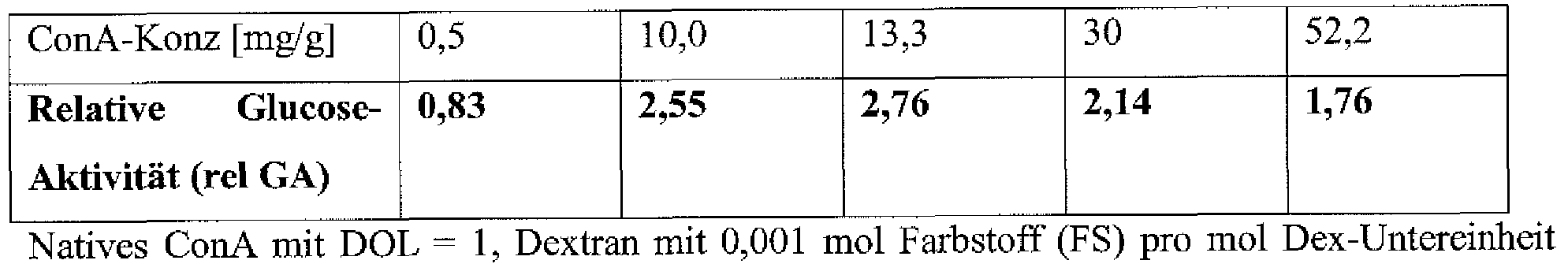WO2012080218A1 - Kompetitiver biosensor mit erhöhter sensitivität - Google Patents
Kompetitiver biosensor mit erhöhter sensitivität Download PDFInfo
- Publication number
- WO2012080218A1 WO2012080218A1 PCT/EP2011/072563 EP2011072563W WO2012080218A1 WO 2012080218 A1 WO2012080218 A1 WO 2012080218A1 EP 2011072563 W EP2011072563 W EP 2011072563W WO 2012080218 A1 WO2012080218 A1 WO 2012080218A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- glucose
- binding protein
- poly
- hydrogel
- concanavalin
- Prior art date
Links
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/543—Immunoassay; Biospecific binding assay; Materials therefor with an insoluble carrier for immobilising immunochemicals
- G01N33/5436—Immunoassay; Biospecific binding assay; Materials therefor with an insoluble carrier for immobilising immunochemicals with ligand physically entrapped within the solid phase
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/536—Immunoassay; Biospecific binding assay; Materials therefor with immune complex formed in liquid phase
- G01N33/542—Immunoassay; Biospecific binding assay; Materials therefor with immune complex formed in liquid phase with steric inhibition or signal modification, e.g. fluorescent quenching
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/66—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving blood sugars, e.g. galactose
Definitions
- the present invention relates to measures for the determination of glucose and for the diagnosis of diseases based on a disturbed glucose metabolism.
- the present invention relates to a device comprising a hydrogel with a stored therein
- a glucose-binding protein and a ligand of the glucose-binding protein wherein the hydrogel comprises a first hydrogel matrix of alginate and a second hydrogel matrix which forms a parent-penetrating network within the first hydrogel matrix.
- the invention further relates to the use of such a device for determining the glucose content in a sample and the use of the device for diagnosing a disturbed glucose
- Z0 range for example in the diagnosis of diseases caused by a disturbed glucose metabolism, e.g. Diabetes mellitus or metabolic syndrome, the rapid and reliable determination of glucose concentration in given solutions plays a central role.
- glucose sensors are used both in devices
- the sensor is a glucose-binding protein and the ligand is a competitor for glucose, which is present in the sensor bound to the glucose sensor molecule.
- the competitor is displaced by the glucose-binding protein. Displacement of the competitor by glucose from - -
- Glucose-binding protein may be modified by a change from a physical or chemical property of the molecules, e.g. by fluorescence resonance energy transfer (FRET).
- FRET fluorescence resonance energy transfer
- the aforementioned systems must be present in a spatially limited area of the sensor.
- hydrogels have been proven, inter alia.
- Suitable hydrogels can be polyethylene glycols, but also alginates (eg US2007 / 0105176, US 6,485,703, Russell 1999, Anal Chem 71: 3126-3132).
- sensors were described in which the aforementioned systems were enclosed in an aqueous medium by a semipermeable membrane.
- membranes may for example consist of regenerated cellulose, polyethylene glycol, polyurethane, layer-by-layer (LBL) layers, polyethersulfones, parylene layers or perforated silica (eg US2007 / 0122829).
- the glucose sensors described in the prior art have a relatively low glucose activity.
- the glucose activity and thus the sensor performance is significantly determined by the binding constants for the complexes of glucose-binding protein and competitor as well as glucose-binding protein and glucose.
- an analyte receptor with almost any high binding constant can be selected, since the analyte does not have to be released again and one usually has a very high binding constant and a very high specificity quality can receive.
- the situation is different, since the sensor must always react reversibly to changes in the concentration of the analyte.
- the binding constant should not be too high, otherwise the sensor would be saturated even at low analyte concentrations and could no longer show concentration changes.
- Another problem of the in vivo sensors is that the range of analyte concentration is fixed and can not be optimized by dilutions or concentrations.
- the prior art thus essentially describes systems of ligand and glucose-binding protein in which average binding constants are realized. Adjustment of the sensitivities by changing the concentrations of either glucose-binding protein or ligand or both are usually due to the low solubility of the corresponding molecules limited. In this respect, in the sensors described in the aforementioned prior art, both the measuring sensitivity and the measuring accuracy of the sensor are limited (see Rounds 2007, J. Fluorec 17: 57-63).
- the object of the present invention is to provide a device which allows a more efficient determination of the level of glucose also in vivo and in which the aforementioned disadvantages are substantially eliminated.
- the invention is solved by the embodiments described in the claims as well as the embodiments which are subsequently disclosed.
- the invention thus relates to a device comprising a hydrogel having incorporated therein a glucose-binding protein and a ligand of the glucose-binding protein, wherein the hydrogel comprises a first hydrogel matrix of alginate and a second hydrogel matrix which forms an interpenetrating network within the first hydrogel matrix
- the device according to the invention is also a composition consisting of the abovementioned components.
- hydrogel describes a water-containing polymer whose molecules are chemically or physically linked to a three-dimensional network.
- the polymer molecules can be linked to one another by covalent or ionic bonds or by looping or interweaving into the three-dimensional network.
- the polymers that form the hydrogel preferably contain hydrophilic polymer components that allow the uptake of aqueous solutions, as well as groups that are able to interact with the glucose-binding protein.
- the hydrogels of the invention comprise a first hydrogel matrix of alginate and a second hydrogel matrix which is capable of forming an interpenetrating network within the hydrogel matrix spanned by the alginate.
- This second hydrogel matrix preferably consists of a water-soluble polymer having at least one crosslinkable group per molecule and a molecular weight of at most 500,000. Most preferred is a molecular weight of at most 250,000, 200,000, 150,000, 100,000 or 50,000.
- the second hydrogel matrix is preferably selected from the group consisting of: polyvinyl alcohols (PVAs), polyethylene glycols (PEGs), poly (2-oxazolines), polyacrylamides (eg dimethylacrylamide), polyhydroxyacrylates (eg polyhydroxymethacrylate, polyhydroxyacrylamides, polyvinylpyrolinones), (2-methyl-3-ethyl [2-hydroxyethyl]) polymers, polyhydroxyalkanoates (PHAs), poly (2-methyl-2-oxazolines), poly (2-methyl) Ethyl-2-oxazolines), poly (2-hydroxyethyl-2-oxazolines), poly (2- (1- (hydroxymethyl) ethyl) -2-oxazolines), poly (hydroxyethyl methacrylate) (PHEMA), poly (hydroxyethyl acrylate) (PHEA Polyvinylpyrolidones, poly (dimethyl) acrylamide, poly (hydroxyethyl) acrylamide, polyvinyl alcohols (including
- An interpenetrating network according to the invention is preferably obtained by polymerizing the monomers of the second polymer in the presence of an already existing first polymer. This can be done particularly preferably by methods which are described in more detail in the exemplary embodiments. This ensures that an interpenetrating network of the second polymer can form in the already existing network of the first polymer.
- the polymer networks are thus intertwined and / or entwined.
- different polymers in mixtures exist as non-interwoven and / or interlaced separate networks side by side.
- the crosslinking mechanisms of the first and second polymers differ so that no mixed crosslinking occurs.
- the aforementioned alginate, which is intended to generate the first hydrogel matrix is crosslinked by ionic interactions.
- the aforementioned second polymers which are to form the interpenetrating network are all crosslinked by free radical or ionic polymerization.
- the second hydrogel matrix is formed from polyvinyl alcohol.
- the polyvinyl alcohol having a molecular weight of 10,000 to 100,000, more preferably 10,000 to 50,000, more preferably 10,000 to 20,000, most preferably 15,000.
- the polyvinyl alcohol particularly preferably has a crosslinker content of at most 0.5 mmol / g, 0.4 mmol / g, 0.35 mmol / g, or 0.3 mmol / g, and very particularly preferably 0.35 mmol / g.
- the polyvinyl alcohol most preferably has a solids content of the prepolymer of less than 40% by weight.
- the hydrogel according to the invention may comprise additives in addition to the first hydrogel matrix and the second hydrogel matrix, for example stabilizers, emulsifiers, antioxidants, UV stabilizers, detergents, and / or UV initiators.
- the hydrogel used according to the invention may be enclosed by a further, preferably semi-permeable, shell material. This enclosure prevents "leaching" of the sensor components from the hydrogel.
- shell material come semi-permeable membranes or other hydrogel matrices in question.
- Semi-permeable membranes may preferably consist of regenerated cellulose, polyethylene glycol, polyurethane layer-by-layer (LBL) layers, polyethersulfones, parylene layers or perforated silica.
- a further hydrogel matrix may preferably be formed from a polymer selected from the group consisting of alginates, sepharoses, hyaluronic acid, chitosan, polyvinyl alcohols (PVAs), polyethylene glycols (PEGs), carrageenans and polyhydroxalkonates (PHAs), poly (2-methyl-2-oxazolines) , Poly (2-ethyl-2-oxazolines), poly (2-hydroxyethyl-2-oxazolines), poly (2- (1- (hydroxymethyl) ethyl) -2-oxazolines), poly (hydroxyethyl methacrylate) (PHEMA), poly (2-hydroxyethyl) (hydroxyethyl acrylate) (PHEA), poly-vinylpyrolidones, poly (dimethyl) acrylamide, poly (hydroxyethyl) acrylamide, polyvinyl alcohols (including copolymers with vinyl acetates and / or ethylene), poly (ethylene-co-viny
- glucose-binding protein in the context of the invention refers to proteins which are able to interact specifically with glucose. Whether a protein is capable of specifically interacting with glucose can readily be determined by one skilled in the art by binding assays known in the art. Particularly preferred is the glucose-binding protein selected from the group consisting of lectins, enzymes that bind glucose as a substrate, and antibodies that specifically recognize glucose.
- glucose-binding protein also includes surrogate molecules that can specifically recognize glucose, preferably aptamers that specifically recognize glucose, and most preferably the glucose-binding protein concanavalin A.
- Nucleic acid sequences and amino acid sequences encoding the aforementioned glucose-binding proteins are known in the art (Yamauchi 1990, FEBS Letters 260 (1): 127-130). Accordingly, the aforementioned proteins can be readily determined by those skilled in the art to be provided. The said proteins can be produced, for example, recombinantly or purified from a biological source. In addition, the proteins can also be chemically synthesized. Most of the aforementioned proteins are also commercially available. Antibodies or aptamers that specifically recognize glucose may be readily provided by those skilled in the art by methods known in the art for antibody or aptamer recovery.
- the aforementioned glucose-binding proteins and in particular concanavalin A may preferably also have chemical modifications which mediate increased water solubility compared to unmodified versions of the glucose-binding proteins.
- Such modifications preferably include functionalization with a water-soluble polymer and may be selected in a particularly preferred embodiment from the group consisting of: pegylation, acetylation, polyoxazolinylation and succinylation.
- the detection of glucose in a sample to be analyzed takes place in the device according to the invention by displacement of the ligand bound to the glucose-binding protein by the glucose contained in the sample (competition between ligand and glucose).
- the ligand has a lower affinity for the glucose-binding protein than the glucose.
- the displacement can preferably be detected by labeling the ligand with a dye or other detectable marker molecule.
- Dyes or other marker molecules have proven to be particularly suitable for the detection of displacement, which cause a change of at least one measurable physical or chemical property on approach of the molecules associated therewith.
- Suitable systems include those in which either a measurable signal is suppressed or generated by means of energy transfer between the dye molecules. Such systems based on energy transfer are described in more detail, for example, in WO2001 / 13783.
- these may be systems in which a fluorescence signal is suppressed by quenching effects when the dye or Withrioleküle- and thus the glucose binding protein and its ligand - are in spatial proximity.
- fluorescence photometers as described in WO2002 / 087429 can be used for the detection.
- fluorescence resonance energy transfer (FRET) based detection systems are so-called fluorescence resonance energy transfer (FRET) based detection systems.
- FRET fluorescence resonance energy transfer
- One component is coupled with an acceptor dye, the other with a donor dye.
- the dyes come into spatial proximity, thereby mediating the FRET effect, in which excitation energy is transferred from the donor to the acceptor dye, thus measurably reducing the intensity of the donor dye.
- glucose can be detected by the increase in the intensity of a signal generated by a dye or marker molecule after separation of the complex of glucose-binding protein and ligand by the analyte.
- the dye or marker molecule is coupled to either the glucose-binding protein or the ligand.
- a donor dye can be coupled to the glucose binding protein or ligand, with the non-donor coupled moiety coupled with a suitable acceptor dye.
- the FRET effect is observed before exposure to a glucose-containing sample as a result of the binding of the ligand to the glucose-binding protein.
- Birch et. al. (Birch 2001, Spectrochimica Acta Part A 57: 2245-2254) have calculated the mathematical solution of chemical equilibrium for the case of concanavalin A as a glucose-binding protein and dextran as a ligand. Simulations with varying concanavalin A and dextran concentrations have shown that the ratio (dextran) / (ConA-dextran complex) only insignificantly depends on the starting concentrations and that the binding constants Koex and KGI UC are the main factors for the sensor performance.
- a combination of a rhodamine and an oxazine dye is clearly superior to the conventionally used combination of a xanthene and a rhodamine dye (FITC-TMR).
- the glucose-binding protein used in the invention is linked to an oxazine dye.
- an oxazine acceptor selected from the group consisting of: AT-TO655, ATTO680, EVObluelO, EVOblue30, EVOblue90 and EVObluelOO.
- ATTO680 is used.
- the oxazine dyes mentioned are commercially available.
- Preferred degree of labeling (DOL) for the glucose-binding protein is 0.1 to 4, more preferably 1 to 4, and most preferably 1 to 3.
- the glucose binding protein is preferably functionalized with PEG.
- Preferred pegylation degree is 0.1 to 5, preferred molecular weight 200 to 10,000, particularly preferably 800 to 8000, further preferably 800 to 5000.
- the improved solubility is particularly relevant when the glucose-binding proteins are to be labeled with a dye, since glucose-binding proteins labeled with dyes, for example a concanavalin A modified with one of the abovementioned oxazine dyes, have a further reduced solubility in aqueous solution , The higher the degree of labeling, the lower the solubility in aqueous solution. However, a high degree of labeling is needed especially for glucose-binding proteins as sensor components in the devices according to the invention.
- the term "ligand of the glucose-binding protein” refers to a molecule capable of specific binding with the glucose-binding protein.
- the molecule interacts with essentially the same binding site as glucose, so that the bound molecule can be displaced by glucose from the binding site on the glucose-binding protein.
- Suitable molecules are therefore structurally related to glucose.
- the ligand of the glucose-binding protein is an oligosaccharide, a glycosylated macromolecule, eg a glycosylated protein or peptide, or a glycosylated nanoparticle.
- the aforementioned molecules, which can be used as ligands of the glucose-binding protein are known in the art and can be readily provided by one skilled in the art. Particular preference is given to using a dextran as the ligand of the glucose-binding protein.
- the ligand of the glucose-binding protein in the device of the present invention is coupled to a rhodamine dye.
- a rhodamine dye particularly preference may be given to using ATTO590, ATTO610, ROX, TMR, rhodamine G6, Alexa fluorine Rhodamine dyes or Dy590 and very particularly preferably ATTO590.
- Preferred degree of labeling (DOL) for the ligand of the glucose-binding protein, eg dextran is 0.00003 to 0.016 (moles of dye) / (mol subunit), more preferably 0.00032 to 0.0065 (moles of dye) / (mol of subunit ) and most preferably from 0.0008 to 0.0035 (moles of dye) / (mol subunit).
- the degree of labeling of the ligand also has an influence on the glucose activity. Too low levels of labeling result in poorer glucose activity as well as excessive. It can be seen from the above that, in a preferred embodiment of the device according to the invention, the glucose-binding protein is concanavalin A.
- the Concanavalin A concentration is greater than 0.5 mg / (g matrix) and most preferably between 2 to 60 mg / (g matrix).
- concanavalin A has increased water solubility as compared to unmodified concanavalin A by chemical modifications, preferably pegylation, acetylation, polyoxazolinylation or succinylation.
- the glucose-binding protein is linked in a preferred embodiment with an oxazine dye and the ligand of the glucose-binding protein with a rhodamine dye.
- a concanavalin A / dextran system is used in the device in which the dextran with a rhodamine donor dye and the Concanavalin A is linked to an oxazine acceptor dye.
- the components are preferably present in a mass ratio (Dextran / ConA) of 1: 1 to 1:40, with mass ratios close to 1:10 being particularly preferred.
- a hydrogel consisting of a first alginate hydrogel matrix and a second hydrogel matrix forming an interpenetrating network within the first one is capable of providing an environment for the sensor components, namely glucose binding protein and the competitive ligand of the glucose-binding protein, which allows an efficient determination of the glucose activity.
- Many fluorescence glucose measurement approaches are successful in solution, but lose their activity when the sensor components are embedded in hydrogel matrices, since the mobility of the sensor components is limited (Rounds 2007, J. Fluoresc. 17: 57-63; 0105176 AI).
- the solubility of the receptor component in free solution is still adversely affected by the addition of the ligand, since the receptor / competitor complex has a lower solubility due to its size and often due to multi valences.
- a Concanavalin A / dextran complex in solution at a mass ratio of 1:10 already starting from a concanavalin A concentration of 0.5 mg / (g solution) begins to precipitate
- the applicable concentration range can be extended 100 times. This is particularly important for applications where analyte concentration is fixed and can not be adjusted by dilution or concentration. Such difficulties occur especially in in vivo applications, such as the determination of the glucose level in on.
- the concentration of the analyte glucose in the in vivo situation does not adapt to the specific assay conditions, but must be taken for granted.
- solubility occurs especially in vivo applications of biological sensors. Due to the higher wavelength, the autofluorescence of the tissue decreases, so that in vivo applications long-wave fluorescent dyes are used. However, these fluorescent dyes are typically apolar due to their molecular structure and size (conjugated systems). If the glucose-binding protein is now labeled with such a dye, the solubility is lowered in addition, so that no high levels of labeling are possible. As already described above, it may therefore be necessary to functionalize the glucose-binding protein with, for example, polyethylene glycol in order to allow increased solubility and, associated therewith, higher levels of labeling.
- the glucose activity in the hydrogel of the device according to the invention is even increased by 4, 3 times.
- the use of the hydro gels in the inventive Device thus allows to set concentration ratios for the glucose-binding protein and its ligand, which allow at appropriate levels of labeling glucose activities that are increased by 4 times over concentrations that are adjustable in aqueous solutions. This advantageously also makes it possible to use the device according to the invention under conditions in which the analyte concentration can not be adapted to the assay conditions, eg in vivo applications.
- an analyte concentration of 50 to 500 mg / dL must be resolved.
- the devices can be used as a sensor both ex vivo and in vivo.
- the sensor device may be subcutaneous, in the eye, e.g. subconjunctival, or placed elsewhere in the body, which allow an evaluation of the measured glucose activity.
- the invention thus also relates to the use of a device according to the invention, as described above, for determining the glucose content in a sample.
- sample is to be understood as meaning a composition, preferably an aqueous composition, which presumably or actually contains glucose, and the sample is preferably a biological sample Specimen about a ⁇ ⁇ ⁇ ⁇ , in particular, tissue fluid (eg, interstitial fluid), blood, plasma, serum, lymph, saliva, tears, sweat or urine.
- tissue fluid eg, interstitial fluid
- the sample is particularly preferably tissue fluid, blood, serum or Plasma.
- the sample is a biological material, eg a body fluid, it may preferably be obtained from a subject who either or not actually has a disturbed glucose metabolism.
- the device according to the invention can thus be used for the ex vivo diagnosis of diseases or disorders of the glucose metabolism, in particular for the diagnosis of diabetes mellitus or the metabolic syndrome.
- the device according to the invention can be used not only for diagnosis, but also for monitoring the glucose level. The device thus also enables the support Therapy decisions, eg insulin doses, which must be administered in response to a change in glucose level.
- the device according to the invention can be introduced into the ex vivo application, for example, in microtiter plates and anchored there. Samples to be measured are then applied to the wells of the microtiter plates and can then be measured with a reader device. Such an approach allows the simultaneous measurement of a large number of samples and is therefore also inexpensive, especially in clinical diagnostics operation.
- the invention also relates to a method for determining the amount of glucose in a sample which actually or presumably contains glucose, comprising the steps:
- the method according to the invention described above may comprise further steps. For example, further steps may involve work-up of the sample, e.g. the recovery of serum from whole blood. Further steps could be taken to set the determined glucose level in relation to pathological changes in glucose metabolism. For this purpose, the determined amount could be compared with reference amounts which are indicative of certain pathological conditions, e.g. Diabetes mellitus or metabolic syndrome. Such methods can then also be used for in vitro diagnosis of diabetes mellitus or metabolic syndrome.
- the method according to the invention or individual steps thereof can be carried out automatically, e.g. through computer implementation and / or robotic systems.
- Quantity refers both to the determination of absolute quantities and of relative quantities.
- the determination of the absolute amount can preferably take place via a calibration curve which is produced from measured values for known glucose quantities with the method according to the invention.
- Relative quantities in the sense of the invention are quantities which are set in relation to a normalizing parameter. It goes without saying that parameters which can be derived from the determined quantity values by mathematical operations can also be determined within the scope of the method according to the invention.
- bringing into contact is intended to allow the sample and thus the glucose contained therein to penetrate into the device. Further, the contacting should allow the competition of glucose with the ligand on the glucose-binding protein embedded in the device.
- the displacement of the ligand is preferably detected in the method of the invention by measuring the increase in intensity of fluorescence emanating from a donor dye as described elsewhere herein.
- the increase results from the displacement of the ligand from the glucose-binding protein because the fluorescence intensity of the donor dye is complexed with the glucose-binding protein. It is understood, however, that other detection techniques for releasing the ligand can also be used.
- the invention also relates to the device according to the invention described above for using the diagnosis of disturbed glucose metabolism in a subject.
- the impaired glucose metabolism is caused by diabetes mellitus or the metabolic syndrome.
- the device In the case of in vivo use of the device according to the invention, it is introduced into the body. It should be noted that the measurement of the glucose level, which is also the basis for the diagnosis, requires that the device comes into contact with a body fluid containing glucose, wherein the concentration of glucose in the fluid representative of the glucose level to be determined is. Suitable body fluids are listed elsewhere in the description. Particularly preferably, the body fluid is tissue liquid. The introduced into the body device then generates a signal that can be evaluated for the diagnosis.
- the device according to the invention is preferably introduced at locations in the body which enable an optical measurement of the signal generated by the device. Suitable are sites with either a low tissue thickness between device and body surface or with transparent tissues that can be well penetrated by the generated signal. Most preferably, the device is administered under the skin (subcutaneously) or in the eye, e.g. subconjunctival, placed. Corresponding methods for implanting the device are known in the art.
- the signal generated by the device according to the invention can also be transmitted out of the body by means of a suitable transmission medium.
- a signal-conducting material can be used as a flexible cable, e.g. a fiber optic cable.
- the transmission of the signal may also be contactless, e.g. as infrared, radio or radio signal.
- the signal generated by the device according to the invention must first be read out by a detector, which must also be mounted in the device or at least in close proximity, and converted into an electromagnetic signal, for example a radio signal. This electromagnetic signal can then be received and evaluated by a receiver located outside the body.
- the present invention relates to the device of the invention as described above for use in determining the need for a therapeutic intervention in a subject with impaired glucose metabolism.
- Appropriate therapeutic measures include those used to treat diabetes mellitus or the metabolic syndrome.
- this also includes the implementation of other therapeutic measures that can be decided on the basis of the detected disturbed glucose metabolism, such as therapeutic interventions, eg gastric bypass surgery, or lifestyle changes, such as the implementation special diets.
- a further device for example a device which controls the delivery of a drug.
- the device of the present invention may be coupled to a device for delivering insulin. The delivery of insulin may then be controlled based on the need determined by the device of the invention.
- a change in the glucose level in a sample determined with the device according to the invention is hereby translated by, for example, a data processing unit in the delivery device into an instruction which determines the need for insulin delivery. This instruction then mediates the delivery of insulin into the blood for as long or in an amount as needed.
- the uses of the device according to the invention described above thus allow an efficient ex vivo and in vivo diagnosis of blood sugar levels and thus the early detection of diseases that accompany impaired glucose metabolism, or by the use in the context of clinical monitoring and the management of such diseases.
- the devices are also suitable for making therapy decisions based on the diagnostic results determined.
- the alginate solution is required via a pump in a two-fluid nozzle. At the same time, compressed air is applied to the second inlet of the nozzle so that the alginate solution is atomized into fine droplets. The droplets are carried by the air flow into a bath with the calcium chloride solution where they gel and sink to the bottom. The gelled balls are then collected.
- Alginate beads are incubated for loading sequentially in a dye-labeled concanavalin A solution and a dye-labeled dextran solution. Then the loaded balls are centrifuged off and the supernatant solution is decanted off. The loaded beads are then optionally subsequently incubated overnight in a solution of a second polymer (e.g., PVA, PEG-based) and optionally separated by centrifugation. Subsequently, the balls are mixed in an aqueous solution of a photochemically crosslinkable polymer. This mixture is then cross-linked with UV light to prevent leaching of the sensor components from the alginate beads.
- a second polymer e.g., PVA, PEG-based
- the amounts depend on the concentration of the analyte to be measured and the degree of labeling is selected as a function of the desired intensity of the fluorescence signal.
- the photochemically crosslinkable polymer for example, Nelfilcon polymer, an acrylamide group-modified polyvinyl alcohol, can be used.
- photochemical crosslinking 0.1% Irgacure 2959 is added. The final solution is dosed into suitable molds and cured by UV light.
- Dye-labeled concanavalin A solution and dye-labeled dextran solution are added sequentially to a water-based prepolymer mixture and stirred for 3 hours. The amounts depend on the concentration of the analyte to be measured and the degree of labeling is selected as a function of the desired intensity of the fluorescence signal.
- the photochemically crosslinkable prepolymer for example, Nelfilcon polymer, an acrylamide group-modified polyvinyl alcohol, can be used.
- photochemical crosslinking 0.1% Irgacure 2959 is added. The final solution is dosed into suitable molds and cured by UV light.
- the fluorescence spectrum of the sensors is determined at different glucose concentrations.
- the change in the fluorescence intensities of the donor with increasing glucose content serves as a measure of the quality of the glucose sensor.
- Glucose concentrations between 50 and 500 mg / dL must be measured, the glucose activity is calculated as follows:
- GA (intensity 5 oomg / dL - intensity 5 o mg / dL) / intensity 50m g / dL
- ConA solution and dextran solution are diluted in buffer solution and stirred for several hours.
- the fluorescence spectrum of the solution is determined at various glucose concentrations.
- Example 3 Determination of the Influence of the Matrix
- the glucose-binding protein and the ligand are incorporated in a hydrogel matrix which has some interaction with the glucose-binding protein.
- the hydrogel matrix is selected so that, on the one hand, the interaction of glucose-binding protein and hydrogel matrix is greater than that between glucose-binding protein and aqueous solution (enrichment of the sensor components).
- the interaction between the glucose-binding protein and the analyte (glucose) must not be affected or at least significantly affected by the interaction between glucose-binding protein and hydrogel matrix.
- hydrogel matrix With a suitable selection of the hydrogel matrix is achieved by the interaction between Glucose-binding protein and hydrogel matrix accumulation of the glucose-binding protein in the matrix far beyond the solubility limit of the glucose-binding protein in aqueous solution.
- concentrations up to 10 times higher than in free solution can be achieved.
- the solubility problems become even more extreme after the addition of the ligand (eg dextran), since the glucose-binding protein-ligand complex has lower solubility due to its size and often due to multivalences.
- ConA concentrations of more than 50 mg / g can be set in a suitable hydrogel matrix at the same mass ratio become.
- the hydrogel matrix can increase the applicable concentration range of the glucose-binding protein, eg ConA, by a factor of 100.
- the glucose activity (GA) achieved at this concentration is set here to 1 for the determination of the relative glucose activity (rel GA).
- the same response is obtained in the enriching matrix as in solution (rel GA-0.83).
- significantly higher ConA concentrations can be set in the accumulating matrix.
- a ConA concentration of 10 mg / g a 2.6-fold increase in glucose activity is obtained in the enriched hydrogel matrix (see Table 1).
- Example 5 Determination of the influence of the degree of labeling Receptor Due to the decreasing autofluorescence of the tissue with higher wavelengths, long-wave fluorescent dyes must be used for in vivo applications. These fluorescent dyes are typically quite non-polar due to the larger conjugated systems. If the glucose-binding protein is labeled with such dyes, its solubility is lowered, so that due to the precipitation often no high label grade are possible. In order to increase the solubility of the glucose-binding protein, it is advantageous to functionalize them, for example, with polyethylene glycol. This increases the solubility of the glucose-binding protein, which allows higher levels of labeling in the synthesis.
- Example 6 Determination of the influence of the fluorescent dyes
- the structure of the fluorescent dyes which are bound to glucose-binding protein or ligand also has an influence on the glucose activity.
- a combination of a rhodamine and an oxazine color substance has proved particularly suitable.
- a rhodamine donor eg ATTO590 or ATTO610-dextran, ROX-dextran
- an oxazine acceptor eg ATT0655 or ATTO680-ConA, Evoblue30-ConA
- the glucose activity is also dependent on the nature of the hydrogel matix.
- the hydrogel matrix may consist of a polymer or of a mixture of several polymers.
- a hydrogel matrix of alginate or a mixture of an alginate and polyvinyl alcohol hydrogel has been found to be advantageous.
- the alginate hydrogel through its interaction with the ConA and the dextran, allows the enrichment of the sensor components in high concentrations.
- the prepolymer of the second hydrogel (eg PVA) can penetrate into the alginate spheres and, after crosslinking, form an interpenetrating network with the alginate.
- the mesh size of the Network affects the mobility of the molecules of the glucose-binding protein and the ligand and thus also the glucose activity.
- the mesh size can be influenced by the type, the molecular weight, the solids content of the polymer and the proportion of crosslinker groups, which determines the number of nodes.
- a lower content of crosslinker groups leads to a higher glucose activity.
- the activity can even be increased by 1.5 times.
- a 4.2-fold improvement in glucose activity can likewise be achieved without an increase in the degree of labeling of the ConA (see Table 7).
- the solids content of the network also has an influence on the glucose activity. The higher the solids content, the lower the glucose activity (see Table 8).
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Immunology (AREA)
- Engineering & Computer Science (AREA)
- Hematology (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Urology & Nephrology (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- Cell Biology (AREA)
- Food Science & Technology (AREA)
- Medicinal Chemistry (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Microbiology (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Diabetes (AREA)
- Investigating Or Analysing Biological Materials (AREA)
- Investigating Or Analysing Materials By The Use Of Chemical Reactions (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013543701A JP5881729B2 (ja) | 2010-12-17 | 2011-12-13 | 高感度を有する競合的バイオセンサー |
| US13/993,556 US10429380B2 (en) | 2010-12-17 | 2011-12-13 | Device comprising a hydrogel having a glucose-binding protein and a ligand of the glucose-binding protein incorporated therein |
| EP11793827.4A EP2652500B1 (de) | 2010-12-17 | 2011-12-13 | Kompetitiver biosensor mit erhöhter sensitivität |
| CA2820841A CA2820841C (en) | 2010-12-17 | 2011-12-13 | Competitive biosensor having elevated sensitivity |
| CN201180065209.0A CN103328980B (zh) | 2010-12-17 | 2011-12-13 | 具有提高的敏感性的竞争性生物传感器 |
| ES11793827.4T ES2580207T3 (es) | 2010-12-17 | 2011-12-13 | Biosensor competitivo de elevada sensibilidad |
| AU2011344290A AU2011344290B2 (en) | 2010-12-17 | 2011-12-13 | Competitive biosensor having elevated sensitivity |
| HK14102880.3A HK1189940A1 (zh) | 2010-12-17 | 2014-03-24 | 具有提高的敏感性的競爭性生物傳感器 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP10195667 | 2010-12-17 | ||
| EP10195667.0 | 2010-12-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012080218A1 true WO2012080218A1 (de) | 2012-06-21 |
Family
ID=43760023
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2011/072563 WO2012080218A1 (de) | 2010-12-17 | 2011-12-13 | Kompetitiver biosensor mit erhöhter sensitivität |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US10429380B2 (de) |
| EP (1) | EP2652500B1 (de) |
| JP (1) | JP5881729B2 (de) |
| CN (1) | CN103328980B (de) |
| AU (1) | AU2011344290B2 (de) |
| CA (1) | CA2820841C (de) |
| ES (1) | ES2580207T3 (de) |
| HK (1) | HK1189940A1 (de) |
| WO (1) | WO2012080218A1 (de) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018046129A1 (de) * | 2016-09-12 | 2018-03-15 | Baden-Württemberg Stiftung Ggmbh | Lektin zur reversiblen immobilisierung von zellen |
| CN109627464A (zh) * | 2018-05-30 | 2019-04-16 | 齐鲁工业大学 | 一种荧光探针聚合物水凝胶及其制备方法 |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9517023B2 (en) | 2009-06-01 | 2016-12-13 | Profusa, Inc. | Method and system for directing a localized biological response to an implant |
| US8741591B2 (en) | 2009-10-09 | 2014-06-03 | The Research Foundation For The State University Of New York | pH-insensitive glucose indicator protein |
| US10010272B2 (en) | 2010-05-27 | 2018-07-03 | Profusa, Inc. | Tissue-integrating electronic apparatus |
| CN103260501B (zh) | 2010-10-06 | 2015-09-02 | 普罗弗萨股份有限公司 | 组织整合性传感器 |
| CN108013881B (zh) | 2013-03-14 | 2021-06-15 | 普罗菲尤萨股份有限公司 | 用于校正光学信号的方法和装置 |
| EP3777656A1 (de) | 2013-06-06 | 2021-02-17 | Profusa, Inc. | Vorrichtung und verfahren zur detektion optischer signale aus implantierten sensoren |
| US9874554B1 (en) | 2014-07-16 | 2018-01-23 | Verily Life Sciences Llc | Aptamer-based in vivo diagnostic system |
| US11331018B2 (en) | 2016-12-22 | 2022-05-17 | Profusa, Inc. | System and single-channel biosensor for and method of determining analyte value |
| CN107422004B (zh) * | 2017-06-26 | 2019-08-02 | 清华大学 | 葡萄糖的检测方法及其检测装置 |
| CN107422016A (zh) * | 2017-07-21 | 2017-12-01 | 上海第二工业大学 | Pva/peg复合水凝胶外膜结构的螺旋形可植入式微创葡萄糖传感器的制备方法 |
| CN107422009B (zh) * | 2017-08-03 | 2019-06-14 | 湖北师范大学 | 一种方便检测葡萄糖的非酶电化学生物传感方法 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001013783A1 (en) | 1999-08-26 | 2001-03-01 | Novartis Ag | Ocular analyte sensor |
| WO2002087429A1 (en) | 2001-04-27 | 2002-11-07 | Novartis Ag | Apparatus for measuring blood glucose concentrations |
| US6485703B1 (en) | 1998-07-31 | 2002-11-26 | The Texas A&M University System | Compositions and methods for analyte detection |
| US20070105176A1 (en) | 2005-09-28 | 2007-05-10 | Ibey Bennett L | Method and apparatus for glucose monitoring |
| US20070122829A1 (en) | 2003-01-07 | 2007-05-31 | Ralph Ballerstadt | Device and method for measuring analytes |
| DE102007024642A1 (de) * | 2007-05-24 | 2008-11-27 | Eyesense Ag | Hydrogel-Implantat für Sensorik von Metaboliten am Auge |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU1623300A (en) * | 1998-11-13 | 2000-06-05 | Sensor Technologies, Inc. | Monodisperse preparations useful with implanted devices |
| CA2328614C (en) * | 1999-02-12 | 2012-06-26 | Biostream, Inc. | Matrices for drug delivery and methods for making and using the same |
| US6627177B2 (en) * | 2000-12-05 | 2003-09-30 | The Regents Of The University Of California | Polyhydroxyl-substituted organic molecule sensing optical in vivo method utilizing a boronic acid adduct and the device thereof |
| EP1281966A3 (de) * | 2001-07-30 | 2003-06-18 | Fuji Photo Film Co., Ltd. | Verfahren und Vorrichtung zur Durchführung einer Rezeptor-Ligand-Reaktion |
| US20050239155A1 (en) * | 2002-01-04 | 2005-10-27 | Javier Alarcon | Entrapped binding protein as biosensors |
| WO2003106589A1 (en) * | 2002-06-13 | 2003-12-24 | Lyotropic Therapeutics, Inc. | A nanoporous particle with a retained target |
| US20090297493A1 (en) * | 2002-06-13 | 2009-12-03 | David Anderson | nanoporous particle with a retained target |
| US20040234962A1 (en) * | 2003-05-02 | 2004-11-25 | Javier Alarcon | Multicoated or multilayer entrapment matrix for protein biosensor |
| WO2005015237A1 (en) * | 2003-08-07 | 2005-02-17 | Novartis Ag | Ophthalmic sensor |
| US20050095174A1 (en) | 2003-10-31 | 2005-05-05 | Wolf David E. | Semipermeable sensors for detecting analyte |
| GB0426823D0 (en) * | 2004-12-07 | 2005-01-12 | Precisense As | Sensor for detection of glucose |
| DE502005005099D1 (de) * | 2005-03-23 | 2008-10-02 | Roche Diagnostics Gmbh | Verfahren zur Bestimmung der Glucosekonzentration durch Fluoreszenzpolarisation |
| DK2989975T3 (en) | 2007-02-06 | 2018-09-24 | Medtronic Minimed Inc | OPTICAL SYSTEMS AND PROCEDURES FOR RATIOMETRIC MEASUREMENT OF BLOOD GLUCOSE CONCENTRATION |
| WO2008124158A1 (en) * | 2007-04-06 | 2008-10-16 | West John R | Method and apparatus for forming and mounting a photovoltaic array |
| CA2686065A1 (en) | 2007-05-10 | 2008-11-20 | Glumetrics, Inc. | Equilibrium non-consuming fluorescence sensor for real time intravascular glucose measurement |
| JP6094762B2 (ja) * | 2010-09-14 | 2017-03-15 | ウィトリシティ コーポレーション | 無線エネルギー分配システム |
| US9244064B2 (en) * | 2010-12-17 | 2016-01-26 | Eyesense Ag | Use of hydrogels for biosensors having elevated sensitivity |
-
2011
- 2011-12-13 AU AU2011344290A patent/AU2011344290B2/en active Active
- 2011-12-13 EP EP11793827.4A patent/EP2652500B1/de active Active
- 2011-12-13 JP JP2013543701A patent/JP5881729B2/ja active Active
- 2011-12-13 US US13/993,556 patent/US10429380B2/en active Active
- 2011-12-13 ES ES11793827.4T patent/ES2580207T3/es active Active
- 2011-12-13 CA CA2820841A patent/CA2820841C/en active Active
- 2011-12-13 CN CN201180065209.0A patent/CN103328980B/zh active Active
- 2011-12-13 WO PCT/EP2011/072563 patent/WO2012080218A1/de active Application Filing
-
2014
- 2014-03-24 HK HK14102880.3A patent/HK1189940A1/zh unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6485703B1 (en) | 1998-07-31 | 2002-11-26 | The Texas A&M University System | Compositions and methods for analyte detection |
| WO2001013783A1 (en) | 1999-08-26 | 2001-03-01 | Novartis Ag | Ocular analyte sensor |
| WO2002087429A1 (en) | 2001-04-27 | 2002-11-07 | Novartis Ag | Apparatus for measuring blood glucose concentrations |
| US20070122829A1 (en) | 2003-01-07 | 2007-05-31 | Ralph Ballerstadt | Device and method for measuring analytes |
| US20070105176A1 (en) | 2005-09-28 | 2007-05-10 | Ibey Bennett L | Method and apparatus for glucose monitoring |
| DE102007024642A1 (de) * | 2007-05-24 | 2008-11-27 | Eyesense Ag | Hydrogel-Implantat für Sensorik von Metaboliten am Auge |
Non-Patent Citations (6)
| Title |
|---|
| BIRCH, SPECTROCHIMICA ACTA, vol. 57, 2001, pages 2245 - 2254 |
| CZIMEROVA ET AL: "Fluorescence resonance energy transfer between two cationic laser dyes in presence of the series of reduced-charge montmorillonites: Effect of the layer charge", JOURNAL OF COLLOID AND INTERFACE SCIENCE, ACADEMIC PRESS, NEW YORK, NY, US, vol. 320, no. 1, 28 January 2008 (2008-01-28), pages 140 - 151, XP022517980, ISSN: 0021-9797, DOI: DOI:10.1016/J.JCIS.2007.10.055 * |
| ROUNDS, J. FLUOREC., vol. 17, 2007, pages 57 - 63 |
| ROUNDS, J. FLUORESC., vol. 17, 2007, pages 57 - 63 |
| RUSSELL, ANAL CHEM, vol. 71, 1999, pages 3126 - 3132 |
| YAMAUCHI, FEBS LETTERS, vol. 260, no. 1, 1990, pages 127 - 130 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018046129A1 (de) * | 2016-09-12 | 2018-03-15 | Baden-Württemberg Stiftung Ggmbh | Lektin zur reversiblen immobilisierung von zellen |
| CN109627464A (zh) * | 2018-05-30 | 2019-04-16 | 齐鲁工业大学 | 一种荧光探针聚合物水凝胶及其制备方法 |
| CN109627464B (zh) * | 2018-05-30 | 2021-07-06 | 齐鲁工业大学 | 一种荧光探针聚合物水凝胶及其制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2013545994A (ja) | 2013-12-26 |
| EP2652500A1 (de) | 2013-10-23 |
| HK1189940A1 (zh) | 2014-06-20 |
| US10429380B2 (en) | 2019-10-01 |
| AU2011344290A1 (en) | 2013-07-04 |
| AU2011344290B2 (en) | 2016-02-25 |
| EP2652500B1 (de) | 2016-05-11 |
| CA2820841A1 (en) | 2012-06-21 |
| CA2820841C (en) | 2018-10-23 |
| CN103328980B (zh) | 2016-01-20 |
| JP5881729B2 (ja) | 2016-03-09 |
| US20130337468A1 (en) | 2013-12-19 |
| ES2580207T3 (es) | 2016-08-22 |
| CN103328980A (zh) | 2013-09-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2652500B1 (de) | Kompetitiver biosensor mit erhöhter sensitivität | |
| EP2652510B1 (de) | Verwendung von hydrogelen für biosensoren mit erhöhter sensitivität | |
| DE60035174T2 (de) | Invasives verfahren zur in vivo analytvermessung | |
| WO2008142158A2 (de) | Hydrogel-implantat für sensorik von metaboliten in körpergewebe | |
| DE102012201892A1 (de) | Bestimmung des Blutzuckerspiegels eines Patienten unter Verwendung eines implantierbaren Sensors und eines elektrischen Funktionspflasters | |
| Bucciarelli et al. | Tidy dataset of the experimental design of the optimization of the alkali degumming process of Bombyx mori silk | |
| EP1290035B1 (de) | Fluoresceinisothiocyanat-(fitc)-sinistrin, seine herstellung und verwendung | |
| EP2942623B1 (de) | Detektionsvorrichtung zur anreicherung von probenmaterial | |
| EP1705485A1 (de) | Verfahren zur Bestimmung der Glucosekonzentration durch Fluoreszenzpolarisation | |
| DE10311623B4 (de) | Membran-Osmometer und Verfahren zur selektiven Bestimmung spezifischer Analyte | |
| EP3576723A1 (de) | Selektives freisetzungssystem für tumortherapeutika und tumordiagnostika sowie biosensor für tumorgewebe | |
| JP7133832B2 (ja) | 蛍光性官能基を有する分子インプリントナノ粒子 | |
| Pashchenko et al. | Smart nanofibres for specific and ultrasensitive nanobiosensors and drug delivery systems | |
| Ostrovidov et al. | Molecularly Imprinted Polymer-Based Sensors for the Detection of Skeletal-and Cardiac-Muscle-Related Analytes | |
| DE102016113166B4 (de) | Verfahren zum Nachweis eines Biofilms und Mittel zum Nachweis eines Biofilms | |
| Ghazi et al. | Synthesis and Characterization of New Boronic Acid-Containing Hydrogels as Glucose-Sensitive Vehicle for Controlled Insulin Release |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11793827 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2820841 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2013543701 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2011344290 Country of ref document: AU Date of ref document: 20111213 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011793827 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13993556 Country of ref document: US |