JP2010095492A - 脂質異常治療薬の副作用防止物質 - Google Patents
脂質異常治療薬の副作用防止物質 Download PDFInfo
- Publication number
- JP2010095492A JP2010095492A JP2008269530A JP2008269530A JP2010095492A JP 2010095492 A JP2010095492 A JP 2010095492A JP 2008269530 A JP2008269530 A JP 2008269530A JP 2008269530 A JP2008269530 A JP 2008269530A JP 2010095492 A JP2010095492 A JP 2010095492A
- Authority
- JP
- Japan
- Prior art keywords
- fibrate
- dyslipidemia
- substance
- group
- treating agent
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 230000000694 effects Effects 0.000 title claims abstract description 26
- 239000000126 substance Substances 0.000 title claims abstract description 10
- 208000032928 Dyslipidaemia Diseases 0.000 title claims abstract description 8
- 208000017170 Lipid metabolism disease Diseases 0.000 title claims abstract description 8
- 230000002411 adverse Effects 0.000 title abstract 4
- 239000003814 drug Substances 0.000 claims abstract description 25
- 229940125753 fibrate Drugs 0.000 claims abstract description 22
- 241000195634 Dunaliella Species 0.000 claims abstract description 14
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 8
- 229940079593 drug Drugs 0.000 claims description 17
- 239000000843 powder Substances 0.000 claims description 5
- 239000002775 capsule Substances 0.000 claims description 4
- 239000008187 granular material Substances 0.000 claims description 3
- 229940126585 therapeutic drug Drugs 0.000 claims description 3
- 230000001258 dyslipidemic effect Effects 0.000 claims description 2
- 239000003826 tablet Substances 0.000 claims description 2
- 206010020880 Hypertrophy Diseases 0.000 abstract description 9
- 239000000556 agonist Substances 0.000 abstract description 8
- 230000015572 biosynthetic process Effects 0.000 abstract description 7
- 238000003786 synthesis reaction Methods 0.000 abstract description 7
- 208000001145 Metabolic Syndrome Diseases 0.000 abstract description 5
- 201000000690 abdominal obesity-metabolic syndrome Diseases 0.000 abstract description 5
- 230000003908 liver function Effects 0.000 abstract description 3
- 102000023984 PPAR alpha Human genes 0.000 abstract 1
- 230000004663 cell proliferation Effects 0.000 abstract 1
- 230000002440 hepatic effect Effects 0.000 abstract 1
- 108091008725 peroxisome proliferator-activated receptors alpha Proteins 0.000 abstract 1
- 230000001737 promoting effect Effects 0.000 abstract 1
- 210000004185 liver Anatomy 0.000 description 16
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 9
- 102000003728 Peroxisome Proliferator-Activated Receptors Human genes 0.000 description 8
- 108090000029 Peroxisome Proliferator-Activated Receptors Proteins 0.000 description 8
- 230000005856 abnormality Effects 0.000 description 8
- 238000002474 experimental method Methods 0.000 description 8
- 150000002632 lipids Chemical class 0.000 description 8
- 210000001789 adipocyte Anatomy 0.000 description 6
- 229960002297 fenofibrate Drugs 0.000 description 6
- YMTINGFKWWXKFG-UHFFFAOYSA-N fenofibrate Chemical compound C1=CC(OC(C)(C)C(=O)OC(C)C)=CC=C1C(=O)C1=CC=C(Cl)C=C1 YMTINGFKWWXKFG-UHFFFAOYSA-N 0.000 description 6
- 238000005259 measurement Methods 0.000 description 6
- 230000007935 neutral effect Effects 0.000 description 5
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 4
- 101100041816 Homo sapiens SCD gene Proteins 0.000 description 3
