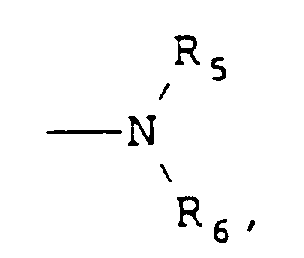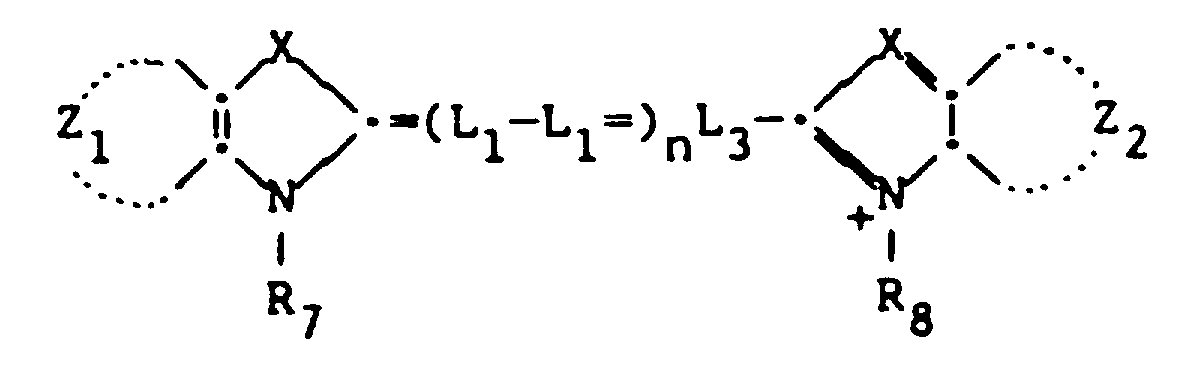EP0346071B1 - Photographisches Verfahren - Google Patents
Photographisches Verfahren Download PDFInfo
- Publication number
- EP0346071B1 EP0346071B1 EP19890305696 EP89305696A EP0346071B1 EP 0346071 B1 EP0346071 B1 EP 0346071B1 EP 19890305696 EP19890305696 EP 19890305696 EP 89305696 A EP89305696 A EP 89305696A EP 0346071 B1 EP0346071 B1 EP 0346071B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- dye
- substituted
- independently
- unsubstituted
- photographic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 238000000034 method Methods 0.000 title claims description 30
- 150000001875 compounds Chemical class 0.000 claims description 35
- -1 silver halide Chemical class 0.000 claims description 35
- 230000003381 solubilizing effect Effects 0.000 claims description 31
- 229910052709 silver Inorganic materials 0.000 claims description 30
- 239000004332 silver Substances 0.000 claims description 30
- 239000000839 emulsion Substances 0.000 claims description 20
- 239000002245 particle Substances 0.000 claims description 16
- 125000000217 alkyl group Chemical group 0.000 claims description 13
- 230000015572 biosynthetic process Effects 0.000 claims description 13
- 125000003118 aryl group Chemical group 0.000 claims description 12
- 125000001424 substituent group Chemical group 0.000 claims description 11
- 239000007787 solid Substances 0.000 claims description 8
- 125000003545 alkoxy group Chemical group 0.000 claims description 5
- 125000004432 carbon atom Chemical group C* 0.000 claims description 5
- 229910052736 halogen Inorganic materials 0.000 claims description 5
- 229910052739 hydrogen Inorganic materials 0.000 claims description 5
- 239000001257 hydrogen Chemical group 0.000 claims description 5
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 5
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 claims description 5
- 125000001434 methanylylidene group Chemical group [H]C#[*] 0.000 claims description 3
- 229910052711 selenium Inorganic materials 0.000 claims description 3
- 229910052717 sulfur Inorganic materials 0.000 claims description 3
- 125000004429 atom Chemical group 0.000 claims description 2
- 150000001768 cations Chemical class 0.000 claims description 2
- 229910052760 oxygen Inorganic materials 0.000 claims description 2
- ANRHNWWPFJCPAZ-UHFFFAOYSA-M thionine Chemical compound [Cl-].C1=CC(N)=CC2=[S+]C3=CC(N)=CC=C3N=C21 ANRHNWWPFJCPAZ-UHFFFAOYSA-M 0.000 claims 4
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims 1
- 125000005843 halogen group Chemical group 0.000 claims 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims 1
- 239000000975 dye Substances 0.000 description 58
- 239000000243 solution Substances 0.000 description 41
- 239000010410 layer Substances 0.000 description 15
- 230000001235 sensitizing effect Effects 0.000 description 15
- 108010010803 Gelatin Proteins 0.000 description 12
- 229920000159 gelatin Polymers 0.000 description 12
- 239000008273 gelatin Substances 0.000 description 12
- 235000019322 gelatine Nutrition 0.000 description 12
- 235000011852 gelatine desserts Nutrition 0.000 description 12
- 239000003795 chemical substances by application Substances 0.000 description 9
- QGKMIGUHVLGJBR-UHFFFAOYSA-M (4z)-1-(3-methylbutyl)-4-[[1-(3-methylbutyl)quinolin-1-ium-4-yl]methylidene]quinoline;iodide Chemical compound [I-].C12=CC=CC=C2N(CCC(C)C)C=CC1=CC1=CC=[N+](CCC(C)C)C2=CC=CC=C12 QGKMIGUHVLGJBR-UHFFFAOYSA-M 0.000 description 8
- 206010070834 Sensitisation Diseases 0.000 description 5
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 5
- 239000007844 bleaching agent Substances 0.000 description 5
- 230000008313 sensitization Effects 0.000 description 5
- 230000003595 spectral effect Effects 0.000 description 5
- 150000002367 halogens Chemical group 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 3
- 230000002378 acidificating effect Effects 0.000 description 3
- 230000002411 adverse Effects 0.000 description 3
- XYXNTHIYBIDHGM-UHFFFAOYSA-N ammonium thiosulfate Chemical compound [NH4+].[NH4+].[O-]S([O-])(=O)=S XYXNTHIYBIDHGM-UHFFFAOYSA-N 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 239000004848 polyfunctional curative Substances 0.000 description 3
- 230000035945 sensitivity Effects 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 229910006069 SO3H Inorganic materials 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 229940125898 compound 5 Drugs 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 125000004356 hydroxy functional group Chemical group O* 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 239000000049 pigment Substances 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 239000011669 selenium Substances 0.000 description 2
- ADZWSOLPGZMUMY-UHFFFAOYSA-M silver bromide Chemical compound [Ag]Br ADZWSOLPGZMUMY-UHFFFAOYSA-M 0.000 description 2
- JHJLBTNAGRQEKS-UHFFFAOYSA-M sodium bromide Chemical compound [Na+].[Br-] JHJLBTNAGRQEKS-UHFFFAOYSA-M 0.000 description 2
- 150000004764 thiosulfuric acid derivatives Chemical class 0.000 description 2
- UMGDCJDMYOKAJW-UHFFFAOYSA-N thiourea Chemical compound NC(N)=S UMGDCJDMYOKAJW-UHFFFAOYSA-N 0.000 description 2
- 229920002554 vinyl polymer Polymers 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 150000005206 1,2-dihydroxybenzenes Chemical class 0.000 description 1
- AIGNCQCMONAWOL-UHFFFAOYSA-N 1,3-benzoselenazole Chemical compound C1=CC=C2[se]C=NC2=C1 AIGNCQCMONAWOL-UHFFFAOYSA-N 0.000 description 1
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical compound C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- 150000005208 1,4-dihydroxybenzenes Chemical class 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- ZKAMEFMDQNTDFK-UHFFFAOYSA-N 1h-imidazo[4,5-b]pyrazine Chemical compound C1=CN=C2NC=NC2=N1 ZKAMEFMDQNTDFK-UHFFFAOYSA-N 0.000 description 1
- CDAWCLOXVUBKRW-UHFFFAOYSA-N 2-aminophenol Chemical class NC1=CC=CC=C1O CDAWCLOXVUBKRW-UHFFFAOYSA-N 0.000 description 1
- JKFYKCYQEWQPTM-UHFFFAOYSA-N 2-azaniumyl-2-(4-fluorophenyl)acetate Chemical compound OC(=O)C(N)C1=CC=C(F)C=C1 JKFYKCYQEWQPTM-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- RKJUIXBNRJVNHR-UHFFFAOYSA-N 3H-indole Chemical compound C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 1
- ALGIYXGLGIECNT-UHFFFAOYSA-N 3h-benzo[e]indole Chemical compound C1=CC=C2C(C=CN3)=C3C=CC2=C1 ALGIYXGLGIECNT-UHFFFAOYSA-N 0.000 description 1
- ZFXPBTZXYNIAJW-UHFFFAOYSA-N 4-[2-(2-phenylethenyl)phenyl]triazine Chemical compound C=1C=CC=CC=1C=CC1=CC=CC=C1C1=CC=NN=N1 ZFXPBTZXYNIAJW-UHFFFAOYSA-N 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 101100172879 Caenorhabditis elegans sec-5 gene Proteins 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- 241000483002 Euproctis similis Species 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- RAXXELZNTBOGNW-UHFFFAOYSA-O Imidazolium Chemical compound C1=C[NH+]=CN1 RAXXELZNTBOGNW-UHFFFAOYSA-O 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- 241000978776 Senegalia senegal Species 0.000 description 1
- 229910021612 Silver iodide Inorganic materials 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Natural products NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 1
- SJOOOZPMQAWAOP-UHFFFAOYSA-N [Ag].BrCl Chemical compound [Ag].BrCl SJOOOZPMQAWAOP-UHFFFAOYSA-N 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 150000001241 acetals Chemical class 0.000 description 1
- 238000007754 air knife coating Methods 0.000 description 1
- 239000012670 alkaline solution Substances 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 239000002216 antistatic agent Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 238000000149 argon plasma sintering Methods 0.000 description 1
- 125000004104 aryloxy group Chemical group 0.000 description 1
- 238000010923 batch production Methods 0.000 description 1
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N benzo-alpha-pyrone Natural products C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 1
- AMTXUWGBSGZXCJ-UHFFFAOYSA-N benzo[e][1,3]benzoselenazole Chemical compound C1=CC=C2C(N=C[se]3)=C3C=CC2=C1 AMTXUWGBSGZXCJ-UHFFFAOYSA-N 0.000 description 1
- KXNQKOAQSGJCQU-UHFFFAOYSA-N benzo[e][1,3]benzothiazole Chemical compound C1=CC=C2C(N=CS3)=C3C=CC2=C1 KXNQKOAQSGJCQU-UHFFFAOYSA-N 0.000 description 1
- WMUIZUWOEIQJEH-UHFFFAOYSA-N benzo[e][1,3]benzoxazole Chemical compound C1=CC=C2C(N=CO3)=C3C=CC2=C1 WMUIZUWOEIQJEH-UHFFFAOYSA-N 0.000 description 1
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000004061 bleaching Methods 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 235000001671 coumarin Nutrition 0.000 description 1
- 125000000332 coumarinyl group Chemical class O1C(=O)C(=CC2=CC=CC=C12)* 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000007766 curtain coating Methods 0.000 description 1
- 238000003618 dip coating Methods 0.000 description 1
- 238000007606 doctor blade method Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-O hydron;1,3-oxazole Chemical compound C1=COC=[NH+]1 ZCQWOFVYLHDMMC-UHFFFAOYSA-O 0.000 description 1
- SMWDFEZZVXVKRB-UHFFFAOYSA-O hydron;quinoline Chemical compound [NH+]1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-O 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 239000011229 interlayer Substances 0.000 description 1
- 229910052741 iridium Inorganic materials 0.000 description 1
- GKOZUEZYRPOHIO-UHFFFAOYSA-N iridium atom Chemical compound [Ir] GKOZUEZYRPOHIO-UHFFFAOYSA-N 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- AWJUIBRHMBBTKR-UHFFFAOYSA-O isoquinolin-2-ium Chemical compound C1=[NH+]C=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-O 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 150000002734 metacrylic acid derivatives Chemical class 0.000 description 1
- LVWZTYCIRDMTEY-UHFFFAOYSA-N metamizole Chemical compound O=C1C(N(CS(O)(=O)=O)C)=C(C)N(C)N1C1=CC=CC=C1 LVWZTYCIRDMTEY-UHFFFAOYSA-N 0.000 description 1
- 239000003607 modifier Substances 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 229910052762 osmium Inorganic materials 0.000 description 1
- SYQBFIAQOQZEGI-UHFFFAOYSA-N osmium atom Chemical compound [Os] SYQBFIAQOQZEGI-UHFFFAOYSA-N 0.000 description 1
- 239000006174 pH buffer Substances 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 239000001814 pectin Substances 0.000 description 1
- 235000010987 pectin Nutrition 0.000 description 1
- 229920001277 pectin Polymers 0.000 description 1
- 229940044652 phenolsulfonate Drugs 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 150000004986 phenylenediamines Chemical class 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 229920002717 polyvinylpyridine Polymers 0.000 description 1
- NDGRWYRVNANFNB-UHFFFAOYSA-N pyrazolidin-3-one Chemical class O=C1CCNN1 NDGRWYRVNANFNB-UHFFFAOYSA-N 0.000 description 1
- JUJWROOIHBZHMG-UHFFFAOYSA-O pyridinium Chemical compound C1=CC=[NH+]C=C1 JUJWROOIHBZHMG-UHFFFAOYSA-O 0.000 description 1
- WVIICGIFSIBFOG-UHFFFAOYSA-N pyrylium Chemical compound C1=CC=[O+]C=C1 WVIICGIFSIBFOG-UHFFFAOYSA-N 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 238000007763 reverse roll coating Methods 0.000 description 1
- 229910052702 rhenium Inorganic materials 0.000 description 1
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 description 1
- 238000007761 roller coating Methods 0.000 description 1
- 150000003839 salts Chemical group 0.000 description 1
- ZUNKMNLKJXRCDM-UHFFFAOYSA-N silver bromoiodide Chemical compound [Ag].IBr ZUNKMNLKJXRCDM-UHFFFAOYSA-N 0.000 description 1
- 229940045105 silver iodide Drugs 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 235000019345 sodium thiosulphate Nutrition 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- PJANXHGTPQOBST-UHFFFAOYSA-N stilbene Chemical class C=1C=CC=CC=1C=CC1=CC=CC=C1 PJANXHGTPQOBST-UHFFFAOYSA-N 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229910052714 tellurium Inorganic materials 0.000 description 1
- PORWMNRCUJJQNO-UHFFFAOYSA-N tellurium atom Chemical compound [Te] PORWMNRCUJJQNO-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000000147 tetrahydroquinolinyl group Chemical class N1(CCCC2=CC=CC=C12)* 0.000 description 1
- 150000003567 thiocyanates Chemical class 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
Classifications
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C1/00—Photosensitive materials
- G03C1/005—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein
- G03C1/06—Silver halide emulsions; Preparation thereof; Physical treatment thereof; Incorporation of additives therein with non-macromolecular additives
- G03C1/08—Sensitivity-increasing substances
- G03C1/10—Organic substances
- G03C1/12—Methine and polymethine dyes
- G03C1/14—Methine and polymethine dyes with an odd number of CH groups
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C5/00—Photographic processes or agents therefor; Regeneration of such processing agents
- G03C5/26—Processes using silver-salt-containing photosensitive materials or agents therefor
Definitions
- This invention relates to photography, especially photographic processes for elements having silver halide sensitized with a cyanine dye.
- Silver halide elements are usually subjected to some type of photographic processing after exposure in order to form images suitable for viewing.
- the processing generally involves contacting the element with a developer solution to cause formation of a silver image corresponding to a latent image formed on the silver halide during exposure, and then with a fixing solution to solubilize and remove silver halide that was not converted to silver during development.
- a developer solution to cause formation of a silver image corresponding to a latent image formed on the silver halide during exposure
- a fixing solution to solubilize and remove silver halide that was not converted to silver during development.
- Any of a number of other known processing solutions may also be used, such as stop baths to halt image development, bleach solutions to remove already formed silver from the element, and a number of other known solutions (e.g., stabilizer baths, hardener solutions, wash baths, and the like).
- the silver halide that is used in photographic elements is usually inherently sensitive only to blue light. It is therefore often desirable, in order to provide sensitivity to light of other regions of the spectrum or to enhance the sensitivity to blue light, to spectrally sensitize the silver halide by adsorbing a sensitizing dye to the silver halide, thus imparting to the silver halide sensitivity to the wavelength of light absorbed by the dye.
- the sensitizing dye can come out of the element and enter one or more of the processing solutions. Indeed, it is usually desirable to remove (usually in the fixing bath)the sensitizing dye from the element during processing so as to minimize adverse effects on image quality that can occur as a result of sensitizing dye remaining in the element after processing.
- sensitizing dye instead of coming out of the element and dissolving in a processing solution, is removed from the element, but is not fully solubilized in the processing solution and forms solid particles. These particles can have a number of adverse effects. They can adhere to the surface of the element, forming visible specks or blotches on the image. They can also adhere to processing equipment, impeding flow or mixing of processing solutions, and necessitating costly and time-consuming cleaning procedures.
- JP-A-62-166 335 describes the use of triazinyl stilbene whitening agents to improve whiteness, the precipitates that are produced in the presence of such compounds, and a developer solution which reduces precipitate formation.
- Research Disclosure No. 207, July 1981, p. 268 describes the use of stilbene compounds to reduce retained sensitizing dye stain.
- R1, R2, R3, and R4 are each independently halogen (e.g. fluoro, chloro, bromo, iodo), substituted or unsubstituted alkoxy of from 1 to 6 carbon atoms (e.g., methoxy, ethoxy, propoxy).
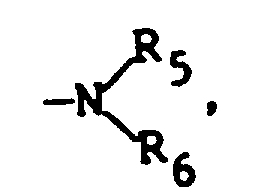
- R5 and R6 are each independently hydrogen, substituted or unsubstituted alkyl of from 1 to 7 carbon atoms (e.g., methyl, ethyl, propyl, isopropyl, t -butyl, n-hexyl, etc.), or substituted or unsubstituted aryl (e.g., phenyl, naphthyl, and the like).
- Useful substituents for the above R groups can include any of a number of substituents commonly utilized for such groups that do not adversely affect the properties of the compound to a significant degree. Examples of such substituents include halogen, alkyl, aryl, and alkoxy as described above, as well as other known substituents, such as allyl, thioethers, etc. . . .
- solubilizing groups are groups that tend to increase the solubility of organic compounds in water.
- solubilizing groups are well-known in the art and include, for example, sulfo, carboxy, and hydroxy.
- anionic sulfo is meant an ionized -SO ⁇ 3 group associated with a counterion, as opposed to an acidic -SO3H group.
- the sulfo group may be in acid form prior to such contact.
- the solubilizing compound of formula (I) is being used in conjunction with a developer solution with a high pH (usually 9 or above), then the solubilizing group may be an acidic -SO3H group until contact with the developing solution, at which time the high pH would ionize the sulfo group.
- solubilizing groups include any group whose presence as a substituent on the compound of formula (I) will tend to render it more soluble in aqueous solutions than without it.
- the compounds of formula (I) can be made by methods well-known in the art. Such methods are described in, for example, U.S. Patents 3,617,295, 3,615,641, 2,937,089, and 3,615,632.
- the dye solubilizing compound of formula (I) can be incorporated in any processing solution where the sensitizing dye would be present in the solution at a concentration sufficient to cause formation of solid dye particles in the absence of the dye solubilizing compound.
- the dye solubilizing compound can be incorporated in the photographic element so that the dye solubilizing compound is present when the element is contacted with the solution that is susceptible to dye particle formation.
- the processing solution may for example, have from 0.01 to 1.0 g/l of the cyanine dye.
- the amount of dye solubilizing compound in the processing solution may, for example, be from 0.1 to 10g/l
- the compound of formula (I) can be used in conjunction with any combination of photographic element and process that is susceptible to formation of solid particles of cyanine sensitizing dye in any of the processing solutions.
- the cyanine sensitizing dye can be any of a number of such dyes, which are well-known in the art.
- the cyanine spectral sensitizing dyes include, joined by a methine linkage, two basic heterocyclic nuclei, such as those derived from quinolinium, pyridinium, isoquinolinium, 3H-indolium, benz[e]indolium, oxazolium, thiazolium, selenazolinium, imidazolium, benzoxazolinium, benzothiazolium, benzoselenazolium, benzimidazolium, naphthoxazolium, naphthothiazolium, naphthoselenazolium, thiazolinium dihydronaphthothiazolium, pyrylium, and imidazopyrazinium quaternary salts. Further description and examples of such dyes can be found in James, The Theory of the Photographic Process 4th ,
- Useful cyanine dyes include those according to the formula:
- Z1 and Z2 represent the atoms necessary to complete a substituted or unsubstituted aromatic ring structure.
- R7 and R8 are each independently substituted or unsubstituted alkyl of 1 to 12 carbon atoms (e.g., methyl, ethyl, propyl, butyl). R7 and R8 may be substituted with substituents as is known in the art, such as halogen, alkoxy, aryl, aryloxy, sulfo, carboxyl, and the like).
- L1, L2, and L3 are each independently substituted (with substituents known in the art, e.g., alkyl) or unsubstituted methine linkages.
- X is O, S, Se, -C-, or N- and n is 0, 1, or 2.
- Photographic elements useful in the practice of the invention generally comprise a support having thereon at least one silver halide emulsion layer.
- the silver halide emulsion can comprise any type of silver halide that is useful in photographic emulsions. This includes silver bromide, silver iodide, silver chlorobromide, silver bromoiodide, silver chlorobromoiodide, or mixtures thereof.
- the silver halide can be in the form of grains bounded by 100, 111, or 110 crystal planes, as described in Research Disclosure I , or in the form of tabular grains, as described in Research Disclosure , January, 1983, item 22534.
- the silver halide can be present in any amount known in the art to be useful in photographic emulsions.
- the silver halide is generally present in the emulsion in an amount such that when it is coated as a layer in a photographic element, the coverage of silver will be from 1.1 to 3.2 g/m.
- the amount of sensitizing dye in the emulsion can be any amount that is known in the art to be useful for spectral sensitization of silver halide, subject to the requirement that it be sufficient to cause formation of solid dye particles in a solution for processing the element.
- the quantity of dye employed will vary with the specific dye or dye combination chosen as well as the size and aspect ration of the grains. It is known in the photographic art that optimum spectral sensitization is generally obtained with organic dyes at 25 to 100 percent of monolayer coverage of the total available surface area of surface sensitive silver halide grains, as disclosed for example, in West et al, "The Adsorption of Sensitizing Dyes in Photographic Emulsions," Journal of Phys. Chem. , Vol. 56, p.
- Optimum dye concentration levels can be chosen by procedures taught by Mees, Theory of the Photographic Process , Macmillan (1942), pp. 1067-69.
- Preferred dye amounts in photographic elements useful according to the present invention range from 1.1 to 10.8 g/m.
- One or more spectral sensitizing dyes may be used to achieve a desired spectral sensitization of the silver halide as well as to achieve results such as supersensitization, as discussed by Gilman in Photographic Science and Engineering , Vol. 18, 1974, pp. 418-30.
- supersensitizing dye combinations include those disclosed in Mcfall et al U.S. Patent 2,933,390, Jones et al U.S. Patent 2,937,089, Motter U.S. Patent 3,506,443, and Schwan et al U.S. Patent 3,672,898.
- the emulsion preferably includes a vehicle for coating the emulsion as a layer of a photographic element.
- vehicle include both naturally occurring substances such as proteins, protein derivatives, cellulose derivatives (e.g., cellulose esters), gelatin (e.g., alkali-treated gelatin such as cattle bone or hide gelatin, or acid-treated gelatin such as pigskin gelatin), gelatin derivatives (e.g., acetylated gelatin, phthalated gelatin, and the like), polysaccharides (e.g., dextran, gum arabic, casein, pectin, and the like), and others, as described in Research Disclosure I .
- Also useful as vehicles or vehicle extenders are hydrophilic water-permeable colloids.
- polystyrene resin examples include synthetic polymeric peptizers, carriers, and/or binders such as poly(vinyl alcohol), poly(vinyl lactams), acrylamide polymers, polyvinyl acetals, polymers of alkyl and sulfoalkyl acrylates and methacrylates, hydrolyzed polyvinyl acetates, polyamides, polyvinyl pyridine, methacrylamide copolymers, and the like, as described in Research Disclosure I .
- the vehicle can be present in the emulsion in any amount known to be useful in photographic emulsions.
- the silver halide emulsion can also include any of the addenda known to be useful in photographic emulsions. These include chemical sensitizers, such as active gelatin, sulfur, selenium, tellurium, gold, platinum, palladium, iridium, osmium, rhenium, phosphorous, or combinations thereof. Chemical sensitization is generally carried out at pAg levels of from 5 to 10, pH levels of from 5 to 8, and temperatures of from 30 to 80°C, as illustrated in Research Disclosure , June, 1975, item 13452 and U.S. Patent 3,772,031.
- chemical sensitizers such as active gelatin, sulfur, selenium, tellurium, gold, platinum, palladium, iridium, osmium, rhenium, phosphorous, or combinations thereof. Chemical sensitization is generally carried out at pAg levels of from 5 to 10, pH levels of from 5 to 8, and temperatures of from 30 to 80°C, as illustrated in Research Disclosure , June, 1975, item 13452
- addenda include brighteners, antifoggants, stabilizers, filter dyes, light absorbing or reflecting pigments, vehicle hardeners such as gelatin hardeners, coating aids, dye-forming couplers, and development modifiers such as development inhibitor releasing couplers and bleach accelerators.
- the emulsion and other layers of photographic elements can be coated onto a support using techniques well-known in the art. These techniques include immersion or dip coating, roller coating, reverse roll coating, air knife coating, doctor blade coating, stretch-flow coating, and curtain coating.
- the silver halide emulsion layer of the photographic element can be coated simultaneously or sequentially with other emulsion layers, subbing layers, filter dye layers, or interlayers or overcoat layers containing various addenda known to be included in photographic elements, such as antifoggants, oxidized developer scavengers, DIR couplers, antistatic agents, optical brighteners, light-absorbing or light-scattering pigments, and the like.
- the coated layers of the photographic element may be chill-set or dried, or both. Drying may be accelerated by known techniques such as conduction, convection, radiation heating, or a combination thereof.
- the photographic element useful in the practice of the invention can be black and white or color.
- Certain black and white elements requiring moderately high density in imaged areas, minimum density in non-imaged areas, and sufficient contrast to yield high quality definition images e.g., microfilm or microfiche elements
- contain sufficient amounts of silver halide i.e., 1.1 to 2.2 g/m) so that when they are sensitized with a cyanine dye, the amount of cyanine dye is sufficient to cause dye particle formation in processing solutions.
- a color photographic element generally contains three silver halide emulsion layers: a blue-sensitive layer having a yellow color coupler associated therewith, a green-sensitive layer having a magenta color coupler associated therewith, and a red-sensitive layer having a cyan color coupler associated therewith.
- the compound of formula (I) can be used in conjunction with essentially any known process, the exact type of which is not critical. Such processes generally include contacting the element with an aqueous alkaline solution of a developing agent, and then contacting the element with a solution of fixing agent or with bleaching and fixing agent(s). Examples of processing formulations and techniques are described in L. Mason, Photographic Processing Chemistry , Focal Press, London, 1966; Processing Chemicals and Formulas , Publication J-1, Eastman Kodak Company, 1985; Photo-Lab Index , Morgan & Morgan, Inc., Dobbs Ferry, New York, 1977; and Neblette's Handbook of Photography and Reprography Materials, Processes and Systems , VanNostrand Reinhold Co., 7th Ed., 1977.
- the compound of formula (I) can be incorporated in the element or in any processing solution in which solid dye particle formation is a problem.
- the compound is to be included in a processing solution, it is preferably incorporated in a developing solution or a fixing solution.
- Developing solutions generally have a pH of 9 or more, although that is not a requirement of the invention.
- the use of the compound of formula (I) in an alkaline developing solution, as indicated above, does allow for ionization of any acidic sulfo solubilizing groups on the compound.
- the developing agent can be any of the known developing agents, as described in James, The Theory of the Photographic Process , 4th, Macmillan, 1977 [hereinafter "James"], Chapter 11.
- Useful developing agents include hydroquinones, catechols, aminophenols, pyrazolidones, phenylenediamines, tetrahydroquinolines, bis(pyridone)amines, cycloalkenones, pyramidines, reductones, and coumarins.
- Fixing agents are also known in the art, as described in James, Chapter 15.
- Useful fixing agents include thiosulfates, thiocyanates, cyanides, sulfite, ammonia, thiourea, thioacids, thiosugars, concentrated alkali halides, and organic solvents such as acetone.
- Thiosulfates, such as sodium thiosulfate or ammonium thiosulfate are among the most commonly-used fixing agents.
- wash baths consisting primarily of water and optionally pH buffers or other stabilizers, bleach or bleach/fix solutions, color developer solutions (which may or may not contain photographic couplers, stop baths for arresting photographic development, gelatin hardening solutions, and other known photographic processing solutions. All these solutions and processing steps are well-known in the art.
- a photographic element was prepared, having an emulsion layer containing gelatin at a coverage of 1.56 g/m and silver bromide at a coverage of 1.61 g/m sensitized with a dye of the formula: at a coverage of 5.4 mg/m. Samples of the element were processed in a semicontinuous batch process in which the element moves successively from one tank to the next of well-mixed non-replenished processing solutions.
- tanks 1 and 2 also contained 0.5 g/l of compound 5 from Table I. After 18.6 m of film had been processed, no dye particles were observed in any of the tanks.
- tank 3 instead of tanks 1 and 2 contained 0.5 g/l of compound 5 from Table I. After 9.3 m of film had been processed, no dye particles were observed in any of the tanks.
Landscapes
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Chemical & Material Sciences (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Silver Salt Photography Or Processing Solution Therefor (AREA)
Claims (11)
- Verfahren zur Entwicklung eines photographischen Elementes mit einer Schicht mit einer Silberhalogenidemulsion, die mit einem Cyaninfarbstoff sensibilisiert ist, bei dem man das Element mit mindestens einer Entwicklungslösung in Kontakt bringt, dadurch gekennzeichnet, daß die Lösung eine einen Farbstoff löslichmachende Verbindung der folgenden Formel enthält:
M ein Kation, und
wobei der Cyaninfarbstoff in der Entwicklungslösung in einer Konzentration vorliegt, die ausreicht, um die Bildung von festen Farbstoffpartikeln in Abwesenheit der Farbstoff löslichmachenden Verbindung zu verursachen. - Verfahren nach Anspruch 1, dadurch gekennzeichnet, daß die Entwicklungslösung eine photographische Entwicklerlösung ist.
- Verfahren nach Anspruch 1, bei dem die Entwicklungslösung eine photographische Fixierlösung ist.
- Verfahren nach Ansprüchen 1 - 3, bei dem der Farbstoff der folgenden Formel entspricht:
worin bedeuten: Z₁ und Z₂ stellen die Atome dar, die zur Vervollständigung einer substituierten oder unsubstituierten aromatischen Ringstruktur erforderlich sind,R₇ und R₈ stehen jeweils unabhängig voneinander für substituiertes oder unsubstituiertes Alkyl,L₁, L₂ und L₃ stehen jeweils unabhängig voneinander für substituierte oder unsubstituierte Methinbindungen,X steht für 0, S, Se, -C- oder N-, undn ist gleich 0, 1 oder 2. - Verfahren nach Ansprüchen 1 - 4, bei dem das photographische Element ein Schwarz-Weiß-Element ist.
- Verfahren nach Ansprüchen 1 - 5, bei dem die Entwicklungslösung 0,01 bis 1,0 g/l des Cyaninfarbstoffes enthält.
- Verfahren nach Ansprüchen 1 - 6, bei dem die Entwicklungslösung 0,1 bis 10 g/l der den Farbstoff löslichmachenden Verbindung enthält.
- Verfahren nach Ansprüchen 1 - 7, bei dem R₁, R₂, R₃ und R₄ jeweils unabhängig voneinander stehen für
- Verfahren nach Ansprüchen 1 - 8, bei dem die löslichmachenden Gruppen Sulfo-, Hydroxy- oder Carboxygruppen sind.
- Verfahren nach Ansprüchen 1 - 9, bei dem der Cyaninfarbstoff in der Entwicklungslösung in einer Konzentration vorliegt, die ausreicht, um in Abwesenheit der Farbstoff löslichmachenden Verbindung die Bildung von festen Farbstoffpartikeln hervorzurufen, die einen mittleren Durchmesser von mindestens 0,3 µm aufweisen.
- Verfahren nach Ansprüchen 1 - 10, bei dem R₁ und R₃ jeweils stehen für
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US20263288A | 1988-06-06 | 1988-06-06 | |
| US202632 | 1988-06-06 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0346071A2 EP0346071A2 (de) | 1989-12-13 |
| EP0346071A3 EP0346071A3 (de) | 1991-02-27 |
| EP0346071B1 true EP0346071B1 (de) | 1996-03-13 |
Family
ID=22750685
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19890305696 Expired - Lifetime EP0346071B1 (de) | 1988-06-06 | 1989-06-06 | Photographisches Verfahren |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP0346071B1 (de) |
| JP (1) | JPH0228649A (de) |
| CA (1) | CA1333967C (de) |
| DE (1) | DE68925924T2 (de) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2576900B2 (ja) * | 1988-05-09 | 1997-01-29 | 富士写真フイルム株式会社 | ハロゲン化銀感光材料の現像処理方法 |
| JP3133874B2 (ja) * | 1993-09-16 | 2001-02-13 | 富士写真フイルム株式会社 | ハロゲン化銀写真感光材料 |
| US5955248A (en) * | 1998-07-06 | 1999-09-21 | Eastman Kodak Company | Concentrated photographic fixer additive and fixing compositions containing triazinylstilbene and method of photographic processing |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0675178B2 (ja) * | 1986-01-20 | 1994-09-21 | コニカ株式会社 | ハロゲン化銀カラ−写真感光材料用発色現像液 |
| JPS62257154A (ja) * | 1986-04-30 | 1987-11-09 | Konika Corp | ハロゲン化銀カラ−写真感光材料の処理方法 |
-
1989
- 1989-05-11 CA CA 599470 patent/CA1333967C/en not_active Expired - Fee Related
- 1989-06-05 JP JP14127289A patent/JPH0228649A/ja active Pending
- 1989-06-06 DE DE1989625924 patent/DE68925924T2/de not_active Expired - Fee Related
- 1989-06-06 EP EP19890305696 patent/EP0346071B1/de not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| EP0346071A3 (de) | 1991-02-27 |
| EP0346071A2 (de) | 1989-12-13 |
| DE68925924T2 (de) | 1996-09-05 |
| DE68925924D1 (de) | 1996-04-18 |
| JPH0228649A (ja) | 1990-01-30 |
| CA1333967C (en) | 1995-01-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US4948717A (en) | Solid particle dye dispersions for photographic filter layers | |
| JPH0614175B2 (ja) | 直接陽画写真乳剤の製造方法 | |
| US4521508A (en) | Silver halide photographic light-sensitive materials | |
| EP0317950B1 (de) | Photographische Elemente, die einen Bleich-Beschleuniger-Vorläufer enthalten | |
| EP0059144B1 (de) | Silberhalogenidemulsion, enthaltend einen Stabilisator für das latente Bild, und fotografisches Element | |
| EP0498302A1 (de) | Silberhalogenidemulsionen für die Verwendung in Entwicklungsverfahren, bei denen eine physikalische Entwicklung in Lösung erfolgt | |
| DE68922239T2 (de) | Photographische Silberhalogenidmaterialien. | |
| EP0351593A2 (de) | Bei Licht handhabbares photographisches Element mit einer eine Dispersion fester Partikel eines Filterfarbstoffes enthaltenden Schicht | |
| US3816121A (en) | Direct positive photographic material containing a color coupler under one micron in size and fogged silver halide grains with substantially no internal sensitivity having absorbed on the surface a desensitizing dye containing a solubilizing group | |
| EP0346071B1 (de) | Photographisches Verfahren | |
| EP0304323B1 (de) | Farbphotographisches lichtempfindliches direkt-positives Silberhalogenidmaterial | |
| EP0605917B1 (de) | Rotsensibilisatoren für silberchloridreiche Emulsionen | |
| US3149970A (en) | Production of photographic silver images by physical development | |
| US5238793A (en) | Photographic process | |
| EP0017148B1 (de) | Innendotierte, chloridreiche Silberhalogenidemulsionen, Verfahren zu deren Herstellung und photographische Elemente | |
| JPH01280750A (ja) | ハロゲン化銀写真感光材料 | |
| US4506008A (en) | Silver halide photographic light-sensitive materials | |
| JPH07199390A (ja) | 写真要素及び写真方法 | |
| JPS60203936A (ja) | ハロゲン化銀写真感光材料 | |
| JP2831020B2 (ja) | 高塩化物ハロゲン化銀写真組成物用増感染料 | |
| US5077190A (en) | Process for spectral sensitization of a silver halide emulsion | |
| CA1146001A (en) | High contrast photographic emulsions containing a phenyl hydrazine derivative | |
| US3854949A (en) | Silver halide photographic compositions and processes for obtaining positive images | |
| JPS5856857B2 (ja) | ハロゲン化銀写真感光材料の処理方法 | |
| US4808516A (en) | Photographic emulsion and element |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE CH DE ES FR GB IT LI NL SE |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AT BE CH DE ES FR GB IT LI NL SE |
|
| 17P | Request for examination filed |
Effective date: 19910819 |
|
| 17Q | First examination report despatched |
Effective date: 19940516 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): BE CH DE FR GB LI NL |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE CH DE FR GB LI NL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Effective date: 19960313 Ref country code: CH Effective date: 19960313 |
|
| REF | Corresponds to: |
Ref document number: 68925924 Country of ref document: DE Date of ref document: 19960418 |
|
| ET | Fr: translation filed | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19970331 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19970610 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19980505 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 19980625 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990101 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990226 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 19990101 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990606 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19990606 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000503 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20000714 Year of fee payment: 12 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20010630 |
|
| BERE | Be: lapsed |
Owner name: EASTMAN KODAK CY (A NEW JERSEY CORP.) Effective date: 20010630 |