EP2236053A1 - Method for the steam treatment of hair - Google Patents
Method for the steam treatment of hair Download PDFInfo
- Publication number
- EP2236053A1 EP2236053A1 EP10158979A EP10158979A EP2236053A1 EP 2236053 A1 EP2236053 A1 EP 2236053A1 EP 10158979 A EP10158979 A EP 10158979A EP 10158979 A EP10158979 A EP 10158979A EP 2236053 A1 EP2236053 A1 EP 2236053A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- shaping
- steam
- hair
- les
- cosmetic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 238000000034 method Methods 0.000 title claims abstract description 47
- 238000011282 treatment Methods 0.000 title claims abstract description 28
- 239000000835 fiber Substances 0.000 claims abstract description 56
- 238000007493 shaping process Methods 0.000 claims abstract description 44
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 7
- 239000002537 cosmetic Substances 0.000 claims description 41
- 238000009499 grossing Methods 0.000 claims description 34
- 239000000203 mixture Substances 0.000 claims description 22
- 239000003795 chemical substances by application Substances 0.000 claims description 11
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 6
- 238000004040 coloring Methods 0.000 claims description 6
- 239000004480 active ingredient Substances 0.000 claims description 4
- 239000003638 chemical reducing agent Substances 0.000 claims description 3
- 238000005406 washing Methods 0.000 claims description 3
- STIAPHVBRDNOAJ-UHFFFAOYSA-N carbamimidoylazanium;carbonate Chemical compound NC(N)=N.NC(N)=N.OC(O)=O STIAPHVBRDNOAJ-UHFFFAOYSA-N 0.000 claims description 2
- 239000008406 cosmetic ingredient Substances 0.000 claims description 2
- 125000000217 alkyl group Chemical group 0.000 description 36
- -1 aliphatic radical Chemical class 0.000 description 33
- 229920000642 polymer Polymers 0.000 description 26
- 125000004432 carbon atom Chemical group C* 0.000 description 25
- 150000001450 anions Chemical class 0.000 description 24
- 125000004429 atom Chemical group 0.000 description 24
- 150000003254 radicals Chemical class 0.000 description 17
- 125000002091 cationic group Chemical group 0.000 description 16
- 150000002148 esters Chemical group 0.000 description 16
- 229920001577 copolymer Polymers 0.000 description 14
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 13
- 229920006317 cationic polymer Polymers 0.000 description 13
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 12
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 12
- 229910019142 PO4 Inorganic materials 0.000 description 11
- 235000021317 phosphate Nutrition 0.000 description 11
- 229920000768 polyamine Polymers 0.000 description 10
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 9
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 9
- 150000004820 halides Chemical class 0.000 description 8
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 8
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 7
- 150000001408 amides Chemical group 0.000 description 7
- 150000001412 amines Chemical class 0.000 description 7
- 125000003118 aryl group Chemical group 0.000 description 7
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 7
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 7
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 6
- 239000004215 Carbon black (E152) Substances 0.000 description 6
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 6
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 6
- 150000001875 compounds Chemical class 0.000 description 6
- 239000006185 dispersion Substances 0.000 description 6
- 238000001035 drying Methods 0.000 description 6
- 229930195733 hydrocarbon Natural products 0.000 description 6
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 6
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 238000010438 heat treatment Methods 0.000 description 5
- 229920001519 homopolymer Polymers 0.000 description 5
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- 238000007254 oxidation reaction Methods 0.000 description 5
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 5
- 239000010452 phosphate Substances 0.000 description 5
- 229920001296 polysiloxane Polymers 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- 239000002453 shampoo Substances 0.000 description 5
- JKNCOURZONDCGV-UHFFFAOYSA-N 2-(dimethylamino)ethyl 2-methylprop-2-enoate Chemical compound CN(C)CCOC(=O)C(C)=C JKNCOURZONDCGV-UHFFFAOYSA-N 0.000 description 4
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical compound OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 4
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 4
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 4
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 4
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 4
- GTDPSWPPOUPBNX-UHFFFAOYSA-N ac1mqpva Chemical compound CC12C(=O)OC(=O)C1(C)C1(C)C2(C)C(=O)OC1=O GTDPSWPPOUPBNX-UHFFFAOYSA-N 0.000 description 4
- 125000002947 alkylene group Chemical group 0.000 description 4
- 125000003277 amino group Chemical group 0.000 description 4
- 239000000975 dye Substances 0.000 description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 4
- 229910052500 inorganic mineral Inorganic materials 0.000 description 4
- 150000003893 lactate salts Chemical class 0.000 description 4
- 239000011707 mineral Substances 0.000 description 4
- 125000004433 nitrogen atom Chemical group N* 0.000 description 4
- 150000007524 organic acids Chemical class 0.000 description 4
- 230000003647 oxidation Effects 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 238000010025 steaming Methods 0.000 description 4
- 229940095064 tartrate Drugs 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical class CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- BRLQWZUYTZBJKN-UHFFFAOYSA-N Epichlorohydrin Chemical compound ClCC1CO1 BRLQWZUYTZBJKN-UHFFFAOYSA-N 0.000 description 3
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 3
- 150000001242 acetic acid derivatives Chemical class 0.000 description 3
- 150000003926 acrylamides Chemical class 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 230000015556 catabolic process Effects 0.000 description 3
- 239000006071 cream Substances 0.000 description 3
- 238000006731 degradation reaction Methods 0.000 description 3
- 125000000623 heterocyclic group Chemical group 0.000 description 3
- 125000001183 hydrocarbyl group Chemical group 0.000 description 3
- 150000007522 mineralic acids Chemical class 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 239000002304 perfume Substances 0.000 description 3
- 239000011593 sulfur Substances 0.000 description 3
- 229910052717 sulfur Inorganic materials 0.000 description 3
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 3
- QLAJNZSPVITUCQ-UHFFFAOYSA-N 1,3,2-dioxathietane 2,2-dioxide Chemical compound O=S1(=O)OCO1 QLAJNZSPVITUCQ-UHFFFAOYSA-N 0.000 description 2
- OSSNTDFYBPYIEC-UHFFFAOYSA-N 1-ethenylimidazole Chemical compound C=CN1C=CN=C1 OSSNTDFYBPYIEC-UHFFFAOYSA-N 0.000 description 2
- WDRZVZVXHZNSFG-UHFFFAOYSA-N 1-ethenylpyridin-1-ium Chemical compound C=C[N+]1=CC=CC=C1 WDRZVZVXHZNSFG-UHFFFAOYSA-N 0.000 description 2
- OSCJHTSDLYVCQC-UHFFFAOYSA-N 2-ethylhexyl 4-[[4-[4-(tert-butylcarbamoyl)anilino]-6-[4-(2-ethylhexoxycarbonyl)anilino]-1,3,5-triazin-2-yl]amino]benzoate Chemical compound C1=CC(C(=O)OCC(CC)CCCC)=CC=C1NC1=NC(NC=2C=CC(=CC=2)C(=O)NC(C)(C)C)=NC(NC=2C=CC(=CC=2)C(=O)OCC(CC)CCCC)=N1 OSCJHTSDLYVCQC-UHFFFAOYSA-N 0.000 description 2
- KGIGUEBEKRSTEW-UHFFFAOYSA-N 2-vinylpyridine Chemical compound C=CC1=CC=CC=N1 KGIGUEBEKRSTEW-UHFFFAOYSA-N 0.000 description 2
- MXRGSJAOLKBZLU-UHFFFAOYSA-N 3-ethenylazepan-2-one Chemical compound C=CC1CCCCNC1=O MXRGSJAOLKBZLU-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- 229920000209 Hexadimethrine bromide Polymers 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- 102000011782 Keratins Human genes 0.000 description 2
- 108010076876 Keratins Proteins 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- QUBHKIHVNBLQBS-UHFFFAOYSA-N N-benzyl-N',N'-dimethyl-N-(3-methylphenyl)-1-phenylethane-1,2-diamine 2,3-dihydroxybutanedioic acid Chemical compound OC(=O)C(O)C(O)C(O)=O.C=1C=CC=CC=1C(CN(C)C)N(C=1C=C(C)C=CC=1)CC1=CC=CC=C1 QUBHKIHVNBLQBS-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 239000001361 adipic acid Substances 0.000 description 2
- 235000011037 adipic acid Nutrition 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 125000003282 alkyl amino group Chemical group 0.000 description 2
- 230000029936 alkylation Effects 0.000 description 2
- 238000005804 alkylation reaction Methods 0.000 description 2
- 150000003863 ammonium salts Chemical class 0.000 description 2
- 239000003093 cationic surfactant Substances 0.000 description 2
- NEHMKBQYUWJMIP-NJFSPNSNSA-N chloro(114C)methane Chemical compound [14CH3]Cl NEHMKBQYUWJMIP-NJFSPNSNSA-N 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- 230000003750 conditioning effect Effects 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 238000007334 copolymerization reaction Methods 0.000 description 2
- 150000004985 diamines Chemical class 0.000 description 2
- YIOJGTBNHQAVBO-UHFFFAOYSA-N dimethyl-bis(prop-2-enyl)azanium Chemical compound C=CC[N+](C)(C)CC=C YIOJGTBNHQAVBO-UHFFFAOYSA-N 0.000 description 2
- 239000000982 direct dye Substances 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- 150000002430 hydrocarbons Chemical class 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- MTNDZQHUAFNZQY-UHFFFAOYSA-N imidazoline Chemical compound C1CN=CN1 MTNDZQHUAFNZQY-UHFFFAOYSA-N 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- FQPSGWSUVKBHSU-UHFFFAOYSA-N methacrylamide Chemical class CC(=C)C(N)=O FQPSGWSUVKBHSU-UHFFFAOYSA-N 0.000 description 2
- 125000005395 methacrylic acid group Chemical group 0.000 description 2
- JZMJDSHXVKJFKW-UHFFFAOYSA-M methyl sulfate(1-) Chemical compound COS([O-])(=O)=O JZMJDSHXVKJFKW-UHFFFAOYSA-M 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 235000010446 mineral oil Nutrition 0.000 description 2
- ZIUHHBKFKCYYJD-UHFFFAOYSA-N n,n'-methylenebisacrylamide Chemical compound C=CC(=O)NCNC(=O)C=C ZIUHHBKFKCYYJD-UHFFFAOYSA-N 0.000 description 2
- 150000002823 nitrates Chemical class 0.000 description 2
- 150000002825 nitriles Chemical class 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 239000007800 oxidant agent Substances 0.000 description 2
- 125000004193 piperazinyl group Chemical group 0.000 description 2
- 229920001281 polyalkylene Polymers 0.000 description 2
- 238000006068 polycondensation reaction Methods 0.000 description 2
- 238000006116 polymerization reaction Methods 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000001453 quaternary ammonium group Chemical group 0.000 description 2
- 150000003335 secondary amines Chemical group 0.000 description 2
- 150000003457 sulfones Chemical class 0.000 description 2
- 239000002562 thickening agent Substances 0.000 description 2
- RRHXZLALVWBDKH-UHFFFAOYSA-M trimethyl-[2-(2-methylprop-2-enoyloxy)ethyl]azanium;chloride Chemical compound [Cl-].CC(=C)C(=O)OCC[N+](C)(C)C RRHXZLALVWBDKH-UHFFFAOYSA-M 0.000 description 2
- 230000000007 visual effect Effects 0.000 description 2
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 1
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 1
- GQWWGRUJOCIUKI-UHFFFAOYSA-N 2-[3-(2-methyl-1-oxopyrrolo[1,2-a]pyrazin-3-yl)propyl]guanidine Chemical group O=C1N(C)C(CCCN=C(N)N)=CN2C=CC=C21 GQWWGRUJOCIUKI-UHFFFAOYSA-N 0.000 description 1
- ACGQRMRFZCXYHQ-UHFFFAOYSA-N 3-[2-(2-aminoethylamino)ethyl-(dimethylamino)amino]propan-1-ol Chemical compound OCCCN(N(C)C)CCNCCN ACGQRMRFZCXYHQ-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 1
- 125000002853 C1-C4 hydroxyalkyl group Chemical group 0.000 description 1
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- QEVGZEDELICMKH-UHFFFAOYSA-N Diglycolic acid Chemical compound OC(=O)COCC(O)=O QEVGZEDELICMKH-UHFFFAOYSA-N 0.000 description 1
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-M Methanesulfonate Chemical compound CS([O-])(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-M 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 229930182559 Natural dye Natural products 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 229920002873 Polyethylenimine Polymers 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 239000004904 UV filter Substances 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000002535 acidifier Substances 0.000 description 1
- 229940095602 acidifiers Drugs 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 125000002252 acyl group Chemical group 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 125000002723 alicyclic group Chemical group 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000002877 alkyl aryl group Chemical group 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- NUKAPDHENUQUOI-UHFFFAOYSA-N benzyl(18-methylnonadecyl)azanium;chloride Chemical class [Cl-].CC(C)CCCCCCCCCCCCCCCCC[NH2+]CC1=CC=CC=C1 NUKAPDHENUQUOI-UHFFFAOYSA-N 0.000 description 1
- 230000001588 bifunctional effect Effects 0.000 description 1
- 230000001680 brushing effect Effects 0.000 description 1
- 239000004568 cement Substances 0.000 description 1
- WOWHHFRSBJGXCM-UHFFFAOYSA-M cetyltrimethylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCC[N+](C)(C)C WOWHHFRSBJGXCM-UHFFFAOYSA-M 0.000 description 1
- RLGQACBPNDBWTB-UHFFFAOYSA-N cetyltrimethylammonium ion Chemical compound CCCCCCCCCCCCCCCC[N+](C)(C)C RLGQACBPNDBWTB-UHFFFAOYSA-N 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229920006037 cross link polymer Polymers 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 239000003431 cross linking reagent Substances 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 125000004427 diamine group Chemical group 0.000 description 1
- UVQMSPMFCMIROL-UHFFFAOYSA-N diazanium propane dichloride Chemical compound [NH4+].[NH4+].[Cl-].[Cl-].CCC UVQMSPMFCMIROL-UHFFFAOYSA-N 0.000 description 1
- 125000004990 dihydroxyalkyl group Chemical group 0.000 description 1
- VAYGXNSJCAHWJZ-UHFFFAOYSA-N dimethyl sulfate Chemical compound COS(=O)(=O)OC VAYGXNSJCAHWJZ-UHFFFAOYSA-N 0.000 description 1
- DKJLEUVQMKPSHB-UHFFFAOYSA-N dimethyl-[3-(octadecanoylamino)propyl]-(2-oxo-2-tetradecoxyethyl)azanium;chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC(=O)NCCC[N+](C)(C)CC(=O)OCCCCCCCCCCCCCC DKJLEUVQMKPSHB-UHFFFAOYSA-N 0.000 description 1
- GQOKIYDTHHZSCJ-UHFFFAOYSA-M dimethyl-bis(prop-2-enyl)azanium;chloride Chemical compound [Cl-].C=CC[N+](C)(C)CC=C GQOKIYDTHHZSCJ-UHFFFAOYSA-M 0.000 description 1
- UCYFZDNMZYZSPN-UHFFFAOYSA-N docosyl(trimethyl)azanium Chemical compound CCCCCCCCCCCCCCCCCCCCCC[N+](C)(C)C UCYFZDNMZYZSPN-UHFFFAOYSA-N 0.000 description 1
- 229940082150 encore Drugs 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- QUPDWYMUPZLYJZ-UHFFFAOYSA-N ethyl Chemical compound C[CH2] QUPDWYMUPZLYJZ-UHFFFAOYSA-N 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 239000000834 fixative Substances 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 125000003827 glycol group Chemical group 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 125000002636 imidazolinyl group Chemical group 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 229910003480 inorganic solid Inorganic materials 0.000 description 1
- 239000004922 lacquer Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- WCYWZMWISLQXQU-UHFFFAOYSA-N methyl Chemical compound [CH3] WCYWZMWISLQXQU-UHFFFAOYSA-N 0.000 description 1
- 150000005451 methyl sulfates Chemical class 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- 239000000978 natural dye Substances 0.000 description 1
- 229920001542 oligosaccharide Polymers 0.000 description 1
- 150000002482 oligosaccharides Polymers 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 229940083254 peripheral vasodilators imidazoline derivative Drugs 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 150000004885 piperazines Chemical class 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 239000000419 plant extract Substances 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 150000003141 primary amines Chemical group 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 238000005956 quaternization reaction Methods 0.000 description 1
- DUIOPKIIICUYRZ-UHFFFAOYSA-N semicarbazide Chemical compound NNC(N)=O DUIOPKIIICUYRZ-UHFFFAOYSA-N 0.000 description 1
- 239000003352 sequestering agent Substances 0.000 description 1
- 229920005573 silicon-containing polymer Polymers 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 150000003871 sulfonates Chemical class 0.000 description 1
- 125000001273 sulfonato group Chemical group [O-]S(*)(=O)=O 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 229910021653 sulphate ion Inorganic materials 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 239000012209 synthetic fiber Substances 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 229920001897 terpolymer Polymers 0.000 description 1
- 150000003512 tertiary amines Chemical group 0.000 description 1
- 150000005621 tetraalkylammonium salts Chemical group 0.000 description 1
- CWERGRDVMFNCDR-UHFFFAOYSA-N thioglycolic acid Chemical class OC(=O)CS CWERGRDVMFNCDR-UHFFFAOYSA-N 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-M toluene-4-sulfonate Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-M 0.000 description 1
- USFMMZYROHDWPJ-UHFFFAOYSA-N trimethyl-[2-(2-methylprop-2-enoyloxy)ethyl]azanium Chemical compound CC(=C)C(=O)OCC[N+](C)(C)C USFMMZYROHDWPJ-UHFFFAOYSA-N 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 229920001567 vinyl ester resin Polymers 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A45—HAND OR TRAVELLING ARTICLES
- A45D—HAIRDRESSING OR SHAVING EQUIPMENT; EQUIPMENT FOR COSMETICS OR COSMETIC TREATMENTS, e.g. FOR MANICURING OR PEDICURING
- A45D7/00—Processes of waving, straightening or curling hair
- A45D7/06—Processes of waving, straightening or curling hair combined chemical and thermal
-
- A—HUMAN NECESSITIES
- A45—HAND OR TRAVELLING ARTICLES
- A45D—HAIRDRESSING OR SHAVING EQUIPMENT; EQUIPMENT FOR COSMETICS OR COSMETIC TREATMENTS, e.g. FOR MANICURING OR PEDICURING
- A45D7/00—Processes of waving, straightening or curling hair
- A45D7/02—Processes of waving, straightening or curling hair thermal
-
- A—HUMAN NECESSITIES
- A45—HAND OR TRAVELLING ARTICLES
- A45D—HAIRDRESSING OR SHAVING EQUIPMENT; EQUIPMENT FOR COSMETICS OR COSMETIC TREATMENTS, e.g. FOR MANICURING OR PEDICURING
- A45D1/00—Curling-tongs, i.e. tongs for use when hot; Curling-irons, i.e. irons for use when hot; Accessories therefor
- A45D2001/008—Curling-tongs, i.e. tongs for use when hot; Curling-irons, i.e. irons for use when hot; Accessories therefor with vapor generation, e.g. steam
Definitions
- the present invention relates to a novel method for treating hair fibers, in particular hair.
- this steam treatment may be associated with different cosmetic hair treatments.
- a hair treatment process which comprises the application of steam for less than 2 minutes at a temperature of at least 75 ° C on keratin fibers maintained under mechanical tension and on which a special cosmetic composition comprising, for example, an oil, a silicone, oxidation dyes, natural dyes, direct dyes and the like has previously been applied.
- a special cosmetic composition comprising, for example, an oil, a silicone, oxidation dyes, natural dyes, direct dyes and the like has previously been applied.
- EP 659,395 , EP 659 393 , EP 659,396 , EP 659,397 are the documents EP 659,395 , EP 659 393 , EP 659,396 , EP 659,397 .
- the purpose of the present invention is to develop a new process for treating keratin fibers which makes it possible to obtain an improvement in cosmetic and visual properties of the hair fibers with easy and quick processes to implement, and which are persistent to at least one wash.
- the steam application step and the shaping step are not carried out simultaneously on the same portion of hair fibers, these two steps can nevertheless be implemented with the same device configured to implement successively these two steps.
- two separate devices can be used to implement these dissociated steps.
- the method of the present invention makes it possible in particular to obtain rapid shaping of the hair fibers while improving their cosmetic properties. Moreover, the method of the present invention makes it possible to obtain an improvement in the cosmetic properties that are at least one wash and that the application is user-friendly.
- the hair fibers treated by the process of the invention thus have an improved smoothness with a brighter appearance than that obtained with the methods of the technique and this even in the absence of additional cosmetic active ingredients.
- hair fibers keratinous fibers such as hair or synthetic fibers called “extensions” which are intended to be reported on the hair of a person by various means including bonding and this to change the appearance of a person's natural hair, for example, by changing the volume, color or appearance of the hair.
- the application of the steam may be carried out before or after the step of shaping the hair fibers.
- the shaping of the hair fibers is performed after the steam treatment of these hair fibers.
- the application of the steam can be repeated several times on the same fibers, however, it is possible to obtain a very good cosmetic result after a single application of steam.
- the amount of steam is between 1 and 4 g / min.
- the water vapor that is applied to the hair contains one or more cosmetic ingredients and / or active ingredients such as a perfume, a shaping or conditioning active ingredient, etc.
- the application of steam may be carried out using any apparatus known per se for generating the amount of steam useful in the process of the invention.
- this apparatus is portable, that is to say that the reservoir for generating the steam is in contact with the part of the device comprising the steam distribution orifices.
- the shaping step of the hair fibers can be carried out with any type of means known in the art for obtaining a temperature on the fibers of at least 50 ° C.
- the shaping means of the capillary fibers are for example devices having a heating surface adapted to come into contact with the capillary fibers and to apply to these fibers a tension, even slight, to smooth, comb, disentangle the hair fibers.
- the shaping temperature is greater than 90 ° C, preferably between 90 ° C and 230 ° C, in particular between 150 and 200 ° C.
- the shaping step is carried out by means of pliers for smoothing.
- pliers for smoothing mention may be made of flat pliers for smoothing described in patents US5957140 , US5046516 , US7044139 , US5223694 , US5091629 .
- the method of the present invention makes it possible in particular when the application of the steam is carried out before shaping, to hydrate the fibers sufficiently to reduce the degradation thereof during the implementation of the shaping. at a temperature above 50 ° C.
- the shaping step is carried out after the steam treatment.
- the method of the invention further comprises a step of cosmetic treatment of the hair fibers by means of a composition comprising one or more cosmetic active agents.
- This cosmetic treatment step may be carried out before or after the application of the steam and / or before or after the shaping of the hair fibers. It should be noted in particular that this cosmetic treatment step is not carried out simultaneously with the application of steam.
- the method of the invention comprises, in order, the cosmetic treatment step, followed by the application of the steam and then the shaping of the hair fibers.
- the method of the invention comprises in order the application of the steam, followed by the shaping of the hair fibers, and then the cosmetic treatment step.
- the method of the invention comprises in order the application of steam, followed by the cosmetic treatment step and then the step of shaping the hair fibers.
- the method of the invention may comprise a second treatment step in post or pretreatment of the hair fibers, identical or different from the first cosmetic treatment step.
- the cosmetic treatment step may be a step of care of the hair fibers, which may or may not be followed by rinsing, a step of washing the fibers, a shaping step or control of the shape, for example a fixing gel, a shaping foam, a lacquer, a non-rinsed conditioner in the form of a cream, a permanent, semi-permanent or temporary coloring step, a permanent deformation step at using a reducing agent and optionally a fixing agent, an alkaline straightening step with sodium hydroxide or with guanidine carbonate.
- a shaping step or control of the shape for example a fixing gel, a shaping foam, a lacquer, a non-rinsed conditioner in the form of a cream, a permanent, semi-permanent or temporary coloring step, a permanent deformation step at using a reducing agent and optionally a fixing agent, an alkaline straightening step with sodium hydroxide or with guanidine carbonate.
- coloring precursors As active cosmetic agents, mention may be made of coloring precursors, direct dyes, silicone polymers or not, fixing or not, oils mineral, vegetable or synthetic, waxes, reducing agents, oxidants, UV filters, conditioning agents, anti-free radical agents, sequestering or stabilizing agents, antioxidants, preservatives, acidifiers, alkaline agents perfumes, volatile or nonvolatile silicones, chemically reactive or inert polymers, pigments, organic or inorganic solid particles, thickeners, vitamins, plant extracts, pro-penetrating or swelling agents of the fiber.
- non-polymeric, non-coloring cationic cosmetic agents containing one or more cationic units and having one or more fatty chains.
- dye not having in its chromophore structure having maximum absorption in the visible.
- fatty chain is meant in the sense of the present invention, any hydrocarbon chain comprising at least 8 carbon atoms, preferably 8 to 30 carbon atoms.
- the cationic cosmetic agent (s) defined above may be chosen from primary, secondary, tertiary and quaternary, aliphatic or cyclic amine groups.
- the amine groups are quaternary.
- the cationic cosmetic agent (s) used according to the invention are generally chosen from cationic surfactants.
- salts of primary, secondary or tertiary fatty amines optionally polyoxyalkylenated; quaternary ammonium salts with a charge not included in a cycle and without ester function; quaternary imidazoline derivatives; quaternary diammonium salts; quaternary ammonium salts with ester function (s); and / or one of their mixtures.
- the alkyl radicals R 15 may be linear or branched and more particularly linear.
- R 15 denotes a methyl, ethyl, hydroxyethyl or dihydroxypropyl radical and more particularly a methyl or ethyl radical.
- the sum x + y + z is from 1 to 10.
- R 16 is a radical R 20 hydrocarbon, it may be long and contain from 12 to 22 carbon atoms or short and have from 1 to 3 carbon atoms.
- R 18 is a hydrocarbon radical R 22 , it preferably has 1 to 3 carbon atoms.
- x and z are 0 or 1.
- y is 1.
- n, p and r are 2 or 3 and even more particularly are equal to 2.
- the anion is preferably a halide (chloride, bromide or iodide) or an alkyl sulphate more particularly methyl sulphate.
- a halide chloride, bromide or iodide
- an alkyl sulphate more particularly methyl sulphate.
- methanesulphonate, phosphate, nitrate, tosylate an anion derived from an organic acid such as acetate or lactate or any other anion compatible with ammonium with an ester function.
- the X- anion is even more particularly chloride or methylsulfate.
- hydrocarbon radicals are linear.
- tetraalkylammonium chlorides for example alkyltrimethylammonium chlorides, in which the alkyl radical contains approximately from 12 to 22 carbon atoms, are particularly preferred.
- ammonium salts containing at least one ester function described in the patent US Patent 4874554 .
- Quaternary ammonium salts of formula (IV) that are suitable for use in the invention include, for example, dipalmitoylethyl hydroxyethylmethyl methosulphate, marketed by STEPAN under the reference STEPANQUAT GA 90.
- the preferred cationic molecules that may be used in the composition according to the invention are the compounds of formula (I) or of formula (IV).
- Cetyl trimethyl ammonium chloride is preferred.
- the cationic molecule (s) may also be chosen from cationic proteins.
- the cationic cosmetic agent or agents advantageously represent from 0.1% to 10% by weight of the total weight of the composition, provided that if the cationic cosmetic agent is a tetraalkylammonium salt containing only one fatty chain, it is present at a content greater than or equal to 1% by weight relative to the total weight of the composition.
- the cationic cosmetic agent (s) used in the invention whatever their nature, represent from 1 to 10%, better still from 1 to 5%, by weight of the total weight of the composition.
- the cationic cosmetic agent (s) when the cationic cosmetic agent (s) comprise two fatty chains, it has no amide function.
- a cosmetic agent useful in the process of the invention of cationic polymers which are non-coloring and which are not derived from a sugar or a silicone, and which have a cationic charge density greater than 1.5. meq / g.
- the charge density can be determined according to the Kjedahl method. It is generally measured at a pH of the order of 3 to 9.
- the cationic polymers used in the present invention have a cationic charge density of less than 10 meq / g.
- cationic polymer any polymer containing cationic groups and / or ionizable groups into cationic groups.
- dye not having in its chromophore structure having a maximum of absorption in the visible.
- sugar unit mono, oligo or polysaccharide
- unit containing one or more silicon atoms By not derived from a sugar or a silicone, one understands not including in its structure of sugar unit (mono, oligo or polysaccharide) or of unit containing one or more silicon atoms.
- the cationic polymers that may be used in accordance with the present invention may be chosen from all those already known per se as improving the cosmetic properties of the hair treated with detergent compositions, namely in particular those described in the patent application.
- the preferred cationic polymers are chosen from those containing units containing primary, secondary, tertiary and / or quaternary amine groups that may either be part of the main polymer chain or may be carried by a lateral substituent directly connected thereto.
- the cationic polymers used have a weight average molecular weight of greater than 10 5 , preferably greater than 10 6 and more preferably 10 6 to 10 8 .
- the cationic polymers are not polyurethanes.
- cationic polymers that may be used according to the invention, mention may be made more particularly of polymers of the polyamine, polyaminoamide and quaternary polyammonium type. These are known products.
- Strands of hair are washed and wrung out. Pre-drying of the hair is then carried out until a drying of about 80% is obtained.
- steam is applied with a flow rate of the order of 3 g / min, this steam application being followed by the shaping / smoothing step by means of a smoothing pliers heated to minus 100 ° C. This treatment is done with a wick on the whole of the hair.
- a 5.64 (copper red light brownish brown Majirouge) permanent oxidation dye is applied, which consists of applying to the hair fibers one or more oxidation bases and one or more couplers well known in the field of permanent coloring. in the presence of an oxidizing agent (hydrogen peroxide).
- fixative is then applied to the entire hair. After a period of exposure, the locks are rinsed, dewatered and dried.
- Thermal Cement of Kerastase which is a care product not rinsed including silicones and cationic surfactants. A pre-drying of the locks is then used until a drying of about 80% is obtained.
- Strands of hair are washed and wrung out. Pre-drying of the hair is then carried out until a drying of about 80% is obtained.
- steam is applied under the conditions defined in the present invention, this steam application being followed by the shaping / smoothing step by means of a smoothing tongs heated to at least 100 °. vs. This treatment is done with a wick on the whole of the hair.
Landscapes
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Cosmetics (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Chemical Or Physical Treatment Of Fibers (AREA)
- Yarns And Mechanical Finishing Of Yarns Or Ropes (AREA)
Abstract
Description
La présente invention concerne un nouveau procédé de traitement des fibres capillaire, en particulier des cheveux.The present invention relates to a novel method for treating hair fibers, in particular hair.
Il est connu de lisser les fibres capillaires avec des pinces de lissage. Ces pinces permettent d'obtenir un lissage des fibres kératiniques à température élevée sans tirer sur les cheveux contrairement aux brushings. Cependant, pour obtenir un bon aspect lissé, il est nécessaire d'effectuer plusieurs passages de la pince ce qui allonge notablement le temps de traitement des cheveux. Par ailleurs, l'application répétée des pinces plates de lissage peut engendrer une dégradation des fibres capillaires due à la température d'application de la pince de lissage.It is known to smooth the hair fibers with pliers of smoothing. These clamps make it possible to obtain a smoothing of the keratinous fibers at high temperature without pulling on the hair, unlike brushings. However, to obtain a smooth appearance, it is necessary to perform several passes of the clamp which significantly increases the time of hair treatment. Moreover, the repeated application of the smoothing flat grippers may cause a degradation of the hair fibers due to the application temperature of the smoothing gripper.
Il est aussi connu d'utiliser de la vapeur pour le traitement des cheveux, ce traitement à la vapeur pouvant être associé à différents traitements cosmétiques capillaires. En particulier, il est connu dans le domaine de la mise en plis (mise en forme non permanente) un procédé de traitement des cheveux qui comprend l'application de vapeur pendant un temps inférieur à 2 minutes à une température d'au moins 75°C sur des fibres kératiniques maintenues sous tension mécanique et sur lesquelles on a au préalable appliqué une composition cosmétique particulière comprenant par exemple, une huile, une silicone, des colorants d'oxydation, des colorants naturels, des colorants directs et autres. On peut citer à titre d'exemples les documents
Il est d'autre part connu de traiter les cheveux avec des pinces à lisser délivrant de la vapeur d'eau. Cependant, ces pinces ne permettent pas d'obtenir un effet cosmétique satisfaisant car la chaleur dispensée par ces pinces est moins importante que celle dispensée par les pinces de lissage classiques. On peut notamment citer le document
Ces procédés bien qu'améliorant les propriétés cosmétiques et visuelles des fibres capillaires sont longs et souvent difficiles à mettre en oeuvre. L'effet cosmétique obtenu est de plus non rémanent au lavage.These methods, while improving the cosmetic and visual properties of the hair fibers, are long and often difficult to implement. The cosmetic effect obtained is also non-retentive washing.
Ainsi le but de la présente invention est de développer un nouveau procédé de traitement des fibres kératiniques qui permet d'obtenir une amélioration des propriétés cosmétiques et visuelles des fibres capillaires avec des procédés faciles et rapides à mettre en oeuvre, et qui sont rémanents à au moins un lavage.Thus, the purpose of the present invention is to develop a new process for treating keratin fibers which makes it possible to obtain an improvement in cosmetic and visual properties of the hair fibers with easy and quick processes to implement, and which are persistent to at least one wash.
Ce but est atteint par la présente invention qui a pour objet un procédé de traitement des fibres capillaires qui comprend :
- (i) l'application sur les fibres capillaires de la vapeur d'eau en quantité inférieure à 5 g/min,
- (ii) la mise en forme de ces fibres capillaires à une température supérieure à 50°C,
l'application de la vapeur et de la mise en forme étant dissociée.
- (i) the application on the capillary fibers of the water vapor in an amount of less than 5 g / min,
- (ii) shaping these hair fibers at a temperature above 50 ° C,
the application of steam and shaping being dissociated.
Autrement dit, pour une mèche de fibres capillaires traitée, l'étape d'application de vapeur et l'étape de mise en forme ne se font pas simultanément sur une même portion de fibres capillaires, ces deux étapes pouvant néanmoins être mises en oeuvre avec un même appareil configuré pour mettre en oeuvre successivement ces deux étapes. Alternativement, deux appareils distincts peuvent être utilisés pour mettre en oeuvre ces étapes dissociées.In other words, for a treated hair fiber wick, the steam application step and the shaping step are not carried out simultaneously on the same portion of hair fibers, these two steps can nevertheless be implemented with the same device configured to implement successively these two steps. Alternatively, two separate devices can be used to implement these dissociated steps.
Le procédé de la présente invention permet en particulier d'obtenir une mise en forme rapide des fibres capillaires tout en améliorant leurs propriétés cosmétiques. Par ailleurs, le procédé de la présente invention permet d'obtenir une amélioration des propriétés cosmétiques rémanente à au moins un lavage et du confort d'application pour l'utilisateur.The method of the present invention makes it possible in particular to obtain rapid shaping of the hair fibers while improving their cosmetic properties. Moreover, the method of the present invention makes it possible to obtain an improvement in the cosmetic properties that are at least one wash and that the application is user-friendly.
Les fibres capillaires traitées par le procédé de l'invention présentent ainsi un lissage amélioré avec un aspect plus brillant que celui obtenu avec le procédés de la technique et ceci même en l'absence d'actifs cosmétiques additionnels.The hair fibers treated by the process of the invention thus have an improved smoothness with a brighter appearance than that obtained with the methods of the technique and this even in the absence of additional cosmetic active ingredients.
Dans la cadre de la présente invention, on entend par « fibres capillaires », des fibres kératiniques telles que les cheveux ou des fibres synthétiques appelées « extensions » qui sont destinées à être rapportées sur la chevelure d'une personne par différents moyens notamment par collage et ceci afin de modifier l'aspect de la chevelure naturelle d'une personne par exemple, en modifiant le volume, la couleur ou l'aspect de la chevelure.In the context of the present invention, the term "hair fibers", keratinous fibers such as hair or synthetic fibers called "extensions" which are intended to be reported on the hair of a person by various means including bonding and this to change the appearance of a person's natural hair, for example, by changing the volume, color or appearance of the hair.
L'application de la vapeur peut être mise en oeuvre avant ou après l'étape de mise en forme des fibres capillaires. Selon un mode de réalisation particulier, la mise en forme des fibres capillaires est effectuée après le traitement à la vapeur de ces fibres capillaires.The application of the steam may be carried out before or after the step of shaping the hair fibers. According to a particular embodiment, the shaping of the hair fibers is performed after the steam treatment of these hair fibers.
L'application de la vapeur peut être répétée plusieurs fois sur les mêmes fibres, cependant, il est possible d'obtenir un très bon résultat cosmétique après une seule application de vapeur.The application of the steam can be repeated several times on the same fibers, however, it is possible to obtain a very good cosmetic result after a single application of steam.
Selon un mode de réalisation particulier de l'invention, la quantité de vapeur est comprise entre 1 et 4 g/min.According to a particular embodiment of the invention, the amount of steam is between 1 and 4 g / min.
Selon un mode de réalisation, particulier, la vapeur d'eau qui est appliquée sur les cheveux contient un ou plusieurs ingrédients et/ou actifs cosmétiques tels qu'un parfum, un actif de mise en forme ou de conditionnement, etc.According to one particular embodiment, the water vapor that is applied to the hair contains one or more cosmetic ingredients and / or active ingredients such as a perfume, a shaping or conditioning active ingredient, etc.
L'application de la vapeur peut être mise en oeuvre à l'aide de tout appareil connu en soi pour générer la quantité de vapeur utile dans le procédé de l'invention. Selon un mode de réalisation particulier, cet appareil est portable, c'est à dire que le réservoir permettant de générer la vapeur est en contact avec la partie du dispositif comprenant les orifices de distribution de la vapeurThe application of steam may be carried out using any apparatus known per se for generating the amount of steam useful in the process of the invention. According to a particular embodiment, this apparatus is portable, that is to say that the reservoir for generating the steam is in contact with the part of the device comprising the steam distribution orifices.
L'étape de mise en forme des fibres capillaires peut être mise en oeuvre avec tout type de moyens connus de la technique permettant d'obtenir une température sur les fibres d'au moins 50°C. Les moyens de mise en forme des fibres capillaires sont par exemple des dispositifs présentant une surface chauffante apte à venir en contact avec les fibres capillaires et à appliquer sur ces fibres une tension, même légère, permettant de lisser, coiffer, démêler les fibres capillaires.The shaping step of the hair fibers can be carried out with any type of means known in the art for obtaining a temperature on the fibers of at least 50 ° C. The shaping means of the capillary fibers are for example devices having a heating surface adapted to come into contact with the capillary fibers and to apply to these fibers a tension, even slight, to smooth, comb, disentangle the hair fibers.
Selon un mode de réalisation particulier du procédé de l'invention, la température de mise en forme est supérieure à 90°C, de préférence entre 90°C et 230°C, notamment entre 150 et 200°C.According to a particular embodiment of the process of the invention, the shaping temperature is greater than 90 ° C, preferably between 90 ° C and 230 ° C, in particular between 150 and 200 ° C.
Selon une variante, l'étape de mise en forme est mise en oeuvre aux moyens de pinces de lissage. Notamment, on peut citer les pinces plates de lissage décrites dans les brevets
Bien que la mise en forme, notamment le lissage des fibres capillaires, puisse être mise en oeuvre plusieurs fois, on obtient des résultats déjà très satisfaisants avec un seul passage de la pince de lissage.Although the shaping, in particular the smoothing of the hair fibers, can be carried out several times, very satisfactory results are obtained with a single passage of the smoothing pliers.
Le procédé de la présente invention permet notamment lorsque l'application de la vapeur est mise en oeuvre avant la mise en forme, d'hydrater suffisamment les fibres pour réduire la dégradation de celles-ci lors de la mise en oeuvre de la mise en forme à une température supérieure à 50°C.The method of the present invention makes it possible in particular when the application of the steam is carried out before shaping, to hydrate the fibers sufficiently to reduce the degradation thereof during the implementation of the shaping. at a temperature above 50 ° C.
Lorsque l'application de la vapeur a lieu après la mise en forme, la vapeur vient réhydrater les fibres réduisant de la même façon la dégradation de fibres capillaires.When steam is applied after shaping, the steam re-hydrates the fibers, reducing the degradation of hair fibers in the same way.
De façon préférée, l'étape de mise en forme est mise en oeuvre après le traitement à la vapeur.Preferably, the shaping step is carried out after the steam treatment.
Selon un mode de réalisation particulier, le procédé de l'invention comprend de plus une étape de traitement cosmétique des fibres capillaires au moyen d'une composition comprenant un ou plusieurs actifs cosmétiques.According to a particular embodiment, the method of the invention further comprises a step of cosmetic treatment of the hair fibers by means of a composition comprising one or more cosmetic active agents.
Cette étape de traitement cosmétique peut être mise en oeuvre avant ou après l'application de la vapeur et/ou avant ou après la mise en forme des fibres capillaires. Il faut en particulier noter que cette étape de traitement cosmétique n'est pas mise en oeuvre simultanément à l'application de la vapeur.This cosmetic treatment step may be carried out before or after the application of the steam and / or before or after the shaping of the hair fibers. It should be noted in particular that this cosmetic treatment step is not carried out simultaneously with the application of steam.
Selon une première variante, le procédé de l'invention comprend dans l'ordre l'étape de traitement cosmétique, suivi de l'application de la vapeur puis de la mise en forme des fibres capillaires.According to a first variant, the method of the invention comprises, in order, the cosmetic treatment step, followed by the application of the steam and then the shaping of the hair fibers.
Selon une seconde variante, le procédé de l'invention comprend dans l'ordre l'application de la vapeur, suivi de la mise en forme des fibres capillaires, puis de l'étape de traitement cosmétique.According to a second variant, the method of the invention comprises in order the application of the steam, followed by the shaping of the hair fibers, and then the cosmetic treatment step.
Selon une troisième variante, le procédé de l'invention comprend dans l'ordre l'application de la vapeur, suivi de l'étape de traitement cosmétique puis de l'étape de la mise en forme des fibres capillaires.According to a third variant, the method of the invention comprises in order the application of steam, followed by the cosmetic treatment step and then the step of shaping the hair fibers.
Selon ces deux variantes, le procédé de l'invention peut comprendre une seconde étape de traitement en post ou prétraitement des fibres capillaires, identique ou différente de la première étape de traitement cosmétique.According to these two variants, the method of the invention may comprise a second treatment step in post or pretreatment of the hair fibers, identical or different from the first cosmetic treatment step.
L'étape de traitement cosmétique peut être une étape de soin des fibres capillaires, qui peut être suivie ou non d'un rinçage, une étape de lavage des fibres, une étape de mise en forme ou de la maitrise de la forme par exemple au moyen d'un gel fixant, d'une mousse de mise en forme, d'une laque, d'un après shampooing non rincé sous forme de crème, une étape de coloration permanente, semi permanente ou temporaire, une étape de déformation permanente au moyen d'un agent réducteur et éventuellement d'un agent fixateur, une étape de défrisage alcalin à la soude ou avec du carbonate de guanidine.The cosmetic treatment step may be a step of care of the hair fibers, which may or may not be followed by rinsing, a step of washing the fibers, a shaping step or control of the shape, for example a fixing gel, a shaping foam, a lacquer, a non-rinsed conditioner in the form of a cream, a permanent, semi-permanent or temporary coloring step, a permanent deformation step at using a reducing agent and optionally a fixing agent, an alkaline straightening step with sodium hydroxide or with guanidine carbonate.
A titre d'agents cosmétiques actifs, on peut citer les précurseurs de coloration, les colorants directs, les polymères siliconés ou non, fixants ou non, les huiles minérales, végétales ou de synthèse, les cires, les agents réducteurs, les oxydants, les filtres UV, les agents conditionneurs, les agents anti-radicaux libres, les agents sequestrants ou stabilisants, les antioxidants, les conservateurs, les acidifiants, les agents alcalins, les parfums, les silicones volatiles ou non, les polymères réactifs ou inertes chimiquement, les pigments, les particules solides organiques ou minérales, les épaississants, les vitamines, les extraits végétaux, les agents pro-pénétrants ou de gonflement de la fibre.As active cosmetic agents, mention may be made of coloring precursors, direct dyes, silicone polymers or not, fixing or not, oils mineral, vegetable or synthetic, waxes, reducing agents, oxidants, UV filters, conditioning agents, anti-free radical agents, sequestering or stabilizing agents, antioxidants, preservatives, acidifiers, alkaline agents perfumes, volatile or nonvolatile silicones, chemically reactive or inert polymers, pigments, organic or inorganic solid particles, thickeners, vitamins, plant extracts, pro-penetrating or swelling agents of the fiber.
A titre d'exemple, on peut citer en particulier les agents cosmétiques cationiques non polymériques, non colorantes, contenant un ou plusieurs motifs cationiques et présentant une ou plusieurs chaînes grasses.By way of example, mention may be made in particular of non-polymeric, non-coloring cationic cosmetic agents containing one or more cationic units and having one or more fatty chains.
Par non colorant, on entend ne présentant pas dans sa structure de chromophore présentant des maximum d'absorption dans le visible.By dye is meant not having in its chromophore structure having maximum absorption in the visible.
Par chaîne grasse, on entend au sens de la présente invention, toute chaîne hydrocarbonée comprenant au moins 8 atomes de carbone, de préférence de 8 à 30 atomes de carbone.By fatty chain is meant in the sense of the present invention, any hydrocarbon chain comprising at least 8 carbon atoms, preferably 8 to 30 carbon atoms.
Le ou les agents cosmétiques cationiques définis ci-dessus peuvent être choisis parmi les groupements amines primaires, secondaires, tertiaires et quaternaires, aliphatiques ou cycliques. De préférence, les groupements amines sont quaternaires.The cationic cosmetic agent (s) defined above may be chosen from primary, secondary, tertiary and quaternary, aliphatic or cyclic amine groups. Preferably, the amine groups are quaternary.
Ainsi, le ou les agents cosmétiques cationiques utilisés selon l'invention sont généralement choisies parmi les tensioactifs cationiques.Thus, the cationic cosmetic agent (s) used according to the invention are generally chosen from cationic surfactants.
A titre d'exemple, on peut citer les sels d'amines grasses primaires, secondaires ou tertiaires, éventuellement polyoxyalkylénées ; les sels d'ammonium quaternaire avec charge non incluse dans un cycle et sans fonction ester ; les dérivés quaternaires d'imidazoline ; les sels de diammonium quaternaire ; les sels d'ammonium quaternaire à fonction(s) ester ; et/ou l'un de leurs mélanges.By way of example, mention may be made of salts of primary, secondary or tertiary fatty amines, optionally polyoxyalkylenated; quaternary ammonium salts with a charge not included in a cycle and without ester function; quaternary imidazoline derivatives; quaternary diammonium salts; quaternary ammonium salts with ester function (s); and / or one of their mixtures.
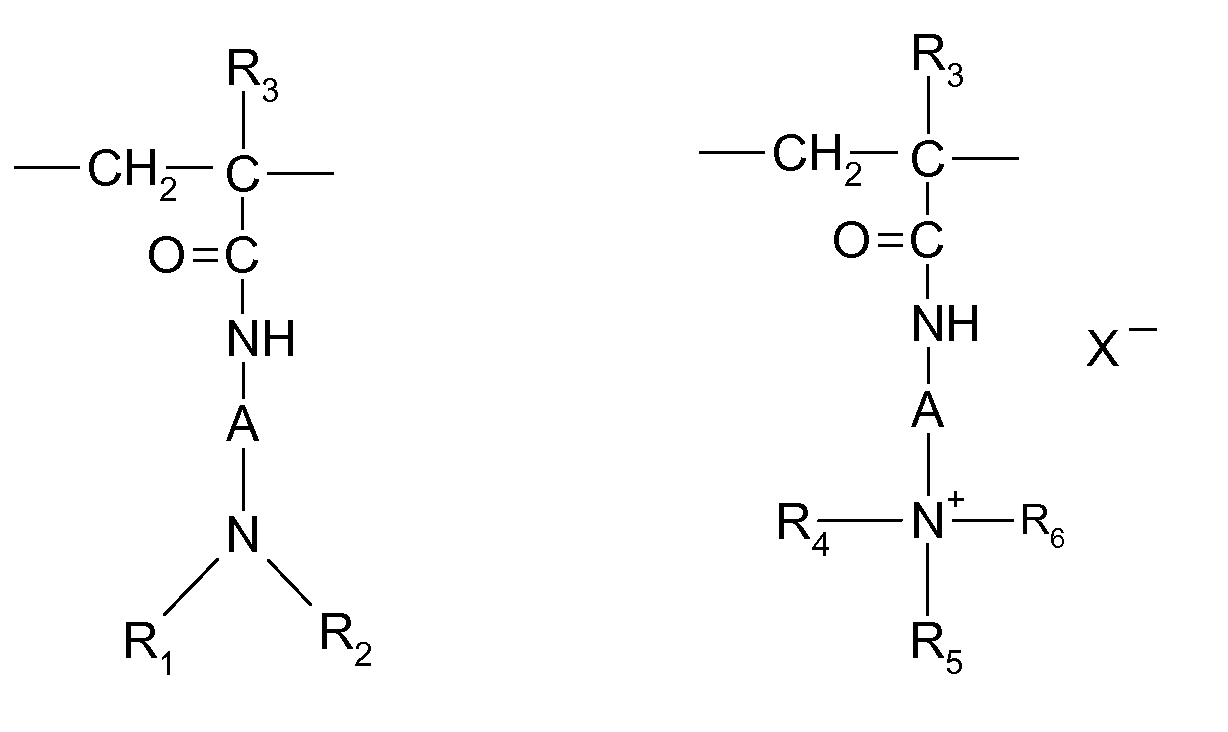
Les sels d'ammonium quaternaires avec charge non incluse dans un cycle et sans fonction ester sont par exemple :
- ceux qui présentent la formule générale (I) suivante :
- les sels d'ammonium quaternaire de l'imidazoline, comme par exemple celui de formule (II) suivante :
- les sels de diammonium quaternaire de formule (III) :
- les sels d'ammonium quaternaire contenant au moins une fonction ester, par exemple ceux de formule (IV) suivante :
- R15 est choisi parmi les radicaux alkyles en C1-C6 et les radicaux hydroxyalkyles ou dihydroxyalkyles en C1-C6;
- R16 est choisi parmi :
- le radical
- les radicaux R20 hydrocarbonés en C1-C22 linéaires ou ramifiés, saturés ou insaturés,
- l'atome d'hydrogène,
- R18 est choisi parmi :
- le radical
- les radicaux R22 hydrocarbonés en C1-C6 linéaires ou ramifiés, saturés ou insaturés,
- l'atome d'hydrogène,
- R17, R19 et R21, identiques ou différents, sont choisis parmi les radicaux hydrocarbonés en C7-C21, linéaires ou ramifiés, saturés ou insaturés;
- n, p et r, identiques ou différents, sont des entiers valant de 2 à 6 ;
- y est un entier valant de 1 à 10 ;
- x et z, identiques ou différents, sont des entiers valant de 0 à 10 ;
- X- est un anion simple ou complexe, organique ou inorganique.
- those who have the following general formula (I):
- the quaternary ammonium salts of imidazoline, such as for example that of formula (II) below:
- the quaternary diammonium salts of formula (III):
- quaternary ammonium salts containing at least one ester function, for example those of formula (IV) below:
- R 15 is selected from alkyl C 1 -C 6 hydroxyalkyl or dihydroxyalkyl radicals C 1 -C 6 alkyl;
- R 16 is chosen from:
- The radical
- R 20 hydrocarbon radicals in C 1 -C 22 linear or branched, saturated or unsaturated,
- the hydrogen atom,
- R 18 is selected from:
- The radical
- linear or branched, saturated or unsaturated C 1 -C 6 hydrocarbon radicals R 22 ,
- the hydrogen atom,
- R 17 , R 19 and R 21 , which are identical or different, are chosen from linear or branched, saturated or unsaturated C 7 -C 21 hydrocarbon radicals;
- n, p and r, which may be identical or different, are integers ranging from 2 to 6;
- y is an integer from 1 to 10;
- x and z, which are identical or different, are integers ranging from 0 to 10;
- X- is a simple or complex anion, organic or inorganic.
Les radicaux alkyles R15 peuvent être linéaires ou ramifiés et plus particulièrement linéaires.The alkyl radicals R 15 may be linear or branched and more particularly linear.
De préférence R15 désigne un radical méthyle, éthyle, hydroxyéthyle ou dihydroxypropyle et plus particulièrement un radical méthyle ou éthyle.Preferably R 15 denotes a methyl, ethyl, hydroxyethyl or dihydroxypropyl radical and more particularly a methyl or ethyl radical.
Avantageusement, la somme x + y + z vaut de 1 à 10.Advantageously, the sum x + y + z is from 1 to 10.
Lorsque R16 est un radical R20 hydrocarboné, il peut être long et avoir de 12 à 22 atomes de carbone ou court et avoir de 1 à 3 atomes de carbone.When R 16 is a radical R 20 hydrocarbon, it may be long and contain from 12 to 22 carbon atoms or short and have from 1 to 3 carbon atoms.
Lorsque R18 est un radical R22 hydrocarboné, il a de préférence 1 à 3 atomes de carbone.When R 18 is a hydrocarbon radical R 22 , it preferably has 1 to 3 carbon atoms.
De préférence, x et z, identiques ou différents, valent 0 ou 1.Preferably, x and z, identical or different, are 0 or 1.
Avantageusement, y est égal à 1.Advantageously, y is 1.
De préférence, n, p et r, identiques ou différents, valent 2 ou 3 et encore plus particulièrement sont égaux à 2.Preferably, n, p and r, identical or different, are 2 or 3 and even more particularly are equal to 2.
L'anion est de préférence un halogénure (chlorure, bromure ou iodure) ou un alkylsulfate plus particulièrement méthylsulfate. On peut cependant utiliser le méthanesulfonate, le phosphate, le nitrate, le tosylate, un anion dérivé d'acide organique tel que l'acétate ou le lactate ou tout autre anion compatible avec l'ammonium à fonction ester.The anion is preferably a halide (chloride, bromide or iodide) or an alkyl sulphate more particularly methyl sulphate. However, it is possible to use methanesulphonate, phosphate, nitrate, tosylate, an anion derived from an organic acid such as acetate or lactate or any other anion compatible with ammonium with an ester function.
L'anion X- est encore plus particulièrement le chlorure ou le méthylsulfate.The X- anion is even more particularly chloride or methylsulfate.
Avantageusement, les radicaux hydrocarbonés sont linéaires.Advantageously, the hydrocarbon radicals are linear.
Parmi les sels d'ammonium quaternaire de formule (I) on préfère, d'une part, les chlorures de tétraalkylammonium comme par exemple les chlorures d'alkyltriméthylammonium, dans lesquels le radical alkyl comporte environ de 12 à 22 atomes de carbone, en particulier les chlorures de béhényl-triméthylammonium, de cétyltriméthylammonium, de benzyldiméthylstéaryl ammonium ou encore, d'autre part, le chlorure de palmitylamidopropyltriméthylammonium ou le chlorure de stéaramido-propyldiméthyl (myristyl acétate) ammonium commercialisé sous la dénomination "CERAPHYL 70" par la société VAN DYK.Among the quaternary ammonium salts of formula (I), tetraalkylammonium chlorides, for example alkyltrimethylammonium chlorides, in which the alkyl radical contains approximately from 12 to 22 carbon atoms, are particularly preferred. behenyltrimethylammonium, cetyltrimethylammonium, benzyldimethylstearylammonium chlorides or else palmitylamidopropyltrimethylammonium chloride or stearamido-propyldimethyl (myristylacetate) ammonium chloride sold under the name "Ceraphyl 70" by Van Dyk. .
On peut aussi utiliser les sels d'ammonium contenant au moins une fonction ester décrits dans le brevet
Des sels d'ammonium quaternaire de formule (IV) convenant à l'invention comprennent notamment le dipalmitoyléthyl hydroxyéthylmmonium méthosulfate, commercialisé par la société STEPAN sous la référence STEPANQUAT GA 90.Quaternary ammonium salts of formula (IV) that are suitable for use in the invention include, for example, dipalmitoylethyl hydroxyethylmethyl methosulphate, marketed by STEPAN under the reference STEPANQUAT GA 90.
Les molécules cationiques préférées utilisables dans la composition selon l'invention sont les composés de formule (I) ou de formule (IV).The preferred cationic molecules that may be used in the composition according to the invention are the compounds of formula (I) or of formula (IV).
On préférera le chlorure de cétyl triméthyl ammonium.Cetyl trimethyl ammonium chloride is preferred.
Comme expliqué précédemment, la ou les molécules cationiques peuvent également être choisies parmi les protéines cationiques.As explained above, the cationic molecule (s) may also be chosen from cationic proteins.
La ou les agents cosmétiques cationiques représentent avantageusement de 0,1% à 10% en poids du poids total de la composition, sous réserve que si l'agent cosmétique cationique est un sel de tétraalkylammonium ne contenant qu'une seule chaîne grasse, il soit présent à une teneur supérieure ou égale à 1 % en poids par rapport au poids total de la composition. De préférence, la ou les agents cosmétiques cationiques utilisés dans l'invention, quelle que soit leur nature, représentent de 1 à 10%, mieux de 1 à 5%, en poids du poids total de la composition.The cationic cosmetic agent or agents advantageously represent from 0.1% to 10% by weight of the total weight of the composition, provided that if the cationic cosmetic agent is a tetraalkylammonium salt containing only one fatty chain, it is present at a content greater than or equal to 1% by weight relative to the total weight of the composition. Preferably, the cationic cosmetic agent (s) used in the invention, whatever their nature, represent from 1 to 10%, better still from 1 to 5%, by weight of the total weight of the composition.
Selon un mode de réalisation particulier, lorsque le ou les agents cosmétiques cationiques comporte deux chaînes grasse, elle ne comporte aucune fonction amide.According to a particular embodiment, when the cationic cosmetic agent (s) comprise two fatty chains, it has no amide function.
A titre d'exemple, on peut citer comme agent cosmétique utile dans le procédé de l'invention, les polymères cationiques non colorants, non dérivés d'un sucre ou d'une silicone, présentant une densité de charge cationique supérieure à 1,5 meq/g.By way of example, mention may be made, as a cosmetic agent useful in the process of the invention, of cationic polymers which are non-coloring and which are not derived from a sugar or a silicone, and which have a cationic charge density greater than 1.5. meq / g.
La densité de charge peut être déterminée selon la méthode Kjedahl. Elle est mesurée en général à un pH de l'ordre de 3 à 9.The charge density can be determined according to the Kjedahl method. It is generally measured at a pH of the order of 3 to 9.
De préférence, les polymères cationiques utilisés dans la présente invention présentent une densité de charge cationique inférieure à 10 meq/g.Preferably, the cationic polymers used in the present invention have a cationic charge density of less than 10 meq / g.
Par "polymère cationique", on entend tout polymère contenant des groupements cationiques et/ou des groupements ionisables en groupements cationiques.By "cationic polymer" is meant any polymer containing cationic groups and / or ionizable groups into cationic groups.
Par non colorant, on entend ne présentant pas dans sa structure de chromophore présentant un maximum d'absorption dans le visible.By dye is meant not having in its chromophore structure having a maximum of absorption in the visible.
Par non dérivé d'un sucre ou d'une silicone, on entend ne comprenant pas dans sa structure de motif sucre (mono, oligo ou polysaccharide) ou de motif contenant un ou plusieurs atomes de silicium.By not derived from a sugar or a silicone, one understands not including in its structure of sugar unit (mono, oligo or polysaccharide) or of unit containing one or more silicon atoms.
Les polymères cationiques utilisables conformément à la présente invention peuvent être choisis parmi tous ceux déjà connus en soi comme améliorant les propriétés cosmétiques des cheveux traités par des compositions détergentes, à savoir notamment ceux décrits dans la demande de brevet
Les polymères cationiques préférés sont choisis parmi ceux qui contiennent des motifs comportant des groupements amine primaires, secondaires, tertiaires et/ou quaternaires pouvant soit faire partie de la chaîne principale polymère, soit être portés par un substituant latéral directement relié à celle-ci.The preferred cationic polymers are chosen from those containing units containing primary, secondary, tertiary and / or quaternary amine groups that may either be part of the main polymer chain or may be carried by a lateral substituent directly connected thereto.
Les polymères cationiques utilisés ont une masse moléculaire moyenne en poids supérieure à 105, de préférence supérieure à 106 et mieux encore comprise entre 106 et 108.The cationic polymers used have a weight average molecular weight of greater than 10 5 , preferably greater than 10 6 and more preferably 10 6 to 10 8 .
De préférence, les polymères cationiques ne sont pas des polyuréthanes.Preferably, the cationic polymers are not polyurethanes.
Parmi les polymères cationiques utilisables selon l'invention, on peut citer plus particulièrement les polymères du type polyamine, polyaminoamide et polyammonium quaternaire. Ce sont des produits connus.Among the cationic polymers that may be used according to the invention, mention may be made more particularly of polymers of the polyamine, polyaminoamide and quaternary polyammonium type. These are known products.
Les polymères du type polyamine, polyaminoamide, polyammonium quaternaire, que l'on peut utiliser dans la composition de la présente invention, sont ceux décrits dans les brevets français nos
- (1) les homopolymères ou copolymères dérivés d'esters ou d'amides acryliques ou méthacryliques et comportant au moins un des motifs de formules suivantes:
- R1 et R2, identiques ou différents, représentent un atome d'hydrogène ou un groupe alkyle ayant de 1 à 6 atomes de carbone, et de préférence un groupe méthyle ou éthyle ;
- R3, identiques ou différents, désignent un atome d'hydrogène ou un groupe CH3 ;
- les symboles A, identiques ou différents, représentent un groupe alkyle, linéaire ou ramifié, comportant de 1 à 6 atomes de carbone, de préférence 2 ou 3 atomes de carbone, ou un groupe hydroxyalkyle comportant de 1 à 4 atomes de carbone ;
- R4, R5, R6, identiques ou différents, représentent un groupe alkyle ayant de 1 à 18 atomes de carbone ou un groupe benzyle, et de préférence un groupe alkyle ayant de 1 à 6 atomes de carbone ;
- X désigne un anion dérivé d'un acide minéral ou organique tel qu'un anion méthosulfate ou un halogénure comme le chlorure ou le bromure.
Ainsi, parmi ces copolymères de la famille (1), on peut citer :- les copolymères d'acrylamide et de méthacrylate de diméthylaminoéthyle quaternisé au sulfate de diméthyle ou avec un halogénure de diméthyle,
- les copolymères d'acrylamide et de chlorure de méthacryloyloxyéthyltriméthylammonium décrits, par exemple, dans la demande de brevet
EP-A-080976 - les copolymères d'acrylamide et de méthosulfate de méthacryloyloxyéthyltriméthylammonium,
- les copolymères vinylpyrrolidone/acrylate ou méthacrylate de dialkylaminoalkyle quaternisés ou non. Ces polymères sont décrits en détail dans les brevets français
2077143 2393573 - les terpolymères méthacrylate de diméthylaminoéthyle/vinylcaprolactame/vinylpyrrolidone,
- les copolymères vinylpyrrolidone/méthacrylamidopropyl-di-méthylamine,
- les copolymères vinylpyrrolidone/méthacrylamide de diméthylaminopropyle quaternisé, et
- les polymères réticulés de sels de méthacryloyloxyalkyl(C1-C4) trialkyl(C1-C4)ammonium tels que les polymères obtenus par homopolymérisation du diméthylaminoéthylméthacrylate quaternisé par le chlorure de méthyle, ou par copolymérisation de l'acrylamide avec le diméthylaminoéthylméthacrylate quaternisé par le chlorure de méthyle, l'homo ou la copolymérisation étant suivie d'une réticulation par un composé à insaturation oléfinique, en particulier le méthylène bis acrylamide. On peut plus particulièrement utiliser un copolymère réticulé acrylamide/chlorure de méthacryloyloxyéthyl triméthylammonium (20/80 en poids) sous forme de dispersion contenant 50% en poids dudit copolymère dans de l'huile minérale. Cette dispersion est commercialisée sous le nom de « SALCARE® SC 92 » par la Société CIBA. On peut également utiliser un homopolymère réticulé du chlorure de méthacryloyloxyéthyl triméthylammonium contenant environ 50 % en poids de l'homopolymère dans de l'huile minérale ou dans un ester liquide. Ces dispersions sont commercialisées sous les noms de « SALCARE® SC 95 » et « SALCARE® SC 96 » par la Société CIBA.
- (2) Les polymères constitués de motifs pipérazinyle et de groupes divalents alkylène ou hydroxyalkylène à chaînes droites ou ramifiées, éventuellement interrompues par des atomes d'oxygène, de soufre, d'azote ou par des cycles aromatiques ou hétérocycliques, ainsi que les produits d'oxydation et/ou de quaternisation de ces polymères. De tels polymères sont notamment décrits dans les brevets français
2 162 025 2 280 361 - (3) Les polyaminoamides solubles dans l'eau, préparés en particulier par polycondensation d'un composé acide avec une polyamine ; ces polyaminoamides peuvent être réticulés par une épihalohydrine, un diépoxyde, un dianhydride, un dianhydride non saturé, un dérivé bis-insaturé, une bis-halohydrine, un bis-azétidinium, une bis-haloacyldiamine, un bis-halogénure d'alkyle ou encore par un oligomère résultant de la réaction d'un composé bifonctionnel réactif vis-à-vis d'une bis-halohydrine, d'un bis-azétidinium, d'une bis-haloacyldiamine, d'un bis-halogénure d'alkyle, d'une épilhalohydrine, d'un diépoxyde ou d'un dérivé bis-insaturé ; l'agent réticulant étant utilisé dans des proportions allant de 0,025 à 0,35 mole par groupement amine du polyaminoamide ; ces polyaminoamides peuvent être alkylés ou s'ils comportent une ou plusieurs fonctions amines tertiaires, quaternisées. De tels polymères sont notamment décrits dans les brevets français
2252840 2368508 - (4) Les dérivés de polyaminoamides résultant de la condensation de polyalkylènes-polyamines avec des acides polycarboxyliques, suivie d'une alkylation par des agents bifonctionnels. On peut citer, par exemple, les polymères acide adipique/dialkylaminohydroxyalkyl-dialkylènetriamine dans lesquels le groupe alkyle comporte de 1 à 4 atomes de carbone et désigne de préférence un groupe méthyle, éthyle, propyle, et le groupe alkylène comporte de 1 à 4 atomes de carbone, et désigne de préférence le groupe éthylène. De tels polymères sont notamment décrits dans le brevet français
1 583 363 - (5) Les polymères obtenus par réaction d'une polyalkylène-polyamine comportant deux groupements amine primaire et au moins un groupement amine secondaire, avec un acide dicarboxylique choisi parmi l'acide diglycolique et les acides dicarboxyliques aliphatiques saturés ayant de 3 à 8 atomes de carbone. Le rapport molaire entre la polyalkylène-polylamine et l'acide dicarboxylique étant compris entre 0,8 : 1 et 1,4 : 1 ; le polyaminoamide en résultant étant amené à réagir avec l'épichlorhydrine dans un rapport molaire d'épichlorhydrine par rapport au groupement amine secondaire du polyaminoamide compris entre 0,5 : 1 et 1,8 : 1. De tels polymères sont notamment décrits dans les brevets américains 3 227 615 et 2 961 347.
- (6) Les cyclopolymères d'alkyldiallylamine ou de dialkyldiallylammonium tels que les homopolymères ou copolymères comportant, comme constituant principal de la chaîne, des motifs répondant aux formules (Ia) ou (Ib) :
2 080 759 2 190 406
R10 et R11, indépendamment l'un de l'autre, désignent de préférence un groupement alkyle ayant de 1 à 4 atomes de carbone.
Parmi les polymères définis ci-dessus, on peut citer plus particulièrement l'homopolymère de diméthyldiallyl-ammonium notamment le chlorure de diméthyldiallylammonium vendu sous la dénomination "MERQUAT® 100" par la société NALCO (et ses homologues de faibles masses moléculaires moyenne en poids), les copolymères de chlorure de diallyldiméthylammonium et d'acrylamide commercialisés sous la dénomination "MERQUAT® 550" et les copolymères de diallyldiméthylammonium et d'acide acrylique tels que le "MERQUAT® 295" commercialisé par la société NALCO. - (7) Les polymères de diammonium quaternaire par exemple ceux contenant des motifs récurrents répondant à la formule (II) suivante :
- R13, R14, R15 et R16, identiques ou différents, représentent des groupes aliphatiques, alicycliques ou arylaliphatiques contenant de 1 à 20 atomes de carbone ou des groupes hydroxyalkylaliphatiques inférieurs, ou bien R13, R14, R15 et R16, ensemble ou séparément, constituent avec les atomes d'azote auxquels ils sont rattachés des hétérocycles contenant éventuellement un second hétéroatome autre que l'azote, ou bien R13, R14, R15 et R16 représentent un groupe alkyle en C1-C6, linéaire ou ramifié, substitué par un groupement nitrile, ester, acyle, amide ou -CO-O-R17-E ou -CO-NH-R17-E où R17 est un groupe alkylène et E un groupement ammonium quaternaire ;
- A1 et B1 représentent des groupements polyméthyléniques contenant de 2 à 20 atomes de carbone, pouvant être linéaires ou ramifiés, saturés ou insaturés, et pouvant contenir, liés à ou intercalés dans la chaîne principale, un ou plusieurs cycles aromatiques, ou un ou plusieurs atomes d'oxygène, de soufre ou des groupements sulfoxyde, sulfone, disulfure, amino, alkylamino, hydroxyle, ammonium quaternaire, uréido, amide ou ester, et
- X- désigne un anion dérivé d'un acide minéral ou organique tel que acétate, borate, citrate, tartrate, bisulfate, bisulfite, sulfate, phosphate, méthosulfate ou un halogénure comme le chlorure ou le bromure,
- A1, R13 et R15 peuvent former avec les deux atomes d'azote auxquels ils sont rattachés un cycle pipérazinique ; en outre, si A1 désigne un groupe alkylène ou hydroxyalkylène linéaire ou ramifié, saturé ou insaturé, B1 peut également désigner un groupement :
-
-(CH2)n-CO-E'-OC-(CH2)n-
dans lequel E' désigne :- a) un reste de glycol de formule -O-Z-O-, où Z désigne un groupe hydrocarboné linéaire ou ramifié, ou un groupement répondant à l'une des formules suivantes :
-(CH2-CH2-O)x-CH2-CH2-
-[CH2-CH(CH3)-O]y-CH2-CH(CH3)-
où x et y désignent un nombre entier de 1 à 4, représentant un degré de polymérisation défini et unique ou un nombre quelconque de 1 à 4 représentant un degré de polymérisation moyen ; - b) un reste de diamine bis-secondaire tel qu'un dérivé de pipérazine ;
- c) un reste de diamine bis-primaire de formule -NH-Y-NH-, où Y désigne un groupe hydrocarboné linéaire ou ramifié, ou bien le groupe divalent -CH2-CH2-S-S-CH2-CH2- ;
- d) un groupement uréylène de formule -NH-CO-NH-.
- a) un reste de glycol de formule -O-Z-O-, où Z désigne un groupe hydrocarboné linéaire ou ramifié, ou un groupement répondant à l'une des formules suivantes :
-
- De préférence, X- est un anion tel que halogénure et notamment le chlorure ou le bromure.
- Des polymères de ce type sont notamment décrits dans les brevets français
2 320 330 2 270 846 2 316 271 2 336 434 2 413 907 US 2 273 780 2 375 853 2 388 614 2 454 547 3 206 462 2 261 002 2 271 378 3 874 870 4 001 432 3 929 990 3 966 904 4 005 193 4 025 617 4 025 627 4 025 653 4 026 945 4 027 020 - On peut utiliser plus particulièrement les polymères qui sont constitués de motifs récurrents répondant à la formule :
- Un composé de formule (III) particulièrement préféré est celui pour lequel R13, R14, R15 et R16, représentent un radical méthyle et n = 3, p = 6 et X = CI, dénommé Hexadimethrine chloride selon la nomenclature INCI (CTFA).
- (8) Les polymères de polyammonium quaternaire constitués de motifs de formule (IV) :
- R18, R19, R20 et R21, identiques ou différents, représentent un atome d'hydrogène ou un groupe méthyle, éthyle, propyle, β-hydroxyéthyle, β-hydroxypropyle ou -CH2CH2(OCH2CH2)pOH, où p est égal à 0 ou à un nombre entier compris entre 1 et 6, sous réserve que R18, R19, R20 et R21 ne représentent pas simultanément un atome d'hydrogène,
- r et s, identiques ou différents, sont des nombres entiers compris entre 1 et 6,
- q est égal à 0 ou à un nombre entier compris entre 1 et 34,
- X- désigne un anion tel qu'un halogénure,
- A désigne un radical d'un dihalogénure ou représente de préférence
-CH2-CH2-O-CH2-CH2-.
- De tels composés sont notamment décrits dans la demande de brevet
EP-A-122 324
- (9) Les polymères quaternaires de vinylpyrrolidone et de vinylimidazole tels que par exemple les produits commercialisés sous les dénominations Luviquat® FC 905, FC 550 et FC 370 par la société B.A.S.F.
D'autres polymères cationiques utilisables dans le cadre de l'invention sont des polyalkylèneimines, en particulier des polyéthylèneimines, des polymères contenant des motifs vinylpyridine ou vinylpyridinium, des condensats de polyamines et d'épichlorhydrine, et des polyuréylènes quaternaires.
Selon une variante préférée, les polymères cationiques utilisables selon l'invention présentent une charge cationique permanente correspondant à la présence de motifs comportant des groupements amines quaternaires.
De préférence, les polymères cationiques de l'invention sont choisis parmi les cyclopolymères de dialkyldiallylammonium et les polymères de diammonium quaternaire de formule (II).
Le ou les polymères cationiques représentent avantageusement de 0,1 à 10%, mieux de 0,2 à 8%, en poids du poids total de la composition.
L'étape de traitement cosmétique peut être mise en oeuvre à partir d'une composition sous la forme d'une lotion, épaissie ou non, d'une crème, d'un gel, ou de toute autre forme appropriée.
Les compositions utilisées sont généralement des compositions aqueuses pouvant contenir des ingrédients habituellement utilisés dans les compositions cosmétiques, tels que des solvants, des agents tensioactifs, des épaississants, des agents conservateurs, des parfums ou tout autre additif utilisé dans ce type de composition.
Selon un mode de réalisation particulier, le procédé de l'invention peut comprendre une étape finale supplémentaire de mise en forme, par exemple une étape de lissage, notamment si l'étape précédente est une étape de traitement cosmétique.
Les exemples qui suivent illustrent le procédé de l'invention mis en oeuvre dans plusieurs applications cosmétiques capillaires.
- (1) homopolymers or copolymers derived from acrylic or methacrylic esters or amides and comprising at least one of the following units of formulas:
- R 1 and R 2 , which may be identical or different, represent a hydrogen atom or an alkyl group having from 1 to 6 carbon atoms, and preferably a methyl or ethyl group;
- R 3 , which may be identical or different, denote a hydrogen atom or a CH 3 group;
- the symbols A, identical or different, represent a linear or branched alkyl group containing from 1 to 6 carbon atoms, preferably 2 or 3 carbon atoms, or a hydroxyalkyl group containing from 1 to 4 carbon atoms;
- R 4 , R 5 , R 6 , which may be identical or different, represent an alkyl group having from 1 to 18 carbon atoms or a benzyl group, and preferably an alkyl group having from 1 to 6 carbon atoms;
- X denotes an anion derived from a mineral or organic acid such as a methosulphate anion or a halide such as chloride or bromide.
Thus, among these copolymers of the family (1), mention may be made of:- copolymers of acrylamide and of dimethylaminoethyl methacrylate quaternized with dimethyl sulphate or with a dimethyl halide,
- copolymers of acrylamide and methacryloyloxyethyltrimethylammonium chloride described, for example, in the patent application
EP-A-080976 - copolymers of acrylamide and methacryloyloxyethyltrimethylammonium methosulphate,
- the vinylpyrrolidone / dialkylaminoalkyl acrylate or methacrylate copolymers quaternized or otherwise. These polymers are described in detail in French patents
2077143 2393573 - dimethylaminoethyl methacrylate / vinylcaprolactam / vinylpyrrolidone terpolymers,
- vinylpyrrolidone / methacrylamidopropyl-dimethylamine copolymers,
- the vinylpyrrolidone / quaternized dimethylaminopropyl methacrylamide copolymers, and
- crosslinked polymers of methacryloyloxyalkyl (C 1 -C 4 ) trialkyl (C 1 -C 4 ) ammonium salts, such as the polymers obtained by homopolymerization of dimethylaminoethyl methacrylate quaternized with methyl chloride, or by copolymerization of acrylamide with quaternized dimethylaminoethyl methacrylate by methyl chloride, the homo or copolymerization being followed by crosslinking with an olefinically unsaturated compound, in particular methylenebisacrylamide. It is more particularly possible to use a crosslinked acrylamide / methacryloyloxyethyltrimethylammonium chloride copolymer (20/80 by weight) in the form of a dispersion containing 50% by weight of said copolymer in mineral oil. This dispersion is sold under the name "Salcare ® SC 92" by the company Ciba. It is also possible to use a crosslinked homopolymer of methacryloyloxyethyltrimethylammonium chloride containing about 50% by weight of the homopolymer in mineral oil or in a liquid ester. These dispersions are sold under the names "Salcare ® SC 95" and "Salcare ® SC 96" by the company Ciba.
- (2) Polymers consisting of piperazinyl units and straight or branched chain alkylene or hydroxyalkylene divalent groups, optionally interrupted by oxygen, sulfur, nitrogen or aromatic or heterocyclic rings, as well as oxidation and / or quaternization of these polymers. Such polymers are in particular described in French patents
2,162,025 2,280,361 - (3) water-soluble polyaminoamides prepared in particular by polycondensation of an acidic compound with a polyamine; these polyaminoamides may be crosslinked by an epihalohydrin, a diepoxide, a dianhydride, an unsaturated dianhydride, a bis-unsaturated derivative, a bis-halohydrin, a bis-azetidinium, a bis-haloacyldiamine, a bis-alkyl halide or else by an oligomer resulting from the reaction of a bifunctional compound which is reactive with a bis-halohydrin, a bis-azetidinium, a bis-haloacyldiamine, an alkyl bis-halogenide, epilhalohydrin, diepoxide or bis-unsaturated derivative; the crosslinking agent being used in proportions ranging from 0.025 to 0.35 moles per amine group of the polyaminoamide; these polyaminoamides can be alkylated or if they comprise one or more tertiary amine functional groups, quaternized. Such polymers are in particular described in French patents
2252840 2368508 - (4) Polyamino amide derivatives resulting from the condensation of polyalkylene polyamines with polycarboxylic acids followed by alkylation with difunctional agents. Mention may be made, for example, of adipic acid / dialkylaminohydroxyalkyl-dialkylenetriamine polymers in which the alkyl group contains from 1 to 4 carbon atoms and preferably denotes a methyl, ethyl or propyl group, and the alkylene group contains from 1 to 4 atoms. carbon, and preferably denotes the ethylene group. Such polymers are described in particular in the French patent
1,583,363 - (5) The polymers obtained by reacting a polyalkylene polyamine comprising two primary amine groups and at least one secondary amine group, with a dicarboxylic acid chosen from diglycolic acid and saturated aliphatic dicarboxylic acids having from 3 to 8 carbon atoms. carbon. The molar ratio between the polyalkylene-polylamine and the dicarboxylic acid being between 0.8: 1 and 1.4: 1; the polyaminoamide resulting therefrom being reacted with epichlorohydrin in a molar ratio of epichlorohydrin relative to the secondary amine group of the polyaminoamide of between 0.5: 1 and 1.8: 1. Such polymers are described in particular in the patents US 3,227,615 and 2,961,347.
- (6) Alkyldiallylamine or dialkyldiallylammonium cyclopolymers such as homopolymers or copolymers comprising, as main constituent of the chain, units corresponding to formulas (Ia) or (Ib):
2,080,759 2,190,406
R 10 and R 11 independently from each other, preferably denote an alkyl group having 1 to 4 carbon atoms.
Among the polymers defined above, there may be mentioned more particularly the homopolymer dimethyldiallyl ammonium including dimethyldiallylammonium chloride sold under the name "Merquat ® 100" by the company NALCO (and its counterparts low molecular weight average) , diallyldimethylammonium chloride copolymers and acrylamide sold under the name "Merquat ® 550" and copolymers of diallyldimethylammonium and acrylic acid such as "Merquat ® 295" sold by the company Nalco. - (7) Quaternary diammonium polymers, for example those containing recurring units corresponding to the following formula (II):
- R 13 , R 14 , R 15 and R 16 , which may be identical or different, represent aliphatic, alicyclic or arylaliphatic groups containing from 1 to 20 carbon atoms or lower hydroxyalkylaliphatic groups, or else R 13 , R 14 , R 15 and R 16, together or separately, constitute, with the nitrogen atoms to which they are attached, heterocycles optionally containing a second heteroatom other than nitrogen, or alternatively R 13, R 14, R 15 and R 16 represent an alkyl group having 1 -C 6 , linear or branched, substituted with a nitrile, ester, acyl, amide or -CO-OR 17 -E or -CO-NH-R 17 -E group where R 17 is an alkylene group and E a quaternary ammonium group ;
- A 1 and B 1 represent polymethylene groups containing from 2 to 20 carbon atoms, which may be linear or branched, saturated or unsaturated, and may contain, bound to or intercalated in the main chain, one or more aromatic rings, or one or a plurality of oxygen, sulfur or sulfoxide, sulfone, disulfide, amino, alkylamino, hydroxyl, quaternary ammonium, ureido, amide or ester groups, and
- X- denotes an anion derived from a mineral or organic acid such as acetate, borate, citrate, tartrate, bisulfate, bisulphite, sulfate, phosphate, methosulphate or a halide such as chloride or bromide,
- A 1 , R 13 and R 15 can form with the two nitrogen atoms to which they are attached a piperazine ring; in addition, if A 1 denotes a linear or branched, saturated or unsaturated alkylene or hydroxyalkylene group, B 1 can also denote a group:
-
- (CH 2 ) n -CO-E'-OC- (CH 2 ) n -
in which E 'designates:- a) a glycol residue of formula -OZO-, where Z denotes a linear or branched hydrocarbon group, or a group corresponding to one of the following formulas:
- (CH 2 -CH 2 -O) x -CH 2 -CH 2 -
- [CH 2 -CH (CH 3 ) -O] y -CH 2 -CH (CH 3 ) -
where x and y denote an integer from 1 to 4, representing a defined and unique degree of polymerization or any number from 1 to 4 representing an average degree of polymerization; - b) a bis-secondary diamine residue such as a piperazine derivative;
- c) a bis-primary diamine residue of formula -NH-Y-NH-, where Y denotes a linear or branched hydrocarbon group, or the divalent group -CH 2 -CH 2 -SS-CH 2 -CH 2 -;
- d) a ureylene group of formula -NH-CO-NH-.
- a) a glycol residue of formula -OZO-, where Z denotes a linear or branched hydrocarbon group, or a group corresponding to one of the following formulas:
-
- Preferably, X- is an anion such as halide and especially chloride or bromide.
- Polymers of this type are in particular described in French patents
2 320 330 2,270,846 2,316,271 2,336,434 2,413,907 US 2,273,780 2,375,853 2,388,614 2,454,547 3,206,462 2,261,002 2,271,378 3,874,870 4,001,432 3,929,990 3,966,904 4,005,193 4,025,617 4,025,627 4,025,653 4,026,945 4,027,020 - It is more particularly possible to use polymers which consist of recurring units corresponding to the formula:
- A compound of formula (III) which is particularly preferred is that for which R 13 , R 14 , R 15 and R 16 represent a methyl radical and n = 3, p = 6 and X = Cl, referred to as Hexadimethrine chloride according to the INCI nomenclature ( CTFA).
- (8) Quaternary polyammonium polymers consisting of units of formula (IV):
- R 18 , R 19 , R 20 and R 21 , which may be identical or different, represent a hydrogen atom or a methyl, ethyl, propyl, β-hydroxyethyl, β-hydroxypropyl or -CH 2 CH 2 (OCH 2 CH 2 ) group; p OH, where p is 0 or an integer between 1 and 6, with the proviso that R 18 , R 19 , R 20 and R 21 do not simultaneously represent a hydrogen atom,
- r and s, which are identical or different, are integers between 1 and 6,
- q is 0 or an integer from 1 to 34,
- X - denotes an anion such as a halide,
- A denotes a radical of a dihalide or is preferably
-CH 2 -CH 2 -O-CH 2 -CH 2 -.
- Such compounds are in particular described in the patent application
EP-A-122,324
- (9) Quaternary polymers of vinylpyrrolidone and of vinylimidazole, such as, for example, the products sold under the names Luviquat® FC 905, FC 550 and FC 370 by the company BASF
Other cationic polymers that may be used in the context of the invention are polyalkyleneimines, in particular polyethyleneimines, polymers containing vinylpyridine or vinylpyridinium units, condensates of polyamines and of epichlorohydrin, and quaternary polyureylenes.
According to a preferred variant, the cationic polymers that can be used according to the invention have a permanent cationic charge corresponding to the presence of units comprising quaternary amine groups.
Preferably, the cationic polymers of the invention are chosen from dialkyldiallylammonium cyclopolymers and quaternary diammonium polymers of formula (II).
The cationic polymer or polymers advantageously represent from 0.1 to 10%, better still from 0.2 to 8%, by weight of the total weight of the composition.
The cosmetic treatment step may be carried out from a composition in the form of a lotion, thickened or not, a cream, a gel, or any other suitable form.
The compositions used are generally aqueous compositions which may contain ingredients usually used in cosmetic compositions, such as solvents, surfactants, thickeners, preserving agents, perfumes or any other additive used in this type of composition.
According to a particular embodiment, the method of the invention may comprise an additional final shaping step, for example a smoothing step, especially if the preceding step is a cosmetic treatment step.
The examples which follow illustrate the process of the invention used in several cosmetic hair applications.
Des mèches de cheveux sont lavées et essorées. Un pré séchage des cheveux est alors mis en oeuvre jusqu'à obtenir un séchage d'environ 80%.Strands of hair are washed and wrung out. Pre-drying of the hair is then carried out until a drying of about 80% is obtained.
Sur ces mèches pré-séchées, on applique de la vapeur avec un débit de l'ordre de 3g/min, cette application de vapeur étant suivie de l'étape de mise en forme/lissage au moyen d'une pince lissante chauffée à au moins 100°C. Ce traitement est effectué mèche à mèche sur l'ensemble de la chevelure.On these pre-dried bits, steam is applied with a flow rate of the order of 3 g / min, this steam application being followed by the shaping / smoothing step by means of a smoothing pliers heated to minus 100 ° C. This treatment is done with a wick on the whole of the hair.
On applique ensuite une coloration d'oxydation permanente de nuance 5.64 (chatain clair rouge cuivré de la gamme Majirouge) qui consiste à appliquer sur les fibres capillaires une ou plusieurs bases d'oxydation et un ou plusieurs coupleurs bien connus du domaine de la coloration permanente, en présence d'un agent oxydant (peroxyde d'hydrogène).Subsequently, a 5.64 (copper red light brownish brown Majirouge) permanent oxidation dye is applied, which consists of applying to the hair fibers one or more oxidation bases and one or more couplers well known in the field of permanent coloring. in the presence of an oxidizing agent (hydrogen peroxide).
Parallèlement, on réalise à nouveau l'essai ci-dessus mais sans mettre en oeuvre l'étape d'application de vapeur ni l'étape de mise en forme dans les conditions précédentes mais en effectuant un lissage avec une pince de lissage chauffante classiqueAt the same time, the above test is carried out again, but without carrying out the steaming step nor the shaping step under the preceding conditions but by smoothing with a conventional heating smoothing plier.
On observe que les cheveux traités avec la vapeur sont plus brillants et plus lisses que ceux traités uniquement avec la pince de lissage chauffante classique.It is observed that the hair treated with steam are brighter and smoother than those treated only with the conventional heat straightener.
Après plusieurs shampooings, on constate une meilleure ténacité de la couleur sur les cheveux traités avec la vapeur.After several shampoos, there is a better tenacity of the color on the hair treated with steam.
Sur des mèches de cheveux lavés et essorés, on applique au pinceau une crème réductrice comprenant comprenant des sels d'acide thioglycolique. Après le temps de pose, les mèches sont à nouveau rincées puis essorées et préséchées à 80% au sèche-cheveux.On hair strands washed and dewatered, is applied by brush a reducing cream comprising thioglycolic acid salts. After the exposure time, the locks are rinsed again and then wrung out and dried at 80% with a hair dryer.
Sur ces fibres pré-séchées, on applique de la vapeur dans les conditions définies précédemment, cette application de vapeur étant suivie de l'étape de mise en forme/lissage au moyen d'une pince lissante chauffée à au moins 100°C. Ce traitement est effectué mèche à mèche sur l'ensemble de la chevelure.On these pre-dried fibers, steam is applied under the conditions defined above, this steam application being followed by the shaping / smoothing step by means of a smoothing pliers heated to at least 100 ° C. This treatment is done with a wick on the whole of the hair.
On applique ensuite le fixateur sur la totalité de la chevelure. Après un temps de pose, les mèches sont rincées, essorées et séchées.The fixative is then applied to the entire hair. After a period of exposure, the locks are rinsed, dewatered and dried.
Parallèlement, on réalise à nouveau l'essai ci-dessus mais sans mettre en oeuvre l'étape d'application de vapeur ni l'étape de mise en forme dans les conditions précédentes mais en effectuant un lissage avec une pince de lissage chauffante classiqueAt the same time, the above test is carried out again but without carrying out the steam application step or the shaping step under the conditions but smoothing with conventional heated smoothing pliers
On observe que les cheveux traités avec le procédé de l'invention sont plus brillants et plus lisses au toucher et visuellement. Les cheveux sont de plus nettement mieux défrisés avec le procédé de l'invention.It is observed that the hair treated with the process of the invention is brighter and smoother to the touch and visually. The hair is also much better relaxed with the method of the invention.
Après plusieurs shampooings, on constate que l'effet lissant persiste lorsqu'il a été obtenu avec le procédé de l'invention.After several shampoos, it is found that the smoothing effect persists when it has been obtained with the method of the invention.
Sur des mèches de cheveux lavées et essorées, on applique le soin «Ciment thermique» de Kerastase qui est un produit de soin non rincé comprenant des silicones et des tensio-actifs cationiques. Un pré séchage des mèches est alors mis en oeuvre jusqu'à obtenir un séchage d'environ 80%.On wicks of hair washed and dewatered, one applies the care "Thermal Cement" of Kerastase which is a care product not rinsed including silicones and cationic surfactants. A pre-drying of the locks is then used until a drying of about 80% is obtained.
Sur ces mèches pré-séchées, on applique de la vapeur dans les conditions définies précédemment, cette application de vapeur étant suivie de l'étape de mise en forme/lissage au moyen d'une pince lissante chauffée à au moins 100°C.On these pre-dried locks, steam is applied under the conditions defined above, this steam application being followed by the shaping / smoothing step by means of a smoothing tongs heated to at least 100 ° C.
Parallèlement, on réalise à nouveau l'essai ci-dessus mais sans mettre en oeuvre l'étape d'application de vapeur ni l'étape de mise en forme dans les conditions précédentes mais en effectuant un lissage avec une pince de lissage chauffante classiqueAt the same time, the above test is carried out again, but without carrying out the steaming step nor the shaping step under the preceding conditions but by smoothing with a conventional heating smoothing plier.
On observe que les mèches traitées selon le procédé de la présente invention sont plus brillants et plus lisses au toucher et visuellement. Les cheveux sont nettement mieux lissés.It is observed that the locks treated according to the process of the present invention are brighter and smoother to the touch and visually. The hair is much better smoothed.
Après plusieurs shampooings, on constate que l'effet lissant persiste lorsqu'il a été obtenu avec le procédé de l'invention.After several shampoos, it is found that the smoothing effect persists when it has been obtained with the method of the invention.
Sur des mèches de cheveux lavées et essorées, on applique un après shampoing « Nutricéramide « de la marque Elsève. Après le temps de pose, les mèches sont rincées puis essorées et préséchées à 80% au sèche-cheveux.On hair strands washed and wrung out, we apply a shampoo "Nutricéramide" of the mark Elsève. After the exposure time, the locks are rinsed and then wrung out and dried at 80% with a hair dryer.
Sur ces mèches pré-séchées, on applique de la vapeur dans les conditions définies précédemment cette application de vapeur étant suivie de l'étape de mise en forme/lissage au moyen d'une pince lissante chauffée à au moins 100°C.On these pre-dried locks, steam is applied under the conditions defined above, this steam application being followed by the shaping / smoothing step by means of a smoothing tongs heated to at least 100 ° C.
Parallèlement, on réalise à nouveau l'essai ci-dessus mais sans mettre en oeuvre l'étape d'application de vapeur ni l'étape de mise en forme dans les conditions précédentes mais en effectuant un lissage avec une pince de lissage chauffante classiqueAt the same time, the above test is carried out again, but without carrying out the steaming step nor the shaping step under the preceding conditions but by smoothing with a conventional heating smoothing plier.
On observe que les mèches traitées avec le procédé de la présente invention sont plus brillants et plus lisses au toucher et visuellement. Les mèches sont nettement mieux lissées.It is observed that the locks treated with the process of the present invention are brighter and smoother to the touch and visually. The locks are much better smoothed.
Après plusieurs shampooings, on constate que l'effet lissant persiste lorsqu'il a été obtenu avec le procédé de l'invention.After several shampoos, it is found that the smoothing effect persists when it has been obtained with the method of the invention.
Des mèches de cheveux sont lavées et essorées. Un pré séchage des cheveux est alors mis en oeuvre jusqu'à obtenir un séchage d'environ 80%.Strands of hair are washed and wrung out. Pre-drying of the hair is then carried out until a drying of about 80% is obtained.
Sur ces mèches pré-séchées, on applique de la vapeur dans les conditions définies dans la présente invention, cette application de vapeur étant suivie de l'étape de mise en forme/lissage au moyen d'une pince lissante chauffée à au moins 100°C. Ce traitement est effectué mèche à mèche sur l'ensemble de la chevelure.On these pre-dried locks, steam is applied under the conditions defined in the present invention, this steam application being followed by the shaping / smoothing step by means of a smoothing tongs heated to at least 100 °. vs. This treatment is done with a wick on the whole of the hair.
Parallèlement, on réalise à nouveau l'essai ci-dessus mais sans mettre en oeuvre l'étape d'application de vapeur en effectuant un lissage avec une pince de lissage chauffante classique sans application de vapeurAt the same time, the above test is carried out again but without carrying out the steaming step by smoothing with a conventional heating smoothing plier without the application of steam.
On observe que les cheveux traités avec la vapeur sont plus brillants et plus lisses que ceux traités uniquement avec la pince de lissage chauffante classique.It is observed that the hair treated with steam are brighter and smoother than those treated only with the conventional heat straightener.
Claims (13)
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0952200A FR2943895B1 (en) | 2009-04-03 | 2009-04-03 | PROCESS FOR TREATING HAIR WITH WATER VAPOR. |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP2236053A1 true EP2236053A1 (en) | 2010-10-06 |
| EP2236053B1 EP2236053B1 (en) | 2016-03-30 |
| EP2236053B2 EP2236053B2 (en) | 2021-03-03 |
Family
ID=41665324
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP10158979.4A Active EP2236053B2 (en) | 2009-04-03 | 2010-04-01 | Method for the steam treatment of hair |
Country Status (9)
| Country | Link |
|---|---|
| US (2) | US9603429B2 (en) |
| EP (1) | EP2236053B2 (en) |
| JP (4) | JP2010241812A (en) |
| KR (1) | KR101235394B1 (en) |
| CN (2) | CN105831947A (en) |
| BR (1) | BRPI1001018B1 (en) |
| ES (1) | ES2576735T3 (en) |
| FR (1) | FR2943895B1 (en) |
| MX (1) | MX338820B (en) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR3032119B1 (en) * | 2015-01-29 | 2017-01-20 | Oreal | METHOD FOR TREATING HAIR WITH AT LEAST ONE SILICONE FUNCTIONALIZED BY AT LEAST ONE ALCOXYSILANE MOTIF, AND STEAM |
| FR3032117B1 (en) | 2015-01-29 | 2018-03-16 | L'oreal | COMPOSITION COMPRISING A SILICONE FUNCTIONALIZED BY AT LEAST ONE ALCOXYSILANE MOTIF AND A NONIONIC OR ANIONIC FIXING POLYMER |
| FR3032118B1 (en) | 2015-01-29 | 2018-03-16 | L'oreal | AEROSOL COMPOSITION COMPRISING A SILICONE FUNCTIONALIZED BY AT LEAST ONE ALCOXYSILANE MOTIF AND AN AMPHOTERE AND / OR CATIONIC FIXING POLYMER |
| FR3039374B1 (en) * | 2015-07-29 | 2019-12-20 | L'oreal | PROCESS FOR TREATING HAIR FIBERS |
| FR3043549B1 (en) * | 2015-11-12 | 2018-12-07 | L'oreal | PROCESS FOR TREATING KERATIN FIBERS WITH A COMPOSITION COMPRISING A RETICULATING AGENT AND A STEAM IRON |
| JP6317515B1 (en) * | 2017-10-23 | 2018-04-25 | 株式会社フルメタルジャケット | How to apply straight perm to hair |
Citations (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2261002A (en) | 1941-06-17 | 1941-10-28 | Du Pont | Organic nitrogen compounds |
| US2271378A (en) | 1939-08-30 | 1942-01-27 | Du Pont | Pest control |
| US2273780A (en) | 1939-12-30 | 1942-02-17 | Du Pont | Wax acryalte ester blends |
| US2375853A (en) | 1942-10-07 | 1945-05-15 | Du Pont | Diamine derivatives |
| US2388614A (en) | 1942-05-05 | 1945-11-06 | Du Pont | Disinfectant compositions |
| US2454547A (en) | 1946-10-15 | 1948-11-23 | Rohm & Haas | Polymeric quaternary ammonium salts |
| US3206462A (en) | 1962-10-31 | 1965-09-14 | Dow Chemical Co | Quaternary poly(oxyalkylene)alkylbis(diethylenetriamine) compounds |
| FR1583363A (en) | 1967-09-28 | 1969-10-24 | ||
| FR2077143A5 (en) | 1970-01-30 | 1971-10-15 | Gaf Corp | |
| FR2080759A1 (en) | 1970-02-25 | 1971-11-19 | Gillette Co | |
| FR2162025A1 (en) | 1971-11-29 | 1973-07-13 | Oreal | |
| FR2190406A2 (en) | 1972-06-29 | 1974-02-01 | Gillette Co | |
| US3874870A (en) | 1973-12-18 | 1975-04-01 | Mill Master Onyx Corp | Microbiocidal polymeric quarternary ammonium compounds |
| FR2252840A1 (en) | 1973-11-30 | 1975-06-27 | Oreal | |
| FR2270846A1 (en) | 1974-05-16 | 1975-12-12 | Oreal | |
| US3929990A (en) | 1973-12-18 | 1975-12-30 | Millmaster Onyx Corp | Microbiocidal polymeric quaternary ammonium compounds |
| FR2280361A2 (en) | 1974-08-02 | 1976-02-27 | Oreal | HAIR TREATMENT AND CONDITIONING COMPOSITIONS |
| US3966904A (en) | 1974-10-03 | 1976-06-29 | Millmaster Onyx Corporation | Quaternary ammonium co-polymers for controlling the proliferation of bacteria |
| US4001432A (en) | 1974-10-29 | 1977-01-04 | Millmaster Onyx Corporation | Method of inhibiting the growth of bacteria by the application thereto of capped polymers |
| US4005193A (en) | 1974-08-07 | 1977-01-25 | Millmaster Onyx Corporation | Microbiocidal polymeric quaternary ammonium compounds |
| FR2316271A1 (en) | 1975-07-04 | 1977-01-28 | Oreal | NEW QUATERNIZED POLYMERS, THEIR PREPARATION PROCESS, AND THEIR APPLICATION |
| FR2320330A1 (en) | 1974-01-25 | 1977-03-04 | Calgon Corp | COMPOSITION OF FUNCTIONAL IONEN POLYMERS AND THEIR USE |
| US4025617A (en) | 1974-10-03 | 1977-05-24 | Millmaster Onyx Corporation | Anti-microbial quaternary ammonium co-polymers |
| US4025627A (en) | 1973-12-18 | 1977-05-24 | Millmaster Onyx Corporation | Microbiocidal polymeric quaternary ammonium compounds |
| US4025653A (en) | 1975-04-07 | 1977-05-24 | Millmaster Onyx Corporation | Microbiocidal polymeric quaternary ammonium compounds |
| US4027020A (en) | 1974-10-29 | 1977-05-31 | Millmaster Onyx Corporation | Randomly terminated capped polymers |
| US4026945A (en) | 1974-10-03 | 1977-05-31 | Millmaster Onyx Corporation | Anti-microbial quaternary ammonium co-polymers |
| FR2336434A1 (en) | 1975-12-23 | 1977-07-22 | Ciba Geigy Ag | QUATERNARY POLYMERIC AMMONIUM SALTS, THEIR PREPARATION PROCESS AND THEIR USE |
| FR2368508A2 (en) | 1977-03-02 | 1978-05-19 | Oreal | HAIR CONDITIONING COMPOSITION |
| FR2383660A1 (en) | 1977-03-15 | 1978-10-13 | Oreal | COMPOSITION AND PROCESS FOR TREATMENT OF KERATINIC MATERIALS WITH CATIONIC AND ANIONIC POLYMERS |
| FR2393573A1 (en) | 1977-06-10 | 1979-01-05 | Gaf Corp | HAIR PREPARATIONS CONTAINING A VINYLPYRROLIDONE COPOLYMER |
| FR2413907A1 (en) | 1977-09-20 | 1979-08-03 | Oreal | COSMETIC COMPOSITIONS BASED ON QUATERNARY POLYAMMONIUM POLYMERS |
| FR2470596A1 (en) | 1979-11-28 | 1981-06-12 | Oreal | COMPOSITION FOR THE TREATMENT OF KERATIN FIBERS BASED ON AMPHOTERIC POLYMERS AND CATIONIC POLYMERS |
| FR2505348A1 (en) | 1981-05-08 | 1982-11-12 | Oreal | COMPOSITION IN THE FORM OF AEROSOL FOAM BASED ON CATIONIC POLYMER AND ANIONIC POLYMER |
| EP0080976A1 (en) | 1981-11-30 | 1983-06-08 | Ciba-Geigy Ag | Mixtures of quaternary polymeric acrylic ammonium salts, quaternary mono- or oligomeric ammonium salts and surfactants, their preparation and their use in cosmetic compositions |
| FR2519863A1 (en) | 1982-01-15 | 1983-07-22 | Oreal | COSMETIC COMPOSITION FOR THE TREATMENT OF KERATIN FIBERS AND METHOD FOR THE TREATMENT THEREOF |
| FR2542997A1 (en) | 1983-03-23 | 1984-09-28 | Oreal | A THICKENED OR GELIFIED HAIR CONDITIONING COMPOSITION CONTAINING AT LEAST ONE CATIONIC POLYMER, AT LEAST ONE ANIONIC POLYMER AND AT LEAST ONE XANTHAN GUM |
| EP0122324A1 (en) | 1983-04-15 | 1984-10-24 | Miranol Inc. | Polyquaternary ammonium compounds and cosmetic compositions containing them |
| US4481961A (en) * | 1980-05-30 | 1984-11-13 | L'oreal | Method for setting hair and a device for carrying out the said method |
| FR2598611A1 (en) | 1986-05-16 | 1987-11-20 | Oreal | COSMETIC COMPOSITIONS CONTAINING A CATIONIC POLYMER AND AN ANIONIC POLYMER AS A THICKENING AGENT |
| US4874554A (en) | 1986-07-10 | 1989-10-17 | Henkel Kommanditgesellschaft Auf Aktien | Quaternary ammonium compounds |
| EP0337354A1 (en) | 1988-04-12 | 1989-10-18 | Kao Corporation | Low-irritation detergent composition |
| US5046516A (en) | 1988-10-31 | 1991-09-10 | George Barradas | Hair curling iron |
| US5091629A (en) | 1990-10-09 | 1992-02-25 | Mcgee Robert J | Hair curling iron for creating an inwardly bending curl and heating apparatus therefor |
| US5223694A (en) | 1991-01-28 | 1993-06-29 | Matsushita Electric Works, Ltd. | Hair styling iron for straightening and curling |
| EP0659395A1 (en) | 1993-12-22 | 1995-06-28 | L'oreal | Process for temporary hair deformation |
| EP0659397A1 (en) | 1993-12-22 | 1995-06-28 | L'oreal | Process for direct coloration of human keratinic fibers by natural dyes and water steam |
| EP0659396A1 (en) | 1993-12-22 | 1995-06-28 | L'oreal | Process for oxidative hair dying using steam |
| EP0659393A1 (en) | 1993-12-22 | 1995-06-28 | L'oreal | Process for non-permanent hair deformation |
| US5520200A (en) * | 1993-12-22 | 1996-05-28 | L'oreal | Process for the non-lasting reshaping of keratinous fibres |
| US5957140A (en) | 1998-11-19 | 1999-09-28 | Mcgee; Robert J. | Hair styling iron for straightening and curling |
| WO2003039291A2 (en) * | 2001-11-02 | 2003-05-15 | Kenra, Llc | Hair relaxer |
| US20040000319A1 (en) * | 2002-06-26 | 2004-01-01 | The Procter & Gamble Company | Device for achieving smooth and straight hair |
| WO2004002262A1 (en) | 2002-06-26 | 2004-01-08 | Matsushita Electric Works, Ltd. | Hair iron |
| US7044139B2 (en) | 2003-08-01 | 2006-05-16 | Hometek International Ltd. | Hair curling iron and shaping tool |
| FR2921804A1 (en) * | 2007-10-03 | 2009-04-10 | Seb Sa | Portable hair treating apparatus for e.g. bleaching application, has steam generator supplied with liquid i.e. water, by electrical pump of liquid supply units to produce steam flow, which has specific range of value in gram per minute |
Family Cites Families (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3745306A (en) | 1971-04-07 | 1973-07-10 | Shigeru Kawayama | Steam generating instrument for hairdressing |
| US4341229A (en) * | 1974-06-10 | 1982-07-27 | L'oreal | Method and apparatus for setting hair |
| LU70281A1 (en) * | 1974-06-10 | 1976-04-13 | ||
| US4205692A (en) | 1978-06-26 | 1980-06-03 | Losenno Luigi G | Vapor hood |
| US4911185A (en) | 1983-08-19 | 1990-03-27 | Gino, Ltd. | Method and system for the application of hair treatment solution |
| JPS62181232U (en) | 1986-05-06 | 1987-11-17 | ||
| JPH0751119A (en) | 1993-08-20 | 1995-02-28 | Kazumi Segami | Operation method for permanent treatment, operation method for iron permanent wave and treatment agent |
| JPH07163416A (en) * | 1993-12-13 | 1995-06-27 | Hoyu Co Ltd | Hair treating method |
| FR2713895B1 (en) * | 1993-12-22 | 1996-02-16 | Oreal | Set intended for working steam hair. |
| FR2723311B1 (en) * | 1994-08-02 | 1996-10-25 | Oreal | PROCESS FOR TREATING HUMAN KERATINIC FIBERS USING FATTY CHAIN AMIDES AND WATER VAPOR |
| US5585307A (en) | 1995-02-27 | 1996-12-17 | Taiwan Semiconductor Manufacturing Company Ltd. | Forming a semi-recessed metal for better EM and Planarization using a silo mask |
| DE19810598A1 (en) | 1998-03-12 | 1999-09-23 | Braun Gmbh | Hair treatment device which allows a treatment agent to be applied |
| US6521219B1 (en) | 2000-09-06 | 2003-02-18 | Phild Co., Ltd. | Method of repairing or restoring damaged or imperfect hair |
| CN100344245C (en) * | 2002-06-26 | 2007-10-24 | 宝洁公司 | Device for achieving smooth and straight hair |
| JP2005179210A (en) * | 2003-12-17 | 2005-07-07 | Lion Corp | Hair setting agent composition, two-package hair setting agent composition, and composition for hair setting agent |
| FR2866232B1 (en) * | 2004-02-12 | 2007-02-09 | Oreal | PROCESS FOR FORMING KERATINIC FIBERS BY APPLYING A COSMETIC COMPOSITION COMPRISING AN ANIONIC FIXING POLYMER AND A NON-IONIC COMPOUND AND APPLYING A HEATING MEANS, CORRESPONDING COMPOSITION |
| US20050196368A1 (en) * | 2004-02-12 | 2005-09-08 | Ludivine Laurent | Cosmetic composition comprising at least one anionic fixing polymer and at least one nonionic compound, and a process for shaping keratin fibers therewith by applying heat |
| JP2005230350A (en) * | 2004-02-20 | 2005-09-02 | Akihiro Watanabe | Perming device and method |
| DE602005011383D1 (en) | 2004-11-01 | 2009-01-15 | K I C A Inc | hair iron |
| FR2887440B1 (en) * | 2005-06-28 | 2009-07-10 | Oreal | PROCESS FOR PHOTOPROTECTIVE TREATMENT OF KERATIN FIBERS BY HEAT APPLICATION |
| TWI377921B (en) | 2006-08-08 | 2012-12-01 | Takara Belmont | Permanent wave treatment method and apparatus thereof |
| FR2921805B1 (en) * | 2007-10-03 | 2011-04-01 | Oreal | METHOD OF TREATING HAIR WITH WATER VAPOR |
| FR2921803B1 (en) * | 2007-10-03 | 2011-04-29 | Seb Sa | APPARATUS FOR TREATING HAIR WITH STEAM |
| FR2967017B1 (en) | 2010-11-05 | 2014-04-04 | Seb Sa | VAPOR HAIRSTYLE APPARATUS |
| FR2967020B1 (en) | 2010-11-05 | 2012-12-21 | Seb Sa | STEAM HAIRSTUFFING APPARATUS COMPRISING A BASE AND A PORTABLE UNIT |
-
2009
- 2009-04-03 FR FR0952200A patent/FR2943895B1/en not_active Expired - Fee Related
-
2010
- 2010-03-31 BR BRPI1001018-1A patent/BRPI1001018B1/en active IP Right Grant
- 2010-03-31 MX MX2010003623A patent/MX338820B/en active IP Right Grant
- 2010-04-01 ES ES10158979.4T patent/ES2576735T3/en active Active
- 2010-04-01 EP EP10158979.4A patent/EP2236053B2/en active Active
- 2010-04-02 KR KR1020100030464A patent/KR101235394B1/en active IP Right Grant
- 2010-04-02 CN CN201610381774.2A patent/CN105831947A/en active Pending
- 2010-04-02 CN CN201010174103A patent/CN101856172A/en active Pending
- 2010-04-02 JP JP2010085876A patent/JP2010241812A/en active Pending
- 2010-04-02 US US12/753,341 patent/US9603429B2/en active Active
-
2015
- 2015-09-25 JP JP2015188560A patent/JP2016025937A/en active Pending
-
2017
- 2017-02-07 US US15/426,567 patent/US20170143091A1/en not_active Abandoned
- 2017-09-08 JP JP2017172953A patent/JP7020826B2/en active Active
-
2020
- 2020-09-10 JP JP2020151979A patent/JP2020203128A/en active Pending
Patent Citations (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2271378A (en) | 1939-08-30 | 1942-01-27 | Du Pont | Pest control |
| US2273780A (en) | 1939-12-30 | 1942-02-17 | Du Pont | Wax acryalte ester blends |
| US2261002A (en) | 1941-06-17 | 1941-10-28 | Du Pont | Organic nitrogen compounds |
| US2388614A (en) | 1942-05-05 | 1945-11-06 | Du Pont | Disinfectant compositions |
| US2375853A (en) | 1942-10-07 | 1945-05-15 | Du Pont | Diamine derivatives |
| US2454547A (en) | 1946-10-15 | 1948-11-23 | Rohm & Haas | Polymeric quaternary ammonium salts |
| US3206462A (en) | 1962-10-31 | 1965-09-14 | Dow Chemical Co | Quaternary poly(oxyalkylene)alkylbis(diethylenetriamine) compounds |
| FR1583363A (en) | 1967-09-28 | 1969-10-24 | ||
| FR2077143A5 (en) | 1970-01-30 | 1971-10-15 | Gaf Corp | |
| FR2080759A1 (en) | 1970-02-25 | 1971-11-19 | Gillette Co | |
| FR2162025A1 (en) | 1971-11-29 | 1973-07-13 | Oreal | |
| FR2190406A2 (en) | 1972-06-29 | 1974-02-01 | Gillette Co | |
| FR2252840A1 (en) | 1973-11-30 | 1975-06-27 | Oreal | |
| US3874870A (en) | 1973-12-18 | 1975-04-01 | Mill Master Onyx Corp | Microbiocidal polymeric quarternary ammonium compounds |
| US4025627A (en) | 1973-12-18 | 1977-05-24 | Millmaster Onyx Corporation | Microbiocidal polymeric quaternary ammonium compounds |
| US3929990A (en) | 1973-12-18 | 1975-12-30 | Millmaster Onyx Corp | Microbiocidal polymeric quaternary ammonium compounds |
| FR2320330A1 (en) | 1974-01-25 | 1977-03-04 | Calgon Corp | COMPOSITION OF FUNCTIONAL IONEN POLYMERS AND THEIR USE |
| FR2270846A1 (en) | 1974-05-16 | 1975-12-12 | Oreal | |
| FR2280361A2 (en) | 1974-08-02 | 1976-02-27 | Oreal | HAIR TREATMENT AND CONDITIONING COMPOSITIONS |
| US4005193A (en) | 1974-08-07 | 1977-01-25 | Millmaster Onyx Corporation | Microbiocidal polymeric quaternary ammonium compounds |
| US3966904A (en) | 1974-10-03 | 1976-06-29 | Millmaster Onyx Corporation | Quaternary ammonium co-polymers for controlling the proliferation of bacteria |
| US4025617A (en) | 1974-10-03 | 1977-05-24 | Millmaster Onyx Corporation | Anti-microbial quaternary ammonium co-polymers |
| US4026945A (en) | 1974-10-03 | 1977-05-31 | Millmaster Onyx Corporation | Anti-microbial quaternary ammonium co-polymers |
| US4001432A (en) | 1974-10-29 | 1977-01-04 | Millmaster Onyx Corporation | Method of inhibiting the growth of bacteria by the application thereto of capped polymers |
| US4027020A (en) | 1974-10-29 | 1977-05-31 | Millmaster Onyx Corporation | Randomly terminated capped polymers |
| US4025653A (en) | 1975-04-07 | 1977-05-24 | Millmaster Onyx Corporation | Microbiocidal polymeric quaternary ammonium compounds |
| FR2316271A1 (en) | 1975-07-04 | 1977-01-28 | Oreal | NEW QUATERNIZED POLYMERS, THEIR PREPARATION PROCESS, AND THEIR APPLICATION |
| FR2336434A1 (en) | 1975-12-23 | 1977-07-22 | Ciba Geigy Ag | QUATERNARY POLYMERIC AMMONIUM SALTS, THEIR PREPARATION PROCESS AND THEIR USE |
| FR2368508A2 (en) | 1977-03-02 | 1978-05-19 | Oreal | HAIR CONDITIONING COMPOSITION |
| FR2383660A1 (en) | 1977-03-15 | 1978-10-13 | Oreal | COMPOSITION AND PROCESS FOR TREATMENT OF KERATINIC MATERIALS WITH CATIONIC AND ANIONIC POLYMERS |
| FR2393573A1 (en) | 1977-06-10 | 1979-01-05 | Gaf Corp | HAIR PREPARATIONS CONTAINING A VINYLPYRROLIDONE COPOLYMER |
| FR2413907A1 (en) | 1977-09-20 | 1979-08-03 | Oreal | COSMETIC COMPOSITIONS BASED ON QUATERNARY POLYAMMONIUM POLYMERS |
| FR2470596A1 (en) | 1979-11-28 | 1981-06-12 | Oreal | COMPOSITION FOR THE TREATMENT OF KERATIN FIBERS BASED ON AMPHOTERIC POLYMERS AND CATIONIC POLYMERS |
| US4481961A (en) * | 1980-05-30 | 1984-11-13 | L'oreal | Method for setting hair and a device for carrying out the said method |
| FR2505348A1 (en) | 1981-05-08 | 1982-11-12 | Oreal | COMPOSITION IN THE FORM OF AEROSOL FOAM BASED ON CATIONIC POLYMER AND ANIONIC POLYMER |
| EP0080976A1 (en) | 1981-11-30 | 1983-06-08 | Ciba-Geigy Ag | Mixtures of quaternary polymeric acrylic ammonium salts, quaternary mono- or oligomeric ammonium salts and surfactants, their preparation and their use in cosmetic compositions |
| FR2519863A1 (en) | 1982-01-15 | 1983-07-22 | Oreal | COSMETIC COMPOSITION FOR THE TREATMENT OF KERATIN FIBERS AND METHOD FOR THE TREATMENT THEREOF |
| FR2542997A1 (en) | 1983-03-23 | 1984-09-28 | Oreal | A THICKENED OR GELIFIED HAIR CONDITIONING COMPOSITION CONTAINING AT LEAST ONE CATIONIC POLYMER, AT LEAST ONE ANIONIC POLYMER AND AT LEAST ONE XANTHAN GUM |
| EP0122324A1 (en) | 1983-04-15 | 1984-10-24 | Miranol Inc. | Polyquaternary ammonium compounds and cosmetic compositions containing them |
| FR2598611A1 (en) | 1986-05-16 | 1987-11-20 | Oreal | COSMETIC COMPOSITIONS CONTAINING A CATIONIC POLYMER AND AN ANIONIC POLYMER AS A THICKENING AGENT |
| US4874554A (en) | 1986-07-10 | 1989-10-17 | Henkel Kommanditgesellschaft Auf Aktien | Quaternary ammonium compounds |
| EP0337354A1 (en) | 1988-04-12 | 1989-10-18 | Kao Corporation | Low-irritation detergent composition |
| US5046516A (en) | 1988-10-31 | 1991-09-10 | George Barradas | Hair curling iron |
| US5091629A (en) | 1990-10-09 | 1992-02-25 | Mcgee Robert J | Hair curling iron for creating an inwardly bending curl and heating apparatus therefor |
| US5223694A (en) | 1991-01-28 | 1993-06-29 | Matsushita Electric Works, Ltd. | Hair styling iron for straightening and curling |
| EP0659395A1 (en) | 1993-12-22 | 1995-06-28 | L'oreal | Process for temporary hair deformation |
| EP0659397A1 (en) | 1993-12-22 | 1995-06-28 | L'oreal | Process for direct coloration of human keratinic fibers by natural dyes and water steam |
| EP0659396A1 (en) | 1993-12-22 | 1995-06-28 | L'oreal | Process for oxidative hair dying using steam |
| EP0659393A1 (en) | 1993-12-22 | 1995-06-28 | L'oreal | Process for non-permanent hair deformation |
| US5520200A (en) * | 1993-12-22 | 1996-05-28 | L'oreal | Process for the non-lasting reshaping of keratinous fibres |
| US5957140A (en) | 1998-11-19 | 1999-09-28 | Mcgee; Robert J. | Hair styling iron for straightening and curling |
| WO2003039291A2 (en) * | 2001-11-02 | 2003-05-15 | Kenra, Llc | Hair relaxer |
| US20040000319A1 (en) * | 2002-06-26 | 2004-01-01 | The Procter & Gamble Company | Device for achieving smooth and straight hair |
| WO2004002262A1 (en) | 2002-06-26 | 2004-01-08 | Matsushita Electric Works, Ltd. | Hair iron |
| US7044139B2 (en) | 2003-08-01 | 2006-05-16 | Hometek International Ltd. | Hair curling iron and shaping tool |
| FR2921804A1 (en) * | 2007-10-03 | 2009-04-10 | Seb Sa | Portable hair treating apparatus for e.g. bleaching application, has steam generator supplied with liquid i.e. water, by electrical pump of liquid supply units to produce steam flow, which has specific range of value in gram per minute |
Also Published As
| Publication number | Publication date |
|---|---|
| MX2010003623A (en) | 2010-10-13 |
| US9603429B2 (en) | 2017-03-28 |
| JP2018023795A (en) | 2018-02-15 |
| BRPI1001018A2 (en) | 2011-04-26 |
| US20100258141A1 (en) | 2010-10-14 |
| CN101856172A (en) | 2010-10-13 |
| KR101235394B1 (en) | 2013-02-21 |
| BRPI1001018B1 (en) | 2020-09-15 |
| ES2576735T3 (en) | 2016-07-11 |
| KR20100110748A (en) | 2010-10-13 |
| JP2020203128A (en) | 2020-12-24 |
| CN105831947A (en) | 2016-08-10 |
| JP7020826B2 (en) | 2022-02-16 |
| US20170143091A1 (en) | 2017-05-25 |
| FR2943895A1 (en) | 2010-10-08 |
| MX338820B (en) | 2016-05-03 |
| JP2016025937A (en) | 2016-02-12 |
| JP2010241812A (en) | 2010-10-28 |
| EP2236053B2 (en) | 2021-03-03 |
| FR2943895B1 (en) | 2011-05-06 |
| EP2236053B1 (en) | 2016-03-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1935275B1 (en) | Method for permanently deforming keratinous fibres including a step of applying a reducing composition with a low concentration and an intermediate drying step | |
| EP1029533B1 (en) | Cosmetic compositions containing a hydroxyalkylether anionic surfactant and a cationic polymer, and their uses | |
| EP2916806B1 (en) | Method for straightening the hair using a composition containing glyoxylic acid and/or a derivative thereof | |
| EP1093808B1 (en) | Cosmetic compositions containing a vinyldimethicone/dimethicone copolymer and a cationic polymer, and their uses | |
| EP1291001B2 (en) | Cosmetic compositions containing a copolymer of methacrylic acid and an oil and uses thereof | |
| EP2236053B1 (en) | Method for the steam treatment of hair | |
| FR2910276A1 (en) | Use of cationic polymers in hair styling compositions containing alkoxysilanes with solubilizing functional groups, to avoid degradation in hair feel | |
| CA2400983A1 (en) | Cosmetic compositions containing a methacrylic acid copolymer, a silicone and a cationic polymer, and use thereof | |
| FR2968940A1 (en) | COSMETIC COMPOSITION COMPRISING A PARTICULAR ZINC SALT AND AN AMINE SILICONE | |
| FR2968944A1 (en) | COMPOSITION COMPRISING A NON-NITROGEN ZINC SALT AND A PARTICULAR CATIONIC SURFACTANT | |
| FR2964319A1 (en) | COSMETIC COMPOSITION COMPRISING AT LEAST ONE CATIONIC POLYMER AND AT LEAST TWO SURFACTANTS | |
| FR3060326A1 (en) | COMPOSITION COMPRISING AT LEAST TWO ANIONIC SURFACTANTS, A NON-IONIC SURFACTANT AND AN AMPHOTERIC SURFACTANT, AND AT LEAST ONE ORGANOSILANE | |
| FR3060381A1 (en) | COSMETIC COMPOSITION COMPRISING AN ORGANOSILANE, AT LEAST ONE CATIONIC POLYMER AND A PARTICULAR ASSOCIATION OF SURFACTANTS | |
| EP1291002A2 (en) | Cosmetic compositions containing a copolymer of methacrylic acid, a silicone and a cationic polymer and uses thereof | |
| EP1353634B1 (en) | Cosmetic compositions containing a fructan and a cationic polymer and their uses | |
| FR2802086A1 (en) | COSMETIC COMPOSITIONS CONTAINING QUATERNIZED SILICONE AND CATIONIC POLYMER AND USES THEREOF | |
| EP1047403B1 (en) | Cosmetic compositions containing an anionic alkylpolyglycoside ester surfactant and a cationic polymer and their uses | |
| FR2802085A1 (en) | COSMETIC COMPOSITIONS CONTAINING QUATERNIZED SILICONE AND NON-AMINOUS SILICONE AND USES THEREOF | |
| FR2984164A1 (en) | Cosmetic composition, useful for washing/cleaning body or conditioning hair, comprises hydrophobically modified celluloses having hydrophobic groups, and anionic surfactants having sulfur trioxide group containing compound | |
| EP1486196A1 (en) | Reducing composition for permanently deforming keratin fibers and process for the permanent deformation | |
| FR2848830A1 (en) | COSMETIC COMPOSITIONS CONTAINING AN AMPHOTERIC SURFACTANT AND A NACRANT AGENT AND USES THEREOF | |
| FR3087340A1 (en) | COSMETIC COMPOSITION COMPRISING CARBOXYLIC ACID, POLYSACCHARIDE, AND SURFACTANTS, PROCESS AND USE THEREOF | |
| FR2968943A1 (en) | COSMETIC COMPOSITION COMPRISING A ZINC SALT, A CATIONIC POLYMER AND A PROPELLING AGENT | |
| FR2802087A1 (en) | COSMETIC COMPOSITIONS CONTAINING QUATERNIZED SILICONE AND A NACRANT AGENT AND USES THEREOF | |
| EP1634569B1 (en) | Cosmetic composition based on a cationic surfactant, a fatty alcohol, a cationic non-silicone polymer and a diol |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20100401 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA ME RS |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20150827 |
|
| INTG | Intention to grant announced |
Effective date: 20151126 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 784332 Country of ref document: AT Kind code of ref document: T Effective date: 20160415 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: FRENCH |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 7 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602010031574 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: FP |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2576735 Country of ref document: ES Kind code of ref document: T3 Effective date: 20160711 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160630 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160701 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 784332 Country of ref document: AT Kind code of ref document: T Effective date: 20160330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160730 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160801 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R026 Ref document number: 602010031574 Country of ref document: DE |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160430 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160430 |
|
| 26 | Opposition filed |
Opponent name: KAO GERMANY GMBH Effective date: 20161228 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160401 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20100401 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160401 Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 |
|
| APBM | Appeal reference recorded |
Free format text: ORIGINAL CODE: EPIDOSNREFNO |
|
| APBP | Date of receipt of notice of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA2O |
|
| APAH | Appeal reference modified |
Free format text: ORIGINAL CODE: EPIDOSCREFNO |
|
| APBQ | Date of receipt of statement of grounds of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA3O |
|
| APAH | Appeal reference modified |
Free format text: ORIGINAL CODE: EPIDOSCREFNO |
|
| APBU | Appeal procedure closed |
Free format text: ORIGINAL CODE: EPIDOSNNOA9O |
|
| PUAH | Patent maintained in amended form |
Free format text: ORIGINAL CODE: 0009272 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT MAINTAINED AS AMENDED |
|
| 27A | Patent maintained in amended form |
Effective date: 20210303 |
|
| AK | Designated contracting states |
Kind code of ref document: B2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R102 Ref document number: 602010031574 Country of ref document: DE |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20210312 Year of fee payment: 12 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20160330 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20210506 Year of fee payment: 12 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210303 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20220310 Year of fee payment: 13 |
|
| P01 | Opt-out of the competence of the unified patent court (upc) registered |
Effective date: 20230527 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230401 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20240229 Year of fee payment: 15 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20240308 Year of fee payment: 15 Ref country code: BE Payment date: 20240319 Year of fee payment: 15 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20240306 Year of fee payment: 15 |