CN1020249C - 噻唑啉类农用杀菌剂 - Google Patents
噻唑啉类农用杀菌剂 Download PDFInfo
- Publication number
- CN1020249C CN1020249C CN 88107909 CN88107909A CN1020249C CN 1020249 C CN1020249 C CN 1020249C CN 88107909 CN88107909 CN 88107909 CN 88107909 A CN88107909 A CN 88107909A CN 1020249 C CN1020249 C CN 1020249C
- Authority
- CN
- China
- Prior art keywords
- compound
- xah
- weight ratio
- preparation
- bactericide
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 230000000844 anti-bacterial effect Effects 0.000 title abstract description 8
- 239000003899 bactericide agent Substances 0.000 title abstract description 8
- -1 thiazoline compound Chemical class 0.000 claims abstract description 5
- 150000001875 compounds Chemical class 0.000 claims description 13
- 238000002360 preparation method Methods 0.000 claims description 8
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 7
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 claims description 6
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 4
- YDEXUEFDPVHGHE-GGMCWBHBSA-L disodium;(2r)-3-(2-hydroxy-3-methoxyphenyl)-2-[2-methoxy-4-(3-sulfonatopropyl)phenoxy]propane-1-sulfonate Chemical compound [Na+].[Na+].COC1=CC=CC(C[C@H](CS([O-])(=O)=O)OC=2C(=CC(CCCS([O-])(=O)=O)=CC=2)OC)=C1O YDEXUEFDPVHGHE-GGMCWBHBSA-L 0.000 claims description 4
- 239000000843 powder Substances 0.000 claims description 4
- 239000000375 suspending agent Substances 0.000 claims description 4
- 238000009736 wetting Methods 0.000 claims description 4
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 3
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 3
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 claims description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 3
- 229910052794 bromium Inorganic materials 0.000 claims description 3
- 229910052801 chlorine Inorganic materials 0.000 claims description 3
- 239000000460 chlorine Substances 0.000 claims description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 3
- 229920002134 Carboxymethyl cellulose Polymers 0.000 claims description 2
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 claims description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 claims description 2
- 239000000440 bentonite Substances 0.000 claims description 2
- 229910000278 bentonite Inorganic materials 0.000 claims description 2
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical compound O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 claims description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 claims description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 claims description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 claims description 2
- 230000018044 dehydration Effects 0.000 claims description 2
- 238000006297 dehydration reaction Methods 0.000 claims description 2
- 239000000645 desinfectant Substances 0.000 claims description 2
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 2
- 238000000034 method Methods 0.000 claims description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 claims description 2
- 229920002545 silicone oil Polymers 0.000 claims description 2
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 claims 1
- 229940049964 oleate Drugs 0.000 claims 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 claims 1
- 229940075582 sorbic acid Drugs 0.000 claims 1
- 235000010199 sorbic acid Nutrition 0.000 claims 1
- 239000004334 sorbic acid Substances 0.000 claims 1
- 241000209140 Triticum Species 0.000 abstract description 5
- 235000021307 Triticum Nutrition 0.000 abstract description 5
- 235000016068 Berberis vulgaris Nutrition 0.000 abstract description 4
- 241000335053 Beta vulgaris Species 0.000 abstract description 4
- 206010039509 Scab Diseases 0.000 abstract description 4
- 229920000742 Cotton Polymers 0.000 abstract description 3
- 241000219146 Gossypium Species 0.000 abstract description 3
- 235000007688 Lycopersicon esculentum Nutrition 0.000 abstract description 3
- 240000003768 Solanum lycopersicum Species 0.000 abstract description 3
- 241001495426 Macrophomina phaseolina Species 0.000 abstract 1
- 244000141359 Malus pumila Species 0.000 abstract 1
- 239000003905 agrochemical Substances 0.000 abstract 1
- 235000021016 apples Nutrition 0.000 abstract 1
- 239000007788 liquid Substances 0.000 abstract 1
- 239000000725 suspension Substances 0.000 abstract 1
- CBDKQYKMCICBOF-UHFFFAOYSA-N thiazoline Chemical compound C1CN=CS1 CBDKQYKMCICBOF-UHFFFAOYSA-N 0.000 abstract 1
- 239000004563 wettable powder Substances 0.000 abstract 1
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 239000003814 drug Substances 0.000 description 6
- 241000219310 Beta vulgaris subsp. vulgaris Species 0.000 description 4
- 241001157813 Cercospora Species 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- 235000021536 Sugar beet Nutrition 0.000 description 4
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000003995 emulsifying agent Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 239000004094 surface-active agent Substances 0.000 description 3
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 2
- QGJOPFRUJISHPQ-UHFFFAOYSA-N Carbon disulfide Chemical compound S=C=S QGJOPFRUJISHPQ-UHFFFAOYSA-N 0.000 description 2
- 229920001732 Lignosulfonate Polymers 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- BPMFZUMJYQTVII-UHFFFAOYSA-N guanidinoacetic acid Chemical compound NC(=N)NCC(O)=O BPMFZUMJYQTVII-UHFFFAOYSA-N 0.000 description 2
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 2
- 239000003456 ion exchange resin Substances 0.000 description 2
- 229920003303 ion-exchange polymer Polymers 0.000 description 2
- 230000003449 preventive effect Effects 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- HUWSZNZAROKDRZ-RRLWZMAJSA-N (3r,4r)-3-azaniumyl-5-[[(2s,3r)-1-[(2s)-2,3-dicarboxypyrrolidin-1-yl]-3-methyl-1-oxopentan-2-yl]amino]-5-oxo-4-sulfanylpentane-1-sulfonate Chemical compound OS(=O)(=O)CC[C@@H](N)[C@@H](S)C(=O)N[C@@H]([C@H](C)CC)C(=O)N1CCC(C(O)=O)[C@H]1C(O)=O HUWSZNZAROKDRZ-RRLWZMAJSA-N 0.000 description 1
- WSWCOQWTEOXDQX-MQQKCMAXSA-M (E,E)-sorbate Chemical compound C\C=C\C=C\C([O-])=O WSWCOQWTEOXDQX-MQQKCMAXSA-M 0.000 description 1
- WZZBNLYBHUDSHF-DHLKQENFSA-N 1-[(3s,4s)-4-[8-(2-chloro-4-pyrimidin-2-yloxyphenyl)-7-fluoro-2-methylimidazo[4,5-c]quinolin-1-yl]-3-fluoropiperidin-1-yl]-2-hydroxyethanone Chemical compound CC1=NC2=CN=C3C=C(F)C(C=4C(=CC(OC=5N=CC=CN=5)=CC=4)Cl)=CC3=C2N1[C@H]1CCN(C(=O)CO)C[C@@H]1F WZZBNLYBHUDSHF-DHLKQENFSA-N 0.000 description 1
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 1
- TWFZGCMQGLPBSX-UHFFFAOYSA-N Carbendazim Natural products C1=CC=C2NC(NC(=O)OC)=NC2=C1 TWFZGCMQGLPBSX-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 241000223195 Fusarium graminearum Species 0.000 description 1
- VLYXMZXKAWASTM-UHFFFAOYSA-N I.CC=1SCC(N1)S Chemical compound I.CC=1SCC(N1)S VLYXMZXKAWASTM-UHFFFAOYSA-N 0.000 description 1
- 235000019738 Limestone Nutrition 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 239000005842 Thiophanate-methyl Substances 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 229940095054 ammoniac Drugs 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 239000006013 carbendazim Substances 0.000 description 1
- JNPZQRQPIHJYNM-UHFFFAOYSA-N carbendazim Chemical compound C1=C[CH]C2=NC(NC(=O)OC)=NC2=C1 JNPZQRQPIHJYNM-UHFFFAOYSA-N 0.000 description 1
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 description 1
- 239000003729 cation exchange resin Substances 0.000 description 1
- 239000003093 cationic surfactant Substances 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000003337 fertilizer Substances 0.000 description 1
- 230000000855 fungicidal effect Effects 0.000 description 1
- 239000000417 fungicide Substances 0.000 description 1
- 230000002070 germicidal effect Effects 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical group 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 239000006028 limestone Substances 0.000 description 1
- 235000014666 liquid concentrate Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000000575 pesticide Substances 0.000 description 1
- ULWHHBHJGPPBCO-UHFFFAOYSA-N propane-1,1-diol Chemical class CCC(O)O ULWHHBHJGPPBCO-UHFFFAOYSA-N 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 229940075554 sorbate Drugs 0.000 description 1
- 238000010025 steaming Methods 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 150000003440 styrenes Chemical class 0.000 description 1
- 229910021653 sulphate ion Inorganic materials 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- QGHREAKMXXNCOA-UHFFFAOYSA-N thiophanate-methyl Chemical group COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC QGHREAKMXXNCOA-UHFFFAOYSA-N 0.000 description 1
- BAZVSMNPJJMILC-UHFFFAOYSA-N triadimenol Chemical compound C1=NC=NN1C(C(O)C(C)(C)C)OC1=CC=C(Cl)C=C1 BAZVSMNPJJMILC-UHFFFAOYSA-N 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
Landscapes
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
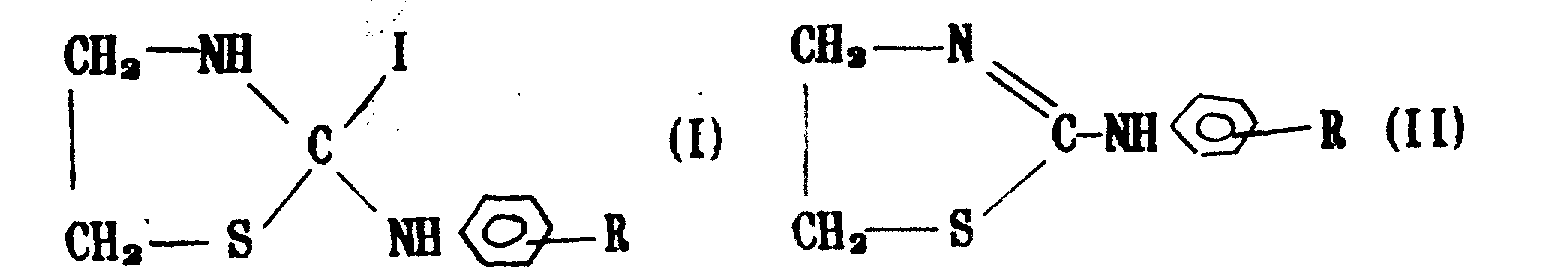
具有结构(I)和(Ⅱ)的噻唑啉化合物可用作农作物(甜菜、棉花、小麦等)的杀菌剂,用它可配成乳油、悬浮液和可湿性粉剂,使用该农药可防治甜菜褐斑病、小麦赤霉病、苹果轮纹病、番茄灰霉病、棉花立枯病,还能对甜菜起增产增糖作用。
Description
本发明属于农用杀菌剂。
目前,对于小麦赤霉病、甜菜褐斑病、苹果轮纹病、番茄灰霉病、棉花立枯病的防治,使用的杀菌剂品种较少,例如小麦赤霉病,一般使用多菌灵和甲基托布津,但这些药品长期使用其药效则下降,而甜菜褐斑病则会使作物减产、减糖。
本发明的目的是寻求一类高效杀菌剂,以防治上述农作物的各种病害。
实验证明,具有下列结构的两大类化合物(Ⅰ)和(Ⅱ)对上述农作物的病害都有很好的防治效果:
其中R可为卤素、烷氧基、烷基、羧甲基、羧基等,其各具体化合物代号见表1及表2。
表1
代号 化合物(Ⅰ)
XAH-1 (R=邻甲氧基)
XAH-2 (R=对甲基)
XAH-3 (R=对溴)
XAH-4 (R=对氯)
XAH-5 (R=羰甲基)
XAH-6 (R=羧基)
表2
代号 化合物(Ⅱ)
XAH-7 (R=邻甲氧基)
XAH-8 (R=对甲基)
XAH-9 (R=对溴)
XAH-10 (R=对氯)
XAH-11 (R=羰甲基)
XAH-12 (R=羧基)
上述化合物的制备方法是以乙醇胺为原料,与硫酸反应生成硫酸氨基乙酸内盐,然后将内盐与二硫化碳反应生成2-噻唑啉硫酮,再制备成第Ⅰ类化合物经离子交换树脂交换,制成第Ⅱ类化合物。
本发明还提供这类化合物的制剂,例如可配成乳剂、悬浮剂、可湿性粉剂等。所用溶剂有甲苯、二甲苯、丙二醇、乙二醇、乙醇和二甲基甲酰胺。用水、溶剂、润湿剂、表面活性剂和本药剂可制成悬浮剂;以石灰石、砂土、粘土、硅藻土、木质素磺酸盐、固体肥料等配制为可湿性粉剂;以溶剂和适当的乳化剂配成乳油。制备粉剂的载体可以是天然的物质,也可以是合成的物质。所采用的表面活性剂可以是阴离子表面活性剂,非离子表面活性剂和阳离子表面活性剂,例如脂肪族醇硫酸盐、十二烷基磺酸钠、脂肪酸酯、木质素磺酸盐等,也可以与其他的杀虫、杀菌剂混配使用。
通过药效测定和大田试验,证明本杀菌剂对小麦赤霉病、甜菜褐斑病均有很好的杀菌防病作用。另外,对苹果轮纹病、蕃茄灰霉病、棉花立枯病也有较好的防效作用;本药剂对甜菜有增产和增糖的作用。例如使用XAH-8杀菌剂,每亩只需用12克纯药,使用250ppm
的浓度,即可防治甜菜褐斑病,防效达65.0%,增产17.5%,增糖1.1度,而XAH-1号药剂,在室内以平板含量介质法测定,测定25ppm对小麦赤霉病菌有100%的抑制,而对照药BAYTAN 25ppm抑菌率为70.1%。
实例1
α-(邻甲氧基苯胺)基-2-噻唑啉碘氢化物(XAH-1)的制备
将2-甲硫基-2-噻唑啉碘氢化物52.0克(0.2mol)与对甲氧基苯胺24.6克(0.2mol)及甲醇200ml的四口瓶中,搅拌回流1小时,反应完毕后减压蒸去甲醇,残留物重结晶,得白色晶体60克,m.p.188℃-190℃,收率89.3%。
C H N
元素分析值% 35.94 3.99 8.19
理论值% 35.71 3.81 8.33
实例2
α-(邻甲氧基苯胺)-噻唑啉(XAH-7)的制备
将上述制得的XAH-160克,用200ml无水乙醇溶解,通过250ml的P-6905号离子交换树脂(强碱性苯乙烯系阳离子交换树脂)的交换柱,将流出液集中浓缩,得黄色晶体30.5克,收率73.3%,m.p.120-121℃
C H N
元素分析值% 57.95 5.80 13.35
理论值% 57.69 5.77 13.46
实例3
将XAH-9号化合物(合成方法同XAH-7)40克,加木质素磺酸钠4克,十二烷基磺酸钠0.5克,及聚乙烯醇0.5克,水55克和乙烯甘醇
0.1克、硅油0.1克共磨,粒度控制在2-5μ,则制成悬浮剂。
实例4
将XAH-8号化合物30克,加膨润土62克、羧甲基纤维素2.0克、木质素磺酸钠6克,则可配制成可湿性粉剂。
实例5
将XAH-12号化合物40克,加二甲苯40克,及乳化剂(失水山梨酸酯)15克、乳化剂(脂肪环氧乙烷缩合物)15克,配成一种乳油。
Claims (1)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 88107909 CN1020249C (zh) | 1988-11-16 | 1988-11-16 | 噻唑啉类农用杀菌剂 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 88107909 CN1020249C (zh) | 1988-11-16 | 1988-11-16 | 噻唑啉类农用杀菌剂 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1042706A CN1042706A (zh) | 1990-06-06 |
| CN1020249C true CN1020249C (zh) | 1993-04-14 |
Family
ID=4834830
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 88107909 Expired - Fee Related CN1020249C (zh) | 1988-11-16 | 1988-11-16 | 噻唑啉类农用杀菌剂 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN1020249C (zh) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2792503A1 (en) * | 2010-03-10 | 2011-09-15 | Galleon Pharmaceuticals, Inc. | Analgesic compounds, compositions, and uses thereof |
| CN101914096A (zh) * | 2010-08-11 | 2010-12-15 | 中国农业大学 | 2-取代芳杂环基-1,3-噻唑啉衍生物及其制备方法和应用 |
-
1988
- 1988-11-16 CN CN 88107909 patent/CN1020249C/zh not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| CN1042706A (zh) | 1990-06-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1152869C (zh) | 噻二唑类金属络合物及其制备方法和用途 | |
| AU2013228038A1 (en) | Use of a metal salt of 2-mercaptobenzothiazole | |
| RO106740B1 (ro) | Derivati ai 5-tiazolilcarboxanilidei, compozitii fungicide si metoda de tratament al plantelor cu acestea | |
| CN1020249C (zh) | 噻唑啉类农用杀菌剂 | |
| DD155384A5 (de) | Fungizide und/oder bakterizide zusammensetzungen | |
| US3917476A (en) | Diethyl alpha phosphonate as an (antimicrobial agent) algaecide | |
| DE3874462T2 (de) | Fungizide zusammensetzungen. | |
| CN101427682A (zh) | 酚菌酮与烯酰吗啉杀菌组合物 | |
| JPH0655708B2 (ja) | スルホンアミド系化合物及び農業用殺菌剤 | |
| CN102835413A (zh) | 防治赤霉病的复配杀菌剂 | |
| CN102150675A (zh) | 一种含有克菌丹与硫代氨基甲酸酯类化合物的杀菌组合物 | |
| CN118252147A (zh) | 一种含氟咯菌腈的杀菌剂及其制备方法 | |
| CN101703053B (zh) | 一种含有噁唑菌酮的杀菌组合物 | |
| DE2123830A1 (de) | Neue Thiocyanatomethylhydantoinverbindungen | |
| CN107691456B (zh) | 一种含有氟嘧菌酯和申嗪霉素的杀菌组合物 | |
| CN109984139A (zh) | 一种含氯氟醚菌唑和宁南霉素的杀菌组合物 | |
| US3318767A (en) | Biocidal compositions containing 4, 5-dicyano-1, 3-dithiole-2-thione oxide | |
| RU2832090C1 (ru) | Фунгицидный и бактерицидный препарат и способ его использования | |
| CN104430425B (zh) | 一种含有硝苯菌酯和丙硫咪唑的杀菌组合物 | |
| CN116326591B (zh) | 一种农业杀螨组合物及其应用 | |
| AU2015224520A1 (en) | Use of a metal salt of 2-mercaptobenzothiazole | |
| SU296304A1 (zh) | ||
| CN102283225A (zh) | 一种含丁香菌酯和保护性杀菌剂的杀菌组合物 | |
| CN102067860B (zh) | 一种多菌灵微乳剂及其制备方法 | |
| CN109042688A (zh) | 一种苯醚甲环唑·丙环唑杀菌组合物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C19 | Lapse of patent right due to non-payment of the annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |