EP0390142B1 - Verfahren zum Herstellen kornorientierter Elektrobleche mit hoher magnetischer Flussdichte - Google Patents
Verfahren zum Herstellen kornorientierter Elektrobleche mit hoher magnetischer Flussdichte Download PDFInfo
- Publication number
- EP0390142B1 EP0390142B1 EP19900106018 EP90106018A EP0390142B1 EP 0390142 B1 EP0390142 B1 EP 0390142B1 EP 19900106018 EP19900106018 EP 19900106018 EP 90106018 A EP90106018 A EP 90106018A EP 0390142 B1 EP0390142 B1 EP 0390142B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- primary
- recrystallization
- annealing
- final
- temperature
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 230000004907 flux Effects 0.000 title claims description 32
- 238000000034 method Methods 0.000 title claims description 26
- 230000008569 process Effects 0.000 title claims description 25
- 229910001224 Grain-oriented electrical steel Inorganic materials 0.000 title claims description 9
- 238000001953 recrystallisation Methods 0.000 claims description 48
- 238000000137 annealing Methods 0.000 claims description 43
- 238000010438 heat treatment Methods 0.000 claims description 41
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 38
- 229910000831 Steel Inorganic materials 0.000 claims description 34
- 239000010959 steel Substances 0.000 claims description 34
- 238000005121 nitriding Methods 0.000 claims description 28
- 229910052757 nitrogen Inorganic materials 0.000 claims description 25
- 238000005097 cold rolling Methods 0.000 claims description 23
- 230000009467 reduction Effects 0.000 claims description 22
- 229910052710 silicon Inorganic materials 0.000 claims description 12
- 239000012535 impurity Substances 0.000 claims description 8
- 238000000746 purification Methods 0.000 claims description 7
- 239000012467 final product Substances 0.000 claims description 6
- 238000005098 hot rolling Methods 0.000 claims description 6
- 229910052742 iron Inorganic materials 0.000 claims description 6
- 229910052782 aluminium Inorganic materials 0.000 claims description 3
- 230000014509 gene expression Effects 0.000 claims description 2
- 239000003112 inhibitor Substances 0.000 description 26
- 239000000047 product Substances 0.000 description 16
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 12
- 239000002244 precipitate Substances 0.000 description 12
- 229910052717 sulfur Inorganic materials 0.000 description 8
- 239000013078 crystal Substances 0.000 description 6
- 238000005261 decarburization Methods 0.000 description 6
- 229910052748 manganese Inorganic materials 0.000 description 6
- 239000011572 manganese Substances 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 238000002474 experimental method Methods 0.000 description 5
- 238000004519 manufacturing process Methods 0.000 description 5
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 229910000616 Ferromanganese Inorganic materials 0.000 description 3
- 238000011065 in-situ storage Methods 0.000 description 3
- DALUDRGQOYMVLD-UHFFFAOYSA-N iron manganese Chemical compound [Mn].[Fe] DALUDRGQOYMVLD-UHFFFAOYSA-N 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 238000001556 precipitation Methods 0.000 description 3
- 238000009825 accumulation Methods 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 230000000996 additive effect Effects 0.000 description 2
- SJKRCWUQJZIWQB-UHFFFAOYSA-N azane;chromium Chemical compound N.[Cr] SJKRCWUQJZIWQB-UHFFFAOYSA-N 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 239000000696 magnetic material Substances 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 230000009466 transformation Effects 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 1
- NRTOMJZYCJJWKI-UHFFFAOYSA-N Titanium nitride Chemical compound [Ti]#N NRTOMJZYCJJWKI-UHFFFAOYSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- RRZKHZBOZDIQJG-UHFFFAOYSA-N azane;manganese Chemical compound N.[Mn] RRZKHZBOZDIQJG-UHFFFAOYSA-N 0.000 description 1
- SKKMWRVAJNPLFY-UHFFFAOYSA-N azanylidynevanadium Chemical compound [V]#N SKKMWRVAJNPLFY-UHFFFAOYSA-N 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000010952 in-situ formation Methods 0.000 description 1
- 229910052745 lead Inorganic materials 0.000 description 1
- 230000005389 magnetism Effects 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229910052758 niobium Inorganic materials 0.000 description 1
- 150000004767 nitrides Chemical class 0.000 description 1
- 238000009828 non-uniform distribution Methods 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 238000005204 segregation Methods 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000006104 solid solution Substances 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 229910052718 tin Inorganic materials 0.000 description 1
- 239000011135 tin Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 239000011800 void material Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/12—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of articles with special electromagnetic properties
- C21D8/1244—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of articles with special electromagnetic properties the heat treatment(s) being of interest
- C21D8/1255—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of articles with special electromagnetic properties the heat treatment(s) being of interest with diffusion of elements, e.g. decarburising, nitriding
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D8/00—Modifying the physical properties by deformation combined with, or followed by, heat treatment
- C21D8/12—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of articles with special electromagnetic properties
- C21D8/1244—Modifying the physical properties by deformation combined with, or followed by, heat treatment during manufacturing of articles with special electromagnetic properties the heat treatment(s) being of interest
- C21D8/1272—Final recrystallisation annealing
Definitions
- the present invention relates to a process for producing a grain-oriented electrical steel sheet used as a soft magnetic material for an iron or magnet core of electrical equipments.
- a grain-oriented electrical steel sheet has a crystal grain orientation referred to as "Goss-orientation", in which grains are ⁇ 110 ⁇ 001>-oriented in terms of the Miller index, and usually has a Si content of 4.5% or less and a sheet thickness of from 0.10 to 0.35 mm.
- the steel sheet should have an excellent magnetic characteristic, particularly the magnetic flux density and the watt-loss characteristics and, to meet that requirement, it is important that the crystal grains are highly uniformly aligned in the Goss-orientation. This extremely high accumulation to the Goss-orientation is achieved by utilizing a catastrophic grain growth referred to as "secondary recrystallization".
- the inhibitor suppresses the growth of the primary-recrystallized grains which are out of the Goss-orientation, and thereby, promotes the preferential growth of grains which are in the Goss-orientation.
- Typical precipitates are MnS as proposed by M. F. Littman in Japanese Examined Patent Publication (Kokoku) No. 30-3651 or by J. E. May and D. Turnbull in Trans. Met. Soc. A.I.M.E. 212, 1958, p769-781, AlN as proposed by Taguchi and Sakakura in Japanese Examined Patent Publication (Kokoku) No. 40-15644, MnSe as proposed by Imanaka et al. in Japanese Examined Patent Publication (Kokoku) No. 51-13469, and (Al, Si)N as proposed by Komatsu et al in Japanese Examined Patent Publication (Kokoku) No. 62-45285.
- the first type utilizes a two-step cold rolling using MnS disclosed by M. F. Littman in Japanese Examined Patent Publication (Kokoku) No. 30-3651
- the second type utilizes a large reduction of 80% or more in the final cold rolling step using AlN and MnS disclosed by Taguchi and Sakakura in Japanese Examined Patent Publication (Kokoku) No. 40-15644
- the third type utilizes a two-step cold rolling using MnS (or MnSe) and Sb disclosed by Imanaka et al. in Japanese Examined Patent Publication (Kokoku) No. 51-13469.
- a steel slab is heated at a high temperature, such as 1260°C or higher in the first type process, 1350°C or higher in the second type process when the slab contains 3% Si as disclosed in Japanese Unexamined Patent Publication (Kokai) No. 48-51852 although the temperature varies with the silicon content, or 1230°C or higher in the third type process as disclosed in Japanese Unexamined Patent Publication (Kokai) No. 51-20716 including an example in which an extremely high temperature of 1320°C is adopted to obtain a particularly high flux density. Under such a high temperature of slab heating, coarse precipitates present in steel matrix are once dissolved in steel to form a solid solution and then a fine precipitation occurs during hot rolling and/or the subsequent heat treatment.
- a high temperature such as 1260°C or higher in the first type process, 1350°C or higher in the second type process when the slab contains 3% Si as disclosed in Japanese Unexamined Patent Publication (Kokai) No. 48-51852 although the temperature varies with the silicon
- Japanese Examined Patent Publication (Kokoku) No. 54-14568 proposed that chromium nitride, titanium nitride, vanadium nitride or the like is added to an annealing separator to ensure the nitrogen partial pressure in the atmosphere during final annealing in which the secondary recrystallization is effected and Japanese Examined Patent Publication (Kokoku) No. 53-50008 proposed that a sulfide such as Fe 2 S is added to ensure the sulfur partial pressure and suppress decomposition of the precipitates so that the secondary recrystallization is stabilized.
- a sulfide such as Fe 2 S
- the precipitation occurs under a non-equilibrium condition and is strongly affected by the prior heat and strain history.
- different portions of a steel slab have different heat and strain histories and a steel slab per se has a nonuniform crystal structure due to a macro-segregation of component elements over the slab thickness and to a local dispersion of the ⁇ - and the ⁇ -phases.
- the process for producing a grain-oriented electrical steel sheet based on the control of inhibitor is not essentially stable when used in industry.
- the object of the present invention is to provide a process for industrially stably producing a grain-oriented electrical steel sheet having an excellent magnetic characteristic.
- a process for producing a grain-oriented electrical steel sheet having a high magnetic flux density comprising the steps of:
- the present invention provides a process for stably producing a steel sheet product having a high flux density by defining the primary-recrystallized texture and the secondary recrystallization temperature.
- the present inventors carried out a detailed study on the growth behavior of secondary-recrystallized grains and found the following novel point.
- the secondary-recrystallized grains substantially completely grow up in the temperature region of from 1000 to 1100°C in a material having a primary-recrystallized texture having a ⁇ 111 ⁇ 112> orientation as a main orientation, which is established under a final cold rolling reduction of 80% or more, grains having the Goss-orientation can grow preferentially, and under this condition, a mere nitriding treatment is sufficiently effective to ensure a certain amount of inhibitors for obtaining a good magnetic characteristic.
- a steel slab comprising 3.3 wt% Si, 0.027 wt% acid-soluble Al, 0.007 wt% N, 0.054 wt% C, 0.13 wt% Mn, 0.007 wt% S, and the balance consisting of Fe and unavoidable impurities was hot-rolled to form a 2.3 mm thick hot-rolled strip, which was then annealed at 1100°C for 2 min and cold-rolled at a reduction of 88% to a final product thickness of 0.2 mm.
- the cold-rolled strip was subjected to a primary recrystallization annealing, during which a decarburization treatment was also effected, followed by a nitriding treatment in an ammonia atmosphere to increase the nitrogen content of the steel strip by 0.005% or 0.018%.
- MgO was applied on the samples from the steel strip, which samples were then heated to 900°C at a heating rate of 30°C/hr in an atmosphere of 10% N 2 plus 90% H 2 and rapidly heated to temperatures of from 950 to 1200°C and held there for 20 hours to effect an annealing so that secondary-recrystallized grains fully grew up.
- some samples were taken out of the heating furnace when they were heated to 900°C and an observation showed that the primary-recrystallized structure remained unchanged.
- Figure 1 shows the relationship between the magnetic flux density (B 8 value) and the secondary recrystallization temperature for the thus obtained sample products.
- Figure 2 shows the flux density (B 8 value) for the thus obtained sample products.
- Figure 3 shows the relationship between the flux density (B 8 ) of the product sheets and the partial nitrogen pressure of the atmosphere when the secondary-recrystallized grains grow at 1050°C in a material preliminarily subjected to a nitriding treatment to increase the nitrogen content by 0.018%.
- the optimum temperature range of from 1000 to 1100°C is considered to enable the preferential growth of grains having a sharp Goss-orientation when the primary-recrystallized texture has as a main orientation a ⁇ 111 ⁇ 112>-orientation established through a cold rolling reduction of 80% or higher.
- a cold rolling reduction 80% or higher.
- the basic fact is that grains having the Goss-orientation grow preferentially in the specified temperature range of from 1000 to 1100°C in response to the primary-recrystallized texture established through the final cold rolling reduction of 80% or higher.
- the secondary-recrystallized grains are allowed to grow in the specified temperature range, merely nitriding or increasing the partial nitrogen pressure of the atmosphere is sufficient to ensure a certain amount of inhibitors and to suppress the reduction rate of the inhibitor amount during the secondary recrystallization, so that the conventional problems due to nonuniform distribution of inhibitors is solved to enable the stable production of a grain-oriented electrical steel sheet having a high flux density.
- Japanese Unexamined Patent Publication (Kokai) No. 48-72025 disclosed a process in which the secondary recrystallization temperature is limited in the range of from 1000 to 1100°C, the primary-recrystallized texture was not taken into consideration, and moreover, MnS used as an inhibitor is unstable in that temperature range as shown by W. M. Swift in Metallurgical Transaction, 4, 1973, p153-157 with the result that a low flux density of merely 1.8 Tesla was obtained.
- a steel slab used in the present invention contains 1.8 to 4.8 wt% Si, 0.012 to 0.050 wt% acid-soluble Al, 0.010 wt% or less N, and the balance consisting of Fe and unavoidable impurities, but may contain elements other than those specified above.
- a material containing Si in an amount more than 4.8 wt% cannot be cold-rolled because cracking easily occurs during cold rolling.
- the Si content is reduced, an ⁇ -to- ⁇ transformation occurs during final annealing and the orientation of crystal grains is broken.
- the Si content of 1.8 wt% or more does not substantially affect the crystal orientation due to the ⁇ -to- ⁇ transformation.
- the acid-soluble Al is bonded with N to form AlN or (Al, Si) N, which functions as an inhibitor.
- Al is utilized for this purpose by a later nitriding treatment, it is particularly effective that Al is present as a free Al.
- the acid-soluble Al content is limited in the range of from 0.012 to 0.050 wt%, in which a high flux density is obtained.
- the N content must not exceed 0.010 wt% because a void referred to as a "blister" is formed in a steel sheet for a higher amount of N.
- Additive elements such as Mn, S, Se, B, Bi, Nb, Cr, Sn, and Ti may be used as inhibitor forming elements.
- the slab heating temperature is not necessarily limited and need not be as high as that used in the conventional process because inhibitors can be formed in-situ in a later step of nitriding treatment.
- the slab heating temperature should not preferably exceed 1300°C from the viewpoint of production cost.
- the slab heating temperature is more specifically controlled in accordance with the Al and the N contents not to exceed a temperature above which AlN is completely dissolved in steel.
- the slab heating temperature is not preferably lower than 1000°C because the deformation resistance of the slab increases with lowering of the heating temperature and the steel sheet shape becomes difficult to ensure.
- oxidation of the slab surface excessively occurs to form a melt referred to as "scum".
- the slab heating temperature is preferably in the range of from 1000 to 1270°C.
- the primary-recrystallized grain size is determined by the condition of primary recrystallization annealing including the annealing temperature and duration time and is affected more essentially by the inhibitors which are present before the primary recrystallization annealing.
- the primary recrystallization annealing must be carried out at a higher temperature and/or for a longer time duration to undesirably raise the production cost, in order to obtain a grain size comparable with that obtained by the present invention, for example, the average grain diameter (D) of about 15 ⁇ m or greater.
- a higher temperature and/or a longer time may cause an abnormal grain growth during the primary recrystallization temperature, with the result that the secondary recrystallization becomes unstable.
- the slab heating temperature can be determined from the Al and the N contents by using the above equation.
- the heated steel slab is subsequently hot-rolled to form a hot-rolled strip.
- the hot-rolled strip is annealed, if necessary, at a temperature of from 750 to 1200°C for 30 sec to 30 min.
- the hot-rolled strip is cold-rolled to a final product sheet thickness under a final cold rolling reduction of 80% or more by a single step of cold rolling or by two or more steps of cold rolling with an intermediate annealing therebetween.
- the reduction of 80% or more is essential for obtaining a desired primary-recrystallized texture.
- a cold-rolled strip is subjected to a primary-recrystallization annealing, in which a decarburization is effected to remove carbon usually contained in steel.
- the annealing condition including temperature and duration time should be determined so that the primary-recrystallized grains have an average grain diameter of about 15 ⁇ m or greater.
- the strip thus obtained is coated with an annealing separator and is then subjected to a final annealing for effecting a secondary recrystallization and a purification.
- the strip which has been primary-recrystallization-annealed is subjected to a nitriding treatment before the secondary recrystallization in the final annealing step occurs and that the secondary-recrystallized grains are allowed to substantially completely grow in the temperature region of from 1000 to 1100°C.
- the nitriding treatment may be carried out in any conventional way for nitriding, for example, nitriding using a gas atmosphere having a nitriding ability such as ammonia gas, nitriding during the final annealing by using an annealing separator containing a metal nitride additive having a nitriding ability such as manganese nitride, chromium nitride, or the like.
- a gas atmosphere having a nitriding ability such as ammonia gas
- nitriding during the final annealing by using an annealing separator containing a metal nitride additive having a nitriding ability such as manganese nitride, chromium nitride, or the like.
- the nitriding carried out after the primary recrystallization annealing and before the beginning of the secondary recrystallization strengthens the previously formed weak inhibitor to stabilize the secondary recrystallization.
- the final annealing of the primary-recrystallization-annealed strip is carried out so that secondary-recrystallized grains substantially grow in a temperature region T defined in the following expressions (1) and (2), as specified in claim 2: T ⁇ 20D + 700 1000 ⁇ T ⁇ 1100 where "D" denotes the average grain diameter of primary-recrystallized grains, in ⁇ m.
- Steel slabs comprising 3.2 to 3.3 wt% Si, 0.010 to 0.045 wt% acid-soluble Al, 0.0030 to 0.0090 wt% N, 0.020 to 0.090 wt% C, 0.070 to 0.500 wt% Mn, 0.0030 to 0.0300 wt% S, and the balance Fe and unavoidable impurities were heated to different temperatures of from 1150 to 1400°C and hot-rolled to form 2.3 mm thick hot-rolled strips, which were then annealed at different temperatures of from 900 to 1200°C and cold-rolled at a reduction of 88% to a final thickness of 0.285 mm.
- the cold-rolled strips were primary-recrystallization-annealed at temperatures of from 830 to 1000°C, during which a decarburization was also effected.
- An annealing separator containing MgO as a main component was then applied on the strips.
- Samples from the strips were heated to 900°C at a heating rate of 20°C/hour in an atmosphere of 10% N 2 plus 90% H 2 and then rapidly heated to predetermined different temperatures of from 950 to 1200°C and held there for 20 hours so that the secondary-recrystallized grains were allowed to fully grow. During this sequence, some samples were taken out of the heating furnace when they were heated to 900°C and an observation showed that the primary-recrystallized grain sizes remained unchanged.
- Fig. 7 shows the relationship among the magnetic flux density (B 8 ), the average grain diameter of primary-recrystallized grains, and the secondary recrystallization temperature for the above-obtained sample products.
- the secondary recrystallization is a phenomenon in which the thermal change of primary-recrystallized structure and the thermal change of inhibitor are competing. Namely, as the inhibitor becomes weak during final annealing, the grains having orientations close to the Goss-orientation, which are present in a scattered condition, form a nucleus and begin to grow.
- the growth rate V (cm/sec) of secondary-recrystallized grains is generally expressed by the following equation: V ⁇ exp(-Q/RT) ⁇ 1 D where Q is the activation energy for the grain growth and R is a gas constant.
- the secondary recrystallization temperature is defined in accordance with the primary-recrystallized grain diameter (D).
- the secondary recrystallization temperature (T) is further limited by the primary-recrystallized grain size (D)
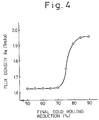
- a sharp Goss-orientation as shown in Figs. 3 and 4 can be more easily ensured than in the case in which the temperature is only limited to the range between 1000 and 1100°C.
- the method of controlling the secondary recrystallization temperature i.e., the temperature at which the secondary-recrystallized grains are allowed to grow is not limited and may carried out by holding or slow heating in the corresponding temperature region.
- a steel slab consisting of 3.3 wt% Si, 0.030 wt% acid-soluble Al, 0.008 wt% N, 0.05 wt% C, 0.14 wt% Mn, 0.007 wt% S and the balance Fe and unavoidable impurities was hot-rolled to form a 1.8 mm thick hot-rolled strip.
- the hot-rolled strip was annealed at 1100°C for 2 min and then cold-rolled at a reduction of 88% to a final product thickness of 0.20 mm.
- the cold-rolled strip was subjected to a primary recrystallization annealing at 830°C, during which a decarburization was also effected.
- Steel slabs comprising 3.28 wt% Si, 0.027 wt% acid-soluble Al, 0.0060 wt% N, 0.14 wt% Mn, 0.007 wt% S and the balance Fe and unavoidable impurities were heated to different temperatures of 1150 and 1300°C and hot-rolled to form 1.8 mm thick hot-rolled strips.
- the strips were annealed by heating at 1150°C for 30 sec and subsequently holding at 900°C for 30 sec.
- the strips were then cold-rolled at a reduction of 89% to a final sheet thickness of 0.20 mm.
- the cold-rolled strips were primary-recrystallization-annealed at 850°C for 90 sec, during which a decarburization was also effected.
- a steel slab comprising 3.3 wt% Si, 0.030 wt% acid-soluble Al, 0.003 wt% N, 0.048 wt% C, 0.13 wt% Mn, 0.010 wt% S, and the balance Fe and unavoidable impurities was heated to 1100°C and hot-rolled to a 2.0 mm thick hot-rolled strip.
- the strip was annealed at 1000°C and cold-rolled at a reduction of 89% to a final thickness of 0.23 mm. Samples from the cold-rolled strip were primary-recrystallization-annealed at different temperatures of 800, 850, and 900°C for 120 sec, during which a decarburization was also effected.
- the samples were then subjected to a nitriding treatment in an atmosphere of ammonia gas so that the nitrogen content was increased by 0.02 to 0.03 wt%.
- An annealing separator was applied on the nitrided samples, which were then final-annealed by heating to 1000°C at a heating rate of 25°C/hr in an atmosphere of 10% N 2 plus 90% H 2 , then heating to 1100°C at a heating rate of 5°C/hr, subsequently heating to 1200°C at a heating rate of 25°C/hr and holding there in a changed atmosphere of 100% H 2 to effect purification.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Physics & Mathematics (AREA)
- Crystallography & Structural Chemistry (AREA)
- Thermal Sciences (AREA)
- Manufacturing & Machinery (AREA)
- Electromagnetism (AREA)
- Mechanical Engineering (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Manufacturing Of Steel Electrode Plates (AREA)
- Solid-Phase Diffusion Into Metallic Material Surfaces (AREA)
Claims (7)
- Verfahren zur Herstellung eines kornorientierten Elektrostahlblechs mit hoher magnetischer Flußdichte, mit den folgenden Schritten:Erwärmen einer Stahlbramme mit 1,8 bis 4,8 Gew.-% Si, 0,012 bis 0,050 Gew.-% säurelöslichem Al, 0,010 Gew.-% oder weniger N, wobei der Rest aus Fe und unvermeidlichen Verunreinigungen besteht, auf eine Warmwalztemperatur;Warmwalzen der erwärmten Bramme zu einem warmgewalzten Band;Kaltwalzen des warmgewalzten Bandes auf eine Endprodukt-Blechdicke mit einem Kaltwalz-Endreduktionsgrad von 80% oder mehr in einem einzigen Kaltwalzschritt oder in zwei oder mehr Kaltwalzschritten mit dazwischenliegendem Glühschritt;Primärrekristallisationsglühen des kalzgewalzten Bandes;Fertigglühen des primär-rekristallisationsgeglühten Bandes derart, daß sekundär-rekristallisierte Körner im wesentlichen vollständig in einem Temperaturbereich von 1000 bis 1100°C wachsen, wonach eine Reinigung bei einer Temperatur über 1100°C ausgeführt wird; undAusführen einer Nitrierbehandlung des primär-rekristallisationsgeglühten Stahlbandes, bevor während des Fertigglühens eine Sekundärrekristallisation stattfindet.
- Verfahren nach Anspruch 1, wobei das Fertigglühen des primär-rekristallisationsgeglühten Bandes so ausgeführt wird, daß sekundär-rekristallisierte Körner im wesentlichen vollständig in einem Temperaturbereich T (°C) wachsen, der durch die folgenden Ausdrücke (1) und (2) definiert wird:
- Verfahren nach Anspruch 1 oder 2, wobei die Atmosphäre während des Fertigglühens so kontrolliert wird, daß der Stickstoffpartialdruck der Atmosphäre gleich 10% oder höher ist, wenn die sekundär-rekristallisierten Körner in dem Temperaturbereich von 1000 bis 1100°C wachsen.
- Verfahren nach Anspruch 3, wobei der Stickstoffpartialdruck gleich 75% oder höher ist.
- Verfahren nach Anspruch 1 oder 2, wobei die Nitrierbehandlung so ausgeführt wird, daß der Stickstoffgehalt des Stahlbandes um 0,005% oder mehr erhöht wird.
- Verfahren nach Anspruch 1 oder 2, wobei die Nitrierbehandlung so ausgeführt wird, daß der Stickstoffgehalt des Stahlbandes um 0,02% oder mehr erhöht wird.
- Verfahren nach einem der Ansprüche 1 bis 6, wobei die Erwärmung der Stahlbramme auf eine Temperatur erfolgt, bei der Al und N nicht vollständig im Stahl gelöst sind.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP1079992A JPH0689405B2 (ja) | 1989-03-30 | 1989-03-30 | 磁束密度の高い一方向性電磁鋼板の製造方法 |
| JP1079991A JPH0689404B2 (ja) | 1989-03-30 | 1989-03-30 | 磁束密度の高い一方向性電磁鋼板の製造方法 |
| JP79991/89 | 1989-03-30 | ||
| JP79992/89 | 1989-03-30 |
Publications (4)
| Publication Number | Publication Date |
|---|---|
| EP0390142A2 EP0390142A2 (de) | 1990-10-03 |
| EP0390142A3 EP0390142A3 (de) | 1992-09-30 |
| EP0390142B1 true EP0390142B1 (de) | 1996-06-26 |
| EP0390142B2 EP0390142B2 (de) | 1999-04-28 |
Family
ID=26420973
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19900106018 Expired - Lifetime EP0390142B2 (de) | 1989-03-30 | 1990-03-29 | Verfahren zum Herstellen kornorientierter Elektrobleche mit hoher magnetischer Flussdichte |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP0390142B2 (de) |
| DE (1) | DE69027553T3 (de) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE10311215A1 (de) * | 2003-03-14 | 2004-10-07 | Thyssenkrupp Electrical Steel Gmbh | Verfahren zum Herstellen von kornorientiertem, kaltgewalztem Elektroblech oder -band |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5354389A (en) * | 1991-07-29 | 1994-10-11 | Nkk Corporation | Method of manufacturing silicon steel sheet having grains precisely arranged in Goss orientation |
| JP2519615B2 (ja) * | 1991-09-26 | 1996-07-31 | 新日本製鐵株式会社 | 磁気特性の優れた方向性電磁鋼板の製造方法 |
| JP2620438B2 (ja) * | 1991-10-28 | 1997-06-11 | 新日本製鐵株式会社 | 磁束密度の高い一方向性電磁鋼板の製造方法 |
| KR960010811B1 (ko) * | 1992-04-16 | 1996-08-09 | 신니뽄세이데스 가부시끼가이샤 | 자성이 우수한 입자배향 전기 강 시트의 제조방법 |
| US6858095B2 (en) | 1992-09-04 | 2005-02-22 | Nippon Steel Corporation | Thick grain-oriented electrical steel sheet exhibiting excellent magnetic properties |
| JP2659655B2 (ja) * | 1992-09-04 | 1997-09-30 | 新日本製鐵株式会社 | 磁気特性の優れた厚い板厚の方向性電磁鋼板 |
| US5858126A (en) * | 1992-09-17 | 1999-01-12 | Nippon Steel Corporation | Grain-oriented electrical steel sheet and material having very high magnetic flux density and method of manufacturing same |
| DE69328998T2 (de) * | 1992-09-17 | 2001-03-01 | Nippon Steel Corp., Tokio/Tokyo | Kornorientierte Elektrobleche und Material mit sehr hoher magnetischer Flussdichte und Verfahren zur Herstellung dieser |
| US5288736A (en) * | 1992-11-12 | 1994-02-22 | Armco Inc. | Method for producing regular grain oriented electrical steel using a single stage cold reduction |
| DE4311151C1 (de) * | 1993-04-05 | 1994-07-28 | Thyssen Stahl Ag | Verfahren zur Herstellung von kornorientierten Elektroblechen mit verbesserten Ummagnetisierungsverlusten |
| EP1179603B1 (de) * | 2000-08-08 | 2011-03-23 | Nippon Steel Corporation | Verfahren zur Herstellung eines kornorientierten Elektrobleches mit hoher magnetischer Flussdichte |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BE563544A (de) * | 1956-12-31 | |||
| US4473416A (en) * | 1982-07-08 | 1984-09-25 | Nippon Steel Corporation | Process for producing aluminum-bearing grain-oriented silicon steel strip |
| JPS59215419A (ja) * | 1983-05-20 | 1984-12-05 | Nippon Steel Corp | 磁束密度の高い一方向性珪素鋼板の製造方法 |
| JPS6240315A (ja) * | 1985-08-15 | 1987-02-21 | Nippon Steel Corp | 磁束密度の高い一方向性珪素鋼板の製造方法 |
| JPH0617512B2 (ja) * | 1986-03-22 | 1994-03-09 | 新日本製鐵株式会社 | 磁束密度の極めて高い一方向性電磁鋼板の製造方法 |
| JPS6475627A (en) * | 1987-09-18 | 1989-03-22 | Nippon Steel Corp | Production of grain oriented electrical steel sheet having extremely high magnetic flux density |
-
1990
- 1990-03-29 DE DE1990627553 patent/DE69027553T3/de not_active Expired - Lifetime
- 1990-03-29 EP EP19900106018 patent/EP0390142B2/de not_active Expired - Lifetime
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE10311215A1 (de) * | 2003-03-14 | 2004-10-07 | Thyssenkrupp Electrical Steel Gmbh | Verfahren zum Herstellen von kornorientiertem, kaltgewalztem Elektroblech oder -band |
| DE10311215B4 (de) * | 2003-03-14 | 2005-09-15 | Thyssenkrupp Electrical Steel Gmbh | Verfahren zum Herstellen von kornorientiertem, kaltgewalztem Elektroblech oder -band |
Also Published As
| Publication number | Publication date |
|---|---|
| DE69027553T2 (de) | 1997-02-20 |
| DE69027553D1 (de) | 1996-08-01 |
| EP0390142A2 (de) | 1990-10-03 |
| DE69027553T3 (de) | 1999-11-11 |
| EP0390142A3 (de) | 1992-09-30 |
| EP0390142B2 (de) | 1999-04-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5643370A (en) | Grain oriented electrical steel having high volume resistivity and method for producing same | |
| EP2025767B1 (de) | Verfahren zur herstellung von kornorientiertem elektrischem stahlblech mit hoher magnetischer flussdichte | |
| EP0390140B1 (de) | Verfahren zur Herstellung von kornorientierten Elektrostahlblechen mit hervorragenden magnetischen Eigenschaften | |
| EP0420238B1 (de) | Herstellungsverfahren für unidirektionale Siliziumstahlbleche mit hoher magnetischer Flussdichte | |
| US4806176A (en) | Process for producing a grain-oriented electromagnetic steel sheet having a high magnetic flux density | |
| EP0390142B1 (de) | Verfahren zum Herstellen kornorientierter Elektrobleche mit hoher magnetischer Flussdichte | |
| KR930001330B1 (ko) | 자속밀도가 높은 일방향성 전자강판의 제조방법 | |
| EP0229846B1 (de) | Herstellungsverfahren für siliziumblattstahl mit weichmagnetischen merkmalen | |
| US3163564A (en) | Method for producing silicon steel strips having cube-on-face orientation | |
| US5186762A (en) | Process for producing grain-oriented electrical steel sheet having high magnetic flux density | |
| EP0378131B1 (de) | Verfahren zum Herstellen eines kornorientierten Elektrostahlbandes | |
| KR950005791B1 (ko) | 고스(Goss) 방위로 집적한 결정방위를 갖는 방향성 규소강판의 제조방법 | |
| EP0076109B2 (de) | Methode zur Erzeugung von kornorientierten Siliziumstahlblechen mit ausgezeichneten magnetischen Eigenschaften | |
| KR100288351B1 (ko) | 한단계의 냉간압연공정을 사용하는 표준 결정립 방향성 전기강 제조 방법 | |
| JP2000045052A (ja) | コイル幅方向端部の形状に優れる低鉄損方向性電磁鋼板及びその製造方法 | |
| EP0357796B1 (de) | Verfahren zur herstellung nichtorientierter elektrofeinbleche | |
| EP0798392A1 (de) | Verfahren zum Herstellen kornorientierter Elektrobleche mir sehr guten magnetischen Eigenschaften | |
| JP2002212636A (ja) | 磁束密度の高い方向性電磁鋼板の製造方法 | |
| JP2607331B2 (ja) | 磁気特性の優れた一方向性電磁鋼板の製造方法 | |
| JP2853552B2 (ja) | 磁気特性の優れた無方向性電磁鋼板とその製造方法 | |
| JP2688146B2 (ja) | 高い磁束密度を有する一方向性電磁鋼板の製造方法 | |
| JP2001060505A (ja) | 一方向性電磁鋼板用の一次再結晶焼鈍板 | |
| JP2521586B2 (ja) | 磁気特性の優れた一方向性電磁鋼板の製造方法 | |
| CA1141631A (en) | Electro magnetic steels | |
| JPH0689404B2 (ja) | 磁束密度の高い一方向性電磁鋼板の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): DE FR GB IT SE |
|
| 17P | Request for examination filed |
Effective date: 19901228 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): DE FR GB IT SE |
|
| 17Q | First examination report despatched |
Effective date: 19940727 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE FR GB IT SE |
|
| REF | Corresponds to: |
Ref document number: 69027553 Country of ref document: DE Date of ref document: 19960801 |
|
| ET | Fr: translation filed | ||
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| ITF | It: translation for a ep patent filed | ||
| PLBQ | Unpublished change to opponent data |
Free format text: ORIGINAL CODE: EPIDOS OPPO |
|
| PLAV | Examination of admissibility of opposition |
Free format text: ORIGINAL CODE: EPIDOS OPEX |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PLAV | Examination of admissibility of opposition |
Free format text: ORIGINAL CODE: EPIDOS OPEX |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| 26 | Opposition filed |
Opponent name: USINOR SACILOR S.A. Effective date: 19970325 |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| PLBF | Reply of patent proprietor to notice(s) of opposition |
Free format text: ORIGINAL CODE: EPIDOS OBSO |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| PLAW | Interlocutory decision in opposition |
Free format text: ORIGINAL CODE: EPIDOS IDOP |
|
| PUAH | Patent maintained in amended form |
Free format text: ORIGINAL CODE: 0009272 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT MAINTAINED AS AMENDED |
|
| 27A | Patent maintained in amended form |
Effective date: 19990428 |
|
| AK | Designated contracting states |
Kind code of ref document: B2 Designated state(s): DE FR GB IT SE |
|
| ITF | It: translation for a ep patent filed | ||
| ET3 | Fr: translation filed ** decision concerning opposition | ||
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20030306 Year of fee payment: 14 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20040330 |
|
| EUG | Se: european patent has lapsed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20070622 Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20090325 Year of fee payment: 20 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080329 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20090327 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20090316 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: PE20 Expiry date: 20100328 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20100328 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20100329 |