, CH), Aureobasidium pullulans (e.g. BOTECTOR® from bio-ferm GmbH, Germany), Bacillus pumilus (e.g. NRRL Accession No. B-30087 in SONATA® and BALLAD® Plus from AgraQuest Inc., USA), Bacillus subtilis (e.g. isolate NRRL-Nr. B-21661 in RHAPSODY®, SERENADE® MAX and SERENADE® ASO from AgraQuest Inc., USA), Bacillus subtilis var. amyloliquefaciens FZB24 (e.g. TAEGRO® from Novozyme Biologicals, Inc., USA), Candida oleophila I-82 (e.g. ASPIRE® from Ecogen Inc., USA), Candida saitoana (e.g. BIOCURE® (in mixture with lysozyme) and BIOCOAT® from Micro Flo Company, USA (BASF SE) and Arysta), Chitosan (e.g.
ARMOUR-ZEN from BotriZen Ltd., NZ), Clonostachys rosea f. catenulata, also named Gliocladium catenulatum (e.g. isolate J1446: PRESTOP® from Verdera, Finland),
Coniothyrium minitans (e.g. CONTANS® from Prophyta, Germany), Cryphonectria parasitica (e.g. Endothia parasitica from CNICM, France), Cryptococcus albidus (e.g. YIELD PLUS® from Anchor Bio-Technologies, South Africa), Fusarium oxysporum (e.g. BIOFOX® from S.I.A.P.A., Italy, FUSACLEAN® from Natural Plant Protection, France), Metschnikowia fructicola (e.g. SHEMER® from Agrogreen, Israel), Microdochium dimerum
(e.g. ANTIBOT® from Agrauxine, France), Phlebiopsis gigantea (e.g. ROTSOP® from Verdera, Finland), Pseudozyma flocculosa (e.g. SPORODEX® from Plant Products Co. Ltd., Canada), Pythium oligandrum DV74 (e.g. POLYVERSUM® from Remeslo SSRO, Biopreparaty, Czech Rep.), Reynoutria sachlinensis (e.g. REGALIA® from Marrone Biolnnovations, USA), Talaromyces flavus V1 17b (e.g. PROTUS® from Prophyta,
Germany), Trichoderma asperellum SKT-1 (e.g. ECO-HOPE® from Kumiai Chemical Industry Co., Ltd., Japan), T. atroviride LC52 (e.g. SENTINEL® from Agrimm
Technologies Ltd, NZ), T. harzianum T-22 (e.g. PLANTSHIELD® der Firma BioWorks Inc., USA), T. harzianum TH 35 (e.g. ROOT PRO® from Mycontrol Ltd., Israel), T.
harzianum T-39 (e.g. TRICHODEX® and TRICHODERMA 2000® from Mycontrol Ltd., Israel and Makhteshim Ltd., Israel), T. harzianum and T. viride (e.g. TRICHOPEL from Agrimm Technologies Ltd, NZ), T. harzianum ICC012 and T. viride ICC080 (e.g.
REMEDIER® WP from Isagro Ricerca, Italy), T. polysporum and T. harzianum (e.g.
BINAB® from BINAB Bio-Innovation AB, Sweden), T. stromaticum (e.g. TRICOVAB® from
C.E.P.L.A.C., Brazil), T. virens GL-21 (e.g. SOILGARD® from Certis LLC, USA), T. viride (e.g. TRIECO® from Ecosense Labs. (India) Pvt. Ltd., Indien, BIO-CURE® F from T. Stanes & Co. Ltd., Indien), T. viride TV1 (e.g. T. viride TV1 from Agribiotec srl, Italy), Ulocladium oudemansii HRU3 (e.g. BOTRY-ZEN® from Botry-Zen Ltd, NZ);
M) Growth regulators
abscisic acid, amidochlor, ancymidol, 6-benzylaminopurine, brassinolide, butralin, chlormequat (chlormequat chloride), choline chloride, cyclanilide, daminozide, dikegulac, dimethipin, 2,6-dimethylpuridine, ethephon, flumetralin, flurprimidol, fluthiacet,
forchlorfenuron, gibberellic acid, inabenfide, indole-3-acetic acid , maleic hydrazide, mefluidide, mepiquat (mepiquat chloride), naphthaleneacetic acid, N-6-benzyladenine, paclobutrazol, prohexadione (prohexadione-calcium), prohydrojasmon, thidiazuron, triapenthenol, tributyl phosphorotrithioate, 2,3,5-tri-iodobenzoic acid , trinexapac-ethyl and uniconazole;
N) Herbicides
- acetamides: acetochlor, alachlor, butachlor, dimethachlor, dimethenamid, flufenacet, mefenacet, metolachlor, metazachlor, napropamide, naproanilide, pethoxamid, pretilachlor, propachlor, thenylchlor;
- amino acid derivatives: bilanafos, glyphosate, glufosinate, sulfosate;
- aryloxyphenoxypropionat.es: clodinafop, cyhalofop-butyl, fenoxaprop, fluazifop, haloxyfop, metamifop, propaquizafop, quizalofop, quizalofop-P-tefuryl;
- Bipyridyls: diquat, paraquat;
- (thio)carbamates: asulam, butylate, carbetamide, desmedipham, dimepiperate, eptam (EPTC), esprocarb, molinate, orbencarb, phenmedipham, prosulfocarb,
pyributicarb, thiobencarb, triallate;
- cyclohexanediones: butroxydim, clethodim, cycloxydim, profoxydim, sethoxydim, tepraloxydim, tralkoxydim;
- dinitroanilines: benfluralin, ethalfluralin, oryzalin, pendimethalin, prodiamine, trifluralin;
- diphenyl ethers: acifluorfen, aclonifen, bifenox, diclofop, ethoxyfen, fomesafen, lactofen, oxyfluorfen;
- hydroxybenzonitriles: bomoxynil, dichlobenil, ioxynil;
- imidazolinones: imazamethabenz, imazamox, imazapic, imazapyr, imazaquin, imazethapyr;
- phenoxy acetic acids: clomeprop, 2,4-dichlorophenoxyacetic acid (2,4-D), 2,4-DB, dichlorprop, MCPA, MCPA-thioethyl, MCPB, Mecoprop;
- pyrazines: chloridazon, flufenpyr-ethyl, fluthiacet, norflurazon, pyridate;
- pyridines: aminopyralid, clopyralid, diflufenican, dithiopyr, fluridone, fluroxypyr, picloram, picolinafen, thiazopyr;
- sulfonyl ureas: amidosulfuron, azimsulfuron, bensulfuron, chlorimuron-ethyl,
chlorsulfuron, cinosulfuron, cyclosulfamuron, ethoxysulfuron, flazasulfuron,
flucetosulfuron, flupyrsulfuron, foramsulfuron, halosulfuron, imazosulfuron, iodosulfuron, mesosulfuron, metazosulfuron, metsulfuron-methyl, nicosulfuron, oxasulfuron,
primisulfuron, prosulfuron, pyrazosulfuron, rimsulfuron, sulfometuron, sulfosulfuron, thifensulfuron, triasulfuron, tribenuron, trifloxysulfuron, triflusulfuron, tritosulfuron, 1 -((2- chloro-6-propyl-imidazo[1 ,2-b]pyridazin-3-yl)sulfonyl)-3-(4,6-dimethoxy-pyrimidin-2 yl)urea;
- triazines: ametryn, atrazine, cyanazine, dimethametryn, ethiozin, hexazinone, metamitron, metribuzin, prometryn, simazine, terbuthylazine, terbutryn, triaziflam;
- ureas: chlorotoluron, daimuron, diuron, fluometuron, isoproturon, linuron, metha- benzthiazuron,tebuthiuron;
- other acetolactate synthase inhibitors: bispyribac-sodium, cloransulam-methyl, diclosulam, florasulam, flucarbazone, flumetsulam, metosulam, ortho-sulfamuron, penoxsulam, propoxycarbazone, pyribambenz-propyl, pyribenzoxim, pyriftalid, pyriminobac-methyl, pyrimisulfan, pyrithiobac, pyroxasulfone, pyroxsulam;
- others: amicarbazone, aminotriazole, anilofos, beflubutamid, benazolin,
bencarbazone,benfluresate, benzofenap, bentazone, benzobicyclon, bicyclopyrone, bromacil, bromobutide, butafenacil, butamifos, cafenstrole, carfentrazone, cinidon-ethyl, chlorthal, cinmethylin, clomazone, cumyluron, cyprosulfamide, dicamba, difenzoquat, diflufenzopyr, Drechslera monoceras, endothal, ethofumesate, etobenzanid,
fenoxasulfone, fentrazamide, flumiclorac-pentyl, flumioxazin, flupoxam, flurochloridone, flurtamone, indanofan, isoxaben, isoxaflutole, lenacil, propanil, propyzamide, quinclorac, quinmerac, mesotrione, methyl arsonic acid, naptalam, oxadiargyl, oxadiazon, oxaziclomefone, pentoxazone, pinoxaden, pyraclonil, pyraflufen-ethyl, pyrasulfotole, pyrazoxyfen, pyrazolynate, quinoclamine, saflufenacil, sulcotrione, sulfentrazone, terbacil, tefuryltrione, tembotrione, thiencarbazone, topramezone, (3-[2-chloro-4-fluoro-5-(3- methyl-2,6-dioxo-4-trifluoromethyl-3,6-dihydro-2H-pyrimidin-1 -yl)-phenoxy]-pyridin-2- yloxy)-acetic acid ethyl ester, 6-amino-5-chloro-2-cyclopropyl-pyrimidine-4-carboxylic acid methyl ester, 6-chloro-3-(2-cyclopropyl-6-methyl-phenoxy)-pyridazin-4-ol, 4-amino-3- chloro-6-(4-chloro-phenyl)-5-fluoro-pyridine-2-carboxylic acid, 4-amino-3-chloro-6-(4- chloro-2-fluoro-3-methoxy-phenyl)-pyridine-2-carboxylic acid methyl ester, and 4-amino-3- chloro-6-(4-chloro-3-dimethylamino-2-fluoro-phenyl)-pyridine-2-carboxylic acid methyl ester.
O) Insecticides
- organo(thio)phosphates: acephate, azamethiphos, azinphos-methyl, chlorpyrifos, chlorpyrifos-methyl, chlorfenvinphos, diazinon, dichlorvos, dicrotophos, dimethoate, disulfoton, ethion, fenitrothion, fenthion, isoxathion, malathion, methamidophos, methidathion, methyl-parathion, mevinphos, monocrotophos, oxydemeton-methyl, paraoxon, parathion, phenthoate, phosalone, phosmet, phosphamidon, phorate, phoxim, pirimiphos-methyl, profenofos, prothiofos, sulprophos, tetrachlorvinphos, terbufos, triazophos, trichlorfon;
- carbamates: alanycarb, aldicarb, bendiocarb, benfuracarb, carbaryl, carbofuran, carbosulfan, fenoxycarb, furathiocarb, methiocarb, methomyl, oxamyl, pirimicarb, propoxur, thiodicarb, triazamate;
- pyrethroids: allethrin, bifenthrin, cyfluthrin, cyhalothrin, cyphenothrin, cypermethrin, alpha-cypermethrin, beta-cypermethrin, zeta-cypermethrin, deltamethrin, esfenvalerate, etofenprox, fenpropathrin, fenvalerate, imiprothrin, lambda-cyhalothrin, permethrin, prallethrin, pyrethrin I and II, resmethrin, silafluofen, tau-fluvalinate, tefluthrin, tetramethrin, tralomethrin, transfluthrin, profluthrin, dimefluthrin;
- insect growth regulators: a) chitin synthesis inhibitors: benzoylureas: chlorfluazuron, cyramazin, diflubenzuron, flucycloxuron, flufenoxuron, hexaflumuron, lufenuron, novaluron, teflubenzuron, triflumuron; buprofezin, diofenolan, hexythiazox, etoxazole, clofentazine; b) ecdysone antagonists: halofenozide, methoxyfenozide, tebufenozide, azadirachtin; c) juvenoids: pyriproxyfen, methoprene, fenoxycarb; d) lipid biosynthesis inhibitors: spirodiclofen, spiromesifen, spirotetramat;
- nicotinic receptor agonists/antagonists compounds: clothianidin, dinotefuran, imidacloprid, thiamethoxam, nitenpyram, acetamiprid, thiacloprid, 1 -(2-chloro-thiazol-5- ylmethyl)-2-nitrimino-3,5-dimethyl-[1 ,3,5]triazinane;
- GABA antagonist compounds: endosulfan, ethiprole, fipronil, vaniliprole,
pyrafluprole, pyriprole, 5-amino-1 -(2,6-dichloro-4-methyl-phenyl)-4-sulfinamoyl- 1 H-pyrazole-3-carbothioic acid amide;
- macrocyclic lactone insecticides: abamectin, emamectin, milbemectin, lepimectin, spinosad, spinetoram;
- mitochondrial electron transport inhibitor (METI) I acaricides: fenazaquin, pyridaben, tebufenpyrad, tolfenpyrad, flufenerim;
- METI II and III compounds: acequinocyl, fluacyprim, hydramethylnon;
- Uncouplers: chlorfenapyr;
- oxidative phosphorylation inhibitors: cyhexatin, diafenthiuron, fenbutatin oxide, propargite;
- moulting disruptor compounds: cryomazine;
- mixed function oxidase inhibitors: piperonyl butoxide;
- sodium channel blockers: indoxacarb, metaflumizone;
- others: benclothiaz, bifenazate, cartap, flonicamid, pyridalyl, pymetrozine, sulfur, thiocyclam, flubendiamide, chlorantraniliprole, cyazypyr (HGW86), cyenopyrafen, flupyrazofos, cyflumetofen, amidoflumet, imicyafos, bistrifluron, pyrifluquinazon and 1 ,1 '- [(3S,4R,4aR,6S,6aS, 12R, 12aS, 12bS)-4-[[(2-cyclopropylacetyl)oxy]methyl]- 1 ,3,4,4a,5,6,6a,12,12a,12b-decahydro-12-hydroxy-4,6a,12b-trimethyl-1 1 -oxo-9-(3- pyridinyl)-2H,1 1 H-naphtho[2,1 -b]pyrano[3,4-e]pyran-3,6-diyl] cyclopropaneacetic acid ester.
The present invention furthermore relates to agrochemical compositions comprising a mixture of at least one compound I (component 1 ) and at least one further active substance useful for plant protection, e. g. selected from the groups A) to O) (component 2), in particular one further fungicide, e. g. one or more fungicide from the groups A) to L), as described above, and if desired one suitable solvent or solid carrier. Those mixtures are of particular interest, since many of them at the same application rate show higher efficiencies against harmful fungi. Furthermore, combating harmful fungi with a mixture of compounds I and at least one fungicide from groups A) to L), as described above, is more efficient than combating those fungi with
individual compounds I or individual fungicides from groups A) to L). By applying compounds I together with at least one active substance from groups A) to O) a synergistic effect can be obtained, i.e. more then simple addition of the individual effects is obtained (synergistic mixtures).
This can be obtained by applying the compounds I and at least one further active substance simultaneously, either jointly (e. g. as tank-mix) or separately, or in succession, wherein the time interval between the individual applications is selected to ensure that the active substance applied first still occurs at the site of action in a sufficient amount at the time of application of the further active substance(s). The order of application is not essential for working of the present invention.
In binary mixtures, i.e. compositions according to the invention comprising one compound I (component 1 ) and one further active substance (component 2), e. g. one active substance from groups A) to O), the weight ratio of component 1 and component 2 generally depends from the properties of the active substances used, usually it is in the range of from 1 :100 to 100:1 , regularly in the range of from 1 :50 to 50:1 , preferably in the range of from 1 :20 to 20:1 , more preferably in the range of from 1 :10 to 10:1 and in particular in the range of from 1 :3 to 3:1 .
In ternary mixtures, i.e. compositions according to the invention comprising one compound I (component 1 ) and a first further active substance (component 2) and a second further active substance (component 3), e. g. two active substances from groups A) to O), the weight ratio of component 1 and component 2 depends from the properties of the active substances used, preferably it is in the range of from 1 :50 to 50:1 and particularly in the range of from 1 :10 to 10:1 , and the weight ratio of component 1 and component 3 preferably is in the range of from 1 :50 to 50:1 and particularly in the range of from 1 :10 to 10:1.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group A) (component 2) and particularly selected from azoxystrobin, dimoxystrobin, fluoxastrobin, kresoxim-methyl, orysastrobin, picoxystrobin, pyraclostrobin, trifloxystrobin; famoxadone, fenamidone; bixafen, boscalid, fluopyram, fluxapyroxad, isopyrazam, penflufen, penthiopyrad, sedaxane; ametoctradin, cyazofamid, fluazinam, fentin salts, such as fentin acetate.
Preference is given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group B) (component 2) and particularly selected from cyproconazole, difenoconazole, epoxiconazole, fluquinconazole, flusilazole, flutriafol, metconazole, myclobutanil, penconazole, propiconazole, prothioconazole, triadimefon, triadimenol, tebuconazole, tetraconazole, triticonazole, prochloraz, fenarimol, triforine;
dodemorph, fenpropimorph, tridemorph, fenpropidin, spiroxamine; fenhexamid.
Preference is given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group C) (component 2) and particularly selected from metalaxyl, (metalaxyl-M) mefenoxam, ofurace.
Preference is given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group D) (component 2) and particularly selected from benomyl, carbendazim, thiophanate-methyl, ethaboxam, fluopicolide, zoxamide, metrafenone, pyriofenone.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group E) (component 2) and particularly selected from
cyprodinil, mepanipyrim, pyrimethanil.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group F) (component 2) and particularly selected from iprodione, fludioxonil, vinclozolin, quinoxyfen.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group G) (component 2) and particularly selected from dimethomorph, flumorph, iprovalicarb, benthiavalicarb, mandipropamid, propamocarb.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group H) (component 2) and particularly selected from copper acetate, copper hydroxide, copper oxychloride, copper sulfate, sulfur, mancozeb, metiram, propineb, thiram, captafol, folpet, chlorothalonil, dichlofluanid, dithianon.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group I) (component 2) and particularly selected from carpropamid and fenoxanil.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group J) (component 2) and particularly selected from acibenzolar-S-methyl, probenazole, tiadinil, fosetyl, fosetyl-aluminium, H3PO3 and salts thereof.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group K) (component 2) and particularly selected from cymoxanil, proquinazid and N-methyl-2-{1 -[(5-methyl-3-trifluoromethyl-1 H-pyrazol-1 -yl)-acetyl]- piperidin-4-yl}-N-[(1 R)-1 ,2,3,4-tetrahydronaphthalen-1 -yl]-4-thiazolecarboxamide.
Preference is also given to mixtures comprising a compound I (component 1 ) and at least one active substance selected from group L) (component 2) and particularly selected from Bacillus subtilis strain NRRL No. B-21661 , Bacillus pumilus strain NRRL No. B-30087 and Ulocladium oudemansii .
Accordingly, the present invention furthermore relates to compositions comprising one compound I (component 1 ) and one further active substance (component 2), which further active substance is selected from the column "Component 2" of the lines D-1 to D-370 of Table D.
A further embodiment relates to the compositions D-1 to D-370 listed in Table D, where a row of Table D corresponds in each case to a fungicidal composition comprising one of the in the present specification individualized compounds of formula I (component 1 ) and the respective further active substance from groups A) to O) (component 2) stated in the row in question. Preferably, the compositions described comprise the active substances in
synergistically effective amounts.
Table D: Composition comprising one indiviualized compound I and one further active substance from groups A) to O)
Mixture Component 1 Component 2
D-1 one individualized compound I Azoxystrobin
D-2 one individualized compound I Coumethoxystrobin
D-3 one individualized compound I Coumoxystrobin
D-4 one individualized compound I Dimoxystrobin
Mixture Component 1 Component 2
D-5 one individualized compound I Enestroburin
D-6 one individualized compound I Fenaminstrobin
D-7 one individualized compound I Fenoxystrobin/Flufenoxystrobin
D-8 one individualized compound I Fluoxastrobin
D-9 one individualized compound I Kresoxim-methyl
D-10 one individualized compound I Metominostrobin
D-1 1 one individualized compound I Orysastrobin
D-12 one individualized compound I Picoxystrobin
D-13 one individualized compound I Pyraclostrobin
D-14 one individualized compound I Pyrametostrobin
D-15 one individualized compound I Pyraoxystrobin
D-16 one individualized compound I Pyribencarb
D-17 one individualized compound I Trifloxystrobin
D-18 one individualized compound I Triclopyricarb/Chlorodincarb
2-[2-(2,5-dimethyl-phenoxymethyl)-
D-19 one individualized compound I phenyl]-3-methoxy-acrylic acid methyl ester
2-(2-(3-(2,6-dichlorophenyl)-1 -methyl-
D-20 one individualized compound I allylideneaminooxymethyl)-phenyl)-
2-methoxyimino-N-methyl-acetamide
D-21 one individualized compound I Benalaxyl
D-22 one individualized compound I Benalaxyl-M
D-23 one individualized compound I Benodanil
D-24 one individualized compound I Benzovindiflupyr
D-25 one individualized compound I Bixafen
D-26 one individualized compound I Boscalid
D-27 one individualized compound I Carboxin
D-28 one individualized compound I Fenfuram
D-29 one individualized compound I Fenhexamid
D-30 one individualized compound I Flutolanil
D-31 one individualized compound I Fluxapyroxad
D-32 one individualized compound I Furametpyr
D-33 one individualized compound I Isopyrazam
D-34 one individualized compound I Isotianil
D-35 one individualized compound I Kiralaxyl
D-36 one individualized compound I Mepronil
D-37 one individualized compound I Metalaxyl
D-38 one individualized compound I Metalaxyl-M
D-39 one individualized compound I Ofurace
D-40 one individualized compound I Oxadixyl
D-41 one individualized compound I Oxycarboxin
D-42 one individualized compound I Penflufen
Mixture Component 1 Component 2
D-43 one individualized compound I Penthiopyrad
D-44 one individualized compound I Sedaxane
D-45 one individualized compound I Tecloftalam
D-46 one individualized compound I Thifluzamide
D-47 one individualized compound I Tiadinil
2-Amino-4-methyl-thiazole-5-carboxylic
D-48 one individualized compound I
acid anilide
N-(4'-trifluoromethylthiobiphenyl-2-yl)-
D-49 one individualized compound I 3- difluoromethyl-1 -methyl-1 H-pyrazole-
4- carboxamide
N-(2-(1 ,3,3-trimethyl-butyl)-phenyl)-
D-50 one individualized compound I 1 ,3-dimethyl-5-fluoro-1 H-pyrazole- 4-carboxamide
3-(difluoromethyl)-1 -methyl-N-(1 ,1 ,3-tri-
D-51 one individualized compound I methylindan-4-yl)pyrazole-4-carbox- amide
3-(trifluoromethyl)-1 -methyl-N-(1 ,1 ,3-tri-
D-52 one individualized compound I methylindan-4-yl)pyrazole-4-carbox- amide
1 ,3-dimethyl-N-(1 ,1 ,3-trimethylindan-
D-53 one individualized compound I
4-yl)pyrazole-4-carboxamide
3- (trifluoromethyl)-1 ,5-dimethyl-
D-54 one individualized compound I N-(1 ,1 ,3-trimethylindan-4-yl)pyrazole-
4- carboxamide
3- (difluoromethyl)-1 ,5-dimethyl-
D-55 one individualized compound I N-(1 ,1 ,3-trimethylindan-4-yl)pyrazole-
4- carboxamide
1 ,3,5-trimethyl-N-(1 ,1 ,3-trimethylindan-
D-56 one individualized compound I
4-yl)pyrazole-4-carboxamide
D-57 one individualized compound I Dimethomorph
D-58 one individualized compound I Flumorph
D-59 one individualized compound I Pyrimorph
D-60 one individualized compound I Flumetover
D-61 one individualized compound I Fluopicolide
D-62 one individualized compound I Fluopyram
D-63 one individualized compound I Zoxamide
D-64 one individualized compound I Carpropamid
D-65 one individualized compound I Diclocymet
D-66 one individualized compound I Mandipropamid
D-67 one individualized compound I Oxytetracyclin
D-68 one individualized compound I Silthiofam
Mixture Component 1 Component 2
N-(6-methoxy-pyridin-3-yl)
D-69 one individualized compound I
cyclopropanecarboxylic acid amide
D-70 one individualized compound I Azaconazole
D-71 one individualized compound I Bitertanol
D-72 one individualized compound I Bromuconazole
D-73 one individualized compound I Cyproconazole
D-74 one individualized compound I Difenoconazole
D-75 one individualized compound I Diniconazole
D-76 one individualized compound I Diniconazole-M
D-77 one individualized compound I Epoxiconazole
D-78 one individualized compound I Fenbuconazole
D-79 one individualized compound I Fluquinconazole
D-80 one individualized compound I Flusilazole
D-81 one individualized compound I Flutriafol
D-82 one individualized compound I Hexaconazol
D-83 one individualized compound I Imibenconazole
D-84 one individualized compound I Ipconazole
D-85 one individualized compound I Metconazole
D-86 one individualized compound I Myclobutanil
D-87 one individualized compound I Oxpoconazol
D-88 one individualized compound I Paclobutrazol
D-89 one individualized compound I Penconazole
D-90 one individualized compound I Propiconazole
D-91 one individualized compound I Prothioconazole
D-92 one individualized compound I Simeconazole
D-93 one individualized compound I Tebuconazole
D-94 one individualized compound I Tetraconazole
D-95 one individualized compound I Triadimefon
D-96 one individualized compound I Triadimenol
D-97 one individualized compound I Triticonazole
D-98 one individualized compound I Uniconazole
1 - [re/-(2S;3R)-3-(2-chlorophenyl)-
D-99 one individualized compound I 2- (2,4-difluorophenyl)-oxiranylmethyl]- 5-thiocyanato-1 H-[1 ,2,4]triazole,
2-[re/-(2S;3R)-3-(2-chlorophenyl)-
D-100 one individualized compound I 2-(2,4-difluorophenyl)-oxiranylmethyl]-
2H-[1 ,2,4]triazole-3-thiol
D-101 one individualized compound I Cyazofamid
D-102 one individualized compound I Amisulbrom
D-103 one individualized compound I Imazalil
D-104 one individualized compound I Imazalil-sulfate
D-105 one individualized compound I Pefurazoate
Mixture Component 1 Component 2
D-106 one individualized compound I Prochloraz
D-107 one individualized compound I Triflumizole
D-108 one individualized compound I Benomyl
D-109 one individualized compound I Carbendazim
D-1 10 one individualized compound I Fuberidazole
D-1 1 1 one individualized compound I Thiabendazole
D-1 12 one individualized compound I Ethaboxam
D-1 13 one individualized compound I Etridiazole
D-1 14 one individualized compound I Hymexazole
2-(4-Chloro-phenyl)-N-[4-(3,4-dimeth-
D-1 15 one individualized compound I oxy-phenyl)-isoxazol-5-yl]-2-prop-2-yn- yloxy-acetamide
D-1 16 one individualized compound I Fluazinam
D-1 17 one individualized compound I Pyrifenox
3-[5-(4-Chloro-phenyl)-2,3-dimethyl-is-
D-1 18 one individualized compound I
oxazolidin-3-yl]-pyridine (Pyrisoxazole)
3-[5-(4-Methyl-phenyl)-2,3-dimethyl-
D-1 19 one individualized compound I
isoxazolidin-3-yl]-pyridine
D-120 one individualized compound I Bupirimate
D-121 one individualized compound I Cyprodinil
D-122 one individualized compound I 5-Fluorocytosine
5-Fluoro-2-(p-tolylmethoxy)pyrimidin-
D-123 one individualized compound I
4-amine
5-Fluoro-2-(4-fluorophenylmethoxy)-
D-124 one individualized compound I
pyrimidin-4-amine
D-125 one individualized compound I Diflumetorim
(5,8-Difluoroquinazolin-4-yl)-{2-[2-fluo-
D-126 one individualized compound I ro-4-(4-trifluoromethylpyridin-2-yloxy)- phenyl]-ethyl}-amine
D-127 one individualized compound I Fenarimol
D-128 one individualized compound I Ferimzone
D-129 one individualized compound I Mepanipyrim
D-130 one individualized compound I Nitra pyrin
D-131 one individualized compound I Nuarimol
D-132 one individualized compound I Pyrimethanil
D-133 one individualized compound I Triforine
D-134 one individualized compound I Fenpiclonil
D-135 one individualized compound I Fludioxonil
D-136 one individualized compound I Aldimorph
D-137 one individualized compound I Dodemorph
D-138 one individualized compound I Dodemorph-acetate
D-139 one individualized compound I Fenpropimorph
Mixture Component 1 Component 2
D-140 one individualized compound I Tridemorph
D-141 one individualized compound I Fenpropidin
D-142 one individualized compound I Fluoroimid
D-143 one individualized compound I Iprodione
D-144 one individualized compound I Procymidone
D-145 one individualized compound I Vinclozolin
D-146 one individualized compound I Famoxadone
D-147 one individualized compound I Fenamidone
D-148 one individualized compound I Flutianil
D-149 one individualized compound I Octhilinone
D-150 one individualized compound I Probenazole
D-151 one individualized compound I Fenpyrazamine
D-152 one individualized compound I Acibenzolar-S-methyl
D-153 one individualized compound I Ametoctradin
D-154 one individualized compound I Amisulbrom
[(3S,6S,7R,8R)-8-benzyl-3-[(3-isobuty- ryloxymethoxy-4-methoxypyridine-
D-155 one individualized compound I
2-carbonyl)amino]-6-methyl-4,9-dioxo- [1 ,5]dioxonan-7-yl] 2-methylpropanoate
[(3S,6S,7R,8R)-8-benzyl-3-[(3-acetoxy- 4-methoxy-pyridine-2-carbonyl)amino]-
D-156 one individualized compound I
6-methyl-4,9-dioxo-1 ,5-dioxonan-7-yl] 2-methylpropanoate
[(3S,6S,7R,8R)-8-benzyl-3-[[3-(acet- oxymethoxy)-4-methoxy-pyridine-
D-157 one individualized compound I
2-carbonyl]amino]-6-methyl-4,9-dioxo- 1 ,5-dioxonan-7-yl] 2-methylpropanoate
[(3S,6S,7R,8R)-8-benzyl-3-[(3-isobut- oxycarbonyloxy-4-methoxy-pyridine-
D-158 one individualized compound I
2-carbonyl)amino]-6-methyl-4,9-dioxo- 1 ,5-dioxonan-7-yl] 2-methylpropanoate
[(3S,6S,7R,8R)-8-benzyl-3-[[3-(1 ,3-ben- zodioxol-5-ylmethoxy)-4-methoxy-pyri-
D-159 one individualized compound I dine-2-carbonyl]amino]-6-methyl-4,9-di- oxo-1 ,5-dioxonan-7-yl] 2-methylpropanoate
(3S,6S,7R,8R)-3-[[(3-hydroxy-4-meth- oxy-2-pyridinyl)carbonyl]amino]-
D-160 one individualized compound I
6-methyl-4,9-dioxo-8-(phenylmethyl)- 1 ,5-dioxonan-7-yl 2-methylpropanoate
D-161 one individualized compound I Anilazin
D-162 one individualized compound I Blasticidin-S
Mixture Component 1 Component 2
D-163 one individualized compound I Captafol
D-164 one individualized compound I Captan
D-165 one individualized compound I Chinomethionat
D-166 one individualized compound I Dazomet
D-167 one individualized compound I Debacarb
D-168 one individualized compound I Diclomezine
D-169 one individualized compound I Difenzoquat,
D-170 one individualized compound I Difenzoquat-methylsulfate
D-171 one individualized compound I Fenoxanil
D-172 one individualized compound I Folpet
D-173 one individualized compound I Oxolinsaure
D-174 one individualized compound I Piperalin
D-175 one individualized compound I Proquinazid
D-176 one individualized compound I Pyroquilon
D-177 one individualized compound I Quinoxyfen
D-178 one individualized compound I Triazoxid
D-179 one individualized compound I Tricyclazole
2-Butoxy-6-iodo-3-propyl-chromen-4-
D-180 one individualized compound I
one
5-Chloro-1 -(4,6-dimethoxy-pyrimidin-2-
D-181 one individualized compound I
yl)-2-methyl-1 H-benzoimidazole
5- Chloro-7-(4-methyl-piperidin-1 -yl)-
D-182 one individualized compound I 6- (2,4,6-trifluoro-phenyl)-[1 ,2,4]tri- azolo[1 ,5-a]pyrimidine
D-183 one individualized compound I Ferbam
D-184 one individualized compound I Mancozeb
D-185 one individualized compound I Maneb
D-186 one individualized compound I Metam
D-187 one individualized compound I Methasulphocarb
D-188 one individualized compound I Metiram
D-189 one individualized compound I Propineb
D-190 one individualized compound I Thiram
D-191 one individualized compound I Zineb
D-192 one individualized compound I Ziram
D-193 one individualized compound I Diethofencarb
D-194 one individualized compound I Benthiavalicarb
D-195 one individualized compound I Iprovalicarb
D-196 one individualized compound I Propamocarb
D-197 one individualized compound I Propamocarb hydrochlorid
D-198 one individualized compound I Valifenalate
Mixture Component 1 Component 2
N-(1 -(1 -(4-cyanophenyl)ethanesulfon-
D-199 one individualized compound I yl)-but-2-yl) carbamic acid-(4-fluoro- phenyl) ester
D-200 one individualized compound I Dodine
D-201 one individualized compound I Dodine free base
D-202 one individualized compound I Guazatine
D-203 one individualized compound I Guazatine-acetate
D-204 one individualized compound I Iminoctadine
D-205 one individualized compound I Iminoctadine-triacetate
D-206 one individualized compound I Iminoctadine-tris(albesilate)
D-207 one individualized compound I Kasugamycin
D-208 one individualized compound I Kasugamycin-hydrochloride-hydrate
D-209 one individualized compound I Polyoxine
D-210 one individualized compound I Streptomycin
D-21 1 one individualized compound I Validamycin A
D-212 one individualized compound I Binapacryl
D-213 one individualized compound I Dicloran
D-214 one individualized compound I Dinobuton
D-215 one individualized compound I Dinocap
D-216 one individualized compound I Nitrothal-isopropyl
D-217 one individualized compound I Tecnazen
D-218 one individualized compound I Fentin salts
D-219 one individualized compound I Dithianon
2,6-dimethyl-1 H,5H-[1 ,4]dithiino
D-220 one individualized compound I [2,3-c:5,6-c']dipyrrole-
1 ,3,5,7(2H,6H)-tetraone
D-221 one individualized compound I Isoprothiolane
D-222 one individualized compound I Edifenphos
D-223 one individualized compound I Fosetyl, Fosetyl-aluminium
D-224 one individualized compound I Iprobenfos
Phosphorous acid (H3PO3) and
D-225 one individualized compound I
derivatives
D-226 one individualized compound I Pyrazophos
D-227 one individualized compound I Tolclofos-methyl
D-228 one individualized compound I Chlorothalonil
D-229 one individualized compound I Dichlofluanid
D-230 one individualized compound I Dichlorophen
D-231 one individualized compound I Flusulfamide
D-232 one individualized compound I Hexachlorbenzene
D-233 one individualized compound I Pencycuron
D-234 one individualized compound I Pentachlorophenol and salts
D-235 one individualized compound I Phthalide
Mixture Component 1 Component 2
D-236 one individualized compound I Quintozene
D-237 one individualized compound I Thiophanate Methyl
D-238 one individualized compound I Tolylfluanid
N-(4-chloro-2-nitro-phenyl)-N-ethyl-
D-239 one individualized compound I
4-methyl-benzenesulfonamide
D-240 one individualized compound I Bordeaux mixture
D-241 one individualized compound I Copper acetate
D-242 one individualized compound I Copper hydroxide
D-243 one individualized compound I Copper oxychloride
D-244 one individualized compound I basic Copper sulfate
D-245 one individualized compound I Sulfur
D-246 one individualized compound I Biphenyl
D-247 one individualized compound I Bronopol
D-248 one individualized compound I Cyflufenamid
D-249 one individualized compound I Cymoxanil
D-250 one individualized compound I Diphenylamin
D-251 one individualized compound I Metrafenone
D-252 one individualized compound I Pyriofenone
D-253 one individualized compound I Mildiomycin
D-254 one individualized compound I Oxin-copper
D-255 one individualized compound I Oxathiapiprolin
D-256 one individualized compound I Prohexadione calcium
D-257 one individualized compound I Spiroxamine
D-258 one individualized compound I Tebufloquin
D-259 one individualized compound I Tolylfluanid
N-(Cyclopropylmethoxyimino-(6-
D-260 one individualized compound I difluoromethoxy-2,3-difluoro-phenyl)- methyl)-2-phenyl acetamide
N'-(4-(4-chloro-3-trifluoromethyl-
D-261 one individualized compound I phenoxy)-2,5-dimethyl-phenyl)-N-ethyl- N-methyl formamidine
N'-(4-(4-fluoro-3-trifluoromethyl-
D-262 one individualized compound I phenoxy)-2,5-dimethyl-phenyl)-N-ethyl- N-methyl formamidine
N'-(2-methyl-5-trifluoromethyl-4-(3-tri-
D-263 one individualized compound I methylsilanyl-propoxy)-phenyl)-N-ethyl- N-methyl formamidine
N'-(5-difluoromethyl-2-methyl-4-(3-tri-
D-264 one individualized compound I methylsilanyl-propoxy)-phenyl)-N-ethyl- N-methyl formamidine
Methoxy-acetic acid 6-tert-butyl-8-
D-265 one individualized compound I
fluoro-2,3-dimethyl-quinolin-4-yl ester
Mixture Component 1 Component 2
D-266 one individualized compound I Bacillus subtilis NRRL No. B-21661
D-267 one individualized compound I Bacillus pumilus NRRL No. B-30087
D-268 one individualized compound I Ulocladium oudemansii
D-269 one individualized compound I Carbaryl
D-270 one individualized compound I Carbofuran
D-271 one individualized compound I Carbosulfan
D-272 one individualized compound I Methomylthiodicarb
D-273 one individualized compound I Bifenthrin
D-274 one individualized compound I Cyfluthrin
D-275 one individualized compound I Cypermethrin
D-276 one individualized compound I alpha-Cypermethrin
D-277 one individualized compound I zeta-Cypermethrin
D-278 one individualized compound I Deltamethrin
D-279 one individualized compound I Esfenvalerate
D-280 one individualized compound I Lambda-cyhalothrin
D-281 one individualized compound I Permethrin
D-282 one individualized compound I Tefluthrin
D-283 one individualized compound I Diflubenzuron
D-284 one individualized compound I Flufenoxuron
D-285 one individualized compound I Lufenuron
D-286 one individualized compound I Teflubenzuron
D-287 one individualized compound I Spirotetramate
D-288 one individualized compound I Clothianidin
D-289 one individualized compound I Dinotefuran
D-290 one individualized compound I Imidacloprid
D-291 one individualized compound I Thiamethoxam
D-292 one individualized compound I Flupyradifurone
D-293 one individualized compound I Acetamiprid
D-294 one individualized compound I Thiacloprid
D-295 one individualized compound I Endosulfan
D-296 one individualized compound I Fipronil
D-297 one individualized compound I Abamectin
D-298 one individualized compound I Emamectin
D-299 one individualized compound I Spinosad
D-300 one individualized compound I Spinetoram
D-301 one individualized compound I Hydramethylnon
D-302 one individualized compound I Chlorfenapyr
D-303 one individualized compound I Fenbutatin oxide
D-304 one individualized compound I Indoxacarb
D-305 one individualized compound I Metaflumizone
D-306 one individualized compound I Flonicamid
D-307 one individualized compound I Lubendiamide
Mixture Component 1 Component 2
D-308 one individualized compound I Chlorantraniliprole
D-309 one individualized compound I Cyazypyr (HGW86)
D-310 one individualized compound I Cyflumetofen
D-31 1 one individualized compound I Acetochlor
D-312 one individualized compound I Dimethenamid
D-313 one individualized compound I metolachlor
D-314 one individualized compound I Metazachlor
D-315 one individualized compound I Glyphosate
D-316 one individualized compound I Glufosinate
D-317 one individualized compound I Sulfosate
D-318 one individualized compound I Clodinafop
D-319 one individualized compound I Fenoxaprop
D-320 one individualized compound I Fluazifop
D-321 one individualized compound I Haloxyfop
D-322 one individualized compound I Paraquat
D-323 one individualized compound I Phenmedipham
D-324 one individualized compound I Clethodim
D-325 one individualized compound I Cycloxydim
D-326 one individualized compound I Profoxydim
D-327 one individualized compound I Sethoxydim
D-328 one individualized compound I Tepraloxydim
D-329 one individualized compound I Pendimethalin
D-330 one individualized compound I Prodiamine
D-331 one individualized compound I Trifluralin
D-332 one individualized compound I Acifluorfen
D-333 one individualized compound I Bromoxynil
D-334 one individualized compound I Imazamethabenz
D-335 one individualized compound I Imazamox
D-336 one individualized compound I Imazapic
D-337 one individualized compound I Imazapyr
D-338 one individualized compound I Imazaquin
D-339 one individualized compound I Imazethapyr
D-340 one individualized compound I 2,4-Dichlorophenoxyacetic acid (2,4-D)
D-341 one individualized compound I Chloridazon
D-342 one individualized compound I Clopyralid
D-343 one individualized compound I Fluroxypyr
D-344 one individualized compound I Picloram
D-345 one individualized compound I Picolinafen
D-346 one individualized compound I Bensulfuron
D-347 one individualized compound I Chlorimuron-ethyl
D-348 one individualized compound I Cyclosulfamuron
D-349 one individualized compound I lodosulfuron
Mixture Component 1 Component 2
D-350 one individua ized compound I Mesosulfuron
D-351 one individua ized compound I Metsulfuron-methyl
D-352 one individua ized compound I Nicosulfuron
D-353 one individua ized compound I Rimsulfuron
D-354 one individua ized compound I Triflusulfuron
D-355 one individua ized compound I Atrazine
D-356 one individua ized compound I Hexazinone
D-357 one individua ized compound I Diuron
D-358 one individua ized compound I Florasulam
D-359 one individua ized compound I Pyroxasulfone
D-360 one individua ized compound I Bentazone
D-361 one individua ized compound I Cinidon-ethyl
D-362 one individua ized compound I Cinmethylin
D-363 one individua ized compound I Dicamba
D-364 one individua ized compound I Diflufenzopyr
D-365 one individua ized compound I Quinclorac
D-366 one individua ized compound I Quinmerac
D-367 one individua ized compound I Mesotrione
D-368 one individua ized compound I Saflufenacil
D-369 one individua ized compound I Topramezone
1 ,1 '-[(3S,4R,4aR,6S,6aS,12R,12aS,
12bS)-4-[[(2-cyclopropylacetyl)oxy]me- thyl]-1 ,3,4,4a,5,6,6a,12,12a,12b-deca-
D-370 one individualized compound I hydro-12-hydroxy-4,6a,12b-trimethyl-
1 1 -oxo-9-(3-pyridinyl)-2H,1 1 H-naph- tho[2,1 -b]pyrano[3,4-e]pyran-3,6-diyl] cyclopropaneacetic acid ester
The active substances referred to as component 2, their preparation and their activity against harmful fungi is known (cf.: http://www.alanwood.net/pesticides/); these substances are commercially available. The compounds described by lUPAC nomenclature, their preparation and their fungicidal activity are also known (cf. Can. J. Plant Sci. 48(6), 587-94, 1968; EP-A 141 317; EP-A 152 031 ; EP-A 226 917; EP-A 243 970; EP-A 256 503; EP-A 428 941 ; EP-
A 532 022; EP-A 1 028 125; EP-A 1 035 122; EP-A 1 201 648; EP-A 1 122 244,
JP 2002316902; DE 19650197; DE 10021412; DE 102005009458; US 3,296,272;
US 3,325,503; WO 98/46608; WO 99/14187; WO 99/24413; WO 99/27783; WO 00/29404;
WO 00/46148; WO 00/65913; WO 01/54501 ; WO 01/56358; WO 02/22583; WO 02/40431 ; WO 03/10149; WO 03/1 1853; WO 03/14103; WO 03/16286; WO 03/53145; WO 03/61388;
WO 03/66609; WO 03/74491 ; WO 04/49804; WO 04/83193; WO 05/120234; WO 05/123689;
WO 05/123690; WO 05/63721 ; WO 05/87772; WO 05/87773; WO 06/15866; WO 06/87325;
WO 06/87343; WO 07/82098; WO 07/90624, WO 1 1/028657).
