EP1275342B1 - Vorrichtung zur Herzzeitvolumenbestimmung eines Lebewesens - Google Patents
Vorrichtung zur Herzzeitvolumenbestimmung eines Lebewesens Download PDFInfo
- Publication number
- EP1275342B1 EP1275342B1 EP02254832.5A EP02254832A EP1275342B1 EP 1275342 B1 EP1275342 B1 EP 1275342B1 EP 02254832 A EP02254832 A EP 02254832A EP 1275342 B1 EP1275342 B1 EP 1275342B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- signals
- terminals
- impedance
- ecg
- data
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/02—Detecting, measuring or recording for evaluating the cardiovascular system, e.g. pulse, heart rate, blood pressure or blood flow
- A61B5/02028—Determining haemodynamic parameters not otherwise provided for, e.g. cardiac contractility or left ventricular ejection fraction
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/02—Detecting, measuring or recording for evaluating the cardiovascular system, e.g. pulse, heart rate, blood pressure or blood flow
- A61B5/026—Measuring blood flow
- A61B5/029—Measuring blood output from the heart, e.g. minute volume
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/02—Detecting, measuring or recording for evaluating the cardiovascular system, e.g. pulse, heart rate, blood pressure or blood flow
- A61B5/026—Measuring blood flow
- A61B5/0295—Measuring blood flow using plethysmography, i.e. measuring the variations in the volume of a body part as modified by the circulation of blood therethrough, e.g. impedance plethysmography
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/24—Detecting, measuring or recording bioelectric or biomagnetic signals of the body or parts thereof
- A61B5/25—Bioelectric electrodes therefor
- A61B5/271—Arrangements of electrodes with cords, cables or leads, e.g. single leads or patient cord assemblies
- A61B5/273—Connection of cords, cables or leads to electrodes
- A61B5/274—Connection of cords, cables or leads to electrodes using snap or button fasteners
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/24—Detecting, measuring or recording bioelectric or biomagnetic signals of the body or parts thereof
- A61B5/25—Bioelectric electrodes therefor
- A61B5/279—Bioelectric electrodes therefor specially adapted for particular uses
- A61B5/28—Bioelectric electrodes therefor specially adapted for particular uses for electrocardiography [ECG]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/68—Arrangements of detecting, measuring or recording means, e.g. sensors, in relation to patient
- A61B5/6801—Arrangements of detecting, measuring or recording means, e.g. sensors, in relation to patient specially adapted to be attached to or worn on the body surface
- A61B5/6843—Monitoring or controlling sensor contact pressure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/24—Detecting, measuring or recording bioelectric or biomagnetic signals of the body or parts thereof
- A61B5/25—Bioelectric electrodes therefor
- A61B5/279—Bioelectric electrodes therefor specially adapted for particular uses
- A61B5/28—Bioelectric electrodes therefor specially adapted for particular uses for electrocardiography [ECG]
- A61B5/282—Holders for multiple electrodes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/72—Signal processing specially adapted for physiological signals or for diagnostic purposes
- A61B5/7225—Details of analogue processing, e.g. isolation amplifier, gain or sensitivity adjustment, filtering, baseline or drift compensation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/72—Signal processing specially adapted for physiological signals or for diagnostic purposes
- A61B5/7235—Details of waveform analysis
- A61B5/7253—Details of waveform analysis characterised by using transforms
- A61B5/726—Details of waveform analysis characterised by using transforms using Wavelet transforms
Definitions
- the present invention relates generally to the field of biomedical analysis, and particularly to an apparatus for non-invasively determining the cardiac output in a living subject using impedance cardiography.
- Noninvasive estimates of cardiac output can be obtained using impedance cardiography.
- impedance cardiography also known as thoracic bioimpedance or impedance plethysmography, is used to measure the stroke volume of the heart.
- cardiac output is obtained.
- CO Stroke volume ⁇ heart rate .
- the heart rate is obtained from an electrocardiogram.
- the basic method of correlating thoracic, or chest cavity, impedance, Z T ( t ), with stroke volume was developed by Kubicek, et al. at the University of Minnesota for use by NASA, and disclosed in U.S. Reissue Patent No.
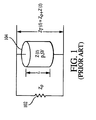
- the method generally comprises modeling the thoracic impedance Z T (t) as a constant impedance, Z o , and time-varying impedance, ⁇ Z(t) , as illustrated schematically in Fig. 1 .
- the time-varying impedance is measured by way of an impedance waveform derived from electrodes placed on various locations of the subject's thorax; changes in the impedance over time can then be related to the change in fluidic volume (i.e., stroke volume), and ultimately cardiac output via Eqn. (1) above.
- Electrodes utilizing a weak adhesive may also be displaced laterally to a different location on the skin through subject movement, tension on the electrical leads connected to the electrodes, or even incidental contact. This so-called “motion artifact” can also reflect itself as reduced accuracy of the cardiac output measurements obtained using the impedance cardiography device.
- a second disability associated with prior art impedance cardiography techniques relates to the detection of a degraded electrical connection or loss of electrical continuity between the terminals of the electrode and the electrical leads used to connect thereto.
- the electrolyte of the electrode may lose contact with the skin, and/or the electrical leads may become partially or completely disconnected from the terminals of the electrode.
- Electrodes As part of impedance cardiographic measurements is the downward or normal pressure applied to the subject in applying the electrode to the skin, and connecting the electrical leads to the electrode. It is desirable to minimize the amount of pressure needed to securely affix the electrode to the subject's skin (as well as engage the electrical lead to the electrode), especially in subjects whose skin has been compromised by way of surgery or other injury, since significant pressure can result in pain, and reopening of wounds.
- the individual stand-alone monitors are also proprietary, such that there is limited if any interface between them for sharing data.
- a common parametric measurement e.g., ECG waveform or blood pressure
- one monitor frequently cannot transmit this data to the other monitor due to the lack of interface, thereby necessitating repeating the measurement.
- ICG impedance cardiography
- Typical patient monitors include modules for several physiologic measurements such as ECG, blood pressure, temperature, and arterial pulse oximetry.
- ECG electrocardial pressure
- ICG blood pressure
- prior art ICG devices do not provide the facility for direct transmission of the data obtained from the subject, or other parameters generated by the ICG device after processing the input data, to a remote location for analysis or storage. Rather, the prior art approaches are localized to the bedside or monitoring location. This is a distinct disability with respect to the aforementioned outpatient applications, since the subject being monitored must either manually relay the information to the caregiver (such as by telephone, mail, or visit), or perform the analysis or interpretation themselves. Additionally, it is often desirable to perform more sophisticated (e.g. algorithmic) comparative or trend analysis of the subject's data, either with respect to prior data for that same subject, or data for other subjects.
- more sophisticated e.g. algorithmic
- an improved apparatus for measuring cardiac output in a living subject.
- Such improved apparatus ideally would allow the clinician to repeatedly and consistently place the electrodes at the optimal locations. Additionally, such an improved apparatus would also permit the detection of degraded electrical continuity between the electrode terminal and skin, or the electrode terminal and electrical leads of the measurement system, and be adapted to minimize the normal pressure on the subject's tissue when applying the electrodes and electrical leads.
- the apparatus would further be adapted to interface with other monitoring/display systems and parametric measurement modules that may be coincidently in use, and have connectivity beyond the immediate locale of the apparatus to permit the ready transfer of data to and from one or more remote locations or devices. Facility for selecting the highest quality input from a number of different sources would also ideally be provided.
- the present invention satisfies the aforementioned needs by providing an improved apparatus for measuring the cardiac output of a living subject.
- the invention is described herein in terms of an apparatus for determining cardiac output suitable for use on the thorax of a human subject, the invention may also conceivably be embodied or adapted to monitor cardiac output at other locations on the human body, as well as monitoring cardiac output on other warm-blooded species.
- digital processor is meant generally to include all types of digital processing devices including, without limitation, digital signal processors (DSPs), reduced instruction set computers (RISC), general-purpose (CISC) processors, microprocessors, and application-specific integrated circuits (ASICs).
- DSPs digital signal processors
- RISC reduced instruction set computers
- CISC general-purpose processors
- microprocessors microprocessors
- ASICs application-specific integrated circuits
- monitoring device and “monitoring device” are used generally to refer to devices adapted to perform monitoring, display, user interface, and/or control functions. Such devices may be dedicated to a particular function, or multi-purpose devices adaptable to performing a variety of functions and/or interfacing with a number of functional modules.
- FIG. 3a-5 the general methodology of measuring cardiac output in a living subject is described.
- the thoracic impedance Z T ( t ) of a living subject may be modeled as comprising a constant impedance, Z o , and time-varying impedance, ⁇ Z(t) .
- this change in thoracic impedance, ⁇ Z (t) is related to the pulsatile blood volume change.
- effectively constant tissue impedances such as bone, muscle, and fat are modeled as a conducting volume Z o 102 in parallel with the pulsatile impedance of the blood ⁇ Z (t) 104.
- This second impedance 104 is a time-varying fluid column with resistivity, p, cylindrical length, L , and a time-varying cross-sectional area that oscillates between zero and a value A, the latter which correlates to the stroke volume V.
- Z o the conducting tissues and fluids
- the ventricular ejection time is estimated from features in the impedance waveform, which is obtained from the measurement terminals of the electrode arrays 302, 304, 306, 308 placed on various locations of the subject's thorax as illustrated in Figs. 3a and 3b .
- a value of 150 ohm-cm is used for the resistivity of the blood, although it will be recognized that other values may be substituted as appropriate.
- the typical impedance associated with a human subject's skin is 2 to 10 times the value of the underlying thoracic impedance Z T ( t ).
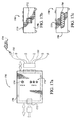
- the present invention may use at least two, and typically four electrode arrays 302, 304, 306, 308 for measurement, as shown in Fig. 3a . The physical construction and these electrode arrays is described in detail with reference to Figs. 7a-8 herein.
- one electrode array 302 comprising a stimulation electrode terminal 310 and a measurement electrode terminal 312 is applied above the thorax 300 of the subject, while a second electrode array 304 (having stimulation electrode terminal 314 and measurement electrode terminal 316) is applied below the thorax 300.
- the AC current from the current source is supplied to the stimulation electrode terminals 310, 314.
- current flows from each stimulation electrode terminal 310, 314 through each constant skin impedance, Z sk1 or Z sk4 , each constant body tissue impedance, Z b1 or Z b1 , and each constant skin impedance, Z sk2 or Z sk3 , to each measurement electrode terminal 312, 316.
- the voltages at the measurement electrode terminals 312, 316 are measured and input to a differential amplifier to obtain the differential voltage, V T (t).
- the desired thoracic impedance, Z T (t) is then obtained using the relationship of Eqn. (4).
- two sets of electrode arrays may advantageously be used to monitor the impedance associated with the left and right portion of the thorax 300 in the present invention.
- the four measurement arrays are also used to obtain an electrocardiogram (ECG), based on one of four vectors modified from Lead I, II, III, or IV.
- ECG electrocardiogram
- the resulting electrocardiograms are based on the original Lead configurations, but are not of diagnostic quality.
- the Q wave of the ECG QRS interval is used to determine the heart rate and to trigger measurements of VET within the dZ t dt waveform.
- Fig. 5 illustrates the logical flow of the method of measuring cardiac output which may be performed by apparatus according to the invention.
- the method 500 generally comprises first providing a plurality of electrode "arrays" of the type previously described herein per step 502.
- the electrode arrays are positioned at predetermined locations above and below the thoracic cavity per step 504, as illustrated in Fig. 3a herein. In one embodiment of the method, these locations are chosen to be on the right and left sides of the abdomen of the subject, and the right and left sides of the neck. These locations, with prior art band electrodes, were first used by Kubicek. Other locations and/or combinations of arrays may be substituted with equal success.
- a substantially constant AC current is generated in step 506, and the current applied to the stimulation electrode terminal 310, 314 of each of the electrode arrays in step 508.
- the voltage generated at the measurement electrode terminal 312, 316 of each electrode array is next measured in step 510. As previously discussed, this voltage is generally reduced from that applied to the stimulation electrode by virtue of the impedance of, inter alia, the thoracic cavity. Note that the measured voltage may be absolute, or relative (i.e., a differential voltage) as desired.
- step 512 the cardiac stroke volume from the measured voltage, using for example the relationship of Eqn. (3) above. Cardiac rate (step 514) is also determined by using the measurement electrodes to sense the ECG potentials generated by the heart of the subject.
- cardiac output is determined based on the stroke volume determined in step 512 and the cardiac rate in step 514 using the relationship of Eqn. 1 above.
- the system 600 generally comprises an alternating current (AC) current source 604 capable of generating a substantially constant current, a plurality of electrical leads in the form of a multi-ended lead assembly 606 for connecting the instrument monitor 607 to the individual terminals of the electrode arrays 302, 304, 306, 308, a processor 608 with associated algorithms capable of running thereon for performing analysis of the signals measured from the measurement terminals, data and program memory 609, 610 in data communication with the processor 608 for storing and retrieving program instructions and data; an I/O interface 611 (including analog-to-digital converter) for interfacing data between the measurement electrodes and the processor 608; a mass storage device 612 in data communication with the processor for storing and retrieving data; a display device 614 (with associated display driver, not shown) for providing an output display to the system operator,
- AC alternating current
- processor 608, memory 609, 610, I/O interface 611, mass storage device 612, display device 614, and input device 616 may be embodied in any variety of forms, such as a personal computer (PC), hand-held computer, or other computing device.
- PC personal computer
- I/O interface 611 I/O interface 611
- mass storage device 612 mass storage device 612
- display device 614 input device 616
- input device 616 may be embodied in any variety of forms, such as a personal computer (PC), hand-held computer, or other computing device.
- the applied current derived from the current source 604 is a 70 kHz sine wave of approximately 2.5 mA peak-to-peak.
- the measured voltage associated with the aforementioned sine wave is on the order of 75 mV peak-to-peak.
- the electrode lead assembly 606 of the illustrated embodiment contains a ten wire assembly (two wires are left unused) that branches to eight individual connectors 606a-h.
- the conductors 613a-j of the lead assembly are fashioned from electrically conductive material such as copper or aluminum, and are insulated using a polymer-based insulation having the desired dielectric strength as is well known in the electrical arts.
- the length of the conductors may also be controlled so as to match the impedance of each individual conductor to that of the others within the assembly 606.
- the body surface potential is measured between two measurement electrodes.
- This time-varying voltage reflects the electrical activity of the heart, and contains one QRS interval per cardiac cycle.
- the biopotential is analyzed to identify each QRS complex.
- the frequency of QRS complexes determines the heart rate.
- the Q wave within the QRS complex is then used to trigger identification of VET within the dZ t dt waveform, as the opening of the aortic valve (the beginning of VET) occurs after the appearance of the Q wave.
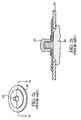
- each array comprises a flexible substrate 704 having a plurality of apertures 706, 708 formed therein.
- two terminals 310, 312 are disposed through the apertures such that the top portions 716, 718 of the terminals project above the plane of the substrate 704.
- the two terminals 310, 312 comprise a stimulation terminal 310 and measurement terminal 312 as previously described with respect to Fig. 3a .
- the stimulation terminal 310 is used to apply the potential necessary to generate the current flowing through the thoracic cavity of the subject.
- the asymmetric shape of the substrate 704 of the embodiment of Figs. 7a-7c may be used to assist the clinician in rapidly determining which electrode is the stimulation electrode and which the measurement electrode, such as by assigning a convention that the end of the array having a given shape always contains the stimulation electrode.
- the substrate may be shaped to adapt to certain physical features of the patient, such as by using a substrate having a broader width so as to better conform to the generally cylindrical shape of the subject's neck. Any number of different substrate shapes may be employed; Fig. 7d illustrates one such alternative shape.
- the terminals 310, 312 are firmly held in place within the substrate 704 at a predetermined distance 705 by a mounting element 707 or any one of a variety of other constructions as will be described in greater detail below.
- the distance (measured centerline-to-centerline on the terminals 310, 312) is approximately 5 cm in the embodiment of Fig. 7a , although it will be recognized that other distances may be substituted. Desired distances may be determined through experimentation, anecdotal observations, calculations, or any other suitable method; however, experimental evidence obtained by the Applicant herein indicates that a distance of 5 cm is optimal for impedance cardiography measurements.
- the substrate 704 in the embodiment of Fig. 7a is formed from a Polyethylene foam, although other materials such as cloth or vinyl may be substituted.
- the polyethylene foam is chosen for its compliance and flexibility, thereby allowing it to conform somewhat to the contours of the subject's anatomy, while still maintaining sufficient rigidity for maintaining the terminals 312, 314 in the desired position and orientation.
- the terminals 310, 312 of each array comprise a generally cylindrical shaped sidewall portion 720 having a first diameter 722, and a top portion 724 having a second diameter 726, the second diameter 726 being greater than the first diameter 722 in order to assist in retaining a connector mated to the terminal 310, 312 as described in greater detail below.
- the outer wall 721 of the sidewall portion 720 is essentially vertical in orientation (i.e., parallel to the central axis 725 of the terminal 310, 312), while the top portion is progressively rounded as shown.
- the terminals may be manufactured from an extruded metal such nickel, with a coating of brass, or may be molded from carbon.
- the terminals may be molded of plastic, and coated with a metal such as gold or impregnated with carbon.
- a metal such as gold or impregnated with carbon.
- the extruded metal possesses the advantage of low cost, while the molded plastic impregnated with carbon possesses the advantage of radiolucency.
- a terminal molded of plastic and coated with gold may possess low noise artifact.
- the terminals 310, 312 of the electrode array comprise a two piece construction, having an upper terminal element 730 and a lower terminal element 732 as shown in Figs. 7a and 7b .
- the post 734 of the lower terminal element 732 is adapted to be frictionally received within the cavity 736 of the upper terminal element when the two components are mated.
- the upper and lower elements 730, 732 form a single unit when assembled, with the mounting element 707 being frictionally held or "pinched" between the lower surface 740 of the upper element 730 and the upper surface 742 of the lower element 732.
- the post 734 of the lower element perforates the mounting element 707, or alternatively penetrates through a pre-existing aperture 738 formed therein.
- the lower elements 730, 732 of the electrode array terminals 310, 312 are coated with Ag/AgCl, although other materials with the desirable mechanical and electrochemical properties such as Zinc Chloride may be used if desired.
- the electrolytic element 750 of each electrode array comprises an electrolytic gel of the type well known in the bio-electrical arts; in the present embodiment, the gel comprises an ultraviolet (UV) cured potassium chloride (KCl) gel, although it will be recognized that other types of compounds or materials may be used. UV curing of the gel allows the element 750 to have a more solidified consistency and improved mechanical properties, thereby preventing excessive spreading or thinning of the element when the array is applied to the subject while still maintaining its overall adhesiveness and electrolytic properties. As shown in Figs. 7b and 7c , the element 750 is sized so as to encompass the edges 752 of the respective aperture 706, 708 in the substrate 704 over which it is placed when assembled, although other configurations may be used.
- UV ultraviolet
- KCl potassium chloride
- the top portion 755 of the element 750 fits at least partially within the aperture 706, 708 and conforms substantially thereto, thereby effecting contact with the bottom surface 760 of the bottom terminal element 732. In this way, ions are passed between the skin of the subject and the terminals of the array via the gel element 750.
- the gel also provides for adhesion of the array to the skin of the subject, although the array of the present embodiment also includes a separate adhesive 762 which is applied to the bottom surface of the substrate 704, as shown in Fig. 7c .
- the gel of the element 750 is advantageously placed in the embodiment of Figs. 7a-c so as to be symmetric with respect to the terminal 310, 312. It will be recognized, however, that the element(s) 750 may also be placed so as to produce certain desired electrolytic conditions. Similarly, the element 750 may be split into two or more component parts if desired.
- Figs. 7a-c employs two fixed terminals that are effectively immovable within the substrate
- means for allowing adjustment or change of the relative position of the terminals may be substituted.
- a terminal array having three terminal posts may be used, the second post 802 being spaced a first distance 804 from the first post 806, and the third post 810 being spaced a second distance 808 from the first post 806, such that the clinician can select one of two terminal spacings as desired.
- each electrode lead assembly connector 606a-h is designed to mitigate the downward force required to mate the connector with its respective electrode array terminal.
- each connector 606a-h contains two spring-biased conductive jaws 902 that are spread apart by the cam surface 904 of an actuator button 906 disposed on the front 907 of the connector body 908.
- the connector jaws 902 and bias mechanism are designed to allow the upper and sidewall portions 724, 720 of the electrode terminal 310, 312 ( Fig. 7b ) to be received within the recess 910 of the jaws 902 when the button 906 is fully depressed. In this fashion, effectively no downward force is required to engage the connector to its respective terminal.
- the jaws 902 are contoured to engage substantially the entire surface of the sidewall portion 720 of the terminal when the actuator button 906 is released. Since the sidewall portion 720 of the terminal is effectively circular in cross-section, the connector may advantageously rotate around the axis of the terminal 310, 312 when lateral tension is applied to the conductor attached to that connector.
- U.S. Patent No. 5,895,298 issued April 20, 1999 entitled “DC Biopotential Electrode Connector and Connector Condition Sensor,” and incorporated herein by reference in its entirety, describes a bias jaw electrical connector of the type referenced above in greater detail.
- each connector 606a-h is fastened to one of the two terminals 310, 312 of an electrode array.
- the 68 kHz constant current is applied from the current source to four electrode terminals (i.e., one terminal per array).
- complete circuits are formed between the current source and the I/O device 611 of the system 600 via the electrical conductors and connectors associated with the stimulation electrode terminals, the stimulation electrode terminals themselves, the thorax of the subject, the measurement terminals, and the electrical conductors and connectors associated with the measurement terminals.
- Fig. 10 the method of evaluating the electrical continuity of one or more leads within the system is described. Note that while the following description is based on the two-terminal array configuration ( Figs 7a-7c ) and the use of four arrays as shown in Fig. 3a , the method may be applied to many alternate configurations with equal success.
- the electrode arrays are disposed on the skin of the subject.
- the position at which the electrode arrays are disposed on the subject are measured in relation to the thoracic cavity as illustrated in Fig. 3a , or alternatively may be inferred by the weight and height of the subject.
- a current is generated between the stimulation electrodes and the measurement electrodes of the respective arrays in step 1004. As previously discussed, the current passes through at least a portion of the subject's thoracic cavity, encountering a time-variant impedance therein.
- An impedance waveform is then measured from two or more of the measurement terminals of the arrays in step 1006.
- the waveforms comprise measurements of impedance as a function of time, which is well known in the cardiographic arts. These measured waveforms are then compared to one another in step 1008 to detect changes or variations between them.
- two waveforms are differenced by way of a simple differencing algorithm resident on the processor 608 of the system 600 ( Fig. 6 ), although it will be recognized that other approaches may be used.
- the base impedance may be calculated for the left and right sides. The larger base impedance may then be subtracted from the smaller base impedance, with this difference then divided by the smaller impedance.
- the resulting percentage ratio when greater than a predetermined threshold value, may represent the presence of detached or loose electrodes. While some variation between the waveforms is normal, significant variations are indicative of either a degraded electrical connection, such as between the electrode array terminal and its respective connector, or between the electrolytic gel and the skin of the patient, or even the gel and the terminal of the array or between the cable and connector.
- a threshold value is determined and set by the operator of the system in step 1010 such that when the threshold "difference" is exceeded as determined by the aforementioned algorithm (step 1012), the operator will be alerted to the degraded condition such as by a visual or audible alarm in step 1014.
- the difference in impedance (or voltage) between the individual terminals 310, 312 of one or more electrode arrays is measured and used as the basis for the continuity determination. Specifically, the difference in the values measured from one terminal 310 with respect to another terminal 312 of the exemplary two-terminal array 302 illustrated herein is measured; when this value exceeds a certain threshold difference value (e.g., 650 Ohms, although other values may be substituted based on any number of factors), a loose electrode or otherwise degraded connection is suspected. It will be recognized that this methodology may also be employed when more that two electrical terminals are electrically connected to the system.
- a certain threshold difference value e.g., 650 Ohms, although other values may be substituted based on any number of factors
- the algorithm would be adapted to measure the difference between each of the non-repeating permutations (i.e., 1-2, 1-3, and 2-3) and compare such differences to the threshold.
- the aforementioned threshold value may be algorithmically determined and/or parametrically variant. For example, based on data obtained by the system before and/or during operation, the apparatus of the present invention may periodically or continuously calculate new threshold values as a function of time. Alternatively, the system may be adapted to calculate a plurality of such impedance difference values across each of the terminal arrays in use, and average the values periodically to maintain a "moving average" of impedance differences. As yet another alternative, other physiological parameters of the subject being monitored could be used as "triggers" for revised threshold determination and/or impedance difference calculation.
- the use of the multi- terminal electrode arrays having predetermined and substantially equal terminal spacing as previously described allows such comparisons between electrode waveforms to be made; errors resulting from uncontrolled spacing of the terminals are effectively eliminated.
- the aforementioned method would be largely ineffective, since these error sources would force the threshold value to be set artificially high, thereby potentially masking conditions of degraded electrical continuity which could affect the ultimate accuracy of and cardiac output estimation made by the system.
- the ICG module utilizes the electrical bioimpedance measurements previously described herein to continuously generate a signal indicative of pulsatile thoracic impedance changes.
- This pulsatile thoracic impedance signal is processed to produce signals indicative of other related parameters, such as the ventricular ejection time (VET) and the maximum rate of change of pulsatile thoracic impedance, which are used to calculate the volume of blood pumped per stroke according to equations previously discussed.
- VET ventricular ejection time
- VET maximum rate of change of pulsatile thoracic impedance
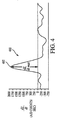
- the voltage developed across the thoracic impedance at any instant in time can vary due to a number of different factors. Specifically, the voltage is affected by four primary components: (i) base impedance; (ii) respiration; (iii) cardiac changes; and (iv) patient motion.
- the base impedance comprises the largest component of the modulated waveform, and represents the nominal conductivity of the thorax. It is a function of the tissue and fluid distribution within the interrogated area.
- the average value of the base impedance, or TFI is roughly 30 Ohms, but can vary from 5 to 60 Ohms in adult humans.
- the ventilation cycle is relatively long, typically 0.2 to 0.7 Hz with variable magnitude.
- Impedance changes occur due to the cardiac cycle, such as after ventricular depolarization (QRS complex), and have very small magnitudes (approximately 0.05 - 0.3 Ohms). These impedance changes are the result of aortic expansion after blood is ejected from the ventricle, and contraction as blood is flows into the circulatory system.
- QRS complex ventricular depolarization
- Movement by the patient causes significant impedance changes due to fluid shifts and density/volume changes in the thorax, with varying frequency and magnitude.
- the motion component of impedance is effectively eliminated when the patient is monitored at rest.
- Fig. 11 is a graph illustrating the frequency ranges of these four signal components for a typical adult human subject.
- the ICG module advantageously addresses the foregoing components of thoracic impedance through its signal processing circuitry and algorithms, now described in detail.
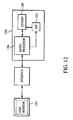
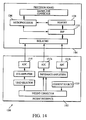
- the ICG module 1200 is electrically coupled to the host monitor 1202 via the interface device 1204.
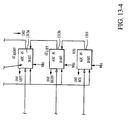
- the ICG module 1200 generally comprises a microprocessor 1206, storage device 1208, and digital signal processor 1210, as described in greater detail below with respect to Fig. 13 .
- the module 1200 communicates with a host monitor 1202 to continuously monitor and display the cardiac output and pulse rate of the subject under evaluation. Communications are in the illustrated embodiment conducted according to a predetermined protocol (such as a serial interface protocol of the type well known in the art), although other approaches may be substituted with equal success.
- the module further includes other features such as input power conditioning and "soft-start" current limiting functionality, software/firmware download from the host device, and electrical isolation (e.g., 4000 V) for the subject being monitored.
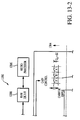
- the exemplary embodiment of the module 1200 comprises two component boards 1302, 1304, identified herein as the "patient board” 1302 and the “processor board” 1304. It will be recognized that these may or may not be separate physical boards or substrates.
- the patient board 1302 provides a number of functions, including (i) interface with the external signal sources; e.g., the patient leads and electrodes which provide, inter alia, the impedance and ECG waveform signals to the module; (ii) ECG vector select (described in greater detail below); and (iii) input signal filtration, conditioning, and domain conversion.
- the patient board 1302 also isolates the electrical (ECG) the mechanical ( ⁇ Z) components of cardiac activity from each other, and from the components of respiration and motion present in the signals derived from the subject under evaluation.
- a first high-pass filter (pole at 0.33 Hz) 1306 filters the input impedance waveform 1307 and ECG waveform 1308.
- Band-pass filters 1309a, 1309b comprising a low-pass filter with pole at 1.59 MHz and high-pass filter with pole at 9.72 kHz are used to further filter the respective high-pass filtered impedance waveforms of each input channel (a channel being defined for the purposes of this exemplary discussion as a pair of electrodes; i.e., "left" channel and "right” channel).
- Fixed gain amplifiers 1310a, 1310b receive the output of the band-pass filters 1309a, 1309b for each channel, and provide a fixed gain output signal to respective digital potentiometers 1311a, 1311b, the output of which is supplied to respective demodulators/filters 1312a, 1312b.
- the output of the demodulator/filter units 1312a, 1312b is passed through 5 M Ohm resistors and subsequently input to respective analog-to-digital converters (ADCs) 1313a, 1313b, which are clocked according to a 2 MHz clock signal (described below).
- ADCs analog-to-digital converters
- the patient board 1302 further comprises a plurality of ECG inputs 1314 which are obtained from the aforementioned electrode pairs and input to a crosspoint switch 1315 (e.g., a 16 x 16 analog multiplexer such as the AD75019 device manufactured by Analog Devices), which selects the best "quality” input from among the four inputs as described in detail below with respect to Figs. 20-22 .
- the selected ECG signal is low-pass filtered 1316, amplified to a fixed gain 1317, high-pass filtered 1318, and then supplied (via 1 M Ohm resistor) to the ECG ADC 1319.
- the three ADCs 1313a, 1313b, 1319 output digitized signals to the DSP 1210 on the processor board 1304, described below.
- the patient board 1302 also uses this crosspoint switch to provide several other functions, including connection of leads to the patient, provision of multiple ECG vectors, loose electrode testing (previously described), cable identification, and calibration.
- the processor board 1304 comprises a digital signal processor (DSP) 1210 with direct memory access (DMA) of the type well known in the electronic arts, a microprocessor 1206, a storage device 1208 coupled with the DSP 1210 and microprocessor 1206 via a data bus, a first signal (constant current) source 1330 generating a nominal 70 kHz output signal, a second signal source 1332 generating a nominal 2 MHz output signal, a clock signal generator (12 MHz nominal), and digital-to-analog converter (DAC) 1334.
- DSP digital signal processor
- DMA direct memory access
- the module is configured with a DC/DC converter operating at 90 kHz, and the aforementioned ICG current source at 70 +/- 6 kHz.
- Fig. 13a graphically illustrates the impedance signal extraction process performed by the ICG module 1200.
- the microprocessor 1206 comprises a microcomputer running at a crystal frequency of approximately 32.7 KHz, although it will be recognized that other platforms may be substituted with equal success.
- the system clock is generated by an on-chip phase-locked loop (PLL) and is software programmed for an operating frequency of 16 MHz.
- PLL phase-locked loop
- the device is operated in the 8-bit bus operating mode with all data transfers occurring on data lines 8 through 15.
- the processor also has a QSPI built in, which in the present embodiment is used for communications to the host device, such as by using a serial interface protocol of the type previously referenced herein.
- the ICG module 1200 utilizes a three-part software architecture comprising three modules: (i) "Initialization” module; (ii) “Operating” module; and (iii) "Processing” module. Any one of the three software code modules can be independently downloaded.
- the Initialization operating system of the microprocessor 1206 comprises a variant of the "C Executive" system manufactured by JMI Software Consultants, Inc., although it will be recognized that other operating systems may be substituted.
- C Executive comprises a real-time, memory-resident, event driven monitor program designed for embedded systems which require multi-tasking functionality and ROM storage.
- the initialization module software uses the initialization OS for process scheduling, input and output, and inter-process communication.
- Fig. 13b graphically illustrates the high-level program flow of the Operating software module of the illustrated embodiment.
- the processing module executes the bioimpedance algorithms. It also controls peripheral functions, such as the gain of the impedance amplifiers, the setting the ECG vector, reading of the impedance and ECG A/D converters, and detection of electrical continuity.
- the DSP 1210 adapted for use with the invention comprises an Analog Devices ADSP-2181 device, although it will be recognized that any digital processing device adapted for algorithms such as those described herein may be used with proper adaptation. For example, members of the Texas Instruments 'C4x family of floating point DSPs, 'C5x family of fixed point processors, 'C6x family of VLIW processors, the Lucent DSP 16000 family, or even a user-customized processor core or ASIC may be used. Many other types of digital signal processors exist, any number of which may be adapted for use with the present invention.
- Parameters determined from the digitized data are communicated to the microprocessor 1206 via the storage device 1208, specifically by writing data words to predetermined locations within the storage device.
- a dual-port RAM (DPR) is selected to allow dual-port access and two-way communication by the DSP 1210 (via, e.g., the BDM port) and microprocessor 1206 via first and second memory ports, respectively; however, it will be recognized that other types and configurations of storage device including DRAM, SDRAM, SRAM, dual data rate synchronous DRAM (DDR-SDRAM), ROM, or even non-semiconductor storage devices may be substituted.
- DRAM dual-port RAM
- SRAM dual data rate synchronous DRAM
- ROM dual data rate synchronous DRAM
- non-semiconductor storage devices may be substituted.
- Fig. 13 further comprises a DMA unit of the type well known in the art, thereby facilitating direct memory accesses by the processor(s).
- the module 1200 is further configured such that address and data bus interfaces exist between the microprocessor 1206 and storage device 1208, and between the DSP 1210 and storage device 1208, thereby providing for memory addressing and data transfer by both processors.
- Fig. 13c illustrates one exemplary embodiment of the memory map used with the DPR 1208 of the ICG module.
- An address range is also specified at the microprocessor 1206 and connected to the DSP's DMA port 1350 for DSP code download from the microprocessor 1206 (and host 1202) to the DSP 1210 during operation.
- the impedance and ECG waveform data present within the module consists of all measured and calculated parameters related to a specific cardiac event, without any averaging.
- the start of data is placed at the Q point of the cardiac cycle measured.
- Table 2 illustrates the data write operations into memory performed by the DSP 1210 at a 200Hz rate: Table 2 Data Memory address Description ⁇ Z 7FA0 ECG 7FA2 Respiration 7FA4 dZ/dt 7FA6 pacer impulse 7FA8 Pace enhanced ECG 7FAA ECG + pacer spikes ECG gain factor 7FAC Loose Electrode 7FAE
- the module 1200 further utilizes a 512K x 8 static RAM (SRAM) array for temporary data storage.
- SRAM static RAM
- the static RAM is also used as temporary storage of ICG Monitor program code during program download of the software.
- the program code is stored in a 128K x 8 sectored "flash" EPROM. This device can be erased on an individual sector basis.
- the first sector of the flash memory is used for storing the initialization (boot-up) code. In general, this sector of code is not modified, thereby ensuring that even if a download of code fails, the module will still be able to attempt another download.
- the other seven sectors of the flash memory are used for storage of the Operating code.
- the Operating code is the code which is run during normal operation of the module. This code can be updated using the host monitor or other external storage device.
- the SPORT0 is a standard port of the DSP which is used to transmit setup control to the ADCs 1313a, 1313b, 1319, digital potentiometers 1311a, 1311b, and the crosspoint switch, and receive data from the ADCs.
- DSP port SPORT1 is used to transmit data to the DAC 1334.
- Fig. 13d graphically illustrates the SPORT0 and ADC data acquisition and timing relationships of the present embodiment in detail.
- Fig. 14 illustrates portions of the data and signal flow within and between the patient and processor boards (and associated components) of the ICG module of Figs. 12-13 .
- Appendix I hereto provides a listing of the various parameters utilized within or generated by the ICG module 1200.
- VEPT Volume of Electrically Participating Tissue
- BSA Body Surface Area
- Hemodynamic parameters are calculated from the values HR, PEP, VET, TFI, dZ t dt max and d 2 ⁇ Z t dt 2 max which are extracted from the aforementioned ECG, dZ t dt and d 2 ⁇ Z t dt 2 waveforms.
- Indexed parameters are obtained by dividing the parameter (e.g., BSA, CO, CI, SI) by the appropriate index.
- Appendix II details the communications protocol (including memory address) for the patient data communicated by the exemplary embodiment of the module to the host device.
- Two important parameters present in estimations of cardiac output are (i) the maximum negative change in the impedance signal ( Z ( t )) as a function of time, dZ t dt max ; and (ii) the ventricular ejection time (VET).
- These parameters, as well as other related parameters, are found from features referred to as "fiducial points" that are present in the inverted first derivative of the impedance waveform, dZ t dt .
- the maximum value of dZ t dt referred to as dZ t dt max
- C point the time at which the inverted derivative value has the highest amplitude
- VET also known as LVET, relating to the left ventricle of the heart in a human
- That point in time associated with aortic valve opening also commonly known as the "B point”
- B point is generally determined as the time associated with the onset of the rapid upstroke (a slight inflection) in dZ t dt before the occurrence of the C point.
- the time associated with aortic valve closing also known as the "X point” is generally determined as the time associated with the inverted derivative global minimum, which occurs after the C point.
- the O point represents the time of opening of the mitral valve of the heart.
- the O point is generally determined as the time associated with the first peak after the X point.
- the time difference between aortic valve closing and mitral valve opening is known as the iso-volumetric relaxation time, IVRT.
- Impedance cardiography further requires recording of the subject's electrocardiogram (ECG) in conjunction with the thoracic impedance waveform previously described.
- ECG electrocardiogram
- Processing of the impedance waveform for hemodynamic analysis requires the use of ECG fiducial points as landmarks.
- Processing of the impedance waveform is generally performed on a beat-by-beat basis, with the ECG being used for beat detection.
- detection of some fiducial points of the impedance signal may require the use of ECG fiducial points as landmarks.
- individual beats are identified by detecting the presence of QRS complexes within the ECG.
- the peak of the R wave (commonly referred to as the "R point") in the QRS complex is also detected, as well as the onset of depolarization of the QRS complex ("Q point").
- R point The peak of the R wave
- Q point the onset of depolarization of the QRS complex
- the ICG module for use in the present invention is further modified to incorporate fiducial point detection within the aforementioned impedance and/or ECG waveforms provided as inputs to the module.
- “event markers” are placed within the waveform buffers to indicate the algorithm detection points with reference to the waveform samples.
- Table 4 shows some of the marker values used for the various fiducial points: Table 4 Fiducial Point Marker Value Q point 0x10 B point 0x20 X point 0x30 dZ/dt max 0x50 d 2 Z/dt 2 max 0x60
- LVET ventricular ejection time
- fiducial point detection within the ICG module is conducted using the wavelet transform methodology as disclosed in co-pending U.S. patent application publication number US 20020138014 .
- the fiducial points of the ⁇ Z and dZ/dt waveforms (e.g., B, C, X, and O) are detected in this variant using discrete wavelet transforms, rather than by empirical detection, which is based on processing features in the first and second derivatives of ⁇ Z(t).
- the wavelet transform methodology advantageously requires only simple additions and multiplications of real numbers, thereby substantially simplifying the processing associated with the cardiac output (CO) determination performed by the DSP 1210 and associated algorithms.
- the wavelet transform methodology compared to the empirical methodology, is much less sensitive to noise artifact.
- fiducial points are utilized in evaluating the electrocardiogram (ECG) waveform of the subject, with specific individual "beats" of the subject's cardiac muscle being identified through detection of one or more fiducial points, either by the aforementioned wavelet transforms or by other means.
- ECG electrocardiogram
- the peak of the R wave (R point) in the QRS complex as well as the onset of depolarization of the QRS complex (Q point) are also detected.
- the time interval between the R waves is also used to calculate the subject's heart rate.
- Figs. 15a-c one embodiment of the ICG module for use in the present invention adapted for rack mounting is described.
- the module 1500 is fitted with a faceplate 1502 disposed generally at the front portion 1506 of the module housing 1504, as well as a plurality of electrical connectors 1510, 1512, one connector 1510 disposed generally at the front portion 1506 of the housing, and one connector 1512 at the rear portion 1515 of the housing 1504.
- a debug port connector (not shown) is also provided to facilitate debug of the microprocessor 1206.
- Optional module status indicators 1520 are also disposed on the faceplate 1502 so as to be viewable by a user or clinician during operation of the module when the module is received within an equipment "rack" (described in greater detail below).
- the module housing 1504 is shaped and sized so as to be received within the rack adjacent or generally in proximity to other modules, such that space is economized.
- the front panel connector 1510 comprises the ICG module interface with the patient being monitored, including electrical connection to the measurement and stimulation electrode terminals previously described herein.
- the front panel connector 1510 may be of any configuration, such as a multi-pin standardized male or female electrical connector of the type well known in the art, although literally any configuration (proprietary or otherwise) may be substituted.
- the rear panel connector 1512 allows for electrical connection of the module to the host monitor/interface unit, such as for example via a multi-pin female connector for mating with backplane connectors of the host monitoring equipment (including any voltage supply associated therewith). It will be recognized, however, that other types and "pin-outs" of connector or data/power interface may be substituted with equal success, dependent primarily on the host equipment with which the module must interface.
- the equipment module 1500 of Fig. 15 may also be configured with a network data interface (described in detail below with respect to Fig. 19 ), thereby allowing the distribution of data to a plurality of different local and/or remote nodes for analysis, storage, or other functions.
- a network data interface described in detail below with respect to Fig. 19
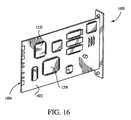
- Fig. 16 illustrates yet another embodiment of the ICG module for use in the invention, configured as a plug-in circuit card 1600 for use within a host device such as a dedicated stand-alone monitor or host monitoring device (e.g., one that has a primary function which may or may not be related to ICG or cardiography), personal computer, laptop computer, hand-held computer, minicomputer, or SUN UNIX workstation.
- the circuit card 1600 integrates all of the functionality of the embodiment of Fig. 1300, including processor and patient interface boards, onto one card substrate 1602.
- a standardized edge-type electrical connector 1604 is also provided to permit interface with the card receptacle of the host device (not shown), which may be configured according to any electrical interface standard (such PCMCIA, PC Card, or otherwise).
- the ICG module for use in the invention comprises a card generally similar to that shown in Fig. 16 , except that the edge-type connector 1604 is replaced with a ribbon-cable and associated connector (or other type of connector) of the type well known in the electrical arts for interfacing the module with other circuit elements and boards within the host device.
- the ICG module may be plugged directly into another module within the host device. It will be recognized that literally any type of electrical interconnection scheme and protocol between the ICG module (whether in "card” form as in Fig. 16 or otherwise) and the host device with which the module is used may be employed consistent with the invention.
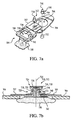
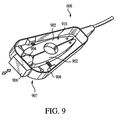
- Figs. 17a-c illustrate yet another embodiment of the ICG module, configured as a "yoke" 1700 adapted for mobility and electrical interface with a monitoring device.
- the term yoke is meant to include any configuration of mobile or transportable device which is used to facilitate centralization of a plurality of patient signals.
- the yoke 1700 is adapted to receive a plurality of electrical leads 1702 (whether as individual leads, in one variant, or as a single multi-terminal electrical connector 1708, in another variant) which are connected to the electrodes 1704 disposed on the thorax of the patient being monitored.
- the yoke 1700 is configured to be light weight and rugged, and utilizes a molded plastic impact-resistant housing 1706 of the type well known in the polymer arts, although other materials may be used.
- the yoke housing 1706 contains the electronics of the ICG module, including processor and patient interface boards (not shown), and further optionally includes an LED 1710 or other status indication for the ICG module.
- the output of the ICG module electronics in the yoke 1700 is transferred to the monitoring device (not shown) via a data interface 1712, in the present embodiment a universal serial bus (USB) connection and cable of the type well known in the electrical arts.
- USB universal serial bus
- a wireless interface between the yoke 1700 and host monitor may be used.
- an RF transceiver and modulator device are provided and adapted to generally comply with the well known "BluetoothTM" wireless interface standard.
- the Bluetooth "3G” wireless technology allows users to make wireless and instant connections between various communication devices, such as mobile devices (e.g., cellular telephones, PDAs, notebook computers, local or remote patient monitoring stations, and the like) and desktop computers or other fixed devices.
- the Bluetooth topology supports both point-to-point and point-to-multipoint connections. Multiple "slave" devices can be set to communicate with a 'master' device.
- the yoke 1700 when outfitted with a Bluetooth wireless suite, may communicate directly with other Bluetooth compliant mobile or fixed devices including a receiver disposed at the host monitor, or alternatively other Bluetooth-capable devices such as a cellular telephone, PDA, notebook computer, or desktop computer. Alternatively, WMTS telemetry may be utilized.
- the operation of the wireless interface is effectively transparent to the yoke 1700 and host monitor, although it will be recognized that data may be "buffered" within one or more intermediary storage devices (not shown) if desired.
- the signal processing and transceiver/modulator components of the interface may be embodied in a fully integrated "system on a chip” (SoC) application specific integrated circuit (ASIC) of the type generally known in the semiconductor fabrication arts (not shown).
- SoC ASIC incorporates, inter alia, a digital signal processor (DSP) core, embedded program and data random access memories, RF transceiver circuitry, modulator, analog-to-digital converter (ADC), and analog interface circuitry necessary to support sampling, conversion, processing, and transmission of the cardiac output (or other) data to the host monitor's receiver.
- DSP digital signal processor
- ADC analog-to-digital converter
- a number of different subjects undergoing cardiac monitoring/analysis using the yoke 1700 may be monitored in real time at a centralized location using a single monitor receiver.
- the monitor receiver (not shown) and transceiver are adapted to receive a plurality (currently seven, under prevailing Bluetooth architecture, although such number may be increased or decreased) of signals from remote ICG module devices, whereby the individual signals may be multiplexed or alternatively processed in parallel by the host monitor and interface (with the addition of appropriate multiplexing or parallel processing hardware of the type well known in the electronic arts).
- a host monitor configured to receive such multiplexed or parallel channel data may be used to monitor the cardiac output and other related parameters of multiple subjects at once.
- Bluetooth-compliant devices operate in the 2.4 GHz ISM band.
- the ISM band is dedicated to unlicensed users, including medical facilities, thereby advantageously allowing for unrestricted spectral access.
- Maximum radiated power levels from the yoke's transceiver are in the mW range, thereby having no deleterious effect on the physiology of the subject due to radiated electromagnetic energy.
- radiated power from the antenna assembly (not shown) of the yoke transceiver may also be controlled and adjusted based on relative proximity of the transceiver, thereby further reducing electromagnetic whole body dose to the subject.
- the modulator of the yoke uses one or more variants of frequency shift keying, such as Gaussian Frequency Shift Keying (GFSK) or Gaussian Minimum Shift keying (GMSK) of the type well known in the art to modulate data onto the carrier(s), although other types of modulation (such as phase modulation or amplitude modulation) may be used.
- GFSK Gaussian Frequency Shift Keying
- GMSK Gaussian Minimum Shift keying
- Spectral access of the device may be accomplished via frequency divided multiple access (FDMA), frequency hopping spread spectrum (FHSS), direct sequence spread spectrum (DSSS, including code division multiple access) using a pseudo-noise spreading code, or even time division multiple access, depending on the needs of the user.
- FDMA frequency divided multiple access
- FHSS frequency hopping spread spectrum
- DSSS direct sequence spread spectrum
- any wireless interface capable of accommodating the bandwidth requirements of the system may be used, such as the new WMTS biomedical band of 608 - 614 MHz.
- an infrared device e.g., Infrared Data Association "IrDA”
- IrDA Infrared Data Association
- Fig. 18 is a perspective view of the yoke of Figs. 17a-c , adapted for wireless communication with the monitoring device as just described.
- a network interface device 1902 e.g., LAN card
- the network interface card 1902 and connector 1904 allow the cardiac output (CO) and other data generated by the module to be transferred to one or more remote nodes, such as the various stations of a local area network or wide area network.
- the network interface device 1902 in the present embodiment comprises an IEEE 802 Ethernet card adapted for packetized data transfer, although it will be recognized that any number of different network hardware environments (including, e.g., X.25, Token Ring, SONET, FDDI, Gibagbit Ethernet, or ATM) and protocols (e.g., TCP/IP, RTP, or FTP) may be utilized.
- the ICG module may be outfitted with a modulator/demodulator apparatus of the type well known in the data communication arts, or DSL, ADSL, or DOCSIS device.
- any data network device including satellite uplink/downlink, can be used for transferring cardiac data to/from the ICG module consistent with the invention.
- the ICG module consistent with the present invention may include provision, via the aforementioned patient board 1302, for receiving a plurality of input signals (“vectors") that may be used in the cardiac output determination.
- These input vectors typically include ECG signals that are derived, for example, from the various ICG/ECG electrodes disposed on the subject's body.
- signal sources may comprise one or more other modules or devices. Regardless of source or type of signal, these input vectors may vary significantly in terms of signal quality and/or continuity. Therefore, the present invention may be advantageously adapted to automatically (and continuously, if desired) analyze and arbitrate between the various input vectors based on their relative attributes (e.g., signal quality). This feature possible with the invention is now described in detail with respect to Figs. 20-22 .
- the ECG vector of the ICG module is selected using a vector select multiplexer 1315.
- the vector is selectable from a plurality of electrode pairs located at various points on the thorax of the subject, as shown in Table 5 below: Table 5 Vector ECG Channel 1 EF 2 CF 3 DF 4 ED
- Fig. 20 illustrates the methodology of ECG vector selection.
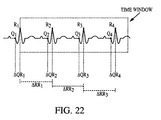
- each electrical lead providing a signal input is tested within a predetermined (e.g., six second) time window, beginning at the first detected R point (step 2002) in the input vector under analysis. If the total number of R points detected within the window is less than a given value n (e.g., 4) per step 2006, the lead is rejected per step 2008.
- n e.g., 4
- each lead is evaluated per step 2010 of the present method 2000 based upon three factors: (i) R-wave magnitude, (ii) QR interval difference, and (iii) RR interval difference.
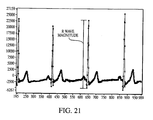
- R-wave magnitude (RM) is the peak-to-peak magnitude of the R point, which is calculated per step 2012 as shown in Fig. 20a by taking the value of the R peak (step 2014) and subtracting the value of the preceding local minimum (step 2016). If there is no local minimum found within a "back" sample or temporal window of a given size (e.g., 80 samples) per step 2018, the R point is rejected.
- Fig. 21 illustrates the calculation process graphically in terms of a typical QRS complex.
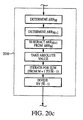
- the QR interval difference ( QR score ) and the RR interval difference ( RR score ) are determined per steps 2020 ( Fig. 20b ) and 2030 ( Fig. 20c ), respectively, as follows.
- Each lead factor is also optionally normalized, to between 0 and 1 in the illustrated embodiment.
- all of the lead magnitudes are divided by the maximum value among the leads.
- the QR score and RR score values the number are first inverted, and then normalized based upon the maximum value among the leads.
- the value are set to a small constant before inversion (or, alternatively, any other approach which accounts for the infinite value when inverting zero may be employed).
- the Assignee hereof has determined that optimal values to be used during testing are 0.01 and 0.3125 for QR score and RR score , respectively, although other values may clearly be substituted.
- the lead (vector) choice is based upon the maximum value of the sum total of the three factors for each lead.
- the leads are ranked from best to worst as: Lead 2, Lead 3, Lead 1, and Lead 4.
- the default lead order is selected based upon an ideal mean electrical axis of the heart.
- Lead 2 should, theoretically, have the largest ECG amplitude because the electrical projection onto this lead is the greatest with the electrodes 'CF' used for the ECG.
- the other rankings are determined based upon this theory, using the expected relative voltage from the lead. If two or more leads have the same score, the lead is selected based upon this ranking system.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Molecular Biology (AREA)
- Surgery (AREA)
- Biophysics (AREA)
- Pathology (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Medical Informatics (AREA)
- Cardiology (AREA)
- Physics & Mathematics (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Hematology (AREA)
- Physiology (AREA)
- Measurement And Recording Of Electrical Phenomena And Electrical Characteristics Of The Living Body (AREA)
- Measuring Pulse, Heart Rate, Blood Pressure Or Blood Flow (AREA)
Claims (25)
- Vorrichtung zur Herzzeitvolumenbestimmung, die Folgendes umfasst:eine Quelle der Stimulierung, die so adaptiert wurde, dass sie einen Reizstrom produziert,eine erste Schnittstelle, die zum Empfang des Folgenden adaptiert wurde:(i) den ersten Signalen von einer ersten Signalquelle, wobei die ersten Signale wenigstens teilweise mit dem Reizstrom in Zusammenhang stehen, und(ii) EKG-Signale von einer zweiten Signalquelle,ein Schlichtungsinstrument, das so adaptiert wurde, dass es automatisch zwischen einer Vielzahl von EKG-Signalen unterscheidet, wobei es wenigstens auf ihre Qualität bezogen ist und das Schlichtungsinstrument für Folgendes konfiguriert wurde:Generieren einer R-Wellen-Größe, einer QR-Intervalldifferenz und einer RR-Intervalldifferenz auf der Basis der empfangenen einzelnen EKG-Signale,Generieren einer Summe, die auf wenigstens zwei der R-Wellen-Größen, der QR-Intervalldifferenz und der RR-Intervalldifferenz für die einzelnen EKG-Signale basiert,Auswählen von nur einer der EKG-Signale zur Verwendung bei der Herzzeitvolumenbestimmung auf der Basis eines Maximalwertes der generierten Summe für die einzelnen EKG-Signale, undeine zweite Schnittstelle, die so adaptiert wurde, dass sie wenigstens Ausgabedaten an ein Überwachungsvorrichtung bereitstellt.
- Vorrichtung nach Anspruch 1, die ferner ein Datenverarbeitungsgerät umfasst, wobei das Verarbeitungsgerät so adaptiert wurde, dass es wenigstens einen Abschnitt der ersten und EKG-Signale zum Generieren der Ausgabedaten verarbeitet.
- Vorrichtung nach Anspruch 2, wobei das Datenverarbeitungsgerät Folgendes umfasst:wenigstens einen Analog-Digital-Wandler, der so adaptiert wurde, dass er wenigstens die ersten Signale von der analogen Domäne zur digitalen Domäne verwandelt,einen digitalen Prozessor, der operativ an wenigstens einem Wandler angekoppelt ist, und so adaptiert wurde, dass er die Signale der digitalen Domäne verarbeitet.
- Vorrichtung nach Anspruch 3, wobei der digitale Signalprozessor einen digitalen Signalprozessor (DSP) mit einem Computerprogramm umfasst, das darauf läuft, wobei das Computerprogramm ein Programm umfasst, das so adaptiert wurde, dass es wenigstens einen fiduziellen Punkt innerhalb der wenigstens ersten Signale umfasst, die eine Wavelet-Transformation verwenden.
- Vorrichtung nach Anspruch 4, wobei die ersten Signale eine Impedanzwellenform umfassen, und das Computerprogramm so adaptiert wurde, dass es wenigstens einen fiduziellen Punkt innerhalb jeder der Impedanzwellenformen und EKG-Signale zu identifizieren.
- Vorrichtung nach Anspruch 1, wobei die zweite Schnittstelle ferner ein Netzwerkschnittstellengerät umfasst, das so adaptiert wurde, dass es die Übertragung der Ausgabedaten zum Überwachungsgerät über ein Datennetz erleichtert.
- Vorrichtung nach Anspruch 1, wobei die zweite Schnittstelle eine drahtlose Schnittstelle umfasst, die so adaptiert wurde, dass sie die Übertragung der Ausgabedaten zum Überwachungsgerät über eine drahtlose Datenverbindung ermöglicht.
- Vorrichtung nach Anspruch 7, wobei das Uberwachungsgerät ein persönliches Elektronikgerät (PED) umfasst, das so adaptiert wurde, dass es wenigstens einen Abschnitt der Ausgangsdaten darin speichert.
- Vorrichtung nach Anspruch 3, die ferner einen Mikroprozessor umfasst, wobei der Mikroprozessor so konfiguriert wurde, dass er mindestens einen Abschnitt des Vorgangs der Herzzeitvolumenbestimmung und der zweiten Schnittstelle kontrolliert.
- Vorrichtung nach Anspruch 1, wobei die zweite Schnittstelle den Apparat umfasst, das so adaptiert wurde, dass er automatisch zwischen der Vielzahl von EKG-Signalen unterscheidet.
- Vorrichtung nach Anspruch 2, wobei der Vorgang der Auswahl Folgendes umfasst:Empfang der EKG-Signale von vier Kabeln des Verarbeitungsgerätes, wobei jedes der EKG-Signale eine Vielzahl von R-Wellengrößen, QR-Intervalldifferenzen und RR-Intervalldifferenzen hat, die damit assoziiert werden,
- Vorrichtung nach Anspruch 3, die ferner ein Signal-Filtrationsgerät umfasst, das so adaptiert wurde, dass wenigstens einen Abschnitt der ersten und EKG-Signale filtriert werden, bevor sie durch das Verarbeitungsgerät verarbeitet werden.
- Vorrichtung nach Anspruch 12, die ferner ein Demodulatorgerät umfasst, das so adaptiert wurde, dass es die gefilterten ersten Signale vor ihrer Umwandlung zur digitalen Domäne demoduliert.
- Vorrichtung nach Anspruch 3, die ferner eine Vorrichtung umfasst, die so adaptiert wurde, dass sie die Differenz in wenigstens zwei der ersten Signale misst, wobei die Differenz mit einem ersten vorbestimmten Wert verglichen wird, um die elektrische Kontinuität der wenigstens einen der elektrischen Klemmen zu untersuchen, die mit der ersten Signalquelle assoziiert wurden.
- Vorrichtung nach Anspruch 1, wobei die erste Schnittstelle weiter adaptiert wurde, sodass sie Blutdrucksignale von einem Dialysesystem empfängt.
- Vorrichtung nach Anspruch 15, wobei das Dialysesystem und das Herzmessgerät im Wesentlichen gemeinsam untergebracht sind und so adaptiert wurden, dass sie gleichzeitig funktionieren.
- Vorrichtung nach Anspruch 15, wobei das Überwachungsgerät in Datenkommunikation mit dem Dialysesystem und dem Herzmessgerät ist, sodass Daten vom Dialysesystem und Herzmessgerät überwacht werden können, wobei das Überwachungsgerät verwendet wird.
- Vorrichtung nach Anspruch 1, die ferner Folgendes umfasst:eine Vielzahl von Elektrodengruppen, wobei jede Elektrodengruppe eine Vielzahl von Klemmen hat, und wobei wenigstens zwei der Vielzahl der Klemmen in einem vorbestimmten Abstand voneinander angebracht sind,eine Vielzahl von elektrischen Kabeln, die die Quelle der Stimulierung mit einzelnen der Klemmen der Elektrodengruppen verbinden,ein erste Schaltung, die so adaptiert wurde, dass die Differenz in der Spannung an den Klemmen, die sich vom Fluss des Stimulationsstroms durch das Lebewesen ergeben und Klemmen unter verschiedenen Herzleiden eines Lebewesens verwendet werden.eine zweite Schaltung, die so adaptiert wurde, dass sie die EKG-Potenziale von wenigstens einer der Elektrodengruppen misst, undwenigstens eine Verarbeitungsschaltung, die so adaptiert wurde, dass sie die Spannung und EKG-Potenziale verarbeitet und eine Schätzung des Herzzeitvolumens davon entwickelt.
- Vorrichtung nach Anspruch 18, die weiter eine dritte Schaltung umfasst, die so adaptiert wurde, dass sie die Differenz in der Impedanz von wenigstens zwei der Klemmen als Funktion der Zeit misst, wobei die Differenz mit einem ersten Wert zum Evaluieren der elektrischen Kontinuität von wenigstens einer der Klemmen verglichen wird.
- Vorrichtung nach Anspruch 18, wobei wenigsten eine Verarbeitungsschaltung Folgendes umfasst:(i) wenigsten einen Analog-Digital-Wandler, und(ii) wenigstens einen digitalen Prozessor in Datenkommunikation mit wenigstens einem Wandler, wobei der mindestens eine digitale Prozessor wenigstens ein Computerprogramm hat, das darauf läuft.
- Vorrichtung nach Anspruch 18, wobei die zweite Schaltung eine Schaltung umfasst, die so adaptiert wurde, dass sie die Potenziale der Körperoberfläche zwischen wenigstens zwei der Klemmen misst, um eine Vielzahl von komplizierten QRS-Ereignissen innerhalb des Lebewesens zu identifizieren.
- Vorrichtung nach Anspruch 21, wobei die komplizierten QRS-Komplexereignisse wenigstens teilweise unter Verwendung einer Wavelet-Transformation identifiziert werden.
- Vorrichtung nach Anspruch 20, die ferner eine Vorrichtung umfasst, die operativ an der zweiten Schaltung angekoppelt ist und so adaptiert wurde, dass sie eine automatische Auswahl zwischen einer Vielzahl von EKG-Potenzialen macht, was auf wenigstens einem Parameter basiert.
- Vorrichtung nach Anspruch 18, wobei jede Klemme Folgendes umfasst:eine zentrale Achse,einen Seitenwandabschnitt, der im Wesentlichen parallel zur Achse verläuft, undeinen oberen Abschnitt, wobei der obere Abschnitt einen Durchmesser hat, der größer als der des vertikalen Seitenwandabschnitts ist.
- Vorrichtung nach Anspruch 1, wobei das Überwachungsgerät so konfiguriert ist, dass es Informationen anzeigt, die mit dem Herzzeitvolumen in Zusammenhang stehen.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US09/903,473 US6602201B1 (en) | 2000-07-10 | 2001-07-10 | Apparatus and method for determining cardiac output in a living subject |
| US903473 | 2001-07-10 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP1275342A2 EP1275342A2 (de) | 2003-01-15 |
| EP1275342A3 EP1275342A3 (de) | 2004-04-14 |
| EP1275342B1 true EP1275342B1 (de) | 2013-06-19 |
Family
ID=25417562
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP02254832.5A Expired - Lifetime EP1275342B1 (de) | 2001-07-10 | 2002-07-10 | Vorrichtung zur Herzzeitvolumenbestimmung eines Lebewesens |
Country Status (2)
| Country | Link |
|---|---|
| US (1) | US6602201B1 (de) |
| EP (1) | EP1275342B1 (de) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8483818B2 (en) | 2005-05-11 | 2013-07-09 | Cardiac Pacemakers, Inc. | Enhancements to the detection of pulmonary edema when using transthoracic impedance |
| US9089275B2 (en) | 2005-05-11 | 2015-07-28 | Cardiac Pacemakers, Inc. | Sensitivity and specificity of pulmonary edema detection when using transthoracic impedance |
| US9265950B2 (en) | 2001-07-31 | 2016-02-23 | Cardiac Pacemakers, Inc. | Contractility modulation based on impedance signal |
Families Citing this family (111)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AUPQ113799A0 (en) | 1999-06-22 | 1999-07-15 | University Of Queensland, The | A method and device for measuring lymphoedema |
| US6912420B2 (en) * | 2001-04-10 | 2005-06-28 | Cardiac Pacemakers, Inc. | Cardiac rhythm management system for hypotension |
| US6907288B2 (en) * | 2001-04-10 | 2005-06-14 | Cardiac Pacemakers, Inc. | Cardiac rhythm management system adjusting rate response factor for treating hypotension |
| US7933642B2 (en) * | 2001-07-17 | 2011-04-26 | Rud Istvan | Wireless ECG system |
| US7047076B1 (en) | 2001-08-03 | 2006-05-16 | Cardiac Pacemakers, Inc. | Inverted-F antenna configuration for an implantable medical device |
| US20050101875A1 (en) * | 2001-10-04 | 2005-05-12 | Right Corporation | Non-invasive body composition monitor, system and method |
| US7628760B2 (en) * | 2007-02-28 | 2009-12-08 | Semler Scientific, Inc. | Circulation monitoring system and method |
| US7100011B2 (en) * | 2002-03-01 | 2006-08-29 | Arris International, Inc. | Method and system for reducing storage requirements for program code in a communication device |
| AU2002951925A0 (en) * | 2002-10-09 | 2002-10-24 | Queensland University Of Technology | An Impedence Cardiography Device |
| US20040092801A1 (en) * | 2002-11-13 | 2004-05-13 | Budimir Drakulic | System for, and method of, acquiring physiological signals of a patient |
| US7043293B1 (en) | 2002-12-24 | 2006-05-09 | Cardiodynamics International Corporation | Method and apparatus for waveform assessment |
| US20040243192A1 (en) * | 2003-06-02 | 2004-12-02 | Hepp Dennis G. | Physiologic stimulator tuning apparatus and method |
| US7455643B1 (en) | 2003-07-07 | 2008-11-25 | Nellcor Puritan Bennett Ireland | Continuous non-invasive blood pressure measurement apparatus and methods providing automatic recalibration |
| US20050192837A1 (en) * | 2004-02-27 | 2005-09-01 | Cardiac Pacemakers, Inc. | Systems and methods for uploading and distributing medical data sets |
| US20050192843A1 (en) * | 2004-02-27 | 2005-09-01 | Cardiac Pacemakers, Inc. | Systems and methods for validating patient and medical devices information |
| US20050192649A1 (en) * | 2004-02-27 | 2005-09-01 | Cardiac Pacemakers, Inc. | Systems and methods for providing variable medical information |
| US7751894B1 (en) | 2004-03-04 | 2010-07-06 | Cardiac Pacemakers, Inc. | Systems and methods for indicating aberrant behavior detected by an implanted medical device |
| US7806830B2 (en) | 2004-06-16 | 2010-10-05 | Cordeus, Inc. | Apparatus and method for determination of stroke volume using the brachial artery |
| US7261697B2 (en) * | 2004-06-16 | 2007-08-28 | Bernstein Donald P | Apparatus for determination of stroke volume using the brachial artery |
| US8068906B2 (en) | 2004-06-21 | 2011-11-29 | Aorora Technologies Pty Ltd | Cardiac monitoring system |
| US20060004295A1 (en) * | 2004-06-29 | 2006-01-05 | Randolph Prydekker | Electrode connectivity determination system |
| US7387610B2 (en) | 2004-08-19 | 2008-06-17 | Cardiac Pacemakers, Inc. | Thoracic impedance detection with blood resistivity compensation |
| US20060111641A1 (en) * | 2004-11-19 | 2006-05-25 | Applied Cardiac Systems, Inc. | System and method for ICG recording and analysis |
| US7570989B2 (en) * | 2004-11-22 | 2009-08-04 | Cardiodynamics International Corporation | Method and apparatus for signal assessment including event rejection |
| US7270580B2 (en) * | 2004-11-22 | 2007-09-18 | Cardio Dynamics International Corporation | Methods and apparatus for conducting electrical current |
| US7299083B2 (en) | 2004-12-09 | 2007-11-20 | Signalife, Inc. | Electrode for, and method of, indicating signal characteristics at particular positions in a patient's body |
| US7395109B2 (en) | 2004-12-09 | 2008-07-01 | Signalife, Inc. | System for, and method of, monitoring heartbeats of a patient |
| US7856261B2 (en) * | 2004-12-13 | 2010-12-21 | Koninklijke Philips Electronics N.V. | Cable with spacer for positioning multiple medical sensors |
| JP4981688B2 (ja) * | 2005-01-31 | 2012-07-25 | コーニンクレッカ フィリップス エレクトロニクス エヌ ヴィ | 医療センサのためのマルチコンダクタ接続デバイス |
| GB2458389B (en) * | 2005-04-14 | 2010-03-24 | Justin Pisani | Wearable physiological monitoring device |
| EP3539463A1 (de) * | 2005-04-14 | 2019-09-18 | Hidalgo Limited | Überwachungsvorrichtung und -system |
| US7630763B2 (en) * | 2005-04-20 | 2009-12-08 | Cardiac Pacemakers, Inc. | Thoracic or intracardiac impedance detection with automatic vector selection |
| US7603170B2 (en) * | 2005-04-26 | 2009-10-13 | Cardiac Pacemakers, Inc. | Calibration of impedance monitoring of respiratory volumes using thoracic D.C. impedance |
| US7340296B2 (en) | 2005-05-18 | 2008-03-04 | Cardiac Pacemakers, Inc. | Detection of pleural effusion using transthoracic impedance |
| CA2613524C (en) * | 2005-07-01 | 2015-12-29 | Impedance Cardiology Systems Inc. | Pulmonary monitoring system |
| CA2609111C (en) | 2005-07-01 | 2016-10-18 | Scott Chetham | A method and apparatus for performing impedance measurements in accordance with determining an electrode arrangement using a displayed representation |
| CA2608962C (en) | 2005-07-01 | 2016-12-06 | Scott Chetham | Monitoring system |
| CA2615845A1 (en) * | 2005-07-20 | 2007-01-25 | Impedance Cardiology Systems, Inc. | Index determination |
| US9839781B2 (en) | 2005-08-22 | 2017-12-12 | Cardiac Pacemakers, Inc. | Intracardiac impedance and its applications |
| WO2007041783A1 (en) * | 2005-10-11 | 2007-04-19 | Impedance Cardiology Systems, Inc. | Hydration status monitoring |
| US20070167866A1 (en) * | 2005-11-29 | 2007-07-19 | Lopez George A | Cardiac output measurement devices and methods |
| KR20070066417A (ko) * | 2005-12-22 | 2007-06-27 | 삼성전자주식회사 | 휴대형 심전도 측정 장치 |
| ES2545730T3 (es) | 2006-05-30 | 2015-09-15 | Impedimed Limited | Mediciones de impedancia |
| US7569020B2 (en) * | 2006-06-19 | 2009-08-04 | St. Jude Medical Ab | Method for extracting timing parameters using a cardio-mechanical sensor |
| EP2091425A4 (de) | 2006-11-30 | 2012-07-25 | Impedimed Ltd | Messgerät |
| US8290577B2 (en) * | 2007-03-23 | 2012-10-16 | Brooks Donald J | Methods and apparatus for enhanced fiducial point determination and non-invasive hemodynamic parameter determination |
| JP5419861B2 (ja) | 2007-04-20 | 2014-02-19 | インぺディメッド リミテッド | インピーダンス測定装置および方法 |
| US20090105555A1 (en) * | 2007-04-30 | 2009-04-23 | Dacso Clifford C | Non-invasive monitoring of physiological measurements in a distributed health care environment |
| US20090318778A1 (en) * | 2007-04-30 | 2009-12-24 | Clifford Dacso | Non-invasive monitoring of physiological measurements in a distributed health care environment |
| US20110046505A1 (en) | 2007-08-09 | 2011-02-24 | Impedimed Limited | Impedance measurement process |
| US8460189B2 (en) | 2007-09-14 | 2013-06-11 | Corventis, Inc. | Adherent cardiac monitor with advanced sensing capabilities |
| US20090076343A1 (en) | 2007-09-14 | 2009-03-19 | Corventis, Inc. | Energy Management for Adherent Patient Monitor |
| EP2194864B1 (de) | 2007-09-14 | 2018-08-29 | Medtronic Monitoring, Inc. | System und verfahren für die drahtlose überwachung von körperflüssigkeiten |
| WO2009036327A1 (en) | 2007-09-14 | 2009-03-19 | Corventis, Inc. | Adherent device for respiratory monitoring and sleep disordered breathing |
| EP2194847A1 (de) | 2007-09-14 | 2010-06-16 | Corventis, Inc. | Haftende vorrichtung mit mehreren physiologischen sensoren |
| EP2194858B1 (de) | 2007-09-14 | 2017-11-22 | Corventis, Inc. | Automatischer start eines medizinprodukts bei kontakt mit patientengewebe |
| US8684925B2 (en) | 2007-09-14 | 2014-04-01 | Corventis, Inc. | Injectable device for physiological monitoring |
| EP2257216B1 (de) | 2008-03-12 | 2021-04-28 | Medtronic Monitoring, Inc. | Auf herzrhythmus basierende vorhersage von herzinsuffizienzdekompensation |
| WO2009146214A1 (en) | 2008-04-18 | 2009-12-03 | Corventis, Inc. | Method and apparatus to measure bioelectric impedance of patient tissue |
| US8398556B2 (en) | 2008-06-30 | 2013-03-19 | Covidien Lp | Systems and methods for non-invasive continuous blood pressure determination |
| US8660799B2 (en) | 2008-06-30 | 2014-02-25 | Nellcor Puritan Bennett Ireland | Processing and detecting baseline changes in signals |
| US8506498B2 (en) | 2008-07-15 | 2013-08-13 | Nellcor Puritan Bennett Ireland | Systems and methods using induced perturbation to determine physiological parameters |
| US7950971B2 (en) * | 2008-08-08 | 2011-05-31 | Cardiodynamics International Corporation | Electrical connector apparatus and methods |
| US20150201858A1 (en) * | 2008-08-15 | 2015-07-23 | Global Cardiac Monitors, Inc. | Diagnostic device for remote sensing and transmitting biophysiological signals |
| US8532751B2 (en) | 2008-09-30 | 2013-09-10 | Covidien Lp | Laser self-mixing sensors for biological sensing |
| US9301697B2 (en) | 2008-09-30 | 2016-04-05 | Nellcor Puritan Bennett Ireland | Systems and methods for recalibrating a non-invasive blood pressure monitor |
| US9314168B2 (en) | 2008-09-30 | 2016-04-19 | Nellcor Puritan Bennett Ireland | Detecting sleep events using localized blood pressure changes |
| US9687161B2 (en) | 2008-09-30 | 2017-06-27 | Nellcor Puritan Bennett Ireland | Systems and methods for maintaining blood pressure monitor calibration |
| EP2348987B1 (de) | 2008-11-28 | 2017-03-22 | Impedimed Limited | Impedanz-messverfahren |
| JP5326654B2 (ja) * | 2009-02-26 | 2013-10-30 | オムロンヘルスケア株式会社 | 電圧−周波数変換回路およびそれを備えた血圧測定装置 |
| US8216136B2 (en) | 2009-03-05 | 2012-07-10 | Nellcor Puritan Bennett Llc | Systems and methods for monitoring heart rate and blood pressure correlation |
| US20100324404A1 (en) * | 2009-06-22 | 2010-12-23 | Analogic Corporation | Icg/ecg monitoring apparatus |
| US8290730B2 (en) | 2009-06-30 | 2012-10-16 | Nellcor Puritan Bennett Ireland | Systems and methods for assessing measurements in physiological monitoring devices |
| US9198582B2 (en) | 2009-06-30 | 2015-12-01 | Nellcor Puritan Bennett Ireland | Determining a characteristic physiological parameter |
| US8628477B2 (en) | 2009-07-31 | 2014-01-14 | Nellcor Puritan Bennett Ireland | Systems and methods for non-invasive determination of blood pressure |
| US9974463B2 (en) | 2009-08-21 | 2018-05-22 | Beth Israel Deaconess Medical Center, Inc. | Hand-held device for electrical impedance myography |
| US9220440B2 (en) | 2009-09-21 | 2015-12-29 | Nellcor Puritan Bennett Ireland | Determining a characteristic respiration rate |
| US9066660B2 (en) | 2009-09-29 | 2015-06-30 | Nellcor Puritan Bennett Ireland | Systems and methods for high-pass filtering a photoplethysmograph signal |
| US8463347B2 (en) | 2009-09-30 | 2013-06-11 | Nellcor Puritan Bennett Ireland | Systems and methods for normalizing a plethysmograph signal for improved feature analysis |
| US8790259B2 (en) | 2009-10-22 | 2014-07-29 | Corventis, Inc. | Method and apparatus for remote detection and monitoring of functional chronotropic incompetence |
| US9615767B2 (en) | 2009-10-26 | 2017-04-11 | Impedimed Limited | Fluid level indicator determination |
| US9585593B2 (en) | 2009-11-18 | 2017-03-07 | Chung Shing Fan | Signal distribution for patient-electrode measurements |
| US9451897B2 (en) | 2009-12-14 | 2016-09-27 | Medtronic Monitoring, Inc. | Body adherent patch with electronics for physiologic monitoring |
| US9451887B2 (en) | 2010-03-31 | 2016-09-27 | Nellcor Puritan Bennett Ireland | Systems and methods for measuring electromechanical delay of the heart |
| US8965498B2 (en) | 2010-04-05 | 2015-02-24 | Corventis, Inc. | Method and apparatus for personalized physiologic parameters |
| US8898037B2 (en) | 2010-04-28 | 2014-11-25 | Nellcor Puritan Bennett Ireland | Systems and methods for signal monitoring using Lissajous figures |
| US8456295B2 (en) | 2010-05-26 | 2013-06-04 | General Electric Company | Alarm generation method for patient monitoring, physiological monitoring apparatus and computer program product for a physiological monitoring apparatus |
| US8825428B2 (en) | 2010-11-30 | 2014-09-02 | Neilcor Puritan Bennett Ireland | Methods and systems for recalibrating a blood pressure monitor with memory |
| US9259160B2 (en) | 2010-12-01 | 2016-02-16 | Nellcor Puritan Bennett Ireland | Systems and methods for determining when to measure a physiological parameter |
| US9357934B2 (en) | 2010-12-01 | 2016-06-07 | Nellcor Puritan Bennett Ireland | Systems and methods for physiological event marking |
| US8665096B2 (en) | 2010-12-21 | 2014-03-04 | General Electric Company | Alarm control method, physiological monitoring apparatus, and computer program product for a physiological monitoring apparatus |
| GB201101858D0 (en) * | 2011-02-03 | 2011-03-23 | Isansys Lifecare Ltd | Health monitoring |
| US9060695B2 (en) | 2011-11-30 | 2015-06-23 | Covidien Lp | Systems and methods for determining differential pulse transit time from the phase difference of two analog plethysmographs |
| AU2012351988B2 (en) | 2011-12-14 | 2017-05-04 | Impedimed Limited | Devices, systems and methods for determining the relative spatial change in subsurface resistivities across frequencies in tissue |
| US8755873B2 (en) | 2012-09-21 | 2014-06-17 | Welch Allyn, Inc. | Evaluation of the quality of electrode contact with a skin surface |
| CN103083011B (zh) * | 2013-01-22 | 2014-11-05 | 西安交通大学 | 利用胸阻抗二阶差分图辅助实时定位心电r波峰的方法 |
| AT514017B1 (de) | 2013-02-22 | 2020-11-15 | Dr Skrabal Falko | Hämodynamisches EKG |
| US9211110B2 (en) | 2013-03-15 | 2015-12-15 | The Regents Of The University Of Michigan | Lung ventillation measurements using ultrasound |
| KR102164705B1 (ko) * | 2013-06-17 | 2020-10-12 | 삼성전자주식회사 | 착탈형 용량성 결합 능동전극을 이용한 생체 신호 측정 장치 및 방법 |
| JP2016527943A (ja) * | 2013-07-01 | 2016-09-15 | インペディメッド・リミテッド | 組織の湿潤度を決定する小型でウェアラブルな家庭用装置 |
| DE102013011141A1 (de) * | 2013-07-03 | 2015-01-08 | Dräger Medical GmbH | Messvorrichtung zur Messung einer Körperfunktion und Verfahren zum Betrieb einer solchen Messvorrichtung |
| ES2537351B1 (es) * | 2013-11-04 | 2015-12-03 | Universidad De Sevilla | Sensor inteligente de bioimpedancia para aplicaciones biomédicas |
| EP3094247A1 (de) * | 2014-01-17 | 2016-11-23 | Koninklijke Philips N.V. | Erfassung und verarbeitung zuverlässiger ekg-signale und taktimpulse in einer magnetischen resonanzumgebung |
| US10149628B2 (en) | 2014-05-20 | 2018-12-11 | Samsung Electronics Co., Ltd. | Signal processing method and apparatus |
| KR102455629B1 (ko) * | 2014-12-29 | 2022-10-17 | 삼성전자주식회사 | 생체 신호 처리 장치 및 생체 신호 처리 방법 |
| ES2765195T3 (es) | 2015-06-30 | 2020-06-08 | Sunstar Suisse Sa | Sustrato adhesivo desechable adaptado para disponerse en un dispositivo médico |
| WO2017214198A1 (en) * | 2016-06-06 | 2017-12-14 | Tosense, Inc. | Physiological monitor for monitoring patients undergoing hemodialysis |
| US11241198B2 (en) | 2016-09-20 | 2022-02-08 | Nokia Technologies Oy | Method and apparatus for synchronizing impedance cardiography with electrocardiography to lower patient auxiliary current |
| US12303455B2 (en) * | 2020-08-03 | 2025-05-20 | Hill-Rom Services, Inc. | Therapeutic technique using electrical impedance spectroscopy |
| EP4111972B1 (de) * | 2021-06-30 | 2025-01-22 | Wojskowa Akademia Techniczna | Elektrodensegment und elektrodenmatrix für elektrokardiographische und/oder bioimpedanzmessungen |
| CN116098601B (zh) * | 2023-02-09 | 2023-09-19 | 山东埃尔法智慧医疗科技有限公司 | 一种无创心输出量参数的验证方法及设备 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5713367A (en) * | 1994-01-26 | 1998-02-03 | Cambridge Heart, Inc. | Measuring and assessing cardiac electrical stability |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| USRE30101E (en) | 1964-08-19 | 1979-09-25 | Regents Of The University Of Minnesota | Impedance plethysmograph |
| US4577639A (en) * | 1984-11-08 | 1986-03-25 | Spacelabs, Inc. | Apparatus and method for automatic lead selection in electrocardiography |
| US5178151A (en) | 1988-04-20 | 1993-01-12 | Sackner Marvin A | System for non-invasive detection of changes of cardiac volumes and aortic pulses |
| US5103828A (en) * | 1988-07-14 | 1992-04-14 | Bomed Medical Manufacturing, Ltd. | System for therapeutic management of hemodynamic state of patient |
| DE3867234D1 (de) | 1988-08-06 | 1992-02-06 | Hewlett Packard Gmbh | Verfahren und anlage zur datenuebermittlung zwischen einer datenverarbeitungszentrale und nebenstellen. |
| US5511553A (en) | 1989-02-15 | 1996-04-30 | Segalowitz; Jacob | Device-system and method for monitoring multiple physiological parameters (MMPP) continuously and simultaneously |
| US5178154A (en) * | 1990-09-18 | 1993-01-12 | Sorba Medical Systems, Inc. | Impedance cardiograph and method of operation utilizing peak aligned ensemble averaging |
| US5184620A (en) | 1991-12-26 | 1993-02-09 | Marquette Electronics, Inc. | Method of using a multiple electrode pad assembly |
| US5590648A (en) | 1992-11-30 | 1997-01-07 | Tremont Medical | Personal health care system |
| US5505209A (en) * | 1994-07-07 | 1996-04-09 | Reining International, Ltd. | Impedance cardiograph apparatus and method |
| JPH09173310A (ja) * | 1995-11-29 | 1997-07-08 | Hewlett Packard Co <Hp> | Ecg波形の心搏を分類する方法および装置 |
| US6016445A (en) * | 1996-04-16 | 2000-01-18 | Cardiotronics | Method and apparatus for electrode and transthoracic impedance estimation |
| US6141575A (en) | 1996-08-16 | 2000-10-31 | Price; Michael A. | Electrode assemblies |
| US5876351A (en) | 1997-04-10 | 1999-03-02 | Mitchell Rohde | Portable modular diagnostic medical device |
| US5895298A (en) | 1997-05-29 | 1999-04-20 | Biofield Corp. | DC biopotential electrode connector and connector condition sensor |
| WO1998053737A1 (en) * | 1997-05-30 | 1998-12-03 | N.I. Medical Ltd. | Method and system for non-invasive determination of the main cardiorespiratory parameters of the human body |
| US6217525B1 (en) * | 1998-04-30 | 2001-04-17 | Medtronic Physio-Control Manufacturing Corp. | Reduced lead set device and method for detecting acute cardiac ischemic conditions |
| US6223831B1 (en) | 1998-10-28 | 2001-05-01 | Flexi-Coil Ltd. | Differential connecting rod and draft cable for agricultural tillage device |
| US6186955B1 (en) | 1998-11-16 | 2001-02-13 | Gail D. Baura | Noninvasive continuous cardiac output monitor |
| US6416471B1 (en) * | 1999-04-15 | 2002-07-09 | Nexan Limited | Portable remote patient telemonitoring system |
-
2001
- 2001-07-10 US US09/903,473 patent/US6602201B1/en not_active Expired - Lifetime
-
2002
- 2002-07-10 EP EP02254832.5A patent/EP1275342B1/de not_active Expired - Lifetime
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5713367A (en) * | 1994-01-26 | 1998-02-03 | Cambridge Heart, Inc. | Measuring and assessing cardiac electrical stability |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9265950B2 (en) | 2001-07-31 | 2016-02-23 | Cardiac Pacemakers, Inc. | Contractility modulation based on impedance signal |
| US8483818B2 (en) | 2005-05-11 | 2013-07-09 | Cardiac Pacemakers, Inc. | Enhancements to the detection of pulmonary edema when using transthoracic impedance |
| US9089275B2 (en) | 2005-05-11 | 2015-07-28 | Cardiac Pacemakers, Inc. | Sensitivity and specificity of pulmonary edema detection when using transthoracic impedance |
Also Published As
| Publication number | Publication date |
|---|---|
| US6602201B1 (en) | 2003-08-05 |
| EP1275342A2 (de) | 2003-01-15 |
| EP1275342A3 (de) | 2004-04-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1275342B1 (de) | Vorrichtung zur Herzzeitvolumenbestimmung eines Lebewesens | |
| US7251524B1 (en) | Apparatus and method for determining cardiac output in a living subject | |
| Hafid et al. | Full impedance cardiography measurement device using raspberry PI3 and system-on-chip biomedical instrumentation solutions | |
| US6636754B1 (en) | Apparatus and method for determining cardiac output in a living subject | |
| US9211073B2 (en) | Necklace-shaped physiological monitor | |
| US8449469B2 (en) | Two-part patch sensor for monitoring vital signs | |
| CN105832317B (zh) | 用于心输出量、每搏输出量、心力以及血压的测量系统 | |
| CA2352403C (en) | Apparatus and method for determining an approximation of the stroke volume and the cardiac output of the heart | |
| US5511553A (en) | Device-system and method for monitoring multiple physiological parameters (MMPP) continuously and simultaneously | |
| JP2022176978A (ja) | 血液透析を受けている患者を監視するための生理学的監視装置 | |
| US10314496B2 (en) | Necklace-shaped physiological monitor | |
| US20140236037A1 (en) | Necklace-shaped physiological monitor | |
| US12251200B2 (en) | Necklace-shaped physiological monitor | |
| US12502092B2 (en) | Bio electric impedance monitors, electrode arrays and method of use | |
| Sel et al. | ImpediBands: Body coupled bio-impedance patches for physiological sensing proof of concept | |
| US20140213878A1 (en) | Magnetically connected electrode for measuring physiological signals | |
| Hayirlioglu et al. | PhysioPatch: A multimodal and adaptable wearable patch for cardiovascular and cardiopulmonary assessment | |
| US20140236027A1 (en) | Necklace-shaped physiological monitor | |
| US20140213881A1 (en) | Magnetically connected electrode for measuring physiological signals | |
| Manjunatha et al. | ECG and PPG Data Capture using Novel Three Lead Electrode | |
| US20260090753A1 (en) | System and Apparatus for Wearable Hemodynamic Monitoring | |
| US20140213876A1 (en) | Magnetically connected electrode for measuring physiological signals | |
| Alves | A Wearable System for Remote Cardiorespiratory Fitness Monitoring | |
| HK1227666B (zh) | 用於心输出量、每搏输出量、心力以及血压的测量系统 | |
| HK1227666A (en) | Body-worn system for continous, noninvasive measurement of cardiac output, stroke volume, cardiac power, and blood pressure |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;MK;RO;SI |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: 7A 61B 5/00 A Ipc: 7A 61B 5/05 B Ipc: 7A 61B 5/029 B |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK RO SI |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: 7A 61B 5/0424 B Ipc: 7A 61B 5/053 B Ipc: 7A 61B 5/029 A Ipc: 7A 61B 5/00 B Ipc: 7A 61B 5/0452 B |
|
| 17P | Request for examination filed |
Effective date: 20041012 |
|
| AKX | Designation fees paid |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| 17Q | First examination report despatched |
Effective date: 20070402 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: BAURA, GAIL D., C/O CARDIO DYNAMICS INT. CORP. Inventor name: HEPP, DENNIS G., C/O CARDIO DYNAMICS INT. CORP. Inventor name: MALECHA, JEREMY R., C/O CARDIO DYNAMICS INT. CORP. Inventor name: NG, SAU KUEN, C/O CARDIO DYNAMICS INT. CORP. Inventor name: ELF, JAMES O., C/O CARDIO DYNAMICS INT. CORP. |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR IE IT LI LU MC NL PT SE SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 617191 Country of ref document: AT Kind code of ref document: T Effective date: 20130715 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 60245111 Country of ref document: DE Effective date: 20130814 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130920 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130930 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 617191 Country of ref document: AT Kind code of ref document: T Effective date: 20130619 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130919 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20130619 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130531 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20131021 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 Ref country code: CY Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130619 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20140331 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130731 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130731 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 |
|
| 26N | No opposition filed |
Effective date: 20140320 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20130919 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130819 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 60245111 Country of ref document: DE Effective date: 20140320 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130919 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130710 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 60245111 Country of ref document: DE Representative=s name: DREISS PATENTANWAELTE PARTG MBB, DE |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 60245111 Country of ref document: DE Representative=s name: DREISS PATENTANWAELTE PARTG MBB, DE Effective date: 20150223 Ref country code: DE Ref legal event code: R081 Ref document number: 60245111 Country of ref document: DE Owner name: FUJIFILM SONOSITE, INC., BOTHELL, US Free format text: FORMER OWNER: CARDIODYNAMICS INTERNATIONAL CORP., SAN DIEGO, CALIF., US Effective date: 20150223 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20130619 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20130710 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20210616 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R071 Ref document number: 60245111 Country of ref document: DE |