EP3382002A1 - Composition for contact lens - Google Patents
Composition for contact lens Download PDFInfo
- Publication number
- EP3382002A1 EP3382002A1 EP17190771.0A EP17190771A EP3382002A1 EP 3382002 A1 EP3382002 A1 EP 3382002A1 EP 17190771 A EP17190771 A EP 17190771A EP 3382002 A1 EP3382002 A1 EP 3382002A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- composition
- contact lens
- lens according
- algefacient
- surfactant
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 31
- 239000004094 surface-active agent Substances 0.000 claims abstract description 17
- -1 polyoxyethylene Polymers 0.000 claims abstract description 14
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims abstract description 13
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims abstract description 13
- 239000001267 polyvinylpyrrolidone Substances 0.000 claims abstract description 13
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims abstract description 13
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 claims abstract description 12
- 239000004359 castor oil Substances 0.000 claims abstract description 7
- 235000019438 castor oil Nutrition 0.000 claims abstract description 7
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 claims abstract description 7
- 229920000136 polysorbate Polymers 0.000 claims abstract description 7
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 claims abstract description 6
- SFNALCNOMXIBKG-UHFFFAOYSA-N ethylene glycol monododecyl ether Chemical compound CCCCCCCCCCCCOCCO SFNALCNOMXIBKG-UHFFFAOYSA-N 0.000 claims abstract description 6
- NTYZDAJPNNBYED-UHFFFAOYSA-M sodium;2-(2-dodecanoyloxypropanoyloxy)propanoate Chemical compound [Na+].CCCCCCCCCCCC(=O)OC(C)C(=O)OC(C)C([O-])=O NTYZDAJPNNBYED-UHFFFAOYSA-M 0.000 claims abstract description 6
- 229920003169 water-soluble polymer Polymers 0.000 claims description 11
- 239000000872 buffer Substances 0.000 claims description 8
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims description 7
- 229920002565 Polyethylene Glycol 400 Polymers 0.000 claims description 5
- NOOLISFMXDJSKH-UTLUCORTSA-N (+)-Neomenthol Chemical compound CC(C)[C@@H]1CC[C@@H](C)C[C@@H]1O NOOLISFMXDJSKH-UTLUCORTSA-N 0.000 claims description 4
- NOOLISFMXDJSKH-UHFFFAOYSA-N DL-menthol Natural products CC(C)C1CCC(C)CC1O NOOLISFMXDJSKH-UHFFFAOYSA-N 0.000 claims description 4
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 claims description 4
- 239000004327 boric acid Substances 0.000 claims description 4
- 229920002674 hyaluronan Polymers 0.000 claims description 4
- 229960003160 hyaluronic acid Drugs 0.000 claims description 4
- 229940041616 menthol Drugs 0.000 claims description 4
- DTGKSKDOIYIVQL-WEDXCCLWSA-N (+)-borneol Chemical compound C1C[C@@]2(C)[C@@H](O)C[C@@H]1C2(C)C DTGKSKDOIYIVQL-WEDXCCLWSA-N 0.000 claims description 3
- REPVLJRCJUVQFA-UHFFFAOYSA-N (-)-isopinocampheol Natural products C1C(O)C(C)C2C(C)(C)C1C2 REPVLJRCJUVQFA-UHFFFAOYSA-N 0.000 claims description 3
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 claims description 3
- DSSYKIVIOFKYAU-XCBNKYQSSA-N (R)-camphor Chemical compound C1C[C@@]2(C)C(=O)C[C@@H]1C2(C)C DSSYKIVIOFKYAU-XCBNKYQSSA-N 0.000 claims description 3
- JDRMYOQETPMYQX-UHFFFAOYSA-M 4-methoxy-4-oxobutanoate Chemical compound COC(=O)CCC([O-])=O JDRMYOQETPMYQX-UHFFFAOYSA-M 0.000 claims description 3
- 241000723346 Cinnamomum camphora Species 0.000 claims description 3
- UJNOLBSYLSYIBM-WISYIIOYSA-N [(1r,2s,5r)-5-methyl-2-propan-2-ylcyclohexyl] (2r)-2-hydroxypropanoate Chemical compound CC(C)[C@@H]1CC[C@@H](C)C[C@H]1OC(=O)[C@@H](C)O UJNOLBSYLSYIBM-WISYIIOYSA-N 0.000 claims description 3
- 229910021538 borax Inorganic materials 0.000 claims description 3
- CKDOCTFBFTVPSN-UHFFFAOYSA-N borneol Natural products C1CC2(C)C(C)CC1C2(C)C CKDOCTFBFTVPSN-UHFFFAOYSA-N 0.000 claims description 3
- 229940116229 borneol Drugs 0.000 claims description 3
- JDRMYOQETPMYQX-UHFFFAOYSA-N butanedioic acid monomethyl ester Natural products COC(=O)CCC(O)=O JDRMYOQETPMYQX-UHFFFAOYSA-N 0.000 claims description 3
- 229930008380 camphor Natural products 0.000 claims description 3
- 229960000846 camphor Drugs 0.000 claims description 3
- UQGFMSUEHSUPRD-UHFFFAOYSA-N disodium;3,7-dioxido-2,4,6,8,9-pentaoxa-1,3,5,7-tetraborabicyclo[3.3.1]nonane Chemical compound [Na+].[Na+].O1B([O-])OB2OB([O-])OB1O2 UQGFMSUEHSUPRD-UHFFFAOYSA-N 0.000 claims description 3
- DTGKSKDOIYIVQL-UHFFFAOYSA-N dl-isoborneol Natural products C1CC2(C)C(O)CC1C2(C)C DTGKSKDOIYIVQL-UHFFFAOYSA-N 0.000 claims description 3
- 239000004328 sodium tetraborate Substances 0.000 claims description 3
- 235000010339 sodium tetraborate Nutrition 0.000 claims description 3
- NFLGAXVYCFJBMK-RKDXNWHRSA-N (+)-isomenthone Natural products CC(C)[C@H]1CC[C@@H](C)CC1=O NFLGAXVYCFJBMK-RKDXNWHRSA-N 0.000 claims description 2
