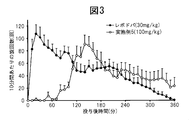
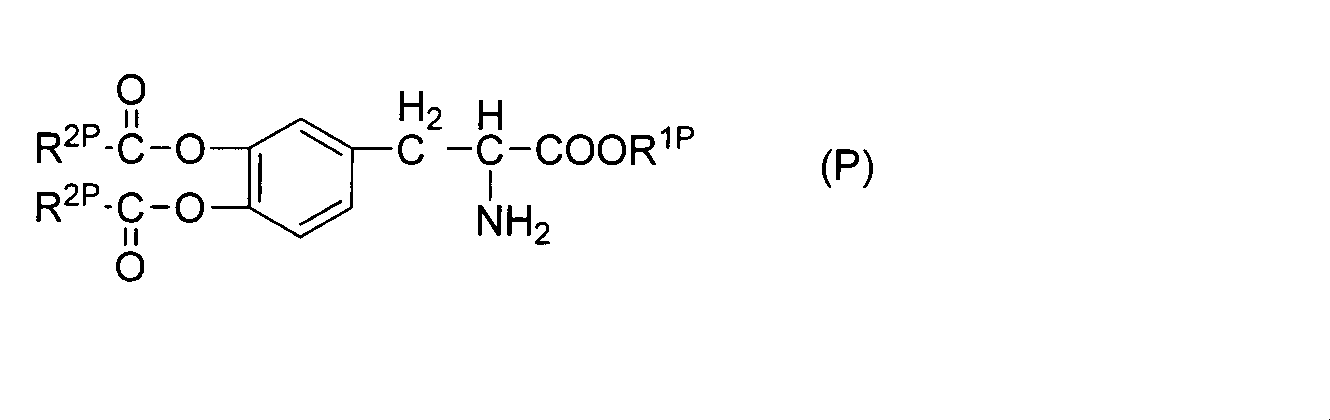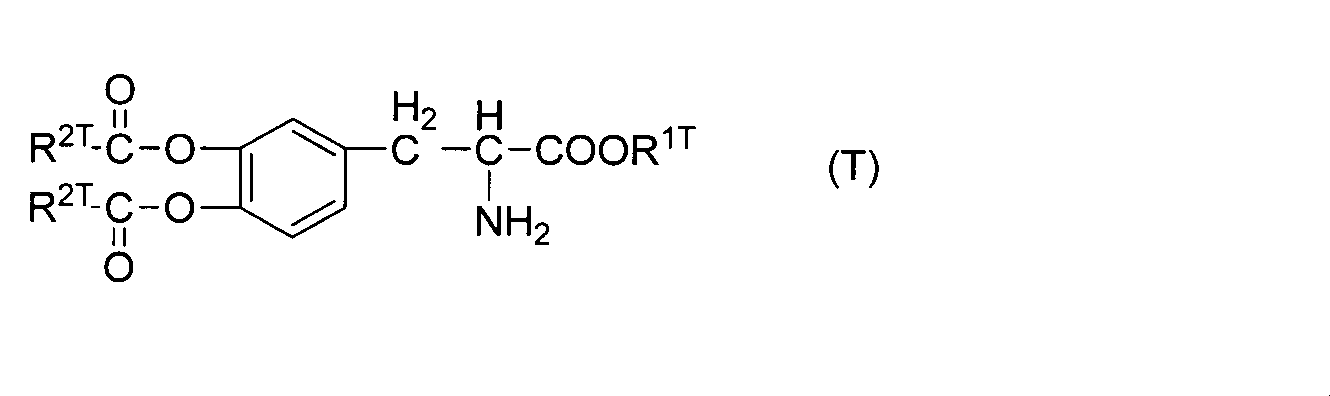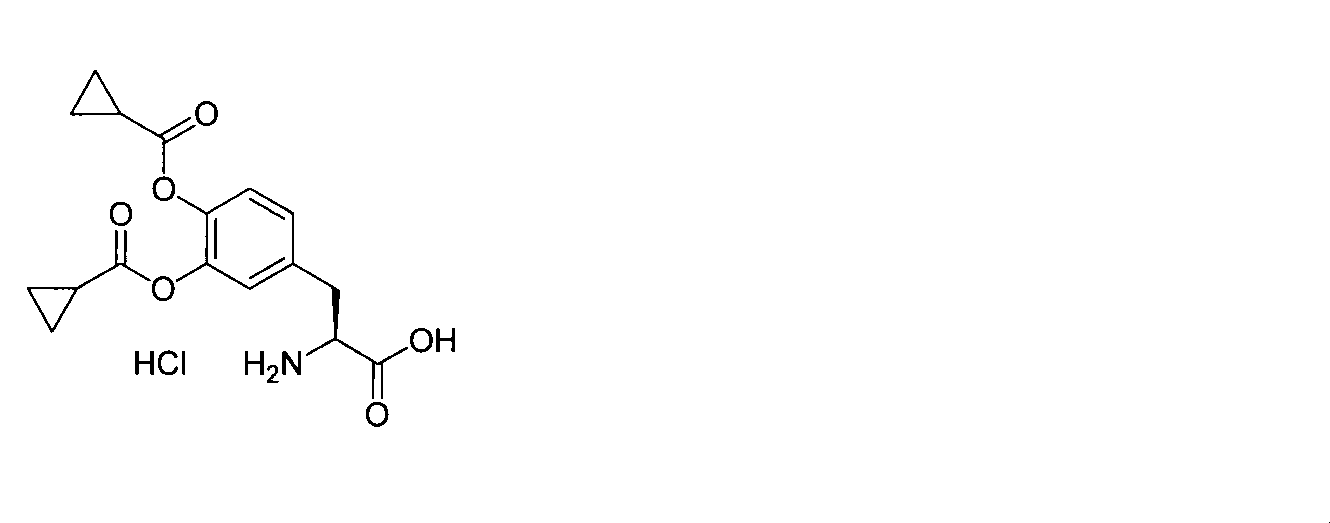WO2012074069A1 - 新規化合物及びその医薬用途 - Google Patents
新規化合物及びその医薬用途 Download PDFInfo
- Publication number
- WO2012074069A1 WO2012074069A1 PCT/JP2011/077834 JP2011077834W WO2012074069A1 WO 2012074069 A1 WO2012074069 A1 WO 2012074069A1 JP 2011077834 W JP2011077834 W JP 2011077834W WO 2012074069 A1 WO2012074069 A1 WO 2012074069A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino
- compound
- oxy
- bis
- phenyl
- Prior art date
Links
- 150000001875 compounds Chemical class 0.000 title claims description 354
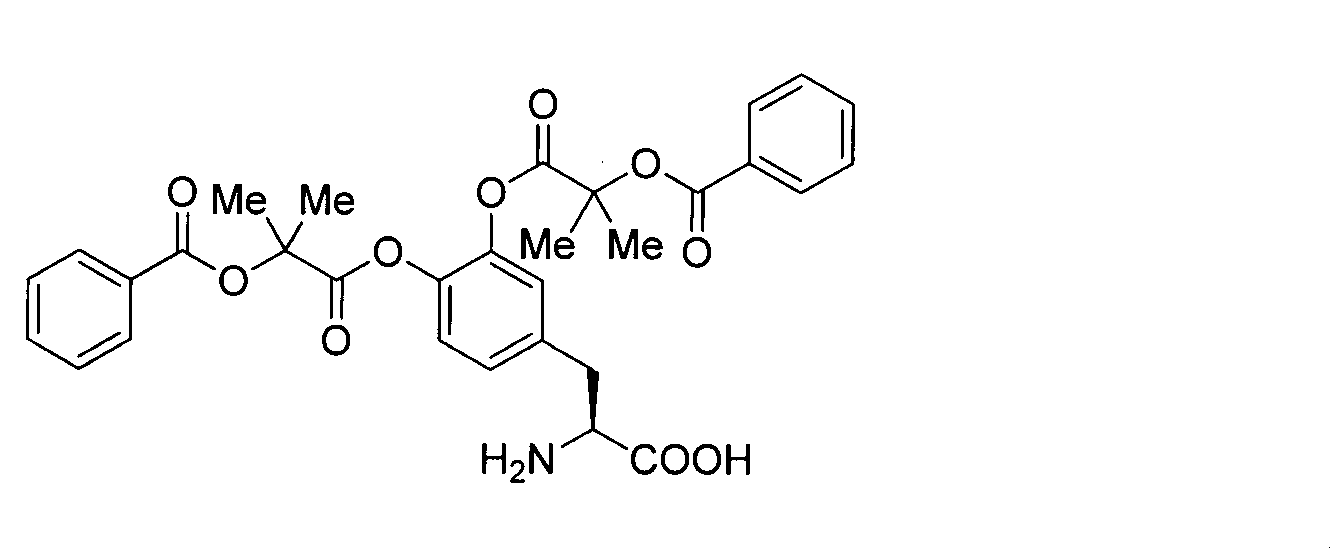
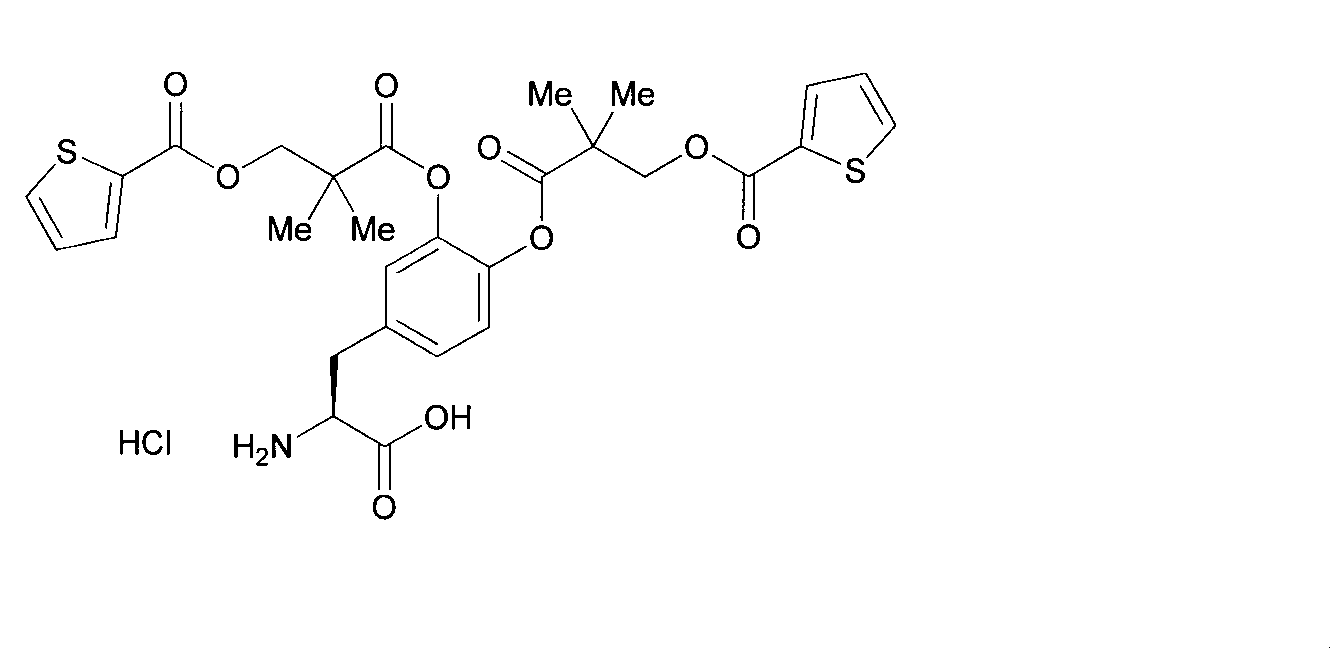
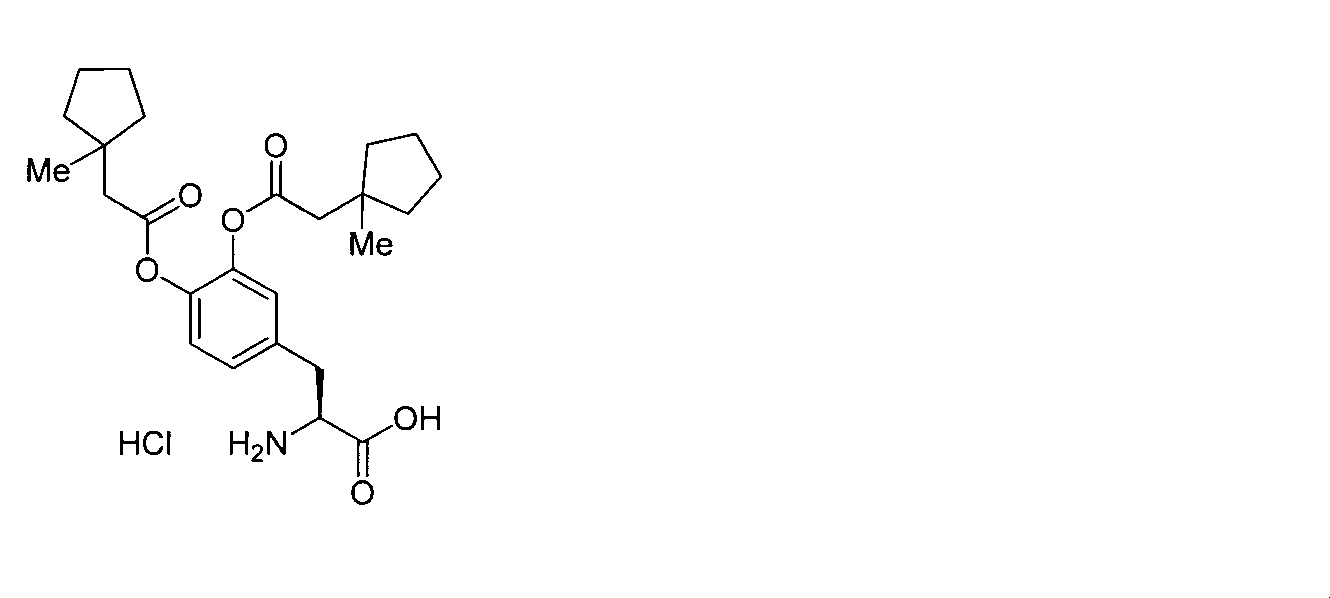
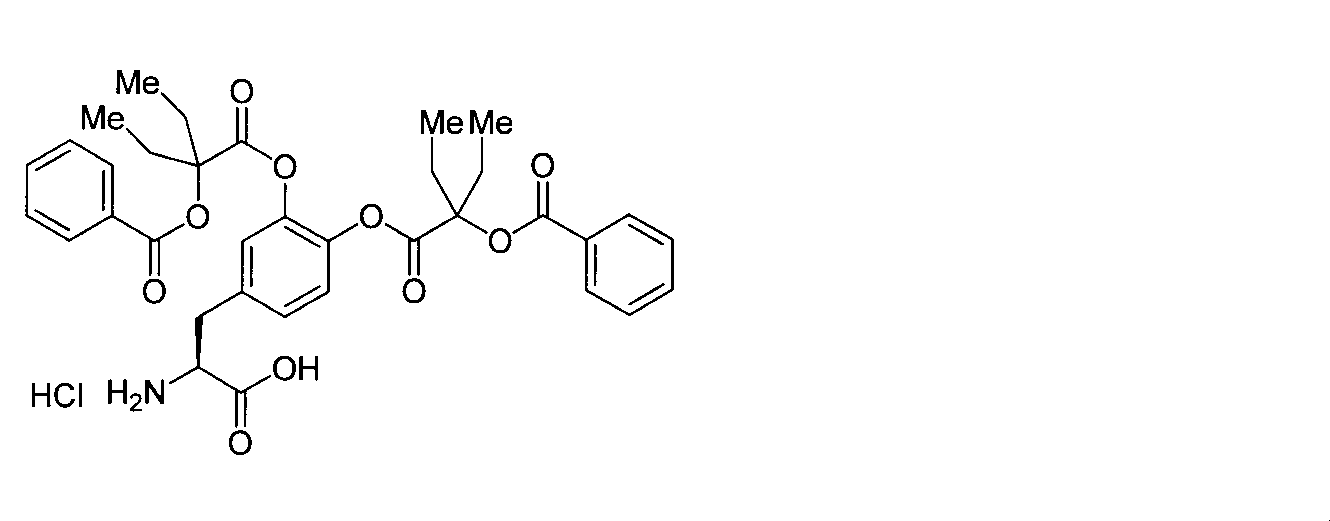
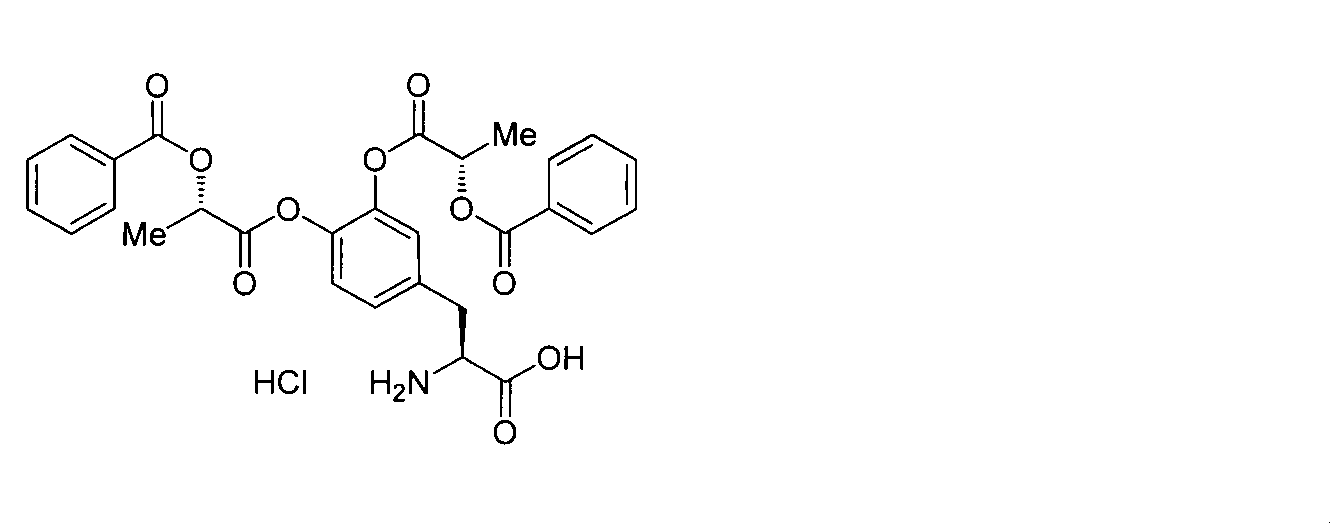
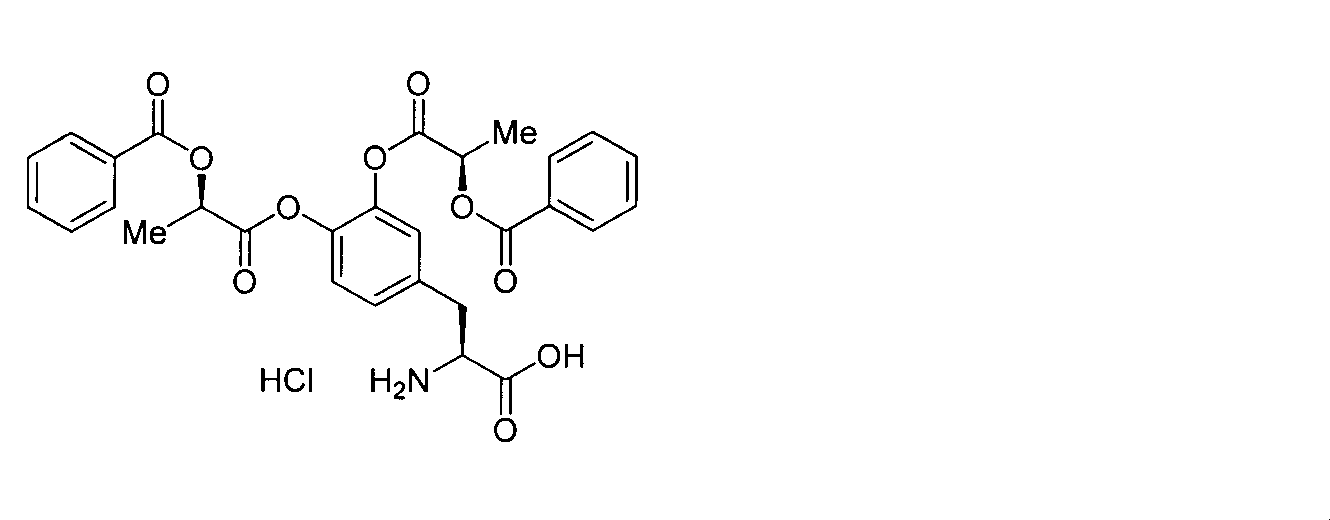
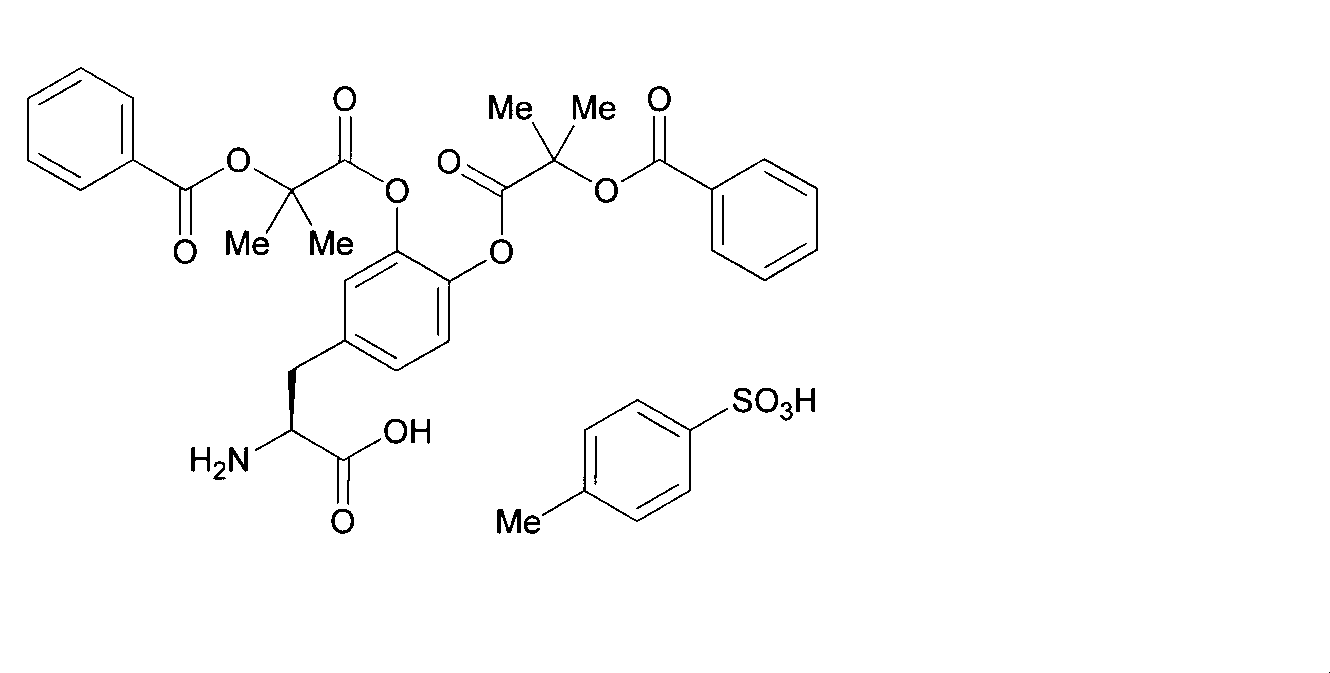
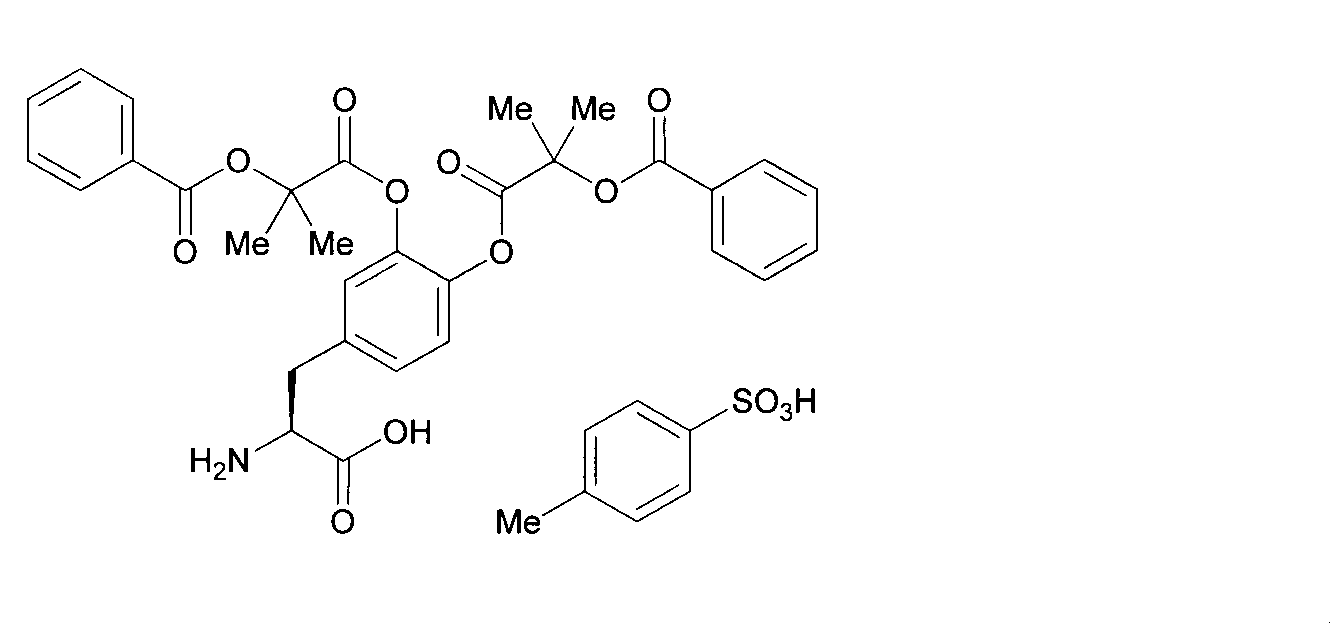
- OJIGBNLGFYOFHY-QFIPXVFZSA-N (2s)-2-amino-3-[3,4-bis[(2-benzoyloxy-2-methylpropanoyl)oxy]phenyl]propanoic acid Chemical compound C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)C(C)(C)OC(=O)C=2C=CC=CC=2)C=1OC(=O)C(C)(C)OC(=O)C1=CC=CC=C1 OJIGBNLGFYOFHY-QFIPXVFZSA-N 0.000 claims abstract description 67
- 208000018737 Parkinson disease Diseases 0.000 claims abstract description 36
- 150000003839 salts Chemical class 0.000 claims abstract description 31
- 239000012453 solvate Substances 0.000 claims abstract description 31
- 206010034010 Parkinsonism Diseases 0.000 claims abstract description 28
- 230000003449 preventive effect Effects 0.000 claims abstract description 6
- 239000013078 crystal Substances 0.000 claims description 150
- -1 (benzoyloxy) -2-methylpropanoyl Chemical group 0.000 claims description 77
- 238000000634 powder X-ray diffraction Methods 0.000 claims description 55
- 238000001228 spectrum Methods 0.000 claims description 55
- 238000000113 differential scanning calorimetry Methods 0.000 claims description 47
- 239000003814 drug Substances 0.000 claims description 47
- LALZWIYGMTXISZ-FTBISJDPSA-N (2s)-2-amino-3-[3,4-bis[(2-benzoyloxy-2-methylpropanoyl)oxy]phenyl]propanoic acid;4-methylbenzenesulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)C(C)(C)OC(=O)C=2C=CC=CC=2)C=1OC(=O)C(C)(C)OC(=O)C1=CC=CC=C1 LALZWIYGMTXISZ-FTBISJDPSA-N 0.000 claims description 38
- 238000002844 melting Methods 0.000 claims description 31
- 230000008018 melting Effects 0.000 claims description 31
- 239000003954 decarboxylase inhibitor Substances 0.000 claims description 21
- 229940123736 Decarboxylase inhibitor Drugs 0.000 claims description 20
- HMSXZFWUONAIFN-FTBISJDPSA-N (2s)-2-amino-3-[3,4-bis[(2-benzoyloxy-2-methylpropanoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)C(C)(C)OC(=O)C=2C=CC=CC=2)C=1OC(=O)C(C)(C)OC(=O)C1=CC=CC=C1 HMSXZFWUONAIFN-FTBISJDPSA-N 0.000 claims description 19
- 229960004205 carbidopa Drugs 0.000 claims description 13
- 102000003823 Aromatic-L-amino-acid decarboxylases Human genes 0.000 claims description 11
- 108090000121 Aromatic-L-amino-acid decarboxylases Proteins 0.000 claims description 11
- BNQDCRGUHNALGH-UHFFFAOYSA-N benserazide Chemical compound OCC(N)C(=O)NNCC1=CC=C(O)C(O)=C1O BNQDCRGUHNALGH-UHFFFAOYSA-N 0.000 claims description 9
- 239000003543 catechol methyltransferase inhibitor Substances 0.000 claims description 8
- 229940000425 combination drug Drugs 0.000 claims description 8
- 229940099362 Catechol O methyltransferase inhibitor Drugs 0.000 claims description 7
- 229940124597 therapeutic agent Drugs 0.000 claims description 7
- 125000003118 aryl group Chemical group 0.000 claims description 6
- JRURYQJSLYLRLN-BJMVGYQFSA-N entacapone Chemical group CCN(CC)C(=O)C(\C#N)=C\C1=CC(O)=C(O)C([N+]([O-])=O)=C1 JRURYQJSLYLRLN-BJMVGYQFSA-N 0.000 claims description 6
- 229960003337 entacapone Drugs 0.000 claims description 6
- MRFOLGFFTUGAEB-UHFFFAOYSA-N 1-(3,4-dihydroxy-5-nitrophenyl)-2-phenylethanone Chemical compound [O-][N+](=O)C1=C(O)C(O)=CC(C(=O)CC=2C=CC=CC=2)=C1 MRFOLGFFTUGAEB-UHFFFAOYSA-N 0.000 claims description 4
- MEYPAYJYAZKERR-JLHYYAGUSA-N CGP 28014 Chemical compound CCCN(CCC)\C=N\C1=CC=CC(=O)N1 MEYPAYJYAZKERR-JLHYYAGUSA-N 0.000 claims description 4
- UPMRZALMHVUCIN-UHFFFAOYSA-N Nitecapone Chemical compound CC(=O)C(C(C)=O)=CC1=CC(O)=C(O)C([N+]([O-])=O)=C1 UPMRZALMHVUCIN-UHFFFAOYSA-N 0.000 claims description 4
- 229960001335 benserazide hydrochloride Drugs 0.000 claims description 4
- 229950008980 nitecapone Drugs 0.000 claims description 4
- 239000008194 pharmaceutical composition Substances 0.000 claims description 4
- 229960004603 tolcapone Drugs 0.000 claims description 4
- MIQPIUSUKVNLNT-UHFFFAOYSA-N tolcapone Chemical compound C1=CC(C)=CC=C1C(=O)C1=CC(O)=C(O)C([N+]([O-])=O)=C1 MIQPIUSUKVNLNT-UHFFFAOYSA-N 0.000 claims description 4
- 150000001413 amino acids Chemical class 0.000 claims description 3
- USROQQUKEBHOFF-UHFFFAOYSA-N 3-[4-[4-(aminomethyl)cyclohexanecarbonyl]oxyphenyl]propanoic acid;hydron;chloride Chemical compound Cl.C1CC(CN)CCC1C(=O)OC1=CC=C(CCC(O)=O)C=C1 USROQQUKEBHOFF-UHFFFAOYSA-N 0.000 claims description 2
- TZFNLOMSOLWIDK-JTQLQIEISA-N carbidopa (anhydrous) Chemical group NN[C@@](C(O)=O)(C)CC1=CC=C(O)C(O)=C1 TZFNLOMSOLWIDK-JTQLQIEISA-N 0.000 claims 1
- WTDRDQBEARUVNC-LURJTMIESA-N L-DOPA Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C(O)=C1 WTDRDQBEARUVNC-LURJTMIESA-N 0.000 abstract description 176
- WTDRDQBEARUVNC-UHFFFAOYSA-N L-Dopa Natural products OC(=O)C(N)CC1=CC=C(O)C(O)=C1 WTDRDQBEARUVNC-UHFFFAOYSA-N 0.000 abstract description 175
- 229960004502 levodopa Drugs 0.000 abstract description 171
- 239000008280 blood Substances 0.000 abstract description 57
- 210000004369 blood Anatomy 0.000 abstract description 57
- 230000000694 effects Effects 0.000 abstract description 27
- 229940002612 prodrug Drugs 0.000 abstract description 19
- 239000000651 prodrug Substances 0.000 abstract description 19
- 230000033001 locomotion Effects 0.000 abstract description 4
- 239000000825 pharmaceutical preparation Substances 0.000 abstract description 4
- 208000027089 Parkinsonian disease Diseases 0.000 abstract description 3
- 238000011161 development Methods 0.000 abstract description 3
- 229940126585 therapeutic drug Drugs 0.000 abstract description 2
- 230000000004 hemodynamic effect Effects 0.000 abstract 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 231
- 238000000034 method Methods 0.000 description 220
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 126
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 98
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 71
- 239000000243 solution Substances 0.000 description 68
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 58
- 238000006243 chemical reaction Methods 0.000 description 55
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 54
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 52
- 239000002904 solvent Substances 0.000 description 50
- 238000004809 thin layer chromatography Methods 0.000 description 49
- 238000005481 NMR spectroscopy Methods 0.000 description 47
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 46
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 45
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 41
- 230000000704 physical effect Effects 0.000 description 39
- 230000002829 reductive effect Effects 0.000 description 34
- 229940079593 drug Drugs 0.000 description 33
- 239000000203 mixture Substances 0.000 description 33
- 238000000862 absorption spectrum Methods 0.000 description 32
- VYFYYTLLBUKUHU-UHFFFAOYSA-N dopamine Chemical compound NCCC1=CC=C(O)C(O)=C1 VYFYYTLLBUKUHU-UHFFFAOYSA-N 0.000 description 32
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 30
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 29
- 239000003960 organic solvent Substances 0.000 description 28
- 238000004458 analytical method Methods 0.000 description 27
- PDUCEWQLOYAIKX-INIZCTEOSA-N (2s)-2-amino-3-[4-(2-benzoyloxy-2-methylpropanoyl)oxy-3-(2-hydroxy-2-methylpropanoyl)oxyphenyl]propanoic acid Chemical compound CC(C)(O)C(=O)OC1=CC(C[C@H](N)C(O)=O)=CC=C1OC(=O)C(C)(C)OC(=O)C1=CC=CC=C1 PDUCEWQLOYAIKX-INIZCTEOSA-N 0.000 description 26
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 26
- 238000010511 deprotection reaction Methods 0.000 description 26
- 239000012046 mixed solvent Substances 0.000 description 26
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 26
- ZZPDREPBZFYLQJ-AWEZNQCLSA-N (2s)-2-amino-3-[3-(2-benzoyloxy-2-methylpropanoyl)oxy-4-hydroxyphenyl]propanoic acid Chemical compound C=1C(C[C@H](N)C(O)=O)=CC=C(O)C=1OC(=O)C(C)(C)OC(=O)C1=CC=CC=C1 ZZPDREPBZFYLQJ-AWEZNQCLSA-N 0.000 description 25
- 239000012043 crude product Substances 0.000 description 25
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 24
- 238000004519 manufacturing process Methods 0.000 description 23
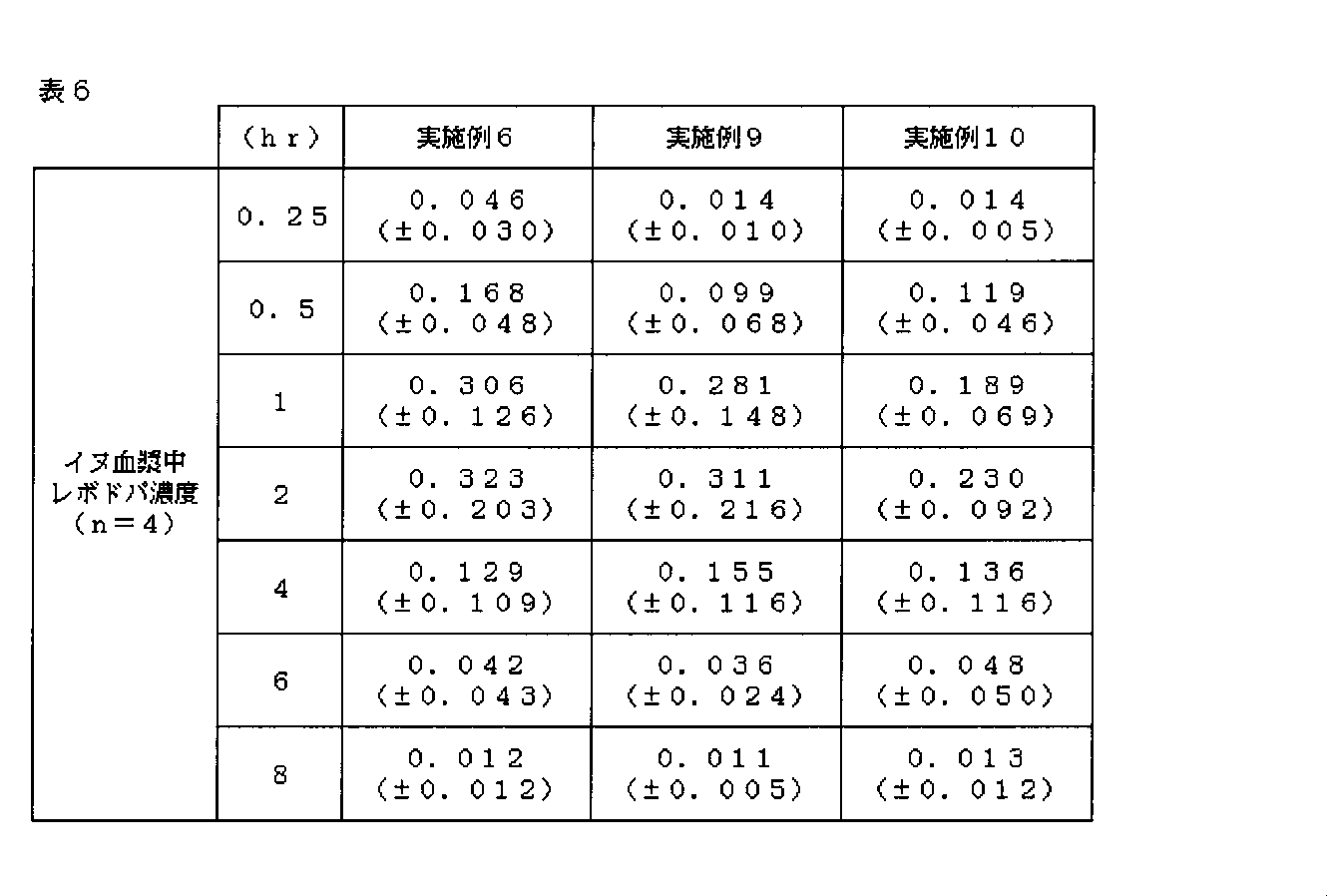
- 230000036470 plasma concentration Effects 0.000 description 23
- 239000007924 injection Substances 0.000 description 22
- 238000002347 injection Methods 0.000 description 22
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 22
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 21
- 239000002253 acid Substances 0.000 description 21
- 239000003795 chemical substances by application Substances 0.000 description 21
- 238000002360 preparation method Methods 0.000 description 21
- 238000012360 testing method Methods 0.000 description 21
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 20
- 241000282472 Canis lupus familiaris Species 0.000 description 19
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 19
- 208000024891 symptom Diseases 0.000 description 19
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 18
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 18
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 18
- 239000002585 base Substances 0.000 description 18
- 238000005259 measurement Methods 0.000 description 17
- 201000010099 disease Diseases 0.000 description 16
- 230000009102 absorption Effects 0.000 description 15
- 238000010521 absorption reaction Methods 0.000 description 15
- 125000000217 alkyl group Chemical group 0.000 description 15
- 229960003638 dopamine Drugs 0.000 description 15
- 150000004820 halides Chemical class 0.000 description 15
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 14
- 238000009472 formulation Methods 0.000 description 13
- 230000002265 prevention Effects 0.000 description 13
- IAVPVBNGDDKIBA-VWLOTQADSA-N (2s)-3-[3,4-bis[(2-benzoyloxy-2-methylpropanoyl)oxy]phenyl]-2-[(2-methylpropan-2-yl)oxycarbonylamino]propanoic acid Chemical compound C=1C=CC=CC=1C(=O)OC(C)(C)C(=O)OC1=CC(C[C@H](NC(=O)OC(C)(C)C)C(O)=O)=CC=C1OC(=O)C(C)(C)OC(=O)C1=CC=CC=C1 IAVPVBNGDDKIBA-VWLOTQADSA-N 0.000 description 12
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 12
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 12
- 208000012661 Dyskinesia Diseases 0.000 description 12
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 12
- QTAOMKOIBXZKND-PPHPATTJSA-N carbidopa Chemical group O.NN[C@@](C(O)=O)(C)CC1=CC=C(O)C(O)=C1 QTAOMKOIBXZKND-PPHPATTJSA-N 0.000 description 12
- 238000010438 heat treatment Methods 0.000 description 12
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 12
- 241000282412 Homo Species 0.000 description 11
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 11
- 241000700159 Rattus Species 0.000 description 11
- 239000013543 active substance Substances 0.000 description 11
- 238000001035 drying Methods 0.000 description 11
- 150000007530 organic bases Chemical class 0.000 description 11
- 230000007704 transition Effects 0.000 description 11
- 150000007529 inorganic bases Chemical class 0.000 description 10
- 239000007788 liquid Substances 0.000 description 10
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 10
- 235000019341 magnesium sulphate Nutrition 0.000 description 10
- 239000000843 powder Substances 0.000 description 10
- 238000003756 stirring Methods 0.000 description 10
- GIPZXQASQLZNAB-INIZCTEOSA-N (2s)-2-amino-3-[3-(2-benzoyloxy-2-methylpropanoyl)oxy-4-(2-hydroxy-2-methylpropanoyl)oxyphenyl]propanoic acid Chemical compound CC(C)(O)C(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)C(C)(C)OC(=O)C1=CC=CC=C1 GIPZXQASQLZNAB-INIZCTEOSA-N 0.000 description 9
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 238000011033 desalting Methods 0.000 description 9
- 239000003755 preservative agent Substances 0.000 description 9
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 9
- 239000007787 solid Substances 0.000 description 9
- CBSQMDGBGLVZPW-JTQLQIEISA-N (2s)-2-amino-3-[3,4-bis[(2-hydroxy-2-methylpropanoyl)oxy]phenyl]propanoic acid Chemical compound CC(C)(O)C(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)C(C)(C)O CBSQMDGBGLVZPW-JTQLQIEISA-N 0.000 description 8
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 8
- 239000012300 argon atmosphere Substances 0.000 description 8
- 125000001231 benzoyloxy group Chemical group C(C1=CC=CC=C1)(=O)O* 0.000 description 8
- 238000009835 boiling Methods 0.000 description 8
- 238000001816 cooling Methods 0.000 description 8
- DIVDFFZHCJEHGG-UHFFFAOYSA-N oxidopamine Chemical compound NCCC1=CC(O)=C(O)C=C1O DIVDFFZHCJEHGG-UHFFFAOYSA-N 0.000 description 8
- 230000002335 preservative effect Effects 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- 239000000725 suspension Substances 0.000 description 8
- 230000001225 therapeutic effect Effects 0.000 description 8
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 8
- AGKSCUASPSBHEM-AWEZNQCLSA-N (2s)-2-amino-3-[4-(2-benzoyloxy-2-methylpropanoyl)oxy-3-hydroxyphenyl]propanoic acid Chemical compound C=1C=C(C[C@H](N)C(O)=O)C=C(O)C=1OC(=O)C(C)(C)OC(=O)C1=CC=CC=C1 AGKSCUASPSBHEM-AWEZNQCLSA-N 0.000 description 7
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 7
- 150000008065 acid anhydrides Chemical class 0.000 description 7
- 235000001014 amino acid Nutrition 0.000 description 7
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 7
- 239000000796 flavoring agent Substances 0.000 description 7
- 235000013355 food flavoring agent Nutrition 0.000 description 7
- 238000002955 isolation Methods 0.000 description 7
- PSHKMPUSSFXUIA-UHFFFAOYSA-N n,n-dimethylpyridin-2-amine Chemical compound CN(C)C1=CC=CC=N1 PSHKMPUSSFXUIA-UHFFFAOYSA-N 0.000 description 7
- 239000012044 organic layer Substances 0.000 description 7
- 238000001953 recrystallisation Methods 0.000 description 7
- 239000002002 slurry Substances 0.000 description 7
- 125000000547 substituted alkyl group Chemical group 0.000 description 7
- SFLSHLFXELFNJZ-QMMMGPOBSA-N (-)-norepinephrine Chemical compound NC[C@H](O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-QMMMGPOBSA-N 0.000 description 6
- 125000004209 (C1-C8) alkyl group Chemical group 0.000 description 6
- PAQZWJGSJMLPMG-UHFFFAOYSA-N 2,4,6-tripropyl-1,3,5,2$l^{5},4$l^{5},6$l^{5}-trioxatriphosphinane 2,4,6-trioxide Chemical compound CCCP1(=O)OP(=O)(CCC)OP(=O)(CCC)O1 PAQZWJGSJMLPMG-UHFFFAOYSA-N 0.000 description 6
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 6
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 6
- 239000003963 antioxidant agent Substances 0.000 description 6
- 230000003078 antioxidant effect Effects 0.000 description 6
- 229910052786 argon Inorganic materials 0.000 description 6
- 210000004556 brain Anatomy 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 230000008859 change Effects 0.000 description 6
- 235000014113 dietary fatty acids Nutrition 0.000 description 6
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 6
- 239000000194 fatty acid Substances 0.000 description 6
- 229930195729 fatty acid Natural products 0.000 description 6
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 6
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 6
- 231100000299 mutagenicity Toxicity 0.000 description 6
- 230000007886 mutagenicity Effects 0.000 description 6
- 231100000150 mutagenicity / genotoxicity testing Toxicity 0.000 description 6
- DUWWHGPELOTTOE-UHFFFAOYSA-N n-(5-chloro-2,4-dimethoxyphenyl)-3-oxobutanamide Chemical compound COC1=CC(OC)=C(NC(=O)CC(C)=O)C=C1Cl DUWWHGPELOTTOE-UHFFFAOYSA-N 0.000 description 6
- 229960002748 norepinephrine Drugs 0.000 description 6
- SFLSHLFXELFNJZ-UHFFFAOYSA-N norepinephrine Natural products NCC(O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-UHFFFAOYSA-N 0.000 description 6
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 6
- 235000019260 propionic acid Nutrition 0.000 description 6
- 238000010898 silica gel chromatography Methods 0.000 description 6
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 6
- 239000003826 tablet Substances 0.000 description 6
- IVTMXOXVAHXCHI-YXLMWLKOSA-N (2s)-2-amino-3-(3,4-dihydroxyphenyl)propanoic acid;(2s)-3-(3,4-dihydroxyphenyl)-2-hydrazinyl-2-methylpropanoic acid Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C(O)=C1.NN[C@@](C(O)=O)(C)CC1=CC=C(O)C(O)=C1 IVTMXOXVAHXCHI-YXLMWLKOSA-N 0.000 description 5
- ICEUWZGFDJYGKA-UHFFFAOYSA-N 4-methylbenzenesulfonic acid;propanoic acid Chemical compound CCC(O)=O.CC1=CC=C(S(O)(=O)=O)C=C1 ICEUWZGFDJYGKA-UHFFFAOYSA-N 0.000 description 5
- 208000010201 Exanthema Diseases 0.000 description 5
- 208000001089 Multiple system atrophy Diseases 0.000 description 5
- 239000002202 Polyethylene glycol Substances 0.000 description 5
- 125000002252 acyl group Chemical group 0.000 description 5
- 229910052782 aluminium Inorganic materials 0.000 description 5
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 5
- 150000001412 amines Chemical class 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 239000012298 atmosphere Substances 0.000 description 5
- 229960000911 benserazide Drugs 0.000 description 5
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 5
- 238000007796 conventional method Methods 0.000 description 5
- 201000005884 exanthem Diseases 0.000 description 5
- 238000001914 filtration Methods 0.000 description 5
- 210000000056 organ Anatomy 0.000 description 5
- 238000007911 parenteral administration Methods 0.000 description 5
- 229920001223 polyethylene glycol Polymers 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 125000006239 protecting group Chemical group 0.000 description 5
- 238000000746 purification Methods 0.000 description 5
- 206010037844 rash Diseases 0.000 description 5
- 238000004088 simulation Methods 0.000 description 5
- 238000004611 spectroscopical analysis Methods 0.000 description 5
- 239000007921 spray Substances 0.000 description 5
- ASOKPJOREAFHNY-UHFFFAOYSA-N 1-Hydroxybenzotriazole Chemical compound C1=CC=C2N(O)N=NC2=C1 ASOKPJOREAFHNY-UHFFFAOYSA-N 0.000 description 4
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Chemical compound CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 4
- PAMIQIKDUOTOBW-UHFFFAOYSA-N 1-methylpiperidine Chemical compound CN1CCCCC1 PAMIQIKDUOTOBW-UHFFFAOYSA-N 0.000 description 4
- YYROPELSRYBVMQ-UHFFFAOYSA-N 4-toluenesulfonyl chloride Chemical compound CC1=CC=C(S(Cl)(=O)=O)C=C1 YYROPELSRYBVMQ-UHFFFAOYSA-N 0.000 description 4
- 241000282465 Canis Species 0.000 description 4
- 102000018208 Cannabinoid Receptor Human genes 0.000 description 4
- 108050007331 Cannabinoid receptor Proteins 0.000 description 4
- 102000013392 Carboxylesterase Human genes 0.000 description 4
- 108010051152 Carboxylesterase Proteins 0.000 description 4
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical compound C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 4
- 108010010803 Gelatin Proteins 0.000 description 4
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 4
- 238000004566 IR spectroscopy Methods 0.000 description 4
- 241001465754 Metazoa Species 0.000 description 4
- 102000010909 Monoamine Oxidase Human genes 0.000 description 4
- 108010062431 Monoamine oxidase Proteins 0.000 description 4
- 206010031127 Orthostatic hypotension Diseases 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 4
- 150000001298 alcohols Chemical class 0.000 description 4
- 125000003545 alkoxy group Chemical group 0.000 description 4
- 125000003277 amino group Chemical group 0.000 description 4
- 210000003169 central nervous system Anatomy 0.000 description 4
- 238000002425 crystallisation Methods 0.000 description 4
- 230000008025 crystallization Effects 0.000 description 4
- LJOODBDWMQKMFB-UHFFFAOYSA-N cyclohexylacetic acid Chemical compound OC(=O)CC1CCCCC1 LJOODBDWMQKMFB-UHFFFAOYSA-N 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 4
- 239000003995 emulsifying agent Substances 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 230000002496 gastric effect Effects 0.000 description 4
- 239000008273 gelatin Substances 0.000 description 4
- 229920000159 gelatin Polymers 0.000 description 4
- 229940014259 gelatin Drugs 0.000 description 4
- 235000019322 gelatine Nutrition 0.000 description 4
- 235000011852 gelatine desserts Nutrition 0.000 description 4
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 4
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 4
- 239000003112 inhibitor Substances 0.000 description 4
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 4
- 210000004962 mammalian cell Anatomy 0.000 description 4
- 238000000691 measurement method Methods 0.000 description 4
- 210000004245 medial forebrain bundle Anatomy 0.000 description 4
- 230000004060 metabolic process Effects 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- 239000003921 oil Substances 0.000 description 4
- 235000019198 oils Nutrition 0.000 description 4
- 229940044551 receptor antagonist Drugs 0.000 description 4
- 239000002464 receptor antagonist Substances 0.000 description 4
- 238000010992 reflux Methods 0.000 description 4
- 238000009256 replacement therapy Methods 0.000 description 4
- UHSKFQJFRQCDBE-UHFFFAOYSA-N ropinirole Chemical compound CCCN(CCC)CCC1=CC=CC2=C1CC(=O)N2 UHSKFQJFRQCDBE-UHFFFAOYSA-N 0.000 description 4
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 4
- 235000017557 sodium bicarbonate Nutrition 0.000 description 4
- 229910052938 sodium sulfate Inorganic materials 0.000 description 4
- 235000011152 sodium sulphate Nutrition 0.000 description 4
- 239000003381 stabilizer Substances 0.000 description 4
- 125000003107 substituted aryl group Chemical group 0.000 description 4
- 238000010998 test method Methods 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 3
- FJKROLUGYXJWQN-UHFFFAOYSA-N 4-hydroxybenzoic acid Chemical compound OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 3
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 3
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 3
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 3
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 3
- 201000002832 Lewy body dementia Diseases 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- 108060001084 Luciferase Proteins 0.000 description 3
- 239000005089 Luciferase Substances 0.000 description 3
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- 229920002125 Sokalan® Polymers 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- 235000021355 Stearic acid Nutrition 0.000 description 3
- 239000003655 absorption accelerator Substances 0.000 description 3
- 230000002378 acidificating effect Effects 0.000 description 3
- 230000010933 acylation Effects 0.000 description 3
- 238000005917 acylation reaction Methods 0.000 description 3
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 3
- 239000003513 alkali Substances 0.000 description 3
- 229910052783 alkali metal Inorganic materials 0.000 description 3
- 150000001340 alkali metals Chemical class 0.000 description 3
- 235000003704 aspartic acid Nutrition 0.000 description 3
- RQPZNWPYLFFXCP-UHFFFAOYSA-L barium dihydroxide Chemical compound [OH-].[OH-].[Ba+2] RQPZNWPYLFFXCP-UHFFFAOYSA-L 0.000 description 3
- 229910001863 barium hydroxide Inorganic materials 0.000 description 3
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 230000036765 blood level Effects 0.000 description 3
- 239000006172 buffering agent Substances 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 3
- 239000001768 carboxy methyl cellulose Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 208000010877 cognitive disease Diseases 0.000 description 3
- 150000004696 coordination complex Chemical class 0.000 description 3
- 239000006071 cream Substances 0.000 description 3
- 239000003405 delayed action preparation Substances 0.000 description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 3
- 208000035475 disorder Diseases 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 239000003136 dopamine receptor stimulating agent Substances 0.000 description 3
- 230000001804 emulsifying effect Effects 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 150000004665 fatty acids Chemical class 0.000 description 3
- 235000019253 formic acid Nutrition 0.000 description 3
- 239000000499 gel Substances 0.000 description 3
- 235000013922 glutamic acid Nutrition 0.000 description 3
- 239000004220 glutamic acid Substances 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 3
- 239000001257 hydrogen Substances 0.000 description 3
- 229910052739 hydrogen Inorganic materials 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- 239000000314 lubricant Substances 0.000 description 3
- UKVIEHSSVKSQBA-UHFFFAOYSA-N methane;palladium Chemical compound C.[Pd] UKVIEHSSVKSQBA-UHFFFAOYSA-N 0.000 description 3
- 229920000609 methyl cellulose Polymers 0.000 description 3
- 235000010981 methylcellulose Nutrition 0.000 description 3
- 239000001923 methylcellulose Substances 0.000 description 3
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 3
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 3
- 230000001575 pathological effect Effects 0.000 description 3
- 229940127557 pharmaceutical product Drugs 0.000 description 3
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 3
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 3
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 239000002994 raw material Substances 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 230000007958 sleep Effects 0.000 description 3
- 229910000029 sodium carbonate Inorganic materials 0.000 description 3
- PRAKJMSDJKAYCZ-UHFFFAOYSA-N squalane Chemical compound CC(C)CCCC(C)CCCC(C)CCCCC(C)CCCC(C)CCCC(C)C PRAKJMSDJKAYCZ-UHFFFAOYSA-N 0.000 description 3
- 239000008107 starch Substances 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- 239000008117 stearic acid Substances 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- 239000000375 suspending agent Substances 0.000 description 3
- 230000002459 sustained effect Effects 0.000 description 3
- 239000002562 thickening agent Substances 0.000 description 3
- QIQITDHWZYEEPA-UHFFFAOYSA-N thiophene-2-carbonyl chloride Chemical compound ClC(=O)C1=CC=CS1 QIQITDHWZYEEPA-UHFFFAOYSA-N 0.000 description 3
- 238000012546 transfer Methods 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 description 2
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 2
- GHXNRYVDXNZXID-UHFFFAOYSA-N 2,2-diethylbutanoic acid Chemical compound CCC(CC)(CC)C(O)=O GHXNRYVDXNZXID-UHFFFAOYSA-N 0.000 description 2
- JVSFQJZRHXAUGT-UHFFFAOYSA-N 2,2-dimethylpropanoyl chloride Chemical compound CC(C)(C)C(Cl)=O JVSFQJZRHXAUGT-UHFFFAOYSA-N 0.000 description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 2
- NGNBDVOYPDDBFK-UHFFFAOYSA-N 2-[2,4-di(pentan-2-yl)phenoxy]acetyl chloride Chemical compound CCCC(C)C1=CC=C(OCC(Cl)=O)C(C(C)CCC)=C1 NGNBDVOYPDDBFK-UHFFFAOYSA-N 0.000 description 2
- YOETUEMZNOLGDB-UHFFFAOYSA-N 2-methylpropyl carbonochloridate Chemical compound CC(C)COC(Cl)=O YOETUEMZNOLGDB-UHFFFAOYSA-N 0.000 description 2
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 2
- 102100022738 5-hydroxytryptamine receptor 1A Human genes 0.000 description 2
- 101710138638 5-hydroxytryptamine receptor 1A Proteins 0.000 description 2
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 2
