RU2379231C2 - Способ получения кристаллов динитрамида аммония (adn), кристаллы adn и содержащий их энергетический композит - Google Patents
Способ получения кристаллов динитрамида аммония (adn), кристаллы adn и содержащий их энергетический композит Download PDFInfo
- Publication number
- RU2379231C2 RU2379231C2 RU2007135718/15A RU2007135718A RU2379231C2 RU 2379231 C2 RU2379231 C2 RU 2379231C2 RU 2007135718/15 A RU2007135718/15 A RU 2007135718/15A RU 2007135718 A RU2007135718 A RU 2007135718A RU 2379231 C2 RU2379231 C2 RU 2379231C2
- Authority
- RU
- Russia
- Prior art keywords
- calcium
- magnesium
- crystals
- adn
- crystal growth
- Prior art date
Links
- 239000013078 crystal Substances 0.000 title claims abstract description 115
- 238000000034 method Methods 0.000 title claims abstract description 47
- BRUFJXUJQKYQHA-UHFFFAOYSA-O ammonium dinitramide Chemical compound [NH4+].[O-][N+](=O)[N-][N+]([O-])=O BRUFJXUJQKYQHA-UHFFFAOYSA-O 0.000 title claims abstract description 18
- 239000002131 composite material Substances 0.000 title claims abstract description 13
- 239000011575 calcium Substances 0.000 claims abstract description 79
- 239000003607 modifier Substances 0.000 claims abstract description 28
- 238000002425 crystallisation Methods 0.000 claims abstract description 24
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 claims abstract description 18
- 229910052791 calcium Inorganic materials 0.000 claims abstract description 18
- 239000002904 solvent Substances 0.000 claims abstract description 15
- 150000003839 salts Chemical group 0.000 claims abstract description 9
- 159000000003 magnesium salts Chemical class 0.000 claims abstract description 6
- 229910052784 alkaline earth metal Inorganic materials 0.000 claims abstract description 4
- 239000011777 magnesium Substances 0.000 claims description 43
- 230000008025 crystallization Effects 0.000 claims description 20
- 229960005069 calcium Drugs 0.000 claims description 17
- 239000000203 mixture Substances 0.000 claims description 14
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 claims description 9
- 229910052749 magnesium Inorganic materials 0.000 claims description 9
- 229940091250 magnesium supplement Drugs 0.000 claims description 9
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 claims description 8
- ZCCIPPOKBCJFDN-UHFFFAOYSA-N calcium nitrate Chemical compound [Ca+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O ZCCIPPOKBCJFDN-UHFFFAOYSA-N 0.000 claims description 8
- 238000001816 cooling Methods 0.000 claims description 8
- 229910020366 ClO 4 Inorganic materials 0.000 claims description 4
- FEWJPZIEWOKRBE-LWMBPPNESA-L D-tartrate(2-) Chemical compound [O-]C(=O)[C@@H](O)[C@H](O)C([O-])=O FEWJPZIEWOKRBE-LWMBPPNESA-L 0.000 claims description 4
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 claims description 4
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 claims description 4
- 229910000019 calcium carbonate Inorganic materials 0.000 claims description 4
- 235000012241 calcium silicate Nutrition 0.000 claims description 4
- 229910052918 calcium silicate Inorganic materials 0.000 claims description 4
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 claims description 4
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 claims description 4
- YIXJRHPUWRPCBB-UHFFFAOYSA-N magnesium nitrate Chemical compound [Mg+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O YIXJRHPUWRPCBB-UHFFFAOYSA-N 0.000 claims description 4
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 claims description 4
- XWKBMOUUGHARTI-UHFFFAOYSA-N tricalcium;diphosphite Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])[O-].[O-]P([O-])[O-] XWKBMOUUGHARTI-UHFFFAOYSA-N 0.000 claims description 4
- -1 alkaline earth metal cations Chemical class 0.000 claims description 3
- 239000011230 binding agent Substances 0.000 claims description 3
- 159000000007 calcium salts Chemical class 0.000 claims description 3
- RWYRUDPAALLKPX-UHFFFAOYSA-N 2,2-difluoro-n-methylethanamine;hydrochloride Chemical compound Cl.CNCC(F)F RWYRUDPAALLKPX-UHFFFAOYSA-N 0.000 claims description 2
- WHQOKFZWSDOTQP-UHFFFAOYSA-N 2,3-dihydroxypropyl 4-aminobenzoate Chemical compound NC1=CC=C(C(=O)OCC(O)CO)C=C1 WHQOKFZWSDOTQP-UHFFFAOYSA-N 0.000 claims description 2
- WLAMNBDJUVNPJU-UHFFFAOYSA-M 2-methylbutyrate Chemical compound CCC(C)C([O-])=O WLAMNBDJUVNPJU-UHFFFAOYSA-M 0.000 claims description 2
- 229910004762 CaSiO Inorganic materials 0.000 claims description 2
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 claims description 2
- CBOCVOKPQGJKKJ-UHFFFAOYSA-L Calcium formate Chemical compound [Ca+2].[O-]C=O.[O-]C=O CBOCVOKPQGJKKJ-UHFFFAOYSA-L 0.000 claims description 2
- 239000001749 Calcium fumarate Substances 0.000 claims description 2
- 239000001736 Calcium glycerylphosphate Substances 0.000 claims description 2
- ROZZMLUWBPPEMU-GRVYQHKQSA-L Calcium linoleate Chemical compound [Ca+2].CCCCC\C=C/C\C=C/CCCCCCCC([O-])=O.CCCCC\C=C/C\C=C/CCCCCCCC([O-])=O ROZZMLUWBPPEMU-GRVYQHKQSA-L 0.000 claims description 2
- BCZXFFBUYPCTSJ-UHFFFAOYSA-L Calcium propionate Chemical compound [Ca+2].CCC([O-])=O.CCC([O-])=O BCZXFFBUYPCTSJ-UHFFFAOYSA-L 0.000 claims description 2
- MPCRDALPQLDDFX-UHFFFAOYSA-L Magnesium perchlorate Chemical compound [Mg+2].[O-]Cl(=O)(=O)=O.[O-]Cl(=O)(=O)=O MPCRDALPQLDDFX-UHFFFAOYSA-L 0.000 claims description 2
- MQHWFIOJQSCFNM-UHFFFAOYSA-L Magnesium salicylate Chemical compound [Mg+2].OC1=CC=CC=C1C([O-])=O.OC1=CC=CC=C1C([O-])=O MQHWFIOJQSCFNM-UHFFFAOYSA-L 0.000 claims description 2
- 229910004298 SiO 2 Inorganic materials 0.000 claims description 2
- 229910004283 SiO 4 Inorganic materials 0.000 claims description 2
- GIYXJPMTXHIINV-UHFFFAOYSA-N [Mg++].[Bi+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O Chemical compound [Mg++].[Bi+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O GIYXJPMTXHIINV-UHFFFAOYSA-N 0.000 claims description 2
- 239000002253 acid Substances 0.000 claims description 2
- 150000001298 alcohols Chemical class 0.000 claims description 2
- 239000001110 calcium chloride Substances 0.000 claims description 2
- 229910001628 calcium chloride Inorganic materials 0.000 claims description 2
- FNAQSUUGMSOBHW-UHFFFAOYSA-H calcium citrate Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O.[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O FNAQSUUGMSOBHW-UHFFFAOYSA-H 0.000 claims description 2
- 239000001354 calcium citrate Substances 0.000 claims description 2
- 229940044172 calcium formate Drugs 0.000 claims description 2
- 235000019255 calcium formate Nutrition 0.000 claims description 2
- 239000004281 calcium formate Substances 0.000 claims description 2
- 235000019296 calcium fumarate Nutrition 0.000 claims description 2
- NEEHYRZPVYRGPP-IYEMJOQQSA-L calcium gluconate Chemical compound [Ca+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O NEEHYRZPVYRGPP-IYEMJOQQSA-L 0.000 claims description 2
- UHHRFSOMMCWGSO-UHFFFAOYSA-L calcium glycerophosphate Chemical compound [Ca+2].OCC(CO)OP([O-])([O-])=O UHHRFSOMMCWGSO-UHFFFAOYSA-L 0.000 claims description 2
- 229940095618 calcium glycerophosphate Drugs 0.000 claims description 2
- 235000019299 calcium glycerylphosphate Nutrition 0.000 claims description 2
- 229940064002 calcium hypophosphite Drugs 0.000 claims description 2
- 229910001382 calcium hypophosphite Inorganic materials 0.000 claims description 2
- MKJXYGKVIBWPFZ-UHFFFAOYSA-L calcium lactate Chemical compound [Ca+2].CC(O)C([O-])=O.CC(O)C([O-])=O MKJXYGKVIBWPFZ-UHFFFAOYSA-L 0.000 claims description 2
- 239000001527 calcium lactate Substances 0.000 claims description 2
- 235000011086 calcium lactate Nutrition 0.000 claims description 2
- 229960002401 calcium lactate Drugs 0.000 claims description 2
- 239000001362 calcium malate Substances 0.000 claims description 2
- OLOZVPHKXALCRI-UHFFFAOYSA-L calcium malate Chemical compound [Ca+2].[O-]C(=O)C(O)CC([O-])=O OLOZVPHKXALCRI-UHFFFAOYSA-L 0.000 claims description 2
- 229940016114 calcium malate Drugs 0.000 claims description 2
- 235000011038 calcium malates Nutrition 0.000 claims description 2
- ROPDWRCJTIRLTR-UHFFFAOYSA-L calcium metaphosphate Chemical compound [Ca+2].[O-]P(=O)=O.[O-]P(=O)=O ROPDWRCJTIRLTR-UHFFFAOYSA-L 0.000 claims description 2
- QXDMQSPYEZFLGF-UHFFFAOYSA-L calcium oxalate Chemical compound [Ca+2].[O-]C(=O)C([O-])=O QXDMQSPYEZFLGF-UHFFFAOYSA-L 0.000 claims description 2
- 235000010331 calcium propionate Nutrition 0.000 claims description 2
- 239000004330 calcium propionate Substances 0.000 claims description 2
- 239000000378 calcium silicate Substances 0.000 claims description 2
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 claims description 2
- 235000013539 calcium stearate Nutrition 0.000 claims description 2
- 239000008116 calcium stearate Substances 0.000 claims description 2
- GUPPESBEIQALOS-UHFFFAOYSA-L calcium tartrate Chemical compound [Ca+2].[O-]C(=O)C(O)C(O)C([O-])=O GUPPESBEIQALOS-UHFFFAOYSA-L 0.000 claims description 2
- 239000001427 calcium tartrate Substances 0.000 claims description 2
- 235000011035 calcium tartrate Nutrition 0.000 claims description 2
- FPCWKNLZGKFYTL-RWUXNGIBSA-L calcium;(e)-3-phenylprop-2-enoate Chemical compound [Ca+2].[O-]C(=O)\C=C\C1=CC=CC=C1.[O-]C(=O)\C=C\C1=CC=CC=C1 FPCWKNLZGKFYTL-RWUXNGIBSA-L 0.000 claims description 2
- HDRTWMBOUSPQON-ODZAUARKSA-L calcium;(z)-but-2-enedioate Chemical compound [Ca+2].[O-]C(=O)\C=C/C([O-])=O HDRTWMBOUSPQON-ODZAUARKSA-L 0.000 claims description 2
- AVVIDTZRJBSXML-UHFFFAOYSA-L calcium;2-carboxyphenolate;dihydrate Chemical compound O.O.[Ca+2].OC1=CC=CC=C1C([O-])=O.OC1=CC=CC=C1C([O-])=O AVVIDTZRJBSXML-UHFFFAOYSA-L 0.000 claims description 2
- BVTQVYBMHFSYPL-UHFFFAOYSA-L calcium;2-methylpropanoate Chemical compound [Ca+2].CC(C)C([O-])=O.CC(C)C([O-])=O BVTQVYBMHFSYPL-UHFFFAOYSA-L 0.000 claims description 2
- PBUBJNYXWIDFMU-UHFFFAOYSA-L calcium;butanedioate Chemical compound [Ca+2].[O-]C(=O)CCC([O-])=O PBUBJNYXWIDFMU-UHFFFAOYSA-L 0.000 claims description 2
- FYPVXEILSNEKOO-UHFFFAOYSA-L calcium;butanoate Chemical compound [Ca+2].CCCC([O-])=O.CCCC([O-])=O FYPVXEILSNEKOO-UHFFFAOYSA-L 0.000 claims description 2
- HIAAVKYLDRCDFQ-UHFFFAOYSA-L calcium;dodecanoate Chemical compound [Ca+2].CCCCCCCCCCCC([O-])=O.CCCCCCCCCCCC([O-])=O HIAAVKYLDRCDFQ-UHFFFAOYSA-L 0.000 claims description 2
- HRBZRZSCMANEHQ-UHFFFAOYSA-L calcium;hexadecanoate Chemical compound [Ca+2].CCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCC([O-])=O HRBZRZSCMANEHQ-UHFFFAOYSA-L 0.000 claims description 2
- ZCZLQYAECBEUBH-UHFFFAOYSA-L calcium;octadec-9-enoate Chemical compound [Ca+2].CCCCCCCCC=CCCCCCCCC([O-])=O.CCCCCCCCC=CCCCCCCCC([O-])=O ZCZLQYAECBEUBH-UHFFFAOYSA-L 0.000 claims description 2
- YZLMERHFSCVBKZ-UHFFFAOYSA-L calcium;pentanoate Chemical compound [Ca+2].CCCCC([O-])=O.CCCCC([O-])=O YZLMERHFSCVBKZ-UHFFFAOYSA-L 0.000 claims description 2
- MLMODXBPCNLAGP-UHFFFAOYSA-L calcium;propanedioate Chemical compound [Ca+2].[O-]C(=O)CC([O-])=O MLMODXBPCNLAGP-UHFFFAOYSA-L 0.000 claims description 2
- HQYGGVLSRVLMEE-UHFFFAOYSA-N dimagnesium dioxidoboranyloxy(dioxido)borane Chemical compound [Mg+2].[Mg+2].[O-]B([O-])OB([O-])[O-] HQYGGVLSRVLMEE-UHFFFAOYSA-N 0.000 claims description 2
- UEGPKNKPLBYCNK-UHFFFAOYSA-L magnesium acetate Chemical compound [Mg+2].CC([O-])=O.CC([O-])=O UEGPKNKPLBYCNK-UHFFFAOYSA-L 0.000 claims description 2
- 239000011654 magnesium acetate Substances 0.000 claims description 2
- 235000011285 magnesium acetate Nutrition 0.000 claims description 2
- 229940069446 magnesium acetate Drugs 0.000 claims description 2
- PJJZFXPJNUVBMR-UHFFFAOYSA-L magnesium benzoate Chemical compound [Mg+2].[O-]C(=O)C1=CC=CC=C1.[O-]C(=O)C1=CC=CC=C1 PJJZFXPJNUVBMR-UHFFFAOYSA-L 0.000 claims description 2
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 claims description 2
- 239000001095 magnesium carbonate Substances 0.000 claims description 2
- 229910000021 magnesium carbonate Inorganic materials 0.000 claims description 2
- 229910001629 magnesium chloride Inorganic materials 0.000 claims description 2
- OVGXLJDWSLQDRT-UHFFFAOYSA-L magnesium lactate Chemical compound [Mg+2].CC(O)C([O-])=O.CC(O)C([O-])=O OVGXLJDWSLQDRT-UHFFFAOYSA-L 0.000 claims description 2
- 239000000626 magnesium lactate Substances 0.000 claims description 2
- 235000015229 magnesium lactate Nutrition 0.000 claims description 2
- 229960004658 magnesium lactate Drugs 0.000 claims description 2
- 229940105112 magnesium myristate Drugs 0.000 claims description 2
- UHNWOJJPXCYKCG-UHFFFAOYSA-L magnesium oxalate Chemical compound [Mg+2].[O-]C(=O)C([O-])=O UHNWOJJPXCYKCG-UHFFFAOYSA-L 0.000 claims description 2
- 229940063002 magnesium palmitate Drugs 0.000 claims description 2
- GVALZJMUIHGIMD-UHFFFAOYSA-H magnesium phosphate Chemical compound [Mg+2].[Mg+2].[Mg+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O GVALZJMUIHGIMD-UHFFFAOYSA-H 0.000 claims description 2
- 229940072082 magnesium salicylate Drugs 0.000 claims description 2
- 235000019359 magnesium stearate Nutrition 0.000 claims description 2
- 229910052943 magnesium sulfate Inorganic materials 0.000 claims description 2
- 235000019341 magnesium sulphate Nutrition 0.000 claims description 2
- 229940062135 magnesium thiosulfate Drugs 0.000 claims description 2
- GMDNUWQNDQDBNQ-UHFFFAOYSA-L magnesium;diformate Chemical compound [Mg+2].[O-]C=O.[O-]C=O GMDNUWQNDQDBNQ-UHFFFAOYSA-L 0.000 claims description 2
- TZKHCTCLSRVZEY-UHFFFAOYSA-L magnesium;dioxido-oxo-sulfanylidene-$l^{6}-sulfane Chemical compound [Mg+2].[O-]S([O-])(=O)=S TZKHCTCLSRVZEY-UHFFFAOYSA-L 0.000 claims description 2
- BJZBHTNKDCBDNQ-UHFFFAOYSA-L magnesium;dodecanoate Chemical compound [Mg+2].CCCCCCCCCCCC([O-])=O.CCCCCCCCCCCC([O-])=O BJZBHTNKDCBDNQ-UHFFFAOYSA-L 0.000 claims description 2
- ABSWXCXMXIZDSN-UHFFFAOYSA-L magnesium;hexadecanoate Chemical compound [Mg+2].CCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCC([O-])=O ABSWXCXMXIZDSN-UHFFFAOYSA-L 0.000 claims description 2
- LOCZQLKNTOXJDV-UHFFFAOYSA-N magnesium;oxido(oxo)borane Chemical compound [Mg+2].[O-]B=O.[O-]B=O LOCZQLKNTOXJDV-UHFFFAOYSA-N 0.000 claims description 2
- DMRBHZWQMKSQGR-UHFFFAOYSA-L magnesium;tetradecanoate Chemical compound [Mg+2].CCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCC([O-])=O DMRBHZWQMKSQGR-UHFFFAOYSA-L 0.000 claims description 2
- AXLHVTKGDPVANO-UHFFFAOYSA-N methyl 2-amino-3-[(2-methylpropan-2-yl)oxycarbonylamino]propanoate Chemical compound COC(=O)C(N)CNC(=O)OC(C)(C)C AXLHVTKGDPVANO-UHFFFAOYSA-N 0.000 claims description 2
- CNALVHVMBXLLIY-IUCAKERBSA-N tert-butyl n-[(3s,5s)-5-methylpiperidin-3-yl]carbamate Chemical compound C[C@@H]1CNC[C@@H](NC(=O)OC(C)(C)C)C1 CNALVHVMBXLLIY-IUCAKERBSA-N 0.000 claims description 2
- YTXZXYYENFXEBZ-UHFFFAOYSA-N tetracalcium disilicate Chemical compound [Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-] YTXZXYYENFXEBZ-UHFFFAOYSA-N 0.000 claims description 2
- 235000013337 tricalcium citrate Nutrition 0.000 claims description 2
- NFMWFGXCDDYTEG-UHFFFAOYSA-N trimagnesium;diborate Chemical compound [Mg+2].[Mg+2].[Mg+2].[O-]B([O-])[O-].[O-]B([O-])[O-] NFMWFGXCDDYTEG-UHFFFAOYSA-N 0.000 claims description 2
- 230000000694 effects Effects 0.000 abstract description 3
- 239000000126 substance Substances 0.000 abstract description 3
- 231100000252 nontoxic Toxicity 0.000 abstract description 2
- 230000003000 nontoxic effect Effects 0.000 abstract description 2
- 150000001768 cations Chemical class 0.000 abstract 1
- 230000000877 morphologic effect Effects 0.000 abstract 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 9
- 238000012986 modification Methods 0.000 description 8
- 230000004048 modification Effects 0.000 description 8
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- 239000002360 explosive Substances 0.000 description 5
- 238000005469 granulation Methods 0.000 description 5
- 230000003179 granulation Effects 0.000 description 5
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- 239000003380 propellant Substances 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 230000005540 biological transmission Effects 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 239000002808 molecular sieve Substances 0.000 description 2
- JTJMJGYZQZDUJJ-UHFFFAOYSA-N phencyclidine Chemical compound C1CCCCN1C1(C=2C=CC=CC=2)CCCCC1 JTJMJGYZQZDUJJ-UHFFFAOYSA-N 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 239000002760 rocket fuel Substances 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- GDDNTTHUKVNJRA-UHFFFAOYSA-N 3-bromo-3,3-difluoroprop-1-ene Chemical compound FC(F)(Br)C=C GDDNTTHUKVNJRA-UHFFFAOYSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical class O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- RNNQKMIGHYYROS-UHFFFAOYSA-N ethyl carbamate;lead Chemical compound [Pb].CCOC(N)=O RNNQKMIGHYYROS-UHFFFAOYSA-N 0.000 description 1
- YSJKYAHYBFJVKV-UHFFFAOYSA-N ethyl n-nitrocarbamate Chemical compound CCOC(=O)N[N+]([O-])=O YSJKYAHYBFJVKV-UHFFFAOYSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000006396 nitration reaction Methods 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 238000013021 overheating Methods 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- BTAAXEFROUUDIL-UHFFFAOYSA-M potassium;sulfamate Chemical compound [K+].NS([O-])(=O)=O BTAAXEFROUUDIL-UHFFFAOYSA-M 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 239000012429 reaction media Substances 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- IIACRCGMVDHOTQ-UHFFFAOYSA-M sulfamate Chemical compound NS([O-])(=O)=O IIACRCGMVDHOTQ-UHFFFAOYSA-M 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B21/00—Nitrogen; Compounds thereof
- C01B21/082—Compounds containing nitrogen and non-metals and optionally metals
- C01B21/087—Compounds containing nitrogen and non-metals and optionally metals containing one or more hydrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C06—EXPLOSIVES; MATCHES
- C06B—EXPLOSIVES OR THERMIC COMPOSITIONS; MANUFACTURE THEREOF; USE OF SINGLE SUBSTANCES AS EXPLOSIVES
- C06B23/00—Compositions characterised by non-explosive or non-thermic constituents
- C06B23/002—Sensitisers or density reducing agents, foam stabilisers, crystal habit modifiers
-
- C—CHEMISTRY; METALLURGY
- C06—EXPLOSIVES; MATCHES
- C06B—EXPLOSIVES OR THERMIC COMPOSITIONS; MANUFACTURE THEREOF; USE OF SINGLE SUBSTANCES AS EXPLOSIVES
- C06B31/00—Compositions containing an inorganic nitrogen-oxygen salt
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
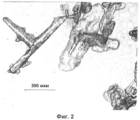
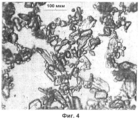
- C01P2004/01—Particle morphology depicted by an image
- C01P2004/02—Particle morphology depicted by an image obtained by optical microscopy
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/30—Particle morphology extending in three dimensions
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/54—Particles characterised by their aspect ratio, i.e. the ratio of sizes in the longest to the shortest dimension
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Peptides Or Proteins (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0503618A FR2884244B1 (fr) | 2005-04-12 | 2005-04-12 | Obtention de cristaux de dinitroamidure d'ammonium (adn); cristaux d'adn et les composites energetiques en contenant. |
| FR0503618 | 2005-04-12 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| RU2007135718A RU2007135718A (ru) | 2009-05-20 |
| RU2379231C2 true RU2379231C2 (ru) | 2010-01-20 |
Family
ID=35615519
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2007135718/15A RU2379231C2 (ru) | 2005-04-12 | 2006-04-11 | Способ получения кристаллов динитрамида аммония (adn), кристаллы adn и содержащий их энергетический композит |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US7789980B2 (enExample) |
| EP (1) | EP1868940B1 (enExample) |
| JP (1) | JP5291455B2 (enExample) |
| FR (1) | FR2884244B1 (enExample) |
| NO (1) | NO20075094L (enExample) |
| RU (1) | RU2379231C2 (enExample) |
| WO (1) | WO2006108991A2 (enExample) |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2935969B1 (fr) * | 2008-09-18 | 2011-05-13 | Snpe Materiaux Energetiques | Procede d'obtention de cristaux d'adn par cristallisation en milieu visqueux ; cristaux d'adn et les materiaux energetiques en contenant |
| US8873507B2 (en) * | 2009-10-02 | 2014-10-28 | Futurewei Technologies, Inc. | Distributed local mobility anchors for achieving optimized mobility routing |
| FR2973799B1 (fr) | 2011-04-08 | 2013-05-10 | Sme | Procede d'obtention d'une charge de cristaux d'hexanitrohexaazaisowurtzitane a morphologie arrondie ; charge et materiau energetique correspondant |
| CN103880569B (zh) * | 2014-03-24 | 2016-04-27 | 西安近代化学研究所 | 一种二硝酰胺铵表面改性包覆方法 |
| FR3019177A1 (fr) | 2014-03-28 | 2015-10-02 | Eurenco France | Procede et dispositif pour la recuperation, a partir de suspensions renfermant des charges explosives, desdites charges explosives, seches |
| FR3027598B1 (fr) | 2014-10-28 | 2018-05-04 | Arianegroup Sas | Produit pyrotechnique composite avec charges d'adn et de rdx dans un liant de type pag et sa preparation |
| FR3081864B1 (fr) * | 2018-05-30 | 2022-03-18 | Arianegroup Sas | Obtention de cristaux de dinitroamidure d'ammmonium (adn) ; cristaux d'adn et les composites energetiques en contenant |
| CN110422859B (zh) * | 2019-07-24 | 2023-04-07 | 北京理工大学 | 一种准球形二硝酰胺铵晶体的制备方法 |
| CN113336241B (zh) * | 2021-07-08 | 2022-04-01 | 北京理工大学 | 一种高能氧化剂二硝酰胺铵的分离制备的方法 |
| CN116062774B (zh) * | 2021-11-03 | 2024-11-26 | 中国科学院大连化学物理研究所 | 一种高纯度二硝酰胺铵的合成方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1999001409A1 (en) * | 1997-07-03 | 1999-01-14 | Akiko Itai | Method for inferring protein functions with the use of ligand data base |
| SE512395C2 (sv) * | 1999-03-08 | 2000-03-13 | Foersvarets Forskningsanstalt | Sätt att framställa ADN-prills samt bimodal blandning innehållande sådana prills |
| EP1331213A2 (de) * | 2002-01-19 | 2003-07-30 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Verfahren zur Herstellung von mit additiven versetztem Ammoniumdinitramid (ADN) |
| RU2215724C2 (ru) * | 1996-06-11 | 2003-11-10 | Юнайтед Текнолоджиз Корпорейшн | Способ получения сферических частиц энергетических соединений |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6136115A (en) * | 1997-07-02 | 2000-10-24 | Cordant Technologies Inc. | Thermally-stabilized prilled ammonium dinitramide particles, and process for making the same |
| DE19816853A1 (de) * | 1998-04-16 | 1999-10-21 | Fraunhofer Ges Forschung | Verfahren zur Herstellung von Partikeln schmelzfähiger Treib- und Explosivstoffe |
| US6787119B2 (en) * | 2002-03-13 | 2004-09-07 | Council Of Scientific And Industrial Research | Process for the preparation of dinitramidic acid and salts thereof |
-
2005
- 2005-04-12 FR FR0503618A patent/FR2884244B1/fr not_active Expired - Fee Related
-
2006
- 2006-04-11 WO PCT/FR2006/050326 patent/WO2006108991A2/fr not_active Ceased
- 2006-04-11 RU RU2007135718/15A patent/RU2379231C2/ru not_active IP Right Cessation
- 2006-04-11 EP EP06726332.7A patent/EP1868940B1/fr not_active Not-in-force
- 2006-04-11 JP JP2008505941A patent/JP5291455B2/ja not_active Expired - Fee Related
- 2006-04-11 US US11/918,018 patent/US7789980B2/en not_active Expired - Fee Related
-
2007
- 2007-10-09 NO NO20075094A patent/NO20075094L/no not_active Application Discontinuation
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2215724C2 (ru) * | 1996-06-11 | 2003-11-10 | Юнайтед Текнолоджиз Корпорейшн | Способ получения сферических частиц энергетических соединений |
| WO1999001409A1 (en) * | 1997-07-03 | 1999-01-14 | Akiko Itai | Method for inferring protein functions with the use of ligand data base |
| SE512395C2 (sv) * | 1999-03-08 | 2000-03-13 | Foersvarets Forskningsanstalt | Sätt att framställa ADN-prills samt bimodal blandning innehållande sådana prills |
| EP1331213A2 (de) * | 2002-01-19 | 2003-07-30 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Verfahren zur Herstellung von mit additiven versetztem Ammoniumdinitramid (ADN) |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2006108991A3 (fr) | 2007-03-22 |
| NO20075094L (no) | 2008-01-09 |
| FR2884244A1 (fr) | 2006-10-13 |
| US20090090441A1 (en) | 2009-04-09 |
| RU2007135718A (ru) | 2009-05-20 |
| JP5291455B2 (ja) | 2013-09-18 |
| EP1868940A2 (fr) | 2007-12-26 |
| JP2008537723A (ja) | 2008-09-25 |
| EP1868940B1 (fr) | 2017-09-20 |
| FR2884244B1 (fr) | 2007-07-06 |
| WO2006108991A2 (fr) | 2006-10-19 |
| US7789980B2 (en) | 2010-09-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2379231C2 (ru) | Способ получения кристаллов динитрамида аммония (adn), кристаллы adn и содержащий их энергетический композит | |
| US4300962A (en) | Ammonium nitrate explosive systems | |
| US4450110A (en) | Azido nitramine | |
| JPH11217212A (ja) | 低シリカフォージャサイト型ゼオライトおよびその製造方法 | |
| CZ297535B6 (cs) | Způsob krystalizace inhibitoru na bázi benzoxazinonu | |
| WO1996007331A1 (en) | Composition comprising co-crystals, method for its manufacture, and its use | |
| JP2007531683A (ja) | α−アランの製造方法 | |
| US5145535A (en) | Method for intermolecular explosive with viscosity modifier | |
| US8491736B2 (en) | Method for obtaining ADN crystals through crystallization in a viscous medium | |
| JPH10259085A (ja) | 低残渣エアバッグ用ガス発生剤組成物 | |
| CN106187927B (zh) | 一种Lesinurad中间体的制备方法 | |
| AU2015259339B2 (en) | Method for preparation of silver azide | |
| JP2025031139A (ja) | ジヒドロスフィンゴミエリンの結晶、及び、その製造方法、並びに、医薬組成物 | |
| JPH0217199A (ja) | 2’−デオキシ−β−アデノシンの製造方法 | |
| CN101628927B (zh) | 以1,3,5-三-O-苯甲酰基-α-D-呋喃核糖为原料立体选择性制备β-二氟胞苷盐酸盐的方法 | |
| US3291664A (en) | Preparation of explosive substances containing carboxymethyl cellulose | |
| RU2161123C1 (ru) | Способ получения окисленного графита | |
| NL2032474B1 (en) | Preparation method of perdeuterated 1,5-diaminotetrazole | |
| RU2211822C2 (ru) | Способ получения гранулированного гексогена | |
| CN108238609B (zh) | 一种四水八硼酸钠的制备方法 | |
| RU2433993C2 (ru) | Соли стифниновой кислоты | |
| US4786754A (en) | Crystalline upsilon-form bestatin and processes for its preparation | |
| CN1067646A (zh) | 无梯岩石硝铵炸药 | |
| US3101379A (en) | Synthesis of hexanitroethane | |
| US8016959B2 (en) | Thermal pre-ignition agents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PD4A | Correction of name of patent owner | ||
| PC43 | Official registration of the transfer of the exclusive right without contract for inventions |
Effective date: 20140225 |
|
| MM4A | The patent is invalid due to non-payment of fees |
Effective date: 20170412 |