JP2005534429A - 塞栓症予防デバイス - Google Patents
塞栓症予防デバイス Download PDFInfo
- Publication number
- JP2005534429A JP2005534429A JP2004526448A JP2004526448A JP2005534429A JP 2005534429 A JP2005534429 A JP 2005534429A JP 2004526448 A JP2004526448 A JP 2004526448A JP 2004526448 A JP2004526448 A JP 2004526448A JP 2005534429 A JP2005534429 A JP 2005534429A
- Authority
- JP
- Japan
- Prior art keywords
- embolic protection
- protection device
- tube
- polymer
- embolism prevention
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 208000005189 Embolism Diseases 0.000 title claims abstract description 157
- 230000002265 prevention Effects 0.000 title claims abstract description 100
- 230000003073 embolic effect Effects 0.000 claims abstract description 133
- 229920000642 polymer Polymers 0.000 claims abstract description 127
- 239000000126 substance Substances 0.000 claims abstract description 34
- 230000000975 bioactive effect Effects 0.000 claims abstract description 26
- 239000003527 fibrinolytic agent Substances 0.000 claims abstract description 10
- 229960000103 thrombolytic agent Drugs 0.000 claims abstract description 10
- 239000000017 hydrogel Substances 0.000 claims description 42
- 238000000034 method Methods 0.000 claims description 40
- 238000012384 transportation and delivery Methods 0.000 claims description 39
- 229920002401 polyacrylamide Polymers 0.000 claims description 31
- 239000000203 mixture Substances 0.000 claims description 30
- 239000012867 bioactive agent Substances 0.000 claims description 29
- 239000004744 fabric Substances 0.000 claims description 20
- 239000000835 fiber Substances 0.000 claims description 19
- 239000002245 particle Substances 0.000 claims description 19
- 229920002635 polyurethane Polymers 0.000 claims description 17
- 239000004814 polyurethane Substances 0.000 claims description 17
- 239000007864 aqueous solution Substances 0.000 claims description 13
- 229920001577 copolymer Polymers 0.000 claims description 12
- 229920000728 polyester Polymers 0.000 claims description 12
- 229920000431 shape-memory polymer Polymers 0.000 claims description 11
- 239000006260 foam Substances 0.000 claims description 10
- 239000013598 vector Substances 0.000 claims description 10
- 210000000709 aorta Anatomy 0.000 claims description 9
- 229920001400 block copolymer Polymers 0.000 claims description 8
- 210000004351 coronary vessel Anatomy 0.000 claims description 8
- 230000036760 body temperature Effects 0.000 claims description 7
- 238000001914 filtration Methods 0.000 claims description 7
- 230000002411 adverse Effects 0.000 claims description 6
- 238000001727 in vivo Methods 0.000 claims description 5
- 239000003146 anticoagulant agent Substances 0.000 claims description 4
- 230000000694 effects Effects 0.000 claims description 4
- 239000003102 growth factor Substances 0.000 claims description 4
- 238000010438 heat treatment Methods 0.000 claims description 4
- 229920000331 Polyhydroxybutyrate Polymers 0.000 claims description 3
- 229940127219 anticoagulant drug Drugs 0.000 claims description 3
- 229940127218 antiplatelet drug Drugs 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 claims description 3
- 239000005015 poly(hydroxybutyrate) Substances 0.000 claims description 3
- 229920002451 polyvinyl alcohol Polymers 0.000 claims description 3
- BWLBGMIXKSTLSX-UHFFFAOYSA-N 2-hydroxyisobutyric acid Chemical compound CC(C)(O)C(O)=O BWLBGMIXKSTLSX-UHFFFAOYSA-N 0.000 claims description 2
- 108010010803 Gelatin Chemical class 0.000 claims description 2
- 229920001612 Hydroxyethyl starch Chemical class 0.000 claims description 2
- 229920000954 Polyglycolide Polymers 0.000 claims description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 claims description 2
- 229920000159 gelatin Chemical class 0.000 claims description 2
- 239000008273 gelatin Chemical class 0.000 claims description 2
- 235000019322 gelatine Nutrition 0.000 claims description 2
- 235000011852 gelatine desserts Nutrition 0.000 claims description 2
- 150000004676 glycans Chemical class 0.000 claims description 2
- 150000001261 hydroxy acids Chemical class 0.000 claims description 2
- 229940050526 hydroxyethylstarch Drugs 0.000 claims description 2
- 229920000747 poly(lactic acid) Polymers 0.000 claims description 2
- 229920002187 poly[N-2-(hydroxypropyl) methacrylamide] polymer Polymers 0.000 claims description 2
- 229920001610 polycaprolactone Polymers 0.000 claims description 2
- 239000004633 polyglycolic acid Substances 0.000 claims description 2
- 239000004626 polylactic acid Substances 0.000 claims description 2
- 229920001282 polysaccharide Polymers 0.000 claims description 2
- 239000005017 polysaccharide Substances 0.000 claims description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 claims description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims description 2
- 230000005779 cell damage Effects 0.000 claims 4
- 208000037887 cell injury Diseases 0.000 claims 1
- 238000003780 insertion Methods 0.000 claims 1
- 230000037431 insertion Effects 0.000 claims 1
- 230000001568 sexual effect Effects 0.000 claims 1
- 239000000463 material Substances 0.000 abstract description 67
- 108090000373 Tissue Plasminogen Activator Proteins 0.000 description 82
- 102000003978 Tissue Plasminogen Activator Human genes 0.000 description 82
- 229960000187 tissue plasminogen activator Drugs 0.000 description 81
- 239000000499 gel Substances 0.000 description 21
- 239000011148 porous material Substances 0.000 description 19
- 108010073385 Fibrin Proteins 0.000 description 18
- 102000009123 Fibrin Human genes 0.000 description 18
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 18
- 229950003499 fibrin Drugs 0.000 description 18
- 239000005020 polyethylene terephthalate Substances 0.000 description 16
- 229920000139 polyethylene terephthalate Polymers 0.000 description 15
- MWUXSHHQAYIFBG-UHFFFAOYSA-N Nitric oxide Chemical compound O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 14
- 238000004132 cross linking Methods 0.000 description 13
- 239000000243 solution Substances 0.000 description 13
- 238000012360 testing method Methods 0.000 description 13
- 210000004369 blood Anatomy 0.000 description 12
- 239000008280 blood Substances 0.000 description 12
- 239000003795 chemical substances by application Substances 0.000 description 12
- 239000011159 matrix material Substances 0.000 description 12
- 210000004204 blood vessel Anatomy 0.000 description 11
- 239000000306 component Substances 0.000 description 11
- 238000011144 upstream manufacturing Methods 0.000 description 11
- 230000000875 corresponding effect Effects 0.000 description 10
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 10
- 239000002953 phosphate buffered saline Substances 0.000 description 10
- 229920000578 graft copolymer Polymers 0.000 description 8
- 210000002216 heart Anatomy 0.000 description 8
- 239000002609 medium Substances 0.000 description 8
- 230000008961 swelling Effects 0.000 description 8
- 208000000913 Kidney Calculi Diseases 0.000 description 7
- 206010029148 Nephrolithiasis Diseases 0.000 description 7
- 208000007536 Thrombosis Diseases 0.000 description 7
- 230000008859 change Effects 0.000 description 7
- 238000004090 dissolution Methods 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 238000006116 polymerization reaction Methods 0.000 description 7
- 108090000623 proteins and genes Proteins 0.000 description 7
- -1 red blood cells Proteins 0.000 description 7
- 230000002829 reductive effect Effects 0.000 description 7
- 238000001356 surgical procedure Methods 0.000 description 7
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical group NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 6
- ROOXNKNUYICQNP-UHFFFAOYSA-N ammonium persulfate Chemical compound [NH4+].[NH4+].[O-]S(=O)(=O)OOS([O-])(=O)=O ROOXNKNUYICQNP-UHFFFAOYSA-N 0.000 description 6
- 238000013459 approach Methods 0.000 description 6
- 210000001367 artery Anatomy 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 230000000747 cardiac effect Effects 0.000 description 6
- 238000007675 cardiac surgery Methods 0.000 description 6
- 230000002612 cardiopulmonary effect Effects 0.000 description 6
- 230000015556 catabolic process Effects 0.000 description 6
- 239000002131 composite material Substances 0.000 description 6
- 150000001875 compounds Chemical class 0.000 description 6
- 238000006731 degradation reaction Methods 0.000 description 6
- 238000013461 design Methods 0.000 description 6
- 239000012530 fluid Substances 0.000 description 6
- 238000000338 in vitro Methods 0.000 description 6
- 229920002959 polymer blend Polymers 0.000 description 6
- 239000000523 sample Substances 0.000 description 6
- 238000011282 treatment Methods 0.000 description 6
- 230000002792 vascular Effects 0.000 description 6
- 239000000853 adhesive Substances 0.000 description 5
- 230000001070 adhesive effect Effects 0.000 description 5
- 210000004556 brain Anatomy 0.000 description 5
- 210000004027 cell Anatomy 0.000 description 5
- 238000000576 coating method Methods 0.000 description 5
- 238000010586 diagram Methods 0.000 description 5
- 238000010790 dilution Methods 0.000 description 5
- 239000012895 dilution Substances 0.000 description 5
- 230000010102 embolization Effects 0.000 description 5
- 239000012634 fragment Substances 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 230000007576 microinfarct Effects 0.000 description 5
- 230000002093 peripheral effect Effects 0.000 description 5
- 229920001184 polypeptide Polymers 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 102000004196 processed proteins & peptides Human genes 0.000 description 5
- 108090000765 processed proteins & peptides Proteins 0.000 description 5
- 102000004169 proteins and genes Human genes 0.000 description 5
- KWYHDKDOAIKMQN-UHFFFAOYSA-N N,N,N',N'-tetramethylethylenediamine Chemical compound CN(C)CCN(C)C KWYHDKDOAIKMQN-UHFFFAOYSA-N 0.000 description 4
- 208000012902 Nervous system disease Diseases 0.000 description 4
- 208000025966 Neurological disease Diseases 0.000 description 4
- 239000004677 Nylon Substances 0.000 description 4
- 208000010378 Pulmonary Embolism Diseases 0.000 description 4
- 230000009286 beneficial effect Effects 0.000 description 4
- 229920002988 biodegradable polymer Polymers 0.000 description 4
- 239000004621 biodegradable polymer Substances 0.000 description 4
- 238000006243 chemical reaction Methods 0.000 description 4
- 239000011248 coating agent Substances 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 238000010828 elution Methods 0.000 description 4
- 238000001125 extrusion Methods 0.000 description 4
- 239000011521 glass Substances 0.000 description 4
- 230000036571 hydration Effects 0.000 description 4
- 238000006703 hydration reaction Methods 0.000 description 4
- 229920001477 hydrophilic polymer Polymers 0.000 description 4
- 239000007943 implant Substances 0.000 description 4
- 208000015181 infectious disease Diseases 0.000 description 4
- 230000007774 longterm Effects 0.000 description 4
- 208000010125 myocardial infarction Diseases 0.000 description 4
- 229920001778 nylon Polymers 0.000 description 4
- 229940012957 plasmin Drugs 0.000 description 4
- 229920000570 polyether Polymers 0.000 description 4
- 229920000247 superabsorbent polymer Polymers 0.000 description 4
- 229920002554 vinyl polymer Polymers 0.000 description 4
- 239000013603 viral vector Substances 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- 239000004971 Cross linker Substances 0.000 description 3
- 238000002965 ELISA Methods 0.000 description 3
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- 239000004721 Polyphenylene oxide Substances 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 206010000891 acute myocardial infarction Diseases 0.000 description 3
- 229910001870 ammonium persulfate Inorganic materials 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 239000007975 buffered saline Substances 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 230000006735 deficit Effects 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 238000000605 extraction Methods 0.000 description 3
- 125000000524 functional group Chemical group 0.000 description 3
- 230000014509 gene expression Effects 0.000 description 3
- 229960002897 heparin Drugs 0.000 description 3
- 229920000669 heparin Polymers 0.000 description 3
- 238000002513 implantation Methods 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- 239000000178 monomer Substances 0.000 description 3
- 238000000465 moulding Methods 0.000 description 3
- 230000000926 neurological effect Effects 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 210000001147 pulmonary artery Anatomy 0.000 description 3
- 239000004575 stone Substances 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 208000024891 symptom Diseases 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- 230000002537 thrombolytic effect Effects 0.000 description 3
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 3
- 210000001635 urinary tract Anatomy 0.000 description 3
- 238000007631 vascular surgery Methods 0.000 description 3
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- REKYPYSUBKSCAT-UHFFFAOYSA-N 3-hydroxypentanoic acid Chemical compound CCC(O)CC(O)=O REKYPYSUBKSCAT-UHFFFAOYSA-N 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 2
- 229920004934 Dacron® Polymers 0.000 description 2
- 108010001336 Horseradish Peroxidase Proteins 0.000 description 2
- 206010033799 Paralysis Diseases 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 102000003990 Urokinase-type plasminogen activator Human genes 0.000 description 2
- 108090000435 Urokinase-type plasminogen activator Proteins 0.000 description 2
- 208000012931 Urologic disease Diseases 0.000 description 2
- 108010073929 Vascular Endothelial Growth Factor A Proteins 0.000 description 2
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 description 2
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 2
- 206010047571 Visual impairment Diseases 0.000 description 2
- 206010052428 Wound Diseases 0.000 description 2
- 208000027418 Wounds and injury Diseases 0.000 description 2
- OIRDTQYFTABQOQ-KQYNXXCUSA-N adenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O OIRDTQYFTABQOQ-KQYNXXCUSA-N 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 229920000249 biocompatible polymer Polymers 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- 239000006172 buffering agent Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 210000000038 chest Anatomy 0.000 description 2
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 2
- 230000004087 circulation Effects 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 230000006835 compression Effects 0.000 description 2
- 238000013270 controlled release Methods 0.000 description 2
- 239000003431 cross linking reagent Substances 0.000 description 2
- 230000034994 death Effects 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 210000003743 erythrocyte Anatomy 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 239000006261 foam material Substances 0.000 description 2
- 210000003709 heart valve Anatomy 0.000 description 2
- 210000005260 human cell Anatomy 0.000 description 2
- 238000003384 imaging method Methods 0.000 description 2
- 238000010348 incorporation Methods 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- 230000001788 irregular Effects 0.000 description 2
- 210000002414 leg Anatomy 0.000 description 2
- 210000004072 lung Anatomy 0.000 description 2
- 238000001000 micrograph Methods 0.000 description 2
- IDSXLJLXYMLSJM-UHFFFAOYSA-N morpholine;propane-1-sulfonic acid Chemical compound C1COCCN1.CCCS(O)(=O)=O IDSXLJLXYMLSJM-UHFFFAOYSA-N 0.000 description 2
- 210000004165 myocardium Anatomy 0.000 description 2
- 230000000474 nursing effect Effects 0.000 description 2
- 239000013612 plasmid Substances 0.000 description 2
- 239000000106 platelet aggregation inhibitor Substances 0.000 description 2
- 229920000515 polycarbonate Polymers 0.000 description 2
- 239000004417 polycarbonate Substances 0.000 description 2
- 229920002338 polyhydroxyethylmethacrylate Polymers 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 239000004583 superabsorbent polymers (SAPs) Substances 0.000 description 2
- 208000014001 urinary system disease Diseases 0.000 description 2
- 229960005356 urokinase Drugs 0.000 description 2
- 208000019553 vascular disease Diseases 0.000 description 2
- 210000005166 vasculature Anatomy 0.000 description 2
- 208000029257 vision disease Diseases 0.000 description 2
- 230000004393 visual impairment Effects 0.000 description 2
- 239000002699 waste material Substances 0.000 description 2
- IHPYMWDTONKSCO-UHFFFAOYSA-N 2,2'-piperazine-1,4-diylbisethanesulfonic acid Chemical compound OS(=O)(=O)CCN1CCN(CCS(O)(=O)=O)CC1 IHPYMWDTONKSCO-UHFFFAOYSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- CQVWXNBVRLKXPE-UHFFFAOYSA-N 2-octyl cyanoacrylate Chemical compound CCCCCCC(C)OC(=O)C(=C)C#N CQVWXNBVRLKXPE-UHFFFAOYSA-N 0.000 description 1
- BUOYTFVLNZIELF-UHFFFAOYSA-N 2-phenyl-1h-indole-4,6-dicarboximidamide Chemical compound N1C2=CC(C(=N)N)=CC(C(N)=N)=C2C=C1C1=CC=CC=C1 BUOYTFVLNZIELF-UHFFFAOYSA-N 0.000 description 1
- BSYNRYMUTXBXSQ-FOQJRBATSA-N 59096-14-9 Chemical compound CC(=O)OC1=CC=CC=C1[14C](O)=O BSYNRYMUTXBXSQ-FOQJRBATSA-N 0.000 description 1
- 208000012639 Balance disease Diseases 0.000 description 1
- 239000004604 Blowing Agent Substances 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 239000002126 C01EB10 - Adenosine Substances 0.000 description 1
- 241001631457 Cannula Species 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 206010010904 Convulsion Diseases 0.000 description 1
- 229920001651 Cyanoacrylate Polymers 0.000 description 1
- 229920000045 Dermatan sulfate Polymers 0.000 description 1
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 1
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical class [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 108010080379 Fibrin Tissue Adhesive Proteins 0.000 description 1
- 102000008946 Fibrinogen Human genes 0.000 description 1
- 108010049003 Fibrinogen Proteins 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- 208000032843 Hemorrhage Diseases 0.000 description 1
- 108090000481 Heparin Cofactor II Proteins 0.000 description 1
- 102100030500 Heparin cofactor 2 Human genes 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101001078143 Homo sapiens Integrin alpha-IIb Proteins 0.000 description 1
- 101000801481 Homo sapiens Tissue-type plasminogen activator Proteins 0.000 description 1
- WOBHKFSMXKNTIM-UHFFFAOYSA-N Hydroxyethyl methacrylate Chemical compound CC(=C)C(=O)OCCO WOBHKFSMXKNTIM-UHFFFAOYSA-N 0.000 description 1
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 1
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 1
- 206010061216 Infarction Diseases 0.000 description 1
- 102100025306 Integrin alpha-IIb Human genes 0.000 description 1
- 206010060820 Joint injury Diseases 0.000 description 1
- 206010049694 Left Ventricular Dysfunction Diseases 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- NOOJLZTTWSNHOX-UWVGGRQHSA-N Melanostatin Chemical compound NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@@H]1CCCN1 NOOJLZTTWSNHOX-UWVGGRQHSA-N 0.000 description 1
- 208000028389 Nerve injury Diseases 0.000 description 1
- 229910000990 Ni alloy Inorganic materials 0.000 description 1
- SNIOPGDIGTZGOP-UHFFFAOYSA-N Nitroglycerin Chemical compound [O-][N+](=O)OCC(O[N+]([O-])=O)CO[N+]([O-])=O SNIOPGDIGTZGOP-UHFFFAOYSA-N 0.000 description 1
- 239000000006 Nitroglycerin Substances 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 102000004264 Osteopontin Human genes 0.000 description 1
- 108010081689 Osteopontin Proteins 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 208000002193 Pain Diseases 0.000 description 1
- 229930040373 Paraformaldehyde Natural products 0.000 description 1
- 208000007542 Paresis Diseases 0.000 description 1
- 102000013566 Plasminogen Human genes 0.000 description 1
- 108010051456 Plasminogen Proteins 0.000 description 1
- 102100031574 Platelet glycoprotein 4 Human genes 0.000 description 1
- 101710202087 Platelet glycoprotein 4 Proteins 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920005830 Polyurethane Foam Polymers 0.000 description 1
- 241000932075 Priacanthus hamrur Species 0.000 description 1
- 101710145796 Staphylokinase Proteins 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 108010023197 Streptokinase Proteins 0.000 description 1
- 101710172711 Structural protein Proteins 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 108010051181 TNK-tissue plasminogen activator Proteins 0.000 description 1
- 229920004933 Terylene® Polymers 0.000 description 1
- 208000001435 Thromboembolism Diseases 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 102100028262 U6 snRNA-associated Sm-like protein LSm4 Human genes 0.000 description 1
- 208000026723 Urinary tract disease Diseases 0.000 description 1
- 206010047249 Venous thrombosis Diseases 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- YWXYYJSYQOXTPL-JGWLITMVSA-N [(3r,3ar,6s,6as)-3-hydroxy-2,3,3a,5,6,6a-hexahydrofuro[3,2-b]furan-6-yl] nitrate Chemical compound [O-][N+](=O)O[C@H]1CO[C@@H]2[C@H](O)CO[C@@H]21 YWXYYJSYQOXTPL-JGWLITMVSA-N 0.000 description 1
- YUZKVBQMDLFRSU-UHFFFAOYSA-N [Na+].[Na+].C[NH-].C[NH-] Chemical compound [Na+].[Na+].C[NH-].C[NH-] YUZKVBQMDLFRSU-UHFFFAOYSA-N 0.000 description 1
- HZEWFHLRYVTOIW-UHFFFAOYSA-N [Ti].[Ni] Chemical compound [Ti].[Ni] HZEWFHLRYVTOIW-UHFFFAOYSA-N 0.000 description 1
- 239000002250 absorbent Substances 0.000 description 1
- 230000002745 absorbent Effects 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 229960005305 adenosine Drugs 0.000 description 1
- 238000002266 amputation Methods 0.000 description 1
- 238000002583 angiography Methods 0.000 description 1
- 238000002399 angioplasty Methods 0.000 description 1
- 230000000582 anti-calcifiying effect Effects 0.000 description 1
- 230000002965 anti-thrombogenic effect Effects 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 229960005348 antithrombin iii Drugs 0.000 description 1
- 210000000702 aorta abdominal Anatomy 0.000 description 1
- 210000002376 aorta thoracic Anatomy 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 238000003491 array Methods 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 239000000560 biocompatible material Substances 0.000 description 1
- 229920001222 biopolymer Polymers 0.000 description 1
- 208000034158 bleeding Diseases 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 239000012503 blood component Substances 0.000 description 1
- 238000000071 blow moulding Methods 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- 210000004958 brain cell Anatomy 0.000 description 1
- QXDMQSPYEZFLGF-UHFFFAOYSA-L calcium oxalate Chemical compound [Ca+2].[O-]C(=O)C([O-])=O QXDMQSPYEZFLGF-UHFFFAOYSA-L 0.000 description 1
- 238000003490 calendering Methods 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 210000001168 carotid artery common Anatomy 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- 230000002490 cerebral effect Effects 0.000 description 1
- 235000012000 cholesterol Nutrition 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 230000001112 coagulating effect Effects 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 238000000748 compression moulding Methods 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 230000003750 conditioning effect Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 230000001276 controlling effect Effects 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- NLCKLZIHJQEMCU-UHFFFAOYSA-N cyano prop-2-enoate Chemical class C=CC(=O)OC#N NLCKLZIHJQEMCU-UHFFFAOYSA-N 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- AVJBPWGFOQAPRH-FWMKGIEWSA-L dermatan sulfate Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@H](OS([O-])(=O)=O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](C([O-])=O)O1 AVJBPWGFOQAPRH-FWMKGIEWSA-L 0.000 description 1
- 229940051593 dermatan sulfate Drugs 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 230000010339 dilation Effects 0.000 description 1
- 229910001882 dioxygen Inorganic materials 0.000 description 1
- 238000003618 dip coating Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000007922 dissolution test Methods 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000005553 drilling Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 230000004064 dysfunction Effects 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- 229940012952 fibrinogen Drugs 0.000 description 1
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 1
- 238000005187 foaming Methods 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 238000001415 gene therapy Methods 0.000 description 1
- 229960003711 glyceryl trinitrate Drugs 0.000 description 1
- 229940125672 glycoprotein IIb/IIIa inhibitor Drugs 0.000 description 1
- 230000035876 healing Effects 0.000 description 1
- 210000004394 hip joint Anatomy 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 239000012729 immediate-release (IR) formulation Substances 0.000 description 1
- 230000007574 infarction Effects 0.000 description 1
- 239000003999 initiator Substances 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 238000001746 injection moulding Methods 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 208000028867 ischemia Diseases 0.000 description 1
- 238000005304 joining Methods 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 238000012153 long-term therapy Methods 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 230000002101 lytic effect Effects 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000000386 microscopy Methods 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 238000012978 minimally invasive surgical procedure Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 230000002107 myocardial effect Effects 0.000 description 1