- 239000000775 AMPA receptor antagonist Substances 0.000 description 2
- 229940098747 AMPA receptor antagonist Drugs 0.000 description 2
- 244000215068 Acacia senegal Species 0.000 description 2
- 102000009346 Adenosine receptors Human genes 0.000 description 2
- 108050000203 Adenosine receptors Proteins 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- 208000019901 Anxiety disease Diseases 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- 208000028698 Cognitive impairment Diseases 0.000 description 2
- 206010010774 Constipation Diseases 0.000 description 2
- YVHAIVPPUIZFBA-UHFFFAOYSA-N Cyclopentylacetic acid Chemical compound OC(=O)CC1CCCC1 YVHAIVPPUIZFBA-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- 229940081615 DOPA decarboxylase inhibitor Drugs 0.000 description 2
- 101150049660 DRD2 gene Proteins 0.000 description 2
- HCYAFALTSJYZDH-UHFFFAOYSA-N Desimpramine Chemical compound C1CC2=CC=CC=C2N(CCCNC)C2=CC=CC=C21 HCYAFALTSJYZDH-UHFFFAOYSA-N 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 208000002603 Dopa-responsive dystonia Diseases 0.000 description 2
- 206010013642 Drooling Diseases 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 208000010228 Erectile Dysfunction Diseases 0.000 description 2
- 229940086575 Glutamate release inhibitor Drugs 0.000 description 2
- 229920000084 Gum arabic Polymers 0.000 description 2
- 208000004547 Hallucinations Diseases 0.000 description 2
- 208000008454 Hyperhidrosis Diseases 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 102000000521 Immunophilins Human genes 0.000 description 2
- 108010016648 Immunophilins Proteins 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 208000009829 Lewy Body Disease Diseases 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 2
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 2
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 2
- 102100025180 Mitogen-activated protein kinase kinase kinase 12 Human genes 0.000 description 2
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 2
- 108010025020 Nerve Growth Factor Proteins 0.000 description 2
- 102000007072 Nerve Growth Factors Human genes 0.000 description 2
- 102000019315 Nicotinic acetylcholine receptors Human genes 0.000 description 2
- 108050006807 Nicotinic acetylcholine receptors Proteins 0.000 description 2
- 239000005642 Oleic acid Substances 0.000 description 2
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- 208000002193 Pain Diseases 0.000 description 2
- 235000021314 Palmitic acid Nutrition 0.000 description 2
- QGMRQYFBGABWDR-UHFFFAOYSA-M Pentobarbital sodium Chemical compound [Na+].CCCC(C)C1(CC)C(=O)NC(=O)[N-]C1=O QGMRQYFBGABWDR-UHFFFAOYSA-M 0.000 description 2
- XYFCBTPGUUZFHI-UHFFFAOYSA-N Phosphine Chemical compound P XYFCBTPGUUZFHI-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- 206010037180 Psychiatric symptoms Diseases 0.000 description 2
- 208000005793 Restless legs syndrome Diseases 0.000 description 2
- XDXHAEQXIBQUEZ-UHFFFAOYSA-N Ropinirole hydrochloride Chemical compound Cl.CCCN(CCC)CCC1=CC=CC2=C1CC(=O)N2 XDXHAEQXIBQUEZ-UHFFFAOYSA-N 0.000 description 2
- 208000008630 Sialorrhea Diseases 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 206010044565 Tremor Diseases 0.000 description 2
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 238000002441 X-ray diffraction Methods 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- AMHPTVWBZSYFSS-BZUAXINKSA-N [(1r,3r,5r)-6,6,9-trimethyl-9-azabicyclo[3.3.1]nonan-3-yl] 2-hydroxy-2,2-dithiophen-2-ylacetate Chemical compound O([C@H]1C[C@@H]2C(C)(C)CC[C@H](C1)N2C)C(=O)C(O)(C=1SC=CC=1)C1=CC=CS1 AMHPTVWBZSYFSS-BZUAXINKSA-N 0.000 description 2
- PRLGAHUPIXAOLK-UHFFFAOYSA-N [I+].C[N+]1=CC=CC=C1Cl Chemical compound [I+].C[N+]1=CC=CC=C1Cl PRLGAHUPIXAOLK-UHFFFAOYSA-N 0.000 description 2
- 235000010489 acacia gum Nutrition 0.000 description 2
- 239000000205 acacia gum Substances 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 239000001361 adipic acid Substances 0.000 description 2
- 235000011037 adipic acid Nutrition 0.000 description 2
- 239000000443 aerosol Substances 0.000 description 2
- 239000000556 agonist Substances 0.000 description 2
- VZTDIZULWFCMLS-UHFFFAOYSA-N ammonium formate Chemical compound [NH4+].[O-]C=O VZTDIZULWFCMLS-UHFFFAOYSA-N 0.000 description 2
- RDOXTESZEPMUJZ-UHFFFAOYSA-N anisole Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 2
- 239000005557 antagonist Substances 0.000 description 2
- 239000001961 anticonvulsive agent Substances 0.000 description 2
- 239000000935 antidepressant agent Substances 0.000 description 2
- 229940005513 antidepressants Drugs 0.000 description 2
- 239000000739 antihistaminic agent Substances 0.000 description 2
- 239000000164 antipsychotic agent Substances 0.000 description 2
- 229940005529 antipsychotics Drugs 0.000 description 2
- 230000036506 anxiety Effects 0.000 description 2
- 229960004046 apomorphine Drugs 0.000 description 2
- VMWNQDUVQKEIOC-CYBMUJFWSA-N apomorphine Chemical compound C([C@H]1N(C)CC2)C3=CC=C(O)C(O)=C3C3=C1C2=CC=C3 VMWNQDUVQKEIOC-CYBMUJFWSA-N 0.000 description 2
- 229960000686 benzalkonium chloride Drugs 0.000 description 2
- PASDCCFISLVPSO-UHFFFAOYSA-N benzoyl chloride Chemical compound ClC(=O)C1=CC=CC=C1 PASDCCFISLVPSO-UHFFFAOYSA-N 0.000 description 2
- AGEZXYOZHKGVCM-UHFFFAOYSA-N benzyl bromide Chemical compound BrCC1=CC=CC=C1 AGEZXYOZHKGVCM-UHFFFAOYSA-N 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 2
- WGQKYBSKWIADBV-UHFFFAOYSA-N benzylamine Chemical class NCC1=CC=CC=C1 WGQKYBSKWIADBV-UHFFFAOYSA-N 0.000 description 2
- 229960002802 bromocriptine Drugs 0.000 description 2
- OZVBMTJYIDMWIL-AYFBDAFISA-N bromocriptine Chemical compound C1=CC(C=2[C@H](N(C)C[C@@H](C=2)C(=O)N[C@]2(C(=O)N3[C@H](C(N4CCC[C@H]4[C@]3(O)O2)=O)CC(C)C)C(C)C)C2)=C3C2=C(Br)NC3=C1 OZVBMTJYIDMWIL-AYFBDAFISA-N 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- PFKFTWBEEFSNDU-UHFFFAOYSA-N carbonyldiimidazole Chemical compound C1=CN=CN1C(=O)N1C=CN=C1 PFKFTWBEEFSNDU-UHFFFAOYSA-N 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- 229940105329 carboxymethylcellulose Drugs 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 238000002659 cell therapy Methods 0.000 description 2
- 208000026106 cerebrovascular disease Diseases 0.000 description 2
- 229960000541 cetyl alcohol Drugs 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 238000013329 compounding Methods 0.000 description 2
- 238000012790 confirmation Methods 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- 230000007850 degeneration Effects 0.000 description 2
- 229960003914 desipramine Drugs 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 239000000534 dopa decarboxylase inhibitor Substances 0.000 description 2
- 239000000221 dopamine uptake inhibitor Substances 0.000 description 2
- 206010013990 dysuria Diseases 0.000 description 2
- 230000008451 emotion Effects 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 150000002148 esters Chemical group 0.000 description 2
- 239000003687 estradiol congener Substances 0.000 description 2
- RIFGWPKJUGCATF-UHFFFAOYSA-N ethyl chloroformate Chemical compound CCOC(Cl)=O RIFGWPKJUGCATF-UHFFFAOYSA-N 0.000 description 2
- 239000003925 fat Substances 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 125000000524 functional group Chemical group 0.000 description 2
- 238000001415 gene therapy Methods 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 150000002430 hydrocarbons Chemical class 0.000 description 2
- 238000007327 hydrogenolysis reaction Methods 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 201000001881 impotence Diseases 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 239000011261 inert gas Substances 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 239000000543 intermediate Substances 0.000 description 2
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 2
- 239000007951 isotonicity adjuster Substances 0.000 description 2
- 229940043355 kinase inhibitor Drugs 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 229950011566 leteprinim Drugs 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 239000000865 liniment Substances 0.000 description 2
- 231100000053 low toxicity Toxicity 0.000 description 2
- 235000019359 magnesium stearate Nutrition 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 229960000219 mazaticol Drugs 0.000 description 2
- XBBDACCLCFWBSI-ZETCQYMHSA-N melevodopa Chemical compound COC(=O)[C@@H](N)CC1=CC=C(O)C(O)=C1 XBBDACCLCFWBSI-ZETCQYMHSA-N 0.000 description 2
- 229960001794 melevodopa Drugs 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 2
- 239000008108 microcrystalline cellulose Substances 0.000 description 2
- 229940016286 microcrystalline cellulose Drugs 0.000 description 2
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 2
- 108090001035 mitogen-activated protein kinase kinase kinase 12 Proteins 0.000 description 2
- 150000004682 monohydrates Chemical class 0.000 description 2
- 231100000219 mutagenic Toxicity 0.000 description 2
- 230000003505 mutagenic effect Effects 0.000 description 2
- WQEPLUUGTLDZJY-UHFFFAOYSA-N n-Pentadecanoic acid Natural products CCCCCCCCCCCCCCC(O)=O WQEPLUUGTLDZJY-UHFFFAOYSA-N 0.000 description 2
- 239000006199 nebulizer Substances 0.000 description 2
- 239000004090 neuroprotective agent Substances 0.000 description 2
- 239000003900 neurotrophic factor Substances 0.000 description 2
- ZWLPBLYKEWSWPD-UHFFFAOYSA-N o-toluic acid Chemical compound CC1=CC=CC=C1C(O)=O ZWLPBLYKEWSWPD-UHFFFAOYSA-N 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- KJIFKLIQANRMOU-UHFFFAOYSA-N oxidanium;4-methylbenzenesulfonate Chemical compound O.CC1=CC=C(S(O)(=O)=O)C=C1 KJIFKLIQANRMOU-UHFFFAOYSA-N 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 235000019271 petrolatum Nutrition 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 235000021317 phosphate Nutrition 0.000 description 2
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 2
- NKJQZSDCCLDOQH-UHFFFAOYSA-N piroheptine Chemical compound CC1N(CC)CCC1=C1C2=CC=CC=C2CCC2=CC=CC=C21 NKJQZSDCCLDOQH-UHFFFAOYSA-N 0.000 description 2
- 229950009232 piroheptine Drugs 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 208000028893 postprandial hypotension Diseases 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229960003975 potassium Drugs 0.000 description 2
- 235000015497 potassium bicarbonate Nutrition 0.000 description 2
- 229910000028 potassium bicarbonate Inorganic materials 0.000 description 2
- 239000011736 potassium bicarbonate Substances 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 2
- MICLTPPSCUXHJT-UHFFFAOYSA-M potassium;4-[3-(6-oxo-3h-purin-9-yl)propanoylamino]benzoate Chemical compound [K+].C1=CC(C(=O)[O-])=CC=C1NC(=O)CCN1C(NC=NC2=O)=C2N=C1 MICLTPPSCUXHJT-UHFFFAOYSA-M 0.000 description 2
- APVQOOKHDZVJEX-QTPLPEIMSA-N pramipexole hydrochloride Chemical compound O.Cl.Cl.C1[C@@H](NCCC)CCC2=C1SC(N)=N2 APVQOOKHDZVJEX-QTPLPEIMSA-N 0.000 description 2
- 239000008213 purified water Substances 0.000 description 2
- 229940044601 receptor agonist Drugs 0.000 description 2
- 239000000018 receptor agonist Substances 0.000 description 2
- 229960001879 ropinirole Drugs 0.000 description 2
- 229960002349 ropinirole hydrochloride Drugs 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 239000000952 serotonin receptor agonist Substances 0.000 description 2
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 2
- 229940001089 sinemet Drugs 0.000 description 2
- 208000019116 sleep disease Diseases 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 239000000021 stimulant Substances 0.000 description 2
- 210000002784 stomach Anatomy 0.000 description 2
- 125000005346 substituted cycloalkyl group Chemical group 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 208000011580 syndromic disease Diseases 0.000 description 2
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 2
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 230000009466 transformation Effects 0.000 description 2
- 238000000844 transformation Methods 0.000 description 2
- AQRLNPVMDITEJU-UHFFFAOYSA-N triethylsilane Chemical compound CC[SiH](CC)CC AQRLNPVMDITEJU-UHFFFAOYSA-N 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- 235000015112 vegetable and seed oil Nutrition 0.000 description 2
- 239000008158 vegetable oil Substances 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- 239000001993 wax Substances 0.000 description 2
- 239000000080 wetting agent Substances 0.000 description 2
- AHOUBRCZNHFOSL-YOEHRIQHSA-N (+)-Casbol Chemical compound C1=CC(F)=CC=C1[C@H]1[C@H](COC=2C=C3OCOC3=CC=2)CNCC1 AHOUBRCZNHFOSL-YOEHRIQHSA-N 0.000 description 1
- LTFSOLPFYIMGLX-UHFFFAOYSA-N (2,2-dimethyl-3-oxo-3-phenylmethoxypropyl) 2-methoxybenzoate Chemical compound COC1=CC=CC=C1C(=O)OCC(C)(C)C(=O)OCC1=CC=CC=C1 LTFSOLPFYIMGLX-UHFFFAOYSA-N 0.000 description 1
- PRVJQIVXFSRWMF-UHFFFAOYSA-N (2,2-dimethyl-3-oxo-3-phenylmethoxypropyl) benzoate Chemical compound C=1C=CC=CC=1COC(=O)C(C)(C)COC(=O)C1=CC=CC=C1 PRVJQIVXFSRWMF-UHFFFAOYSA-N 0.000 description 1
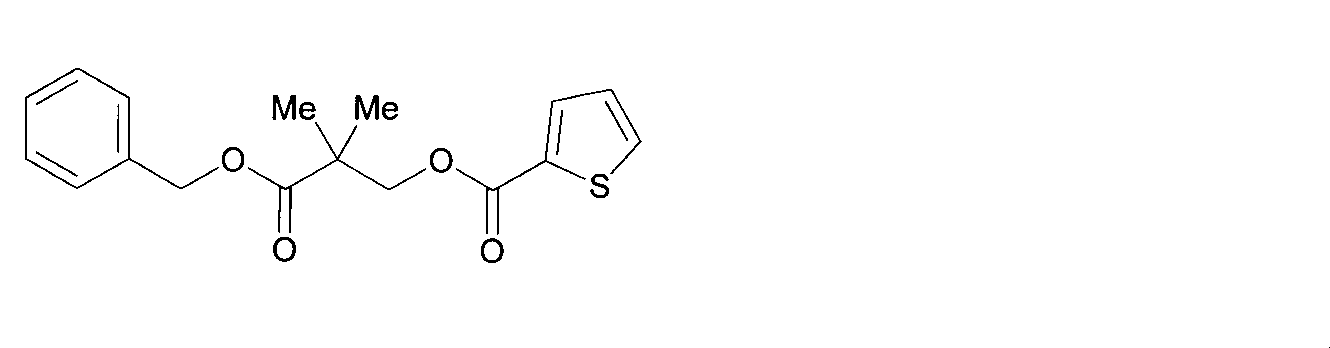
- GXPHGCHZOPTABT-UHFFFAOYSA-N (2,2-dimethyl-3-oxo-3-phenylmethoxypropyl) thiophene-3-carboxylate Chemical compound C=1C=CC=CC=1COC(=O)C(C)(C)COC(=O)C=1C=CSC=1 GXPHGCHZOPTABT-UHFFFAOYSA-N 0.000 description 1
- GMBQZIIUCVWOCD-UQHLGXRBSA-N (25R)-5beta-spirostan-3beta-ol Chemical compound O([C@@H]1[C@@H]([C@]2(CC[C@@H]3[C@@]4(C)CC[C@H](O)C[C@H]4CC[C@H]3[C@@H]2C1)C)[C@@H]1C)[C@]11CC[C@@H](C)CO1 GMBQZIIUCVWOCD-UQHLGXRBSA-N 0.000 description 1
- VXLBSYHAEKDUSU-JXMROGBWSA-N (2e)-2-(fluoromethylidene)-4-(4-fluorophenyl)butan-1-amine Chemical compound NC\C(=C\F)CCC1=CC=C(F)C=C1 VXLBSYHAEKDUSU-JXMROGBWSA-N 0.000 description 1
- OBETXYAYXDNJHR-SSDOTTSWSA-M (2r)-2-ethylhexanoate Chemical compound CCCC[C@@H](CC)C([O-])=O OBETXYAYXDNJHR-SSDOTTSWSA-M 0.000 description 1
- XWAVPOFYNPXXEL-MRVPVSSYSA-N (2r)-2-phenylmethoxypropanoic acid Chemical compound OC(=O)[C@@H](C)OCC1=CC=CC=C1 XWAVPOFYNPXXEL-MRVPVSSYSA-N 0.000 description 1
- DSEKYWAQQVUQTP-XEWMWGOFSA-N (2r,4r,4as,6as,6as,6br,8ar,12ar,14as,14bs)-2-hydroxy-4,4a,6a,6b,8a,11,11,14a-octamethyl-2,4,5,6,6a,7,8,9,10,12,12a,13,14,14b-tetradecahydro-1h-picen-3-one Chemical compound C([C@H]1[C@]2(C)CC[C@@]34C)C(C)(C)CC[C@]1(C)CC[C@]2(C)[C@H]4CC[C@@]1(C)[C@H]3C[C@@H](O)C(=O)[C@@H]1C DSEKYWAQQVUQTP-XEWMWGOFSA-N 0.000 description 1
- FOPALECPEUVCTL-QMMMGPOBSA-N (2s)-1-(3,3-dimethyl-2-oxopentanoyl)pyrrolidine-2-carboxylic acid Chemical compound CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(O)=O FOPALECPEUVCTL-QMMMGPOBSA-N 0.000 description 1
- FFZGHUAZJKPSSP-FERBBOLQSA-N (2s)-2-amino-3-[3,4-bis(2,2-diethylbutanoyloxy)phenyl]propanoic acid;hydrochloride Chemical compound Cl.CCC(CC)(CC)C(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)C(CC)(CC)CC FFZGHUAZJKPSSP-FERBBOLQSA-N 0.000 description 1
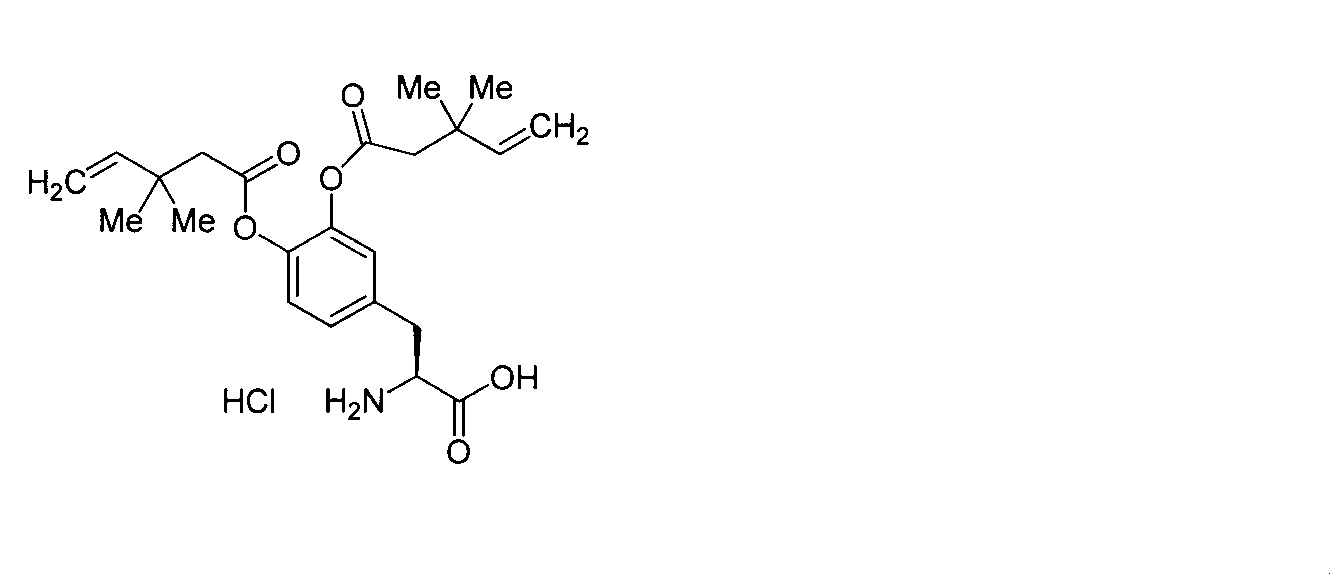
- DHOKBWZZLXPDJG-NTISSMGPSA-N (2s)-2-amino-3-[3,4-bis(3,3-dimethylpent-4-enoyloxy)phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=CC(C)(C)CC(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)CC(C)(C)C=C DHOKBWZZLXPDJG-NTISSMGPSA-N 0.000 description 1
- ZXYHDZDIEFJGLA-NTISSMGPSA-N (2s)-2-amino-3-[3,4-bis(4,4-dimethylpentanoyloxy)phenyl]propanoic acid;hydrochloride Chemical compound Cl.CC(C)(C)CCC(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)CCC(C)(C)C ZXYHDZDIEFJGLA-NTISSMGPSA-N 0.000 description 1
- ITZXHLACSKYLAR-YDALLXLXSA-N (2s)-2-amino-3-[3,4-bis(cyclopropanecarbonyloxy)phenyl]propanoic acid;hydrochloride Chemical compound Cl.C1CC1C(=O)OC1=CC(C[C@H](N)C(O)=O)=CC=C1OC(=O)C1CC1 ITZXHLACSKYLAR-YDALLXLXSA-N 0.000 description 1
- PITCVNZWSHKVFV-YDALLXLXSA-N (2s)-2-amino-3-[3,4-bis[(1-methylcyclopropanecarbonyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)C2(C)CC2)C=1OC(=O)C1(C)CC1 PITCVNZWSHKVFV-YDALLXLXSA-N 0.000 description 1
- XUXMZHOFVDNUNF-UQKRIMTDSA-N (2s)-2-amino-3-[3,4-bis[(2-acetyloxy-2-methylpropanoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.CC(=O)OC(C)(C)C(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)C(C)(C)OC(C)=O XUXMZHOFVDNUNF-UQKRIMTDSA-N 0.000 description 1
- UZIRYMVAAQFWIK-SNYZSRNZSA-N (2s)-2-amino-3-[3,4-bis[(2-benzoyloxy-2-ethylbutanoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)C(CC)(CC)OC(=O)C=2C=CC=CC=2)C=1OC(=O)C(CC)(CC)OC(=O)C1=CC=CC=C1 UZIRYMVAAQFWIK-SNYZSRNZSA-N 0.000 description 1
- GWBXZGFQNASYMZ-BDQAORGHSA-N (2s)-2-amino-3-[3,4-bis[(2-cyclohexylacetyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C1CCCCC1CC(=O)OC1=CC(C[C@H](N)C(O)=O)=CC=C1OC(=O)CC1CCCCC1 GWBXZGFQNASYMZ-BDQAORGHSA-N 0.000 description 1
- XWPBQVYHWSSJGL-FERBBOLQSA-N (2s)-2-amino-3-[3,4-bis[(2-cyclopentylacetyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C1CCCC1CC(=O)OC1=CC(C[C@H](N)C(O)=O)=CC=C1OC(=O)CC1CCCC1 XWPBQVYHWSSJGL-FERBBOLQSA-N 0.000 description 1
- MWDPRTGEMSNCMG-NTISSMGPSA-N (2s)-2-amino-3-[3,4-bis[(2-ethyl-2-methylbutanoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.CCC(C)(CC)C(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)C(C)(CC)CC MWDPRTGEMSNCMG-NTISSMGPSA-N 0.000 description 1
- JSFSTPNXPCTIHW-BDQAORGHSA-N (2s)-2-amino-3-[3,4-bis[(2-methylbenzoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.CC1=CC=CC=C1C(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)C1=CC=CC=C1C JSFSTPNXPCTIHW-BDQAORGHSA-N 0.000 description 1
- NDHXBZFGFRZORE-JIDHJSLPSA-N (2s)-2-amino-3-[3,4-bis[(3-benzoyloxy-2,2-dimethylpropanoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)C(C)(C)COC(=O)C=2C=CC=CC=2)C=1OC(=O)C(C)(C)COC(=O)C1=CC=CC=C1 NDHXBZFGFRZORE-JIDHJSLPSA-N 0.000 description 1
- UQDJJWDHKWCFAF-FERBBOLQSA-N (2s)-2-amino-3-[3,4-bis[(3-ethyl-3-methylpentanoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.CCC(C)(CC)CC(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)CC(C)(CC)CC UQDJJWDHKWCFAF-FERBBOLQSA-N 0.000 description 1
- RWHZXUNWFYULLV-FERBBOLQSA-N (2s)-2-amino-3-[3,4-bis[(4-hydroxybenzoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(O)C=CC=1C(=O)OC1=CC(C[C@H](N)C(O)=O)=CC=C1OC(=O)C1=CC=C(O)C=C1 RWHZXUNWFYULLV-FERBBOLQSA-N 0.000 description 1
- XFMMUEHKBXUVIV-FTBISJDPSA-N (2s)-2-amino-3-[3,4-bis[(4-methyl-3-propan-2-ylpentanoyl)oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.CC(C)C(C(C)C)CC(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1OC(=O)CC(C(C)C)C(C)C XFMMUEHKBXUVIV-FTBISJDPSA-N 0.000 description 1
- FKSRENSSJUUXQI-KJGMQWEMSA-N (2s)-2-amino-3-[3,4-bis[[(2r)-2-benzoyloxypropanoyl]oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.O([C@H](C)C(=O)OC=1C(=CC(C[C@H](N)C(O)=O)=CC=1)OC(=O)[C@@H](C)OC(=O)C=1C=CC=CC=1)C(=O)C1=CC=CC=C1 FKSRENSSJUUXQI-KJGMQWEMSA-N 0.000 description 1
- FKSRENSSJUUXQI-JIXBSQRZSA-N (2s)-2-amino-3-[3,4-bis[[(2s)-2-benzoyloxypropanoyl]oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.O([C@@H](C)C(=O)OC=1C(=CC(C[C@H](N)C(O)=O)=CC=1)OC(=O)[C@H](C)OC(=O)C=1C=CC=CC=1)C(=O)C1=CC=CC=C1 FKSRENSSJUUXQI-JIXBSQRZSA-N 0.000 description 1
- YCYKPEVQHPTZOL-FERBBOLQSA-N (2s)-2-amino-3-[3,4-bis[[2,2-dimethyl-3-(thiophene-2-carbonyloxy)propanoyl]oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)C(C)(C)COC(=O)C=2SC=CC=2)C=1OC(=O)C(C)(C)COC(=O)C1=CC=CS1 YCYKPEVQHPTZOL-FERBBOLQSA-N 0.000 description 1
- OXIJPDYBZOUDNN-BDQAORGHSA-N (2s)-2-amino-3-[3,4-bis[[2,2-dimethyl-3-(thiophene-3-carbonyloxy)propanoyl]oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)C(C)(C)COC(=O)C2=CSC=C2)C=1OC(=O)C(C)(C)COC(=O)C=1C=CSC=1 OXIJPDYBZOUDNN-BDQAORGHSA-N 0.000 description 1
- FFCTVDVFTHSQJI-BDQAORGHSA-N (2s)-2-amino-3-[3,4-bis[[2-(1-methylcyclohexyl)acetyl]oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)CC2(C)CCCCC2)C=1OC(=O)CC1(C)CCCCC1 FFCTVDVFTHSQJI-BDQAORGHSA-N 0.000 description 1
- NCWIVUYDCNUNRW-FERBBOLQSA-N (2s)-2-amino-3-[3,4-bis[[2-(1-methylcyclopentyl)acetyl]oxy]phenyl]propanoic acid;hydrochloride Chemical compound Cl.C=1C=C(C[C@H](N)C(O)=O)C=C(OC(=O)CC2(C)CCCC2)C=1OC(=O)CC1(C)CCCC1 NCWIVUYDCNUNRW-FERBBOLQSA-N 0.000 description 1
- ARFWITPWHIUQQJ-SFHVURJKSA-N (2s)-2-amino-3-[3,4-bis[[2-(trifluoromethyl)benzoyl]oxy]phenyl]propanoic acid Chemical compound C=1C=CC=C(C(F)(F)F)C=1C(=O)OC1=CC(C[C@H](N)C(O)=O)=CC=C1OC(=O)C1=CC=CC=C1C(F)(F)F ARFWITPWHIUQQJ-SFHVURJKSA-N 0.000 description 1
- ODRCDAPMBBAREL-QMMMGPOBSA-N (2s)-2-amino-3-[3-hydroxy-4-(2-hydroxy-2-methylpropanoyl)oxyphenyl]propanoic acid Chemical compound CC(C)(O)C(=O)OC1=CC=C(C[C@H](N)C(O)=O)C=C1O ODRCDAPMBBAREL-QMMMGPOBSA-N 0.000 description 1
- OUXFBCFJKVIFMW-QMMMGPOBSA-N (2s)-2-amino-3-[4-hydroxy-3-(2-hydroxy-2-methylpropanoyl)oxyphenyl]propanoic acid Chemical compound CC(C)(O)C(=O)OC1=CC(C[C@H](N)C(O)=O)=CC=C1O OUXFBCFJKVIFMW-QMMMGPOBSA-N 0.000 description 1
- XWAVPOFYNPXXEL-QMMMGPOBSA-N (2s)-2-phenylmethoxypropanoic acid Chemical compound OC(=O)[C@H](C)OCC1=CC=CC=C1 XWAVPOFYNPXXEL-QMMMGPOBSA-N 0.000 description 1
- FZTWZSXBKWSEAJ-MHZLTWQESA-N (2s)-3-[3,4-bis[(3-benzoyloxy-2,2-dimethylpropanoyl)oxy]phenyl]-2-[(2-methylpropan-2-yl)oxycarbonylamino]propanoic acid Chemical compound C=1C=CC=CC=1C(=O)OCC(C)(C)C(=O)OC1=CC(C[C@H](NC(=O)OC(C)(C)C)C(O)=O)=CC=C1OC(=O)C(C)(C)COC(=O)C1=CC=CC=C1 FZTWZSXBKWSEAJ-MHZLTWQESA-N 0.000 description 1
- XUTANPWEKDKDKQ-NRFANRHFSA-N (2s)-3-[3,4-bis[[2,2-dimethyl-3-(thiophene-2-carbonyloxy)propanoyl]oxy]phenyl]-2-[(2-methylpropan-2-yl)oxycarbonylamino]propanoic acid Chemical compound C=1C=CSC=1C(=O)OCC(C)(C)C(=O)OC1=CC(C[C@H](NC(=O)OC(C)(C)C)C(O)=O)=CC=C1OC(=O)C(C)(C)COC(=O)C1=CC=CS1 XUTANPWEKDKDKQ-NRFANRHFSA-N 0.000 description 1
- TYHXRVDMISGDLS-QHCPKHFHSA-N (2s)-3-[3,4-bis[[2,2-dimethyl-3-(thiophene-3-carbonyloxy)propanoyl]oxy]phenyl]-2-[(2-methylpropan-2-yl)oxycarbonylamino]propanoic acid Chemical compound C1=CSC=C1C(=O)OCC(C)(C)C(=O)OC1=CC(C[C@H](NC(=O)OC(C)(C)C)C(O)=O)=CC=C1OC(=O)C(C)(C)COC(=O)C=1C=CSC=1 TYHXRVDMISGDLS-QHCPKHFHSA-N 0.000 description 1
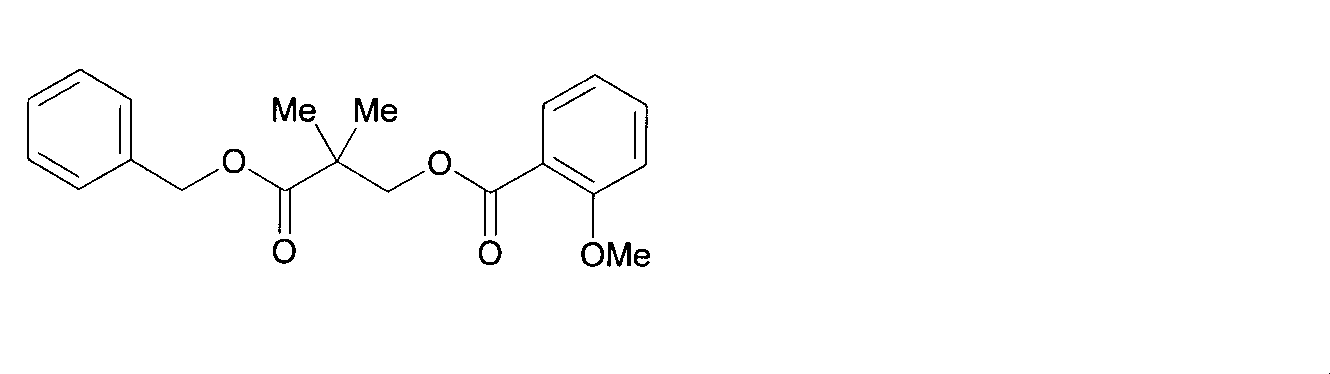
- CXISZLRUJLFYAZ-MHZLTWQESA-N (2s)-3-[3,4-bis[[3-(2-methoxybenzoyl)oxy-2,2-dimethylpropanoyl]oxy]phenyl]-2-[(2-methylpropan-2-yl)oxycarbonylamino]propanoic acid Chemical compound COC1=CC=CC=C1C(=O)OCC(C)(C)C(=O)OC1=CC=C(C[C@H](NC(=O)OC(C)(C)C)C(O)=O)C=C1OC(=O)C(C)(C)COC(=O)C1=CC=CC=C1OC CXISZLRUJLFYAZ-MHZLTWQESA-N 0.000 description 1
- YYGNTYWPHWGJRM-UHFFFAOYSA-N (6E,10E,14E,18E)-2,6,10,15,19,23-hexamethyltetracosa-2,6,10,14,18,22-hexaene Chemical compound CC(C)=CCCC(C)=CCCC(C)=CCCC=C(C)CCC=C(C)CCC=C(C)C YYGNTYWPHWGJRM-UHFFFAOYSA-N 0.000 description 1
- BGOQGUHWXBGXJW-RHSMWYFYSA-N (6aR,12bS)-5,6,6a,7,8,12b-hexahydrobenzo[a]phenanthridine-10,11-diol Chemical compound N1CC2=CC=CC=C2[C@H]2[C@H]1CCC1=C2C=C(O)C(O)=C1 BGOQGUHWXBGXJW-RHSMWYFYSA-N 0.000 description 1
- SKYZYDSNJIOXRL-BTQNPOSSSA-N (6ar)-6-methyl-5,6,6a,7-tetrahydro-4h-dibenzo[de,g]quinoline-10,11-diol;hydrochloride Chemical compound Cl.C([C@H]1N(C)CC2)C3=CC=C(O)C(O)=C3C3=C1C2=CC=C3 SKYZYDSNJIOXRL-BTQNPOSSSA-N 0.000 description 1
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 1
- RTHCYVBBDHJXIQ-MRXNPFEDSA-N (R)-fluoxetine Chemical compound O([C@H](CCNC)C=1C=CC=CC=1)C1=CC=C(C(F)(F)F)C=C1 RTHCYVBBDHJXIQ-MRXNPFEDSA-N 0.000 description 1
- SYTBZMRGLBWNTM-SNVBAGLBSA-N (R)-flurbiprofen Chemical compound FC1=CC([C@H](C(O)=O)C)=CC=C1C1=CC=CC=C1 SYTBZMRGLBWNTM-SNVBAGLBSA-N 0.000 description 1
- XHBLRJRZRFZSGW-UHFFFAOYSA-N (S)-2-amino-3-(3,4-methylenedioxyphenyl)propanoic acid Natural products OC(=O)C(N)CC1=CC=C2OCOC2=C1 XHBLRJRZRFZSGW-UHFFFAOYSA-N 0.000 description 1
- ZEUITGRIYCTCEM-KRWDZBQOSA-N (S)-duloxetine Chemical compound C1([C@@H](OC=2C3=CC=CC=C3C=CC=2)CCNC)=CC=CS1 ZEUITGRIYCTCEM-KRWDZBQOSA-N 0.000 description 1
- BCPPKHPWLRPWBJ-OURKGEEVSA-N (z)-but-2-enedioic acid;3-ethynyl-5-[(2s)-1-methylpyrrolidin-2-yl]pyridine Chemical compound OC(=O)\C=C/C(O)=O.CN1CCC[C@H]1C1=CN=CC(C#C)=C1 BCPPKHPWLRPWBJ-OURKGEEVSA-N 0.000 description 1
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 1
- 229940058015 1,3-butylene glycol Drugs 0.000 description 1
- CNEWKIDCGDXBDE-UHFFFAOYSA-N 1-[2-(4-phenylphenyl)ethyl]-4-[3-(trifluoromethyl)phenyl]-3,6-dihydro-2h-pyridine Chemical compound FC(F)(F)C1=CC=CC(C=2CCN(CCC=3C=CC(=CC=3)C=3C=CC=CC=3)CC=2)=C1 CNEWKIDCGDXBDE-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- DIZKLZKLNKQFGB-UHFFFAOYSA-N 1-methylcyclopropane-1-carboxylic acid Chemical compound OC(=O)C1(C)CC1 DIZKLZKLNKQFGB-UHFFFAOYSA-N 0.000 description 1
- VOXZDWNPVJITMN-SFFUCWETSA-N 17α-estradiol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 VOXZDWNPVJITMN-SFFUCWETSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- BXCUSXZFYQLWLL-UHFFFAOYSA-N 2,2-dimethyl-3-(thiophene-2-carbonyloxy)propanoic acid Chemical compound OC(=O)C(C)(C)COC(=O)C1=CC=CS1 BXCUSXZFYQLWLL-UHFFFAOYSA-N 0.000 description 1
- FTZCWISJGSNDEZ-UHFFFAOYSA-N 2,2-dimethyl-3-(thiophene-3-carbonyloxy)propanoic acid Chemical compound OC(=O)C(C)(C)COC(=O)C=1C=CSC=1 FTZCWISJGSNDEZ-UHFFFAOYSA-N 0.000 description 1
- ZHQAIBYWBFMHHQ-UHFFFAOYSA-N 2-(1-methylcyclopentyl)acetic acid Chemical compound OC(=O)CC1(C)CCCC1 ZHQAIBYWBFMHHQ-UHFFFAOYSA-N 0.000 description 1
- 125000003821 2-(trimethylsilyl)ethoxymethyl group Chemical group [H]C([H])([H])[Si](C([H])([H])[H])(C([H])([H])[H])C([H])([H])C(OC([H])([H])[*])([H])[H] 0.000 description 1
- YFGHCGITMMYXAQ-UHFFFAOYSA-N 2-[(diphenylmethyl)sulfinyl]acetamide Chemical compound C=1C=CC=CC=1C(S(=O)CC(=O)N)C1=CC=CC=C1 YFGHCGITMMYXAQ-UHFFFAOYSA-N 0.000 description 1
- OQDPVLVUJFGPGQ-UHFFFAOYSA-N 2-[4-(1,3-benzodioxol-5-ylmethyl)-1-piperazinyl]pyrimidine Chemical compound C=1C=C2OCOC2=CC=1CN(CC1)CCN1C1=NC=CC=N1 OQDPVLVUJFGPGQ-UHFFFAOYSA-N 0.000 description 1
- NIWFXRKHWKGFMT-UHFFFAOYSA-N 2-acetyloxy-2-methylpropanoic acid Chemical compound CC(=O)OC(C)(C)C(O)=O NIWFXRKHWKGFMT-UHFFFAOYSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- YSGASDXSLKIKOD-UHFFFAOYSA-N 2-amino-N-(1,2-diphenylpropan-2-yl)acetamide Chemical compound C=1C=CC=CC=1C(C)(NC(=O)CN)CC1=CC=CC=C1 YSGASDXSLKIKOD-UHFFFAOYSA-N 0.000 description 1
- HYQMIUSWZXGTCC-UHFFFAOYSA-N 2-amino-n-(1,2-diphenylpropan-2-yl)acetamide;hydron;chloride Chemical compound Cl.C=1C=CC=CC=1C(C)(NC(=O)CN)CC1=CC=CC=C1 HYQMIUSWZXGTCC-UHFFFAOYSA-N 0.000 description 1
- ABFPKTQEQNICFT-UHFFFAOYSA-M 2-chloro-1-methylpyridin-1-ium;iodide Chemical compound [I-].C[N+]1=CC=CC=C1Cl ABFPKTQEQNICFT-UHFFFAOYSA-M 0.000 description 1
- LHJPKLWGGMAUAN-UHFFFAOYSA-N 2-ethyl-2-methyl-butanoic acid Chemical compound CCC(C)(CC)C(O)=O LHJPKLWGGMAUAN-UHFFFAOYSA-N 0.000 description 1
- PZYPAXZIPRVVKM-UHFFFAOYSA-N 2-ethyl-2-phenylmethoxybutanoic acid Chemical compound CCC(CC)(C(O)=O)OCC1=CC=CC=C1 PZYPAXZIPRVVKM-UHFFFAOYSA-N 0.000 description 1
- XULHFMYCBKQGEE-UHFFFAOYSA-N 2-hexyl-1-Decanol Chemical compound CCCCCCCCC(CO)CCCCCC XULHFMYCBKQGEE-UHFFFAOYSA-N 0.000 description 1
- RFVNOJDQRGSOEL-UHFFFAOYSA-N 2-hydroxyethyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCO RFVNOJDQRGSOEL-UHFFFAOYSA-N 0.000 description 1
- RZNHSEZOLFEFGB-UHFFFAOYSA-N 2-methoxybenzoyl chloride Chemical compound COC1=CC=CC=C1C(Cl)=O RZNHSEZOLFEFGB-UHFFFAOYSA-N 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-M 2-methylbenzenesulfonate Chemical compound CC1=CC=CC=C1S([O-])(=O)=O LBLYYCQCTBFVLH-UHFFFAOYSA-M 0.000 description 1
- BSKHPKMHTQYZBB-UHFFFAOYSA-N 2-methylpyridine Chemical compound CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 description 1
- FYYOTZLMLLTWAP-UHFFFAOYSA-N 3,3-dimethylpent-4-enoic acid Chemical compound C=CC(C)(C)CC(O)=O FYYOTZLMLLTWAP-UHFFFAOYSA-N 0.000 description 1
- WGTASENVNYJZBK-UHFFFAOYSA-N 3,4,5-trimethoxyamphetamine Chemical class COC1=CC(CC(C)N)=CC(OC)=C1OC WGTASENVNYJZBK-UHFFFAOYSA-N 0.000 description 1
- KNCDEKVABZGNKW-UHFFFAOYSA-N 3-(2-methoxybenzoyl)oxy-2,2-dimethylpropanoic acid Chemical compound COC1=CC=CC=C1C(=O)OCC(C)(C)C(O)=O KNCDEKVABZGNKW-UHFFFAOYSA-N 0.000 description 1
- HQSBCDPYXDGTCL-UHFFFAOYSA-N 3-[(4-amino-3-methylphenyl)methyl]-7-(furan-2-yl)triazolo[4,5-d]pyrimidin-5-amine Chemical compound C1=C(N)C(C)=CC(CN2C3=NC(N)=NC(=C3N=N2)C=2OC=CC=2)=C1 HQSBCDPYXDGTCL-UHFFFAOYSA-N 0.000 description 1
