JP2021519798A - 弾性食感を有する多粒子固体剤形 - Google Patents
弾性食感を有する多粒子固体剤形 Download PDFInfo
- Publication number
- JP2021519798A JP2021519798A JP2020553655A JP2020553655A JP2021519798A JP 2021519798 A JP2021519798 A JP 2021519798A JP 2020553655 A JP2020553655 A JP 2020553655A JP 2020553655 A JP2020553655 A JP 2020553655A JP 2021519798 A JP2021519798 A JP 2021519798A
- Authority
- JP
- Japan
- Prior art keywords
- solid dosage
- dosage form
- mixture
- microcapsules
- particle solid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
- A61K9/1605—Excipients; Inactive ingredients
- A61K9/1629—Organic macromolecular compounds
- A61K9/1652—Polysaccharides, e.g. alginate, cellulose derivatives; Cyclodextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/0056—Mouth soluble or dispersible forms; Suckable, eatable, chewable coherent forms; Forms rapidly disintegrating in the mouth; Lozenges; Lollipops; Bite capsules; Baked products; Baits or other oral forms for animals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2072—Pills, tablets, discs, rods characterised by shape, structure or size; Tablets with holes, special break lines or identification marks; Partially coated tablets; Disintegrating flat shaped forms
- A61K9/2077—Tablets comprising drug-containing microparticles in a substantial amount of supporting matrix; Multiparticulate tablets
- A61K9/2081—Tablets comprising drug-containing microparticles in a substantial amount of supporting matrix; Multiparticulate tablets with microcapsules or coated microparticles according to A61K9/50
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L27/00—Spices; Flavouring agents or condiments; Artificial sweetening agents; Table salts; Dietetic salt substitutes; Preparation or treatment thereof
- A23L27/70—Fixation, conservation, or encapsulation of flavouring agents
- A23L27/72—Encapsulation
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/115—Fatty acids or derivatives thereof; Fats or oils
- A23L33/12—Fatty acids or derivatives thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23P—SHAPING OR WORKING OF FOODSTUFFS, NOT FULLY COVERED BY A SINGLE OTHER SUBCLASS
- A23P10/00—Shaping or working of foodstuffs characterised by the products
- A23P10/30—Encapsulation of particles, e.g. foodstuff additives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/20—Carboxylic acids, e.g. valproic acid having a carboxyl group bound to a chain of seven or more carbon atoms, e.g. stearic, palmitic, arachidic acids
- A61K31/202—Carboxylic acids, e.g. valproic acid having a carboxyl group bound to a chain of seven or more carbon atoms, e.g. stearic, palmitic, arachidic acids having three or more double bonds, e.g. linolenic
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/0056—Mouth soluble or dispersible forms; Suckable, eatable, chewable coherent forms; Forms rapidly disintegrating in the mouth; Lozenges; Lollipops; Bite capsules; Baked products; Baits or other oral forms for animals
- A61K9/0058—Chewing gums
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
- A61K9/1605—Excipients; Inactive ingredients
- A61K9/1629—Organic macromolecular compounds
- A61K9/1641—Organic macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyethylene glycol, poloxamers
- A61K9/1647—Polyesters, e.g. poly(lactide-co-glycolide)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
- A61K9/1682—Processes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/205—Polysaccharides, e.g. alginate, gums; Cyclodextrin
- A61K9/2059—Starch, including chemically or physically modified derivatives; Amylose; Amylopectin; Dextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2072—Pills, tablets, discs, rods characterised by shape, structure or size; Tablets with holes, special break lines or identification marks; Partially coated tablets; Disintegrating flat shaped forms
- A61K9/2077—Tablets comprising drug-containing microparticles in a substantial amount of supporting matrix; Multiparticulate tablets
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5021—Organic macromolecular compounds
- A61K9/5036—Polysaccharides, e.g. gums, alginate; Cyclodextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5021—Organic macromolecular compounds
- A61K9/5052—Proteins, e.g. albumin
- A61K9/5057—Gelatin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/02—Nutrients, e.g. vitamins, minerals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5089—Processes
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nutrition Science (AREA)
- Physiology (AREA)
- Zoology (AREA)
- Food Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Hematology (AREA)
- Mycology (AREA)
- Diabetes (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Obesity (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Medicinal Preparation (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
本発明は、経口投与のための多粒子固体剤形の製造に関する。
国際公開第2013/092589号パンフレットは、経口投与のための多単位ペレット錠剤を開示している。国際公開第2013/092589号パンフレットの4頁では、ペレットの圧縮に関する問題が考察されている。
マイクロカプセルは、蜜ろう、デンプン、ゼラチン、親水コロイド又はポリアクリル酸などのシェル材料の薄いコーティング内の固体の小粒子又は液体の液滴として定義される。これらは、例えば、酸化に対して保護するため、且つ/又は酵素、香味、栄養素及び薬物などの活性成分の放出速度を制御するために使用される。
本発明の多粒子固体剤形は、好ましくは、100℃未満、より好ましくは90℃未満、最も好ましくは85℃未満の温度で指定の混合物を流し込み成形することによって得られる。錠剤プレスは、使用されない。
本発明は、以下の実施例によってさらに例証される。
実施例1において、ドコサヘキサエン酸(DHA)がモデル物質として選択された。DHAは、オメガ−3脂肪酸である。他の大部分のオメガ−3脂肪酸のように、DHAは、酸化しやすい。酸化すると、DHAは、魚臭い異臭を発し、非常に少ない量でさえ摂取する際に容易に認識される。
実施例1を繰り返した。しかしながら、実施例2では、糖の代わりに甘味料(マルチトールシロップ剤及びステビオール配糖体)が使用される。弾性食感を有する円錐体が実施例1と同様に流し込み成形された。
実施例1を繰り返した。しかしながら、実施例3では、MEG−3(登録商標)DHA H粉末の代わりにLife’s DHA(登録商標)S24−P100粉末(これもDSM(登録商標)から入手可能)を使用した。Life’s DHA(登録商標)S24−P100は、菜食主義者用原料由来のDHAを封入するマイクロカプセルを含む。
Claims (15)
- 弾性食感を有する多粒子固体剤形を製造する方法であって、
水、マイクロカプセル及びデンプン粒子を含む混合物は、流し込み成形され、
前記混合物は、前記混合物の全重量に基づいて少なくとも5重量%の、300を超える重合度を有する少なくとも1種のデンプンを含む、方法において、
前記マイクロカプセルは、少なくとも1種の医薬品及び/又は少なくとも1種の微量栄養素を封入することを特徴とする方法。 - 前記混合物は、流し込み成形中、60Pa・s未満の動的粘度を有し、及び/又は前記混合物の前記粘度は、その調製から2時間後の前記混合物の前記粘度を、その調製から6時間後のその前記粘度と比較した場合、冷却なし及び加熱なしで少なくとも100%だけ増加する、請求項1に記載の方法。
- 前記混合物は、前記混合物の全重量に基づいて10重量%〜40重量%の粒状デンプン、好ましくは15重量%〜35重量%の粒状デンプン、最も好ましくは20重量%〜25重量%の粒状デンプンを含む、請求項1又は2に記載の方法。
- 前記混合物は、第1マイクロカプセルの凝集塊を含み、それぞれの個々の第1マイクロカプセルは、第1シェルを有し、及び前記凝集塊は、外部シェルによって封入される、請求項1〜3のいずれか一項に記載の方法。
- 前記第1シェル及び/又は前記外部シェルは、ゼラチン、ポリリン酸塩、アラビアゴム、アルジネート、キトサン、カラゲナン、ペクチン、カルボキシメチルセルロース又はその混合物を含む、請求項4に記載の方法。
- 前記マイクロカプセルは、疎水性液体を封入し、前記液体は、好ましくは、少なくとも1種の多価不飽和脂肪酸を含む、請求項1〜5のいずれか一項に記載の方法。
- 前記微量栄養素は、多価不飽和脂肪酸であり、前記多価不飽和脂肪酸は、遊離脂肪酸、塩、エステル、モノアシルグリセロール(MAG)、ジアシルグリセロール(DAG)、トリアシルグリセロール(TAG)、リン脂質(PL)又はその混合物の形態である、請求項1〜6のいずれか一項に記載の方法。
- 前記多価不飽和脂肪酸は、オメガ−3脂肪酸、オメガ−3脂肪酸のエステル、オメガ−6脂肪酸、オメガ−6脂肪酸のエステル又はその混合物である、請求項6又は7に記載の方法。
- 少なくとも100個のマイクロカプセルを含む流し込み成形多粒子固体剤形であって、1g〜6gの重量を有し、且つ/又は少なくとも10mgの少なくとも1種の多価不飽和脂肪酸を含む流し込み成形多粒子固体剤形。
- 請求項1〜8のいずれか一項に記載の方法によって得られ、且つ/又は好ましくは前記多粒子固体剤形の全重量に基づいて5重量%未満の水を含む、請求項9に記載の流し込み成形多粒子固体剤形。
- 2g〜5g、好ましくは3g〜4gの重量を有し、且つ/又は少なくとも20mg、好ましくは少なくとも30mgの少なくとも1種の多価不飽和脂肪酸を含む、請求項9又は10に記載の流し込み成形多粒子固体剤形。
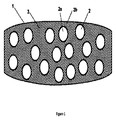
- 錠剤の形状を有さず、且つ好ましくは円錐形、球形、円柱形又はピラミッド形の形状を有する、請求項9〜11のいずれか一項に記載の流し込み成形多粒子固体剤形。
- 少なくとも100個のマイクロカプセルを含む多粒子固体剤形を流し込み成形するためのモーグルラインの使用。
- 前記多粒子固体剤形は、請求項9〜12のいずれか一項に記載の流し込み成形多粒子固体剤形である、請求項13に記載の使用。
- 請求項9〜12のいずれか一項に記載の少なくとも1種の多粒子固体剤形を含むパッケージであって、前記多粒子固体剤形は、円錐形の形状を有する、パッケージ。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP18166672 | 2018-04-10 | ||
| EP18166672.8 | 2018-04-10 | ||
| PCT/EP2019/059005 WO2019197432A1 (en) | 2018-04-10 | 2019-04-10 | Multiparticulate solid dosage form having an elastic texture |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2021519798A true JP2021519798A (ja) | 2021-08-12 |
| JPWO2019197432A5 JPWO2019197432A5 (ja) | 2022-02-01 |
Family
ID=61965824
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020553655A Pending JP2021519798A (ja) | 2018-04-10 | 2019-04-10 | 弾性食感を有する多粒子固体剤形 |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20210030681A1 (ja) |
| EP (1) | EP3773521A1 (ja) |
| JP (1) | JP2021519798A (ja) |
| KR (1) | KR20200143697A (ja) |
| CN (1) | CN111936123A (ja) |
| BR (1) | BR112020020567A2 (ja) |
| TW (1) | TW201943410A (ja) |
| WO (1) | WO2019197432A1 (ja) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0952850A (ja) * | 1995-06-09 | 1997-02-25 | Meiji Seika Kaisha Ltd | グミ製剤 |
| JP2009500034A (ja) * | 2005-07-07 | 2009-01-08 | オーシャン・ニュートリション・カナダ・リミテッド | デリバリーデバイスを有する食料品およびその調製方法 |
| US20090068333A1 (en) * | 2006-05-05 | 2009-03-12 | Innogel Ag | Low temperature mogul method |
| US20160038428A1 (en) * | 2014-08-05 | 2016-02-11 | Advanced Bionutrition Corp. | Encapsulation of hydrophobic biologically active compounds |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6974592B2 (en) | 2002-04-11 | 2005-12-13 | Ocean Nutrition Canada Limited | Encapsulated agglomeration of microcapsules and method for the preparation thereof |
| CA2620507C (en) * | 2005-08-30 | 2011-07-19 | Firmenich Sa | Encapsulated active ingredients, methods of preparation and their use |
| KR100758664B1 (ko) * | 2006-08-16 | 2007-09-13 | (주)케비젠 | 불포화지방산 함유 마이크로캡슐의 제조방법, 이 방법에의해 제조된 마이크로캡슐 및 이를 포함하는 제품 |
| EP2173321A2 (en) | 2007-07-19 | 2010-04-14 | DSM IP Assets B.V. | Tablettable formulations of lipophilic health ingredients |
| ES2337935T3 (es) | 2007-11-09 | 2010-04-30 | Acino Pharma Ag | Comprimidos de accion retardada con hidromorfona. |
| WO2009126993A1 (en) * | 2008-04-14 | 2009-10-22 | Cadbury Enterprises Pte Limited | Use of vibrational energy in producing jelly confectionery |
| CH700621A1 (de) | 2009-03-27 | 2010-09-30 | Innogel Ag | Verbessertes Mogul Verfahren. |
| CN104023727B (zh) * | 2011-10-28 | 2017-04-05 | 鲁美纳医药公司 | 用于治疗小儿胆汁淤积性肝病的胆汁酸再循环抑制剂 |
| EP2606879A1 (en) | 2011-12-21 | 2013-06-26 | Hexal AG | Multiple unit pellet tablet formulation comprising an opioid |
| WO2013136183A2 (en) * | 2012-03-12 | 2013-09-19 | Innolipid, As | Oxidixable fatty acid composition delivery form |
| CN103071437B (zh) * | 2012-10-09 | 2015-02-25 | 临沂宝力佳食品有限公司 | 二十二碳六烯酸微胶囊二次包埋方法 |
-
2019
- 2019-04-09 TW TW108112367A patent/TW201943410A/zh unknown
- 2019-04-10 WO PCT/EP2019/059005 patent/WO2019197432A1/en unknown
- 2019-04-10 CN CN201980024374.8A patent/CN111936123A/zh active Pending
- 2019-04-10 US US17/046,107 patent/US20210030681A1/en not_active Abandoned
- 2019-04-10 JP JP2020553655A patent/JP2021519798A/ja active Pending
- 2019-04-10 EP EP19715932.0A patent/EP3773521A1/en active Pending
- 2019-04-10 KR KR1020207031629A patent/KR20200143697A/ko not_active Application Discontinuation
- 2019-04-10 BR BR112020020567-2A patent/BR112020020567A2/pt not_active IP Right Cessation
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0952850A (ja) * | 1995-06-09 | 1997-02-25 | Meiji Seika Kaisha Ltd | グミ製剤 |
| JP2009500034A (ja) * | 2005-07-07 | 2009-01-08 | オーシャン・ニュートリション・カナダ・リミテッド | デリバリーデバイスを有する食料品およびその調製方法 |
| US20090068333A1 (en) * | 2006-05-05 | 2009-03-12 | Innogel Ag | Low temperature mogul method |
| US20160038428A1 (en) * | 2014-08-05 | 2016-02-11 | Advanced Bionutrition Corp. | Encapsulation of hydrophobic biologically active compounds |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201943410A (zh) | 2019-11-16 |
| BR112020020567A2 (pt) | 2021-01-12 |
| CN111936123A (zh) | 2020-11-13 |
| EP3773521A1 (en) | 2021-02-17 |
| US20210030681A1 (en) | 2021-02-04 |
| WO2019197432A1 (en) | 2019-10-17 |
| KR20200143697A (ko) | 2020-12-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101454942B1 (ko) | 채식주의용 마이크로캡슐 | |
| CA2640529C (en) | Chewable capsules | |
| EP2825057B1 (en) | Oxidixable fatty acid composition delivery form | |
| WO2008025034A2 (en) | Food fortification with polyunsaturated fatty acids | |
| US20080044481A1 (en) | Microparticles for Oral Delivery | |
| CN101801416A (zh) | 无缝胶囊 | |
| US20120156296A1 (en) | Antioxidants in fish oil powder and tablets | |
| WO2019008101A1 (en) | ENTERICALLY COATED SOLID DOSAGE FORM COMPRISING AMINO ACIDS SALTS OF OMEGA-3 FATTY ACIDS | |
| US20230157964A1 (en) | A stable food-grade microcapsule for the delivery of unstable and food-incompatible active ingredients to food products | |
| JP2021519798A (ja) | 弾性食感を有する多粒子固体剤形 | |
| KR100732199B1 (ko) | 저작성 캡슐 및 그 제조 방법 | |
| US20130108745A1 (en) | Coated effervescent tablet | |
| CN117120039A (zh) | 明胶胶囊剂 | |
| US20210015753A1 (en) | Freeze-dried multiparticulate solid dosage form | |
| CN1708290A (zh) | 含有稳定胆碱的剂型及其制备方法 | |
| CN113727705A (zh) | 新型多不饱和脂肪酸递送系统 | |
| JPWO2019197432A5 (ja) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220124 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20220124 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20221125 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20221213 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20230704 |