WO2005100496A2 - Chemical-mechanical polishing composition and method for using the same - Google Patents
Chemical-mechanical polishing composition and method for using the same Download PDFInfo
- Publication number
- WO2005100496A2 WO2005100496A2 PCT/US2005/008411 US2005008411W WO2005100496A2 WO 2005100496 A2 WO2005100496 A2 WO 2005100496A2 US 2005008411 W US2005008411 W US 2005008411W WO 2005100496 A2 WO2005100496 A2 WO 2005100496A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polishing composition
- alumina
- chemical
- abrasive
- substrate
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 291
- 238000005498 polishing Methods 0.000 title claims abstract description 286
- 238000000034 method Methods 0.000 title claims abstract description 40
- 239000000758 substrate Substances 0.000 claims abstract description 107
- PNEYBMLMFCGWSK-UHFFFAOYSA-N Alumina Chemical compound [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims abstract description 101
- 229910052751 metal Inorganic materials 0.000 claims abstract description 51
- 239000002184 metal Substances 0.000 claims abstract description 51
- 150000002500 ions Chemical class 0.000 claims abstract description 41
- 239000007788 liquid Substances 0.000 claims abstract description 31
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims abstract description 27
- 239000011777 magnesium Substances 0.000 claims abstract description 27
- 229910052749 magnesium Inorganic materials 0.000 claims abstract description 27
- 229910052788 barium Inorganic materials 0.000 claims abstract description 26
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 claims abstract description 26
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 25
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 claims abstract description 23
- 229910052791 calcium Inorganic materials 0.000 claims abstract description 23
- 239000011575 calcium Substances 0.000 claims abstract description 23
- 229910052712 strontium Inorganic materials 0.000 claims abstract description 23
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 claims abstract description 23
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 claims abstract description 19
- 229910052725 zinc Inorganic materials 0.000 claims abstract description 19
- 239000011701 zinc Substances 0.000 claims abstract description 19
- 229910003460 diamond Inorganic materials 0.000 claims abstract description 12
- 239000010432 diamond Substances 0.000 claims abstract description 12
- 229910010271 silicon carbide Inorganic materials 0.000 claims abstract description 12
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 claims abstract description 12
- NRTOMJZYCJJWKI-UHFFFAOYSA-N Titanium nitride Chemical compound [Ti]#N NRTOMJZYCJJWKI-UHFFFAOYSA-N 0.000 claims abstract description 11
- 229910052580 B4C Inorganic materials 0.000 claims abstract description 10
- INAHAJYZKVIDIZ-UHFFFAOYSA-N boron carbide Chemical compound B12B3B4C32B41 INAHAJYZKVIDIZ-UHFFFAOYSA-N 0.000 claims abstract description 10
- UONOETXJSWQNOL-UHFFFAOYSA-N tungsten carbide Chemical compound [W+]#[C-] UONOETXJSWQNOL-UHFFFAOYSA-N 0.000 claims abstract description 10
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 82
- 229910052697 platinum Inorganic materials 0.000 claims description 41
- 229910000510 noble metal Inorganic materials 0.000 claims description 19
- 238000007517 polishing process Methods 0.000 claims description 12
- 239000000126 substance Substances 0.000 claims description 7
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 claims description 6
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 claims description 4
- 229910052741 iridium Inorganic materials 0.000 claims description 4
- GKOZUEZYRPOHIO-UHFFFAOYSA-N iridium atom Chemical compound [Ir] GKOZUEZYRPOHIO-UHFFFAOYSA-N 0.000 claims description 4
- 229910052703 rhodium Inorganic materials 0.000 claims description 4
- 239000010948 rhodium Substances 0.000 claims description 4
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 claims description 4
- 229910052707 ruthenium Inorganic materials 0.000 claims description 4
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 claims description 3
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 claims description 3
- 229910052737 gold Inorganic materials 0.000 claims description 3
- 239000010931 gold Substances 0.000 claims description 3
- 229910052762 osmium Inorganic materials 0.000 claims description 3
- SYQBFIAQOQZEGI-UHFFFAOYSA-N osmium atom Chemical compound [Os] SYQBFIAQOQZEGI-UHFFFAOYSA-N 0.000 claims description 3
- 229910052763 palladium Inorganic materials 0.000 claims description 3
- 229910052709 silver Inorganic materials 0.000 claims description 3
- 239000004332 silver Substances 0.000 claims description 3
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 14
- 230000000052 comparative effect Effects 0.000 description 14
- -1 heterocyclic organic compound Chemical class 0.000 description 13
- 239000010410 layer Substances 0.000 description 12
- 230000007797 corrosion Effects 0.000 description 10
- 238000005260 corrosion Methods 0.000 description 10
- 230000001590 oxidative effect Effects 0.000 description 10
- 239000002245 particle Substances 0.000 description 10
- 229910021645 metal ion Inorganic materials 0.000 description 9
- 239000003112 inhibitor Substances 0.000 description 8
- 239000000463 material Substances 0.000 description 8
- 150000001875 compounds Chemical class 0.000 description 7
- 229910001629 magnesium chloride Inorganic materials 0.000 description 7
- 150000003839 salts Chemical class 0.000 description 7
- 239000002253 acid Substances 0.000 description 6
- 239000000654 additive Substances 0.000 description 6
- 239000002518 antifoaming agent Substances 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- 230000003115 biocidal effect Effects 0.000 description 5
- 239000003139 biocide Substances 0.000 description 5
- 239000002738 chelating agent Substances 0.000 description 5
- 239000008139 complexing agent Substances 0.000 description 5
- 230000033001 locomotion Effects 0.000 description 5
- 238000004519 manufacturing process Methods 0.000 description 5
- WDIHJSXYQDMJHN-UHFFFAOYSA-L barium chloride Chemical compound [Cl-].[Cl-].[Ba+2] WDIHJSXYQDMJHN-UHFFFAOYSA-L 0.000 description 4
- 229910001626 barium chloride Inorganic materials 0.000 description 4
- 150000007942 carboxylates Chemical class 0.000 description 4
- 229910044991 metal oxide Inorganic materials 0.000 description 4
- 150000004706 metal oxides Chemical class 0.000 description 4
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 4
- 239000004094 surface-active agent Substances 0.000 description 4
- JIAARYAFYJHUJI-UHFFFAOYSA-L zinc dichloride Chemical compound [Cl-].[Cl-].[Zn+2] JIAARYAFYJHUJI-UHFFFAOYSA-L 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical group [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 229910052782 aluminium Inorganic materials 0.000 description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 3
- 239000002736 nonionic surfactant Substances 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 238000007254 oxidation reaction Methods 0.000 description 3
- 239000006179 pH buffering agent Substances 0.000 description 3
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical class OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 description 3
- 239000004065 semiconductor Substances 0.000 description 3
- BNGXYYYYKUGPPF-UHFFFAOYSA-M (3-methylphenyl)methyl-triphenylphosphanium;chloride Chemical compound [Cl-].CC1=CC=CC(C[P+](C=2C=CC=CC=2)(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 BNGXYYYYKUGPPF-UHFFFAOYSA-M 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- 150000001242 acetic acid derivatives Chemical class 0.000 description 2
- 150000003863 ammonium salts Chemical class 0.000 description 2
- 229910001422 barium ion Inorganic materials 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- YCIMNLLNPGFGHC-UHFFFAOYSA-N catechol Chemical compound OC1=CC=CC=C1O YCIMNLLNPGFGHC-UHFFFAOYSA-N 0.000 description 2
- 238000005229 chemical vapour deposition Methods 0.000 description 2
- 150000001860 citric acid derivatives Chemical class 0.000 description 2
- 239000004020 conductor Substances 0.000 description 2
- 230000007547 defect Effects 0.000 description 2
- 239000008367 deionised water Substances 0.000 description 2
- 229910021641 deionized water Inorganic materials 0.000 description 2
- 238000000151 deposition Methods 0.000 description 2
- 239000003989 dielectric material Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 125000000524 functional group Chemical group 0.000 description 2
- LNTHITQWFMADLM-UHFFFAOYSA-N gallic acid Chemical compound OC(=O)C1=CC(O)=C(O)C(O)=C1 LNTHITQWFMADLM-UHFFFAOYSA-N 0.000 description 2
- YBMRDBCBODYGJE-UHFFFAOYSA-N germanium dioxide Chemical compound O=[Ge]=O YBMRDBCBODYGJE-UHFFFAOYSA-N 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 150000002739 metals Chemical class 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 238000012544 monitoring process Methods 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 229920000620 organic polymer Polymers 0.000 description 2
- 125000005498 phthalate group Chemical class 0.000 description 2
- 238000005240 physical vapour deposition Methods 0.000 description 2
- 238000000623 plasma-assisted chemical vapour deposition Methods 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 239000011592 zinc chloride Substances 0.000 description 2
- 235000005074 zinc chloride Nutrition 0.000 description 2
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- 125000001399 1,2,3-triazolyl group Chemical group N1N=NC(=C1)* 0.000 description 1
- TUSDEZXZIZRFGC-UHFFFAOYSA-N 1-O-galloyl-3,6-(R)-HHDP-beta-D-glucose Natural products OC1C(O2)COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC1C(O)C2OC(=O)C1=CC(O)=C(O)C(O)=C1 TUSDEZXZIZRFGC-UHFFFAOYSA-N 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- KAESVJOAVNADME-UHFFFAOYSA-N 1H-pyrrole Natural products C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 1
- NSPMIYGKQJPBQR-UHFFFAOYSA-N 4H-1,2,4-triazole Chemical compound C=1N=CNN=1 NSPMIYGKQJPBQR-UHFFFAOYSA-N 0.000 description 1
- KLSJWNVTNUYHDU-UHFFFAOYSA-N Amitrole Chemical group NC1=NC=NN1 KLSJWNVTNUYHDU-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- OCUCCJIRFHNWBP-IYEMJOQQSA-L Copper gluconate Chemical class [Cu+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O OCUCCJIRFHNWBP-IYEMJOQQSA-L 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical class OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 239000001263 FEMA 3042 Substances 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical class OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 1
- JLVVSXFLKOJNIY-UHFFFAOYSA-N Magnesium ion Chemical compound [Mg+2] JLVVSXFLKOJNIY-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- LRBQNJMCXXYXIU-PPKXGCFTSA-N Penta-digallate-beta-D-glucose Natural products OC1=C(O)C(O)=CC(C(=O)OC=2C(=C(O)C=C(C=2)C(=O)OC[C@@H]2[C@H]([C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(OC(=O)C=3C=C(O)C(O)=C(O)C=3)C(O)=C(O)C=2)O)=C1 LRBQNJMCXXYXIU-PPKXGCFTSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 239000003082 abrasive agent Substances 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 125000005595 acetylacetonate group Chemical group 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 150000001414 amino alcohols Chemical class 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- 239000002280 amphoteric surfactant Substances 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- QRUDEWIWKLJBPS-UHFFFAOYSA-N benzotriazole Chemical compound C1=CC=C2N[N][N]C2=C1 QRUDEWIWKLJBPS-UHFFFAOYSA-N 0.000 description 1
- 239000012964 benzotriazole Substances 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical class O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- 150000001728 carbonyl compounds Chemical class 0.000 description 1
- 239000003093 cationic surfactant Substances 0.000 description 1
- CETPSERCERDGAM-UHFFFAOYSA-N ceric oxide Chemical compound O=[Ce]=O CETPSERCERDGAM-UHFFFAOYSA-N 0.000 description 1
- 229910000422 cerium(IV) oxide Inorganic materials 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- QDOXWKRWXJOMAK-UHFFFAOYSA-N dichromium trioxide Chemical compound O=[Cr]O[Cr]=O QDOXWKRWXJOMAK-UHFFFAOYSA-N 0.000 description 1
- 150000002009 diols Chemical class 0.000 description 1
- QLBHNVFOQLIYTH-UHFFFAOYSA-L dipotassium;2-[2-[bis(carboxymethyl)amino]ethyl-(carboxylatomethyl)amino]acetate Chemical compound [K+].[K+].OC(=O)CN(CC([O-])=O)CCN(CC(O)=O)CC([O-])=O QLBHNVFOQLIYTH-UHFFFAOYSA-L 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 229940058180 edetate dipotassium anhydrous Drugs 0.000 description 1
- 229940048820 edetates Drugs 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 230000003628 erosive effect Effects 0.000 description 1
- 125000003916 ethylene diamine group Chemical group 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 229910021485 fumed silica Inorganic materials 0.000 description 1
- 229940074391 gallic acid Drugs 0.000 description 1
- 235000004515 gallic acid Nutrition 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 239000011229 interlayer Substances 0.000 description 1
- MGIYRDNGCNKGJU-UHFFFAOYSA-N isothiazolinone Chemical compound O=C1C=CSN1 MGIYRDNGCNKGJU-UHFFFAOYSA-N 0.000 description 1
- 150000003893 lactate salts Chemical class 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 229910001425 magnesium ion Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 150000004701 malic acid derivatives Chemical class 0.000 description 1
- 238000001465 metallisation Methods 0.000 description 1
- 238000004377 microelectronic Methods 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 150000002823 nitrates Chemical class 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 239000006259 organic additive Substances 0.000 description 1
- 150000002894 organic compounds Chemical group 0.000 description 1
- 150000003891 oxalate salts Chemical class 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 238000007747 plating Methods 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- IWZKICVEHNUQTL-UHFFFAOYSA-M potassium hydrogen phthalate Chemical compound [K+].OC(=O)C1=CC=CC=C1C([O-])=O IWZKICVEHNUQTL-UHFFFAOYSA-M 0.000 description 1
- OTYBMLCTZGSZBG-UHFFFAOYSA-L potassium sulfate Chemical compound [K+].[K+].[O-]S([O-])(=O)=O OTYBMLCTZGSZBG-UHFFFAOYSA-L 0.000 description 1
- 229910052939 potassium sulfate Inorganic materials 0.000 description 1
- 235000011151 potassium sulphates Nutrition 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 229940079877 pyrogallol Drugs 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 229910001631 strontium chloride Inorganic materials 0.000 description 1
- AHBGXTDRMVNFER-UHFFFAOYSA-L strontium dichloride Chemical compound [Cl-].[Cl-].[Sr+2] AHBGXTDRMVNFER-UHFFFAOYSA-L 0.000 description 1
- 150000003890 succinate salts Chemical class 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- LRBQNJMCXXYXIU-NRMVVENXSA-N tannic acid Chemical compound OC1=C(O)C(O)=CC(C(=O)OC=2C(=C(O)C=C(C=2)C(=O)OC[C@@H]2[C@H]([C@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(OC(=O)C=4C=C(O)C(O)=C(O)C=4)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(OC(=O)C=3C=C(O)C(O)=C(O)C=3)C(O)=C(O)C=2)O)=C1 LRBQNJMCXXYXIU-NRMVVENXSA-N 0.000 description 1
- 235000015523 tannic acid Nutrition 0.000 description 1
- 229940033123 tannic acid Drugs 0.000 description 1
- 229920002258 tannic acid Polymers 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 1
- MZLGASXMSKOWSE-UHFFFAOYSA-N tantalum nitride Chemical compound [Ta]#N MZLGASXMSKOWSE-UHFFFAOYSA-N 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 150000003892 tartrate salts Chemical class 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 238000012876 topography Methods 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09G—POLISHING COMPOSITIONS; SKI WAXES
- C09G1/00—Polishing compositions
- C09G1/02—Polishing compositions containing abrasives or grinding agents
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01F—COMPOUNDS OF THE METALS BERYLLIUM, MAGNESIUM, ALUMINIUM, CALCIUM, STRONTIUM, BARIUM, RADIUM, THORIUM, OR OF THE RARE-EARTH METALS
- C01F7/00—Compounds of aluminium
- C01F7/02—Aluminium oxide; Aluminium hydroxide; Aluminates
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K3/00—Materials not provided for elsewhere
- C09K3/14—Anti-slip materials; Abrasives
- C09K3/1454—Abrasive powders, suspensions and pastes for polishing
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L21/00—Processes or apparatus adapted for the manufacture or treatment of semiconductor or solid state devices or of parts thereof
- H01L21/02—Manufacture or treatment of semiconductor devices or of parts thereof
- H01L21/04—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer
- H01L21/18—Manufacture or treatment of semiconductor devices or of parts thereof the devices having potential barriers, e.g. a PN junction, depletion layer or carrier concentration layer the devices having semiconductor bodies comprising elements of Group IV of the Periodic Table or AIIIBV compounds with or without impurities, e.g. doping materials
- H01L21/30—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26
- H01L21/302—Treatment of semiconductor bodies using processes or apparatus not provided for in groups H01L21/20 - H01L21/26 to change their surface-physical characteristics or shape, e.g. etching, polishing, cutting
- H01L21/306—Chemical or electrical treatment, e.g. electrolytic etching
- H01L21/30625—With simultaneous mechanical treatment, e.g. mechanico-chemical polishing
Definitions
- This invention pertains to a chemical-mechanical polishing composition and a method of polishing a substrate using the same.
- PVD physical vapor deposition
- CVD chemical vapor deposition
- PECVD plasma-enhanced chemical vapor deposition
- ECP electrochemical plating
- Planarizing a surface is a process where material is removed from the surface of the substrate to form a generally even, planar surface. Planarization is useful in removing undesired surface topography and surface defects, such as rough surfaces, agglomerated materials, crystal lattice damage, scratches, and contaminated layers or materials. Planarization is also useful in forming features on a substrate by removing excess deposited material used to fill the features and to provide an even surface for subsequent levels of metallization and processing.
- CMP chemical-mechanical planarization
- a substrate carrier or polishing head is mounted on a carrier assembly and positioned in contact with a polishing pad in a CMP apparatus.
- the carrier assembly provides a controllable pressure to the substrate, urging the substrate against the polishing pad.
- the pad is moved relative to the substrate by an external driving force. The relative movement of the pad and substrate serves to abrade the surface of the substrate to remove a portion of the material from the substrate surface, thereby polishing the substrate.
- the polishing of the substrate by the relative movement of the pad and the substrate typically is further aided by the chemical activity of the polishing composition and/or the mechanical activity of an abrasive suspended in the polishing composition.
- DRAMs dynamic random access memory
- FeRAMs ferrroelectric random access memory
- the use of noble metals can provide increased performance in such devices, the use of noble metals can — and often does — present unique manufacturing challenges.
- noble metals are mechanically hard and chemically resistant. Indeed, the term noble metal was adopted to describe the metals' superior resistance to corrosion and oxidation. This mechanical hardness and relative chemical resistance make noble metals much more difficult to efficiently polish using conventional chemical-mechanical polishing compositions and techniques.
- WO 01/44396 Al discloses a polishing composition for noble metals comprising sulfur-containing compounds, abrasive particles, and water-soluble organic additives which purportedly improve the dispersion of the abrasive particles and enhance metal removal rates and selectivity.
- each of the aforementioned chemical-mechanical polishing compositions might be capable of polishing noble metals more efficiently than conventional chemical- mechanical polishing compositions
- the compositions also can produce defects on the surface of the substrate that can negatively impact the performance of any integrated circuit later manufactured from the substrate.
- the halogen- and sulfur-containing compounds utilized in the aforementioned polishing compositions can be highly toxic (which can complicate the polishing process by requiring specialized handling equipment and/or procedures), expensive to produce, and/or expensive to properly dispose of in accordance with environmental regulations.
- the invention provides such a chemical-mechanical polishing composition and related method of using the same to polish a substrate.
- the invention provides a chemical-mechanical polishing composition
- a chemical-mechanical polishing composition comprising: (a) an abrasive comprising ⁇ -alumina, (b) 0.05 to 50 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of calcium, strontium, barium, and mixtures thereof, based on the total weight of the polishing composition, and (c) a liquid carrier comprising water.
- the invention also provides a chemical-mechanical polishing composition
- a chemical-mechanical polishing composition comprising: (a) an abrasive selected from the group consisting of ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, diamond, boron carbide, silicon carbide, tungsten carbide, titanium nitride, and mixtures thereof, (b) 0.05 to 3.5 mmol/kg of ions of at least one metal selected from the group consisting of calcium, strontium, barium, magnesium, zinc, and mixtures thereof, based on the total weight of the polishing composition, and (c) a liquid carrier comprising water.
- the invention further provides a method of polishing a substrate comprising the steps of: (a) providing a substrate, (b) providing a chemical-mechanical polishing composition comprising: (i) an abrasive comprising ⁇ -alumina, (ii) 0.05 to 50 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of calcium, strontium, barium, and mixtures thereof, based on the total weight of the polishing composition, and (iii) a liquid carrier comprising water, (c) applying the chemical- mechanical polishing composition to at least a portion of the substrate, and (d) abrading at least a portion of the substrate with the polishing composition to polish the substrate.
- a chemical-mechanical polishing composition comprising: (i) an abrasive comprising ⁇ -alumina, (ii) 0.05 to 50 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of calcium, strontium, barium,
- the invention additionally provides a method of polishing a substrate comprising the steps of: (a) providing a substrate, (b) providing a chemical-mechanical polishing composition comprising: (i) an abrasive selected from the group consisting of ⁇ - alumina, ⁇ -alumina, ⁇ -alumina, diamond, boron carbide, silicon carbide, tungsten carbide, titanium nitride, and mixtures thereof, (ii) 0.05 to 3.5 mmol/kg of ions of at least one metal selected from the group consisting of calcium, strontium, barium, magnesium, zinc, and mixtures thereof, based on the total weight of the polishing composition, and (iii) a liquid carrier comprising water, (c) applying the chemical- mechanical polishing composition to at least a portion of the substrate, and (d) abrading at least a portion of the substrate with the polishing composition to polish the substrate.
- a chemical-mechanical polishing composition comprising: (i) an abrasive selected from the group consisting
- the invention provides a chemical-mechanical polishing composition
- a chemical-mechanical polishing composition comprising (a) an abrasive, (b) ions of at least one metal selected from the group consisting of calcium, strontium, barium, magnesium, zinc, and mixtures thereof, and (c) a liquid carrier comprising water.
- the chemical-mechanical polishing composition comprises (a) an abrasive comprising ⁇ -alumina, (b) 0.05 to 50 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of calcium, strontium, barium, and mixtures thereof, based on the total weight of the polishing composition, and (c) a liquid carrier comprising water.
- the chemical-mechanical polishing composition comprises (a) an abrasive comprising ⁇ - alumina, (b) 0.05 to 3.5 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of magnesium, zinc, and mixtures thereof, based on the total weight of the polishing composition, and (c) a liquid carrier comprising water.
- the chemical-mechanical polishing composition comprises (a) an abrasive selected from the group consisting of ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, diamond, boron carbide, silicon carbide, tungsten carbide, titanium nitride, and mixtures thereof, (b) 0.05 to 3.5 mmol/kg of ions of at least one metal selected from the group consisting of calcium, strontium, barium, magnesium, zinc, and mixtures thereof, based on the total weight of the polishing composition, and (c) a liquid carrier comprising water.
- the polishing composition comprises an abrasive, and, in certain embodiments, the abrasive comprises ⁇ -alumina.
- alumina i.e., aluminum oxide
- alumina exists is several different crystalline phases, which include ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, and p-alumina.
- the ⁇ -alumina when present in the abrasive, can be present in any suitable form.
- the ⁇ -alumina can be present in the form of distinct abrasive particles consisting essentially of, or consisting of, ⁇ -alumina, or the ⁇ -alumina can be present in abrasive particles comprising ⁇ -alumina and other suitable abrasive components (e.g., a metal oxide, such as fumed alumina).
- suitable abrasive components e.g., a metal oxide, such as fumed alumina.
- the abrasive when the abrasive comprises ⁇ -alumina, the abrasive preferably comprises 10 wt.% or more, more preferably 20 wt.% or more, still more preferably 30 wt.% or more, even more preferably 40 wt.% or more, and most preferably 50 wt.% or more (e.g., 55 wt.% or more, or 60 wt.% or more) ⁇ -alumina based on the total weight of the abrasive.
- 10 wt.% or more more preferably 20 wt.% or more, still more preferably 30 wt.% or more, even more preferably 40 wt.% or more, and most preferably 50 wt.% or more (e.g., 55 wt.% or more, or 60 wt.% or more) ⁇ -alumina based on the total weight of the abrasive.
- the invention also provides a chemical-mechanical polishing composition
- a chemical-mechanical polishing composition comprising an abrasive selected from the group consisting of ⁇ -alumina, ⁇ - alumina, ⁇ -alumina, ⁇ -alumina, diamond, boron carbide, silicon carbide, tungsten carbide, titanium nitride, and mixtures thereof.
- the abrasive preferably is selected from the group consisting of ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, diamond, silicon carbide, titanium nitride, and mixtures thereof.
- the abrasive is selected from the group consisting of ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, diamond, silicon carbide, and mixtures thereof. Most preferably, the abrasive is selected from the group consisting of ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, and mixtures thereof. [0016] In addition to the aforementioned abrasive components, the abrasive of the polishing composition can further comprise other suitable abrasive components.
- Suitable additional abrasive components include, but are not limited to, metal oxide abrasives, such as other forms of alumina (e.g., fumed alumina), silica (e.g., colloidally dispersed condensation-polymerized silica, fumed or pyrogenic silica, and precipitated silica), ceria, titania, zirconia, chromia, iron oxide, germania, magnesia, co-formed products thereof, and combinations thereof.
- metal oxide abrasives such as other forms of alumina (e.g., fumed alumina), silica (e.g., colloidally dispersed condensation-polymerized silica, fumed or pyrogenic silica, and precipitated silica), ceria, titania, zirconia, chromia, iron oxide, germania, magnesia, co-formed products thereof, and combinations thereof.
- the abrasive can be present in the polishing composition in any suitable amount.
- the abrasive is present in the polishing composition in an amount of 0.01 wt.% or more, preferably 0.05 wt.% or more, more preferably 0.1 wt.% or more, still more preferably 0.5 wt.% or more, and most preferably 1 wt.%> or more, based on the total weight of the polishing composition.
- the abrasive typically is present in the polishing composition in an amount of 25 wt.% or less, preferably 20 wt.% or less, more preferably 15 wt.% or less, still more preferably 10 wt.% or less, and most preferably 5 wt.%> or less, based on the total weight of the polishing composition.
- the polishing composition comprises ions of at least one metal selected from the group consisting of calcium, strontium, barium, and mixtures thereof.
- the polishing composition comprises ions of at least one metal selected from the group consisting of magnesium, zinc, and mixtures thereof.
- the polishing composition comprises ions of at least one metal selected from the group consisting of calcium, strontium, barium, magnesium, zinc, and mixtures thereof.
- the ions of the metal(s) contained in the polishing composition can be derived from any suitable source.
- the ions of the metal(s) contained in the polishing composition are derived from at least one water-soluble metal salt.
- the ions of the metal can be present in the polishing composition in any suitable amount.
- the ions of the metal are present in the polishing composition in an amount of 0.05 millimoles per kilogram (mmol/kg) or more, preferably 0.06 mmol/kg or more, more preferably 0.07 mmol/kg or more, and most preferably 1 mmol/kg or more, based on the total weight of the polishing composition.
- the ions of the metal generally are present in the polishing composition in an amount of 50 mmol/kg or less, preferably 40 mmol/kg or less, more preferably 30 mmol/kg or less, and most preferably 20 mmol/kg or less (e.g., 10 mmol/kg or less, 5 mmol/kg or less, or 3.5 mmol/kg or less), based on the total weight of the polishing composition.
- the polishing composition comprises ions of at least one metal selected from the group consisting of calcium, strontium, barium, and mixtures thereof
- the ions of the metal preferably are present in the polishing composition in an amount of 0.05 to 50 millimoles per kilogram (mmol/kg), more preferably 0.05 to 40 mmol/kg (e.g., 0.05 to 30 mmol/kg, 0.05 to 25 mmol/kg, 0.05 to 20 mmol/kg, or 0.05 to 15 mmol/kg), still more preferably 0.05 to 10 mmol/kg, and most preferably 0.05 to 5 mmol/kg, based on the total weight of the polishing composition.
- the polishing composition comprises ions of at least one metal selected from the group consisting of magnesium, zinc, and mixtures thereof, or the polishing composition comprises an abrasive selected from the group consisting of ⁇ -alumina, ⁇ - alumina, ⁇ -alumina, ⁇ -alumina, diamond, boron carbide, silicon carbide, tungsten carbide, titanium nitride, and mixtures thereof
- the ions of the metal preferably are present in the polishing composition in an amount of 0.05 to 3.5 millimoles per kilogram (mmol/kg), more preferably 0.05 to 3.4 mmol/kg (e.g., 0.05 to 3.3 mmol/kg, 0.05 to 3.25 mmol/kg, 0.05 to 3.2 mmol/kg, or 0.05 to 3.1 mmol/kg), and most preferably 0.05 to 3 mmol/kg, based on the total weight of the polishing composition.
- a liquid carrier is used to facilitate the application of the abrasive, metal ions, and any other additives to the surface of a suitable substrate to be polished or planarized.
- the liquid carrier can be any suitable liquid carrier.
- the liquid carrier comprises water.
- the water is deionized water.
- the liquid carrier can further comprise a suitable water-miscible solvent.
- the liquid carrier consists essentially of, or consists of, water, more preferably deionized water.
- the polishing composition can have any suitable pH (e.g., 1 to 13).
- the polishing composition has a pH of 1 to 7, more preferably 2 to 5.
- the pH of the chemical-mechanical polishing system can be achieved and/or maintained by any suitable means. More specifically, the polishing composition can further comprise a pH adjustor, a pH buffering agent, or a combination thereof.
- the pH adjustor can be any suitable pH- adjusting compound.
- the pH adjustor can be potassium hydroxide, sodium hydroxide, ammonium hydroxide, or a combination thereof.
- the pH buffering agent can be any suitable buffering agent, for example, phosphates, acetates, borates, ammonium salts, and the like.
- the chemical-mechanical polishing system can comprise any suitable amount of a pH adjustor and/or a pH buffering agent, provided such amount is sufficient to achieve and/or maintain the pH of the polishing system within the ranges set forth herein.
- the polishing composition can further comprise an acid.
- the acid can be any suitable acid, such as an inorganic or an organic acid, or a combination thereof.
- the polishing composition can comprise an inorganic acid selected from the group consisting of nitric acid, phosphoric acid, sulfuric acid, salts thereof, and combinations thereof.
- the polishing composition can comprise (alternatively or in addition to the inorganic acid) an organic acid selected from the group consisting of oxalic acid, malic acid, malonic acid, tartaric acid, acetic acid, lactic acid, propionic acid, phthalic acid, benzoic acid, citric acid, succinic acid, salts thereof, and combinations thereof.
- the polishing composition also can comprise a corrosion inhibitor (i.e., a film- forming agent).
- the corrosion inhibitor can be any suitable corrosion inhibitor.
- the corrosion inhibitor is an organic compound containing a heteroatom-containing functional group.
- the corrosion inhibitor can be a heterocyclic organic compound with at least one 5- or 6-member heterocyclic ring as the active functional group, wherein the heterocyclic ring contains at least one nitrogen atom, for example, an azole compound.
- the corrosion inhibitor contains at least one azole group.
- the corrosion inhibitor is selected from the group consisting of 1,2,3-triazole, 1,2,4-triazole, benzotriazole, benzimidazole, benzothiazole, and mixtures thereof.
- the amount of corrosion inhibitor used in the polishing system typically is 0.0001 wt.%> to 3 wt.%) (preferably 0.001 wt.% to 2 wt.%) based on the total weight of the polishing composition.
- the polishing composition optionally further comprises a chelating or complexing agent.
- the complexing agent is any suitable chemical additive that enhances the removal rate of the substrate layer being removed.
- Suitable chelating or complexing agents can include, for example, carbonyl compounds (e.g., acetylacetonates, and the like), simple carboxylates (e.g., acetates, aryl carboxylates, and the like), carboxylates containing one or more hydroxyl groups (e.g., glycolates, lactates, gluconates, gallic acid and salts thereof, and the like), di-, tri-, and poly-carboxylates (e.g., oxalates, phthalates, citrates, succinates, tartrates, malates, edetates (e.g., dipotassium EDTA), mixtures thereof, and the like), carboxylates containing one or more sulfonic and/or phosphonic groups, and the like.
- Suitable chelating or complexing agents also can include, for example, di-, tri-, or polyalcohols (e.g., ethylene glycol, pyrocatechol, pyrogallol, tannic acid, and the like) and amine-containing compounds (e.g., ammonia, amino acids, amino alcohols, di-, tri-, and polyamines, and the like).
- amine-containing compounds e.g., ammonia, amino acids, amino alcohols, di-, tri-, and polyamines, and the like.
- the choice of chelating or complexing agent will depend on the type of substrate layer being removed.
- a salt e.g., a metal salt, an ammonium salt, or the like
- an acid or as a partial salt.
- citrates include citric acid, as well as mono-, di-, and tri-salts thereof; phthalates include phthalic acid, as well as mono-salts (e.g., potassium hydrogen phthalate) and di-salts thereof; perchlorates include the corresponding acid (i.e., perchloric acid), as well as salts thereof.
- certain compounds or reagents may perform more than one function. For example, some compounds can function both as a chelating agent and an oxidizing agent (e.g., certain ferric nitrates and the like).
- the polishing composition can further comprise a surfactant.
- Suitable surfactants can include, for example, cationic surfactants, anionic surfactants, nonionic surfactants, amphoteric surfactants, mixtures thereof, and the like.
- the polishing composition comprises a nonionic surfactant.
- a suitable nonionic surfactant is an ethylenediamine polyoxyethylene surfactant.
- the amount of surfactant typically is 0.0001 wt.% to 1 wt.% (preferably 0.001 wt.% to 0.1 wt.%, and more preferably 0.005 wt.% to 0.05 wt.%) based on the total weight of the polishing composition.
- the polishing composition can further comprise an antifoaming agent.
- the anti- foaming agent can be any suitable anti-foaming agent. Suitable antifoaming agents include, but are not limited to, silicon-based and acetylenic diol-based antifoaming agents.
- the amount of anti-foaming agent present in the polishing composition typically is 10 ppm to 140 ppm.
- the polishing composition can also comprise a biocide.
- the biocide can be any suitable biocide, for example an isothiazolinone biocide.
- the amount of biocide used in the polishing composition typically is 1 to 50 ppm, preferably 10 to 20 ppm.
- the polishing composition preferably is colloidally stable.
- colloid refers to the suspension of the abrasive (e.g., abrasive particles) in the liquid carrier.
- Colloidal stability refers to the maintenance of that suspension through time.
- a polishing composition is considered colloidally stable if, when the polishing composition is placed into a 100 ml graduated cylinder and allowed to stand unagitated for a time of 2 hours, the difference between the concentration of abrasive (e.g., abrasive particles) in the bottom 50 ml of the graduated cylinder ([B] in terms of g/ml) and the concentration of abrasive (e.g., abrasive particles) in the top 50 ml of the graduated cylinder ([T] in terms of g/ml) divided by the initial concentration of abrasive (e.g., abrasive particles) in the polishing composition ([C] in terms of g/ml) is less than or equal to 0.5 (i.e., ⁇ [B] - [T] ⁇ /[C] ⁇ 0.5).
- abrasive e.g., abrasive particles
- the value of [B]-[T]/[C] is less than or equal to 0.3, more preferably is less than or equal to 0.1, even more preferably is less than or equal to 0.05, and most preferably is less than or equal to 0.01.
- the average particle size of the polishing composition preferably remains essentially unchanged throughout the useful life of the polishing composition.
- the average particle size of the polishing composition preferably increases by less than 40% (e.g., less than 35%, less than 30%, less than 25%>, less than 20%, less than 15%>, or less than 10%)) throughout the useful life of the polishing composition (e.g., 90 days or more, 180 days or more, or 365 days or more).
- the invention further provides methods of polishing a substrate with the polishing compositions as described herein.
- the methods generally comprise the steps of (i) providing a substrate, (ii) providing a polishing composition as described herein, (iii) applying the polishing composition to a portion of the substrate, and (iv) abrading a portion of the substrate to polish the substrate.
- the method of polishing a substrate comprises the steps of: (a) providing a substrate, (b) providing a chemical-mechanical polishing composition comprising: (i) an abrasive comprising ⁇ -alumina, (ii) 0.05 to 50 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of calcium, strontium, barium, and mixtures thereof, based on the total weight of the polishing composition, and (iii) a liquid carrier comprising water, (c) applying the chemical-mechanical polishing composition to at least a portion of the substrate, and (d) abrading at least a portion of the substrate with the polishing composition to polish the substrate.
- a chemical-mechanical polishing composition comprising: (i) an abrasive comprising ⁇ -alumina, (ii) 0.05 to 50 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of calcium, strontium, barium, and mixtures thereof,
- the polishing composition utilized in this method embodiment of the invention comprises: (a) an abrasive comprising ⁇ -alumina, (b) 0.05 to 50 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of calcium, strontium, barium, and mixtures thereof, based on the total weight of the polishing composition, and (c) a liquid carrier comprising water.
- the other characteristics of the chemical-mechanical polishing composition utilized in this method embodiment of the invention e.g., the amount of abrasive, the liquid carrier, the pH, and other suitable additives
- the chemical-mechanical polishing composition of the invention can be the same as set forth above for the chemical-mechanical polishing composition of the invention.
- the method of polishing a substrate comprises the steps of: (a) providing a substrate, (b) providing a chemical-mechanical polishing composition comprising: (i) an abrasive comprising ⁇ -alumina, (ii) 0.05 to 3.5 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of magnesium, zinc, and mixtures thereof, based on the total weight of the polishing composition, and (iii) a liquid carrier comprising water, (c) applying the chemical-mechanical polishing composition to at least a portion of the substrate, and (d) abrading at least a portion of the substrate with the polishing composition to polish the substrate.
- a chemical-mechanical polishing composition comprising: (i) an abrasive comprising ⁇ -alumina, (ii) 0.05 to 3.5 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of magnesium, zinc, and mixtures thereof, based on the total weight of the polish
- the polishing composition utilized in this method embodiment of the invention comprises: (a) an abrasive comprising ⁇ -alumina, (b) 0.05 to 3.5 millimoles per kilogram (mmol/kg) of ions of at least one metal selected from the group consisting of magnesium, zinc, and mixtures thereof, based on the total weight of the polishing composition, and (c) a liquid carrier comprising water.
- the other characteristics of the chemical-mechanical polishing composition utilized in this method embodiment of the invention e.g., the amount of abrasive, the liquid carrier, the pH, and other suitable additives
- the chemical-mechanical polishing composition of the invention can be the same as set forth above for the chemical-mechanical polishing composition of the invention.
- the method of polishing a substrate comprises the steps of: (a) providing a substrate, (b) providing a chemical-mechanical polishing composition comprising: (i) an abrasive selected from the group consisting of ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, diamond, boron carbide, silicon carbide, tungsten carbide, titanium nitride, and mixtures thereof, (ii) 0.05 to 3.5 mmol/kg of ions of at least one metal selected from the group consisting of calcium, strontium, barium, magnesium, zinc, and mixtures thereof, based on the total weight of the polishing composition, and (iii) a liquid carrier comprising water, (c) applying the chemical-mechanical polishing composition to at least a portion of the substrate, and (d) abrading at least a portion of the substrate with the polishing composition to polish the substrate.
- a chemical-mechanical polishing composition comprising: (i) an abrasive selected from the group consisting of
- the polishing composition utilized in this method embodiment of the invention comprises: (a) an abrasive selected from the group consisting of ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, ⁇ -alumina, diamond, boron carbide, silicon carbide, tungsten carbide, titanium nitride, and mixtures thereof, (b) 0.05 to 3.5 mmol/kg of ions of at least one metal selected from the group consisting of calcium, strontium, barium, magnesium, zinc, and mixtures thereof, based on the total weight of the polishing composition, and (c) a liquid carrier comprising water.
- the other characteristics of the chemical-mechanical polishing composition utilized in this method embodiment of the invention can be the same as set forth above for the chemical-mechanical polishing composition of the invention.
- the substrate to be polished using the methods of the invention can be any suitable substrate. Suitable substrates include, but are not limited to, integrated circuits, memory or rigid disks, metals, interlayer dielectric (ILD) devices, semiconductors, micro- electro-mechanical systems, ferroelectrics, and magnetic heads.
- the metal layer can comprise any suitable metal.
- the metal layer can comprise copper, tantalum (e.g., tantalum nitride), titanium, aluminum, nickel, platinum, ruthenium, iridium, or rhodium.
- the substrate can further comprise at least one insulating layer.
- the insulating layer can be a metal oxide, porous metal oxide, glass, organic polymer, fluorinated organic polymer, or any other suitable high or low- ⁇ insulating layer.
- the substrate comprises a noble metal, and at least a portion of the noble metal is abraded with the polishing composition to polish the substrate.
- Suitable noble metal include, but are not limited to, platinum, iridium, ruthenium, rhodium, palladium, silver, osmium, gold, and combinations thereof.
- the substrate comprises platinum, and at least a portion of the platinum is abraded with the polishing composition to polish the substrate.
- the polishing methods of the invention are particularly suited for use in conjunction with a chemical-mechanical polishing (CMP) apparatus.
- CMP chemical-mechanical polishing
- the apparatus comprises a platen, which, when in use, is in motion and has a velocity that results from orbital, linear, or circular motion, a polishing pad in contact with the platen and moving with the platen when in motion, and a carrier that holds a substrate to be polished by contacting and moving relative to the surface of the polishing pad.
- the polishing of the substrate takes place by the substrate being placed in contact with the polishing pad and the polishing composition of the invention and then the polishing pad moving relative to the substrate, so as to abrade at least a portion of the substrate to polish the substrate.
- the CMP apparatus further comprises an in situ polishing endpoint detection system, many of which are known in the art.
- the inspection or monitoring of the progress of the polishing process with respect to a substrate being polished enables the determination of the polishing end-point, i.e., the determination of when to terminate the polishing process with respect to a particular substrate.
- the CMP apparatus can further comprise a means for oxidizing the substrate.
- the means for oxidizing the substrate preferably comprises a device for applying a time-varying potential (e.g., anodic potential) to the substrate (e.g., electronic potentiostat).
- the device for applying time-varying potential to the substrate can be any suitable such device.
- the means for oxidizing the substrate preferably comprises a device for applying a first potential (e.g., a more oxidizing potential) during an initial stage of the polishing and applying a second potential (e.g., a less oxidizing potential) at or during a later stage of polishing, or a device for changing the first potential to the second potential during an intermediate stage of polishing, e.g., continuously reducing the potential during the intermediate stage or rapidly reducing the potential from a first, higher oxidizing potential to a second, lower oxidizing potential after a predetermined interval at the first, higher oxidizing potential.
- a first potential e.g., a more oxidizing potential
- a second potential e.g., a less oxidizing potential
- a relatively high oxidizing potential is applied to the substrate to promote a relatively high rate of oxidation/dissolution/removal of the substrate.
- the applied potential is reduced to a level producing a substantially lower or negligible rate of oxidation/dissolution/removal of the substrate, thereby eliminating or substantially reducing dishing, corrosion, and erosion.
- the time-varying electrochemical potential is preferably applied using a controllably variable DC power supply, e.g., an electronic potentiostat.
- U.S. Patent 6,379,223 further describes a means for oxidizing a substrate by applying a potential.
- polishing composition of the invention Similar substrates comprising platinum were polished using four different polishing compositions (polishing compositions 1A, IB, 1C, and ID). Polishing composition 1A (comparative) did not comprise an appreciable amount of metals ions. Polishing composition IB (invention) comprised 0.38 mmol/kg (approximately 15 ppm) of calcium (as calcium chloride).
- Polishing composition 1C (invention) comprised 0.38 mmol/kg (approximately 33 ppm) of strontium (as strontium chloride).
- Polishing composition ID (invention) comprised 0.37 mmol/kg (approximately 51 ppm) of barium (as barium chloride).
- Each of the aforementioned polishing compositions also comprised 3 wt.%> of an abrasive comprising, based on the total weight of the abrasive, approximately 60 wt.%> ⁇ -alumina and approximately 40 wt.%> fumed alumina, and had a pH of 3.
- the values for the platinum removal rate (in angstroms per minute) were measured for each of the polishing compositions. The results are summarized in Table 1. Table 1: Platinum removal rates.
- polishing composition of the invention exhibits a high platinum removal rate as compared to similar polishing compositions comprising no appreciable amount of metal ions.
- polishing compositions IBID invention
- polishing compositions IBID invention
- polishing compositions 1A comparative
- This example demonstrates the enhanced polishing rate exhibited by the polishing composition of the invention.
- Polishing composition 2A comparative
- Polishing composition 2B comparative
- Polishing composition 2C comparative
- Each of the aforementioned polishing compositions also comprised 3 wt.%> of an abrasive comprising, based on the total weight of the abrasive, approximately 60 wt.% ⁇ -alumina and approximately 40 wt.% fumed alumina, and had a pH of 3.
- the values for the platinum removal rate (in angstroms per minute) were measured for each of the polishing compositions. The results are summarized in Table 2. Table 2: Platinum removal rates.
- polishing composition of the invention exhibits a high platinum removal rate as compared to similar polishing compositions comprising no appreciable amount of metal ions.
- polishing compositions 2B and 2C which comprised approximately 0.4 and 0.74 mmol/kg of ions of magnesium, each exhibited a platinum removal rate that was approximately 30%) and 75%>, respectively, greater that the platinum removal rate for polishing composition 2A (comparative), which did not contain an appreciable amount of magnesium ions.
- EXAMPLE 3 [0047] This example demonstrates the enhanced polishing rate exhibited by the polishing composition of the invention.
- polishing compositions 3A, 3B, 3C, 3D, 3E, and 3F Similar substrates comprising platinum were polished using six different polishing compositions (polishing compositions 3A, 3B, 3C, 3D, 3E, and 3F).
- Polishing composition 3A comparative
- Polishing composition 3B comparative
- Polishing composition 3C comprisesd 3.0 mmol/kg of aluminum (as aluminum nitrate).
- Polishing composition 3D (invention) comprised 0.74 mmol/kg (approximately 18 ppm) of magnesium (as magnesium chloride).
- Polishing composition 3E (invention) comprised 0.75 mmol/kg (approximately 49 ppm) of zinc (as zinc chloride).
- Polishing composition 3F (invention) comprised 1.5 mmol/kg (approximately 96 ppm) of zinc (as zinc chloride).
- Each of the aforementioned polishing compositions also comprised 3 wt.%> of an abrasive comprising, based on the total weight of the abrasive, approximately 60 wt.%> ⁇ -alumina and approximately 40 wt.%> fumed alumina, and had a pH of 3.
- the values for the platinum removal rate (in angstroms per minute) were measured for each of the polishing compositions. The results are summarized in Table 3. Table 3: Platinum removal rates.
- polishing composition of the invention exhibits a high platinum removal rate as compared to similar polishing compositions comprising no appreciable amount of metal ions or a similar amount of different metal ions.
- polishing compositions 3D-3F invention
- polishing compositions 3A-3C comparative, which comprised approximately 0.74 to 1.5 mmol/kg of ions of a metal selected from the group consisting of magnesium and zinc, each exhibited a platinum removal rate that was approximately 80%> or more higher that the platinum removal rate for polishing compositions 3A-3C (comparative), which did not contain an appreciable amount of ions of magnesium or zinc.
- EXAMPLE 4 [0049] This example demonstrates the enhanced polishing rate exhibited by the polishing composition of the invention.
- polishing compositions 4A, 4B, 4C, 4D, 4E, 4F, and 4G Similar substrates comprising platinum (from a different lot than those used in Examples 1, 2, 3, and 5) were polished using seven different polishing compositions (polishing compositions 4A, 4B, 4C, 4D, 4E, 4F, and 4G). Polishing composition 4A (comparative) did not comprise an appreciable amount of metals ions. Polishing composition 4B (comparative) comprised 0.74 mmol/kg (approximately 29 ppm) of potassium (as potassium chloride). Polishing composition 4C (comparative) comprised 0.74 mmol/kg (approximately 29 ppm) of potassium (as potassium sulfate).
- Polishing composition 4D comprised 0.74 mmol/kg (approximately 18 ppm) of magnesium (as magnesium chloride).
- Polishing composition 4E comprised 1.5 mmol/kg (approximately 36 ppm) of magnesium (as magnesium chloride).
- Polishing composition 4F comprised 3.0 mmol/kg (approximately 72 ppm) of magnesium (as magnesium chloride).
- Polishing composition 4G (invention) comprised 5.9 mmol/kg (approximately 144 ppm) of magnesium (as magnesium chloride).
- Each of the aforementioned polishing compositions also comprised 3 wt.% of an abrasive comprising, based on the total weight of the abrasive, approximately 60 wt.%> ⁇ -alumina and approximately 40 wt.%> fumed alumina, and had a pH of 3.
- the values for the platinum removal rate (in angstroms per minute) were measured for each of the polishing compositions. The results are summarized in Table 4. Table 4: Platinum removal rates.
- polishing composition of the invention exhibits a high platinum removal rate as compared to similar polishing compositions comprising no appreciable amount of metal ions or a similar amount of different metal ions.
- polishing compositions 4D-4G which comprised approximately 0.74 to 5.9 mmol/kg of magnesium, each exhibited a platinum removal rate that was greater than or equal to 4000 A/min.
- Such a removal rate was significantly greater than that observed for polishing compositions 4A-4C (comparative), which did not contain appreciable amounts of magnesium.
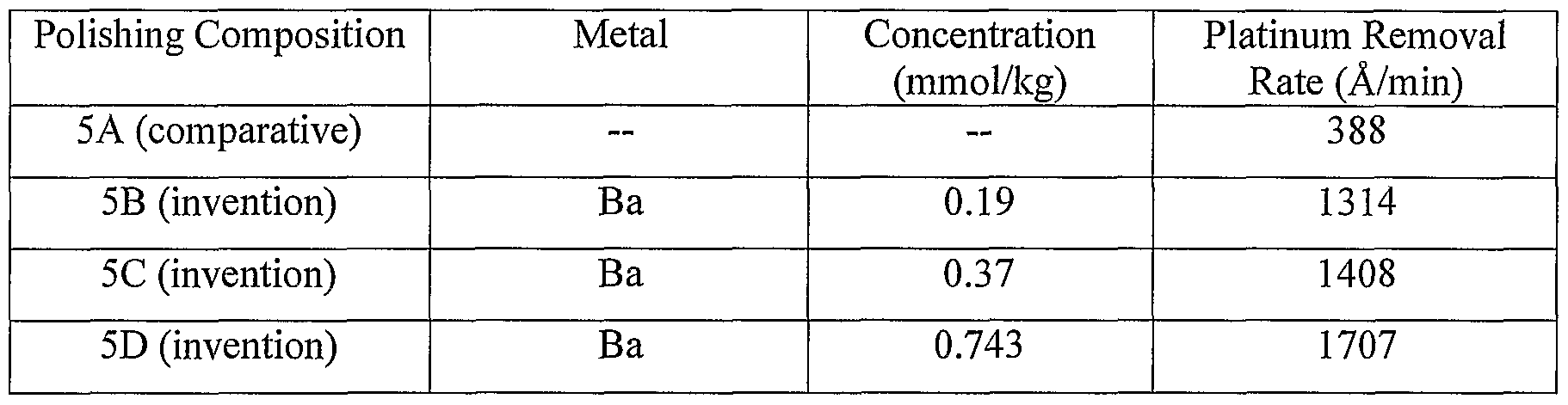
- polishing compositions 4D-4G were polished using four different polishing compositions (polishing compositions 5A, 5B, 5C, and 5D). Polishing composition 5A (comparative) did not comprise an appreciable amount of metals ions. Polishing composition 5B (invention) comprised 0.19 mmol/kg (approximately 26 ppm) of barium (as barium chloride).
- Polishing composition 5C (invention) comprised 0.37 mmol/kg (approximately 51 ppm) of barium (as barium chloride).
- Polishing composition 5D (invention) comprised 0.743 mmol/kg (approximately 102 ppm) of barium (as barium chloride).
- Each of the aforementioned polishing compositions also comprised 3 wt.% of an abrasive comprising, based on the total weight of the abrasive, approximately 60 wt.% ⁇ -alumina and approximately 40 wt.%> fumed alumina, and had a pH of 3.
- the values for the platinum removal rate (in angstroms per minute) were measured for each of the polishing compositions. The results are summarized in Table 5. Table 5: Platinum removal rates.
- polishing composition of the invention exhibits a high platinum removal rate as compared to similar polishing compositions comprising no appreciable amount of metal ions.
- polishing compositions 5B- 5D which comprised approximately 0.19 to 0.743 mmol/kg of barium ions, each exhibited a platinum removal rate that was approximately 240%> or more higher that the platinum removal rate for polishing composition 1A (comparative), which did not contain an appreciable amount of barium ions.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Life Sciences & Earth Sciences (AREA)
- Geology (AREA)
- Inorganic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Materials Engineering (AREA)
- General Physics & Mathematics (AREA)
- Manufacturing & Machinery (AREA)
- Computer Hardware Design (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Power Engineering (AREA)
- Mechanical Treatment Of Semiconductor (AREA)
- Finish Polishing, Edge Sharpening, And Grinding By Specific Grinding Devices (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2007505001A JP2007531274A (en) | 2004-03-24 | 2005-03-14 | Chemical mechanical polishing composition and method of use thereof |
| AT05725517T ATE540093T1 (en) | 2004-03-24 | 2005-03-14 | COMPOSITION FOR CHEMICAL-MECHANICAL POLISHING AND USE |
| EP05725517A EP1730246B1 (en) | 2004-03-24 | 2005-03-14 | Chemical-mechanical polishing composition and method for using the same |
| CN2005800096376A CN1938392B (en) | 2004-03-24 | 2005-03-14 | Chemical-mechanical polishing composition and method for using the same |
| IL176669A IL176669A0 (en) | 2004-03-24 | 2006-07-02 | Chemical-mechanical polishing composition and method for using the same |
| KR1020067019562A KR101082154B1 (en) | 2004-03-24 | 2006-09-22 | - chemical-mechanical polishing composition and method for using the same |
| IL219496A IL219496A (en) | 2004-03-24 | 2012-04-30 | Chemical-mechanical polishing composition and method for using the same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/807,944 | 2004-03-24 | ||
| US10/807,944 US20050211950A1 (en) | 2004-03-24 | 2004-03-24 | Chemical-mechanical polishing composition and method for using the same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005100496A2 true WO2005100496A2 (en) | 2005-10-27 |
| WO2005100496A3 WO2005100496A3 (en) | 2005-12-29 |
Family
ID=34962673
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2005/008411 WO2005100496A2 (en) | 2004-03-24 | 2005-03-14 | Chemical-mechanical polishing composition and method for using the same |
Country Status (11)
| Country | Link |
|---|---|
| US (2) | US20050211950A1 (en) |
| EP (1) | EP1730246B1 (en) |
| JP (2) | JP2007531274A (en) |
| KR (1) | KR101082154B1 (en) |
| CN (1) | CN1938392B (en) |
| AT (1) | ATE540093T1 (en) |
| IL (2) | IL176669A0 (en) |
| MY (1) | MY146598A (en) |
| SG (1) | SG150494A1 (en) |
| TW (1) | TWI299747B (en) |
| WO (1) | WO2005100496A2 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2419134B (en) * | 2004-09-09 | 2009-10-14 | Fujimi Inc | Polishing composition and polishing method using the same |
| JP2010509755A (en) * | 2006-11-02 | 2010-03-25 | キャボット マイクロエレクトロニクス コーポレイション | CMP of copper / ruthenium / tantalum substrates |
| EP2268761A2 (en) * | 2008-03-21 | 2011-01-05 | Cabot Microelectronics Corporation | Compositions for polishing aluminum/copper and titanium in damascene structures |
| CN102212334A (en) * | 2011-04-19 | 2011-10-12 | 浙江露笑光电有限公司 | Coarse grinding fluid for sapphire substrate and preparation method thereof |
| US8372305B2 (en) | 2007-05-24 | 2013-02-12 | Basf Se | Chemical-mechanical polishing composition comprising metal-organic framework materials |
| US9799532B2 (en) | 2010-02-15 | 2017-10-24 | Hitachi Chemical Company, Ltd. | CMP polishing solution and polishing method |
| US10796921B2 (en) | 2009-07-16 | 2020-10-06 | Hitachi Chemical Company, Ltd. | CMP fluid and method for polishing palladium |
| EP3894497A4 (en) * | 2018-12-10 | 2022-09-14 | CMC Materials, Inc. | Oxidizer free slurry for ruthenium cmp |
Families Citing this family (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7247567B2 (en) * | 2004-06-16 | 2007-07-24 | Cabot Microelectronics Corporation | Method of polishing a tungsten-containing substrate |
| US20060108325A1 (en) * | 2004-11-19 | 2006-05-25 | Everson William J | Polishing process for producing damage free surfaces on semi-insulating silicon carbide wafers |
| US7955519B2 (en) * | 2005-09-30 | 2011-06-07 | Cabot Microelectronics Corporation | Composition and method for planarizing surfaces |
| US7998866B2 (en) * | 2006-09-05 | 2011-08-16 | Cabot Microelectronics Corporation | Silicon carbide polishing method utilizing water-soluble oxidizers |
| JP5429169B2 (en) * | 2008-08-06 | 2014-02-26 | 日立化成株式会社 | CMP polishing liquid and substrate polishing method using this CMP polishing liquid |
| KR101492969B1 (en) * | 2008-11-14 | 2015-02-16 | 일진다이아몬드(주) | High hardness coated powder and method of manufacturing the same |
| CN102699811B (en) * | 2012-05-29 | 2015-07-29 | 上海瑞钼特金属新材料有限公司 | Refractory metal alloy paillon foil part of surface best bright finish and preparation method thereof |
| US9011207B2 (en) | 2012-10-29 | 2015-04-21 | Wayne O. Duescher | Flexible diaphragm combination floating and rigid abrading workholder |
| US8845394B2 (en) | 2012-10-29 | 2014-09-30 | Wayne O. Duescher | Bellows driven air floatation abrading workholder |
| US9199354B2 (en) | 2012-10-29 | 2015-12-01 | Wayne O. Duescher | Flexible diaphragm post-type floating and rigid abrading workholder |
| US9233452B2 (en) | 2012-10-29 | 2016-01-12 | Wayne O. Duescher | Vacuum-grooved membrane abrasive polishing wafer workholder |
| US8998677B2 (en) | 2012-10-29 | 2015-04-07 | Wayne O. Duescher | Bellows driven floatation-type abrading workholder |
| US9604339B2 (en) | 2012-10-29 | 2017-03-28 | Wayne O. Duescher | Vacuum-grooved membrane wafer polishing workholder |
| US8998678B2 (en) | 2012-10-29 | 2015-04-07 | Wayne O. Duescher | Spider arm driven flexible chamber abrading workholder |
| US9039488B2 (en) | 2012-10-29 | 2015-05-26 | Wayne O. Duescher | Pin driven flexible chamber abrading workholder |
| US8920667B2 (en) * | 2013-01-30 | 2014-12-30 | Cabot Microelectronics Corporation | Chemical-mechanical polishing composition containing zirconia and metal oxidizer |
| CN103254799A (en) * | 2013-05-29 | 2013-08-21 | 陈玉祥 | Hydrophilic diamond-suspended grinding and polishing solution and preparation method thereof |
| US9434859B2 (en) * | 2013-09-24 | 2016-09-06 | Cabot Microelectronics Corporation | Chemical-mechanical planarization of polymer films |
| WO2015057433A1 (en) * | 2013-10-18 | 2015-04-23 | Cabot Microelectronics Corporation | Polishing composition and method for nickel-phosphorous coated memory disks |
| CN104592935B (en) * | 2015-01-04 | 2016-04-27 | 江苏中晶科技有限公司 | Mechanically resistant material grinding accelerator |
| US10926378B2 (en) | 2017-07-08 | 2021-02-23 | Wayne O. Duescher | Abrasive coated disk islands using magnetic font sheet |
| US11286403B2 (en) | 2018-07-20 | 2022-03-29 | Dongjin Semichem Co., Ltd | Chemical mechanical polishing composition, chemical mechanical polishing slurry and method for polishing substrate |
| US11691241B1 (en) * | 2019-08-05 | 2023-07-04 | Keltech Engineering, Inc. | Abrasive lapping head with floating and rigid workpiece carrier |
| CN111421391A (en) * | 2020-03-09 | 2020-07-17 | 大连理工大学 | Double-sided chemical mechanical polishing method for single crystal diamond wafer |
| CN115926748B (en) * | 2022-12-21 | 2024-07-12 | 广东红日星实业有限公司 | Grinding fluid and preparation method and application thereof |
Family Cites Families (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01257563A (en) * | 1988-04-08 | 1989-10-13 | Showa Denko Kk | Composition for polishing aluminium magnetic disc |
| US4959113C1 (en) * | 1989-07-31 | 2001-03-13 | Rodel Inc | Method and composition for polishing metal surfaces |
| US5693239A (en) * | 1995-10-10 | 1997-12-02 | Rodel, Inc. | Polishing slurries comprising two abrasive components and methods for their use |
| US6110396A (en) * | 1996-11-27 | 2000-08-29 | International Business Machines Corporation | Dual-valent rare earth additives to polishing slurries |
| US20030077221A1 (en) * | 2001-10-01 | 2003-04-24 | Shivkumar Chiruvolu | Aluminum oxide powders |
| US6099604A (en) * | 1997-08-21 | 2000-08-08 | Micron Technology, Inc. | Slurry with chelating agent for chemical-mechanical polishing of a semiconductor wafer and methods related thereto |
| JPH1180708A (en) * | 1997-09-09 | 1999-03-26 | Fujimi Inkooporeetetsudo:Kk | Composition for polishing |
| US6432828B2 (en) * | 1998-03-18 | 2002-08-13 | Cabot Microelectronics Corporation | Chemical mechanical polishing slurry useful for copper substrates |
| US5972124A (en) | 1998-08-31 | 1999-10-26 | Advanced Micro Devices, Inc. | Method for cleaning a surface of a dielectric material |
| JP2000212776A (en) * | 1999-01-18 | 2000-08-02 | Jsr Corp | Aqueous dispersion for mechanochemical polishing |
| US6443812B1 (en) * | 1999-08-24 | 2002-09-03 | Rodel Holdings Inc. | Compositions for insulator and metal CMP and methods relating thereto |
| WO2001044395A1 (en) * | 1999-12-14 | 2001-06-21 | Rodel Holdings, Inc. | Polishing compositions for semiconductor substrates |
| US20030006396A1 (en) * | 1999-12-14 | 2003-01-09 | Hongyu Wang | Polishing composition for CMP having abrasive particles |
| US20020039839A1 (en) * | 1999-12-14 | 2002-04-04 | Thomas Terence M. | Polishing compositions for noble metals |
| JP2001187876A (en) * | 1999-12-28 | 2001-07-10 | Nec Corp | Slurry for chemical mechanical polishing |
| JP2001210640A (en) * | 2000-01-27 | 2001-08-03 | Inst Of Physical & Chemical Res | Method for forming protective film of semiconductor |
| JP2001308042A (en) * | 2000-04-26 | 2001-11-02 | Okamoto Machine Tool Works Ltd | Polishing agent slurry for substrate |
| US6468913B1 (en) * | 2000-07-08 | 2002-10-22 | Arch Specialty Chemicals, Inc. | Ready-to-use stable chemical-mechanical polishing slurries |
| DE10048477B4 (en) * | 2000-09-29 | 2008-07-03 | Qimonda Ag | Process for the chemical-mechanical polishing of layers of platinum group metals |
| US6649523B2 (en) * | 2000-09-29 | 2003-11-18 | Nutool, Inc. | Method and system to provide material removal and planarization employing a reactive pad |
| JP4153657B2 (en) * | 2000-11-21 | 2008-09-24 | 富士フイルム株式会社 | Magnetic recording medium |
| EP1211024A3 (en) * | 2000-11-30 | 2004-01-02 | JSR Corporation | Polishing method |
| EP1356502A1 (en) * | 2001-01-16 | 2003-10-29 | Cabot Microelectronics Corporation | Ammonium oxalate-containing polishing system and method |
| JP2002231666A (en) * | 2001-01-31 | 2002-08-16 | Fujimi Inc | Composition for polishing, and polishing method using the composition |
| US6812193B2 (en) * | 2001-08-31 | 2004-11-02 | International Business Machines Corporation | Slurry for mechanical polishing (CMP) of metals and use thereof |
| US6730592B2 (en) * | 2001-12-21 | 2004-05-04 | Micron Technology, Inc. | Methods for planarization of metal-containing surfaces using halogens and halide salts |
| US6884723B2 (en) * | 2001-12-21 | 2005-04-26 | Micron Technology, Inc. | Methods for planarization of group VIII metal-containing surfaces using complexing agents |
| US20030162398A1 (en) * | 2002-02-11 | 2003-08-28 | Small Robert J. | Catalytic composition for chemical-mechanical polishing, method of using same, and substrate treated with same |
| KR20040000009A (en) * | 2002-06-19 | 2004-01-03 | 주식회사 하이닉스반도체 | Solution for Platinum-Chemical Mechanical Planarization |
| US20040029494A1 (en) * | 2002-08-09 | 2004-02-12 | Souvik Banerjee | Post-CMP cleaning of semiconductor wafer surfaces using a combination of aqueous and CO2 based cryogenic cleaning techniques |
| US7968465B2 (en) * | 2003-08-14 | 2011-06-28 | Dupont Air Products Nanomaterials Llc | Periodic acid compositions for polishing ruthenium/low K substrates |
-
2004
- 2004-03-24 US US10/807,944 patent/US20050211950A1/en not_active Abandoned
-
2005
- 2005-03-01 TW TW094105987A patent/TWI299747B/en not_active IP Right Cessation
- 2005-03-14 JP JP2007505001A patent/JP2007531274A/en not_active Withdrawn
- 2005-03-14 CN CN2005800096376A patent/CN1938392B/en not_active Expired - Fee Related
- 2005-03-14 WO PCT/US2005/008411 patent/WO2005100496A2/en active Application Filing
- 2005-03-14 EP EP05725517A patent/EP1730246B1/en not_active Not-in-force
- 2005-03-14 AT AT05725517T patent/ATE540093T1/en active
- 2005-03-14 SG SG200900786-5A patent/SG150494A1/en unknown
- 2005-03-22 MY MYPI20051241A patent/MY146598A/en unknown
-
2006
- 2006-07-02 IL IL176669A patent/IL176669A0/en not_active IP Right Cessation
- 2006-09-22 KR KR1020067019562A patent/KR101082154B1/en active IP Right Grant
-
2009
- 2009-02-26 US US12/393,489 patent/US8101093B2/en not_active Expired - Fee Related
-
2011
- 2011-12-02 JP JP2011264253A patent/JP5781906B2/en not_active Expired - Fee Related
-
2012
- 2012-04-30 IL IL219496A patent/IL219496A/en not_active IP Right Cessation
Non-Patent Citations (1)
| Title |
|---|
| None |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2419134B (en) * | 2004-09-09 | 2009-10-14 | Fujimi Inc | Polishing composition and polishing method using the same |
| JP2010509755A (en) * | 2006-11-02 | 2010-03-25 | キャボット マイクロエレクトロニクス コーポレイション | CMP of copper / ruthenium / tantalum substrates |
| US8372305B2 (en) | 2007-05-24 | 2013-02-12 | Basf Se | Chemical-mechanical polishing composition comprising metal-organic framework materials |
| EP2268761A2 (en) * | 2008-03-21 | 2011-01-05 | Cabot Microelectronics Corporation | Compositions for polishing aluminum/copper and titanium in damascene structures |
| EP2268761A4 (en) * | 2008-03-21 | 2011-12-07 | Cabot Microelectronics Corp | Compositions for polishing aluminum/copper and titanium in damascene structures |
| US10796921B2 (en) | 2009-07-16 | 2020-10-06 | Hitachi Chemical Company, Ltd. | CMP fluid and method for polishing palladium |
| US9799532B2 (en) | 2010-02-15 | 2017-10-24 | Hitachi Chemical Company, Ltd. | CMP polishing solution and polishing method |
| CN102212334A (en) * | 2011-04-19 | 2011-10-12 | 浙江露笑光电有限公司 | Coarse grinding fluid for sapphire substrate and preparation method thereof |
| EP3894497A4 (en) * | 2018-12-10 | 2022-09-14 | CMC Materials, Inc. | Oxidizer free slurry for ruthenium cmp |
Also Published As
| Publication number | Publication date |
|---|---|
| MY146598A (en) | 2012-08-30 |
| IL219496A (en) | 2013-04-30 |
| TWI299747B (en) | 2008-08-11 |
| EP1730246B1 (en) | 2012-01-04 |
| SG150494A1 (en) | 2009-03-30 |
| CN1938392A (en) | 2007-03-28 |
| EP1730246A2 (en) | 2006-12-13 |
| JP2012049570A (en) | 2012-03-08 |
| US20050211950A1 (en) | 2005-09-29 |
| JP2007531274A (en) | 2007-11-01 |
| IL176669A0 (en) | 2006-10-31 |
| WO2005100496A3 (en) | 2005-12-29 |
| IL219496A0 (en) | 2012-06-28 |
| US20090152240A1 (en) | 2009-06-18 |
| KR20060134996A (en) | 2006-12-28 |
| TW200540240A (en) | 2005-12-16 |
| US8101093B2 (en) | 2012-01-24 |
| CN1938392B (en) | 2010-09-01 |
| KR101082154B1 (en) | 2011-11-09 |
| JP5781906B2 (en) | 2015-09-24 |
| ATE540093T1 (en) | 2012-01-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1730246B1 (en) | Chemical-mechanical polishing composition and method for using the same | |
| JP5449248B2 (en) | Chemical mechanical polishing composition | |
| EP3049216B1 (en) | Chemical-mechanical planarization of polymer films | |
| US8591763B2 (en) | Halide anions for metal removal rate control | |
| EP1812523B1 (en) | Metal ion-containing cmp composition and method for using the same | |
| TWI753987B (en) | Chemical mechanical polishing method for tungsten | |
| KR20080059397A (en) | Polishing fluids and methods for cmp | |
| US20050126588A1 (en) | Chemical mechanical polishing slurries and cleaners containing salicylic acid as a corrosion inhibitor | |
| EP1356502A1 (en) | Ammonium oxalate-containing polishing system and method | |
| JP2008034818A (en) | Polishing solution for polishing noble metal films and polishing method of noble metal films |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 176669 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020067019562 Country of ref document: KR Ref document number: 2007505001 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580009637.6 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Country of ref document: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005725517 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005725517 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020067019562 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 219496 Country of ref document: IL |