- OXJGJKIURHREKH-UHFFFAOYSA-O CC(=C)C(=O)OCCP(=O)=C(O)C[N+](C)(C)C Chemical compound CC(=C)C(=O)OCCP(=O)=C(O)C[N+](C)(C)C OXJGJKIURHREKH-UHFFFAOYSA-O 0.000 claims description 2
- NFLGAXVYCFJBMK-UHFFFAOYSA-N Menthone Chemical compound CC(C)C1CCC(C)CC1=O NFLGAXVYCFJBMK-UHFFFAOYSA-N 0.000 claims description 2
- 229930007503 menthone Natural products 0.000 claims description 2
- 229940068918 polyethylene glycol 400 Drugs 0.000 claims description 2
- 230000035807 sensation Effects 0.000 description 12
- 239000000243 solution Substances 0.000 description 6
- 230000000052 comparative effect Effects 0.000 description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 208000001491 myopia Diseases 0.000 description 4
- 230000004379 myopia Effects 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- SOGAXMICEFXMKE-UHFFFAOYSA-N Butylmethacrylate Chemical compound CCCCOC(=O)C(C)=C SOGAXMICEFXMKE-UHFFFAOYSA-N 0.000 description 2
- 206010013774 Dry eye Diseases 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 239000000017 hydrogel Substances 0.000 description 2
- 230000005923 long-lasting effect Effects 0.000 description 2
- 238000005461 lubrication Methods 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 239000003761 preservation solution Substances 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- ISIJQEHRDSCQIU-UHFFFAOYSA-N tert-butyl 2,7-diazaspiro[4.5]decane-7-carboxylate Chemical compound C1N(C(=O)OC(C)(C)C)CCCC11CNCC1 ISIJQEHRDSCQIU-UHFFFAOYSA-N 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- 206010015958 Eye pain Diseases 0.000 description 1
- 206010052140 Eye pruritus Diseases 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 229920001616 Polymacon Polymers 0.000 description 1
- 208000003464 asthenopia Diseases 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000003139 buffering effect Effects 0.000 description 1
- CQEYYJKEWSMYFG-UHFFFAOYSA-N butyl acrylate Chemical compound CCCCOC(=O)C=C CQEYYJKEWSMYFG-UHFFFAOYSA-N 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- BNIILDVGGAEEIG-UHFFFAOYSA-L disodium hydrogen phosphate Chemical compound [Na+].[Na+].OP([O-])([O-])=O BNIILDVGGAEEIG-UHFFFAOYSA-L 0.000 description 1
- 229910000397 disodium phosphate Inorganic materials 0.000 description 1
- 235000019800 disodium phosphate Nutrition 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 238000000034 method Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 229910000403 monosodium phosphate Inorganic materials 0.000 description 1
- 235000019799 monosodium phosphate Nutrition 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- JLFNLZLINWHATN-UHFFFAOYSA-N pentaethylene glycol Chemical compound OCCOCCOCCOCCOCCO JLFNLZLINWHATN-UHFFFAOYSA-N 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/0005—Other compounding ingredients characterised by their effect
- C11D3/0078—Compositions for cleaning contact lenses, spectacles or lenses
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/02—Anionic compounds
- C11D1/04—Carboxylic acids or salts thereof
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/02—Anionic compounds
- C11D1/12—Sulfonic acids or sulfuric acid esters; Salts thereof
- C11D1/14—Sulfonic acids or sulfuric acid esters; Salts thereof derived from aliphatic hydrocarbons or mono-alcohols
- C11D1/146—Sulfuric acid esters
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/02—Anionic compounds
- C11D1/12—Sulfonic acids or sulfuric acid esters; Salts thereof
- C11D1/29—Sulfates of polyoxyalkylene ethers
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/02—Anionic compounds
- C11D1/34—Derivatives of acids of phosphorus
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/66—Non-ionic compounds
- C11D1/667—Neutral esters, e.g. sorbitan esters
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/66—Non-ionic compounds
- C11D1/72—Ethers of polyoxyalkylene glycols
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/18—Hydrocarbons
- C11D3/188—Terpenes
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/20—Organic compounds containing oxygen
- C11D3/2003—Alcohols; Phenols
- C11D3/2006—Monohydric alcohols
- C11D3/2024—Monohydric alcohols cyclic; polycyclic
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/20—Organic compounds containing oxygen
- C11D3/2003—Alcohols; Phenols
- C11D3/2006—Monohydric alcohols
- C11D3/2037—Terpenes
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/20—Organic compounds containing oxygen
- C11D3/2003—Alcohols; Phenols
- C11D3/2041—Dihydric alcohols
- C11D3/2062—Terpene
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/20—Organic compounds containing oxygen
- C11D3/2093—Esters; Carbonates
Definitions
- the instant disclosure relates to a composition for contact lenses; in particular, to a composition for contact lenses which can serve as a contact lens preservation solution.
- Smartphones and computers are used frequently in today's information society, resulting in an increase in the population with myopia and a decrease in the age group of myopia.
- it is generally a good choice for people with myopia to wear contact lenses.
- most people with myopia need to wear contact lenses for extended periods of time.
- the material of the conventional contact lenses have been constantly improved to meet various requirements such as high water content and high oxygen permeability, the wearers thereof would still suffer from eye dryness due to decreased water content and experience eye discomfort due to dirt or secretion. Therefore, there is an urgent need for a maintenance solution that can be used to clean, rehydrate and preserve contact lenses.
- One aspect of the instant disclosure relates to a composition for contact lens which allows a contact lens wearer's eyes to feel comfortable during the period of initial wear and extended periods of continuous wear.
- the composition for contact lenses includes an algefacient and a surfactant, wherein the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80TM and GEROPONTM SBFA-30.
- the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80TM and GEROPONTM SBFA-30.
- the algefacient and the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80TM and GEROPONTM SBFA-30
- the expression of the algefacient in the composition of a contact lens can be performed effectively. Therefore, the discomfort and foreign matter sensation in the eyes of contact lens wearers can be reduced or relieved, providing long-lasting cool and moist sensations to the eyes.
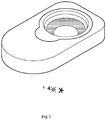
- Fig. 1 is a perspective view showing a contact lens and a composition for contact lenses according to the instant disclosure in a container.
- the instant disclosure provides a composition for contact lenses which can serve as a preservation solution and be hermetically sealed with the contact lenses in a container.
- the contact lenses are immersed in direct contact with the composition.
- the user takes the contact lens out of the composition and put the contact lenses on his/her eyes. More specifically, the ingredients of the composition absorb onto the surface of the contact lenses and then transfer into the user's eyes.
- the composition according to the instant embodiment is intended for use in connection with soft and rigid contact lenses.
- soft contact lens can refer to a hydrous soft contact lens made from hydrogels (i.e., hydrogel contact lenses) and a non-hydrous soft contact lens made from copolymers of butyl acrylate and butyl methacrylate.
- the composition according to the instant embodiment is applicable to the soft contact lenses in U.S. FDA category Group I-IV.
- Group I-IV lenses often contain materials identified in the market as: alphafilcon, asmofilcon, balafilcon, ethafilcon, hefilcon, hilafilcon, lidofilcon, lotrafilcon, methafilcon, nelfilcon, ocufilcon, omafilcon, phemfilcon, polymacon, tefilcon, tetrafilcon, vasurfilcon, vifilcon, senofilcon, galyfilcon, enfilcon, comfilcon, narafilcon, delefilcon, efrofilcon, and filicon II 3.
- the composition according to the instant embodiment mainly includes a base solution, an algefacient, a surfactant, and a water soluble polymer.
- the algefacient, the surfactant, and the water soluble polymer are uniformly dispersed in the base solution.
- the base solution is a physiological saline solution with a buffer containing boric acid or phosphorous acid.
- the buffer has a pH buffering capacity and is configured to adjust the pH of the composition to a pH between 6.8 and 7.6.
- the base solution can include an appropriate amount of sodium chloride or potassium chloride to maintain the osmolality of the composition at a value between 250 Osmol/Kg and 350 Osmol/Kg.
- the buffer can be selected from boric acid, borate (e.g., sodium tetraborate), phosphorous acid, or phosphate (e.g., sodium hydrogen phosphate and sodium dihydrogen phosphate). It should be noted that a buffer may be used alone or two or more different buffers may be used in combination in the base solution. In addition, the concentration of the buffer may be appropriately adjusted depending on its kind or the kind of the other ingredients.
- the algefacient is added to reduce or relieve the discomfort and foreign matter sensation in the eyes and give the eyes a cool, fresh sensation.
- the algefacient includes one or a combination of menthol, camphor, borneol, menthyl lactate, menthone glycerol ketal, monomethyl succinate, and p-menthane-3,8-doil.
- the content of the algefacient is in the range between 0.001% (w/v) and 0.5 % (w/v), preferably between 0.001% (w/v) and 0.25 % (w/v), to avoid irritating the eye.
- the surfactant is added to clean and moisturize a contact lens, in which protein and lipid deposits on the surfaces of the contact lens can be removed.
- the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80TM and GEROPONTM SBFA-30.
- the content of the surfactant is in the range between 0.001% (w/v) and 2% (w/v), and preferably between 0.001% (w/v) and 0.5 % (w/v).
- the expression of the algefacient in the composition for contact lenses can be performed effectively. Therefore, eye related symptoms such as eye fatigue, eye dryness, eye pruritus, and eye pain can be reduced or relieved, and a cool fresh sensation in the eyes can be maintained for a long period of time.
- the water soluble polymer is added to moisten and lubricate the eyes, and also helps to keep the ingredients beneficial to the eyes in contact with the contact lens.
- the composition for contact lenses includes one or more vitamins (e.g., A, B and E)
- the vitamin(s) can be attached to the surface of the contact lens by interacting with the water soluble polymer.
- the water soluble polymer includes one or a combination of polyethylene glycol 400 (PEG 400), methacryloyloxyethyl phosphorylcholine (MPC), and hyaluronic acid.
- the water soluble polymer includes PEG 400 and MPC in a molar ratio of 1:20.
- the content of the water soluble polymer is in the range between 0.05% (w/v) and 5% (w/v).
- Examples 1-5 and Comparative Examples 1-2 are shown in Table 1. Each of the compositions obtained respectively in Examples 1-5 and Comparative Examples 1-2 was used in full contact with the contact lenses. The contact lenses were then put on the user's eyes, and a VAS (Visual analogue scale) method was used to evaluate the sensations of lubrication, comfort, coolness, and moisture in the eyes during the period of initial wear (time 1) and after wearing for 30 minutes (time 2) and 60 minutes (time 3). The evaluation results of for the Examples 1-5 and the Comparative Examples 1-2 are shown in Table 2. Scores for each of the items, as shown in Table 2, are average scores calculated from three users (i.e., six eyes).
- Table 1 unit g Examples Comparative ex 1 2 3 4 5 1 2 water 2000 2000 2000 2000 2000 2000 2000 2000 2000 2000 2000 2000 sodium chloride 10-20 10-20 10-20 10-20 10-20 10-20 boric acid 8-16 8-16 8-16 8-16 8-16 8-16 sodium tetraborate 0.5-4.5 0.5-4.5 0.5-4.5 0.5-4.5 0.5-4.5 0.5-4.5 TWEEN 80TM 0.01-5 0.01-5 - 0.01-5 0.01-5 - - PVP 0.01-5 0.01-5 0.01-5 0 0.01-5 - - GEROPONTM SBFA-30 0 0.01-5 0.01-5 0 0.01-5 - - polyoxyethylene hardened castor oil - - - 0.01-2.5 0.01-2.5 - - Hyaluronic acid (HA) 0-50 0-50 0-50 0-50 0-50 - 0-50 PEG400 0-50 0-50 0-50 0-50 0-50 - 0-50 MPC 0-50 0-50 0-50 0-50 0-50
- composition for contact lenses in which the algefacient and the surfactant are cooperatively used and the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80TM and GEROPONTM SBFA-30. Accordingly, the expression of the algefacient can be performed effectively, so that the discomfort and foreign matter sensation in the eyes of contact lens wearers can be reduced or relieved, and long-lasting cool and moist sensations can be provided to the eyes.
Abstract
Description
- The instant disclosure relates to a composition for contact lenses; in particular, to a composition for contact lenses which can serve as a contact lens preservation solution.
- Smartphones and computers are used frequently in today's information society, resulting in an increase in the population with myopia and a decrease in the age group of myopia. In consideration of user convenience and aesthetics, it is generally a good choice for people with myopia to wear contact lenses. However, most people with myopia need to wear contact lenses for extended periods of time. Although the material of the conventional contact lenses have been constantly improved to meet various requirements such as high water content and high oxygen permeability, the wearers thereof would still suffer from eye dryness due to decreased water content and experience eye discomfort due to dirt or secretion. Therefore, there is an urgent need for a maintenance solution that can be used to clean, rehydrate and preserve contact lenses.
