JP5102377B2 - ガスミスト圧浴システム - Google Patents
ガスミスト圧浴システム Download PDFInfo
- Publication number
- JP5102377B2 JP5102377B2 JP2011010648A JP2011010648A JP5102377B2 JP 5102377 B2 JP5102377 B2 JP 5102377B2 JP 2011010648 A JP2011010648 A JP 2011010648A JP 2011010648 A JP2011010648 A JP 2011010648A JP 5102377 B2 JP5102377 B2 JP 5102377B2
- Authority
- JP
- Japan
- Prior art keywords
- gas
- gas mist
- pressure bath
- living body
- mist
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000003595 mist Substances 0.000 title claims description 233
- 239000007789 gas Substances 0.000 claims description 356
- 239000007788 liquid Substances 0.000 claims description 72
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 claims description 42
- 239000001569 carbon dioxide Substances 0.000 claims description 21
- 229910002092 carbon dioxide Inorganic materials 0.000 claims description 21
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 20
- 229910052760 oxygen Inorganic materials 0.000 claims description 20
- 239000001301 oxygen Substances 0.000 claims description 20
- 238000003860 storage Methods 0.000 claims description 20
- 210000004400 mucous membrane Anatomy 0.000 claims description 17
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 17
- 239000003795 chemical substances by application Substances 0.000 claims description 13
- 230000000694 effects Effects 0.000 claims description 9
- 238000007599 discharging Methods 0.000 claims description 7
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 claims description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 6
- KFSLWBXXFJQRDL-UHFFFAOYSA-N Peracetic acid Chemical compound CC(=O)OO KFSLWBXXFJQRDL-UHFFFAOYSA-N 0.000 claims description 6
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 6
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 claims description 6
- 239000011941 photocatalyst Substances 0.000 claims description 6
- 239000008213 purified water Substances 0.000 claims description 6
- -1 anesthetic Chemical compound 0.000 claims description 5
- NOOLISFMXDJSKH-UTLUCORTSA-N (+)-Neomenthol Chemical compound CC(C)[C@@H]1CC[C@@H](C)C[C@@H]1O NOOLISFMXDJSKH-UTLUCORTSA-N 0.000 claims description 3
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 claims description 3
- FPIPGXGPPPQFEQ-UHFFFAOYSA-N 13-cis retinol Natural products OCC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-UHFFFAOYSA-N 0.000 claims description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 claims description 3
- ACTIUHUUMQJHFO-UHFFFAOYSA-N Coenzym Q10 Natural products COC1=C(OC)C(=O)C(CC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)C)=C(C)C1=O ACTIUHUUMQJHFO-UHFFFAOYSA-N 0.000 claims description 3
- 229920000858 Cyclodextrin Polymers 0.000 claims description 3
- NOOLISFMXDJSKH-UHFFFAOYSA-N DL-menthol Natural products CC(C)C1CCC(C)CC1O NOOLISFMXDJSKH-UHFFFAOYSA-N 0.000 claims description 3
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 claims description 3
- 241000241413 Propolis Species 0.000 claims description 3
- 239000005708 Sodium hypochlorite Substances 0.000 claims description 3
- 241000700605 Viruses Species 0.000 claims description 3
- 229930003427 Vitamin E Natural products 0.000 claims description 3
