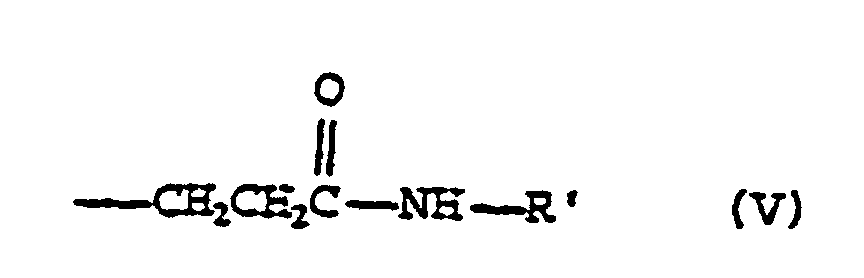EP1027348B1 - 2-(2'-hydroxyphenyl)benzotriazole verwendbar als uv-stabilisatoren - Google Patents
2-(2'-hydroxyphenyl)benzotriazole verwendbar als uv-stabilisatoren Download PDFInfo
- Publication number
- EP1027348B1 EP1027348B1 EP19980955498 EP98955498A EP1027348B1 EP 1027348 B1 EP1027348 B1 EP 1027348B1 EP 19980955498 EP19980955498 EP 19980955498 EP 98955498 A EP98955498 A EP 98955498A EP 1027348 B1 EP1027348 B1 EP 1027348B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- group
- general formula
- linear
- hydroxyphenyl
- branched
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
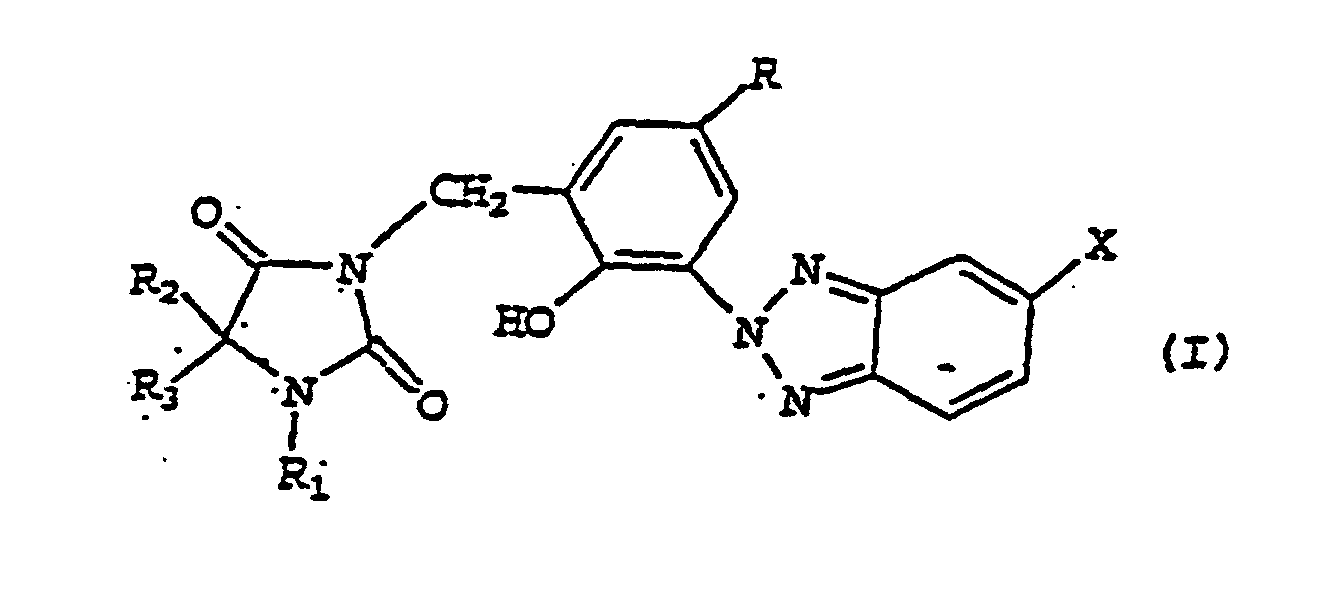
- FJGQBLRYBUAASW-UHFFFAOYSA-N 2-(benzotriazol-2-yl)phenol Chemical class OC1=CC=CC=C1N1N=C2C=CC=CC2=N1 FJGQBLRYBUAASW-UHFFFAOYSA-N 0.000 title claims abstract description 29
- 239000003381 stabilizer Substances 0.000 title claims description 8
- 229920000620 organic polymer Polymers 0.000 claims abstract description 18
- 239000004611 light stabiliser Substances 0.000 claims abstract description 10
- -1 2-ethylhexyl Chemical group 0.000 claims description 90
- 239000000203 mixture Substances 0.000 claims description 56
- 150000001875 compounds Chemical class 0.000 claims description 48
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 28
- 125000000623 heterocyclic group Chemical group 0.000 claims description 24
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 21
- 229910052757 nitrogen Inorganic materials 0.000 claims description 20
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 19
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 claims description 18
- 125000006702 (C1-C18) alkyl group Chemical group 0.000 claims description 16
- 238000000034 method Methods 0.000 claims description 16
- 125000004429 atom Chemical group 0.000 claims description 14
- 125000003342 alkenyl group Chemical group 0.000 claims description 13
- 239000003054 catalyst Substances 0.000 claims description 13
- 229910052760 oxygen Inorganic materials 0.000 claims description 13
- 229910052717 sulfur Inorganic materials 0.000 claims description 13
- 125000005915 C6-C14 aryl group Chemical group 0.000 claims description 12
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 12
- 125000003118 aryl group Chemical group 0.000 claims description 12
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 12
- 125000005842 heteroatom Chemical group 0.000 claims description 12
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 claims description 12
- 239000001301 oxygen Substances 0.000 claims description 12
- 239000011593 sulfur Substances 0.000 claims description 12
- 125000000304 alkynyl group Chemical group 0.000 claims description 11
- 239000004417 polycarbonate Substances 0.000 claims description 11
- 125000002877 alkyl aryl group Chemical group 0.000 claims description 10
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 10
- 229920000515 polycarbonate Polymers 0.000 claims description 9
- GWHJZXXIDMPWGX-UHFFFAOYSA-N 1,2,4-trimethylbenzene Chemical compound CC1=CC=C(C)C(C)=C1 GWHJZXXIDMPWGX-UHFFFAOYSA-N 0.000 claims description 8
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 claims description 8
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 claims description 8
- 125000003545 alkoxy group Chemical group 0.000 claims description 8
- 238000000576 coating method Methods 0.000 claims description 8
- 239000002904 solvent Substances 0.000 claims description 8
- 229920002554 vinyl polymer Polymers 0.000 claims description 8
- 229930040373 Paraformaldehyde Natural products 0.000 claims description 7
- 238000001704 evaporation Methods 0.000 claims description 7
- 239000003960 organic solvent Substances 0.000 claims description 7
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 7
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 6
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 6
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 claims description 6
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 6
- 125000000217 alkyl group Chemical group 0.000 claims description 6
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 6
- 229910052794 bromium Inorganic materials 0.000 claims description 6
- 239000000460 chlorine Substances 0.000 claims description 6
- 229910052801 chlorine Inorganic materials 0.000 claims description 6
- 239000011248 coating agent Substances 0.000 claims description 6
- 230000008020 evaporation Effects 0.000 claims description 6
- 125000005843 halogen group Chemical group 0.000 claims description 6
- WJRBRSLFGCUECM-UHFFFAOYSA-N hydantoin Chemical compound O=C1CNC(=O)N1 WJRBRSLFGCUECM-UHFFFAOYSA-N 0.000 claims description 6
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 6
- 238000002360 preparation method Methods 0.000 claims description 6
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 5
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 5
- XNMQEEKYCVKGBD-UHFFFAOYSA-N 2-butyne Chemical compound CC#CC XNMQEEKYCVKGBD-UHFFFAOYSA-N 0.000 claims description 4
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 claims description 4
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 claims description 4
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 claims description 4
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 claims description 4
- KDKYADYSIPSCCQ-UHFFFAOYSA-N but-1-yne Chemical compound CCC#C KDKYADYSIPSCCQ-UHFFFAOYSA-N 0.000 claims description 4
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 claims description 4
- 125000004185 ester group Chemical group 0.000 claims description 4
- 238000001914 filtration Methods 0.000 claims description 4
- UAEPNZWRGJTJPN-UHFFFAOYSA-N methylcyclohexane Chemical compound CC1CCCCC1 UAEPNZWRGJTJPN-UHFFFAOYSA-N 0.000 claims description 4
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 claims description 3
- 125000002252 acyl group Chemical group 0.000 claims description 3
- 150000001298 alcohols Chemical class 0.000 claims description 3
- 125000003368 amide group Chemical group 0.000 claims description 3
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 3
- 238000009835 boiling Methods 0.000 claims description 3
- 238000002425 crystallisation Methods 0.000 claims description 3
- 230000008025 crystallization Effects 0.000 claims description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 3
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 claims description 3
- 238000010422 painting Methods 0.000 claims description 3
- 150000003335 secondary amines Chemical class 0.000 claims description 3
- HCNHNBLSNVSJTJ-UHFFFAOYSA-N 1,1-Bis(4-hydroxyphenyl)ethane Chemical group C=1C=C(O)C=CC=1C(C)C1=CC=C(O)C=C1 HCNHNBLSNVSJTJ-UHFFFAOYSA-N 0.000 claims description 2
- VYKNVAHOUNIVTQ-UHFFFAOYSA-N 1,2,2,3,3-pentamethylpiperidine Chemical compound CN1CCCC(C)(C)C1(C)C VYKNVAHOUNIVTQ-UHFFFAOYSA-N 0.000 claims description 2
- KKZZSTCINUMTGD-UHFFFAOYSA-N 1,2,2,3-tetramethylpiperidin-4-ol Chemical compound CC1C(O)CCN(C)C1(C)C KKZZSTCINUMTGD-UHFFFAOYSA-N 0.000 claims description 2
- YAXWOADCWUUUNX-UHFFFAOYSA-N 1,2,2,3-tetramethylpiperidine Chemical compound CC1CCCN(C)C1(C)C YAXWOADCWUUUNX-UHFFFAOYSA-N 0.000 claims description 2
- UXBUDIGMOGHUIP-UHFFFAOYSA-N 2,2,3,3,4-pentamethylmorpholine Chemical compound CN1CCOC(C)(C)C1(C)C UXBUDIGMOGHUIP-UHFFFAOYSA-N 0.000 claims description 2
- RKMGAJGJIURJSJ-UHFFFAOYSA-N 2,2,6,6-Tetramethylpiperidine Substances CC1(C)CCCC(C)(C)N1 RKMGAJGJIURJSJ-UHFFFAOYSA-N 0.000 claims description 2
- REYQMSSIXYTZLP-UHFFFAOYSA-N 2,3,3,4-tetramethylmorpholine Chemical compound CC1OCCN(C)C1(C)C REYQMSSIXYTZLP-UHFFFAOYSA-N 0.000 claims description 2
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 claims description 2
- 125000006491 4-t-butyl benzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])*)C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 claims description 2
- 150000001346 alkyl aryl ethers Chemical class 0.000 claims description 2
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 claims description 2
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 claims description 2
- 150000004945 aromatic hydrocarbons Chemical class 0.000 claims description 2
- 239000003849 aromatic solvent Substances 0.000 claims description 2
- 125000004432 carbon atom Chemical group C* 0.000 claims description 2
- UOCJDOLVGGIYIQ-PBFPGSCMSA-N cefatrizine Chemical group S([C@@H]1[C@@H](C(N1C=1C(O)=O)=O)NC(=O)[C@H](N)C=2C=CC(O)=CC=2)CC=1CSC=1C=NNN=1 UOCJDOLVGGIYIQ-PBFPGSCMSA-N 0.000 claims description 2
- 238000001816 cooling Methods 0.000 claims description 2
- RWGFKTVRMDUZSP-UHFFFAOYSA-N cumene Chemical compound CC(C)C1=CC=CC=C1 RWGFKTVRMDUZSP-UHFFFAOYSA-N 0.000 claims description 2
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 2
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 claims description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 2
- GYNNXHKOJHMOHS-UHFFFAOYSA-N methyl-cycloheptane Natural products CC1CCCCCC1 GYNNXHKOJHMOHS-UHFFFAOYSA-N 0.000 claims description 2
- GRVDJDISBSALJP-UHFFFAOYSA-N methyloxidanyl Chemical group [O]C GRVDJDISBSALJP-UHFFFAOYSA-N 0.000 claims description 2
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 2
- 125000001624 naphthyl group Chemical group 0.000 claims description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 2
- MWWATHDPGQKSAR-UHFFFAOYSA-N propyne Chemical compound CC#C MWWATHDPGQKSAR-UHFFFAOYSA-N 0.000 claims description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 2
- 150000003852 triazoles Chemical class 0.000 claims description 2
- 125000006193 alkinyl group Chemical group 0.000 claims 1
- 229910052799 carbon Inorganic materials 0.000 claims 1
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 54
- 229920001577 copolymer Polymers 0.000 description 38
- 229920000642 polymer Polymers 0.000 description 26
- 239000003973 paint Substances 0.000 description 20
- 239000004952 Polyamide Substances 0.000 description 18
- 229920002647 polyamide Polymers 0.000 description 18
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 17
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical compound C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 description 13
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 13
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 12
- 229920000178 Acrylic resin Polymers 0.000 description 11
- 239000004925 Acrylic resin Substances 0.000 description 11
- 229910052751 metal Inorganic materials 0.000 description 11
- 239000002184 metal Substances 0.000 description 11
- 229920005989 resin Polymers 0.000 description 11
- 239000011347 resin Substances 0.000 description 11
- 239000005977 Ethylene Substances 0.000 description 10
- 239000004743 Polypropylene Substances 0.000 description 10
- 229920002857 polybutadiene Polymers 0.000 description 10
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 9
- 239000005062 Polybutadiene Substances 0.000 description 9
- 150000002148 esters Chemical class 0.000 description 9
- 229920001684 low density polyethylene Polymers 0.000 description 9
- 239000004702 low-density polyethylene Substances 0.000 description 9
- 239000000243 solution Substances 0.000 description 9
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 8
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 8
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 8
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 8
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 8
- 229920000728 polyester Polymers 0.000 description 8
- 229920006324 polyoxymethylene Polymers 0.000 description 8
- 239000004721 Polyphenylene oxide Substances 0.000 description 7
- 150000001993 dienes Chemical class 0.000 description 7
- 239000010410 layer Substances 0.000 description 7
- 239000003446 ligand Substances 0.000 description 7
- 229920001169 thermoplastic Polymers 0.000 description 7
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- 229920000877 Melamine resin Polymers 0.000 description 6
- 150000001412 amines Chemical class 0.000 description 6
- 150000001565 benzotriazoles Chemical class 0.000 description 6
- 238000006243 chemical reaction Methods 0.000 description 6
- 229920001228 polyisocyanate Polymers 0.000 description 6
- 239000005056 polyisocyanate Substances 0.000 description 6
- 229920006380 polyphenylene oxide Polymers 0.000 description 6
- 229920001155 polypropylene Polymers 0.000 description 6
- 239000004814 polyurethane Substances 0.000 description 6
- 239000004800 polyvinyl chloride Substances 0.000 description 6
- 229920000915 polyvinyl chloride Polymers 0.000 description 6
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 5
- 229920000122 acrylonitrile butadiene styrene Polymers 0.000 description 5
- 239000000654 additive Substances 0.000 description 5
- 239000007859 condensation product Substances 0.000 description 5
- 229920001971 elastomer Polymers 0.000 description 5
- 229940093476 ethylene glycol Drugs 0.000 description 5
- 239000010408 film Substances 0.000 description 5