- CVQFAMQDTWVJSV-LQGIQHHOSA-N 3-[(6aR,9S)-7-methyl-6,6a,8,9-tetrahydro-4H-indolo[4,3-fg]quinolin-9-yl]-1,1-diethylurea (E)-but-2-enedioic acid Chemical compound OC(=O)\C=C\C(O)=O.C1=CC(C=2[C@H](N(C)C[C@H](C=2)NC(=O)N(CC)CC)C2)=C3C2=CNC3=C1 CVQFAMQDTWVJSV-LQGIQHHOSA-N 0.000 description 1
- ZSJHWNREWOLDBF-UHFFFAOYSA-N 3-benzoyloxy-2,2-dimethylpropanoic acid Chemical compound OC(=O)C(C)(C)COC(=O)C1=CC=CC=C1 ZSJHWNREWOLDBF-UHFFFAOYSA-N 0.000 description 1
- LYLOGSAMXQCTRG-UHFFFAOYSA-N 3-ethyl-3-methylpentanoic acid Chemical compound CCC(C)(CC)CC(O)=O LYLOGSAMXQCTRG-UHFFFAOYSA-N 0.000 description 1
- NUPUDYKEEJNZRG-LBPRGKRZSA-N 3-ethynyl-5-[(2s)-1-methylpyrrolidin-2-yl]pyridine Chemical compound CN1CCC[C@H]1C1=CN=CC(C#C)=C1 NUPUDYKEEJNZRG-LBPRGKRZSA-N 0.000 description 1
- IBWPUTAKVGZXRB-UHFFFAOYSA-N 4-(1,3-benzodioxol-5-yl)butan-2-ylhydrazine Chemical compound NNC(C)CCC1=CC=C2OCOC2=C1 IBWPUTAKVGZXRB-UHFFFAOYSA-N 0.000 description 1
- 229940090248 4-hydroxybenzoic acid Drugs 0.000 description 1
- DFMRDHIALJTRDQ-UHFFFAOYSA-N 4-methyl-3-propan-2-ylpentanoic acid Chemical compound CC(C)C(C(C)C)CC(O)=O DFMRDHIALJTRDQ-UHFFFAOYSA-N 0.000 description 1
- KXSUAWAUCNFBQJ-UHFFFAOYSA-N 5-(2-ethyl-5-fluoro-1,3-dihydroinden-2-yl)-1h-imidazole Chemical compound C1C2=CC=C(F)C=C2CC1(CC)C1=CN=CN1 KXSUAWAUCNFBQJ-UHFFFAOYSA-N 0.000 description 1
- DHSSDEDRBUKTQY-UHFFFAOYSA-N 6-prop-2-enyl-4,5,7,8-tetrahydrothiazolo[4,5-d]azepin-2-amine Chemical compound C1CN(CC=C)CCC2=C1N=C(N)S2 DHSSDEDRBUKTQY-UHFFFAOYSA-N 0.000 description 1
- NQRIKTDKFHAOKC-UHFFFAOYSA-N 7-(4-methylpiperazin-1-yl)-3h-1,3-benzoxazol-2-one;hydrochloride Chemical compound Cl.C1CN(C)CCN1C1=CC=CC2=C1OC(=O)N2 NQRIKTDKFHAOKC-UHFFFAOYSA-N 0.000 description 1
- RGCLHVKCJVVHLN-QMMMGPOBSA-N Ac-DOPA-OH Natural products CC(=O)N[C@H](C(O)=O)CC1=CC=C(O)C(O)=C1 RGCLHVKCJVVHLN-QMMMGPOBSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 229940122578 Acetylcholine receptor agonist Drugs 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 206010001541 Akinesia Diseases 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 201000009487 Amblyopia Diseases 0.000 description 1
- 108050005273 Amino acid transporters Proteins 0.000 description 1
- 102000034263 Amino acid transporters Human genes 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 208000007415 Anhedonia Diseases 0.000 description 1
- 206010002942 Apathy Diseases 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- CEUORZQYGODEFX-UHFFFAOYSA-N Aripirazole Chemical compound ClC1=CC=CC(N2CCN(CCCCOC=3C=C4NC(=O)CCC4=CC=3)CC2)=C1Cl CEUORZQYGODEFX-UHFFFAOYSA-N 0.000 description 1
- 102100038238 Aromatic-L-amino-acid decarboxylase Human genes 0.000 description 1
- 206010003591 Ataxia Diseases 0.000 description 1
- 208000006096 Attention Deficit Disorder with Hyperactivity Diseases 0.000 description 1
- 208000036864 Attention deficit/hyperactivity disease Diseases 0.000 description 1
- 206010003805 Autism Diseases 0.000 description 1
- 208000020706 Autistic disease Diseases 0.000 description 1
- CYGODHVAJQTCBG-UHFFFAOYSA-N Bifeprunox Chemical compound C=12OC(=O)NC2=CC=CC=1N(CC1)CCN1CC(C=1)=CC=CC=1C1=CC=CC=C1 CYGODHVAJQTCBG-UHFFFAOYSA-N 0.000 description 1
- 208000020925 Bipolar disease Diseases 0.000 description 1
- 108030001720 Bontoxilysin Proteins 0.000 description 1
- 208000003174 Brain Neoplasms Diseases 0.000 description 1
- 208000009079 Bronchial Spasm Diseases 0.000 description 1
- 208000014181 Bronchial disease Diseases 0.000 description 1
- 206010006482 Bronchospasm Diseases 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- OXBZSWRVJMWDON-UHFFFAOYSA-N C=1C=CC=CC=1COC(=O)C(C)(C)CC=1C=CSC=1C(O)=O Chemical compound C=1C=CC=CC=1COC(=O)C(C)(C)CC=1C=CSC=1C(O)=O OXBZSWRVJMWDON-UHFFFAOYSA-N 0.000 description 1
- 0 CC(C)(C)CCC(Oc1cc(C[C@@](C(O)=O)N)ccc1OC(CCC(C)(C)*)=O)=O Chemical compound CC(C)(C)CCC(Oc1cc(C[C@@](C(O)=O)N)ccc1OC(CCC(C)(C)*)=O)=O 0.000 description 1
- MJFJHCRUAJPBHW-UHFFFAOYSA-N CC(C)(C)OC(NC(Cc(cc1)cc(O)c1O)C(OCc1ccccc1)=O)=O Chemical compound CC(C)(C)OC(NC(Cc(cc1)cc(O)c1O)C(OCc1ccccc1)=O)=O MJFJHCRUAJPBHW-UHFFFAOYSA-N 0.000 description 1
- DCEZZDIXWQSLQG-JIDHJSLPSA-N CC(C)(COC(=O)C1=CC=CC=C1OC)C(=O)OC2=C(C=C(C=C2)C[C@@H](C(=O)O)N)OC(=O)C(C)(C)COC(=O)C3=CC=CC=C3OC.Cl Chemical compound CC(C)(COC(=O)C1=CC=CC=C1OC)C(=O)OC2=C(C=C(C=C2)C[C@@H](C(=O)O)N)OC(=O)C(C)(C)COC(=O)C3=CC=CC=C3OC.Cl DCEZZDIXWQSLQG-JIDHJSLPSA-N 0.000 description 1
- MAMZIHHYBBJYOS-SFHVURJKSA-N CC(C)(COC(c1ccc[s]1)=O)C(Oc(ccc(C[C@@H](C(O)=O)N)c1)c1OC(C(C)(C)COC(c1ccc[s]1)=O)=O)=O Chemical compound CC(C)(COC(c1ccc[s]1)=O)C(Oc(ccc(C[C@@H](C(O)=O)N)c1)c1OC(C(C)(C)COC(c1ccc[s]1)=O)=O)=O MAMZIHHYBBJYOS-SFHVURJKSA-N 0.000 description 1
- KORNTPPJEAJQIU-KJXAQDMKSA-N Cabaser Chemical compound C1=CC([C@H]2C[C@H](CN(CC=C)[C@@H]2C2)C(=O)N(CCCN(C)C)C(=O)NCC)=C3C2=CNC3=C1 KORNTPPJEAJQIU-KJXAQDMKSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 206010007559 Cardiac failure congestive Diseases 0.000 description 1
- 102000006378 Catechol O-methyltransferase Human genes 0.000 description 1
- 108020002739 Catechol O-methyltransferase Proteins 0.000 description 1
- 101000957724 Catostomus commersonii Corticoliberin-1 Proteins 0.000 description 1
- 241000283153 Cetacea Species 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- GDLIGKIOYRNHDA-UHFFFAOYSA-N Clomipramine Chemical compound C1CC2=CC=C(Cl)C=C2N(CCCN(C)C)C2=CC=CC=C21 GDLIGKIOYRNHDA-UHFFFAOYSA-N 0.000 description 1
- RGHNJXZEOKUKBD-SQOUGZDYSA-M D-gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O RGHNJXZEOKUKBD-SQOUGZDYSA-M 0.000 description 1
- AEMOLEFTQBMNLQ-AQKNRBDQSA-N D-glucopyranuronic acid Chemical compound OC1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O AEMOLEFTQBMNLQ-AQKNRBDQSA-N 0.000 description 1
- 230000033616 DNA repair Effects 0.000 description 1
- 206010012239 Delusion Diseases 0.000 description 1
- 206010012289 Dementia Diseases 0.000 description 1
- 206010067889 Dementia with Lewy bodies Diseases 0.000 description 1
- 206010012335 Dependence Diseases 0.000 description 1
- 206010054089 Depressive symptom Diseases 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- BWLUMTFWVZZZND-UHFFFAOYSA-N Dibenzylamine Chemical class C=1C=CC=CC=1CNCC1=CC=CC=C1 BWLUMTFWVZZZND-UHFFFAOYSA-N 0.000 description 1
- XBPCUCUWBYBCDP-UHFFFAOYSA-N Dicyclohexylamine Chemical compound C1CCCCC1NC1CCCCC1 XBPCUCUWBYBCDP-UHFFFAOYSA-N 0.000 description 1
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 1
- 208000007590 Disorders of Excessive Somnolence Diseases 0.000 description 1
- 229940098778 Dopamine receptor agonist Drugs 0.000 description 1
- 208000030453 Drug-Related Side Effects and Adverse reaction Diseases 0.000 description 1
- 108010000912 Egg Proteins Proteins 0.000 description 1
- 102000002322 Egg Proteins Human genes 0.000 description 1
- 208000021473 Ejaculation disease Diseases 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- BRLQWZUYTZBJKN-UHFFFAOYSA-N Epichlorohydrin Chemical compound ClCC1CO1 BRLQWZUYTZBJKN-UHFFFAOYSA-N 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 208000027776 Extrapyramidal disease Diseases 0.000 description 1
- 239000001293 FEMA 3089 Substances 0.000 description 1
- 208000001640 Fibromyalgia Diseases 0.000 description 1
- 102100027346 GTP cyclohydrolase 1 Human genes 0.000 description 1
- 201000004311 Gilles de la Tourette syndrome Diseases 0.000 description 1
- 102000034615 Glial cell line-derived neurotrophic factor Human genes 0.000 description 1
- 108091010837 Glial cell line-derived neurotrophic factor Proteins 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- 206010019280 Heart failures Diseases 0.000 description 1
- 101000862581 Homo sapiens GTP cyclohydrolase 1 Proteins 0.000 description 1
- 208000023105 Huntington disease Diseases 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 229920001479 Hydroxyethyl methyl cellulose Polymers 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- 208000004044 Hypesthesia Diseases 0.000 description 1
- 208000030990 Impulse-control disease Diseases 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 206010022941 Iridocyclitis Diseases 0.000 description 1
- 206010022945 Iris adhesions Diseases 0.000 description 1
- PWWVAXIEGOYWEE-UHFFFAOYSA-N Isophenergan Chemical compound C1=CC=C2N(CC(C)N(C)C)C3=CC=CC=C3SC2=C1 PWWVAXIEGOYWEE-UHFFFAOYSA-N 0.000 description 1
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 239000004166 Lanolin Substances 0.000 description 1
- JMFKTFLARGGXCC-UHFFFAOYSA-N Lazabemide hydrochloride Chemical compound Cl.NCCNC(=O)C1=CC=C(Cl)C=N1 JMFKTFLARGGXCC-UHFFFAOYSA-N 0.000 description 1
- 208000009625 Lesch-Nyhan syndrome Diseases 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- DUGOZIWVEXMGBE-UHFFFAOYSA-N Methylphenidate Chemical compound C=1C=CC=CC=1C(C(=O)OC)C1CCCCN1 DUGOZIWVEXMGBE-UHFFFAOYSA-N 0.000 description 1
- UEQUQVLFIPOEMF-UHFFFAOYSA-N Mianserin Chemical compound C1C2=CC=CC=C2N2CCN(C)CC2C2=CC=CC=C21 UEQUQVLFIPOEMF-UHFFFAOYSA-N 0.000 description 1
- 229940121948 Muscarinic receptor antagonist Drugs 0.000 description 1
- 208000002740 Muscle Rigidity Diseases 0.000 description 1
- 206010052904 Musculoskeletal stiffness Diseases 0.000 description 1
- 235000021360 Myristic acid Nutrition 0.000 description 1
- TUNFSRHWOTWDNC-UHFFFAOYSA-N Myristic acid Natural products CCCCCCCCCCCCCC(O)=O TUNFSRHWOTWDNC-UHFFFAOYSA-N 0.000 description 1
- JTVPZMFULRWINT-UHFFFAOYSA-N N-[2-(diethylamino)ethyl]-2-methoxy-5-methylsulfonylbenzamide Chemical compound CCN(CC)CCNC(=O)C1=CC(S(C)(=O)=O)=CC=C1OC JTVPZMFULRWINT-UHFFFAOYSA-N 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- 229940127523 NMDA Receptor Antagonists Drugs 0.000 description 1
- 229940099433 NMDA receptor antagonist Drugs 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 241000772415 Neovison vison Species 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 208000008705 Nocturnal Myoclonus Syndrome Diseases 0.000 description 1
- PHVGLTMQBUFIQQ-UHFFFAOYSA-N Nortryptiline Chemical compound C1CC2=CC=CC=C2C(=CCCNC)C2=CC=CC=C21 PHVGLTMQBUFIQQ-UHFFFAOYSA-N 0.000 description 1
- ILUJQPXNXACGAN-UHFFFAOYSA-M O-methylsalicylate Chemical compound COC1=CC=CC=C1C([O-])=O ILUJQPXNXACGAN-UHFFFAOYSA-M 0.000 description 1
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 description 1
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 description 1
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 1
- 206010033557 Palpitations Diseases 0.000 description 1
- 208000007542 Paresis Diseases 0.000 description 1
- AHOUBRCZNHFOSL-UHFFFAOYSA-N Paroxetine hydrochloride Natural products C1=CC(F)=CC=C1C1C(COC=2C=C3OCOC3=CC=2)CNCC1 AHOUBRCZNHFOSL-UHFFFAOYSA-N 0.000 description 1
- 201000004316 Perry syndrome Diseases 0.000 description 1
- 201000005702 Pertussis Diseases 0.000 description 1
- 239000004264 Petrolatum Substances 0.000 description 1
- 201000011252 Phenylketonuria Diseases 0.000 description 1
- 208000005374 Poisoning Diseases 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- 206010036376 Postherpetic Neuralgia Diseases 0.000 description 1
- HDSBZMRLPLPFLQ-UHFFFAOYSA-N Propylene glycol alginate Chemical compound OC1C(O)C(OC)OC(C(O)=O)C1OC1C(O)C(O)C(C)C(C(=O)OCC(C)O)O1 HDSBZMRLPLPFLQ-UHFFFAOYSA-N 0.000 description 1
- YZCKVEUIGOORGS-IGMARMGPSA-N Protium Chemical compound [1H] YZCKVEUIGOORGS-IGMARMGPSA-N 0.000 description 1
- 239000007868 Raney catalyst Substances 0.000 description 1
- NPXOKRUENSOPAO-UHFFFAOYSA-N Raney nickel Chemical compound [Al].[Ni] NPXOKRUENSOPAO-UHFFFAOYSA-N 0.000 description 1
- 229910000564 Raney nickel Inorganic materials 0.000 description 1
- BKRGVLQUQGGVSM-KBXCAEBGSA-N Revanil Chemical compound C1=CC(C=2[C@H](N(C)C[C@H](C=2)NC(=O)N(CC)CC)C2)=C3C2=CNC3=C1 BKRGVLQUQGGVSM-KBXCAEBGSA-N 0.000 description 1
- FTALBRSUTCGOEG-UHFFFAOYSA-N Riluzole Chemical compound C1=C(OC(F)(F)F)C=C2SC(N)=NC2=C1 FTALBRSUTCGOEG-UHFFFAOYSA-N 0.000 description 1
- 229920002536 Scavenger resin Polymers 0.000 description 1
- 208000008039 Secondary Parkinson Disease Diseases 0.000 description 1
- 206010039897 Sedation Diseases 0.000 description 1
- 201000001880 Sexual dysfunction Diseases 0.000 description 1
- 208000005439 Sleep paralysis Diseases 0.000 description 1
- 208000022890 Sleep-related eating disease Diseases 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 1
- 206010041349 Somnolence Diseases 0.000 description 1
- AWMVMTVKBNGEAK-UHFFFAOYSA-N Styrene oxide Chemical compound C1OC1C1=CC=CC=C1 AWMVMTVKBNGEAK-UHFFFAOYSA-N 0.000 description 1
- 208000032851 Subarachnoid Hemorrhage Diseases 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- JACAAXNEHGBPOQ-LLVKDONJSA-N Talampanel Chemical compound C([C@H](N(N=1)C(C)=O)C)C2=CC=3OCOC=3C=C2C=1C1=CC=C(N)C=C1 JACAAXNEHGBPOQ-LLVKDONJSA-N 0.000 description 1
- 206010043118 Tardive Dyskinesia Diseases 0.000 description 1
- GUGOEEXESWIERI-UHFFFAOYSA-N Terfenadine Chemical compound C1=CC(C(C)(C)C)=CC=C1C(O)CCCN1CCC(C(O)(C=2C=CC=CC=2)C=2C=CC=CC=2)CC1 GUGOEEXESWIERI-UHFFFAOYSA-N 0.000 description 1
- JOAHPSVPXZTVEP-YXJHDRRASA-N Terguride Chemical compound C1=CC([C@H]2C[C@@H](CN(C)[C@@H]2C2)NC(=O)N(CC)CC)=C3C2=CNC3=C1 JOAHPSVPXZTVEP-YXJHDRRASA-N 0.000 description 1
- BHEOSNUKNHRBNM-UHFFFAOYSA-N Tetramethylsqualene Natural products CC(=C)C(C)CCC(=C)C(C)CCC(C)=CCCC=C(C)CCC(C)C(=C)CCC(C)C(C)=C BHEOSNUKNHRBNM-UHFFFAOYSA-N 0.000 description 1
- 208000000323 Tourette Syndrome Diseases 0.000 description 1
- 208000016620 Tourette disease Diseases 0.000 description 1
- HWHLPVGTWGOCJO-UHFFFAOYSA-N Trihexyphenidyl Chemical group C1CCCCC1C(C=1C=CC=CC=1)(O)CCN1CCCCC1 HWHLPVGTWGOCJO-UHFFFAOYSA-N 0.000 description 1
- 108010035075 Tyrosine decarboxylase Proteins 0.000 description 1
- 238000002083 X-ray spectrum Methods 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- QDLVYMYXOLGZOD-ZMBIFBSDSA-N [(2r)-3,4-dihydro-2h-chromen-2-yl]methyl-[[5-(4-fluorophenyl)pyridin-3-yl]methyl]azanium;chloride Chemical compound Cl.C1=CC(F)=CC=C1C1=CN=CC(CNC[C@@H]2OC3=CC=CC=C3CC2)=C1 QDLVYMYXOLGZOD-ZMBIFBSDSA-N 0.000 description 1
- LQYRDXFYYFLCGT-HNNXBMFYSA-N [4-[(2s)-2-amino-3-methoxy-3-oxopropyl]-2-(3,3-dimethylbutanoyloxy)phenyl] 3,3-dimethylbutanoate Chemical compound COC(=O)[C@@H](N)CC1=CC=C(OC(=O)CC(C)(C)C)C(OC(=O)CC(C)(C)C)=C1 LQYRDXFYYFLCGT-HNNXBMFYSA-N 0.000 description 1
- XAKBSHICSHRJCL-UHFFFAOYSA-N [CH2]C(=O)C1=CC=CC=C1 Chemical group [CH2]C(=O)C1=CC=CC=C1 XAKBSHICSHRJCL-UHFFFAOYSA-N 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 229940124532 absorption promoter Drugs 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 235000010419 agar Nutrition 0.000 description 1
- 239000005456 alcohol based solvent Substances 0.000 description 1
- 150000008044 alkali metal hydroxides Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 125000005819 alkenylalkoxy group Chemical group 0.000 description 1
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 1
- OBETXYAYXDNJHR-UHFFFAOYSA-N alpha-ethylcaproic acid Natural products CCCCC(CC)C(O)=O OBETXYAYXDNJHR-UHFFFAOYSA-N 0.000 description 1
- 229950001297 altinicline Drugs 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- WMGSQTMJHBYJMQ-UHFFFAOYSA-N aluminum;magnesium;silicate Chemical compound [Mg+2].[Al+3].[O-][Si]([O-])([O-])[O-] WMGSQTMJHBYJMQ-UHFFFAOYSA-N 0.000 description 1
- 229960003805 amantadine Drugs 0.000 description 1
- DKNWSYNQZKUICI-UHFFFAOYSA-N amantadine Chemical compound C1C(C2)CC3CC2CC1(N)C3 DKNWSYNQZKUICI-UHFFFAOYSA-N 0.000 description 1
- WOLHOYHSEKDWQH-UHFFFAOYSA-N amantadine hydrochloride Chemical compound [Cl-].C1C(C2)CC3CC2CC1([NH3+])C3 WOLHOYHSEKDWQH-UHFFFAOYSA-N 0.000 description 1
- 229960001280 amantadine hydrochloride Drugs 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 229960000836 amitriptyline Drugs 0.000 description 1
- KRMDCWKBEZIMAB-UHFFFAOYSA-N amitriptyline Chemical compound C1CC2=CC=CC=C2C(=CCCN(C)C)C2=CC=CC=C21 KRMDCWKBEZIMAB-UHFFFAOYSA-N 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 239000003708 ampul Substances 0.000 description 1
- 239000010775 animal oil Substances 0.000 description 1
- 201000004612 anterior uveitis Diseases 0.000 description 1
- 230000001078 anti-cholinergic effect Effects 0.000 description 1
- 230000001387 anti-histamine Effects 0.000 description 1
- 230000002421 anti-septic effect Effects 0.000 description 1
- 229960003965 antiepileptics Drugs 0.000 description 1
- 229940125715 antihistaminic agent Drugs 0.000 description 1
- 229940064004 antiseptic throat preparations Drugs 0.000 description 1
- 229960003990 apomorphine hydrochloride Drugs 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 229960004372 aripiprazole Drugs 0.000 description 1
- 230000037007 arousal Effects 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 208000015802 attention deficit-hyperactivity disease Diseases 0.000 description 1
- 238000005102 attenuated total reflection Methods 0.000 description 1
- 230000002567 autonomic effect Effects 0.000 description 1
- 210000004227 basal ganglia Anatomy 0.000 description 1
- 235000013871 bee wax Nutrition 0.000 description 1
- 239000012166 beeswax Substances 0.000 description 1
- 230000006399 behavior Effects 0.000 description 1
- 229940077388 benzenesulfonate Drugs 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- UYCBEQGAKVJJOE-UHFFFAOYSA-N benzyl 3-hydroxy-2,2-dimethylpropanoate 3-hydroxy-2,2-dimethylpropanoic acid Chemical compound CC(C(=O)O)(CO)C.OCC(C(=O)OCC1=CC=CC=C1)(C)C UYCBEQGAKVJJOE-UHFFFAOYSA-N 0.000 description 1
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 229950009087 bifeprunox Drugs 0.000 description 1
- 229960003003 biperiden Drugs 0.000 description 1
- YSXKPIUOCJLQIE-UHFFFAOYSA-N biperiden Chemical compound C1C(C=C2)CC2C1C(C=1C=CC=CC=1)(O)CCN1CCCCC1 YSXKPIUOCJLQIE-UHFFFAOYSA-N 0.000 description 1
- 125000000319 biphenyl-4-yl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 208000028683 bipolar I disease Diseases 0.000 description 1
- 230000008499 blood brain barrier function Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 210000001218 blood-brain barrier Anatomy 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 229940053031 botulinum toxin Drugs 0.000 description 1
- 229960002452 budipine Drugs 0.000 description 1
- QIHLUZAFSSMXHQ-UHFFFAOYSA-N budipine Chemical compound C1CN(C(C)(C)C)CCC1(C=1C=CC=CC=1)C1=CC=CC=C1 QIHLUZAFSSMXHQ-UHFFFAOYSA-N 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 235000019437 butane-1,3-diol Nutrition 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229960004596 cabergoline Drugs 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- 238000007707 calorimetry Methods 0.000 description 1
- 210000000692 cap cell Anatomy 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 125000002837 carbocyclic group Chemical group 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 229940084030 carboxymethylcellulose calcium Drugs 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 206010008118 cerebral infarction Diseases 0.000 description 1
- 229940082500 cetostearyl alcohol Drugs 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000000812 cholinergic antagonist Substances 0.000 description 1
- 238000013375 chromatographic separation Methods 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 229960004606 clomipramine Drugs 0.000 description 1
- 229960004170 clozapine Drugs 0.000 description 1
- QZUDBNBUXVUHMW-UHFFFAOYSA-N clozapine Chemical compound C1CN(C)CCN1C1=NC2=CC(Cl)=CC=C2NC2=CC=CC=C12 QZUDBNBUXVUHMW-UHFFFAOYSA-N 0.000 description 1
- 239000003245 coal Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000005094 computer simulation Methods 0.000 description 1
- 230000001054 cortical effect Effects 0.000 description 1
- 238000012926 crystallographic analysis Methods 0.000 description 1
- NISGSNTVMOOSJQ-UHFFFAOYSA-N cyclopentanamine Chemical class NC1CCCC1 NISGSNTVMOOSJQ-UHFFFAOYSA-N 0.000 description 1
- YMGUBTXCNDTFJI-UHFFFAOYSA-N cyclopropanecarboxylic acid Chemical compound OC(=O)C1CC1 YMGUBTXCNDTFJI-UHFFFAOYSA-N 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 231100000868 delusion Toxicity 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- MIOUTPXNKVBIFR-UHFFFAOYSA-J dicalcium 2-hydroxyacetate Chemical compound C(CO)(=O)[O-].[Ca+2].[Ca+2].C(CO)(=O)[O-].C(CO)(=O)[O-].C(CO)(=O)[O-] MIOUTPXNKVBIFR-UHFFFAOYSA-J 0.000 description 1
- YNHIGQDRGKUECZ-UHFFFAOYSA-N dichloropalladium;triphenylphosphanium Chemical compound Cl[Pd]Cl.C1=CC=CC=C1[PH+](C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1[PH+](C=1C=CC=CC=1)C1=CC=CC=C1 YNHIGQDRGKUECZ-UHFFFAOYSA-N 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- LVTYICIALWPMFW-UHFFFAOYSA-N diisopropanolamine Chemical compound CC(O)CNCC(C)O LVTYICIALWPMFW-UHFFFAOYSA-N 0.000 description 1
- 229940043276 diisopropanolamine Drugs 0.000 description 1
- FSBVERYRVPGNGG-UHFFFAOYSA-N dimagnesium dioxido-bis[[oxido(oxo)silyl]oxy]silane hydrate Chemical compound O.[Mg+2].[Mg+2].[O-][Si](=O)O[Si]([O-])([O-])O[Si]([O-])=O FSBVERYRVPGNGG-UHFFFAOYSA-N 0.000 description 1
- BADXJIPKFRBFOT-UHFFFAOYSA-N dimedone Chemical compound CC1(C)CC(=O)CC(=O)C1 BADXJIPKFRBFOT-UHFFFAOYSA-N 0.000 description 1
- 239000004205 dimethyl polysiloxane Substances 0.000 description 1
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 1
- 230000005750 disease progression Effects 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- IQQBRKLVEALROM-UHFFFAOYSA-N drinabant Chemical compound C=1C(F)=CC(F)=CC=1N(S(=O)(=O)C)C(C1)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 IQQBRKLVEALROM-UHFFFAOYSA-N 0.000 description 1
- QXWYKJLNLSIPIN-JGVFFNPUSA-N droxidopa Chemical compound OC(=O)[C@@H](N)[C@H](O)C1=CC=C(O)C(O)=C1 QXWYKJLNLSIPIN-JGVFFNPUSA-N 0.000 description 1
- 229960001104 droxidopa Drugs 0.000 description 1
- 206010013663 drug dependence Diseases 0.000 description 1
- 229960002866 duloxetine Drugs 0.000 description 1
- 230000008482 dysregulation Effects 0.000 description 1
- 235000013345 egg yolk Nutrition 0.000 description 1
- 210000002969 egg yolk Anatomy 0.000 description 1
- 239000012156 elution solvent Substances 0.000 description 1
- 206010014599 encephalitis Diseases 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 206010015037 epilepsy Diseases 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- NULMGOSOSZBEQL-QMMMGPOBSA-N etilevodopa Chemical compound CCOC(=O)[C@@H](N)CC1=CC=C(O)C(O)=C1 NULMGOSOSZBEQL-QMMMGPOBSA-N 0.000 description 1
- 229960001820 etilevodopa Drugs 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000008921 facial expression Effects 0.000 description 1
- 206010016256 fatigue Diseases 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 229950010445 fipamezole Drugs 0.000 description 1
- 229960002464 fluoxetine Drugs 0.000 description 1
- CJOFXWAVKWHTFT-XSFVSMFZSA-N fluvoxamine Chemical compound COCCCC\C(=N/OCCN)C1=CC=C(C(F)(F)F)C=C1 CJOFXWAVKWHTFT-XSFVSMFZSA-N 0.000 description 1
- 229960004038 fluvoxamine Drugs 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 238000005194 fractionation Methods 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 229960003692 gamma aminobutyric acid Drugs 0.000 description 1
- BTCSSZJGUNDROE-UHFFFAOYSA-N gamma-aminobutyric acid Chemical compound NCCCC(O)=O BTCSSZJGUNDROE-UHFFFAOYSA-N 0.000 description 1
- 210000004211 gastric acid Anatomy 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 239000003349 gelling agent Substances 0.000 description 1
- 229940050410 gluconate Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 229940097042 glucuronate Drugs 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- SCMLRESZJCKCTC-KMYQRJGFSA-N gtpl8173 Chemical compound C12=CC=C(CSCC)C=C2C2=C(CNC3=O)C3=C3C4=CC(CSCC)=CC=C4N4C3=C2N1[C@]1(C)[C@@](O)(C(=O)OC)C[C@H]4O1 SCMLRESZJCKCTC-KMYQRJGFSA-N 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- 229910000042 hydrogen bromide Inorganic materials 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 description 1
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 description 1
- 208000013403 hyperactivity Diseases 0.000 description 1
- 230000037315 hyperhidrosis Effects 0.000 description 1
- 208000034783 hypoesthesia Diseases 0.000 description 1
- 230000002218 hypoglycaemic effect Effects 0.000 description 1
- 229960004801 imipramine Drugs 0.000 description 1
- BCGWQEUPMDMJNV-UHFFFAOYSA-N imipramine Chemical compound C1CC2=CC=CC=C2N(CCCN(C)C)C2=CC=CC=C21 BCGWQEUPMDMJNV-UHFFFAOYSA-N 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 208000035231 inattentive type attention deficit hyperactivity disease Diseases 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 229940041682 inhalant solution Drugs 0.000 description 1
- 208000030603 inherited susceptibility to asthma Diseases 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- SUMDYPCJJOFFON-UHFFFAOYSA-N isethionic acid Chemical compound OCCS(O)(=O)=O SUMDYPCJJOFFON-UHFFFAOYSA-N 0.000 description 1
- IQVRBWUUXZMOPW-PKNBQFBNSA-N istradefylline Chemical compound CN1C=2C(=O)N(CC)C(=O)N(CC)C=2N=C1\C=C\C1=CC=C(OC)C(OC)=C1 IQVRBWUUXZMOPW-PKNBQFBNSA-N 0.000 description 1
- 229950009028 istradefylline Drugs 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 235000019388 lanolin Nutrition 0.000 description 1
- 229940039717 lanolin Drugs 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- 229950005862 lazabemide Drugs 0.000 description 1
- 210000004558 lewy body Anatomy 0.000 description 1
- 229940040145 liniment Drugs 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 229960003587 lisuride Drugs 0.000 description 1
- 229960000418 lisuride maleate Drugs 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 229960003511 macrogol Drugs 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 159000000003 magnesium salts Chemical class 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 201000005857 malignant hypertension Diseases 0.000 description 1
- 229960004090 maprotiline Drugs 0.000 description 1
- QSLMDECMDJKHMQ-GSXCWMCISA-N maprotiline Chemical compound C12=CC=CC=C2[C@@]2(CCCNC)C3=CC=CC=C3[C@@H]1CC2 QSLMDECMDJKHMQ-GSXCWMCISA-N 0.000 description 1
- 230000007721 medicinal effect Effects 0.000 description 1
- 229910000000 metal hydroxide Inorganic materials 0.000 description 1
- 150000004692 metal hydroxides Chemical class 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- JDBJJCWRXSVHOQ-UTONKHPSSA-N methanesulfonic acid;(1r)-n-prop-2-ynyl-2,3-dihydro-1h-inden-1-amine Chemical compound CS(O)(=O)=O.C1=CC=C2[C@H](NCC#C)CCC2=C1 JDBJJCWRXSVHOQ-UTONKHPSSA-N 0.000 description 1
- RAOHHYUBMJLHNC-UHFFFAOYSA-N methixene hydrochloride Chemical compound [H+].O.[Cl-].C1N(C)CCCC1CC1C2=CC=CC=C2SC2=CC=CC=C21 RAOHHYUBMJLHNC-UHFFFAOYSA-N 0.000 description 1
- UZKWTJUDCOPSNM-UHFFFAOYSA-N methoxybenzene Substances CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 229960001344 methylphenidate Drugs 0.000 description 1
- 229960005103 metixene Drugs 0.000 description 1
- MJFJKKXQDNNUJF-UHFFFAOYSA-N metixene Chemical compound C1N(C)CCCC1CC1C2=CC=CC=C2SC2=CC=CC=C21 MJFJKKXQDNNUJF-UHFFFAOYSA-N 0.000 description 1
- 229960003955 mianserin Drugs 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 229960001785 mirtazapine Drugs 0.000 description 1
- RONZAEMNMFQXRA-UHFFFAOYSA-N mirtazapine Chemical compound C1C2=CC=CN=C2N2CCN(C)CC2C2=CC=CC=C21 RONZAEMNMFQXRA-UHFFFAOYSA-N 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 229960001165 modafinil Drugs 0.000 description 1
- 229950010854 mofegiline Drugs 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- 239000003703 n methyl dextro aspartic acid receptor blocking agent Substances 0.000 description 1
- ACTNHJDHMQSOGL-UHFFFAOYSA-N n',n'-dibenzylethane-1,2-diamine Chemical class C=1C=CC=CC=1CN(CCN)CC1=CC=CC=C1 ACTNHJDHMQSOGL-UHFFFAOYSA-N 0.000 description 1
- JZXRLKWWVNUZRB-UHFFFAOYSA-N n-(2-aminoethyl)-5-chloropyridine-2-carboxamide Chemical compound NCCNC(=O)C1=CC=C(Cl)C=N1 JZXRLKWWVNUZRB-UHFFFAOYSA-N 0.000 description 1
- SQAZQLMBEHYFJA-BTJKTKAUSA-N n-(benzo[b][1]benzoxepin-5-ylmethyl)-n-methylprop-2-yn-1-amine;(z)-but-2-enedioic acid Chemical compound OC(=O)\C=C/C(O)=O.C#CCN(C)CC1=CC2=CC=CC=C2OC2=CC=CC=C12 SQAZQLMBEHYFJA-BTJKTKAUSA-N 0.000 description 1
