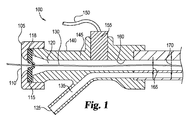
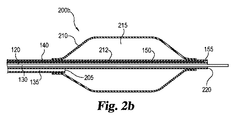
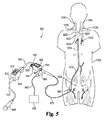
JP6499578B2 - 細胞療法に有用なカテーテルシステム及び方法 - Google Patents
細胞療法に有用なカテーテルシステム及び方法 Download PDFInfo
- Publication number
- JP6499578B2 JP6499578B2 JP2015516167A JP2015516167A JP6499578B2 JP 6499578 B2 JP6499578 B2 JP 6499578B2 JP 2015516167 A JP2015516167 A JP 2015516167A JP 2015516167 A JP2015516167 A JP 2015516167A JP 6499578 B2 JP6499578 B2 JP 6499578B2
- Authority
- JP
- Japan
- Prior art keywords
- catheter
- lumen
- port
- pressure
- catheter shaft
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 238000000034 method Methods 0.000 title description 50
- 238000002659 cell therapy Methods 0.000 title description 7
- 239000012530 fluid Substances 0.000 claims description 60
- 210000004369 blood Anatomy 0.000 claims description 14
- 239000008280 blood Substances 0.000 claims description 14
- 210000004204 blood vessel Anatomy 0.000 claims description 14
- 238000004891 communication Methods 0.000 claims description 13
- 238000001802 infusion Methods 0.000 claims description 9
- 239000007788 liquid Substances 0.000 claims description 7
- 238000007789 sealing Methods 0.000 claims description 4
- 210000004027 cell Anatomy 0.000 description 59
- 239000003814 drug Substances 0.000 description 58
- 229940124597 therapeutic agent Drugs 0.000 description 55
- 210000003462 vein Anatomy 0.000 description 36
- 210000003748 coronary sinus Anatomy 0.000 description 28
- 239000000463 material Substances 0.000 description 25
- 210000000130 stem cell Anatomy 0.000 description 25
- 210000001519 tissue Anatomy 0.000 description 20
- 210000002216 heart Anatomy 0.000 description 19
- 210000005245 right atrium Anatomy 0.000 description 19
- 230000005540 biological transmission Effects 0.000 description 11
- 238000003780 insertion Methods 0.000 description 11
- 230000037431 insertion Effects 0.000 description 11
- 238000002604 ultrasonography Methods 0.000 description 11
- 238000012544 monitoring process Methods 0.000 description 10
- 239000000126 substance Substances 0.000 description 10
- 238000002560 therapeutic procedure Methods 0.000 description 9
- 210000002620 vena cava superior Anatomy 0.000 description 9
- 210000003191 femoral vein Anatomy 0.000 description 8
- 230000023597 hemostasis Effects 0.000 description 8
- 230000001225 therapeutic effect Effects 0.000 description 8
- 210000001631 vena cava inferior Anatomy 0.000 description 8
- 210000004413 cardiac myocyte Anatomy 0.000 description 7
- 210000003205 muscle Anatomy 0.000 description 7
- 210000004165 myocardium Anatomy 0.000 description 7
- 230000010412 perfusion Effects 0.000 description 7
- 210000000689 upper leg Anatomy 0.000 description 7
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 6
- 239000000306 component Substances 0.000 description 6
- 210000004731 jugular vein Anatomy 0.000 description 6
- 229910052760 oxygen Inorganic materials 0.000 description 6
- 239000001301 oxygen Substances 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 230000008569 process Effects 0.000 description 6
- 210000001321 subclavian vein Anatomy 0.000 description 6
- 210000005166 vasculature Anatomy 0.000 description 6
- 230000000747 cardiac effect Effects 0.000 description 5
- 230000003511 endothelial effect Effects 0.000 description 5
- 210000004013 groin Anatomy 0.000 description 5
- 210000003111 iliac vein Anatomy 0.000 description 5
- 239000003550 marker Substances 0.000 description 5
- 210000000056 organ Anatomy 0.000 description 5
- 230000035515 penetration Effects 0.000 description 5
- 239000012779 reinforcing material Substances 0.000 description 5
- 206010061216 Infarction Diseases 0.000 description 4
- 210000001789 adipocyte Anatomy 0.000 description 4
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- 238000003384 imaging method Methods 0.000 description 4
- 230000001965 increasing effect Effects 0.000 description 4
- 230000007574 infarction Effects 0.000 description 4
- 210000004072 lung Anatomy 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 241001465754 Metazoa Species 0.000 description 3
- 238000004026 adhesive bonding Methods 0.000 description 3
- 210000001367 artery Anatomy 0.000 description 3
- 210000003129 brachiocephalic vein Anatomy 0.000 description 3
- 238000010276 construction Methods 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 210000002889 endothelial cell Anatomy 0.000 description 3
- 239000000945 filler Substances 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 210000004185 liver Anatomy 0.000 description 3
- 238000002595 magnetic resonance imaging Methods 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 230000002787 reinforcement Effects 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 230000002792 vascular Effects 0.000 description 3
- 238000012800 visualization Methods 0.000 description 3
- 101000738771 Homo sapiens Receptor-type tyrosine-protein phosphatase C Proteins 0.000 description 2
- 102100037422 Receptor-type tyrosine-protein phosphatase C Human genes 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 210000000709 aorta Anatomy 0.000 description 2
- 230000004872 arterial blood pressure Effects 0.000 description 2
- 238000005452 bending Methods 0.000 description 2
- 229920000249 biocompatible polymer Polymers 0.000 description 2
- 239000012503 blood component Substances 0.000 description 2
- 238000009530 blood pressure measurement Methods 0.000 description 2
- 210000000988 bone and bone Anatomy 0.000 description 2
- 210000002449 bone cell Anatomy 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 210000001612 chondrocyte Anatomy 0.000 description 2
- 230000001332 colony forming effect Effects 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 239000002872 contrast media Substances 0.000 description 2
- 238000013480 data collection Methods 0.000 description 2
- 230000001066 destructive effect Effects 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 210000002919 epithelial cell Anatomy 0.000 description 2
- 230000009969 flowable effect Effects 0.000 description 2
- 238000002594 fluoroscopy Methods 0.000 description 2
- 210000004392 genitalia Anatomy 0.000 description 2
- 239000003102 growth factor Substances 0.000 description 2
- 210000000936 intestine Anatomy 0.000 description 2
- 210000003734 kidney Anatomy 0.000 description 2
- 210000005246 left atrium Anatomy 0.000 description 2
- 210000005240 left ventricle Anatomy 0.000 description 2
- 239000004973 liquid crystal related substance Substances 0.000 description 2
- 239000004005 microsphere Substances 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 230000002107 myocardial effect Effects 0.000 description 2
- 208000010125 myocardial infarction Diseases 0.000 description 2
- 210000002569 neuron Anatomy 0.000 description 2
- 210000000496 pancreas Anatomy 0.000 description 2
- 239000008188 pellet Substances 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- 108090000765 processed proteins & peptides Proteins 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 230000001172 regenerating effect Effects 0.000 description 2
- 230000003014 reinforcing effect Effects 0.000 description 2
- 210000005241 right ventricle Anatomy 0.000 description 2
- 210000002027 skeletal muscle Anatomy 0.000 description 2
- 210000000952 spleen Anatomy 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 238000013519 translation Methods 0.000 description 2
- 230000014616 translation Effects 0.000 description 2
- 208000030090 Acute Disease Diseases 0.000 description 1
- 102100021569 Apoptosis regulator Bcl-2 Human genes 0.000 description 1
- 208000031104 Arterial Occlusive disease Diseases 0.000 description 1
- 102100024153 Cadherin-15 Human genes 0.000 description 1
- 208000017667 Chronic Disease Diseases 0.000 description 1
- 108020004414 DNA Proteins 0.000 description 1
- 102100036912 Desmin Human genes 0.000 description 1
- 108010044052 Desmin Proteins 0.000 description 1
- 102100031573 Hematopoietic progenitor cell antigen CD34 Human genes 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000971171 Homo sapiens Apoptosis regulator Bcl-2 Proteins 0.000 description 1
- 101000777663 Homo sapiens Hematopoietic progenitor cell antigen CD34 Proteins 0.000 description 1
- 101000946889 Homo sapiens Monocyte differentiation antigen CD14 Proteins 0.000 description 1
- 108010018562 M-cadherin Proteins 0.000 description 1
- 102100035877 Monocyte differentiation antigen CD14 Human genes 0.000 description 1
- 102000004364 Myogenin Human genes 0.000 description 1
- 108010056785 Myogenin Proteins 0.000 description 1
- 102100024616 Platelet endothelial cell adhesion molecule Human genes 0.000 description 1
- 102000016971 Proto-Oncogene Proteins c-kit Human genes 0.000 description 1
- 108010014608 Proto-Oncogene Proteins c-kit Proteins 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 108010017842 Telomerase Proteins 0.000 description 1
- 101001023030 Toxoplasma gondii Myosin-D Proteins 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 108010022164 acetyl-LDL Proteins 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 210000000577 adipose tissue Anatomy 0.000 description 1
- 210000004504 adult stem cell Anatomy 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 238000002583 angiography Methods 0.000 description 1
- 239000000427 antigen Substances 0.000 description 1
- 102000036639 antigens Human genes 0.000 description 1
- 108091007433 antigens Proteins 0.000 description 1
- 230000001640 apoptogenic effect Effects 0.000 description 1
- 230000008321 arterial blood flow Effects 0.000 description 1
- 208000021328 arterial occlusion Diseases 0.000 description 1
- 210000002565 arteriole Anatomy 0.000 description 1
- 230000001174 ascending effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 210000000013 bile duct Anatomy 0.000 description 1
- 210000003969 blast cell Anatomy 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 230000015624 blood vessel development Effects 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 210000004271 bone marrow stromal cell Anatomy 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- 210000001217 buttock Anatomy 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 230000002648 chondrogenic effect Effects 0.000 description 1
- 238000010968 computed tomography angiography Methods 0.000 description 1
- 210000004351 coronary vessel Anatomy 0.000 description 1
- 210000000028 corpus adiposum pararenale Anatomy 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 210000000805 cytoplasm Anatomy 0.000 description 1
- 238000007405 data analysis Methods 0.000 description 1
- 210000005045 desmin Anatomy 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 230000002526 effect on cardiovascular system Effects 0.000 description 1
- 210000001671 embryonic stem cell Anatomy 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 201000010063 epididymitis Diseases 0.000 description 1
- 210000004700 fetal blood Anatomy 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 210000002837 heart atrium Anatomy 0.000 description 1
- 208000019622 heart disease Diseases 0.000 description 1
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 1
- 230000002439 hemostatic effect Effects 0.000 description 1
- 210000003897 hepatic stem cell Anatomy 0.000 description 1
- 210000003494 hepatocyte Anatomy 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 210000004153 islets of langerhan Anatomy 0.000 description 1
- 210000001865 kupffer cell Anatomy 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 210000005265 lung cell Anatomy 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000013507 mapping Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 238000013160 medical therapy Methods 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 210000000663 muscle cell Anatomy 0.000 description 1
- 210000001665 muscle stem cell Anatomy 0.000 description 1
- 210000003098 myoblast Anatomy 0.000 description 1
- 210000000107 myocyte Anatomy 0.000 description 1
- 230000001338 necrotic effect Effects 0.000 description 1
- 210000001178 neural stem cell Anatomy 0.000 description 1
- 210000001428 peripheral nervous system Anatomy 0.000 description 1
- 239000002831 pharmacologic agent Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 210000001778 pluripotent stem cell Anatomy 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 210000000229 preadipocyte Anatomy 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 210000002363 skeletal muscle cell Anatomy 0.000 description 1
- 210000004927 skin cell Anatomy 0.000 description 1
- 210000001057 smooth muscle myoblast Anatomy 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 210000000352 storage cell Anatomy 0.000 description 1
- 210000002536 stromal cell Anatomy 0.000 description 1
- 210000004003 subcutaneous fat Anatomy 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- -1 treatment areas Substances 0.000 description 1
- 238000012285 ultrasound imaging Methods 0.000 description 1
- 210000004509 vascular smooth muscle cell Anatomy 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M25/1011—Multiple balloon catheters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B34/00—Computer-aided surgery; Manipulators or robots specially adapted for use in surgery
- A61B34/20—Surgical navigation systems; Devices for tracking or guiding surgical instruments, e.g. for frameless stereotaxis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0021—Catheters; Hollow probes characterised by the form of the tubing
- A61M25/0023—Catheters; Hollow probes characterised by the form of the tubing by the form of the lumen, e.g. cross-section, variable diameter
- A61M25/0026—Multi-lumen catheters with stationary elements
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0021—Catheters; Hollow probes characterised by the form of the tubing
- A61M25/0041—Catheters; Hollow probes characterised by the form of the tubing pre-formed, e.g. specially adapted to fit with the anatomy of body channels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0067—Catheters; Hollow probes characterised by the distal end, e.g. tips
- A61M25/0068—Static characteristics of the catheter tip, e.g. shape, atraumatic tip, curved tip or tip structure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0067—Catheters; Hollow probes characterised by the distal end, e.g. tips
- A61M25/0068—Static characteristics of the catheter tip, e.g. shape, atraumatic tip, curved tip or tip structure
- A61M25/007—Side holes, e.g. their profiles or arrangements; Provisions to keep side holes unblocked
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/0105—Steering means as part of the catheter or advancing means; Markers for positioning
- A61M25/0108—Steering means as part of the catheter or advancing means; Markers for positioning using radio-opaque or ultrasound markers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M25/1018—Balloon inflating or inflation-control devices
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M37/00—Other apparatus for introducing media into the body; Percutany, i.e. introducing medicines into the body by diffusion through the skin
- A61M37/0092—Other apparatus for introducing media into the body; Percutany, i.e. introducing medicines into the body by diffusion through the skin using ultrasonic, sonic or infrasonic vibrations, e.g. phonophoresis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M2025/0001—Catheters; Hollow probes for pressure measurement
- A61M2025/0003—Catheters; Hollow probes for pressure measurement having an additional lumen transmitting fluid pressure to the outside for measurement
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M2025/0004—Catheters; Hollow probes having two or more concentrically arranged tubes for forming a concentric catheter system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0021—Catheters; Hollow probes characterised by the form of the tubing
- A61M2025/0042—Microcatheters, cannula or the like having outside diameters around 1 mm or less
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/01—Introducing, guiding, advancing, emplacing or holding catheters
- A61M25/09—Guide wires
- A61M2025/091—Guide wires having a lumen for drug delivery or suction
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M25/1011—Multiple balloon catheters
- A61M2025/1015—Multiple balloon catheters having two or more independently movable balloons where the distance between the balloons can be adjusted, e.g. two balloon catheters concentric to each other forming an adjustable multiple balloon catheter system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M2025/1043—Balloon catheters with special features or adapted for special applications
- A61M2025/1052—Balloon catheters with special features or adapted for special applications for temporarily occluding a vessel for isolating a sector
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M2025/1043—Balloon catheters with special features or adapted for special applications
- A61M2025/1061—Balloon catheters with special features or adapted for special applications having separate inflations tubes, e.g. coaxial tubes or tubes otherwise arranged apart from the catheter tube
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/10—Balloon catheters
- A61M2025/1043—Balloon catheters with special features or adapted for special applications
- A61M2025/1093—Balloon catheters with special features or adapted for special applications having particular tip characteristics
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Engineering & Computer Science (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Biomedical Technology (AREA)
- General Health & Medical Sciences (AREA)
- Anesthesiology (AREA)
- Hematology (AREA)
- Pulmonology (AREA)
- Biophysics (AREA)
- Child & Adolescent Psychology (AREA)
- Medical Informatics (AREA)
- Dermatology (AREA)
- Surgery (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Robotics (AREA)
- Molecular Biology (AREA)
- Media Introduction/Drainage Providing Device (AREA)
- Infusion, Injection, And Reservoir Apparatuses (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201261655976P | 2012-06-05 | 2012-06-05 | |
| US61/655,976 | 2012-06-05 | ||
| PCT/US2013/044287 WO2013184782A2 (en) | 2012-06-05 | 2013-06-05 | Catheter systems and methods useful for cell therapy |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2015523884A JP2015523884A (ja) | 2015-08-20 |
| JP2015523884A5 JP2015523884A5 (enExample) | 2016-07-21 |
| JP6499578B2 true JP6499578B2 (ja) | 2019-04-10 |
Family
ID=48746641
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2015516167A Expired - Fee Related JP6499578B2 (ja) | 2012-06-05 | 2013-06-05 | 細胞療法に有用なカテーテルシステム及び方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (3) | US9604037B2 (enExample) |
| EP (2) | EP3597254A1 (enExample) |
| JP (1) | JP6499578B2 (enExample) |
| KR (1) | KR20150016970A (enExample) |
| CA (2) | CA2875516C (enExample) |
| WO (1) | WO2013184782A2 (enExample) |
Families Citing this family (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10155099B2 (en) | 2009-09-21 | 2018-12-18 | Cook Regentec Llc | Method for infusing stem cells |
| US9539081B2 (en) | 2009-12-02 | 2017-01-10 | Surefire Medical, Inc. | Method of operating a microvalve protection device |
| WO2014025255A1 (en) * | 2012-08-06 | 2014-02-13 | Wellinq Medical B.V. | Pressure sensor catheter and associated method |
| CA2923419A1 (en) * | 2013-09-09 | 2015-03-12 | Pryor Medical Devices, Inc. | Low-profile occlusion catheter |
| US9968740B2 (en) | 2014-03-25 | 2018-05-15 | Surefire Medical, Inc. | Closed tip dynamic microvalve protection device |
| WO2015179710A1 (en) * | 2014-05-22 | 2015-11-26 | Conkwest, Inc. | Treating solid tumours with nk-92 cells applied by microcatheter |
| KR102557862B1 (ko) | 2015-01-09 | 2023-07-20 | 애커러트 메디컬 테라퓨틱스 엘티디. | 색전술 마이크로카테터 |
| WO2016149653A2 (en) | 2015-03-19 | 2016-09-22 | Prytime Medical Devices, Inc. | System and method for low-profile occlusion balloon catheter |
| US20160287839A1 (en) | 2015-03-31 | 2016-10-06 | Surefire Medical, Inc. | Apparatus and Method for Infusing an Immunotherapy Agent to a Solid Tumor for Treatment |
| EP3322348A1 (en) * | 2015-07-14 | 2018-05-23 | Boston Scientific Scimed Inc. | Gel delivery catheters, systems, and methods |
| US12268824B2 (en) | 2018-07-27 | 2025-04-08 | Embolx, Inc. | Shaped catheter tip for tracking over a guidewire through turns in the vasculature |
| US10350382B1 (en) * | 2018-06-08 | 2019-07-16 | Embolx, Inc. | High torque catheter and methods of manufacture |
| CN107343980A (zh) * | 2016-04-21 | 2017-11-14 | 青岛智辰生物科技有限公司 | 一种多腔道球囊导管 |
| CA3021780C (en) | 2016-05-04 | 2020-09-08 | Accurate Medical Therapeutics Ltd. | Embolization microcatheter head having slitted pattern |
| CA2990479C (en) | 2016-06-02 | 2019-03-26 | Prytime Medical Devices, Inc. | System and method for low-profile occlusion balloon catheter |
| US11400263B1 (en) | 2016-09-19 | 2022-08-02 | Trisalus Life Sciences, Inc. | System and method for selective pressure-controlled therapeutic delivery |
| US10780250B1 (en) | 2016-09-19 | 2020-09-22 | Surefire Medical, Inc. | System and method for selective pressure-controlled therapeutic delivery |
| US20200197682A1 (en) * | 2016-10-17 | 2020-06-25 | Prytime Medical Devices, Inc. | Access device with guidewire and related methods |
| WO2018132623A1 (en) | 2017-01-12 | 2018-07-19 | The Regents Of The University Of California | Endovascular perfusion augmentation for critical care |
| WO2018164907A1 (en) | 2017-03-10 | 2018-09-13 | Promedica Health System, Inc. | Aortic valve no exchange catheter |
| US10588636B2 (en) | 2017-03-20 | 2020-03-17 | Surefire Medical, Inc. | Dynamic reconfigurable microvalve protection device |
| CN110769749B (zh) | 2017-04-21 | 2023-05-09 | 加利福尼亚大学董事会 | 用于部分主动脉闭塞的主动脉流量计和泵 |
| US11185245B2 (en) | 2017-06-03 | 2021-11-30 | Sentinel Medical Technologies, Llc. | Catheter for monitoring pressure for muscle compartment syndrome |
| US11045128B2 (en) | 2017-06-03 | 2021-06-29 | Sentinel Medical Technologies, LLC | Catheter for monitoring intra-abdominal pressure |
| US11045143B2 (en) | 2017-06-03 | 2021-06-29 | Sentinel Medical Technologies, LLC | Catheter with connectable hub for monitoring pressure |
| KR102870709B1 (ko) | 2017-11-02 | 2025-10-16 | 애커러트 메디컬 테라퓨틱스 엘티디. | 필터가 내장된 색전술용 마이크로카테터 |
| EP3533484A1 (en) * | 2018-03-01 | 2019-09-04 | Mehmet Ali Özer | Controlled pressure reperfusion catheter |
| US12097345B2 (en) | 2018-06-01 | 2024-09-24 | Penumbra, Inc. | Infusion catheter and methods of use |
| EP3801730A4 (en) * | 2018-06-01 | 2022-03-30 | Penumbra, Inc. | Venous infusion catheter and methods for its use |
| US10960177B2 (en) | 2018-06-29 | 2021-03-30 | Promedica Health System, Inc. | Aortic valve no exchange catheter and methods of using the same |
| US11850398B2 (en) | 2018-08-01 | 2023-12-26 | Trisalus Life Sciences, Inc. | Systems and methods for pressure-facilitated therapeutic agent delivery |
| EP3833273A4 (en) | 2018-08-06 | 2022-06-29 | Prytime Medical Devices, Inc. | System and method for low profile occlusion balloon catheter |
| US11338117B2 (en) | 2018-10-08 | 2022-05-24 | Trisalus Life Sciences, Inc. | Implantable dual pathway therapeutic agent delivery port |
| WO2020081835A1 (en) * | 2018-10-17 | 2020-04-23 | University Of Kentucky Research Foundation | Percutaneous pulmonary artery drainage device |
| US11672457B2 (en) | 2018-11-24 | 2023-06-13 | Sentinel Medical Technologies, Llc. | Catheter for monitoring pressure |
| US11779263B2 (en) | 2019-02-08 | 2023-10-10 | Sentinel Medical Technologies, Llc. | Catheter for monitoring intra-abdominal pressure for assessing preeclampsia |
| CN114173687A (zh) | 2019-05-23 | 2022-03-11 | 阿克瑞特医学治疗有限公司 | 用于微球的无回流递送的栓塞导管 |
| US12433597B2 (en) | 2019-06-04 | 2025-10-07 | Trisalus Life Sciences, Inc. | Atraumatic occlusive system with compartment for measurement of vascular pressure change |
| EP4009860B1 (en) | 2019-08-08 | 2024-07-24 | Sentinel Medical Technologies, LLC | Cable for use with pressure monitoring catheters |
| EP4017568A4 (en) | 2019-08-20 | 2023-10-18 | Embolx, Inc. | CATHETERS AND METHODS OF MANUFACTURE AND USE |
| US11617543B2 (en) | 2019-12-30 | 2023-04-04 | Sentinel Medical Technologies, Llc. | Catheter for monitoring pressure |
| AU2021239935A1 (en) | 2020-03-16 | 2022-10-06 | Certus Critical Care, Inc. | Blood flow control devices, systems, and methods and error detection thereof |
| CN111419304A (zh) * | 2020-04-16 | 2020-07-17 | 上海科赐医疗技术有限公司 | 弯曲球囊导管牵开器 |
| EP4084703B1 (en) | 2021-03-18 | 2024-03-13 | Prytime Medical Devices, Inc. | Vascular occlusion catheter |
| WO2023278594A1 (en) | 2021-07-02 | 2023-01-05 | Embolx, Inc. | Catheters adapted for agent delivery |
| US20240188974A1 (en) * | 2022-12-12 | 2024-06-13 | Boston Scientific Scimed, Inc. | Medical device for ultrasound-assisted drug delivery |
| CN115844481B (zh) * | 2023-02-15 | 2023-06-02 | 乐普(北京)医疗器械股份有限公司 | 一种冠状窦脉冲球囊导管及其控制方法 |
| US20240366309A1 (en) * | 2023-05-01 | 2024-11-07 | Mg Stroke Analytics Inc. | System and methods for navigation of catheters within vessels |
| WO2025014589A1 (en) * | 2023-07-13 | 2025-01-16 | Becton, Dickinson And Company | Catheter system having a fluid chamber proximate a pressure transducer |
| EP4501387A1 (en) * | 2023-07-31 | 2025-02-05 | Albert-Ludwigs-Universität Freiburg | A dual catheter arrangement and system for reperfusion of an ischemic tissue region via a coronary vessel |
| WO2025136869A1 (en) * | 2023-12-21 | 2025-06-26 | Medtronic Vascular, Inc. | Local drug delivery to lesion treatment site in blood vessel |
Family Cites Families (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59131364A (ja) * | 1983-01-18 | 1984-07-28 | テルモ株式会社 | 弁体付カテ−テル |
| US4848344A (en) * | 1987-11-13 | 1989-07-18 | Cook, Inc. | Balloon guide |
| US5328470A (en) | 1989-03-31 | 1994-07-12 | The Regents Of The University Of Michigan | Treatment of diseases by site-specific instillation of cells or site-specific transformation of cells and kits therefor |
| US5706809A (en) * | 1993-01-29 | 1998-01-13 | Cardima, Inc. | Method and system for using multiple intravascular sensing devices to detect electrical activity |
| GB9317539D0 (en) | 1993-08-24 | 1993-10-06 | Shiu Man F | Catheter |
| US5478309A (en) * | 1994-05-27 | 1995-12-26 | William P. Sweezer, Jr. | Catheter system and method for providing cardiopulmonary bypass pump support during heart surgery |
| US5573007A (en) * | 1994-08-08 | 1996-11-12 | Innerspace, Inc. | Gas column pressure monitoring catheters |
| US5518205A (en) | 1994-09-06 | 1996-05-21 | Rockwell International Corporation | High altitude, long duration surveillance system |
| US5858003A (en) | 1994-10-20 | 1999-01-12 | Children's Medical Center Corporation | Systems and methods for promoting tissue growth |
| US5707358A (en) * | 1996-05-13 | 1998-01-13 | Wright; John T. M. | Dual concentric balloon catheter for retrograde cardioplegia perfusion |
| US6132397A (en) * | 1997-05-01 | 2000-10-17 | Chase Medical Inc. | Integral aortic arch infusion clamp catheter |
| DE69833665T2 (de) * | 1997-08-08 | 2006-11-09 | Duke University | Zusammensetzungen zur vereinfachung von chirurgischen verfahren |
| US6866842B1 (en) | 1998-05-01 | 2005-03-15 | University Of Pittsburgh | Muscle-derived cells (MDCs) for treating muscle-or bone-related injury or dysfunction |
| US6777231B1 (en) | 1999-03-10 | 2004-08-17 | The Regents Of The University Of California | Adipose-derived stem cells and lattices |
| JP2000300524A (ja) * | 1999-04-16 | 2000-10-31 | Aisin Seiki Co Ltd | バルーンカテーテル |
| AU4848200A (en) * | 1999-05-14 | 2000-12-05 | Salient Interventional Systems, Inc. | Intravascular device and methods of manufacture and use |
| CN1433325A (zh) | 1999-11-05 | 2003-07-30 | 加利福尼亚大学董事会 | 通过体内基因递送治疗心血管疾病的技术和组合物 |
| US6500145B1 (en) * | 2000-02-08 | 2002-12-31 | California Medical Laboratories, Inc. | Retrograde cardioplegia catheter |
| US20040009940A1 (en) | 2000-10-20 | 2004-01-15 | Coleman Michael E. | Gene delivery formulations and methods for treatment of ischemic conditions |
| US6616597B2 (en) * | 2000-12-12 | 2003-09-09 | Datascope Investment Corp. | Intra-aortic balloon catheter having a dual sensor pressure sensing system |
| US7155417B1 (en) | 2001-06-05 | 2006-12-26 | Intervoice Limited Partnership | System and method for detecting fraud in prepaid accounts |
| US6805860B1 (en) | 2001-09-30 | 2004-10-19 | Eckhard Alt | Method of transluminal application of myogenic cells for repair or replacement of heart tissue |
| US7452532B2 (en) | 2001-09-30 | 2008-11-18 | Scicotec Gmbh | Transluminal application of adult stem cells for body organ tissue repair |
| CA2463415C (en) * | 2001-10-25 | 2012-02-07 | Emory University | Catheter for modified perfusion |
| US7595043B2 (en) | 2001-12-07 | 2009-09-29 | Cytori Therapeutics, Inc. | Method for processing and using adipose-derived stem cells |
| WO2005011794A1 (ja) * | 2003-07-30 | 2005-02-10 | Zeon Corporation | 大動脈内バルーンカテーテル |
| US20050059930A1 (en) * | 2003-09-16 | 2005-03-17 | Michi Garrison | Method and apparatus for localized drug delivery |
| US20050059931A1 (en) * | 2003-09-16 | 2005-03-17 | Venomatrix | Methods and apparatus for localized and semi-localized drug delivery |
| US20050266556A1 (en) | 2004-02-09 | 2005-12-01 | Yoder Mervin C | Isolation, expansion and use of clonogenic endothelial progenitor cells |
| US8100883B1 (en) * | 2004-08-11 | 2012-01-24 | Cardiac Pacemakers, Inc. | Right-side coronary sinus lead delivery catheter |
| US7819856B2 (en) | 2004-10-05 | 2010-10-26 | Nexeon Medical Systems, Inc. | Methods and apparatus for treating infarcted regions of tissue following acute myocardial infarction |
| US8142468B2 (en) * | 2005-12-22 | 2012-03-27 | Cordis Corporation | Guidewire with distal expansion feature and method for enhancing the deliverability and crossability of medical devices |
| US7959055B2 (en) * | 2006-04-05 | 2011-06-14 | Senju Metal Industry Co., Ltd. | Wave soldering tank |
| JP4942462B2 (ja) * | 2006-11-22 | 2012-05-30 | テルモ株式会社 | 弁体および医療器具 |
| US10123803B2 (en) * | 2007-10-17 | 2018-11-13 | Covidien Lp | Methods of managing neurovascular obstructions |
| US20110105960A1 (en) * | 2009-10-06 | 2011-05-05 | Wallace Michael P | Ultrasound-enhanced Stenosis therapy |
| US20120215099A1 (en) * | 2009-10-06 | 2012-08-23 | Wallace Michael P | Methods and Apparatus for Endovascular Ultrasound Delivery |
| US20110270131A1 (en) * | 2010-04-28 | 2011-11-03 | Allergan, Inc. | Method and system for determining the pressure of a fluid in a syringe, an access port, a catheter, and a gastric band |
| US11337707B2 (en) * | 2010-05-25 | 2022-05-24 | Miracor Medical Sa | Treating heart tissue |
| EP2389974B1 (en) * | 2010-05-25 | 2014-04-23 | Miracor Medical Systems GmbH | A balloon catheter for introduction into a body vessel, in particular the coronary sinus |
-
2013
- 2013-06-05 CA CA2875516A patent/CA2875516C/en not_active Expired - Fee Related
- 2013-06-05 EP EP19189191.0A patent/EP3597254A1/en not_active Withdrawn
- 2013-06-05 CA CA3092408A patent/CA3092408A1/en not_active Abandoned
- 2013-06-05 JP JP2015516167A patent/JP6499578B2/ja not_active Expired - Fee Related
- 2013-06-05 WO PCT/US2013/044287 patent/WO2013184782A2/en not_active Ceased
- 2013-06-05 US US13/910,693 patent/US9604037B2/en active Active
- 2013-06-05 KR KR1020147035679A patent/KR20150016970A/ko not_active Abandoned
- 2013-06-05 EP EP13734222.6A patent/EP2854923B1/en active Active
-
2017
- 2017-03-08 US US15/453,194 patent/US10532193B2/en active Active
-
2020
- 2020-01-14 US US16/742,061 patent/US20200147354A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| WO2013184782A3 (en) | 2014-01-30 |
| US20130338637A1 (en) | 2013-12-19 |
| US20200147354A1 (en) | 2020-05-14 |
| CA3092408A1 (en) | 2013-12-12 |
| US20170173309A1 (en) | 2017-06-22 |
| JP2015523884A (ja) | 2015-08-20 |
| KR20150016970A (ko) | 2015-02-13 |
| EP2854923A2 (en) | 2015-04-08 |
| EP2854923B1 (en) | 2019-08-21 |
| US10532193B2 (en) | 2020-01-14 |
| US9604037B2 (en) | 2017-03-28 |
| EP3597254A1 (en) | 2020-01-22 |
| CA2875516A1 (en) | 2013-12-12 |
| CA2875516C (en) | 2020-11-03 |
| WO2013184782A2 (en) | 2013-12-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6499578B2 (ja) | 細胞療法に有用なカテーテルシステム及び方法 | |
| US6165164A (en) | Catheter for injecting therapeutic and diagnostic agents | |
| CA2273467C (en) | Catheter for injecting therapeutic and diagnostic agents | |
| US7416547B2 (en) | Injection catheter | |
| US6905476B2 (en) | Catheter with injection needle | |
| US6623474B1 (en) | Injection catheter with needle stop | |
| JP6559681B2 (ja) | 血管内圧を評価するためのデバイス、システム及び方法 | |
| US20090177183A1 (en) | Cell delivery catheters with distal tip high fidelity sensors | |
| US7905864B2 (en) | Injection catheter with multi-directional delivery injection needle | |
| RU2442617C2 (ru) | Конвекционный доставляющий катетер со съемным элементом жесткости и способ его применения | |
| US6540725B1 (en) | Injection catheter with controllably extendable injection needle | |
| WO2017098198A1 (en) | Microcatheter apparatus | |
| JP2011517582A (ja) | 光ファイバ検出構成要素を有するガイドワイヤ及び送達カテーテル、並びに関連システム及び方法 | |
| CN107854763A (zh) | 可自主控制的拉线注射导管、包括所述导管的机器人系统及其操作方法 | |
| US20100226903A1 (en) | Method of delivering cell therapy to a target site | |
| CN204352347U (zh) | 分段式柔性渐变血管微导管 | |
| CN204446908U (zh) | 血管微导管 | |
| CN204352348U (zh) | 一体式柔性渐变血管微导管 | |
| CN204352346U (zh) | 带有编织缠丝加强层的一体式柔性渐变血管微导管 | |
| CN206350868U (zh) | 一种具有血管内压力测量功能的导引导管 | |
| US10285648B2 (en) | Multifunctional, electrophysiological diagnostic catheter for treatments in electrocardiology | |
| CN204446909U (zh) | 具有可变柔性的血管微导管 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20160602 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20160602 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20170530 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170829 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20180130 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20180501 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20180904 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190104 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20190115 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20190226 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20190315 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6499578 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| LAPS | Cancellation because of no payment of annual fees |