- ZIUHHBKFKCYYJD-UHFFFAOYSA-N n,n'-methylenebisacrylamide Chemical compound C=CC(=O)NCNC(=O)C=C ZIUHHBKFKCYYJD-UHFFFAOYSA-N 0.000 description 1
- 230000008764 nerve damage Effects 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 150000002823 nitrates Chemical class 0.000 description 1
- 230000001453 nonthrombogenic effect Effects 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 102000039446 nucleic acids Human genes 0.000 description 1
- 108020004707 nucleic acids Proteins 0.000 description 1
- 150000007523 nucleic acids Chemical class 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000013307 optical fiber Substances 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 230000002018 overexpression Effects 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 108010085108 pamiteplase Proteins 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 229920002866 paraformaldehyde Polymers 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 239000013618 particulate matter Substances 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000010106 peripheral embolization Effects 0.000 description 1
- 230000002572 peristaltic effect Effects 0.000 description 1
- 102000013415 peroxidase activity proteins Human genes 0.000 description 1
- 108040007629 peroxidase activity proteins Proteins 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 239000002570 phosphodiesterase III inhibitor Substances 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 229920001692 polycarbonate urethane Polymers 0.000 description 1
- 229920005646 polycarboxylate Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 238000010094 polymer processing Methods 0.000 description 1
- 239000011496 polyurethane foam Substances 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 238000003672 processing method Methods 0.000 description 1
- 230000000750 progressive effect Effects 0.000 description 1
- 230000017854 proteolysis Effects 0.000 description 1
- 238000011555 rabbit model Methods 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 230000010410 reperfusion Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 210000003752 saphenous vein Anatomy 0.000 description 1
- 238000004513 sizing Methods 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- KXDHHKIAGHRLRO-UHFFFAOYSA-N sodium methylazanide Chemical compound [Na+].[NH-]C KXDHHKIAGHRLRO-UHFFFAOYSA-N 0.000 description 1
- 239000002993 sponge (artificial) Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 229960005202 streptokinase Drugs 0.000 description 1
- 210000003270 subclavian artery Anatomy 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 239000003894 surgical glue Substances 0.000 description 1
- 230000002522 swelling effect Effects 0.000 description 1
- 238000007910 systemic administration Methods 0.000 description 1
- 238000012385 systemic delivery Methods 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 239000004753 textile Substances 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 238000011287 therapeutic dose Methods 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- 230000001732 thrombotic effect Effects 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 231100000816 toxic dose Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 101150010939 tpa gene Proteins 0.000 description 1
- 238000002627 tracheal intubation Methods 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 210000000626 ureter Anatomy 0.000 description 1
- 230000004865 vascular response Effects 0.000 description 1
- 229940124549 vasodilator Drugs 0.000 description 1
- 239000003071 vasodilator agent Substances 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 238000011179 visual inspection Methods 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
- 238000007794 visualization technique Methods 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 239000011800 void material Substances 0.000 description 1
- PJVWKTKQMONHTI-UHFFFAOYSA-N warfarin Chemical compound OC=1C2=CC=CC=C2OC(=O)C=1C(CC(=O)C)C1=CC=CC=C1 PJVWKTKQMONHTI-UHFFFAOYSA-N 0.000 description 1
- 229960005080 warfarin Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12027—Type of occlusion
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12027—Type of occlusion
- A61B17/12036—Type of occlusion partial occlusion
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12099—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder
- A61B17/12109—Occluding by internal devices, e.g. balloons or releasable wires characterised by the location of the occluder in a blood vessel
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/12168—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device having a mesh structure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/12168—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device having a mesh structure
- A61B17/12172—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device having a mesh structure having a pre-set deployed three-dimensional shape
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/12—Surgical instruments, devices or methods for ligaturing or otherwise compressing tubular parts of the body, e.g. blood vessels or umbilical cord
- A61B17/12022—Occluding by internal devices, e.g. balloons or releasable wires
- A61B17/12131—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device
- A61B17/12181—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device formed by fluidized, gelatinous or cellular remodelable materials, e.g. embolic liquids, foams or extracellular matrices
- A61B17/1219—Occluding by internal devices, e.g. balloons or releasable wires characterised by the type of occluding device formed by fluidized, gelatinous or cellular remodelable materials, e.g. embolic liquids, foams or extracellular matrices expandable in contact with liquids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/04—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
- A61F2002/016—Filters implantable into blood vessels made from wire-like elements
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/01—Filters implantable into blood vessels
- A61F2002/018—Filters implantable into blood vessels made from tubes or sheets of material, e.g. by etching or laser-cutting
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0063—Three-dimensional shapes
- A61F2230/0073—Quadric-shaped
- A61F2230/008—Quadric-shaped paraboloidal
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0063—Three-dimensional shapes
- A61F2230/0091—Three-dimensional shapes helically-coiled or spirally-coiled, i.e. having a 2-D spiral cross-section
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0058—Additional features; Implant or prostheses properties not otherwise provided for
- A61F2250/0067—Means for introducing or releasing pharmaceutical products into the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/36—Materials or treatment for tissue regeneration for embolization or occlusion, e.g. vaso-occlusive compositions or devices
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Surgery (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Vascular Medicine (AREA)
- Public Health (AREA)
- Heart & Thoracic Surgery (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- Medical Informatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Reproductive Health (AREA)
- Epidemiology (AREA)
- Cardiology (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Transplantation (AREA)
- Surgical Instruments (AREA)
- Materials For Medical Uses (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- External Artificial Organs (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US40034102P | 2002-08-01 | 2002-08-01 | |
| US10/414,909 US7303575B2 (en) | 2002-08-01 | 2003-04-16 | Embolism protection devices |
| PCT/US2003/024459 WO2004012587A2 (en) | 2002-08-01 | 2003-08-01 | Embolism protection devices |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2005534429A true JP2005534429A (ja) | 2005-11-17 |
| JP2005534429A5 JP2005534429A5 (enExample) | 2006-09-07 |
Family
ID=31498612
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2004526448A Pending JP2005534429A (ja) | 2002-08-01 | 2003-08-01 | 塞栓症予防デバイス |
Country Status (5)
| Country | Link |
|---|---|
| US (2) | US7303575B2 (enExample) |
| EP (1) | EP1545322B1 (enExample) |
| JP (1) | JP2005534429A (enExample) |
| AU (1) | AU2003257182A1 (enExample) |
| WO (1) | WO2004012587A2 (enExample) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010505526A (ja) * | 2006-10-04 | 2010-02-25 | ボストン サイエンティフィック リミテッド | アテローム硬化性プラークを治療するための装置及び方法 |
| JP2012521258A (ja) * | 2009-03-25 | 2012-09-13 | リシブロック リミテッド | フィルター装置及びそれを使用する方法 |
| US10368974B2 (en) | 2014-10-27 | 2019-08-06 | Lithiblock Ltd. | Gallbladder implants and systems and methods for the delivery thereof |
| JP2021013745A (ja) * | 2019-07-11 | 2021-02-12 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 再挿入のための血塊回収装置の洗浄 |
Families Citing this family (160)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7335220B2 (en) | 2004-11-05 | 2008-02-26 | Access Closure, Inc. | Apparatus and methods for sealing a vascular puncture |
| US7314477B1 (en) * | 1998-09-25 | 2008-01-01 | C.R. Bard Inc. | Removable embolus blood clot filter and filter delivery unit |
| US6575997B1 (en) | 1999-12-23 | 2003-06-10 | Endovascular Technologies, Inc. | Embolic basket |
| US6402771B1 (en) | 1999-12-23 | 2002-06-11 | Guidant Endovascular Solutions | Snare |
| US6660021B1 (en) | 1999-12-23 | 2003-12-09 | Advanced Cardiovascular Systems, Inc. | Intravascular device and system |
| US6695813B1 (en) | 1999-12-30 | 2004-02-24 | Advanced Cardiovascular Systems, Inc. | Embolic protection devices |
| US7918820B2 (en) | 1999-12-30 | 2011-04-05 | Advanced Cardiovascular Systems, Inc. | Device for, and method of, blocking emboli in vessels such as blood arteries |
| US6540722B1 (en) | 1999-12-30 | 2003-04-01 | Advanced Cardiovascular Systems, Inc. | Embolic protection devices |
| US6964670B1 (en) | 2000-07-13 | 2005-11-15 | Advanced Cardiovascular Systems, Inc. | Embolic protection guide wire |
| US6537294B1 (en) | 2000-10-17 | 2003-03-25 | Advanced Cardiovascular Systems, Inc. | Delivery systems for embolic filter devices |
| US6893451B2 (en) | 2000-11-09 | 2005-05-17 | Advanced Cardiovascular Systems, Inc. | Apparatus for capturing objects beyond an operative site utilizing a capture device delivered on a medical guide wire |
| US6506203B1 (en) | 2000-12-19 | 2003-01-14 | Advanced Cardiovascular Systems, Inc. | Low profile sheathless embolic protection system |
| US7044952B2 (en) | 2001-06-06 | 2006-05-16 | Sdgi Holdings, Inc. | Dynamic multilock anterior cervical plate system having non-detachably fastened and moveable segments |
| US6599307B1 (en) | 2001-06-29 | 2003-07-29 | Advanced Cardiovascular Systems, Inc. | Filter device for embolic protection systems |
| US7338510B2 (en) | 2001-06-29 | 2008-03-04 | Advanced Cardiovascular Systems, Inc. | Variable thickness embolic filtering devices and method of manufacturing the same |
| US6638294B1 (en) | 2001-08-30 | 2003-10-28 | Advanced Cardiovascular Systems, Inc. | Self furling umbrella frame for carotid filter |
| US6592606B2 (en) | 2001-08-31 | 2003-07-15 | Advanced Cardiovascular Systems, Inc. | Hinged short cage for an embolic protection device |
| US8262689B2 (en) | 2001-09-28 | 2012-09-11 | Advanced Cardiovascular Systems, Inc. | Embolic filtering devices |
| US7241304B2 (en) | 2001-12-21 | 2007-07-10 | Advanced Cardiovascular Systems, Inc. | Flexible and conformable embolic filtering devices |
| US9204956B2 (en) | 2002-02-20 | 2015-12-08 | C. R. Bard, Inc. | IVC filter with translating hooks |
| US6887258B2 (en) | 2002-06-26 | 2005-05-03 | Advanced Cardiovascular Systems, Inc. | Embolic filtering devices for bifurcated vessels |
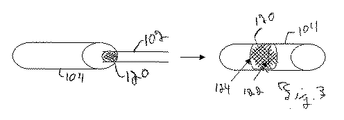
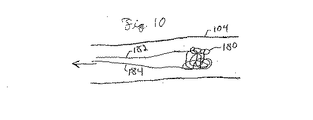
| US7303575B2 (en) * | 2002-08-01 | 2007-12-04 | Lumen Biomedical, Inc. | Embolism protection devices |
| US7252675B2 (en) | 2002-09-30 | 2007-08-07 | Advanced Cardiovascular, Inc. | Embolic filtering devices |
| US7331973B2 (en) | 2002-09-30 | 2008-02-19 | Avdanced Cardiovascular Systems, Inc. | Guide wire with embolic filtering attachment |
| US20040088000A1 (en) | 2002-10-31 | 2004-05-06 | Muller Paul F. | Single-wire expandable cages for embolic filtering devices |
| US8591540B2 (en) | 2003-02-27 | 2013-11-26 | Abbott Cardiovascular Systems Inc. | Embolic filtering devices |
| US8048042B2 (en) * | 2003-07-22 | 2011-11-01 | Medtronic Vascular, Inc. | Medical articles incorporating surface capillary fiber |
| US7879062B2 (en) * | 2003-07-22 | 2011-02-01 | Lumen Biomedical, Inc. | Fiber based embolism protection device |
| US20070135907A1 (en) | 2003-10-02 | 2007-06-14 | The Regents Of The University Of California | Stent with expandable foam |
| US7892251B1 (en) | 2003-11-12 | 2011-02-22 | Advanced Cardiovascular Systems, Inc. | Component for delivering and locking a medical device to a guide wire |
| US20050107867A1 (en) * | 2003-11-17 | 2005-05-19 | Taheri Syde A. | Temporary absorbable venous occlusive stent and superficial vein treatment method |
| EP1557128B1 (en) * | 2004-01-23 | 2007-03-07 | Cathenet Corporation | Atherectomy head and atherectomy catheter using the same |
| US8092483B2 (en) | 2004-03-06 | 2012-01-10 | Medtronic, Inc. | Steerable device having a corewire within a tube and combination with a functional medical component |
| US7678129B1 (en) | 2004-03-19 | 2010-03-16 | Advanced Cardiovascular Systems, Inc. | Locking component for an embolic filter assembly |
| US7976516B2 (en) * | 2004-06-25 | 2011-07-12 | Lumen Biomedical, Inc. | Medical device having mechanically interlocked segments |
| US20060020269A1 (en) * | 2004-07-20 | 2006-01-26 | Eric Cheng | Device to aid in stone removal and laser lithotripsy |
| US7704267B2 (en) | 2004-08-04 | 2010-04-27 | C. R. Bard, Inc. | Non-entangling vena cava filter |
| US20060047301A1 (en) * | 2004-09-02 | 2006-03-02 | Ogle Matthew F | Emboli removal system with oxygenated flow |
| US7927346B2 (en) * | 2004-09-10 | 2011-04-19 | Stryker Corporation | Diversion device to increase cerebral blood flow |
| US8262693B2 (en) * | 2004-11-05 | 2012-09-11 | Accessclosure, Inc. | Apparatus and methods for sealing a vascular puncture |
| US7794473B2 (en) * | 2004-11-12 | 2010-09-14 | C.R. Bard, Inc. | Filter delivery system |
| WO2006064056A2 (en) * | 2004-12-16 | 2006-06-22 | Nolabs Ab | Anti-pathogen and anti-cancer filtering device and method comprising nitric oxide |
| EP1681068A1 (en) * | 2004-12-16 | 2006-07-19 | NOLabs AB | Anti-pathogen and anti-cancer filtering device and method comprising nitric oxide |
| US20060161241A1 (en) * | 2005-01-14 | 2006-07-20 | Denise Barbut | Methods and devices for treating aortic atheroma |
| EP1841368B1 (en) * | 2005-01-25 | 2015-06-10 | Covidien LP | Structures for permanent occlusion of a hollow anatomical structure |
| US8267954B2 (en) | 2005-02-04 | 2012-09-18 | C. R. Bard, Inc. | Vascular filter with sensing capability |
| US8945169B2 (en) * | 2005-03-15 | 2015-02-03 | Cook Medical Technologies Llc | Embolic protection device |
| US9259305B2 (en) | 2005-03-31 | 2016-02-16 | Abbott Cardiovascular Systems Inc. | Guide wire locking mechanism for rapid exchange and other catheter systems |
| US20060224178A1 (en) * | 2005-04-05 | 2006-10-05 | Boston Scientific Scimed, Inc. | Expandable medical retrieval device and related methods of use |
| WO2006124405A2 (en) | 2005-05-12 | 2006-11-23 | C.R. Bard Inc. | Removable embolus blood clot filter |
| US12115057B2 (en) | 2005-05-12 | 2024-10-15 | C.R. Bard, Inc. | Tubular filter |
| US20080172066A9 (en) * | 2005-07-29 | 2008-07-17 | Galdonik Jason A | Embolectomy procedures with a device comprising a polymer and devices with polymer matrices and supports |
| US8062327B2 (en) | 2005-08-09 | 2011-11-22 | C. R. Bard, Inc. | Embolus blood clot filter and delivery system |
| US20070112372A1 (en) * | 2005-11-17 | 2007-05-17 | Stephen Sosnowski | Biodegradable vascular filter |
| US9131999B2 (en) | 2005-11-18 | 2015-09-15 | C.R. Bard Inc. | Vena cava filter with filament |
| US8052714B2 (en) | 2005-11-22 | 2011-11-08 | Medtronic Vascular, Inc. | Radiopaque fibers and filtration matrices |
| JP2009519049A (ja) * | 2005-12-02 | 2009-05-14 | シー・アール・バード・インコーポレイテツド | 螺旋形大静脈フィルタ |
| EP1986707A2 (en) * | 2006-01-30 | 2008-11-05 | Surgica Corporation | Compressible intravascular embolization particles and related methods and delivery systems |
| WO2007090130A2 (en) * | 2006-01-30 | 2007-08-09 | Surgica Corporation | Porous intravascular embolization particles and related methods |
| US20070185520A1 (en) * | 2006-02-07 | 2007-08-09 | Boston Scientific Scimed, Inc. | Detachable medical immobilization device and related methods of use |
| US20070203564A1 (en) * | 2006-02-28 | 2007-08-30 | Boston Scientific Scimed, Inc. | Biodegradable implants having accelerated biodegradation properties in vivo |
| US8795709B2 (en) | 2006-03-29 | 2014-08-05 | Incept Llc | Superabsorbent, freeze dried hydrogels for medical applications |
| US7846175B2 (en) | 2006-04-03 | 2010-12-07 | Medrad, Inc. | Guidewire and collapsable filter system |
| US9017361B2 (en) | 2006-04-20 | 2015-04-28 | Covidien Lp | Occlusive implant and methods for hollow anatomical structure |
| WO2007133366A2 (en) | 2006-05-02 | 2007-11-22 | C. R. Bard, Inc. | Vena cava filter formed from a sheet |
| US20100030251A1 (en) * | 2006-05-24 | 2010-02-04 | Mayo Foundation For Medical Education And Research | Devices and methods for crossing chronic total occlusions |
| US9326842B2 (en) | 2006-06-05 | 2016-05-03 | C. R . Bard, Inc. | Embolus blood clot filter utilizable with a single delivery system or a single retrieval system in one of a femoral or jugular access |
| US8162970B2 (en) | 2006-07-19 | 2012-04-24 | Novate Medical Limited | Vascular filter |
| US9339367B2 (en) | 2006-09-11 | 2016-05-17 | Edwards Lifesciences Ag | Embolic deflection device |
| US9480548B2 (en) | 2006-09-11 | 2016-11-01 | Edwards Lifesciences Ag | Embolic protection device and method of use |
| US8460335B2 (en) | 2006-09-11 | 2013-06-11 | Embrella Cardiovascular, Inc. | Method of deflecting emboli from the cerebral circulation |
| US8216209B2 (en) | 2007-05-31 | 2012-07-10 | Abbott Cardiovascular Systems Inc. | Method and apparatus for delivering an agent to a kidney |
| US7867273B2 (en) | 2007-06-27 | 2011-01-11 | Abbott Laboratories | Endoprostheses for peripheral arteries and other body vessels |
| US20090069790A1 (en) * | 2007-09-07 | 2009-03-12 | Edward Maxwell Yokley | Surface properties of polymeric materials with nanoscale functional coating |
| US8962097B1 (en) | 2007-09-07 | 2015-02-24 | Edward Maxwell Yokley | Surface properties of polymeric materials with nanoscale functional coating |
| US9138307B2 (en) | 2007-09-14 | 2015-09-22 | Cook Medical Technologies Llc | Expandable device for treatment of a stricture in a body vessel |
| US9034007B2 (en) | 2007-09-21 | 2015-05-19 | Insera Therapeutics, Inc. | Distal embolic protection devices with a variable thickness microguidewire and methods for their use |
| JP2011502582A (ja) | 2007-11-02 | 2011-01-27 | インセプト,エルエルシー | 血管穿刺を塞ぐ装置および方法 |
| US20090287120A1 (en) | 2007-12-18 | 2009-11-19 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Circulatory monitoring systems and methods |
| US9717896B2 (en) | 2007-12-18 | 2017-08-01 | Gearbox, Llc | Treatment indications informed by a priori implant information |
| US8636670B2 (en) | 2008-05-13 | 2014-01-28 | The Invention Science Fund I, Llc | Circulatory monitoring systems and methods |
| US20100152646A1 (en) * | 2008-02-29 | 2010-06-17 | Reshma Girijavallabhan | Intravitreal injection device and method |
| JP5391407B2 (ja) * | 2008-04-03 | 2014-01-15 | クック・メディカル・テクノロジーズ・リミテッド・ライアビリティ・カンパニー | 自浄式装置、システムおよびその使用方法 |
| EP2303384B1 (en) | 2008-06-23 | 2015-08-12 | Lumen Biomedical, Inc. | Embolic protection during percutaneous heart valve replacement and similar procedures |
| US8070694B2 (en) | 2008-07-14 | 2011-12-06 | Medtronic Vascular, Inc. | Fiber based medical devices and aspiration catheters |
| CN102143996A (zh) * | 2008-10-30 | 2011-08-03 | 大卫·刘 | 微球形多孔可生物相容支架及制造该支架的方法和装置 |
| BRPI0923160A2 (pt) * | 2008-12-19 | 2016-02-10 | Tyco Healthcare Croup Lp | método e aparelho para armazenamento e/ou introdução de implante para estrutura anatômica oca. |
| US8388644B2 (en) | 2008-12-29 | 2013-03-05 | Cook Medical Technologies Llc | Embolic protection device and method of use |
| US20170202657A1 (en) | 2009-01-16 | 2017-07-20 | Claret Medical, Inc. | Intravascular blood filters and methods of use |
| WO2011034718A2 (en) | 2009-09-21 | 2011-03-24 | Claret Medical, Inc. | Intravascular blood filters and methods of use |
| US9326843B2 (en) | 2009-01-16 | 2016-05-03 | Claret Medical, Inc. | Intravascular blood filters and methods of use |
| ES2516066T3 (es) * | 2009-01-16 | 2014-10-30 | Claret Medical, Inc. | Filtro sanguíneo intravascular |
| US8057507B2 (en) | 2009-01-16 | 2011-11-15 | Novate Medical Limited | Vascular filter |
| EP2381893B1 (en) | 2009-01-16 | 2018-03-07 | Novate Medical Ltd. | A vascular filter device |
| US20100228281A1 (en) * | 2009-01-16 | 2010-09-09 | Paul Gilson | Vascular filter system |
| EP2381891B1 (en) * | 2009-01-16 | 2018-04-11 | Novate Medical Limited | A vascular filter device |
| WO2010088520A2 (en) * | 2009-01-29 | 2010-08-05 | Claret Medical, Inc. | Illuminated intravascular blood filter |
| US20100228280A1 (en) * | 2009-03-09 | 2010-09-09 | Adam Groothuis | Methods and devices for treatment of lumenal systems |
| US20100274277A1 (en) * | 2009-04-27 | 2010-10-28 | Cook Incorporated | Embolic protection device with maximized flow-through |
| WO2010129510A2 (en) | 2009-05-04 | 2010-11-11 | Incept. Llc | Biomaterials for track and puncture closure |
| US8828053B2 (en) | 2009-07-24 | 2014-09-09 | Depuy Mitek, Llc | Methods and devices for repairing and anchoring damaged tissue |
| US8814903B2 (en) | 2009-07-24 | 2014-08-26 | Depuy Mitek, Llc | Methods and devices for repairing meniscal tissue |
| EP2459120A4 (en) | 2009-07-27 | 2017-11-01 | Claret Medical, Inc. | Dual endovascular filter and methods of use |
| EP3505136B1 (en) | 2009-07-29 | 2025-06-25 | C. R. Bard, Inc. | Tubular filter |
| BR112012003804B1 (pt) * | 2009-08-21 | 2019-02-19 | Novan, Inc. | Curativo para ferimentos, método para formar um curativo para ferimentos, e, kit de curativo para ferimento |
| JP2013509963A (ja) | 2009-11-09 | 2013-03-21 | スポットライト テクノロジー パートナーズ エルエルシー | 断片化ヒドロゲル |
| WO2011057131A1 (en) | 2009-11-09 | 2011-05-12 | Spotlight Technology Partners Llc | Polysaccharide based hydrogels |
| JP5808792B2 (ja) | 2010-04-13 | 2015-11-10 | ミビ・ニューロサイエンス・リミテッド・ライアビリティ・カンパニーMivi Neuroscience LLC | 急性脳卒中治療装置およびシステム |
| WO2011137159A1 (en) | 2010-04-27 | 2011-11-03 | Synthes Usa, Llc | Anchor assembly including expandable anchor |
| US9743919B2 (en) | 2010-04-27 | 2017-08-29 | DePuy Synthes Products, Inc. | Stitch lock for attaching two or more structures |
| US9451938B2 (en) | 2010-04-27 | 2016-09-27 | DePuy Synthes Products, Inc. | Insertion instrument for anchor assembly |
| US9597064B2 (en) | 2010-04-27 | 2017-03-21 | DePuy Synthes Products, Inc. | Methods for approximating a tissue defect using an anchor assembly |
| US9307978B2 (en) | 2010-11-04 | 2016-04-12 | Linvatec Corporation | Method and apparatus for securing an object to bone, including the provision and use of a novel suture assembly for securing an object to bone |
| US9307977B2 (en) | 2010-11-04 | 2016-04-12 | Conmed Corporation | Method and apparatus for securing an object to bone, including the provision and use of a novel suture assembly for securing suture to bone |
| US9259306B2 (en) | 2010-12-30 | 2016-02-16 | Claret Medical, Inc. | Aortic embolic protection device |
| US12150851B2 (en) | 2010-12-30 | 2024-11-26 | Claret Medical, Inc. | Method of isolating the cerebral circulation during a cardiac procedure |
| WO2012100091A2 (en) | 2011-01-19 | 2012-07-26 | Accessclosure, Inc. | Apparatus and methods for sealing a vascular puncture |
| US9820728B2 (en) | 2011-01-19 | 2017-11-21 | Access Closure, Inc. | Apparatus and methods for sealing a vascular puncture |
| AU2012217577B2 (en) | 2011-02-16 | 2016-06-09 | Linvatec Corporation | Method and apparatus for securing an object to bone |
| US10327790B2 (en) | 2011-08-05 | 2019-06-25 | Route 92 Medical, Inc. | Methods and systems for treatment of acute ischemic stroke |
| US10779855B2 (en) | 2011-08-05 | 2020-09-22 | Route 92 Medical, Inc. | Methods and systems for treatment of acute ischemic stroke |
| WO2013109623A1 (en) | 2012-01-17 | 2013-07-25 | Lumen Biomedical, Inc. | Aortic arch filtration system for carotid artery protection |
| US9888994B2 (en) | 2012-05-15 | 2018-02-13 | Transverse Medical, Inc. | Catheter-based apparatuses and methods |
| GB2507053A (en) * | 2012-10-16 | 2014-04-23 | Jonathan Featherstone | Nephroureterectomy apparatus |
| US10285797B2 (en) | 2013-01-22 | 2019-05-14 | St. Jude Medical, Llc | Protecting against cerebral embolism |
| GB2510151B (en) * | 2013-01-25 | 2015-07-22 | Cook Medical Technologies Llc | Vascular plug |
| WO2014140088A2 (en) | 2013-03-15 | 2014-09-18 | Novate Medical Limited | A vascular filter device |
| US8679150B1 (en) | 2013-03-15 | 2014-03-25 | Insera Therapeutics, Inc. | Shape-set textile structure based mechanical thrombectomy methods |
| US8715314B1 (en) | 2013-03-15 | 2014-05-06 | Insera Therapeutics, Inc. | Vascular treatment measurement methods |
| CN105228688B (zh) | 2013-03-15 | 2019-02-19 | 伊瑟拉医疗公司 | 脉管治疗装置和方法 |
| US8715315B1 (en) | 2013-03-15 | 2014-05-06 | Insera Therapeutics, Inc. | Vascular treatment systems |
| EP2996630B1 (en) | 2013-05-14 | 2022-04-06 | Transverse Medical, Inc. | Catheter-based apparatuses |
| WO2017142874A2 (en) | 2016-02-16 | 2017-08-24 | Insera Therapeutics, Inc. | Aspiration devices and anchored flow diverting devices |
| US9265512B2 (en) | 2013-12-23 | 2016-02-23 | Silk Road Medical, Inc. | Transcarotid neurovascular catheter |
| CN103751856B (zh) * | 2014-01-22 | 2015-07-29 | 同济大学 | 一种具有良好分散性的聚乳酸类栓塞微球 |
| US9782247B2 (en) | 2014-02-18 | 2017-10-10 | Cook Medical Technologies, LLC | Flexible embolic double filter |
| US9968758B2 (en) * | 2014-03-21 | 2018-05-15 | Boston Scientific Scimed, Inc. | Devices and methods for treating a lung |
| US9820761B2 (en) | 2014-03-21 | 2017-11-21 | Route 92 Medical, Inc. | Rapid aspiration thrombectomy system and method |
| DE202016009165U1 (de) | 2015-02-04 | 2023-04-26 | Route 92 Medical, Inc. | System für schnelle Aspirationsthrombektomie |
| US11065019B1 (en) | 2015-02-04 | 2021-07-20 | Route 92 Medical, Inc. | Aspiration catheter systems and methods of use |
| US9566144B2 (en) | 2015-04-22 | 2017-02-14 | Claret Medical, Inc. | Vascular filters, deflectors, and methods |
| WO2017019563A1 (en) | 2015-07-24 | 2017-02-02 | Route 92 Medical, Inc. | Anchoring delivery system and methods |
| WO2017040681A1 (en) | 2015-09-01 | 2017-03-09 | Mivi Neuroscience, Inc. | Thrombectomy devices and treatment of acute ischemic stroke with thrombus engagement |
| US10716915B2 (en) | 2015-11-23 | 2020-07-21 | Mivi Neuroscience, Inc. | Catheter systems for applying effective suction in remote vessels and thrombectomy procedures facilitated by catheter systems |
| WO2018067844A1 (en) | 2016-10-06 | 2018-04-12 | Mivi Neuroscience, Inc. | Hydraulic displacement and removal of thrombus clots, and catheters for performing hydraulic displacement |
| WO2018132387A1 (en) | 2017-01-10 | 2018-07-19 | Route 92 Medical, Inc. | Aspiration catheter systems and methods of use |
| CN110461401B (zh) | 2017-01-20 | 2022-06-07 | 92号医疗公司 | 单操作者颅内医疗装置输送系统和使用方法 |
| EP4052679B1 (en) | 2017-02-22 | 2025-04-23 | Boston Scientific Scimed, Inc. | Systems for protecting the cerebral vasculature |
| WO2018183624A1 (en) | 2017-03-29 | 2018-10-04 | The Regents Of The University Of Colorado, A Body Corporate | Reverse thermal gels and their use as vascular embolic repair agents |
| US11234723B2 (en) | 2017-12-20 | 2022-02-01 | Mivi Neuroscience, Inc. | Suction catheter systems for applying effective aspiration in remote vessels, especially cerebral arteries |
| US10478535B2 (en) | 2017-05-24 | 2019-11-19 | Mivi Neuroscience, Inc. | Suction catheter systems for applying effective aspiration in remote vessels, especially cerebral arteries |
| EP4378421A3 (en) | 2017-10-27 | 2024-08-07 | Boston Scientific Scimed, Inc. | Systems and methods for protecting the cerebral vasculature |
| US11033411B2 (en) * | 2017-12-14 | 2021-06-15 | Boston Scientific Scimed, Inc. | Stent including an expandable member |
| US11154390B2 (en) | 2017-12-19 | 2021-10-26 | Claret Medical, Inc. | Systems for protection of the cerebral vasculature during a cardiac procedure |
| EP3784168B1 (en) | 2018-04-26 | 2024-03-20 | Boston Scientific Scimed, Inc. | Systems for protecting the cerebral vasculature |
| CN115920204B (zh) | 2018-05-17 | 2025-06-20 | 92号医疗公司 | 抽吸导管系统和使用方法 |
| CN108742927B (zh) * | 2018-06-27 | 2020-02-04 | 李林 | 一种血栓诱发实验装置 |
| EP3840691B1 (en) | 2018-08-21 | 2025-01-29 | Boston Scientific Scimed, Inc. | Systems for protecting the cerebral vasculature |
| US11617865B2 (en) | 2020-01-24 | 2023-04-04 | Mivi Neuroscience, Inc. | Suction catheter systems with designs allowing rapid clearing of clots |
| WO2022076893A1 (en) | 2020-10-09 | 2022-04-14 | Route 92 Medical, Inc. | Aspiration catheter systems and methods of use |
Family Cites Families (80)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4422939A (en) | 1979-11-07 | 1983-12-27 | Texas Medical Products, Inc. | Blood and perfusate filter |
| US4429939A (en) * | 1981-09-09 | 1984-02-07 | Thomas & Betts Corporation | Electrical cable assembly |
| SE434715B (sv) * | 1982-01-20 | 1984-08-13 | Tetra Pak Dev | Sett vid sammanfogning av laminatmaterial innefattande lager av orienteringsstreckt polyester |
| US4494531A (en) * | 1982-12-06 | 1985-01-22 | Cook, Incorporated | Expandable blood clot filter |
| US4512338A (en) * | 1983-01-25 | 1985-04-23 | Balko Alexander B | Process for restoring patency to body vessels |
| US4550126A (en) | 1985-01-25 | 1985-10-29 | Hydromer, Inc. | Hydrophilic, flexible, open cell polyurethane-poly(N-vinyl lactam) interpolymer foam and dental and biomedical products fabricated therefrom |
| US4793348A (en) | 1986-11-15 | 1988-12-27 | Palmaz Julio C | Balloon expandable vena cava filter to prevent migration of lower extremity venous clots into the pulmonary circulation |
| US5061276A (en) * | 1987-04-28 | 1991-10-29 | Baxter International Inc. | Multi-layered poly(tetrafluoroethylene)/elastomer materials useful for in vivo implantation |
| US4817600A (en) | 1987-05-22 | 1989-04-04 | Medi-Tech, Inc. | Implantable filter |
| US4832055A (en) | 1988-07-08 | 1989-05-23 | Palestrant Aubrey M | Mechanically locking blood clot filter |
| US5972505A (en) * | 1989-04-04 | 1999-10-26 | Eastman Chemical Company | Fibers capable of spontaneously transporting fluids |
| US5059205A (en) | 1989-09-07 | 1991-10-22 | Boston Scientific Corporation | Percutaneous anti-migration vena cava filter |
| DE69113450T3 (de) | 1990-02-20 | 2001-06-13 | The Procter & Gamble Company, Cincinnati | Struktur mit offenen kapillarkanälen, verfahren zu deren herstellung und extrusionsdüse zum gebrauch darin. |
| WO1991017724A1 (en) | 1990-05-17 | 1991-11-28 | Harbor Medical Devices, Inc. | Medical device polymer |
| GB9102660D0 (en) | 1991-02-07 | 1991-03-27 | Ultra Lab Ltd | Wound dressing materials |
| US5350398A (en) | 1991-05-13 | 1994-09-27 | Dusan Pavcnik | Self-expanding filter for percutaneous insertion |
| US5288489A (en) | 1991-08-28 | 1994-02-22 | Orion Therapeutic Systems, Inc. | Fibrinolysis and fibrinogenolysis treatment |
| US5277976A (en) * | 1991-10-07 | 1994-01-11 | Minnesota Mining And Manufacturing Company | Oriented profile fibers |
| US5626605A (en) | 1991-12-30 | 1997-05-06 | Scimed Life Systems, Inc. | Thrombosis filter |
| US5527338A (en) * | 1992-09-02 | 1996-06-18 | Board Of Regents, The University Of Texas System | Intravascular device |
| US5807306A (en) * | 1992-11-09 | 1998-09-15 | Cortrak Medical, Inc. | Polymer matrix drug delivery apparatus |
| US5836868A (en) | 1992-11-13 | 1998-11-17 | Scimed Life Systems, Inc. | Expandable intravascular occlusion material removal devices and methods of use |
| US5540707A (en) * | 1992-11-13 | 1996-07-30 | Scimed Life Systems, Inc. | Expandable intravascular occlusion material removal devices and methods of use |
| DK0659389T3 (da) * | 1993-10-20 | 1999-02-15 | Schneider Europ Ag | Endoprotese |
| US5599298A (en) * | 1993-12-30 | 1997-02-04 | Boston Scientific Corporation | Bodily sample collection balloon catheter method |
| DE9409484U1 (de) | 1994-06-11 | 1994-08-04 | Naderlinger, Eduard, 50127 Bergheim | Vena-cava Thromben-Filter |
| US6099864A (en) | 1994-12-02 | 2000-08-08 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | In situ activation of microcapsules |
| US5651765A (en) | 1995-04-27 | 1997-07-29 | Avecor Cardiovascular Inc. | Blood filter with concentric pleats and method of use |
| US5938645A (en) | 1995-05-24 | 1999-08-17 | Boston Scientific Corporation Northwest Technology Center Inc. | Percutaneous aspiration catheter system |
| US5704910A (en) | 1995-06-05 | 1998-01-06 | Nephros Therapeutics, Inc. | Implantable device and use therefor |
| US6491965B1 (en) | 1995-11-30 | 2002-12-10 | Hamilton Civic Hospitals Research Development, Inc. | Medical device comprising glycosaminoglycan-antithrombin III/heparin cofactor II conjugates |
| NL1002423C2 (nl) * | 1996-02-22 | 1997-08-25 | Cordis Europ | Tijdelijk-filtercatheter. |
| US5935139A (en) | 1996-05-03 | 1999-08-10 | Boston Scientific Corporation | System for immobilizing or manipulating an object in a tract |
| US5839443A (en) | 1996-05-16 | 1998-11-24 | The Trustees Of Columbia University In The City Of New York | Method for inhibiting thrombosis in a patient whose blood is subjected to extracorporeal circulation |
| US6544276B1 (en) | 1996-05-20 | 2003-04-08 | Medtronic Ave. Inc. | Exchange method for emboli containment |
| US5972019A (en) * | 1996-07-25 | 1999-10-26 | Target Therapeutics, Inc. | Mechanical clot treatment device |
| US5977429A (en) | 1996-08-22 | 1999-11-02 | Eastman Chemical Company | Synthetic polyester absorbent materials |
| US6103376A (en) | 1996-08-22 | 2000-08-15 | Eastman Chemical Company | Bundles of fibers useful for moving liquids at high fluxes and acquisition/distribution structures that use the bundles |
| US6391044B1 (en) * | 1997-02-03 | 2002-05-21 | Angioguard, Inc. | Vascular filter system |
| US5882329A (en) * | 1997-02-12 | 1999-03-16 | Prolifix Medical, Inc. | Apparatus and method for removing stenotic material from stents |
| US5814064A (en) * | 1997-03-06 | 1998-09-29 | Scimed Life Systems, Inc. | Distal protection device |
| US5911734A (en) * | 1997-05-08 | 1999-06-15 | Embol-X, Inc. | Percutaneous catheter and guidewire having filter and medical device deployment capabilities |
| US6245088B1 (en) | 1997-07-07 | 2001-06-12 | Samuel R. Lowery | Retrievable umbrella sieve and method of use |
| US5928260A (en) * | 1997-07-10 | 1999-07-27 | Scimed Life Systems, Inc. | Removable occlusion system for aneurysm neck |
| US6361545B1 (en) * | 1997-09-26 | 2002-03-26 | Cardeon Corporation | Perfusion filter catheter |
| US6395014B1 (en) * | 1997-09-26 | 2002-05-28 | John A. Macoviak | Cerebral embolic protection assembly and associated methods |
| US6066149A (en) | 1997-09-30 | 2000-05-23 | Target Therapeutics, Inc. | Mechanical clot treatment device with distal filter |
| ATE404123T1 (de) | 1997-11-12 | 2008-08-15 | Genesis Technologies Llc | Vorrichtung zum entfernen von okklusionen in biologischen durchgängen |
| US6254563B1 (en) * | 1997-12-15 | 2001-07-03 | Cardeon Corporation | Perfusion shunt apparatus and method |
| ATE266434T1 (de) | 1998-02-23 | 2004-05-15 | Massachusetts Inst Technology | Bioabbaubare polymere mit formgedächtnis |
| US6123681A (en) | 1998-03-31 | 2000-09-26 | Global Vascular Concepts, Inc. | Anti-embolism stocking device |
| US6450989B2 (en) * | 1998-04-27 | 2002-09-17 | Artemis Medical, Inc. | Dilating and support apparatus with disease inhibitors and methods for use |
| EP1082072B8 (en) * | 1998-06-04 | 2014-03-05 | New York University | Endovascular thin film devices for treating and preventing stroke |
| US6306163B1 (en) | 1998-08-04 | 2001-10-23 | Advanced Cardiovascular Systems, Inc. | Assembly for collecting emboli and method of use |
| US6083239A (en) | 1998-11-24 | 2000-07-04 | Embol-X, Inc. | Compliant framework and methods of use |
| US6373970B1 (en) | 1998-12-29 | 2002-04-16 | General Electric Company | Image registration using fourier phase matching |
| US6368338B1 (en) * | 1999-03-05 | 2002-04-09 | Board Of Regents, The University Of Texas | Occlusion method and apparatus |
| US6231589B1 (en) | 1999-03-22 | 2001-05-15 | Microvena Corporation | Body vessel filter |
| US6267776B1 (en) | 1999-05-03 | 2001-07-31 | O'connell Paul T. | Vena cava filter and method for treating pulmonary embolism |
| EP1176995A1 (en) | 1999-05-07 | 2002-02-06 | Salviac Limited | A tissue engineering scaffold |
| WO2000067670A1 (en) * | 1999-05-07 | 2000-11-16 | Salviac Limited | An embolic protection device |
| US6371970B1 (en) | 1999-07-30 | 2002-04-16 | Incept Llc | Vascular filter having articulation region and methods of use in the ascending aorta |
| US6346116B1 (en) | 1999-08-03 | 2002-02-12 | Medtronic Ave, Inc. | Distal protection device |
| US6273901B1 (en) | 1999-08-10 | 2001-08-14 | Scimed Life Systems, Inc. | Thrombosis filter having a surface treatment |
| US6364895B1 (en) | 1999-10-07 | 2002-04-02 | Prodesco, Inc. | Intraluminal filter |
| US6368344B1 (en) * | 1999-12-16 | 2002-04-09 | Advanced Cardiovascular Systems, Inc. | Stent deployment system with reinforced inner member |
| ES2282246T3 (es) * | 2000-03-10 | 2007-10-16 | Anthony T. Don Michael | Dispositivo de prevencion de la embolia vascular que utiliza filtros. |
| US6514273B1 (en) | 2000-03-22 | 2003-02-04 | Endovascular Technologies, Inc. | Device for removal of thrombus through physiological adhesion |
| US6602271B2 (en) * | 2000-05-24 | 2003-08-05 | Medtronic Ave, Inc. | Collapsible blood filter with optimal braid geometry |
| US6527746B1 (en) * | 2000-08-03 | 2003-03-04 | Ev3, Inc. | Back-loading catheter |
| US6485501B1 (en) * | 2000-08-11 | 2002-11-26 | Cordis Corporation | Vascular filter system with guidewire and capture mechanism |
| EP1355692B1 (en) | 2001-01-09 | 2011-08-10 | Microvention, Inc. | Embolectomy catheters |
| US6610077B1 (en) | 2001-01-23 | 2003-08-26 | Endovascular Technologies, Inc. | Expandable emboli filter and thrombectomy device |
| US20020128680A1 (en) * | 2001-01-25 | 2002-09-12 | Pavlovic Jennifer L. | Distal protection device with electrospun polymer fiber matrix |
| US6878153B2 (en) * | 2001-07-02 | 2005-04-12 | Rubicon Medical, Inc. | Methods, systems, and devices for providing embolic protection and removing embolic material |
| US6616682B2 (en) | 2001-09-19 | 2003-09-09 | Jomed Gmbh | Methods and apparatus for distal protection during a medical procedure |
| US7052500B2 (en) | 2001-10-19 | 2006-05-30 | Scimed Life Systems, Inc. | Embolus extractor |
| US20050021075A1 (en) | 2002-12-30 | 2005-01-27 | Bonnette Michael J. | Guidewire having deployable sheathless protective filter |
| US7303575B2 (en) * | 2002-08-01 | 2007-12-04 | Lumen Biomedical, Inc. | Embolism protection devices |
| US7220271B2 (en) * | 2003-01-30 | 2007-05-22 | Ev3 Inc. | Embolic filters having multiple layers and controlled pore size |
-
2003
- 2003-04-16 US US10/414,909 patent/US7303575B2/en not_active Expired - Lifetime
- 2003-08-01 EP EP03767200.3A patent/EP1545322B1/en not_active Expired - Lifetime
- 2003-08-01 JP JP2004526448A patent/JP2005534429A/ja active Pending
- 2003-08-01 AU AU2003257182A patent/AU2003257182A1/en not_active Abandoned
- 2003-08-01 WO PCT/US2003/024459 patent/WO2004012587A2/en not_active Ceased
-
2004
- 2004-03-22 US US10/806,311 patent/US8123775B2/en active Active
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010505526A (ja) * | 2006-10-04 | 2010-02-25 | ボストン サイエンティフィック リミテッド | アテローム硬化性プラークを治療するための装置及び方法 |
| JP2012521258A (ja) * | 2009-03-25 | 2012-09-13 | リシブロック リミテッド | フィルター装置及びそれを使用する方法 |
| US9427299B2 (en) | 2009-03-25 | 2016-08-30 | Lithiblock Ltd. | Filter apparatuses and methods of using same |
| US10368974B2 (en) | 2014-10-27 | 2019-08-06 | Lithiblock Ltd. | Gallbladder implants and systems and methods for the delivery thereof |
| US10702368B2 (en) | 2014-10-27 | 2020-07-07 | Lithiblock Ltd. | Gallbladder implant and systems and methods for the delivery thereof |
| JP2021013745A (ja) * | 2019-07-11 | 2021-02-12 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 再挿入のための血塊回収装置の洗浄 |
| JP7532121B2 (ja) | 2019-07-11 | 2024-08-13 | デピュイ・シンセス・プロダクツ・インコーポレイテッド | 再挿入のための血塊回収装置の洗浄 |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2003257182A1 (en) | 2004-02-23 |
| WO2004012587A2 (en) | 2004-02-12 |
| EP1545322A2 (en) | 2005-06-29 |
| WO2004012587A3 (en) | 2004-06-10 |
| US8123775B2 (en) | 2012-02-28 |
| EP1545322B1 (en) | 2019-10-02 |
| AU2003257182A8 (en) | 2004-02-23 |
| EP1545322A4 (en) | 2008-12-03 |
| US20040220611A1 (en) | 2004-11-04 |
| US7303575B2 (en) | 2007-12-04 |
| US20040093015A1 (en) | 2004-05-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2005534429A (ja) | 塞栓症予防デバイス | |
| US7879067B2 (en) | Fiber based embolism protection device | |
| JP4916854B2 (ja) | ぜい弱性プラークを治療するための装置 | |
| US5823198A (en) | Method and apparatus for intravasculer embolization | |
| US5891192A (en) | Ion-implanted protein-coated intralumenal implants | |
| US8048042B2 (en) | Medical articles incorporating surface capillary fiber | |
| JP4856339B2 (ja) | ヒドロゲルからなる血管塞栓材及びそれを使用した治療方法 | |
| JP4416151B2 (ja) | 環境の変化に応答して体積が膨潤するヒドロゲルならびにそれらの製造法および利用法 | |
| US5456693A (en) | Embolization plugs for blood vessels | |
| US20030014075A1 (en) | Methods, materials and apparatus for deterring or preventing endoleaks following endovascular graft implanation | |
| JP2007521041A (ja) | 重合性の再拘束可能で再配置可能で取外し可能な経皮的血管内ステントグラフト | |
| JP2004531331A (ja) | 薬剤投与器具 | |
| WO1997009006A1 (en) | Endovascular support device and method of use | |
| JPWO2001030411A1 (ja) | ヒドロゲルからなる血管塞栓材及びそれを使用した治療方法 | |
| Cohn et al. | In situ generated medical devices | |
| JP4687174B2 (ja) | ステント | |
| JPH0341963A (ja) | 人工血管およびその製造方法 | |
| JP2024075523A (ja) | 塞栓物 | |
| JP2004527268A (ja) | 罹患した動脈の血管再構築のための方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20060721 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20060721 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090602 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090827 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20100224 |