- JXTPJDDICSTXJX-UHFFFAOYSA-N n-Triacontane Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCC JXTPJDDICSTXJX-UHFFFAOYSA-N 0.000 description 1
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 1
- 201000003631 narcolepsy Diseases 0.000 description 1
- 239000007923 nasal drop Substances 0.000 description 1
- 229940100662 nasal drops Drugs 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 230000004770 neurodegeneration Effects 0.000 description 1
- 208000015122 neurodegenerative disease Diseases 0.000 description 1
- 201000001119 neuropathy Diseases 0.000 description 1
- 230000007823 neuropathy Effects 0.000 description 1
- 230000000324 neuroprotective effect Effects 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 206010029446 nocturia Diseases 0.000 description 1
- 229960001158 nortriptyline Drugs 0.000 description 1
- 231100000862 numbness Toxicity 0.000 description 1
- 229940060184 oil ingredients Drugs 0.000 description 1
- 239000003883 ointment base Substances 0.000 description 1
- 229960005017 olanzapine Drugs 0.000 description 1
- KVWDHTXUZHCGIO-UHFFFAOYSA-N olanzapine Chemical compound C1CN(C)CCN1C1=NC2=CC=CC=C2NC2=C1C=C(C)S2 KVWDHTXUZHCGIO-UHFFFAOYSA-N 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 235000021313 oleic acid Nutrition 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 229960003104 ornithine Drugs 0.000 description 1
- 150000002924 oxiranes Chemical class 0.000 description 1
- MUMZUERVLWJKNR-UHFFFAOYSA-N oxoplatinum Chemical compound [Pt]=O MUMZUERVLWJKNR-UHFFFAOYSA-N 0.000 description 1
- NXJCBFBQEVOTOW-UHFFFAOYSA-L palladium(2+);dihydroxide Chemical compound O[Pd]O NXJCBFBQEVOTOW-UHFFFAOYSA-L 0.000 description 1
- YJVFFLUZDVXJQI-UHFFFAOYSA-L palladium(ii) acetate Chemical compound [Pd+2].CC([O-])=O.CC([O-])=O YJVFFLUZDVXJQI-UHFFFAOYSA-L 0.000 description 1
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 1
- 229960002296 paroxetine Drugs 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 229960001412 pentobarbital Drugs 0.000 description 1
- 229960004851 pergolide Drugs 0.000 description 1
- YEHCICAEULNIGD-MZMPZRCHSA-N pergolide Chemical compound C1=CC([C@H]2C[C@@H](CSC)CN([C@@H]2C2)CCC)=C3C2=CNC3=C1 YEHCICAEULNIGD-MZMPZRCHSA-N 0.000 description 1
- UWCVGPLTGZWHGS-ZORIOUSZSA-N pergolide mesylate Chemical compound CS(O)(=O)=O.C1=CC([C@H]2C[C@@H](CSC)CN([C@@H]2C2)CCC)=C3C2=CNC3=C1 UWCVGPLTGZWHGS-ZORIOUSZSA-N 0.000 description 1
- 229960001511 pergolide mesylate Drugs 0.000 description 1
- 208000023515 periodic limb movement disease Diseases 0.000 description 1
- 208000033808 peripheral neuropathy Diseases 0.000 description 1
- 230000002688 persistence Effects 0.000 description 1
- 229940066842 petrolatum Drugs 0.000 description 1
- UYWQUFXKFGHYNT-UHFFFAOYSA-N phenylmethyl ester of formic acid Natural products O=COCC1=CC=CC=C1 UYWQUFXKFGHYNT-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 229910000073 phosphorus hydride Inorganic materials 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229960003300 pimavanserin Drugs 0.000 description 1
- RGSULKHNAKTFIZ-CEAXSRTFSA-N pimavanserin tartrate Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1=CC(OCC(C)C)=CC=C1CNC(=O)N(C1CCN(C)CC1)CC1=CC=C(F)C=C1.C1=CC(OCC(C)C)=CC=C1CNC(=O)N(C1CCN(C)CC1)CC1=CC=C(F)C=C1 RGSULKHNAKTFIZ-CEAXSRTFSA-N 0.000 description 1
- 229960004310 piribedil Drugs 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229910003446 platinum oxide Inorganic materials 0.000 description 1
- 231100000572 poisoning Toxicity 0.000 description 1
- 230000000607 poisoning effect Effects 0.000 description 1
- 210000004508 polar body Anatomy 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 239000004584 polyacrylic acid Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 230000001144 postural effect Effects 0.000 description 1
- ZUFQCVZBBNZMKD-UHFFFAOYSA-M potassium 2-ethylhexanoate Chemical compound [K+].CCCCC(CC)C([O-])=O ZUFQCVZBBNZMKD-UHFFFAOYSA-M 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 235000011181 potassium carbonates Nutrition 0.000 description 1
- 229940086066 potassium hydrogencarbonate Drugs 0.000 description 1
- 229960003089 pramipexole Drugs 0.000 description 1
- FASDKYOPVNHBLU-ZETCQYMHSA-N pramipexole Chemical compound C1[C@@H](NCCC)CCC2=C1SC(N)=N2 FASDKYOPVNHBLU-ZETCQYMHSA-N 0.000 description 1
- 201000011461 pre-eclampsia Diseases 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 229960002262 profenamine Drugs 0.000 description 1
- CDOZDBSBBXSXLB-UHFFFAOYSA-N profenamine Chemical compound C1=CC=C2N(CC(C)N(CC)CC)C3=CC=CC=C3SC2=C1 CDOZDBSBBXSXLB-UHFFFAOYSA-N 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 229960003910 promethazine Drugs 0.000 description 1
- 235000010409 propane-1,2-diol alginate Nutrition 0.000 description 1
- 239000000770 propane-1,2-diol alginate Substances 0.000 description 1
- 125000001325 propanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 210000004129 prosencephalon Anatomy 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 208000002815 pulmonary hypertension Diseases 0.000 description 1
- 229960004431 quetiapine Drugs 0.000 description 1
- URKOMYMAXPYINW-UHFFFAOYSA-N quetiapine Chemical compound C1CN(CCOCCO)CCN1C1=NC2=CC=CC=C2SC2=CC=CC=C12 URKOMYMAXPYINW-UHFFFAOYSA-N 0.000 description 1
- 229960005197 quetiapine fumarate Drugs 0.000 description 1
- 230000036385 rapid eye movement (rem) sleep Effects 0.000 description 1
- 229960000245 rasagiline Drugs 0.000 description 1
- RUOKEQAAGRXIBM-GFCCVEGCSA-N rasagiline Chemical compound C1=CC=C2[C@H](NCC#C)CCC2=C1 RUOKEQAAGRXIBM-GFCCVEGCSA-N 0.000 description 1
- 229960001956 rasagiline mesylate Drugs 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 230000011514 reflex Effects 0.000 description 1
- 229950000659 remacemide Drugs 0.000 description 1
- 230000029058 respiratory gaseous exchange Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 229960004181 riluzole Drugs 0.000 description 1
- 229960001534 risperidone Drugs 0.000 description 1
- RAPZEAPATHNIPO-UHFFFAOYSA-N risperidone Chemical compound FC1=CC=C2C(C3CCN(CC3)CCC=3C(=O)N4CCCCC4=NC=3C)=NOC2=C1 RAPZEAPATHNIPO-UHFFFAOYSA-N 0.000 description 1
- 229960003179 rotigotine Drugs 0.000 description 1
- KFQYTPMOWPVWEJ-INIZCTEOSA-N rotigotine Chemical compound CCCN([C@@H]1CC2=CC=CC(O)=C2CC1)CCC1=CC=CS1 KFQYTPMOWPVWEJ-INIZCTEOSA-N 0.000 description 1
- NEMGRZFTLSKBAP-LBPRGKRZSA-N safinamide Chemical compound C1=CC(CN[C@@H](C)C(N)=O)=CC=C1OCC1=CC=CC(F)=C1 NEMGRZFTLSKBAP-LBPRGKRZSA-N 0.000 description 1
- 229950002652 safinamide Drugs 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- HKFMQJUJWSFOLY-OAQYLSRUSA-N sarizotan Chemical compound C1=CC(F)=CC=C1C1=CN=CC(CNC[C@@H]2OC3=CC=CC=C3CC2)=C1 HKFMQJUJWSFOLY-OAQYLSRUSA-N 0.000 description 1
- 229950007903 sarizotan Drugs 0.000 description 1
- XMXGGIWSFRIOKL-UHFFFAOYSA-N sch-63390 Chemical compound N1=CC=2C3=NC(C=4OC=CC=4)=NN3C(N)=NC=2N1CCCC1=CC=CC=C1 XMXGGIWSFRIOKL-UHFFFAOYSA-N 0.000 description 1
- MEZLKOACVSPNER-GFCCVEGCSA-N selegiline Chemical compound C#CCN(C)[C@H](C)CC1=CC=CC=C1 MEZLKOACVSPNER-GFCCVEGCSA-N 0.000 description 1
- 229960003946 selegiline Drugs 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- UQDJGEHQDNVPGU-UHFFFAOYSA-N serine phosphoethanolamine Chemical compound [NH3+]CCOP([O-])(=O)OCC([NH3+])C([O-])=O UQDJGEHQDNVPGU-UHFFFAOYSA-N 0.000 description 1
- 229960002073 sertraline Drugs 0.000 description 1
- VGKDLMBJGBXTGI-SJCJKPOMSA-N sertraline Chemical compound C1([C@@H]2CC[C@@H](C3=CC=CC=C32)NC)=CC=C(Cl)C(Cl)=C1 VGKDLMBJGBXTGI-SJCJKPOMSA-N 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 229950002275 setiptiline Drugs 0.000 description 1
- GVPIXRLYKVFFMK-UHFFFAOYSA-N setiptiline Chemical compound C12=CC=CC=C2CC2=CC=CC=C2C2=C1CN(C)CC2 GVPIXRLYKVFFMK-UHFFFAOYSA-N 0.000 description 1
- 231100000872 sexual dysfunction Toxicity 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 229920002545 silicone oil Polymers 0.000 description 1
- 208000022925 sleep disturbance Diseases 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- VYPDUQYOLCLEGS-UHFFFAOYSA-M sodium;2-ethylhexanoate Chemical compound [Na+].CCCCC(CC)C([O-])=O VYPDUQYOLCLEGS-UHFFFAOYSA-M 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 229940032094 squalane Drugs 0.000 description 1
- 229940031439 squalene Drugs 0.000 description 1
- TUHBEKDERLKLEC-UHFFFAOYSA-N squalene Natural products CC(=CCCC(=CCCC(=CCCC=C(/C)CCC=C(/C)CC=C(C)C)C)C)C TUHBEKDERLKLEC-UHFFFAOYSA-N 0.000 description 1
- 229940012831 stearyl alcohol Drugs 0.000 description 1
- 239000008223 sterile water Substances 0.000 description 1
- 239000008227 sterile water for injection Substances 0.000 description 1
- 208000011117 substance-related disease Diseases 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- RKZSNTNMEFVBDT-MRVPVSSYSA-N sumanirole Chemical compound C([C@H](C1)NC)C2=CC=CC3=C2N1C(=O)N3 RKZSNTNMEFVBDT-MRVPVSSYSA-N 0.000 description 1
- 229950011111 sumanirole Drugs 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000003447 supported reagent Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 230000009747 swallowing Effects 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 229950004608 talampanel Drugs 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 229950008418 talipexole Drugs 0.000 description 1
- 239000008399 tap water Substances 0.000 description 1
- 235000020679 tap water Nutrition 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 229960004558 terguride Drugs 0.000 description 1
- YZUAOVCUGSBIPP-UHFFFAOYSA-N tert-butyl N-[1-([1,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl]carbamate Chemical compound C1=CC=CN2C(C(NC(=O)OC(C)(C)C)C)=NN=C21 YZUAOVCUGSBIPP-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- DZLFLBLQUQXARW-UHFFFAOYSA-N tetrabutylammonium Chemical class CCCC[N+](CCCC)(CCCC)CCCC DZLFLBLQUQXARW-UHFFFAOYSA-N 0.000 description 1
- TUNFSRHWOTWDNC-HKGQFRNVSA-N tetradecanoic acid Chemical compound CCCCCCCCCCCCC[14C](O)=O TUNFSRHWOTWDNC-HKGQFRNVSA-N 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical class C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- OULAJFUGPPVRBK-UHFFFAOYSA-N tetratriacontyl alcohol Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO OULAJFUGPPVRBK-UHFFFAOYSA-N 0.000 description 1
- QERYCTSHXKAMIS-UHFFFAOYSA-M thiophene-2-carboxylate Chemical compound [O-]C(=O)C1=CC=CS1 QERYCTSHXKAMIS-UHFFFAOYSA-M 0.000 description 1
- QTWBEVAYYDZLQL-UHFFFAOYSA-N thiophene-3-carbonyl chloride Chemical compound ClC(=O)C=1C=CSC=1 QTWBEVAYYDZLQL-UHFFFAOYSA-N 0.000 description 1
- YNVOMSDITJMNET-UHFFFAOYSA-M thiophene-3-carboxylate Chemical compound [O-]C(=O)C=1C=CSC=1 YNVOMSDITJMNET-UHFFFAOYSA-M 0.000 description 1
- 229960005344 tiapride Drugs 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 238000002834 transmittance Methods 0.000 description 1
- DBGVGMSCBYYSLD-UHFFFAOYSA-N tributylstannane Chemical compound CCCC[SnH](CCCC)CCCC DBGVGMSCBYYSLD-UHFFFAOYSA-N 0.000 description 1
- 125000006000 trichloroethyl group Chemical group 0.000 description 1
- 125000004044 trifluoroacetyl group Chemical group FC(C(=O)*)(F)F 0.000 description 1
- 229960001032 trihexyphenidyl Drugs 0.000 description 1
- QDWJJTJNXAKQKD-UHFFFAOYSA-N trihexyphenidyl hydrochloride Chemical compound Cl.C1CCCCC1C(C=1C=CC=CC=1)(O)CCN1CCCCC1 QDWJJTJNXAKQKD-UHFFFAOYSA-N 0.000 description 1
- 229960004479 trihexyphenidyl hydrochloride Drugs 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 125000002221 trityl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C([*])(C1=C(C(=C(C(=C1[H])[H])[H])[H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 239000003871 white petrolatum Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
- 229960002911 zonisamide Drugs 0.000 description 1
- UBQNRHZMVUUOMG-UHFFFAOYSA-N zonisamide Chemical compound C1=CC=C2C(CS(=O)(=O)N)=NOC2=C1 UBQNRHZMVUUOMG-UHFFFAOYSA-N 0.000 description 1
- XJSMBWUHHJFJFV-VTIMJTGVSA-N α-dihydroergocryptine Chemical compound C([C@H]1N(C)C2)C([C]34)=CN=C4C=CC=C3[C@H]1C[C@H]2C(=O)N[C@@]1(C(C)C)C(=O)N2[C@@H](CC(C)C)C(=O)N3CCC[C@H]3[C@]2(O)O1 XJSMBWUHHJFJFV-VTIMJTGVSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/02—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C229/04—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C229/26—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having more than one amino group bound to the carbon skeleton, e.g. lysine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/02—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C229/34—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton containing six-membered aromatic rings
- C07C229/36—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton containing six-membered aromatic rings with at least one amino group and one carboxyl group bound to the same carbon atom of the carbon skeleton
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/12—Ketones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/12—Ketones
- A61K31/122—Ketones having the oxygen directly attached to a ring, e.g. quinones, vitamin K1, anthralin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/195—Carboxylic acids, e.g. valproic acid having an amino group
- A61K31/197—Carboxylic acids, e.g. valproic acid having an amino group the amino and the carboxyl groups being attached to the same acyclic carbon chain, e.g. gamma-aminobutyric acid [GABA], beta-alanine, epsilon-aminocaproic acid or pantothenic acid
- A61K31/198—Alpha-amino acids, e.g. alanine or edetic acid [EDTA]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/216—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acids having aromatic rings, e.g. benactizyne, clofibrate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/235—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids having an aromatic ring attached to a carboxyl group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/235—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids having an aromatic ring attached to a carboxyl group
- A61K31/24—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids having an aromatic ring attached to a carboxyl group having an amino or nitro group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/275—Nitriles; Isonitriles
- A61K31/277—Nitriles; Isonitriles having a ring, e.g. verapamil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4412—Non condensed pyridines; Hydrogenated derivatives thereof having oxo groups directly attached to the heterocyclic ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/13—Crystalline forms, e.g. polymorphs
Definitions
- the present invention relates to (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropatide) useful for the prevention and / or treatment of Parkinson's disease and / or Parkinson's syndrome.
- Parkinson's disease is one of the typical neurodegenerative diseases of the elderly caused by depaminergic degeneration / dropout, and is designated as one of the specific diseases (intractable diseases).
- the prevalence of Parkinson's disease is 100 to 300 people per 100,000 population, and clinical symptoms can be roughly classified into motor symptoms and non-motor symptoms.
- Motor symptoms include extrapyramidal symptoms such as tremor, akinesia, rigidity, and postural instability, including tremor.
- the three symptoms of immobility and muscle stiffness are known as the three main symptoms of Parkinson's disease.
- non-motor symptoms for example, digestive tract symptoms such as constipation, drooling, orthostatic hypotension, postprandial hypotension, hyperhidrosis, facial expression, dysuria, erectile dysfunction, etc., or emotion Symptoms such as apathy, loss of pleasure (anhedonia), depression, anxiety, and hallucinations appear. Furthermore, patients with Parkinson's disease often have cognitive impairment such as dementia.
- Parkinson's disease symptoms similar to Parkinson's disease may be caused by diseases such as cerebrovascular disorder, brain tumor, encephalitis, side effects of drugs, poisoning and the like. Those secondary to symptoms similar to Parkinson's disease are generally referred to as symptomatic parkinsonism, and may be referred to as Parkinson's syndrome together with essential parkinsonism such as Parkinson's disease.
- Dopamine replacement therapy is performed as a typical treatment for Parkinson's disease and / or Parkinson's syndrome.
- Levodopa L-dopa or L-3,4-dihydroxyphenylalanine
- Levodopa one of the drugs used in dopamine replacement therapy, was developed in the late 1960s, but is still the first in Parkinson's disease treatment today. Used as a single-choice drug.
- levodopa has pharmacokinetic problems and is one of the drugs that are difficult to control in the vicinity of the effective blood concentration.
- levodopa When levodopa is administered orally, levodopa is rapidly absorbed by the amino acid transporter present in the upper small intestine.
- the blood concentration of levodopa reaches its maximum in about 30 minutes to 2 hours after oral administration, but the blood half-life is as short as about 1 hour.
- the absorption process of levodopa is susceptible to the gastric residence time and the acidity of gastric acid and is unstable.
- AADC aromatic L-amino acid decarboxylase
- levodopa has a pharmacokinetic problem of unstable absorption, short blood duration, and low rate of transfer to the central nervous system (brain transfer rate). Some patients require 12 doses a day.
- levodopa also has a problem that its effectiveness gradually weakens after several years from the start of treatment. This is because as the disease progresses, the ability to store dopamine in the brain declines, and the blood concentration range (treatment area) of levodopa that provides an appropriate therapeutic effect is narrowed. Because of this undesired nature, even patients who are currently taking treatment with levodopa three times a day may have to take more times in a few years. visit.
- the brain transfer rate of levodopa is improved and the dose of levodopa is about 1/5 compared to the case of taking levodopa alone.
- the blood half-life of levodopa is still about 1 hour when DCI is added, and it does not improve at all in terms of maintaining the blood levodopa concentration.
- levodopa On the other hand, high-dose administration of levodopa is not practical in terms of side effects.
- the blood concentration of levodopa has a narrow effective range and a small deviation from the toxic range.
- side effects such as gastric symptoms, orthostatic hypotension, and palpitations occurred at the initial stage of treatment, and treatment started 2 After 3 months, serious central side effects such as dyskinesia and psychiatric symptoms occurred. Many patients who need levodopa cannot take a sufficient amount of levodopa because of these side effects.
- Levodopa's“ large number of doses ” cannot be improved by combining levodopa with an inhibitor of levodopa metabolism or by changing the administration method of levodopa itself.
- side effects such as dyskinesia that develop due to frequent exposure to levodopa exceeding the effective blood concentration are also a problem that is eagerly desired for patients who require levodopa.
- a prodrug body of levodopa itself for solving the problems caused by the blood kinetics of levodopa, such as “frequent administration” and “occurrence of side effects due to frequent administration”. Many reports have been reported.
- R 1C represents C1-8 alkyl, substituted C1-8 alkyl, C1-8 alkoxy and the like
- R 2C and R 3C each independently represent C1-8 alkyl, substituted C1-8 alkyl
- C1-8 R 4C and R 5C each independently represents a hydrogen atom, C1-8 alkyl, substituted C1-8 alkyl, etc.) (wherein the definitions of each group are excerpts) (See Patent Document 3);
- R 1D and R 2D each independently represent —C ( ⁇ O) R 5D or —C ( ⁇ O) OR 5D , or at least one of R 1D and R 2D represents a hydrogen atom.
- each of R 3D and R 4D independently represents a hydrogen atom, an optionally substituted C1-C6 alkyl, or C3- R 5D represents a hydrogen atom, an optionally substituted C 1-6 alkyl, or —CH 2 Q D ;
- Q D represents an optionally substituted 3-6 membered monocyclic group A compound represented by a carbocyclic or heterocyclic ring (note that the definitions of each group are excerpts) (see Patent Document 4);
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid, a salt thereof or a solvent thereof disclosed in the present invention Japanese products and their uses are not described in any prior art and are not derived from any prior art combination.
- An object of the present invention is to overcome the pharmacokinetic problems of levodopa in patients with Parkinson's disease and / or Parkinson's syndrome, and to provide an effective blood concentration of levodopa with a small number of administrations.
- the goal is to develop a reduced prodrug of levodopa.
- the present invention [1] (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid, a salt thereof or a solvate thereof; [2] (2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid, (2S) -2-amino-3 -(3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate, or (2S) -2-amino-3- (3,4-bis ( The compound of the above-mentioned [1], which is (2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride; [3] The above [3]
- [44] Contains (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid, a salt thereof or a solvate thereof
- a pharmaceutical composition comprising: [45] The pharmaceutical composition according to the above [44], which is a preventive and / or therapeutic agent for Parkinson's disease and / or Parkinson's syndrome; [46] (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid, a salt thereof or a solvate thereof;
- a pharmaceutical comprising a combination of an aromatic L-amino acid decarboxylase inhibitor and / or a catechol-O-methyltransferase inhibitor; [47] The medicament according to [46] above, wherein the aromatic L-amino acid decarboxylase inhibitor is carbidop
- the compound of the present invention may be abbreviated as “the compound of the present invention.”) Is a prodrug form of levodopa that can overcome the pharmacokinetic problems of levodopa and can provide an effective blood concentration of levodopa with a small number of administrations.
- patients with Parkinson's disease and / or Parkinson's syndrome who have been taking levodopa preferably Parkinson's disease and / or Patients with Parkinson's syndrome can maintain the effective blood concentration of levodopa for approximately 16 hours by administration twice a day (at most three times a day).
- the ability to obtain the same efficacy with twice a day administration also leads to improved medication compliance.
- the compound of the present invention is a prodrug that can give levodopa at an effective blood concentration in humans (effective plasma concentration: 0.4-1 ⁇ g / mL) for a long period of time, and has a flat blood concentration transition of levodopa. By doing so, the possibility of developing side effects such as dyskinesia and wear-off is minimized.
- the compound of the present invention is a drug with no concern for mutagenicity.
- some prodrugs of levodopa particularly some compounds having excellent persistence in blood, are compounds that are found to be mutagenic in a mutagenicity test method using mammalian cells. It turns out that there is not a little.
- the compound of the present invention does not have mutagenicity, patients can take it with peace of mind even when they have to take the drug for several years or decades, such as Parkinson's disease and / or Parkinson's syndrome. Can continue.
- the transition of plasma levodopa concentration when levodopa or a compound of the present invention is administered to dogs in combination with DCI is shown.
- the simulation of the levodopa concentration transition in human plasma obtained using the kinetic data of levodopa or the compound of the present invention in combination with canine DCI is shown.
- 6 shows changes in swirl movement when 6-hydroxydopamine medial forebrain bundle injection model rats were administered levodopa or a compound of the present invention in combination with DCI (benserazide).
- Crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid obtained in Example 10 (B-type crystal) ) Shows a differential scanning calorimetry chart.
- Crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid obtained in Example 10 (B-type crystal) ) Shows an infrared absorption spectrum chart.
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is represented by the following formula: It is a compound shown by these.
- isomers include all of them unless otherwise specified.
- isomers R-form, S-form, ⁇ -configuration, ⁇ -configuration, enantiomer, diastereomer
- optically active compounds having optical activity D-form, L-form, d-form, l-form
- polar bodies high-polar bodies, low-polar bodies
- equilibrium compounds for example, tautomers generated in amide bonds, etc.
- rotamers mixtures of these in any proportion, racemic mixtures
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is obtained by a known method using a corresponding salt. Is converted to The salt is preferably water-soluble.
- Suitable salts include acid addition salts (eg, hydrochloride, hydrobromide, hydroiodide, sulfate, phosphate, nitrate, and other inorganic acid salts such as acetate, lactate, Tartrate, benzoate, citrate, methanesulfonate, ethanesulfonate, benzenesulfonate, toluenesulfonate, p-toluenesulfonate (tosylate), isethionate, glucuronate , Organic acid salts such as gluconate, etc.), salts of acidic natural amino acids (eg, aspartic acid, glutamic acid, etc.), salts of alkali metals (eg, potassium, sodium, etc.), alkaline earth metals (eg, calcium, Magnesium salts, ammonium salts, tetramethylammonium salts, tetrabutylammonium salts, pharmaceutically acceptable
- (2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid and its salts can also be converted into solvates. It can. It is preferable that the solvate has low toxicity and water solubility. Suitable solvates include, for example, solvates with water and alcohol solvents (for example, ethanol).
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid, a salt thereof or a solvate thereof That is, the compound of the present invention is not limited by the crystalline form, and may be crystalline or non-crystalline. Or the mixture of the crystal
- the crystal form of the compound of the present invention is determined by, for example, a known analysis method used for crystallographic analysis, such as powder X-ray diffraction spectrum method, differential scanning calorimetry method, infrared absorption spectrum method, melting point measurement method and the like. It can be determined by measuring alone or in combination.
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid has at least two It has been confirmed that it can take a crystal form (in this specification, for the sake of convenience, it is described separately from an A-type crystal and a B-type crystal).
- a type crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is, for example, a powder X-ray diffraction spectrum In the analysis by the method, preferably having at least about 4.03, 7.21, 9.98, 10.72, 17.93, and 19.20 degrees 2 ⁇ peaks, preferably about 4.03, 7.21.
- Type 2 crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid include, for example, differential scanning calorimetry In the analysis according to Fig. 5, it is preferable to have an exothermic peak near 148.7 ° C and endothermic peaks near 184.7, 194.7, and 200.3 ° C, so that the differential scanning calorimetry chart shown in FIG. It is characterized by showing the same data.
- Type A crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid are obtained, for example, by infrared absorption spectroscopy.
- 1771, 1720, 1632, 1602, 1543, 1506, 1469, 1451, 1387, 1359, 1316, 1287, 1203, 1165, 1093, 1069, 1026, 957, 937, 898, 863, 802, 742, 710 , 687, 615, 557, 526, 490, 482, 452, 424, 416, and 408 cm ⁇ 1 preferably exhibit substantially the same data as the infrared absorption spectrum chart shown in FIG. It is characterized by that.
- Type 2 crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid are analyzed, for example, by a melting point measurement method. Is characterized by showing a melting point of about 177.0 to 181.9 ° C.
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid are, for example, powder X-ray diffraction spectra In analysis by the method, preferably having peaks of at least about 4.62 degrees 2 ⁇ , preferably peaks of about 4.62, 8.40, 9.54, 12.08, 15.38, and 18.16 degrees 2 ⁇ . More preferably, by showing the data described in Table 4 of Example 10 described later, particularly preferably by showing substantially the same data as the powder X-ray diffraction spectrum chart shown in FIG. Characterized.
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid include, for example, differential scanning calorimetry In the analysis according to Fig. 8, it is preferably substantially the same as the differential scanning calorimetry chart shown in Fig. 8 by having an exothermic peak around 183.3 ° C and endothermic peaks around 192.2 and 200.8 ° C. Characterized by showing data.
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid are obtained, for example, by infrared absorption spectroscopy.
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid are analyzed, for example, by a melting point measurement method. Is characterized by showing a melting point of about 174.7 to 179.0 ° C.
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate It has been confirmed that can take at least two crystal forms (in the present specification, for the sake of convenience, they are described separately from A-type crystals and B-type crystals).
- a type crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate is, for example, powder X
- powder X By having peaks of at least about 10.97, 11.58, 14.83, 16.36, 16.70, 19.42, 20.58, and 21.69 degrees 2 ⁇ in analysis by line diffraction spectroscopy. , Preferably about 5.15, 6.97, 7.46, 10.97, 11.58, 13.74, 14.83, 15.20, 16.10, 16.36, 16.70, 17.
- Example 6 More preferably by having peaks of 35, 18.30, 18.83, 19.42, 19.95, 20.58, 21.69, 22.63, 22.84, and 24.00 degrees 2 ⁇ .
- Example 6 described later By showing the data listed in Table 1, it is particularly preferably characterized by showing substantially the same data as the powder X-ray diffraction spectrum chart shown in FIG.
- Type A crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate include, for example, differential scanning In the analysis by the calorimetry method, it is characterized by having an endothermic peak around 135.95 ° C., preferably by showing substantially the same data as the differential scanning calorimetry chart shown in FIG.
- a type crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate has, for example, infrared absorption In the analysis by the spectral method, 1780, 1712, 1599, 1508, 1452, 1388, 1316, 1289, 1217, 1166, 1120, 1090, 1071, 1036, 1026, 1010, 957, 900, 864, 817, 742, 713, By having absorptions of 680, 622, 567, 550, 472, and 440 cm ⁇ 1 , it is preferably characterized by showing substantially the same data as the infrared absorption spectrum chart shown in FIG.
- Type 2 crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate for example, melting point measurement In the analysis by the method, it is characterized by showing a melting point of about 132.0-136.0 ° C.
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate are, for example, powder X At least about 0.011, 11.88, 13.87, 15.01, 15.87, 16.07, 17.81, 18.65, 19.17, and 22.11 in analysis by line diffraction spectroscopy. By having a peak at a degree 2 ⁇ , preferably about 4.04, 5.04, 5.54, 6.11, 6.60, 7.96, 8.62, 10.10, 10.32, 11.
- FIG. Is characterized by showing substantially the same data as the powder X-ray diffraction spectrum chart shown in FIG.
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate include, for example, differential scanning In the analysis by the calorimetric method, it is characterized by having an endothermic peak near 134.54 ° C., preferably by showing substantially the same data as the differential scanning calorimetry chart shown in FIG.
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate include, for example, infrared absorption In the analysis by the spectrum method, 1781, 1711, 1600, 1507, 1315, 1287, 1220, 1203, 1166, 1119, 1088, 1070, 1036, 1027, 1010, 944, 898, 863, 816, 713, 681, 617, By having absorptions of 567, 531, 517, 507, 484, 470, 452, 437, 421, and 413 cm ⁇ 1 , preferably exhibit substantially the same data as the infrared absorption spectrum chart shown in FIG. Is characterized by
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate for example, melting point measurement Characterized by showing a melting point of about 132.3-135.3 ° C. in the analysis by the method.
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride is It has been confirmed that it can take an amorphous crystalline form.
- the amorphous form of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride is, for example, powder X-ray diffraction It is characterized by the absence of a crystal-like peak in the analysis by the spectral method.
- the amorphous of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride is, for example, differential scanning calorimetry Analysis by the method is characterized by having an endothermic peak around 82.83 ° C., preferably by showing substantially the same data as the differential scanning calorimetry chart shown in FIG.
- the non-crystalline (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride is obtained, for example, by infrared absorption spectroscopy. 3409, 2992, 2944, 2865, 2629, 1970, 1774, 1718, 1655, 1601, 1585, 1508, 1470, 1452, 1428, 1388, 1369, 1317, 1290, 1258, 1204, 1168, 1125, Infrared absorption preferably shown in FIG.
- the amorphous form of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride is obtained, for example, by a melting point measurement method. In the analysis, it is characterized by showing a melting point of about 112.0-117.0 ° C.
- crystal form of the compound of the present invention is specified by the physicochemical properties disclosed in the present specification, but since each data can vary somewhat in its properties, it should not be interpreted strictly. Absent.
- the data obtained by the powder X-ray diffraction spectrum method for example, the relative intensity can vary somewhat depending on the nature of the crystal growth direction, particle size, measurement conditions, etc.
- the diffraction angle (2 ⁇ ) and the overall pattern are important.
- the full width at half maximum may be read from the powder X-ray diffraction spectrum chart and used in combination with the diffraction angle (2 ⁇ ), the overall pattern, and the relative intensity. .
- the data obtained by the differential scanning calorimetry method and infrared absorption spectrum method may vary somewhat depending on the measurement conditions, etc., due to its nature. is important.
- the overall pattern of at least one data obtained by powder X-ray diffraction spectroscopy, differential scanning calorimetry, and infrared absorption spectroscopy is similar to that of the crystalline forms of the compounds disclosed herein. All are included in the present invention.
- the powder X-ray diffraction spectrum chart shown in the drawings described later represents the diffraction angle (2 ⁇ ) (degrees) on the horizontal axis (2-Theta-Scale).
- the diffraction intensity is shown on the vertical axis (Lin (Counts)), and the differential scanning calorimetry chart shows time (minutes) or temperature (° C.) on the horizontal axis and heat flow on the vertical axis.
- the infrared absorption spectrum chart shows the wavelength on the horizontal axis (Wavenumber [cm -1 ]) and the transmittance on the vertical axis (% T).
- the compound of the present invention is a prodrug form of levodopa and undergoes metabolism in vivo, for example, the following intermediates (1) to (7): (1) (S) -2-Amino-3- (3-((2- (benzoyloxy) -2-methylpropanoyl) oxy) -4-((2-hydroxy-2-methylpropanoyl) oxy) Phenyl) propanoic acid; (2) (S) -2-Amino-3- (4-((2- (benzoyloxy) -2-methylpropanoyl) oxy) -3-((2-hydroxy-2-methylpropanoyl) oxy) Phenyl) propanoic acid; (3) (S) -2-Amino-3- (3-((2- (benzoyloxy) -2-methylpropanoyl) oxy) -4-hydroxyphenyl) propanoic acid; (4) (S) -2-Amino-3- (3,4-bis ((2-hydroxy-2-methylpropan
- the compound of the present invention is a prodrug that can give levodopa at an effective blood concentration (effective plasma concentration 0.4-1 ⁇ g / mL) for a long time in humans. By making it flat, the possibility of side effects such as dyskinesia and wear-off is minimized.
- this property of the compound of the present invention is due to the pharmacokinetic parameters of the compound of the present invention, for example, (1) changes in blood concentration of levodopa when the compound of the present invention is administered. Calculated from the “area under the blood concentration-time curve (AUC)” calculated from (2) and the change in blood concentration of levodopa when the compound of the present invention was administered, “6 hours after oral administration” It is brought about by a combination of “ratio of plasma concentration and maximum plasma concentration (Cmax) (Cmax / C6hr)”.
- AUC blood concentration-time curve
- the compound of the present invention is administered, for example, at a dose of 3 mg / kg as levodopa, for example, 0.6 ⁇ g ⁇ hr / mL or more, preferably 0.7 ⁇ g ⁇ hr / mL or more, more preferably 0.8 ⁇ g ⁇ hr / mL or more, particularly preferably 0.85 ⁇ g ⁇ hr / mL or more.
- the upper limit value of each of these ranges is disclosed as the AUC value of levodopa when a considerable amount of levodopa (here 3 mg / kg) is orally administered (0.96 ⁇ g ⁇ hr / mL as an example value). ).
- the compound of the present invention has a value of, for example, 100 or less, preferably 75 or less, more preferably 50 or less, particularly preferably 20 or less, particularly preferably 10 or less in Cmax / C6hr of (2) above. Show.
- the compound of the present invention shows a combination of the above preferred values of (1) AUC and (2) Cmax / C6hr in a kinetic test of dogs, so that “a prodrug that can give an effective blood concentration of levodopa for a long time in humans” And “a prodrug body in which the possibility of the occurrence of side effects such as dyskinesia and wear-off by giving a flat blood concentration of levodopa is reduced”.
- the compound of the present invention can be produced by the methods shown in the examples described later. Moreover, although it can manufacture also according to the method shown below or the method according to these, it is not limited to this.
- the compound of the present invention is levodopa: Can be produced according to the procedure of (A) protection of amino group ⁇ (B) protection of carboxyl group ⁇ (C) acylation of hydroxyl group ⁇ (D) deprotection of protecting group. Moreover, you may interchange the operation procedure of (A) and (B) if desired.
- (A) Protection of amino group The protection reaction of amino group is well known. For example, (1) a method using an acid halide, (2) a method using a mixed acid anhydride, (3) a method using a condensing agent, etc. Is mentioned.
- the method using an acid halide includes, for example, carboxylic acid in an organic solvent (chloroform, dichloromethane, diethyl ether, tetrahydrofuran, etc.) or without a solvent, and an acid halide agent (oxalyl chloride, thionyl chloride, etc.) and The reaction is carried out at 20 ° C.
- an organic solvent chloroform, dichloromethane, diethyl ether, tetrahydrofuran, etc.
- an acid halide agent oxalyl chloride, thionyl chloride, etc.
- the obtained acid halide can also be reacted with an amine at 0 to 40 ° C. in an organic solvent (dioxane, tetrahydrofuran, etc.) using an alkaline aqueous solution (aqueous sodium bicarbonate solution, sodium hydroxide solution, etc.).
- the method using a mixed acid anhydride is, for example, a method in which a carboxylic acid is used in an organic solvent (chloroform, dichloromethane, diethyl ether, tetrahydrofuran, etc.) or without a solvent, and a base (pyridine, triethylamine, dimethylaniline, dimethylaminopyridine, In the presence of diisopropylethylamine, etc., acid halides (pivaloyl chloride, tosyl chloride, mesyl chloride, etc.), acid derivatives (ethyl chloroformate, isobutyl chloroformate, etc.), or acid anhydride derivatives (di-tert-butyl-di)
- the reaction is carried out by reacting the resulting mixed acid anhydride with an amine at 0-40 ° C. in an organic solvent (chloroform, dichloromethane, diethyl ether, tetrahydrofuran
- a method using a condensing agent includes, for example, combining a carboxylic acid and an amine in an organic solvent (chloroform, dichloromethane, dimethylformamide, diethyl ether, tetrahydrofuran, etc.) or without a solvent (base, pyridine, triethylamine, dimethylaniline).
- organic solvent chloroform, dichloromethane, dimethylformamide, diethyl ether, tetrahydrofuran, etc.
- base pyridine, triethylamine, dimethylaniline
- amino-protecting groups include benzyloxycarbonyl (Cbz) group, tert-butoxycarbonyl (Boc) group, allyloxycarbonyl (Alloc) group, 1-methyl-1- (4-biphenyl) ethoxycarbonyl (Bpoc). ) Group, trifluoroacetyl group, 9-fluorenylmethoxycarbonyl (Fmoc) group, benzyl (Bn) group, p-methoxybenzyl group, benzyloxymethyl (BOM) group, 2- (trimethylsilyl) ethoxymethyl (SEM) Groups and the like.
- (B) Protection of carboxyl group The protection reaction of carboxyl group is well known. For example, (1) a method using an acid halide, (2) a method using a mixed acid anhydride, (3) a method using a condensing agent, etc. Is mentioned.
- the method using an acid halide includes, for example, carboxylic acid in an organic solvent (chloroform, dichloromethane, diethyl ether, tetrahydrofuran, etc.) or without a solvent, and an acid halide agent (oxalyl chloride, thionyl chloride, etc.) and The reaction is carried out at 20 ° C.
- an organic solvent chloroform, dichloromethane, diethyl ether, tetrahydrofuran, etc.
- an acid halide agent oxalyl chloride, thionyl chloride, etc.
- the resulting acid halide is used in the presence or absence of a base (pyridine, triethylamine, dimethylaniline, dimethylaminopyridine, diisopropylethylamine, etc.) or an alcohol and an organic solvent (chloroform, dichloromethane, In diethyl ether, tetrahydrofuran, etc.) at 0 ° C. to the reflux temperature of the solvent used.
- the reaction can be carried out by reacting with an acid halide at 0 to 40 ° C. in an organic solvent (dioxane, tetrahydrofuran, etc.) using an aqueous alkali solution (such as aqueous sodium bicarbonate or sodium hydroxide).
- the method using a mixed acid anhydride is, for example, a method in which a carboxylic acid is used in an organic solvent (chloroform, dichloromethane, diethyl ether, tetrahydrofuran, etc.) or without a solvent, and a base (pyridine, triethylamine, dimethylaniline, dimethylaminopyridine, Obtained by reacting with an acid halide (pivaloyl chloride, tosyl chloride, mesyl chloride, etc.) or an acid derivative (ethyl chloroformate, isobutyl chloroformate, etc.) at 0 to 40 ° C. in the presence of diisopropylethylamine, etc.
- the mixed acid anhydride is reacted with an alcohol at 0 to 40 ° C. in an organic solvent (chloroform, dichloromethane, diethyl ether, tetrahydrofuran, etc.).
- a method using a condensing agent includes, for example, using a carboxylic acid and an alcohol in an organic solvent (chloroform, dichloromethane, dimethylformamide, diethyl ether, tetrahydrofuran, etc.) or without a solvent in a base (pyridine, triethylamine, dimethylaniline).
- an organic solvent chloroform, dichloromethane, dimethylformamide, diethyl ether, tetrahydrofuran, etc.
- pyridine triethylamine, dimethylaniline
- Dimethylaminopyridine and the like in the presence or absence of a condensing agent (1,3-dicyclohexylcarbodiimide (DCC), 1-ethyl-3- [3- (dimethylamino) propyl] carbodiimide (EDC), 1, Using 1′-carbonyldiimidazole (CDI), 2-chloro-1-methylpyridinium iodine, 1-propanephosphonic acid cyclic anhydride (T3P), etc., 1-hydroxybenztriazole (HOBt) ) With or without 0 It is carried out by reacting at 40 ° C..
- DCC dicyclohexylcarbodiimide
- EDC 1-ethyl-3- [3- (dimethylamino) propyl] carbodiimide
- CDI 1′-carbonyldiimidazole
- T3P 1-propanephosphonic acid cyclic anhydride
- HOBt 1-hydroxybenztriazo
- Examples of the protecting group for the carboxyl group include methyl, ethyl, tert-butyl, trichloroethyl, benzyl (Bn), phenacyl, p-methoxybenzyl, trityl, 2-chlorotrityl and the like.
- the acylation of the hydroxyl group of the compound represented by The carboxylic acid represented by is, for example, in an organic solvent (chloroform, dichloromethane, diethyl ether, tetrahydrofuran, etc.) or without a solvent, with an acid halide agent (oxalyl chloride, thionyl chloride, etc.) at ⁇ 20 ° C. to reflux temperature.
- the acid halide obtained is reacted in an alcohol and an organic solvent (chloroform, dichloromethane, diethyl ether, tetrahydrofuran, acetonitrile, etc.).
- the reaction is carried out at a temperature of ⁇ 40 ° C.
- the reaction can be carried out by reacting with an acid halide at 0 to 40 ° C. in an organic solvent (dioxane, tetrahydrofuran, etc.) using an alkaline aqueous solution (aqueous sodium bicarbonate solution, sodium hydroxide solution, etc.).
- (D) Deprotection of protecting group The deprotection reaction of the protecting groups represented by R 101 and R 102 is known and can be carried out by the following method. For example, (1) Deprotection reaction by alkali hydrolysis, (2) Deprotection reaction under acidic conditions, (3) Deprotection reaction by hydrogenolysis, (4) Deprotection reaction of silyl group, (5) Deprotection reaction using metal, (6) Deprotection reaction using a metal complex and the like.
- the deprotection reaction by alkali hydrolysis is carried out, for example, in an organic solvent (methanol, tetrahydrofuran, dioxane, etc.), alkali metal hydroxide (sodium hydroxide, potassium hydroxide, lithium hydroxide, etc.), alkaline earth It is carried out at a temperature of 0 to 40 ° C. using a metal hydroxide (barium hydroxide, calcium hydroxide, etc.), carbonate (sodium carbonate, potassium carbonate, etc.), an aqueous solution thereof, or a mixture thereof.
- organic solvent methanol, tetrahydrofuran, dioxane, etc.
- alkali metal hydroxide sodium hydroxide, potassium hydroxide, lithium hydroxide, etc.
- alkaline earth It is carried out at a temperature of 0 to 40 ° C. using a metal hydroxide (barium hydroxide, calcium hydroxide, etc.), carbonate (sodium carbonate, potassium carbonate
- the deprotection reaction under acid conditions is carried out, for example, by using an organic acid (acetic acid, trifluoroacetic acid, methanesulfonic acid, p-toluenesulfonic acid) in an organic solvent (dichloromethane, chloroform, dioxane, ethyl acetate, anisole, etc.). Etc.), or an inorganic acid (hydrochloric acid, sulfuric acid, etc.), or a mixture thereof (hydrogen bromide / acetic acid, etc.) at a temperature of 0 to 100 ° C.
- organic acid acetic acid, trifluoroacetic acid, methanesulfonic acid, p-toluenesulfonic acid
- organic solvent diichloromethane, chloroform, dioxane, ethyl acetate, anisole, etc.
- Etc. an organic acid
- an inorganic acid hydroochloric acid, sulfuric acid, etc.
- Deprotection reaction by hydrogenolysis includes, for example, solvent (ether (tetrahydrofuran, dioxane, dimethoxyethane, diethyl ether, etc.), alcohol (methanol, ethanol, etc.), benzene (benzene, toluene, etc.), ketone In catalyst (acetone, methyl ethyl ketone, etc.), nitrile (acetonitrile, etc.), amide (dimethylformamide, etc.), water, ethyl acetate, acetic acid or a mixed solvent of two or more thereof, catalyst (palladium-carbon, palladium black) , Palladium hydroxide, platinum oxide, Raney nickel, etc.) in a hydrogen atmosphere under normal pressure or under pressure, or in the presence of ammonium formate, at a temperature of 0 to 200 ° C.
- solvent ether (tetrahydrofuran, dioxane, dimethoxyethane, diethyl
- the deprotection reaction of the silyl group is performed at a temperature of 0 to 40 ° C. using tetrabutylammonium fluoride in an organic solvent miscible with water (tetrahydrofuran, acetonitrile, etc.), for example.
- the deprotection reaction using a metal is carried out, for example, in an acidic solvent (acetic acid, a buffer solution of pH 4.2 to 7.2 or a mixture thereof with an organic solvent such as tetrahydrofuran) in the presence of powdered zinc. If necessary, it is performed at a temperature of 0 to 40 ° C. while applying ultrasonic waves.
- an acidic solvent acetic acid, a buffer solution of pH 4.2 to 7.2 or a mixture thereof with an organic solvent such as tetrahydrofuran
- the deprotection reaction using a metal complex is performed by, for example, trapping reagent (tributyltin hydride) in an organic solvent (dichloromethane, dimethylformamide, tetrahydrofuran, ethyl acetate, acetonitrile, dioxane, ethanol, etc.), water, or a mixed solvent thereof.
- trapping reagent tributyltin hydride
- organic solvent dichloromethane, dimethylformamide, tetrahydrofuran, ethyl acetate, acetonitrile, dioxane, ethanol, etc.
- Triethylsilane dimedone, morpholine, diethylamine, pyrrolidine, etc.
- organic acids acetic acid, formic acid, 2-ethylhexanoic acid, etc.
- organic acid salts sodium 2-ethylhexanoate, potassium 2-ethylhexanoate, etc.
- a phosphine-based reagent such as triphenylphosphine
- a metal complex tetrakistriphenylphosphine palladium (0), bis (triphenylphosphine) palladium dichloride (II), palladium acetate ( II), Tris chloride (Trife Nylphosphine) rhodium (I) and the like
- the intended compound of the present invention can be easily produced by properly using these deprotection reactions.
- the compound of the present invention is not limited by its crystal form. That is, the compound of the present invention may be crystalline or non-crystalline. Or the mixture of the crystal
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate is prepared by the method described below,
- the crystal can be produced by a method according to these methods or a method described in Examples.
- type 2 crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate The following method 1 to method 2: (Method 1) (2S) -3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) -2-((tert-butoxycarbonyl) amino) propanoic acid (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropaline) without isolation after deprotection using p-toluenesulfonic acid A method for producing a solvate of noyl) oxy) phenyl) propanoic acid tosylate salt, and heating and drying under reduced pressure to produce A-type crystals; (Method 2) (2S) -3- (3,4-bis ((
- Method 1 comprises (2S) -3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) -2-((tert-butoxycarbonyl) amino) propanoic acid.
- a solvate of noyl) oxy) phenyl) propanoic acid tosylate salt is produced and dried under heat under reduced pressure to produce A-type crystals.
- Deprotection of the tert-butoxycarbonyl group with p-toluenesulfonic acid is known, for example, an organic solvent (acetonitrile, ethanol, ethyl acetate, tert-butyl methyl ether, n-heptane, isopropyl ether, or two or more thereof.
- an organic solvent acetonitrile, ethanol, ethyl acetate, tert-butyl methyl ether, n-heptane, isopropyl ether, or two or more thereof.
- the reaction is carried out at a temperature of 0 ° C. to the boiling point of the solvent used, preferably 0 to 90 ° C.
- the solvate obtained by stirring the slurry at a temperature of 0 ° C. to the boiling point of the solvent used, preferably 0 to 90 ° C., without performing an isolation operation is preferably 30 to 100 ° C. under reduced pressure.
- the solvent used in Method 1 is preferably a mixed solvent of acetonitrile and tert-butyl methyl ether, a mixed solvent of ethyl acetate and tert-butyl methyl ether, or a mixed solvent of acetonitrile and water.
- Method 2 comprises (2S) -3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) -2-((tert-butoxycarbonyl) amino) propanoic acid. This is a method for producing an A-type crystal by taking out an amorphous substance after performing a deprotection reaction using p-toluenesulfonic acid, and performing amorphous recrystallization.
- Amorphous for recrystallization can be produced by concentrating the solvent used under reduced pressure after the deprotection reaction described in (Method 1) above.
- the obtained amorphous is in an organic solvent (such as acetonitrile, ethanol, ethyl acetate, tert-butyl methyl ether, n-heptane, isopropyl ether, or a mixed solvent of two or more thereof), or a mixed solvent thereof with water.
- the solvate obtained by recrystallization, or by stirring the slurry at a temperature of 0 ° C. to the boiling point of the solvent used, preferably 0 to 90 ° C. is preferably 30 to 100 ° C., preferably A-type crystals can be produced by heating and drying at 30 to 70 ° C.
- the solvent used for the deprotection reaction is preferably acetonitrile, ethyl acetate, a mixed solvent of acetonitrile and water, or a mixed solvent of ethyl acetate and water, and particularly preferably a mixed solvent of acetonitrile and water.
- the recrystallization operation was performed in a mixed solvent of ethyl acetate and tert-butyl methyl ether, a mixed solvent of acetonitrile and tert-butyl methyl ether, a mixed solvent of ethyl acetate and n-heptane, or a mixed solvent of ethanol and isopropyl ether. It is preferable to carry out, particularly in a mixed solvent of acetonitrile and tert-butyl methyl ether.
- B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate are Method 3 to Method 4: (Method 3) (2S) -3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) -2-((tert-butoxycarbonyl) amino) propanoic acid (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropaline) without isolation after deprotection using p-toluenesulfonic acid
- Method 3 comprises (2S) -3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) -2-((tert-butoxycarbonyl) amino) propanoic acid.
- a solvate of noyl) oxy) phenyl) propanoic acid tosylate salt is produced and dried under heat under reduced pressure to produce a B-type crystal.
- (2S) -3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) -2-((tert-butoxycarbonyl) amino) propanoic acid is 0.5 to 10 equivalents, preferably 0.5 to 3 equivalents, more preferably 0.5 to 1.5 equivalents of p-toluenesulfonic acid or its monohydrate in acetone or a mixed solvent of acetone and water In the presence of.
- the solvate obtained by stirring the slurry at a temperature of 0 to 60 ° C. without performing an isolation operation is dried by heating at 30 to 100 ° C., preferably 30 to 70 ° C. under reduced pressure.
- B-type crystals can be produced.
- acetone is preferable as a solvent used in the method 3.
- Method 4 is obtained by recrystallizing in an organic solvent (acetone or the like) or a mixed solvent of water and water after producing the A-type crystal by the method 1 or method 2 described above, or by stirring the slurry. This is a method for producing a B-type crystal by heating and drying the obtained solvate under reduced pressure.
- a solvate obtained by suspending a type crystal in acetone or a mixed solvent of acetone and water and stirring the slurry at 0 to 60 ° C. under reduced pressure is 25 to 100 ° C., preferably 25 to 70 ° C.
- a B-type crystal can be produced by drying with heating.
- acetone is preferable.
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride is converted to an inorganic base, Methods 5-7 using organic bases or organic epoxide compounds:
- (Method 5) (2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride is desalted with an inorganic base Method;
- (Method 6) (2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride is desalted with an organic base Method;
- (Method 7) (2S) -2-amino-3- (3,4-bis ((2- (benz
- Method 5 is a method of desalting (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride with an inorganic base. It is. Desalting reaction of amino acid hydrochloride with an inorganic base is known.
- an inorganic base such as acetonitrile, ethyl acetate, tetrahydrofuran, dioxane, methanol, ethanol, water, or a mixed solvent of two or more thereof
- a solvent such as acetonitrile, ethyl acetate, tetrahydrofuran, dioxane, methanol, ethanol, water, or a mixed solvent of two or more thereof
- the reaction is carried out at the boiling point of the solvent used, preferably at 0 to 40 ° C.
- Method 6 is a method of desalting (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride with an organic base. It is. Desalting reactions of amino acid hydrochlorides with organic bases are known.
- organic bases acetonitrile, ethyl acetate, tetrahydrofuran, dioxane, methanol, ethanol, water, or a mixed solvent of two or more thereof
- organic base Using triethylamine, diisopropylethylamine, N-methylpiperidine, etc.
- the reaction is preferably performed at 0 to 40 ° C.
- Method 7 desalinates (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride with an organic epoxide compound
- Desalting reaction of amino acid hydrochloride with organic epoxide compound is known, for example, organic epoxide in a solvent (acetonitrile, ethyl acetate, tetrahydrofuran, dioxane, methanol, ethanol, water, or a mixed solvent of two or more thereof).
- the reaction is carried out using 0.5 to 10 equivalents of a compound (epichlorohydrin, ethylene oxide, styrene oxide, etc.) at 0 ° C. to the boiling point of the solvent used, preferably 0 to 60 ° C.
- a compound epichlorohydrin, ethylene oxide, styrene oxide, etc.
- Method 8 desalinates (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate with an inorganic base.
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate as a raw material was a crystal. It may be non-crystalline. Or the mixture of the crystal
- the desalting reaction of amino acid tosylate with an inorganic base is known, for example, an inorganic base in a solvent (acetonitrile, ethyl acetate, tetrahydrofuran, dioxane, methanol, ethanol, water, or a mixed solvent of two or more thereof).
- a solvent acetonitrile, ethyl acetate, tetrahydrofuran, dioxane, methanol, ethanol, water, or a mixed solvent of two or more thereof.
- Sodium hydroxide, water cyclized potassium, barium hydroxide, etc. or an aqueous solution thereof is used in an amount of 0.5 to 10 equivalents, preferably 0.5 to 3 equivalents, more preferably 0.5 to 1.5 equivalents.
- the reaction is carried out at a boiling point of the solvent used, preferably 0 to 40 ° C.
- an organic base in a solvent acetonitrile, ethyl acetate, tetrahydrofuran, dioxane, methanol, ethanol, water, or a mixed solvent of two or more thereof
- a solvent acetonitrile, ethyl acetate, tetrahydrofuran, dioxane, methanol, ethanol, water, or a mixed solvent of two or more thereof
- Triethylamine, diisopropylethylamine, N-methylpiperidine, etc. is used in an amount of 0.5 to 10 equivalents, preferably 0.5 to 3 equivalents, more preferably 0.5 to 1.5 equivalents.
- the reaction is carried out at the boiling point, preferably 0 to 40 ° C.
- A-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid are Method 10-11: (Method 10) A crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is converted into the above-mentioned Method 5 to A method for producing type A crystals after isolation in 9 and without isolating the crude product; (Method 11) A crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is prepared by the above-described Method 5 to A method of isolating the crude product after the production in 9 and converting it into A-type crystals;
- Method 10 is a method in which a crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is converted into the above-described Method 5 to This is a method for producing the A-type crystal without isolating the crude product after the production in 9.
- the crude product produced by the above-mentioned methods 5 to 9, preferably method 7 or method 9 is used at a temperature of 0 to 80 ° C., preferably 0 to 50 ° C., more preferably 0 in a solvent (acetonitrile, etc.) used for the desalting reaction.
- a solvent acetonitrile, etc.
- A-type crystals can be produced by heat drying at 25-100 ° C., preferably 25-70 ° C. under reduced pressure.
- acetonitrile is preferable as a solvent used in the method 10.
- Method 11 is a method for preparing a crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid as described in the above methods 5 to 9. In this method, the crude product is isolated and converted into A-type crystals.
- the crude product produced by the above-mentioned methods 5 to 9 is isolated and then suspended in an organic solvent (acetonitrile or the like) and slurried at 0 to 80 ° C., preferably 0 to 50 ° C., more preferably 0 to 30 ° C. After stirring, the A-type crystal can be produced by heating and drying at 25 to 100 ° C., preferably 25 to 70 ° C. under reduced pressure.
- an organic solvent acetonitrile or the like
- a B-type crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is obtained by the following method 12- 13:
- Method 12 A crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is prepared by the above-described Method 5 to A method for producing a B-type crystal without isolating a crude product after the production in 9;
- Method 13 A crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is converted into the above-mentioned Method 5 to A method of isolating the crude product after the production in 9 and
- Method 12 is a method in which a crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is converted into the above-described Method 5 to 9 is a method for producing a B-type crystal without isolating a crude product.
- the crude product produced by the above-described methods 5 to 9 is prepared by using a solvent (such as acetonitrile) and water used for the desalting reaction in a ratio of 100: 1 to 100: 50, preferably 100: 1 to 100: 10, and more preferably.
- a solvent such as acetonitrile
- water used for the desalting reaction in a ratio of 100: 1 to 100: 50, preferably 100: 1 to 100: 10, and more preferably.
- recrystallization is performed by adding an organic solvent (acetonitrile, etc.), and the obtained crystals are reduced under reduced pressure.
- B-type crystals can be produced by heat drying at 25 to 100 ° C., preferably 25 to 70 ° C.
- acetonitrile is preferable.
- Method 13 is a method in which a crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid is converted into the above-described Method 5 to In this method, the crude product is isolated and converted into B-type crystals.
- the crude product produced by the above-mentioned methods 5 to 9 is isolated and then 100: 1 to 100: 50, preferably 100: 1 to 100: 10, more preferably 100: 5 to 100, of an organic solvent and water. : Dissolved in a mixed solvent of 10 and stirred at 0 to 80 ° C., preferably 0 to 60 ° C., and then recrystallized by adding an organic solvent (acetonitrile, etc.).
- B-type crystals can be produced by heating and drying at 100 ° C., preferably 25 to 70 ° C.
- acetonitrile is preferable.
- the B-type crystal can be obtained by subjecting the A-type crystal to the reaction of the method 12 or the method 13 described above.
- the A-type crystal can be obtained by subjecting the B-type crystal to the reaction of the method 10 or the method 11 described above.
- the compound of the present invention may be produced by known methods such as Comprehensive Organic Transformations: A Guide to Functional Group Preparations, Second Edition (Comprehensive® Organic Transformations). : A Guide to Functional Group Preparations, 2nd Edition (Richard C. Larock, John Wiley & Sons Inc, 1999)), etc., or a method that is partially modified from known methods can do.
- the compound used as a raw material is known per se or can be easily produced by a known method.
- reaction involving heating can be performed using a water bath, an oil bath, a sand bath, or a microwave, as will be apparent to those skilled in the art.
- a solid-phase-supported reagent supported on a polymer for example, polystyrene, polyacrylamide, polypropylene, polyethylene glycol, etc.
- a polymer for example, polystyrene, polyacrylamide, polypropylene, polyethylene glycol, etc.
- the reaction product is obtained by a usual purification means, for example, distillation under normal pressure or reduced pressure, high performance liquid chromatography using silica gel or magnesium silicate, thin layer chromatography, ion exchange resin, It can be purified by a scavenger resin, column chromatography, washing or recrystallization. Purification may be performed for each reaction or after completion of several reactions.
- the compound of the present invention Since the compound of the present invention has low toxicity, it can be safely used as a pharmaceutical product. In particular, since the compound of the present invention has no mutagenicity, patients can continue to take drugs with peace of mind even when they have to take drugs for several years or decades, such as Parkinson's disease and / or Parkinson's syndrome. be able to.
- the compound of the present invention is useful for the prevention and / or treatment of Parkinson's disease and / or Parkinson's syndrome.
- the prevention and / or treatment of Parkinson's disease and / or Parkinson's syndrome includes the meaning of words such as prevention and treatment of Parkinson's disease and Parkinson's syndrome, for example, prevention of onset of dyskinesia, reduction of severity of dyskinesia, Parkinson's Disease progression and Parkinson's syndrome progression inhibition (dopamine neuroprotective action), Parkinson's disease and non-motor symptoms of Parkinson's syndrome (eg, sleep disorders (eg, sleep disturbances, frequent sleep awakening, REM sleep behavior abnormalities, sleep disordered breathing, Arousal disorder, sleep phase regression syndrome, night wonder, nocturia, sleep paralysis, sleep-related eating disorder, etc.), psychiatric symptoms (eg, depressive symptoms, anxiety, dullness of emotion, loss of pleasure, hallucinations, delusions, impulse control disorder) , Dopamine dysregulation, etc.), autonomic
- levodopa is used as a therapeutic agent or is expected to be effective for other diseases such as Lewy body disease, depression, attention deficit disorder, integration Ataxia, manic depression, cognitive impairment, restless leg syndrome (restless legs syndrome), periodic limb movement disorder, tardive dyskinesia, Huntington's disease, Tourette syndrome, hypertension, addiction disorder, congestive heart failure, diabetic Pain associated with neuropathy, postherpetic neuralgia, fibromyalgia, autism, drug dependence, narcolepsy or excessive daytime sleepiness, dopa-responsive dystonia, plant condition, Perry syndrome, Segawa disease , Malignant syndrome, ejaculation disorder, gastric paresis, Lesch-Nyhan disease, amblyopia, pulmonary hypertension, cortical basal ganglia degeneration, phenylketonuria, panic Work, decreased libido, swallowing reflex disorder, also useful in the
- the Lewy body disease may be any disease in which Lewy bodies are observed pathologically, and includes, for example, dementia with Lewy bodies.
- the compound of the present invention is also useful for the prevention and / or treatment of diseases that are expected to be improved by dopamine stimulation and diseases that are induced by a decrease in noradrenaline.
- examples of diseases that are expected to be improved by dopamine stimulation include childhood hyperactivity, pregnancy toxemia, malignant hypertension, epilepsy and the like.
- diseases induced by a decrease in noradrenaline include, for example, orthostatic hypotension, subarachnoid hemorrhage, cerebral infarction, bronchospasm associated with bronchial asthma and pertussis, hypoglycemic symptoms due to insulin administration, iridocyclitis Examples include iris adhesions.
- the compound of the present invention is, for example, (1) supplemented and / or enhanced for its prevention, treatment and / or symptom improvement effect, (2) its kinetics / absorption improvement, dose reduction and / or (3) reduction of its side effects.
- Parkinson's disease and / or Parkinson's syndrome eg, levodopa or analogs thereof, aromatic L-amino acid decarboxylase inhibitors, catechol-O-methyltransferase inhibition Drugs, combination drugs for combined dopamine replacement therapy, dopamine receptor agonists, dopamine releaser, monoamine oxidase (MAO) inhibitor, dopamine uptake inhibitor, anticholinergic, nicotinic acetylcholine receptor Drugs, noradrenaline receptor agonists, ⁇ 2 receptor antagonists, serotonin receptor agonists, 5-HT1A selective / D2 receptor antagonist, adenosine receptor (A2A) antagonist, NMDA receptor antagonist, can
- levodopa or its analogs include levodopa, melevodopa, etilevodopa, and the like.
- aromatic L-amino acid decarboxylase inhibitor examples include benserazide, benserazide hydrochloride, carbidopa, carbidopa hydrate and the like.
- catechol-O-methyltransferase inhibitors examples include entacapone, tolcapone, nitecapone, BIA-3-202, CGP-28014 and the like.
- a combination drug for dopamine replacement therapy combining these, for example, a levodopa / benserazide combination preparation, a levodopa / carbidopa combination ⁇ preparation, a levodopa / carbidopa / entacapone combination (Levodopa / carbidopa / entacapone combination preparation), melevodopa / carbidopa combination preparation and the like.
- dopamine receptor agonists examples include cabergoline, pergolide, pergolide mesylate, bromocryptine, bromocryptine mesodate, pramipexole, and pramipexole hydrochloride.
- Japanese products pramipexole hydrochloride hydrate), ropinirole (ropinirole), ropinirole hydrochloride (ropinirole hydrochloride), talipexole, ⁇ -dihydroergocryptine, apomorphine, apomorphine hydrochloride (Sumanirole), terguride, bifeprunox, piribedil, lisuride, lisuride maleate, rotigotine DAR-0100, SLV-308, and the like.
- Examples of the dopamine-releasing drug include amantadine, amantadine hydrochloride, budipine, and the like.
- Monoamine oxidase (MAO) inhibitors include, for example, selegiline, safinamide, safrazine, deprnil, mofegiline, rasagiline, rasagiline mesylate, Examples include lazabemide and lazabemide hydrochloride.
- dopamine uptake inhibitors examples include modafinil, NS-2330, and the like.
- anticholinergic agents examples include trihexyphenidyl, trihexyphenidyl hydrochloride, biperiden, profenamine, metixene, metixene hydrochloride, and pyroheptin.
- pyroheptine pyroheptine hydrochloride
- mazaticol mazaticol hydrochloride
- mazaticol hydrochloride and the like.
- nicotinic acetylcholine receptor agonist examples include articline (altinicline), articline maleate (altinicline maleate) and the like.
- noradrenaline receptor agonists examples include droxidopa and the like.
- ⁇ 2 receptor antagonist examples include fipamezole and the like.
- Examples of serotonin receptor agonists include ACP-103.
- Examples of the 5-HT1A selective action / D2 receptor antagonist include sarizotan, sarizotan hydrochloride, and the like.
- adenosine receptor (A2A) antagonist examples include istradefylline, Sch-63390, VR-2006 and the like.
- NMDA receptor antagonists examples include remacemide, remacemide hydrochloride, and the like.
- CBD1 agonists examples include AVE-1625.
- AMPA receptor antagonists include, for example, talampanel and E-2007.
- glutamate release inhibitor examples include riluzole.
- antihistamines examples include promethazine and the like.
- antiepileptic drugs examples include zonisamide.
- Antidepressants include, for example, nortriptyline, imipramine, amitriptyline, clomipramine, desipramine, maprotiline, mianserin, setiptiline, fluxetine fluoxetine), fluvoxamine, sertraline, paroxetine, mirtazapine, duloxetine and the like.
- stimulants include methylphenidate.
- mixed lineage kinase inhibitors examples include CEP-1347.
- estrogen analogs examples include MITO-4509.
- antipsychotics examples include clozapine, quetiapine, quetiapine fumarate, olanzapine, risperidone, tiapride, and aripiprazole.
- neurotrophic factor examples include GDNF, PYM-50028, SR-57667, leteprinim potassium (leteprinimpotassium) and the like.
- neuroprotective drugs examples include TCH-346.
- immunophilin ligands examples include GPI-1485.
- gene therapy examples include CERE-120, NLX-XI, and P63.
- Examples of cell therapy include spheramine.
- the compound of the present invention when administered, it is an aromatic L-amino acid decarboxylase inhibitor carbidopa, carbidopa hydrate, benserazide, benserazide hydrochloride, and / or catechol-O-methyltransferase inhibitor.
- the sustained blood level transition of levodopa caused by the compound of the present invention is further prolonged, preferably 3 times a day, preferably
- the blood concentration of levodopa is 0.1-1.5 ⁇ g / mL, preferably 0.2-1.4 ⁇ g / mL, more preferably 0.3-1.2 ⁇ g / mL , Particularly preferably in the range of 0.4-1 ⁇ g / mL, which is considered to be the effective blood concentration of levodopa, is generally about 12 hours or more, preferably 14 hours or more, particularly preferably It can be maintained for six hours.
- the combination of the compound of the present invention and these other drugs may be administered in the form of a combination drug containing both components in one preparation, or may be administered in separate preparations.
- simultaneous administration and administration by time difference are included.
- administration with a time difference may be such that the compound of the present invention is administered first, the other drug may be administered later, the other drug may be administered first, and the compound of the present invention may be administered later.
- the administration method may be the same or different.
- the dose of the other drug can be appropriately selected based on the clinically used dose.
- the compounding ratio of the compound of the present invention and other drugs can be appropriately selected depending on the age and weight of the administration subject, administration method, administration time, target disease, symptom, combination and the like. For example, 0.01 to 100 parts by mass of another drug may be used with respect to 1 part by mass of the compound of the present invention. Two or more other drugs may be administered in combination at an appropriate ratio.
- the other drugs include not only those found so far but also those found in the future.
- the compound of the present invention or the concomitant agent of the compound of the present invention and another drug for the above purpose it is usually administered systemically or locally in an oral or parenteral form.
- the dose of the compound of the present invention varies depending on age, body weight, symptom, therapeutic effect, administration method, treatment time, etc., but is usually once to several times a day in the range of 100 mg to 3 g per adult. Administered orally or parenterally once or several times a day in the range of 10 mg to 1 g per adult, or continued intravenously in the range of 1 to 24 hours per day Be administered.
- a solid preparation or liquid for internal use for oral administration a sustained-release preparation for oral administration, or an injection for parenteral administration It is used as an agent, external preparation, inhalant, suppository, etc.
- solid preparations for internal use for oral administration include tablets, pills, capsules, powders and granules.
- Capsules include hard capsules and soft capsules.
- one or more active substances are left as they are, or excipients (eg, lactose, mannitol, glucose, microcrystalline cellulose, starch, etc.), binders (eg, hydroxypropylcellulose) , Polyvinyl pyrrolidone, magnesium aluminate metasilicate, etc.), disintegrant (eg, calcium calcium glycolate), lubricant (eg, magnesium stearate), stabilizer, solubilizer (eg, glutamic acid, aspartic acid) Etc.) and the like, and formulated into a conventional method.
- excipients eg, lactose, mannitol, glucose, microcrystalline cellulose, starch, etc.
- binders eg, hydroxypropylcellulose
- Polyvinyl pyrrolidone e.g, magnesium aluminate metasilicate, etc.
- disintegrant eg, calcium calcium glycolate
- lubricant eg, magnesium stea
- cover may coat
- a coating agent for example, sucrose, gelatin, hydroxypropyl cellulose, hydroxypropyl methylcellulose phthalate etc.
- cover may be coated with two or more layers.
- capsules of absorbable materials such as gelatin.
- Oral liquids for oral administration include pharmaceutically acceptable solutions, suspensions, emulsions, syrups and elixirs.
- a solution one or more active substances are dissolved, suspended or emulsified in a commonly used diluent (for example, purified water, ethanol, or a mixture thereof).
- this liquid agent may contain a wetting agent, a suspending agent, an emulsifier, a sweetening agent, a flavoring agent, a fragrance, a preservative or a buffering agent.
- sustained-release preparations for oral administration are also effective.
- the gel-forming substance used in these sustained-release preparations is a substance that swells with a solvent, the colloidal particles are connected to each other, take a three-dimensional network structure, and can form a jelly-like object that loses fluidity It is. On the formulation, it is mainly used as a binder, thickener and sustained release base.
- gum arabic for example, gum arabic, agar, polyvinylpyrrolidone, sodium alginate, propylene glycol alginate, carboxyvinyl polymer, carboxymethylcellulose, sodium carboxymethylcellulose, guar gum, gelatin, hydroxypropylmethylcellulose, hydroxypropylcellulose, polyvinyl alcohol, methylcellulose or hydroxyethylmethylcellulose Can be used.
- injections for parenteral administration are solutions, suspensions, emulsions and solid injections used by dissolving or suspending in a solvent at the time of use.
- An injection is used by dissolving, suspending or emulsifying one or more active substances in a solvent.
- the solvent for example, distilled water for injection, physiological saline, vegetable oil, propylene glycol, polyethylene glycol, alcohols such as ethanol, and combinations thereof are used.
- this injection contains a stabilizer, a solubilizing agent (for example, glutamic acid, aspartic acid, polysorbate 80 (registered trademark), etc.), a suspending agent, an emulsifying agent, a soothing agent, a buffering agent or a preservative. May be.
- a solubilizing agent for example, glutamic acid, aspartic acid, polysorbate 80 (registered trademark), etc.
- a suspending agent for example, glutamic acid, aspartic acid, polysorbate 80 (registere
- Examples of external dosage forms for parenteral administration include sprays, inhalants, sprays, aerosols, ointments, gels, creams, poultices, patches, liniments and nasal drops. Is included. These contain one or more active substances and are prepared by known methods or commonly used formulations.
- Sprays, inhalants, and sprays include buffers that provide isotonicity with stabilizers such as sodium bisulfite in addition to commonly used diluents, such as sodium chloride, sodium citrate or citric acid.
- stabilizers such as sodium bisulfite
- diluents such as sodium chloride, sodium citrate or citric acid.
- An isotonic agent such as A method for producing a spray is described in detail in, for example, US Pat. Nos. 2,868,691 and 30,955,355.
- Inhalants for parenteral administration include aerosols, inhalable powders or inhalable solutions, which are used by dissolving or suspending them in water or other suitable media at the time of use. It may be.
- inhalants are produced according to known methods.
- preservatives eg, benzalkonium chloride, parabens, etc.
- colorants e.g., sodium phosphate, sodium acetate, etc.
- buffering agents e.g, sodium phosphate, sodium acetate, etc.
- isotonic agents e.g, chloride
- thickeners for example, carboxyvinyl polymer, etc.
- absorption accelerators and the like, as appropriate.
- lubricants eg, stearic acid and its salts
- binders eg, starch, dextrin, etc.
- excipients eg, lactose, cellulose, etc.
- colorants eg, antiseptics
- an agent for example, benzalkonium chloride, paraben, etc. or an absorption accelerator is appropriately selected as necessary.
- a nebulizer for example, an atomizer, a nebulizer, etc.
- an inhalation administration device for a powder drug is usually used.
- the ointment is manufactured by a known or commonly used formulation. For example, it is prepared by mixing or melting one or more active substances in a base.
- the ointment base is selected from known or commonly used ones.
- higher fatty acid or higher fatty acid ester for example, adipic acid, myristic acid, palmitic acid, stearic acid, oleic acid, adipic acid ester, myristic acid ester, palmitic acid ester, stearic acid ester, oleic acid ester, etc.
- waxes E.g., beeswax, whale wax, ceresin, etc.
- surfactants e.g., polyoxyethylene alkyl ether phosphates, etc.
- higher alcohols e.g., cetanol, stearyl alcohol, cetostearyl alcohol, etc.
- silicone oils e.g., Dimethylpolysiloxane, etc.
- hydrocarbons eg, hydro
- Gel is manufactured by a known or commonly used formulation. For example, it is prepared by melting one or more active substances in a base.
- the gel base is selected from known or commonly used ones.
- lower alcohol eg, ethanol, isopropyl alcohol, etc.
- gelling agent eg, carboxymethylcellulose, hydroxyethylcellulose, hydroxypropylcellulose, ethylcellulose, etc.
- neutralizing agent eg, triethanolamine, diisopropanolamine, etc.
- surfactants for example, polyethylene glycol monostearate
- gums water
- absorption promoters and anti-rash agents are used alone or in admixture of two or more.
- a preservative, an antioxidant or a flavoring agent may be included.
- Creams are produced by known or commonly used formulations. For example, it is produced by melting or emulsifying one or more active substances in a base.
- the cream base is selected from known or commonly used ones. For example, higher fatty acid esters, lower alcohols, hydrocarbons, polyhydric alcohols (eg, propylene glycol, 1,3-butylene glycol, etc.), higher alcohols (eg, 2-hexyldecanol, cetanol, etc.), emulsifiers (eg, polyoxy (Ethylene alkyl ethers, fatty acid esters, etc.), water, absorption accelerators and anti-rash agents are used alone or in admixture of two or more. Furthermore, a preservative, an antioxidant or a flavoring agent may be included.
- the poultice is manufactured by a known or commonly used formulation. For example, it is produced by melting one or more active substances in a base material, and applying it as a kneaded product on a support.
- the poultice base is selected from known or commonly used ones.
- thickeners eg, polyacrylic acid, polyvinyl pyrrolidone, gum arabic, starch, gelatin, methyl cellulose, etc.
- wetting agents eg, urea, glycerin, propylene glycol, etc.
- fillers eg, kaolin, zinc oxide, Those selected from talc, calcium, magnesium, etc.
- solubilizers, tackifiers and anti-rash agents are used alone or in admixture of two or more.
- a preservative, an antioxidant or a flavoring agent may be included.
- the patch is manufactured by a known or commonly used formulation. For example, it is produced by melting one or more active substances in a base and spreading and coating them on a support.
- the base for patch is selected from known or commonly used ones. For example, those selected from polymer bases, fats and oils, higher fatty acids, tackifiers and anti-rash agents may be used alone or in admixture of two or more. Furthermore, a preservative, an antioxidant or a flavoring agent may be included.
- the liniment is produced by a known or commonly used formulation.
- one or more active substances may be selected from water, alcohol (eg, ethanol, polyethylene glycol, etc.), higher fatty acids, glycerin, soap, emulsifier, suspending agent, etc. alone or in two or more types It is prepared by suspending or emulsifying.
- a preservative, an antioxidant or a flavoring agent may be included.
- compositions for parenteral administration include suppositories for rectal administration and pessaries for intravaginal administration, which contain one or more active substances and are formulated by conventional methods.
- the solvent in parentheses shown in the location of separation by chromatography and in TLC indicates the elution solvent or developing solvent used, and the ratio indicates the volume ratio.
- the numerical value shown in the NMR part is the measured value of 1 H-NMR when using the described measuring solvent.
- Example 1 2- (Benzoyloxy) -2-methylpropanoic acid 2-hydroxyisobutyric acid (50 g) was dissolved in acetonitrile (480 mL). To this solution was added pyridine (78 mL) followed by benzoyl chloride (56 mL). The solution was stirred at room temperature for 40 minutes. 2N-hydrochloric acid (300 mL) was added to the reaction solution to acidify the solution, and the mixture was extracted with ethyl acetate (400 ⁇ 2 mL). The organic layers were combined and dried over magnesium sulfate. After magnesium sulfate was filtered off, the solvent was concentrated under reduced pressure.
- Example 2 (2S) -Benzyl 2-((tert-butoxycarbonyl) amino) -3- (3,4-dihydroxyphenyl) propanoate
- Purified water (30 mL) was added to (S) -3,4-dihydroxyphenylalanine (L-DOPA, 10.0 g) under an argon atmosphere to obtain a suspension.
- To this solution was added triethylamine (14.2 mL), followed by a solution of di-tert-butyl-dicarbonate (Boc 2 O, 13.3 g) in tetrahydrofuran (30 mL) at room temperature. The solution was stirred at room temperature for 14 hours.
- reaction solution was acidified with 2N hydrochloric acid (61 mL) under ice-cooling, and extracted with ethyl acetate (200 mL ⁇ 2). The organic layers were combined, washed with saturated brine (200 mL), and dried over magnesium sulfate. After magnesium sulfate was filtered off, the solvent was concentrated under reduced pressure. The residue was used in the next step without purification.
- TLC Rf value
- Example 3 (2S)-((4- (3- (benzyloxy) -2-((tert-butoxycarbonyl) amino) -3-oxopropyl) -1,2-phenylene) bis (oxy)) bis (2-methyl -1-oxopropane-2,1-diyl) dibenzoate
- Toluene (227 mL) was added to the compound produced in Example 1 (90.7 g) to give a suspension.
- N, N-dimethylformamide (0.8 mL) was added under an argon atmosphere, followed by thionyl chloride (38.2 mL). The reaction was stirred at 70 ° C. for 1 hour.
- the reaction solution was cooled to room temperature and concentrated under reduced pressure.
- Toluene (170 mL) was added to the residue and concentrated under reduced pressure. This operation was repeated twice to obtain acid chloride.
- Example 2 The compound prepared in Example 2 (76.7 g) was dissolved in acetonitrile (100 mL) under an argon atmosphere. To this solution, triethylamine (83 mL) was added under ice-cooling, and then the acid chloride prepared in the previous reaction was added over 15 minutes. This solution was stirred for 1 hour under ice cooling. To the reaction solution was added 10% aqueous sodium hydrogen carbonate solution (800 mL), and the mixture was extracted with ethyl acetate (800 mL ⁇ 2). The organic layers were combined, washed with saturated brine (800 mL), and dried over sodium sulfate. Sodium sulfate was filtered off and concentrated under reduced pressure.
- Example 4 (2S) -3- (3,4-Bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) -2-((tert-butoxycarbonyl) amino) propanoic acid
- ethanol 400 mL
- 10% palladium-carbon 50% water content, 14.6 g
- Ethyl acetate 400 mL was added to the reaction mixture, and the mixture was filtered through Celite (trade name). The filtrate was concentrated under reduced pressure.
- Example 5 (2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid hydrochloride
- 4N-hydrogen chloride-dioxane solution 500 mL was added. The solution was stirred at room temperature for 1 hour. The reaction solution was concentrated under reduced pressure to obtain the title compound (93.2 g, 94%) having the following physical property values.
- the obtained title compound was amorphous, and the melting point was about 112.0 to 117.0 ° C. (measured by the capillary method described in the Japanese Pharmacopoeia).
- Powder X-ray diffraction spectrum method [measurement conditions] Device: BRUKER DISCOVER with GADDS (C2) Target: Cu Filter: None Voltage: 40kV Current: 40 mA Exposure time: 180 sec [result] In the powder X-ray diffraction spectrum method using Cu—K ⁇ ray, no crystal-like peak was observed.
- Reference example 2 3- (Benzyloxy) -2,2-dimethyl-3-oxopropylthiophene-2-carboxylate
- the compound prepared in Reference Example 1 (2.0 g) was dissolved in dichloromethane (30 mL). Triethylamine (2.7 mL) was added to this solution, and then 2-thiophenecarboxylic acid chloride (1.5 mL) was added under ice cooling. This solution was stirred for 2 hours under ice cooling. A saturated aqueous sodium carbonate solution (30 mL) was added to the reaction solution, and the mixture was extracted with ethyl acetate (100 ⁇ 2 mL).
- Reference Example 3 2,2-Dimethyl-3-((thiophen-2-carbonyl) oxy) propanoic acid
- the compound (3.0 g) produced in Reference Example 2 was dissolved in ethanol (20 mL).
- 10% palladium-carbon (50% water content, 500 mg) was added under an argon atmosphere.
- the solution was stirred at room temperature for 1 hour under a hydrogen atmosphere.
- Ethyl acetate (20 mL) was added to the reaction solution, and filtered through Celite (trade name). The filtrate was concentrated under reduced pressure.
- Reference example 7 3- (Benzyloxy) -2,2-dimethyl-3-oxopropyl thiophene-3-carboxylate
- 3-thiophenecarboxylic acid chloride 2.1 g
- 2-thiophenecarboxylic acid chloride The title compound having a value (3.1 g, 100%) was obtained.
- Reference Example 8 2,2-Dimethyl-3-((thiophene-3-carbonyl) oxy) propanoic acid
- the same operation as in Reference Example 3 was carried out using the compound (3.1 g) prepared in Reference Example 7 instead of the compound prepared in Reference Example 2, and the title compound (354 mg, 16%) having the following physical property values was obtained. Obtained.
- TLC Rf value: 0.12 (ethyl acetate); NMR (CDCl 3 ): ⁇ 8.08-8.10 (m, 1H), 7.48-7.51 (m, 1H), 7.27-7.31 (m, 1H), 4.32 (s, 2H), 1.33 (s, 6H).
- Reference Example 12 3- (Benzyloxy) -2,2-dimethyl-3-oxopropyl 2-methoxybenzoate
- 2-methoxybenzoyl chloride 1.5 mL
- 2-thiophenecarboxylic acid chloride The title compound was obtained (1.8 g, 72%).
- Reference Example 13 3-((2-Methoxybenzoyl) oxy) -2,2-dimethylpropanoic acid
- the title compound (1.3 g, 91%) having the following physical property values was obtained by performing the same operation as in Reference Example 3 using the compound (1.8 g) prepared in Reference Example 12 instead of the compound prepared in Reference Example 2.
- Reference Example 36 3- (Benzyloxy) -2,2-dimethyl-3-oxopropyl benzoate
- the compound produced in Reference Example 1 (8 g) was dissolved in dichloromethane (40 mL). To this solution, triethylamine (8 mL) was added, followed by benzoyl chloride (5.4 mL) under ice cooling. The solution was stirred at room temperature for 4 hours. A saturated aqueous sodium carbonate solution (30 mL) was added to the reaction solution, and the mixture was extracted with dichloromethane (100 ⁇ 2 mL). The organic layers were combined, washed with saturated brine (30 mL), and dried over sodium sulfate.
- Reference Example 37 3- (Benzoyloxy) -2,2-dimethylpropanoic acid
- the same operation as in Reference Example 3 was carried out using the compound (12 g) produced in Reference Example 36 instead of the compound produced in Reference Example 2, and the title compound (5.7 g, 67%) having the following physical property values was obtained. Obtained.
- Example 6 Crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate (A-type crystal)
- acetonitrile 4.2 mL
- water 1.13 mL
- acetonitrile 37) of the compound prepared in Example 4 (8.32 g). .8 mL) solution was added.
- the solution was stirred at 70 ° C. for 2 hours.
- the reaction solution was cooled to room temperature, and tert-butyl methyl ether (254 mL) was added.
- A-type crystals of the title compound having the following physical properties (6.87 g, 92% ) was obtained as white crystals.
- the melting point of the crystal was about 132.0 to 136.0 ° C. (measured by the capillary method described in the Japanese Pharmacopoeia).
- Type A of the (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate salt thus obtained
- the powder X-ray diffraction spectrum chart of the crystal is shown in FIG. 10, the differential scanning calorimetry chart is shown in FIG. 11, and the infrared absorption spectrum chart is shown in FIG.
- Powder X-ray diffraction spectrum method [measurement conditions] Device: BRUKER DISCOVER with GADDS (C2) Target: Cu Filter: None Voltage: 40kV Current: 40 mA Exposure time: 180 sec [result] Table 1 shows the results of diffraction angle (2 ⁇ ) (degrees) and relative intensity (%) obtained by powder X-ray diffraction spectroscopy using Cu—K ⁇ rays. The relative intensity is obtained by calculating the height of each peak with the highest peak height (Lin (Counts)) being 100%.
- Example 7 Crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate (B-type crystal)
- the precipitated crystals were filtered and then dried under reduced pressure at 60 ° C. for 16 hours to obtain B-type crystals (7.1 g, 89%) of the title compound as white crystals.
- the melting point of the crystal was about 132.3 to 135.3 ° C. (measured by the capillary method described in the Japanese Pharmacopoeia).
- Form B of the (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate thus obtained
- the powder X-ray diffraction spectrum chart of the crystal is shown in FIG. 13, the differential scanning calorimetry chart is shown in FIG. 14, and the infrared absorption spectrum chart is shown in FIG.
- Powder X-ray diffraction spectrum method [measurement conditions] Device: BRUKER DISCOVER with GADDS (C2) Target: Cu Filter: None Voltage: 40kV Current: 40 mA Exposure time: 180 sec [result] Table 2 shows the results of diffraction angle (2 ⁇ ) (degrees) and relative intensity (%) obtained by the powder X-ray diffraction spectrum method using Cu—K ⁇ rays. The relative intensity is obtained by calculating the height of each peak with the highest peak height (Lin (Counts)) being 100%.
- Example 8 Crude product of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid
- (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid tosylate salt prepared in Example 7
- To a solution of crystals (167 g) in acetonitrile (2080 mL) and water (42 mL) a solution of triethylamine (21.4 g) in acetonitrile (420 mL) was added dropwise at room temperature.
- Example 9 Crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid (A-type crystal)
- a crude product (31 g) of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid prepared in Example 8 was obtained.
- the suspension was suspended in acetonitrile (470 mL) under an argon atmosphere, and the mixture was stirred at room temperature (internal temperature 23 to 24 ° C.) for 24 hours.
- the crystals were collected by filtration and washed with acetonitrile (94 mL). The crystals were dried under reduced pressure at 60 ° C. for 24 hours to give A-type crystals (31 g, 99%) of the title compound as white crystals.
- the melting point of the crystal was about 177.0 to 181.9 ° C. (measured by the capillary method described in the Japanese Pharmacopoeia).
- FIG. 4 shows an X-ray diffraction spectrum chart
- FIG. 5 shows a differential scanning calorimetry chart
- FIG. 6 shows an infrared absorption spectrum chart.
- Powder X-ray diffraction spectrum method [measurement conditions] Device: BRUKER DISCOVER with GADDS (C2) Target: Cu Filter: None Voltage: 40kV Current: 40 mA Exposure time: 180 sec [result] Table 3 shows the results of diffraction angle (2 ⁇ ) (degrees) and relative intensity (%) obtained by powder X-ray diffraction spectroscopy using Cu—K ⁇ rays. The relative intensity is obtained by calculating the height of each peak with the highest peak height (Lin (Counts)) being 100%.
- Example 10 Crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid (B-type crystal)
- a crude product (104 g) of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid prepared in Example 8 was obtained. Under argon atmosphere, it was dissolved by heating in acetonitrile (520 mL) and water (104 mL).
- Powder X-ray diffraction spectrum method [measurement conditions] Device: BRUKER DISCOVER with GADDS (C2) Target: Cu Filter: None Voltage: 40kV Current: 40 mA Exposure time: 180 sec [result] Table 4 shows the results of diffraction angle (2 ⁇ ) (degrees) and relative intensity (%) obtained by the powder X-ray diffraction spectrum method using Cu—K ⁇ rays. The relative intensity is obtained by calculating the height of each peak with the highest peak height (Lin (Counts)) being 100%.
- Example 11 Crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid (A-type crystal) Prepared in Example 10 instead of the crude (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid ( 2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid B-type crystals were used in the same manner as in Example 9. (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid was converted to an A-type crystal.
- Example 12 Crystal of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid (B-type crystal) Prepared in Example 9 instead of the crude (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid ( 2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid A-type crystals were used in the same manner as in Example 10. And converted to B-type crystals of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid.
- Biological Example 1 (1) Dog kinetic study In patients taking levodopa, the relationship between blood concentration of levodopa and side effects is gradually being clarified. For example, it is believed that dyskinesia is caused by frequent exposure of levodopa above effective blood levels, and wear-off is caused by levodopa that is below effective blood levels. To reduce the number of doses of levodopa and avoid side effects such as dyskinesia and wear-off in patients taking levodopa, it is necessary to keep the blood concentration of levodopa as long as possible within the therapeutic range. .
- the present inventors compared the compound of the present invention produced in Example 5 with levodopa for the purpose of clarifying that the compound of the present invention is a levodopa prodrug having such a flat blood concentration transition.
- Compound group used (compounds prepared in Reference Examples 6, 11, 16, 18, 19, 21-23, 25-35, and 40, and 3,3-dimethyl- described in WO 2009/022098 pamphlet)
- Compound X 4-((S) -2-amino-2-methoxycarbonyl-ethyl) butyric acid-2- (3,3-dimethyl-butyryloxy) -phenyl ester
- carboxyesterase plays the most important role among many enzymes in the process of producing levodopa after metabolism after being administered to a living body. It is thought that bears. Therefore, as an animal species to be subjected to a kinetic test, a dog whose organ distribution of carboxyesterase is most similar to that of a human was selected as an evaluation target.
- the compound of the present invention (Example 5) was found to have both AUC and Cmax / C6hr as compared with the compounds prepared in Reference Examples 6, 11, 16, 18, 19, 21, 23, and 25-35, and Compound X. It was confirmed that the compound is good, has a high rate of exposure to levodopa after being converted to levodopa, and gives a flat levodopa plasma concentration transition.
- the compounds used for comparison for example, the compounds produced in Reference Examples 6, 11, 19, 23, 27-29, 32 and 35, showed a value of 0.5 or more in AUC, but the value of Cmax / C6hr was about 15 at the smallest, and did not give a flat levodopa plasma concentration transition.
- Example 6 the compound of the present invention produced in Example 6, the compound of the present invention produced in Example 9, and the compound of the present invention produced in Example 10 were observed in plasma when orally administered to dogs.
- Example 6 The compound of the present invention produced in Example 6, the compound of the present invention produced in Example 9, and Example 10 in the same manner as the compound of the present invention (hydrochloride, non-crystalline) described in Example 5 shown in the preceding item (1).
- Any of the compounds of the present invention produced in (1) can be absorbed by oral administration, and the levodopa concentration in plasma observed after administration did not show any particular difference exceeding the range of variation. From the above results, it is clear that the compound of the present invention can be administered orally regardless of the salt or crystal form, and has the absorbability necessary for the development of drug efficacy and can be used uniformly as a pharmaceutical product. became.
- the compound of the present invention is a prodrug of levodopa, if it can be confirmed that levodopa is produced when administered to a living body, it is sure to show the same effectiveness as levodopa. In other words, a kinetic test for confirming that levodopa is produced by administration of the compound of the present invention can be regarded as equivalent to a pharmacological test for evaluating the effectiveness.
- the compound of the present invention produces levodopa after being administered to a living body. Therefore, the compound of the present invention can be administered by increasing or decreasing the dose as needed.
- the target efficacy can be obtained by a pharmacological test showing the effectiveness.
- Example 5 Each of the compound of the present invention and levodopa produced in Example 5 was weighed and then dissolved in 1 mg / mL in terms of levodopa using a medium to prepare a drug solution for oral administration. This drug solution was fasted from the previous day in advance, and a dog (beagle dog / male) administered with an effective amount (60 mg / kg) of carbidopa (AK Scientific Inc, 70566) was forcibly injected into the stomach using an oral sonde. Was administered. The dosage is 3 mg / kg for levodopa and 18.6 mg / kg for the compound of the present invention (Example 5) (6 mg / kg in terms of levodopa).
- 1 mL of blood was collected from the cephalic vein using a heparinized syringe at 15, 30 minutes, 1, 2, 4, 6 and 8 hours after administration of the drug solution.
- the collected blood was immediately centrifuged at 14500 rpm for 45 seconds in a table centrifuge, 2 volumes of 0.1125% formic acid-containing acetonitrile was added to 1 volume of plasma, stirred, and stored at ⁇ 20 ° C. until measurement.
- FIG. 1 shows the change in plasma concentration of levodopa when the compound of the present invention was administered to dogs.
- levodopa was administered in combination with carbidopa, which is a general-purpose DCI in clinical practice
- the levodopa blood concentration transition after administration of the compound of the present invention was compared.
- the plasma levodopa concentration after administration of levodopa was Although it decreased rapidly after reaching the maximum plasma concentration (Cmax) of 2.1 ⁇ g / mL in 15 minutes, the plasma levodopa concentration when the compound of the present invention was administered instead of levodopa was It gradually decreased after reaching Cmax of 1.0 ⁇ g / mL gradually in 4 hours.
- Cmax maximum plasma concentration
- the time (duration) in which the plasma levodopa concentration was in the range of 0.4-1 ⁇ g / mL was 0.6 hours when levodopa was administered. However, it was 4.3 hours when the compound of the present invention was administered.
- the compound of the present invention provides a sustained plasma concentration of levodopa after oral administration even in combination with carbidopa, which is widely used in clinical practice, and is effective in blood compared with the case of administering levodopa. It was found to increase the duration of the concentration by approximately 7 times. As described above, since the organ distribution of carboxyesterase is similar to that of humans, the compound of the present invention can give sustained plasma concentrations of levodopa even in humans based on the results of kinetic tests using these dogs. It was thought that.
- the levodopa and blood concentration concentrations of the compounds of the present invention obtained in the canine kinetic study are input to the kinetic analysis software Phoenix WinNonLin version 6.1 (Farsight) to fit the levodopa plasma concentration. By doing so, we built a prediction model to simulate levodopa blood dynamics.
- levodopa develops dyskinesia as a side effect when its plasma concentration is too high, and wear-off when it is too low. Therefore, if an intermediate plasma concentration that does not show these side effects can be maintained, it can be an excellent method for treating Parkinson's disease and / or Parkinson's syndrome.
- the compound of the present invention when taken at 600 mg (200 mg as levodopa amount), the time during which the plasma levodopa concentration was within the therapeutic range was calculated to be 7.8 hours. Therefore, the compound of the present invention gives a therapeutic range of levodopa for about 3.4 times longer than the existing levodopa formulation in humans, and in particular, when combined with DCI, it is administered twice a day for about 16 hours. It turns out that you can give levodopa.
- Biological Example 2 Mutagenicity test In the treatment of Parkinson's disease and / or Parkinson's syndrome, levodopa prodrugs may continue to be taken over long periods of years to decades. Therefore, the presence or absence of mutagenicity of the compounds of the present invention was evaluated by a mutagenicity test method using mammalian cells.
- Example 3 6-hydroxydopamine medial forebrain bundle injection model An experimental Parkinson's disease model using animals for the purpose of confirming that the compound of the present invention is metabolized to levodopa after oral administration and shows effectiveness against Parkinson's disease. -The efficacy of the compounds of the present invention in the hydroxydopamine medial forebrain bundle injection model was evaluated.
- 6-OHDA is placed inside according to Paxinos and Watson brain maps.
- the injection needle was left for 2 minutes or more in order to prevent the back flow of the liquid. Thereafter, the hole was closed with an instantaneous adhesive, and sutures and iodine tincture were disinfected.
- the compound of the present invention exhibited the same activity as when levodopa was administered after a certain period of time after oral administration. This is a result consistent with the characteristics of the compound of the present invention, in which the compound of the present invention does not exhibit levodopa-like activity by itself but exhibits metabolism and becomes levodopa.
- Formulation Example 1 (2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid 5 mg-containing tablet
- the following ingredients were mixed by a conventional method. After tableting, 10,000 tablets containing 5 mg of the active ingredient in one tablet were obtained.
- (2S) -2-Amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid 50 g ⁇ Carboxymethylcellulose calcium (disintegrant): 20 g ⁇ Magnesium stearate (lubricant): 10g ⁇ Microcrystalline cellulose: 920 g
- Formulation Example 2 Injection containing 20 mg of (2S) -2-amino-3- (3,4-bis ((2- (benzoyloxy) -2-methylpropanoyl) oxy) phenyl) propanoic acid After that, the solution was sterilized by a conventional method, filled into ampoules in 5 mL portions, and freeze-dried by a conventional method to obtain 10,000 ampoules containing 20 mg of active ingredient in one ampule.
- the compound of the present invention is a levodopa prodrug, a disease in which levodopa is used as a therapeutic agent such as Parkinson's disease and / or Parkinson's syndrome, or a disease that is expected to be effective, a disease that is expected to be improved by dopamine stimulation, or noradrenaline It is useful as a preventive and / or therapeutic agent for diseases induced by a decrease in
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Emergency Medicine (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Psychology (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
及び
[1](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物;
[2](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩、又は(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩である前記[1]記載の化合物;
[3](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の結晶である前記[2]記載の化合物;
[4]融点が、約177.0℃乃至約181.9℃である前記[3]記載の化合物;
[5]粉末X線回折スペクトルにおいて、少なくとも約4.03、7.21、9.98、10.72、17.93、及び19.20度2θにピークを有する前記[3]又は[4]記載の化合物;
[6]粉末X線回折スペクトルにおいて、約4.03、7.21、9.98、10.72、11.93、12.90、13.48、14.65、15.23、15.99、16.56、17.23、17.93、19.20、20.88、21.66、22.36、22.50、及び24.58度2θにピークを有する前記[3]乃至[5]のいずれか一項に記載の化合物;
[7]図4に示される粉末X線回折スペクトルチャートを特徴とする前記[3]乃至[6]のいずれか一項に記載の化合物;
[8]示差走査熱量測定において、約148.7℃の発熱ピーク、ならびに約184.7℃、約194.7℃、及び約200.3℃の吸熱ピークを有する前記[3]乃至[7]のいずれか一項に記載の化合物;
[9]図5に示される示差走査熱量測定チャートを特徴とする前記[3]乃至[8]のいずれか一項に記載の化合物;
[10]赤外線吸収スペクトルにおいて、1771、1720、1632、1602、1543、1506、1469、1451、1387、1359、1316、1287、1203、1165、1093、1069、1026、957、937、898、863、802、742、710、687、615、557、526、490、482、452、424、416、及び408cm-1に吸収を有する前記[3]乃至[9]のいずれか一項に記載の化合物;
[11]図6に示される赤外線吸収スペクトルチャートを特徴とする前記[3]乃至[10]のいずれか一項に記載の化合物;
[12]融点が、約174.7℃乃至約179.0℃である前記[3]記載の化合物;
[13]粉末X線回折スペクトルにおいて、少なくとも約4.62度2θにピークを有する前記[3]又は[12]記載の化合物;
[14]粉末X線回折スペクトルにおいて、約4.62、8.40、9.54、12.08、15.38、及び18.16度2θにピークを有する前記[3]、[12]、及び[13]のいずれか一項に記載の化合物;
[15]図7に示される粉末X線回折スペクトルチャートを特徴とする前記[3]、及び[12]乃至[14]のいずれか一項に記載の化合物;
[16]示差走査熱量測定において、約183.3℃の発熱ピーク、ならびに約192.2℃、及び約200.8℃の吸熱ピークを有する前記[3]、及び[12]乃至[15]のいずれか一項に記載の化合物;
[17]図8に示される示差走査熱量測定チャートを特徴とする前記[3]、及び[12]乃至[16]のいずれか一項に記載の化合物;
[18]赤外線吸収スペクトルにおいて、1771、1715、1608、1505、1469、1452、1411、1386、1368、1352、1315、1288、1256、1201、1166、1092、1070、1026、955、895、865、803、744、711、675、617、605、472、444、432、及び414cm-1に吸収を有する前記[3]、及び[12]乃至[17]のいずれか一項に記載の化合物;
[19]図9に示される赤外線吸収スペクトルチャートを特徴とする前記[3]、及び[12]乃至[18]のいずれか一項に記載の化合物;
[20](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩の結晶である前記[2]記載の化合物;
[21]融点が、約132.0℃乃至約136.0℃である前記[20]記載の化合物;
[22]粉末X線回折スペクトルにおいて、少なくとも約10.97、11.58、14.83、16.36、16.70、19.42、20.58、及び21.69度2θにピークを有する前記[20]又は[21]記載の化合物;
[23]粉末X線回折スペクトルにおいて、約5.15、6.97、7.46、10.97、11.58、13.74、14.83、15.20、16.10、16.36、16.70、17.35、18.30、18.83、19.42、19.95、20.58、21.69、22.63、22.84、及び24.00度2θにピークを有する前記[20]乃至[22]のいずれか一項に記載の化合物;
[24]図10に示される粉末X線回折スペクトルチャートを特徴とする前記[20]乃至[23]のいずれか一項に記載の化合物;
[25]示差走査熱量測定において、約135.95℃に吸熱ピークを有する前記[20]乃至[24]のいずれか一項に記載の化合物;
[26]図11に示される示差走査熱量測定チャートを特徴とする前記[20]乃至[25]のいずれか一項に記載の化合物;
[27]赤外線吸収スペクトルにおいて、1780、1712、1599、1508、1452、1388、1316、1289、1217、1166、1120、1090、1071、1036、1026、1010、957、900、864、817、742、713、680、622、567、550、472、及び440cm-1に吸収を有する前記[20]乃至[26]のいずれか一項に記載の化合物;
[28]図12に示される赤外線吸収スペクトルチャートを特徴とする前記[20]乃至[27]のいずれか一項に記載の化合物;
[29]融点が、約132.3℃乃至約135.3℃である前記[20]記載の化合物;
[30]粉末X線回折スペクトルにおいて、少なくとも約10.01、11.88、13.87、15.01、15.87、16.07、17.81、18.65、19.17、及び22.11度2θにピークを有する前記[20]又は[29]記載の化合物;
[31]粉末X線回折スペクトルにおいて、約4.04、5.04、5.54、6.11、6.60、7.96、8.62、10.01、10.32、11.88、12.88、13.87、15.01、15.87、16.07、16.74、17.17、17.81、18.65、19.17、19.72、20.27、20.93、21.67、22.11、22.56、23.11、23.47、及び24.21度2θにピークを有する前記[20]、[29]、及び[30]のいずれか一項に記載の化合物;
[32]図13に示される粉末X線回折スペクトルチャートを特徴とする前記[20]、及び[29]乃至[31]のいずれか一項に記載の化合物;
[33]示差走査熱量測定において、約134.54℃に吸熱ピークを有する前記[20]、及び[29]乃至[32]のいずれか一項に記載の化合物;
[34]図14に示される示差走査熱量測定チャートを特徴とする前記[20]、及び[29]乃至[33]のいずれか一項に記載の化合物;
[35]赤外線吸収スペクトルにおいて、1781、1711、1600、1507、1315、1287、1220、1203、1166、1119、1088、1070、1036、1027、1010、944、898、863、816、713、681、617、567、531、517、507、484、470、452、437、421、及び413cm-1に吸収を有する前記[20]、及び[29]乃至[34]のいずれか一項に記載の化合物;
[36]図15に示される赤外線吸収スペクトルチャートを特徴とする前記[20]、及び[29]乃至[35]のいずれか一項に記載の化合物;
[37](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩の非結晶である前記[2]記載の化合物;
[38]融点が、約112.0℃乃至約117.0℃である前記[37]記載の化合物;
[39]図16に示される粉末X線回折スペクトルチャートを特徴とする前記[37]又は[38]記載の化合物;
[40]示差走査熱量測定において、約82.83℃に吸熱ピークを有する前記[37]乃至[39]記載の化合物;
[41]図17に示される示差走査熱量測定チャートを特徴とする前記[37]乃至[40]のいずれか一項に記載の化合物;
[42]赤外線吸収スペクトルにおいて、3409、2992、2944、2865、2629、1970、1774、1718、1655、1601、1585、1508、1470、1452、1428、1388、1369、1317、1290、1258、1204、1168、1125、1093、1070、1026、1003、958、866、806、741、714、687、617、530、496、467、447、及び419cm-1に吸収を有する前記[37]乃至[41]のいずれか一項に記載の化合物;
[43]図18に示される赤外線吸収スペクトルチャートを特徴とする前記[37]乃至[42]のいずれか一項に記載の化合物;
[44](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物を含有してなる医薬組成物;
[45]パーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療剤である前記[44]記載の医薬組成物;
[46](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物と、芳香族L-アミノ酸デカルボキシラーゼ阻害薬及び/又はカテコール-O-メチル基転移酵素阻害薬とを組み合わせてなる医薬;
[47]芳香族L-アミノ酸デカルボキシラーゼ阻害薬が、カルビドパ水和物、又は塩酸ベンセラジドである前記[46]記載の医薬;
[48]カテコール-O-メチル基転移酵素阻害薬が、エンタカポン、トルカポン、ニテカポン、BIA-3-202、又はCGP-28014である前記[46]記載の医薬;
[49]配合剤である前記[46]乃至[48]のいずれか一項に記載の医薬;
[50](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物の有効量を哺乳動物に投与することを特徴とするパーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療方法;
[51]更に、芳香族L-アミノ酸デカルボキシラーゼ阻害薬及び/又はカテコール-O-メチル基転移酵素阻害薬の有効量を投与する前記[50]記載の予防及び/又は治療方法;
[52]パーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療のための(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物;
[53]パーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療に際し、芳香族L-アミノ酸デカルボキシラーゼ阻害薬及び/又はカテコール-O-メチル基転移酵素阻害薬と組合わせて用いるための(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物;
[54]パーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療剤の製造のための(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物の使用;
[55](2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物を含有してなるパーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療剤;
[56](S)-((4-(3-(ベンジルオキシ)-2-((tert-ブトキシカルボニル)アミノ)-3-オキソプロピル)-1,2-フェニレン)ビス(オキシ))ビス(2-メチル-1-オキソプロパン-2,1-ジイル) ジベンゾアート又はその塩;及び
[57](S)-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸又はその塩等に関する。
(1)(S)-2-アミノ-3-(3-((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)-4-((2-ヒドロキシ-2-メチルプロパノイル)オキシ)フェニル)プロパン酸;
(2)(S)-2-アミノ-3-(4-((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)-3-((2-ヒドロキシ-2-メチルプロパノイル)オキシ)フェニル)プロパン酸;
(3)(S)-2-アミノ-3-(3-((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)-4-ヒドロキシフェニル)プロパン酸;
(4)(S)-2-アミノ-3-(3,4-ビス((2-ヒドロキシ-2-メチルプロパノイル)オキシ)フェニル)プロパン酸;
(5)(S)-2-アミノ-3-(4-((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)-3-ヒドロキシフェニル)プロパン酸;
(6)(S)-2-アミノ-3-(4-ヒドロキシ-3-((2-ヒドロキシ-2-メチルプロパノイル)オキシ)フェニル)プロパン酸;及び
(7)(S)-2-アミノ-3-(3-ヒドロキシ-4-((2-ヒドロキシ-2-メチルプロパノイル)オキシ)フェニル)プロパン酸;
の全て若しくは幾つかを経て、レボドパを生成する。
本発明化合物は、後述の実施例に示す方法で製造することができる。また、以下に示す方法、若しくはこれらに準ずる方法に従っても製造することができるが、これに限定されるものではない。
アミノ基の保護反応はよく知られており、例えば、(1)酸ハライドを用いる方法、(2)混合酸無水物を用いる方法、(3)縮合剤を用いる方法等が挙げられる。
(1)酸ハライドを用いる方法は、例えば、カルボン酸を有機溶媒(クロロホルム、ジクロロメタン、ジエチルエーテル、テトラヒドロフラン等)中、又は無溶媒で、酸ハライド化剤(オキザリルクロリド、チオニルクロリド等)と-20℃~還流温度で反応させ、得られた酸ハライドを塩基(ピリジン、トリエチルアミン、ジメチルアニリン、ジメチルアミノピリジン、ジイソプロピルエチルアミン等)の存在下、アミンと有機溶媒(クロロホルム、ジクロロメタン、ジエチルエーテル、テトラヒドロフラン等)中、0~40℃の温度で反応させることにより行われる。また、得られた酸ハライドを有機溶媒(ジオキサン、テトラヒドロフラン等)中、アルカリ水溶液(重曹水又は水酸化ナトリウム溶液等)を用いて、アミンと0~40℃で反応させることにより行うこともできる。
カルボキシル基の保護反応はよく知られており、例えば、(1)酸ハライドを用いる方法、(2)混合酸無水物を用いる方法、(3)縮合剤を用いる方法等が挙げられる。
(1)酸ハライドを用いる方法は、例えば、カルボン酸を有機溶媒(クロロホルム、ジクロロメタン、ジエチルエーテル、テトラヒドロフラン等)中、又は無溶媒で、酸ハライド化剤(オキザリルクロリド、チオニルクロリド等)と-20℃~還流温度で反応させ、得られた酸ハライドを塩基(ピリジン、トリエチルアミン、ジメチルアニリン、ジメチルアミノピリジン、ジイソプロピルエチルアミン等)の存在下、又は非存在下、アルコールと有機溶媒(クロロホルム、ジクロロメタン、ジエチルエーテル、テトラヒドロフラン等)中、0℃~用いる溶媒の還流温度で反応させることにより行われる。また、有機溶媒(ジオキサン、テトラヒドロフラン等)中、アルカリ水溶液(重曹水又は水酸化ナトリウム溶液等)を用いて、酸ハライドと0~40℃で反応させることにより行うこともできる。
レボドパのアミノ基とカルボキシル基を保護した化合物、すなわち、
で示される化合物の水酸基のアシル化は、
R101、R102で示した保護基の脱保護反応は公知であり、以下の方法で行うことができる。例えば、
(1)アルカリ加水分解による脱保護反応、
(2)酸性条件下における脱保護反応、
(3)加水素分解による脱保護反応、
(4)シリル基の脱保護反応、
(5)金属を用いる脱保護反応、
(6)金属錯体を用いる脱保護反応等が挙げられる。
(1)アルカリ加水分解による脱保護反応は、例えば、有機溶媒(メタノール、テトラヒドロフラン、ジオキサン等)中、アルカリ金属の水酸化物(水酸化ナトリウム、水酸化カリウム、水酸化リチウム等)、アルカリ土類金属の水酸化物(水酸化バリウム、水酸化カルシウム等)、又は炭酸塩(炭酸ナトリウム、炭酸カリウム等)、或いはその水溶液、若しくはこれらの混合物を用いて、0~40℃の温度で行われる。
(方法1)(2S)-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸をp-トルエンスルホン酸を用いた脱保護反応に付した後、単離することなく(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩の溶媒和物を製造し、減圧下、加熱乾燥することでA型結晶を製造する方法;
(方法2)(2S)-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸をp-トルエンスルホン酸を用いた脱保護反応に付した後、非晶質を取り出し、非晶質の再結晶を行うことでA型結晶を製造する方法;
の反応によって製造することができる。
(方法1)
方法1は、(2S)-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸をp-トルエンスルホン酸を用いた脱保護反応に付した後、単離することなく(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩の溶媒和物を製造し、減圧下、加熱乾燥することでA型結晶を製造する方法である。
方法2は、(2S)-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸をp-トルエンスルホン酸を用いた脱保護反応に付した後、非晶質を取り出し、非晶質の再結晶を行うことでA型結晶を製造する方法である。
(方法3)(2S)-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸をp-トルエンスルホン酸を用いた脱保護反応に付した後、単離することなく(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩の溶媒和物を製造し、減圧下、加熱乾燥することでB型結晶を製造する方法;
(方法4)前述した方法1又は方法2でA型結晶を製造した後、有機溶媒(アセトン等)中、若しくはそれらと水の混合溶媒中、再結晶することにより、或いはスラリー撹拌することにより得られた溶媒和物を、減圧下、加熱乾燥することでB型結晶を製造する方法;
の反応によって製造することができる。
方法3は、(2S)-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸をp-トルエンスルホン酸を用いた脱保護反応に付した後、単離することなく(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩の溶媒和物を製造し、減圧下、加熱乾燥することでB型結晶を製造する方法である。
なお、方法3において使用する溶媒としては、アセトンが好ましい。
方法4は、前述した方法1又は方法2でA型結晶を製造した後、有機溶媒(アセトン等)中、若しくはそれらと水の混合溶媒中、再結晶することにより、或いはスラリー撹拌することにより得られた溶媒和物を、減圧下、加熱乾燥することでB型結晶を製造する方法である。
なお、方法4において使用する溶媒としては、アセトンが好ましい。
(方法5)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩を無機塩基で脱塩する方法;
(方法6)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩を有機塩基で脱塩する方法;
(方法7)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩を有機エポキシド化合物で脱塩する方法;
の反応に付すか、或いは、(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩を、無機塩基、又は有機塩基を用いて方法8~9:
(方法8)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩を無機塩基で脱塩する方法;
(方法9)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩を有機塩基で脱塩する方法;
の反応に付して製造することができる。
(方法5)
方法5は(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩を無機塩基で脱塩する方法である。アミノ酸の塩酸塩の無機塩基による脱塩反応は公知であり、例えば、溶媒(アセトニトリル、酢酸エチル、テトラヒドロフラン、ジオキサン、メタノール、エタノール、水、又はそれらの2以上の混合溶媒等)中、無機塩基(水酸化ナトリウム、水酸化カリウム、水酸化バリウム等)若しくはその水溶液を0.5~10当量、好ましくは0.5~3当量、より好ましくは0.5~1.5当量用いて、0℃~用いた溶媒の沸点、好ましくは0~40℃で反応させることにより行われる。
方法6は(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩を有機塩基で脱塩する方法である。アミノ酸の塩酸塩の有機塩基による脱塩反応は公知であり、例えば、溶媒(アセトニトリル、酢酸エチル、テトラヒドロフラン、ジオキサン、メタノール、エタノール、水、又はそれらの2以上の混合溶媒等)中、有機塩基(トリエチルアミン、ジイソプロピルエチルアミン、N-メチルピペリジン等)を0.5~10当量、好ましくは0.5~3当量、より好ましくは0.5~1.5当量用いて、0℃~用いた溶媒の沸点、好ましくは0~40℃で反応させることにより行われる。
方法7は(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩を有機エポキシド化合物で脱塩する方法である。アミノ酸の塩酸塩の有機エポキシド化合物による脱塩反応は公知であり、例えば、溶媒(アセトニトリル、酢酸エチル、テトラヒドロフラン、ジオキサン、メタノール、エタノール、水、又はそれらの2以上の混合溶媒等)中、有機エポキシド化合物(エピクロロヒドリン、エチレンオキシド、スチレンオキシド等)を0.5~10当量用いて、0℃~用いた溶媒の沸点、好ましくは0~60℃で反応させることにより行われる。
方法8は(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩を無機塩基で脱塩する方法である。ここで、原料としての(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩は結晶であってもよく、また、非結晶であってもよい。或いは、結晶と非結晶の任意の割合の混合物であってもよい。アミノ酸のトシル酸塩の無機塩基による脱塩反応は公知であり、例えば、溶媒(アセトニトリル、酢酸エチル、テトラヒドロフラン、ジオキサン、メタノール、エタノール、水、又はそれらの2以上の混合溶媒等)中、無機塩基(水酸化ナトリウム、水環化カリウム、水酸化バリウム等)若しくはその水溶液を0.5~10当量、好ましくは0.5~3当量、より好ましくは0.5~1.5当量用いて、0℃~用いた溶媒の沸点、好ましくは0~40℃で反応させることにより行われる。
方法9は(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩を有機塩基で脱塩する方法である。ここで、原料としての(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩は結晶であってもよく、また、非結晶であってもよい。或いは、結晶と非結晶の任意の割合の混合物であってもよい。アミノ酸のトシル酸塩の有機塩基による脱塩反応は公知であり、例えば、溶媒(アセトニトリル、酢酸エチル、テトラヒドロフラン、ジオキサン、メタノール、エタノール、水、又はそれらの2以上の混合溶媒等)中、有機塩基(トリエチルアミン、ジイソプロピルエチルアミン、N-メチルピペリジン等)を0.5~10当量、好ましくは0.5~3当量、より好ましくは0.5~1.5当量用いて、0℃~用いた溶媒の沸点、好ましくは0~40℃で反応させることにより行われる。
(方法10)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体を前述の方法5~9で製造した後、粗体を単離することなくA型結晶を製造する方法;
(方法11)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体を前述の方法5~9で製造した後、粗体を単離して、A型結晶へと変換する方法;
の反応によって製造することができる。
(方法10)
方法10は、(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体を前述の方法5~9で製造した後、粗体を単離することなくA型結晶を製造する方法である。
なお、方法10において使用する溶媒としては、アセトニトリルが好ましい。
方法11は(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体を前述の方法5~9で製造した後、粗体を単離して、A型結晶へと変換する方法である。
なお、方法11において使用する溶媒としては、アセトニトリルが好ましい。
(方法12)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体を前述の方法5~9で製造した後、粗体を単離することなくB型結晶を製造する方法;
(方法13)(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体を前述の方法5~9で製造した後、粗体を単離して、B型結晶へと変換する方法;
の反応によって製造することができる。
(方法12)
方法12は、(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体を前述の方法5~9で製造した後、粗体を単離することなくB型結晶を製造する方法である。
なお、方法12において使用する溶媒としては、アセトニトリルが好ましい。
方法13は、(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体を前述の方法5~9で製造した後、粗体を単離して、B型結晶へと変換する方法である。
なお、方法13において使用する溶媒としては、アセトニトリルが好ましい。
本発明化合物の毒性は低いものであるため、医薬品として安全に使用することができる。特に本発明化合物は変異原性を有しないため、パーキンソン病及び/又はパーキンソン症候群のような、実に数年や数十年にわたって薬物を服用しなけれなならない場合にも、患者は安心して服用を続けることができる。
本発明化合物は、パーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療に有用である。ここで、パーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療には、パーキンソン病やパーキンソン症候群の予防や治療といった文言通りの意味のほか、例えば、ジスキネジアの発症予防、ジスキネジアの重症度軽減、パーキンソン病やパーキンソン症候群の病態進行抑制(ドパミン神経保護作用)、パーキンソン病やパーキンソン症候群の非運動症状(例えば、睡眠障害(例:入眠障害、頻回中途覚醒、レム睡眠行動異常、睡眠時呼吸障害、覚醒障害、睡眠相後退症候群、夜驚症、夜間頻尿、睡眠麻痺、睡眠関連摂食障害等)、精神症状(例:うつ症状、不安、感情鈍麻、快感喪失、幻覚、妄想、衝動制御障害、ドパミン調節障害等)、自律神経症状(例:嘔気、便秘、垂涎等の消化器症状や、起立性低血圧、食後性低血圧、発汗発作、あぶら顔、排尿障害、勃起不全等)、認知機能障害、疲労、性機能障害、しびれ、痛み等)の予防及び/又は治療も含まれる。そして、本発明化合物はレボドパのプロドラッグ体であるがゆえに、レボドパが治療薬として用いられ、或いは有効性が期待されるその他の疾患、例えば、レビー小体病、鬱病、注意力欠如障害、統合失調症、躁鬱病、認知機能障害、下肢静止不能症候群(レストレスレッグス症候群)、周期性四肢運動障害、遅発性ジスキネジー、ハンチントン病、トゥレット症候群、高血圧症、嗜癖障害、鬱血性心不全、糖尿病性神経障害に伴う疼痛、帯状疱疹後神経痛、繊維筋痛症、自閉症、薬物依存症、ナルコレプシー又は日中の過度の眠気等の疾患、ドパ反応性ジストニア、植物状態、ペリー症候群、瀬川病、悪性症候群、射精障害、胃不全麻痺、レッシュ・ナイハン病、弱視、肺高血圧症、大脳皮質基底核変性症、フェニルケトン尿症、パニック発作、性欲減退、嚥下反射障害、多系統萎縮症等の予防及び/又は治療にも有用である。
例えば、吸入用液剤の場合には、防腐剤(例えば、塩化ベンザルコニウム、パラベン等)、着色剤、緩衝化剤(例えば、リン酸ナトリウム、酢酸ナトリウム等)、等張化剤(例えば、塩化ナトリウム、濃グリセリン等)、増粘剤(例えば、カルボキシビニルポリマー等)、吸収促進剤等を必要に応じて適宜選択して調製される。
2-(ベンゾイルオキシ)-2-メチルプロパン酸
TLC(Rf値):0.37(酢酸エチル);
NMR(300MHz,CDCl3):δ 8.20-9.40 (br, 1H), 8.01-8.06 (m, 2H), 7.53-7.59 (m, 1H), 7.40-7.46 (m, 2H), 1.73 (s, 6H)。
(2S)-ベンジル 2-((tert-ブトキシカルボニル)アミノ)-3-(3,4-ジヒドロキシフェニル)プロパノアート
TLC(Rf値):0.37(ジクロロメタン:メタノール:酢酸=17:3:1)。
TLC(Rf値):0.64(n-ヘキサン:酢酸エチル:酢酸=50:50:1);
NMR(300MHz,CDCl3):δ 7.31-7.40 (m, 5H), 6.98 (d, J=7.8Hz, 1H), 6.44 (dd, J=7.8, 1.8Hz, 1H), 6.40 (d, J=1.8Hz, 1H), 5.26-5.64 (br, 2H), 5.05-5.23 (m, 2H), 5.00 (d, J=8.1Hz, 1H), 4.50-4.58 (m, 1H), 2.94 (d, J=5.7Hz, 2H), 1.41 (s, 9H)。
(2S)-((4-(3-(ベンジルオキシ)-2-((tert-ブトキシカルボニル)アミノ)-3-オキソプロピル)-1,2-フェニレン)ビス(オキシ))ビス(2-メチル-1-オキソプロパン-2,1-ジイル) ジベンゾアート
TLC(Rf値):0.44(n-ヘキサン:酢酸エチル=3:1);
NMR(300MHz,CDCl3):δ 8.01-8.07 (m, 4H), 7.52-7.60 (m, 2H), 7.38-7.49 (m, 4H), 7.25-7.32 (m, 5H), 7.15 (d, J=8.1Hz, 1H), 7.05 (s, 1H), 6.91 (d, J=8.1Hz, 1H), 5.12 (s, 2H), 5.02 (d, J=7.8Hz, 1H), 4.55-4.63 (m, 1H), 3.09 (d, J=5.4Hz, 2H), 1.81-1.85 (m, 12H), 1.39 (s, 9H)。
(2S)-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸
TLC(Rf値):0.38(n-ヘキサン:酢酸エチル:酢酸=100:100:1);
NMR(300MHz,CD3OD):δ 8.02-8.07 (m, 4H), 7.59-7.66 (m, 2H), 7.46-7.52 (m, 4H), 7.13-7.21 (m, 3H), 4.33 (dd, J=9.0, 5.1Hz, 1H), 3.18 (dd, J=13.5, 5.1Hz, 1H), 2.93 (dd, J=13.5, 9.0Hz, 1H), 1.82 (s, 12H), 1.33 (s, 9H)。
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.64(酢酸エチル:酢酸:水=5:5:1);
NMR(300MHz,CD3OD):δ 8.02-8.06 (m, 4H), 7.60-7.67 (m, 2H), 7.46-7.52 (m, 4H), 7.25-7.30 (m, 3H), 4.24 (dd, J=8.4, 5.1Hz, 1H), 3.37 (dd, J=15.0, 5.1Hz, 1H), 3.13 (dd, J=15.0, 8.4Hz, 1H), 1.83 (s, 6H), 1.82 (s, 6H)。
[測定条件]
装置:BRUKER DISCOVER with GADDS(C2)
ターゲット:Cu
フィルター:なし
電圧:40kV
電流:40mA
露光時間:180sec
[結果]
Cu-Kα線を使用した粉末X線回折スペクトル法において、結晶様のピークは認められなかった。
[測定条件]
装置:SEIKO INSTRUMENT DSC6200
試料量:3.73mg
試料セル:Aluminium Standard 40μL(ピンホール付蓋あり)
窒素流量:40mL/min
昇温速度:10℃/min
昇温開始温度:25℃
[結果]
その結果、82.83℃付近に吸熱ピークを有することがわかった。
[測定条件]
装置:日本分光 FTIR-660plus/SENSIR DURASCOPE
分解能:4cm-1
スキャン回数:32
[結果]
IR(Attenuated total reflectance法(以下ATR法と略記する)):3409、2992、2944、2865、2629、1970、1774、1718、1655、1601、1585、1508、1470、1452、1428、1388、1369、1317、1290、1258、1204、1168、1125、1093、1070、1026、1003、958、866、806、741、714、687、617、530、496、467、447、419cm-1。
ベンジル 3-ヒドロキシ-2,2-ジメチルプロパノアート
TLC(Rf値):0.39(n-ヘキサン:酢酸エチル=3:1);
NMR(CDCl3):δ 7.29-7.41 (m, 5H), 5.15 (s, 2H), 3.57 (d, J=6.3Hz, 2H), 1.22 (s, 6H)。
3-(ベンジルオキシ)-2,2-ジメチル-3-オキソプロピルチオフェン-2-カルボキシラート
TLC(Rf値):0.65(n-ヘキサン:酢酸エチル=3:1);
NMR(CDCl3):δ 7.67-7.69 (m, 1H), 7.52-7.55 (m, 1H), 7.26-7.34 (m, 5H), 7.05-7.08 (m, 1H), 5.16 (s, 2H), 4.34 (s, 2H), 1.28 (s, 6H)。
2,2-ジメチル-3-((チオフェン-2-カルボニル)オキシ)プロパン酸
TLC(Rf値):0.60(酢酸エチル);
NMR(CDCl3):δ 7.77-7.79 (m, 1H), 7.53-7.55 (m, 1H), 7.06-7.10 (m, 1H), 4.34 (s, 2H), 1.33 (s, 6H)。
(2S)-((4-(3-(ベンジルオキシ)-2-((tert-ブトキシカルボニル)アミノ)-3-オキソプロピル)-1,2-フェニレン)ビス(オキシ))ビス(2,2-ジメチル-3-オキソプロパン-3,1-ジイル) ビス(チオフェン-2-カルボキシラート)
TLC(Rf値):0.60(n-ヘキサン:酢酸エチル=1:1);
NMR(CDCl3):δ 7.79-7.82 (m, 2H), 7.53-7.58 (m, 2H), 7.26-7.31 (m, 5H), 7.06-7.11 (m, 2H), 7.00 (d, J=8.4Hz, 1H), 6.87-6.94 (m, 2H), 5.10 (s, 2H), 4.96-5.04 (m, 1H), 4.52-4.59 (m, 1H), 4.43 (s, 2H), 4.42 (s, 2H), 2.99-3.06 (m, 2H), 1.34-1.35 (m, 21H)。
(2S)-3-(3,4-ビス((2,2-ジメチル-3-((チオフェン-2-カルボニル)オキシ)プロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸
TLC(Rf値):0.12(酢酸エチル);
NMR(CDCl3):δ 7.74-7.85 (m, 2H), 7.03-7.18 (m, 4H), 6.99-7.12 (m, 3H), 4.43 (s, 2H), 4.42 (s, 2H), 4.05-4.22 (m, 1H), 3.06-3.14 (m, 1H), 2.83-2.91 (m, 1H), 1.36-1.48 (m, 21H)。
(2S)-2-アミノ-3-(3,4-ビス((2,2-ジメチル-3-((チオフェン-2-カルボニル)オキシ)プロパノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.74(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 7.75-7.84 (m, 4H), 7.14-7.23 (m, 5H), 4.45 (s, 2H), 4.44 (s, 2H), 4.17-4.23 (m, 1H), 3.35-3.38 (m, 1H), 3.09 (dd, J=14.4, 8.4Hz, 1H), 1.41-1.44 (m, 12H)。
3-(ベンジルオキシ)-2,2-ジメチル-3-オキソプロピル チオフェン-3-カルボキシラート
TLC(Rf値):0.50(n-ヘキサン:酢酸エチル=5:1);
NMR(CDCl3):δ 7.89-7.91 (m, 1H), 7.39-7.42 (m, 1H), 7.24-7.38 (m, 6H), 5.16 (s, 2H), 4.31 (s, 2H), 1.32 (s, 6H)。
2,2-ジメチル-3-((チオフェン-3-カルボニル)オキシ)プロパン酸
TLC(Rf値):0.12(酢酸エチル);
NMR(CDCl3):δ 8.08-8.10 (m, 1H), 7.48-7.51 (m, 1H), 7.27-7.31 (m, 1H), 4.32 (s, 2H), 1.33 (s, 6H)。
(2S)-((4-(3-(ベンジルオキシ)-2-((tert-ブトキシカルボニル)アミノ)-3-オキソプロピル)-1,2-フェニレン)ビス(オキシ))ビス(2,2-ジメチル-3-オキソプロパン-3,1-ジイル) ビス(チオフェン-3-カルボキシラート)
TLC(Rf値):0.47(n-ヘキサン:酢酸エチル=2:1);
NMR(CDCl3):δ 8.10-8.12 (m, 2H), 7.49-7.52 (m, 2H), 7.23-7.33 (m, 7H), 6.85-6.97 (m, 3H), 5.10 (s, 2H), 4.98 (d, J=8.7Hz, 1H), 4.52-4.58 (m, 1H), 4.40 (s, 2H), 4.41 (s, 2H), 2.96-3.08 (m, 2H), 1.32-1.36 (m, 21H)。
(2S)-3-(3,4-ビス((2,2-ジメチル-3-((チオフェン-3-カルボニル)オキシ)プロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸
TLC(Rf値):0.12(酢酸エチル);
NMR(CDCl3):δ 8.20-8.24 (m, 2H), 7.45-7.52 (m, 4H), 6.99-7.12 (m, 3H), 4.41 (s, 2H), 4.40 (s, 2H), 4.19-4.26 (m, 1H), 3.06-3.14 (m, 1H), 2.84-2.90 (m, 1H), 1.24-1.34 (m, 21H)。
(2S)-2-アミノ-3-(3,4-ビス((2,2-ジメチル-3-((チオフェン-3-カルボニル)オキシ)プロパノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.57(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 8.20-8.24 (m, 2H), 7.46-7.51 (m, 4H), 7.12-7.21 (m, 3H), 4.42 (s, 2H), 4.41 (s, 2H), 4.14-4.21 (m, 1H), 3.31-3.35 (m, 1H), 3.05-3.10 (m, 1H), 1.41-1.43 (m, 12H)。
3-(ベンジルオキシ)-2,2-ジメチル-3-オキソプロピル 2-メトキシベンゾアート
TLC(Rf値):0.66(n-ヘキサン:酢酸エチル=2:1);
NMR(CDCl3):δ 7.68 (dd, J=7.5, 2.1Hz, 1H), 7.43-7.49 (m, 1H), 7.25-7.32 (m, 5H), 6.89-6.96 (m, 2H), 5.15 (s, 2H), 4.35 (s, 2H), 3.84 (s, 3H), 1.33 (s, 6H)。
3-((2-メトキシベンゾイル)オキシ)-2,2-ジメチルプロパン酸
TLC(Rf値):0.12(n-ヘキサン:酢酸エチル=3:1);
NMR(CDCl3):δ 7.78-7.82 (m, 1H), 7.43-7.47 (m, 1H), 6.93-6.99 (m, 2H), 4.34 (s, 2H), 3.86 (s, 3H), 1.34 (s, 6H)。
(2S)-((4-(3-(ベンジルオキシ)-2-((tert-ブトキシカルボニル)アミノ)-3-オキソプロピル)-1,2-フェニレン)ビス(オキシ))ビス(2,2-ジメチル-3-オキソプロパン-3,1-ジイル) ビス(2-メトキシベンゾアート)
TLC(Rf値):0.74(n-ヘキサン:酢酸エチル=1:1);
NMR(CDCl3):δ 7.76-7.81 (m, 2H), 7.41-7.50 (m, 2H), 7.25-7.31 (m, 5H), 6.82-6.99 (m, 7H), 5.07 (s, 2H), 4.94 (d, J=7.8Hz, 1H), 4.48-4.55 (m, 1H), 4.41 (s, 4H), 3.84 (s, 6H), 2.94-3.03 (m, 2H), 1.28-1.39 (m, 21H)。
(2S)-3-(3,4-ビス((3-((2-メトキシベンゾイル)オキシ)-2,2-ジメチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸
TLC(Rf値):0.12(n-ヘキサン:酢酸エチル=1:1);
NMR(CDCl3):δ 7.77-7.86 (m, 2H), 7.44-7.51 (m, 2H), 6.93-7.05 (m, 7H), 4.94-5.01 (m, 1H), 4.32-4.59 (m, 5H), 3.86 (s, 3H), 3.84 (s, 3H), 3.07 (d, J=5.4Hz, 2H), 1.34-1.41 (m, 21H)。
(2S)-2-アミノ-3-(3,4-ビス((3-((2-メトキシベンゾイル)オキシ)-2,2-ジメチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.45(酢酸エチル:酢酸:水=5:1:1);
NMR(CD3OD):δ 7.70-7.75 (m, 2H), 7.49-7.53 (m, 2H), 7.02-7.21 (m, 5H), 6.95-7.02 (m, 2H), 4.40 (s, 2H), 4.38 (s, 2H), 4.14 (dd, J=8.7, 5.1Hz, 1H), 3.82-3.83 (m, 6H), 3.25-3.30 (m, 1H), 3.04 (dd, J=14.7, 8.7Hz, 1H), 1.37-1.46 (m, 12H)。
(2S)-2-アミノ-3-(3,4-ビス((2,2-ジエチルブタノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.78(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 7.23 (dd, J=8.4, 1.8Hz, 1H), 7.17 (d, J=8.4Hz, 1H), 7.13 (d, J=1.8Hz, 1H), 4.25 (dd, J=8.1, 5.4Hz, 1H), 3.35 (dd, J=14.7, 5.4Hz, 1H), 3.14 (dd, J=14.7, 8.1Hz, 1H), 1.70-1.79 (m, 12H), 0.87-0.96 (m, 18H)。
(2S)-2-アミノ-3-(3,4-ビス((2-エチル-2-メチルブタノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.75(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 7.23 (dd, J=8.4, 2.1Hz, 1H), 7.15 (d, J=8.4Hz, 1H), 7.13 (d, J=2.1Hz, 1H), 4.25 (dd, J=8.1, 5.1Hz, 1H), 3.36 (dd, J=14.7, 5.1Hz, 1H), 3.14 (dd, J=14.7, 8.1Hz, 1H), 1.59-1.86 (m, 8H), 1.25 (s, 3H), 1.24 (s, 3H), 0.93-1.03 (m, 12H)。
(2S)-2-アミノ-3-(3,4-ビス((4,4-ジメチルペンタノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.63(酢酸エチル:酢酸:水=6:1:1);
NMR(CD3OD):δ 7.18-7.24 (m, 3H), 4.23 (dd, J=8.4, 5.1Hz, 1H), 3.38 (dd, J=14.7, 5.1Hz, 1H), 3.12 (dd, J=14.7, 8.4Hz, 1H), 2.52-2.58 (m, 4H), 1.61-1.67 (m, 4H), 0.89-1.17 (m, 18H)。
(2S)-2-アミノ-3-(3,4-ビス((3,3-ジメチルペンタ-4-エノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.45(酢酸エチル:酢酸:水=6:1:1);
NMR(CD3OD):δ 7.13-7.25 (m, 3H), 5.99 (dd, J=17.4, 10.5Hz, 1H), 5.01-5.10 (m, 2H), 4.23 (dd, J=8.4, 5.1Hz, 1H), 3.37 (dd, J=14.7, 5.1Hz, 1H), 3.11 (dd, J=14.7, 8.4Hz, 1H), 2.57 (s, 2H), 2.56 (s, 2H), 1.21-1.22 (m, 12H)。
(2S)-2-アミノ-3-(3,4-ビス((3-エチル-3-メチルペンタノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.51(酢酸エチル:酢酸:水=5:1:1);
NMR(CD3OD):δ 7.15-7.26 (m, 3H), 4.23 (dd, J=8.4, 5.1Hz, 1H), 3.38 (dd, J=14.7, 5.1Hz, 1H), 3.17 (dd, J=14.7, 8.4Hz, 1H), 2.45 (s, 2H), 2.44 (s, 2H), 1.42-1.50 (m, 8H), 1.04 (s, 3H), 1.03 (s, 3H), 0.87-0.92 (m, 12H)。
(2S)-2-アミノ-3-(3,4-ビス((3-イソプロピル-4-メチルペンタノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.67(酢酸エチル:酢酸=3:1);
NMR(CD3OD):δ 7.15-7.26 (m, 3H), 4.13 (dd, J=8.4, 5.1Hz, 1H), 3.20-3.30 (m, 1H), 3.05-3.13 (m, 1H), 2.47 (d, J=5.7Hz, 2H), 2.46 (d, J=5.7Hz, 2H), 1.79-1.90 (m, 4H), 1.65-1.72 (m, 2H), 0.89-0.99 (m, 24H)。
(2S)-2-アミノ-3-(3,4-ビス((2-メチルベンゾイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.79(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 7.93-7.97 (m, 2H), 7.34-7.47 (m, 5H), 7.15-7.35 (m, 4H), 4.30-4.36 (m, 1H), 3.47 (dd, J=14.7, 5.1Hz, 1H), 3.25 (dd, J=14.7, 8.4Hz, 1H), 2.49 (s, 3H), 2.48 (s, 3H)。
(2S)-2-アミノ-3-(3,4-ビス((4-ヒドロキシベンゾイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.62(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 7.82-7.88 (m, 4H), 7.30-7.40 (m, 3H), 6.73-6.78 (m, 4H), 4.23 (dd, J=8.7, 4.8Hz, 1H), 3.44 (dd, J=14.7, 4.8Hz, 1H), 3.16 (dd, J=14.7, 8.7Hz, 1H)。
(2S)-2-アミノ-3-(3,4-ビス((2-(トリフルオロメチル)ベンゾイル)オキシ)フェニル)プロパン酸
TLC(Rf値):0.80(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 7.84-7.91 (m, 4H), 7.65-7.79 (m, 4H), 7.39-7.49 (m, 3H), 4.35 (dd, J=7.8, 5.1Hz, 1H), 3.43 (dd, J=14.4, 5.1Hz, 1H), 3.20-3.27 (m, 1H)。
(2S)-2-アミノ-3-(3,4-ビス((シクロプロパンカルボニル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.49(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 7.17-7.23 (m, 3H), 4.27 (dd, J=8.7, 5.1Hz, 1H), 3.37 (dd, J=14.7, 5.1Hz, 1H), 3.12 (dd, J=14.7, 8.7Hz, 1H), 1.84-1.90 (m, 2H), 1.08-1.13 (m, 8H)。
(2S)-2-アミノ-3-(3,4-ビス((1-メチルシクロプロパンカルボニル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.23(酢酸エチル:酢酸:水=6:1:1);
NMR(CD3OD):δ 7.15-7.25 (m, 3H), 4.26 (dd, J=8.4, 4.8Hz, 1H), 3.35 (dd, J=14.7, 4.8Hz, 1H), 3.12 (dd, J=14.7, 8.4Hz, 1H), 1.33-1.46 (m, 10H), 0.93-0.97 (m, 4H)。
(2S)-2-アミノ-3-(3,4-ビス(2-シクロペンチルアセトキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.28(酢酸エチル:酢酸:水=6:1:1);
NMR(CD3OD):δ 7.17-7.27 (m, 3H), 4.27 (dd, J=8.4, 5.1Hz, 1H), 3.28 (dd, J=14.7, 5.1Hz, 1H), 3.13 (dd, J=14.7, 8.4Hz, 1H), 2.56-2.60 (m, 4H), 2.24-2.35 (m, 2H), 1.86-1.96 (m, 4H), 1.54-1.75 (m, 8H), 1.20-1.32 (m, 4H)。
(2S)-2-アミノ-3-(3,4-ビス(2-シクロヘキシルアセトキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.30(酢酸エチル:酢酸:水=6:1:1);
NMR(CD3OD):δ 7.16-7.56 (m, 3H), 4.23 (dd, J=8.7, 5.1Hz, 1H), 3.37 (dd, J=14.7, 5.1Hz, 1H), 3.11 (dd, J=14.7, 8.4Hz, 1H), 2.44 (d, J=6.6Hz, 2H), 2.43 (d, J=6.6Hz, 2H), 1.61-1.88 (m, 12H), 1.01-1.40 (m, 10H)。
(2S)-2-アミノ-3-(3,4-ビス(2-(1-メチルシクロヘキシル)アセトキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.43(酢酸エチル:酢酸=3:1);
NMR(CD3OD):δ 7.21-7.31 (m, 3H), 4.28 (dd, J=8.4, 4.8Hz, 1H), 3.30-3.46 (m, 1H), 3.17 (dd, J=14.4, 8.4Hz, 1H), 2.56 (s, 2H), 2.55 (s, 2H), 1.49-1.70 (m, 20H), 1.18 (s, 3H), 1.17 (s, 3H)。
(2S)-2-アミノ-3-(3,4-ビス(2-(1-メチルシクロペンチル)アセトキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.29(酢酸エチル:酢酸=3:1);
NMR(CD3OD):δ 7.16-7.26 (m, 3H), 4.16 (dd, J=8.7, 4.8Hz, 1H), 3.22-3.40 (m, 1H), 3.05-3.13 (m, 1H), 2.56 (s, 2H), 2.57 (s, 2H), 1.50-1.73 (m, 16H), 1.16 (s, 3H), 1.15 (s, 3H)。
(2S)-2-アミノ-3-(3,4-ビス((2-アセトキシ-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.70(酢酸エチル:酢酸:水=3:1:1);
NMR(CD3OD):δ 7.18-7.27 (m, 3H), 4.25 (dd, J=8.4, 5.4Hz, 1H), 3.37 (dd, J=14.7, 5.4Hz, 1H), 3.13 (dd, J=14.7, 8.4Hz, 1H), 2.09 (s, 3H), 2.07 (s, 3H), 1.66-1.68 (m, 12H)。
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-エチルブタノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.51(酢酸エチル:酢酸:水=5:1:1);
NMR(CD3OD):δ 8.01-8.06 (m, 4H), 7.61-7.64 (m, 2H), 7.49-7.52 (m, 4H), 7.25-7.47 (m, 3H), 4.22 (dd, J=8.4, 5.1Hz, 1H), 3.32-3.35 (m, 1H), 3.05-3.16 (m, 1H), 2.20-2.37 (m, 8H), 0.97-1.03 (m, 12H)。
(2S)-2-アミノ-3-(3,4-ビス(((S)-2-(ベンゾイルオキシ)プロパノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.38(酢酸エチル:酢酸:水=10:2:1);
NMR(CD3OD):δ 8.06-8.09 (m, 4H), 7.61-7.66 (m, 2H), 7.46-7.52 (m, 4H), 7.26-7.29 (m, 3H), 5.61-5.68 (m, 2H), 4.21 (dd, J=8.4, 5.4Hz, 1H), 3.30-3.40 (m, 1H), 3.13 (dd, J=14.7, 8.1Hz, 1H), 1.81 (d, J=7.2Hz, 3H), 1.80 (d, J=7.2Hz, 3H)。
(2S)-2-アミノ-3-(3,4-ビス(((R)-2-(ベンゾイルオキシ)プロパノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.38(酢酸エチル:酢酸:水=10:2:1);
NMR(CD3OD):δ 8.06-8.09 (m, 4H), 7.61-7.66 (m, 2H), 7.46-7.52 (m, 4H), 7.26-7.29 (m, 3H), 5.61-5.68 (m, 2H), 4.21 (dd, J=8.4, 5.4Hz, 1H), 3.30-3.40 (m, 1H), 3.13 (dd, J=14.7, 8.1Hz, 1H), 1.81 (d, J=7.2Hz, 3H), 1.80 (d, J=7.2Hz, 3H)。
3-(ベンジルオキシ)-2,2-ジメチル-3-オキソプロピル ベンゾアート
TLC(Rf値):0.70(n-ヘキサン:酢酸エチル=2:1);
NMR(300MHz,CDCl3):δ 7.89-7.93 (m, 2H), 7.50-7.57 (m, 1H), 7.36-7.42 (m, 2H), 7.23-7.33 (m, 5H), 5.16 (s, 2H), 4.37 (s, 2H), 1.34 (s, 6H)。
3-(ベンゾイルオキシ)-2,2-ジメチルプロパン酸
TLC(Rf値):0.22(n-ヘキサン:酢酸エチル=3:1);
NMR(CDCl3):δ 7.99-8.03 (m, 2H), 7.48-7.61 (m, 1H), 7.38-7.46 (m, 2H), 4.37 (s, 2H), 1.35 (s, 6H)。
(S)-((4-(3-(ベンジルオキシ)-2-((tert-ブトキシカルボニル)アミノ)-3-オキソプロピル)-1,2-フェニレン)ビス(オキシ))ビス(2,2-ジメチル-3-オキソプロパン-3,1-ジイル) ジベンゾアート
TLC(Rf値):0.51(n-ヘキサン:酢酸エチル=2:1);
NMR(CDCl3):δ 8.00-8.05 (m, 4H), 7.50-7.59 (m, 2H), 7.39-7.46 (m, 4H), 7.23-7.34 (m, 5H), 6.83-6.97 (m, 3H), 5.09 (s, 2H), 4.97 (d, J=8.4Hz, 1H), 4.50-4.57 (m, 1H), 4.45 (s, 4H), 2.94-3.03 (m, 2H), 1.39-1.43 (m, 21H)。
(S)-3-(3,4-ビス((3-(ベンゾイルオキシ)-2,2-ジメチルプロパノイル)オキシ)フェニル)-2-((tert-ブトキシカルボニル)アミノ)プロパン酸
TLC(Rf値):0.12(n-ヘキサン:酢酸エチル=2:1);
NMR(CDCl3):δ 8.00-8.04 (m, 4H), 7.54-7.61 (m, 2H), 7.41-7.48 (m, 4H), 6.97-7.05 (m, 2H), 6.91 (d, J=1.8Hz, 1H), 4.99 (d, J=7.5Hz, 1H), 4.42-4.50 (m, 5H), 2.98-3.11 (m, 2H), 1.40-1.42 (m, 21H)。
(S)-2-アミノ-3-(3,4-ビス((3-(ベンゾイルオキシ)-2,2-ジメチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩
TLC(Rf値):0.34(酢酸エチル:酢酸:水=6:1:1);
NMR(CD3OD):δ 7.97-8.02 (m, 4H), 7.57-7.63 (m, 2H), 7.43-7.51 (m, 4H), 7.12-7.18 (m, 3H), 4.54-4.66 (m, 4H), 4.09 (dd, J=8.7, 4.8Hz, 1H), 3.06-3.25 (m, 1H), 2.99-3.07 (m, 1H), 1.43-1.44 (m, 12H)。
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩の結晶(A型結晶)
TLC(Rf値):0.56(酢酸エチル:酢酸:水=10:1:1);
NMR(300MHz,CD3OD):δ 8.06-8.03 (m, 4H), 7.71-7.62 (m, 4H), 7.52-7.48 (m, 4H), 7.31-7.20 (m, 5H), 4.27 (dd, J=8.4, 5.1Hz, 1H), 3.37 (dd, J=14.7, 5.1Hz, 1H), 3.13 (dd, J=14.7, 8.4Hz, 1H), 2.36 (s, 3H), 1.83 (s, 12H)。
[測定条件]
装置:BRUKER DISCOVER with GADDS(C2)
ターゲット:Cu
フィルター:なし
電圧:40kV
電流:40mA
露光時間:180sec
[結果]
Cu-Kα線を使用した粉末X線回折スペクトル法で得られた回折角(2θ)(度)及び相対強度(%)の結果を表1に示す。なお、相対強度は最も高いピークの高さ(Lin(Counts))を100%として各々のピークの高さを算出したものである。
[測定条件]
装置:SEIKO INSTRUMENT DSC6200
試料量:4.22mg
試料セル:Aluminium Standard 40μL(ピンホール付蓋あり)
アルゴンガス流量:40mL/min
昇温速度:5℃/min
昇温開始温度:25℃
[結果]
その結果、135.95℃付近に吸熱ピークを有することがわかった。
[測定条件]
装置:日本分光 FTIR-660plus/SENSIR DURASCOPE
分解能:4cm-1
スキャン回数:32
[結果]
IR(ATR法):1780、1712、1599、1508、1452、1388、1316、1289、1217、1166、1120、1090、1071、1036、1026、1010、957、900、864、817、742、713、680、622、567、550、472、440cm-1。
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩の結晶(B型結晶)
[測定条件]
装置:BRUKER DISCOVER with GADDS(C2)
ターゲット:Cu
フィルター:なし
電圧:40kV
電流:40mA
露光時間:180sec
[結果]
Cu-Kα線を使用した粉末X線回折スペクトル法で得られた回折角(2θ)(度)及び相対強度(%)の結果を表2に示す。なお、相対強度は最も高いピークの高さ(Lin(Counts))を100%として各々のピークの高さを算出したものである。
[測定条件]
装置:SEIKO INSTRUMENT DSC6200
試料量:3.08mg
試料セル:Aluminium Standard 40μL(ピンホール付蓋あり)
アルゴンガス流量:40mL/min
昇温速度:5℃/min
昇温開始温度:25℃
[結果]
その結果、134.54℃付近に吸熱ピークを有することがわかった。
[測定条件]
装置:日本分光 FTIR-660plus/SENSIR DURASCOPE
分解能:4cm-1
スキャン回数:32
[結果]
IR(ATR法):1781、1711、1600、1507、1315、1287、1220、1203、1166、1119、1088、1070、1036、1027、1010、944、898、863、816、713、681、617、567、531、517、507、484、470、452、437、421、413cm-1。
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の粗体
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の結晶(A型結晶)
[測定条件]
装置:BRUKER DISCOVER with GADDS(C2)
ターゲット:Cu
フィルター:なし
電圧:40kV
電流:40mA
露光時間:180sec
[結果]
Cu-Kα線を使用した粉末X線回折スペクトル法で得られた回折角(2θ)(度)及び相対強度(%)の結果を表3に示す。なお、相対強度は最も高いピークの高さ(Lin(Counts))を100%として各々のピークの高さを算出したものである。
[測定条件]
装置:SEIKO INSTRUMENT DSC6200
試料量:6.07mg
試料セル:Aluminium Standard 40μL(ピンホール付蓋あり)
アルゴンガス流量:40mL/min
昇温速度:10℃/min
昇温開始温度:25℃
[結果]
その結果、148.7℃付近に発熱ピークを、184.7、194.7、及び200.3℃付近に吸熱ピークを有することがわかった。
[測定条件]
装置:日本分光 FTIR-660plus/SENSIR DURASCOPE
分解能:4cm-1
スキャン回数:32
[結果]
IR(ATR法):1771、1720、1632、1602、1543、1506、1469、1451、1387、1359、1316、1287、1203、1165、1093、1069、1026、957、937、898、863、802、742、710、687、615、557、526、490、482、452、424、416、408cm-1。
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の結晶(B型結晶)
[測定条件]
装置:BRUKER DISCOVER with GADDS(C2)
ターゲット:Cu
フィルター:なし
電圧:40kV
電流:40mA
露光時間:180sec
[結果]
Cu-Kα線を使用した粉末X線回折スペクトル法で得られた回折角(2θ)(度)及び相対強度(%)の結果を表4に示す。なお、相対強度は最も高いピークの高さ(Lin(Counts))を100%として各々のピークの高さを算出したものである。
[測定条件]
装置:SEIKO INSTRUMENT DSC6200
試料量:5.68mg
試料セル:Aluminium Standard 40μL(ピンホール付蓋あり)
アルゴンガス流量:40mL/min
昇温速度:10℃/min
昇温開始温度:25℃
[結果]
その結果、183.3℃付近に発熱ピークを、192.2、及び200.8℃付近に吸熱ピークを有することがわかった。
[測定条件]
装置:日本分光 FTIR-660plus/SENSIR DURASCOPE
分解能:4cm-1
スキャン回数:32
[結果]
IR(ATR法):1771、1715、1608、1505、1469、1452、1411、1386、1368、1352、1315、1288、1256、1201、1166、1092、1070、1026、955、895、865、803、744、711、675、617、605、472、444、432、414cm-1。
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の結晶(A型結晶)
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の結晶(B型結晶)
(1)イヌ動態試験
レボドパ服用患者における、レボドパの血中濃度と副作用の関係は次第に明らかにされつつある。例えば、ジスキネジアは有効血中濃度を上回るレボドパの頻回の曝露によってもたらされ、ウェアリングオフは有効血中濃度に満たないレボドパによってもたらされると考えられている。レボドパ服用患者においてレボドパの服用回数を減らし、且つジスキネジアやウェアリングオフといった副作用を回避するためには、レボドパの血中濃度を、治療域の濃度範囲の中で、できるだけ長時間推移させる必要がある。本発明者らは、本発明化合物がこのような平坦な血中濃度推移を示すレボドパプロドラッグ体であることを明らかにする目的で、実施例5で製造した本発明化合物と、レボドパ、比較に用いた化合物群(参考例6、11、16、18、19、21-23、25-35、及び40で製造した化合物、ならびに国際公開第2009/022098号パンフレットに記載の3,3-ジメチル-酪酸 4-((S)-2-アミノ-2-メトキシカルボニル-エチル)-2-(3,3-ジメチル-ブチリルオキシ)-フェニルエステル(以下、化合物Xと表記する。)の各々について、動態試験を実施した。
本発明化合物(実施例5)と、レボドパ、比較に用いた化合物群(参考例6、11、16、18、19、21-23、25-35、及び40で製造した化合物、ならびに化合物Xの各々について、秤量後、媒体を用い、レボドパ換算で1mg/mLに溶解し、経口投与用の薬液を調製した。
この薬液を、予め前日から絶食しておいたイヌ(ビーグル犬/雄)に、経口ゾンデを用いて強制的に胃内へ投与した。血液は、薬液投与後15、30分、1、2、4、6及び8時間後に橈側皮静脈よりヘパリン加シリンジを用いて1mL採取した。採取した血液は、直ちに卓上遠心機で14500rpmで45秒間遠心し、血漿1容に対して2容の0.1125%ギ酸含有アセトニトリルを添加して撹拌後、測定時まで-20℃で保存した。
測定日に解凍したサンプルは、撹拌後13000rpmで3分間遠心(4℃)し、上清をフィルターろ過後、LC/MS/MSにて分析した。
LC/MS/MSによる分析は、以下の条件で行った。
測定装置:API-5000(Applied Biosystems社製);
分析カラム:CAPCELL PAK CR(1:4)(4.6mm I.D.×250mm、5μm);
分析カラム温度:40℃;
流速:1mL/分;
移動相:A:5mMギ酸アンモニウム(pH3.9)、B:アセトニトリル(A/B=17/3);
スキャンタイプ:MRM;
Polarity:Negative;
検出(レボドパ):m/z(precursor):196.19;m/z(product):134.99;
DP(Declustering Potential):-60;
CE(Collision Energy):-25;
CXP(Collision Cell Exit Potential):-17。
一般に、塩や結晶形態が異なると、それぞれの溶解性の違い等が血中動態に影響を与え、場合によっては薬効に強弱の差を及ぼすこともあるとされている。前項(1)では実施例5に記載の本発明化合物(塩酸塩、非結晶)が経口投与によって吸収されうることを確認しているが、それ以外の本発明化合物が、経口投与下での吸収性を有するものであるか否かを確認した。
実施例6で製造した本発明化合物((2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩のA型結晶)、実施例9で製造した本発明化合物((2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸のA型結晶)、及び実施例10で製造した本発明化合物((2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸のB型結晶)の各々について、秤量後、0.5w/v%メチルセルロース400を用い、レボドパ換算で6mg/3mLに懸濁し、経口投与用の薬液を調製した。
前項(1)に準じて実施した。ただし投与量は6mg/3mL/kgとした。
[分析試料の調製ならびに分析]
前項(1)に準じて実施した。
[結果]
イヌ動態試験の結果を表6に示す。
上記(1)の結果により示された、本発明化合物を投与することによって与えられるレボドパの平坦な血中濃度推移が、臨床において汎用されているDCI併用下においてどのように変化するかを、DCIとしてカルビドパを用いることにより検討した。
実施例5で製造した本発明化合物とレボドパの各々について、秤量後、媒体を用い、レボドパ換算で1mg/mLに溶解し、経口投与用の薬液を調製した。
この薬液を、予め前日から絶食し、カルビドパ(AKサイエンティフィックInc、70566)の有効量(60mg/kg)を投与したイヌ(ビーグル犬/雄)に、経口ゾンデを用いて強制的に胃内へ投与した。投与量は、レボドパが3mg/kg、本発明化合物(実施例5)が18.6mg/kg(レボドパ換算で6mg/kg)である。血液は、薬液投与後15、30分、1、2、4、6及び8時間後に橈側皮静脈よりヘパリン加シリンジを用いて1mL採取した。採取した血液は、直ちに卓上遠心機で14500rpmで45秒間遠心し、血漿1容に対して2容の0.1125%ギ酸含有アセトニトリルを添加して撹拌後、測定時まで-20℃で保存した。
分析試料の調製ならびに分析は、生物学的実施例1(1)の同項に記載の方法に準じて行った。
イヌに本発明化合物を投与した際の、レボドパの血漿中の濃度推移を図1に示す。
臨床において汎用のDCIであるカルビドパ併用下において、レボドパを投与した場合と本発明化合物を投与した場合のレボドパ血中濃度推移を比較したところ、レボドパを投与した場合の血漿中レボドパ濃度は、投与後15分で2.1μg/mLの最高血漿中濃度(Cmax)に達したあと速やかに下降するものであったが、レボドパの代わりに本発明化合物を投与した場合の血漿中レボドパ濃度は、投与後4時間で漸く1.0μg/mLのCmaxに達したあと緩やかに下降するものであった。
イヌ動態試験で観察された本発明化合物の長時間にわたるレボドパの遊離作用が、ヒトにおいても観察されるか否かを検証する目的で、前記(3)のイヌ動態試験の結果を元にしたヒト血中動態シミュレーションを実施した。
前記(3)で得られたイヌの血中動態試験の値を用いて、ヒトの血中動態をシミュレートする予測モデルを構築した。
本発明化合物のヒト血中動態シミュレーションの結果、ならびに市販のレボドパ・カルビドパ配合剤(シネメット:SINEMET[登録商標])の100mg錠(100mgのレボドパを含有)を服用した場合のレボドパ血中動態に関する文献値(ヨーロピアン・ジャーナル・オブ・クリニカル・ファーマコロジー(Eur. J. Clin. Pharmacol.)、1993年、45巻、419-423頁:V.V.Myllylaら)を図2に示す。
従って、本発明化合物は、ヒトにおいて既存のレボドパ製剤の約3.4倍の長時間にわたって治療域のレボドパを与え、殊、DCI併用下においては、1日2回投与で約16時間にわたって治療域のレボドパを与え得ることがわかった。
変異原性試験
パーキンソン病及び/又はパーキンソン症候群の治療に際しては、レボドパのプロドラッグ体は、数年乃至数十年の長期間にわたって服用され続ける可能性がある。そこで、本発明化合物の変異原性の有無について、哺乳動物細胞を用いた変異原性試験法により、評価を行った。
哺乳動物細胞を用いた変異原性試験法は、日清食品株式会社への委託試験として実施した。NESMAGET法ともよばれる本法は、DNAの修復遺伝子であるp53R2の発現をルシフェラーゼ活性でみるものであり、具体的な実験手技は、特開2005-000024号公報、特許第4243716号公報に記載のものである。当該試験結果の陽性判定に際しては、p53R2のルシフェラーゼ活性が、溶媒対照(0.3%ジメチルスルホキシド)を100%として、200%を超える濃度を陽性と判定した。
本発明化合物(実施例5)、参考例6で製造した化合物、参考例11で製造した化合物、参考例16で製造した化合物、及び参考例40で製造した化合物について、変異原性試験の結果を表7に示す。
6-ヒドロキシドパミン内側前脳束注入モデル
本発明化合物が経口投与後にレボドパに代謝され、パーキンソン病に対して有効性を示すことを確認する目的で、動物を用いた実験的パーキンソン病モデルである6-ヒドロキシドパミン内側前脳束注入モデルにおける本発明化合物の有効性を評価した。
実験には、5週齢で入荷した雄性Crl:CD(SD)IGSラット(日本チャールス・リバー株式会社)を使用した。ラットは実験に供するまで、エコンケージに1ケージあたり4匹以下を収容し、固型飼料CRF-1(オリエンタル酵母株式会社)及び上水道水(給水瓶)を自由摂取させて飼育した。
入荷後、馴化を終えた6週齢のCrl:CD(SD)IGSラットをペントバルビタールナトリウム(ソムノペンチル(登録商標)注射液、35mg/kg、腹腔内投与)で麻酔した。次いで、6-ヒドロキシドパミン(6-OHDA)によるノルエピネフリン神経の傷害を防止する目的で、デシプラミン(25mg/kg)を腹腔内投与し、脳固定装置でラットを保定した。ハンドドリルで目的の部位に小骨窓を開け、針先を45°にカットした30Gカニューレ(ブレインサイエンス・イデア)を用い、パキシノス(Paxinos)とワトソン(Watson)の脳地図に従って、6-OHDAを内側前脳束に注入した(注入部位はブレグマよりA=-4.5mm、L=+1.2mm、V=-7.8mm;注入量:各8μg/4μL/8min/site)。注入後、液の逆流を防ぐために、2分間以上注入針を留置した。その後、穴を瞬間接着剤で塞ぎ、縫合、ヨードチンキ消毒を行った。
6-OHDA注入から2週間後に、病態が惹起されている個体の選別を行った。具体的には、下記のRotation Testを用いて、被験物質としてドパミン受容体アゴニストであるアポモルフィン(0.05mg/kg)を皮下投与した場合に、投与15-20分後の旋回数が、5分間で20回以上の個体のみを病態が惹起されているラットとして選択した。
Rotation testには、口径35cm、底径17cmの黒塗りのボウルの中に、直径30cm、高さ35cmの黒いプラスチックの円筒を立てて置いた装置を用いた(ボウル底部から円筒下端までの曲面の高さを7cmとする)。ラットを1匹につき1台の装置に入れ30分間馴化を行った。その後、被験物質を投与し、再び同じ装置内に戻し、ビデオカメラで撮影した。撮影したビデオ映像を解析し、5分間又は10分間毎の旋回数を測定した。旋回数は同一方向への360°旋回を1回転とした。旋回途中で方向(体の向き、進行方向)が変化した場合、その旋回に関しては無効とした。
病態惹起の確認から1週間以上あけ、実施例5で製造した本発明化合物(レボドパ量として100mg/kg)を経口投与した場合の旋回運動を投与6時間後まで評価した(N=11)。また、レボドパ(30mg/kg)を経口投与した場合の旋回運動についても同様に、投与6時間後まで評価した(N=11、クロスオーバー)。なお、いずれの場合もドパ脱炭酸酵素阻害であるベンセラジド(benserazide)(8mg/kg)を同時に経口投与した。
レボドパ(30mg/kg)投与群ならびに実施例5で製造した本発明化合物(レボドパ量として100mg/kg)投与群の、投与後6時間(360分)までのラットの10分間あたりの旋回数(回)を図1に示した。なお、図中では、レボドパ(30mg/kg)投与群の値をL-dopa(30mg/kg)とし、実施例5で製造した本発明化合物(レボドパ量として100mg/kg)投与群の値をCompound of Ex.5(100mg/kg)として表記した。また、それぞれの値は、各群11匹ずつのラットを用いて得られた値の平均値とその標準誤差である。
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸 5mg含有錠
以下の各成分を常法により混合した後打錠して、一錠中に5mgの活性成分を含有する錠剤1万錠を得た。
・(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸:50g
・カルボキシメチルセルロースカルシウム(崩壊剤):20g
・ステアリン酸マグネシウム(潤滑剤):10g
・微結晶セルロース:920g
製剤例2:
(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸 20mg含有注射剤
以下の各成分を常法により混合した後、溶液を常法により滅菌し、5mLずつアンプルに充填し、常法により凍結乾燥し、1アンプル中20mgの活性成分を含有するアンプル1万本を得た。
・(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸:200g
・マンニトール:20g
・蒸留水:50L
Claims (39)
- (2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物。
- (2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩、又は(2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩である請求項1記載の化合物。
- (2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸の結晶である請求項2記載の化合物。
- 融点が、約177.0℃乃至約181.9℃である請求項3記載の化合物。
- 粉末X線回折スペクトルにおいて、少なくとも約4.03、7.21、9.98、10.72、17.93、及び19.20度2θにピークを有する請求項3記載の化合物。
- 粉末X線回折スペクトルにおいて、約4.03、7.21、9.98、10.72、11.93、12.90、13.48、14.65、15.23、15.99、16.56、17.23、17.93、19.20、20.88、21.66、22.36、22.50、及び24.58度2θにピークを有する請求項5記載の化合物。
- 図4に示される粉末X線回折スペクトルチャートを特徴とする請求項6記載の化合物。
- 示差走査熱量測定において、約148.7℃の発熱ピーク、ならびに約184.7℃、約194.7℃、及び約200.3℃の吸熱ピークを有する請求項3記載の化合物。
- 図5に示される示差走査熱量測定チャートを特徴とする請求項8記載の化合物。
- 融点が、約174.7℃乃至約179.0℃である請求項3記載の化合物。
- 粉末X線回折スペクトルにおいて、少なくとも約4.62度2θにピークを有する請求項3記載の化合物。
- 粉末X線回折スペクトルにおいて、約4.62、8.40、9.54、12.08、15.38、及び18.16度2θにピークを有する請求項11記載の化合物。
- 図7に示される粉末X線回折スペクトルチャートを特徴とする請求項12記載の化合物。
- 示差走査熱量測定において、約183.3℃の発熱ピーク、ならびに約192.2℃、及び約200.8℃の吸熱ピークを有する請求項3記載の化合物。
- 図8に示される示差走査熱量測定チャートを特徴とする請求項14記載の化合物。
- (2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸トシル酸塩の結晶である請求項2記載の化合物。
- 融点が、約132.0℃乃至約136.0℃である請求項16記載の化合物。
- 粉末X線回折スペクトルにおいて、少なくとも約10.97、11.58、14.83、16.36、16.70、19.42、20.58、及び21.69度2θにピークを有する請求項16記載の化合物。
- 粉末X線回折スペクトルにおいて、約5.15、6.97、7.46、10.97、11.58、13.74、14.83、15.20、16.10、16.36、16.70、17.35、18.30、18.83、19.42、19.95、20.58、21.69、22.63、22.84、及び24.00度2θにピークを有する請求項18記載の化合物。
- 図10に示される粉末X線回折スペクトルチャートを特徴とする請求項19記載の化合物。
- 示差走査熱量測定において、約135.95℃の吸熱ピークを有する請求項16記載の化合物。
- 図11に示される示差走査熱量測定チャートを特徴とする請求項21記載の化合物。
- 融点が、約132.3℃乃至約135.3℃である請求項16記載の化合物。
- 粉末X線回折スペクトルにおいて、少なくとも約10.01、11.88、13.87、15.01、15.87、16.07、17.81、18.65、19.17、及び22.11度2θにピークを有する請求項16記載の化合物。
- 粉末X線回折スペクトルにおいて、約4.04、5.04、5.54、6.11、6.60、7.96、8.62、10.01、10.32、11.88、12.88、13.87、15.01、15.87、16.07、16.74、17.17、17.81、18.65、19.17、19.72、20.27、20.93、21.67、22.11、22.56、23.11、23.47、及び24.21度2θにピークを有する請求項24記載の化合物。
- 図13に示される粉末X線回折スペクトルチャートを特徴とする請求項25記載の化合物。
- 示差走査熱量測定において、約134.54℃に吸熱ピークを有する請求項16記載の化合物。
- 図14に示される示差走査熱量測定チャートを特徴とする請求項27記載の化合物。
- (2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸塩酸塩の非結晶である請求項2記載の化合物。
- 融点が、約112.0℃乃至約117.0℃である請求項29記載の化合物。
- 図16に示される粉末X線回折スペクトルチャートを特徴とする請求項29記載の化合物。
- 示差走査熱量測定において、約82.83℃に吸熱ピークを有する請求項29記載の化合物。
- 図17に示される示差走査熱量測定チャートを特徴とする請求項32記載の化合物。
- (2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物を含有してなる医薬組成物。
- パーキンソン病及び/又はパーキンソン症候群の予防及び/又は治療剤である請求項34記載の医薬組成物。
- (2S)-2-アミノ-3-(3,4-ビス((2-(ベンゾイルオキシ)-2-メチルプロパノイル)オキシ)フェニル)プロパン酸、その塩又はその溶媒和物と、芳香族L-アミノ酸デカルボキシラーゼ阻害薬及び/又はカテコール-O-メチル基転移酵素阻害薬とを組み合わせてなる医薬。
- 芳香族L-アミノ酸デカルボキシラーゼ阻害薬が、カルビドパ水和物、又は塩酸ベンセラジドである請求項36記載の医薬。
- カテコール-O-メチル基転移酵素阻害薬が、エンタカポン、トルカポン、ニテカポン、BIA-3-202、又はCGP-28014である請求項36記載の医薬。
- 配合剤である請求項36乃至38のいずれか一項に記載の医薬。
Priority Applications (18)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2818161A CA2818161A1 (en) | 2010-12-02 | 2011-12-01 | Novel compound and medical use thereof |
| JP2012546945A JP5423905B2 (ja) | 2010-12-02 | 2011-12-01 | 新規化合物及びその医薬用途 |
| US13/991,025 US8748485B2 (en) | 2010-12-02 | 2011-12-01 | Compound and medical use thereof |
| KR1020137016942A KR20130121902A (ko) | 2010-12-02 | 2011-12-01 | 신규 화합물 및 그 의약 용도 |
| MX2013006128A MX342798B (es) | 2010-12-02 | 2011-12-01 | Compuesto de ácido (2s)-2-amino-3-(3,4-bis((2-(benzoiloxi)-2-metil propanoil)oxi)-fenil)propanóico y uso médico del mismo. |
| ES11844106.2T ES2529129T3 (es) | 2010-12-02 | 2011-12-01 | Nuevo compuesto y uso médico del mismo |
| PL11844106T PL2647619T3 (pl) | 2010-12-02 | 2011-12-01 | Nowy związek i jego zastosowanie medyczne |
| CN201180057807.3A CN103228618B (zh) | 2010-12-02 | 2011-12-01 | 新化合物及其医药用途 |
| EP11844106.2A EP2647619B1 (en) | 2010-12-02 | 2011-12-01 | Novel compound and medical use thereof |
| NZ612636A NZ612636A (en) | 2010-12-02 | 2011-12-01 | Levodopa prodrugs |
| BR112013013551A BR112013013551A2 (pt) | 2010-12-02 | 2011-12-01 | composto e uso médico do mesmo |
| DK11844106.2T DK2647619T3 (en) | 2010-12-02 | 2011-12-01 | NEW RELATIONSHIP AND MEDICAL USE THEREOF |
| AU2011337596A AU2011337596B2 (en) | 2010-12-02 | 2011-12-01 | Novel compound and medical use thereof |
| RU2013129917/04A RU2570900C2 (ru) | 2010-12-02 | 2011-12-01 | Новое соединение и его применение в медицине |
| IL226672A IL226672A (en) | 2010-12-02 | 2013-05-30 | Prodrug compounds for vodopa, preparations containing them and their uses |
| ZA2013/04012A ZA201304012B (en) | 2010-12-02 | 2013-05-31 | Novel compound and medical use thereof |
| HK13112160.4A HK1184779A1 (en) | 2010-12-02 | 2013-10-29 | Novel compound and medical use thereof |
| US14/197,381 US20140187630A1 (en) | 2010-12-02 | 2014-03-05 | Novel compound and medical use thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010269046 | 2010-12-02 | ||
| JP2010-269046 | 2010-12-02 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/991,025 A-371-Of-International US8748485B2 (en) | 2010-12-02 | 2011-12-01 | Compound and medical use thereof |
| US14/197,381 Division US20140187630A1 (en) | 2010-12-02 | 2014-03-05 | Novel compound and medical use thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012074069A1 true WO2012074069A1 (ja) | 2012-06-07 |
Family
ID=46171994
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2011/077834 WO2012074069A1 (ja) | 2010-12-02 | 2011-12-01 | 新規化合物及びその医薬用途 |
Country Status (21)
| Country | Link |
|---|---|
| US (2) | US8748485B2 (ja) |
| EP (1) | EP2647619B1 (ja) |
| JP (1) | JP5423905B2 (ja) |
| KR (1) | KR20130121902A (ja) |
| CN (1) | CN103228618B (ja) |
| AU (1) | AU2011337596B2 (ja) |
| BR (1) | BR112013013551A2 (ja) |
| CA (1) | CA2818161A1 (ja) |
| DK (1) | DK2647619T3 (ja) |
| ES (1) | ES2529129T3 (ja) |
| HK (1) | HK1184779A1 (ja) |
| IL (1) | IL226672A (ja) |
| MX (1) | MX342798B (ja) |
| MY (1) | MY160914A (ja) |
| NZ (1) | NZ612636A (ja) |
| PL (1) | PL2647619T3 (ja) |
| PT (1) | PT2647619E (ja) |
| RU (1) | RU2570900C2 (ja) |
| TW (1) | TWI534125B (ja) |
| WO (1) | WO2012074069A1 (ja) |
| ZA (1) | ZA201304012B (ja) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111362980B (zh) | 2014-10-21 | 2022-10-18 | 艾伯维公司 | 卡比多巴和左旋多巴前药及它们治疗帕金森病的用途 |
| WO2019097120A1 (en) | 2017-11-16 | 2019-05-23 | Orion Corporation | New use and pharmaceutical dosage forms |
| RU2678977C1 (ru) * | 2018-04-12 | 2019-02-05 | Федеральное государственное автономное образовательное учреждение высшего образования "Белгородский государственный национальный исследовательский университет" (НИУ "БелГУ") | Способ коррекции тремора в эксперименте |
| CN109820862A (zh) * | 2019-02-21 | 2019-05-31 | 山东大学 | D-甘露糖在制备抵抗多巴胺减少和/或提升多巴胺水平产品中的应用 |
Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2868691A (en) | 1956-03-21 | 1959-01-13 | Riker Laboratories Inc | Self-propelling compositions for inhalation therapy containing a salt of isoproterenol or epinephrine |
| US3095355A (en) | 1961-10-12 | 1963-06-25 | Revlon | Aerosol composition |
| DE2153800A1 (ja) | 1970-10-30 | 1972-05-04 | ||
| JPS4872150A (ja) | 1971-12-28 | 1973-09-29 | ||
| JPS4961135A (ja) | 1972-06-17 | 1974-06-13 | ||
| JPS5029527A (ja) | 1972-08-02 | 1975-03-25 | ||
| US3998799A (en) | 1973-11-02 | 1976-12-21 | Interx Research Corporation | Novel, transient pro-drug forms of l-dopa |
| US4065566A (en) | 1975-04-17 | 1977-12-27 | Interx Research Corporation | N-Nicotinoyl-3,4-dinicotinoyloxy-L-phenylalanine and derivatives pharmaceutical compositions and methods containing same |
| EP0728469A2 (en) | 1995-02-17 | 1996-08-28 | Shiseido Company Limited | An endermic liniment |
| JP2005000024A (ja) | 2003-06-10 | 2005-01-06 | Nissin Food Prod Co Ltd | 哺乳動物細胞を用いた変異原性試験法 |
| WO2005121069A1 (en) | 2004-06-04 | 2005-12-22 | Xenoport, Inc. | Levodopa prodrugs, and compositions and uses thereof |
| WO2005121070A1 (en) | 2004-06-04 | 2005-12-22 | Xenoport, Inc. | Levodopa prodrugs, and compositions and uses thereof |
| WO2006119758A2 (de) | 2005-05-13 | 2006-11-16 | Ellneuroxx Ltd. | Derivate von dihydroxyphenylalanin |
| WO2007104959A1 (en) | 2006-03-16 | 2007-09-20 | Proximagen Ltd. | Amino acid derivatives |
| WO2007109882A1 (en) | 2006-03-24 | 2007-10-04 | National Research Council Of Canada | Anti-diabetic cataract compounds and their uses |
| WO2008076458A1 (en) * | 2006-12-21 | 2008-06-26 | Xenoport, Inc. | Levodopa dimethyl-substituted diester prodrugs, compositions, and methods of use |
| WO2008079387A1 (en) | 2006-12-21 | 2008-07-03 | Xenoport, Inc. | Catechol protected levodopa diester prodrugs, compositions, and methods of use |
| WO2009022098A1 (en) | 2007-08-11 | 2009-02-19 | Proximagen Ltd. | Amino acid derivatives |
| JP2009520690A (ja) * | 2005-12-05 | 2009-05-28 | ゼノポート,インコーポレーテッド | レボドパプロドラッグメシラート、その組成物、およびその使用法 |
| WO2009101616A1 (en) | 2008-02-11 | 2009-08-20 | Ramot At Tel Aviv University Ltd. | Novel conjugates for treating neurodegenerative diseases and disorders |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2277575A1 (fr) * | 1974-07-10 | 1976-02-06 | Synthelabo | Nouveaux derives de la dihydroxy-3,4 phenylalanine, leurs sels, leur preparation et les medicaments qui en contiennent |
| CN1175248A (zh) * | 1995-01-13 | 1998-03-04 | 诺瓦提斯公司 | 具有农药性能的4-芳基-和4-杂芳基-5-氧代吡唑啉衍生物 |
| KR100708644B1 (ko) | 2004-02-26 | 2007-04-17 | 삼성에스디아이 주식회사 | 박막 트랜지스터, 이를 구비한 평판 표시장치, 박막트랜지스터의 제조방법, 평판 표시장치의 제조방법, 및도너 시트의 제조방법 |
| JP4961135B2 (ja) | 2005-11-24 | 2012-06-27 | 国立大学法人東京農工大学 | 立体画像記録体 |
| WO2009079387A2 (en) | 2007-12-14 | 2009-06-25 | E. I. Du Pont De Nemours And Company | Organic solvent free pigment dispersant for waterborne electrocoating |
| US8045264B2 (en) | 2009-03-20 | 2011-10-25 | Skyline Displays, Inc. | Projection backwall apparatus and system |
-
2011
- 2011-12-01 CN CN201180057807.3A patent/CN103228618B/zh not_active Expired - Fee Related
- 2011-12-01 RU RU2013129917/04A patent/RU2570900C2/ru not_active IP Right Cessation
- 2011-12-01 BR BR112013013551A patent/BR112013013551A2/pt not_active IP Right Cessation
- 2011-12-01 NZ NZ612636A patent/NZ612636A/en not_active IP Right Cessation
- 2011-12-01 US US13/991,025 patent/US8748485B2/en not_active Expired - Fee Related
- 2011-12-01 TW TW100144155A patent/TWI534125B/zh not_active IP Right Cessation
- 2011-12-01 ES ES11844106.2T patent/ES2529129T3/es active Active
- 2011-12-01 WO PCT/JP2011/077834 patent/WO2012074069A1/ja active Application Filing
- 2011-12-01 JP JP2012546945A patent/JP5423905B2/ja not_active Expired - Fee Related
- 2011-12-01 PT PT118441062T patent/PT2647619E/pt unknown
- 2011-12-01 MY MYPI2013700909A patent/MY160914A/en unknown
- 2011-12-01 KR KR1020137016942A patent/KR20130121902A/ko not_active Application Discontinuation
- 2011-12-01 MX MX2013006128A patent/MX342798B/es active IP Right Grant
- 2011-12-01 PL PL11844106T patent/PL2647619T3/pl unknown
- 2011-12-01 AU AU2011337596A patent/AU2011337596B2/en not_active Ceased
- 2011-12-01 DK DK11844106.2T patent/DK2647619T3/en active
- 2011-12-01 CA CA2818161A patent/CA2818161A1/en not_active Abandoned
- 2011-12-01 EP EP11844106.2A patent/EP2647619B1/en not_active Not-in-force
-
2013
- 2013-05-30 IL IL226672A patent/IL226672A/en not_active IP Right Cessation
- 2013-05-31 ZA ZA2013/04012A patent/ZA201304012B/en unknown
- 2013-10-29 HK HK13112160.4A patent/HK1184779A1/xx not_active IP Right Cessation
-
2014
- 2014-03-05 US US14/197,381 patent/US20140187630A1/en not_active Abandoned
Patent Citations (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2868691A (en) | 1956-03-21 | 1959-01-13 | Riker Laboratories Inc | Self-propelling compositions for inhalation therapy containing a salt of isoproterenol or epinephrine |
| US3095355A (en) | 1961-10-12 | 1963-06-25 | Revlon | Aerosol composition |
| DE2153800A1 (ja) | 1970-10-30 | 1972-05-04 | ||
| JPS4872150A (ja) | 1971-12-28 | 1973-09-29 | ||
| JPS4961135A (ja) | 1972-06-17 | 1974-06-13 | ||
| JPS5029527A (ja) | 1972-08-02 | 1975-03-25 | ||
| US3998799A (en) | 1973-11-02 | 1976-12-21 | Interx Research Corporation | Novel, transient pro-drug forms of l-dopa |
| US4065566A (en) | 1975-04-17 | 1977-12-27 | Interx Research Corporation | N-Nicotinoyl-3,4-dinicotinoyloxy-L-phenylalanine and derivatives pharmaceutical compositions and methods containing same |
| EP0728469A2 (en) | 1995-02-17 | 1996-08-28 | Shiseido Company Limited | An endermic liniment |
| JP4243716B2 (ja) | 2003-06-10 | 2009-03-25 | 日清食品ホールディングス株式会社 | 哺乳動物細胞を用いた変異原性試験法 |
| JP2005000024A (ja) | 2003-06-10 | 2005-01-06 | Nissin Food Prod Co Ltd | 哺乳動物細胞を用いた変異原性試験法 |
| WO2005121069A1 (en) | 2004-06-04 | 2005-12-22 | Xenoport, Inc. | Levodopa prodrugs, and compositions and uses thereof |
| JP2008501704A (ja) * | 2004-06-04 | 2008-01-24 | ゼノポート,インコーポレーテッド | レボドパプロドラッグおよびその組成物ならびにその使用 |
| WO2005121070A1 (en) | 2004-06-04 | 2005-12-22 | Xenoport, Inc. | Levodopa prodrugs, and compositions and uses thereof |
| WO2006119758A2 (de) | 2005-05-13 | 2006-11-16 | Ellneuroxx Ltd. | Derivate von dihydroxyphenylalanin |
| JP2009520690A (ja) * | 2005-12-05 | 2009-05-28 | ゼノポート,インコーポレーテッド | レボドパプロドラッグメシラート、その組成物、およびその使用法 |
| WO2007104959A1 (en) | 2006-03-16 | 2007-09-20 | Proximagen Ltd. | Amino acid derivatives |
| WO2007109882A1 (en) | 2006-03-24 | 2007-10-04 | National Research Council Of Canada | Anti-diabetic cataract compounds and their uses |
| WO2008076458A1 (en) * | 2006-12-21 | 2008-06-26 | Xenoport, Inc. | Levodopa dimethyl-substituted diester prodrugs, compositions, and methods of use |
| WO2008079387A1 (en) | 2006-12-21 | 2008-07-03 | Xenoport, Inc. | Catechol protected levodopa diester prodrugs, compositions, and methods of use |
| WO2009022098A1 (en) | 2007-08-11 | 2009-02-19 | Proximagen Ltd. | Amino acid derivatives |
| WO2009101616A1 (en) | 2008-02-11 | 2009-08-20 | Ramot At Tel Aviv University Ltd. | Novel conjugates for treating neurodegenerative diseases and disorders |
Non-Patent Citations (8)
| Title |
|---|
| D. R. C. ROBERTSON, BR. J. CLIN. PHARM., vol. 28, 1989, pages 61 - 69 |
| MANUELA CONTIN; DIETZ ET AL., CLINICAL PHARMACOLOGY & THERAPEUTICS, vol. 70, 2001, pages 33 - 41 |
| NEUROPSYCHOBIOLOGY, vol. 19, no. 4, 1988, pages 180 - 185 |
| RICHARD C. LAROCK: "Comprehensive Organic Transformations: A Guide to Functional Group Preparations 2nd Edition", 1999, JOHN WILEY & SONS. INC |
| See also references of EP2647619A4 |
| T. W. GREENE: "Protective Groups in Organic Synthesis", 1999, JOHN WILEY & SONS, INC. |
| THERAPEUTIC DRUG MONITORING, vol. 23, 2001, pages 621 - 629 |
| V. V. MYLLYLA, EUR. J. CLIN. PHARMACOL., vol. 45, 1993, pages 419 - 423 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20130121902A (ko) | 2013-11-06 |
| PL2647619T3 (pl) | 2015-08-31 |
| ES2529129T3 (es) | 2015-02-17 |
| CN103228618A (zh) | 2013-07-31 |
| EP2647619A4 (en) | 2013-10-09 |
| PT2647619E (pt) | 2015-02-20 |
| DK2647619T3 (en) | 2015-03-02 |
| MX342798B (es) | 2016-10-13 |
| AU2011337596B2 (en) | 2016-07-07 |
| RU2570900C2 (ru) | 2015-12-20 |
| US20130245074A1 (en) | 2013-09-19 |
| JPWO2012074069A1 (ja) | 2014-05-19 |
| NZ612636A (en) | 2015-01-30 |
| RU2013129917A (ru) | 2015-01-10 |
| TW201307257A (zh) | 2013-02-16 |
| IL226672A (en) | 2016-04-21 |
| CA2818161A1 (en) | 2012-06-07 |
| HK1184779A1 (en) | 2014-01-30 |
| US8748485B2 (en) | 2014-06-10 |
| ZA201304012B (en) | 2014-02-26 |
| EP2647619A1 (en) | 2013-10-09 |
| JP5423905B2 (ja) | 2014-02-19 |
| TWI534125B (zh) | 2016-05-21 |
| MX2013006128A (es) | 2013-07-03 |
| MY160914A (en) | 2017-03-31 |
| AU2011337596A1 (en) | 2013-07-18 |
| US20140187630A1 (en) | 2014-07-03 |
| BR112013013551A2 (pt) | 2016-10-11 |
| EP2647619B1 (en) | 2015-01-21 |
| CN103228618B (zh) | 2016-02-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2022516575A (ja) | ケタミン誘導体およびその組成物 | |
| JP5319920B2 (ja) | ナトリウムおよび/またはカルシウムチャンネル選択的調節因子として活性のある医薬の製造のための(ハロベンジルオキシ)ベンジルアミノ−プロパンアミドの使用 | |
| JPH08510479A (ja) | 眼の炎症障害を処置するための3‐ベンゾイルフェニル酢酸誘導体を含む局所的に投与可能な組成物 | |
| JP5423905B2 (ja) | 新規化合物及びその医薬用途 | |
| JP2012144545A (ja) | アルファ−2−デルタリガンドとセロトニン/ノルアドレナリン再取込み阻害薬を含む組合せ | |
| US20210330615A1 (en) | S-enantiomerically enriched compositions of beta blockers for treating amyotrophic lateral sclerosis | |
| EP3706744B1 (en) | Small molecule inhibitors of shared epitope-calreticulin interactions and methods of use | |
| US20040186177A1 (en) | Alpha2delta ligands for fibromyalgia and other disorders | |
| JPWO2008099781A1 (ja) | アミノアルコール誘導体を有効成分とする脱髄性疾患の治療剤又は予防剤 | |
| JP5435165B2 (ja) | 医薬用途 | |
| AU2003303040B2 (en) | Pregabalin derivatives for the treatment of fibromyalgia and other disorders | |
| JP2005535716A (ja) | 医薬用の置換グリシン誘導体 | |
| CN113082023B (zh) | P2x3抑制剂和p2x4抑制剂的药物组合及其应用 | |
| JP2024041865A (ja) | パ-キンソン病治療用の新規高透過薬物及びその組成物 | |
| WO2005016331A1 (ja) | N−(ベンゾイル)アミノ酸誘導体を有効成分とするニューロパシー性疼痛治療剤 | |
| JP2023502893A (ja) | プロスタグランジンアナログおよびその用途 | |
| KR20080088631A (ko) | 트리아진 유도체 및 PPARα 작용제의 조합물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201180057807.3 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11844106 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2818161 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 226672 Country of ref document: IL |
|
| ENP | Entry into the national phase |
Ref document number: 2012546945 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13991025 Country of ref document: US Ref document number: 12013501117 Country of ref document: PH Ref document number: MX/A/2013/006128 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2013129917 Country of ref document: RU Kind code of ref document: A Ref document number: 20137016942 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011844106 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2011337596 Country of ref document: AU Date of ref document: 20111201 Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013013551 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112013013551 Country of ref document: BR Kind code of ref document: A2 Effective date: 20130531 |