WO2022260145A1 - 防曇膜付きガラス物品及び防曇膜形成用塗布液 - Google Patents
防曇膜付きガラス物品及び防曇膜形成用塗布液 Download PDFInfo
- Publication number
- WO2022260145A1 WO2022260145A1 PCT/JP2022/023355 JP2022023355W WO2022260145A1 WO 2022260145 A1 WO2022260145 A1 WO 2022260145A1 JP 2022023355 W JP2022023355 W JP 2022023355W WO 2022260145 A1 WO2022260145 A1 WO 2022260145A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- film
- water
- glass
- fogging
- antifogging film
- Prior art date
Links
- 239000011521 glass Substances 0.000 title claims abstract description 102
- 239000007788 liquid Substances 0.000 title claims description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 69
- 239000000758 substrate Substances 0.000 claims abstract description 44
- 239000006087 Silane Coupling Agent Substances 0.000 claims abstract description 26
- 229920000642 polymer Polymers 0.000 claims abstract description 21
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 claims abstract description 19
- 150000002009 diols Chemical class 0.000 claims abstract description 18
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 16
- 238000000576 coating method Methods 0.000 claims description 34
- 239000011248 coating agent Substances 0.000 claims description 33
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 claims description 18
- 239000005329 float glass Substances 0.000 claims description 12
- 229920002554 vinyl polymer Polymers 0.000 claims description 8
- DHKHKXVYLBGOIT-UHFFFAOYSA-N 1,1-Diethoxyethane Chemical compound CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 claims description 7
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 6
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 claims description 6
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 claims description 5
- 239000011354 acetal resin Substances 0.000 claims description 4
- 230000015572 biosynthetic process Effects 0.000 claims description 4
- CDQSJQSWAWPGKG-UHFFFAOYSA-N butane-1,1-diol Chemical compound CCCC(O)O CDQSJQSWAWPGKG-UHFFFAOYSA-N 0.000 claims description 4
- 229920006324 polyoxymethylene Polymers 0.000 claims description 4
- ACCCMOQWYVYDOT-UHFFFAOYSA-N hexane-1,1-diol Chemical compound CCCCCC(O)O ACCCMOQWYVYDOT-UHFFFAOYSA-N 0.000 claims description 2
- UWJJYHHHVWZFEP-UHFFFAOYSA-N pentane-1,1-diol Chemical compound CCCCC(O)O UWJJYHHHVWZFEP-UHFFFAOYSA-N 0.000 claims description 2
- 239000004721 Polyphenylene oxide Substances 0.000 abstract description 4
- 229920000570 polyether Polymers 0.000 abstract description 4
- 239000002250 absorbent Substances 0.000 abstract 1
- 230000002745 absorbent Effects 0.000 abstract 1
- 238000011156 evaluation Methods 0.000 description 26
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 23
- -1 B2O3 Chemical compound 0.000 description 18
- 125000000524 functional group Chemical group 0.000 description 12
- 229910052710 silicon Inorganic materials 0.000 description 10
- 239000010703 silicon Substances 0.000 description 10
- 239000000377 silicon dioxide Substances 0.000 description 10
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 9
- 239000010419 fine particle Substances 0.000 description 8
- 238000000034 method Methods 0.000 description 8
- 229920001296 polysiloxane Polymers 0.000 description 8
- 238000006359 acetalization reaction Methods 0.000 description 7
- 125000003277 amino group Chemical group 0.000 description 7
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzenecarboxaldehyde Natural products O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 7
- 230000000052 comparative effect Effects 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 7
- WYTZZXDRDKSJID-UHFFFAOYSA-N (3-aminopropyl)triethoxysilane Chemical compound CCO[Si](OCC)(OCC)CCCN WYTZZXDRDKSJID-UHFFFAOYSA-N 0.000 description 6
- 239000004372 Polyvinyl alcohol Substances 0.000 description 6
- 150000001241 acetals Chemical class 0.000 description 6
- 150000002430 hydrocarbons Chemical group 0.000 description 6
- 229920002451 polyvinyl alcohol Polymers 0.000 description 6
- 238000005299 abrasion Methods 0.000 description 5
- 125000003545 alkoxy group Chemical group 0.000 description 5
- 239000002585 base Substances 0.000 description 5
- 125000003700 epoxy group Chemical group 0.000 description 5
- 239000003822 epoxy resin Substances 0.000 description 5
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 5
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 5
- 229920000647 polyepoxide Polymers 0.000 description 5
- 239000005341 toughened glass Substances 0.000 description 5
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 4
- 125000000217 alkyl group Chemical group 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 238000009833 condensation Methods 0.000 description 4
- 230000005494 condensation Effects 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 4
- 238000007654 immersion Methods 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 229920005862 polyol Polymers 0.000 description 4
- SJECZPVISLOESU-UHFFFAOYSA-N 3-trimethoxysilylpropan-1-amine Chemical compound CO[Si](OC)(OC)CCCN SJECZPVISLOESU-UHFFFAOYSA-N 0.000 description 3
- 238000010521 absorption reaction Methods 0.000 description 3
- 150000003934 aromatic aldehydes Chemical class 0.000 description 3
- 150000003935 benzaldehydes Chemical group 0.000 description 3
- 230000001588 bifunctional effect Effects 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 238000010828 elution Methods 0.000 description 3
- 239000005357 flat glass Substances 0.000 description 3
- 239000012528 membrane Substances 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- 238000006116 polymerization reaction Methods 0.000 description 3
- 150000003077 polyols Chemical class 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- FZHAPNGMFPVSLP-UHFFFAOYSA-N silanamine Chemical compound [SiH3]N FZHAPNGMFPVSLP-UHFFFAOYSA-N 0.000 description 3
- 125000001424 substituent group Chemical group 0.000 description 3
- 230000002087 whitening effect Effects 0.000 description 3
- 239000001431 2-methylbenzaldehyde Chemical group 0.000 description 2
- ZTQSAGDEMFDKMZ-UHFFFAOYSA-N Butyraldehyde Chemical compound CCCC=O ZTQSAGDEMFDKMZ-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 238000006124 Pilkington process Methods 0.000 description 2
- 239000006096 absorbing agent Substances 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 229910052681 coesite Inorganic materials 0.000 description 2
- 239000008119 colloidal silica Substances 0.000 description 2
- 229910052906 cristobalite Inorganic materials 0.000 description 2
- 239000011353 cycloaliphatic epoxy resin Substances 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical compound C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 2
- 239000004744 fabric Substances 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- FXLOVSHXALFLKQ-UHFFFAOYSA-N p-tolualdehyde Chemical group CC1=CC=C(C=O)C=C1 FXLOVSHXALFLKQ-UHFFFAOYSA-N 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 238000007127 saponification reaction Methods 0.000 description 2
- 125000000467 secondary amino group Chemical group [H]N([*:1])[*:2] 0.000 description 2
- 150000003377 silicon compounds Chemical class 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 238000004528 spin coating Methods 0.000 description 2
- 229910052682 stishovite Inorganic materials 0.000 description 2
- 239000002344 surface layer Substances 0.000 description 2
- 229920002803 thermoplastic polyurethane Polymers 0.000 description 2
- 229910052905 tridymite Inorganic materials 0.000 description 2
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 1
- FPYUJUBAXZAQNL-UHFFFAOYSA-N 2-chlorobenzaldehyde Chemical group ClC1=CC=CC=C1C=O FPYUJUBAXZAQNL-UHFFFAOYSA-N 0.000 description 1
- PRKPGWQEKNEVEU-UHFFFAOYSA-N 4-methyl-n-(3-triethoxysilylpropyl)pentan-2-imine Chemical compound CCO[Si](OCC)(OCC)CCCN=C(C)CC(C)C PRKPGWQEKNEVEU-UHFFFAOYSA-N 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- 229910011255 B2O3 Inorganic materials 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 239000004593 Epoxy Substances 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- FUJCRWPEOMXPAD-UHFFFAOYSA-N Li2O Inorganic materials [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 1
- KKCBUQHMOMHUOY-UHFFFAOYSA-N Na2O Inorganic materials [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- 239000011358 absorbing material Substances 0.000 description 1
- IKHGUXGNUITLKF-XPULMUKRSA-N acetaldehyde Chemical compound [14CH]([14CH3])=O IKHGUXGNUITLKF-XPULMUKRSA-N 0.000 description 1
- 125000003668 acetyloxy group Chemical group [H]C([H])([H])C(=O)O[*] 0.000 description 1
- 239000003377 acid catalyst Substances 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 125000003302 alkenyloxy group Chemical group 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- IYABWNGZIDDRAK-UHFFFAOYSA-N allene Chemical group C=C=C IYABWNGZIDDRAK-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 239000005354 aluminosilicate glass Substances 0.000 description 1
- XJDFBLQCLSBCGQ-UHFFFAOYSA-N anthracene-1-carbaldehyde Chemical compound C1=CC=C2C=C3C(C=O)=CC=CC3=CC2=C1 XJDFBLQCLSBCGQ-UHFFFAOYSA-N 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- BPBSLBDLLKIFRL-UHFFFAOYSA-N benzaldehyde;2-methylbenzaldehyde Chemical group O=CC1=CC=CC=C1.CC1=CC=CC=C1C=O BPBSLBDLLKIFRL-UHFFFAOYSA-N 0.000 description 1
- 239000005388 borosilicate glass Substances 0.000 description 1
- ODINCKMPIJJUCX-UHFFFAOYSA-N calcium oxide Inorganic materials [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 238000006482 condensation reaction Methods 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 229910052593 corundum Inorganic materials 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- OTARVPUIYXHRRB-UHFFFAOYSA-N diethoxy-methyl-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](C)(OCC)CCCOCC1CO1 OTARVPUIYXHRRB-UHFFFAOYSA-N 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- XUCJHNOBJLKZNU-UHFFFAOYSA-M dilithium;hydroxide Chemical compound [Li+].[Li+].[OH-] XUCJHNOBJLKZNU-UHFFFAOYSA-M 0.000 description 1
- WHGNXNCOTZPEEK-UHFFFAOYSA-N dimethoxy-methyl-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](C)(OC)CCCOCC1CO1 WHGNXNCOTZPEEK-UHFFFAOYSA-N 0.000 description 1
- 238000003618 dip coating Methods 0.000 description 1
- 238000011978 dissolution method Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 125000005678 ethenylene group Chemical group [H]C([*:1])=C([H])[*:2] 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229910052736 halogen Chemical group 0.000 description 1
- 150000002367 halogens Chemical group 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 1
- 239000004611 light stabiliser Substances 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- OVWYEQOVUDKZNU-UHFFFAOYSA-N m-tolualdehyde Chemical group CC1=CC=CC(C=O)=C1 OVWYEQOVUDKZNU-UHFFFAOYSA-N 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- WSFSSNUMVMOOMR-NJFSPNSNSA-N methanone Chemical compound O=[14CH2] WSFSSNUMVMOOMR-NJFSPNSNSA-N 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 239000012768 molten material Substances 0.000 description 1
- INJVFBCDVXYHGQ-UHFFFAOYSA-N n'-(3-triethoxysilylpropyl)ethane-1,2-diamine Chemical compound CCO[Si](OCC)(OCC)CCCNCCN INJVFBCDVXYHGQ-UHFFFAOYSA-N 0.000 description 1
- PHQOGHDTIVQXHL-UHFFFAOYSA-N n'-(3-trimethoxysilylpropyl)ethane-1,2-diamine Chemical compound CO[Si](OC)(OC)CCCNCCN PHQOGHDTIVQXHL-UHFFFAOYSA-N 0.000 description 1
- MQWFLKHKWJMCEN-UHFFFAOYSA-N n'-[3-[dimethoxy(methyl)silyl]propyl]ethane-1,2-diamine Chemical compound CO[Si](C)(OC)CCCNCCN MQWFLKHKWJMCEN-UHFFFAOYSA-N 0.000 description 1
- KBJFYLLAMSZSOG-UHFFFAOYSA-N n-(3-trimethoxysilylpropyl)aniline Chemical compound CO[Si](OC)(OC)CCCNC1=CC=CC=C1 KBJFYLLAMSZSOG-UHFFFAOYSA-N 0.000 description 1
- RMTGISUVUCWJIT-UHFFFAOYSA-N n-[3-[3-aminopropoxy(dimethoxy)silyl]propyl]-1-phenylprop-2-en-1-amine;hydrochloride Chemical compound Cl.NCCCO[Si](OC)(OC)CCCNC(C=C)C1=CC=CC=C1 RMTGISUVUCWJIT-UHFFFAOYSA-N 0.000 description 1
- 150000004002 naphthaldehydes Chemical class 0.000 description 1
- 238000007500 overflow downdraw method Methods 0.000 description 1
- AFEQENGXSMURHA-UHFFFAOYSA-N oxiran-2-ylmethanamine Chemical compound NCC1CO1 AFEQENGXSMURHA-UHFFFAOYSA-N 0.000 description 1
- 239000013500 performance material Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920002037 poly(vinyl butyral) polymer Polymers 0.000 description 1
- 229920001228 polyisocyanate Polymers 0.000 description 1
- 239000005056 polyisocyanate Substances 0.000 description 1
- 229920005749 polyurethane resin Polymers 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 239000011118 polyvinyl acetate Substances 0.000 description 1
- NOTVAPJNGZMVSD-UHFFFAOYSA-N potassium monoxide Inorganic materials [K]O[K] NOTVAPJNGZMVSD-UHFFFAOYSA-N 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 125000006410 propenylene group Chemical group 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 239000008213 purified water Substances 0.000 description 1
- 239000005871 repellent Substances 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 238000007650 screen-printing Methods 0.000 description 1
- 239000005361 soda-lime glass Substances 0.000 description 1
- RCIJACVHOIKRAP-UHFFFAOYSA-N sodium;1,4-dioctoxy-1,4-dioxobutane-2-sulfonic acid Chemical compound [Na+].CCCCCCCCOC(=O)CC(S(O)(=O)=O)C(=O)OCCCCCCCC RCIJACVHOIKRAP-UHFFFAOYSA-N 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 125000001302 tertiary amino group Chemical group 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- JXUKBNICSRJFAP-UHFFFAOYSA-N triethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](OCC)(OCC)CCCOCC1CO1 JXUKBNICSRJFAP-UHFFFAOYSA-N 0.000 description 1
- DQZNLOXENNXVAD-UHFFFAOYSA-N trimethoxy-[2-(7-oxabicyclo[4.1.0]heptan-4-yl)ethyl]silane Chemical compound C1C(CC[Si](OC)(OC)OC)CCC2OC21 DQZNLOXENNXVAD-UHFFFAOYSA-N 0.000 description 1
- HGBOYTHUEUWSSQ-UHFFFAOYSA-N valeric aldehyde Natural products CCCCC=O HGBOYTHUEUWSSQ-UHFFFAOYSA-N 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 229910001845 yogo sapphire Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D129/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal, or ketal radical; Coating compositions based on hydrolysed polymers of esters of unsaturated alcohols with saturated carboxylic acids; Coating compositions based on derivatives of such polymers
- C09D129/14—Homopolymers or copolymers of acetals or ketals obtained by polymerisation of unsaturated acetals or ketals or by after-treatment of polymers of unsaturated alcohols
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B60—VEHICLES IN GENERAL
- B60J—WINDOWS, WINDSCREENS, NON-FIXED ROOFS, DOORS, OR SIMILAR DEVICES FOR VEHICLES; REMOVABLE EXTERNAL PROTECTIVE COVERINGS SPECIALLY ADAPTED FOR VEHICLES
- B60J1/00—Windows; Windscreens; Accessories therefor
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C17/00—Surface treatment of glass, not in the form of fibres or filaments, by coating
- C03C17/28—Surface treatment of glass, not in the form of fibres or filaments, by coating with organic material
- C03C17/30—Surface treatment of glass, not in the form of fibres or filaments, by coating with organic material with silicon-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C17/00—Surface treatment of glass, not in the form of fibres or filaments, by coating
- C03C17/28—Surface treatment of glass, not in the form of fibres or filaments, by coating with organic material
- C03C17/32—Surface treatment of glass, not in the form of fibres or filaments, by coating with organic material with synthetic or natural resins
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C17/00—Surface treatment of glass, not in the form of fibres or filaments, by coating
- C03C17/34—Surface treatment of glass, not in the form of fibres or filaments, by coating with at least two coatings having different compositions
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C17/00—Surface treatment of glass, not in the form of fibres or filaments, by coating
- C03C17/34—Surface treatment of glass, not in the form of fibres or filaments, by coating with at least two coatings having different compositions
- C03C17/3405—Surface treatment of glass, not in the form of fibres or filaments, by coating with at least two coatings having different compositions with at least two coatings of organic materials
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C17/00—Surface treatment of glass, not in the form of fibres or filaments, by coating
- C03C17/34—Surface treatment of glass, not in the form of fibres or filaments, by coating with at least two coatings having different compositions
- C03C17/42—Surface treatment of glass, not in the form of fibres or filaments, by coating with at least two coatings having different compositions at least one coating of an organic material and at least one non-metal coating
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D183/00—Coating compositions based on macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon, with or without sulfur, nitrogen, oxygen, or carbon only; Coating compositions based on derivatives of such polymers
- C09D183/04—Polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/20—Diluents or solvents
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/05—Alcohols; Metal alcoholates
- C08K5/053—Polyhydroxylic alcohols
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/5403—Silicon-containing compounds containing no other elements than carbon or hydrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/544—Silicon-containing compounds containing nitrogen
Definitions
- the present invention relates to a glass article with an antifogging film and a coating liquid for forming an antifogging film.
- Glass substrates are required to have various properties. For example, anti-fogging properties are required for glass substrates. In order to obtain an anti-fogging glass substrate, techniques for forming an anti-fogging film on the surface of the glass substrate have been investigated.
- Patent Document 1 describes a windshield in which a water-absorbing material suitable for preventing condensation of water vapor in the optical path space is arranged.
- this windshield for example, it is described that the water-absorbing film disposed on the glass has excellent water-absorbing properties in a high-humidity region.
- an object of the present invention is to provide a novel glass article with an anti-fogging film.
- the present invention comprising a glass substrate and an anti-fog coating on the surface of the glass substrate; It was immersed in water at 25°C for 24 hours and then removed from the water, and the antifogging film was exposed for 30 seconds to water vapor generated from water at 90°C to 100°C which was placed vertically downward from the antifogging film at a distance of 60 mm.
- Can read information of QR code having a size of 40 mm square
- a glass article with an anti-fog coating Provided is a glass article with an anti-fog coating.
- the QR code encodes the character string "Rank: B” as the information according to the Japanese Industrial Standard (JIS) X 0510: 2018, with a symbol size of 21 ⁇ 21 modules and a level H error correction specification. It is a two-dimensional code.
- the present invention provides comprising a glass substrate and an anti-fog coating on the surface of the glass substrate;
- the anti-fogging film contains a water-absorbing polymer, It is immersed in water at 25° C. for 24 hours and then removed from the water, and the anti-fogging film is exposed for 30 seconds to water vapor generated from water at 90° C. to 100° C. placed at a distance of 60 mm vertically downward from the anti-fogging film.
- a transparent continuous film is formed on the surface of the antifogging film exposed to the water vapor when the test is performed.
- a glass article with an anti-fog coating is provided.
- the present invention provides comprising a glass substrate and an anti-fog coating on the surface of the glass substrate;
- the anti-fogging film comprises at least one selected from the group consisting of a silane coupling agent and a crosslinked structure derived from the silane coupling agent, a water-absorbing polymer, a polyether-modified siloxane, and a diol having 2 to 8 carbon atoms.
- a glass article with an anti-fog coating is provided.
- the present invention provides A silane coupling agent, a water-absorbing polymer, a polyether-modified siloxane, and a diol having 2 to 8 carbon atoms, A coating liquid for forming an antifogging film is provided.
- a novel glass article with an anti-fogging film is provided.
- FIG. 3 is a cross-sectional view showing another example of the glass article with an antifogging film according to the present embodiment.
- It is a schematic diagram for demonstrating the outline
- It is another schematic diagram for demonstrating the outline
- It is an example of a QR code (size 10 mm ⁇ 10 mm, recorded information “Rank: SSS”) used for high-temperature steam evaluation.
- a QR code size 15 mm ⁇ 15 mm, recorded information “Rank: SS” used for high-temperature steam evaluation.
- QR code size 20 mm ⁇ 20 mm, recorded information “Rank: S” used for high-temperature steam evaluation. It is an example of a QR code (size 30 mm ⁇ 30 mm, recorded information “Rank: A”) used for high-temperature steam evaluation. It is an example of a QR code (size 40 mm ⁇ 40 mm, recorded information “Rank: B”) used for high-temperature steam evaluation.
- the term "main component” means the component with the highest content.
- the “principal surface” of a plate-like article means two surfaces facing opposite sides separated by a predetermined distance called thickness.
- anti-fogging films are often blended with water-absorbing polymers. As the content of the water-absorbing polymer increases, the antifogging property of the film can be expected to improve. On the other hand, as the water-absorbing polymer content increases, the abrasion resistance of the membrane usually decreases. For this reason, as disclosed in Patent Document 1, an inorganic component, typically a silica component such as colloidal silica, is often added to the antifogging film to compensate for the decrease in abrasion resistance. In contrast, anti-fogging films are not required to have a high level of abrasion resistance, but may be required to have other properties.
- an inorganic component typically a silica component such as colloidal silica
- the anti-fogging film even when the anti-fogging film is exposed to harsh environments for a long period of time, it is sometimes required that the anti-fogging property can be maintained.
- the glass article with an anti-fogging film according to the present embodiment was obtained by further studies from this point of view.
- the anti-fogging film does not scatter light at a high level even when exposed to harsh environments It can exhibit the function of allowing it to pass through.
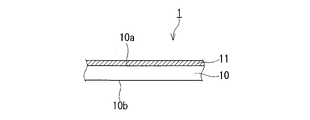
- FIG. 1 is a cross-sectional view showing a glass article with an anti-fogging film according to this embodiment.
- a glass article 1 with an antifogging film includes a plate-like glass substrate 10 , that is, a glass substrate, and an antifogging film 11 formed on the surface of the glass substrate 10 .
- the anti-fogging film 11 is formed on at least part of the surface of the glass substrate 10 , for example, the main surface of the glass substrate 10 .
- the antifogging film 11 may be formed on both main surfaces 10a and 10b of the plate-like glass substrate 10, but as shown in FIG. 1, it is formed only on one main surface 10a. may
- the shape and material of the glass substrate 10 are not particularly limited.
- the glass substrate 10 is, for example, a glass plate.
- the glass composition constituting the glass plate is not particularly limited, and may be soda-lime glass, aluminosilicate glass, borosilicate glass, alkali-free glass, or multicomponent glass called C glass, E glass, or the like.
- the multicomponent glass contains SiO2 as a main component, and a group consisting of components other than SiO2 , such as B2O3 , Al2O3 , MgO, CaO, Li2O, Na2O , and K2O . It further contains at least one oxide selected from the above.
- the glass plate may be made of silica glass.
- the glass plate may be float glass.
- Float glass is formed by a so-called float method. Since float glass is formed in a float bath with one main surface in contact with molten tin, tin diffuses to the main surface. Thus, float glass has a tin-diffused surface layer on one major surface, called the bottom surface, and this surface layer is absent on the other major surface, called the top surface. Stated from another point of view, in float glass, the concentration of tin on one major surface is higher than the concentration of tin on the other major surface.
- the glass plate may be formed by a manufacturing method other than the float method, such as an overflow down-draw method.
- the major surface 10a may be the top surface, but is preferably the bottom surface. Since the bottom surface has more hydroxyl groups than the top surface, it is suitable as a surface for forming an antifogging film with excellent water resistance.
- the thickness of the glass plate is, for example, 0.5 to 7.0 mm, and may be 0.5 to 5.0 mm.
- the thickness of the glass plate which is non-tempered glass, is preferably 3.5 mm or more. However, in the case of tempered glass, if the thickness is 1.8 mm or more, the glass plate can have sufficient impact resistance.
- the tempered glass may be air-cooled tempered glass or chemically tempered glass.
- the glass substrate 10 may be in the shape of a flat plate whose main surface is flat.
- the main surface of the glass substrate may be curved.
- the glass substrate may be obtained by bending a flat glass substrate.
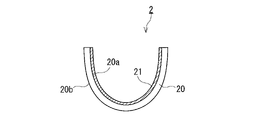
- the glass substrate may be a molded body having a curved surface that is directly molded from a molten material without passing through a flat glass substrate. An example of such a compact is shown in FIG.
- the main surfaces of the glass substrate 20 shown in FIG. 2 are both curved surfaces, one main surface 20a being concave and the other main surface 20b being convex.
- An anti-fogging film 21 is formed on the main surface 20a, which is a concave surface.
- An anti-fogging film may be formed on the main surface 20b, which is a convex surface.
- a base film may be formed on the main surface of the glass substrate.
- the base film is interposed between the surface of the glass base material and the anti-fogging film.
- the base film is not particularly limited, but may be, for example, a barrier film that prevents elution of alkali metals from glass.
- the barrier film is composed of, for example, a silica film.
- a film other than the anti-fogging film may be formed on the other main surface.
- films include antireflection films, water-repellent films, hydrophilic films, colored films, and the like.
- the film thickness of the antifogging films 11 and 21 is not limited to a specific value, and is 0.1 to 10 ⁇ m, preferably 0.5 to 5.0 ⁇ m, particularly preferably 0.8 to 2.0 ⁇ m.
- the antifogging films 11 and 21 contain, for example, a silane coupling agent and/or a crosslinked structure derived from the silane coupling agent, a water-absorbing polymer, a polyether-modified siloxane, and a diol having 2 to 8 carbon atoms. .
- a silane coupling agent and/or a crosslinked structure derived from the silane coupling agent a water-absorbing polymer, a polyether-modified siloxane, and a diol having 2 to 8 carbon atoms. .
- At least one selected from the group consisting of urethane resins, epoxy resins, acrylic resins, polyvinyl acetal resins, and polyvinyl alcohol resins can be exemplified as the water absorbing polymer.
- Urethane resins include polyurethane resins composed of polyisocyanate and polyol.
- Polyols include acrylic polyols and polyoxyalkylene-based polyols.
- epoxy-based resins include glycidyl ether-based epoxy resins, glycidyl ester-based epoxy resins, glycidylamine-based epoxy resins, and cycloaliphatic epoxy resins.
- Preferred epoxy resins are cycloaliphatic epoxy resins.
- a polyvinyl acetal resin hereinafter simply referred to as "polyvinyl acetal", which is a preferred water-absorbing polymer, will be described below.
- Polyvinyl acetal can be obtained by condensation reaction of polyvinyl alcohol with aldehyde to acetalize it.
- Acetalization of polyvinyl alcohol may be carried out using a known method such as a precipitation method using an aqueous medium in the presence of an acid catalyst, or a dissolution method using a solvent such as alcohol.
- Acetalization can also be carried out in parallel with the saponification of polyvinyl acetate.
- the degree of acetalization is preferably 2 to 40 mol %, more preferably 3 to 30 mol %, especially 5 to 20 mol %, and in some cases 5 to 15 mol %.
- the degree of acetalization can be measured, for example, by 13 C nuclear magnetic resonance spectroscopy.
- a polyvinyl acetal having a degree of acetalization within the above range is suitable for forming an anti-fogging film having good water absorption and water resistance.
- the average degree of polymerization of polyvinyl alcohol is preferably 200-4500, more preferably 500-4500.
- a high average degree of polymerization is advantageous for forming an anti-fogging film with good water absorption and water resistance, but if the average degree of polymerization is too high, the viscosity of the solution becomes too high, which may interfere with the formation of the film.
- the degree of saponification of polyvinyl alcohol is preferably 75 to 99.8 mol %.
- Aldehydes that are condensed with polyvinyl alcohol include aliphatic aldehydes such as formaldehyde, acetaldehyde, butyraldehyde, hexylcarbaldehyde, octylcarbaldehyde, and decylcarbaldehyde.
- benzaldehyde 2-methylbenzaldehyde, 3-methylbenzaldehyde, 4-methylbenzaldehyde, other alkyl group-substituted benzaldehyde; chlorobenzaldehyde, other halogen atom-substituted benzaldehyde; hydroxyl group, alkoxy group, amino group, alkyl such as cyano group
- Aromatic aldehydes such as substituted benzaldehyde in which a hydrogen atom is substituted by a functional group other than the group; condensed aromatic ring aldehydes such as naphthaldehyde and anthraldehyde.
- Aromatic aldehydes with strong hydrophobicity are advantageous in forming a water-absorbing film with a low degree of acetalization and excellent water resistance.
- the use of an aromatic aldehyde is also advantageous in forming a highly water-absorbing film while leaving many hydroxyl groups.
- the polyvinyl acetal preferably contains an acetal structure derived from aromatic aldehydes, especially benzaldehyde.
- the content of the water-absorbing polymer in the antifogging film is, for example, 45-95% by mass, preferably 55-85% by mass, more preferably 65-80% by mass.
- the water-absorbing polymer may be the main component of the anti-fogging film.
- the polyether-modified siloxane is a compound having at least one polyether chain selected from molecular chains bonded to the ends of the main chain of siloxane and molecular chains bonded as side chains of the main chain of siloxane.
- Siloxane is a compound having a siloxane bond (Si--O--Si) as a skeleton.
- Constituent units of the polyether chain are not particularly limited, but are, for example, ethylene oxide and propylene oxide.
- strand may contain only 1 type as a structural unit, and may contain 2 or more types.
- the polyether-modified siloxane may be polyether-modified silicone.
- Silicone is a polymer having a siloxane bond (Si--O--Si) as a skeleton.
- Examples of polyether-modified silicones are BYK-345, BYK-347, BYK-349 from BYK-Chemie, and TSF-4440 from Momentive.
- the content of polyether-modified siloxane in the antifogging film is, for example, 2 to 30% by mass, preferably 5 to 25% by mass, more preferably 8 to 20% by mass.
- Diols having 2 to 8 carbon atoms are for example selected from the group consisting of ethylene glycol, diethylene glycol, propylene glycol, dipropylene glycol, butanediol, pentanediol and hexanediol. may include at least one
- the diol may be a diol having 2 to 6 carbon atoms.
- Preferred diols include at least one selected from the group consisting of butanediol, propylene glycol, and dipropylene glycol.
- the antifogging film may contain propylene glycol and/or dipropylene glycol as particularly preferred diols.
- the content of the diol having 2 to 8 carbon atoms in the antifogging film is, for example, 0.01 to 30% by mass, preferably 0.05 to 20% by mass, more preferably 0.1 to 10% by mass.
- a silane coupling agent can be exemplified by a silicon compound having a hydrolyzable group represented by the following formula (I).
- the silicon compound having a hydrolyzable group represented by formula (I) may be used alone or in combination of two or more.
- n represents an integer of 1-3.
- X is a hydrolyzable group or a halogen atom.
- hydrolyzable groups include at least one selected from alkoxyl groups, acetoxy groups, alkenyloxy groups and amino groups.
- Preferred alkoxyl groups include alkoxyl groups having 1 to 4 carbon atoms (methoxy, ethoxy, propoxy, butoxy). Chlorine can be exemplified as a preferable halogen atom.
- Y is an alkyl group having 1 to 3 carbon atoms.
- Preferred alkyl groups are methyl and ethyl groups.
- L is a hydrocarbon group.
- a hydrocarbon group is preferably an alkylene group.
- the number of carbon atoms in the hydrocarbon group is, for example, 1-10, preferably 1-6, more preferably 1-3.
- Preferred examples of hydrocarbon groups are methylene, ethylene, n-propylene, isopropylene, vinylene and propenylene groups.
- R is a substituent or a hydrocarbon group which may have a substituent.
- the number of carbon atoms in this hydrocarbon group is not particularly limited, and is, for example, 4 to 12, but may be 3 or less.
- a substituent may be a reactive functional group.
- the reactive functional group is, for example, at least one selected from the group consisting of an epoxy group, an amino group, a mercapto group, an isocyanate group, an acrylic group, and a methacrylic group, preferably selected from the group consisting of an epoxy group and an amino group.
- Epoxy groups may be included as part of the glycidyl ether group.
- a silane coupling agent having an epoxy group may be described as "epoxysilane".
- the amino group may be a primary amino group, a secondary amino group or a tertiary amino group, preferably a primary amino group and a secondary amino group.
- a silane coupling agent having an amino group is sometimes referred to as "aminosilane
- silicon alkoxides Compounds in which X in formula (I) is an alkoxyl group are called silicon alkoxides.
- n is 2 or 3. That is, the silane coupling agent is preferably a bifunctional silicon alkoxide having a reactive functional group represented by RL-SiY 1 X 2 in formula (I) or RL- It is a trifunctional silicon alkoxide having a reactive functional group represented by SiX3 .
- bifunctional silicon alkoxides having reactive functional groups are glycidoxyalkylalkyldialkoxysilanes and aminoalkylalkyldialkoxysilanes.
- glycidoxyalkylalkyldialkoxysilanes are 3-glycidoxypropylmethyldimethoxysilane and 3-glycidoxypropylmethyldiethoxysilane.
- aminoalkylalkyldialkoxysilanes are N-2-(aminoethyl)-3-aminopropylmethyldimethoxysilane and N-2-(aminoethyl)-3-aminopropylethyldiethoxysilane.
- trifunctional silicon alkoxides having reactive functional groups are glycidoxyalkyltrialkoxysilane and aminoalkyltrialkoxysilane.
- glycidoxyalkyltrialkoxysilanes are 2-(3,4-epoxycyclohexyl)ethyltrimethoxysilane, 3-glycidoxypropyltrimethoxysilane (GPTMS), and 3-glycidoxypropyltriethoxysilane. be.
- aminoalkyltrialkoxysilanes are 3-aminopropyltrimethoxysilane (APTMS), 3-aminopropyltriethoxysilane (APTES), 3-triethoxysilyl-N-(1,3-dimethyl-butylidene)propylamine , 3-(N-phenyl)aminopropyltrimethoxysilane, N-(vinylbenzyl)-2-aminoethyl-3-aminopropyltrimethoxysilane hydrochloride, N-2-(aminoethyl)-3-aminopropyl trimethoxysilane, and N-2-(aminoethyl)-3-aminopropyltriethoxysilane.
- APIMS 3-aminopropyltrimethoxysilane
- APTES 3-aminopropyltriethoxysilane
- APTES 3-triethoxysilyl-N-(1,
- silane coupling agent only at least one selected from the group consisting of a bifunctional silicon alkoxide having a reactive functional group and a trifunctional silicon alkoxide having a reactive functional group may be used, Multiple types may be used.
- a trifunctional silicon alkoxide having a reactive functional group can preferably be used as the silane coupling agent.
- the trifunctional silicon alkoxide having a reactive functional group at least one selected from the group consisting of glycidoxyalkyltrialkoxysilane and aminoalkyltrialkoxysilane may be used alone, or a plurality of types may be used. good.
- trifunctional silicon alkoxides having reactive functional groups 3-glycidoxypropyltrimethoxysilane (GPTMS) and 3-aminopropyltriethoxysilane (APTES) can preferably be used.
- At least part of the silane coupling agent reacts with other components in the antifogging film to form a crosslinked structure.
- Other components include organic components such as water-absorbing polymers, and hydroxyl groups on the substrate surface.
- a particularly preferable combination of silane coupling agents is the combined use of an epoxy group-containing silane coupling agent (epoxysilane) and an amino group-containing silane coupling agent (aminosilane).
- Epoxysilane and aminosilane may be added in a weight ratio of, for example, 1:3 to 3:1, particularly 1:2 to 2:1.
- the content of the silane coupling agent in the antifogging film is, for example, 2 to 35% by mass, preferably 5 to 30% by mass, more preferably 8 to 25% by mass.
- the anti-fogging film may appropriately contain ultraviolet absorbers, infrared absorbers, leveling agents (surface conditioners), light stabilizers, etc., in addition to the components described above. However, these components are desirably added in an amount of 20% by mass or less, more preferably 10% by mass or less, and particularly 5% by mass or less of the antifogging film. Colloidal silica and other silica fine particles may or may not be contained in the antifogging film.
- the content of silica fine particles in the antifogging film may be, for example, 10 to 60% by mass, but may be limited to less than 5% by mass, further less than 3% by mass, and particularly less than 1% by mass.
- the content of oxide fine particles including silica fine particles may also be at the same level as described above for silica fine particles.
- the antifogging film may or may not contain oxide fine particles typified by silica fine particles. However, even if it does not contain silica fine particles, the anti-fogging film may contain siloxane components contained in polyether-modified siloxane, silane coupling agents, and the like.
- the antifogging film according to the present embodiment exerts a function of transmitting transmitted light at a high level without scattering even after being placed in a harsh environment for the antifogging film, for example, in contact with water for a long period of time. sell.
- the anti-fogging film desirably has transparency, water absorbency, water resistance, and hydrophilicity.
- An anti-fogging film with insufficient water resistance may elute its components when in contact with water for a long period of time.
- the anti-fogging film is hydrophilic enough to retain surplus water as a continuous film on its surface when the water absorption by itself reaches a saturated state, transmitted light will not be excessively scattered. permeable.
- the glass article with an anti-fogging film according to this embodiment can transmit transmitted light without excessive scattering.
- the anti-fogging coated glass article may have a haze ratio of, for example, 5% or less, even 3% or less, particularly 1% or less, and in some cases 0.4% or less.
- the haze ratio is specified in JIS K 7136:2018.
- both water resistance and hydrophilicity can be achieved. These properties can be evaluated by a method called hot steam evaluation, which is described in detail in the Examples section.
- hot steam evaluation which is described in detail in the Examples section.
- high-temperature and excessive water vapor is supplied to the anti-fogging film with the anti-fogging film facing vertically downward.
- a transparent continuous film of water is formed on the surface of the anti-fogging film, which is hydrophilic and excellent in water resistance, in the portion directly exposed to water vapor.
- Whether or not it is a "transparent continuous film” can be determined by visually confirming that continuity as a film is ensured and that the film is not clouded. Cloudiness of the film can be caused by whitening of the film due to insufficient water resistance or condensation on the film surface due to insufficient anti-fogging properties.
- On the surface of the film lacking hydrophilicity water is not retained as a continuous film, but is dispersed and deposited as water droplets. A film with insufficient water resistance is observed to be whitened by contact with high-temperature steam, and elution of film components and film defects may occur. Hydrophilicity of a surface is generally evaluated by the contact angle of water.
- the transparent continuous film may cover 80% or more, or even 90% or more of the surface of the antifogging film exposed to water vapor.
- the glass article with an antifogging film according to the present embodiment preferably has a QR code "A” having a size of 30 mm square, more preferably a QR code "A” having a size of 20 mm square, even after being immersed in water at room temperature for 24 hours.
- the coating liquid for forming an antifogging film contains a silane coupling agent, a water-absorbing polymer, a polyether-modified siloxane, and a diol having 2 to 8 carbon atoms.
- a silane coupling agent for example, the compounds described above can be used.
- the content of each component in the coating solution for forming an antifogging film may be appropriately adjusted so that the content of each component in the antifogging film falls within the above-described range.
- the coating liquid for forming an antifogging film may contain other components. Examples of other ingredients are water and alcohols.
- the anti-fogging film is formed by applying a coating liquid for forming the anti-fogging film (coating liquid for forming an anti-fogging film) to the surface of the glass substrate, and heating the glass substrate on which the coating film is formed with the coating liquid. , can be deposited.
- a coating liquid for forming the anti-fogging film coating liquid for forming an anti-fogging film
- Conventionally known materials and methods may be used for the solvent used for preparing the coating liquid and the coating method for the coating liquid. Examples of application methods are spin coating, roll coating, spray coating, dip coating, flow coating, screen printing and brush coating.
- the coating may optionally be dried prior to heating.
- the heating temperature of the glass substrate on which the coating film is formed is not particularly limited, but is, for example, 100 to 180° C., and the heating time is, for example, 5.0 minutes to 1.0 hour.
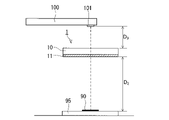
- FIG. 3A a glass article with an anti-fogging film is placed above a stainless steel heat-retaining cup 80 holding boiled water 70 so that the surface on which the anti-fogging film 11 is formed faces the heat-retaining cup 80 side. 1 was held horizontally. The temperature of water 70 was maintained at 90-100° C. while supplying steam. A distance D 1 between the antifogging film 11 and the water surface was set to 60 mm. The internal space of the heat-retaining cup 80 is cylindrical with an opening having a diameter of 64 mm, and the volume of the water 70 is about 130 cc.
- the antifogging film-attached glass article 1 was held on the heat-retaining cup 80 for 30 seconds, and high-temperature steam was supplied to the antifogging film 11 .
- the heat retaining cup 80 was removed and replaced with a mount 95 on which a predetermined QR code 90 was printed.
- the distance D 2 between the antifogging film 11 and the mount 95 was set to 110 mm.
- the QR code 90 was photographed with the camera 100 from above through the glass article 1 with the antifogging film, and it was confirmed whether the information possessed by the QR code 90 could be read.
- the distance D3 between the glass substrate 10 and the lens 101 of the camera 100 was set to 80 mm. It took less than 30 seconds from the removal of the heat retaining cup 80, that is, the stop of the supply of the high-temperature steam to the photographing of the QR code.
- QR code SSS 10 mm x 10 mm
- information "Rank: SSS” Fig. 4A
- QR code "SS” 15 mm x 15 mm
- information "Rank: SS” Fig. 4B
- QR code "S” 20 mm x 20 mm
- information "Rank: S” Fig. 4C
- information "Rank: A” Fig. 4D
- the QR code "SSS” described in FIG. 4A is based on JIS X 0510:2018, and the above character string is encoded as information according to the symbol size of 25 ⁇ 25 modules and the specification of error correction of level H.
- the QR code "SS” to QR code “B” described in FIGS. 4B to 4E conform to JIS X 0510:2018, and according to the specification of the symbol size of the 21 ⁇ 21 module and the error correction of level H, each of the above characters as information Encoded columns.
- Each character string is composed of half-width characters (1-byte code) instead of full-width characters.
- the camera used was Sony's smartphone "Xperia XZ2" (model name: SO-03K, OS: Android (registered trademark) (ver.10)).
- the QR code reading function of the LINE (registered trademark) application (ver.11.7.2) was used to read the QR code.
- Example 1 (Preparation of coating liquid) Polyvinyl acetal resin-containing solution (“S-Lec KX-5” manufactured by Sekisui Chemical Co., Ltd., solid content 8% by mass, degree of acetalization 9 mol%, including acetal structure derived from benzaldehyde) 48.2% by mass, polyether-modified siloxane (“BYK-345” manufactured by BYK Chemie) 0.5% by mass, 3-aminopropyltriethoxysilane (APTES, “KBE-903” manufactured by Shin-Etsu Silicone Co., Ltd.) 1.0% by mass, 3-glycidoxypropyltrimethoxy Silane (GPTMS, "KBM-403” manufactured by Shin-Etsu Silicone Co., Ltd.) 0.7% by mass, propylene glycol 20.0% by mass, purified water 18.5% by mass, alcohol solvent ("Neoethanol P-7” manufactured by Daishin Chemical Co., Ltd.
- a coating liquid was applied by spin coating (1500 rpm, 10 seconds) to the bottom surface of a float glass (size: 50 mm ⁇ 50 mm, thickness: 1.1 mm) preliminarily washed with alkali to form a coating film.
- the float glass with the coating film formed thereon was heated at 100° C. for 30 minutes to obtain a glass article with an anti-fogging film.
- Example 2 A glass article with an anti-fogging film was obtained in the same manner as in Example 1, except that the raw materials and amounts shown in Table 1 were used in the preparation of the coating liquid.
- the amount of the alcohol solvent was increased by the decrease in the amount of the epoxysilane.
- the amount of alcohol solvent was reduced by the amount of polyether-modified siloxane increased.
- BYK-347, 349, and 3455 are polyether-modified silicones manufactured by BYK-Chemie
- TSF-4440 is a polyether-modified silicone manufactured by Momentive Performance Materials.
- Comparative Examples 1 to 10 A glass article with an anti-fogging film was obtained in the same manner as in Example 1, except that the raw materials and amounts shown in Table 2 were used in the preparation of the coating liquid.
- "A-80" (Rapisol A-80) used in Comparative Example 10 is a product name manufactured by NOF Corporation.
- the amount of the alcohol solvent was increased by the amount of the component that was not added.
- the glass articles with an anti-fog film according to each example had a high-temperature steam evaluation of "SSS" to "S” after the water immersion test.
- the antifogging films of the glass articles according to the examples when the supply of high-temperature water vapor was stopped, a continuous film of water having a uniform thickness was formed on the surface, and the film itself was whitened and fogged due to dew condensation. None of the above was observed, and the transparency of the glass article with the antifogging film was ensured.
- the transparent continuous film evaluated as "SSS" to "S” is 90% or more of the surface of the antifogging film exposed to water vapor, more specifically, substantially all of the above surface. , was covered.
- Comparative Examples 7 to 9 the antifogging film was eluted after the water immersion test.
- Comparative Examples 1 to 6 and 10 when the supply of high-temperature steam was stopped, fogging due to dew condensation was observed, or large water droplets were dispersed and adhered to the surface of the antifogging film. Therefore, even the largest QR code could not be read in these comparative examples.
- Glass articles with anti-fogging films according to the present invention include window glass for vehicles and buildings; glass for facades of buildings; mirrors for houses, housing equipment, vehicles and mobile phones; optical members such as lenses and optical filters; headlights of vehicles; liquid crystal displays with touch panels such as electronic blackboards; head-mounted displays used in VR (virtual reality) goggles and the like.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Geochemistry & Mineralogy (AREA)
- General Chemical & Material Sciences (AREA)
- Wood Science & Technology (AREA)
- Mechanical Engineering (AREA)
- Surface Treatment Of Glass (AREA)
- Surface Treatment Of Optical Elements (AREA)
- Laminated Bodies (AREA)
Abstract
Description
ガラス基材と、前記ガラス基材の表面上の防曇膜と、を備え、
25℃の水に24時間浸漬させて前記水から取り出し、前記防曇膜から鉛直方向下方に60mm離間させて配置した90℃~100℃の水から発生する水蒸気に前記防曇膜を30秒間曝し、前記防曇膜から前記方向下方に110mm離れて配置したQRコードの情報を前記防曇膜が形成された側とは反対側からカメラを使用して読み取ることができるかを判定する試験において、40mm四方のサイズを有するQRコードの情報を読み取ることができる、
防曇膜付きガラス物品、を提供する。
ここで、前記QRコードは、日本産業規格(JIS)X 0510:2018に従って、21×21モジュールのシンボルサイズ及びレベルHの誤り訂正の仕様により、前記情報として文字列「Rank:B」を符号化した二次元コードである。
ガラス基材と、前記ガラス基材の表面上の防曇膜と、を備え、
前記防曇膜は、吸水性ポリマーを含み、
25℃の水に24時間浸漬させて前記水から取り出し、前記防曇膜から鉛直方向下方に60mm離間させて配置した90℃~100℃の水から発生する水蒸気に前記防曇膜を30秒間曝す試験を実施したときに、前記水蒸気に曝された前記防曇膜の前記表面に透明な連続膜が形成される、
防曇膜付きガラス物品、を提供する。
ガラス基材と、前記ガラス基材の表面上の防曇膜と、を備え、
前記防曇膜は、シランカップリング剤及びシランカップリング剤に由来する架橋構造からなる群より選択される少なくとも1つと、吸水性ポリマーと、ポリエーテル変性シロキサンと、炭素数2~8のジオールと、を含む、
防曇膜付きガラス物品、を提供する。
シランカップリング剤と、吸水性ポリマーと、ポリエーテル変性シロキサンと、炭素数2~8のジオールと、を含む、
防曇膜形成用塗布液、を提供する。
ガラス基材10は、その形状及び材料に特段の制限はない。ガラス基材10は、例えば、ガラス板である。
防曇膜11及び21の膜厚は、特定の値に限定されず、0.1~10μm、好ましくは0.5~5.0μm、特に好ましくは0.8~2.0μm、である。
吸水性ポリマーとしては、ウレタン樹脂、エポキシ樹脂、アクリル樹脂、ポリビニルアセタール樹脂、及びポリビニルアルコール樹脂からなる群より選択される少なくとも1種を例示できる。ウレタン樹脂としては、ポリイソシアネートとポリオールとで構成されるポリウレタン樹脂が挙げられる。ポリオールとしては、アクリルポリオール及びポリオキシアルキレン系ポリオールが挙げられる。エポキシ系樹脂としては、グリシジルエーテル系エポキシ樹脂、グリシジルエステル系エポキシ樹脂、グリシジルアミン系エポキシ樹脂、環式脂肪族エポキシ樹脂が挙げられる。好ましいエポキシ樹脂は、環式脂肪族エポキシ樹脂である。以下、好ましい吸水性ポリマーであるポリビニルアセタール樹脂(以下、単に「ポリビニルアセタール」)について説明する。
ポリエーテル変性シロキサンは、シロキサンの主鎖の末端に結合した分子鎖、及びシロキサンの主鎖の側鎖として結合した分子鎖から選択される少なくとも1つとしてポリエーテル鎖を有する化合物である。シロキサンは、シロキサン結合(Si-O-Si)を骨格とする化合物である。ポリエーテル鎖の構成単位は、特に限定されないが、例えば、エチレンオキサイド、プロピレンオキサイドである。ポリエーテル鎖は、構成単位として、1種のみを含んでいてもよく、2種以上を含んでいてもよい。
炭素数2~8のジオール、言い換えると2~8個の炭素原子を有するジオールは、例えば、エチレングリコール、ジエチレングリコール、プロピレングリコール、ジプロピレングリコール、ブタンジオール、ペンタンジオール、及びヘキサンジオールからなる群より選択される少なくとも1つを含んでいてもよい。ジオールは、炭素数2~6のジオールであってもよい。好ましいジオールとしては、ブタンジオール、プロピレングリコール、及びジプロピレングリコールからなる群より選択される少なくとも1つを例示できる。防曇膜は、特に好ましいジオールとして、プロピレングリコール及び/又はジプロピレングリコールを含んでいてもよい。
シランカップリング剤は、以下の式(I)で表される加水分解性基を有するシリコン化合物を例示できる。式(I)で表される加水分解性基を有するシリコン化合物は、単独で使用してもよく、2種以上を併用してもよい。
防曇膜は、上述の成分以外にも、適宜、紫外線吸収剤、赤外線吸収剤、レベリング剤(表面調整剤)、光安定化剤等を含んでいてもよい。ただし、これらの成分は、防曇膜の20質量%以下、さらに10質量%以下、特に5質量%以下の範囲で添加することが望ましい。防曇膜において、コロイダルシリカその他のシリカ微粒子は、含まれていてもよく、含まれていなくてもよい。防曇膜におけるシリカ微粒子の含有率は、例えば10~60質量%であってもよいが、5質量%未満、さらに3質量%未満、特に1質量%未満に制限されていてもよい。シリカ微粒子を含む酸化物微粒子の含有率も、シリカ微粒子について上述した程度であってもよい。防曇膜は、シリカ微粒子に代表される酸化物微粒子を含んでいてもよく、含んでいなくてもよい。ただし、シリカ微粒子を含まない場合であっても、防曇膜は、ポリエーテル変性シロキサン、シランカップリング剤等に含まれるシロキサン成分を含みうる。
本実施形態による防曇膜は、防曇膜にとって厳しい環境、例えば水と接触した状態で長い期間置かれた後であっても、高いレベルで透過光を散乱させずに透過させる機能を発揮しうる。この機能を発揮するために、防曇膜は、透明性及び吸水性と共に、耐水性を有し、さらに親水性を有することが望ましい。耐水性が十分でない防曇膜は、水分に長時間接するとその成分が溶出することがある。また、防曇膜は、自身による吸水が飽和状態に達したときに、その表面に余剰の水が連続した膜として保持される程度に親水性であれば、透過光は過度に散乱することなく透過しうる。
本実施形態による防曇膜付きガラス物品は、透過光を過度に散乱することなく透過させうる。具体的に、防曇膜付きガラス物品は、例えば、5%以下、さらに3%以下、特に1%以下、場合によっては0.4%以下、のヘイズ率を有しうる。ヘイズ率は、JIS K 7136:2018に規定されている。
本実施形態による防曇膜付きガラス物品によれば、耐水性と親水性とが両立しうる。これらの特性は、実施例の欄で詳細を記述する高温水蒸気評価と呼ぶ方法により評価できる。この方法では、防曇膜を鉛直方向下方に向けた状態で防曇膜に高温かつ過剰の水蒸気が供給される。この水蒸気に接した際、親水性かつ耐水性に優れた防曇膜の表面には、水蒸気に直接曝された部分において水の透明な連続膜が形成される。「透明な連続膜」であるかは、水滴ではなく膜としての連続性が確保され、かつその膜が白濁していないことを目視により確認することにより判断できる。膜の白濁は、耐水性の不足による膜の白化により、又は防曇性の不足による膜表面の結露により、生じうる。親水性が不足している膜の表面では、水は、連続膜として保持されず、水滴として分散して付着する。耐水性が不足している膜には、高温水蒸気との接触により膜の白化が観察され、膜の成分の溶出や膜の欠損が生じることもある。表面の親水性は、一般に、水の接触角により評価される。しかし、この評価法では、ごく少量の水滴が膜に滴下されるのみであるから、厳しい環境を十分に再現したことにはならない。なお、透明な連続膜は、水蒸気に曝された防曇膜の表面の80%以上、さらに90%以上を被覆していてもよい。
防曇膜は、防曇膜を形成するための塗布液(防曇膜形成用塗布液)をガラス基材の表面に塗布し、塗布液により塗膜を形成したガラス基材を加熱することにより、成膜できる。塗布液の調製に用いる溶媒、塗布液の塗布方法は、従来から公知の材料及び方法を用いればよい。塗布方法の例は、スピンコート法、ロールコート法、スプレーコート法、ディップコート法、フローコート法、スクリーン印刷法、及び刷毛塗法である。塗膜は、加熱の前に適宜乾燥させてもよい。
室温(約25℃)の純水を保持したプラスチック製容器に防曇膜付きガラス物品を浸漬させ、この状態で24時間保持した。その後、防曇膜付きガラス物品を取り出し、ホルダーに立てかけて乾燥させた。乾燥後のサンプルについて、以下の外観評価及び高温水蒸気評価を実施した。
膜面の状態が下記のいずれに該当するかを肉眼で評価した。
G :試験前と比較して変化なし。
F :若干の白化が観察される。
NG:白化が観察される。
Y :膜の溶出が観察される。
図3Aに示すように、沸騰させた水70を内部に保持したステンレス製保温カップ80の上方に、防曇膜11が形成された面が保温カップ80側を向くように防曇膜付きガラス物品1を水平に保持した。水蒸気を供給する間、水70の温度は90~100℃に維持されていた。防曇膜11と水面との間の距離D1は60mmとした。なお、保温カップ80の内部空間は開口部の直径が64mmの円柱状であり、水70の体積は約130ccとした。保温カップ80上で防曇膜付きガラス物品1を30秒間保持して防曇膜11に高温の水蒸気を供給した。その後、図3Bに示すように、保温カップ80を撤去し、これに代えて所定のQRコード90を印刷した台紙95を配置した。防曇膜11と台紙95との間の距離D2は110mmとした。この状態で、防曇膜付きガラス物品1を介してその上方からカメラ100によりQRコード90を撮影し、QRコード90が有する情報を読み取れるかを確認した。ガラス基材10とカメラ100のレンズ101との間の距離D3は80mmとした。保温カップ80の撤去、すなわち高温水蒸気の供給の停止からQRコードの撮影までは30秒以内に実施した。
QRコード「SSS」:10mm×10mm、情報「Rank:SSS」(図4A)
QRコード「SS」 :15mm×15mm、情報「Rank:SS」(図4B)
QRコード「S」 :20mm×20mm、情報「Rank:S」(図4C)
QRコード「A」 :30mm×30mm、情報「Rank:A」(図4D)
QRコード「B」 :40mm×40mm、情報「Rank:B」(図4E)
(塗布液の調製)
ポリビニルアセタール樹脂含有溶液(積水化学工業社製「エスレックKX-5」、固形分8質量%、アセタール化度9モル%、ベンズアルデヒドに由来するアセタール構造を含む)48.2質量%、ポリエーテル変性シロキサン(ビックケミー社製「BYK-345」)0.5質量%、3-アミノプロピルトリエトキシシラン(APTES、信越シリコーン社製「KBE-903」)1.0質量%、3-グリシドキシプロピルトリメトキシシラン(GPTMS、信越シリコーン社製「KBM-403」)0.7質量%、プロピレングリコール20.0質量%、精製水18.5質量%、アルコール溶媒(大伸化学社製「ネオエタノールP-7」)11.0質量%、塩酸0.1質量%、レベリング剤(信越シリコーン社製「KP-341」)0.01質量%をガラス製容器に投入して攪拌し、塗布液を調製した。
アルカリ洗浄によって予め洗浄したフロートガラス(サイズ:50mm×50mm、厚み:1.1mm)のボトム面に、スピンコート法(1500rpm、10秒)により塗布液を塗布して塗膜を形成した。次に、100℃、30分間の条件で塗膜を形成したフロートガラスを加熱し、防曇膜付きガラス物品を得た。
塗布液の調製において、表1に記載した原料及び量を用いたことを除き、実施例1と同様にして防曇膜付きガラス物品を得た。なお、実施例2では、エポキシシランの減量分だけアルコール溶媒を増量した。実施例3では、ポリエーテル変性シロキサンの増量分だけアルコール溶媒を減量した。BYK-347、349、3455はビックケミー社製の、TSF-4440はモメンティブ・パフォーマンス・マテリアルズ社製のポリエーテル変性シリコーンである。
塗布液の調製において、表2に記載した原料及び量を用いたことを除き、実施例1と同様にして防曇膜付きガラス物品を得た。なお、比較例10で使用した「A-80」(ラピゾールA-80)は日油社製の製品名である。また、比較例1~6では、添加しないこととした成分の量だけアルコール溶媒を増量した。
防曇膜を形成した初期状態で、上記と同様にして、外観評価を実施し、さらにヘイズ率を測定した。
防曇膜を形成した初期状態において、高温水蒸気評価と同様にして、高温の水蒸気を防曇膜に供給した。その後、防曇膜付きガラス物品を取り出し、ホルダーに立てかけて乾燥させた。乾燥後のサンプルに、再度、高温の水蒸気を防曇膜に供給し、乾燥させることを繰り返し、高温の水蒸気を防曇膜に10回供給した。その後、上記と同様にして、外観評価及び高温水蒸気評価を実施した。なお、高温水蒸気評価において、QRコードの撮影は、11回目の水蒸気の供給を省いて(10回目の水蒸気供給を終えた後直ちに)実施した。
エタノールを主成分とするアルコール溶媒(双葉化学薬品社製「ファインエターA-10」)0.5ccを25mm幅にカットした不織布ウエス(旭化成社製「ベンコットM-3II」)に滴下して染み込ませた後、防曇膜付きガラス物品と共に往復摩耗試験にセットした。ウエスに400gの荷重を加えた状態で、長さ30mmを20往復させた。その後、上記と同様にして、外観評価及び高温水蒸気評価を実施した。
Claims (9)
- ガラス基材と、前記ガラス基材の表面上の防曇膜と、を備え、
25℃の水に24時間浸漬させて前記水から取り出し、前記防曇膜から鉛直方向下方に60mm離間させて配置した90℃~100℃の水から発生する水蒸気に前記防曇膜を30秒間曝し、前記防曇膜から前記方向下方に110mm離れて配置したQRコードの情報を前記防曇膜が形成された側とは反対側からカメラを使用して読み取ることができるかを判定する試験において、40mm四方のサイズを有するQRコードの情報を読み取ることができる、
防曇膜付きガラス物品。
ここで、前記QRコードは、日本産業規格(JIS)X 0510:2018に従って、21×21モジュールのシンボルサイズ及びレベルHの誤り訂正の仕様により、前記情報として文字列「Rank:B」を符号化した二次元コードである。 - ガラス基材と、前記ガラス基材の表面上の防曇膜と、を備え、
前記防曇膜は、吸水性ポリマーを含み、
25℃の水に24時間浸漬させて前記水から取り出し、前記防曇膜から鉛直方向下方に60mm離間させて配置した90℃~100℃の水から発生する水蒸気に前記防曇膜を30秒間曝す試験を実施したときに、前記水蒸気に曝された前記防曇膜の前記表面に透明な連続膜が形成される、
防曇膜付きガラス物品。 - 前記防曇膜は、シランカップリング剤及びシランカップリング剤に由来する架橋構造からなる群より選択される少なくとも1つと、吸水性ポリマーと、ポリエーテル変性シロキサンと、炭素数2~8のジオールと、を含む、請求項1又は2に記載の防曇膜付きガラス物品。
- ガラス基材と、前記ガラス基材の表面上の防曇膜と、を備え、
前記防曇膜は、シランカップリング剤及びシランカップリング剤に由来する架橋構造からなる群より選択される少なくとも1つと、吸水性ポリマーと、ポリエーテル変性シロキサンと、炭素数2~8のジオールと、を含む、
防曇膜付きガラス物品。 - 前記ジオールは、エチレングリコール、ジエチレングリコール、プロピレングリコール、ジプロピレングリコール、ブタンジオール、ペンタンジオール、及びヘキサンジオールからなる群より選択される少なくとも1つを含む、請求項3又は4に記載の防曇膜付きガラス物品。
- 前記ジオールは、ブタンジオール、プロピレングリコール、及びジプロピレングリコールからなる群より選択される少なくとも1つを含む、請求項5に記載の防曇膜付きガラス物品。
- 前記吸水性ポリマーは、ポリビニルアセタール樹脂を含む、請求項3~6のいずれか1項に記載の防曇膜付きガラス物品。
- 前記ガラス基材は、フロートガラスであり、前記防曇膜は、前記フロートガラスのボトム面に形成されている、請求項1~7のいずれか1項に記載の防曇膜付きガラス物品。
- シランカップリング剤と、吸水性ポリマーと、ポリエーテル変性シロキサンと、炭素数2~8のジオールと、を含む、
防曇膜形成用塗布液。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP22820324.6A EP4353695A1 (en) | 2021-06-11 | 2022-06-09 | Antifogging film-including glass article, and antifogging film-forming application liquid |
| JP2022568664A JP7281608B2 (ja) | 2021-06-11 | 2022-06-09 | 防曇膜付きガラス物品及び防曇膜形成用塗布液 |
| CN202280041498.9A CN117858853A (zh) | 2021-06-11 | 2022-06-09 | 带有防雾膜的玻璃物品及防雾膜形成用涂布液 |
| JP2023036737A JP2023085291A (ja) | 2021-06-11 | 2023-03-09 | 防曇膜付きガラス物品及び防曇膜形成用塗布液 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021-098342 | 2021-06-11 | ||
| JP2021098342 | 2021-06-11 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022260145A1 true WO2022260145A1 (ja) | 2022-12-15 |
Family
ID=84424608
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/023355 WO2022260145A1 (ja) | 2021-06-11 | 2022-06-09 | 防曇膜付きガラス物品及び防曇膜形成用塗布液 |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP4353695A1 (ja) |
| JP (2) | JP7281608B2 (ja) |
| CN (1) | CN117858853A (ja) |
| TW (1) | TW202313864A (ja) |
| WO (1) | WO2022260145A1 (ja) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015186360A1 (ja) * | 2014-06-05 | 2015-12-10 | 日本板硝子株式会社 | 防曇膜つき透明物品 |
| JP2016113468A (ja) * | 2014-12-10 | 2016-06-23 | 積水化学工業株式会社 | 防曇性樹脂組成物 |
| WO2018110465A1 (ja) | 2016-12-12 | 2018-06-21 | 日本板硝子株式会社 | ウインドシールド、ウインドシールド用ガラス製品及び防曇部材 |
| WO2020111128A1 (ja) * | 2018-11-30 | 2020-06-04 | ダイキン工業株式会社 | 防曇塗料用組成物 |
-
2022
- 2022-06-09 CN CN202280041498.9A patent/CN117858853A/zh active Pending
- 2022-06-09 EP EP22820324.6A patent/EP4353695A1/en active Pending
- 2022-06-09 JP JP2022568664A patent/JP7281608B2/ja active Active
- 2022-06-09 WO PCT/JP2022/023355 patent/WO2022260145A1/ja active Application Filing
- 2022-06-10 TW TW111121670A patent/TW202313864A/zh unknown
-
2023
- 2023-03-09 JP JP2023036737A patent/JP2023085291A/ja active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015186360A1 (ja) * | 2014-06-05 | 2015-12-10 | 日本板硝子株式会社 | 防曇膜つき透明物品 |
| JP2016113468A (ja) * | 2014-12-10 | 2016-06-23 | 積水化学工業株式会社 | 防曇性樹脂組成物 |
| WO2018110465A1 (ja) | 2016-12-12 | 2018-06-21 | 日本板硝子株式会社 | ウインドシールド、ウインドシールド用ガラス製品及び防曇部材 |
| WO2020111128A1 (ja) * | 2018-11-30 | 2020-06-04 | ダイキン工業株式会社 | 防曇塗料用組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202313864A (zh) | 2023-04-01 |
| JPWO2022260145A1 (ja) | 2022-12-15 |
| CN117858853A (zh) | 2024-04-09 |
| JP7281608B2 (ja) | 2023-05-25 |
| EP4353695A1 (en) | 2024-04-17 |
| JP2023085291A (ja) | 2023-06-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10400130B2 (en) | Anti-fogging coated transparent article | |
| US7842753B2 (en) | Composite resin, coating composition containing such resin, and coated article | |
| EP2216379B1 (en) | Photocurable coating composition, film forming method, and coated article | |
| EP3138880A1 (en) | Liquid composition and antibacterial article | |
| WO2011004873A1 (ja) | 防曇性物品およびその製造方法、防曇膜形成用コーティングキット | |
| US20230057817A1 (en) | Transparent laminate | |
| JP2012017394A (ja) | 防曇性物品 | |
| JP2009256563A (ja) | 熱硬化性ハードコート剤組成物、成形品、およびレンズ | |
| JP2014037326A (ja) | 窓ガラスおよびその製造方法 | |
| WO2011090156A1 (ja) | 防曇性物品 | |
| JP7281608B2 (ja) | 防曇膜付きガラス物品及び防曇膜形成用塗布液 | |
| US20220234947A1 (en) | A 3d structure for easy-to-clean coatings | |
| JP7281607B2 (ja) | セキュリティカメラ用防曇膜付き透明物品及びセキュリティカメラ | |
| JP7210812B1 (ja) | 防曇膜付き樹脂基板、防曇膜付き透明物品、及び塗布液 | |
| EP4353693A1 (en) | Antifog film-equipped glass article for security cameras, security camera, and method for producing antifog film-equipped glass article for security cameras | |
| JP2021107198A (ja) | 飛行体 | |
| JPWO2017183700A1 (ja) | ウインドシールド | |
| JP2011153164A (ja) | 耐熱性防曇膜及びその形成方法並びに耐熱性防曇膜形成用塗布剤 | |
| JP4446654B2 (ja) | 防曇剤及び防曇性被膜の形成方法 | |
| JP2012017220A (ja) | 防曇性物品 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2022568664 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22820324 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280041498.9 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18569088 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022820324 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022820324 Country of ref document: EP Effective date: 20240111 |