The mixtures of active substances can be prepared as compositions comprising besides the active ingridients at least one inert ingredient by usual means, e. g. by the means given for the compositions of compounds I.
Concerning usual ingredients of such compositions reference is made to the explanations given for the compositions containing compounds I.
The mixtures of active substances according to the present invention are suitable as fungicides, as are the compounds of formula I. They are distinguished by an outstanding effectiveness against a broad spectrum of phytopathogenic fungi, especially from the classes of the Ascomycetes, Basidiomycetes, Deuteromycetes and Peronosporomycetes (syn.
Oomycetes). In addition, it is refered to the explanations regarding the fungicidal activity of the compounds and the compositions containing compounds I, respectively. I. Synthesis examples
With appropriate modification of the starting materials, the procedures given in the synthesis examples below were used to obtain further compounds I. The compounds produced in this manner are listed in Table V below including corresponding physical data. 2-(2-Chloro-4- hydroxyphenyl)ethylamine-feri-butyl carbamate was prepared as described in WO 2004026305 A1 . 2-(2-Fluoro-4-methoxyphenyl)ethylamine hydrochloride was prepared from 2-fluoro-4- methoxybenzaldehyde as described in US 201 10054173 A1 .
Example 1 : Preparation of 2-(2-fluoro-4-hydroxyphenyl)ethylamine-fert-butyl carbamate
2-(2-Fluoro-4-methoxyphenyl)ethylamine hydrochloride (270 g, 1 .6 mol) was carefully added portionwise to 48% HBr (1 .5 L) and stirred for 4 h at 140 °C. The reaction solution was reduced in vacuo, then acetonitrile (1 L) was added to the residue and was evaporated to dryness. The crude product was stirred in diisopropylether and recovered by filtration to afford 222 g (940 mmol, 59%) of desired phenol. The purified phenol was dissolved in dichloromethane (DCM, 1 .5 L) to which was added triethylamine (275 ml_), feri-butyloxycarbonyl anhydride (215 g, 990 mmol) in a solution of DCM (500 ml_), and was stirred for 30 min at room temperature. The reaction solution was washed with water, dried over Na2S04, then concentrated in vacuo. The residue was purified by flash silica column chromatography (4:1 cyclohexane: MTBE) to afford 220 g (860 mmol, 92%) of the desired product.
Example 2: Preparation of 2-[4-[[2-(trifluoromethyl)-4-pyridyl]oxy]phenyl]ethanamine hydrochloride To a solution of N-feri-butyloxycarbonyl-protected tyramine (130 g, 550 mmol), potassium carbonate (76 g, 550 mmol), and catalytic amounts of potassium iodide in N-methyl-2- pyrrolidone (NMP, 400 ml.) that was stirred for 10 minutes at room temperature was added 4- chloro-2-trifluoromethylpyridine (100 g, 550 mmol). The reaction mixture was then heated to 160°C for 6 h, cooled, poured into water and extracted with methyl-feri-butylether (MTBE, 3x). The combined organic layers were washed with succesivly with 3 N NaOH and water, dried over Na2S04, and reduced in vacuo. The crude residue was purified by flash silica gel column chromatography to afford a 48% (102 g, 265 mmol) yield of N-tert-butyloxycarbonyl-protected 2-[4-[[2-(trifluoromethyl)-4-pyridyl]oxy]phenyl]ethanamine. To a solution of this material in dioxane (200 ml.) was added 4 M HCI in dioxane (250 ml_). The solution was left overnight to
stir at room temperature, which resulted in precipitation of 2-[4-[[2-(trifluoromethyl)-4- pyridyl]oxy]phenyl]ethanamine hydrochloride. The reaction solution was evaporated and the residue was stirred with pentane and filtered to provide 100% (37 g, 104 mmol) of the desired amine hydrochloride.
Example 3: Preparation of 6-chloro-N-[2-[4-(6-chloro-5-methyl-pyrimidin-4- yl)oxyphenyl]ethyl]-5-methyl-pyrimidin-4-amine (V-8)
To a solution of 4-hydroxyphenylethylamine hydrochloride (300 mg) in dimethyl formamide (DMF, 5 mL) was added diisopropylethylamine (355 mg, 2 equiv.). The solution was stirred for 5 min before the addition of 4,6-dichloro-5-methylpyrimidine (224 mg, 1 equiv.). The reaction mixture was then stirred for 16 h at room temperature before it was concentrated in vacuo. The residue was dissolved in DMF (10 mL) and was stirred with sodium hydride (66 mg, 2.2 equiv.) for 20 min at room temperature. Another portion of 4,6-dichloro-5-methylpyrimidine (269 mg, 1 .2 equiv.) was introduced into the reaction mixture, which was then left to stir at room temperature for 48 h. The DMF was then removed in vacuo and the product was purified by flash silica column chromatography to afford 130 mg (24% yield) of the product as a white solid.
Example 4: Preparation of 6-chloro-N-[2-[2-fluoro-4-[[4-(trifluoromethyl)-2- pyridyl]oxy]phenyl]ethyl]-5-methyl-pyrimidin-4-amine (V-3)
To a solution of 2-[2-fluoro-4-[[4-(trifluoromethyl)-2-pyridyl]oxy]phenyl]ethanamine hydrochloride (378 mg, 1 mmol) in NMP (5 mL) was added diisopropylethylamine (0.40 mL, 2.3 mmol). The solution was stirred for 5 min at room temperature at which time 4,6-dichloro-5- methylpyrimidine (150 mg, 0.9 mmol) was added. The reaction mixture was stirred at 80 °C overnight then allowed to cool to room temperature. Water was added and was extracted with MTBE (3x). The combined organic layers were washed with water, dried over Na2S04, and concentrated in vacuo. The residue was purified by flash silica column chromatography to provide 330 mg (0.80 mmol, 84%) of the light yellow solid product.
Example 5: Preparation of methyl 6-[2-[2-fluoro-4-[[2-(trifluoromethyl)-4- pyridyl]oxy]phenyl]ethylamino]-5-methyl-pyrimidine-4-carboxylate (V-2)
To a solution of 6-chloro-N-[2-[2-fluoro-4-[[2-(trifluoromethyl)-4-pyridyl]oxy]phenyl]ethyl]-5- methyl-pyrimidin-4-amine (4.27 g, 0.01 mol) in MeOH (20 mL) in a high pressure reactor was added Pd(CH3CN)2Cl2,(100 mg), 1 ,1 '-bis-diphenylphosphino-ferrocene (100 mg) and triethylamine (3 g, 0.03 mol). The reactor was sealed and flushed with carbon monoxide. The reaction was heated to 100 °C overnight under 5 bar carbon monoxide. The catalyst was separated from the reaction by filtration and the methanol was removed in vacuo. The product was purified by flash silica gel column chromatography (50:1 MTBE:cyclohexanes) to afford 2.4 g (53%) of the desired ester.
Example 6: Preparation of 6-[2-[2-fluoro-4-[[2-(trifluoromethyl)-4- pyridyl]oxy]phenyl]ethylamino]-5-methyl-pyrimidine-4-carbonitrile (V-5)
A solution of methyl 6-[2-[2-fluoro-4-[[2-(trifluoromethyl)-4-pyridyl]oxy]phenyl]ethylamino]-5- methyl-pyrimidine-4-carboxylate (1 .2 g, 2.7 mmol) in
7 M NH3 in MeOH (12 mL) was stirred at 120°C overnight under a closed system. The residue was purified by flash silica column chromatography (1 :1 to 100:1 ethyl acetate : cyclohexanes) to provide the corresponding amide (0.40 g, 46%). To a cooled solution of the resulting amide (0.30 g, 0.7 mmol) in DCM (15 mL) was added triethylamine (0.15 mL, 1 .52 mmol) followed by slow addition of trifluoroacetic acid anhydride (0.17 g, 0.83 mmol). The reaction was stirred for 30 min and then concentrated in vacuo. The crude product was purified by flash silica column chromatography (1 :1 ethyl acetate:cyclohexanes) to provide 105 mg (36% yield) of desired product.
The compounds listed in Table V have been prepared in an analogous manner.
Table V: Compounds V-1 to V-20 of formula 1.2 as defined herein.
* The position of Rb on the phenyl ring is defined relative to the CR1R2-moiety bound to the phenyl ring as being in ortho (o-) or meta (m-) position, n = 0 indicates that no substituent Rb is present on the phenyl ring. m.p. = melting point (°C).
HPLC: HPLC-column Kinetex XB C18 1 ,7μ (50 x 2,1 mm); eluent: acetonitrile / water + 0.1 %
TFA (gradient from 5:95 to 100 : 0 in 1 .5 min at 60°C, flow gradient from 0.8 to 1.0 ml/min in 1 .5 min). MS: Quadrupol Electrospray lonisation, 80 V (positive mode).
II. Biological examples for fungicidal activity
The fungicidal action of the compounds I was demonstrated by the following experiments:
A. Glass house trials
The spray solutions were prepared in several steps: The stock solution were prepared: a mixture of acetone and/or dimethylsulfoxide and the wetting agent/emulsifier Wettol, which is based on ethoxylated alkylphenoles, in a relation (volume) solvent-emulsifier of 99 to 1 was added to 25 mg of the compound to give a total of 5 ml. Water was then added to total volume of 100 ml.
This stock solution was diluted with the described solvent-emulsifier-water mixture to the given concentration.
After the final cultivation period, the extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
Use example 1 : Control of late blight on tomatoes caused by Phytophthora infestans
Young seedlings of tomato plants were grown in pots. These plants were sprayed to run-off with an aqueous suspension, containing the concentration of active ingredient or their mixture mentioned in the table below. The next day, the treated plants were inoculated with an aqueous suspension of sporangia of Phytophthora infestans. After inoculation, the trial plants were immediately transferred to a humid chamber. After 6 days at 18 to 20°C and a relative humidity close to 100% the extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
In this test, the plants which had been treated with 250 ppm of the active compound
V-2, V-3, V-4, V-7, V-9, V-17, V-19 or V-20 showed a diseased leaf area of at most 20%, whereas the untreated plants showed 78% diseased leaf area. Use example 2: Preventative control of leaf blotch on wheat caused by Septoria tritici
Leaves of pot-grown wheat seedling were sprayed to run-off with an aqueous suspension of the active compound or their mixture, prepared as described. The plants were allowed to air-dry. At the following day the plants were inoculated with an aqueous spore suspension of Septoria tritici. Then the trial plants were immediately transferred to a humid chamber at 18 to 22°C and a relative humidity close to 100%. After 4 days the plants were transferred to a chamber with 18 to 22°C and a relative humidity close to 70%. After 4 weeks the extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
In this test, the plants which had been treated with 250 ppm of the active compound
V-1 , V-2, V-3, V-4, V-5, V-6, V-8, V-9, V-1 1 , V-12, V-13 or V-20 showed a diseased leaf area of at most 15%, whereas the untreated plants showed 92% diseased leaf area.
Use example 3: Preventative control of brown rust on wheat caused by Puccinia recondita. The first two developed leaves of pot-grown wheat seedling were sprayed to run-off with an aqueous suspension, containing the concentration of active ingredient or their mixture as described below.
The next day the plants were inoculated with spores of Puccinia recondita. To ensure the success the artificial inoculation, the plants were transferred to a humid chamber without light and a relative humidity of 95 to 99% and 20 to 24°C for 24 h. Then the trial plants were cultivated for 6 days in a greenhouse chamber at 20 to 24°C and a relative humidity between 65 and 70%. The extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
In this test, the plants which had been treated with 250 ppm of the active compound
V-1 , V-4, V-10, V-1 1 , V-14, V-19 or V-20 showed a diseased leaf area of at most 20%, whereas the untreated plants showed 84% diseased leaf area.
Use example 4: Protective control of soy bean rust on soy beans caused by Phakopsora pachyrhizi Leaves of pot-grown soy bean seedlings were sprayed to run-off with an aqueous suspension, containing the concentration of active ingredient or their mixture as described below. The plants were allowed to air-dry. The trial plants were cultivated for 1 day in a greenhouse chamber at 23 to 27°C and a relative humidity between 60 and 80%. Then the plants were inoculated with spores of Phakopsora pachyrhizi. To ensure the success the artificial inoculation, the plants were transferred to a humid chamber with a relative humidity of about 95% and 20 to 24°C for 24 h. The trial plants were cultivated for fourteen days in a greenhouse chamber at 23 to 27°C and a relative humidity between 60 and 80%. The extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
In this test, the plants which had been treated with 250 ppm of the active compound
V-1 , V-3, V-5, V-13, V-15 or V-20 showed a diseased leaf area of at most 20%, whereas the untreated plants showed 92% diseased leaf area.
Use example 5: Preventative fungicidal control of Botrytis cinerea on leaves of green pepper Young seedlings of green pepper were grown in pots to the 4 to 5 leaf stage. These plants were sprayed to run-off with an aqueous suspension, containing the concentration of active ingredient or their mixture mentioned in the table below. The next day the plants were inoculated with a aqueous biomalt solution containing the spore suspension of Botrytis cinerea. Then the plants were immediately transferred to a humid chamber and kept for 5 days at 22 to 24°C and a relative humidity close to 100 %.
In this test, the plants which had been treated with 250 ppm of the active compound V-6 or V-19 showed a diseased leaf area of at most 15%, whereas the untreated plants showed 96% diseased leaf area. Use example 6: Control of late blight on tomatoes caused by Phytophthora infestans
Young seedlings of tomato plants were grown in pots. These plants were sprayed to run-off with an aqueous suspension, containing the concentration of active ingredient or their mixture mentioned in the table below. After seven days the treated plants were inoculated with an aqueous suspension of sporangia of Phytophthora infestans. After inoculation, the trial plants were immediately transferred to a humid chamber and kept for 6 days at 18 to 20°C and a relative humidity close to 100%.
In this test, the plants which had been treated with 250 ppm of the active compound V-16 or V-18 showed a diseased leaf area of at most 20%, whereas the untreated plants showed 74% diseased leaf area.
Claims
We claim:
Compounds of formula I
wherein:
Ra5 is methyl or halomethyl;
Ra2, Ra6 independently of each other are hydrogen, halogen, CN , NO2, OH, SH, C1-C4- alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci-C4-alkylthio, C1-C4- haloalkylthio, Ci-C4-alkylsulfinyl, Ci-C4-haloalkylsulfinyl,
Ci-C4-alkylsulfonyl, Ci-C4-haloalkylsulfonyl, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy- Ci-C4-alkoxy, C2-C4-alkenyl, C2-C4-alkynyl, C2-C4-haloalkenyl, C2-C4-haloalkynyl, C3- Ce-cycloalkyl, C3-C8-cycloalkyloxy, C3-C8-cycloalkyl-Ci-C4-alkyl, NRARB, C(=0)R', C(=NOR")R"' or C(=NH)-0-R"';
RA, RB independently of one another are hydrogen, Ci-C4-alkyl,
C2-C4-alkenyl, C2-C4-alkynyl, phenyl, benzyl, Cs-Cs-cycloalkyI, Cs-Cs-cycloalk- enyl or C(=0)-R';
R' is hydrogen, OH, NH2, Ci-C4-alkyl, Ci-C4-haloalkyl, C2-C4-alkenyl, C2-C4-alkynyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci-C4-alkylamino or di(Ci-C4-alkyl)amino;
R" is hydrogen, Ci-C4-alkyl, Ci-C4-haloalkyl, C2-C4-alkenyl, C2-C4-alkynyl or
C 1 -C4-a I koxy-C 1 -C4-a I ky I ,
R'" is hydrogen or Ci-C4-alkyl;
R is hydrogen, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy,
Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-haloalkoxy-Ci-C4-alkyl, C2-C4-alkenyl, C2-C4-haloalkenyl, C2-C4-alkynyl, C2-C4-haloalkynyl, CN, CH2CN, NRARB or
CH2-0-C(=0)R';
R1 , R2 independently of each other are hydrogen, halogen, OH , CN,
Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci-C4-a I koxy-C 1-C4- alkyl, Ci-C4-haloalkoxy-Ci-C4-alkyl, C2-C4-alkenyl, C2-C4-haloalkenyl, C2-C4-alkynyl, C2-C4-haloalkynyl, Cs-Cs-cycloalkyI, Cs-Cs-cycloalkyloxy, C3-Cs-cycloalkyl-Ci-C4- alkyl, NRARB, C(=0)R', C(=NOR")R"', C(=NH)-0-R"' or benzyl,
wherein the phenyl moiety of benzyl is unsubstituted or carries 1 , 2 , 3, 4 or 5 substituents selected from the group consisting of CN, halogen,
Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, (Ci-C4-alkoxy)carbonyl and di(Ci-C4-alkyl)aminocarbonyl, or two radicals R1 and R2 that are bound to the same carbon atom form together with said carbon atom a saturated or partially unsaturated
3-, 4-, 5-, 6- or 7-membered carbocycle or a saturated or partially unsaturated 3-, 4-, 5-, 6- or 7-membered heterocycle, wherein the ring member atoms of the
abovementioned heterocycle include beside carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S, and wherein the abovementioned cycle is unsubstituted or carries 1 , 2, 3 or 4 substituents selected from halogen, CN, OH, SH, Ci-C4-alkyl, Ci-C4-alkoxy or Ci-C4-alkylthio; and
one or two CH2 groups of the abovementioned cycles may be replaced by one or two C(=0) or C(=S) groups respectively; n indicates the number of substituents Rb on the phenyl ring and n is 0, 1 , 2, 3 or 4;
Rb is halogen, CN, NO2, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, C2-C4-alkenyl, C2-C4-haloalkenyl, C2-C4-alkynyl, C2-C4-haloalkynyl, NRARB,
C(=NOR")R"' or C(=NH)-0-R"',
it being possible for n = 2, 3 or 4 that Rb are identical or different;
Het is a 5- or 6-membered heteroaryl, wherein the ring member atoms of the heteroaryl include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N,
O and S and wherein the heteroaryl is unsubstituted or carries 1 , 2, 3 or 4 identical or different groups Rc:
Rc is halogen, CN, N02, Ci-C6-alkyl, d-C6-haloalkyl, Ci-C6-alkoxy, Ci-C6-halo- alkoxy, Ci-C6-alkylthio, Ci-C6-haloalkylthio, Ci-C6-alkylsulfinyl, Ci-C6-halo- alkylsulfinyl, Ci-C6-alkylsulfonyl, Ci-C6-haloalkylsulfonyl, Ci-C6-alkoxy- Ci-C4-alkyl, Ci-C6-haloalkoxy-Ci-C4-alkyl, C2-C6-alkenyl, C2-C6-alkynyl, NRARB, C(=0)R', C(=NOR")R'", C(=NH)-0-R'", C3-C8-cycloalkyl, C3-C8-cycloalkyl-Ci-C4-alkyl, phenyl, phenyl-Ci-C4-alkyl, phenoxy, phenoxy- Ci-C4-alkyl or a 5- or 6-membered heteroaryl, wherein the ring member atoms of the heteroaryl include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S, and wherein the aforementioned cyclic radicals are unsubstituted or carry 1 , 2, 3 or 4 identical or different substituents Rd:
Rd is halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy or Ci-C4-haloalkoxy;
or two radicals Rc that are bound to adjacent ring member atoms of the Het group form together with said ring member atoms a fused
5-, 6- or 7-membered saturated, partially unsaturated or aromatic carbocycle or heterocycle, wherein the ring member atoms of the fused heterocycle include besides carbon atoms 1 , 2, 3 or 4 heteroatoms selected from the group of N, O and S, and wherein the fused carbocycle or heterocycle is unsubstituted or carries 1 , 2, 3 or 4 identical or different radicals groups Re:
Re is halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy or
Ci-C4-haloalkoxy; and the N-oxides and the agriculturally acceptable salts thereof. Compounds according to claim 1 , wherein Ra5 is methyl. Compounds according to claim 1 , wherein Ra5 is CF3.
Compounds according to any of claims 1 to 3, wherein Ra2 and Ra6 independently of each other are halogen, CN, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci- C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkoxy or (Ci-C4-alkoxy)carbonyl, and it being possible that one of both, Ra2 or Ra6, can in addition be hydrogen.
Compounds according to any of claims 1 to 4, wherein R is hydrogen.
Compounds according to any of claims 1 to 5, wherein n is 1 , 2, 3 or 4 and Rb is selected from the group of halogen, CN, NO2, Ci-C4-alkyl, Ci-C4-haloalkyl, Ci-C4-alkoxy,
Ci-C4-haloalkoxy and Ci-C4-alkoxy-Ci-C4-alkyl.
Compounds according to any of claims 1 to 6, wherein Het is selected from pyrimidin-2-yl, pyrimidin-3-yl, pyrimidin-4-yl, pyridin-2-yl, pyridin-3-yl, thiazol-2-yl, pyrazin-2-yl, pyridazin- 3-yl, 1 ,3,5-triazin-2-yl, and 1 ,2,4-triazin-3-yl.
Compounds according to any of claims 1 to 7, wherein Het carries 1 or 2 radicals Rc which are selected from F, CI, Br, CN, Ci-C2-alkylsulfonyl, Ci-C2-alkoxycarbonyl, aminocarbonyl, Ci-C2-alkylaminocarbonyl, di(Ci-C2-alkyl)aminocarbonyl, Ci-C2-alkoxy, CF3, CHF2, OCF3
A process for preparing compounds I as defined in claim 1 , which comprises reacting a compound of formula II
wherein Ra2, Ra5 and Ra6 are as defined in claim 1 and Hal is fluorine, chlorine or bromine, with a compound of formula III
wherein R, R1, R2, Rb, n, X and Het are as defined in claim 1 in the presence of a base or a catalyst or a combination of a base and a catalyst and in the presence of a solvent.
10. An agrochemical composition which comprises an auxiliary and at least one compound of formula I or an N-oxide or an agriculturally acceptable salt thereof, according to any of claims 1 to 8.
1 1 . An agrochemical composition according to claim 10 comprising at least one further active substance. 12. A method for combating phytopathogenic harmful fungi, which process comprises treating the fungi or the materials, plants, the soil or seeds to be protected against fungal attack, with an effective amount of at least one compound of formula I of an or an N-oxide or an agriculturally acceptable salt thereof, as defined in any of claims 1 to 8. 13. The use of compounds of formula I, their N-oxides and their agriculturally acceptable salts, as defined in any of claims 1 to 8 for combating phytopathogenic harmful fungi. 14. Seed comprising a compound of formula I, or an N-oxide or an agriculturally acceptable salt thereof, as defined in any of claims 1 to 8, in an amount of from 0.1 g to 10 kg per 100 kg of seed.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP12153758.3 | 2012-02-03 | ||
| EP12153758 | 2012-02-03 | ||
| EP12154196.5 | 2012-02-07 | ||
| EP12154196 | 2012-02-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013113787A1 true WO2013113787A1 (en) | 2013-08-08 |
Family
ID=47624098
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2013/051854 WO2013113787A1 (en) | 2012-02-03 | 2013-01-31 | Fungicidal pyrimidine compounds |
| PCT/EP2013/051855 WO2013113788A1 (en) | 2012-02-03 | 2013-01-31 | Fungicidal pyrimidine compounds |
| PCT/EP2013/051859 WO2013113791A1 (en) | 2012-02-03 | 2013-01-31 | Fungicidal pyrimidine compounds |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2013/051855 WO2013113788A1 (en) | 2012-02-03 | 2013-01-31 | Fungicidal pyrimidine compounds |
| PCT/EP2013/051859 WO2013113791A1 (en) | 2012-02-03 | 2013-01-31 | Fungicidal pyrimidine compounds |
Country Status (2)
| Country | Link |
|---|---|
| AR (3) | AR089881A1 (en) |
| WO (3) | WO2013113787A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014063642A1 (en) * | 2012-10-25 | 2014-05-01 | 中国中化股份有限公司 | Substituted pyrimidine compound and uses thereof |
| WO2015036058A1 (en) * | 2013-09-16 | 2015-03-19 | Basf Se | Fungicidal pyrimidine compounds |
| CN105753791A (en) * | 2014-12-19 | 2016-07-13 | 沈阳中化农药化工研发有限公司 | Substituted pyrimidine compound and use thereof |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111789120B (en) * | 2020-07-29 | 2022-04-01 | 湖南省植物保护研究所 | Bactericide imazalil ionic liquid, and preparation method and application thereof |
Citations (91)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3296272A (en) | 1965-04-01 | 1967-01-03 | Dow Chemical Co | Sulfinyl- and sulfonylpyridines |
| US3325503A (en) | 1965-02-18 | 1967-06-13 | Diamond Alkali Co | Polychloro derivatives of mono- and dicyano pyridines and a method for their preparation |
| GB2059955A (en) | 1979-09-13 | 1981-04-29 | Merck & Co Inc | Process for preparing methylvanillyl ketone |
| EP0141317A2 (en) | 1983-10-21 | 1985-05-15 | BASF Aktiengesellschaft | 7-Amino-azolo[1,5-a]pyrimidines and fungicides containing them |
| EP0152031A2 (en) | 1984-02-03 | 1985-08-21 | Shionogi & Co., Ltd. | Azolyl cycloalkanol derivatives and agricultural fungicides |
| EP0226917A1 (en) | 1985-12-20 | 1987-07-01 | BASF Aktiengesellschaft | Acrylic acid esters and fungicides containing these compounds |
| EP0243970A1 (en) | 1986-05-02 | 1987-11-04 | Stauffer Chemical Company | Fungicidal pyridyl imidates |
| EP0256503A2 (en) | 1986-08-12 | 1988-02-24 | Mitsubishi Kasei Corporation | Pyridinecarboxamide derivatives and their use as fungicide |
| EP0264217A2 (en) | 1986-10-08 | 1988-04-20 | Ube Industries, Ltd. | Aralkylaminopyrimidine derivative, process for producing thereof and insecticide, acaricide and fungicide containing said derivative as active ingredient |
| EP0276406A1 (en) * | 1986-12-12 | 1988-08-03 | Bayer Ag | Substituted 4-amino-pyrimidines |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0428941A1 (en) | 1989-11-10 | 1991-05-29 | Agro-Kanesho Co., Ltd. | Hexahydrotriazine compounds and insecticides |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0519211A1 (en) * | 1991-05-17 | 1992-12-23 | Hoechst Schering AgrEvo GmbH | Substituted 4-aminopyrimidine, process for their preparation and their use as parasiticide |
| EP0532022A1 (en) | 1991-09-13 | 1993-03-17 | Ube Industries, Ltd. | Acrylate compound, preparation process thereof and fungicide using the same |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995018795A1 (en) | 1994-01-07 | 1995-07-13 | Dowelanco | N-(4-pyrimidinyl)amide pesticides |
| EP0665225A1 (en) | 1994-02-01 | 1995-08-02 | Ube Industries, Ltd. | 4-Phenethylamino pyrimidine derivative, process for preparing the same and agricultural and horticultural chemical for controlling noxious organisms containing the same |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| DE19518073A1 (en) | 1995-05-17 | 1996-11-21 | Hoechst Ag | Substituted benzyloxycarbonylguanidines, process for their preparation, their use as a medicament or diagnostic agent and medicament containing them |
| WO1997028133A1 (en) | 1996-02-01 | 1997-08-07 | Bayer Aktiengesellschaft | Acylated 4-amino- and 4-hydrazinopyrimidines and their use as pesticides |
| WO1998003272A1 (en) | 1996-07-19 | 1998-01-29 | Ube Industries, Ltd. | Processes for the preparation of 4-amino-5-chloro-6-(1-fluoroethyl)pyrimidine compounds |
| DE19650197A1 (en) | 1996-12-04 | 1998-06-10 | Bayer Ag | 3-thiocarbamoylpyrazole derivatives |
| WO1998046608A1 (en) | 1997-04-14 | 1998-10-22 | American Cyanamid Company | Fungicidal trifluoromethylalkylamino-triazolopyrimidines |
| WO1999014187A1 (en) | 1997-09-18 | 1999-03-25 | Basf Aktiengesellschaft | Benzamidoxim derivatives, intermediate products and methods for preparing and using them as fungicides |
| WO1999024413A2 (en) | 1997-11-12 | 1999-05-20 | Bayer Aktiengesellschaft | Isothiazole carboxylic acid amides and the application thereof in order to protect plants |
| WO1999027783A1 (en) | 1997-12-04 | 1999-06-10 | Dow Agrosciences Llc | Fungicidal compositions and methods, and compounds and methods for the preparation thereof |
| WO2000029404A1 (en) | 1998-11-17 | 2000-05-25 | Kumiai Chemical Industry Co., Ltd. | Pyrimidinylbenzimidazole and triazinylbenzimidazole derivatives and agricultura/horticultural bactericides |
| WO2000046148A1 (en) | 1999-02-02 | 2000-08-10 | Sintokogio, Ltd. | Silica gel carrying titanium oxide photocatalyst in high concentration and method for preparation thereof |
| EP1028125A1 (en) | 1998-11-30 | 2000-08-16 | Isagro Ricerca S.r.l. | Dipeptide compounds having fungicidal activity and their agronomic use |
| EP1035122A1 (en) | 1999-03-11 | 2000-09-13 | Rohm And Haas Company | Heterocyclic subsituted isoxazolidines and their use as fungicides |
| WO2000065913A1 (en) | 1999-04-28 | 2000-11-09 | Takeda Chemical Industries, Ltd. | Sulfonamide derivatives |
| DE10021412A1 (en) | 1999-12-13 | 2001-06-21 | Bayer Ag | Fungicidal active ingredient combinations |
| WO2001054501A2 (en) | 2000-01-25 | 2001-08-02 | Syngenta Participations Ag | Herbicidal composition |
| EP1122244A1 (en) | 2000-02-04 | 2001-08-08 | Sumitomo Chemical Company, Limited | Uracil compounds and their use |
| WO2001056358A2 (en) | 2000-01-28 | 2001-08-09 | Rohm And Haas Company | Enhanced propertied pesticides |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2002022583A2 (en) | 2000-09-18 | 2002-03-21 | E. I. Du Pont De Nemours And Company | Pyridinyl amides and imides for use as fungicides |
| EP1201648A1 (en) | 1999-08-05 | 2002-05-02 | Kumiai Chemical Industry Co., Ltd. | Carbamate derivatives and agricultural/horticultural bactericides |
| WO2002040431A2 (en) | 2000-11-17 | 2002-05-23 | Dow Agrosciences Llc | Compounds having fungicidal activity and processes to make and use same |
| JP2002316902A (en) | 2001-04-20 | 2002-10-31 | Sumitomo Chem Co Ltd | Plant blight-preventing agent composition |
| WO2003010149A1 (en) | 2001-07-25 | 2003-02-06 | Bayer Cropscience Ag | Pyrazolylcarboxanilides as fungicides |
| WO2003011853A1 (en) | 2001-07-30 | 2003-02-13 | Dow Agrosciences Llc | 6-aryl-4-aminopicolinates and their use as herbicides |
| WO2003014103A1 (en) | 2001-08-03 | 2003-02-20 | Bayer Cropscience S.A. | Iodobenzopyran-4-one derivatives having fungicidal activity |
| WO2003016286A1 (en) | 2001-08-17 | 2003-02-27 | Sankyo Agro Company, Limited | 3-phenoxy-4-pyridazinol derivative and herbicide composition containing the same |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2003053145A1 (en) | 2001-12-21 | 2003-07-03 | Nissan Chemical Industries, Ltd. | Bactericidal composition |
| WO2003061388A1 (en) | 2002-01-18 | 2003-07-31 | Sumitomo Chemical Takeda Agro Company, Limited | Fused heterocyclic sulfonylurea compound, herbicide containing the same, and method of controlling weed with the same |
| WO2003064572A1 (en) | 2002-01-31 | 2003-08-07 | Exxonmobil Research And Engineering Company | Lubricating oil compositions with improved friction properties |
| WO2003066609A1 (en) | 2002-02-04 | 2003-08-14 | Bayer Cropscience Aktiengesellschaft | Disubstituted thiazolyl carboxanilides and their use as microbicides |
| WO2003074491A1 (en) | 2002-03-05 | 2003-09-12 | Syngenta Participations Ag | O-cyclopropyl-carboxanilides and their use as fungicides |
| WO2004026305A1 (en) | 2002-09-19 | 2004-04-01 | Eli Lilly And Company | Diaryl ethers as opioid receptor antagonist |
| WO2004049804A2 (en) | 2002-11-29 | 2004-06-17 | Syngenta Participations Ag | Fungicidal combinations for crop potection |
| WO2004083193A1 (en) | 2003-03-17 | 2004-09-30 | Sumitomo Chemical Company, Limited | Amide compound and bactericide composition containing the same |
| WO2005063721A1 (en) | 2003-12-19 | 2005-07-14 | E.I. Dupont De Nemours And Company | Herbicidal pyrimidines |
| WO2005087772A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005087773A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005095357A2 (en) | 2004-03-30 | 2005-10-13 | Taisho Pharmaceutical Co., Ltd. | Pyrimidine derivatives and methods of treatment related to the use thereof |
| WO2005120234A2 (en) | 2004-06-03 | 2005-12-22 | E.I. Dupont De Nemours And Company | Fungicidal mixtures of amidinylphenyl compounds |
| WO2005123690A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-difluoromethyl-pyrazol-4-carbonic acid-(ortho-phenyl)-anilides, and use thereof as a fungicide |
| WO2005123689A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-trifluoromethyl-pyrazole-4-carboxylic acid (ortho-phenyl)-anilides and to use thereof as fungicide |
| WO2006015866A1 (en) | 2004-08-12 | 2006-02-16 | Syngenta Participations Ag | Method for protecting useful plants or plant propagation material |
| AU2006201959A1 (en) | 1997-12-22 | 2006-06-01 | Bayer Healthcare Llc | Inhibition of Raf Kinase using Substituted Heterocyclic Ureas |
| WO2006072831A1 (en) | 2005-01-10 | 2006-07-13 | Pfizer Inc. | Pyrrolopyrazoles, potent kinase inhibitors |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| WO2006087325A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | 5-alkoxyalkyl-6-alkyl-7-amino-azolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said substances |
| DE102005009458A1 (en) | 2005-03-02 | 2006-09-07 | Bayer Cropscience Ag | pyrazolylcarboxanilides |
| WO2007006714A1 (en) | 2005-07-07 | 2007-01-18 | Medivir Ab | Cysteine protease inhibitors |
| WO2007020227A1 (en) | 2005-08-15 | 2007-02-22 | Boehringer Ingelheim International Gmbh | Method for producing betamimetics |
| WO2007020381A2 (en) | 2005-08-15 | 2007-02-22 | Syngenta Participations Ag | Process for the synthesis of mandipropamid and derivatives thereof |
| WO2007046809A1 (en) | 2005-10-21 | 2007-04-26 | Dow Agrosciences Llc | Thieno-pyrimidine compounds having fungicidal activity |
| WO2007082098A2 (en) | 2006-01-13 | 2007-07-19 | Dow Agrosciences Llc | 6-(poly-substituted aryl)-4-aminopicolinates and their use as herbicides |
| WO2007090624A2 (en) | 2006-02-09 | 2007-08-16 | Syngenta Participations Ag | A method of protecting a plant propagation material, a plant, and/or plant organs |
| WO2007096647A2 (en) | 2006-02-27 | 2007-08-30 | Sterix Limited | Diaryl compounds as non-steroidal inhibitors of 17-beta hydroxysteroid dehydrogenase and/or steroid sulphatase for the treatment of oestrogen-related diseases such as hormone dependent breast cancer |
| WO2008039882A1 (en) | 2006-09-30 | 2008-04-03 | Sanofi-Aventis U.S. Llc | A combination of niacin and a prostaglandin d2 receptor antagonist |
| WO2008046598A1 (en) | 2006-10-20 | 2008-04-24 | Laboratorios Almirall, S.A. | DERIVATIVES OF 4-(2-AMINO-1-HYDROXYETHYL)PHENOL AS AGONISTS OF THE β2 ADRENERGIC RECEPTOR |
| WO2008065393A1 (en) | 2006-11-29 | 2008-06-05 | Astrazeneca Ab | Hydantoin derivatives used as mmp inhibitors |
| US20080171761A1 (en) | 2007-01-12 | 2008-07-17 | Tomoharu Iino | Substituted spirochromanone derivatives |
| WO2008124757A1 (en) | 2007-04-10 | 2008-10-16 | Bristol-Myers Squibb Company | Thiazolyl compounds useful as kinase inhibitors |
| WO2008156726A1 (en) | 2007-06-20 | 2008-12-24 | Merck & Co., Inc. | Inhibitors of janus kinases |
| WO2010025451A2 (en) | 2008-08-29 | 2010-03-04 | Dow Agrosciences Llc | 5,8-difluoro-4-(2-(4-(heteroaryloxy)-phenyl)ethylamino)quinazolines and their use as agrochemicals |
| WO2010081692A1 (en) | 2009-01-19 | 2010-07-22 | Almirall, S.A. | Oxadiazole derivatives as slpl receptor agonists |
| WO2011025505A1 (en) | 2009-08-31 | 2011-03-03 | Dow Agrosciences Llc | Pteridines and their use as agrochemicals |
| US20110054173A1 (en) | 2009-08-31 | 2011-03-03 | Dow Agrosciences Llc | Pteridines and their use as agrochemicals |
| WO2011028657A1 (en) | 2009-09-01 | 2011-03-10 | Dow Agrosciences Llc | Synergistic fungicidal compositions containing a 5-fluoropyrimidine derivative for fungal control in cereals |
| WO2011032277A1 (en) | 2009-09-18 | 2011-03-24 | Boehringer Ingelheim International Gmbh | Quinazolinone derivatives as viral polymerase inhibitors |
| WO2011053835A1 (en) | 2009-10-30 | 2011-05-05 | Aton Pharma, Inc. | Stereoselective synthesis of metyrosine |
| WO2011088181A1 (en) | 2010-01-13 | 2011-07-21 | Tempero Pharmaceuticals, Inc. | Compounds and methods |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS54115388A (en) | 1978-03-01 | 1979-09-07 | Nippon Chemiphar Co Ltd | Acetic acid derivative and its preparation |
| US5439911A (en) | 1992-12-28 | 1995-08-08 | Shionogi & Co., Ltd. | Aminopyrimidine derivatives and their production and use |
| JP2007526230A (en) | 2003-06-25 | 2007-09-13 | エラン ファーマシューティカルズ,インコーポレイテッド | Methods and compositions for treating rheumatoid arthritis |
| EP1654260A4 (en) | 2003-08-01 | 2008-09-24 | Pfizer Prod Inc | 6-menbered heteroaryl compounds for the treatment of neurodegenerative disorders |
| WO2005014532A1 (en) | 2003-08-08 | 2005-02-17 | Transtech Pharma, Inc. | Aryl and heteroaryl compounds, compositions and methods of use |
| US7459562B2 (en) | 2004-04-23 | 2008-12-02 | Bristol-Myers Squibb Company | Monocyclic heterocycles as kinase inhibitors |
| MX2007008410A (en) | 2005-01-14 | 2007-08-21 | Hoffmann La Roche | Thiazole-4-carboxamide derivatives as mglur5 antagonists. |
| MX2007010972A (en) | 2005-03-09 | 2007-09-19 | Schering Corp | Fused thieno [2, 3-b] pyridine and thiazolo [5, 4-b] pyridine compounds for inhibiting ksp kinesin activity. |
| US20070021435A1 (en) | 2005-06-10 | 2007-01-25 | Gaul Michael D | Aminopyrimidines as kinase modulators |
| US8450351B2 (en) | 2005-12-20 | 2013-05-28 | Incyte Corporation | N-hydroxyamidinoheterocycles as modulators of indoleamine 2,3-dioxygenase |
| BRPI0716952A2 (en) | 2006-09-15 | 2013-10-29 | Reviva Pharmaceuticals Inc | COMPOUND, METHODS FOR TREATING AND / OR PREVENTING OBESITY OR CO-MORBID INDICATION RELATED TO OBESITY, AND FOR SYNTHESIZING CYCLOalkylmethylamine Derivatives |
| US8779154B2 (en) | 2006-09-26 | 2014-07-15 | Qinglin Che | Fused ring compounds for inflammation and immune-related uses |
| DK2562162T3 (en) | 2008-01-22 | 2015-11-23 | Dow Agrosciences Llc | N-cyano-4-amino-5-fluoro-pyrimidine derivatives as fungicides |
| PL2303846T3 (en) | 2008-06-24 | 2015-10-30 | Sanofi Sa | Substituted isoquinolines and isoquinolinones as rho kinase inhibitors |
| DE102008040168A1 (en) | 2008-07-04 | 2010-01-07 | Robert Bosch Gmbh | Solenoid valve for a fuel injector and fuel injector |
| US20100068197A1 (en) | 2008-07-11 | 2010-03-18 | Myriad Pharmaceuticals, Inc. | Pharmaceutical compounds as inhibitors of cell proliferation and the use thereof |
| NZ591366A (en) | 2008-09-11 | 2012-05-25 | Pfizer | Heteroaryls amide derivatives and their use as glucokinase activators |
| AR074209A1 (en) | 2008-11-24 | 2010-12-29 | Boehringer Ingelheim Int | USEFUL PYRIMIDINE DERIVATIVES FOR CANCER TREATMENT |
| KR101442897B1 (en) | 2009-05-28 | 2014-09-23 | 노파르티스 아게 | Substituted aminopropionic derivatives as neprilysin inhibitors |
| AU2011215963A1 (en) | 2010-02-10 | 2012-08-02 | Lexicon Pharmaceuticals, Inc. | Tryptophan hydroxylase inhibitors for the treatment of metastatic bone disease |
| CN102260263A (en) | 2010-05-26 | 2011-11-30 | 四川大学 | Diphenylamine purine derivatives, and preparation method and medicinal application thereof |
-
2013
- 2013-01-31 WO PCT/EP2013/051854 patent/WO2013113787A1/en active Application Filing
- 2013-01-31 WO PCT/EP2013/051855 patent/WO2013113788A1/en active Application Filing
- 2013-01-31 WO PCT/EP2013/051859 patent/WO2013113791A1/en active Application Filing
- 2013-02-01 AR ARP130100325 patent/AR089881A1/en unknown
- 2013-02-01 AR ARP130100326 patent/AR089882A1/en unknown
- 2013-02-01 AR ARP130100327 patent/AR089883A1/en unknown
Patent Citations (91)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3325503A (en) | 1965-02-18 | 1967-06-13 | Diamond Alkali Co | Polychloro derivatives of mono- and dicyano pyridines and a method for their preparation |
| US3296272A (en) | 1965-04-01 | 1967-01-03 | Dow Chemical Co | Sulfinyl- and sulfonylpyridines |
| GB2059955A (en) | 1979-09-13 | 1981-04-29 | Merck & Co Inc | Process for preparing methylvanillyl ketone |
| EP0141317A2 (en) | 1983-10-21 | 1985-05-15 | BASF Aktiengesellschaft | 7-Amino-azolo[1,5-a]pyrimidines and fungicides containing them |
| EP0152031A2 (en) | 1984-02-03 | 1985-08-21 | Shionogi & Co., Ltd. | Azolyl cycloalkanol derivatives and agricultural fungicides |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0226917A1 (en) | 1985-12-20 | 1987-07-01 | BASF Aktiengesellschaft | Acrylic acid esters and fungicides containing these compounds |
| EP0243970A1 (en) | 1986-05-02 | 1987-11-04 | Stauffer Chemical Company | Fungicidal pyridyl imidates |
| EP0256503A2 (en) | 1986-08-12 | 1988-02-24 | Mitsubishi Kasei Corporation | Pyridinecarboxamide derivatives and their use as fungicide |
| EP0264217A2 (en) | 1986-10-08 | 1988-04-20 | Ube Industries, Ltd. | Aralkylaminopyrimidine derivative, process for producing thereof and insecticide, acaricide and fungicide containing said derivative as active ingredient |
| EP0276406A1 (en) * | 1986-12-12 | 1988-08-03 | Bayer Ag | Substituted 4-amino-pyrimidines |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0428941A1 (en) | 1989-11-10 | 1991-05-29 | Agro-Kanesho Co., Ltd. | Hexahydrotriazine compounds and insecticides |
| EP0519211A1 (en) * | 1991-05-17 | 1992-12-23 | Hoechst Schering AgrEvo GmbH | Substituted 4-aminopyrimidine, process for their preparation and their use as parasiticide |
| EP0532022A1 (en) | 1991-09-13 | 1993-03-17 | Ube Industries, Ltd. | Acrylate compound, preparation process thereof and fungicide using the same |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995018795A1 (en) | 1994-01-07 | 1995-07-13 | Dowelanco | N-(4-pyrimidinyl)amide pesticides |
| EP0665225A1 (en) | 1994-02-01 | 1995-08-02 | Ube Industries, Ltd. | 4-Phenethylamino pyrimidine derivative, process for preparing the same and agricultural and horticultural chemical for controlling noxious organisms containing the same |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| DE19518073A1 (en) | 1995-05-17 | 1996-11-21 | Hoechst Ag | Substituted benzyloxycarbonylguanidines, process for their preparation, their use as a medicament or diagnostic agent and medicament containing them |
| WO1997028133A1 (en) | 1996-02-01 | 1997-08-07 | Bayer Aktiengesellschaft | Acylated 4-amino- and 4-hydrazinopyrimidines and their use as pesticides |
| WO1998003272A1 (en) | 1996-07-19 | 1998-01-29 | Ube Industries, Ltd. | Processes for the preparation of 4-amino-5-chloro-6-(1-fluoroethyl)pyrimidine compounds |
| DE19650197A1 (en) | 1996-12-04 | 1998-06-10 | Bayer Ag | 3-thiocarbamoylpyrazole derivatives |
| WO1998046608A1 (en) | 1997-04-14 | 1998-10-22 | American Cyanamid Company | Fungicidal trifluoromethylalkylamino-triazolopyrimidines |
| WO1999014187A1 (en) | 1997-09-18 | 1999-03-25 | Basf Aktiengesellschaft | Benzamidoxim derivatives, intermediate products and methods for preparing and using them as fungicides |
| WO1999024413A2 (en) | 1997-11-12 | 1999-05-20 | Bayer Aktiengesellschaft | Isothiazole carboxylic acid amides and the application thereof in order to protect plants |
| WO1999027783A1 (en) | 1997-12-04 | 1999-06-10 | Dow Agrosciences Llc | Fungicidal compositions and methods, and compounds and methods for the preparation thereof |
| AU2006201959A1 (en) | 1997-12-22 | 2006-06-01 | Bayer Healthcare Llc | Inhibition of Raf Kinase using Substituted Heterocyclic Ureas |
| WO2000029404A1 (en) | 1998-11-17 | 2000-05-25 | Kumiai Chemical Industry Co., Ltd. | Pyrimidinylbenzimidazole and triazinylbenzimidazole derivatives and agricultura/horticultural bactericides |
| EP1028125A1 (en) | 1998-11-30 | 2000-08-16 | Isagro Ricerca S.r.l. | Dipeptide compounds having fungicidal activity and their agronomic use |
| WO2000046148A1 (en) | 1999-02-02 | 2000-08-10 | Sintokogio, Ltd. | Silica gel carrying titanium oxide photocatalyst in high concentration and method for preparation thereof |
| EP1035122A1 (en) | 1999-03-11 | 2000-09-13 | Rohm And Haas Company | Heterocyclic subsituted isoxazolidines and their use as fungicides |
| WO2000065913A1 (en) | 1999-04-28 | 2000-11-09 | Takeda Chemical Industries, Ltd. | Sulfonamide derivatives |
| EP1201648A1 (en) | 1999-08-05 | 2002-05-02 | Kumiai Chemical Industry Co., Ltd. | Carbamate derivatives and agricultural/horticultural bactericides |
| DE10021412A1 (en) | 1999-12-13 | 2001-06-21 | Bayer Ag | Fungicidal active ingredient combinations |
| WO2001054501A2 (en) | 2000-01-25 | 2001-08-02 | Syngenta Participations Ag | Herbicidal composition |
| WO2001056358A2 (en) | 2000-01-28 | 2001-08-09 | Rohm And Haas Company | Enhanced propertied pesticides |
| EP1122244A1 (en) | 2000-02-04 | 2001-08-08 | Sumitomo Chemical Company, Limited | Uracil compounds and their use |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2002022583A2 (en) | 2000-09-18 | 2002-03-21 | E. I. Du Pont De Nemours And Company | Pyridinyl amides and imides for use as fungicides |
| WO2002040431A2 (en) | 2000-11-17 | 2002-05-23 | Dow Agrosciences Llc | Compounds having fungicidal activity and processes to make and use same |
| JP2002316902A (en) | 2001-04-20 | 2002-10-31 | Sumitomo Chem Co Ltd | Plant blight-preventing agent composition |
| WO2003010149A1 (en) | 2001-07-25 | 2003-02-06 | Bayer Cropscience Ag | Pyrazolylcarboxanilides as fungicides |
| WO2003011853A1 (en) | 2001-07-30 | 2003-02-13 | Dow Agrosciences Llc | 6-aryl-4-aminopicolinates and their use as herbicides |
| WO2003014103A1 (en) | 2001-08-03 | 2003-02-20 | Bayer Cropscience S.A. | Iodobenzopyran-4-one derivatives having fungicidal activity |
| WO2003016286A1 (en) | 2001-08-17 | 2003-02-27 | Sankyo Agro Company, Limited | 3-phenoxy-4-pyridazinol derivative and herbicide composition containing the same |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2003053145A1 (en) | 2001-12-21 | 2003-07-03 | Nissan Chemical Industries, Ltd. | Bactericidal composition |
| WO2003061388A1 (en) | 2002-01-18 | 2003-07-31 | Sumitomo Chemical Takeda Agro Company, Limited | Fused heterocyclic sulfonylurea compound, herbicide containing the same, and method of controlling weed with the same |
| WO2003064572A1 (en) | 2002-01-31 | 2003-08-07 | Exxonmobil Research And Engineering Company | Lubricating oil compositions with improved friction properties |
| WO2003066609A1 (en) | 2002-02-04 | 2003-08-14 | Bayer Cropscience Aktiengesellschaft | Disubstituted thiazolyl carboxanilides and their use as microbicides |
| WO2003074491A1 (en) | 2002-03-05 | 2003-09-12 | Syngenta Participations Ag | O-cyclopropyl-carboxanilides and their use as fungicides |
| WO2004026305A1 (en) | 2002-09-19 | 2004-04-01 | Eli Lilly And Company | Diaryl ethers as opioid receptor antagonist |
| WO2004049804A2 (en) | 2002-11-29 | 2004-06-17 | Syngenta Participations Ag | Fungicidal combinations for crop potection |
| WO2004083193A1 (en) | 2003-03-17 | 2004-09-30 | Sumitomo Chemical Company, Limited | Amide compound and bactericide composition containing the same |
| WO2005063721A1 (en) | 2003-12-19 | 2005-07-14 | E.I. Dupont De Nemours And Company | Herbicidal pyrimidines |
| WO2005087772A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005087773A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005095357A2 (en) | 2004-03-30 | 2005-10-13 | Taisho Pharmaceutical Co., Ltd. | Pyrimidine derivatives and methods of treatment related to the use thereof |
| WO2005120234A2 (en) | 2004-06-03 | 2005-12-22 | E.I. Dupont De Nemours And Company | Fungicidal mixtures of amidinylphenyl compounds |
| WO2005123690A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-difluoromethyl-pyrazol-4-carbonic acid-(ortho-phenyl)-anilides, and use thereof as a fungicide |
| WO2005123689A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-trifluoromethyl-pyrazole-4-carboxylic acid (ortho-phenyl)-anilides and to use thereof as fungicide |
| WO2006015866A1 (en) | 2004-08-12 | 2006-02-16 | Syngenta Participations Ag | Method for protecting useful plants or plant propagation material |
| WO2006072831A1 (en) | 2005-01-10 | 2006-07-13 | Pfizer Inc. | Pyrrolopyrazoles, potent kinase inhibitors |
| WO2006087325A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | 5-alkoxyalkyl-6-alkyl-7-amino-azolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said substances |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| DE102005009458A1 (en) | 2005-03-02 | 2006-09-07 | Bayer Cropscience Ag | pyrazolylcarboxanilides |
| WO2007006714A1 (en) | 2005-07-07 | 2007-01-18 | Medivir Ab | Cysteine protease inhibitors |
| WO2007020227A1 (en) | 2005-08-15 | 2007-02-22 | Boehringer Ingelheim International Gmbh | Method for producing betamimetics |
| WO2007020381A2 (en) | 2005-08-15 | 2007-02-22 | Syngenta Participations Ag | Process for the synthesis of mandipropamid and derivatives thereof |
| WO2007046809A1 (en) | 2005-10-21 | 2007-04-26 | Dow Agrosciences Llc | Thieno-pyrimidine compounds having fungicidal activity |
| WO2007082098A2 (en) | 2006-01-13 | 2007-07-19 | Dow Agrosciences Llc | 6-(poly-substituted aryl)-4-aminopicolinates and their use as herbicides |
| WO2007090624A2 (en) | 2006-02-09 | 2007-08-16 | Syngenta Participations Ag | A method of protecting a plant propagation material, a plant, and/or plant organs |
| WO2007096647A2 (en) | 2006-02-27 | 2007-08-30 | Sterix Limited | Diaryl compounds as non-steroidal inhibitors of 17-beta hydroxysteroid dehydrogenase and/or steroid sulphatase for the treatment of oestrogen-related diseases such as hormone dependent breast cancer |
| WO2008039882A1 (en) | 2006-09-30 | 2008-04-03 | Sanofi-Aventis U.S. Llc | A combination of niacin and a prostaglandin d2 receptor antagonist |
| WO2008046598A1 (en) | 2006-10-20 | 2008-04-24 | Laboratorios Almirall, S.A. | DERIVATIVES OF 4-(2-AMINO-1-HYDROXYETHYL)PHENOL AS AGONISTS OF THE β2 ADRENERGIC RECEPTOR |
| WO2008065393A1 (en) | 2006-11-29 | 2008-06-05 | Astrazeneca Ab | Hydantoin derivatives used as mmp inhibitors |
| US20080171761A1 (en) | 2007-01-12 | 2008-07-17 | Tomoharu Iino | Substituted spirochromanone derivatives |
| WO2008124757A1 (en) | 2007-04-10 | 2008-10-16 | Bristol-Myers Squibb Company | Thiazolyl compounds useful as kinase inhibitors |
| WO2008156726A1 (en) | 2007-06-20 | 2008-12-24 | Merck & Co., Inc. | Inhibitors of janus kinases |
| WO2010025451A2 (en) | 2008-08-29 | 2010-03-04 | Dow Agrosciences Llc | 5,8-difluoro-4-(2-(4-(heteroaryloxy)-phenyl)ethylamino)quinazolines and their use as agrochemicals |
| WO2010081692A1 (en) | 2009-01-19 | 2010-07-22 | Almirall, S.A. | Oxadiazole derivatives as slpl receptor agonists |
| WO2011025505A1 (en) | 2009-08-31 | 2011-03-03 | Dow Agrosciences Llc | Pteridines and their use as agrochemicals |
| US20110054173A1 (en) | 2009-08-31 | 2011-03-03 | Dow Agrosciences Llc | Pteridines and their use as agrochemicals |
| WO2011028657A1 (en) | 2009-09-01 | 2011-03-10 | Dow Agrosciences Llc | Synergistic fungicidal compositions containing a 5-fluoropyrimidine derivative for fungal control in cereals |
| WO2011032277A1 (en) | 2009-09-18 | 2011-03-24 | Boehringer Ingelheim International Gmbh | Quinazolinone derivatives as viral polymerase inhibitors |
| WO2011053835A1 (en) | 2009-10-30 | 2011-05-05 | Aton Pharma, Inc. | Stereoselective synthesis of metyrosine |
| WO2011088181A1 (en) | 2010-01-13 | 2011-07-21 | Tempero Pharmaceuticals, Inc. | Compounds and methods |
Non-Patent Citations (65)
| Title |
|---|
| "McCutcheon's, Vol.1: Emulsifiers & Detergents", 2008, MCCUTCHEON'S DIRECTORIES |
| "Technical Monograph No. 2, 6th Ed.", May 2008, CROPLIFE INTERNATIONAL., article "Catalogue of pesticide formulation types and international coding system" |
| ACTA PHARMACEUTICA SUECICA, vol. 13, no. 1, 1976, pages 65 - 74 |
| ADV. SYNTH. CATAL., vol. 353, no. 2, 3, 2011, pages 501 - 507 |
| ARCHIV DER PHARMAZIE, vol. 344, no. 6, 2011, pages 372 - 385 |
| BIOORG. MED. CHEM. LETT., vol. 17, no. 4, 2007, pages 974 - 977 |
| BIOORG. MED. CHEM., vol. 12, no. 15, 2004, pages 4055 - 4066 |
| CAN. J. PLANT SCI., vol. 48, no. 6, 1968, pages 587 - 94 |
| CHEM. BIODIVERSITY, vol. 2, no. 9, 2005, pages 1217 - 1231 |
| CHEM. PHARM. BULL., vol. 51, no. 6, 2003, pages 702 - 709 |
| CHEMISTRY A EUR. J., vol. 17, no. 44, 2011, pages 12276 - 12279 |
| CHIRALITY, vol. 21, no. 9, 2009, pages 836 - 842 |
| EUR. J. INORG. CHEM., 2011, pages 3381 - 3386 |
| EUR. J. MED. CHEM., vol. 44, no. 5, 2009, pages 2246 - 2251 |
| EUR. J. MED. CHEM., vol. 45, no. 1, 2010, pages 11 - 18 |
| EUR. J. MED. CHEM., vol. 46, no. 9, 2011, pages 3986 - 3995 |
| EUR. J. ORG. CHEM., no. 19, 2002, pages 3243 - 3249 |
| HETEROCYCLES, vol. 78, no. 7, 2009, pages 1627 - 1665 |
| J. AM CHEM. SOC., vol. 127, no. 29, 2005, pages 10186 - 10187 |
| J. AM. CHEM. SOC., vol. 107, no. 12, 1985, pages 3601 - 6 |
| J. AM. CHEM. SOC., vol. 121, no. 18, 1999, pages 4369 - 4378 |
| J. AM. CHEM. SOC., vol. 123, no. 25, 2001, pages 5962 - 5973 |
| J. AM. CHEM. SOC., vol. 133, no. 31, 2011, pages 12197 - 12219 |
| J. AM. CHEM. SOC., vol. 133, no. 33, 2011, pages 12914 - 12917 |
| J. AM. CHEM. SOC., vol. 55, 1933, pages 2593 - 7 |
| J. AM. CHEM. SOC., vol. 70, 1948, pages 3738 - 40 |
| J. AM. CHEM. SOC., vol. 74, 1952, pages 4611 - 15 |
| J. CHEM. SOC., 1959, pages 1780 - 2 |
| J. HETEROCYC. CHEM., vol. 18, no. 7, 1981, pages 1305 - 1308 |
| J. MASS SPECTROMETRY, vol. 43, no. 4, 2008, pages 528 - 534 |
| J. MED. CHEM., vol. 19, no. 6, 1976, pages 763 - 6 |
| J. MED. CHEM., vol. 38, no. 11, 1995, pages 1892 - 1903 |
| J. MED. CHEM., vol. 50, no. 20, 2007, pages 5003 - 5011 |
| J. ORG. CHEM, vol. 67, no. 14, 2002, pages 4875 - 4881 |
| J. ORG. CHEM., vol. 46, no. 4, 1981, pages 783 - 8 |
| J. ORG. CHEM., vol. 50, no. 20, 1985, pages 3878 - 81 |
| J. ORG. CHEM., vol. 60, no. 13, 1995, pages 4204 - 12 |
| J. ORG. CHEM., vol. 70, no. 14, 2005, pages 5519 - 5527 |
| J. ORG. CHEM., vol. 72, no. 22, 2007, pages 8422 - 8426 |
| J. ORG. CHEM., vol. 73, no. 18, 2008, pages 7373 - 7375 |
| JINGXI HUAGONG ZHONGJIANTI, vol. 40, no. 3, 2010, pages 26 - 28 |
| JUSTUS LIEBIGS ANNALEN DER CHEMIE, vol. 564, 1949, pages 49 - 54 |
| JUSTUS LIEBIGS ANNALEN DER CHEMIE, vol. 605, 1957, pages 200 - 11 |
| KNOWLES: "Agrow Reports DS243", 2005, T&F INFORMA, article "New developments in crop protection product formulation" |
| KNOWLES: "Agrow Reports DS256", 2006, T&F INFORMA UK, article "Adjuvants and additives" |
| MOLLET; GRUBEMANN: "Formulation technology", 2001, WILEY VCH |
| NEW J. CHEM., vol. 18, no. 6, 1994, pages 701 - 8 |
| ORG. BIOMOL. CHEM., vol. 9, no. 23, 2011, pages 8171 - 8177 |
| ORG. LETT., vol. 3, no. 26, 2001, pages 4315 - 4317 |
| ORG. LETT., vol. 8, no. 20, 2006, pages 4481 - 4484 |
| ORG. LETT., vol. 9, no. 2, 2007, pages 203 - 206 |
| ORGANIC REACTIONS, 2000, pages 56 |
| PESTIC. SCI., vol. 44, no. 4, 1995, pages 341 - 355 |
| SCIENCE OF SYNTHESIS, vol. 16, 2004, pages 379 - 572 |
| SYNLETT, 2008, pages 1857 - 1861 |
| SYNLETT, no. 3, 2003, pages 353 - 356 |
| TARGETS IN HETEROCYCLIC SYSTEMS, vol. 12, 2008, pages 59 - 84 |
| TETRAHEDRON LETT., vol. 26, no. 36, 1985, pages 4299 - 300 |
| TETRAHEDRON LETT., vol. 44, no. 12, 2003, pages 2557 - 2560 |
| TETRAHEDRON LETT., vol. 48, no. 38, 2007, pages 6779 - 6784 |
| TETRAHEDRON, vol. 44, no. 15, 1988, pages 4737 - 46 |
| TETRAHEDRON, vol. 55, no. 43, 1999, pages 12493 - 12514 |
| TETRAHEDRON, vol. 57, no. 40, 2001, pages 8573 - 8580 |
| TETRAHEDRON, vol. 58, no. 8, 2002, pages 1513 - 1518 |
| TETRAHEDRON, vol. 67, no. 42, 2011, pages 8183 - 8186 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014063642A1 (en) * | 2012-10-25 | 2014-05-01 | 中国中化股份有限公司 | Substituted pyrimidine compound and uses thereof |
| CN104684900A (en) * | 2012-10-25 | 2015-06-03 | 中国中化股份有限公司 | Substituted pyrimidine compound and uses thereof |
| WO2015036058A1 (en) * | 2013-09-16 | 2015-03-19 | Basf Se | Fungicidal pyrimidine compounds |
| CN105722833A (en) * | 2013-09-16 | 2016-06-29 | 巴斯夫欧洲公司 | Fungicidal pyrimidine compounds |
| CN105753791A (en) * | 2014-12-19 | 2016-07-13 | 沈阳中化农药化工研发有限公司 | Substituted pyrimidine compound and use thereof |
| CN105753791B (en) * | 2014-12-19 | 2019-09-13 | 沈阳中化农药化工研发有限公司 | Substituted uracil compound and application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2013113791A1 (en) | 2013-08-08 |
| AR089882A1 (en) | 2014-09-24 |
| AR089881A1 (en) | 2014-09-24 |
| AR089883A1 (en) | 2014-09-24 |
| WO2013113788A1 (en) | 2013-08-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2016299729B2 (en) | Pyridine compounds useful for combating phytopathogenic fungi | |
| US9072301B2 (en) | Fungicidal pyrimidine compounds | |
| US9055750B2 (en) | Fungicidal pyrimidine compounds | |
| EP3359530A1 (en) | Pyridine derivatives for combating phytopathogenic fungi | |
| WO2014095548A1 (en) | Substituted [1,2,4]triazole compounds and their use as fungicides | |
| WO2015036058A1 (en) | Fungicidal pyrimidine compounds | |
| WO2014095655A1 (en) | Substituted [1,2,4]triazole and imidazole compounds | |
| WO2014095381A1 (en) | Fungicidal imidazolyl and triazolyl compounds | |
| WO2013113776A1 (en) | Fungicidal pyrimidine compounds | |
| WO2015036059A1 (en) | Fungicidal pyrimidine compounds | |
| WO2013113787A1 (en) | Fungicidal pyrimidine compounds | |
| WO2013113781A1 (en) | Fungicidal pyrimidine compounds i | |
| WO2014095637A1 (en) | Substituted [1,2,4]triazole and imidazole compounds | |
| JP2015512891A (en) | Bactericidal pyrimidine compounds | |
| WO2013113778A1 (en) | Fungicidal pyrimidine compounds | |
| WO2013113773A1 (en) | Fungicidal pyrimidine compounds | |
| WO2013113716A1 (en) | Fungicidal pyrimidine compounds | |
| WO2013135672A1 (en) | Fungicidal pyrimidine compounds | |
| WO2013113719A1 (en) | Fungicidal pyrimidine compounds ii | |
| WO2013113720A1 (en) | Fungicidal pyrimidine compounds | |
| WO2013113782A1 (en) | Fungicidal pyrimidine compounds | |
| EP2746264A1 (en) | Substituted [1,2,4]triazole and imidazole compounds | |
| EP2746274A1 (en) | Substituted [1,2,4]triazole compounds | |
| EP2745691A1 (en) | Substituted imidazole compounds and their use as fungicides |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13702046 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13702046 Country of ref document: EP Kind code of ref document: A1 |