- 230000003444 anaesthetic effect Effects 0.000 claims description 3
- 239000002260 anti-inflammatory agent Substances 0.000 claims description 3
- 229940121363 anti-inflammatory agent Drugs 0.000 claims description 3
- 239000000043 antiallergic agent Substances 0.000 claims description 3
- 229940121375 antifungal agent Drugs 0.000 claims description 3
- 239000003429 antifungal agent Substances 0.000 claims description 3
- 239000002220 antihypertensive agent Substances 0.000 claims description 3
- 229940030600 antihypertensive agent Drugs 0.000 claims description 3
- 239000002246 antineoplastic agent Substances 0.000 claims description 3
- 229910052586 apatite Inorganic materials 0.000 claims description 3
- 229960000686 benzalkonium chloride Drugs 0.000 claims description 3
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 claims description 3
- 229960003333 chlorhexidine gluconate Drugs 0.000 claims description 3
- YZIYKJHYYHPJIB-UUPCJSQJSA-N chlorhexidine gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O.C1=CC(Cl)=CC=C1NC(=N)NC(=N)NCCCCCCNC(=N)NC(=N)NC1=CC=C(Cl)C=C1 YZIYKJHYYHPJIB-UUPCJSQJSA-N 0.000 claims description 3
- 235000017471 coenzyme Q10 Nutrition 0.000 claims description 3
- 229940110767 coenzyme Q10 Drugs 0.000 claims description 3
- ACTIUHUUMQJHFO-UPTCCGCDSA-N coenzyme Q10 Chemical compound COC1=C(OC)C(=O)C(C\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CCC=C(C)C)=C(C)C1=O ACTIUHUUMQJHFO-UPTCCGCDSA-N 0.000 claims description 3
- 239000002537 cosmetic Substances 0.000 claims description 3
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 claims description 3
- 230000003779 hair growth Effects 0.000 claims description 3
- 229920002674 hyaluronan Polymers 0.000 claims description 3
- 229960003160 hyaluronic acid Drugs 0.000 claims description 3
- 206010022000 influenza Diseases 0.000 claims description 3
- 229960003971 influenza vaccine Drugs 0.000 claims description 3
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 claims description 3
- 229940041616 menthol Drugs 0.000 claims description 3
- 239000002245 particle Substances 0.000 claims description 3
- VSIIXMUUUJUKCM-UHFFFAOYSA-D pentacalcium;fluoride;triphosphate Chemical compound [F-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O VSIIXMUUUJUKCM-UHFFFAOYSA-D 0.000 claims description 3
- 239000002504 physiological saline solution Substances 0.000 claims description 3
- 229940069949 propolis Drugs 0.000 claims description 3
- 229960003471 retinol Drugs 0.000 claims description 3
- 235000020944 retinol Nutrition 0.000 claims description 3
- 239000011607 retinol Substances 0.000 claims description 3
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 claims description 3
- 239000000377 silicon dioxide Substances 0.000 claims description 3
- SUKJFIGYRHOWBL-UHFFFAOYSA-N sodium hypochlorite Chemical compound [Na+].Cl[O-] SUKJFIGYRHOWBL-UHFFFAOYSA-N 0.000 claims description 3
- 229910000031 sodium sesquicarbonate Inorganic materials 0.000 claims description 3
- 235000018341 sodium sesquicarbonate Nutrition 0.000 claims description 3
- 150000003431 steroids Chemical class 0.000 claims description 3
- 239000002562 thickening agent Substances 0.000 claims description 3
- WCTAGTRAWPDFQO-UHFFFAOYSA-K trisodium;hydrogen carbonate;carbonate Chemical compound [Na+].[Na+].[Na+].OC([O-])=O.[O-]C([O-])=O WCTAGTRAWPDFQO-UHFFFAOYSA-K 0.000 claims description 3
- 235000015112 vegetable and seed oil Nutrition 0.000 claims description 3
- 150000003700 vitamin C derivatives Chemical class 0.000 claims description 3
- 235000019165 vitamin E Nutrition 0.000 claims description 3
- 229940046009 vitamin E Drugs 0.000 claims description 3
- 239000011709 vitamin E Substances 0.000 claims description 3
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 claims description 2
- 229960004756 ethanol Drugs 0.000 claims description 2
- 238000005259 measurement Methods 0.000 claims description 2
- 239000003907 antipyretic analgesic agent Substances 0.000 claims 1
- 238000010521 absorption reaction Methods 0.000 description 17
- 238000010586 diagram Methods 0.000 description 13
- 210000003141 lower extremity Anatomy 0.000 description 10
- 239000003814 drug Substances 0.000 description 7
- 238000003825 pressing Methods 0.000 description 7
- 229940079593 drug Drugs 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- 230000000844 anti-bacterial effect Effects 0.000 description 4
- 230000017531 blood circulation Effects 0.000 description 4
- 210000004027 cell Anatomy 0.000 description 4
- 238000010298 pulverizing process Methods 0.000 description 4
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 239000000853 adhesive Substances 0.000 description 3
- 230000001070 adhesive effect Effects 0.000 description 3
- 229940035676 analgesics Drugs 0.000 description 3
- 239000000730 antalgic agent Substances 0.000 description 3
- 230000003110 anti-inflammatory effect Effects 0.000 description 3
- UBAZGMLMVVQSCD-UHFFFAOYSA-N carbon dioxide;molecular oxygen Chemical compound O=O.O=C=O UBAZGMLMVVQSCD-UHFFFAOYSA-N 0.000 description 3
- 229920001971 elastomer Polymers 0.000 description 3
- 210000003743 erythrocyte Anatomy 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 230000002093 peripheral effect Effects 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 239000002699 waste material Substances 0.000 description 3
- 102000001554 Hemoglobins Human genes 0.000 description 2
- 108010054147 Hemoglobins Proteins 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 239000002280 amphoteric surfactant Substances 0.000 description 2
- 230000001754 anti-pyretic effect Effects 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 239000002221 antipyretic Substances 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 230000036772 blood pressure Effects 0.000 description 2
- 210000000245 forearm Anatomy 0.000 description 2
- 230000036039 immunity Effects 0.000 description 2
- 210000003127 knee Anatomy 0.000 description 2
- 210000004877 mucosa Anatomy 0.000 description 2
- 235000019645 odor Nutrition 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 230000035515 penetration Effects 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 230000033764 rhythmic process Effects 0.000 description 2
- 229920002379 silicone rubber Polymers 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N C1CCCCC1 Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 229920001328 Polyvinylidene chloride Polymers 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 229940035674 anesthetics Drugs 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 230000003796 beauty Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- CEJLBZWIKQJOAT-UHFFFAOYSA-N dichloroisocyanuric acid Chemical compound ClN1C(=O)NC(=O)N(Cl)C1=O CEJLBZWIKQJOAT-UHFFFAOYSA-N 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 235000012489 doughnuts Nutrition 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 239000003193 general anesthetic agent Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 230000003780 keratinization Effects 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 238000000034 method Methods 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- 229920003052 natural elastomer Polymers 0.000 description 1
- 229920001194 natural rubber Polymers 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 230000037368 penetrate the skin Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 230000001766 physiological effect Effects 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 229920003225 polyurethane elastomer Polymers 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 239000011118 polyvinyl acetate Substances 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 239000005033 polyvinylidene chloride Substances 0.000 description 1
- 230000010349 pulsation Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 239000004945 silicone rubber Substances 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000001360 synchronised effect Effects 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M37/00—Other apparatus for introducing media into the body; Percutany, i.e. introducing medicines into the body by diffusion through the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H33/00—Bathing devices for special therapeutic or hygienic purposes
- A61H33/02—Bathing devices for use with gas-containing liquid, or liquid in which gas is led or generated, e.g. carbon dioxide baths
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H33/00—Bathing devices for special therapeutic or hygienic purposes
- A61H33/06—Artificial hot-air or cold-air baths; Steam or gas baths or douches, e.g. sauna or Finnish baths
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H33/00—Bathing devices for special therapeutic or hygienic purposes
- A61H33/06—Artificial hot-air or cold-air baths; Steam or gas baths or douches, e.g. sauna or Finnish baths
- A61H33/10—Devices on tubs for steam baths
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H33/00—Bathing devices for special therapeutic or hygienic purposes
- A61H33/14—Devices for gas baths with ozone, hydrogen, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H9/00—Pneumatic or hydraulic massage
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H33/00—Bathing devices for special therapeutic or hygienic purposes
- A61H33/14—Devices for gas baths with ozone, hydrogen, or the like
- A61H2033/143—Devices for gas baths with ozone, hydrogen, or the like with oxygen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H33/00—Bathing devices for special therapeutic or hygienic purposes
- A61H33/14—Devices for gas baths with ozone, hydrogen, or the like
- A61H2033/145—Devices for gas baths with ozone, hydrogen, or the like with CO2
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/02—Characteristics of apparatus not provided for in the preceding codes heated or cooled
- A61H2201/0207—Characteristics of apparatus not provided for in the preceding codes heated or cooled heated
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/10—Characteristics of apparatus not provided for in the preceding codes with further special therapeutic means, e.g. electrotherapy, magneto therapy or radiation therapy, chromo therapy, infrared or ultraviolet therapy
- A61H2201/105—Characteristics of apparatus not provided for in the preceding codes with further special therapeutic means, e.g. electrotherapy, magneto therapy or radiation therapy, chromo therapy, infrared or ultraviolet therapy with means for delivering media, e.g. drugs or cosmetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/16—Physical interface with patient
- A61H2201/1602—Physical interface with patient kind of interface, e.g. head rest, knee support or lumbar support
- A61H2201/1619—Thorax
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/16—Physical interface with patient
- A61H2201/1602—Physical interface with patient kind of interface, e.g. head rest, knee support or lumbar support
- A61H2201/1635—Hand or arm, e.g. handle
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/16—Physical interface with patient
- A61H2201/1602—Physical interface with patient kind of interface, e.g. head rest, knee support or lumbar support
- A61H2201/164—Feet or leg, e.g. pedal
- A61H2201/1642—Holding means therefor
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/16—Physical interface with patient
- A61H2201/1602—Physical interface with patient kind of interface, e.g. head rest, knee support or lumbar support
- A61H2201/165—Wearable interfaces
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/16—Physical interface with patient
- A61H2201/1683—Surface of interface
- A61H2201/1688—Surface of interface disposable
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/50—Control means thereof
- A61H2201/5007—Control means thereof computer controlled
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2201/00—Characteristics of apparatus not provided for in the preceding codes
- A61H2201/50—Control means thereof
- A61H2201/5058—Sensors or detectors
- A61H2201/5082—Temperature sensors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2205/00—Devices for specific parts of the body
- A61H2205/06—Arms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2205/00—Devices for specific parts of the body
- A61H2205/06—Arms
- A61H2205/065—Hands
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2205/00—Devices for specific parts of the body
- A61H2205/08—Trunk
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2205/00—Devices for specific parts of the body
- A61H2205/10—Leg
- A61H2205/106—Leg for the lower legs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2205/00—Devices for specific parts of the body
- A61H2205/10—Leg
- A61H2205/108—Leg for the upper legs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61H—PHYSICAL THERAPY APPARATUS, e.g. DEVICES FOR LOCATING OR STIMULATING REFLEX POINTS IN THE BODY; ARTIFICIAL RESPIRATION; MASSAGE; BATHING DEVICES FOR SPECIAL THERAPEUTIC OR HYGIENIC PURPOSES OR SPECIFIC PARTS OF THE BODY
- A61H2205/00—Devices for specific parts of the body
- A61H2205/12—Feet
Landscapes
- Health & Medical Sciences (AREA)
- Public Health (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Physical Education & Sports Medicine (AREA)
- Rehabilitation Therapy (AREA)
- Pain & Pain Management (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Dermatology (AREA)
- Medical Informatics (AREA)
- Anesthesiology (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Hematology (AREA)
- Devices For Medical Bathing And Washing (AREA)
Description
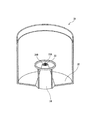
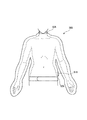
図1は、本発明の第1の実施形態に係るガスミスト圧浴システムの全体概略図である。図1に示すように、本実施形態のガスミスト圧浴システムは、酸素、炭酸ガス、又は酸素と炭酸ガスの混合ガス(以下、単にガスという)を供給するガス供給手段10と、ガスミスト生成手段としてのガスミスト生成器30と、供給されたガスミストを内部に封入する生体カバー手段としての生体圧浴カバー50と、制御手段としての制御装置60と、から構成される。
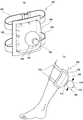
図11は、本発明の第2の実施形態に係るガスミスト圧浴システムの全体概略図である。本実施形態では、生体圧浴カバー内の加圧を容易にする加圧手段をさらに備えたガスミスト圧浴システムについて説明する。なお、図1に示す第1の実施形態と同一の部分については、同一の符号を付して詳細な説明は省略する。
図14は、本発明の第3の実施形態に係るガスミスト圧浴システムの全体概略図である。本実施形態では、発生させたミストを帯電させるための電荷付与手段をさらに備えたガスミスト圧浴システムについて説明する。なお、図1に示す第1の実施形態と同一の部分については、同一の符号を付して詳細な説明は省略する。
21 電源装置
22 電極
30 ガスミスト生成器
31 生成器本体
31A、31B 栓
31C フィルム
32 液体貯留部
33 ノズル
33A 先端開口
34 吸液管形成部材
34A 吸液管
34B 先端部
35 ガス供給手段接続部
35A ガス吐出口
35B 分岐部
35C 流量調整器
36 蓋部
37 ガス導入部
38 バッフル
38A バッフル支持部
39 ガスミスト排出部
41 ガスミスト供給管
50、50A、50B、50C、50D、50E、50F 生体圧浴カバー
51、51A、51B、51C、51D、51E、51F 供給口
52、52A、52B、52C、52D、52E、52F 止着部
53A 止着部
53F 固定部
54F カバー部
60 制御装置
71 圧力計
72 温度計
80、80A、80B、80C、80D 生体圧浴カバー
81、81A、81B、81C、81D 供給口
82、82A、82B、82C、82D 止着部
83D 固定部
84D カバー部
90、90A、90B、90C、90D 加圧手段
91、91A、91B、91C、91D ガス溜部
92、92A、92B、92C、92D 供給口
Claims (19)
- 所定値以上の濃度の炭酸ガス、又は酸素、又は炭酸ガスと酸素の混合ガス(以下、「ガス」という)と液体を粉砕溶解させたミスト(以下、「ガスミスト」という)を生体の皮膚又は粘膜に接触させるためのシステムであって、
ガス供給手段と、
該ガス供給手段との接続部と、液体を供給する液体貯留部と、前記ガスが供給されるノズルと、該ノズル先端に前記液体を送液する吸液管と、前記ノズルからのガス流によって吹き上げられた液体を衝突させる衝突部材と、前記ガスが供給され前記ノズルの上部までガスを導く円筒状のガス導入部と、前記ガスミストを収集し排出するドーナツ形のガスミスト排出部と、前記ガス供給手段からのガスを前記ノズルと前記ガス導入部に分岐させる分岐部と、を有するガスミスト生成手段と、
前記生体の皮膚及び粘膜を覆い、前記ガスミスト生成手段から供給されるガスミストを内部に封入する空間を形成する生体カバー部材と、
を備え、
前記ガスミスト生成手段において、少なくとも前記液体貯留部を取り外し可能にして他の液体貯留部と取替え可能に構成したことを特徴とするガスミスト圧浴システム。 - 前記生体カバー部材内の気圧、温度、酸素濃度、炭酸ガス濃度及び湿度等の測定値を検知するセンサ類と、
該センサ類の測定値に基づき前記生体カバー部材内を予め設定された設定値の範囲内に制御する制御手段と、
をさらに備えることを特徴とする請求項1に記載のガスミスト圧浴システム。 - 前記生体カバー部材内を加圧する加圧手段をさらに備えることを特徴とする請求項1又は2に記載のガスミスト圧浴システム。
- 前記加圧手段が、前記生体カバー部材と連設され、該生体カバー部材内にガスミストを排出可能な中空のガス溜からなることを特徴とする請求項3に記載のガスミスト圧浴システム。
- 前記制御手段により、前記ガスミストを間歇的に前記生体カバー部材内に供給することによって、該生体カバー部材をインターバル加圧することを特徴とする請求項2に記載のガスミスト圧浴システム。
- 前記加圧手段が、前記生体カバー部材内に間歇的にガスミストを排出することによって、該生体カバー部材をインターバル加圧することを特徴とする請求項4に記載のガスミスト圧浴システム。
- 前記ガスミスト生成手段が、外気を取り込むための通気孔を備えることを特徴とする請求項1乃至6の何れか一項に記載のガスミスト圧浴システム。
- 前記液体は、水、イオン水、オゾン水、生理食塩水、精製水、又は滅菌精製水の何れか一つ又は複数の組み合わせであることを特徴とする請求項1乃至7の何れか一項に記載のガスミスト圧浴システム。
- 前記液体は、メンソール、ビタミンE、ビタミンC誘導体、レチノール、麻酔薬、シクロデキストリン、光触媒、光触媒とアパタイトの複合体、ヒアルロン酸、コエンザイムQ10、シードオイル、プロポリス、エタノール、グルコン酸クロルヘキシジン、両性界面活性剤、塩化ベンザルコニウム、酢酸アルキルジアミノエテルグリシン、次亜塩素酸ナトリウム、過酢酸、セスキ炭酸ナトリウム、シリカ、ポピドンヨード、炭酸水素ナトリウム、高濃度炭酸泉剤、抗アレルギー剤、抗炎症剤、解熱鎮痛剤、抗真菌剤、抗インフルエンザウィルス剤、インフルエンザワクチン、ステロイド剤、抗ガン剤、血圧降下剤、化粧剤、増毛剤、発毛剤、又は育毛剤のうち、何れか一つ又は複数をさらに含有することを特徴とする請求項8に記載のガスミスト圧浴システム。
- 前記ガスミスト生成手段から前記生体カバー部材内に供給される前記ミストの粒径が、10μm以下であることを特徴とする請求項1乃至9の何れか一項に記載のガスミスト圧浴システム。
- 前記制御手段は、ガスミスト圧浴時における前記生体カバー部材内の圧力を1.02乃至2.5気圧に保持することを特徴とする請求項2に記載のガスミスト圧浴システム。
- 前記ガスミスト生成手段が供給するミストに電荷を付与する電荷付与手段をさらに備えることを特徴とする請求項1乃至11の何れか一項に記載のガスミスト圧浴システム。
- 前記電荷が、マイナス電荷であることを特徴とする請求項12に記載のガスミスト圧浴システム。
- 前記ガスミスト生成手段が、前記生体圧浴カバーへガスミストを供給するためのガスミスト供給管を有し、
該ガスミスト供給管が、管内に付着する液滴を除去するためのフィルターを備えることを特徴とする請求項1又は2に記載のガスミスト圧浴システム。 - 前記ガスミスト生成手段が、前記生体圧浴カバーへガスミストを供給するためのガスミスト供給管を有し、
該ガスミスト供給管の全部又は一部がジャバラ状の管から構成されることを特徴とする請求項1又は2に記載のガスミスト圧浴システム。 - 前記ガスミスト生成手段が、前記生体圧浴カバーへガスミストを供給するためのガスミスト供給管を有し、
該ガスミスト供給管に、逆止弁を設けることを特徴とする請求項1又は2に記載のガスミスト圧浴システム。 - 前記生体カバー部材のガスミスト供給口に、逆止弁を設けることを特徴とする請求項1又は2に記載のガスミスト圧浴システム。
- 前記制御手段は、前記生体カバー部材内の圧力値が所定値以上になった際は前記ガス供給手段からのガスの供給を停止することを特徴とする請求項1又は2に記載のガスミスト圧浴システム。
- 前記ガスミスト生成手段の取り外し可能部分が、予め滅菌処理されていることを特徴とする請求項1又は2に記載のガスミスト圧浴システム。
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011010648A JP5102377B2 (ja) | 2011-01-21 | 2011-01-21 | ガスミスト圧浴システム |
| PCT/JP2012/051249 WO2012099252A1 (ja) | 2011-01-21 | 2012-01-20 | ガスミスト圧浴システム |
| US13/884,474 US9278204B2 (en) | 2011-01-21 | 2012-01-20 | Pressurized gas mist bathing system |
| KR1020137010524A KR20130141494A (ko) | 2011-01-21 | 2012-01-20 | 가스 미스트 압욕 시스템 |
| EP12737170.6A EP2626053A1 (en) | 2011-01-21 | 2012-01-20 | Pressurized gas mist bathing system |
| CN201280003166.8A CN103140204B (zh) | 2011-01-21 | 2012-01-20 | 加压气雾洗浴系统 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011010648A JP5102377B2 (ja) | 2011-01-21 | 2011-01-21 | ガスミスト圧浴システム |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2012148022A JP2012148022A (ja) | 2012-08-09 |
| JP2012148022A5 JP2012148022A5 (ja) | 2012-10-04 |
| JP5102377B2 true JP5102377B2 (ja) | 2012-12-19 |
Family
ID=46515869
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2011010648A Expired - Fee Related JP5102377B2 (ja) | 2011-01-21 | 2011-01-21 | ガスミスト圧浴システム |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US9278204B2 (ja) |
| EP (1) | EP2626053A1 (ja) |
| JP (1) | JP5102377B2 (ja) |
| KR (1) | KR20130141494A (ja) |
| CN (1) | CN103140204B (ja) |
| WO (1) | WO2012099252A1 (ja) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103338804B (zh) * | 2011-11-04 | 2016-07-06 | 中村正一 | 气雾吸入器 |
| WO2013065777A1 (ja) * | 2011-11-04 | 2013-05-10 | 日本エー・シー・ピー株式会社 | ガスミスト圧浴システム |
| CA3085086C (en) | 2011-12-06 | 2023-08-08 | Delta Faucet Company | Ozone distribution in a faucet |
| CN108463437B (zh) | 2015-12-21 | 2022-07-08 | 德尔塔阀门公司 | 包括消毒装置的流体输送系统 |
| US10682283B2 (en) * | 2016-04-07 | 2020-06-16 | James L. Evavold | Wearable soaking garment |
| CN105748227A (zh) * | 2016-04-27 | 2016-07-13 | 青岛心康电子科技有限公司 | 一种防褥疮氧疗头枕 |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE667501C (de) * | 1936-10-21 | 1938-11-12 | Dr Ernst Schubert | Zerstaeubungsvorrichtung zum Inhalieren von Medikamenten |
| DE2062632A1 (de) * | 1970-12-18 | 1972-06-29 | Paul Ritzau Pari-Werk KG, 8135 Söcking | Inhalationsgerät |
| US4224941A (en) * | 1978-11-15 | 1980-09-30 | Stivala Oscar G | Hyperbaric treatment apparatus |
| US5263476A (en) * | 1991-07-08 | 1993-11-23 | Henson-Thiery Corporation | Enclosure system for burn victims |
| US5516517A (en) * | 1994-05-02 | 1996-05-14 | Exfoliation Cleansing Hydration Oxygenation Corporation | Method for nutritional oxygenation of the skin |
| US5584285A (en) * | 1995-06-07 | 1996-12-17 | Salter Labs | Breathing circuit apparatus for a nebulizer |
| TW317509B (ja) * | 1995-10-31 | 1997-10-11 | Sanyo Electric Co | |
| JP2000245841A (ja) * | 1999-02-26 | 2000-09-12 | Kao Corp | アイケア用噴霧装置 |
| US6432077B1 (en) * | 2000-12-26 | 2002-08-13 | Sensormedics Corporation | Device and method for treatment of surface infections with nitric oxide |
| ES2592274T3 (es) * | 2006-03-30 | 2016-11-29 | Carefusion 2200, Inc. | Nebulizador con control de fluido basado en el flujo |
| JP5023747B2 (ja) * | 2007-03-13 | 2012-09-12 | オムロンヘルスケア株式会社 | ネブライザ用吸入補助具 |
| US20110060257A1 (en) * | 2009-02-06 | 2011-03-10 | Shoichi Nakamura | Gas mist pressure bath system |
| JP5674637B2 (ja) * | 2009-02-26 | 2015-02-25 | 中村 正一 | ガスミスト吸入器 |
| CN201591884U (zh) * | 2009-04-29 | 2010-09-29 | 杭州京泠医疗器械有限公司 | 医用雾化器 |
| CN201603223U (zh) * | 2009-12-12 | 2010-10-13 | 王仁红 | 一种气切用药物雾化装置 |
| CN201701589U (zh) * | 2010-06-02 | 2011-01-12 | 天卓睿丰医疗科技(北京)有限公司 | 呼吸道给药的喷雾器 |
| JP3163837U (ja) * | 2010-08-23 | 2010-11-04 | 中村 正一 | ガスミスト圧浴用カバー |
-
2011
- 2011-01-21 JP JP2011010648A patent/JP5102377B2/ja not_active Expired - Fee Related
-
2012
- 2012-01-20 KR KR1020137010524A patent/KR20130141494A/ko not_active Application Discontinuation
- 2012-01-20 US US13/884,474 patent/US9278204B2/en not_active Expired - Fee Related
- 2012-01-20 EP EP12737170.6A patent/EP2626053A1/en not_active Withdrawn
- 2012-01-20 WO PCT/JP2012/051249 patent/WO2012099252A1/ja active Application Filing
- 2012-01-20 CN CN201280003166.8A patent/CN103140204B/zh not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| JP2012148022A (ja) | 2012-08-09 |
| CN103140204B (zh) | 2016-09-21 |
| EP2626053A1 (en) | 2013-08-14 |
| WO2012099252A1 (ja) | 2012-07-26 |
| US9278204B2 (en) | 2016-03-08 |
| US20130226076A1 (en) | 2013-08-29 |
| CN103140204A (zh) | 2013-06-05 |
| KR20130141494A (ko) | 2013-12-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5088905B2 (ja) | ガスミスト圧浴システム | |
| JP4922459B2 (ja) | ガスミスト圧浴システム | |
| JP5406214B2 (ja) | 炭酸ガスミスト圧浴システム | |
| JP5995373B2 (ja) | ガスミスト圧浴システム | |
| JP5102377B2 (ja) | ガスミスト圧浴システム | |
| JP5088906B2 (ja) | 炭酸ガスミスト圧浴システム | |
| JP5553416B2 (ja) | ガスミスト圧浴システム | |
| JP5474042B2 (ja) | ガスミスト圧浴システム | |
| JP5414927B2 (ja) | ガスミスト圧浴システム | |
| JP5517260B2 (ja) | ガスミスト圧浴システム | |
| JP5305550B2 (ja) | ガスミスト圧浴システム | |
| JP5473942B2 (ja) | ガスミスト圧浴システム | |
| JP5517261B2 (ja) | ガスミスト圧浴システム | |
| JP5743587B2 (ja) | ガスミスト圧浴システム |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120817 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20120817 |
|
| A871 | Explanation of circumstances concerning accelerated examination |
Free format text: JAPANESE INTERMEDIATE CODE: A871 Effective date: 20120817 |
|
| TRDD | Decision of grant or rejection written | ||
| A975 | Report on accelerated examination |
Free format text: JAPANESE INTERMEDIATE CODE: A971005 Effective date: 20120918 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20120925 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20120927 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20151005 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5102377 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20151005 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20221005 Year of fee payment: 10 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |