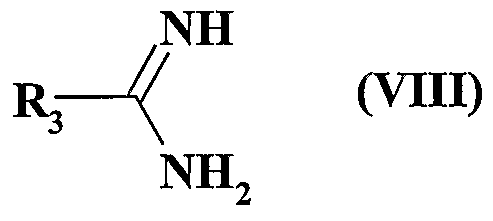WO2003053979A1 - Nouveaux derives de benzothiazine et benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent - Google Patents
Nouveaux derives de benzothiazine et benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent Download PDFInfo
- Publication number
- WO2003053979A1 WO2003053979A1 PCT/FR2002/004485 FR0204485W WO03053979A1 WO 2003053979 A1 WO2003053979 A1 WO 2003053979A1 FR 0204485 W FR0204485 W FR 0204485W WO 03053979 A1 WO03053979 A1 WO 03053979A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- group
- linear
- branched
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/02—1,2-Thiazines; Hydrogenated 1,2-thiazines
Definitions
- the present invention relates to new benzothiazine and benzothiadiazine derivatives, their preparation process and the pharmaceutical compositions containing them.
- the AMPA receptor (“ ⁇ -amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid”) appears to be the most involved in the phenomena of physiological neuronal excitability and in particular in those involved in the memorization process. For example, learning has been shown to be associated with increased binding of AMPA to its receptor in the hippocampus, one of the brain areas essential for mnemocognitive processes. Likewise, nootropic agents such as aniracetam have been described very recently as positively modulating the AMPA receptors of neuronal cells (Journal of Neurochemistry, 1992, 58, 1199-1204).
- patent EP 692 484 describes a benzothiadiazine derivative having a facilitating activity on the AMPA current and patent application WO 99/42456 describes inter alia certain benzothiadiazine derivatives as modulators of AMPA receptors.
- benzothiazine and benzothiadiazine derivatives which are the subject of the present invention, in addition to the fact that they are new, have, surprisingly, pharmacological activities on the AMPA current clearly superior to those of compounds of similar structures described in the Art.
- Prior. They are useful as AMPA modulators for the treatment or prevention of mnemocognitive disorders associated with age, anxiety or depressive syndromes, progressive neurogenerative diseases, Alzheimer's disease, Pick's disease, chorea. Huntington's, schizophrenia, sequelae of acute neurodegenerative diseases, sequelae of ischemia and sequelae of epilepsy.
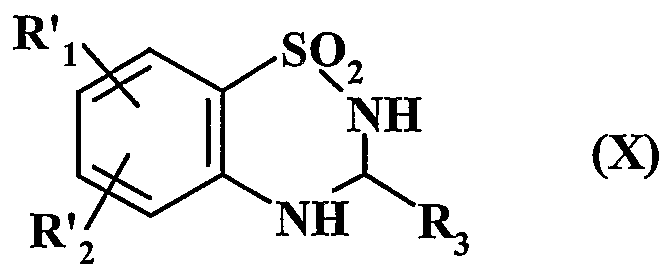
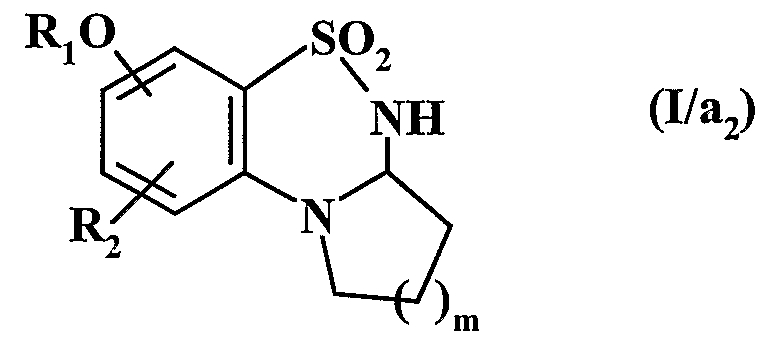
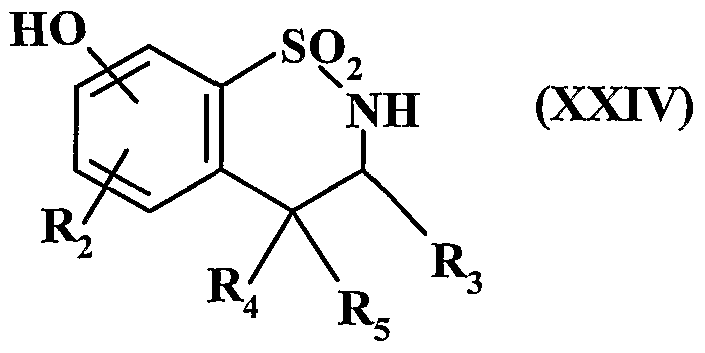
- Ri represents an aryl or heteroaryl group
- R 2 represents a hydrogen atom, a halogen atom or a hydroxy group
- A represents a CR R 5 group or an NI i group
- R 3 represents a hydrogen atom, a linear (C ⁇ -C 6 ) alkyl group or branched or a cycloalkyl group (C 3 -C)
- R represents a hydrogen atom or an alkyl group (C ⁇ -C 6 ) linear or branched
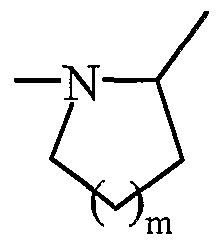