- 108010013563 Lipoprotein Lipase Proteins 0.000 description 3
- 102100022119 Lipoprotein lipase Human genes 0.000 description 3
- 206010067125 Liver injury Diseases 0.000 description 3
- 101150097713 SCD1 gene Proteins 0.000 description 3
- 102100028897 Stearoyl-CoA desaturase Human genes 0.000 description 3
- 108010021111 Uncoupling Protein 2 Proteins 0.000 description 3
- 230000022131 cell cycle Effects 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 231100000234 hepatic damage Toxicity 0.000 description 3
- 230000008818 liver damage Effects 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 229940124597 therapeutic agent Drugs 0.000 description 3
- 241000195633 Dunaliella salina Species 0.000 description 2
- 206010019842 Hepatomegaly Diseases 0.000 description 2
- 241000699666 Mus <mouse, genus> Species 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- 241000283984 Rodentia Species 0.000 description 2
- 102000008219 Uncoupling Protein 2 Human genes 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 239000003638 chemical reducing agent Substances 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 238000005265 energy consumption Methods 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 208000019423 liver disease Diseases 0.000 description 2
- 230000005976 liver dysfunction Effects 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 102000006311 Cyclin D1 Human genes 0.000 description 1
- 108010058546 Cyclin D1 Proteins 0.000 description 1
- 206010053759 Growth retardation Diseases 0.000 description 1
- 206010023126 Jaundice Diseases 0.000 description 1
- 238000008214 LDL Cholesterol Methods 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 210000000577 adipose tissue Anatomy 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 238000002485 combustion reaction Methods 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 230000003467 diminishing effect Effects 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 235000009200 high fat diet Nutrition 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 210000001596 intra-abdominal fat Anatomy 0.000 description 1
- 230000037356 lipid metabolism Effects 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 238000007449 liver function test Methods 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 229940127557 pharmaceutical product Drugs 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000003449 preventive effect Effects 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108091006084 receptor activators Proteins 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 150000003626 triacylglycerols Chemical class 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/02—Algae
- A61K36/05—Chlorophycota or chlorophyta (green algae), e.g. Chlorella
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Animal Behavior & Ethology (AREA)
- Biotechnology (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Botany (AREA)
- Organic Chemistry (AREA)
- Mycology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microbiology (AREA)
- Medical Informatics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Alternative & Traditional Medicine (AREA)
- Diabetes (AREA)
- Obesity (AREA)
- Gastroenterology & Hepatology (AREA)
- Hematology (AREA)
- Medicines Containing Plant Substances (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
【解決手段】 本発明に係る脂質異常治療薬の副作用防止物質は、脂質異常治療薬のフィブレート系薬剤と併用投与して、該フィブレート系薬剤による副作用を防止するデュナリエラ属の微細藻から抽出した抽出物からなるものであり、フィブレート系薬剤のPPAR−αアゴニストによって起こる肝肥大を抑制し、脂肪燃焼促進、脂肪合成抑制、細胞増殖抑制の作用があり、それによる肝機能障害を予防する。
【選択図】なし
Description
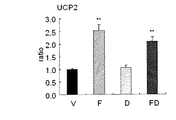
KKAyマウスを高脂肪食負荷で飼育し、そのマウスをV群(コントロール群)と、F群(フィブレート系薬剤単独投与群)と、D群(デュナリエラ抽出物単独投与群)と、FD群(フィブレート系薬剤とデュナリエラ抽出物併用群)に分け、フィブレート系薬剤は0.1%に、デュナリエラ抽出物は0.4%になるように飼料に配合して投与した。投与8週間後に解剖して肝臓重量、ACO、UCP2、LPL、SCD1を測定した。測定結果は、図1〜図5に示したとおりである。なお、全肝重量あたりのTG含量と、細胞周期についても計測した。その結果は図6と図7に示したとおりであった。
1).図1から明らかなように、肝臓重量が、F群と比較してFD群で約30%肝肥大を 抑制した。
2).図2から明らかなように、脂肪燃焼に関わるACOの発現は、V群に比べてF群と FD群で高く、脂肪燃焼が促進されたことを示している。
3).図3から明らかなように、エネルギー消費に関わる遺伝子UCP2の発現が、V群 に比べてF群とFD群で上昇し、脂肪燃焼したことを示している。
4).図4から明らかなように、中性脂肪の分解に関わるLPLの発現は、V群に比べて F群とFD群で上昇し、中性脂肪を分解したことを示している。
5).図5から明らかなように、脂肪合成に関わるSCD1は、F群に比べてFD群で約 61.5%抑制され、脂肪の合成が抑制されたことを示している。また、V群に比べ てD群でも脂肪の合成を抑制していることより、脂肪蓄積による肝肥大の抑制がある と推定される。
6).図6から明らかなように、肝臓重量当たりのトリグリセライド(TG)は、F群に 比べてFD群で50%低い値を示し、FD群とV群とは差がないことからトリグリセ ライド増加を抑制したと推定される。
7).図7から明らかなように、細胞周期に関わる遺伝子(Cyclin D1)は、F群で異常 に高く、肝肥大を生起させることが示されているが、FD群では完全に抑制している ことを示している。
このことは脂質異常治療薬として使用されているフィブレート系薬剤(fenofibrate)などのPPAR−αアゴニストによる副作用を抑制すると考えられる。
Claims (2)
- 脂質異常治療薬のフィブレート系薬剤と併用投与して、該フィブレート系薬剤による副作用を防止するデュナリエラ属の微細藻から抽出した抽出物からなる脂質異常治療薬の副作用防止物質。
- 前記抽出物を、カプセル状、錠剤状、顆粒状または粉末状にしたこと
を特徴とする請求項1に記載の脂質異常治療薬の副作用防止物質。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008269530A JP5733592B2 (ja) | 2008-10-20 | 2008-10-20 | 脂質異常治療薬の副作用防止物質 |
| US12/385,961 US20100098720A1 (en) | 2008-10-20 | 2009-04-24 | Substance preventing adverse actions of therapeutic agents for dyslipidemia |
| US12/852,704 US20100303855A1 (en) | 2008-10-20 | 2010-08-09 | Method of reducing adverse effects of therapeutic agents for dyslipidemia |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008269530A JP5733592B2 (ja) | 2008-10-20 | 2008-10-20 | 脂質異常治療薬の副作用防止物質 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010095492A true JP2010095492A (ja) | 2010-04-30 |
| JP5733592B2 JP5733592B2 (ja) | 2015-06-10 |
Family
ID=42108859
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2008269530A Active JP5733592B2 (ja) | 2008-10-20 | 2008-10-20 | 脂質異常治療薬の副作用防止物質 |
Country Status (2)
| Country | Link |
|---|---|
| US (2) | US20100098720A1 (ja) |
| JP (1) | JP5733592B2 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011105656A (ja) * | 2009-11-18 | 2011-06-02 | Microalgae Corporation | 脱共役タンパク質発現誘導剤及び冷え改善剤 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5310554A (en) * | 1992-10-27 | 1994-05-10 | Natural Carotene Corporation | High purity beta-carotene |
| US5480909A (en) * | 1994-08-08 | 1996-01-02 | University Of Pittsburgh Medical Center | Method for inhibiting generation of free-radicals |
| JPH11187894A (ja) * | 1997-12-26 | 1999-07-13 | Oyo Seikagaku Kenkyusho | 高純度 9−シス−β−カロチン含有組成物及びその製法 |
| US7264813B2 (en) * | 2003-09-24 | 2007-09-04 | Nikken Sohonsha Corporation | Therapeutic uses of Dunaliella powder |
| JP2006213613A (ja) * | 2005-02-02 | 2006-08-17 | Sun Chlorella Corp | 肝障害抑制剤 |
-
2008
- 2008-10-20 JP JP2008269530A patent/JP5733592B2/ja active Active
-
2009
- 2009-04-24 US US12/385,961 patent/US20100098720A1/en not_active Abandoned
-
2010
- 2010-08-09 US US12/852,704 patent/US20100303855A1/en not_active Abandoned
Non-Patent Citations (1)
| Title |
|---|
| JPN6013004107; Shaish A, et al: '9-cis beta-carotene-rich powder of the alga Dunaliella bardawil increases plasma HDL-cholesterol in' Atherosclerosis 189(1), 2006, 215-221 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2011105656A (ja) * | 2009-11-18 | 2011-06-02 | Microalgae Corporation | 脱共役タンパク質発現誘導剤及び冷え改善剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20100303855A1 (en) | 2010-12-02 |
| JP5733592B2 (ja) | 2015-06-10 |
| US20100098720A1 (en) | 2010-04-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Fetterman Jr et al. | Therapeutic potential of n-3 polyunsaturated fatty acids in disease | |
| Lavie et al. | Omega-3 polyunsaturated fatty acids and cardiovascular diseases | |
| Huang et al. | 5‐Caffeoylquinic acid decreases diet‐induced obesity in rats by modulating PPARα and LXRα transcription | |
| DK2800563T3 (en) | DPA Enriched Compositions of Multi-Saturated Omega-3 Fatty Acids in Free Acid Form | |
| O'Keefe Jr et al. | From Inuit to implementation: omega-3 fatty acids come of age | |
| WO2011097273A1 (en) | Methods and compositions for treating non-alcoholic fatty liver disease with docosahexaenoic acid and n-acetyl lcystenine | |
| Aluko et al. | Bioactive lipids | |
| EP2682116B1 (en) | Metabolic syndrome ameliorating agent | |
| CN104983737B (zh) | 药物组合物及其在制备防治动脉粥样硬化、血脂异常药物中的应用 | |
| JP5733592B2 (ja) | 脂質異常治療薬の副作用防止物質 | |
| JP6840671B2 (ja) | 一価不飽和脂肪酸組成物およびアテローム性動脈硬化症を処置するための使用 | |
| Kuo et al. | Caffeamide 36-13 regulates the antidiabetic and hypolipidemic signs of high-fat-fed mice on glucose transporter 4, AMPK phosphorylation, and regulated hepatic glucose production | |
| Yang et al. | Chlorella unsaturated fatty acids suppress high‐fat diet‐induced obesity in C57/BL6J mice | |
| JP2006273788A (ja) | Pparリガンド活性を有する植物由来の組成物 | |
| KR20100088007A (ko) | 율피 추출물을 포함하는 지방간의 예방 또는 치료용 조성물 | |
| JP2018127452A (ja) | 炎症を伴う肝疾患の治療または予防剤 | |
| JP5317175B2 (ja) | インスリン抵抗性改善剤 | |
| Nakamura et al. | Effects of cilostazol on lipid and fatty acid metabolism | |
| KR20190003570A (ko) | 안전한·안정된 플라스마로겐과 그 제제 및 인지증의 미병 상태의 판정 방법 | |
| JP2009114138A (ja) | コレステロール上昇抑制剤 | |
| Singh et al. | Association of High W-6/W-3 Ratio Paleolithic Style Diets and Risk of Cardiovascular Diseases and Other Chronic Diseases: Is the Tissue the Main Issue? | |
| Harris et al. | 26 Omega-3 Fatty Acids | |
| Du et al. | Conjugated Linolenic Acid (CLnA) vs Conjugated Linoleic Acid (CLA): A Comprehensive Review of Potential Advantages in Molecular Characteristics, Health Benefits, and Production Techniques | |
| Rohman et al. | An overview of Beta vulgaris L.: Its functions as antidyslipidemia | |
| CN117338870A (zh) | 一种辅助降血脂的软胶囊及其制备方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110422 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20110422 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130205 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130405 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20130611 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130906 |
|
| A911 | Transfer to examiner for re-examination before appeal (zenchi) |
Free format text: JAPANESE INTERMEDIATE CODE: A911 Effective date: 20130918 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20150216 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20150402 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5733592 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |