KR20220156808A - laminate - Google Patents
laminate Download PDFInfo
- Publication number
- KR20220156808A KR20220156808A KR1020227028038A KR20227028038A KR20220156808A KR 20220156808 A KR20220156808 A KR 20220156808A KR 1020227028038 A KR1020227028038 A KR 1020227028038A KR 20227028038 A KR20227028038 A KR 20227028038A KR 20220156808 A KR20220156808 A KR 20220156808A
- Authority
- KR
- South Korea
- Prior art keywords
- polyimide film
- film
- self
- acid
- bis
- Prior art date
Links
- 229920001721 polyimide Polymers 0.000 claims abstract description 142
- 239000002904 solvent Substances 0.000 claims abstract description 30
- 239000000203 mixture Substances 0.000 claims description 26
- 238000004132 cross linking Methods 0.000 claims description 22
- 229920000642 polymer Polymers 0.000 claims description 18
- 238000000034 method Methods 0.000 claims description 17
- 238000002834 transmittance Methods 0.000 claims description 17
- 229910003460 diamond Inorganic materials 0.000 claims description 3
- 239000010432 diamond Substances 0.000 claims description 3
- 239000000126 substance Substances 0.000 abstract description 17
- 238000005452 bending Methods 0.000 abstract description 8
- 238000012545 processing Methods 0.000 abstract description 5
- 239000010410 layer Substances 0.000 description 52
- -1 polyethylene terephthalate Polymers 0.000 description 42
- 239000004642 Polyimide Substances 0.000 description 31
- 238000001035 drying Methods 0.000 description 30
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 27
- 229920005575 poly(amic acid) Polymers 0.000 description 26
- 238000004519 manufacturing process Methods 0.000 description 21
- 238000000576 coating method Methods 0.000 description 18
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 16
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 16
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 14
- 238000010438 heat treatment Methods 0.000 description 13
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- 150000008065 acid anhydrides Chemical class 0.000 description 12
- 239000013557 residual solvent Substances 0.000 description 12
- 239000011248 coating agent Substances 0.000 description 11
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 10
- 238000003756 stirring Methods 0.000 description 10
- NVKGJHAQGWCWDI-UHFFFAOYSA-N 4-[4-amino-2-(trifluoromethyl)phenyl]-3-(trifluoromethyl)aniline Chemical group FC(F)(F)C1=CC(N)=CC=C1C1=CC=C(N)C=C1C(F)(F)F NVKGJHAQGWCWDI-UHFFFAOYSA-N 0.000 description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 9
- 229920000570 polyether Polymers 0.000 description 9
- 150000000000 tetracarboxylic acids Chemical class 0.000 description 9
- 239000004962 Polyamide-imide Substances 0.000 description 8
- 150000004984 aromatic diamines Chemical class 0.000 description 8
- 125000005442 diisocyanate group Chemical group 0.000 description 8
- 229910052757 nitrogen Inorganic materials 0.000 description 8
- 229920002312 polyamide-imide Polymers 0.000 description 8
- 238000005259 measurement Methods 0.000 description 7
- 238000002360 preparation method Methods 0.000 description 7
- 239000001294 propane Substances 0.000 description 7
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 6
- 239000004721 Polyphenylene oxide Substances 0.000 description 6
- 239000002253 acid Substances 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 229920001577 copolymer Polymers 0.000 description 6
- 238000006358 imidation reaction Methods 0.000 description 6
- 238000006116 polymerization reaction Methods 0.000 description 6
- AIVVXPSKEVWKMY-UHFFFAOYSA-N 4-(3,4-dicarboxyphenoxy)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1OC1=CC=C(C(O)=O)C(C(O)=O)=C1 AIVVXPSKEVWKMY-UHFFFAOYSA-N 0.000 description 5
- YGYCECQIOXZODZ-UHFFFAOYSA-N 4415-87-6 Chemical compound O=C1OC(=O)C2C1C1C(=O)OC(=O)C12 YGYCECQIOXZODZ-UHFFFAOYSA-N 0.000 description 5
- QQGYZOYWNCKGEK-UHFFFAOYSA-N 5-[(1,3-dioxo-2-benzofuran-5-yl)oxy]-2-benzofuran-1,3-dione Chemical compound C1=C2C(=O)OC(=O)C2=CC(OC=2C=C3C(=O)OC(C3=CC=2)=O)=C1 QQGYZOYWNCKGEK-UHFFFAOYSA-N 0.000 description 5
- 239000001273 butane Substances 0.000 description 5
- 150000001991 dicarboxylic acids Chemical class 0.000 description 5
- IJDNQMDRQITEOD-UHFFFAOYSA-N n-butane Chemical compound CCCC IJDNQMDRQITEOD-UHFFFAOYSA-N 0.000 description 5
- OFBQJSOFQDEBGM-UHFFFAOYSA-N n-pentane Natural products CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 229920006299 self-healing polymer Polymers 0.000 description 5
- 230000003746 surface roughness Effects 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 4
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 4
- MHABMANUFPZXEB-UHFFFAOYSA-N O-demethyl-aloesaponarin I Natural products O=C1C2=CC=CC(O)=C2C(=O)C2=C1C=C(O)C(C(O)=O)=C2C MHABMANUFPZXEB-UHFFFAOYSA-N 0.000 description 4
- KKEYFWRCBNTPAC-UHFFFAOYSA-N Terephthalic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-N 0.000 description 4
- 150000008064 anhydrides Chemical group 0.000 description 4
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 150000004985 diamines Chemical class 0.000 description 4
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 229910021645 metal ion Inorganic materials 0.000 description 4
- 230000000149 penetrating effect Effects 0.000 description 4
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 239000002243 precursor Substances 0.000 description 4
- 238000010926 purge Methods 0.000 description 4
- 238000011084 recovery Methods 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 3
- LJGHYPLBDBRCRZ-UHFFFAOYSA-N 3-(3-aminophenyl)sulfonylaniline Chemical compound NC1=CC=CC(S(=O)(=O)C=2C=C(N)C=CC=2)=C1 LJGHYPLBDBRCRZ-UHFFFAOYSA-N 0.000 description 3
- UITKHKNFVCYWNG-UHFFFAOYSA-N 4-(3,4-dicarboxybenzoyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1 UITKHKNFVCYWNG-UHFFFAOYSA-N 0.000 description 3
- XPAQFJJCWGSXGJ-UHFFFAOYSA-N 4-amino-n-(4-aminophenyl)benzamide Chemical compound C1=CC(N)=CC=C1NC(=O)C1=CC=C(N)C=C1 XPAQFJJCWGSXGJ-UHFFFAOYSA-N 0.000 description 3
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- MQJKPEGWNLWLTK-UHFFFAOYSA-N Dapsone Chemical compound C1=CC(N)=CC=C1S(=O)(=O)C1=CC=C(N)C=C1 MQJKPEGWNLWLTK-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 239000004952 Polyamide Substances 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- BDWOQDZGSYLSCZ-UHFFFAOYSA-N [1,3]oxazolo[4,5-f][1,3]benzoxazole Chemical compound C1=C2OC=NC2=CC2=C1OC=N2 BDWOQDZGSYLSCZ-UHFFFAOYSA-N 0.000 description 3
- PMJNNCUVWHTTMV-UHFFFAOYSA-N [1,3]oxazolo[5,4-f][1,3]benzoxazole Chemical compound C1=C2OC=NC2=CC2=C1N=CO2 PMJNNCUVWHTTMV-UHFFFAOYSA-N 0.000 description 3
- 229920003232 aliphatic polyester Polymers 0.000 description 3
- JFDZBHWFFUWGJE-UHFFFAOYSA-N benzonitrile Chemical compound N#CC1=CC=CC=C1 JFDZBHWFFUWGJE-UHFFFAOYSA-N 0.000 description 3
- 239000011247 coating layer Substances 0.000 description 3
- VKIRRGRTJUUZHS-UHFFFAOYSA-N cyclohexane-1,4-diamine Chemical compound NC1CCC(N)CC1 VKIRRGRTJUUZHS-UHFFFAOYSA-N 0.000 description 3
- 230000001747 exhibiting effect Effects 0.000 description 3
- 239000012948 isocyanate Substances 0.000 description 3
- 150000002513 isocyanates Chemical class 0.000 description 3
- 239000011254 layer-forming composition Substances 0.000 description 3
- 239000012299 nitrogen atmosphere Substances 0.000 description 3
- 229920002647 polyamide Polymers 0.000 description 3
- 229920000728 polyester Polymers 0.000 description 3
- 229920006267 polyester film Polymers 0.000 description 3
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 3
- 229920001169 thermoplastic Polymers 0.000 description 3
- DVKJHBMWWAPEIU-UHFFFAOYSA-N toluene 2,4-diisocyanate Chemical compound CC1=CC=C(N=C=O)C=C1N=C=O DVKJHBMWWAPEIU-UHFFFAOYSA-N 0.000 description 3
- 150000003628 tricarboxylic acids Chemical class 0.000 description 3
- 125000000355 1,3-benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 2
- PXGZQGDTEZPERC-UHFFFAOYSA-N 1,4-cyclohexanedicarboxylic acid Chemical compound OC(=O)C1CCC(C(O)=O)CC1 PXGZQGDTEZPERC-UHFFFAOYSA-N 0.000 description 2
- CDMDQYCEEKCBGR-UHFFFAOYSA-N 1,4-diisocyanatocyclohexane Chemical compound O=C=NC1CCC(N=C=O)CC1 CDMDQYCEEKCBGR-UHFFFAOYSA-N 0.000 description 2
- MXPYJVUYLVNEBB-UHFFFAOYSA-N 2-[2-(2-carboxybenzoyl)oxycarbonylbenzoyl]oxycarbonylbenzoic acid Chemical compound OC(=O)C1=CC=CC=C1C(=O)OC(=O)C1=CC=CC=C1C(=O)OC(=O)C1=CC=CC=C1C(O)=O MXPYJVUYLVNEBB-UHFFFAOYSA-N 0.000 description 2
- WXUAQHNMJWJLTG-UHFFFAOYSA-N 2-methylbutanedioic acid Chemical compound OC(=O)C(C)CC(O)=O WXUAQHNMJWJLTG-UHFFFAOYSA-N 0.000 description 2
- SFHLLWPKGUSQIK-UHFFFAOYSA-N 2-methylcyclohexane-1,4-diamine Chemical compound CC1CC(N)CCC1N SFHLLWPKGUSQIK-UHFFFAOYSA-N 0.000 description 2
- LJMPOXUWPWEILS-UHFFFAOYSA-N 3a,4,4a,7a,8,8a-hexahydrofuro[3,4-f][2]benzofuran-1,3,5,7-tetrone Chemical compound C1C2C(=O)OC(=O)C2CC2C(=O)OC(=O)C21 LJMPOXUWPWEILS-UHFFFAOYSA-N 0.000 description 2
- UPMLOUAZCHDJJD-UHFFFAOYSA-N 4,4'-Diphenylmethane Diisocyanate Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=C(N=C=O)C=C1 UPMLOUAZCHDJJD-UHFFFAOYSA-N 0.000 description 2
- HYDATEKARGDBKU-UHFFFAOYSA-N 4-[4-[4-(4-aminophenoxy)phenyl]phenoxy]aniline Chemical group C1=CC(N)=CC=C1OC1=CC=C(C=2C=CC(OC=3C=CC(N)=CC=3)=CC=2)C=C1 HYDATEKARGDBKU-UHFFFAOYSA-N 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 2
- WVOLTBSCXRRQFR-SJORKVTESA-N Cannabidiolic acid Natural products OC1=C(C(O)=O)C(CCCCC)=CC(O)=C1[C@@H]1[C@@H](C(C)=C)CCC(C)=C1 WVOLTBSCXRRQFR-SJORKVTESA-N 0.000 description 2
- 229920002284 Cellulose triacetate Polymers 0.000 description 2
- 229920000858 Cyclodextrin Polymers 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- OTMSDBZUPAUEDD-UHFFFAOYSA-N Ethane Chemical compound CC OTMSDBZUPAUEDD-UHFFFAOYSA-N 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- DAKWPKUUDNSNPN-UHFFFAOYSA-N Trimethylolpropane triacrylate Chemical compound C=CC(=O)OCC(CC)(COC(=O)C=C)COC(=O)C=C DAKWPKUUDNSNPN-UHFFFAOYSA-N 0.000 description 2
- NNLVGZFZQQXQNW-ADJNRHBOSA-N [(2r,3r,4s,5r,6s)-4,5-diacetyloxy-3-[(2s,3r,4s,5r,6r)-3,4,5-triacetyloxy-6-(acetyloxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6s)-4,5,6-triacetyloxy-2-(acetyloxymethyl)oxan-3-yl]oxyoxan-2-yl]methyl acetate Chemical compound O([C@@H]1O[C@@H]([C@H]([C@H](OC(C)=O)[C@H]1OC(C)=O)O[C@H]1[C@@H]([C@@H](OC(C)=O)[C@H](OC(C)=O)[C@@H](COC(C)=O)O1)OC(C)=O)COC(=O)C)[C@@H]1[C@@H](COC(C)=O)O[C@@H](OC(C)=O)[C@H](OC(C)=O)[C@H]1OC(C)=O NNLVGZFZQQXQNW-ADJNRHBOSA-N 0.000 description 2
- 239000002390 adhesive tape Substances 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- TUQQUUXMCKXGDI-UHFFFAOYSA-N bis(3-aminophenyl)methanone Chemical compound NC1=CC=CC(C(=O)C=2C=C(N)C=CC=2)=C1 TUQQUUXMCKXGDI-UHFFFAOYSA-N 0.000 description 2
- WVOLTBSCXRRQFR-DLBZAZTESA-N cannabidiolic acid Chemical compound OC1=C(C(O)=O)C(CCCCC)=CC(O)=C1[C@H]1[C@H](C(C)=C)CCC(C)=C1 WVOLTBSCXRRQFR-DLBZAZTESA-N 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- SMEJCQZFRMVYGC-UHFFFAOYSA-N cyclohexane-1,2,3,4-tetracarboxylic acid Chemical compound OC(=O)C1CCC(C(O)=O)C(C(O)=O)C1C(O)=O SMEJCQZFRMVYGC-UHFFFAOYSA-N 0.000 description 2
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 2
- BGTOWKSIORTVQH-UHFFFAOYSA-N cyclopentanone Chemical compound O=C1CCCC1 BGTOWKSIORTVQH-UHFFFAOYSA-N 0.000 description 2
- 230000018044 dehydration Effects 0.000 description 2
- 238000006297 dehydration reaction Methods 0.000 description 2
- 230000006866 deterioration Effects 0.000 description 2
- 125000006159 dianhydride group Chemical group 0.000 description 2
- KZTYYGOKRVBIMI-UHFFFAOYSA-N diphenyl sulfone Chemical compound C=1C=CC=CC=1S(=O)(=O)C1=CC=CC=C1 KZTYYGOKRVBIMI-UHFFFAOYSA-N 0.000 description 2
- 238000006073 displacement reaction Methods 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000010419 fine particle Substances 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- 239000003999 initiator Substances 0.000 description 2
- QQVIHTHCMHWDBS-UHFFFAOYSA-N isophthalic acid Chemical compound OC(=O)C1=CC=CC(C(O)=O)=C1 QQVIHTHCMHWDBS-UHFFFAOYSA-N 0.000 description 2
- AWJUIBRHMBBTKR-UHFFFAOYSA-N isoquinoline Chemical compound C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 238000000691 measurement method Methods 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- 229910000096 monohydride Inorganic materials 0.000 description 2
- BDJRBEYXGGNYIS-UHFFFAOYSA-N nonanedioic acid Chemical compound OC(=O)CCCCCCCC(O)=O BDJRBEYXGGNYIS-UHFFFAOYSA-N 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- WLJVNTCWHIRURA-UHFFFAOYSA-N pimelic acid Chemical compound OC(=O)CCCCCC(O)=O WLJVNTCWHIRURA-UHFFFAOYSA-N 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 229920000139 polyethylene terephthalate Polymers 0.000 description 2
- 239000005020 polyethylene terephthalate Substances 0.000 description 2
- 230000000379 polymerizing effect Effects 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 239000004814 polyurethane Substances 0.000 description 2
- 229920002635 polyurethane Polymers 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- CYIDZMCFTVVTJO-UHFFFAOYSA-N pyromellitic acid Chemical compound OC(=O)C1=CC(C(O)=O)=C(C(O)=O)C=C1C(O)=O CYIDZMCFTVVTJO-UHFFFAOYSA-N 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 238000006798 ring closing metathesis reaction Methods 0.000 description 2
- CXMXRPHRNRROMY-UHFFFAOYSA-N sebacic acid Chemical compound OC(=O)CCCCCCCCC(O)=O CXMXRPHRNRROMY-UHFFFAOYSA-N 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- TYFQFVWCELRYAO-UHFFFAOYSA-N suberic acid Chemical compound OC(=O)CCCCCCC(O)=O TYFQFVWCELRYAO-UHFFFAOYSA-N 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 229920005992 thermoplastic resin Polymers 0.000 description 2
- RUELTTOHQODFPA-UHFFFAOYSA-N toluene 2,6-diisocyanate Chemical compound CC1=C(N=C=O)C=CC=C1N=C=O RUELTTOHQODFPA-UHFFFAOYSA-N 0.000 description 2
- ARCGXLSVLAOJQL-UHFFFAOYSA-N trimellitic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1 ARCGXLSVLAOJQL-UHFFFAOYSA-N 0.000 description 2
- SRPWOOOHEPICQU-UHFFFAOYSA-N trimellitic anhydride Chemical compound OC(=O)C1=CC=C2C(=O)OC(=O)C2=C1 SRPWOOOHEPICQU-UHFFFAOYSA-N 0.000 description 2
- LWBHHRRTOZQPDM-UHFFFAOYSA-N undecanedioic acid Chemical compound OC(=O)CCCCCCCCCC(O)=O LWBHHRRTOZQPDM-UHFFFAOYSA-N 0.000 description 2
- STIUJDCDGZSXGO-UHFFFAOYSA-N (3-amino-4-phenoxyphenyl)-(3-aminophenyl)methanone Chemical compound NC1=CC=CC(C(=O)C=2C=C(N)C(OC=3C=CC=CC=3)=CC=2)=C1 STIUJDCDGZSXGO-UHFFFAOYSA-N 0.000 description 1
- GSHMRKDZYYLPNZ-UHFFFAOYSA-N (3-amino-4-phenoxyphenyl)-(4-amino-3-phenoxyphenyl)methanone Chemical compound NC1=CC=C(C(=O)C=2C=C(N)C(OC=3C=CC=CC=3)=CC=2)C=C1OC1=CC=CC=C1 GSHMRKDZYYLPNZ-UHFFFAOYSA-N 0.000 description 1
- PHPTWVBSQRENOR-UHFFFAOYSA-N (3-amino-4-phenoxyphenyl)-(4-aminophenyl)methanone Chemical compound C1=CC(N)=CC=C1C(=O)C(C=C1N)=CC=C1OC1=CC=CC=C1 PHPTWVBSQRENOR-UHFFFAOYSA-N 0.000 description 1
- YKNMIGJJXKBHJE-UHFFFAOYSA-N (3-aminophenyl)-(4-aminophenyl)methanone Chemical compound C1=CC(N)=CC=C1C(=O)C1=CC=CC(N)=C1 YKNMIGJJXKBHJE-UHFFFAOYSA-N 0.000 description 1
- HFAMSBMTCKNPRG-UHFFFAOYSA-N (4-amino-3-phenoxyphenyl)-(3-aminophenyl)methanone Chemical compound NC1=CC=CC(C(=O)C=2C=C(OC=3C=CC=CC=3)C(N)=CC=2)=C1 HFAMSBMTCKNPRG-UHFFFAOYSA-N 0.000 description 1
- NILYJZJYFZUPPO-UHFFFAOYSA-N (4-amino-3-phenoxyphenyl)-(4-aminophenyl)methanone Chemical compound C1=CC(N)=CC=C1C(=O)C1=CC=C(N)C(OC=2C=CC=CC=2)=C1 NILYJZJYFZUPPO-UHFFFAOYSA-N 0.000 description 1
- AVQQQNCBBIEMEU-UHFFFAOYSA-N 1,1,3,3-tetramethylurea Chemical compound CN(C)C(=O)N(C)C AVQQQNCBBIEMEU-UHFFFAOYSA-N 0.000 description 1
- GEYOCULIXLDCMW-UHFFFAOYSA-N 1,2-phenylenediamine Chemical compound NC1=CC=CC=C1N GEYOCULIXLDCMW-UHFFFAOYSA-N 0.000 description 1
- CYSGHNMQYZDMIA-UHFFFAOYSA-N 1,3-Dimethyl-2-imidazolidinon Chemical compound CN1CCN(C)C1=O CYSGHNMQYZDMIA-UHFFFAOYSA-N 0.000 description 1
- RTTZISZSHSCFRH-UHFFFAOYSA-N 1,3-bis(isocyanatomethyl)benzene Chemical compound O=C=NCC1=CC=CC(CN=C=O)=C1 RTTZISZSHSCFRH-UHFFFAOYSA-N 0.000 description 1
- WZCQRUWWHSTZEM-UHFFFAOYSA-N 1,3-phenylenediamine Chemical compound NC1=CC=CC(N)=C1 WZCQRUWWHSTZEM-UHFFFAOYSA-N 0.000 description 1
- OHLKMGYGBHFODF-UHFFFAOYSA-N 1,4-bis(isocyanatomethyl)benzene Chemical compound O=C=NCC1=CC=C(CN=C=O)C=C1 OHLKMGYGBHFODF-UHFFFAOYSA-N 0.000 description 1
- 125000001140 1,4-phenylene group Chemical group [H]C1=C([H])C([*:2])=C([H])C([H])=C1[*:1] 0.000 description 1
- CBCKQZAAMUWICA-UHFFFAOYSA-N 1,4-phenylenediamine Chemical compound NC1=CC=C(N)C=C1 CBCKQZAAMUWICA-UHFFFAOYSA-N 0.000 description 1
- GEWWCWZGHNIUBW-UHFFFAOYSA-N 1-(4-nitrophenyl)propan-2-one Chemical compound CC(=O)CC1=CC=C([N+]([O-])=O)C=C1 GEWWCWZGHNIUBW-UHFFFAOYSA-N 0.000 description 1
- 239000012956 1-hydroxycyclohexylphenyl-ketone Substances 0.000 description 1
- LFSYUSUFCBOHGU-UHFFFAOYSA-N 1-isocyanato-2-[(4-isocyanatophenyl)methyl]benzene Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=CC=C1N=C=O LFSYUSUFCBOHGU-UHFFFAOYSA-N 0.000 description 1
- ICLCCFKUSALICQ-UHFFFAOYSA-N 1-isocyanato-4-(4-isocyanato-3-methylphenyl)-2-methylbenzene Chemical compound C1=C(N=C=O)C(C)=CC(C=2C=C(C)C(N=C=O)=CC=2)=C1 ICLCCFKUSALICQ-UHFFFAOYSA-N 0.000 description 1
- QFGCFKJIPBRJGM-UHFFFAOYSA-N 12-[(2-methylpropan-2-yl)oxy]-12-oxododecanoic acid Chemical compound CC(C)(C)OC(=O)CCCCCCCCCCC(O)=O QFGCFKJIPBRJGM-UHFFFAOYSA-N 0.000 description 1
- BEBVJSBFUZVWMS-UHFFFAOYSA-N 2-(2-methylpropyl)cyclohexane-1,4-diamine Chemical compound CC(C)CC1CC(N)CCC1N BEBVJSBFUZVWMS-UHFFFAOYSA-N 0.000 description 1
- IKSUMZCUHPMCQV-UHFFFAOYSA-N 2-(3-aminophenyl)-1,3-benzoxazol-5-amine Chemical compound NC1=CC=CC(C=2OC3=CC=C(N)C=C3N=2)=C1 IKSUMZCUHPMCQV-UHFFFAOYSA-N 0.000 description 1
- VSMRWFMFAFOGGD-UHFFFAOYSA-N 2-(3-aminophenyl)-1,3-benzoxazol-6-amine Chemical compound NC1=CC=CC(C=2OC3=CC(N)=CC=C3N=2)=C1 VSMRWFMFAFOGGD-UHFFFAOYSA-N 0.000 description 1
- UMGYJGHIMRFYSP-UHFFFAOYSA-N 2-(4-aminophenyl)-1,3-benzoxazol-5-amine Chemical compound C1=CC(N)=CC=C1C1=NC2=CC(N)=CC=C2O1 UMGYJGHIMRFYSP-UHFFFAOYSA-N 0.000 description 1
- DIXHWJYQQGNWTI-UHFFFAOYSA-N 2-[4-(5-amino-1,3-benzoxazol-2-yl)phenyl]-1,3-benzoxazol-5-amine Chemical compound NC1=CC=C2OC(C3=CC=C(C=C3)C=3OC4=CC=C(C=C4N=3)N)=NC2=C1 DIXHWJYQQGNWTI-UHFFFAOYSA-N 0.000 description 1
- SFZGLHDSSSDCHH-UHFFFAOYSA-N 2-[4-(6-amino-1,3-benzoxazol-2-yl)phenyl]-1,3-benzoxazol-6-amine Chemical compound C1=C(N)C=C2OC(C3=CC=C(C=C3)C3=NC4=CC=C(C=C4O3)N)=NC2=C1 SFZGLHDSSSDCHH-UHFFFAOYSA-N 0.000 description 1
- IAEVEDMYEYMTSE-UHFFFAOYSA-N 2-butan-2-ylcyclohexane-1,4-diamine Chemical compound CCC(C)C1CC(N)CCC1N IAEVEDMYEYMTSE-UHFFFAOYSA-N 0.000 description 1
- HIEBSORULQQANM-UHFFFAOYSA-N 2-butylcyclohexane-1,4-diamine Chemical compound CCCCC1CC(N)CCC1N HIEBSORULQQANM-UHFFFAOYSA-N 0.000 description 1
- FMZFBOHWVULQIY-UHFFFAOYSA-N 2-ethylcyclohexane-1,4-diamine Chemical compound CCC1CC(N)CCC1N FMZFBOHWVULQIY-UHFFFAOYSA-N 0.000 description 1
- ALBWHZWLFAHNRI-UHFFFAOYSA-N 2-propan-2-ylcyclohexane-1,4-diamine Chemical compound CC(C)C1CC(N)CCC1N ALBWHZWLFAHNRI-UHFFFAOYSA-N 0.000 description 1
- RKBNPLYPKZRNEF-UHFFFAOYSA-N 2-tert-butylcyclohexane-1,4-diamine Chemical compound CC(C)(C)C1CC(N)CCC1N RKBNPLYPKZRNEF-UHFFFAOYSA-N 0.000 description 1
- NBAUUNCGSMAPFM-UHFFFAOYSA-N 3-(3,4-dicarboxyphenyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C1=CC=CC(C(O)=O)=C1C(O)=O NBAUUNCGSMAPFM-UHFFFAOYSA-N 0.000 description 1
- LXJLFVRAWOOQDR-UHFFFAOYSA-N 3-(3-aminophenoxy)aniline Chemical compound NC1=CC=CC(OC=2C=C(N)C=CC=2)=C1 LXJLFVRAWOOQDR-UHFFFAOYSA-N 0.000 description 1
- ZBMISJGHVWNWTE-UHFFFAOYSA-N 3-(4-aminophenoxy)aniline Chemical compound C1=CC(N)=CC=C1OC1=CC=CC(N)=C1 ZBMISJGHVWNWTE-UHFFFAOYSA-N 0.000 description 1
- ZMPZWXKBGSQATE-UHFFFAOYSA-N 3-(4-aminophenyl)sulfonylaniline Chemical compound C1=CC(N)=CC=C1S(=O)(=O)C1=CC=CC(N)=C1 ZMPZWXKBGSQATE-UHFFFAOYSA-N 0.000 description 1
- ZDBWYUOUYNQZBM-UHFFFAOYSA-N 3-(aminomethyl)aniline Chemical compound NCC1=CC=CC(N)=C1 ZDBWYUOUYNQZBM-UHFFFAOYSA-N 0.000 description 1
- RHRNYXVSZLSRRP-UHFFFAOYSA-N 3-(carboxymethyl)cyclopentane-1,2,4-tricarboxylic acid Chemical compound OC(=O)CC1C(C(O)=O)CC(C(O)=O)C1C(O)=O RHRNYXVSZLSRRP-UHFFFAOYSA-N 0.000 description 1
- CKOFBUUFHALZGK-UHFFFAOYSA-N 3-[(3-aminophenyl)methyl]aniline Chemical compound NC1=CC=CC(CC=2C=C(N)C=CC=2)=C1 CKOFBUUFHALZGK-UHFFFAOYSA-N 0.000 description 1
- FGWQCROGAHMWSU-UHFFFAOYSA-N 3-[(4-aminophenyl)methyl]aniline Chemical compound C1=CC(N)=CC=C1CC1=CC=CC(N)=C1 FGWQCROGAHMWSU-UHFFFAOYSA-N 0.000 description 1
- DKKYOQYISDAQER-UHFFFAOYSA-N 3-[3-(3-aminophenoxy)phenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=C(OC=3C=C(N)C=CC=3)C=CC=2)=C1 DKKYOQYISDAQER-UHFFFAOYSA-N 0.000 description 1
- GBUNNYTXPDCASY-UHFFFAOYSA-N 3-[3-[2-[3-(3-aminophenoxy)phenyl]-1,1,1,3,3,3-hexafluoropropan-2-yl]phenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=C(C=CC=2)C(C=2C=C(OC=3C=C(N)C=CC=3)C=CC=2)(C(F)(F)F)C(F)(F)F)=C1 GBUNNYTXPDCASY-UHFFFAOYSA-N 0.000 description 1
- LBPVOEHZEWAJKQ-UHFFFAOYSA-N 3-[4-(3-aminophenoxy)phenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=CC(OC=3C=C(N)C=CC=3)=CC=2)=C1 LBPVOEHZEWAJKQ-UHFFFAOYSA-N 0.000 description 1
- UQHPRIRSWZEGEK-UHFFFAOYSA-N 3-[4-[1-[4-(3-aminophenoxy)phenyl]ethyl]phenoxy]aniline Chemical compound C=1C=C(OC=2C=C(N)C=CC=2)C=CC=1C(C)C(C=C1)=CC=C1OC1=CC=CC(N)=C1 UQHPRIRSWZEGEK-UHFFFAOYSA-N 0.000 description 1
- PHVQYQDTIMAIKY-UHFFFAOYSA-N 3-[4-[1-[4-(3-aminophenoxy)phenyl]propyl]phenoxy]aniline Chemical compound C=1C=C(OC=2C=C(N)C=CC=2)C=CC=1C(CC)C(C=C1)=CC=C1OC1=CC=CC(N)=C1 PHVQYQDTIMAIKY-UHFFFAOYSA-N 0.000 description 1
- MFTFTIALAXXIMU-UHFFFAOYSA-N 3-[4-[2-[4-(3-aminophenoxy)phenyl]-1,1,1,3,3,3-hexafluoropropan-2-yl]phenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=CC(=CC=2)C(C=2C=CC(OC=3C=C(N)C=CC=3)=CC=2)(C(F)(F)F)C(F)(F)F)=C1 MFTFTIALAXXIMU-UHFFFAOYSA-N 0.000 description 1
- BDROEGDWWLIVJF-UHFFFAOYSA-N 3-[4-[2-[4-(3-aminophenoxy)phenyl]ethyl]phenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=CC(CCC=3C=CC(OC=4C=C(N)C=CC=4)=CC=3)=CC=2)=C1 BDROEGDWWLIVJF-UHFFFAOYSA-N 0.000 description 1
- NYRFBMFAUFUULG-UHFFFAOYSA-N 3-[4-[2-[4-(3-aminophenoxy)phenyl]propan-2-yl]phenoxy]aniline Chemical compound C=1C=C(OC=2C=C(N)C=CC=2)C=CC=1C(C)(C)C(C=C1)=CC=C1OC1=CC=CC(N)=C1 NYRFBMFAUFUULG-UHFFFAOYSA-N 0.000 description 1
- TZFAMRKTHYOODK-UHFFFAOYSA-N 3-[4-[3-[4-(3-aminophenoxy)phenyl]propyl]phenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=CC(CCCC=3C=CC(OC=4C=C(N)C=CC=4)=CC=3)=CC=2)=C1 TZFAMRKTHYOODK-UHFFFAOYSA-N 0.000 description 1
- UCQABCHSIIXVOY-UHFFFAOYSA-N 3-[4-[4-(3-aminophenoxy)phenyl]phenoxy]aniline Chemical group NC1=CC=CC(OC=2C=CC(=CC=2)C=2C=CC(OC=3C=C(N)C=CC=3)=CC=2)=C1 UCQABCHSIIXVOY-UHFFFAOYSA-N 0.000 description 1
- JERFEOKUSPGKGV-UHFFFAOYSA-N 3-[4-[4-(3-aminophenoxy)phenyl]sulfanylphenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=CC(SC=3C=CC(OC=4C=C(N)C=CC=4)=CC=3)=CC=2)=C1 JERFEOKUSPGKGV-UHFFFAOYSA-N 0.000 description 1
- VTHWGYHNEDIPTO-UHFFFAOYSA-N 3-[4-[4-(3-aminophenoxy)phenyl]sulfinylphenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=CC(=CC=2)S(=O)C=2C=CC(OC=3C=C(N)C=CC=3)=CC=2)=C1 VTHWGYHNEDIPTO-UHFFFAOYSA-N 0.000 description 1
- WCXGOVYROJJXHA-UHFFFAOYSA-N 3-[4-[4-(3-aminophenoxy)phenyl]sulfonylphenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=CC(=CC=2)S(=O)(=O)C=2C=CC(OC=3C=C(N)C=CC=3)=CC=2)=C1 WCXGOVYROJJXHA-UHFFFAOYSA-N 0.000 description 1
- YSMXOEWEUZTWAK-UHFFFAOYSA-N 3-[4-[[4-(3-aminophenoxy)phenyl]methyl]phenoxy]aniline Chemical compound NC1=CC=CC(OC=2C=CC(CC=3C=CC(OC=4C=C(N)C=CC=4)=CC=3)=CC=2)=C1 YSMXOEWEUZTWAK-UHFFFAOYSA-N 0.000 description 1
- RXXCIBALSKQCAE-UHFFFAOYSA-N 3-methylbutoxymethylbenzene Chemical compound CC(C)CCOCC1=CC=CC=C1 RXXCIBALSKQCAE-UHFFFAOYSA-N 0.000 description 1
- YBRVSVVVWCFQMG-UHFFFAOYSA-N 4,4'-diaminodiphenylmethane Chemical compound C1=CC(N)=CC=C1CC1=CC=C(N)C=C1 YBRVSVVVWCFQMG-UHFFFAOYSA-N 0.000 description 1
- BKQWDTFZUNGWNV-UHFFFAOYSA-N 4-(3,4-dicarboxycyclohexyl)cyclohexane-1,2-dicarboxylic acid Chemical compound C1C(C(O)=O)C(C(=O)O)CCC1C1CC(C(O)=O)C(C(O)=O)CC1 BKQWDTFZUNGWNV-UHFFFAOYSA-N 0.000 description 1
- LFBALUPVVFCEPA-UHFFFAOYSA-N 4-(3,4-dicarboxyphenyl)phthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1C1=CC=C(C(O)=O)C(C(O)=O)=C1 LFBALUPVVFCEPA-UHFFFAOYSA-N 0.000 description 1
- AVCOFPOLGHKJQB-UHFFFAOYSA-N 4-(3,4-dicarboxyphenyl)sulfonylphthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1S(=O)(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1 AVCOFPOLGHKJQB-UHFFFAOYSA-N 0.000 description 1
- QYIMZXITLDTULQ-UHFFFAOYSA-N 4-(4-amino-2-methylphenyl)-3-methylaniline Chemical group CC1=CC(N)=CC=C1C1=CC=C(N)C=C1C QYIMZXITLDTULQ-UHFFFAOYSA-N 0.000 description 1
- MITHMOYLTXMLRB-UHFFFAOYSA-N 4-(4-aminophenyl)sulfinylaniline Chemical compound C1=CC(N)=CC=C1S(=O)C1=CC=C(N)C=C1 MITHMOYLTXMLRB-UHFFFAOYSA-N 0.000 description 1
- NEQFBGHQPUXOFH-UHFFFAOYSA-N 4-(4-carboxyphenyl)benzoic acid Chemical compound C1=CC(C(=O)O)=CC=C1C1=CC=C(C(O)=O)C=C1 NEQFBGHQPUXOFH-UHFFFAOYSA-N 0.000 description 1
- BFWYZZPDZZGSLJ-UHFFFAOYSA-N 4-(aminomethyl)aniline Chemical compound NCC1=CC=C(N)C=C1 BFWYZZPDZZGSLJ-UHFFFAOYSA-N 0.000 description 1
- HLBLWEWZXPIGSM-UHFFFAOYSA-N 4-Aminophenyl ether Chemical compound C1=CC(N)=CC=C1OC1=CC=C(N)C=C1 HLBLWEWZXPIGSM-UHFFFAOYSA-N 0.000 description 1
- JHCBFGGESJQAIQ-UHFFFAOYSA-N 4-[(4-amino-3,5-dimethylcyclohexyl)methyl]-2,6-dimethylcyclohexan-1-amine Chemical compound C1C(C)C(N)C(C)CC1CC1CC(C)C(N)C(C)C1 JHCBFGGESJQAIQ-UHFFFAOYSA-N 0.000 description 1
- HESXPOICBNWMPI-UHFFFAOYSA-N 4-[2-[4-[2-(4-aminophenyl)propan-2-yl]phenyl]propan-2-yl]aniline Chemical compound C=1C=C(C(C)(C)C=2C=CC(N)=CC=2)C=CC=1C(C)(C)C1=CC=C(N)C=C1 HESXPOICBNWMPI-UHFFFAOYSA-N 0.000 description 1
- JCRRFJIVUPSNTA-UHFFFAOYSA-N 4-[4-(4-aminophenoxy)phenoxy]aniline Chemical compound C1=CC(N)=CC=C1OC(C=C1)=CC=C1OC1=CC=C(N)C=C1 JCRRFJIVUPSNTA-UHFFFAOYSA-N 0.000 description 1
- QXCRYCTXLXDDST-UHFFFAOYSA-N 4-[4-[2-[3-[2-[4-(4-amino-2-fluorophenoxy)phenyl]propan-2-yl]phenyl]propan-2-yl]phenoxy]-3-fluoroaniline Chemical compound C=1C=CC(C(C)(C)C=2C=CC(OC=3C(=CC(N)=CC=3)F)=CC=2)=CC=1C(C)(C)C(C=C1)=CC=C1OC1=CC=C(N)C=C1F QXCRYCTXLXDDST-UHFFFAOYSA-N 0.000 description 1
- QJZDVFLOHJFIAK-UHFFFAOYSA-N 4-[4-[2-[3-[2-[4-(4-amino-2-methylphenoxy)phenyl]propan-2-yl]phenyl]propan-2-yl]phenoxy]-3-methylaniline Chemical compound CC1=CC(N)=CC=C1OC1=CC=C(C(C)(C)C=2C=C(C=CC=2)C(C)(C)C=2C=CC(OC=3C(=CC(N)=CC=3)C)=CC=2)C=C1 QJZDVFLOHJFIAK-UHFFFAOYSA-N 0.000 description 1
- RIMWCPQXJPPTHR-UHFFFAOYSA-N 4-[4-[2-[3-[2-[4-[4-amino-2-(trifluoromethyl)phenoxy]phenyl]propan-2-yl]phenyl]propan-2-yl]phenoxy]-3-(trifluoromethyl)aniline Chemical compound C=1C=CC(C(C)(C)C=2C=CC(OC=3C(=CC(N)=CC=3)C(F)(F)F)=CC=2)=CC=1C(C)(C)C(C=C1)=CC=C1OC1=CC=C(N)C=C1C(F)(F)F RIMWCPQXJPPTHR-UHFFFAOYSA-N 0.000 description 1
- HHLMWQDRYZAENA-UHFFFAOYSA-N 4-[4-[2-[4-(4-aminophenoxy)phenyl]-1,1,1,3,3,3-hexafluoropropan-2-yl]phenoxy]aniline Chemical compound C1=CC(N)=CC=C1OC1=CC=C(C(C=2C=CC(OC=3C=CC(N)=CC=3)=CC=2)(C(F)(F)F)C(F)(F)F)C=C1 HHLMWQDRYZAENA-UHFFFAOYSA-N 0.000 description 1
- KMKWGXGSGPYISJ-UHFFFAOYSA-N 4-[4-[2-[4-(4-aminophenoxy)phenyl]propan-2-yl]phenoxy]aniline Chemical compound C=1C=C(OC=2C=CC(N)=CC=2)C=CC=1C(C)(C)C(C=C1)=CC=C1OC1=CC=C(N)C=C1 KMKWGXGSGPYISJ-UHFFFAOYSA-N 0.000 description 1
- LDFYRFKAYFZVNH-UHFFFAOYSA-N 4-[4-[4-(4-aminophenoxy)phenoxy]phenoxy]aniline Chemical compound C1=CC(N)=CC=C1OC(C=C1)=CC=C1OC(C=C1)=CC=C1OC1=CC=C(N)C=C1 LDFYRFKAYFZVNH-UHFFFAOYSA-N 0.000 description 1
- SXTPNMJRVQKNRN-UHFFFAOYSA-N 4-[4-[4-(4-aminophenoxy)phenyl]sulfanylphenoxy]aniline Chemical compound C1=CC(N)=CC=C1OC(C=C1)=CC=C1SC(C=C1)=CC=C1OC1=CC=C(N)C=C1 SXTPNMJRVQKNRN-UHFFFAOYSA-N 0.000 description 1
- UTDAGHZGKXPRQI-UHFFFAOYSA-N 4-[4-[4-(4-aminophenoxy)phenyl]sulfonylphenoxy]aniline Chemical compound C1=CC(N)=CC=C1OC1=CC=C(S(=O)(=O)C=2C=CC(OC=3C=CC(N)=CC=3)=CC=2)C=C1 UTDAGHZGKXPRQI-UHFFFAOYSA-N 0.000 description 1
- CNABHHDNHRETRU-UHFFFAOYSA-N 4-[4-[4-[4-[4-(4-aminophenoxy)phenoxy]phenyl]sulfonylphenoxy]phenoxy]aniline Chemical compound C1=CC(N)=CC=C1OC(C=C1)=CC=C1OC1=CC=C(S(=O)(=O)C=2C=CC(OC=3C=CC(OC=4C=CC(N)=CC=4)=CC=3)=CC=2)C=C1 CNABHHDNHRETRU-UHFFFAOYSA-N 0.000 description 1
- LACZRKUWKHQVKS-UHFFFAOYSA-N 4-[4-[4-amino-2-(trifluoromethyl)phenoxy]phenoxy]-3-(trifluoromethyl)aniline Chemical compound FC(F)(F)C1=CC(N)=CC=C1OC(C=C1)=CC=C1OC1=CC=C(N)C=C1C(F)(F)F LACZRKUWKHQVKS-UHFFFAOYSA-N 0.000 description 1
- RUGBKNDJNDMKGS-UHFFFAOYSA-N CC1=C(C(C2=C3C=CC=C2)(C(C=C2)=C(C)C=C2OC2=CC=C(C(O)=O)C(C(O)=O)=C2)OC3=O)C=CC(OC2=CC=C(C(O)=O)C(C(O)=O)=C2)=C1 Chemical compound CC1=C(C(C2=C3C=CC=C2)(C(C=C2)=C(C)C=C2OC2=CC=C(C(O)=O)C(C(O)=O)=C2)OC3=O)C=CC(OC2=CC=C(C(O)=O)C(C(O)=O)=C2)=C1 RUGBKNDJNDMKGS-UHFFFAOYSA-N 0.000 description 1
- ZPAKUZKMGJJMAA-UHFFFAOYSA-N Cyclohexane-1,2,4,5-tetracarboxylic acid Chemical compound OC(=O)C1CC(C(O)=O)C(C(O)=O)CC1C(O)=O ZPAKUZKMGJJMAA-UHFFFAOYSA-N 0.000 description 1
- JJHHIJFTHRNPIK-UHFFFAOYSA-N Diphenyl sulfoxide Chemical compound C=1C=CC=CC=1S(=O)C1=CC=CC=C1 JJHHIJFTHRNPIK-UHFFFAOYSA-N 0.000 description 1
- 239000005057 Hexamethylene diisocyanate Substances 0.000 description 1
- 239000005058 Isophorone diisocyanate Substances 0.000 description 1
- LJGFYZPEFLGTSS-UHFFFAOYSA-N NC(C=C1)=CC=C1C(OC(C=C1)=CC2=C1OC1=CC(OC(C(C=C3)=CC=C3N)=O)=CC=C1C21C(C=CC=C2)=C2C2=CC=CC=C12)=O Chemical compound NC(C=C1)=CC=C1C(OC(C=C1)=CC2=C1OC1=CC(OC(C(C=C3)=CC=C3N)=O)=CC=C1C21C(C=CC=C2)=C2C2=CC=CC=C12)=O LJGFYZPEFLGTSS-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- OMBNUQNGWAHQRP-UHFFFAOYSA-N O=C1OC(C2=C1C=CC=C2)(C2=C(C=C(C(=C2)C)OC=2C=C(C(=CC2)C(=O)O)C(=O)O)C)C2=C(C=C(C(=C2)C)OC=2C=C(C(=CC2)C(=O)O)C(=O)O)C Chemical compound O=C1OC(C2=C1C=CC=C2)(C2=C(C=C(C(=C2)C)OC=2C=C(C(=CC2)C(=O)O)C(=O)O)C)C2=C(C=C(C(=C2)C)OC=2C=C(C(=CC2)C(=O)O)C(=O)O)C OMBNUQNGWAHQRP-UHFFFAOYSA-N 0.000 description 1
- LAKTUTOJZZAKOD-UHFFFAOYSA-N OC(C(C(C(O)=O)=C1)=CC=C1C(OC(C=C1)=CC2=C1OC1=CC(OC(C3=CC=C(C(O)=O)C(C(O)=O)=C3)=O)=CC=C1C21C(C=CC=C2)=C2C2=CC=CC=C12)=O)=O Chemical compound OC(C(C(C(O)=O)=C1)=CC=C1C(OC(C=C1)=CC2=C1OC1=CC(OC(C3=CC=C(C(O)=O)C(C(O)=O)=C3)=O)=CC=C1C21C(C=CC=C2)=C2C2=CC=CC=C12)=O)=O LAKTUTOJZZAKOD-UHFFFAOYSA-N 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical class O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 1
- HJNTWZFZPOUDDG-UHFFFAOYSA-N [2-(3-aminophenoxy)phenyl]-phenylmethanone Chemical compound NC1=CC=CC(OC=2C(=CC=CC=2)C(=O)C=2C=CC=CC=2)=C1 HJNTWZFZPOUDDG-UHFFFAOYSA-N 0.000 description 1
- HVVWZTWDBSEWIH-UHFFFAOYSA-N [2-(hydroxymethyl)-3-prop-2-enoyloxy-2-(prop-2-enoyloxymethyl)propyl] prop-2-enoate Chemical compound C=CC(=O)OCC(CO)(COC(=O)C=C)COC(=O)C=C HVVWZTWDBSEWIH-UHFFFAOYSA-N 0.000 description 1
- VSZMHWHJMHITJR-UHFFFAOYSA-N [3-(3-amino-4-phenoxybenzoyl)phenyl]-(3-amino-4-phenoxyphenyl)methanone Chemical compound NC1=CC(C(=O)C=2C=C(C=CC=2)C(=O)C=2C=C(N)C(OC=3C=CC=CC=3)=CC=2)=CC=C1OC1=CC=CC=C1 VSZMHWHJMHITJR-UHFFFAOYSA-N 0.000 description 1
- JYYBEGXDDHNJMX-UHFFFAOYSA-N [3-(4-amino-3-phenoxybenzoyl)phenyl]-(4-amino-3-phenoxyphenyl)methanone Chemical compound NC1=CC=C(C(=O)C=2C=C(C=CC=2)C(=O)C=2C=C(OC=3C=CC=CC=3)C(N)=CC=2)C=C1OC1=CC=CC=C1 JYYBEGXDDHNJMX-UHFFFAOYSA-N 0.000 description 1
- WYYLAHMAYZBJOI-UHFFFAOYSA-N [3-[4-(3-aminophenoxy)benzoyl]phenyl]-[4-(3-aminophenoxy)phenyl]methanone Chemical compound NC1=CC=CC(OC=2C=CC(=CC=2)C(=O)C=2C=C(C=CC=2)C(=O)C=2C=CC(OC=3C=C(N)C=CC=3)=CC=2)=C1 WYYLAHMAYZBJOI-UHFFFAOYSA-N 0.000 description 1
- CTEMSXOAFRWUOU-UHFFFAOYSA-N [4-(3-amino-4-phenoxybenzoyl)phenyl]-(3-amino-4-phenoxyphenyl)methanone Chemical compound NC1=CC(C(=O)C=2C=CC(=CC=2)C(=O)C=2C=C(N)C(OC=3C=CC=CC=3)=CC=2)=CC=C1OC1=CC=CC=C1 CTEMSXOAFRWUOU-UHFFFAOYSA-N 0.000 description 1
- VQXJDOIQDHMFPQ-UHFFFAOYSA-N [4-(4-amino-3-phenoxybenzoyl)phenyl]-(4-amino-3-phenoxyphenyl)methanone Chemical compound NC1=CC=C(C(=O)C=2C=CC(=CC=2)C(=O)C=2C=C(OC=3C=CC=CC=3)C(N)=CC=2)C=C1OC1=CC=CC=C1 VQXJDOIQDHMFPQ-UHFFFAOYSA-N 0.000 description 1
- OXIKYYJDTWKERT-UHFFFAOYSA-N [4-(aminomethyl)cyclohexyl]methanamine Chemical compound NCC1CCC(CN)CC1 OXIKYYJDTWKERT-UHFFFAOYSA-N 0.000 description 1
- JZFJDKQWTNQXPN-UHFFFAOYSA-N [6'-(4-aminobenzoyl)oxyspiro[fluorene-9,9'-xanthene]-3'-yl] 4-aminobenzoate Chemical compound C1=CC=C2C(=C1)C3=CC=CC=C3C24C5=C(C=C(C=C5)OC(=O)C6=CC=C(C=C6)N)OC7=C4C=CC(=C7)OC(=O)C8=CC=C(C=C8)N JZFJDKQWTNQXPN-UHFFFAOYSA-N 0.000 description 1
- GTDPSWPPOUPBNX-UHFFFAOYSA-N ac1mqpva Chemical compound CC12C(=O)OC(=O)C1(C)C1(C)C2(C)C(=O)OC1=O GTDPSWPPOUPBNX-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- ZIXLDMFVRPABBX-UHFFFAOYSA-N alpha-methylcyclopentanone Natural products CC1CCCC1=O ZIXLDMFVRPABBX-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 230000003712 anti-aging effect Effects 0.000 description 1
- 230000003373 anti-fouling effect Effects 0.000 description 1
- 239000002519 antifouling agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 238000007611 bar coating method Methods 0.000 description 1
- HFACYLZERDEVSX-UHFFFAOYSA-N benzidine Chemical group C1=CC(N)=CC=C1C1=CC=C(N)C=C1 HFACYLZERDEVSX-UHFFFAOYSA-N 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- CJYIPJMCGHGFNN-UHFFFAOYSA-N bicyclo[2.2.1]heptane-2,3,5,6-tetracarboxylic acid Chemical compound C1C2C(C(O)=O)C(C(=O)O)C1C(C(O)=O)C2C(O)=O CJYIPJMCGHGFNN-UHFFFAOYSA-N 0.000 description 1
- KVMMOSKKPJEDNG-UHFFFAOYSA-N bicyclo[2.2.1]heptane-2,5-diamine Chemical compound C1C2C(N)CC1C(N)C2 KVMMOSKKPJEDNG-UHFFFAOYSA-N 0.000 description 1
- XQBSPQLKNWMPMG-UHFFFAOYSA-N bicyclo[2.2.2]octane-2,3,5,6-tetracarboxylic acid Chemical compound C1CC2C(C(O)=O)C(C(=O)O)C1C(C(O)=O)C2C(O)=O XQBSPQLKNWMPMG-UHFFFAOYSA-N 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 125000006267 biphenyl group Chemical group 0.000 description 1
- JMHUKWKCCHMXEL-UHFFFAOYSA-N bis(1,3-dioxo-2-benzofuran-5-yl) benzene-1,4-dicarboxylate Chemical compound C1=C2C(=O)OC(=O)C2=CC(OC(=O)C2=CC=C(C=C2)C(OC=2C=C3C(=O)OC(=O)C3=CC=2)=O)=C1 JMHUKWKCCHMXEL-UHFFFAOYSA-N 0.000 description 1
- SONDVQSYBUQGDH-UHFFFAOYSA-N bis(3-amino-4-phenoxyphenyl)methanone Chemical compound NC1=CC(C(=O)C=2C=C(N)C(OC=3C=CC=CC=3)=CC=2)=CC=C1OC1=CC=CC=C1 SONDVQSYBUQGDH-UHFFFAOYSA-N 0.000 description 1
- LRSFHOCOLGECMQ-UHFFFAOYSA-N bis(4-amino-3-phenoxyphenyl)methanone Chemical compound NC1=CC=C(C(=O)C=2C=C(OC=3C=CC=CC=3)C(N)=CC=2)C=C1OC1=CC=CC=C1 LRSFHOCOLGECMQ-UHFFFAOYSA-N 0.000 description 1
- ZLSMCQSGRWNEGX-UHFFFAOYSA-N bis(4-aminophenyl)methanone Chemical compound C1=CC(N)=CC=C1C(=O)C1=CC=C(N)C=C1 ZLSMCQSGRWNEGX-UHFFFAOYSA-N 0.000 description 1
- MQDJYUACMFCOFT-UHFFFAOYSA-N bis[2-(1-hydroxycyclohexyl)phenyl]methanone Chemical compound C=1C=CC=C(C(=O)C=2C(=CC=CC=2)C2(O)CCCCC2)C=1C1(O)CCCCC1 MQDJYUACMFCOFT-UHFFFAOYSA-N 0.000 description 1
- BBRLKRNNIMVXOD-UHFFFAOYSA-N bis[4-(3-aminophenoxy)phenyl]methanone Chemical compound NC1=CC=CC(OC=2C=CC(=CC=2)C(=O)C=2C=CC(OC=3C=C(N)C=CC=3)=CC=2)=C1 BBRLKRNNIMVXOD-UHFFFAOYSA-N 0.000 description 1
- WKDNYTOXBCRNPV-UHFFFAOYSA-N bpda Chemical compound C1=C2C(=O)OC(=O)C2=CC(C=2C=C3C(=O)OC(C3=CC=2)=O)=C1 WKDNYTOXBCRNPV-UHFFFAOYSA-N 0.000 description 1
- CDQSJQSWAWPGKG-UHFFFAOYSA-N butane-1,1-diol Chemical compound CCCC(O)O CDQSJQSWAWPGKG-UHFFFAOYSA-N 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 238000003851 corona treatment Methods 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- CURBACXRQKTCKZ-UHFFFAOYSA-N cyclobutane-1,2,3,4-tetracarboxylic acid Chemical compound OC(=O)C1C(C(O)=O)C(C(O)=O)C1C(O)=O CURBACXRQKTCKZ-UHFFFAOYSA-N 0.000 description 1
- QSAWQNUELGIYBC-UHFFFAOYSA-N cyclohexane-1,2-dicarboxylic acid Chemical compound OC(=O)C1CCCCC1C(O)=O QSAWQNUELGIYBC-UHFFFAOYSA-N 0.000 description 1
- XBZSBBLNHFMTEB-UHFFFAOYSA-N cyclohexane-1,3-dicarboxylic acid Chemical compound OC(=O)C1CCCC(C(O)=O)C1 XBZSBBLNHFMTEB-UHFFFAOYSA-N 0.000 description 1
- WOSVXXBNNCUXMT-UHFFFAOYSA-N cyclopentane-1,2,3,4-tetracarboxylic acid Chemical compound OC(=O)C1CC(C(O)=O)C(C(O)=O)C1C(O)=O WOSVXXBNNCUXMT-UHFFFAOYSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000002542 deteriorative effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000004031 devitrification Methods 0.000 description 1
- 125000004386 diacrylate group Chemical group 0.000 description 1
- 238000007607 die coating method Methods 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- UHESRSKEBRADOO-UHFFFAOYSA-N ethyl carbamate;prop-2-enoic acid Chemical compound OC(=O)C=C.CCOC(N)=O UHESRSKEBRADOO-UHFFFAOYSA-N 0.000 description 1
- 229920006228 ethylene acrylate copolymer Polymers 0.000 description 1
- 229920001038 ethylene copolymer Polymers 0.000 description 1
- 238000007756 gravure coating Methods 0.000 description 1
- 239000012760 heat stabilizer Substances 0.000 description 1
- RRAMGCGOFNQTLD-UHFFFAOYSA-N hexamethylene diisocyanate Chemical compound O=C=NCCCCCCN=C=O RRAMGCGOFNQTLD-UHFFFAOYSA-N 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 230000001678 irradiating effect Effects 0.000 description 1
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229940018564 m-phenylenediamine Drugs 0.000 description 1
- 235000019988 mead Nutrition 0.000 description 1
- OMGMPQSKRWSUHO-UHFFFAOYSA-N naphthalene-1,2,5-tricarboxylic acid Chemical compound OC(=O)C1=CC=CC2=C(C(O)=O)C(C(=O)O)=CC=C21 OMGMPQSKRWSUHO-UHFFFAOYSA-N 0.000 description 1
- KYTZHLUVELPASH-UHFFFAOYSA-N naphthalene-1,2-dicarboxylic acid Chemical compound C1=CC=CC2=C(C(O)=O)C(C(=O)O)=CC=C21 KYTZHLUVELPASH-UHFFFAOYSA-N 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 125000005740 oxycarbonyl group Chemical group [*:1]OC([*:2])=O 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 1
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 238000009832 plasma treatment Methods 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 239000009719 polyimide resin Substances 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 125000003003 spiro group Chemical group 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 238000000967 suction filtration Methods 0.000 description 1
- HXJUTPCZVOIRIF-UHFFFAOYSA-N sulfolane Chemical compound O=S1(=O)CCCC1 HXJUTPCZVOIRIF-UHFFFAOYSA-N 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 239000004634 thermosetting polymer Substances 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 229920006352 transparent thermoplastic Polymers 0.000 description 1
- 239000006097 ultraviolet radiation absorber Substances 0.000 description 1
- 230000002087 whitening effect Effects 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
- C08J7/042—Coating with two or more layers, where at least one layer of a composition contains a polymer binder
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/34—Layered products comprising a layer of synthetic resin comprising polyamides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2379/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen, or carbon only, not provided for in groups C08J2361/00 - C08J2377/00
- C08J2379/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08J2379/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
Abstract
취급시에 손상되어 시인성이 저하되지 않도록 내찰상성을 부여하기 위한 자기 수복층을 적층한, 높은 인장 탄성률과 낮은 CTE 및 내열성이나 내약품성을 갖는 폴리이미드 필름에 있어서, 가공 프로세스시 또는 굴곡시에 자기 수복층의 박리나 탈락을 일으키지 않는 적층체를 제공한다. MD 방향 및 TD 방향의 양쪽의 인장 탄성률이 3 GPa 이상, MD 방향 및 TD 방향의 양쪽의 CTE가 -5 ppm/℃∼+55 ppm/℃, 용매 함유량이 0.5∼5.0 질량%인 폴리이미드 필름과, 상기 폴리이미드 필름의 적어도 한쪽 면에 형성된 자기 수복층을 포함하는 것을 특징으로 하는 적층체. In a polyimide film having a high tensile modulus, low CTE, and heat resistance or chemical resistance, laminated with a self-healing layer for imparting scratch resistance so as not to deteriorate visibility due to damage during handling, self-repairing during processing or bending A laminate that does not cause peeling or falling off of a repair layer is provided. A polyimide film having a tensile modulus of elasticity of 3 GPa or more in both the MD and TD directions, a CTE of -5 ppm/°C to +55 ppm/°C in both the MD and TD directions, and a solvent content of 0.5 to 5.0% by mass; , A laminate comprising a self-healing layer formed on at least one side of the polyimide film.
Description
본 발명은, 자기 수복층과 폴리이미드 필름을 포함하는 적층체에 관한 것이다. 보다 상세하게는, 우수한 투명성, 자기 수복성, 유연성을 나타내고, 자기 수복층과의 밀착성이 우수한 폴리이미드 필름으로 이루어진 적층체에 관한 것이다. The present invention relates to a laminate comprising a self-healing layer and a polyimide film. More specifically, it relates to a laminate made of a polyimide film that exhibits excellent transparency, self-healing properties and flexibility, and has excellent adhesion to a self-healing layer.
종래, 터치패널이나 디스플레이 등의 화상 표시 장치의 전면판, 전극 주변에는, 취급시에 손상되어 시인성이 저하되지 않도록 내찰상성을 부여하는 것을 목적으로, 투명 기재 필름에 하드코트층을 형성한 하드코트 필름이 이용되고 있다(특허문헌 1). 한편, 최근, 높은 휴대성과 큰 화면을 모두 제공하기 위해서, 화상 표시 부분이 절첩 가능한 터치패널이나 디스플레이 등이 제안되고 있어(특허문헌 2), 하드코트 필름에 종래보다 우수한 유연성이 요구되게 되었다. Conventionally, around front plates and electrodes of image display devices such as touch panels and displays, a hard coat layer is formed on a transparent substrate film for the purpose of imparting scratch resistance so that visibility is not deteriorated due to damage during handling. A film is used (Patent Document 1). On the other hand, in recent years, in order to provide both high portability and a large screen, a touch panel or display in which an image display portion can be folded has been proposed (Patent Document 2), and flexibility superior to that of the conventional hard coat film has been required.
하드코트 필름에 형성되는 하드코트층은 내찰상성을 부여하기 위해 높은 표면 경도를 갖지만, 높은 표면 경도를 갖는 코트층은 취약해지는 경향이 있기 때문에, 우수한 유연성과 양립시키는 것은 어려웠다. 따라서 하드코트층을 대신하는 수단으로서, 유연하고 강인한 코트층을 형성하여, 취급시 또는 가공시 등의 응력에 의해 생긴 변형이나 손상을 자발적으로 소실하는 기능을 갖는 자기 치유성을 갖는 코트층을 형성한 필름이 제안되었다(특허문헌 3). The hard coat layer formed on the hard coat film has a high surface hardness to impart scratch resistance, but since a coat layer having a high surface hardness tends to be brittle, it has been difficult to achieve both excellent flexibility and flexibility. Therefore, as a means to replace the hard coat layer, a flexible and tough coat layer is formed to form a self-healing coat layer having a function of spontaneously disappearing deformation or damage caused by stress during handling or processing. One film has been proposed (Patent Document 3).
이러한 자기 수복성 필름에 이용되는 기재 필름으로는, 일반적으로 폴리에틸렌테레프탈레이트(PET), 아크릴, 폴리카보네이트(PC), 트리아세틸셀룰로오스(TAC), 폴리올레핀 등으로 이루어진 투명한 열가소성 수지 필름이 이용되고 있다. 이들 열가소성 수지 필름은 투명성이나 내굴곡성이 우수하고, 이(易)접착 처리나 가공이 용이하기 때문에 자기 수복 코트층의 밀착성도 우수하다(특허문헌 4). A transparent thermoplastic resin film made of polyethylene terephthalate (PET), acryl, polycarbonate (PC), triacetyl cellulose (TAC), polyolefin, or the like is generally used as a base film for such a self-healing film. These thermoplastic resin films are excellent in transparency and resistance to bending, and because they are easy to bond and process, they are also excellent in the adhesiveness of the self-healing coating layer (Patent Document 4).
한편, 기재 필름 상에 전극, 디스플레이 소자 등 기능 소자를 직접 형성하는 기술 개발이 행해지고, 하드코트 필름용의 기재 필름에 일반적인 열가소성 수지 필름 대신에, 내열성이나 내약품성을 갖는 폴리이미드 필름을 이용하는 것이 시도되고 있다(특허문헌 5∼6). On the other hand, technology development for directly forming functional elements such as electrodes and display elements on a base film has been carried out, and attempts have been made to use a polyimide film having heat resistance and chemical resistance instead of a general thermoplastic resin film for a base film for a hard coat film. It has become (Patent Documents 5 to 6).
전극, 디스플레이 소자 등 기능 소자를 직접 형성하기 위해 기재 필름에는 높은 인장 탄성률과 낮은 CTE 및 내열성이나 내약품성이 요구된다. 그와 같은 기재 필름에 적합한 폴리이미드 필름을 특허문헌 4에 예시되는 유연성이나 내굴곡성이 우수한 자기 수복성 필름의 기재 필름으로서 이용하고자 하는 경우, 폴리이미드 필름의 표면이 평탄하고 저활성이기 때문에, 폴리이미드 필름에 대한 밀착성이 불충분하고, 가공 프로세스시 또는 굴곡시에 코팅층의 박리나 탈락이 생기기 쉬워진다. In order to directly form functional elements such as electrodes and display elements, a high tensile modulus, low CTE, and heat resistance or chemical resistance are required for base films. When a polyimide film suitable for such a base film is to be used as a base film for a self-healing film excellent in flexibility and bending resistance as exemplified in Patent Document 4, since the surface of the polyimide film is flat and low in activity, the polyimide film is Adhesion to the mid film is insufficient, and peeling or falling off of the coating layer tends to occur during the processing process or during bending.
한편, 자기 수복층은 취급시 또는 가공시 등에 의한 외력을 흡수하여 변형이나 상처를 수복시키는 기능을 갖는 것 외에, 화상 표시 장치의 전면에 형성되기 때문에, 저태크성이나 방오성, 내약품성을 겸비할 필요가 있어, 높은 가교도와 조성적으로 저활성인 성분이 이용된다. 그 때문에 자기 수복층에 대해서는, 하드코트층용의 특허문헌 5에 예시되는 이접착층은 밀착성이 불충분하여, 가공 프로세스시 또는 굴곡시에 자기 수복층의 박리나 탈락이 생기기 쉬워진다.On the other hand, the self-healing layer has a function of absorbing an external force during handling or processing to repair deformation or damage, and since it is formed on the entire surface of the image display device, it can have low tack, stain resistance, and chemical resistance. If necessary, a component with a high degree of crosslinking and compositionally low activity is used. Therefore, regarding the self-healing layer, the easily bonding layer exemplified in Patent Document 5 for the hard coat layer has insufficient adhesiveness, and peeling or falling off of the self-healing layer occurs easily during the processing process or during bending.
이와 같이 폴리이미드 필름과 자기 수복층이 모두 저활성이기 때문에, 우수한 투명성, 자기 수복성, 유연성을 나타내면서, 자기 수복층과 폴리이미드 필름과의 밀착성을 얻는 것이 과제였다. Thus, since both the polyimide film and the self-healing layer have low activity, it has been a problem to obtain adhesion between the self-healing layer and the polyimide film while exhibiting excellent transparency, self-healing properties, and flexibility.
본 발명자들은, 상기 과제를 해결하기 위해 예의 검토한 결과, 이러한 과제를 해결할 수 있는 것을 발견하여 본 발명에 도달했다. 즉 본 발명은 이하의 구성을 갖는 것이다. The inventors of the present invention, as a result of intensive studies to solve the above problems, found that these problems could be solved and reached the present invention. That is, this invention has the following structures.
MD 방향 및 TD 방향의 양쪽의 인장 탄성률이 3 GPa 이상, MD 방향 및 TD 방향의 양쪽의 CTE가 -5 ppm/℃∼+55 ppm/℃, 용매 함유량이 0.5∼5.0 질량%인 폴리이미드 필름과, 상기 폴리이미드 필름의 적어도 한쪽 면에 형성된 자기 수복층을 포함하는 것을 특징으로 하는 적층체. A polyimide film having a tensile modulus of elasticity of 3 GPa or more in both the MD and TD directions, a CTE of -5 ppm/°C to +55 ppm/°C in both the MD and TD directions, and a solvent content of 0.5 to 5.0% by mass; , A laminate comprising a self-healing layer formed on at least one side of the polyimide film.
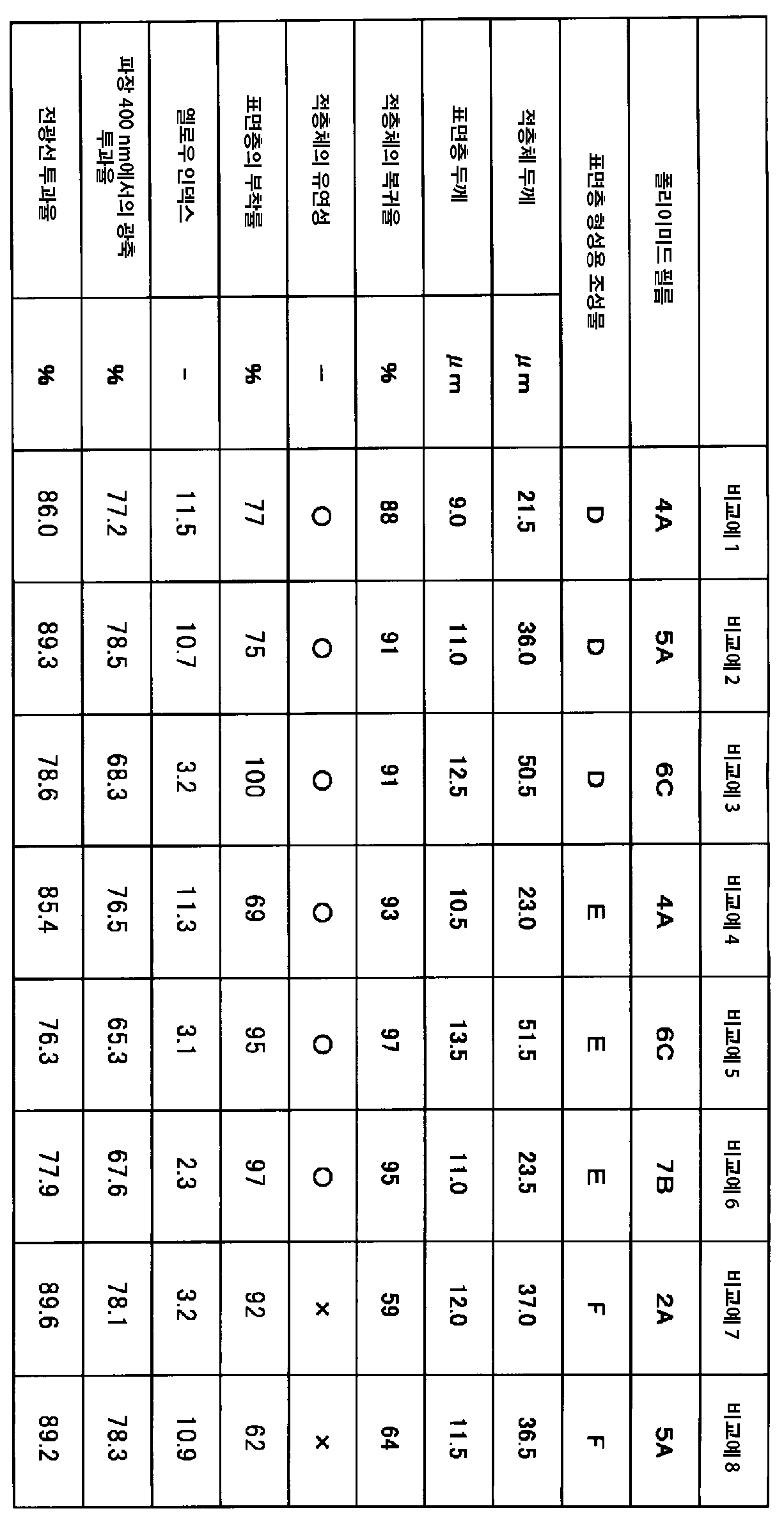
상기 적층체는, 미소 경도 시험기로 비커스 사각뿔 다이아몬드 압자로부터 미소한 하중 0.5 mN을 표면에 부여하여 5초간 유지한 후에, 0.005 mN까지 하중을 제거하여 60초간 유지한 후의 복귀율이 80% 이상인 것이 바람직하다. 또한, 상기 적층체는, JIS K 5600-5-6(1999)의 크로스커트법에 의한, 격자형으로 절단된 상기 자기 수복층의 상기 폴리이미드 필름에 대한 부착률이 80% 이상인 것이 바람직하다. 또한, 상기 적층체는, 옐로우 인덱스가 10 이하, 파장 400 nm에서의 광선 투과율이 70% 이상, 전광선 투과율이 85% 이상인 것이 바람직하다. 상기 자기 수복층은, 가교점을 포함하는 중합체 조성물이며, 상기 가교점의 일부 또는 전부가 가동성을 갖는 가교점인 것이 바람직하다. It is preferable that the laminate has a recovery rate of 80% or more after applying a minute load of 0.5 mN to the surface from a Vickers square pyramid diamond indenter with a micro hardness tester and holding it for 5 seconds, then removing the load to 0.005 mN and holding it for 60 seconds. do. Further, in the laminate, it is preferable that the adhesion rate of the self-healing layer cut in a lattice shape to the polyimide film by the crosscut method of JIS K 5600-5-6 (1999) is 80% or more. Further, the laminate preferably has a yellow index of 10 or less, a light transmittance of 70% or more at a wavelength of 400 nm, and a total light transmittance of 85% or more. It is preferable that the self-healing layer is a polymer composition containing crosslinking points, and some or all of the crosslinking points are crosslinking points having mobility.
본 발명에 의하면, 저활성의 표면을 갖는 폴리이미드 필름을 기재 필름으로서 이용하더라도, 우수한 자기 수복성을 나타내면서, 자기 수복층과의 밀착성이 우수한 적층체를 제공할 수 있다. 또한, 우수한 투명성 및 유연성을 갖기 때문에, 화상 표시 부분이 절첩 가능한 터치패널이나 디스플레이 등의 화상 표시 장치의 전면판, 전극 주변에 적합한 투명 적층체를 제공할 수 있다. According to the present invention, even when a polyimide film having a low activity surface is used as a base film, a laminate having excellent self-healing properties and excellent adhesion to a self-healing layer can be provided. Moreover, since it has excellent transparency and flexibility, it is possible to provide a transparent laminate suitable for front plates and around electrodes of image display devices such as touch panels and displays in which an image display portion can be folded.
이하, 본 발명의 실시형태의 적층체에 관해 설명한다. 본 발명의 적층체는 폴리이미드 필름과, 상기 폴리이미드 필름의 적어도 한쪽 면에 형성된 자기 수복층을 포함하는 적층체이다. 본 발명의 적층체를 구성하는 폴리이미드 필름은, 주쇄에 이미드 결합을 갖는 고분자의 필름이며, 바람직하게는 폴리이미드 필름 또는 폴리아미드이미드 필름이며, 보다 바람직하게는 폴리이미드 필름이다. Hereinafter, the laminate of the embodiment of the present invention will be described. The laminate of the present invention is a laminate comprising a polyimide film and a self-healing layer formed on at least one side of the polyimide film. The polyimide film constituting the laminate of the present invention is a polymer film having an imide bond in the main chain, preferably a polyimide film or a polyamideimide film, more preferably a polyimide film.
본 발명의 폴리이미드 필름은, 이하의 어느 제조 방법에 의해 얻어지는 것이 바람직하다. 우선, 첫번째 방법은, 용매 중에서 디아민류와 테트라카르복실산류를 중합 반응시켜 얻어지는 폴리아미드산(폴리이미드 전구체) 용액을, 폴리이미드 필름 제작용 지지체에 도포, 건조하여 그린 필름(「전구체 필름」, 「폴리아미드산 필름」 또는 「폴리아믹산 필름」이라고도 함)으로 하고, 또한 폴리이미드 필름 제작용 지지체 상에서, 혹은 상기 지지체로부터 박리한 상태에서, 그린 필름을 고온 열처리하여 탈수 폐환 중합반응을 행하게 함으로써 얻어진다. 또한, 두번째 방법으로서, 용매 중에서 디아민류와 테트라카르복실산류와의 탈수 폐환 중합 반응에 의해 얻어지는 폴리이미드 용액을 폴리이미드 필름 제작용 지지체에 도포, 건조하여 1∼50 질량%의 용매를 포함하는 폴리이미드 필름으로 하고, 또한 폴리이미드 필름 제작용 지지체 상에서, 혹은 상기 지지체로부터 박리한 상태에서, 1∼50 질량%의 용매를 포함하는 폴리이미드 필름을 고온 처리하여 건조시키는 것에 의해서도 얻어진다. It is preferable that the polyimide film of this invention is obtained by any of the following manufacturing methods. First, in the first method, a polyamic acid (polyimide precursor) solution obtained by polymerizing diamines and tetracarboxylic acids in a solvent is applied to a support for producing a polyimide film and dried to form a green film ("precursor film", Also referred to as "polyamic acid film" or "polyamic acid film"), and obtained by subjecting a green film to a high-temperature heat treatment on a support for producing a polyimide film or in a state peeled from the support to cause a dehydration ring closure polymerization reaction lose In addition, as a second method, a polyimide solution obtained by dehydration ring-closure polymerization of diamines and tetracarboxylic acids in a solvent is applied to a support for producing a polyimide film and dried to form a polyimide solution containing 1 to 50% by mass of a solvent. It is also obtained by setting it as a mid-film and further treating a polyimide film containing 1 to 50% by mass of a solvent at a high temperature on a support for polyimide film production or in a state peeled from the support and drying it.
또한, 세번째 방법은, 용매 중에서 디이소시아네이트류와 트리카르본류를 중합 반응시켜 얻어지는 폴리아미드이미드 용액을, 폴리아미드이미드 필름 제작용 지지체에 도포, 건조하여 1∼50 질량%의 용매를 포함하는 폴리아미드이미드 필름으로 하고, 또한 폴리아미드이미드 제작용 지지체 상에서, 혹은 상기 지지체로부터 박리한 상태에서, 1∼50 질량%의 용매를 포함하는 폴리아미드이미드 필름을 고온 처리하여 건조시킴으로써 얻어진다. 한편, 상기 3개의 제조 방법에 있어서, 디카르복실산류를 적절하게 사용할 수도 있다. Further, in the third method, a polyamideimide solution obtained by polymerizing diisocyanates and tricarbones in a solvent is applied to a support for producing a polyamideimide film and dried to obtain polyamide containing 1 to 50% by mass of a solvent. It is obtained by treating a polyamide-imide film containing 1 to 50% by mass of a solvent at a high temperature and drying it as a mid film, on a support for producing polyamide-imide, or in a state peeled from the support. On the other hand, in the above three production methods, dicarboxylic acids may be appropriately used.
상기 테트라카르복실산류, 트리카르복실산류 및 디카르복실산류로는, 폴리이미드 합성 또는 폴리아미드이미드 합성에 통상 이용되는 방향족 테트라카르복실산류(그 산무수물을 포함), 지방족 테트라카르복실산류(그 산무수물을 포함), 지환식 테트라카르복실산류(그 산무수물을 포함), 방향족 트리카르복실산류(그 산무수물을 포함), 지방족 트리카르복실산류(그 산무수물을 포함), 지환식 트리카르복실산류(그 산무수물을 포함), 방향족 디카르복실산류, 지방족 디카르복실산류 및 지환식 디카르복실산류 등을 이용할 수 있다. 그 중에서도, 방향족 테트라카르복실산무수물류 또는 지환식 테트라카르복실산무수물류가 바람직하고, 내열성의 관점에서는 방향족 테트라카르복실산무수물류가 보다 바람직하고, 광투과성(투명성)의 관점에서는 지환식 테트라카르복실산류가 보다 바람직하다. 테트라카르복실산류가 산무수물인 경우, 분자 내에 무수물 구조는 1개이어도 좋고 2개이어도 좋지만, 바람직하게는 2개의 무수물 구조를 갖는 것(2무수물)이 좋다. 테트라카르복실산류, 트리카르복실산류, 디카르복실산류는 단독으로 이용해도 좋고, 2종 이상을 병용해도 좋다. As the above tetracarboxylic acids, tricarboxylic acids and dicarboxylic acids, aromatic tetracarboxylic acids (including acid anhydrides thereof) commonly used in polyimide synthesis or polyamideimide synthesis, aliphatic tetracarboxylic acids (these acid anhydrides), alicyclic tetracarboxylic acids (including their acid anhydrides), aromatic tricarboxylic acids (including their acid anhydrides), aliphatic tricarboxylic acids (including their acid anhydrides), alicyclic tricarboxylic acids Boxylic acids (including acid anhydrides thereof), aromatic dicarboxylic acids, aliphatic dicarboxylic acids, alicyclic dicarboxylic acids, and the like can be used. Among them, aromatic tetracarboxylic acid anhydrides or alicyclic tetracarboxylic acid anhydrides are preferable, aromatic tetracarboxylic acid anhydrides are more preferable from the viewpoint of heat resistance, and alicyclic tetracarboxylic acid anhydrides are more preferable from the viewpoint of light transmission (transparency). Carboxylic acids are more preferred. When the tetracarboxylic acid is an acid anhydride, it may have one or two anhydride structures in the molecule, but preferably one having two anhydride structures (dianhydride) is good. Tetracarboxylic acids, tricarboxylic acids, and dicarboxylic acids may be used alone or in combination of two or more.
본 발명에서의 내열성이 높은 폴리이미드를 얻기 위한 방향족 테트라카르복실산류로는, 4,4'-(2,2-헥사플루오로이소프로필리덴)디프탈산, 4,4'-옥시디프탈산, 비스(1,3-디옥소-1,3-디히드로-2-벤조푸란-5-카르복실산)1,4-페닐렌, 비스(1,3-디옥소-1,3-디히드로-2-벤조푸란-5-일)벤젠-1,4-디카르복실레이트, 4,4'-[4,4'-(3-옥소-1,3-디히드로-2-벤조푸란-1,1-디일)비스(벤젠-1,4-디일옥시)]디벤젠-1,2-디카르복실산, 3,3',4,4'-벤조페논테트라카르복실산, 4,4'-[(3-옥소-1,3-디히드로-2-벤조푸란-1,1-디일)비스(톨루엔-2,5-디일옥시)]디벤젠-1,2-디카르복실산, 4,4'-[(3-옥소-1,3-디히드로-2-벤조푸란-1,1-디일)비스(1,4-크실렌-2,5-디일옥시)]디벤젠-1,2-디카르복실산, 4,4'-[4,4'-(3-옥소-1,3-디히드로-2-벤조푸란-1,1-디일)비스(4-이소프로필-톨루엔-2,5-디일옥시)]디벤젠-1,2-디카르복실산, 4,4'-[4,4'-(3-옥소-1,3-디히드로-2-벤조푸란-1,1-디일)비스(나프탈렌-1,4-디일옥시)]디벤젠-1,2-디카르복실산, 4,4'-[4,4'-(3H-2,1-벤즈옥사티올-1,1-디옥시드-3,3-디일)비스(벤젠-1,4-디일옥시)]디벤젠-1,2-디카르복실산, 4,4'-벤조페논테트라카르복실산, 4,4'-[(3H-2,1-벤즈옥사티올-1,1-디옥시드-3,3-디일)비스(톨루엔-2,5-디일옥시)]디벤젠-1,2-디카르복실산, 4,4'-[(3H-2,1-벤즈옥사티올-1,1-디옥시드-3,3-디일)비스(1,4-크실렌-2,5-디일옥시)]디벤젠-1,2-디카르복실산, 4,4'-[4,4'-(3H-2,1-벤즈옥사티올-1,1-디옥시드-3,3-디일)비스(4-이소프로필-톨루엔-2,5-디일옥시)]디벤젠-1,2-디카르복실산, 4,4'-[4,4'-(3H-2,1-벤즈옥사티올-1,1-디옥시드-3,3-디일)비스(나프탈렌-1,4-디일옥시)]디벤젠-1,2-디카르복실산, 3,3',4,4'-벤조페논테트라카르복실산, 3,3',4,4'-벤조페논테트라카르복실산, 3,3',4,4'-디페닐술폰테트라카르복실산, 3,3',4,4'-비페닐테트라카르복실산, 2,3,3',4'-비페닐테트라카르복실산, 피로멜리트산, 4,4'-[스피로(크산텐-9,9'-플루오렌)-2,6-디일비스(옥시카르보닐)]디벤젠-1,2-디카르복실산, 4,4'-[스피로(크산텐-9,9'-플루오렌)-3,6-디일비스(옥시카르보닐)]디벤젠-1,2-디카르복실산, 등의 테트라카르복실산 및 이들의 산무수물을 들 수 있다. 이들 중에서도, 2개의 산무수물 구조를 갖는 2무수물이 적합하며, 특히, 4,4'-(2,2-헥사플루오로이소프로필리덴)디프탈산2무수물, 4,4'-옥시디프탈산2무수물이 바람직하다. 한편, 방향족 테트라카르복실산류는 단독으로 이용해도 좋고, 2종 이상을 병용해도 좋다. 방향족 테트라카르복실산류는, 내열성을 중시하는 경우에는, 예컨대, 전체 테트라카르복실산류의 50 질량% 이상이 바람직하고, 보다 바람직하게는 60 질량% 이상, 더욱 바람직하게는 70 질량% 이상, 더욱 더 바람직하게는 80 질량% 이상이다. As aromatic tetracarboxylic acids for obtaining polyimide with high heat resistance in the present invention, 4,4'-(2,2-hexafluoroisopropylidene)diphthalic acid, 4,4'-oxydiphthalic acid, bis (1,3-dioxo-1,3-dihydro-2-benzofuran-5-carboxylic acid) 1,4-phenylene, bis(1,3-dioxo-1,3-dihydro-2 -benzofuran-5-yl)benzene-1,4-dicarboxylate, 4,4'-[4,4'-(3-oxo-1,3-dihydro-2-benzofuran-1,1 -Diyl)bis(benzene-1,4-diyloxy)]dibenzene-1,2-dicarboxylic acid, 3,3',4,4'-benzophenonetetracarboxylic acid, 4,4'- [(3-oxo-1,3-dihydro-2-benzofuran-1,1-diyl)bis(toluene-2,5-diyloxy)]dibenzene-1,2-dicarboxylic acid, 4 ,4'-[(3-oxo-1,3-dihydro-2-benzofuran-1,1-diyl)bis(1,4-xylene-2,5-diyloxy)]dibenzene-1, 2-dicarboxylic acid, 4,4'-[4,4'-(3-oxo-1,3-dihydro-2-benzofuran-1,1-diyl)bis(4-isopropyl-toluene- 2,5-diyloxy)]dibenzene-1,2-dicarboxylic acid, 4,4'-[4,4'-(3-oxo-1,3-dihydro-2-benzofuran-1 ,1-diyl)bis(naphthalene-1,4-diyloxy)]dibenzene-1,2-dicarboxylic acid, 4,4'-[4,4'-(3H-2,1-benzoxa Thiol-1,1-dioxide-3,3-diyl)bis(benzene-1,4-diyloxy)]dibenzene-1,2-dicarboxylic acid, 4,4'-benzophenonetetracarboxylic Acid, 4,4'-[(3H-2,1-benzoxathiol-1,1-dioxide-3,3-diyl)bis(toluene-2,5-diyloxy)]dibenzene-1, 2-dicarboxylic acid, 4,4'-[(3H-2,1-benzoxathiol-1,1-dioxide-3,3-diyl)bis(1,4-xylene-2,5-di yloxy)]dibenzene-1,2-dicarboxylic acid, 4,4'-[4,4'-(3H-2,1-benzoxathiol-1,1-dioxide-3,3-diyl )bis(4-isopropyl-toluene-2,5-diyloxy)]dibenzene-1,2-dicarboxylic acid, 4,4'-[4,4'-(3H-2,1-benz Oxathiol-1,1-dioxide-3,3-diyl)bis(naphthalene-1,4-diyloxy)]dibenzene-1,2-dicarboxylic acid, 3,3',4,4' -benzophenone tetracarboxylic acid, 3,3',4,4 '-benzophenonetetracarboxylic acid, 3,3',4,4'-diphenylsulfonetetracarboxylic acid, 3,3',4,4'-biphenyltetracarboxylic acid, 2,3,3' ,4'-biphenyltetracarboxylic acid, pyromellitic acid, 4,4'-[spiro(xanthene-9,9'-fluorene)-2,6-diylbis(oxycarbonyl)]dibenzene- 1,2-dicarboxylic acid, 4,4'-[spiro(xanthene-9,9'-fluorene)-3,6-diylbis(oxycarbonyl)]dibenzene-1,2-dicar Tetracarboxylic acids and acid anhydrides thereof, such as a boxylic acid, are mentioned. Among these, dianhydrides having two acid anhydride structures are suitable, particularly 4,4'-(2,2-hexafluoroisopropylidene)diphthalic dianhydride and 4,4'-oxydiphthalic dianhydride. this is preferable On the other hand, aromatic tetracarboxylic acids may be used independently or may use 2 or more types together. Aromatic tetracarboxylic acids are preferably 50% by mass or more, more preferably 60% by mass or more, still more preferably 70% by mass or more, and even more preferably 50% by mass or more of all tetracarboxylic acids, when importance is placed on heat resistance. Preferably it is 80 mass % or more.
무색 투명성이 높은 폴리이미드를 얻기 위한 지환식 테트라카르복실산류로는, 1,2,3,4-시클로부탄테트라카르복실산, 1,2,3,4-시클로펜탄테트라카르복실산, 3-(카르복시메틸)시클로펜탄-1,2,4-트리카르복실산, 1,2,3,4-시클로헥산테트라카르복실산, 1,2,4,5-시클로헥산테트라카르복실산, 3,3',4,4'-비시클로헥실테트라카르복실산, 비시클로[2,2,1]헵탄-2,3,5,6-테트라카르복실산, 비시클로[2,2,2]옥탄-2,3,5,6-테트라카르복실산, 비시클로[2,2,2]옥토-7-엔-2,3,5,6-테트라카르복실산, 테트라데카히드로안트라센-2,3,6,7-테트라카르복실산, 테트라데카히드로-1,4:5,8:9,10-트리메타노안트라센-2,3,6,7-테트라카르복실산, 데카히드로나프탈렌-2,3,6,7-테트라카르복실산, 데카히드로-1,4:5,8-디메타노나프탈렌-2,3,6,7-테트라카르복실산, 데카히드로-1,4-에타노-5,8-메타노나프탈렌-2,3,6,7-테트라카르복실산, 노보난-2-스피로-α-시클로펜타논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산(별칭 「노보난-2-스피로-2'-시클로펜타논-5'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산」), 메틸노보난-2-스피로-α-시클로펜타논-α'-스피로-2''-(메틸노보난)-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로헥사논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산(별칭 「노보난-2-스피로-2'-시클로헥사논-6'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산」), 메틸노보난-2-스피로-α-시클로헥사논-α'-스피로-2''-(메틸노보난)-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로프로파논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로부타논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로헵타논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로옥타논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로노나논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로데카논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로운데카논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로도데카논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로트리데카논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로테트라데카논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-시클로펜타데카논-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-(메틸시클로펜타논)-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 노보난-2-스피로-α-(메틸시클로헥사논)-α'-스피로-2''-노보난-5,5'',6,6''-테트라카르복실산, 등의 테트라카르복실산 및 이들의 산무수물을 들 수 있다. 이들 중에서도, 2개의 산무수물 구조를 갖는 2무수물이 적합하며, 특히, 1,2,3,4-시클로부탄테트라카르복실산2무수물, 1,2,3,4-시클로헥산테트라카르복실산2무수물, 1,2,4,5-시클로헥산테트라카르복실산2무수물이 바람직하고, 1,2,3,4-시클로부탄테트라카르복실산2무수물, 1,2,4,5-시클로헥산테트라카르복실산2무수물이 보다 바람직하고, 1,2,3,4-시클로부탄테트라카르복실산2무수물이 더욱 바람직하다. 또, 이들은 단독으로 이용해도 좋고, 2종 이상을 병용해도 좋다. 지환식 테트라카르복실산류는, 투명성을 중시하는 경우에는, 예컨대, 전체 테트라카르복실산류의 50 질량% 이상이 바람직하고, 보다 바람직하게는 60 질량% 이상, 더욱 바람직하게는 70 질량% 이상, 더욱 더 바람직하게는 80 질량% 이상이다. As alicyclic tetracarboxylic acids for obtaining colorless and highly transparent polyimide, 1,2,3,4-cyclobutane tetracarboxylic acid, 1,2,3,4-cyclopentane tetracarboxylic acid, 3- (Carboxymethyl)cyclopentane-1,2,4-tricarboxylic acid, 1,2,3,4-cyclohexanetetracarboxylic acid, 1,2,4,5-cyclohexanetetracarboxylic acid, 3, 3',4,4'-bicyclohexyltetracarboxylic acid, bicyclo[2,2,1]heptane-2,3,5,6-tetracarboxylic acid, bicyclo[2,2,2]octane -2,3,5,6-tetracarboxylic acid, bicyclo[2,2,2]octo-7-ene-2,3,5,6-tetracarboxylic acid, tetradecahydroanthracene-2,3 ,6,7-tetracarboxylic acid, tetradecahydro-1,4:5,8:9,10-trimethanoanthracene-2,3,6,7-tetracarboxylic acid, decahydronaphthalene-2, 3,6,7-tetracarboxylic acid, decahydro-1,4:5,8-dimethanonaphthalene-2,3,6,7-tetracarboxylic acid, decahydro-1,4-ethano- 5,8-Methanonaphthalene-2,3,6,7-tetracarboxylic acid, norbonane-2-spiro-α-cyclopentanone-α'-spiro-2''-norbonane-5,5' ',6,6''-tetracarboxylic acid (aka "norbonane-2-spiro-2'-cyclopentanone-5'-spiro-2''-norbonane-5,5'',6,6 ''-tetracarboxylic acid'), methylnorbonane-2-spiro-α-cyclopentanone-α'-spiro-2''-(methylnorbornane)-5,5'',6,6'' -Tetracarboxylic acid, norbonane-2-spiro-α-cyclohexanone-α'-spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid (aka " Norbonane-2-spiro-2'-cyclohexanone-6'-spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid"), methylnorbornane-2 -Spiro-α-cyclohexanone-α'-spiro-2''-(methylnorbornane)-5,5'',6,6''-tetracarboxylic acid, norbonane-2-spiro-α- Cyclopropanone-α'-spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid, norbonane-2-spiro-α-cyclobutanone-α'-spiro -2''-norbonane-5,5'',6,6''-tetracarboxylic acid, norbonane-2-spiro-α-cycloheptanone-α'-spiro-2''-norbonane- 5,5'',6,6 ''-tetracarboxylic acid, norbonane-2-spiro-α-cyclooctanone-α'-spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid, Norbonane-2-spiro-α-cyclononanone-α'-spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid, norbonane-2-spiro-α -Cyclodecanone-α'-spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid, norbonane-2-spiro-α-cycloundecanone-α'- Spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid, norbonane-2-spiro-α-cyclododecanone-α'-spiro-2''-norbonane -5,5'',6,6''-tetracarboxylic acid, norbonane-2-spiro-α-cyclotridecanone-α'-spiro-2''-norbonane-5,5'', 6,6''-tetracarboxylic acid, norbonane-2-spiro-α-cyclotetradecanone-α'-spiro-2''-norbonane-5,5'',6,6''-tetra Carboxylic acid, norbonane-2-spiro-α-cyclopentadecanone-α'-spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid, norbonane- 2-spiro-α-(methylcyclopentanone)-α'-spiro-2''-norbonane-5,5'',6,6''-tetracarboxylic acid, norbonane-2-spiro-α Tetracarboxylic acids such as -(methylcyclohexanone)-α'-spiro-2''-norbornane-5,5'',6,6''-tetracarboxylic acid, and acid anhydrides thereof. can Among these, dianhydrides having two acid anhydride structures are suitable, particularly 1,2,3,4-cyclobutanetetracarboxylic dianhydride and 1,2,3,4-cyclohexanetetracarboxylic acid 2 Anhydride, 1,2,4,5-cyclohexanetetracarboxylic dianhydride is preferred, 1,2,3,4-cyclobutanetetracarboxylic dianhydride, 1,2,4,5-cyclohexanetetra Carboxylic dianhydride is more preferred, and 1,2,3,4-cyclobutanetetracarboxylic dianhydride is still more preferred. Moreover, these may be used independently and may use 2 or more types together. Alicyclic tetracarboxylic acids are preferably 50% by mass or more, more preferably 60% by mass or more, still more preferably 70% by mass or more, of all tetracarboxylic acids, for example, when importance is placed on transparency. More preferably, it is 80 mass % or more.
트리카르복실산류로는, 트리멜리트산, 1,2,5-나프탈렌트리카르복실산, 디페닐에테르-3,3',4'-트리카르복실산, 디페닐술폰-3,3',4'-트리카르복실산 등의 방향족 트리카르복실산, 혹은 헥사히드로트리멜리트산 등의 상기 방향족 트리카르복실산의 수소 첨가물, 에틸렌글리콜비스트리멜리테이트, 프로필렌글리콜비스트리멜리테이트, 1,4-부탄디올비스트리멜리테이트, 폴리에틸렌글리콜비스트리멜리테이트 등의 알킬렌글리콜비스트리멜리테이트, 및 이들의 1무수물, 에스테르화물을 들 수 있다. 이들 중에서도, 1개의 산무수물 구조를 갖는 1무수물이 적합하며, 특히, 트리멜리트산무수물, 헥사히드로트리멜리트산무수물이 바람직하다. 또한, 이들은 단독으로 사용해도 좋고 복수를 조합하여 사용해도 좋다. Examples of tricarboxylic acids include trimellitic acid, 1,2,5-naphthalene tricarboxylic acid, diphenyl ether-3,3',4'-tricarboxylic acid, and diphenylsulfone-3,3',4 Aromatic tricarboxylic acids such as '-tricarboxylic acid, or hydrogenated products of the aromatic tricarboxylic acids such as hexahydrotrimellitic acid, ethylene glycol bistrimellitate, propylene glycol bistrimellitate, 1,4- alkylene glycol bistrimellitate such as butanediol bistrimellitate and polyethylene glycol bistrimellitate; and monohydrides and esterified products thereof. Among these, a monohydride having a single acid anhydride structure is preferable, and trimellitic anhydride and hexahydrotrimellitic anhydride are particularly preferable. In addition, these may be used individually or may be used combining plurality.
디카르복실산류로는, 테레프탈산, 이소프탈산, 오르토프탈산, 나프탈렌디카르복실산, 비페닐-4,4'-디카르복실산, 4,4'-옥시디벤젠디카르복실산 등의 방향족 디카르복실산, 혹은 1,4-시클로헥산디카르복실산, 1,3-시클로헥산디카르복실산, 1,2-시클로헥산디카르복실산 등의 상기 방향족 디카르복실산의 수소 첨가물, 옥살산, 숙신산, 글루탈산, 아디프산, 헵탄2산, 옥탄2산, 아젤라산, 세바스산, 운데카2산, 도데칸2산, 2-메틸숙신산 및 이것들의 산염화물 혹은 에스테르화물 등을 들 수 있다. 이들 중에서 방향족 디카르복실산 및 그 수소 첨가물이 적합하며, 특히, 테레프탈산, 1,4-시클로헥산디카르복실산, 4,4'-옥시디벤젠디카르복실산이 바람직하다. 또한, 디카르복실산류는 단독으로 사용해도 좋고 복수를 조합하여 사용해도 좋다. Examples of the dicarboxylic acids include aromatic dicarboxylic acids such as terephthalic acid, isophthalic acid, orthophthalic acid, naphthalene dicarboxylic acid, biphenyl-4,4'-dicarboxylic acid, and 4,4'-oxydibenzenedicarboxylic acid. Hydrogenated products of aromatic dicarboxylic acids such as carboxylic acid, 1,4-cyclohexanedicarboxylic acid, 1,3-cyclohexanedicarboxylic acid, 1,2-cyclohexanedicarboxylic acid, or oxalic acid , succinic acid, glutalic acid, adipic acid, heptanedioic acid, octanedioic acid, azelaic acid, sebacic acid, undecanedioic acid, dodecanedioic acid, 2-methylsuccinic acid, and acid chlorides or esters thereof. . Among these, aromatic dicarboxylic acids and hydrogenated products thereof are suitable, and terephthalic acid, 1,4-cyclohexanedicarboxylic acid, and 4,4'-oxydibenzenedicarboxylic acid are particularly preferred. In addition, dicarboxylic acids may be used independently or may be used in combination of plurality.
본 발명에서의 내열성 및/또는 무색 투명성이 높은 폴리이미드를 얻기 위한 디아민류 혹은 이소시아네이트류로는, 특별히 제한은 없고, 폴리이미드 합성 또는 폴리아미드이미드 합성에 통상 이용되는 방향족 디아민류, 지방족 디아민류, 지환식 디아민류, 방향족 디이소시아네이트류, 지방족 디이소시아네이트류, 지환식 디이소시아네이트류 등을 이용할 수 있다. 내열성의 관점에서는, 방향족 디아민류 또는 방향족 디이소시아네이트류가 바람직하고, 투명성의 관점에서는 지환식 디아민류 또는 지환식 디이소시아네이트류가 바람직하다. 또한, 벤조옥사졸 구조를 갖는 방향족 디아민류 또는 디이소시아네이트류를 이용하면, 높은 내열성과 함께, 고탄성률, 저열수축성, 저선팽창계수를 발현시키는 것이 가능해지기 때문에 바람직하다. 상기 디아민류 및 이소시아네이트류는, 단독으로 이용해도 좋고 2종 이상을 병용해도 좋다. Diamines or isocyanates for obtaining polyimide with high heat resistance and/or colorless transparency in the present invention are not particularly limited, and aromatic diamines and aliphatic diamines commonly used in polyimide synthesis or polyamideimide synthesis, Alicyclic diamines, aromatic diisocyanates, aliphatic diisocyanates, alicyclic diisocyanates and the like can be used. From the viewpoint of heat resistance, aromatic diamines or aromatic diisocyanates are preferred, and from the viewpoint of transparency, alicyclic diamines or alicyclic diisocyanates are preferred. In addition, when aromatic diamines or diisocyanates having a benzoxazole structure are used, they are preferable because they can exhibit a high modulus of elasticity, low heat shrinkage, and low coefficient of linear expansion along with high heat resistance. The said diamines and isocyanates may be used independently or may use 2 or more types together.
방향족 디아민류로는, 예컨대, 2,2'-디메틸-4,4'-디아미노비페닐, 1,4-비스[2-(4-아미노페닐)-2-프로필]벤젠, 1,4-비스(4-아미노-2-트리플루오로메틸페녹시)벤젠, 2,2'-디트리플루오로메틸-4,4'-디아미노비페닐, 4,4'-비스(4-아미노페녹시)비페닐, 4,4'-비스(3-아미노페녹시)비페닐, 비스[4-(3-아미노페녹시)페닐]케톤, 비스[4-(3-아미노페녹시)페닐]술피드, 비스[4-(3-아미노페녹시)페닐]술폰, 2,2-비스[4-(3-아미노페녹시)페닐]프로판, 2,2-비스[4-(3-아미노페녹시)페닐]-1,1,1,3,3,3-헥사플루오로프로판, m-페닐렌디아민, o-페닐렌디아민, p-페닐렌디아민, m-아미노벤질아민, p-아미노벤질아민, 4-아미노-N-(4-아미노페닐)벤즈아미드, 3,3'-디아미노디페닐에테르, 3,4'-디아미노디페닐에테르, 4,4'-디아미노디페닐에테르, 2,2'-트리플루오로메틸-4,4'-디아미노디페닐에테르, 3,3'-디아미노디페닐술피드, 3,3'-디아미노디페닐술폭시드, 3,4'-디아미노디페닐술폭시드, 4,4'-디아미노디페닐술폭시드, 3,3'-디아미노디페닐술폰, 3,4'-디아미노디페닐술폰, 4,4'-디아미노디페닐술폰, 3,3'-디아미노벤조페논, 3,4'-디아미노벤조페논, 4,4'-디아미노벤조페논, 3,3'-디아미노디페닐메탄, 3,4'-디아미노디페닐메탄, 4,4'-디아미노디페닐메탄, 비스[4-(4-아미노페녹시)페닐]메탄, 1,1-비스[4-(4-아미노페녹시)페닐]에탄, 1,2-비스[4-(4-아미노페녹시)페닐]에탄, 1,1-비스[4-(4-아미노페녹시)페닐]프로판, 1,2-비스[4-(4-아미노페녹시)페닐]프로판, 1,3-비스[4-(4-아미노페녹시)페닐]프로판, 2,2-비스[4-(4-아미노페녹시)페닐]프로판, 1,1-비스[4-(4-아미노페녹시)페닐]부탄, 1,3-비스[4-(4-아미노페녹시)페닐]부탄, 1,4-비스[4-(4-아미노페녹시)페닐]부탄, 2,2-비스[4-(4-아미노페녹시)페닐]부탄, 2,3-비스[4-(4-아미노페녹시)페닐]부탄, 2-[4-(4-아미노페녹시)페닐]-2-[4-(4-아미노페녹시)-3-메틸페닐]프로판, 2,2-비스[4-(4-아미노페녹시)-3-메틸페닐]프로판, 2-[4-(4-아미노페녹시)페닐]-2-[4-(4-아미노페녹시)-3,5-디메틸페닐]프로판, 2,2-비스[4-(4-아미노페녹시)-3,5-디메틸페닐]프로판, 2,2-비스[4-(4-아미노페녹시)페닐]-1,1,1,3,3,3-헥사플루오로프로판, 1,4-비스(3-아미노페녹시)벤젠, 1,3-비스(3-아미노페녹시)벤젠, 1,4-비스(4-아미노페녹시)벤젠, 4,4'-비스(4-아미노페녹시)비페닐, 비스[4-(4-아미노페녹시)페닐]케톤, 비스[4-(4-아미노페녹시)페닐]술피드, 비스[4-(4-아미노페녹시)페닐]술폭시드, 비스[4-(4-아미노페녹시)페닐]술폰, 비스[4-(3-아미노페녹시)페닐]에테르, 비스[4-(4-아미노페녹시)페닐]에테르, 1,3-비스[4-(4-아미노페녹시)벤조일]벤젠, 1,3-비스[4-(3-아미노페녹시)벤조일]벤젠, 1,4-비스[4-(3-아미노페녹시)벤조일]벤젠, 4,4'-비스[(3-아미노페녹시)벤조일]벤젠, 1,1-비스[4-(3-아미노페녹시)페닐]프로판, 1,3-비스[4-(3-아미노페녹시)페닐]프로판, 3,4'-디아미노디페닐술피드, 2,2-비스[3-(3-아미노페녹시)페닐]-1,1,1,3,3,3-헥사플루오로프로판, 비스[4-(3-아미노페녹시)페닐]메탄, 1,1-비스[4-(3-아미노페녹시)페닐]에탄, 1,2-비스[4-(3-아미노페녹시)페닐]에탄, 비스[4-(3-아미노페녹시)페닐]술폭시드, 4,4'-비스[3-(4-아미노페녹시)벤조일]디페닐에테르, 4,4'-비스[3-(3-아미노페녹시)벤조일]디페닐에테르, 4,4'-비스[4-(4-아미노-α,α-디메틸벤질)페녹시]벤조페논, 4,4'-비스[4-(4-아미노-α,α-디메틸벤질)페녹시]디페닐술폰, 비스[4-{4-(4-아미노페녹시)페녹시}페닐]술폰, 1,4-비스[4-(4-아미노페녹시)페녹시-α,α-디메틸벤질]벤젠, 1,3-비스[4-(4-아미노페녹시)페녹시-α,α-디메틸벤질]벤젠, 1,3-비스[4-(4-아미노-6-트리플루오로메틸페녹시)-α,α-디메틸벤질]벤젠, 1,3-비스[4-(4-아미노-6-플루오로페녹시)-α,α-디메틸벤질]벤젠, 1,3-비스[4-(4-아미노-6-메틸페녹시)-α,α-디메틸벤질]벤젠, 1,3-비스[4-(4-아미노-6-시아노페녹시)-α,α-디메틸벤질]벤젠, 3,3'-디아미노-4,4'-디페녹시벤조페논, 4,4'-디아미노-5,5'-디페녹시벤조페논, 3,4'-디아미노-4,5'-디페녹시벤조페논, 3,3'-디아미노-4-페녹시벤조페논, 4,4'-디아미노-5-페녹시벤조페논, 3,4'-디아미노-4-페녹시벤조페논, 3,4'-디아미노-5'-페녹시벤조페논, 3,3'-디아미노-4,4'-디비페녹시벤조페논, 4,4'-디아미노-5,5'-디비페녹시벤조페논, 3,4'-디아미노-4,5'-디비페녹시벤조페논, 3,3'-디아미노-4-비페녹시벤조페논, 4,4'-디아미노-5-비페녹시벤조페논, 3,4'-디아미노-4-비페녹시벤조페논, 3,4'-디아미노-5'-비페녹시벤조페논, 1,3-비스(3-아미노-4-페녹시벤조일)벤젠, 1,4-비스(3-아미노-4-페녹시벤조일)벤젠, 1,3-비스(4-아미노-5-페녹시벤조일)벤젠, 1,4-비스(4-아미노-5-페녹시벤조일)벤젠, 1,3-비스(3-아미노-4-비페녹시벤조일)벤젠, 1,4-비스(3-아미노-4-비페녹시벤조일)벤젠, 1,3-비스(4-아미노-5-비페녹시벤조일)벤젠, 1,4-비스(4-아미노-5-비페녹시벤조일)벤젠, 2,6-비스[4-(4-아미노α,α-디메틸벤질)페녹시]벤조니트릴, 4,4'-[스피로(크산텐-9,9'-플루오렌)-2,6-디일비스(옥시카르보닐)]비스아닐린, 4,4'-[스피로(크산텐-9,9'-플루오렌)-3,6-디일비스(옥시카르보닐)]비스아닐린 등을 들 수 있다. 또한, 상기 방향족 디아민의 방향환 상의 수소 원자의 일부 혹은 전부가, 할로겐 원자, 탄소수 1∼3의 알킬기 혹은 알콕실기, 또는 시아노기로 치환되어 있어도 좋고, 또한 상기 탄소수 1∼3의 알킬기 혹은 알콕실기의 수소 원자의 일부 혹은 전부가 할로겐 원자로 치환되어 있어도 좋다. 또한, 방향족 디아민류는 단독으로 사용해도 좋고 복수를 조합하여 사용해도 좋다. Examples of aromatic diamines include 2,2'-dimethyl-4,4'-diaminobiphenyl, 1,4-bis[2-(4-aminophenyl)-2-propyl]benzene, and 1,4- Bis (4-amino-2-trifluoromethylphenoxy) benzene, 2,2'-ditrifluoromethyl-4,4'-diaminobiphenyl, 4,4'-bis (4-aminophenoxy ) Biphenyl, 4,4'-bis (3-aminophenoxy) biphenyl, bis [4- (3-aminophenoxy) phenyl] ketone, bis [4- (3-aminophenoxy) phenyl] sulfide , bis[4-(3-aminophenoxy)phenyl]sulfone, 2,2-bis[4-(3-aminophenoxy)phenyl]propane, 2,2-bis[4-(3-aminophenoxy) Phenyl] -1,1,1,3,3,3-hexafluoropropane, m-phenylenediamine, o-phenylenediamine, p-phenylenediamine, m-aminobenzylamine, p-aminobenzylamine, 4-amino-N-(4-aminophenyl)benzamide, 3,3'-diaminodiphenyl ether, 3,4'-diaminodiphenyl ether, 4,4'-diaminodiphenyl ether, 2, 2'-trifluoromethyl-4,4'-diaminodiphenyl ether, 3,3'-diaminodiphenyl sulfide, 3,3'-diaminodiphenylsulfoxide, 3,4'-diamino Diphenylsulfoxide, 4,4'-diaminodiphenylsulfoxide, 3,3'-diaminodiphenylsulfone, 3,4'-diaminodiphenylsulfone, 4,4'-diaminodiphenylsulfone, 3,3'-diaminobenzophenone, 3,4'-diaminobenzophenone, 4,4'-diaminobenzophenone, 3,3'-diaminodiphenylmethane, 3,4'-diaminodiphenyl Methane, 4,4'-diaminodiphenylmethane, bis[4-(4-aminophenoxy)phenyl]methane, 1,1-bis[4-(4-aminophenoxy)phenyl]ethane, 1,2 -bis[4-(4-aminophenoxy)phenyl]ethane, 1,1-bis[4-(4-aminophenoxy)phenyl]propane, 1,2-bis[4-(4-aminophenoxy) Phenyl]propane, 1,3-bis[4-(4-aminophenoxy)phenyl]propane, 2,2-bis[4-(4-aminophenoxy)phenyl]propane, 1,1-bis[4- (4-aminophenoxy)phenyl]butane, 1,3-bis[4-(4-aminophenoxy)phenyl]butane, 1,4-bis[4-(4-aminophenoxy)phenyl]butane, 2 ,2-bis[4-(4-aminophenoxy)phenyl]butane, 2,3-bis[4-(4-aminophenoxy)phenyl]butane, 2-[4- (4-aminophenoxy)phenyl]-2-[4-(4-aminophenoxy)-3-methylphenyl]propane, 2,2-bis[4-(4-aminophenoxy)-3-methylphenyl]propane , 2-[4-(4-aminophenoxy)phenyl]-2-[4-(4-aminophenoxy)-3,5-dimethylphenyl]propane, 2,2-bis[4-(4-amino) Phenoxy) -3,5-dimethylphenyl] propane, 2,2-bis [4- (4-aminophenoxy) phenyl] -1,1,1,3,3,3-hexafluoropropane, 1, 4-bis(3-aminophenoxy)benzene, 1,3-bis(3-aminophenoxy)benzene, 1,4-bis(4-aminophenoxy)benzene, 4,4'-bis(4-amino Phenoxy)biphenyl, bis[4-(4-aminophenoxy)phenyl]ketone, bis[4-(4-aminophenoxy)phenyl]sulfide, bis[4-(4-aminophenoxy)phenyl] Sulfoxide, bis[4-(4-aminophenoxy)phenyl]sulfone, bis[4-(3-aminophenoxy)phenyl]ether, bis[4-(4-aminophenoxy)phenyl]ether, 1, 3-bis[4-(4-aminophenoxy)benzoyl]benzene, 1,3-bis[4-(3-aminophenoxy)benzoyl]benzene, 1,4-bis[4-(3-aminophenoxy) ) Benzoyl] benzene, 4,4'-bis [(3-aminophenoxy) benzoyl] benzene, 1,1-bis [4- (3-aminophenoxy) phenyl] propane, 1,3-bis [4- (3-aminophenoxy)phenyl]propane, 3,4'-diaminodiphenylsulfide, 2,2-bis[3-(3-aminophenoxy)phenyl]-1,1,1,3,3 ,3-hexafluoropropane, bis[4-(3-aminophenoxy)phenyl]methane, 1,1-bis[4-(3-aminophenoxy)phenyl]ethane, 1,2-bis[4- (3-aminophenoxy)phenyl]ethane, bis[4-(3-aminophenoxy)phenyl]sulfoxide, 4,4'-bis[3-(4-aminophenoxy)benzoyl]diphenyl ether, 4 ,4'-bis[3-(3-aminophenoxy)benzoyl]diphenyl ether, 4,4'-bis[4-(4-amino-α,α-dimethylbenzyl)phenoxy]benzophenone, 4, 4′-bis[4-(4-amino-α,α-dimethylbenzyl)phenoxy]diphenylsulfone, bis[4-{4-(4-aminophenoxy)phenoxy}phenyl]sulfone, 1,4 -bis[4-(4-aminophenoxy)phenoxy-α,α-dimethylbenzyl]benzene, 1,3-bis[4-(4-aminophenoxy)phenoxy-α,α-dimethylbenzyl]benzene , 1,3-bis[4-(4- Amino-6-trifluoromethylphenoxy)-α,α-dimethylbenzyl]benzene, 1,3-bis[4-(4-amino-6-fluorophenoxy)-α,α-dimethylbenzyl]benzene , 1,3-bis[4-(4-amino-6-methylphenoxy)-α,α-dimethylbenzyl]benzene, 1,3-bis[4-(4-amino-6-cyanophenoxy) -α,α-dimethylbenzyl]benzene, 3,3'-diamino-4,4'-diphenoxybenzophenone, 4,4'-diamino-5,5'-diphenoxybenzophenone, 3, 4'-diamino-4,5'-diphenoxybenzophenone, 3,3'-diamino-4-phenoxybenzophenone, 4,4'-diamino-5-phenoxybenzophenone, 3,4 '-diamino-4-phenoxybenzophenone, 3,4'-diamino-5'-phenoxybenzophenone, 3,3'-diamino-4,4'-dibiphenoxybenzophenone, 4,4 '-diamino-5,5'-dibiphenoxybenzophenone, 3,4'-diamino-4,5'-dibiphenoxybenzophenone, 3,3'-diamino-4-biphenoxybenzophenone , 4,4'-diamino-5-biphenoxybenzophenone, 3,4'-diamino-4-biphenoxybenzophenone, 3,4'-diamino-5'-biphenoxybenzophenone, 1,3-bis(3-amino-4-phenoxybenzoyl)benzene, 1,4-bis(3-amino-4-phenoxybenzoyl)benzene, 1,3-bis(4-amino-5-phenoxy Benzoyl) benzene, 1,4-bis (4-amino-5-phenoxybenzoyl) benzene, 1,3-bis (3-amino-4-biphenoxybenzoyl) benzene, 1,4-bis (3-amino -4-biphenoxybenzoyl) benzene, 1,3-bis (4-amino-5-biphenoxybenzoyl) benzene, 1,4-bis (4-amino-5-biphenoxybenzoyl) benzene, 2, 6-bis[4-(4-aminoα,α-dimethylbenzyl)phenoxy]benzonitrile, 4,4′-[spiro(xanthene-9,9′-fluorene)-2,6-diylbis( oxycarbonyl)] bisaniline, 4,4'-[spiro(xanthen-9,9'-fluorene)-3,6-diylbis(oxycarbonyl)]bisaniline, and the like. Further, some or all of the hydrogen atoms on the aromatic ring of the aromatic diamine may be substituted with a halogen atom, an alkyl group or alkoxyl group having 1 to 3 carbon atoms, or a cyano group, or an alkyl group or alkoxyl group having 1 to 3 carbon atoms. Some or all of the hydrogen atoms of may be substituted with halogen atoms. Moreover, aromatic diamines may be used individually or may be used combining plurality.
또한, 상기 벤조옥사졸 구조를 갖는 방향족 디아민류로는 특별히 한정되지 않고, 예컨대, 5-아미노-2-(p-아미노페닐)벤조옥사졸, 6-아미노-2-(p-아미노페닐)벤조옥사졸, 5-아미노-2-(m-아미노페닐)벤조옥사졸, 6-아미노-2-(m-아미노페닐)벤조옥사졸, 2,2'-p-페닐렌비스(5-아미노벤조옥사졸), 2,2'-p-페닐렌비스(6-아미노벤조옥사졸), 1-(5-아미노벤조옥사졸로)-4-(6-아미노벤조옥사졸로)벤젠, 2,6-(4,4'-디아미노디페닐)벤조[1,2-d:5,4-d']비스옥사졸, 2,6-(4,4'-디아미노디페닐)벤조[1,2-d:4,5-d']비스옥사졸, 2,6-(3,4'-디아미노디페닐)벤조[1,2-d:5,4-d']비스옥사졸, 2,6-(3,4'-디아미노디페닐)벤조[1,2-d:4,5-d']비스옥사졸, 2,6-(3,3'-디아미노디페닐)벤조[1,2-d:5,4-d']비스옥사졸, 2,6-(3,3'-디아미노디페닐)벤조[1,2-d:4,5-d']비스옥사졸 등을 들 수 있다. 이들 중에서, 특히, 2,2'-디트리플루오로메틸-4,4'-디아미노비페닐, 4-아미노-N-(4-아미노페닐)벤즈아미드, 4,4'-디아미노디페닐술폰 또는 3,3'-디아미노벤조페논이 바람직하다. 또한, 옥사졸 구조를 갖는 방향족 디아민류는 단독으로 사용해도 좋고 복수를 조합하여 사용해도 좋다. In addition, the aromatic diamine having the benzoxazole structure is not particularly limited, and examples thereof include 5-amino-2-(p-aminophenyl)benzoxazole and 6-amino-2-(p-aminophenyl)benzo. Oxazole, 5-amino-2-(m-aminophenyl)benzoxazole, 6-amino-2-(m-aminophenyl)benzoxazole, 2,2'-p-phenylenebis(5-aminobenzo oxazole), 2,2'-p-phenylenebis(6-aminobenzoxazole), 1-(5-aminobenzooxazolo)-4-(6-aminobenzooxazolo)benzene, 2,6- (4,4'-diaminodiphenyl)benzo[1,2-d:5,4-d']bisoxazole, 2,6-(4,4'-diaminodiphenyl)benzo[1,2 -d:4,5-d']bisoxazole, 2,6-(3,4'-diaminodiphenyl)benzo[1,2-d:5,4-d']bisoxazole, 2, 6-(3,4'-diaminodiphenyl)benzo[1,2-d:4,5-d']bisoxazole, 2,6-(3,3'-diaminodiphenyl)benzo[1 ,2-d:5,4-d']bisoxazole, 2,6-(3,3'-diaminodiphenyl)benzo[1,2-d:4,5-d']bisoxazole, etc. can be heard Among these, in particular, 2,2'-ditrifluoromethyl-4,4'-diaminobiphenyl, 4-amino-N-(4-aminophenyl)benzamide, 4,4'-diaminodiphenyl Sulfone or 3,3'-diaminobenzophenone is preferred. Moreover, aromatic diamines which have an oxazole structure may be used independently or may be used combining plurality.
지환식 디아민류로는, 예컨대, 1,4-디아미노시클로헥산, 1,4-디아미노-2-메틸시클로헥산, 1,4-디아미노-2-에틸시클로헥산, 1,4-디아미노-2-n-프로필시클로헥산, 1,4-디아미노-2-이소프로필시클로헥산, 1,4-디아미노-2-n-부틸시클로헥산, 1,4-디아미노-2-이소부틸시클로헥산, 1,4-디아미노-2-sec-부틸시클로헥산, 1,4-디아미노-2-tert-부틸시클로헥산, 4,4'-메틸렌비스(2,6-디메틸시클로헥실아민), 시클로헥산-1,4-디일디메탄아민, 비시클로[2,2,1]헵탄-2,5-디아민 등을 들 수 있다. 이들 중에서, 특히, 1,4-디아미노시클로헥산 또는 1,4-디아미노-2-메틸시클로헥산이 바람직하고, 1,4-디아미노시클로헥산이 보다 바람직하다. 또한, 지환식 디아민류는 단독으로 사용해도 좋고 복수를 조합하여 사용해도 좋다. Examples of alicyclic diamines include 1,4-diaminocyclohexane, 1,4-diamino-2-methylcyclohexane, 1,4-diamino-2-ethylcyclohexane, and 1,4-diamino. -2-n-propylcyclohexane, 1,4-diamino-2-isopropylcyclohexane, 1,4-diamino-2-n-butylcyclohexane, 1,4-diamino-2-isobutylcyclo Hexane, 1,4-diamino-2-sec-butylcyclohexane, 1,4-diamino-2-tert-butylcyclohexane, 4,4'-methylenebis(2,6-dimethylcyclohexylamine), Cyclohexane-1,4-diyl dimethaneamine, bicyclo[2,2,1]heptane-2,5-diamine, etc. are mentioned. Among these, 1,4-diaminocyclohexane or 1,4-diamino-2-methylcyclohexane is particularly preferred, and 1,4-diaminocyclohexane is more preferred. Moreover, alicyclic diamines may be used independently or may be used combining plurality.
디이소시아네이트류로는, 예컨대, 디페닐메탄-2,4'-디이소시아네이트, 3,2'- 또는 3,3'- 또는 4,2'- 또는 4,3'- 또는 5,2'- 또는 5,3'- 또는 6,2'- 또는 6,3'-디메틸디페닐메탄-2,4'-디이소시아네이트, 3,2'- 또는 3,3'- 또는 4,2'- 또는 4,3'- 또는 5,2'- 또는 5,3'- 또는 6,2'- 또는 6,3'-디에틸디페닐메탄-2,4'-디이소시아네이트, 3,2'- 또는 3,3'- 또는 4,2'- 또는 4,3'- 또는 5,2'- 또는 5,3'- 또는 6,2'- 또는 6,3'-디메톡시디페닐메탄-2,4'-디이소시아네이트, 디페닐메탄-4,4'-디이소시아네이트, 디페닐메탄-3,3'-디이소시아네이트, 디페닐메탄-3,4'-디이소시아네이트, 디페닐에테르-4,4'-디이소시아네이트, 벤조페논-4,4'-디이소시아네이트, 디페닐술폰-4,4'-디이소시아네이트, 톨릴렌-2,4-디이소시아네이트, 톨릴렌-2,6-디이소시아네이트, m-크실릴렌디이소시아네이트, p-크실릴렌디이소시아네이트, 나프탈렌-2,6-디이소시아네이트, 4,4'-(2,2-비스(4-페녹시페닐)프로판)디이소시아네이트, 3,3'- 또는 2,2'-디메틸비페닐-4,4'-디이소시아네이트, 3,3'- 또는 2,2'-디에틸비페닐-4,4'-디이소시아네이트, 3,3'-디메톡시비페닐-4,4'-디이소시아네이트, 3,3'-디에톡시비페닐-4,4'-디이소시아네이트 등의 방향족 디이소시아네이트류, 및 이들 중 어느 하나를 수소 첨가한 디이소시아네이트(예컨대, 이소포론디이소시아네이트, 1,4-시클로헥산디이소시아네이트, 1,3-시클로헥산디이소시아네이트, 4,4'-디시클로헥실메탄디이소시아네이트, 헥사메틸렌디이소시아네이트) 등을 들 수 있다. 이들 중에서는, 저흡습성, 치수 안정성, 가격 및 중합성의 점에서 디페닐메탄-4,4'-디이소시아네이트, 톨릴렌-2,4-디이소시아네이트, 톨릴렌-2,6-디이소시아네이트, 3,3'-디메틸비페닐-4,4'-디이소시아네이트, 나프탈렌-2,6-디이소시아네이트, 4,4'-디시클로헥실메탄디이소시아네이트, 또는 1,4-시클로헥산디이소시아네이트가 바람직하다. 또한, 디이소시아네이트류는 단독으로 사용해도 좋고 복수를 조합하여 사용해도 좋다. Diisocyanates include, for example, diphenylmethane-2,4'-diisocyanate, 3,2'- or 3,3'- or 4,2'- or 4,3'- or 5,2'- or 5,3'- or 6,2'- or 6,3'-dimethyldiphenylmethane-2,4'-diisocyanate, 3,2'- or 3,3'- or 4,2'- or 4, 3'- or 5,2'- or 5,3'- or 6,2'- or 6,3'-diethyldiphenylmethane-2,4'-diisocyanate, 3,2'- or 3,3 '- or 4,2'- or 4,3'- or 5,2'- or 5,3'- or 6,2'- or 6,3'-dimethoxydiphenylmethane-2,4'-di Isocyanate, diphenylmethane-4,4'-diisocyanate, diphenylmethane-3,3'-diisocyanate, diphenylmethane-3,4'-diisocyanate, diphenylether-4,4'-diisocyanate, Benzophenone-4,4'-diisocyanate, diphenylsulfone-4,4'-diisocyanate, tolylene-2,4-diisocyanate, tolylene-2,6-diisocyanate, m-xylylene diisocyanate, p-xylylenediisocyanate, naphthalene-2,6-diisocyanate, 4,4'-(2,2-bis(4-phenoxyphenyl)propane)diisocyanate, 3,3'- or 2,2'- Dimethylbiphenyl-4,4'-diisocyanate, 3,3'- or 2,2'-diethylbiphenyl-4,4'-diisocyanate, 3,3'-dimethoxybiphenyl-4,4' -Aromatic diisocyanates such as diisocyanate and 3,3'-diethoxybiphenyl-4,4'-diisocyanate, and diisocyanates obtained by hydrogenating any of these diisocyanates (e.g., isophorone diisocyanate, 1,4 -Cyclohexane diisocyanate, 1,3-cyclohexane diisocyanate, 4,4'-dicyclohexylmethane diisocyanate, hexamethylene diisocyanate) and the like. Among these, diphenylmethane-4,4'-diisocyanate, tolylene-2,4-diisocyanate, tolylene-2,6-diisocyanate, 3, tolylene-2,4-diisocyanate, 3, 3'-dimethylbiphenyl-4,4'-diisocyanate, naphthalene-2,6-diisocyanate, 4,4'-dicyclohexylmethane diisocyanate, or 1,4-cyclohexane diisocyanate is preferred. In addition, diisocyanates may be used independently or may be used in combination of plurality.
본 발명의 폴리아미드산 용액, 폴리이미드 용액 및 폴리아미드이미드 용액에 이용되는 용매로는, 폴리이미드계 수지 또는 그 전구체를 용해시키는 것이라면 특별히 특정되지 않지만, 예컨대 N-메틸-2-피롤리돈, N,N-디메틸아세트아미드, N,N-디메틸포름아미드, 1,3-디메틸-2-이미다졸리디논, 테트라메틸우레아, 술포란, 디메틸술폭시드, γ-부티로락톤, 시클로헥사논, 시클로펜타논 등이 있다. 이들은 단독으로 사용해도 좋고, 2종 이상을 병용해도 좋다. 특히 생산성이나 필름의 광학 특성을 고려하면 유기 용제의 주성분으로서 N,N-디메틸아세트아미드를 이용하는 것이 바람직하다. 또한 이들 유기 용매와 더불어, 톨루엔, 크실렌 등의 빈용매를 폴리이미드계 수지 또는 그 전구체가 석출되지 않을 정도로 사용해도 좋다. The solvent used in the polyamic acid solution, polyimide solution and polyamideimide solution of the present invention is not particularly specified as long as it dissolves the polyimide resin or its precursor, but for example, N-methyl-2-pyrrolidone; N,N-dimethylacetamide, N,N-dimethylformamide, 1,3-dimethyl-2-imidazolidinone, tetramethylurea, sulfolane, dimethylsulfoxide, γ-butyrolactone, cyclohexanone, and cyclopentanone. These may be used independently and may use 2 or more types together. In particular, considering productivity and optical properties of the film, it is preferable to use N,N-dimethylacetamide as the main component of the organic solvent. In addition to these organic solvents, poor solvents such as toluene and xylene may be used to the extent that the polyimide-based resin or its precursor does not precipitate.
본 발명에서의 폴리이미드 필름의 두께는 3 μm 이상이 바람직하고, 보다 바람직하게는 11 μm 이상이고, 더욱 바람직하게는 24 μm 이상이다. 상기 폴리이미드 필름의 두께의 상한은 특별히 제한되지 않지만, 플렉시블 전자 디바이스로서 이용하기 위해서는 250 μm 이하인 것이 바람직하고, 보다 바람직하게는 150 μm 이하이며, 더욱 바람직하게는 100 μm 이하이다. The thickness of the polyimide film in the present invention is preferably 3 μm or more, more preferably 11 μm or more, still more preferably 24 μm or more. The upper limit of the thickness of the polyimide film is not particularly limited, but for use as a flexible electronic device, it is preferably 250 μm or less, more preferably 150 μm or less, still more preferably 100 μm or less.
본 발명의 폴리이미드 필름의 인장 탄성률은, MD 방향 및 TD 방향 모두 3 GPa 이상이 필요하고, 바람직하게는 4 GPa 이상이고, 보다 바람직하게는 5 GPa 이상이다. 상기 인장 탄성률이 3 GPa 이상이면, 폴리이미드 필름과 기능 소자가 박리되는 것을 회피할 수 있어, 취급성이 우수하다. 상기 MD 방향 및 TD 방향의 두 방향의 인장 탄성률은, 20 GPa 이하가 바람직하고, 보다 바람직하게는 12 GPa 이하이며, 더욱 바람직하게는 10 GPa 이하이다. 상기 인장 탄성률이 20 GPa 이하이면, 상기 폴리이미드 필름을 플렉시블한 필름으로서 사용할 수 있다. 또, 상기 폴리이미드 필름의 인장 탄성률이란, 폴리이미드 필름의 유동 방향(MD 방향)의 인장 탄성률 및 폭방향(TD 방향)의 인장 탄성률의 평균치를 가리킨다. 상기 폴리이미드 필름의 인장 탄성률의 측정 방법은, 실시예에 기재된 방법에 따른다. The tensile modulus of elasticity of the polyimide film of the present invention is required to be 3 GPa or more in both MD and TD directions, preferably 4 GPa or more, and more preferably 5 GPa or more. When the tensile modulus is 3 GPa or more, peeling between the polyimide film and the functional element can be avoided, and the handleability is excellent. The tensile modulus of elasticity in both the MD and TD directions is preferably 20 GPa or less, more preferably 12 GPa or less, still more preferably 10 GPa or less. If the said tensile modulus is 20 GPa or less, the said polyimide film can be used as a flexible film. In addition, the tensile modulus of elasticity of the polyimide film refers to the average value of the tensile modulus of elasticity in the flow direction (MD direction) and the tensile modulus of elasticity in the width direction (TD direction) of the polyimide film. The method for measuring the tensile modulus of elasticity of the polyimide film is based on the method described in Examples.
본 발명에서의 폴리이미드 필름의 30℃부터 300℃ 사이의 평균 CTE는, -5 ppm/℃∼+55 ppm/℃인 것이 필요하다. 바람직하게는 -4 ppm/℃∼+45 ppm/℃이며, 보다 바람직하게는 -3 ppm/℃∼+35 ppm/℃이며, 더욱 바람직하게는 -2 ppm/℃∼+20 ppm/℃이며, 더욱 더 바람직하게는 +1 ppm/℃∼+10 ppm/℃이다. CTE가 상기 범위이면, 기능 소자와의 선팽창계수의 차를 작게 유지할 수 있고, 열을 가하는 프로세스에 제공하더라도 폴리이미드 필름과 기능 소자가 박리되는 것을 회피할 수 있어, 가공성이 우수하다. 여기서 CTE란 온도에 대하여 가역적인 신축을 나타내는 요인이다. 또, 상기 폴리이미드 필름의 CTE란, 폴리이미드 필름의 유동 방향(MD 방향)의 CTE 및 폭방향(TD 방향)의 CTE의 평균치를 가리킨다. 상기 폴리이미드 필름의 CTE의 측정 방법은, 실시예에 기재된 방법에 따른다. The average CTE between 30°C and 300°C of the polyimide film in the present invention is required to be -5 ppm/°C to +55 ppm/°C. Preferably -4 ppm/°C to +45 ppm/°C, more preferably -3 ppm/°C to +35 ppm/°C, still more preferably -2 ppm/°C to +20 ppm/°C, Even more preferably, it is +1 ppm/°C to +10 ppm/°C. When the CTE is within the above range, the difference in coefficient of linear expansion with the functional element can be kept small, and separation between the polyimide film and the functional element can be avoided even when subjected to a heat application process, resulting in excellent workability. Here, CTE is a factor representing reversible expansion and contraction with respect to temperature. In addition, the CTE of the said polyimide film refers to the average value of CTE of the flow direction (MD direction) of a polyimide film, and CTE of the width direction (TD direction). The method for measuring the CTE of the polyimide film is based on the method described in Examples.
본 발명의 인장 탄성률과 CTE를 나타내는 폴리이미드 필름은, 폴리이미드 필름의 성막 과정에서 연신을 행함으로써 실현할 수 있다. 이러한 연신 조작은, 폴리이미드 용액을 폴리이미드 필름 제작용 지지체에 도포, 건조하여 1∼50 질량%의 용매를 포함하는 폴리이미드 필름으로 하고, 또한 폴리이미드 필름 제작용 지지체 상에서, 혹은 상기 지지체로부터 박리한 상태에서, 1∼50 질량%의 용매를 포함하는 폴리이미드 필름을 고온 처리하여 건조시키는 과정에서, MD 방향으로 1.5배로부터 4.0배로, TD 방향으로 1.4배로부터 3.0배로 연신함으로써 실현할 수 있다. 이 때 폴리이미드 필름 제작용 지지체에 미연신의 열가소성 고분자 필름을 이용하여, 열가소성 고분자 필름과 폴리이미드 필름을 동시에 연신한 후에 열가소성 고분자 필름으로부터 연신후의 폴리이미드 필름을 박리하는 것에 의해, 특히 MD 방향의 연신시에 폴리이미드 필름에 생기는 손상을 방지할 수 있고, 보다 고품위의 무색 투명성이 높은 폴리이미드 필름을 얻을 수 있다. MD 방향의 바람직한 연신 배율은 1.7배∼3.5배이며, 보다 바람직하게는 2.0배∼3.0배이다. 또한, TD 방향의 바람직한 연신 배율은 1.7배∼3.5배이며, 보다 바람직하게는 2.0배∼3.0배이다. MD 방향의 연신 배율과 TD 방향의 연신 배율의 비율(MD/TD)은, 1 초과인 것이 바람직하고, 보다 바람직하게는 1.01 이상이고, 더욱 바람직하게는 1.05 이상이고, 보다 더 바람직하게는 1.08 이상이고, 특히 바람직하게는 1.1 이상이다. 또한, 2.0 이하인 것이 바람직하고, 보다 바람직하게는 1.8 이하이며, 더욱 바람직하게는 1.5 이하이며, 특히 바람직하게는 1.2 이하이다. The polyimide film exhibiting the tensile modulus and CTE of the present invention can be realized by stretching in the film forming process of the polyimide film. In this stretching operation, the polyimide solution is applied to a support for production of a polyimide film and dried to obtain a polyimide film containing 1 to 50% by mass of a solvent, and then peeled on or from the support for production of a polyimide film. It can be realized by stretching a polyimide film containing 1 to 50% by mass of a solvent at a high temperature and drying it, by stretching it from 1.5 times to 4.0 times in the MD direction and from 1.4 times to 3.0 times in the TD direction. At this time, an unstretched thermoplastic polymer film is used as a support for producing a polyimide film, the thermoplastic polymer film and the polyimide film are simultaneously stretched, and then the stretched polyimide film is separated from the thermoplastic polymer film, particularly in the MD direction. Damage to the polyimide film at the time of application can be prevented, and a higher quality, colorless and transparent polyimide film can be obtained. The preferred stretch ratio in the MD direction is 1.7 to 3.5 times, more preferably 2.0 to 3.0 times. In addition, the preferred draw ratio in the TD direction is 1.7 to 3.5 times, more preferably 2.0 to 3.0 times. The ratio of the draw ratio in the MD direction to the draw ratio in the TD direction (MD/TD) is preferably greater than 1, more preferably 1.01 or more, still more preferably 1.05 or more, still more preferably 1.08 or more. , and is particularly preferably 1.1 or more. Further, it is preferably 2.0 or less, more preferably 1.8 or less, still more preferably 1.5 or less, and particularly preferably 1.2 or less.
본 발명의 폴리이미드 필름의 용매 함유량은, 0.5∼5.0 질량%인 것이 필요하다. 바람직하게는 0.7∼4.0 질량%의 범위, 보다 바람직하게는 1.0∼3.0 질량%의 범위이다. 용매 함유량을 상기 하한치 이상으로 하는 것에 의해, 과잉 고온 처리에 의해 폴리이미드 필름 표면이 과도하게 불활성이 되는 일이 없고, 후술하는 자기 수복층을 형성하는 중합체 조성물에 대한 밀착성이 유지되고, 또한, 옐로우 인덱스의 악화를 억제할 수 있다. 또한, 용매 함유량을 상기 상한치 이하로 하는 것에 의해, 인장 탄성률 및 CTE를 바람직한 범위로 하는 것이 용이해지고, 또한 자기 수복층에 잔류 용매가 이행하여 백화, 실투하는 것을 억제하여, 파장 400 nm에서의 광선 투과율이나 전광선 투과율의 악화를 억제할 수 있다. The solvent content of the polyimide film of the present invention needs to be 0.5 to 5.0% by mass. It is preferably in the range of 0.7 to 4.0% by mass, more preferably in the range of 1.0 to 3.0% by mass. By setting the solvent content to the above lower limit or more, the surface of the polyimide film does not become excessively inactive due to excessive high-temperature treatment, and adhesion to the polymer composition forming the self-healing layer described later is maintained, and yellow Deterioration of the index can be suppressed. Further, by setting the solvent content to the above upper limit or less, it becomes easy to set the tensile modulus and CTE within a preferable range, and whitening and devitrification due to transfer of residual solvent to the self-healing layer are suppressed, and light at a wavelength of 400 nm is suppressed. Deterioration of transmittance or total light transmittance can be suppressed.
폴리이미드 필름에 함유하는 용매는, 폴리이미드 필름 제작 과정(예컨대, 폴리이미드 필름을 고온 처리하여 건조시키는 과정)에서의 용매의 잔류물(잔류 용매)이라도 상관없고, 폴리이미드 필름을 제작한 후에 용매를 첨가한 것이라도 상관없다. 바람직하게는 폴리이미드 필름 제작 과정에서의 잔류 용매이다. The solvent contained in the polyimide film may be a residue (residual solvent) of the solvent in the polyimide film production process (eg, a process of drying the polyimide film by treating it at a high temperature), and the solvent after the polyimide film is produced. It doesn't matter if you added . Preferably, it is a residual solvent in the polyimide film manufacturing process.
폴리이미드 필름 및/또는 그린 필름의 건조 조건으로는 특별히 한정되지 않고, 1단계로 건조시켜도 좋고, 다단계로 건조시켜도 좋다. 다단계로 건조시키는 것이 바람직하다. 다단계 건조시의 건조 단계수로는, 특별히 한정되지 않고, 2단계 이상인 것이 바람직하고, 보다 바람직하게는 3단계 이상이다. 또한, 10단계 이하인 것이 바람직하고, 보다 바람직하게는 5단계 이하이다. 다단계에서의 건조 온도는 후단이 될수록 고온으로 하는 것이 바람직하다. 예컨대, 2단계 건조의 경우, 1단계째는 100℃ 이상 250℃ 이하인 것이 바람직하고, 보다 바람직하게는 120℃ 이상 200℃ 이하이며, 더욱 바람직하게는 150℃ 이상 180℃ 이하이다. 2단계째는 250℃ 초과 500℃ 이하인 것이 바람직하고, 보다 바람직하게는 280℃ 이상 450℃ 이하이며, 더욱 바람직하게는 300℃ 이상 400℃ 이하이다. Drying conditions for the polyimide film and/or green film are not particularly limited, and may be dried in one step or in multiple steps. Drying in multiple stages is preferred. The number of drying stages in multi-stage drying is not particularly limited, and is preferably two or more stages, and more preferably three or more stages. Moreover, it is preferable that it is 10 steps or less, More preferably, it is 5 steps or less. It is preferable to make the drying temperature in a multi-stage higher as it becomes the next stage. For example, in the case of two-step drying, the first step is preferably 100°C or more and 250°C or less, more preferably 120°C or more and 200°C or less, and still more preferably 150°C or more and 180°C or less. In the second stage, it is preferably more than 250°C and less than 500°C, more preferably more than 280°C and less than 450°C, and still more preferably more than 300°C and less than 400°C.
또한, 건조 시간은 상기 건조 온도나 막두께, 용제의 종류, 사용하는 건조 기기에 따라 설정할 수 있다. 예컨대, 텐터를 사용하고 상기 2단계 건조인 경우라면, 1단계째의 건조 시간은 2분 이상 15분 이하인 것이 바람직하고, 보다 바람직하게는 3분 이상 12분 이하이며, 더욱 바람직하게는 5분 이상 8분 이하이다. 2단계째의 건조 시간은 1분 초과 10분 이하인 것이 바람직하고, 보다 바람직하게는 2분 이상 8분 이하이며, 더욱 바람직하게는 3분 이상 5분 이하이다. 상기 범위 내로 함으로써 폴리이미드 필름의 용매 함유량을 소정 범위 내로 할 수 있다. In addition, the drying time can be set according to the said drying temperature, film thickness, the kind of solvent, and the drying apparatus used. For example, in the case of the two-step drying using a tenter, the drying time of the first step is preferably 2 minutes or more and 15 minutes or less, more preferably 3 minutes or more and 12 minutes or less, and still more preferably 5 minutes or more less than 8 minutes The drying time in the second step is preferably more than 1 minute and 10 minutes or less, more preferably 2 minutes or more and 8 minutes or less, still more preferably 3 minutes or more and 5 minutes or less. By setting it in the said range, the solvent content of a polyimide film can be made into a predetermined range.
또한, 3단계 건조의 경우, 1단계째는 100℃ 이상 200℃ 이하인 것이 바람직하고, 보다 바람직하게는 120℃ 이상 190℃ 이하이며, 더욱 바람직하게는 150℃ 이상 180℃ 이하이다. 2단계째는 200℃ 초과 300℃ 이하인 것이 바람직하고, 보다 바람직하게는 210℃ 이상 280℃ 이하이며, 더욱 바람직하게는 220℃ 이상 250℃ 이하이다. 3단계째는 300℃ 초과 500℃ 이하인 것이 바람직하고, 보다 바람직하게는 320℃ 이상 450℃ 이하이며, 더욱 바람직하게는 340℃ 이상 400℃ 이하이다. 또한, 건조 시간은 건조 온도에 따라 설정할 수 있다. 예컨대, 상기 3단계 건조인 경우라면, 1단계째의 건조 시간은 30초 이상 10분 이하인 것이 바람직하고, 보다 바람직하게는 1분 이상 8분 이하이며, 더욱 바람직하게는 2분 이상 5분 이하이다. 2단계째의 건조 시간은 30초 이상 10분 이하인 것이 바람직하고, 보다 바람직하게는 1분 이상 8분 이하이며, 더욱 바람직하게는 2분 이상 5분 이하이다. 3단계째의 건조 시간은 1분 초과 10분 미만인 것이 바람직하고, 보다 바람직하게는 2분 이상 9분 이하이며, 더욱 바람직하게는 3분 이상 8분 이하이다. 상기 범위 내로 함으로써 폴리이미드 필름의 용매 함유량을 소정 범위 내로 할 수 있다. Further, in the case of three-stage drying, the first stage is preferably 100°C or more and 200°C or less, more preferably 120°C or more and 190°C or less, and still more preferably 150°C or more and 180°C or less. In the second step, it is preferably more than 200°C and not more than 300°C, more preferably 210°C or more and 280°C or less, still more preferably 220°C or more and 250°C or less. The third step is preferably more than 300°C and less than 500°C, more preferably more than 320°C and less than 450°C, still more preferably more than 340°C and less than 400°C. In addition, the drying time can be set according to the drying temperature. For example, in the case of the three-step drying, the drying time of the first step is preferably 30 seconds or more and 10 minutes or less, more preferably 1 minute or more and 8 minutes or less, and still more preferably 2 minutes or more and 5 minutes or less. . The drying time in the second step is preferably 30 seconds or more and 10 minutes or less, more preferably 1 minute or more and 8 minutes or less, still more preferably 2 minutes or more and 5 minutes or less. The drying time in the third step is preferably more than 1 minute and less than 10 minutes, more preferably 2 minutes or more and 9 minutes or less, still more preferably 3 minutes or more and 8 minutes or less. By setting it in the said range, the solvent content of a polyimide film can be made into a predetermined range.
본 발명의 적층체는 상기 인장 탄성률과 CTE를 나타내는 폴리이미드 필름의 적어도 한쪽 면에 자기 수복층이 적층된 것이며, 이것에 의해 적층체의 자기 수복성이 확보된다. In the laminate of the present invention, a self-healing layer is laminated on at least one side of the polyimide film exhibiting the tensile modulus and CTE, and thereby the self-healing property of the laminate is secured.
- 자기 수복성의 정의-- Definition of self-healing -
여기서, 자기 수복성이란, 응력 집중에 의한 왜곡에 의해 생긴 변형이나 손상을 응력의 하중 제거시에 복원하는 성질을 가리키며, 구체적으로는 실시예에 기재된 측정 방법에 의해 구해지는 「복귀율」에 의해 평가된다. Here, the self-healing property refers to the property of restoring deformation or damage caused by distortion due to stress concentration when the stress load is removed. Specifically, by the "recovery rate" obtained by the measurement method described in the examples It is evaluated.
본 발명의 적층체는 자기 수복성으로서, 상기 복귀율의 측정에 의해 계산되는 복귀율이 80% 이상인 것이 바람직하고, 85% 이상이 보다 바람직하고, 90% 이상이 더욱 바람직하고, 95% 이상이 보다 더 바람직하고, 100%가 특히 바람직하다. The laminate of the present invention has self-healing properties, and the recovery rate calculated by measuring the recovery rate is preferably 80% or more, more preferably 85% or more, more preferably 90% or more, and 95% or more It is even more preferable, and 100% is particularly preferable.
본 발명의 적층체를 구성하는 자기 수복층은, 주로 화상 표시 장치의 전면(前面)에 형성되기 때문에 저태크성이나 방오성, 내약품성을 필요로 하는 관점에서 가교점을 가지며, 또한 자기 수복성으로서 복귀율이 80% 이상을 달성하는 관점에서, 가교점의 일부 또는 전부가 가동성을 갖는 가교점을 포함하는 중합체 조성물(이하, 「자기 수복성 중합체 조성물」이라고도 함)인 것이 바람직하다. Since the self-healing layer constituting the laminate of the present invention is mainly formed on the front surface of an image display device, it has a crosslinking point from the viewpoint of requiring low tack, antifouling, and chemical resistance, and also has self-healing properties. From the viewpoint of achieving a return ratio of 80% or more, it is preferable that part or all of the crosslinking points are a polymer composition (hereinafter also referred to as "self-healing polymer composition") containing movable crosslinking points.
본 발명에서의 가동성을 갖는 가교점은, 제1 형태로서 공유 결합 이외의 화학 결합에 의해 형성된 가교점을 나타내고, 공유 결합 이외의 화학 결합으로는, 이온 결합, 수소 결합을 들 수 있다. 이러한 화학 결합은, 응력 집중에 의해 생긴 왜곡이 전파되면, 결합이 절단되어 변형이나 손상이 생기지만, 응력을 제거한 후에 분자 운동에 의해 결합이 재생성되기 때문에, 변형이나 손상이 소실되어 자기 수복성을 갖는다. The movable cross-linking point in the present invention represents a cross-linking point formed by a chemical bond other than a covalent bond as a first form, and an ionic bond or a hydrogen bond is exemplified as a chemical bond other than a covalent bond. When distortion caused by stress concentration propagates, these chemical bonds break and cause deformation or damage. However, since the bond is regenerated by molecular motion after the stress is removed, the deformation or damage disappears and exhibits self-healing properties. have
본 발명에서의 가동성을 갖는 가교점은, 제2 형태로서 화학 결합에 의하지 않는 기하형상에 의한 구속으로 이루어진 가교점을 나타내고, 기하형상에 의한 구속으로는, 고리형 분자의 개구부에 직쇄형 분자가 관통한 구속, 고리형 분자끼리 개구부를 관통한 구속이 있다. 이러한 구속은, 응력 집중에 의해 생긴 왜곡이 전파되면, 분자 결합이 절단되는 대신에 구속 개소가 이동하여 변형이나 손사이 생기지만, 응력을 제거한 후에 분자 운동에 의해 구속 개소가 재구성되기 때문에, 변형이나 손사이 소실되어 자기 수복성을 갖는다. The movable cross-linking point in the present invention represents a cross-linking point consisting of constraints by geometric shapes that do not depend on chemical bonds as a second form, and in the constraints by geometric shapes, straight-chain molecules are formed at the openings of cyclic molecules. There are constraints that pass through, and constraints in which ring-shaped molecules pass through openings. When the distortion caused by stress concentration propagates, the constraint points move instead of breaking the molecular bonds, resulting in deformation or loss. Damage is lost and it has self-healing properties.
본 발명에서의 가동성을 갖는 가교점으로서 제1 형태인 공유 결합 이외의 화학 결합에 의해 형성된 가교점을 갖는 중합체 조성물로는, 이온 결합을 이용한 중합체 조성물로서, 금속 이온 함유 아크릴산에틸렌 공중합체, 금속 이온 함유 (메트)아크릴산에틸렌 공중합체, 금속 이온 함유 술폰산폴리에스테르 중합체, 금속 이온 함유 술폰산폴리에스테르폴리에테르 공중합체를 들 수 있고, 수소 결합을 이용한 중합체 조성물로서, 폴리에테르우레탄 공중합체, 지방족 폴리에스테르우레탄 공중합체, 폴리에테르폴리에스테르우레탄 공중합체, 폴리우레탄아크릴레이트 공중합체, 폴리에테르폴리아미드 공중합체, 지방족 폴리에스테르폴리아미드 공중합체를 들 수 있다. As the crosslinking point having mobility in the present invention, the polymer composition having a crosslinking point formed by a chemical bond other than a covalent bond, which is the first form, is a polymer composition using an ionic bond, such as a metal ion-containing ethylene acrylate copolymer, a metal ion (meth)acrylate-containing ethylene copolymers, metal ion-containing sulfonic acid polyester polymers, and metal ion-containing sulfonic acid polyester polyether copolymers, as polymer compositions utilizing hydrogen bonds, polyetherurethane copolymers and aliphatic polyesterurethanes. copolymers, polyether polyester urethane copolymers, polyurethane acrylate copolymers, polyether polyamide copolymers, and aliphatic polyester polyamide copolymers.
본 발명에서의 가동성을 갖는 가교점으로서 제2 형태인 기하형상에 의한 구속으로 이루어진 가교점을 갖는 중합체 조성물로는, 고리형 폴리에테르 고리 관통 폴리에테르 중합체, 고리형 폴리에테르 고리 관통 지방족 폴리에스테르 공중합체, 고리형 폴리에테르 고리 관통 폴리아크릴 중합체, 시클로덱스트린 고리 관통 폴리에테르 중합체, 시클로덱스트린 고리 관통 지방족 폴리에스테르 공중합체를 들 수 있다. As the crosslinking point having mobility in the present invention, the polymer composition having a crosslinking point composed of constraints by the geometric shape, which is the second type, is a cyclic polyether ring piercing polyether polymer, a cyclic polyether ring piercing aliphatic polyester copolymer polymers, cyclic polyether penetrating polyacryl polymers, cyclodextrin penetrating polyether polymers, and cyclodextrin penetrating aliphatic polyester copolymers.
자기 수복층에 포함되는 상기 자기 수복성 중합체 조성물의 함유량은, 90 질량% 이상인 것이 바람직하고, 보다 바람직하게는 95 질량% 이상이고, 더욱 바람직하게는 99 질량% 이상이고, 특히 바람직하게는 99.9 질량% 이상이고, 100 질량%이라도 지장없다. The content of the self-healing polymer composition contained in the self-healing layer is preferably 90% by mass or more, more preferably 95% by mass or more, even more preferably 99% by mass or more, and particularly preferably 99.9% by mass. It is % or more, and even if it is 100 mass %, there is no problem.
상기 가교점의 일부 또는 전부가 가동성을 갖는 가교점을 포함하는 중합체 조성물은, 단독으로 사용해도 좋고 복수를 조합하여 사용해도 좋고, 필요에 따라서 용매, 연화제, 가소제, 노화 방지제, 산화 방지제, 자외선 흡수제, 광안정제, 표면 윤활제, 레벨링제, 방오제, 이형제, 내열안정제, 윤활제, 무기 미립자, 계면 활성제 등, 본 발명이 속하는 기술분야에서 통상 사용되는 첨가제를 추가적으로 포함할 수 있다. 또한, 그 함유량은, 본 발명의 적층체의 물성을 저하시키지 않는 범위 내에서 다양하게 조절할 수 있기 때문에, 특별히 제한은 없지만, 예컨대 상기 자기 수복층 100 질량부에 대하여 약 0.1∼ 약 10 질량부로 포함되어도 좋다. The polymer composition, in which some or all of the crosslinking points contain a crosslinking point having mobility, may be used alone or in combination of a plurality of them, and, if necessary, a solvent, a softener, a plasticizer, an antiaging agent, an antioxidant, an ultraviolet absorber , light stabilizers, surface lubricants, leveling agents, antifouling agents, release agents, heat stabilizers, lubricants, inorganic fine particles, surfactants, and the like, additives commonly used in the art to which the present invention pertains may be further included. In addition, the content can be variously adjusted within the range of not deteriorating the physical properties of the laminate of the present invention, so there is no particular limitation, but, for example, it is included in about 0.1 to about 10 parts by mass based on 100 parts by mass of the self-healing layer. It may be.
본 발명의 적층체를 구성하는 자기 수복층의 두께는, 밀착성을 확보하고, 자기 수복 기능을 충분히 발현시킨다고 하는 관점에서, 5 μm 이상이 바람직하고, 8 μm 이상이 보다 바람직하고, 10 μm 이상이 특히 바람직하다. 한편, 투명성, 가공성, 성형성을 확보하는 관점에서, 상한의 두께는 100 μm 이하가 바람직하고, 80 μm 이하가 보다 바람직하고, 50 μm 이하가 특히 바람직하다. The thickness of the self-healing layer constituting the laminate of the present invention is preferably 5 μm or more, more preferably 8 μm or more, and more preferably 10 μm or more, from the viewpoint of securing adhesion and sufficiently expressing the self-healing function. particularly preferred. On the other hand, from the viewpoint of ensuring transparency, workability and moldability, the upper limit of the thickness is preferably 100 μm or less, more preferably 80 μm or less, and particularly preferably 50 μm or less.
본 발명의 적층체를 구성하는 자기 수복층은, 폴리이미드 필름 상에 자기 수복성 중합체 조성물을 도포하고, 필요에 따라서 건조시킨 후, 열 혹은 활성 에너지선을 조사하고 경화시키는 것에 의해 형성할 수 있다. 즉, 본 발명에 이러한 자기 수복성층을 형성하기 위한 자기 수복성 중합체 조성물은, 열경화성 중합체 조성물 혹은 활성 에너지선 경화성 중합체 조성물인 것이 바람직하다. The self-healing layer constituting the laminate of the present invention can be formed by applying a self-healing polymer composition on a polyimide film, drying it as necessary, and then curing it by irradiating heat or active energy rays. . That is, the self-healing polymer composition for forming such a self-healing layer in the present invention is preferably a thermosetting polymer composition or an active energy ray-curable polymer composition.
자기 수복성 중합체 조성물의 도포에 이용되는 도포 방법으로는, 웨트 코팅법이 바람직하다. 이러한 웨트 코팅법으로는, 예컨대 리버스코트법, 스프레이코트법, 바코트법, 그라비아코트법, 로드코트법, 다이코트법, 롤코트법, 립코트법 등을 들 수 있다. As a coating method used for coating the self-healing polymer composition, a wet coating method is preferred. Examples of such a wet coating method include a reverse coating method, a spray coating method, a bar coating method, a gravure coating method, a rod coating method, a die coating method, a roll coating method, and a lip coating method.
적층체의 바람직한 두께는, 8 μm 이상이고, 보다 바람직하게는 15 μm이며, 더욱 바람직하게는 20 μm 이상이다. 또한, 300 μm 이하인 것이 바람직하고, 보다 바람직하게는 200 μm 이하이며, 더욱 바람직하게는 100 μm 이하이다. The thickness of the laminate is preferably 8 μm or more, more preferably 15 μm or more, still more preferably 20 μm or more. Further, it is preferably 300 μm or less, more preferably 200 μm or less, and still more preferably 100 μm or less.
본 발명의 적층체는, 폴리이미드 필름, 자기 수복층이 이 순으로 적층된 것이며, 자기 수복층은 폴리이미드 필름에 대하여 밀착성을 가지며, 그 밀착성은 구체적으로 실시예에 기재된 측정 방법에 의해 구해지는 「부착률」에 의해 평가된다. In the laminate of the present invention, a polyimide film and a self-healing layer are laminated in this order, and the self-healing layer has adhesion to the polyimide film, and the adhesion can be specifically obtained by the measurement method described in the Examples. It is evaluated by "attachment rate".
본 발명의 적층체는 자기 수복층의 밀착성으로서, 상기 부착률이 80% 이상인 것이 바람직하고, 85% 이상이 보다 바람직하고, 90% 이상이 더욱 바람직하고, 95% 이상이 보다 더 바람직하고, 100%가 특히 바람직하다. 부착률은 전술한 바와 같이, 폴리이미드 필름에 포함되는 용매 함유량을 전술한 하한치 이상으로 하는 것에 의해 바람직한 값을 달성할 수 있다. 또한, 폴리이미드 필름에는, 투명성이나 자기 수복층과의 밀착성을 저하시키지 않는 범위에서, 표면에 전처리로서 코로나 처리, 플라즈마 처리, 오존 처리, 약품 처리, 용매 처리 등이 실시되어도 좋다. As the adhesiveness of the self-healing layer of the laminate of the present invention, the adhesion rate is preferably 80% or more, more preferably 85% or more, still more preferably 90% or more, still more preferably 95% or more, and 100% or more. % is particularly preferred. As described above, the adhesion rate can achieve a desirable value by setting the solvent content in the polyimide film to the lower limit value or more. In addition, the polyimide film may be subjected to corona treatment, plasma treatment, ozone treatment, chemical treatment, solvent treatment, etc. as a pretreatment on the surface within a range that does not reduce the transparency or adhesion to the self-healing layer.
본 발명의 적층체는 주로 터치패널이나 디스플레이 등의 화상 표시 장치의 전면판, 전극 주변에 이용되기 때문에, 황색도 지수(옐로우 인덱스)는 10 이하가 바람직하고, 보다 바람직하게는 7 이하이며, 더욱 바람직하게는 5 이하이며, 한층 더 바람직하게는 3 이하이다. 상기 폴리이미드 필름의 황색도의 하한은 특별히 제한되지 않지만, 플렉시블 전자 디바이스로서 이용하기 위해서는 0.1 이상인 것이 바람직하고, 보다 바람직하게는 0.2 이상이고, 더욱 바람직하게는 0.3 이상이다. Since the laminate of the present invention is mainly used around the front plate and electrodes of image display devices such as touch panels and displays, the yellowness index (yellow index) is preferably 10 or less, more preferably 7 or less, and even more Preferably it is 5 or less, More preferably, it is 3 or less. The lower limit of the yellowness of the polyimide film is not particularly limited, but for use as a flexible electronic device, it is preferably 0.1 or more, more preferably 0.2 or more, still more preferably 0.3 or more.
본 발명의 적층체는 주로 터치패널이나 디스플레이 등의 화상 표시 장치의 전면판, 전극 주변에 이용되기 때문에, 파장 400 nm에서의 광선 투과율은 70% 이상이 바람직하고, 보다 바람직하게는 72% 이상이고, 더욱 바람직하게는 75% 이상이고, 한층 더 바람직하게는 80% 이상이다. 상기 폴리이미드 필름의 파장 400 nm의 광선 투과율의 상한은 특별히 제한되지 않지만, 플렉시블 전자 디바이스로서 이용하기 위해서는 99% 이하인 것이 바람직하고, 보다 바람직하게는 98% 이하이며, 더욱 바람직하게는 97% 이하이다. Since the laminate of the present invention is mainly used around the front plate and electrodes of image display devices such as touch panels and displays, the light transmittance at a wavelength of 400 nm is preferably 70% or more, more preferably 72% or more, , More preferably, it is 75% or more, and even more preferably, it is 80% or more. The upper limit of the light transmittance of the polyimide film at a wavelength of 400 nm is not particularly limited, but for use as a flexible electronic device, it is preferably 99% or less, more preferably 98% or less, still more preferably 97% or less. .
본 발명의 적층체는 주로 터치패널이나 디스플레이 등의 화상 표시 장치의 전면판, 전극 주변에 이용되기 때문에, 전광선 투과율은 85% 이상이 바람직하고, 보다 바람직하게는 86% 이상이고, 더욱 바람직하게는 87% 이상이고, 한층 더 바람직하게는 88% 이상이다. 상기 폴리이미드 필름의 전광선 투과율의 상한은 특별히 제한되지 않지만, 플렉시블 전자 디바이스로서 이용하기 위해서는 99% 이하인 것이 바람직하고, 보다 바람직하게는 98% 이하이며, 더욱 바람직하게는 97% 이하이다. Since the laminate of the present invention is mainly used around the front plate and electrodes of image display devices such as touch panels and displays, the total light transmittance is preferably 85% or more, more preferably 86% or more, still more preferably It is 87% or more, and more preferably 88% or more. The upper limit of the total light transmittance of the polyimide film is not particularly limited, but for use as a flexible electronic device, it is preferably 99% or less, more preferably 98% or less, still more preferably 97% or less.
실시예Example
이하, 본 발명에 관해 실시예를 이용하여 상세히 설명하지만, 본 발명은 그 요지를 벗어나지 않는 한, 이하의 실시예에 한정되는 것이 아니다. Hereinafter, the present invention will be described in detail using Examples, but the present invention is not limited to the following Examples unless departing from the gist thereof.
한편 실시예, 비교예에서의 각 측정치는, 특별히 언급하지 않는 한 이하의 방법으로 측정했다. On the other hand, each measured value in Examples and Comparative Examples was measured by the following method unless otherwise noted.
<폴리아미드산 및 폴리이미드의 환원 점도> <Reduced viscosity of polyamic acid and polyimide>
폴리머 농도가 0.2 g/dl이 되도록 N-메틸-2-피롤리돈(또는 N,N-디메틸아세트아미드)에 용해한 용액을 우베로데형의 점도관에 의해 30℃에서 측정했다. (폴리아미드산 용액의 조제에 사용한 용매가 N,N-디메틸아세트아미드인 경우는, N,N-디메틸아세트아미드를 사용하여 폴리머를 용해하여 측정했다.)A solution dissolved in N-methyl-2-pyrrolidone (or N,N-dimethylacetamide) to a polymer concentration of 0.2 g/dl was measured at 30°C with an Uberode viscometer. (When the solvent used to prepare the polyamic acid solution was N,N-dimethylacetamide, the measurement was performed by dissolving the polymer using N,N-dimethylacetamide.)
<폴리이미드 필름 및 적층체의 두께> <Thickness of polyimide film and laminate>
마이크로미터(파인류프사 제조, 밀리트론 1245D)를 이용하여 측정했다. It was measured using a micrometer (Militron 1245D, manufactured by Fine Lupe Co., Ltd.).
<폴리이미드 필름의 인장 탄성률> <Tensile modulus of elasticity of polyimide film>
폴리이미드 필름의 유동 방향(MD 방향) 및 폭방향(TD 방향)으로 각각 100 mm×10 mm의 세그먼트형으로 잘라낸 것을 시험편으로 했다. 시험편은, 폭방향 중앙 부분으로부터 잘라냈다. 인장 시험기(시마즈 제작소 제조, 오토그래프(R), 기종명 AG-5000A)를 이용하여, 온도 25℃, 인장 속도 50 mm/분, 척간 거리 40 mm의 조건으로, MD 방향, TD 방향 각각 샘플 5점에 관해 인장 탄성률을 측정하고, 전체 측정치의 평균치를 구했다. What was cut into segments of 100 mm x 10 mm in the flow direction (MD direction) and width direction (TD direction) of the polyimide film, respectively, was used as a test piece. The test piece was cut out from the central part in the width direction. Using a tensile tester (manufactured by Shimadzu Corporation, Autograph (R), model name AG-5000A), 5 samples each in the MD direction and the TD direction under the conditions of a temperature of 25 ° C, a tensile speed of 50 mm / min, and a distance between chucks of 40 mm. The tensile elastic modulus was measured, and the average value of all the measured values was obtained.
<폴리이미드 필름의 선팽창계수(CTE)> <Coefficient of linear expansion (CTE) of polyimide film>
폴리이미드 필름의 유동 방향(MD 방향) 및 폭방향(TD 방향) 각각 샘플 5점에서, 하기 조건으로 신축률을 측정하고, 30℃∼45℃, 45℃∼60℃와 같이 15℃ 간격에서의 신축률/온도를 측정하고, 이 측정을 300℃까지 행하여, 전체 측정치의 평균치를 CTE로서 산출했다. Stretching ratio was measured at 5 samples each in the flow direction (MD direction) and width direction (TD direction) of the polyimide film under the following conditions, and at 15 ° C intervals such as 30 ° C to 45 ° C and 45 ° C to 60 ° C. Stretching ratio/temperature was measured, this measurement was performed up to 300°C, and the average value of all measured values was calculated as CTE.
기기명 : MAC 사이언스사 제조 TMA4000SDevice name: TMA4000S manufactured by MAC Science
시료 길이 : 20 mmSample length: 20 mm
시료폭 : 2 mmSample width: 2 mm
승온 개시 온도 : 25℃Heating start temperature: 25°C
승온 종료 온도 : 400℃Heating end temperature: 400℃
승온 속도: 5℃/minHeating rate: 5°C/min
분위기 : 아르곤Atmosphere: Argon
<폴리이미드 필름 중의 용매 함유량> <Solvent content in polyimide film>
폴리이미드 필름 중의 용매 함유량은 열중량 분석 장치(TA Instruments 제조, 기종명 TGA2950)를 이용하여 측정했다. 약 10 mg의 샘플을 알루미늄제의 마이크로셀에 셋팅하여, 5℃/분의 속도로 500℃까지 승온했다. 측정은 질소 분위기하에 행하고, 100℃∼300℃의 사이에 감소한 중량을 용매 함유량으로 했다. The solvent content in the polyimide film was measured using a thermogravimetric analyzer (manufactured by TA Instruments, model name TGA2950). About 10 mg of the sample was set in an aluminum microcell, and the temperature was raised to 500°C at a rate of 5°C/min. The measurement was performed in a nitrogen atmosphere, and the weight decreased between 100°C and 300°C was used as the solvent content.
<적층체의 복귀율> <Return rate of laminated body>
측정 장치로서 미소 경도 시험기 피셔 스코프 HM2000(피셔사 제조)을 이용하고, 적층체를 슬라이드 글래스에 순간 접착제 아론알파 221F(도아고세이사 제조)로 고정하여, 상기 측정 장치에 셋팅한다. 슬라이드 글래스에 고정된 적층체에 측정 온도 30℃에서 비커스 사각뿔 다이아몬드 압자로부터 0.5 mN까지 15초에 걸쳐 하중을 가하여 0.5 mNd로 5초간 유지한다. 그 때의 최대 변위를 (h1)로 한다. 그 후, 15초에 걸쳐 0.005 mN까지 하중을 제거하고, 0.005 mN로 60초간 유지했을 때의 변위를 (h2)로 하여, 복귀율〔{(h1-h2)/h1}×100(%)〕을 계산했다. Using a micro hardness tester Fischer Scope HM2000 (manufactured by Fisher Co., Ltd.) as a measuring device, the laminate was fixed to a slide glass with instant adhesive Aaron Alpha 221F (manufactured by Toagosei Co., Ltd.), and set in the measuring device. A load was applied over 15 seconds to 0.5 mN from a Vickers square pyramid diamond indenter at a measurement temperature of 30 DEG C to the laminated body fixed to the slide glass, and held at 0.5 mNd for 5 seconds. The maximum displacement at that time is (h1). Thereafter, the load was removed to 0.005 mN over 15 seconds, and the displacement when held at 0.005 mN for 60 seconds was (h2), and the return rate [{(h1-h2)/h1} × 100 (%)] Calculated.
<적층체의 유연성 평가> <Evaluation of the flexibility of the laminate>
JIS K 5600-5-1(1999)에 준거한 굴곡 시험법에 기초하여, 굴곡 시험기 타입 1(이모토 제작소사 제조, 모델 IMC-AOF2, 맨드릴 직경 φ20 mm)을 이용하여 1000회의 굴곡 시험을 행한 후, 적층체 샘플 표면을 육안으로 관찰하고, 하기의 기준에 따라서 유연성을 평가했다. Based on the bending test method conforming to JIS K 5600-5-1 (1999), a bending test of 1000 times was performed using a bending tester type 1 (manufactured by Imoto Manufacturing Co., Ltd., model IMC-AOF2, mandrel diameter φ20 mm) After that, the surface of the laminate sample was visually observed, and flexibility was evaluated according to the following criteria.
0 : 적층체 샘플 표면에 미소한 균열이 관찰되지 않는다 0: No minute cracks are observed on the surface of the laminate sample
× : 적층체 샘플 표면에 미소한 균열이 관찰된다×: Minor cracks are observed on the surface of the laminate sample
<적층체의 부착률> <Adhesion rate of laminated body>
JIS K 5600-5-6(1999)의 크로스커트법에 의해, 항온 항습 조건하(23℃, 50%RH)에, 적층체 샘플의 자기 수복층 상에, 크로스커트 박리 시험 지그를 이용하여 세로방향과 가로방향으로 각각 1 mm 간격의 직선형 커트를 11개 넣어, 1 평방 mm의 크로스커트를 100개 제작했다. 이 크로스커트 스퀘어 상에 세키스이 화학 공업 주식회사 제조의 점착 테이프 No.252를 그 위에 접착하고, 주걱을 이용하여 균일하게 누른 후에, 적층체에 대하여 점착 테이프를 90도 방향으로 박리한다. 부착률은 자기 수복층 크로스커트의 폴리이미드 필름 상의 잔존 갯수로서 구할 수 있다. 평가는 100개 중 잔존 갯수를 「%」로 구했다. By the crosscut method of JIS K 5600-5-6 (1999), under constant temperature and humidity conditions (23 ° C., 50% RH), on the self-healing layer of the laminate sample, a crosscut peel test jig was used to perform a longitudinal test. 100 crosscuts of 1 square mm were produced by inserting 11 straight cuts at intervals of 1 mm in the direction and transverse direction, respectively. On this crosscut square, adhesive tape No. 252 manufactured by Sekisui Chemical Industry Co., Ltd. is adhered thereon, and after pressing uniformly using a spatula, the adhesive tape is peeled in the direction of 90 degrees with respect to the layered product. The adhesion rate can be obtained as the remaining number of self-healing layer crosscuts on the polyimide film. For evaluation, the remaining number out of 100 was determined as "%".
<적층체의 황색도 지수(옐로우 인덱스, YI)> <Yellowness Index (Yellow Index, YI) of Laminates>
컬러 미터(ZE6000, 니폰덴쇼쿠사 제조) 및 C2 광원을 사용하여, ASTM D1925에 준하여 폴리이미드 필름의 삼자극값 XYZ값을 측정하여, 하기 식에 의해 황색도 지수(YI)를 산출했다. 한편, 동일한 측정을 3회 행하여 그 산술 평균치를 채용했다. Using a color meter (ZE6000, manufactured by Nippon Denshoku Co., Ltd.) and a C2 light source, the tristimulus value XYZ value of the polyimide film was measured according to ASTM D1925, and the yellowness index (YI) was calculated by the following formula. On the other hand, the same measurement was performed three times and the arithmetic average value was adopted.
YI=100×(1.28X-1.06Z)/Y YI=100×(1.28X-1.06Z)/Y
<적층체의 400 nm 광선 투과율> <400 nm light transmittance of the laminate>
분광 광도계(히타치 제작소 제조 「U-2001」)를 이용하여 파장 400 nm에서의 광선 투과율을 측정하고, 얻어진 값을 런벨트·베일의 법칙에 따르는 것으로서 20 μm의 두께로 환산하여, 얻어진 값을 폴리이미드 필름의 400 nm 광선 투과율로 했다. 또한, 동일한 측정을 3회 행하여 그 산술 평균치를 채용했다. The light transmittance at a wavelength of 400 nm was measured using a spectrophotometer ("U-2001" manufactured by Hitachi, Ltd.), and the obtained value was converted to a thickness of 20 µm as according to Runbelt-Vale's law, and the obtained value was The transmittance of mid-film was 400 nm. In addition, the same measurement was performed three times and the arithmetic average value was adopted.
<적층체의 전광선 투과율(TT)> <Total light transmittance (TT) of the laminate>
HAZEMETER(NDH5000, 니폰덴쇼쿠사 제조)를 이용하여 폴리이미드 필름의 전광선 투과율(TT)을 측정했다. 광원으로는 D65 램프를 사용했다. 또한, 동일한 측정을 3회 행하여 그 산술 평균치를 채용했다. The total light transmittance (TT) of the polyimide film was measured using HAZEMETER (NDH5000, manufactured by Nippon Denshoku Co., Ltd.). A D65 lamp was used as the light source. In addition, the same measurement was performed three times and the arithmetic average value was adopted.
〔폴리아미드산 용액 A의 조제〕[Preparation of polyamic acid solution A]
질소 도입관, 온도계, 교반 막대를 구비한 반응 용기 내를 질소 치환한 후, 반응 용기에 질소 분위기하, 176.5 g(0.900 mol)의 1,2,3,4-시클로부탄테트라카르복실산2무수물(CBDA), 31.0 g(0.100 mol)의 4,4'-옥시디프탈산(ODPA), 160.1 g(0.500 mol)의 2,2'-디트리플루오로메틸-4,4'-디아미노비페닐(TFMB), 113.6 g(0.500 mol)의 4-아미노-N-(4-아미노페닐)벤즈아미드(DABAN), 2000 g의 N,N-디메틸아세트아미드를 넣고 용해시킨 후, 실온에서 24시간 교반하여 중합 반응을 행했다. 그 후, 1000 g의 N,N-디메틸아세트아미드로 희석하여, 환원 점도 4.50 dl/g의 폴리아미드산 용액 A를 얻었다. After purging the inside of the reaction vessel equipped with a nitrogen inlet tube, thermometer, and stirring rod with nitrogen, 176.5 g (0.900 mol) of 1,2,3,4-cyclobutanetetracarboxylic dianhydride was added to the reaction vessel under a nitrogen atmosphere. (CBDA), 31.0 g (0.100 mol) of 4,4'-oxydiphthalic acid (ODPA), 160.1 g (0.500 mol) of 2,2'-ditrifluoromethyl-4,4'-diaminobiphenyl (TFMB), 113.6 g (0.500 mol) of 4-amino-N- (4-aminophenyl) benzamide (DABAN), and 2000 g of N, N-dimethylacetamide were added and dissolved, followed by stirring at room temperature for 24 hours A polymerization reaction was performed. Then, it was diluted with 1000 g of N,N-dimethylacetamide to obtain a polyamic acid solution A having a reduced viscosity of 4.50 dl/g.
〔폴리이미드 용액 B의 조제〕[Preparation of Polyimide Solution B]
질소 도입관, 온도계, 교반 막대를 구비한 반응 용기 내를 질소 치환한 후, 반응 용기에 질소 분위기하, 461 g의 N,N-디메틸아세트아미드(DMAC)와 64.0 g(0.200 mol)의 2,2'-디트리플루오로메틸-4,4'-디아미노비페닐(TFMB)를 넣어 교반하고, TFMB를 DMAC 중에 용해시켰다. 이어서, 반응 용기 내를 교반하면서, 질소 기류하에, 89.737 g(0.202 mol)의 4,4'-(2,2-헥사플루오로이소프로필리덴)디프탈산2무수물(6FDA)을 10분 정도에 걸쳐 투입하고, 그대로 온도가 20∼40℃의 온도 범위가 되도록 조정하면서 6시간 계속 교반하여 중합 반응을 행하여, 점조의 폴리아미드산 용액을 얻었다. After purging the inside of the reaction vessel equipped with a nitrogen inlet pipe, thermometer, and stirring rod with nitrogen, 461 g of N,N-dimethylacetamide (DMAC) and 64.0 g (0.200 mol) of 2, 2'-ditrifluoromethyl-4,4'-diaminobiphenyl (TFMB) was added and stirred, and TFMB was dissolved in DMAC. Then, 89.737 g (0.202 mol) of 4,4'-(2,2-hexafluoroisopropylidene)diphthalic dianhydride (6FDA) was added over 10 minutes under a nitrogen stream while stirring the inside of the reaction vessel. After adding, the polymerization reaction was carried out by continuously stirring for 6 hours while adjusting the temperature as it was to be in the temperature range of 20 to 40°C, and a viscous polyamic acid solution was obtained.
다음으로, 얻어진 폴리아미드산 용액에 410 g의 DMAC를 가하여 희석한 후, 이미드화 촉진제로서 25.83 g의 이소퀴놀린을 가하여, 폴리아미드산 용액을 교반하면서 30∼40℃의 온도 범위로 유지하고, 거기에 이미드화제로서 122.5 g(1.20 몰)의 무수아세트산을 약 10분에 걸쳐 천천히 적하하면서 투입하고, 그 후 액온을 30∼40℃로 유지하고 12시간 계속 교반하여 화학 이미드화 반응를 더 행하여, 폴리이미드 용액을 얻었다. Next, after diluting by adding 410 g of DMAC to the obtained polyamic acid solution, 25.83 g of isoquinoline was added as an imidation accelerator, and the polyamic acid solution was maintained at a temperature range of 30 to 40 ° C. while stirring, there As an imidation agent, 122.5 g (1.20 mol) of acetic anhydride was slowly added dropwise over about 10 minutes, and then the liquid temperature was maintained at 30 to 40° C. and stirred continuously for 12 hours to further carry out a chemical imidation reaction. Mead solution was obtained.
다음으로, 얻어진 이미드화제 및 이미드화 촉진제를 포함하는 폴리이미드 용액 1000 g을, 교반 장치와 교반 날개를 구비한 반응 용기로 옮기고, 120 rpm의 속도로 교반하면서 15∼25℃의 온도로 유지하고, 거기에 1500 g의 메탄올을 10 g/분의 속도로 적하시켰다. 약 800 g의 메탄올을 투입한 지점에서 폴리이미드 용액이 흐려진 것이 확인되고, 분체형의 폴리이미드의 석출이 확인되었다. 계속해서 1500 g 전량의 메탄올을 투입하고, 폴리이미드의 석출을 완료시켰다. 계속해서, 반응 용기의 내용물을 흡인 여과 장치에 의해 여과 분리하고, 또한 1000 g의 메탄올을 이용하여 세정·여과 분리했다. 그 후, 여과 분리한 폴리이미드 분체 50 g을 국소 배기 장치가 부착된 건조기를 이용하여, 50℃에서 24시간 건조시키고, 260℃에서 2시간 더 건조시켜, 남아 있는 휘발 성분을 제거하고, 폴리이미드 분체를 얻었다. 얻어진 폴리이미드 분체의 환원 점도는 5.40 dl/g였다. 다음으로, 얻어진 폴리이미드 분체 40 g을 300 g의 DMAC에 용해시켜, 폴리이미드 용액 B를 얻었다. Next, 1000 g of the obtained polyimide solution containing the imidation agent and the imidation accelerator was transferred to a reaction vessel equipped with a stirring device and a stirring blade, and maintained at a temperature of 15 to 25 ° C. while stirring at a speed of 120 rpm, , 1500 g of methanol was added dropwise thereto at a rate of 10 g/min. It was confirmed that the polyimide solution became cloudy at the point where about 800 g of methanol was injected, and precipitation of powdery polyimide was confirmed. Subsequently, 1500 g of methanol was injected to complete the precipitation of polyimide. Subsequently, the contents of the reaction vessel were separated by filtration with a suction filtration device, and further washed and filtered using 1000 g of methanol. Thereafter, 50 g of the polyimide powder separated by filtration was dried at 50° C. for 24 hours using a dryer equipped with a local exhaust system and further dried at 260° C. for 2 hours to remove remaining volatile components, and polyimide powder was obtained. The reduced viscosity of the obtained polyimide powder was 5.40 dl/g. Next, 40 g of the obtained polyimide powder was dissolved in 300 g of DMAC to obtain a polyimide solution B.
〔폴리이미드 용액 C의 조제〕[Preparation of polyimide solution C]
질소 도입관, 딘·스타크관 및 환류관, 온도계, 교반 막대를 구비한 반응 용기에 질소 가스를 도입하면서, 120.5 g(0.485 mol)의 4,4'-디아미노디페닐술폰(4,4'-DDS), 51.6 g(0.208 mol)의 3,3'-디아미노디페닐술폰(3,3'-DDS), 500 g의γ-부티로락톤(GBL)을 가했다. 계속해서 217.1 g(0.700 mol)의 4,4'-옥시디프탈산2무수물(ODPA), 223 g의 GBL, 260 g의 톨루엔을 실온에서 가한 후, 내온 160℃까지 승온하고, 160℃에서 1시간 가열 환류하여 이미드화를 행했다. 이미드화 완료후, 180℃까지 승온하고, 톨루엔을 추출하면서 반응을 계속했다. 12시간 중합 반응후, 오일배스를 제거하여 실온으로 되돌리고, 고형분이 20 질량% 농도가 되도록 GBL을 가하여 환원 점도 2.50 dl/g의 폴리이미드 용액 C를 얻었다. 120.5 g (0.485 mol) of 4,4'-diaminodiphenylsulfone (4,4' -DDS), 51.6 g (0.208 mol) of 3,3'-diaminodiphenylsulfone (3,3'-DDS), and 500 g of γ-butyrolactone (GBL) were added. Subsequently, after adding 217.1 g (0.700 mol) of 4,4'-oxydiphthalic dianhydride (ODPA), 223 g of GBL, and 260 g of toluene at room temperature, the internal temperature was raised to 160°C, and the temperature was raised to 160°C for 1 hour. Imidization was performed by heating to reflux. After completion of the imidation, the temperature was raised to 180°C, and the reaction was continued while extracting toluene. After the polymerization reaction for 12 hours, the oil bath was removed, the temperature was returned to room temperature, and GBL was added so that the solid content was 20% by mass to obtain a polyimide solution C having a reduced viscosity of 2.50 dl/g.
〔폴리아미드산 용액 G의 조제〕[Preparation of polyamic acid solution G]
질소 도입관, 온도계, 교반 막대를 구비한 반응 용기 내를 질소 치환한 후, 반응 용기에 질소 분위기하, 94.1 g(0.480 mol)의 1,2,3,4-시클로부탄테트라카르복실산2무수물(CBDA), 108.9 g(0.370 mol)의 3,3',4,4'-비페닐테트라카르복실산2무수물(BPDA), 46.5 g(0.150 mol)의 4,4'-옥시디프탈산(ODPA), 320.2 g(1.000 mol)의 2,2'-디트리플루오로메틸-4,4'-디아미노비페닐(TFMB), 2000 g의 N,N-디메틸아세트아미드를 넣고 용해시킨 후, 실온에서 24시간 교반하여 중합 반응을 행했다. 그 후, 1000 g의 N,N-디메틸아세트아미드로 희석하여, 환원 점도 3.50 dl/g의 폴리아미드산 용액 G을 얻었다. After purging the inside of the reaction vessel equipped with a nitrogen inlet tube, thermometer, and stirring rod with nitrogen, 94.1 g (0.480 mol) of 1,2,3,4-cyclobutanetetracarboxylic dianhydride was added to the reaction vessel under a nitrogen atmosphere. (CBDA), 108.9 g (0.370 mol) of 3,3',4,4'-biphenyltetracarboxylic dianhydride (BPDA), 46.5 g (0.150 mol) of 4,4'-oxydiphthalic acid (ODPA) ), 320.2 g (1.000 mol) of 2,2'-ditrifluoromethyl-4,4'-diaminobiphenyl (TFMB), and 2000 g of N, N-dimethylacetamide were added and dissolved, followed by dissolution at room temperature. was stirred for 24 hours to carry out a polymerization reaction. Then, it was diluted with 1000 g of N,N-dimethylacetamide to obtain a polyamic acid solution G having a reduced viscosity of 3.50 dl/g.
〔폴리이미드 필름의 제작예 1〕[Production Example 1 of Polyimide Film]
폴리아미드산 용액 A를, 다이코터를 이용하여, 필름 제작 지지체인 경면 마무리한 스테인레스스틸제의 무단 연속 벨트 상에 도포하고(도공폭 1240 mm), 90∼115℃에서 10분간 건조시켰다. 건조후에 자기 지지성이 된 폴리아미드산 필름(잔류 용매를 9 질량% 포함)을 지지체로부터 박리하고 양쪽 단부를 커트하여 그린 필름을 얻었다. 얻어진 그린 필름을 핀 텐터에 의해 최종 핀 시트 간격이 1140 mm가 되도록 반송하고, 1단계 170℃에서 2분간, 2단계 230℃에서 2분간, 3단계 350℃에서 6분간 열처리하고, 잔류 용매를 소정 값의 범위 내가 되도록 제거했다. 그 후, 2분간 실온까지 냉각시키고, 필름의 양쪽 단부의 평면성이 나쁜 부분을 슬리터로 잘라내고, 롤형으로 감아, 표 1에 나타내는 폴리이미드 필름 1A를 얻었다. 이하 동일하게 폴리아미드산 용액 A를 폴리이미드 용액 B 또는 폴리아미드산 용액 G로 바꾸고, 또한 지지체에 대한 도포 두께를 바꿔, 표 1에 나타내는 폴리이미드 필름 1B 또는 폴리이미드 필름 1G를 얻었다. Polyamic acid solution A was applied using a die coater onto a mirror-finished stainless steel endless continuous belt (coating width: 1240 mm) as a film production support, and dried at 90 to 115° C. for 10 minutes. After drying, the self-supporting polyamic acid film (containing 9% by mass of residual solvent) was peeled off from the support and both ends were cut to obtain a green film. The obtained green film was conveyed by a pin tenter so that the final pin sheet interval was 1140 mm, heat treatment was performed at 170 ° C. for 2 minutes in the first step, 230 ° C. for 2 minutes in the second step, and 350 ° C. for 6 minutes in the third step. It was removed to be within the range of values. Thereafter, it was cooled to room temperature for 2 minutes, and portions with poor flatness at both ends of the film were cut out with a slitter and wound into a roll shape to obtain a polyimide film 1A shown in Table 1. Polyimide film 1B or polyimide film 1G shown in Table 1 was obtained by changing the polyamic acid solution A to polyimide solution B or polyamic acid solution G in the same manner and changing the coating thickness to the support.
〔폴리이미드 필름의 제작예 2〕[Production Example 2 of Polyimide Film]
폴리아미드산 용액 A를, 필름 제작 지지체인 지점의 영역 표면 거칠기(Sa)가 1 nm, 최대 돌기 높이(Sp)가 7 nm, 산정점 밀도(Spd)가 20/평방 μm 이하이며, 표면에 코트층을 갖지 않는 폴리에스테르 필름에 콤마코터를 이용하여 도포하고(도공폭 1240 mm), 90∼115℃에서 10분간 건조시켰다. 건조후에 자기 지지성이 된 폴리아미드산 필름(잔류 용매를 10 질량% 포함)을 지지체로부터 박리하고 양쪽 단부를 커트하여 그린 필름을 얻었다. 얻어진 그린 필름을 핀 텐터에 의해 최종 핀 시트 간격이 1140 mm가 되도록 반송하고, 1단계 170℃에서 2분간, 2단계 230℃에서 2분간, 3단계 350℃에서 6분간 열처리하고, 잔류 용매를 소정 값의 범위 내가 되도록 제거했다. 그 후, 2분간 실온까지 냉각시키고, 필름의 양쪽 단부의 평면성이 나쁜 부분을 슬리터로 잘라내고, 롤형으로 감아, 표 1에 나타내는 폴리이미드 필름 2A를 얻었다. 이하 동일하게 폴리아미드산 용액 A를 폴리이미드 용액 C 또는 폴리아미드산 용액 G로 바꾸고, 또한 지지체에 대한 도포 두께를 바꿔, 표 1에 나타내는 폴리이미드 필름 2C 또는 폴리이미드 필름 2G를 얻었다. Polyamic acid solution A was coated on the surface with a surface roughness (Sa) of 1 nm, a maximum protrusion height (Sp) of 7 nm, a peak point density (Spd) of 20/square μm or less, and It was applied to a non-layered polyester film using a comma coater (coating width: 1240 mm), and dried at 90 to 115° C. for 10 minutes. After drying, the self-supporting polyamic acid film (containing 10% by mass of residual solvent) was peeled off from the support and both ends were cut to obtain a green film. The obtained green film was conveyed by a pin tenter so that the final pin sheet interval was 1140 mm, heat treatment was performed at 170 ° C. for 2 minutes in the first step, 230 ° C. for 2 minutes in the second step, and 350 ° C. for 6 minutes in the third step. It was removed to be within the range of values. Thereafter, it was cooled to room temperature for 2 minutes, and portions with poor flatness at both ends of the film were cut out with a slitter, and wound into a roll shape to obtain a polyimide film 2A shown in Table 1. Polyimide film 2C or polyimide film 2G shown in Table 1 was obtained by changing the polyamic acid solution A to polyimide solution C or polyamic acid solution G in the same manner and changing the coating thickness to the support.
〔폴리이미드 필름의 제작예 3〕[Production Example 3 of Polyimide Film]
폴리이미드 용액 B를, 필름 제작 지지체인 지점의 영역 표면 거칠기(Sa)가 3 nm, 최대 돌기 높이(Sp)가 12 nm, 산정점 밀도(Spd)가 25/평방 μm 이하인 미연신 폴리프로필렌 필름에 콤마코터를 이용하여 도포하고(도공폭 450 mm), 85∼105℃에서 30분간 건조시켜, 지지체와 폴리이미드 필름(잔류 용매 약 8 질량%을 포함)의 2층 필름을 얻었다. 이어서, 이 2층 필름을, 2층 동시에 롤의 주속도차를 이용하여 MD 방향으로 2.8배로 연신했다. 또, 주속도차가 있는 롤과 롤의 사이에서는 2층 필름의 폴리이미드 필름측의 면에 롤이 접촉하지 않도록 배치했다. MD 방향으로의 연신후에, 클립 텐터로 2층 필름의 양쪽 단부를 파지하고, 최종 핀 시트 간격이 1140 mm, 즉 TD 방향으로 2.5배 연신이 되도록 150℃에서 6분간의 열처리를 하면서 반송하고, 그 후에 2층 필름의 지지체로부터 폴리이미드 필름을 박리하고, 350℃에서 3분간의 열처리를 더 행하는 것에 의해 잔류 용매를 소정 값의 범위 내가 되도록 제거했다. 그 후, 2분간 실온까지 냉각시키고, 필름의 양쪽 단부의 평면성이 나쁜 부분을 슬리터로 잘라내고, 롤형으로 감아, 표 1에 나타내는 폴리이미드 필름 3B를 얻었다. 이하 동일하게 폴리이미드 용액 B를 폴리이미드 용액 C로 바꾸고, 또한 지지체에 대한 도포 두께를 바꿔, 표 1에 나타내는 폴리이미드 필름 3C를 얻었다. Polyimide solution B was applied to an unstretched polypropylene film having a surface roughness (Sa) of 3 nm, a maximum protrusion height (Sp) of 12 nm, and a peak point density (Spd) of 25/square μm or less at the points of the film production support. It was applied using a comma coater (coating width: 450 mm) and dried at 85 to 105° C. for 30 minutes to obtain a two-layer film of a support and a polyimide film (including about 8% by mass of residual solvent). Next, this two-layer film was stretched 2.8 times in the MD direction using the difference in circumferential speed of the rolls at the same time. Moreover, between the rolls with the circumferential speed difference, it arrange|positioned so that a roll might not contact the surface of the polyimide film side of a two-layer film. After stretching in the MD direction, both ends of the two-layer film are held by a clip tenter, and transported while heat treatment at 150 ° C. for 6 minutes so that the final pin sheet interval is 1140 mm, that is, stretched 2.5 times in the TD direction, After that, the polyimide film was peeled off from the support of the two-layer film, and a heat treatment at 350° C. for 3 minutes was further performed to remove the residual solvent so as to be within a predetermined range. Thereafter, it was cooled to room temperature for 2 minutes, and portions with poor flatness at both ends of the film were cut out with a slitter, and rolled up to obtain a polyimide film 3B shown in Table 1. In the same manner, polyimide solution B was replaced with polyimide solution C, and the coating thickness to the support was changed to obtain a polyimide film 3C shown in Table 1.
〔폴리이미드 필름의 제작예 4〕[Production Example 4 of Polyimide Film]
폴리아미드산 용액 A를, 다이코터를 이용하여, 필름 제작 지지체인 경면 마무리한 스테인레스스틸제의 무단 연속 벨트 상에 도포하고(도공폭 1240 mm), 90∼115℃에서 10분간 건조시켰다. 건조후에 자기 지지성이 된 폴리아미드산 필름(잔류 용매를 9 질량% 포함)을 지지체로부터 박리하고 양쪽 단부를 커트하여 그린 필름을 얻었다. 얻어진 그린 필름을 핀 텐터에 의해 최종 핀 시트 간격이 1140 mm가 되도록 반송하고, 1단계 170℃에서 2분간, 2단계 230℃에서 2분간, 3단계 350℃에서 10분간 열처리하고, 잔류 용매를 소정 값의 범위 내가 되도록 제거했다. 그 후, 2분간 실온까지 냉각시키고, 필름의 양쪽 단부의 평면성이 나쁜 부분을 슬리터로 잘라내고, 롤형으로 감아, 표 1에 나타내는 폴리이미드 필름 4A를 얻었다. Polyamic acid solution A was applied using a die coater onto a mirror-finished stainless steel endless continuous belt (coating width: 1240 mm) as a film production support, and dried at 90 to 115° C. for 10 minutes. After drying, the self-supporting polyamic acid film (containing 9% by mass of residual solvent) was peeled off from the support and both ends were cut to obtain a green film. The obtained green film was conveyed by a pin tenter so that the final pin sheet interval was 1140 mm, heat treatment was performed at 170 ° C. for 2 minutes in the first step, 230 ° C. for 2 minutes in the second step, and 350 ° C. for 10 minutes in the third step. It was removed to be within the range of values. Thereafter, it was cooled to room temperature for 2 minutes, and portions with poor flatness at both ends of the film were cut out with a slitter, and wound into a roll shape to obtain a polyimide film 4A shown in Table 1.
〔폴리이미드 필름의 제작예 5〕[Production Example 5 of Polyimide Film]
폴리아미드산 용액 A를, 필름 제작 지지체인 지점의 영역 표면 거칠기(Sa)가 1 nm, 최대 돌기 높이(Sp)가 7 nm, 산정점 밀도(Spd)가 20/평방 μm 이하이며, 표면에 코트층을 갖지 않는 폴리에스테르 필름에 콤마코터를 이용하여 도포하고(도공폭 1240 mm), 90∼115℃에서 10분간 건조시켰다. 건조후에 자기 지지성이 된 폴리아미드산 필름(잔류 용매를 10 질량% 포함)을 지지체로부터 박리하고 양쪽 단부를 커트하여 그린 필름을 얻었다. 얻어진 그린 필름을 핀 텐터에 의해 최종 핀 시트 간격이 1140 mm가 되도록 반송하고, 1단계 170℃에서 2분간, 2단계 230℃에서 2분간, 3단계 350℃에서 10분간 열처리하고, 잔류 용매를 소정 값의 범위 내가 되도록 제거했다. 그 후, 2분간 실온까지 냉각시키고, 필름의 양쪽 단부의 평면성이 나쁜 부분을 슬리터로 잘라내고, 롤형으로 감아, 표 1에 나타내는 폴리이미드 필름 5A를 얻었다. Polyamic acid solution A was coated on the surface with a surface roughness (Sa) of 1 nm, a maximum protrusion height (Sp) of 7 nm, a peak point density (Spd) of 20/square μm or less, and It was applied to a non-layered polyester film using a comma coater (coating width: 1240 mm), and dried at 90 to 115° C. for 10 minutes. After drying, the self-supporting polyamic acid film (containing 10% by mass of residual solvent) was peeled off from the support and both ends were cut to obtain a green film. The obtained green film was conveyed by a pin tenter so that the final pin sheet interval was 1140 mm, heat treatment was performed at 170 ° C. for 2 minutes in the first step, 230 ° C. for 2 minutes in the second step, and 350 ° C. for 10 minutes in the third step. It was removed to be within the range of values. Thereafter, it was cooled to room temperature for 2 minutes, and portions with poor flatness at both ends of the film were cut out with a slitter, and wound into a roll shape to obtain a polyimide film 5A shown in Table 1.
〔폴리이미드 필름의 제작예 6〕[Production Example 6 of Polyimide Film]
폴리이미드 용액 C를, 필름 제작 지지체인 지점의 영역 표면 거칠기(Sa)가 1 nm, 최대 돌기 높이(Sp)가 7 nm, 산정점 밀도(Spd)가 20/평방 μm 이하이며, 표면에 코트층을 갖지 않는 폴리에스테르 필름에 콤마코터를 이용하여 도포하고(도공폭 1240 mm), 90∼115℃에서 10분간 건조시켰다. 건조후에 자기 지지성이 된 폴리아미드산 필름(잔류 용매를 10 질량% 포함)을 지지체로부터 박리하고 양쪽 단부를 커트하여 그린 필름을 얻었다. 얻어진 그린 필름을 핀 텐터에 의해 최종 핀 시트 간격이 1140 mm가 되도록 반송하고, 1단계 170℃에서 2분간, 2단계 230℃에서 2분간, 3단계 350℃에서 1분간 열처리하고, 잔류 용매를 소정 값의 범위 내가 되도록 제거했다. 그 후, 2분간 실온까지 냉각시키고, 필름의 양쪽 단부의 평면성이 나쁜 부분을 슬리터로 잘라내고, 롤형으로 감아, 표 1에 나타내는 폴리이미드 필름 6C를 얻었다. Polyimide solution C has a surface roughness (Sa) of 1 nm, a maximum protrusion height (Sp) of 7 nm, a peak point density (Spd) of 20/square μm or less, and a coating layer on the surface of the point, which is a film production support. It was applied using a comma coater to a polyester film without (coating width: 1240 mm), and dried at 90 to 115° C. for 10 minutes. After drying, the self-supporting polyamic acid film (containing 10% by mass of residual solvent) was peeled off from the support and both ends were cut to obtain a green film. The obtained green film was conveyed by a pin tenter so that the final pin sheet interval was 1140 mm, heat treatment was performed at 170 ° C. for 2 minutes in the first step, 230 ° C. for 2 minutes in the second step, and 350 ° C. for 1 minute in the third step. It was removed to be within the range of values. Thereafter, it was cooled to room temperature for 2 minutes, and portions with poor flatness at both ends of the film were cut out with a slitter and wound into a roll to obtain a polyimide film 6C shown in Table 1.
〔폴리이미드 필름의 제작예 7〕[Production Example 7 of Polyimide Film]
폴리이미드 용액 B를, 필름 제작 지지체인 지점의 영역 표면 거칠기(Sa)가 3 nm, 최대 돌기 높이(Sp)가 12 nm, 산정점 밀도(Spd)가 25/평방 μm 이하인 미연신 폴리프로필렌 필름에 콤마코터를 이용하여 도포하고(도공폭 450 mm), 85∼105℃에서 30분간 건조시켜, 지지체와 폴리이미드 필름(잔류 용매 약 8 질량%을 포함)의 2층 필름을 얻었다. 이어서, 이 2층 필름을, 2층 동시에 롤의 주속도차를 이용하여 MD 방향으로 2.8배로 연신했다. 또, 주속도차가 있는 롤과 롤의 사이에서는 2층 필름의 폴리이미드 필름측의 면에 롤이 접촉하지 않도록 배치했다. MD 방향으로의 연신후에, 클립 텐터로 2층 필름의 양쪽 단부를 파지하고, 최종 핀 시트 간격이 1140 mm, 즉 TD 방향으로 2.5배 연신이 되도록 150℃에서 열처리하면서 반송하고, 그 후에 2층 필름의 지지체로부터 폴리이미드 필름을 박리하고, 350℃에서 1분간의 열처리를 더 행하는 것에 의해 잔류 용매를 소정 값의 범위 내가 되도록 제거했다. 그 후, 2분간 실온까지 냉각시키고, 필름의 양쪽 단부의 평면성이 나쁜 부분을 슬리터로 잘라내고, 롤형으로 감아, 표 1에 나타내는 폴리이미드 필름 7B를 얻었다. Polyimide solution B was applied to an unstretched polypropylene film having a surface roughness (Sa) of 3 nm, a maximum protrusion height (Sp) of 12 nm, and a peak point density (Spd) of 25/square μm or less at the points of the film production support. It was applied using a comma coater (coating width: 450 mm) and dried at 85 to 105° C. for 30 minutes to obtain a two-layer film of a support and a polyimide film (including about 8% by mass of residual solvent). Next, this two-layer film was stretched 2.8 times in the MD direction using the difference in circumferential speed of the rolls at the same time. Moreover, between the rolls with the circumferential speed difference, it arrange|positioned so that a roll might not contact the surface of the polyimide film side of a two-layer film. After stretching in the MD direction, both ends of the two-layer film are held by a clip tenter, and the final pin sheet interval is 1140 mm, that is, 2.5 times in the TD direction. The polyimide film was peeled from the support and further subjected to heat treatment at 350°C for 1 minute to remove residual solvent so as to be within a predetermined range. Thereafter, it was cooled to room temperature for 2 minutes, and portions with poor flatness at both ends of the film were cut out with a slitter and wound into a roll to obtain a polyimide film 7B shown in Table 1.
〔자기 수복층 형성용 조성물 D의 조제〕[Preparation of composition D for forming self-healing layer]
공유 결합 이외의 화학 결합에 의해 형성된 가교점을 갖는 중합체 조성물로서 폴리우레탄아크릴레이트 공중합체를 형성하는 조성물을 하기와 같이 계량하고, 실온에서 혼합 조제했다. A composition for forming a polyurethane acrylate copolymer as a polymer composition having a crosslinking point formed by a chemical bond other than a covalent bond was weighed as follows, and mixed and prepared at room temperature.
AUP-787(광중합 개시제 함유 우레탄아크릴레이트, (주)토쿠시키 제조) : 100 질량부AUP-787 (urethane acrylate containing photopolymerization initiator, manufactured by Tokushiki Co., Ltd.): 100 parts by mass
메틸에틸케톤 : 50 질량부Methyl ethyl ketone: 50 parts by mass
프로필렌글리콜모노메틸에테르 : 30 질량부 Propylene glycol monomethyl ether: 30 parts by mass
BYK-381(계면 활성제, 빅케미·재팬사 제조) : 1 질량부BYK-381 (surfactant, manufactured by Big Chemie Japan): 1 part by mass
〔자기 수복층 형성용 조성물 E의 조제〕[Preparation of composition E for forming a self-healing layer]
화학 결합에 의하지 않은 기하형상에 의한 구속으로 이루어진 가교점을 갖는 중합체 조성물로서 시클로덱스트린 고리 관통 폴리에테르 중합체를 형성하는 조성물을 하기와 같이 계량하고, 실온에서 혼합 조제했다. A composition for forming a cyclodextrin ring penetrating polyether polymer as a polymer composition having a crosslinking point composed of constraints by geometric shapes not by chemical bonds was weighed as follows, and mixed and prepared at room temperature.
SM3405P(수식 폴리로탁산, 어드밴스트 소프트 마테리알사 제조) : 50 질량부 SM3405P (modified polyrotaxane, manufactured by Advanced Soft Material Co., Ltd.): 50 parts by mass
M-309(트리메틸올프로판트리아크릴레이트, 도아고세이 제조) : 35 질량부 M-309 (trimethylolpropane triacrylate, manufactured by Toagosei): 35 parts by mass
M284(폴리에틸렌글리콜디아크릴레이트, 도요케미컬즈 제조) : 15 질량부M284 (polyethylene glycol diacrylate, manufactured by Toyo Chemicals): 15 parts by mass
메틸에틸케톤 : 40 질량부Methyl ethyl ketone: 40 parts by mass
프로필렌글리콜모노메틸에테르 : 10 질량부 Propylene glycol monomethyl ether: 10 parts by mass
Omnirad184(광중합 개시제, IGMResins 제조) : 4 질량부Omnirad184 (photopolymerization initiator, manufactured by IGMResins): 4 parts by mass
〔하드코트층 형성용 조성물 F의 조제〕 [Preparation of Composition F for Forming Hard Coat Layer]
하드코트층을 형성하는 중합체 조성물로서 (메트)아크릴레이트 공중합체를 형성하는 조성물을 하기와 같이 계량하고, 실온에서 혼합 조제했다. A composition for forming a (meth)acrylate copolymer as a polymer composition for forming a hard coat layer was weighed as follows, and mixed and prepared at room temperature.
OPSTAR Z7530(유기 수식 실리카 미립자와 다작용 아크릴레이트의 혼합물, 아라카와 화학 공업(주)사 제조) : 100 질량부OPSTAR Z7530 (a mixture of organically modified silica fine particles and polyfunctional acrylate, manufactured by Arakawa Chemical Industry Co., Ltd.): 100 parts by mass
펜타에리트리톨트리아크릴레이트 : 34 질량부Pentaerythritol triacrylate: 34 parts by mass
1-히드록시시클로헥실페닐케톤 : 1.8 질량부 1-hydroxycyclohexylphenyl ketone: 1.8 parts by mass
BYK-300(레벨링제, 빅케미·재팬사 제조) : 0.1 질량부BYK-300 (leveling agent, manufactured by Big Chemie Japan): 0.1 part by mass
프로필렌글리콜모노메틸에테르 : 80 질량부Propylene glycol monomethyl ether: 80 parts by mass
실시예 1Example 1
<적층체의 제작> <Manufacture of laminated body>
제작예 1에서 얻은 폴리이미드 필름 1A 전면에, 자기 수복층 형성 조성물 D를 롤코터에 의해 도포했다. 이어서, 80℃에서 건조시킨 후, 산소 농도가 1.0 체적% 이하의 분위기가 되도록 질소 퍼지하면서, 자외선 램프를 이용하여 조사부의 조도가 100 mW/평방 cm이고, 조사량을 0.3 J/평방 cm로 하여 도포층을 경화시켜, 적층체 두께 22.0 μm, 드라이층 두께 9.5 μm의 자기 수복층이 형성된 적층체를 작성했다. The self-healing layer-forming composition D was applied to the entire surface of the polyimide film 1A obtained in Production Example 1 by a roll coater. Subsequently, after drying at 80 ° C., while purging with nitrogen so that the oxygen concentration is 1.0% by volume or less, the irradiation area is 100 mW / square cm, and the irradiation amount is 0.3 J / square cm using an ultraviolet lamp. The layer was cured to create a laminate with a self-healing layer having a laminate thickness of 22.0 µm and a dry layer thickness of 9.5 µm.
실시예 2∼13 Examples 2 to 13
이하 동일하게 표 1에 나타내는 폴리이미드 필름과 자기 수복층 형성 조성물 D 및 E를 이용하여 적층체를 제작하여, 적층체의 특성을 평가했다. 결과를 표 2에 나타낸다. In the same manner, a laminate was produced using the polyimide film and the self-healing layer-forming compositions D and E shown in Table 1, and the characteristics of the laminate were evaluated. The results are shown in Table 2.
비교예 1∼8 Comparative Examples 1 to 8
이하 동일하게 표 1에 나타내는 폴리이미드 필름과 자기 수복층 형성 조성물 D, E 및 F를 이용하여 적층체를 제작하고, 적층체의 특성을 평가했다. 결과를 표 3에 나타낸다. Similarly, laminates were prepared using the polyimide films and the self-healing layer-forming compositions D, E, and F shown in Table 1, and the characteristics of the laminates were evaluated. The results are shown in Table 3.
이상 설명한 바와 같이, 본 발명의 적층체는, 우수한 투명성, 자기 수복성, 유연성을 나타내고, 자기 수복층과의 밀착성이 우수하기 때문에, 화상 표시 부분이 절첩 가능한 터치패널이나 디스플레이 등의 화상 표시 장치의 전면판, 전극 주변에 매우 유용하다. As described above, the laminate of the present invention exhibits excellent transparency, self-healing properties, and flexibility, and has excellent adhesion to the self-healing layer, so that the image display portion can be folded. Very useful around the front plate and electrodes.
Claims (5)
미소 경도 시험기로 비커스 사각뿔 다이아몬드 압자로부터 미소한 하중 0.5 mN을 표면에 부여하여 5초간 유지한 후에, 0.005 mN까지 하중을 제거하여 60초간 유지한 후의 복귀율이 80% 이상인 것을 특징으로 하는 적층체. According to claim 1,
After applying a minute load of 0.5 mN to the surface from a Vickers square pyramid diamond indenter with a micro hardness tester and holding it for 5 seconds, the return rate after removing the load to 0.005 mN and holding it for 60 seconds is 80% or more Laminate, characterized in that.
JIS K 5600-5-6(1999)의 크로스커트법에 의한, 격자형으로 절단된 상기 자기 수복층의 상기 폴리이미드 필름에 대한 부착률이 80% 이상인 것을 특징으로 하는 적층체. According to claim 1 or 2,
A laminate characterized in that the adhesion rate of the self-healing layer cut in a lattice shape to the polyimide film by the crosscut method of JIS K 5600-5-6 (1999) is 80% or more.
옐로우 인덱스가 10 이하, 파장 400 nm에서의 광선 투과율이 70% 이상, 전광선 투과율이 85% 이상인 것을 특징으로 하는 적층체. According to any one of claims 1 to 3,
A laminate characterized by having a yellow index of 10 or less, a light transmittance of 70% or more at a wavelength of 400 nm, and a total light transmittance of 85% or more.
상기 자기 수복층이 가교점을 포함하는 중합체 조성물로서, 상기 가교점의 일부 또는 전부가 가동성을 갖는 가교점인 것을 특징으로 하는 적층체. According to any one of claims 1 to 4,
A laminate characterized in that the self-healing layer is a polymer composition containing crosslinking points, and some or all of the crosslinking points are crosslinking points having mobility.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020050726 | 2020-03-23 | ||
| JPJP-P-2020-050726 | 2020-03-23 | ||
| PCT/JP2021/006845 WO2021192789A1 (en) | 2020-03-23 | 2021-02-24 | Laminate |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20220156808A true KR20220156808A (en) | 2022-11-28 |
Family
ID=77891622
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020227028038A KR20220156808A (en) | 2020-03-23 | 2021-02-24 | laminate |
Country Status (4)
| Country | Link |
|---|---|
| JP (1) | JPWO2021192789A1 (en) |
| KR (1) | KR20220156808A (en) |
| CN (1) | CN115175816B (en) |
| WO (1) | WO2021192789A1 (en) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013037323A (en) | 2011-08-11 | 2013-02-21 | Lintec Corp | Hard coat film |
| JP2016516846A (en) | 2013-03-15 | 2016-06-09 | エルジー・ケム・リミテッド | Plastic film |
| KR20160147416A (en) | 2015-06-15 | 2016-12-23 | 김동하 | Cooking device for controling water level automatically |
| KR20190051644A (en) | 2017-11-07 | 2019-05-15 | 주식회사 일진글로벌 | Sealing device for wheel bearing and wheel bearing assembly comprising the same |
| KR20190105829A (en) | 2018-03-06 | 2019-09-18 | 주식회사 엘지화학 | Method for predicting crack of the plate glass |
| JP2019211778A (en) | 2018-06-01 | 2019-12-12 | 三星電子株式会社Samsung Electronics Co.,Ltd. | Electronic device containing fordable display |
Family Cites Families (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3178279B2 (en) * | 1994-11-18 | 2001-06-18 | 宇部興産株式会社 | Polyimide film with adhesive and laminate |
| JP2003283077A (en) * | 2002-03-20 | 2003-10-03 | Teijin Ltd | Metal wiring circuit board |
| EP1867675A4 (en) * | 2005-03-28 | 2010-06-09 | Teijin Ltd | Aromatic polyimide film and method for producing same |
| JP2007076231A (en) * | 2005-09-15 | 2007-03-29 | Toyobo Co Ltd | Laminated polyimide film |
| JP2007106838A (en) * | 2005-10-12 | 2007-04-26 | Toyobo Co Ltd | Polyimide film |
| JP2007106837A (en) * | 2005-10-12 | 2007-04-26 | Toyobo Co Ltd | Polyimide film |
| JP2008044230A (en) * | 2006-08-16 | 2008-02-28 | Toyobo Co Ltd | Multilayered polyimide film and its forming method |
| JP2008094015A (en) * | 2006-10-13 | 2008-04-24 | Toyobo Co Ltd | Polyimide molded body, and its manufacturing method |
| US8222365B2 (en) * | 2006-12-12 | 2012-07-17 | Toyo Boseki Kabushiki Kaisha | Polyamideimide resin, as well as a colorless and transparent flexible metal-clad laminate and circuit board obtained therefrom |
| JP2008229866A (en) * | 2007-03-16 | 2008-10-02 | Toyobo Co Ltd | Adhesive film and semiconductor device |
| JP5224011B2 (en) * | 2011-04-15 | 2013-07-03 | 東洋紡株式会社 | LAMINATE, MANUFACTURING METHOD THEREOF, AND DEVICE STRUCTURE MANUFACTURING METHOD USING THE SAME |
| JP5894811B2 (en) * | 2012-02-03 | 2016-03-30 | 東レ・デュポン株式会社 | Polyimide sheet and method for producing the same |
| KR102034762B1 (en) * | 2012-06-20 | 2019-10-21 | 도요보 가부시키가이샤 | Process for producing layered product, layered product, process for producing layered product with device using said layered product, and layered product with device |
| KR101473968B1 (en) * | 2012-08-14 | 2014-12-18 | 국립대학법인 울산과학기술대학교 산학협력단 | Negative electrode active material for rechargeable lithium battery, method for preparing the same, and rechargeable lithium battery including the same |
| JP5931672B2 (en) * | 2012-09-24 | 2016-06-08 | 新日鉄住金化学株式会社 | Polyimide laminate and method for producing the same |
| JP5750424B2 (en) * | 2012-11-30 | 2015-07-22 | 株式会社カネカ | Isotropic adhesive film, method for producing the same, and flexible metal laminate using the adhesive film |
| JP6257302B2 (en) * | 2012-12-20 | 2018-01-10 | 旭化成株式会社 | POLYIMIDE PRECURSOR, RESIN COMPOSITION CONTAINING THE SAME, POLYIMIDE FILM AND ITS MANUFACTURING METHOD, AND LAMINATE AND ITS MANUFACTURING METHOD |
| JP5650347B1 (en) * | 2013-07-18 | 2015-01-07 | 日本合成化学工業株式会社 | Resin molded body, protective plate for display and touch panel substrate, and self-repairing method of resin molded body |
| KR102151077B1 (en) * | 2014-01-27 | 2020-09-02 | 에스케이이노베이션 주식회사 | Structure of Polyimide Composite Film and Fabrication Method |
| KR102305462B1 (en) * | 2015-04-30 | 2021-09-27 | 삼성디스플레이 주식회사 | Flexible window substrate and flexible display device having the same |
| JP6910133B2 (en) * | 2016-11-29 | 2021-07-28 | 旭化成株式会社 | Polyimide film laminate |
| KR20180091265A (en) * | 2017-02-06 | 2018-08-16 | 삼성전자주식회사 | Anti-fingerpringing composition with self-healing property, film, laminate and device |
| JP6912287B2 (en) * | 2017-06-27 | 2021-08-04 | 旭化成株式会社 | Polyimide film and its manufacturing method |
| JP2019137864A (en) * | 2018-02-13 | 2019-08-22 | 大日本印刷株式会社 | Polyimide film, laminate, display member, touch panel member, liquid crystal display device and organic electroluminescence display device |
| JP7099019B2 (en) * | 2018-04-09 | 2022-07-12 | 大日本印刷株式会社 | Method for manufacturing polyimide laminate and method for manufacturing polyimide film |
| JP2019210422A (en) * | 2018-06-07 | 2019-12-12 | 旭化成株式会社 | Polymer and varnish |
-
2021
- 2021-02-24 KR KR1020227028038A patent/KR20220156808A/en active Search and Examination
- 2021-02-24 JP JP2021559197A patent/JPWO2021192789A1/ja active Pending
- 2021-02-24 WO PCT/JP2021/006845 patent/WO2021192789A1/en active Application Filing
- 2021-02-24 CN CN202180017406.9A patent/CN115175816B/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013037323A (en) | 2011-08-11 | 2013-02-21 | Lintec Corp | Hard coat film |
| JP2016516846A (en) | 2013-03-15 | 2016-06-09 | エルジー・ケム・リミテッド | Plastic film |
| KR20160147416A (en) | 2015-06-15 | 2016-12-23 | 김동하 | Cooking device for controling water level automatically |
| KR20190051644A (en) | 2017-11-07 | 2019-05-15 | 주식회사 일진글로벌 | Sealing device for wheel bearing and wheel bearing assembly comprising the same |
| KR20190105829A (en) | 2018-03-06 | 2019-09-18 | 주식회사 엘지화학 | Method for predicting crack of the plate glass |
| JP2019211778A (en) | 2018-06-01 | 2019-12-12 | 三星電子株式会社Samsung Electronics Co.,Ltd. | Electronic device containing fordable display |
Also Published As
| Publication number | Publication date |
|---|---|
| CN115175816A (en) | 2022-10-11 |
| WO2021192789A1 (en) | 2021-09-30 |
| CN115175816B (en) | 2024-03-08 |
| JPWO2021192789A1 (en) | 2021-09-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN108699270B (en) | Polyimide film, method for producing polyimide film, and polyimide precursor resin composition | |
| WO2019026806A1 (en) | Polyimide varnish and method for producing same | |
| WO2022102451A1 (en) | Polyimide film and production method therefor | |
| JP7268791B2 (en) | laminate | |
| JP2023139144A (en) | Resin film and method for manufacturing resin film | |
| JP7287536B2 (en) | Polyimide film and its manufacturing method | |
| WO2022004852A1 (en) | Resin film and production method thereof | |
| KR20220156808A (en) | laminate | |
| JPH08225645A (en) | Colorless clear polyimide molding and production thereof | |
| JP7103534B2 (en) | Polyimide film and its manufacturing method | |
| WO2021241574A1 (en) | Laminate including transparent film with high heat resistance | |
| WO2022018994A1 (en) | Multilayer body and method for producing flexible device | |
| WO2023157839A1 (en) | Laminate | |
| WO2022102449A1 (en) | Polyimide film and production method therefor | |
| WO2021256298A1 (en) | Colorless multilayer polyimide film, multilayer body and method for producing flexible electronic device | |
| WO2022102450A1 (en) | Colorless multilayer polyimide film, laminate body, and flexible electronic device manufacturing method | |
| KR20230151517A (en) | A laminate of an inorganic substrate/polymer film layer with a protective film, a stack of the laminate, a storage method for the laminate, and a method for transporting the laminate. | |
| KR20240007114A (en) | Manufacturing method of resin film and film before cutting | |
| WO2023243693A1 (en) | Laminate | |
| JP2023001599A (en) | laminate |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination |