WO2023209788A1 - Appareil et procédé de production d'hydroxyapatite contenant un métal antibactérien - Google Patents
Appareil et procédé de production d'hydroxyapatite contenant un métal antibactérien Download PDFInfo
- Publication number
- WO2023209788A1 WO2023209788A1 PCT/JP2022/018811 JP2022018811W WO2023209788A1 WO 2023209788 A1 WO2023209788 A1 WO 2023209788A1 JP 2022018811 W JP2022018811 W JP 2022018811W WO 2023209788 A1 WO2023209788 A1 WO 2023209788A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- container
- solution
- suspension
- agha
- injection device
- Prior art date
Links
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 50
- 230000000844 anti-bacterial effect Effects 0.000 title claims abstract description 36
- 229910052588 hydroxylapatite Inorganic materials 0.000 title claims abstract description 21
- XYJRXVWERLGGKC-UHFFFAOYSA-D pentacalcium;hydroxide;triphosphate Chemical compound [OH-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O XYJRXVWERLGGKC-UHFFFAOYSA-D 0.000 title claims abstract description 21
- 229910052751 metal Inorganic materials 0.000 title claims abstract description 20
- 239000002184 metal Substances 0.000 title claims abstract description 20
- 239000000243 solution Substances 0.000 claims abstract description 119
- 239000000725 suspension Substances 0.000 claims abstract description 119
- 238000002347 injection Methods 0.000 claims abstract description 88
- 239000007924 injection Substances 0.000 claims abstract description 88
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims abstract description 68
- SQGYOTSLMSWVJD-UHFFFAOYSA-N silver(1+) nitrate Chemical compound [Ag+].[O-]N(=O)=O SQGYOTSLMSWVJD-UHFFFAOYSA-N 0.000 claims abstract description 52
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 claims abstract description 49
- 239000000920 calcium hydroxide Substances 0.000 claims abstract description 48
- 229910001861 calcium hydroxide Inorganic materials 0.000 claims abstract description 48
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims abstract description 34
- 229910001961 silver nitrate Inorganic materials 0.000 claims abstract description 26
- 239000011259 mixed solution Substances 0.000 claims abstract description 18
- 229910000365 copper sulfate Inorganic materials 0.000 claims abstract description 8
- XTVVROIMIGLXTD-UHFFFAOYSA-N copper(II) nitrate Chemical compound [Cu+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O XTVVROIMIGLXTD-UHFFFAOYSA-N 0.000 claims abstract description 8
- ARUVKPQLZAKDPS-UHFFFAOYSA-L copper(II) sulfate Chemical compound [Cu+2].[O-][S+2]([O-])([O-])[O-] ARUVKPQLZAKDPS-UHFFFAOYSA-L 0.000 claims abstract description 8
- 239000004745 nonwoven fabric Substances 0.000 claims description 100
- 239000007788 liquid Substances 0.000 claims description 52
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 41
- 239000000203 mixture Substances 0.000 claims description 24
- 238000004062 sedimentation Methods 0.000 claims description 24
- 238000001035 drying Methods 0.000 claims description 15
- NDVLTYZPCACLMA-UHFFFAOYSA-N silver oxide Chemical compound [O-2].[Ag+].[Ag+] NDVLTYZPCACLMA-UHFFFAOYSA-N 0.000 claims description 14
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 claims description 7
- 229910017604 nitric acid Inorganic materials 0.000 claims description 7
- 229910001923 silver oxide Inorganic materials 0.000 claims description 7
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical group [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 claims description 6
- 229910052709 silver Inorganic materials 0.000 claims description 6
- 239000004332 silver Substances 0.000 claims description 6
- 239000007921 spray Substances 0.000 claims 1
- 238000002156 mixing Methods 0.000 abstract description 8
- 239000002245 particle Substances 0.000 description 14
- 238000010586 diagram Methods 0.000 description 13
- 238000000034 method Methods 0.000 description 13
- 238000003756 stirring Methods 0.000 description 13
- 230000015572 biosynthetic process Effects 0.000 description 10
- 238000003786 synthesis reaction Methods 0.000 description 10
- 239000006228 supernatant Substances 0.000 description 9
- 239000011248 coating agent Substances 0.000 description 8
- 238000000576 coating method Methods 0.000 description 8
- 238000003860 storage Methods 0.000 description 8
- 241000894006 Bacteria Species 0.000 description 7
- 230000001877 deodorizing effect Effects 0.000 description 7
- 229910052710 silicon Inorganic materials 0.000 description 7
- 239000010703 silicon Substances 0.000 description 7
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 6
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 6
- 229910052791 calcium Inorganic materials 0.000 description 6
- 239000011575 calcium Substances 0.000 description 6
- 239000010949 copper Substances 0.000 description 6
- 230000002950 deficient Effects 0.000 description 6
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 5
- 238000002474 experimental method Methods 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 238000004804 winding Methods 0.000 description 5
- 229910001385 heavy metal Inorganic materials 0.000 description 4
- 230000002209 hydrophobic effect Effects 0.000 description 4
- 230000002776 aggregation Effects 0.000 description 3
- 238000004220 aggregation Methods 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- 241000588724 Escherichia coli Species 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- -1 copper Chemical class 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 239000000551 dentifrice Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 150000002739 metals Chemical class 0.000 description 2
- 239000002105 nanoparticle Substances 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 238000009736 wetting Methods 0.000 description 2
- 229910014497 Ca10(PO4)6(OH)2 Inorganic materials 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 238000007664 blowing Methods 0.000 description 1
- 229910052793 cadmium Inorganic materials 0.000 description 1
- BDOSMKKIYDKNTQ-UHFFFAOYSA-N cadmium atom Chemical compound [Cd] BDOSMKKIYDKNTQ-UHFFFAOYSA-N 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 238000007598 dipping method Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 239000006196 drop Substances 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- GPRLSGONYQIRFK-UHFFFAOYSA-N hydron Chemical compound [H+] GPRLSGONYQIRFK-UHFFFAOYSA-N 0.000 description 1
- 230000008676 import Effects 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 229910052753 mercury Inorganic materials 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 150000003376 silicon Chemical class 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- 238000007711 solidification Methods 0.000 description 1
- 230000008023 solidification Effects 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 229910052716 thallium Inorganic materials 0.000 description 1
- BKVIYDNLLOSFOA-UHFFFAOYSA-N thallium Chemical compound [Tl] BKVIYDNLLOSFOA-UHFFFAOYSA-N 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N59/00—Biocides, pest repellants or attractants, or plant growth regulators containing elements or inorganic compounds
- A01N59/26—Phosphorus; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K6/00—Preparations for dentistry
- A61K6/70—Preparations for dentistry comprising inorganic additives
- A61K6/71—Fillers
- A61K6/74—Fillers comprising phosphorus-containing compounds
- A61K6/75—Apatite
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
- A61K8/24—Phosphorous; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/18—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing inorganic materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/02—Inorganic materials
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B25/00—Phosphorus; Compounds thereof
- C01B25/16—Oxyacids of phosphorus; Salts thereof
- C01B25/26—Phosphates
- C01B25/45—Phosphates containing plural metal, or metal and ammonium
Definitions
- the present invention relates to an apparatus and method for producing antibacterial metal-containing hydroxyapatite.
- HA Hydroxyapatite
- Ca 10 (PO 4 ) 6 (OH) 2 An example of a method for producing HA is a solution method. Generally, HA is transported, sold, and used in dry powder form. HA aggregates when dried. The particle size of aggregated HA becomes micro-sized.
- Embodiments of the present invention provide an apparatus and method for producing antibacterial metal-containing HA that has a large bacterium-inhibiting effect.
- a manufacturing apparatus includes a first injection device, a second injection device, and a control device.
- a first injection device injects calcium hydroxide into the container.
- the second injection device injects silver nitrate, copper nitrate, or a mixed solution of copper sulfate and phosphoric acid into the solution contained in the container.
- the control device controls at least one of the concentration and injection rate of the liquid mixture injected by the second injection device so that the pH of the solution in the container as measured by the pH meter falls within a predetermined range. , prepare a light gray or white antibacterial metal-containing hydroxyapatite suspension in a container.
- a manufacturing device includes a first injection device, a second injection device, and a control device.
- the first injection device injects silver nitrate, copper nitrate, or a mixed solution of copper sulfate and calcium hydroxide into the container.
- the second injection device injects phosphoric acid into the solution contained in the container.
- the control device controls at least one of the concentration and injection rate of the phosphoric acid injected by the second injection device so that the pH of the solution in the container as measured by the pH meter falls within a predetermined range. , prepare a light gray or white antibacterial metal-containing hydroxyapatite suspension in a container.
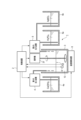
- FIG. 1 is a block diagram showing an example of the configuration of a manufacturing apparatus according to the first embodiment.
- FIG. 2 is a conceptual diagram showing an example of a mixed state of calcium hydroxide and a first liquid mixture containing phosphoric acid and silver nitrate by the manufacturing apparatus according to the first embodiment.
- FIG. 3 is a flowchart illustrating an example of a suspension production method executed by the production apparatus according to the first embodiment.
- FIG. 4 is a conceptual diagram showing an example of natural sedimentation in the suspension preparation method according to the first embodiment.
- FIG. 5 is a diagram showing an example of the relationship between the pH and color of a solution.
- FIG. 6 is a diagram showing an example of the results of applying gray AgHA, light gray AgHA, and white AgHA to a nonwoven fabric and investigating the antibacterial activity value using Escherichia coli. It is a figure which shows the example of the measurement result of the antibacterial activity value of.
- Figure 7 shows a nonwoven fabric coated with a HA solution with a concentration of 0.5% by weight, a nonwoven fabric coated with a light gray AgHA solution with a concentration of 0.5%, and a nonwoven fabric coated with a white AgHA solution with a concentration of 0.5%. It is a graph showing an example of deodorizing effect with.
- FIG. 8 is a conceptual diagram showing an example of a mixed state of a second liquid mixture containing calcium hydroxide and silver nitrate and phosphoric acid by the manufacturing apparatus according to the second embodiment.
- FIG. 9 is a conceptual diagram showing an example of the configuration of a coating device for coating a nonwoven fabric with AgHA according to the third embodiment.
- FIG. 10 is a side view showing an example of a shower head and an ultrasonic vibrator.
- FIG. 11 is a perspective view showing an example of the upper surface of the support base.
- FIG. 12 is a flowchart illustrating an example of a method for applying AgHA to a nonwoven fabric, which is executed by the application apparatus according to the third embodiment.
- a production apparatus and method for producing antibacterial metal-containing HA having a large bacteria-inhibiting effect and deodorizing effect using a solution method will be described. More specifically, in the first embodiment, a production apparatus and method for producing light gray or white antibacterial metal-containing HA by controlling pH (hydrogen ion concentration index) will be described.
- the antibacterial metal may be a heavy metal.
- heavy metals having antibacterial properties include silver, copper, palladium, platinum, cadmium, nickel, cobalt, zinc, manganese, thallium, lead, and mercury.
- AgHA an apparatus and method for producing HA (hereinafter referred to as AgHA) containing silver (Ag) among metals having antibacterial properties will be described as an example. Note that it is also possible to produce HA containing other antibacterial metals, such as copper, by appropriately modifying the first embodiment.
- AgHA contained in the AgHA suspension produced according to the first embodiment can maintain a nanosize.
- FIG. 1 is a block diagram showing an example of the configuration of a manufacturing apparatus 1 according to the first embodiment.
- the manufacturing apparatus 1 injects (for example, drops) a first mixed solution containing phosphoric acid and silver nitrate into calcium hydroxide, and produces light gray by controlling the pH of the solution containing calcium hydroxide and the first mixed solution. Or make white AgHA.
- CuHA may be produced using copper nitrate or copper sulfate instead of silver nitrate.
- the production apparatus 1 may produce light gray or white AgHA by injecting a mixed solution of silver oxide, nitric acid, and phosphoric acid into calcium hydroxide.
- a mixed solution of silver oxide, nitric acid, and phosphoric acid it is difficult to produce light gray or white AgHA.
- a mixture of silver oxide, nitric acid, and phosphoric acid is used, light gray or white AgHA can be produced.
- the production apparatus 1 uses synthetic conditions that are lower than when using the first mixed solution containing phosphoric acid and silver nitrate. Set the pH used as a low value.
- the production device 1 includes a control device 2, a first container 3, a first injection device 4, a second injection device 5, a pH meter 6, a thermometer 7, a temperature adjustment device 8, a stirrer 37, and a first pouring device.
- a device 9 and a second pouring device 10 are provided.
- a light gray or white AgHA suspension 11 produced and concentrated by the production apparatus 1 is stored in a second container 12 .
- the various devices constituting the manufacturing apparatus 1 may be combined as appropriate.
- the pH meter 6 and the thermometer 7 may be one device.
- the first pouring device 9 and the second pouring device 10 may be one pump.
- the various devices constituting the manufacturing apparatus 1 may be separated as appropriate.
- the first container 3 is a container used for dropping a first liquid mixture containing phosphoric acid and silver nitrate onto calcium hydroxide, and the solution 13 which is a mixture of calcium hydroxide and the first liquid mixture. It may be divided into a container used for carrying out natural sedimentation.
- the control device 2 receives a signal indicating the pH measured by the pH meter 6 and a signal indicating the temperature measured by the thermometer 7.
- the control device 2 controls the amount of calcium hydroxide injected into the first container 3 by the first injection device 4 based on the pH indicated by the signal received from the pH meter 6 and the temperature indicated by the signal received from the thermometer 7.
- concentration, at least one of the concentration and injection rate (for example, dropping rate) of the first liquid mixture injected (for example, dropped) into the first container 3 by the second injection device 5, the pH of the solution 13 (synthesis pH), the temperature control of the solution 13 during AgHA synthesis and natural precipitation by the temperature control device 8 is controlled.
- the control device 2 may perform control to intentionally create calcium-deficient AgHA by additionally dropping the first mixed liquid after dropping the first mixed liquid and lowering the pH of the solution 13.
- control device 2 controls the concentration of calcium hydroxide so that the concentration of calcium hydroxide falls within the first range.
- the first range may be, for example, 0.01% or more and 50% or less.
- the control device 2 controls the concentration of the first liquid mixture so that the concentration of phosphoric acid is within the second range and the concentration of silver nitrate is within the third range.
- the second range may be, for example, 0.01% or more and 50% or less.
- the third range may be, for example, 0.01% or more and 50% or less.
- the control device 2 controls the injection rate of the first mixed liquid so that the injection rate of the first mixed liquid is within the fourth range with respect to the production amount of AgHA.
- the fourth range may be, for example, 0.01 ml/min/g or more and 100 ml/min/g or less.
- the fourth range may preferably be 0.1 ml/min/g or more and 10 ml/min/g or less.
- the control device 2 controls the injection amount of at least one of calcium hydroxide and the first liquid mixture so that the pH of the solution 13 in the first container 3 falls within the fifth range.
- the fifth range may be, for example, the pH at which light gray or white AgHA is produced.
- AgHA has a color such as brown, gray, light gray, or white, for example.
- the controller 2 controls the pH of the solution 13 in the first container 3 and controls the first mixing so that the solution 13 in the first container 3 is light gray or white. Control the amount of liquid injected.
- control device 2 may perform control so that the pH of the solution 13 in the first container 3 is 4 or more and 12 or less, thereby producing light gray AgHA.
- control device 2 may perform control so that the pH of the solution 13 in the first container 3 is 2 or more and 10 or less to produce white AgHA.
- the synthetic pH of AgHA used in the control device 2 is adjusted as appropriate depending on the purity, temperature, and Ag content of the raw materials.
- the control device 2 performs control so that the content of Ag with respect to HA is 0.01% by weight or more and 30% by weight or less, more preferably 0.1% by weight or more and 15% by weight or less.
- the content of Ag relative to HA is 0.4% by weight or more, the antibacterial properties of AgHA are greatly improved.
- the control device 2 controls the synthesis temperature at which the AgHA suspension is synthesized by mixing calcium hydroxide and the first mixed liquid, and the temperature during the reaction between the calcium hydroxide and the first mixed liquid at a sixth temperature.
- the temperature of the solution 13 is controlled to be within the range.
- the sixth range may be, for example, 5° C. or higher and 50° C. or lower.
- the sixth range may preferably be, for example, 5° C. or higher and 30° C. or lower.
- the temperature of the solution 13 tends to rise due to reaction heat, but the operation of the control device 2 and temperature adjustment device 8 can realize a state suitable for AgHA production.
- the control device 2 may include, for example, a storage device 2a and a processor 2b.
- the storage device 2a stores the software 2c.
- the storage device 2a may include a non-temporary storage device and a temporary storage device such as a cache memory.
- the software 2c may include a program and data.
- the processor 2b executes the software 2c stored in the storage device 2a, and executes various controls for producing the light gray or white AgHA suspension 11.
- the control device 2 executes control to cause the stirrer 37 to stir the solution 13 in the container 3 during mixing of calcium hydroxide and the first liquid mixture (AgHA synthesis). More specifically, the control device 2 transmits a signal indicating the rotational speed to the stirrer 37 when the second injection device 5 performs the dropping process of the first liquid mixture. For example, the control device 2 transmits to the stirrer 37 a signal for rotating the stirrer 37 at, for example, 50 rpm or more and 1000 rpm or less. More preferably, the control device 2 transmits to the stirrer 37 a signal for rotating the stirrer 37 at, for example, 100 rpm or more and 500 rpm or less.
- the controller 2 After pouring out the supernatant portion of the solution 13 from the first container 3 and before pouring out the concentrated AgHA suspension 11 from the first container 3, the controller 2 causes the stirrer 37 to The control for stirring the concentrated AgHA suspension 11 in the container 3 is executed. More specifically, the control device 2 transmits an instruction indicating the rotational speed to the stirrer 37 before pouring out the concentrated AgHA suspension 11. For example, the control device 2 transmits to the stirrer 37 a signal for rotating the stirrer 37 at, for example, 50 rpm or more and 1000 rpm or less. More preferably, the control device 2 transmits to the stirrer 37 a signal for rotating the stirrer 37 at, for example, 100 rpm or more and 500 rpm or less. By stirring with the stirrer 37, the concentration of the concentrated light gray or white AgHA suspension 11 can be made uniform.
- the control device 2 instructs the first pouring device 9 to pour out the supernatant portion of the solution 13 contained in the first container 3.
- the control device 2 instructs the second pouring device 10 to pour out the sedimented portion (i.e., the concentrated AgHA suspension 11) of the solution 13 contained in the first container 3. do.
- the first container 3 contains calcium hydroxide (liquid) injected from the first injection device 4.
- the first container 3 receives the first liquid mixture injected (for example, dropped) by the second injection device 4.
- the first injection device 4 receives a signal indicating the concentration of calcium hydroxide from the control device 2.
- the first injection device 4 adjusts the concentration of calcium hydroxide based on the received signal, and injects the adjusted concentration calcium hydroxide into the first container 3.
- the purity of calcium hydroxide may be 90% or more and 100% or less.
- the second injection device 5 receives from the control device 2 a signal indicating the concentration of phosphoric acid, a signal indicating the concentration of silver nitrate, and a signal indicating the injection speed of the first mixed liquid.
- the second injection device 5 mixes phosphoric acid at a concentration indicated by the received signal and silver nitrate at a concentration indicated by the signal to be received to generate a first mixed solution, and It is dripped into the solution 13 containing calcium hydroxide stored in the first container 3 at the injection rate indicated by the signal.
- the pH meter 6 measures the pH of the solution 13 stored in the first container 3 and sends a signal indicating the pH to the control device 2.
- the thermometer 7 measures the temperature of the solution 13 stored in the first container 3 and sends a signal indicating the temperature to the control device 2.
- the temperature adjustment device 8 receives a signal indicating the temperature from the control device 2.
- the temperature control device 8 controls the signal received by the solution 13 in the first container 3 during mixing of calcium hydroxide and the first liquid mixture (during AgHA synthesis) and while performing natural sedimentation on the solution 13.
- the temperature of the solution 13 in the first container 3 is controlled (eg, heated or cooled) so that the temperature indicated by is reached.
- the stirrer 37 receives a signal indicating the rotation speed from the control device 2.
- the stirrer 37 stirs the solution 13 in the container 3 according to the rotational speed indicated by the received signal.
- the stirrer 37 stirs the solution 13 when dropping the first liquid mixture (during AgHA synthesis). Further, the stirrer 37 stirs the concentrated AgHA suspension 11 before pouring out the concentrated AgHA suspension 11. There is a concentration gradient in the settling part after the supernatant part is poured out.
- the concentration of the sedimented portion can be made uniform by stirring the sedimented portion.
- the first dispensing device 9 receives a signal indicating a dispensing instruction from the control device 2.
- the first pouring device 9 pours out the supernatant portion of the solution 13 from the first container 3 based on the received signal, and pours out the sedimented portion of the solution 13 (i.e., the concentrated light gray or white AgHA suspension). 11) is left in the first container 3.
- the water intake port of the first pouring device 9 is movable up and down depending on the boundary position between the supernatant portion and the sedimentation portion. More specifically, the height of the water intake port of the first pouring device 9 is adjusted so that it is above the boundary position between the supernatant portion and the settling portion. Thereby, the supernatant portion can be efficiently poured out and the sedimented portion can be left in the first container 3.
- the second dispensing device 10 receives a signal indicating a dispensing instruction from the control device 2. Based on the received signal, the second pouring device 10 converts the sedimented portion of the solution 13 in the first container 3 into a concentrated light gray or white AgHA suspension 11 into a second container 12 for transportation. Inject into.
- FIG. 2 is a conceptual diagram showing an example of the mixing state of calcium hydroxide and the first mixed liquid (AgHA synthesis state) by the production apparatus 1 according to the first embodiment.
- the first pouring device 9 and the second pouring device 10 of the manufacturing apparatus 1 are omitted.
- the third container 4a contains a calcium hydroxide solution.
- the first injection device 4 adjusts the concentration of calcium hydroxide to the concentration indicated by the signal received from the control device 2, and injects the calcium hydroxide with the adjusted concentration into the first container 3.
- the fourth container 5a contains a solution of phosphoric acid.
- the fifth container 5b contains a silver nitrate solution.
- the second injection device 5 adjusts the concentration of phosphoric acid to the concentration indicated by the signal received from the control device 2. Further, the second injection device 5 adjusts the concentration of silver nitrate to the concentration indicated by the signal received from the control device 2. Further, the second injection device 5 mixes phosphoric acid and nitric acid to generate a first mixed liquid. Then, the second injection device 5 drips the first liquid mixture whose concentration has been adjusted into the first container 3 at a dropping speed indicated by the signal received from the control device 2 .
- the control device 2 receives a signal indicating the pH of the solution 13 from the pH meter 6, and controls the operation of the first injection device 4 and the second injection device 5 and the stirring of the stirrer 37 based on the pH of the solution 13. Control processing.
- the control device 2 receives a signal indicating the temperature of the solution 13 from the thermometer 7, and controls the operation of the temperature adjustment device 8 based on the temperature of the solution 13.
- the first injection device 4 and the second injection device 5 may be, for example, tube pumps.
- a fourth container 5a containing phosphoric acid and a fifth container 5b containing silver nitrate are used separately.
- the fourth container 5a and the fifth container 5b may be combined into one container.
- this one container contains a first liquid mixture obtained by mixing phosphoric acid and silver nitrate in advance.
- the second injection device 5 sucks the first liquid mixture in this one container and drips it into the solution 13 in the first container 3.
- FIG. 3 is a flowchart illustrating an example of a method for producing a light gray or white AgHA suspension 11 performed by the production apparatus 1 according to the first embodiment. The manufacturing method shown in FIG. 3 is executed under the control of the control device 2.
- the first injection device 4 injects calcium hydroxide at a concentration specified by the control device 2 into the first container 3.
- the stirrer 37 operates at the rotation speed specified by the control device 2 to stir the solution 13 in the first container 3.
- the second injection device 5 drops the first liquid mixture into the solution 13 (calcium hydroxide) in the first container 3 at the concentration and injection rate specified by the control device 2.
- the pH meter 6 measures the pH of the solution 13 in the first container 3 and sends a signal indicating the pH to the control device 2, and the thermometer 7 measures the temperature of the solution 13 in the first container 3. A signal indicating the measured temperature is transmitted to the control device 2.
- control device 2 determines that the relationship between the concentration of calcium hydroxide, the concentration and injection rate of the first mixed liquid, the pH of the solution 13 in the first container 3, and the temperature of the solution 13 is that AgHA is light gray or white. Determine whether or not the synthesis condition is satisfied.
- control device 2 determines that the synthesis conditions are not satisfied, in S306, the control device 2 controls the new concentration of calcium hydroxide, the new concentration of the first mixed liquid, the new injection rate of phosphoric acid, and the new concentration of the first mixed liquid. Determine the pH, the new temperature of the solution 13 in the first container 3. The control device 2 then transmits a signal indicating the new concentration of calcium hydroxide to the first injection device 4. The first injection device 4 adjusts the concentration of calcium hydroxide based on the signal received from the control device 2. The control device 2 transmits a signal indicating the concentration of the new first mixed liquid and a signal indicating the injection speed of the new first mixed liquid to the second injection device 5.
- the second injection device 5 adjusts the concentration and injection rate of the first mixed liquid based on the signal received from the control device 2.
- the control device 2 sends a signal indicating the new temperature to the temperature adjustment device 8.
- Temperature adjustment device 8 adjusts the temperature of solution 13 based on the signal received from control device 2 . After that, the process returns to S301.
- control device 2 does not need to determine the new concentration of calcium hydroxide, and sends a signal indicating the new concentration of calcium hydroxide to the first injection device 4. There is no need to send it to . In this case, the process returns to S302.
- control device 2 determines in S305 that the synthesis conditions are satisfied, in S307 the control device 2 determines whether the termination conditions for the operation of dropping the first mixed liquid onto calcium hydroxide (dropping termination conditions) are satisfied. to judge.
- the dropping end condition may be, for example, that the pH of the solution 13 in the first container 3 has reached the target range (target pH).
- the target pH is, for example, a pH at which the ratio of AgHA produced is equal to or higher than a threshold value and the AgHA produced becomes light gray or white.
- the control device 2 causes the second injection device 5 to drip the first mixed liquid again as appropriate, so that the pH of the solution 13 increases.
- the condition is stable, it may be determined that the drop termination condition is satisfied.
- the dropping termination conditions are, for example, that the ratio of AgHA to be produced is equal to or higher than a threshold value, the pH is such that the AgHA to be produced is light gray or white, and the amount of solution 13 in the first container 3 is below the threshold value. It may also be said to exceed.
- the control device 2 may intentionally produce calcium-deficient AgHA by additionally dropping the first mixed liquid after dropping the first mixed liquid and lowering the pH of the solution 13.
- control device 2 determines that the termination conditions are not satisfied, the process returns to S301. Note that in S306 of the first embodiment, if the control device 2 does not determine a new concentration of calcium hydroxide, the process may return to S302.
- control device 2 determines that the end condition is satisfied, in S308, the control device 2 transmits a stop signal to the first injection device 4, the second injection device 5, and the stirrer 37.
- the first injection device 4 and the second injection device 5 stop their injection operations in accordance with the signal received from the control device 2.
- the stirrer 37 stops its stirring operation in accordance with the signal received from the control device 2.
- control device 2 transmits a signal indicating a temperature suitable for natural sedimentation to the temperature adjustment device 8.
- Temperature adjustment device 8 adjusts the temperature of solution 13 based on the signal received from control device 2 .
- step S310 natural sedimentation of the solution 13 in the first container 3 is performed for a predetermined period of time or more.
- the first pouring device 9 pours out the supernatant portion of the solution 13 in the first container 3 under the control of the control device 2, and leaves the sedimented portion in the first container 3.
- the stirring device 37 stirs the concentrated light gray or white AgHA suspension 11, which is the sedimented portion in the first container 3, under the control of the control device 2.
- the second pouring device 10 pours out the concentrated light gray or white AgHA suspension 11 in the first container 3 under the control of the control device 2, and stores it in the second container 12. do.
- FIG. 4 is a conceptual diagram showing an example of natural sedimentation in the method for producing the AgHA suspension 11 according to the first embodiment.
- the AgHA-containing solution 13 prepared in the first container 3 is subjected to natural sedimentation for a predetermined period of time.
- the predetermined time for natural sedimentation may be, for example, 1 day or more and 60 days or less, more preferably 9 days or more and 28 days or less.
- Nanosized AgHA in the solution 13 slowly settles. Removal of the supernatant portion of solution 13 leaves a concentrated AgHA suspension 11, which is the sedimented portion of solution 13. AgHA contained in this AgHA suspension 11 maintains a nanosize.
- a dentifrice in which nano-sized AgHA is diffused can be produced.
- the target pH is a pH at which the concentrated AgHA suspension 11 becomes light gray or white.
- the AgHA suspension 11 is prepared using a first mixed solution of phosphoric acid and silver nitrate.
- a manufacturing method that uses silver oxide instead of silver nitrate as the Ag source, but since silver oxide does not dissolve in phosphoric acid, silver nitrate is used as the Ag source in the first embodiment.
- Cu may also be used as the antibacterial heavy metal.
- Cu(NO 3 ), Cu(NO 4 ), or Cu(NO 3 ) 2 is used instead of silver nitrate to prepare a CuHA suspension containing CuHA.
- FIG. 5 is a diagram showing an example of the relationship between the pH and color of the solution 13.
- control device 2 controls the pH of the solution 13 so that the color of AgHA is light gray or white and the ratio of AgHA in the solution 13 exceeds a predetermined value. .
- a crystal of calcium-deficient AgHA has a structure in which the amount of Ag uptake is greater than that of an AgHA crystal. Therefore, the higher the ratio of calcium-deficient AgHA, the higher the antibacterial effect of the produced AgHA suspension 11.
- White AgHA contains more calcium-deficient AgHA and has a higher Ag uptake than gray AgHA.
- FIG. 6 is a diagram showing an example of the results of applying gray AgHA, light gray AgHA, and white AgHA to a nonwoven fabric and investigating the antibacterial activity value using Escherichia coli.
- FIG. 6 shows the results of investigating antibacterial activity values for a nonwoven fabric coated with a 0.1% AgHA solution and a nonwoven fabric coated with a 0.5% AgHA solution.
- AgHA contained in the AgHA solution the Ag content with respect to HA is 2% by weight.
- An antibacterial activity value of 2 or more and less than 3 indicates an antibacterial effect, and a value of 3 or more indicates a large antibacterial effect.
- Figure 7 shows a nonwoven fabric coated with a HA solution with a concentration of 0.5% by weight, a nonwoven fabric coated with a light gray AgHA solution with a concentration of 0.5%, and a nonwoven fabric coated with a white AgHA solution with a concentration of 0.5%. It is a graph showing an example of deodorizing effect with. In FIG. 7, the vertical axis indicates the reduction rate of ammonia odor. In the AgHA shown in FIG. 7, the Ag content with respect to HA is 2% by weight.
- Light gray AgHA has a higher deodorizing effect than HA and gray AgHA.
- the deodorizing effect of white AgHA is approximately 4 to 5 times higher than that of light gray AgHA.
- the light gray or white AgHA produced in the first embodiment has strong bactericidal and deodorizing effects.
- a solution 13 is prepared and concentrated, and the concentrated AgHA suspension 11 is stored in a second container 12. 2 containers 12 are transported.
- the solution 13 containing AgHA having a small particle size can be adjusted.
- the number of bacteria can be controlled by preparing and efficiently concentrating the solution 13 and adjusting the pH of the solution 13.
- AgHA is shipped and used as a suspension. Therefore, aggregation of AgHA particles can be suppressed, and AgHA with small particle size can be used.
- concentration is performed by natural sedimentation.
- concentration using a centrifuge causes AgHA particles to aggregate, making redispersion difficult.
- concentration since concentration is performed by natural sedimentation, aggregation of AgHA particles can be suppressed. By concentrating it, the transport of the AgHA suspension 11 can be made more efficient and easier to handle.
- the period required for natural sedimentation varies depending on the temperature of the solution 13 in the first container 3. For example, in experiments, the number of days required to concentrate to about 3% was 9 to 28 days. It was obtained from data obtained by preparing 200 100 L AgHA suspensions 11 that natural sedimentation is affected by the temperature atmosphere. Based on this result, in the first embodiment, the control device 2 controls the temperature of the solution 13 in the first container 3 to a temperature suitable for natural sedimentation. In the production apparatus 1 according to the first embodiment, the sedimentation period can be shortened by performing the natural sedimentation at a temperature of 5° C. or higher and 40° C. or lower, and the solution 13 can be efficiently concentrated to form the AgHA suspension 11. can be created.
- the concentrated suspension 11 is stirred by the stirrer 37 and then stored in the second container 12. Therefore, in the first embodiment, the concentration of the concentrated suspension 11 can be made uniform.
- the effect of suppressing the number of bacteria can be obtained by controlling the storage temperature of the solution 13 and the concentrated AgHA suspension 11 to a range of 2° C. or higher and 30° C. or lower. More preferably, the number of bacteria can be suppressed by controlling the storage temperature of the solution 13 and the concentrated AgHA suspension 11 to a range of 2° C. or higher and 20° C. or lower.
- control device 2 and the temperature adjustment device 8 control the temperature of the solution 13 and the temperature of the concentrated AgHA suspension 11 in a range of 2°C or more and 30°C or less, more preferably 2°C or more and 20°C or less. It may be controlled within a range.
- the control device 2 and the temperature adjustment device 8 control the temperature of the solution 13 during the sedimentation period to a range of 15° C. or more and 25° C. or less in order to shorten the sedimentation period, and after the sedimentation period has elapsed.
- the temperature of the solution 13 and the concentrated HA suspension 11 may be controlled within the range of 2° C. or higher and 20° C. or lower.
- the temperature control device 8 controls the temperature of the concentrated AgHA suspension 11 in the second container 12 in addition to controlling the temperature of the solution 13 in the first container 3. Good too.
- the control device 2 may use the temperature adjustment device 8 to control the temperature of the concentrated AgHA suspension 11 in the second container 12 to a range of 2° C. or more and 20° C. or less.
- the solution 13 is concentrated by natural sedimentation.
- the concentration may also be carried out by centrifugation using very low centrifugal force to maintain the dispersion of AgHA in solution 13.
- the AgHA suspension concentrated by natural sedimentation may be further concentrated using a centrifuge using a weak centrifugal force.
- the stirrer 37 may vigorously stir the AgHA suspension in order to disperse the AgHA within the concentrated AgHA suspension.
- the prepared solution 13 (AgHA suspension) is concentrated is explained as an example.
- the solution 13 may be stirred with the stirrer 37 and transferred to the second container 11.
- the solution 13 may be diluted with water, stirred with a stirrer 37, and transferred to the second container 11.
- the second injection device 5 In the first embodiment, the second injection device 5 generates a first mixed solution of phosphoric acid and silver nitrate, and drops the first mixed solution into the solution 13 in the container 3 .
- the first injection device 4 mixes the calcium hydroxide in the container 4a and the silver nitrate in the container 5b, and A liquid is generated, the second mixed liquid is injected into the container 3, and then the second injection device 5 drops the calcium hydroxide in the container 5a to the solution 13 in the container 3.
- an AgHA suspension 11 containing AgHA having a large antibacterial effect and deodorizing effect and a small particle size can be produced. I can do it.
- AgHA is applied to a nonwoven fabric by dipping the nonwoven fabric in an AgHA suspension.
- application is assumed to be a technique that utilizes wetting and solidification.
- FIG. 9 is a conceptual diagram showing an example of the configuration of a coating device 15 for coating a nonwoven fabric 14 with AgHA according to the third embodiment.
- FIG. 9 illustrates a side sectional view of the coating device 15.
- the coating device 15 mainly includes a delivery section 16 that sends out the nonwoven fabric 14 before treatment, a water tank section 17, a drying section 18, a winding section 19 that winds up the nonwoven fabric 14 after treatment, and a droplet receiving section 20.
- the continuous nonwoven fabric 14 is fed out from the feeding section 16, its position is adjusted by the rollers 211 to 214 of the water tank section 17 and the roller 22 of the drying section 18, and the continuous nonwoven fabric 14 is moved from left to right in FIG. 9 by winding up by the winding section 19. do.
- the direction from right to left in FIG. 9 is referred to as the nonwoven fabric feeding direction.
- the water tank section 17 includes rollers 211 to 214, a water tank 23, a first pump 24, a second pump 25, a shower head 26, an ultrasonic vibrator 27, and a cooler 28.
- the rollers 211 and 212 guide the nonwoven fabric 14 fed out from the feeding section 16 into the AgHA suspension 29 (corresponding to the AgHA suspension 11) stored in the water tank 23.
- the roller 212 further guides the nonwoven fabric 14 between the shower head 26 and the ultrasonic vibrator 27.
- the rollers 213 and 214 guide the nonwoven fabric 14 that has passed between the shower head 26 and the ultrasonic vibrator 27 out of the suspension 29 stored in the water tank 23.
- the roller 214 further returns the excess suspension contained in the nonwoven fabric 14 from the nonwoven fabric 14 to the water tank 23.
- the water tank 23 stores the suspension 29.
- the water tank 29 includes a first outlet 30 , an inlet 31 , and a second outlet 32 .
- the first outlet 30 is provided on the first side surface 23a of the water tank 23.
- the inlet 31 is provided on the second side surface 23b of the water tank 23.
- the second side surface 23b may be a surface facing the first side surface 23a.
- the first pump 24 discharges the suspension 29 from the first outlet 30 to the outside of the water tank 23 and causes the suspension 29 to flow into the water tank 23 from the inlet 31. As a result, the suspension 29 flows within the water tank 23.
- the second outlet 32 is provided on the bottom surface 23c of the water tank 23.
- the second pump 25 discharges the suspension 29 from the second outlet 32 to the outside of the water tank 23 and supplies the suspension 29 to the shower head 26.
- the suspension 29 that exists deep in the water tank 23 and has a high concentration of AgHA is supplied to the shower head 26 , the suspension 29 is spouted from the shower head 26 , and the suspension 29 is flowed in the water tank 23 . can be done.
- the shower head 26 spouts a suspension 29 from its liquid jetting surface.
- the liquid ejection surface of the shower head 26 faces the vibration surface of the ultrasonic vibrator 27 with a gap therebetween.
- the ultrasonic transducer 27 is an example of an ultrasonic generator.
- the ultrasonic vibrator 27 vibrates at a high frequency and emits ultrasonic waves from its ultrasonic emission surface.
- the ultrasonic transducer 27 may be, for example, a device that emits powerful ultrasonic waves for cell disruption.
- the ultrasonic wave emitting surface of the ultrasonic vibrator 27 faces the liquid discharge surface of the shower head 26 via a gap.
- the shower head 26 is placed at the top and the ultrasonic vibrator 27 is placed at the bottom.
- the gap between the liquid discharge surface of the shower head 26 and the vibration surface of the ultrasonic vibrator 27 is, for example, larger than the thickness of the nonwoven fabric 14 and 3 mm or less. From the results of the experiment, it was possible to infiltrate the suspension 29 into the nonwoven fabric 14 even when the gap was, for example, 6.5 mm or less. By making the ultrasonic waves more powerful and/or making the water flow of the suspension 29 more powerful, the gap can be applied in a range of 50 mm or less.
- the nonwoven fabric 14 that has passed through the gap between the shower head 26 and the ultrasonic vibrator 27 is soaked with the suspension 29.
- the cooler 28 suppresses the temperature rise of the suspension 29 in the water tank 23 due to ultrasonic waves. More specifically, the cooler 28 operates when the temperature of the suspension 29 in the water tank 23 exceeds a threshold value and lowers the temperature of the suspension 29.
- the droplet receiving section 20 is arranged between the water tank section 17 and the drying section 18.
- the droplet receiver 20 receives droplets of the suspension 29 dripping from the nonwoven fabric 14 .
- the drying section 18 includes a housing 33, a plurality of rollers 22, an air outlet 34, and a support stand 35.
- the surface of the casing 33 on the side into which the nonwoven fabric 14 is carried may be, for example, a transparent acrylic plate 33a.
- the acrylic plate 33a has an opening 33c for carrying the nonwoven fabric 14 into the housing 33 from the outside.
- the surface of the casing 33 on the side from which the nonwoven fabric 14 is carried out may be, for example, a flexible silicon plate 33b.
- a flexible silicon plate 33b For example, only the upper portion of the silicon plate 33b is connected to the upper surface of the casing 33, and the silicon plate 33b is arranged like a banner.
- the operator can set and change the inside of the housing 33 by raising this silicon plate 33b.
- the flexible silicon plate 33b in this manner, the operator can easily observe and change the internal state of the drying section 18.
- gas such as air inside the housing 33 can be flexibly discharged.
- the silicon plate 33b has an opening 33d for transporting the nonwoven fabric 14 from the inside of the casing 33 to the outside.
- the plurality of rollers 22 are arranged so that the nonwoven fabric 14 carried in through an opening 33c formed in an acrylic plate 33a of the drying section 18 is carried out through an opening 33d formed in a silicone plate 33b of the drying section 18. , move.
- the air outlet 34 discharges air (for example, warm air) for drying the nonwoven fabric 14.
- the air outlet 34 discharges air in a direction perpendicular to the plane of the nonwoven fabric 14 on the side of the acrylic plate 33a inside the housing 33. More specifically, the air outlet 34 is installed on the upper surface of the casing 33 on the import side of the nonwoven fabric 14 inside the casing 33, and discharges downward air to the nonwoven fabric 14.
- the shape of the air outlet 34 is preferably circular, for example, but other shapes such as an ellipse or a rectangle may also be used.
- a support stand 35 is installed on the roller 22 side of the nonwoven fabric 14 opposite to the blowout port 34 side to prevent the nonwoven fabric 14 exposed to the wind from getting caught up in the roller 22.
- the support stand 35 is inside the casing 33 and supports the nonwoven fabric 14 on the carry-in side where the nonwoven fabric 14 receives wind.
- the upper surface of the support stand 35 (the surface that supports the nonwoven fabric 14) is net-shaped.
- FIG. 10 is a side view showing an example of the shower head 26 and the ultrasonic vibrator 27.
- the shower head 26 and the ultrasonic vibrator 27 are provided to face each other with a gap 36, for example, larger than the thickness of the nonwoven fabric 14 and 3 mm or less.
- a shower head 26 is provided on the top and an ultrasonic transducer 27 is provided on the bottom.
- other arrangement relationships may be applied, such as the shower head 26 being at the bottom and the ultrasonic transducer 27 being at the top.
- a plurality of holes are formed in the lower surface of the shower head 26.
- Suspension liquid 29 is ejected from holes in the lower surface of shower head 26 toward nonwoven fabric 14 .
- the ultrasonic vibrator 27 vibrates the nonwoven fabric 14 and the suspension 29 present in the gap 36 with ultrasonic waves.
- the nonwoven fabric 14 immersed in the suspension 29 in the water tank 23 contains air bubbles.
- suspension 29 ejected from shower head 26 is pressed against nonwoven fabric 14 . Due to the synergistic effect of the ejection of the suspension 29 and the ultrasonic waves generated from the ultrasonic vibrator 27, air bubbles are discharged from the nonwoven fabric 14, and the hydrophobic nonwoven fabric 14 is wetted by the suspension 29.
- FIG. 11 is a perspective view showing an example of the upper surface of the support stand 35.
- the efficiency with which the air discharged from the blowing port 34 blows through the nonwoven fabric 14 can be improved, and furthermore, the nonwoven fabric 14 can be prevented from being caught up in the rotor 22 under the support stand 35. can be prevented.
- FIG. 12 is a flowchart illustrating an example of a method for applying AgHA to the nonwoven fabric 14, which is executed by the application device 15 according to the third embodiment.
- step S1201 the nonwoven fabric 14 is set in the coating device 15 in a state that it can be moved from the feeding section 16 to the winding section 19 via the water tank section 17 and the drying section 18 in the nonwoven fabric feeding direction.
- step S1202 the water tank 23 stores the suspension 29.
- step S1203a the first pump 24 circulates the suspension 29 in the water tank 23.
- step S1203b the second pump 25 supplies the suspension 29 in the water tank 23 to the shower head 26, and causes the shower head 26 to spout the suspension 29.
- step S1203c the ultrasonic vibrator 27 emits ultrasonic waves to the nonwoven fabric 14 by vibration operation to remove air bubbles from the nonwoven fabric 14.
- step S1204 the feeding unit 16, rollers 211 to 214, roller 22, and winding unit 19 move the nonwoven fabric 14 in the nonwoven fabric feeding direction.
- step S1205 the drying unit 18 dries the nonwoven fabric 14 soaked with the suspension 29 by using the air discharged from the air outlet 34.
- the particle size of AgHA in suspension 29 is smaller than that of dried AgHA.
- AgHA having a small particle size is applied to the nonwoven fabric 14 by impregnating the suspension 29 into the nonwoven fabric 14 to make it wet, and then drying it.
- the nonwoven fabric 14 is hydrophobic, simply soaking the nonwoven fabric 14 in the suspension 29 may not sufficiently soak the suspension 29 into the nonwoven fabric 14, and it may not be possible to sufficiently apply AgHA to the nonwoven fabric 14.
- the nonwoven fabric 14 is vibrated by the ultrasonic vibrator 27, and the suspension 29 is ejected from the shower head 26 toward the nonwoven fabric 14 to generate a water flow. Air bubbles are expelled to make the nonwoven fabric 14 wet with the suspension 29, and then the nonwoven fabric 14 is quickly dried.
- the shower head 26, in addition to ejecting the homogeneous suspension 29, is also responsible for expelling air bubbles.
- AgHA which is a suspended component and has a small particle size, can be attached to the nonwoven fabric 14.
- the concentration of the suspension 29 in the water tank 23 can be made uniform by vibrating the suspension 29 using the ultrasonic vibrator 27.
- the nonwoven fabric 14 passes through the gap 36 between the shower head 26 and the ultrasonic vibrator 27 that face each other.
- the width of this gap 36 larger than the thickness of the nonwoven fabric 14 and 3 mm or less, permeation of the suspension 29 into the nonwoven fabric 14 can be promoted.
- the concentration of the suspension 29 may be set to 0.005% or more and 5.0% or less.
- AgHA can be sufficiently adhered to the nonwoven fabric 14, and it is possible to prevent AgHA from excessively adhering to the fibers of the nonwoven fabric 14 and causing the AgHA powder to fall off.
- the appropriate concentration of suspension 29 will vary depending on particle size.
- the suspension 29 may be replenished into the water tank 23 so that the concentration of the suspension 29 in the water tank 23 is equal to or higher than a predetermined value.
- the inside of the nonwoven fabric 14 can be efficiently
- the nonwoven fabric 14 can be wetted by the suspension 29 by expelling the air bubbles.
- a shower head 26 and an ultrasonic vibrator 27 are used to wet the hydrophobic nonwoven fabric 14.
- the nonwoven fabric is hydrophilic, the nonwoven fabric can be wetted without using the ultrasonic transducer 27.
- the nonwoven fabric 14 passes between the shower head 26 and the ultrasonic vibrator 27 , the extra suspension attached to the nonwoven fabric 14 is squeezed out by the roller 214 , and the nonwoven fabric 14 returns to the water tank 23 .
- the moderately wet nonwoven fabric 14 is then conveyed into the drying section 18.
- the suspension 29 discharged from the first outlet 30 is caused to flow in from the inlet 31, thereby further reducing the suspension in the water tank 23.
- the liquid 29 can be stirred and circulated. Thereby, the concentration of the suspension 29 in the water tank 23 can be made uniform, and AgHA can be uniformly applied to the nonwoven fabric 14.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Environmental Sciences (AREA)
- Birds (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Wood Science & Technology (AREA)
- Dentistry (AREA)
- Plant Pathology (AREA)
- Pest Control & Pesticides (AREA)
- Zoology (AREA)
- Hematology (AREA)
- Materials Engineering (AREA)
- Heart & Thoracic Surgery (AREA)
- Surgery (AREA)
- Vascular Medicine (AREA)
- Agronomy & Crop Science (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Dans le présent mode de réalisation, l'appareil de production est pourvu d'un premier dispositif d'injection, d'un second dispositif d'injection, d'un dispositif de commande et d'un dispositif verseur. Le premier dispositif d'injection injecte de l'hydroxyde de calcium dans un récipient. Le second dispositif d'injection injecte une solution mélangée préparée par mélange de nitrate d'argent, de nitrate de cuivre ou de sulfate de cuivre avec de l'acide phosphorique dans la solution stockée dans le récipient. L'unité de commande commande la concentration et/ou la vitesse d'injection de la solution mélangée devant être injectée par le second dispositif d'injection de façon à ce que la valeur du pH mesurée par un pH-mètre se trouve dans une plage prédéfinie, produisant ainsi une suspension d'hydroxyapatite blanche ou gris clair contenant un métal antibactérien dans le récipient.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2022/018811 WO2023209788A1 (fr) | 2022-04-26 | 2022-04-26 | Appareil et procédé de production d'hydroxyapatite contenant un métal antibactérien |
| JP2022567621A JPWO2023209788A1 (fr) | 2022-04-26 | 2022-04-26 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2022/018811 WO2023209788A1 (fr) | 2022-04-26 | 2022-04-26 | Appareil et procédé de production d'hydroxyapatite contenant un métal antibactérien |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023209788A1 true WO2023209788A1 (fr) | 2023-11-02 |
Family
ID=88518150
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/018811 WO2023209788A1 (fr) | 2022-04-26 | 2022-04-26 | Appareil et procédé de production d'hydroxyapatite contenant un métal antibactérien |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JPWO2023209788A1 (fr) |
| WO (1) | WO2023209788A1 (fr) |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS62252307A (ja) * | 1986-04-25 | 1987-11-04 | Sekisui Plastics Co Ltd | 水酸アパタイトを湿式合成する方法 |
| JPH02180270A (ja) * | 1988-09-29 | 1990-07-13 | Sangi:Kk | 抗菌性ハイドロキシアパタイト組成物及びその製造法 |
| JPH06200238A (ja) * | 1992-12-28 | 1994-07-19 | Sumitomo Cement Co Ltd | 抗菌・防カビ性シーリング材 |
| JP2007075595A (ja) * | 2005-08-19 | 2007-03-29 | San Medical Gijutsu Kenkyusho:Kk | インプラント医療機器用シート状被覆部材 |
| JP2010273847A (ja) * | 2009-05-28 | 2010-12-09 | Tokyo Institute Of Technology | 高密度多孔質複合体 |
| JP2019155209A (ja) * | 2018-03-07 | 2019-09-19 | 住友金属鉱山株式会社 | ホウ素含有水の処理設備および処理方法 |
| JP2019528364A (ja) * | 2017-01-12 | 2019-10-10 | 株式会社テラハイムTerraheim Co., Ltd. | 銀ナノ粒子が含浸された銀ナノ有・無機複合樹脂の製造方法及びこれを利用して製造された抗生物性上水道管 |
-
2022
- 2022-04-26 WO PCT/JP2022/018811 patent/WO2023209788A1/fr active Application Filing
- 2022-04-26 JP JP2022567621A patent/JPWO2023209788A1/ja active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS62252307A (ja) * | 1986-04-25 | 1987-11-04 | Sekisui Plastics Co Ltd | 水酸アパタイトを湿式合成する方法 |
| JPH02180270A (ja) * | 1988-09-29 | 1990-07-13 | Sangi:Kk | 抗菌性ハイドロキシアパタイト組成物及びその製造法 |
| JPH06200238A (ja) * | 1992-12-28 | 1994-07-19 | Sumitomo Cement Co Ltd | 抗菌・防カビ性シーリング材 |
| JP2007075595A (ja) * | 2005-08-19 | 2007-03-29 | San Medical Gijutsu Kenkyusho:Kk | インプラント医療機器用シート状被覆部材 |
| JP2010273847A (ja) * | 2009-05-28 | 2010-12-09 | Tokyo Institute Of Technology | 高密度多孔質複合体 |
| JP2019528364A (ja) * | 2017-01-12 | 2019-10-10 | 株式会社テラハイムTerraheim Co., Ltd. | 銀ナノ粒子が含浸された銀ナノ有・無機複合樹脂の製造方法及びこれを利用して製造された抗生物性上水道管 |
| JP2019155209A (ja) * | 2018-03-07 | 2019-09-19 | 住友金属鉱山株式会社 | ホウ素含有水の処理設備および処理方法 |
Non-Patent Citations (3)
| Title |
|---|
| AL-ESSA KHANSAA, RADHA A V, NAVROTSKY ALEXANDRA: "Drop Solution Calorimetric Studies of Interface Enthalpy of Cubic Silver (I) Oxide (Ag<sub>2</sub>O) Nanocrystals", KEY ENGINEERING MATERIALS, vol. 878, pages 73 - 80, XP093104608, DOI: 10.4028/www.scientific.net/KEM.878.73 * |
| GOITSUKA, YUKARI: "Experiments Forum. Silver reactions. ", CHEMICAL EDUCATION, NIPPON KAGAKKAI, JP, vol. 66, no. 12, 1 December 2018 (2018-12-01), JP , pages 580 - 581, XP009550500, ISSN: 0386-2151 * |
| MOHD PU'AD N.A.S., ABDUL HAQ R.H., MOHD NOH H., ABDULLAH H.Z., IDRIS M.I., LEE T.C.: "Synthesis method of hydroxyapatite: A review", MATERIALS TODAY: PROCEEDINGS, ELSEVIER, NL, vol. 29, 1 January 2020 (2020-01-01), NL , pages 233 - 239, XP093104604, ISSN: 2214-7853, DOI: 10.1016/j.matpr.2020.05.536 * |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2023209788A1 (fr) | 2023-11-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN108472672B (zh) | 喷施器系统 | |
| US3425669A (en) | Dry chemical feeder method and apparatus | |
| WO2023209788A1 (fr) | Appareil et procédé de production d'hydroxyapatite contenant un métal antibactérien | |
| CN102666942A (zh) | 电镀装置 | |
| US3974307A (en) | Method for coating wood chips with resinous liquid | |
| WO2023195119A1 (fr) | Appareil et méthode de préparation d'une suspension d'hydroxyapatite | |
| CN110961165A (zh) | 催化剂浸渍干燥一体化装置和方法及其应用 | |
| JP2024012323A (ja) | 抗菌性金属含有ハイドロキシアパタイトの作製装置 | |
| US5328261A (en) | Method and apparatus for dissolving powder in a liquid | |
| US4300446A (en) | Apparatus for salting cheese | |
| CN110961164B (zh) | 催化剂连续浸渍设备和方法及其应用 | |
| CN112315798A (zh) | 一种智能制造的药片糖衣包裹装置 | |
| KR101813004B1 (ko) | 제올라이트 혼합액을 이용한 제올라이트 처리장치 | |
| KR102024691B1 (ko) | 원단 처리 장치 및 방법 | |
| USRE27681E (en) | Dry chemical feeder method and apparatus | |
| CN212492631U (zh) | 一种水溶性二氧化钛纳米颗粒制备用配比设备 | |
| US7005151B1 (en) | Method and apparatus for coating a product | |
| WO2023175846A1 (fr) | Dispositif et procédé pour l'application d'hydroxyapatite sur un tissu non-tissé | |
| CN206652401U (zh) | 混合搅拌装置 | |
| CN107097327B (zh) | 铁氧体成形体生产线以及铁氧体成形体生产方法 | |
| RU2186867C1 (ru) | Способ и устройство для получения литых композиционных материалов на основе алюминия | |
| CN218784938U (zh) | 一种纸盒生产用快速喷胶机构 | |
| CN210356773U (zh) | 一种制备脱氧剂的混合机用液体搅拌机 | |
| CN108588688A (zh) | 一种磁性磨料化学复合镀装置 | |
| CN210278887U (zh) | 一种高速混合机 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022567621 Country of ref document: JP |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22940068 Country of ref document: EP Kind code of ref document: A1 |