CLAIM OF PRIORITY
-
This application claims priority to U.S. Ser. No. 61/160,253, filed Mar. 13, 2009; U.S. Ser. No. 61/160,664, filed Mar. 16, 2009; U.S. Ser. No. 61/173,518, filed Apr. 28, 2009; U.S. Ser. No. 61/180,609, filed May 22, 2009; U.S. Ser. No. 61/220,543, filed Jun. 25, 2009; U.S. Ser. No. 61/227,649, filed Jul. 22, 2009; U.S. Ser. No. 61/229,689, filed Jul. 29, 2009; U.S. Ser. No. 61/253,820, filed Oct. 21, 2009; and U.S. Ser. No. 61/266,929, filed Dec. 4, 2009, the contents of each of which are incorporated herein by reference.
FIELD OF THE INVENTION
-
The invention relates to methods and compositions for evaluating and treating cell proliferation-related disorders, e.g., proliferative disorders such as cancer.
BACKGROUND
-
Isocitrate dehydrogenase, also known as IDH, is an enzyme which participates in the citric acid cycle. It catalyzes the third step of the cycle: the oxidative decarboxylation of isocitrate, producing alpha-ketoglutarate (α-ketoglutarate or α-KG) and CO2 while converting NAD+ to NADH. This is a two-step process, which involves oxidation of isocitrate (a secondary alcohol) to oxalosuccinate (a ketone), followed by the decarboxylation of the carboxyl group beta to the ketone, forming alpha-ketoglutarate. Another isoform of the enzyme catalyzes the same reaction; however this reaction is unrelated to the citric acid cycle, is carried out in the cytosol as well as the mitochondrion and peroxisome, and uses NADP+ as a cofactor instead of NAD+.
SUMMARY OF THE INVENTION
-
Methods and compositions disclosed herein relate to the role played in disease by neoactive products produced by neoactive mutant enzymes, e.g., mutant metabolic pathway enzymes. The inventors have discovered, inter alia, a neoactivity associated with IDH mutants and that the product of the neoactivity can be significantly elevated in cancer cells. Disclosed herein are methods and compositions for treating, and methods of evaluating, subjects having or at risk for a disorder, e.g., a cell proliferation-related disorder characterized by a neoactivity in a metabolic pathway enzyme, e.g., IDH neoactivity. Such disorders include e.g., proliferative disorders such as cancer. The inventors have discovered and disclosed herein novel therapeutic agents for the treatment of disorders, e.g., cancers, characterized by, e.g., by a neoactivity, neoactive protein, neoactive mRNA, or neoactive mutations. In embodiments a therapeutic agent reduces levels of neoactivity or neoactive product or ameliorates an effect of a neoactive product. Methods described herein also allow the identification of a subject, or identification of a treatment for the subject, on the basis of neaoctivity genotype or phenotype. This evaluation can allow for optimal matching of subject with treatment, e.g., where the selection of subject, treatment, or both, is based on an analysis of neoactivity genotype or phenotype. E.g., methods describe herein can allow selection of a treatment regimen comprising administration of a novel compound, e.g., a novel compound disclosed herein, or a known compound, e.g., a known compound not previously recommended for a selected disorder. In embodiments the known compound reduces levels of neoactivity or neoactive product or ameliorates an effect of a neoactive product. Methods described herein can guide and provide a basis for selection and administration of a novel compound or a known compound, or combination of compounds, not previously recommended for subjects having a disorder characterized by a somatic neoactive mutation in a metabolic pathway enzyme. In embodiments the neoactive genotype or phenotype can act as a biomarker the presence of which indicates that a compound, either novel, or previously known, should be administered, to treat a disorder characterized by a somatic neoactive mutation in a metabolic pathway enzyme. Neoactive mutants of IDH1 having a neoactivity that results in the production of 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate and associated disorders are discussed in detail herein. They are exemplary, but not limiting, examples of embodiments of the invention.
-
While not wishing to be bound by theory it is believed that the balance between the production and elimination of neoactive product, e.g., 2HG, e.g., R-2HG, is important in disease. Neoactive mutants, to varying degrees for varying mutations, increase the level of neoactive product, while other processes, e.g., in the case of 2HG, e.g., R-2HG, enzymatic degradation of 2HG, e.g., by 2HG dehydrogenase, reduce the level of neoative product. An incorrect balance is associated with disease. In embodiments, the net result of a neoactive mutation at IDH1 or IDH2 result in increased levels, in affected cells, of neoactive product, 2HG, e.g., R-2HG,
-
Accordingly, in one aspect, the invention features, a method of treating a subject having a cell proliferation-related disorder, e.g., a disorder characterized by unwanted cell proliferation, e.g., cancer, or a precancerous disorder. The cell proliferation-related disorder is characterized by a somatic mutation in a metabolic pathway enzyme. The mutation is associated with a neoactivity that results in the production of a neoactivity product. The method comprises: administering to the subject a therapeutically effective amount of a therapeutic agent described herein, e.g., a therapeutic agent that decreases the level of neoactivity product encoded by a selected or mutant somatic allele, e.g., an inhibitor of a neoactivity of the metabolic pathway enzyme (the neoactive enzyme), a therapeutic agent that ameliorates an unwanted affect of the neoactivity product, or a nucleic acid based inhibitor, e.g., a dRNA which targets the neoactive enzyme mRNA, to thereby treat the subject.
-
In an embodiment the subject is a subject not having, or not diagnosed as having, 2-hydroxyglutaric aciduria.
-
In an embodiment the subject has a cell proliferation-related disorder, e.g., a cancer, characterized by the neoactivity of the metabolic pathway enzyme encoded by selected or mutant allele.
-
In an embodiment the subject has a cell proliferation-related disorder, e.g., a cancer, characterized by the product formed by the neoactivity of the metabolic pathway enzyme encoded by selected or mutant allele.
-
In one embodiment, the metabolic pathway is selected from a metabolic pathway leading to fatty acid biosynthesis, glycolysis, glutaminolysis, the pentose phosphate shunt, nucleotide biosynthetic pathways, or the fatty acid biosynthetic pathway.
-
In an embodiment the therapeutic agent is a therapeutic agent described herein.
-
In an embodiment the method comprises selecting a subject on the basis of having a cancer characterized by the selected or mutant allele, the neoactivity, or an elevated level of neaoctivity product.
-
In an embodiment the method comprises selecting a subject on the basis of having a cancer characterized by the product formed by the neoactivity of the protein encoded by selected or mutant allele, e.g., by the imaging and/or spectroscopic analysis, e.g., magnetic resonance-based analysis, e.g., MRI (magnetic resonance imaging) and/or MRS (magnetic resonance spectroscopy), to determine the presence, distribution or level of the product of the neoactivity, e.g., in the case of an IDH1 allele described herein, 2-hydroxyglutarate (sometimes referred to herein as 2HG), e.g., R-2-hydroxyglutarate (sometimes referred to herein as R-2HG).
-
In an embodiment the method comprises confirming or determining, e.g., by direct examination or evaluation of the subject, or sample e.g., tissue, product (e.g., feces, sweat, semen, exhalation, hair or nails), or bodily fluid (e.g., blood (e.g., blood plasma), urine, lymph, or cerebrospinal fluid or other sample sourced disclosed herein) therefrom, (e.g., by DNA sequencing, immuno analysis, or assay for enzymatic activity), or receiving such information about the subject, that the cancer is characterized by the selected or mutant allele.
-
In an embodiment the method comprises confirming or determining, e.g., by direct examination or evaluation of the subject, the level of neoactivity or the level of the product of the neoactivity, or receiving such information about the subject. In an embodiment the presence, distribution or level of the product of the neoactivity, e.g., in the case of an IDH1 allele described herein, 2HG, e.g., R-2HG, is determined non-invasively, e.g., by imaging methods, e.g., by magnetic resonance-based methods.
-
In an embodiment the method comprises administering a second anti-cancer agent or therapy to the subject, e.g., surgical removal or administration of a chemotherapeutic.
-
In another aspect, the invention features, a method of treating a subject having a cell proliferation-related disorder, e.g., a precancerous disorder, or cancer. In an embodiment the subject does not have, or has not been diagnosed as having, 2-hydroxyglutaric aciduria. The cell proliferation-related disorder is characterized by a somatic allele, e.g., a preselected allele, or mutant allele, of an IDH, e.g., IDH1 or IDH2, which encodes a mutant IDH, e.g., IDH1 or IDH2, enzyme having a neoactivity.
-
In embodiments the neoactivity is alpha hydroxy neoactivity. As used herein, alpha hydroxy neoactivity refers to the ability to convert an alpha ketone to an alpha hydroxy. In embodiments alpha hydroxy neoactivity proceeds with a reductive cofactor, e.g., NADPH or NADH. In embodiments the alpha hydroxyl neoactivity is 2HG neoactivity. 2HG neoactivity, as used herein, refers to the ability to convert alpha ketoglutarate to 2-hydroxyglutarate (sometimes referred to herein as 2HG), e.g., R-2-hydroxyglutarate (sometimes referred to herein as R-2HG). In embodiments 2HG neoactivity proceeds with a reductive cofactor, e.g., NADPH or NADH. In an embodiment a neoactive enzyme, e.g., an alpha hydroxyl, e.g., a 2HG, neoactive enzyme, can act on more than one substrate, e.g., more than one alpha hydroxy substrate.
-
The method comprises administering to the subject an effective amount of a therapeutic agent of type described herein to thereby treat the subject.
-
In an embodiment the therapeutic agent: results in lowering the level of a neoactivity product, e.g., an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the method comprises administering a therapeutic agent that lowers neoactivity, e.g., 2HG neoactivity. In an embodiment the method comprises administering an inhibitor of a mutant IDH protein, e.g., a mutant IDH1 or mutant IDH2 protein, having a neoactivity, e.g., alpha hydroxy neoactivity, e.g., 2HG neoactivity.
-
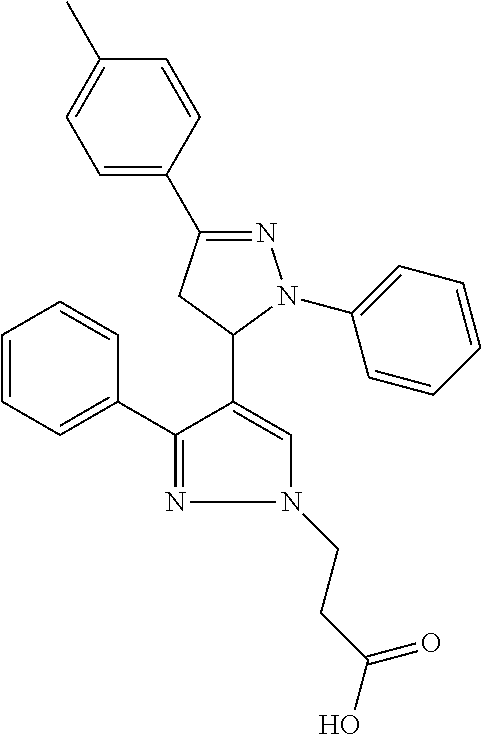
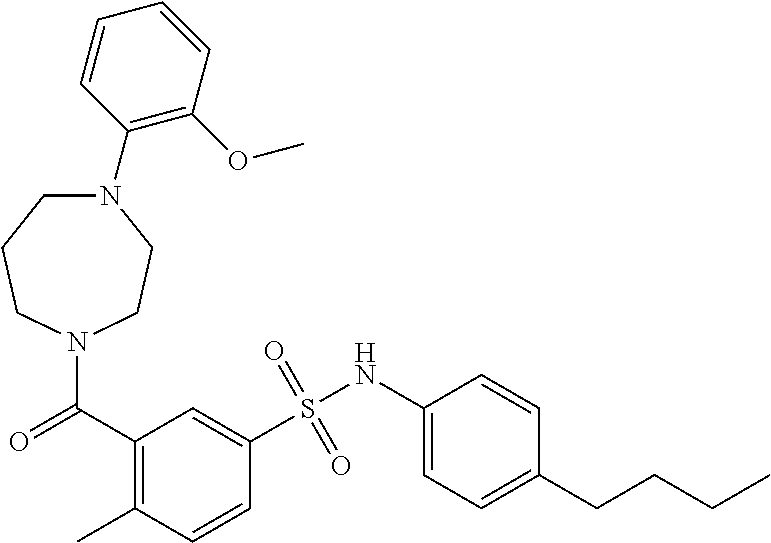
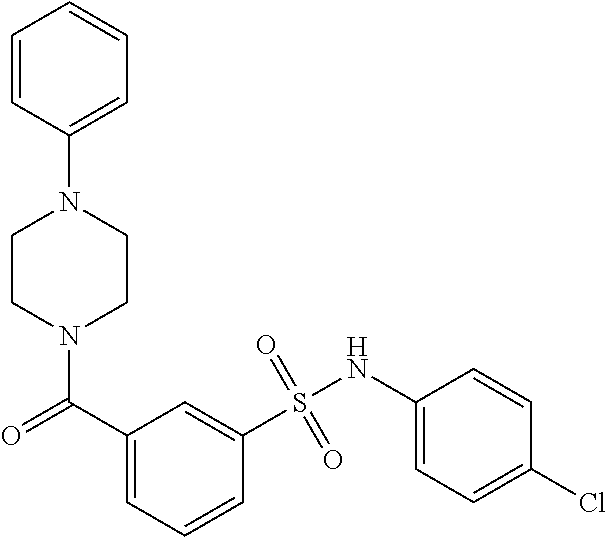
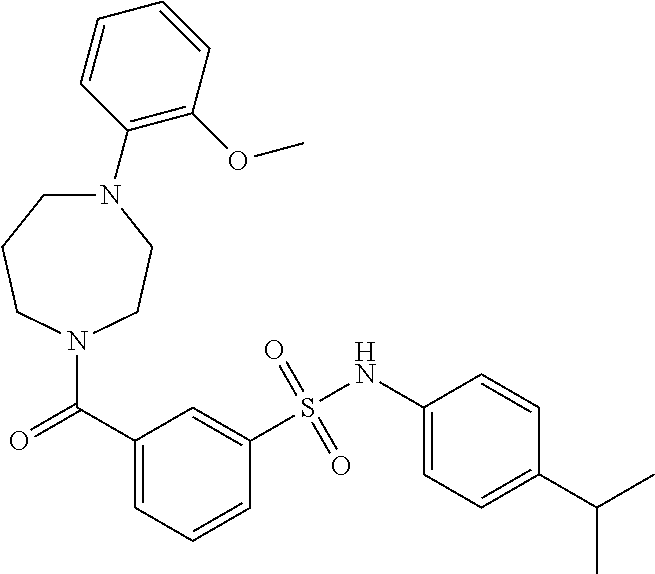
In an embodiment the therapeutic agent comprises a compound from Table 24a or Table 24b or a compound having the structure of Formula (X) or (Formula (XI) described herein.
-
In an embodiment the therapeutic agent comprises nucleic acid-based therapeutic agent, e.g., a dsRNA, e.g., a dsRNA described herein.
-
In an embodiment the therapeutic agent is an inhibitor, e.g., a polypeptide, peptide, or small molecule (e.g., a molecule of less than 1,000 daltons), or aptomer, that binds to an IDH1 mutant or wildtype subunit and inhibits neoactivity, e.g., by inhibiting formation of a dimer, e.g., a homodimer of mutant IDH1 subunits or a heterodimer of a mutant and a wildype subunit. In an embodiment the inhibitor is a polypeptide. In an embodiment the polypeptide acts as a dominant negative with respect to the neoactivity of the mutant enzyme. The polypeptide can correspond to full length IDH1 or a fragment thereof. The polypeptide need not be identical with the corresponding residues of wildtype IDH1, but in embodiments has at least 60, 70, 80, 90 or 95% homology with wildtype IDH1.
-
In an embodiment the therapeutic agent decreases the affinity of an IDH, e.g., IDH1 or IDH2 neoactive mutant protein for NADH, NADPH or a divalent metal ion, e.g., Mg2+ or Mn2+, or decreases the levels or availability of NADH, NADPH or divalent metal ion, e.g., Mg2+ or Mn2+, e.g., by competing for binding to the mutant enzyme. In an embodiment the enzyme is inhibited by replacing Mg2+ or Mn2+ with Ca2+.
-
In an embodiment the therapeutic agent is an inhibitor that reduces the level a neoactivity of an IDH, e.g., IDH1 or IDH2, e.g., 2HG neoactivity.
-
In an embodiment the therapeutic agent is an inhibitor that reduces the level of the product of a mutant having a neoactivity of an IDH, e.g., IDH1 or IDH2 mutant, e.g., it reduces the level of 2HG, e.g., R-2HG.
-
In an embodiment the therapeutic agent is an inhibitor that:
-
inhibits, e.g., specifically, a neoactivity of an IDH, e.g., IDH1 or IDH2, e.g., a neoactivity described herein, e.g., 2HG neoactivity; or
-
inhibits both the wildtype activity and a neoactivity of an IDH, e.g., IDH1 or IDH2, e.g., a neoactivity described herein, e.g, 2HG neoactivity.
-
In an embodiment the therapeutic agent is an inhibitor that is selected on the basis that it:
-
inhibits, e.g., specifically, a neoactivity of an IDH, e.g., IDH1 or IDH2, e.g., a neoactivity described herein e.g., 2HG neoactivity; or
-
inhibits both the wildtype activity and a neoactivity of an IDH1, e.g., IDH1 or IDH2, e.g., a neoactivity described herein, e.g., 2HG neoactivity.
-
In an embodiment the therapeutic agent is an inhibitor that reduces the amount of a mutant IDH, e.g., IDH1 or IDH2, protein or mRNA.
-
In an embodiment the therapeutic agent is an inhibitor that interacts directly with, e.g., it binds to, the mutant IDH, e.g., IDH1 or IDH2 mRNA.
-
In an embodiment the therapeutic agent is an inhibitor that interacts directly with, e.g., it binds to, the mutant IDH, e.g., IDH1 or IDH2, protein.
-
In an embodiment the therapeutic agent is an inhibitor that reduces the amount of neoactive enzyme activity, e.g., by interacting with, e.g., binding to, mutant IDH, e.g., IDH1 or IDH2, protein. In an embodiment the inhibitor is other than an antibody.
-
In an embodiment the therapeutic agent is an inhibitor that is a small molecule and interacts with, e.g., binds, the mutant RNA, e.g., mutant IDH1 or IDH2 mRNA (e.g., mutant IDH1 mRNA).
-
In an embodiment the therapeutic agent is an inhibitor that interacts directly with, e.g., binds, either the mutant IDH, e.g., IDH1 or IDH2, protein or interacts directly with, e.g., binds, the mutant IDH mRNA, e.g., IDH1 or IDH2 mRNA.
-
In an embodiment the IDH is IDH1 and the neoactivity is alpha hydroxy neoactivity, e.g., 2HG neoactivity. Mutations in IDH1 associated with 2HG neoactivity include mutations at residue 132, e.g., R132H, R132C, R132S, R132G, R132L, or R132V (e.g., R132H or R132C).
-
In an embodiment the IDH is IDH2 and the neoactivity of the IDH2 mutant is alpha hydroxy neoactivity, e.g., 2HG neoactivity. Mutations in IDH2 associated with 2HG neoactivity include mutations at residue 172, e.g., R172K, R172M, R172S, R172G, or R172W.
-
Treatment methods described herein can comprise evaluating a neoactivity genotype or phenotype. Methods of obtaining and analyzing samples, and the in vivo analysis in subjects, described elsewhere herein, e.g., in the section entitled, “Methods of evaluating samples and/or subjects,” can be combined with this method.
-
In an embodiment, prior to or after treatment, the method includes evaluating the growth, size, weight, invasiveness, stage or other phenotype of the cell proliferation-related disorder.
-
In an embodiment, prior to or after treatment, the method includes evaluating the IDH, e.g., IDH1 or IDH2, alpha hydroxyl neoactivity genotype, e.g., 2HG, genotype, or alpha hydroxy neoactivity phenotype, e.g., 2HG, e.g., R-2HG, phenotype. Evaluating the alpha hydroxyl, e.g., 2HG, genotype can comprise determining if an IDH1 or IDH2 mutation having alpha hydroxy neoactivity, e.g., 2HG neoactivity, is present, e.g., a mutation disclosed herein having alpha hydroxy neoactivity, e.g., 2HG neoactivity. Alpha hydroxy neoactivity phenotype, e.g., 2HG, e.g., R-2HG, phenotype, as used herein, refers to the level of alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, level of alpha hydroxy neoactivity, e.g., 2HG neoactivity, or level of mutant enzyme having alpha hydroxy neoactivity, e.g., 2HG neoactivity (or corresponding mRNA). The evaluation can be by a method described herein.
-
In an embodiment the subject can be evaluated, before or after treatment, to determine if the cell proliferation-related disorder is characterized by an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment a cancer, e.g., a glioma or brain tumor in a subject, can be analyzed, e.g., by imaging and/or spectroscopic analysis, e.g., magnetic resonance-based analysis, e.g., MRI and/or MRS, e.g., before or after treatment, to determine if it is characterized by presence of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the method comprises evaluating, e.g., by direct examination or evaluation of the subject, or a sample from the subject, or receiving such information about the subject, the IDH, e.g., IDH1 or IDH2, genotype, or an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG phenotype of, the subject, e.g., of a cell, e.g., a cancer cell, characterized by the cell proliferation-related disorder. (As described in more detail elsewhere herein the evaluation can be, e.g., by DNA sequencing, immuno analysis, evaluation of the presence, distribution or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, e.g., from spectroscopic analysis, e.g., magnetic resonance-based analysis, e.g., MRI and/or MRS measurement, sample analysis such as serum or spinal cord fluid analysis, or by analysis of surgical material, e.g., by mass-spectroscopy). In embodiments this information is used to determine or confirm that a proliferation-related disorder, e.g., a cancer, is characterized by an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG. In embodiments this information is used to determine or confirm that a cell proliferation-related disorder, e.g., a cancer, is characterized by an IDH, e.g., IDH1 or IDH2, allele described herein, e.g., an IDH1 allele having a mutation, e.g., a His, Ser, Cys, Gly, Val, Pro or Leu (e.g., His, Ser, Cys, Gly, Val, or Leu at residue 132, more specifically, His or Cys, or an IDH2 allele having a mutation at residue 172, e.g., a K, M, S, G, or W.
-
In an embodiment, before and/or after treatment has begun, the subject is evaluated or monitored by a method described herein, e.g., the analysis of the presence, distribution, or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, e.g., to select, diagnose or prognose the subject, to select an inhibitor, or to evaluate response to the treatment or progression of disease.
-
In an embodiment the cell proliferation-related disorder is a tumor of the CNS, e.g., a glioma, a leukemia, e.g., AML or ALL, e.g., B-ALL or T-ALL, prostate cancer, fibrosarcoma, paraganglioma, or myelodysplasia or myelodysplastic syndrome (e.g., B-ALL or T-ALL, prostate cancer, or myelodysplasia or myelodysplastic syndrome) and the evaluation is: evaluation of the presence, distribution, or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG; or evaluation of the presence, distribution, or level of a neoactivity, e.g., an alpha hydroxy neoactivity, e.g., 2HG neoactivity, of an IDH1 or IDH2, mutant protein.
-
In an embodiment the disorder is other than a solid tumor. In an embodiment the disorder is a tumor that, at the time of diagnosis or treatment, does not have a necrotic portion. In an embodiment the disorder is a tumor in which at least 30, 40, 50, 60, 70, 80 or 90% of the tumor cells carry an IHD, e.g., IDH1 or IDH2, mutation having 2HG neoactivity, at the time of diagnosis or treatment.
-
In an embodiment the cell proliferation-related disorder is a cancer, e.g., a cancer described herein, characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the tumor is characterized by increased levels of an alpha hydroxy neoactivity product, 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type.
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by unwanted (i.e., increased) levels of an alpha hydroxy neoactivity, product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is a tumor of the CNS, e.g., a glioma, e.g., wherein the tumor is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. Gliomas include astrocytic tumors, oligodendroglial tumors, oligoastrocytic tumors, anaplastic astrocytomas, and glioblastomas. In an embodiment the tumor is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type. E.g., in an embodiment, the IDH1 allele encodes an IDH1 having other than an Arg at residue 132. E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu (e.g., His, Ser, Cys, Gly, Val, or Leu), or any residue described in Yan et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21). In an embodiment the allele encodes an IDH1 having His at residue 132. In an embodiment the allele encodes an IDH1 having Ser at residue 132.
-
In an embodiment the IDH1 allele has an A (or any other nucleotide other than C) at nucleotide position 394, or an A (or any other nucleotide other than G) at nucleotide position 395. In an embodiment the allele is a C394A, a C394G, a C394T, a G395C, a G395T or a G395A mutation; specifically a C394A or a G395A mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having a glioma, wherein the cancer is characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having His, Ser, Cys, Gly, Val, Pro or Leu at residue 132 (SEQ ID NO:8), more specifically His, Ser, Cys, Gly, Val, or Leu; or His or Cys.
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by an IDH1 allele described herein, e.g., an IDH1 allele having His, Ser, Cys, Gly, Val, Pro or Leu at residue 132 (SEQ ID NO:8), more specifically His, Ser, Cys, Gly, Val, or Leu; or His or Cys.
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity, product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the method comprises selecting a subject having a fibrosarcoma or paraganglioma wherein the cancer is characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having a fibrosarcoma or paraganglioma, on the basis of the cancer being characterized by an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having a fibrosarcoma or paraganglioma, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity, product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is localized or metastatic prostate cancer, e.g., prostate adenocarcinoma, e.g., wherein the cancer is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the cancer is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type.
-
E.g., in an embodiment, the IDH1 allele encodes an IDH1 having other than an Arg at residue 132. E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Kang et al, 2009, Int. J. Cancer, 125: 353-355 at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21) (e.g., His, Ser, Cys, Gly, Val, or Leu). In an embodiment the allele encodes an IDH1 having His or Cys at residue 132.
-
In an embodiment the IDH1 allele has a T (or any other nucleotide other than C) at nucleotide position 394, or an A (or any other nucleotide other than G) at nucleotide position 395. In an embodiment the allele is a C394T or a G395A mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having prostate cancer, e.g., prostate adenocarcinoma, wherein the cancer is characterized by an IDH1 allele described herein, e.g., an IDH1 allele having His or Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having prostate cancer, e.g., prostate adenocarcinoma, on the basis of the cancer being characterized by an IDH1 allele described herein, e.g., an IDH1 allele having His or Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having prostate cancer, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is a hematological cancer, e.g., a leukemia, e.g., AML, or ALL, wherein the hematological cancer is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the cancer is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type.
-
In an embodiment the cell proliferation-related disorder is acute lymphoblastic leukemia (e.g., an adult or pediatric form), e.g., wherein the acute lymphoblastic leukemia (sometimes referred to herein as ALL) is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. The ALL can be, e.g., B-ALL or T-ALL. In an embodiment the cancer is characterized by increased levels of 2 an alpha hydroxy neoactivity product, e.g., HG, e.g., R-2HG, as compared to non-diseased cells of the same type. E.g., in an embodiment, the IDH1 allele is an IDH1 having other than an Arg at residue 132 (SEQ ID NO:8). E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Kang et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21), more specifically His, Ser, Cys, Gly, Val, or Leu. In an embodiment the allele encodes an IDH1 having Cys at residue 132.
-
In an embodiment the IDH1 allele has a T (or any other nucleotide other than C) at nucleotide position 394. In an embodiment the allele is a C394T mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, characterized by an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 according to the sequence of SEQ ID NO:8.
-
In an embodiment the method comprises selecting a subject ALL, e.g., B-ALL or T-ALL, on the basis of cancer being characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is acute myelogenous leukemia (e.g., an adult or pediatric form), e.g., wherein the acute myelogenous leukemia (sometimes referred to herein as AML) is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the cancer is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type. E.g., in an embodiment, the IDH1 allele is an IDH1 having other than an Arg at residue 132 (SEQ ID NO:8). E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Kang et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21). In an embodiment the allele encodes an IDH1 having Cys, His or Gly at residue 132, more specifically, Cys at residue 132.
-
In an embodiment the IDH1 allele has a T (or any other nucleotide other than C) at nucleotide position 394. In an embodiment the allele is a C394T mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having acute myelogenous lymphoplastic leukemia (AML) characterized by an IDH1 allele described herein, e.g., an IDH1 allele having Cys, His, or Gly at residue 132 according to the sequence of SEQ ID NO:8, more specifically, Cys at residue 132.
-
In an embodiment the method comprises selecting a subject having acute myelogenous lymphoplastic leukemia (AML) on the basis of cancer being characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having Cys, His, or Gly at residue 132 (SEQ ID NO:8), more specifically, Cys at residue 132.
-
In an embodiment the method comprises selecting a subject having acute myelogenous lymphoplastic leukemia (AML), on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the method further comprises evaluating the subject for the presence of a mutation in the NRAS or NPMc gene.
-
In an embodiment the cell proliferation-related disorder is myelodysplasia or myelodysplastic syndrome, e.g., wherein the myelodysplasia or myelodysplastic syndrome is characterized by having an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the disorder is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type. E.g., in an embodiment, the IDH1 allele is an IDH1 having other than an Arg at residue 132 (SEQ ID NO:8). E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Kang et al., according to the sequence of SEQ ID NO:8 (see also FIG. 21), more specifically His, Ser, Cys, Gly, Val, or Leu. In an embodiment the allele encodes an IDH1 having Cys at residue 132.
-
In an embodiment the IDH1 allele has a T (or any other nucleotide other than C) at nucleotide position 394. In an embodiment the allele is a C394T mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome characterized by an IDH1 allele described herein, e.g., an IDH1 allele having Cys, His, or Gly at residue 132 according to the sequence of SEQ ID NO:8, more specifically, Cys at residue 132.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome on the basis of cancer being characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having Cys, His, or Gly at residue 132 (SEQ ID NO:8), more specifically, Cys at residue 132.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is a glioma, characterized by a mutation, or preselected allele, of IDH2 associated with an alpha hydroxy neoactivity, e.g., 2HG neoactivity. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), more specifically Lys, Gly, Met, Trp, or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment the method comprises selecting a subject having a glioma, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys, Gly, Met, Trp, Thr, or Ser at residue 172 (SEQ ID NO:10), more specifically Lys, Gly, Met, Trp, or Ser; or Lys or Met.
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys, Gly, Met, Trp, Thr, or Ser at residue 172 (SEQ ID NO:10), more specifically Lys, Gly, Met, Trp, or Ser; or Lys or Met.
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is a prostate cancer, e.g., prostate adenocarcinoma, characterized by a mutation, or preselected allele, of IDH2 associated with an alpha hydroxy neoactivity, e.g., 2HG neoactivity. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), more specifically Lys, Gly, Met, Trp, or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment the method comprises selecting a subject having a prostate cancer, e.g., prostate adenocarcinoma, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having a prostate cancer, e.g., prostate adenocarcinoma, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having a prostate cancer, e.g., prostate adenocarcinoma, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is ALL, e.g., B-ALL or T-ALL, characterized by a mutation, or preselected allele, of IDH2 associated with an alpha hydroxy neoactivity, e.g., 2HG neoactivity. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22). In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is AML, characterized by a mutation, or preselected allele, of IDH2 associated with an alpha hydroxy neoactivity, e.g., 2HG neoactivity. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), more specifically Lys, Gly, Met, or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172. In an embodiment the allele encodes an IDH2 having Gly at residue 172.
-
In an embodiment the method comprises selecting a subject having AML, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys, Gly or Met at residue 172 (SEQ ID NO:10), more specifically Lys or Met.
-
In an embodiment the method comprises selecting a subject having AML, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys, Gly, or Met at residue 172 (SEQ ID NO:10), more specifically Lys or Met.
-
In an embodiment the method comprises selecting a subject having AML, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is myelodysplasia or myelodysplastic syndrome, characterized by a mutation, or preselected allele, of IDH2. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), more specifically Lys, Gly, Met, Trp or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172. In an embodiment the allele encodes an IDH2 having Gly at residue 172.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys, Gly, or Met at residue 172 (SEQ ID NO:10), in specific embodiments, Lys or Met.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys, Gly, or Met at residue 172 (SEQ ID NO:10), in specific embodiments, Lys or Met.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment a product of the neoactivity is 2HG (e.g., R-2HG) which acts as a metabolite. In another embodiment a product of the neoactivity is 2HG (e.g., R-2HG) which acts as a toxin, e.g., a carcinogen.
-
In some embodiments, the methods described herein can result in reduced side effects relative to other known methods of treating cancer.
-
Therapeutic agents and methods of subject evaluation described herein can be combined with other therapeutic mocalities, e.g., with art-known treatments.
-
In an embodiment the method comprises providing a second treatment, to the subject, e.g., surgical removal, irradiation or administration of a chemotherapeutitc agent, e.g., an administration of an alkylating agent. Administration (or the establishment of therapeutic levels) of the second treatment can: begin prior to the beginning or treatment with (or prior to the establishment of therapeutic levels of) the inhibitor; begin after the beginning or treatment with (or after the establishment of therapeutic levels of) the inhibitor, or can be administered concurrently with the inhibitor, e.g., to achieve therapeutic levels of both concurrently.
-
In an embodiment the cell proliferation-related disorder is a CNS tumor, e.g., a glioma, and the second therapy comprises administration of one or more of: radiation; an alkylating agent, e.g., temozolomide, e.g., Temoader®, or BCNU; or an inhibitor of HER1/EGFR tyrosine kinase, e.g., erlotinib, e.g., Tarceva®.
-
The second therapy, e.g., in the case of glioma, can comprise implantation of BCNU or carmustine in the brain, e.g., implantation of a Gliadel® wafer.
-
The second therapy, e.g., in the case of glioma, can comprise administration of imatinib, e.g., Gleevec®.
-
In an embodiment the cell proliferation-related disorder is prostate cancer and the second therapy comprises one or more of: androgen ablation; administration of a microtubule stabilizer, e.g., docetaxol, e.g., Taxotere®; or administration of a topoisomerase II inhibitor, e.g., mitoxantrone.
-
In an embodiment the cell proliferation-related disorder is ALL, e.g., B-ALL or T-ALL, and the second therapy comprises one or more of:
-
induction phase treatment comprising the administration of one or more of: a steroid; an inhibitor of microtubule assembly, e.g., vincristine; an agent that reduces the availability of asparagine, e.g., asparaginase; an anthracycline; or an antimetabolite, e.g., methotrexate, e.g., intrathecal methotrexate, or 6-mercaptopurine;
-
consolidation phase treatment comprising the administration of one or more of: a drug listed above for the induction phase; an antimetabolite, e.g., a guanine analog, e.g., 6-thioguanine; an alkylating agent, e.g., cyclophosphamide; an anti-metabolite, e.g., AraC or cytarabine; or an inhibitor of topoisomerase I, e.g., etoposide; or
-
maintenance phase treatment comprising the administration of one or more of the drugs listed above for induction or consolidation phase treatment.
-
In an embodiment the cell proliferation-related disorder is AML and the second therapy comprises administration of one or more of: an inhibitor of topoisomerase II, e.g., daunorubicin, idarubicin, topotecan or mitoxantrone; an inhibitor of topoisomerase I, e.g., etoposide; or an anti-metabolite, e.g., AraC or cytarabine.
-
In another aspect, the invention features, a method of evaluating, e.g. diagnosing, a subject, e.g., a subject not having, or not diagnosed as having, 2-hydroxyglutaric aciduria. The method comprises analyzing a parameter related to the neoactivity genotype or phenotype of the subject, e.g., analyzing one or more of:
-
a) the presence, distribution, or level of a neoactive product, e.g., the product of an alpha hydroxy neoactivity, e.g., 2HG, e.g., R-2HG, e.g., an increased level of product, 2HG, e.g., R-2HG (as used herein, an increased level of a product of an alpha hydroxy neoactivity, e.g., 2HG, e.g., R-2HG, or similar term, e.g., an increased level of neoactive product or neoactivity product, means increased as compared with a reference, e.g., the level seen in an otherwise similar cell lacking the IDH mutation, e.g., IDH1 or IDH2 mutation, or in a tissue or product from a subject not having the mutation (the terms increased and elevated as referred to the level of a product of alpha hydroxyl neoactivity as used herein, are used interchangably);
-
b) the presence, distribution, or level of a neoactivity, e.g., alpha hydroxy neoactivity, e.g., 2HG neoactivity, of an IDH1 or IDH2, mutant protein;
-
c) the presence, distribution, or level of a neoactive mutant protein, e.g., an IDH, e.g., an IDH1 or IDH2, mutant protein which has a neoactivity, e.g., alpha hydroxy neoactivity, e.g., 2HG neoactivity, or a corresponding RNA; or
-
d) the presence of a selected somatic allele or mutation conferring neoactivity, e.g., an IDH, e.g., IDH1 or IDH2, which encodes a protein with a neoactivity, e.g., alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., an allele disclosed herein, in cells characterized by a cell proliferation-related disorder from the subject, thereby evaluating the subject.
-
In an embodiment analyzing comprises performing a procedure, e.g., a test, to provide data or information on one or more of a-d, e.g., performing a method which results in a physical change in a sample, in the subject, or in a device or reagent used in the analysis, or which results in the formation of an image representative of the data. Methods of obtaining and analyzing samples, and the in vivo analysis in subjects, described elsewhere herein, e.g., in the section entitled, “Methods of evaluating samples and/or subjects.,” can be combined with this method. In another embodiment analyzing comprises receiving data or information from such test from another party. In an embodiment the analyzing comprises receiving data or information from such test from another party and, the method comprises, responsive to that data or information, administering a treatment to the subject.
-
As described herein, the evaluation can be used in a number of applications, e.g., for diagnosis, prognosis, staging, determination of treatment efficacy, patent selection, or drug selection.
-
Thus, in an embodiment method further comprises, e.g., responsive to the analysis of one or more of a-d:
-
diagnosing the subject, e.g., diagnosing the subject as having a cell proliferation-related disorder, e.g., a disorder characterized by unwanted cell proliferation, e.g., cancer, or a precancerous disorder;
-
staging the subject, e.g., determining the stage of a cell proliferation-related disorder, e.g., a disorder characterized by unwanted cell proliferation, e.g., cancer, or a precancerous disorder;
-
providing a prognosis for the subject, e.g., providing a prognosis for a cell proliferation-related disorder, e.g., a disorder characterized by unwanted cell proliferation, e.g., cancer, or a precancerous disorder;
-
determining the efficacy of a treatment, e.g., the efficacy of a chemotherapeutic agent, irradiation or surgery;
-
determining the efficacy of a treatment with a therapeutic agent, e.g., an inhibitor, described herein;
-
selecting the subject for a treatment for a cell proliferation-related disorder, e.g., a disorder characterized by unwanted cell proliferation, e.g., cancer, or a precancerous disorder. The selection can be based on the need for a reduction in neoactivity or on the need for amelioration of a condition associated with or resulting from neoactivity. For example, if it is determined that the subject has a cell proliferation-related disorder, e.g., e.g., cancer, or a precancerous disorder characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, or by a mutant IDH1 or IDH2, having alpha hydroxyl neoactivity, e.g., 2HG, neaoctivity, selecting the subject for treatment with a therapeutic agent described herein, e.g., an inhibitor (e.g., a small molecule or a nucleic acid-based inhibitor) of the neoactivity of that mutant (e.g., conversion of alpha-ketoglutarate to 2HG, e.g., R-2HG);
-
correlating the analysis with an outcome or a prognosis;
-
providing a value for an analysis on which the evaluation is based, e.g., the value for a parameter correlated to the presence, distribution, or level of an alpha hydroxyl neoactivity product, e.g., 2HG, e.g., R-2HG;
-
providing a recommendation for treatment of the subject; or
-
memorializing a result of, or output from, the method, e.g., a measurement made in the course of performing the method, and optionally transmitting the memorialization to a party, e.g., the subject, a healthcare provider, or an entity that pays for the subject's treatment, e.g., a government, insurance company, or other third party payer.
-
As described herein, the evaluation can provide information on which a number of decisions or treatments can be based.
-
Thus, in an embodiment the result of the evaluation, e.g., an increased level of an alpha hydroxyl neoactivity product, e.g., 2HG, e.g., R-2HG, the presence of an IDH, e.g., IDH1 or IDH2, neoactivity, e.g., alpha hydroxyl neoactivity, e.g., 2HG neoactivity, the presence of an IDH, e.g., IDH1 or IDH2, mutant protein (or corresponding RNA) which has alpha hydroxyl neoactivity, e.g., 2HG neoactivity, the presence of a mutant allele of IDH, e.g., IDH1 or IDH2, having alpha hydroxyl neoactivity, 2HG neoactivity, e.g., an allele disclosed herein, is indicative of:
-
a cell proliferation-related disorder, e.g., cancer, e.g., it is indicative of a primary or metastatic lesion;
-
the stage of a cell proliferation-related disorder;
-
a prognosis or outcome for a cell proliferation-related disorder, e.g., it is indicative of a less aggressive form of the disorder, e.g., cancer. E.g., in the case of glioma, presence of an alpha hydroxyl neoactivity product, e.g., 2HG, e.g., R-2HG, can indicate a less aggressive form of the cancer;
-
the efficacy of a treatment, e.g., the efficacy of a chemotherapeutic agent, irradiation or surgery;
-
the need of a therapy disclosed herein, e.g., inhibition a neoactivity of an IDH, e.g., IDH1 or IDH2, neoactive mutant described herein. In an embodiment relatively higher levels (or the presence of the mutant) is correlated with need of inhibition a neoactivity of an IDH, e.g., IDH1 or IDH2, mutant described herein; or
-
responsiveness to a treatment. The result can be used as a noninvasive biomarker for clinical response. E.g., elevated levels can be predictive on better outcome in glioma patients (e.g., longer life expectancy).
-
As described herein, the evaluation can provide for the selection of a subject.
-
Thus, in an embodiment the method comprises, e.g., responsive to the analysis of one or more of a-d, selecting a subject, e.g., for a treatment. The subject can be selected on a basis described herein, e.g., on the basis of:
-
said subject being at risk for, or having, higher than normal levels of an alpha hydroxy neoactivity product, e.g., 2-hydroxyglurarate (e.g., R-2HG) in cell having a cell proliferation-related disorder, e.g., a leukemia such as AML or ALL, e.g., B-ALL or T-ALL, or a tumor lesion, e.g., a glioma or a prostate tumor;
-
said subject having a proliferation-related disorder characterized by a selected IDH, e.g., IDH1 or IDH2 allele, e.g., an IDH1 or IDH2 mutation, having alpha hydroxyl neoactivity, e.g., 2HG neoactivity;
-
said subject having a selected IDH allele, e.g., a selected IDH1 or IDH2 allele; having alpha hydroxyl neoactivity, e.g., 2HG neoactivity;
-
said subject having a proliferation-related disorder;
-
said subject being in need of, or being able to benefit from, a therapeutic agent of a type described herein;
-
said subject being in need of, or being able to benefit from, a compound that inhibits alpha hydroxyl neoactivity, e.g., 2HG neoactivity;
-
said subject being in need of, or being able to benefit from, a compound that lowers the level of an alpha hydroxyl neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment evaluation comprises selecting the subject, e.g., for treatment with an anti-neoplastic agent, on the establishment of, or determination that, the subject has increased alpha hydroxyl neoactivity product, e.g., 2HG, e.g., R-2HG, or increased alpha hydroxyl neoactivity, e.g., 2HG neoactivity, or that the subject is in need of inhibition of a neoactivity of an IDH, e.g., IDH1 or IDH2, mutant described herein.
-
As described herein, the evaluations provided for by methods described herein allow the selection of optimal treatment regimens.
-
Thus, in an embodiment the method comprises, e.g., responsive to the analysis of one or more of a-d, selecting a treatment for the subject, e.g., selecting a treatment on a basis disclosed herein. The treatment can be the administration of a therapeutic agent disclosed herein. The treatment can be selected on the basis that:
-
it us useful in treating a disorder characterized by one or more of alpha hydroxyl neoactivity, e.g., 2HG neoactivity, an IDH1 or IDH2, mutant protein having alpha hydroxyl neoactivity, e.g., 2HG neoactivity (or a corresponding RNA);
-
it is useful in treating a disorder characterized by a selected somatic allele or mutation of an IDH, e.g., IDH1 or IDH2, which encodes a protein with alpha hydroxyl neoactivity, e.g., 2HG neoactivity, e.g., an allele disclosed herein, in cells characterized by a cell proliferation-related disorder from the subject;
-
it reduces the level of an alpha hydroxyl neoactivity product, e.g., 2HG, e.g., R-2HG;
-
it reduces the level of alpha hydroxyl neoactivity, e.g., 2HG neoactivity.
-
In an embodiment evaluation comprises selecting the subject, e.g., for treatment.
-
In embodiments the treatment is the administration of a therapeutic agent described herein.
-
The methods can also include treating a subject, e.g, with a treatment selected in response to, or on the basis of, an evaluation made in the method.
-
Thus, in an embodiment the method comprises, e.g., responsive to the analysis of one or more of a-d, administering a treatment to the subject, e.g., the administration of a therapeutic agent of a type described herein.
-
In an embodiment the therapeutic agent comprises a compound from Table 24a or Table 24b or a compound having the structure of Formula (X) or (XI) described below.
-
In an embodiment the therapeutic agent comprises nucleic acid, e.g., dsRNA, e.g., a dsRNA described herein.
-
In an embodiment the therapeutic agent is an inhibitor, e.g., a polypeptide, peptide, or small molecule (e.g., a molecule of less than 1,000 daltons), or aptomer, that binds to an IDH1 or IDH2 mutant (e.g., an aptomer that binds to an IDH1 mutant) or wildtype subunit and inhibits neoactivity, e.g., by inhibiting formation of a dimer, e.g., a homodimer of mutant IDH1 or IDH2 subunits (e.g., a homodimer of mutant IDH1 subunits) or a heterodimer of a mutant and a wildype subunit. In an embodiment the inhibitor is a polypeptide. In an embodiment the polypeptide acts as a dominant negative with respect to the neoactivity of the mutant enzyme. The polypeptide can correspond to full length IDH1 or IDH2 or a fragment thereof (e.g., the polypeptide corresponds to full length IDH1 or a fragment thereof). The polypeptide need not be identical with the corresponding residues of wildtype IDH1 or IDH2 (e.g., wildtype IDH1), but in embodiments has at least 60, 70, 80, 90 or 95% homology with wildtype IDH1 or IDH2 (e.g., wildtype IDH1).
-
In an embodiment the therapeutic agent decreases the affinity of an IDH, e.g., IDH1 or IDH2 neoactive mutant protein for NADH, NADPH or a divalent metal ion, e.g., Mg2+ or Mn2+, or decreases the levels or availability of NADH, NADPH or divalent metal ion, e.g., Mg2+ or Mn2+, e.g., by competing for binding to the mutant enzyme. In an embodiment the enzyme is inhibited by replacing Mg2+ or Mn2+ with Ca2+.
-
In an embodiment the therapeutic agent is an inhibitor that reduces the level a neoactivity of an IDH, e.g., IDH1 or IDH2, e.g., 2HG neoactivity.
-
In an embodiment the therapeutic agent is an inhibitor that reduces the level of the product of a mutant having a neoactivity of an IDH, e.g., IDH1 or IDH2 mutant, e.g., it reduces the level of 2HG, e.g., R-2HG.
-
In an embodiment the therapeutic agent is an inhibitor that:
-
inhibits, e.g., specifically, a neoactivity of an IDH, e.g., IDH1 or IDH2, e.g., a neoactivity described herein, e.g., 2HG neoactivity; or
-
inhibits both the wildtype activity and a neoactivity of an IDH, e.g., IDH1 or IDH2, e.g., a neoactivity described herein, e.g, 2HG neoactivity.
-
In an embodiment the therapeutic agent is an inhibitor that is selected on the basis that it:
-
inhibits, e.g., specifically, a neoactivity of an IDH, e.g., IDH1 or IDH2, e.g., a neoactivity described herein e.g., 2HG neoactivity; or
-
inhibits both the wildtype activity and a neoactivity of an IDH1, e.g., IDH1 or IDH2, e.g., a neoactivity described herein, e.g., 2HG neoactivity.
-
In an embodiment the therapeutic agent is an inhibitor that reduces the amount of a mutant IDH, e.g., IDH1 or IDH2, protein or mRNA.
-
In an embodiment the therapeutic agent is an inhibitor that interacts directly with, e.g., it binds to, the mutant IDH, e.g., IDH1 or IDH2 mRNA.
-
In an embodiment the therapeutic agent is an inhibitor that interacts directly with, e.g., it binds to, the mutant IDH, e.g., IDH1 or IDH2, protein.
-
In an embodiment the therapeutic agent is an inhibitor that reduces the amount of neoactive enzyme activity, e.g., by interacting with, e.g., binding to, mutant IDH, e.g., IDH1 or IDH2, protein. In an embodiment the inhibitor is other than an antibody.
-
In an embodiment the therapeutic agent is an inhibitor that is a small molecule and interacts with, e.g., binds, the mutant RNA, e.g., mutant IDH1 mRNA.
-
In an embodiment the therapeutic agent is an inhibitor that interacts directly with, e.g., binds, either the mutant IDH, e.g., IDH1 or IDH2, protein or interacts directly with, e.g., binds, the mutant IDH mRNA, e.g., IDH1 or IDH2 mRNA.
-
In an embodiment the therapeutic agent is administered.
-
In an embodiment the treatment: inhibits, e.g., specifically, a neoactivity of IDH1 or IDH2 (e.g., a neoactivity of IDH1), e.g., a neoactivity described herein; or inhibits both the wildtype and activity and a neoactivity of IDH1 or IDH2 (e.g., a neoactivity of IDH1), e.g., a neoactivity described herein In an embodiment, the subject is subsequently evaluated or monitored by a method described herein, e.g., the analysis of the presence, distribution, or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, e.g., to evaluate response to the treatment or progression of disease.
-
In an embodiment the treatment is selected on the basis that it: inhibits, e.g., specifically, a neoactivity of IDH1 or IDH2 (e.g., a neoactivity of IDH1), e.g., alpha hydroxy neoactivity, e.g., 2HG neoactivity; or inhibits both the wildtype and activity and a neoactivity of IDH1 or IDH2 (e.g., a neoactivity of IDH1), e.g., a neoactivity described herein.
-
In an embodiment, the method comprises determining the possibility of a mutation other than a mutation in IDH1 or in IDH2. In embodiments a relatively high level of 2HG, e.g., R-2HG is indicative of another mutation.
-
In an embodiment, which embodiment includes selecting or administering a treatment for the subject, the subject:
-
has not yet been treated for the subject the cell proliferation-related disorder and the selected or administered treatment is the initial or first line treatment;
-
has already been treated for the cell proliferation-related and the selected or administered treatment results in an alteration of the existing treatment;
-
has already been treated for the cell proliferation-related, and the selected treatment results in continuation of the existing treatment; or
-
has already been treated for the cell proliferation-related disorder and the selected or administered treatment is different, e.g., as compared to what was administered prior to the evaluation or to what would be administered in the absence of elevated levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment, which embodiment includes selecting or administering a treatment for the subject, the selected or administered treatment can comprise:
-
a treatment which includes administration of a therapeutic agent at different, e.g., a greater (or lesser) dosage (e.g., different as compared to what was administered prior to the evaluation or to what would be administered in the absence of elevated levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG);
-
a treatment which includes administration of a therapeutic agent at a different frequency, e.g., more or less frequently, or not at all (e.g., different as compared to what was administered prior to the evaluation or to what would be administered in the absence of elevated levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG); or
-
a treatment which includes administration of a therapeutic agent in a different therapeutic setting (e.g., adding or deleting a second treatment from the treatment regimen) (e.g., different as compared to what was administered prior to the evaluation or to what would be administered in the absence of elevated levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG).
-
Methods of evaluating a subject described herein can comprise evaluating a neoactivity genotype or phenotype. Methods of obtaining and analyzing samples, and the in vivo analysis in subjects, described elsewhere herein, e.g., in the section entitled, “Methods of evaluating samples and/or subjects,” can be combined with this method.
-
In an embodiment the method comprises:
-
subjecting the subject (e.g., a subject not having 2-hydroxyglutaric aciduria) to imaging and/or spectroscopic analysis, e.g., magnetic resonance-based analysis, e.g., MRI and/or MRS e.g., imaging analysis, to provide a determination of the presence, distribution, or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, e.g., as associated with a tumor, e.g., a glioma, in the subject;
-
optionally storing a parameter related to the determination, e.g., the image or a value related to the image from the imaging analysis, in a tangible medium; and
-
responsive to the determination, performing one or more of: correlating the determination with outcome or with a prognosis; providing an indication of outcome or prognosis; providing a value for an analysis on which the evaluation is based, e.g., the presence, distribution, or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG; providing a recommendation for treatment of the subject; selecting a course of treatment for the subject, e.g., a course of treatment described herein, e.g., selecting a course of treatment that includes inhibiting a neoactivity of a mutant IDH, e.g., IDH1 or IDH2, allele, e.g., a neoactivity described herein; administering a course of treatment to the subject, e.g., a course of treatment described herein, e.g., a course of treatment that includes inhibiting a neoactivity of a mutant IDH, e.g., IDH1 or IDH2, allele, e.g., a neoactivity described herein; and memorializing a result of the method or a measurement made in the course of the method, e.g., one or more of the above and/or transmitting memorialization of one or more of the above to a party, e.g., the subject, a healthcare provider, or an entity that pays for the subject's treatment, e.g., a government, insurance company, or other third party payer.
-
In an embodiment the method comprises confirming or determining, e.g., by direct examination or evaluation of the subject, or sample e.g., tissue or bodily fluid (e.g., blood (e.g., blood plasma), urine, lymph, or cerebrospinal fluid) therefrom, (e.g., by DNA sequencing or immuno analysis or evaluation of the presence, distribution or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG), or receiving such information about the subject, that the subject has a cancer characterized by an IDH, e.g., IDH1 or IDH2, allele described herein, e.g., an IDH1 allele having His, Ser, Cys, Gly, Val, Pro or Leu at residue 132 (SEQ ID NO:8), in specific embodiments, an IDH1 allele having His, Ser, Cys, Gly, Val, or Leu at residue 132 or an IDH1 allele having His or Cys at residue 132; or an IDH2 allele having Lys, Gly, Met, Trp, Thr, or Ser at residue 172 (SEQ ID NO:10).
-
In an embodiment, prior to or after treatment, the method includes evaluating the growth, size, weight, invasiveness, stage or other phenotype of the cell proliferation-related disorder.
-
In an embodiment the cell proliferation-related disorder is a tumor of the CNS, e.g., a glioma, a leukemia, e.g., AML or ALL, e.g., B-ALL or T-ALL, prostate cancer, or myelodysplasia or myelodysplastic syndrome and the evaluation is a or b. In an embodiment the method comprises evaluating a sample, e.g., a sample described herein, e.g., a tissue, e.g., a cancer sample, or a bodily fluid, e.g., serum or blood, for increased alpha neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment, a subject is subjected to MRS and the evaluation comprises evaluating the presence or elevated amount of a peak correlated to or corresponding to 2HG, e.g., R-2HG, as determined by magnetic resonance. For example, a subject can be analyzed for the presence and/or strength of a signal at about 2.5 ppm to determine the presence and/or amount of 2HG, e.g., R-2HG in the subject.
-
In an embodiment the method comprises obtaining a sample from the subject and analyzing the sample, or analyzing the subject, e.g., by imaging the subject and optionally forming a representation of the image on a computer.
-
In an embodiment the results of the analysis is compared to a reference.
-
In an embodiment a value for a parameter correlated to the presence, distribution, or level, e.g., of 2HG, e.g., R-2HG, is determined. It can be compared with a reference value, e.g., the value for a reference subject not having abnormal presence, level, or distribution, e.g., a reference subject cell not having a mutation in IDH, e.g., IDH1 or IDH2, having a neoactivity described herein.
-
In an embodiment the method comprises determining if an IDH, e.g., IDH1 or IDH2, mutant allele that is associated with 2HG neoactivity is present. E.g., in the case of IDH1, the presence of a mutation at residue 132 associated with 2HG neoactivity can be determined. In the case of IDH2, the presence of a mutation at residue 172 associated with 2HG neoactivity can be determined. The determination can comprise sequencing a nucleic acid, e.g., genomic DNA or cDNA, from an affected cell, which encodes the relevant amino acid(s). The mutation can be a deletion, insertion, rearrangement, or substitution. The mutation can involve a single nucleotide, e.g., a single substitution, or more than one nucleotide, e.g., a deletion of more than one nucleotides.
-
In an embodiment the method comprises determining the sequence at position 394 or 395 of the IDH1 gene, or determining the identity of amino acid residue 132 (SEQ ID NO:8) in the IDH1 gene in a cell characterized by the cell proliferation related disorder.
-
In an embodiment the method comprises determining the amino acid sequence, e.g., by DNA sequencing, at position 172 of the IDH2 gene in a cell characterized by the cell proliferation related disorder.
-
In an embodiment a product of the neoactivity is 2-HG, e.g., R-2HG, which acts as a metabolite. In another embodiment a product of the neoactivity is 2HG, e.g., R-2HG, which acts as a toxin, e.g., a carcinogen.
-
In an embodiment the disorder is other than a solid tumor. In an embodiment the disorder is a tumor that, at the time of diagnosis or treatment, does not have a necrotic portion. In an embodiment the disorder is a tumor in which at least 30, 40, 50, 60, 70, 80 or 90% of the tumor cells carry an IHD, e.g., IDH1 or IDH2, mutation having 2HG neoactivity, at the time of diagnosis or treatment.
-
In an embodiment the cell proliferation-related disorder is a cancer, e.g., a cancer described herein, characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the tumor is characterized by increased levels of an alpha hydroxy neoactivity product, 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type.
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity, product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is a tumor of the CNS, e.g., a glioma, e.g., wherein the tumor is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. Gliomas include astrocytic tumors, oligodendroglial tumors, oligoastrocytic tumors, anaplastic astrocytomas, and glioblastomas. In an embodiment the tumor is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type. E.g., in an embodiment, the IDH1 allele encodes an IDH1 having other than an Arg at residue 132. E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Yan et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21). In an embodiment the allele encodes an IDH1 having His at residue 132. In an embodiment the allele encodes an IDH1 having Ser at residue 132.
-
In an embodiment the IDH1 allele has an A (or any other nucleotide other than C) at nucleotide position 394, or an A (or any other nucleotide other than G) at nucleotide position 395. In an embodiment the allele is a C394A, a C394G, a C394T, a G395C, a G395T or a G395A mutation, specifically C394A or a G395A mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having a glioma, wherein the cancer is characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having His, Ser, Cys, Gly, Val, Pro or Leu at residue 132 (SEQ ID NO:8) (e.g., His, Ser, Cys, Gly, Val, or Leu; or His or Cys).
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by an IDH1 allele described herein, e.g., an IDH1 allele having His, Ser, Cys, Gly, Val, Pro or Leu at residue 132 (SEQ ID NO:8) (e.g., His, Ser, Cys, Gly, Val, or Leu; or His or Cys).
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity, product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment, the cell proliferation disorder is fibrosarcoma or paraganglioma wherein the cancer is characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment, the cell proliferation disorder is fibrosarcoma or paraganglioma wherein the cancer is characterized by an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment, the cell proliferation disorder is fibrosarcoma or paraganglioma wherein the cancer is characterized by increased levels of an alpha hydroxy neoactivity, product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is localized or metastatic prostate cancer, e.g., prostate adenocarcinoma, e.g., wherein the cancer is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the cancer is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type.
-
E.g., in an embodiment, the IDH1 allele encodes an IDH1 having other than an Arg at residue 132. E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Kang et al, 2009, Int. J. Cancer, 125: 353-355 at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21) (e.g., His, Ser, Cys, Gly, Val, or Leu). In an embodiment the allele encodes an IDH1 having His or Cys at residue 132.
-
In an embodiment the IDH1 allele has a T (or any other nucleotide other than C) at nucleotide position 394, or an A (or any other nucleotide other than G) at nucleotide position 395. In an embodiment the allele is a C394T or a G395A mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having prostate cancer, e.g., prostate adenocarcinoma, wherein the cancer is characterized by an IDH1 allele described herein, e.g., an IDH1 allele having His or Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having prostate cancer, e.g., prostate adenocarcinoma, on the basis of the cancer being characterized by an IDH1 allele described herein, e.g., an IDH1 allele having His or Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having prostate cancer, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is a hematological cancer, e.g., a leukemia, e.g., AML, or ALL, wherein the hematological cancer is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the cancer is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type. In an embodiment the method comprises evaluating a serum or blood sample for increased alpha neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is acute lymphoblastic leukemia (e.g., an adult or pediatric form), e.g., wherein the acute lymphoblastic leukemia (sometimes referred to herein as ALL) is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. The ALL can be, e.g., B-ALL or T-ALL. In an embodiment the cancer is characterized by increased levels of 2 an alpha hydroxy neoactivity product, e.g., HG, e.g., R-2HG, as compared to non-diseased cells of the same type. E.g., in an embodiment, the IDH1 allele is an IDH1 having other than an Arg at residue 132 (SEQ ID NO:8). E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Kang et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21) (e.g., His, Ser, Cys, Gly, Val, or Leu). In an embodiment the allele encodes an IDH1 having Cys at residue 132.
-
In an embodiment the IDH1 allele has a T (or any other nucleotide other than C) at nucleotide position 394. In an embodiment the allele is a C394T mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, characterized by an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 according to the sequence of SEQ ID NO:8.
-
In an embodiment the method comprises selecting a subject ALL, e.g., B-ALL or T-ALL, on the basis of cancer being characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is acute myelogenous leukemia (e.g., an adult or pediatric form), e.g., wherein the acute myelogenous leukemia (sometimes referred to herein as AML) is characterized by an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the cancer is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type. E.g., in an embodiment, the IDH1 allele is an IDH1 having other than an Arg at residue 132 (SEQ ID NO:8). E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Kang et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21) (e.g., His, Ser, Cys, Gly, Val or Leu). In an embodiment the allele encodes an IDH1 having Cys, His or Gly at residue 132, specifically, Cys.
-
In an embodiment the IDH1 allele has a T (or any other nucleotide other than C) at nucleotide position 394. In an embodiment the allele is a C394T mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having acute myelogenous lymphoplastic leukemia (AML) characterized by an IDH1 allele described herein, e.g., an IDH1 allele having Cys, His or Gly at residue 132 according to the sequence of SEQ ID NO:8, specifically, Cys.
-
In an embodiment the method comprises selecting a subject having acute myelogenous lymphoplastic leukemia (AML) on the basis of cancer being characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having Cys, His or Gly at residue 132 (SEQ ID NO:8), specifically, Cys.
-
In an embodiment the method comprises selecting a subject having acute myelogenous lymphoplastic leukemia (AML), on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG. In an embodiment the method comprises evaluating a serum or blood sample for increased alpha neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the method further comprises evaluating the subject for the presence of a mutation in the NRAS or NPMc gene.
-
In an embodiment the cell proliferation-related disorder is myelodysplasia or myelodysplastic syndrome, e.g., wherein the myelodysplasia or myelodysplastic syndrome is characterized by having an IDH1 somatic mutant having alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., a mutant described herein. In an embodiment the disorder is characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, as compared to non-diseased cells of the same type. E.g., in an embodiment, the IDH1 allele is an IDH1 having other than an Arg at residue 132 (SEQ ID NO:8). E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Kang et al., according to the sequence of SEQ ID NO:8 (see also FIG. 21), specifically, His, Ser, Cys, Gly, Val, or Leu. In an embodiment the allele encodes an IDH1 having Cys at residue 132.
-
In an embodiment the IDH1 allele has a T (or any other nucleotide other than C) at nucleotide position 394. In an embodiment the allele is a C394T mutation according to the sequence of SEQ ID NO:5.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome characterized by an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 according to the sequence of SEQ ID NO:8.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome on the basis of cancer being characterized by having an IDH1 allele described herein, e.g., an IDH1 allele having Cys at residue 132 (SEQ ID NO:8).
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG. In an embodiment the method comprises evaluating a serum or blood sample for increased alpha neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is a glioma, characterized by a mutation, or preselected allele, of IDH2 associated with an alpha hydroxy neoactivity, e.g., 2HG neoactivity. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), specifically, Lys, Gly, Met, Trp or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment the method comprises selecting a subject having a glioma, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys, Gly, Met, Trp, Thr, or Ser at residue 172 (SEQ ID NO:10), specifically Lys, Gly, Met, Trp, or Ser; or Lys or Met.
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys, Gly, Met, Trp, Thr, or Ser at residue 172 (SEQ ID NO:10), specifically Lys, Gly, Met, Trp, or Ser; or Lys or Met.
-
In an embodiment the method comprises selecting a subject having a glioma, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is a prostate cancer, e.g., prostate adenocarcinoma, characterized by a mutation, or preselected allele, of IDH2 associated with an alpha hydroxy neoactivity, e.g., 2HG neoactivity. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), specifically Lys, Gly, Met, Trp, or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment the method comprises selecting a subject having a prostate cancer, e.g., prostate adenocarcinoma, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having a prostate cancer, e.g., prostate adenocarcinoma, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having a prostate cancer, e.g., prostate adenocarcinoma, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is ALL, e.g., B-ALL or T-ALL, characterized by a mutation, or preselected allele, of IDH2 associated with an alpha hydroxy neoactivity, e.g., 2HG neoactivity. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), specifically Lys, Gly, Met, Trp, or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having ALL, e.g., B-ALL or T-ALL, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG. In an embodiment the method comprises evaluating a serum or blood sample for increased alpha neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is AML, characterized by a mutation, or preselected allele, of IDH2 associated with an alpha hydroxy neoactivity, e.g., 2HG neoactivity. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), specifically Lys, Gly, Met, Trp, or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment the method comprises selecting a subject having AML, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having AML, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having AML, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG. In an embodiment the method comprises evaluating a serum or blood sample for increased alpha neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In an embodiment the cell proliferation-related disorder is myelodysplasia or myelodysplastic syndrome, characterized by a mutation, or preselected allele, of IDH2. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), specifically Lys, Gly, Met, Trp, or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome, wherein the cancer is characterized by having an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome, on the basis of the cancer being characterized by an IDH2 allele described herein, e.g., an IDH2 allele having Lys or Met at residue 172 (SEQ ID NO:10).
-
In an embodiment the method comprises selecting a subject having myelodysplasia or myelodysplastic syndrome, on the basis of the cancer being characterized by increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG. In an embodiment the method comprises evaluating a serum or blood sample for increased alpha neoactivity product, e.g., 2HG, e.g., R-2HG.
-
In another aspect the invention features a pharmaceutical composition of an inhibitor (e.g., a small molecule or a nucleic acid-based inhibitor) described herein.
-
In an embodiment a mutant protein specific reagent, e.g., an antibody that specifically binds an IDH mutant protein, e.g., an antibody that specifically binds an IDH1-R132H mutant protein, can be used to detect neoactive mutant enzyme see, for example, that described by Y. Kato et al., “A monoclonal antibody IMab-1 specifically recognizes IDH1R132H, the most common glioma-derived mutation: (Kato, Biochem. Biophys. Res. Commun. (2009), which is hereby incorporated by reference in its entirety.
-
In another aspect, the invention features, a method of evaluating a candidate compound, e.g., for the ability to inhibit a neoactivity of a mutant enzyme, e.g., for use as an anti-proliferative or anti-cancer agent. In an embodiment the mutant enzyme is an IDH, e.g., an IDH1 or IDH2 mutant, e.g., a mutant described herein. In an embodiment the neaoctivity is alpha hydroxy neoactivity, e.g., 2HG neoactivity. The method comprises:
-
optionally supplying the candidate compound;
-
contacting the candidate compound with a mutant enzyme having a neoactivity, or with another enzyme, a referred to herein as a proxy enzyme, having an activity, referred to herein as a proxy activity, which is the same as the neoactivity (or with a cell or cell lysate comprising the same); and
-
evaluating the ability of the candidate compound to modulate, e.g., inhibit or promote, the neoactivity or the proxy activity, thereby evaluating the candidate compound.
-
In an embodiment the mutant enzyme is a mutant IDH1, e.g., an IDH1 mutant described herein, and the neoactivity is an alpha hydroxy neoactivity, e.g., 2HG neoactivity. Mutations associated with 2HG neoactivity in IDH1 include mutations at residue 132, e.g., R132H, R132C, R132S, R132G, R132L, or R132V, more specifically, R132H or R132C.
-
In an embodiment the mutant enzyme is a mutant IDH2, e.g., an IDH2 mutant described herein, and the neoactivity is an alpha hydroxy neoactivity, e.g., 2HG neoactivity. Mutations associated with 2HG neoactivity in IDH2 include mutations at residue 172, e.g., R172K, R172M, R172S, R172G, or R172W.
-
In an embodiment the method includes evaluating the ability of the candidate compound to inhibit the neoactivity or the proxy activity.
-
In an embodiment the method further comprises evaluating the ability of the candidate compound to inhibit the forward reaction of non-mutant or wild type enzyme activity, e.g., in the case of IDH, e.g., IDH1 or IDH2, the conversion of isocitrate to α-ketoglutarate (or an intermediate thereof, including the reduced hydroxyl intermediate).
-
In an embodiment, the contacting step comprises contacting the candidate compound with a cell, or a cell lysate thereof, wherein the cell comprises a mutant enzyme having the neoactivity or an enzyme having the activity.
-
In an embodiment, the cell comprises a mutation, or preselected allele, of a mutant IDH1 gene. E.g., in an embodiment, the IDH1 allele encodes an IDH1 having other than an Arg at residue 132. E.g., the allele can encode His, Ser, Cys, Gly, Val, Pro or Leu, or any other residue described in Yan et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21), specifically His, Ser, Cys, Gly, Val, or Leu.
-
In an embodiment the allele encodes an IDH1 having His at residue 132.
-
In an embodiment the allele encodes an IDH1 having Ser at residue 132.
-
In an embodiment the allele is an Arg132His mutation, or an Arg132Ser mutation, according to the sequence of SEQ ID NO:8 (see FIGS. 2 and 21).
-
In an embodiment, the cell comprises a mutation, or preselected allele, of a mutant IDH2 gene. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), specifically, Lys, Gly, Met, Trp, or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment, the cell includes a heterologous copy of a mutant IDH gene, e.g., a mutant IDH1 or IDH2 gene. (Heterologous copy refers to a copy introduced or formed by a genetic engineering manipulation.)
-
In an embodiment, the cell is transfected (e.g., transiently or stably transfected) or transduced (e.g., transiently or stably transduced) with a nucleic acid sequence encoding an IDH, e.g., IDH1 or IDH2, described herein, e.g., an IDH1 having other than an Arg at residue 132. In an embodiment, the IDH, e.g., IDH1 or IDH2, is epitope-tagged, e.g., myc-tagged.
-
In an embodiment, the cell, e.g., a cancer cell, is non-mutant or wild type for the IDH, e.g., IDH1 or IDH2, allele. The cell can include a heterologous IDH1 or IDH2 mutant.
-
In an embodiment, the cell is a cultured cell, e.g., a primary cell, a secondary cell, or a cell line. In an embodiment, the cell is a cancer cell, e.g., a glioma cell (e.g., a glioblastoma cell), a prostate cancer cell, a leukemia cell (e.g., an ALL, e.g., B-ALL or T-ALL, cell or AML cell) or a cell characterized by myelodysplasia or myelodysplastic syndrome. In embodiment, the cell is a 293T cell, a U87MG cell, or an LN-18 cell (e.g., ATCC HTB-14 or CRL-2610).
-
In an embodiment, the cell is from a subject, e.g., a subject having cancer, e.g., a cancer characterized by an IDH, e.g., IDH1 or IDH2, allele described herein, e.g., an IDH1 allele having His, Ser, Cys, Gly, Val, Pro or Leu at residue 132 (SEQ ID NO:8); specifically His or Cys; or an IDH2 allele having Lys, Gly, Met, Trp, Thr, or Ser at residue 172 (SEQ ID NO:10), specifically Lys, Gly, Met, Trp, or Ser.
-
In an embodiment, the evaluating step comprises evaluating the presence and/or amount of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, e.g., in the cell lysate or culture medium, e.g., by LC-MS.
-
In an embodiment, the evaluating step comprises evaluating the presence and/or amount of an alpha hydroxy neoactivity, e.g., 2HG neoactivity, in the cell lysate or culture medium.
-
In an embodiment, the method further comprises evaluating the presence/amount one or more of TCA metabolite(s), e.g., citrate, α-KG, succinate, fumarate, and/or malate, e.g., by LC-MS, e.g., as a control.
-
In an embodiment, the method further comprises evaluating the oxidation state of NADPH, e.g., the absorbance at 340 nm, e.g., by spectrophotometer.
-
In an embodiment, the method further comprises evaluating the ability of the candidate compound to inhibit a second enzymatic activity, e.g., the forward reaction of non-mutant or wild type enzyme activity, e.g., in the case of IDH1 or IDH2 (e.g., IDH1), the conversion of isocitrate to α-ketoglutarate (or an intermediate thereof, including the reduced hydroxyl intermediate).
-
In an embodiment, the candidate compound is a small molecule, a polypeptide, peptide, a carbohydrate based molecule, or an aptamer (e.g., a nucleic acid aptamer, or a peptide aptamer). The method can be used broadly and can, e.g., be used as one or more of a primary screen, to confirm candidates produced by this or other methods or screens, or generally to guide drug discovery or drug candidate optimization.
-
In an embodiment, the method comprises evaluating, e.g., confirming, the ability of a candidate compound (e.g., a candidate compound which meets a predetermined level of inhibition in the evaluating step) to inhibit the neoactivity or proxy activity in a second assay.
-
In an embodiment, the second assay comprises repeating one or more of the contacting and/or evaluating step(s) of the basic method.
-
In another embodiment, the second assay is different from the first. E.g., where the first assay can use a cell or cell lysate or other non-whole animal model the second assay can use an animal model, e.g., a tumor transplant model, e.g., a mouse having an IDH, e.g., IDH1 or IDH2, mutant cell or tumor transplanted in it. E.g., a U87 cell, or glioma, e.g., glioblastoma, cell, harboring a transfected IDH, e.g., IDH1 or IDH2, neoactive mutant can be implanted as a xenograft and used in an assay. Primary human glioma or AML tumor cells can be grafted into mice to allow prorogation of the tumor and used in an assay. A genetically engineered mouse model (GEMM) harboring an IDH1 or IDH2 mutation and/or other mutation, e.g., a p53 null mutation, can also be used in an assay.
-
In an embodiment the method comprises:
-
optionally supplying the candidate compound;
-
contacting the candidate compound with a cell comprising a nucleic acid sequence, e.g., a heterologous sequence, encoding an IDH1 having other than an Arg at residue 132 (e.g., IDH1R132H) or an IDH2 having other than an Arg at residue 172 (specifically an IDH1 having other than an Arg at residue 132); and
-
evaluating the presence and/or amount of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, in the cell lysate or culture medium, by LC-MS, thereby evaluating the compound.
-
In an embodiment the result of the evaluation is compared with a reference, e.g., the level of product, e.g., an alpha hydroxy neoactivity product, e.g., 2HG. e.g., R-2HG, in a control cell, e.g., a cell having inserted therein a wild type or non-mutant copy of IDH1 or IDH2 (e.g., IDH1).
-
In another aspect, the invention features, a method of evaluating a candidate compound, e.g., for the ability to inhibit an RNA encoding a mutant enzyme having a neoactivity, e.g., for use as an anti-proliferative or anti-cancer agent. In an embodiment the mutant enzyme is an IDH, e.g., an IDH1 or IDH2 mutant, e.g., a mutant described herein. In an embodiment the neaoctivity is alpha hydroxy neoactivity, e.g., 2HG neoactivity. The method comprises:
-
optionally supplying the candidate compound, e.g., a nucleic acid based inhibitor (e.g., a dsRNA (e.g., siRNA or shRNA), an antisense, or a microRNA);
-
contacting the candidate compound with an RNA, e.g., an mRNA, which encodes IDH, e.g., an IDH1 or IDH2, e.g., an RNA that encode mutant enzyme having a neoactivity (or with a cell or cell lysate comprising the same); and
-
evaluating the ability of the candidate compound to inhibit the RNA, thereby evaluating the candidate compound. By inhibit the RNA means, e.g., to cleave or otherwise inactivate the RNA.
-
In an embodiment the RNA encodes a fusion of all or part of the IDH, e.g., IDH1 or IDH2, wildtype or mutant protein to a second protein, e.g., a reporter protein, e.g., a fluorescent protein, e.g., a green or red fluorescent protein.
-
In an embodiment the mutant enzyme is a mutant IDH1, e.g., an IDH1 mutant described herein, and the neoactivity is an alpha hydroxy neoactivity, e.g., 2HG neoactivity.
-
In an embodiment the mutant enzyme is a mutant IDH2, e.g., an IDH2 mutant described herein, and the neoactivity is an alpha hydroxy neoactivity, e.g., 2HG neoactivity.
-
In an embodiment, the contacting step comprises contacting the candidate compound with a cell, or a cell lysate thereof, wherein the cell comprises RNA encoding IDH, e.g., IDH1 or IDH2, e.g., a mutant IDH, e.g., IDH1 or IDH2, enzyme having the neoactivity.
-
In an embodiment, the cell comprises a mutation, or preselected allele, of a mutant IDH1 gene. E.g., in an embodiment, the IDH1 allele encodes an IDH1 having other than an Arg at residue 132. E.g., the allele can encode His, Ser, Cys, Gly, Val, Pro or Leu, or any other residue described in Yan et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21), specifically His, Ser, Cys, Gly, Val, or Leu.
-
In an embodiment the allele encodes an IDH1 having His at residue 132.
-
In an embodiment the allele encodes an IDH1 having Ser at residue 132.
-
In an embodiment the allele is an Arg132His mutation, or an Arg132Ser mutation, according to the sequence of SEQ ID NO:8 (see FIGS. 2 and 21).
-
In an embodiment, the cell comprises a mutation, or preselected allele, of a mutant IDH2 gene. E.g., in an embodiment, the IDH2 allele encodes an IDH2 having other than an Arg at residue 172. E.g., the allele encodes Lys, Gly, Met, Trp, Thr, Ser, or any residue described in described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22), specifically Lys, Gly, Met, Trp or Ser. In an embodiment the allele encodes an IDH2 having Lys at residue 172. In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment, the cell includes a heterologous copy of a wildtype or mutant IDH gene, e.g., a wildtype or mutant IDH1 or IDH2 gene. (Heterologous copy refers to a copy introduced or formed by a genetic engineering manipulation.) In an embodiment the heterologous gene comprises a fusion to a reporter protein, e.g., a fluorescent protein, e.g., a green or red fluorescent protein.
-
In an embodiment, the cell is transfected (e.g., transiently or stably transfected) or transduced (e.g., transiently or stably transduced) with a nucleic acid sequence encoding an IDH, e.g., IDH1 or IDH2, described herein, e.g., an IDH1 having other than an Arg at residue 132 or an IDH2 having other than an Arg at residue 172 (e.g., an IDH1 having other than an Arg at residue 132). In an embodiment, the IDH, e.g., IDH1 or IDH2, is epitope-tagged, e.g., myc-tagged.
-
In an embodiment, the cell, e.g., a cancer cell, is non-mutant or wild type for the IDH, e.g., IDH1 or IDH2, allele. The cell can include a heterologous IDH1 or IDH2 mutant.
-
In an embodiment, the cell is a cultured cell, e.g., a primary cell, a secondary cell, or a cell line. In an embodiment, the cell is a cancer cell, e.g., a glioma cell (e.g., a glioblastoma cell), a prostate cancer cell, a leukemia cell (e.g., an ALL, e.g., B-ALL or T-ALL cell or AML cell) or a cell characterized by myelodysplasia or myelodysplastic syndrome. In embodiment, the cell is a 293T cell, a U87MG cell, or an LN-18 cell (e.g., ATCC HTB-14 or CRL-2610).
-
In an embodiment, the cell is from a subject, e.g., a subject having cancer, e.g., a cancer characterized by an IDH, e.g., IDH1 or IDH2, allele described herein, e.g., an IDH1 allele having His, Ser, Cys, Gly, Val, Pro or Leu at residue 132 (SEQ ID NO:8); specifically His or Cys. In an embodiment, the cancer is characterized by an IDH2 allele having Lys, Gly, Met, Trp, Thr, or Ser at residue 172 (SEQ ID NO:10), specifically Lys, Gly, Met, Trp, or Ser.
-
In an embodiment, the method comprises a second assay and the second assay comprises repeating one or more of the contacting and/or evaluating step(s) of the basic method.
-
In another embodiment, the second assay is different from the first. E.g., where the first assay can use a cell or cell lysate or other non-whole animal model the second assay can use an animal model
-
In an embodiment the efficacy of the candidate is evaluated by its effect on reporter protein activity.
-
In another aspect, the invention features, a method of evaluating a candidate compound, e.g., for the ability to inhibit transcription of an RNA encoding a mutant enzyme having a neoactivity, e.g., for use as an anti-proliferative or anti-cancer agent. In an embodiment the mutant enzyme is an IDH, e.g., an IDH1 or IDH2 mutant, e.g., a mutant described herein. In an embodiment the neaoctivity is alpha hydroxy neoactivity, e.g., 2HG neoactivity. The method comprises:
-
optionally supplying the candidate compound, e.g., a small molecule, polypeptide, peptide, aptomer, a carbohydrate-based molecule or nucleic acid based molecule;
-
contacting the candidate compound with a system comprising a cell or cell lysate; and
-
evaluating the ability of the candidate compound to inhibit the translation of IDH, e.g., IDH1 or IDH2, RNA, e.g, thereby evaluating the candidate compound.
-
In an embodiment the system comprises a fusion gene encoding of all or part of the IDH, e.g., IDH1 or IDH2, wildtype or mutant protein to a second protein, e.g., a reporter protein, e.g., a fluorescent protein, e.g., a green or red fluorescent protein.
-
In an embodiment the mutant enzyme is a mutant IDH1, e.g., an IDH1 mutant described herein, and the neoactivity is alpha hydroxy neoactivity, e.g., 2HG neoactivity.
-
In an embodiment the mutant enzyme is a mutant IDH2, e.g., an IDH2 mutant described herein, and the neoactivity is alpha hydroxy neoactivity, e.g., 2HG neoactivity.
-
In an embodiment, the system includes a heterologous copy of a wildtype or mutant IDH gene, e.g., a wildtype or mutant IDH1 or IDH2 gene. (Heterologous copy refers to a copy introduced or formed by a genetic engineering manipulation.) In an embodiment the heterologous gene comprises a fusion to a reporter protein, e.g., a fluorescent protein, e.g., a green or red fluorescent protein.
-
In an embodiment the cell, e.g., a cancer cell, is non-mutant or wild type for the IDH, e.g., IDH1 or IDH2, allele. The cell can include a heterologous IDH1 or IDH2 mutant.
-
In an embodiment, the cell is a cultured cell, e.g., a primary cell, a secondary cell, or a cell line. In an embodiment, the cell is a cancer cell, e.g., a glioma cell (e.g., a glioblastoma cell), a prostate cancer cell, a leukemia cell (e.g., an ALL, e.g., B-ALL or T-ALL, cell or AML cell) or a cell characterized by myelodysplasia or myelodysplastic syndrome. In embodiment, the cell is a 293T cell, a U87MG cell, or an LN-18 cell (e.g., ATCC HTB-14 or CRL-2610).
-
In an embodiment, the cell is from a subject, e.g., a subject having cancer, e.g., a cancer characterized by an IDH, e.g., IDH1 or IDH2, allele described herein, e.g., an IDH1 allele having His, Ser, Cys, Gly, Val, Pro or Leu at residue 132 (SEQ ID NO:8); specifically His, Ser, Cys, Gly, Val, or Leu. In an embodiment, the cancer is characterized an IDH2 allele having Lys, Gly, Met, Trp, Thr, or Ser at residue 172 (SEQ ID NO:10).
-
In an embodiment, the method comprises a second assay and the second assay comprises repeating the method.
-
In another embodiment, the second assay is different from the first. E.g., where the first assay can use a cell or cell lysate or other non-whole animal model the second assay can use an animal model.
-
In an embodiment the efficacy of the candidate is evaluated by its effect on reporter protein activity.
-
In another aspect, the invention features, a method of evaluating a candidate compound, e.g., a therapeutic agent, or inhibitor, described herein in an animal model. The candidate compound can be, e.g., a small molecule, polypeptide, peptide, aptomer, a carbohydrate-based molecule or nucleic acid based molecule. The method comprises, contacting the candidate with the animal model and evaluating the animal model.
-
In an embodiment evaluating comprises;
-
determining an effect of the compound on the general health of the animal;
-
determining an effect of the compound on the weight of the animal;
-
determining an effect of the compound on liver function, e.g, on a liver enzyme;
-
determining an effect of the compound on the cardiovascular system of the animal;
-
determining an effect of the compound on neurofunction, e.g., on neuromuscular control or response;
-
determining an effect of the compound on eating or drinking;
-
determining the distribution of the compound in the animal;
-
determining the persistence of the compound in the animal or in a tissue or organ of the animal, e.g., determining plasma half-life; or
-
determining an effect of the compound on a selected cell in the animal;
-
determining an effect of the compound on the growth, size, weight, invasiveness or other phenotype of a tumor, e.g., an endogenous tumor or a tumor arising from introduction of cells from the same or a different species.
-
In an embodiment the animal is a non-human primate, e.g., a cynomolgus monkey or chimpanzee.
-
In an embodiment the animal is a rodent, e.g., a rat or mouse.
-
In an embodiment the animal is a large animal, e.g., a dog or pig, other than a non-human primate.
-
In an embodiment the evaluation is memorialized and optionally transmitted to another party.
-
In one aspect, the invention provides, a method of evaluating or processing a therapeutic agent, e.g., a therapeutic agent referred to herein, e.g., a therapeutic agent that results in a lowering of the level of a product of an IDH, e.g., IDH1 or IDH2, mutant having a neoactivity. In an embodiment the neoactivity is an alpha hydroxy neoactivity, e.g., 2HG neoactivity, and the level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG is lowered.
-
The method includes:
-
providing, e.g., by testing a sample, a value (e.g., a test value) for a parameter related to a property of the therapeutic agent, e.g., the ability to inhibit the conversion of alpha ketoglutarate to 2 hydroxyglutarate (i.e., 2HG), e.g., R-2 hydroxyglutarate (i.e., R-2HG), and,
-
optionally, providing a determination of whether the value determined for the parameter meets a preselected criterion, e.g., is present, or is present within a preselected range,
-
thereby evaluating or processing the therapeutic agent.
-
In an embodiment the therapeutic agent is approved for use in humans by a government agency, e.g., the FDA.
-
In an embodiment the parameter is correlated to the ability to inhibit 2HG neoactivity, and, e.g., the therapeutic agent is an inhibitor which binds to IDH1 or IDH2 protein and reduces an alpha hydroxy neoactivity, e.g., 2HG neoactivity.
-
In an embodiment the parameter is correlated to the level of mutant IDH, e.g., IDH1 or IDH2, protein, and, e.g., the therapeutic agent is an inhibitor which reduces the level of IDH1 or IDH2 mutant protein.
-
In an embodiment the parameter is correlated to the level of an RNA that encodes a mutant IDH, e.g., IDH1 or IDH2, protein, and, e.g., the therapeutic agent reduces the level of RNA, e.g., mRNA, that encodes IDH1 or IDH2 mutant protein.
-
In an embodiment the method includes contacting the therapeutic agent with a mutant IDH, e.g., IDH1 or IDH2, protein (or corresponding RNA).
-
In an embodiment, the method includes providing a comparison of the value determined for a parameter with a reference value or values, to thereby evaluate the therapeutic agent. In an embodiment, the comparison includes determining if a test value determined for the therapeutic agent has a preselected relationship with the reference value, e.g., determining if it meets the reference value. The value need not be a numerical value but, e.g., can be merely an indication of whether an activity is present.
-
In an embodiment the method includes determining if a test value is equal to or greater than a reference value, if it is less than or equal to a reference value, or if it falls within a range (either inclusive or exclusive of one or both endpoints). In an embodiment, the test value, or an indication of whether the preselected criterion is met, can be memorialized, e.g., in a computer readable record.
-
In an embodiment, a decision or step is taken, e.g., a sample containing the therapeutic agent, or a batch of the therapeutic agent, is classified, selected, accepted or discarded, released or withheld, processed into a drug product, shipped, moved to a different location, formulated, labeled, packaged, contacted with, or put into, a container, e.g., a gas or liquid tight container, released into commerce, or sold or offered for sale, or a record made or altered to reflect the determination, depending on whether the preselected criterion is met. E.g., based on the result of the determination or whether an activity is present, or upon comparison to a reference standard, the batch from which the sample is taken can be processed, e.g., as just described.
-
The evaluation of the presence or level of activity can show if the therapeutic agent meets a reference standard.
-
In an embodiment, methods and compositions disclosed herein are useful from a process standpoint, e.g., to monitor or ensure batch-to-batch consistency or quality, or to evaluate a sample with regard to a reference, e.g., a preselected value.
-
In an embodiment, the method can be used to determine if a test batch of a therapeutic agent can be expected to have one or more of the properties. Such properties can include a property listed on the product insert of a therapeutic agent, a property appearing in a compendium, e.g., the US Pharmacopea, or a property required by a regulatory agency, e.g., the FDA, for commercial use.
-
In an embodiment the method includes testing the therapeutic agent for its effect on the wildtype activity of an IDH, e.g., IDH1 or IDH2, protein, and providing a determination of whether the value determined meets a preselected criterion, e.g., is present, or is present within a preselected range.
-
In an embodiment the method includes:
-
contacting a therapeutic agent that is an inhibitor of IDH1 an alpha hydroxy neoactivity, e.g., 2HG neoactivity, with an IDH1 mutant having an alpha hydroxy neoactivity, e.g., 2HG neoactivity,
-
determining a value related to the inhibition of an alpha hydroxy neoactivity, e.g., 2HG neoactivity, and
-
comparing the value determined with a reference value, e.g., a range of values, for the inhibition of an alpha hydroxy neoactivity, e.g., 2HG neoactivity. In an embodiment the reference value is an FDA required value, e.g., a release criteria.
-
In an embodiment the method includes:
-
contacting a therapeutic agent that is an inhibitor of mRNA which encodes a mutant IDH1 having an alpha hydroxy neoactivity, e.g., 2HG neoactivity, with an mRNA that encodes an IDH1 mutant having an alpha hydroxy neoactivity, e.g., 2HG neoactivity,
-
determining a value related to the inhibition of the mRNA, and,
-
comparing the value determined with a reference value, e.g., a range of values for inhibition of the mRNA. In an embodiment the reference value is an FDA required value, e.g., a release criteria.
-
In one aspect, the invention features a method of evaluating a sample of a therapeutic agent, e.g., a therapeutic agent referred to herein, that includes receiving data with regard to an activity of the therapeutic agent; providing a record which includes said data and optionally includes an identifier for a batch of therapeutic agent; submitting said record to a decision-maker, e.g., a government agency, e.g., the FDA; optionally, receiving a communication from said decision maker; optionally, deciding whether to release market the batch of therapeutic agent based on the communication from the decision maker. In one embodiment, the method further includes releasing, or other wise processing, e.g., as described herein, the sample.
-
In another aspect, the invention features, a method of selecting a payment class for treatment with a therapeutic agent described herein, e.g., an inhibitor of IDH, e.g., IDH1 or IDH2, neoactivity, for a subject having a cell proliferation-related disorder. The method includes:
-
providing (e.g., receiving) an evaluation of whether the subject is positive for increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, or neoactivity, e.g., an alpha hydroxy neoactivity, e.g., 2HG neoactivity, a mutant IDH1 or IDH2 having neoactivity, e.g., an alpha hydroxy neoactivity, e.g., 2HG neoactivity, (or a corresponding RNA), or a mutant IDH, e.g., IDH1 or IDH2, somatic gene, e.g., a mutant described herein, and
-
performing at least one of (1) if the subject is positive selecting a first payment class, and (2) if the subject is a not positive selecting a second payment class.
-
In an embodiment the selection is memorialized, e.g., in a medical records system.
-
In an embodiment the method includes evaluation of whether the subject is positive for increased levels of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, or neoactivity, e.g., an alpha hydroxy neoactivity, e.g., 2HG neoactivity.
-
In an embodiment the method includes requesting the evaluation.
-
In an embodiment the evaluation is performed on the subject by a method described herein.
-
In an embodiment, the method comprises communicating the selection to another party, e.g., by computer, compact disc, telephone, facsimile, email, or letter.
-
In an embodiment, the method comprises making or authorizing payment for said treatment.
-
In an embodiment, payment is by a first party to a second party. In some embodiments, the first party is other than the subject. In some embodiments, the first party is selected from a third party payor, an insurance company, employer, employer sponsored health plan, HMO, or governmental entity. In some embodiments, the second party is selected from the subject, a healthcare provider, a treating physician, an HMO, a hospital, a governmental entity, or an entity which sells or supplies the drug. In some embodiments, the first party is an insurance company and the second party is selected from the subject, a healthcare provider, a treating physician, an HMO, a hospital, a governmental entity, or an entity which sells or supplies the drug. In some embodiments, the first party is a governmental entity and the second party is selected from the subject, a healthcare provider, a treating physician, an HMO, a hospital, an insurance company, or an entity which sells or supplies the drug.
-
As used herein, a cell proliferation-related disorder is a disorder characterized by unwanted cell proliferation or by a predisposition to lead to unwanted cell proliferation (sometimes referred to as a precancerous disorder). Examples of disorders characterized by unwanted cell proliferation include cancers, e.g., tumors of the CNS, e.g., a glioma. Gliomas include astrocytic tumors, oligodendroglial tumors, oligoastrocytic tumors, anaplastic astrocytomas, and glioblastomas. Other examples include hematological cancers, e.g., a leukemia, e.g., AML (e.g., an adult or pediatric form) or ALL, e.g., B-ALL or T-ALL (e.g., an adult or pediatric form), localized or metastatic prostate cancer, e.g., prostate adenocarcinoma, fibrosarcoma, and paraganglioma; specifically a leukemia, e.g., AML (e.g., an adult or pediatric form) or ALL, e.g., B-ALL or T-ALL (e.g., an adult or pediatric form), localized or metastatic prostate cancer, e.g., prostate adenocarcinoma. Examples of disorders characterized by a predisposition to lead to unwanted cell proliferation include myelodysplasia or myelodysplastic syndrome, which are a diverse collection of hematological conditions marked by ineffective production (or dysplasia) of myeloid blood cells and risk of transformation to AML.
-
As used herein, specifically inhibits a neoactivity (and similar language), means the neoactivity of the mutant enzyme is inhibited to a significantly greater degree than is the wildtype enzyme activity. By way of example, “specifically inhibits the 2HG neoactivity of mutant IDH1 (or IDH2)” means the 2HG neoactivity is inhibited to a significantly greater degree than is the forward reaction (the conversion of isocitrate to alpha ketoglutarate) of wildtype IDH1 (or IDH2) activity. In embodiments the neoactivity is inhibited at least 2, 5, 10, or 100 fold more than the wildtype activity. In embodiments an inhibitor that is specific for the 2HG neaoctivity of IDH, e.g., IDH1 or IDH2, will also inhibit another dehydrogenase, e.g., malate dehydrogenase. In other embodiments the specific inhibitor does inhibit other dehydrogenases, e.g., malate dehydrogenase.
-
As used herein, a cell proliferation-related disorder, e.g., a cancer, characterized by a mutation or allele, means a cell proliferation-related disorder having a substantial number of cells which carry that mutation or allele. In an embodiment at least 10, 25, 50, 75, 90, 95 or 99% of the cell proliferation-related disorder cells, e.g., the cells of a cancer, or a representative, average or typical sample of cancer cells, e.g., from a tumor or from affected blood cells, carry at least one copy of the mutation or allele. A cell proliferation-related disorder, characterized by a mutant IDH, e.g., a mutant IDH1 or mutant IDH2, having 2HG neoactivity is exemplary. In an embodiment the mutation or allele is present as a heterozygote at the indicated frequencies.
-
As used herein, a “SNP” is a DNA sequence variation occurring when a single nucleotide (A, T, C, or G) in the genome (or other shared sequence) differs between members of a species (or between paired chromosomes in an individual).
-
As used herein, a subject can be a human or non-human subject. Non-human subjects include non-human primates, rodents, e.g., mice or rats, or other non-human animals.
-
The details of one or more embodiments of the invention are set forth in the description below. Other features, objects, and advantages of the invention will be apparent from the description and the drawings, and from the claims.
BRIEF DESCRIPTION OF THE DRAWINGS
-
FIG. 1 depicts DNA sequence verification of pET41a-IDH1 and alignment against published IDH1 CDS. The sequence of IDH1 (CDS) corresponds to SEQ ID NO:5. The sequence of pET41a-IDH1 corresponds to SEQ ID NO:6, and the “consensus” sequence corresponds to SEQ ID NO:7.
-
FIG. 2 depicts DNA sequence verification of R132S and R132H mutants according to the SEQ ID NO:8. The amino acid sequence of IDH1 (SEQ ID NO:8) is provided in FIG. 21.
-
FIG. 3 depicts separation of wild type IDH1 protein on Ni-Sepharose column.
-
FIG. 4 depicts protein analysis of wild type IDH1 on SDS gel pre and post Ni column fractionation. T: total protein; I: insoluble fractions; S: soluble fraction; L: sample for loading on Ni-column. The numbers in the figure indicates the fraction numbers. Fractions #17˜#27 were collected for further purification.
-
FIG. 5A depicts separation of wild type IDH1 protein through SEC column S-200.
-
FIG. 5B depicts protein analysis of wild type IDH1 on SDS gel pre and post S-200 column fractionation. M: molecular weight marker; Ni: nickel column fraction prior to S-200; S200: fraction from SEC column.
-
FIG. 6 depicts separation of mutant R132S protein on Ni-Sepharose column.
-
FIG. 7 depicts protein analysis of mutant R132S on SDS gel pre and post Ni column fractionation. M: protein marker (KDa): 116, 66.2, 45, 35, 25, 18.4, 14.4; T: total cell protein; So: soluble fraction; In: insoluble fraction; Ft: flow through. #3-#7 indicate the corresponding eluted fraction numbers.
-
FIG. 8A depicts separation of mutant R132S protein through SEC column S-200.
-
FIG. 8B depicts protein analysis of mutant R132S on SDS gel post S-200 column fractionation. M: molecular weight marker; R132S: fraction from SEC column.
-
FIG. 9 depicts separation of mutant R132H protein on Ni-Sepharose column.
-
FIG. 10 depicts protein analysis of mutant R132H on SDS gel pre and post Ni column fractionation. M: protein marker (KDa): 116, 66.2, 45, 35, 25, 18.4, 14.4; T: total cell protein; So: soluble fraction; In: insoluble fraction; Ft: flow through; #5-#10 indicate the corresponding eluted fraction numbers; Ni: sample from Ni-Sepharose column, pool #5-#10 together.
-
FIG. 11A depicts separation of mutant R132H protein through SEC column S-200.
-
FIG. 11B depicts protein analysis of mutant R132H on SDS gel post S-200 column fractionation. M: molecular weight marker; R132H: fraction from SEC column.
-
FIG. 12A depicts Michaelis-Menten plot of IDH1 wild-type in the oxidative decarboxylation of ioscitrate to α-ketoglutarate.
-
FIG. 12B depicts Michaelis-Menten plot of R132H mutant enzyme in the oxidative decarboxylation of ioscitrate to α-ketoglutarate.
-
FIG. 12C depicts Michaelis-Menten plot of R132S mutant enzyme in the oxidative decarboxylation of ioscitrate to α-ketoglutarate.
-
FIG. 13A depicts α-KG inhibition of IDH1 wild-type.
-
FIG. 13B depicts α-KG inhibition of R132H mutant enzyme.
-
FIG. 13C depicts α-KG inhibition of R132S mutant enzyme.
-
FIG. 14 depicts IDH1 wt, R132H, and R132S in the conversion α-ketoglutarate to 2-hydroxyglutarate.
-
FIG. 15A depicts Substrate-Concentration velocity plot for R132H mutant enzyme.
-
FIG. 15B depicts Substrate-Concentration velocity plot for R132S mutant enzyme.
-
FIG. 16 depicts IDH1 wt, R132H, and R132S in the conversion α-ketoglutarate to 2-hydroxyglutarate with NADH.
-
FIG. 17A depicts oxalomalate inhibition to IDH1 wt.
-
FIG. 17B depicts oxalomalate inhibition to R132H.
-
FIG. 17C depicts oxalomalate inhibition to R132S.
-
FIG. 18A depicts LC-MS/MS analysis of the control reaction.
-
FIG. 18B depicts LC-MS/MS analysis of the reaction containing enzyme.
-
FIG. 18C depicts LC-MS/MS analysis of the spiked control reaction.
-
FIG. 19 depicts LC-MS/MS analysis of alpha-hydroxyglutarate.
-
FIG. 20 depicts LC-MS/MS analysis showing that R132H consumes α-KG to produce 2-hydroxyglutaric acid.
-
FIG. 21 depicts the amino acid sequence of IDH1 (SEQ ID NO:13) as described in GenBank Accession No. NP—005887.2 (GI No. 28178825) (record dated May 10, 2009).
-
FIG. 21A is the cDNA sequence of IDH1 as presented at GenBank Accession No. NM—005896.2 (Record dated May 10, 2009; GI No. 28178824) (SEQ ID NO:8).
-
FIG. 21B depicts the mRNA sequence of IDH1 as described in GenBank Accession No. NM—005896.2 (Record dated May 10, 2009; GI No. 28178824) (SEQ ID NO:9).
-
FIG. 22 is the amino acid sequence of IDH2 as presented at GenBank Accession No. NM—002168.2 (Record dated Aug. 16, 2009; GI28178831) (SEQ ID NO:10).
-
FIG. 22A is the cDNA sequence of IDH2 as presented at GenBank Accession No. NM—002168 (Record dated Aug. 16, 2009; GI28178831) (SEQ ID NO:11).
-
FIG. 22B is the mRNA sequence of IDH2 as presented at GenBank Accession No. NM—002168.2 (Record dated Aug. 16, 2009; GI28178831) (SEQ ID NO:12).
-
FIG. 23 depicts the progress of forward reactions (isocitrate to α-KG) for the mutant enzyme R132H and R132S.
-
FIG. 24A depicts LC-MS/MS analysis of derivatized 2-HG racemic mixture.
-
FIG. 24B depicts LC-MS/MS analysis of derivatized R-2HG standard.
-
FIG. 24C depicts LC-MS/MS analysis of a coinjection of derivatized 2-HG racemate and R-2-HG standard.
-
FIG. 24D depicts LC-MS/MS analysis of the derivatized neoactivity reaction product.
-
FIG. 24E depicts LC-MS/MS analysis of a coinjection of the neoactivity enzyme reaction product and the R-2-HG standard.
-
FIG. 24F depicts LC-MS/MS analysis of a coinjection of the neoactivity enzyme reaction product and the 2-HG racemic mixture.
-
FIG. 25 depicts the inhibitory effect of 2-HG derived from the reduction of α-KG by ICDH1 R132H on the wild-type ICDH1 catalytic oxidative decarboxylation of isocitrate to α-KG.
-
FIG. 26A depicts levels of 2-HG in CRL-2610 cell lines expressing wildtype or IDH-1 R132H mutant protein.
-
FIG. 26B depicts levels of 2-HG in HTB-14 cell lines expressing wildtype or IDH-1 R132H mutant protein.
-
FIG. 27 depicts human IDH1 genomic DNA: intron/2nd exon sequence.
-
FIG. 28 depicts concentrations of 2HG in human malignant gliomas containing R132 mutations in IDH1. Human glioma samples obtained by surgical resection were snap frozen, genotyped to stratify as wild-type (WT) (N=10) or carrying an R132 mutant allele (Mutant) (n=12) and metabolites extracted for LC-MS analysis. Among the 12 mutant tumors, 10 carried a R132H mutation, one an R132S mutation, and one an R132G mutation. Each symbol represents the amount of the listed metabolite found in each tumor sample. Red lines indicate the group sample means. The difference in 2HG observed between WT and R132 mutant IDH1 mutant tumors was statistically significant by Student's t-test (p<0.0001). There were no statistically significant differences in αKG, malate, fumarate, succinate, or isocitrate levels between the WT and R132 mutant IDH1 tumors.
-
FIG. 29A depicts the structural analysis of R132H mutant IDH1. On left is shown an overlay structure of R132H mutant IDH1 and WT IDH1 in the ‘closed’ conformation. On the right is shown an overlay structure of WT IDH1 in the ‘open’ conformation with mutant IDH1 for comparison.
-
FIG. 29B depicts the close-up structural comparison of the R132H IDH1 (left) and wild-type (WT) IDH1 (right) active-site containing both αKG and NADPH. In addition to changes at residue 132, the position of the catalytic residues Tyr 139 and Lys 212 are different and αKG is oriented differently relative to NADPH for catalytic hydride transfer in the WT versus R132H mutant enzymes.
-
FIG. 30A depicts the enzymatic properties of IDH1 R132H mutants when recombinant human wild-type (WT) and R132H mutant (R132H) IDH1 enzymes were assessed for oxidative decarboxylation of isocitrate to αKG with NADP as cofactor. Different concentrations of enzyme were used to generate the curves.
-
FIG. 30B depicts the enzymatic properties of IDH R132 mutants when WT and R132H mutant IDH1 enzymes were assessed for reduction of αKG with NADPH as cofactor. Different concentrations of enzyme were used to generate the curves.
-
FIG. 30C depicts kinetic parameters of oxidative and reductive reactions as measured for WT and R132H IDH1 enzymes are shown. Km and kcat values for the reductive activity of the WT enzyme were unable to be determined as no measurable enzyme activity was detectable at any substrate concentration.
-
FIG. 31A depicts the LC-MS/MS analysis identifying 2HG as the reductive reaction product of recombinant human R132H mutant IDH1.
-
FIG. 31B depicts the diacetyl-L-tartaric anhydride derivatization and LC-MS/MS analysis of the chirality of 2HG produced by R132H mutant IDH1. Normalized LC-MS/MS signal for the reductive reaction (r×n) product alone, an R(−)-2HG standard alone, and the two together (Rxn+R(−)-2HG) are shown as is the signal for a racemic mixture of R(−) and S(+) forms (2HG Racemate) alone or with the reaction products (Rxn+Racemate).
-
FIG. 32A depicts SDS-PAGE and Western blot analyses of C-terminal affinity-purification tagged IDH1 R132S protein used for crystallization.
-
FIG. 32B depicts the chromatogram of FPLC analysis of the IDH1 R132S protein sample.
-
FIG. 33 depicts crystals obtained from a protein solution contained 5 mM NADP, 5 mM isocitrate, 10 mM Ca2+. Precipitant solution contained 100 mM MES (pH 6.0) and 20% PEG 6000 using a hanging drop method of crystallization.
-
FIG. 34 depicts crystal obtained from a protein solution contained 5 mM NADP, 5 mM α-ketoglutarate, 10 mM Ca2+. Precipitant contained 100 mM MES (pH 6.5) and 12% PEG 20000.
-
FIG. 35 is a bar graph depicting elevated NADPH reductive catalysis activity in IDH2-R172K mutant enzyme as compared to wildtype IDH2.
-
FIGS. 36A-C are graphs depicting the following: (A) Extracts from IDH1/2 wt (n=10), and IDH1/2 mutant (n=16) patient leukemia cells obtained at presentation and relapse, and IDH1 R132 mutant leukemia cells grown in culture for 14 days (n=14) analyzed by LC-MS to measure levels of 2-HG; and (B) 2-HG measured in serum of patients with IDH1 wt or IDH1 R132 mutant leukemia. In (A) and (B), each point represents an individual patient sample. Diamonds represent wildtype, circles represent IDH1 mutants, and triangles represent IDH2 mutants. Horizontal bars indicate the mean. (*) indicates a statistically significant difference relative to wild-type patient cells (p<0.05). (C) depicts In vitro growth curves of IDH1 R132 mutant and IDH1 wild-type AML cells.
-
FIG. 37 is a graph depicting the results of extracts from leukemia cells of AML patients carrying an IDH1/2 mutant (n=16) or wild-type (n=10) allele obtained at initial presentation and relapse assayed by LC-MS for levels of α-KG, succinate, malate, and fumarate. Each point represents an individual patient sample. Open circles represent wild-types, closed circles represent IDH1 mutants, and triangles represent IDH2 mutants. Horizontal bars represent the mean. There were no statistically significant differences between the wild-type and IDH1/2 mutant AML samples.
-
FIG. 38 depicts graphical representations of LC-MS analysis of in vitro reactions using recombinant IDH1 R132C and IDH2 R172K confirming that 2-HG and not isocitrate is the end product of the mutant enzyme reactions.
-
FIGS. 39A and B depict (A) the wild-type IDH1 enzyme catalysis of the oxidative decarboxylation of isocitrate to alpha-ketoglutarate with the concomitant reduction of NADP to NADPH; and (B) the IDH1 R132C mutant reduction of alpha-ketoglutarate to 2-hydroxyglutarate while oxidizing NADPH to NADP. These are referred to as the “forward” and “partial reverse” reactions, respectively.
DETAILED DESCRIPTION
-
The inventors have discovered that certain mutated forms of an enzyme (e.g., IDH1 or IDH2) have a gain of function, referred to herein as a neoactivity, which can be targeted in the treatment of a cell proliferation-related disorder, e.g., a proliferative disorder such as cancer. For example, in the case of a metabolic pathway enzyme, a gain of function or neoactivity can serve as a target for treatment of cancer. Described herein are methods and compositions for the treatment of a cell proliferation-related disorder, e.g., a proliferative disorder such as cancer. The methods include, e.g., treating a subject having a glioma or brain tumor characterized by a preselected IDH1 allele, e.g., an allele having A at position 394, such as a C394A, a C394G, a C394T, a G395C, a G395T or a G395A mutation, (e.g., a C394A mutant) or an A at position 395 (e.g., a G395A mutant) according to the sequence of SEQ ID NO:5, that encodes an IDH1 having His, Ser, Cys, Gly, Val, Pro or Leu at position 132 (e.g., His); or a preselected IDH2 allele that encodes an IDH2 having Lys, Gly, Met, Trp, Thr, or Ser at position 172 and having a neoactivity disclosed herein, by administering to the subject a therapeutically effective amount of an inhibitor of IDH1 or IDH2 (e.g., IDH1), e.g., a small molecule or nucleic acid. The nucleic acid based inhibitor is, for example, a dsRNA, e.g., a dsRNA that comprises the primary sequences of the sense strand and antisense strands of Tables 7-14. The dsRNA is composed of two separate strands, or a single strand folded to form a hairpin structure (e.g., a short hairpin RNA (shRNA)). In some embodiments, the nucleic acid based inhibitor is an antisense nucleic acid, such as an antisense having a sequence that overlaps, or includes, an antisense sequence provided in Tables 7-14.
Neoactivity of an Enzyme
-
Neoactivity, as used herein, means an activity that arises as a result of a mutation, e.g., a point mutation, e.g., a substitution, e.g., in the active site of an enzyme. In an embodiment the neoactivity is substantially absent from wild type or non-mutant enzyme. This is sometimes referred to herein as a first degree neoactivity. An example of a first degree neoactivity is a “gain of function” wherein the mutant enzyme gains a new catalytic activity. In an embodiment the neoactivity is present in wild type or non-mutant enzyme but at a level which is less than 10, 5, 1, 0.1, 0.01 or 0.001% of what is seen in the mutant enzyme. This is sometimes referred to herein as a second degree neoactivity. An example of a second degree neoactivity is a “gain of function” wherein the mutant enzyme has an increase, for example, a 5 fold increase in the rate of a catalytic activity possessed by the enzyme when lacking the mutation.
-
In some embodiments, a non-mutant form the enzyme, e.g., a wild type form, converts substance A (e.g., isocitrate) to substance B (e.g., α-ketoglutarate), and the neoactivity converts substance B (e.g., α-ketoglutarate) to substance C, sometimes referred to as the neoactivity product (e.g., 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate). In some embodiments, the enzyme is in a metabolic pathway, e.g., a metabolic pathway leading to fatty acid biosynthesis, glycolysis, glutaminolysis, the pentose phosphate shunt, the nucleotide biosynthetic pathway, or the fatty acid biosynthetic pathway, e.g., IDH1 or IDH2.
-
In some embodiments, a non-mutant form the enzyme, e.g., a wild type form, converts substance A to substance B, and the neoactivity converts substance B to substance A. In some embodiments, the enzyme is in a metabolic pathway, e.g., a metabolic pathway leading to fatty acid biosynthesis, glycolysis, glutaminolysis, the pentose phosphate shunt, the nucleotide biosynthetic pathway, or the fatty acid biosynthetic pathway.
Isocitrate Dehydrogenases
-
Isocitrate dehydrogenases (IDHs) catalyze the oxidative decarboxylation of isocitrate to 2-oxoglutarate (i.e., α-ketoglutarate). These enzymes belong to two distinct subclasses, one of which utilizes NAD(+) as the electron acceptor and the other NADP(+). Five isocitrate dehydrogenases have been reported: three NAD(+)-dependent isocitrate dehydrogenases, which localize to the mitochondrial matrix, and two NADP(+)-dependent isocitrate dehydrogenases, one of which is mitochondrial and the other predominantly cytosolic. Each NADP(+)-dependent isozyme is a homodimer.
-
IDH1 (isocitrate dehydrogenase 1 (NADP+), cytosolic) is also known as IDH; IDP; IDCD; IDPC or PICD. The protein encoded by this gene is the NADP(+)-dependent isocitrate dehydrogenase found in the cytoplasm and peroxisomes. It contains the PTS-1 peroxisomal targeting signal sequence. The presence of this enzyme in peroxisomes suggests roles in the regeneration of NADPH for intraperoxisomal reductions, such as the conversion of 2,4-dienoyl-CoAs to 3-enoyl-CoAs, as well as in peroxisomal reactions that consume 2-oxoglutarate, namely the alpha-hydroxylation of phytanic acid. The cytoplasmic enzyme serves a significant role in cytoplasmic NADPH production.
-
The human IDH1 gene encodes a protein of 414 amino acids. The nucleotide and amino acid sequences for human IDH1 can be found as GenBank entries NM—005896.2 and NP—005887.2 respectively. The nucleotide and amino acid sequences for IDH1 are also described in, e.g., Nekrutenko et al., Mol. Biol. Evol. 15:1674-1684 (1998); Geisbrecht et al., J. Biol. Chem. 274:30527-30533 (1999); Wiemann et al., Genome Res. 11:422-435 (2001); The MGC Project Team, Genome Res. 14:2121-2127 (2004); Lubec et al., Submitted (DEC-2008) to UniProtKB; Kullmann et al., Submitted (JUN-1996) to the EMBL/GenBank/DDBJ databases; and Sjoeblom et al., Science 314:268-274 (2006).
-
IDH2 (isocitrate dehydrogenase 2 (NADP+), mitochondrial) is also known as IDH; IDP; IDHM; IDPM; ICD-M; or mNADP-IDH. The protein encoded by this gene is the NADP(+)-dependent isocitrate dehydrogenase found in the mitochondria. It plays a role in intermediary metabolism and energy production. This protein may tightly associate or interact with the pyruvate dehydrogenase complex. Human IDH2 gene encodes a protein of 452 amino acids. The nucleotide and amino acid sequences for IDH2 can be found as GenBank entries NM—002168.2 and NP—002159.2 respectively. The nucleotide and amino acid sequence for human IDH2 are also described in, e.g., Huh et al., Submitted (NOV-1992) to the EMBL/GenBank/DDBJ databases; and The MGC Project Team, Genome Res. 14:2121-2127 (2004).
-
Non-mutant, e.g., wild type, IDH1 catalyzes the oxidative decarboxylation of ioscitrate to α-ketoglutarate thereby reducing NAD+ (NADP+) to NADP (NADPH), e.g., in the forward reaction:
-
Isocitrate+NAD+ (NADP+)→α-KG+CO2+NADH(NADPH)+H+
-
In some embodiments, the neoactivity of a mutant IDH1 can have the ability to convert α-ketoglutarate to 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate:
-
α-KG+NADH(NADPH)+H+2-hydroxyglutarate, e.g., R-2-hydroxyglutarate+NAD+ (NADP+).
-
In some embodiments, the neoactivity can be the reduction of pyruvate or malate to the corresponding α-hydroxyl compounds.
-
In some embodiments, the neoactivity of a mutant IDH1 can arise from a mutant IDH1 having a His, Ser, Cys, Gly, Val, Pro or Leu, or any other mutations described in Yan et al., at residue 132 (e.g., His, Ser, Cys, Gly, Val or Leu; or His, Ser, Cys or Lys). In some embodiments, the neoactivity of a mutant IDH2 can arise from a mutant IDH2 having a Lys, Gly, Met, Trp, Thr, or Ser (e.g., Lys, Gly, Met, Trp, or Ser; or Gly, Met or Lys), or any other mutations described in Yan H et al., at residue 172. Exemplary mutations include the following: R132H, R132C, R132S, R132G, R132L, and R132V.
-
In some embodiments, the mutant IDH1 and/or IDH2 (e.g., a mutant IDH1 and/or IDH2 having a neoactivity described herein) could lead to an increased level of 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate in a subject. The accumulation of 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate in a subject, e.g., in the brain of a subject, can be harmful. For example, in some embodiments, elevated levels of 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate can lead to and/or be predictive of cancer in a subject such as a cancer of the central nervous system, e.g., brain tumor, e.g., glioma, e.g., glioblastoma multiforme (GBM). Accordingly, in some embodiments, a method described herein includes administering to a subject an inhibitor of the neo activity.
-
Detection of 2-hydroxyglutarate
-
2-hydroxyglutarate can be detected, e.g., by LC/MS. To detect secreted 2-hydroxyglutarate in culture media, 500 μL aliquots of conditioned media can be collected, mixed 80:20 with methanol, and centrifuged at 3,000 rpm for 20 minutes at 4 degrees Celsius. The resulting supernatant can be collected and stored at −80 degrees Celsius prior to LC-MS/MS to assess 2-hydroxyglutarate levels. To measure whole-cell associated metabolites, media can be aspirated and cells can be harvested, e.g., at a non-confluent density. A variety of different liquid chromatography (LC) separation methods can be used. Each method can be coupled by negative electrospray ionization (ESI, −3.0 kV) to triple-quadrupole mass spectrometers operating in multiple reaction monitoring (MRM) mode, with MS parameters optimized on infused metabolite standard solutions. Metabolites can be separated by reversed phase chromatography using 10 mM tributyl-amine as an ion pairing agent in the aqueous mobile phase, according to a variant of a previously reported method (Luo et al. J Chromatogr A 1147, 153-64, 2007). One method allows resolution of TCA metabolites: t=0, 50% B; t=5, 95% B; t=7, 95% B; t=8, 0% B, where B refers to an organic mobile phase of 100% methanol. Another method is specific for 2-hydroxyglutarate, running a fast linear gradient from 50%-95% B (buffers as defined above) over 5 minutes. A Synergi Hydro-RP, 100 mm×2 mm, 2.1 μm particle size (Phenomonex) can be used as the column, as described above. Metabolites can be quantified by comparison of peak areas with pure metabolite standards at known concentration. Metabolite flux studies from 13C-glutamine can be performed as described, e.g., in Munger et al. Nat Biotechnol 26, 1179-86, 2008.
-
In an embodiment 2HG, e.g., R-2HG, is evaluated and the analyte on which the determination is based is 2HG, e.g., R-2HG. In an embodiment the analyte on which the determination is based is a derivative of 2HG, e.g., R-2HG, formed in process of performing the analytic method. By way of example such a derivative can be a derivative formed in MS analysis. Derivatives can include a salt adduct, e.g., a Na adduct, a hydration variant, or a hydration variant which is also a salt adduct, e.g., a Na adduct, e.g., as formed in MS analysis. Exemplary 2HG derivatives include dehydrated derivatives such as the compounds provided below or a salt adduct thereof:
-
-
Methods of Evaluating Samples and/or Subjects
-
This section provides methods of obtaining and analyzing samples and of analyzing subjects.
-
Embodiments of the method comprise evaluation of one or more parameters related to IDH, e.g., IDH1 or IDH2, an alpha hydroxy neoactivity, e.g., 2HG neoactivity, e.g., to evaluate the IDH1 or IDH2 2HG neoactivity genotype or phenotype. The evaluation can be performed, e.g., to select, diagnose or prognose the subject, to select a therapeutic agent, e.g., an inhibitor, or to evaluate response to the treatment or progression of disease. In an embodiment the evaluation, which can be performed before and/or after treatment has begun, is based, at least in part, on analysis of a tumor sample, cancer cell sample, or precancerous cell sample, from the subject. E.g., a sample from the patient can be analyzed for the presence or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, by evaluating a parameter correlated to the presence or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG. An alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, in the sample can be determined by a chromatographic method, e.g., by LC-MS analysis. It can also be determined by contact with a specific binding agent, e.g., an antibody, which binds the alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, and allows detection. In an embodiment the sample is analyzed for the level of neoactivity, e.g., an alpha hydroxy neoactivity, e.g., 2HG neoactivity. In an embodiment the sample is analysed for the presence of a mutant IDH, e.g., IDH1 or IDH2, protein having an alpha hydroxy neoactivity, e.g., 2HG neoactivity (or a corresponding RNA). E.g., a mutant protein specific reagent, e.g., an antibody that specifically binds an IDH mutant protein, e.g., an antibody that specifically binds an IDH1-R132H mutant protein or an IDH2-R172 mutant protein (e.g., an IDH1-R132H mutant protein), can be used to detect neoactive mutant enzyme In an embodiment a nucleic acid from the sample is sequenced to determine if a selected allele or mutation of IDH1 or IDH2 disclosed herein is present. In an embodiment the analysis is other than directly determining the presence of a mutant IDH, e.g., IDH1 or IDH2, protein (or corresponding RNA) or sequencing of an IDH, e.g., IDH1 or IDH2 gene. In an embodiment the analysis is other than directly determining, e.g., it is other than sequencing genomic DNA or cDNA, the presence of a mutation at residue 132 of IDH1 and/or a mutation at residue 172 of IDH2. E.g., the analysis can be the detection of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, or the measurement of the mutation's an alpha hydroxy neoactivity, e.g., 2HG neoactivity. In an embodiment the sample is removed from the patient and analyzed. In an embodiment the evaluation can include one or more of performing the analysis of the sample, requesting analysis of the sample, requesting results from analysis of the sample, or receiving the results from analysis of the sample. (Generally herein, analysis can include one or both of performing the underlying method or receiving data from another who has performed the underlying method.)
-
In an embodiment the evaluation, which can be performed before and/or after treatment has begun, is based, at least in part, on analysis of a tissue (e.g., a tissue other than a tumor sample), or bodily fluid, or bodily product. Exemplary tissues include lymph node, skin, hair follicles and nails. Exemplary bodily fluids include blood, plasma, urine, lymph, tears, sweat, saliva, semen, and cerebrospinal fluid. Exemplary bodily products include exhaled breath. E.g., the tissue, fluid or product can be analyzed for the presence or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, by evaluating a parameter correlated to the presence or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG. An alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, in the sample can be determined by a chromatographic method, e.g., by LC-MS analysis. It can also be determined by contact with a specific binding agent, e.g., an antibody, which binds the alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, and allows detection. In embodiments where sufficient levels are present, the tissue, fluid or product can be analyzed for the level of neoactivity, e.g., an alpha hydroxy neoactivity, e.g., the 2HG neoactivity. In an embodiment the sample is analysed for the presence of a mutant IDH, e.g., IDH1 or IDH2, protein having an alpha hydroxy neoactivity, e.g., 2HG neoactivity (or a corresponding RNA). E.g., a mutant protein specific reagent, e.g., an antibody that specifically binds an IDH mutant protein, e.g., an antibody that specifically binds an IDH1-R132H mutant protein or an IDH2-R172 mutant protein (e.g., an IDH1-R132H mutant protein), can be used to detect neoactive mutant enzyme. In an embodiment a nucleic acid from the sample is sequenced to determine if a selected allele or mutation of IDH1 or IDH2 disclosed herein is present. In an embodiment the analysis is other than directly determining the presence of a mutant IDH, e.g., IDH1 or IDH2, protein (or corresponding RNA) or sequencing of an IDH, e.g., IDH1 or IDH2 gene. E.g., the analysis can be the detection of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, or the measurement of 2HG neoactivity. In an embodiment the tissue, fluid or product is removed from the patient and analyzed. In an embodiment the evaluation can include one or more of performing the analysis of the tissue, fluid or product, requesting analysis of the tissue, fluid or product, requesting results from analysis of the tissue, fluid or product, or receiving the results from analysis of the tissue, fluid or product.
-
In an embodiment the evaluation, which can be performed before and/or after treatment has begun, is based, at least in part, on alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, imaging of the subject. In embodiments magnetic resonance methods are is used to evaluate the presence, distribution, or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, in the subject. In an embodiment the subject is subjected to imaging and/or spectroscopic analysis, e.g., magnetic resonance-based analysis, e.g., MRI and/or MRS e.g., analysis, and optionally an image corresponding to the presence, distribution, or level of an alpha hydroxy neoactivity product, e.g., 2HG, e.g., R-2HG, or of the tumor, is formed. Optionally the image or a value related to the image is stored in a tangible medium and/or transmitted to a second site. In an embodiment the evaluation can include one or more of performing imaging analysis, requesting imaging analysis, requesting results from imaging analysis, or receiving the results from imaging analysis.
Methods of Treating a Proliferative Disorder
-
Described herein are methods of treating a cell proliferation-related disorder, e.g., a cancer, e.g., a glioma, e.g., by inhibiting a neoactivity of a mutant enzyme, e.g., an enzyme in a metabolic pathway, e.g., a metabolic pathway leading to fatty acid biosynthesis, glycolysis, glutaminolysis, the pentose phosphate shunt, the nucleotide biosynthetic pathway, or the fatty acid biosynthetic pathway, e.g., IDH1 or IDH2. The cancer can be characterized by the presence of a neoactivity, such as a gain of function in one or more mutant enzymes (e.g., an enzyme in the metabolic pathway, e.g., a metabolic pathway leading to fatty acid biosynthesis, glycolysis, glutaminolysis, the pentose phosphate shunt, the nucleotide biosynthetic pathway, or the fatty acid biosynthetic pathway e.g., IDH1 or IDH2). In some embodiments, the gain of function is the conversion of α-ketoglutarate to 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate.
Compounds for the Treatment of Cancer
-
A candidate compound can be evaluated for modulation (e.g., inhibition) of neoactivity, for example, using an assay described herein. A candidate compound can also be evaluated for modulation (e.g., inhibition) of wild type or non-mutant activity. For example, the formation of a product or by-product of any activity (e.g., enzymatic activity) can be assayed, thus evaluating a candidate compound. In some embodiments, the activity (e.g., wild type/non-mutant or neoactivity) can be evaluated by measuring one or more readouts from an enzymatic assay. For example, the change in nature and/or amount of substrate and/or product can be measured, e.g., using methods such as fluorescent or radiolabeled substrates. Exemplary substrates and/or products include α-ketoglutarate, CO2, NADP, NADPH, NAD, NADH, and 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate. In some embodiments, the rate of reaction of the enzyme can also be evaluated as can the nature and/or amount of a product of the enzymatic reaction. In addition to the measurement of potential enzymatic activities, activity (e.g., wild type/non-mutant or neoactivity) can be detected by the quenching of protein fluorescence upon binding of a potential substrate, cofactor, or enzymatic activity modulator to the enzyme.
-
In one embodiment, assay progress can be monitored by changes in the OD340 or fluorescence of the NAD or NADP cofactor. In another embodiment, the reaction progress can be coupled to a secondary enzyme assay system in continuous mode or endpoint mode for increasing the dynamic range of the assay. For example, an endpoint assay can be performed by adding to the reaction an excess of diaphorase and rezasarin. Diaphorase consumes the remaining NADPH or NADH while producing resorufin from rezasarin. Resorufin is a highly fluorescent product which can be measured by fluorescence at Ex544 Em590. This not only terminates the reaction but also generates an easily detectable signal with greater quantum yield than the fluorescence of the cofactor.
-
A continuous assay can be implemented through coupling a product of the primary reaction to a secondary enzyme reaction that yields detectable results of greater dynamic range or more convenient detection mode. For example, inclusion in the reaction mix of aldehyde dehydrogenase (ALDH), which is an NADP+ dependent enzyme, and 6-methoxy-2-napthaldehye, a chromogenic substrate for ALDH, will result in the production of the fluorescent product 6-methoxy-2-napthoate (Ex310 Em 360) at a rate dependent on the production of NADP+ by isocitrate dehydrogenase. The inclusion of a coupling enzyme such as aldehyde dehydrogenase has the additional benefit of allowing screening of neoactivity irrespective of whether NADP+ or NAD+ is produced, since this enzyme is capable of utilizing both. Additionally, since the NADPH or NADH cofactor required for the “reverse” assay is regenerated, a coupled enzyme system which cycles the cofactor back to the IDH enzyme has the further advantage of permitting continuous assays to be conducted at cofactor concentrations much below Km for the purpose of enhancing the detection of competitive inhibitors of cofactor binding.
-
In yet a third embodiment of an activity (e.g., wild type/non-mutant or neoactivity) screen, one or a number of IDH substrates, cofactors, or products can be isotopically labeled with radioactive or “heavy” elements at defined atoms for the purpose of following specific substrates or atoms of substrates through the chemical reaction. For example, the alpha carbon of α-KG, isocitrate, or 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate may be 14C or 13C. Amount, rate, identity and structure of products formed can be analyzed by means known to those of skill in the art, for example mass spectroscopy or radiometric HPLC.
-
Compounds that inhibit a neoactivity, e.g., a neoactivity described herein, can include, e.g., small molecule, nucleic acid, protein and antibody.
-
Exemplary small molecules include, e.g, small molecules that bind to enzymes and decrease their activity, e.g., a neoactivity described herein. The binding of an inhibitor can stop a substrate from entering the enzyme's active site and/or hinder the enzyme from catalyzing its reaction. Inhibitor binding is either reversible or irreversible. Irreversible inhibitors usually react with the enzyme and change it chemically. These inhibitors can modify key amino acid residues needed for enzymatic activity. In contrast, reversible inhibitors bind non-covalently and different types of inhibition are produced depending on whether these inhibitors bind the enzyme, the enzyme-substrate complex, or both.
-
In some embodiments, the small molecule is oxalomalate, oxalofumarate, or oxalosuccinate.
-
In some embodiments, the small molecule is a compound of formula (X), or a compound as listed in Table 24a. The compound of formula (X) is provided below:
-
-
wherein X is C1-C6 alkylene (e.g., methylene), C(O), or C(O)C1-C6 alkylene;
-
wherein X is optionally substituted;
-
R1 is halo (e.g., fluoro), C1-C6 alkyl, C1-C6 haloalkyl, hydroxyl, C1-C6 alkoxy, cyano, nitro, amino, alkylamino, dialkylamino, amido, —C(O)OH, or C(O)OC1-C6alkyl; and
-
m is 0, 1, 2, or 3.
-
In some embodiments, the compound is a compound of formula (XI) or a pharmaceutically acceptable salt thereof or a compound listed in Table 24b
-
-
wherein:
W, X, Y and Z are each independently selected from CH or N;
B and B1 are independently selected from hydrogen, alkyl or when taken together with the carbon to which they are attached form a carbonyl group;
Q is C═O or SO2;
-
D and D1 are independently selected from a bond, oxygen or NRc;
A is optionally substituted aryl or optionally substituted heteroaryl;
R1 is independently selected from alkyl, acyl, cycloalkyl, aryl, heteroaryl, heterocyclyl, heterocyclylalkyl, cycloalkylalkyl, aralkyl, and heteroaralkyl; each of which may be optionally substituted with 0-3 occurrences of Rd;
each R3 is independently selected from halo, haloalkyl, alkyl and —ORa;
each Ra is independently selected from alkyl, and haloalkyl;
each Rb is independently alkyl;
each Rc is independently selected from hydrogen, alkyl and alkenyl;
each Rd is independently selected from halo, haloalkyl, alkyl, nitro, cyano, and —ORa, or two Rd taken together with the carbon atoms to which they are attached form an optionally substituted heterocyclyl;
n is 0, 1, or 2;
h is 0, 1, 2; and
g is 0, 1 or 2.
-
In some embodiments, the small molecule is a selective inhibitor of the neoactivity (e.g., relative to the wild type activity).
-
Nucleic acids can be used to inhibit a neoactivity, e.g., a neoactivity described herein, e.g., by decreasing the expression of the enzyme. Exemplary nucleic acids include, e.g., siRNA, shRNA, antisense RNA, aptamer and ribozyme. Art-known methods can be used to select inhibitory molecules, e.g., siRNA molecules, for a particular gene sequence.
-
Proteins can also be used to inhibit a neoactivity, e.g., a neoactivity described herein, by directly or indirectly binding to the enzyme and/or substrate, or competing binding to the enzyme and/or substrate. Exemplary proteins include, e.g., soluble receptors, peptides and antibodies. Exemplary antibodies include, e.g., whole antibody or a fragment thereof that retains its ability to bind to the enzyme or substrate.
-
Exemplary candidate compounds, which can be tested for inhibiting of a neoactivity described herein (e.g., a neoactivity associated with mutant IDH1), are described in the following references, each of which are incorporated herein by reference: Bioorganic & Medicinal Chemistry (2008), 16(7), 3580-3586; Free Radical Biology & Medicine (2007), 42(1), 44-51; KR 2005036293 A; Applied and Environmental Microbiology (2005), 71(9), 5465-5475; KR 2002095553 A; U.S. Pat. Appl. US 2004067234 A1; PCT Int. Appl. (2002), WO 2002033063 A1; Journal of Organic Chemistry (1996), 61(14), 4527-4531; Biochimica et Biophysica Acta, Enzymology (1976), 452(2), 302-9; Journal of Biological Chemistry (1975), 250(16), 6351-4; Bollettino—Societa Italiana di Biologia Sperimentale (1972), 48(23), 1031-5; Journal of Biological Chemistry (1969), 244(20), 5709-12.
Isomers
-
Certain compounds may exist in one or more particular geometric, optical, enantiomeric, diasteriomeric, epimeric, atropic, stereoisomer, tautomeric, conformational, or anomeric forms, including but not limited to, cis- and trans-forms; E- and Z-forms; c-, t-, and r-forms; endo- and exo-forms; R-, S-, and meso-forms; D- and L-forms; d- and l-forms; (+) and (−) forms; keto-, enol-, and enolate-forms; syn- and anti-forms; synclinal- and anticlinal-forms; α- and β-forms; axial and equatorial forms; boat-, chair-, twist-, envelope-, and halfchair-forms; and combinations thereof, hereinafter collectively referred to as “isomers” (or “isomeric forms”).
-
In one embodiment, a compound described herein, e.g., an inhibitor of a neoactivity or 2-HG is an enantiomerically enriched isomer of a stereoisomer described herein. For example, the compound has an enantiomeric excess of at least about 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99%. Enantiomer, when used herein, refers to either of a pair of chemical compounds whose molecular structures have a mirror-image relationship to each other.
-
In one embodiment, a preparation of a compound disclosed herein is enriched for an isomer of the compound having a selected stereochemistry, e.g., R or S, corresponding to a selected stereocenter, e.g., the 2-position of 2-hydroxyglutaric acid. 2HG can be purchased from commercial sources or can be prepared using methods known in the art, for example, as described in Org. Syn. Coll vol., 7, P-99, 1990. For example, the compound has a purity corresponding to a compound having a selected stereochemistry of a selected stereocenter of at least about 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99%.
-
In one embodiment, a composition described herein includes a preparation of a compound disclosed herein that is enriched for a structure or structures having a selected stereochemistry, e.g., R or S, at a selected stereocenter, e.g., the 2-position of 2-hydroxyglutaric acid. Exemplary R/S configurations can be those provided in an example described herein.
-
An “enriched preparation,” as used herein, is enriched for a selected stereoconfiguration of one, two, three or more selected stereocenters within the subject compound. Exemplary selected stereocenters and exemplary stereoconfigurations thereof can be selected from those provided herein, e.g., in an example described herein. By enriched is meant at least 60%, e.g., of the molecules of compound in the preparation have a selected stereochemistry of a selected stereocenter. In an embodiment it is at least 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, or 99%. Enriched refers to the level of a subject molecule(s) and does not connote a process limitation unless specified.
-
Note that, except as discussed below for tautomeric forms, specifically excluded from the term “isomers,” as used herein, are structural (or constitutional) isomers (i.e., isomers which differ in the connections between atoms rather than merely by the position of atoms in space). For example, a reference to a methoxy group, —OCH3, is not to be construed as a reference to its structural isomer, a hydroxymethyl group, —CH2OH. Similarly, a reference to ortho-chlorophenyl is not to be construed as a reference to its structural isomer, meta-chlorophenyl. However, a reference to a class of structures may well include structurally isomeric forms falling within that class (e.g., C1-7 alkyl includes n-propyl and iso-propyl; butyl includes n-, iso-, sec-, and tert-butyl; methoxyphenyl includes ortho-, meta-, and para-methoxyphenyl).
-
The above exclusion does not pertain to tautomeric forms, for example, keto-, enol-, and enolate-forms, as in, for example, the following tautomeric pairs: keto/enol (illustrated below), imine/enamine, amide/imino alcohol, amidine/amidine, nitroso/oxime, thioketone/enethiol, N-nitroso/hydroxyazo, and nitro/aci-nitro.
-
-
Note that specifically included in the term “isomer” are compounds with one or more isotopic substitutions. For example, H may be in any isotopic form, including 1H, 2H (D), and 3H (T); C may be in any isotopic form, including 12C, 13C, and 14C; O may be in any isotopic form, including 16O and 18O; and the like. Unless otherwise specified, a reference to a particular compound includes all such isomeric forms, including (wholly or partially) racemic and other mixtures thereof. Methods for the preparation (e.g., asymmetric synthesis) and separation (e.g., fractional crystallisation and chromatographic means) of such isomeric forms are either known in the art or are readily obtained by adapting the methods taught herein, or known methods, in a known manner.
Salts
-
It may be convenient or desirable to prepare, purify, and/or handle a corresponding salt of the active compound, for example, a pharmaceutically-acceptable salt. Examples of pharmaceutically acceptable salts are discussed in Berge et al., 1977, “Pharmaceutically Acceptable Salts.” J. Pharm. ScL. Vol. 66, pp. 1-19.
-
For example, if the compound is anionic, or has a functional group which may be anionic (e.g., —COOH may be —COO″), then a salt may be formed with a suitable cation. Examples of suitable inorganic cations include, but are not limited to, alkali metal ions such as Na+ and K+, alkaline earth cations such as Ca2+ and Mg2+, and other cations such as Al+3. Examples of suitable organic cations include, but are not limited to, ammonium ion (i.e., NH4+) and substituted ammonium ions (e.g., NH3R+, NH2R2+, NHR3+, NR4+). Examples of some suitable substituted ammonium ions are those derived from: ethylamine, diethylamine, dicyclohexylamine, triethylamine, butylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine, benzylamine, phenylbenzylamine, choline, meglumine, and tromethamine, as well as amino acids, such as lysine and arginine. An example of a common quaternary ammonium ion is N(CH3)4+.
-
If the compound is cationic, or has a functional group that may be cationic (e.g., —NH2 may • be —NH3+), then a salt may be formed with a suitable anion. Examples of suitable inorganic anions include, but are not limited to, those derived from the following inorganic acids: hydrochloric, hydrobromic, hydroiodic, sulfuric, sulfurous, nitric, nitrous, phosphoric, and phosphorous.
-
Examples of suitable organic anions include, but are not limited to, those derived from the following organic acids: 2-acetyoxybenzoic, acetic, ascorbic, aspartic, benzoic, camphorsulfonic, cinnamic, citric, edetic, ethanedisulfonic, ethanesulfonic, fumaric, glucheptonic, gluconic, glutamic, glycolic, hydroxymaleic, hydroxynaphthalene carboxylic, isethionic, lactic, lactobionic, lauric, maleic, malic, methanesulfonic, mucic, oleic, oxalic, palmitic, pamoic, pantothenic, phenylacetic, phenylsulfonic, propionic, pyruvic, salicylic, stearic, succinic, sulfanilic, tartaric, toluenesulfonic, and valeric. Examples of suitable polymeric organic anions include, but are not limited to, those derived from the following polymeric acids: tannic acid, carboxymethyl cellulose.
-
Unless otherwise specified, a reference to a particular compound also includes salt forms thereof.
Chemically Protected Forms
-
It may be convenient or desirable to prepare, purify, and/or handle the active compound in a chemically protected form. The term “chemically protected form” is used herein in the conventional chemical sense and pertains to a compound in which one or more reactive functional groups are protected from undesirable chemical reactions under specified conditions (e.g., pH, temperature, radiation, solvent, and the like). In practice, well known chemical methods are employed to reversibly render unreactive a functional group, which otherwise would be reactive, under specified conditions. In a chemically protected form, one or more reactive functional groups are in the form of a protected or protecting group (also known as a masked or masking group or a blocked or blocking group). By protecting a reactive functional group, reactions involving other unprotected reactive functional groups can be performed, without affecting the protected group; the protecting group may be removed, usually in a subsequent step, without substantially affecting the remainder of the molecule. See, for example, Protective Groups in Organic Synthesis (T. Green and P. Wuts; 3rd Edition; John Wiley and Sons, 1999). Unless otherwise specified, a reference to a particular compound also includes chemically protected forms thereof.
-
A wide variety of such “protecting,” “blocking,” or “masking” methods are widely used and well known in organic synthesis. For example, a compound which has two nonequivalent reactive functional groups, both of which would be reactive under specified conditions, may be derivatized to render one of the functional groups “protected,” and therefore unreactive, under the specified conditions; so protected, the compound may be used as a reactant which has effectively only one reactive functional group. After the desired reaction (involving the other functional group) is complete, the protected group may be “deprotected” to return it to its original functionality.
-
For example, a hydroxy group may be protected as an ether (—OR) or an ester (—OC(═O)R), for example, as: a t-butyl ether; a benzyl, benzhydryl (diphenylmethyl), or trityl (triphenylmethyl)ether; a trimethylsilyl or t-butyldimethylsilyl ether; or an acetyl ester (—OC(═O)CH3, —OAc).
-
For example, an aldehyde or ketone group may be protected as an acetal (R—CH(OR)2) or ketal (R2C(OR)2), respectively, in which the carbonyl group (>C═O) is converted to a diether (>C(OR)2), by reaction with, for example, a primary alcohol. The aldehyde or ketone group is readily regenerated by hydrolysis using a large excess of water in the presence of acid.
-
For example, an amine group may be protected, for example, as an amide (—NRCO—R) or a urethane (—NRCO—OR), for example, as: a methyl amide (—NHCO—CH3); a benzyloxy amide (—NHCO—OCH2C6H5, —NH-Cbz); as a t-butoxy amide (—NHCO—OC(CH3)3, —NH-Boc); a 2-biphenyl-2-propoxy amide (—NHCO—OC(CH3)2C6H4C6H5, —NH-Bpoc), as a 9-fluorenylmethoxy amide (—NH-Fmoc), as a 6-nitroveratryloxy amide (—NH-Nvoc), as a 2-trimethylsilylethyloxy amide (—NH-Teoc), as a 2,2,2-trichloroethyloxy amide (—NH-Troc), as an allyloxy amide (—NH-Alloc), as a 2(-phenylsulphonyl)ethyloxy amide (—NH-Psec); or, in suitable cases (e.g., cyclic amines), as a nitroxide radical (>N—O<<).
-
For example, a carboxylic acid group may be protected as an ester for example, as: an Ĉalkyl ester (e.g., a methyl ester; a t-butyl ester); a Cvrhaloalkyl ester (e.g., a C1-7-trihaloalkyl ester); a triC1-7alkylsilyl-Ci.7alkyl ester; or a C5.2oaryl-C1-7alkyl ester (e.g., a benzyl ester; a nitrobenzyl ester); or as an amide, for example, as a methyl amide.
-
For example, a thiol group may be protected as a thioether (—SR), for example, as: a benzyl thioether; an acetamidomethyl ether (—S—CH2NHC(═O)CH3).
Nucleic Acid Based Inhibitors
-
Nucleic acid-based inhibitors for inhibition IDH, e.g., IDH1, can be, e.g., double stranded RNA (dsRNA) that function, e.g., by an RNA interference (RNAi mechanism), an antisense RNA, or a microRNA (miRNA). In an embodiment the nucleic-acid based inhibitor binds to the target mRNA and inhibits the production of protein therefrom, e.g., by cleavage of the targent mRNA.
-
Double Stranded RNA (dsRNA)
-
A nucleic acid based inhibitor useful for decreasing IDH1 or IDH2 mutant function is, e.g., a dsRNA, such as a dsRNA that acts by an RNAi mechanism. RNAi refers to the process of sequence-specific post-transcriptional gene silencing in animals mediated by short interfering RNAs (siRNAs). dsRNAs as used herein are understood to include siRNAs. Typically, inhibition of IDH, e.g., IDH1, by dsRNAs does not trigger the interferon response that results from dsRNA-mediated activation of protein kinase PKR and 2′,5′-oligoadenylate synthetase resulting in non-specific cleavage of mRNA by ribonuclease L.
-
dsRNAs targeting an IDH, e.g., IDH1, enzyme, e.g., a wildtype or mutant IDH1, can be unmodified or chemically modified. The dsRNA can be chemically synthesized, expressed from a vector or enzymatically synthesized. The invention also features various chemically modified synthetic dsRNA molecules capable of modulating IDH1 gene expression or activity in cells by RNA interference (RNAi). The use of chemically modified dsRNA improves various properties of native dsRNA molecules, such as through increased resistance to nuclease degradation in vivo and/or through improved cellular uptake.
-
The dsRNAs targeting nucleic acid can be composed of two separate RNAs, or of one RNA strand, which is folded to form a hairpin structure. Hairpin dsRNAs are typically referred to as shRNAs.
-
An shRNA that targets IDH, e.g., a mutant or wildtype IDH1 gene can be expressed from a vector, e.g., viral vector, such as a lentiviral or adenoviral vector. In certain embodiments, a suitable dsRNA for inhibiting expression of an IDH1 gene will be identified by screening an siRNA library, such as an adenoviral or lentiviral siRNA library.
-
In an embodiment, a dsRNA that targets IDH, e.g., IDH1, is about 15 to about 30 base pairs in length (e.g., about 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, or 29) basepairs in length. In another embodiment, the dsRNA includes overhanging ends of about 1 to about 3 (e.g., about 1, 2, or 3) nucleotides. By “overhang” is meant that 3′-end of one strand of the dsRNA extends beyond the 5′-end of the other strand, or vice versa. The dsRNA can have an overhang on one or both ends of the dsRNA molecule. In some embodiments, the single-stranded overhang is located at the 3′-terminal end of the antisense strand, or, alternatively, at the 3′-terminal end of the sense strand. In some embodiments, the overhang is a TT or UU dinucleotide overhang, e.g., a TT or UU dinucleotide overhang. For example, in an embodiment, the dsRNA includes a 21-nucleotide antisense strand, a 19 base pair duplex region, and a 3′-terminal dinucleotide. In yet another embodiment, a dsRNA includes a duplex nucleic acid where both ends are blunt, or alternatively, where one of the ends is blunt.
-
In an embodiment, the dsRNA includes a first and a second strand, each strand is about 18 to about 28 nucleotides in length, e.g., about 19 to about 23 nucleotides in length, the first strand of the dsRNA includes a nucleotide sequence having sufficient complementarity to the IDH, e.g., IDH1, RNA for the dsRNA to direct cleavage of the IDH, e.g., IDH1, mRNA via RNA interference, and the second strand of the dsRNA includes a nucleotide sequence that is complementary to the first strand.
-
In an embodiment, a dsRNA targeting an IDH, e.g., IDH1, gene can target wildtype and mutant forms of the gene, or can target different allelic isoforms of the same gene. For example, the dsRNA will target a sequence that is identical in two or more of the different isoforms. In an embodiment, the dsRNA targets an IDH1 having G at position 395 or C at position 394 (e.g., a wildtype IDH1 RNA) and an IDH1 having A at position 395 or A at position 394, such as a C394A, a C394G, a C394T, a G395C, a G395T or a G395A mutation, (e.g., an IDH1 RNA carrying a G395A and/or a C394A mutation) (FIG. 2).
-
In an embodiment, a dsRNA will preferentially or specifically target a mutant IDH RNA, or a particular IDH polymorphism. In some embodiments, the IDH has a mutation at position 394 or 395 such as a C394A, a C394G, a C394T, a G395C, a G395T or a G395A mutation. For example, in an embodiment, the dsRNA targets an IDH1 RNA carrying an A at position 395, e.g., G395A, and in another embodiment, the dsRNA targets an IDH1 RNA carrying an A at position 394, e.g., C394A mutation.
-
In an embodiment, a dsRNA targeting an IDH RNA includes one or more chemical modifications. Non-limiting examples of such chemical modifications include without limitation phosphorothioate internucleotide linkages, 2′-deoxyribonucleotides, 2′-O-methyl ribonucleotides, 2′-deoxy-2′-fluoro ribonucleotides, “universal base” nucleotides, “acyclic” nucleotides, 5-C-methyl nucleotides, and terminal glyceryl and/or inverted deoxy a basic residue incorporation. Such chemical modifications have been shown to preserve RNAi activity in cells while at the same time, dramatically increasing the serum stability of these compounds. Furthermore, one or more phosphorothioate substitutions are well-tolerated and have been shown to confer substantial increases in serum stability for modified dsRNA constructs.
-
In an embodiment, a dsRNA targeting an IDH, e.g., IDH1, RNA includes modified nucleotides while maintaining the ability to mediate RNAi. The modified nucleotides can be used to improve in vitro or in vivo characteristics such as stability, activity, and/or bioavailability. For example, the dsRNA can include modified nucleotides as a percentage of the total number of nucleotides present in the molecule. As such, the dsRNA can generally include about 5% to about 100% modified nucleotides (e.g., about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95% or 100% modified nucleotides).
-
In some embodiments, the dsRNA targeting IDH, e.g., IDH1, is about 21 nucleotides long. In another embodiment, the dsRNA does not contain any ribonucleotides, and in another embodiment, the dsRNA includes one or more ribonucleotides. In an embodiment, each strand of the dsRNA molecule independently includes about 15 to about 30 (e.g., about 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30) nucleotides, wherein each strand includes about 15 to about 30 (e.g., about 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30) nucleotides that are complementary to the nucleotides of the other strand. In an embodiment, one of the strands of the dsRNA includes a nucleotide sequence that is complementary to a nucleotide sequence or a portion thereof of the IDH1 or IDH2 gene, and the second strand of the dsRNA includes a nucleotide sequence substantially similar to the nucleotide sequence of the IDH1 or IDH2 gene or a portion thereof.
-
In an embodiment, the dsRNA targeting IDH1 or IDH2 includes an antisense region having a nucleotide sequence that is complementary to a nucleotide sequence of the IDH1 or IDH2 gene or a portion thereof, and a sense region having a nucleotide sequence substantially similar to the nucleotide sequence of the IDH1 or IDH2 gene or a portion thereof. In an embodiment, the antisense region and the sense region independently include about 15 to about 30 (e.g., about 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30) nucleotides, where the antisense region includes about 15 to about 30 (e.g., about 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30) nucleotides that are complementary to nucleotides of the sense region.
-
As used herein, the term “dsRNA” is meant to include nucleic acid molecules that are capable of mediating sequence specific RNAi, such as short interfering RNA (siRNA), short hairpin RNA (shRNA), short interfering oligonucleotide, short interfering nucleic acid, short interfering modified oligonucleotide, chemically modified siRNA, post-transcriptional gene silencing RNA (ptgsRNA), and others. In addition, as used herein, the term “RNAi” is meant to include sequence specific RNA interference, such as post transcriptional gene silencing, translational inhibition, or epigenetics.
-
Nucleic Acid-Based IDH Inhibitors
-
In an embodiment the inhibitor is a nucleic acid-based inhibitor, such as a double stranded RNA (dsRNA) or antisense RNA that targets a mutant IDH, e.g., mutant IDH1 or IDH2.
-
In one embodiment, the nucleic acid based inhibitor, e.g., a dsRNA or antisense molecule, decreases or inhibits expression of an IDH1 having other than an Arg, e.g., having a His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Yan et al., N. Eng. J. Med. 360:765-73, at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21). In one embodiment, the nucleic acid based inhibitor decreases or inhibits expression of an IDH1 enzyme having His at residue 132
-
In an embodiment the nucleic acid-based inhibitor is a dsRNA that targets an mRNA that encodes an IDH1 allele described herein, e.g., an IDH1 allele having other than an Arg at residue 132. E.g., the allele encodes His, Ser, Cys, Gly, Val, Pro or Leu, or any residue described in Yan et al., at residue 132, according to the sequence of SEQ ID NO:8 (see also FIG. 21).
-
In an embodiment the allele encodes an IDH1 having His at residue 132.
-
In an embodiment the allele encodes an IDH1 having Ser at residue 132.
-
In an embodiment, the nucleic acid-based inhibitor is a dsRNA that targets IDH1, e.g., an IDH1 having an A or a T (or a nucleotide other than C) at nucleotide position 394 or an A (or a nucleotide other than G) at nucleotide position 395, e.g., a mutant allele carrying a C394T mutation or a G395A mutation according to the IDH1 sequence of SEQ ID NO:8 (see also FIG. 21A).
-
In an embodiment, the dsRNA targets an IDH1 having other than C, e.g., a T or an A, at nucleotide position 394 or and other than G, e.g., an A, at 395 (e.g., a mutant) and an IDH1 having a C at nucleotide position 394 or a G at nucleotide position 395 (e.g., a wildtype), e.g., by targeting a region of the IDH1 mRNA that is identical between the wildtype and mutant transcripts. In yet another embodiment, the dsRNA targets a particular mutant or polymorphism (such as a single nucleotide polymorphism (SNP)), but not a wildtype allele. In this case, the nucleic acid based inhibitor, e.g., a dsRNA, targets the region of the IDH1 containing the mutation.
-
In some embodiments, the nucleic acid based inhibitor, e.g., a dsRNA preferentially or specifically inhibits the product of a mutant IDH1 as compared to the product of a wildtype IDH1. In some embodiments, the IDH has a mutation at position 394 or 395 such as a C394A, a C394G, a C394T, a G395C, a G395T or a G395A mutation. For example, in one embodiment, a dsRNA targets a region of an IDH1 mRNA that carries the mutation (e.g., a C394A of C394T or a G395A mutation according to SEQ ID NO:5).
-
In one embodiment, the nucleic acid-based inhibitor is a dsRNA including a sense strand and an antisense strand having a primary sequence presented in Tables 7-14. In another embodiment, the nucleic acid based inhibitor is an antisense oligonucleotide that includes all or a part of an antisense primary sequence presented in Tables 7-14 or which targets the same or substantially the same region as does a dsRNA from Tables 7-14.
-
In one embodiment, the nucleic acid based inhibitor decreases or inhibits expression of an IDH2 having Lys, Gly, Met, Trp, Thr, Ser, or any residue described in Yan et al., at residue 172, according to the amino acid sequence of SEQ ID NO:10 (see also FIG. 22). In one embodiment, the nucleic acid based inhibitor decreases or inhibits expression of an IDH2 enzyme having Lys at residue 172.
-
In an embodiment the nucleic acid-based inhibitor is a dsRNA that targets an mRNA that encodes an IDH2 allele described herein, e.g., an IDH2 allele having other than an Arg at residue 172. E.g., the allele can have Lys, Gly, Met, Trp, Thr, Ser, or any residue described in Yan et al., at residue 172, according to the sequence of SEQ ID NO:10 (see also FIG. 22).
-
In an embodiment the allele encodes an IDH2 having Lys at residue 172.
-
In an embodiment the allele encodes an IDH2 having Met at residue 172.
-
In an embodiment, the nucleic acid-based inhibitor is a dsRNA that targets IDH2, e.g., an IDH2 having a G or a T (or a nucleotide other than A or C) at nucleotide position 514 or an A or T or C (or a nucleotide other than G) at nucleotide position 515, e.g., a mutant allele carrying a A514G mutation or a G515T or a G515A mutation according to the IDH2 sequence of SEQ ID NO:10 (FIG. 22A). In one embodiment, the nucleic acid-based inhibitor is a dsRNA that targets IDH2, e.g., an IDH2 having a C or a T (or a nucleotide other than G or A) at nucleotide position 516 according to the IDH2 sequence of SEQ ID NO:10.
-
In an embodiment, the nucleic acid-based inhibitor is a dsRNA that targets IDH2, e.g., an IDH2 having a G at nucleotide position 514 or a T at nucleotide position 515 or an A at position 515, according to the IDH2 sequence of SEQ ID NO:10.
-
In an embodiment, the dsRNA targets an IDH2 having other than A, e.g., a G or a T, at nucleotide position 514, or other than G, e.g., an A or C or T at position 515 (e.g., a mutant), or other than G, e.g., C or T, and an IDH2 having an A at nucleotide position 514 or a G at nucleotide position 515 or a G at position 516 (e.g., a wildtype), e.g., by targeting a region of the IDH2 mRNA that is identical between the wildtype and mutant transcripts. In yet another embodiment, the dsRNA targets a particular mutant or polymorphism (such as a single nucleotide polymorphism (SNP)), but not a wildtype allele. In this case, the nucleic acid based inhibitor, e.g., a dsRNA, targets the region of the IDH2 containing the mutation.
-
In some embodiments, the nucleic acid based inhibitor, e.g., a dsRNA, preferentially or specifically inhibits the product of a mutant IDH2 as compared to the product of a wildtype IDH2. For example, in one embodiment, a dsRNA targets a region of an IDH2 mRNA that carries the mutation (e.g., an A514G or G515T or a G515U mutation according to SEQ ID NO:10).
-
In one embodiment, the nucleic acid-based inhibitor is a dsRNA including a sense strand and an antisense strand having a primary sequence presented in Tables 15-23. In another embodiment, the nucleic acid based inhibitor is an antisense oligonucleotide that includes all or a part of an antisense primary sequence presented in Tables 15-23 or which targets the same or substantially the same region as does a dsRNA from Tables 15-23.
-
In an embodiment, the nucleic acid based inhibitor is delivered to the brain, e.g., directly to the brain, e.g., by intrathecal or intraventricular delivery. The nucleic acid based inhibitor can also be delivered from an implantable device. In an embodiment, the nucleic acid-based inhibitor is delivered by infusion using, e.g., a catheter, and optionally, a pump.
-
Antisense
-
Suitable nucleic acid based inhibitors include antisense nucleic acids. While not being bound by theory it is believed that antisense inhibition is typically based upon hydrogen bonding-based hybridization of oligonucleotide strands or segments such that at least one strand or segment is cleaved, degraded, or otherwise rendered inoperable.
-
An antisense agent can bind IDH1 or IDH2 DNA. In embodiments it inhibits replication and transcription. While not being bound by theory it is believed that an antisense agent can also function to inhibit target RNA translocation, e.g., to a site of protein translation, translation of protein from the RNA, splicing of the RNA to yield one or more RNA species, and catalytic activity or complex formation involving the RNA.
-
An antisense agents can have a chemical modification described above as being suitable for dsRNA.
-
Antisense agents can include, for example, from about 8 to about 80 nucleobases (i.e., from about 8 to about 80 nucleotides), e.g., about 8 to about 50 nucleobases, or about 12 to about 30 nucleobases. Antisense compounds include ribozymes, external guide sequence (EGS) oligonucleotides (oligozymes), and other short catalytic RNAs or catalytic oligonucleotides which hybridize to the target nucleic acid and modulate its expression. Anti-sense compounds can include a stretch of at least eight consecutive nucleobases that are complementary to a sequence in the target gene. An oligonucleotide need not be 100% complementary to its target nucleic acid sequence to be specifically hybridizable. An oligonucleotide is specifically hybridizable when binding of the oligonucleotide to the target interferes with the normal function of the target molecule to cause a loss of utility, and there is a sufficient degree of complementarity to avoid non-specific binding of the oligonucleotide to non-target sequences under conditions in which specific binding is desired, i.e., under physiological conditions in the case of in vivo assays or therapeutic treatment or, in the case of in vitro assays, under conditions in which the assays are conducted.
-
Hybridization of antisense oligonucleotides with mRNA (e.g., an mRNA encoding IDH1 or IDH2) can interfere with one or more of the normal functions of mRNA. While not being bound by theory it is believed that the functions of mRNA to be interfered with include all key functions such as, for example, translocation of the RNA to the site of protein translation, translation of protein from the RNA, splicing of the RNA to yield one or more mRNA species, and catalytic activity which may be engaged in by the RNA. Binding of specific protein(s) to the RNA may also be interfered with by antisense oligonucleotide hybridization to the RNA.
-
Exemplary antisense compounds include DNA or RNA sequences that specifically hybridize to the target nucleic acid, e.g., the mRNA encoding IDH1 or IDH2. The complementary region can extend for between about 8 to about 80 nucleobases. The compounds can include one or more modified nucleobases. Modified nucleobases may include, e.g., 5-substituted pyrimidines such as 5-iodouracil, 5-iodocytosine, and C5-propynyl pyrimidines such as C5-propynylcytosine and C5-propynyluracil. Other suitable modified nucleobases include N4—(C1-C12) alkylaminocytosines and N4,N4—(C1-C12) dialkylaminocytosines. Modified nucleobases may also include 7-substituted-5-aza-7-deazapurines and 7-substituted-7-deazapurines such as, for example, 7-iodo-7-deazapurines, 7-cyano-7-deazapurines, 7-aminocarbonyl-7-deazapurines. Examples of these include 6-amino-7-iodo-7-deazapurines, 6-amino-7-cyano-7-deazapurines, 6-amino-7-aminocarbonyl-7-deazapurines, 2-amino-6-hydroxy-7-iodo-7-deazapurines, 2-amino-6-hydroxy-7-cyano-7-deazapurines, and 2-amino-6-hydroxy-7-aminocarbonyl-7-deazapurines. Furthermore, N6—(C1-C12) alkylaminopurines and N6,N6—(C1-C12) dialkylaminopurines, including N6-methylaminoadenine and N6,N6-dimethylaminoadenine, are also suitable modified nucleobases. Similarly, other 6-substituted purines including, for example, 6-thioguanine may constitute appropriate modified nucleobases. Other suitable nucleobases include 2-thiouracil, 8-bromoadenine, 8-bromoguanine, 2-fluoroadenine, and 2-fluoroguanine. Derivatives of any of the aforementioned modified nucleobases are also appropriate. Substituents of any of the preceding compounds may include C1-C30 alkyl, C2-C30 alkenyl, C2-C30 alkynyl, aryl, aralkyl, heteroaryl, halo, amino, amido, nitro, thio, sulfonyl, carboxyl, alkoxy, alkylcarbonyl, alkoxycarbonyl, and the like.
-
MicroRNA
-
In some embodiments, the nucleic acid-based inhibitor suitable for targeting IDH, e.g., IDH1, is a microRNA (miRNA). A miRNA is a single stranded RNA that regulates the expression of target mRNAs either by mRNA cleavage, translational repression/inhibition or heterochromatic silencing. The miRNA is 18 to 25 nucleotides, typically 21 to 23 nucleotides in length. In some embodiments, the miRNA includes chemical modifications, such as one or more modifications described herein.
-
In some embodiments, a nucleic acid based inhibitor targeting IDH has partial complementarity (i.e., less than 100% complementarity) with the target IDH, e.g., IDH1 or IDH2, mRNA. For example, partial complementarity can include various mismatches or non-base paired nucleotides (e.g., 1, 2, 3, 4, 5 or more mismatches or non-based paired nucleotides, such as nucleotide bulges), which can result in bulges, loops, or overhangs that result between the antisense strand or antisense region of the nucleic acid-based inhibitor and the corresponding target nucleic acid molecule.
-
The nucleic acid-based inhibitors described herein, e.g., antisense nucleic acid described herein, can be incorporated into a gene construct to be used as a part of a gene therapy protocol to deliver nucleic acids that can be used to express and produce agents within cells. Expression constructs of such components may be administered in any biologically-effective carrier, e.g., any formulation or composition capable of effectively delivering the component gene to cells in vivo. Approaches include insertion of the subject gene in viral vectors including recombinant retroviruses, adenovirus, adeno-associated virus, lentivirus, and herpes simplex virus-1, or recombinant bacterial or eukaryotic plasmids. Viral vectors transfect cells directly; plasmid DNA can be delivered with the help of, for example, cationic liposomes (lipofectin) or derivatized (e.g., antibody conjugated) polylysine conjugates, gramacidin S, artificial viral envelopes or other such intracellular earners, as well as direct injection of the gene construct or CaPO4 precipitation carried out in vivo.
-
In an embodiment, in vivo introduction of nucleic acid into a cell includes use of a viral vector containing nucleic acid, e.g., a cDNA. Infection of cells with a viral vector has the advantage that a large proportion of the targeted cells can receive the nucleic acid. Additionally, molecules encoded within the viral vector, e.g., by a cDNA contained in the viral vector, are expressed efficiently in cells which have taken up viral vector nucleic acid.
-
Retroviral vectors and adeno-associated virus vectors can be used as a recombinant gene delivery system for the transfer of exogenous genes in vivo particularly into humans. These vectors provide efficient delivery of genes into cells, and the transferred nucleic acids are stably integrated into the chromosomal DNA of the host. Protocols for producing recombinant retroviruses and for infecting cells in vitro or in vivo with such viruses can be found in Current Protocols in Molecular Biology, Ausubel, F. M. et al. (eds.) Greene Publishing Associates (1989), Sections 9.10-9.14 and other standard laboratory manuals. Examples of suitable retroviruses include pLJ, pZIP, pWE, and pEM which are known to those skilled in the art. Examples of suitable packaging virus lines for preparing both ecotropic and amphotropic retroviral systems include Crip, Cre, 2, and Am. Retroviruses have been used to introduce a variety of genes into many different cell types, including epithelial cells, in vitro and/or in vivo (see, for example, Eglitis et al. (1985) Science 230:1395-1398; Danos and Mulligan (1988) Proc. Natl. Acad. Sci. USA 85:6460-6464; Wilson et al. (1988) Proc. Natl. Acad. Sci. USA 85:3014-3018; Armentano et al. (1990) Proc. Natl. Acad. Sci. USA 87:6141-6145; Huber et al. (1991) Proc. Natl. Acad. Sci. USA 88:8039-8043; Ferry et al. (1991) Proc. Natl. Acad. Sci. USA 88:8377-8381; Chowdhury et al. (1991) Science 254:1802-1805; van Beusechem et al. (1992) Proc. Natl. Acad. Sci. USA 89:7640-7644; Kay et al. (1992) Human Gene Therapy 3:641-647; Dai et al. (1992) Proc. Natl. Acad. Sci. USA 89:10892-10895; Hwu et al. (1993) J. Immunol. 150:4104-4115; U.S. Pat. Nos. 4,868,116 and 4,980,286; PCT Pub. Nos. WO 89/07136, WO 89/02468, WO 89/05345, and WO 92/07573).
-
Another viral gene delivery system utilizes adenovirus-derived vectors. See, for example, Berkner et al. (1988) BioTechniques 6:616; Rosenfeld et al. (1991) Science 252:431-434; and Rosenfeld et al. (1992) Cell 68:143-155. Suitable adenoviral vectors derived from the adenovirus strain Ad type 5 d1324 or other strains of adenovirus (e.g., Ad2, Ad3, Ad7 etc.) are known to those skilled in the art.
-
Yet another viral vector system useful for delivery of the subject gene is the adeno-associated virus (AAV). See, for example, Flotte et al. (1992) Am. J. Respir. Cell. Mol. Biol. 7:349-356; Samulski et al. (1989) J. Virol. 63:3822-3828; and McLaughlin et al. (1989) J. Virol. 62:1963-1973.
Pharmaceutical Compositions
-
The compositions delineated herein include the compounds delineated herein, as well as additional therapeutic agents if present, in amounts effective for achieving a modulation of disease or disease symptoms, including those described herein.
-
The term “pharmaceutically acceptable carrier or adjuvant” refers to a carrier or adjuvant that may be administered to a patient, together with a compound of this invention, and which does not destroy the pharmacological activity thereof and is nontoxic when administered in doses sufficient to deliver a therapeutic amount of the compound.
-
Pharmaceutically acceptable carriers, adjuvants and vehicles that may be used in the pharmaceutical compositions of this invention include, but are not limited to, ion exchangers, alumina, aluminum stearate, lecithin, self-emulsifying drug delivery systems (SEDDS) such as d-α-tocopherol polyethyleneglycol 1000 succinate, surfactants used in pharmaceutical dosage forms such as Tweens or other similar polymeric delivery matrices, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene-polyoxypropylene-block polymers, polyethylene glycol and wool fat. Cyclodextrins such as α-, β-, and γ-cyclodextrin, or chemically modified derivatives such as hydroxyalkylcyclodextrins, including 2- and 3-hydroxypropyl-β-cyclodextrins, or other solubilized derivatives may also be advantageously used to enhance delivery of compounds of the formulae described herein.
-
The pharmaceutical compositions containing inhibitors of IDH, e.g., IDH1, may be administered directly to the central nervous system, such as into the cerebrospinal fluid or into the brain. Delivery can be, for example, in a bolus or by continuous pump infusion. In certain embodiments, delivery is by intrathecal delivery or by intraventricular injection directly into the brain. A catheter and, optionally, a pump can be used for delivery. The inhibitors can be delivered in and released from an implantable device, e.g., a device that is implanted in association with surgical removal of tumor tissue. E.g., for delivery to the brain, the delivery can be analogous to that with Gliadel, a biopolymer wafer designed to deliver carmustine directly into the surgical cavity created when a brain tumor is resected. The Gliadel wafer slowly dissolves and delivers carmustine.
-
The therapeutics disclosed herein, e.g., nucleic acid based inhibitors, e.g. siRNAs can be administered directly to the CNS, e.g., the brain, e.g., using a pump and/or catheter system. In one embodiment, the pump is implanted under the skin. In an embodiment and a catheter attached to a pump is inserted into the CNS, e.g., into the brain or spine. In one embodiment, the pump (such as the IsoMed Drug Pump from Medtronic) delivers dosing, e.g, constant dosing, of a nucleic acid based inhibitor. In an embodiment, the pump is programmable to administer variable or constant doses at predetermined time intervals. For example, the IsoMed Drug pump from Medtronic (or a similar device) can be used to administer a constant supply of the inhibitor, or the SynchroMedII Drug Pump (or a similar device) can be used to administer a variable dosing regime.
-
Methods and devices described in U.S. Pat. Nos. 7,044,932, 6,620,151, 6,283,949, and 6,685,452 can be used in methods described herein.
-
The pharmaceutical compositions of this invention may be administered orally, parenterally, by inhalation, topically, rectally, nasally, buccally, vaginally or via an implanted reservoir, preferably by oral administration or administration by injection. The pharmaceutical compositions of this invention may contain any conventional non-toxic pharmaceutically-acceptable carriers, adjuvants or vehicles. In some cases, the pH of the formulation may be adjusted with pharmaceutically acceptable acids, bases or buffers to enhance the stability of the formulated compound or its delivery form. The term parenteral as used herein includes subcutaneous, intracutaneous, intravenous, intramuscular, intraarticular, intraarterial, intrasynovial, intrasternal, intrathecal, intralesional and intracranial injection or infusion techniques.
-
The pharmaceutical compositions may be in the form of a sterile injectable preparation, for example, as a sterile injectable aqueous or oleaginous suspension. This suspension may be formulated according to techniques known in the art using suitable dispersing or wetting agents (such as, for example, Tween 80) and suspending agents. The sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally acceptable diluent or solvent, for example, as a solution in 1,3-butanediol. Among the acceptable vehicles and solvents that may be employed are mannitol, water, Ringer's solution and isotonic sodium chloride solution. In addition, sterile, fixed oils are conventionally employed as a solvent or suspending medium. For this purpose, any bland fixed oil may be employed including synthetic mono- or diglycerides. Fatty acids, such as oleic acid and its glyceride derivatives are useful in the preparation of injectables, as are natural pharmaceutically-acceptable oils, such as olive oil or castor oil, especially in their polyoxyethylated versions. These oil solutions or suspensions may also contain a long-chain alcohol diluent or dispersant, or carboxymethyl cellulose or similar dispersing agents which are commonly used in the formulation of pharmaceutically acceptable dosage forms such as emulsions and or suspensions. Other commonly used surfactants such as Tweens or Spans and/or other similar emulsifying agents or bioavailability enhancers which are commonly used in the manufacture of pharmaceutically acceptable solid, liquid, or other dosage forms may also be used for the purposes of formulation.
-
The pharmaceutical compositions of this invention may be orally administered in any orally acceptable dosage form including, but not limited to, capsules, tablets, emulsions and aqueous suspensions, dispersions and solutions. In the case of tablets for oral use, carriers which are commonly used include lactose and corn starch. Lubricating agents, such as magnesium stearate, are also typically added. For oral administration in a capsule form, useful diluents include lactose and dried corn starch. When aqueous suspensions and/or emulsions are administered orally, the active ingredient may be suspended or dissolved in an oily phase is combined with emulsifying and/or suspending agents. If desired, certain sweetening and/or flavoring and/or coloring agents may be added.
-
The pharmaceutical compositions of this invention may also be administered in the form of suppositories for rectal administration. These compositions can be prepared by mixing a compound of this invention with a suitable non-irritating excipient which is solid at room temperature but liquid at the rectal temperature and therefore will melt in the rectum to release the active components. Such materials include, but are not limited to, cocoa butter, beeswax and polyethylene glycols.
-
Topical administration of the pharmaceutical compositions of this invention is useful when the desired treatment involves areas or organs readily accessible by topical application. For application topically to the skin, the pharmaceutical composition should be formulated with a suitable ointment containing the active components suspended or dissolved in a carrier. Carriers for topical administration of the compounds of this invention include, but are not limited to, mineral oil, liquid petroleum, white petroleum, propylene glycol, polyoxyethylene polyoxypropylene compound, emulsifying wax and water. Alternatively, the pharmaceutical composition can be formulated with a suitable lotion or cream containing the active compound suspended or dissolved in a carrier with suitable emulsifying agents. Suitable carriers include, but are not limited to, mineral oil, sorbitan monostearate, polysorbate 60, cetyl esters wax, cetearyl alcohol, 2-octyldodecanol, benzyl alcohol and water. The pharmaceutical compositions of this invention may also be topically applied to the lower intestinal tract by rectal suppository formulation or in a suitable enema formulation. Topically-transdermal patches are also included in this invention.
-
The pharmaceutical compositions of this invention may be administered by nasal aerosol or inhalation. Such compositions are prepared according to techniques well-known in the art of pharmaceutical formulation and may be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other solubilizing or dispersing agents known in the art.
-
When the compositions of this invention comprise a combination of a compound of the formulae described herein and one or more additional therapeutic or prophylactic agents, both the compound and the additional agent should be present at dosage levels of between about 1 to 100%, and more preferably between about 5 to 95% of the dosage normally administered in a monotherapy regimen. The additional agents may be administered separately, as part of a multiple dose regimen, from the compounds of this invention. Alternatively, those agents may be part of a single dosage form, mixed together with the compounds of this invention in a single composition.
-
The compounds described herein can, for example, be administered by injection, intravenously, intraarterially, subdermally, intraperitoneally, intramuscularly, or subcutaneously; or orally, buccally, nasally, transmucosally, topically, in an ophthalmic preparation, or by inhalation, with a dosage ranging from about 0.02 to about 100 mg/kg of body weight, alternatively dosages between 1 mg and 1000 mg/dose, every 4 to 120 hours, or according to the requirements of the particular drug. The methods herein contemplate administration of an effective amount of compound or compound composition to achieve the desired or stated effect. Typically, the pharmaceutical compositions of this invention will be administered from about 1 to about 6 times per day or alternatively, as a continuous infusion. Such administration can be used as a chronic or acute therapy. The amount of active ingredient that may be combined with the carrier materials to produce a single dosage form will vary depending upon the host treated and the particular mode of administration. A typical preparation will contain from about 5% to about 95% active compound (w/w). Alternatively, such preparations contain from about 20% to about 80% active compound.
-
Lower or higher doses than those recited above may be required. Specific dosage and treatment regimens for any particular patient will depend upon a variety of factors, including the activity of the specific compound employed, the age, body weight, general health status, sex, diet, time of administration, rate of excretion, drug combination, the severity and course of the disease, condition or symptoms, the patient's disposition to the disease, condition or symptoms, and the judgment of the treating physician.
-
Upon improvement of a patient's condition, a maintenance dose of a compound, composition or combination of this invention may be administered, if necessary. Subsequently, the dosage or frequency of administration, or both, may be reduced, as a function of the symptoms, to a level at which the improved condition is retained when the symptoms have been alleviated to the desired level. Patients may, however, require intermittent treatment on a long-term basis upon any recurrence of disease symptoms.
Kits
-
A compound described herein can be provided in a kit.
-
In an embodiment the kit includes (a) a compound described herein, e.g., a composition that includes a compound described herein (wherein, e.g., the compound can be an inhibitor described herein), and, optionally (b) informational material. The informational material can be descriptive, instructional, marketing or other material that relates to the methods described herein and/or the use of a compound described herein for the methods described herein.
-
In an embodiment the kit provides materials for evaluating a subject. The evaluation can be, e.g., for: identifying a subject having unwanted levels (e.g., higher than present in normal or wildtype cells) of any of 2HG, 2HG neoactivity, or mutant IDH1 or IDH2 protein having 2HG neoactivity (or corresponding RNA), or having a somatic mutation in IDH1 or IDH2 characterized by 2HG neoactivity; diagnosing, prognosing, or staging, a subject, e.g., on the basis of having increased levels of 2HG, 2HG neoactivity, or mutant IDH1 or IDH2 protein having 2HG neoactivity (or corresponding RNA), or having a somatic mutation in IDH1 or IDH2 characterized by 2HG neoactivity; selecting a treatment for, or evaluating the efficacy of, a treatment, e.g., on the basis of the subject having increased levels of 2HG, 2HG neoactivity, or mutant IDH1 or IDH2 protein having 2HG neoactivity (or corresponding RNA), or having a somatic mutation in IDH1 or IDH2 characterized by 2HG neoactivity. The kit can include one or more reagent useful in the evaluation, e.g., reagents mentioned elsewhere herein. A detection reagent, e.g., an antibody or other specific binding reagent can be included. Standards or reference samples, e.g., a positive or negative control standard can be included. E.g., if the evaluation is based on the presence of 2HG the kit can include a reagent, e.g, a positive or negative control standards for an assay, e.g., a LC-MS assay.
-
If the evaluation is based on the presence of 2HG neoactivity, the kit can include a reagent, e.g., one or more of those mentioned elsewhere herein, for assaying 2HG neoactivity. If the evaluation is based on sequencing, the kit can include primers or other materials useful for sequencing the relevant nucleic acids for identifying an IHD, e.g., IDH1 or IDH2, neoactive mutant. E.g., the kit can contain a reagent that provides for interrogation of the identity, i.e., sequencing of, residue 132 of IDH1 to determine if a neoactive mutant is present. The kit can include nucleic acids, e.g., an oligomer, e.g., primers, which allow sequencing of the nucleotides that encode residue 132 of IDH1. In an embodiment the kit includes a nucleic acid whose hybridization, or ability to be amplified, is dependent on the identity of residue 132 of IDH1. In other embodiments the kit includes a reagent, e.g., an antibody or other specific binding molecule that can identify the presence of a neoactive mutant, e.g., a protein encoded by a neoactive mutant at 132 of IDH1. As described below, a kit can also include buffers, solvents, and information related to the evaluation.
-
In one embodiment, the informational material can include information about production of the compound, molecular weight of the compound, concentration, date of expiration, batch or production site information, and so forth. In one embodiment, the informational material relates to methods for administering the compound.
-
In one embodiment, the informational material can include instructions to administer a compound described herein in a suitable manner to perform the methods described herein, e.g., in a suitable dose, dosage form, or mode of administration (e.g., a dose, dosage form, or mode of administration described herein). In another embodiment, the informational material can include instructions to administer a compound described herein to a suitable subject, e.g., a human, e.g., a human having or at risk for a disorder described herein.
-
The informational material of the kits is not limited in its form. In many cases, the informational material, e.g., instructions, is provided in printed matter, e.g., a printed text, drawing, and/or photograph, e.g., a label or printed sheet. However, the informational material can also be provided in other formats, such as Braille, computer readable material, video recording, or audio recording. In another embodiment, the informational material of the kit is contact information, e.g., a physical address, email address, website, or telephone number, where a user of the kit can obtain substantive information about a compound described herein and/or its use in the methods described herein. Of course, the informational material can also be provided in any combination of formats.
-
In addition to a compound described herein, the composition of the kit can include other ingredients, such as a solvent or buffer, a stabilizer, a preservative, a flavoring agent (e.g., a bitter antagonist or a sweetener), a fragrance or other cosmetic ingredient, and/or a second agent for treating a condition or disorder described herein. Alternatively, the other ingredients can be included in the kit, but in different compositions or containers than a compound described herein. In such embodiments, the kit can include instructions for admixing a compound described herein and the other ingredients, or for using a compound described herein together with the other ingredients.
-
A compound described herein can be provided in any form, e.g., liquid, dried or lyophilized form. It is preferred that a compound described herein be substantially pure and/or sterile. When a compound described herein is provided in a liquid solution, the liquid solution preferably is an aqueous solution, with a sterile aqueous solution being preferred. When a compound described herein is provided as a dried form, reconstitution generally is by the addition of a suitable solvent. The solvent, e.g., sterile water or buffer, can optionally be provided in the kit.
-
The kit can include one or more containers for the composition containing a compound described herein. In some embodiments, the kit contains separate containers, dividers or compartments for the composition and informational material. For example, the composition can be contained in a bottle, vial, or syringe, and the informational material can be contained in a plastic sleeve or packet. In other embodiments, the separate elements of the kit are contained within a single, undivided container. For example, the composition is contained in a bottle, vial or syringe that has attached thereto the informational material in the form of a label. In some embodiments, the kit includes a plurality (e.g., a pack) of individual containers, each containing one or more unit dosage forms (e.g., a dosage form described herein) of a compound described herein. For example, the kit includes a plurality of syringes, ampules, foil packets, or blister packs, each containing a single unit dose of a compound described herein. The containers of the kits can be air tight, waterproof (e.g., impermeable to changes in moisture or evaporation), and/or light-tight.
-
The kit optionally includes a device suitable for administration of the composition, e.g., a syringe, inhalant, pipette, forceps, measured spoon, dropper (e.g., eye dropper), swab (e.g., a cotton swab or wooden swab), or any such delivery device. In an embodiment, the device is a medical implant device, e.g., packaged for surgical insertion.
Combination Therapies
-
In some embodiments, a compound or composition described herein, is administered together with an additional cancer treatment. Exemplary cancer treatments include, for example: surgery, chemotherapy, targeted therapies such as antibody therapies, immunotherapy, and hormonal therapy. Examples of each of these treatments are provided below.
Chemotherapy
-
In some embodiments, a compound or composition described herein, is administered with a chemotherapy. Chemotherapy is the treatment of cancer with drugs that can destroy cancer cells. “Chemotherapy” usually refers to cytotoxic drugs which affect rapidly dividing cells in general, in contrast with targeted therapy. Chemotherapy drugs interfere with cell division in various possible ways, e.g., with the duplication of DNA or the separation of newly formed chromosomes. Most forms of chemotherapy target all rapidly dividing cells and are not specific for cancer cells, although some degree of specificity may come from the inability of many cancer cells to repair DNA damage, while normal cells generally can.
-
Examples of chemotherapeutic agents used in cancer therapy include, for example, antimetabolites (e.g., folic acid, purine, and pyrimidine derivatives) and alkylating agents (e.g., nitrogen mustards, nitrosoureas, platinum, alkyl sulfonates, hydrazines, triazenes, aziridines, spindle poison, cytotoxic agents, toposimerase inhibitors and others). Exemplary agents include Aclarubicin, Actinomycin, Alitretinon, Altretamine, Aminopterin, Aminolevulinic acid, Amrubicin, Amsacrine, Anagrelide, Arsenic trioxide, Asparaginase, Atrasentan, Belotecan, Bexarotene, endamustine, Bleomycin, Bortezomib, Busulfan, Camptothecin, Capecitabine, Carboplatin, Carboquone, Carmofur, Carmustine, Celecoxib, Chlorambucil, Chlonnethine, Cisplatin, Cladribine, Clofarabine, Crisantaspase, Cyclophosphamide, Cytarabine, Dacarbazine, Dactinomycin, Daunorubicin, Decitabine, Demecolcine, Docetaxel, Doxorubicin, Efaproxiral, Elesclomol, Elsamitrucin, Enocitabine, Epirubicin, Estramustine, Etoglucid, Etoposide, Floxuridine, Fludarabine, Fluorouracil (5FU), Fotemustine, Gemcitabine, Gliadel implants, Hydroxycarbamide, Hydroxyurea, Idarubicin, Ifosfamide, Irinotecan, Irofulven, Ixabepilone, Larotaxel, Leucovorin, Liposomal doxorubicin, Liposomal daunorubicin, Lonidamine, Lomustine, Lucanthone, Mannosulfan, Masoprocol, Melphalan, Mercaptopurine, Mesna, Methotrexate, Methyl aminolevulinate, Mitobronitol, Mitoguazone, Mitotane, Mitomycin, Mitoxantrone, Nedaplatin, Nimustine, Oblimersen, Omacetaxine, Ortataxel, Oxaliplatin, Paclitaxel, Pegaspargase, Pemetrexed, Pentostatin, Pirarubicin, Pixantrone, Plicamycin, Porfimer sodium, Prednimustine, Procarbazine, Raltitrexed, Ranimustine, Rubitecan, Sapacitabine, Semustine, Sitimagene ceradenovec, Strataplatin, Streptozocin, Talaporfin, Tegafur-uracil, Temoporfin, Temozolomide, Teniposide, Tesetaxel, Testolactone, Tetranitrate, Thiotepa, Tiazofurine, Tioguanine, Tipifarnib, Topotecan, Trabectedin, Triaziquone, Triethylenemelamine, Triplatin, Tretinoin, Treosulfan, Trofosfamide, Uramustine, Valrubicin, Verteporfin, Vinblastine, Vincristine, Vindesine, Vinflunine, Vinorelbine, Vorinostat, Zorubicin, and other cytostatic or cytotoxic agents described herein.
-
Because some drugs work better together than alone, two or more drugs are often given at the same time. Often, two or more chemotherapy agents are used as combination chemotherapy. In some embodiments, the chemotherapy agents (including combination chemotherapy) can be used in combination with a compound described herein, e.g., phenformin.
-
Targeted Therapy
-
In some embodiments, a compound or composition described herein, is administered with a targeted therapy. Targeted therapy constitutes the use of agents specific for the deregulated proteins of cancer cells. Small molecule targeted therapy drugs are generally inhibitors of enzymatic domains on mutated, overexpressed, or otherwise critical proteins within the cancer cell. Prominent examples are the tyrosine kinase inhibitors such as Axitinib, Bosutinib, Cediranib, desatinib, erlotinib, imatinib, gefitinib, lapatinib, Lestaurtinib, Nilotinib, Semaxanib, Sorafenib, Sunitinib, and Vandetanib, and also cyclin-dependent kinase inhibitors such as Alvocidib and Seliciclib. Monoclonal antibody therapy is another strategy in which the therapeutic agent is an antibody which specifically binds to a protein on the surface of the cancer cells. Examples include the anti-HER2/neu antibody trastuzumab (HERCEPTIN®) typically used in breast cancer, and the anti-CD20 antibody rituximab and Tositumomab typically used in a variety of B-cell malignancies. Other exemplary antibodies include Cetuximab, Panitumumab, Trastuzumab, Alemtuzumab, Bevacizumab, Edrecolomab, and Gemtuzumab. Exemplary fusion proteins include Aflibercept and Denileukin diftitox. In some embodiments, the targeted therapy can be used in combination with a compound described herein, e.g., a biguanide such as metformin or phenformin, preferably phenformin.
-
Targeted therapy can also involve small peptides as “homing devices” which can bind to cell surface receptors or affected extracellular matrix surrounding the tumor. Radionuclides which are attached to these peptides (e.g., RGDs) eventually kill the cancer cell if the nuclide decays in the vicinity of the cell. An example of such therapy includes BEXXAR®.
-
Immunotherapy
-
In some embodiments, a compound or composition described herein, is administered with an immunotherapy. Cancer immunotherapy refers to a diverse set of therapeutic strategies designed to induce the patient's own immune system to fight the tumor. Contemporary methods for generating an immune response against tumors include intravesicular BCG immunotherapy for superficial bladder cancer, and use of interferons and other cytokines to induce an immune response in renal cell carcinoma and melanoma patients.
-
Allogeneic hematopoietic stem cell transplantation can be considered a form of immunotherapy, since the donor's immune cells will often attack the tumor in a graft-versus-tumor effect. In some embodiments, the immunotherapy agents can be used in combination with a compound or composition described herein.
-
Hormonal Therapy
-
In some embodiments, a compound or composition described herein, is administered with a hormonal therapy. The growth of some cancers can be inhibited by providing or blocking certain hormones. Common examples of hormone-sensitive tumors include certain types of breast and prostate cancers. Removing or blocking estrogen or testosterone is often an important additional treatment. In certain cancers, administration of hormone agonists, such as progestogens may be therapeutically beneficial. In some embodiments, the hormonal therapy agents can be used in combination with a compound or a composition described herein.
-
In some embodiments, a compound or composition described herein, is administered together with an additional cancer treatment (e.g., surgical removal), in treating cancer in nervous system, e.g., cancer in central nervous system, e.g., brain tumor, e.g., glioma, e.g., glioblastoma multiforme (GBM).
-
Several studies have suggested that more than 25% of glioblastoma patients obtain a significant survival benefit from adjuvant chemotherapy. Meta-analyses have suggested that adjuvant chemotherapy results in a 6-10% increase in 1-year survival rate.
-
Temozolomide is an orally active alkylating agent that is used for persons newly diagnosed with glioblastoma multiforme. It was approved by the United States Food and Drug Administration (FDA) in March 2005. Studies have shown that the drug was well tolerated and provided a survival benefit. Adjuvant and concomitant temozolomide with radiation was associated with significant improvements in median progression-free survival over radiation alone (6.9 vs 5 mo), overall survival (14.6 vs 12.1 mo), and the likelihood of being alive in 2 years (26% vs 10%).
-
Nitrosoureas: BCNU (carmustine)-polymer wafers (Gliadel) were approved by the FDA in 2002. Though Gliadel wafers are used by some for initial treatment, they have shown only a modest increase in median survival over placebo (13.8 vs. 11.6 months) in the largest such phase III trial, and are associated with increased rates of CSF leak and increased intracranial pressure secondary to edema and mass effect.
-
MGMT is a DNA repair enzyme that contributes to temozolomide resistance. Methylation of the MGMT promoter, found in approximately 45% of glioblastoma multiformes, results in an epigenetic silencing of the gene, decreasing the tumor cell's capacity for DNA repair and increasing susceptibility to temozolomide.
-
When patients with and without MGMT promoter methylation were treated with temozolomide, the groups had median survivals of 21.7 versus 12.7 months, and 2-year survival rates of 46% versus 13.8%, respectively.
-
Though temozolomide is currently a first-line agent in the treatment of glioblastoma multiforme, unfavorable MGMT methylation status could help select patients appropriate for future therapeutic investigations.
-
O6-benzylguanine and other inhibitors of MGMT as well as RNA interference-mediated silencing of MGMT offer promising avenues to increase the effectiveness of temozolomide and other alkylating antineoplastics, and such agents are under active study.
-
Carmustine (BCNU) and cis-platinum (cisplatin) have been the primary chemotherapeutic agents used against malignant gliomas. All agents in use have no greater than a 30-40% response rate, and most fall into the range of 10-20%.
-
Data from the University of California at San Francisco indicate that, for the treatment of glioblastomas, surgery followed by radiation therapy leads to 1-, 3-, and 5-year survival rates of 44%, 6%, and 0%, respectively. By comparison, surgery followed by radiation and chemotherapy using nitrosourea-based regimens resulted in 1-, 3-, and 5-year survival rates of 46%, 18%, and 18%, respectively.
-
A major hindrance to the use of chemotherapeutic agents for brain tumors is the fact that the blood-brain barrier (BBB) effectively excludes many agents from the CNS. For this reason, novel methods of intracranial drug delivery are being developed to deliver higher concentrations of chemotherapeutic agents to the tumor cells while avoiding the adverse systemic effects of these medications.
-
Pressure-driven infusion of chemotherapeutic agents through an intracranial catheter, also known as convection-enhanced delivery (CED), has the advantage of delivering drugs along a pressure gradient rather than by simple diffusion. CED has shown promising results in animal models with agents including BCNU and topotecan.
-
Initial attempts investigated the delivery of chemotherapeutic agents via an intraarterial route rather than intravenously. Unfortunately, no survival advantage was observed.
-
Chemotherapy for recurrent glioblastoma multiforme provides modest, if any, benefit, and several classes of agents are used. Carmustine wafers increased 6-month survival from 36% to 56% over placebo in one randomized study of 222 patients, though there was a significant association between the treatment group and serious intracranial infections.
-
Genotyping of brain tumors may have applications in stratifying patients for clinical trials of various novel therapies.
-
The anti-angiogenic agent bevacizumab, when used with irinotecan improved 6-month survival in recurrent glioma patients to 46% compared with 21% in patients treated with temozolomide. This bevacizumab and irinotecan combination for recurrent glioblastoma multiforme has been shown to improve survival over bevacizumab alone. Anti-angiogenic agents also decrease peritumoral edema, potentially reducing the necessary corticosteroid dose.
-
Some glioblastomas responds to gefitinib or erlotinib (tyrosine kinase inhibitors). The simultaneous presence in glioblastoma cells of mutant EGFR (EGFRviii) and PTEN was associated with responsiveness to tyrosine kinase inhibitors, whereas increased p-akt predicts a decreased effect. Other targets include PDGFR, VEGFR, mTOR, farnesyltransferase, and PI3K.
-
Other possible therapy modalities include imatinib, gene therapy, peptide and dendritic cell vaccines, synthetic chlorotoxins, and radiolabeled drugs and antibodies.
Patient Selection/Monitoring
-
Described herein are methods of treating a cell proliferation-related disorder, e.g., cancer, in a subject and methods of identifying a subject for a treatment described herein. Also described herein are methods of predicting a subject who is at risk of developing cancer (e.g., a cancer associate with a mutation in an enzyme (e.g., an enzyme in the metabolic pathway such as IDH1 and/or IDH2)). The cancer is generally characterized by the presence of a neoactivity, such as a gain of function in one or more mutant enzymes (e.g., an enzyme in the metabolic pathway leading to fatty acid biosynthesis, glycolysis, glutaminolysis, the pentose phosphate shunt, the nucleotide biosynthetic pathway, or the fatty acid biosynthetic pathway, e.g., IDH1 or IDH2). The subject can be selected on the basis of the subject having a mutant gene having a neoactivity, e.g., a neoactivity described herein. As used herein, “select” means selecting in whole or part on said basis.
-
In some embodiments, a subject is selected for treatment with a compound described herein based on a determination that the subject has a mutant enzyme described herein (e.g., an enzyme in the metabolic pathway, e.g., a metabolic pathway leading to fatty acid biosynthesis, glycolysis, glutaminolysis, the pentose phosphate shunt, the nucleotide biosynthetic pathway, or the fatty acid biosynthetic pathway, e.g., IDH1 or IDH2). In some embodiments, the mutant enzyme has a neoactivity and the patient is selected on that basis. The neoactivity of the enzyme can be identified, for example, by evaluating the subject or sample (e.g., tissue or bodily fluid) therefrom, for the presence or amount of a substrate, cofactor and/or product of the enzyme. The presence and/or amount of substrate, cofactor and/or product can correspond to the wild-type/non-mutant activity or can correspond to the neoactivity of the enzyme. Exemplary bodily fluid that can be used to identify (e.g., evaluate) the neoactivity of the enzyme include amniotic fluid surrounding a fetus, aqueous humour, blood (e.g., blood plasma), Cerebrospinal fluid, cerumen, chyme, Cowper's fluid, female ejaculate, interstitial fluid, lymph, breast milk, mucus (e.g., nasal drainage or phlegm), pleural fluid, pus, saliva, sebum, semen, serum, sweat, tears, urine, vaginal secretion, or vomit.
-
In some embodiments, a subject can be evaluated for neoactivity of an enzyme using magnetic resonance. For example, where the mutant enzyme is IDH1 or IDH2 and the neoactivity is conversion of α-ketoglutarate to 2-hydroxyglutarate, the subject can be evaluated for the presence of and/or an elevated amount of 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate relative to the amount of 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate present in a subject who does not have a mutation in IDH1 or IDH2 having the above neoactivity. In some embodiments, neoactivity of IDH1 or IDH2 can be determined by the presence or elevated amount of a peak corresponding to 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate as determined by magnetic resonance. For example, a subject can be evaluated for the presence and/or strength of a signal at about 2.5 ppm to determine the presence and/or amount of 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate in the subject. This can be correlated to and/or predictive of a neoactivity described herein for the mutant enzyme IDH. Similarly, the presence, strength and/or absence of a signal at about 2.5 ppm could be predictive of a response to treatment and thereby used as a noninvasive biomarker for clinical response.
-
Neoactivity of a mutant enzyme such as IDH can also be evaluated using other techniques known to one skilled in the art. For example, the presence or amount of a labeled substrate, cofactor, and/or reaction product can be measured such as a 13C or 14C labeled substrate, cofactor, and/or reaction product. The neoactivity can be evaluated by evaluating the forward reaction of the wild-type/non mutant enzyme (such as the oxidative decarboxylation of ioscitrate to α-ketoglutarate in a mutant IDH1 or IDH2 enzyme, specifically a mutant IDH1 enzyme) and/or the reaction corresponding to the neoactivity (e.g., the conversion of α-ketoglutarate to 2-hydroxyglutarate, e.g., R-2-hydroxyglutarate in a mutant IDH1 or IDH2 enzyme, specifically a mutant IDH1 enzyme).
Disorders
-
The IDH-related methods disclosed herein, e.g., methods of evaluating or treating subjects, are directed to subjects having a cell proliferation-related disorder characterized by an IDH mutant, e.g., an IDH1 or IDH2, mutant having neoactivity, e.g., 2HG neoactivity. Examples of some of the disorders below have been shown to be characterized by an IDH1 or IDH2 mutation. Others can be analyzed, e.g., by sequencing cell samples to determine the presence of a somatic mutation at amino acid 132 of IDH1 or at amino acid 172 of IDH2. Without being bound by theory it is expected that a portion of the tumors of given type of cancer will have an IDH, e.g., IDH1 or IDH2, mutant having 2HG neoactivity.
-
The disclosed methods are useful in evaluating or treating proliferative disorders, e.g. evaluating or treating solid tumors, soft tissue tumors, and metastases thereof wherein the solid tumor, soft tissue tumor or metastases thereof is a cancer described herein. Exemplary solid tumors include malignancies (e.g., sarcomas, adenocarcinomas, and carcinomas) of the various organ systems, such as those of brain, lung, breast, lymphoid, gastrointestinal (e.g., colon), and genitourinary (e.g., renal, urothelial, or testicular tumors) tracts, pharynx, prostate, and ovary. Exemplary adenocarcinomas include colorectal cancers, renal-cell carcinoma, liver cancer, non-small cell carcinoma of the lung, and cancer of the small intestine. The disclosed methods are also useful in evaluating or treating non-solid cancers.
-
The methods described herein can be used with any cancer, for example those described by the National Cancer Institute. A cancer can be evaluated to determine whether it is using a method described herein. Exemplary cancers described by the National Cancer Institute include: Acute Lymphoblastic Leukemia, Adult; Acute Lymphoblastic Leukemia, Childhood; Acute Myeloid Leukemia, Adult; Adrenocortical Carcinoma; Adrenocortical Carcinoma, Childhood; AIDS-Related Lymphoma; AIDS-Related Malignancies; Anal Cancer; Astrocytoma, Childhood Cerebellar; Astrocytoma, Childhood Cerebral; Bile Duct Cancer, Extrahepatic; Bladder Cancer; Bladder Cancer, Childhood; Bone Cancer, Osteosarcoma/Malignant Fibrous Histiocytoma; Brain Stem Glioma, Childhood; Brain Tumor, Adult; Brain Tumor, Brain Stem Glioma, Childhood; Brain Tumor, Cerebellar Astrocytoma, Childhood; Brain Tumor, Cerebral Astrocytoma/Malignant Glioma, Childhood; Brain Tumor, Ependymoma, Childhood; Brain Tumor, Medulloblastoma, Childhood; Brain Tumor, Supratentorial Primitive Neuroectodermal Tumors, Childhood; Brain Tumor, Visual Pathway and Hypothalamic Glioma, Childhood; Brain Tumor, Childhood (Other); Breast Cancer; Breast Cancer and Pregnancy; Breast Cancer, Childhood; Breast Cancer, Male; Bronchial Adenomas/Carcinoids, Childhood; Carcinoid Tumor, Childhood; Carcinoid Tumor, Gastrointestinal; Carcinoma, Adrenocortical; Carcinoma, Islet Cell; Carcinoma of Unknown Primary; Central Nervous System Lymphoma, Primary; Cerebellar Astrocytoma, Childhood; Cerebral Astrocytoma/Malignant Glioma, Childhood; Cervical Cancer; Childhood Cancers; Chronic Lymphocytic Leukemia; Chronic Myelogenous Leukemia; Chronic Myeloproliferative Disorders; Clear Cell Sarcoma of Tendon Sheaths; Colon Cancer; Colorectal Cancer, Childhood; Cutaneous T-Cell Lymphoma; Endometrial Cancer; Ependymoma, Childhood; Epithelial Cancer, Ovarian; Esophageal Cancer; Esophageal Cancer, Childhood; Ewing's Family of Tumors; Extracranial Germ Cell Tumor, Childhood; Extragonadal Germ Cell Tumor; Extrahepatic Bile Duct Cancer; Eye Cancer, Intraocular Melanoma; Eye Cancer, Retinoblastoma; Gallbladder Cancer; Gastric (Stomach) Cancer; Gastric (Stomach) Cancer, Childhood; Gastrointestinal Carcinoid Tumor; Germ Cell Tumor, Extracranial, Childhood; Germ Cell Tumor, Extragonadal; Germ Cell Tumor, Ovarian; Gestational Trophoblastic Tumor; Glioma, Childhood Brain Stem; Glioma, Childhood Visual Pathway and Hypothalamic; Hairy Cell Leukemia; Head and Neck Cancer; Hepatocellular (Liver) Cancer, Adult (Primary); Hepatocellular (Liver) Cancer, Childhood (Primary); Hodgkin's Lymphoma, Adult; Hodgkin's Lymphoma, Childhood; Hodgkin's Lymphoma During Pregnancy; Hypopharyngeal Cancer; Hypothalamic and Visual Pathway Glioma, Childhood; Intraocular Melanoma; Islet Cell Carcinoma (Endocrine Pancreas); Kaposi's Sarcoma; Kidney Cancer; Laryngeal Cancer; Laryngeal Cancer, Childhood; Leukemia, Acute Lymphoblastic, Adult; Leukemia, Acute Lymphoblastic, Childhood; Leukemia, Acute Myeloid, Adult; Leukemia, Acute Myeloid, Childhood; Leukemia, Chronic Lymphocytic; Leukemia, Chronic Myelogenous; Leukemia, Hairy Cell; Lip and Oral Cavity Cancer; Liver Cancer, Adult (Primary); Liver Cancer, Childhood (Primary); Lung Cancer, Non-Small Cell; Lung Cancer, Small Cell; Lymphoblastic Leukemia, Adult Acute; Lymphoblastic Leukemia, Childhood Acute; Lymphocytic Leukemia, Chronic; Lymphoma, AIDS-Related; Lymphoma, Central Nervous System (Primary); Lymphoma, Cutaneous T-Cell; Lymphoma, Hodgkin's, Adult; Lymphoma, Hodgkin's, Childhood; Lymphoma, Hodgkin's During Pregnancy; Lymphoma, Non-Hodgkin's, Adult; Lymphoma, Non-Hodgkin's, Childhood; Lymphoma, Non-Hodgkin's During Pregnancy; Lymphoma, Primary Central Nervous System; Macroglobulinemia, Waldenstrom's; Male Breast Cancer; Malignant Mesothelioma, Adult; Malignant Mesothelioma, Childhood; Malignant Thymoma; Medulloblastoma, Childhood; Melanoma; Melanoma, Intraocular; Merkel Cell Carcinoma; Mesothelioma, Malignant; Metastatic Squamous Neck Cancer with Occult Primary; Multiple Endocrine Neoplasia Syndrome, Childhood; Multiple Myeloma/Plasma Cell Neoplasm; Mycosis Fungoides; Myelodysplastic Syndromes; Myelogenous Leukemia, Chronic; Myeloid Leukemia, Childhood Acute; Myeloma, Multiple; Myeloproliferative Disorders, Chronic; Nasal Cavity and Paranasal Sinus Cancer; Nasopharyngeal Cancer; Nasopharyngeal Cancer, Childhood; Neuroblastoma; Non-Hodgkin's Lymphoma, Adult; Non-Hodgkin's Lymphoma, Childhood; Non-Hodgkin's Lymphoma During Pregnancy; Non-Small Cell Lung Cancer; Oral Cancer, Childhood; Oral Cavity and Lip Cancer; Oropharyngeal Cancer; Osteosarcoma/Malignant Fibrous Histiocytoma of Bone; Ovarian Cancer, Childhood; Ovarian Epithelial Cancer; Ovarian Germ Cell Tumor; Ovarian Low Malignant Potential Tumor; Pancreatic Cancer; Pancreatic Cancer, Childhood; Pancreatic Cancer, Islet Cell; Paranasal Sinus and Nasal Cavity Cancer; Parathyroid Cancer; Penile Cancer; Pheochromocytoma; Pineal and Supratentorial Primitive Neuroectodermal Tumors, Childhood; Pituitary Tumor; Plasma Cell Neoplasm/Multiple Myeloma; Pleuropulmonary Blastoma; Pregnancy and Breast Cancer; Pregnancy and Hodgkin's Lymphoma; Pregnancy and Non-Hodgkin's Lymphoma; Primary Central Nervous System Lymphoma; Primary Liver Cancer, Adult; Primary Liver Cancer, Childhood; Prostate Cancer; Rectal Cancer; Renal Cell (Kidney) Cancer; Renal Cell Cancer, Childhood; Renal Pelvis and Ureter, Transitional Cell Cancer; Retinoblastoma; Rhabdomyosarcoma, Childhood; Salivary Gland Cancer; Salivary Gland Cancer, Childhood; Sarcoma, Ewing's Family of Tumors; Sarcoma, Kaposi's; Sarcoma (Osteosarcoma)/Malignant Fibrous Histiocytoma of Bone; Sarcoma, Rhabdomyosarcoma, Childhood; Sarcoma, Soft Tissue, Adult; Sarcoma, Soft Tissue, Childhood; Sezary Syndrome; Skin Cancer; Skin Cancer, Childhood; Skin Cancer (Melanoma); Skin Carcinoma, Merkel Cell; Small Cell Lung Cancer; Small Intestine Cancer; Soft Tissue Sarcoma, Adult; Soft Tissue Sarcoma, Childhood; Squamous Neck Cancer with Occult Primary, Metastatic; Stomach (Gastric) Cancer; Stomach (Gastric) Cancer, Childhood; Supratentorial Primitive Neuroectodermal Tumors, Childhood; T-Cell Lymphoma, Cutaneous; Testicular Cancer; Thymoma, Childhood; Thymoma, Malignant; Thyroid Cancer; Thyroid Cancer, Childhood; Transitional Cell Cancer of the Renal Pelvis and Ureter; Trophoblastic Tumor, Gestational; Unknown Primary Site, Cancer of, Childhood; Unusual Cancers of Childhood; Ureter and Renal Pelvis, Transitional Cell Cancer; Urethral Cancer; Uterine Sarcoma; Vaginal Cancer; Visual Pathway and Hypothalamic Glioma, Childhood; Vulvar Cancer; Waldenstrom's Macro globulinemia; and Wilms' Tumor. Metastases of the aforementioned cancers can also be treated or prevented in accordance with the methods described herein.
-
The methods described herein are useful in treating cancer in nervous system, e.g., brain tumor, e.g., glioma, e.g., glioblastoma multiforme (GBM), e.g., by inhibiting a neoactivity of a mutant enzyme, e.g., an enzyme in a metabolic pathway, e.g., a metabolic pathway leading to fatty acid biosynthesis, glycolysis, glutaminolysis, the pentose phosphate shunt, the nucleotide biosynthetic pathway, or the fatty acid biosynthetic pathway, e.g., IDH1 or IDH2.
-
Gliomas, a type of brain tumors, can be classified as grade Ito grade IV on the basis of histopathological and clinical criteria established by the World Health Organization (WHO). WHO grade I gliomas are often considered benign. Gliomas of WHO grade II or III are invasive, progress to higher-grade lesions. WHO grade IV tumors (glioblastomas) are the most invasive form. Exemplary brain tumors include, e.g., astrocytic tumor (e.g., pilocytic astrocytoma, subependymal giant-cell astrocytoma, diffuse astrocytoma, pleomorphic xanthoastrocytoma, anaplastic astrocytoma, astrocytoma, giant cell glioblastoma, glioblastoma, secondary glioblastoma, primary adult glioblastoma, and primary pediatric glioblastoma); oligodendroglia) tumor (e.g., oligodendroglioma, and anaplastic oligodendroglioma); oligoastrocytic tumor (e.g., oligoastrocytoma, and anaplastic oligoastrocytoma); ependymoma (e.g., myxopapillary ependymoma, and anaplastic ependymoma); medulloblastoma; primitive neuroectodermal tumor, schwannoma, meningioma, meatypical meningioma, anaplastic meningioma; and pituitary adenoma. Exemplary cancers are described in Acta Neuropathol (2008) 116:597-602 and N Engl J. Med. 2009 Feb. 19; 360(8):765-73, the contents of which are each incorporated herein by reference.
-
In embodiments the disorder is glioblastoma.
-
In an embodiment the disorder is prostate cancer, e.g., stage T1 (e.g., T1a, T1b and T1c), T2 (e.g., T2a, T2b and T2c), T3 (e.g., T3a and T3b) and T4, on the TNM staging system. In embodiments the prostate cancer is grade G1, G2, G3 or G4 (where a higher number indicates greater difference from normal tissue). Types of prostate cancer include, e.g., prostate adenocarcinoma, small cell carcinoma, squamous carcinoma, sarcomas, and transitional cell carcinoma.
-
Methods and compositions of the invention can be combined with art-known treatment. Art-known treatment for prostate cancer can include, e.g., active surveillance, surgery (e.g., radical prostatectomy, transurethral resection of the prostate, orchiectomy, and cryosurgegry), radiation therapy including brachytherapy (prostate brachytherapy) and external beam radiation therapy, High-Intensity Focused Ultrasound (HIFU), chemotherapy, cryosurgery, hormonal therapy (e.g., antiandrogens (e.g., flutamide, bicalutamide, nilutamide and cyproterone acetate, ketoconazole, aminoglutethimide), GnRH antagonists (e.g., Abarelix)), or a combination thereof.
-
All references described herein are expressly incorporated herein by reference.
EXAMPLES
Example 1
IDH1 Cloning, Mutagenesis, Expression and Purification
-
1. Wild Type IDH1 was Cloned into pET41a, Creating His8 Tag at C-Terminus.
-
The IDH1 gene coding region (cDNA) was purchased from Invitrogen in pENTR221 vector (www.invitrogen.com, Cat#B-068487_Ultimate_ORF). Oligo nucleotides were designed to PCR out the coding region of IDH1 with NdeI at the 5′ end and XhoI at the 3′. (IDH1-f: TAATCATATGTCCAAAAAAATCAGT (SEQ ID NO:1), IDH1-r: TAATCTCGAGTGAAAGTTTGGCCTGAGCTAGTT (SEQ ID NO:2)). The PCR product is cloned into the NdeI/XhoI cleaved pET41a vector. NdeI/XhoI cleavage of the vector pET41a releases the GST portion of the plasmid, and creating a C-terminal His8 tag (SEQ ID NO:3) without the N-terminal GST fusion. The original stop codon of IDH1 is change to serine, so the junction sequence in final IDH1 protein is: Ser-Leu-Glu-His-His-His-His-His-His-His-His-Stop (SEQ ID NO:4).
-
The C-terminal His tag strategy instead of N-terminal His tag strategy was chosen, because C-terminal tag might not negatively impact IDH1 protein folding or activity. See, e.g., Xu X et al, J Biol Chem. 2004 Aug. 6; 279(32):33946-57.
-
The sequence for pET41a-IDH1 plasmid is confirmed by DNA sequencing. FIG. 1 shows detailed sequence verification of pET41a-IDH1 and alignment against published IDH1 CDS below.
2. IDH1 Site Directed Mutagenesis to Create the IDHr132s and IDHr132H Mutants.
-
Site directed mutagenesis was performed to convert R132 to S or H, DNA sequencing confirmed that G395 is mutated to A (creating Arg→His mutation in the IDH1 protein), and C394 is mutated to A (creating Arg→Ser in the IDH1 protein). Detailed method for site directed mutagenesis is described in the user manual for QuikChange® MultiSite-Directed Mutagenesis Kit (Stratagene, cat#200531). FIG. 2 shows DNA sequence verification of such mutations. Highlighted nucleotides were successfully changed in the mutagenesis: G395→A mutation allows amino acid Arg132→His; C394→A mutation allows amino acid Arg132→Ser.
3. IDH1 Protein Expression and Purification.
-
IDHwt, IDHR132S, and IDHR132H proteins were expressed in the E. coli strain Rosetta and purified according to the detailed procedure below. Active IDH1 proteins are in dimer form, and SEC column fraction/peak that correspond to the dimer form were collected for enzymology analysis and cross comparison of catalytic activities of these proteins.
A. Cell Culturing:
-
Cells were grown in LB (20 μg/ml Kanamycin) at 37° C. with shaking until OD600 reaches 0.6. The temperature was changed to 18° C. and protein was induced by adding IPTG to final concentration of 1 mM. Cells were collected 12-16 hours after IPTG induction.
B. Buffer System:
-
Lysis buffer: 20 mM Tris, pH7.4, 0.1% Triton X-100, 500 mM NaCl, 1 mM PMSF, 5 mM (3-mercaptoethanol, 10% glycerol.
-
Ni-Column Buffer A: 20 mM Tris, pH7.4, 500 mM NaCl, 5 mM β-mercaptoethanol, 10% glycerol.
-
Ni-column Buffer B: 20 mM Tris, pH7.4, 500 mM NaCl, 5 mM β-mercaptoethanol, 500 mM Imidazole, 10% glycerol
-
Gel filtration Buffer C: 200 mM NaCl, 50 mM Tris 7.5, 5 mM (3-mercaptoethanol, 2 mM MnSO4, 10% glycerol.
C. Protein Purification Procedure
-
1. Cell pellet were resuspended in the lysis buffer (1 gram cell/5-10 ml buffer).
2. Cells were broken by passing the cell through Microfludizer with at a pressure of 15,000 psi for 3 times.
3. Soluble protein was collected from supernatant after centrifugation at 20,000 g (Beckman Avanti J-26XP) for 30 min at 4° C.
4. 5-10 ml of Ni-column was equilibrated by Buffer A until the A280 value reached baseline. The supernatant was loaded onto a 5-ml Ni-Sepharose column (2 ml/min). The column was washed by 10-20 CV of washing buffer (90% buffer A+10% buffer B) until A280 reach the baseline (2 ml/min).
5. The protein was eluted by liner gradient of 10-100% buffer B (20 CV) with the flow rate of 2 ml/min and the sample fractions were collected as 2 ml/tube.
6. The samples were analyzed on SDS-PAGE gel.
7. The samples were collected and dialyzed against 200× Gel filtration buffer for 2 times (1 hour and >4 hours).
8. The samples were concentrated to 10 ml.
9. 200 ml of S-200 Gel-filtration column was equilibrated by buffer C until the A280 value reached baseline. The samples were loaded onto Gel filtration column (0.5 ml/min).
10. The column was washed by 10 CV of buffer C, collect fractions as 2-4 ml/tube.
11. The samples were analyzed on SDS-PAGE gel and protein concentration was determined.
D. Protein Purification Results
-
The results for purification of wild type IDH1 are shown in FIGS. 3, 4, 5A and 5B.
-
The results for purification of mutant IDH1R132S are shown in FIGS. 6, 7, 8A and 8B.
-
The results for purification of wild type IDH1R132H are shown in FIGS. 9, 10, 11A and 11B.
Example 2
Enzymology Analysis of IDH1 Wild Type and Mutants
1. Analysis of IDH1 Wild-Type and Mutants R132H and R132S in the Oxidative Decarboxylation of Isocitrate to α-Ketoglutarate (α-KG).
A. Methods
-
To determine the catalytic efficiency of enzymes in the oxidative decarboxylation of isocitrate to α-Ketoglutarate (α-KG) direction, reactions were performed to determine Vmax and Km for isocitrate. In these reactions, the substrate was varied while the cofactor was held constant at 500 uM. All reactions were performed in 150 mM NaCl, 20 mM Tris-Cl, pH 7.5, 10% glycerol, and 0.03% (w/v) BSA). Reaction progress was followed by spectroscopy at 340 nM monitoring the change in oxidation state of the cofactor. Sufficient enzyme was added to give a linear change in absorbance for 10 minutes.
-
B. ICDH1 R132H and ICDH1 R132S are impaired for conversion of isocitrate to α-KG.
-
Michaelis-Menten plots for the relationship of isocitrate concentration to reaction velocity are presented in FIGS. 12A-12C. Kinetic parameters are summarized in the Table 1. All data was fit to the Hill equation by least-squares regression analysis.
-
| TABLE 1 |
| |
| |
|
|
|
|
Relative |
| |
Vmax |
Km |
Hill |
|
Catalytic |
| Enzyme |
(umol/min/mg) |
(uM) |
Constant |
Vmax/Km |
Efficiency |
| |
| |
| Wt |
30.5 |
56.8 |
1.8 |
0.537 |
100% |
|
| R132H |
0.605 |
171.7 |
0.6 |
0.0035 |
0.35% |
| R132S |
| |
95 |
>1e6 |
0.479 |
<9.5e7 |
<.001% |
| |
-
Both mutant enzymes display a reduced Hill coefficient and an increase in Km for isocitrate, suggesting a loss of co-operatively in substrate binding and/or reduced affinity for substrate. R132H enzyme also displays a reduced Vmax, suggestive of a lower kcat. R132S displays an increase in Vmax, suggesting an increase in kcat, although this comes at the expense of a 20,000 fold increase in Km so that the overall effect on catalytic efficiency is a great decrease as compared to the wild-type enzyme. The relative catalytic efficiency, described as Vmax/Km, is dramatically lower for the mutants as compared to wild-type. The in vivo effect of these mutations would be to decrease the flux conversion of isocitrate to α-KG.
C. The ICDH1 R132H and R132S Mutants Display Reduced Product Inhibition in the Oxidative Decarboxylation of Isocitrate to α-Ketoglutarate (α-Kg).
-
A well-known regulatory mechanism for control of metabolic enzymes is feedback inhibition, in which the product of the reaction acts as a negative regulator for the generating enzyme. To examine whether the R132S or R132H mutants maintain this regulatory mechanism, the Ki for α-KG in the oxidative decarboxylation of ioscitrate to α-ketoglutarate was determined. Data is presented in FIGS. 13A-13C and summarized in Table 2. In all cases, α-KG acts as a competitive inhibitor of the isocitrate substrate. However, R132H and R132S display a 20-fold and 13-fold increase in sensitivity to feedback inhibition as compared to the wild-type enzyme.
-
| |
Wt |
612.2 |
| |
R132H |
28.6 |
| |
R132S |
45.3 |
| |
|
D. The Effect of MnCl7 in Oxidative Decarboxylation of Isocitrate to α-Ketoglutarate (α-KG).
-
MnCl2 can be substituted with MgCl2 to examine if there is any difference in oxidative decarboxylation of isocitrate to α-Ketoglutarate (α-KG).
E. The Effect of R132 Mutations on the Inhibitory Effect of Oxalomalate on IDH1
-
The purpose of this example is to examine the susceptibility of IDH1R132S and IDH1R132H in oxidative decarboxylation of isocitrate to α-Ketoglutarate (α-KG) to the known IDH1 inhibitor oxalomalate. Experiments were performed to examine if R132 mutations circumvent the inhibition by oxalomalate.
-
Final concentrations: Tris 7.5 20 mM, NaCl 150 mM, MnCl 2 2 mM, Glycerol 10%, BSA 0.03%, NADP 0.5 mM, IDH1 wt 1.5 ug/ml, IDH1R132S 30 ug/ml, IDH1R132H 60 ug/ml, DL-isocitrate (5-650 uM). The results are summarized in FIG. 17 and Table 3. The R132S mutation displays approximately a two-fold increase in susceptibility to inhibition by oxalomalate, while the R132H mutation is essentially unaffected. In all three cases, the same fully competitive mode of inhibition with regards to isocitrate was observed.
-
| |
TABLE 3 |
| |
|
| |
Enzyme |
Oxalomalate Ki (uM) |
| |
|
| |
| |
wt |
955.4 |
| |
R132S |
510 |
| |
R132H |
950.8 |
| |
|
F. Forward Reactions (Isocitrate to α-KG) of Mutant Enzyme do not go to Completion.
-
Forward reactions containing ICDH1 R132S or ICDH1 R132H were assembled and reaction progress monitored by an increase in the OD340 of the reduced NADPH cofactor. It was observed (FIG. 23), that these reactions proceed in the forward direction for a period of time and then reverse direction and oxidize the cofactor reduced in the early stages of the reaction, essentially to the starting concentration present at the initiation of the experiment. Addition of further isocitrate re-initiated the forward reaction for a period of time, but again did not induce the reaction to proceed to completion. Rather, the system returned to initial concentrations of NADPH. This experiment suggested that the mutant enzymes were performing a reverse reaction other than the conversion of α-KG to isocitrate.
2. Analysis of IDH1 Wild-Type and Mutants R132H and R132S in the Reduction of α-Ketoglutarate (α-KG).
A. Methods
-
To determine the catalytic efficiency of enzymes in the reduction of α-Ketoglutarate (α-KG), reactions were performed to determine Vmax and Km for α-KG. In these reactions, substrate was varied while the cofactor was held constant at 500 uM. All reactions were performed in 50 mM potassium phosphate buffer, pH 6.5, 10% glycerol, 0.03% (w/v) BSA, 5 mM MgCl2, and 40 mM sodium hydrocarbonate. Reaction progress was followed by spectroscopy at 340 nM monitoring the change in oxidation state of the cofactor. Sufficient enzyme was added to give a linear change in absorbance for 10 minutes.
B. The R132H and R132S Mutant Enzymes, but not the Wild-Type Enzyme, Support the Reduction of α-KG.
-
To test the ability of the mutant and wild-type enzymes to perform the reduction of α-KG, 40 ug/ml of enzyme was incubated under the conditions for the reduction of α-Ketoglutarate (α-KG) as described above. Results are presented in FIG. 14. The wild-type enzyme was unable to consume NADPH, while R132S and R132H reduced α-KG and consumed NADPH.
C. The Reduction of α-KG by the R132H and R132S Mutants Occurs In Vitro at Physiologically Relevant Concentrations of α-Kg.
-
To determine the kinetic parameters of the reduction of α-KG performed by the mutant enzymes, a substrate titration experiment was performed, as presented in FIGS. 15A-15B. R132H maintained the Hill-type substrate interaction as seen in the oxidative decarboxylation of isocitrate, but displayed positive substrate co-operative binding. R132S showed a conversion to Michaelis-Menten kinetics with the addition of uncompetitive substrate inhibition, as compared to wild-type enzyme in the oxidative decarboxylation of isocitrate. The enzymatic parameters of the mutant enzyme are presented in Table 4. Since the wild-type enzyme did not consume measurable NADPH in the experiment described above, a full kinetic workup was not performed.
-
| TABLE 4 |
| |
| |
Vmax |
Km |
Hill |
Ki |
|
| Enzyme |
(umol/min/mg) |
(mM) |
Constant |
(mM) |
Vmax/Km |
| |
| |
| R132H |
1.3 |
0.965 |
1.8 |
|
1.35 |
| R132S |
2.7 |
0.181 |
0.479 |
24.6 |
14.92 |
| |
-
The relative catalytic efficiency of reduction of α-KG is approximately ten-fold higher in the R132S mutant than in the R132H mutant. The biological consequence is that the rate of metabolic flux should be greater in cells expressing R132S as compared to R132H.
-
D. Analysis of IDH1 Wild-Type and Mutants R132H and R132S in the Reduction of Alpha-Ketoglutarate with NADH.
-
In order to evaluate the ability of the mutant enzymes to utilize NADH in the reduction of alpha-ketoglutarate, the following experiment was conducted. Final concentrations: NaHCO3 40 mM, MgCl2 5 mM, Glycerol 10%, K2HPO4 50 mM, BSA 0.03%, NADH 0.5 mM, IDH1 wt 5 ug/ml, R132S 30 ug/ml, R132H 60 ug/ml, alpha-Ketoglutarate 5 mM.
-
The results are shown in FIG. 16 and Table 5. The R132S mutant demonstrated the ability to utilize NADH while the wild type and R132H show no measurable consumption of NADH in the presence of alpha-ketoglutarate.
-
| TABLE 5 |
| |
| Consumption of NADH by R132S in the |
| presence of alpha-ketoglutarate |
| Rate (ΔA/sec) |
0.001117 |
0.001088 |
0.001103 |
2.05E−05 |
| Umol/min/mg |
0.718328 |
0.699678 |
0.709003 |
0.013187 |
| |
Summary
-
To understand how R132 mutations alter the enzymatic properties of IDH1, wild-type and R132H mutant IDH1 proteins were produced and purified from E. coli. When NADP+-dependent oxidative decarboxylation of isocitrate was measured using purified wild-type or R132H mutant IDH1 protein, it was confirmed that R132H mutation impairs the ability of IDH1 to catalyze this reaction (Yan, H. et al. N Engl J Med 360, 765-73 (2009); Zhao, S. et al. Science 324, 261-5 (2009)), as evident by the loss in binding affinity for both isocitrate and MgCl2 along with a 1000-fold decrease in catalytic turnover (FIGS. 30A and 30C). In contrast, when NADPH-dependent reduction of αKG was assessed using either wild-type or R132H mutant IDH1 protein, only R132H mutant could catalyze this reaction at a measurable rate (FIGS. 30 and 30C). Part of this increased rate of αKG reduction results from an increase in binding affinity for both the cofactor NADPH and substrate αKG in the R132H mutant IDH1 (FIG. 30C). Taken together, these data demonstrate that while the R132H mutation leads to a loss of enzymatic function for oxidative decarboxylation of isocitrate, this mutation also results in a gain of enzyme function for the NADPH-dependent reduction of αKG.
2: Analysis of Mutant IDH1
-
The R132H Mutant does not Result in the Conversion of α-KG to Isocitrate.
-
Using standard experimental methods, an API2000 mass spectrometer was configured for optimal detection of α-KG and isocitrate (Table 6). MRM transitions were selected and tuned such that each analyte was monitored by a unique transition. Then, an enzymatic reaction containing 1 mM α-KG, 1 mM NADPH, and ICDH1 R132H were assembled and run to completion as judged by the decrease to baseline of the optical absorbance at 340 nM. A control reaction was performed in parallel from which the enzyme was omitted. Reactions were quenched 1:1 with methanol, extracted, and subjected to analysis by LC-MS/MS.
-
FIG. 18A presents the control reaction indicating that αKG was not consumed in the absence of enzyme, and no detectable isocitrate was present. FIG. 18B presents the reaction containing R132H enzyme, in which the α-KG has been consumed, but no isocitrate was detected. FIG. 18C presents a second analysis of the reaction containing enzyme in which isocitrate has been spiked to a final concentration of 1 mM, demonstrating that had α-KG been converted to isocitrate at any appreciable concentration greater than 0.01%, the configured analytical system would have been capable of detecting its presence in the reaction containing enzyme. The conclusion from this experiment is that while α-KG was consumed by R132H, isocitrate was not produced. This experiment indicates that one neoactivity of the R132H mutant is the reduction of α-KG to a compound other than isocitrate.
-
| TABLE 6 |
| |
| Instrument settings for MRM detection of compounds |
| Compound |
Q1 |
Q3 |
DP |
FP |
EP |
CEP |
CE |
CXP |
| |
| α-KG |
144.975 |
100.6 |
−6 |
−220 |
−10 |
−16 |
−10 |
−22 |
| isocitrate |
191.235 |
110.9 |
−11 |
−230 |
−4.5 |
−14 |
−16 |
−24 |
| a-hydroxy- |
147.085 |
128.7 |
−11 |
−280 |
−10 |
−22 |
−12 |
−24 |
| glutarate |
| |
-
The R132H Mutant Reduces α-KG to 2-Hydroxyglutaric Acid.
-
Using standard experimental methods, an API2000 mass spectrometer was configured for optimal detection 2-hydroxyglutarate (Table 6 and FIG. 19). The reaction products of the control and enzyme-containing reactions from above were investigated for the presence of 2-hydroxyglutaric acid, FIG. 20. In the control reaction, no 2-hydroxyglutaric acid was detected, while in reaction containing R132H, 2-hydroxyglutaric acid was detected. This data confirms that one neoactivity of the R132H mutant is the reduction of α-KG to 2-hydroxyglutaric acid.
-
To determine whether R132H mutant protein directly produced 2HG from αKG, the product of the mutant IDH1 reaction was examined using negative ion mode triple quadrupole electrospray LC-MS. These experiments confirmed that 2HG was the direct product of NADPH-dependent αKG reduction by the purified R132H mutant protein through comparison with a known metabolite standards (FIG. 31A). Conversion of αKG to isocitrate was not observed.
-
One can determine the enantiomeric specificity of the reaction product through derivitazation with DATAN (diacetyl-L-tartaric acid) and comparing the retention time to that of known R and S standards. This method is described in Struys et al. Clin Chem 50:1391-1395 (2004). The stereo-specific production of either the R or S enantomer of alpha-hydroxyglutaric acid by ICDH1 R132H may modify the biological activity of other enzymes present in the cell. The racemic production may also occur.
-
For example, one can measure the inhibitory effect of alpha-hydroxyglutaric acid on the enzymatic activity of enzymes which utilize α-KG as a substrate. In one embodiment, alpha-hydroxyglutaric acid may be a substrate- or product-analogue inhibitor of wild-type ICDH1. In another embodiment alpha-hydroxyglutaric acid may be a substrate- or product-analogue inhibitor of HIF1 prolyl hydroxylase. In the former case, inhibition of wild type ICDH1 by the enzymatic product of R132H will reduce the circulating levels of αKG in the cell. In the latter case, inhibition of HIF1 prolyl hydroxylase will result in the stabilization of HIF1 and an induction of the hypoxic response cohort of cellular responses.
ICDH R132H Reduces αKG to the R-Enantiomer of 2-Hydroxyglutarate.
-
There are two possible enantiomers of the ICDHR132H reductive reaction product, converting alpha-ketoglutarate to 2-hydroxyglutarate, with the chiral center being located at the alpha-carbon position. Exemplary products are depicted below.
-
-
These are referred to by those with knowledge in the art as the R (or pro-R) and S (or pro-S) enantiomers, respectively. In order to determine which form or both is produced as a result of the ICDH1 neoactivity described above, the relative amount of each chiral form in the reaction product was determined in the procedure described below.
-
Reduction of α-KG to 2-HG was performed by ICDHR132H in the presence of NADPH as described above, and the reaction progress was monitored by a change in extinction coefficient of the nucleotide cofactor at 340 nM; once the reaction was judged to be complete, the reaction was extracted with methanol and dried down completely in a stream of nitrogen gas. In parallel, samples of chirally pure R-2-HG and a racemic mixture of R- and S-2-HG (produced by a purely chemical reduction of α-KG to 2-HG) were resuspended in ddH2O, similarly extracted with methanol, and dried.
-
The reaction products or chiral standards were then resuspended in a solution of dichloromethane:acetic acid (4:1) containing 50 g/L DATAN and heated to 75° C. for 30 minutes to promote the derivitization of 2-HG in the scheme described below:
-
-
After cooling to room temperature, the derivitization reactions were dried to completion and resuspended in ddH2O for analysis on an LC-MS/MS system. Analysis of reaction products and chiral standards was performed on an API2000 LC-MS/MS system using a 2×150 mM C18 column with an isocratic flow of 200 μl/min of 90:10 (ammonium formate, pH 3.6:methanol) and monitoring the retention times of the 2-HG-DATAN complex using XIC and the diagnostic MRM transition of 363/147 in the negative ion mode.
-
It should be noted that retention times in the experiments described below are approximate and accurate to within +/−1 minute; the highly reproducible peak seen at 4 minutes is an artifact of a column switching valve whose presence has no result on the conclusions drawn from the experiment.
-
Injection of the racemic mixture gave two peaks of equal area at retention times of 8 and 10 minutes (FIG. 24A), while injection of the R-2-HG standard resulted in a major peak of >95% area at 10 minutes and a minor peak <5% area at 8 minutes (FIG. 24B); indicating that the R-2-HG standard is approximately 95% R and 5% S. Thus, this method allows us to separate the R and S-2-HG chiral forms and to determine the relative amounts of each in a given sample. Coinjection of the racemic mixture and the R-2-HG standard resulted in two peaks at 8 and 10 minutes, with a larger peak at 10 minutes resulting from the addition of surplus pro-R-form (the standard) to a previously equal mixture of R- and S-2-HG (FIG. 24C). These experiments allow us to assign the 8 minute peak to the S-2-HG form and the 10 minute peak to the R-2-HG form.
-
Injection of the derivatized neoactivity enzyme reaction product alone yields a single peak at 10 minutes, suggesting that the neoactivity reaction product is chirally pure R-2-HG (FIG. 24D). Coinjection of the neoactivity reaction product with the R-2-HG standard results in a major peak of >95% area at 10 minutes (FIG. 24E) and a single minor peak of <5% area at 8 minutes (previously observed in injection of the R-2-HG standard alone) confirming the chirality of the neoactivity product as R. Coinjection of a racemic mixture and the neoactivity reaction product (FIG. 24F) results in a 60% area peak at 10 minutes and a 40% area peak at 8 minutes; this deviation from the previously symmetrical peak areas observed in the racemate sample being due to the excess presence of R-2-HG form contributed by the addition of the neoactivity reaction product.
-
These experiments allow us to conclude that the ICDH1 neoactivity is a highly specific chiral reduction of α-KG to R-2-HG.
Enzyme Properties of Other IDH1 Mutations
-
To determine whether the altered enzyme properties resulting from R132H mutation were shared by other R132 mutations found in human gliomas, recombinant R132C, R132L and R132S mutant IDH1 proteins were generated and the enzymatic properties assessed. Similar to R132H mutant protein, R132C, R132L, and R132S mutations all result in a gain-of-function for NADPH-dependent reduction of αKG (data not shown). Thus, in addition to impaired oxidative decarboxylation of isocitrate, one common feature shared among the IDH1 mutations found in human gliomas is the ability to catalyze direct NADPH-dependent reduction of αKG.
Identification of 2-HG Production in Glioblastoma Cell Lines Containing the IDH-1 R132H Mutant Protein.
-
Generation of genetic engineered glioblastoma cell lines expressing wildtype or mutant IDH-1 protein. A carboxy-terminal Myc-DDK-tagged open reading frame (ORF) clone of human isocitrate dehydrogenase 1 (IDH1; Ref. ID: NM—005896) cloned in vector pCMV6 was obtained from commercial vendor Origen Inc. Vector pCMV6 contains both kanamycin and neomycin resistance cassettes for selection in both bacterial and mammalian cell systems. Standard molecular biology mutagenesis techniques were utilized to alter the DNA sequence at base pair 364 of the ORF to introduce base pair change from guanine to adenine resulting in a change in the amino acid code at position 132 from argentine (wt) to histidine (mutant; or R132H). Specific DNA sequence alteration was confirmed by standard methods for DNA sequence analysis. Parental vector pCMV6 (no insert), pCMV6-wt IDH1 or pCMV6-R132H were transfected into immortalized human glioblastoma cell lines ATCC® CRL-2610 (LN-18) or HTB-14 (U-87) in standard growth medium (DMEM; Dulbecco's modified Eagles Medium containing 10% fetal bovine serum). Approximately 24 hrs after transfection, the cell cultures were transitioned to DMEM containing G418 sodium salt at concentrations of either 750 ug/ml (CRL-2610) or 500 ug/ml (HTB-14) to select those cells in culture that expressed the integrated DNA cassette expressing both the neomycin selectable marker and the ORF for human wild type or R132H. Pooled populations of G418 resistant cells were generated and expression of either wild type IDH1 or R132 IDH1 was confirmed by standard Western blot analysis of cell lysates using commercial antibodies recognizing either human IDH1 antigen or the engineered carboxy-terminal MYC-DDK expression tag. These stable clonal pools were then utilized for metaobolite preparation and analysis.
-
Procedure for metabolite preparation and analysis. Glioblastoma cell lines (CRL-2610 and HTB-14) expressing wildtype or mutant IDH-1 protein were grown using standard mammalian tissue culture techniques on DMEM media containing 10% FCS, 25 mM glucose, 4 mM glutamine, and G418 antibiotic (CRL-2610 at 750 ug/mL; HTB-14 at 500 ug/mL) to insure ongoing selection to preserve the transfected mutant expression sequences. In preparation for metabolite extraction experiments, cells were passaged into 10 cm round culture dishes at a density of 1×106 cells. Approximately 12 hours prior to metabolite extraction, the culture media was changed (8 mL per plate) to DMEM containing 10% dialyzed FCS (10,000 mwco), 5 mM glucose, 4 mM glutamine, and G-418 antibiotic as before; the dialyzed FCS removes multiple small molecules form the culture media and enables cell culture-specific assessment of metabolite levels. The media was again changed 2 hours prior to metabolite extraction. Metabolite extraction was accomplished by quickly aspirating the media from the culture dishes in a sterile hood, immediately placing the dishes in a tray containing dry ice to cool them to −80° C., and as quickly as possible, adding 2.6 mL of 80% MeOH/20% water, pre-chilled to −80° C. in a dry-ice/acetone bath. These chilled, methanol extracted cells were then physically separated from the culture dish by scraping with a sterile polyethylene cell lifter (Corning #3008), brought into suspension and transferred to a 15 mL conical vial, then chilled to −20° C. An additional 1.0 mL of 80% MeOH/20% water was applied to the chilled culture dish and the cell lifting procedure repeated, to give a final extraction volume of 3.6 mL. The extracts were centrifuged at 20,000×g for 30 minutes to sediment the cell debris, and 3.0 mL of the supernatants was transferred to a screw-cap freezer vial and stored at −80° C. until ready for analysis.
-
In preparation for analysis, the extracts were removed from the freezer and dried on a nitrogen blower to remove methanol. The 100% aqueous samples were analyzed by LCMS as follows. The extract (10 μL) was injected onto a reverse-phase HPLC column (Synergi 150 mm×2 mm, Phenomenex Inc.) and eluted using a linear gradient of LCMS-grade methanol (Buffer B) in Aq. 10 mM tributylamine, 15 mM Acetic acid (Buffer A), running from 3% Buffer B to 95% Buffer B over 45 minutes at 200 μL/min. Eluted metabolite ions were detected using a triple-quadrapole mass spectrometer, tuned to detect in negative mode with multiple-reaction-monitoring mode transition set (MRM's) according to the molecular weights and fragmentation patterns for 38 known central metabolites, including 2-hydroxyglutarate (MRM parameters were optimized by prior infusion of known compound standards). Data was processed using Analyst Software (Applied Biosystems, Inc.) and metabolite signal intensities were converted into absolute concentrations using signal build-up curves from injected mixtures of metabolite standards at known concentrations. Final metabolite concentrations were reported as mean of at least three replicates, +/− standard deviation.
