JP5679458B2 - 内臓脂肪重量の低下剤 - Google Patents
内臓脂肪重量の低下剤 Download PDFInfo
- Publication number
- JP5679458B2 JP5679458B2 JP2011533064A JP2011533064A JP5679458B2 JP 5679458 B2 JP5679458 B2 JP 5679458B2 JP 2011533064 A JP2011533064 A JP 2011533064A JP 2011533064 A JP2011533064 A JP 2011533064A JP 5679458 B2 JP5679458 B2 JP 5679458B2
- Authority
- JP
- Japan
- Prior art keywords
- group
- compound
- fgf21
- fat weight
- visceral fat
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 210000001596 intra-abdominal fat Anatomy 0.000 title claims description 25
- 229940124024 weight reducing agent Drugs 0.000 title claims description 5
- 239000013585 weight reducing agent Substances 0.000 title claims description 4
- 150000001875 compounds Chemical class 0.000 claims description 46
- -1 Benzoxazol-2-yl Chemical group 0.000 claims description 39
- 108090000376 Fibroblast growth factor 21 Proteins 0.000 claims description 33
- 102000003973 Fibroblast growth factor 21 Human genes 0.000 claims description 33
- 150000003839 salts Chemical class 0.000 claims description 32
- 239000012453 solvate Substances 0.000 claims description 30
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 claims description 24
- 238000004519 manufacturing process Methods 0.000 claims description 16
- 125000004202 aminomethyl group Chemical group [H]N([H])C([H])([H])* 0.000 claims description 12
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims description 12
- 239000004480 active ingredient Substances 0.000 claims description 8
- 230000001603 reducing effect Effects 0.000 claims description 7
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 6
- 230000001737 promoting effect Effects 0.000 claims description 4
- 210000001835 viscera Anatomy 0.000 claims description 3
- 239000008194 pharmaceutical composition Substances 0.000 claims 2
- 208000008589 Obesity Diseases 0.000 description 25
- 235000020824 obesity Nutrition 0.000 description 24
- 229960002297 fenofibrate Drugs 0.000 description 23
- YMTINGFKWWXKFG-UHFFFAOYSA-N fenofibrate Chemical compound C1=CC(OC(C)(C)C(=O)OC(C)C)=CC=C1C(=O)C1=CC=C(Cl)C=C1 YMTINGFKWWXKFG-UHFFFAOYSA-N 0.000 description 23
- 229940126062 Compound A Drugs 0.000 description 21
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 21
- 238000000034 method Methods 0.000 description 15
- 239000003814 drug Substances 0.000 description 14
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 14
- 230000000694 effects Effects 0.000 description 13
- 241000700159 Rattus Species 0.000 description 11
- 230000009471 action Effects 0.000 description 11
- 208000001145 Metabolic Syndrome Diseases 0.000 description 10
- 201000000690 abdominal obesity-metabolic syndrome Diseases 0.000 description 10
- CZMRCDWAGMRECN-UHFFFAOYSA-N 2-{[3,4-dihydroxy-2,5-bis(hydroxymethyl)oxolan-2-yl]oxy}-6-(hydroxymethyl)oxane-3,4,5-triol Chemical compound OCC1OC(CO)(OC2OC(CO)C(O)C(O)C2O)C(O)C1O CZMRCDWAGMRECN-UHFFFAOYSA-N 0.000 description 9
- 102000023984 PPAR alpha Human genes 0.000 description 9
- 108091008725 peroxisome proliferator-activated receptors alpha Proteins 0.000 description 9
- 229940079593 drug Drugs 0.000 description 8
- 201000010063 epididymitis Diseases 0.000 description 8
- 241001465754 Metazoa Species 0.000 description 7
- 238000002360 preparation method Methods 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- 239000007864 aqueous solution Substances 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 6
- 241000699670 Mus sp. Species 0.000 description 5
- 230000003247 decreasing effect Effects 0.000 description 5
- 206010012601 diabetes mellitus Diseases 0.000 description 5
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 5
- 125000004430 oxygen atom Chemical group O* 0.000 description 5
- 230000002265 prevention Effects 0.000 description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- 102000003728 Peroxisome Proliferator-Activated Receptors Human genes 0.000 description 4
- 108090000029 Peroxisome Proliferator-Activated Receptors Proteins 0.000 description 4
- 239000000556 agonist Substances 0.000 description 4
- 230000004069 differentiation Effects 0.000 description 4
- 229920000609 methyl cellulose Polymers 0.000 description 4
- 239000001923 methylcellulose Substances 0.000 description 4
- 108090000623 proteins and genes Proteins 0.000 description 4
- 230000002829 reductive effect Effects 0.000 description 4
- 229940124597 therapeutic agent Drugs 0.000 description 4
- 230000009278 visceral effect Effects 0.000 description 4
- 125000004769 (C1-C4) alkylsulfonyl group Chemical group 0.000 description 3
- 241000252212 Danio rerio Species 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 108010013563 Lipoprotein Lipase Proteins 0.000 description 3
- 102000043296 Lipoprotein lipases Human genes 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 238000009395 breeding Methods 0.000 description 3
- 230000001488 breeding effect Effects 0.000 description 3
- 210000003743 erythrocyte Anatomy 0.000 description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 3
- 229940125753 fibrate Drugs 0.000 description 3
- 230000037356 lipid metabolism Effects 0.000 description 3
- 210000004698 lymphocyte Anatomy 0.000 description 3
- 239000013642 negative control Substances 0.000 description 3
- 230000003449 preventive effect Effects 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 2
- 125000004768 (C1-C4) alkylsulfinyl group Chemical group 0.000 description 2
- 102000000452 Acetyl-CoA carboxylase Human genes 0.000 description 2
- 108010016219 Acetyl-CoA carboxylase Proteins 0.000 description 2
- 108010018763 Biotin carboxylase Proteins 0.000 description 2
- 102000018233 Fibroblast Growth Factor Human genes 0.000 description 2
- 108050007372 Fibroblast Growth Factor Proteins 0.000 description 2
- 101000895303 Homo sapiens Cyclic AMP-responsive element-binding protein 3-like protein 3 Proteins 0.000 description 2
- 101100120063 Homo sapiens FGF21 gene Proteins 0.000 description 2
- 101000846529 Homo sapiens Fibroblast growth factor 21 Proteins 0.000 description 2
- 208000031226 Hyperlipidaemia Diseases 0.000 description 2
- 206010021033 Hypomenorrhoea Diseases 0.000 description 2
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 210000000577 adipose tissue Anatomy 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 2
- 125000003282 alkyl amino group Chemical group 0.000 description 2
- 125000005196 alkyl carbonyloxy group Chemical group 0.000 description 2
- 125000005278 alkyl sulfonyloxy group Chemical group 0.000 description 2
- 125000004414 alkyl thio group Chemical group 0.000 description 2
- 125000005530 alkylenedioxy group Chemical group 0.000 description 2
- 125000004397 aminosulfonyl group Chemical group NS(=O)(=O)* 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 2
- 125000006297 carbonyl amino group Chemical group [H]N([*:2])C([*:1])=O 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 2
- 235000014113 dietary fatty acids Nutrition 0.000 description 2
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 239000000194 fatty acid Substances 0.000 description 2
- 229930195729 fatty acid Natural products 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 230000037406 food intake Effects 0.000 description 2
- 235000012631 food intake Nutrition 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- 125000002816 methylsulfanyl group Chemical group [H]C([H])([H])S[*] 0.000 description 2
- 125000006216 methylsulfinyl group Chemical group [H]C([H])([H])S(*)=O 0.000 description 2
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 2
- 238000003672 processing method Methods 0.000 description 2
- 230000000069 prophylactic effect Effects 0.000 description 2
- 102000005962 receptors Human genes 0.000 description 2
- 108020003175 receptors Proteins 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 125000006296 sulfonyl amino group Chemical group [H]N(*)S(*)(=O)=O 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 125000004434 sulfur atom Chemical group 0.000 description 2
- 230000001629 suppression Effects 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 239000008399 tap water Substances 0.000 description 2
- 235000020679 tap water Nutrition 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- 238000013518 transcription Methods 0.000 description 2
- 230000035897 transcription Effects 0.000 description 2
- 238000011144 upstream manufacturing Methods 0.000 description 2
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical compound C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- 102000030169 Apolipoprotein C-III Human genes 0.000 description 1
- 108010056301 Apolipoprotein C-III Proteins 0.000 description 1
- 206010003210 Arteriosclerosis Diseases 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 102000002666 Carnitine O-palmitoyltransferase Human genes 0.000 description 1
- 108010018424 Carnitine O-palmitoyltransferase Proteins 0.000 description 1
- 241000725101 Clea Species 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 102100021306 Cyclic AMP-responsive element-binding protein 3-like protein 3 Human genes 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- 208000002249 Diabetes Complications Diseases 0.000 description 1
- 208000007342 Diabetic Nephropathies Diseases 0.000 description 1
- 206010012655 Diabetic complications Diseases 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- 238000008157 ELISA kit Methods 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- HEMJJKBWTPKOJG-UHFFFAOYSA-N Gemfibrozil Chemical compound CC1=CC=C(C)C(OCCCC(C)(C)C(O)=O)=C1 HEMJJKBWTPKOJG-UHFFFAOYSA-N 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 102000001554 Hemoglobins Human genes 0.000 description 1
- 108010054147 Hemoglobins Proteins 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 208000035150 Hypercholesterolemia Diseases 0.000 description 1
- 206010020710 Hyperphagia Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 206010022489 Insulin Resistance Diseases 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 206010067482 No adverse event Diseases 0.000 description 1
- 108020005497 Nuclear hormone receptor Proteins 0.000 description 1
- 229920002565 Polyethylene Glycol 400 Polymers 0.000 description 1
- 108091027981 Response element Proteins 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 238000010162 Tukey test Methods 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 150000001242 acetic acid derivatives Chemical class 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 150000001413 amino acids Chemical group 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000019552 anatomical structure morphogenesis Effects 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 208000011775 arteriosclerosis disease Diseases 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 210000003651 basophil Anatomy 0.000 description 1
- 229960000516 bezafibrate Drugs 0.000 description 1
- IIBYAHWJQTYFKB-UHFFFAOYSA-N bezafibrate Chemical compound C1=CC(OC(C)(C)C(O)=O)=CC=C1CCNC(=O)C1=CC=C(Cl)C=C1 IIBYAHWJQTYFKB-UHFFFAOYSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 229940084030 carboxymethylcellulose calcium Drugs 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000019522 cellular metabolic process Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920006184 cellulose methylcellulose Polymers 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- 235000012000 cholesterol Nutrition 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 210000004443 dendritic cell Anatomy 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 208000033679 diabetic kidney disease Diseases 0.000 description 1
- 125000001664 diethylamino group Chemical group [H]C([H])([H])C([H])([H])N(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000008034 disappearance Effects 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 210000003979 eosinophil Anatomy 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000006125 ethylsulfonyl group Chemical group 0.000 description 1
- 125000004705 ethylthio group Chemical group C(C)S* 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 229960003627 gemfibrozil Drugs 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 208000019622 heart disease Diseases 0.000 description 1
- 208000014951 hematologic disease Diseases 0.000 description 1
- 208000018706 hematopoietic system disease Diseases 0.000 description 1
- 210000003494 hepatocyte Anatomy 0.000 description 1
- 102000056713 human FGF21 Human genes 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 201000001421 hyperglycemia Diseases 0.000 description 1
- 230000001227 hypertriglyceridemic effect Effects 0.000 description 1
- 208000026278 immune system disease Diseases 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 125000003253 isopropoxy group Chemical group [H]C([H])([H])C([H])(O*)C([H])([H])[H] 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 238000002350 laparotomy Methods 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 210000003593 megakaryocyte Anatomy 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- 125000003506 n-propoxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 210000000822 natural killer cell Anatomy 0.000 description 1
- 230000017074 necrotic cell death Effects 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 102000006255 nuclear receptors Human genes 0.000 description 1
- 108020004017 nuclear receptors Proteins 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- JLFNLZLINWHATN-UHFFFAOYSA-N pentaethylene glycol Chemical compound OCCOCCOCCOCCOCCO JLFNLZLINWHATN-UHFFFAOYSA-N 0.000 description 1
- 239000003614 peroxisome proliferator Substances 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- LCPDWSOZIOUXRV-UHFFFAOYSA-N phenoxyacetic acid Chemical class OC(=O)COC1=CC=CC=C1 LCPDWSOZIOUXRV-UHFFFAOYSA-N 0.000 description 1
- 208000022530 polyphagia Diseases 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 230000002250 progressing effect Effects 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 229940043274 prophylactic drug Drugs 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 238000011552 rat model Methods 0.000 description 1
- 230000000384 rearing effect Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 238000007619 statistical method Methods 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 230000002381 testicular Effects 0.000 description 1
- 229940126585 therapeutic drug Drugs 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 125000005270 trialkylamine group Chemical group 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 230000005740 tumor formation Effects 0.000 description 1
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 1
- 238000012762 unpaired Student’s t-test Methods 0.000 description 1
- 150000004670 unsaturated fatty acids Chemical class 0.000 description 1
- 235000021122 unsaturated fatty acids Nutrition 0.000 description 1
- 230000004584 weight gain Effects 0.000 description 1
- 235000019786 weight gain Nutrition 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/42—Oxazoles
- A61K31/423—Oxazoles condensed with carbocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/54—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings condensed with carbocyclic rings or ring systems
- C07D231/56—Benzopyrazoles; Hydrogenated benzopyrazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D263/00—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings
- C07D263/52—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings condensed with carbocyclic rings or ring systems
- C07D263/54—Benzoxazoles; Hydrogenated benzoxazoles
- C07D263/58—Benzoxazoles; Hydrogenated benzoxazoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/60—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings condensed with carbocyclic rings or ring systems
- C07D277/62—Benzothiazoles
- C07D277/68—Benzothiazoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached in position 2
- C07D277/82—Nitrogen atoms
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Diabetes (AREA)
- Obesity (AREA)
- Hematology (AREA)
- Epidemiology (AREA)
- Urology & Nephrology (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Child & Adolescent Psychology (AREA)
- Endocrinology (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Vascular Medicine (AREA)
- Emergency Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Heterocyclic Compounds That Contain Two Or More Ring Oxygen Atoms (AREA)
Description
PPARαアゴニストとしては、不飽和脂肪酸やフィブラート系薬剤、例えばフェノフィブラート、ベザフィブラート、ゲムフィブロジル等の薬剤が知られている(非特許文献2)。PPARαアゴニストは、脂肪酸のβ酸化に関与するアセチル−CoAカルボキシラーゼ(ACC)やカルニチンパルミトイルトランスフェラーゼ1(CPT−1)を活性化することも報告されている。
従って、FGF21は、造血幹細胞の赤血球−骨髄球系列細胞(例えば、赤血球、巨核球、好酸球、好中球、好塩基球、単球、樹状細胞)への分化、あるいは造血幹細胞のリンパ球系列細胞(例えば、T細胞、B細胞、NK細胞)への分化を制御し得ると考えられる。また、FGF21の発現又は機能を調節する物質は、造血幹細胞の赤血球−骨髄球系列細胞又はリンパ球系列細胞への分化調節能を有し、血液細胞の異常に起因する疾患(例えば、造血疾患、免疫疾患、アレルギー疾患)に対する医薬あるいは研究用試薬の開発などに有用であることが報告されている(特許文献1)。
これまでに血中のFGF21濃度を著明に増加させる方法としては、FGF21タンパク質の注射(非特許文献10)、FGF21遺伝子の導入(非特許文献11)あるいはFGF21の発現を促進するもうひとつの転写因子CREB−Hを遺伝子導入により活性化する方法(非特許文献12)が報告されているのみである。しかも、これらの方法は、安全性が確立されていない。特に、FGF21遺伝子の導入やCREB−H遺伝子の導入には、ベクターを用いる必要があり、もしヒトに用いた場合には、高い確率で副作用が発現すると考えられる。
で表される化合物、若しくはその塩又はこれらの溶媒和物を有効成分とするFGF21産生促進剤を提供するものである。
また、本発明は、前記一般式(1)で表される化合物、若しくはその塩又はこれらの溶媒和物を有効成分とする、内臓脂肪重量の低下剤を提供するものである。
また、本発明は、FGF21産生促進のための、前記一般式(1)で表される化合物、若しくはその塩又はこれらの溶媒和物を提供するものである。
また、本発明は、内臓脂肪重量の低下のための、前記一般式(1)で表される化合物、若しくはその塩又はこれらの溶媒和物を提供するものである。
また、本発明は、肥満、より詳細には、内臓脂肪型肥満の予防及び/又は治療のための、前記一般式(1)で表される化合物、若しくはその塩又はこれらの溶媒和物を提供するものである。
さらに、本発明は、メタボリックシンドロームの予防及び/又は治療のための、前記一般式(1)で表される化合物、若しくはその塩又はこれらの溶媒和物を提供するものである。
本試験には、前記特許文献2に実施例14として開示されている化合物の光学活性体である、(R)−2−[3−[[N−(ベンズオキサゾール−2−イル)−N−3−(4−メトキシフェノキシ)プロピル]アミノメチル]フェノキシ]酪酸(以下、化合物Aと表記する)を使用した。化合物A及びフェノフィブラートをZucker fatty ラットに投与したときの血漿中トリグリセリド(TG)、血漿中FGF21濃度、及び精巣上体脂肪重量に対する効果を、次法に従って測定した。
多食による肥満を伴い高脂血症を呈するZucker fatty ラット(Crlj:ZUC−Lepr<fa> Genotype:fa/fa)及び肥満を呈さないZucker lean ラット(Crlj:ZUC−Lepr<fa> Genotype:fa/+ or +/+)を日本チャールス・リバー株式会社より購入し、8週齢で供試した。
実験期間を通じて、明暗サイクル(室内光による明るい期間:午前7時〜午後7時)、温度23±3℃、湿度55±15%に維持された飼育室で飼育し、固形飼料(CE−2;オリエンタル酵母工業(株))及び水道水を自由摂取させた。
化合物A及びフェノフィブラートのそれぞれを、メチルセルロース(メトローズ(商標登録)、SM−400、信越化学工業(株))の0.5質量%水溶液に懸濁し、水溶液の投与量が2mL/kgになるように調製した。懸濁液は遮光ビンにて冷蔵(4℃)保存し、調製は7日ごとに行った。
ラットを体重、血漿中総コレステロール濃度及びトリグリセライド濃度が平均化されるように、以下の4群(各群6匹)、すなわち、第一群として対照群、第二群として化合物A(1mg/kg)投与群、第三群として化合物A(3mg/kg)投与群、第四群としてフェノフィブラート(300mg/kg)投与群に群分けした。また、陰性対照群としてzucker lean rat(5匹)を用いた。
各薬剤は、1日1回(午前中)に14日間反復経口投与した。第一群の対照群には、メチルセルロース0.5質量%水溶液2mL/kgを経口投与した。
投与開始から14日目に、投与より4時間絶食した後、採血を行い、血漿中のトリグリセライド濃度を酵素法により、FGF21濃度をELISA法(Human FGF−21 ELISA Kit,Biovender社)にて測定した。採血の翌日、麻酔下に開腹後、精巣脂肪上体脂肪を摘出し、その重量を測定した。なお、精巣上体脂肪重量は、内臓脂肪の全体重量と高く相関していることから、薬剤の内臓脂肪重量を評価するための指標として使用される(FASEB J.20,1203−1205 (2006))。
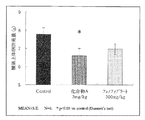
結果は、平均値±標準誤差で示した。対照群とZucker leanラットとの比較はunpaired Student’s t−testで行い、対照群と薬物投与群との比較はDunnettの多重比較検定またはTukeyの多重比較検定で行い、危険率5%未満を有意差ありと判定した。
図1に血漿中トリグリセライドの濃度を示す。横軸は左側から第一群の対照群、第二群の化合物A(1mg/kg)投与群、第三群の化合物A(3mg/kg)投与群、第四群のフェノフィブラート(300mg/kg)投与群、及び陰性対照群(zucker lean rat)である。図1中の*印はp<0.05、#はp<0.001で有意差があることを示している。
その結果、血漿中のトリグリセライド濃度は、第三群では対照群に比べて大幅な低下が認められ、危険率p<0.05で明らかな血漿トリグリセライド低下作用を示した。一方、第四群では僅かに低下傾向を示すに過ぎなかった。
その結果、血漿中のFGF21濃度は、第三群では対照群に比べて大幅な上昇が認められ、危険率p<0.001で明らかな血漿FGF21産生促進作用を示した。一方、第四群では対照群に比して僅かに上昇傾向を示すに過ぎなかった(P=0.2)。
以上のデータが示すように、本モデル動物において化合物Aは血漿中のTGを低下させ、新規の糖・脂質代謝改善因子であるFGF21の発現を著しく増大させる作用を有することが確認され、化合物Aは高度な脂質代謝異常を有する病態でも血漿中のFGF21濃度を増大させて脂質代謝を改善できる可能性が示唆された。
以上のことから、化合物AがFGF21を著明に増加させ、なおかつ、その結果として、内臓脂肪重量を強力に低下させることが確認された。さらに、これらの効果が既存のフィブラート、すなわちPPARαアゴニストで最も優れているフェノフィブラートと比較しても、明らかに強力であり、高い安全性を示すことが確認された。
本試験には、前記化合物A、及び前記特許文献2に実施例229として開示されている化合物の光学活性体である、(R)−2−[3−[[N−(5−フルオロベンズオキサゾール−2−イル)−N−2−フェノキシエチル]アミノメチル]フェノキシ]酪酸(以下、化合物Bと表記する)を使用した。化合物A、化合物B及びフェノフィブラートをKK−Ayマウスに投与したときの精巣上体脂肪重量、腸間膜脂肪重量に対する効果を、次法に従って測定した。
方法:雄性KK−Ayマウス(KK−Ay/TaJcl)を日本クレアより購入し、10週齢で供試した。マウスに化合物を4週間、1日1回経口投与した。実験期間を通じて、明暗サイクル(室内光による明るい期間:午前7時〜午後7時)、温度23±3℃、湿度55±15%に維持された飼育室で飼育し、固形飼料(CE−2;オリエンタル酵母工業(株))及び水道水を自由摂取させた。
化合物A、B及びフェノフィブラートのそれぞれを、メチルセルロース(メトローズ(商標登録)、SM−400、信越化学工業(株))の0.5質量%水溶液に懸濁し、水溶液の投与量が5mL/kgになるように調製した。懸濁液は遮光ビンにて冷蔵(4℃)保存し、調製は7日ごとに行った。
動物を、以下の4群(各群6匹)、すなわち、第一群として対照群、第二群として化合物A(1mg/kg)投与群、第三群として化合物B(3mg/kg)投与群、第四群としてフェノフィブラート(300mg/kg)投与群に群分けした。
各薬剤は、1日1回(午前中)に35日間反復経口投与した。第一群の対照群には、メチルセルロース0.5質量%水溶液5mL/kgを経口投与した。
投与終了後に、精巣上体脂肪及び腸間膜脂肪を摘出し、重量を測定した。
結果は、対照群における脂肪重量に対する各化合物投与群における脂肪重量の低下率について、平均値で示した。
表1に結果を示す。化合物A(1mg/kg)投与群及び化合物B(3mg/kg)投与群は、フェノフィブラート(300mg/kg)投与群よりも強力な脂肪重量の低下作用を有していた。なお、化合物A及び化合物Bの投与期間中に特に有害事象は確認されなかった。
Claims (4)
- (R)−2−[3−[[N−(ベンズオキサゾール−2−イル)−N−3−(4−メトキシフェノキシ)プロピル]アミノメチル]フェノキシ]酪酸、(R)−2−[3−[[N−(5−フルオロベンズオキサゾール−2−イル)−N−2−フェノキシエチル]アミノメチル]フェノキシ]酪酸、これらの塩及びこれらの溶媒和物から選ばれる化合物を有効成分とするFGF21産生促進剤。
- (R)−2−[3−[[N−(ベンズオキサゾール−2−イル)−N−3−(4−メトキシフェノキシ)プロピル]アミノメチル]フェノキシ]酪酸、(R)−2−[3−[[N−(5−フルオロベンズオキサゾール−2−イル)−N−2−フェノキシエチル]アミノメチル]フェノキシ]酪酸、これらの塩及びこれらの溶媒和物から選ばれる化合物を有効成分とする内臓脂肪重量の低下剤。
- (R)−2−[3−[[N−(ベンズオキサゾール−2−イル)−N−3−(4−メトキシフェノキシ)プロピル]アミノメチル]フェノキシ]酪酸、(R)−2−[3−[[N−(5−フルオロベンズオキサゾール−2−イル)−N−2−フェノキシエチル]アミノメチル]フェノキシ]酪酸、これらの塩及びこれらの溶媒和物から選ばれる化合物、並びに薬学的に許容される担体を含有してなるFGF21産生促進用医薬組成物。
- (R)−2−[3−[[N−(ベンズオキサゾール−2−イル)−N−3−(4−メトキシフェノキシ)プロピル]アミノメチル]フェノキシ]酪酸、(R)−2−[3−[[N−(5−フルオロベンズオキサゾール−2−イル)−N−2−フェノキシエチル]アミノメチル]フェノキシ]酪酸、これらの塩及びこれらの溶媒和物から選ばれる化合物、並びに薬学的に許容される担体を含有してなる内臓脂肪重量の低下用医薬組成物。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011533064A JP5679458B2 (ja) | 2009-09-28 | 2010-09-27 | 内臓脂肪重量の低下剤 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009222853 | 2009-09-28 | ||
| JP2009222853 | 2009-09-28 | ||
| PCT/JP2010/066678 WO2011037223A1 (ja) | 2009-09-28 | 2010-09-27 | 内臓脂肪重量の低下剤 |
| JP2011533064A JP5679458B2 (ja) | 2009-09-28 | 2010-09-27 | 内臓脂肪重量の低下剤 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JPWO2011037223A1 JPWO2011037223A1 (ja) | 2013-02-21 |
| JP5679458B2 true JP5679458B2 (ja) | 2015-03-04 |
Family
ID=43795964
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2011533064A Active JP5679458B2 (ja) | 2009-09-28 | 2010-09-27 | 内臓脂肪重量の低下剤 |
Country Status (5)
| Country | Link |
|---|---|
| US (9) | US20120165377A1 (ja) |
| EP (1) | EP2484361B1 (ja) |
| JP (1) | JP5679458B2 (ja) |
| ES (1) | ES2559238T3 (ja) |
| WO (1) | WO2011037223A1 (ja) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NZ701573A (en) | 2012-05-17 | 2017-04-28 | Extend Biosciences Inc | Carriers for improved drug delivery |
| TWI696462B (zh) * | 2013-07-10 | 2020-06-21 | 日商興和股份有限公司 | 非酒精性脂肪性肝疾病治療劑 |
| US10293026B2 (en) | 2014-09-08 | 2019-05-21 | Osaka University | Agent for preventing or treating demyelinating disease |
| US9585934B2 (en) | 2014-10-22 | 2017-03-07 | Extend Biosciences, Inc. | Therapeutic vitamin D conjugates |
| US9616109B2 (en) | 2014-10-22 | 2017-04-11 | Extend Biosciences, Inc. | Insulin vitamin D conjugates |
| US9789197B2 (en) | 2014-10-22 | 2017-10-17 | Extend Biosciences, Inc. | RNAi vitamin D conjugates |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005023777A1 (ja) * | 2003-09-03 | 2005-03-17 | Kowa Co., Ltd. | Ppar活性化化合物及びこれを含有する医薬組成物 |
| WO2008120472A1 (ja) * | 2007-03-29 | 2008-10-09 | Kowa Company, Ltd. | 高脂血症の予防及び/又は治療剤 |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9606805D0 (en) * | 1996-03-30 | 1996-06-05 | Glaxo Wellcome Inc | Medicaments |
| AU718500B2 (en) * | 1997-01-23 | 2000-04-13 | Sumitomo Pharmaceuticals Company, Limited | Remedies for diabetes |
| AU2002347985A1 (en) | 2001-11-15 | 2003-06-10 | Eli Lilly And Company | Peroxisome proliferator activated receptor alpha agonists |
| US6987118B2 (en) | 2003-05-21 | 2006-01-17 | Pfizer Inc. | Tetrahydroisoquinoline derivatives as PPAR-alpha activators |
| JPWO2005009942A1 (ja) | 2003-07-28 | 2006-09-07 | 杏林製薬株式会社 | 光学活性置換フェニルプロピオン酸誘導体 |
| DE102004016845A1 (de) | 2004-04-07 | 2005-10-27 | Bayer Healthcare Ag | Phenylthioessigsäure-Derivate und ihre Verwendung |
| CA2581072A1 (en) | 2004-09-16 | 2006-03-30 | Merck & Co., Inc. | Compounds for the treatment of dyslipidemia and other lipid disorders |
| US20070190079A1 (en) * | 2004-10-29 | 2007-08-16 | Kalypsys, Inc. | Methods for the selective modulation of ppar |
| TWI359810B (en) | 2004-11-04 | 2012-03-11 | Mitsubishi Tanabe Pharma Corp | Carboxylic acid derivative containing thiazole rin |
| JP2006246823A (ja) | 2005-03-11 | 2006-09-21 | Kyoto Univ | 造血因子としてのFgf21の使用 |
| US7795291B2 (en) | 2006-07-07 | 2010-09-14 | Bristol-Myers Squibb Company | Substituted acid derivatives useful as anti-atherosclerotic, anti-dyslipidemic, anti-diabetic and anti-obesity agents and method |
| WO2009047240A1 (en) | 2007-10-09 | 2009-04-16 | Smithkline Beecham Corporation | Indole derivatives useful as ppar activators |
| DE102007061757A1 (de) | 2007-12-20 | 2009-06-25 | Bayer Healthcare Ag | Substituierte 2-Phenylpyrimidin-5-carbonsäuren und ihre Verwendung |
-
2010
- 2010-09-27 ES ES10818895.4T patent/ES2559238T3/es active Active
- 2010-09-27 WO PCT/JP2010/066678 patent/WO2011037223A1/ja active Application Filing
- 2010-09-27 EP EP10818895.4A patent/EP2484361B1/en active Active
- 2010-09-27 US US13/393,164 patent/US20120165377A1/en not_active Abandoned
- 2010-09-27 JP JP2011533064A patent/JP5679458B2/ja active Active
-
2016
- 2016-05-03 US US15/145,090 patent/US20160243086A1/en not_active Abandoned
-
2017
- 2017-10-18 US US15/787,049 patent/US20180078530A1/en not_active Abandoned
-
2019
- 2019-02-07 US US16/270,193 patent/US20190167644A1/en not_active Abandoned
- 2019-10-02 US US16/590,458 patent/US20200030299A1/en not_active Abandoned
-
2020
- 2020-04-22 US US16/855,722 patent/US20200246315A1/en not_active Abandoned
-
2021
- 2021-02-26 US US17/186,226 patent/US20210205274A1/en not_active Abandoned
-
2022
- 2022-09-02 US US17/902,119 patent/US20230019803A1/en not_active Abandoned
-
2023
- 2023-05-24 US US18/322,909 patent/US20230321051A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005023777A1 (ja) * | 2003-09-03 | 2005-03-17 | Kowa Co., Ltd. | Ppar活性化化合物及びこれを含有する医薬組成物 |
| WO2008120472A1 (ja) * | 2007-03-29 | 2008-10-09 | Kowa Company, Ltd. | 高脂血症の予防及び/又は治療剤 |
Non-Patent Citations (5)
| Title |
|---|
| JPN6014026822; YAMAZAKI, Y. et al.: 'Design and synthesis of highly potent and selective human peroxisome proliferator-activated receptor' BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, PERGAMON, ELSEVIER SCIENCE Vol.17, No.16, 2007, p.4689-4693 * |
| JPN6014026826; LUNDASEN, T. et al.: 'PPARalpha is a key regulator of hepatic FGF2' BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS Vol.360, No.2, 2007, p.437-440 * |
| JPN7014001945; BADMAN, M. K. et al.: 'Hepatic Fibroblast Growth Factor 21 Is Regulatedby PPARalpha and Is a Key Mediatorof Hepatic Lipid Meta' CELL METABOLISM Vol.5, 2007, p.426-437 * |
| JPN7014001946; GALMAN, C. et al.: 'The Circulating Metabolic RegulatorFGF21 Is Induced by Prolonged Fastingand PPARalpha Activation in Man' CELL METABOLISM Vol.8, 2008, p.169-174 * |
| JPN7014001947; INAGAKI, T. et al.: 'Endocrine Regulation of the Fasting Responseby PPARalpha-Mediated Inductionof Fibroblast Growth Factor' CELL METABOLISM Vol.5, 2007, p.415-425 * |
Also Published As
| Publication number | Publication date |
|---|---|
| US20180078530A1 (en) | 2018-03-22 |
| ES2559238T3 (es) | 2016-02-11 |
| US20210205274A1 (en) | 2021-07-08 |
| US20190167644A1 (en) | 2019-06-06 |
| EP2484361B1 (en) | 2015-11-11 |
| EP2484361A1 (en) | 2012-08-08 |
| US20200246315A1 (en) | 2020-08-06 |
| US20230321051A1 (en) | 2023-10-12 |
| WO2011037223A1 (ja) | 2011-03-31 |
| US20230019803A1 (en) | 2023-01-19 |
| EP2484361A4 (en) | 2013-05-22 |
| JPWO2011037223A1 (ja) | 2013-02-21 |
| US20200030299A1 (en) | 2020-01-30 |
| US20160243086A1 (en) | 2016-08-25 |
| US20120165377A1 (en) | 2012-06-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230321051A1 (en) | Agent for reducing visceral fat weight | |
| JP6391572B2 (ja) | 非アルコール性脂肪性肝疾患治療剤 | |
| JP6238460B2 (ja) | 脂質異常症治療剤 | |
| US8426455B2 (en) | Prophylactic and/or therapeutic agent for hyperlipidemia | |
| WO2018131626A1 (ja) | 非アルコール性脂肪性肝疾患の予防及び治療薬 | |
| US11938122B2 (en) | 5-[[4-[2-[5-(1-hydroxyethyl)pyridin-2-yl]ethoxy]phenyl]methyl]-1,3- thiazolidine-2,4-dione for treating nonalcoholic fatty liver disease | |
| US9968592B2 (en) | Dyslipidemia therapeutic agent | |
| RU2664442C2 (ru) | Способ снижения веса | |
| AU2007257854B2 (en) | Compounds for the treatment of metabolic disorders | |
| US20240316010A1 (en) | Agent for reducing visceral fat weight | |
| WO2016047800A1 (ja) | 脂質異常症治療剤 | |
| JP2016535099A (ja) | 糖尿病及びそれから生じる疾患合併症の治療のための化合物 | |
| KR20160045042A (ko) | 지질 이상증 치료제 | |
| JPWO2017082393A1 (ja) | 緑内障予防治療剤 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130605 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20130605 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140701 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140901 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20141224 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20141226 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5679458 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R153 | Grant of patent term extension |
Free format text: JAPANESE INTERMEDIATE CODE: R153 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R153 | Grant of patent term extension |
Free format text: JAPANESE INTERMEDIATE CODE: R153 |