EP4393395A2 - Dermale schichtanalytmessvorrichtungen und verfahren - Google Patents
Dermale schichtanalytmessvorrichtungen und verfahren Download PDFInfo
- Publication number
- EP4393395A2 EP4393395A2 EP24152037.8A EP24152037A EP4393395A2 EP 4393395 A2 EP4393395 A2 EP 4393395A2 EP 24152037 A EP24152037 A EP 24152037A EP 4393395 A2 EP4393395 A2 EP 4393395A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- sensor
- insertion needle
- dermal
- layer
- analyte
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/145—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue
- A61B5/1468—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue using chemical or electrochemical methods, e.g. by polarographic means
- A61B5/1473—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue using chemical or electrochemical methods, e.g. by polarographic means invasive, e.g. introduced into the body by a catheter
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/145—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue
- A61B5/14507—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue specially adapted for measuring characteristics of body fluids other than blood
- A61B5/1451—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue specially adapted for measuring characteristics of body fluids other than blood for interstitial fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/145—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue
- A61B5/14532—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue for measuring glucose, e.g. by tissue impedance measurement
Definitions
- the detection of the level of glucose or other analytes, such as lactate, oxygen or the like, in certain individuals is vitally important to their health.
- the monitoring of glucose is particularly important to individuals with diabetes. Diabetics may need to monitor glucose levels to determine when insulin is needed to reduce glucose levels in their bodies or when additional glucose is needed to raise the level of glucose in their bodies.
- Blood vessel sensors are more invasive than subcutaneous sensors, but have the advantage of providing analyte concentrations directly from the blood.
- Subcutaneous analyte sensors are therefore used, but they too have certain limitations.
- the insertion of the analyte sensor in the subcutaneous tissue results in skin/tissue trauma that causes immunological response that can cause inaccurate sensor readings, at least for a period of time.
- the trauma may cause an over-consumption of glucose in the positioned sensor vicinity by erythrocytes released by localized bleeding.
- the glucose response from a subcutaneously positioned sensor lags the response of a venous-positioned sensor, primarily due to a physiological lag between subcutaneous and venous glucose.
- Analyte sensors, applicators for inserting them, and methods of making and using are described that provide benefits of blood vessel and subcutaneous analyte sensors without their key limitations.
- in vivo analyte sensors and in vivo sensor applicator sets that insert at least a sensing portion of an in vivo analyte sensor into a subject
- embodiments of methods of using the sensor applicator sets to insert a sensing portion of an in vivo analyte sensor into a subject and methods of determining in vivo analyte presence and concentration using the in vivo analyte sensors.
- analyte concentration data obtained using a dermal analyte sensor correlates more closely to the analyte concentration in blood as compared to in vivo ISF sensors.
- the dermal insertion needle is configured to create an insertion path at a skin site of a subject to place a sensor in the dermal layer and no further.
- the dermal sensor is inserted into the insertion path and at the dermal layer, but not through a dermal layer.
- the insertion needle creates an insertion path for the sensor before the insertion of the sensor into, but not through, a dermal layer is initiated.
- the reference point may be the skin surface to be breached for dermal insertion, or may be a reference or component of the sensor applicator set.
- the needle may be disposed at an angle relative to the sensor. For example, when designed so that that the needle is angled relative to the sensor, the needle creates a leading edge for the sensor during operation of the applicator set.
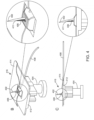
- FIG. 1 panel B.
- An applicator set according to embodiments of the present disclosure is schematically illustrated in FIG. 1 , Panel B.
- the sensor and/or needle are angled relative to a reference point.
- one of the sensor or insertion needle may be angled relative to the other, or one or both may be angled relative to a skin surface or another element of the applicator set.
- the angles of each may differ and the needle may be angled a first angle and the sensor may be angled a second angle. As shown in Fig.
- the forces applied by the tissue surface or underlying tissue during insertion are primarily applied to and absorbed by the insertion needle - indicated by line 115 at the tip of insertion needle 114.
- the insertion needle can also assist in stabilizing the sensor by counteracting forces applied by the tissue of the subject to the sensor in the direction of the insertion needle.
- embodiments include longitudinal axis Ln 1 of the insertion needle disposed at an acute angle 122 relative to the base and axis Ln 1 and the longest axis of the sensor Ls 1 converge towards each other at area C. While angle 123 is shown in Panel B as a 90° angle, it can be anywhere from 80°-100°.
- a given needle may have a constant, i.e., uniform, width along its entire length, or may have a varying, i.e., changing, width along at least a portion of its length, such as the tip portion used to pierce the surface of the skin.
- An insertion needle has a length to insert a dermal sensor just into the dermal layer, and no more. Insertion depth may be controlled by the length of the needle and/or configuration of the base other applicator components that limit insertion depth.
- the insertion needle can be configured to extend beyond the dermal space, such as into (or even fully through) subcutaneous tissue (e.g., 3 mm to 10 mm beneath the surface of the skin depending on the location of the skin on the body).
- subcutaneous tissue e.g., 3 mm to 10 mm beneath the surface of the skin depending on the location of the skin on the body.
- Any of the insertion needles described herein may be solid insertion needles, where by “solid” is meant that the needles do not have an internal space and/or lumen (e.g., are not hollow), or they may include an internal space or lumen.
- An insertion needle of the subject applicator sets may be bladed or non-bladed.
- the dimensions (e.g., the length) of the sensor may be selected according to the body site of the subject in which the sensor is to be inserted, as the depth and thickness of the epidermis and dermis exhibit a degree of variability depending on skin location.
- the epidermis is only about 0.05 mm thick on the eyelids, but about 1.5 mm thick on the palms and the soles of the feet.
- the dermis is the thickest of the three layers of skin and ranges from about 1.5 mm to 4 mm thick, depending on the skin location.
- the length of the inserted portion of the sensor should be greater than the thickness of the epidermis and dermis, but should not exceed the combined thickness of the epidermis, dermis, and subcutaneous tissue (when inserted at a normal angle to the skin surface - a non-normal angle allows an insertion portion of the sensor that can exceed these combined thicknesses since the absolute depth of penetration is less than the length of the inserted portion of the sensor).
- a sensor of the subject applicator sets may be made of a conductive material which may exhibit sufficient conductivity to obviate any need for an additional conductive coating to provide a working electrode capable of electrochemical determination of the concentration of the analyte.
- the conductive material is a conductive plastic.
- the conductive plastic may include a polymer matrix (e.g., a non-conductive or substantially non-conductive polymer matrix) that includes a conductive material interspersed therein.
- the polymer matrix may include any polymer suitable for forming a conductive plastic, including but not limited to, a polyether block amide polymer.
- the conductive material may be a conductive material that confers sufficient conductivity to the polymer matrix.
- Such materials may include conductive particulate matter, such as conductive metal particles, microspheres coated with a conductive material, carbon, and/or the like.
- the conductive material interspersed in the polymer matrix is carbon.
- the carbon may be present in the polymer matrix in an amount ranging from 5% to 30% w/w.
- the carbon may be present in the polymer matrix in an amount ranging from 7% to 20% w/w, such as from 8% to 12% w/w, e.g., 10% w/w.
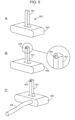
- FIG. 3 A sensor that can be used in an applicator set according to one embodiment of the present disclosure is schematically illustrated in FIG. 3 .
- the sensor includes substrate 302 having groove 304, with the sensor positioned on base 306.
- the substrate and base are made of plastic.
- a conductive layer e.g., Au layer 308
- sputter coating or any other suitable approach for providing a conductive layer on a plastic substrate (Panel B).
- Additional layers may be applied to the substrate and/or base, such as carbon layer 310 disposed on the Au-coated substrate (e.g., by dip coating, spraying or submersion), as shown in Panel C.
- a sensing layer, a protective and/or mass transport limiting layer, and/or the like may further be provided on carbon layer 310 to enhance the performance of and/or provide additional functionalities to the sensor.
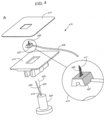
- FIG 4 Various components of a disassembled sensor applicator set 416 according to one embodiment of the present disclosure are shown in FIG 4 , Panel A.
- the proximal end of insertion needle 402 is disposed within or on molded hub 404.
- Insertion needle 402 is disposed at an angle 403 (such as between 9°and 13°, e.g., 11°) relative to vertical axis 405 extending from the top surface of molded hub 404, and/or angle 415 may also be formed similar to angle 122 of Panel B of Fig. 1 .
- the applicator set includes dermal sensor 406 having an appropriate size, shape, and dimension for insertion into a dermal layer of a subject and for generating analyte-responsive signals in response to analyte present in the dermal fluid.
- Sensor 406 may be the same as, similar to, or different from the sensor shown in FIG. 3 , Panel C.
- Wire 408 is connected to a conductive coating on the base 407 of sensor 406 and facilitates electrical communication between sensor 406 and components of an analyte sensing system, e.g., a voltage source, control electronics, and/or the like.
- View 410 is a magnified view of sensor 406 and wire 408 connected thereto.
- dermal sensor 406 includes groove 411 having a shape and angle complementary to a portion of insertion needle 402.
- the applicator set includes assembly mount 412 for mounting/fixing the sensor and insertion needle in the desired position relative to each other, as well as adhesive 414 for securing the dermal sensor in its final position upon insertion into the subject.
- Assembled sensor applicator set 416 which includes dermal sensor 406 and insertion needle 402 disposed at an angle relative to sensor 406, is shown in FIG. 4 , Panel B (left).
- Adhesive 414 is cut-away to show the other elements of applicator set 416, and a magnified view of the sensor and insertion needle associated with each other via their complementary shapes is also shown in FIG. 4 , Panel B.
- a side view of assembled applicator set 416 is provided in FIG. 4 , Panel C, including a magnified side view of dermal sensor 406 and insertion needle 402 on the right.
- applicator sets of the present disclosure may include a dermal sensor molded (e.g., by injection molding) from a nonconductive or conductive plastic.
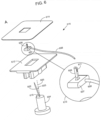
- An exemplary applicator set 502 according to this embodiment is shown in FIG. 5 .
- Dermal sensor 501 and base 503, e.g., molded from conductive plastic, is shown in FIG. 5 , Panel A.
- the conductive plastic may include a polymer matrix (e.g., a polyether block amide polymer) having a conductive material (e.g., carbon particles) disposed therein, as described hereinabove.
- a distal portion of the sensor 501 includes a divot.
- An exemplary divot 504 is shown in FIG.
- a dermal sensor includes divot or through hole, sensing reagents or the like may be deposited therein. If the divot is to be used for deposition of sensing reagents therein, the area and/or volume of the divot may be selected during the manufacturing design process to provide one or more desired sensor characteristics (e.g., working electrode characteristics), such as a particular sensitivity in response to the analyte, e.g., where the sensitivity (e.g., analyte- responsive signal) may be increased or decreased by increasing or decreasing the area and/or volume of the recess, respectively.
- desired sensor characteristics e.g., working electrode characteristics
- a dermal sensor may therefore include one or more structural features (e.g., a molded divot for deposition of sensor reagents, a molded groove having a shape complementary to the insertion needle, and/or the like) at one or more regions of the sensor.
- Electrical connecting wire 506 connected to the conductive plastic of the base is shown in FIG. 5 , Panel C.
- the wire may be employed to facilitate electrical communication between sensor and base 502 and components of an analyte sensing system, e.g., a voltage source, control electronics, and/or the like.
- FIG 6 Components of a disassembled sensor applicator set 616 according to one embodiment of the present disclosure are shown in FIG 6 , Panel A.
- the proximal end of insertion needle 602 is disposed within or on hub 604.
- Insertion needle 602 is disposed at an angle 603 (such as between 9°and 13°, e.g., 11°) relative to vertical axis 605 extending from the top surface of molded hub 604, which may be its angle relative to the sensor upon assembly of the applicator set, and/or angle 615 may also be formed similar to angle 122 of Panel B of Fig. 1 .
- the applicator set includes molded dermal sensor 606 and base 607 configured (e.g., sized, etc.) for insertion into a dermal layer of a subject and for generating analyte-responsive signals in response to analyte present in the dermal fluid.
- Molded dermal sensor 606 may be the same as, similar to, or different from the molded sensor shown in FIG. 5 .
- Electrical connecting wire 608 is connected to the base 607 of molded sensor 606 and facilitates electrical communication between molded sensor 606 and components of an analyte sensing system, e.g., a voltage source, control electronics, and/or the like.
- View 610 is a magnified view of molded dermal sensor 606 and wire 608 connected thereto.
- molded dermal sensor 606 includes a groove 611 having a shape and angle complementary to a body portion of insertion needle 602.
- the applicator set includes assembly mount 612 for mounting/fixing the sensor and insertion needle in the desired position relative to each other, as well as adhesive 614 for securing the sensor in its final position upon insertion into the subject.
- Assembled sensor applicator set 616 which includes dermal sensor 606 and insertion needle 602 disposed at an angle relative to sensor 606, is shown in FIG. 6 , Panel B (left).
- Adhesive 614 is cut-away in FIG. 6 , Panel B to show the other elements of applicator set 616.
- a magnified view of the dermal sensor and the insertion needle associated with each other via their complementary shapes is shown in FIG. 6 , Panel B (right).
- a side view of assembled applicator set 616 is provided in FIG. 6 , Panel C (left), including a magnified view of dermal sensor 606 and insertion needle 602 on the right.
- a dermal sensor employed by a subject applicator set is a dermal sensor that includes one or more conductive materials, e.g., in the form of conductive layers, integrated particles, and the like.
- the one or more conductive materials confer conductivity to the sensor, e.g., when the sensor includes a non-conductive or substantially non-conductive material (e.g., a non-conductive plastic material).
- a conductive layer disposed on a non-conductive material may constitute the working electrode of the sensor.
- a second conductive material e.g., a conductive layer, electrically isolated from the first conductive material or layer, e.g., by an electrically insulating layer, may be added to provide a second electrode in addition to the working electrode (e.g., a counter or counter/reference electrode, a reference electrode, or the like).
- the working electrode e.g., a counter or counter/reference electrode, a reference electrode, or the like.
- a dermal sensor described herein may include one or more conductive layers made of a material independently selected from gold, carbon, platinum, ruthenium, palladium, silver, silver chloride, silver bromide, and combinations thereof.
- a conductive layer may be a layer of gold, tin oxide, platinum, ruthenium dioxide or palladium, indium tin oxide, zinc oxide, fluorine doped tin oxide, as well as other non-corroding materials known to those skilled in the art.
- the conductive layer can be a combination of two or more conductive materials.
- the conductive layer may be constructed from a layer of gold on a first region of the sensor and a layer of carbon on a second region of the sensor.
- the conductive layer may be applied to the sensor or a layer thereof by being deposited, such as by vapor deposition or vacuum deposition or otherwise sputtered, printed on a flat surface or in an embossed or otherwise recessed surface, transferred from a separate carrier or liner, etched, or molded.
- Suitable methods of printing include screen-printing, piezoelectric printing, ink jet printing, laser printing, photolithography, painting, gravure roll printing, transfer printing, and other known printing methods.
- At least one or more working electrodes of a dermal sensor may include a sensing layer that includes sensing reagents to facilitate determination of a concentration of an analyte of interest.
- the sensing layer includes one or more components designed to facilitate the electrolysis of the analyte.
- the sensing layer may include, for example, an analyte responsive enzyme to catalyze a reaction of the analyte and produce a response at the working electrode, an electron transfer agent to indirectly or directly transfer electrons between the analyte and the working electrode, or both.
- the sensing layer is deposited on the conductive material of a working electrode.
- the sensing layer may extend beyond the conductive material of the working electrode.
- the sensing layer may also extend over other electrodes, e.g., over a counter electrode and/or reference electrode (or counter/reference electrode).
- the sensing layer may be integral with the material of an electrode.
- a sensor for sensing glucose may include a first sensing layer which is spaced apart from the working electrode and contains a glucose-responsive enzyme, for example, glucose oxidase or glucose dehydrogenase.
- a glucose-responsive enzyme for example, glucose oxidase or glucose dehydrogenase.
- the reaction of glucose the presence of the appropriate enzyme forms hydrogen peroxide.
- a second sensing layer may be provided directly on the working electrode and contains a peroxidase enzyme and an electron transfer agent to generate a signal at the electrode in response to the hydrogen peroxide.
- the level of hydrogen peroxide indicated by the sensor then correlates to the level of glucose.
- Another sensor which operates similarly can be made using a single sensing layer with both the glucose and the peroxidase being deposited in the single sensing layer.
- a sensing layer that is in direct contact with the working electrode may contain an electron transfer agent to transfer electrons directly or indirectly between the analyte and the working electrode, and/or an enzyme to facilitate a reaction of the analyte.
- a glucose, or oxygen electrode may be formed having a sensing layer which contains an enzyme, such as glucose oxidase, and an electron transfer agent that facilitates the electro-oxidation of the glucose.
- the sensing layer is not deposited directly on the working electrode. Instead, the sensing layer may be spaced apart from the working electrode, and separated from the working electrode, e.g., by a separation layer.
- a separation layer may include one or more membranes or films or a physical distance.
- the separation layer may also act as a mass transport limiting layer and/or an interferent eliminating layer and/or a biocompatible layer.
- one or more of the working electrodes do not have a corresponding sensing layer, or have a sensing layer which does not contain one or more components (e.g., an electron transfer agent and/or enzyme) needed to electrolyze the analyte.
- the signal at this working electrode corresponds to background signal which may be removed from the analyte signal obtained from one or more other working electrodes that are associated with fully-functional sensing layers by, for example, subtracting the signal.
- a type of polymeric electron transfer agent that may be used with the dermal sensors contains a redox species covalently bound in a polymeric composition, as described, e.g., in U.S. Patent Nos. 6,605,200 and 6,605,201 , the disclosures of which are incorporated herein by reference in their entireties for all purposes.
- An example of this type of mediator is poly(vinylferrocene).
- Another type of electron transfer agent contains an ionically-bound redox species.
- This type of mediator may include a charged polymer coupled to an oppositely charged redox species. Examples of this type of mediator include a negatively charged polymer coupled to a positively charged redox species such as an osmium or ruthenium polypyridyl cation.
- an ionically-bound mediator is a positively charged polymer such as quaternized poly(4- vinyl pyridine) or poly(1-vinyl imidazole) coupled to a negatively charged redox species such as ferricyanide or ferrocyanide.
- electron transfer agents include a redox species coordinatively bound to a polymer.
- the mediator may be formed by coordination of an osmium or cobalt 2,2'-bipyridyl complex to poly(1-vinyl imidazole) or poly(4-vinyl pyridine).
- Suitable electron transfer agents are osmium transition metal complexes with one or more ligands, each ligand having a nitrogen-containing heterocycle such as 2,2'- bipyridine, 1.10-phenanthroline, 1-methyl, 2-pyridyl biimidazole, or derivatives thereof.
- the electron transfer agents may also have one or more ligands covalently bound in a polymer, each ligand having at least one nitrogen-containing heterocycle, such as pyridine, imidazole, or derivatives thereof.
- An example of an electron transfer agent includes (a) a polymer or copolymer having pyridine or imidazole functional groups and (b) osmium cations complexed with two ligands, each ligand containing 2,2'-bipyridine, 1,10-phenanthroline, or derivatives thereof, the two ligands not necessarily being the same.
- Some derivatives of 2,2'-bipyridine for complexation with the osmium cation include but are not limited to 4,4'-dimethy)-2,2'-bipyridine and mono-, di-, and polyalkoxy-2,2'-bipyridines, such as 4,4'-dimethoxy-2,2'-bipyridine.
- Derivatives of 1,10- phenanthroline for complexation with the osmium cation include but are not limited to 4,7-dimethyl-1,10-ptienanthrolitic and mono, di-, and polyalkoxy-1,10-phenanthrolines, such as 4,7-dimethoxy-1,10-plienanthroline.
- Polymers for complexation with the osmium cation include but are not limited to polymers and copolymers of poly(1-vinylimidazole) (referred to as "PVI”) and poly(4-vinyl pyridine) (referred to as "PVP").
- Suitable copolymer substituents of poly(1-vinyl imidazole) include acrylonitrile, acrylamide, and substituted or quaternized N-vinyl imidazole, e.g., electron transfer agents with osmium complexed to a polymer or copolymer of poly(1-vinyl imidazole).
- Embodiments may employ electron transfer agents having a redox potential ranging from about -200 mV to about +200 mV versus the standard calomel electrode (SCE).
- the sensing layer may also include an enzyme which is capable of catalyzing a reaction of the analyte.
- the enzyme may also, in some embodiments, act as an electron transfer agent.
- One example of a suitable enzyme is an enzyme which catalyzes a reaction of the analyte.
- an enzyme such as a glucose oxidase, glucose dehydrogenase (e.g., pyrroloquinoline quinone (PQQ) dependent glucose dehydrogenase, flavine adenine dinucleotide (FAD) dependent glucose dehydrogenase, or nicotinamide adenine dinucleotide (NAD) dependent glucose dehydrogenase), may be used when the analyte of interest is glucose.
- PQQ pyrroloquinoline quinone
- FAD flavine adenine dinucleotide
- NAD nicotinamide adenine dinucleotide dependent glucose dehydrogenase
- a lactate oxidase or lactate dehydrogenase may be used when the analyte of interest is lactate.
- Laccase may be used when the analyte of interest is oxygen or when oxygen is generated or consumed in response to a reaction of the analyte.
- an enzyme may be attached to a polymer, cross linking the enzyme with another electron transfer agent (which, as described above, may be polymeric).
- a second enzyme may also be used in certain embodiments. This second enzyme may be used to catalyze a reaction of a product compound resulting from the catalyzed reaction of the analyte. The second enzyme may operate with an electron transfer agent to electrolyze the product compound to generate a signal at the working electrode.
- a second enzyme may be provided in an interferent-eliminating layer to catalyze reactions that remove interferents.
- Certain embodiments include a sensing layer that works at a gentle oxidizing potential, e.g., a potential of about +40 mV.
- This sensing layer uses an osmium (Os)- based mediator designed for low potential operation and is stably anchored in a polymeric layer.
- the sensing element is redox active component that includes (1) Osmium-based mediator molecules attached by stable (bidente) ligands anchored to a polymeric backbone, and (2) glucose oxidase enzyme molecules. These two constituents are crosslinked together.
- Such sensing layers are described in, for example, U.S. Patent No. 5,262,035 , which is incorporated herein by reference in its entirety for all purposes.
- Optional hydrophilic modifiers such as poly(ethylene glycol), hydroxyl or polyhydroxyl modifiers, may be used to enhance the biocompatibility of the polymer or the resulting membrane.
- Mass transport limiting layers that may be adapted for use wityh present disclosure are described, e.g., in U.S. Patent No. 6,932,894 , the disclosure of which is incorporated herein by reference in its entirety for all purposes.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Physics & Mathematics (AREA)
- Biomedical Technology (AREA)
- Medical Informatics (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Biophysics (AREA)
- Pathology (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Optics & Photonics (AREA)
- Molecular Biology (AREA)
- Surgery (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Emergency Medicine (AREA)
- Measurement Of The Respiration, Hearing Ability, Form, And Blood Characteristics Of Living Organisms (AREA)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201261738776P | 2012-12-18 | 2012-12-18 | |
| PCT/US2013/075658 WO2014099907A1 (en) | 2012-12-18 | 2013-12-17 | Dermal layer analyte sensing devices and methods |
| EP20181886.1A EP3735896B1 (de) | 2012-12-18 | 2013-12-17 | Erfassungsvorrichtungen und -verfahren für hautschichtanalyt |
| EP13865733.3A EP2934315B1 (de) | 2012-12-18 | 2013-12-17 | Erfassungsvorrichtungen für hautschichtanalyte |
Related Parent Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20181886.1A Division EP3735896B1 (de) | 2012-12-18 | 2013-12-17 | Erfassungsvorrichtungen und -verfahren für hautschichtanalyt |
| EP13865733.3A Division EP2934315B1 (de) | 2012-12-18 | 2013-12-17 | Erfassungsvorrichtungen für hautschichtanalyte |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP4393395A2 true EP4393395A2 (de) | 2024-07-03 |
| EP4393395A3 EP4393395A3 (de) | 2024-10-30 |
Family
ID=50931699
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20181886.1A Active EP3735896B1 (de) | 2012-12-18 | 2013-12-17 | Erfassungsvorrichtungen und -verfahren für hautschichtanalyt |
| EP24152037.8A Pending EP4393395A3 (de) | 2012-12-18 | 2013-12-17 | Dermale schichtanalytmessvorrichtungen und verfahren |
| EP13865733.3A Active EP2934315B1 (de) | 2012-12-18 | 2013-12-17 | Erfassungsvorrichtungen für hautschichtanalyte |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20181886.1A Active EP3735896B1 (de) | 2012-12-18 | 2013-12-17 | Erfassungsvorrichtungen und -verfahren für hautschichtanalyt |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP13865733.3A Active EP2934315B1 (de) | 2012-12-18 | 2013-12-17 | Erfassungsvorrichtungen für hautschichtanalyte |
Country Status (4)
| Country | Link |
|---|---|
| US (3) | US9636060B2 (de) |
| EP (3) | EP3735896B1 (de) |
| ES (1) | ES2978657T3 (de) |
| WO (1) | WO2014099907A1 (de) |
Families Citing this family (71)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9788771B2 (en) | 2006-10-23 | 2017-10-17 | Abbott Diabetes Care Inc. | Variable speed sensor insertion devices and methods of use |
| US9402544B2 (en) | 2009-02-03 | 2016-08-02 | Abbott Diabetes Care Inc. | Analyte sensor and apparatus for insertion of the sensor |
| DK3622883T3 (da) | 2010-03-24 | 2021-07-19 | Abbott Diabetes Care Inc | Indførerer til medicinsk indretning og fremgangsmåder til at indføre og anvende medicinske indretninger |
| AU2012352560B2 (en) | 2011-12-11 | 2017-01-19 | Abbott Diabetes Care Inc. | Analyte sensor devices, connections, and methods |
| US9668686B2 (en) | 2013-03-15 | 2017-06-06 | Abbott Diabetes Care Inc. | In vivo glucose sensing in an increased perfusion dermal layer |
| WO2014197995A1 (en) * | 2013-06-13 | 2014-12-18 | Microdermics Inc. | Metallic microneedles |
| CN115444410B (zh) | 2017-01-23 | 2025-08-15 | 雅培糖尿病护理公司 | 用于插入体内分析物传感器的施加器及组件 |
| US12109032B1 (en) | 2017-03-11 | 2024-10-08 | Biolinq Incorporated | Methods for achieving an isolated electrical interface between an anterior surface of a microneedle structure and a posterior surface of a support structure |
| US11045142B1 (en) | 2017-04-29 | 2021-06-29 | Biolinq, Inc. | Heterogeneous integration of silicon-fabricated solid microneedle sensors and CMOS circuitry |
| CN110769872B (zh) * | 2017-05-01 | 2022-11-11 | 目标点技术有限公司 | 注射设备和使用方法 |
| EP3928688B1 (de) | 2017-06-23 | 2022-06-01 | Dexcom, Inc. | Transkutane analytsensoren, applikatoren dafür und nadelnabe mit verdrehsicherung |
| US11199556B2 (en) * | 2017-07-04 | 2021-12-14 | Arkray, Inc. | Measuring apparatus, computer readable medium storing measuring program and measuring method |
| US11246512B2 (en) * | 2017-07-04 | 2022-02-15 | Arkray, Inc. | Measuring apparatus, computer readable medium storing measuring program and measuring method |
| US11918348B2 (en) | 2017-12-05 | 2024-03-05 | Abbott Diabetes Care Inc. | Medical devices having a dynamic surface profile and methods for production and use thereof |
| US11311212B2 (en) | 2017-12-05 | 2022-04-26 | Abbott Diabetes Care Inc. | Guard rings for on-body analyte sensors |
| US12433510B2 (en) | 2017-12-21 | 2025-10-07 | Georgia Tech Research Corporation | Methods and systems for improved collection of interstitial fluid |
| US20210236028A1 (en) | 2018-05-17 | 2021-08-05 | Abbott Diabetes Care Inc. | Analyte sensor antimicrobial configurations and adhesives |
| USD926325S1 (en) | 2018-06-22 | 2021-07-27 | Dexcom, Inc. | Wearable medical monitoring device |
| US12016821B2 (en) | 2018-08-23 | 2024-06-25 | Abbott Diabetes Care Inc. | Needle assemblies containing oriented acupuncture needles and methods for production thereof |
| CN113195022A (zh) | 2018-12-21 | 2021-07-30 | 雅培糖尿病护理公司 | 用于分析物传感器插入的系统、装置和方法 |
| CN109589121A (zh) * | 2019-02-01 | 2019-04-09 | 浙江澍源智能技术有限公司 | 一种探测体内组织液葡萄糖浓度信息的电化学方法及其装置 |
| CN109758162A (zh) * | 2019-02-01 | 2019-05-17 | 浙江澍源智能技术有限公司 | 一种探测体内组织液葡萄糖信号的光学装置及方法 |
| JP7319742B2 (ja) | 2020-07-29 | 2023-08-02 | バイオリンク インコーポレイテッド | 微小針アレイを伴う持続的分析物監視システム |
| US12239463B2 (en) | 2020-08-31 | 2025-03-04 | Abbott Diabetes Care Inc. | Systems, devices, and methods for analyte sensor insertion |
| JP2023540086A (ja) | 2020-08-31 | 2023-09-21 | アボット ダイアベティス ケア インコーポレイテッド | 検体センサー取付器のためのシステム、装置、及び方法 |
| CN116033869A (zh) | 2020-09-15 | 2023-04-28 | 雅培糖尿病护理公司 | 用于分析物监测的系统、装置和方法 |
| MY199727A (en) | 2020-09-15 | 2023-11-21 | Abbott Diabetes Care Inc | Systems, devices, and methods for an analyte sensor |
| CA3197412A1 (en) * | 2020-12-10 | 2022-06-16 | Yi Wang | Systems, devices, and methods for analyte sensor insertion |
| MX2023006880A (es) | 2020-12-10 | 2023-06-23 | Abbott Diabetes Care Inc | Sensores de analitos para detectar cetonas y metodos para usar los mismos. |
| EP4262557B1 (de) | 2020-12-15 | 2026-01-21 | Abbott Diabetes Care Inc. | Nad(p)-depot für nad(p)-abhängige enzymbasierte sensoren |
| CN116801802A (zh) | 2020-12-18 | 2023-09-22 | 美国雅培糖尿病护理公司 | 用于分析物检测的系统和方法 |
| AU2021401430B2 (en) | 2020-12-18 | 2025-04-17 | Abbott Diabetes Care Inc. | Systems and methods for analyte detection |
| JP7612871B2 (ja) | 2020-12-23 | 2025-01-14 | アボット ダイアベティス ケア インコーポレイテッド | 連続カリウムセンサおよびその使用方法 |
| EP4267752A1 (de) | 2020-12-23 | 2023-11-01 | Abbott Diabetes Care, Inc. | Analytsensoren zur erfassung von glutamat und verfahren zur verwendung davon |
| WO2022146888A1 (en) | 2020-12-30 | 2022-07-07 | Abbott Diabetes Care Inc. | Analyte sensors for detecting asparagine and aspartate and methods of use thereof |
| JP2024501962A (ja) | 2020-12-30 | 2024-01-17 | アボット ダイアベティス ケア インコーポレイテッド | 検体モニタのためのデバイス |
| ES3009941T3 (en) | 2020-12-31 | 2025-03-31 | Abbott Diabetes Care Inc | Systems, devices, and methods for analyte monitoring |
| US20220202326A1 (en) | 2020-12-31 | 2022-06-30 | Abbott Diabetes Care Inc. | Analyte sensors with metal-containing redox mediators and methods of using the same |
| EP4271273A1 (de) | 2020-12-31 | 2023-11-08 | Abbott Diabetes Care, Inc. | Analytsensor und scharfes produkt zur abgabe eines therapeutischen mittels in unmittelbarer nähe zu einem analytsensor und verfahren dafür |
| CA3202248A1 (en) | 2020-12-31 | 2022-07-07 | Zenghe Liu | Analyte sensors and methods of use thereof |
| JP7341583B6 (ja) | 2021-05-08 | 2023-09-29 | バイオリンク インコーポレイテッド | 微小針アレイベースの持続的分析物監視デバイスのための障害検出 |
| CN117396973A (zh) | 2021-06-03 | 2024-01-12 | 雅培糖尿病护理公司 | 生理参数分析和相关图形用户界面的系统、设备和方法 |
| CA3222449A1 (en) | 2021-07-16 | 2023-01-19 | Abbott Diabetes Care Inc. | Systems, devices, and methods for analyte monitoring |
| JP2024535833A (ja) | 2021-09-15 | 2024-10-02 | アボット ダイアベティス ケア インコーポレイテッド | 異なるタイプの生理学的センサとの拡張可能通信のためのモジュール式検体接続性システム |
| JP2024538580A (ja) | 2021-09-27 | 2024-10-23 | アボット ダイアベティス ケア インコーポレイテッド | 検体モニタシステムのための適応センサ感度 |
| EP4681635A3 (de) | 2021-09-28 | 2026-03-25 | Biolinq Incorporated | Mikronadelgehäuse und applikatorvorrichtung für eine vorrichtung zur kontinuierlichen analytüberwachung auf mikronadelarraybasis |
| EP4409598A1 (de) | 2021-09-29 | 2024-08-07 | Abbott Diabetes Care, Inc. | Mobile anwendungsaktualisierungen für analytdatenempfangsvorrichtungen |
| AU2022363525A1 (en) | 2021-10-11 | 2024-03-14 | Abbott Diabetes Care Inc. | Systems, devices, and methods for communication between an analyte sensor and external devices |
| US20250221636A1 (en) | 2021-10-29 | 2025-07-10 | Abbott Diabetes Care Inc. | Systems, devices, and methods for analyte monitoring |
| USD1033641S1 (en) | 2021-12-17 | 2024-07-02 | Biolinq Incorporated | Microneedle array sensor applicator device |
| AU2022425086A1 (en) | 2021-12-30 | 2024-05-30 | Abbott Diabetes Care Inc. | Methods and systems for sensing a plurality of analytes |
| JP2025503520A (ja) | 2021-12-31 | 2025-02-04 | アボット ダイアベティス ケア インコーポレイテッド | 検体モニタのためのシステム及び方法 |
| CN118414119A (zh) | 2022-01-04 | 2024-07-30 | 雅培糖尿病护理公司 | 用于分析物监测的系统、装置和方法 |
| WO2023150363A1 (en) | 2022-02-04 | 2023-08-10 | Abbott Diabetes Care Inc. | Systems, devices, and methods for an analyte sensor |
| US20250152052A1 (en) | 2022-02-22 | 2025-05-15 | Abbott Diabetes Care Inc. | Systems, devices, and methods for an analyte sensor |
| JP2025509070A (ja) | 2022-03-16 | 2025-04-11 | アボット ダイアベティス ケア インコーポレイテッド | 検体モニタのためのシステム、デバイス、及び方法 |
| USD1057153S1 (en) | 2022-04-29 | 2025-01-07 | Biolinq Incorporated | Microneedle array sensor applicator device |
| CA3261124A1 (en) | 2022-07-11 | 2024-01-18 | Abbott Diabetes Care Inc. | ANALYTICAL MONITORING SYSTEMS, DEVICES AND METHODS |
| US20240065586A1 (en) | 2022-08-31 | 2024-02-29 | Abbott Diabetes Care Inc. | System, apparatus, and devices for analyte monitoring |
| JP2025536929A (ja) | 2022-10-24 | 2025-11-12 | アボット ダイアベティス ケア インコーポレイテッド | リモート検体モニタシステムへの持続接続の促進 |
| WO2024145693A1 (en) | 2022-12-30 | 2024-07-04 | Abbott Diabetes Care Inc. | Drug delivery compositions and methods of controlling drug delivery rates of subcutaneous sensors |
| CN120548138A (zh) | 2023-01-18 | 2025-08-26 | 雅培糖尿病护理公司 | 用于分析物监测的系统、装置和方法 |
| AU2024212125A1 (en) | 2023-01-25 | 2025-08-14 | Abbott Diabetes Care Inc. | Systems, devices, and methods for analyte monitoring |
| WO2024163950A2 (en) | 2023-02-02 | 2024-08-08 | Biolinq Incorporated | Method for improved sensor sensitivity of a microneedle-based continuous analyte monitoring system |
| CN120882369A (zh) | 2023-02-17 | 2025-10-31 | 雅培糖尿病护理公司 | 用于分析物监测的系统、装置和方法 |
| CN121194737A (zh) | 2023-05-12 | 2025-12-23 | 雅培糖尿病护理公司 | 多天线无线通信系统 |
| AU2024276818A1 (en) | 2023-05-24 | 2025-12-04 | Abbott Diabetes Care Inc. | Methods for incorporating therapeutic agents into analyte sensors |
| WO2024254033A1 (en) | 2023-06-09 | 2024-12-12 | Abbott Diabetes Care Inc. | Systems, devices, and methods for analyte monitoring |
| WO2025106332A1 (en) | 2023-11-13 | 2025-05-22 | Abbott Diabetes Care Inc. | Improved sharp for insertion of analyte sensor |
| WO2025165935A1 (en) | 2024-02-02 | 2025-08-07 | Abbott Diabetes Care Inc. | Recyclable analyte monitoring devices |
| WO2025231220A1 (en) | 2024-05-03 | 2025-11-06 | Abbott Diabetes Care Inc. | Devices for skin adhesive liner removal |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5262035A (en) | 1989-08-02 | 1993-11-16 | E. Heller And Company | Enzyme electrodes |
| US6605201B1 (en) | 1999-11-15 | 2003-08-12 | Therasense, Inc. | Transition metal complexes with bidentate ligand having an imidazole ring and sensor constructed therewith |
| US6932894B2 (en) | 2001-05-15 | 2005-08-23 | Therasense, Inc. | Biosensor membranes composed of polymers containing heterocyclic nitrogens |
| US20100326842A1 (en) | 2009-06-30 | 2010-12-30 | Abbott Diabetes Care Inc. | Extruded Electrode Structures and Methods of Using Same |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4627445A (en) * | 1985-04-08 | 1986-12-09 | Garid, Inc. | Glucose medical monitoring system |
| GB8813571D0 (en) | 1988-06-08 | 1988-07-13 | Showell A W Sugicraft Ltd | Biosensor introducers &c |
| IE72524B1 (en) * | 1994-11-04 | 1997-04-23 | Elan Med Tech | Analyte-controlled liquid delivery device and analyte monitor |
| US5951521A (en) * | 1998-09-25 | 1999-09-14 | Minimed Inc. | Subcutaneous implantable sensor set having the capability to remove deliver fluids to an insertion site |
| US7509153B2 (en) | 2000-09-26 | 2009-03-24 | Sensys Medical, Inc. | Method and apparatus for control of skin perfusion for indirect glucose measurement |
| CA2412401C (en) | 2000-06-09 | 2011-05-10 | Piet Moerman | Cap for a lancing device |
| GB0030929D0 (en) | 2000-12-19 | 2001-01-31 | Inverness Medical Ltd | Analyte measurement |
| US20050054908A1 (en) | 2003-03-07 | 2005-03-10 | Blank Thomas B. | Photostimulation method and apparatus in combination with glucose determination |
| US6936006B2 (en) * | 2002-03-22 | 2005-08-30 | Novo Nordisk, A/S | Atraumatic insertion of a subcutaneous device |
| WO2004002312A1 (ja) * | 2002-07-01 | 2004-01-08 | Terumo Kabushiki Kaisha | 体液採取具 |
| US20040149578A1 (en) * | 2003-01-30 | 2004-08-05 | Chun-Mu Huang | Method for manufacturing electrochemical sensor and structure thereof |
| US20060253085A1 (en) | 2005-05-06 | 2006-11-09 | Medtronic Minimed, Inc. | Dual insertion set |
| US8088150B2 (en) * | 2008-02-04 | 2012-01-03 | Aleeva Medical Inc. | Device for disc shunt implantation and peri-shunt injection |
| EP2437697A1 (de) | 2009-06-04 | 2012-04-11 | C8 Medisensors Inc. | Vorrichtung zur erhöhung der blutperfusion und zur verbesserten kühlung der haut |
| US10376213B2 (en) * | 2009-06-30 | 2019-08-13 | Waveform Technologies, Inc. | System, method and apparatus for sensor insertion |
| US20110319738A1 (en) * | 2010-06-29 | 2011-12-29 | Abbott Diabetes Care Inc. | Medical Devices and Insertion Systems and Methods |
| EP2621339B1 (de) | 2010-09-29 | 2020-01-15 | Dexcom, Inc. | Erweitertes system für kontinuierliche analytüberwachung |
-
2013
- 2013-12-17 WO PCT/US2013/075658 patent/WO2014099907A1/en not_active Ceased
- 2013-12-17 EP EP20181886.1A patent/EP3735896B1/de active Active
- 2013-12-17 EP EP24152037.8A patent/EP4393395A3/de active Pending
- 2013-12-17 ES ES20181886T patent/ES2978657T3/es active Active
- 2013-12-17 US US14/108,964 patent/US9636060B2/en active Active
- 2013-12-17 EP EP13865733.3A patent/EP2934315B1/de active Active
-
2017
- 2017-03-28 US US15/471,867 patent/US10624567B2/en active Active
-
2020
- 2020-03-11 US US16/815,438 patent/US12396666B2/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5262035A (en) | 1989-08-02 | 1993-11-16 | E. Heller And Company | Enzyme electrodes |
| US6605201B1 (en) | 1999-11-15 | 2003-08-12 | Therasense, Inc. | Transition metal complexes with bidentate ligand having an imidazole ring and sensor constructed therewith |
| US6605200B1 (en) | 1999-11-15 | 2003-08-12 | Therasense, Inc. | Polymeric transition metal complexes and uses thereof |
| US6932894B2 (en) | 2001-05-15 | 2005-08-23 | Therasense, Inc. | Biosensor membranes composed of polymers containing heterocyclic nitrogens |
| US20100326842A1 (en) | 2009-06-30 | 2010-12-30 | Abbott Diabetes Care Inc. | Extruded Electrode Structures and Methods of Using Same |
Also Published As
| Publication number | Publication date |
|---|---|
| US12396666B2 (en) | 2025-08-26 |
| EP4393395A3 (de) | 2024-10-30 |
| US9636060B2 (en) | 2017-05-02 |
| US20200305771A1 (en) | 2020-10-01 |
| US20170196487A1 (en) | 2017-07-13 |
| ES2978657T3 (es) | 2024-09-17 |
| WO2014099907A1 (en) | 2014-06-26 |
| EP2934315A4 (de) | 2016-08-17 |
| EP3735896B1 (de) | 2024-01-17 |
| EP2934315B1 (de) | 2020-07-22 |
| EP3735896A1 (de) | 2020-11-11 |
| US10624567B2 (en) | 2020-04-21 |
| US20140171771A1 (en) | 2014-06-19 |
| EP2934315A1 (de) | 2015-10-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12396666B2 (en) | Analyte sensing devices and methods | |
| EP2720610B1 (de) | Gestapelter analytsensor mit einer ersten elektrode, die schmaler als einer zweiten elektrode des sensors ist | |
| US8986208B2 (en) | Analyte sensor sensitivity attenuation mitigation | |
| US20170127989A1 (en) | Shallow implantable analyte sensor with rapid physiological response | |
| US20210361204A1 (en) | Electrochemical sensors and methods for making electrochemical sensors using advanced printing technology | |
| EP2935615B1 (de) | Verfahren zur verbesserung der messgenauigkeit sowie dazugehörige vorrichtungen und systeme | |
| WO2009148849A1 (en) | Extended lifetime reference electrodes for amperometric sensors | |
| WO2009148845A1 (en) | Extended lifetime reference electrodes for long term amperometric sensors | |
| WO2009148846A1 (en) | Reference electrodes having an extended lifetime for use in long term amperometric sensors | |
| WO2012149410A2 (en) | Methods of collecting and analyzing samples | |
| EP2925229A1 (de) | Sensormodul und verfahren zur verwendung eines sensormoduls | |
| WO2012158202A2 (en) | Analyte sensors and methods of fabricating them | |
| WO2010014391A1 (en) | Analyte sensors comprising plasticizers | |
| US20250064355A1 (en) | Analyte monitoring systems and methods for monitoring multiple analytes | |
| US20250213152A1 (en) | Background interference mitigation for high sensitivity ketone sensing | |
| WO2013059624A1 (en) | Analyte sensor membranes and methods for making same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN PUBLISHED |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 2934315 Country of ref document: EP Kind code of ref document: P Ref document number: 3735896 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R079 Free format text: PREVIOUS MAIN CLASS: A61B0005150000 Ipc: A61B0005050000 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: A61B 5/15 20060101ALI20240924BHEP Ipc: A61B 5/05 20210101AFI20240924BHEP |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20250408 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: A61B 5/05 20210101AFI20260217BHEP Ipc: A61B 5/15 20060101ALI20260217BHEP Ipc: A61B 5/145 20060101ALI20260217BHEP |