- 229920001903 high density polyethylene Polymers 0.000 description 5
- 239000004700 high-density polyethylene Substances 0.000 description 5
- 150000002739 metals Chemical class 0.000 description 5
- 229920000647 polyepoxide Polymers 0.000 description 5
- 150000005846 sugar alcohols Polymers 0.000 description 5
- KAKZBPTYRLMSJV-UHFFFAOYSA-N vinyl-ethylene Natural products C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 5
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 description 4
- BPXVHIRIPLPOPT-UHFFFAOYSA-N 1,3,5-tris(2-hydroxyethyl)-1,3,5-triazinane-2,4,6-trione Chemical compound OCCN1C(=O)N(CCO)C(=O)N(CCO)C1=O BPXVHIRIPLPOPT-UHFFFAOYSA-N 0.000 description 4
- ALVZNPYWJMLXKV-UHFFFAOYSA-N 1,9-Nonanediol Chemical compound OCCCCCCCCCO ALVZNPYWJMLXKV-UHFFFAOYSA-N 0.000 description 4
- CKPKHTKLLYPGFM-UHFFFAOYSA-N 6,6-dimethylheptane-1,1-diol Chemical compound CC(CCCCC(O)O)(C)C CKPKHTKLLYPGFM-UHFFFAOYSA-N 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- 229920002292 Nylon 6 Polymers 0.000 description 4
- YIKSCQDJHCMVMK-UHFFFAOYSA-N Oxamide Chemical compound NC(=O)C(N)=O YIKSCQDJHCMVMK-UHFFFAOYSA-N 0.000 description 4
- 239000004698 Polyethylene Substances 0.000 description 4
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 4
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 4
- 238000010521 absorption reaction Methods 0.000 description 4
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 4
- 125000001931 aliphatic group Chemical group 0.000 description 4
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical compound C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 4
- 239000007795 chemical reaction product Substances 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- 229910052736 halogen Inorganic materials 0.000 description 4
- 150000002367 halogens Chemical class 0.000 description 4
- XXMIOPMDWAUFGU-UHFFFAOYSA-N hexane-1,6-diol Chemical compound OCCCCCCO XXMIOPMDWAUFGU-UHFFFAOYSA-N 0.000 description 4
- 239000012948 isocyanate Substances 0.000 description 4
- 239000000178 monomer Substances 0.000 description 4
- DNIAPMSPPWPWGF-UHFFFAOYSA-N monopropylene glycol Natural products CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 4
- SLCVBVWXLSEKPL-UHFFFAOYSA-N neopentyl glycol Chemical compound OCC(C)(C)CO SLCVBVWXLSEKPL-UHFFFAOYSA-N 0.000 description 4
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 4
- 229920000058 polyacrylate Polymers 0.000 description 4
- 229920000570 polyether Polymers 0.000 description 4
- 229920000573 polyethylene Polymers 0.000 description 4
- 229920002635 polyurethane Polymers 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 235000013772 propylene glycol Nutrition 0.000 description 4
- 229920001897 terpolymer Polymers 0.000 description 4
- 239000004416 thermosoftening plastic Substances 0.000 description 4
- 238000004809 thin layer chromatography Methods 0.000 description 4
- YODZTKMDCQEPHD-UHFFFAOYSA-N thiodiglycol Chemical compound OCCSCCO YODZTKMDCQEPHD-UHFFFAOYSA-N 0.000 description 4
- ZIBGPFATKBEMQZ-UHFFFAOYSA-N triethylene glycol Chemical compound OCCOCCOCCO ZIBGPFATKBEMQZ-UHFFFAOYSA-N 0.000 description 4
- PKTNMTDLDLSRDE-UHFFFAOYSA-N C1OC2(P(=O)=O)OCC1(CO)CO2 Chemical compound C1OC2(P(=O)=O)OCC1(CO)CO2 PKTNMTDLDLSRDE-UHFFFAOYSA-N 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- 229920002943 EPDM rubber Polymers 0.000 description 3
- 229920002396 Polyurea Polymers 0.000 description 3
- 229920000180 alkyd Polymers 0.000 description 3
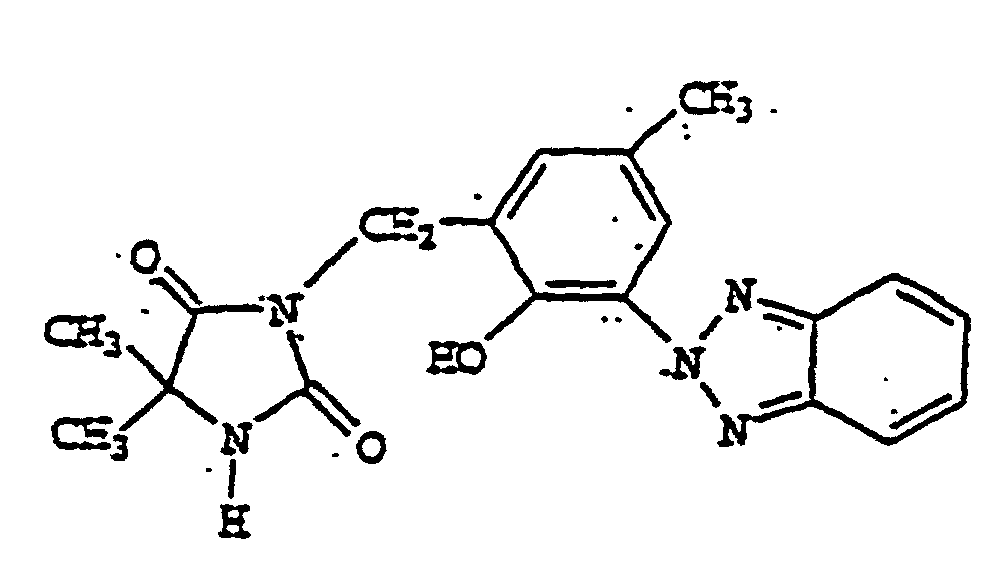
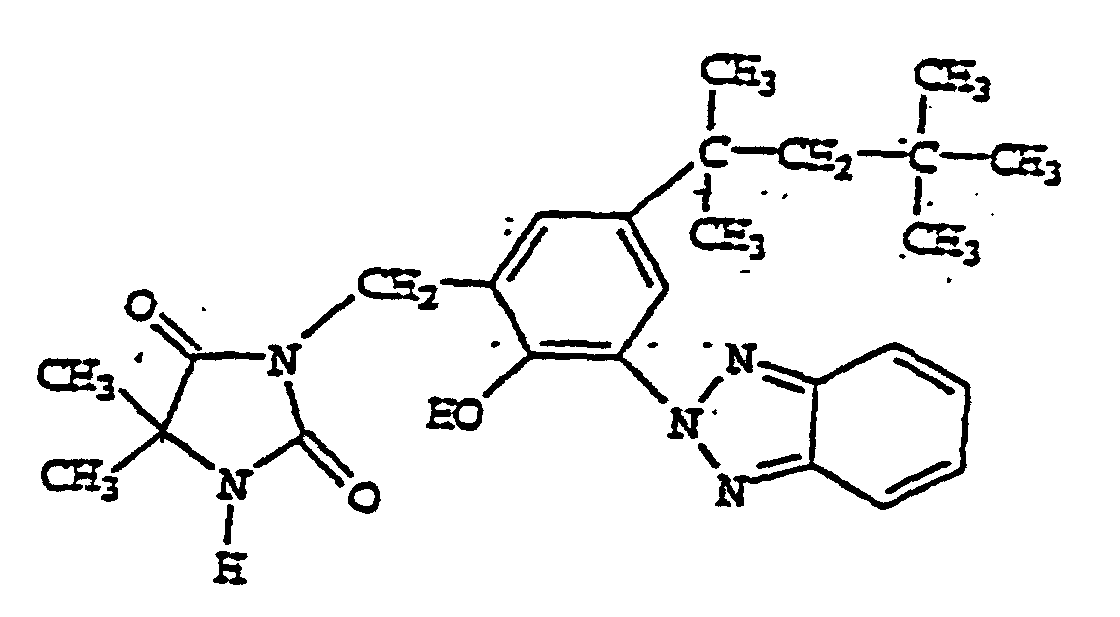
- FQUNFJULCYSSOP-UHFFFAOYSA-N bisoctrizole Chemical compound N1=C2C=CC=CC2=NN1C1=CC(C(C)(C)CC(C)(C)C)=CC(CC=2C(=C(C=C(C=2)C(C)(C)CC(C)(C)C)N2N=C3C=CC=CC3=N2)O)=C1O FQUNFJULCYSSOP-UHFFFAOYSA-N 0.000 description 3
- 239000000806 elastomer Substances 0.000 description 3
- 239000003822 epoxy resin Substances 0.000 description 3
- HQQADJVZYDDRJT-UHFFFAOYSA-N ethene;prop-1-ene Chemical group C=C.CC=C HQQADJVZYDDRJT-UHFFFAOYSA-N 0.000 description 3
- 239000005038 ethylene vinyl acetate Substances 0.000 description 3
- 229920006226 ethylene-acrylic acid Polymers 0.000 description 3
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 3
- 150000002815 nickel Chemical class 0.000 description 3
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 3
- 229920000139 polyethylene terephthalate Polymers 0.000 description 3
- 239000005020 polyethylene terephthalate Substances 0.000 description 3
- 238000006116 polymerization reaction Methods 0.000 description 3
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 3
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 229940117958 vinyl acetate Drugs 0.000 description 3
- ZEBMSMUPGIOANU-UHFFFAOYSA-N (3,5-ditert-butyl-4-hydroxyphenyl)methylphosphonic acid Chemical compound CC(C)(C)C1=CC(CP(O)(O)=O)=CC(C(C)(C)C)=C1O ZEBMSMUPGIOANU-UHFFFAOYSA-N 0.000 description 2
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- YHMYGUUIMTVXNW-UHFFFAOYSA-N 1,3-dihydrobenzimidazole-2-thione Chemical compound C1=CC=C2NC(S)=NC2=C1 YHMYGUUIMTVXNW-UHFFFAOYSA-N 0.000 description 2
- 150000005208 1,4-dihydroxybenzenes Chemical class 0.000 description 2
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 2
- HECLRDQVFMWTQS-RGOKHQFPSA-N 1755-01-7 Chemical compound C1[C@H]2[C@@H]3CC=C[C@@H]3[C@@H]1C=C2 HECLRDQVFMWTQS-RGOKHQFPSA-N 0.000 description 2
- 238000005160 1H NMR spectroscopy Methods 0.000 description 2
- SLUKQUGVTITNSY-UHFFFAOYSA-N 2,6-di-tert-butyl-4-methoxyphenol Chemical compound COC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 SLUKQUGVTITNSY-UHFFFAOYSA-N 0.000 description 2
- OEPOKWHJYJXUGD-UHFFFAOYSA-N 2-(3-phenylmethoxyphenyl)-1,3-thiazole-4-carbaldehyde Chemical compound O=CC1=CSC(C=2C=C(OCC=3C=CC=CC=3)C=CC=2)=N1 OEPOKWHJYJXUGD-UHFFFAOYSA-N 0.000 description 2
- IYAZLDLPUNDVAG-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-4-(2,4,4-trimethylpentan-2-yl)phenol Chemical compound CC(C)(C)CC(C)(C)C1=CC=C(O)C(N2N=C3C=CC=CC3=N2)=C1 IYAZLDLPUNDVAG-UHFFFAOYSA-N 0.000 description 2
- QMNZHUUXEVHCPY-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-6-[(dipropylamino)methyl]-4-(2,4,4-trimethylpentan-2-yl)phenol Chemical compound CCCN(CCC)CC1=CC(C(C)(C)CC(C)(C)C)=CC(N2N=C3C=CC=CC3=N2)=C1O QMNZHUUXEVHCPY-UHFFFAOYSA-N 0.000 description 2
- KVLHIJXLYHBARH-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-6-[(dipropylamino)methyl]-4-methylphenol Chemical compound CCCN(CCC)CC1=CC(C)=CC(N2N=C3C=CC=CC3=N2)=C1O KVLHIJXLYHBARH-UHFFFAOYSA-N 0.000 description 2
- RXFCIXRFAJRBSG-UHFFFAOYSA-N 3,2,3-tetramine Chemical compound NCCCNCCNCCCN RXFCIXRFAJRBSG-UHFFFAOYSA-N 0.000 description 2
- HCILJBJJZALOAL-UHFFFAOYSA-N 3-(3,5-ditert-butyl-4-hydroxyphenyl)-n'-[3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoyl]propanehydrazide Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CCC(=O)NNC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 HCILJBJJZALOAL-UHFFFAOYSA-N 0.000 description 2
- WPMYUUITDBHVQZ-UHFFFAOYSA-N 3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoic acid Chemical compound CC(C)(C)C1=CC(CCC(O)=O)=CC(C(C)(C)C)=C1O WPMYUUITDBHVQZ-UHFFFAOYSA-N 0.000 description 2
- ACZGCWSMSTYWDQ-UHFFFAOYSA-N 3h-1-benzofuran-2-one Chemical class C1=CC=C2OC(=O)CC2=C1 ACZGCWSMSTYWDQ-UHFFFAOYSA-N 0.000 description 2
- YIROYDNZEPTFOL-UHFFFAOYSA-N 5,5-Dimethylhydantoin Chemical compound CC1(C)NC(=O)NC1=O YIROYDNZEPTFOL-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 2
- 239000013032 Hydrocarbon resin Substances 0.000 description 2
- OWYWGLHRNBIFJP-UHFFFAOYSA-N Ipazine Chemical compound CCN(CC)C1=NC(Cl)=NC(NC(C)C)=N1 OWYWGLHRNBIFJP-UHFFFAOYSA-N 0.000 description 2
- 229920010126 Linear Low Density Polyethylene (LLDPE) Polymers 0.000 description 2
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical compound O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 description 2
- BAPJBEWLBFYGME-UHFFFAOYSA-N Methyl acrylate Chemical compound COC(=O)C=C BAPJBEWLBFYGME-UHFFFAOYSA-N 0.000 description 2
- 229920002367 Polyisobutene Polymers 0.000 description 2
- 239000004734 Polyphenylene sulfide Substances 0.000 description 2
- KKEYFWRCBNTPAC-UHFFFAOYSA-N Terephthalic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-N 0.000 description 2
- 239000007983 Tris buffer Substances 0.000 description 2
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 2
- BZHJMEDXRYGGRV-UHFFFAOYSA-N Vinyl chloride Chemical compound ClC=C BZHJMEDXRYGGRV-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 229920000800 acrylic rubber Polymers 0.000 description 2
- 229920002877 acrylic styrene acrylonitrile Polymers 0.000 description 2
- 239000012190 activator Substances 0.000 description 2
- 239000001361 adipic acid Substances 0.000 description 2
- 235000011037 adipic acid Nutrition 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 125000005250 alkyl acrylate group Chemical group 0.000 description 2
- XYLMUPLGERFSHI-UHFFFAOYSA-N alpha-Methylstyrene Chemical compound CC(=C)C1=CC=CC=C1 XYLMUPLGERFSHI-UHFFFAOYSA-N 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 150000008064 anhydrides Chemical group 0.000 description 2
- 229940051881 anilide analgesics and antipyretics Drugs 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- DQXBYHZEEUGOBF-UHFFFAOYSA-N but-3-enoic acid;ethene Chemical compound C=C.OC(=O)CC=C DQXBYHZEEUGOBF-UHFFFAOYSA-N 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 229910002091 carbon monoxide Inorganic materials 0.000 description 2
- 238000005266 casting Methods 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 238000004040 coloring Methods 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 238000004132 cross linking Methods 0.000 description 2
- 239000003431 cross linking reagent Substances 0.000 description 2
- LPIQUOYDBNQMRZ-UHFFFAOYSA-N cyclopentene Chemical compound C1CC=CC1 LPIQUOYDBNQMRZ-UHFFFAOYSA-N 0.000 description 2
- 150000001991 dicarboxylic acids Chemical class 0.000 description 2
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical class C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 2
- WEHWNAOGRSTTBQ-UHFFFAOYSA-N dipropylamine Chemical compound CCCNCCC WEHWNAOGRSTTBQ-UHFFFAOYSA-N 0.000 description 2
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical group [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- MCPKSFINULVDNX-UHFFFAOYSA-N drometrizole Chemical compound CC1=CC=C(O)C(N2N=C3C=CC=CC3=N2)=C1 MCPKSFINULVDNX-UHFFFAOYSA-N 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 238000000921 elemental analysis Methods 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 150000002170 ethers Chemical class 0.000 description 2
- VXNZUUAINFGPBY-UHFFFAOYSA-N ethyl ethylene Natural products CCC=C VXNZUUAINFGPBY-UHFFFAOYSA-N 0.000 description 2
- 238000001125 extrusion Methods 0.000 description 2
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 2
- 150000004820 halides Chemical class 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- NAQMVNRVTILPCV-UHFFFAOYSA-N hexane-1,6-diamine Chemical compound NCCCCCCN NAQMVNRVTILPCV-UHFFFAOYSA-N 0.000 description 2
- 229920001519 homopolymer Polymers 0.000 description 2
- 150000001469 hydantoins Chemical class 0.000 description 2
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 2
- 229920006270 hydrocarbon resin Polymers 0.000 description 2
- 229920000554 ionomer Polymers 0.000 description 2
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 2
- 229920000092 linear low density polyethylene Polymers 0.000 description 2
- 239000004707 linear low-density polyethylene Substances 0.000 description 2
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 2
- JDSHMPZPIAZGSV-UHFFFAOYSA-N melamine Chemical compound NC1=NC(N)=NC(N)=N1 JDSHMPZPIAZGSV-UHFFFAOYSA-N 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 150000002734 metacrylic acid derivatives Chemical class 0.000 description 2
- UJRDRFZCRQNLJM-UHFFFAOYSA-N methyl 3-[3-(benzotriazol-2-yl)-5-tert-butyl-4-hydroxyphenyl]propanoate Chemical compound CC(C)(C)C1=CC(CCC(=O)OC)=CC(N2N=C3C=CC=CC3=N2)=C1O UJRDRFZCRQNLJM-UHFFFAOYSA-N 0.000 description 2
- 150000005673 monoalkenes Chemical class 0.000 description 2
- UKJARPDLRWBRAX-UHFFFAOYSA-N n,n'-bis(2,2,6,6-tetramethylpiperidin-4-yl)hexane-1,6-diamine Chemical compound C1C(C)(C)NC(C)(C)CC1NCCCCCCNC1CC(C)(C)NC(C)(C)C1 UKJARPDLRWBRAX-UHFFFAOYSA-N 0.000 description 2
- 230000000737 periodic effect Effects 0.000 description 2
- QQVIHTHCMHWDBS-UHFFFAOYSA-N perisophthalic acid Natural products OC(=O)C1=CC=CC(C(O)=O)=C1 QQVIHTHCMHWDBS-UHFFFAOYSA-N 0.000 description 2
- ZQBAKBUEJOMQEX-UHFFFAOYSA-N phenyl salicylate Chemical compound OC1=CC=CC=C1C(=O)OC1=CC=CC=C1 ZQBAKBUEJOMQEX-UHFFFAOYSA-N 0.000 description 2
- AQSJGOWTSHOLKH-UHFFFAOYSA-N phosphite(3-) Chemical class [O-]P([O-])[O-] AQSJGOWTSHOLKH-UHFFFAOYSA-N 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 2
- 229920002239 polyacrylonitrile Polymers 0.000 description 2
- 229920001281 polyalkylene Polymers 0.000 description 2
- 239000004645 polyester resin Substances 0.000 description 2
- 229920001225 polyester resin Polymers 0.000 description 2
- 229920000098 polyolefin Polymers 0.000 description 2
- 229920000069 polyphenylene sulfide Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 239000005060 rubber Substances 0.000 description 2
- 230000006641 stabilisation Effects 0.000 description 2
- 238000011105 stabilization Methods 0.000 description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-N succinic acid Chemical compound OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 2
- 239000002344 surface layer Substances 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- KKEYFWRCBNTPAC-UHFFFAOYSA-L terephthalate(2-) Chemical compound [O-]C(=O)C1=CC=C(C([O-])=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-L 0.000 description 2
- HVLLSGMXQDNUAL-UHFFFAOYSA-N triphenyl phosphite Chemical compound C=1C=CC=CC=1OP(OC=1C=CC=CC=1)OC1=CC=CC=C1 HVLLSGMXQDNUAL-UHFFFAOYSA-N 0.000 description 2
- BOXSVZNGTQTENJ-UHFFFAOYSA-L zinc dibutyldithiocarbamate Chemical compound [Zn+2].CCCCN(C([S-])=S)CCCC.CCCCN(C([S-])=S)CCCC BOXSVZNGTQTENJ-UHFFFAOYSA-L 0.000 description 2
- WGVKWNUPNGFDFJ-DQCZWYHMSA-N β-tocopherol Chemical compound OC1=CC(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C WGVKWNUPNGFDFJ-DQCZWYHMSA-N 0.000 description 2
- GZIFEOYASATJEH-VHFRWLAGSA-N δ-tocopherol Chemical compound OC1=CC(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1 GZIFEOYASATJEH-VHFRWLAGSA-N 0.000 description 2
- OUBISKKOUYNDML-UHFFFAOYSA-N (2,2,6,6-tetramethylpiperidin-4-yl) 2-[bis[2-oxo-2-(2,2,6,6-tetramethylpiperidin-4-yl)oxyethyl]amino]acetate Chemical compound C1C(C)(C)NC(C)(C)CC1OC(=O)CN(CC(=O)OC1CC(C)(C)NC(C)(C)C1)CC(=O)OC1CC(C)(C)NC(C)(C)C1 OUBISKKOUYNDML-UHFFFAOYSA-N 0.000 description 1
- HQEPZWYPQQKFLU-UHFFFAOYSA-N (2,6-dihydroxyphenyl)-phenylmethanone Chemical compound OC1=CC=CC(O)=C1C(=O)C1=CC=CC=C1 HQEPZWYPQQKFLU-UHFFFAOYSA-N 0.000 description 1
- HJIAMFHSAAEUKR-UHFFFAOYSA-N (2-hydroxyphenyl)-phenylmethanone Chemical class OC1=CC=CC=C1C(=O)C1=CC=CC=C1 HJIAMFHSAAEUKR-UHFFFAOYSA-N 0.000 description 1
- ATLWFAZCZPSXII-UHFFFAOYSA-N (2-octylphenyl) 2-hydroxybenzoate Chemical compound CCCCCCCCC1=CC=CC=C1OC(=O)C1=CC=CC=C1O ATLWFAZCZPSXII-UHFFFAOYSA-N 0.000 description 1
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- FKFOHTUAFNQANW-UHFFFAOYSA-N (3,5-ditert-butyl-4-hydroxyphenyl) octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 FKFOHTUAFNQANW-UHFFFAOYSA-N 0.000 description 1
- GOZHNJTXLALKRL-UHFFFAOYSA-N (5-benzoyl-2,4-dihydroxyphenyl)-phenylmethanone Chemical compound OC1=CC(O)=C(C(=O)C=2C=CC=CC=2)C=C1C(=O)C1=CC=CC=C1 GOZHNJTXLALKRL-UHFFFAOYSA-N 0.000 description 1
- OJOWICOBYCXEKR-KRXBUXKQSA-N (5e)-5-ethylidenebicyclo[2.2.1]hept-2-ene Chemical compound C1C2C(=C/C)/CC1C=C2 OJOWICOBYCXEKR-KRXBUXKQSA-N 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N (R)-alpha-Tocopherol Natural products OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
- VCMZIKKVYXGKCI-UHFFFAOYSA-N 1,1-bis(2,4-ditert-butyl-6-methylphenyl)-2,2-bis(hydroxymethyl)propane-1,3-diol dihydroxyphosphanyl dihydrogen phosphite Chemical compound OP(O)OP(O)O.C(C)(C)(C)C1=C(C(=CC(=C1)C(C)(C)C)C)C(O)(C(CO)(CO)CO)C1=C(C=C(C=C1C)C(C)(C)C)C(C)(C)C VCMZIKKVYXGKCI-UHFFFAOYSA-N 0.000 description 1
- RGASRBUYZODJTG-UHFFFAOYSA-N 1,1-bis(2,4-ditert-butylphenyl)-2,2-bis(hydroxymethyl)propane-1,3-diol dihydroxyphosphanyl dihydrogen phosphite Chemical compound OP(O)OP(O)O.C(C)(C)(C)C1=C(C=CC(=C1)C(C)(C)C)C(O)(C(CO)(CO)CO)C1=C(C=C(C=C1)C(C)(C)C)C(C)(C)C RGASRBUYZODJTG-UHFFFAOYSA-N 0.000 description 1
- LENJMLXEFOXTRG-UHFFFAOYSA-N 1,1-bis(2,5-ditert-butyl-4-methylphenyl)-2,2-bis(hydroxymethyl)propane-1,3-diol dihydroxyphosphanyl dihydrogen phosphite Chemical compound OP(O)OP(O)O.C(C)(C)(C)C=1C=C(C(=CC1C)C(C)(C)C)C(O)(C(CO)(CO)CO)C1=CC(=C(C=C1C(C)(C)C)C)C(C)(C)C LENJMLXEFOXTRG-UHFFFAOYSA-N 0.000 description 1
- VNQNXQYZMPJLQX-UHFFFAOYSA-N 1,3,5-tris[(3,5-ditert-butyl-4-hydroxyphenyl)methyl]-1,3,5-triazinane-2,4,6-trione Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CN2C(N(CC=3C=C(C(O)=C(C=3)C(C)(C)C)C(C)(C)C)C(=O)N(CC=3C=C(C(O)=C(C=3)C(C)(C)C)C(C)(C)C)C2=O)=O)=C1 VNQNXQYZMPJLQX-UHFFFAOYSA-N 0.000 description 1
- MYMKXVFDVQUQLG-UHFFFAOYSA-N 1,3,7,9-tetratert-butyl-11-fluoro-5-methyl-5h-benzo[d][1,3,2]benzodioxaphosphocine Chemical compound CC1C2=CC(C(C)(C)C)=CC(C(C)(C)C)=C2OP(F)OC2=C1C=C(C(C)(C)C)C=C2C(C)(C)C MYMKXVFDVQUQLG-UHFFFAOYSA-N 0.000 description 1
- VMDYMJSKWCVEEB-UHFFFAOYSA-N 1-[3,5-bis[3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoyl]-1,3,5-triazinan-1-yl]-3-(3,5-ditert-butyl-4-hydroxyphenyl)propan-1-one Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CCC(=O)N2CN(CN(C2)C(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)C(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 VMDYMJSKWCVEEB-UHFFFAOYSA-N 0.000 description 1
- KEXRSLVRFLEMHJ-UHFFFAOYSA-N 1-o,4-o-bis[(4-tert-butyl-3-hydroxy-2,6-dimethylphenyl)methyl] benzene-1,4-dicarbothioate Chemical compound CC1=CC(C(C)(C)C)=C(O)C(C)=C1COC(=S)C1=CC=C(C(=S)OCC=2C(=C(O)C(=CC=2C)C(C)(C)C)C)C=C1 KEXRSLVRFLEMHJ-UHFFFAOYSA-N 0.000 description 1
- TUYBEVLJKZQJPO-UHFFFAOYSA-N 19-(3,5-ditert-butyl-4-hydroxyphenyl)heptatriacontan-19-ylphosphonic acid Chemical compound CCCCCCCCCCCCCCCCCCC(CCCCCCCCCCCCCCCCCC)(P(O)(O)=O)C1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 TUYBEVLJKZQJPO-UHFFFAOYSA-N 0.000 description 1
- CDWOTAMGTNNLHY-UHFFFAOYSA-N 19-(3-tert-butyl-4-hydroxy-5-methylphenyl)heptatriacontan-19-ylphosphonic acid Chemical compound CCCCCCCCCCCCCCCCCCC(CCCCCCCCCCCCCCCCCC)(P(O)(O)=O)C1=CC(C)=C(O)C(C(C)(C)C)=C1 CDWOTAMGTNNLHY-UHFFFAOYSA-N 0.000 description 1
- KGRVJHAUYBGFFP-UHFFFAOYSA-N 2,2'-Methylenebis(4-methyl-6-tert-butylphenol) Chemical compound CC(C)(C)C1=CC(C)=CC(CC=2C(=C(C=C(C)C=2)C(C)(C)C)O)=C1O KGRVJHAUYBGFFP-UHFFFAOYSA-N 0.000 description 1
- IOHCSOXUCWUZQJ-UHFFFAOYSA-J 2,2'-spirobi[1,3,2-benzodioxastannole] Chemical compound O1c2ccccc2O[Sn]11Oc2ccccc2O1 IOHCSOXUCWUZQJ-UHFFFAOYSA-J 0.000 description 1
- BWJKLDGAAPQXGO-UHFFFAOYSA-N 2,2,6,6-tetramethyl-4-octadecoxypiperidine Chemical compound CCCCCCCCCCCCCCCCCCOC1CC(C)(C)NC(C)(C)C1 BWJKLDGAAPQXGO-UHFFFAOYSA-N 0.000 description 1
- DCOZBPTXZNTCFM-UHFFFAOYSA-N 2,2-bis(2,2,6,6-tetramethyl-1-octoxypiperidin-3-yl)decanedioic acid Chemical compound CC1(C)N(OCCCCCCCC)C(C)(C)CCC1C(CCCCCCCC(O)=O)(C(O)=O)C1C(C)(C)N(OCCCCCCCC)C(C)(C)CC1 DCOZBPTXZNTCFM-UHFFFAOYSA-N 0.000 description 1
- VKCMZEUOQWUNAR-UHFFFAOYSA-N 2,2-bis(2,4-ditert-butyl-6-methylphenyl)ethyl dihydrogen phosphite Chemical compound CC1=CC(C(C)(C)C)=CC(C(C)(C)C)=C1C(COP(O)O)C1=C(C)C=C(C(C)(C)C)C=C1C(C)(C)C VKCMZEUOQWUNAR-UHFFFAOYSA-N 0.000 description 1
- GXURZKWLMYOCDX-UHFFFAOYSA-N 2,2-bis(hydroxymethyl)propane-1,3-diol;dihydroxyphosphanyl dihydrogen phosphite Chemical compound OP(O)OP(O)O.OCC(CO)(CO)CO GXURZKWLMYOCDX-UHFFFAOYSA-N 0.000 description 1
- HIQBOYNRKGWHAU-UHFFFAOYSA-N 2,3-bis(2,2,6,6-tetramethylpiperidin-1-yl)butanedioic acid Chemical compound CC1(N(C(CCC1)(C)C)C(C(C(=O)O)N1C(CCCC1(C)C)(C)C)C(=O)O)C HIQBOYNRKGWHAU-UHFFFAOYSA-N 0.000 description 1
- BJELTSYBAHKXRW-UHFFFAOYSA-N 2,4,6-triallyloxy-1,3,5-triazine Chemical compound C=CCOC1=NC(OCC=C)=NC(OCC=C)=N1 BJELTSYBAHKXRW-UHFFFAOYSA-N 0.000 description 1
- UUAIOYWXCDLHKT-UHFFFAOYSA-N 2,4,6-tricyclohexylphenol Chemical compound OC1=C(C2CCCCC2)C=C(C2CCCCC2)C=C1C1CCCCC1 UUAIOYWXCDLHKT-UHFFFAOYSA-N 0.000 description 1
- OSPBEQGPLJSTKW-UHFFFAOYSA-N 2,4,6-tris[(3,5-ditert-butyl-4-hydroxyphenyl)methyl]phenol Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CC=2C=C(CC=3C=C(C(O)=C(C=3)C(C)(C)C)C(C)(C)C)C(O)=C(CC=3C=C(C(O)=C(C=3)C(C)(C)C)C(C)(C)C)C=2)=C1 OSPBEQGPLJSTKW-UHFFFAOYSA-N 0.000 description 1
- OPLCSTZDXXUYDU-UHFFFAOYSA-N 2,4-dimethyl-6-tert-butylphenol Chemical compound CC1=CC(C)=C(O)C(C(C)(C)C)=C1 OPLCSTZDXXUYDU-UHFFFAOYSA-N 0.000 description 1
- LXWZXEJDKYWBOW-UHFFFAOYSA-N 2,4-ditert-butyl-6-[(3,5-ditert-butyl-2-hydroxyphenyl)methyl]phenol Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=CC(CC=2C(=C(C=C(C=2)C(C)(C)C)C(C)(C)C)O)=C1O LXWZXEJDKYWBOW-UHFFFAOYSA-N 0.000 description 1
- DXCHWXWXYPEZKM-UHFFFAOYSA-N 2,4-ditert-butyl-6-[1-(3,5-ditert-butyl-2-hydroxyphenyl)ethyl]phenol Chemical compound C=1C(C(C)(C)C)=CC(C(C)(C)C)=C(O)C=1C(C)C1=CC(C(C)(C)C)=CC(C(C)(C)C)=C1O DXCHWXWXYPEZKM-UHFFFAOYSA-N 0.000 description 1
- CZNRFEXEPBITDS-UHFFFAOYSA-N 2,5-bis(2-methylbutan-2-yl)benzene-1,4-diol Chemical compound CCC(C)(C)C1=CC(O)=C(C(C)(C)CC)C=C1O CZNRFEXEPBITDS-UHFFFAOYSA-N 0.000 description 1
- JZODKRWQWUWGCD-UHFFFAOYSA-N 2,5-di-tert-butylbenzene-1,4-diol Chemical compound CC(C)(C)C1=CC(O)=C(C(C)(C)C)C=C1O JZODKRWQWUWGCD-UHFFFAOYSA-N 0.000 description 1
- FLLRQABPKFCXSO-UHFFFAOYSA-N 2,5-ditert-butyl-4-methoxyphenol Chemical compound COC1=CC(C(C)(C)C)=C(O)C=C1C(C)(C)C FLLRQABPKFCXSO-UHFFFAOYSA-N 0.000 description 1
- JFGVTUJBHHZRAB-UHFFFAOYSA-N 2,6-Di-tert-butyl-1,4-benzenediol Chemical compound CC(C)(C)C1=CC(O)=CC(C(C)(C)C)=C1O JFGVTUJBHHZRAB-UHFFFAOYSA-N 0.000 description 1
- RPLXHDXNCZNHRA-UHFFFAOYSA-N 2,6-bis(dodecylsulfanylmethyl)-4-nonylphenol Chemical compound CCCCCCCCCCCCSCC1=CC(CCCCCCCCC)=CC(CSCCCCCCCCCCCC)=C1O RPLXHDXNCZNHRA-UHFFFAOYSA-N 0.000 description 1
- LKALLEFLBKHPTQ-UHFFFAOYSA-N 2,6-bis[(3-tert-butyl-2-hydroxy-5-methylphenyl)methyl]-4-methylphenol Chemical compound OC=1C(CC=2C(=C(C=C(C)C=2)C(C)(C)C)O)=CC(C)=CC=1CC1=CC(C)=CC(C(C)(C)C)=C1O LKALLEFLBKHPTQ-UHFFFAOYSA-N 0.000 description 1
- BVUXDWXKPROUDO-UHFFFAOYSA-N 2,6-di-tert-butyl-4-ethylphenol Chemical compound CCC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 BVUXDWXKPROUDO-UHFFFAOYSA-N 0.000 description 1
- FRAQIHUDFAFXHT-UHFFFAOYSA-N 2,6-dicyclopentyl-4-methylphenol Chemical compound OC=1C(C2CCCC2)=CC(C)=CC=1C1CCCC1 FRAQIHUDFAFXHT-UHFFFAOYSA-N 0.000 description 1
- JBYWTKPHBLYYFJ-UHFFFAOYSA-N 2,6-ditert-butyl-4-(2-methylpropyl)phenol Chemical compound CC(C)CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 JBYWTKPHBLYYFJ-UHFFFAOYSA-N 0.000 description 1
- GJDRKHHGPHLVNI-UHFFFAOYSA-N 2,6-ditert-butyl-4-(diethoxyphosphorylmethyl)phenol Chemical compound CCOP(=O)(OCC)CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 GJDRKHHGPHLVNI-UHFFFAOYSA-N 0.000 description 1
- SCXYLTWTWUGEAA-UHFFFAOYSA-N 2,6-ditert-butyl-4-(methoxymethyl)phenol Chemical compound COCC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 SCXYLTWTWUGEAA-UHFFFAOYSA-N 0.000 description 1
- UDFARPRXWMDFQU-UHFFFAOYSA-N 2,6-ditert-butyl-4-[(3,5-ditert-butyl-4-hydroxyphenyl)methylsulfanylmethyl]phenol Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CSCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 UDFARPRXWMDFQU-UHFFFAOYSA-N 0.000 description 1
- QHPKIUDQDCWRKO-UHFFFAOYSA-N 2,6-ditert-butyl-4-[2-(3,5-ditert-butyl-4-hydroxyphenyl)propan-2-yl]phenol Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(C(C)(C)C=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 QHPKIUDQDCWRKO-UHFFFAOYSA-N 0.000 description 1
- JMCKNCBUBGMWAY-UHFFFAOYSA-N 2,6-ditert-butyl-4-[[4-(3,5-ditert-butyl-4-hydroxyphenoxy)-6-octylsulfanyl-1,3,5-triazin-2-yl]oxy]phenol Chemical compound N=1C(OC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=NC(SCCCCCCCC)=NC=1OC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 JMCKNCBUBGMWAY-UHFFFAOYSA-N 0.000 description 1
- RTOZVEXLKURGKW-UHFFFAOYSA-N 2,6-ditert-butyl-4-[[4-[(3,5-ditert-butyl-4-hydroxyphenyl)methyl]-2,3,5,6-tetramethylphenyl]methyl]phenol Chemical compound CC=1C(C)=C(CC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)C(C)=C(C)C=1CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 RTOZVEXLKURGKW-UHFFFAOYSA-N 0.000 description 1
- HWRLEEPNFJNTOP-UHFFFAOYSA-N 2-(1,3,5-triazin-2-yl)phenol Chemical class OC1=CC=CC=C1C1=NC=NC=N1 HWRLEEPNFJNTOP-UHFFFAOYSA-N 0.000 description 1
- QRBPVMCWQACMMY-UHFFFAOYSA-N 2-(2,2,6,6-tetramethyl-1-octoxypiperidin-3-yl)butanedioic acid Chemical compound CCCCCCCCON1C(C)(C)CCC(C(CC(O)=O)C(O)=O)C1(C)C QRBPVMCWQACMMY-UHFFFAOYSA-N 0.000 description 1
- DEHILEUXPOWXIS-UHFFFAOYSA-N 2-(2,5-ditert-butyl-4-hydroxyphenyl)propan-2-ylphosphonic acid Chemical compound CC(C)(C)C1=CC(C(C)(C)P(O)(O)=O)=C(C(C)(C)C)C=C1O DEHILEUXPOWXIS-UHFFFAOYSA-N 0.000 description 1
- LBOGPIWNHXHYHN-UHFFFAOYSA-N 2-(2-hydroxy-5-octylphenyl)sulfanyl-4-octylphenol Chemical compound CCCCCCCCC1=CC=C(O)C(SC=2C(=CC=C(CCCCCCCC)C=2)O)=C1 LBOGPIWNHXHYHN-UHFFFAOYSA-N 0.000 description 1
- QLMGIWHWWWXXME-UHFFFAOYSA-N 2-(3,5-ditert-butyl-4-hydroxyphenyl)acetic acid Chemical compound CC(C)(C)C1=CC(CC(O)=O)=CC(C(C)(C)C)=C1O QLMGIWHWWWXXME-UHFFFAOYSA-N 0.000 description 1
- ZMWRRFHBXARRRT-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-4,6-bis(2-methylbutan-2-yl)phenol Chemical compound CCC(C)(C)C1=CC(C(C)(C)CC)=CC(N2N=C3C=CC=CC3=N2)=C1O ZMWRRFHBXARRRT-UHFFFAOYSA-N 0.000 description 1
- OLFNXLXEGXRUOI-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-4,6-bis(2-phenylpropan-2-yl)phenol Chemical compound C=1C(N2N=C3C=CC=CC3=N2)=C(O)C(C(C)(C)C=2C=CC=CC=2)=CC=1C(C)(C)C1=CC=CC=C1 OLFNXLXEGXRUOI-UHFFFAOYSA-N 0.000 description 1
- LHPPDQUVECZQSW-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-4,6-ditert-butylphenol Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=CC(N2N=C3C=CC=CC3=N2)=C1O LHPPDQUVECZQSW-UHFFFAOYSA-N 0.000 description 1
- WXHVQMGINBSVAY-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-4-tert-butylphenol Chemical compound CC(C)(C)C1=CC=C(O)C(N2N=C3C=CC=CC3=N2)=C1 WXHVQMGINBSVAY-UHFFFAOYSA-N 0.000 description 1
- ITLDHFORLZTRJI-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-5-octoxyphenol Chemical compound OC1=CC(OCCCCCCCC)=CC=C1N1N=C2C=CC=CC2=N1 ITLDHFORLZTRJI-UHFFFAOYSA-N 0.000 description 1
- RTNVDKBRTXEWQE-UHFFFAOYSA-N 2-(benzotriazol-2-yl)-6-butan-2-yl-4-tert-butylphenol Chemical compound CCC(C)C1=CC(C(C)(C)C)=CC(N2N=C3C=CC=CC3=N2)=C1O RTNVDKBRTXEWQE-UHFFFAOYSA-N 0.000 description 1
- XQESJWNDTICJHW-UHFFFAOYSA-N 2-[(2-hydroxy-5-methyl-3-nonylphenyl)methyl]-4-methyl-6-nonylphenol Chemical compound CCCCCCCCCC1=CC(C)=CC(CC=2C(=C(CCCCCCCCC)C=C(C)C=2)O)=C1O XQESJWNDTICJHW-UHFFFAOYSA-N 0.000 description 1
- YQQAAUCBTNZUQQ-UHFFFAOYSA-N 2-[1-(2-hydroxy-3,5-dimethylphenyl)butyl]-4,6-dimethylphenol Chemical compound C=1C(C)=CC(C)=C(O)C=1C(CCC)C1=CC(C)=CC(C)=C1O YQQAAUCBTNZUQQ-UHFFFAOYSA-N 0.000 description 1
- WQYFETFRIRDUPJ-UHFFFAOYSA-N 2-[2-hydroxy-5-(2,4,4-trimethylpentan-2-yl)phenyl]sulfanyl-4-(2,4,4-trimethylpentan-2-yl)phenol Chemical compound CC(C)(C)CC(C)(C)C1=CC=C(O)C(SC=2C(=CC=C(C=2)C(C)(C)CC(C)(C)C)O)=C1 WQYFETFRIRDUPJ-UHFFFAOYSA-N 0.000 description 1
- DVIFVFGPATZWCB-UHFFFAOYSA-N 2-[4,4-bis(1,2,2,6,6-pentamethylpiperidin-3-yl)butyl]-2-[(3,5-ditert-butyl-4-hydroxyphenyl)methyl]propanedioic acid Chemical compound CC1(C)N(C)C(C)(C)CCC1C(C1C(N(C)C(C)(C)CC1)(C)C)CCCC(C(O)=O)(C(O)=O)CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 DVIFVFGPATZWCB-UHFFFAOYSA-N 0.000 description 1
- ZSSVCEUEVMALRD-UHFFFAOYSA-N 2-[4,6-bis(2,4-dimethylphenyl)-1,3,5-triazin-2-yl]-5-(octyloxy)phenol Chemical compound OC1=CC(OCCCCCCCC)=CC=C1C1=NC(C=2C(=CC(C)=CC=2)C)=NC(C=2C(=CC(C)=CC=2)C)=N1 ZSSVCEUEVMALRD-UHFFFAOYSA-N 0.000 description 1
- DBYBHKQEHCYBQV-UHFFFAOYSA-N 2-[4,6-bis(2,4-dimethylphenyl)-1,3,5-triazin-2-yl]-5-dodecoxyphenol Chemical compound OC1=CC(OCCCCCCCCCCCC)=CC=C1C1=NC(C=2C(=CC(C)=CC=2)C)=NC(C=2C(=CC(C)=CC=2)C)=N1 DBYBHKQEHCYBQV-UHFFFAOYSA-N 0.000 description 1
- WPMUMRCRKFBYIH-UHFFFAOYSA-N 2-[4,6-bis(2-hydroxy-4-octoxyphenyl)-1,3,5-triazin-2-yl]-5-octoxyphenol Chemical compound OC1=CC(OCCCCCCCC)=CC=C1C1=NC(C=2C(=CC(OCCCCCCCC)=CC=2)O)=NC(C=2C(=CC(OCCCCCCCC)=CC=2)O)=N1 WPMUMRCRKFBYIH-UHFFFAOYSA-N 0.000 description 1
- HHIVRACNDKRDTF-UHFFFAOYSA-N 2-[4-(2,4-dimethylphenyl)-6-(2-hydroxy-4-propoxyphenyl)-1,3,5-triazin-2-yl]-5-propoxyphenol Chemical compound OC1=CC(OCCC)=CC=C1C1=NC(C=2C(=CC(C)=CC=2)C)=NC(C=2C(=CC(OCCC)=CC=2)O)=N1 HHIVRACNDKRDTF-UHFFFAOYSA-N 0.000 description 1
- RDDIIAYGQFICIL-UHFFFAOYSA-N 2-[4-(5,7-ditert-butyl-2-oxo-3h-1-benzofuran-3-yl)phenoxy]ethyl acetate Chemical compound C1=CC(OCCOC(=O)C)=CC=C1C1C(C=C(C=C2C(C)(C)C)C(C)(C)C)=C2OC1=O RDDIIAYGQFICIL-UHFFFAOYSA-N 0.000 description 1
- YJWCUAHFSOAUKV-UHFFFAOYSA-N 2-[4-(5,7-ditert-butyl-2-oxo-3h-1-benzofuran-3-yl)phenoxy]ethyl octadecanoate Chemical compound C1=CC(OCCOC(=O)CCCCCCCCCCCCCCCCC)=CC=C1C1C(C=C(C=C2C(C)(C)C)C(C)(C)C)=C2OC1=O YJWCUAHFSOAUKV-UHFFFAOYSA-N 0.000 description 1
- XUQNLOIVFHUMTR-UHFFFAOYSA-N 2-[[2-hydroxy-5-nonyl-3-(1-phenylethyl)phenyl]methyl]-4-nonyl-6-(1-phenylethyl)phenol Chemical compound OC=1C(C(C)C=2C=CC=CC=2)=CC(CCCCCCCCC)=CC=1CC(C=1O)=CC(CCCCCCCCC)=CC=1C(C)C1=CC=CC=C1 XUQNLOIVFHUMTR-UHFFFAOYSA-N 0.000 description 1
- HHPDFYDITNAMAM-UHFFFAOYSA-N 2-[cyclohexyl(2-hydroxyethyl)amino]ethanol Chemical compound OCCN(CCO)C1CCCCC1 HHPDFYDITNAMAM-UHFFFAOYSA-N 0.000 description 1
- LUPQXHNQUVRJFK-UHFFFAOYSA-N 2-butyl-2-[(3,5-ditert-butyl-2-hydroxyphenyl)-bis(1,2,2,6,6-pentamethylpiperidin-3-yl)methyl]propanedioic acid Chemical compound CCCCC(C(=O)O)(C(=O)O)C(C1CCC(N(C1(C)C)C)(C)C)(C2CCC(N(C2(C)C)C)(C)C)C3=CC(=CC(=C3O)C(C)(C)C)C(C)(C)C LUPQXHNQUVRJFK-UHFFFAOYSA-N 0.000 description 1
- AKNMPWVTPUHKCG-UHFFFAOYSA-N 2-cyclohexyl-6-[(3-cyclohexyl-2-hydroxy-5-methylphenyl)methyl]-4-methylphenol Chemical compound OC=1C(C2CCCCC2)=CC(C)=CC=1CC(C=1O)=CC(C)=CC=1C1CCCCC1 AKNMPWVTPUHKCG-UHFFFAOYSA-N 0.000 description 1
- NCWTZPKMFNRUAK-UHFFFAOYSA-N 2-ethyl-4,6-bis(octylsulfanylmethyl)phenol Chemical compound CCCCCCCCSCC1=CC(CC)=C(O)C(CSCCCCCCCC)=C1 NCWTZPKMFNRUAK-UHFFFAOYSA-N 0.000 description 1
- LHHLLQVLJAUUDT-UHFFFAOYSA-N 2-ethylhexyl 3-[3-(benzotriazol-2-yl)-5-tert-butyl-4-hydroxyphenyl]propanoate Chemical compound CC(C)(C)C1=CC(CCC(=O)OCC(CC)CCCC)=CC(N2N=C3C=CC=CC3=N2)=C1O LHHLLQVLJAUUDT-UHFFFAOYSA-N 0.000 description 1
- AWEVLIFGIMIQHY-UHFFFAOYSA-N 2-ethylhexyl 3-[3-tert-butyl-5-(5-chlorobenzotriazol-2-yl)-4-hydroxyphenyl]propanoate Chemical compound CC(C)(C)C1=CC(CCC(=O)OCC(CC)CCCC)=CC(N2N=C3C=C(Cl)C=CC3=N2)=C1O AWEVLIFGIMIQHY-UHFFFAOYSA-N 0.000 description 1
- UORSDGBOJHYJLV-UHFFFAOYSA-N 2-hydroxy-n'-(2-hydroxybenzoyl)benzohydrazide Chemical compound OC1=CC=CC=C1C(=O)NNC(=O)C1=CC=CC=C1O UORSDGBOJHYJLV-UHFFFAOYSA-N 0.000 description 1
- MZZYGYNZAOVRTG-UHFFFAOYSA-N 2-hydroxy-n-(1h-1,2,4-triazol-5-yl)benzamide Chemical compound OC1=CC=CC=C1C(=O)NC1=NC=NN1 MZZYGYNZAOVRTG-UHFFFAOYSA-N 0.000 description 1
- WBJWXIQDBDZMAW-UHFFFAOYSA-N 2-hydroxynaphthalene-1-carbonyl chloride Chemical compound C1=CC=CC2=C(C(Cl)=O)C(O)=CC=C21 WBJWXIQDBDZMAW-UHFFFAOYSA-N 0.000 description 1
- GAODDBNJCKQQDY-UHFFFAOYSA-N 2-methyl-4,6-bis(octylsulfanylmethyl)phenol Chemical compound CCCCCCCCSCC1=CC(C)=C(O)C(CSCCCCCCCC)=C1 GAODDBNJCKQQDY-UHFFFAOYSA-N 0.000 description 1
- VSKJLJHPAFKHBX-UHFFFAOYSA-N 2-methylbuta-1,3-diene;styrene Chemical compound CC(=C)C=C.C=CC1=CC=CC=C1.C=CC1=CC=CC=C1 VSKJLJHPAFKHBX-UHFFFAOYSA-N 0.000 description 1
- ZYJXQDCMXTWHIV-UHFFFAOYSA-N 2-tert-butyl-4,6-bis(octylsulfanylmethyl)phenol Chemical compound CCCCCCCCSCC1=CC(CSCCCCCCCC)=C(O)C(C(C)(C)C)=C1 ZYJXQDCMXTWHIV-UHFFFAOYSA-N 0.000 description 1
- YFHKLSPMRRWLKI-UHFFFAOYSA-N 2-tert-butyl-4-(3-tert-butyl-4-hydroxy-5-methylphenyl)sulfanyl-6-methylphenol Chemical compound CC(C)(C)C1=C(O)C(C)=CC(SC=2C=C(C(O)=C(C)C=2)C(C)(C)C)=C1 YFHKLSPMRRWLKI-UHFFFAOYSA-N 0.000 description 1
- HXIQYSLFEXIOAV-UHFFFAOYSA-N 2-tert-butyl-4-(5-tert-butyl-4-hydroxy-2-methylphenyl)sulfanyl-5-methylphenol Chemical compound CC1=CC(O)=C(C(C)(C)C)C=C1SC1=CC(C(C)(C)C)=C(O)C=C1C HXIQYSLFEXIOAV-UHFFFAOYSA-N 0.000 description 1
- RKLRVTKRKFEVQG-UHFFFAOYSA-N 2-tert-butyl-4-[(3-tert-butyl-4-hydroxy-5-methylphenyl)methyl]-6-methylphenol Chemical compound CC(C)(C)C1=C(O)C(C)=CC(CC=2C=C(C(O)=C(C)C=2)C(C)(C)C)=C1 RKLRVTKRKFEVQG-UHFFFAOYSA-N 0.000 description 1
- BCHSGIGAOYQMPC-UHFFFAOYSA-N 2-tert-butyl-4-[1-(5-tert-butyl-4-hydroxy-2-methylphenyl)-3-dodecylsulfanylbutyl]-5-methylphenol Chemical compound C=1C(C(C)(C)C)=C(O)C=C(C)C=1C(CC(C)SCCCCCCCCCCCC)C1=CC(C(C)(C)C)=C(O)C=C1C BCHSGIGAOYQMPC-UHFFFAOYSA-N 0.000 description 1
- PFANXOISJYKQRP-UHFFFAOYSA-N 2-tert-butyl-4-[1-(5-tert-butyl-4-hydroxy-2-methylphenyl)butyl]-5-methylphenol Chemical compound C=1C(C(C)(C)C)=C(O)C=C(C)C=1C(CCC)C1=CC(C(C)(C)C)=C(O)C=C1C PFANXOISJYKQRP-UHFFFAOYSA-N 0.000 description 1
- JJBOJSJSDIRUGY-UHFFFAOYSA-N 2-tert-butyl-4-[2-(5-tert-butyl-4-hydroxy-2-methylphenyl)-4-dodecylsulfanylbutan-2-yl]-5-methylphenol Chemical compound C=1C(C(C)(C)C)=C(O)C=C(C)C=1C(C)(CCSCCCCCCCCCCCC)C1=CC(C(C)(C)C)=C(O)C=C1C JJBOJSJSDIRUGY-UHFFFAOYSA-N 0.000 description 1
- XMUNJUUYEJAAHG-UHFFFAOYSA-N 2-tert-butyl-5-methyl-4-[1,5,5-tris(5-tert-butyl-4-hydroxy-2-methylphenyl)pentyl]phenol Chemical compound CC1=CC(O)=C(C(C)(C)C)C=C1C(C=1C(=CC(O)=C(C=1)C(C)(C)C)C)CCCC(C=1C(=CC(O)=C(C=1)C(C)(C)C)C)C1=CC(C(C)(C)C)=C(O)C=C1C XMUNJUUYEJAAHG-UHFFFAOYSA-N 0.000 description 1
- MQWCQFCZUNBTCM-UHFFFAOYSA-N 2-tert-butyl-6-(3-tert-butyl-2-hydroxy-5-methylphenyl)sulfanyl-4-methylphenol Chemical compound CC(C)(C)C1=CC(C)=CC(SC=2C(=C(C=C(C)C=2)C(C)(C)C)O)=C1O MQWCQFCZUNBTCM-UHFFFAOYSA-N 0.000 description 1
- GPNYZBKIGXGYNU-UHFFFAOYSA-N 2-tert-butyl-6-[(3-tert-butyl-5-ethyl-2-hydroxyphenyl)methyl]-4-ethylphenol Chemical compound CC(C)(C)C1=CC(CC)=CC(CC=2C(=C(C=C(CC)C=2)C(C)(C)C)O)=C1O GPNYZBKIGXGYNU-UHFFFAOYSA-N 0.000 description 1
- UHLYPUYAVHSKBN-UHFFFAOYSA-N 2-tert-butyl-6-[1-[3-tert-butyl-2-hydroxy-5-(2-methylpropyl)phenyl]ethyl]-4-(2-methylpropyl)phenol Chemical compound CC(C)(C)C1=CC(CC(C)C)=CC(C(C)C=2C(=C(C=C(CC(C)C)C=2)C(C)(C)C)O)=C1O UHLYPUYAVHSKBN-UHFFFAOYSA-N 0.000 description 1
- HNTWDNMNGNBFEA-UHFFFAOYSA-N 3,3,5,5-tetramethylpiperazin-2-one Chemical compound CC1(C)CNC(=O)C(C)(C)N1 HNTWDNMNGNBFEA-UHFFFAOYSA-N 0.000 description 1
- BVGUNYMSTQHLIC-UHFFFAOYSA-N 3,5-ditert-butyl-2-(2,4-ditert-butyl-6-methylphenyl)-4-hydroxybenzoic acid Chemical compound CC1=CC(C(C)(C)C)=CC(C(C)(C)C)=C1C1=C(C(O)=O)C=C(C(C)(C)C)C(O)=C1C(C)(C)C BVGUNYMSTQHLIC-UHFFFAOYSA-N 0.000 description 1
- NMAGCVWUISAHAP-UHFFFAOYSA-N 3,5-ditert-butyl-2-(2,4-ditert-butylphenyl)-4-hydroxybenzoic acid Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=CC=C1C1=C(C(O)=O)C=C(C(C)(C)C)C(O)=C1C(C)(C)C NMAGCVWUISAHAP-UHFFFAOYSA-N 0.000 description 1
- YLUZWKKWWSCRSR-UHFFFAOYSA-N 3,9-bis(8-methylnonoxy)-2,4,8,10-tetraoxa-3,9-diphosphaspiro[5.5]undecane Chemical compound C1OP(OCCCCCCCC(C)C)OCC21COP(OCCCCCCCC(C)C)OC2 YLUZWKKWWSCRSR-UHFFFAOYSA-N 0.000 description 1
- PZRWFKGUFWPFID-UHFFFAOYSA-N 3,9-dioctadecoxy-2,4,8,10-tetraoxa-3,9-diphosphaspiro[5.5]undecane Chemical compound C1OP(OCCCCCCCCCCCCCCCCCC)OCC21COP(OCCCCCCCCCCCCCCCCCC)OC2 PZRWFKGUFWPFID-UHFFFAOYSA-N 0.000 description 1
- DBHUTHZPCWZNRW-UHFFFAOYSA-N 3-(3,5-dicyclohexyl-4-hydroxyphenyl)propanoic acid Chemical compound OC=1C(C2CCCCC2)=CC(CCC(=O)O)=CC=1C1CCCCC1 DBHUTHZPCWZNRW-UHFFFAOYSA-N 0.000 description 1
- KJEKRODBOPOEGG-UHFFFAOYSA-N 3-(3,5-ditert-butyl-4-hydroxyphenyl)-n-[3-[3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoylamino]propyl]propanamide Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CCC(=O)NCCCNC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 KJEKRODBOPOEGG-UHFFFAOYSA-N 0.000 description 1
- FLZYQMOKBVFXJS-UHFFFAOYSA-N 3-(3-tert-butyl-4-hydroxy-5-methylphenyl)propanoic acid Chemical compound CC1=CC(CCC(O)=O)=CC(C(C)(C)C)=C1O FLZYQMOKBVFXJS-UHFFFAOYSA-N 0.000 description 1
- YCGKJPVUGMBDDS-UHFFFAOYSA-N 3-(6-azabicyclo[3.1.1]hepta-1(7),2,4-triene-6-carbonyl)benzamide Chemical compound NC(=O)C1=CC=CC(C(=O)N2C=3C=C2C=CC=3)=C1 YCGKJPVUGMBDDS-UHFFFAOYSA-N 0.000 description 1
- SAEZGDDJKSBNPT-UHFFFAOYSA-N 3-dodecyl-1-(1,2,2,6,6-pentamethylpiperidin-4-yl)pyrrolidine-2,5-dione Chemical compound O=C1C(CCCCCCCCCCCC)CC(=O)N1C1CC(C)(C)N(C)C(C)(C)C1 SAEZGDDJKSBNPT-UHFFFAOYSA-N 0.000 description 1
- FBIXXCXCZOZFCO-UHFFFAOYSA-N 3-dodecyl-1-(2,2,6,6-tetramethylpiperidin-4-yl)pyrrolidine-2,5-dione Chemical compound O=C1C(CCCCCCCCCCCC)CC(=O)N1C1CC(C)(C)NC(C)(C)C1 FBIXXCXCZOZFCO-UHFFFAOYSA-N 0.000 description 1
- CIFFBTOJCKSRJY-UHFFFAOYSA-N 3α,4,7,7α-tetrahydro-1h-isoindole-1,3(2h)-dione Chemical compound C1C=CCC2C(=O)NC(=O)C21 CIFFBTOJCKSRJY-UHFFFAOYSA-N 0.000 description 1
- MDWVSAYEQPLWMX-UHFFFAOYSA-N 4,4'-Methylenebis(2,6-di-tert-butylphenol) Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 MDWVSAYEQPLWMX-UHFFFAOYSA-N 0.000 description 1
- HPFWYRKGZUGGPB-UHFFFAOYSA-N 4,6-dichloro-n-(2,4,4-trimethylpentan-2-yl)-1,3,5-triazin-2-amine Chemical compound CC(C)(C)CC(C)(C)NC1=NC(Cl)=NC(Cl)=N1 HPFWYRKGZUGGPB-UHFFFAOYSA-N 0.000 description 1
- VGEJJASMUCILJT-UHFFFAOYSA-N 4-[2-[4,6-bis[2-(3,5-ditert-butyl-4-hydroxyphenyl)ethyl]-1,3,5-triazin-2-yl]ethyl]-2,6-ditert-butylphenol Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CCC=2N=C(CCC=3C=C(C(O)=C(C=3)C(C)(C)C)C(C)(C)C)N=C(CCC=3C=C(C(O)=C(C=3)C(C)(C)C)C(C)(C)C)N=2)=C1 VGEJJASMUCILJT-UHFFFAOYSA-N 0.000 description 1
- PRWJPWSKLXYEPD-UHFFFAOYSA-N 4-[4,4-bis(5-tert-butyl-4-hydroxy-2-methylphenyl)butan-2-yl]-2-tert-butyl-5-methylphenol Chemical compound C=1C(C(C)(C)C)=C(O)C=C(C)C=1C(C)CC(C=1C(=CC(O)=C(C=1)C(C)(C)C)C)C1=CC(C(C)(C)C)=C(O)C=C1C PRWJPWSKLXYEPD-UHFFFAOYSA-N 0.000 description 1
- FROCQMFXPIROOK-UHFFFAOYSA-N 4-[4,6-bis(2,4-dimethylphenyl)-1,3,5-triazin-2-yl]benzene-1,3-diol Chemical compound CC1=CC(C)=CC=C1C1=NC(C=2C(=CC(C)=CC=2)C)=NC(C=2C(=CC(O)=CC=2)O)=N1 FROCQMFXPIROOK-UHFFFAOYSA-N 0.000 description 1
- QVXGXGJJEDTQSU-UHFFFAOYSA-N 4-[4-hydroxy-2,5-di(pentan-2-yl)phenyl]sulfanyl-2,5-di(pentan-2-yl)phenol Chemical compound C1=C(O)C(C(C)CCC)=CC(SC=2C(=CC(O)=C(C(C)CCC)C=2)C(C)CCC)=C1C(C)CCC QVXGXGJJEDTQSU-UHFFFAOYSA-N 0.000 description 1
- IYUSCCOBICHICG-UHFFFAOYSA-N 4-[[2,4-bis(3,5-ditert-butyl-4-hydroxyphenoxy)-1h-triazin-6-yl]oxy]-2,6-ditert-butylphenol Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(ON2N=C(OC=3C=C(C(O)=C(C=3)C(C)(C)C)C(C)(C)C)C=C(OC=3C=C(C(O)=C(C=3)C(C)(C)C)C(C)(C)C)N2)=C1 IYUSCCOBICHICG-UHFFFAOYSA-N 0.000 description 1
- VSAWBBYYMBQKIK-UHFFFAOYSA-N 4-[[3,5-bis[(3,5-ditert-butyl-4-hydroxyphenyl)methyl]-2,4,6-trimethylphenyl]methyl]-2,6-ditert-butylphenol Chemical compound CC1=C(CC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)C(C)=C(CC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)C(C)=C1CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 VSAWBBYYMBQKIK-UHFFFAOYSA-N 0.000 description 1
- BOQNWBDBDUWBMT-UHFFFAOYSA-N 4-[[bis[(3,5-ditert-butyl-4-hydroxyphenyl)methyl]amino]methyl]-2,6-ditert-butylphenol Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CN(CC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)CC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 BOQNWBDBDUWBMT-UHFFFAOYSA-N 0.000 description 1
- WTWGHNZAQVTLSQ-UHFFFAOYSA-N 4-butyl-2,6-ditert-butylphenol Chemical compound CCCCC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 WTWGHNZAQVTLSQ-UHFFFAOYSA-N 0.000 description 1
- OVARTXYXUGDZHU-UHFFFAOYSA-N 4-hydroxy-n-phenyldodecanamide Chemical compound CCCCCCCCC(O)CCC(=O)NC1=CC=CC=C1 OVARTXYXUGDZHU-UHFFFAOYSA-N 0.000 description 1
- WSSSPWUEQFSQQG-UHFFFAOYSA-N 4-methyl-1-pentene Chemical compound CC(C)CC=C WSSSPWUEQFSQQG-UHFFFAOYSA-N 0.000 description 1
- UXMKUNDWNZNECH-UHFFFAOYSA-N 4-methyl-2,6-di(nonyl)phenol Chemical compound CCCCCCCCCC1=CC(C)=CC(CCCCCCCCC)=C1O UXMKUNDWNZNECH-UHFFFAOYSA-N 0.000 description 1
- LZAIWKMQABZIDI-UHFFFAOYSA-N 4-methyl-2,6-dioctadecylphenol Chemical compound CCCCCCCCCCCCCCCCCCC1=CC(C)=CC(CCCCCCCCCCCCCCCCCC)=C1O LZAIWKMQABZIDI-UHFFFAOYSA-N 0.000 description 1
- JJHKARPEMHIIQC-UHFFFAOYSA-N 4-octadecoxy-2,6-diphenylphenol Chemical compound C=1C(OCCCCCCCCCCCCCCCCCC)=CC(C=2C=CC=CC=2)=C(O)C=1C1=CC=CC=C1 JJHKARPEMHIIQC-UHFFFAOYSA-N 0.000 description 1
- KDVYCTOWXSLNNI-UHFFFAOYSA-N 4-t-Butylbenzoic acid Chemical compound CC(C)(C)C1=CC=C(C(O)=O)C=C1 KDVYCTOWXSLNNI-UHFFFAOYSA-N 0.000 description 1
- DBOSBRHMHBENLP-UHFFFAOYSA-N 4-tert-Butylphenyl Salicylate Chemical compound C1=CC(C(C)(C)C)=CC=C1OC(=O)C1=CC=CC=C1O DBOSBRHMHBENLP-UHFFFAOYSA-N 0.000 description 1
- CXUYVOHLQSZWCW-UHFFFAOYSA-N 5,7-ditert-butyl-3-(4-ethoxyphenyl)-3h-1-benzofuran-2-one Chemical compound C1=CC(OCC)=CC=C1C1C(C=C(C=C2C(C)(C)C)C(C)(C)C)=C2OC1=O CXUYVOHLQSZWCW-UHFFFAOYSA-N 0.000 description 1
- UWSMKYBKUPAEJQ-UHFFFAOYSA-N 5-Chloro-2-(3,5-di-tert-butyl-2-hydroxyphenyl)-2H-benzotriazole Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=CC(N2N=C3C=C(Cl)C=CC3=N2)=C1O UWSMKYBKUPAEJQ-UHFFFAOYSA-N 0.000 description 1
- XUEVNEQLDIFVAO-UHFFFAOYSA-N 6-methylheptyl 2-[(3,5-ditert-butyl-4-hydroxyphenyl)methylsulfanyl]acetate Chemical compound CC(C)CCCCCOC(=O)CSCC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 XUEVNEQLDIFVAO-UHFFFAOYSA-N 0.000 description 1
- VPOKLVDHXARWQB-UHFFFAOYSA-N 7,7,9,9-tetramethyl-3-octyl-1,3,8-triazaspiro[4.5]decane-2,4-dione Chemical compound O=C1N(CCCCCCCC)C(=O)NC11CC(C)(C)NC(C)(C)C1 VPOKLVDHXARWQB-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- OGBVRMYSNSKIEF-UHFFFAOYSA-N Benzylphosphonic acid Chemical class OP(O)(=O)CC1=CC=CC=C1 OGBVRMYSNSKIEF-UHFFFAOYSA-N 0.000 description 1
- 239000004604 Blowing Agent Substances 0.000 description 1
- NLZUEZXRPGMBCV-UHFFFAOYSA-N Butylhydroxytoluene Chemical compound CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NLZUEZXRPGMBCV-UHFFFAOYSA-N 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- IOBYPSNWPGHFNA-UHFFFAOYSA-N CC1(NC(CC(C1)OC(=O)C(C)(C(CC(=O)OC1CC(NC(C1)(C)C)(C)C)C(=O)OC1CC(NC(C1)(C)C)(C)C)C(=O)OC1CC(NC(C1)(C)C)(C)C)(C)C)C Chemical compound CC1(NC(CC(C1)OC(=O)C(C)(C(CC(=O)OC1CC(NC(C1)(C)C)(C)C)C(=O)OC1CC(NC(C1)(C)C)(C)C)C(=O)OC1CC(NC(C1)(C)C)(C)C)(C)C)C IOBYPSNWPGHFNA-UHFFFAOYSA-N 0.000 description 1
- DJRCMVCFUPOPKC-UHFFFAOYSA-N CCCCCCCCCCCC(=NO)C1=CC=C(C)C=C1O Chemical compound CCCCCCCCCCCC(=NO)C1=CC=C(C)C=C1O DJRCMVCFUPOPKC-UHFFFAOYSA-N 0.000 description 1
- AUYGZHLYMDQOIG-UHFFFAOYSA-N CCCCCCCCCCCC[S+](CCCCCCCCCCCC)CCOC(C(CC(C=C1C(C)(C)C)=CC(C(C)(C)C)=C1O)(CC(C=C1C(C)(C)C)=CC(C(C)(C)C)=C1O)C([O-])=O)=O Chemical compound CCCCCCCCCCCC[S+](CCCCCCCCCCCC)CCOC(C(CC(C=C1C(C)(C)C)=CC(C(C)(C)C)=C1O)(CC(C=C1C(C)(C)C)=CC(C(C)(C)C)=C1O)C([O-])=O)=O AUYGZHLYMDQOIG-UHFFFAOYSA-N 0.000 description 1
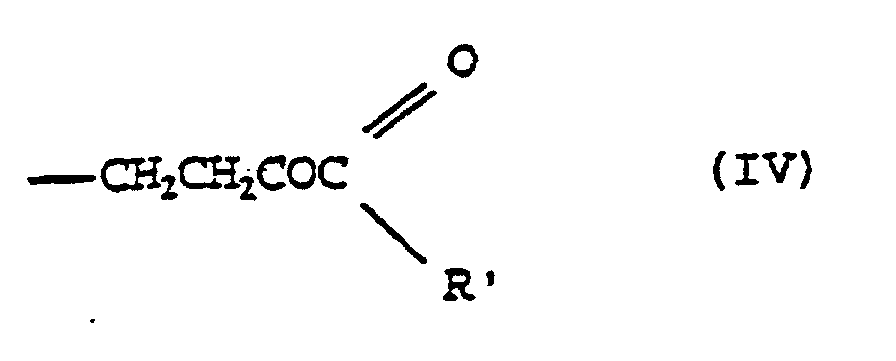
- 0 CI(*)C(C(*(*)C1=O)O)(*1N)*=* Chemical compound CI(*)C(C(*(*)C1=O)O)(*1N)*=* 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-N Carbamic acid Chemical class NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 description 1
- 229920001634 Copolyester Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- GZIFEOYASATJEH-UHFFFAOYSA-N D-delta tocopherol Natural products OC1=CC(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1 GZIFEOYASATJEH-UHFFFAOYSA-N 0.000 description 1
- GHKOFFNLGXMVNJ-UHFFFAOYSA-N Didodecyl thiobispropanoate Chemical compound CCCCCCCCCCCCOC(=O)CCSCCC(=O)OCCCCCCCCCCCC GHKOFFNLGXMVNJ-UHFFFAOYSA-N 0.000 description 1
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 1
- 239000003508 Dilauryl thiodipropionate Substances 0.000 description 1
- 239000002656 Distearyl thiodipropionate Substances 0.000 description 1
- BRLQWZUYTZBJKN-UHFFFAOYSA-N Epichlorohydrin Chemical compound ClCC1CO1 BRLQWZUYTZBJKN-UHFFFAOYSA-N 0.000 description 1
- 239000004593 Epoxy Chemical class 0.000 description 1
- 239000004262 Ethyl gallate Substances 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 239000004716 Ethylene/acrylic acid copolymer Substances 0.000 description 1
- 239000001828 Gelatine Substances 0.000 description 1
- OKOBUGCCXMIKDM-UHFFFAOYSA-N Irganox 1098 Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CCC(=O)NCCCCCCNC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 OKOBUGCCXMIKDM-UHFFFAOYSA-N 0.000 description 1
- VQTUBCCKSQIDNK-UHFFFAOYSA-N Isobutene Chemical group CC(C)=C VQTUBCCKSQIDNK-UHFFFAOYSA-N 0.000 description 1
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Natural products CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 1
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical class [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 1
- 239000004594 Masterbatch (MB) Substances 0.000 description 1
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 1
- GYCMBHHDWRMZGG-UHFFFAOYSA-N Methylacrylonitrile Chemical compound CC(=C)C#N GYCMBHHDWRMZGG-UHFFFAOYSA-N 0.000 description 1
- 229920000571 Nylon 11 Polymers 0.000 description 1
- 229920000299 Nylon 12 Polymers 0.000 description 1
- 229920001007 Nylon 4 Polymers 0.000 description 1
- 229920002302 Nylon 6,6 Polymers 0.000 description 1
- IWGBGUINMSOOKP-UHFFFAOYSA-N OC1=C(C(=O)CCCCCCCCCCC)C=NN1C1=CC=CC=C1 Chemical compound OC1=C(C(=O)CCCCCCCCCCC)C=NN1C1=CC=CC=C1 IWGBGUINMSOOKP-UHFFFAOYSA-N 0.000 description 1
- 229920007019 PC/ABS Polymers 0.000 description 1
- 229920006778 PC/PBT Polymers 0.000 description 1
- 239000002033 PVDF binder Substances 0.000 description 1
- JKIJEFPNVSHHEI-UHFFFAOYSA-N Phenol, 2,4-bis(1,1-dimethylethyl)-, phosphite (3:1) Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=CC=C1OP(OC=1C(=CC(=CC=1)C(C)(C)C)C(C)(C)C)OC1=CC=C(C(C)(C)C)C=C1C(C)(C)C JKIJEFPNVSHHEI-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical class OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 229920003006 Polybutadiene acrylonitrile Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- 229920001328 Polyvinylidene chloride Polymers 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-N Propionic acid Chemical class CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 239000004433 Thermoplastic polyurethane Substances 0.000 description 1
- 239000003490 Thiodipropionic acid Substances 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 239000004904 UV filter Substances 0.000 description 1
- 239000012963 UV stabilizer Substances 0.000 description 1
- 229920001807 Urea-formaldehyde Polymers 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- FDLQZKYLHJJBHD-UHFFFAOYSA-N [3-(aminomethyl)phenyl]methanamine Chemical compound NCC1=CC=CC(CN)=C1 FDLQZKYLHJJBHD-UHFFFAOYSA-N 0.000 description 1
- OLXVZPUAKFOWDD-UHFFFAOYSA-N [3-hydroxy-2,2-bis(hydroxymethyl)propyl] 2,2,3,3-tetrakis(dodecan-2-ylsulfanyl)propanoate;1-(octadecyldisulfanyl)octadecane Chemical compound CCCCCCCCCCCCCCCCCCSSCCCCCCCCCCCCCCCCCC.CCCCCCCCCCC(C)SC(SC(C)CCCCCCCCCC)C(SC(C)CCCCCCCCCC)(SC(C)CCCCCCCCCC)C(=O)OCC(CO)(CO)CO OLXVZPUAKFOWDD-UHFFFAOYSA-N 0.000 description 1
- ZSFWKZUHCKWKGK-UHFFFAOYSA-N [4-(5,7-ditert-butyl-2-oxo-3h-1-benzofuran-3-yl)-2,6-dimethylphenyl] 2,2-dimethylpropanoate Chemical compound CC1=C(OC(=O)C(C)(C)C)C(C)=CC(C2C3=C(C(=CC(=C3)C(C)(C)C)C(C)(C)C)OC2=O)=C1 ZSFWKZUHCKWKGK-UHFFFAOYSA-N 0.000 description 1
- FXOMJIWKCOCWOU-UHFFFAOYSA-N [4-(5,7-ditert-butyl-2-oxo-3h-1-benzofuran-3-yl)-2,6-dimethylphenyl] acetate Chemical compound C1=C(C)C(OC(=O)C)=C(C)C=C1C1C(C=C(C=C2C(C)(C)C)C(C)(C)C)=C2OC1=O FXOMJIWKCOCWOU-UHFFFAOYSA-N 0.000 description 1
- YIMQCDZDWXUDCA-UHFFFAOYSA-N [4-(hydroxymethyl)cyclohexyl]methanol Chemical compound OCC1CCC(CO)CC1 YIMQCDZDWXUDCA-UHFFFAOYSA-N 0.000 description 1
- HHFMFWAFQGUGOB-UHFFFAOYSA-N [5-(4-tert-butylbenzoyl)-2,4-dihydroxyphenyl]-(4-tert-butylphenyl)methanone Chemical compound C1=CC(C(C)(C)C)=CC=C1C(=O)C1=CC(C(=O)C=2C=CC(=CC=2)C(C)(C)C)=C(O)C=C1O HHFMFWAFQGUGOB-UHFFFAOYSA-N 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- DHKHKXVYLBGOIT-UHFFFAOYSA-N acetaldehyde Diethyl Acetal Chemical class CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 1
- 150000001241 acetals Chemical class 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 150000001336 alkenes Chemical class 0.000 description 1
- 229940087168 alpha tocopherol Drugs 0.000 description 1
- PYHXGXCGESYPCW-UHFFFAOYSA-N alpha-phenylbenzeneacetic acid Natural products C=1C=CC=CC=1C(C(=O)O)C1=CC=CC=C1 PYHXGXCGESYPCW-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 150000003931 anilides Chemical class 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000002216 antistatic agent Substances 0.000 description 1
- 239000004760 aramid Substances 0.000 description 1
- 229920003235 aromatic polyamide Polymers 0.000 description 1
- 239000010425 asbestos Substances 0.000 description 1
- 229940116224 behenate Drugs 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 150000001559 benzoic acids Chemical class 0.000 description 1
- 229940066595 beta tocopherol Drugs 0.000 description 1
- 230000008033 biological extinction Effects 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- SMISHRXKWQZCCQ-UHFFFAOYSA-N bis(1,2,2,6,6-pentamethylpiperidin-3-yl) decanedioate Chemical compound CC1(C)N(C)C(C)(C)CCC1OC(=O)CCCCCCCCC(=O)OC1C(C)(C)N(C)C(C)(C)CC1 SMISHRXKWQZCCQ-UHFFFAOYSA-N 0.000 description 1
- YWDBZVIHZORXHG-UHFFFAOYSA-N bis(2,2,6,6-tetramethylpiperidin-1-yl) decanedioate Chemical compound CC1(C)CCCC(C)(C)N1OC(=O)CCCCCCCCC(=O)ON1C(C)(C)CCCC1(C)C YWDBZVIHZORXHG-UHFFFAOYSA-N 0.000 description 1
- YTKWTCYBDMELQK-UHFFFAOYSA-N bis(2,4-ditert-butyl-6-methylphenyl)methyl dihydrogen phosphite Chemical compound CC1=CC(C(C)(C)C)=CC(C(C)(C)C)=C1C(OP(O)O)C1=C(C)C=C(C(C)(C)C)C=C1C(C)(C)C YTKWTCYBDMELQK-UHFFFAOYSA-N 0.000 description 1
- OJZRGIRJHDINMJ-UHFFFAOYSA-N bis(3,5-ditert-butyl-4-hydroxyphenyl) hexanedioate Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(OC(=O)CCCCC(=O)OC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 OJZRGIRJHDINMJ-UHFFFAOYSA-N 0.000 description 1
- JTWMYTDTAUIDCU-UHFFFAOYSA-N bis[4-(2,4,4-trimethylpentan-2-yl)phenyl] 2,2-bis[(3,5-ditert-butyl-4-hydroxyphenyl)methyl]propanedioate Chemical compound C1=CC(C(C)(C)CC(C)(C)C)=CC=C1OC(=O)C(C(=O)OC=1C=CC(=CC=1)C(C)(C)CC(C)(C)C)(CC=1C=C(C(O)=C(C=1)C(C)(C)C)C(C)(C)C)CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 JTWMYTDTAUIDCU-UHFFFAOYSA-N 0.000 description 1
- 229920000402 bisphenol A polycarbonate polymer Polymers 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 230000001680 brushing effect Effects 0.000 description 1
- OCWYEMOEOGEQAN-UHFFFAOYSA-N bumetrizole Chemical compound CC(C)(C)C1=CC(C)=CC(N2N=C3C=C(Cl)C=CC3=N2)=C1O OCWYEMOEOGEQAN-UHFFFAOYSA-N 0.000 description 1
- HQABUPZFAYXKJW-UHFFFAOYSA-N butan-1-amine Chemical compound CCCCN HQABUPZFAYXKJW-UHFFFAOYSA-N 0.000 description 1
- 150000004648 butanoic acid derivatives Chemical class 0.000 description 1
- CQEYYJKEWSMYFG-UHFFFAOYSA-N butyl acrylate Chemical group CCCCOC(=O)C=C CQEYYJKEWSMYFG-UHFFFAOYSA-N 0.000 description 1
- QHIWVLPBUQWDMQ-UHFFFAOYSA-N butyl prop-2-enoate;methyl 2-methylprop-2-enoate;prop-2-enoic acid Chemical compound OC(=O)C=C.COC(=O)C(C)=C.CCCCOC(=O)C=C QHIWVLPBUQWDMQ-UHFFFAOYSA-N 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical class OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 229920001727 cellulose butyrate Polymers 0.000 description 1
- 229920003086 cellulose ether Polymers 0.000 description 1
- 229920006218 cellulose propionate Polymers 0.000 description 1
- 239000008199 coating composition Substances 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 150000001879 copper Chemical class 0.000 description 1
- 229920006037 cross link polymer Polymers 0.000 description 1
- 150000004292 cyclic ethers Chemical class 0.000 description 1
- 150000001925 cycloalkenes Chemical class 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 235000010389 delta-tocopherol Nutrition 0.000 description 1
- 230000002542 deteriorative effect Effects 0.000 description 1
- 150000004985 diamines Chemical class 0.000 description 1
- QGBSISYHAICWAH-UHFFFAOYSA-N dicyandiamide Chemical compound NC(N)=NC#N QGBSISYHAICWAH-UHFFFAOYSA-N 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- ZJIPHXXDPROMEF-UHFFFAOYSA-N dihydroxyphosphanyl dihydrogen phosphite Chemical compound OP(O)OP(O)O ZJIPHXXDPROMEF-UHFFFAOYSA-N 0.000 description 1
- 235000019304 dilauryl thiodipropionate Nutrition 0.000 description 1
- VNSRQBDLLINZJV-UHFFFAOYSA-N dioctadecyl 2,2-bis[(3,5-ditert-butyl-2-hydroxyphenyl)methyl]propanedioate Chemical compound C=1C(C(C)(C)C)=CC(C(C)(C)C)=C(O)C=1CC(C(=O)OCCCCCCCCCCCCCCCCCC)(C(=O)OCCCCCCCCCCCCCCCCCC)CC1=CC(C(C)(C)C)=CC(C(C)(C)C)=C1O VNSRQBDLLINZJV-UHFFFAOYSA-N 0.000 description 1
- KQEPQKRGTBAQRR-UHFFFAOYSA-N dioctadecyl 2-[(3-tert-butyl-4-hydroxy-5-methylphenyl)methyl]propanedioate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)C(C(=O)OCCCCCCCCCCCCCCCCCC)CC1=CC(C)=C(O)C(C(C)(C)C)=C1 KQEPQKRGTBAQRR-UHFFFAOYSA-N 0.000 description 1
- 150000002009 diols Chemical class 0.000 description 1
- 238000007598 dipping method Methods 0.000 description 1
- PWWSSIYVTQUJQQ-UHFFFAOYSA-N distearyl thiodipropionate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)CCSCCC(=O)OCCCCCCCCCCCCCCCCCC PWWSSIYVTQUJQQ-UHFFFAOYSA-N 0.000 description 1
- 235000019305 distearyl thiodipropionate Nutrition 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000032050 esterification Effects 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- SWRGUMCEJHQWEE-UHFFFAOYSA-N ethanedihydrazide Chemical compound NNC(=O)C(=O)NN SWRGUMCEJHQWEE-UHFFFAOYSA-N 0.000 description 1
- UHESRSKEBRADOO-UHFFFAOYSA-N ethyl carbamate;prop-2-enoic acid Chemical class OC(=O)C=C.CCOC(N)=O UHESRSKEBRADOO-UHFFFAOYSA-N 0.000 description 1
- 125000004494 ethyl ester group Chemical group 0.000 description 1
- 229920006228 ethylene acrylate copolymer Polymers 0.000 description 1
- 229920001038 ethylene copolymer Polymers 0.000 description 1
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000003063 flame retardant Substances 0.000 description 1
- IVJISJACKSSFGE-UHFFFAOYSA-N formaldehyde;1,3,5-triazine-2,4,6-triamine Chemical compound O=C.NC1=NC(N)=NC(N)=N1 IVJISJACKSSFGE-UHFFFAOYSA-N 0.000 description 1
- WOLATMHLPFJRGC-UHFFFAOYSA-N furan-2,5-dione;styrene Chemical compound O=C1OC(=O)C=C1.C=CC1=CC=CC=C1 WOLATMHLPFJRGC-UHFFFAOYSA-N 0.000 description 1
- 235000010382 gamma-tocopherol Nutrition 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- AHAREKHAZNPPMI-UHFFFAOYSA-N hexa-1,3-diene Chemical compound CCC=CC=C AHAREKHAZNPPMI-UHFFFAOYSA-N 0.000 description 1
- NZYMWGXNIUZYRC-UHFFFAOYSA-N hexadecyl 3,5-ditert-butyl-4-hydroxybenzoate Chemical compound CCCCCCCCCCCCCCCCOC(=O)C1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NZYMWGXNIUZYRC-UHFFFAOYSA-N 0.000 description 1
- 229920005669 high impact polystyrene Polymers 0.000 description 1
- 239000004797 high-impact polystyrene Substances 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 229920002681 hypalon Polymers 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- JYGFTBXVXVMTGB-UHFFFAOYSA-N indolin-2-one Chemical class C1=CC=C2NC(=O)CC2=C1 JYGFTBXVXVMTGB-UHFFFAOYSA-N 0.000 description 1
- 238000001746 injection moulding Methods 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 150000002513 isocyanates Chemical class 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 239000004922 lacquer Substances 0.000 description 1
- 150000003951 lactams Chemical class 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 238000003475 lamination Methods 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 150000002690 malonic acid derivatives Chemical class 0.000 description 1
- 150000002696 manganese Chemical class 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 150000007974 melamines Chemical class 0.000 description 1
- 239000006078 metal deactivator Substances 0.000 description 1
- 229910052987 metal hydride Inorganic materials 0.000 description 1
- 150000004681 metal hydrides Chemical class 0.000 description 1
- 229910000000 metal hydroxide Inorganic materials 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- VRBLLGLKTUGCSG-UHFFFAOYSA-N methyl 3-[3-tert-butyl-5-(5-chlorobenzotriazol-2-yl)-4-hydroxyphenyl]propanoate Chemical compound CC(C)(C)C1=CC(CCC(=O)OC)=CC(N2N=C3C=C(Cl)C=CC3=N2)=C1O VRBLLGLKTUGCSG-UHFFFAOYSA-N 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 229960002900 methylcellulose Drugs 0.000 description 1
- ZGEGCLOFRBLKSE-UHFFFAOYSA-N methylene hexane Natural products CCCCCC=C ZGEGCLOFRBLKSE-UHFFFAOYSA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 239000003607 modifier Substances 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- RYKIMYINWPEUBW-UHFFFAOYSA-N n',n'-diphenyloxamide Chemical compound C=1C=CC=CC=1N(C(=O)C(=O)N)C1=CC=CC=C1 RYKIMYINWPEUBW-UHFFFAOYSA-N 0.000 description 1
- YIMHRDBSVCPJOV-UHFFFAOYSA-N n'-(2-ethoxyphenyl)-n-(2-ethylphenyl)oxamide Chemical compound CCOC1=CC=CC=C1NC(=O)C(=O)NC1=CC=CC=C1CC YIMHRDBSVCPJOV-UHFFFAOYSA-N 0.000 description 1
- FPQJEXTVQZHURJ-UHFFFAOYSA-N n,n'-bis(2-hydroxyethyl)oxamide Chemical compound OCCNC(=O)C(=O)NCCO FPQJEXTVQZHURJ-UHFFFAOYSA-N 0.000 description 1
- ZJFPXDGPJMHQMW-UHFFFAOYSA-N n,n'-bis[3-(dimethylamino)propyl]oxamide Chemical compound CN(C)CCCNC(=O)C(=O)NCCCN(C)C ZJFPXDGPJMHQMW-UHFFFAOYSA-N 0.000 description 1
- FTWUXYZHDFCGSV-UHFFFAOYSA-N n,n'-diphenyloxamide Chemical compound C=1C=CC=CC=1NC(=O)C(=O)NC1=CC=CC=C1 FTWUXYZHDFCGSV-UHFFFAOYSA-N 0.000 description 1
- YASWBJXTHOXPGK-UHFFFAOYSA-N n-(4-hydroxyphenyl)octadecanamide Chemical compound CCCCCCCCCCCCCCCCCC(=O)NC1=CC=C(O)C=C1 YASWBJXTHOXPGK-UHFFFAOYSA-N 0.000 description 1
- DARUEKWVLGHJJT-UHFFFAOYSA-N n-butyl-1-[4-[4-(butylamino)-2,2,6,6-tetramethylpiperidin-1-yl]-6-chloro-1,3,5-triazin-2-yl]-2,2,6,6-tetramethylpiperidin-4-amine Chemical compound CC1(C)CC(NCCCC)CC(C)(C)N1C1=NC(Cl)=NC(N2C(CC(CC2(C)C)NCCCC)(C)C)=N1 DARUEKWVLGHJJT-UHFFFAOYSA-N 0.000 description 1
- BLBLVDQTHWVGRA-UHFFFAOYSA-N n-butyl-3-[4-[4-(butylamino)-1,2,2,6,6-pentamethylpiperidin-3-yl]-6-chloro-1,3,5-triazin-2-yl]-1,2,2,6,6-pentamethylpiperidin-4-amine Chemical compound CCCCNC1CC(C)(C)N(C)C(C)(C)C1C1=NC(Cl)=NC(C2C(N(C)C(C)(C)CC2NCCCC)(C)C)=N1 BLBLVDQTHWVGRA-UHFFFAOYSA-N 0.000 description 1
- 229920005615 natural polymer Polymers 0.000 description 1
- 150000002816 nickel compounds Chemical class 0.000 description 1
- JFNLZVQOOSMTJK-KNVOCYPGSA-N norbornene Chemical compound C1[C@@H]2CC[C@H]1C=C2 JFNLZVQOOSMTJK-KNVOCYPGSA-N 0.000 description 1
- 239000002667 nucleating agent Substances 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- IXVLEAZXSJJKFR-UHFFFAOYSA-N octadecyl 2-[(4-hydroxy-3,5-dimethylphenyl)methylsulfanyl]acetate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)CSCC1=CC(C)=C(O)C(C)=C1 IXVLEAZXSJJKFR-UHFFFAOYSA-N 0.000 description 1
- RNVAPPWJCZTWQL-UHFFFAOYSA-N octadecyl 3,5-ditert-butyl-4-hydroxybenzoate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)C1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 RNVAPPWJCZTWQL-UHFFFAOYSA-N 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- XQAABEDPVQWFPN-UHFFFAOYSA-N octyl 3-[3-(benzotriazol-2-yl)-5-tert-butyl-4-hydroxyphenyl]propanoate Chemical compound CC(C)(C)C1=CC(CCC(=O)OCCCCCCCC)=CC(N2N=C3C=CC=CC3=N2)=C1O XQAABEDPVQWFPN-UHFFFAOYSA-N 0.000 description 1
- DMFXLIFZVRXRRR-UHFFFAOYSA-N octyl 3-[3-tert-butyl-5-(5-chlorobenzotriazol-2-yl)-4-hydroxyphenyl]propanoate Chemical compound CC(C)(C)C1=CC(CCC(=O)OCCCCCCCC)=CC(N2N=C3C=C(Cl)C=CC3=N2)=C1O DMFXLIFZVRXRRR-UHFFFAOYSA-N 0.000 description 1
- NTTIENRNNNJCHQ-UHFFFAOYSA-N octyl n-(3,5-ditert-butyl-4-hydroxyphenyl)carbamate Chemical compound CCCCCCCCOC(=O)NC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NTTIENRNNNJCHQ-UHFFFAOYSA-N 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- RGSFGYAAUTVSQA-UHFFFAOYSA-N pentamethylene Natural products C1CCCC1 RGSFGYAAUTVSQA-UHFFFAOYSA-N 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- 229960000969 phenyl salicylate Drugs 0.000 description 1
- ZPNJBTBYIHBSIG-UHFFFAOYSA-N phenyl-(2,2,6,6-tetramethylpiperidin-4-yl)methanone Chemical compound C1C(C)(C)NC(C)(C)CC1C(=O)C1=CC=CC=C1 ZPNJBTBYIHBSIG-UHFFFAOYSA-N 0.000 description 1
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 1
- XRBCRPZXSCBRTK-UHFFFAOYSA-N phosphonous acid Chemical class OPO XRBCRPZXSCBRTK-UHFFFAOYSA-N 0.000 description 1
- 239000012994 photoredox catalyst Substances 0.000 description 1
- XNGIFLGASWRNHJ-UHFFFAOYSA-L phthalate(2-) Chemical compound [O-]C(=O)C1=CC=CC=C1C([O-])=O XNGIFLGASWRNHJ-UHFFFAOYSA-L 0.000 description 1
- DOIRQSBPFJWKBE-UHFFFAOYSA-N phthalic acid di-n-butyl ester Natural products CCCCOC(=O)C1=CC=CC=C1C(=O)OCCCC DOIRQSBPFJWKBE-UHFFFAOYSA-N 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920001627 poly(4-methyl styrene) Polymers 0.000 description 1
- 229920001084 poly(chloroprene) Polymers 0.000 description 1
- 229920001643 poly(ether ketone) Polymers 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920003251 poly(α-methylstyrene) Polymers 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 229920001515 polyalkylene glycol Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 229920001707 polybutylene terephthalate Polymers 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229940068886 polyethylene glycol 300 Drugs 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 229920001195 polyisoprene Polymers 0.000 description 1
- 229920000193 polymethacrylate Polymers 0.000 description 1
- 229920005606 polypropylene copolymer Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 239000011118 polyvinyl acetate Substances 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 229920002620 polyvinyl fluoride Polymers 0.000 description 1
- 239000005033 polyvinylidene chloride Substances 0.000 description 1
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 239000012429 reaction media Substances 0.000 description 1
- 239000012744 reinforcing agent Substances 0.000 description 1
- 229940066675 ricinoleate Drugs 0.000 description 1
- 229910052895 riebeckite Inorganic materials 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 238000009987 spinning Methods 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000001384 succinic acid Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- MHSKRLJMQQNJNC-UHFFFAOYSA-N terephthalamide Chemical compound NC(=O)C1=CC=C(C(N)=O)C=C1 MHSKRLJMQQNJNC-UHFFFAOYSA-N 0.000 description 1
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 description 1
- 238000002411 thermogravimetry Methods 0.000 description 1
- 229920002803 thermoplastic polyurethane Polymers 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- YONPGGFAJWQGJC-UHFFFAOYSA-K titanium(iii) chloride Chemical compound Cl[Ti](Cl)Cl YONPGGFAJWQGJC-UHFFFAOYSA-K 0.000 description 1
- 229960000984 tocofersolan Drugs 0.000 description 1
- AOBORMOPSGHCAX-DGHZZKTQSA-N tocofersolan Chemical compound OCCOC(=O)CCC(=O)OC1=C(C)C(C)=C2O[C@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C AOBORMOPSGHCAX-DGHZZKTQSA-N 0.000 description 1
- 229930003799 tocopherol Natural products 0.000 description 1
- 239000011732 tocopherol Substances 0.000 description 1
- 235000019149 tocopherols Nutrition 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 150000003918 triazines Chemical class 0.000 description 1
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- IVIIAEVMQHEPAY-UHFFFAOYSA-N tridodecyl phosphite Chemical compound CCCCCCCCCCCCOP(OCCCCCCCCCCCC)OCCCCCCCCCCCC IVIIAEVMQHEPAY-UHFFFAOYSA-N 0.000 description 1
- CNUJLMSKURPSHE-UHFFFAOYSA-N trioctadecyl phosphite Chemical compound CCCCCCCCCCCCCCCCCCOP(OCCCCCCCCCCCCCCCCCC)OCCCCCCCCCCCCCCCCCC CNUJLMSKURPSHE-UHFFFAOYSA-N 0.000 description 1
- WGKLOLBTFWFKOD-UHFFFAOYSA-N tris(2-nonylphenyl) phosphite Chemical compound CCCCCCCCCC1=CC=CC=C1OP(OC=1C(=CC=CC=1)CCCCCCCCC)OC1=CC=CC=C1CCCCCCCCC WGKLOLBTFWFKOD-UHFFFAOYSA-N 0.000 description 1
- 229920006305 unsaturated polyester Polymers 0.000 description 1
- 150000003672 ureas Chemical class 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 150000003751 zinc Chemical class 0.000 description 1
- XOOUIPVCVHRTMJ-UHFFFAOYSA-L zinc stearate Chemical compound [Zn+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O XOOUIPVCVHRTMJ-UHFFFAOYSA-L 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
- 235000007680 β-tocopherol Nutrition 0.000 description 1
- 239000011590 β-tocopherol Substances 0.000 description 1
- 239000002478 γ-tocopherol Substances 0.000 description 1
- QUEDXNHFTDJVIY-UHFFFAOYSA-N γ-tocopherol Chemical class OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1 QUEDXNHFTDJVIY-UHFFFAOYSA-N 0.000 description 1
- QUEDXNHFTDJVIY-DQCZWYHMSA-N γ-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1 QUEDXNHFTDJVIY-DQCZWYHMSA-N 0.000 description 1
- 239000002446 δ-tocopherol Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/34—Heterocyclic compounds having nitrogen in the ring
- C08K5/3467—Heterocyclic compounds having nitrogen in the ring having more than two nitrogen atoms in the ring
- C08K5/3472—Five-membered rings
- C08K5/3475—Five-membered rings condensed with carbocyclic rings
Definitions
- the present invention relates to 2-(2'-hydroxyphenyl)benzotriazoles.
