EP3936136A1 - Spezifische sirna-moleküle, zusammensetzung und verwendung davon zur behandlung von dreifach negativem brustkrebs - Google Patents
Spezifische sirna-moleküle, zusammensetzung und verwendung davon zur behandlung von dreifach negativem brustkrebs Download PDFInfo
- Publication number
- EP3936136A1 EP3936136A1 EP21181521.2A EP21181521A EP3936136A1 EP 3936136 A1 EP3936136 A1 EP 3936136A1 EP 21181521 A EP21181521 A EP 21181521A EP 3936136 A1 EP3936136 A1 EP 3936136A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- sirna
- erbb
- seq
- spt3
- cells
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 208000003721 Triple Negative Breast Neoplasms Diseases 0.000 title claims abstract description 127
- 208000022679 triple-negative breast carcinoma Diseases 0.000 title claims abstract description 127
- 239000000203 mixture Substances 0.000 title claims abstract description 62
- 238000011282 treatment Methods 0.000 title claims abstract description 49
- 108020004459 Small interfering RNA Proteins 0.000 title claims abstract description 37
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 120
- 238000000034 method Methods 0.000 claims abstract description 83
- 108090000623 proteins and genes Proteins 0.000 claims abstract description 82
- 208000026310 Breast neoplasm Diseases 0.000 claims abstract description 50
- 206010006187 Breast cancer Diseases 0.000 claims abstract description 49
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims abstract description 44
- 230000002401 inhibitory effect Effects 0.000 claims abstract description 10
- 101150029707 ERBB2 gene Proteins 0.000 claims description 256
- 108010029485 Protein Isoforms Proteins 0.000 claims description 121
- 102000001708 Protein Isoforms Human genes 0.000 claims description 121
- 108020004999 messenger RNA Proteins 0.000 claims description 69
- 125000003729 nucleotide group Chemical group 0.000 claims description 60
- 239000002773 nucleotide Substances 0.000 claims description 41
- 239000000523 sample Substances 0.000 claims description 41
- 230000004048 modification Effects 0.000 claims description 28
- 238000012986 modification Methods 0.000 claims description 28
- 230000008685 targeting Effects 0.000 claims description 25
- 239000003795 chemical substances by application Substances 0.000 claims description 22
- 239000003814 drug Substances 0.000 claims description 22
- 230000000692 anti-sense effect Effects 0.000 claims description 20
- 238000003556 assay Methods 0.000 claims description 19
- 230000002209 hydrophobic effect Effects 0.000 claims description 19
- 230000035755 proliferation Effects 0.000 claims description 18
- 239000003550 marker Substances 0.000 claims description 17
- 238000002560 therapeutic procedure Methods 0.000 claims description 16
- 238000000338 in vitro Methods 0.000 claims description 15
- 108091081021 Sense strand Proteins 0.000 claims description 14
- 239000008194 pharmaceutical composition Substances 0.000 claims description 14
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 claims description 14
- 108091032973 (ribonucleotides)n+m Proteins 0.000 claims description 13
- 239000003937 drug carrier Substances 0.000 claims description 12
- 230000000295 complement effect Effects 0.000 claims description 10
- 238000007901 in situ hybridization Methods 0.000 claims description 9
- 238000012163 sequencing technique Methods 0.000 claims description 9
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 claims description 8
- 239000002246 antineoplastic agent Substances 0.000 claims description 8
- 239000003112 inhibitor Substances 0.000 claims description 7
- 238000002512 chemotherapy Methods 0.000 claims description 6
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 6
- 108091007960 PI3Ks Proteins 0.000 claims description 5
- 102000003993 Phosphatidylinositol 3-kinases Human genes 0.000 claims description 5
- 108090000430 Phosphatidylinositol 3-kinases Proteins 0.000 claims description 5
- 230000002601 intratumoral effect Effects 0.000 claims description 5
- 238000001990 intravenous administration Methods 0.000 claims description 5
- RYYWUUFWQRZTIU-UHFFFAOYSA-K thiophosphate Chemical compound [O-]P([O-])([O-])=S RYYWUUFWQRZTIU-UHFFFAOYSA-K 0.000 claims description 5
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 claims description 4
- 102100033810 RAC-alpha serine/threonine-protein kinase Human genes 0.000 claims description 4
- 108010065917 TOR Serine-Threonine Kinases Proteins 0.000 claims description 4
- 102000013530 TOR Serine-Threonine Kinases Human genes 0.000 claims description 4
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 claims description 4
- 229960003852 atezolizumab Drugs 0.000 claims description 4
- 229960004397 cyclophosphamide Drugs 0.000 claims description 4
- 229940127089 cytotoxic agent Drugs 0.000 claims description 4
- 238000009261 endocrine therapy Methods 0.000 claims description 4
- 102000005962 receptors Human genes 0.000 claims description 4
- 108020003175 receptors Proteins 0.000 claims description 4
- 238000007920 subcutaneous administration Methods 0.000 claims description 4
- 241000196324 Embryophyta Species 0.000 claims description 3
- 108060008682 Tumor Necrosis Factor Proteins 0.000 claims description 3
- 239000003242 anti bacterial agent Substances 0.000 claims description 3
- 230000002280 anti-androgenic effect Effects 0.000 claims description 3
- 229940034984 endocrine therapy antineoplastic and immunomodulating agent Drugs 0.000 claims description 3
- 229960001592 paclitaxel Drugs 0.000 claims description 3
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 claims description 3
- GRZXWCHAXNAUHY-NSISKUIASA-N (2S)-2-(4-chlorophenyl)-1-[4-[(5R,7R)-7-hydroxy-5-methyl-6,7-dihydro-5H-cyclopenta[d]pyrimidin-4-yl]-1-piperazinyl]-3-(propan-2-ylamino)-1-propanone Chemical compound C1([C@H](C(=O)N2CCN(CC2)C=2C=3[C@H](C)C[C@@H](O)C=3N=CN=2)CNC(C)C)=CC=C(Cl)C=C1 GRZXWCHAXNAUHY-NSISKUIASA-N 0.000 claims description 2
- LKJPYSCBVHEWIU-KRWDZBQOSA-N (R)-bicalutamide Chemical compound C([C@@](O)(C)C(=O)NC=1C=C(C(C#N)=CC=1)C(F)(F)F)S(=O)(=O)C1=CC=C(F)C=C1 LKJPYSCBVHEWIU-KRWDZBQOSA-N 0.000 claims description 2
- SGTNSNPWRIOYBX-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(propan-2-yl)pentanenitrile Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCCC(C#N)(C(C)C)C1=CC=C(OC)C(OC)=C1 SGTNSNPWRIOYBX-UHFFFAOYSA-N 0.000 claims description 2
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 claims description 2
- CWHUFRVAEUJCEF-UHFFFAOYSA-N BKM120 Chemical compound C1=NC(N)=CC(C(F)(F)F)=C1C1=CC(N2CCOCC2)=NC(N2CCOCC2)=N1 CWHUFRVAEUJCEF-UHFFFAOYSA-N 0.000 claims description 2
- 108010006654 Bleomycin Proteins 0.000 claims description 2
- COVZYZSDYWQREU-UHFFFAOYSA-N Busulfan Chemical compound CS(=O)(=O)OCCCCOS(C)(=O)=O COVZYZSDYWQREU-UHFFFAOYSA-N 0.000 claims description 2
- GAGWJHPBXLXJQN-UORFTKCHSA-N Capecitabine Chemical compound C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1[C@H]1[C@H](O)[C@H](O)[C@@H](C)O1 GAGWJHPBXLXJQN-UORFTKCHSA-N 0.000 claims description 2
- GAGWJHPBXLXJQN-UHFFFAOYSA-N Capecitabine Natural products C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1C1C(O)C(O)C(C)O1 GAGWJHPBXLXJQN-UHFFFAOYSA-N 0.000 claims description 2
- DLGOEMSEDOSKAD-UHFFFAOYSA-N Carmustine Chemical compound ClCCNC(=O)N(N=O)CCCl DLGOEMSEDOSKAD-UHFFFAOYSA-N 0.000 claims description 2
- 108010092160 Dactinomycin Proteins 0.000 claims description 2
- 108010041356 Estrogen Receptor beta Proteins 0.000 claims description 2
- HKVAMNSJSFKALM-GKUWKFKPSA-N Everolimus Chemical compound C1C[C@@H](OCCO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 HKVAMNSJSFKALM-GKUWKFKPSA-N 0.000 claims description 2
- 108010068250 Herpes Simplex Virus Protein Vmw65 Proteins 0.000 claims description 2
- GQYIWUVLTXOXAJ-UHFFFAOYSA-N Lomustine Chemical compound ClCCN(N=O)C(=O)NC1CCCCC1 GQYIWUVLTXOXAJ-UHFFFAOYSA-N 0.000 claims description 2
- 229930192392 Mitomycin Natural products 0.000 claims description 2
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 claims description 2
- 239000012828 PI3K inhibitor Substances 0.000 claims description 2
- 229930012538 Paclitaxel Natural products 0.000 claims description 2
- 229940127397 Poly(ADP-Ribose) Polymerase Inhibitors Drugs 0.000 claims description 2
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 claims description 2
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 claims description 2
- 229940009456 adriamycin Drugs 0.000 claims description 2
- 229930013930 alkaloid Natural products 0.000 claims description 2
- 229940100198 alkylating agent Drugs 0.000 claims description 2
- 239000002168 alkylating agent Substances 0.000 claims description 2
- 229960002932 anastrozole Drugs 0.000 claims description 2
- YBBLVLTVTVSKRW-UHFFFAOYSA-N anastrozole Chemical compound N#CC(C)(C)C1=CC(C(C)(C#N)C)=CC(CN2N=CN=C2)=C1 YBBLVLTVTVSKRW-UHFFFAOYSA-N 0.000 claims description 2
- 230000002137 anti-vascular effect Effects 0.000 claims description 2
- 239000000051 antiandrogen Substances 0.000 claims description 2
- 229940088710 antibiotic agent Drugs 0.000 claims description 2
- 229950002916 avelumab Drugs 0.000 claims description 2
- 229960000397 bevacizumab Drugs 0.000 claims description 2
- 229960000997 bicalutamide Drugs 0.000 claims description 2
- 229960001561 bleomycin Drugs 0.000 claims description 2
- OYVAGSVQBOHSSS-UAPAGMARSA-O bleomycin A2 Chemical compound N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C OYVAGSVQBOHSSS-UAPAGMARSA-O 0.000 claims description 2
- 229960002092 busulfan Drugs 0.000 claims description 2
- 229960004117 capecitabine Drugs 0.000 claims description 2
- 229960004562 carboplatin Drugs 0.000 claims description 2
- 229960005243 carmustine Drugs 0.000 claims description 2
- 229960005395 cetuximab Drugs 0.000 claims description 2
- HWGQMRYQVZSGDQ-HZPDHXFCSA-N chembl3137320 Chemical compound CN1N=CN=C1[C@H]([C@H](N1)C=2C=CC(F)=CC=2)C2=NNC(=O)C3=C2C1=CC(F)=C3 HWGQMRYQVZSGDQ-HZPDHXFCSA-N 0.000 claims description 2
- 229960004630 chlorambucil Drugs 0.000 claims description 2
- JCKYGMPEJWAADB-UHFFFAOYSA-N chlorambucil Chemical compound OC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 JCKYGMPEJWAADB-UHFFFAOYSA-N 0.000 claims description 2
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 claims description 2
- 229960004316 cisplatin Drugs 0.000 claims description 2
- 229960002271 cobimetinib Drugs 0.000 claims description 2
- RESIMIUSNACMNW-BXRWSSRYSA-N cobimetinib fumarate Chemical compound OC(=O)\C=C\C(O)=O.C1C(O)([C@H]2NCCCC2)CN1C(=O)C1=CC=C(F)C(F)=C1NC1=CC=C(I)C=C1F.C1C(O)([C@H]2NCCCC2)CN1C(=O)C1=CC=C(F)C(F)=C1NC1=CC=C(I)C=C1F RESIMIUSNACMNW-BXRWSSRYSA-N 0.000 claims description 2
- 229960000640 dactinomycin Drugs 0.000 claims description 2
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 claims description 2
- 229960000975 daunorubicin Drugs 0.000 claims description 2
- 229960004679 doxorubicin Drugs 0.000 claims description 2
- 229950009791 durvalumab Drugs 0.000 claims description 2
- 229960004671 enzalutamide Drugs 0.000 claims description 2
- WXCXUHSOUPDCQV-UHFFFAOYSA-N enzalutamide Chemical compound C1=C(F)C(C(=O)NC)=CC=C1N1C(C)(C)C(=O)N(C=2C=C(C(C#N)=CC=2)C(F)(F)F)C1=S WXCXUHSOUPDCQV-UHFFFAOYSA-N 0.000 claims description 2
- YJGVMLPVUAXIQN-UHFFFAOYSA-N epipodophyllotoxin Natural products COC1=C(OC)C(OC)=CC(C2C3=CC=4OCOC=4C=C3C(O)C3C2C(OC3)=O)=C1 YJGVMLPVUAXIQN-UHFFFAOYSA-N 0.000 claims description 2
- 229960005167 everolimus Drugs 0.000 claims description 2
- 230000037440 gene silencing effect Effects 0.000 claims description 2
- 230000009459 hedgehog signaling Effects 0.000 claims description 2
- 238000009169 immunotherapy Methods 0.000 claims description 2
- 229950006331 ipatasertib Drugs 0.000 claims description 2
- 229960002247 lomustine Drugs 0.000 claims description 2
- 229940124302 mTOR inhibitor Drugs 0.000 claims description 2
- 239000003628 mammalian target of rapamycin inhibitor Substances 0.000 claims description 2
- 229960001924 melphalan Drugs 0.000 claims description 2
- SGDBTWWWUNNDEQ-LBPRGKRZSA-N melphalan Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N(CCCl)CCCl)C=C1 SGDBTWWWUNNDEQ-LBPRGKRZSA-N 0.000 claims description 2
- 239000002829 mitogen activated protein kinase inhibitor Substances 0.000 claims description 2
- 229960004857 mitomycin Drugs 0.000 claims description 2
- 229940035036 multi-peptide vaccine Drugs 0.000 claims description 2
- 229960003301 nivolumab Drugs 0.000 claims description 2
- 229960000572 olaparib Drugs 0.000 claims description 2
- FAQDUNYVKQKNLD-UHFFFAOYSA-N olaparib Chemical compound FC1=CC=C(CC2=C3[CH]C=CC=C3C(=O)N=N2)C=C1C(=O)N(CC1)CCN1C(=O)C1CC1 FAQDUNYVKQKNLD-UHFFFAOYSA-N 0.000 claims description 2
- 229960002621 pembrolizumab Drugs 0.000 claims description 2
- 229940043441 phosphoinositide 3-kinase inhibitor Drugs 0.000 claims description 2
- YJGVMLPVUAXIQN-XVVDYKMHSA-N podophyllotoxin Chemical compound COC1=C(OC)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@H](O)[C@@H]3[C@@H]2C(OC3)=O)=C1 YJGVMLPVUAXIQN-XVVDYKMHSA-N 0.000 claims description 2
- 229960001237 podophyllotoxin Drugs 0.000 claims description 2
- YVCVYCSAAZQOJI-UHFFFAOYSA-N podophyllotoxin Natural products COC1=C(O)C(OC)=CC(C2C3=CC=4OCOC=4C=C3C(O)C3C2C(OC3)=O)=C1 YVCVYCSAAZQOJI-UHFFFAOYSA-N 0.000 claims description 2
- 230000003439 radiotherapeutic effect Effects 0.000 claims description 2
- 229950004550 talazoparib Drugs 0.000 claims description 2
- XFCLJVABOIYOMF-QPLCGJKRSA-N toremifene Chemical compound C1=CC(OCCN(C)C)=CC=C1C(\C=1C=CC=CC=1)=C(\CCCl)C1=CC=CC=C1 XFCLJVABOIYOMF-QPLCGJKRSA-N 0.000 claims description 2
- 229960005026 toremifene Drugs 0.000 claims description 2
- 102000003390 tumor necrosis factor Human genes 0.000 claims description 2
- 229940124676 vascular endothelial growth factor receptor Drugs 0.000 claims description 2
- 229950011257 veliparib Drugs 0.000 claims description 2
- JNAHVYVRKWKWKQ-CYBMUJFWSA-N veliparib Chemical compound N=1C2=CC=CC(C(N)=O)=C2NC=1[C@@]1(C)CCCN1 JNAHVYVRKWKWKQ-CYBMUJFWSA-N 0.000 claims description 2
- 229960001722 verapamil Drugs 0.000 claims description 2
- 229960003048 vinblastine Drugs 0.000 claims description 2
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 claims description 2
- 229960004528 vincristine Drugs 0.000 claims description 2
- OGWKCGZFUXNPDA-XQKSVPLYSA-N vincristine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-XQKSVPLYSA-N 0.000 claims description 2
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 claims description 2
- 229960004449 vismodegib Drugs 0.000 claims description 2
- BPQMGSKTAYIVFO-UHFFFAOYSA-N vismodegib Chemical compound ClC1=CC(S(=O)(=O)C)=CC=C1C(=O)NC1=CC=C(Cl)C(C=2N=CC=CC=2)=C1 BPQMGSKTAYIVFO-UHFFFAOYSA-N 0.000 claims description 2
- 102000000509 Estrogen Receptor beta Human genes 0.000 claims 1
- 190000008236 carboplatin Chemical compound 0.000 claims 1
- 230000009368 gene silencing by RNA Effects 0.000 abstract description 38
- 238000012228 RNA interference-mediated gene silencing Methods 0.000 abstract description 34
- 201000011510 cancer Diseases 0.000 abstract description 23
- 230000012010 growth Effects 0.000 abstract description 13
- 230000009702 cancer cell proliferation Effects 0.000 abstract description 4
- 210000004027 cell Anatomy 0.000 description 209
- 230000014509 gene expression Effects 0.000 description 81
- 102000004169 proteins and genes Human genes 0.000 description 53
- 235000018102 proteins Nutrition 0.000 description 48
- 150000001875 compounds Chemical class 0.000 description 42
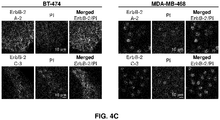
- 238000010166 immunofluorescence Methods 0.000 description 33
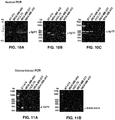
- 238000003752 polymerase chain reaction Methods 0.000 description 31
- 108091034117 Oligonucleotide Proteins 0.000 description 30
- 239000002585 base Substances 0.000 description 30
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 30
- 230000001225 therapeutic effect Effects 0.000 description 30
- 238000004458 analytical method Methods 0.000 description 27
- 102000039446 nucleic acids Human genes 0.000 description 27
- 108020004707 nucleic acids Proteins 0.000 description 27
- 150000007523 nucleic acids Chemical group 0.000 description 27
- 238000003364 immunohistochemistry Methods 0.000 description 26
- 210000001519 tissue Anatomy 0.000 description 26
- 101150023582 spt4 gene Proteins 0.000 description 25
- 101100403102 Mus musculus Mucl2 gene Proteins 0.000 description 24
- 201000010099 disease Diseases 0.000 description 24
- 238000002347 injection Methods 0.000 description 24
- 239000007924 injection Substances 0.000 description 24
- 238000002474 experimental method Methods 0.000 description 23
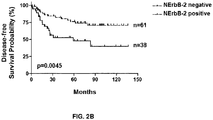
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 description 20
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 description 20
- 150000002632 lipids Chemical class 0.000 description 20
- 239000002502 liposome Substances 0.000 description 20
- 238000001727 in vivo Methods 0.000 description 19
- 230000004083 survival effect Effects 0.000 description 19
- 238000013519 translation Methods 0.000 description 19
- 238000013459 approach Methods 0.000 description 18
- 230000000694 effects Effects 0.000 description 18
- 210000004940 nucleus Anatomy 0.000 description 18
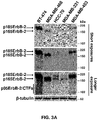
- 238000001262 western blot Methods 0.000 description 18
- 108700024394 Exon Proteins 0.000 description 17
- 208000035269 cancer or benign tumor Diseases 0.000 description 17
- 108091026890 Coding region Proteins 0.000 description 16
- 238000001514 detection method Methods 0.000 description 16
- 230000030279 gene silencing Effects 0.000 description 16
- 235000000346 sugar Nutrition 0.000 description 16
- 238000013461 design Methods 0.000 description 15
- 108020003589 5' Untranslated Regions Proteins 0.000 description 14
- 238000009472 formulation Methods 0.000 description 14
- 239000013598 vector Substances 0.000 description 14
- 239000012528 membrane Substances 0.000 description 13
- -1 purine compound Chemical class 0.000 description 13
- 238000010186 staining Methods 0.000 description 13
- 206010027476 Metastases Diseases 0.000 description 12
- 238000004624 confocal microscopy Methods 0.000 description 12
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 11
- 108010076504 Protein Sorting Signals Proteins 0.000 description 11
- 102000004243 Tubulin Human genes 0.000 description 11
- 108090000704 Tubulin Proteins 0.000 description 11
- 150000001413 amino acids Chemical class 0.000 description 11
- 208000036815 beta tubulin Diseases 0.000 description 11
- 125000002091 cationic group Chemical group 0.000 description 11
- 229940079593 drug Drugs 0.000 description 11
- 238000007857 nested PCR Methods 0.000 description 11
- 238000011529 RT qPCR Methods 0.000 description 10
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 10
- 230000000875 corresponding effect Effects 0.000 description 10
- 239000000412 dendrimer Substances 0.000 description 10
- 229920000736 dendritic polymer Polymers 0.000 description 10
- 238000011068 loading method Methods 0.000 description 10
- 230000007935 neutral effect Effects 0.000 description 10
- 102000040430 polynucleotide Human genes 0.000 description 10
- 108091033319 polynucleotide Proteins 0.000 description 10
- 239000002157 polynucleotide Substances 0.000 description 10
- 238000012360 testing method Methods 0.000 description 10
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 9
- 108020004414 DNA Proteins 0.000 description 9
- 102100034343 Integrase Human genes 0.000 description 9
- 241000699670 Mus sp. Species 0.000 description 9
- 108010092799 RNA-directed DNA polymerase Proteins 0.000 description 9
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 9
- 229940024606 amino acid Drugs 0.000 description 9
- 235000001014 amino acid Nutrition 0.000 description 9
- 210000001808 exosome Anatomy 0.000 description 9
- 102000006602 glyceraldehyde-3-phosphate dehydrogenase Human genes 0.000 description 9
- 108020004445 glyceraldehyde-3-phosphate dehydrogenase Proteins 0.000 description 9
- 210000004962 mammalian cell Anatomy 0.000 description 9
- 239000002105 nanoparticle Substances 0.000 description 9
- 238000011002 quantification Methods 0.000 description 9
- 230000002441 reversible effect Effects 0.000 description 9
- 238000001890 transfection Methods 0.000 description 9
- FWBHETKCLVMNFS-UHFFFAOYSA-N 4',6-Diamino-2-phenylindol Chemical compound C1=CC(C(=N)N)=CC=C1C1=CC2=CC=C(C(N)=N)C=C2N1 FWBHETKCLVMNFS-UHFFFAOYSA-N 0.000 description 8
- 241000699666 Mus <mouse, genus> Species 0.000 description 8
- 102400000058 Neuregulin-1 Human genes 0.000 description 8
- 108090000556 Neuregulin-1 Proteins 0.000 description 8
- 230000004913 activation Effects 0.000 description 8
- 239000013543 active substance Substances 0.000 description 8
- 239000000090 biomarker Substances 0.000 description 8
- 230000001413 cellular effect Effects 0.000 description 8
- 230000008859 change Effects 0.000 description 8
- 238000003776 cleavage reaction Methods 0.000 description 8
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 8
- 230000009401 metastasis Effects 0.000 description 8
- 210000000056 organ Anatomy 0.000 description 8
- 102000003998 progesterone receptors Human genes 0.000 description 8
- 108090000468 progesterone receptors Proteins 0.000 description 8
- 230000002829 reductive effect Effects 0.000 description 8
- 230000007017 scission Effects 0.000 description 8
- 230000004988 N-glycosylation Effects 0.000 description 7
- 230000003321 amplification Effects 0.000 description 7
- 210000000481 breast Anatomy 0.000 description 7
- 210000000170 cell membrane Anatomy 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 230000005764 inhibitory process Effects 0.000 description 7
- 230000001404 mediated effect Effects 0.000 description 7
- 238000003199 nucleic acid amplification method Methods 0.000 description 7
- 238000011160 research Methods 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- 230000002103 transcriptional effect Effects 0.000 description 7
- 239000003981 vehicle Substances 0.000 description 7
- 108020005345 3' Untranslated Regions Proteins 0.000 description 6
- 101150043871 MAPK7 gene Proteins 0.000 description 6
- 241000283973 Oryctolagus cuniculus Species 0.000 description 6
- 239000002202 Polyethylene glycol Substances 0.000 description 6
- 101150099493 STAT3 gene Proteins 0.000 description 6
- 210000004556 brain Anatomy 0.000 description 6
- 239000000872 buffer Substances 0.000 description 6
- 238000004422 calculation algorithm Methods 0.000 description 6
- 230000005907 cancer growth Effects 0.000 description 6
- 230000015556 catabolic process Effects 0.000 description 6
- 230000002860 competitive effect Effects 0.000 description 6
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical compound NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 6
- 238000006731 degradation reaction Methods 0.000 description 6
- 238000000326 densiometry Methods 0.000 description 6
- 208000035475 disorder Diseases 0.000 description 6
- 238000005516 engineering process Methods 0.000 description 6
- 230000001965 increasing effect Effects 0.000 description 6
- 210000000265 leukocyte Anatomy 0.000 description 6
- 210000001165 lymph node Anatomy 0.000 description 6
- 230000007246 mechanism Effects 0.000 description 6
- 230000000394 mitotic effect Effects 0.000 description 6
- 229920001223 polyethylene glycol Polymers 0.000 description 6
- 238000003762 quantitative reverse transcription PCR Methods 0.000 description 6
- 238000009097 single-agent therapy Methods 0.000 description 6
- RWQNBRDOKXIBIV-UHFFFAOYSA-N thymine Chemical compound CC1=CNC(=O)NC1=O RWQNBRDOKXIBIV-UHFFFAOYSA-N 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 5
- 108010051109 Cell-Penetrating Peptides Proteins 0.000 description 5
- 102000020313 Cell-Penetrating Peptides Human genes 0.000 description 5
- 108020004635 Complementary DNA Proteins 0.000 description 5
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 5
- 108010001336 Horseradish Peroxidase Proteins 0.000 description 5
- 101710205482 Nuclear factor 1 A-type Proteins 0.000 description 5
- 102100022165 Nuclear factor 1 B-type Human genes 0.000 description 5
- 101710170464 Nuclear factor 1 B-type Proteins 0.000 description 5
- 101710113455 Nuclear factor 1 C-type Proteins 0.000 description 5
- 101710140810 Nuclear factor 1 X-type Proteins 0.000 description 5
- 102220497176 Small vasohibin-binding protein_T47D_mutation Human genes 0.000 description 5
- 210000004369 blood Anatomy 0.000 description 5
- 239000008280 blood Substances 0.000 description 5
- 210000004899 c-terminal region Anatomy 0.000 description 5
- 229910052799 carbon Inorganic materials 0.000 description 5
- 238000004113 cell culture Methods 0.000 description 5
- 238000007385 chemical modification Methods 0.000 description 5
- 235000012000 cholesterol Nutrition 0.000 description 5
- 230000001419 dependent effect Effects 0.000 description 5
- 239000000262 estrogen Substances 0.000 description 5
- 238000010348 incorporation Methods 0.000 description 5
- 238000011534 incubation Methods 0.000 description 5
- 208000026534 luminal B breast carcinoma Diseases 0.000 description 5
- 239000006166 lysate Substances 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 206010061289 metastatic neoplasm Diseases 0.000 description 5
- 238000000491 multivariate analysis Methods 0.000 description 5
- 239000013642 negative control Substances 0.000 description 5
- 230000002018 overexpression Effects 0.000 description 5
- 229920000656 polylysine Polymers 0.000 description 5
- XJMOSONTPMZWPB-UHFFFAOYSA-M propidium iodide Chemical compound [I-].[I-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CCC[N+](C)(CC)CC)=C1C1=CC=CC=C1 XJMOSONTPMZWPB-UHFFFAOYSA-M 0.000 description 5
- 238000004445 quantitative analysis Methods 0.000 description 5
- 238000003753 real-time PCR Methods 0.000 description 5
- 230000004044 response Effects 0.000 description 5
- 239000000725 suspension Substances 0.000 description 5
- 231100000419 toxicity Toxicity 0.000 description 5
- 230000001988 toxicity Effects 0.000 description 5
- 239000012096 transfection reagent Substances 0.000 description 5
- 239000012103 Alexa Fluor 488 Substances 0.000 description 4
- 108091093088 Amplicon Proteins 0.000 description 4
- 102000008096 B7-H1 Antigen Human genes 0.000 description 4
- 108010074708 B7-H1 Antigen Proteins 0.000 description 4
- 108010014303 DNA-directed DNA polymerase Proteins 0.000 description 4
- 102000016928 DNA-directed DNA polymerase Human genes 0.000 description 4
- 102000004190 Enzymes Human genes 0.000 description 4
- 108090000790 Enzymes Proteins 0.000 description 4
- 238000000729 Fisher's exact test Methods 0.000 description 4
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 4
- 241001465754 Metazoa Species 0.000 description 4
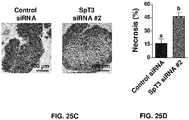
- 206010028851 Necrosis Diseases 0.000 description 4
- KDCGOANMDULRCW-UHFFFAOYSA-N Purine Natural products N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 description 4
- 108091028664 Ribonucleotide Proteins 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 108010006785 Taq Polymerase Proteins 0.000 description 4
- IQFYYKKMVGJFEH-XLPZGREQSA-N Thymidine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 IQFYYKKMVGJFEH-XLPZGREQSA-N 0.000 description 4
- ISAKRJDGNUQOIC-UHFFFAOYSA-N Uracil Chemical compound O=C1C=CNC(=O)N1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 0.000 description 4
- 241000700605 Viruses Species 0.000 description 4
- MBMBGCFOFBJSGT-KUBAVDMBSA-N all-cis-docosa-4,7,10,13,16,19-hexaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCC(O)=O MBMBGCFOFBJSGT-KUBAVDMBSA-N 0.000 description 4
- 238000000137 annealing Methods 0.000 description 4
- 238000002869 basic local alignment search tool Methods 0.000 description 4
- 230000027455 binding Effects 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 238000004364 calculation method Methods 0.000 description 4
- 239000013311 covalent triazine framework Substances 0.000 description 4
- 230000001086 cytosolic effect Effects 0.000 description 4
- 230000003247 decreasing effect Effects 0.000 description 4
- 230000018109 developmental process Effects 0.000 description 4
- UKMSUNONTOPOIO-UHFFFAOYSA-N docosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCC(O)=O UKMSUNONTOPOIO-UHFFFAOYSA-N 0.000 description 4
- 210000003743 erythrocyte Anatomy 0.000 description 4
- 229940011871 estrogen Drugs 0.000 description 4
- 239000000284 extract Substances 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- 238000012226 gene silencing method Methods 0.000 description 4
- 230000009036 growth inhibition Effects 0.000 description 4
- UYTPUPDQBNUYGX-UHFFFAOYSA-N guanine Chemical compound O=C1NC(N)=NC2=C1N=CN2 UYTPUPDQBNUYGX-UHFFFAOYSA-N 0.000 description 4
- 238000009396 hybridization Methods 0.000 description 4
- 229910052739 hydrogen Inorganic materials 0.000 description 4
- 239000001257 hydrogen Substances 0.000 description 4
- 230000000977 initiatory effect Effects 0.000 description 4
- 230000003993 interaction Effects 0.000 description 4
- 230000002452 interceptive effect Effects 0.000 description 4
- 239000003446 ligand Substances 0.000 description 4
- 230000004807 localization Effects 0.000 description 4
- 230000001394 metastastic effect Effects 0.000 description 4
- 230000005012 migration Effects 0.000 description 4
- 238000013508 migration Methods 0.000 description 4
- 230000017074 necrotic cell death Effects 0.000 description 4
- 238000011580 nude mouse model Methods 0.000 description 4
- 231100000590 oncogenic Toxicity 0.000 description 4
- 230000002246 oncogenic effect Effects 0.000 description 4
- 238000001543 one-way ANOVA Methods 0.000 description 4
- 239000002245 particle Substances 0.000 description 4
- 238000006366 phosphorylation reaction Methods 0.000 description 4
- 229920000729 poly(L-lysine) polymer Polymers 0.000 description 4
- 229920000642 polymer Polymers 0.000 description 4
- 102000004196 processed proteins & peptides Human genes 0.000 description 4
- 108090000765 processed proteins & peptides Proteins 0.000 description 4
- 238000001959 radiotherapy Methods 0.000 description 4
- 239000002336 ribonucleotide Substances 0.000 description 4
- 125000002652 ribonucleotide group Chemical group 0.000 description 4
- 230000000638 stimulation Effects 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- 208000024891 symptom Diseases 0.000 description 4
- 230000009885 systemic effect Effects 0.000 description 4
- 230000004614 tumor growth Effects 0.000 description 4
- VYEWZWBILJHHCU-OMQUDAQFSA-N (e)-n-[(2s,3r,4r,5r,6r)-2-[(2r,3r,4s,5s,6s)-3-acetamido-5-amino-4-hydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-6-[2-[(2r,3s,4r,5r)-5-(2,4-dioxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]-2-hydroxyethyl]-4,5-dihydroxyoxan-3-yl]-5-methylhex-2-enamide Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@H]2O)O)C(O)C[C@@H]2[C@H](O)[C@H](O)[C@H]([C@@H](O2)O[C@@H]2[C@@H]([C@@H](O)[C@H](N)[C@@H](CO)O2)NC(C)=O)NC(=O)/C=C/CC(C)C)C=CC(=O)NC1=O VYEWZWBILJHHCU-OMQUDAQFSA-N 0.000 description 3
- HSTOKWSFWGCZMH-UHFFFAOYSA-N 3,3'-diaminobenzidine Chemical compound C1=C(N)C(N)=CC=C1C1=CC=C(N)C(N)=C1 HSTOKWSFWGCZMH-UHFFFAOYSA-N 0.000 description 3
- 229930024421 Adenine Natural products 0.000 description 3
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 3
- 108020004774 Alkaline Phosphatase Proteins 0.000 description 3
- 102000002260 Alkaline Phosphatase Human genes 0.000 description 3
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- 108020004705 Codon Proteins 0.000 description 3
- 125000000824 D-ribofuranosyl group Chemical group [H]OC([H])([H])[C@@]1([H])OC([H])(*)[C@]([H])(O[H])[C@]1([H])O[H] 0.000 description 3
- 102000053602 DNA Human genes 0.000 description 3
- 230000004544 DNA amplification Effects 0.000 description 3
- 108010043121 Green Fluorescent Proteins Proteins 0.000 description 3
- 102000004144 Green Fluorescent Proteins Human genes 0.000 description 3
- 102000001554 Hemoglobins Human genes 0.000 description 3
- 108010054147 Hemoglobins Proteins 0.000 description 3
- 108700020796 Oncogene Proteins 0.000 description 3
- 108091000080 Phosphotransferase Proteins 0.000 description 3
- RJKFOVLPORLFTN-LEKSSAKUSA-N Progesterone Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H](C(=O)C)[C@@]1(C)CC2 RJKFOVLPORLFTN-LEKSSAKUSA-N 0.000 description 3
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 3
- 238000000692 Student's t-test Methods 0.000 description 3
- 102000040945 Transcription factor Human genes 0.000 description 3
- 108091023040 Transcription factor Proteins 0.000 description 3
- YJQCOFNZVFGCAF-UHFFFAOYSA-N Tunicamycin II Natural products O1C(CC(O)C2C(C(O)C(O2)N2C(NC(=O)C=C2)=O)O)C(O)C(O)C(NC(=O)C=CCCCCCCCCC(C)C)C1OC1OC(CO)C(O)C(O)C1NC(C)=O YJQCOFNZVFGCAF-UHFFFAOYSA-N 0.000 description 3
- 108091023045 Untranslated Region Proteins 0.000 description 3
- 229960000643 adenine Drugs 0.000 description 3
- 210000000577 adipose tissue Anatomy 0.000 description 3
- 239000002671 adjuvant Substances 0.000 description 3
- 239000011543 agarose gel Substances 0.000 description 3
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 description 3
- 230000009286 beneficial effect Effects 0.000 description 3
- 230000008499 blood brain barrier function Effects 0.000 description 3
- 210000001218 blood-brain barrier Anatomy 0.000 description 3
- 239000012830 cancer therapeutic Substances 0.000 description 3
- 239000013592 cell lysate Substances 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 238000002648 combination therapy Methods 0.000 description 3
- 239000002299 complementary DNA Substances 0.000 description 3
- 229940104302 cytosine Drugs 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 238000002716 delivery method Methods 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 238000003745 diagnosis Methods 0.000 description 3
- 238000002405 diagnostic procedure Methods 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 239000000975 dye Substances 0.000 description 3
- 108700015053 epidermal growth factor receptor activity proteins Proteins 0.000 description 3
- 102000052116 epidermal growth factor receptor activity proteins Human genes 0.000 description 3
- 238000005194 fractionation Methods 0.000 description 3
- 239000012634 fragment Substances 0.000 description 3
- 102000054767 gene variant Human genes 0.000 description 3
- 239000005090 green fluorescent protein Substances 0.000 description 3
- 238000007490 hematoxylin and eosin (H&E) staining Methods 0.000 description 3
- 238000007489 histopathology method Methods 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 230000002519 immonomodulatory effect Effects 0.000 description 3
- 238000001802 infusion Methods 0.000 description 3
- 238000007913 intrathecal administration Methods 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 210000004185 liver Anatomy 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 230000002503 metabolic effect Effects 0.000 description 3
- 230000000813 microbial effect Effects 0.000 description 3
- YOHYSYJDKVYCJI-UHFFFAOYSA-N n-[3-[[6-[3-(trifluoromethyl)anilino]pyrimidin-4-yl]amino]phenyl]cyclopropanecarboxamide Chemical compound FC(F)(F)C1=CC=CC(NC=2N=CN=C(NC=3C=C(NC(=O)C4CC4)C=CC=3)C=2)=C1 YOHYSYJDKVYCJI-UHFFFAOYSA-N 0.000 description 3
- 230000017111 nuclear migration Effects 0.000 description 3
- 210000003463 organelle Anatomy 0.000 description 3
- 238000007911 parenteral administration Methods 0.000 description 3
- 230000037361 pathway Effects 0.000 description 3
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 description 3
- 230000026731 phosphorylation Effects 0.000 description 3
- 102000020233 phosphotransferase Human genes 0.000 description 3
- 230000023603 positive regulation of transcription initiation, DNA-dependent Effects 0.000 description 3
- 230000003389 potentiating effect Effects 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 239000000583 progesterone congener Substances 0.000 description 3
- 239000002096 quantum dot Substances 0.000 description 3
- 229930002330 retinoic acid Natural products 0.000 description 3
- 229920002477 rna polymer Polymers 0.000 description 3
- 238000007480 sanger sequencing Methods 0.000 description 3
- 230000035945 sensitivity Effects 0.000 description 3
- 230000011664 signaling Effects 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 241000894007 species Species 0.000 description 3
- 210000000278 spinal cord Anatomy 0.000 description 3
- 238000001356 surgical procedure Methods 0.000 description 3
- 229940124597 therapeutic agent Drugs 0.000 description 3
- 229940113082 thymine Drugs 0.000 description 3
- 231100000331 toxic Toxicity 0.000 description 3
- 230000002588 toxic effect Effects 0.000 description 3
- 238000013518 transcription Methods 0.000 description 3
- 230000035897 transcription Effects 0.000 description 3
- 230000009466 transformation Effects 0.000 description 3
- 229960000575 trastuzumab Drugs 0.000 description 3
- 229960001727 tretinoin Drugs 0.000 description 3
- MEYZYGMYMLNUHJ-UHFFFAOYSA-N tunicamycin Natural products CC(C)CCCCCCCCCC=CC(=O)NC1C(O)C(O)C(CC(O)C2OC(C(O)C2O)N3C=CC(=O)NC3=O)OC1OC4OC(CO)C(O)C(O)C4NC(=O)C MEYZYGMYMLNUHJ-UHFFFAOYSA-N 0.000 description 3
- 238000007473 univariate analysis Methods 0.000 description 3
- SNKAWJBJQDLSFF-NVKMUCNASA-N 1,2-dioleoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/CCCCCCCC SNKAWJBJQDLSFF-NVKMUCNASA-N 0.000 description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 2
- 102100036475 Alanine aminotransferase 1 Human genes 0.000 description 2
- 108010082126 Alanine transaminase Proteins 0.000 description 2
- 102100032187 Androgen receptor Human genes 0.000 description 2
- 108010003415 Aspartate Aminotransferases Proteins 0.000 description 2
- 102000004625 Aspartate Aminotransferases Human genes 0.000 description 2
- 235000021357 Behenic acid Nutrition 0.000 description 2
- BPYKTIZUTYGOLE-IFADSCNNSA-N Bilirubin Chemical compound N1C(=O)C(C)=C(C=C)\C1=C\C1=C(C)C(CCC(O)=O)=C(CC2=C(C(C)=C(\C=C/3C(=C(C=C)C(=O)N\3)C)N2)CCC(O)=O)N1 BPYKTIZUTYGOLE-IFADSCNNSA-N 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- 241000283707 Capra Species 0.000 description 2
- IELOKBJPULMYRW-NJQVLOCASA-N D-alpha-Tocopheryl Acid Succinate Chemical compound OC(=O)CCC(=O)OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C IELOKBJPULMYRW-NJQVLOCASA-N 0.000 description 2
- HMFHBZSHGGEWLO-SOOFDHNKSA-N D-ribofuranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H]1O HMFHBZSHGGEWLO-SOOFDHNKSA-N 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- 102000001301 EGF receptor Human genes 0.000 description 2
- 108060006698 EGF receptor Proteins 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- 108060002716 Exonuclease Proteins 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- 102100037907 High mobility group protein B1 Human genes 0.000 description 2
- 101710168537 High mobility group protein B1 Proteins 0.000 description 2
- 108010033040 Histones Proteins 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 101000950687 Homo sapiens Mitogen-activated protein kinase 7 Proteins 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 238000010824 Kaplan-Meier survival analysis Methods 0.000 description 2
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 2
- 239000002136 L01XE07 - Lapatinib Substances 0.000 description 2
- 239000000232 Lipid Bilayer Substances 0.000 description 2
- SMEROWZSTRWXGI-UHFFFAOYSA-N Lithocholsaeure Natural products C1CC2CC(O)CCC2(C)C2C1C1CCC(C(CCC(O)=O)C)C1(C)CC2 SMEROWZSTRWXGI-UHFFFAOYSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 108091027974 Mature messenger RNA Proteins 0.000 description 2
- 108010052285 Membrane Proteins Proteins 0.000 description 2
- 102100037805 Mitogen-activated protein kinase 7 Human genes 0.000 description 2
- 101710163270 Nuclease Proteins 0.000 description 2
- 108091028043 Nucleic acid sequence Proteins 0.000 description 2
- 108700026244 Open Reading Frames Proteins 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- 229920002873 Polyethylenimine Polymers 0.000 description 2
- 108010039918 Polylysine Proteins 0.000 description 2
- 238000012181 QIAquick gel extraction kit Methods 0.000 description 2
- 102000004278 Receptor Protein-Tyrosine Kinases Human genes 0.000 description 2
- 108090000873 Receptor Protein-Tyrosine Kinases Proteins 0.000 description 2
- 101710100968 Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 description 2
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 2
- 108010017324 STAT3 Transcription Factor Proteins 0.000 description 2
- 102100024040 Signal transducer and activator of transcription 3 Human genes 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 229940123237 Taxane Drugs 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 125000002015 acyclic group Chemical group 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 108010080146 androgen receptors Proteins 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 229940045799 anthracyclines and related substance Drugs 0.000 description 2
- 239000000427 antigen Substances 0.000 description 2
- 108091007433 antigens Proteins 0.000 description 2
- 102000036639 antigens Human genes 0.000 description 2
- 230000006907 apoptotic process Effects 0.000 description 2
- 239000012736 aqueous medium Substances 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- 229940116226 behenic acid Drugs 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 239000003012 bilayer membrane Substances 0.000 description 2
- 210000000988 bone and bone Anatomy 0.000 description 2
- 238000010804 cDNA synthesis Methods 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 229920006317 cationic polymer Polymers 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 230000004663 cell proliferation Effects 0.000 description 2
- 230000004700 cellular uptake Effects 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 230000003081 coactivator Effects 0.000 description 2
- 230000021615 conjugation Effects 0.000 description 2
- DDRJAANPRJIHGJ-UHFFFAOYSA-N creatinine Chemical compound CN1CC(=O)NC1=N DDRJAANPRJIHGJ-UHFFFAOYSA-N 0.000 description 2
- VFLDPWHFBUODDF-FCXRPNKRSA-N curcumin Chemical compound C1=C(O)C(OC)=CC(\C=C\C(=O)CC(=O)\C=C\C=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-FCXRPNKRSA-N 0.000 description 2
- 230000001351 cycling effect Effects 0.000 description 2
- 230000002939 deleterious effect Effects 0.000 description 2
- 238000012217 deletion Methods 0.000 description 2
- 230000037430 deletion Effects 0.000 description 2
- 239000005547 deoxyribonucleotide Substances 0.000 description 2
- 125000002637 deoxyribonucleotide group Chemical group 0.000 description 2
- 235000020669 docosahexaenoic acid Nutrition 0.000 description 2
- KFEVDPWXEVUUMW-UHFFFAOYSA-N docosanoic acid Natural products CCCCCCCCCCCCCCCCCCCCCC(=O)OCCC1=CC=C(O)C=C1 KFEVDPWXEVUUMW-UHFFFAOYSA-N 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 230000003828 downregulation Effects 0.000 description 2
- 238000009509 drug development Methods 0.000 description 2
- 238000007876 drug discovery Methods 0.000 description 2
- 230000002900 effect on cell Effects 0.000 description 2
- 235000020673 eicosapentaenoic acid Nutrition 0.000 description 2
- 238000004520 electroporation Methods 0.000 description 2
- 230000009881 electrostatic interaction Effects 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 230000002327 eosinophilic effect Effects 0.000 description 2
- 229960005542 ethidium bromide Drugs 0.000 description 2
- ZMMJGEGLRURXTF-UHFFFAOYSA-N ethidium bromide Chemical compound [Br-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CC)=C1C1=CC=CC=C1 ZMMJGEGLRURXTF-UHFFFAOYSA-N 0.000 description 2
- 230000007717 exclusion Effects 0.000 description 2
- 102000013165 exonuclease Human genes 0.000 description 2
- 238000010195 expression analysis Methods 0.000 description 2
- 235000019197 fats Nutrition 0.000 description 2
- 238000009093 first-line therapy Methods 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 230000013595 glycosylation Effects 0.000 description 2
- 230000002489 hematologic effect Effects 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 102000051957 human ERBB2 Human genes 0.000 description 2
- 210000005260 human cell Anatomy 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- FDGQSTZJBFJUBT-UHFFFAOYSA-N hypoxanthine Chemical compound O=C1NC=NC2=C1NC=N2 FDGQSTZJBFJUBT-UHFFFAOYSA-N 0.000 description 2
- 238000003119 immunoblot Methods 0.000 description 2
- 238000011532 immunohistochemical staining Methods 0.000 description 2
- 230000001976 improved effect Effects 0.000 description 2
- 230000001939 inductive effect Effects 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 238000011221 initial treatment Methods 0.000 description 2
- 239000000543 intermediate Substances 0.000 description 2
- 230000003834 intracellular effect Effects 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 230000003447 ipsilateral effect Effects 0.000 description 2
- 238000012729 kappa analysis Methods 0.000 description 2
- 230000003907 kidney function Effects 0.000 description 2
- BCFGMOOMADDAQU-UHFFFAOYSA-N lapatinib Chemical compound O1C(CNCCS(=O)(=O)C)=CC=C1C1=CC=C(N=CN=C2NC=3C=C(Cl)C(OCC=4C=C(F)C=CC=4)=CC=3)C2=C1 BCFGMOOMADDAQU-UHFFFAOYSA-N 0.000 description 2
- 229960004891 lapatinib Drugs 0.000 description 2
- SMEROWZSTRWXGI-HVATVPOCSA-N lithocholic acid Chemical compound C([C@H]1CC2)[C@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@@H](CCC(O)=O)C)[C@@]2(C)CC1 SMEROWZSTRWXGI-HVATVPOCSA-N 0.000 description 2
- 238000001325 log-rank test Methods 0.000 description 2
- 208000026535 luminal A breast carcinoma Diseases 0.000 description 2
- 210000004072 lung Anatomy 0.000 description 2
- 210000004698 lymphocyte Anatomy 0.000 description 2
- 229910001629 magnesium chloride Inorganic materials 0.000 description 2
- 239000002122 magnetic nanoparticle Substances 0.000 description 2
- 238000007726 management method Methods 0.000 description 2
- 108010082117 matrigel Proteins 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 230000010534 mechanism of action Effects 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- POULHZVOKOAJMA-UHFFFAOYSA-N methyl undecanoic acid Natural products CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 239000002679 microRNA Substances 0.000 description 2
- 238000000520 microinjection Methods 0.000 description 2
- 238000000386 microscopy Methods 0.000 description 2
- 238000011201 multiple comparisons test Methods 0.000 description 2
- 230000001338 necrotic effect Effects 0.000 description 2
- 210000000440 neutrophil Anatomy 0.000 description 2
- 231100000956 nontoxicity Toxicity 0.000 description 2
- 238000010606 normalization Methods 0.000 description 2
- 230000008689 nuclear function Effects 0.000 description 2
- 238000012758 nuclear staining Methods 0.000 description 2
- 230000005937 nuclear translocation Effects 0.000 description 2
- 239000002777 nucleoside Substances 0.000 description 2
- 150000003833 nucleoside derivatives Chemical class 0.000 description 2
- 230000030648 nucleus localization Effects 0.000 description 2
- 230000009437 off-target effect Effects 0.000 description 2
- 238000011275 oncology therapy Methods 0.000 description 2
- 239000010452 phosphate Substances 0.000 description 2
- 150000003904 phospholipids Chemical class 0.000 description 2
- 239000013612 plasmid Substances 0.000 description 2
- 229920000962 poly(amidoamine) Polymers 0.000 description 2
- 229920002643 polyglutamic acid Polymers 0.000 description 2
- 229920001184 polypeptide Polymers 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 239000003531 protein hydrolysate Substances 0.000 description 2
- 230000006337 proteolytic cleavage Effects 0.000 description 2
- 230000010837 receptor-mediated endocytosis Effects 0.000 description 2
- 125000000548 ribosyl group Chemical group C1([C@H](O)[C@H](O)[C@H](O1)CO)* 0.000 description 2
- 235000003441 saturated fatty acids Nutrition 0.000 description 2
- 150000004671 saturated fatty acids Chemical class 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000001509 sodium citrate Substances 0.000 description 2
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 2
- 230000006641 stabilisation Effects 0.000 description 2
- 238000011105 stabilization Methods 0.000 description 2
- 230000004960 subcellular localization Effects 0.000 description 2
- 150000008163 sugars Chemical class 0.000 description 2
- 230000001629 suppression Effects 0.000 description 2
- 230000002459 sustained effect Effects 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 238000002626 targeted therapy Methods 0.000 description 2
- 230000002110 toxicologic effect Effects 0.000 description 2
- 231100000027 toxicology Toxicity 0.000 description 2
- 238000010361 transduction Methods 0.000 description 2
- 230000026683 transduction Effects 0.000 description 2
- 239000000439 tumor marker Substances 0.000 description 2
- 229940035893 uracil Drugs 0.000 description 2
- 108700043108 vasectrin III Proteins 0.000 description 2
- 238000012800 visualization Methods 0.000 description 2
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 1
- UEMGWPRHOOEKTA-UHFFFAOYSA-N 1,3-difluorobenzene Chemical compound FC1=CC=CC(F)=C1 UEMGWPRHOOEKTA-UHFFFAOYSA-N 0.000 description 1
- UIDRIVJQZGXVCM-XVFCMESISA-N 1-[(2r,3r,4r,5r)-4-hydroxy-5-(hydroxymethyl)-3-sulfanyloxolan-2-yl]pyrimidine-2,4-dione Chemical compound S[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C=C1 UIDRIVJQZGXVCM-XVFCMESISA-N 0.000 description 1
- 108010058566 130-nm albumin-bound paclitaxel Proteins 0.000 description 1
- AEQDJSLRWYMAQI-UHFFFAOYSA-N 2,3,9,10-tetramethoxy-6,8,13,13a-tetrahydro-5H-isoquinolino[2,1-b]isoquinoline Chemical compound C1CN2CC(C(=C(OC)C=C3)OC)=C3CC2C2=C1C=C(OC)C(OC)=C2 AEQDJSLRWYMAQI-UHFFFAOYSA-N 0.000 description 1
- XKZQKPRCPNGNFR-UHFFFAOYSA-N 2-(3-hydroxyphenyl)phenol Chemical compound OC1=CC=CC(C=2C(=CC=CC=2)O)=C1 XKZQKPRCPNGNFR-UHFFFAOYSA-N 0.000 description 1
- XWNJMSJGJFSGRY-UHFFFAOYSA-N 2-(benzylamino)-3,7-dihydropurin-6-one Chemical compound N1C=2N=CNC=2C(=O)N=C1NCC1=CC=CC=C1 XWNJMSJGJFSGRY-UHFFFAOYSA-N 0.000 description 1
- ASJSAQIRZKANQN-CRCLSJGQSA-N 2-deoxy-D-ribose Chemical compound OC[C@@H](O)[C@@H](O)CC=O ASJSAQIRZKANQN-CRCLSJGQSA-N 0.000 description 1
- SVNCRRZKBNSMIV-UHFFFAOYSA-N 3-Aminoquinoline Chemical compound C1=CC=CC2=CC(N)=CN=C21 SVNCRRZKBNSMIV-UHFFFAOYSA-N 0.000 description 1
- CYDQOEWLBCCFJZ-UHFFFAOYSA-N 4-(4-fluorophenyl)oxane-4-carboxylic acid Chemical compound C=1C=C(F)C=CC=1C1(C(=O)O)CCOCC1 CYDQOEWLBCCFJZ-UHFFFAOYSA-N 0.000 description 1
- QCXGJTGMGJOYDP-UHFFFAOYSA-N 4-methyl-1h-benzimidazole Chemical compound CC1=CC=CC2=C1N=CN2 QCXGJTGMGJOYDP-UHFFFAOYSA-N 0.000 description 1
- MSSXOMSJDRHRMC-UHFFFAOYSA-N 9H-purine-2,6-diamine Chemical compound NC1=NC(N)=C2NC=NC2=N1 MSSXOMSJDRHRMC-UHFFFAOYSA-N 0.000 description 1
- 101800000112 Acidic peptide Proteins 0.000 description 1
- 241000589158 Agrobacterium Species 0.000 description 1
- 230000007730 Akt signaling Effects 0.000 description 1
- ZAINTDRBUHCDPZ-UHFFFAOYSA-M Alexa Fluor 546 Substances [H+].[Na+].CC1CC(C)(C)NC(C(=C2OC3=C(C4=NC(C)(C)CC(C)C4=CC3=3)S([O-])(=O)=O)S([O-])(=O)=O)=C1C=C2C=3C(C(=C(Cl)C=1Cl)C(O)=O)=C(Cl)C=1SCC(=O)NCCCCCC(=O)ON1C(=O)CCC1=O ZAINTDRBUHCDPZ-UHFFFAOYSA-M 0.000 description 1
- 239000012099 Alexa Fluor family Substances 0.000 description 1
- 108700028369 Alleles Proteins 0.000 description 1
- 102100034613 Annexin A2 Human genes 0.000 description 1
- 108090000668 Annexin A2 Proteins 0.000 description 1
- 101100519158 Arabidopsis thaliana PCR2 gene Proteins 0.000 description 1
- 208000003174 Brain Neoplasms Diseases 0.000 description 1
- 241000244203 Caenorhabditis elegans Species 0.000 description 1
- 101000715943 Caenorhabditis elegans Cyclin-dependent kinase 4 homolog Proteins 0.000 description 1
- 208000005623 Carcinogenesis Diseases 0.000 description 1
- 206010057248 Cell death Diseases 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- 108010025468 Cyclin-Dependent Kinase 6 Proteins 0.000 description 1
- 102100026804 Cyclin-dependent kinase 6 Human genes 0.000 description 1
- 102100033270 Cyclin-dependent kinase inhibitor 1 Human genes 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- FBPFZTCFMRRESA-KAZBKCHUSA-N D-altritol Chemical compound OC[C@@H](O)[C@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KAZBKCHUSA-N 0.000 description 1
- 241000702421 Dependoparvovirus Species 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- 101150039808 Egfr gene Proteins 0.000 description 1
- 108010093099 Endoribonucleases Proteins 0.000 description 1
- 102000002494 Endoribonucleases Human genes 0.000 description 1
- 241000283074 Equus asinus Species 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 108090000371 Esterases Proteins 0.000 description 1
- 102100029951 Estrogen receptor beta Human genes 0.000 description 1
- 239000001116 FEMA 4028 Substances 0.000 description 1
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 description 1
- 102000003974 Fibroblast growth factor 2 Human genes 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 102100034051 Heat shock protein HSP 90-alpha Human genes 0.000 description 1
- 101710154606 Hemagglutinin Proteins 0.000 description 1
- 206010019851 Hepatotoxicity Diseases 0.000 description 1
- 102000006479 Heterogeneous-Nuclear Ribonucleoproteins Human genes 0.000 description 1
- 108010019372 Heterogeneous-Nuclear Ribonucleoproteins Proteins 0.000 description 1
- 102100033636 Histone H3.2 Human genes 0.000 description 1
- 101000907904 Homo sapiens Endoribonuclease Dicer Proteins 0.000 description 1
- 101001016865 Homo sapiens Heat shock protein HSP 90-alpha Proteins 0.000 description 1
- 101000611943 Homo sapiens Programmed cell death protein 4 Proteins 0.000 description 1
- 101000587434 Homo sapiens Serine/arginine-rich splicing factor 3 Proteins 0.000 description 1
- 101000666340 Homo sapiens Tenascin Proteins 0.000 description 1
- 101000626125 Homo sapiens Tetranectin Proteins 0.000 description 1
- UGQMRVRMYYASKQ-UHFFFAOYSA-N Hypoxanthine nucleoside Natural products OC1C(O)C(CO)OC1N1C(NC=NC2=O)=C2N=C1 UGQMRVRMYYASKQ-UHFFFAOYSA-N 0.000 description 1