- A represents a nitrogen atom and forms with the group -CHR - adjacent the cycle in which m represents 1, 2 or 3
- Rs represents a hydrogen or halogen atom
- aryl group is understood a monocyclic aromatic group or a bicyclic group in which at least one of the rings is aromatic, optionally substituted by a or several groups, identical or different, halogen, linear or branched (C ⁇ -C 6 ) alkyl (optionally substituted by one or more hydroxy groups), linear or branched alkoxy (C ⁇ -C 6 ), perhaloalkyl (C ⁇ -C 6 ) or branched, linear or branched alkoxy carbonyl (C ⁇ -C 6 ), linear or branched alkylthio (Ci- C 6 ), carboxy, acyl (C ⁇ -C 6 ) linear or branched, linear or branched perhaloalkoxy (C ⁇ -C 6 ), hydroxy, cyano, nitro, amino (optionally substituted by one or more alkyl (C ⁇ -C6) linear or branched acyl or (C
- hydrochloric hydrobromic, sulfuric and phosphonic acids.
- acetic trifluoroacetic, lactic, pyruvic, malonic, succinic, glutaric, fumaric, tartaric, maleic, citric, ascorbic, methane sulphonic, camphoric, etc ...
- the RiO- group is preferably in position b of the phenyl which carries it.
- the preferred group Ri is an optionally substituted aryl group, especially phenyl group.
- the substituent is preferably meta.
- the preferred group R 2 is the hydrogen atom.
- the preferred compounds of the invention are the compounds such that A represents an atom
- the invention also extends to the process for the preparation of the compounds of formula (I).
- R'i represents a linear or branched alkoxy group (C ⁇ -C 6 ),
- R ' 2 represents a hydrogen, halogen atom or a linear or branched alkoxy group (C ⁇ -C 6 ), which is:
- R is such a defined in formula (I),
- R'i represents a linear or branched alkoxy group (C ⁇ -C 6 ),
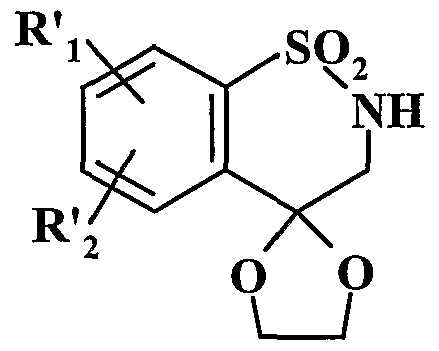
- R ' 2 represents a hydrogen, halogen atom or a linear or branched alkoxy group (C ⁇ -C 6 ), which undergoes the action of chloroacetone in the presence of dimethylformamide, to lead to the compound of formula (XVI):
- R'i and R ' 2 have the same meaning as above, which is deacetylated by heating at reflux in a benzene medium in the presence of an excess of ethylene glycol and of a catalytic amount of acid /> - toluene sulfonic acid, to lead to the compound of formula (XVIII):
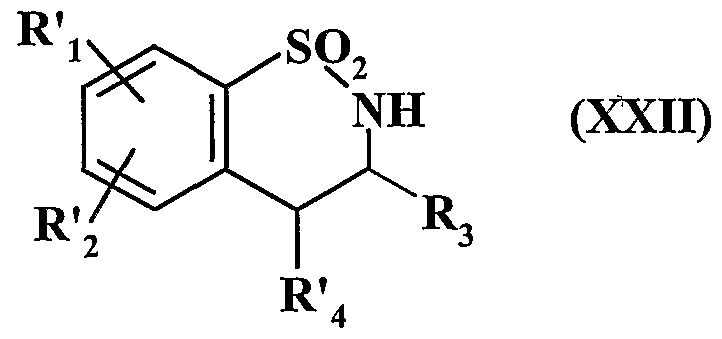
- R'i and R ' 2 have the same meaning as above, including, optionally, depending on the nature of the group R 3 which it is desired to obtain, the nitrogen atom is protected by a protective group, then, after treatment with a strong base, which is treated with a compound of formula R ' 3 -P, in which R' 3 represents a linear or branched alkyl group (C ⁇ -C 6 ) or a cycloalkyl group (C -C) and P represents a leaving group, to lead after deprotection of the nitrogen atom, to the compound of formula (XIX '):
- R'i, R ' 2 and R' 3 have the same meaning as above, composed of formula (XIXa) and (XlX'a) represented by the formula (XIX):
- R'i and R ' 2 have the same meaning and R 3 is as defined in formula (I), which:
- R'i and R ' 2 have the same meaning as above, R' 5 represents a halogen atom,
- R ' 4 MgBr represents a linear or branched (C ⁇ -C 6 ) alkyl group
- R'i, R ' 2 and R' 4 have the same meaning as above and R ' 5 represents a halogen atom
- the invention also extends to pharmaceutical compositions containing as active ingredient a compound of formula (I) with one or more inert, non-toxic and suitable excipients.
- pharmaceutical compositions according to the invention mention may be made more particularly of those which are suitable for oral, parenteral (intravenous or subcutaneous), nasal administration, simple or coated tablets, sublingual tablets, capsules, tablets, suppositories, creams, ointments, dermal gels, injections, oral suspensions, etc.
- the useful dosage is adaptable according to the nature and severity of the disease, the route of administration as well as the age and weight of the patient. This dosage varies from 1 to 500 mg per day in one or more doses.
- the starting materials used are known products or prepared according to known procedures.
- Stage D 5,5-Dioxido-2,3,3a, 4-téirahvdro ⁇ l ⁇ - rrolof2, l-cJFl 2,4J benzothiadiazin- 7-al
- the compounds of Examples 2 and 3 are obtained by separation of isomers of Example 1, on a chiral column, Chiralcel OC ® using as elution solvent a mixture of isopropanol / diethylamine 1000 / 0.5. After separation, each isomer is purified by chromatography on a silica column using a dichloromethane / ethyl acetate mixture (20/10) as eluent.
- Example 14a and 14b were obtained by separation of the enantiomers of Example 14 on a chiral column, Chiralcel OC ® under the same conditions as described for Examples 2 and 3.
- the expected product is obtained from the compound described in Example 9. To this, after dissolution in methylene chloride, cooling in an ice bath, is added a solution of boron tribromide (IM) in CH 2 C1 2 . After returning to ambient temperature, stirring overnight, the medium is cooled in an ice bath. After addition of water, extraction with CH 2 C1 2 , drying and evaporation, the expected product is obtained after filtration of the residue and taken up in ether and filtration.
- IM boron tribromide
- EXAMPLE 25 ⁇ 3 - [(5 5 5-Dioxido-2,3,3a, 4-têtrahydro-1H-pyrrolo [2, lc] [1,2,4] benzothîadiazin-7-yl) oxy] phenyl ⁇ ethanone
- a suspension of 3.0 g of 2-amino-5-methoxy-benzenesulfonamide is stirred for 1 night at 80 ° C. in the presence of 1.31 g of formamidine hydrochloride and 2.27 ml of triethylamine in 50 ml of toluene.
- the toluene is evaporated under vacuum. The residue is taken up in water and the precipitate is filtered.
- the expected product is obtained from the compound described in the preceding stage according to the process described in stage E of Example 1.
- a solution of sodium ethanolate in ethanol is prepared by dissolving 1.08 g of sodium in 23 ml of ethanol at reflux. The temperature of the solution is brought back to 40 ° C. and 6.30 g of the product from stage A is added thereto, with stirring. The reaction medium solidifies. 5 ml of ethanol are added to allow stirring to take place and the mixture is heated
- Stage C 7-Methoxy ⁇ 2,3-dihvdro-4H-1,2,2-benzothiazine-4,4 - ethylenedioxy 1,1-dioxide
- the mixture is stirred at reflux in a flask surmounted by a Dean-Stark 5.35 g of the product obtained in the preceding stage, 200 mg of paratoluene sulfonic acid, 5.6 ml of ethylene glycol in 200 ml of benzene.
- the benzene is evaporated under vacuum.
- the residue is dissolved in ethyl acetate and the organic phase is washed with water and then saturated NaCl. It is dried, filtered, evaporated and an oil is obtained which is crystallized from a mixture of ethyl ether / isopropyl ether.
- the expected product is obtained from the compound described in the preceding stage according to the process described in stage E of Example 1.
- EXAMPLE 34 7- (3-MethvlsuIfanvlphenoxv) -2, 3a, 4-tetrahvdro-1H-Dvrrolor2 -cl [1,2,4] benzothiadiazine-5,5-dioxide
- Example 38 7- (3-Bromophenoxy) -2,3,3a, 4-tetrahydro-1JÎ-pyrrolo [2, l-c] [1,2,4] benzothiadiazine-5,5-doxide
- This compound is obtained according to the protocol of Example 39 by replacing the ammonia with dimethylamine in the presence of pyridine.
- This compound is obtained according to the protocol of Example 39 by replacing rammoniac with methylamine in the presence of pyridine.
- Example 15 The product of Example 15 (2.19 mmol) is dissolved in a mixture of 100 ml of ethyl acetate and 50 ml of ethanol, 100 mg of 10% palladium on charcoal are added and hydrogenation is carried out under pressure. atmospheric during lh. The catalyst is filtered, the filtrate evaporated to dryness and the residue is concretized in ether to give, after filtration, the expected product.
- EXAMPLE 43 N- ⁇ 3 - [(5,5-Dioxido-2,3,3a, 4-tetrahydro-l-ff-pyrrolo [2, lc] [1,2,4] benzothiadiazin-7-yl) oxy ] phenyl ⁇ -N- (methylsulfonyl) methane-sulfonamide
- a spatula tip of DMAP is added, then dropwise 0.90 mmol of methanesulfonic anhydride diluted in 8 ml of CH 2 C1 2 .
- the reaction is washed (IN HCl, saturated NaCl), dried (MgSO 4 ).
- the 2 products formed are purified by chromatography on silica, eluting with a CH 2 C1 2 100% gradient -> CH 2 Cl 2 / MeOH 95/5.
- the first product chosen corresponds to the title di-methylsulfonylated product.
- the second, mono-methylsulfonylated corresponds to the product described in the following example.
- the title product corresponds to the second product isolated by chromatography under the conditions mentioned.
- This compound is obtained by reaction of the product from stage A of Example 49 with the corresponding amine.
- This compound is obtained by reaction of the product from stage A of Example 49 with the corresponding amine.
- EXAMPLE 54 Acid 3 - [(5,5-dioxido-2,3,3a, 4-tetrahydro-1H-pyrrolo [2, lc] [1,2,4] benzothiadiazin-7-yl) oxy] phenylphosphonic Agitation is carried out for 1 h at reflux of acetomtrile (20 ml), 0.66 mmol of the product of Example 52 and 1.98 mmol of bromotrimethylsilane. The solvent and the excess of reagent are evaporated under vacuum and the residue is taken up in solution in 10 ml of methanol. After 30 min of stirring, the solution is evaporated to dryness and the residue is taken up in IN HCl. A gum is obtained which is managed to crystallize by adding a little CH 2 C1 2 . By filtration, the expected product is recovered.
- EXAMPLE 56 4-Ethyl-7 - [(3-methylsulfanyl) phenoxy] -3,4-dihydro-2H- [1,2,4] benzothiadiazine-1,1-dioxide Melting point: 127-130 ° C Elemental microanalysis:
- This compound is obtained by reduction of the nitro product of Example 15 according to the process described in Example 42.
- This compound is obtained by amidification of the product of Example 65 according to the method of Example 39.
- This compound is obtained by amidification of the product of Example 65 according to the method of Example 40.
- EXAMPLE 68 3-Phenoxy-6,6a, 7,8,9,10-hexahydropyrido [2,1-c] [1,2,4] benzothiadiazine 5,5-dioxide This compound is obtained according to the process described in Example 1 using in stage A the chloride of 5-chloropropanoic acid.
- MRNAs are prepared from the cerebral cortex of a male Wistar rat by the guanidium thiocyanate / phenol / chloroform method.
- Poly (A + ) mRNAs are isolated by chromatography on oligo-dT cellulose and injected at a rate of 50 ng per oocyte. The oocytes are left for 2 to 3 days in incubation at 18 ° C to allow expression of the receptors and then stored at 8-10 ° C.
- the electrophysiological recording is carried out in a plexiglass® chamber at 20-
- the compounds of the invention very strongly potentiate the excitatory effects of AMPA and their activity is very clearly superior to that of the reference compounds.
- the compound of Example 3 has in particular an EC2X equal to 0.8 ⁇ M and an EC5X equal to 3.6 ⁇ M.
- the compound of Example 29 has an EC2X equal to 1.4 ⁇ M and an EC5X equal to 4.5 ⁇ M.
Landscapes
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Biomedical Technology (AREA)
- Psychiatry (AREA)
- Pain & Pain Management (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Vascular Medicine (AREA)
- Urology & Nephrology (AREA)
- Psychology (AREA)
- Hospice & Palliative Care (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen- Or Sulfur-Containing Heterocyclic Ring Compounds With Rings Of Six Or More Members (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
Abstract
Description
Claims
Priority Applications (14)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR0214973-7A BR0214973A (pt) | 2001-12-21 | 2002-12-20 | Derivados de benzotiazina e benzotiadiazina, seu processo de preparação e as composições farmacêuticas que os contêm |
| KR1020047009302A KR100586685B1 (ko) | 2001-12-21 | 2002-12-20 | 벤조티아진 및 벤조티아디아진 유도체, 이들의 제조 방법및 이들을 함유하는 약제 조성물 |
| CA002471263A CA2471263A1 (fr) | 2001-12-21 | 2002-12-20 | Nouveaux derives de benzothiazine et benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| UA20040706011A UA77737C2 (en) | 2001-12-21 | 2002-12-20 | Benzothiazine and benzothiadizine derivatives, a process for preparation thereof and pharmaceutical composition containing them |
| MXPA04005837A MXPA04005837A (es) | 2001-12-21 | 2002-12-20 | Nuevos derivados de benzotiazina y benzotiadiazina, metodo para su preparacion y composiciones farmaceuticas que los contienen. |
| AU2002364674A AU2002364674B2 (en) | 2001-12-21 | 2002-12-20 | Novel benzothiazine and benzothiadiazine derivatives, method for preparing same and pharmaceutical compositions containing same |
| US10/498,948 US7253161B2 (en) | 2001-12-21 | 2002-12-20 | Benzothiazine and benzothiadiazine derivatives method for preparing same and pharmaceutical compositions containing same |
| EP02805408A EP1480986A1 (fr) | 2001-12-21 | 2002-12-20 | Nouveaux derives de benzothiazine et benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| HU0402585A HUP0402585A3 (en) | 2001-12-21 | 2002-12-20 | Novel benzothiazine and benzothiadiazine derivatives, method for preparing same, their use and pharmaceutical compositions containing same |
| JP2003554695A JP2005518386A (ja) | 2001-12-21 | 2002-12-20 | 新規なベンゾチアジンおよびベンゾチアジアジン化合物、それらの製造方法、ならびにそれらを含む医薬組成物 |
| NZ533328A NZ533328A (en) | 2001-12-21 | 2002-12-20 | Novel benzothiazine and benzothiadiazine derivatives, method for preparing same and pharmaceutical compositions containing same |
| EA200400758A EA007227B1 (ru) | 2001-12-21 | 2002-12-20 | Соединения бензотиазина и бензотиадиазина, способ их получения и фармацевтические композиции, которые их содержат |
| NO20042978A NO20042978L (no) | 2001-12-21 | 2004-07-14 | Nye benzotiazin- og benzotiadiazinforbindelser, fremgangsmate for deres fremstilling og farmasoytiske sammensetninger inneholdende dem |
| HK05104494A HK1071749A1 (en) | 2001-12-21 | 2005-05-30 | Novel benzothiazine and benzothiadiazine derivatives, method for preparing same and pharmaceutical compositions containing same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR01/16621 | 2001-12-21 | ||
| FR0116621A FR2833956B1 (fr) | 2001-12-21 | 2001-12-21 | Nouveaux derives de benzothiazine et de benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2003053979A1 true WO2003053979A1 (fr) | 2003-07-03 |
Family
ID=8870809
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2002/004485 WO2003053979A1 (fr) | 2001-12-21 | 2002-12-20 | Nouveaux derives de benzothiazine et benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
Country Status (22)
| Country | Link |
|---|---|
| US (1) | US7253161B2 (fr) |
| EP (1) | EP1480986A1 (fr) |
| JP (1) | JP2005518386A (fr) |
| KR (1) | KR100586685B1 (fr) |
| CN (1) | CN1289509C (fr) |
| AR (1) | AR038028A1 (fr) |
| AU (1) | AU2002364674B2 (fr) |
| BR (1) | BR0214973A (fr) |
| CA (1) | CA2471263A1 (fr) |
| EA (1) | EA007227B1 (fr) |
| FR (1) | FR2833956B1 (fr) |
| GE (1) | GEP20063759B (fr) |
| HK (1) | HK1071749A1 (fr) |
| HU (1) | HUP0402585A3 (fr) |
| MA (1) | MA27090A1 (fr) |
| MX (1) | MXPA04005837A (fr) |
| NO (1) | NO20042978L (fr) |
| NZ (1) | NZ533328A (fr) |
| PL (1) | PL370159A1 (fr) |
| UA (1) | UA77737C2 (fr) |
| WO (1) | WO2003053979A1 (fr) |
| ZA (1) | ZA200404294B (fr) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2877342A1 (fr) * | 2004-11-03 | 2006-05-05 | Servier Lab | Nouveaux derives de benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| FR2877338A1 (fr) * | 2004-11-03 | 2006-05-05 | Servier Lab | Nouveaux derives de benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| EP1669072A1 (fr) | 2004-12-10 | 2006-06-14 | Les Laboratoires Servier | Dérivés de benzothiazine et benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent |
| SG139548A1 (en) * | 2003-06-13 | 2008-02-29 | Servier Lab | New benzothiazine and benzothiadiazine compounds, a process for their preparation and pharmaceutical compositions containing them |
| SG148023A1 (en) * | 2003-06-13 | 2008-12-31 | Servier Lab | New benzothiazine and benzothiadiazine compounds, a process for their preparation and pharmaceutical compositions containing them |
| WO2011083265A1 (fr) * | 2010-01-08 | 2011-07-14 | Les Laboratoires Servier | Nouveaux derives de benzothiadiazines cyclopropylees, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| EP2813508A4 (fr) * | 2012-02-08 | 2015-10-21 | Takeda Pharmaceutical | Composé hétérocyclique et application associée |
| US11939327B2 (en) | 2017-10-06 | 2024-03-26 | Takeda Pharmaceutical Company Limited | Heterocyclic compounds |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107641197B (zh) * | 2017-10-30 | 2019-11-22 | 河南工程学院 | 一种以二氧化碳与环氧环己烷为单体的共聚反应催化剂 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0692484A1 (fr) * | 1994-07-12 | 1996-01-17 | Adir Et Compagnie | Nouveau dérivé de benzothiadiazine, son procédé de préparation et les compositions pharmaceutiques qui le contiennent |
| WO1999042456A2 (fr) * | 1998-02-18 | 1999-08-26 | Neurosearch A/S | Nouveaux composes et leur utilisation comme modulateurs positifs du recepteur ampa |
| EP1176148A1 (fr) * | 2000-07-28 | 2002-01-30 | Les Laboratoires Servier | Nouveaux dérivés de benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent |
-
2001
- 2001-12-21 FR FR0116621A patent/FR2833956B1/fr not_active Expired - Fee Related
-
2002
- 2002-12-18 AR ARP020104932A patent/AR038028A1/es unknown
- 2002-12-20 JP JP2003554695A patent/JP2005518386A/ja active Pending
- 2002-12-20 HU HU0402585A patent/HUP0402585A3/hu unknown
- 2002-12-20 WO PCT/FR2002/004485 patent/WO2003053979A1/fr active Application Filing
- 2002-12-20 NZ NZ533328A patent/NZ533328A/en unknown
- 2002-12-20 GE GE5645A patent/GEP20063759B/en unknown
- 2002-12-20 CA CA002471263A patent/CA2471263A1/fr not_active Abandoned
- 2002-12-20 KR KR1020047009302A patent/KR100586685B1/ko not_active IP Right Cessation
- 2002-12-20 PL PL02370159A patent/PL370159A1/xx not_active Application Discontinuation
- 2002-12-20 US US10/498,948 patent/US7253161B2/en not_active Expired - Fee Related
- 2002-12-20 MX MXPA04005837A patent/MXPA04005837A/es active IP Right Grant
- 2002-12-20 CN CNB028258126A patent/CN1289509C/zh not_active Expired - Fee Related
- 2002-12-20 BR BR0214973-7A patent/BR0214973A/pt not_active IP Right Cessation
- 2002-12-20 EA EA200400758A patent/EA007227B1/ru not_active IP Right Cessation
- 2002-12-20 AU AU2002364674A patent/AU2002364674B2/en not_active Ceased
- 2002-12-20 UA UA20040706011A patent/UA77737C2/uk unknown
- 2002-12-20 EP EP02805408A patent/EP1480986A1/fr not_active Withdrawn
-
2004
- 2004-06-01 ZA ZA200404294A patent/ZA200404294B/en unknown
- 2004-06-04 MA MA27719A patent/MA27090A1/fr unknown
- 2004-07-14 NO NO20042978A patent/NO20042978L/no not_active Application Discontinuation
-
2005
- 2005-05-30 HK HK05104494A patent/HK1071749A1/xx not_active IP Right Cessation
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0692484A1 (fr) * | 1994-07-12 | 1996-01-17 | Adir Et Compagnie | Nouveau dérivé de benzothiadiazine, son procédé de préparation et les compositions pharmaceutiques qui le contiennent |
| WO1999042456A2 (fr) * | 1998-02-18 | 1999-08-26 | Neurosearch A/S | Nouveaux composes et leur utilisation comme modulateurs positifs du recepteur ampa |
| EP1176148A1 (fr) * | 2000-07-28 | 2002-01-30 | Les Laboratoires Servier | Nouveaux dérivés de benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SG148023A1 (en) * | 2003-06-13 | 2008-12-31 | Servier Lab | New benzothiazine and benzothiadiazine compounds, a process for their preparation and pharmaceutical compositions containing them |
| SG139548A1 (en) * | 2003-06-13 | 2008-02-29 | Servier Lab | New benzothiazine and benzothiadiazine compounds, a process for their preparation and pharmaceutical compositions containing them |
| EP1655030A1 (fr) * | 2004-11-03 | 2006-05-10 | Les Laboratoires Servier | Derives de benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| EP1655299A1 (fr) * | 2004-11-03 | 2006-05-10 | Les Laboratoires Servier | Dérivés de benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent |
| KR100818431B1 (ko) * | 2004-11-03 | 2008-04-01 | 르 라보레또레 쎄르비에르 | 벤조티아디아진 화합물, 이를 제조하는 방법 및 이를함유하는 약제 조성물 |
| FR2877342A1 (fr) * | 2004-11-03 | 2006-05-05 | Servier Lab | Nouveaux derives de benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| FR2877338A1 (fr) * | 2004-11-03 | 2006-05-05 | Servier Lab | Nouveaux derives de benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| CN100396673C (zh) * | 2004-11-03 | 2008-06-25 | 瑟维尔实验室 | 苯并噻二嗪化合物及其制备方法和含有它们的药物组合物 |
| EP1669072A1 (fr) | 2004-12-10 | 2006-06-14 | Les Laboratoires Servier | Dérivés de benzothiazine et benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent |
| FR2879201A1 (fr) * | 2004-12-10 | 2006-06-16 | Servier Lab | Nouveaux derives de benzothiazine et benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| WO2011083265A1 (fr) * | 2010-01-08 | 2011-07-14 | Les Laboratoires Servier | Nouveaux derives de benzothiadiazines cyclopropylees, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| FR2955106A1 (fr) * | 2010-01-08 | 2011-07-15 | Servier Lab | Nouveaux derives de benzothiadiazines cyclopropylees, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
| EP2813508A4 (fr) * | 2012-02-08 | 2015-10-21 | Takeda Pharmaceutical | Composé hétérocyclique et application associée |
| US9493484B2 (en) | 2012-02-08 | 2016-11-15 | Takeda Pharmaceutical Company Limited | Heterocyclic compound and use thereof |
| US11939327B2 (en) | 2017-10-06 | 2024-03-26 | Takeda Pharmaceutical Company Limited | Heterocyclic compounds |
Also Published As
| Publication number | Publication date |
|---|---|
| UA77737C2 (en) | 2007-01-15 |
| HUP0402585A2 (hu) | 2005-03-29 |
| AR038028A1 (es) | 2004-12-22 |
| NO20042978L (no) | 2004-07-14 |
| BR0214973A (pt) | 2004-12-14 |
| US20050124606A1 (en) | 2005-06-09 |
| HUP0402585A3 (en) | 2009-01-28 |
| CN1606560A (zh) | 2005-04-13 |
| HK1071749A1 (en) | 2005-07-29 |
| MA27090A1 (fr) | 2004-12-20 |
| KR20040073480A (ko) | 2004-08-19 |
| CN1289509C (zh) | 2006-12-13 |
| CA2471263A1 (fr) | 2003-07-03 |
| JP2005518386A (ja) | 2005-06-23 |
| NZ533328A (en) | 2006-10-27 |
| US7253161B2 (en) | 2007-08-07 |
| FR2833956B1 (fr) | 2004-01-30 |
| EA200400758A1 (ru) | 2005-06-30 |
| GEP20063759B (en) | 2006-02-27 |
| FR2833956A1 (fr) | 2003-06-27 |
| KR100586685B1 (ko) | 2006-06-08 |
| ZA200404294B (en) | 2005-06-01 |
| EA007227B1 (ru) | 2006-08-25 |
| MXPA04005837A (es) | 2004-09-10 |
| AU2002364674A1 (en) | 2003-07-09 |
| EP1480986A1 (fr) | 2004-12-01 |
| PL370159A1 (en) | 2005-05-16 |
| AU2002364674B2 (en) | 2007-11-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1655030B1 (fr) | Dérivés de benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent | |
| CA2153549C (fr) | Nouveau derive de benzothiadiazine, son procede de preparation et les compositions pharmeceutiques qui le contiennent | |
| WO2003053979A1 (fr) | Nouveaux derives de benzothiazine et benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent | |
| EP1456189B1 (fr) | Derives de benzothia(dia)zine et leur utilisation comme modulateurs ampa | |
| EP1176148B1 (fr) | Nouveaux dérivés de benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent | |
| EP1486503B1 (fr) | Dérivés de benzothiazine et benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent | |
| CA2492161C (fr) | Nouveaux derives de benzothiadiazines fluorees, leur procede de preparation et les compositions pharmaceutiques qui les contiennent | |
| EP1620441A1 (fr) | Derives de thiadiazine et leur utilisation comme modulateurs positifs des recepteurs ampa | |
| FR2856064A1 (fr) | Nouveaux derives de benzothiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent. | |
| WO2003053978A1 (fr) | Nouveaux derives de benzothiazine et benzothiadiazine, leur procede de preparation et les compositions pharmaceutiques qui les contiennent | |
| EP1655299A1 (fr) | Dérivés de benzothiadiazine, leur procédé de préparation et les compositions pharmaceutiques qui les contiennent | |
| FR2696457A1 (fr) | Dérivés de 3,4-dihydro-2H-1,2,4-benzothiadiazine 1,1-dioxyde, leur préparation et les médicaments les contenant. | |
| FR2955106A1 (fr) | Nouveaux derives de benzothiadiazines cyclopropylees, leur procede de preparation et les compositions pharmaceutiques qui les contiennent | |
| WO2011083264A1 (fr) | Derives thiochromanes, leur procede de preparation et les compositions pharmaceutiques qui les contiennent |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LU MC NL PT SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2004/04294 Country of ref document: ZA Ref document number: 200404294 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 533328 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002805408 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002364674 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2004/005837 Country of ref document: MX Ref document number: 2003554695 Country of ref document: JP Ref document number: 10498948 Country of ref document: US Ref document number: 1020047009302 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2471263 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20028258126 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200400758 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 8322 Country of ref document: GE Ref document number: 5645 Country of ref document: GE |
|
| WWP | Wipo information: published in national office |
Ref document number: 2002805408 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2002364674 Country of ref document: AU Date of ref document: 20021220 Kind code of ref document: B |