KR20160078986A - 대상체의 기도로 효소를 투여하기 위한 조성물 및 방법 - Google Patents
대상체의 기도로 효소를 투여하기 위한 조성물 및 방법 Download PDFInfo
- Publication number
- KR20160078986A KR20160078986A KR1020167012122A KR20167012122A KR20160078986A KR 20160078986 A KR20160078986 A KR 20160078986A KR 1020167012122 A KR1020167012122 A KR 1020167012122A KR 20167012122 A KR20167012122 A KR 20167012122A KR 20160078986 A KR20160078986 A KR 20160078986A
- Authority
- KR
- South Korea
- Prior art keywords
- composition
- plasminogen activator
- solution
- subject
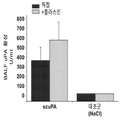
- scupa
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 80
- 238000000034 method Methods 0.000 title claims abstract description 74
- 102000004190 Enzymes Human genes 0.000 title claims abstract description 69
- 108090000790 Enzymes Proteins 0.000 title claims abstract description 69
- TXEYQDLBPFQVAA-UHFFFAOYSA-N tetrafluoromethane Chemical compound FC(F)(F)F TXEYQDLBPFQVAA-UHFFFAOYSA-N 0.000 claims abstract description 52
- 102000001938 Plasminogen Activators Human genes 0.000 claims abstract description 51
- 108010001014 Plasminogen Activators Proteins 0.000 claims abstract description 51
- 229940127126 plasminogen activator Drugs 0.000 claims abstract description 51
- 206010069351 acute lung injury Diseases 0.000 claims abstract description 34
- 239000000779 smoke Substances 0.000 claims abstract description 17
- 230000002685 pulmonary effect Effects 0.000 claims abstract description 5
- 208000015181 infectious disease Diseases 0.000 claims abstract 2
- 239000000243 solution Substances 0.000 claims description 69
- 229940088598 enzyme Drugs 0.000 claims description 65
- 108090000373 Tissue Plasminogen Activator Proteins 0.000 claims description 62
- 102000003978 Tissue Plasminogen Activator Human genes 0.000 claims description 62
- 229960000187 tissue plasminogen activator Drugs 0.000 claims description 62
- 108090000435 Urokinase-type plasminogen activator Proteins 0.000 claims description 31
- 238000000889 atomisation Methods 0.000 claims description 20
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 claims description 15
- 239000002953 phosphate buffered saline Substances 0.000 claims description 15
- 102000003990 Urokinase-type plasminogen activator Human genes 0.000 claims description 14
- 229950011087 perflunafene Drugs 0.000 claims description 12
- UWEYRJFJVCLAGH-IJWZVTFUSA-N perfluorodecalin Chemical compound FC1(F)C(F)(F)C(F)(F)C(F)(F)[C@@]2(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)[C@@]21F UWEYRJFJVCLAGH-IJWZVTFUSA-N 0.000 claims description 12
- 208000004852 Lung Injury Diseases 0.000 claims description 9
- 206010069363 Traumatic lung injury Diseases 0.000 claims description 9
- 231100000515 lung injury Toxicity 0.000 claims description 9
- WTWWXOGTJWMJHI-UHFFFAOYSA-N perflubron Chemical compound FC(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)Br WTWWXOGTJWMJHI-UHFFFAOYSA-N 0.000 claims description 9
- 229960001217 perflubron Drugs 0.000 claims description 9
- 206010001052 Acute respiratory distress syndrome Diseases 0.000 claims description 8
- 208000032376 Lung infection Diseases 0.000 claims description 7
- 150000003839 salts Chemical class 0.000 claims description 7
- 239000007864 aqueous solution Substances 0.000 claims description 5
- 239000002245 particle Substances 0.000 claims description 5
- 206010051011 Fibrinous bronchitis Diseases 0.000 claims description 4
- 208000006673 asthma Diseases 0.000 claims description 4
- 208000018916 plastic bronchitis Diseases 0.000 claims description 4
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 3
- 239000006174 pH buffer Substances 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 238000005507 spraying Methods 0.000 claims description 2
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 claims 4
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 claims 4
- 206010006451 bronchitis Diseases 0.000 claims 1
- 229920003023 plastic Polymers 0.000 claims 1
- 239000004033 plastic Substances 0.000 claims 1
- 230000000694 effects Effects 0.000 description 22
- 102100031358 Urokinase-type plasminogen activator Human genes 0.000 description 18
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 17
- 108090000765 processed proteins & peptides Proteins 0.000 description 17
- 210000004072 lung Anatomy 0.000 description 14
- 102000009123 Fibrin Human genes 0.000 description 13
- 108010073385 Fibrin Proteins 0.000 description 13
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 13
- 229950003499 fibrin Drugs 0.000 description 13
- 102000004196 processed proteins & peptides Human genes 0.000 description 12
- 201000010099 disease Diseases 0.000 description 11
- 229920001184 polypeptide Polymers 0.000 description 11
- 108010088842 Fibrinolysin Proteins 0.000 description 10
- 241001465754 Metazoa Species 0.000 description 10
- 229920006926 PFC Polymers 0.000 description 10
- 239000007788 liquid Substances 0.000 description 9
- 208000000884 Airway Obstruction Diseases 0.000 description 8
- 229940012957 plasmin Drugs 0.000 description 8
- 239000000725 suspension Substances 0.000 description 8
- 206010057190 Respiratory tract infections Diseases 0.000 description 7
- 102000012479 Serine Proteases Human genes 0.000 description 7
- 108010022999 Serine Proteases Proteins 0.000 description 7
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 7
- 239000000443 aerosol Substances 0.000 description 7
- 150000001413 amino acids Chemical class 0.000 description 7
- 150000001875 compounds Chemical class 0.000 description 7
- 102000013566 Plasminogen Human genes 0.000 description 6
- 108010051456 Plasminogen Proteins 0.000 description 6
- 208000035475 disorder Diseases 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 239000011780 sodium chloride Substances 0.000 description 6
- 230000001225 therapeutic effect Effects 0.000 description 6
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 5
- 241000282412 Homo Species 0.000 description 5
- 241000124008 Mammalia Species 0.000 description 5
- 102100038567 Properdin Human genes 0.000 description 5
- 230000008021 deposition Effects 0.000 description 5
- 239000003814 drug Substances 0.000 description 5
- 229960002897 heparin Drugs 0.000 description 5
- 229920000669 heparin Polymers 0.000 description 5
- 230000007774 longterm Effects 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- 230000000241 respiratory effect Effects 0.000 description 5
- 208000024891 symptom Diseases 0.000 description 5
- 210000001519 tissue Anatomy 0.000 description 5
- 108050006955 Tissue-type plasminogen activator Proteins 0.000 description 4
- 102100033571 Tissue-type plasminogen activator Human genes 0.000 description 4
- 239000000427 antigen Substances 0.000 description 4
- 102000036639 antigens Human genes 0.000 description 4
- 108091007433 antigens Proteins 0.000 description 4
- 210000003123 bronchiole Anatomy 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 4
- 238000009826 distribution Methods 0.000 description 4
- 239000003937 drug carrier Substances 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 238000006213 oxygenation reaction Methods 0.000 description 4
- 230000003449 preventive effect Effects 0.000 description 4
- 108090000623 proteins and genes Proteins 0.000 description 4
- 108010006654 Bleomycin Proteins 0.000 description 3
- 208000032843 Hemorrhage Diseases 0.000 description 3
- 241000699670 Mus sp. Species 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 208000034158 bleeding Diseases 0.000 description 3
- 230000000740 bleeding effect Effects 0.000 description 3
- 229960001561 bleomycin Drugs 0.000 description 3
- OYVAGSVQBOHSSS-UAPAGMARSA-O bleomycin A2 Chemical compound N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C OYVAGSVQBOHSSS-UAPAGMARSA-O 0.000 description 3
- 210000000621 bronchi Anatomy 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 230000003480 fibrinolytic effect Effects 0.000 description 3
- 208000014674 injury Diseases 0.000 description 3
- 238000005399 mechanical ventilation Methods 0.000 description 3
- 239000002243 precursor Substances 0.000 description 3
- 210000002345 respiratory system Anatomy 0.000 description 3
- 241000283690 Bos taurus Species 0.000 description 2
- 241000282472 Canis lupus familiaris Species 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 206010053567 Coagulopathies Diseases 0.000 description 2
- 241000283086 Equidae Species 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- 101000801481 Homo sapiens Tissue-type plasminogen activator Proteins 0.000 description 2
- 101000638886 Homo sapiens Urokinase-type plasminogen activator Proteins 0.000 description 2
- 241001494479 Pecora Species 0.000 description 2
- 208000013616 Respiratory Distress Syndrome Diseases 0.000 description 2
- 108010039185 Tenecteplase Proteins 0.000 description 2
- 208000027418 Wounds and injury Diseases 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 201000000028 adult respiratory distress syndrome Diseases 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 210000004027 cell Anatomy 0.000 description 2
- 230000015271 coagulation Effects 0.000 description 2
- 238000005345 coagulation Methods 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 210000000981 epithelium Anatomy 0.000 description 2
- 229940001501 fibrinolysin Drugs 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000012634 fragment Substances 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 210000000867 larynx Anatomy 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 239000000155 melt Substances 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 239000011859 microparticle Substances 0.000 description 2
- 210000000056 organ Anatomy 0.000 description 2
- LOQGSOTUHASIHI-UHFFFAOYSA-N perfluoro-1,3-dimethylcyclohexane Chemical compound FC(F)(F)C1(F)C(F)(F)C(F)(F)C(F)(F)C(F)(C(F)(F)F)C1(F)F LOQGSOTUHASIHI-UHFFFAOYSA-N 0.000 description 2
- FYJQJMIEZVMYSD-UHFFFAOYSA-N perfluoro-2-butyltetrahydrofuran Chemical compound FC(F)(F)C(F)(F)C(F)(F)C(F)(F)C1(F)OC(F)(F)C(F)(F)C1(F)F FYJQJMIEZVMYSD-UHFFFAOYSA-N 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 102000040430 polynucleotide Human genes 0.000 description 2
- 108091033319 polynucleotide Proteins 0.000 description 2
- 239000002157 polynucleotide Substances 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 230000006337 proteolytic cleavage Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 230000029058 respiratory gaseous exchange Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 239000007921 spray Substances 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 230000009885 systemic effect Effects 0.000 description 2
- 229960000216 tenecteplase Drugs 0.000 description 2
- 229960005356 urokinase Drugs 0.000 description 2
- YFYNOWXBIBKGHB-FBCQKBJTSA-N (1s,3r)-1-aminocyclopentane-1,3-dicarboxylic acid Chemical compound OC(=O)[C@]1(N)CC[C@@H](C(O)=O)C1 YFYNOWXBIBKGHB-FBCQKBJTSA-N 0.000 description 1
- OZFAFGSSMRRTDW-UHFFFAOYSA-N (2,4-dichlorophenyl) benzenesulfonate Chemical compound ClC1=CC(Cl)=CC=C1OS(=O)(=O)C1=CC=CC=C1 OZFAFGSSMRRTDW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 108010057987 Desmodus rotundus salivary plasminogen activator alpha 1 Proteins 0.000 description 1
- 239000012591 Dulbecco’s Phosphate Buffered Saline Substances 0.000 description 1
- 102000010911 Enzyme Precursors Human genes 0.000 description 1
- 108010062466 Enzyme Precursors Proteins 0.000 description 1
- 102000008946 Fibrinogen Human genes 0.000 description 1
- 108010049003 Fibrinogen Proteins 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 101100043639 Glycine max ACPD gene Proteins 0.000 description 1
- 241001272567 Hominoidea Species 0.000 description 1
- PWKSKIMOESPYIA-BYPYZUCNSA-N L-N-acetyl-Cysteine Chemical compound CC(=O)N[C@@H](CS)C(O)=O PWKSKIMOESPYIA-BYPYZUCNSA-N 0.000 description 1
- 206010025102 Lung infiltration Diseases 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 102000015636 Oligopeptides Human genes 0.000 description 1
- 108010038807 Oligopeptides Proteins 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 101150034459 Parpbp gene Proteins 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 241000288906 Primates Species 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 208000021063 Respiratory fume inhalation disease Diseases 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 102000002262 Thromboplastin Human genes 0.000 description 1
- 108010000499 Thromboplastin Proteins 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 229960004308 acetylcysteine Drugs 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 125000000539 amino acid group Chemical group 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 238000005842 biochemical reaction Methods 0.000 description 1
- 229940088623 biologically active substance Drugs 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- 201000009267 bronchiectasis Diseases 0.000 description 1
- 239000007975 buffered saline Substances 0.000 description 1
- 210000004899 c-terminal region Anatomy 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 230000035602 clotting Effects 0.000 description 1
- 238000003271 compound fluorescence assay Methods 0.000 description 1
- 238000004590 computer program Methods 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 231100000517 death Toxicity 0.000 description 1
- 230000006735 deficit Effects 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- 229950001282 desmoteplase Drugs 0.000 description 1
- 230000010339 dilation Effects 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 239000013583 drug formulation Substances 0.000 description 1
- 235000006694 eating habits Nutrition 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 238000001839 endoscopy Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 229940012952 fibrinogen Drugs 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 238000007917 intracranial administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000012669 liquid formulation Substances 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 210000000885 nephron Anatomy 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 150000004812 organic fluorine compounds Chemical class 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 238000010951 particle size reduction Methods 0.000 description 1
- YVBBRRALBYAZBM-UHFFFAOYSA-N perfluorooctane Chemical compound FC(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F YVBBRRALBYAZBM-UHFFFAOYSA-N 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 239000002574 poison Substances 0.000 description 1
- 231100000614 poison Toxicity 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 239000000955 prescription drug Substances 0.000 description 1
- 239000000651 prodrug Substances 0.000 description 1
- 229940002612 prodrug Drugs 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 229940024999 proteolytic enzymes for treatment of wounds and ulcers Drugs 0.000 description 1
- 208000005069 pulmonary fibrosis Diseases 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 239000012266 salt solution Substances 0.000 description 1
- -1 sc-tPA and dc-tPA) Proteins 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 230000003319 supportive effect Effects 0.000 description 1
- 238000009120 supportive therapy Methods 0.000 description 1
- 230000002277 temperature effect Effects 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 239000011573 trace mineral Substances 0.000 description 1
- 235000013619 trace mineral Nutrition 0.000 description 1
- 238000002627 tracheal intubation Methods 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 230000007306 turnover Effects 0.000 description 1
- 210000001215 vagina Anatomy 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 230000002861 ventricular Effects 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/43—Enzymes; Proenzymes; Derivatives thereof
- A61K38/46—Hydrolases (3)
- A61K38/48—Hydrolases (3) acting on peptide bonds (3.4)
- A61K38/49—Urokinase; Tissue plasminogen activator
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/007—Pulmonary tract; Aromatherapy
- A61K9/0073—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy
- A61K9/0078—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy for inhalation via a nebulizer such as a jet nebulizer, ultrasonic nebulizer, e.g. in the form of aqueous drug solutions or dispersions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/43—Enzymes; Proenzymes; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/007—Pulmonary tract; Aromatherapy
- A61K9/0073—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy
- A61K9/008—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy comprising drug dissolved or suspended in liquid propellant for inhalation via a pressurized metered dose inhaler [MDI]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y304/00—Hydrolases acting on peptide bonds, i.e. peptidases (3.4)
- C12Y304/21—Serine endopeptidases (3.4.21)
- C12Y304/21068—Tissue plasminogen activator (3.4.21.68), i.e. tPA
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/12—Aerosols; Foams
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y304/00—Hydrolases acting on peptide bonds, i.e. peptidases (3.4)
- C12Y304/21—Serine endopeptidases (3.4.21)
- C12Y304/21073—Serine endopeptidases (3.4.21) u-Plasminogen activator (3.4.21.73), i.e. urokinase
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Pulmonology (AREA)
- Otolaryngology (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Gastroenterology & Hepatology (AREA)
- Organic Chemistry (AREA)
- Dispersion Chemistry (AREA)
- Biomedical Technology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicinal Preparation (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361899739P | 2013-11-04 | 2013-11-04 | |
| US61/899,739 | 2013-11-04 | ||
| PCT/US2014/063784 WO2015066664A2 (en) | 2013-11-04 | 2014-11-04 | Compositions and methods for administration of an enzyme to a subject's airway |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20160078986A true KR20160078986A (ko) | 2016-07-05 |
Family
ID=53005414
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020167012122A Withdrawn KR20160078986A (ko) | 2013-11-04 | 2014-11-04 | 대상체의 기도로 효소를 투여하기 위한 조성물 및 방법 |
Country Status (11)
| Country | Link |
|---|---|
| US (2) | US20160279209A1 (enExample) |
| EP (1) | EP3065705A4 (enExample) |
| JP (1) | JP2016535782A (enExample) |
| KR (1) | KR20160078986A (enExample) |
| CN (1) | CN105764488A (enExample) |
| AU (1) | AU2014341862A1 (enExample) |
| CA (1) | CA2928759A1 (enExample) |
| CL (1) | CL2016001062A1 (enExample) |
| MX (1) | MX2016005720A (enExample) |
| PE (1) | PE20160789A1 (enExample) |
| WO (1) | WO2015066664A2 (enExample) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4438044A3 (en) | 2013-03-15 | 2024-12-04 | Board of Regents, The University of Texas System | Peptide fttftvt for use in treating fibrosis |
| AU2019339260B2 (en) | 2018-09-10 | 2025-06-26 | Rein Therapeutics, Inc. | Modified peptide fragments of Cav-1 protein and the use thereof in the treatment of fibrosis |
| WO2021180068A1 (zh) * | 2020-03-09 | 2021-09-16 | 泰伦基国际有限公司 | 一种治疗2019新型冠状病毒引发疾病的方法和药物 |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4790305A (en) * | 1986-06-23 | 1988-12-13 | The Johns Hopkins University | Medication delivery system |
| EP0266032A1 (en) | 1986-08-29 | 1988-05-04 | Beecham Group Plc | Modified fibrinolytic enzyme |
| US5302390A (en) | 1987-07-01 | 1994-04-12 | Beecham Group Plc | Hybrid proteins of human plasminogen and human t-PA, pharmaceutical compositions and methods of treatment |
| US5531219A (en) * | 1994-11-04 | 1996-07-02 | Alliance Pharmaceutical Corp. | Use of liquid fluorocarbons to facilitate pulmonary drug delivery |
| WO1998006426A1 (en) * | 1996-08-14 | 1998-02-19 | Gram Joergen Brodersen | Use of a plasminogen activator for the treatment of pulmonary disorders |
| US20030113271A1 (en) * | 1997-01-29 | 2003-06-19 | University Technology Corporation | Formulations for pulmonary delivery |
| CN1620334A (zh) * | 2001-12-21 | 2005-05-25 | 环境清洁技术公司 | 锰氧化物的预处理与再生 |
| US20050036951A1 (en) * | 2003-01-09 | 2005-02-17 | Arizeke Pharmaceuticals, Inc. | Methods of treating lung diseases |
| US20050169908A1 (en) | 2004-01-23 | 2005-08-04 | Kazunori Murakami | Use of aerosolized antithrombin to treat acute lung injury |
| US20060198940A1 (en) | 2005-03-04 | 2006-09-07 | Mcmorrow David | Method of producing particles utilizing a vibrating mesh nebulizer for coating a medical appliance, a system for producing particles, and a medical appliance |
| US20070157931A1 (en) | 2005-07-11 | 2007-07-12 | Richard Parker | System and method for optimized delivery of an aerosol to the respiratory tract |
| EP1962869B1 (en) * | 2005-12-21 | 2013-03-20 | SolAeroMed Inc. | Treatment of respiratory diseases |
| SI22865A (sl) | 2008-09-25 | 2010-03-31 | Univerza@V@Ljubljani | Peptid uroaktivin kot aktivator encima urokinaze |
| JP2012509941A (ja) * | 2008-11-26 | 2012-04-26 | ファイブ・プライム・セラピューティクス,インコーポレイテッド | Serpine2によるコラーゲンおよび平滑筋アクチンの発現を調節するための組成物および方法 |
| DE102009031274A1 (de) * | 2009-06-30 | 2011-01-13 | Justus-Liebig-Universität Giessen | Liposomen zur pulmonalen Applikation |
| WO2013029627A1 (en) * | 2011-08-29 | 2013-03-07 | Trifoilium Aps | Compositions and methods for treating or preventing radiation- or chemotherapy-induced pulmonary dysfunction |
-
2014
- 2014-11-04 AU AU2014341862A patent/AU2014341862A1/en not_active Abandoned
- 2014-11-04 CA CA2928759A patent/CA2928759A1/en not_active Abandoned
- 2014-11-04 EP EP14857307.4A patent/EP3065705A4/en not_active Withdrawn
- 2014-11-04 WO PCT/US2014/063784 patent/WO2015066664A2/en not_active Ceased
- 2014-11-04 US US15/034,019 patent/US20160279209A1/en not_active Abandoned
- 2014-11-04 CN CN201480060878.2A patent/CN105764488A/zh active Pending
- 2014-11-04 JP JP2016552448A patent/JP2016535782A/ja active Pending
- 2014-11-04 KR KR1020167012122A patent/KR20160078986A/ko not_active Withdrawn
- 2014-11-04 PE PE2016000563A patent/PE20160789A1/es not_active Application Discontinuation
- 2014-11-04 MX MX2016005720A patent/MX2016005720A/es unknown
-
2016
- 2016-05-03 CL CL2016001062A patent/CL2016001062A1/es unknown
-
2019
- 2019-01-28 US US16/259,112 patent/US11033611B2/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| US20190255157A1 (en) | 2019-08-22 |
| PE20160789A1 (es) | 2016-08-17 |
| EP3065705A2 (en) | 2016-09-14 |
| CA2928759A1 (en) | 2015-05-07 |
| WO2015066664A2 (en) | 2015-05-07 |
| AU2014341862A1 (en) | 2016-05-19 |
| CN105764488A (zh) | 2016-07-13 |
| US11033611B2 (en) | 2021-06-15 |
| MX2016005720A (es) | 2016-11-25 |
| CL2016001062A1 (es) | 2017-01-13 |
| US20160279209A1 (en) | 2016-09-29 |
| JP2016535782A (ja) | 2016-11-17 |
| WO2015066664A3 (en) | 2015-09-17 |
| EP3065705A4 (en) | 2017-10-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| ES2792682T3 (es) | Métodos para el tratamiento de enfermedades pulmonares con estabilizadores de mastocitos | |
| ES2970401T3 (es) | Compuestos y composiciones para uso en un método de tratamiento del virus de parainfluenza | |
| US8709496B2 (en) | Use of deuterium oxide for the treatment of virus-based diseases of the respiratory tract | |
| JP6397984B2 (ja) | 乾燥粉末ペプチド医薬 | |
| JP5970465B2 (ja) | ペプチドおよびウイルス・ノイラミニダーゼ阻害剤を含んでなる組成物 | |
| JP2023511763A (ja) | 炎症性肺疾患の吸入治療用のヒト抗炎症性ペプチド | |
| US11033611B2 (en) | Compositions and methods for administration of an enzyme to a subject's airway | |
| WO2022009081A1 (en) | Drug for use against the novel coronavirus disease, covid-19 | |
| KR102664833B1 (ko) | 나파모스타트 또는 카모스타트를 포함하는 흡입용 제제 | |
| AU2021326382B2 (en) | Use of a heparin composition in the treatment of viral lung diseases, acute and/or chronic lung diseases by soft mist inhaler or vibration mesh technology nebulizer through inhalation route | |
| CN115843267A (zh) | 呼吸道病毒感染的治疗 | |
| US20180140547A1 (en) | Compositions and methods for administration of an enzyme to a subject's airway | |
| TWI788779B (zh) | 一種治療病毒性肺炎的方法和藥物 | |
| RU2843523C1 (ru) | Применение композиции на основе гепарина при лечении вирусных заболеваний легких, острых и/или хронических заболеваний легких с помощью ингалятора «мягкого тумана» или небулайзера с технологией вибрирующей сетки посредством ингаляционного пути введения | |
| CN102369012B (zh) | 氧化氘用于治疗病毒性呼吸道疾病的用途 | |
| WO2025210089A1 (en) | A combination of vasoactive intestinal peptide (vip) receptor agonist and a mucolytic agent against inflammatory diseases of the respiratory tract | |
| WO2022140817A1 (en) | Anti-viral proteases and methods of use | |
| WO2021188334A1 (en) | Use of st266 to treat severe systemic inflammation and post-acute covid-19 syndrome | |
| HK40084754A (en) | Method and drug for treating viral pneumonia | |
| Abdalaziz et al. | Formulation strategy for hydroxychloroquine as inhaler dosage from as a potential for COVID-19 treatment |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20160509 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PC1203 | Withdrawal of no request for examination | ||
| WITN | Application deemed withdrawn, e.g. because no request for examination was filed or no examination fee was paid |