- XQFRJNBWHJMXHO-RRKCRQDMSA-N IDUR Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(I)=C1 XQFRJNBWHJMXHO-RRKCRQDMSA-N 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 102000012334 Integrin beta4 Human genes 0.000 description 1
- 108010022238 Integrin beta4 Proteins 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- 108010063045 Lactoferrin Proteins 0.000 description 1
- 102100032241 Lactotransferrin Human genes 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 108060001084 Luciferase Proteins 0.000 description 1
- 239000005089 Luciferase Substances 0.000 description 1
- 241000023320 Luma <angiosperm> Species 0.000 description 1
- 102000018697 Membrane Proteins Human genes 0.000 description 1
- 108060004795 Methyltransferase Proteins 0.000 description 1
- 108700011259 MicroRNAs Proteins 0.000 description 1
- 108091092878 Microsatellite Proteins 0.000 description 1
- 241000699660 Mus musculus Species 0.000 description 1
- 206010028470 Mycoplasma infections Diseases 0.000 description 1
- MRWXACSTFXYYMV-UHFFFAOYSA-N Nebularine Natural products OC1C(O)C(CO)OC1N1C2=NC=NC=C2N=C1 MRWXACSTFXYYMV-UHFFFAOYSA-N 0.000 description 1
- 238000011887 Necropsy Methods 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- 102000006570 Non-Histone Chromosomal Proteins Human genes 0.000 description 1
- 108010008964 Non-Histone Chromosomal Proteins Proteins 0.000 description 1
- 108091092724 Noncoding DNA Proteins 0.000 description 1
- 102000007999 Nuclear Proteins Human genes 0.000 description 1
- 108010089610 Nuclear Proteins Proteins 0.000 description 1
- 102000007399 Nuclear hormone receptor Human genes 0.000 description 1
- 108020005497 Nuclear hormone receptor Proteins 0.000 description 1
- 101710093908 Outer capsid protein VP4 Proteins 0.000 description 1
- 101710135467 Outer capsid protein sigma-1 Proteins 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- 101150102573 PCR1 gene Proteins 0.000 description 1
- 208000005228 Pericardial Effusion Diseases 0.000 description 1
- 108090001050 Phosphoric Diester Hydrolases Proteins 0.000 description 1
- 102000004861 Phosphoric Diester Hydrolases Human genes 0.000 description 1
- 241000590419 Polygonia interrogationis Species 0.000 description 1
- 102100040992 Programmed cell death protein 4 Human genes 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- 102100038280 Prostaglandin G/H synthase 2 Human genes 0.000 description 1
- 108050003267 Prostaglandin G/H synthase 2 Proteins 0.000 description 1
- 102000007327 Protamines Human genes 0.000 description 1
- 108010007568 Protamines Proteins 0.000 description 1
- 229940124158 Protease/peptidase inhibitor Drugs 0.000 description 1
- 101710176177 Protein A56 Proteins 0.000 description 1
- 108010001267 Protein Subunits Proteins 0.000 description 1
- 102000002067 Protein Subunits Human genes 0.000 description 1
- 102100024924 Protein kinase C alpha type Human genes 0.000 description 1
- 101710109947 Protein kinase C alpha type Proteins 0.000 description 1
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 description 1
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 description 1
- 229930185560 Pseudouridine Natural products 0.000 description 1
- PTJWIQPHWPFNBW-UHFFFAOYSA-N Pseudouridine C Natural products OC1C(O)C(CO)OC1C1=CNC(=O)NC1=O PTJWIQPHWPFNBW-UHFFFAOYSA-N 0.000 description 1
- 102000013009 Pyruvate Kinase Human genes 0.000 description 1
- 108020005115 Pyruvate Kinase Proteins 0.000 description 1
- 108020005067 RNA Splice Sites Proteins 0.000 description 1
- 238000002123 RNA extraction Methods 0.000 description 1
- 239000013614 RNA sample Substances 0.000 description 1
- 102000000574 RNA-Induced Silencing Complex Human genes 0.000 description 1
- 108010016790 RNA-Induced Silencing Complex Proteins 0.000 description 1
- 230000004570 RNA-binding Effects 0.000 description 1
- 101710100969 Receptor tyrosine-protein kinase erbB-3 Proteins 0.000 description 1
- 101710100963 Receptor tyrosine-protein kinase erbB-4 Proteins 0.000 description 1
- 108020004511 Recombinant DNA Proteins 0.000 description 1
- 108010083644 Ribonucleases Proteins 0.000 description 1
- 102000006382 Ribonucleases Human genes 0.000 description 1
- 108091006627 SLC12A9 Proteins 0.000 description 1
- 108091058545 Secretory proteins Proteins 0.000 description 1
- 102000040739 Secretory proteins Human genes 0.000 description 1
- 241000710961 Semliki Forest virus Species 0.000 description 1
- 102100029665 Serine/arginine-rich splicing factor 3 Human genes 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 108091081024 Start codon Proteins 0.000 description 1
- 229930182558 Sterol Natural products 0.000 description 1
- 101710192266 Tegument protein VP22 Proteins 0.000 description 1
- RYYWUUFWQRZTIU-UHFFFAOYSA-N Thiophosphoric acid Chemical class OP(O)(S)=O RYYWUUFWQRZTIU-UHFFFAOYSA-N 0.000 description 1
- 108010022394 Threonine synthase Proteins 0.000 description 1
- 102000005497 Thymidylate Synthase Human genes 0.000 description 1
- 238000008050 Total Bilirubin Reagent Methods 0.000 description 1
- 108010018242 Transcription Factor AP-1 Proteins 0.000 description 1
- 102100023132 Transcription factor Jun Human genes 0.000 description 1
- 102000002015 Transforming Protein 1 Src Homology 2 Domain-Containing Human genes 0.000 description 1
- 108010040625 Transforming Protein 1 Src Homology 2 Domain-Containing Proteins 0.000 description 1
- 108700019146 Transgenes Proteins 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 229920004890 Triton X-100 Polymers 0.000 description 1
- 239000013504 Triton X-100 Substances 0.000 description 1
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 1
- 108010040002 Tumor Suppressor Proteins Proteins 0.000 description 1
- 102000001742 Tumor Suppressor Proteins Human genes 0.000 description 1
- 206010054094 Tumour necrosis Diseases 0.000 description 1
- 241000700618 Vaccinia virus Species 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- BFPAJRYMGFFFLA-UHFFFAOYSA-N [N+](=O)([O-])C1=CNC=C1.[N+](=O)([O-])C=1C=C2C=CNC2=CC1 Chemical compound [N+](=O)([O-])C1=CNC=C1.[N+](=O)([O-])C=1C=C2C=CNC2=CC1 BFPAJRYMGFFFLA-UHFFFAOYSA-N 0.000 description 1
- 239000003070 absorption delaying agent Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 238000011374 additional therapy Methods 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- JAZBEHYOTPTENJ-JLNKQSITSA-N all-cis-5,8,11,14,17-icosapentaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O JAZBEHYOTPTENJ-JLNKQSITSA-N 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- WLDHEUZGFKACJH-UHFFFAOYSA-K amaranth Chemical compound [Na+].[Na+].[Na+].C12=CC=C(S([O-])(=O)=O)C=C2C=C(S([O-])(=O)=O)C(O)=C1N=NC1=CC=C(S([O-])(=O)=O)C2=CC=CC=C12 WLDHEUZGFKACJH-UHFFFAOYSA-K 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000002583 anti-histone Effects 0.000 description 1
- 230000000840 anti-viral effect Effects 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 229940045985 antineoplastic platinum compound Drugs 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 230000001640 apoptogenic effect Effects 0.000 description 1
- 239000008365 aqueous carrier Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 210000004507 artificial chromosome Anatomy 0.000 description 1
- 210000003567 ascitic fluid Anatomy 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 230000002238 attenuated effect Effects 0.000 description 1
- 230000035578 autophosphorylation Effects 0.000 description 1
- VSRXQHXAPYXROS-UHFFFAOYSA-N azanide;cyclobutane-1,1-dicarboxylic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OC(=O)C1(C(O)=O)CCC1 VSRXQHXAPYXROS-UHFFFAOYSA-N 0.000 description 1
- 210000003651 basophil Anatomy 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 229960004217 benzyl alcohol Drugs 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- WGDUUQDYDIIBKT-UHFFFAOYSA-N beta-Pseudouridine Natural products OC1OC(CN2C=CC(=O)NC2=O)C(O)C1O WGDUUQDYDIIBKT-UHFFFAOYSA-N 0.000 description 1
- WHGYBXFWUBPSRW-FOUAGVGXSA-N beta-cyclodextrin Chemical compound OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO WHGYBXFWUBPSRW-FOUAGVGXSA-N 0.000 description 1
- 235000011175 beta-cyclodextrine Nutrition 0.000 description 1
- 229960004853 betadex Drugs 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 239000012472 biological sample Substances 0.000 description 1
- 229960000074 biopharmaceutical Drugs 0.000 description 1
- 238000001574 biopsy Methods 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 229910021538 borax Inorganic materials 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- 210000005013 brain tissue Anatomy 0.000 description 1
- 201000008275 breast carcinoma Diseases 0.000 description 1
- 238000000339 bright-field microscopy Methods 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 210000004900 c-terminal fragment Anatomy 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 239000003560 cancer drug Substances 0.000 description 1
- 230000036952 cancer formation Effects 0.000 description 1
- 239000002041 carbon nanotube Substances 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 231100000504 carcinogenesis Toxicity 0.000 description 1
- 230000022131 cell cycle Effects 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- 230000005754 cellular signaling Effects 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 210000001175 cerebrospinal fluid Anatomy 0.000 description 1
- 238000001311 chemical methods and process Methods 0.000 description 1
- 230000000973 chemotherapeutic effect Effects 0.000 description 1
- 238000000546 chi-square test Methods 0.000 description 1
- 229940045110 chitosan Drugs 0.000 description 1
- 210000002314 coated vesicle Anatomy 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 229940109239 creatinine Drugs 0.000 description 1
- 210000004748 cultured cell Anatomy 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 229940109262 curcumin Drugs 0.000 description 1
- 235000012754 curcumin Nutrition 0.000 description 1
- 239000004148 curcumin Substances 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- 210000000805 cytoplasm Anatomy 0.000 description 1
- 231100000135 cytotoxicity Toxicity 0.000 description 1
- 230000003013 cytotoxicity Effects 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- 238000004925 denaturation Methods 0.000 description 1
- 230000036425 denaturation Effects 0.000 description 1
- 230000001687 destabilization Effects 0.000 description 1
- 230000000368 destabilizing effect Effects 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- VFLDPWHFBUODDF-UHFFFAOYSA-N diferuloylmethane Natural products C1=C(O)C(OC)=CC(C=CC(=O)CC(=O)C=CC=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-UHFFFAOYSA-N 0.000 description 1
- ZPTBLXKRQACLCR-XVFCMESISA-N dihydrouridine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)CC1 ZPTBLXKRQACLCR-XVFCMESISA-N 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 239000002612 dispersion medium Substances 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 229940090949 docosahexaenoic acid Drugs 0.000 description 1
- 229940000406 drug candidate Drugs 0.000 description 1
- 241001493065 dsRNA viruses Species 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 230000002500 effect on skin Effects 0.000 description 1
- 229960005135 eicosapentaenoic acid Drugs 0.000 description 1
- JAZBEHYOTPTENJ-UHFFFAOYSA-N eicosapentaenoic acid Natural products CCC=CCC=CCC=CCC=CCC=CCCCC(O)=O JAZBEHYOTPTENJ-UHFFFAOYSA-N 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 230000002357 endometrial effect Effects 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 210000003979 eosinophil Anatomy 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 230000003628 erosive effect Effects 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 210000003527 eukaryotic cell Anatomy 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000011124 ex vivo culture Methods 0.000 description 1
- 239000013604 expression vector Substances 0.000 description 1
- 239000012894 fetal calf serum Substances 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 238000000799 fluorescence microscopy Methods 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 229920001109 fluorescent polymer Polymers 0.000 description 1
- 230000037433 frameshift Effects 0.000 description 1
- 150000002243 furanoses Chemical class 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- 230000000799 fusogenic effect Effects 0.000 description 1
- 238000001502 gel electrophoresis Methods 0.000 description 1
- 229940014259 gelatin Drugs 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 238000011331 genomic analysis Methods 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 239000003481 heat shock protein 90 inhibitor Substances 0.000 description 1
- 230000003067 hemagglutinative effect Effects 0.000 description 1
- 239000000185 hemagglutinin Substances 0.000 description 1
- 238000005534 hematocrit Methods 0.000 description 1
- 230000007686 hepatotoxicity Effects 0.000 description 1
- 231100000304 hepatotoxicity Toxicity 0.000 description 1
- 108010084091 heregulin beta1 Proteins 0.000 description 1
- FBPFZTCFMRRESA-UHFFFAOYSA-N hexane-1,2,3,4,5,6-hexol Chemical compound OCC(O)C(O)C(O)C(O)CO FBPFZTCFMRRESA-UHFFFAOYSA-N 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 230000002962 histologic effect Effects 0.000 description 1
- 238000010562 histological examination Methods 0.000 description 1
- 238000000265 homogenisation Methods 0.000 description 1
- 102000051308 human DICER1 Human genes 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 210000002865 immune cell Anatomy 0.000 description 1
- 230000008105 immune reaction Effects 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 230000003308 immunostimulating effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000000411 inducer Substances 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000015788 innate immune response Effects 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 238000000185 intracerebroventricular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000007919 intrasynovial administration Methods 0.000 description 1
- 208000030776 invasive breast carcinoma Diseases 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 239000007951 isotonicity adjuster Substances 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- CSSYQJWUGATIHM-IKGCZBKSSA-N l-phenylalanyl-l-lysyl-l-cysteinyl-l-arginyl-l-arginyl-l-tryptophyl-l-glutaminyl-l-tryptophyl-l-arginyl-l-methionyl-l-lysyl-l-lysyl-l-leucylglycyl-l-alanyl-l-prolyl-l-seryl-l-isoleucyl-l-threonyl-l-cysteinyl-l-valyl-l-arginyl-l-arginyl-l-alanyl-l-phenylal Chemical compound C([C@H](N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(O)=O)C1=CC=CC=C1 CSSYQJWUGATIHM-IKGCZBKSSA-N 0.000 description 1
- 229940078795 lactoferrin Drugs 0.000 description 1
- 235000021242 lactoferrin Nutrition 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 108020001756 ligand binding domains Proteins 0.000 description 1
- 238000012417 linear regression Methods 0.000 description 1
- 210000002751 lymph Anatomy 0.000 description 1
- 230000002132 lysosomal effect Effects 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 210000002540 macrophage Anatomy 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 210000005075 mammary gland Anatomy 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 238000001906 matrix-assisted laser desorption--ionisation mass spectrometry Methods 0.000 description 1
- 229960002985 medroxyprogesterone acetate Drugs 0.000 description 1
- PSGAAPLEWMOORI-PEINSRQWSA-N medroxyprogesterone acetate Chemical compound C([C@@]12C)CC(=O)C=C1[C@@H](C)C[C@@H]1[C@@H]2CC[C@]2(C)[C@@](OC(C)=O)(C(C)=O)CC[C@H]21 PSGAAPLEWMOORI-PEINSRQWSA-N 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 230000009245 menopause Effects 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- OSWPMRLSEDHDFF-UHFFFAOYSA-N methyl salicylate Chemical compound COC(=O)C1=CC=CC=C1O OSWPMRLSEDHDFF-UHFFFAOYSA-N 0.000 description 1
- 230000011987 methylation Effects 0.000 description 1
- 238000007069 methylation reaction Methods 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- YACKEPLHDIMKIO-UHFFFAOYSA-N methylphosphonic acid Chemical class CP(O)(O)=O YACKEPLHDIMKIO-UHFFFAOYSA-N 0.000 description 1
- 108091027943 miR-16 stem-loop Proteins 0.000 description 1
- 108091062762 miR-21 stem-loop Proteins 0.000 description 1
- 108091041631 miR-21-1 stem-loop Proteins 0.000 description 1
- 108091044442 miR-21-2 stem-loop Proteins 0.000 description 1
- 108091070501 miRNA Proteins 0.000 description 1
- 239000000693 micelle Substances 0.000 description 1
- 230000011278 mitosis Effects 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000007479 molecular analysis Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 230000003562 morphometric effect Effects 0.000 description 1
- 238000013425 morphometry Methods 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 229920005615 natural polymer Polymers 0.000 description 1
- MRWXACSTFXYYMV-FDDDBJFASA-N nebularine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C2=NC=NC=C2N=C1 MRWXACSTFXYYMV-FDDDBJFASA-N 0.000 description 1
- 238000011227 neoadjuvant chemotherapy Methods 0.000 description 1
- 210000005170 neoplastic cell Anatomy 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 108091008104 nucleic acid aptamers Proteins 0.000 description 1
- 125000003835 nucleoside group Chemical group 0.000 description 1
- 238000006384 oligomerization reaction Methods 0.000 description 1
- 230000005959 oncogenic signaling Effects 0.000 description 1
- 239000003002 pH adjusting agent Substances 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 231100000915 pathological change Toxicity 0.000 description 1
- 230000036285 pathological change Effects 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000007170 pathology Effects 0.000 description 1
- 108010043655 penetratin Proteins 0.000 description 1
- MCYTYTUNNNZWOK-LCLOTLQISA-N penetratin Chemical compound C([C@H](NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)CCCNC(N)=N)[C@@H](C)CC)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(N)=O)C1=CC=CC=C1 MCYTYTUNNNZWOK-LCLOTLQISA-N 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 150000002972 pentoses Chemical class 0.000 description 1
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 1
- 230000010412 perfusion Effects 0.000 description 1
- 210000004912 pericardial fluid Anatomy 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical group CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 1
- 150000004713 phosphodiesters Chemical class 0.000 description 1
- 150000008298 phosphoramidates Chemical class 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 1
- 238000000053 physical method Methods 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 239000013600 plasmid vector Substances 0.000 description 1
- 150000003058 platinum compounds Chemical class 0.000 description 1
- 229920000333 poly(propyleneimine) Polymers 0.000 description 1
- 230000008488 polyadenylation Effects 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 102000054765 polymorphisms of proteins Human genes 0.000 description 1
- 238000010837 poor prognosis Methods 0.000 description 1
- 230000034190 positive regulation of NF-kappaB transcription factor activity Effects 0.000 description 1
- 230000029279 positive regulation of transcription, DNA-dependent Effects 0.000 description 1
- 230000009819 post translational phosphorylation Effects 0.000 description 1
- 230000004481 post-translational protein modification Effects 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 238000002203 pretreatment Methods 0.000 description 1
- 230000001480 pro-metastatic effect Effects 0.000 description 1
- 239000000186 progesterone Substances 0.000 description 1
- 229960003387 progesterone Drugs 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 208000023958 prostate neoplasm Diseases 0.000 description 1
- 210000001938 protoplast Anatomy 0.000 description 1
- PTJWIQPHWPFNBW-GBNDHIKLSA-N pseudouridine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1C1=CNC(=O)NC1=O PTJWIQPHWPFNBW-GBNDHIKLSA-N 0.000 description 1
- 230000002685 pulmonary effect Effects 0.000 description 1
- 150000003212 purines Chemical class 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 108700022487 rRNA Genes Proteins 0.000 description 1
- 230000002285 radioactive effect Effects 0.000 description 1
- 239000012132 radioimmunoprecipitation assay buffer Substances 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 102000027426 receptor tyrosine kinases Human genes 0.000 description 1
- 108091008598 receptor tyrosine kinases Proteins 0.000 description 1
- 238000010188 recombinant method Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000002040 relaxant effect Effects 0.000 description 1
- 210000005084 renal tissue Anatomy 0.000 description 1
- 230000001177 retroviral effect Effects 0.000 description 1
- 238000003757 reverse transcription PCR Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 238000010079 rubber tapping Methods 0.000 description 1
- 210000003296 saliva Anatomy 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 230000011218 segmentation Effects 0.000 description 1
- 239000012679 serum free medium Substances 0.000 description 1
- 230000001743 silencing effect Effects 0.000 description 1
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 1
- 229910010271 silicon carbide Inorganic materials 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 239000004055 small Interfering RNA Substances 0.000 description 1
- 229940126586 small molecule drug Drugs 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 235000011083 sodium citrates Nutrition 0.000 description 1
- 239000000176 sodium gluconate Substances 0.000 description 1
- 235000012207 sodium gluconate Nutrition 0.000 description 1
- 229940005574 sodium gluconate Drugs 0.000 description 1
- 239000001540 sodium lactate Substances 0.000 description 1
- 229940005581 sodium lactate Drugs 0.000 description 1
- 235000011088 sodium lactate Nutrition 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 235000011008 sodium phosphates Nutrition 0.000 description 1
- 235000010339 sodium tetraborate Nutrition 0.000 description 1
- 210000004872 soft tissue Anatomy 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000002047 solid lipid nanoparticle Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000000527 sonication Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 238000011255 standard chemotherapy Methods 0.000 description 1
- 238000007619 statistical method Methods 0.000 description 1
- 102000005969 steroid hormone receptors Human genes 0.000 description 1
- 108020003113 steroid hormone receptors Proteins 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 150000003432 sterols Chemical class 0.000 description 1
- 235000003702 sterols Nutrition 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 210000002536 stromal cell Anatomy 0.000 description 1
- YCUVUDODLRLVIC-VPHDGDOJSA-N sudan black b Chemical compound C1=CC(=C23)NC(C)(C)NC2=CC=CC3=C1\N=N\C(C1=CC=CC=C11)=CC=C1\N=N\C1=CC=CC=C1 YCUVUDODLRLVIC-VPHDGDOJSA-N 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- DKPFODGZWDEEBT-QFIAKTPHSA-N taxane Chemical class C([C@]1(C)CCC[C@@H](C)[C@H]1C1)C[C@H]2[C@H](C)CC[C@@H]1C2(C)C DKPFODGZWDEEBT-QFIAKTPHSA-N 0.000 description 1
- 231100001274 therapeutic index Toxicity 0.000 description 1
- 230000004797 therapeutic response Effects 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- 210000000779 thoracic wall Anatomy 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 229940043263 traditional drug Drugs 0.000 description 1
- 108010062760 transportan Proteins 0.000 description 1
- PBKWZFANFUTEPS-CWUSWOHSSA-N transportan Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(C)C)C(N)=O)[C@@H](C)CC)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)CN)[C@@H](C)O)C1=CC=C(O)C=C1 PBKWZFANFUTEPS-CWUSWOHSSA-N 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 239000001226 triphosphate Substances 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- BSVBQGMMJUBVOD-UHFFFAOYSA-N trisodium borate Chemical compound [Na+].[Na+].[Na+].[O-]B([O-])[O-] BSVBQGMMJUBVOD-UHFFFAOYSA-N 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 231100000588 tumorigenic Toxicity 0.000 description 1
- 230000000381 tumorigenic effect Effects 0.000 description 1
- 238000007492 two-way ANOVA Methods 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 229940121358 tyrosine kinase inhibitor Drugs 0.000 description 1
- 239000005483 tyrosine kinase inhibitor Substances 0.000 description 1
- 150000004917 tyrosine kinase inhibitor derivatives Chemical class 0.000 description 1
- 238000002604 ultrasonography Methods 0.000 description 1
- 241000712461 unidentified influenza virus Species 0.000 description 1
- 241001515965 unidentified phage Species 0.000 description 1
- 238000012762 unpaired Student’s t-test Methods 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 238000010200 validation analysis Methods 0.000 description 1
- 238000012795 verification Methods 0.000 description 1
- 239000013603 viral vector Substances 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 239000008215 water for injection Substances 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- AFVLVVWMAFSXCK-UHFFFAOYSA-N α-cyano-4-hydroxycinnamic acid Chemical compound OC(=O)C(C#N)=CC1=CC=C(O)C=C1 AFVLVVWMAFSXCK-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/71—Receptors; Cell surface antigens; Cell surface determinants for growth factors; for growth regulators
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1135—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against oncogenes or tumor suppressor genes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
- C12Q1/6886—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material for cancer
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering nucleic acids [NA]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/315—Phosphorothioates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/321—2'-O-R Modification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
- C12N2310/3515—Lipophilic moiety, e.g. cholesterol
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/30—Special therapeutic applications
- C12N2320/31—Combination therapy
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/30—Special therapeutic applications
- C12N2320/32—Special delivery means, e.g. tissue-specific
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/30—Special therapeutic applications
- C12N2320/34—Allele or polymorphism specific uses
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/156—Polymorphic or mutational markers
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/158—Expression markers
Definitions
- the present invention generally relates to the field of molecular biology and RNA interference (RNAi). More specifically, the methods of the present invention relates to specific double stranded RNA (dsRNA) molecules, referred to as small (or short) interfering RNAs (siRNAs), its composition and use thereof, as well as a method of treating cancer and methods of inhibiting cancer cell proliferation, particularly a method of treating breast cancer.
- dsRNA specific double stranded RNA
- siRNAs small (or short) interfering RNAs
- the method of the present invention is a method for inhibiting in vitro proliferation of triple negative breast cancer (TNBC) cells and for the blockade of in vivo grow of TN breast tumors.
- TNBC triple negative breast cancer
- the invention provides specific siRNA molecules, comprising nucleotide sequences as depicted in SEQ ID NO: 1 and SEQ ID NO: 2.
- siRNA molecules are suitable for the treatment of breast cancer, particularly, TNBC.
- Aspects of the invention also relate to methods for detecting a diagnostic marker in a sample of a subject suspected of having a TNBC tumor, the methods comprising providing a diagnostic sample of the subject, contacting the diagnostic sample of the subject with the siRNA molecules of the invention, and determining the presence of a diagnostic marker in said sample.
- Double stranded RNA has the capacity to inhibit protein expression, which represents an extraordinary therapy for a large number of human diseases.
- therapeutic dsRNA are emerging as a new class of drugs.
- RNA interference RNA interference
- mRNA homologous host messenger RNA
- siRNAs small interfering RNAs
- siRNA-based drug can target any mRNAs of interest, regardless of the cellular location of the translated proteins; iii) the sequence-specific basis of RNAi allows to target any mRNA and to discriminate against different alleles and alternatively spliced isoforms of protein-coding mRNAs ( Goldberg, M.S. et al. (2012) Pyruvate kinase M2-specific siRNA induces apoptosis and tumor regression.
- siRNAs are especially advantageous for therapeutic applications in heterogeneous diseases that lack common molecular signatures.
- triple-negative breast cancer refers to tumors that do not express clinically significant levels of the steroid hormone receptors estrogen (ER) and progesterone (PR), and lack overexpression at the cytoplasmic membrane of ErbB-2/HER2 (MErbB-2), a member of the ErbBs family of receptor tyrosine kinases (EGFR/ErbB-1, ErbB-2, ErbB-3, ErbB-4) or ERBB2 gene amplification.
- TNBC triple-negative breast cancer
- ER steroid hormone receptors
- PR progesterone
- EGFR/ErbB-1, ErbB-2, ErbB-3, ErbB-4 ERBB2 gene amplification.
- GE profile studies in TNBC cohorts revealed that this subtype is indeed a heterogeneous group and identified six different GE profile clusters: basal-like 1 (BL1), basal-like 2 (BL2), immunomodulatory (IM), mesenchymal (M), mesenchymal stem-like (MSL) and luminal androgen receptor (LAR) ( Lehmann, B.D. et al. (2011) Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. JClinInvest 121, 2750-2767 ).
- BL1 basal-like 1
- BL2 basal-like 2
- IM immunomodulatory
- M mesenchymal
- MSL mesenchymal stem-like
- LAR luminal androgen receptor
- TNBC-4type BL1, BL2, M and LAR
- PloS one 11, e0157368 ; Prat, A. et al. (2013a) Molecular characterization of basal-like and non-basal-like triple-negative breast cancer. The oncologist 18, 123-133 ).
- TNBC transcriptomes identified four subtypes: LAR, M, basal-like immune suppressed (BLIS), and BL immune activated (BLIA) ( Burstein, M.D. et al. (2015) Comprehensive genomic analysis identifies novel subtypes and targets of triple-negative breast cancer. ClinCancer Res. 21, 1688-1698 ). Correlation of the transcriptional profiles of TN tumors to those of the intrinsic BC molecular subtypes (luminal A (LumA), luminal B (LumB), basal-like (BL), and ErbB-2-enriched (ErbB-2E)) ( Perou, C.M. et al. (2000) Molecular portraits of human breast tumours. Nature 406, 747-752 ; Prat, A.
- TNBCs segregated into the ErbB-2E subtype which is defined by MErbB-2 overexpression and/or ERBB2 gene amplification (Prat, A. et al. (2013a) op. cit.).
- TNBCs that segregated into the ErbB-2E subtype showed only five genes significantly downregulated as compared to ErbB-2E tumors, including ErbB-2 (Prat, A. et al. (2013a) op. cit.).
- MErbB-2 role as a potent inducer of BC growth and metastasis is well acknowledged ( Henderson, I.C. et al. (1998) The relationship between prognostic and predictive factors in the management of breast cancer. Breast Cancer ResTreat 52, 261-288 ; Ross, J.S. et al. (2009) The HER-2 receptor and breast cancer: ten years of targeted anti-HER-2 therapy and personalized medicine. Oncologist 14, 320-368 ; Slamon, D.J. et al. (1989) Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 244, 707-712 ).
- MErbB-2 migrates to the nucleus (NErbB-2) of BC cells where it binds promoters/enhancers of target genes and functions as a transcription factor (TF) or as a coactivator to promote BC growth, metastasis and resistance to anti-MErbB-2 therapies ( Beguelin, W. et al. (2010) Progesterone receptor induces ErbB-2 nuclear translocation to promote breast cancer growth via a novel transcriptional effect: ErbB-2 function as a coactivator of Stat3. MolCell Biol 30, 5456-5472 ; Cordo Russo, R.I. et al.
- ErbB-2 refers to the tyrosine kinase receptor ErbB-2 that belongs to the epidermal growth factor receptor family. ErbB-2 can be natural or synthetic (e.g., derived from PCR and/or recombinant DNA techniques). ErbB-2 can be from a mammal, such as a human. An exemplary wild-type ErbB-2 nucleic acid sequence is NCBI GenBank Accession No. NG_007503.1 (SEQ ID NO: 3).
- WT wild-type
- p165ErbB-2 an ErbB-2 isoform having a molecular weight (MW) of 165 kDa on sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) (herein also referred to as p165ErbB-2) in cell lines from the four TNBC subtypes identified at present.
- WT wild-type
- p165ErbB-2 sodium dodecyl sulfate polyacrylamide gel electrophoresis
- MDA-MB-453 cells luminal androgen receptor, LAR subtype express wild-type (WT) ErbB-2 (MW of 185 kDa, also referred to as p185ErbB-2).
- WT wild-type
- MDA-MB-468 cells (basal-like 1, BL1 subtype) express only p165ErbB-2.
- HCC-70 basal-like 2, BL2 subtype
- MDA-MB-231 cells (mesenchymal, M subtype) express both p185ErbB-2 (WTErbB-2) and p165ErbB-2.
- ErbB-2 protein variants are placed in a new and unanticipated scenario: the nucleus of TNBC.
- PCR polymerase chain reaction
- RNAi RNA interference
- NP_001276865 encoded by the ErbB-2 alternative transcript 3
- SpT3, NM_001289936 is the contributor to the low MW variant p165ErbB-2. This is the first report of the expression of the non-canonical ErbB-2 isoform c in any normal or malignant cell type.
- the present inventors have designed novel and specific siRNA molecules that target the abovementioned alternative transcript variant encoding isoform c of ErbB-2 and propose the use of said specific siRNAs for the treatment of breast cancer, in particular, triple negative breast cancer.
- the present invention provides a new alternative for treating cancer and methods of inhibiting cancer cell proliferation, more particularly, for the treatment of triple negative breast cancer, through specific siRNAs directed against the ErbB-2 isoform c.
- kits for gene silencing wherein said kit is comprised of at least two siRNA duplexes, wherein one strand of each duplex comprises at least eighteen contiguous bases that are complementary to a region of one target messenger RNA (mRNA).
- mRNA target messenger RNA
- the present invention provides specific siRNA molecules targeting ErbB-2 alternative transcript variant 3 (SpT3) which encodes the non-canonical ErbB-2 isoform c, including, but not restricted to those having the sequences depicted in SEQ ID NO: 1 and SEQ ID NO: 2.
- SpT3 ErbB-2 alternative transcript variant 3
- the present invention provides a specific siRNA molecule targeted to ErbB-2 alternative transcript variant 3 (SpT3, NM_001289936, SEQ ID NO: 32), which encodes the non-canonical isoform c (NP_001276865) of ErbB-2 having a MW of -165 kDa on SDS-PAGE, said siRNA molecule comprising a sequence as depicted in SEQ ID NO: 1 or in SEQ ID NO: 2 or any other sequence having a sequence identity greater than 90% between the siRNA and the portion of the target gene.
- the aforesaid siRNA molecule is composed by a duplex region and displays either no overhangs or at least one overhang, wherein each overhang has up to six or fewer nucleotides, wherein the duplex region comprises a sense strand and an antisense strand, wherein said sense strand and said antisense strand together form said duplex region and said duplex region is 18-30 base pairs in length and said antisense strand comprises a sequence that is complementary to the target RNA, SEQ ID NO: 32.
- the sense strand and the antisense strand each comprise one or more chemically-modified nucleotides; or the sense strand and the antisense strand each consists of chemically-modified nucleotides.
- a second object of the present invention is to provide a pharmaceutical composition comprising a siRNA molecule as disclosed herein, in particular an effective amount thereof, together with a pharmaceutically acceptable excipient or carrier.
- a pharmaceutical composition comprising an effective amount of a siRNA molecule as disclosed herein, in particular an effective amount thereof, further comprising an additional anti-cancer agent, and a pharmaceutically acceptable excipient or carrier.
- the present invention also provides the use of the siRNAs disclosed herein for the manufacture of a medicament for the treatment of breast cancer.
- the breast cancer is triple negative breast cancer.
- a method for the treatment of triple negative breast cancer comprising administration of one or more of the siRNA molecules according to the invention, or a composition comprising said siRNAs according to the invention and a pharmaceutically acceptable excipient or carrier, or a composition comprising said siRNAs, an additional anti-cancer agent, and a pharmaceutically acceptable excipient or carrier.
- SpT3 siRNA #2 SEQ ID NO: 2 is used.
- the present invention concerns a siRNA molecule according to the invention for use in a method for the treatment of triple negative breast cancer, comprising the administration of one or more of said siRNA,
- the present invention concerns a composition according to the present invention for use in a method for the treatment of triple negative breast cancer, comprising the administration of said composition.
- SpT3 siRNA #2 (SEQ ID NO: 2) is used.
- SpT3 siRNA#1 (SEQ ID NO: 1) is used.
- Another aspect of the present invention involves the administration of a pool of the two siRNAS designed by the inventors: SEQ ID NO: 1 and SEQ ID NO: 2 to increase the gene silencing activity.
- SEQ ID NO: 1 and SEQ ID NO: 2 may be used either alone or as a pool.
- kits for detecting a diagnostic marker in a sample of a subject suspected of having a TNBC tumor comprising providing a diagnostic sample of the subject, contacting the diagnostic sample of the subject with the siRNA molecules of the invention, and determining the presence of a diagnostic marker in said sample.
- the diagnostic marker is alternative transcript variant 3 (SpT3), which encodes isoform c of ErbB-2, having a MW of 165 kDa on SDS-PAGE (p165ErbB-2).
- said method is based on the ability of a siRNA molecular beacon (MB) to recognize a unique mRNA sequence at the 5' UTR and 5' coding region of SpT3, the transcript variant encoding ErbB-2 isoform c which displays a MW of 165 kDa (p165ErbB-2).
- MB siRNA molecular beacon
- the siRNA molecules of the invention having the sequences depicted in SEQ ID NO: 1 or SEQ ID NO: 2 are targeted to this unique region.
- SpT3 MB siRNA molecular beacon
- Yet another object of the present invention is directed to a binary molecular beacon consisting of SpT3 MB displaying SpT3 siRNA #1 of SEQ ID NO: 1 and SpT3 MB displaying SpT3 siRNA #2 of SEQ ID NO: 2.
- MBs may include also other specific siRNAs sequences targeting SpT3 as explained above.
- the method for detecting a diagnostic marker involves the use of a highly sensitive, branched fluorescence in situ hybridization (FISH) technology named RNAscope ® ( Wang, F. et al. (2012) RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissues. JMD 14, 22-29 ) for determining the presence of alternative ErbB-2 transcripts in breast cancer, in particular TNBC.
- FISH fluorescence in situ hybridization
- the target probes of the RNAscope ® assays are designed to specifically recognize a unique mRNA sequence at the 5' UTR and 5' coding region of SpT3, the transcript variant encoding ErbB-2 isoform c which displays a MW of 165 kDa (p165ErbB-2).
- RNAscope ® is well described into the following issued patents, in: U.S. Pat. Nos. 7,709,198 ; 8,604,182 ; 8,658,361 ; 8,951,726 ; 7,709,198 ; 8,658,361 ; and 7,709,198 .
- a kit for detection of ErbB-2 alternative transcript variant 3 comprising agents for conducting an RNA ISH assay including, but not restricted to, one or more target probe sets having the sequences depicted in SEQ ID NO: 1 and SEQ ID NO: 2, for detection of ErbB-2 alternative transcript variant 3 (SpT3) diagnostic marker.
- the aforementioned ISH assay is an RNAscope ® assay.”
- Further objects of the invention are a method for inhibiting in vitro proliferation of triple negative breast cancer (TNBC) cells, by using the aforesaid siRNA molecules; a method for the blockade of in vivo grow of TN breast tumors, comprising the administration to a patient of the aforesaid siRNA molecules.
- TNBC triple negative breast cancer
- FIGS 1A-1C NErbB-2 expression in TNBC patients.
- Low membrane ErbB-2 (MErbB-2) levels (1+ score) are not considered as membrane ErbB-2 overexpression in the clinical setting.
- Thick arrows indicate MErbB-2 and slim arrows, NErbB-2. Nuclei were stained with DAPI.
- NErbB-2 IF staining using the C-18 ErbB-2 polyclonal antibody (upper panel) or the ErbB-2 A-2 monoclonal antibody raised against amino acids 1180-1197 at the ErbB-2 C-terminus (lower panel).
- One representative tumor displaying 3+ NErbB-2 score is shown. Nuclei were stained with DAPI.
- (1C) NErbB-2 expression was detected with the C-18 antibody by IF, as described in Fig. 1A , or by immunohistochemistry (IHC). Shown are representative images of tumors displaying 3+ NErbB-2 score. Boxed areas in the IHC staining are shown in detail in the inset (400X).
- NErbB-2 shows clinical relevance in TNBC. Relationship between NErbB-2 positivity and survival in terms of overall survival (2A), disease-free survival (2B), local relapse-free survival (2C) and distant metastasis-free survival (2D) probabilities (%), as assessed by a Kaplan-Meier analysis and log-rank test.
- the TNBC clinical study is based on the monitoring of 99 patients, 38 therefrom being NErbB-2-positive (full line) and 61 NErbB-2-negative (dotted line).
- FIGS 3A-3I ErbB-2 isoforms expression and activation in BC cells.
- MDA-MB-453 cells (LAR subtype) expressed full-length ErbB-2 (185 kDa, p185ErbB-2, WTErbB-2) comparable to the canonical ErbB-2 present in BT-474 cells, used as a control.
- MDA-MB-468 (BL1 subtype) showed an ErbB-2 variant of lower MW (-165 kDa, p165ErbB-2).
- HCC-70 and MDA-MB-231 cells presented both p185ErbB-2 (WTErbB-2) and p165ErbB-2. Additionally, the ErbB-2 truncated isoforms p95ErbB-2/CTFs (-90-115 kDa) produced by proteolytic cleavage and alternative initiation of translation were observed.
- 3B Signal intensities of p185ErbB-2 (black bars) and p165ErbB-2 (grey bars) in four independent WBs performed as in Fig. 3A , were analyzed by densitometry and normalized to ⁇ -tubulin protein bands.
- FIGS 4A-4D ErbB-2 subcellular localization in BC cells.
- 4A ErbB-2 localization was studied by IF and confocal microscopy using the ErbB-2 C-18 antibody followed by incubation with an IgG-Alexa Fluor 488 secondary antibody. Thick arrows indicate the presence of MErbB-2 and slim ones show NErbB-2 presence in TNBC cell lines and in the control BT-474 cells upon heregulin (HRG) stimulation. Nuclei were stained with DAPI.
- 4B Percentage of nuclear ErbB-2 presence in confocal images from Fig. 4A .
- Integrated density (mean fluorescence intensity per unit area) of subcellular compartments was quantified in 50 cells from each cell line and was analyzed as percentages (mean ⁇ SD), relative to the total content (integrated density) of ErbB-2 in each cell, which was set to 100%.
- One-way ANOVA with Dunnett's multiple comparisons test was applied to determined significant differences between control and HRG- ⁇ 1-treated cells. For b vs a, and c vs a: P ⁇ 0.001.
- Figures 5A-5B N-glycosylation pattern of ErbB-2 in TNBC cells.
- 5A NetNGlyc 1.0 server (http://www.cbs.dtu.dk/services/NetNGlyc/) was used to predict N-glycosylation sites by analyzing the Asn-Xaa-Ser/Thr sequons defined as a pre-requisite for N-glycosylation ( Blom, N. et al. (2004) Prediction of post-translational glycosylation and phosphorylation of proteins from the amino acid sequence. Proteomics 4, 1633-1649 ).
- Each graph illustrates putative N-glycosylation sites across the protein chain of different ErbB-2 isoforms (https://www.ncbi.nlm.nih.gov/refseq/).
- X-axis represents protein length from N- to C-terminal.
- a position with a vertical grey line crossing the threshold (black horizontal line at 0.5) is predicted to be N-glycosylated.
- BT-474 and MDA-MB-468 cells were treated with tunicamycin (10 ⁇ g/ml) or DMSO as control for 15 h. ErbB-2 expression was then evaluated by WB with the C-18 antibody. MW calculation of protein bands was performed using the relative migration distance (Rf) method.
- FIGS 6A-6C Schematic representation of protein-coding ErbB-2 transcript variants annotated on RefSeq database (NCBI RefSeq, 2020 https://www.ncbi.nlm.nih.gov/refseq/).
- Consensus coding sequences (CCDSs) and untranslated regions (5' and 3' UTRs) are represented as lines and dashed lines, respectively. Numbers inside boxes and below lines indicate the sequence length in nucleotides (nt).
- the position of the translation start site (ATG) and the translation stop codon (TGA) are also shown.
- Variant 1 corresponds to the canonical sequence that encodes the ErbB-2 isoform a, p185ErbB-2 (WTErbB-2).
- Variant 2 differs in the 5' UTR, it lacks a portion of the 5' CCDS, initiating translation at a downstream start codon.
- the box indicating region from position 1 to position 558 nt shows the differential region of variant 2 compared to variant 1.
- Variant 3 (SpT3) has an alternate 5' UTR and 5' CCDS.
- the box indicating region from position 1 to position 607 nt indicates the differential region of variant 3 with respect to the canonical transcript.
- Variant 3 also presents a unique sequence of 51 nt (exon 5) spanning its 5' UTR and 5' CCDS (box indicating region from position 560 to position 610 nt).
- Variant 4 (SpT4) lacks the sequence that corresponds to exon 26 in the 3' CCDS of variant 1 (box indicating region from position 3421 to position 3673 nt) resulting in a translational frameshift. The box indicating region from position 3421 to position 4411 nt shows the differential 3' UTR and 3' CCDS region of variant 4.
- 6C Schematic representation of ErbB-2 ⁇ 16 spliced transcript compared with variant 1.
- FIG. 7 Schematic representation of ErbB-2 isoforms. The regions that are relevant to ErbB-2 function are depicted as grey boxes. Numbers below lines indicate the protein length in amino acids (aa). The extracellular portion is composed by 4 domains (I-IV): two leucine-rich (L) ligand-binding domains, I/LI and III/LII, and two cysteine-rich (C) domains, II/CI and IV/CII, which facilitate the formation of disulfide bonds. The position of the signal peptide (SP) in the amino-terminus is also shown. ErbB-2 contains a transmembrane domain (TM region) and a juxtamembrane domain, which bears the nuclear localization sequence (NLS).
- TM region transmembrane domain
- NLS nuclear localization sequence
- the intracellular portion includes the catalytic protein tyrosine kinase domain (PTKc domain) which contains the activation loop (A-loop), and a proline-rich transactivation domain near the carboxy-terminus.
- PTKc domain catalytic protein tyrosine kinase domain
- A-loop activation loop
- proline-rich transactivation domain near the carboxy-terminus.
- the major phosphorylation sites are indicated (grey dashed lines).
- FIG 8 Signal peptide prediction. Graphical output of SignalP 4.1 Server (http://www.cbs.dtu.dk/services/SignaIP/) for signal peptide (SP) prediction ( Nielsen, H. (2017) Predicting Secretory Proteins with SignaIP. Methods in molecular biology 1611, 59-73 ; Emanuelsson, O. et al. (2007) Locating proteins in the cell using TargetP, SignaIP and related tools. Nature protocols 2, 953-971 ).
- C-score raw cleavage site score, dark grey continuous line
- the C-score is trained to be high at the position immediately after the cleavage site (the first residue in the mature protein).
- S-score signal peptide score, light grey dashed line
- Y-score combined cleavage site score, dark grey dotted line
- Y-score combined cleavage site score, dark grey dotted line
- Figures 9A-9E Amplification of the entire coding region of ErbB-2 transcript variants by long range (LR)-PCR.
- LR-PCRs were performed with RANGER DNA polymerase using cDNA as template and two sets of primers: (9A, 9B, 9C) forward primer of SEQ ID NO: 4 and reverse primer of SEQ ID NO: 5, spanning the complete coding region of variants 1, 4 and ErbB-2 ⁇ 16 on one side (LR-PCR1), and on the other, (9D, 9E) forward primer of SEQ ID NO: 6 and reverse primer of SEQ ID NO: 5, spanning the complete coding region of variants 2 and 3 (LR-PCR2).
- Analysis of LR-PCR products on 0.7% agarose gels showed fragments ranging between ⁇ 4 to 5 kb long (indicated by arrows) corresponding to the size of expected products, which are indicated.
- FIGS 10A-10C Representative gel images for the nested PCR strategy.
- LR-PCR1 product was used as template for nested PCR with primers for SpT1
- LR-PCR2 product as template for nested PCR with primers for SpT2 and SpT3.
- 10A SpT1 expression was studied using primers spanning its differential region between exons 16 to 26 (forward primer of SEQ ID NO: 7 and reverse primer of SEQ ID NO: 8).
- Expression of SpT2 (10B) and SpT3 (10C) was assesed using variants specific primers spanning their differential region between exons 1 to 5.
- Primers for SpT2 are depicted as SEQ ID NOs: 9 and 10
- primers for SpT3 have SEQ ID NOs: 11 and 12.
- FIGS 11A-11B Representative gel images for the conventional PCR strategy. Expression of SpT4 (11A) and of ErbB-2 ⁇ 16 (11B) was studied by conventional PCR using spliced-specific primers: for SpT4 primers of SEQ ID NOs: 13 and 14; and primers for ErbB-2 ⁇ 16 have SEQ ID NOs: 15 and 16.
- Figures 12A-12D Representative gel images of the competitive PCR strategy.
- Amplification products corresponding to competitors (variants with exon 26 or 16 inclusion) and targets (SpT4 or ErbB-2 ⁇ 16, respectively) were quantified by densitometry and plotted as % of total ErbB-2 mRNA levels.
- targets SpT4 or ErbB-2 ⁇ 16, respectively
- FIG. 13 Total ErbB-2 mRNA levels assessed by Reverse Transcriptase (RT)-quantitative PCR (qPCR). Fold change was calculated by normalizing the absolute levels of ErbB-2 mRNA to those of GAPDH, used as an internal control, setting the value of BT-474 cells to 1. Data are presented as mean ⁇ S.D. For b vs a: P ⁇ 0.001. Primers for total ErbB-2 are depicted as SEQ ID NOs: 21 and 22, and primers for GAPDH have SEQ ID NOs: 25 and 26.
- FIGS 14A-14B SpT3 expression levels assessed by RT-qPCR.
- 14A Total ErbB-2 and SpT3 mRNA levels were measured by RT-qPCR. Fold change was calculated by normalizing the absolute levels of total ErbB-2 or SpT3 mRNA to those of GAPDH, used as an internal control, setting the value of BT-474 cells to 1. Data are presented as mean ⁇ S.D. For b vs a: P ⁇ 0.001.
- FIGS 15A-15G siRNAs targeting the common coding region of transcript variants 1 to 4, and of ErbB-2 ⁇ 16 are unable to silence p165ErbB-2 expression.
- 15A Schematic diagram of the target sequences of a pool of 4 siRNAs (ErbB-2 siRNA, siGENOME SMARTpool siRNAs for human ErbB-2 (2064) cat #M-0031126-04-0020, Dharmacon, Lafayette, CO, USA) designed to target the common coding region of all ErbB-2 variants analyzed in the present application (grey boxes indicating regions between nucleotides (nt): 1080-1098, 1558-1576, 1559-1577, and 2565-2583).
- CCDSs Consensus coding sequences
- UTRs untranslated regions
- Numbers below the lines indicate the sequence length of mRNA in nt. Shown are the position of the translation start site (ATG) and the translation stop codon (TGA).
- the pool of siRNAs targeting ErbB-2 have the sequences: GGACGAAUUCUGCACAAUG (SEQ ID NO: 27); GACGAAUUCUGCACAAUGG (SEQ ID NO: 28); CUACAACACAGACACGUUU (SEQ ID NO: 29) and AGACGAAGCAUACGUGAUG (SEQ ID NO: 30).
- BT-474 and TNBC cells were transfected with either Control siRNA (siGENOME Non-Targeting siRNA #5, cat #D-001210-05-20, Dharmacon, Lafayette, CO, USA), shown in the sequence listing as SEQ ID NO: 31 (UGGUUUACAUGUCGACUAA), or with the abovementioned pool of 4 siRNAs (ErbB-2 siRNA) at a final concentration of 100 nM.
- Protein extracts 50 ⁇ g were examined for ErbB-2 isoforms expression with the C-18 antibody.
- ⁇ -tubulin was used as a loading control.
- ErbB-2 siRNA pool had no effect on NErbB-2 levels in TNBC cells only expressing p165ErbB-2.
- Cell lines were transfected with control siRNA (SEQ ID NO: 31) (upper panels) or with ErbB-2 siRNA pool (SEQ ID NOs: 27-30) (lower panels) at a final concentration of 100 nM. Nuclei were stained with propidium iodide (PI). ErbB-2 levels were visualized by IF and confocal microscopy using the ErbB-2 C-18 antibody in BT-474 (16A) and TNBC cell lines: MDA-MB-468 (16B), HCC-70 (16C), MDA-MB-231 (16D) and MDA-MB-453 (16E).
- FIGS 17A-17E ErbB-2 siRNA pool blocks proliferation in BC cells expressing only p185ErbB-2.
- BT-474 (17A), MDA-MB-468 (17B), HCC-70 (17C), MDA-MB-231 (17D) and MDA-MB-453 (17E) cells were transfected with Control siRNA (SEQ ID NO: 31) or with the ErbB-2 siRNA pool (SEQ ID NOs: 27-30) at a final concentration of 100 nM and proliferation was assessed by [ 3 H]-thymidine uptake. Data are presented as mean ⁇ S.D. of three independent experiments. For b vs a: P ⁇ 0.001.
- FIGS 18A-18D siRNAs targeting variant 3 (SpT3) which encodes the non-canonical isoform c, silence p165ErbB-2 expression.
- SpT3 siRNA #1 SEQ ID NO: 1
- SpT3 siRNA #2 SEQ ID NO: 2
- SpT3 siRNAs have the sequences: GUGAGAUACUUCAAAGAUU (SEQ ID NO: 1) and CAAAGAUUCCAGAAGAUAU (SEQ ID NO: 2).
- Consensus coding sequences and untranslated regions (5' and 3' UTRs) are represented as full lines and dashed lines, respectively. Numbers below lines indicate the sequence length of mRNA in nucleotides (nt). The position of the translation start site (ATG) and the translation stop codon (TGA) are also shown.
- Target sequences of SpT3 siRNA #1 (SEQ ID NO: 1) and SpT3 siRNA #2 (SEQ ID NO: 2) are depicted as a dark grey box indicating region between nt 555-573, and as a black box indicating region between nt 566-584, respectively.
- BT-474 and TNBC cells were transfected with Control siRNA (SEQ ID NO: 31) or with SpT3 siRNA #2 (SEQ ID NO: 2) targeting SpT3, at a final concentration of 100 nM.
- Protein extracts (10 ⁇ g) were examined for ErbB-2 isoforms expression with the C-18 antibody.
- ⁇ -tubulin was used as a loading control.
- Signal intensities of (18C) p185ErbB-2 and (18D) p165ErbB-2 (ErbB-2 isoform c) were analyzed by densitometry from three independent WBs performed as in Fig. 18B . Fold change was calculated by normalizing the absolute levels of each ErbB-2 isoform to those of ⁇ -tubulin, setting the value of Control siRNA-transfected cells to 1.
- b vs a P ⁇ 0.001.
- SpT3 siRNA #2 reduces nuclear ErbB-2 levels in TNBC cells expressing only p165ErbB-2 (ErbB-2 isoform c) or both p165ErbB-2 (ErbB-2 isoform c) and p185ErbB-2.
- TNBC cell lines were transfected with Control siRNA (SEQ ID NO: 31) (upper panels) or with SpT3 siRNA #2 (SEQ ID NO: 2) (lower panels) at a final concentration of 100 nM. Nuclei were stained with DAPI.
- ErbB-2 levels were visualized by IF and confocal microscopy using the ErbB-2 C-18 antibody in MDA-MB-468 (19A), MDA-MB-231 (19B) and MDA-MB-453 cells (19C). Quantitative analysis of NErbB-2 expression in confocal images from Figs. 19A to 19C was performed. Fluorescence intensities from nuclear compartment (black bars) were quantified in 50 cells from MDA-MB-468 (19D), MDA-MB-231 (19E) and MDA-MB-453 (19F), and plotted (mean ⁇ S.D.). For b vs a: P ⁇ 0.001. Experiments in Figs. 19A to 19C and its quantification were repeated thrice with similar results.
- FIGS 20A-20E SpT3 siRNA #2 inhibits in vitro proliferation of TNBC cells expressing only p165ErbB-2 (ErbB-2 isoform c) or both p165ErbB-2 (ErbB-2 isoform c) and p185ErbB-2.
- (20A) BT-474, (20B) MDA-MB-468, (20C) HCC-70, (20D) MDA-MB-231 and (20E) MDA-MB-453 cells were transfected with Control siRNA (SEQ ID NO: 31) or with SpT3 siRNA #2 (SEQ ID NO: 2) targeting SpT3, at a final concentration of 100 nM and proliferation was assessed by [ 3 H]-thymidine uptake. Data are presented as mean ⁇ S.D. of three independent experiments. For b vs a: P ⁇ 0.001.
- FIGS 21A-21F Effects of SpT3 siRNA #1.
- (21A) BT-474 and TNBC cells (MDA-MB-468, HCC-70, MDA-MB-231 and MDA-MB-453) were transfected with Control siRNA (SEQ ID NO: 31) or with SpT3 siRNA #1 (SEQ ID NO: 1) at a final concentration of 100 nM.
- Protein extracts 50 ⁇ g were examined for ErbB-2 isoforms expression with the C-18 antibody.
- ⁇ -tubulin was used as a loading control. Images shown are representative of three independent experiments.
- FIG 22 Effects of SpT3 siRNA #2 on tumor explants.
- Triple negative tumor explants established from MDA-MB-468 xenografts were cultured with medium containing SpT3 siRNA #2 (SEQ ID NO: 2) or Control siRNA (SEQ ID NO: 31) at a final concentration of 100 nM.
- WB analyses of p165ErbB-2 (ErbB-2 isoform c) and Erk5 were performed in tumor lysates after 24 hours of transfection. Numbers under each blot represent the corresponding densitometric quantification.
- Fold change of protein levels was calculated by normalizing the absolute levels of p165ErbB-2 (ErbB-2 isoform c) and Erk5 bands to those of ⁇ -tubulin, used as loading control, setting the value of control siRNA-transfected tumors to 1.
- FIG. 23A-23B Preclinical model of the blockade of p165ErbB-2 (ErbB-2 isoform c) expression with SpT3 siRNA #2.
- (23B) Mice were sacrificed two days after the last treatment and tumor lysates were analyzed by WB with the C-18 antibody. Shown are two representative tumors from each group.
- FIGS. 24A-24B Silencing of p165ErbB-2 (ErbB-2 isoform c) with SpT3 siRNA #2 in the MDA-MB-468 preclinical model reduces nuclear ErbB-2 expression.
- (24) IF and confocal microscopy of histological sections from MDA-MB-468 tumors excised at the end of the experiment. ErbB-2 was detected using the C-18 antibody, followed by incubation with an IgG-Alexa Fluor 488 secondary antibody. Nuclei were stained with DAPI. Representative images are shown.
- 24B Quantitative analysis of NErbB-2 expression in images from Fig. 24A .
- FIGS 25A-25D Histopathological analysis.
- 25A Representative hematoxylin and eosin (H&E) staining of histological sections from MDA-MB-468 tumors excised at the end of the experiment. Mitotic figures are indicated with black arrows.
- 25B Tumor proliferation was quantified by mitotic figures count per high power field (HPF) in MDA-MB-468 histological sections. Data are presented as mean ⁇ S.D. For b vs a: P ⁇ 0.001.
- 25C Representative H&E staining of histological sections from MDA-MB-468 tumors showing areas with extensive necrosis (pale eosinophilic areas) after SpT3 siRNA #2 treatment.
- FIGS 26A-26D Preclinical toxicological analysis of animal blood samples.
- ALT Alanine transaminase
- AST aspartate aminotransferase
- 26C total bilirubin levels were determined for evaluating hepatotoxicity in SpT3 siRNA #2 injected mice.
- 26D Creatinine levels were also measured to assess SpT3 siRNA #2 effects on renal function. n.s., non-significat by unpaired Student's t test.
- nucleotide base is a heterocyclic pyrimidine or purine compound that is a component of a nucleotide, and includes the primary purine bases adenine (A) and guanine (G), and the primary pyrimidine bases cytosine (C), thymine (T), and uracil (U).
- a nucleobase may further be modified to include, without limitation, universal bases, hydrophobic bases, promiscuous bases, size-expanded bases, and fluorinated bases. (See. e.g., Modified Nucleosides in Biochemistry, Biotechnology and Medicine, Herdewijn, P. ed. Wiley-VCH, 2008 ). The synthesis of such modified nucleobases (including phosphoramidite compounds that include modified nucleobases) is known in the art.
- nucleoside refers to a molecule made up of a nucleotide base and its sugar.
- nucleotide refers to a nucleoside having a phosphate group on its 3' or 5' sugar hydroxyl group.
- oligonucleotide means a polymer of linked nucleotides each of which can be independently modified or unmodified.
- the polynucleotides can be “deoxyribonucleic acid” (DNA), “ribonucleic acid” (RNA), or derivatives or modified versions thereof.
- the polynucleotide may be single-stranded or double-stranded.
- deoxynucleotide refers to a nucleotide or polynucleotide lacking a hydroxyl group (OH group) at the 2' and/or 3' position of a sugar moiety. Instead, it has a hydrogen bonded to the 2' and/or 3' carbon.
- deoxynucleotide refers to the lack of an OH group at the 2' position of the sugar moiety, having instead a hydrogen bonded directly to the 2' carbon.
- deoxyribonucleotide and “DNA” refer to a nucleotide or polynucleotide comprising at least one sugar moiety that has an H, rather than an OH, at its 2' and/or 3' position.
- ribonucleotide and the phrase “ribonucleic acid” (RNA), refer to a modified or unmodified nucleotide or polynucleotide comprising at least one ribonucleotide unit.
- a ribonucleotide unit comprises an hydroxyl group attached to the 2' position of a ribosyl moiety that has a nitrogenous base attached in N-glycosidic linkage at the 1' position of a ribosyl moiety, and a moiety that either allows for linkage to another nucleotide or precludes linkage
- 3' end refers to the end of a nucleic acid that contains an unmodified hydroxyl group at the 3' carbon of its ribose ring.
- 5' end refers to the end of a nucleic acid that contains a phosphate group attached to the 5' carbon of its ribose ring.
- mRNA messenger RNA
- mRNA transcripts include, but are not limited to pre-mRNA transcript(s), transcript processing intermediates, mature mRNA(s) ready for translation and transcripts of the gene or genes, or nucleic acids derived from the mRNA transcript(s). Processing of pre-mRNA transcripts, along with the possible use of alternative promoters and alternative polyadenylation sites, allows a single gene to generate many different mature RNAs, by varying the pattern of splicing in a process known as alternative splicing. Alternative splicing can also introduce or remove regulatory elements to affect mRNA translation, localization or stability.
- alternatively spliced mRNAs are translated into alternative splice form proteins, here referred to as protein isoforms, that contain different amino acid sequences than the corresponding wild-type or canonical protein produced by normally spliced mRNA.
- RNAi agent and "RNA silencing agent” means a composition that contains an RNA or RNA-like (e.g., chemically modified RNA) oligonucleotide molecule that is capable of degrading or inhibiting (e.g., degrades or inhibits under appropriate conditions) translation of messenger RNA (mRNA) transcripts of a target mRNA in a sequence specific manner.
- RNAi agents may operate through the RNA interference mechanism (i.e., inducing RNA interference through interaction with the RNA interference pathway machinery (RNA-induced silencing complex or RISC) of mammalian cells), or by any alternative mechanism(s) or pathway(s). While it is believed that RNAi agents, as that term is used herein, operate primarily through the RNA interference mechanism, the disclosed RNAi agents are not bound by or limited to any particular pathway or mechanism of action.
- siRNAs small (or short) interfering RNA
- siRNAs consist of two RNA strands, an antisense (or guide) strand and a sense (or passenger) strand. These molecules display generally between 18-30 basepairs and contain varying degrees of complementarity to their target mRNA in the antisense strand.
- Some, but not all, siRNAs include structures with overhangs. Overhangs have been described to be advantageous and may be present on the 5' ends or on the 3' ends of either strand as they reduce recognition by RNAses. Some authors recommend including overhangs on both 3' ends of the molecules, whereas others consider one overhang to be sufficient.
- overhangs are said to further enhance resistance to nuclease (RNase) degradation.
- RNase nuclease
- tail refers to 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20 or more sequential nucleotides at the 3' end of one or both of the sense strand and the antisense strand that are single-stranded.
- RNA includes duplexes of two separate strands, as well as single strands that can form hairpin structures comprising a duplex region.
- antisense (or guide) strand refers to the strand of an siRNA duplex that contains some degree of complementarity to a target gene or mRNA and contains complementarity to the sense strand of the siRNA duplex.
- sense (or passenger) strand refers to the strand of an siRNA duplex that contains complementarity to the antisense strand of the siRNA duplex.
- duplex region refers to the region in two complementary or partially complementary polynucleotides (e.g., a sense strand or an antisense strand) that form base pairs with one another.
- a base pair is a unit consisting of two nucleobases connected by hydrogen bonds.
- DNA contains four bases: the two purines adenine (A) and guanine (G) and the two pyrimidines cytosine (C) and thymine (T).
- RNA thymine is replaced by uracil (U).
- Non-Watson-Crick base-pairing models display alternative hydrogen-bonding patterns; examples are Hoogsteen base pairs, which are A-T or C-G analogues.
- nucleotide or “nucleotide analog” or “altered nucleotide” or “modified nucleotide” refer to a non-standard nucleotide, including non-naturally occurring ribonucleotides or deoxyribonucleotides.
- nucleotide analogs may be modified to change chemical properties of the nucleotide, maintaining the ability of the nucleotide analog to perform its function.
- Nucleotide analogs include nucleotides having modifications in the chemical structure of the base, sugar and/or phosphate.
- the nucleobase moiety may be modified by the replacement or addition of one or more atoms or groups.
- a pyrimidine base may be modified at the 2, 3, 4, 5, and/or 6 position of the pyrimidine ring.
- the exocyclic amine of cytosine may also be modified.
- a purine base may be modified at the 1, 2, 3, 6, 7, or 8 position.
- the exocyclic amine of adenine may also be modified.
- Base modifications are known to those of ordinary skill in the art. Some examples include alkylated, halogenated, thiolated, aminated, amidated, or acetylated bases, or other heterocycles. Nucleobase modifications in small amounts (up to 10%) could reduce immune reactions and improve the thermodynamic siRNA profile.
- nucleobases substitutions Reviewed in Chernikov, I.V. et al. (2019) Current Development of siRNA Bioconjugates: From Research to the Clinic. Frontiers in pharmacology 10, 444 ) with various modified analogs (pseudouridine, 2'thiouridine, dihydrouridine, 2,4-difluorobenzene, 4-methylbenzimidazole, hypoxanthine, 7-deazaguanin, N2-alkyl-8-oxoguanine, N2-benzylguanine, and 2,6-diaminopurine) may be used to increase the thermal stability of the siRNA duplex.
- modified analogs pseudouridine, 2'thiouridine, dihydrouridine, 2,4-difluorobenzene, 4-methylbenzimidazole, hypoxanthine, 7-deazaguanin, N2-alkyl-8-oxoguanine, N2-benzylguanine, and 2,6-dia
- Non-limiting examples of modified bases are described in U.S. Pat. Nos. 10,011,836 ; 8,008,474 ; 7,816,512 and 7,977,471 , all of which are incorporated herein by reference.
- nucleotide analogs may also display modifications at the sugar moiety.
- Sugar moieties include natural, unmodified sugars, e.g., monosaccharide (such as pentose, e.g., ribose, deoxyribose), modified sugars and sugar analogs.
- modifications of sugar moiety include substitutions in the 2'-position, as the 2'OH group participates in the cleavage of RNA by endoribonucleases.
- modifications include but are nor limited to, the replacement of the 2'OH group by a group selected from H, OR, R, F, Cl, Br, I, SH, SR, NH2, NHR, NR2, COOR, or OR, wherein R is substituted or unsubstituted C1-C6 alkyl, alkenyl, alkynyl, aryl, etc, as described in U.S. Pat. No. 9,080,171 and U.S. Pat. Application No. 2019/0024082 , all of which are incorporated herein by reference.
- Nucleotide analogs with a modified structure of the furanose cycle such as derivatives containing 6-membered (hexitol (HNA), cyclohexenic (CeNA) and altritol (ANA) nucleic acids), and 7-membered rings (oxepanic nucleic acid (ONA)), bicyclic (locked nucleic acids (LNA), 2'-deoxymethanocarbanucleosides (MCs)), tricyclic (tricyclo-DNA (tc-DNA)), and acyclic (unlocked nucleic acid (UNA)) derivatives, can be used.
- HNA 6-membered
- CeNA cyclohexenic
- ANA altritol
- LNA locked nucleic acids
- MCs 2'-deoxymethanocarbanucleosides
- tc-DNA tricyclic
- UPA unlocked nucleic acid
- nucleotide analogs may also display nonnatural phosphodiester linkages such as methylphosphonates, phosphorothioates and peptides.
- nucleotide analogs may also comprise hydrophobic modifications at the base moiety.
- hydrophobic modifications are phenyl, indolyl, isobutyl, butyl, aminobenzyl, benzyl, thiophene, ethynyl, and imidazole.
- nucleotide also includes what are known in the art as universal bases. These nucleotide analogues lack hydrogen bonding sites and are generally hydrophobic aromatic base residues. Some examples are 3-nitropyrrole 5-nitroindole, or nebularine.
- nucleotide is also meant to include the nucleotide analogues N3' to P5' phosphoramidates. These compounds contain 3'-amino group replacing the ribosyl 3'-O-atom.
- nucleotide also includes those species that have a detectable label, such as for example a radioactive or fluorescent moiety, or mass label attached to the nucleotide.
- gene silencing or “knockdown” refers to a process by which the expression of a specific gene product is lessened or attenuated.
- shilenced or “knockdown”, when referring to expression of a given gene, mean that the expression of the gene, as measured by the level of RNA transcribed from the gene or the level of polypeptide, protein, alternative isoform of a given protein, e.g.
- ErbB-2, or protein subunit translated from the mRNA in a cell, group of cells, tissue, organ, or subject in which the gene is transcribed is reduced when the cell, group of cells, tissue, organ, or subject is treated with the siRNAis described herein as compared to a second cell, group of cells, tissue, organ, or subject that has not or have not been so treated.
- off-target silencing and off-target interference are defined as degradation of mRNA other than the intended target mRNA due to overlapping and/or partial homology with secondary mRNA messages.
- Such off-target effects are primarily mediated by the sequence-specific interaction between the siRNA seed regions (position 2-8 of either siRNA strand counting from the 5'-end) and complementary sequences in the 3' UTR of off-targets ( Bramsen, J.B. et al. (2010) A screen of chemical modifications identifies position-specific modification by UNA to most potently reduce siRNA off-target effects. Nucleic acids research 38, 5761-5773 ).
- siRNA score refers to a number determined by applying any of the design algorithms or formulas (e.g., siDESIGN Center, Dharmacon, Lafayette, CO, USA) to a given siRNA sequence.
- the present invention provides specific siRNA molecules for use to silence a particular mRNA (e.g., ErbB-2 alternative transcript variant 3, SpT3) which have been selected on the basis of this method.
- Target mRNA refers to a messenger RNA (mRNA) to which a given siRNA can be directed against.
- Target sequence refers to a contiguous portion of the nucleotide sequence of a target mRNA molecule formed during the transcription of a target gene, including mRNA that is a product of RNA processing of a primary transcription product.
- siRNA target can refer to the gene, mRNA, or protein against which an siRNA is directed.
- Non-targeting siRNA As used herein, “Non-targeting siRNA”, “Control siRNA” (siGENOME Non-Targeting siRNA#5 cat #D-001210-05-20, Dharmacon, Lafayette, CO, USA), refers to a highly functional, chemically synthesized negative control siRNA duplex for experiments in human, mouse, and rat cells.
- the control siRNA oligonucleotide used herein does not target any known mammalian gene. Neither the mRNA nor protein level of the experimental gene is affected by this control siRNA. Cell viability as well as cell phenotype of samples treated with the control siRNA remains comparable to those of untreated samples.
- control siRNA does not bind to ErbB-2 alternative transcript variant 3 (SpT3), which encodes isoform c of ErbB-2.
- SpT3 ErbB-2 alternative transcript variant 3
- the control siRNA used in the assays carried out in the present invention consists of the sequence UGGUUUACAUGUCGACUAA shown as SEQ ID NO: 31.
- an RNA silencing agent (or any portion thereof), e.g., the siRNA molecules of the present invention targeting ErbB-2 alternative transcript SpT3 which encodes ErbB-2 isoform c, as described herein, may be modified such that the activity of the RNA silencing agent is further improved.
- the RNA silencing agents described in the present invention may be modified with any of the modifications described herein. The modifications can, in part, serve to further enhance target discrimination, to enhance stability of the agent (e.g., to prevent degradation), to promote cellular uptake, to enhance the target efficiency, to improve efficacy in binding (e.g., to the targets), to improve patient tolerance to the agent, and/or to reduce toxicity.
- certain modifications may be incorporated to facilitate preferential selection of the antisense strand of the siRNA molecule by the cellular RNAi machinery, such that the antisense strand preferentially guides cleavage or translational repression of a target mRNA, and thus increasing the efficiency of target cleavage and silencing.
- Non-limiting examples of structural and chemical modifications used to achieve preferential strand selection include the development of internally destabilized duplexes, asymmetric duplexes and 2'OMe modifications of the two nucleotides at the 5' end of the sense strand.
- the siRNAs of the invention may be substituted with a destabilizing nucleotide.
- a destabilizing nucleotide Non-limiting examples include acyclic nucleotide residues which decrease the stability when incorporated into RNA duplexes.
- Unlocked nucleic acid (UNA) is reported to destabilize RNA-RNA interactions and can be strategically used in siRNA design to induce local thermodynamical destabilization (Bramsen, J.B. et al. (2010) op. cit. )
- the siRNAs of the invention may be altered according to asymmetry design rules.
- Asymmetric siRNA compounds have previously been developed. Non-limiting examples include: i) siRNAs with deletions at the sense and/or antisense strand, with a duplex region of 16 nucleotides (nt) ( Chu, C.Y. and Rana, T.M. (2008) Potent RNAi by short RNA triggers. Rna 14, 1714-1719 ); ii) asymmetric interfering RNA (aiRNA) compounds with a short sense strand (12-15 nt) and a regular length antisense strand (21 nt) ( Sun, X. et al.
- sd-rxRNA ® a new class of small, hydrophobic, asymmetric RNAi compounds, termed "self-delivering rxRNAs"
- sd-rxRNA ® are extensively modified siRNAs compounds, with a small duplex region of ⁇ 15 nt, and a fully phosphorothioated single-stranded tail.
- sd-rxRNAs are functional in vitro and in vivo without the aid of any delivery vehicle ( Byrne, M. et al. (2013) Novel hydrophobically modified asymmetric RNAi compounds (sd-rxRNA) demonstrate robust efficacy in the eye. J Ocul Pharmacol Ther 29, 855-864 ).
- modifications that increase or decrease sugar flexibility including locked nucleic acid (LNA) and unlocked nucleic acid (UNA), may be used to introduce chemical asymmetry into duplex siRNAs (Reviewed in Khvorova, A. and Watts, J.K. (2017) The chemical evolution of oligonucleotide therapies of clinical utility. Nature biotechnology 35, 238-248 ).
- LNA locked nucleic acid
- UPA unlocked nucleic acid
- siRNA molecules of the present invention may be modified at various locations in order to promote metabolic stability and enhance silencing activity.
- modifications include, but are not limited to: a) sugar modifications such as 2' ribo modifications, including particularly combinations of 2'-O-methyl (2'OMe), 2'-fluoro (2'F), 2'-deoxy and others; modifications that increase or decrease sugar flexibility, including locked nucleic acid (LNA) and unlocked nucleic acid (UNA); b) backbone modifications such as phosphorothioate internucleotide linkages (referred to as phosphorothioate modifications) at both ends of both strands of the siRNA duplex; and methylation of the 5' carbon to give (S)-5'-C-methyl-RNA; and c) 5'-phosphate stabilization of the siRNA guide strand by incorporation of phosphate analogs such as 5'E-vinyl phosphonate (5'-E-VP), 5'-methyl phosphonate, 5'-C-methyl
- immune stimulation may be beneficial for the treatment of cancer and siRNAs with both RNAi and immunostimulatory activity (isiRNAs)
- siRNAs with both RNAi and immunostimulatory activity
- isiRNAs RNAi and immunostimulatory activity
- 5'-triphosphate modifications known to activate the cytosolic antiviral helicase retinoic acid-induced protein I (Rig-I) and, therefore induce innate immunity, may be introduced to the siRNAs of the present invention (Poeck, H. et al. (2008) op.cit.).
- siRNA molecules of the present invention may be associated with a hydrophobic moiety for targeting and/or delivery of the molecule to a cell.
- the hydrophobic moiety is associated (covalently or noncovalently) with the nucleic acid molecule through a linker. Any linker known in the art may be used to associate the nucleic acid with the hydrophobic moiety.
- the linker is capable of being cleaved from the nucleic acidunder physiological conditions (e.g., acid labile linkers are cleaved in the acidic endosomal/lysosomal compartments, and disulfide linkers are cleaved at the reductive environment of cytosolic space).
- the linker is capable of being cleaved by an enzyme (e.g., an esterase or phosphodiesterase).
- an enzyme e.g., an esterase or phosphodiesterase.
- linkers employed for hydrophobic/lipid conjugation include: trans-4-hydroxyprolinol linker, disulfide linker, aminohexyl linker, triethyl glycol (TEG) linker and 2-aminobutyl-1-3-propanediol (C7) linker.
- the hydrophobic moiety is linked at various positions of the siRNA molecule. In a preferred embodiment, the hydrophobic moiety is linked to either the 3' or 5' end of the sense strand. In an exemplary embodiment, the hydrophobic moiety is selected from the group consisting of saturated fatty acids (e.g. docosanoic acid (DCA)), non-saturated fatty acids (e.g. docosahexaenoic acid (DHA, 22:6 n-3) and eicosapentaenoic acid (EPA, 20:5 n-3)), sterols (e.g. cholesterol (Chol) and lithocholic acid (LA)) and vitamins (e.g.
- DCA docosanoic acid
- DHA docosahexaenoic acid
- LA lithocholic acid
- RA retinoic acid
- TS ⁇ -tocopheryl succinate
- cationic dyes e.g., Cy3
- the siRNA molecules of the present invention contain 5'and/or 3'-end overhangs.
- the number and/or sequence of nucleotides overhang on one end of the polynucleotide may be the same or different from the other end of the polynucleotide.
- one or more of the overhang nucleotides may contain chemical modification(s), such as phosphorothioate or 2'OMe modification.
- the 3' and 5' end of the siRNA molecules of the present invention can be substantially protected from nucleases by modifying the 3' or 5' linkages (e.g., U.S. Pat. No. 5,849,902 and WO 98/13526 ).
- End-blocking groups or “exonuclease blocking groups” may also be used to protect the 5' and 3' end of the oligonucleotides.
- Such groups include modified nucleotides and non-nucleotide exonuclease resistant structures. Exemplary end-blocking groups are described in U.S. Pat. No. 9,080,171 .
- Oligonucleotides and oligonucleotide compositions of the present invention are contacted with (i.e., brought into contact with, also referred to herein as administered or delivered to) and taken up by one or more cells.
- the term "cells" includes eukaryotic cells, preferably vertebrate cells, and, more preferably, mammalian cells.
- the oligonucleotide compositions of the invention are contacted with human cells.
- siRNAs of the present invention can be delivered or administered to a cell (e.g., a cancer cell) in vitro, in vivo, or ex vivo.
- a cell e.g., a cancer cell
- novel siRNAs disclosed herein are suitable for any known delivery method, including intratumor or systemic routes.
- Delivery of the siRNAs of the present invention to an organelle, cell, tissue, and/or organism can be done by any method known to those skilled in the art.
- One exemplary means of delivering or introducing siRNAs into a cell is by transfection or transduction procedures.
- Transfection refers to the acquisition by a cell of siRNAs by incorporation of added siRNAs molecules. Transfection can occur by physical or chemical methods. Transduction refers to the process of transferring nucleic acid into a cell using a DNA or RNA virus.
- Such methods for delivering siRNAs to an organelle, cell, tissue, and/or organism include, but are not limited to, direct delivery of RNA such as by ex vivo transfection, by injection, including microinjection; by electroporation; by calcium phosphate precipitation; by using DEAE-dextran followed by polyethylene glycol; by direct sonic loading; by liposome mediated transfection and receptor-mediated transfection; by microprojectile bombardment; by agitation with silicon carbide fibers; by Agrobacterium-mediated transformation; by PEG-mediated transformation of protoplasts; by desiccation/inhibition-mediated RNA uptake, naked plasmid adsorption, and any combination of such methods.
- organelle(s), cell(s), tissue(s) or organism(s) may be stably or transiently transformed.
- a vector may be used in some embodiments as a carrier for the siRNA.
- the vector may comprise deoxyribonucleic acids (DNA) and/or ribonucleic acids (RNA).
- Vectors include plasmids, cosmids, viruses (bacteriophage, animal viruses, and plant viruses), and artificial chromosomes (e.g., YACs).
- plasmid vectors such as E. coli; phage vectors; and viral vectors such as adenoviral vectors, adeno-associated virus (AAV) vectors, retroviral vectors, vaccinia viruses, and Semliki Forest virus vectors.
- AAV adeno-associated virus
- an oligonucleotide is associated with a carrier or vehicle.
- the siRNAs of the present invention can be delivered or administered to a cell by suitable art recognized methods including but not limited to: a) lipid-based siRNA delivery, b) polymer-based siRNA delivery, c) bioconjugated siRNA delivery, d) inorganic nanoparticles, and e) extracellular vesicles.
- Lipid-based systems for delivering siRNAs embody varied lipid nanoparticles, including liposomes, micelles, emulsions, and solid lipid nanoparticles.
- lipid composition, drug-to-lipid ratio, particle size, and the manufacturing process should be optimized.
- Liposomal formulations containing the compounds (e.g., siRNAs), disclosed herein or compositions thereof may be lyophilized, to produce a lyophilizate, which may be reconstituted with a pharmaceutically acceptable carrier, such as water, to regenerate a liposomal suspension.
- a pharmaceutically acceptable carrier such as water
- Cationic liposomes are vesicular structures characterized by a phospholipid bilayer membrane and an inner aqueous medium. Multilamellar liposomes have multiple lipid layers separated by aqueous medium. They form spontaneously when phospholipids are suspended in an excess of aqueous solution. The diameters of the liposomes generally range from about 15 nm to about 5 microns.
- the components mainly include cationic lipids and neutral adjuvant lipids. Cationic lipids can be readily mixed and complexed with negatively charged siRNA molecules to form nanoparticles by electrostatic interaction. Diphenol phthalocyanine ethanolamine (DOPE) and cholesterol are commonly used as neutral auxiliary lipid molecules.
- DOPE Diphenol phthalocyanine ethanolamine
- cholesterol are commonly used as neutral auxiliary lipid molecules.
- the technology for forming liposomal suspensions is well known in the art.
- the compound or composition thereof is an aqueous-soluble composition
- using conventional liposome technology the same may be incorporated into lipid vesicles.
- the compound or composition will be substantially entrained within the hydrophilic center or core of the liposomes.
- the lipid layer employed may be of any conventional composition and may either contain cholesterol or may be cholesterol-free.
- the compound or composition of interest is water-insoluble, again employing conventional liposome formation technology, the composition may be substantially entrained within the hydrophobic lipid bilayer that forms the structure of the liposome. In either instance, the liposomes that are produced may be reduced in size, as through the use of standard sonication and homogenization techniques.
- cationic liposomes are stable nucleic acid lipid particles (SNALPs).
- SNALPs the siRNA is surrounded by a lipid bilayer containing a mixture of cationic lipids, fused lipids, cholesterol, and polyethylene glycol (PEG)-modified neutral lipids.
- PEG polyethylene glycol
- nucleic acids associated with the invention can be hydrophobically modified and can be encompassed within neutral nanotransporters (further description of neutral nanotransporters is incorporated in U.S. Pat. No. 10,138,485 ). Such particles enable quantitative oligonucleotide incorporation into noncharged lipid mixtures. The lack of toxic levels of cationic lipids in such neutral nanotransporter compositions is an important feature.
- the neutral nanotransporters compositions enable efficient loading of oligonucleotide into neutral fat formulation.
- the composition includes an oligonucleotide that is modified in a manner such that the hydrophobicity of the molecule is increased (for example a hydrophobic molecule is attached (covalently or no-covalently) to a hydrophobic molecule on the oligonucleotide terminus or a non-terminal nucleotide, base, sugar, or backbone), the modified oligonucleotide being mixed with a neutral fat formulation (for example containing at least 25% of cholesterol and 25% of 1,2-dioleoyl-sn-glycero-3-phosphatidylcholine (DOPC) or analogs thereof).
- DOPC 1,2-dioleoyl-sn-glycero-3-phosphatidylcholine
- a cargo molecule such as another lipid can also be included in the composition.
- lipid-like delivery molecules termed lipidoids may also be used to deliver the siRNAs of the invention ( Akinc, A. et al. (2008) A combinatorial library of lipid-like materials for delivery of RNAi therapeutics. Nature biotechnology 26, 561-569 ).
- a cationic polymer-based delivery system may be used for siRNA administration.
- the siRNA is condensed within different kinds of cationic polymers that form nanoparticles, and the surface of the nanoparticles is decorated with PEG and targeting moieties.
- Said molecules include, but are not limited to, natural polymer materials such as chitosan, beta-cyclodextrin, hyaluronic acid, and gelatin, and synthetic polymers such as poly-L-lysine (PLL), poly-L-glutamic acid (PGA), and polyethyleneimine (PEI).
- dendrimers may be used for siRNA administration.
- Dendrimers are synthetic, highly branched macromolecules with three-dimensional nanometric structure. Dendrimers have high surface charge density, which also uses electrostatic interactions to load siRNA drugs effectively. Cationic dendrimers have proven useful in masking the charge of siRNA long enough for in vivo delivery.
- dendrimers are polyamidoamine dendrimers (PAMAM), polylylamine dendrimers (poly-L-lysine dendrimers), and polypropylene imine dendrimers (polypropylenimine dendrimers), among others.
- PAMAM polyamidoamine dendrimers
- polylylamine dendrimers poly-L-lysine dendrimers
- polypropylene imine dendrimers polypropylenimine dendrimers
- the delivery of siRNAs can also be improved by targeting the siRNAs to a cellular receptor.
- the targeting moieties can be conjugated to the oligonucleotides or attached to a carrier group (i.e., poly(L-lysine) or liposomes) linked to the oligonucleotides. This method is well suited to cells that display specific receptor-mediated endocytosis.
- Non-limiting examples of targeting moieties linked to oligonucleotides include 6-phosphomannosylated proteins and nucleic-acid aptamers.
- Non-limiting examples of specific ligands linked to the polylysine component of polylysine-based delivery systems include transferrin-polylysine, adenovirus-polylysine, and influenza virus hemagglutinin HA-2 N-terminal fusogenic peptides-polylysine conjugates.
- targeting ligands may be included in the siRNA delivery vehicles, e.g., the use of fusion proteins made by an antibody and the cationic protein protamine ( De Paula, D. et al. (2007) Hydrophobization and bioconjugation for enhanced siRNA delivery and targeting. RNA 13, 431-456 ).
- a liposome may be complexed with a hemagglutinating virus (HVJ). This has been shown to facilitate fusion with the cell membrane and promote cell entry of liposome-encapsulated DNA.
- a liposome may be complexed or employed in conjunction with nuclear non-histone chromosomal proteins (HMG-1).
- HMG-1 nuclear non-histone chromosomal proteins
- a liposome may be complexed or employed in conjunction with both HVJ and HMG-1.
- a delivery vehicle may comprise a ligand and a liposome.
- siRNAs can also be conjugated with cell-penetrating peptides (CPPs), which can enhance the internalization of siRNAs into cells.
- CPPs cell-penetrating peptides
- the composition includes an oligonucleotide which is complementary to a target nucleic acid molecule encoding the protein, and a covalently attached CPP.
- CPPs are cationic or amphiphilic peptides, usually up to 30 amino acids.
- CPPs may be natural or synthetic and can cross the plasma membrane and the blood-brain barrier.
- Non-limiting examples of CPPs include: HIV TAT transcription factor, lactoferrin, Herpes VP22 protein, fibroblast growth factor 2, penetratin and transportan.
- the guide and/or passenger strands can be attached to a conjugate.
- the conjugate is hydrophobic.
- the hydrophobic molecule is or includes a lipophilic group. Conjugation with lipids may enhance siRNA uptake via receptor-mediated endocytosis or by an increased membrane permeability of the otherwise negatively charged siRNA. The presence of such conjugate can influence the ability of the siRNA molecule to be taken into a cell with or without a lipid transfection reagent.
- molecules associated with the invention are "self-delivering" RNA (sdRNA) including rxRNA ® (RXi Therapeutics).
- sdRNA self-delivering RNA
- rxRNA ® rxRNA ®
- self-delivery refers to the ability of a molecule to be delivered into a cell without the need for an additional delivery vehicle such as a transfection reagent. Further description of self-delivering siRNAs are incorporated by reference from U.S. Patent Application Nos. 2019/0024082 and 2016/0319278 and U.S. Patent No. 9,080,171 .
- the delivery of siRNAs can also be achieved using inorganic nanoparticles.
- inorganic nanoparticles include but are not limited to: gold nanoparticles (AuNPs), magnetic nanoparticles (MNPs), mesoporous silica nanoparticles (MSNPs), carbon nanotubes (CNTs) and quantum dots (QDs).
- siRNAs of the present invention can also be delivered by extracellular vesicles (EVs).
- EVs are bilayer membrane-coated vesicles, 30-100 nm in diameter, which are secreted by cells.
- EVs are "exosomes”.
- Methods generally used to load therapeutic siRNA into the exosomes include but are not limited to: (1) plasma membrane anchors are used to induce a high degree of protein oligomerization on the surface of exosomes; (2) a zipper-like sequence is added to the non-coding region of the mRNA to promote mRNA loading into the exosomes; (3) siRNA is over-expressed in the donor cell, which positively loads it into the exosomes; (4) exosomes are infected with an AAV containing therapeutic information; or (5) small molecule drugs or siRNA are loaded into exosomes via physical methods, including mixing (e.g., for curcumin) and electroporation.
- plasma membrane anchors are used to induce a high degree of protein oligomerization on the surface of exosomes
- a zipper-like sequence is added to the non-coding region of the mRNA to promote mRNA loading into the exosomes
- siRNA is over-expressed in the donor cell, which positively loads it into the exosomes
- hydrophobically modified siRNAs can efficiently be loaded into exosomes upon simple co-incubation without altering vesicle size distribution or intactness ( Didiot, M.C. et al. (2016) Exosome-mediated Delivery of Hydrophobically Modified siRNA for Huntingtin mRNA Silencing. Mol Ther 24, 1836-1847 ).
- a pharmaceutical composition comprising a therapeutically effective amount of one or more siRNA compounds as described herein, and a pharmaceutically acceptable carrier.
- a method of treating or managing breast cancer, particularly TNBC comprising administering to a patient in need of such treatment or management a therapeutically effective amount of an active agent, siRNA, or nucleic acid as described herein, or a pharmaceutical composition comprising said active agent, siRNA, or nucleic acid.
- administration refers to contacting cells with siRNAs of the present invention and can be performed in vitro, in vivo or ex vivo.
- an effective amount refers to the amount of a therapeutic active agent (e.g., siRNA) that produces the desired therapeutic result when administered or delivered to a subject by an appropriate dose and regimen.
- a therapeutic active agent e.g., siRNA
- pharmaceutically acceptable means that the active agent is suitable for administration or delivery to a subject to achieve the treatments described herein, without unduly deleterious side effects in light of the severity of the disease and necessity of the treatment.
- pharmaceutically acceptable carrier is intended to include any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like, compatible with pharmaceutical administration.
- the use of such media and agents for pharmaceutically active substances is well known in the art. Except insofar as any conventional media or agent is incompatible with the active compound, use thereof in the compositions is contemplated. Supplementary active compounds can also be incorporated into the compositions.
- the active agents and/or compositions thereof described herein may be formulated for administration or delivery together with a pharmaceutically acceptable excipient or carrier in accordance with known techniques in the art (See, e.g., Remington, The Science and Practice of Pharmacy (9.sup.th Ed. 1995 )).
- a pharmaceutically acceptable excipient or carrier in accordance with known techniques in the art (See, e.g., Remington, The Science and Practice of Pharmacy (9.sup.th Ed. 1995 )).
- the active compound(s) including the physiologically acceptable salts thereof
- the carrier must, of course, be acceptable in the sense of being compatible with any other ingredients in the formulation and must not be deleterious to the patient.
- the carrier may be a solid or a liquid, or both, and is preferably formulated with the compound(s) as a unit-dose formulation, for example, a tablet, which may contain from 0.01 or 0.5% to 95% or 99% by weight of the active compound.
- One or more active compounds may be incorporated in the formulations of the invention, which may be prepared by any of the well-known techniques of pharmacy comprising admixing the components, optionally including one or more accessory ingredients.
- parenteral administration which in some instances is preferred, includes administration by the following routes: intravenous; intramuscular; interstitially; intraarterially; subcutaneous; intrasynovial; intradermal; intracerebroventricular; intrathecal; transepithelial, including transdermal; pulmonary via inhalation; sublingual and buccal; topically, including dermal; vaginal; rectal; and nasal inhalation via insufflation.
- routes e.g., parenterally, orally, or intraperitoneally.
- Parenteral administration which in some instances is preferred, includes administration by the following routes: intravenous; intramuscular; interstitially; intraarterially; subcutaneous; intrasynovial; intradermal; intracerebroventricular; intrathecal; transepithelial, including transdermal; pulmonary via inhalation; sublingual and buccal; topically, including dermal; vaginal; rectal; and nasal inhalation via insufflation.
- the most suitable route in any given case
- Formulations of the present invention suitable for parenteral administration comprise sterile aqueous and non-aqueous injection solutions of the active compound, which preparations are preferably isotonic with the blood of the intended recipient. These preparations may contain anti-oxidants, buffers, bacteriostats and solutes that render the formulation isotonic with the blood of the intended recipient.
- Aqueous and non-aqueous sterile suspensions may include suspending agents and thickening agents.
- the formulations may be presented in unitary-dose or multi-dose containers, for example sealed ampoules and vials, and may be stored in a freeze-dried (lyophilized) form requiring only the addition of the sterile liquid carrier, for example, saline or waterfor-injection, immediately prior to use.
- Extemporaneous injection solutions and suspensions may be prepared from sterile powders, granules and tablets of the kind previously described.
- an injectable, stable, sterile composition comprising an active compound or composition in a unit dosage form in a sealed container.
- the compound or composition is provided in the form of a lyophilizate that is capable of being reconstituted with a suitable pharmaceutically acceptable carrier to form a liquid composition suitable for injection thereof into a subject.
- a sufficient amount of emulsifying agent that is physiologically acceptable may be employed in sufficient quantity to emulsify the compound or composition in an aqueous carrier.
- emulsifying agent is phosphatidyl choline.
- the pharmaceutical compositions may contain other additives, such as pH-adjusting additives.
- useful pH-adjusting agents include acids, such as hydrochloric acid, bases or buffers, such as sodium lactate, sodium acetate, sodium phosphate, sodium citrate, sodium borate, or sodium gluconate.
- the compositions may contain microbial preservatives.
- Useful microbial preservatives include methylparaben, propylparaben, and benzyl alcohol. The microbial preservative is typically employed when the formulation is placed in a vial designed for multidose use.
- the pharmaceutical compositions of the present invention may be lyophilized using techniques well known in the art.
- Oligonucleotides of the present invention may be incorporated into liposomes or liposomes modified with polyethylene glycol or admixed with cationic lipids for parenteral administration. Incorporation of additional substances into the liposome, for example, antibodies reactive against membrane proteins found on specific target cells, can help target the oligonucleotides to specific cell types.
- Liposomal formulations containing the compounds (e.g., siRNAs), disclosed herein or compositions thereof may be lyophilized, as disclosed above, to produce a lyophilizate, which may be reconstituted with a pharmaceutically acceptable carrier, such as water, to regenerate a liposomal suspension.
- a pharmaceutically acceptable carrier such as water
- Toxicity and therapeutic efficacy of such compounds can be determined by standard pharmaceutical procedures in cell cultures or experimental animals, e.g., for determining the LD50 (the dose lethal to 50% of the population) and the ED50 (the dose therapeutically effective in 50% of the population).
- the dose ratio between toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio LD50/ED50.
- Compounds that exhibit large therapeutic indices are particularly suitable. Although compounds that exhibit toxic side effects may be used, care should be taken to design a delivery system that targets such compounds to the site of affected tissue in order to minimize potential damage to uninfected cells and, thereby, reduce side effects.
- the data obtained from the cell culture assays and animal studies can be used in formulating a range of dosage for use in humans.
- the dosage of such compounds lies typically within a range of circulating concentrations that include the ED50 with little or no toxicity.
- the dosage may vary within this range depending upon the dosage form employed and the route of administration utilized.
- the therapeutically effective dose can be estimated initially from cell culture assays.
- a dose may be formulated in animal models to achieve a circulating plasma concentration range that includes the EC50 (i.e., the concentration of the test compound which achieves a half-maximal response) as determined in cell culture.
- Such information can be used to more accurately determine useful doses in humans.
- Levels in plasma may be measured, for example, by high performance liquid chromatography.
- the active agent of the present invention may optionally be administered in conjunction with other compounds and/or therapies useful in the treatment of cancer.
- siRNAs of the present invention may be administered in both the neoadjuvant and adjuvant settings of a patient's treatment.
- therapies include, but are not limited to, radiotherapeutic agents and factors; surgery; antibiotics such as doxorubicin, daunorubicin, mitomycin, actinomycin D, and bleomycin; chemotherapeutic agents such as cisplatin, VP16, adriamycin, verapamil, and podophyllotoxin; tumor necrosis factor; plant alkaloids such as taxol, vincristine, and vinblastine; and alkylating agents such as carmustine, melphalan, cyclophosphamide, chlorambucil, busulfan, and lomustine.
- radiotherapeutic agents and factors include, but are not limited to, radiotherapeutic agents and factors; surgery; antibiotics such as doxorubicin, daunorubicin, mitomycin, actinomycin D, and bleomycin; chemotherapeutic agents such as cisplatin, VP16, adriamycin, verapamil, and podophyl
- cancer therapies for TNBC which may be used in conjuntion to siRNAs disclosed herein include, but are not limited to, standard anthracycline- and/or taxanebased chemotherapy.
- Additional therapies for TNBC which may be used alone and/or with anthracycline and/or taxane chemotherapeutic regimens, include: a) chemotherapy treatments with carboplatin, capecitabine and cyclophosphamide; b) anti-androgen receptor (AR) therapies using bicalutamide or enzalutamide; c) treatment with the anti-PD-I1 antibodies nivolumab, pembrolizumab, atezolizumab, avelumab and durvalumab; d) endocrine therapy for estrogen receptor-beta-positive TNBC, using toremifene or anastrozole; e) immunotherapy with the PVX-410 multipeptide vaccine; treatment with: f) the anti-EGF-R antibody cetuximab;
- the other compounds and/or therapies may optionally be administered concurrently or sequentially.
- the active agents may be mixed prior to administration or delivery, or may be administered or delivered at the same point in time but at different anatomic sites and/or by using different routes of administration.
- the siRNA is delivered as a single-agent therapy to treat the TNBC.
- a "single-agent therapy,” as used herein, is one in which no other agent or therapy is utilized to treat the TNBC or to sensitize the cancer cell to the siRNA, i.e., the siRNA, is administered or delivered as a single therapeutic or agent to treat the TNBC.
- the siRNA is delivered as a single-agent therapy in the first-line therapeutic approach.
- first-line therapeutic approach “first-line therapy” and grammatical variations thereof, as used herein, refer to a therapeutic utilized in the initial treatment of a disease or disorder.
- the first-line therapeutic approach as used herein is not limited to single-agent therapies, but may also apply to combination therapies.
- the siRNAs are utilized as a first-line therapy for the initial treatment of cancer, wherein the siRNAs are delivered as single-agent therapy or as a combination therapy.
- the siRNAs are utilized as a therapeutic in the second-line therapeutic approach or in any subsequent therapeutic approach.
- the second-line therapeutic approach and any subsequent therapeutic approaches refer to therapeutic approaches after the initial therapeutic approach, i.e., the first-line therapeutic approach. These approaches may be the same as or different from the first-line therapeutic approach and may comprise a single-agent therapy or a combination therapy.
- the present invention also relates to vectors expressing the nucleic acids of the invention, and cells comprising such vectors or the nucleic acids.
- the cell may be a mammalian cell in vivo or in culture, such as a human cell.
- the invention further relates to compositions comprising the RNAi constructs, and a pharmaceutically acceptable carrier or diluent.
- Another aspect of the invention provides a method for inhibiting the expression of a target gene in a mammalian cell, comprising contacting the mammalian cell with any of the RNAi constructs. The method may be carried out in vitro, ex vivo, or in vivo.
- the target cells e.g., breast cancer cells
- siRNAs of the present invention are targeted to a neoplasm or neoplastic cells of epithelial origin.
- the neoplasm of epithelial origin is breast cancer.
- breast cancer is triple negative breast cancer (TNBC).
- Treatment is defined as the application or administration of a therapeutic agent (e.g., a RNAi agent or vector or transgene encoding same) to a patient, or application or administration of a therapeutic agent to an isolated tissue or cell line from a patient, who has the disease or disorder, a symptom of disease or disorder or a predisposition toward a disease or disorder, with the purpose to cure, heal, alleviate, relieve, alter, remedy, ameliorate, improve or affect the disease or disorder, the symptoms of the disease or disorder, or the predisposition toward disease.
- a therapeutic agent e.g., a RNAi agent or vector or transgene encoding same
- Treatment of cells, or contacting cells can be performed in vitro (e.g., by culturing the cell with the agent), in vivo (e.g., by administering the agent to a subject), or ex vivo (e.g., by administering the agent to cells removed from a subject).
- cells are isolated from an animal (e.g., a human), transformed (i.e., transduced or transfected in vitro) with a delivery vehicle containing the siRNA and then administered to a recipient.
- a delivery vehicle containing the siRNA i.e., a delivery vehicle containing the siRNA
- Procedures for removing cells from mammals are well known to those of ordinary skill in the art.
- tissue or (whole or parts of) organs may be removed, treated ex vivo and then returned to the patient.
- cells, tissue or organs may be cultured, bathed, perfused and the like under conditions for introducing the siRNAs of the invention into the desired cells.
- in vivo treatment cells of a subject are transformed in vivo with the siRNAs of the invention.
- the in vivo treatment may involve, but is not limited to, systemic intravenous treatment, local internal treatment such as by localized perfusion or topical treatment, and the like.
- the siRNAs are delivered to the cell or subject by injection.
- the injection e.g., needle injection
- the injection may comprise one or more injections and can be, for example, subcutaneous, intradermal, intramuscular, intervenous, intraperitoneal, intrathecal, and/or intratumor.
- Methods of injection are well known to those of ordinary skill in the art (e.g., injection of a composition comprising a saline solution).
- Further embodiments of the present invention include the introduction of the siRNAs by direct microinjection.
- Chemically modified siRNAs of the present invention can be administered by any known delivery method, including local (intratumoral) or systemic routes.
- the siRNAs are delivered directly to the neoplasm, for example, by injection using a needle and syringe. Injection into the neoplasm permits large quantities of the siRNAs to be delivered directly to the target cells while minimizing delivery to systemic sites. By direct injection into the neoplasm, an effective amount to promote RNA interference by the siRNAs is distributed throughout at least a substantial volume of the neoplasm. In some instances, delivery of the siRNAs requires a single injection into the neoplasm. In other instances, delivery of the siRNAs requires multiple injections into separate regions of the neoplasm such that the entire mass of the neoplasm is invested with an effective amount to promote RNA interference by the siRNAs. See U.S. Pat. Nos. 5,162,115 and 5,051,257 , each of which is incorporated herein by reference.
- the total dose, concentration, volume of the siRNAs delivered, and rate of delivery can be optimized for a given neoplasm type, size and architecture.
- the zone of RNA interference can be controlled by optimizing these parameters.
- the volume and concentration of the siRNAs delivered into the neoplasm must be sufficient to promote RNA interference throughout the tumor. Depending on the number of injections, and their placement with respect to neoplasm architecture, it can be useful to administer total siRNAs volumes less than the neoplasm volume, greater than the neoplasm volume, or approximately equal to the neoplasm volume.
- the siRNAs are delivered directly to the neoplasm using an implantable device.
- siRNAs injection into a neoplasm can be accompanied by ultrasound guidance.
- the siRNAs are administered systemically, for example, intravenously, intraarterially, intramuscularly, or subcutaneously.
- siRNA to brain tumors. Any appropriate delivery mechanism for delivering an siRNA to the brain or spinal cord can be applied. In some embodiments, delivery to the brain or spinal cord occurs by infusion, intrathecal delivery, parenchymal delivery, intravenous delivery or direct injection into the brain or spinal cord. In some embodiments, an siRNA is delivered to a specific region of the brain. An siRNA can be modified or formulated appropriately to pass the blood-brain barrier. In other embodiments, an siRNA is administered in such a way that it does not need to cross the blood-brain barrier.
- an "effective amount" of an oligonucleotide of the present invention is defined as an amount, at dosages and for periods of time necessary to achieve the desired result.
- an effective amount of an oligonucleotide may vary according to factors such as the type of cell, the oligonucleotide used, and for in vivo uses the disease state, age, sex, and weight of the individual, and the ability of the oligonucleotide to elicit a desired response in the individual.
- Establishment of therapeutic levels of oligonucleotides within the cell is dependent upon the rates of uptake and efflux or degradation. Decreasing the degree of degradation prolongs the intracellular half-life of the oligonucleotide.
- chemically-modified oligonucleotides e.g., with modification of the phosphate backbone, may require different dosing.
- oligonucleotide and number of doses administered will depend upon the data generated experimentally and in clinical trials. Several factors such as the desired effect, the delivery vehicle, disease indication, and the route of administration, will affect the dosage. Dosages can be readily determined by one of ordinary skill in the art and formulated into the subject pharmaceutical compositions. Preferably, the duration of treatment will extend at least through the course of the disease symptoms.
- the initial pharmaceutically effective amount of the active compound or composition administered parenterally may be in the range of about 0.1 to 50 mg/kg of patient body weight per day.
- the desired dosage can be delivered by a single bolus administration, by multiple bolus administrations, by continuous infusion administration, or by intermittent infusion administration of active compound, depending on the pattern of pharmacokinetic decay that the practitioner wishes to achieve.
- the active compound(s) is/are administered to the patient at one time or over a series of treatments. Depending on the type and severity of the disease, about 1 mg/kg to 15 mg/kg of active compound(s) is an initial candidate dosage for administration to the patient.
- a typical daily dosage might range from about 0.1, 0.5, 1, 10 or 100 micrograms/kg up to 100, 200 or 500 mg/kg, or more, including any intermediate values, depending on the factors mentioned above.
- a more particular dosage of the active compound will be in the range from about 0.05 mg/kg to about 10 mg/kg.
- one or more doses of about 0.5 mg/kg, 2.0 mg/kg, 4.0 mg/kg or 10 mg/kg (or any combination thereof) may be administered to the patient.
- Such doses may be administered intermittently, e.g. every week or every three weeks (e.g., such that the patient receives from about two to about twenty, e.g. about six doses of siRNA).
- An initial higher loading dose, followed by one or more lower doses may be administered.
- An exemplary dosing regimen comprises administering an initial loading dose of about 0.5 to 10 mg/kg, followed by a weekly maintenance dose of about 0.5 to 10 mg/kg of the active compound.
- an initial loading dose of about 0.5 to 10 mg/kg
- a weekly maintenance dose of about 0.5 to 10 mg/kg of the active compound.
- Other dosage regimens may be useful.
- the progress of this therapy can be monitored by conventional techniques and assays.
- Subjects treated by the methods of the present invention can also be administered one or more additional therapeutic agents.
- Chemotherapeutic agents may be administered by methods well known to the skilled practitioner, including systemically, direct injection into the cancer, or by localization at the site of the cancer by associating the desired chemotherapeutic agent with an appropriate slow release material or intra-arterial perfusing of the tumor.
- the preferred dose may be chosen by the practitioner based on the nature of the cancer to be treated, and other factors routinely considered in administering.
- Subjects may also be treated by radiation therapy, including, but not limited to, external beam radiotherapy, which may be at any suitable dose (e.g., 20 to 70 Gy or more per tumor, typically delivered over a fractionated schedule).
- external beam radiotherapy may be at any suitable dose (e.g., 20 to 70 Gy or more per tumor, typically delivered over a fractionated schedule).
- the diagnostic marker is alternative transcript variant 3 (SpT3), which encodes isoform c of ErbB-2, having a MW of 165 kDa on SDS-PAGE (p165ErbB-2).
- sample refers to a material known or suspected of expressing or containing one or more polynucleotide or polypeptide cancer markers.
- the diagnostic sample may be any cell, tissue or organ and may be removed by standard biopsy. Exemplary tissues for use in methods described herein include, but are not limited to, blood, bone, brain tissue, endometrial tissue, kidney tissue, stomach tissue, mammary tissue, mammary gland tissue, muscle tissue, nervous tissue and soft tissue.
- the diagnostic sample may also be a bodily fluid, including, but not limited to, cerebrospinal fluid, pericardial fluid, peritoneal fluid, saliva, serum, and urine.
- the diagnostic method disclosed herein is based on the ability of a siRNA molecular beacon (SpT3 MB) to recognize the unique mRNA sequence at the 5' UTR and 5' coding region of SpT3, the transcript variant encoding the isoform c of ErbB-2, which displays a MW of 165kDa (p165ErbB-2).
- SpT3 MB siRNA molecular beacon
- targeted siRNA molecules including, but not limited to, those of the present invention, comprising the sequences depicted in SEQ ID NO: 1 or SEQ ID NO: 2.
- a binary MB approach may also be used consisting of the simultaneous contact of the diagnostic sample with both the SpT3 MB displaying SEQ ID NO: 1 and the SpT3 MB displaying SEQ ID NO: 2.
- the SpT3 MB is designed to have a stem-loop structure, with the nucleotides in the stem complementary to each other forming a four to seven base pairs double stranded stem and the loop consisting of SEQ ID NO: 1 or SEQ ID NO: 2. Furthermore, conjugated to the 5' and 3' ends of this molecular beacon are a fluorophore and a quencher, respectively. Replacement of the organic fluorophores with other brighter signaling materials, such as quantum dots and fluorescent polymers is also considered. Chemical modifications to promote metabolic stability and improve target cell penetration may be introduced to the SpT3 MBs, including but not limited to 2'OMe-modified siRNAs.
- the diagnostic method disclosed in the present invention enables the detection of gene expression in fixed as well as viable cells, thus allowing identification of the expression of SpT3 in clinical samples.
- the diagnostic method disclosed herein concerns an RNA in situ hybridization (ISH) assay for determining the presence of alternative ErbB-2 transcripts (e.g., SpT3).
- ISH RNA in situ hybridization
- the aforementioned ISH assay is an RNAscope ® assay.
- RNAscope ® platform presents a unique probe design strategy that allows simultaneous signal amplification and background suppression to achieve single-molecule visualization while preserving tissue morphology.
- RNAscope ® is compatible with routine formalin-fixed, paraffin embedded (PE) tissue specimens and can use either conventional chromogenic dyes for bright-field microscopy or fluorescent dyes for multiplex analysis (Wang, F. et al. (2012) op. cit.).
- Target probes for the RNAscope ® assay displaying, but not limited to, the sequences depicted in SEQ ID NO: 1 or SEQ ID NO: 2 may be designed to specifically recognize a unique mRNA sequence at the 5' UTR and 5' coding region of SpT3, the transcript variant encoding ErbB-2 isoform c which displays a MW of 165 kDa (p165ErbB-2).
- SpT3 ErbB-2 alternative transcript variant 3
- a series of target probes to hybridize SpT3 may be designed according to methods described in published U.S. Patent Nos. 7,709,198 ; 8,604,182 ; 8,658,361 ; 8,951,726 ; 7,709,198 ; 8,658,361 and 7,709,198 .
- each target probe may contain an 18 to 25-base region complementary to the target mRNA (e.g., SpT3), a spacer sequence, and a 14-base tail sequence (conceptualized as Z).
- a pair of target probes (double Z), each possessing a different type of tail sequence may hybridize contiguously to a target region (-50 bases).
- the two tail sequences together form a 28-base hybridization site for the preamplifier, which contains 20 binding sites for the amplifier, which, in turn, contains 20 binding sites for the label probe.
- a 1 kb region on the RNA molecule could be targeted by 20 probe pairs; thus, sequential hybridizations with the preamplifier, amplifier, and label probe can theoretically yield up to 8000 labels for each target RNA molecule.
- the label probe can be either fluorescently labeled for direct visualization under an epifluorescent microscope or conjugated to an alkaline phosphatase or horseradish peroxidase (HRP) molecule for chromogenic reactions (Fast Red with alkaline phosphatase and 3,3'-diaminobenzidine (DAB) with HRP).
- HRP horseradish peroxidase
- the alkaline phosphatase or HRP-labeled probes have an added advantage, in that chromogen-stained slides can be viewed under a standard bright-field microscope similar to immunohistochemistry (IHC) procedures, making RNAscope ® assay results easier to read and archive in a clinical setting.
- the target probes for different ErbB-2 transcripts could have the same tail sequence recognized by the same signal amplification system, generating a pooled signal; alternatively, multiple signal amplification systems with different label probes can be used to detect each mRNA species, allowing for multiplex detection of multiple target RNAs.
- Hybridization chromogenic detection and estimation of mRNA copy number may be performed as described (Wang, F. et al. (2012), op. cit.).
- ErbB-2 isoform c encoded by SpT3, is the major contributor to the p165ErbB-2 protein isoform.
- ErbB-2 was placed in a new and unanticipated scenario, i.e., the nucleus of TNBC. Also, it was revealed that alternative ErbB-2 transcript 3 (SpT3), encoding ErbB-2 isoform c, is the major oncogenic driver of TNBC proliferation. The authors' clinical findings also identified nuclear ErbB-2 as a biomarker of poor clinical outcome in TNBC.
- TNBC needs to be further subclassified as either nuclear ErbB-2-positive or nuclear ErbB-2-negative.
- nuclear wild-type (WT) ErbB-2 p185ErbB-2, WTErbB-22
- WT nuclear wild-type
- p185ErbB-2, WTErbB-2 p185ErbB-2, WTErbB-2
- WTErbB-2 nuclear wild-type ErbB-2
- WTErbB-2 nuclear wild-type
- the nuclear ErbB-2 isoform c is postulated as a novel common biomarker and target of therapy in BL and M TNBC subtypes.
- Example 1 - NErbB-2 correlates with poor prognosis in TNBC
- Kaplan-Meier analysis revealed that TNBC patients bearing NErbB-2-positive tumors showed significantly shorter overall survival (OS) and disease-free survival (DFS) compared to patients whose tumors lacked NErbB-2 ( FIG. 2A and FIG. 2B ).
- OS overall survival
- DFS disease-free survival
- LRFS Local relapse-free survival
- DMFS distant metastasis-free survival
- FIG. 2C and FIG. 2D Univariate analysis revealed that NErbB-2 and higher clinical stage were associated with lower OS, DFS and DMFS (see Table 4 below). Higher lymph node status was also associated with shorter OS and DMFS.
- OS overall survival
- DFS disease-free survival
- DMFS distant metastasis-free survival
- the present inventors identified nuclear ErbB-2 as a biomarker of poor clinical outcome in TNBC.
- the present inventors explored the presence and activation state of ErbB-2 in TNBC cell lines from all four TNBC molecular subtypes (TNBC-4type). While ErbB-2 expression was detected in cell lysates from all lines, using the C-18 antibody, ErbB-2 variants of different molecular weight (MW) where found among subtypes. Comparable to control BT-474 cells, from the ErbB-2-enriched (ErbB-2E) intrinsic BC subtype, MDA-MB-453 cells (LAR subtype) express wild-type (WT) ErbB-2 (MW of 185 kDa, p185ErbB-2) ( FIG. 3A ).
- MDA-MB-468 cells (BL1 subtype) express only an isoform with a SDS-PAGE MW of 165 kDa (p165ErbB-2) ( FIG. 3A ).
- TNBC lines express significantly lower p185ErbB-2 levels than those in BT-474 cells ( FIG. 3A and FIG. 3B ).
- Levels of p165ErbB-2 isoform in MDA-MB-468, HCC-70 and MDA-MB-231 cells were also lower than those of p185ErbB-2 in BT-474 cells ( FIG. 3A and FIG. 3B ).
- Proteolytic cleavage and alternative initiation of translation of ErbB-2 result in carboxy-terminal fragments from 90 to 115 kDa in BC cells and tumors, collectively referred to as p95ErbB-2/CTFs variants ( Anido, J. et al. (2006) Biosynthesis of tumorigenic HER2 C-terminal fragments by alternative initiation of translation. EMBO J 25, 3234-3244 ), which are associated with nodal metastasis, resistance to anti-MErbB-2 and to endocrine therapies (Anido, J. et al. (2006) op. cit. ; Scaltriti, M. et al.
- NErbB-2 is phosphorylated at tyrosine (Tyr) 877 (Beguelin, W. et al. (2010) op. cit. ; Cordo Russo, R.I. et al. (2015) op. cit.), a site different from the autophosphorylation ones, located at the kinase domain ( Guo, W. et al.
- Beta 4 integrin amplifies ErbB2 signaling to promote mammary tumorigenesis.
- Cell 126, 489-502 Xu, W. et al. (2007) Loss of Hsp90 association upregulates Src-dependent ErbB2 activity. MolCell Biol 27, 220-228 ).
- This phosphorylation appears to be mandatory for ErbB-2 nuclear migration in said cells (Beguelin, W. et al. (2010) op. cit. ; Cordo Russo, R.I. et al. (2015) op. cit.).
- ErbB-2 Tyr877 phosphorylation was found in all TNBC cells at both p165 and p185 ErbB-2 ( FIG. 3I ).
- NErbB-2 presence in TNBC cells by immunofluorescence (IF) and confocal microscopy with the C-18 antibody.
- TNBC cells also display very low to moderate levels of MErbB-2 staining ( FIG. 4A and FIG. 4B ).
- Subcellular fractionation and immunoblotting studies, using C-18 antibody showed that the major ErbB-2 nuclear isoform reflects its abundance in each cell type ( FIG. 4D ).
- the extracellular domain of ErbB-2 displays 7 putative sites for N-glycosylation ( FIG. 5A ), 5 of which were validated in BC cells ( Frei, A.P. et al. (2012) Direct identification of ligand-receptor interactions on living cells and tissues. Nature biotechnology 30, 997-1001 ; Watanabe, M. et al. (2013) Improvement of mass spectrometry analysis of glycoproteins by MALDI-MS using 3-aminoquinoline/alpha-cyano-4-hydroxycinnamic acid. Analytical and bioanalytical chemistry 405, 4289-4293 ). This posttranslational modification alters the migration pattern of proteins on SDS-PAGE.
- SpTs spliced transcripts
- SpPs protein isoforms
- Alternative transcript variant 3 SpT3 is the longest one ( FIG. 6A ).
- Transcript variant 2 SpT2
- TSS transcriptional start site
- SpT3 differs from SpT3 in that it excludes the exon 5 by alternative splicing ( FIG. 6A ).
- the transcriptional unit of ERBB2 has another TSS, downstream of the one that generates SpT2 and SpT3, from which other ErbB-2 transcripts are generated, among them, SpT1 and SpT4 ( FIG. 6B ).
- SpT1 is considered the canonical transcript which translates into p185ErbB-2 WTErbB-2, herein referred as isoform a ( FIG. 7 ).
- SpT4 lacks the penultimate exon (26, considering the order of exons in SpT1), and uses an alternative 3' splice site in the last exon (27, according to the order in SpT1) ( FIG. 6B ).
- the protein encoded by SpT4 has a shorter C-terminus compared to isoform a (and FIG. 7 ).
- translation of SpT2 and SpT3 give rise to proteins (isoforms b and c, respectively) with the same C-termini as isoform a, but with shorter N-termini, lacking the signal peptide (SP) ( FIG. 7 and FIG. 8 ).
- SP signal peptide
- ErbB-2 ⁇ 16 an alternative-spliced ErbB-2 isoform (ErbB-2 ⁇ 16) was detected in BC and associated with high oncogenic and metastatic potential ( Castiglioni, F. et al. (2006) Role of exon-16-deleted HER2 in breast carcinomas. Endocr Relat Cancer 13, 221-232 ; Kwong, K.Y. and Hung, M.C. (1998) A novel splice variant of HER2 with increased transformation activity. Mol Carcinog 23, 62-68 ; Mitra, D. et al. (2009) An oncogenic isoform of HER2 associated with locally disseminated breast cancer and trastuzumab resistance. Molecular cancer therapeutics 8, 2152-2162 ; Turpin, J.
- ErbB2DeltaEx16 splice variant is a major oncogenic driver in breast cancer that promotes a pro-metastatic tumor microenvironment. Oncogene 35, 6053-6064 ).
- ErbB-2 ⁇ 16 lacks exon 16 (considering the order of exons in SpT1) ( FIG. 6C ). The exclusion of this exon results in an in-frame deletion of 16 amino acids (aa) at the juxtamembrane domain ( FIG. 7 ). There is no mRNA sequence of ErbB-2 ⁇ 16 logged to any publicly available databases.
- ErbB-2 ⁇ 16 protein runs slightly faster than p185ErbB-2 on SDS-PAGE, in a position comparable to that of p165ErbB-2, its MW was not disclosed. Therefore, the inventors also investigated whether ErbB-2 ⁇ 16 protein contributes to the p165ErbB-2 band detected by WB. ErbB-2 C- and N-terminal antibodies would recognize ErbB-2 ⁇ 16, which displays intact Cand N-terminus (see Table 6 below). TABLE 6. Anti-ErbB-2 antibodies used in this study. Target amino acids sequences (aa) corresponding to the amino- and carboxy-terminus of all ErbB-2 isoforms are indicated.
- Anti-ErbB-2 antibody ErbB-2 isoform ErbB-2 C-18 aa ErbB-2 A-2 aa ErbB-2 C-3 aa Isoform a 1248-1255 1180-1197 251-450 Isoform b 1218-1225 1150-1167 221-420 Isoform c 1233-1240 1165-1182 236-435 Isoform d - - 251-450 ErbB-2 ⁇ 16 1232-1239 1164-1181 251-450
- PCR polymerase chain reaction
- RNA from all cells was reverse transcribed. Due to the high sequence homology among ErbB-2 transcripts, the first strategy used by the present inventors was to use cDNA for long-range (LR)-PCR. Two sets of primers were used: one of them amplifies the entire coding region of transcripts 1, 4, and ErbB-2A16 (LR-PCR1: forward primer SEQ ID NO: 4; reverse primer SEQ ID NO: 5) while the other, amplifies the entire coding region of transcripts 2 and 3 (LR-PCR2: forward primer SEQ ID NO: 6; reverse primer SEQ ID NO: 5).
- Forward primer for LR-PCR1 (SEQ ID NO: 4) anneal to the common 5' end of SpT1, SpT4 and ErbB-2 ⁇ 16.
- Forward primer for LR-PCR2 (SEQ ID NO: 6) anneal to the common 5' end of SpT2 and SpT3.
- the same reverse primer (SEQ ID NO: 5), which anneals to the common 3' end of all five transcripts, was used for both LR-PCRs.
- each of the LR-PCR products were purified and used as templates for nested PCR with variant specific primers for SpTs 1, 2 or 3.
- the inventors designed forward and reverse primers annealing to exons 16 and 26, respectively.
- the inventors designed two set of primers spanning their differential region between exons 1 to 5 (additional exons upstream of the canonical exon 1).
- SpT1 was detected in all cell lines where expression of p185ErbB-2 (WT ErbB-2) was found, but not in TNBC MDA-MB-468 cells, which only express the p165ErbB-2 isoform ( FIG. 10A ).
- Direct Sanger sequencing in both directions of the PCR products confirmed SpT1 identity in BT-474 (SEQ IDs 34 and 35) and HCC-70 cells (SEQ IDs 36 and 37).
- SpT2 and SpT3 were present in all TNBC lines and in BT-474 cells ( FIGS. 10B and 10C ). Sequencing of the corresponding amplicons verified transcript identities.
- cDNAs from all cell lines were used for competitive PCR to quantify the relative abundance of SpT4 in comparison to those of variants that include exon 26 using primers that amplify the region between exons 25 and 27.
- All cell lines express SpT4 which represents 4 to 6% of the total ErbB-2 mRNA ( FIGS. 12A and 12C ).
- the inventors also performed competitive PCR to quantify the relative levels of ErbB-2 ⁇ 16 in comparison to those of variants that include exon 16 using primers spanning exons 15 and 17.
- ErbB-2 ⁇ 16 transcript accounts for 9 to 12% of total ErbB-2 mRNA ( FIGS. 12B and 12D ).
- the inventors studied the expression levels of SpT3 by reverse transcriptase (RT)-quantitative PCR (qPCR) in BT-474 and MDA-MB-468 cells. Consistent with their findings of lower total ErbB-2 message in TNBC as compared with ErbB-2E cells ( FIG. 13 ), SpT3 expression was decreased in MDA-MB-468 cells with respect to BT-474 ( FIG. 14A ). However, the ratio of SpT3/total ErbB-2 was higher in MDA-MB-468 cells ( FIG. 14B ).
- RT reverse transcriptase
- qPCR quantitative PCR
- Example 4 Nuclear ErbB-2 isoform c induces proliferation of TNBC of basal and mesenchymal subtypes
- ErbB-2 siRNA To further dissect the identity of the p165ErbB-2 protein, a pool of 4 different siRNAs (siGENOME SMARTpool Human ERBB2, Dharmacon, Lafayette, CO, USA), herein referred as ErbB-2 siRNA ( FIG.15A ) was used. These siRNAs target the common coding region of transcript variants 1 to 4, and ErbB-2 ⁇ 16. Unexpectedly, these series of siRNAs silenced only p185ErbB-2 but were ineffective to knockdown p165ErbB-2 ( FIGS. 15B-15D ). Levels of ErbB-2 mRNA were silenced in cells expressing p185ErbB-2 ( FIG. 15G ) or both p185 and p165ErbB-2 ( FIG. 15F ), but remained unaffected in cells which only express p165ErbB-2 ( FIG. 15E ).
- FIGS. 17A and 17E blocked in vitro proliferation of cells that only express p185ErbB-2 ( FIGS. 17A and 17E ) but were unable to inhibit proliferation of TNBC cells which express p165ErbB-2 ( FIG. 17B ) or both p165 and p185ErbB-2 ( FIGS. 17C and 17D ).
- SpTs 2 and 3 were identified as the main contributors to the p165ErbB-2 protein. However, since the secondary structure of said variants remains unknown, the use of total ErbB-2 siRNAs was possibly inappropriate to target them. As shown in FIGS. 6A-6C , SpT3 includes a specific exon (5, according to the order of exons in SpT3), spanning its 5' UTR and 5' coding region from nucleotides (nt) 560 to 610, which is neither present in any of the other transcripts here studied.
- At least two optimized siRNAs that have been selected according to the aforementioned algorithms are used to silence a gene, more preferably at least three and most preferably at least four.
- the siRNAs may be used individually or together in a pool or kit. Further, they may be applied to a cell simultaneously or separately. Preferably, the at least two siRNAs are applied simultaneously. Pools are particularly beneficial for many research applications. However, for therapeutics, it may be more desirable to employ a single siRNA.
- siDESIGN Center was used for identifying functional siRNAs against ErbB-2 alternative transcript variant 3 (SpT3) (SEQ ID NO: 32), which encodes the non-canonical isoform c (SEQ ID NO: 33).
- siRNA molecules include, but are not limited to those having the sequences depicted in SEQ ID NO: 1 and SEQ ID NO: 2.
- any other sequence selection algorithm or method may be used for optimizing siRNAs for specific targets (e.g., ERBB2 alternative transcript variant 3, NM_001289936, SEQ ID NO: 32).
- the first step of siDESIGN Center comprises selecting the target nucleotide sequence for evaluation by the siDESIGN algorithm.
- Target Gene Variants Table with the Accession number(s) for all known RefSeq variants associated with the target gene (e.g., ERBB2 human) was displayed. Specific variant(s) were selected in the Target Gene Variants Table (e.g., ERBB2 alternative transcript variant 3, NM_001289936) and targeted by resulting siRNA designs.
- target mRNA i.e., 5' UTR, open reading frame (ORF), 3' UTR
- BLAST analysis was also considered to further enhance target specificity.
- candidate siRNA design regions are also selected by the siDESIGN algorithm with other important criteria.
- Several of these key criteria include the exclusion of: i) motifs correlated with toxicity; ii) known miRNA seed region motifs and iii) known single nucleotide polymorphisms (SNPs).
- SNPs single nucleotide polymorphisms
- BLAST analysis revealed that both siRNAs were highly sequence specific (100% sequence identity between each siRNA and SpT3), targeting SpT3 mRNA at positions 555-573 (SpT3 siRNA #1, SEQ ID NO: 1) and 566-584 (SpT3 siRNA #2, SEQ ID NO: 2) ( FIG. 18A ).
- Example 5 - SpT3 siRNA #2 of the present invention abrogates tumor growth in TNBC
- the present inventors evaluated the effect of silencing in triple negative tumors the expression of p165ErbB-2 (isoform c) with the siRNA #2. They used an ex vivo methodology to culture MDA-MB-468 human tumor xenografts. This procedure is a modified version of a previously reported technique ( Centenera, M.M. et al. (2012) Evidence for efficacy of new Hsp90 inhibitors revealed by ex vivo culture of human prostate tumors. Clin Cancer Res 18, 3562-3570 ; Dean, J.L. et al. (2012) Therapeutic response to CDK4/6 inhibition in breast cancer defined by ex vivo analyses of human tumors.
- the inventors established a human TN breast tumor explant that allowed to study, on one hand, TNBC response to their invented siRNAs drugs and, on the other, to reveal in the preclinical setting the role of ErbB-2 isoform c as biomarker in TN tumors.
- TNBC tumor necrosis factor
- MDA-MB-468 cells 5 ⁇ 10 6 /mouse
- MDA-MB-468 tumors were established (volumes of 50 ⁇ 70 mm 3 ), they were surgically removed and dissected into approximately 1-mm 3 using a scalpel. Four tumor pieces were placed on a presoaked gelatin sponge (Johnson and Johnson) in 6-well plates containing 1 mL DMEM F12 with 10% FBS. Tumor explants were cultured at 37°C in a 5% CO 2 -enriched atmosphere for 6 hours.
- tumor explants were treated for 24 hours with SpT3 siRNA #2 (SEQ ID NO: 2), or Control siRNA (SEQ ID NO: 25) at a final concentration of 100 nM, by using DharmaFECT transfection reagent (Dharmacon) according to the manufacturer's instructions.
- DharmaFECT transfection reagent Dharmacon
- cultured tumor explants were lysed in ice-cold radioimmunoprecipitation assay buffer (10 mmol/L Tris-HCI, 150 mmol/L NaCl, 1 mmol/L EDTA, 1% Triton X-100) containing protease inhibitor cocktail (Sigma), using the Precellys24 tissue homogenizer (Bertin Technologies).
- Example 6 - SpT3 siRNA #2 of the present invention abrogates TNBC growth in vivo
- tumor xenografts were developed in nude mice using MDA-MB-468 cells. Mice received intratumoral injections of SpT3 siRNA #2 (SEQ ID NO: 2), or Control siRNA (SEQ ID NO: 31), thrice a week. Tumor volume was measured as a function of time (days) in the xenografts generated for this assay ( FIG. 23A and Table 8 below). TABLE 8. Tumor growth analysis of MDA-MB-468 xenografts treated with either SpT3 siRNA #2 (SEQ ID NO: 2), or Control siRNA (SEQ ID NO: 31). Mean tumor volume (mm 3 ) ⁇ S.D.
- ErbB-2 isoform c encoded by SpT3 and displaying a SDS-PAGE MW of 165 kDa, is located in the nucleus and drives in vitro and in vivo growth of BL and M TNBC subtypes. Accordingly, SpT3 siRNA #2 of the invention proves to be an excellent therapy for TNBC tumors.
- DFS disease-free survival
- DMFS distant metastasis-free survival
- LRFS local relapse-free survival
- DMFS and LRFS were defined as the time from BC diagnosis to the first recording of a distant metastasis or a local recurrence, respectively.
- Local relapse was defined as recurrences of BC occurring in the ipsilateral breast, regional lymph nodes, and skin from the breast.
- Distant relapse was defined as recurrences of BC occurring beyond the confines of the ipsilateral breast, chest wall, or regional lymph nodes.
- Sites of distant relapse included: brain (and central nervous system), liver, lung, bone, pleural/peritoneal, and supraclavicular nodes.
- the secondary endpoint was the overall survival (OS).
- Pre-treatment patient staging was classified according to the American Joint Committee on Cancer (AJCC) system ( Singletary, S.E.
- MDA-MB-468, HCC-70, MDA-MB-231, BT-474, SK-BR-3 and T47D cells were obtained from American Type Culture Collection (Manassas, VA, USA). MDA-MB-453 and HCC-1419 were a gift from DJ Slamon (University of California, Los Angeles, CA, USA). Luciferase-expressing MDA-MB-231 cells (MDA-MB-231-luc) were kindly provided by MC Hung (University of Texas, M.D. Anderson Cancer Center, Houston, TX, USA). BT-474, SK-BR-3 and T47D cells were cultured as described previously (Beguelin, W. et al. (2010) op. cit.
- MDA-MB-453 and HCC-1419 cells were cultured as described by DJ Slamon ( O'Brien, N.A. et al. (2010) Activated phosphoinositide 3-kinase/AKTsignaling confers resistance to trastuzumab but not lapatinib. MolCancer Ther 9, 1489-1502 ).
- MDA-MB-468, HCC-70 and MDA-MB-231 were maintained according to the supplier's instructions.
- MDA-MB-231-luc cells were cultured as described by MC Hung ( Xie, X. et al. (2012) Targeted expression of BikDD eliminates breast cancer with virtually no toxicity in noninvasive imaging models. Molecular cancer therapeutics 11, 1915-1924 ).
- rabbit polyclonal anti-ErbB-2 clone C-18 (sc-284, raised against the C-terminus), mouse monoclonal anti-ErbB-2 clone A-2 (sc-393712, raised against the C-terminus), mouse monoclonal anti-ErbB-2 clone C-3 (sc-377344, raised against the N-terminus), and rabbit polyclonal anti-Histone H3 clone C-16 (sc-8654-R), all from Santa Cruz Biotechnology; rabbit polyclonal anti-pErbB-2 Tyr877 (2241) from Cell Signaling Technology; mouse monoclonal anti- ⁇ -Tubulin clone T0198 (T0198) from Sigma-Aldrich; rabbit monoclonal anti-Erk5 clone EP791Y (ab40809) from Abcam; and HRP-conjugated secondary antibodies from Vector Laboratories.
- the antibodies used for immunofluorescence (IF) and confocal microscopy were anti-ErbB-2 C-18, anti-ErbB-2 A-2, anti-ErbB-2 C-3, and mouse monoclonal anti-green fluorescence protein (GFP) clone B-2 (SC-9996), all from Santa Cruz Biotechnology, and Alexa Fluor-conjugated secondary antibodies from Invitrogen.
- the antibodies used for immunohistochemistry (IHC) were anti-ErbB-2 C-18 and anti-ErbB-2 A-2, from Santa Cruz Biotechnology.
- Total lysates were obtained from cells growing in complete media or subjected to the different treatments. 10-50 ⁇ g of lysates were separated on a 6-12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), transferred to a nitrocellulose membrane and blotted as described (Beguelin, W. et al. (2010) op. cit.) with the antibodies detailed in each experiment. Signal intensities of phospho-ErbB-2 bands were analyzed by densitometry using Image J software (National Institutes of Health) and normalized to total ErbB-2 protein bands. Experiments assessing total protein content were also repeated three to five times and signal intensities were normalized to ⁇ -tubulin bands, used as loading control.
- SDS-PAGE sodium dodecyl sulfate-polyacrylamide gel electrophoresis
- Subcellular protein fractionation kit for cultured cells (78840, Thermo Fisher Scientific Inc., Waltham, MA, USA) was performed in order to obtain cytoplasmic, membrane, and nuclear protein extracts, according to manufacturer's instructions.
- Molecular weight (MW) determination was performed as described ( Guan, Y. et al. (2015) An equation to estimate the difference between theoretically predicted and SDS PAGE-displayed molecular weights for an acidic peptide. Scientific reports 5, 13370 ). Briefly, for each western blot (WB), a MW standard protein ladder (Amersham ECL Rainbow Marker Full range, GE Healthcare) was loaded along with different protein samples.
- an Rf value defined as the migration distance of a protein through the gel divided by the migration distance of the dye front, was calculated for each standard protein.
- the Rf value for each protein sample was then obtained and used for the calculation of the SDS-PAGE-displayed MW of the ErbB-2 protein band.
- Unsupervised hierarchical clustering of BC cells was carried out on the quantification profiles of p185-, p165- and p95-ErbB-2 from WBs. Normalized signal intensities of the different ErbB-2 isoforms were log2 transformed and clustering analysis was performed using the R's heatmap.2 function. The heatmap and associated dendrogram were generated with the Euclidean distance and complete linkage clustering method without additional normalization. Color scale reflects the standardized expression of each isoform with red indicating highest expression and green indicating lowest expression.
- BT-474 and MDA-MB-468 cells were treated for 15 h with 10 ⁇ g/ml tunicamycin (Enzo Life Sciences, Inc.) or dimethyl sulfoxide (DMSO, Merck). Total cells lysates were processed as indicated for WB analysis using the ErbB-2 C-18 antibody. MW calculation was performed as indicated above.
- RT-qPCRs were performed in a StepOne Real Time PCR System (Applied Biosystems) with FastStart Universal SYBR Green Master mix (Roche) using the following cycling conditions: 40 cycles with 15 s of denaturing at 95°C and annealing/extension at 60°C for 1 min.
- Expression analysis of the canonical SpT1 and the transcript variants SpT2 and SpT3 was performed using a long-range PCR (LR-PCR) and a nested PCR approach.
- LR-PCR long-range PCR
- cDNAs from all cell lines were subjected to LR-PCR using two sets of primers, one of them amplifies the entire coding region of SpT1, SpT4, and ErbB-2 ⁇ 16 (LR-PCR1), and the other the entire coding region of SpT2 and SpT3 (LR-PCR2).
- Nested PCR was then performed using first-round LR-PCR products and variant specific primers spanning the differential regions of SpT1, SpT2 and SpT3.
- LR-PCRs were performed with a ready-to-use RANGER Mix 2X (BIOLINE), based on the high-fidelity hot-start RANGER DNA Polymerase. Briefly, after cDNA synthesis, 1 ⁇ l of RT-PCR product was amplified in a reaction mixture containing 25 ⁇ l of manufacturer's supplied buffer which includes RANGER DNA polymerase along with dNTPs and MgCl 2 , 0.3 ⁇ M of each primer, 5% DMSO and nuclease-free water up to 50 ⁇ l.
- LR-PCR reactions were carried out in a Veriti 96 Well Thermal Cycler (Applied Biosystems) under the following conditions: polymerase activation was at 95°C for 3 min followed by 35 cycles of amplification (98°C for 20 s, 55°C for 15 s, 72°C for 5 min) and a final extension step at 72°C for 5 min.
- the LR-PCR products were run on 0.7% agarose gels, visualized by staining with 0.5 ⁇ g/ml ethidium bromide (Invitrogen) and purified with the QIAquick gel extraction kit (QIAGEN) according to manufacturer's instructions.
- LR-PCR1 1 ⁇ l of purified LR-PCR1 product was subjected to nested PCR using RANGER DNA polymerase (BIOLINE) and specific primers as indicated above.
- RANGER DNA polymerase BIOLINE
- purified LR-PCR2 products were used for nested PCR with Taq DNA polymerase (Invitrogen) and variant-specific primers.
- This PCR mixture contained 1 ⁇ l of LR-PCR2 product, 200 ⁇ M dNTPs, 0.5 ⁇ M of each primer, 1.5 mM MgCl 2 , 0.05 U of Taq DNA polymerase in 10X PCR buffer and nuclease-free water up to 20 ⁇ l.
- Nested PCR reactions for SpT2 and SpT3 were also performed in a Veriti 96 Well Thermal Cycler (Applied Biosystems) under the following conditions: a denaturation step at 94°C for 2 min, followed by 30 cycles at 94°C for 30 s, 55°C for 30 s, 72 °C for 40 s, and a final extension step at 72°C for 5 min.
- Expression analysis of SpT4 and ErbB-2 ⁇ 16 was performed by conventional PCR using Taq DNA polymerase (Invitrogen) and cDNAs from all cell lines as templates. Cycling conditions were similar to those used in nested PCR for SpT2 and SpT3, setting the annealing temperature at 60°C.
- SpT3 siRNA #2 or Control siRNA were appropriately diluted in 25 ⁇ l serum free medium (DMEM/F12), and complexed with 1.5 ⁇ l DharmaFECT1 transfection reagent (Dharmacon) previously diluted in 25 ⁇ l DMEM/F12, according to manufacturer's instructions. Mice were sacrificed two days after the last treatment.
- Tumor volumes, growth rates and percentage of growth inhibition were calculated as described ( Proietti, C. et al. (2005) Progestins induce transcriptional activation of signal transducer and activator of transcription 3 (Stat3) via a Jak- and Src-dependent mechanism in breast cancer cells. MolCell Biol 25, 4826-4840 ).
- Stat3 signal transducer and activator of transcription 3
- MolCell Biol 25, 4826-4840 tumor pieces for molecular studies were stored at -80°C. Tumor pieces and whole organ specimens for histopathological examination were fixed in 10% formalin. Blood samples were also collected for performing hematology tests and evaluating serum biochemistry parameters.
- Hematoxylin and eosin (H&E) staining was performed on 5 ⁇ m slide sections of MDA-MB-468 tumors, and used for histopathological examination. Mitotic figure counts were performed in 10 consecutive high power fields (HPF, 400 X magnification) using a Leica DM500 light microscope (0.45 mm diameter of the HPF). The identification of well-defined mitotic figures was performed as previously described ( Baak, J.P. et al. (2005) Prospective multicenter validation of the independent prognostic value of the mitotic activity index in lymph node-negative breast cancer patients younger than 55 years. J Clin Oncol 23, 5993-6001 ; van Diest, P.J. et al.
- Necrotic areas were characterized by cell and nuclear swelling, pale eosinophilic cytoplasm, nuclear dissolution (karyolysis), nuclear fragments (karyorrhexis) and loss of cellular detail with shadows of tumor cells visible to variable extent. Some degree of nuclear condensation (pyknosis) may be present. Adjacent cellular debris and inflammation (neutrophils, macrophages, etc.) may also be present if cell membrane leakage or rupture has occurred.
- ErbB-2 was localized using either a rabbit polyclonal (C-18) or a mouse monoclonal (C-3 or A-2) ErbB-2 antibody (all from Santa Cruz Biotechnology). Secondary antibodies for ErbB-2 C-18 were goat anti-rabbit IgG-Alexa Fluor 488 or donkey anti-rabbit IgG-Alexa Fluor 546. For ErbB-2 C-3 or A-2 a goat anti-mouse IgG-Alexa 488 was used, all from Invitrogen. Negative controls were carried out using PBS instead of primary antibodies. When cells were transfected with hErbB-2 ⁇ NLS, green fluorescent protein (GFP) from this expression vector was visualized by direct fluorescence imaging.
- GFP green fluorescent protein
- cytoplasmic membrane compartment was defined as the difference between the image of the cell and a binary erosion (iterations: 5-25), and the nuclear compartment was defined according to the nuclear stain (DAPI (4',6-diamidino-2-phenylindole) or propidium iodide).
- An integrated density value (mean fluorescence intensity per unit area) was obtained for total ErbB-2 (tErbB-2), for membrane ErbB-2 (mErbB-2), and for nuclear ErbB-2 (nErbB-2).
- tErbB-2 total ErbB-2
- mErbB-2 membrane ErbB-2
- nErbB-2 nuclear ErbB-2
- NErbB-2 ratio of integrated density of nErbB-2/tErbB-2
- MErbB-2 mErbB-2/tErbB-2
- Membrane ErbB-2 (MErbB-2) expression levels detected by IF were semiquantified using the same scores as those used in IHC staining (see below). Nuclear ErbB-2 levels detected by IF were scored considering both the percentage of positive cells and staining intensity. A score of 0 represents faint or no staining in less than 10% of cells, 1+ weak nuclear staining in 10-25%, 2+ moderate staining in 26-50%, and 3+ strong staining in > 50% of cells. Scores of 2+ and 3+ were considered positive for NErbB-2 presence (Schillaci, R. et al. (2012) op. cit.).
- NErbB-2 was also evaluated by IHC with the ErbB-2 C-18 antibody as follows: heat-induced antigen retrieval was performed in 10 mM Tris, 1 mM EDTA pH 9 for 30 min. Slides were incubated with the ErbB-2 C-18 antibody (dilution 1:200) overnight at 4°C. Sections were subsequently incubated with the anti-rabbit EnVision+ System-HRP Labelled Polymer (K4003, Dako, Agilent) and developed using 3,3'-diaminobenzidine chromogen solution (Cell Marque, MilliporeSigma) according to manufacturer's protocol. The score was performed as detailed for IF.
- MErbB-2 expression was evaluated by IHC with c-erb-B2 clone A0485 (Dako), as we already described (Schillaci, R. et al. (2012) op. cit. ) .
- MErbB-2 was scored according to the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guidelines ( Wolff, A.C. et al. (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. JClinOncol 31, 3997-4013 ). Scores 2+ in which FISH confirmed ErbB-2 amplification and 3+ were considered positive for MErbB-2 overexpression.
- Estrogen (ER) and progesterone receptor (PR) were evaluated by IHC with clone 6F11 (Novocastra Laboratories) and clone hPRa2+hPRa3 (NeoMarkers), respectively and scored as described (Schillaci, R. et al. (2012) op. cit.).
- Nuclear epidermal growth factor receptor is a functional molecular target in triple-negative breast cancer. Molecular cancer therapeutics 13, 1356-1368 .
- RNA 13, 431-456 Hydrophobization and bioconjugation for enhanced siRNA delivery and targeting.
- Beta 4 integrin amplifies ErbB2 signaling to promote mammary tumorigenesis. Cell 126, 489-502 .
- Lapatinib a dual EGFR and HER2 tyrosine kinase inhibitor, downregulates thymidylate synthase by inhibiting the nuclear translocation of EGFR and HER2.
- PLoSOne 4, e5933 a dual EGFR and HER2 tyrosine kinase inhibitor
- Prat, A. et al. (2013a) Molecular characterization of basal-like and non-basal-like triple-negative breast cancer. The oncologist 18, 123-133 .
- RNAscope a novel in situ RNA analysis platform for formalinfixed, paraffin-embedded tissues. JMD 14, 22-29 .
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Zoology (AREA)
- Biomedical Technology (AREA)
- Wood Science & Technology (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biochemistry (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Physics & Mathematics (AREA)
- Microbiology (AREA)
- Epidemiology (AREA)
- Immunology (AREA)
- Oncology (AREA)
- Plant Pathology (AREA)
- Analytical Chemistry (AREA)
- Pathology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Hospice & Palliative Care (AREA)
- Dermatology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Gastroenterology & Hepatology (AREA)
- Toxicology (AREA)
- Cell Biology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16/910,481 US11459567B2 (en) | 2020-06-24 | 2020-06-24 | Specific siRNA molecules, composition and use thereof for the treatment of triple negative breast cancer |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP3936136A1 true EP3936136A1 (de) | 2022-01-12 |
Family
ID=76601130
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP21181521.2A Pending EP3936136A1 (de) | 2020-06-24 | 2021-06-24 | Spezifische sirna-moleküle, zusammensetzung und verwendung davon zur behandlung von dreifach negativem brustkrebs |
Country Status (2)
| Country | Link |
|---|---|
| US (1) | US11459567B2 (de) |
| EP (1) | EP3936136A1 (de) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN119139500B (zh) * | 2024-11-14 | 2025-02-18 | 上海交通大学医学院附属仁济医院 | 一种糖核酸分子影像探针及其制备方法和使用方法 |
Citations (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5051257A (en) | 1989-05-09 | 1991-09-24 | Pietronigro Dennis D | Antineoplastic solution and method for treating neoplasms |
| US5162115A (en) | 1989-05-09 | 1992-11-10 | Pietronigro Dennis D | Antineoplastic solution and method for treating neoplasms |
| WO1998013526A1 (en) | 1996-09-26 | 1998-04-02 | Oligos Etc. Inc. | Three component chimeric antisense oligonucleotides |
| EP1527176A2 (de) | 2002-08-05 | 2005-05-04 | atugen AG | Weitere neue formen von interferierende rns moleküle |
| WO2008104978A2 (en) | 2007-02-28 | 2008-09-04 | Quark Pharmaceuticals, Inc. | Novel sirna structures |
| US7709198B2 (en) | 2005-06-20 | 2010-05-04 | Advanced Cell Diagnostics, Inc. | Multiplex detection of nucleic acids |
| US7816512B2 (en) | 2002-11-14 | 2010-10-19 | Dharmacon, Inc. | siRNA targeting proto-oncogene MET |
| US7977471B2 (en) | 2002-11-14 | 2011-07-12 | Dharmacon, Inc. | siRNA targeting TNFα |
| US8008474B2 (en) | 2002-11-14 | 2011-08-30 | Dharmacon, Inc. | siRNA targeting KRAS |
| US8658361B2 (en) | 2010-10-21 | 2014-02-25 | Advanced Cell Diagnostics, Inc. | Ultra sensitive method for in situ detection of nucleic acids |
| US9080171B2 (en) | 2010-03-24 | 2015-07-14 | RXi Parmaceuticals Corporation | Reduced size self-delivering RNAi compounds |
| US20160319278A1 (en) | 2015-04-03 | 2016-11-03 | University Of Massachusetts | Fully stabilized asymmetric sirna |
| US10011836B2 (en) | 2002-11-14 | 2018-07-03 | Thermo Fisher Scientific Inc. | Methods and compositions for selecting siRNA of improved functionality |
| US10138485B2 (en) | 2008-09-22 | 2018-11-27 | Rxi Pharmaceuticals Corporation | Neutral nanotransporters |
| US20190024082A1 (en) | 2017-06-23 | 2019-01-24 | University Of Massachusetts | Two-tailed self-delivering sirna |
| WO2019018841A2 (en) * | 2017-07-21 | 2019-01-24 | The Regents Of The University Of Colorado, A Body Corporate | COMPOSITIONS AND METHODS FOR IDENTIFICATION OF COMBINATORIAL IMMUNO-ONCOLOGIC THERAPEUTIC AGENTS |
| WO2020096248A1 (ko) * | 2018-11-09 | 2020-05-14 | 주식회사 셀레믹스 | 폐암 조직 내 세포 유래 돌연변이를 검출하기 위한 프로브 제조 및 검출 방법 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7148342B2 (en) * | 2002-07-24 | 2006-12-12 | The Trustees Of The University Of Pennyslvania | Compositions and methods for sirna inhibition of angiogenesis |
| US20170044540A1 (en) * | 2014-04-22 | 2017-02-16 | Mina Therapeutics Limited | Sarna compositions and methods of use |
-
2020
- 2020-06-24 US US16/910,481 patent/US11459567B2/en active Active
-
2021
- 2021-06-24 EP EP21181521.2A patent/EP3936136A1/de active Pending
Patent Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5162115A (en) | 1989-05-09 | 1992-11-10 | Pietronigro Dennis D | Antineoplastic solution and method for treating neoplasms |
| US5051257A (en) | 1989-05-09 | 1991-09-24 | Pietronigro Dennis D | Antineoplastic solution and method for treating neoplasms |
| WO1998013526A1 (en) | 1996-09-26 | 1998-04-02 | Oligos Etc. Inc. | Three component chimeric antisense oligonucleotides |
| US5849902A (en) | 1996-09-26 | 1998-12-15 | Oligos Etc. Inc. | Three component chimeric antisense oligonucleotides |
| EP1527176A2 (de) | 2002-08-05 | 2005-05-04 | atugen AG | Weitere neue formen von interferierende rns moleküle |
| US8008474B2 (en) | 2002-11-14 | 2011-08-30 | Dharmacon, Inc. | siRNA targeting KRAS |
| US10011836B2 (en) | 2002-11-14 | 2018-07-03 | Thermo Fisher Scientific Inc. | Methods and compositions for selecting siRNA of improved functionality |
| US7816512B2 (en) | 2002-11-14 | 2010-10-19 | Dharmacon, Inc. | siRNA targeting proto-oncogene MET |
| US7977471B2 (en) | 2002-11-14 | 2011-07-12 | Dharmacon, Inc. | siRNA targeting TNFα |
| US8604182B2 (en) | 2005-06-20 | 2013-12-10 | Advanced Cell Diagnostics, Inc. | Multiplex detection of nucleic acids |
| US7709198B2 (en) | 2005-06-20 | 2010-05-04 | Advanced Cell Diagnostics, Inc. | Multiplex detection of nucleic acids |
| US8951726B2 (en) | 2005-06-20 | 2015-02-10 | Advanced Cell Diagnostics, Inc. | Multiplex detection of nucleic acids |
| WO2008104978A2 (en) | 2007-02-28 | 2008-09-04 | Quark Pharmaceuticals, Inc. | Novel sirna structures |
| US10138485B2 (en) | 2008-09-22 | 2018-11-27 | Rxi Pharmaceuticals Corporation | Neutral nanotransporters |
| US9080171B2 (en) | 2010-03-24 | 2015-07-14 | RXi Parmaceuticals Corporation | Reduced size self-delivering RNAi compounds |
| US8658361B2 (en) | 2010-10-21 | 2014-02-25 | Advanced Cell Diagnostics, Inc. | Ultra sensitive method for in situ detection of nucleic acids |
| US20160319278A1 (en) | 2015-04-03 | 2016-11-03 | University Of Massachusetts | Fully stabilized asymmetric sirna |
| US20190024082A1 (en) | 2017-06-23 | 2019-01-24 | University Of Massachusetts | Two-tailed self-delivering sirna |
| WO2019018841A2 (en) * | 2017-07-21 | 2019-01-24 | The Regents Of The University Of Colorado, A Body Corporate | COMPOSITIONS AND METHODS FOR IDENTIFICATION OF COMBINATORIAL IMMUNO-ONCOLOGIC THERAPEUTIC AGENTS |
| WO2020096248A1 (ko) * | 2018-11-09 | 2020-05-14 | 주식회사 셀레믹스 | 폐암 조직 내 세포 유래 돌연변이를 검출하기 위한 프로브 제조 및 검출 방법 |
Non-Patent Citations (94)
Also Published As
| Publication number | Publication date |
|---|---|
| US11459567B2 (en) | 2022-10-04 |
| US20210403914A1 (en) | 2021-12-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Dews et al. | Augmentation of tumor angiogenesis by a Myc-activated microRNA cluster | |
| Fang et al. | Targeting the Wnt/β-catenin pathway in human osteosarcoma cells | |
| Zaytseva et al. | Inhibition of fatty acid synthase attenuates CD44-associated signaling and reduces metastasis in colorectal cancer | |
| Matà et al. | IGF-I induces upregulation of DDR1 collagen receptor in breast cancer cells by suppressing MIR-199a-5p through the PI3K/AKT pathway | |
| Zhang et al. | MiR-221 and miR-222 target PUMA to induce cell survival in glioblastoma | |
| Wu et al. | 2′-OMe-phosphorodithioate-modified siRNAs show increased loading into the RISC complex and enhanced anti-tumour activity | |
| Parmar et al. | Combinational siRNA delivery using hyaluronic acid modified amphiphilic polyplexes against cell cycle and phosphatase proteins to inhibit growth and migration of triple-negative breast cancer cells | |
| US20190276831A1 (en) | Lung cancer diagnostics and therapeutics with mir-660 | |
| Yang et al. | Targeting oncogenic KRAS in non-small cell lung cancer with EGFR aptamer-conjugated multifunctional RNA nanoparticles | |
| JP5931897B2 (ja) | マイクロrna−21、ミスマッチ修復および結腸直腸癌に関連する物質および方法 | |
| JP2017006137A (ja) | 肺がんの腫瘍発生および化学療法耐性を低減するために有用なmiRNAならびに関連する組成物および方法 | |
| US9334500B2 (en) | Methods and pharmaceutical compositions for treating cancer | |
| Jiang et al. | LOC102724163 promotes breast cancer cell proliferation and invasion by stimulating MUC19 expression | |
| WO2017207623A1 (en) | Mirna as biomarkers and regulators of cancer stem cells | |
| ES2873377T3 (es) | Procedimientos y composiciones farmacéuticas para el tratamiento de cáncer de pulmón | |
| Tang et al. | MicroRNAs in breast cancer therapy | |
| US11459567B2 (en) | Specific siRNA molecules, composition and use thereof for the treatment of triple negative breast cancer | |
| US20160319285A1 (en) | Use of miR-494 to Modulate TRAIL-induced Apoptosis through BIM Down-regulation | |
| JPWO2011040613A1 (ja) | 腫瘍治療剤 | |
| Zhang et al. | Mir-486-3p suppresses osteosarcoma proliferation and migration by targeting the SPRED1-MAPK/ERK pathway | |
| Wei et al. | Repression of metadherin inhibits biological behavior of prostate cancer cells and enhances their sensitivity to cisplatin | |
| US8198254B2 (en) | Annexin A9 (ANXA9) biomarker and therapeutic target in epithelial cancer | |
| WO2021064431A1 (en) | Targeting expression of kras and related methods | |
| Takahashi et al. | Small interfering RNA-mediated silencing of the ribophorin II gene: advances in the treatment of malignant breast cancer | |
| Adekunle | Functional CircRNAs in EGFR Activation Disease Progression of Lung Cancer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN PUBLISHED |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| B565 | Issuance of search results under rule 164(2) epc |
Effective date: 20211209 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20220629 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |