JP6761345B2 - 生体構造非依存性偏向装置 - Google Patents
生体構造非依存性偏向装置 Download PDFInfo
- Publication number
- JP6761345B2 JP6761345B2 JP2016545785A JP2016545785A JP6761345B2 JP 6761345 B2 JP6761345 B2 JP 6761345B2 JP 2016545785 A JP2016545785 A JP 2016545785A JP 2016545785 A JP2016545785 A JP 2016545785A JP 6761345 B2 JP6761345 B2 JP 6761345B2
- Authority
- JP
- Japan
- Prior art keywords
- wire
- filter
- catheter
- attached
- transverse structure
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 210000002376 aorta thoracic Anatomy 0.000 claims description 29
- 239000011324 bead Substances 0.000 claims description 18
- 230000007246 mechanism Effects 0.000 claims description 16
- 208000005189 Embolism Diseases 0.000 claims description 15
- 230000005855 radiation Effects 0.000 claims description 7
- 229910052751 metal Inorganic materials 0.000 claims description 6
- 239000002184 metal Substances 0.000 claims description 6
- 239000003550 marker Substances 0.000 claims description 2
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 claims description 2
- 229920000642 polymer Polymers 0.000 claims 1
- 210000004204 blood vessel Anatomy 0.000 description 30
- 238000013461 design Methods 0.000 description 14
- 238000000034 method Methods 0.000 description 11
- 239000002245 particle Substances 0.000 description 11
- 239000000463 material Substances 0.000 description 10
- 239000013618 particulate matter Substances 0.000 description 10
- 210000000709 aorta Anatomy 0.000 description 9
- 210000001367 artery Anatomy 0.000 description 6
- 210000004369 blood Anatomy 0.000 description 6
- 239000008280 blood Substances 0.000 description 6
- 210000004556 brain Anatomy 0.000 description 6
- 230000006378 damage Effects 0.000 description 6
- 229910001000 nickel titanium Inorganic materials 0.000 description 5
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 5
- 210000003743 erythrocyte Anatomy 0.000 description 4
- 210000000265 leukocyte Anatomy 0.000 description 4
- 239000011148 porous material Substances 0.000 description 4
- 239000004677 Nylon Substances 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- 229920001778 nylon Polymers 0.000 description 3
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 3
- -1 polytetrafluoroethylene Polymers 0.000 description 3
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 3
- 239000004810 polytetrafluoroethylene Substances 0.000 description 3
- 239000002033 PVDF binder Substances 0.000 description 2
- 229920006362 Teflon® Polymers 0.000 description 2
- 208000007536 Thrombosis Diseases 0.000 description 2
- 206010000891 acute myocardial infarction Diseases 0.000 description 2
- 210000001765 aortic valve Anatomy 0.000 description 2
- 230000000747 cardiac effect Effects 0.000 description 2
- 210000001326 carotid sinus Anatomy 0.000 description 2
- 239000000788 chromium alloy Substances 0.000 description 2
- 238000002591 computed tomography Methods 0.000 description 2
- 230000003073 embolic effect Effects 0.000 description 2
- 230000010102 embolization Effects 0.000 description 2
- 238000009434 installation Methods 0.000 description 2
- 238000002595 magnetic resonance imaging Methods 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 2
- 230000008439 repair process Effects 0.000 description 2
- 229910001285 shape-memory alloy Inorganic materials 0.000 description 2
- 210000003270 subclavian artery Anatomy 0.000 description 2
- 229910052715 tantalum Inorganic materials 0.000 description 2
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 2
- 206010065558 Aortic arteriosclerosis Diseases 0.000 description 1
- 229910000684 Cobalt-chrome Inorganic materials 0.000 description 1
- 208000002330 Congenital Heart Defects Diseases 0.000 description 1
- 229920004934 Dacron® Polymers 0.000 description 1
- 206010014498 Embolic stroke Diseases 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- 229920000271 Kevlar® Polymers 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 241000124033 Salix Species 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- 238000002583 angiography Methods 0.000 description 1
- 201000001962 aortic atherosclerosis Diseases 0.000 description 1
- 229920003235 aromatic polyamide Polymers 0.000 description 1
- 206010003119 arrhythmia Diseases 0.000 description 1
- 230000003143 atherosclerotic effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 230000036770 blood supply Effects 0.000 description 1
- 210000001168 carotid artery common Anatomy 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 208000028831 congenital heart disease Diseases 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 238000002716 delivery method Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 229910003460 diamond Inorganic materials 0.000 description 1
- 239000010432 diamond Substances 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000002592 echocardiography Methods 0.000 description 1
- 206010014665 endocarditis Diseases 0.000 description 1
- 230000003511 endothelial effect Effects 0.000 description 1
- 229920000295 expanded polytetrafluoroethylene Polymers 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 229920002313 fluoropolymer Polymers 0.000 description 1
- 239000004811 fluoropolymer Substances 0.000 description 1
- 238000002594 fluoroscopy Methods 0.000 description 1
- 210000004491 foramen ovale Anatomy 0.000 description 1
- 208000014951 hematologic disease Diseases 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 238000003780 insertion Methods 0.000 description 1
- 230000037431 insertion Effects 0.000 description 1
- 238000002608 intravascular ultrasound Methods 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 210000004115 mitral valve Anatomy 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 230000033764 rhythmic process Effects 0.000 description 1
- 238000009958 sewing Methods 0.000 description 1
- 239000004753 textile Substances 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 230000001732 thrombotic effect Effects 0.000 description 1
- 238000002604 ultrasonography Methods 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
- A61F2/011—Instruments for their placement or removal
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
- A61F2/013—Distal protection devices, i.e. devices placed distally in combination with another endovascular procedure, e.g. angioplasty or stenting
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
- A61F2002/016—Filters implantable into blood vessels made from wire-like elements
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
- A61F2002/018—Filters implantable into blood vessels made from tubes or sheets of material, e.g. by etching or laser-cutting
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2220/00—Fixations or connections for prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2220/0025—Connections or couplings between prosthetic parts, e.g. between modular parts; Connecting elements
- A61F2220/0041—Connections or couplings between prosthetic parts, e.g. between modular parts; Connecting elements using additional screws, bolts, dowels or rivets, e.g. connecting screws
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0002—Two-dimensional shapes, e.g. cross-sections
- A61F2230/0004—Rounded shapes, e.g. with rounded corners
- A61F2230/0008—Rounded shapes, e.g. with rounded corners elliptical or oval
Description
Claims (22)
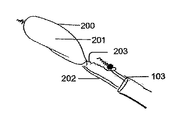
- 塞栓を偏向させるために、大動脈弓に配置されるデバイスであって、
細長い形状を取り囲む横方向構造体(200)と、
前記横方向構造体(200)の長さ及び幅に取り付けられた細長いフィルタ(201)とを備えるデバイスにおいて、
前記横方向構造体の前記長さに沿って配置され、前記デバイスが前記大動脈弓に配置されたときに、前記横方向構造体の遠位端部に、または前記フィルタ(201)の遠位端部に直接または間接的に接続された第1のワイヤ(202)と、
前記横方向構造体又は前記フィルタ(201)の近位端部に取り付けられた第2のワイヤ(203)とを備え、
前記フィルタ(201)が前記第1のワイヤ(202)と類似の形状に曲げられるように、前記第1のワイヤ(202)が、予め曲げられた形状を有し、前記フィルタ(201)の中央に沿って取り付けられており、
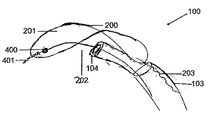
前記第1のワイヤ(202)及び前記第2のワイヤ(203)は、前記デバイス(100)の位置、配向、若しくは偏向を制御するように、拡張したフィルタ(201)に対して配置され、前記デバイスが前記大動脈弓に配置されたときに前記第1のワイヤ(202)が前記横方向構造体の遠位端部または前記フィルタ(201)の遠位端部から取り外し可能である、デバイス。 - 前記第1のワイヤ(202)は、さらに、その遠位端部に配置されたねじ様機構(401)を備えており、前記フィルタ(201)又は前記横方向構造体(200)は、その遠位端部に、前記ねじ様機構(401)を受け入れるのに適した接続構成要素を備えている、請求項1に記載のデバイス。
- 前記接続構成要素は、前記フィルタ(201)又は前記横方向構造体(200)の前記遠位端部に取り付けられたビーズ(400)を備えている、請求項2に記載のデバイス。
- 前記フィルタ(201)は、前記横方向構造体(200)によって取り囲まれた周囲を越えて延びている、請求項1から3のいずれか一項に記載のデバイス。
- 前記第1のワイヤ(202)が前記第2のワイヤ(203)より硬いか、又は、前記第2のワイヤ(203)が前記第1のワイヤ(202)より硬い、請求項1から3のいずれか一項に記載のデバイス。
- 前記第1のワイヤ(202)及び前記第2のワイヤ(203)は、等しい硬度のものである、請求項1から3のいずれか一項に記載のデバイス。
- 前記デバイスは、金属ワイヤ又は重合体ワイヤから作製されている、請求項1から6のいずれか一項に記載のデバイス。
- 前記デバイスは、ニチノールワイヤから作製されている、請求項1から6のいずれか一項に記載のデバイス。
- 前記ワイヤの直径は、約1.0mm又はそれ以下である、請求項1から8のいずれか一項に記載のデバイス。
- 前記横方向構造体(200)は、約1.0mm未満の直径を有する前記ワイヤの二つ以上のねじりを備えている、請求項9に記載のデバイス。
- 前記デバイスは、さらに、放射線不透過性マーカを備えている、請求項1から10のいずれか一項に記載のデバイス。
- 大動脈弓内における設置時、前記横方向構造体は、前記大動脈弓の壁と共にシールを形成している、請求項1から11のいずれか一項に記載のデバイス。
- 前記フィルタ(201)は、50、100、又は200μmより大きい寸法を有する塞栓を有意に通さない、請求項1から12のいずれか一項に記載のデバイス。
- 前記第1のワイヤ又は前記第2のワイヤは、前記フィルタに取り付けられている、請求項1から13のいずれか一項に記載のデバイス。
- 前記第2のワイヤ(203)は、中空チューブである、請求項1から14のいずれか一項に記載のデバイス。
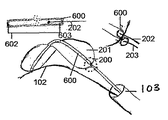
- 前記横方向構造体(200)は、さらに、二つのボール又はチップ(700)を前記遠位端部及び前記近位端部に備えている、請求項1から15のいずれか一項に記載のデバイス。
- 前記第1のワイヤ(202)及び/又は前記第2のワイヤ(203)は、スネア(701)を含み、前記フィルタ(201)の近位端部及び/又は遠位端部は、前記スネアと接合するビーズ(400)又は他の受け入れ要素を含んでいる、請求項1から16のいずれか一項に記載のデバイス。
- 前記フィルタ(201)は、さらに、前記フィルタの長さに沿って配置された中空チューブ(600)を備えている、請求項1から17のいずれか一項に記載のデバイス。
- 請求項1から9のいずれか一項に記載のデバイスを備えるシステムであって、前記デバイスがカテーテル(103)内に装填されている、システム。
- 前記カテーテル(103)は、さらに、ガイドワイヤ(102)を備えている、請求項19に記載のシステム。
- 前記デバイス(100)の前記横方向構造体(200)及び前記フィルタ(201)に対する前記カテーテル(103)の後退時、前記デバイス(100)の前記横方向構造体(200)及び前記フィルタ(201)は拡張する、請求項19または20に記載のシステム。
- 前記カテーテル(103)の開口部は、前記カテーテル(103)の長手方向軸に対して垂直でない、請求項19から21のいずれか一項に記載のシステム。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020105495A JP6978548B2 (ja) | 2014-01-10 | 2020-06-18 | 生体構造非依存性偏向装置 |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201461926142P | 2014-01-10 | 2014-01-10 | |
| US61/926,142 | 2014-01-10 | ||
| PCT/IB2015/000410 WO2015104645A2 (en) | 2014-01-10 | 2015-01-09 | Anatomy independent deflector |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020105495A Division JP6978548B2 (ja) | 2014-01-10 | 2020-06-18 | 生体構造非依存性偏向装置 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2017501840A JP2017501840A (ja) | 2017-01-19 |
| JP2017501840A5 JP2017501840A5 (ja) | 2019-06-06 |
| JP6761345B2 true JP6761345B2 (ja) | 2020-09-23 |
Family
ID=52997479
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016545785A Active JP6761345B2 (ja) | 2014-01-10 | 2015-01-09 | 生体構造非依存性偏向装置 |
| JP2020105495A Active JP6978548B2 (ja) | 2014-01-10 | 2020-06-18 | 生体構造非依存性偏向装置 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020105495A Active JP6978548B2 (ja) | 2014-01-10 | 2020-06-18 | 生体構造非依存性偏向装置 |
Country Status (5)
| Country | Link |
|---|---|
| US (2) | US20160324621A1 (ja) |
| EP (1) | EP3091937B1 (ja) |
| JP (2) | JP6761345B2 (ja) |
| CN (2) | CN110215312A (ja) |
| WO (1) | WO2015104645A2 (ja) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2759746C (en) | 2001-12-05 | 2018-05-22 | Smt Research And Development Ltd. | Endovascular device for entrapment of particulate matter and method for use |
| WO2015177322A1 (en) | 2014-05-21 | 2015-11-26 | Swat Medical Ab | Improved embolic protection device and method |
| WO2016116816A2 (en) * | 2015-01-20 | 2016-07-28 | Keystone Heart Ltd. | Intravascular devices and delivery systems and uses thereof |
| WO2016168616A1 (en) | 2015-04-16 | 2016-10-20 | Sanford Health | Vessel filter and methods for use |
| WO2017042808A1 (en) * | 2015-09-07 | 2017-03-16 | Filterlex Medical Ltd. | Intra-aortic emboli protection filter device |
| AU2015411406B2 (en) * | 2015-10-09 | 2019-02-14 | Transverse Medical, Inc. | Catheter-based apparatuses and methods |
| US11007045B2 (en) | 2016-03-02 | 2021-05-18 | C.R. Bard, Inc. | Embolic protection basket apparatus |
| US20190076231A1 (en) | 2016-03-10 | 2019-03-14 | Keystone Heart Ltd. | Intra-Aortic Device |
| DE102016012869A1 (de) * | 2016-10-28 | 2018-05-03 | Protembis Gmbh | Embolieschutzvorrichtung, Verfahren zu deren Faltung und Formvorrichtung |
| US11337790B2 (en) * | 2017-02-22 | 2022-05-24 | Boston Scientific Scimed, Inc. | Systems and methods for protecting the cerebral vasculature |
| EP3432825A4 (en) * | 2017-03-27 | 2019-05-22 | Transverse Medical, Inc. | APPARATUS AND METHODS FOR FILTER |
| EP3400901A1 (en) * | 2017-05-12 | 2018-11-14 | Keystone Heart Ltd. | A device for filtering embolic material in a vascular system |
| KR20200130351A (ko) * | 2018-03-07 | 2020-11-18 | 이너베이티브 카디오배스큘러 솔류션스, 엘엘씨 | 색전 보호 디바이스 |
| GB2576334B (en) | 2018-08-14 | 2021-06-02 | Mokita Medical Gmbh | Embolic protection systems and devices |
| JP2022524892A (ja) * | 2018-12-15 | 2022-05-10 | ラウル グェルラ、エリック | 血栓摘出カテーテルおよび使用方法 |
| US11707351B2 (en) | 2019-08-19 | 2023-07-25 | Encompass Technologies, Inc. | Embolic protection and access system |
| CN113017909A (zh) * | 2019-12-25 | 2021-06-25 | 上海微创医疗器械(集团)有限公司 | 一种血栓阻隔装置及栓塞保护系统 |
| WO2024042527A1 (en) * | 2022-08-25 | 2024-02-29 | Capricon Medical Ltd | Fixation of debris protection devices in body lumens |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6258120B1 (en) * | 1997-12-23 | 2001-07-10 | Embol-X, Inc. | Implantable cerebral protection device and methods of use |
| US6361545B1 (en) * | 1997-09-26 | 2002-03-26 | Cardeon Corporation | Perfusion filter catheter |
| US6371935B1 (en) * | 1999-01-22 | 2002-04-16 | Cardeon Corporation | Aortic catheter with flow divider and methods for preventing cerebral embolization |
| US6908474B2 (en) * | 1998-05-13 | 2005-06-21 | Gore Enterprise Holdings, Inc. | Apparatus and methods for reducing embolization during treatment of carotid artery disease |
| US6245087B1 (en) * | 1999-08-03 | 2001-06-12 | Embol-X, Inc. | Variable expansion frame system for deploying medical devices and methods of use |
| US20030023263A1 (en) * | 2001-07-24 | 2003-01-30 | Incept Llc | Apparatus and methods for aspirating emboli |
| CA2759746C (en) | 2001-12-05 | 2018-05-22 | Smt Research And Development Ltd. | Endovascular device for entrapment of particulate matter and method for use |
| US20080255603A1 (en) | 2005-06-10 | 2008-10-16 | Sagax, Inc. | Implant Device Particularly Useful For Implantation In the Intravascular System For Diverting Emboli |
| US8062324B2 (en) | 2006-05-08 | 2011-11-22 | S.M.T. Research And Development Ltd. | Device and method for vascular filter |
| US20100324589A1 (en) * | 2006-09-11 | 2010-12-23 | Carpenter Judith T | Embolic deflection device |
| KR101720149B1 (ko) * | 2008-09-04 | 2017-03-27 | 앤덜스 존슨 | 일시적인 색전 방지 장치 및 그것의 전달을 위한 의료 방법 |
| WO2011017103A2 (en) * | 2009-07-27 | 2011-02-10 | Claret Medical, Inc. | Dual endovascular filter and methods of use |
| US20130184739A1 (en) * | 2010-04-28 | 2013-07-18 | Eamon Brady | Clot engagement and removal systems |
| WO2012085916A2 (en) * | 2010-12-23 | 2012-06-28 | Smt Research And Development Ltd. | Device and method for deflecting emboli in an aorta |
| EP2522307B1 (en) * | 2011-05-08 | 2020-09-30 | ITSO Medical AB | Device for delivery of medical devices to a cardiac valve |
| WO2012160556A1 (en) * | 2011-05-26 | 2012-11-29 | Keystone Heart Ltd. | An embolic filter device and method of use thereof |
| US20130123835A1 (en) * | 2011-11-14 | 2013-05-16 | Boston Scientific Scimed, Inc. | Embolic protection device and method |
| US20150039016A1 (en) * | 2012-03-09 | 2015-02-05 | Keystone Heart Ltd. | Device and method for deflecting emboli in an aorta |
| US9888994B2 (en) * | 2012-05-15 | 2018-02-13 | Transverse Medical, Inc. | Catheter-based apparatuses and methods |
| EP2732794A1 (en) * | 2012-11-14 | 2014-05-21 | Contego AB | Improved embolic protection device and method |
| CN103349577B (zh) * | 2012-11-30 | 2015-05-06 | 宁波健世生物科技有限公司 | 带远端保护的经皮主动脉支架或主动脉瓣膜支架系统 |
-
2015
- 2015-01-09 JP JP2016545785A patent/JP6761345B2/ja active Active
- 2015-01-09 CN CN201910661478.1A patent/CN110215312A/zh active Pending
- 2015-01-09 US US15/110,764 patent/US20160324621A1/en not_active Abandoned
- 2015-01-09 EP EP15717961.5A patent/EP3091937B1/en active Active
- 2015-01-09 CN CN201580013235.7A patent/CN106102654B/zh active Active
- 2015-01-09 WO PCT/IB2015/000410 patent/WO2015104645A2/en active Application Filing
-
2020
- 2020-06-18 JP JP2020105495A patent/JP6978548B2/ja active Active
-
2023
- 2023-08-11 US US18/448,839 patent/US20230414336A1/en active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| CN110215312A (zh) | 2019-09-10 |
| EP3091937B1 (en) | 2024-03-20 |
| CN106102654A (zh) | 2016-11-09 |
| JP6978548B2 (ja) | 2021-12-08 |
| CN106102654B (zh) | 2019-06-28 |
| JP2020163184A (ja) | 2020-10-08 |
| EP3091937A2 (en) | 2016-11-16 |
| JP2017501840A (ja) | 2017-01-19 |
| US20230414336A1 (en) | 2023-12-28 |
| WO2015104645A3 (en) | 2015-11-19 |
| WO2015104645A2 (en) | 2015-07-16 |
| US20160324621A1 (en) | 2016-11-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6978548B2 (ja) | 生体構造非依存性偏向装置 | |
| JP7157131B2 (ja) | 血管内装置および送達システム | |
| JP7191339B2 (ja) | 改善された塞栓防御デバイスおよび方法 | |
| US11020211B2 (en) | Accessory device to provide neuroprotection during interventional procedures | |
| CN105392432B (zh) | 具有压力和超声波特征的远端保护系统和方法 | |
| US10973618B2 (en) | Embolic protection device | |
| US9480548B2 (en) | Embolic protection device and method of use | |
| JP6232000B2 (ja) | 遠位保護フィルタ | |
| JP5896575B2 (ja) | 血管造影用カテーテル | |
| US9089406B2 (en) | Embolic filter devices, systems, and methods for capturing emboli during medical procedures | |
| US20160151141A1 (en) | High Flow Embolic Protection Device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| RD01 | Notification of change of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7426 Effective date: 20161215 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20161215 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20171212 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20171212 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20180912 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20180925 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20181219 |
|
| A524 | Written submission of copy of amendment under article 19 pct |
Free format text: JAPANESE INTERMEDIATE CODE: A524 Effective date: 20190322 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7422 Effective date: 20190322 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190401 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20190401 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20190611 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20190910 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20191111 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20191209 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20200218 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20200618 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20200630 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20200818 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20200824 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20200901 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20200904 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6761345 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |