EP0826657A1 - Verbindungen zur Modulierung von Hormonrezeptoren, diese enthaltende Zusammensetzungen und ihre Verwendung in Therapie - Google Patents
Verbindungen zur Modulierung von Hormonrezeptoren, diese enthaltende Zusammensetzungen und ihre Verwendung in Therapie Download PDFInfo
- Publication number
- EP0826657A1 EP0826657A1 EP97401999A EP97401999A EP0826657A1 EP 0826657 A1 EP0826657 A1 EP 0826657A1 EP 97401999 A EP97401999 A EP 97401999A EP 97401999 A EP97401999 A EP 97401999A EP 0826657 A1 EP0826657 A1 EP 0826657A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- radical
- disorders
- acne
- skin
- formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000000203 mixture Substances 0.000 title claims description 36
- 150000001875 compounds Chemical class 0.000 title claims description 29
- 108091008039 hormone receptors Proteins 0.000 title 1
- 238000002560 therapeutic procedure Methods 0.000 title 1
- 150000003839 salts Chemical class 0.000 claims abstract description 9
- 125000000623 heterocyclic group Chemical group 0.000 claims abstract description 4
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 4
- 125000003545 alkoxy group Chemical group 0.000 claims abstract description 3
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 claims abstract description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 19
- 208000035475 disorder Diseases 0.000 claims description 18
- 102000003702 retinoic acid receptors Human genes 0.000 claims description 13
- 108090000064 retinoic acid receptors Proteins 0.000 claims description 13
- 208000002874 Acne Vulgaris Diseases 0.000 claims description 12
- 206010000496 acne Diseases 0.000 claims description 12
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 7
- 206010003645 Atopy Diseases 0.000 claims description 6
- 108091027981 Response element Proteins 0.000 claims description 6
- 239000003814 drug Substances 0.000 claims description 5
- 230000003780 keratinization Effects 0.000 claims description 5
- NKCCODPFBDGPRJ-UHFFFAOYSA-N nitridocarbon(1+) Chemical compound N#[C+] NKCCODPFBDGPRJ-UHFFFAOYSA-N 0.000 claims description 5
- 230000007170 pathology Effects 0.000 claims description 5
- 230000035755 proliferation Effects 0.000 claims description 5
- 108090000623 proteins and genes Proteins 0.000 claims description 5
- 230000000699 topical effect Effects 0.000 claims description 5
- 206010003694 Atrophy Diseases 0.000 claims description 4
- 241000713772 Human immunodeficiency virus 1 Species 0.000 claims description 4
- 201000004681 Psoriasis Diseases 0.000 claims description 4
- 230000032683 aging Effects 0.000 claims description 4
- 208000010668 atopic eczema Diseases 0.000 claims description 4
- 239000002537 cosmetic Substances 0.000 claims description 4
- 230000002500 effect on skin Effects 0.000 claims description 4
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 4
- HZVOZRGWRWCICA-UHFFFAOYSA-N methanediyl Chemical compound [CH2] HZVOZRGWRWCICA-UHFFFAOYSA-N 0.000 claims description 4
- 239000008194 pharmaceutical composition Substances 0.000 claims description 4
- 102000005962 receptors Human genes 0.000 claims description 4
- 108020003175 receptors Proteins 0.000 claims description 4
- 201000010153 skin papilloma Diseases 0.000 claims description 4
- 230000003612 virological effect Effects 0.000 claims description 4
- 229910052727 yttrium Inorganic materials 0.000 claims description 4
- 241000700721 Hepatitis B virus Species 0.000 claims description 3
- 208000012641 Pigmentation disease Diseases 0.000 claims description 3
- 241000700605 Viruses Species 0.000 claims description 3
- 239000003246 corticosteroid Substances 0.000 claims description 3
- 229960001334 corticosteroids Drugs 0.000 claims description 3
- 230000002757 inflammatory effect Effects 0.000 claims description 3
- 229910052757 nitrogen Inorganic materials 0.000 claims description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 3
- 208000003154 papilloma Diseases 0.000 claims description 3
- 230000019612 pigmentation Effects 0.000 claims description 3
- 201000004384 Alopecia Diseases 0.000 claims description 2
- 206010003210 Arteriosclerosis Diseases 0.000 claims description 2
- 208000027932 Collagen disease Diseases 0.000 claims description 2
- XFXPMWWXUTWYJX-UHFFFAOYSA-N Cyanide Chemical compound N#[C-] XFXPMWWXUTWYJX-UHFFFAOYSA-N 0.000 claims description 2
- 208000002506 Darier Disease Diseases 0.000 claims description 2
- 201000004624 Dermatitis Diseases 0.000 claims description 2
- 206010012441 Dermatitis bullous Diseases 0.000 claims description 2
- 208000004898 Herpes Labialis Diseases 0.000 claims description 2
- 206010021197 Ichthyoses Diseases 0.000 claims description 2
- 206010023369 Keratosis follicular Diseases 0.000 claims description 2
- 206010067152 Oral herpes Diseases 0.000 claims description 2
- 206010033554 Palmoplantar keratoderma Diseases 0.000 claims description 2
- 201000001263 Psoriatic Arthritis Diseases 0.000 claims description 2
- 208000036824 Psoriatic arthropathy Diseases 0.000 claims description 2
- 241001303601 Rosacea Species 0.000 claims description 2
- 206010039792 Seborrhoea Diseases 0.000 claims description 2
- 206010040799 Skin atrophy Diseases 0.000 claims description 2
- 206010040925 Skin striae Diseases 0.000 claims description 2
- 206010041303 Solar dermatitis Diseases 0.000 claims description 2
- 208000031439 Striae Distensae Diseases 0.000 claims description 2
- 102000040945 Transcription factor Human genes 0.000 claims description 2
- 108091023040 Transcription factor Proteins 0.000 claims description 2
- 208000009621 actinic keratosis Diseases 0.000 claims description 2
- 231100000360 alopecia Toxicity 0.000 claims description 2
- 208000011775 arteriosclerosis disease Diseases 0.000 claims description 2
- 206010003246 arthritis Diseases 0.000 claims description 2
- 230000037444 atrophy Effects 0.000 claims description 2
- 210000000748 cardiovascular system Anatomy 0.000 claims description 2
- 201000004306 epidermodysplasia verruciformis Diseases 0.000 claims description 2
- 208000009197 gingival hypertrophy Diseases 0.000 claims description 2
- TUJKJAMUKRIRHC-UHFFFAOYSA-N hydroxyl Chemical compound [OH] TUJKJAMUKRIRHC-UHFFFAOYSA-N 0.000 claims description 2
- 206010021198 ichthyosis Diseases 0.000 claims description 2
- 230000001506 immunosuppresive effect Effects 0.000 claims description 2
- 230000004968 inflammatory condition Effects 0.000 claims description 2
- 201000004607 keratosis follicularis Diseases 0.000 claims description 2
- 208000002741 leukoplakia Diseases 0.000 claims description 2
- 230000003211 malignant effect Effects 0.000 claims description 2
- 230000003287 optical effect Effects 0.000 claims description 2
- 201000008743 palmoplantar keratosis Diseases 0.000 claims description 2
- 208000029211 papillomatosis Diseases 0.000 claims description 2
- 230000000241 respiratory effect Effects 0.000 claims description 2
- 102000027483 retinoid hormone receptors Human genes 0.000 claims description 2
- 108091008679 retinoid hormone receptors Proteins 0.000 claims description 2
- 201000004700 rosacea Diseases 0.000 claims description 2
- 230000037390 scarring Effects 0.000 claims description 2
- 208000008742 seborrheic dermatitis Diseases 0.000 claims description 2
- 125000004434 sulfur atom Chemical group 0.000 claims description 2
- 230000009885 systemic effect Effects 0.000 claims description 2
- 206010004146 Basal cell carcinoma Diseases 0.000 claims 1
- 206010061598 Immunodeficiency Diseases 0.000 claims 1
- 208000029462 Immunodeficiency disease Diseases 0.000 claims 1
- 125000004093 cyano group Chemical group *C#N 0.000 claims 1
- 230000007813 immunodeficiency Effects 0.000 claims 1
- 208000029443 keratinization disease Diseases 0.000 claims 1
- 206010041823 squamous cell carcinoma Diseases 0.000 claims 1
- 125000000217 alkyl group Chemical group 0.000 abstract description 4
- IPZJQDSFZGZEOY-UHFFFAOYSA-N dimethylmethylene Chemical group C[C]C IPZJQDSFZGZEOY-UHFFFAOYSA-N 0.000 abstract description 2
- 101100295738 Gallus gallus COR3 gene Proteins 0.000 abstract 1
- 125000000000 cycloalkoxy group Chemical group 0.000 abstract 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 abstract 1
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 abstract 1
- 150000002790 naphthalenes Chemical class 0.000 abstract 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 abstract 1
- 150000003254 radicals Chemical class 0.000 description 25
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 21
- -1 radical thiol Chemical class 0.000 description 21
- 239000000243 solution Substances 0.000 description 20
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 19
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 12
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 11
- WYURNTSHIVDZCO-UHFFFAOYSA-N tetrahydrofuran Substances C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 11
- 108010038912 Retinoid X Receptors Proteins 0.000 description 9
- 102000034527 Retinoid X Receptors Human genes 0.000 description 9
- 239000003795 chemical substances by application Substances 0.000 description 8
- 239000012074 organic phase Substances 0.000 description 8
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 8
- 241001080024 Telles Species 0.000 description 7
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 6
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 239000002253 acid Substances 0.000 description 6
- 239000012230 colorless oil Substances 0.000 description 6
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 6
- 239000000377 silicon dioxide Substances 0.000 description 6
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 5
- 238000010908 decantation Methods 0.000 description 5
- 150000002148 esters Chemical class 0.000 description 5
- 238000003818 flash chromatography Methods 0.000 description 5
- 230000002018 overexpression Effects 0.000 description 5
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 125000006239 protecting group Chemical group 0.000 description 5
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 4
- GVJHHUAWPYXKBD-IEOSBIPESA-N α-tocopherol Chemical compound OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- 206010037211 Psychomotor hyperactivity Diseases 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 229940082150 encore Drugs 0.000 description 3
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 3
- 235000019341 magnesium sulphate Nutrition 0.000 description 3
- 238000000034 method Methods 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 239000003921 oil Substances 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 239000012429 reaction media Substances 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 3
- ZKZMJYZYHYWFHN-HMAPJEAMSA-N (z)-3-[3-[5-[tert-butyl(dimethyl)silyl]oxypentoxy]-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-yl]but-2-enal Chemical compound CC1(C)CCC(C)(C)C2=C1C=C(OCCCCCO[Si](C)(C)C(C)(C)C)C(C(=C\C=O)/C)=C2 ZKZMJYZYHYWFHN-HMAPJEAMSA-N 0.000 description 2
- ZIMGGGWCDYVHOY-UHFFFAOYSA-N 3-hydroxy-2-imino-6-(1-piperidinyl)-4-pyrimidinamine Chemical compound N=C1N(O)C(N)=CC(N2CCCCC2)=N1 ZIMGGGWCDYVHOY-UHFFFAOYSA-N 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 2
- ULGZDMOVFRHVEP-RWJQBGPGSA-N Erythromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@@](C)(O)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 ULGZDMOVFRHVEP-RWJQBGPGSA-N 0.000 description 2
- 241000725303 Human immunodeficiency virus Species 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 2
- 102000004310 Ion Channels Human genes 0.000 description 2
- ZFMITUMMTDLWHR-UHFFFAOYSA-N Minoxidil Chemical compound NC1=[N+]([O-])C(N)=CC(N2CCCCC2)=N1 ZFMITUMMTDLWHR-UHFFFAOYSA-N 0.000 description 2
- CXOFVDLJLONNDW-UHFFFAOYSA-N Phenytoin Chemical compound N1C(=O)NC(=O)C1(C=1C=CC=CC=1)C1=CC=CC=C1 CXOFVDLJLONNDW-UHFFFAOYSA-N 0.000 description 2
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 2
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 description 2
- 125000000746 allylic group Chemical group 0.000 description 2
- 150000001280 alpha hydroxy acids Chemical group 0.000 description 2
- 150000004716 alpha keto acids Chemical class 0.000 description 2
- 229940087168 alpha tocopherol Drugs 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- SIPUZPBQZHNSDW-UHFFFAOYSA-N bis(2-methylpropyl)aluminum Chemical compound CC(C)C[Al]CC(C)C SIPUZPBQZHNSDW-UHFFFAOYSA-N 0.000 description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 2
- 229910052794 bromium Inorganic materials 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 108010011716 cellular retinoic acid binding protein II Proteins 0.000 description 2
- 238000013270 controlled release Methods 0.000 description 2
- GDLBFKVLRPITMI-UHFFFAOYSA-N diazoxide Chemical compound ClC1=CC=C2NC(C)=NS(=O)(=O)C2=C1 GDLBFKVLRPITMI-UHFFFAOYSA-N 0.000 description 2
- GUVUOGQBMYCBQP-UHFFFAOYSA-N dmpu Chemical compound CN1CCCN(C)C1=O GUVUOGQBMYCBQP-UHFFFAOYSA-N 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 230000014509 gene expression Effects 0.000 description 2
- 229910052736 halogen Inorganic materials 0.000 description 2
- 239000005556 hormone Substances 0.000 description 2
- 229940088597 hormone Drugs 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- RCBVKBFIWMOMHF-UHFFFAOYSA-L hydroxy-(hydroxy(dioxo)chromio)oxy-dioxochromium;pyridine Chemical compound C1=CC=NC=C1.C1=CC=NC=C1.O[Cr](=O)(=O)O[Cr](O)(=O)=O RCBVKBFIWMOMHF-UHFFFAOYSA-L 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- OVEHNNQXLPJPPL-UHFFFAOYSA-N lithium;n-propan-2-ylpropan-2-amine Chemical compound [Li].CC(C)NC(C)C OVEHNNQXLPJPPL-UHFFFAOYSA-N 0.000 description 2
- 239000004005 microsphere Substances 0.000 description 2
- 229960003632 minoxidil Drugs 0.000 description 2
- 239000002077 nanosphere Substances 0.000 description 2
- BDJRBEYXGGNYIS-UHFFFAOYSA-N nonanedioic acid Chemical compound OC(=O)CCCCCCCC(O)=O BDJRBEYXGGNYIS-UHFFFAOYSA-N 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 235000011181 potassium carbonates Nutrition 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 238000001403 relative X-ray reflectometry Methods 0.000 description 2
- 229930002330 retinoic acid Natural products 0.000 description 2
- 239000012312 sodium hydride Substances 0.000 description 2
- 229910000104 sodium hydride Inorganic materials 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 229960000984 tocofersolan Drugs 0.000 description 2
- QAIPRVGONGVQAS-DUXPYHPUSA-N trans-caffeic acid Chemical compound OC(=O)\C=C\C1=CC=C(O)C(O)=C1 QAIPRVGONGVQAS-DUXPYHPUSA-N 0.000 description 2
- 230000007704 transition Effects 0.000 description 2
- 229960001727 tretinoin Drugs 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 235000019166 vitamin D Nutrition 0.000 description 2
- 239000011710 vitamin D Substances 0.000 description 2
- 235000004835 α-tocopherol Nutrition 0.000 description 2
- 239000002076 α-tocopherol Substances 0.000 description 2
- XMAYWYJOQHXEEK-OZXSUGGESA-N (2R,4S)-ketoconazole Chemical compound C1CN(C(=O)C)CCN1C(C=C1)=CC=C1OC[C@@H]1O[C@@](CN2C=NC=C2)(C=2C(=CC(Cl)=CC=2)Cl)OC1 XMAYWYJOQHXEEK-OZXSUGGESA-N 0.000 description 1
- GJWJBXMTBMFERZ-RZLPOJDTSA-N (2e,4e,6z)-7-[3-(5-hydroxypentoxy)-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-yl]-3-methylocta-2,4,6-trienoic acid Chemical compound CC1(C)CCC(C)(C)C2=C1C=C(OCCCCCO)C(C(\C)=C/C=C/C(/C)=C/C(O)=O)=C2 GJWJBXMTBMFERZ-RZLPOJDTSA-N 0.000 description 1
- SSSTZBUOUJTGKG-RWGNZMNTSA-N (2e,4e,6z)-7-[3-[5-[tert-butyl(dimethyl)silyl]oxypentoxy]-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-yl]-3-methylocta-2,4,6-trienoic acid Chemical compound CC1(C)CCC(C)(C)C2=C1C=C(OCCCCCO[Si](C)(C)C(C)(C)C)C(C(\C)=C/C=C/C(/C)=C/C(O)=O)=C2 SSSTZBUOUJTGKG-RWGNZMNTSA-N 0.000 description 1
- ACEAELOMUCBPJP-UHFFFAOYSA-N (E)-3,4,5-trihydroxycinnamic acid Natural products OC(=O)C=CC1=CC(O)=C(O)C(O)=C1 ACEAELOMUCBPJP-UHFFFAOYSA-N 0.000 description 1
- PJDMKDZLTZAYHO-UZYVYHOESA-N (Z)-3-[3-[5-[tert-butyl(dimethyl)silyl]oxypentoxy]-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-yl]but-2-enoic acid Chemical compound CC1(C)CCC(C)(C)C2=C1C=C(OCCCCCO[Si](C)(C)C(C)(C)C)C(C(=C\C(O)=O)/C)=C2 PJDMKDZLTZAYHO-UZYVYHOESA-N 0.000 description 1
- KZKRRZFCAYOXQE-UHFFFAOYSA-N 1$l^{2}-azinane Chemical compound C1CC[N]CC1 KZKRRZFCAYOXQE-UHFFFAOYSA-N 0.000 description 1
- GMRQFYUYWCNGIN-ZVUFCXRFSA-N 1,25-dihydroxy vitamin D3 Chemical compound C1([C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@@H](CCCC(C)(C)O)C)=CC=C1C[C@@H](O)C[C@H](O)C1=C GMRQFYUYWCNGIN-ZVUFCXRFSA-N 0.000 description 1
- PNCWHIAZZSDHPU-UHFFFAOYSA-N 2-benzylsulfanylethanamine Chemical compound NCCSCC1=CC=CC=C1 PNCWHIAZZSDHPU-UHFFFAOYSA-N 0.000 description 1
- YWEGYQWOTPKCMT-UHFFFAOYSA-N 3-bromo-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-ol Chemical compound BrC1=C(O)C=C2C(C)(C)CCC(C)(C)C2=C1 YWEGYQWOTPKCMT-UHFFFAOYSA-N 0.000 description 1
- NNHUDXYYPUFACA-UHFFFAOYSA-N 5-[(3-bromo-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-yl)oxy]pentan-1-ol Chemical compound OCCCCCOC1=C(Br)C=C2C(C)(C)CCC(C)(C)C2=C1 NNHUDXYYPUFACA-UHFFFAOYSA-N 0.000 description 1
- IWYIIFZUBDNBLG-UHFFFAOYSA-N 5-[(3-bromo-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-yl)oxy]pentoxy-tert-butyl-dimethylsilane Chemical compound CC1(C)CCC(C)(C)C2=C1C=C(Br)C(OCCCCCO[Si](C)(C)C(C)(C)C)=C2 IWYIIFZUBDNBLG-UHFFFAOYSA-N 0.000 description 1
- LQNRCMLZZMZVCZ-UHFFFAOYSA-N 5-[(3-bromo-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-yl)oxy]pentyl acetate Chemical compound CC1(C)CCC(C)(C)C2=C1C=C(Br)C(OCCCCCOC(=O)C)=C2 LQNRCMLZZMZVCZ-UHFFFAOYSA-N 0.000 description 1
- JIHJLQWUVZUKCH-UHFFFAOYSA-N 5-bromopentyl acetate Chemical compound CC(=O)OCCCCCBr JIHJLQWUVZUKCH-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 239000004342 Benzoyl peroxide Substances 0.000 description 1
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 1
- NLZUEZXRPGMBCV-UHFFFAOYSA-N Butylhydroxytoluene Chemical compound CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NLZUEZXRPGMBCV-UHFFFAOYSA-N 0.000 description 1
- UUNFIJJROXBJPD-PYCFMQQDSA-N C/C(=C/C(=O)O)/C1=CC2=C(C=C1OCCCCCO[Si](C)(C)CC(C)(C)C)C(CCC2(C)C)(C)C Chemical compound C/C(=C/C(=O)O)/C1=CC2=C(C=C1OCCCCCO[Si](C)(C)CC(C)(C)C)C(CCC2(C)C)(C)C UUNFIJJROXBJPD-PYCFMQQDSA-N 0.000 description 1
- 0 CCOC(C=C(C)C=CC=C(C)[C@@]1C=C(C(C)(C)CCC2(C)C)C2=CC1OC**O)=O Chemical compound CCOC(C=C(C)C=CC=C(C)[C@@]1C=C(C(C)(C)CCC2(C)C)C2=CC1OC**O)=O 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 201000009030 Carcinoma Diseases 0.000 description 1
- 102100038503 Cellular retinoic acid-binding protein 1 Human genes 0.000 description 1
- 102100038504 Cellular retinoic acid-binding protein 2 Human genes 0.000 description 1
- ACTIUHUUMQJHFO-UHFFFAOYSA-N Coenzym Q10 Natural products COC1=C(OC)C(=O)C(CC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)C)=C(C)C1=O ACTIUHUUMQJHFO-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 101001099865 Homo sapiens Cellular retinoic acid-binding protein 1 Proteins 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 206010020880 Hypertrophy Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 150000000996 L-ascorbic acids Chemical class 0.000 description 1
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 1
- 241000701076 Macacine alphaherpesvirus 1 Species 0.000 description 1
- 239000004909 Moisturizer Substances 0.000 description 1
- FNJSWIPFHMKRAT-UHFFFAOYSA-N Monomethyl phthalate Chemical compound COC(=O)C1=CC=CC=C1C(O)=O FNJSWIPFHMKRAT-UHFFFAOYSA-N 0.000 description 1
- 229930193140 Neomycin Natural products 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 101100030361 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) pph-3 gene Proteins 0.000 description 1
- JCXJVPUVTGWSNB-UHFFFAOYSA-N Nitrogen dioxide Chemical compound O=[N]=O JCXJVPUVTGWSNB-UHFFFAOYSA-N 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- 229920002565 Polyethylene Glycol 400 Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 208000006994 Precancerous Conditions Diseases 0.000 description 1
- GBFLZEXEOZUWRN-VKHMYHEASA-N S-carboxymethyl-L-cysteine Chemical compound OC(=O)[C@@H](N)CSCC(O)=O GBFLZEXEOZUWRN-VKHMYHEASA-N 0.000 description 1
- 102000019197 Superoxide Dismutase Human genes 0.000 description 1
- 108010012715 Superoxide dismutase Proteins 0.000 description 1
- 239000004098 Tetracycline Substances 0.000 description 1
- AUYYCJSJGJYCDS-LBPRGKRZSA-N Thyrolar Chemical class IC1=CC(C[C@H](N)C(O)=O)=CC(I)=C1OC1=CC=C(O)C(I)=C1 AUYYCJSJGJYCDS-LBPRGKRZSA-N 0.000 description 1
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 1
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 description 1
- 101001099854 Xenopus laevis Cellular retinoic acid-binding protein 2 Proteins 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- XDNRTTCEIPCXNN-UHFFFAOYSA-N [3-[5-[tert-butyl(dimethyl)silyl]oxypentoxy]-5,5,8,8-tetramethyl-6,7-dihydronaphthalen-2-yl]boronic acid Chemical compound CC1(C)CCC(C)(C)C2=C1C=C(B(O)O)C(OCCCCCO[Si](C)(C)C(C)(C)C)=C2 XDNRTTCEIPCXNN-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- OENHQHLEOONYIE-UKMVMLAPSA-N all-trans beta-carotene Natural products CC=1CCCC(C)(C)C=1/C=C/C(/C)=C/C=C/C(/C)=C/C=C/C=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C OENHQHLEOONYIE-UKMVMLAPSA-N 0.000 description 1
- 230000000172 allergic effect Effects 0.000 description 1
- 210000004102 animal cell Anatomy 0.000 description 1
- NUZWLKWWNNJHPT-UHFFFAOYSA-N anthralin Chemical compound C1C2=CC=CC(O)=C2C(=O)C2=C1C=CC=C2O NUZWLKWWNNJHPT-UHFFFAOYSA-N 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000003255 anti-acne Effects 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 230000002682 anti-psoriatic effect Effects 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960002255 azelaic acid Drugs 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 235000019400 benzoyl peroxide Nutrition 0.000 description 1
- TUPZEYHYWIEDIH-WAIFQNFQSA-N beta-carotene Natural products CC(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/C1=C(C)CCCC1(C)C)C=CC=C(/C)C=CC2=CCCCC2(C)C TUPZEYHYWIEDIH-WAIFQNFQSA-N 0.000 description 1
- 235000013734 beta-carotene Nutrition 0.000 description 1
- 239000011648 beta-carotene Substances 0.000 description 1
- 229960002747 betacarotene Drugs 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- CZBZUDVBLSSABA-UHFFFAOYSA-N butylated hydroxyanisole Chemical compound COC1=CC=C(O)C(C(C)(C)C)=C1.COC1=CC=C(O)C=C1C(C)(C)C CZBZUDVBLSSABA-UHFFFAOYSA-N 0.000 description 1
- 229940074360 caffeic acid Drugs 0.000 description 1
- 235000004883 caffeic acid Nutrition 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 235000021466 carotenoid Nutrition 0.000 description 1
- 150000001747 carotenoids Chemical class 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 230000030833 cell death Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 108010012671 cellular retinoic acid binding protein I Proteins 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- QAIPRVGONGVQAS-UHFFFAOYSA-N cis-caffeic acid Natural products OC(=O)C=CC1=CC=C(O)C(O)=C1 QAIPRVGONGVQAS-UHFFFAOYSA-N 0.000 description 1
- 229960002227 clindamycin Drugs 0.000 description 1
- KDLRVYVGXIQJDK-AWPVFWJPSA-N clindamycin Chemical compound CN1C[C@H](CCC)C[C@H]1C(=O)N[C@H]([C@H](C)Cl)[C@@H]1[C@H](O)[C@H](O)[C@@H](O)[C@@H](SC)O1 KDLRVYVGXIQJDK-AWPVFWJPSA-N 0.000 description 1
- 235000017471 coenzyme Q10 Nutrition 0.000 description 1
- ACTIUHUUMQJHFO-UPTCCGCDSA-N coenzyme Q10 Chemical compound COC1=C(OC)C(=O)C(C\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CC\C=C(/C)CCC=C(C)C)=C(C)C1=O ACTIUHUUMQJHFO-UPTCCGCDSA-N 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- WZHCOOQXZCIUNC-UHFFFAOYSA-N cyclandelate Chemical compound C1C(C)(C)CC(C)CC1OC(=O)C(O)C1=CC=CC=C1 WZHCOOQXZCIUNC-UHFFFAOYSA-N 0.000 description 1
- 238000006264 debenzylation reaction Methods 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- 229960004042 diazoxide Drugs 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 229960002311 dithranol Drugs 0.000 description 1
- 230000003828 downregulation Effects 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 239000003974 emollient agent Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 230000002124 endocrine Effects 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 208000010932 epithelial neoplasm Diseases 0.000 description 1
- 229960003276 erythromycin Drugs 0.000 description 1
- 229940011871 estrogen Drugs 0.000 description 1
- 239000000262 estrogen Substances 0.000 description 1
- GNCUVFPHYFZIRH-UHFFFAOYSA-N ethyl 6-methylpyridazine-3-carboxylate Chemical compound CCOC(=O)C1=CC=C(C)N=N1 GNCUVFPHYFZIRH-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 239000003889 eye drop Substances 0.000 description 1
- 229940012356 eye drops Drugs 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 239000000833 heterodimer Substances 0.000 description 1
- 230000003054 hormonal effect Effects 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 229960004337 hydroquinone Drugs 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229960004125 ketoconazole Drugs 0.000 description 1
- BEJNERDRQOWKJM-UHFFFAOYSA-N kojic acid Chemical compound OCC1=CC(=O)C(O)=CO1 BEJNERDRQOWKJM-UHFFFAOYSA-N 0.000 description 1
- 229960004705 kojic acid Drugs 0.000 description 1
- WZNJWVWKTVETCG-UHFFFAOYSA-N kojic acid Natural products OC(=O)C(N)CN1C=CC(=O)C(O)=C1 WZNJWVWKTVETCG-UHFFFAOYSA-N 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 230000001333 moisturizer Effects 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- 229960004927 neomycin Drugs 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 229960002036 phenytoin Drugs 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 230000001185 psoriatic effect Effects 0.000 description 1
- NPCOQXAVBJJZBQ-UHFFFAOYSA-N reduced coenzyme Q9 Natural products COC1=C(O)C(C)=C(CC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)CCC=C(C)C)C(O)=C1OC NPCOQXAVBJJZBQ-UHFFFAOYSA-N 0.000 description 1
- 150000004492 retinoid derivatives Chemical class 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 238000007127 saponification reaction Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 239000002453 shampoo Substances 0.000 description 1
- 208000017520 skin disease Diseases 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 230000003637 steroidlike Effects 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229940032362 superoxide dismutase Drugs 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- BCNZYOJHNLTNEZ-UHFFFAOYSA-N tert-butyldimethylsilyl chloride Chemical compound CC(C)(C)[Si](C)(C)Cl BCNZYOJHNLTNEZ-UHFFFAOYSA-N 0.000 description 1
- 235000019364 tetracycline Nutrition 0.000 description 1
- 150000003522 tetracyclines Chemical class 0.000 description 1
- 229940040944 tetracyclines Drugs 0.000 description 1
- 239000005495 thyroid hormone Substances 0.000 description 1
- 229940036555 thyroid hormone Drugs 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- CIWZUQUKZAMSIZ-UHFFFAOYSA-N trimethoxy borate Chemical compound COOB(OOC)OOC CIWZUQUKZAMSIZ-UHFFFAOYSA-N 0.000 description 1
- AALUTIYNYXEFNT-UHFFFAOYSA-N trimethylsilane hydroiodide Chemical compound C[SiH](C)C.I AALUTIYNYXEFNT-UHFFFAOYSA-N 0.000 description 1
- 229940035936 ubiquinone Drugs 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- MECHNRXZTMCUDQ-RKHKHRCZSA-N vitamin D2 Chemical class C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)/C=C/[C@H](C)C(C)C)=C\C=C1\C[C@@H](O)CCC1=C MECHNRXZTMCUDQ-RKHKHRCZSA-N 0.000 description 1
- 235000005282 vitamin D3 Nutrition 0.000 description 1
- 239000011647 vitamin D3 Substances 0.000 description 1
- QYSXJUFSXHHAJI-YRZJJWOYSA-N vitamin D3 Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C\C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-YRZJJWOYSA-N 0.000 description 1
- 229940021056 vitamin d3 Drugs 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- OENHQHLEOONYIE-JLTXGRSLSA-N β-Carotene Chemical compound CC=1CCCC(C)(C)C=1\C=C\C(\C)=C\C=C\C(\C)=C\C=C\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C OENHQHLEOONYIE-JLTXGRSLSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/66—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety
- C07C69/73—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of unsaturated acids
- C07C69/734—Ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/02—Nutrients, e.g. vitamins, minerals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/20—Antivirals for DNA viruses
- A61P31/22—Antivirals for DNA viruses for herpes viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C47/00—Compounds having —CHO groups
- C07C47/20—Unsaturated compounds having —CHO groups bound to acyclic carbon atoms
- C07C47/277—Unsaturated compounds having —CHO groups bound to acyclic carbon atoms containing ether groups, groups, groups, or groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/58—Unsaturated compounds containing ether groups, groups, groups, or groups
- C07C59/72—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings and other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/10—One of the condensed rings being a six-membered aromatic ring the other ring being six-membered, e.g. tetraline
Definitions
- the present invention relates to new modulator compounds families of cellular hormonal receptors such as those described by Guiguère or Evans (Guiguère V., Endocrine Reviews, 15.1, 1994.61; Mangelsdorf D. & Evans R., Cell, 83, 1995, 841).
- these new compounds exert biological activity on animal cells and tissues with regard to their differentiation, their proliferation or their implication in cell death (apoptosis).
- These substances act in combination with known ligands from the super family, for example retinoic acid and retinoids, vitamin D3, thyroid hormone, estrogen, etc., so as to improve their risk / benefit ratio, both by reducing their toxicity and by improving their pharmacological efficacy.
- the compounds according to the invention allow the prevention and / or the treatment of disorders and / or conditions linked to overregulation of receptors related to these hormones or vitamins. They may also be involved in the control of the different stages of the metabolism or catabolism of these hormones or vitamins.
- salts these are preferably salts of an alkali or alkaline earth metal or still zinc or an organic amine.
- lower alkyl radical means a radical having from 1 to 6 carbon atoms, linear or branched, preferably the methyl, ethyl, isopropyl, butyl, tert-butyl and hexyl radicals. This definition also applies to the alkyl residues of the alkoxy radicals.
- cycloalkoxyl is preferably understood according to the invention the radicals the cycloalkyl radical of which consists of a mono group or polycyclic, comprising 3 to 12 carbon atoms, optionally substituted by one or more lower alkyl radicals.
- heterocycle preferably means a piperidino radical, morpholino, pyrrolidino or pizzaino, optionally substituted in position 4 by a lower alkyl radical.
- Y represents a cyano radical (-CN) or a -COR 3 radical, R 3 being defined above, n represents 2, 3 or 4 and o represents 1, 2 or 3.
- Z represents a methylene radical (-CH 2 -)
- Y represents a -thiol (-SH), trimethylammonium (-N + (CH 3 ) 3 ) or cyano radical (-CN).
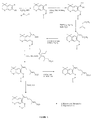
- These compounds of formula (I) can be obtained in particular by reacting a hydroxylated compound of formula (II) in which X and R 2 are defined above, with a radical of formula (III) X- (CH 2 ) n -Z- (CH 2 ) o -Y in which Z, Y, n and o are defined above, and X represents a leaving group, such as a halogen, in particular bromine or iodine.
- the hydroxylated compound of formula (II) is prepared from the derivative corresponding alkoxylated described in patent application DE 28 19 213, in particular the methoxylated derivative described in example 9.
- the hydrolysis of the function ether is produced according to the usual methods of the technique, with protection and appropriate deprotection of functions sensitive to hydrolysis conditions.
- R 2 represents the radical -COOH
- the compounds are prepared by protecting R 2 with a protective group of alkyl, allylic, benzyl or tert-butyl type.
- the phenol of formula is transformed general (II) into a compound of formula (I) with the radical of formula (III) in presence of potassium carbonate (K2CO3) in methyl ether ketone (MEK) or in the presence of sodium hydride in a solvent such as dimethylformamide (DMF).
- K2CO3 potassium carbonate
- MEK methyl ether ketone
- DMF dimethylformamide
- W represents an atom halogen such as bromine or iodine
- ⁇ represents a protective group, such as terbutyldimethylsilane (TBDMSi)
- R the radical - (CH2) n-Z- (CH2) o- and Y is such than in the request.
- Reactions 1 / of Mitsunobu and 2 / of saponification are possibly performed if Y represents an oxygen atom and we want the transform into sulfur or nitrogen atom.
- the examples below illustrate this reaction scheme.
- the present invention also relates to the use of the compounds according to the invention as medicament and cosmetic compositions or pharmaceuticals comprising at least one compound of general formula (I) such as defined above and at least one vehicle cosmetically or pharmaceutically acceptable.
- Cosmetic or pharmaceutical compositions containing at least one compound of formula (I) are in particular intended for the treatment of disorders or conditions linked to overregulation of transcription factors called retinoid receptors which include RARs (Retinoic Acid Receptors), including including the subtypes ⁇ , ⁇ , ⁇ , the Retinoic Receptors X (RXRs), including the subtypes ⁇ , ⁇ , ⁇ , or the genes comprising the response elements RARs and RXRs.
- RARs Retinoic Acid Receptors
- RXRs Retinoic Receptors X
- overregulation of the RARs and / or RXRs receptors it is understood according to the invention an overexpression of the RARs and / or RXRs receptors, and / or a biological overactivity of RARs and / or RXRs receptors.
- the biological overactivity of the RARs and / or RXRs receptors can be due to a chemical modification of the RARs and / or RXRs receptors, but it can may also be due to a factor other than the receiver itself. So the biological overactivity may be due to overexpression of an endogenous gene or to expression of an exogenous gene comprising the RARE response element (retinoic acid response element) on which a heterodimer including the RAR receptor, the latter carrying an agonist ligand.
- RARE response element retinoic acid response element
- an endogenous gene comprising the RARE response element we may include the CRABP II gene (cellular retinoic acid binding protein II), the overexpression has been demonstrated in psoriasis ("Overexpression of CRABP Il and down-regulation of CRABP I in psoriatic skin ", G. Siegenthaler et al., Dermatology 1992; 185: 251-256).
- gene expression exogenous comprising the RARE response element we can cite the HIV-1 genome (human immunodeficiency virus) (Proc. Natl. Acad. Sci. USA, Lee et al., Vol. 91, pp.
- the compounds of formula (I) can be advantageously used in combination with other compounds with activity of retinoid type, with vitamins D or their derivatives, with corticosteroids, with anti-free radicals , ⁇ -hydroxy or ⁇ -keto acids or their derivatives, or alternatively with ion channel blockers.
- vitamins D or their derivatives is meant, for example, the derivatives of vitamin D 2 or D 3 and in particular 1,25-dihydroxyvitamin D 3 .
- anti-free radicals is meant, for example, ⁇ -tocopherol, Super Oxide Dismutase, Ubiquinone or certain metal chelators.
- ⁇ -hydroxy or ⁇ -keto acids or their derivatives is meant for example lactic, malic, citric, glycolic, mandelic, tartaric, glyceric or ascorbic acids or their salts, amides or esters.
- ion channel blockers is meant, for example, Minoxidil (2,4-diamino-6-piperidino-pyrimidine-3-oxide) and its derivatives.
- Cosmetic or pharmaceutical compositions comprising a effective amount of at least one compound of formula (I), one of its analogs chiral or one of its salts comprises a cosmetically or pharmaceutically acceptable and compatible with the mode of administration restrained.
- the effective amount depends of course on the desired treatment and the nature of the compound chosen is therefore determined by a person skilled in the art.
- the administration of the compounds according to the invention can be carried out by enteral, parenteral, topical or ocular.
- the composition can be in the form of tablets, capsules, dragees, syrups, suspensions, solutions, powders, granules, emulsions, microspheres or nanospheres or lipid vesicles or polymers allowing controlled release.
- the composition, more particularly the pharmaceutical composition can be present in the form of solutions or suspensions for infusion or for injection.
- the compounds according to the invention are generally administered to a daily dose of approximately 0.01 mg / kg to 100 mg / kg in body weight, and this at reason for 1 to 3 takes.
- the composition is more particularly intended for treatment of the skin and mucous membranes and can then be in the form ointments, creams, milks, ointments, powders, soaked tampons, solutions, gels, sprays, lotions, suspensions or shampoos. It can also be in the form of microspheres or nanospheres or lipid or polymer vesicles or polymer patches and hydrogels allowing controlled release.
- This topical composition can by elsewhere be present either in anhydrous form or in an aqueous form.
- composition for topical or ocular use contains at least one compound of formula (I) as defined above, or one of its analogs chirals or one of its salts, preferably at a concentration between 0.001% and 5% by weight relative to the total weight of the composition.
- composition according to the invention may also contain additives inert or even pharmacodynamically or cosmetically active or combinations of these additives, and in particular: wetting agents; agents depigmenters such as hydroquinone, azelaic acid, caffeic acid or kojic acid; emollients; moisturizers such as glycerol, PEG 400, thiamorpholinone, and its derivatives or even urea; agents antiseborrhoeic or anti-acne, such as S-carboxymethylcysteine, S-benzyl-cysteamine, their salts or derivatives, or benzoyl peroxide; of antibiotics like erythromycin and its esters, neomycin, clindamycin and its esters, tetracyclines; antifungal agents such as ketoconazole or polymethylene-4,5 isothiazolidones-3; agents promoting regrowth hair, such as Minoxidil (2,4-diamino-6-piperidin
- composition may also contain agents for improving flavor, preservatives such as acid esters parahydroxybenzoic, stabilizing agents, regulating agents humidity, pH regulating agents, pressure modifying agents osmotic, emulsifying agents, UV-A and UV-B filters, antioxidants, such as ⁇ -tocopherol, butylhydroxyanisole or butylhydroxytoluene.
- preservatives such as acid esters parahydroxybenzoic, stabilizing agents, regulating agents humidity, pH regulating agents, pressure modifying agents osmotic, emulsifying agents, UV-A and UV-B filters, antioxidants, such as ⁇ -tocopherol, butylhydroxyanisole or butylhydroxytoluene.
- tert-butyldimethylsilyl chloride (2.64 g) is added to a mixture of 5- (3-bromo-5,5,8 , 8-tetramethyl-5,6,7,8-tetrahydro-naphthalene-2-yloxy) -pentan-1-ol (4.3g, 11.7 mmol) and 80% sodium hydride (422 mg) in THF (20ml). The mixture is stirred at room temperature for 2 h. The solution is poured into a mixture of water and ethyl acetate.
- the organic phase is washed twice with water, dried over magnesium sulfate, and concentrated on a rotary evaporator under vacuum at 40 ° C.
- the product is purified by flash chromatography on a silica column. Yellow oil. Yield: 64%.
- the filtrate is poured into an ethyl ether / water mixture.
- the organic phase is washed with water, dried over anhydrous magnesium sulfate, concentrated on a rotary evaporator under vacuum at 40 ° C.
- the resulting oil is dissolved in 30 ml of dichloromethane and 2 g of pyridinium dichromate (PDC) are added.
- PDC pyridinium dichromate
- the mixture is stirred for 4 hours at room temperature, filtered through silica.
- the filtrate is concentrated on a rotary evaporator under vacuum at 40 ° C and purified by flash chromatography on a silica column. Colorless oil. Yield: 61%.
- the mixture is acidified to pH 1 with a solution of concentrated hydrochloric acid. After decantation, the organic phase is washed twice with water, dried over anhydrous magnesium sulfate and concentrated on a rotary evaporator under vacuum at 40 ° C. Colorless oil. Yield: 100%.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Virology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Hematology (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Nutrition Science (AREA)
- Diabetes (AREA)
- Biotechnology (AREA)
- Obesity (AREA)
- Dermatology (AREA)
- Molecular Biology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Steroid Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Cosmetics (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Furan Compounds (AREA)
- Pyrane Compounds (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR9610686A FR2752837B1 (fr) | 1996-09-02 | 1996-09-02 | Nouveaux composes modulateurs des recepteurs hormonaux, les compositions les comprenant et leur utilisation en therapie |
| FR9610686 | 1996-09-02 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0826657A1 true EP0826657A1 (de) | 1998-03-04 |
| EP0826657B1 EP0826657B1 (de) | 2001-08-01 |
Family
ID=9495359
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP97401999A Expired - Lifetime EP0826657B1 (de) | 1996-09-02 | 1997-08-27 | Verbindungen zur Modulierung von Hormonrezeptoren, diese enthaltende Zusammensetzungen und ihre Verwendung in Therapie |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US5910508A (de) |
| EP (1) | EP0826657B1 (de) |
| JP (1) | JP2910995B2 (de) |
| AT (1) | ATE203743T1 (de) |
| AU (1) | AU703076B2 (de) |
| CA (1) | CA2213609C (de) |
| DE (1) | DE69705902T2 (de) |
| DK (1) | DK0826657T3 (de) |
| ES (1) | ES2163723T3 (de) |
| FR (1) | FR2752837B1 (de) |
| GR (1) | GR3036997T3 (de) |
| PT (1) | PT826657E (de) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009156675A2 (fr) | 2008-05-30 | 2009-12-30 | Galderma Research & Development | Nouvelles compositions dépigmentantes sous forme d'une composition anhydre sans vaseline et sans élastomère comprenant un dérivé phénolique solubilisé et un rétinoïde |
| EP2460562A1 (de) | 2006-12-21 | 2012-06-06 | Galderma Research & Development | Creme-Gel mit mindestens einem Retinoid und Benzoylperoxid |
| EP2460561A1 (de) | 2006-12-21 | 2012-06-06 | Galderma Research & Development | Emulsion mit mindestens einem Retinoid und Benzoylperoxid |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7655699B1 (en) | 1992-04-22 | 2010-02-02 | Eisai Inc. | Compounds having selective activity for retinoid X receptors, and means for modulation of processes mediated by retinoid X receptors |
| TR200200507T2 (tr) | 1999-08-27 | 2002-10-21 | Ligand Pharmaceuticals Inc | Androjen reseptörü modülatör bileşikleri ve metotları |
| US6566372B1 (en) | 1999-08-27 | 2003-05-20 | Ligand Pharmaceuticals Incorporated | Bicyclic androgen and progesterone receptor modulator compounds and methods |
| WO2001016133A2 (en) | 1999-08-27 | 2001-03-08 | Ligand Pharmaceuticals Incorporated | 8-substituted-6-trifluoromethyl-9-pyrido[3,2-g]quinoline compounds as androgen receptor modulators |
| WO2001019770A2 (en) * | 1999-09-14 | 2001-03-22 | Ligand Pharmaceuticals Incorporated | Rxr modulators with improved pharmacologic profile |
| US8026280B2 (en) | 2001-03-27 | 2011-09-27 | Errant Gene Therapeutics, Llc | Histone deacetylase inhibitors |
| US7312247B2 (en) * | 2001-03-27 | 2007-12-25 | Errant Gene Therapeutics, Llc | Histone deacetylase inhibitors |
| US7842727B2 (en) * | 2001-03-27 | 2010-11-30 | Errant Gene Therapeutics, Llc | Histone deacetylase inhibitors |
| US6495719B2 (en) | 2001-03-27 | 2002-12-17 | Circagen Pharmaceutical | Histone deacetylase inhibitors |
| CA2486303C (en) * | 2002-05-22 | 2013-04-30 | Errant Gene Therapeutics, Llc | Histone deacetylase inhibitors based on alpha-ketoepoxide compounds |
| CA2506504A1 (en) * | 2002-11-20 | 2004-06-03 | Errant Gene Therapeutics, Llc | Treatment of lung cells with histone deacetylase inhibitors |
| WO2006052916A2 (en) * | 2004-11-08 | 2006-05-18 | Errant Gene Therapeutics, Inc. | Histone deacetylase inhibitors |
| EP2742350B1 (de) | 2011-08-08 | 2019-10-30 | The Coca-Cola Company | Zelllinien mit endogenen geschmacksrezptoren und deren verwendungen |
| KR102170307B1 (ko) * | 2012-11-15 | 2020-10-26 | 주식회사 알엔에스 | 탈모방지 및 모발성장 촉진제 조성물 |
| JP2021503464A (ja) * | 2017-11-17 | 2021-02-12 | アイオー セラピューティクス インコーポレイテッド | レチノイドx受容体特異的なレチノイドの調製のための化合物および合成方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2390428A1 (fr) * | 1977-05-04 | 1978-12-08 | Hoffmann La Roche | Nouveaux composes polyeniques, leur preparation et leur application en tant que medicaments |
| EP0568898A1 (de) * | 1992-05-07 | 1993-11-10 | F. Hoffmann-La Roche Ag | Benzothiepin-, Benzothiopyran- und Benzothiophenderivate, deren Herstellung und Verwendung als Heilmittel |
| WO1995004036A1 (en) * | 1993-01-11 | 1995-02-09 | Ligand Pharmaceuticals Inc. | Compounds having selective activity for retinoid x receptors, and means for modulation of processes mediated by retinoid x receptors |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU626881B2 (en) * | 1988-07-14 | 1992-08-13 | F. Hoffmann-La Roche Ag | Benzofused heterocyclics used as pharmaceuticals |
| EP0983992B1 (de) * | 1992-04-22 | 2005-10-26 | Ligand Pharmaceuticals Incorporated | Verbindungen mit Retinoid x rezeptorselektivität |
-
1996
- 1996-09-02 FR FR9610686A patent/FR2752837B1/fr not_active Expired - Fee Related
-
1997
- 1997-08-27 ES ES97401999T patent/ES2163723T3/es not_active Expired - Lifetime
- 1997-08-27 DE DE69705902T patent/DE69705902T2/de not_active Expired - Fee Related
- 1997-08-27 EP EP97401999A patent/EP0826657B1/de not_active Expired - Lifetime
- 1997-08-27 PT PT97401999T patent/PT826657E/pt unknown
- 1997-08-27 AT AT97401999T patent/ATE203743T1/de not_active IP Right Cessation
- 1997-08-27 DK DK97401999T patent/DK0826657T3/da active
- 1997-08-28 AU AU36059/97A patent/AU703076B2/en not_active Ceased
- 1997-08-29 CA CA002213609A patent/CA2213609C/fr not_active Expired - Fee Related
- 1997-09-01 JP JP9236309A patent/JP2910995B2/ja not_active Expired - Fee Related
- 1997-09-02 US US08/921,591 patent/US5910508A/en not_active Expired - Fee Related
-
2001
- 2001-10-24 GR GR20010401870T patent/GR3036997T3/el not_active IP Right Cessation
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2390428A1 (fr) * | 1977-05-04 | 1978-12-08 | Hoffmann La Roche | Nouveaux composes polyeniques, leur preparation et leur application en tant que medicaments |
| EP0568898A1 (de) * | 1992-05-07 | 1993-11-10 | F. Hoffmann-La Roche Ag | Benzothiepin-, Benzothiopyran- und Benzothiophenderivate, deren Herstellung und Verwendung als Heilmittel |
| WO1995004036A1 (en) * | 1993-01-11 | 1995-02-09 | Ligand Pharmaceuticals Inc. | Compounds having selective activity for retinoid x receptors, and means for modulation of processes mediated by retinoid x receptors |
Non-Patent Citations (1)
| Title |
|---|
| KOCH S S C ET AL: "Identification of the First Retinoid X Receptor Homodimer Antagonist", J. MED. CHEM. (JMCMAR,00222623);96; VOL.39 (17); PP.3229-3234, LIGAND PHARMACEUTICALS INC.;DEPARTMENT OF RETINOID CHEMISTRY RESEARCH; SAN DIEGO; 92121; CA; USA (US), XP002030143 * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2460562A1 (de) | 2006-12-21 | 2012-06-06 | Galderma Research & Development | Creme-Gel mit mindestens einem Retinoid und Benzoylperoxid |
| EP2460561A1 (de) | 2006-12-21 | 2012-06-06 | Galderma Research & Development | Emulsion mit mindestens einem Retinoid und Benzoylperoxid |
| EP3025763A1 (de) | 2006-12-21 | 2016-06-01 | Galderma Research & Development | Cremegel, das mindestens ein retinoid und benzoylperoxid umfasst |
| WO2009156675A2 (fr) | 2008-05-30 | 2009-12-30 | Galderma Research & Development | Nouvelles compositions dépigmentantes sous forme d'une composition anhydre sans vaseline et sans élastomère comprenant un dérivé phénolique solubilisé et un rétinoïde |
Also Published As
| Publication number | Publication date |
|---|---|
| ATE203743T1 (de) | 2001-08-15 |
| JPH10101641A (ja) | 1998-04-21 |
| AU3605997A (en) | 1998-03-05 |
| US5910508A (en) | 1999-06-08 |
| FR2752837B1 (fr) | 1999-11-12 |
| CA2213609A1 (fr) | 1998-03-02 |
| GR3036997T3 (en) | 2002-01-31 |
| JP2910995B2 (ja) | 1999-06-23 |
| DE69705902T2 (de) | 2001-11-15 |
| ES2163723T3 (es) | 2002-02-01 |
| CA2213609C (fr) | 2002-06-25 |
| DK0826657T3 (da) | 2001-10-22 |
| FR2752837A1 (fr) | 1998-03-06 |
| AU703076B2 (en) | 1999-03-11 |
| PT826657E (pt) | 2001-12-28 |
| EP0826657B1 (de) | 2001-08-01 |
| DE69705902D1 (de) | 2001-09-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0826657B1 (de) | Verbindungen zur Modulierung von Hormonrezeptoren, diese enthaltende Zusammensetzungen und ihre Verwendung in Therapie | |
| CA2224528C (fr) | Composes stilbeniques a groupement adamantyl, compositions les contenant et utilisations | |
| CA2147806C (fr) | Composes polyeniques, compositions pharmaceutiques et cosmetiques les contenant et utilisations | |
| FR2733684A1 (fr) | Utilisation de retinoides dans une composition cosmetique ou pour la fabrication d'une composition pharmaceutique | |
| FR2763588A1 (fr) | Composes triaromatiques, compositions les contenant et utilisations | |
| EP0915823B1 (de) | Bisaromatische verbindungen, sie enthaltende pharmazeutische und kosmetische zusammensetzungen und verwendungen | |
| CA2191790C (fr) | Composes biaromatiques portant un groupement adamantyl en para, compositions pharmaceutiques et cosmetiques les contenant et utilisations de ces compositions | |
| EP0832081A1 (de) | Bicyclische aromatische verbindungen | |
| EP0832057B1 (de) | Diaromatische propynyl- oder dienylverbindungen | |
| FR2776657A1 (fr) | Composes bicycliques-aromatiques et leur utilisation en medecine humaine ou veterinaire ainsi qu'en cosmetologie | |
| EP0977749B1 (de) | Bi-aromatische verbindungen und diese enthaltende pharmazeutische und kosmetische zusammenstellungen | |
| LU86345A1 (fr) | Nouveaux derives benzofuranniques,leurs procedes de preparation et compositions medicamenteuse et cosmetique les contenant | |
| EP0816352A1 (de) | Biaryl heterocyclische Verbindungen und ihre Verwendung in der Human- oder Tiermedizin und in der Kosmetik | |
| EP1086080B1 (de) | Diarylselenid-verbindungen und ihre verwendung in menschliche oder tierische medizin wie in kosmetologie | |
| EP0986537B1 (de) | Hetero-ethynyl-verbindungen und diese enthaltende zusammensetzungen und kosmetika | |
| WO1998024778A1 (fr) | Derives d'acides benzofuranne-acryliques et leur utilisation comme modulateurs des recepteurs rxrs ou rars | |
| EP0934295B1 (de) | Bioaromatische dibenzofuranderivate und verwendung in der human- oder tiermedizin und in der kosmetik | |
| EP0952975A1 (de) | Eine heteroethynylen-brücke enthaltende bi-aromatische verbindungen und diese enthaltende pharmazeutische und kosmetische zusammensetzungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| AX | Request for extension of the european patent |
Free format text: AL;LT;LV;RO;SI |
|
| DAX | Request for extension of the european patent (deleted) | ||
| 17P | Request for examination filed |
Effective date: 19980131 |
|
| AKX | Designation fees paid |
Free format text: AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| 17Q | First examination report despatched |
Effective date: 19990510 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE |
|
| REF | Corresponds to: |
Ref document number: 203743 Country of ref document: AT Date of ref document: 20010815 Kind code of ref document: T |
|
| ITF | It: translation for a ep patent filed | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20010827 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: FRENCH |
|
| REF | Corresponds to: |
Ref document number: 69705902 Country of ref document: DE Date of ref document: 20010906 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 |
|
| GBT | Gb: translation of ep patent filed (gb section 77(6)(a)/1977) |
Effective date: 20011101 |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Free format text: AVAILABILITY OF NATIONAL TRANSLATION Effective date: 20011008 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20010401870 Country of ref document: GR |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2163723 Country of ref document: ES Kind code of ref document: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020301 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20040803 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20040806 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20040810 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IE Payment date: 20040811 Year of fee payment: 8 Ref country code: AT Payment date: 20040811 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FI Payment date: 20040812 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DK Payment date: 20040813 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20040825 Year of fee payment: 8 Ref country code: ES Payment date: 20040825 Year of fee payment: 8 Ref country code: PT Payment date: 20040825 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GR Payment date: 20040827 Year of fee payment: 8 Ref country code: CH Payment date: 20040827 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20040902 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20041021 Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 20050827 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050827 Ref country code: FI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050827 Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050827 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050828 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050829 Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050829 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050831 Ref country code: DK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050831 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050831 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050831 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060227 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060301 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060301 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060302 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP |
|
| EUG | Se: european patent has lapsed | ||
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20050827 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20060428 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 20060301 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20060428 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20050829 |
|
| BERE | Be: lapsed |
Owner name: CENTRE INTERNATIONAL DE RECHERCHES DERMATOLOGIQUES Effective date: 20050831 |