-
Results. Analyses reveal significantly higher levels of 2-HG in cells that express the IDH-1 R132H mutant protein. As shown in FIG. 26A, levels of 2-HG in CRL-2610 cell lines expressing the IDH-1 R132H mutant protein are approximately 28-fold higher than identical lines expressing the wild-type protein. Similarly, levels of 2-HG in HTB-14 cell lines expressing the IDH-1 R132H mutant protein are approximately 38-fold higher than identical lines expressing the wild-type protein, as shown in FIG. 26B.
Evaluation of 2-Hydroxyglutarate (2-HG) Production in Human Glioblastoma Tumors Containing Mutations in Isocitrate Dehydrogenase 1 (IDH1) at Amino Acid 132.
-
Heterozygous somatic mutations at nucleotide position 395 (amino acid codon 132) in the transcript encoding isocitrate dehydrogenase 1 (IDH1) can occur in brain tumors.
-
Tissue source: Human brain tumors were obtained during surgical resection, flash frozen in liquid nitrogen and stored at −80° C. Clinical classification of the tissue as gliomas was performed using standard clinical pathology categorization and grading.
-
Genomic sequence analysis to identify brain tumor samples containing either wild type isocitrate dehydrogenase (IDH1) or mutations altering amino acid 132. Genomic DNA was isolated from 50-100 mgs of brain tumor tissue using standard methods. A polymerase chain reaction (PCR) procedure was then performed on the isolated genomic DNA to amplify a 295 base pair fragment of the genomic DNA that contains both intron and 2nd exon sequences of human IDH1 (FIG. 27). In FIG. 27, intron sequence is shown in lower case font; 2nd exon IDH1 DNA sequence is shown in upper case font; forward (5′) and reverse (3′) primer sequences are shown in underlined font; guanine nucleotide mutated in a subset of human glioma tumors is shown in bold underlined font.
-
The amplified DNA fragment was then sequenced using standard protocols and sequence alignments were performed to classify the sequences as either wild type or mutant at the guanine nucleotide at base pair 170 of the amplified PCR fragment. Tumors were identified that contained genomic DNA having either two copies of guanine (wild type) or a mixed or monoalellic combination of one IDH1 allele containing guanine and the other an adenine (mutant) sequence at base pair 170 of the amplified product (Table 15). The nucleotide change results in a change at amino acid position 132 of human IDH1 protein from arginine (wild type) to histidine (mutant) as has been previously reported.
-
| TABLE 15 |
| |
| Sequence variance at base pair 170 of the amplified |
| genomic DNA from human glioma samples. |
| |
Sample |
Base |
IDH1 Amino Acid |
|
| |
ID |
170 |
132 |
Genotype |
| |
|
| |
1102 |
G |
arginine |
wild type |
| |
1822 |
A |
histidine |
mutant |
| |
496 |
G |
arginine |
wild type |
| |
1874 |
A |
histidine |
mutant |
| |
816 |
A |
histidine |
mutant |
| |
534 |
G |
arginine |
wild type |
| |
AP-1 |
A |
histidine |
mutant |
| |
AP-2 |
A |
histidine |
mutant |
| |
|
-
Procedure for metabolite preparation and analysis. Metabolite extraction was accomplished by adding a 10× volume (m/v ratio) of −80 C methanol:water mix (80%:20%) to the brain tissue (approximately 100 mgs) followed by 30 s homogenization at 4 C. These chilled, methanol extracted homogenized tissues were then centrifuged at 14,000 rpm for 30 minutes to sediment the cellular and tissue debris and the cleared tissue supernatants were transferred to a screw-cap freezer vial and stored at −80° C. For analysis, a 2× volume of tributylamine (10 mM) acetic acid (10 mM) pH 5.5 was added to the samples and analyzed by LCMS as follows. Sample extracts were filtered using a Millex-FG 0.20 micron disk and 10 μL were injected onto a reverse-phase HPLC column (Synergi 150 mm×2 mm, Phenomenex Inc.) and eluted using a linear gradient LCMS-grade methanol (50%) with 10 mM tributylamine and 10 mM acetic acid) ramping to 80% methanl:10 mM tributylamine: 10 mM acetic acid over 6 minutes at 200 μL/min. Eluted metabolite ions were detected using a triple-quadrapole mass spectrometer, tuned to detect in negative mode with multiple-reaction-monitoring mode transition set (MRM's) according to the molecular weights and fragmentation patterns for 8 known central metabolites, including 2-hydroxyglutarate (MRM parameters were optimized by prior infusion of known compound standards). Data was processed using Analyst Software (Applied Biosystems, Inc.) and metabolite signal intensities were obtained by standard peak integration methods.
-
Results. Analyses revealed dramatically higher levels of 2-HG in cells tumor samples that express the IDH-1 R132H mutant protein. Data is summarized in Table 16 and FIG. 28.
-
| TABLE 16 |
| |
| |
|
|
Tumor |
|
|
|
|
|
|
|
|
|
| |
|
|
Cells |
| |
|
|
in |
| |
|
|
Tumor |
|
Nucleo- |
| mple |
Primary Specimen |
|
Foci |
Geno- |
tide |
|
2HG |
□GKG |
Malate |
Fumarate |
Succinate |
Isocitrate |
| ID |
Diagnosis |
Grade |
(%) |
type |
change |
Codon |
(□mole/g) |
(□mole/g) |
(□mole/g) |
(□mole/g) |
(□mole/g) |
(□mole/g) |
| |
| |
| 1 |
Glioblastoma, |
WHO |
n/a |
wild |
wild |
R132 |
0.18 |
0.161 |
1.182 |
0.923 |
1.075 |
0.041 |
| |
residual/recurrent |
grade |
|
type |
type |
| |
|
IV |
| 2 |
Glioblastoma |
WHO |
n/a |
wild |
wild |
R132 |
0.16 |
0.079 |
1.708 |
1.186 |
3.156 |
0.100 |
| |
|
grade |
|
type |
type |
| |
|
IV |
| 3 |
Glioblastoma |
WHO |
n/a |
wild |
wild |
R132 |
0.13 |
0.028 |
0.140 |
0.170 |
0.891 |
0.017 |
| |
|
grade |
|
type |
type |
| |
|
IV |
| 4 |
Oligoastrocytoma |
WHO |
n/a |
wild |
wild |
R132 |
0.21 |
0.016 |
0.553 |
1.061 |
1.731 |
0.089 |
| |
|
grade |
|
type |
type |
| |
|
II |
| 5 |
Glioblastoma |
WHO |
n/a |
mutant |
G364A |
R132H |
16.97 |
0.085 |
1.091 |
0.807 |
1.357 |
0.058 |
| |
|
grade |
| |
|
IV |
| 6 |
Glioblastoma |
WHO |
n/a |
mutant |
G364A |
R132H |
19.42 |
0.023 |
0.462 |
0.590 |
1.966 |
0.073 |
| |
|
grade |
| |
|
IV |
| 7 |
Glioblastoma |
WHO |
n/a |
mutant |
G364A |
R132H |
31.56 |
0.068 |
0.758 |
0.503 |
2.019 |
0.093 |
| |
|
grade |
| |
|
IV |
| 8 |
Oligodendroglioma, |
WHO |
75 |
mutant |
G364A |
R132H |
12.49 |
0.033 |
0.556 |
0.439 |
0.507 |
0.091 |
| |
anaplastic |
grade |
| |
|
III |
| 9 |
Oligodendroglioma, |
WHO |
90 |
mutant |
G364A |
R132H |
4.59 |
0.029 |
1.377 |
1.060 |
1.077 |
0.574 |
| |
anaplastic |
grade |
| |
|
III |
| 10 |
Oligoastrocytoma |
WHO |
n/a |
mutant |
G364A |
R132H |
6.80 |
0.038 |
0.403 |
0.503 |
1.561 |
0.065 |
| |
|
grade |
| |
|
II |
| 11 |
Glioblastoma |
WHO |
n/a |
wild |
wild |
R132 |
0.686 |
0.686 |
0.686 |
0.686 |
0.686 |
0.007 |
| |
|
grade |
|
type |
type |
| |
|
IV |
| 12 |
Glioblastoma |
WHO |
n/a |
mutant |
G364A |
R132H |
18.791 |
18.791 |
18.791 |
18.791 |
18.791 |
0.031 |
| |
|
grade |
| |
|
IV |
| 13 |
Glioblastoma |
WHO |
n/a |
mutant |
G364A |
R132H |
4.59 |
0.029 |
1.377 |
1.060 |
1.077 |
0.043 |
| |
|
grade |
| |
|
IV |
| 14 |
Glioblastoma |
WHO |
n/a |
wild |
wild |
R132 |
0.199 |
0.046 |
0.180 |
0.170 |
0.221 |
0.014 |
| |
|
grade |
|
type |
type |
| |
|
IV |
| 15 |
Glioblastoma |
WHO |
n/a |
mutant |
C363G |
R132G |
13.827 |
0.030 |
0.905 |
0.599 |
1.335 |
0.046 |
| |
|
grade |
| |
|
IV |
| 16 |
Glioblastoma |
WHO |
n/a |
mutant |
G364A |
R132H |
28.364 |
0.068 |
0.535 |
0.488 |
2.105 |
0.054 |
| |
|
grade |
| |
|
IV |
| 17 |
Glioblastoma |
WHO |
n/a |
mutant |
C363A |
R132S |
9.364 |
0.029 |
1.038 |
0.693 |
2.151 |
0.121 |
| |
|
grade |
| |
|
IV |
| 18 |
Glioblastoma |
WHO |
n/a |
wild |
wild |
R132 |
0.540 |
0.031 |
0.468 |
0.608 |
1.490 |
0.102 |
| |
|
grade |
|
type |
type |
| |
|
IV |
| 19 |
Glioma, malignant, |
WHO |
80 |
mutant |
G364A |
R132H |
19.000 |
0.050 |
0.654 |
0.391 |
2.197 |
0.171 |
| |
astrocytoma |
grade |
| |
|
IV |
| 20 |
Oligodendroglioma |
WHO |
80 |
wild |
wild |
R132 |
0.045 |
0.037 |
1.576 |
0.998 |
1.420 |
0.018 |
| |
|
grade |
|
type |
type |
| |
|
III |
| 21 |
Glioma, malignant, |
WHO |
95 |
wild |
wild |
R132 |
0.064 |
0.034 |
0.711 |
0.710 |
2.105 |
0.165 |
| |
astrocytoma |
grade |
|
type |
type |
| |
|
IV |
| 22 |
Glioblastoma |
WHO |
70 |
wild |
wild |
R132 |
0.171 |
0.041 |
2.066 |
1.323 |
0.027 |
0.072 |
| |
|
grade |
|
type |
type |
| |
|
IV |
| |
| indicates data missing or illegible when filed |
-
To determine if 2HG production is characteristic of tumors harboring mutations in IDH1, metabolites were extracted from human malignant gliomas that were either wild-type or mutant for IDH1. It has been suggested that αKG levels are decreased in cells transfected with mutant IDH1 (Zhao, S. et al. Science 324, 261-5 (2009)). The average αKG level from 12 tumor samples harboring various R132 mutations was slightly less than the average αKG level observed in 10 tumors which are wild-type for IDH1. This difference in αKG was not statistically significant, and a range of αKG levels was observed in both wild-type and mutant tumors. In contrast, increased 2HG levels were found in all tumors that contained an R132 IDH1 mutation. All R132 mutant IDH1 tumors examined had between 5 and 35 μmol of 2HG per gram of tumor, while tumors with wild-type IDH1 had over 100 fold less 2HG. This increase in 2HG in R132 mutant tumors was statistically significant (p<0.0001). It was confirmed that (R)-2HG was the isomer present in tumor samples (data not shown). Together these data establish that the novel enzymatic activity associated with R132 mutations in IDH1 results in the production of 2HG in human brain tumors that harbor these mutations.
-
2HG is known to accumulate in the inherited metabolic disorder 2-hydroxyglutaric aciduria. This disease is caused by deficiency in the enzyme 2-hydroxyglutarate dehydrogenase, which converts 2HG to αKG (Struys, E. A. et al. Am J Hum Genet. 76, 358-60 (2005)). Patients with 2-hydroxyglutarate dehydrogenase deficiencies accumulate 2HG in the brain as assessed by MRI and CSF analysis, develop leukoencephalopathy, and have an increased risk of developing brain tumors (Aghili, M., Zahedi, F. & Rafiee, J Neurooncol 91, 233-6 (2009); Kolker, S., Mayatepek, E. & Hoffmann, G. F. Neuropediatrics 33, 225-31 (2002); Wajner, M., Latini, A., Wyse, A. T. & Dutra-Filho, C. S. J Inherit Metab Dis 27, 427-48 (2004)). Furthermore, elevated brain levels of 2HG result in increased ROS levels (Kolker, S. et al. Eur J Neurosci 16, 21-8 (2002); Latini, A. et al. Eur J Neurosci 17, 2017-22 (2003)), potentially contributing to an increased risk of cancer. The ability of 2HG to act as an NMDA receptor agonist may contribute to this effect (Kolker, S. et al. Eur J Neurosci 16, 21-8 (2002)). 2HG may also be toxic to cells by competitively inhibiting glutamate and/or αKG utilizing enzymes. These include transaminases which allow utilization of glutamate nitrogen for amino and nucleic acid biosynthesis, and αKG-dependent prolyl hydroxylases such as those which regulate Hif1α levels. Alterations in Hif1α have been reported to result from mutant IDH1 protein expression (Zhao, S. et al. Science 324, 261-5 (2009)). Regardless of mechanism, it appears likely that the gain-of-function ability of cells to produce 2HG as a result of R132 mutations in IDH1 contributes to tumorigenesis. Patients with 2-hydroxyglutarate dehydrogenase deficiency have a high risk of CNS malignancy (Aghili, M., Zahedi, F. & Rafiee, E. J Neurooncol 91, 233-6 (2009)). The ability of mutant IDH1 to directly act on αKG may explain the prevalence of IDH1 mutations in tumors from CNS tissue, which are unique in their high level of glutamate uptake and its ready conversion to αKG in the cytosol (Tsacopoulos, M. J Physiol Paris 96, 283-8 (2002)), thereby providing high levels of substrate for 2HG production. The apparent co-dominance of the activity of mutant IDH1 with that of the wild-type enzyme is consistent with the genetics of the disease, in which only a single copy of the gene is mutated. As discussed above, the wild-type IDH1 could directly provide NADPH and αKG to the mutant enzyme. These data also demonstrate that mutation of R132 to histidine, serine, cysteine, glycine or leucine share a common ability to catalyze the NADPH-dependent conversion of αKG to 2HG. These findings help clarify why mutations at other amino acid residues of IDH1, including other residues essential for catalytic activity, are not found. Finally, these findings have clinical implications in that they suggest that 2HG production will identify patients with IDH1 mutant brain tumors. This will be important for prognosis as patients with IDH1 mutations live longer than patients with gliomas characterized by other mutations (Parsons, D. W. et al. Science 321, 1807-12 (2008)). In addition, patients with lower grade gliomas may benefit by the therapeutic inhibition of 2HG production. Inhibition of 2HG production by mutant IDH1 might slow or halt conversion of lower grade glioma into lethal secondary glioblastoma, changing the course of the disease.
The Reaction Product of ICDH1 R132H Reduction of α-Kg Inhibits the Oxidative Decarboxylation of Isocitrate by Wild-Type ICDH1.
-
A reaction containing the wild-type ICDH1, NADP, and α-KG was assembled (under conditions as described above) to which was added in a titration series either (R)-2-hydroxyglutarate or the reaction product of the ICDH1 R1321H mutant reduction of α-KG to 2-hydroxyglutarate. The reaction product 2-HG was shown to inhibit the oxidative decarboxylation of isocitrate by the wild-type ICDH1, while the (R)-2-hydroxyglutarate did not show any effect on the rate of the reaction. Since there are only two possible chiral products of the ICDH1 R132H mutant reduction of α-KG to 2-HG, and the (R)-2-HG did not show inhibition in this assay, it follows that the product of the mutant reaction is the (S)-2-HG form. This experiment is presented in FIG. 25.
-
To determine the chirality of the 2HG produced, the products of the R132H reaction was derivatized with diacetyl-L-tartaric anhydride, which allowed separating the (S) and (R) enantiomers of 2HG by simple reverse-phase LC and detecting the products by tandem mass spectrometry (Strays, E. A., Jansen, E. E., Verhoeven, N. M. & Jakobs, C. Clin Chem 50, 1391-5 (2004)) (FIG. 31B). The peaks corresponding to the (S) and (R) isomers of 2HG were confirmed using racemic and R(−)-2HG standards. The reaction product from R132H co-eluted with R(−)-2HG peak, demonstrating that the R(−) stereoisomer is the product produced from αKG by R132H mutant IDH1.
-
The observation that the reaction product of the mutant enzyme is capable of inhibiting a metabolic reaction known to occur in cells suggests that this reaction product might also inhibit other reactions which utilize α-KG, isocitrate, or citrate as substrates or produce them as products in vivo or in vitro.
Example 3
Metabolomics Analysis of IDH1 Wild Type and Mutants
-
Metabolomics research can provide mechanistic basis for why R132 mutations confer survival advantage for GBM patients carrying such mutations.
1. Metabolomics of GBM Tumor Cell Lines: Wild Type Vs R132 Mutants
-
Cell lines with R132 mutations can be identified and profiled. Experiments can be performed in proximal metabolite pool with a broad scope of metabolites.
2. Oxalomalate Treatment of GBM Cell Lines
-
Oxalomalate is a competitive inhibitor of IDH1. Change of NADPH (metabolomics) when IDH1 is inhibited by a small molecule can be examined.
3. Metabolomics of Primary GBM Tumors: Wild Type Vs R132 Mutations
-
Primary tumors with R132 mutations can be identified. Experiments can be performed in proximal metabolite pool with a broad scope of metabolites.
-
4. Detection of 2-Hydroxyglutarate in Cells that Overexpress IDH1 132 Mutants
-
Overexpression of an IDH1 132 mutant in cells may cause an elevated level of 2-hydroxyglutarate and/or a reduced level of alpha-ketoglutarate. One can perform a metabolomic experiment to demonstrate the consequence of this mutation on the cellular metabolite pool.
Example 4
Evaluation of IDH1 as a Cancer Target
-
shRNAmir inducible knockdown can be performed to examine the cellular phenotype and metabolomics profiles. HTS grade IDH1 enzymes are available. The IDH mutations described herein can be used for patient selection.
Example 5
siRNAs
-
IDH1
-
Exemplary siRNAs are presented in the following tables. Art-known methods can be used to select other siRNAs. siRNAs can be evaluated, e.g., by determining the ability of an siRNA to silence an IDH, e.g., IDH1, e.g., in an in vitro system, e.g., in cultured cells, e.g., HeLa cells or cultured glioma cells. siRNAs known in the art for silencing the target can also be used, see, e.g., Silencing of cytosolic NADP+ dependent isoccitrate dehydrogenase by small interfering RNA enhances the sensitivity of HeLa cells toward stauropine, Lee et al., 2009, Free Radical Research, 43: 165-173.
-
The siRNAs in Table 7 (with the exception of entry 1356) were generated using the siRNA selection tool available on the worldwide web at jura.wi.mit.edu/bioc/siRNAext/. (Yuan et al. Nucl. Acids. Res. 2004 32:W130-W134.) Other selection tools can be used as well. Entry 1356 was adapted from Silencing of cytosolic NADP+ dependent isoccitrate dehydrogenase by small interfering RNA enhances the sensitivity of HeLa cells toward stauropine, Lee et al., 2009, Free Radical Research, 43: 165-173.
-
The siRNAs in Tables 7, 8, 9, 10, 11, 12, 13 and 14 represent candidates spanning the IDH1 mRNA at nucleotide positions 628 and 629 according to the sequence at GenBank Accession No. NM—005896.2 (SEQ ID NO:9, FIG. 22).
-
The RNAs in the tables can be modified, e.g., as described herein. Modifications include chemical modifications to enhance properties, e.g., resistance to degradation, or the use of overhangs. For example, either one or both of the sense and antisense strands in the tables can include an additional dinucleotide at the 3′ end, e.g., TT, UU, dTdT.
-
| TABLE 7 |
| |
| siRNAs targeting wildtype IDH1 |
| on mRNA |
|
SEQ |
|
SEQ |
| (FIG. 21B) |
sense (5′ to 3′) |
ID NO: |
antisense (5′ to 3′) |
ID NO: |
| |
| 13 |
GGUUUCUGCAGAGUCUAC |
14 |
AGUAGACUCUGCAGAAAC |
15 |
| |
U |
|
C |
|
| |
| 118 |
CUCUUCGCCAGCAUAUCA |
16 |
AUGAUAUGCUGGCGAAGA |
17 |
| |
U |
|
G |
|
| |
| 140 |
GGCAGGCGAUAAACUACA |
18 |
AUGUAGUUUAUCGCCUGC |
19 |
| |
U |
|
C |
|
| |
| 145 |
GCGAUAAACUACAUUCAG |
20 |
ACUGAAUGUAGUUUAUCG |
21 |
| |
U |
|
C |
|
| |
| 199 |
GAAAUCUAUUCACUGUCA |
22 |
UUGACAGUGAAUAGAUUU |
23 |
| |
A |
|
C |
|
| |
| 257 |
GUUCUGUGGUAGAGAUGC |
24 |
UGCAUCUCUACCACAGAA |
25 |
| |
A |
|
C |
|
| |
| 272 |
GCAAGGAGAUGAAAUGAC |
26 |
UGUCAUUUCAUCUCCUUG |
27 |
| |
A |
|
C |
|
| |
| 277 |
GGAGAUGAAAUGACACGA |
28 |
UUCGUGUCAUUUCAUCUC |
29 |
| |
A |
|
C |
|
| |
| 278 |
GAGAUGAAAUGACACGAA |
30 |
AUUCGUGUCAUUUCAUCU |
31 |
| |
U |
|
C |
|
| |
| 280 |
GAUGAAAUGACACGAAUC |
32 |
UGAUUCGUGUCAUUUCAU |
33 |
| |
A |
|
C |
|
| |
| 292 |
CGAAUCAUUUGGGAAUUG |
34 |
UCAAUUCCCAAAUGAUUC |
35 |
| |
A |
|
G |
|
| |
| 302 |
GGGAAUUGAUUAAAGAGA |
36 |
UUCUCUUUAAUCAAUUCC |
37 |
| |
A |
|
C |
|
| |
| 332 |
CCUACGUGGAAUUGGAUC |
38 |
AGAUCCAAUUCCACGUAG |
39 |
| |
U |
|
G |
|
| |
| 333 |
CUACGUGGAAUUGGAUCU |
40 |
UAGAUCCAAUUCCACGUA |
41 |
| |
A |
|
G |
|
| |
| 345 |
GGAUCUACAUAGCUAUGA |
42 |
AUCAUAGCUAUGUAGAUC |
43 |
| |
U |
|
C |
|
| |
| 356 |
GCUAUGAUUUAGGCAUAG |
44 |
UCUAUGCCUAAAUCAUAG |
45 |
| |
A |
|
C |
|
| |
| 408 |
GGAUGCUGCAGAAGCUAU |
46 |
UAUAGCUUCUGCAGCAUC |
47 |
| |
A |
|
C |
|
| |
| 416 |
CAGAAGCUAUAAAGAAGC |
48 |
UGCUUCUUUAUAGCUUCU |
49 |
| |
A |
|
G |
|
| |
| 418 |
GAAGCUAUAAAGAAGCAU |
50 |
UAUGCUUCUUUAUAGCUU |
51 |
| |
A |
|
C |
|
| |
| 432 |
GCAUAAUGUUGGCGUCAA |
52 |
UUUGACGCCAACAUUAUG |
53 |
| |
A |
|
C |
|
| |
| 467 |
CUGAUGAGAAGAGGGUUG |
54 |
UCAACCCUCUUCUCAUCA |
55 |
| |
A |
|
G |
|
| |
| 481 |
GUUGAGGAGUUCAAGUUG |
56 |
UCAACUUGAACUCCUCAA |
57 |
| |
A |
|
C |
|
| |
| 487 |
GAGUUCAAGUUGAAACAA |
58 |
UUUGUUUCAACUUGAACU |
59 |
| |
A |
|
C |
|
| |
| 495 |
GUUGAAACAAAUGUGGAA |
60 |
UUUCCACAUUUGUUUCAA |
61 |
| |
A |
|
C |
|
| |
| 502 |
CAAAUGUGGAAAUCACCA |
62 |
UUGGUGAUUUCCACAUUU |
63 |
| |
A |
|
G |
|
| |
| 517 |
CCAAAUGGCACCAUACGA |
64 |
UUCGUAUGGUGCCAUUUG |
65 |
| |
A |
|
G |
|
| |
| 528 |
CAUACGAAAUAUUCUGGG |
66 |
ACCCAGAAUAUUUCGUAU |
67 |
| |
U |
|
G |
|
| |
| 560 |
GAGAAGCCAUUAUCUGCA |
68 |
UUGCAGAUAAUGGCUUCU |
69 |
| |
A |
|
C |
|
| |
| 614 |
CUAUCAUCAUAGGUCGUC |
70 |
UGACGACCUAUGAUGAUA |
71 |
| |
A |
|
G |
|
| |
| 618 |
CAUCAUAGGUCGUCAUGC |
72 |
AGCAUGACGACCUAUGAU |
73 |
| |
U |
|
G |
|
| |
| 621 |
CAUAGGUCGUCAUGCUUA |
74 |
AUAAGCAUGACGACCUAU |
75 |
| |
U |
|
G |
|
| |
| 691 |
GAGAUAACCUACACACCA |
76 |
UUGGUGUGUAGGUUAUCU |
77 |
| |
A |
|
C |
|
| |
| 735 |
CCUGGUACAUAACUUUGA |
78 |
UUCAAAGUUAUGUACCAG |
79 |
| |
A |
|
G |
|
| |
| 747 |
CUUUGAAGAAGGUGGUGG |
80 |
ACCACCACCUUCUUCAAA |
81 |
| |
U |
|
G |
|
| |
| 775 |
GGGAUGUAUAAUCAAGAU |
82 |
UAUCUUGAUUAUACAUCC |
83 |
| |
A |
|
C |
|
| |
| 811 |
GCACACAGUUCCUUCCAA |
84 |
UUUGGAAGGAACUGUGUG |
85 |
| |
A |
|
C |
|
| |
| 818 |
GUUCCUUCCAAAUGGCUC |
86 |
AGAGCCAUUUGGAAGGAA |
87 |
| |
U |
|
C |
|
| |
| 844 |
GGUUGGCCUUUGUAUCUG |
88 |
UCAGAUACAAAGGCCAAC |
89 |
| |
A |
|
C |
|
| |
| 851 |
CUUUGUAUCUGAGCACCA |
90 |
UUGGUGCUCAGAUACAAA |
91 |
| |
A |
|
G |
|
| |
| 882 |
GAAGAAAUAUGAUGGGCG |
92 |
ACGCCCAUCAUAUUUCUU |
93 |
| |
U |
|
C |
|
| |
| 942 |
GUCCCAGUUUGAAGCUCA |
94 |
UUGAGCUUCAAACUGGGA |
95 |
| |
A |
|
C |
|
| |
| 968 |
GGUAUGAGCAUAGGCUCA |
96 |
AUGAGCCUAUGCUCAUAC |
97 |
| |
U |
|
C |
|
| |
| 998 |
GGCCCAAGCUAUGAAAUC |
98 |
UGAUUUCAUAGCUUGGGC |
99 |
| |
A |
|
C |
|
| |
| 1001 |
CCCAAGCUAUGAAAUCAG |
100 |
UCUGAUUUCAUAGCUUGG |
101 |
| |
A |
|
G |
|
| |
| 1127 |
CAGAUGGCAAGACAGUAG |
102 |
UCUACUGUCUUGCCAUCU |
103 |
| |
A |
|
G |
|
| |
| 1133 |
GCAAGACAGUAGAAGCAG |
104 |
UCUGCUUCUACUGUCUUG |
105 |
| |
A |
|
C |
|
| |
| 1184 |
GCAUGUACCAGAAAGGAC |
106 |
UGUCCUUUCUGGUACAUG |
107 |
| |
A |
|
C |
|
| |
| 1214 |
CCAAUCCCAUUGCUUCCA |
108 |
AUGGAAGCAAUGGGAUUG |
109 |
| |
U |
|
G |
|
| |
| 1257 |
CCACAGAGCAAAGCUUGA |
110 |
AUCAAGCUUUGCUCUGUG |
111 |
| |
U |
|
G |
|
| |
| 1258 |
CACAGAGCAAAGCUUGAU |
112 |
UAUCAAGCUUUGCUCUGU |
113 |
| |
A |
|
G |
|
| |
| 1262 |
GAGCAAAGCUUGAUAACA |
114 |
UUGUUAUCAAGCUUUGCU |
115 |
| |
A |
|
C |
|
| |
| 1285 |
GAGCUUGCCUUCUUUGCA |
116 |
UUGCAAAGAAGGCAAGCU |
117 |
| |
A |
|
C |
|
| |
| 1296 |
CUUUGCAAAUGCUUUGGA |
118 |
UUCCAAAGCAUUUGCAAA |
119 |
| |
A |
|
G |
|
| |
| 1301 |
CAAAUGCUUUGGAAGAAG |
120 |
ACUUCUUCCAAAGCAUUU |
121 |
| |
U |
|
G |
|
| |
| 1307 |
CUUUGGAAGAAGUCUCUA |
122 |
AUAGAGACUUCUUCCAAA |
123 |
| |
U |
|
G |
|
| |
| 1312 |
GAAGAAGUCUCUAUUGAG |
124 |
UCUCAAUAGAGACUUCUU |
125 |
| |
A |
|
C |
|
| |
| 1315 |
GAAGUCUCUAUUGAGACA |
126 |
UUGUCUCAAUAGAGACUU |
127 |
| |
A |
|
C |
|
| |
| 1356 |
GGACUUGGCUGCUUGCAU |
128 |
AAUGCAAGCAGCCAAGUC |
129 |
| |
U |
|
C |
|
| |
| 1359 |
CUUGGCUGCUUGCAUUAA |
130 |
UUUAAUGCAAGCAGCCAA |
131 |
| |
A |
|
G |
|
| |
| 1371 |
CAUUAAAGGUUUACCCAA |
132 |
AUUGGGUAAACCUUUAAU |
133 |
| |
U |
|
G |
|
| |
| 1385 |
CCAAUGUGCAACGUUCUG |
134 |
UCAGAACGUUGCACAUUG |
135 |
| |
A |
|
G |
|
| |
| 1390 |
GUGCAACGUUCUGACUAC |
136 |
AGUAGUCAGAACGUUGCA |
137 |
| |
U |
|
C |
|
| |
| 1396 |
CGUUCUGACUACUUGAAU |
138 |
UAUUCAAGUAGUCAGAAC |
139 |
| |
A |
|
G |
|
| |
| 1415 |
CAUUUGAGUUCAUGGAUA |
140 |
UUAUCCAUGAACUCAAAU |
141 |
| |
A |
|
G |
|
| |
| 1422 |
GUUCAUGGAUAAACUUGG |
142 |
UCCAAGUUUAUCCAUGAA |
143 |
| |
A |
|
C |
|
| |
| 1425 |
CAUGGAUAAACUUGGAGA |
144 |
UUCUCCAAGUUUAUCCAU |
145 |
| |
A |
|
G |
|
| |
| 1455 |
CAAACUAGCUCAGGCCAA |
146 |
UUUGGCCUGAGCUAGUUU |
147 |
| |
A |
|
G |
|
| |
| 1487 |
CCUGAGCUAAGAAGGAUA |
148 |
UUAUCCUUCUUAGCUCAG |
149 |
| |
A |
|
G |
|
| |
| 1493 |
CUAAGAAGGAUAAUUGUC |
150 |
AGACAAUUAUCCUUCUUA |
151 |
| |
U |
|
G |
|
| |
| 1544 |
CUGUGUUACACUCAAGGA |
152 |
AUCCUUGAGUGUAACACA |
153 |
| |
U |
|
G |
|
| |
| 1546 |
GUGUUACACUCAAGGAUA |
154 |
UUAUCCUUGAGUGUAACA |
155 |
| |
A |
|
C |
|
| |
| 1552 |
CACUCAAGGAUAAAGGCA |
156 |
UUGCCUUUAUCCUUGAGU |
157 |
| |
A |
|
G |
|
| |
| 1581 |
GUAAUUUGUUUAGAAGCC |
158 |
UGGCUUCUAAACAAAUUA |
159 |
| |
A |
|
C |
|
| |
| 1646 |
GUUAUUGCCACCUUUGUG |
160 |
UCACAAAGGUGGCAAUAA |
161 |
| |
A |
|
C |
|
| |
| 1711 |
CAGCCUAGGAAUUCGGUU |
162 |
UAACCGAAUUCCUAGGCU |
163 |
| |
A |
|
G |
|
| |
| 1713 |
GCCUAGGAAUUCGGUUAG |
164 |
ACUAACCGAAUUCCUAGG |
165 |
| |
U |
|
C |
|
| |
| 1714 |
CCUAGGAAUUCGGUUAGU |
166 |
UACUAACCGAAUUCCUAG |
167 |
| |
A |
|
G |
|
| |
| 1718 |
GGAAUUCGGUUAGUACUC |
168 |
UGAGUACUAACCGAAUUC |
169 |
| |
A |
|
C |
|
| |
| 1719 |
GAAUUCGGUUAGUACUCA |
170 |
AUGAGUACUAACCGAAUU |
171 |
| |
U |
|
C |
|
| |
| 1725 |
GGUUAGUACUCAUUUGUA |
172 |
AUACAAAUGAGUACUAAC |
173 |
| |
U |
|
C |
|
| |
| 1730 |
GUACUCAUUUGUAUUCAC |
174 |
AGUGAAUACAAAUGAGUA |
175 |
| |
U |
|
C |
|
| |
| 1804 |
GGUAAAUGAUAGCCACAG |
176 |
ACUGUGGCUAUCAUUUAC |
177 |
| |
U |
|
C |
|
| |
| 1805 |
GUAAAUGAUAGCCACAGU |
178 |
UACUGUGGCUAUCAUUUA |
179 |
| |
A |
|
C |
|
| |
| 1816 |
CCACAGUAUUGCUCCCUA |
180 |
UUAGGGAGCAAUACUGUG |
181 |
| |
A |
|
G |
|
| |
| 1892 |
GGGAAGUUCUGGUGUCAU |
182 |
UAUGACACCAGAACUUCC |
183 |
| |
A |
|
C |
|
| |
| 1897 |
GUUCUGGUGUCAUAGAUA |
184 |
AUAUCUAUGACACCAGAA |
185 |
| |
U |
|
c |
|
| |
| 1934 |
GCUGUGCAUUAAACUUGC |
186 |
UGCAAGUUUAAUGCACAG |
187 |
| |
A |
|
C |
|
| |
| 1937 |
GUGCAUUAAACUUGCACA |
188 |
AUGUGCAAGUUUAAUGCA |
189 |
| |
U |
|
C |
|
| |
| 1939 |
GCAUUAAACUUGCACAUG |
190 |
UCAUGUGCAAGUUUAAUG |
191 |
| |
A |
|
C |
|
| |
| 1953 |
CAUGACUGGAACGAAGUA |
192 |
AUACUUCGUUCCAGUCAU |
193 |
| |
U |
|
G |
|
| |
| 1960 |
GGAACGAAGUAUGAGUGC |
194 |
UGCACUCAUACUUCGUUC |
195 |
| |
A |
|
C |
|
| |
| 1961 |
GAACGAAGUAUGAGUGCA |
196 |
UUGCACUCAUACUUCGUU |
197 |
| |
A |
|
C |
|
| |
| 1972 |
GAGUGCAACUCAAAUGUG |
198 |
ACACAUUUGAGUUGCACU |
199 |
| |
U |
|
C |
|
| |
| 1976 |
GCAACUCAAAUGUGUUGA |
200 |
UUCAACACAUUUGAGUUG |
201 |
| |
A |
|
C |
|
| |
| 1982 |
CAAAUGUGUUGAAGAUAC |
202 |
AGUAUCUUCAACACAUUU |
203 |
| |
U |
|
G |
|
| |
| 1987 |
GUGUUGAAGAUACUGCAG |
204 |
ACUGCAGUAUCUUCAACA |
205 |
| |
U |
|
C |
|
| |
| 1989 |
GUUGAAGAUACUGCAGUC |
206 |
UGACUGCAGUAUCUUCAA |
207 |
| |
A |
|
C |
|
| |
| 2020 |
CCUUGCUGAAUGUUUCCA |
208 |
UUGGAAACAUUCAGCAAG |
209 |
| |
A |
|
G |
|
| |
| 2021 |
CUUGCUGAAUGUUUCCAA |
210 |
AUUGGAAACAUUCAGCAA |
211 |
| |
U |
|
G |
|
| |
| 2024 |
GCUGAAUGUUUCCAAUAG |
212 |
UCUAUUGGAAACAUUCAG |
213 |
| |
A |
|
C |
|
| |
| 2035 |
CCAAUAGACUAAAUACUG |
214 |
ACAGUAUUUAGUCUAUUG |
215 |
| |
U |
|
G |
|
| |
| 2067 |
GAGUUUGGAAUCCGGAAU |
216 |
UAUUCCGGAUUCCAAACU |
217 |
| |
A |
|
C |
|
| |
| 2073 |
GGAAUCCGGAAUAAAUAC |
218 |
AGUAUUUAUUCCGGAUUC |
219 |
| |
U |
|
C |
|
| |
| 2074 |
GAAUCCGGAAUAAAUACU |
220 |
UAGUAUUUAUUCCGGAUU |
221 |
| |
A |
|
C |
|
| |
| 2080 |
GGAAUAAAUACUACCUGG |
222 |
UCCAGGUAGUAUUUAUUC |
223 |
| |
A |
|
C |
|
| |
| 2133 |
GGCCUGGCCUGAAUAUUA |
224 |
AUAAUAUUCAGGCCAGGC |
225 |
| |
U |
|
C |
|
| |
| 2134 |
GCCUGAAUAUUAUACUAC |
226 |
AGUAGUAUAAUAUUCAGG |
227 |
| |
U |
|
C |
|
| |
| 2136 |
CUGGCCUGAAUAUUAUAC |
228 |
AGUAUAAUAUUCAGGCCA |
229 |
| |
U |
|
G |
|
| |
| 2166 |
CAUAUUUCAUCCAAGUGC |
230 |
UGCACUUGGAUGAAAUAU |
231 |
| |
A |
|
G |
|
| |
| 2180 |
GUGCAAUAAUGUAAGCUG |
232 |
UCAGCUUACAUUAUUGCA |
233 |
| |
A |
|
C |
|
| |
| 2182 |
GCAAUAAUGUAAGCUGAA |
234 |
AUUCAGCUUACAUUAUUG |
235 |
| |
U |
|
C |
|
| |
| 2272 |
CACUAUCUUAUCUUCUCC |
236 |
AGGAGAAGAUAAGAUAGU |
237 |
| |
U |
|
G |
|
| |
| 2283 |
CUUCUCCUGAACUGUUGA |
238 |
AUCAACAGUUCAGGAGAA |
239 |
| |
U |
|
G |
| |
-
| TABLE 8 |
| |
| siRNAs targeting wildtype IDH1 |
| Position |
|
|
|
|
| on mRNA |
|
SEQ |
|
SEQ |
| (FIG. 21B) |
sense (5′ to 3′) |
ID NO: |
antisense (5′ to 3′) |
ID NO: |
| |
| 611 |
AACCUAUCAUCAUAGGUC |
240 |
CGACCUAUGAUGAUAGGU |
241 |
| |
G |
|
U |
|
| |
| 612 |
ACCUAUCAUCAUAGGUCG |
242 |
ACGACCUAUGAUGAUAGG |
243 |
| |
U |
|
U |
|
| |
| 613 |
CCUAUCAUCAUAGGUCGU |
244 |
GACGACCUAUGAUGAUAG |
245 |
| |
C |
|
G |
|
| |
| 614 |
CUAUCAUCAUAGGUCGUC |
246 |
UGACGACCUAUGAUGAUA |
247 |
| |
A |
|
G |
|
| |
| 615 |
UAUCAUCAUAGGUCGUCA |
248 |
AUGACGACCUAUGAUGAU |
249 |
| |
U |
|
A |
|
| |
| 616 |
AUCAUCAUAGGUCGUCAU |
250 |
CAUGACGACCUAUGAUGA |
251 |
| |
G |
|
U |
|
| |
| 617 |
UCAUCAUAGGUCGUCAUG |
252 |
GCAUGACGACCUAUGAUG |
253 |
| |
C |
|
A |
|
| |
| 618 |
CAUCAUAGGUCGUCAUGC |
254 |
AGCAUGACGACCUAUGAU |
255 |
| |
U |
|
G |
|
| |
| 619 |
AUCAUAGGUCGUCAUGCU |
256 |
AAGCAUGACGACCUAUGA |
257 |
| |
U |
|
U |
|
| |
| 620 |
UCAUAGGUCGUCAUGCUU |
258 |
UAAGCAUGACGACCUAUG |
259 |
| |
A |
|
A |
|
| |
| 621 |
CAUAGGUCGUCAUGCUUA |
260 |
AUAAGCAUGACGACCUAU |
261 |
| |
U |
|
G |
|
| |
| 622 |
AUAGGUCGUCAUGCUUAU |
262 |
CAUAAGCAUGACGACCUA |
263 |
| |
G |
|
U |
|
| |
| 623 |
UAGGUCGUCAUGCUUAUG |
264 |
CCAUAAGCAUGACGACCU |
265 |
| |
G |
|
A |
|
| |
| 624 |
AGGUCGUCAUGCUUAUGG |
266 |
CCCAUAAGCAUGACGACC |
267 |
| |
G |
|
U |
|
| |
| 625 |
GGUCGUCAUGCUUAUGGG |
268 |
CCCCAUAAGCAUGACGAC |
269 |
| |
G |
|
C |
|
| |
| 626 |
GUCGUCAUGCUUAUGGGG |
270 |
UCCCAUAAGCAUGACGAC |
271 |
| |
A |
|
C |
|
| |
| 627 |
UCGUCAUGCUUAUGGGGA |
272 |
AUCCCAUAAGCAUGACGA |
273 |
| |
U |
|
C |
| |
-
| TABLE 9 |
| |
| siRNAs targeting G395A mutant IDH1 (SEQ ID NO: 5) |
| (equivalent to G629A of SEQ ID NO: 9 (FIG. 21B)) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 21B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 611 |
AACCUAUCAUCAUAGG |
274 |
UGACCUAUGAUGAUAG |
275 |
| |
UCA |
|
GUU |
|
| |
| 612 |
ACCUAUCAUCAUAGGU |
276 |
AUGACCUAUGAUGAUA |
277 |
| |
CAU |
|
GGU |
|
| |
| 613 |
CCUAUCAUCAUAGGUC |
278 |
GAUGACCUAUGAUGAU |
279 |
| |
AUC |
|
AGG |
|
| |
| 614 |
CUAUCAUCAUAGGUCA |
280 |
UGAUGACCUAUGAUGA |
281 |
| |
UCA |
|
UAG |
|
| |
| 615 |
UAUCAUCAUAGGUCAU |
282 |
AUGAUGACCUAUGAUG |
283 |
| |
CAU |
|
AUA |
|
| |
| 616 |
AUCAUCAUAGGUCAUC |
284 |
CAUGAUGACCUAUGAU |
285 |
| |
AUG |
|
GAU |
|
| |
| 617 |
UCAUCAUAGGUCAUCA |
286 |
GCAUGAUGACCUAUGA |
287 |
| |
UGC |
|
UGA |
|
| |
| 618 |
CAUCAUAGGUCAUCAU |
288 |
AGCAUGAUGACCUAUG |
289 |
| |
GCU |
|
AUG |
|
| |
| 619 |
AUCAUAGGUCAUCAUG |
290 |
AAGCAUGAUGACCUAU |
291 |
| |
CUU |
|
GAU |
|
| |
| 620 |
UCAUAGGUCAUCAUGC |
292 |
UAAGCAUGAUGACCUA |
293 |
| |
UUA |
|
UGA |
|
| |
| 621 |
CAUAGGUCAUCAUGCU |
294 |
AUAAGCAUGAUGACCU |
295 |
| |
UAU |
|
AUG |
|
| |
| 622 |
AUAGGUCAUCAUGCUU |
296 |
CAUAAGCAUGAUGACC |
297 |
| |
AUG |
|
UAU |
|
| |
| 623 |
UAGGUCAUCAUGCUUA |
298 |
CCAUAAGCAUGAUGAC |
299 |
| |
UGG |
|
CUA |
|
| |
| 624 |
AGGUCAUCAUGCUUAU |
300 |
CCCAUAAGCAUGAUGA |
301 |
| |
GGG |
|
CCU |
|
| |
| 625 |
GGUCAUCAUGCUUAUG |
302 |
CCCCAUAAGCAUGAUG |
303 |
| |
GGG |
|
ACC |
|
| |
| 626 |
GUCAUCAUGCUUAUGG |
304 |
UCCCCAUAAGCAUGAU |
305 |
| |
GGA |
|
GAC |
|
| |
| 627 |
UCAUCAUGCUUAUGGG |
306 |
AUCCCCAUAAGCAUGA |
307 |
| |
GAU |
|
UGA |
| |
-
| TABLE 10 |
| |
| siRNAs targeting C394A mutant IDH1 (SEQ ID NO: 5) |
| (equivalent to C628A of SEQ ID NO: 9 (FIG. 21B)) |
| (Arg132Ser (SEQ ID NO: 8)) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 21B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 611 |
AACCUAUCAUCAUAGG |
308 |
CUACCUAUGAUGAUAG |
309 |
| |
UAG |
|
GUU |
|
| |
| 612 |
ACCUAUCAUCAUAGGU |
310 |
ACUACCUAUGAUGAUA |
311 |
| |
AGU |
|
GGU |
|
| |
| 613 |
CCUAUCAUCAUAGGUA |
312 |
GACUACCUAUGAUGAU |
313 |
| |
GUC |
|
AGG |
|
| |
| 614 |
CUAUCAUCAUAGGUAG |
314 |
UGACUACCUAUGAUGA |
315 |
| |
UCA |
|
UAG |
|
| |
| 615 |
UAUCAUCAUAGGUAGU |
316 |
AUGACUACCUAUGAUG |
317 |
| |
CAU |
|
AUA |
|
| |
| 616 |
AUCAUCAUAGGUAGUC |
318 |
CAUGACUACCUAUGAU |
319 |
| |
AUG |
|
GAU |
|
| |
| 617 |
UCAUCAUAGGUAGUCA |
320 |
GCAUGACUACCUAUGA |
321 |
| |
UGC |
|
UGA |
|
| |
| 618 |
CAUCAUAGGUAGUCAU |
322 |
AGCAUGACUACCUAUG |
323 |
| |
GCU |
|
AUG |
|
| |
| 619 |
AUCAUAGGUAGUCAUG |
324 |
AAGCAUGACUACCUAU |
325 |
| |
CUU |
|
GAU |
|
| |
| 620 |
UCAUAGGUAGUCAUGC |
326 |
UAAGCAUGACUACCUA |
327 |
| |
UUA |
|
UGA |
|
| |
| 621 |
CAUAGGUAGUCAUGCU |
328 |
AUAAGCAUGACUACCU |
329 |
| |
UAU |
|
AUG | |
| |
| 622 |
AUAGGUAGUCAUGCUU |
| |
330 |
CAUAAGCAUGACUACC |
331 |
| |
AUG |
|
UAU |
|
| |
| 623 |
UAGGUAGUCAUGCUUA |
332 |
CCAUAAGCAUGACUAC |
333 |
| |
UGG |
|
CUA |
|
| |
| 624 |
AGGUAGUCAUGCUUAU |
334 |
CCCAUAAGCAUGACUA |
335 |
| |
GGG |
|
CCU |
|
| |
| 625 |
GGUAGUCAUGCUUAUG |
336 |
CCCCAUAAGCAUGACU |
337 |
| |
GGG |
|
ACC |
|
| |
| 626 |
GUAGUCAUGCUUAUGG |
338 |
UCCCCAUAAGCAUGAC |
339 |
| |
GGA |
|
UAC |
|
| |
| 627 |
UAGUCAUGCUUAUGGG |
340 |
AUCCCCAUAAGCAUGA |
341 |
| |
GAU |
|
CUA |
| |
-
| TABLE 11 |
| |
| siRNAs targeting C394U mutant IDH1 (SEQ ID NO: 5) |
| (equivalent to C628U of SEQ ID NO: 9 (FIG. 21B)) |
| (Arg132Cys (SEQ ID NO: 8)) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 21B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 611 |
AACCUAUCAUCAUAGG |
342 |
CAACCUAUGAUGAUAG |
343 |
| |
UUG |
|
GUU |
|
| |
| 612 |
ACCUAUCAUCAUAGGU |
344 |
ACAACCUAUGAUGAUA |
345 |
| |
UGU |
|
GGU |
|
| |
| 613 |
CCUAUCAUCAUAGGUU |
346 |
GACAACCUAUGAUGAU |
347 |
| |
GUC |
|
AGG |
|
| |
| 614 |
CUAUCAUCAUAGGUUG |
348 |
UGACAACCUAUGAUGA |
349 |
| |
UCA |
|
UAG |
|
| |
| 615 |
UAUCAUCAUAGGUUGU |
350 |
AUGACAACCUAUGAUG |
351 |
| |
CAU |
|
AUA |
|
| |
| 616 |
AUCAUCAUAGGUUGUC |
352 |
CAUGACAACCUAUGAU |
353 |
| |
AUG |
|
GAU |
|
| |
| 617 |
UCAUCAUAGGUUGUCA |
354 |
GCAUGACAACCUAUGA |
355 |
| |
UGC |
|
UGA |
|
| |
| 618 |
CAUCAUAGGUUGUCAU |
356 |
AGCAUGACAACCUAUG |
357 |
| |
GCU |
|
AUG |
|
| |
| 619 |
AUCAUAGGUUGUCAUG |
358 |
AAGCAUGACAACCUAU |
359 |
| |
CUU |
|
GAU |
|
| |
| 620 |
UCAUAGGUUGUCAUGC | 360 |
UAAGCAUGACAACCUA | |
361 |
| |
UUA |
|
UGA |
|
| |
| 621 |
CAUAGGUUGUCAUGCU |
362 |
AUAAGCAUGACAACCU |
363 |
| |
UAU |
|
AUG |
|
| |
| 622 |
AUAGGUUGUCAUGCUU |
364 |
CAUAAGCAUGACAACC |
365 |
| |
AUG |
|
UAU |
|
| |
| 623 |
UAGGUUGUCAUGCUUA |
366 |
CCAUAAGCAUGACAAC |
367 |
| |
UGG |
|
CUA |
|
| |
| 624 |
AGGUUGUCAUGCUUAU |
368 |
CCCAUAAGCAUGACAA |
369 |
| |
GGG |
|
CCU |
|
| |
| 625 |
GGUUGUCAUGCUUAUG |
370 |
CCCCAUAAGCAUGACA |
371 |
| |
GGG |
|
ACC |
|
| |
| 626 |
GUUGUCAUGCUUAUGG |
372 |
UCCCCAUAAGCAUGAC |
373 |
| |
GGA |
|
AAC |
|
| |
| 627 |
UUGUCAUGCUUAUGGG |
374 |
AUCCCCAUAAGCAUGA |
375 |
| |
GAU |
|
CAA |
| |
-
| TABLE 12 |
| |
| siRNAs targeting C394G mutant IDH1 (SEQ ID NO: 5) |
| (equivalent to C628G of SEQ ID NO: 9 (FIG. 21B)) |
| (Arg132Gly (SEQ ID NO: 8)) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 21B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 611 |
AACCUAUCAUCAUAGG |
376 |
CCACCUAUGAUGAUAG |
377 |
| |
UGG |
|
GUU |
|
| |
| 612 |
ACCUAUCAUCAUAGGU |
378 |
ACCACCUAUGAUGAUA |
379 |
| |
GGU |
|
GGU |
|
| |
| 613 |
CCUAUCAUCAUAGGUG |
380 |
GACCACCUAUGAUGAU |
381 |
| |
GUC |
|
AGG |
|
| |
| 614 |
CUAUCAUCAUAGGUGG |
382 |
UGACCACCUAUGAUGA |
383 |
| |
UCA |
|
UAG |
|
| |
| 615 |
UAUCAUCAUAGGUGGU |
384 |
AUGACCACCUAUGAUG |
385 |
| |
CAU |
|
AUA |
|
| |
| 616 |
AUCAUCAUAGGUGGUC |
386 |
CAUGACCACCUAUGAU |
387 |
| |
AUG |
|
GAU |
|
| |
| 617 |
UCAUCAUAGGUGGUCA |
388 |
GCAUGACCACCUAUGA |
389 |
| |
UGC |
|
UGA |
|
| |
| 618 |
CAUCAUAGGUGGUCAU |
390 |
AGCAUGACCACCUAUG |
391 |
| |
GCU |
|
AUG |
|
| |
| 619 |
AUCAUAGGUGGUCAUG |
392 |
AAGCAUGACCACCUAU |
393 |
| |
CUU |
|
GAU |
|
| |
| 620 |
UCAUAGGUGGUCAUGC |
394 |
UAAGCAUGACCACCUA |
395 |
| |
UUA |
|
UGA |
|
| |
| 621 |
CAUAGGUGGUCAUGCU |
396 |
AUAAGCAUGACCACCU |
397 |
| |
UAU |
|
AUG |
|
| |
| 622 |
AUAGGUGGUCAUGCUU |
398 |
CAUAAGCAUGACCACC |
399 |
| |
AUG |
|
UAU |
|
| |
| 623 |
UAGGUGGUCAUGCUUA |
400 |
CCAUAAGCAUGACCAC |
401 |
| |
UGG |
|
CUA |
|
| |
| 624 |
AGGUUGUCAUGCUUAU |
402 |
CCCAUAAGCAUGACCA |
403 |
| |
GGG |
|
CCU |
|
| |
| 625 |
GGUUGUCAUGCUUAUG |
404 |
CCCCAUAAGCAUGACC |
405 |
| |
GGG |
|
ACC |
|
| |
| 626 |
GUUGUCAUGCUUAUGG |
406 |
UCCCCAUAAGCAUGAC |
407 |
| |
GGA |
|
CAC |
|
| |
| 627 |
UUGUCAUGCUUAUGGG |
408 |
AUCCCCAUAAGCAUGA |
409 |
| |
GAU |
|
CCA |
| |
-
| TABLE 13 |
| |
| siRNAs targeting G395C mutant IDH1 (SEQ ID NO: 5) |
| (equivalent to G629C of SEQ ID NO: 9 (FIG. 21B)) |
| (Arg132Pro (SEQ ID NO: 8)) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 21B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 611 |
AACCUAUCAUCAUAGG |
410 |
CGACCUAUGAUGAUAG |
411 |
| |
UCG |
|
GUU |
|
| |
| 612 |
ACCUAUCAUCAUAGGU |
412 |
ACGACCUAUGAUGAUA |
413 |
| |
CGU |
|
GGU |
|
| |
| 613 |
CCUAUCAUCAUAGGUC |
414 |
GACGACCUAUGAUGAU |
415 |
| |
GUC |
|
AGG |
|
| |
| 614 |
CUAUCAUCAUAGGUCG |
416 |
UGACGACCUAUGAUGA |
417 |
| |
UCA |
|
UAG |
|
| |
| 615 |
UAUCAUCAUAGGUCGU |
418 |
AUGACGACCUAUGAUG |
419 |
| |
CAU |
|
AUA |
|
| |
| 616 |
AUCAUCAUAGGUCGUC |
420 |
CAUGACGACCUAUGAU |
421 |
| |
AUG |
|
GAU |
|
| |
| 617 |
UCAUCAUAGGUCGUCA |
422 |
GCAUGACGACCUAUGA |
423 |
| |
UGC |
|
UGA |
|
| |
| 618 |
CAUCAUAGGUCGUCAU |
424 |
AGCAUGACGACCUAUG |
425 |
| |
GCU |
|
AUG |
|
| |
| 619 |
AUCAUAGGUCGUCAUG |
426 |
AAGCAUGACGACCUAU |
427 |
| |
CUU |
|
GAU |
|
| |
| 620 |
UCAUAGGUCGUCAUGC |
428 |
UAAGCAUGACGACCUA |
429 |
| |
UUA |
|
UGA |
|
| |
| 621 |
CAUAGGUCGUCAUGCU |
430 |
AUAAGCAUGACGACCU |
431 |
| |
UAU |
|
AUG |
|
| |
| 622 |
AUAGGUCGUCAUGCUU |
432 |
CAUAAGCAUGACGACC |
433 |
| |
AUG |
|
UAU |
|
| |
| 623 |
UAGGUCGUCAUGCUUA |
434 |
CCAUAAGCAUGACGAC |
435 |
| |
UGG |
|
CUA |
|
| |
| 624 |
AGGUCGUCAUGCUUAU |
436 |
CCCAUAAGCAUGACGA |
437 |
| |
GGG |
|
CCU |
|
| |
| 625 |
GGUCGUCAUGCUUAUG |
438 |
CCCCAUAAGCAUGACG |
439 |
| |
GGG |
|
ACC |
|
| |
| 626 |
GUCGUCAUGCUUAUGG |
440 |
UCCCCAUAAGCAUGAC |
441 |
| |
GGA |
|
GAC |
|
| |
| 627 |
UCGUCAUGCUUAUGGG |
442 |
AUCCCCAUAAGCAUGA |
443 |
| |
GAU |
|
CGA |
| |
-
| TABLE 14 |
| |
| siRNAs targeting G395U mutant IDH1 (SEQ ID NO: 5) |
| (equivalent to G629U of SEQ ID NO: 9 (FIG. 21B)) |
| (Arg132Leu (SEQ ID NO: 8)) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 21B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 611 |
AACCUAUCAUCAUAGG |
444 |
AGACCUAUGAUGAUAG |
445 |
| |
UCU |
|
GUU |
|
| |
| 612 |
ACCUAUCAUCAUAGGU |
446 |
AAGACCUAUGAUGAUA |
447 |
| |
CUU |
|
GGU |
|
| |
| 613 |
CCUAUCAUCAUAGGUC |
448 |
GAAGACCUAUGAUGAU |
449 |
| |
UUC |
|
AGG |
|
| |
| 614 |
CUAUCAUCAUAGGUCU |
450 |
UGAAGACCUAUGAUGA |
451 |
| |
UCA |
|
UAG |
|
| |
| 615 |
UAUCAUCAUAGGUCUU |
452 |
AUGAAGACCUAUGAUG |
453 |
| |
CAU |
|
AUA |
|
| |
| 616 |
AUCAUCAUAGGUCUUC |
454 |
CAUGAAGACCUAUGAU |
455 |
| |
AUG |
|
GAU |
|
| |
| 617 |
UCAUCAUAGGUCUUCA |
456 |
GCAUGAAGACCUAUGA |
457 |
| |
UGC |
|
UGA |
|
| |
| 618 |
CAUCAUAGGUCUUCAU |
458 |
AGCAUGAAGACCUAUG |
459 |
| |
GCU |
|
AUG |
|
| |
| 619 |
AUCAUAGGUCUUCAUG |
460 |
AAGCAUGAAGACCUAU |
461 |
| |
CUU |
|
GAU |
|
| |
| 620 |
UCAUAGGUCUUCAUGC |
462 |
UAAGCAUGAAGACCUA |
463 |
| |
UUA |
|
UGA |
|
| |
| 621 |
CAUAGGUCUUCAUGCU |
464 |
AUAAGCAUGAAGACCU |
465 |
| |
UAU |
|
AUG |
|
| |
| 622 |
AUAGGUCUUCAUGCUU |
466 |
CAUAAGCAUGAAGACC |
467 |
| |
AUG |
|
UAU |
|
| |
| 623 |
UAGGUCUUCAUGCUUA |
468 |
CCAUAAGCAUGAAGAC |
469 |
| |
UGG |
|
CUA |
|
| |
| 624 |
AGGUCUUCAUGCUUAU |
470 |
CCCAUAAGCAUGAAGA |
471 |
| |
GGG |
|
CCU |
|
| |
| 625 |
GGUCUUCAUGCUUAUG |
472 |
CCCCAUAAGCAUGAAG |
473 |
| |
GGG |
|
ACC |
|
| |
| 626 |
GUCUUCAUGCUUAUGG |
474 |
UCCCCAUAAGCAUGAA |
475 |
| |
GGA |
|
GAC |
|
| |
| 627 |
UCUUCAUGCUUAUGGG |
476 |
AUCCCCAUAAGCAUGA |
477 |
| |
GAU |
|
AGA |
| |
IDH2
-
Exemplary siRNAs are presented in the following tables. Art-known methods can be used to select other siRNAs. siRNAs can be evaluated, e.g., by determining the ability of an siRNA to silence an e.g., IDH2, e.g., in an in vitro system, e.g., in cultured cells, e.g., HeLa cells or cultured glioma cells. e.g.,
-
The siRNAs in Table 15 were generated using the siRNA selection tool available on the worldwide web at jura.wi.mit.edu/bioc/siRNAext/. (Yuan et al. Nucl. Acids. Res. 2004 32:W130-W134.) Other selection tools can be used as well. Entry 1356 was adapted from Silencing of cytosolic NADP+ dependent isoccitrate dehydrogenase by small interfering RNA enhances the sensitivity of HeLa cells toward stauropine, Lee et al., 2009, Free Radical Research, 43: 165-173.
-
The siRNAs in Tables 16-23 represent candidates spanning the IDH2 mRNA at nucleotide positions 600, 601, and 602 according to the mRNA sequence presented at GenBank Accession No. NM—002168.2 (Record dated Aug. 16, 2009; GI28178831) (SEQ ID N012, FIG. 22B; equivalent to nucleotide positions 514, 515, and 516 of the cDNA sequence represented by SEQ ID NO:11, FIG. FIG. 22A).
-
The RNAs in the tables can be modified, e.g., as described herein. Modifications include chemical modifications to enhance properties, e.g., resistance to degradation, or the use of overhangs. For example, either one or both of the sense and antisense strands in the tables can include an additional dinucleotide at the 3′ end, e.g., TT, UU, dTdT.
-
| TABLE 15 |
| |
| siRNAs targeting wildtype IDH2 |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 250 |
GUGAUGAGAUGACCCG |
478 |
AUACGGGUCAUCUCAU |
479 |
| |
UAU |
|
CAC |
|
| |
| 252 |
GAUGAGAUGACCCGUA |
480 |
UAAUACGGGUCAUCUC |
481 |
| |
UUA |
|
AUC |
|
| |
| 264 |
CGUAUUAUCUGGCAGU |
482 |
UGAACUGCCAGAUAAU |
483 |
| |
UCA |
|
ACG |
|
| |
| 274 |
GGCAGUUCAUCAAGGA |
484 |
UUCUCCUUGAUGAACU |
485 |
| |
GAA |
|
GCC |
|
| |
| 451 |
GUGUGGAAGAGUUCAA |
486 |
AGCUUGAACUCUUCCA |
487 |
| |
GCU |
|
CAC |
|
| |
| 453 |
GUGGAAGAGUUCAAGC |
488 |
UCAGCUUGAACUCUUC |
489 |
| |
UGA |
|
CAC |
|
| |
| 456 |
GAAGAGUUCAAGCUGA |
490 |
UCUUCAGCUUGAACUC |
491 |
| |
AGA |
|
UUC |
|
| |
| 795 |
CAGUAUGCCAUCCAGA |
492 |
UCUUCUGGAUGGCAUA |
493 |
| |
AGA |
|
CUG |
|
| |
| 822 |
CUGUACAUGAGCACCA |
494 |
UCUUGGUGCUCAUGUA |
495 |
| |
AGA |
|
CAG |
|
| |
| 832 |
GCACCAAGAACACCAU |
496 |
AGUAUGGUGUUCUUGG |
497 |
| |
ACU |
|
UGC |
|
| |
| 844 |
CCAUACUGAAAGCCUA |
498 |
UCGUAGGCUUUCAGUA |
499 |
| |
CGA |
|
UGG |
|
| |
| 845 |
CAUACUGAAAGCCUAC |
500 |
AUCGUAGGCUUUCAGU |
501 |
| |
GAU |
|
AUG |
|
| |
| 868 |
GUUUCAAGGACAUCUU |
502 |
UGGAAGAUGUCCUUGA |
503 |
| |
CCA |
|
AAC |
|
| |
| 913 |
CCGACUUCGACAAGAA |
504 |
UUAUUCUUGUCGAAGU |
505 |
| |
UAA |
|
CGG |
|
| |
| 915 |
GACUUCGACAAGAAUA |
506 |
UCUUAUUCUUGUCGAA |
507 |
| |
AGA |
|
GUC |
|
| |
| 921 |
GACAAGAAUAAGAUCU |
508 |
ACCAGAUCUUAUUCUU |
509 |
| |
GGU |
|
GUC |
|
| |
| 949 |
GGCUCAUUGAUGACAU |
510 |
ACCAUGUCAUCAAUGA |
511 |
| |
GGU |
|
GCC |
|
| |
| 1009 |
GCAAGAACUAUGACGG |
512 |
UCUCCGUCAUAGUUCU |
513 |
| |
AGA |
|
UGC |
|
| |
| 1010 |
CAAGAACUAUGACGGA |
514 |
AUCUCCGUCAUAGUUC |
515 |
| |
GAU |
|
UUG |
|
| |
| 1024 |
GAGAUGUGCAGUCAGA |
516 |
AUGUCUGACUGCACAU |
517 |
| |
CAU |
|
CUC |
|
| |
| 1096 |
CUGAUGGGAAGACGAU |
518 |
UCAAUCGUCUUCCCAU |
519 |
| |
UGA |
|
CAG |
|
| |
| 1354 |
GCAAUGUGAAGCUGAA |
520 |
UCGUUCAGCUUCACAU |
521 |
| |
CGA |
|
UGC |
|
| |
| 1668 |
CUGUAAUUUAUAUUGC |
522 |
AGGGCAAUAUAAAUUA |
523 |
| |
CCU |
|
CAG |
|
| |
| 1694 |
CAUGGUGCCAUAUUUA |
524 |
AGCUAAAUAUGGCACC |
525 |
| |
GCU |
|
AUG |
|
| |
| 1697 |
GGUGCCAUAUUUAGCU |
526 |
AGUAGCUAAAUAUGGC |
527 |
| |
ACU |
|
ACC |
|
| |
| 1698 |
GUGCCAUAUUUAGCUA |
528 |
UAGUAGCUAAAUAUGG |
529 |
| |
CUA |
|
CAC |
|
| |
| 1700 |
GCCAUAUUUAGCUACU |
530 |
UUUAGUAGCUAAAUAU |
531 |
| |
AAA |
|
GGC |
| |
-
| TABLE 16 |
| |
| siRNAs targeting wildtype IDH2 |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 584 |
GCCCAUCACCAUUGGC |
532 |
CCUGCCAAUGGUGAUG |
533 |
| |
AGG |
|
GGC |
|
| |
| 585 |
CCCAUCACCAUUGGCA |
534 |
GCCUGCCAAUGGUGAU |
535 |
| |
GGC |
|
GGG |
|
| |
| 586 |
CCAUCACCAUUGGCAG |
536 |
UGCCUGCCAAUGGUGA |
537 |
| |
GCA |
|
UGG |
|
| |
| 587 |
CAUCACCAUUGGCAGG |
538 |
GUGCCUGCCAAUGGUG |
539 |
| |
CAC |
|
AUG |
|
| |
| 588 |
AUCACCAUUGGCAGGC |
540 |
CGUGCCUGCCAAUGGU |
541 |
| |
ACG |
|
GAU |
|
| |
| 589 |
UCACCAUUGGCAGGCA |
542 |
GCGUGCCUGCCAAUGG |
543 |
| |
CGC |
|
UGA |
|
| |
| 590 |
CACCAUUGGCAGGCAC |
544 |
GGCGUGCCUGCCAAUG |
545 |
| |
GCC |
|
GUG |
|
| |
| 591 |
ACCAUUGGCAGGCACG |
546 |
GGGCGUGCCUGCCAAU |
547 |
| |
CCC |
|
GGU |
|
| |
| 592 |
CCAUUGGCAGGCACGC |
548 |
UGGGCGUGCCUGCCAA |
549 |
| |
CCA |
|
UGG |
|
| |
| 593 |
CAUUGGCAGGCACGCC |
550 |
AUGGGCGUGCCUGCCA |
551 |
| |
CAU |
|
AUG |
|
| |
| 594 |
AUUGGCAGGCACGCCC |
552 |
CAUGGGCGUGCCUGCC |
553 |
| |
AUG |
|
AAU |
|
| |
| 595 |
UUGGCAGGCACGCCCA |
554 |
CCAUGGGCGUGCCUGC |
555 |
| |
UGG |
|
CAA |
|
| |
| 596 |
UGGCAGGCACGCCCAU |
556 |
GCCAUGGGCGUGCCUG |
557 |
| |
GGC |
|
CCA |
|
| |
| 597 |
GGCAGGCACGCCCAUG |
558 |
CGCCAUGGGCGUGCCU |
559 |
| |
GCG |
|
GCC |
|
| |
| 598 |
GCAGGCACGCCCAUGG |
560 |
UCGCCAUGGGCGUGCC |
561 |
| |
CGA |
|
UGC |
|
| |
| 599 |
CAGGCACGCCCAUGGC |
562 |
GUCGCCAUGGGCGUGC |
563 |
| |
GAC |
|
CUG |
|
| |
| 600 |
AGGCACGCCCAUGGCG |
564 |
GGUCGCCAUGGGCGUG |
565 |
| |
ACC |
|
CCU |
| |
-
| TABLE 17 |
| |
| siRNAs targeting A514G mutant IDH2 (equivalent |
| to A600G of SEQ ID NO: 12, (FIG. 22B) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 584 |
GCCCAUCACCAUUGGC |
566 |
CCCGCCAAUGGUGAUG |
567 |
| |
GGG |
|
GGC |
|
| |
| 585 |
CCCAUCACCAUUGGCG |
568 |
GCCCGCCAAUGGUGAU |
569 |
| |
GGC |
|
GGG |
|
| |
| 586 |
CCAUCACCAUUGGCGG |
570 |
UGCCCGCCAAUGGUGA |
571 |
| |
GCA |
|
UGG |
|
| |
| 587 |
CAUCACCAUUGGCGGG |
572 |
GUGCCCGCCAAUGGUG |
573 |
| |
CAC |
|
AUG |
|
| |
| 588 |
AUCACCAUUGGCGGGC |
574 |
CGUGCCCGCCAAUGGU |
575 |
| |
ACG |
|
GAU |
|
| |
| 589 |
UCACCAUUGGCGGGCA |
576 |
GCGUGCCCGCCAAUGG |
577 |
| |
CGC |
|
UGA |
|
| |
| 590 |
CACCAUUGGCGGGCAC |
578 |
GGCGUGCCCGCCAAUG |
579 |
| |
GCC |
|
GUG |
|
| |
| 591 |
ACCAUUGGCGGGCACG |
580 |
GGGCGUGCCCGCCAAU |
581 |
| |
CCC |
|
GGU |
|
| |
| 592 |
CCAUUGGCGGGCACGC |
582 |
UGGGCGUGCCCGCCAA |
583 |
| |
CCA |
|
UGG |
|
| |
| 593 |
CAUUGGCGGGCACGCC |
584 |
AUGGGCGUGCCCGCCA |
585 |
| |
CAU |
|
AUG |
|
| |
| 594 |
AUUGGCGGGCACGCCC |
586 |
CAUGGGCGUGCCCGCC |
587 |
| |
AUG |
|
AAU |
|
| |
| 595 |
UUGGCGGGCACGCCCA |
588 |
CCAUGGGCGUGCCCGC |
589 |
| |
UGG |
|
CAA |
|
| |
| 596 |
UGGCGGGCACGCCCAU |
590 |
GCCAUGGGCGUGCCCG |
591 |
| |
GGC |
|
CCA |
|
| |
| 597 |
GGCGGGCACGCCCAUG |
592 |
CGCCAUGGGCGUGCCC |
593 |
| |
GCG |
|
GCC |
|
| |
| 598 |
GCGGGCACGCCCAUGG |
594 |
UCGCCAUGGGCGUGCC |
595 |
| |
CGA |
|
CGC |
|
| |
| 599 |
CGGGCACGCCCAUGGC |
596 |
GUCGCCAUGGGCGUGC |
597 |
| |
GAC |
|
CCG |
|
| |
| 600 |
GGGCACGCCCAUGGCG |
598 |
GGUCGCCAUGGGCGUG |
599 |
| |
ACC |
|
CCC |
| |
-
| TABLE 18 |
| |
| siRNAs targeting A514U mutant IDH2 (equivalent |
| to A600U of SEQ ID NO: 12, (FIG. 22B) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 584 |
GCCCAUCACCAUUGGC |
600 |
CCAGCCAAUGGUGAUG |
601 |
| |
UGG |
|
GGC |
|
| |
| 585 |
CCCAUCACCAUUGGCU |
602 |
GCCAGCCAAUGGUGAU |
603 |
| |
GGC |
|
GGG |
|
| |
| 586 |
CCAUCACCAUUGGCUG |
604 |
UGCCAGCCAAUGGUGA |
605 |
| |
GCA |
|
UGG |
|
| |
| 587 |
CAUCACCAUUGGCUGG |
606 |
GUGCCAGCCAAUGGUG |
607 |
| |
CAC |
|
AUG |
|
| |
| 588 |
AUCACCAUUGGCUGGC |
608 |
CGUGCCAGCCAAUGGU |
609 |
| |
ACG |
|
GAU |
|
| |
| 589 |
UCACCAUUGGCUGGCA |
610 |
GCGUGCCAGCCAAUGG |
611 |
| |
CGC |
|
UGA |
|
| |
| 590 |
CACCAUUGGCUGGCAC |
612 |
GGCGUGCCAGCCAAUG |
613 |
| |
GCC |
|
GUG |
|
| |
| 591 |
ACCAUUGGCUGGCACG |
614 |
GGGCGUGCCAGCCAAU |
615 |
| |
CCC |
|
GGU |
|
| |
| 592 |
CCAUUGGCUGGCACGC |
616 |
UGGGCGUGCCAGCCAA |
617 |
| |
CCA |
|
UGG |
|
| |
| 593 |
CAUUGGCUGGCACGCC |
618 |
AUGGGCGUGCCAGCCA |
619 |
| |
CAU |
|
AUG |
|
| |
| 594 |
AUUGGCUGGCACGCCC |
620 |
CAUGGGCGUGCCAGCC |
621 |
| |
AUG |
|
AAU |
|
| |
| 595 |
UUGGCUGGCACGCCCA |
622 |
CCAUGGGCGUGCCAGC |
623 |
| |
UGG |
|
CAA |
|
| |
| 596 |
UGGCUGGCACGCCCAU |
624 |
GCCAUGGGCGUGCCAG |
625 |
| |
GGC |
|
CCA |
|
| |
| 597 |
GGCUGGCACGCCCAUG |
626 |
CGCCAUGGGCGUGCCA |
627 |
| |
GCG |
|
GCC |
|
| |
| 598 |
GCUGGCACGCCCAUGG |
628 |
UCGCCAUGGGCGUGCC |
629 |
| |
CGA |
|
AGC |
|
| |
| 599 |
CUGGCACGCCCAUGGC |
630 |
GUCGCCAUGGGCGUGC |
631 |
| |
GAC |
|
CAG |
|
| |
| 600 |
UGGCACGCCCAUGGCG |
632 |
GGUCGCCAUGGGCGUG |
633 |
| |
ACC |
|
CCA |
| |
-
| TABLE 19 |
| |
| siRNAs targeting G515A mutant IDH2 (equivalent |
| to G601A of SEQ ID NO: 12, (FIG. 22B) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 584 |
GCCCAUCACCAUUGGC |
634 |
CUUGCCAAUGGUGAUG |
635 |
| |
AAG |
|
GGC |
|
| |
| 585 |
CCCAUCACCAUUGGCA |
636 |
GCUUGCCAAUGGUGAU |
637 |
| |
AGC |
|
GGG |
|
| |
| 586 |
CCAUCACCAUUGGCAA |
638 |
UGCUUGCCAAUGGUGA |
639 |
| |
GCA |
|
UGG |
|
| |
| 587 |
CAUCACCAUUGGCAAG |
640 |
GUGCUUGCCAAUGGUG |
641 |
| |
CAC |
|
AUG |
|
| |
| 588 |
AUCACCAUUGGCAAGC |
642 |
CGUGCUUGCCAAUGGU |
643 |
| |
ACG |
|
GAU |
|
| |
| 589 |
UCACCAUUGGCAAGCA |
644 |
GCGUGCUUGCCAAUGG |
645 |
| |
CGC |
|
UGA |
|
| |
| 590 |
CACCAUUGGCAAGCAC |
646 |
GGCGUGCUUGCCAAUG |
647 |
| |
GCC |
|
GUG |
|
| |
| 591 |
ACCAUUGGCAAGCACG |
648 |
GGGCGUGCUUGCCAAU |
649 |
| |
CCC |
|
GGU |
|
| |
| 592 |
CCAUUGGCAAGCACGC |
650 |
UGGGCGUGCUUGCCAA |
651 |
| |
CCA |
|
UGG |
|
| |
| 593 |
CAUUGGCAAGCACGCC |
652 |
AUGGGCGUGCUUGCCA |
653 |
| |
CAU |
|
AUG |
|
| |
| 594 |
AUUGGCAAGCACGCCC |
654 |
CAUGGGCGUGCUUGCC |
655 |
| |
AUG |
|
AAU |
|
| |
| 595 |
UUGGCAAGCACGCCCA |
656 |
CCAUGGGCGUGCUUGC |
657 |
| |
UGG |
|
CAA |
|
| |
| 596 |
UGGCAAGCACGCCCAU |
658 |
GCCAUGGGCGUGCUUG |
659 |
| |
GGC |
|
CCA |
|
| |
| 597 |
GGCAAGCACGCCCAUG |
660 |
CGCCAUGGGCGUGCUU |
661 |
| |
GCG |
|
GCC |
|
| |
| 598 |
GCAAGCACGCCCAUGG |
662 |
UCGCCAUGGGCGUGCU |
663 |
| |
CGA |
|
UGC |
|
| |
| 599 |
CAAGCACGCCCAUGGC |
664 |
GUCGCCAUGGGCGUGC |
665 |
| |
GAC |
|
UUG |
|
| |
| 600 |
AAGCACGCCCAUGGCG |
666 |
GGUCGCCAUGGGCGUG |
667 |
| |
ACC |
|
CUU |
| |
-
| TABLE 20 |
| |
| siRNAs targeting G515C mutant IDH2 (equivalent |
| to G601C of SEQ ID NO: 12, (FIG. 22B) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 584 |
GCCCAUCACCAUUGGC |
668 |
CGUGCCAAUGGUGAUG |
669 |
| |
ACG |
|
GGC |
|
| |
| 585 |
CCCAUCACCAUUGGCA |
670 |
GCGUGCCAAUGGUGAU |
671 |
| |
CGC |
|
GGG |
|
| |
| 586 |
CCAUCACCAUUGGCAC |
672 |
UGCGUGCCAAUGGUGA |
673 |
| |
GCA |
|
UGG |
|
| |
| 587 |
CAUCACCAUUGGCACG |
674 |
GUGCGUGCCAAUGGUG |
675 |
| |
CAC |
|
AUG |
|
| |
| 588 |
AUCACCAUUGGCACGC |
676 |
CGUGCGUGCCAAUGGU |
677 |
| |
ACG |
|
GAU |
|
| |
| 589 |
UCACCAUUGGCACGCA |
678 |
GCGUGCGUGCCAAUGG |
679 |
| |
CGC |
|
UGA |
|
| |
| 590 |
CACCAUUGGCACGCAC |
680 |
GGCGUGCGUGCCAAUG |
681 |
| |
GCC |
|
GUG |
|
| |
| 591 |
ACCAUUGGCACGCACG |
682 |
GGGCGUGCGUGCCAAU |
683 |
| |
CCC |
|
GGU |
|
| |
| 592 |
CCAUUGGCACGCACGC |
684 |
UGGGCGUGCGUGCCAA |
685 |
| |
CCA |
|
UGG |
|
| |
| 593 |
CAUUGGCACGCACGCC |
686 |
AUGGGCGUGCGUGCCA |
687 |
| |
CAU |
|
AUG |
|
| |
| 594 |
AUUGGCACGCACGCCC |
688 |
CAUGGGCGUGCGUGCC |
689 |
| |
AUG |
|
AAU |
|
| |
| 595 |
UUGGCACGCACGCCCA |
690 |
CCAUGGGCGUGCGUGC |
691 |
| |
UGG |
|
CAA |
|
| |
| 596 |
UGGCACGCACGCCCAU |
692 |
GCCAUGGGCGUGCGUG |
693 |
| |
GGC |
|
CCA |
|
| |
| 597 |
GGCACGCACGCCCAUG |
694 |
CGCCAUGGGCGUGCGU |
695 |
| |
GCG |
|
GCC |
|
| |
| 598 |
GCACGCACGCCCAUGG |
696 |
UCGCCAUGGGCGUGCG |
697 |
| |
CGA |
|
UGC |
|
| |
| 599 |
CACGCACGCCCAUGGC |
698 |
GUCGCCAUGGGCGUGC |
699 |
| |
GAC |
|
GUG |
|
| |
| 600 |
ACGCACGCCCAUGGCG |
700 |
GGUCGCCAUGGGCGUG |
701 |
| |
ACC |
|
CGU |
| |
-
| TABLE 21 |
| |
| siRNAs targeting G515U mutant IDH2 (equivalent |
| to G601U of SEQ ID NO: 12, (FIG. 22B) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 584 |
GCCCAUCACCAUUGGC |
702 |
CAUGCCAAUGGUGAUG |
703 |
| |
AUG |
|
GGC |
|
| |
| 585 |
CCCAUCACCAUUGGCA |
704 |
GCAUGCCAAUGGUGAU |
705 |
| |
UGC |
|
GGG |
|
| |
| 586 |
CCAUCACCAUUGGCAU |
706 |
UGCAUGCCAAUGGUGA |
707 |
| |
GCA |
|
UGG |
|
| |
| 587 |
CAUCACCAUUGGCAUG |
708 |
GUGCAUGCCAAUGGUG |
709 |
| |
CAC |
|
AUG |
|
| |
| 588 |
AUCACCAUUGGCAUGC |
710 |
CGUGCAUGCCAAUGGU |
711 |
| |
ACG |
|
GAU |
|
| |
| 589 |
UCACCAUUGGCAUGCA |
712 |
GCGUGCAUGCCAAUGG |
713 |
| |
CGC |
|
UGA |
|
| |
| 590 |
CACCAUUGGCAUGCAC |
714 |
GGCGUGCAUGCCAAUG |
715 |
| |
GCC |
|
GUG |
|
| |
| 591 |
ACCAUUGGCAUGCACG |
716 |
GGGCGUGCAUGCCAAU |
717 |
| |
CCC |
|
GGU |
|
| |
| 592 |
CCAUUGGCAUGCACGC |
718 |
UGGGCGUGCAUGCCAA |
719 |
| |
CCA |
|
UGG |
|
| |
| 593 |
CAUUGGCAUGCACGCC |
720 |
AUGGGCGUGCAUGCCA |
721 |
| |
CAU |
|
AUG |
|
| |
| 594 |
AUUGGCAUGCACGCCC |
722 |
CAUGGGCGUGCAUGCC |
723 |
| |
AUG |
|
AAU |
|
| |
| 595 |
UUGGCAUGCACGCCCA |
724 |
CCAUGGGCGUGCAUGC |
725 |
| |
UGG |
|
CAA |
|
| |
| 596 |
UGGCAUGCACGCCCAU |
726 |
GCCAUGGGCGUGCAUG |
727 |
| |
GGC |
|
CCA |
|
| |
| 597 |
GGCAUGCACGCCCAUG |
728 |
CGCCAUGGGCGUGCAU |
729 |
| |
GCG |
|
GCC |
|
| |
| 598 |
GCAUGCACGCCCAUGG |
730 |
UCGCCAUGGGCGUGCA |
731 |
| |
CGA |
|
UGC |
|
| |
| 599 |
CAUGCACGCCCAUGGC |
732 |
GUCGCCAUGGGCGUGC |
733 |
| |
GAC |
|
AUG |
|
| |
| 600 |
AUGCACGCCCAUGGCG |
734 |
GGUCGCCAUGGGCGUG |
735 |
| |
ACC |
|
CAU |
| |
-
| TABLE 22 |
| |
| siRNAs targeting G516C mutant IDH2 (equivalent |
| to G602C of SEQ ID NO: 12, (FIG. 22B) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 584 |
GCCCAUCACCAUUGGC |
736 |
GCUGCCAAUGGUGAUG |
737 |
| |
AGC |
|
GGC |
|
| |
| 585 |
CCCAUCACCAUUGGCA |
738 |
GGCUGCCAAUGGUGAU |
739 |
| |
GCC |
|
GGG |
|
| |
| 586 |
CCAUCACCAUUGGCAG |
740 |
UGGCUGCCAAUGGUGA |
741 |
| |
CCA |
|
UGG |
|
| |
| 587 |
CAUCACCAUUGGCAGC |
742 |
GUGGCUGCCAAUGGUG |
743 |
| |
CAC |
|
AUG |
|
| |
| 588 |
AUCACCAUUGGCAGCC |
744 |
CGUGGCUGCCAAUGGU |
745 |
| |
ACG |
|
GAU |
|
| |
| 589 |
UCACCAUUGGCAGCCA |
746 |
GCGUGGCUGCCAAUGG |
747 |
| |
CGC |
|
UGA |
|
| |
| 590 |
CACCAUUGGCAGCCAC |
748 |
GGCGUGGCUGCCAAUG |
749 |
| |
GCC |
|
GUG |
|
| |
| 591 |
ACCAUUGGCAGCCACG |
750 |
GGGCGUGGCUGCCAAU |
751 |
| |
CCC |
|
GGU |
|
| |
| 592 |
CCAUUGGCAGCCACGC |
752 |
UGGGCGUGGCUGCCAA |
753 |
| |
CCA |
|
UGG |
|
| |
| 593 |
CAUUGGCAGCCACGCC |
754 |
AUGGGCGUGGCUGCCA |
755 |
| |
CAU |
|
AUG |
|
| |
| 594 |
AUUGGCAGCCACGCCC |
756 |
CAUGGGCGUGGCUGCC |
757 |
| |
AUG |
|
AAU |
|
| |
| 595 |
UUGGCAGCCACGCCCA |
758 |
CCAUGGGCGUGGCUGC |
759 |
| |
UGG |
|
CAA |
|
| |
| 596 |
UGGCAGCCACGCCCAU |
760 |
GCCAUGGGCGUGGCUG |
761 |
| |
GGC |
|
CCA |
|
| |
| 597 |
GGCAGCCACGCCCAUG |
762 |
CGCCAUGGGCGUGGCU |
763 |
| |
GCG |
|
GCC |
|
| |
| 598 |
GCAGCCACGCCCAUGG |
764 |
UCGCCAUGGGCGUGGC |
765 |
| |
CGA |
|
UGC |
|
| |
| 599 |
CAGCCACGCCCAUGGC |
766 |
GUCGCCAUGGGCGUGG |
767 |
| |
GAC |
|
CUG |
|
| |
| 600 |
AGCCACGCCCAUGGCG |
768 |
GGUCGCCAUGGGCGUG |
769 |
| |
ACC |
|
GCU |
| |
-
| TABLE 23 |
| |
| siRNAs targeting G516U mutant IDH2 (equivalent |
| to G602U of SEQ ID NO: 12, (FIG. 22B) |
| Position |
|
SEQ |
|
SEQ |
| on mRNA |
sense |
ID |
antisense |
ID |
| (FIG. 22B) |
(5′ to 3′) |
NO: |
(5′ to 3′) |
NO: |
| |
| 584 |
GCCCAUCACCAUUGGC |
770 |
ACUGCCAAUGGUGAUG |
771 |
| |
AGU |
|
GGC |
|
| |
| 585 |
CCCAUCACCAUUGGCA |
772 |
GACUGCCAAUGGUGAU |
773 |
| |
GUC |
|
GGG |
|
| |
| 586 |
CCAUCACCAUUGGCAG |
774 |
UGACUGCCAAUGGUGA |
775 |
| |
UCA |
|
UGG |
|
| |
| 587 |
CAUCACCAUUGGCAGU |
776 |
GUGACUGCCAAUGGUG |
777 |
| |
CAC |
|
AUG |
|
| |
| 588 |
AUCACCAUUGGCAGUC |
778 |
CGUGACUGCCAAUGGU |
779 |
| |
ACG |
|
GAU |
|
| |
| 589 |
UCACCAUUGGCAGUCA |
780 |
GCGUGACUGCCAAUGG |
781 |
| |
CGC |
|
UGA |
|
| |
| 590 |
CACCAUUGGCAGUCAC |
782 |
GGCGUGACUGCCAAUG |
783 |
| |
GCC |
|
GUG |
|
| |
| 591 |
ACCAUUGGCAGUCACG |
784 |
GGGCGUGACUGCCAAU |
785 |
| |
CCC |
|
GGU |
|
| |
| 592 |
CCAUUGGCAGUCACGC |
786 |
UGGGCGUGACUGCCAA |
787 |
| |
CCA |
|
UGG |
|
| |
| 593 |
CAUUGGCAGUCACGCC |
788 |
AUGGGCGUGACUGCCA |
789 |
| |
CAU |
|
AUG |
|
| |
| 594 |
AUUGGCAGUCACGCCC |
790 |
CAUGGGCGUGACUGCC |
791 |
| |
AUG |
|
AAU |
|
| |
| 595 |
UUGGCAGUCACGCCCA |
792 |
CCAUGGGCGUGACUGC |
793 |
| |
UGG |
|
CAA |
|
| |
| 596 |
UGGCAGUCACGCCCAU |
794 |
GCCAUGGGCGUGACUG |
795 |
| |
GGC |
|
CCA |
|
| |
| 597 |
GGCAGUCACGCCCAUG |
796 |
CGCCAUGGGCGUGACU |
797 |
| |
GCG |
|
GCC |
|
| |
| 598 |
GCAGUCACGCCCAUGG |
798 |
UCGCCAUGGGCGUGAC |
799 |
| |
CGA |
|
UGC |
|
| |
| 599 |
CAGUCACGCCCAUGGC |
800 |
GUCGCCAUGGGCGUGA |
801 |
| |
GAC |
|
CUG |
|
| |
| 600 |
AGUCACGCCCAUGGCG |
802 |
GGUCGCCAUGGGCGUG |
803 |
| |
ACC |
|
ACU |
| |
Example 6
Structural Analysis of R132H Mutant IDH1
-
To define how R132 mutations alter the enzymatic properties of IDH1, the crystal structure of R132H mutant IDH1 bound to αKG, NADPH, and Ca2+ was solved at 2.1 Å resolution.
-
The overall quaternary structure of the homodimeric R132H mutant enzyme adopts the same closed catalytically competent conformation (shown as a monomer in FIG. 29A) that has been previously described for the wild-type enzyme (Xu, X. et al. J Biol Chem 279, 33946-57 (2004)). NADPH is positioned as expected for hydride transfer to αKG in an orientation that would produce R(−)-2HG, consistent with our chiral determination of the 2HG product.
-
Two important features were noted by the change of R132 to histidine: the effect on catalytic conformation equilibrium and the reorganization of the active-site. Locating atop a n-sheet in the relatively rigid small domain, R132 acts as a gate-keeper residue and appears to orchestrate the hinge movement between the open and closed conformations. The guanidinium moiety of R132 swings from the open to the closed conformation with a distance of nearly 8 Å. Substitution of histidine for arginine is likely to change the equilibrium in favor of the closed conformation that forms the catalytic cleft for cofactor and substrate to bind efficiently, which partly explains the high-affinity for NADPH exhibited by the R132H mutant enzyme. This feature may be advantageous for the NADPH-dependent reduction of αKG to R(−)-2HG in an environment where NADPH concentrations are low. Secondly, closer examination of the catalytic pocket of the mutant IDH1 structure in comparison to the wild-type enzyme showed not only the expected loss of key salt-bridge interactions between the guanidinium of R132 and the α/β carboxylates of isocitrate, as well as changes in the network that coordinates the metal ion, but also an unexpected reorganization of the active-site. Mutation to histidine resulted in a significant shift in position of the highly conserved residues Y139 from the A subunit and K212′ from the B subunit (FIG. 29B), both of which are thought to be critical for catalysis of this enzyme family (Aktas, D. F. & Cook, P. F. Biochemistry 48, 3565-77 (2009)). In particular, the hydroxyl moiety of Y139 now occupies the space of the β-carboxylate of isocitrate. In addition, a significant repositioning of αKG compared to isocitrate where the distal carboxylate of αKG now points upward to make new contacts with N96 and S94 was observed. Overall, this single R132 mutation results in formation of a distinct active site compared to wild-type IDH1.
Example 7
Materials and Methods
Summary
-
R132H, R132C, R132L and R132S mutations were introduced into human IDH1 by standard molecular biology techniques. 293T and the human glioblastoma cell lines U87MG and LN-18 were cultured in DMEM, 10% fetal bovine serum. Cells were transfected and selected using standard techniques. Protein expression levels were determined by Western blot analysis using IDHc antibody (Santa Cruz Biotechnology), IDH1 antibody (proteintech), MYC tag antibody (Cell Signaling Technology), and IDH2 antibody (Abeam). Metabolites were extracted from cultured cells and from tissue samples according to close variants of a previously reported method (Lu, W., Kimball, E. & Rabinowitz, J. D. J Am Soc Mass Spectrom 17, 37-50 (2006)), using 80% aqueous methanol (−80° C.) and either tissue scraping or homogenization to disrupt cells. Enzymatic activity in cell lysates was assessed by following a change in NADPH fluorescence over time in the presence of isocitrate and NADP, or αKG and NADPH. For enzyme assays using recombinant IDH1 enzyme, proteins were produced in E. coli and purified using Ni affinity chromatography followed by Sephacryl S-200 size-exclusion chromatography. Enzymatic activity for recombinant IDH1 protein was assessed by following a change in NADPH UV absorbance at 340 nm using a stop-flow spectrophotometer in the presence of isocitrate and NADP or αKG and NADPH. Chirality of 2HG was determined as described previously (Struys, E. A., Jansen, E. E., Verhoeven, N. M. & Jakobs, C. Clin Chem 50, 1391-5 (2004)). For crystallography studies, purified recombinant IDH1 (R132H) at 10 mg/mL in 20 mM Tris pH 7.4, 100 mM NaCl was pre-incubated for 60 min with 10 mM NADPH, 10 mM calcium chloride, and 75 mM αKG. Crystals were obtained at 20° C. by vapor diffusion equilibration using 3 μL drops mixed 2:1 (protein:precipitant) against a well-solution of 100 mM MES pH 6.5, 20% PEG 6000. Patient tumor samples were obtained after informed consent as part of a UCLA IRB-approved research protocol. Brain tumor samples were obtained after surgical resection, snap frozen in isopentane cooled by liquid nitrogen and stored at −80 C. The IDH1 mutation status of each sample was determined using standard molecular biology techniques as described previously (Yan, H. et al. N Engl J Med 360, 765-73 (2009)). Metabolites were extracted and analyzed by LC-MS/MS as described above. Full methods are available in the supplementary material.
Supplementary Methods
-
Cloning, Expression, and Purification of ICDH1 wt and mutants in E. coli. The open reading frame (ORF) clone of human isocitrate dehydrogenase 1 (cDNA) (IDH1; ref. ID NM—005896) was purchased from Invitrogen in pENTR221 (Carlsbad, Calif.) and Origene Inc. in pCMV6 (Rockville, Md.). To transfect cells with wild-type or mutant IDH1, standard molecular biology mutagenesis techniques were utilized to alter the DNA sequence at base pair 395 of the ORF in pCMV6 to introduce base pair change from guanine to adenine, which resulted in a change in the amino acid code at position 132 from arginine (wt) to histidine (mutant; or R132H), and confirmed by standard DNA sequencing methods. For 293T cell transfection, wild-type and R132H mutant IDH1 were subcloned into pCMV-Sport6 with or without a carboxy-terminal Myc-DDK-tag. For stable cell line generation, constructs in pCMV6 were used. For expression in E. coli, the coding region was amplified from pENTR221 by PCR using primers designed to add NDEI and XHO1 restrictions sites at the 5′ and 3′ ends respectively. The resultant fragment was cloned into vector pET41a (EMD Biosciences, Madison, Wis.) to enable the E. coli expression of C-terminus His8-tagged protein. Site directed mutagenesis was performed on the pET41a-ICHD1 plasmid using the QuikChange® MultiSite-Directed Mutagenesis Kit (Stratagene, La Jolla, Calif.) to change G395 to A, resulting in the Arg to His mutation. R132C, R132L and R132S mutants were introduced into pET41a-ICHD1 in an analogous way.
-
Wild-type and mutant proteins were expressed in and purified from the E. coli Rosetta™ strain (Invitrogen, Carlsbad, Calif.) as follows. Cells were grown in LB (20 μg/ml Kanamycin) at 37° C. with shaking until OD600 reaches 0.6. The temperature was changed to 18° C. and protein expression was induced by adding IPTG to final concentration of 1 mM. After 12-16 hours of IPTG induction, cells were resuspended in Lysis Buffer (20 mM Tris, pH7.4, 0.1% Triton X-100, 500 mM NaCl, 1 mM PMSF, 5 mM β-mercaptoethanol, 10% glycerol) and disrupted by microfluidation. The 20,000 g supernatant was loaded on metal chelate affinity resin (MCAC) equilibrated with Nickel Column Buffer A (20 mM Tris, pH7.4, 500 mM NaCl, 5 mM β-mercaptoethanol, 10% glycerol) and washed for 20 column volumes. Elution from the column was effected by a 20 column-volume linear gradient of 10% to 100% Nickel Column Buffer B (20 mM Tris, pH7.4, 500 mM NaCl, 5 mM β-mercaptoethanol, 500 mM Imidazole, 10% glycerol) in Nickel Column Buffer A). Fractions containing the protein of interest were identified by SDS-PAGE, pooled, and dialyzed twice against a 200-volume excess of Gel Filtration Buffer (200 mM NaCl, 50 mM Tris 7.5, 5 mM β-mercaptoethanol, 2 mM MnSO4, 10% glycerol), then concentrated to 10 ml using Centricon (Millipore, Billerica, Mass.) centrifugal concentrators. Purification of active dimers was achieved by applying the concentrated eluent from the MCAC column to a Sephacryl S-200 (GE Life Sciences, Piscataway, N.J.) column equilibrated with Gel Filtration Buffer and eluting the column with 20 column volumes of the same buffer. Fractions corresponding to the retention time of the dimeric protein were identified by SDS-PAGE and pooled for storage at −80° C.
-
Cell lines and Cell Culture. 293T cells were cultured in DMEM (Dulbecco's modified Eagles Medium) with 10% fetal bovine serum and were transfected using pCMV-6-based IDH-1 constructs in six-well plates with Fugene 6 (Roche) or Lipofectamine 2000 (Invitrogen) according to manufacturer's instructions. Parental vector pCMV6 (no insert), pCMV6-wt IDH1 or pCMV6-R132H were transfected into human glioblastoma cell lines (U87MG; LN-18 (ATCC, HTB-14 and CRL-2610; respectively) cultured in DMEM with 10% fetal bovine serum. Approximately 24 hrs after transfection, the cell cultures were transitioned to medium containing G418 sodium salt at concentrations of either 500 ug/ml (U87MG) or 750 ug/ml (LN-18) to select stable transfectants. Pooled populations of G418 resistant cells were generated and expression of either wild-type IDH1 or R132 IDH1 was confirmed by standard Western blot analysis.
-
Western blot. For transient transfection experiments in 293 cells, cells were lysed 72 hours after transfection with standard RIPA buffer. Lysates were separated by SDS-PAGE, transferred to nitrocellulose and probed with goat-anti-IDHc antibody (Santa Cruz Biotechnology sc49996) or rabbit-anti-MYC tag antibody (Cell Signaling Technology #2278) and then detected with HRP-conjugated donkey anti-goat or HRP-conjugated goat-anti-rabbit antibody (Santa Cruz Biotechnology sc2004). IDH1 antibody to confirm expression of both wild-type and R132H IDH1 was obtained from Proteintech. The IDH2 mouse monoclonal antibody used was obtained from Abcam.
-
Detection of isocitrate, αKG, and 2HG in purified enzyme reactions by LC-MS/MS. Enzyme reactions performed as described in the text were run to completion as judged by measurement of the oxidation state of NADPH at 340 nm. Reactions were extracted with eight volumes of methanol, and centrifuged to remove precipitated protein. The supernatant was dried under a stream of nitrogen and resuspended in H2O. Analysis was conducted on an API2000 LC-MS/MS (Applied Biosystems, Foster City, Calif.). Sample separation and analysis was performed on a 150×2 mm, 4 uM Synergi Hydro-RP 80 A column, using a gradient of Buffer A (10 mM tributylamine, 15 mM acetic acid, 3% (v/v) methanol, in water) and Buffer B (methanol) using MRM transitions.
-
Cell lysates based enzyme assays. 293T cell lysates for measuring enzymatic activity were obtained 48 hours after transfection with M-PER lysis buffer supplemented with protease and phosphatase inhibitors. After lysates were sonicated and centrifuged at 12,000 g, supernatants were collected and normalized for total protein concentration. To measure IDH oxidative activity, 3 μg of lysate protein was added to 200 μl of an assay solution containing 33 mM Tris-acetate buffer (pH 7.4), 1.3 mM MgCl2, 0.33 mM EDTA, 100 μM β-NADP, and varying concentrations of D-(+)-threo-isocitrate. Absorbance at 340 nm, reflecting NADPH production, was measured every 20 seconds for 30 min on a SpectraMax 190 spectrophotometer (Molecular Devices). Data points represent the mean activity of 3 replicates per lysate, averaged among 5 time points centered at every 5 min. To measure IDH reductive activity, 3 μg of lysate protein was added to 200 μl of an assay solution which contained 33 mM Tris-acetate (pH 7.4), 1.3 mM MgCl2, 25 μM β-NADPH, 40 mM NaHCO3, and 0.6 mM αKG. The decrease in 340 nm absorbance over time was measured to assess NADPH consumption, with 3 replicates per lysate.
-
Recombinant IDH1 Enzyme Assays. All reactions were performed in standard enzyme reaction buffer (150 mM NaCl, 20 mM Tris-Cl, pH 7.5, 10% glycerol, 5 mM MgCl2 and 0.03% (w/v) bovine serum albumin). For determination of kinetic parameters, sufficient enzyme was added to give a linear reaction for 1 to 5 seconds. Reaction progress was monitored by observation of the reduction state of the cofactor at 340 nm in an SFM-400 stopped-flow spectrophotometer (BioLogic, Knoxyille, Tenn.). Enzymatic constants were determined using curve fitting algorithms to standard kinetic models with the Sigmaplot software package (Systat Software, San Jose, Calif.).
-
Determination of chirality of reaction products from enzyme reactions and tumors. Enzyme reactions were run to completion and extracted with methanol as described above, then derivatized with enantiomerically pure tartaric acid before resolution and analysis by LC-MS/MS. After being thoroughly dried, samples were resuspended in freshly prepared 50 mg/ml (2R,3R)-(+)-Tartaric acid in dichloromethane:acetic acid (4:1) and incubated for 30 minutes at 75° C. After cooling to room temperature, samples were briefly centrifuged at 14,000 g, dried under a stream of nitrogen, and resuspended in H 20. Analysis was conducted on an API200 LC-MS/MS (Applied Biosystems, Foster City, Calif.), using an isocratic flow of 90:10 (2 mM ammonium formate, pH 3.6:MeOH) on a Luna C18(2) 150×2 mm, 5 uM column. Tartaric-acid derivatized 2HG was detected using the 362.9/146.6 MRM transition and the following instrument settings: DP-1, FP-310, EP-4, CE-12, CXP-26. Analysis of the (R)-2HG standard, 2HG racemic mixture, and methanol-extracted tumor biomass (q.v.) was similarly performed.
-
Crystallography conditions. Crystals were obtained at 20° C. by vapor diffusion equilibration using 3 μL drops mixed 2:1 (protein:precipitant) against a well-solution of 100 mM MES pH 6.5, 20% PEG 6000.
-
Protein characterization. Approximately 90 mg of human cytosolic isocitrate dehydrogenase (HcIDH) was supplied to Xtal BioStructures by Agios. This protein was an engineered mutant form, R132S, with an 11-residue C-terminal affinity-purification tag (sequence SLEHHHHHHHH). The calculated monomeric molecular weight was 48.0 kDa and the theoretical pI was 6.50. The protein, at about 6 mg/mL concentration, was stored in 1-mL aliquots in 50 mM Tris-HCl (pH 7.4), 500 mM NaCl, 5 mM β-mercaptoethanol and 10% glycerol at −80° C. As shown in FIG. 32A, SDS-PAGE was performed to test protein purity and an anti-histidine Western blot was done to demonstrate the protein was indeed his-tagged. A sample of the protein was injected into an FPLC size-exclusion column to evaluate the sample purity and to determine the polymeric state in solution. FIG. 32B is a chromatogram of this run showing a single peak running at an estimated 87.6 kDa, suggesting IDH exists as a dimer at pH 7.4. Prior to crystallization, the protein was exchanged into 20 mM Tris-HCl (pH 7.4) and 100 mM NaCl using Amicon centrifugal concentrators. At this time, the protein was also concentrated to approximately 15 mg/mL. At this protein concentration and ionic strength, the protein tended to form a detectable level of precipitate. After spinning out the precipitate, the solution was stable at ˜10 mg/mL at 4° C.
-
Initial attempts at crystallization. The strategy for obtaining diffraction-quality crystals was derived from literature conditions, specifically “Structures of Human Cytosolic NADP-dependent Isocitrate Dehydrogenase Reveal a Novel Self-regulatory Mechanism of Activity,” Xu, et al. (2005) J. Biol. Chem. 279: 33946-56. In this study, two crystal forms of HcIDH wildtype protein were produced. One contained their “binary complex”, IDH-NADP, which crystallized from hanging drops in the tetragonal space group P4 3212. The drops were formed from equal parts of protein solution (15 mg/mL IDH, 10 mM NADP) and precipitant consisting of 100 mM MES (pH 6.5) and 12% PEG 20000. The other crystal form contained their “quaternary complex”, IDH-NADP/isocitrate/Ca2+, which crystallized in the monoclinic space group P21 using 100 mM MES (pH 5.9) and 20% PEG 6000 as the precipitant. Here they had added 10 mM DL-isocitrate and 10 mM calcium chloride to the protein solution. First attempts at crystallizing the R132S mutant in this study centered around these two reported conditions with little variation. The following lists the components of the crystallization that could be varied; several different combinations of these components were tried in the screening process.
-
| HcIDH(R132S) |
always ~10 mg/mL or ~0.2 mM |
| Tris-HCl (pH 7.4) |
always 20 mM |
| NaCl |
always 100 mM |
| NADP+/NADPH |
absent or 5 mM NADP+ (did not try NADPH) |
| DL-isocitic acid, |
absent or 5 mM |
| trisodium salt |
| calcium chloride |
absent or 10 mM |
| In the precipitant: |
100 mM MES (pH 6.5) and 12% PEG 20000 |
| |
100 mM MES (pH 6.0) and 20% PEG 6000 |
| Drop size: |
always 3 μL |
| Drop ratios: |
2:1, 1:1 or 1:2 (protein:precipitant) |
| |
-
Upon forming the hanging drops, a milky precipitate was always observed. On inspection after 2-4 days at 20° C. most drops showed dense precipitation or phase separation. In some cases, the precipitate subsided and it was from these types of drops small crystals had grown, for example, as shown in FIG. 33.
-
Crystal optimization. Once bonafide crystals were achieved, the next step was to optimize the conditions to obtain larger and more regularly-shaped crystals of IDH-NADP/isocitrate/Ca2+ in a timely and consistent manner. The optimal screen focused on varying the pH from 5.7 to 6.2, the MES concentration from 50 to 200 mM and the PEG 6000 concentration from 20 to 25%. Also, bigger drops were set up (5-6 μl) and the drop ratios were again varied. These attempts failed to produce larger, diffraction-quality crystals but did reproduce the results reported above. Either a dense precipitate, oily phase separation or small crystals were observed.
-
Using α-Ketoglutarate. Concurrent to the optimization of the isocitrate crystals, other screens were performed to obtain crystals of IDH(R132S) complexed with α-ketoglutarate instead. The protein solution was consistently 10 mg/mL IDH in 20 mM Tris-HCl (pH 7.4) and 100 mM NaCl. The following were added in this order: 5 mM NADP, 5 mM α-ketoglutaric acid (free acid, pH balanced with NaOH) and 10 mM calcium chloride. The protein was allowed to incubate with these compounds for at least an hour before the drops were set up. The precipitant was either 100 mM MES (pH 6.5) and 12% PEG 20000 or 100 mM MES (pH 6.5) and 20% PEG 6000. Again, precipitation or phase separation was primarily seen, but in some drops small crystals did form. At the edge of one of the drops, a single large crystal formed, pictured below. This was the single crystal used in the following structure determination. FIG. 34 shows crystal obtained from a protein solution contained 5 mM NADP, 5 mM α-ketoglutarate, 10 mM Ca2+. Precipitant contained 100 mM MES (pH 6.5) and 12% PEG 20000.
-
Cryo conditions. In order to ship the crystal to the X-ray source and protect it during cryo-crystallography, a suitable cryo-protectant was needed. Glycerol is quite widely used and was the first choice. A cryo solution was made, basically as a mixture of the protein buffer and precipitant solution plus glycerol: 20 mM Tris-HCl (pH 7.5), 100 mM NaCl, 5 mM NADP, 5 mM α-ketoglutaric acid, 10 mM calcium chloride, 100 mM MES (pH 6.5), 12% PEG 20000 and either 12.5% glycerol or 25% glycerol. The crystal was transferred to the cryo solution in two steps. First, 5 μL of the 12.5% glycerol solution was added directly to the drop and incubated for 10 minutes, watching for possible cracking of the crystal. The liquid was removed from the drop and 10 μL of the 25% glycerol solution was added on top of the crystal. Again, this incubated for 10 minutes, harvested into a nylon loop and plunged into liquid nitrogen. The crystal was stored submerged in a liquid nitrogen dewar for transport.
-
Data collection and processing. The frozen crystal was mounted on a Rigaku RAXIS IV X-ray instrument under a stream of nitrogen gas at temperatures near −170° C. A 200° dataset was collected with the image plate detector using 1.54 Å wavelength radiation from a rotating copper anode home source, 1° oscillations and 10 minute exposures. The presence of 25% glycerol as a cryoprotectant was sufficient for proper freezing, as no signs of crystal cracking (split spots or superimposed lattices) were observed. A diffuse ring was observed at 3.6 Å resolution, most likely caused by icing. The X-ray diffraction pattern showed clear lattice planes and reasonable spot separation, although the spacing along one reciprocal axis was rather small (b=275.3). The data was indexed to 2.7 Å resolution into space group P2 1212 with HKL2000 (Otwinowski and Minor, 1997). Three structures for HcIDH are known, designated the closed form (1T0L), the open form (1T09 subunit A) and semi-open form (1T09 subunit B). Molecular replacement was performed with the CCP4 program PHASER (Bailey, 1994) using only the protein atoms from these three form's. Only the closed form yielded a successful molecular replacement result with 6 protein subunits in the asymmetric unit. The unit cell contains approximately 53.8% solvent.
-
Model refinement. Using the CCP4 program REFMAC5, rigid-body refinement was performed to fit each of the 6 IDH subunits in the asymmetric unit. This was followed by rigid-body refinement of the three domains in each protein subunit. Restrained refinement utilizing non-crystallographic symmetry averaging of related pairs of subunits yielded an initial structure with Rcryst of 33% and Rfree of 42%. Model building and real-space refinement were performed using the graphics program COOT (Emsley and Cowtan, 2004). A difference map was calculated and this showed strong electron density into which six individual copies of the NADP ligand and calcium ion were manually fit with COOT. Density for the α-ketoglutarate structure was less defined and was fit after the binding-site protein residues were fit using a 2Fo−Fc composite omit map. Automated Ramachandran-plot optimization coupled with manual real-space density fitting was applied to improve the overall geometry and fit. A final round of restrained refinement with NCS yielded an Rcryst of 30.1% and Rfree of 35.2%.
-
| |
| |
|
|
|
|
|
Unit cell |
|
| a, Å |
b, Å |
c, Å |
α |
B |
γ |
volume, Å3 |
Z |
| |
| 116.14 |
275.30 |
96.28 |
90° |
90° |
90° |
3.08 × 106 |
24 |
| |
-
| |
|
| |
Reflections in working set/test set |
68,755/3,608 (5.0%) |
| |
Rcryst |
30.1% |
| |
Rfree |
35.2% |
| |
|
-
X-ray data and refinement statistics for IDH(R132S)—NADP/α-ketoglutarate/Ca2+
-
| |
|
| |
Crystal parameters |
|
| |
Space group | P2 | 1212 |
| |
Unit cell dimensions |
| |
a, b, c, Å |
116.139, 275.297, 96.283 |
| |
α, β, γ, ° |
90.0, 90.0, 90.0 |
| |
Volume, Å3 |
3,078,440 |
| |
No. protein molecules in |
6 |
| |
asymmetric unit |
| |
No. protein molecules in |
24 |
| |
unit cell, Z |
| |
Data collection |
| |
Beam line |
| |
Date of collection |
Apr. 25, 2009 |
| |
λ, Å |
1.5418 |
| |
Detector |
Rigaku Raxis IV |
| |
Data set (phi), ° |
200 |
| |
Resolution, Å |
25-2.7 (2.8-2.7) |
| |
Unique reflections (N, |
73,587 |
| |
F > 0) |
| |
Completeness, % |
85.4 (48.4) |
| |
<I>/σI |
9.88 (1.83) |
| |
R-merge |
0.109 (0.33) |
| |
Redundancy |
4.3 (1.8) |
| |
Mosaicity |
0.666 |
| |
Wilson B factor |
57.9 |
| |
Anisotropy B factor, Å2 |
−1.96 |
| |
Refinement Statistics |
| |
Resolution limit, Å |
20.02-2.70 |
| |
No. of reflections used |
68,755/3608 |
| |
for R-worka/R-freeb |
| |
Protein atoms |
19788 |
| |
Ligand atoms |
348 |
| |
No. of waters |
357 |
| |
Ions etc. |
6 |
| |
Matthews coeff. Å3/ |
2.68 |
| |
Dalton |
| |
Solvent, % |
53.8 |
| |
R-worka/R-freeb, (%) |
30.1/35.2 |
| |
Figure-of-meritc |
0.80 (0.74) |
| |
Average B factors |
31.0 |
| |
Coordinates error |
0.484 |
| |
(Luzzati plot), Å |
| |
R.M.S. deviations |
| |
Bond lengths, Å |
0.026 |
| |
Bond angles, ° |
2.86 |
| |
|
-
Completeness and R-merge are given for all data and for data in the highest resolution shell. Highest shell values are in parentheses.
-
aR factor=Σhkl/Fo−Fc|/ΣhklFo, where Fo and Fc are the observed and calculated structure factor amplitudes, respectively for all reflections hkl used in refinement.
bR-free is calculated for 5% of the data that were not used in refinement.
cFigure of merit=√{square root over (x2+y2)}, where x=(Σ0 2πP(α)cos α)/(Σ0 2πP(α)), y=(Σ0 2πP(α)sin α)/(Σ0 2πP(α)), and the phase probability P(α)=exp(A cos α+B sin α+C cos(2α)+D sin(2α), where A, B, C, and D are the Hendrickson-Lattman coefficients and α is the phase.
Stereochemistry of IDH(R132S)—NADP/α-Ketoglurate/Ca2+
-
-
| |
| |
No. of |
|
| |
amino |
% of |
| Ramachandran plot statistics |
acids |
Residues |
| |
| |
| Residues in most favored regions [A, B, L] |
1824 |
82.2 |
| Residues in additional allowed regions |
341 |
15.4 |
| [a, b, l, p] |
| Residues in generously allowed regions |
38 |
1.7 |
| [−a, −b, −l, −p] |
| Residues in disallowed regions |
17 |
0.8 |
| Number of non-glycine and non-proline residues |
2220 |
100 |
| Number of end-residues (excl. Gly and Pro) |
387 |
| Number of glycine residues |
198 |
| Number of proline residues |
72 |
| Total number of residues |
2877 |
| Overall <G>-factord score (>−1.0) |
−0.65 |
| |
-
Generated by PROCHECK (Laskowski R A, MacArthur M W, Moss D S, Thornton J M (1993) J Appl Crystallogr 26:283-291.)
-
dG-factors for main-chain and side-chain dihedral angles, and main-chain covalent forces (bond lengths and bond angles). Values should be ideally −0.5 or above −1.0.
-
| |
|
| |
Radiation wavelength, Å |
1.54 |
| |
Resolution, Å (outer shell) |
20-2.70 (2.80-2.70) |
| |
Unique reflections |
73,587 |
| |
Completeness (outer shell) |
85.4% (48.4%) |
| |
Redundancy (outer shell) |
4.3 (1.8) |
| |
Rmerge (outer shell) |
10.9% (33%) |
| |
<I>/<σ(I)> (outer shell) |
9.88 (1.83) |
| |
|
-
Clinical Specimens, metabolite extraction and analysis. Human brain tumors were obtained during surgical resection, snap frozen in isopentane cooled by liquid nitrogen and stored at −80 C. Clinical classification of the tissue was performed using standard clinical pathology categorization and grading as established by the WHO. Genomic sequence analysis was deployed to identify brain tumor samples containing either wild-type isocitrate dehydrogenase (IDH1) or mutations altering amino acid 132. Genomic DNA was isolated from 50-100 mgs of brain tumor tissue using standard methods. A polymerase chain reaction on the isolated genomic DNA was used to amplify a 295 base pair fragment of the genomic DNA that contains both the intron and 2nd exon sequences of human IDH1 and mutation status assessed by standard molecular biology techniques.
-
Metabolite extraction was accomplished by adding a 10× volume (m/v ratio) of −80° C. methanol:water mix (80%:20%) to the brain tissue (approximately 100 mgs) followed by 30 s homogenization at 4 C. These chilled, methanol extracted homogenized tissues were then centrifuged at 14,000 rpm for 30 minutes to sediment the cellular and tissue debris and the cleared tissue supernatants were transferred to a screw-cap freezer vial and stored at −80° C. For analysis, a 2× volume of tributylamine (10 mM) acetic acid (10 mM) pH 5.5 was added to the samples and analyzed by LCMS as follows. Sample extracts were filtered using a Millex-FG 0.20 micron disk and 10 μl were injected onto a reverse-phase HPLC column (Synergi 150 mm×2 mm, Phenomenex Inc.) and eluted using a linear gradient LCMS-grade methanol (50%) with 10 mM tributylamine and 10 mM acetic acid) ramping to 80% methanol:10 mM tributylamine: 10 mM acetic acid over 6 minutes at 200 μL/min. Eluted metabolite ions were detected using a triple-quadrupole mass spectrometer, tuned to detect in negative mode with multiple-reaction-monitoring mode transition set (MRM's) according to the molecular weights and fragmentation patterns for 8 known central metabolites, including 2-hydroxyglutarate as described above. Data was processed using Analyst Software (Applied Biosystems, Inc.) and metabolite signal intensities were obtained by standard peak integration methods.
Example 9
Compounds that Inhibit IDH1 R132H
-
Assays were conducted in a volume of 76 ul assay buffer (150 mM NaCl, 10 mM MgCl2, 20 mM Tris pH 7.5, 0.03% bovine serum albumin) as follows in a standard 384-well plate: To 25 ul of substrate mix (8 uM NADPH, 2 mM αKG), 1 ul of test compound was added in DMSO. The plate was centrifuged briefly, and then 25 ul of enzyme mix was added (0.2 ug/ml ICDH1 R132H) followed by a brief centrifugation and shake at 100 RPM. The reaction was incubated for 50 minutes at room temperature, then 25 ul of detection mix (30 uM resazurin, 36 ug/ml) was added and the mixture further incubated for 5 minutes at room temperature. The conversion of resazurin to resorufin was detected by fluorescent spectroscopy at Ex544 Em590 c/o 590.
-
Table 24a shows the wild type vs mutant selectivity profile of 5 examples of IDH1R132H inhibitors. The IDH1 wt assay was performed at 1×Km of NADPH as opposed to IDHR132H at 10× or 100×Km of NADPH. The second example showed no inhibition, even at 100 uM. Also, the first example has IC50=5.74 uM but is shifted significantly when assayed at 100×Km, indicating direct NADPH-competitive inhibitor. The selectivity between wild type vs mutant could be >20-fold.
-
| TABLE 24a |
| |
| |
|
|
ICDH IC50 |
ICDH IC50 |
|
|
| |
|
|
(uM) @ 4 uM |
(uM) @ |
IC50 |
IDH1wt |
| |
LDHa |
LDHb |
(10x Km) |
40 uM |
Ratio |
IC50 @ 1x |
| STRUCTURE |
IC50 |
IC50 |
NADPH |
NADPH |
(40/4) |
Km (uM) |
| |
| |
|
|
25.43 |
64.07 |
5.74 |
>100 |
17.42 |
16.22 |
| |
|
|
5.92 |
17.40 |
12.26 |
41.40 |
3.38 |
NO inhibition |
| |
|
|
8.61 |
>100 |
12.79 |
14.70 |
1.15 |
19.23 |
| |
|
|
33.75 |
>100 |
14.98 |
19.17 |
1.28 |
46.83 |
| |
|
|
12.76 |
>100 |
23.80 |
33.16 |
1.39 |
69.33 |
| |
-
Additional exemplary compounds that inhibit IDH1R132H are provided below in Table 24b.
-
|
|
1 |
| |
|
|
2 |
| |
|
|
3 |
| |
|
|
4 |
| |
|
|
5 |
| |
|
|
6 |
| |
|
|
7 |
| |
|
|
8 |
| |
|
|
9 |
| |
|
|
10 |
| |
|
|
11 |
| |
|
|
12 |
| |
|
|
13 |
| |
|
|
14 |
| |
|
|
15 |
| |
|
|
16 |
| |
|
|
17 |
| |
|
|
18 |
| |
|
|
19 |
| |
|
|
20 |
| |
|
|
21 |
| |
|
|
22 |
| |
|
|
23 |
| |
|
|
24 |
| |
|
|
25 |
| |
|
|
26 |
| |
|
|
27 |
| |
|
|
28 |
| |
|
|
29 |
| |
|
|
30 |
| |
|
|
31 |
| |
|
|
32 |
| |
|
|
33 |
| |
|
|
34 |
| |
|
|
35 |
| |
|
|
36 |
| |
|
|
37 |
| |
|
|
38 |
| |
|
|
39 |
| |
|
|
40 |
| |
|
|
41 |
| |
|
|
42 |
| |
|
|
43 |
| |
|
|
44 |
| |
|
|
45 |
| |
|
|
46 |
| |
|
|
47 |
| |
|
|
48 |
| |
|
|
49 |
| |
|
|
50 |
| |
|
|
51 |
| |
|
|
52 |
| |
|
|
53 |
| |
|
|
54 |
| |
|
|
55 |
| |
|
|
56 |
| |
|
|
57 |
| |
|
|
58 |
| |
|
|
59 |
| |
|
|
60 |
| |
|
|
61 |
| |
|
|
62 |
| |
|
|
63 |
| |
|
|
64 |
| |
|
|
65 |
| |
|
|
66 |
| |
|
|
67 |
| |
|
|
68 |
| |
|
|
69 |
| |
|
|
70 |
| |
|
|
71 |
| |
|
|
72 |
| |
|
|
73 |
| |
|
|
74 |
| |
|
|
75 |
| |
|
|
76 |
| |
|
|
77 |
| |
|
|
78 |
| |
|
|
79 |
| |
|
|
80 |
| |
|
|
81 |
| |
|
|
82 |
| |
|
|
83 |
| |
|
|
84 |
| |
|
|
85 |
| |
|
|
86 |
| |
|
|
87 |
| |
|
|
88 |
| |
|
|
89 |
| |
|
|
90 |
| |
|
|
91 |
| |
|
|
92 |
| |
Example 10
The Mutant Enzyme IDH2-R172K has Elevated NADPH Reductive Catalysis Activity as Compared to Wildtype IDH2 Enzyme
-
NADPH reduction activity was measured for the enzymes IDH2-R172K, IDH2-wildtype, IDH1-R132H and IDH1-wildtype. The final reactant concentrations for each reaction were as follows: 20 mM Tris 7.5, 150 mM NaCl, 2 mM MnCl2, 10% glycerol, 0.03% BSA, enzyme (1-120 μg/mL), 1 mM NADPH, and 5 mM αKG (alpha ketoglutarate). The resulting specific activities (μmol/min/mg) are presented in the graph in FIG. 35. The results indicate that the mutant IDH2 has elevated reductive activity as compared to wildtype IDH2, even though both the mutant and wildtype IDH2 enzymes were able to make 2HG (2-hydroxyglutarate) at saturating levels of reactants αKG and NADPH.
Example 11
2-HG Accumulates in AML with IDH1/2 Mutations
Patients and Clinical Data
-
Peripheral blood and bone marrow were collected from AML patients at the time of diagnosis and at relapse, following REB approved informed consent. The cells were separated by ficol hypaque centrifugation, and stored at −150° C. in 10% DMSO, 40% FCS and 50% alpha-MEM medium. Patient sera were stored at −80° C. Cytogenetics and molecular testing were performed in the diagnostic laboratory of the University Health Network (Toronto, Canada). A subgroup of patients (n=132) was given consistent initial treatment using a standard induction and consolidation chemotherapy regimen consisting of daunorubicin and cytarabine.
IDH1 and IDH2 Genotyping
-
DNA was extracted from leukemic cells and cell lines using the Qiagen Puregene kit (Valencia Calif.). For a subset of samples (n=96), RNA was extracted from leukemic cells using a Qiagen RNeasy kit, and reverse transcribed into cDNA for IDH1 and IDH2 genotyping. IDH1 and IDH2 genotype was determined at the Analytical Genetics Technology Centre at the University Health Network (Toronto, Canada) using a Sequenom MassARRAY™ platform (Sequenom, San Diego, Calif.). Positive results were confirmed by direct sequencing on an ABI PRISM 3130XL genetic analyzer (Applied Biosystems, Foster City, Calif.).
Cell Lines
-
AML cell lines (OCI/AML-1, OCI/AML-2, OCI/AML-3, OCI/AML-4, OCI/AML-5, HL-60, MV-4-11, THP-1, K562, and KG1A) and 5637 cells were obtained from the laboratory of Mark Minden (Ontario Cancer Institute, Toronto, Canada). Primary AML cells were cultured in alpha-MEM media supplemented with 20% fetal bovine serum, and 10% 5637 cell conditioned media as previously described13. Growth curves were generated by counting viable cells as assessed by trypan blue exclusion on a Vi-CELL automated cell counter (Beckman Coulter, Fullarton, Calif.).
Expression/Purification of IDH1 and IDH2 Proteins
-
The human IDH1 cDNA (ref. ID NM—005896) and IDH2 cDNA (ref ID NM—002168) were purchased from OriGene Technologies (Rockville, Md.). For expression in E. coli, the coding region was amplified by PCR using primers designed to add NDEI and XHO1 restrictions sites at the 5′ and 3′ ends respectively. The resultant fragments for IDH1 (full length) and IDH2 (residues 40-452) were cloned into vector pET41a (EMD Biosciences, Madison, Wis.) to enable the E. coli expression of C-terminal His8-tagged protein. Site directed mutagenesis was performed on the pET41a-IDH1 and pET41a-IDH2 plasmid using the QuikChange® Lightning Site-Directed Mutagenesis Kit (Stratagene, La Jolla, Calif.) to change C394 to T in the IDH1 cDNA, resulting in the R132C mutation, and to change G515 to A in the IDH2 cDNA, resulting in the R172K mutation. Wild-type and mutant IDH1 proteins were expressed in and purified from the E. coli Rosetta™ (DE3) strain according to manufacturer's instructions (Invitrogen, Carlsbad, Calif.). Overexpression of IDH2 protein was accomplished by co-transfection of expression plasmids encoding respective IDH2 clones and pG-KJE8 expressing chaperone proteins.
IDH1/2 Activity Assays
-
Enzymatic activity was assessed by following the change in NADPH absorbance at 340 nm over time in an SFM-400 stopped-flow spectrophotometer (BioLogic, Knoxyille, Tenn.) in the presence of isocitrate and NADP+ (forward reaction), or α-KG and NADPH (reverse reaction). All reactions were performed in standard enzyme reaction buffer (150 mM NaCl, 20 mM Tris-Cl, pH 7.5, 10 mM MgCl2 and 0.03% (w/v) bovine serum albumin). For determination of kinetic parameters, sufficient enzyme was added to give a linear reaction for 1 to 5 seconds. Enzymatic binding constants were determined using curve fitting algorithms to standard kinetic models with the Sigmaplot software package (Systat Software, San Jose, Calif.). For determination of kcat, enzyme was incubated with 5×Km of substrate and cofactor; consumption of NADPH or NADP was determined by a change in the OD340 over time. In both cases an extinction coefficient of 6200 M−1 cm−1 was used for NADPH.
2-HG and Metabolite Analysis
-
Metabolites were extracted from cultured cells, primary leukemic cells, and sera using 80% aqueous methanol (−80° C.) as previously described. For cell extraction, frozen biopsies were thawed quickly at 37° C., and an aliquot of 2 million cells was spun down at 4° C. The pellet was resuspended in −80° C. 80% methanol. For serum extraction, 1 ml of serum was thawed quickly and mixed with 4 ml −80° C. methanol. All extracts were spun at 13000 rpm at 4° C. to remove precipitate, dried at room temperature, and stored at −80° C. until analysis by LC-MS. Metabolite levels (2-HG, α-KG, succinate, fumarate, and malate) were determined by ion paired reverse phase LC coupled to negative mode electrospray triple-quadrupole MS using multiple reaction monitoring, and integrated elution peaks were compared with metabolite standard curves for absolute quantification as described.
Statistical Analysis
-
Fisher's exact test was used to test for differences in categorical variables between IDH1/2 wt and IDH1/2 mutant patients. One way ANOVA followed by a student's t-test with correction for multiple comparisons was used to test for differences in IDH1 activity and metabolite concentrations. Differences with p<0.05 were considered significant.
Results
-
In order to investigate the role of IDH1 R132 mutations in AML, leukemic cells obtained at initial presentation, from a series of 145 AML patients treated at the Princess Margaret Hospital with the aim of identifying mutant samples in our viable cell tissue bank were genotyped. Heterozygous IDH1 R132 mutations were found in 11 (8%) of these patients (Table 25). The spectrum of IDH1 mutations observed in AML appears to differ from that seen in CNS tumors. In the CNS, the majority of mutations (80-90%) are IDH1 R132H substitutions, whereas 5, 4, and 2 patients with IDH1 R132H, R132C, and R132G mutations, respectively (Table 25), were observed. In four cases, leukemic cells were also available from samples taken at the time of relapse. The IDH1 mutation was retained in 4/4 of these samples (Table 25). One of the patients harboring an IDH1 mutation had progressed to AML from an earlier myelodysplastic syndrome (MDS). When cells from the prior MDS in this patient were analyzed, IDH1 was found to be wild-type. An additional 14 patients with MDS were genotyped, and all patients were found to be wild-type for IDH1, suggesting that IDH1 mutations are not a common feature of this disease. In samples from a subset of IDH1 mutant patients (n=8), reverse transcribed RNA was used for genotyping in order to assess the relative expression of mutant and wild-type alleles. Seqenom genotyping showed balanced allele peaks for these samples, indicating that both the wild-type and mutant genes are expressed. Ten established AML cell lines were also genotyped (OCI/AML-1, OCI/AML-2, OCI/AML-3, OCI/AML-4, OCI/AML-5, HL-60, MV-4-11, THP-1, K562, and KG1A) and none carried an IDH1 R132 mutation. Table 25: Identification of 13 AML patients bearing an IDH1 R132 or IDH2 R172 mutation*
-
| TABLE 25 |
| |
| Table 25: Identification of 13 AML patients bearing an IDH1 R132 or IDH2 R172 mutation* |
| |
|
|
|
NPM1 and |
|
|
|
| |
|
Amino acid |
FAB |
FLT3 |
|
Genotype at |
2-HG level |
| Patient ID |
Mutation |
change |
subtype |
status |
Cytogenetic profile |
relapse |
(ng/2 × 106 cells) |
| |
| 090108 |
G/A |
R132H |
M4 |
na |
Normal |
na |
2090 |
| 090356 |
G/A |
R132H |
na |
na |
na |
na |
1529 |
| 0034 |
C/T |
R132C |
M5a |
Normal |
Normal |
na |
10285 |
| 0086 |
C/G |
R132G |
M2 |
Normal |
Normal |
na |
10470 |
| 0488 |
C/T |
R132C |
M0 |
Normal |
Normal |
R132C |
13822 |
| 8587 |
G/A |
R132H |
na |
Normal |
Normal |
na |
5742 |
| 8665 |
C/T |
R132C |
M1 |
na |
Normal |
na |
7217 |
| 8741 |
G/A |
R132H |
M4 |
NPM1 |
Normal |
R132H |
6419 |
| 9544 |
C/G |
R132G |
na |
na |
Normal |
R132G |
4962 |
| 0174268 |
G/A |
R132H |
M1 |
NPM1 |
Normal |
R132H |
8464 |
| 090148 |
C/T |
R132C |
M1 |
na |
46, xx, i(7) (p10) [20] |
na |
na |
| 9382 |
G/A |
R172K |
M0 |
Normal |
Normal |
na |
19247 |
| 0831 |
G/A |
R172K |
M1 |
Normal |
Normal |
na |
15877 |
| |
| *NPM1 denotes nucleophosmin 1, and FLT FMS-related tyrosine kinase 3. |
| na indicates that some data was not available for some patients. |
-
A metabolite screening assay to measure 2-HG in this set of AML samples was set up. Levels of 2-HG were approximately 50-fold higher in samples harboring an IDH1 R132 mutation (Table 25, FIG. 36A, Table 26). 2-HG was also elevated in the sera of patients with IDH1 R132 mutant AML (FIG. 36B). There was no relationship between the specific amino acid substitution at residue 132 of IDH1 and the level of 2-HG in this group of patients.
-
| TABLE 26 |
| |
| Metabolite concentrations in individual IDH1/2 mutant and wild-type AML cells* |
| |
|
2-HG |
α-KG |
Malate |
Fumarate |
Succinate |
| |
IDH1/2 |
(ng/2 × 106 |
(ng/2 × 106 |
(ng/2 × 106 |
(ng/2 × 106 |
(ng/2 × 106 |
| Sample |
Genotype |
cells) |
cells) |
cells) |
cells) |
cells) |
| |
| 0034 |
R132C |
10285 |
125 |
192 |
239 |
2651 |
| 0086 |
R132G |
10470 |
124 |
258 |
229 |
3043 |
| 0488 |
R132C |
13822 |
95 |
184 |
193 |
2671 |
| 8587 |
R132H |
5742 |
108 |
97 |
95 |
1409 |
| 8665 |
R132C |
7217 |
137 |
118 |
120 |
1648 |
| 8741 |
R132H |
6419 |
87 |
66 |
61 |
938 |
| 9544 |
R132G |
4962 |
95 |
76 |
72 |
1199 |
| 0174268 |
R132H |
8464 |
213 |
323 |
318 |
2287 |
| 090356 |
R132H |
1529 |
138 |
657 |
366 |
1462 |
| 090108 |
R132H |
2090 |
Na |
246 |
941 |
3560 |
| 090148† |
R132C |
na |
Na |
na |
Na |
Na |
| 8741‡ |
R132H |
2890 |
131 |
113 |
106 |
1509 |
| 9554‡ |
R132G |
7448 |
115 |
208 |
227 |
2658 |
| 0174268‡ |
R132H |
964 |
72 |
134 |
138 |
2242 |
| 0488‡ |
R132C |
7511 |
85 |
289 |
310 |
3448 |
| 9382 |
R172K |
19247 |
790 |
821 |
766 |
5481 |
| 0831 |
R172K |
15877 |
350 |
721 |
708 |
5144 |
| 157 |
Wild type |
212 |
121 |
484 |
437 |
3057 |
| 202 |
Wild type |
121 |
57 |
161 |
136 |
1443 |
| 205 |
Wild type |
147 |
39 |
162 |
153 |
1011 |
| 209 |
Wild type |
124 |
111 |
167 |
168 |
1610 |
| 239 |
Wild type |
112 |
106 |
305 |
361 |
1436 |
| 277 |
Wild type |
157 |
61 |
257 |
257 |
2029 |
| 291 |
Wild type |
113 |
118 |
124 |
128 |
1240 |
| 313 |
Wild type |
116 |
75 |
151 |
181 |
1541 |
| 090158 |
Wild type |
411 |
217 |
658 |
647 |
3202 |
| 090156 |
Wild type |
407 |
500 |
1276 |
1275 |
6091 |
| |
| *IDH1/2 denotes isocitrate dehydrogenase 1 and 2,2-HG 2-hydroxy glutarate, and α-KG alpha-ketogluatarate. |
| Metabolite measurements were not available for all patients. |
| †metabolic measurements were not made due to limited patient sample |
| ‡indicates samples obtained at relapse. |
-
Two samples harboring wild-type IDH1 also showed high levels of 2-HG (Table 25). The high 2-HG concentration prompted sequencing of the IDH2 gene in these two AML samples, which established the presence of IDH2 R172K mutations in both samples (Table 25).
-
Evaluation of the clinical characteristics of patients with or without IDH1/2 mutations revealed a significant correlation between IDH1/2 mutations and normal karyotype (p=0.05), but no other differences between these two groups (Table 27). Notably, there was no difference in treatment response for a subgroup of patients who received consistent treatment (n=136). These findings are consistent with the initial report identifying IDH1 mutations in AML.
-
| TABLE 27 |
| |
| Characteristics of IDH1/2 mutant and wild-type patients* |
| |
IDH1/2 Wild-type |
IDH1/2 Mutant |
|
| |
(N = 132) |
(N = 13) |
P Value |
| |
|
| Variable |
|
|
|
| Age (yr) |
58.8 ± 16.2 |
52.6 ± 7.0 |
0.17† |
| Sex (% male) |
53 (70/132) |
62 (8/13) |
0.77‡ |
| WBC at diagnosis (109 |
40.7 ± 50.6 |
28.7 ± 34.1 |
0.38† |
| cells/L) |
| Initial treatment response |
70 (85/122) |
62 (8/13) |
0.54‡ |
| (% complete remission) |
| Cytogenetic profile (% |
62 (72/117) |
92 (11/12) |
0.05‡ |
| normal) |
| Additional mutations |
| FLT3 (%) |
17 (8/47) |
0 (0/8) |
0.58‡ |
| NPM1 (%) |
30 (14/47) |
25 (2/8) |
1.0‡ |
| |
| *For plus-minus values, the value indicates the mean, and ± indicates the standard deviation. IDH1/2 denotes isocitrate dehydrogenase 1 and 2, WBC white blood cell count, FLT3 FMS-related tyrosine kinase 3, and NPM1 nucleophosmin 1. |
| †P-value was calculated using the student's t-test. |
| ‡P-value was calculated using Fisher's exact test. |
-
Panels of AML cells from wild-type and IDH1 mutant patients were cultured in vitro. There was no difference in the growth rates or viability of the IDH1 R132 mutant and wild-type cells, with both groups showing high variability in their ability to proliferate in culture, as is characteristic of primary AML cells (FIG. 36C). There was no relationship between 2-HG levels in the IDH1 R132 mutant cells and their growth rate or viability in culture. After 14 days in culture, the mutant AML cells retained their IDH1 R132 mutations (11/11), and continued to accumulate high levels of 2-HG (FIG. 36A), further confirming that IDH1 R132 mutations lead to the production and accumulation of 2-HG in AML cells.
-
To investigate the effect of IDH1/2 mutations on the concentration of cellular metabolites proximal to the IDH reaction, α-KG, succinate, malate, and fumarate levels were measured in AML cells with IDH1/2 mutations and in a set of wild-type AML cells matched for AML subtype and cytogenetic profile. None of the metabolites were found to be greatly altered in the IDH1 mutants compared to the IDH1 wild-type cells (FIG. 27, Supplementary Table 26). The mean level of α-KG was not altered in the IDH1/2 mutant AML cells, suggesting that the mutation does not decrease the concentration of this metabolite as has been previously hypothesized. To confirm that the R132C mutation of IDH1, and the R172K mutation of IDH2 confer a novel enzymatic activity that produces 2-HG, recombinant mutant enzymes were assayed for the NADPH-dependent reduction of α-KG. When samples were analyzed by LC-MS upon completion of the enzyme assay, 2-HG was identified as the end product for both the IDH1 R132C and IDH2 R172K mutant enzymes (FIG. 38). No isocitrate was detectable by LC-MS, indicating that 2-HG is the sole product of this reaction (FIG. 38). This observation held true even when the reductive reaction was performed in buffer containing NaHCO3 saturated with CO2. A large proportion of IDH1 mutant patients in AML have an IDH1 R132C mutation (Table 25). In order to biochemically characterize mutant IDH1 R132C, the enzymatic properties of recombinant R132C protein were assessed in vitro. Kinetic analyses showed that the R132C substitution severely impairs the oxidative decarboxylation of isocitrate to α-KG, with a significant decrease in kcat, even though the affinity for the co-factor NADP+ remains essentially unchanged (Table 28). However, unlike the R132H mutant enzyme described previously the R132C mutation leads to a dramatic loss of affinity for isocitrate (KM), and a drop in net isocitrate metabolism efficiency (kcat/KM) of more than six orders of magnitude (Table 28). This suggests a potential difference in the substrate-level regulation of enzyme activity in the context of AML. While substitution of cysteine at R132 inactivates the canonical conversion of isocitrate to α-KG, the IDH1 R132C mutant enzyme acquires the ability to catalyze the reduction of α-KG to 2-HG in an NADPH dependent manner (FIG. 39). This reductive reaction of mutant IDH1 R132C is highly efficient (kcat/KM) compared to the wild-type enzyme, due to the considerable increase in binding affinity of both the NADPH and α-KG substrates (KM) (Table 28).
-
| TABLE 28 |
| |
| Kinetic parameters of the IDH1 R132C mutant enzyme |
| |
Oxidative (→ NADPH) |
|
|
| |
KM, NADP+ (μM) |
49 |
21 |
| |
KM, isocitrate (μM) |
57 |
8.7 × 104 |
| |
KM, MgCl2 (μM) |
29 |
4.5 × 102 |
| |
Ki, αKG (μM) |
6.1 × 102 |
61 |
| |
kcat (s−1) |
1.3 × 105 |
7.1 × 102 |
| |
kcat/KM, isoc(M−1 · s−1) |
2.3 × 109 |
8.2 × 103 |
| |
Reductive (→ NADP+) |
| |
KM, NADPH (μM) |
n/a* |
0.3 |
| |
KM, αKG (μM) |
n/a |
295 |
| |
kcat (s−1) |
~7 (est.) |
5.5 × 102 |
| |
|
| |
*n/a indicates no measureable activity |