- the present invention relates to 2-(2'-hydroxyphenyl)benzotriazoles containing a 2,4-imidazolidinedione group or a 2,4-imidazolidinedione-5,5-disubstituted group in the molecule, a process for their preparation and their use as light stabilizers for organic polymers.
- the present invention also relates to the polymeric compositions stabilized with the above benzotriazoles and to the end-products obtained from these compositions.
- EP-A-0 057 160 discloses 2-(2'-hydroxyphenyl-3'-tert.-butyl)benzotriazoles comprising a hydrocarbon chain which contains a carbonyl group and their use as UV stabilizers.
- US Patent 4,077,971 discloses tetrahydrophthalimide methyl-2-phenylbenzatriazoles useful as ultraviolet absorbers.
- US Patent 3,629,192, CH Patent 476 745 and FR Patent 1 324 899 disclose 2-(2'-hydroxyphenyl-)benzotriazoles comprising amine or amide groups. The compounds are said to act as light-stabilizers.
- EP-A-0867435 disclosing 2-(2'hydroxyphenyl)benzotriazoles and their use as light stabilizers for organic polymers.
- the benzotriazoles however have various disadvantages. In fact, they are often rather volatile, they have a low thermal stability and, as they sometimes have a significant absorption at 400 nm, they give the polymers, into which they are incorporated, a yellow colouring.
- the present invention therefore relates to 2-(2'-hydroxyphenyl)benzotriazoles having general formula (I): wherein:
- the compounds having general formula (I) can be used as light stabilizers for organic polymers.
- the C 5 -C 18 cycloalkyl groups, the C 6 -C 14 aryl groups and the heterocyclic groups with 5 or 6 atoms are defined as being optionally substituted, these groups are substituted with: halogen atoms selected from chlorine and bromine, linear or branched C 1 -C 18 alkyl groups, linear or branched C 2 -C 18 alkenyl groups; linear or branched C 2 -C 18 alkynyl groups, OH groups, NH groups, SH groups.
- C 1 -C 18 alkyl groups are: methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, t-butyl, t-amyl, 2-ethylhexyl, n-octyl, 1,1,3,3-tetramethylbutyl, n-dodecyl, 1,1,7,7-tetramethyloctyl, n-octadecyl, etc.
- C 2 -C 18 alkenyl groups are: vinyl, propylene, butylene, pentylene, hexylene, etc.
- C 2 -C 18 alkynyl groups are: acetylene, propyne, butyne, 2-butyne, etc.
- C 5 -C 18 cycloalkyl groups optionally substituted, are: cyclohexyl, cyclopentyl, methylcyciohexyl, etc.
- C 7 -C 15 arylalkyl or alkylaryl groups are: benzyl, 2-phenylethyl, 4-t-butylbenzyl, etc.
- C 6 -C 14 aryl groups optionally substituted, are: phenyl, naphthyl, anthracenyl, 2-hydroxyphenyl, etc.
- C 1 -C 18 alkoxyl groups are: methoxyl, ethoxyl, propoxyl, n-butoxyl, etc.
- heterocyclic groups with 5 or 6 atoms are: piperidine, morpholine, piperazine, triazole, tetramethylpiperidine, pentamethylpiperidine, tetramethylmorpholine, pentamethylmorpholine, 4-hydroxy-tetramethylpiperidine, etc.
- the compounds having general formula (I) of the present invention can be prepared with various processes.
- a further object of the present invention relates to a process for the preparation of 2-(2'-hydroxyphenyl)benzotriazoles having general formula (I).
- a process for the preparation of compounds having general formula (I) comprises:
- the 2-(2'-hydroxyphenyl)benzotriazoles having general formula (IX) can be prepared as described, for example, in German patent application DE 4.237.817.
- the secondary amines having general formula (X) and 2,4-imidazolidinediones having general formula (XII), are commercially available compounds.
- the compounds having general formula (I) of the present invention can be used as light stabilizers for a wide range of organic polymers.
- Organic polymers capable of being stabilized with the compounds of the present invention are:
- the compounds having formula (I) of the present invention are particularly useful in the stabilization of polycarbonates.
- a further object of the present invention relates to polymeric compositions containing an organic polymer and an effective quantity of one or more compounds having general formula (I).
- the compounds having general formula (I) of the present invention can be used as such or combined with other stabilizers, in the above polymeric compositions.
- the above compounds having general formula (I) are used in a quantity ranging from about 0.1% to about 5% by weight of the weight of the polymeric compositions to be stabilized, although the quantity used varies according to the substrate to be stabilized and the final application. They are preferably added in a quantity ranging from about 0.5% to about 3% by weight of the weight of the polymeric compositions to be stabilized.
- the compounds having general formula (I), optionally in the presence of other additives, can be easily incorporated in the organic polymers to be stabilized using the conventional techniques. This incorporation can take place before or during the formation of the end-product, for example, by mixing the compounds having general formula (I) in powder form with the polymer to be stabilized, or by adding these compounds to the polymer to be stabilized in the molten state or in solution, or applying a solution or suspension of these compounds to the polymer to be stabilized, optionally evaporating the solvent used.
- the elastomers can be stabilized as latexes.
- Another method for incorporating the compounds having general formula (I) in the organic polymers comprises the addition of these before or during the polymerization of the corresponding monomers or before the cross-linking.
- the compounds having general formula (I) or their mixtures can be added to the polymer to be stabilized also in masterbatch form which comprises these compounds in a concentration ranging, for example, from 2.5% to 25% by weight.
- the compounds having general formula (I) can be conveniently incorporated in the organic polymers to be stabilized by means of the following methods:
- polymeric compositions stabilized as said above can be converted to end-products such as, for example, fibers, films, tapes, sheets, multi-layer sheets, containers, tubes and other forms, by means of methods known in the art such as, for example, casting, hot moulding, spinning, extrusion or injection moulding.
- the present invention therefore also relates to the use of the above polymeric compositions for the production of end-products.
- multi-layer systems in which one of the above compositions having a relatively high content of a compound having general formula (I), for example, between 5% and 15% by weight, is applied in the form of a thin film (10-100 ⁇ m in thickness) to a shaped article consisting of a polymer not containing or containing a small quantity of a compound having general formula (I).
- This application can be carried out during the formation of said article, for example, by means of a co-extrusion.
- the application can also be effected however on the end shaped-article, for example, by lamination with a film or by coating with a solution.
- the surface layer or layers of the end-article act as a UV filter which protects the inside of the articles from the deteriorating action of UV light.
- the upper layer preferably contains from 5% to 15% by weight, more preferably from 5% to 10% by weight, of at least one compound having general formula (I).
- the polymers stabilized as described above have a high resistance to degradation caused by atmospheric agents, in particular a high resistance to UV light. They are therefore capable of maintaining their colour and brightness for a long period even when exposed to external agents.
- compositions described above can also be used as compositions for coating or painting ("coating compositions") such as, for example, paints, lacquers, plastic-based compositions.
- organic polymer is selected from:
- thermoplastic polymers (a) containing heteroatoms, in particular nitrogen, sulfur and/or oxygen, in the main chain are listed above under points 13 to 20.
- polycarbonates polyesters, polyamides, polyacetals, polyphenylene oxides and polyphenylene sulfides are preferred; particularly preferred are polycarbonates, polyesters such as, for example, polyethylene terephthalate (PET), and polyamides (PA) such as, for example, PA 6 and PA 6/6; even more preferred are polycarbonates.
- PET polyethylene terephthalate
- PA polyamides
- Paint ligands (b) can comprise at least one of the organic polymers specified herebelow. Specific examples of paints containing specific ligands are:
- the paints can be applied as one or two layers of coating ("one- or two-coat") and the stabilizing compounds having formula (I) are preferably added to the upper colourless coating.
- the paints can be applied to the substrate (metal, plastic, wood, etc.) using the conventional methods such as, for example, brushing, spraying, pouring, dipping or electrophoresis.
- a preferred embodiment of the present invention consists in paints or coatings (for example car coatings) comprising at least one compound having general formula (I).
- Ligands which can be used for the purpose are, for example, those listed above.
- the compounds having general formula (I) of the present invention can be combined, as already mentioned above, with other conventional additives or their mixtures. These additives are added in a quantity ranging-from about 0.1% to about 5% by weight of the weight of the polymeric compositions to be stabilized, preferably from about 0.5% to about 3% by weight. Some of the additives used are listed below as an example.
- Films with a thickness of 100 ⁇ m are prepared from the above solutions by casting.
- the films obtained are subjected to accelerated aging in an Atlas CI 65 Weatherometer under the following conditions:
- the yellow index (YI) of the above films is measured using the method ASTM E 313. The crushing time is also analyzed.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Cosmetics (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Claims (23)
- 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) worin:X ein Wasserstoffatom, ein unter Chlor und Brom ausgewähltes Halogenatom, eine geradekettige oder verzweigte C1-C18-Alkylgruppe, eine geradekettige oder verzweigte C1-C18-Alkoxygruppe, eine Cyangruppe darstellt,R ein unter Chlor und Brom ausgewähltes Halogenatom, eine geradekettige oder verzweigte C1-C18-Alkylgruppe, eine geradekettige oder verzweigte C2-C18-Alkenylgruppe, eine geradekettige oder verzweigte C2-C18-Alkinylgruppe, eine C5-C18-Cycloalkylgruppe, wobei die Cycloalkylgruppe gegebenenfalls substituiert ist, eine C7-C15-Arylalkyl- oder Alkylaryl-Gruppe, eine C6-C14-Arylgruppe, wobei die Arylgruppe gegebenenfalls substituiert ist, eine geradekettige oder verzweigte C1-C18-Alkoxygruppe, eine heterocyclische Gruppe mit 5 oder 6 Atomen, die mindestens ein unter Sauerstoff, Stickstoff und Schwefel ausgewähltes Heteroatom enthält, wobei die heterocyclische Gruppe gegebenenfalls substituiert ist, eine Gruppe der Formel eine -COR4-Gruppe oder eine -NR5R6-Gruppe darstellt, worin R4, R5 und R6 gleich oder verschieden sind und eine geradekettige oder verzweigte C1-C18-Alkylgruppe, eine geradekettige oder verzweigte C2-C18-Alkenylgruppe, eine geradekettige oder verzweigte C2-C18-Alkinylgruppe, eine C5-C18-Cycloalkylgruppe, die gegebenenfalls substituiert ist, darstellen, eine C7-C15-Arylalkyl- oder Alkylarylgruppe, eine C6-C14-Arylgruppe, wobei die Arylgruppe gegebenenfalls substituiert ist, eine heterocyclische Gruppe mit 5 oder 6 Atomen, die mindestens ein unter Sauerstoff, Stickstoff und Schwefel ausgewähltes Heteroatom enthält und wobei die heterocyclische Gruppe gegebenenfalls substituiert ist, darstellen,
oder R eine Estergruppe der allgemeinen Formel (II), (III) oder (IV) oder eine Amidgruppe der allgemeinen Formel (V) darstellt,
worin R' eine geradekettige oder verzweigte C1-C18-Alkylgruppe, eine geradekettige oder verzweigte C2-C18-Alkenylgruppe, eine geradekettige oder verzweigte C2-C18-Alkinylgruppe, eine C5-C18-Cycloalkylgruppe, wobei die Cycloalkylgruppe gegebenenfalls substituiert ist, eine C7-C15-Arylalkyl- oder Alkylarylgruppe, eine C6-C14-Arylgruppe, wobei die Arylgruppe gegebenenfalls substituiert ist, eine geradekettige oder verzweigte C1-C18-Alkoxygruppe, eine heterocyclische Gruppe mit 5 oder 6 Atomen, die mindestens ein unter Sauerstoff, Stickstoff und Schwefel ausgewähltes Heteroatom enthält, wobei die heterocyclische Gruppe gegebenenfalls substituiert ist, darstellt,
oder R eine 4,4'-Ethylidenbisphenol-Gruppe der Formel (VI) darstellt:R1 ein Wasserstoffatom, eine geradekettige oder verzweigte C1-C18-Alkylgruppe, eine geradekettige oder verzweigte C2-C18-Alkenylgruppe, eine geradekettige oder verzweigte C2-C18-Alkinylgruppe, eine C5-C18-Cycloalkylgruppe, wobei die Cycloalkylgruppe gegebenenfalls substituiert ist, eine C7-C15-Arylalkyl- oder Alkylarylgruppe, eine C6-C14-Arylgruppe, wobei die Arylgruppe-gegebenenfalls substituiert ist, eine heterocyclische Gruppe mit 5 oder 6 Atomen, die mindestens ein unter Sauerstoff, Stickstoff und Schwefel ausgewähltes Heteroatom enthält, wobei die heterocyclische Gruppe gegebenenfalls substituiert ist, eine Acylgruppe der allgemeinen Formel (VII): oder eine Estergruppe der allgemeinen Formel (VIII): worin R' die vorstehend gegebene Bedeutung hat, darstellt, R2und R3 gleich oder verschieden sind und ein Wasserstoffatom, eine geradekettige oder verzweigte C1-C18-Alkylgruppe, eine Phenylgruppe, eine heterocyclische Gruppe mit 5 oder 6 Atomen, die mindestens ein unter Sauerstoff, Stickstoff und Schwefel ausgewähltes Heteröatom enthält, wobei die heterocyclische Gruppe gegebenenfalls substituiert ist, darstellen,
worin die C5-C18-Cycloalkylgruppen, die C6-C14-Arylgruppen und die heterocyclischen Gruppen mit 5 oder 6 Atomen mit Halogenatomen, ausgewählt unter Chlor und Brom, geradekettigen oder verzweigten C1-C18-Alkylgruppen, geradekettigen oder verzweigten C2-C18-Alkenylgruppen, geradekettigen oder verzweigten C2-C18-Alkinylgruppen, OH-Gruppen, NH-Gruppen, SH-Gruppen substituiert sind. - 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) nach Anspruch 1, worin die C1-C18-Alkylgruppen Methyl, Ethyl, Propyl, Isopropyl, n-Butyl, sec-Butyl, t-Butyl, t-Amyl, 2-Ethylhexyl, n-Octyl, 1,1,3,3-Tetramethylbutyl, n-Dodecyl, 1,1,7,7-Tetramethyloctyl oder n-Octadecyl sind.
- 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) nach Anspruch 1, worin die C2-C18-Alkenylgruppen Vinyl, Propylen, Butylen, Pentylen oder Hexylen sind.
- 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) nach Anspruch 1, worin die C2-C18-Alkinylgruppen Acetylen, Propin, Butin oder 2-Butin sind.
- 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) nach Anspruch 1, worin die gegebenenfalls substituierten C5-C18-Cycloalkylgruppen Cyclohexyl, Cyclopentyl oder Methylcyclohexyl sind.
- 2-(2'-Hydroxyphenyl) benzotriazole der allgemeinen Formel (I) nach Anspruch 1, worin die C7-C15-Arylalkyl- oder Alkylarylgruppen Benzyl, 2-Phenylethyl oder 4-t-Butylbenzyl sind.
- 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) nach Anspruch 1, worin die gegebenenfalls substituierten C6-C14-Arylgruppen Phenyl, Naphthyl, Anthracenyl oder 2-Hydroxyphenyl sind.
- 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) nach Anspruch 1, worin die C1-C18-Alkoxygruppen Methoxy, Ethoxy, Propoxy oder n-Butoxy sind.
- 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) nach Anspruch 1, worin die gegebenenfalls substituierten heterocyclischen Gruppen mit 5 oder 6 Atomen Piperidin, Morpholin, Piperazin, Triazol, Tetramethylpiperidin, Pentamethylpiperidin, Tetramethylmorpholin, Pentamethylmorpholin oder 4-Hydroxytetramethylpiperidin sind.
- Verfahren zur Herstellung von 2-(2'-Hydroxyphenyl)benzotriazolen der allgemeinen Formel (I) nach einem der vorhergehenden Patentansprüche, welches umfaßt:(a) die Umsetzung eines 2-(2'-Hydroxyphenyl)benzotriazols der allgemeinen Formel (IX): worin X und R die vorstehend definierten Bedeutungen haben, mit einem sekundären Amin der allgemeinen Formel (X): worin R7 und R8 gleich oder verschieden sind, vorzugsweise gleich sind, und eine geradekettige Alkylgruppe mit 3 oder mehr Kohlenstoffatomen darstellen, in Gegenwart von Formaldehyd oder Paraformaldehyd und eines inerten organischen Lösungsmittels bei einer Temperatur im Bereich von 95°C bis 100°C während einer Dauer im Bereich von 40 Stunden bis 60 Stunden, wobei die Mannich-Base der allgemeinen Formel (XI) erhalten wird: worin R, R7 und R8 die vorstehend definierte Bedeutung haben,(b) Umsetzen der in Stufe (a) erhaltenen Mannich-Base der allgemeinen Formel (XI) mit einem 2,4-Imidazolidindion der allgemeinen Formel (XII): worin R1, R2 und R3 die vorstehend beschriebenen Bedeutungen haben, in Gegenwart eines alkalischen Katalysators und eines inerten organischen Lösungsmittels mit einem Siedepunkt von 160°C bei einer Temperatur im Bereich von 20°C bis 200°C während einer Dauer im Bereich von 20 Stunden bis 30 Stunden, wobei eine Lösung erhalten wird, aus der nach dem Abkühlen auf Raumtemperatur und Entfernen des Katalysators durch Filtration und-des Lösungsmittels durch Verdampfen die gewünschte Verbindung durch Kristallisation in Gegenwart eines inerten organischen Lösungsmittels isoliert wird, das unter Alkoholen, Monoalkylethern, geradekettigen oder cyclischen aliphatischen Kohlenwasserstoffen, aromatischen Kohlenwasserstoffen und chlorierten aromatischen Lösungsmitteln ausgewählt ist.
- Verfahren nach Anspruch 12, wobei die Stufe (a) in Gegenwart von Paraformaldehyd und n-Butanol durchgeführt wird.
- Verfahren nach Anspruch 12, wobei die in Stufe (b) verwendeten Katalysatoren Natriummethylat, Natriumhydroxid, Kaliumhydroxide, Lithiumhydroxid sind.
- Verfahren nach Anspruch 12, wobei die in Stufe (b) verwendeten inerten organischen Lösungsmittel mit einem Siedepunkt von 160°C 1,2,4-Trimethylbenzol oder 4-Isopropylbenzol sind.
- Verfahren nach Anspruch 12, wobei Stufe (b) bei einer Temperatur im Bereich von 40°C bis 170°C durchgeführt wird.
- Verfahren nach Anspruch 12, wobei die in Stufe (b) verwendeten Kristallisationslösungsmittel Methanol, 2-Methoxyethanol, Hexan, Heptan, Cyclohexan, Methylcyclohexan, Toluol oder Chlorbenzol sind.
- Polymerzusammensetzungen, die ein organisches Polymer und eine wirksame Menge eines oder mehrerer 2-(2'-Hydroxyphenyl)-benzotriazole der allgemeinen Formel (I) nach einem der vorhergehenden Patentansprüche enthalten.
- Polymerzusammensetzungen nach Anspruch 18, wobei die 2-(2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) mit anderen Stabilisatoren kombiniert sind.
- Polymerzusammensetzungen nach Anspruch 18 oder 19, wobei das organische Polymer unter Polycarbonaten ausgewählt ist.
- Verwendung der (2'-Hydroxyphenyl)benzotriazole der allgemeinen Formel (I) nach einem der Ansprüche 1 bis 11 als Lichtstabilisatoren für organische Polymere.
- Verwendung der Polymerzusammensetzungen nach Anspruch 18 oder 19 zur Herstellung von Mehrschichtsystemen, wobei die obere Schicht einer Dicke von 10 bis 100 µm diese Zusammensetzungen enthält, während die innere Schicht eine Verbindung der allgemeinen Formel (I) nach einem der Ansprüche 1 bis 11 nicht oder in geringer Menge enthält.
- Verwendung der Polymerzusammensetzungen nach Anspruch 18 oder 19 als Überzugs- oder Anstrich-Zusammensetzungen.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ITMI972436 | 1997-10-30 | ||
| IT97MI002436A IT1295933B1 (it) | 1997-10-30 | 1997-10-30 | 2-(2'-idrossifenil)benzotriazoli e procedimento per la loro preparazione |
| PCT/EP1998/006654 WO1999023093A1 (en) | 1997-10-30 | 1998-10-22 | 2-(2'-hydroxyphenyl)benzotriazoles used as u.v. stabilisers |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1027348A1 EP1027348A1 (de) | 2000-08-16 |
| EP1027348B1 true EP1027348B1 (de) | 2003-07-30 |
Family
ID=11378128
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19980955498 Expired - Lifetime EP1027348B1 (de) | 1997-10-30 | 1998-10-22 | 2-(2'-hydroxyphenyl)benzotriazole verwendbar als uv-stabilisatoren |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US6407254B1 (de) |
| EP (1) | EP1027348B1 (de) |
| JP (1) | JP2001521936A (de) |
| AT (1) | ATE246187T1 (de) |
| DE (1) | DE69816851T2 (de) |
| DK (1) | DK1027348T3 (de) |
| ES (1) | ES2202908T3 (de) |
| IT (1) | IT1295933B1 (de) |
| PT (1) | PT1027348E (de) |
| WO (1) | WO1999023093A1 (de) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6166218A (en) * | 1996-11-07 | 2000-12-26 | Ciba Specialty Chemicals Corporation | Benzotriazole UV absorbers having enhanced durability |
| US6846929B2 (en) * | 2001-04-02 | 2005-01-25 | Ciba Specialty Chemicals Corporation | Benzotriazole/hals molecular combinations and compositions stabilized therewith |
| US7279576B2 (en) | 2002-12-31 | 2007-10-09 | Deciphera Pharmaceuticals, Llc | Anti-cancer medicaments |
| US7144911B2 (en) | 2002-12-31 | 2006-12-05 | Deciphera Pharmaceuticals Llc | Anti-inflammatory medicaments |
| US7202257B2 (en) | 2003-12-24 | 2007-04-10 | Deciphera Pharmaceuticals, Llc | Anti-inflammatory medicaments |
| JP4820185B2 (ja) * | 2006-02-28 | 2011-11-24 | シプロ化成株式会社 | ベンジリデンヒダントイン誘導体化合物 |
| KR20100014811A (ko) | 2007-04-20 | 2010-02-11 | 데시페라 파마슈티칼스, 엘엘씨. | 골수증식성 질환 및 기타 증식성 질환의 치료에 유용한 키나제 억제제 |
| US8461179B1 (en) | 2012-06-07 | 2013-06-11 | Deciphera Pharmaceuticals, Llc | Dihydronaphthyridines and related compounds useful as kinase inhibitors for the treatment of proliferative diseases |
| US10074245B2 (en) | 2017-01-10 | 2018-09-11 | Woodward, Inc. | Force feel using a brushless DC motor |
| IL276398B2 (en) | 2018-01-31 | 2026-03-01 | Deciphera Pharmaceuticals Llc | Combination therapy for mastocytosis |
| KR102708177B1 (ko) | 2018-01-31 | 2024-09-23 | 데시페라 파마슈티칼스, 엘엘씨. | 위장관 기질 종양의 치료를 위한 병용 요법 |
| EP3938363A1 (de) | 2019-03-11 | 2022-01-19 | Teva Pharmaceuticals International GmbH | Festkörperformen von ripretinib |
| LT4013412T (lt) | 2019-08-12 | 2026-03-25 | Deciphera Pharmaceuticals, Llc | Ripretinibas, skirtas virškinamojo trakto stromos navikų gydymui |
| PT4013412T (pt) | 2019-08-12 | 2026-03-18 | Deciphera Pharmaceuticals Llc | Ripretinib para o tratamento de tumores estromais gastrointestinais |
| HRP20231699T1 (hr) | 2019-12-30 | 2024-05-10 | Deciphera Pharmaceuticals, Llc | Formulacije inhibitora amorfne kinaze i postupci njihove primjene |
| CN115243681B (zh) | 2019-12-30 | 2024-08-16 | 德西费拉制药有限责任公司 | 1-(4-溴-5-(1-乙基-7-(甲氨基)-2-侧氧基-1,2-二氢-1,6-萘啶-3-基)-2-氟苯基)-3-苯基脲的组合物 |
| CN116917272A (zh) | 2021-03-05 | 2023-10-20 | 巴斯夫欧洲公司 | 制备n,n'-二(2,2,6,6-四甲基哌啶-4-基)-1,6-己二胺的方法 |
| US11779572B1 (en) | 2022-09-02 | 2023-10-10 | Deciphera Pharmaceuticals, Llc | Methods of treating gastrointestinal stromal tumors |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CH476744A (de) * | 1961-06-16 | 1969-08-15 | Geigy Ag J R | Verfahren zur Herstellung von substituierten 2-(2'-Hydroxyphenyl)-benztriazolverbindungen |
| NL279768A (de) | 1961-06-16 | |||
| FR1324899A (fr) * | 1961-06-16 | 1963-04-19 | Geigy Ag J R | Nouveaux 2-(2'-hydroxyphényl)-benzotriazoles et leur préparation |
| US4044019A (en) * | 1974-05-14 | 1977-08-23 | Ciba-Geigy Corporation | Hydroxyphenylated hydantoins |
| JPS565279B2 (de) * | 1974-08-15 | 1981-02-04 | ||
| CH611284A5 (de) * | 1975-04-10 | 1979-05-31 | Ciba Geigy Ag | |
| EP0057160B1 (de) * | 1981-01-23 | 1985-06-19 | Ciba-Geigy Ag | 2-(2-Hydroxyphenyl)-benztriazole, ihre Verwendung als UV-Absorber und ihre Herstellung |
| ATE230742T1 (de) * | 1997-03-27 | 2003-01-15 | Great Lakes Chemical Europ | 2-(2'-hydroxphenyl)-benzotriazole und ihre verwendung als lichtschutzmittel für organische polymere |
-
1997
- 1997-10-30 IT IT97MI002436A patent/IT1295933B1/it active IP Right Grant
-
1998
- 1998-10-22 ES ES98955498T patent/ES2202908T3/es not_active Expired - Lifetime
- 1998-10-22 US US09/529,410 patent/US6407254B1/en not_active Expired - Fee Related
- 1998-10-22 JP JP2000518964A patent/JP2001521936A/ja not_active Abandoned
- 1998-10-22 PT PT98955498T patent/PT1027348E/pt unknown
- 1998-10-22 EP EP19980955498 patent/EP1027348B1/de not_active Expired - Lifetime
- 1998-10-22 WO PCT/EP1998/006654 patent/WO1999023093A1/en not_active Ceased
- 1998-10-22 DK DK98955498T patent/DK1027348T3/da active
- 1998-10-22 AT AT98955498T patent/ATE246187T1/de not_active IP Right Cessation
- 1998-10-22 DE DE69816851T patent/DE69816851T2/de not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| ES2202908T3 (es) | 2004-04-01 |
| WO1999023093A1 (en) | 1999-05-14 |
| ATE246187T1 (de) | 2003-08-15 |
| US6407254B1 (en) | 2002-06-18 |
| PT1027348E (pt) | 2003-11-28 |
| IT1295933B1 (it) | 1999-05-28 |
| DE69816851T2 (de) | 2004-05-27 |
| JP2001521936A (ja) | 2001-11-13 |
| DE69816851D1 (de) | 2003-09-04 |
| DK1027348T3 (da) | 2003-11-17 |
| EP1027348A1 (de) | 2000-08-16 |
| ITMI972436A1 (it) | 1999-04-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE69810526T2 (de) | Erfassung von Strahldüsenfehlern durch optisches Abtasten eines Probemusters | |
| EP0867435B1 (de) | 2-(2'-Hydroxphenyl)-benzotriazole und ihre Verwendung als Lichtschutzmittel für organische Polymere | |
| EP1027348B1 (de) | 2-(2'-hydroxyphenyl)benzotriazole verwendbar als uv-stabilisatoren | |
| FR2749319A1 (fr) | Melange stabilisant, composition le contenant et procede l'utilisant pour stabiliser une matiere organique | |
| EP1276802B1 (de) | Flüssige stabilisatorgemische für organische polymere | |
| EP1049690B1 (de) | Eine 2,4-imidazolidindiongruppe enthaltende 2-(2'-hydroxyphenyl)benzotriazole und verfahren zu ihrer herstellung | |
| US6353044B1 (en) | Light-stabilized organic polymers | |
| WO1999043666A1 (en) | Photochromatic compositions endowed with light fatigue resistance and photochromatic articles containing them | |
| JP2001509521A (ja) | 有機ポリマー用光安定化相乗混合物 | |
| ITMI982065A1 (it) | Miscele sinergiche stabilizzanti | |
| WO1998047893A1 (en) | Enamine derivatives for use as antioxydants for polymers | |
| ITMI972476A1 (it) | 2-(2'-idrossifenil)benzotriazoli e procedimento per la loro preparazione | |
| ITMI970719A1 (it) | 2-(2'-idrossifenil) benzotriazoli e procedimento per la loro preparazione | |
| ITMI20000326A1 (it) | Composti appartenenti alla classe delle ammine stericamente impedite procedimento per la loro preparazione e loro utilizzo in polimeri organ | |
| ITMI992158A1 (it) | Composti stabilizzanti contenenti un gruppo fenolico stearicamente impedito ed una funzione silica | |
| WO2000008096A1 (en) | Stabilizing mixture having a melting point lower than that of the highest melting component |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20000131 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: GREAT LAKES CHEMICAL (EUROPE) GMBH |
|
| 17Q | First examination report despatched |
Effective date: 20020829 |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: GREAT LAKES CHEMICAL (EUROPE) GMBH |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 69816851 Country of ref document: DE Date of ref document: 20030904 Kind code of ref document: P |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20031021 Year of fee payment: 6 Ref country code: AT Payment date: 20031021 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IE Payment date: 20031022 Year of fee payment: 6 Ref country code: FI Payment date: 20031022 Year of fee payment: 6 Ref country code: DK Payment date: 20031022 Year of fee payment: 6 Ref country code: CH Payment date: 20031022 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PT Payment date: 20031024 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: MC Payment date: 20031027 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GR Payment date: 20031029 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: LU Payment date: 20031031 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20031107 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: NV Representative=s name: AMMANN PATENTANWAELTE AG BERN |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 Ref country code: GR Ref legal event code: EP Ref document number: 20030404230 Country of ref document: GR |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20031125 Year of fee payment: 6 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CY Payment date: 20040213 Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2202908 Country of ref document: ES Kind code of ref document: T3 |
|
| ET | Fr: translation filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20040504 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041022 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041022 Ref country code: FI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041022 Ref country code: CY Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041022 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041022 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041023 Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041023 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041031 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041031 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041031 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041101 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050422 |
|
| BERE | Be: lapsed |
Owner name: *GREAT LAKES CHEMICAL (EUROPE) G.M.B.H. Effective date: 20041031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050504 |
|
| EUG | Se: european patent has lapsed | ||
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: MM4A Free format text: LAPSE DUE TO NON-PAYMENT OF FEES Effective date: 20050422 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20041023 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| BERE | Be: lapsed |
Owner name: *GREAT LAKES CHEMICAL (EUROPE) G.M.B.H. Effective date: 20041031 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20071024 Year of fee payment: 10 Ref country code: DE Payment date: 20071130 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20071029 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20071029 Year of fee payment: 10 Ref country code: FR Payment date: 20071017 Year of fee payment: 10 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20081022 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 20090501 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20090630 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090501 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20081022 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090501 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20081031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20081022 |