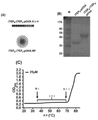
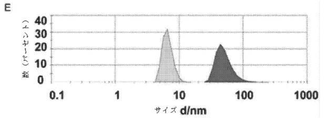
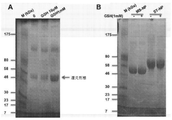
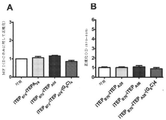
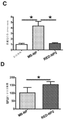
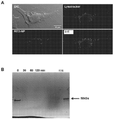
JP6924151B2 - 免疫寛容及び非免疫寛容エラスチン様組換えペプチドならびに使用方法 - Google Patents
免疫寛容及び非免疫寛容エラスチン様組換えペプチドならびに使用方法 Download PDFInfo
- Publication number
- JP6924151B2 JP6924151B2 JP2017561974A JP2017561974A JP6924151B2 JP 6924151 B2 JP6924151 B2 JP 6924151B2 JP 2017561974 A JP2017561974 A JP 2017561974A JP 2017561974 A JP2017561974 A JP 2017561974A JP 6924151 B2 JP6924151 B2 JP 6924151B2
- Authority
- JP
- Japan
- Prior art keywords
- gly
- itep
- val
- seq
- pro
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/64—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent
- A61K47/6435—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent the peptide or protein in the drug conjugate being a connective tissue peptide, e.g. collagen, fibronectin or gelatin
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/78—Connective tissue peptides, e.g. collagen, elastin, laminin, fibronectin, vitronectin or cold insoluble globulin [CIG]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/337—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having four-membered rings, e.g. taxol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/35—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/64—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/76—Albumins
- C07K14/77—Ovalbumin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/60—Medicinal preparations containing antigens or antibodies characteristics by the carrier linked to the antigen
- A61K2039/6031—Proteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Organic Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Toxicology (AREA)
- Gastroenterology & Hepatology (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Zoology (AREA)
- Biochemistry (AREA)
- Oncology (AREA)
- Microbiology (AREA)
- Immunology (AREA)
- Biomedical Technology (AREA)
- Mycology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201562230160P | 2015-05-29 | 2015-05-29 | |
| US62/230,160 | 2015-05-29 | ||
| US201662309113P | 2016-03-16 | 2016-03-16 | |
| US62/309,113 | 2016-03-16 | ||
| PCT/US2016/034530 WO2016196249A1 (en) | 2015-05-29 | 2016-05-27 | Immune tolerant and non-immune tolerant elastin-like recombinant peptides and methods of use |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2018521977A JP2018521977A (ja) | 2018-08-09 |
| JP2018521977A5 JP2018521977A5 (enExample) | 2019-07-04 |
| JP6924151B2 true JP6924151B2 (ja) | 2021-08-25 |
Family
ID=57441707
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2017561974A Active JP6924151B2 (ja) | 2015-05-29 | 2016-05-27 | 免疫寛容及び非免疫寛容エラスチン様組換えペプチドならびに使用方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US11957762B2 (enExample) |
| EP (1) | EP3302531B1 (enExample) |
| JP (1) | JP6924151B2 (enExample) |
| CN (1) | CN107750163A (enExample) |
| CA (1) | CA2986766A1 (enExample) |
| WO (1) | WO2016196249A1 (enExample) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11198722B2 (en) | 2017-10-06 | 2021-12-14 | University Of Utah Research Foundation | Immune tolerant elastin-like peptide tetramer guided nanoparticles and methods of use |
| WO2020048996A1 (en) | 2018-09-06 | 2020-03-12 | ETH Zürich | Self-assembling globular proteins and uses thereof |
| CN111346235A (zh) * | 2018-12-20 | 2020-06-30 | 海南医学院第一附属医院 | 一种基于弹性靶向多肽的载药纳米颗粒及其制备方法和应用 |
| KR20220103916A (ko) * | 2019-08-23 | 2022-07-25 | 유타대학연구재단 | 면역 내성 엘라스틴-유사 재조합 펩티드 및 사용 방법 |
| EP4035682A1 (en) * | 2021-01-29 | 2022-08-03 | Universite De Bordeaux | Bioconjugate comprising a photosensitizer unit, method for its preparation and use thereof |
| KR102718109B1 (ko) * | 2021-10-01 | 2024-10-17 | (주)케어젠 | 탈모 방지 또는 발모 촉진 활성을 갖는 펩타이드와 이의 용도 |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4703004A (en) | 1984-01-24 | 1987-10-27 | Immunex Corporation | Synthesis of protein with an identification peptide |
| US4851341A (en) | 1986-12-19 | 1989-07-25 | Immunex Corporation | Immunoaffinity purification system |
| US5206353A (en) | 1988-07-23 | 1993-04-27 | The United States Of America As Represented By The Department Of Health And Human Services | CD-4/cytotoxic gene fusions |
| AU5670194A (en) | 1992-11-20 | 1994-06-22 | Enzon, Inc. | Linker for linked fusion polypeptides |
| US5914254A (en) | 1993-08-02 | 1999-06-22 | Celtrix Pharmaceuticals, Inc. | Expression of fusion polypeptides transported out of the cytoplasm without leader sequences |
| US5763733A (en) | 1994-10-13 | 1998-06-09 | Enzon, Inc. | Antigen-binding fusion proteins |
| US5981177A (en) | 1995-01-25 | 1999-11-09 | Demirjian; David C. | Protein fusion method and constructs |
| US5994104A (en) | 1996-11-08 | 1999-11-30 | Royal Free Hospital School Of Medicine | Interleukin-12 fusion protein |
| WO1998022577A1 (en) * | 1996-11-15 | 1998-05-28 | Maria Grazia Masucci | Fusion proteins having increased half-lives |
| WO1998036087A1 (en) | 1997-02-13 | 1998-08-20 | American National Red Cross | Immunological tolerance to hiv epitopes |
| US7494788B2 (en) * | 2005-07-11 | 2009-02-24 | Molecular Kinetics, Inc. | Entropic bristle domain sequences and their use in recombinant protein production |
| US8470967B2 (en) | 2010-09-24 | 2013-06-25 | Duke University | Phase transition biopolymers and methods of use |
| US9114128B2 (en) * | 2011-09-30 | 2015-08-25 | Protein Genomics, Inc. | Tropoelastins and uses thereof |
| EP3052125B1 (en) | 2013-10-01 | 2020-09-30 | University of Mississippi Medical Center | Elastin-like polypeptide coupled to therapeutic agent for delivery during pregnancy |
-
2016
- 2016-05-27 WO PCT/US2016/034530 patent/WO2016196249A1/en not_active Ceased
- 2016-05-27 CA CA2986766A patent/CA2986766A1/en active Pending
- 2016-05-27 CN CN201680036648.1A patent/CN107750163A/zh active Pending
- 2016-05-27 EP EP16804094.7A patent/EP3302531B1/en active Active
- 2016-05-27 US US15/577,998 patent/US11957762B2/en active Active
- 2016-05-27 JP JP2017561974A patent/JP6924151B2/ja active Active
Also Published As
| Publication number | Publication date |
|---|---|
| EP3302531B1 (en) | 2021-02-17 |
| JP2018521977A (ja) | 2018-08-09 |
| US20180289830A1 (en) | 2018-10-11 |
| CA2986766A1 (en) | 2016-12-08 |
| US11957762B2 (en) | 2024-04-16 |
| EP3302531A1 (en) | 2018-04-11 |
| WO2016196249A1 (en) | 2016-12-08 |
| EP3302531A4 (en) | 2019-02-20 |
| CN107750163A (zh) | 2018-03-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6924151B2 (ja) | 免疫寛容及び非免疫寛容エラスチン様組換えペプチドならびに使用方法 | |
| Dacoba et al. | Polysaccharide nanoparticles can efficiently modulate the immune response against an HIV peptide antigen | |
| RU2748378C2 (ru) | Комбинация модулятора иммунных контрольных точек и комплекса, содержащего проникающий в клетку пептид, карго-молекулу и пептидный агонист tlr, для применения в медицине | |
| TWI823054B (zh) | IL-7Rα結合化合物 | |
| Moyle et al. | Site-specific incorporation of three toll-like receptor 2 targeting adjuvants into semisynthetic, molecularly defined nanoparticles: application to group a streptococcal vaccines | |
| Barrett et al. | Modular peptide amphiphile micelles improving an antibody-mediated immune response to Group A Streptococcus | |
| Cho et al. | Immune-tolerant elastin-like polypeptides (iTEPs) and their application as CTL vaccine carriers | |
| EP3572429A1 (en) | Pd-l1 binding polypeptide or composite | |
| WO2012099805A2 (en) | Nanoparticle based immunological stimulation | |
| WO2016146261A1 (en) | Combination of an immune checkpoint modulator and a complex comprising a cell penetrating peptide, a cargo and a tlr peptide agonist for use in medicine | |
| JP2017524342A (ja) | プロテアーゼ切断耐性、志賀毒素aサブユニットエフェクターポリペプチド及びそれを含む細胞標的化分子 | |
| ES2781085T3 (es) | Mutantes de griffithsina | |
| WO2011099684A1 (ko) | Muc1에 대한 단일 도메인 항체 | |
| Rudra et al. | Supramolecular peptide nanofibers engage mechanisms of autophagy in antigen-presenting cells | |
| KR101579879B1 (ko) | siRNA 전달을 위한 히알루론산-콜레스테롤 나노파티클 및 이를 포함하는 조성물 | |
| Stahl et al. | Enzyme-triggered antigen release enhances cross-presentation by dendritic cells | |
| TW202521160A (zh) | 靶向傳遞至細胞之藥劑及方法 | |
| KR101430232B1 (ko) | siRNA 전달을 위한 p19-YSA 재조합 단백질 및 이를 포함하는 조성물 | |
| US20220145316A1 (en) | Cancer immunotherapy | |
| Özcan et al. | New generation peptide-based vaccine prototype | |
| Pho | Surface Engineering of Protein Nanoparticles for Intranasal Delivery | |
| WO2010129710A1 (en) | Use of rankl to induce differentiation of microfold cells (m cells) | |
| Lin | Development of gold nanoparticle-based antigen delivery platform for vaccines against HIV-1 | |
| US7314626B2 (en) | Use of peptide vectors to improve the immune response to antigens | |
| Ruseska et al. | Protamine–a review on an oligonucleotide-binding peptide applied in nanopharmaceuticals including vaccines |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20190527 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190603 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20200622 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20200918 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20210107 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210405 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20210701 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20210730 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6924151 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |