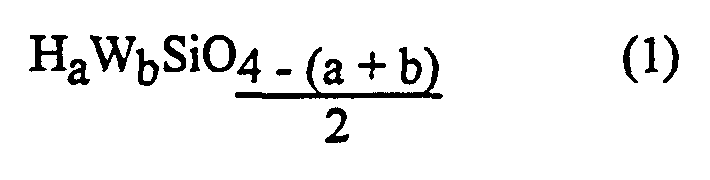WO1995025734A1 - Complexes du platine et catalyseurs d'hydrosilylation photoactivables les contenant - Google Patents
Complexes du platine et catalyseurs d'hydrosilylation photoactivables les contenant Download PDFInfo
- Publication number
- WO1995025734A1 WO1995025734A1 PCT/FR1995/000328 FR9500328W WO9525734A1 WO 1995025734 A1 WO1995025734 A1 WO 1995025734A1 FR 9500328 W FR9500328 W FR 9500328W WO 9525734 A1 WO9525734 A1 WO 9525734A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compounds
- alkyl
- formula
- complexes
- different
- Prior art date
Links
- 239000003054 catalyst Substances 0.000 title claims abstract description 40
- 238000006459 hydrosilylation reaction Methods 0.000 title claims abstract description 31
- 150000003057 platinum Chemical class 0.000 title claims abstract description 14
- 239000000203 mixture Substances 0.000 claims abstract description 45
- 239000003446 ligand Substances 0.000 claims abstract description 30
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 10
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 58
- 230000003197 catalytic effect Effects 0.000 claims description 45
- 150000001875 compounds Chemical class 0.000 claims description 34
- -1 arylalkyl radicals Chemical group 0.000 claims description 33
- 238000000034 method Methods 0.000 claims description 32
- 230000008569 process Effects 0.000 claims description 23
- 150000003254 radicals Chemical class 0.000 claims description 23
- 125000000217 alkyl group Chemical group 0.000 claims description 22
- 230000002186 photoactivation Effects 0.000 claims description 18
- 229910052697 platinum Inorganic materials 0.000 claims description 18
- 125000003342 alkenyl group Chemical group 0.000 claims description 13
- 125000003118 aryl group Chemical group 0.000 claims description 13
- 125000004432 carbon atom Chemical group C* 0.000 claims description 10
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 9
- 229910052760 oxygen Inorganic materials 0.000 claims description 9
- 230000015572 biosynthetic process Effects 0.000 claims description 8
- 125000004122 cyclic group Chemical group 0.000 claims description 8
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 8
- 239000001257 hydrogen Substances 0.000 claims description 7
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 7
- 229910052710 silicon Inorganic materials 0.000 claims description 7
- QYLFHLNFIHBCPR-UHFFFAOYSA-N 1-ethynylcyclohexan-1-ol Chemical compound C#CC1(O)CCCCC1 QYLFHLNFIHBCPR-UHFFFAOYSA-N 0.000 claims description 6
- 150000001298 alcohols Chemical class 0.000 claims description 6
- 125000001931 aliphatic group Chemical group 0.000 claims description 6
- 125000003545 alkoxy group Chemical group 0.000 claims description 6
- 238000003786 synthesis reaction Methods 0.000 claims description 6
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 5
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 5
- 239000002243 precursor Substances 0.000 claims description 5
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 5
- 239000010703 silicon Substances 0.000 claims description 5
- CIUQDSCDWFSTQR-UHFFFAOYSA-N [C]1=CC=CC=C1 Chemical compound [C]1=CC=CC=C1 CIUQDSCDWFSTQR-UHFFFAOYSA-N 0.000 claims description 4
- 125000004429 atom Chemical group 0.000 claims description 4
- 229910052736 halogen Inorganic materials 0.000 claims description 4
- 150000002367 halogens Chemical class 0.000 claims description 4
- 125000005842 heteroatom Chemical group 0.000 claims description 4
- 239000001301 oxygen Substances 0.000 claims description 4
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 4
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 4
- 230000005855 radiation Effects 0.000 claims description 4
- 239000003381 stabilizer Substances 0.000 claims description 4
- 229910052727 yttrium Inorganic materials 0.000 claims description 4
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 3
- 230000009471 action Effects 0.000 claims description 3
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 3
- 230000000694 effects Effects 0.000 claims description 3
- 229910052731 fluorine Inorganic materials 0.000 claims description 3
- 239000011737 fluorine Substances 0.000 claims description 3
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 3
- 125000005023 xylyl group Chemical group 0.000 claims description 3
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 claims description 2
- 125000004975 3-butenyl group Chemical group C(CC=C)* 0.000 claims description 2
- 125000006043 5-hexenyl group Chemical group 0.000 claims description 2
- 229910004283 SiO 4 Inorganic materials 0.000 claims description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 claims description 2
- 125000002252 acyl group Chemical group 0.000 claims description 2
- 230000002411 adverse Effects 0.000 claims description 2
- 125000004450 alkenylene group Chemical group 0.000 claims description 2
- 125000002947 alkylene group Chemical group 0.000 claims description 2
- 125000003368 amide group Chemical group 0.000 claims description 2
- 150000005840 aryl radicals Chemical class 0.000 claims description 2
- 125000004104 aryloxy group Chemical group 0.000 claims description 2
- 238000009835 boiling Methods 0.000 claims description 2
- 229910052799 carbon Inorganic materials 0.000 claims description 2
- 239000000470 constituent Substances 0.000 claims description 2
- 125000003493 decenyl group Chemical group [H]C([*])=C([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 2
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 claims description 2
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 claims description 2
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 claims description 2
- 239000012429 reaction media Substances 0.000 claims description 2
- 239000011541 reaction mixture Substances 0.000 claims description 2
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 2
- 229920002554 vinyl polymer Polymers 0.000 claims description 2
- 125000001183 hydrocarbyl group Chemical group 0.000 claims 2
- 238000004132 cross linking Methods 0.000 abstract description 14
- 229920001296 polysiloxane Polymers 0.000 abstract description 9
- 230000003647 oxidation Effects 0.000 abstract description 7
- 238000007254 oxidation reaction Methods 0.000 abstract description 7
- 239000004593 Epoxy Substances 0.000 abstract description 6
- 238000000576 coating method Methods 0.000 abstract description 5
- 239000011248 coating agent Substances 0.000 abstract description 3
- 239000000654 additive Substances 0.000 abstract description 2
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 15
- 229920002545 silicone oil Polymers 0.000 description 13
- 239000003921 oil Substances 0.000 description 11
- 238000006243 chemical reaction Methods 0.000 description 8
- 239000003504 photosensitizing agent Substances 0.000 description 7
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 6
- 229910004298 SiO 2 Inorganic materials 0.000 description 6
- 238000011065 in-situ storage Methods 0.000 description 6
- 239000000123 paper Substances 0.000 description 6
- AZQWKYJCGOJGHM-UHFFFAOYSA-N 1,4-benzoquinone Chemical compound O=C1C=CC(=O)C=C1 AZQWKYJCGOJGHM-UHFFFAOYSA-N 0.000 description 5
- 0 O=C(*1)C=CC1=O Chemical compound O=C(*1)C=CC1=O 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 4
- 230000004913 activation Effects 0.000 description 4
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 238000002156 mixing Methods 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 239000000853 adhesive Substances 0.000 description 3
- 230000001070 adhesive effect Effects 0.000 description 3
- 239000003112 inhibitor Substances 0.000 description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 3
- 239000000565 sealant Substances 0.000 description 3
- 238000003860 storage Methods 0.000 description 3
- 238000009281 ultraviolet germicidal irradiation Methods 0.000 description 3
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 239000004677 Nylon Substances 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 2
- 239000012965 benzophenone Substances 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- KTWOOEGAPBSYNW-UHFFFAOYSA-N ferrocene Chemical compound [Fe+2].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 KTWOOEGAPBSYNW-UHFFFAOYSA-N 0.000 description 2
- 238000007710 freezing Methods 0.000 description 2
- 230000008014 freezing Effects 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 229920001778 nylon Polymers 0.000 description 2
- 125000002524 organometallic group Chemical group 0.000 description 2
- 229920000728 polyester Polymers 0.000 description 2
- 238000006116 polymerization reaction Methods 0.000 description 2
- 150000003283 rhodium Chemical class 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- YRHRIQCWCFGUEQ-UHFFFAOYSA-N thioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3SC2=C1 YRHRIQCWCFGUEQ-UHFFFAOYSA-N 0.000 description 2
- 125000005389 trialkylsiloxy group Chemical group 0.000 description 2
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- GISACPLIGJIBHT-UHFFFAOYSA-N C#CN(CC1CC1)ON1CC1 Chemical compound C#CN(CC1CC1)ON1CC1 GISACPLIGJIBHT-UHFFFAOYSA-N 0.000 description 1
- UDHXJZHVNHGCEC-UHFFFAOYSA-N Chlorophacinone Chemical compound C1=CC(Cl)=CC=C1C(C=1C=CC=CC=1)C(=O)C1C(=O)C2=CC=CC=C2C1=O UDHXJZHVNHGCEC-UHFFFAOYSA-N 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 1
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 1
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical compound O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 description 1
- HIDBROSJWZYGSZ-UHFFFAOYSA-N O=C(C=CC1=O)N1c1ccccc1 Chemical compound O=C(C=CC1=O)N1c1ccccc1 HIDBROSJWZYGSZ-UHFFFAOYSA-N 0.000 description 1
- 229910008329 Si-V Inorganic materials 0.000 description 1
- 229910006768 Si—V Inorganic materials 0.000 description 1
- 229910008828 WSiO Inorganic materials 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 1
- 239000011111 cardboard Substances 0.000 description 1
- 239000003060 catalysis inhibitor Substances 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 229910052570 clay Inorganic materials 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 239000011243 crosslinked material Substances 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 239000002978 dental impression material Substances 0.000 description 1
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 1
- 229910001882 dioxygen Inorganic materials 0.000 description 1
- 230000008034 disappearance Effects 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 150000002118 epoxides Chemical class 0.000 description 1
- 125000005677 ethinylene group Chemical group [*:2]C#C[*:1] 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 238000007210 heterogeneous catalysis Methods 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 238000004452 microanalysis Methods 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 239000012764 mineral filler Substances 0.000 description 1
- 238000011017 operating method Methods 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 150000003003 phosphines Chemical class 0.000 description 1
- AQSJGOWTSHOLKH-UHFFFAOYSA-N phosphite(3-) Chemical class [O-]P([O-])[O-] AQSJGOWTSHOLKH-UHFFFAOYSA-N 0.000 description 1
- 238000007146 photocatalysis Methods 0.000 description 1
- 230000001699 photocatalysis Effects 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 238000007493 shaping process Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 229920005573 silicon-containing polymer Polymers 0.000 description 1
- 229920002379 silicone rubber Polymers 0.000 description 1
- 239000000779 smoke Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 239000002023 wood Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/0091—Complexes with metal-heteroatom-bonds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/1608—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes the ligands containing silicon
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2282—Unsaturated compounds used as ligands
- B01J31/2291—Olefins
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/0006—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table compounds of the platinum group
- C07F15/0086—Platinum compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L83/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon only; Compositions of derivatives of such polymers
- C08L83/04—Polysiloxanes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2231/00—Catalytic reactions performed with catalysts classified in B01J31/00
- B01J2231/30—Addition reactions at carbon centres, i.e. to either C-C or C-X multiple bonds
- B01J2231/32—Addition reactions to C=C or C-C triple bonds
- B01J2231/323—Hydrometalation, e.g. bor-, alumin-, silyl-, zirconation or analoguous reactions like carbometalation, hydrocarbation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/80—Complexes comprising metals of Group VIII as the central metal
- B01J2531/82—Metals of the platinum group
- B01J2531/828—Platinum
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/06—Preparatory processes
- C08G77/08—Preparatory processes characterised by the catalysts used
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/12—Polysiloxanes containing silicon bound to hydrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/14—Polysiloxanes containing silicon bound to oxygen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/20—Polysiloxanes containing silicon bound to unsaturated aliphatic groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/22—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen
- C08G77/24—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen halogen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/70—Siloxanes defined by use of the MDTQ nomenclature
Definitions
- the present invention relates to the field of catalysis of crosslinking reactions between polymer chains, e.g. silicones, comprising reactive radicals capable of forming interchain bridging, so as to obtain a crosslinked material having a certain hardness and a certain mechanical strength.
- polymer chains e.g. silicones
- reactive radicals capable of forming interchain bridging
- substrates A are intended to react with substrates of type B having, per molecule, at least one reactive unsaturated aliphatic group and / or at least one reactive function, eg epoxide, in the presence of a catalyst comprising at least one platinum complex, such a reaction being initiated or activated by light rays, of wavelength, chosen, preferably, in the field of ultraviolet (UV).
- UV ultraviolet
- the present invention relates, first of all, to new catalytic systems formed by photoactive organoplatinic complexes in the crosslinking by hydrosilylation of polyorganosiloxanes A of Si-H type and of compounds B with aliphatic unsaturation and / or with reactive function. .
- the present invention also relates to a hydrosilylation process in which the above-mentioned catalytic systems are used, as well as compositions which can be crosslinked by photoactivation and which contain, inter alia, the substrates A and B and the above-mentioned catalyst.
- coatings formed from a mixture of silicone oils which can be crosslinked together and which are intended to form non-stick layers on fibrous supports such as paper, dental impressions, adhesives, sealants, jointing materials, etc.
- non-stick paper application among others, cross-linking in situ by photoactivation, eg by UV irradiation, is particularly advantageous insofar as it allows high coating speeds.
- this in situ crosslinking can take place easily at room temperature. It also avoids the use of solvents, the elimination of which is costly and difficult.
- these catalysts are in the form of organometallic complexes (platinum or the like).
- platinum has proven to be the most suitable metal for entering into the composition of these organometallic complexes.
- the source of the platinum in this solution is formed by the catalyst of SPEIER or hexachloroplatinic acid.
- This KARSTEDT catalyst has the drawback of not being part of the photoactivatable hydrosilylation catalysts.
- the activation adapted to this KARSTEDT catalyst is thermoactivation, which is, all the same, much more restrictive and expensive than a simple UV activation, of the type of that targeted by the invention.
- Another major defect of the catalyst of the KARSTEDT solution is that it has a relatively poor storage stability (gel time at 25 ° C less than or equal to 1 min).
- Another objective of the invention is to provide a hydrosilylation process by photoactivation of reactive products, and in particular of silicones, in which the abovementioned catalytic systems are used, said process having to be easy to carry out, fast, and inexpensive.
- Another objective of the invention is to propose compositions of silicone oils crosslinkable by photoactivation:
- compositions stable in storage, can be used, for example, for paper anti-adhesion, dental impressions, sealing or jointing materials, adhesives, or for any other application in which it is advantageous to use performs cross-linking in situ of silicone elastomers.
- R 1 , R 2 , R 3 , R 4 are different or similar to each other and represent an alkyl and / or acyl group, preferably an alkyl having from 1 to 18 carbon atoms, methyl, ethyl, propyl, pentyl groups , hexyl, allyl, acetyl, propionyl being particularly preferred and especially methyl, ⁇ L 1 being a ligand of the following formula:
- ⁇ Z 1 , Z 2 are identical or different and:
- alkyl and alkenyl residues considered above being linear or branched and containing from 1 to 6 carbon atoms
- ⁇ either are two atoms linked to each other and belonging to a preferably aromatic ring.
- ⁇ Y 1 , Y 2 are identical or different and representing an alkyl, aryl radical preferably phenyl, alkyloxy, oryloxy, hydrogen or halogen,
- ⁇ L 2 being a ligand consisting of one of the following two residues (2 1 ), (2 2 ):
- Y 3 to Y 6 on the one hand and E 3 , E 4 on the other hand are identical or different and correspond to the same definition as that given above for Y 1 , Y 2 and E 1 , E 2 respectively,
- Y 3 and Y 4 and / or Y 5 and Y 6 can also together form a cyclic group, preferably an aromatic group comprising one or more aromatic rings having from 6 to 8 members, being in ortho or pericondensed form, and optionally substituted by monovalent radicals corresponding to the definition given above for Y 3 to Y 6 ;
- Z 3 , Z 5 are identical or different and correspond to:
- CR 13 with R 13 H or lower alkyl C 1 , C 6 , N;
- Z 4 is a residue comprising from 2 to 5 phenylene radicals linked together by:
- the ligand L is advantageously a chromophore. It can be varied in nature. This ligand L is also decisive with regard to the definition of two preferred species C 1 and C 2 in accordance with the invention.
- the first species C 1 is that in which there is only one platinum atom of real and formal degree of oxidation 0, complexed with the siloxane residue and the ligand L 1 , via the ⁇ double bonds.
- the second species C 2 has two platinum atoms, also of real and formal oxidation state 0 and complexed each with a siloxane residue and the ligand L 2 : (2 1 ), (2 2 ).
- the subject of the invention is also a new photoactivatable catalytic system - possibly thermoactivable - based on platinum and useful in particular in the hydrosilylation between, on the one hand, polyorganosiloxanes A having, per molecule, at least one reactive radical Si- H and free of silicon atom linked to more than two hydrogen atoms and, on the other hand, of compounds B having at least one reactive group and / or at least one reactive polar function,
- Such catalytic systems are particularly advantageous in the context of in situ crosslinking techniques, for example of silicones, in which “ready-to-use” compositions are used and comprising both polymers intended for react between them and a photosensitive catalytic system, so that this considerably simplifies their application.
- These compositions are known to those skilled in the art under the name "single-component".
- the photoactivatable catalytic systems according to the invention make it possible to control the crosslinking conditions and therefore the final mechanical properties targeted for the crosslinked elastomeric compositions. It is thus possible to adapt these to the intended application (e.g. non-stick, paper).
- the catalytic system of which it is a question in the present presentation can consist of a mixture of complexes C 1 , C 2 with ligands of formulas (1) and / or (2 1 ) and / or (2 2 ).
- the complexes C 1 are chosen from the following compounds, taken alone or as a mixture:
- the C 2 complexes are selected from the following compounds, taken alone or as a mixture
- the catalytic system contains, in the free state, at least one stabilizing agent formed by at least one ligand Ls of formula L 1 or L 2 as defined above.
- the ligand Ls forming the stabilizing agent can be identical or different to the ligands (L 1 , L 2 ) of the complexes (I): C 1 , C 2 .
- the compound Ls in the catalytic system in an excess of 1 to 30, preferably 10 to 20 molar equivalents relative to the platinum present.
- This Ls compound can in fact be considered as a catalyst inhibitor. It is moreover possible to use in combination or as a replacement for Ls other conventional inhibitors of hydrosilylation catalysts. These inhibitors also known under the name of thermal blockers make it possible to increase the "pot life" of the compositions considered (stabilities greater than 3-5 days) without harming the kinetics of hydrosililation.
- R is a linear or branched alkyl radical, or a phenyl radical
- R ' is H or a linear or branched alkyl radical, or a phenyl radical
- the total number of carbon atoms contained in R and R 1 being at least 5, preferably from 9 to 20.
- Said alcohols are preferably chosen from those having a boiling point above 250 ° C. Mention may be made, by way of examples:
- the synthesis of the platinum complexes (I) according to the invention consists in reacting a ligand L with products of average formula [Pt 2 ] [ViMe 2 Si - O - Si Me 2 Vi] 3 . These products are preferably formed by a KARSTEDT catalyst solution.
- This reaction can be carried out in bulk or in a solvent, such as toluene, ethanol or isopropanol.
- the catalytic system can comprise a precursor of the complexes (I), eg C 1 or C 2 .
- a precursor of the complexes (I) eg C 1 or C 2 .
- Such a precursor is advantageously constituted by a composition of the starting materials for the synthesis of the complexes
- the formation of the platinum complexes takes place in situ, before and / or during the use of the crosslinkable composition containing the precursor composition. This greatly simplifies the preparation.
- catalytic system according to the invention is sufficiently reactive and efficient to dispense with the use of photosensitizers, it is entirely possible, for the sake of optimization, to use such products.
- At least one photosensitizer having a triplet energy greater than or equal to 31 Kcal / mole and selected from (poly) aromatic (optionally metallic) and heterocyclic products, preferably in the following list of products: toluene, pyridine, ferrocene, benzene, thioxanthone, anthracene, benzophenone, this selection being made so that the triplet lifetimes of the platinum complexes and of the photosensitizer (s) are preferably of the same order.
- these photosensitizers are used at a rate of 5 ⁇ 10 -3 to
- the photosensitive catalytic system according to the invention is non-reducible, so that it does not tend to react with the reactive Si-Hs of type A polyorganosiloxanes.
- the present invention relates to a hydrosilylation process between, on the one hand, polyorganosiloxanes A having, per molecule, at least one reactive radical Si-H and free of silicon atoms bound to more than two hydrogen atoms, and on the other hand, compounds B preferably chosen from polyorganosiloxanes and having, per molecule, at least one unsaturated aliphatic reactive group and / or at least one reactive function, process in which one implements at least one platinum complex as a catalyst.
- This process differs from its similar in that it consists essentially in reacting compounds A and B in the presence of a catalytic system of the type of that according to the invention described above, and / or by using simultaneously a synthesis of complexes (I) in which a precursor is formed, as described above, formed by a composition of the starting materials (excess ligand + KARSTEDT catalyst) and possibly using at least the constituents of the above catalytic system, the initiation of the reaction being at least partly carried out by photoactivation.
- the compounds A are chosen from polyorganohydrogensiloxanes comprising:
- the symbols W are similar or different and representing a monovalent hydrocarbon group, free from any adverse action on the activity of the catalyst and preferably chosen from (cyclo) alkyl groups having from 1 to 18 carbon atoms included, and, advantageously, among the methyl, ethyl, propyl, octyl and 3,3,3-trifluoropropyl groups and also among the C 6 -C 12 aryl or aralkyl groups and, advantageously, among the xylyl and totyl and phenyl radicals,
- Organopolysiloxane A can only be formed of units of formula (1) or additionally comprise units of formula (2).
- Organopolysiloxane A can have a linear, branched or unbranched, cyclic or network structure. The degree of polymerization is greater than or equal to 2. More generally, it is less than 5,000.
- linear polymers When linear polymers are involved, these essentially consist of "D" W 2 SiO 2/2 , WHSiO 2/2 , and “M” W 3 SiO 1/2 or W 2 HSiO 1/2 units. , WH 2 SiO 1/2 ; the blocking terminal "M” units can be trialkylsiloxy, dialkylarylsiloxy groups.
- terminal "M” units mention may be made of trimethylsiloxy, dimethylphenylsiloxy, dimethylethoxysiloxy, dimethylethyltriethoxysilylsiloxy groups, etc.
- units "D” mention may be made of dimethylsiloxy, methylphenylsiloxy groups.
- Said linear polyorganosiloxanes A can be oils of dynamic viscosity at 25 ° C of the order of 1 to 100,000 mPa.s at 25oC, generally of the order of 10 to 5,000 mPa.s at 25 ° C, or gums having a molecular weight of the order of 1,000,000.
- cyclic polyorganosiloxanes consist of "D" W 2 SiO 2/2 , and WHSiO 2/2 units , which may be of the dialkylsiloxy or alkylarylsiloxy type.
- Said cyclic polyorganosiloxanes have a viscosity of the order of 1 to 5000 mPa.s.
- the dynamic viscosity at 25 ° C all the silicone polymers considered in this presentation can be measured using a BROOKFIELD viscometer, according to AFNOR NFT 76 102 standard of February 1972.
- the viscosity in question in this presentation is the dynamic viscosity at 25 ° C called “Newtonian”, that is to say the dynamic viscosity which is measured, in a manner known per se, at a shear rate gradient sufficiently low so that the viscosity measured is independent of the speed gradient.
- organopolysiloxanes A are:
- compounds B they are selected from polyorganosiloxanes comprising similar or different units of formula:
- the symbols X are similar or different and represent a hydrogen atom or a reactive function, preferably epoxyfunctional, linked to silicon by a divalent radical eg C 1 -C 20 optionally comprising at least one heteroatom, the radicals X more particularly preferred being: 3-glycidoxypropyl, 4-ethanediyl (1,2-epoxycylohexyl) ...,
- Y are similar or different and represent a linear or branched C 1 -C 12 alkenyl residue, and having at least one ethylenic unsaturation at the chain end and / or in the chain and optionally at least one heteroatom;
- ⁇ d, e and f are 0, 1, 2 or 3;
- Y residues they are advantageously chosen from the following list: vinyl, propenyl, 3-butenyl, 5-hexenyl, 9 decenyl, 10-undecenyl, 5.9 -decadienyl, 6-11-dodecadienyl.
- Organopolysiloxanes B can have a linear structure (branched or not) cyclic or network. Their degree of polymerization is preferably between 2 and 5000.
- the terminal blocking "M” units can be trialkylsiloxy, dialkylarylsiloxy, dialkylvinylsiloxy, dialkylalkenylsiloxy groups.
- terminal "M” units examples include trimethylsiloxy, dimethylphenylsiloxy, dimethylvinylsiloxy, dimethylhexenylsiloxy, dimethylethoxysiloxy, dimethylethyltriethoxysilylsiloxy groups, etc.
- units "D” mention may be made of dimethylsiloxy, methylphenylsiloxy, methylvinylsiloxy, methylbutenylsiloxy, methylhexenylsiloxy, methyldecenylesiloxy, methyldecadienylsiloxy, methyl-3-hydroxypropylsiloxy, methylpropyl-3-glycidoxy-methyl-3-propoxy , 4'-epoxycyclohexyl) ethylsiloxy, methylbutoxysiloxy, methyl- ⁇ -trimethoxysilylethylsiloxy, methyl- ⁇ -triethoxysilylethylsiloxy.
- Said linear polyorganosiloxanes B can be oils of dynamic viscosity at 25 ° C of the order of 1 to 100,000 mPa.s at 25 ° C, generally of the order of
- cyclic polyorganosiloxanes B consist of "D" units W 2 SiO 2/2 , WSiO 2/2 , WYSiO 2/2 , which may be of the dialkylsiloxy, alkylarylsiloxy, alkylvinylsiloxy, alkylYsiloxy type. , alkylXsiloxy; examples of such patterns have already been cited above.
- Said cyclic polyorganosiloxanes B have a viscosity of the order of 1 to 5000 mPa.s.
- the compounds B with aliphatic unsaturation useful in the context of the process according to the invention are, for example, those with olefinic or acetylene unsaturation well known in the technical field considered.
- the reaction mixture comprises compounds A and compounds B in an amount such that the molar ratio
- Si-H / unsaturated groups and / or reactive function e.g. epoxy either between 0.4 and 10 preferably between 1 and 4 and, more preferably still, is of the order of
- the catalyst being present in an amount from 1 to 400, preferably from 10 to 300 and, more preferably, from 20 to 200 ppm of platinum metal, expressed by weight relative to the compounds A and B present.
- the technical literature of the field considered gives all the other useful information, as for the reaction conditions of hydrosilylation by photoactivation (A / B mixture and UV irradiation at 25 ° C).
- crosslinking reactions of silicone oils in particular hydrosilylation (e. G: Si-H / Si-Vi), catalyzed by photosensitive complexes of platinum, such as those according to the invention, pass through the formation of particles. colloids, useful as a support for platinum, according to a heterogeneous catalysis process.
- photoactivation preferably takes place in the ultraviolet range.
- thermoactivation is preferably carried out using infrared radiation and it advantageously takes place after the photoactivation.
- a last aspect of the invention which is mentioned in a nonlimiting manner in the present description, relates to a crosslinkable and in particular photocrosslinkable composition, characterized in that it comprises the compounds A and B, and a catalyst as defined above.
- the above composition can also comprise at least one photosensitizer having a triplet energy greater than or equal to 31 Kcal / mole and selected from (poly) aromatic (optionally metallic) and heterocyclic products, and preferably from the following list of products: toluene, pyridine, ferrocene, benzene, thioxanthone, anthracene, benzophenone, this selection being made so that the triplet lifetimes of the platinum complexes and (of ) photosensitizer (s) are, advantageously, of the same order.
- at least one photosensitizer having a triplet energy greater than or equal to 31 Kcal / mole and selected from (poly) aromatic (optionally metallic) and heterocyclic products, and preferably from the following list of products: toluene, pyridine, ferrocene, benzene, thioxanthone, anthracene, benzophenone, this selection being made so that the triplet lifetimes of the platinum complexe
- compositions in accordance with the invention are prepared either before (or even long before) or even immediately before the implementation of the hydrosilylation process. It should be noted that these compositions are particularly stable on storage and that, in accordance with the process of the invention, they offer rapid crosslinking kinetics. In addition, their uncrosslinked state, before exposure to the activation light radiation, offers great ease of handling, application or placement on different supports or other shaping molds.
- compositions and / or the method according to the invention can incorporate different additives, chosen according to the intended final application. It can be, for example, mineral or non-mineral fillers and / or pigments such as synthetic or natural fibers (polymers) ground calcium carbonate, talc, clay, titanium dioxide or silica smoke. This can improve e.g. the mechanical characteristics of the final materials.
- Soluble dyes, oxidation inhibitors and / or any other material which does not interfere with the catalytic activity of the platinum complex and does not absorb in the wavelength range chosen for photoactivation, can also be added. to the composition or used in the context of the process according to the invention.
- the composition can be applied, for various purposes, to the surface of any solid substrate.
- solid supports can be paper, cardboard, wood, plastic (e.g. polyester, nylon, polycarbonate), fibrous supports woven or not made of cotton, polyester, nylon etc, or a metal, glass or ceramic.
- the applications, more particularly, targeted by the catalyst, the process and the compositions according to the invention are, in particular, those of cross-linkable silicone oils "in situ", useful for the preparation of non-stick coatings on fibrous supports of any kind and in particular on paper.
- the above compositions make it possible to achieve very high coating speeds, due to their very rapid crosslinking kinetics.
- dental impression materials adhesives, sealants, sealants, adhesion primers.
- the operating method used consists in reacting a ligand of type ⁇ (maleic anhydride, Benzoquinone, maleimide, etc.) on the KARSTEDT complex of ideal formula: Pt 2 (ViMe 2 Si-O-SiMe 2 Vi) 3 .
- This reaction can be carried out in bulk or in a solvent such as toluene, ethanol or isopropanol, according to the following sequences:
- Fig. 1 attached represents the kinetics of hydrosilylation of silicone oils with Si-H and Si-Vi units.
- the silicone oils used have the following structures: - Oils with Si-H units: Me 3 SiO - (- SiMe 2 O) 15.6 (-SiMeHO) 8] -SiMe 3
- the catalytic systems to be tested and an excess of ligand are introduced into the Si-Vi pattern oil. Then add this mixture to the Si-H pattern oil.
- the platinum level is 200 ppm relative to the total mass.
- the Si-H / Si-Vi molar ratio is 1.7. 20 molar equivalents of ligand are used.
- the irradiation is carried out using an HPK 125 lamp with a power: 120 mJ / cm 2 .mn.
- the thickness of the film studied is 24 ⁇ m.
- the kinetics of disappearance of the Si-H patterns is carried out by FTIR by measuring the decrease in the vibration band v Si-H. The tests are carried out at 25 ° C. TABLE 2 below brings together the main systems used.
- Fig. 2 attached represents the kinetics of hydrosilylation of a mixture of silicone oils with Si-H / Si-Vi units described in Example 2, catalyzed by the following complex:
- Fig. 3 attached represents the comparison of the kinetics observed for the hydrosilylation of silicone oils with Si-H / Si-Vi units (identical to those of Example 2) with the two catalytic systems:
- Fig. 4 represents the various kinetics observed using the catalytic system of the following formula:
- the catalytic mixture is introduced into an oil with Si-Vi units (idem example NO 2) and then into a silicone oil with Si-H units.
- the Si-H / Si-Vi molar ratio 1.7.
- a film with a thickness of 24 ⁇ m is produced.
- the platinum concentration is 200 ppm. 6.3 DETERMINATION OF THE FREEZE TIME AT 250 ° C
- Figure 5 attached shows the hydrosilylation kinetics observed for O, P and Q.
- Figure 6 attached shows the hydrosilylation kinetics observed for R and S.
- the silicone oil with Si-alkenyl patterns is as follows:
- Figure 7 shows the hydrosilylation kinetics obtained.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Inorganic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Silicon Polymers (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Catalysts (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP7524428A JPH09507677A (ja) | 1994-03-18 | 1995-03-17 | 有機白金錯体及びこれらを含有する光活性化性ヒドロシリル化触媒系 |
| EP95913212A EP0750623A1 (fr) | 1994-03-18 | 1995-03-17 | Complexes du platine et catalyseurs d'hydrosilylation photoactivables les contenant |
| AU20762/95A AU680129B2 (en) | 1994-03-18 | 1995-03-17 | Platinum complexes and light-activatable hydrosilylation catalysts containing same |
| FI963668A FI963668A (fi) | 1994-03-18 | 1996-09-17 | Platinakompleksit ja niitä sisältävät, valolla aktivoitavissa olevat hyrosilylointikatalyytit |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR9403442A FR2717482B1 (fr) | 1994-03-18 | 1994-03-18 | Complexes organoplatiniques et systèmes catalytiques photoactivables d'hydrosilylation en contenant. |
| FR94/03442 | 1994-03-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1995025734A1 true WO1995025734A1 (fr) | 1995-09-28 |
Family
ID=9461370
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR1995/000328 WO1995025734A1 (fr) | 1994-03-18 | 1995-03-17 | Complexes du platine et catalyseurs d'hydrosilylation photoactivables les contenant |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP0750623A1 (fr) |
| JP (1) | JPH09507677A (fr) |
| AU (1) | AU680129B2 (fr) |
| CA (1) | CA2185823A1 (fr) |
| FI (1) | FI963668A (fr) |
| FR (1) | FR2717482B1 (fr) |
| WO (1) | WO1995025734A1 (fr) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2750349A1 (fr) * | 1996-06-28 | 1998-01-02 | Rhone Poulenc Chimie | Utilisation de complexes du platine notamment a titre de catalyseurs d'hydrosilylation homogenes et thermoactivables |
| WO2002014407A1 (fr) * | 2000-08-17 | 2002-02-21 | Rhodia Chimie | Procede de preparation d'huiles silicones par hydrosilylation de synthons contenant au moins un cycle hydrocarbone dans lequel est inclus un atome d'oxygene en presence d'un complexe metallique catalytique |
| US6562470B2 (en) | 2001-01-10 | 2003-05-13 | General Electric Company | Method for making coated substrates and articles made thereby |
| WO2003085058A1 (fr) * | 2002-04-04 | 2003-10-16 | 3M Innovative Properties Company | Compositions cuites transparentes aux rayonnements ultraviolets |
| WO2008113763A1 (fr) * | 2007-03-21 | 2008-09-25 | Wacker Chemie Ag | Quinones à substitution si utilisées comme co-catalyseurs dans l'hydrosilylation catalysée par un métal de transition |
| US8207241B2 (en) | 2004-12-09 | 2012-06-26 | Wacker Chemie Ag | Platinum catalysts supported on nanosize titanium dioxide, their use in hydrosilylation and compositions comprising such catalysts |
| WO2023111478A1 (fr) | 2021-12-17 | 2023-06-22 | Elkem Silicones France Sas | Composition silicone réticulable par irradiation |
| WO2023209140A1 (fr) | 2022-04-28 | 2023-11-02 | Elkem Silicones France Sas | Composition silicone réticulable par irradiation comprenant du pt(octane-2,4-dione)2 comme catalyseur |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE452928T1 (de) * | 2005-06-14 | 2010-01-15 | Dow Corning | Angereicherte silikonharzfolie und verfahren für ihre zubereitung |
| US7649071B2 (en) * | 2006-09-01 | 2010-01-19 | Momentive Performance Materials Inc. | Branched polysiloxane composition |
| WO2012084992A1 (fr) | 2010-12-21 | 2012-06-28 | Bluestar Silicones France Sas | Composition silicone reticulable par hydrosilylation et procede de revetement ou de fabrication d'objets a partir de cette composition |
| JP2013087199A (ja) * | 2011-10-18 | 2013-05-13 | Shin-Etsu Chemical Co Ltd | 付加硬化型オルガノポリシロキサン組成物の硬化方法 |
| JP7060132B2 (ja) * | 2020-04-21 | 2022-04-26 | 三菱ケミカル株式会社 | 粘着剤組成物及び粘着シート |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3775452A (en) * | 1971-04-28 | 1973-11-27 | Gen Electric | Platinum complexes of unsaturated siloxanes and platinum containing organopolysiloxanes |
-
1994
- 1994-03-18 FR FR9403442A patent/FR2717482B1/fr not_active Expired - Fee Related
-
1995
- 1995-03-17 EP EP95913212A patent/EP0750623A1/fr not_active Ceased
- 1995-03-17 AU AU20762/95A patent/AU680129B2/en not_active Ceased
- 1995-03-17 JP JP7524428A patent/JPH09507677A/ja active Pending
- 1995-03-17 CA CA002185823A patent/CA2185823A1/fr not_active Abandoned
- 1995-03-17 WO PCT/FR1995/000328 patent/WO1995025734A1/fr not_active Application Discontinuation
-
1996
- 1996-09-17 FI FI963668A patent/FI963668A/fi unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3775452A (en) * | 1971-04-28 | 1973-11-27 | Gen Electric | Platinum complexes of unsaturated siloxanes and platinum containing organopolysiloxanes |
Non-Patent Citations (1)
| Title |
|---|
| PETER B. HITCHCOCK: "Synthesis and structure of a rac-Tris(divinyldisiloxane)diplatinum(o) Complex and its Reaction with Maleic Anhydride", ANGEWANDTE CHEMIE. INTERNATIONAL EDITION, vol. 30, no. 4, 1991, WEINHEIM DE, pages 438 - 440 * |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2750349A1 (fr) * | 1996-06-28 | 1998-01-02 | Rhone Poulenc Chimie | Utilisation de complexes du platine notamment a titre de catalyseurs d'hydrosilylation homogenes et thermoactivables |
| WO1998000463A1 (fr) * | 1996-06-28 | 1998-01-08 | Rhodia Chimie | Utilisation de complexes du platine notamment a titre de catalyseurs d'hydrosilylation homogenes et thermoactivables |
| WO2002014407A1 (fr) * | 2000-08-17 | 2002-02-21 | Rhodia Chimie | Procede de preparation d'huiles silicones par hydrosilylation de synthons contenant au moins un cycle hydrocarbone dans lequel est inclus un atome d'oxygene en presence d'un complexe metallique catalytique |
| FR2813081A1 (fr) * | 2000-08-17 | 2002-02-22 | Rhodia Chimie Sa | Procede de preparation d'huiles silicones par hydrosilylation de synthons contenant au moins un cycle hydrocarbone dans lequel est inclus un atome d'oxygene en presence d'un complexe metallique catalytique |
| CN100390219C (zh) * | 2000-08-17 | 2008-05-28 | 罗狄亚化学公司 | 在催化金属络合物的存在下通过氢化硅烷化含有至少一个其中包括氧原子的烃环的合成子来制备硅油的方法 |
| US6562470B2 (en) | 2001-01-10 | 2003-05-13 | General Electric Company | Method for making coated substrates and articles made thereby |
| GB2401369A (en) * | 2002-04-04 | 2004-11-10 | 3M Innovative Properties Co | Cured compositions transparent to ultraviolet radiation |
| WO2003085058A1 (fr) * | 2002-04-04 | 2003-10-16 | 3M Innovative Properties Company | Compositions cuites transparentes aux rayonnements ultraviolets |
| US8207241B2 (en) | 2004-12-09 | 2012-06-26 | Wacker Chemie Ag | Platinum catalysts supported on nanosize titanium dioxide, their use in hydrosilylation and compositions comprising such catalysts |
| WO2008113763A1 (fr) * | 2007-03-21 | 2008-09-25 | Wacker Chemie Ag | Quinones à substitution si utilisées comme co-catalyseurs dans l'hydrosilylation catalysée par un métal de transition |
| DE102007013608A1 (de) | 2007-03-21 | 2008-09-25 | Wacker Chemie Ag | Si-substituierte Chinone als Co-Katalysatoren in der Übergangsmetall-katalysierten Hydrosilylierung |
| WO2023111478A1 (fr) | 2021-12-17 | 2023-06-22 | Elkem Silicones France Sas | Composition silicone réticulable par irradiation |
| FR3130816A1 (fr) | 2021-12-17 | 2023-06-23 | Elkem Silicones France Sas | Composition silicone réticulable par irradiation |
| WO2023209140A1 (fr) | 2022-04-28 | 2023-11-02 | Elkem Silicones France Sas | Composition silicone réticulable par irradiation comprenant du pt(octane-2,4-dione)2 comme catalyseur |
| FR3135086A1 (fr) | 2022-04-28 | 2023-11-03 | Elkem Silicones France Sas | Composition silicone réticulable par irradiation comprenant du Pt(octane-2,4-dione)2 comme catalyseur |
Also Published As
| Publication number | Publication date |
|---|---|
| FR2717482A1 (fr) | 1995-09-22 |
| AU2076295A (en) | 1995-10-09 |
| AU680129B2 (en) | 1997-07-17 |
| FR2717482B1 (fr) | 1996-06-21 |
| EP0750623A1 (fr) | 1997-01-02 |
| FI963668A (fi) | 1996-11-15 |
| JPH09507677A (ja) | 1997-08-05 |
| FI963668A0 (fi) | 1996-09-17 |
| CA2185823A1 (fr) | 1995-09-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0703236B1 (fr) | Amorceurs de réticulation, par voie cationique, de polymères à groupements organofonctionnels compositions à base de polyorganosiloxanes réticulables et contenant ces amorceurs et application desdites compositions en antiadhérence | |
| EP3215556B1 (fr) | Nouveaux catalyseurs de réticulation de compositions silicones | |
| EP1701992B1 (fr) | Procede de preparation de polyorganosiloxanes (pos) par polymerisation par ouverture de cycle(s) et/ou redistribution de pos, en presence de carbene(s) et compositions de pos mises en oeuvre dans ce procede | |
| FR2575086A1 (fr) | Complexe platine-alcenylcyclohexene comme catalyseur de reaction d'hydrosilylation et son procede de preparation | |
| WO1995025734A1 (fr) | Complexes du platine et catalyseurs d'hydrosilylation photoactivables les contenant | |
| EP0188978A1 (fr) | Complexe platine-triene comme catalyseur de réaction d'hydrosilylation et son procédé de préparation | |
| WO1996016125A1 (fr) | Polyorganosiloxanes fonctionnalises et l'un de leurs procedes de preparation | |
| FR2707993A1 (fr) | Dérivés mono et di-alcényl substitués de l'acide maléique utilisés pour empêcher le durcissement prématuré de compositions de polysiloxanes de moulage, compositions de polysiloxanes de moulage contenant de tels inhibiteurs. | |
| EP1633830B1 (fr) | Composition silicone reticulable en gel adhesif | |
| FR2500842A1 (fr) | Composition de caoutchouc de silicone pour l'enrobage d'elements photovoltaiques | |
| FR2727119A1 (fr) | Polyorganosiloxanes fonctionnalises et l'un de leurs procedes de preparation | |
| FR2831548A1 (fr) | Composition silicone adhesive reticulable comprenant comme agent thixotropant un compose a fonction amine cyclique portee par une chaine siloxanique | |
| EP1969034A1 (fr) | Procédé de lutte contre l'apparition de brouillard lors de l'enduction de supports flexibles avec une composition silicone liquide réticulable, dans un dispositif a cylindres | |
| WO1998000463A1 (fr) | Utilisation de complexes du platine notamment a titre de catalyseurs d'hydrosilylation homogenes et thermoactivables | |
| WO1995025735A1 (fr) | Complexes du platine et catalyseurs d'hydrosilylation photoactivables les contenant | |
| WO2003092890A2 (fr) | Ensemble catalytique pour hydrosilylation, son procede de preparation et compositions silicone l'incorporant | |
| EP0421895B1 (fr) | Composés d'étain (IV) éventuellement chélatés utilisables notamment comme catalyseurs latents | |
| FR2802540A1 (fr) | Procede de preparation de polyorganosiloxanes par polymerisation catalysee par un systeme catalytique a base d'acide triflique ou de derives de l'acide triflique | |
| FR2591916A1 (fr) | Catalyseur d'hydrosilylation, son procede de preparation et son utilisation | |
| FR2600661A1 (fr) | Compositions organopolysiloxanes polyaddition pour l'enduction de materiaux souples | |
| FR2616152A1 (fr) | Nouveaux polysilanes, polysilanes modifies correspondants, leur preparation et leur application dans des compositions reticulables | |
| FR2737215A1 (fr) | Polyorganosiloxanes perhalogenes et leurs procedes d'obtention | |
| EP3559088B1 (fr) | Compose du cobalt utile comme catalyseur d'hydrosilylation, de silylation deshydrogenante et de reticulation de compositions silicones | |
| FR2707655A1 (fr) | Nouveaux polymères siliconés à fonctions oléfiniques, leur procédé de préparation et compositions durcissables comprenant lesdits polymères. | |
| EP0338947A1 (fr) | Composition polyorganosiloxane contenant un catalyseur latent à l'étain et reticulable en couche mince |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AU CA FI JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2185823 Country of ref document: CA Ref document number: 963668 Country of ref document: FI |
|
| ENP | Entry into the national phase |
Ref document number: 1996 716356 Country of ref document: US Date of ref document: 19960918 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1995913212 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1995913212 Country of ref document: EP |
|
| WWR | Wipo information: refused in national office |
Ref document number: 1995913212 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1995913212 Country of ref document: EP |