USRE49582E1 - Therapeutic compounds and compositions - Google Patents
Therapeutic compounds and compositions Download PDFInfo
- Publication number
- USRE49582E1 USRE49582E1 US17/131,562 US202017131562A USRE49582E US RE49582 E1 USRE49582 E1 US RE49582E1 US 202017131562 A US202017131562 A US 202017131562A US RE49582 E USRE49582 E US RE49582E
- Authority
- US
- United States
- Prior art keywords
- compound
- mhz
- nmr
- piperazine
- quinoline
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active, expires
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/02—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings
- C07D241/10—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D241/14—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D241/20—Nitrogen atoms
- C07D241/22—Benzenesulfonamido pyrazines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/16—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D215/36—Sulfur atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4965—Non-condensed pyrazines
- A61K31/497—Non-condensed pyrazines containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/16—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D215/38—Nitrogen atoms
- C07D215/40—Nitrogen atoms attached in position 8
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/32—One oxygen, sulfur or nitrogen atom
- C07D239/42—One nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/02—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings
- C07D241/10—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D241/14—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D241/20—Nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D243/00—Heterocyclic compounds containing seven-membered rings having two nitrogen atoms as the only ring hetero atoms
- C07D243/06—Heterocyclic compounds containing seven-membered rings having two nitrogen atoms as the only ring hetero atoms having the nitrogen atoms in positions 1 and 4
- C07D243/08—Heterocyclic compounds containing seven-membered rings having two nitrogen atoms as the only ring hetero atoms having the nitrogen atoms in positions 1 and 4 not condensed with other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D271/00—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms
- C07D271/12—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms condensed with carbocyclic rings or ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/60—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings condensed with carbocyclic rings or ring systems
- C07D277/62—Benzothiazoles
- C07D277/64—Benzothiazoles with only hydrocarbon or substituted hydrocarbon radicals attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/60—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings condensed with carbocyclic rings or ring systems
- C07D277/62—Benzothiazoles
- C07D277/68—Benzothiazoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/16—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms
- C07D295/18—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms by radicals derived from carboxylic acids, or sulfur or nitrogen analogues thereof
- C07D295/182—Radicals derived from carboxylic acids
- C07D295/192—Radicals derived from carboxylic acids from aromatic carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D319/00—Heterocyclic compounds containing six-membered rings having two oxygen atoms as the only ring hetero atoms
- C07D319/10—1,4-Dioxanes; Hydrogenated 1,4-dioxanes
- C07D319/14—1,4-Dioxanes; Hydrogenated 1,4-dioxanes condensed with carbocyclic rings or ring systems
- C07D319/16—1,4-Dioxanes; Hydrogenated 1,4-dioxanes condensed with carbocyclic rings or ring systems condensed with one six-membered ring
- C07D319/18—Ethylenedioxybenzenes, not substituted on the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D407/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00
- C07D407/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing two hetero rings
- C07D407/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having oxygen atoms as the only ring hetero atoms, not provided for by group C07D405/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/582—Recycling of unreacted starting or intermediate materials
Definitions
- Cancer cells rely primarily on glycolysis to generate cellular energy and biochemical intermediates for biosynthesis of lipids and nucleotides, while the majority of “normal” cells in adult tissues utilize aerobic respiration. This fundamental difference in cellular metabolism between cancer cells and normal cells, termed the Warburg Effect, has been exploited for diagnostic purposes, but has not yet been exploited for therapeutic benefit.
- PK Pyruvate kinase
- L and R isoforms are expressed in liver and red blood cells
- M1 isoform is expressed in most adult tissues
- M2 isoform is a splice variant of M1 expressed during embryonic development. All tumor cells exclusively express the embryonic M2 isoform.
- M2 is a low-activity enzyme that relies on allosteric activation by the upstream glycolytic intermediate, fructose-1,6-bisphosphate (FBP), whereas M1 is a constitutively active enzyme.
- FBP fructose-1,6-bisphosphate
- PKM2 is also expressed in adipose tissue and activated T-cells.
- the modulation (e.g. inhibition or activation) of PKM2 may be effective in the treatment of, e.g., obesity, diabetes, autoimmune conditions, and proliferation-dependent diseases, e.g., benign prostatic hyperplasia (BPH).
- BPH benign prostatic hyperplasia
- Current inhibitors of pyruvate kinase are not selective, making it difficult to treat disease related to pyruvate kinase function.
- PKM2 phosphotyrosine peptide binding to PKM2 leads to a dissociation of FBP from PKM2 and conformational changes of PKM2 from an active, tetrameric form to an inactive form.
- Compounds that bind to PKM2 and lock the enzyme in the active confirmation will lead to the loss of allosteric control of PKM2 needed for shunting biochemical intermediates from glycolysis into biosynthesis of nucleotides and lipids.
- the activation of PKM2 i.e., activators of PKM2
- Described herein are compounds that modulate pyruvate kinase M2 (PKM2) and pharmaceutically acceptable salts, solvates, and hydrates thereof, for example, compounds that activate PKM2.
- PKM2 pyruvate kinase M2
- pharmaceutical compositions comprising a compound provided herewith and the use of such compositions in methods of treating diseases and conditions that are related to pyruvate kinase function (e.g., PKM2 function), including, e.g., cancer, diabetes, obesity, autoimmune disorders, and benign prostatic hyperplasia (BPH).
- composition comprising a compound or a pharmaceutically acceptable salt of formula (I):
- W, X, Y and Z are each independently selected from CH or N;
- D and D 1 are independently selected from a bond or NR b ;
- A is optionally substituted aryl or optionally substituted heteroaryl
- L is a bond, —C(O)—, —(CR c R c ) m —, —OC(O)—, —(CR c R c ) m —OC(O)—, —(CR c R c ) m —C(O—, NR b C (S)—, or —NR b C(O)— (wherein the point of the attachment to R 1 is on the left-hand side);
- R 1 is selected from alkyl, cycloalkyl, aryl, heteroaryl, and heterocyclyl; each of which is substituted with 0-5 occurrences of R d ;
- each R 3 is independently selected from halo, haloalkyl, alkyl, hydroxyl and —OR a , or two adjacent R 3 taken together with the carbon atoms to which they are attached form an optionally substituted heterocyclyl; each R a is independently selected from alkyl, acyl, hydroxyalkyl and haloalkyl;
- each R b is independently selected from hydrogen and alkyl
- each R c is independently selected from hydrogen, halo, alkyl, alkoxy and halo alkoxy or two R c taken together with the carbon atoms to which they are attached form an optionally substituted cycloalkyl;
- each R d is independently selected from halo, haloalkyl, haloalkoxy, alkyl, alkynyl, nitro, cyano, hydroxyl, —C(O)R a , —OC(O)R a , —C(O)OR a , —SR a , NR a R b and —OR a , or two R d taken together with the carbon atoms to which they are attached form an optionally substituted heterocyclyl;
- n 0, 1, or 2;
- n 1, 2 or 3;
- h 0, 1, 2;
- g 0, 1 or 2.
- a method for treating or preventing a disease, condition or disorder as described (e.g., treating) herein comprising administering a compound provided herein, a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof.
- a method of modulating e.g., increasing or decreasing the level of PKM2 activity and/or glycolysis (e.g., modulating the endogenous ability of a cell in the patient to down regulate PKM2) in a patient in need thereof.
- the method comprises the step of administering an effective amount of a compound described herein to the patient in need thereof, thereby modulating (e.g., increasing or decreasing) the level of PKM2 activity and/or glycolysis in the patient.
- a compound or a composition described herein is used to maintain PKM2 in its active conformation or activate pyruvate kinase activity in proliferating cells as a means to divert glucose metabolites into catabolic rather than anabolic processes in the patient.
- a method of inhibiting cell proliferation in a patient in need thereof comprises the step of administering an effective amount of a compound described herein to the patient in need thereof, thereby inhibiting cell proliferation in the patient.
- this method can inhibiting growth of a transformed cell, e.g., a cancer cell, or generally inhibiting growth in a PKM2-dependent cell that undergoes aerobic glycolysis.
- a method of treating a patient suffering from or susceptible to a disease or disorder associated with the function of PKM2 in a patient in need thereof comprises the step of administering an effective amount of a compound described herein to the patient in need thereof, thereby treating, preventing or ameliorating the disease or disorder in the patient.
- the modulator is provided in a pharmaceutical composition.
- the method includes identifying or selecting a patient who would benefit from modulation (e.g., activation) of PKM2.
- the patient can be identified on the basis of the level of PKM2 activity in a cell of the patient for treatment of cancer associated with PKM2 function.
- the selected patient is a patient suffering from or susceptible to a disorder or disease identified herein, e.g., a disorder characterized by unwanted cell growth or proliferation, e.g., cancer, obesity, diabetes, atherosclerosis, restenosis, and autoimmune diseases.
- a disorder or disease identified herein e.g., a disorder characterized by unwanted cell growth or proliferation, e.g., cancer, obesity, diabetes, atherosclerosis, restenosis, and autoimmune diseases.
- the compound described herein is administered at a dosage and frequency sufficient to increase lactate production or oxidative phosphorylation.
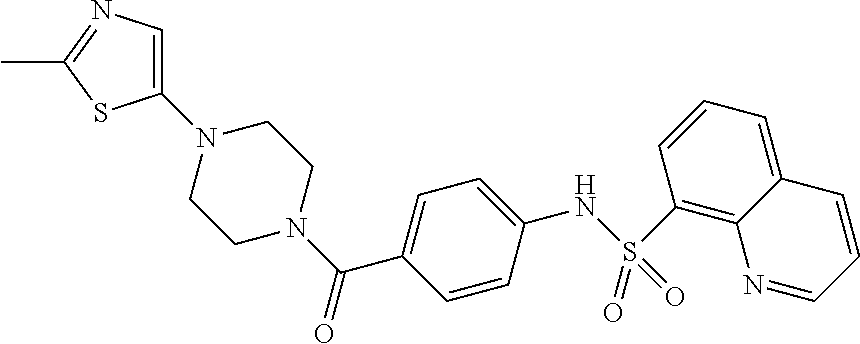
- FIGA. 1 A- 1 A- 44 represent a table of exemplary compounds and the corresponding activity of the compound.
- Compounds that modulate PKM2, e.g., activate PKM2, can be used to treat disorders such as neoplastic disorders (e.g., cancer) or fat related disorders (e.g., obesity).
- a compound of formula (I) or a pharmaceutically acceptable salt thereof or a pharmaceutical composition comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof:
- W, X, Y and Z are each independently selected from CH or N;
- D and D 1 are independently selected from a bond or NR b ;
- A is optionally substituted aryl or optionally substituted heteroaryl
- L is a bond, —C(O)—, —(CR c R c ) m —, —OC(O)—, —(CR c R c ) m —OC(O)—, —(CR c R c ) m —C(O)—, —NR b C (S)—, or —NR b C(O)— (wherein the point of the attachment to R 1 is on the left-hand side);
- R 1 is selected from alkyl, cyclalkyl, aryl, heteroaryl, and heterocyclyl; each of which is substituted with 0-5 occurrences of R d ;
- each R 3 is independently selected from halo, haloalkyl, alkyl, hydroxyl and —OR a , or two adjacent R 3 taken together with the carbon atoms to which they are attached form an optionally substituted heterocyclyl;
- each R a is independently selected from alkyl, acyl, hydroxyalkyl and haloalkyl;
- each R b is independently selected from hydrogen and alkyl
- each R c is independently selected from hydrogen, halo, alkyl, alkoxy and halo alkoxy or two R c taken together with the carbon atoms to which they are attached form an optionally substituted cycloalkyl;
- each R d is independently selected from halo, haloalkyl, haloalkoxy, alkyl, alkynyl, nitro, cyano, hydroxyl, —C(O)R a , —OC(O)R a , —C(O)OR a , —SR a , —NR a R b and —OR a , or two R d taken together with the carbon atoms to which they are attached form an optionally substituted heterocyclyl;
- n 0, 1, or 2;
- n 1, 2 or 3;
- h 0, 1, 2;
- g 0, 1 or 2.
- W, X, Y and Z are each independently selected from CH or N;
- D and D 1 are independently selected from a bond or NR b ;
- A is optionally substituted bicyclic heteroaryl
- L is a bond, —C(O)—, —(CR c R c ) m —, —OC(O)—, —(CR c R c ) m —OC(O)—, —(CR c R c ) m —C(O)—, —NR b C (S)—, or —NR b C(O);
- R 1 is selected from alkyl, cycloalkyl, aryl, heteroaryl, and heterocyclyl; each of which is substituted with 0-5 occurrences of R d ;
- each R 3 is independently selected from halo, haloalkyl, alkyl, hydroxyl and —OR a or two adjacent R 3 taken together with the carbon atoms to which they are attached form an optionally substituted cyclyl; each R a is independently selected from alkyl, acyl, hydroxyalkyl and haloalkyl;
- each R b is independently selected from hydrogen and alkyl
- each R c is independently selected from hydrogen, halo, alkyl, alkoxy and halo alkoxy or two R c taken together with the carbon atoms to which they are attached form an optionally substituted cycloalkyl;
- each R d is independently selected from halo, haloalkyl, haloalkoxy, alkyl, alkynyl, nitro, cyano, hydroxyl, —C(O)R a , —OC(O)R a , —C(O)OR a , —SR a , NR a R b and —OR a , or two R d taken together with the carbon atoms to which they are attached form an optionally substituted heterocyclyl;
- n 0, 1, or 2;
- n 1, 2 or 3;
- h 0, 1, 2;
- g is 0, 1 or 2. In some embodiments, h is 1. In some embodiments, h is 2.
- g is 1. In some embodiments, g is 2.
- both h and g are 1. In some embodiments, h is 1 and g is 2. In some embodiments, g is 1 and h is 2.
- W, X, Y and Z are CH. In some embodiments, at least one of W, X, Y and Z is N. In some embodiments, at least two of W, X, Y and Z are N. In some embodiments, at least three of W, X, Y and Z are N.
- W, X, Y, Z and the carbons to which they are attached form a pyridyl ring. In some embodiments, W, X, Y, Z and the carbon atoms to which they are attached form a pyrimidyl ring. In some embodiments, W, X, Y, Z and the carbon atoms to which they are attached form a pyridazinyl ring.
- W, X and Y are CH and Z is N.
- X, Y and Z are CH and W is N.
- D is NR b and D 1 is a bond. In some embodiments, D is a bond and D 1 is NR b . In some embodiments, both D and D 1 are NR b .
- R b is alkyl (e.g., methyl or ethyl). In some embodiments, R b is hydrogen (H).
- A is a 9-10 membered bicyclic heteroaryl (e.g., quinazolinyl, quinoxalinyl, cinnolinyl, isoquinolyl, indolyl, benzoxazolyl, pyrrolopyridyl, pyrrolopyrimidyl, benzimidazolyl, benzthiazolyl, or benzoxazolyl).
- A is a N-containing 9-10 membered bicyclic heteroaryl.
- A is optionally substituted quinazolinyl (e.g., 8-quinazolinyl or 4-quinazolinyl), optionally substituted quinoxalinyl (e.g., 5-quinoxalinyl), optionally substituted quinolinyl (e.g., 4-quinolinyl or 8-quinolinyl), optionally substituted cinnolinyl (e.g., 8-cinnolinyl), optionally substituted isoquinolinyl, optionally substituted indolyl (7-indolyl), optionally substituted benzoxazolyl (e.g., 7-benzoxazolyl), optionally substituted pyrrolopyridyl (e.g., 4-pyrrolopyridyl), optionally substituted pyrrolopyrimidyl (e.g., 4-pyrrolopyrimidyl), optionally substituted benzimidazolyl (e.g., 7-benzimid,
- A is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-phenyl
- A is optionally substituted
- A is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-phenyl
- L is a bond
- L is —(CR c R c ) m — and m is 1.
- each R c is hydrogen.
- one R c is alkyl (e.g., methyl or ethyl) and the other R c is hydrogen.
- one R c is halo (e.g., fluoro) and one R c is hydrogen.
- both R c are halo (e.g., fluoro).
- one R c is alkoxy (e.g., methoxy or ethoxy) and one R c is hydrogen.
- both R c are alkoxy (e.g., methoxy or ethoxy). In some aspects of these embodiments, two R c taken together with the carbon to which they are attached form a cycloalkyl (e.g., cyclopropyl).
- L is —(CR c R c ) m — and m is 2.
- each R c is hydrogen.
- 1R c is alkyl (e.g., methyl or ethyl) and each of the other R c are hydrogen.
- two R c s taken together with the carbon to which they are attached form a cycloalkyl (e.g., cyclopropyl) and each of the other two R c s are hydrogen.
- L is —(CR c R c ) m — and m is 3. In some aspects of these embodiments each R c is hydrogen.
- L is —C(O)—.
- L is —O—(O)—.
- L is NR b C(O)— and R b is H. In some embodiments, L is NR b C(S)— and R b is H.
- L is —(CR c R c ) m —C(O)— and m is 1.
- each R c is hydrogen.
- one R c is alkyl (e.g., methyl or ethyl) and one R c is hydrogen.
- both R c are alkyl (e.g., methyl or ethyl).
- L is —(CR c R c ) m —C(O)— and m is 2. In some aspects of these embodiments, each R c is hydrogen.
- L is (—(CR c R c ) m —C(O)— and m is 3. In some aspects of these embodiments, each R c is hydrogen.
- R 1 is alkyl substituted with 0-5 occurrences of R d (e.g., methyl, ethyl, n-propyl, i-propyl, or n-butyl). In some embodiments, R 1 is methyl, ethyl, n-propyl, i-propyl, or n-butyl. In some embodiments, R 1 is ethyl or propyl (n-propyl or i-propyl). In some aspects of these embodiments, L is a bond, —CH 2 —, —C(O)—, or —O(CO)—. In some aspects of these embodiments, L is —O(CO)—.
- R 1 is alkyl substituted with 1 occurrence of R d (e.g., methyl, ethyl, n-propyl, i-propyl, or n-butyl). In some embodiments, R 1 is methyl, ethyl, or n-propyl substituted with 1 occurrence of R d . In some aspects of these embodiments, R d is halo (e.g., fluorine or chlorine). In some aspects of these embodiments, R d is —C(O)OR a . In some aspects of these embodiments, R a is alkyl (e.g., methyl or ethyl). In some aspects of these embodiments, L is —NHC (O)—.
- R 1 is alkyl substituted with 2 occurrences of R d (e.g., methyl, ethyl, n-propyl, i-propyl, or n-butyl). In some embodiments, R 1 is methyl, ethyl, or n-propyl substituted with 2 occurrences of R d . In some embodiments, R 1 is n-propyl substituted with 2 occurrences of R d . In some aspects of these embodiments, 1R d is cyano and the other R d is —NR a R b . In some aspects of these embodiments, R a and R b are hydrogen. In some aspects of these embodiments, L is —CH 2 —.
- R 1 is heteroaryl substituted with 0-5 occurrences of R d (e.g., S-containing monocyclic heteroaryl, N-containing monocyclic heteroaryl or N-containing bicyclic heteroaryl). In some embodiments, R 1 is a 5-8 membered monocyclic heteroaryl substituted with 0-5 occurrences of R d (e.g., thiophenyl, pyridyl, pyrimidyl or pyrazyl).
- R 1 is pyridyl substituted with 0-5 occurrences of R d (e.g., 2-pyridyl, 3-pyridyl or 4-pyridyl), pyrimidyl substituted with 0-5 occurrences of R d (e.g., 2-pyrimidyl or 5-pyrimidyl) or pyrazinyl substituted with 0-5 occurrences of R d (e.g., 2-pyrazinyl).
- R 1 is thiazolyl substituted with 0-5 occurrences of R d (e.g., 2-thiazolyl or 5-thiazolyl).
- R 1 is pyrimidyl substituted with 0-5 occurrences of R d (e.g., 2-pyrimidyl).
- R 1 is thiadiazolyl substituted with 0-5 occurrences of R d (e.g., 4-thiadiazolyl).
- R 1 is pyrrolyl substituted with 0-5 occurrences of R d (e.g., 2-pyrrolyl).
- L is a bond, —CH 2 —, —C(O)—, or —O(CO)—.
- R 1 is pyridyl (e.g., 2-pyridyl, 3-pyridyl or 4-pyridyl).
- R 1 is pyridyl (e.g., 2-pyridyl, 3-pyridyl or 4-pyridyl) substituted with 1 occurrence of R d .
- R d is —OC(O)R a .
- R d is —OR a .
- R d is —C(O)OR a .
- R d is alkyl (e.g., methyl or ethyl).
- R d is haloalkyl (e.g., trifluoromethyl).
- R d is halo (e.g., fluorine or chlorine).
- R a is alkyl (e.g., methyl or ethyl).
- L is —CH 2 —.
- R 1 is pyridyl (e.g., 2-pyridyl, 3-pyridyl or 4-pyridyl) substituted with 2 occurrences of R d .
- one R d is —C(O)OR a and the other R d is —OR a .
- R a is alkyl (e.g., methyl or ethyl).
- both R d are halo (e.g., fluoro or chloro).
- L is —CH 2— .
- R 1 is pyrimidyl (e.g., 2-pyrimidyl or 5-pyrimidyl).
- L is a bond.
- R 1 is pyrimidyl (e.g., 2-pyrimidyl or 5-pyrimidyl) substituted with 1 occurrence of R d .
- R d is halo (e.g., fluoro or chloro).
- R 1 is pyrazinyl (e.g., 2-pyrazinyl).
- L is a bond.
- R 1 is thiazolyl (e.g., 2-thiazolyl, 4-thiazolyl, or 5-thiazolyl).
- L is —C(O)—.
- R 1 is thiazolyl (e.g., 2-thiazolyl, 4-thiazolyl, or 5-thiazolyl) substituted with 1 occurrences of R d .
- R d is alkyl (e.g, methyl or ethyl).
- L is —C(O)—.
- R 1 is thiophenyl substituted with 0-5 occurrences of R d (e.g., 2-thiophenyl). In some embodiments, R 1 is thiophenyl.
- R 1 is thiadiazolyl (e.g., 4-thiadiazolyl).
- R 1 is pyrrolyl (e.g., 2-pyrrolyl).
- R 1 is cycloalkyl substituted with 0-5 occurrences of R d (e.g., cyclopropyl, cyclopentyl or cyclohexyl). In some embodiments, R 1 is cyclopropyl. In some embodiments, R 1 is cyclohexyl. In some embodiments, R 1 is cyclopentyl. In some aspect of these embodiments, L is —CH 2 —C(O)—. In some embodiment, R 1 is aryl substituted with 0-5 occurrences of R d . In some aspects of these embodiments, L is a bond, —CH 2 —, —C(O)—, or —O(CO)—.
- R 1 is aryl (e.g., phenyl). In some embodiments, R 1 is phenyl. In some aspects of these embodiments, L is a bond, —CH 2 —, —C(O)—, or —O(CO)—.
- R 1 is phenyl substituted with 1 occurrence of R d .
- R d is ortho substituted.
- R d is meta substituted.
- R d is para substituted.
- R d is halo (e.g., fluorine, bromine or chlorine).
- R d is alkyl (e.g., methyl, ethyl, isopropyl, t-butyl, n-butyl or n-pentyl).
- R d is haloalkyl (e.g., trifluoromethyl).
- R d is —OR a . In some aspects of these embodiments, R d is —C(O)R a . In some aspects of these embodiments, R d is —SR a . In some aspects of these embodiments, R d is —C(O)OR a . In some aspects of these embodiments, R d is cyano. In some aspects of these embodiments, R d is —NR a R b . In some aspects of these embodiments, R d is haloalkoxy (e.g., difluoromethoxy or trifluoromethoxy). In some aspects of these embodiments, R d is hydroxyl.
- R d is —OC (O)R a .
- R d is alkynyl (e.g., 1-hexynyl).
- R d is haloalkyl (e.g., trifluoromethyl).
- R a is alkyl (e.g., methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, t-butyl or n-pentyl).
- R a is hydroxyalkyl (e.g., 2-hydroxylethyl).
- R a and R b are alkyl (e.g., methyl or ethyl).
- R a is acyl (e.g., acetyl) and R b is hydrogen.
- L is a bond, —CH 2 —, —C(O)—, or —O(CO)—.
- R 1 is phenyl substituted with 2 occurrences of R d .
- both R d are halo (e.g., fluorine or chlorine).
- both R d are alkyl (e.g., methyl or ethyl).
- 1R d is alkyl (e.g., methyl or ethyl) and the other is —OR a .
- 1 R d is halo (e.g., fluorine or chlorine) and the other R d is —OR a .
- both R d are —OR a .
- 1 R d is halo (e.g., fluorine or chlorine) and the other R d is hydroxyl. In some aspects of these embodiments, 1 R d is halo (e.g., fluorine or chlorine) and the other is haloalkyl (e.g., trifluoromethyl). In some aspects of these embodiments, 1 R d is —OR a and the other R d is —C(O)OR a . In some aspects of these embodiments, 1 R d is —OR a and the other R d is hydroxyl.
- 1 R d is alkyl (e.g., methyl or ethyl) and the other R d is hydroxyl. In some aspects of these embodiments, both R d are hydroxyl. In some aspects of these embodiments, 1 R d is halo (e.g., fluorine) and the other R d is haloalkyl (e.g., trifluoromethyl). In some aspects of these embodiments, both R d are hydroxyl. In some aspects of these embodiments, one R d is haloalkyl (e.g., trifluoromethyl) and the other R d is alkyl (e.g., methyl).
- two R d together with the carbon atoms to which they are attached, form an optionally substituted heterocyclyl. In some aspects of these embodiments, two R d , together with the carbon atoms to which they are attached, form an optionally substituted 5-7 membered heterocyclyl. In some aspects of these embodiments, two R d , together with the phenyl ring to which they are attached, form the following structure:
- R a is alkyl (e.g., methyl or ethyl).
- L is a bond, —CH 2 —, —C(O)—, or —O(CO)—.
- R 1 is phenyl substituted with 3 occurrences of R d .
- 3 R d are halo (e.g., fluorine or chlorine).
- 2 R d are halo (e.g., fluorine or chlorine) and 1 R d is hydroxyl.
- 1 R d is halo (e.g., fluorine or chlorine), 1 R d is alkyl (e.g., methyl) and 1 R d is hydroxyl.
- 3 R d are alkyl (e.g., methyl or ethyl).
- 2 R d are alkyl (e.g., methyl or ethyl) and 1 R d is hydroxyl. In some aspects of these embodiments, 2 R d are halo (e.g., fluorine or chlorine) and 1 R d is —OR a . In some aspects of these embodiments, R a is alkyl (e.g., methyl or ethyl). In some aspects of these embodiments, 1 R d is hydroxyl and 2 R d are —OR a . In some aspects of these embodiments, R a is alkyl (e.g., methyl or ethyl). In some aspects of these embodiments, 3 R d are —OR a .
- R d are halo (e.g., fluorine or chlorine).
- R a is alkyl (e.g., methyl or ethyl).
- L is a bond, —CH 2 —, —C(O)—, or —O(CO)—.
- R 1 is phenyl substituted with 4 occurrences of R d .
- 1 R d is jhydroxyl
- 1 R d is alkyl (e.g., methyl or ethyl)
- 2 R d are —OR a .
- R a is alkyl (e.g., methyl or ethyl).
- L is a bond, —CH 2 —, —C(O)—, or —O(CO)—.
- R 1 is heterocyclyl substituted with 0-5 occurrences of R d .
- R 1 is tetrahydrofuranyl substituted with 0-5 occurrences of R d (e.g., 2-tetrahydrofuranyl or 3-tetrahydrofuranyl). In some aspects of these embodiments, R 1 is tetrahydrofuranyl (e.g., 2-tetrahydrofuranyl or 3-tetrahydrofuranyl). In some aspects of these embodiments, L is —C(O)—.
- R 1 is azetidinyl substituted with 0-5 occurrences of R d (e.g., 3-azetidinyl). In some embodiments, R 1 is azetidinyl (e.g., 3-azetidinyl). In some embodiments, R 1 is azetidinyl (e.g., 3-azetidinyl) substituted with 1 occurrence of R d . In some aspects of these embodiments, R d is alkyl (e.g., methyl or ethyl). In some aspects of these embodiments, L is —C(O)—.
- R 1 is 10-14 membered bicyclic aryl substituted with 0-5 occurrences of R d .
- R d is naphthyl substituted with 0-5 occurrences of R d .
- R d is naphthyl.
- L is a bond, —(CR c R c ) m —, —NR b C(O)—, —(CR c R c ) m —C(O)—, —C(O)—, or —O(CO)—.
- L is a bond and R 1 is alkyl, aryl or heteroaryl substituted with 0-5 occurrences of R d .
- alkyl, aryl or heteroaryl of R 1 is as described in any one of the embodiments and aspects above.
- L is —(CR c R c ) m — and R 1 is cycloalkyl, aryl, heteroaryl or heterocyclyl substituted with 0-5 occurrences of R d .
- R 1 is cycloalkyl, aryl, heteroaryl or heterocyclyl of R 1 is as described in any one of the embodiments and aspects above.
- L is —NR b C(O)— and R b is hydrogen; and R 1 is aryl substituted with 0-5 occurrences of R d .
- aryl of R 1 is as described in any one of the embodiments and aspects above.
- L is —(CR c R c ) m —C(O)— and R 1 is cycloalkyl, aryl or heteroaryl substituted with 0-5 occurrences of R d .
- cycloalkyl, aryl, or heteroaryl of R 1 is as described in any one of the embodiments and aspects above.
- L is —C(O)— and R 1 is aryl, alkyl, or heteroaryl substituted with 0-5 occurrences of R d .
- R 1 is aryl, alkyl, or heteroaryl substituted with 0-5 occurrences of R d .
- aryl, alkyl, or heteroaryl of R 1 is as described in any one of the embodiments and aspects above.
- L is —OC(O)— and R 1 is alkyl, aryl or heterocyclyl substituted with 0-5 occurrences of R d .
- alkyl, aryl, or heterocyclyl of R 1 is as described in any one of the embodiments and aspects above.
- L is —(CR c R c ) m —OC(O)— and R 1 is heterocyclyl or cycloalkyl substituted with 0-5 occurrences of R d .
- heterocyclyl or cycloalkyl of R 1 is as described in any one of the embodiments and aspects above.
- n is 0. In some embodiments, n is 1.
- R 3 is alkyl (e.g., methyl or ethyl). In some embodiments, R 3 is —OR a . In some aspects of these embodiments, R a is alkyl (e.g., methyl or ethyl). In some embodiments, R 3 is halo (e.g., fluorine or chlorine). In some embodiments, R 3 is hydroxyl. In some embodiments, R 3 is haloalkyl (e.g., trifluoromethyl).
- n is 2.
- two adjacent R 3 taken together with the carbon atoms to which they are attached form a heterocyclyl ring. In some embodiments, both R 3 are —OR a . In some embodiments, two adjacent R 3 taken together with the carbon atoms to which they are attached form
- a compound is of formula (II) or a pharmaceutical acceptable salt thereof:
- L, R 1 , R 3 , R a , R b , R c , R d , Y, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- A is aryl (e.g., phenyl or naphthyl) optionally substituted with 1 or 2 occurrences of R 2 , wherein each R 2 is independently selected from halo, haloalkyl, aryl, heteroaryl, alkyl, —OR a , —COOR c , or —CONR c R c ; and D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- R 2 is independently selected from halo, haloalkyl, aryl, heteroaryl, alkyl, —OR a , —COOR c , or —CONR c R c ; and D, D 1 , L, R 1 , R 3 , R
- D and D 1 are N. In some aspect of these embodiments, at least one of W, X, Y and Z is N. In some aspect of these embodiments, one of W, Y and Z is N; his 1 and g is 1.
- A is heteroaryl (e.g., N-containing monocyclic heteroaryl or N-containing bicyclic heteroaryl); and D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- A is a 5-8 membered monocyclic heteroaryl (e.g., pyridyl, pyrimidyl, or pyrazyl); and D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- pyridyl pyrimidyl, or pyrazyl
- D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- A is a 5-8 membered N-containing monocyclic heteroaryl; and D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- A is optionally substituted pyridyl (e.g., 2-pyridyl, 3-pyridyl or 4-pyridyl), optionally substituted pyrimidyl (e.g., 2-pyrimidyl or 5-pyrimidyl), or optionally substituted pyrazyl (e.g., 2-pyrazyl); and L, R 1 , R 3 , R a , R b , R c , R d , Y, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- pyridyl e.g., 2-pyridyl, 3-pyridyl or 4-pyridyl
- optionally substituted pyrimidyl e.g., 2-pyrimidyl or 5-pyrimidyl
- optionally substituted pyrazyl e.g., 2-pyrazyl
- A is substituted with 1 occurrence of R 2 ; and D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- R 2 is alkyl (e.g., methyl or ethyl).
- R 2 is halo.
- R 2 is fluorine (F).
- R 2 is bromine (Br).
- R 2 is chlorine (Cl).
- R 2 is —OR a .
- R a is alkyl (e.g., methyl).
- A is substituted with 2 occurrences of R 2 ; and D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- both R 2 are halo (e.g., fluorine or fluorine and chlorine).
- both R 2 are alkyl (e.g, methyl).
- both R 2 are —OR a .
- one R 2 is halo and the other is —OR a .
- one R 2 is bromine (BR) and the other is —OR a .
- one R 2 is chlorine (Cl) and the other is —OR a .
- one R 2 is fluorine (F) and the other is —OR a .
- R a is alkyl (e.g., methyl or ethyl).
- both R 2 are —OR a .
- two —OR a taken together with the carbon atoms to which they are attached form a heterocyclyl.
- A is
- D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- a compound of formula (I) or a pharmaceutically acceptable salt thereof or a pharmaceutical composition comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof:
- W, X, Y and Z are each independently selected from CH or N;
- D and D 1 are independently selected from a bond or NR b ;
- A is optionally substituted aryl or optionally substituted heteroaryl
- L is a bond, —C(O)—, —(CR c R c ) m —, —OC(O)—, of —C(O)NR b —;
- R 1 is independently selected from alkyl, cycloalkyl, aryl, heteroaryl, and heterocyclyl; each of which are substituted with 0-3 occurrences of R d ;
- each R 3 is independently selected from halo, haloalkyl, alkyl, hydroxyl and —OR a or two adjacent R 3 taken together with the carbon atoms to which they are attached form an optionally substituted cyclyl;
- each R a is independently selected from alkyl and haloalkyl
- each R b is independently selected from hydrogen and alkyl
- each R c is independently selected from hydrogen, halo, alkyl, alkoxy and halo alkoxy or two R c taken together with the carbon atoms to which they are attached form an optionally substituted cycloalkyl;
- each R d is independently selected from halo, haloalkyl, alkyl, nitro, cyano and —OR a , or two R d taken together with the carbon atoms to which they are attached form an optionally substituted heterocyclyl;
- n 0, 1, or 2;
- n 1, 2 or 3;
- h 0, 1, 2;
- A, D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined in any one of the embodiments or aspects described herein.
- a compound of formula (I) or a pharmaceutically acceptable salt thereof or a pharmaceutical composition comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof:
- W, X, Y and Z are each independently selected from CH or N;
- D and D 1 are independently selected from a bond or NR c ;
- A is optionally substituted aryl or optionally substituted heteroaryl
- R 1 is independently selected from alkyl, optionally substituted aryl, and optionally substituted heteroaryl;
- each R 3 is independently selected from halo, haloalkyl, alkyl, and —OR a ;
- each R a is independently selected from alkyl, haloalkyl and optionally substituted heteroaryl;
- each R b is independently alkyl
- each R c is independently selected from hydrogen or alkyl
- n 0, 1, or 2;
- A, D, D 1 , L, R 1 , R 3 , R a , R b , R c , R d , X, Y, Z, W, n, m, h and g are as defined in any one of the embodiments or aspects described herein.
- a compound or pharmaceutically acceptable salt of formula (Ib) or a pharmaceutical composition comprising a compound or pharmaceutically acceptable salt of formula (Ib):
- A, L, R 1 , R 3 , R a , R b , R c , R d , W, X, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- X, W an d Z are CH. In some embodiments, one of X, W and Z is N and the other two of X, W and Z are CH.
- a pharmaceutical composition comprising a compound or pharmaceutically acceptable salt of formula (Ic) or a pharmaceutical composition comprising a compound or pharmaceutically acceptable salt of formula (Ic):
- A, L, R 1 , R 3 , R a , R b , R c , R d , W, X, Y, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- X, Y and W are CH. In some embodiments, one o f X, Y and W is N and the other two of X, Y and W are CH.
- a compound or pharmaceutically acceptable salt of formula (Id) or a pharmaceutical composition comprising a compound or pharmaceutically acceptable salt of formula (Id):
- A, L, R 1 , R 3 , R a , R b , R c , R d , Y, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- Y and Z are CH. In some embodiments, one of Y and Z is N and one of Y and Z is CH.
- a compound or pharmaceutically acceptable salt of formula (Ie) or a pharmaceutical composition comprising a compound or pharmaceutically acceptable salt of formula (Ie):
- A, L, R 1 , R 3 , R a , R b , R c , R d , W, X, Y, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
- exemplary compounds of Formula I include the compounds described in FIGS. 1 A- 1 A- 44 and in the Examples.
- a compound described herein modulates PKM2 by interacting (e.g., binding) with the FBP binding pocket.
- a compound described herein can compete with FBP binding in PKM2.
- a compound described herein has one or more properties described herein, e.g., one or more of the following properties: it is an allosteric modulator (e.g., activator); it modulates the release of FBP (e.g., promotes); it is a modulator (e.g., agonist) of FBP, e.g., an agonist which binds with a lower, about the same, or higher affinity than does FBP; it modulates (e.g., promotes) the dissolution of tetrameric PKM2; it modulates (e.g., promotes) the assembly of tetrameric PKM2; it selectively modulates (e.g., activates) PKM2 over at least one other isoform of PK, e.g., it is selective for PKM2 over PKR, PKM1, or PKL; is has an affinity for PKM2 which is greater than its affinity for at least one other isoform of PK, e.g., PK
- the activator of PKM2 utilized in the methods and compositions described herein operates by or has one or more of the following mechanisms or properties:
- Scheme 1 above is an exemplary scheme that depicts a representative synthesis of certain copounds described herein.
- Sulfonyl chloride 1 is reacted with amine 1 under standard coupling conditions to produce ester 3. Hydrolysis of 3 using lithium hydroxide generates carboxylic acid 4.
- Piperazine (5) is with the appropriate bromide under standard palladium coupling conditions to provide 7.
- Carboxylic acid 4 is then treated with piperazine derivative 7 to produce final compound 8.
- the compounds provided herein may contain one or more asymmetric centers and thus occur as racemates and racemic mixtures, single enantiomers, individual diastereomers and diastereomeric mixtures. All such isomeric forms of these compounds are expressly included within the scope. Unless otherwise indicated when a compound is named or depicted by a structure without specifying the stereochemistry and has one or more chiral centers, it is understood to represent all possible stereoisomers of the compound.
- the compounds provided herewith may also contain linkages (e.g., carboncarbon bonds) or substituents that can restrict bond rotation, e.g. restriction resulting from the presence of a ring or double bond. Accordingly, all cis/trans and E/Z isomers are expressly included.
- the compounds provided herein may also comprise one or more isotopic substitutions.
- H may be in any isotopic form, including 1 H, 2 H (D or deuterium), and 3 H (T or tritium);
- C may be in any isotopic form, including 12 C, 13 C, and 14 C;
- O may be in any isotopic form, including 16 O and 18 O; and the like.
- the compounds provided herein may also be represented in multiple tautomeric forms, in such instances, expressly includes all tauto- meric forms of the compounds described herein, even though only a single tautomeric form may be represented (e.g., alkylation of a ring system may result in alkylation at multiple sites; all such reaction products are expressly included). All such isomeric forms of such compounds are expressly included. All crystal forms of the compounds described herein are expressly included.
- a salt for example, can be formed between an anion and a positively charged substituent (e.g., amino) on a compound described herein. Suitable anions include chloride, bromide, iodide, sulfate, nitrate, phosphate, citrate, methanesulfonate, trifluoroacetate, and acetate. Likewise, a salt can also be formed between a cation and a negatively charged substituent (e.g., carboxylate) on a compound described herein.
- a positively charged substituent e.g., amino
- Suitable anions include chloride, bromide, iodide, sulfate, nitrate, phosphate, citrate, methanesulfonate, trifluoroacetate, and acetate.
- a salt can also be formed between a cation and a negatively charged substituent (e.g., carboxylate) on a compound described herein.
- Suitable cations include sodium ion, potassium ion, magnesium ion, calcium ion, and an ammonium cation such as tetramethylammonium ion.
- Examples of prodrugs include esters and other pharmaceutically acceptable derivatives, which, upon administration to a subject, are capable of providing active compounds.
- the compounds provided herein may be modified by appending appropriate functionalities to enhance selected biological properties, e.g., targeting to a particular tissue.
- modifications are known in the art and include those which increase biological penetration into a given biological compartment (e.g., blood, lymphatic system, central nervous system), increase oral availability, increase solubility to allow administration by injection, alter metabolism and alter rate of excretion.
- the compounds described herein may be used as platforms or scaffolds that may be utilized in combinatorial chemistry techniques for preparation of derivatives and/or chemical libraries of compounds.
- Such derivatives and libraries of compounds have biological activity and are useful for identifying and designing compounds possessing a particular activity.
- Combinatorial techniques suitable for utilizing the compounds described herein are known in the art as exemplified by Obrecht, D. and Villalgrodo, J.
- one embodiment relates to a method of using the compounds described herein for generating derivatives or chemical libraries comprising: 1) providing a body comprising a plurality of wells; 2) providing one or more compounds identified by methods described herein in each well; 3) providing an additional one or more chemicals in each well; 4) isolating the resulting one or more products from each well.
- An alternate embodiment relates to a method of using the compounds described herein for generating derivatives or chemical libraries comprising: 1) providing one or more compounds described herein attached to a solid support; 2) treating the one or more compounds identified by methods described herein attached to a solid support with one or more additional chemicals; 3) isolating the resulting one or more products from the solid support.
- tags or identifier or labeling moieties may be attached to and/or detached from the compounds described herein or their derivatives, to facilitate tracking, identification or isolation of the desired products or their intermediates.
- moieties are known in the art.
- the chemicals used in the aforementioned methods may include, for example, solvents, reagents, catalysts, protecting group and deprotecting group reagents and the like. Examples of such chemicals are those that appear in the various synthetic and protecting group chemistry texts and treatises referenced herein.
- halo or halogen refers to any radical of fluorine, chlorine, bromine or iodine.
- alkyl refers to a hydrocarbon chain that may be a straight chain or branched chain, containing the indicated number of carbon atoms. For example, C 1 -C 12 alkyl indicates that the group may have from 1 to 12 (inclusive) carbon atoms in it.
- haloalkyl refers to an alkyl in which one or more hydrogen atoms are replaced by halo, and includes alkyl moieties in which all hydrogens have been replaced by halo (e.g., perfluoroalkyl).
- arylalkyl or “aralkyl” refer to an alkyl moiety in which an alkyl hydrogen atom is replaced by an aryl group.
- Aralkyl includes groups in which more than one hydrogen atom has been replaced by an aryl group.
- arylalkyl or “aralkyl” include benzyl, 2-phenylethyl, 3-phenylpropyl, 9-fluorenyl, benzhydryl, and trityl groups.
- alkylene refers to a divalent alkyl, e.g., —CH 2 —, —CH 2 CH 2 —, and —CH 2 CH 2 CH 2 —.
- alkenyl refers to a straight or branched hydrocarbon chain containing 2-12 carbon atoms and having one or more double bonds.
- alkenyl groups include, but are not limited to, allyl, propenyl, 2-butenyl, 3-hexenyl and 3-octenyl groups.
- One of the double bond carbons may optionally be the point of attachment of the alkenyl substituent.
- alkynyl refers to a straight or branched hydrocarbon chain containing 2-12 carbon atoms and characterized in having one or more triple bonds. Examples of alkynyl groups include, but are not limited to, ethynyl, propargyl, and 3-hexynyl.
- One of the triple bond carbons may optionally be the point of attachment of the alkynyl substituent.
- alkylamino and dialkylamino refer to —NH (alkyl) and —NH(alkyl) 2 radicals respectively.
- aralkylamino refers to a —NH(aralkyl) radical.
- alkylaminoalkyl refers to a (alkyl)NH-alkyl-radical; the term dialkylaminoalkyl refers to a (alkyl) 2 N-alkyl-radical
- alkoxy refers to an —O-alkyl radical.
- mercapto refers to an SH radical.
- thioalkoxy refers to an S-alkyl radical.
- thioaryloxy refers to an —S-aryl radical.
- aryl refers to a monocyclic, bicyclic, or tricyclic aromatic hydrocarbon ring system, wherein any ring atom capable of substitution can be substituted (e.g., by one or more substituents).
- aryl moieties include, but are not limited to, phenyl, naphthyl, and anthracenyl.
- cycloalkyl as employed herein includes cyclic, bicyclic, tricyclic, or polycyclic non-aromatic hydrocarbon groups having 3 to 12 carbons. Any substitutable ring atom can be substituted (e.g., by one or more substituents).
- the cycloalkyl groups can contain fused or spiro rings. Fused rings are rings that share a common carbon atom. Examples of cycloalkyl moieties include, but are not limited to, cyclopropyl, cyclohexyl, methylcyclohexyl, adamantyl, and norbornyl.
- heterocyclyl or “heterocyclic group” refer to 3- to 14-membered non-aromatic ring structures (e.g., 3- to 14-membered rings, more preferably 3- to 7-membered rings), whose ring structures include one to four heteroatoms independently selected from O, N and S.
- the heterocyclyl or heterocyclic groups can contain fused or spiro rings.
- Heterocycles can also be polycycles, with each group having, e.g., 5-7 ring members.
- the term “heterocyclyl” or “heterocyclic group” includes saturated and partially saturated heterocyclyl structures.
- heteroaryl refers to a 5-14 membered (i.e., a 5-8 membered monocyclic, 8-12 membered bicyclic, or 11-14 membered tricyclic) aromatic ring system having 1-3 ring heteroatoms if monocyclic, 1-6 ring heteroatoms if bicyclic, or 1-9 ring heteroatoms if tricyclic, said ring heteroatoms independently selected from O, N, and S (e.g., 1-3, 1-6, or 1-9 ring heteroatoms of N, O or S if monocyclic, bicyclic, or tricyclic, respectively). Any substitutable ring atom can be substituted (e.g., by one or more substituents).
- Heterocyclyl and heteroaryl groups include, for example, thiophene, thianthrene, furan, pyran, isobenzofuran, chromene, xanthene, phenoxathiin, pyrrole, imidazole, pyrazole, isothiazole, isoxazole, pyridine, pyrazine, pyrimidine, pyridazine, indolizine, isoindole, indole, indazole, purine, quinolizine, isoquinoline, quinoline, phthalazine, naphthyridine, quinoxaline, quinazoline, cinnoline, pteridine, carbazole, carboline, phenanthridine, acridine, pyrimidine, phenanthroline, phenazine, phenarsazine, phenothiazine, furazan, phenoxazine, pyrrol
- the heterocyclic or heteroaryl ring can be substituted at one or more positions with such substituents as described herein, as for example, halogen, alkyl, aralkyl, alkenyl, alkynyl, cycloalkyl, hydroxyl, amino, nitro, sulfhydryl, imino, amido, phosphate, phosphonate, phosphinate, carbonyl, carboxyl, silyl, ether, alkylthio, sulfonyl, ketone, aldehyde, ester, a heterocyclyl, an aromatic or heteroaromatic moiety, —CF 3 , —CN, or the like.
- substituents as described herein, as for example, halogen, alkyl, aralkyl, alkenyl, alkynyl, cycloalkyl, hydroxyl, amino, nitro, sulfhydryl, imino, amido, phosphate, phosphon
- heterocyclylalkyl refers to an alkyl group substituted with a heterocycle group.
- cycloalkenyl refers to partially unsaturated, nonaromatic, monocyclic, bicyclic, or tricyclic hydrocarbon groups having 5 to 12 carbons, preferably 5 to 8 carbons.
- the unsaturated carbon may optionally be the point of attachment of the cycloalkenyl substituent. Any substitutable ring atom can be substituted (e.g., by one or more substituents).
- the cycloalkenyl groups can contain fused or spiro rings. Fused rings are rings that share a common carbon atom. Examples of cycloalkenyl moieties include, but are not limited to, cyclohexenyl, cyclohexadienyl, or norbornenyl.
- heterocycloalkenyl refers to a partially saturated, nonaromatic 5-10 membered monocyclic, 8-12 membered bicyclic, or 11-14 membered tricyclic ring system having 1-3 heteroatoms if monocyclic, 1-6 heteroatoms if bicyclic, or 1-9 heteroatoms if tricyclic, said heteroatoms independently selected from O, N, and S (e.g., 1-3, 1-6, or 1-9 ring heteroatoms of N, O, or S if monocyclic, bicyclic, or tricyclic, respectively).
- the unsaturated carbon or the heteroatom may optionally be the point of attachment of the heterocycloalkenyl substituent.
- heterocycloalkenyl groups can contain fused rings. Fused rings are rings that share a common carbon atom. Examples of heterocycloalkenyl include but are not limited to tetrahydropyridyl and dihydropyranyl.
- heteroaryl refers to an alkyl group substituted with a heteroaryl group.
- the ring heteroatoms of the compounds provided herein include N—O, S(O), and S(O) 2 .
- oxo refers to an oxygen atom, which forms a carbonyl when attached to carbon, an N-oxide when attached to nitrogen, and a sulfoxide or sulfone when attached to sulfur.
- acyl refers to an alkylcarbonyl, cycloalkylcarbonyl, arylcarbonyl, heterocyclylcarbonyl, or heteroarylcarbonyl substituent, any of which may be further substituted (e.g., by one or more substituents).
- substituted refers to a group “substituted” on an alkyl, cycloalkyl, alkenyl, alkynyl, heterocyclyl, heterocycloalkenyl, cycloalkenyl, aryl, or heteroaryl group at any substitutable atom of that group. Any substitutable atom can be substituted.
- substituents include, without limitation, alkyl (e.g., C1, C2, C3, C4, C5, C6, C7, C8, C9, C10, C11, C12 straight or branched chain alkyl), cycloalkyl, haloalkyl (e.g., perfluoroalkyl such as CF 3 ), aryl, heteroaryl, aralkyl, heteroaralkyl, heterocyclyl, alkenyl, alkynyl, cycloalkenyl, heterocycloalkenyl, alkoxy, haloalkoxy (e.g., perfluoroalkoxy such as OCF 3 ), halo, hydroxy, carboxy, carboxylate, cyano, nitro, amino, alkyl amino, SO 3 H, sulfate, phosphate, methylenedioxy (—O— CH 2 —O— wherein oxygens are attached to vicinal atoms), ethylenedioxy, o
- selective is meant at least 2-fold, 3-fold, 4-fold, 5-fold, 6-fold, or 10-fold greater modulation (e.g., activation) of PKM2 than PKM1.
- activator means an agent that (measurably) increases the activity of a pyruvate kinase (e.g., PKM2) or causes pyruvate kinase (e.g., PKM2) activity to increase to a level that is greater than PKM2's basal levels of activity.
- the activator may mimic the effect caused by a natural ligand (e.g., FBP).
- the activator effect caused by a compound provided herein may be to the same, or to a greater, or to a lesser extent than the activating effect caused by a natural ligand, but the same type of effect is caused.
- a compound provided herein can be evaluated to determine if it is an activator by measuring either directly or indirectly the activity of the pyruvate kinase when subjected to said compound.
- the activity of a compound provided herein can be measured, for example, against a control substance.
- the activity measured of the test compound is for activation of PKM2.
- the activity of PKM2 can be measured, for example, by monitoring the concentration of a substrate such as ATP or NADH.
- Me, Et, Ph, Tf, Nf, Ts, Ms represent methyl, ethyl, phenyl, trifluoromethanesulfonyl, nonafluorobutanesulfonyl, p-toluenesulfonyl and methanesulfonyl, respectively.
- a more comprehensive list of the abbreviations utilized by organic chemists of ordinary skill in the art appears in the first issue of each volume of the Journal of Organic Chemistry; this list is typically presented in a table entitled Standard List of Abbreviations. The abbreviations contained in said list, and all abbreviations utilized by organic chemists of ordinary skill in the art are hereby incorporated by reference.
- the compounds described herein can be evaluated for ability to modulate PKM2 (e.g., activate PKM2) by methods known in the art.
- compounds described herein are evaluated for ability to modulate PKM2 (e.g. activate PKM2) in serine deficient conditions.
- exemplary methods include contacting the compound with a cell-based assay which allows assessment of the ability to modulate (e.g., activate) PKM2.
- the candidate compound can be contacted with a cell and measuring the consumption of oxygen or production of lactate.
- a change in cellular phosphoenolpyruvate, a change in glycerol-phosphate, a change in ribose or deoxyribose, a change in lipid synthesis, or a change in glucose conversion to lipid or nucleic acids or amino acids or protein can also be used to evaluate a compound for its ability to modulate PKM2 (e.g., activate PKM2).
- the evaluation could also include measuring a change in pyruvate or a determination of an alteration in mitochondrial membrane potential, e.g., as measured by fluorescent potentiometric dyes.
- the activity of the PKM enzyme measured in the screening/testing assay may be measured by, e.g., monitoring the concentration of a substrate (e.g., ATP or NADH) present in the reaction mixture.
- a substrate e.g., ATP or NADH
- Pyruvate produced by the enzymatic activity of pyruvate kinase, is converted into lactate by lactate dehydrogenase, which requires the consumption of NADH (NADH ⁇ NAD+).
- the activity of PKM2 can be indirectly measured by monitoring the consumption of NADH through, e.g., fluorescence assays.
- the activity of the PKM2 enzyme can be directly monitored by measuring the production of ATP, as ATP is produced when phosphoenolpyruvate is converted to pyruvate.
- Methods for monitoring the amount of substrate in a reaction mixture include, e.g., absorbance, fluorescence, Raman scattering, phosphorescence, luminescence, luciferas
- the screening procedure requires the presence of specific components in the reaction mixture.
- Components utilized in the assay include, e.g., a nucleoside diphosphate (e.g., ADP), phosphoenolpyruvate, NADH, lactate dehydrogenase, FBP, a reducing agent (e.g., dithiothreitol), a detergent (e.g., Brij 35), glycerol, and a solvent (e.g., DMSO).
- a nucleoside diphosphate e.g., ADP
- phosphoenolpyruvate NADH
- lactate dehydrogenase FBP
- a reducing agent e.g., dithiothreitol
- a detergent e.g., Brij 35
- glycerol e.g., glycerol
- solvent e.g., DMSO
- a compound such as a compound described herein can be evaluated in a cellular/ex vivo assay.
- a cell is treated with a compound described herein (i.e., a PKM2 activator), and the compound is evaluated, for example for its ability to enter the cell and bind to PKM2, inducing an activated conformation of PKM2.
- the excess unbound compound can then be washed away with PBS, and the cells lysed, for example, by snap-freezing on dry ice, followed by addition of a detergent-containing lysis buffer.
- the lysate, in which activated PKM2 remains intact, can then be removed and added to a chemical cocktail including the chemicals necessary to measure pyruvate kinase activity.
- the assay can be coupled to another assay such as an assay that is coupled to the LDHa enzyme.
- the amount of pyruvate kinase activity that is measured can then be normalized to the total protein content in the lysate, and related to the concentration of PKM2 activator that was added to the cell. This can allow an AC 50 (concentration at which PKM2 is activated 50%) value to be derived.
- the total fold-increase in activity over mock-treated cells can also be calculated, and the “maximum level of activation” can be used to distinguish between compounds that fully activate PKM2 and compounds that can only partially activate PKM2.
- tissue for example, in a cell tumor
- animals harboring the tissue/tumor of interest can be dosed with a compound. After a specified period of time in which exposure has been achieved in the target tissue/tumor of interest, the tissue/tumor can then be harvested from the animal, snap-frozen, and then lysed and homogenized. The amount of pyruvate kinase activity in this lysate can then be quantitated as described above.
- PKM1 and PKM2 for use in the screening/testing methods described herein may be produced by any method known in the art for expression of recombinant proteins.
- nucleic acids that encode the desired polypeptide may be introduced into various cell types or cell-free systems for expression.
- Eukaryotic e.g., COS, HEK293T, CHO, and NIH cell lines
- prokaryotic e.g., E. coli expression systems may be generated in which a PKM sequence is introduced into a plasmid or other vector, which is then used to transform living cells.
- PKM cDNA contains the entire open reading frame, or biologically active fragment thereof, are inserted in the correct orientation into an expression plasmid and may be used for protein expression.
- Prokaryotic and eukaryotic expression systems allow for the expression and recovery of fusion proteins in which the PKM protein is covalently linked to a tag molecule on either the amino terminal or carboxy terminal side, which facilitates identification and/or purification.
- tags that can be used include hexahistidine, HA, FLAG, and c-myc epitope tags.
- An enzymatic or chemical cleavage site can be engineered between the PKM protein and the tag molecule so that the tag can be removed following purification.
- Compounds useful as PKM2 activators are those demonstrate specificity and activation of PKM2 enzyme in the absence of FBP to a level greater than that of 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 99, or 100% in the presence of FBP. Furthermore, compounds can be evaluated in the presence or absence of a phosphotyrosine peptide. Phosphotyrosine peptide binding to PKM2 leads to a dissociation of FBP from PKM2 and conformational changes of PKM2 from an active, tetrameric form to an inactive form.
- a method for treating or preventing a disease, condition or disorder as described herein comprising administering a compound, a pharmaceutically acceptable salt of a compound or pharmaceutical composition comprising a compound described herein (e.g., a compound of formula (I), (I-a), (II) or in FIGS. 1 A- 1 A- 44 ).
- the compounds and compositions described herein can be administered to cells in culture, e.g. in vitro or ex vivo, or to a subject, e.g., in vivo, to treat, prevent, and/or diagnose a variety of disorders, including those described herein below.
- the term “treat” or “treatment” is defined as the application or administration of a compound, alone or in combination with, a second compound to a subject, e.g., a patient, or application or administration of the compound to an isolated tissue or cell, e.g., cell line, from a subject, e.g., a patient, who has a disorder (e.g., a disorder as described herein), a symptom of a disorder, or a predisposition toward a disorder, with the purpose to cure, heal, alleviate, relieve, alter, remedy, ameliorate, improve or affect the disorder, one or more symptoms of the disorder or the predisposition toward the disorder (e.g., to prevent at least one symptom of the disorder or to delay onset of at least one symptom of the disorder).
- a disorder e.g., a disorder as described herein
- a symptom of a disorder e.g., a disorder as described herein
- a predisposition toward a disorder e.
- an amount of a compound effective to treat a disorder refers to an amount of the compound which is effective, upon single or multiple dose administration to a subject, in treating a cell, or in curing, alleviating, relieving or improving a subject with a disorder beyond that expected in the absence of such treatment.
- an amount of a compound effective to prevent a disorder refers to an amount effective, upon single- or multiple-dose administration to the subject, in preventing or delaying the occurrence of the onset or recurrence of a disorder or a symptom of the disorder.
- the term “subject” is intended to include human and non-human animals.
- exemplary human subjects include a human patient having a disorder, e.g., a disorder described herein or a normal subject.
- non-human animals includes all vertebrates, e.g., non-mammals (such as chickens, amphibians, reptiles) and mammals, such as non-human primates, domesticated and/or agriculturally useful animals, e.g., sheep, dog, cat, cow, pig, etc.
- a compound or composition described herein can be used to treat a neoplastic disorder.
- a “neoplastic disorder” is a disease or disorder characterized by cells that have the capacity for autonomous growth or replication, e.g., an abnormal state or condition characterized by proliferative cell growth.
- Exemplary neoplastic disorders include: carcinoma, sarcoma, metastatic disorders (e.g., tumors arising from prostate, colon, lung, breast and liver origin), hematopoietic neoplastic disorders, e.g., leukemias, metastatic tumors.
- Prevalent cancers include: breast, prostate, colon, lung, liver, and pancreatic cancers.
- Treatment with the compound may be in an amount effective to ameliorate at least one symptom of the neoplastic disorder, e.g., reduced cell proliferation, reduced tumor mass, etc.
- the disclosed methods are useful in the prevention and treatment of cancer, including for example, solid tumors, soft tissue tumors, and metastases thereof.
- the disclosed methods are also useful in treating non-solid cancers.
- Exemplary solid tumors include malignancies (e.g., sarcomas, adenocarcinomas, and carcinomas) of the various organ systems, such as those of lung, breast, lymphoid, gastrointestinal (e.g., colon), and genitourinary (e.g., renal, urothelial, or testicular tumors) tracts, pharynx, prostate, and ovary.
- Exemplary adenocarcinomas include colorectal cancers, renal-cell carcinoma, liver cancer, non-small cell carcinoma of the lung, and cancer of the small intestine.
- cancers include: Acute Lymphoblastic Leukemia, Adult; Acute Lymphoblastic Leukemia, Childhood; Acute Myeloid Leukemia, Adult; Adrenocortical Carcinoma; Adrenocortical Carcinoma, Childhood; AIDS-Related Lymphoma; AIDS-Related Malignancies; Anal Cancer; Astrocytoma, Childhood Cerebellar; Astrocytoma, Childhood Cerebral; Bile Duct Cancer, Extrahepatic; Bladder Cancer; Bladder Cancer, Childhood; Bone Cancer, Osteosarcoma/Malignant Fibrous Histiocytoma; Brain Stem Glioma, Childhood; Brain Tumor, Adult; Brain Tumor, Brain Stem Glioma, Childhood; Brain Tumor, Cerebellar Astrocytoma, Childhood; Brain Tumor, Cerebral Astrocytoma/Malignant Glioma, Childhood; Brain Tumor, Ependymoma, Childhood; Brain Tu
- a compound described herein is administered together with one or more additional cancer treatments.
- exemplary cancer treatments include, for example: chemotherapy, targeted therapies such as antibody therapies, immunotherapy, and hormonal therapy. Examples of each of these treatments are provided below.
- a compound described herein is administered with one or more chemotherapies.
- Chemotherapy is the treatment of cancer with drugs that can destroy cancer cells. “Chemotherapy” usually refers to cytotoxic drugs which affect rapidly dividing cells in general, in contrast with targeted therapy. Chemotherapy drugs interfere with cell division in various possible ways, e.g., with the duplication of DNA or the separation of newly formed chromosomes. Most forms of chemotherapy target all rapidly dividing cells and are not specific for cancer cells, although some degree of specificity may come from the inability of many cancer cells to repair DNA damage, while normal cells generally can.
- chemotherapeutic agents used in cancer therapy include, for example, antimetabolites (e.g., folic acid, purine, and pyrimidine derivatives) and alkylating agents (e.g., nitrogen mustards, nitrosoureas, platinum, alkyl sulfonates, hydrazines, triazenes, aziridines, spindle poison, cytotoxic agents, toposimerase inhibitors and others).
- antimetabolites e.g., folic acid, purine, and pyrimidine derivatives
- alkylating agents e.g., nitrogen mustards, nitrosoureas, platinum, alkyl sulfonates, hydrazines, triazenes, aziridines, spindle poison, cytotoxic agents, toposimerase inhibitors and others.
- agents include Aclarubicin, Actinomycin, Alitretinon, Altretamine, Aminopterin, Aminolevulinic acid, Amrubicin, Amsacrine, Anagrelide, Arsenic trioxide, Asparaginase, Atrasentan, Belotecan, Bexarotene, endamustine, Bleomycin, Bortezomib, Busulfan, Camptothecin, Capecitabine, Carboplatin, Carboquone, Carmofur, Carmustine, Celecoxib, Chlorambucil, Chlormethine, Cisplatin, Cladribine, Clofarabine, Crisantaspase, Cyclophosphamide, Cytarabine, dacarbazine, Dactinomycin, Daunorubicin, Decitabine, Demecolcine, Docetaxel, Doxorubicin, Efaproxiral, Elesclomol, Elsamitrucin, Enocita
- the chemotherapy agents can be used in combination with a compound described herein.
- a compound described herein is administered with one or more targeted therapies.
- Targeted therapy constitutes the use of agents specific for the deregulated proteins of cancer cells.
- Small molecule targeted therapy drugs are generally inhibitors of enzymatic domains on mutated, overexpressed, or otherwise critical proteins within the cancer cell.
- Prominent examples are the tyrosine kinase inhibitors such as Axitinib, Bosutinib, Cediranib, dasatinib, erlotinib, imatinib, gefitinib, lapatinib, Lestaurtinib, Nilotinib, Semaxanib, Sorafenib, Sunitinib, and Vandetanib, and also cyclin-dependent kinase inhibitors such as Alvocidib and Seliciclib.
- Monoclonal antibody therapy is another strategy in which the therapeutic agent is an antibody which specifically binds to a protein on the surface of the cancer cells.
- Examples include the anti-HER2/neu antibody trastuzumab (HERCEPTIN®) typically used in breast cancer, and the anti-CD20 antibody rituximab and Tositumomab typically used in a variety of B-cell malignancies.
- Other exemplary antibodies include Cetuximab, Panitumumab, Trastuzumab, Alemtuzumab, Bevacizumab, Edrecolomab, and Gemtuzumab.
- Exemplary fusion proteins include Aflibercept and Denileukin diftitox.
- the targeted therapy can be used in combination with a compound described herein.
- Targeted therapy can also involve small peptides as “homing devices” which can bind to cell surface receptors or affected extracellular matrix surrounding the tumor. Radionuclides which are attached to these peptides (e.g., RGDs) eventually kill the cancer cell if the nuclide decays in the vicinity of the cell.
- RGDs Radionuclides which are attached to these peptides
- An example of such therapy includes BEXXAR®.
- a compound described herein is administered with one or more immunotherapies.
- Cancer immunotherapy refers to a diverse set of therapeutic strategies designed to induce the patient's own immune system to fight the tumor.
- Contemporary methods for generating an immune response against tumors include intravesicular BCG immunotherapy for superficial bladder cancer, and use of interferons and other cytokines to induce an immune response in renal cell carcinoma and melanoma patients.
- Allogeneic hematopoietic stem cell transplantation can be considered a form of immunotherapy, since the donor's immune cells will often attack the tumor in a graft-versustumor effect.
- the immunotherapy agents can be used in combination with a compound described herein.
- a compound described herein is administered with one or more hormonal therapies.
- the growth of some cancers can be inhibited by providing or blocking certain hormones.
- hormone-sensitive tumors include certain types of breast and prostate cancers. Removing or blocking estrogen or testosterone is often an important additional treatment.
- administration of hormone agonists, such as progestogens may be therapeutically beneficial.
- the hormonal therapy agents can be used in combination with a compound described herein.
- a compound or composition described herein can be used to treat or prevent obesity, e.g., in a human subject, e.g. a child or adult subject.
- “Obesity” refers to a condition in which a subject has a body mass index of greater than or equal to 30.
- Many compounds described herein can be used to treat or prevent an over-weight condition.
- “Over-weight” refers to a condition in which a subject has a body mass index of greater or equal to 25.0.
- the body mass index (BMI) and other definitions are according to the “NIH Clinical Guidelines on the Identification and Evaluation, and Treatment of Overweight and Obesity in Adults” (1998).
- Treatment with the compound may be in an amount effective to alter the weight of the subject, e.g., by at least 2, 5, 7, 10, 12, 15, 20, 25, 30, 25, 40, 45, 50, or 55%.
- Treatment with a compound may be in an amount effective to reduce the body mass index of the subject, e.g., to less than 30, 28, 27, 25, 22, 20, or 18.
- the compounds can be used to treat or prevent aberrant or inappropriate weight gain, metabolic rate, or fat deposition, e.g., anorexia, bulimia, obesity, diabetes, or hyperlipidemia (e.g., elevated triglycerides and/or elevated cholesterol), as well as disorders of fat or lipid metabolism.
- a compound or composition described herein can be administered to treat obesity associated with Prader-Willi Syndrome (PWS).
- PWS is a genetic disorder associated with obesity (e.g., morbid obesity).
- a compound or composition described herein can be used to reduce body fat, prevent increased body fat, reduce cholesterol (e.g., total cholesterol and/or ratios of total cholesterol to HDL cholesterol), and/or reduce appetite in individuals having PWS associated obesity, and/or reduce comorbidities such as diabetes, cardiovascular disease, and stroke.
- reduce cholesterol e.g., total cholesterol and/or ratios of total cholesterol to HDL cholesterol
- reduce appetite in individuals having PWS associated obesity, and/or reduce comorbidities such as diabetes, cardiovascular disease, and stroke.
- compositions delineated herein include the compounds delineated herein (e.g., a compound described herein), as well as additional therapeutic agents if present, in amounts effective for achieving a modulation of disease or disease symptoms, including those described herein.
- pharmaceutically acceptable carrier or adjuvant refers to a carrier or adjuvant that may be administered to a patient, together with a compound provided herewith, and which does not destroy the pharmacological activity thereof and is nontoxic when administered in doses sufficient to deliver a therapeutic amount of the compound.
- Pharmaceutically acceptable carriers, adjuvants and vehicles that may be used in the pharmaceutical compositions provided herewith include, but are not limited to, ion exchangers, alumina, aluminum stearate, lecithin, self-emulsifying drug delivery systems (SEDDS) such as d- ⁇ -tocopherol polyethyleneglycol 1000 succinate, surfactants used in pharmaceutical dosage forms such as Tweens or other similar polymeric delivery matrices, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene-
- Cyclodextrins such as ⁇ -, ⁇ -, and ⁇ -cyclodextrin, or chemically modified derivatives such as hydroxyalkylcyclodextrins, including 2- and 3-hydroxypropyl- ⁇ -cyclodextrins, or other solubilized derivatives may also be advantageously used to enhance delivery of compounds of the formulae described herein.
- compositions provided herewith may be administered orally, parenterally, by inhalation spray, topically, rectally, nasally, buccally, vaginally or via an implanted reservoir, preferably by oral administration or administration by injection.
- the pharmaceutical compositions provided herewith may contain any conventional non-toxic pharmaceutically-acceptable carriers, adjuvants or vehicles.
- the pH of the formulation may be adjusted with pharmaceutically acceptable acids, bases or buffers to enhance the stability of the formulated compound or its delivery form.
- parenteral as used herein includes subcutaneous, intracutaneous, intravenous, intramuscular, intraarticular, intraarterial, intrasynovial, intrasternal, intrathecal, intralesional and intracranial injection or infusion techniques.
- the pharmaceutical compositions may be in the form of a sterile injectable preparation, for example, as a sterile injectable aqueous or oleaginous suspension.
- This suspension may be formulated according to techniques known in the art using suitable dispersing or wetting agents (such as, for example, Tween 80) and suspending agents.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally acceptable diluent or solvent, for example, as a solution in 1,3-butanediol.
- suitable vehicles and solvents that may be employed are mannitol, water, Ringer's solution and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- Fatty acids, such as oleic acid and its glyceride derivatives are useful in the preparation of injectables, as are natural pharmaceutically-acceptable oils, such as olive oil or castor oil, especially in their polyoxyethylated versions.
- These oil solutions or suspensions may also contain a long-chain alcohol diluent or dispersant, or carboxymethyl cellulose or similar dispersing agents which are commonly used in the formulation of pharmaceutically acceptable dosage forms such as emulsions and or suspensions.
- surfactants such as Tweens or Spans and/or other similar emulsifying agents or bioavailability enhancers which are commonly used in the manufacture of pharmaceutically acceptable solid, liquid, or other dosage forms may also be used for the purposes of formulation.
- compositions provided herewith may be orally administered in any orally acceptable dosage form including, but not limited to, capsules, tablets, emulsions and aqueous suspensions, dispersions and solutions.
- carriers which are commonly used include lactose and corn starch.
- Lubricating agents such as magnesium stearate, are also typically added.
- useful diluents include lactose and dried corn starch.
- aqueous suspensions and/or emulsions are administered orally, the active ingredient may be suspended or dissolved in an oily phase is combined with emulsifying and/or suspending agents. If desired, certain sweetening and/or flavoring and/or coloring agents may be added.
- compositions provided herewith may also be administered in the form of suppositories for rectal administration.
- These compositions can be prepared by mixing a compound provided herewith a suitable non-irritating excipient which is solid at room temperature but liquid at the rectal temperature and therefore will melt in the rectum to release the active components.
- suitable non-irritating excipient include, but are not limited to, cocoa butter, beeswax and polyethylene glycols.
- Topical administration of the pharmaceutical compositions provided herewith is useful when the desired treatment involves areas or organs readily accessible by topical application.
- the pharmaceutical composition should be formulated with a suitable ointment containing the active components suspended or dissolved in a carrier.
- Carriers for topical administration of the compounds provided herewith include, but are not limited to, mineral oil, liquid petroleum, white petroleum, propylene glycol, polyoxyethylene polyoxypropylene compound, emulsifying wax and water.
- the pharmaceutical composition can be formulated with a suitable lotion or cream containing the active compound suspended or dissolved in a carrier with suitable emulsifying agents.
- Suitable carriers include, but are not limited to, mineral oil, sorbitan monostearate, polysorbate 60, cetyl esters wax, cetearyl alcohol, 2-octyldodecanol, benzyl alcohol and water.
- the pharmaceutical compositions provided herewith may also be topically applied to the lower intestinal tract by rectal suppository formulation or in a suitable enema formulation. Topically-transdermal patches are also included.
- compositions provided herewith may be administered by nasal aerosol or inhalation.
- Such compositions are prepared according to techniques well-known in the art of pharmaceutical formulation and may be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other solubilizing or dispersing agents known in the art.
- compositions provided herewith comprise a combination of a compound of the formulae described herein and one or more additional therapeutic or prophylactic agents
- both the compound and the additional agent should be present at dosage levels of between about 1 to 100%, and more preferably between about 5 to 95% of the dosage normally administered in a monotherapy regimen.
- the additional agents may be administered separately, as part of a multiple dose regimen, from the compounds provided herewith. Alternatively, those agents may be part of a single dosage form, mixed together with the compounds provided herewith in a single composition.
- the compounds described herein can, for example, be administered by injection, intravenously, intraarterially, subdermally, intraperitoneally, intramuscularly, or subcutaneously; or orally, buccally, nasally, transmucosally, topically, in an ophthalmic preparation, or by inhalation, with a dosage ranging from about 0.5 to about 100 mg/kg of body weight, alternatively dosages between 1 mg and 1000 mg/dose, every 4 to 120 hours, or according to the requirements of the particular drug.
- the methods herein contemplate administration of an effective amount of compound or compound composition to achieve the desired or stated effect.
- the pharmaceutical compositions provided herewith will be administered from about 1 to about 6 times per day or alternatively, as a continuous infusion.
- Such administration can be used as a chronic or acute therapy.
- the amount of active ingredient that may be combined with the carrier materials to produce a single dosage form will vary depending upon the host treated and the particular mode of administration.
- a typical preparation will contain from about 5% to about 95% active compound (w/w).
- such preparations contain from about 20% to about 80% active compound.
- a maintenance dose of a compound, composition or combination provided herewith may be administered, if necessary. Subsequently, the dosage or frequency of administration, or both, may be reduced, as a function of the symptoms, to a level at which the improved condition is retained when the symptoms have been alleviated to the desired level. Patients may, however, require intermittent treatment on a long-term basis upon any recurrence of disease symptoms.
- the compounds described herein can modulate PKM2. Accordingly, a patient and/or subject can be selected for treatment using a compound described herein by first evaluating the patient and/or subject to determine whether the subject is in need of modulation of PKM2, and if the subject is determined to be in need of modulation of PKM2, then administering to the subject a compound described herein.
- a subject can be evaluated as being in need of modulation of PKM2 using methods known in the art, e.g., by measuring the presence and/or activity of PKM2 in the patient.
- the activity and/or level of PKM2 is evaluated in the cancer.
- a patient receiving a compound described herein can be monitored, for example, for improvement in the condition and/or adverse effects. Improvement of a patient's condition can be evaluated, for example, by monitoring the growth, absence of growth, or regression of the cancer (e.g., a tumor). In some embodiments, the patient is evaluated using a radiological assay or evaluation of hemolytic parameters.
- tert-butyl-4-(2-methoxyphenyl) piperazine-1-carboxylate compound 5, 0.600 g, 0.00205 moles, 1 eq
- ether-HCl 10 mL
- the resulting mixture was stirred for overnight.
- ether was removed under reduced pressure and a solid material was obtained.
- the solid material was washed with ethyl acetate and dried to obtain the amine compound 6 as a white solid (0.425 g, 90.08%).
- Compound 8 was prepared from commercially available 2-Methoxy-5-chlorobenzenesulfonyl chloride as shown in Scheme 2.
- naphthalene-2-sulfonyl chloride was utilized in place of compound 2 used to provide 10.
- reaction was monitored by TLC. Upon completion, the reaction mixture was washed with water and extracted with ethyl acetate. The organic layer was dried over sodium sulfate and concentrated under vacuum. The crude product was purified by crystallization with an ethyl acetate/hexane solvent mixture to obtain compound 24 in 24.6% yield (35 mg).
- Compound 45 was prepared from 4-chlorobenzenesulfonyl chloride following the protocol described in Scheme 7 using the general procedure provided in Scheme 4.
- N 1 -Boc-N 4 -arylpiperazine (86, 1.075 mmol) was taken into a round bottomed flask and was added methanolic-HCl (20 ml, 20%) which resulted in formation of a homogeneous solution and was stirred for 1 h at room temperature. After completion of the reaction (monitored by TLC), the solvent was removed under vacuum. The crude product was washed with ethyl acetate repeatedly and then dried well to obtain compound 87 (90%) as a white solid.
- N 1 -Boc-N 4 -arylpiperazine (94, 1.075 mmol) was taken into a round bottomed flask and was added methanolic-HCl (20 ml, 20%) which resulted in formation of a homogeneous solution and was stirred for 1 h at room temperature. After completion of the reaction (monitored by TLC), the solvent was removed under vacuum. The crude product was washed with ethyl acetate repeatedly and then dried well to obtain hydrochloride salt (95) (90%) as a white solid.
- N 1 -Boc-N 4 -arylhomopiperazine (100, 1.070 mmol) was taken into a round bottomed flask and was added methanolic-HCl (20 ml, 20%) which resulted in formation of a homogeneous solution and was stirred for 1 h at room temperature. After completion of the reaction (monitored by TLC), the solvent was removed under vacuum. The crude product was washed with ethyl acetate repeatedly and then dried well to obtain compound (101) (90%) as a white solid.
- EDCI (3.8 g, 19.8 mmol) and HOBT (2.67 g, 19.8 mmol) were added to a stirred solution of the acid 91 (6.5 g, 19.8 mmol) in anhydrous DMF.
- the temperature of the mixture was reduced to 0° C., at which time DIPEA (11 ml, 59.45 mmol) was added under nitrogen atmosphere and the resultant solution (or suspension) was stirred at room temperature for 30 min.
- Boc-piperazine (3.68 g, 19.8 mmol) was then added at 0° C.
- the reaction mixture was then brought to room temperature and stirred overnight. After completion of the reaction, the reaction mixture was diluted with water and extracted with ethyl acetate (3 ⁇ 70 ml).
Abstract
Description
In some aspects of these embodiments, Ra is alkyl (e.g., methyl or ethyl). In some aspects of these embodiments, L is a bond, —CH2—, —C(O)—, or —O(CO)—.
wherein L, R1, R3, Ra, Rb, Rc, Rd, Y, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
and D, D1, L, R1, R3, Ra, Rb, Rc, Rd, X, Y, Z, W, n, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
wherein A, L, R1, R3, Ra, Rb, Rc, Rd, W, X, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
wherein A, L, R1, R3, Ra, Rb, Rc, Rd, W, X, Y, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
wherein A, L, R1, R3, Ra, Rb, Rc, Rd, Y, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
wherein A, L, R1, R3, Ra, Rb, Rc, Rd, W, X, Y, Z, m, h and g are as defined above in formula (I) or any one of the embodiments or aspects described herein.
-
- a. it is an allosteric activator of PKM2;
- b. it modulates (e.g., stabilizes) the binding of FBP in a binding pocket of PKM2;
- c. it modulates (e.g., promotes) the release of FBP from a binding pocket of PKM2;
- d. it is a modulator (e.g., an agonist), e.g., an analog, of FBP, e.g., an agonist which binds PKM2 with a lower, about the same, or higher affinity than does FBP;
- e. it modulates (e.g., promotes) the dissolution of tetrameric PKM2;
- f. it modulates (e.g., promotes) the assembly of tetrameric PKM2;
- g. it modulates (e.g., stabilizes) the tetrameric conformation of PKM2;
- h. it modulates (e.g., promotes) the binding of a phosphotyrosine containing polypeptide to PKM2;
- i. it modulates (e.g., promotes) the ability of a phosphotyrosine containing polypeptide to induce release of FBP from PKM2, e.g., by inducing a change in the conformation of PKM2, e.g., in the position of Lys 433, thereby hindering the release of FBP;
- k. it binds to or changes the position of Lys 433 relative to the FBP binding pocket;
- l. it selectively modulates (e.g., activates) PKM2 over at least one other isoform of PK, e.g., it is selective for PKM2 over one or more of PKR, PKM1, or PKL;
- m. it has an affinity for PKM2 which is greater than its affinity for at least one other isoform of PK, e.g., PKR, PKM1, or PKL. A compound described herein may be tested for its ability to activate PKM2. For simplicity, the activation activity of these compounds is represented as an AC50 in
FIGS. 1A-1A-44 and throughout the application. Exemplary compounds are shown inFIGS. 1A-1A-44 . As shown inFIGS. 1A-1A-44 , “A” refers to an activator of PKM2 with an EC50<100 nM. “B” refers to an activator of PKM2 with an EC50 between 100 nM and 500 nM. “C” refers to an activator of PKM2 with an EC50 between 500 nM and 1000 nM. “D” refers to an activator of PKM2 with an EC50 between 1 μM and 10 μM. “E” refers to data that is not available.
| TABLE 1 | |||
| Amount in | |||
| Activation | |||
| Component of Reaction Condition | Assay | ||
| ADP | 0.1-5.0 | mM | |
| Phosphoenolpyruvate | 0.1-5.0 | mM | |
| NADH | 10-1000 | μM | |
| Lactate dehydrogenase | 0.1-10 | units | |
| Fructose-1,6- |
0 | ||
| DTT | 0.1-50 | mM |
| Brij 35 | 0.01-1% | |
| Glycerol | 0.1-10% |
| Pyruvate Kinase M2 (used for screen) | 1-100 | pg |
| DMSO | 1-10% | ||
-
- PKM2 stock enzyme solution was diluted in Reaction Buffer
- 2 μL of compound was added into each well first, and then 180 μL of the Reaction Mix was added.
- Reaction mixture with compound (without ADP) were incubated for 30 minutes at 4° C.
- Plates were re-equilibrated to room temperature prior to adding 20 μL ADP to initiate the reaction.
- Reaction progress was measured as changes in absorbance at 340 nm wavelength at room temperature (25° C.)
Reaction Mix:
| TABLE 2 | |||
| S. No. | R | R′ | n |
| 60 | | H | 1 |
| 61 | | H | 1 |
| 62 | | H | 1 |
| 63 | | H | 1 |
| 64 | | H | 1 |
| 65 | | H | 1 |
| 66 | | H | 1 |
| 67 | | H | 1 |
| 68 | | H | 1 |
| 69 | | H | 1 |
| 70 | | H | 1 |
| 71 | | H | 1 |
| 72 | | H | 1 |
| 73 | | H | 1 |
| 74 | | H | 1 |
| 75 | | H | 1 |
| 76 | | Na+ | 1 |
| 77 | | CH3SO3 − | 1 |
| 78 | | H | 1 |
| 79 | | H | 2 |
| 80 | | H | 1 |
| 81 | | H | 1 |
| 82 | | H | 1 |
| 83 | | H | 1 |
Preparation of Compound 55:
Claims (57)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17/131,562 USRE49582E1 (en) | 2009-06-29 | 2020-12-22 | Therapeutic compounds and compositions |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US22143009P | 2009-06-29 | 2009-06-29 | |
| US29236010P | 2010-01-05 | 2010-01-05 | |
| US12/826,630 US8785450B2 (en) | 2009-06-29 | 2010-06-29 | Therapeutic compounds and compositions |
| US17/131,562 USRE49582E1 (en) | 2009-06-29 | 2020-12-22 | Therapeutic compounds and compositions |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/826,630 Reissue US8785450B2 (en) | 2009-06-29 | 2010-06-29 | Therapeutic compounds and compositions |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE49582E1 true USRE49582E1 (en) | 2023-07-18 |
Family
ID=43381412
Family Applications (7)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/826,630 Ceased US8785450B2 (en) | 2009-06-29 | 2010-06-29 | Therapeutic compounds and compositions |
| US14/286,088 Abandoned US20140323467A1 (en) | 2009-06-29 | 2014-05-23 | Therapeutic compounds and compositions |
| US15/412,976 Active US10029987B2 (en) | 2009-06-29 | 2017-01-23 | Therapeutic compounds and compositions |
| US16/010,717 Abandoned US20190241523A1 (en) | 2009-06-29 | 2018-06-18 | Therapeutic compounds and compositions |
| US16/243,247 Active US10988448B2 (en) | 2009-06-29 | 2019-01-09 | Therapeutic compounds and compositions |
| US17/131,562 Active 2031-02-24 USRE49582E1 (en) | 2009-06-29 | 2020-12-22 | Therapeutic compounds and compositions |
| US17/195,243 Active 2030-12-29 US11866411B2 (en) | 2009-06-29 | 2021-03-08 | Therapeutic compounds and compositions |
Family Applications Before (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/826,630 Ceased US8785450B2 (en) | 2009-06-29 | 2010-06-29 | Therapeutic compounds and compositions |
| US14/286,088 Abandoned US20140323467A1 (en) | 2009-06-29 | 2014-05-23 | Therapeutic compounds and compositions |
| US15/412,976 Active US10029987B2 (en) | 2009-06-29 | 2017-01-23 | Therapeutic compounds and compositions |
| US16/010,717 Abandoned US20190241523A1 (en) | 2009-06-29 | 2018-06-18 | Therapeutic compounds and compositions |
| US16/243,247 Active US10988448B2 (en) | 2009-06-29 | 2019-01-09 | Therapeutic compounds and compositions |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US17/195,243 Active 2030-12-29 US11866411B2 (en) | 2009-06-29 | 2021-03-08 | Therapeutic compounds and compositions |
Country Status (39)
| Country | Link |
|---|---|
| US (7) | US8785450B2 (en) |
| EP (3) | EP3241554B1 (en) |
| JP (2) | JP5856052B2 (en) |
| KR (2) | KR101850813B1 (en) |
| CN (2) | CN102481300B (en) |
| AR (1) | AR077292A1 (en) |
| AU (1) | AU2010266386C1 (en) |
| BR (1) | BRPI1011587B1 (en) |
| CA (2) | CA2766882C (en) |
| CL (1) | CL2011003350A1 (en) |
| CO (1) | CO6480955A2 (en) |
| CR (2) | CR20170071A (en) |
| CY (1) | CY1119443T1 (en) |
| DK (1) | DK2448582T3 (en) |
| EC (1) | ECSP11011567A (en) |
| ES (1) | ES2632954T3 (en) |
| FI (1) | FIC20230017I1 (en) |
| FR (1) | FR23C1017I2 (en) |
| HR (1) | HRP20170955T1 (en) |
| HU (2) | HUE032969T2 (en) |
| IL (1) | IL217277A (en) |
| IN (1) | IN2012DN00471A (en) |
| LT (2) | LT2448582T (en) |
| MX (1) | MX2012000256A (en) |
| MY (1) | MY191825A (en) |
| NI (1) | NI201200003A (en) |
| NL (1) | NL301230I2 (en) |
| NO (1) | NO2023017I1 (en) |
| NZ (2) | NZ622505A (en) |
| PE (1) | PE20120693A1 (en) |
| PL (1) | PL2448582T3 (en) |
| PT (1) | PT2448582T (en) |
| RU (1) | RU2561132C2 (en) |
| SG (3) | SG177434A1 (en) |
| SI (1) | SI2448582T1 (en) |
| TW (4) | TWI598337B (en) |
| UA (1) | UA107667C2 (en) |
| WO (1) | WO2011002817A1 (en) |
| ZA (1) | ZA201200054B (en) |
Families Citing this family (73)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2740424T3 (en) | 2009-03-13 | 2020-02-05 | Agios Pharmaceuticals Inc | Methods and compositions for disorders related to cell proliferation |
| JP5756457B2 (en) | 2009-04-06 | 2015-07-29 | アジオス ファーマシューティカルズ, インコーポレイテッド | Pyruvate kinase M2 modulators, therapeutic compositions and related methods of use |
| ES2619557T3 (en) | 2009-05-04 | 2017-06-26 | Agios Pharmaceuticals, Inc. | PKM2 activators for use in cancer treatment |
| BRPI1015135B1 (en) | 2009-06-29 | 2021-08-03 | Incyte Holdings Corporation | PI3K-INHIBITING PYRIMIDINONES, COMPOSITION INCLUDING SUCH COMPOUNDS AS WELL AS USES THEREOF |
| ES2618630T3 (en) | 2009-06-29 | 2017-06-21 | Agios Pharmaceuticals, Inc. | Therapeutic compositions and related methods of use |
| PT2448582T (en) * | 2009-06-29 | 2017-07-10 | Agios Pharmaceuticals Inc | Quinoline-8-sulfonamide derivatives having an anticancer activity |
| EP3561077B1 (en) | 2009-10-21 | 2022-12-21 | Les Laboratoires Servier | Methods for cell-proliferation-related disorders |
| CA2793836C (en) | 2009-10-21 | 2020-03-24 | Agios Pharmaceuticals, Inc. | Methods and compositions for cell-proliferation-related disorders |
| EP2509600B1 (en) * | 2009-12-09 | 2017-08-02 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds for use in the treatment of cancer characterized as having an idh mutation |
| WO2011075643A1 (en) | 2009-12-18 | 2011-06-23 | Incyte Corporation | Substituted heteroaryl fused derivatives as pi3k inhibitors |
| US9193721B2 (en) | 2010-04-14 | 2015-11-24 | Incyte Holdings Corporation | Fused derivatives as PI3Kδ inhibitors |
| WO2011163195A1 (en) | 2010-06-21 | 2011-12-29 | Incyte Corporation | Fused pyrrole derivatives as pi3k inhibitors |
| WO2012056319A1 (en) | 2010-10-27 | 2012-05-03 | Dynamix Pharmaceuticals Ltd. | Sulfonamides for the modulation of pkm2 |
| FR2967674B1 (en) | 2010-11-23 | 2012-12-14 | Pf Medicament | HETEROARYLSULFONAMIDE DERIVATIVES, THEIR PREPARATION AND THEIR APPLICATION IN HUMAN THERAPEUTICS |
| ES2564952T3 (en) | 2010-12-17 | 2016-03-30 | Agios Pharmaceuticals, Inc. | New derivatives of N- (4- (azetidine-1-carbonyl) phenyl) - (hetero-) arylsulfonamide as modulators of pyruvate kinase M2 (PKM2) |
| JP5961187B2 (en) | 2010-12-20 | 2016-08-02 | インサイト・ホールディングス・コーポレイションIncyte Holdings Corporation | N- (1- (substituted phenyl) ethyl) -9H-purin-6-amine as a PI3K inhibitor |
| EP2655350B1 (en) | 2010-12-21 | 2016-03-09 | Agios Pharmaceuticals, Inc. | Bicyclic pkm2 activators |
| TWI549947B (en) | 2010-12-29 | 2016-09-21 | 阿吉歐斯製藥公司 | Therapeutic compounds and compositions |
| US9108984B2 (en) | 2011-03-14 | 2015-08-18 | Incyte Corporation | Substituted diamino-pyrimidine and diamino-pyridine derivatives as PI3K inhibitors |
| US9126948B2 (en) | 2011-03-25 | 2015-09-08 | Incyte Holdings Corporation | Pyrimidine-4,6-diamine derivatives as PI3K inhibitors |
| ES2746558T3 (en) | 2011-05-03 | 2020-03-06 | Agios Pharmaceuticals Inc | Pyruvate kinase R activators for therapy use |
| WO2012151448A1 (en) * | 2011-05-03 | 2012-11-08 | Agios Pharmaceuticals, Inc. | Pyruvate kinase activators for use in therapy |
| MX367387B (en) * | 2011-05-03 | 2019-08-19 | Agios Pharmaceuticals Inc | Pyruvate kinase activators for use in therapy. |
| WO2012151440A1 (en) | 2011-05-03 | 2012-11-08 | Agios Pharmaceuticals, Inc. | Pyruvate kinase activators for use for increasing lifetime of the red blood cells and treating anemia |
| CN102827073A (en) | 2011-06-17 | 2012-12-19 | 安吉奥斯医药品有限公司 | Therapeutically active compositions and application methods thereof |
| CN102827170A (en) | 2011-06-17 | 2012-12-19 | 安吉奥斯医药品有限公司 | Active treatment compositions and use method thereof |
| WO2013033569A1 (en) | 2011-09-02 | 2013-03-07 | Incyte Corporation | Heterocyclylamines as pi3k inhibitors |
| US20140249150A1 (en) * | 2011-10-13 | 2014-09-04 | Agios Pharmaceuticals, Inc | Activators of pyruvate kinase m2 and methods of treating disease |
| US9474779B2 (en) | 2012-01-19 | 2016-10-25 | Agios Pharmaceuticals, Inc. | Therapeutically active compositions and their methods of use |
| CN104334527B (en) * | 2012-02-22 | 2017-05-24 | 桑福德-伯纳姆医学研究院 | Sulfonamide compounds and uses as tnap inhibitors |
| AR090548A1 (en) | 2012-04-02 | 2014-11-19 | Incyte Corp | BICYCLIC AZAHETEROCICLOBENCILAMINS AS PI3K INHIBITORS |
| US9605521B2 (en) | 2012-09-14 | 2017-03-28 | Weatherford Technology Holdings, Llc | Gas lift valve with mixed bellows and floating constant volume fluid chamber |
| CN104822373B (en) | 2012-10-15 | 2018-08-28 | 安吉奥斯医药品有限公司 | Therapeutic compound and composition |
| WO2014071512A1 (en) * | 2012-11-06 | 2014-05-15 | Universite Laval | Combination therapy and methods for the treatment of respiratory diseases |
| CN103808929A (en) * | 2012-11-08 | 2014-05-21 | 中国科学院上海生命科学研究院 | GBSS1 specific enzyme activity determination method |
| NZ707778A (en) | 2012-11-08 | 2019-03-29 | Agios Pharmaceuticals Inc | Therapeutic compounds and compositions and their use as pkm2 modulators |
| WO2014139144A1 (en) | 2013-03-15 | 2014-09-18 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| US9579324B2 (en) | 2013-07-11 | 2017-02-28 | Agios Pharmaceuticals, Inc | Therapeutically active compounds and their methods of use |
| EP3019480B1 (en) | 2013-07-11 | 2020-05-06 | Agios Pharmaceuticals, Inc. | 2,4- or 4,6-diaminopyrimidine compounds as idh2 mutants inhibitors for the treatment of cancer |
| WO2015003355A2 (en) | 2013-07-11 | 2015-01-15 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| WO2015003360A2 (en) | 2013-07-11 | 2015-01-15 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| US20150031627A1 (en) | 2013-07-25 | 2015-01-29 | Agios Pharmaceuticals, Inc | Therapeutically active compounds and their methods of use |
| WO2015138839A1 (en) | 2014-03-14 | 2015-09-17 | Agios Pharmaceuticals, Inc. | Pharmaceutical compositions of therapeutically active compounds |
| US10077277B2 (en) | 2014-06-11 | 2018-09-18 | Incyte Corporation | Bicyclic heteroarylaminoalkyl phenyl derivatives as PI3K inhibitors |
| GB201419579D0 (en) * | 2014-11-03 | 2014-12-17 | Iomet Pharma Ltd | Pharmaceutical compound |
| US11225469B2 (en) | 2015-01-15 | 2022-01-18 | Whitehead Institute For Biomedical Research | Inhibitors of phosphoglycerate dehydrogenase (PHGDH) and uses thereof |
| CR20170389A (en) | 2015-02-27 | 2018-01-26 | Incyte Corp | P13K INHIBITOR SALTS AND PREPARATION PROCESSES |
| CN104803923B (en) * | 2015-04-22 | 2017-09-05 | 江苏恩华药业股份有限公司 | A kind of preparation method of 1 (2 pyrimidine) piperazine hydrochloride |
| US9988401B2 (en) | 2015-05-11 | 2018-06-05 | Incyte Corporation | Crystalline forms of a PI3K inhibitor |
| US9732097B2 (en) | 2015-05-11 | 2017-08-15 | Incyte Corporation | Process for the synthesis of a phosphoinositide 3-kinase inhibitor |
| EP4344703A1 (en) * | 2015-06-11 | 2024-04-03 | Agios Pharmaceuticals, Inc. | Methods of using pyruvate kinase activators |
| EA039829B1 (en) | 2015-10-15 | 2022-03-17 | Аджиос Фармасьютикалз, Инк. | Combination therapy for treating malignancies |
| EP3362065B1 (en) | 2015-10-15 | 2024-04-03 | Les Laboratoires Servier | Combination therapy comprising ivosidenib, cytarabine and daunorubicin or idarubicin for treating acute myelogenous leukemia |
| AR111295A1 (en) | 2017-03-20 | 2019-06-26 | Forma Therapeutics Inc | PIRROLOPIRROL COMPOSITIONS AS PIRUVATO QUINASA ACTIVATORS (PKR) |
| TWI796353B (en) | 2017-08-15 | 2023-03-21 | 美商阿吉歐斯製藥公司 | Pyruvate kinase modulators and use thereof |
| WO2019099651A1 (en) * | 2017-11-16 | 2019-05-23 | Agios Pharmaceuticals, Inc. | Methods of using deuterated pyruvate kinase activators |
| DK3713919T3 (en) * | 2017-11-22 | 2023-09-25 | Agios Pharmaceuticals Inc | CRYSTALLINE FORMS OF N-(4-(4-(CYCLOPROPYLMETHYL)PIPERAZINE-1-CARBONYL)PHENYL)QUINOLINE-8-SULFONAMIDE |
| CN108440505A (en) * | 2018-03-30 | 2018-08-24 | 中南大学 | The total synthesis method of eptapirone |
| US10980788B2 (en) | 2018-06-08 | 2021-04-20 | Agios Pharmaceuticals, Inc. | Therapy for treating malignancies |
| CN113226356A (en) | 2018-09-19 | 2021-08-06 | 福马治疗股份有限公司 | Activating pyruvate kinase R |
| US20200129485A1 (en) | 2018-09-19 | 2020-04-30 | Forma Therapeutics, Inc. | Treating sickle cell disease with a pyruvate kinase r activating compound |
| WO2020132459A1 (en) * | 2018-12-20 | 2020-06-25 | The Regents Of The University Of Michigan | Quinolinyl-pyrazine-carboxamide compounds and uses thereof |
| US20220127267A1 (en) | 2019-02-13 | 2022-04-28 | Agios Pharmaceuticals, Inc. | Thieno[3,2-b] pyrrole[3,2-d]pyridazinone derivatives and their use as pkm2 derivatives for the treatment of cancer, obesity and diabetes related disorders |
| EP3972957A1 (en) * | 2019-05-22 | 2022-03-30 | Agios Pharmaceuticals, Inc. | Crystalline salt forms of n-(4-(4-(cyclopropylmethyl)piperazine-1-carbonyl)phenyl)quinoline-8-sulfonamide |
| US11878049B1 (en) * | 2019-06-14 | 2024-01-23 | Agios Pharmaceuticals, Inc. | Mitapivat therapy and modulators of cytochrome P450 |
| US20230015914A1 (en) * | 2019-11-07 | 2023-01-19 | Crinetics Pharmaceuticals, Inc. | Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereof |
| WO2021154987A1 (en) | 2020-01-28 | 2021-08-05 | Teva Pharmaceuticals International Gmbh | Solid state forms of mitapivat and process for preparation thereof |
| MX2023003431A (en) | 2020-09-25 | 2023-05-12 | Agios Pharmaceuticals Inc | Pharmaceutical formulation. |
| WO2022231627A1 (en) | 2021-04-30 | 2022-11-03 | Agios Pharmaceuticals, Inc. | Methods for titrating mitapivat for use in treating thalassemia |
| AU2022267322A1 (en) | 2021-04-30 | 2023-11-30 | Agios Pharmaceuticals, Inc. | Methods for titrating mitapivat |
| WO2023154036A1 (en) | 2022-02-08 | 2023-08-17 | Agios Pharmaceuticals, Inc. | Methods for titrating mitapivat |
| TW202329935A (en) | 2021-11-16 | 2023-08-01 | 美商阿吉歐斯製藥公司 | Compounds for treating mds-associated anemias and other conditions |
| DE202022106287U1 (en) | 2022-11-09 | 2022-11-21 | Ahmad Salawi | A method for the synthesis and biological evaluation of new sulfonamide analogues |
Citations (260)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2390529A (en) | 1942-02-03 | 1945-12-11 | Ernst A H Friedheim | Hydrazino-1,3,5-triazino derivatives of substituted phenylarsenic compounds |
| US3046122A (en) | 1949-07-23 | 1962-07-24 | Azoplate Corp | Process of making printing plates and light sensitive material suitable for use therein |
| US3097210A (en) | 1959-04-06 | 1963-07-09 | Merck & Co Inc | Substituted pyrazole sulfonamides |
| GB1274436A (en) | 1970-06-09 | 1972-05-17 | Wolfen Filmfab Veb | Process for the sensitization of photographic silver chloride and silver chlorobromide emulsions that may contain colour couplers |
| US3755322A (en) | 1970-07-15 | 1973-08-28 | Ciba Geigy Corp | Diamino-s-triazines |
| US3867383A (en) | 1971-03-29 | 1975-02-18 | Ciba Geigy Corp | Monoanthranilatoanilino-s-triazines |
| US3998828A (en) | 1975-01-31 | 1976-12-21 | Pfizer Inc. | 4-[2-(1,3-Dialkyl-1,2,3,4-tetra-hydropyrimidine-2,4-dione-5-carboxamido)ethyl]-1-piperidine sulfonamide |
| US4084053A (en) | 1974-06-21 | 1978-04-11 | Ciba-Geigy Corporation | Polycyclic compounds |
| US4235871A (en) | 1978-02-24 | 1980-11-25 | Papahadjopoulos Demetrios P | Method of encapsulating biologically active materials in lipid vesicles |
| EP0022958A1 (en) | 1979-07-14 | 1981-01-28 | Bayer Ag | Urea derivatives for use in the treatment of fat metabolism disorders |
| US4315940A (en) | 1979-12-01 | 1982-02-16 | Hoechst Aktiengesellschaft | Antidiabetic 1-piperidine-sulfonylureas |
| DE3314663A1 (en) | 1982-04-27 | 1983-10-27 | Nippon Kayaku K.K., Tokyo | METHOD FOR DYEING CELLULOSE FIBERS OR CELLULOSE MIXED FIBERS AND THE DYES THAT CAN BE USED FOR THIS |
| US4474599A (en) | 1982-07-14 | 1984-10-02 | The Dow Chemical Company | 1-(Pyridyl)-1H-1,2,3-triazole derivatives, and use as herbicidal agents |
| US4501728A (en) | 1983-01-06 | 1985-02-26 | Technology Unlimited, Inc. | Masking of liposomes from RES recognition |
| WO1985001289A1 (en) | 1983-09-22 | 1985-03-28 | Fujisawa Pharmaceutical Co., Ltd. | Benzoxazolone and benzothiazolone derivatives and process for their preparation |
| US4591548A (en) | 1984-07-20 | 1986-05-27 | Minnesota Mining And Manufacturing Company | Cyan dye-forming couplers and photographic elements and processes |
| US4593102A (en) | 1984-04-10 | 1986-06-03 | A. H. Robins Company, Inc. | N-[(amino)alkyl]-1-pyrrolidine, 1-piperidine and 1-homopiperidinecarboxamides (and thiocarboxamides) with sulfur linked substitution in the 2, 3 or 4-position |
| JPS61129129A (en) | 1984-11-28 | 1986-06-17 | Kureha Chem Ind Co Ltd | Antitumor agent |
| EP0189069A2 (en) | 1985-01-18 | 1986-07-30 | Nissan Chemical Industries Ltd. | Pyrazolesulfonamide derivative, process for its production and herbicide containing it |
| DE3512630A1 (en) | 1985-04-06 | 1986-10-23 | Hoechst Ag, 6230 Frankfurt | METHOD FOR COLORING OR PRINTING CELLULOSE FIBERS OR CELLULOSE MIXED FIBERS |
| EP0246749A2 (en) | 1986-05-17 | 1987-11-25 | AgrEvo UK Limited | Triazole herbicides |
| US4775762A (en) | 1987-05-11 | 1988-10-04 | The Dow Chemical Company | Novel (1H-1,2,3-triazol-1-yl)pyridines |
| US4837028A (en) | 1986-12-24 | 1989-06-06 | Liposome Technology, Inc. | Liposomes with enhanced circulation time |
| US4849424A (en) | 1986-08-05 | 1989-07-18 | Nissin Shokuhin Kabushiki Kaisha | Pyrimidine derivatives |
| DE3813886A1 (en) | 1988-04-20 | 1989-11-02 | Schering Ag | 1-Triazinyl-1H-1,2,4-triazole-3-sulphonamides, processes for their preparation, and their use as herbicides, fungicides and plant growth regulators |
| EP0384228A1 (en) | 1989-02-21 | 1990-08-29 | Dainippon Pharmaceutical Co., Ltd. | Medicament for treating cerebral insufficiency diseases containing 2-(1-piperazinyl)-4-phenylcycloalkanopyrimidine derivatives, and process for the production thereof |
| EP0385237A2 (en) | 1989-03-03 | 1990-09-05 | Dainippon Pharmaceutical Co., Ltd. | 2-(1-piperazinyl)-4-phenylcycloalkanopyridine derivatives, processes for the production thereof, and pharmaceutical composition containing the same |
| US4959094A (en) | 1988-04-20 | 1990-09-25 | Schering Aktiengesellschaft | 1-chloropyrimidinyl-1H-1,2,4-triazole-3-sulphonamides as herbicides |
| US5019369A (en) | 1984-10-22 | 1991-05-28 | Vestar, Inc. | Method of targeting tumors in humans |
| JPH0499768A (en) | 1990-08-17 | 1992-03-31 | Dainippon Pharmaceut Co Ltd | 4-(4-phenylpyridin-2-yl)piperazine-1-oxide derivative |
| US5122530A (en) | 1990-06-29 | 1992-06-16 | Sumitomo Chemical Company, Limited | 1-pyridylimidazole derivative and its use |
| WO1992011761A1 (en) | 1990-12-31 | 1992-07-23 | Monsanto Company | Reducing pesticidal interactions in crops |
| US5180732A (en) | 1990-02-20 | 1993-01-19 | Sumitomo Chemical Company, Limited | 4-tert.-butyl imidazole derivative and use |
| US5220028A (en) | 1988-10-27 | 1993-06-15 | Nissan Chemical Industries, Ltd. | Halogeno-4-methylpyrazoles |
| WO1993013072A1 (en) | 1991-12-20 | 1993-07-08 | Italfarmaco S.P.A. | 5-isoquinolinesulfonamide derivatives as protein kinase inhibiting agents |
| US5252590A (en) | 1991-06-28 | 1993-10-12 | Sumitomo Chemical Company, Limited | 1-pyridylimidazole derivative |
| JPH0625177A (en) | 1992-07-09 | 1994-02-01 | Nissan Chem Ind Ltd | Pyrazole derivative and herbicide |
| EP0628551A1 (en) | 1993-06-14 | 1994-12-14 | Shionogi & Co., Ltd. | Semicyclic urea derivatives as antihistaminic agents |
| JPH07165708A (en) | 1993-09-10 | 1995-06-27 | Eisai Co Ltd | Bicyclic heterocycle-containing sulfonamide and sulfonic acid ester derivative |
| US5489591A (en) | 1992-02-28 | 1996-02-06 | Zenyaku Kogyo Kabushiki Kaisha | S-triazine derivative and remedy for estrogen-dependent disease containing said derivative as effective component |
| US5556866A (en) | 1993-04-28 | 1996-09-17 | Kabushiki Kaisha Hayashibara Seibutsu Kagaku Kenkyujo | Hair restorer and its preparation |
| WO1996030343A1 (en) | 1995-03-29 | 1996-10-03 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| FR2735127A1 (en) | 1995-06-09 | 1996-12-13 | Pf Medicament | NOVEL HETEROAROMATIC PIPERAZINES USEFUL AS MEDICAMENTS. |
| CA2235621A1 (en) | 1995-10-25 | 1997-05-01 | Schering Aktiengesellschaft | Imidazole derivatives and their use as nitrogen monoxide synthase inhibitors |
| WO1997028141A1 (en) | 1996-02-02 | 1997-08-07 | Pierre Fabre Medicament | Novel aromatic piperazines derived from substituted cycloazanes, method for preparing same, pharmaceutical compositions, and use thereof as drugs |
| WO1997028128A1 (en) | 1996-02-02 | 1997-08-07 | Zeneca Limited | Heterocyclic compounds useful as pharmaceutical agents |
| WO1997028129A1 (en) | 1996-02-02 | 1997-08-07 | Zeneca Limited | Aminoheterocyclic derivatives as antithrombotic or anticoagulant agents |
| JPH09291034A (en) | 1996-02-27 | 1997-11-11 | Yoshitomi Pharmaceut Ind Ltd | Condensed pyridine compound and its use as medicine |
| WO1997044322A1 (en) | 1996-05-20 | 1997-11-27 | Darwin Discovery Limited | Quinoline sulfonamides as tnf inhibitors and as pde-iv inhibitors |
| WO1998002434A1 (en) | 1996-07-13 | 1998-01-22 | Glaxo Group Limited | Fused heterocyclic compounds as protein tyrosine kinase inhibitors |
| WO1998003350A1 (en) | 1996-07-20 | 1998-01-29 | Golden Records Association International Corporation | Plastic card |
| US5807876A (en) | 1996-04-23 | 1998-09-15 | Vertex Pharmaceuticals Incorporated | Inhibitors of IMPDH enzyme |
| US5843485A (en) | 1996-06-28 | 1998-12-01 | Incoe Corporation | Valve-gate bushing for gas-assisted injection molding |
| WO1999016751A1 (en) | 1997-10-01 | 1999-04-08 | Merck Patent Gmbh | Benzamidine derivatives as factor xa inhibitors |
| JPH11158073A (en) | 1997-09-26 | 1999-06-15 | Takeda Chem Ind Ltd | Adenosine a3 antagonist |
| WO1999032463A1 (en) | 1997-12-22 | 1999-07-01 | Bayer Corporation | INHIBITION OF p38 KINASE USING SYMMETRICAL AND UNSYMMETRICAL DIPHENYL UREAS |
| WO1999047508A1 (en) | 1998-03-17 | 1999-09-23 | Astrazeneca Ab | Benzenesulfonamide-derivatives and their use as medicaments |
| EP0945446A1 (en) | 1996-11-14 | 1999-09-29 | Nissan Chemical Industries, Limited | Cyanoethylmelamine derivatives and process for producing the same |
| WO1999048490A1 (en) | 1998-03-24 | 1999-09-30 | Allos Therapeutics, Inc. | Allosteric inhibitors and activators of pyruvate kinase |
| US5962490A (en) | 1987-09-25 | 1999-10-05 | Texas Biotechnology Corporation | Thienyl-, furyl- and pyrrolyl-sulfonamides and derivatives thereof that modulate the activity of endothelin |
| US5965569A (en) | 1995-10-11 | 1999-10-12 | Medichem, S.A. | Polycyclic aminopyridine compounds which are acetylcholinesterase inhibitors, process for preparing them and their use |
| US5965559A (en) | 1994-09-26 | 1999-10-12 | Zeneca Limited | Aminoheterocyclic derivatives as antithrombotic or anticoagulant |
| US5984882A (en) | 1996-08-19 | 1999-11-16 | Angiosonics Inc. | Methods for prevention and treatment of cancer and other proliferative diseases with ultrasonic energy |
| WO1999062506A1 (en) | 1998-05-29 | 1999-12-09 | Astrazeneca Ab | Use of compounds for the elevation of pyruvate dehydrogenase activity |
| US6020357A (en) | 1996-12-23 | 2000-02-01 | Dupont Pharmaceuticals Company | Nitrogen containing heteroaromatics as factor Xa inhibitors |
| DE19841985A1 (en) | 1998-09-03 | 2000-03-09 | Schering Ag | New heterocyclic alkanesulfonic and alkane carboxylic acid derivatives are VEGF receptor blockers useful in treatment of e.g. psoriasis, rheumatoid arthritis, stroke, tumors and endometriosis |
| WO2000017202A1 (en) | 1998-09-18 | 2000-03-30 | Basf Aktiengesellschaft | 4-aminopyrrolopyrimidines as kinase inhibitors |
| US6106849A (en) | 1998-01-21 | 2000-08-22 | Dragoco Gerberding & Co. Ag | Water soluble dry foam personal care product |
| WO2000053596A2 (en) | 1999-03-08 | 2000-09-14 | Schering Corporation | Imidazole compounds as histamine h3 ligands |
| US6150356A (en) | 1998-09-01 | 2000-11-21 | Bristol-Myers Squibb Company | Potassium channel inhibitors and method |
| US6172005B1 (en) | 1997-03-11 | 2001-01-09 | E. I. Du Pont De Nemours And Company | Heteroaryl azole herbicides |
| WO2001007440A1 (en) | 1999-07-21 | 2001-02-01 | Boehringer Ingelheim Pharmaceuticals, Inc. | Imidazoimidazoles and triazoles as anti-inflammatory agents |
| WO2001016097A1 (en) | 1999-08-27 | 2001-03-08 | Sugen, Inc. | Phosphate mimics and methods of treatment using phosphatase inhibitors |
| WO2001019798A2 (en) | 1999-09-17 | 2001-03-22 | Cor Therapeutics Inc. | INHIBITORS OF FACTOR Xa |
| WO2001019788A2 (en) | 1999-09-17 | 2001-03-22 | Cor Therapeutics, Inc. | BENZAMIDES AND RELATED INHIBITORS OF FACTOR Xa |
| US6262113B1 (en) | 1996-03-20 | 2001-07-17 | Smithkline Beecham Corporation | IL-8 receptor antagonists |
| US6265588B1 (en) | 1997-01-28 | 2001-07-24 | Aventis Pharma Deutschland Gmbh | Isoxazole and crotonamide derivatives and their use as pharmaceuticals and diagnostics |
| US6274620B1 (en) | 1999-06-07 | 2001-08-14 | Biochem Pharma Inc. | Thiophene integrin inhibitors |
| WO2001064642A2 (en) | 2000-02-29 | 2001-09-07 | Cor Therapeutics, Inc. | Benzamides and related inhibitors of factor xa |
| US6359134B1 (en) | 1997-05-30 | 2002-03-19 | Takeda Chemical Industries, Ltd. | Sulfonamide derivatives, their production and use |
| US6399358B1 (en) | 1997-03-31 | 2002-06-04 | Thomas Jefferson University | Human gene encoding human chondroitin 6-sulfotransferase |
| JP2002193710A (en) | 2000-12-25 | 2002-07-10 | Kumiai Chem Ind Co Ltd | Pyrimidine or triazine derivative and fungicide for agriculture and horticulture |
| WO2002072077A2 (en) | 2001-03-13 | 2002-09-19 | Schebo Biotech Ag | Use of amino acids, amino acid analogues, sugar phosphates and sugar phosphate analogues for treatment of tumors, treatment of sepsis and immunosuppression |
| WO2002088101A2 (en) | 2001-04-27 | 2002-11-07 | Vertex Pharmaceuticals Incorporated | Inhibitors of bace |
| WO2002095063A1 (en) | 2001-05-23 | 2002-11-28 | MAX-PLANCK-Gesellschaft zur Förderung der Wissenschaften e.V. | Pyruvate-kinase as a novel target molecule |
| WO2002100822A1 (en) | 2001-06-11 | 2002-12-19 | Biovitrum Ab | Substituted sulfonamide compounds, process for their use as medicament for the treatment of cns disorders, obesity and type ii diabetes |
| WO2002102313A2 (en) | 2001-06-19 | 2002-12-27 | Bristol-Myers Squibb Company | Pyrimidine inhibitors of phosphodiesterase (pde) 7 |
| US6515013B2 (en) | 2000-07-13 | 2003-02-04 | Abbott Laboratories | 1,3-disubstituted and 1,3,3-trisubstituted pyrrolidines as histamine-3 receptor ligands and their therapeutic applications |
| US6525091B2 (en) | 2001-03-07 | 2003-02-25 | Telik, Inc. | Substituted diarylureas as stimulators for Fas-mediated apoptosis |
| WO2003016300A1 (en) | 2001-08-15 | 2003-02-27 | E. I. Du Pont De Nemours And Company | Ortho-substituted aryl amides for controlling invertebrate pests |
| WO2003016289A1 (en) | 2001-08-17 | 2003-02-27 | Ciba Specialty Chemicals Holding Inc. | Triazine derivatives and their use as sunscreens |
| WO2003022277A1 (en) | 2001-09-07 | 2003-03-20 | Bayer Healthcare Ag | Arylsulfonamide derivatives for use as ccr3 antagonists in the treatment of inflammatory and immunological disorders |
| US20030082877A1 (en) | 2001-06-18 | 2003-05-01 | Hitachi, Ltd. | Semiconductor integrated circuit device and a method of manufacturing the same |
| WO2003037252A2 (en) | 2001-10-30 | 2003-05-08 | Merck & Co., Inc. | Tyrosine kinase inhibitors |
| US20030109527A1 (en) | 1998-12-16 | 2003-06-12 | Smithkline Beecham Corporation | Hydroxy diphenyl urea sulfonamides as IL-8 receptor antagonists |
| US20030106381A1 (en) | 2000-03-08 | 2003-06-12 | Krouth Terrance F. | Hydraulic actuator piston measurement apparatus and method |
| WO2003062235A1 (en) | 2002-01-17 | 2003-07-31 | Eli Lilly And Company | Modulators of acetylcholine receptors |
| US20030158232A1 (en) | 2001-05-30 | 2003-08-21 | Cheng Peter T. | Substituted azole acid derivatives useful as antidiabetic and antiobesity agents and method |
| WO2003073999A2 (en) | 2002-03-01 | 2003-09-12 | Pintex Pharmaceuticals, Inc. | Pini-modulating compounds and methods of use thereof |
| WO2003076422A1 (en) | 2002-03-13 | 2003-09-18 | Janssen Pharmaceutica N.V. | Sulfonyl-derivatives as novel inhibitors of histone deacetylase |
| US20030187001A1 (en) | 1997-03-19 | 2003-10-02 | David Calderwood | 4-aminopyrrolopyrimidines as kinase inhibitors |
| WO2003093297A2 (en) | 2002-05-03 | 2003-11-13 | Exelixis, Inc. | Protein kinase modulators and methods of use |
| WO2004004730A2 (en) | 2002-07-06 | 2004-01-15 | Astex Technology Limited | 2-aminopyrazine derivatives as inhibitors of cyclin dependent kinases for the treatment of proliferative disorders |
| WO2004009562A1 (en) | 2002-07-18 | 2004-01-29 | Janssen Pharmaceutica, Nv | Substituted triazine kinase inhibitors |
| WO2004014851A2 (en) | 2002-08-09 | 2004-02-19 | Merck & Co., Inc. | Tyrosine kinase inhibitors |
| US20040048283A1 (en) | 2000-11-28 | 2004-03-11 | Bernard Pau | Novel method for screening bacterial transcription modulators |
| US20040067234A1 (en) | 2002-07-11 | 2004-04-08 | Paz Einat | Isocitrate dehydrogenase and uses thereof |
| JP2004107220A (en) | 2002-09-13 | 2004-04-08 | Mitsubishi Pharma Corp | TNF-alpha PRODUCTION INHIBITOR |
| US6723730B2 (en) | 2000-07-20 | 2004-04-20 | Neurogen Corporation | Capsaicin receptor ligands |
| WO2004037251A1 (en) | 2002-10-24 | 2004-05-06 | Sterix Limited | Inhibitors of 11-beta-hydroxy steroid dehydrogenase type 1 and type 2 |
| WO2004046120A2 (en) | 2002-11-15 | 2004-06-03 | Vertex Pharmaceuticals Incorporated | Diaminotriazoles useful as inhibitors of protein kinases |
| WO2004050033A2 (en) | 2002-12-02 | 2004-06-17 | Arqule, Inc. | Method of treating cancers |
| WO2004074438A2 (en) | 2003-02-14 | 2004-09-02 | Smithkline Beecham Corporation | Ccr8 antagonists |
| WO2004073619A2 (en) | 2003-02-14 | 2004-09-02 | Smithkline Beecham Corporation | Ccr8 antagonists |
| WO2004074288A1 (en) | 2003-02-19 | 2004-09-02 | F. Hoffmann-La Roche Ag | Sulfonamide substituted xanthine derivatives for use as pepck inhibitors |
| US20040198979A1 (en) | 2001-05-07 | 2004-10-07 | Dashyant Dhanak | Sulfonamides |
| WO2004089470A2 (en) | 2003-04-11 | 2004-10-21 | Novo Nordisk A/S | New amide derivatives and pharmaceutical use thereof |
| US6818631B1 (en) | 2003-08-15 | 2004-11-16 | Nippon Soda Co. Ltd. | Fungicidal pyrimidine derivatives |
| WO2004110375A2 (en) | 2003-06-06 | 2004-12-23 | Merck & Co., Inc. | Combination therapy for the treatment of diabetes |
| US6878196B2 (en) | 2002-01-15 | 2005-04-12 | Fuji Photo Film Co., Ltd. | Ink, ink jet recording method and azo compound |
| WO2005035507A2 (en) | 2003-10-10 | 2005-04-21 | Bayer Pharmaceuticals Corporation | 4-aminopyrimidine derivatives for treatment of hyperproliferative disorders |
| WO2005060956A1 (en) | 2003-12-12 | 2005-07-07 | University Of Maryland, Baltimore | IMMUNOMODULATORY COMPOUNDS THAT TARGET AND INHIBIT THE pY+3 BINDING SITE OF TYROSENE KINASE p56 LCK SH2 DOMAIN |
| WO2005065691A1 (en) | 2003-12-24 | 2005-07-21 | Scios, Inc. | Treatment of malignant gliomas with tgf-beta inhibitors |
| WO2005066126A1 (en) | 2003-12-23 | 2005-07-21 | Eli Lilly And Company | Cb1 modulator compounds |
| US20050176675A1 (en) | 2004-02-10 | 2005-08-11 | Philipe Gorny | Stable and active complexes of adenosine and adenosine phosphates with aminoalcohols for the treatment of pulmonary artery hypertension, cardiac failure and other diseases |
| WO2005072642A1 (en) | 2004-01-29 | 2005-08-11 | Colgate-Palmolive Company | Light-based toothbrush |
| EP1586558A2 (en) | 1995-01-20 | 2005-10-19 | G.D. Searle LLC. | Bis-sulfonamide hydroxyethylamino retroviral protease inhinitors |
| WO2005117591A2 (en) | 2004-05-28 | 2005-12-15 | Andrx Labs Llc | Novel pharmaceutical formulation containing a biguanide and an angiotensin antagonist |
| WO2005120474A2 (en) | 2004-06-05 | 2005-12-22 | K.U.Leuven Research And Development | Phophodiesterase 10a inhibitors to amplify the action of glp1-mimetics or dpp-iv inhibitors in diabetes |
| US6979675B2 (en) | 2003-01-10 | 2005-12-27 | Threshold Pharmaceuticals, Inc. | Treatment of cancer with 2-deoxyglucose |
| WO2006004195A1 (en) | 2004-07-02 | 2006-01-12 | Tanabe Seiyaku Co., Ltd. | Piperidine compound and process for preparing the same |
| WO2006016062A1 (en) | 2004-07-12 | 2006-02-16 | Laurent Schwartz | Multitherapy against cancer |
| WO2006033628A1 (en) | 2004-09-24 | 2006-03-30 | Astrazeneca Ab | Benzimidazole derivatives, compositions containing them, preparation thereof and uses thereof |
| WO2006034341A2 (en) | 2004-09-20 | 2006-03-30 | Xenon Pharmaceuticals Inc. | Pyridazine derivatives for inhibiting human stearoyl-coa-desaturase |
| WO2006038594A1 (en) | 2004-10-04 | 2006-04-13 | Ono Pharmaceutical Co., Ltd. | N-type calcium channel inhibitor |
| WO2006043950A1 (en) | 2004-10-20 | 2006-04-27 | Smithkline Beecham Corporation | Il-8 receptor antagonists |
| WO2006052190A1 (en) | 2004-11-11 | 2006-05-18 | Astrazeneca Ab | Indazole sulphonamide derivatives |
| WO2006063294A2 (en) | 2004-12-09 | 2006-06-15 | Kalypsys, Inc. | Novel inhibitors of histone deacetylase for the treatment of disease |
| WO2006070198A1 (en) | 2004-12-30 | 2006-07-06 | Astex Therapeutics Limited | Pyrazole derivatives as that modulate the activity of cdk, gsk and aurora kinases |
| WO2006077821A1 (en) | 2005-01-19 | 2006-07-27 | Dainippon Sumitomo Pharma Co., Ltd. | Aromatic sulfone compound as aldosterone receptor modulator |
| WO2006079791A1 (en) | 2005-01-25 | 2006-08-03 | Astrazeneca Ab | Chemical compounds |
| WO2006117762A2 (en) | 2005-05-03 | 2006-11-09 | Ranbaxy Laboratories Limited | Antimicrobial agents |
| WO2006122546A1 (en) | 2005-05-18 | 2006-11-23 | Forschungsverbund Berlin E.V. | Non-peptidic inhibitors of akap/pka interaction |
| WO2007003934A2 (en) | 2005-07-04 | 2007-01-11 | Sterix Limited | 17beta-hydr0xyster0id dehydrogenase type 3 (17beta-hsd3 ) inhibitors |
| US20070032418A1 (en) | 2003-02-25 | 2007-02-08 | Ambion, Inc | Small-molecule inhibitors of angiogenin and rnases and in vivo and in vitro methods of using same |
| US20070032520A1 (en) | 2003-07-30 | 2007-02-08 | Laboratorios Del Dr. Esteve S.A | Indol-5-yl sulfonamide derivatives, their preparation and their use 5-ht-6 as modulators |
| WO2007023186A1 (en) | 2005-08-26 | 2007-03-01 | Laboratoires Serono S.A. | Pyrazine derivatives and use as pi3k inhibitors |
| US20070127505A1 (en) | 2005-12-02 | 2007-06-07 | Nokia Corporation | Group communication |
| US7235664B2 (en) | 2001-06-12 | 2007-06-26 | Sanofi-Aventis Deutschland Gmbh | Anthranilamides with heteroarylsulfonyl side chain, process of preparation, and use |
| US7241759B2 (en) | 2003-03-28 | 2007-07-10 | Janssen Pharmaceutica N.V. | Benzo[1,2,5]thiadiazole compounds |
| JP2007238458A (en) | 2006-03-06 | 2007-09-20 | D Western Therapeutics Institute Inc | Novel isoquinoline derivative and medicine containing the same |
| WO2007117465A2 (en) | 2006-03-31 | 2007-10-18 | Abbott Laboratories | Indazole compounds |
| US20070244088A1 (en) | 2004-06-28 | 2007-10-18 | Astrazeneca Ab | New Pyridine Analogues II |
| WO2007127505A2 (en) | 2006-01-20 | 2007-11-08 | Smithkline Beecham Corporation | Use of sulfonamide derivatives in the treatment of disorders of the metabolism and the nervous system |
| US20080004269A1 (en) | 2004-11-04 | 2008-01-03 | Yuelian Xu | Pyrazolylmethy Heteroaryl Derivatives |
| WO2008019139A2 (en) | 2006-08-04 | 2008-02-14 | Beth Israel Deaconess Medical Center | Inhibitors of pyruvate kinase and methods of treating disease |
| US20080044833A1 (en) | 2004-11-16 | 2008-02-21 | Connors Timothy D | Diagnostic PKM2 methods and compositions |
| US20080051414A1 (en) | 2003-10-14 | 2008-02-28 | Arizona Board Of Regents On Behalf Of The University Of Arizona | Protein Kinase Inhibitors |
| WO2008024284A2 (en) | 2006-08-21 | 2008-02-28 | Merck & Co., Inc. | Sulfonylated piperazines as cannabinoid-1 receptor modulators |
| WO2008026658A1 (en) | 2006-09-01 | 2008-03-06 | Otsuka Chemical Co., Ltd. | N-pyridylpiperidine compound, method for producing the same, and pest control agent |
| US7358262B2 (en) | 2003-01-29 | 2008-04-15 | Whitehead Institute For Biomedical Research | Identification of genotype-selective anti-tumor agents |
| WO2008043381A1 (en) | 2006-10-12 | 2008-04-17 | Fluid Automation Systems S.A. | Rocker valve mechanism and rocker valve |
| WO2008047198A1 (en) | 2006-10-16 | 2008-04-24 | Pfizer Products Inc. | Therapeutic pyrazolyl thienopyridines |
| WO2008052190A2 (en) | 2006-10-26 | 2008-05-02 | Flynn Gary A | Aquaporin modulators and methods of using them for the treatment of edema and fluid imbalance |
| WO2008050168A1 (en) | 2006-10-27 | 2008-05-02 | Richter Gedeon Nyrt. | New sulfonamide derivatives as bradykinin antagonists |
| JP4099768B2 (en) | 2003-11-10 | 2008-06-11 | 富士電機デバイステクノロジー株式会社 | Electrophotographic photosensitive member and method for determining presence or absence of interference fringes resulting from electrophotographic photosensitive member |
| WO2008070661A1 (en) | 2006-12-04 | 2008-06-12 | Neurocrine Biosciences, Inc. | Substituted pyrimidines as adenosine receptor antagonists |
| WO2008073670A2 (en) | 2006-12-08 | 2008-06-19 | Millennium Pharmaceuticals, Inc. | Unit dose formulations and methods of treating thrombosis with an oral factor xa inhibitor |
| WO2008076883A2 (en) | 2006-12-15 | 2008-06-26 | Abraxis Bioscience, Inc. | Triazine derivatives and their therapeutical applications |
| US20080214495A1 (en) | 2005-07-08 | 2008-09-04 | Astrazeneca Ab | Heterocyclic Sulfonamide Derivatives as Inhibitors of Factor Xa |
| WO2008131547A1 (en) | 2007-04-30 | 2008-11-06 | Prometic Biosciences Inc. | 'triazine derivatives, compositions containing such derivatives, and methods of treatment of cancer and autoimmune diseases using such derivatives' |
| US7452888B2 (en) | 2002-03-27 | 2008-11-18 | Glaxo Group Limited | Quinoline derivatives and their use as 5-ht6 ligands |
| WO2008154026A1 (en) | 2007-06-11 | 2008-12-18 | Miikana Therapeutics, Inc. | Substituted pyrazole compounds |
| WO2009012430A1 (en) | 2007-07-18 | 2009-01-22 | Janssen Pharmaceutica N.V. | Sulfonamides as trpm8 modulators |
| WO2009013126A1 (en) | 2007-07-20 | 2009-01-29 | Nerviano Medical Sciences S.R.L. | Substituted indazole derivatives active as kinase inhibitors |
| WO2009016410A2 (en) | 2007-07-31 | 2009-02-05 | Astrazeneca Ab | Chemical compounds 831 |
| US20090048227A1 (en) | 2005-12-22 | 2009-02-19 | Chakravarty Pasun K | Substituted-1-Phthalazinamines As Vr- 1 Antagonists |
| WO2009025781A1 (en) | 2007-08-16 | 2009-02-26 | Beth Israel Deaconess Medical Center | Activators of pyruvate kinase m2 and methods of treating disease |
| US20090054453A1 (en) | 2006-03-17 | 2009-02-26 | Lilian Alcaraz | Novel Tetralins as 5-HT6 Modulators |
| US20090093526A1 (en) | 1997-12-22 | 2009-04-09 | Scott Miller | Inhibition of p38 kinase using symmetrical and unsymmetrical diphenyl ureas |
| US7524848B2 (en) | 2006-03-23 | 2009-04-28 | Amgen Inc. | Diaza heterocyclic amide compounds and their uses |
| WO2009053102A1 (en) | 2007-10-26 | 2009-04-30 | Syngenta Participations Ag | Novel imidazole derivatives |
| US20090163508A1 (en) | 2007-10-10 | 2009-06-25 | Takeda Pharmaceutical Company Limited | Amide compound |
| US20090163545A1 (en) | 2007-12-21 | 2009-06-25 | University Of Rochester | Method For Altering The Lifespan Of Eukaryotic Organisms |
| US7572913B2 (en) | 2004-02-24 | 2009-08-11 | Bioaxone Therapeutique Inc. | 4-substituted piperidine derivatives |
| US7595322B2 (en) | 2003-03-27 | 2009-09-29 | Cytokinetics, Inc. | Heterocyclic sulfonamides as modulators of cardiac sarcomeres |
| WO2009118567A2 (en) | 2008-03-26 | 2009-10-01 | The University Of Nottingham | Pyrimidines, triazines and their use as pharmaceutical agents |
| JP2009237115A (en) | 2008-03-26 | 2009-10-15 | Fuji Xerox Co Ltd | Electrophotographic photoreceptor, image forming apparatus, process cartridge, and image forming method |
| WO2009126863A2 (en) | 2008-04-11 | 2009-10-15 | Genentech, Inc. | Pyridyl inhibitors of hedgehog signalling |
| US20090270454A1 (en) | 2007-04-23 | 2009-10-29 | Weingarten M David | Sulfonamide containing compounds for treatment of inflammatory disorders |
| US7615553B2 (en) | 2002-03-13 | 2009-11-10 | Janssen Pharmaceutica N.V. | Inhibitors of histone deacetylase |
| CN101575408A (en) | 2008-05-09 | 2009-11-11 | Mca技术有限公司 | Polytriazinyl compounds as flame retardants and light stabilizers |
| WO2009150248A1 (en) | 2008-06-13 | 2009-12-17 | Cytomics Systems | Compounds which can be used for the treatment of cancers |
| WO2010007756A1 (en) | 2008-07-14 | 2010-01-21 | 塩野義製薬株式会社 | Pyridine derivative having ttk inhibition activity |
| WO2010023445A1 (en) | 2008-08-29 | 2010-03-04 | Xention Limited | Novel potassium channel blocker |
| WO2010028099A1 (en) | 2008-09-03 | 2010-03-11 | The Johns Hopkins University | Genetic alterations in isocitrate dehydrogenase and other genes in malignant glioma |
| JP2010079130A (en) | 2008-09-29 | 2010-04-08 | Fuji Xerox Co Ltd | Electrophotographic photoreceptor, process cartridge, and image forming apparatus |
| WO2010042867A2 (en) | 2008-10-09 | 2010-04-15 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Activators of human pyruvate kinase |
| US20100105657A1 (en) | 2005-07-05 | 2010-04-29 | Astrazeneca Ab | Compounds, Process for their Preparation, Intermediates, Pharmaceutical Compositions and their use in the Treatment of 5-HT6 Mediated Disorders such as Alzheimer's Disease, Cognitive Disorders, Cognitive Impairment Associated with Schizophrenia, Obesity and Parkinson's Disease |
| US20100144722A1 (en) | 2008-09-03 | 2010-06-10 | Dr. Reddy's Laboratories Ltd. | Novel heterocyclic compounds as gata modulators |
| US20100179150A1 (en) | 2005-02-18 | 2010-07-15 | Astrazeneca Ab | Antibacterial piperdine derivatives |
| JP2010181540A (en) | 2009-02-04 | 2010-08-19 | Fuji Xerox Co Ltd | Electrophotographic photoreceptor, process cartridge and image forming apparatus |
| WO2010105243A1 (en) | 2009-03-13 | 2010-09-16 | Agios Pharmaceuticals, Inc. | Methods and compositions for cell-proliferation-related disorders |
| WO2010118063A2 (en) | 2009-04-06 | 2010-10-14 | Agios Pharmaceuticals, Inc. | Therapeutic compositions and related methods of use |
| US7820654B2 (en) | 2004-09-23 | 2010-10-26 | Dr. Reddy's Laboratories Ltd. | Pyrimidine compounds, process for their preparation and compositions containing them |
| US20100273808A1 (en) | 2008-11-21 | 2010-10-28 | Millennium Pharmaceticals, Inc. | Lactate salt of 4-[6-methoxy-7-(3-piperidin-1-yl-propoxy)quinazolin-4-yl]piperazine-1-carboxylic acid(4-isopropoxyphenyl)-amide and pharmaceutical compositions thereof for the treatment of cancer and other diseases or disorders |
| WO2010129596A1 (en) | 2009-05-04 | 2010-11-11 | Agios Pharmaceuticals, Inc. | Pmk2 modulators for use in the treatment of cancer |
| WO2010130638A1 (en) | 2009-05-14 | 2010-11-18 | Evotec Ag | Sulfonamide compounds, pharmaceutical compositions and uses thereof |
| WO2010144404A1 (en) | 2009-06-09 | 2010-12-16 | Abraxis Bioscience, Llc | Pyridil-triazine inhibitors of hedgehog signaling |
| WO2010144338A1 (en) | 2009-06-08 | 2010-12-16 | Abraxis Bioscience, Llc | Triazine derivatives and their therapeutical applications |
| US20100331307A1 (en) | 2009-06-29 | 2010-12-30 | Salituro Francesco G | Therapeutic compounds and compositions |
| WO2011002816A1 (en) | 2009-06-29 | 2011-01-06 | Agios Pharmaceuticals, Inc. | Therapeutic compositions and related methods of use |
| WO2011032169A2 (en) | 2009-09-14 | 2011-03-17 | Phusis Therapeutics Inc. | Pharmaceutical compositions and formulations including inhibitors of the pleckstrin homology domain and methods for using same |
| WO2011050210A1 (en) | 2009-10-21 | 2011-04-28 | Agios Pharmaceuticals, Inc. | Methods and compositions for cell-proliferation-related disorders |
| WO2011047432A1 (en) | 2009-10-22 | 2011-04-28 | Fibrotech Therapeutics Pty Ltd | Fused ring analogues of anti-fibrotic agents |
| WO2011072174A1 (en) | 2009-12-09 | 2011-06-16 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds for use in the treatment of cancer characterized as having an idh mutation |
| JP4753336B2 (en) | 2001-09-04 | 2011-08-24 | 日本化薬株式会社 | Novel allyl compound and process for producing the same |
| WO2011109441A1 (en) | 2010-03-01 | 2011-09-09 | Myrexis, Inc. | Compounds and therapeutic uses thereof |
| US20110224252A1 (en) | 2007-10-26 | 2011-09-15 | Syngenta Crop Protection, Inc. | Novel imidazole derivatives |
| WO2011137089A1 (en) | 2010-04-29 | 2011-11-03 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Activators of human pyruvate kinase |
| US8058313B2 (en) | 2004-06-24 | 2011-11-15 | Temple University—Of the Commonwealth System of Higher Education | Alpha, beta-unsaturated sulfones, sulfoxides, sulfonimides, sulfinimides, acylsulfonamides and acylsulfinamides and therapeutic uses thereof |
| US20110312931A1 (en) | 2010-06-04 | 2011-12-22 | Albany Molecular Research, Inc. | Glycine transporter-1 inhibitors, methods of making them, and uses thereof |
| WO2012009678A1 (en) | 2010-07-16 | 2012-01-19 | Agios Pharmaceuticals, Inc. | Therapeutically active compositions and their method of use |
| US8133900B2 (en) | 2005-11-01 | 2012-03-13 | Targegen, Inc. | Use of bi-aryl meta-pyrimidine inhibitors of kinases |
| WO2012069503A1 (en) | 2010-11-23 | 2012-05-31 | Pierre Fabre Medicament | Derivatives of heteroarylsulfonamides, their preparation and their application in human therapy |
| WO2012074999A1 (en) | 2010-11-29 | 2012-06-07 | Galleon Pharmaceuticals, Inc. | Novel compounds as respiratory stimulants for treatment of breathing control disorders or diseases |
| WO2012092442A1 (en) | 2010-12-29 | 2012-07-05 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| US20120202818A1 (en) | 2009-06-09 | 2012-08-09 | California Capital Equity, Llc | Ureidophenyl substituted triazine derivatives and their therapeutical applications |
| CN102659765A (en) | 2011-12-31 | 2012-09-12 | 沈阳药科大学 | Pyrimidine and triazine compound preparation method and application |
| US8278038B2 (en) | 2005-06-08 | 2012-10-02 | Millennium Pharmaceuticals, Inc. | Methods for the identification, assessment, and treatment of patients with cancer therapy |
| WO2012151451A1 (en) | 2011-05-03 | 2012-11-08 | Agios Pharmaceuticals, Inc. | Pyruvate kinase activators for use in therapy |
| WO2012151452A1 (en) | 2011-05-03 | 2012-11-08 | Agios Pharmaceuticals, Inc | Pyruvate kinase activators for use in therapy |
| WO2012160034A1 (en) | 2011-05-24 | 2012-11-29 | Bayer Intellectual Property Gmbh | 4-aryl-n-phenyl-1,3,5-triazin-2-amines containing a sulfoximine group |
| WO2012171506A1 (en) | 2011-06-17 | 2012-12-20 | Agios Pharmaceuticals, Inc. | Compounds, their pharmaceutical compositions and their uses as idh1 mutants inhibitors for treating cancers |
| US20130109643A1 (en) | 2010-05-10 | 2013-05-02 | The Johns Hopkins University | Metabolic inhibitor against tumors having an idh mutation |
| US8465673B2 (en) | 2009-09-30 | 2013-06-18 | Fujifilm Corporation | Humidity dependence improver for polymer film, polymer film, protective film for polarizer, retardation film, polarizer, and liquid crystal display device |
| WO2013102431A1 (en) | 2012-01-06 | 2013-07-11 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| WO2013107291A1 (en) | 2012-01-19 | 2013-07-25 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| WO2013107405A1 (en) | 2012-01-19 | 2013-07-25 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| US20130197106A1 (en) | 2010-04-01 | 2013-08-01 | Agios Pharmaceuticals, Inc | Methods of identifying a candidate compound |
| WO2013133367A1 (en) | 2012-03-09 | 2013-09-12 | カルナバイオサイエンス株式会社 | Novel triazine derivative |
| US8603469B2 (en) | 2002-12-16 | 2013-12-10 | Genmab A/S | Methods of treating cancer with human monoclonal antibodies against interleukin 8 |
| WO2014015422A1 (en) | 2012-07-27 | 2014-01-30 | Ontario Institute For Cancer Research | Cellulose-based nanoparticles for drug delivery |
| US8652534B2 (en) | 2009-10-14 | 2014-02-18 | Berry Pharmaceuticals, LLC | Compositions and methods for treatment of mammalian skin |
| US8673891B2 (en) | 2009-02-06 | 2014-03-18 | Nippon Shinyaku Co., Ltd. | Aminopyrazine derivative and medicine |
| JP2014509458A (en) | 2010-10-20 | 2014-04-17 | ヨタ デバイセズ アイピーアール リミテッド | Portable device |
| JP2014509459A (en) | 2011-02-04 | 2014-04-17 | 日本電気株式会社 | Radio communication system, base station apparatus, radio resource control method, and program |
| US8735392B2 (en) | 2010-10-21 | 2014-05-27 | Biomarin Pharmaceutical Inc. | Crystalline (8S,9R)-5-fluoro-8-(4-fluorophenyl)-9-(1-methyl-1H-1,2,4-triazol-5-yl)-8,9-dihydro-2H-pyrido[4,3,2-de]phthalazin-3(7H)-one tosylate salt |
| US20140213580A1 (en) | 2011-06-17 | 2014-07-31 | Agios Pharmaceuticals, Inc | Therapeutically active compositions and their methods of use |
| US20140249150A1 (en) | 2011-10-13 | 2014-09-04 | Agios Pharmaceuticals, Inc | Activators of pyruvate kinase m2 and methods of treating disease |
| US8883438B2 (en) | 2009-10-21 | 2014-11-11 | Agios Pharmaceuticals, Inc. | Method for diagnosing cell proliferation disorders having a neoactive mutation at residue 97 of isocitrate dehydrogenase 1 |
| US20150031627A1 (en) | 2013-07-25 | 2015-01-29 | Agios Pharmaceuticals, Inc | Therapeutically active compounds and their methods of use |
| US9150505B2 (en) * | 2010-03-04 | 2015-10-06 | Ajinomoto Co., Inc. | Prophylactic or therapeutic agent for diabetes or obesity |
| US9221792B2 (en) | 2010-12-17 | 2015-12-29 | Agios Pharmaceuticals, Inc | N-(4-(azetidine-1-carbonyl) phenyl)-(hetero-) arylsulfonamide derivatives as pyruvate kinase M2 (PMK2) modulators |
| US9328077B2 (en) | 2010-12-21 | 2016-05-03 | Agios Pharmaceuticals, Inc | Bicyclic PKM2 activators |
| US9487815B2 (en) | 2012-03-01 | 2016-11-08 | Dkfz Deutsches Krebsforschungszentrum | Means and methods for the determination of (D)-2-hydroxyglutarate (D2HG) or (D)-2-hydroxyadipic acid |
| US9579324B2 (en) | 2013-07-11 | 2017-02-28 | Agios Pharmaceuticals, Inc | Therapeutically active compounds and their methods of use |
| US10202339B2 (en) | 2012-10-15 | 2019-02-12 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| US20200030322A1 (en) | 2015-06-11 | 2020-01-30 | Agios Pharmaceuticals, Inc. | Methods of using pyruvate kinase activators |
| US11254652B2 (en) * | 2017-11-22 | 2022-02-22 | Agios Pharmaceuticals, Inc. | Crystalline forms of N-(4-(4-(cyclopropymethyl)piperazine-1-carbonyl)phenyl)quinoline-8-sulfonamide |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4643759A (en) | 1984-07-13 | 1987-02-17 | E. I. Du Pont De Nemours And Company | Herbicidal sulfonamides |
| CN1164232A (en) * | 1994-09-26 | 1997-11-05 | 曾尼卡有限公司 | Aminoheterocyclic derivatives as antithrombotic or anticoagulant agents |
| RU2126001C1 (en) * | 1995-01-10 | 1999-02-10 | Самдзин Фармасьютикал Ко., Лтд | Piperazine derivatives and pharmaceutical composition on their basis |
| JPH1158073A (en) | 1997-08-12 | 1999-03-02 | Ishikawajima Harima Heavy Ind Co Ltd | Device for welding hanging piece |
| WO2003066055A1 (en) * | 2002-02-04 | 2003-08-14 | F. Hoffmann-La Roche Ag | Quinoline derivatives as npy antagonists |
| EP2059809B1 (en) | 2006-08-30 | 2014-07-23 | Metanomics GmbH | Means and method for diagnosing hemolytic anemia |
-
2010
- 2010-06-29 PT PT107946683T patent/PT2448582T/en unknown
- 2010-06-29 NZ NZ622505A patent/NZ622505A/en unknown
- 2010-06-29 IN IN471DEN2012 patent/IN2012DN00471A/en unknown
- 2010-06-29 SG SG2011097367A patent/SG177434A1/en unknown
- 2010-06-29 DK DK10794668.3T patent/DK2448582T3/en active
- 2010-06-29 TW TW104100537A patent/TWI598337B/en active
- 2010-06-29 TW TW106118093A patent/TWI649313B/en active
- 2010-06-29 PL PL10794668T patent/PL2448582T3/en unknown
- 2010-06-29 BR BRPI1011587-0A patent/BRPI1011587B1/en active IP Right Grant
- 2010-06-29 KR KR1020177001949A patent/KR101850813B1/en active IP Right Grant
- 2010-06-29 KR KR1020127002311A patent/KR101712035B1/en active IP Right Grant
- 2010-06-29 CA CA2766882A patent/CA2766882C/en active Active
- 2010-06-29 WO PCT/US2010/040486 patent/WO2011002817A1/en active Application Filing
- 2010-06-29 ES ES10794668.3T patent/ES2632954T3/en active Active
- 2010-06-29 EP EP17000407.1A patent/EP3241554B1/en active Active
- 2010-06-29 US US12/826,630 patent/US8785450B2/en not_active Ceased
- 2010-06-29 MX MX2012000256A patent/MX2012000256A/en active IP Right Grant
- 2010-06-29 EP EP10794668.3A patent/EP2448582B1/en active Active
- 2010-06-29 TW TW107138088A patent/TWI691493B/en active
- 2010-06-29 AR ARP100102326A patent/AR077292A1/en active IP Right Grant
- 2010-06-29 PE PE2011002173A patent/PE20120693A1/en active IP Right Grant
- 2010-06-29 AU AU2010266386A patent/AU2010266386C1/en active Active
- 2010-06-29 EP EP20153827.9A patent/EP3708169A1/en active Pending
- 2010-06-29 SG SG10201811480WA patent/SG10201811480WA/en unknown
- 2010-06-29 HU HUE10794668A patent/HUE032969T2/en unknown
- 2010-06-29 UA UAA201200823A patent/UA107667C2/en unknown
- 2010-06-29 SG SG10201403696UA patent/SG10201403696UA/en unknown
- 2010-06-29 CA CA2944788A patent/CA2944788C/en active Active
- 2010-06-29 JP JP2012518591A patent/JP5856052B2/en active Active
- 2010-06-29 SI SI201031488T patent/SI2448582T1/en unknown
- 2010-06-29 CN CN201080037700.8A patent/CN102481300B/en active Active
- 2010-06-29 LT LTEP10794668.3T patent/LT2448582T/en unknown
- 2010-06-29 TW TW99121186A patent/TWI472522B/en active
- 2010-06-29 MY MYPI2011006339A patent/MY191825A/en unknown
- 2010-06-29 CN CN201510106447.1A patent/CN104817498B/en active Active
- 2010-06-29 NZ NZ597379A patent/NZ597379A/en not_active IP Right Cessation
- 2010-06-29 CR CR20170071A patent/CR20170071A/en unknown
- 2010-06-29 RU RU2012102922/04A patent/RU2561132C2/en active
-
2011
- 2011-12-27 CO CO11179296A patent/CO6480955A2/en active IP Right Grant
- 2011-12-29 EC EC2011011567A patent/ECSP11011567A/en unknown
- 2011-12-29 IL IL217277A patent/IL217277A/en active IP Right Grant
- 2011-12-29 CL CL2011003350A patent/CL2011003350A1/en unknown
-
2012
- 2012-01-04 ZA ZA2012/00054A patent/ZA201200054B/en unknown
- 2012-01-05 NI NI201200003A patent/NI201200003A/en unknown
- 2012-01-09 CR CR20120001A patent/CR20120001A/en unknown
-
2014
- 2014-05-23 US US14/286,088 patent/US20140323467A1/en not_active Abandoned
- 2014-11-04 JP JP2014224372A patent/JP2015028083A/en not_active Withdrawn
-
2017
- 2017-01-23 US US15/412,976 patent/US10029987B2/en active Active
- 2017-06-23 HR HRP20170955TT patent/HRP20170955T1/en unknown
- 2017-06-28 CY CY20171100682T patent/CY1119443T1/en unknown
-
2018
- 2018-06-18 US US16/010,717 patent/US20190241523A1/en not_active Abandoned
-
2019
- 2019-01-09 US US16/243,247 patent/US10988448B2/en active Active
-
2020
- 2020-12-22 US US17/131,562 patent/USRE49582E1/en active Active
-
2021
- 2021-03-08 US US17/195,243 patent/US11866411B2/en active Active
-
2023
- 2023-04-27 NO NO2023017C patent/NO2023017I1/en unknown
- 2023-04-27 LT LTPA2023513C patent/LTPA2023513I1/lt unknown
- 2023-04-28 FR FR23C1017C patent/FR23C1017I2/en active Active
- 2023-05-02 NL NL301230C patent/NL301230I2/en unknown
- 2023-05-02 HU HUS2300018C patent/HUS2300018I1/en unknown
- 2023-05-02 FI FIC20230017C patent/FIC20230017I1/en unknown
Patent Citations (338)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2390529A (en) | 1942-02-03 | 1945-12-11 | Ernst A H Friedheim | Hydrazino-1,3,5-triazino derivatives of substituted phenylarsenic compounds |
| US3046122A (en) | 1949-07-23 | 1962-07-24 | Azoplate Corp | Process of making printing plates and light sensitive material suitable for use therein |
| US3097210A (en) | 1959-04-06 | 1963-07-09 | Merck & Co Inc | Substituted pyrazole sulfonamides |
| GB1274436A (en) | 1970-06-09 | 1972-05-17 | Wolfen Filmfab Veb | Process for the sensitization of photographic silver chloride and silver chlorobromide emulsions that may contain colour couplers |
| US3755322A (en) | 1970-07-15 | 1973-08-28 | Ciba Geigy Corp | Diamino-s-triazines |
| US3867383A (en) | 1971-03-29 | 1975-02-18 | Ciba Geigy Corp | Monoanthranilatoanilino-s-triazines |
| US4084053A (en) | 1974-06-21 | 1978-04-11 | Ciba-Geigy Corporation | Polycyclic compounds |
| US3998828A (en) | 1975-01-31 | 1976-12-21 | Pfizer Inc. | 4-[2-(1,3-Dialkyl-1,2,3,4-tetra-hydropyrimidine-2,4-dione-5-carboxamido)ethyl]-1-piperidine sulfonamide |
| US4235871A (en) | 1978-02-24 | 1980-11-25 | Papahadjopoulos Demetrios P | Method of encapsulating biologically active materials in lipid vesicles |
| EP0022958A1 (en) | 1979-07-14 | 1981-01-28 | Bayer Ag | Urea derivatives for use in the treatment of fat metabolism disorders |
| US4315940A (en) | 1979-12-01 | 1982-02-16 | Hoechst Aktiengesellschaft | Antidiabetic 1-piperidine-sulfonylureas |
| DE3314663A1 (en) | 1982-04-27 | 1983-10-27 | Nippon Kayaku K.K., Tokyo | METHOD FOR DYEING CELLULOSE FIBERS OR CELLULOSE MIXED FIBERS AND THE DYES THAT CAN BE USED FOR THIS |
| US4474599A (en) | 1982-07-14 | 1984-10-02 | The Dow Chemical Company | 1-(Pyridyl)-1H-1,2,3-triazole derivatives, and use as herbicidal agents |
| US4501728A (en) | 1983-01-06 | 1985-02-26 | Technology Unlimited, Inc. | Masking of liposomes from RES recognition |
| WO1985001289A1 (en) | 1983-09-22 | 1985-03-28 | Fujisawa Pharmaceutical Co., Ltd. | Benzoxazolone and benzothiazolone derivatives and process for their preparation |
| IT1176770B (en) | 1983-09-22 | 1987-08-18 | Fujisawa Pharmaceutical Co | DERIVATIVES OF BENZOSSAZOLONE AND BENZOTIAZOLONE PRO CEDIMENTS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOUND THAT CONTAINS THEM |
| US4593102A (en) | 1984-04-10 | 1986-06-03 | A. H. Robins Company, Inc. | N-[(amino)alkyl]-1-pyrrolidine, 1-piperidine and 1-homopiperidinecarboxamides (and thiocarboxamides) with sulfur linked substitution in the 2, 3 or 4-position |
| US4591548A (en) | 1984-07-20 | 1986-05-27 | Minnesota Mining And Manufacturing Company | Cyan dye-forming couplers and photographic elements and processes |
| US5019369A (en) | 1984-10-22 | 1991-05-28 | Vestar, Inc. | Method of targeting tumors in humans |
| JPS61129129A (en) | 1984-11-28 | 1986-06-17 | Kureha Chem Ind Co Ltd | Antitumor agent |
| EP0189069A2 (en) | 1985-01-18 | 1986-07-30 | Nissan Chemical Industries Ltd. | Pyrazolesulfonamide derivative, process for its production and herbicide containing it |
| US4881965A (en) | 1985-01-18 | 1989-11-21 | Nissan Chemical Industries, Ltd. | Wheat growing process utilizing selective herebicide |
| DE3512630A1 (en) | 1985-04-06 | 1986-10-23 | Hoechst Ag, 6230 Frankfurt | METHOD FOR COLORING OR PRINTING CELLULOSE FIBERS OR CELLULOSE MIXED FIBERS |
| US4889553A (en) | 1986-05-17 | 1989-12-26 | Schering Agrochemicals | Herbicides |
| EP0246749A2 (en) | 1986-05-17 | 1987-11-25 | AgrEvo UK Limited | Triazole herbicides |
| US4849424A (en) | 1986-08-05 | 1989-07-18 | Nissin Shokuhin Kabushiki Kaisha | Pyrimidine derivatives |
| US4837028A (en) | 1986-12-24 | 1989-06-06 | Liposome Technology, Inc. | Liposomes with enhanced circulation time |
| US4775762A (en) | 1987-05-11 | 1988-10-04 | The Dow Chemical Company | Novel (1H-1,2,3-triazol-1-yl)pyridines |
| US5962490A (en) | 1987-09-25 | 1999-10-05 | Texas Biotechnology Corporation | Thienyl-, furyl- and pyrrolyl-sulfonamides and derivatives thereof that modulate the activity of endothelin |
| DE3813886A1 (en) | 1988-04-20 | 1989-11-02 | Schering Ag | 1-Triazinyl-1H-1,2,4-triazole-3-sulphonamides, processes for their preparation, and their use as herbicides, fungicides and plant growth regulators |
| US4959094A (en) | 1988-04-20 | 1990-09-25 | Schering Aktiengesellschaft | 1-chloropyrimidinyl-1H-1,2,4-triazole-3-sulphonamides as herbicides |
| US5220028A (en) | 1988-10-27 | 1993-06-15 | Nissan Chemical Industries, Ltd. | Halogeno-4-methylpyrazoles |
| EP0384228A1 (en) | 1989-02-21 | 1990-08-29 | Dainippon Pharmaceutical Co., Ltd. | Medicament for treating cerebral insufficiency diseases containing 2-(1-piperazinyl)-4-phenylcycloalkanopyrimidine derivatives, and process for the production thereof |
| US5021421A (en) | 1989-03-03 | 1991-06-04 | Dainippon Pharmaceutical Co., Ltd. | 2-(1-Piperazinyl)-4-phenylcycloalkanopyridine derivatives, processes for the production thereof, and pharmaceutical composition containing the same |
| EP0385237A2 (en) | 1989-03-03 | 1990-09-05 | Dainippon Pharmaceutical Co., Ltd. | 2-(1-piperazinyl)-4-phenylcycloalkanopyridine derivatives, processes for the production thereof, and pharmaceutical composition containing the same |
| US5180732A (en) | 1990-02-20 | 1993-01-19 | Sumitomo Chemical Company, Limited | 4-tert.-butyl imidazole derivative and use |
| US5122530A (en) | 1990-06-29 | 1992-06-16 | Sumitomo Chemical Company, Limited | 1-pyridylimidazole derivative and its use |
| JPH0499768A (en) | 1990-08-17 | 1992-03-31 | Dainippon Pharmaceut Co Ltd | 4-(4-phenylpyridin-2-yl)piperazine-1-oxide derivative |
| WO1992011761A1 (en) | 1990-12-31 | 1992-07-23 | Monsanto Company | Reducing pesticidal interactions in crops |
| US5252590A (en) | 1991-06-28 | 1993-10-12 | Sumitomo Chemical Company, Limited | 1-pyridylimidazole derivative |
| WO1993013072A1 (en) | 1991-12-20 | 1993-07-08 | Italfarmaco S.P.A. | 5-isoquinolinesulfonamide derivatives as protein kinase inhibiting agents |
| US5489591A (en) | 1992-02-28 | 1996-02-06 | Zenyaku Kogyo Kabushiki Kaisha | S-triazine derivative and remedy for estrogen-dependent disease containing said derivative as effective component |
| JPH0625177A (en) | 1992-07-09 | 1994-02-01 | Nissan Chem Ind Ltd | Pyrazole derivative and herbicide |
| US5556866A (en) | 1993-04-28 | 1996-09-17 | Kabushiki Kaisha Hayashibara Seibutsu Kagaku Kenkyujo | Hair restorer and its preparation |
| EP0628551A1 (en) | 1993-06-14 | 1994-12-14 | Shionogi & Co., Ltd. | Semicyclic urea derivatives as antihistaminic agents |
| JPH07165708A (en) | 1993-09-10 | 1995-06-27 | Eisai Co Ltd | Bicyclic heterocycle-containing sulfonamide and sulfonic acid ester derivative |
| US5965559A (en) | 1994-09-26 | 1999-10-12 | Zeneca Limited | Aminoheterocyclic derivatives as antithrombotic or anticoagulant |
| EP1586558A2 (en) | 1995-01-20 | 2005-10-19 | G.D. Searle LLC. | Bis-sulfonamide hydroxyethylamino retroviral protease inhinitors |
| WO1996030343A1 (en) | 1995-03-29 | 1996-10-03 | Merck & Co., Inc. | Inhibitors of farnesyl-protein transferase |
| FR2735127A1 (en) | 1995-06-09 | 1996-12-13 | Pf Medicament | NOVEL HETEROAROMATIC PIPERAZINES USEFUL AS MEDICAMENTS. |
| US5965569A (en) | 1995-10-11 | 1999-10-12 | Medichem, S.A. | Polycyclic aminopyridine compounds which are acetylcholinesterase inhibitors, process for preparing them and their use |
| CA2235621A1 (en) | 1995-10-25 | 1997-05-01 | Schering Aktiengesellschaft | Imidazole derivatives and their use as nitrogen monoxide synthase inhibitors |
| WO1997028129A1 (en) | 1996-02-02 | 1997-08-07 | Zeneca Limited | Aminoheterocyclic derivatives as antithrombotic or anticoagulant agents |
| US6313127B1 (en) | 1996-02-02 | 2001-11-06 | Zeneca Limited | Heterocyclic compounds useful as pharmaceutical agents |
| WO1997028128A1 (en) | 1996-02-02 | 1997-08-07 | Zeneca Limited | Heterocyclic compounds useful as pharmaceutical agents |
| US7173025B1 (en) | 1996-02-02 | 2007-02-06 | Zeneca Limited | Aminoheterocyclic derivatives as antithrombotic or anticoagulant agents |
| WO1997028141A1 (en) | 1996-02-02 | 1997-08-07 | Pierre Fabre Medicament | Novel aromatic piperazines derived from substituted cycloazanes, method for preparing same, pharmaceutical compositions, and use thereof as drugs |
| US20030207882A1 (en) | 1996-02-02 | 2003-11-06 | Zeneca Limited | Aminoheterocyclic derivatives as antithrombotic or anticoagulant agents |
| JPH09291034A (en) | 1996-02-27 | 1997-11-11 | Yoshitomi Pharmaceut Ind Ltd | Condensed pyridine compound and its use as medicine |
| US6262113B1 (en) | 1996-03-20 | 2001-07-17 | Smithkline Beecham Corporation | IL-8 receptor antagonists |
| US5807876A (en) | 1996-04-23 | 1998-09-15 | Vertex Pharmaceuticals Incorporated | Inhibitors of IMPDH enzyme |
| US5834485A (en) | 1996-05-20 | 1998-11-10 | Chiroscience Limited | Quinoline sulfonamides and their therapeutic use |
| WO1997044322A1 (en) | 1996-05-20 | 1997-11-27 | Darwin Discovery Limited | Quinoline sulfonamides as tnf inhibitors and as pde-iv inhibitors |
| US5843485A (en) | 1996-06-28 | 1998-12-01 | Incoe Corporation | Valve-gate bushing for gas-assisted injection molding |
| WO1998002434A1 (en) | 1996-07-13 | 1998-01-22 | Glaxo Group Limited | Fused heterocyclic compounds as protein tyrosine kinase inhibitors |
| WO1998003350A1 (en) | 1996-07-20 | 1998-01-29 | Golden Records Association International Corporation | Plastic card |
| US5984882A (en) | 1996-08-19 | 1999-11-16 | Angiosonics Inc. | Methods for prevention and treatment of cancer and other proliferative diseases with ultrasonic energy |
| EP0945446A1 (en) | 1996-11-14 | 1999-09-29 | Nissan Chemical Industries, Limited | Cyanoethylmelamine derivatives and process for producing the same |
| US6020357A (en) | 1996-12-23 | 2000-02-01 | Dupont Pharmaceuticals Company | Nitrogen containing heteroaromatics as factor Xa inhibitors |
| US6265588B1 (en) | 1997-01-28 | 2001-07-24 | Aventis Pharma Deutschland Gmbh | Isoxazole and crotonamide derivatives and their use as pharmaceuticals and diagnostics |
| US6172005B1 (en) | 1997-03-11 | 2001-01-09 | E. I. Du Pont De Nemours And Company | Heteroaryl azole herbicides |
| US7863444B2 (en) | 1997-03-19 | 2011-01-04 | Abbott Laboratories | 4-aminopyrrolopyrimidines as kinase inhibitors |
| US20030187001A1 (en) | 1997-03-19 | 2003-10-02 | David Calderwood | 4-aminopyrrolopyrimidines as kinase inhibitors |
| US6399358B1 (en) | 1997-03-31 | 2002-06-04 | Thomas Jefferson University | Human gene encoding human chondroitin 6-sulfotransferase |
| US6359134B1 (en) | 1997-05-30 | 2002-03-19 | Takeda Chemical Industries, Ltd. | Sulfonamide derivatives, their production and use |
| JPH11158073A (en) | 1997-09-26 | 1999-06-15 | Takeda Chem Ind Ltd | Adenosine a3 antagonist |
| US6492368B1 (en) | 1997-10-01 | 2002-12-10 | Merck Patent Gmbh | Benzamidine derivatives as factor XA inhibitors |
| WO1999016751A1 (en) | 1997-10-01 | 1999-04-08 | Merck Patent Gmbh | Benzamidine derivatives as factor xa inhibitors |
| US20090093526A1 (en) | 1997-12-22 | 2009-04-09 | Scott Miller | Inhibition of p38 kinase using symmetrical and unsymmetrical diphenyl ureas |
| WO1999032463A1 (en) | 1997-12-22 | 1999-07-01 | Bayer Corporation | INHIBITION OF p38 KINASE USING SYMMETRICAL AND UNSYMMETRICAL DIPHENYL UREAS |
| US6106849A (en) | 1998-01-21 | 2000-08-22 | Dragoco Gerberding & Co. Ag | Water soluble dry foam personal care product |
| WO1999047508A1 (en) | 1998-03-17 | 1999-09-23 | Astrazeneca Ab | Benzenesulfonamide-derivatives and their use as medicaments |
| JP2002506859A (en) | 1998-03-17 | 2002-03-05 | アストラゼネカ アクチボラグ | Benzenesulfonamide derivatives and their use as drugs |
| WO1999048490A1 (en) | 1998-03-24 | 1999-09-30 | Allos Therapeutics, Inc. | Allosteric inhibitors and activators of pyruvate kinase |
| WO1999062506A1 (en) | 1998-05-29 | 1999-12-09 | Astrazeneca Ab | Use of compounds for the elevation of pyruvate dehydrogenase activity |
| US6511977B1 (en) | 1998-09-01 | 2003-01-28 | Bristol-Myers Squibb Company | Potassium channel inhibitors and method |
| US6150356A (en) | 1998-09-01 | 2000-11-21 | Bristol-Myers Squibb Company | Potassium channel inhibitors and method |
| DE19841985A1 (en) | 1998-09-03 | 2000-03-09 | Schering Ag | New heterocyclic alkanesulfonic and alkane carboxylic acid derivatives are VEGF receptor blockers useful in treatment of e.g. psoriasis, rheumatoid arthritis, stroke, tumors and endometriosis |
| WO2000017202A1 (en) | 1998-09-18 | 2000-03-30 | Basf Aktiengesellschaft | 4-aminopyrrolopyrimidines as kinase inhibitors |
| US20030109527A1 (en) | 1998-12-16 | 2003-06-12 | Smithkline Beecham Corporation | Hydroxy diphenyl urea sulfonamides as IL-8 receptor antagonists |
| WO2000053596A2 (en) | 1999-03-08 | 2000-09-14 | Schering Corporation | Imidazole compounds as histamine h3 ligands |
| US6274620B1 (en) | 1999-06-07 | 2001-08-14 | Biochem Pharma Inc. | Thiophene integrin inhibitors |
| WO2001007440A1 (en) | 1999-07-21 | 2001-02-01 | Boehringer Ingelheim Pharmaceuticals, Inc. | Imidazoimidazoles and triazoles as anti-inflammatory agents |
| WO2001016097A1 (en) | 1999-08-27 | 2001-03-08 | Sugen, Inc. | Phosphate mimics and methods of treatment using phosphatase inhibitors |
| WO2001019788A2 (en) | 1999-09-17 | 2001-03-22 | Cor Therapeutics, Inc. | BENZAMIDES AND RELATED INHIBITORS OF FACTOR Xa |
| WO2001019798A2 (en) | 1999-09-17 | 2001-03-22 | Cor Therapeutics Inc. | INHIBITORS OF FACTOR Xa |
| WO2001064643A2 (en) | 2000-02-29 | 2001-09-07 | Cor Therapeutics, Inc. | BENZAMIDES AND RELATED INHIBITORS OF FACTOR Xa |
| WO2001064642A2 (en) | 2000-02-29 | 2001-09-07 | Cor Therapeutics, Inc. | Benzamides and related inhibitors of factor xa |
| US20030106381A1 (en) | 2000-03-08 | 2003-06-12 | Krouth Terrance F. | Hydraulic actuator piston measurement apparatus and method |
| US6515013B2 (en) | 2000-07-13 | 2003-02-04 | Abbott Laboratories | 1,3-disubstituted and 1,3,3-trisubstituted pyrrolidines as histamine-3 receptor ligands and their therapeutic applications |
| US6723730B2 (en) | 2000-07-20 | 2004-04-20 | Neurogen Corporation | Capsaicin receptor ligands |
| US20040048283A1 (en) | 2000-11-28 | 2004-03-11 | Bernard Pau | Novel method for screening bacterial transcription modulators |
| JP2002193710A (en) | 2000-12-25 | 2002-07-10 | Kumiai Chem Ind Co Ltd | Pyrimidine or triazine derivative and fungicide for agriculture and horticulture |
| US6525091B2 (en) | 2001-03-07 | 2003-02-25 | Telik, Inc. | Substituted diarylureas as stimulators for Fas-mediated apoptosis |
| US20040235755A1 (en) | 2001-03-13 | 2004-11-25 | Erich Eigenbrodt | Use of amino acids amino acid alogues, sugar phosphates and sugar phosphate analogues for treatment of tumors, treatment of sepsis and immunosuppression |
| WO2002072077A2 (en) | 2001-03-13 | 2002-09-19 | Schebo Biotech Ag | Use of amino acids, amino acid analogues, sugar phosphates and sugar phosphate analogues for treatment of tumors, treatment of sepsis and immunosuppression |
| JP2004534017A (en) | 2001-04-27 | 2004-11-11 | バーテックス ファーマシューティカルズ インコーポレイテッド | BACE inhibitors |
| WO2002088101A2 (en) | 2001-04-27 | 2002-11-07 | Vertex Pharmaceuticals Incorporated | Inhibitors of bace |
| US20030095958A1 (en) | 2001-04-27 | 2003-05-22 | Bhisetti Govinda R. | Inhibitors of bace |
| US20040198979A1 (en) | 2001-05-07 | 2004-10-07 | Dashyant Dhanak | Sulfonamides |
| WO2002095063A1 (en) | 2001-05-23 | 2002-11-28 | MAX-PLANCK-Gesellschaft zur Förderung der Wissenschaften e.V. | Pyruvate-kinase as a novel target molecule |
| US20080021116A1 (en) | 2001-05-23 | 2008-01-24 | Max-Planck-Gesellschaft Zur Foerderung Der Wissenschaften E.V. | Pyruvate-kinase as a novel target molecule |
| US20040152648A1 (en) | 2001-05-23 | 2004-08-05 | Axel Ullrich | Pyruvate-kinase as a novel target molecule |
| US20030158232A1 (en) | 2001-05-30 | 2003-08-21 | Cheng Peter T. | Substituted azole acid derivatives useful as antidiabetic and antiobesity agents and method |
| WO2002100822A1 (en) | 2001-06-11 | 2002-12-19 | Biovitrum Ab | Substituted sulfonamide compounds, process for their use as medicament for the treatment of cns disorders, obesity and type ii diabetes |
| US7235664B2 (en) | 2001-06-12 | 2007-06-26 | Sanofi-Aventis Deutschland Gmbh | Anthranilamides with heteroarylsulfonyl side chain, process of preparation, and use |
| US20030082877A1 (en) | 2001-06-18 | 2003-05-01 | Hitachi, Ltd. | Semiconductor integrated circuit device and a method of manufacturing the same |
| WO2002102313A2 (en) | 2001-06-19 | 2002-12-27 | Bristol-Myers Squibb Company | Pyrimidine inhibitors of phosphodiesterase (pde) 7 |
| US7288554B2 (en) | 2001-08-15 | 2007-10-30 | E.I. Du Pont De Nemours And Company | Ortho-substituted aryl amides for controlling invertebrate pests |
| WO2003016300A1 (en) | 2001-08-15 | 2003-02-27 | E. I. Du Pont De Nemours And Company | Ortho-substituted aryl amides for controlling invertebrate pests |
| WO2003016289A1 (en) | 2001-08-17 | 2003-02-27 | Ciba Specialty Chemicals Holding Inc. | Triazine derivatives and their use as sunscreens |
| JP4753336B2 (en) | 2001-09-04 | 2011-08-24 | 日本化薬株式会社 | Novel allyl compound and process for producing the same |
| WO2003022277A1 (en) | 2001-09-07 | 2003-03-20 | Bayer Healthcare Ag | Arylsulfonamide derivatives for use as ccr3 antagonists in the treatment of inflammatory and immunological disorders |
| WO2003037252A2 (en) | 2001-10-30 | 2003-05-08 | Merck & Co., Inc. | Tyrosine kinase inhibitors |
| US6878196B2 (en) | 2002-01-15 | 2005-04-12 | Fuji Photo Film Co., Ltd. | Ink, ink jet recording method and azo compound |
| WO2003062235A1 (en) | 2002-01-17 | 2003-07-31 | Eli Lilly And Company | Modulators of acetylcholine receptors |
| WO2003073999A2 (en) | 2002-03-01 | 2003-09-12 | Pintex Pharmaceuticals, Inc. | Pini-modulating compounds and methods of use thereof |
| US7615553B2 (en) | 2002-03-13 | 2009-11-10 | Janssen Pharmaceutica N.V. | Inhibitors of histone deacetylase |
| WO2003076422A1 (en) | 2002-03-13 | 2003-09-18 | Janssen Pharmaceutica N.V. | Sulfonyl-derivatives as novel inhibitors of histone deacetylase |
| US7452888B2 (en) | 2002-03-27 | 2008-11-18 | Glaxo Group Limited | Quinoline derivatives and their use as 5-ht6 ligands |
| WO2003093297A2 (en) | 2002-05-03 | 2003-11-13 | Exelixis, Inc. | Protein kinase modulators and methods of use |
| WO2004004730A2 (en) | 2002-07-06 | 2004-01-15 | Astex Technology Limited | 2-aminopyrazine derivatives as inhibitors of cyclin dependent kinases for the treatment of proliferative disorders |
| US20080300208A1 (en) | 2002-07-11 | 2008-12-04 | Quark Pharmaceuticals, Inc. | Isociatrate dehydrogenase and uses therof |
| US20040067234A1 (en) | 2002-07-11 | 2004-04-08 | Paz Einat | Isocitrate dehydrogenase and uses thereof |
| WO2004009562A1 (en) | 2002-07-18 | 2004-01-29 | Janssen Pharmaceutica, Nv | Substituted triazine kinase inhibitors |
| WO2004014851A2 (en) | 2002-08-09 | 2004-02-19 | Merck & Co., Inc. | Tyrosine kinase inhibitors |
| JP2004107220A (en) | 2002-09-13 | 2004-04-08 | Mitsubishi Pharma Corp | TNF-alpha PRODUCTION INHIBITOR |
| WO2004037251A1 (en) | 2002-10-24 | 2004-05-06 | Sterix Limited | Inhibitors of 11-beta-hydroxy steroid dehydrogenase type 1 and type 2 |
| WO2004046120A2 (en) | 2002-11-15 | 2004-06-03 | Vertex Pharmaceuticals Incorporated | Diaminotriazoles useful as inhibitors of protein kinases |
| WO2004050033A2 (en) | 2002-12-02 | 2004-06-17 | Arqule, Inc. | Method of treating cancers |
| US8603469B2 (en) | 2002-12-16 | 2013-12-10 | Genmab A/S | Methods of treating cancer with human monoclonal antibodies against interleukin 8 |
| US6979675B2 (en) | 2003-01-10 | 2005-12-27 | Threshold Pharmaceuticals, Inc. | Treatment of cancer with 2-deoxyglucose |
| US7358262B2 (en) | 2003-01-29 | 2008-04-15 | Whitehead Institute For Biomedical Research | Identification of genotype-selective anti-tumor agents |
| WO2004074438A2 (en) | 2003-02-14 | 2004-09-02 | Smithkline Beecham Corporation | Ccr8 antagonists |
| WO2004073619A2 (en) | 2003-02-14 | 2004-09-02 | Smithkline Beecham Corporation | Ccr8 antagonists |
| JP2006515337A (en) | 2003-02-19 | 2006-05-25 | エフ.ホフマン−ラ ロシュ アーゲー | Sulfonamide-substituted xanthine derivatives for use as PEPCK inhibitors |
| WO2004074288A1 (en) | 2003-02-19 | 2004-09-02 | F. Hoffmann-La Roche Ag | Sulfonamide substituted xanthine derivatives for use as pepck inhibitors |
| US20070032418A1 (en) | 2003-02-25 | 2007-02-08 | Ambion, Inc | Small-molecule inhibitors of angiogenin and rnases and in vivo and in vitro methods of using same |
| US7595322B2 (en) | 2003-03-27 | 2009-09-29 | Cytokinetics, Inc. | Heterocyclic sulfonamides as modulators of cardiac sarcomeres |
| US7241759B2 (en) | 2003-03-28 | 2007-07-10 | Janssen Pharmaceutica N.V. | Benzo[1,2,5]thiadiazole compounds |
| WO2004089470A2 (en) | 2003-04-11 | 2004-10-21 | Novo Nordisk A/S | New amide derivatives and pharmaceutical use thereof |
| WO2004110375A2 (en) | 2003-06-06 | 2004-12-23 | Merck & Co., Inc. | Combination therapy for the treatment of diabetes |
| US20070032520A1 (en) | 2003-07-30 | 2007-02-08 | Laboratorios Del Dr. Esteve S.A | Indol-5-yl sulfonamide derivatives, their preparation and their use 5-ht-6 as modulators |
| US6818631B1 (en) | 2003-08-15 | 2004-11-16 | Nippon Soda Co. Ltd. | Fungicidal pyrimidine derivatives |
| WO2005035507A2 (en) | 2003-10-10 | 2005-04-21 | Bayer Pharmaceuticals Corporation | 4-aminopyrimidine derivatives for treatment of hyperproliferative disorders |
| US20080051414A1 (en) | 2003-10-14 | 2008-02-28 | Arizona Board Of Regents On Behalf Of The University Of Arizona | Protein Kinase Inhibitors |
| JP4099768B2 (en) | 2003-11-10 | 2008-06-11 | 富士電機デバイステクノロジー株式会社 | Electrophotographic photosensitive member and method for determining presence or absence of interference fringes resulting from electrophotographic photosensitive member |
| WO2005060956A1 (en) | 2003-12-12 | 2005-07-07 | University Of Maryland, Baltimore | IMMUNOMODULATORY COMPOUNDS THAT TARGET AND INHIBIT THE pY+3 BINDING SITE OF TYROSENE KINASE p56 LCK SH2 DOMAIN |
| JP2007519631A (en) | 2003-12-23 | 2007-07-19 | イーライ リリー アンド カンパニー | CB1 modulator compound |
| WO2005066126A1 (en) | 2003-12-23 | 2005-07-21 | Eli Lilly And Company | Cb1 modulator compounds |
| WO2005065691A1 (en) | 2003-12-24 | 2005-07-21 | Scios, Inc. | Treatment of malignant gliomas with tgf-beta inhibitors |
| WO2005072642A1 (en) | 2004-01-29 | 2005-08-11 | Colgate-Palmolive Company | Light-based toothbrush |
| US20050176675A1 (en) | 2004-02-10 | 2005-08-11 | Philipe Gorny | Stable and active complexes of adenosine and adenosine phosphates with aminoalcohols for the treatment of pulmonary artery hypertension, cardiac failure and other diseases |
| US7572913B2 (en) | 2004-02-24 | 2009-08-11 | Bioaxone Therapeutique Inc. | 4-substituted piperidine derivatives |
| WO2005117591A2 (en) | 2004-05-28 | 2005-12-15 | Andrx Labs Llc | Novel pharmaceutical formulation containing a biguanide and an angiotensin antagonist |
| WO2005120474A2 (en) | 2004-06-05 | 2005-12-22 | K.U.Leuven Research And Development | Phophodiesterase 10a inhibitors to amplify the action of glp1-mimetics or dpp-iv inhibitors in diabetes |
| US8058313B2 (en) | 2004-06-24 | 2011-11-15 | Temple University—Of the Commonwealth System of Higher Education | Alpha, beta-unsaturated sulfones, sulfoxides, sulfonimides, sulfinimides, acylsulfonamides and acylsulfinamides and therapeutic uses thereof |
| US20070244088A1 (en) | 2004-06-28 | 2007-10-18 | Astrazeneca Ab | New Pyridine Analogues II |
| WO2006004195A1 (en) | 2004-07-02 | 2006-01-12 | Tanabe Seiyaku Co., Ltd. | Piperidine compound and process for preparing the same |
| WO2006016062A1 (en) | 2004-07-12 | 2006-02-16 | Laurent Schwartz | Multitherapy against cancer |
| US20070280918A1 (en) | 2004-07-12 | 2007-12-06 | Laurent Schwartz | Multitherapy Against Cancer |
| WO2006034341A2 (en) | 2004-09-20 | 2006-03-30 | Xenon Pharmaceuticals Inc. | Pyridazine derivatives for inhibiting human stearoyl-coa-desaturase |
| US7820654B2 (en) | 2004-09-23 | 2010-10-26 | Dr. Reddy's Laboratories Ltd. | Pyrimidine compounds, process for their preparation and compositions containing them |
| WO2006033628A1 (en) | 2004-09-24 | 2006-03-30 | Astrazeneca Ab | Benzimidazole derivatives, compositions containing them, preparation thereof and uses thereof |
| JP2008514590A (en) | 2004-09-24 | 2008-05-08 | アストラゼネカ・アクチエボラーグ | Benzimidazole derivative, composition containing the same, method for producing the same, and use thereof |
| WO2006038594A1 (en) | 2004-10-04 | 2006-04-13 | Ono Pharmaceutical Co., Ltd. | N-type calcium channel inhibitor |
| WO2006043950A1 (en) | 2004-10-20 | 2006-04-27 | Smithkline Beecham Corporation | Il-8 receptor antagonists |
| US20080004269A1 (en) | 2004-11-04 | 2008-01-03 | Yuelian Xu | Pyrazolylmethy Heteroaryl Derivatives |
| WO2006052190A1 (en) | 2004-11-11 | 2006-05-18 | Astrazeneca Ab | Indazole sulphonamide derivatives |
| US20080044833A1 (en) | 2004-11-16 | 2008-02-21 | Connors Timothy D | Diagnostic PKM2 methods and compositions |
| WO2006063294A2 (en) | 2004-12-09 | 2006-06-15 | Kalypsys, Inc. | Novel inhibitors of histone deacetylase for the treatment of disease |
| WO2006070198A1 (en) | 2004-12-30 | 2006-07-06 | Astex Therapeutics Limited | Pyrazole derivatives as that modulate the activity of cdk, gsk and aurora kinases |
| WO2006077821A1 (en) | 2005-01-19 | 2006-07-27 | Dainippon Sumitomo Pharma Co., Ltd. | Aromatic sulfone compound as aldosterone receptor modulator |
| WO2006079791A1 (en) | 2005-01-25 | 2006-08-03 | Astrazeneca Ab | Chemical compounds |
| US20100179150A1 (en) | 2005-02-18 | 2010-07-15 | Astrazeneca Ab | Antibacterial piperdine derivatives |
| WO2006117762A2 (en) | 2005-05-03 | 2006-11-09 | Ranbaxy Laboratories Limited | Antimicrobial agents |
| WO2006122546A1 (en) | 2005-05-18 | 2006-11-23 | Forschungsverbund Berlin E.V. | Non-peptidic inhibitors of akap/pka interaction |
| US8278038B2 (en) | 2005-06-08 | 2012-10-02 | Millennium Pharmaceuticals, Inc. | Methods for the identification, assessment, and treatment of patients with cancer therapy |
| WO2007003934A2 (en) | 2005-07-04 | 2007-01-11 | Sterix Limited | 17beta-hydr0xyster0id dehydrogenase type 3 (17beta-hsd3 ) inhibitors |
| US20100105657A1 (en) | 2005-07-05 | 2010-04-29 | Astrazeneca Ab | Compounds, Process for their Preparation, Intermediates, Pharmaceutical Compositions and their use in the Treatment of 5-HT6 Mediated Disorders such as Alzheimer's Disease, Cognitive Disorders, Cognitive Impairment Associated with Schizophrenia, Obesity and Parkinson's Disease |
| US20080214495A1 (en) | 2005-07-08 | 2008-09-04 | Astrazeneca Ab | Heterocyclic Sulfonamide Derivatives as Inhibitors of Factor Xa |
| CN101296909A (en) | 2005-08-26 | 2008-10-29 | 雪兰诺实验室有限公司 | Pyrazine derivatives and use as pI3K inhibitors |
| WO2007023186A1 (en) | 2005-08-26 | 2007-03-01 | Laboratoires Serono S.A. | Pyrazine derivatives and use as pi3k inhibitors |
| US8133900B2 (en) | 2005-11-01 | 2012-03-13 | Targegen, Inc. | Use of bi-aryl meta-pyrimidine inhibitors of kinases |
| US20070127505A1 (en) | 2005-12-02 | 2007-06-07 | Nokia Corporation | Group communication |
| US20090048227A1 (en) | 2005-12-22 | 2009-02-19 | Chakravarty Pasun K | Substituted-1-Phthalazinamines As Vr- 1 Antagonists |
| WO2007127505A2 (en) | 2006-01-20 | 2007-11-08 | Smithkline Beecham Corporation | Use of sulfonamide derivatives in the treatment of disorders of the metabolism and the nervous system |
| JP2007238458A (en) | 2006-03-06 | 2007-09-20 | D Western Therapeutics Institute Inc | Novel isoquinoline derivative and medicine containing the same |
| US20090054453A1 (en) | 2006-03-17 | 2009-02-26 | Lilian Alcaraz | Novel Tetralins as 5-HT6 Modulators |
| US7524848B2 (en) | 2006-03-23 | 2009-04-28 | Amgen Inc. | Diaza heterocyclic amide compounds and their uses |
| WO2007117465A2 (en) | 2006-03-31 | 2007-10-18 | Abbott Laboratories | Indazole compounds |
| WO2008019139A2 (en) | 2006-08-04 | 2008-02-14 | Beth Israel Deaconess Medical Center | Inhibitors of pyruvate kinase and methods of treating disease |
| US8877791B2 (en) | 2006-08-04 | 2014-11-04 | Beth Israel Deaconess Medical Center, Inc. | Inhibitors of pyruvate kinase and methods of treating disease |
| US20090247499A1 (en) | 2006-08-21 | 2009-10-01 | Fletcher Joan M | Sulfonylated piperazines as cannabinoid-1 receptor modulators |
| WO2008024284A2 (en) | 2006-08-21 | 2008-02-28 | Merck & Co., Inc. | Sulfonylated piperazines as cannabinoid-1 receptor modulators |
| WO2008026658A1 (en) | 2006-09-01 | 2008-03-06 | Otsuka Chemical Co., Ltd. | N-pyridylpiperidine compound, method for producing the same, and pest control agent |
| WO2008043381A1 (en) | 2006-10-12 | 2008-04-17 | Fluid Automation Systems S.A. | Rocker valve mechanism and rocker valve |
| JP2010506122A (en) | 2006-10-12 | 2010-02-25 | フルーイッド・オートメーション・システムズ・ソシエテ・アノニム | Rocker valve mechanism and rocker valve |
| WO2008047198A1 (en) | 2006-10-16 | 2008-04-24 | Pfizer Products Inc. | Therapeutic pyrazolyl thienopyridines |
| WO2008052190A2 (en) | 2006-10-26 | 2008-05-02 | Flynn Gary A | Aquaporin modulators and methods of using them for the treatment of edema and fluid imbalance |
| WO2008050168A1 (en) | 2006-10-27 | 2008-05-02 | Richter Gedeon Nyrt. | New sulfonamide derivatives as bradykinin antagonists |
| WO2008070661A1 (en) | 2006-12-04 | 2008-06-12 | Neurocrine Biosciences, Inc. | Substituted pyrimidines as adenosine receptor antagonists |
| WO2008073670A2 (en) | 2006-12-08 | 2008-06-19 | Millennium Pharmaceuticals, Inc. | Unit dose formulations and methods of treating thrombosis with an oral factor xa inhibitor |
| WO2008076883A2 (en) | 2006-12-15 | 2008-06-26 | Abraxis Bioscience, Inc. | Triazine derivatives and their therapeutical applications |
| US7858782B2 (en) | 2006-12-15 | 2010-12-28 | Abraxis Bioscience, Llc | Triazine derivatives and their therapeutical applications |
| US20090270454A1 (en) | 2007-04-23 | 2009-10-29 | Weingarten M David | Sulfonamide containing compounds for treatment of inflammatory disorders |
| US8258295B2 (en) | 2007-04-30 | 2012-09-04 | Prometic Biosciences Inc. | Triazine derivatives, compositions containing such derivatives, and methods of treatment of cancer and autoimmune diseases using such derivatives |
| WO2008131547A1 (en) | 2007-04-30 | 2008-11-06 | Prometic Biosciences Inc. | 'triazine derivatives, compositions containing such derivatives, and methods of treatment of cancer and autoimmune diseases using such derivatives' |
| WO2008154026A1 (en) | 2007-06-11 | 2008-12-18 | Miikana Therapeutics, Inc. | Substituted pyrazole compounds |
| WO2009012430A1 (en) | 2007-07-18 | 2009-01-22 | Janssen Pharmaceutica N.V. | Sulfonamides as trpm8 modulators |
| WO2009013126A1 (en) | 2007-07-20 | 2009-01-29 | Nerviano Medical Sciences S.R.L. | Substituted indazole derivatives active as kinase inhibitors |
| WO2009016410A2 (en) | 2007-07-31 | 2009-02-05 | Astrazeneca Ab | Chemical compounds 831 |
| WO2009025781A1 (en) | 2007-08-16 | 2009-02-26 | Beth Israel Deaconess Medical Center | Activators of pyruvate kinase m2 and methods of treating disease |
| US20110046083A1 (en) | 2007-08-16 | 2011-02-24 | Beth Israel Deaconess Medical Center | Activators of pyruvate kinase m2 and methods of treating disease |
| US20090163508A1 (en) | 2007-10-10 | 2009-06-25 | Takeda Pharmaceutical Company Limited | Amide compound |
| US20110224252A1 (en) | 2007-10-26 | 2011-09-15 | Syngenta Crop Protection, Inc. | Novel imidazole derivatives |
| WO2009053102A1 (en) | 2007-10-26 | 2009-04-30 | Syngenta Participations Ag | Novel imidazole derivatives |
| US20090163545A1 (en) | 2007-12-21 | 2009-06-25 | University Of Rochester | Method For Altering The Lifespan Of Eukaryotic Organisms |
| CA2709784A1 (en) | 2007-12-21 | 2009-07-09 | University Of Rochester | Method for altering the lifespan of eukaryotic organisms |
| AU2008345225A1 (en) | 2007-12-21 | 2009-07-09 | University Of Rochester | Method for altering the lifespan of eukaryotic organisms |
| JP2011507910A (en) | 2007-12-21 | 2011-03-10 | ユニバーシティー オブ ロチェスター | Methods for changing the lifetime of eukaryotes |
| EP2219646A2 (en) | 2007-12-21 | 2010-08-25 | University Of Rochester | Method for altering the lifespan of eukaryotic organisms |
| WO2009086303A2 (en) | 2007-12-21 | 2009-07-09 | University Of Rochester | Method for altering the lifespan of eukaryotic organisms |
| US8642660B2 (en) | 2007-12-21 | 2014-02-04 | The University Of Rochester | Method for altering the lifespan of eukaryotic organisms |
| WO2009118567A2 (en) | 2008-03-26 | 2009-10-01 | The University Of Nottingham | Pyrimidines, triazines and their use as pharmaceutical agents |
| JP2009237115A (en) | 2008-03-26 | 2009-10-15 | Fuji Xerox Co Ltd | Electrophotographic photoreceptor, image forming apparatus, process cartridge, and image forming method |
| WO2009126863A2 (en) | 2008-04-11 | 2009-10-15 | Genentech, Inc. | Pyridyl inhibitors of hedgehog signalling |
| CN101575408A (en) | 2008-05-09 | 2009-11-11 | Mca技术有限公司 | Polytriazinyl compounds as flame retardants and light stabilizers |
| WO2009150248A1 (en) | 2008-06-13 | 2009-12-17 | Cytomics Systems | Compounds which can be used for the treatment of cancers |
| WO2010007756A1 (en) | 2008-07-14 | 2010-01-21 | 塩野義製薬株式会社 | Pyridine derivative having ttk inhibition activity |
| WO2010023445A1 (en) | 2008-08-29 | 2010-03-04 | Xention Limited | Novel potassium channel blocker |
| WO2010028099A1 (en) | 2008-09-03 | 2010-03-11 | The Johns Hopkins University | Genetic alterations in isocitrate dehydrogenase and other genes in malignant glioma |
| US20100144722A1 (en) | 2008-09-03 | 2010-06-10 | Dr. Reddy's Laboratories Ltd. | Novel heterocyclic compounds as gata modulators |
| JP2010079130A (en) | 2008-09-29 | 2010-04-08 | Fuji Xerox Co Ltd | Electrophotographic photoreceptor, process cartridge, and image forming apparatus |
| WO2010042867A2 (en) | 2008-10-09 | 2010-04-15 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Activators of human pyruvate kinase |
| US20100273808A1 (en) | 2008-11-21 | 2010-10-28 | Millennium Pharmaceticals, Inc. | Lactate salt of 4-[6-methoxy-7-(3-piperidin-1-yl-propoxy)quinazolin-4-yl]piperazine-1-carboxylic acid(4-isopropoxyphenyl)-amide and pharmaceutical compositions thereof for the treatment of cancer and other diseases or disorders |
| JP2010181540A (en) | 2009-02-04 | 2010-08-19 | Fuji Xerox Co Ltd | Electrophotographic photoreceptor, process cartridge and image forming apparatus |
| US8673891B2 (en) | 2009-02-06 | 2014-03-18 | Nippon Shinyaku Co., Ltd. | Aminopyrazine derivative and medicine |
| US20140187435A1 (en) | 2009-03-13 | 2014-07-03 | Agios Pharmaceuticals, Inc | Methods and compositions for cell-proliferation-related disorders |
| US20120121515A1 (en) | 2009-03-13 | 2012-05-17 | Lenny Dang | Methods and compositions for cell-proliferation-related disorders |
| WO2010105243A1 (en) | 2009-03-13 | 2010-09-16 | Agios Pharmaceuticals, Inc. | Methods and compositions for cell-proliferation-related disorders |
| US20140323729A1 (en) | 2009-04-06 | 2014-10-30 | Agios Pharmaceuticals, Inc | Pyruvate kinase m2 modulators, therapeutic compositions and related methods of use |
| WO2010118063A2 (en) | 2009-04-06 | 2010-10-14 | Agios Pharmaceuticals, Inc. | Therapeutic compositions and related methods of use |
| US8742119B2 (en) | 2009-04-06 | 2014-06-03 | Agios Pharmaceuticals, Inc. | Pyruvate kinase M2 modulators, therapeutic compositions and related methods of use |
| EP2417123A2 (en) | 2009-04-06 | 2012-02-15 | Agios Pharmaceuticals, Inc. | Therapeutic compositions and related methods of use |
| US8501953B2 (en) | 2009-05-04 | 2013-08-06 | Agios Pharmaceuticals, Inc | PKM2 modulators for use in the treatment of cancer |
| WO2010129596A1 (en) | 2009-05-04 | 2010-11-11 | Agios Pharmaceuticals, Inc. | Pmk2 modulators for use in the treatment of cancer |
| WO2010130638A1 (en) | 2009-05-14 | 2010-11-18 | Evotec Ag | Sulfonamide compounds, pharmaceutical compositions and uses thereof |
| WO2010144338A1 (en) | 2009-06-08 | 2010-12-16 | Abraxis Bioscience, Llc | Triazine derivatives and their therapeutical applications |
| US20120238576A1 (en) | 2009-06-08 | 2012-09-20 | California Capital Equity, Llc | Triazine Derivatives and their Therapeutical Applications |
| US20120202818A1 (en) | 2009-06-09 | 2012-08-09 | California Capital Equity, Llc | Ureidophenyl substituted triazine derivatives and their therapeutical applications |
| US20120277233A1 (en) | 2009-06-09 | 2012-11-01 | California Capital Equity, Llc | Pyridyl-Triazine Inhibitors of Hedgehog Signaling |
| WO2010144404A1 (en) | 2009-06-09 | 2010-12-16 | Abraxis Bioscience, Llc | Pyridil-triazine inhibitors of hedgehog signaling |
| JP2015028083A (en) | 2009-06-29 | 2015-02-12 | アジオス ファーマシューティカルズ, インコーポレイテッド | Therapeutic compound and composition |
| US10029987B2 (en) | 2009-06-29 | 2018-07-24 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| US20190345109A1 (en) | 2009-06-29 | 2019-11-14 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| US20140323467A1 (en) | 2009-06-29 | 2014-10-30 | Agios Pharmaceuticals, Inc | Therapeutic compounds and compositions |
| WO2011002817A1 (en) | 2009-06-29 | 2011-01-06 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| US20190241523A1 (en) | 2009-06-29 | 2019-08-08 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| TW201103913A (en) | 2009-06-29 | 2011-02-01 | Agios Pharmaceuticals Inc | Therapeutic compounds and compositions |
| WO2011002816A1 (en) | 2009-06-29 | 2011-01-06 | Agios Pharmaceuticals, Inc. | Therapeutic compositions and related methods of use |
| US20100331307A1 (en) | 2009-06-29 | 2010-12-30 | Salituro Francesco G | Therapeutic compounds and compositions |
| US8785450B2 (en) | 2009-06-29 | 2014-07-22 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| TWI598337B (en) | 2009-06-29 | 2017-09-11 | 阿吉歐斯製藥公司 | Therapeutic compounds and compositions |
| US9115086B2 (en) | 2009-06-29 | 2015-08-25 | Agios Pharmaceuticals, Inc. | Therapeutic compositions and related methods of use |
| JP2012532137A (en) | 2009-06-29 | 2012-12-13 | アジオス ファーマシューティカルズ, インコーポレイテッド | Therapeutic compounds and compositions |
| CN102481300A (en) | 2009-06-29 | 2012-05-30 | 安吉奥斯医药品有限公司 | Therapeutic compounds and compositions |
| TWI472522B (en) | 2009-06-29 | 2015-02-11 | Agios Pharmaceuticals Inc | Therapeutic compounds and compositions |
| WO2011032169A2 (en) | 2009-09-14 | 2011-03-17 | Phusis Therapeutics Inc. | Pharmaceutical compositions and formulations including inhibitors of the pleckstrin homology domain and methods for using same |
| US8465673B2 (en) | 2009-09-30 | 2013-06-18 | Fujifilm Corporation | Humidity dependence improver for polymer film, polymer film, protective film for polarizer, retardation film, polarizer, and liquid crystal display device |
| US8652534B2 (en) | 2009-10-14 | 2014-02-18 | Berry Pharmaceuticals, LLC | Compositions and methods for treatment of mammalian skin |
| WO2011050210A1 (en) | 2009-10-21 | 2011-04-28 | Agios Pharmaceuticals, Inc. | Methods and compositions for cell-proliferation-related disorders |
| US9434979B2 (en) | 2009-10-21 | 2016-09-06 | Shin-San Michael Su | Methods and compositions for cell-proliferation-related disorders |
| US20130288284A1 (en) | 2009-10-21 | 2013-10-31 | Leonard Luan C Dang | Methods and compositions for cell-proliferation-related disorders |
| US8883438B2 (en) | 2009-10-21 | 2014-11-11 | Agios Pharmaceuticals, Inc. | Method for diagnosing cell proliferation disorders having a neoactive mutation at residue 97 of isocitrate dehydrogenase 1 |
| US20150240286A1 (en) | 2009-10-21 | 2015-08-27 | Agios Pharmaceuticals, Inc | Methods and compositions for cell-proliferation-related disorders |
| WO2011047432A1 (en) | 2009-10-22 | 2011-04-28 | Fibrotech Therapeutics Pty Ltd | Fused ring analogues of anti-fibrotic agents |
| US20130035329A1 (en) | 2009-12-09 | 2013-02-07 | Saunders Jeffrey O | Therapeutically active compositions and their methods of use |
| WO2011072174A1 (en) | 2009-12-09 | 2011-06-16 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds for use in the treatment of cancer characterized as having an idh mutation |
| US20170166541A1 (en) | 2009-12-09 | 2017-06-15 | Agios Pharmaceuticals, Inc | Therapeutically active compositions and their methods of use |
| WO2011109441A1 (en) | 2010-03-01 | 2011-09-09 | Myrexis, Inc. | Compounds and therapeutic uses thereof |
| US9150505B2 (en) * | 2010-03-04 | 2015-10-06 | Ajinomoto Co., Inc. | Prophylactic or therapeutic agent for diabetes or obesity |
| US20130197106A1 (en) | 2010-04-01 | 2013-08-01 | Agios Pharmaceuticals, Inc | Methods of identifying a candidate compound |
| WO2011137089A1 (en) | 2010-04-29 | 2011-11-03 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Activators of human pyruvate kinase |
| US20130109643A1 (en) | 2010-05-10 | 2013-05-02 | The Johns Hopkins University | Metabolic inhibitor against tumors having an idh mutation |
| US20110312931A1 (en) | 2010-06-04 | 2011-12-22 | Albany Molecular Research, Inc. | Glycine transporter-1 inhibitors, methods of making them, and uses thereof |
| CN103097340A (en) | 2010-07-16 | 2013-05-08 | 安吉奥斯医药品有限公司 | Therapeutically active compositions and their method of use |
| WO2012009678A1 (en) | 2010-07-16 | 2012-01-19 | Agios Pharmaceuticals, Inc. | Therapeutically active compositions and their method of use |
| US20130184222A1 (en) | 2010-07-16 | 2013-07-18 | Agios Pharmaceuticals, Inc | Therapeutically active compositions and their methods of use |
| JP2014509458A (en) | 2010-10-20 | 2014-04-17 | ヨタ デバイセズ アイピーアール リミテッド | Portable device |
| US8735392B2 (en) | 2010-10-21 | 2014-05-27 | Biomarin Pharmaceutical Inc. | Crystalline (8S,9R)-5-fluoro-8-(4-fluorophenyl)-9-(1-methyl-1H-1,2,4-triazol-5-yl)-8,9-dihydro-2H-pyrido[4,3,2-de]phthalazin-3(7H)-one tosylate salt |
| WO2012069503A1 (en) | 2010-11-23 | 2012-05-31 | Pierre Fabre Medicament | Derivatives of heteroarylsulfonamides, their preparation and their application in human therapy |
| WO2012074999A1 (en) | 2010-11-29 | 2012-06-07 | Galleon Pharmaceuticals, Inc. | Novel compounds as respiratory stimulants for treatment of breathing control disorders or diseases |
| US9221792B2 (en) | 2010-12-17 | 2015-12-29 | Agios Pharmaceuticals, Inc | N-(4-(azetidine-1-carbonyl) phenyl)-(hetero-) arylsulfonamide derivatives as pyruvate kinase M2 (PMK2) modulators |
| US9328077B2 (en) | 2010-12-21 | 2016-05-03 | Agios Pharmaceuticals, Inc | Bicyclic PKM2 activators |
| US9199968B2 (en) | 2010-12-29 | 2015-12-01 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| WO2012092442A1 (en) | 2010-12-29 | 2012-07-05 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| US8889667B2 (en) | 2010-12-29 | 2014-11-18 | Agios Pharmaceuticals, Inc | Therapeutic compounds and compositions |
| JP2014509459A (en) | 2011-02-04 | 2014-04-17 | 日本電気株式会社 | Radio communication system, base station apparatus, radio resource control method, and program |
| US9682080B2 (en) | 2011-05-03 | 2017-06-20 | Agios Pharmaceuticals, Inc | Pyruvate kinase activators for use in therapy |
| US10632114B2 (en) | 2011-05-03 | 2020-04-28 | Agios Pharmaceuticals, Inc. | Pyruvate kinase activators for use in therapy |
| US9980961B2 (en) | 2011-05-03 | 2018-05-29 | Agios Pharmaceuticals, Inc. | Pyruvate kinase activators for use in therapy |
| US9404081B2 (en) | 2011-05-03 | 2016-08-02 | Agios Pharmaceuticals, Inc. | Pyruvate kinase activators for use in therapy |
| WO2012151451A1 (en) | 2011-05-03 | 2012-11-08 | Agios Pharmaceuticals, Inc. | Pyruvate kinase activators for use in therapy |
| US9193701B2 (en) | 2011-05-03 | 2015-11-24 | Agios Pharmaceuticals, Inc | Pyruvate kinase activators for use in therapy |
| WO2012151452A1 (en) | 2011-05-03 | 2012-11-08 | Agios Pharmaceuticals, Inc | Pyruvate kinase activators for use in therapy |
| WO2012160034A1 (en) | 2011-05-24 | 2012-11-29 | Bayer Intellectual Property Gmbh | 4-aryl-n-phenyl-1,3,5-triazin-2-amines containing a sulfoximine group |
| US20140213580A1 (en) | 2011-06-17 | 2014-07-31 | Agios Pharmaceuticals, Inc | Therapeutically active compositions and their methods of use |
| WO2012171506A1 (en) | 2011-06-17 | 2012-12-20 | Agios Pharmaceuticals, Inc. | Compounds, their pharmaceutical compositions and their uses as idh1 mutants inhibitors for treating cancers |
| US9662327B2 (en) | 2011-06-17 | 2017-05-30 | Agios Pharmaceuticals, Inc | Phenyl and pyridinyl substituted piperidines and piperazines as inhibitors of IDH1 mutants and their use in treating cancer |
| US20140249150A1 (en) | 2011-10-13 | 2014-09-04 | Agios Pharmaceuticals, Inc | Activators of pyruvate kinase m2 and methods of treating disease |
| CN102659765A (en) | 2011-12-31 | 2012-09-12 | 沈阳药科大学 | Pyrimidine and triazine compound preparation method and application |
| WO2013102431A1 (en) | 2012-01-06 | 2013-07-11 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| US9732062B2 (en) | 2012-01-06 | 2017-08-15 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| WO2013107405A1 (en) | 2012-01-19 | 2013-07-25 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| US9474779B2 (en) | 2012-01-19 | 2016-10-25 | Agios Pharmaceuticals, Inc. | Therapeutically active compositions and their methods of use |
| WO2013107291A1 (en) | 2012-01-19 | 2013-07-25 | Agios Pharmaceuticals, Inc. | Therapeutically active compounds and their methods of use |
| US20150087600A1 (en) | 2012-01-19 | 2015-03-26 | Agios Pharmaceuticals, Inc | Therapeutically active compounds and their methods of use |
| US9487815B2 (en) | 2012-03-01 | 2016-11-08 | Dkfz Deutsches Krebsforschungszentrum | Means and methods for the determination of (D)-2-hydroxyglutarate (D2HG) or (D)-2-hydroxyadipic acid |
| WO2013133367A1 (en) | 2012-03-09 | 2013-09-12 | カルナバイオサイエンス株式会社 | Novel triazine derivative |
| EP2824099A1 (en) | 2012-03-09 | 2015-01-14 | Carna Biosciences Inc. | Novel triazine derivative |
| WO2014015422A1 (en) | 2012-07-27 | 2014-01-30 | Ontario Institute For Cancer Research | Cellulose-based nanoparticles for drug delivery |
| US10202339B2 (en) | 2012-10-15 | 2019-02-12 | Agios Pharmaceuticals, Inc. | Therapeutic compounds and compositions |
| US9579324B2 (en) | 2013-07-11 | 2017-02-28 | Agios Pharmaceuticals, Inc | Therapeutically active compounds and their methods of use |
| US20150031627A1 (en) | 2013-07-25 | 2015-01-29 | Agios Pharmaceuticals, Inc | Therapeutically active compounds and their methods of use |
| US20200030322A1 (en) | 2015-06-11 | 2020-01-30 | Agios Pharmaceuticals, Inc. | Methods of using pyruvate kinase activators |
| US11234976B2 (en) * | 2015-06-11 | 2022-02-01 | Agios Pharmaceuticals, Inc. | Methods of using pyruvate kinase activators |
| US11254652B2 (en) * | 2017-11-22 | 2022-02-22 | Agios Pharmaceuticals, Inc. | Crystalline forms of N-(4-(4-(cyclopropymethyl)piperazine-1-carbonyl)phenyl)quinoline-8-sulfonamide |
Non-Patent Citations (310)
| Title |
|---|
| Adveenko, et al., "Thiocyanation of N-arylsulfonyl-, N-aroyl-, and N-[(N-arylsulfonyl)benzimidoyl]-1,4-benzoquinone imines" Russian Journal of Organic Chemistry, vol. 45, No. 3, (2009), 408-416. |
| Aghili et al., "Hydroxyglutaric aciduria and malignant brain tumor: a case report and literature review", J Neurooncol (2009) 91:233-236. |
| Anastasiou et al., "Pyruvate kinase M2 activators promote tetramer formation and suppress tumorigenesis," Nat Chem Biol. Oct. 2012; 8(10):839-847. |
| Ansell et al., "The interactions of artificial coenzymes with alcohol dehydrogenase and other NAD(P)(H) dependent enzymes," Journal of Molecular Catalysis B: Enzymatic (1999) vol. 6, No. 1-2, pp. 111-123. |
| Balss, "Analysis of the IDH1 codon 132 mutation in brain tumors", Acata Neuropathol (2008) vol. 116, pp. 597-602. |
| Baxter I et al: "Preparation and some reactions of 6-arylsulphonimidobenzoxazol-2(3H)-one" Journal of the Chemical Society, Section C: Organic Chemistry, Chemical Society Letchworth, GB LNKD-DOI:10.1039/J39700000850, Jan. 1, 1970, pp. 850-853. |
| Beger, et. al., "Treatment of Pancreatic Cancer: Challenge of the Facts" World J. Surg., Societe Internationale de Chirurgie, vol. 27, pp. 1075-1083, 2003 |
| Behun et al., "The Chemistry of Pyrazine and Its Derivatives. IV. The Alkylation and Arylation of Methylpyrazine," J Org. Chern., 26(9),3379-3382 (1961). |
| Benesch et al., "The clinicopathological and prognostic relevance of pyruvate kinase M2 and pAkt expression in breast cancer." Anticancer Res.; 30(5):1689-94 (2010). |
| Benner et al., "Evolution, language and analogy in functional genomics", Trends in Genetics (2001) vol. 17, No. 7, pp. 414-418. |
| Beutler et al., "Elevated Pyruvate Kinase Activity in Patients with Hemolytic Anemia Due to Red Cell Pyruvate Kinase ‘Deficiency’," The American Journal of Medicine (1987) vol. 83, pp. 899-904. |
| Bhushan et al., "Reversed-phase liquid chromatographic resolution of diastereomers of protein and non-protein amino acids prepared with newly synthesized chiral derivatizing reagents based on cyanuric chloride," Amino Acids (2011) vol. 40, pp. 403-409. |
| Bleeker et al., "IDH1 Mutations at Residue p. R132 (IDH1 (R132)) Occur Frequently in High-Grade Gliomas But Not in Other Solid Tumors," Human Mutation, (2009), vol. 30, No. 1, pp. 7-11. |
| Bonuccelli et al., "The reverse Warburg effect: Glycolysis inhibitors prevent the tumor promiting effects of caveolin-1 deficient cancer associated fibroblasts." Cell Cycle.;9(10)(2010). |
| Boxer, et al., "Evaluation of Substituted N,N?-Diarylsulfonamides as Activators of the Tumor Cell Specific M2 Isoform of Pruvate Kinase", J Med Chem. Feb. 11, 2010; 53(3):1048. |
| Boxer, et al., "Identification of activators for the M2 isoform of human pyruvate kinase Version 3", Sep. 2009, Probe Reports from the NIH Molecular Libraries Program [Internet]. Bethesda (MD): National Center for Biotechnology Information (US). |
| Braun et al., "Triazine-based polymers: 4. MALDI-MS of triazine-based polyamines," Polymer (1996) vol. 37, No. 5, pp. 777-783. |
| Budinger et al., "Cellular Energy Utilization and Supply During Hypoxia in Embryonic Cardiac Myocytes," Am J Physiol. 270: L44-53 (1996). |
| Buschow et al., "MHC class II-associated proteins in B-cell exosomes and potential functional implications for exosome biogenesis." Immunol Cell Biol. (2010). |
| Cairns et al., "Oncogenic Isocitrate Dehydrogenase Mutations: Mechanisms, Models, and Clinical Opportunities," Cancer Discovery (2013) vol. 3, Iss. 7, pp. 730-741. |
| Cecil Textbook of Medicine, edited by Bennet, J.C., and Plum F., 20th edition, vol. 1, 1004-1010, 1996. |
| Chabner, et al., "Chemotherapy and the war on cancer", Nature Rev. Cancer, Nature Publishing Group, vol. 5, pp. 65-72, 2005. |
| Chan et al., "Multi-domain hydrogen-bond forming metal chelates: X-ray crystal structures of dicyclopalladated 2,3-bis[6-(2-amino-4-phenylamino-1,3,5-triazinyl)]pyrazine(H2L) [Pd2Br2L] and 2,6-bis[6-(2-amino-4-phenylamino-1,3,5-triazinylium)]-pyridine dichloride," Chem. Commun. (1996) No. 1, pp. 81-83. |
| Chan et al., "Synthesis and characterization of poly(amide sulfonamide)s (PASAs)," J Polymer. Sci., 33(15), 2525-2531 (1995). |
| Chapman et al., "Substituted aminopyrimidine protein kinase B (PknB) inhibitors show activity against Mycobacterium tuberculosis," Bioorganic & Medicinal Chemistry Letters 22 (2012), pp. 3349-3353. |
| Charache et al., "Effect of 2,3-Diphosphoglycerate on Oxygen Affinity of Blood in Sickle Cell Anemia," The Journal of Clinical Investigation (1970) vol. 49, pp. 806-812. |
| Chen et al., "Cytotoxicity, Hemolysis, and Acute in Vivo Toxicity of Dendrimers Based on Melamine, Candidate Vehicles for Delivery," J. Am. Chem. Soc. (2004) vol. 126, No. 32, pp. 10044-10048. |
| Chilean Patent Office First Office Action and English Translation for Application No. 3350-2011 dated Feb. 5, 2014. |
| Chilean Patent Office Second Office Action and English Translation for Application No. 3350-2011 dated Jan. 28, 2015. |
| Chinese Patent Office Notice on the First Review Opinion and English Translation for Application No. 201080037700.8 dated Jan. 17, 2013. |
| Chinese Patent Office Notice on the First Review Opinion and English Translation for Application No. 201180068645.3 dated Sep. 2, 2014. |
| Chinese Patent Office Second Review Opinion Notice and English Translation for Application No. 201180068645.3 dated Jun. 23, 2015. |
| Christofk et al. , "The M2 Splice Isoform of Pyruvate Kinase is Important for Cancer Metbolism and Tumour Growth," Nature 452: 230-233 (2008). |
| Christofk et al., "pyruvate Kinase M2 is a Phosphotyrosine-Binding Protein," Nature 452: 181-186 (2008). |
| Clement, et. al., "Production of Intracellular Superoxide Mediates Dithiothreitol-Dependent Inhibition of Apoptotic Cell Death" Antioxidants and Redox Signaling, Mary Ann Liebert, vol. 7, issues 3-4, pp. 456-464, 2005. |
| Cocco et al., "Synthesis of Triflouromethylated Pyridinecarbonitriles," J. Heterocyclic Chem. (1995), vol. 32, pp. 543-545. |
| Cohen et al., "The development and therapeutic potential of protein kinase inhibitors", Current Opinion in Chemical Biology, 3,459-465, 1999. |
| Conti et al., "Su alcuni analoghi ossigenati della benzo-tiazine: 2-3-diidro-3-cheto-benzo-1-4-ossazine 6-sostitute," Bollettino Scientifico Della Facolta Di Chimica Industriale Di Bologna (1957) vol. XV, No. 2, pp. 33-36. |
| Crawford et al., Caplus an 2010:1218943, Sep. 30, 2010. |
| Cuzick, et. al., "Overview of the main outcomes in breast-cancer prevention trials" The Lancet, The Lancet Publishing Group, vol. 361, pp. 296-300, 2003. |
| Dang et al., "Cancer-associated IDH1 mutations produce 2-hydroxyglutarate," Nature, Dec. 10, 2009, vol. 462, No. 7274, pp. 739-744. |
| Dang et al., "IDH mutations in glioma and acute myeloid leukemia," Trends in Molecular Medicine (2010) vol. 16, No. 9, pp. 387-397. |
| Database Chemcats, Chemical Abstracts Service, Columbus, OH, US "Bionet Screening Compounds" Key Organics Ltd., Camelford, Cornwall (2001). |
| Davis et al., "Biochemical, Cellular, and Biophysical Characterization of a Potent Inhibitor of Mutant Isocitrate Dehydrogenase IDH1," The Journal of Biological Chemistry (2014) vol. 289, No. 20, pp. 13717-13725. |
| Denham et al., "Blood diseases in the elderly," Moscow, Medicine, 1989, Chapter 15. |
| Dermer et al., "Another Anniversary for the War on Cancer", Bio/Technology, 1994, 12:320. |
| Docoslis et al., "Characterization of the Distribution, Polymorphism, and Stability of Nimodipine in Its Solid Dispersions in Polyethylene Glycol by Micro-Raman Spectroscopy and Powder X-Ray Diffraction," The AAPS Journal 2007; 9 (3) Article 43, E361-E370. |
| Dohner et al., "Impact of Genetic Features on Treatment Decisions in AML," American Society of Hematology (2011) pp. 36-42. |
| Dombrauckas, et al., Structural Basis for Tumor Pyruvate Kinasa M2 Allosteric Regulation and Catalysis, Biochemistry, vol. 44, p. 9717-9429 (2005). |
| Dong et al., "PKM2 and cancer: The function of PKM2 beyond glycolysis," Oncology Letters (2016), 11:1980-1986. |
| Duanmu et al., "Dendron-Functionalized Superparamagnetic Nanoparticles with Switchable Solubility in Organic and Aqueous Media: Matrices for Homogeneous Catalysis and Potential MRI Contrast Agents," Chem. Mater. (2006) vol. 18, No. 25, pp. 5973-5981. |
| Ecuador Patent Office Action and English Translation for Application No. SP 2011-11567 dated Apr. 17, 2019. |
| Egyptian Patent Office Action and English Translation for Application No. 2011122168 dated Apr. 28, 2019. |
| Egyptian Patent Office Decision and English Translation for Application No. 2011122168 dated Nov. 8, 2017. |
| Eigenbrodt et al., "Double Role for Pyruvate Kinase Type M2 in the Expansion of Phosphometabolite Pools Found in Tumor Cells," Crit Rev Oncog. 3: 91-115 (1992). (Abstract only). |
| Engelman et al., "Allelic Dilution Obscures Detection of a Biologically Significant Resistance Mutation in EGFR-Amplified Lung Cancer," J Clin Invest. 116: 2695-2706 (2006). |
| Eswaran et al., "Crystal Structures and Inhibitor Identifcation for PTPN5, PTPRR and PTPN7: A Family of Human MAPK-Specific Protien Tyrosine Phosphatases," Biochem J. 395: 483-491 (2006). |
| European Communication pursuant to EP Article 94(3) EPC for European Application No. 10714131.9 dated Oct. 17, 2014. |
| European Patent Office Communication (European Application No. 07836571.5), dated Oct. 18, 2010. |
| European Patent Office Communication for European Application No. 10794668.3 dated Feb. 26, 2014. |
| European Patent Office Communication for European Application No. 10794668.3 dated Nov. 18, 2014. |
| European Patent Office Communication for European Application No. 10794668.3 dated Sep. 16, 2015. |
| European Search report for EP Application No. 10 794 667.5 dated Oct. 9, 2013. |
| European Search Report for EP Application No. 10751525.6 dated Dec. 14, 2012. |
| European Search Report for EP Application No. 11763425.3 dated Sep. 23, 2013. |
| European Search Report for European Application No. 10825706.4 dated Apr. 5, 2013. |
| European Search Report for European Application No. 11808773.3 dated Apr. 9, 2014. |
| European Search Report for European Application No. 11811257.2 dated Apr. 23, 2014. |
| European Search Report for European Application No. 12799802.9 dated Oct. 9, 2014. |
| European Search Report for European Application No. 12800001.5 dated Oct. 21, 2014. |
| European Supplementary European Search Report for European Application No. 10825707.2 dated Jun. 28, 2013. |
| Extended European Search Report for European Application No. 07836571.5 dated Oct. 18, 2010. |
| Extended European Search Report for European Application No. 16152308.9 dated Jul. 18, 2016. |
| Extended European Search Report for European Application No. 17000407.1 dated Aug. 4, 2017. |
| Extended European Search Report for European Application No. 20153827.9 dated Jun. 15, 2020. |
| Fabbro et al. "Protein kinases as targets for anticancer agents: from inhibitors to useful drugs." Pharmacology & Therapeutics 93, 79-98, 2002. |
| Freshney et al.,Culture of Animal Cells, a Manual of Basic Technique, Alan R. Liss, Inc., 1983, New York, p. 4 |
| Friedman et al., "Leptin and the regulation of body weight in mammals" Nature. vol. 395, 1996. |
| Furuya et al., Inactiveation of the 3-phosphoglycerate dehydrogenase gene in mice: changes in gene expression and associated regulatory networks resulting from serive deficiency. Funct Integr Genomics (2008) 8:235-249. |
| Ge et al., "Anaplasma phagocytophilum inhibits human neutrophil apoptosis via upregulation of bfl-1, maintenance of mitochondrial membrane potential and prevention of caspase 3 activation," Cellular Microbiology (2005) 7(1), 29-38. |
| Genetics Home Reference, "L2HGDH," accessed at <http://ghr.nlm.nih.gov/gene/L2HGDH> on Sep. 4, 2015. |
| Gewald et al., "Discovery of triazines as potent, selective and orally active PDE4 inhibitors," Bioorganic & Medicinal Chemistry Letters 23 (2013), pp. 4308-4314. |
| Golub et al., "Molecular Classification of Cancer: Class Discovery and Class Prediction by Gene Expression Monitoring", Science, 286, 531-537, 1999. |
| Gupta et al., "Dominant negative mutations affect oligomerisation of human pyruvate kinase M2 isozyme and promote cellular growth and polyploidy." J Biol Chem. (2010). |
| Hartmann et al., "Type and frequency of IDH1 and IDH2 mutations are related to astrocytic and oligodendroglial differentiation and age: a study of 1,010 diffuse gliomas," Acta Neuropathol (2009) 118:469-474. |
| Hitosugi T et al: "Tyrosine Phosphorylation Inhibits PKM2 to Promote the Warburg Effect and Tumor Growth" Science Signaling, American Association for the Advancement of Science, US LNKD-DOI:10.1126/SCISIGNAL.2000431, vol. 2, No. 97, Nov. 17, 2009, pp. RA73-RA1. |
| Hitosugi, et al., "Tyrosine Phosphorylation Ingibits PKM2 to Promote the Warburg Effect and Tumor Growth" Sci. Signal., Nov. 17, 2009, vol. 2, Issue 97, p. ra73. |
| Holmes et al., "750 MHz 1H NMR spectroscopy characterisation of the complex metabolic pattern of urine from patients with inborn errors of metabolism: 2-hydroxyglutaric aciduria and maple syrup urine disease," Journal of Pharmaceutical and Biomedical Analysis 15 (1997), pp. 1647-1659. |
| Hulleman, et al., "Pyruvate kinase M2 and prednisolone resistance in acute lymphoblastic leukemia." Haematologica. Sep. 2009; 94(9): 1322-1324. |
| Indian First Examination Report for Application No. 201918008568 dated Dec. 26, 2019. |
| Indian Patent Office Examination Report for Indian Application No. 471/DELNP/2012 dated Sep. 7, 2018. |
| Indian Patent Office Hearing Notice for Indian Application No. 471/DELNP/2012 dated Nov. 13, 2019. |
| Inglese et al., "Quantitative high-throughput screening: A titration-based approach that efficiently identifies biological activities in large chemical libraries," ProC. Natl. Acad. Sci., 103 (31), 11473-11478 (2006). |
| International Preliminary Report for related application No. PCT/US2010/059778 dated Jun. 12, 2012. |
| International Preliminary Report for related application No. PCT/US2011/067752 dated Apr. 11, 2013. |
| International Preliminary Report on Patenability for PCT/US2010/040486 dated Jan. 12, 2012. |
| International Preliminary Report on patentability for International Application No. PCT/US2007/017519, issued Feb. 10, 2009. |
| International Preliminary Report on Patentability for PCT/CN2012/000841 dated Dec. 17, 2013. |
| International Preliminary Report on Patentability for PCT/CN2012/077096 dated Dec. 17, 2013. |
| International Preliminary Report on Patentability for PCT/US2008/009828, dated Feb. 16, 2010. |
| International Preliminary Report on Patentability for PCT/US2010/027253 dated Sep. 13, 2011. |
| International Preliminary Report on Patentability for PCT/US2010/053623 dated Apr. 24, 2012. |
| International Preliminary Report on Patentability for PCT/US2010/053624 dated Apr. 24, 2012. |
| International Preliminary Report on Patentability for PCT/US2011/030692 dated Oct. 2, 2012. |
| International Preliminary Report on Patentability, Application No. PCT/US2009/060237, dated Apr. 12, 2011. |
| International Search Report & Written Opinion for PCT/US10/030139 dated Dec. 10, 2010. |
| International Search Report & Written Opinion for PCT/US10/40485 dated Aug. 11, 2010. |
| International Search Report and the Written Opinion of the International Search Authority (PCT/US07/17519), mailed Jul. 8, 2008. |
| International Search Report and Written Opinion for International Application No. PCT/CN2013/080105 dated Jul. 11, 2014. |
| International Search Report and Written Opinion for International Application No. PCT/CN2013/081170 dated Apr. 30, 2014. |
| International Search Report and Written Opinion for International Application No. PCT/CN2014/081957 dated Sep. 30, 2014. |
| International Search Report and Written Opinion for International Application No. PCT/CN2014/081958 dated Sep. 29, 2014. |
| International Search Report and Written Opinion for International Application No. PCT/CN2014/082869 dated Sep. 30, 2014. |
| International Search Report and Written Opinion for International Application No. PCT/US2012/036412 dated Jul. 6, 2012. |
| International Search Report and Written Opinion for International Application No. PCT/US2012/036413 dated Jul. 6, 2012. |
| International Search Report and Written Opinion for International Application No. PCT/US2013/064601 dated Feb. 24, 2014. |
| International Search Report and Written Opinion for International Application No. PCT/US2014/046202 dated Sep. 30, 2014. |
| International Search Report and Written Opinion for International Application No. PCT/US2014/049469 dated Jan. 22, 2015. |
| International Search Report and Written Opinion for International Application No. PCT/US2015/020346 dated Jun. 18, 2015. |
| International Search Report and Written Opinion for International Application No. PCT/US2015/020349 dated Jun. 15, 2015. |
| International Search Report and Written Opinion for PCT/CN2013/000009 dated Apr. 18, 2013. |
| International Search Report and Written Opinion for PCT/CN2013/070755 dated Apr. 25, 2013. |
| International Search Report and Written Opinion for PCT/US2010/053624 dated Apr. 7, 2011. |
| International Search Report and Written Opinion for PCT/US2011/044254 dated Oct. 5, 2011. |
| International Search Report dated Apr. 4, 2012 for related Application PCT/US2011/065633. |
| International Search Report dated Mar. 5, 2012 for related international application No. PCT/US2011/067752. |
| International Search Report dated May 3, 2012 for related application PCT/US2011/066595. |
| International Search Report dated Sep. 1, 2010 in related application—PCT/US10/40486. |
| International Search Report for Application No. PCT/US12/60099 dated Jan. 8, 2013. |
| International Search Report for International Application No. PCT/CN2013/079184 dated Jan. 12, 2015. |
| International Search Report for International Application No. PCT/CN2013/079200 dated Jan. 12, 2015. |
| International Search Report for International Application No. PCT/US2014/046204 dated Oct. 1, 2014. |
| International Search Report for PCT/CN2012/000841 dated Sep. 27, 2012. |
| International Search Report for PCT/CN2012/077096 dated Oct. 4, 2012. |
| International Search Report for PCT/CN2013/000068 dated Apr. 25, 2013. |
| International Search Report for PCT/US10/40486 dated Sep. 1, 2010. |
| International Search Report for PCT/US2008/009828, dated Dec. 5, 2008. |
| International Search Report for PCT/US2010/027253 dated Aug. 19, 2010. |
| International Search Report for PCT/US2010/033610 dated Jul. 22, 2010. |
| International Search Report for PCT/US2010/059778 dated Mar. 17, 2011. |
| International Search Report for PCT/US2010/53623 dated Jan. 18, 2011. |
| International Search Report for PCT/US2011/030692 dated Jun. 27, 2011. |
| International Search Report for PCT/US2011/065633 dated Jun. 18, 2013. |
| International Search Report, Application No. PCT/US2009/060237, dated Jun. 16, 2010. |
| International Search Report, Application No. PCT/US2011/033852, dated Aug. 3, 2011. |
| Irikura et al., "New s-Triazine Derivatives as Depressants for Reticuloendothelial Hyperfunction Induced by Bacterial Endotoxin," Journal of Medicinal Chemistry (1970) vol. 31, pp. 1081-1089. |
| Japanese Patent Office Decision of Rejection and English Translation for Application No. 2012-518591 dated Apr. 30, 2015. |
| Japanese Patent Office Notification of Reasons for Rejection and English Translation for Application No. 2012-518591 dated Aug. 1, 2014. |
| Jennings et al., "Expression and Mutagenesis of Mammalian Cytosolic NADP+-Specific Isocitrate Dehydrogenase," Biochemistry (1997) vol. 36, pp. 13743-13747. |
| Jiang et al., "Evaluation of theino [3,2-b]pyrrole[3,2-djpyridazinones as activators of the tumor cell specific M2 isoform of pyruvate kinase." Bioorg. Med. Chern. Lett., 20(11), 3387-3393 (2010). |
| Joshi et al., "Age-related faecal calprotectin, lactoferrin and tumour M2-PK concentrations in healthy volunteers." Ann Clin Biochem.;47(Pt 3):259-63 (2010). |
| Jurica et al., "The Allosteric Regulation of Pyruvate Kinase by Fructose-1 ,6-Bisphosphate," Structure 6: 195-210 (1998). |
| Kaila et al., "A convenient one-pot synthesis of trisubstituted 1,3,5-triazines through intermediary amidinothioureas," Tetrahedron Letters 51 (2010) pp. 1486-1489. |
| Kao et al., "A Small-Molecule Inhibitor of the Ribonucleolytic Activity of Human Angiogenin That Possesses Antitumor Activity," Proc. Nat!. Acad. Sci. USA, 99(15): 10066-10071 (2002). |
| Kelarev et al., "Synthesis and properties of sym-triazines. 10 Synthesis of 2,4-diamino-sym-triazines containing a sterically hindered phenol substituent," Chemistry of Heterocyclic Compounds (1992) vol. 28, No. 10, pp. 1189-1193. |
| Kharalkar et al., "Identification of Novel Allosteric Regulators of Human-Erythrocyte Pyruvate Kinase," Chem Biodivers. 4: 2603-2617 (2007). |
| Kim et al., "Identification and Functional Characterization of a Novel, Tissue-specific NAD+-dependent Isocitrate Dehydrogenase β Subunit Isoform," The Journal of Biological Chemistry, Dec. 24, 1999, vol. 274, No. 52, p. 36866-36875. |
| Kim et al., "Ser95, Asn97, and Thr78 are important for the catalytic function of porcine NADP-dependent isocitrate dehydrogenase," Protein Science (2005) 14:140-147. |
| Klapars et al., "A General and Efficient Copper Catalyst for the Amidation of Aryl Halides and the N-Arylation of Nitrogen Heterocycles," J. Am. Chem. Soc., 123 (31), 7727-7729 (2001). |
| Komoriya et al. "Design, synthesis, and biological activity of non-basic compounds as factor Xa inhibitors: SAR Study of S1 and aryl binding sites" Bioorganic & Medicinal Chemistry 13 (2005) 3927-3954. |
| Koshelev et al.,"Synthesis of N-Substituted 2,4-Diamino-1,3,5-triazines Containing Pyridyl Groups," Russian Journal of Organic Chemistry (1995) vol. 31, No. 2, pp. 260-263. |
| Kranendijk et al., "IDH2 Mutations in Patients with D-2-Hydroxyglutaric Aciduria," Science (2010) vol. 330, p. 336. |
| Krell et al., "IDH mutations in tumorigenesis and their potential role as novel therapeutic targets," Future Oncol. (2013) 9(12), pp. 1923-1935. |
| Kumar et al., "In vivo factors influencing tumour M2-pyruvate kinase level in human pancreatic cancer cell lines." Tumour Biol.;31(2):69-77 (2010). |
| Kung et al. "Small Molecule Activation of PKM2 in Cancer Cells Induces Serine Auxotrophy" Chemistry & Biology, 19, 1187-1198, Sep. 21, 2012. |
| Kung, Charles et al., "AG-348 Activation of Pyruvate Kinase in Vivo Enhances Red Cell Glycolosis in Mice," Database Biosis [Online], database accession No. PREV201500280942, vol. 124, No. 21, 56th Annual Meeting of the American-Society-of-Hematology, San Francisco, CA, 2014. |
| Kusakabe et al., Chemical Abstracts vol. 152, No. 191956, Abstract for WO2010007756 (2010). |
| Lee et al., "An Efficient Synthesis of 2,8-Diazabicyclo[4.3.0]-Nonane Derivatives Via Intramolecular Cyclization Reaction," Synth. Comm., 25 (23), 3741-3746 (1995). |
| Lee et al., "Combinatorial Solid-Phase Synthesis of 6-Aryl-1,3,4-triazines via Suzuki Coupling," Aust. J. Chem. (2011) 64, pp. 540-544. |
| Lee, "Consolidation Effect of Phenylalanine-administration of Anti-tumor Activity of A 5 Fluorouracil," Med. J. Kagoshima Univ. 37(3-4): 285-308 (1985). |
| Lee, et al., "Pyruvate kinase isozyme type M2 (PKM2) interacts and cooperates with Oct-4 in regulating transcription" International J. Biochem. & Cell Biol., vol. 40, # 5,2008, 1043-1054. |
| Li et al., "Quantitative proteome analysis of multidrug resistance in human ovarian cancer cell line." J Cell Biochem.;109(4):625-33 (2010). |
| Li et al., "Screening and identification of interactive proteins of SH2D4A." Yi Chuan.;32(7):712-8 (2010). |
| Liu et al., "Inhibition of Cancer-Associated Mutant Isocitrate Dehydrogenases: Synthesis, Structure—Activity Relationship, and Selective Antitumor Activity," J. Med. Chem. (2014), vol. 57, pp. 8307-8318. |
| Lou, "IDH1: function follows form," SciBX, 2009, pp. 1-2. |
| Luo et al., "Synthesis and Fungicidal Activity of N-Benzo[b][1,4]oxazin-6-y1-2,4-dimethylthiazole-5-carboxamides," Agrochemicals (2009) vol. 48, No. 1, pp. 19-22. |
| Lutker et al., "Crystal Polymorphism in a Carbamazepine Derivative: Oxcarbazepine," NIH Public Access, J Pharm Sci. Feb. 2010; 99(2): 794-803. doi:10.1002/jps.21873. |
| Madsen-Duggan et al., "Lead optimization of 5,6-diarylpyridines as CB1 receptor inverse agonists," Bioorganic & Medicinal Chemistry Letters 17 (2007) pp. 2031-2035. |
| Marry et al., "Human Biochemistry," Moscow, Mir, 1993, Chapter 18. |
| Mass, R. D., "The HER receptor family: a rich target for therapeutic development", Int. J. Radiation Oncology Bio. Phys.vol. 58(3): 932-940, 2004. |
| May, "How Many Species Are There on Earth," Science (1988) vol. 241, p. 1441. |
| Moreno et al., "Identification of diamine linkers with differing reactivity and their application in the synthesis of melamine dendrimers," Tetrahedron Letters 49 (2008), pp. 1152-1154. |
| Moreno et al., "Molecular recognition in dendrimers based on melamine," Polymer Preprints (2005) vol. 46, No. 2, p. 1127. |
| Morshed et al., "Computational approach to the identification of novel Aurora-A inhibitors," Bioorganic & Medicinal Chemistry (2011) vol. 19, No. 2, pp. 907-916. |
| Oeda, "On some 2,5-Dialikl-piperazines," Bull. Chem. Soc., 13, 465-470 (1938). |
| Pan et al., "Research Status of Pyruvate Deficiency," Chinese Journal of Hematology (1999) vol. 20, No. 4, pp. 223. |
| Paraguay Office Action and English Translation for Application No. 25374/2010 dated Sep. 18, 2019. |
| Park, "Prevention of type 2 diabetes mellitus from the viewpoint of genetics." Diabetes Research and Clinical Practice 2004; 66S: S33-S35. |
| Paronikyan et al., "Synthesis and biological activity of 3-piperazinyl-substituted pyrano [3,4-C] pyridines," Armyanskii Khimicheskii Zhurnal (1990) vol. 43, No. 8, pp. 518-523. |
| Parsons et al., "An Integrated Genomic Analysis of Human Glioblastoma Multiforme," Science, vol. 321 (2008) pp. 1807-1812 and Supplemental Data. |
| Patel et al. "Synthesis of some new idolinone derivatives containing piperazine moiety" Bulgarian Chemical Communications, 2003 Bol 35 No. 4 pp. 242-244. |
| Paudler et al., "3,7-Disubstituted octahydro-1,5-diazocines. Their converstion into tetrahydro-1 ,5-diazocines and into ring-contracted products," J. Org. Chern., 32 (8), 2425-2430 (1967). |
| Petz et al., "Increased IgG Molecules Bound to the Surface of Red Blood Cells of Patients With Sickle Cell Anemia," Blood (1984) vol. 64, No. 1, pp. 301-304. |
| Pollard et al., "Cancer. Puzzling patterns of predisposition," Science, Apr. 10, 2009, vol. 324, 1-5, 15-16, 18-22, 35-38, No. 5924, pp. 192-194. |
| Pollard et al., "Some Amides of Piperazines," J. Am. Chem. Soc., 75(2), 491 (1953). |
| Popovici-Muller, Janeta et al., "Discovery of the First Potent Inhibitors of Mutant IDH1 That Lower Tumor 2-HG in Vivo," ACS Medicinal Chemistry Letters, Sep. 17, 2012, vol. 3, No. 10, pp. 850-855. |
| Proisy et al. "Rapid Synthesis of 3-Aminoisoquinoline-5-sulfonamides Using the Buchwald_Hartwig Reaction" Synthesis 2009, No. 4, pp. 0561-0566. |
| Pubchem CID 4078245 [online]; Sep. 13, 2005 [retrieved on Feb. 4, 2012]; retrieved from http://pubchem.ncbi.nlm.nih.gov/; 2d-structure. |
| Pubchem CID 4854170 [online]; Sep. 17, 2005 [retrieved on Feb. 4, 2012]; retrieved from http://pubchem.ncbi.nlm.nih.gov/; 2d-structure. |
| Pujol, et. al., "Is there a case for cisplatin in the treatment of smallcell lung cancer? A meta-analysis of randomized trials of a cisplatin-containing regimen versus a regimen without this alkylating agent" British Journal of Cancer, Cancer Research Campaign, vol. 83, issue 1, pp. 8-15, 2000. |
| Rao et al., "Polymorphism in Drugs and its Significance in Therapeutics," Journal of Scientific & Industrial Research, vol. 46, Oct. 1987, pp. 450-455. |
| Raynaud et al., "Absence of R140Q mutation of isocitrate dehydrogenase 2 in gliomas and breast cancers," Oncology Letters (2010) vol. 1, No. 5, pp. 883-884. |
| Registry (STN) [online], Apr. 13, 2007 [Retrieved on Jan. 29, 2016] CAS Registration No. 929819-92-1. |
| Registry (STN) [online], Apr. 13, 2007 [Retrieved on Jan. 29, 2016] CAS Registration No. 929971-43-7. |
| Registry (STN) [online], Apr. 19, 2009 [Retrieved on Jan. 29, 2016] CAS Registration No. 1136498-70-8. |
| Registry (STN) [online], Aug. 23, 2006 [Retrieved on Jan. 29, 2016] CAS Registration No. 903862-76-0. |
| Registry (STN) [online], Aug. 23, 2006 [Retrieved on Jan. 29, 2016] CAS Registration No. 903869-26-1. |
| Registry (STN) [online], Aug. 27, 2009 [Retrieved on Jan. 29, 2016] CAS Registration No. 1176756-98-1. |
| Reitman et al., "Isocitrate Dehydrogenase 1 and 2 Mutations in Cancer: Alterations at a Crossroads of Cellular Metabolism," Journal of the National Cancer Institute (2010), vol. 102, No. 13, pp. 932-941. |
| Remington's, "Structure Activity Relationship and Drug Design," Pharmaceutical Sciences, pp. 420-425, 1980. |
| Rich, et. al., "Development of novel targeted therapies in the treatment of malignant glioma" Nature Rev. Drug Disc., Nature Publishing Group, vol. 3, pp. 430-446, 2004. |
| Rohle et al., "An Inhibitor of Mutant IDH1 Delays Growth and Promotes Differentiation of Glioma Cells," Science (2013), vol. 340, No. 6132, pp. 626-630. |
| Root et al., "Genome-Scale Loss-of-Function Screening with a Lentiviral RNAi Library," Nat Methods 3: 715-719 (2006). |
| Ruan et al., "HSP60, a protein downregulated by IGFBP7 in colorectal carcinoma." J Exp Clin Cancer Res.;29-41 (2010). |
| Sabatine et al., "Metabolomic Identification of Novel Biomarkers of Myocardial Ischemia," Circulation 112: 3868-3875 (2005). |
| Scharn et al., "Spatially Addressed Synthesis of Amino- and Amino-Oxy-Substituted 1,3,5-Triazine Arrays on Polymeric Membranes," J. Comb. Chem. (2000) vol. 2, No. 4, pp. 361-369. |
| Schneider, et al., "Tumor M2-pyruvate kinase in the follow-up of inoperable lung cancer patients: a pilot study." Cancer Letters, Elsevier, vol. 193, pp. 91-98, 2003. |
| Schroth et al., "RingschluBreaktion von Diacetylen mit Diaminen: Eine Ciniache von 2,3-Dihydro-1 ,4-diazepinen," Zeitschritt Fur Chemie., 6 (4), 143 (1969). |
| Seibel et al., "Synthesis and evaluation of B-lactams (piperazones) as elastase inhibitors," Bioorg. Med. Chern. Ltrs., 13 (3),387-389 (2003). |
| Shi, et al., "Silencing of pkm2 increases the efficacy of docetaxel in human lung cancer xenografts in mice." Cancer Science, vol. 101, #6, 1447-1453, Jun. 2010. |
| Shih et al., "The role of mutations in epigenetic regulators in myeloid malignancies," Nature Reviews Cancer (2012) vol. 12, No. 9, pp. 599-612. |
| Sirakanyan, S.N. et al., "Synthesis of new derivatives of piperazine-substituted pyrano[3,4-c]pyridines," Hayastani Kimiakan Handes 2009, vol. 62, No. 3-4, pp. 378-385. English Abstract Only. |
| Sonoda et al., "Analysis of IDH1 and IDH2 mutations in Japanese glioma patients," Cancer Science (2009), vol. 100, No. 10, pp. 1996-1998. |
| Sosnovik et al., "Emerging concepts in molecular MRI," Current Opinion in Biotechnology 2007, 18:4-10. |
| Steiner et al., "Synthesis and Antihypertensive Activity of New 6-Heteroaryl-3-hydrazinopyridazine Derivatives," J. Med. Chem. (1981), vol. 24, No. 1, pp. 59-63. |
| Stewart et al., "Piperazines. I. Derivavtives of Piperazine-1-Carboxylic and -1,4-Dicarboxylic Acid,", J. Org. Chern., 18(1),1478-1483 (1953). |
| STN File CA, Registry No. 1023444-33-8, entered STN on May 29, 2008, Chemical Abstracts Index Name "Benzenesulfonamide, 3-[[4-(1,3-benzodioxol-5-ylmethyl)-1-piperazinyl]carbonyl]-N-(4-butylphenyl)-4-methyl-". |
| STN File CA, Registry No. 1090629-29-0, entered STN on Dec. 28, 2008, Chemical Abstracts Index Name "Benzenesulfonamide, 3-[[4-[(2,5-dimethoxyphenyl)methyl]-1-piperazinyl]carbonyl]-N-(4-methoxyphenyl)-4-methyl-". |
| STN File CA, Registry No. 134538-28-6, entered STN on Jun. 28, 1991, Chemical Abstracts Index Name "1H-Pyrano[3,4-c]pyridine-5-carbonitrile,3,4-dihydro-3,3-dimethyl-6-[4-(1-oxobutyl)-1-piperazinyl]-8-phenyl-", disclosed in Paronikyan et al. Armyanskii Khimicheskii Zhurnal, 1990, vol. 43, No. 8. |
| STN File CA, Registry No. 134538-29-7, entered STN on Jun. 28, 1991, Chemical Abstracts Index Name "1H-Pyrano[3,4-c]pyridine-5-carbonitrile,3,4-dihydro-3,3-dimethyl-6-[4-(2-methyl-1-oxopropyl)-1-piperazinyl]-8-phenyl-", disclosed in Paronikyan et al. Armyanskii Khimicheskii Zhurnal, 1990, vol. 43, No. 8. |
| STN File CA, Registry No. 134538-30-0, entered STN on Jun. 28, 1991, Chemical Abstracts Index Name "1H-Pyrano[3,4-c]pyridine-5-carbonitrile,6-(4-benzoyl-1-piperazinyl)-3,4-dihydro-3,3-dimethyl-8-phenyl-", disclosed in Paronikyan et al. Armyanskii Khimicheskii Zhurnal, 1990, vol. 43, No. 8. |
| STN File CA, Registry No. 134538-31-1, entered STN on Jun. 28, 1991, Chemical Abstracts Index Name "1H-Pyrano[3,4-c]pyridine-5-carbonitrile,6-[4-(2-furanylcarbonyl)-1-piperazinyl]-3,4-dihydro-3,3-dimethyl-8-phenyl-", disclosed in Paronikyan et al. Armyanskii Khimicheskii Zhurnal, 1990, vol. 43, No. 8. |
| STN File CA, Registry No. 321433-63-0, entered STN on Feb. 12, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-phenyl" Available through Key Organics (under the Bionet brand) Jan. 1994. |
| STN File CA, Registry No. 321433-64-1, entered STN on Feb. 12, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-(4-methphenyl)" Available through Key Organics (under the Bionet brand) Jan. 1994. |
| STN File CA, Registry No. 321433-65-2, entered STN on Feb. 12, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-(3,5-dimethylphenyl)" Available through Key Organics (under the Bionet brand) Jan. 1994. |
| STN File CA, Registry No. 321433-66-3, entered STN on Feb. 12, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-(4-methoxyphenyl)" Available through Key Organics (under the Bionet brand) Jan. 1994. |
| STN File CA, Registry No. 321433-68-5, entered STN on Feb. 12, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-propyl" Available through Key Organics (under the Bionet brand) Jan. 1994. |
| STN File CA, Registry No. 321433-69-6, entered STN on Feb. 12, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-(2-methoxyethyl)" Available through Key Organics (under the Bionet brand) Jan. 1994. |
| STN File CA, Registry No. 338397-92-5, entered STN on May 25, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N,N-dimethyl" Available through Key Organics (under the Bionet brand) Feb. 1993. |
| STN File CA, Registry No. 338397-95-8, entered STN on May 25, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, N-[(4-chlorophenyl)-1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]" Available through Key Organics (under the Bionet brand) Feb. 1993. |
| STN File CA, Registry No. 338397-96-9, entered STN on May 25, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonic acid, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-4-chlorophenyl ester" Available through Key Organics (under the Bionet brand) Feb. 1993. |
| STN File CA, Registry No. 338406-58-9, entered STN on May 25, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-[2-(trifluoromethyl)phenyl]" Available through Key Organics (under the Bionet brand) Mar. 1993. |
| STN File CA, Registry No. 338406-64-7, entered STN on May 25, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-(3-pyridinylmethyl)" Available through Key Organics (under the Bionet brand) Mar. 1993. |
| STN File CA, Registry No. 338406-72-7, entered STN on May 25, 2001, Chemical Abstracts Index Name "1H-Pyrrole-2-sulfonamide, N-[(4-chlorophenyl)methyl]-1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]" Available through Key Organics (under the Bionet brand) Mar. 1993. |
| STN File CA, Registry No. 338407-11-7, entered STN on May 25, 2001, Chemical Abstracts Index Name "1H=Pyrrole-2-sulfonamide, 1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-N-[3-chloro-4-[[5-(trifluoromethyl)-2-pyridinyl]oxy]phenyl]" Available through Key Organics (under the Bionet brand) Mar. 1993. |
| STN File CA, Registry No. 338407-13-9, entered STN on May 25, 2001, Chemical Abstracts Index Name "Benzoic acid, 3-[[[1-[3-chloro-5-(trifluoromethyl)-2-pyridinyl]-1H-pyrrol-2-yl]sulfonyl]amino]" Available through Key Organics (under the Bionet brand) Mar. 1993. |
| STN File CA, Registry No. 380466-24-0 entered STN on Jan. 4, 2002, Chemical Abstracts Index Name "Benzenesulfonamide, N-methyl-N-phenyl-3-[[4-(2-pyridinyl)-1-piperazinyl]carbonyl]". |
| STN File CA, Registry No. 713505-78-3, entered STN on Jul. 21, 2004, Chemical Abstracts Index Name "1-Piperazinecarboxylic acid, 4-[4-methyl-3-[(phenylamino)sulfonyl]benzoyl]-, ethyl ester". |
| STN File CA, Registry No. 736168-79-9 entered STN on Aug. 31, 2004, Chemical Abstracts Index Name "Benzenesulfonamide, 3,4-difluoro-N-[3-334-(phenylmethyl0-1-piperazinyl]carbonyl]phenyl". |
| STN File CA, Registry No. 847757-57-7, entered STN on Apr. 1, 2005, Chemical Abstracts Index Name "Benzenesulfonamide, 3-[[4-(1,3-benzodioxol-5-ylmethyl)-1-piperazinyl]carbonyl]-N-(4-ethoxyphenyl)-N,4-dimethyl-" or "Piperazine, 1-(1,3-benzodioxol-5-ylmethyl)-4-[5- [[(4-ethoxyphenyl)methylamino]sulfonyl]-2-methylbenzoyl]-". |
| STN Registry, L23 Answer 2 of 3 (Cas No. 1032450-21-7), Database: ASINEX Ltd., Entered STN: Jul. 3, 2008 (Jul. 3, 2008). |
| STN Registry. L23 Answer 1 of 3 (Cas No. 1038821-72-5), Database: ChemDB (University of California Irvine), Entered STN: Aug. 5, 2008 (Aug. 5, 2008). |
| STN Tokyo, Registry No. 1001833-18-6, Entered STN on Feb. 6, 2008, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [(4-methyl-1-piperazinyl)carbonyl]phenyl]--". |
| STN Tokyo, Registry No. 1030142-35-8, Entered STN on Jun. 24, 2008, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [[4-[(5-methyl-3-isoxazolyl)methyl]-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 1031531-78-8, Entered STN on Jun. 29, 2008 Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, N-4[4-[(4-acetyl-1-piperazinyl)carbonyl]phenyl]-2,3-dihydro-". |
| STN Tokyo, Registry No. 1057928-35-4, Entered STN on Oct. 7, 2008, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [[4-(2-pyridinyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 1240875-00-6, entered STN on Sep. 14, 2010, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4-[[4-(2-thiazolyl)-1-piperazinyl] carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 748791-86-8, Entered STN on Sep. 21, 2004, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, N-[4- [[4-(2-furanylcarbonyl)-1-piperazinyl]carbonyl]phenyl]-2,3-dihydro-". |
| STN Tokyo, Registry No. 878469-24-0, Entered STN on Mar. 29, 2006, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4-[[4-(2-pyrimidinyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 878474-39-6, Entered STN on Mar. 29, 2006, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4[(4-phenyl-1-piperazinyl)carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 878590-33-1, Entered STN on Mar. 30, 2006, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4-{{4-(tetrahydro-2-furanyl)methyl]-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 878943-66-9 Entered STN on Apr. 2, 2006, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 3,4-dihydro-N-[[4-(2-pyrimidinyl)-1-piperazinyl)carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 878956-06-0, Entered STN on Apr. 2, 2006, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, N-[4- [[4-(cyclopropylcarbonyl)-1-piperazinyl]carbonyl]phenyl]-2,3 -dihydro-". |
| STN Tokyo, Registry No. 920679-46-5, Entered STN on Feb. 13, 2007, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [[4-(4-pyridinyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 920822-52-2, Entered STN on Feb. 14, 2007, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, N-[4- [[4-(4-fluoropheyl)-1-piperazinyl]carbonyl]phenyl]-2,3dihydro-". |
| STN Tokyo, Registry No. 920824-56-2, Entered STN on Feb. 14, 2007, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [[4-(3-thienylmethyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 920847-34-3, Entered STN on Feb. 14, 2007, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [[4-(2-methylphenyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 920875-39-4, Entered STN on Feb. 14, 2007, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [[4-(2-hydroxyphenyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 920902-88-1, Entered STN on Feb. 14, 2007, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [[4-(2-thienylmethyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 920921-09-1 Entered STN on Feb. 14, 2007, Chemical Abstracts Index Name "2H-1, 5-Benzodioxepin-7-sulfonamide, 3,4-dihydro-N-[4-[[4-(2pyridinyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 920924-42-1, Entered STN on Feb. 14, 2007, Chemical Abstracts Index Name "1,4-Benzodioxin-6-sulfonamide, 2,3-dihydro-N-[4- [[4-(2-pyridinylmethyl)-1-piperazinyl]carbonyl]phenyl]-". |
| STN Tokyo, Registry No. 941220-77-5, Entered STN on Jul. 4, 2007, Chemical Abstracts Index Name "2H-1, 5-Benzodioxepin-7-sulfonamide, 3,4-dihydro-N-[4-[(4-methyl-1-piperazinyl)carbonyl]phenyl]-". |
| Struys et al., "Mutations in the D-2-Hydroxyglutarate Dehydrogenase Gene Cause D-2-Hydroxyglutaric Aciduria," Am. J. Hum. Genet. (2005), 76:358-360. |
| Struys et al., Investigations by mass isotopomer analysis of the formation of D-2-hydroxyglutarate by cultured lymphoblasts from two patients with D-2-hydroxyglutaric aciduria, FEBS Letters 557 (2004), pp. 115-120. |
| Struys, et al., "Measurement of Urinary D- and L-2-Hydroxyglutarate Enantiomers by Stable-Isotope-Dilution Liquid Chromatography-Tandem Mass Spectrometry after Derivatization with Diacetyl-L-Tartaric Anhydride," Clinical Chemistry (2004), 50:8, pp. 1391-1395. |
| Supplemental EP Search Report & Written Opinion for EP 10 79 4667, Jan. 22, 2013. |
| Supplementary Search Report for EP10794668 Mailed Oct. 18, 2012. |
| Surh, "Cancer Chemoprevention with Dietary Phytochemicals", Nature Reviews Cancer, Nature Publishing Group, vol. 3, p. 768-780, 2003. |
| Szoka et al., "Comparative properties and methods of preparation of lipid vesicles (liposomes )," Ann. Rev. Biophys. Bioeng., 9, 467-508 (1980). |
| Takagi et al., "Synthesis of poly(triazinylstyrene) containing nitrogen-based ligand and function as metal ion adsorbent and oxidation catalyst," Reactive & Functional Polymers 66 (2006) vol. 31, pp. 1718-1724. |
| Tawaka, et al., Caplus an 1998:794998, 1998. |
| The radiation fact sheet published by the National Cancer Institute, http://www.cancer.gov/about-cancer/treatment/types/radiation-therapy/radiation-fact-sheet, reviewed Jun. 30, 2010. |
| Thompson, "Metabolic Enzymes as Oncogenes or Tumor Suppressors," The New England Journal of Medicine, Feb. 19, 2009, vol. 360, No. 8, pp. 813-815, col. 1; Fig. 1. |
| Tsujino, Kumiko et al., "CBA-Pk-1slc/Pk-1slc mutant mouse in Newborn period does not exhibit hemolytic anemia," Japanese Society of Animal Models for Human Diseases, 1998, vol. 14, p. 24. |
| Uozumi et al., "Catalytic asymmetric construction of morpholines and piperazines by palladium-catalyzed tandem allylic substitution reactions," J. Org. Chem., 58 (24),6826-6832 (1993). |
| Van Emelen et al., Caplus am 2003:737742. |
| Van Schaftingen, et al., "L-2-Hydroxyglutaric aciduria, a disorder of metabolite repair," J Inherit. Metab. Dis. (2009) vol. 32, pp. 135-142. |
| Vander Hedien et al., "Identification of Small Molecule Inhibitors of Pyruvate Kinase M2," Biochemical Pharmacology. 79(8): 1118-1124 (2010). |
| Vander Heiden et al., "Growth Factors Can Influence Cell Growth and Survival Through Effects on Glucose Metabolism," Mol Cell Bioi. 21: 5899-5912 (2001). |
| Villen et al., "Large-Scale Phosphorylation Analysis of Mouse Liver," Proc Nat!Acad Sci USA 104: 1488-1493 (2007). |
| Villoutreix et al., Caplus an 2010:20993, Jan. 7, 2010. |
| Walsh et al. "2-oxo-N-aryl-1,2,3,4-tetrahydroquinoline-6-sulfonamides as activators of the tumor cell specific M2 isoform of pyruvate kinase" Bioorg Med Chem Lett. Nov. 1, 2011; 21(21):6322-6327. |
| Wang et al., "A novel ligand N,N'-di(2-pyridyl)-2,4-diamino-6-phenyl-1,3,5-triazine (dpdapt) and its complexes: [Cu(dpdapt)Cl2] and [Cu(dpdapt)(NO3)(H2O)] NO3 H2O," Polyhedron (2006) vol. 25, Issue 1, pp. 195-202. |
| Wang et al., "Facile Synthesis of 2,4-Diamino-6-alkyl- or 6-Aryl-Pyrimidine Derivatives," J. Heterocyclic Chem. (2010) vol. 47 pp. 1056-1061. |
| Ward et al., "The Common Feature of Leukemia-Associated IDH1 and IDH2 Mutations Is a Neomorphic Enzyme Activity Converting [alpha]-Ketoglutarate to 2-Hydroxyglutarate," Cancer Cell (2010), vol. 17, No. 3, pp. 225-234. |
| Watanabe et al., "IDH1 Mutations Are Early Events in the Development of Astrocytomas and Oligodendrogliomas," The American Journal of Pathology, Apr. 2009 (published online Feb. 26, 2009, vol. 174, No. 4, pp. 1149-1153; Abstract, p. 1150, col. 1. |
| Web posting, Pyruvate kinase M2 isozyme (PKM2), SciBX 5(42), Published online Oct. 25, 2012, Abstract only. |
| Wong et al., "PKM2, a Central Point of Regulation in Cancer Metabolism," International Journal of Cell Biology (2013), vol. 2013, pp. 1-11. |
| Written Opinion for PCT/US2010/027253 dated Aug. 19, 2010. |
| Written Opinion of Search Authority for PCT/US2010/53623 dated Jan. 18, 2011. |
| Written Opinion of the International Searching Authority for PCT/US2008/009828, dated Dec. 5, 2008. |
| Written Opinion of the International Searching Authority for PCT/US2011/067752 dated Mar. 5, 2012. |
| Yamada and Noguchi, "Nutrient and Hormonal Regulation of Pyruvate Kinase Gene Expression," Biochem J. 337: 1-11 (1999). |
| Yamaoka, Takashi, Adenosine deaminase hyperkinasia, Nihon Rinsho (supplementary volume) series of Syndrome for each area 20 Blood Syndrome I, Aug. 12, 1998, p. 308-311. |
| Yan et al., "IDH1 and IDH2 Mutations in Gliomas." The New England Journal of Medicine, 19 Feb. 18-22, 2009, vol. 360, No. 8, pp. 765-773. |
| Yang, Hua et al., "Phase I Single (SAD) and Multiple Ascending Dose (MAD) Studies of the Safety, Tolerability, Pharmacokinetics (PK) and Pharmacodynamics (PD) of AG-348, a First-in-Class Allosteric Activator of Pyruvate Kinase-R, in Healthy Subjects," Database Biosis [Online], database accession No. PREV201500280858, vol. 124, No. 21, 56th Annual Meeting of the American-Society-of-Hematology, San Francisco, CA, 2014. |
| Yar et al., "An Annulation Reaction for the Synthesis of Morpholines, Thiomorpholines, and Piperazines from !3-Heteroatom Amino Compounds and Vinyl Sulfonium Salts," Angewandte Chemie., 47(20),3784-3786 (2008). |
| Ye et al., Pyruvate kinase M2 promotes de novo serive synthesis to sustain mTORC1 activity and cell proliferation, PNAS 109(18), 2012, pp. 6904-6909. |
| Zhao et al., "Glioma-Derived Mutations in IDH1 Dominantly Inhibit IDH1 Catalytic Activity and Induce HIF-1alpha," Science, vol. 324, No. 5924, Apr. 10, 2009 (Apr. 10, 2009), pp. 261-265. |
| Zheng et al., "Synthesis and antitumor evaluation of a novel series of triaminotriazine derivatives," Bioorganic & Medicinal Chemistry 15 (2007), pp. 1815-1827. |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| USRE49582E1 (en) | Therapeutic compounds and compositions | |
| US8889667B2 (en) | Therapeutic compounds and compositions | |
| US9115086B2 (en) | Therapeutic compositions and related methods of use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FEPP | Fee payment procedure |
Free format text: ENTITY STATUS SET TO UNDISCOUNTED (ORIGINAL EVENT CODE: BIG.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| AS | Assignment |
Owner name: AGIOS PHARMACEUTICALS, INC., MASSACHUSETTS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:SALITURO, FRANCESCO G.;SAUNDERS, JEFFREY O.;YAN, SHUNQI;REEL/FRAME:062708/0945 Effective date: 20100831 Owner name: SCHRODINGER, INC., NEW YORK Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:YAN, SHUNQI;REEL/FRAME:062761/0319 Effective date: 20171209 Owner name: AGIOS PHARMACEUTICALS, INC., MASSACHUSETTS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:SCHRODINGER, LLC;REEL/FRAME:062710/0835 Effective date: 20171220 Owner name: SCHRODINGER, LLC, NEW YORK Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:SCHRODINGER, INC.;REEL/FRAME:062710/0255 Effective date: 20171220 |