- One aspect of the instant disclosure relates to a composition for contact lens which allows a contact lens wearer's eyes to feel comfortable during the period of initial wear and extended periods of continuous wear.
- According to one of the embodiments of the instant disclosure, the composition for contact lenses includes an algefacient and a surfactant, wherein the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80™ and GEROPON™ SBFA-30.
- Based on the above, by cooperatively using the algefacient and the surfactant, wherein the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80™ and GEROPON™ SBFA-30, the expression of the algefacient in the composition of a contact lens can be performed effectively. Therefore, the discomfort and foreign matter sensation in the eyes of contact lens wearers can be reduced or relieved, providing long-lasting cool and moist sensations to the eyes.
- In order to further appreciate the characteristics and technical contents of the instant disclosure, references are hereunder made to the detailed descriptions and appended drawings in connection with the instant disclosure. However, the appended drawings are merely shown for exemplary purposes, rather than being used to restrict the scope of the instant disclosure.
-
Fig. 1 is a perspective view showing a contact lens and a composition for contact lenses according to the instant disclosure in a container. - The aforementioned illustrations and following detailed descriptions are exemplary for the purpose of further explaining the scope of the instant disclosure. Other objectives and advantages related to the instant disclosure will be illustrated in the subsequent descriptions and appended drawings.
- Referring to
FIG. 1 , the instant disclosure provides a composition for contact lenses which can serve as a preservation solution and be hermetically sealed with the contact lenses in a container. In practice, the contact lenses are immersed in direct contact with the composition. When the user wishes to wear the contact lenses, the user takes the contact lens out of the composition and put the contact lenses on his/her eyes. More specifically, the ingredients of the composition absorb onto the surface of the contact lenses and then transfer into the user's eyes. - The composition according to the instant embodiment is intended for use in connection with soft and rigid contact lenses. The term "soft contact lens" can refer to a hydrous soft contact lens made from hydrogels (i.e., hydrogel contact lenses) and a non-hydrous soft contact lens made from copolymers of butyl acrylate and butyl methacrylate. Furthermore, the composition according to the instant embodiment is applicable to the soft contact lenses in U.S. FDA category Group I-IV. Group I-IV lenses often contain materials identified in the market as: alphafilcon, asmofilcon, balafilcon, ethafilcon, hefilcon, hilafilcon, lidofilcon, lotrafilcon, methafilcon, nelfilcon, ocufilcon, omafilcon, phemfilcon, polymacon, tefilcon, tetrafilcon, vasurfilcon, vifilcon, senofilcon, galyfilcon, enfilcon, comfilcon, narafilcon, delefilcon, efrofilcon, and filicon II 3.
- The composition according to the instant embodiment mainly includes a base solution, an algefacient, a surfactant, and a water soluble polymer. The algefacient, the surfactant, and the water soluble polymer are uniformly dispersed in the base solution.
- In the instant embodiment, the base solution is a physiological saline solution with a buffer containing boric acid or phosphorous acid. The buffer has a pH buffering capacity and is configured to adjust the pH of the composition to a pH between 6.8 and 7.6. In addition, the base solution can include an appropriate amount of sodium chloride or potassium chloride to maintain the osmolality of the composition at a value between 250 Osmol/Kg and 350 Osmol/Kg.
- The buffer can be selected from boric acid, borate (e.g., sodium tetraborate), phosphorous acid, or phosphate (e.g., sodium hydrogen phosphate and sodium dihydrogen phosphate). It should be noted that a buffer may be used alone or two or more different buffers may be used in combination in the base solution. In addition, the concentration of the buffer may be appropriately adjusted depending on its kind or the kind of the other ingredients.
- The algefacient is added to reduce or relieve the discomfort and foreign matter sensation in the eyes and give the eyes a cool, fresh sensation. The algefacient includes one or a combination of menthol, camphor, borneol, menthyl lactate, menthone glycerol ketal, monomethyl succinate, and p-menthane-3,8-doil. The content of the algefacient is in the range between 0.001% (w/v) and 0.5 % (w/v), preferably between 0.001% (w/v) and 0.25 % (w/v), to avoid irritating the eye.
- The surfactant is added to clean and moisturize a contact lens, in which protein and lipid deposits on the surfaces of the contact lens can be removed. The surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80TM and GEROPONTM SBFA-30. The content of the surfactant is in the range between 0.001% (w/v) and 2% (w/v), and preferably between 0.001% (w/v) and 0.5 % (w/v).
- It should be noted that by cooperatively using the algefacient and the surfactant, the expression of the algefacient in the composition for contact lenses can be performed effectively. Therefore, eye related symptoms such as eye fatigue, eye dryness, eye pruritus, and eye pain can be reduced or relieved, and a cool fresh sensation in the eyes can be maintained for a long period of time.
- The water soluble polymer is added to moisten and lubricate the eyes, and also helps to keep the ingredients beneficial to the eyes in contact with the contact lens. For example, if the composition for contact lenses includes one or more vitamins (e.g., A, B and E), the vitamin(s) can be attached to the surface of the contact lens by interacting with the water soluble polymer. The water soluble polymer includes one or a combination of polyethylene glycol 400 (PEG 400), methacryloyloxyethyl phosphorylcholine (MPC), and hyaluronic acid. Exemplarily, the water soluble polymer includes PEG 400 and MPC in a molar ratio of 1:20. The content of the water soluble polymer is in the range between 0.05% (w/v) and 5% (w/v).
- Examples 1-5 and Comparative Examples 1-2 are shown in Table 1. Each of the compositions obtained respectively in Examples 1-5 and Comparative Examples 1-2 was used in full contact with the contact lenses. The contact lenses were then put on the user's eyes, and a VAS (Visual analogue scale) method was used to evaluate the sensations of lubrication, comfort, coolness, and moisture in the eyes during the period of initial wear (time 1) and after wearing for 30 minutes (time 2) and 60 minutes (time 3). The evaluation results of for the Examples 1-5 and the Comparative Examples 1-2 are shown in Table 2. Scores for each of the items, as shown in Table 2, are average scores calculated from three users (i.e., six eyes).
Table 1 unit: g Examples Comparative ex 1 2 3 4 5 1 2 water 2000 2000 2000 2000 2000 2000 2000 sodium chloride 10-20 10-20 10-20 10-20 10-20 10-20 10-20 boric acid 8-16 8-16 8-16 8-16 8-16 8-16 8-16 sodium tetraborate 0.5-4.5 0.5-4.5 0.5-4.5 0.5-4.5 0.5-4.5 0.5-4.5 0.5-4.5 TWEEN 80™ 0.01-5 0.01-5 - 0.01-5 0.01-5 - - PVP 0.01-5 0.01-5 0.01-5 0 0.01-5 - - GEROPON™ SBFA-30 0 0.01-5 0.01-5 0 0.01-5 - - polyoxyethylene hardened castor oil - - - 0.01-2.5 0.01-2.5 - - Hyaluronic acid (HA) 0-50 0-50 0-50 0-50 0-50 - 0-50 PEG400 0-50 0-50 0-50 0-50 0-50 - 0-50 MPC 0-50 0-50 0-50 0-50 0-50 - 0-50 menthol 0-4 0-4 0-4 0-4 0-4 - 0-4 camphor 0-4 0-4 0-4 0-4 0-4 - 0-4 borneol 0-4 0-4 0-4 0-4 0-4 - 0-4 menthyl lactate 0-4 0-4 0-4 0-4 0-4 - 0-4 menthone-glycerol ketal 0-4 0-4 0-4 0-4 0-4 - 0-4 monomethyl succinate 0-4 0-4 0-4 0-4 0-4 - 0-4 p-menthane-3,8-doil 0-4 0-4 0-4 0-4 0-4 - 0-4 Table 2 evaluation items wear-in time self-feeling score Examples Comparative ex lubrication sensation immediately right after 7 8 after 30 minutes 7 8 after 60 minutes 6 8 comfort sensation immediately right after 7 8 after 30 minutes 7 8 after 60 minutes 7 8 coolness sensation immediately right after NA 9 after 30 minutes NA 6 after 60 minutes NA 4 moisture sensation immediately right after 8 10 after 30 minutes 8 9 after 60 minutes 8 9 - The composition for contact lenses in which the algefacient and the surfactant are cooperatively used and the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80™ and GEROPON™ SBFA-30. Accordingly, the expression of the algefacient can be performed effectively, so that the discomfort and foreign matter sensation in the eyes of contact lens wearers can be reduced or relieved, and long-lasting cool and moist sensations can be provided to the eyes.
- The descriptions illustrated supra set forth simply the preferred embodiments of the instant disclosure; however, the characteristics of the instant disclosure are by no means restricted thereto. All changes, alternations, or modifications conveniently considered by those skilled in the art are deemed to be encompassed within the scope of the instant disclosure delineated by the following claims.
Claims (9)
- A composition for contact lenses which includes an algefacient and a surfactant, wherein the surfactant includes one or a combination of sodium lauroyl lactylate, polyoxyethylene laureth sulfate, polyoxyethylene hardened castor oil, polyvinylpyrrolidone (PVP), sodium dodecyl sulfate (SDS), TWEEN 80™ and GEROPON™ SBFA-30.
- The composition for contact lens according to claim 1, wherein the content of the surfactant is in the range between 0.001% (w/v) and 0.5% (w/v).
- The composition for contact lens according to claim 1, wherein the algefacient includes one or a combination of menthol, camphor, borneol, menthyl lactate, menthone glycerol ketal, monomethyl succinate, and p-menthane-3,8-doil.
- The composition for contact lens according to claim 3, wherein the algefacient is menthol.
- The composition for contact lens according to claim 3 or 4, wherein the content of the algefacient is in the range between 0.001% (w/v) and 0.25 % (w/v).
- The composition for contact lens according to claim 1, further including a water soluble polymer, wherein the water soluble polymer includes one or a combination of polyethylene glycol 400 (PEG 400), methacryloyloxyethyl phosphorylcholine (MPC), and hyaluronic acid.
- The composition for contact lens according to claim 6, wherein the water soluble polymer includes PEG 400 and MPC in a molar ratio of 1:20.
- The composition for contact lens according to claim 6 or 7, wherein the content of the water soluble polymer is in the range between 0.05% (w/v) and 5% (w/v).
- The composition for contact lens according to claim 1, further including a buffer, wherein the buffer includes boric acid and sodium tetraborate.
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| TW106111144A TWI607768B (en) | 2017-03-31 | 2017-03-31 | Composition for contact lenses and method for caring contact lenses |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP3382002A1 true EP3382002A1 (en) | 2018-10-03 |
| EP3382002B1 EP3382002B1 (en) | 2021-08-04 |
Family
ID=60021863
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP17190771.0A Active EP3382002B1 (en) | 2017-03-31 | 2017-09-13 | Composition for contact lens |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP3382002B1 (en) |
| JP (1) | JP6652530B2 (en) |
| TW (1) | TWI607768B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI798452B (en) * | 2019-06-17 | 2023-04-11 | 晶碩光學股份有限公司 | Contact lens product |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996003158A1 (en) * | 1994-07-22 | 1996-02-08 | Alcon Laboratories, Inc. | Use of low molecular weight amino acids in ophthalmic compositions |
| US6228323B1 (en) * | 1996-12-13 | 2001-05-08 | Alcon Laboratories, Inc. | Multi-purpose compositions containing an alkyl-trypsin and methods of use in contact lens cleaning and disinfecting |
| US20080095754A1 (en) * | 2006-10-18 | 2008-04-24 | Burke Susan E | Ophthalmic compositions comprising diglycine |
| US20170056335A1 (en) * | 2014-04-09 | 2017-03-02 | Rhoto Pharmaceutical Co., Ltd. | Ophthalmic Composition |
| US20170087199A1 (en) * | 2016-02-10 | 2017-03-30 | Senomyx, Inc. | Compositions for delivering a cooling sensation |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5494937A (en) * | 1994-07-22 | 1996-02-27 | Alcon Laboratories, Inc. | Saline solution for treating contact lenses |
| JP5437350B2 (en) * | 2000-12-28 | 2014-03-12 | ロート製薬株式会社 | Ophthalmic composition |
| JP4135905B2 (en) * | 2003-03-17 | 2008-08-20 | ロート製薬株式会社 | Eye drops for contact lenses containing a cooling agent |
| JP2012072183A (en) * | 2004-06-08 | 2012-04-12 | Rohto Pharmaceutical Co Ltd | Refreshing composition for ophthalmology |
| JP2007297559A (en) * | 2006-05-02 | 2007-11-15 | Rohto Pharmaceut Co Ltd | AMINO ACID-MODIFIED-(gamma-POLYGLUTAMIC ACID) OR ITS SALT AND THEIR USES |
| JP5401113B2 (en) * | 2008-02-07 | 2014-01-29 | ロート製薬株式会社 | Aqueous composition containing chlorous acid compounds containing menthols |
| JP4457237B1 (en) * | 2009-07-30 | 2010-04-28 | 株式会社シード | Contact lens solution |
| WO2011040572A1 (en) * | 2009-09-30 | 2011-04-07 | ロート製薬株式会社 | Ophthalmic composition |
| TWI480039B (en) * | 2011-03-10 | 2015-04-11 | Pegavision Corp | Aqueous composition for contact lens |
| WO2013188274A2 (en) * | 2012-06-14 | 2013-12-19 | Novartis Ag | Azetidinium-containing copolymers and uses thereof |
| JP6654340B2 (en) * | 2013-04-12 | 2020-02-26 | ロート製薬株式会社 | Contact lens composition and contact lens package using the same |
| KR101490344B1 (en) * | 2013-07-05 | 2015-02-11 | 마준오 | Contact lens with cooling coating layer |
| JP6616137B2 (en) * | 2015-09-15 | 2019-12-04 | 株式会社メニコン | Contact lens composition |
-
2017
- 2017-03-31 TW TW106111144A patent/TWI607768B/en active
- 2017-08-29 JP JP2017164517A patent/JP6652530B2/en active Active
- 2017-09-13 EP EP17190771.0A patent/EP3382002B1/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996003158A1 (en) * | 1994-07-22 | 1996-02-08 | Alcon Laboratories, Inc. | Use of low molecular weight amino acids in ophthalmic compositions |
| US6228323B1 (en) * | 1996-12-13 | 2001-05-08 | Alcon Laboratories, Inc. | Multi-purpose compositions containing an alkyl-trypsin and methods of use in contact lens cleaning and disinfecting |
| US20080095754A1 (en) * | 2006-10-18 | 2008-04-24 | Burke Susan E | Ophthalmic compositions comprising diglycine |
| US20170056335A1 (en) * | 2014-04-09 | 2017-03-02 | Rhoto Pharmaceutical Co., Ltd. | Ophthalmic Composition |
| US20170087199A1 (en) * | 2016-02-10 | 2017-03-30 | Senomyx, Inc. | Compositions for delivering a cooling sensation |
Non-Patent Citations (1)
| Title |
|---|
| ANONYMOUS: "Algefacient | definition of algefacient by Medical dictionary", MEDICAL DICTIONARY, 6 April 2018 (2018-04-06), pages 1 - 2, XP055465196, Retrieved from the Internet <URL:https://medical-dictionary.thefreedictionary.com/algefacient> [retrieved on 20180406] * |
Also Published As
| Publication number | Publication date |
|---|---|
| TWI607768B (en) | 2017-12-11 |
| TW201836650A (en) | 2018-10-16 |
| EP3382002B1 (en) | 2021-08-04 |
| JP2018173618A (en) | 2018-11-08 |
| JP6652530B2 (en) | 2020-02-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Covey et al. | Hypoxic effects on the anterior eye of high-Dk soft contact lens wearers are negligible | |
| Wolffsohn et al. | Crossover evaluation of silicone hydrogel daily disposable contact lenses | |
| Stern et al. | Comparison of the performance of 6-or 30-night extended wear schedules with silicone hydrogel lenses over 3 years | |
| JP6871334B2 (en) | Compositions for contact lenses and contact lens packages using them | |
| Campbell et al. | Clinical benefits of a new multipurpose disinfecting solution in silicone hydrogel and soft contact lens users | |
| González-Méijome et al. | Clinical performance and “ex vivo” dehydration of silicone hydrogel contact lenses with two new multipurpose solutions | |
| Sankaridurg et al. | The future of silicone hydrogels | |
| EP3382002B1 (en) | Composition for contact lens | |
| TWI651095B (en) | Antiallergic ophthalmic product | |
| JP5568174B1 (en) | Contact lens package and packaging solution for contact lens | |
| CN117099025A (en) | WS12 releasing contact lens | |
| EP1845405A1 (en) | Liquid agent composition for contact lens | |
| Efron et al. | The effect of soft lens lubricants on symptoms and lens dehydration | |
| CN108690740A (en) | The maintenance method of contact lenses composition and contact lenses | |
| Luensmann et al. | The impact of cosmetics on the physical dimension and optical performance of contemporary silicone hydrogel contact lenses | |
| CN109718376A (en) | It releives the ophthalmically acceptable product of effect with antiallergic | |
| Guthrie et al. | Exploring the factors which impact overall satisfaction with single vision contact lenses | |
| CN112094582A (en) | Contact lens solution capable of increasing moist feeling and contact lens product containing same | |
| JP6766121B2 (en) | Ophthalmic products and their ophthalmic compositions | |
| NO171885B (en) | Ophthalmic COMFORT KILL SOLUTIONS FOR INTRAOCULAR USE IN HYDROGAL CONTACT LENSES | |
| Bonanno et al. | Tear oxygen under hydrogel and silicone hydrogel contact lenses in humans | |
| CN110564519A (en) | Ophthalmic product and ophthalmic composition thereof | |
| Lakkis et al. | Clinical investigation of asmofilcon a silicone hydrogel lenses | |
| WO2019232717A1 (en) | Ophthalmic product and ophthalmic composition thereof | |
| JP7432643B2 (en) | Liquid compositions for ophthalmic products |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN PUBLISHED |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20190328 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| 17Q | First examination report despatched |
Effective date: 20190611 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20210217 |
|
| GRAJ | Information related to disapproval of communication of intention to grant by the applicant or resumption of examination proceedings by the epo deleted |
Free format text: ORIGINAL CODE: EPIDOSDIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| INTC | Intention to grant announced (deleted) | ||
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 1417015 Country of ref document: AT Kind code of ref document: T Effective date: 20210815 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602017043248 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG9D |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20210804 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 1417015 Country of ref document: AT Kind code of ref document: T Effective date: 20210804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20211104 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20211206 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20211104 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20211105 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602017043248 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20210930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20220506 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20210913 Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20210913 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20210930 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20210930 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20210930 |
|
| P01 | Opt-out of the competence of the unified patent court (upc) registered |
Effective date: 20230524 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20170913 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20230913 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20230829 Year of fee payment: 7 Ref country code: DE Payment date: 20230804 Year of fee payment: 7 |