JP2021519668A - 中枢神経系局所的低体温装置および方法 - Google Patents
中枢神経系局所的低体温装置および方法 Download PDFInfo
- Publication number
- JP2021519668A JP2021519668A JP2021500502A JP2021500502A JP2021519668A JP 2021519668 A JP2021519668 A JP 2021519668A JP 2021500502 A JP2021500502 A JP 2021500502A JP 2021500502 A JP2021500502 A JP 2021500502A JP 2021519668 A JP2021519668 A JP 2021519668A
- Authority
- JP
- Japan
- Prior art keywords
- catheter
- cooling
- drainage
- coolant
- assembly
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000000034 method Methods 0.000 title abstract description 25
- 210000003169 central nervous system Anatomy 0.000 title abstract description 9
- 230000002631 hypothermal effect Effects 0.000 title description 25
- 238000001816 cooling Methods 0.000 claims abstract description 246
- 239000012530 fluid Substances 0.000 claims abstract description 56
- 238000007913 intrathecal administration Methods 0.000 claims abstract description 31
- 239000002826 coolant Substances 0.000 claims description 88
- 210000001175 cerebrospinal fluid Anatomy 0.000 claims description 74
- 238000003780 insertion Methods 0.000 claims description 29
- 230000037431 insertion Effects 0.000 claims description 29
- 210000000278 spinal cord Anatomy 0.000 claims description 28
- 238000005259 measurement Methods 0.000 claims description 24
- 230000004087 circulation Effects 0.000 claims description 20
- 229920002635 polyurethane Polymers 0.000 claims description 3
- 239000004814 polyurethane Substances 0.000 claims description 3
- 238000009428 plumbing Methods 0.000 claims 1
- 230000000712 assembly Effects 0.000 abstract description 4
- 238000000429 assembly Methods 0.000 abstract description 4
- 238000010586 diagram Methods 0.000 abstract description 2
- 230000013011 mating Effects 0.000 description 27
- 206010021113 Hypothermia Diseases 0.000 description 21
- 210000004556 brain Anatomy 0.000 description 18
- 230000006378 damage Effects 0.000 description 11
- 238000007789 sealing Methods 0.000 description 10
- 241001269524 Dura Species 0.000 description 8
- 239000008280 blood Substances 0.000 description 8
- 210000004369 blood Anatomy 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 230000001965 increasing effect Effects 0.000 description 8
- 208000014674 injury Diseases 0.000 description 8
- 230000035515 penetration Effects 0.000 description 8
- 210000001519 tissue Anatomy 0.000 description 8
- 208000027418 Wounds and injury Diseases 0.000 description 7
- 230000036961 partial effect Effects 0.000 description 7
- 230000008901 benefit Effects 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- 230000037361 pathway Effects 0.000 description 6
- 238000001356 surgical procedure Methods 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 238000011282 treatment Methods 0.000 description 5
- 238000003466 welding Methods 0.000 description 5
- 230000029142 excretion Effects 0.000 description 4
- 238000007917 intracranial administration Methods 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 230000010412 perfusion Effects 0.000 description 4
- 238000004886 process control Methods 0.000 description 4
- 230000002829 reductive effect Effects 0.000 description 4
- 238000012546 transfer Methods 0.000 description 4
- 208000010496 Heart Arrest Diseases 0.000 description 3
- 208000006011 Stroke Diseases 0.000 description 3
- 238000004026 adhesive bonding Methods 0.000 description 3
- 239000003570 air Substances 0.000 description 3
- 230000017531 blood circulation Effects 0.000 description 3
- 230000002490 cerebral effect Effects 0.000 description 3
- 208000028867 ischemia Diseases 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 230000002572 peristaltic effect Effects 0.000 description 3
- 230000002980 postoperative effect Effects 0.000 description 3
- 230000009885 systemic effect Effects 0.000 description 3
- 102000015081 Blood Coagulation Factors Human genes 0.000 description 2
- 108010039209 Blood Coagulation Factors Proteins 0.000 description 2
- 206010048962 Brain oedema Diseases 0.000 description 2
- 206010010904 Convulsion Diseases 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- 206010019233 Headaches Diseases 0.000 description 2
- 206010033799 Paralysis Diseases 0.000 description 2
- 241000269800 Percidae Species 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 206010047513 Vision blurred Diseases 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 239000003114 blood coagulation factor Substances 0.000 description 2
- 230000036770 blood supply Effects 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 230000036760 body temperature Effects 0.000 description 2
- 208000006752 brain edema Diseases 0.000 description 2
- 230000006835 compression Effects 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 239000004020 conductor Substances 0.000 description 2
- 239000012809 cooling fluid Substances 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 238000006073 displacement reaction Methods 0.000 description 2
- 230000005484 gravity Effects 0.000 description 2
- 231100000869 headache Toxicity 0.000 description 2
- 238000002513 implantation Methods 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000001361 intraarterial administration Methods 0.000 description 2
- 206010025482 malaise Diseases 0.000 description 2
- 230000000926 neurological effect Effects 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- 238000004806 packaging method and process Methods 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 229920003023 plastic Polymers 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 239000003507 refrigerant Substances 0.000 description 2
- 238000005057 refrigeration Methods 0.000 description 2
- 239000000565 sealant Substances 0.000 description 2
- 239000004065 semiconductor Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 208000020431 spinal cord injury Diseases 0.000 description 2
- 230000008961 swelling Effects 0.000 description 2
- SFLSHLFXELFNJZ-QMMMGPOBSA-N (-)-norepinephrine Chemical compound NC[C@H](O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-QMMMGPOBSA-N 0.000 description 1
- 206010002329 Aneurysm Diseases 0.000 description 1
- 201000001320 Atherosclerosis Diseases 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 206010008164 Cerebrospinal fluid leakage Diseases 0.000 description 1
- 206010053567 Coagulopathies Diseases 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 206010019196 Head injury Diseases 0.000 description 1
- 206010021143 Hypoxia Diseases 0.000 description 1
- 208000032382 Ischaemic stroke Diseases 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 229920002614 Polyether block amide Polymers 0.000 description 1
- 208000001647 Renal Insufficiency Diseases 0.000 description 1
- 208000007536 Thrombosis Diseases 0.000 description 1
- 206010043647 Thrombotic Stroke Diseases 0.000 description 1
- 208000030886 Traumatic Brain injury Diseases 0.000 description 1
- 239000004775 Tyvek Substances 0.000 description 1
- 229920000690 Tyvek Polymers 0.000 description 1
- 208000002223 abdominal aortic aneurysm Diseases 0.000 description 1
- OIPILFWXSMYKGL-UHFFFAOYSA-N acetylcholine Chemical compound CC(=O)OCC[N+](C)(C)C OIPILFWXSMYKGL-UHFFFAOYSA-N 0.000 description 1
- 229960004373 acetylcholine Drugs 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 239000012080 ambient air Substances 0.000 description 1
- 208000007474 aortic aneurysm Diseases 0.000 description 1
- 206010003119 arrhythmia Diseases 0.000 description 1
- 230000004872 arterial blood pressure Effects 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 229940009098 aspartate Drugs 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 210000003050 axon Anatomy 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 230000008499 blood brain barrier function Effects 0.000 description 1
- 208000015294 blood coagulation disease Diseases 0.000 description 1
- 210000001218 blood-brain barrier Anatomy 0.000 description 1
- 208000029028 brain injury Diseases 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 230000001964 calcium overload Effects 0.000 description 1
- 230000002612 cardiopulmonary effect Effects 0.000 description 1
- 210000001715 carotid artery Anatomy 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000002872 contrast media Substances 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 230000004153 glucose metabolism Effects 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 229940049906 glutamate Drugs 0.000 description 1
- 229960002449 glycine Drugs 0.000 description 1
- 210000003128 head Anatomy 0.000 description 1
- 230000002439 hemostatic effect Effects 0.000 description 1
- 230000007954 hypoxia Effects 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 208000037906 ischaemic injury Diseases 0.000 description 1
- 230000000302 ischemic effect Effects 0.000 description 1
- 201000006370 kidney failure Diseases 0.000 description 1
- 238000002350 laparotomy Methods 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000037323 metabolic rate Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 210000000944 nerve tissue Anatomy 0.000 description 1
- 210000002569 neuron Anatomy 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 229960002748 norepinephrine Drugs 0.000 description 1
- SFLSHLFXELFNJZ-UHFFFAOYSA-N norepinephrine Natural products NCC(O)C1=CC=C(O)C(O)=C1 SFLSHLFXELFNJZ-UHFFFAOYSA-N 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 230000008557 oxygen metabolism Effects 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 230000003134 recirculating effect Effects 0.000 description 1
- 210000004761 scalp Anatomy 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 238000004513 sizing Methods 0.000 description 1
- 210000003625 skull Anatomy 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 210000000701 subdural space Anatomy 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 210000000115 thoracic cavity Anatomy 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 230000000472 traumatic effect Effects 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 238000007631 vascular surgery Methods 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F7/12—Devices for heating or cooling internal body cavities
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M39/00—Tubes, tube connectors, tube couplings, valves, access sites or the like, specially adapted for medical use
- A61M39/22—Valves or arrangement of valves
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0021—Catheters; Hollow probes characterised by the form of the tubing
- A61M25/0023—Catheters; Hollow probes characterised by the form of the tubing by the form of the lumen, e.g. cross-section, variable diameter
- A61M25/0026—Multi-lumen catheters with stationary elements
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M25/00—Catheters; Hollow probes
- A61M25/0067—Catheters; Hollow probes characterised by the distal end, e.g. tips
- A61M25/0068—Static characteristics of the catheter tip, e.g. shape, atraumatic tip, curved tip or tip structure
- A61M25/007—Side holes, e.g. their profiles or arrangements; Provisions to keep side holes unblocked
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M27/00—Drainage appliance for wounds or the like, i.e. wound drains, implanted drains
- A61M27/002—Implant devices for drainage of body fluids from one part of the body to another
- A61M27/006—Cerebrospinal drainage; Accessories therefor, e.g. valves
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/44—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests having means for cooling or heating the devices or media
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F2007/0001—Body part
- A61F2007/0002—Head or parts thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F2007/0054—Heating or cooling appliances for medical or therapeutic treatment of the human body with a closed fluid circuit, e.g. hot water
- A61F2007/0056—Heating or cooling appliances for medical or therapeutic treatment of the human body with a closed fluid circuit, e.g. hot water for cooling
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F7/007—Heating or cooling appliances for medical or therapeutic treatment of the human body characterised by electric heating
- A61F2007/0075—Heating or cooling appliances for medical or therapeutic treatment of the human body characterised by electric heating using a Peltier element, e.g. near the spot to be heated or cooled
- A61F2007/0076—Heating or cooling appliances for medical or therapeutic treatment of the human body characterised by electric heating using a Peltier element, e.g. near the spot to be heated or cooled remote from the spot to be heated or cooled
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F7/12—Devices for heating or cooling internal body cavities
- A61F2007/126—Devices for heating or cooling internal body cavities for invasive application, e.g. for introducing into blood vessels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/33—Controlling, regulating or measuring
- A61M2205/3331—Pressure; Flow
- A61M2205/3334—Measuring or controlling the flow rate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/36—General characteristics of the apparatus related to heating or cooling
- A61M2205/3606—General characteristics of the apparatus related to heating or cooling cooled
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/36—General characteristics of the apparatus related to heating or cooling
- A61M2205/366—General characteristics of the apparatus related to heating or cooling by liquid heat exchangers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/36—General characteristics of the apparatus related to heating or cooling
- A61M2205/3673—General characteristics of the apparatus related to heating or cooling thermo-electric, e.g. Peltier effect, thermocouples, semi-conductors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2210/00—Anatomical parts of the body
- A61M2210/10—Trunk
- A61M2210/1003—Spinal column
Abstract
Description
本出願は、2018年3月22日に出願された「Central Nervous System Localized Hypothermia Apparatus and Methods」と題された米国仮特許出願第62/646,539号に対する米国特許法第119条に基づく優先権を主張し、その内容全体が参照により本明細書に組み込まれる。
Claims (20)
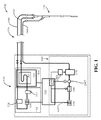
- 脊髄冷却および排出装置であって、
挿入部分および作動端部部分を備える、患者の中への髄腔内挿入のために適合される排出および冷却カテーテルアセンブリであって、前記排出および冷却カテーテルアセンブリの前記挿入部分の最大断面寸法が、約5Frである、排出および冷却カテーテルアセンブリと、
コンソールであって、
排出ポンプと、
前記排出ポンプに動作可能に接続されている排出制御システムであって、患者からの脳脊髄液の排出速度を制御するように構成されている、排出制御システムと、
脳脊髄液収集リザーバであって、前記排出および冷却カテーテルアセンブリに流体的に結合され、かつ前記収集リザーバ内に収集されたある容積の脳脊髄液上に自由に浮遊するようにサイズ決定されたフロートを備える、脳脊髄液収集リザーバと、
冷却液ポンプであって、前記排出および冷却カテーテルアセンブリに結合され、かつ前記排出および冷却カテーテルアセンブリと前記コンソールとの間で冷却液を循環させるように構成されている、冷却液ポンプと、
熱電冷却ユニットを備える冷却機アセンブリと、を備える、コンソールと、を備える装置。 - 前記排出制御システムが、
(a)第1の時点で前記フロートまでの第1の距離を測定して、前記第1の距離および前記第1の時点に対応する第1の信号を生成し、かつ(b)第2の時点で前記フロートまでの第2の距離を測定して、前記第2の距離および前記第2の時点に対応する第2の信号を生成するように適合された、非接触測定システムと、
(a)少なくとも前記第1の信号および前記第2の信号に基づいて流量を計算し、かつ(b)前記計算された流量に基づいて排出ポンプ制御信号を生成するように適合された、コントローラと、を備える、請求項1に記載の装置。 - 前記コンソールが、真空レギュレータをさらに備え、前記排出ポンプが、真空ポンプであり、前記真空ポンプおよび前記真空レギュレータが、前記排出制御システムコントローラに動作可能に接続されている、請求項2に記載の装置。
- 前記真空ポンプが、前記収集リザーバに流体的に接続され、かつ前記真空レギュレータが、前記真空ポンプと前記収集リザーバとの間に装着され、前記真空レギュレータが、前記排出ポンプ制御信号に基づいて、前記収集リザーバに適用される前記真空レベルを調節する、請求項3に記載の装置。
- 前記カテーテルアセンブリが、
近位端部、開放遠位端部、およびそれらの間を通る中央管腔を有する円筒形本体を有する排出カテーテルと、
前記排出カテーテルの前記近位端部を前記収集リザーバに流体的に結合する排出配管セットと、
冷却液入口ラインおよび冷却液出口ラインを備える冷却配管セットと、
近位端部および閉鎖遠位端部を有する冷却カテーテルであって、前記近位端部が、(a)前記冷却液入口ラインを前記冷却カテーテルの第1の内部管腔に、および(b)前記冷却液出口ラインを前記冷却カテーテルの第2の内部管腔に、結合するように適合され、前記第1の内部管腔および前記第2の内部管腔が、前記冷却カテーテルの前記閉鎖遠位端部の近くで流体連通している、冷却カテーテルと、を備え、
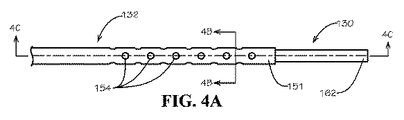
前記冷却液入口ラインおよび冷却液出口ラインが、前記冷却カテーテルを前記冷却機アセンブリに流体的に結合する、請求項1に記載の装置。 - 前記排出カテーテルが、前記円筒形本体の外側表面から前記中央管腔まで通る複数の孔をさらに備え、前記複数の孔が、前記排出カテーテルの前記遠位端部に向かって位置付けられる、請求項5に記載のカテーテルアセンブリ。
- 前記排出カテーテルの中央管腔の内壁と前記冷却カテーテルの外壁との間に脳脊髄液の流れを可能にする隙間が存在するように、前記冷却カテーテルが、前記排出カテーテルの前記中央管腔を通過するように構成される、請求項5に記載のカテーテルアセンブリ。
- 前記排出および冷却カテーテルアセンブリが、内部に埋め込まれた強化メッシュ構造を有するポリウレタンからなる、請求項1に記載の装置。
- 前記排出および冷却カテーテルアセンブリが、排出管腔と、冷却液入口管腔と、冷却液出口管腔と、を備える、多管腔カテーテルを備え、前記排出管腔の遠位端部が、患者の髄腔内空洞と流体連通するように適合され、かつ前記入口管腔および出口管腔が、前記多管腔カテーテルの前記遠位端部で互いに流体連通する、請求項1に記載の装置。
- 脊髄冷却および排出システムであって、
冷却カテーテルを備えるカテーテルアセンブリと、
配管セットと、
コンソールであって、
熱電冷却ユニットおよび冷却液を備える、冷却システムと、
循環システムと、
制御システムと、を備える、コンソールと、を備え、
前記配管セットが、前記冷却システムを前記カテーテルアセンブリに流体的に接続し、かつ前記冷却システムと前記冷却カテーテルとの間の前記冷却液の循環を可能にするように構成され、
前記循環システムが、前記冷却システムと前記カテーテルアセンブリとの間の前記冷却液の流れを制御するように構成される、脊髄冷却および排出システム。 - 前記冷却カテーテルが、近位端部および遠位端部を有し、かつ前記冷却カテーテル内の入口管腔および出口管腔を通した前記冷却液の循環を可能にするように構成され、前記入口管腔および出口管腔が、前記冷却カテーテルの前記遠位端部の近くで互いに流体的に接続され、かつ各々が前記冷却カテーテルの前記近位端部で前記配管セットに流体的に接続される、請求項10に記載の脊髄冷却システム。
- 前記カテーテルアセンブリの遠位部分が、患者の中への挿入のために構成され、かつ約5Frの最大外形寸法を有する、請求項10に記載の脊髄冷却システム。
- 前記カテーテルアセンブリの遠位部分が、患者の中への挿入のために構成され、かつ約4Frの最大外形寸法を有する、請求項10に記載の脊髄冷却システム。
- 排出システムをさらに備え、前記排出システムが、排出カテーテルと、排出配管セットと、収集リザーバと、排出制御装置と、を備え、前記排出配管セットが、前記排出カテーテルを前記収集リザーバに流体的に接続し、前記冷却カテーテルが、前記排出カテーテルの中央管腔を通って取り外し可能に同軸に挿入されるように構成される、請求項10に記載の脊髄冷却システム。
- 前記排出カテーテルが、近位端部および遠位端部を備え、前記排出カテーテルの前記遠位端部が、患者の中への挿入のために構成され、前記排出カテーテルの前記近位端部が、前記排出配管セットへの接続のために構成される、請求項14に記載の脊髄冷却システム。
- 前記排出制御装置が、前記収集リザーバに流体的に接続されている真空源と、前記制御システムに動作可能に接続されているフィードバックシステムと、を備える、請求項14に記載の脊髄冷却システム。
- 前記フィードバックシステムが、非接触測定システムと、前記収集リザーバ内に収集されたある容積の流体上に浮遊するように構成されているフロートと、を備え、前記フィードバックシステムが、前記非接触測定システムからの読み取りに基づいてフィードバックデータを生成するように構成される、請求項16に記載の脊髄冷却システム。
- 前記排出制御システムが、前記排出カテーテルを通るCSF流量を調節するように、前記フィードバックデータに基づいて前記真空源を制御するように構成される、請求項17に記載の脊髄冷却システム。
- 前記コンソールが、熱交換器を備える冷却液回路をさらに備え、前記冷却液回路が、前記流体冷却システム、前記配管セット、および前記カテーテルシステムを流体的に接続し、前記冷却液を前記循環システムによって閉ループ内で循環することを可能にし、前記熱交換器が、前記循環冷却液から熱を除去するように、前記熱電冷却ユニットと相互作用するように適合される、請求項10に記載の脊髄冷却システム。
- 前記熱交換器が、前記熱電冷却システムへ取り外し可能に挿入されるように構成される可撓性ポーチを備える、請求項19に記載の脊髄冷却システム。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201862646539P | 2018-03-22 | 2018-03-22 | |
| US62/646,539 | 2018-03-22 | ||
| PCT/US2019/023556 WO2019183464A1 (en) | 2018-03-22 | 2019-03-22 | Central nervous system localized hypothermia apparatus and methods |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2021519668A true JP2021519668A (ja) | 2021-08-12 |
| JPWO2019183464A5 JPWO2019183464A5 (ja) | 2022-04-04 |
Family
ID=67984476
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2021500502A Pending JP2021519668A (ja) | 2018-03-22 | 2019-03-22 | 中枢神経系局所的低体温装置および方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US11672695B2 (ja) |
| EP (1) | EP3768204A4 (ja) |
| JP (1) | JP2021519668A (ja) |
| CA (1) | CA3094812A1 (ja) |
| WO (1) | WO2019183464A1 (ja) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA3198090A1 (en) * | 2020-10-06 | 2022-04-14 | Enclear Therapies, Inc. | System and method for controlling csf flow and managing intracranial pressure |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5343997A (en) * | 1976-10-02 | 1978-04-20 | Klauschenz & Perrot | Device for dialyzing patient*s blood |
| JPH01204674A (ja) * | 1988-02-10 | 1989-08-17 | Japan Medical Dynamic Marketing Inc | 流量測定式脳室シャント |
| JP2000254238A (ja) * | 1999-03-11 | 2000-09-19 | Olympus Optical Co Ltd | 加温治療用プローブ |
| US20160331282A1 (en) * | 2015-05-15 | 2016-11-17 | Gauss Surgical, Inc. | Systems and methods for assessing fluids from a patient |
| WO2017062606A1 (en) * | 2015-10-06 | 2017-04-13 | Minnetronix, Inc. | Devices and methods for providing focal cooling to the brain and spinal cord |
Family Cites Families (100)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3662598A (en) * | 1969-06-09 | 1972-05-16 | Jordan L Spencer | Flow control system |
| US3985601A (en) | 1975-05-05 | 1976-10-12 | Quantum, Inc. | Method for producing a balloon type catheter having a smooth continuous outer surface |
| US6623516B2 (en) | 1992-08-13 | 2003-09-23 | Mark A. Saab | Method for changing the temperature of a selected body region |
| US5437673A (en) | 1993-02-04 | 1995-08-01 | Cryomedical Sciences, Inc. | Closed circulation tissue warming apparatus and method of using the same in prostate surgery |
| US5837003A (en) | 1993-02-10 | 1998-11-17 | Radiant Medical, Inc. | Method and apparatus for controlling a patient's body temperature by in situ blood temperature modification |
| US6620188B1 (en) | 1998-08-24 | 2003-09-16 | Radiant Medical, Inc. | Methods and apparatus for regional and whole body temperature modification |
| US6036654A (en) | 1994-09-23 | 2000-03-14 | Baxter International Inc. | Multi-lumen, multi-parameter catheter |
| US6231595B1 (en) | 1998-03-31 | 2001-05-15 | Innercool Therapies, Inc. | Circulating fluid hypothermia method and apparatus |
| US7458984B2 (en) | 1998-01-23 | 2008-12-02 | Innercool Therapies, Inc. | System and method for inducing hypothermia with active patient temperature control employing catheter-mounted temperature sensor and temperature projection algorithm |
| US6325818B1 (en) | 1999-10-07 | 2001-12-04 | Innercool Therapies, Inc. | Inflatable cooling apparatus for selective organ hypothermia |
| US6051019A (en) | 1998-01-23 | 2000-04-18 | Del Mar Medical Technologies, Inc. | Selective organ hypothermia method and apparatus |
| US6558412B2 (en) | 1998-01-23 | 2003-05-06 | Innercool Therapies, Inc. | Selective organ hypothermia method and apparatus |
| US6530946B1 (en) | 1998-04-21 | 2003-03-11 | Alsius Corporation | Indwelling heat exchange heat pipe catheter and method of using same |
| US6368304B1 (en) | 1999-02-19 | 2002-04-09 | Alsius Corporation | Central venous catheter with heat exchange membrane |
| US6419643B1 (en) | 1998-04-21 | 2002-07-16 | Alsius Corporation | Central venous catheter with heat exchange properties |
| US6090281A (en) | 1998-08-06 | 2000-07-18 | Buckner; Lynn | Beverage storage and dispensing container |
| US6673098B1 (en) | 1998-08-24 | 2004-01-06 | Radiant Medical, Inc. | Disposable cassette for intravascular heat exchange catheter |
| US6129701A (en) | 1998-11-19 | 2000-10-10 | Sound Surgical Technologies, Llc | Vented aspirator and method |
| EP1011152A3 (en) | 1998-12-16 | 2000-11-15 | Matsushita Electric Industrial Co., Ltd. | Heat transfer apparatus |
| US7014624B2 (en) | 1999-03-03 | 2006-03-21 | The Uab Research Foundation | Direct central nervous system catheter and temperature control system |
| IL129465A (en) | 1999-04-15 | 2005-03-20 | M T R E Advanced Technology Lt | Heat exchanger garment |
| US6264679B1 (en) | 1999-08-20 | 2001-07-24 | Radiant Medical, Inc. | Heat exchange catheter with discrete heat exchange elements |
| US6289600B1 (en) | 1999-11-02 | 2001-09-18 | United States Pipe & Foundry Company | Non-contact measuring device |
| US6626902B1 (en) | 2000-04-12 | 2003-09-30 | University Of Virginia Patent Foundation | Multi-probe system |
| US6482184B1 (en) | 2000-09-29 | 2002-11-19 | Advanced Infusion, Inc. | Attachable catheter |
| US6348678B1 (en) | 2000-10-24 | 2002-02-19 | Patrick V. Loyd, Sr. | Flexible heater assembly |
| EP1384271B1 (en) | 2001-04-24 | 2005-11-09 | Top-Cool Holding B.V. | Electric cooling device |
| US6524302B2 (en) | 2001-04-26 | 2003-02-25 | Scimed Life Systems, Inc. | Multi-lumen catheter |
| US6699269B2 (en) * | 2001-04-30 | 2004-03-02 | Rohit K. Khanna | Selective brain and spinal cord hypothermia method and apparatus |
| US7455666B2 (en) | 2001-07-13 | 2008-11-25 | Board Of Regents, The University Of Texas System | Methods and apparatuses for navigating the subarachnoid space |
| US6929656B1 (en) | 2001-09-14 | 2005-08-16 | Medcool, Inc. | Method and device for reducing secondary brain injury |
| US6645233B1 (en) * | 2001-09-27 | 2003-11-11 | Gregory M. Ayers | Drainage tube with heat transfer function and methods of use |
| EP1453425B1 (en) | 2001-12-03 | 2006-03-08 | Ekos Corporation | Catheter with multiple ultrasound radiating members |
| US7156867B2 (en) | 2001-12-31 | 2007-01-02 | Medcool, Inc. | Uniform selective cerebral hypothermia |
| US7276043B2 (en) | 2002-01-29 | 2007-10-02 | Scimed Life Systems, Inc. | Occlusion-resistant catheter |
| US20030216710A1 (en) | 2002-03-26 | 2003-11-20 | Hurt Robert F. | Catheter |
| WO2003089031A1 (en) * | 2002-04-19 | 2003-10-30 | Neuron Therapeutics | Subarachnoid spinal catheter for transporting cerebrospinal fluid |
| EP1514529A4 (en) | 2002-06-17 | 2006-08-16 | Atsuo Mori | CATHETER FOR TOPICAL COOLING AND TOPICAL COOLING DEVICE USING THE CATHETER |
| US20030236489A1 (en) | 2002-06-21 | 2003-12-25 | Baxter International, Inc. | Method and apparatus for closed-loop flow control system |
| US6916294B2 (en) | 2002-07-09 | 2005-07-12 | George Washington University | Brain retraction sensor |
| US6969399B2 (en) | 2002-07-11 | 2005-11-29 | Life Recovery Systems Hd, Llc | Apparatus for altering the body temperature of a patient |
| US7211066B1 (en) | 2002-07-15 | 2007-05-01 | Focal Cool, Llc | Active surface exchange catheter and method |
| US20040211189A1 (en) | 2002-07-17 | 2004-10-28 | Arnold Anthony P. | Personal heat control device and method |
| JP4890761B2 (ja) | 2002-07-19 | 2012-03-07 | バクスター・インターナショナル・インコーポレイテッド | 腹膜透析を実施するためのシステムおよび方法 |
| DE10261366A1 (de) | 2002-12-30 | 2004-07-08 | BSH Bosch und Siemens Hausgeräte GmbH | Hilfskühlvorrichtung |
| US7004961B2 (en) | 2003-01-09 | 2006-02-28 | Edward Wong | Medical device and method for temperature control and treatment of the brain and spinal cord |
| US6823678B1 (en) | 2003-12-22 | 2004-11-30 | Ferrotec (Usa) Corporation | Air conditioner system for flexible material-based devices |
| US7318834B2 (en) | 2004-02-09 | 2008-01-15 | Philip Chidi Njemanze | Apparatus and method for hypothermia and rewarming by altering the temperature of the cerebrospinal fluid in the brain |
| US8157792B2 (en) | 2004-02-26 | 2012-04-17 | Haemonetics Corporation | Wound drainage suction relief |
| US7127033B2 (en) | 2004-02-28 | 2006-10-24 | Xoft, Inc. | Miniature x-ray tube cooling system |
| IL160738A (en) * | 2004-03-04 | 2007-12-03 | Shai Amisar | Liquid flow monitoring apparatus |
| WO2005122933A1 (ja) | 2004-06-21 | 2005-12-29 | Hiroshi Furuhata | 超音波脳梗塞治療装置 |
| US8353942B2 (en) * | 2004-09-16 | 2013-01-15 | Thomas Lad Merrill | Cooling guide catheter and associated method of use |
| US8672988B2 (en) | 2004-10-22 | 2014-03-18 | Medtronic Cryocath Lp | Method and device for local cooling within an organ using an intravascular device |
| US20060167438A1 (en) | 2005-01-26 | 2006-07-27 | Gary Kalser | Pressure-limiting balloon catheter and method for using the catheter |
| US20060175543A1 (en) * | 2005-02-08 | 2006-08-10 | John Elefteriades | Intra-thecal catheter and method for cooling the spinal cord |
| US20070038171A1 (en) * | 2005-07-25 | 2007-02-15 | Mayer Peter L | Shunt system |
| DE502005002730D1 (de) | 2005-08-02 | 2008-03-20 | Moeller Medical Gmbh & Co Kg | Liquor-Drainagesystem |
| US20070245765A1 (en) | 2005-08-26 | 2007-10-25 | Calvin Casher | Multi-wine dispenser/the wine box cooler |
| GR20050100452A (el) | 2005-09-02 | 2007-04-25 | Estelle Enterprises Limited | Συστημα καθετηρα ανταλλαγης υγρων |
| US7639346B2 (en) | 2005-09-14 | 2009-12-29 | Booker Jr Reginald Earl | Non-contact measurement device |
| CA2634683C (en) | 2005-12-22 | 2016-02-02 | The Trustees Of Columbia University In The City Of New York | Systems and methods for intravascular cooling |
| NL2000032C2 (nl) | 2006-03-20 | 2007-09-21 | Bredel Hose Pumps B V | Peristaltische pomp, werkwijze voor het vervaardigen van een slang daarvoor, en slang voor een dergelijke pomp. |
| US7403384B2 (en) | 2006-07-26 | 2008-07-22 | Dell Products L.P. | Thermal docking station for electronics |
| US7588549B2 (en) | 2006-08-03 | 2009-09-15 | Terumo Cardiovascular Systems Corporation | Thermoelectric temperature control for extracorporeal blood circuit |
| US20080044473A1 (en) | 2006-08-17 | 2008-02-21 | Milan Radojicic | Methods of reducing nervous system glial scar formation using cyclosporine components |
| US7822485B2 (en) | 2006-09-25 | 2010-10-26 | Zoll Circulation, Inc. | Method and apparatus for spinal cooling |
| US8192474B2 (en) | 2006-09-26 | 2012-06-05 | Zeltiq Aesthetics, Inc. | Tissue treatment methods |
| EP2086573B1 (en) | 2006-10-09 | 2020-11-25 | Neurofluidics, Inc. | Cerebrospinal fluid purification system |
| US10569064B2 (en) * | 2013-03-13 | 2020-02-25 | Minnetronix, Inc. | Devices and methods for providing focal cooling to the brain and spinal cord |
| WO2008079202A1 (en) | 2006-12-19 | 2008-07-03 | Lam Research Corporation | Measuring fluid quantities, blending liquid constituents, and dispensing blends |
| US7824357B2 (en) | 2007-06-11 | 2010-11-02 | Ibrahim Rashid Al-Rashdan | Method and system for preventing contrast associated nephropathy |
| AU2008275158A1 (en) | 2007-07-09 | 2009-01-15 | Velomedix, Inc | Hypothermia devices and methods |
| US8002729B2 (en) | 2007-08-21 | 2011-08-23 | Cook Medical Technologies Llc | Multi-lumen catheter assembly |
| US8221366B2 (en) | 2007-09-05 | 2012-07-17 | Integra Lifesciences Corporation | Volume limiting bodily fluid drainage system |
| AU2009206271B8 (en) | 2008-01-25 | 2013-04-11 | Neurosave, Inc, | Rapid cooling of body and/or brain by irrigating with a cooling liquid |
| US8475508B2 (en) | 2008-05-15 | 2013-07-02 | Ryan R Munson | Therapeutic cooling system |
| US8162879B2 (en) | 2008-09-22 | 2012-04-24 | Tyco Healthcare Group Lp | Double balloon catheter and methods for homogeneous drug delivery using the same |
| JP5746973B2 (ja) | 2008-12-05 | 2015-07-08 | フルイセンス アーペーエス | 体液サンプリングデバイスおよびその方法 |
| CA2750473A1 (en) | 2009-02-06 | 2010-08-12 | Velomedix, Inc. | Method and apparatus for inducing therapeutic hypothermia |
| AU2010217849A1 (en) | 2009-02-26 | 2011-09-15 | Advanced Cooling Therapy, Llc | Devices and methods for controlling patient temperature |
| US8905968B2 (en) * | 2009-04-29 | 2014-12-09 | Encephalon Technologies, Llc | System for cooling and pressurizing fluid |
| WO2011041508A1 (en) | 2009-09-30 | 2011-04-07 | Xcellerex, Inc. | Disposable bioreactor condenser bag and filter heater |
| US9078980B2 (en) | 2010-02-02 | 2015-07-14 | Nirva Medical, Llc | Localized therapy delivery and local organ protection |
| AU2011218179A1 (en) | 2010-02-16 | 2012-09-06 | The Johns Hopkins University | Method and device for non-invasive anatomical and systemic cooling and neuroprotection |
| US20120302938A1 (en) | 2010-03-19 | 2012-11-29 | University Of Washington | Drainage systems for excess body fluids and associated methods |
| US8441792B2 (en) | 2010-07-21 | 2013-05-14 | Birchbridge Incorporated | Universal conduction cooling platform |
| EP2625425A2 (en) | 2010-10-07 | 2013-08-14 | Vanderbilt University | Peristaltic micropump and related systems and methods |
| EP2719015A2 (en) | 2011-06-06 | 2014-04-16 | Gentherm Incorporated | Cartridge-based thermoelectric systems |
| EP2736581A4 (en) | 2011-07-25 | 2015-11-04 | Neurosave Inc | NON-INVASIVE SYSTEMS, DEVICES AND METHODS FOR SELECTIVE BRAIN COOLING |
| US9846085B2 (en) | 2012-07-25 | 2017-12-19 | Nxstage Medical, Inc. | Fluid property measurement devices, methods, and systems |
| US8926077B2 (en) | 2013-02-26 | 2015-01-06 | Inx International Ink Company | Ink supply system for ink jet printers |
| US8795219B1 (en) | 2013-03-15 | 2014-08-05 | Ibrahim Rashid Al-Rashdan | System and method for removing blood containing contrast dye |
| DE102013104951B4 (de) | 2013-05-14 | 2015-11-19 | Acandis Gmbh & Co. Kg | Ventrikelkatheter sowie Anordnung und System mit einem Ventrikelkatheter |
| WO2015039104A2 (en) | 2013-09-16 | 2015-03-19 | Neuraxis, Llc | Methods and devices for applying localized thermal therapy |
| US10271987B2 (en) | 2014-02-24 | 2019-04-30 | Lucent Medical Systems, Inc. | Localized therapeutic hypothermia system, device, and method |
| US9440043B2 (en) | 2014-06-13 | 2016-09-13 | Leading Age Supplies LLC | Catheter having a tapered structure and balloon formed above a lower drainage hole |
| US9784263B2 (en) | 2014-11-06 | 2017-10-10 | Zoll Circulation, Inc. | Heat exchange system for patient temperature control with easy loading high performance peristaltic pump |
| EP3407956B1 (en) | 2016-03-03 | 2022-02-09 | Esculon, LLC | Devices and methods for managing chest drainage |
| US11420031B2 (en) * | 2017-10-09 | 2022-08-23 | Alan Richard Turtz | System and method for draining cerebrospinal fluid in hydrocephalus patients |
-
2019
- 2019-03-22 WO PCT/US2019/023556 patent/WO2019183464A1/en active Application Filing
- 2019-03-22 CA CA3094812A patent/CA3094812A1/en active Pending
- 2019-03-22 US US16/361,566 patent/US11672695B2/en active Active
- 2019-03-22 JP JP2021500502A patent/JP2021519668A/ja active Pending
- 2019-03-22 EP EP19772187.1A patent/EP3768204A4/en active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5343997A (en) * | 1976-10-02 | 1978-04-20 | Klauschenz & Perrot | Device for dialyzing patient*s blood |
| JPH01204674A (ja) * | 1988-02-10 | 1989-08-17 | Japan Medical Dynamic Marketing Inc | 流量測定式脳室シャント |
| JP2000254238A (ja) * | 1999-03-11 | 2000-09-19 | Olympus Optical Co Ltd | 加温治療用プローブ |
| US20160331282A1 (en) * | 2015-05-15 | 2016-11-17 | Gauss Surgical, Inc. | Systems and methods for assessing fluids from a patient |
| WO2017062606A1 (en) * | 2015-10-06 | 2017-04-13 | Minnetronix, Inc. | Devices and methods for providing focal cooling to the brain and spinal cord |
| JP2018529473A (ja) * | 2015-10-06 | 2018-10-11 | ミネトロニクス, インコーポレイテッド | 脳および脊髄に局所冷却を提供する装置および方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| US11672695B2 (en) | 2023-06-13 |
| WO2019183464A1 (en) | 2019-09-26 |
| EP3768204A4 (en) | 2022-01-12 |
| CA3094812A1 (en) | 2019-09-26 |
| US20190290481A1 (en) | 2019-09-26 |
| EP3768204A1 (en) | 2021-01-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7156867B2 (en) | Uniform selective cerebral hypothermia | |
| US11752251B2 (en) | System and method for extracorporeal temperature control | |
| US7241307B2 (en) | Method and apparatus for managing temperature in a patient | |
| JP5624599B2 (ja) | 改良型の体内から熱交換を行う装置およびその方法 | |
| CA2357914C (en) | Cooling system for indwelling heat exchange catheter | |
| US7842002B2 (en) | Device for the extravascular recirculation of liquid in body cavities | |
| US20120095537A1 (en) | Method and Apparatus for Inducing Therapeutic Hypothermia | |
| US20140031631A1 (en) | Method and Apparatus for Inducing Therapeutic Hypothermia | |
| WO2005023200A2 (en) | System and method for cooling internal tissue | |
| US20110029050A1 (en) | Intra-ventricular brain cooling catheter | |
| WO2001076655A9 (en) | Method and apparatus for regulating patient temperature by irrigating the bladder with a fluid | |
| JP2021519668A (ja) | 中枢神経系局所的低体温装置および方法 | |
| AU2016203442B2 (en) | Method for adjusting the temperature of a patient | |
| JP2023505752A (ja) | 脱着式冷却装置、関連システムおよび配置方法 | |
| AU2012211501B2 (en) | Method for adjusting the temperature of a patient |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20210601 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20220322 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20220325 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20221223 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20230117 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20230413 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20230613 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20230912 |