WO2020151728A1 - 2-氧代-1,2-二氢吡啶类衍生物、其制备方法及其在医药上的应用 - Google Patents
2-氧代-1,2-二氢吡啶类衍生物、其制备方法及其在医药上的应用 Download PDFInfo
- Publication number
- WO2020151728A1 WO2020151728A1 PCT/CN2020/073668 CN2020073668W WO2020151728A1 WO 2020151728 A1 WO2020151728 A1 WO 2020151728A1 CN 2020073668 W CN2020073668 W CN 2020073668W WO 2020151728 A1 WO2020151728 A1 WO 2020151728A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- general formula
- group
- pharmaceutically acceptable
- tautomer
- racemate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *c(c(*)c1*)c(*)c(C(NC(C=CN2CO*)=CC2=O)=O)c1Nc1c(*)c(O*)c(*)c(*)c1* Chemical compound *c(c(*)c1*)c(*)c(C(NC(C=CN2CO*)=CC2=O)=O)c1Nc1c(*)c(O*)c(*)c(*)c1* 0.000 description 2
- KPAPRPPQXFDQLL-UHFFFAOYSA-N Cc1c(Cc(cc(C(F)(F)F)c(Cl)c2)c2C(NC(C=CN2CCl)=CC2=O)=O)ccc(F)c1 Chemical compound Cc1c(Cc(cc(C(F)(F)F)c(Cl)c2)c2C(NC(C=CN2CCl)=CC2=O)=O)ccc(F)c1 KPAPRPPQXFDQLL-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/66—Phosphorus compounds
- A61K31/675—Phosphorus compounds having nitrogen as a ring hetero atom, e.g. pyridoxal phosphate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/72—Nitrogen atoms
- C07D213/75—Amino or imino radicals, acylated by carboxylic or carbonic acids, or by sulfur or nitrogen analogues thereof, e.g. carbamates
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/553—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having one nitrogen atom as the only ring hetero atom
- C07F9/576—Six-membered rings
Definitions
- the present disclosure belongs to the field of medicine, and relates to a 2-oxo-1,2-dihydropyridine derivative, a preparation method thereof, and application in medicine.
- the present disclosure relates to 2-oxo-1,2-dihydropyridine derivatives represented by general formula (I), methods for their preparation, and pharmaceutical compositions containing the derivatives, and their use as Na V inhibitors And its use in the preparation of medicines for treating and/or reducing pain and pain-related diseases.
- Pain is a complex physical and psychological activity and one of the most common symptoms in clinical practice.
- the International Association for Pain Research defines pain as "an unpleasant feeling and emotional feeling, accompanied by substantial or potential tissue damage, which is a subjective feeling.” Pain can be used as a warning signal to remind the body to pay attention to potential dangers, and has an indispensable protective effect on the body's normal life activities. At the same time, pain is also a common clinical symptom. After the external stimulus that caused the pain disappears, strong or long-lasting pain can cause disorders of physiological functions and seriously affect the quality of life of the living body. Statistics show that about one-fifth of people in the world suffer from moderate to severe chronic pain.
- action potentials nerve impulses
- DRG dorsal root ganglia
- Na V voltage-gated sodium channels
- Na V is a type of transmembrane ion channel protein. These proteins are composed of an alpha subunit with a molecular weight of 260kD and a beta subunit with a molecular weight of 30-40kD. According to the different ⁇ subunits, it can be divided into 9 subtypes, Na V ll ⁇ Na V 1.9. Different subtypes show different tissue distribution and electrophysiological and pharmacological characteristics (Rush AM, et al. J. Physiol. 2007, 579, 1-14). According to whether it can be effectively inhibited by nanomolar tetrodotoxin (TTX), sodium ion channels are divided into TTX-sensitive (TTX-S) and TTX-insensitive (TTX-R).
- TTX-S TTX-sensitive
- TTX-R TTX-insensitive
- Na V 1.1, Na V 1.2, Na V 1.3, and Na V 1.7 are TTX-S type, and the coding genes are located in human chromosome 2q23-24, and they are expressed in large amounts in neurons.
- Na V 1.5, Na V 1.8 and Na V 1.9 are TTX-R type, and the coding gene is located on human chromosome 3p21-24.
- Na V 1.5 is mainly present in cardiomyocytes
- Na V 1.8 and Na V 1. 9 are present in the peripheral nervous system (Goldin A.L., et al. Annu. Rev. Physiol. 2001, 63, 871-894).
- Both Na V 1.4 and Na V 1.6 are of TTX-S type, which are abundantly present in skeletal muscle and central nervous system respectively (Fozzard HA, et al. Physiol. Rev. 1996, 76, 887-926). Local anesthetic lidocaine for pain by inhibiting Na V.
- Non-selective Na V inhibitors such as lamotrigine, lacosamide, and mexiletine, have been successfully used to treat chronic pain.
- Na V 1.8 is TTX-R type
- the coding gene is SCN10A, which mainly exists in trigeminal ganglion neurons and DRG neurons, and has the electrophysiological characteristics of slow inactivation and rapid recovery (Dib-Hajj SD, et al. Annu .Rev.Neurosci.2010,33,325–347).
- the rise of action potential is mainly composed of Na V 1.8 current.
- nerve damage will increase the expression level of Na V 1.8 in axons and neuron cell bodies (Sleeper AA, et al. J. Neurosci. 2000, 20, 7279-7289).
- Na V 1.8 antisense oligonucleotides can significantly relieve pain while reducing the expression of Na V 1.8 (Yoshimura N., et al. J. Neurosci. 2001, 21, 8690-8696).
- carrageenan was injected into the paws of rats, the expression of Na V 1.8 in DRG neurons increased (Tanaka M., et al. G. NeuroReport 1998, 9, 967-972).
- Na V 1.8 knockout mice cannot show normal visceral inflammation pain (Kerr BJ, et al. NeuroReport 2001, 12, 3077-3080).
- the human gene Na V 1.8 gain generation function mutations leads to peripheral neuropathic pain (Faber CG, et al.Proc.Natl.Acad.Sci.USA 2012,109,19444-19449) .
- selective inhibition of Na V 1.8 has the potential to become a new type of analgesic therapy, which can be used for the treatment of inflammatory pain, nerve pain, postoperative pain, cancer pain and other types of pain.
- Na V inhibitors used in clinical practice can inhibit the sodium ion channels expressed in the heart and central nervous system, so the therapeutic window is narrow and the scope of application is limited.
- Na V 1.8 is mainly distributed in the peripheral nervous system, so selective inhibition of Na V 1.8 can effectively reduce side effects.
- the purpose of the present disclosure is to provide a compound represented by general formula (IG), or its tautomer, meso, racemate, enantiomer, diastereomer or its In the form of a mixture, or a pharmaceutically acceptable salt thereof,
- R 0 is a hydrogen atom or -CH 2 OR x ;
- M is CR 7 R 8 or S atom
- Ring A is aryl or heteroaryl
- R 1 is selected from hydrogen atom, halogen, alkyl, alkoxy, haloalkyl, cyano, amino, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocyclic, aryl and heteroaryl;
- R 2 is selected from hydrogen atom, halogen, alkyl, deuterated alkyl, alkoxy, deuterated alkoxy, halogenated alkyl, halogenated alkoxy, cyano, amino, nitro, hydroxyl, hydroxyalkyl, cycloalkane Group, heterocyclyl, aryl and heteroaryl, wherein the alkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl are optionally selected from alkyl, haloalkyl, halogen, amino, nitro Substituted by one or more substituents in the group, cyano, hydroxy, alkoxy, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl;
- R 3 , R 4 , R 5 and R 6 are the same or different, and are each independently selected from a hydrogen atom, a halogen, an alkyl group, an alkoxy group, a halogenated alkyl group, a halogenated alkoxy group, a cyano group, an amino group, a nitro group, a hydroxyl group, Hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl;
- R 7 and R 8 are the same or different, and are each independently selected from hydrogen atom, halogen, alkyl, alkoxy, haloalkyl, cyano, amino, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocycle Group, aryl and heteroaryl;
- the R 7 and R 8 together with the connected carbon atoms form a carbonyl group, a cycloalkyl group and a heterocyclic group, wherein the cycloalkyl group and the heterocyclic group are optionally selected from alkyl, alkoxy, Substituted by one or more substituents among oxo, halogen, amino, cyano, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl;
- R x is selected from a hydrogen atom, -S (O) 2 OH, -S (O) 2 O - Q +, -PO (OR y) 2, -PO (OH) O - Q +, -PO (O -) 2 2Q + and -PO (O -) 2 W 2+ ;
- Q + is a monovalent pharmaceutically acceptable cation;
- W 2+ cation is a pharmaceutically acceptable divalent;
- R y is a hydrogen atom or an alkyl group
- n 0, 1, 2, 3, 4 or 5;
- t 0, 1, or 2.
- the compound represented by the general formula (IG), or its tautomer, meso, racemate, enantiomer, diastereomer Form or a mixture thereof, or a pharmaceutically acceptable salt thereof which is a compound represented by general formula (I), or a tautomer, meso, racemate, enantiomer, Diastereoisomers or mixtures thereof, or pharmaceutically acceptable salts thereof,
- M is CR 7 R 8 or S atom
- Ring A is aryl or heteroaryl
- R 1 is selected from hydrogen atom, halogen, alkyl, alkoxy, haloalkyl, cyano, amino, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocyclic, aryl and heteroaryl;
- R 2 is selected from hydrogen atom, halogen, alkyl, deuterated alkyl, alkoxy, deuterated alkoxy, halogenated alkyl, halogenated alkoxy, cyano, amino, nitro, hydroxyl, hydroxyalkyl, cycloalkane Group, heterocyclyl, aryl and heteroaryl, wherein the alkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl are optionally selected from alkyl, haloalkyl, halogen, amino, nitro Substituted by one or more substituents in the group, cyano, hydroxy, alkoxy, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl;
- R 3 , R 4 , R 5 and R 6 are the same or different, and are each independently selected from a hydrogen atom, a halogen, an alkyl group, an alkoxy group, a halogenated alkyl group, a halogenated alkoxy group, a cyano group, an amino group, a nitro group, a hydroxyl group, Hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl;
- R 7 and R 8 are the same or different, and are each independently selected from hydrogen atom, halogen, alkyl, alkoxy, haloalkyl, cyano, amino, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocycle Group, aryl and heteroaryl;
- the R 7 and R 8 together with the connected carbon atoms form a carbonyl group, a cycloalkyl group and a heterocyclic group, wherein the cycloalkyl group and the heterocyclic group are optionally selected from alkyl, alkoxy, Substituted by one or more substituents among oxo, halogen, amino, cyano, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl;
- n 0, 1, 2, 3, 4 or 5;
- t 0, 1, or 2.
- R 2 is selected from hydrogen atom, halogen, alky
- R 9 , R 10 , R 11 , R 12 and R 13 are the same or different, and are each independently selected from a hydrogen atom, a halogen, an alkyl group, a deuterated alkyl group, an alkoxy group, a deuterated alkoxy group, a halogenated alkyl group, Halogenated alkoxy, cyano, amino, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl, wherein the alkyl, cycloalkyl, heterocyclyl, aryl And heteroaryl are optionally selected from alkyl, haloalkyl, halogen, amino, nitro, cyano, hydroxy, alkoxy, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and One or more substituents in the heteroaryl group are substituted; and
- the compound represented by the general formula (IG), or its tautomer, meso, racemate, enantiomer, diastereomer In the form of a mixture thereof, or a pharmaceutically acceptable salt thereof, and further a compound represented by the general formula (IIG), or a tautomer, meso, racemate, enantiomer, Diastereoisomers or mixtures thereof, or pharmaceutically acceptable salts thereof,
- R 9 , R 10 , R 11 , R 12 and R 13 are the same or different, and are each independently selected from a hydrogen atom, a halogen, an alkyl group, a deuterated alkyl group, an alkoxy group, a deuterated alkoxy group, a halogenated alkyl group, Halogenated alkoxy, cyano, amino, nitro, hydroxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and heteroaryl, wherein the alkyl, cycloalkyl, heterocyclyl, aryl And heteroaryl are optionally selected from alkyl, haloalkyl, halogen, amino, nitro, cyano, hydroxy, alkoxy, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocyclyl, aryl and One or more substituents in the heteroaryl group are substituted; and
- R x , M, R 1 , R 3 to R 6 and t are as defined in the general formula (IG).
- R 9 , R 10 , R 11 , R 12 and R 13 are the same or different, and are each independently selected from a hydrogen atom, a halogen, an alkyl group, and a deuterated alkane Group, alkoxy group, deuterated alkoxy group, cycloalkyl group, halogenated alkyl group and
- R 9 , R 10 , R 11 , R 12 and R 13 are the same or different, and are each independently selected from a hydrogen atom, a halogen, an alkyl group, and an alkoxy group , Halogenated alkyl and halogenated alkoxy, preferably hydrogen atom, halogen and alkyl.
- the compound represented by the general formula (IG), or its tautomer, meso, racemate, enantiomer, diastereomer In the form of a mixture thereof, or a pharmaceutically acceptable salt thereof is further a compound represented by the general formula (III), or a tautomer, meso, racemate, enantiomer, Diastereoisomers or mixtures thereof, or pharmaceutically acceptable salts thereof,
- R 1 , R 4 , R 5 , R 9 , R 11 and t are as defined in the compound of general formula (II).
- R x , R 1 , R 4 , R 5 , R 9 , R 11 and t are as defined in the compound of general formula (IG).
- Typical compounds of general formula (I) include but are not limited to:
- Another aspect of the present disclosure provides a compound represented by general formula (IA), or its tautomer, meso, racemate, enantiomer, diastereomer or In the form of a mixture, or a pharmaceutically acceptable salt thereof,
- R a is an alkyl group, preferably a methyl group
- the compound of general formula (IA) is an intermediate for preparing the compound of general formula (I).
- Typical compounds of general formula (IA) include but are not limited to:
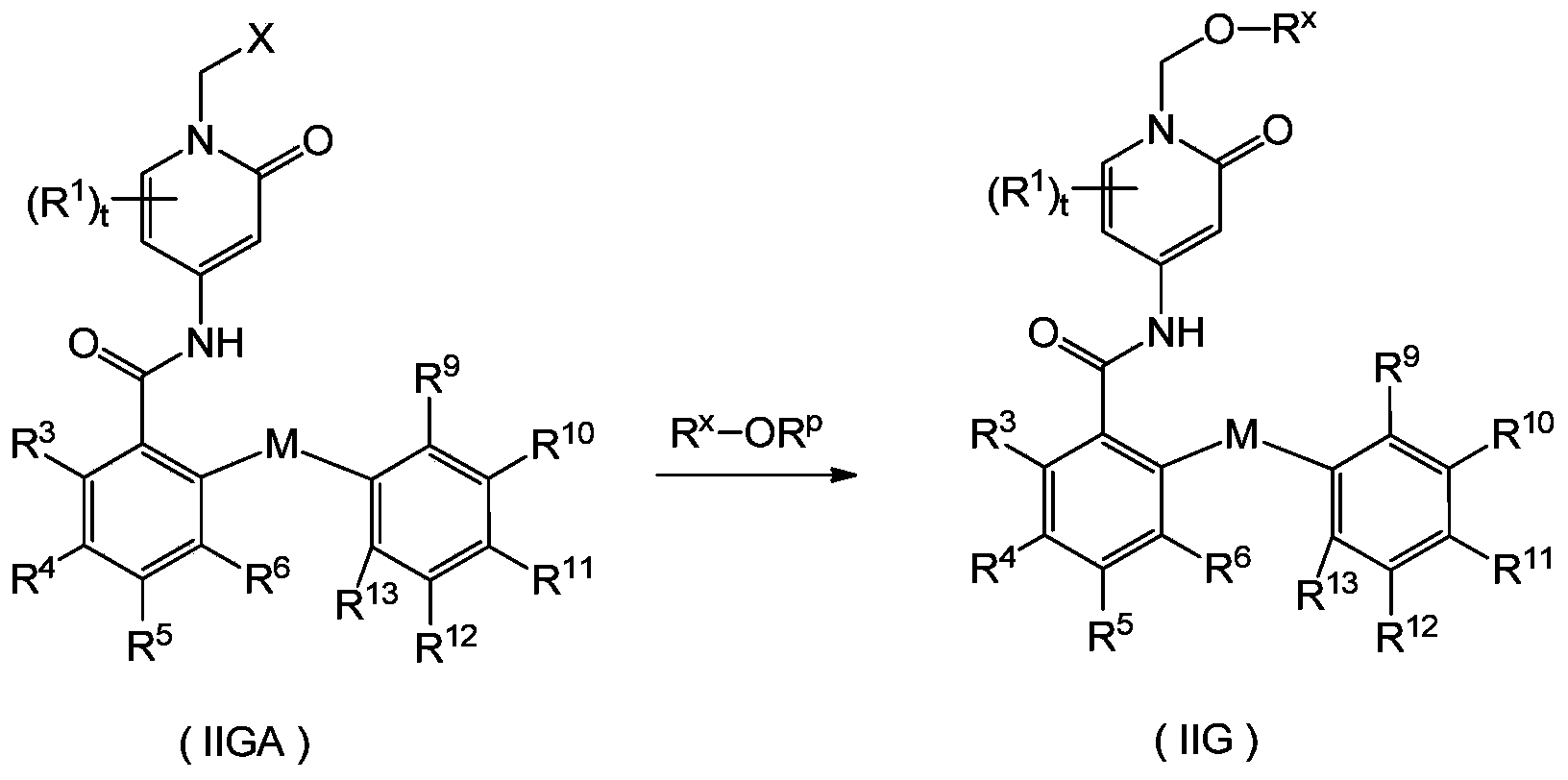
- Another aspect of the present disclosure provides a compound represented by general formula (IIGA), or its tautomer, meso, racemate, enantiomer, diastereomer or In the form of a mixture, or a pharmaceutically acceptable salt thereof,
- X is halogen; preferably Cl;
- R 1 , R 3 to R 6 , R 9 to R 13 and t are as defined in the compound of general formula (IIG).
- the compound of general formula (IIGA) is an intermediate for preparing the compound of general formula (IIG).
- Another aspect of the present disclosure provides a compound represented by general formula (IVGA), or its tautomer, meso, racemate, enantiomer, diastereomer or In the form of a mixture, or a pharmaceutically acceptable salt thereof,
- X is halogen; preferably Cl;
- R 1 , R 4 , R 5 , R 9 , R 11 and t are as defined in the compound of general formula (IVG).
- the compound of general formula (IVGA) is an intermediate for preparing the compound of general formula (IVG).
- Another aspect of the present disclosure relates to a method for preparing a compound represented by general formula (I), or its tautomer, meso, racemate, enantiomer, or diastereomer Or its mixture form, or its pharmaceutically acceptable salt method, the method comprises:
- the compound of general formula (I) is obtained by reacting compound of general formula (IA);
- R a is an alkyl group, preferably a methyl group
- Another aspect of the present disclosure relates to a preparation of the compound represented by general formula (II) or its tautomer, meso, racemate, enantiomer, diastereomer Or its mixture form, or its pharmaceutically acceptable salt method, the method includes:
- the compound of general formula (II) is obtained by reacting the compound of general formula (IIA);
- R a is an alkyl group, preferably a methyl group
- Another aspect of the present disclosure relates to a preparation of a compound represented by general formula (IIG) or its tautomer, meso, racemate, enantiomer, diastereomer Or its mixture form, or its pharmaceutically acceptable salt method, the method includes:
- X is halogen; preferably Cl;
- R p is a hydrogen atom or Q + ;
- Q + is a pharmaceutically acceptable monovalent cation, preferably K + ;
- R x , R 1 , R 3 to R 6 , R 9 to R 13 and t are as defined in the compound of general formula (IIG).
- Another aspect of the present disclosure relates to a preparation of a compound represented by general formula (III) or its tautomer, meso, racemate, enantiomer, diastereomer Or its mixture form, or its pharmaceutically acceptable salt method, the method includes:
- the compound of general formula (III) is obtained by reacting the compound of general formula (IIIA);
- R a is an alkyl group, preferably a methyl group
- Another aspect of the present disclosure relates to a preparation of the compound represented by general formula (IV) or its tautomer, meso, racemate, enantiomer, diastereomer Or its mixture form, or its pharmaceutically acceptable salt method, the method includes:
- the compound of general formula (IV) is obtained by reacting the compound of general formula (IVA);
- R a is an alkyl group, preferably a methyl group
- R 1 , R 4 , R 5 , R 9 , R 11 and t are as defined in the compound of general formula (IV).
- Another aspect of the present disclosure relates to a preparation of a compound represented by general formula (IVG) or its tautomers, mesosomes, racemates, enantiomers, and diastereomers Or its mixture form, or its pharmaceutically acceptable salt method, the method includes:
- X is halogen; preferably Cl;
- R p is a hydrogen atom or Q + ;
- Q + is a pharmaceutically acceptable monovalent cation, preferably K + ;
- R x , R 1 , R 4 , R 5 , R 9 , R 11 and t are as defined in the compound of general formula (IVG).
- compositions which contains the compound represented by the general formula (IG) as described above, or its tautomer, meso, racemate, enantiomer Conformers, diastereomers, or mixtures thereof, or pharmaceutically acceptable salts, and one or more pharmaceutically acceptable carriers, diluents or excipients.
- IG general formula
- the present disclosure also relates to a method for preparing the above-mentioned pharmaceutical composition, which comprises combining the compound represented by the general formula (IG) as described above, or its tautomer, meso, racemate, and The enantiomers, diastereomers, or mixtures thereof, or pharmaceutically acceptable salts thereof are mixed with a pharmaceutically acceptable carrier, diluent or excipient.
- a pharmaceutically acceptable carrier diluent or excipient.
- the present disclosure also relates to a compound represented by the general formula (IG) as described above, or its tautomer, meso, racemate, enantiomer, or diastereomer , Or the form of a mixture thereof, or a pharmaceutically acceptable salt thereof, or the use of the above-mentioned pharmaceutical composition in the preparation of a medicament for inhibiting a voltage-gated sodium ion channel of a subject.
- the voltage-gated sodium ion channel is preferably Na V 1.8.
- the present disclosure also relates to a compound represented by the general formula (IG) as described above, or its tautomer, meso, racemate, enantiomer, or diastereomer , Or a mixture thereof, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition as described above in the preparation for the treatment and/or alleviation of pain and pain-related diseases, multiple sclerosis, Xia-Martin-Tusan syndrome Use in medicines for symptom, incontinence or arrhythmia.
- the pain is preferably selected from chronic pain, acute pain, inflammatory pain, cancer pain, neuropathic pain, musculoskeletal pain, postoperative pain, visceral pain, intestinal pain and idiopathic pain.
- the present disclosure also relates to a method for inhibiting a voltage-gated sodium ion channel in a subject, the method comprising administering a compound represented by the above-mentioned general formula (IG) of the present disclosure, or a tautomer thereof, to a patient in need thereof Isomers, mesosomes, racemates, enantiomers, diastereomers, or mixtures thereof, or pharmaceutically acceptable salts thereof, or pharmaceutical compositions as described above.
- the voltage-gated sodium ion channel is preferably Na V 1.8.
- the present disclosure also relates to a method of treating and/or alleviating pain and pain-related diseases, multiple sclerosis, Char-Martin-Tuson syndrome, incontinence or arrhythmia drugs, the method comprising administering to a patient in need thereof
- the pain is preferably selected from chronic pain, acute pain, inflammatory pain, cancer pain, neuropathic pain, musculoskeletal pain, postoperative pain, visceral pain, intestinal pain and idiopathic pain.
- the present disclosure also relates to a compound represented by the general formula (IG), or a tautomer, meso, racemate, enantiomer, diastereomer or a mixture thereof , Or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition as described above, which is used as a medicine.
- IG general formula
- the present disclosure also relates to a compound represented by the general formula (IG), or a tautomer, meso, racemate, enantiomer, diastereomer or a mixture thereof , Or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition as described above, which is used as a drug for inhibiting voltage-gated sodium channels in a subject.
- the voltage-gated sodium ion channel is preferably Na V 1.8.
- the present disclosure also relates to a compound represented by the general formula (IG), or a tautomer, meso, racemate, enantiomer, diastereomer or a mixture thereof , Or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition as described above, which is used for the treatment and/or alleviation of pain and pain-related diseases, multiple sclerosis, Chama-Martinus syndrome, incontinence or heart rhythm Abnormal.
- the pain mentioned therein is preferably selected from chronic pain, acute pain, inflammatory pain, cancer pain, neuropathic pain, musculoskeletal pain, postoperative pain, visceral pain, intestinal pain and idiopathic pain.
- the neuropathic pain described in the present disclosure is preferably selected from the group consisting of trigeminal neuralgia, postherpetic neuralgia, diabetic neuralgia, painful HIV-related sensory neuralgia, burn syndrome, pain after amputation, pain after spinal cord injury, phantom Pain, painful neuroma, traumatic neuroma, Morton neuroma, nerve crush injury, spinal stenosis, carpal tunnel syndrome, nerve root pain, sciatica, nerve avulsion, brachial plexus avulsion Injuries, complex regional pain syndrome, neuralgia caused by drug therapy, neuralgia caused by cancer chemotherapy, neuralgia caused by antiretroviral therapy, idiopathic small fiber neuropathy, idiopathic sensory neuralgia, and trigeminal Autonomic headache.
- the musculoskeletal pain described in the present disclosure is preferably selected from osteoarthritis pain, back pain, cold pain, burning pain and toothache.
- the intestinal pain described in the present disclosure is preferably selected from inflammatory bowel disease pain, Crohn's disease pain and interstitial cystitis pain.
- the inflammatory pain described in the present disclosure is preferably selected from rheumatoid arthritis pain and vulvar pain.
- the idiopathic pain described in the present disclosure is preferably selected from fibromyalgia.
- the dosage of the compound or composition used in the treatment methods of the present disclosure will generally vary with the severity of the disease, the weight of the patient, and the relative efficacy of the compound.
- a suitable unit dose may be 0.1-1000 mg.
- the pharmaceutical composition of the present disclosure may contain one or more auxiliary materials selected from the following ingredients: fillers (diluents), binders, wetting agents, disintegrants or excipients Wait.
- auxiliary materials selected from the following ingredients: fillers (diluents), binders, wetting agents, disintegrants or excipients Wait.
- the composition may contain 0.1 to 99% by weight of the active compound.
- the pharmaceutical composition containing the active ingredient may be in a form suitable for oral administration, such as tablets, dragees, lozenges, water or oil suspensions, dispersible powders or granules, emulsions, hard or soft capsules, or syrups or Elixirs.
- the oral composition may be prepared according to any method known in the art for preparing pharmaceutical compositions, and such compositions may contain one or more ingredients selected from the group consisting of sweeteners, flavoring agents, coloring agents and preservatives, In order to provide pleasing and delicious medicinal preparations.
- the tablet contains the active ingredient and non-toxic pharmaceutically acceptable excipients suitable for the preparation of tablets for mixing. These excipients can be inert excipients, granulating and disintegrating agents and lubricants. These tablets may be uncoated or may be coated by a known technique that masks the taste of the drug or delays disintegration and absorption in the gastrointestinal tract, thereby providing a sustained release effect over a longer period of time.
- Oral preparations can also be provided in soft gelatin capsules in which the active ingredient is mixed with an inert solid diluent or the active ingredient is mixed with a water-soluble carrier or oil vehicle or olive oil.
- Aqueous suspensions contain the active substance and excipients suitable for the preparation of aqueous suspensions for mixing. Such excipients are suspending agents, dispersing agents or wetting agents.
- the aqueous suspension may also contain one or more preservatives such as ethyl paraben or n-propyl paraben, one or more coloring agents, one or more flavoring agents and one or more sweeteners. Flavoring agent.
- Oil suspensions can be formulated by suspending the active ingredients in vegetable oil or mineral oil.
- the oil suspension may contain thickeners.
- the above-mentioned sweeteners and flavoring agents can be added to provide a palatable preparation. These compositions can be preserved by adding antioxidants.
- dispersible powders and granules suitable for preparing aqueous suspensions can be provided with active ingredients and dispersing or wetting agents for mixing, suspending agents or one or more preservatives. Suitable dispersing or wetting agents and suspending agents can illustrate the above examples. Other excipients such as sweeteners, flavoring agents and coloring agents may also be added.
- the pharmaceutical composition of the present disclosure may also be in the form of an oil-in-water emulsion.
- the oil phase may be vegetable oil or mineral oil such as liquid paraffin or a mixture thereof. Suitable emulsifiers can be naturally occurring phospholipids or partial esters.
- the emulsion may also contain sweetening agents, flavoring agents, preservatives and antioxidants.
- the pharmaceutical composition of the present disclosure may be in the form of a sterile injectable aqueous solution.
- Acceptable solvents or solvents that can be used include water, Ringer's solution and isotonic sodium chloride solution.
- the sterile injection preparation may be a sterile injection oil-in-water microemulsion in which the active ingredient is dissolved in an oil phase. The injection or microemulsion can be injected into the patient's bloodstream by local large-scale injection.
- the pharmaceutical composition of the present disclosure may be in the form of a sterile injection water or oil suspension for intramuscular and subcutaneous administration.
- the suspension can be formulated according to known techniques using those suitable dispersing or wetting agents and suspending agents mentioned above.
- the sterile injection preparation can also be a sterile injection solution or suspension prepared in a parenterally acceptable non-toxic diluent or solvent.
- sterile fixed oil can be conveniently used as a solvent or suspension medium.
- the compounds of the present disclosure can be administered in the form of suppositories for rectal administration.
- These pharmaceutical compositions can be prepared by mixing the drug with a suitable non-irritating excipient that is solid at ordinary temperatures but liquid in the rectum and thus will melt in the rectum to release the drug.
- the dosage of the drug depends on many factors, including but not limited to the following factors: the activity of the specific compound used, the age of the patient, the weight of the patient, the health of the patient, and the behavior of the patient , The patient’s diet, time of administration, mode of administration, rate of excretion, combination of drugs, etc.; in addition, the best mode of treatment such as the mode of treatment, the daily dosage of the compound (I) or the amount of pharmaceutically acceptable salt
- the type can be verified according to the traditional treatment plan.
- This application provides a Na V inhibitor or its prodrug with a novel structure represented by the general formula (IG), which has a significant inhibitory effect on the activity of the Nav1.8 channel, especially R 4 in the general formula (IG)
- R 5 is halogen and R 5 is haloalkyl
- the biological activity of the entire molecule is significantly improved.
- alkyl refers to a saturated aliphatic hydrocarbon group, which is a straight or branched chain group containing 1 to 20 carbon atoms, preferably an alkyl group containing 1 to 12 carbon atoms, more preferably containing 1 to 6 carbon atoms Atom of the alkyl group.
- Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, sec-butyl, n-pentyl, 1,1-dimethylpropyl, 1 ,2-Dimethylpropyl, 2,2-Dimethylpropyl, 1-ethylpropyl, 2-methylbutyl, 3-methylbutyl, n-hexyl, 1-ethyl-2- Methylpropyl, 1,1,2-trimethylpropyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 2,2-dimethylbutyl, 1,3 -Dimethylbutyl, 2-ethylbutyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 2,3-dimethylbutyl, n-heptyl, 2 -Methylhexyl, 3-methylhexyl, 4-methylhe
- lower alkyl groups containing 1 to 6 carbon atoms More preferred are lower alkyl groups containing 1 to 6 carbon atoms.
- Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, and sec-butyl.
- Alkyl groups may be substituted or unsubstituted.
- substituents When substituted, substituents may be substituted at any available attachment point.
- the substituents are preferably one or more of the following groups, which are independently selected from alkanes Group, alkenyl, alkynyl, alkoxy, alkylthio, alkylamino, halogen, mercapto, hydroxyl, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkane Oxy, heterocycloalkoxy, cycloalkylthio, heterocycloalkylthio, oxo, carboxy, or carboxylate.
- alkoxy refers to -O- (alkyl) and -O- (unsubstituted cycloalkyl), where alkyl is as defined above.
- alkoxy groups include: methoxy, ethoxy, propoxy, butoxy, cyclopropoxy, cyclobutoxy, cyclopentyloxy, cyclohexyloxy.
- the alkoxy group may be optionally substituted or unsubstituted.
- the substituent group is preferably one or more of the following groups, which are independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkane Thio, alkylamino, halogen, mercapto, hydroxy, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkoxy, heterocycloalkoxy, cycloalkylthio , Heterocycloalkylthio, carboxy or carboxylate.
- cycloalkyl refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent.
- the cycloalkyl ring contains 3 to 20 carbon atoms, preferably 3 to 12 carbon atoms, more preferably 3 to 6 Carbon atoms (e.g. 3, 4, 5 or 6 carbon atoms), most preferably 5 to 6 carbon atoms.
- Non-limiting examples of monocyclic cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, cycloheptyl, cycloheptatriene Groups, cyclooctyl groups, etc.; polycyclic cycloalkyls include spiro, fused, and bridged cycloalkyls.
- spirocycloalkyl refers to a polycyclic group that shares one carbon atom (called a spiro atom) between 5- to 20-membered monocyclic rings, which may contain one or more double bonds, but none of the rings have complete conjugate ⁇ electronic system. It is preferably 6 to 14 yuan, more preferably 7 to 10 yuan (for example, 7, 8, 9 or 10 yuan).
- the spirocycloalkyl group is classified into a single spirocycloalkyl group, a bispirocycloalkyl group or a polyspirocycloalkyl group, preferably a single spirocycloalkyl group and a bispirocycloalkyl group. More preferably, it is a 4-membered/4-membered, 4-membered/5-membered, 4-membered/6-membered, 5-membered/5-membered, or 5-membered/6-membered monospirocycloalkyl.
- spirocycloalkyl groups include:
- fused cycloalkyl refers to a 5- to 20-membered all-carbon polycyclic group in which each ring in the system shares an adjacent pair of carbon atoms with other rings in the system, wherein one or more rings may contain one or Multiple double bonds, but none of the rings have a fully conjugated ⁇ -electron system. It is preferably 6 to 14 yuan, more preferably 7 to 10 yuan. According to the number of constituent rings, it can be classified into bicyclic, tricyclic, tetracyclic or polycyclic condensed cycloalkyls, preferably bicyclic or tricyclic, more preferably 5-membered/5-membered or 5-membered/6-membered bicyclic alkyl.
- fused cycloalkyl groups include:
- bridged cycloalkyl refers to a 5- to 20-membered, all-carbon polycyclic group with any two rings sharing two carbon atoms that are not directly connected. It may contain one or more double bonds, but no ring has complete Conjugated ⁇ electron system. It is preferably 6 to 14 yuan, more preferably 7 to 10 yuan. According to the number of constituent rings, it can be classified into bicyclic, tricyclic, tetracyclic or polycyclic bridged cycloalkyl groups, preferably bicyclic, tricyclic or tetracyclic, and more preferably bicyclic or tricyclic.
- bridged cycloalkyl groups include:
- the cycloalkyl ring includes the cycloalkyl as described above (including monocyclic, spiro, fused, and bridged rings) fused on an aryl, heteroaryl or heterocycloalkyl ring, where it is connected to the parent structure
- the ring together is a cycloalkyl group, non-limiting examples include indanyl, tetrahydronaphthyl, benzocycloheptyl, etc.; preferably phenylcyclopentyl, tetrahydronaphthyl. Cycloalkyl groups may be optionally substituted or unsubstituted.
- the substituents are preferably one or more of the following groups, which are independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkane Thio, alkylamino, halogen, mercapto, hydroxy, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkoxy, heterocycloalkoxy, cycloalkylthio , Heterocycloalkylthio, oxo, carboxy, or carboxylate.
- heterocyclyl refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent which contains 3 to 20 ring atoms, one or more of which is selected from nitrogen, oxygen or S(O) m (where m is an integer of 0 to 2) heteroatoms, but does not include the ring part of -OO-, -OS- or -SS-, and the remaining ring atoms are carbon. It preferably contains 3 to 12 ring atoms, of which 1 to 4 are heteroatoms; most preferably contains 3 to 8 ring atoms, of which 1 to 3 are heteroatoms; most preferably contains 5 to 6 ring atoms, of which 1 to 2 or 1 to 3 are heteroatoms.
- Non-limiting examples of monocyclic heterocyclic groups include pyrrolidinyl, imidazolidinyl, tetrahydrofuranyl, tetrahydropyranyl, tetrahydrothienyl, dihydroimidazolyl, dihydrofuranyl, dihydropyrazolyl, dihydropyrazole Hydropyrrolyl, piperidinyl, piperazinyl, morpholinyl, thiomorpholinyl, homopiperazinyl, etc., preferably tetrahydropyranyl, piperidinyl, and pyrrolidinyl.
- Polycyclic heterocyclic groups include spiro, fused, and bridged heterocyclic groups.
- spiroheterocyclic group refers to a polycyclic heterocyclic group that shares one atom (called a spiro atom) between 5- to 20-membered monocyclic rings, wherein one or more ring atoms are selected from nitrogen, oxygen or S(O ) m (where m is an integer of 0 to 2) heteroatoms, and the remaining ring atoms are carbon. It can contain one or more double bonds, but none of the rings have a fully conjugated ⁇ -electron system. It is preferably 6 to 14 yuan, more preferably 7 to 10 yuan.
- the spiro heterocyclic group is classified into a single spiro heterocyclic group, a dispiro heterocyclic group or a polyspiro heterocyclic group, preferably a single spiro heterocyclic group and a dispiro heterocyclic group. More preferably, it is a 4-membered/4-membered, 4-membered/5-membered, 4-membered/6-membered, 5-membered/5-membered or 5-membered/6-membered monospiro heterocyclic group.
- Non-limiting examples of spiroheterocyclic groups include:
- fused heterocyclic group refers to a 5- to 20-membered polycyclic heterocyclic group in which each ring in the system shares an adjacent pair of atoms with other rings in the system.
- One or more rings may contain one or more Double bond, but none of the rings have a fully conjugated ⁇ -electron system, one or more of the ring atoms are heteroatoms selected from nitrogen, oxygen or S(O) m (where m is an integer from 0 to 2), and the rest of the ring
- the atom is carbon. It is preferably 6 to 14 yuan, more preferably 7 to 10 yuan.
- bicyclic, tricyclic, tetracyclic or polycyclic fused heterocyclic groups preferably bicyclic or tricyclic, more preferably 5-membered/5-membered or 5-membered/6-membered bicyclic fused heterocyclic group.
- fused heterocyclic groups include:
- bridged heterocyclic group refers to a 5- to 14-membered polycyclic heterocyclic group with any two rings sharing two atoms that are not directly connected. It may contain one or more double bonds, but none of the rings has a complete common A conjugated ⁇ -electron system in which one or more ring atoms are heteroatoms selected from nitrogen, oxygen, or S(O) m (where m is an integer of 0 to 2), and the remaining ring atoms are carbon. It is preferably 6 to 14 yuan, more preferably 7 to 10 yuan.
- bridged heterocyclic groups include:
- the heterocyclyl ring includes the heterocyclic group (including monocyclic, spiro heterocyclic, fused heterocyclic and bridged heterocyclic ring) as described above fused on an aryl, heteroaryl or cycloalkyl ring, wherein
- the rings linked together in the structure are heterocyclic groups, non-limiting examples of which include:
- the heterocyclic group may be optionally substituted or unsubstituted.
- the substituent is preferably one or more of the following groups, which are independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkane Thio, alkylamino, halogen, mercapto, hydroxy, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkoxy, heterocycloalkoxy, cycloalkylthio , Heterocycloalkylthio, oxo, carboxy, or carboxylate.
- aryl refers to a 6 to 14-membered all-carbon monocyclic or fused polycyclic (that is, rings sharing adjacent pairs of carbon atoms) with a conjugated ⁇ -electron system, preferably 6 to 10 members, such as benzene Base and naphthyl.
- the aryl ring includes the aryl ring as described above fused to a heteroaryl, heterocyclic or cycloalkyl ring, wherein the ring connected to the parent structure is an aryl ring, and non-limiting examples include :
- the aryl group may be substituted or unsubstituted.
- the substituent is preferably one or more of the following groups, which are independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkylthio, Alkylamino, halogen, mercapto, hydroxy, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkoxy, heterocycloalkoxy, cycloalkylthio, heterocycle Alkylthio, carboxyl or carboxylate.
- heteroaryl refers to a heteroaromatic system containing 1 to 4 heteroatoms and 5 to 14 ring atoms, where the heteroatoms are selected from oxygen, sulfur and nitrogen.
- Heteroaryl groups are preferably 5 to 10 members, containing 1 to 3 heteroatoms; more preferably 5 or 6 members, containing 1 to 2 heteroatoms; preferably, for example, imidazolyl, furyl, thienyl, thiazolyl, pyridine Azolyl, oxazolyl, pyrrolyl, tetrazolyl, pyridyl, pyrimidinyl, thiadiazole, pyrazinyl and pyridazinyl, etc., preferably pyridazinyl and pyridinyl; more preferably pyridinyl.
- the heteroaryl ring includes the aforementioned heteroaryl ring fused to an aryl, heterocyclic or cycloalkyl ring, wherein the ring connected to the parent structure is a heteroaryl ring, which is not limiting Examples include:
- the heteroaryl group may be optionally substituted or unsubstituted.
- the substituent is preferably one or more of the following groups, which are independently selected from alkyl, alkenyl, alkynyl, alkoxy, alkane Thio, alkylamino, halogen, mercapto, hydroxy, oxo, nitro, cyano, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, cycloalkoxy, heterocycloalkoxy, Cycloalkylthio, heterocycloalkylthio, carboxyl or carboxylate.
- hydroxyalkyl refers to an alkyl group substituted with a hydroxy group, where the alkyl group is as defined above.
- haloalkyl refers to an alkyl group substituted with one or more halogens, where the alkyl group is as defined above.
- deuterated alkyl refers to an alkyl group substituted with one or more deuterium atoms, where the alkyl group is as defined above.
- haloalkoxy refers to an alkoxy group substituted with one or more halogens, where alkoxy is as defined above.
- deuterated alkoxy refers to an alkoxy group substituted with one or more deuterium atoms, where the alkoxy group is as defined above.
- cycloalkylalkyl refers to an alkyl group substituted with one or more cycloalkyl groups, wherein cycloalkyl and alkyl are as defined above.
- heterocyclylalkyl refers to an alkyl group substituted with one or more heterocyclyl groups, wherein heterocyclyl and alkyl are as defined above.
- arylalkyl refers to an alkyl group substituted with one or more aryl groups, where aryl and alkyl are as defined above.
- hydroxy refers to the -OH group.
- halogen refers to fluorine, chlorine, bromine or iodine.
- amino refers to -NH 2 .
- cyano refers to -CN.
- nitro refers to -NO 2 .
- carboxylate group refers to -C(O)O(alkyl) or -C(O)O(cycloalkyl), where alkyl and cycloalkyl are as defined above.
- acyl halide refers to a compound containing a -C(O)-halogen group.
- Q + pharmaceutically acceptable monovalent cation
- R y is H or C 1 -C 4 alkyl
- alkali metal ions such as potassium, sodium and lithium ions
- dicyclohexylamine ion and N-methyl D-reduced glucosamine ion.
- the term "pharmaceutically acceptable divalent cation” includes alkaline earth metal ions, such as calcium and magnesium ions, and divalent aluminum ions. It also includes amino acid cations, such as monovalent or divalent ions such as arginine, lysine, ornithine.
- the pharmaceutically acceptable divalent cation (W 2+ ) can be replaced by two pharmaceutically acceptable monovalent cations (Q + ).
- the compounds of the present disclosure may also include isotopic derivatives thereof.
- isotopic derivative refers to a compound that differs in structure only in the presence of one or more isotopically enriched atoms.
- isotopic derivative refers to a compound that differs in structure only in the presence of one or more isotopically enriched atoms.
- isotopic derivative refers to a compound that differs in structure only in the presence of one or more isotopically enriched atoms.
- isotopic derivative refers to a compound that differs in structure only in the presence of one or more isotopically enriched atoms.
- 18 F-fluorine label 18 F isotope
- 11 C-, 13 C-, or 14 C-rich Compounds in which collective carbons ( 11 C-, 13 C-, or 14 C-carbon labels; 11 C-, 13 C-, or 14 C- isotopes) replace carbon atoms are within the scope of the present disclosure.
- Such compounds can be used, for example, as
- the compounds of the present disclosure also include compounds of formula (I) in various deuterated forms. Each available hydrogen atom connected to a carbon atom can be independently replaced by a deuterium atom. Those skilled in the art can synthesize the compound of formula (I) in the deuterated form with reference to relevant literature. Commercially available deuterated starting materials can be used when preparing the deuterated form of the compound of formula (I), or they can be synthesized using conventional techniques using deuterated reagents.
- Deuterated reagents include but are not limited to deuterated borane, tri-deuterated Borane tetrahydrofuran solution, deuterated lithium aluminum hydride, deuterated ethyl iodide and deuterated methyl iodide, etc.
- heterocyclic group optionally substituted by an alkyl group means that an alkyl group may but does not have to be present.
- the description includes the case where the heterocyclic group is substituted by an alkyl group and the case where the heterocyclic group is not substituted by an alkyl group. .
- Substituted refers to one or more hydrogen atoms in the group, preferably up to 5, more preferably 1 to 3 hydrogen atoms independently of each other substituted by a corresponding number of substituents. It goes without saying that the substituents are only in their possible chemical positions, and those skilled in the art can determine (by experiment or theory) possible or impossible substitutions without too much effort. For example, an amino group or a hydroxyl group with free hydrogen may be unstable when combined with a carbon atom with an unsaturated (eg, olefinic) bond.
- “Pharmaceutical composition” means a mixture containing one or more of the compounds described herein or their physiologically/pharmaceutically acceptable salts or prodrugs and other chemical components, and other components such as physiological/pharmaceutically acceptable carriers And excipients.
- the purpose of the pharmaceutical composition is to promote the administration to the organism, which is beneficial to the absorption of the active ingredient and thus the biological activity.
- “Pharmaceutically acceptable salt” refers to the salt of the compound of the present disclosure. Such salt is safe and effective when used in mammals, and has due biological activity.
- the present disclosure provides novel inhibitors of NaV having formula (I) structure, a compound having such a structure has an excellent inhibitory effect of 1.8 to Na V.
- the compound represented by the general formula (I) of the present disclosure or its tautomer, meso, racemate, enantiomer, diastereomer, or mixture form thereof, or
- the preparation method of medicinal salt includes the following steps:
- R a is an alkyl group, preferably a methyl group
- the compound of general formula (I-1) and the compound of general formula (I-2) react under basic conditions in the presence of a chlorinating reagent to obtain a compound of general formula (IA);
- the compound of general formula (IA) is reacted under acidic conditions to obtain the compound of general formula (I).
- the chlorinating reagent includes, but is not limited to, phosphorus oxychloride, phosphorus trichloride, thionyl chloride and phosphorus pentachloride, preferably phosphorus oxychloride.
- the reagents that provide alkaline conditions include organic bases and inorganic bases.
- the organic bases include but are not limited to pyridine, hexahydropyridine, triethylamine, N,N-diisopropylethylamine, n-butyllithium, Lithium diisopropylamide, potassium acetate, sodium tert-butoxide and potassium tert-butoxide
- the inorganic bases include but are not limited to sodium hydride, potassium phosphate, sodium carbonate, sodium acetate, potassium carbonate, potassium acetate, cesium carbonate , Sodium hydroxide and lithium hydroxide.
- Reagents that provide acidic conditions include, but are not limited to, pyridine hydrobromide, hydrobromic acid, acetic acid, hydrochloric acid, nitric acid, phosphoric acid, p-toluenesulfonic acid, trifluoroacetic acid, and sulfuric acid, preferably pyridine hydrobromide.
- the above reaction is preferably carried out in a solvent.
- the solvents used include but are not limited to: acetic acid, trifluoroacetic acid, methanol, ethanol, acetonitrile, pyridine, toluene, tetrahydrofuran, dichloromethane, petroleum ether, ethyl acetate, n-hexane, dimethyl Sulfoxide, 1,4-dioxane, water, N,N-dimethylformamide and mixtures thereof.
- the compound represented by the general formula (II) of the present disclosure or its tautomer, meso, racemate, enantiomer, diastereomer, or mixture thereof, or
- the preparation method of medicinal salt includes the following steps:
- R a is an alkyl group, preferably a methyl group
- the compound of general formula (II-1) and the compound of general formula (I-2) react under basic conditions in the presence of a chlorinating reagent to obtain a compound of general formula (IIA);
- the compound of general formula (IIA) is reacted under acidic conditions to obtain the compound of general formula (II).
- the chlorinating reagent includes, but is not limited to, phosphorus oxychloride, phosphorus trichloride, thionyl chloride and phosphorus pentachloride, preferably phosphorus oxychloride.
- the reagents that provide alkaline conditions include organic bases and inorganic bases.
- the organic bases include but are not limited to pyridine, hexahydropyridine, triethylamine, N,N-diisopropylethylamine, n-butyllithium, Lithium diisopropylamide, potassium acetate, sodium tert-butoxide and potassium tert-butoxide
- the inorganic bases include but are not limited to sodium hydride, potassium phosphate, sodium carbonate, sodium acetate, potassium carbonate, potassium acetate, cesium carbonate , Sodium hydroxide and lithium hydroxide.
- Reagents that provide acidic conditions include, but are not limited to, pyridine hydrobromide, hydrobromic acid, acetic acid, hydrochloric acid, nitric acid, phosphoric acid, p-toluenesulfonic acid, trifluoroacetic acid, and sulfuric acid, preferably pyridine hydrobromide.
- the above reaction is preferably carried out in a solvent.
- the solvents used include but are not limited to: acetic acid, trifluoroacetic acid, methanol, ethanol, acetonitrile, pyridine, toluene, tetrahydrofuran, dichloromethane, petroleum ether, ethyl acetate, n-hexane, dimethyl Sulfoxide, 1,4-dioxane, water, N,N-dimethylformamide and mixtures thereof.
- the compound represented by the general formula (III) of the present disclosure or its tautomer, meso, racemate, enantiomer, diastereomer, or mixture form thereof, or
- the preparation method of medicinal salt includes the following steps:
- R a is an alkyl group, preferably a methyl group
- a compound of general formula (III-1) and a compound of general formula (I-2) react under basic conditions in the presence of a chlorinating reagent to obtain a compound of general formula (IIIA);
- the compound of general formula (IIIA) is reacted under acidic conditions to obtain the compound of general formula (III).
- the chlorinating reagent includes, but is not limited to, phosphorus oxychloride, phosphorus trichloride, thionyl chloride and phosphorus pentachloride, preferably phosphorus oxychloride.
- the reagents that provide alkaline conditions include organic bases and inorganic bases.
- the organic bases include but are not limited to pyridine, hexahydropyridine, triethylamine, N,N-diisopropylethylamine, n-butyllithium, Lithium diisopropylamide, potassium acetate, sodium tert-butoxide and potassium tert-butoxide
- the inorganic bases include but are not limited to sodium hydride, potassium phosphate, sodium carbonate, sodium acetate, potassium carbonate, potassium acetate, cesium carbonate , Sodium hydroxide and lithium hydroxide.
- Reagents that provide acidic conditions include, but are not limited to, pyridine hydrobromide, hydrobromic acid, acetic acid, hydrochloric acid, nitric acid, phosphoric acid, p-toluenesulfonic acid, trifluoroacetic acid, and sulfuric acid, preferably pyridine hydrobromide.
- the above reaction is preferably carried out in a solvent.
- the solvents used include but are not limited to: acetic acid, trifluoroacetic acid, methanol, ethanol, acetonitrile, pyridine, toluene, tetrahydrofuran, dichloromethane, petroleum ether, ethyl acetate, n-hexane, dimethyl Sulfoxide, 1,4-dioxane, water, N,N-dimethylformamide and mixtures thereof.
- the compound represented by the general formula (IV) of the present disclosure or its tautomer, meso, racemate, enantiomer, diastereomer, or mixture thereof, or
- the preparation method of medicinal salt includes the following steps:
- R a is an alkyl group, preferably a methyl group
- R 1 , R 4 , R 5 , R 9 , R 11 and t are as defined in the compound of general formula (IV).
- the compound of general formula (IV-1) and the compound of general formula (I-2) react under basic conditions in the presence of a chlorinating reagent to obtain a compound of general formula (IVA);
- the compound of general formula (IVA) is reacted under acidic conditions to obtain the compound of general formula (IV).
- the chlorinating reagent includes, but is not limited to, phosphorus oxychloride, phosphorus trichloride, thionyl chloride and phosphorus pentachloride, preferably phosphorus oxychloride.
- the reagents that provide alkaline conditions include organic bases and inorganic bases.
- the organic bases include but are not limited to pyridine, hexahydropyridine, triethylamine, N,N-diisopropylethylamine, n-butyllithium, Lithium diisopropylamide, potassium acetate, sodium tert-butoxide and potassium tert-butoxide
- the inorganic bases include but are not limited to sodium hydride, potassium phosphate, sodium carbonate, sodium acetate, potassium carbonate, potassium acetate, cesium carbonate , Sodium hydroxide and lithium hydroxide.
- Reagents that provide acidic conditions include, but are not limited to, pyridine hydrobromide, hydrobromic acid, acetic acid, hydrochloric acid, nitric acid, phosphoric acid, p-toluenesulfonic acid, trifluoroacetic acid, and sulfuric acid, preferably pyridine hydrobromide.
- the above reaction is preferably carried out in a solvent.
- the solvents used include but are not limited to: acetic acid, trifluoroacetic acid, methanol, ethanol, acetonitrile, pyridine, toluene, tetrahydrofuran, dichloromethane, petroleum ether, ethyl acetate, n-hexane, dimethyl Sulfoxide, 1,4-dioxane, water, N,N-dimethylformamide and mixtures thereof.
- the preparation method of medicinal salt includes the following steps:
- the compound of general formula (IIGA) and R x -OR p react in the presence of a phase transfer catalyst (preferably tetrabutylammonium iodide) to obtain a compound of general formula (IIG); when R x is a phosphate (preferably tert-butyl phosphate) Ester), it can be further hydrolyzed under weak acid (preferably acetic acid) conditions to obtain the final compound,
- a phase transfer catalyst preferably tetrabutylammonium iodide
- X is halogen; preferably Cl;
- R p is a hydrogen atom or Q + ;
- Q + is a pharmaceutically acceptable monovalent cation, preferably K + ;
- R x , R 1 , R 3 to R 6 , R 9 to R 13 and t are as defined in the compound of general formula (IIG).
- the above reaction is preferably carried out in a solvent.
- the solvents used include but are not limited to: acetic acid, trifluoroacetic acid, methanol, ethanol, acetonitrile, pyridine, toluene, tetrahydrofuran, dichloromethane, petroleum ether, ethyl acetate, n-hexane, dimethyl Sulfoxide, 1,4-dioxane, water, N,N-dimethylformamide and mixtures thereof.
- the compound represented by the general formula (IVG) of the present disclosure or its tautomer, mesosome, racemate, enantiomer, diastereomer or its mixture form, or its
- the preparation method of medicinal salt includes the following steps:
- the compound of general formula (IVGA) and R x -OR p are reacted in the presence of a phase transfer catalyst (preferably tetrabutylammonium iodide) to obtain a compound of general formula (IVG); when R x is a phosphate (preferably tertiary phosphoric acid) Butyl ester), the final compound can be further hydrolyzed under weak acid (preferably acetic acid) conditions,
- a phase transfer catalyst preferably tetrabutylammonium iodide
- X is halogen; preferably Cl;
- R p is a hydrogen atom or Q + ;
- Q + is a pharmaceutically acceptable monovalent cation, preferably K + ;
- R x , R 1 , R 4 , R 5 , R 9 , R 11 and t are as defined in the compound of general formula (IVG).
- the above reaction is preferably carried out in a solvent.
- the solvents used include but are not limited to: acetic acid, trifluoroacetic acid, methanol, ethanol, acetonitrile, pyridine, toluene, tetrahydrofuran, dichloromethane, petroleum ether, ethyl acetate, n-hexane, dimethyl Sulfoxide, 1,4-dioxane, water, N,N-dimethylformamide and mixtures thereof.
- the structure of the compound is determined by nuclear magnetic resonance (NMR) or/and mass spectrometry (MS).
- NMR shift ( ⁇ ) is given in units of 10 -6 (ppm).
- the NMR was measured with Bruker AVANCE-400 nuclear magnetic instrument, and the solvent was deuterated dimethyl sulfoxide (DMSO-d 6 ), deuterated chloroform (CDCl 3 ), deuterated methanol (CD 3 OD), and the internal standard was four Methylsilane (TMS).
- DMSO-d 6 dimethyl sulfoxide
- CDCl 3 deuterated chloroform
- CD 3 OD deuterated methanol
- TMS Methylsilane
- MS uses Agilent 1200/1290 DAD-6110/6120 Quadrupole MS liquid mass spectrometer (manufacturer: Agilent, MS model: 6110/6120 Quadrupole MS).
- HPLC High performance liquid chromatography analysis uses Agilent HPLC1200DAD, Agilent HPLC 1200VWD and Waters HPLC e2695-2489 high pressure liquid chromatograph.
- HPLC preparation uses Waters 2545-2767, Waters 2767-SQ Detecor2, Shimadzu LC-20AP and Gilson GX-281 preparative chromatographs.
- CombiFlash rapid preparation instrument uses Combiflash Rf200 (TELEDYNE ISCO).
- the thin layer chromatography silica gel plate uses Yantai Huanghai HSGF254 or Qingdao GF254 silica gel plate, the size of the silica gel plate used in thin layer chromatography (TLC) is 0.15mm ⁇ 0.2mm, and the size of thin layer chromatography separation and purification products is 0.4mm ⁇ 0.5mm.
- the silica gel column chromatography generally uses Yantai Huanghai silica gel 200-300 mesh silica gel as the carrier.
- the known starting materials of the present disclosure can be synthesized by or according to methods known in the art, or can be purchased from ABCR GmbH & Co. KG, Acros Organics, Aldrich Chemical Company, Accela ChemBio Inc., Darui Chemicals and other companies.
- reaction can all be carried out under an argon atmosphere or a nitrogen atmosphere.
- the argon atmosphere or nitrogen atmosphere means that the reaction flask is connected to an argon or nitrogen balloon with a volume of about 1L.
- the hydrogen atmosphere means that the reaction flask is connected to a hydrogen balloon with a volume of about 1L.
- the pressure hydrogenation reaction uses Parr 3916EKX hydrogenator and Qinglan QL-500 hydrogen generator or HC2-SS hydrogenator.
- the hydrogenation reaction is usually evacuated, filled with hydrogen, and repeated three times.
- the microwave reaction uses CEM Discover-S 908860 type microwave reactor.
- the solution refers to an aqueous solution.
- reaction temperature is room temperature, which is 20°C to 30°C.
- the monitoring of the reaction progress in the examples adopts thin-layer chromatography (TLC), the developing reagent used in the reaction, the eluent system of column chromatography used in the purification of the compound, and the developing reagent system of thin-layer chromatography include: A: Dichloromethane/methanol system, B: n-hexane/ethyl acetate system, C: petroleum ether/ethyl acetate system, the volume ratio of the solvent is adjusted according to the polarity of the compound, and a small amount of triethylamine and Adjust with alkaline or acidic reagents such as acetic acid.
- TLC thin-layer chromatography
- the polished magnesium bar (760mg, 31.7mmol, medicine) was cut into small pieces and added to tetrahydrofuran (80mL), protected by argon.
- drop in trimethylchlorosilane (345mg, 3.17mmol, Shaoyuan Technology (Shanghai) Co., Ltd.).
- 1-bromo-4-fluoro-2-methylbenzene 1a (1.5g, 7.9mmol, Shaoyuan Technology (Shanghai) Co., Ltd.)
- compound 1a 4.5g, 23.7mmol, Shaoyuan Technology (Shanghai) Co., Ltd.
- the reaction was heated at 45°C for 1 hour, all the magnesium bars disappeared and a gray homogeneous liquid was formed to obtain the title compound 1b solution (0.4M, 80 mL).
- the product was directly used in the next reaction without purification.
- Tetrahydrofuran (100 mL) and lithium hexamethyldisilazide (1M, 120 mL, 120 mmol, Titan Chemical) were cooled to -78°C under argon protection.
- 4-bromo-2-chloro-1-(trifluoromethyl)benzene 1c 25g, 96.36mmol, Shaoyuan Technology (Shanghai) Co., Ltd.
- N,N-dimethylformamide (14.1g, 192.9mmol, Bailingwei Technology Co., Ltd.
- Water was added and extracted with ethyl acetate (50 mL ⁇ 3).
- Dissolve compound 1e (4g, 10.1mmol) in dichloromethane (50mL), cool to 0°C, add trifluoroacetic acid (10mL, Titan Chemical), dropwise add triethylsilane (6mL, Shaoyuan Technology (Shanghai) Co., Ltd. ), react at 0°C for 1 hour. The reaction was quenched by adding water, and extracted with ethyl acetate (10 mL ⁇ 3). The organic phase was dried with anhydrous sodium sulfate. After filtration, the filtrate was concentrated under reduced pressure, and purified by silica gel column chromatography with the developing solvent system B to obtain the title compound 1f (3.2 g) with a yield of 83%.
- Dissolve compound 1i (1.4g, 3.1mmol) in N,N-dimethylformamide, add pyridine hydrobromide (2.5g, 15.7mmol, Shaoyuan Technology (Shanghai) Co., Ltd.), and raise the temperature to 100°C. React for 1.5 hours. Cooled, filtered, and the filtrate was purified by high performance liquid chromatography (Waters 2767-SQ Detecor 2, elution system: ammonium bicarbonate, water, acetonitrile) to obtain the title compound 1 (1 g) with a yield of 74%.
- Dissolve compound 2c (130mg, 0.26mmol) in toluene (5mL), add tetrakistriphenylphosphine palladium (32mg, 0.03mmol, Bailingwei Technology Co., Ltd.), potassium fluoride (60mg, 1.03mmol, Sinopharm Chemical Reagent Co., Ltd. ), potassium bromide (44mg, 0.37mmol, Sinopharm Chemical Reagent Co., Ltd.) and cyclopropylboronic acid (44mg, 0.51mmol, Shaoyuan Technology (Shanghai) Co., Ltd.), filled with argon gas, and placed in a microwave reactor , React at 100°C for 1 hour. Concentrate under reduced pressure to obtain the crude title compound 2d (120 mg). The product was directly used in the next reaction without purification.
- Test Example 1 Determination of the inhibitory activity of the compound of the present disclosure on Nav1.8
- the purpose of the experiment is to investigate the effect of the compound on the Na V 1.8 ion channel in an in vitro experiment, and the Na V 1.8 ion channel is stably expressed on HEK293 cells. After the Na V 1.8 current is stable, comparing the Na V 1.8 current before and after the compound application, the effect of the compound on the Na V 1.8 ion channel can be obtained.
- Patch clamp amplifier patch clamp PC-505B (WARNER instruments)/MultiClamp 700A (Axon instrument)
- Tetrodotoxin AF3014 (Affix Scientific)
- the extracellular fluid is: NaCl, 137; KCl, 4; CaCl 2 , 1.8; MgCl 2, 1; HEPES, 10; glucose 10; pH 7.4 (NaOH titration).
- Intracellular fluid is aspartic acid, 140; MgCl2, 2; EGTA 11; HEPES, 10; pH 7.2 (CsOH titration). All test compound and control compound solutions contained 1 ⁇ M TTX.
- the storage concentration of the test compound is 9 mM, dissolved in dimethyl sulfoxide (DMSO). Re-dissolve in the extracellular fluid on the day of the test and prepare the required concentration.
- DMSO dimethyl sulfoxide
- the data will be stored in the computer system for analysis. Data collection and analysis will use pCLAMP 10 (Molecular Devices, Union City, CA), and management personnel will review the analysis results.
- Current stability means that the current changes within a limited range over time. The magnitude of the current stabilized is used to calculate the effect of the compound's solubility.
- the inhibitory activity of the compound of the present disclosure on Nav1.8 was determined by the above test, and the measured IC 50 value is shown in Table 1.
- Table 1 The IC 50 of the compounds of the present disclosure for inhibition of Nav1.8 channel activity
- the LC/MS/MS method was used to determine the concentration of the drug in plasma at different times after the rats were intragastrically administered the compound of Example 3 and the compound of Example 7. To study the pharmacokinetic behavior of the compound of the present disclosure in rats and evaluate its pharmacokinetic characteristics.
- Example 3 compound and Example 7 compound Example 3 compound and Example 7 compound.
- SD rats were fasted overnight and then administered by gavage.
- the dose was 2.0 mg/kg and the volume was 10.0 mL/kg.
- Rats were given the compound of Example 3 and Example 7 by intragastric administration. 0.2 mL of blood was collected from the orbit before and 0.5, 1.0, 2.0, 4.0, 6.0, 8.0, 11.0, 24.0 hours after the administration, and placed in heparinized In the test tube, the plasma was separated by centrifugation at 3500 rpm for 10 minutes at 4°C, stored at -20°C, and food was taken 2 hours after administration.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Rheumatology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pain & Pain Management (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本公开涉及2-氧代-1,2-二氢吡啶类衍生物、其制备方法及其在医药上的应用。特别地,本公开涉及通式(IG)所示的2-氧代-1,2-二氢吡啶类衍生物或其前药、其制备方法及含有该衍生物或其前药的药物组合物,以及其作为NaV抑制剂的用途和其在制备治疗和/或预防疼痛和疼痛相关疾病的药物中的用途。其中通式(IG)的各取代基与说明书中的定义相同。
Description
本公开属于医药领域,涉及一种2-氧代-1,2-二氢吡啶类衍生物、其制备方法及其在医药上的应用。特别地,本公开涉及通式(I)所示的2-氧代-1,2-二氢吡啶类衍生物、其制备方法及含有该衍生物的药物组合物,以及其作为Na
V抑制剂的用途和其在制备治疗和/或减轻疼痛和疼痛相关疾病的药物中的用途。
疼痛是一种复杂的生理心理活动,是临床上最常见的症状之一。国际疼痛研究协会将疼痛定义为“一种令人不快的感觉和情绪上的感受,伴有实质上的或潜在的组织损伤,它是一种主观感受”。疼痛可以作为一种警戒信号,提醒机体注意潜在的危险,对机体正常的生命活动具有不可或缺的保护作用。同时,疼痛也是一种常见的临床症状,在引发疼痛的外界刺激消失后,强烈或持久的疼痛会造成生理功能的紊乱,严重影响生命体的生活质量。统计数据显示,全世界约五分之一的人患有中度至重度慢性疼痛。
疼痛起源于周围神经系统的伤害感受器。这是一种游离的神经末梢,广泛分布于全身的皮肤、肌肉、关节和内脏组织中,它可以将感受到的热的、机械的或化学的刺激转化为神经冲动(动作电位)并经由传入神经纤维传递到其位于背根神经节(dorsal root ganglia,DRG)的胞体部分,最终传递到高级神经中枢,引起痛觉。而神经元中动作电位的产生和传导又依赖于细胞膜上的电压门控钠离子通道(voltage-gated sodium channels,Na
V)。当细胞膜去极化时,钠离子通道激活,通道打开,引起钠离子内流,使细胞膜进一步去极化,导致动作电位的产生。因此,抑制异常的钠离子通道活动有助于疼痛的治疗、缓解。
Na
V是一类跨膜离子通道蛋白。这些蛋白由分子量260kD的α亚基和分子量为30-40kD的β亚基组成。根据α亚基的不同可以分为9种亚型,Na
Vl.l~Na
V1.9。不同亚型表现出不同的组织分布和电生理、药理学特征(Rush A.M.,et al.J.Physiol.2007,579,1–14)。根据能否被纳摩尔河豚毒素(tetrodotoxin,TTX)有效抑制,钠离子通道被分为TTX敏感型(TTX-S)和TTX不敏感型(TTX-R)。其中,Na
V1.1、Na
V1.2、Na
V1.3和Na
V1.7为TTX-S型,编码基因位于人类染色体2q23-24,它们在神经元中大量表达。Na
V1.5、Na
V1.8和Na
V1.9为TTX-R型,编码基因位于人类染色体3p21-24。其中,Na
V1.5主要存在于心肌细胞中,Na
V1.8、Na
Vl.9存在于外周神经系统(GoldinA.L.,et al.Annu.Rev.Physiol.2001,63,871–894)。Na
V1.4 和Na
V1.6都为TTX-S型,分别在骨骼肌和中枢神经系统中大量存在(Fozzard H.A.,et al.Physiol.Rev.1996,76,887–926)。局部麻醉药利多卡因通过抑制Na
V来止痛。而非选择性的Na
V抑制剂,例如拉莫三嗪、拉科酰胺、美西律已经成功地用于治疗慢性疼痛。
Na
V1.8为TTX-R型,编码基因为SCN10A,主要存在于三叉神经节神经元和DRG神经元中,具有慢速失活、迅速恢复的电生理特征(Dib-Hajj S.D.,et al.Annu.Rev.Neurosci.2010,33,325–347)。在表达Na
V1.8的神经元内,动作电位的上升主要由Na
V1.8电流构成。在研究神经性疼痛的一些模型中,神经损伤会使Na
V1.8在轴突和神经元胞体中的表达水平上升(Sleeper A.A.,et al.J.Neurosci.2000,20,7279–7289)。使用Na
V1.8反义寡核苷酸在降低Na
V1.8表达的同时可以明显地缓解疼痛(Yoshimura N.,et al.J.Neurosci.2001,21,8690-8696)。大鼠爪内注射角叉菜胶(carrageenan)后,DRG神经元中Na
V1.8的表达有所上升(Tanaka M.,et al.G.NeuroReport 1998,9,967–972)。Na
V1.8敲除小鼠不能表现出正常的内脏炎症痛(Kerr B.J.,et al.NeuroReport2001,12,3077–3080)。人类的Na
V1.8基因产生功能增益突变后,会导致外周神经痛(Faber C.G.,et al.Proc.Natl.Acad.Sci.USA 2012,109,19444–19449)。根据一系列动物实验以及人类基因证据,选择性抑制Na
V1.8具有成为新型镇痛疗法的潜力,可以用于炎性疼痛、神经疼痛、手术后疼痛、癌痛等多种疼痛类型的治疗。
临床中使用的Na
V抑制剂由于缺乏亚型选择性,能够抑制表达在心脏和中枢神经系统中的钠离子通道,因此治疗窗口较窄,应用范围受到限制。Na
V1.8主要分布在外周神经系统,所以选择性地抑制Na
V1.8可以有效地减少副作用。因此,有必要开发活性更高,选择性更好,药代动力学性质更佳,副作用更少的Na
V1.8抑制剂。
发明内容
本公开的目的在于提供一种通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
R
0为氢原子或-CH
2O-R
x;
M为CR
7R
8或S原子;
环A为芳基或杂芳基;
R
1选自氢原子、卤素、烷基、烷氧基、卤代烷基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;
R
2选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;
R
3、R
4、R
5和R
6相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;
R
7和R
8相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;
或者,所述R
7和R
8与相连接的碳原子一起形成羰基、环烷基和杂环基,其中所述的环烷基和杂环基任选被选自烷基、烷氧基、氧代基、卤素、氨基、氰基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;
R
x选自氢原子、-S(O)
2OH、-S(O)
2O
-Q
+、-PO(OR
y)
2、-PO(OH)O
-Q
+、-PO(O
-)
22Q
+和-PO(O
-)
2W
2+;Q
+为药学上可接受的单价阳离子;W
2+为药学上可接受的二价阳离子;
R
y为氢原子或烷基;
n为0、1、2、3、4或5;且
t为0、1或2。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其为通式(I)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
M为CR
7R
8或S原子;
环A为芳基或杂芳基;
R
1选自氢原子、卤素、烷基、烷氧基、卤代烷基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;
R
2选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;
R
3、R
4、R
5和R
6相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;
R
7和R
8相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;
或者,所述R
7和R
8与相连接的碳原子一起形成羰基、环烷基和杂环基,其中所述的环烷基和杂环基任选被选自烷基、烷氧基、氧代基、卤素、氨基、氰基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;
n为0、1、2、3、4或5;且
t为0、1或2。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R
2选自氢原子、卤素、烷基、烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中环A为苯基或吡啶基。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,进一步为通式(II)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
R
9、R
10、R
11、R
12和R
13相同或不同,且各自独立地选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;且
M、R
1、R
3~R
6和t如通式(I)化合物中所定义。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,进一步为通式(IIG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
R
9、R
10、R
11、R
12和R
13相同或不同,且各自独立地选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;且
R
x、M、R
1、R
3~R
6和t如通式(IG)中所定义。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R
3、R
4、R
5和R
6相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基和卤代烷氧基,优选氢原子、卤素和卤代烷基。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R
3和R
6为氢原子。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R
9、R
10、R
11、R
12和R
13相同或不同,且各自独立地选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、环烷基、卤代烷基和卤代烷氧基,优选氢原子、卤素和烷基。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R
9、R
10、R
11、R
12和R
13相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基和卤代烷氧基,优选氢原子、卤素和烷基。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,进一步为通式(III)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
M、R
1、R
4、R
5、R
9、R
11和t如通式(II)化合物中所定义。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中M为CR
7R
8;R
7和R
8如通式(IG)中所定义。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,进一步为通式(IV)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
R
1、R
4、R
5、R
9、R
11和t如通式(II)化合物中所定义。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,进一步为通式(IVG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
R
x、R
1、R
4、R
5、R
9、R
11和t如通式(IG)化合物中所定义。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R
1为氢原子。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R
4为卤素,优选为氯;且R
5为卤代烷基,优选为三氟甲基。
在本公开一些实施方案中,所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R
x为-PO(OH)
2。
通式(I)的典型化合物,包括但不限于:
或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐。
本公开另一方面提供一种通式(IA)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
R
a为烷基,优选为甲基;
M、环A、R
1~R
6、n和t如通式(I)化合物中所定义。通式(IA)化合物为制备通式(I)化合物的中间体。
通式(IA)的典型化合物,包括但不限于:
或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐。
本公开另一方面提供一种通式(IIGA)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
X为卤素;优选为Cl;且
M、R
1、R
3~R
6、R
9~R
13和t如通式(IIG)化合物中所定义。通式(IIGA)化合物为制备通式(IIG)化合物的中间体。
本公开另一方面提供一种通式(IVGA)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,
其中:
X为卤素;优选为Cl;
R
1、R
4、R
5、R
9、R
11和t如通式(IVG)化合物中所定义。通式(IVGA)化合物为制备通式(IVG)化合物的中间体。
通式(IIGA)或通式(IVGA)的典型化合物,包括但不限于:
或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐。
本公开的另一方面涉及一种制备通式(I)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐的方法,该方法包括:
由通式(IA)化合物反应得到通式(I)化合物;
其中:
R
a为烷基,优选为甲基;且
M、环A、R
1~R
6、n和t如通式(I)化合物中所定义。
本公开的另一方面涉及一种制备通式(II)所示的化合物的或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐方法,该方法包括:
由通式(IIA)化合物反应得到通式(II)化合物;
其中:
R
a为烷基,优选为甲基;且
M、R
1、R
3~R
6、R
9~R
13和t如通式(II)化合物中所定义。
本公开的另一方面涉及一种制备通式(IIG)所示的化合物的或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐方法,该方法包括:
通式(IIGA)化合物与R
x-OR
p反应得到通式(IIG)化合物;
其中:
X为卤素;优选为Cl;
R
p为氢原子或Q
+;Q
+为药学上可接受的单价阳离子,优选为K
+;且
M、R
x、R
1、R
3~R
6、R
9~R
13和t如通式(IIG)化合物中所定义。
本公开的另一方面涉及一种制备通式(III)所示的化合物的或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐方法,该方法包括:
由通式(IIIA)化合物反应得到通式(III)化合物;
其中:
R
a为烷基,优选为甲基;且
M、R
1、R
4、R
5、R
9、R
11和t如通式(III)化合物中所定义。
本公开的另一方面涉及一种制备通式(IV)所示的化合物的或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐方法,该方法包括:
由通式(IVA)化合物反应得到通式(IV)化合物;
其中:
R
a为烷基,优选为甲基;
R
1、R
4、R
5、R
9、R
11和t如通式(IV)化合物中所定义。
本公开的另一方面涉及一种制备通式(IVG)所示的化合物的或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐方法,该方法包括:
通式(IVGA)化合物与R
x-OR
p反应得到通式(IVG)化合物;
其中:
X为卤素;优选为Cl;
R
p为氢原子或Q
+;Q
+为药学上可接受的单价阳离子,优选为K
+;且
R
x、R
1、R
4、R
5、R
9、R
11和t如通式(IVG)化合物中所定义。
本公开的另一方面涉及一种药物组合物,其含有如上所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体、或其混合物形式、或可药用的盐,以及一种或多种药学上可接受的载体、稀释剂或赋形剂。
本公开还涉及一种制备上述药物组合物的方法,其包括将如上所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体、或其混合物形式、或其可药用的盐与药学上可接受的载体、稀释剂或赋形剂相混合。
本公开还涉及一种如上所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体、或其混合物形式、或其可药用的盐,或如上所述的药物组合物在制备抑制受试者电压门控钠离子通道的药物中的用途。其中所述的电压门控钠离子通道优选为Na
V1.8。
本公开还涉及一种如上所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体、或其混合物形式、或其可药用的盐,或如上所述的药物组合物在制备用于治疗和/或减轻疼痛和疼痛相关疾病、多发性硬化症、夏-马-图三氏综合症、失禁或心律失常的药物中的用途。所述的疼痛优选选自慢性疼痛、急性疼痛、炎性疼痛、癌症疼痛、神经性疼痛、肌肉骨骼痛、手术后疼痛、内脏痛、肠痛和特发性疼痛。
本公开还涉及一种抑制受试者电压门控钠离子通道的方法,该方法包括向需要其的患者施用本公开如上所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体、或其混合物形式、或其可药用的盐,或如上所述的药物组合物。所述的电压门控钠离子通道优选为Na
V1.8。
本公开还涉及一种治疗和/或减轻疼痛和疼痛相关疾病、多发性硬化症、夏-马-图三氏综合症、失禁或心律失常的药物的方法,该方法包括向需要其的患者施用本公开如上所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体、或其混合物形式、或其可药用的盐,或如上所述的药物组合物。所述的疼痛优选选自慢性疼痛、急性疼痛、炎性疼痛、癌症疼痛、神经性疼痛、肌肉骨骼痛、手术后疼痛、内脏痛、肠痛和特发性疼痛。
本公开还涉及一种通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐,或如上所述的药物组合物,其用作药物。
本公开还涉及一种通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐,或如上所述的药物组合物,其用作抑制受试者电压门控钠通道的药物。所述的电压门控钠离子通道优选为Na
V1.8。
本公开还涉及一种通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐,或如上所述的药物组合物,其用于治疗和/或减轻疼痛和疼痛相关疾病、多发性硬化症、夏-马-图三氏综合症、失禁或心律失常。其中所述的疼痛优选选自慢性疼痛、急性疼痛、炎性疼痛、癌症疼痛、神经性疼痛、肌肉骨骼痛、手术后疼痛、内脏痛、肠痛和特发性疼痛。
本公开中所述的神经性疼痛优选选自三叉神经痛、疱疹后神经痛、糖尿病性神经痛、痛性HIV相关性感觉神经痛、灼伤综合症、截肢术后疼痛、脊髓损伤后疼痛、幻痛、痛性神经瘤、创伤性神经瘤、莫顿(Morton)神经瘤、神经挤压损伤、脊管狭窄、腕管综合症、神经根痛、坐骨神经痛、神经撕脱伤、臂丛撕脱伤、复杂性区域疼痛综合征、药物疗法引起的神经痛、癌症化学疗法引起的神经痛、抗逆转录病毒疗法引起的神经痛、特发性小纤维神经病变、特发性感觉神经痛和三叉自主神经性头痛。
本公开中所述的肌肉骨骼痛优选选自骨关节炎疼痛、背痛、冷痛、灼烧疼痛和牙痛。
本公开中所述的肠痛优选选自发炎性肠病疼痛、克罗恩病疼痛和间质性膀胱炎疼痛。
本公开中所述的炎性疼痛优选选自类风湿性关节炎疼痛和外阴痛。
本公开中所述的特发性疼痛优选选自纤维肌痛。
本公开治疗方法中所用化合物或组合物的剂量通常将随疾病的严重性、患者的体重和化合物的相对功效而改变。不过,作为一般性指导,合适的单位剂量可以是0.1~1000mg。
本公开的药物组合物除活性化合物外,可含有一种或多种辅料,所述辅料选自以下成分:填充剂(稀释剂)、粘合剂、润湿剂、崩解剂或赋形剂等。根据给药方法的不同,组合物可含有0.1至99重量%的活性化合物。
含活性成分的药物组合物可以是适用于口服的形式,例如片剂、糖锭剂、锭剂、水或油混悬液、可分散粉末或颗粒、乳液、硬或软胶囊,或糖浆剂或酏剂。可按照本领域任何已知制备药用组合物的方法制备口服组合物,此类组合物可含有一种或多种选自以下的成分:甜味剂、矫味剂、着色剂和防腐剂,以提供悦目和可口的药用制剂。片剂含有活性成分和用于混合的适宜制备片剂的无毒的可药用的赋形剂。这些赋形剂可以是惰性赋形剂,造粒剂和崩解剂和润滑剂。这些片 剂可以不包衣或可通过掩盖药物的味道或在胃肠道中延迟崩解和吸收,因而在较长时间内提供缓释作用的已知技术将其包衣。
也可用其中活性成分与惰性固体稀释剂或其中活性成分与水溶性载体或油溶媒或橄榄油混合的软明胶胶囊提供口服制剂。
水悬浮液含有活性物质和用于混合的适宜制备水悬浮液的赋形剂。此类赋形剂是悬浮剂,分散剂或湿润剂。水混悬液也可以含有一种或多种防腐剂例如尼泊金乙酯或尼泊金正丙酯、一种或多种着色剂、一种或多种矫味剂和一种或多种甜味剂。
油混悬液可通过使活性成分悬浮于植物油或矿物油中配制而成。油悬浮液可含有增稠剂。可加入上述的甜味剂和矫味剂,以提供可口的制剂。可通过加入抗氧化剂保存这些组合物。
通过加入水可使适用于制备水混悬液的可分散粉末和颗粒提供活性成分和用于混合的分散剂或湿润剂、悬浮剂或一种或多种防腐剂。适宜的分散剂或湿润剂和悬浮剂可说明上述的例子。也可加入其他赋形剂例如甜味剂、矫味剂和着色剂。
本公开的药物组合物也可以是水包油乳剂的形式。油相可以是植物油或矿物油例如液体石蜡或其混合物。适宜的乳化剂可以是天然产生的磷脂或偏酯。乳剂也可以含有甜味剂、矫味剂、防腐剂和抗氧剂。
本公开的药物组合物可以是无菌注射水溶液形式。可以使用的可接受的溶媒或溶剂有水、林格氏液和等渗氯化钠溶液。无菌注射制剂可以是其中活性成分溶于油相的无菌注射水包油微乳。可通过局部大量注射,将注射液或微乳注入患者的血流中。
本公开的药物组合物可以是用于肌内和皮下给药的无菌注射水或油混悬液的形式。可按已知技术,用上述那些适宜的分散剂或湿润剂和悬浮剂配制该混悬液。无菌注射制剂也可以是在肠胃外可接受的无毒稀释剂或溶剂中制备的无菌注射溶液或混悬液。此外,可方便地用无菌固定油作为溶剂或悬浮介质。
可按用于直肠给药的栓剂形式给予本公开化合物。可通过将药物与在普通温度下为固体但在直肠中为液体,因而在直肠中会溶化而释放药物的适宜的无刺激性赋形剂混合来制备这些药物组合物。
如本领域技术人员所熟知的,药物的给药剂量依赖于多种因素,包括但并非限定于以下因素:所用具体化合物的活性、患者的年龄、患者的体重、患者的健康状况、患者的行为、患者的饮食、给药时间、给药方式、排泄的速率、药物的组合等;另外,最佳的治疗方式如治疗的模式、通式化合物(I)的日用量或可药用的盐的种类可以根据传统的治疗方案来验证。
本申请提供了一种如通式(IG)所示的新型结构的Na
V抑制剂或其前药,其对Nav1.8通道活性具有明显的抑制效果,特别是通式(IG)中R
4为卤素,R
5为卤代烷基时,整个分子的生物活性有显著提高。
术语定义
除非有相反陈述,在说明书和权利要求书中使用的术语具有下述含义。
术语“烷基”指饱和脂肪族烃基团,其为包含1至20个碳原子的直链或支链基团,优选含有1至12个碳原子的烷基,更优选含有1至6个碳原子的烷基。非限制性实例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、仲丁基、正戊基、1,1-二甲基丙基、1,2-二甲基丙基、2,2-二甲基丙基、1-乙基丙基、2-甲基丁基、3-甲基丁基、正己基、1-乙基-2-甲基丙基、1,1,2-三甲基丙基、1,1-二甲基丁基、1,2-二甲基丁基、2,2-二甲基丁基、1,3-二甲基丁基、2-乙基丁基、2-甲基戊基、3-甲基戊基、4-甲基戊基、2,3-二甲基丁基、正庚基、2-甲基己基、3-甲基己基、4-甲基己基、5-甲基己基、2,3-二甲基戊基、2,4-二甲基戊基、2,2-二甲基戊基、3,3-二甲基戊基、2-乙基戊基、3-乙基戊基、正辛基、2,3-二甲基己基、2,4-二甲基己基、2,5-二甲基己基、2,2-二甲基己基、3,3-二甲基己基、4,4-二甲基己基、2-乙基己基、3-乙基己基、4-乙基己基、2-甲基-2-乙基戊基、2-甲基-3-乙基戊基、正壬基、2-甲基-2-乙基己基、2-甲基-3-乙基己基、2,2-二乙基戊基、正癸基、3,3-二乙基己基、2,2-二乙基己基,及其各种支链异构体等。更优选的是含有1至6个碳原子的低级烷基,非限制性实施例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、仲丁基、正戊基、1,1-二甲基丙基、1,2-二甲基丙基、2,2-二甲基丙基、1-乙基丙基、2-甲基丁基、3-甲基丁基、正己基、1-乙基-2-甲基丙基、1,1,2-三甲基丙基、1,1-二甲基丁基、1,2-二甲基丁基、2,2-二甲基丁基、1,3-二甲基丁基、2-乙基丁基、2-甲基戊基、3-甲基戊基、4-甲基戊基、2,3-二甲基丁基等。烷基可以是取代的或非取代的,当被取代时,取代基可以在任何可使用的连接点上被取代,所述取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基、氧代基、羧基或羧酸酯基。
术语“烷氧基”指-O-(烷基)和-O-(非取代的环烷基),其中烷基如上所定义。烷氧基的非限制性实例包括:甲氧基、乙氧基、丙氧基、丁氧基、环丙氧基、环丁氧基、环戊氧基、环己氧基。烷氧基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基、羧基或羧酸酯基。
术语“环烷基”指饱和或部分不饱和单环或多环环状烃取代基,环烷基环包含3至20个碳原子,优选包含3至12个碳原子,更优选包含3至6个碳原子(例如3、4、5或6个碳原子),最优选包含5至6个碳原子。单环环烷基的非限制性 实例包括环丙基、环丁基、环戊基、环戊烯基、环己基、环己烯基、环己二烯基、环庚基、环庚三烯基、环辛基等;多环环烷基包括螺环、稠环和桥环的环烷基。
术语“螺环烷基”指5至20元的单环之间共用一个碳原子(称螺原子)的多环基团,其可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统。优选为6至14元,更优选为7至10元(例如7、8、9或10元)。根据环与环之间共用螺原子的数目将螺环烷基分为单螺环烷基、双螺环烷基或多螺环烷基,优选为单螺环烷基和双螺环烷基。更优选为4元/4元、4元/5元、4元/6元、5元/5元或5元/6元单螺环烷基。螺环烷基的非限制性实例包括:
术语“稠环烷基”指5至20元,系统中的每个环与体系中的其他环共享毗邻的一对碳原子的全碳多环基团,其中一个或多个环可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环稠环烷基,优选为双环或三环,更优选为5元/5元或5元/6元双环烷基。稠环烷基的非限制性实例包括:
术语“桥环烷基”指5至20元,任意两个环共用两个不直接连接的碳原子的全碳多环基团,其可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环桥环烷基,优选为双环、三环或四环,更优选为双环或三环。桥环烷基的非限制性实例包括:
所述环烷基环包括如上所述的环烷基(包括单环、螺环、稠环和桥环)稠合于芳基、杂芳基或杂环烷基环上,其中与母体结构连接在一起的环为环烷基,非限制性实例包括茚满基、四氢萘基、苯并环庚烷基等;优选苯基并环戊基、四氢萘基。环烷基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以 下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基、氧代基、羧基或羧酸酯基。
术语“杂环基”指饱和或部分不饱和单环或多环环状烃取代基,其包含3至20个环原子,其中一个或多个环原子为选自氮、氧或S(O)
m(其中m是整数0至2)的杂原子,但不包括-O-O-、-O-S-或-S-S-的环部分,其余环原子为碳。优选包含3至12个环原子,其中1~4个是杂原子;最优选包含3至8个环原子,其中1~3个是杂原子;最优选包含5至6个环原子,其中1~2或1~3个是杂原子。单环杂环基的非限制性实例包括吡咯烷基、咪唑烷基、四氢呋喃基、四氢吡喃基、四氢噻吩基、二氢咪唑基、二氢呋喃基、二氢吡唑基、二氢吡咯基、哌啶基、哌嗪基、吗啉基、硫代吗啉基、高哌嗪基等,优选四氢吡喃基、哌啶基、吡咯烷基。多环杂环基包括螺环、稠环和桥环的杂环基。
术语“螺杂环基”指5至20元的单环之间共用一个原子(称螺原子)的多环杂环基团,其中一个或多个环原子为选自氮、氧或S(O)
m(其中m是整数0至2)的杂原子,其余环原子为碳。其可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统。优选为6至14元,更优选为7至10元。根据环与环之间共用螺原子的数目将螺杂环基分为单螺杂环基、双螺杂环基或多螺杂环基,优选为单螺杂环基和双螺杂环基。更优选为4元/4元、4元/5元、4元/6元、5元/5元或5元/6元单螺杂环基。螺杂环基的非限制性实例包括:
术语“稠杂环基”指5至20元,系统中的每个环与体系中的其他环共享毗邻的一对原子的多环杂环基团,一个或多个环可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统,其中一个或多个环原子为选自氮、氧或S(O)
m(其中m是整数0至2)的杂原子,其余环原子为碳。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环稠杂环基,优选为双环或三环,更优选为5元/5元或5元/6元双环稠杂环基。稠杂环基的非限制性实例包括:
术语“桥杂环基”指5至14元,任意两个环共用两个不直接连接的原子的多环杂环基团,其可以含有一个或多个双键,但没有一个环具有完全共轭的π电子系统,其中一个或多个环原子为选自氮、氧或S(O)
m(其中m是整数0至2)的杂原子,其余环原子为碳。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环桥杂环基,优选为双环、三环或四环,更优选为双环或三环。桥杂环基的非限制性实例包括:
所述杂环基环包括如上所述的杂环基(包括单环、螺杂环、稠杂环和桥杂环)稠合于芳基、杂芳基或环烷基环上,其中与母体结构连接在一起的环为杂环基,其非限制性实例包括:
杂环基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基、氧代基、羧基或羧酸酯基。
术语“芳基”指具有共轭的π电子体系的6至14元全碳单环或稠合多环(也就是共享毗邻碳原子对的环)基团,优选为6至10元,例如苯基和萘基。所述芳基环包括如上所述的芳基环稠合于杂芳基、杂环基或环烷基环上,其中与母体结构连接在一起的环为芳基环,其非限制性实例包括:
芳基可以是取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基、羧基或羧酸酯基。
术语“杂芳基”指包含1至4个杂原子、5至14个环原子的杂芳族体系,其中杂原子选自氧、硫和氮。杂芳基优选为5至10元,含1至3个杂原子;更优选为5元或6元,含1至2个杂原子;优选例如咪唑基、呋喃基、噻吩基、噻唑基、吡唑基、噁唑基、吡咯基、四唑基、吡啶基、嘧啶基、噻二唑、吡嗪基和哒嗪基等,优选为哒嗪基和吡啶基;更优选吡啶基。所述杂芳基环包括如上所述的杂芳基环稠合于芳基、杂环基或环烷基环上,其中与母体结构连接在一起的环为杂芳基环,其非限制性实例包括:
杂芳基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、巯基、羟基、氧代基、硝基、氰基、环烷基、杂环烷基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基、羧基或羧酸酯基。
术语“羟烷基”指被羟基取代的烷基,其中烷基如上所定义。
术语“卤代烷基”指烷基被一个或多个卤素取代,其中烷基如上所定义。
术语“氘代烷基”指烷基被一个或多个氘原子取代,其中烷基如上所定义。
术语“卤代烷氧基”指烷氧基被一个或多个卤素取代,其中烷氧基如上所定义。
术语“氘代烷氧基”指烷氧基被一个或多个氘原子取代,其中烷氧基如上所定义。
术语“环烷基烷基”指烷基被一个或多个环烷基取代,其中环烷基和烷基如上所定义。
术语“杂环基烷基”指烷基被一个或多个杂环基取代,其中杂环基和烷基如上所定义。
术语“芳基烷基”指烷基被一个或多个芳基取代,其中芳基和烷基如上所定义。
术语“羟基”指-OH基团。
术语“卤素”指氟、氯、溴或碘。
术语“羰基”指C=O。
术语“氨基”指-NH
2。
术语“氰基”指-CN。
术语“硝基”指-NO
2。
术语“羧基”指-C(O)OH。
术语“羧酸酯基”指-C(O)O(烷基)或-C(O)O(环烷基),其中烷基、环烷基如上所定义。
术语“酰卤”指含有-C(O)-卤素的基团的化合物。
术语“药学上可接受的单价阳离子”(Q
+)包括(例如N(R
y)
4,其中R
y为H或C
1-C
4烷基)、碱金属离子(例如钾、钠及锂离子)、二环己胺离子及N-甲基D-还原葡糖胺离子。
术语“药学上可接受的二价阳离子”(W
2+)包括碱土金属离子,例如钙及镁离子,以及二价铝离子。还包括氨基酸阳离子,例如精氨酸、赖氨酸、鸟氨酸等的单价或二价离子。药学上可接受的二价阳离子(W
2+)可以被两个药学上可接受的单价阳离子(Q
+)替换。
本公开的化合物还可包含其同位素衍生物。术语“同位素衍生物”指结构不同仅在于存在一种或多种同位素富集原子的化合物。例如,具有本公开的结构,除了用“氘”或“氚”代替氢,或者用
18F-氟标记(
18F同位素)代替氟,或者用
11C-,
13C-,或者
14C-富集的碳(
11C-,
13C-,或者
14C-碳标记;
11C-,
13C-,或者
14C-同位素)代替碳原子的化合物处于本公开的范围内。这样的化合物可用作例如生物学测定中的分析工具或探针,或者可以用作疾病的体内诊断成像示踪剂,或者作为药效学、药动学或受体研究的示踪剂。
本公开的化合物还包括各种氘化形式的式(I)化合物。与碳原子连接的各个可用的氢原子可独立地被氘原子替换。本领域技术人员能够参考相关文献合成氘化形式的式(I)化合物。在制备氘代形式的式(I)化合物时可使用市售的氘代起始物质,或它们可使用常规技术采用氘代试剂合成,氘代试剂包括但不限于氘代硼烷、三氘代硼烷四氢呋喃溶液、氘代氢化锂铝、氘代碘乙烷和氘代碘甲烷等。
“任选”或“任选地”意味着随后所描述的事件或环境可以但不必发生,该说明包括该事件或环境发生或不发生的场合。例如,“任选被烷基取代的杂环基团”意味着烷基可以但不必须存在,该说明包括杂环基团被烷基取代的情形和杂环基团不被烷基取代的情形。
“取代的”指基团中的一个或多个氢原子,优选为最多5个,更优选为1~3个氢原子彼此独立地被相应数目的取代基取代。不言而喻,取代基仅处在它们的可能的化学位置,本领域技术人员能够在不付出过多努力的情况下确定(通过实验或理论)可能或不可能的取代。例如,具有游离氢的氨基或羟基与具有不饱和(如烯属)键的碳原子结合时可能是不稳定的。
“药物组合物”表示含有一种或多种本文所述化合物或其生理学上/可药用的盐或前体药物与其他化学组分的混合物,以及其他组分例如生理学/可药用的载体和赋形剂。药物组合物的目的是促进对生物体的给药,利于活性成分的吸收进而发挥生物活性。
“可药用的盐”是指本公开化合物的盐,这类盐用于哺乳动物体内时具有安全性和有效性,且具有应有的生物活性。
本公开提供了一种具有通式(I)结构的新型NaV抑制剂,具有此类结构的化合物对Na
V1.8具有优异的抑制效果。
本公开化合物的合成方法
为了完成本公开的目的,本公开采用如下技术方案:
方案一
本公开通式(I)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐制备方法,包括以下步骤:
其中:
R
a为烷基,优选为甲基;
M、环A、R
1~R
6、n和t如通式(I)化合物中所定义。
第一步,通式(I-1)化合物和通式(I-2)化合物在碱性条件下,在氯化试剂的存在下,发生反应得到通式(IA)化合物;
第二步,通式(IA)化合物在酸性条件下,反应得到通式(I)化合物。
所述氯化试剂包括但不限于三氯氧磷、三氯化磷、二氯亚砜和五氯化磷,优选三氯氧磷。
提供碱性条件的试剂包括有机碱和无机碱类,所述的有机碱类包括但不限于吡啶、六氢吡啶、三乙胺、N,N-二异丙基乙胺、正丁基锂、二异丙基氨基锂、醋酸钾、叔丁醇钠和叔丁醇钾,所述的无机碱类包括但不限于氢化钠、磷酸钾、碳酸钠、醋酸钠、碳酸钾、醋酸钾、碳酸铯、氢氧化钠和氢氧化锂。
提供酸性条件的试剂包括但不限于吡啶氢溴酸盐、氢溴酸、乙酸、盐酸、硝酸、磷酸、对苯甲磺酸、三氟乙酸和硫酸,优选吡啶氢溴酸盐。
上述反应优选在溶剂中进行,所用溶剂包括但不限于:乙酸、三氟乙酸、甲醇、乙醇、乙腈、吡啶、甲苯、四氢呋喃、二氯甲烷、石油醚、乙酸乙酯、正己烷、二甲基亚砜、1,4-二氧六环、水、N,N-二甲基甲酰胺及其混合物。
方案二
本公开通式(II)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐制备方法,包括以下步骤:
其中:
R
a为烷基,优选为甲基;
M、R
1、R
3~R
6、R
9~R
13和t如通式(II)化合物中所定义。
第一步,通式(II-1)化合物和通式(I-2)化合物在碱性条件下,在氯化试剂的存在下,发生反应得到通式(IIA)化合物;
第二步,通式(IIA)化合物在酸性条件下,反应得到通式(II)化合物。
所述氯化试剂包括但不限于三氯氧磷、三氯化磷、二氯亚砜和五氯化磷,优选三氯氧磷。
提供碱性条件的试剂包括有机碱和无机碱类,所述的有机碱类包括但不限于吡啶、六氢吡啶、三乙胺、N,N-二异丙基乙胺、正丁基锂、二异丙基氨基锂、醋酸钾、叔丁醇钠和叔丁醇钾,所述的无机碱类包括但不限于氢化钠、磷酸钾、碳酸钠、醋酸钠、碳酸钾、醋酸钾、碳酸铯、氢氧化钠和氢氧化锂。
提供酸性条件的试剂包括但不限于吡啶氢溴酸盐、氢溴酸、乙酸、盐酸、硝酸、磷酸、对苯甲磺酸、三氟乙酸和硫酸,优选吡啶氢溴酸盐。
上述反应优选在溶剂中进行,所用溶剂包括但不限于:乙酸、三氟乙酸、甲醇、乙醇、乙腈、吡啶、甲苯、四氢呋喃、二氯甲烷、石油醚、乙酸乙酯、正己烷、二甲基亚砜、1,4-二氧六环、水、N,N-二甲基甲酰胺及其混合物。
方案三
本公开通式(III)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐制备方法,包括以下步骤:
其中:
R
a为烷基,优选为甲基;
M、R
1、R
4、R
5、R
9、R
11和t如通式(III)化合物中所定义。
第一步,通式(III-1)化合物和通式(I-2)化合物在碱性条件下,在氯化试剂的存在下,发生反应得到通式(IIIA)化合物;
第二步,通式(IIIA)化合物在酸性条件下,反应得到通式(III)化合物。
所述氯化试剂包括但不限于三氯氧磷、三氯化磷、二氯亚砜和五氯化磷,优选三氯氧磷。
提供碱性条件的试剂包括有机碱和无机碱类,所述的有机碱类包括但不限于吡啶、六氢吡啶、三乙胺、N,N-二异丙基乙胺、正丁基锂、二异丙基氨基锂、醋酸钾、叔丁醇钠和叔丁醇钾,所述的无机碱类包括但不限于氢化钠、磷酸钾、碳酸钠、醋酸钠、碳酸钾、醋酸钾、碳酸铯、氢氧化钠和氢氧化锂。
提供酸性条件的试剂包括但不限于吡啶氢溴酸盐、氢溴酸、乙酸、盐酸、硝酸、磷酸、对苯甲磺酸、三氟乙酸和硫酸,优选吡啶氢溴酸盐。
上述反应优选在溶剂中进行,所用溶剂包括但不限于:乙酸、三氟乙酸、甲醇、乙醇、乙腈、吡啶、甲苯、四氢呋喃、二氯甲烷、石油醚、乙酸乙酯、正己烷、二甲基亚砜、1,4-二氧六环、水、N,N-二甲基甲酰胺及其混合物。
方案四
本公开通式(IV)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐制备方法,包括以下步骤:
其中:
R
a为烷基,优选为甲基;
R
1、R
4、R
5、R
9、R
11和t如通式(IV)化合物中所定义。
第一步,通式(IV-1)化合物和通式(I-2)化合物在碱性条件下,在氯化试剂的存在下,发生反应得到通式(IVA)化合物;
第二步,通式(IVA)化合物在酸性条件下,反应得到通式(IV)化合物。
所述氯化试剂包括但不限于三氯氧磷、三氯化磷、二氯亚砜和五氯化磷,优选三氯氧磷。
提供碱性条件的试剂包括有机碱和无机碱类,所述的有机碱类包括但不限于吡啶、六氢吡啶、三乙胺、N,N-二异丙基乙胺、正丁基锂、二异丙基氨基锂、醋酸钾、叔丁醇钠和叔丁醇钾,所述的无机碱类包括但不限于氢化钠、磷酸钾、碳酸钠、醋酸钠、碳酸钾、醋酸钾、碳酸铯、氢氧化钠和氢氧化锂。
提供酸性条件的试剂包括但不限于吡啶氢溴酸盐、氢溴酸、乙酸、盐酸、硝酸、磷酸、对苯甲磺酸、三氟乙酸和硫酸,优选吡啶氢溴酸盐。
上述反应优选在溶剂中进行,所用溶剂包括但不限于:乙酸、三氟乙酸、甲醇、乙醇、乙腈、吡啶、甲苯、四氢呋喃、二氯甲烷、石油醚、乙酸乙酯、正己烷、二甲基亚砜、1,4-二氧六环、水、N,N-二甲基甲酰胺及其混合物。
方案五
本公开通式(IIG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐制备方法,包括以下步骤:
通式(IIGA)化合物与R
x-OR
p在相转移催化剂(优选为四丁基碘化铵)存在下,反应得到通式(IIG)化合物;当R
x为磷酸酯(优选为磷酸叔丁酯)时,可进一步在弱酸性(优选为乙酸)条件下水解得到最终化合物,
其中:
X为卤素;优选为Cl;
R
p为氢原子或Q
+;Q
+为药学上可接受的单价阳离子,优选为K
+;
M、R
x、R
1、R
3~R
6、R
9~R
13和t如通式(IIG)化合物中所定义。
上述反应优选在溶剂中进行,所用溶剂包括但不限于:乙酸、三氟乙酸、甲醇、乙醇、乙腈、吡啶、甲苯、四氢呋喃、二氯甲烷、石油醚、乙酸乙酯、正己烷、二甲基亚砜、1,4-二氧六环、水、N,N-二甲基甲酰胺及其混合物。
方案六
本公开通式(IVG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式、或其可药用的盐制备方法,包括以下步骤:
通式(IVGA)化合物与R
x-OR
p在在相转移催化剂(优选为四丁基碘化铵)存在下,反应得到通式(IVG)化合物;当R
x为磷酸酯(优选为磷酸叔丁酯)时,可进一步在弱酸性(优选为乙酸)条件下水解得到最终化合物,
其中:
X为卤素;优选为Cl;
R
p为氢原子或Q
+;Q
+为药学上可接受的单价阳离子,优选为K
+;
R
x、R
1、R
4、R
5、R
9、R
11和t如通式(IVG)化合物中所定义。
上述反应优选在溶剂中进行,所用溶剂包括但不限于:乙酸、三氟乙酸、甲醇、乙醇、乙腈、吡啶、甲苯、四氢呋喃、二氯甲烷、石油醚、乙酸乙酯、正己烷、二甲基亚砜、1,4-二氧六环、水、N,N-二甲基甲酰胺及其混合物。
以下结合实施例进一步描述本公开,但这些实施例并非限制着本公开的范围。
实施例
化合物的结构是通过核磁共振(NMR)或/和质谱(MS)来确定的。NMR位移(δ)以10
-6(ppm)的单位给出。NMR的测定是用Bruker AVANCE-400核磁仪,测定溶剂为氘代二甲基亚砜(DMSO-d
6)、氘代氯仿(CDCl
3)、氘代甲醇(CD
3OD),内标为四甲基硅烷(TMS)。
MS的测定用Agilent 1200/1290 DAD-6110/6120 Quadrupole MS液质联用仪(生产商:Agilent,MS型号:6110/6120 Quadrupole MS)。
waters ACQuity UPLC-QD/SQD(生产商:waters,MS型号:waters ACQuity Qda Detector/waters SQ Detector)THERMO Ultimate3000-Q Exactive(生产商:THERMO,MS型号:THERMO Q Exactive)。
高效液相色谱法(HPLC)分析使用Agilent HPLC1200DAD、Agilent HPLC 1200VWD和Waters HPLC e2695-2489高压液相色谱仪。
手性HPLC分析测定使用Agilent 1260 DAD高效液相色谱仪。
高效液相制备使用Waters 2545-2767、Waters2767-SQ Detecor2、Shimadzu LC-20AP和Gilson GX-281制备型色谱仪。
手性制备使用Shimadzu LC-20AP制备型色谱仪。
CombiFlash快速制备仪使用Combiflash Rf200(TELEDYNE ISCO)。
薄层层析硅胶板使用烟台黄海HSGF254或青岛GF254硅胶板,薄层色谱法(TLC)使用的硅胶板采用的规格是0.15mm~0.2mm,薄层层析分离纯化产品采用的规格是0.4mm~0.5mm。
硅胶柱色谱法一般使用烟台黄海硅胶200~300目硅胶为载体。
激酶平均抑制率及IC
50值的测定用NovoStar酶标仪(德国BMG公司)。
本公开的已知的起始原料可以采用或按照本领域已知的方法来合成,或可购买自ABCR GmbH&Co.KG,Acros Organics,Aldrich Chemical Company,韶远化学科技(Accela ChemBio Inc)、达瑞化学品等公司。
实施例中无特殊说明,反应能够均在氩气氛或氮气氛下进行。
氩气氛或氮气氛是指反应瓶连接一个约1L容积的氩气或氮气气球。
氢气氛是指反应瓶连接一个约1L容积的氢气气球。
加压氢化反应使用Parr 3916EKX型氢化仪和清蓝QL-500型氢气发生器或HC2-SS型氢化仪。
氢化反应通常抽真空,充入氢气,反复操作3次。
微波反应使用CEM Discover-S 908860型微波反应器。
实施例中无特殊说明,溶液是指水溶液。
实施例中无特殊说明,反应的温度为室温,为20℃~30℃。
实施例中的反应进程的监测采用薄层色谱法(TLC),反应所使用的展开剂,纯化化合物采用的柱层析的洗脱剂的体系和薄层色谱法的展开剂体系包括:A:二氯甲烷/甲醇体系,B:正己烷/乙酸乙酯体系,C:石油醚/乙酸乙酯体系,溶剂的体积比根据化合物的极性不同而进行调节,也可以加入少量的三乙胺和醋酸等碱性或酸性试剂进行调节。
实施例1
5-氯-2-(4-氟-2-甲基苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺1
第一步
(4-氟-2-甲基苯基)溴化镁1b
将打磨光亮的镁条(760mg,31.7mmol,药),剪碎,加入四氢呋喃(80mL)中,氩气保护。室温下,滴入三甲基氯硅烷(345mg,3.17mmol,韶远科技(上海)有限公司)。加入1-溴-4-氟-2-甲基苯1a(1.5g,7.9mmol,韶远科技(上海)有限公司),加热引发反应后,再加入化合物1a(4.5g,23.7mmol,韶远科技(上海)有限公司)。加热45℃反应1小时,镁条全部消失,形成灰色均匀液体,得到标题化合物1b溶液(0.4M,80mL),产物不经纯化,直接用于下一步反应。
第二步
2-溴-4-氯-5-(三氟甲基)苯甲醛1d
将四氢呋喃(100mL)和六甲基二硅基胺基锂(1M,120mL,120mmol,泰坦化学)在氩气保护冷却至-78℃。滴加4-溴-2-氯-1-(三氟甲基)苯1c(25g,96.36mmol,韶远科技(上海)有限公司),保持低温反应2小时。滴加N,N-二甲基甲酰胺(14.1g,192.9mmol,百灵威科技有限公司),逐渐升至室温反应16小时。加入水,乙酸乙酯萃取(50mL×3)。有机相用无水硫酸钠干燥。过滤,滤液减压浓缩,用硅胶柱色谱法以展开剂体系B纯化,得到标题化合物1d(3.14g),产率:28%。
1H NMR(400MHz,CDCl
3)δ10.32(s,1H),8.23(s,1H),7.87(s,1H)。
第三步
2-溴-4-氯-5-(三氟甲基)苯基)(4-氟-2-甲基苯基)甲醇1e
将化合物1d(900mg,3.13mmol)溶于四氢呋喃(10mL),滴加现制的化合物1b溶液(7.97mmol,19.92mL),室温反应1小时。加入饱和氯化铵溶液,乙酸乙酯萃取(10mL×3)。有机相用无水硫酸钠干燥。过滤,滤液减压浓缩,用硅胶柱色谱法以展开剂体系B纯化,得到标题化合物1e(900mg),产率72%。
1HNMR(400MHz,CD
3OD)δ8.04(s,1H),7.93(s,1H),7.0-7.03(m,1H),6.87-6.90(m,2H),6.17(s,1H),2.5(s,3H)。
第四步
1-溴-5-氯-2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯1f
将化合物1e(4g,10.1mmol)溶于二氯甲烷(50mL),冷却至0℃加入三氟醋酸(10mL,泰坦化学),滴加三乙基硅烷(6mL,韶远科技(上海)有限公司),0℃反应1小时。加水淬灭反应,乙酸乙酯萃取(10mL×3)。有机相用无水硫酸钠干燥。过滤,滤液减压浓缩,用硅胶柱色谱法以展开剂体系B纯化,得到标题化合物1f(3.2g),产率83%。
第五步
5-氯-2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯甲酸甲酯1g
将化合物1f(3.3g,8.64mmol)溶于甲醇(60mL)。加入醋酸钯(388.31mg,1.73mmol,百灵威科技有限公司),1,1′-双(二苯基膦)二茂铁(960mg,1.73mmol,韶远科技(上海)有限公司),三乙胺(2.63g,25.94mmol,国药集团化学试剂有限公司)。反应体系连接一氧化碳气球,60℃反应16小时。硅藻土过滤,滤液减压浓缩,用硅胶柱色谱法以展开剂体系B纯化,得到标题化合物1g(2.2g),为白色固体,产率71%。
MS m/z(ESI):359.1[M-1]。
第六步
5-氯-2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯甲酸1h
将化合物1g(2.2g,6.1mmol)溶于甲醇(40mL)和水(20mL)中,加入氢氧化钠溶液(5M,6mL,30mmol,国药集团化学试剂有限公司),升温至40℃反应3小时。冷却,4M盐酸调节pH为2,二氯甲烷萃取(10mL×3)。有机相用无水硫酸钠干燥。过滤,滤液减压浓缩,得标题粗品化合物1h(2.1g),,产物不经纯化,直接用于下一步反应。
MS m/z(ESI):345.1[M-1]。
第七步
5-氯-2-(4-氟-2-甲基苄基)-N-(2-甲氧基吡啶-4-基)-4-(三氟甲基)苯甲酰胺1i
化合物1h(1.1g,3.17mmol)溶于吡啶(20mL),冷却至-20℃,滴加三氯氧磷 (4.86g,31.7mmol,国药集团化学试剂有限公司)。滴加完毕,再加入4-氨基-2-甲氧基吡啶(788mg,6.34mmol,韶远科技(上海)有限公司),-20℃反应半小时。反应液倒入冰水,二氯甲烷萃取(10mL×3),滤液减压浓缩,得粗品标题化合物1i(1.4g),产物不经纯化,直接用于下一步反应。
MS m/z(ESI):451.2[M-1]。
第八步
5-氯-2-(4-氟-2-甲基苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺1
将化合物1i(1.4g,3.1mmol)溶于N,N-二甲基甲酰胺,加入吡啶氢溴酸盐(2.5g,15.7mmol,韶远科技(上海)有限公司),升温至100℃,反应1.5小时。冷却,过滤,滤液用高效液相色谱制备(Waters 2767-SQ Detecor2,洗脱体系:碳酸氢铵,水,乙腈)纯化,得到标题化合物1(1g),产率74%。
MS m/z(ESI):439.1[M+1]。
1H NMR(400MHz,DMSO-d
6)δ11.25(s,1H),10.53(s,1H),7.90(s,1H),7.56(s,1H),7.27(d,1H),6.88-6.98(m,3H),6.88(s,1H),6.28(d,1H),4.08(s,2H),2.10(s,3H)。
实施例2
5-氯-2-(2-环丙基-4-氟苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺2
第一步
5-氯-2-(4-氟-2-羟基苄基)-4-(三氟甲基)苯甲酸甲酯2b
采用实施例1的合成路线,将第一步原料4-氟-2-甲基溴苯替换为化合物4-氟-2-甲氧基溴苯,制得5-氯-2-(4-氟-2-甲氧基苄基)-4-(三氟甲基)苯甲酸甲酯2a(250mg)。将化合物2a(250mg,0.66mmol)溶于四氢呋喃,冰水浴冷却,滴加三溴化硼(3.33g,13.3mmol,国药集团化学试剂有限公司),室温下反应过夜。加入甲醇反应,减压浓缩,用硅胶柱色谱法以展开剂体系B纯化,得到标题化合物2b(120mg),产率:50%。
第二步
5-氯-2-(4-氟-2-(((三氟甲基)磺酰基)氧基)苄基)-4-(三氟甲基)苯甲酸甲酯2c
冰浴下,将化合物2b(120mg,0.33mmol)和N-苯基双(三氟甲烷磺酸亚胺)(178mg,0.49mmol,韶远科技(上海)有限公司)溶于二氯甲烷(3mL)和乙腈(3mL)的混合溶剂(v/v=1/1)。加入碳酸铯(216mg,0.66mmol),升至室温反应3小时。减压浓缩,用硅胶柱色谱法以展开剂体系B纯化,得到标题化合物2c(130mg),产率:79%。
第三步
5-氯-2-(2-环丙基-4-氟苄基)-4-(三氟甲基)苯甲酸甲酯2d
将化合物2c(130mg,0.26mmol)溶于甲苯(5mL),加入四三苯基膦钯(32mg,0.03mmol,百灵威科技有限公司),氟化钾(60mg,1.03mmol,国药集团化学试剂有限公司),溴化钾(44mg,0.37mmol,国药集团化学试剂有限公司)和环丙基硼酸(44mg,0.51mmol,韶远科技(上海)有限公司),充入氩气,置于微波反应器中,100℃反应1小时。减压浓缩,得到粗品标题化合物2d(120mg),产物不经纯化,直接用于下一步反应。
第四步
5-氯-2-(2-环丙基-4-氟苄基)-4-(三氟甲基)苯甲酸2e
将化合物2d(120mg,0.31mmol)溶于甲醇(4mL)和水(2mL)中,加入氢氧化钠(25mg,0.625mmol),升温至40℃反应3小时。冷却,用稀盐酸调节至pH=2,二氯甲烷萃取(10mL×3)。有机相用无水硫酸钠干燥,过滤,滤液减压浓缩,得粗品标题化合物2e(115mg),产物不经纯化,直接用于下一步反应。
第五步
5-氯-2-(2-环丙基-4-氟苄基)-N-(2-甲氧基吡啶-4-基)-4-(三氟甲基)苯甲酰胺2f
化合物2e(115mg,0.31mmol)溶于吡啶(3mL),冷却至-20℃,滴加三氯氧磷(142mg,0.93mmol,国药集团化学试剂有限公司)。滴加完毕,再加入4-氨基-2-甲氧基吡啶(77mg,0.62mmol,韶远科技(上海)有限公司),-20℃反应半小时。反应液倒入冰水,二氯甲烷萃取(10mL×3)。有机相用无水硫酸钠干燥,过滤,滤液减压浓缩,得粗品标题化合物2f(143mg),产物不经纯化,直接用于下一步反应。
第六步
5-氯-2-(2-环丙基-4-氟苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺2
将化合物2f(143mg,0.3mmol)溶于N,N-二甲基甲酰胺,加入吡啶氢溴酸盐(300mg,1.89mmol,韶远科技(上海)有限公司),升温至100℃,反应1.5小时。冷却,过滤,滤液用高效液相色谱制备(Waters2767-SQ Detecor2,洗脱体系:碳酸氢铵,水,乙腈)纯化,得到标题化合物2(30mg)。
MS m/z(ESI):463.1[M-1]。
1H NMR(400MHz,CD
3OD)δ7.77(s,1H),7.55(s,1H),7.39-7.40(m,1H),6.98-6.99(m,2H),6.80-6.81(m,1H),6.73(m,1H),6.63-6.65(m,1H),4.36(s,2H),1.79(m,1H),0.84-0.87(m,2H),0.57-0.59(m,2H)。
实施例3
5-氯-2-(4-氟-2-甲氧基苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺
采用实施例1的合成路线,将第一步原料1-溴-4-氟-2-甲基苯替换为1-溴-4-氟-2-甲氧基苯,制得标题化合物3(150mg)。
MS m/z(ESI):455.1[M+1]。
1H NMR(400MHz,CD
3OD)δ7.69(s,2H),7.44(d,1H),7.11-7.06(m,2H),6.70-6.66(m,2H),6.55-6.51(m,1H),4.12(s,2H),3.69(s,3H)。
实施例4
5-氯-2-(2-乙基-4-氟苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺
采用实施例1的合成路线,将第一步原料1-溴-4-氟-2-甲基苯替换为化合物1-溴-4-氟-2-乙基苯,制得标题化合物4(50mg)。
MS m/z(ESI):451.2[M-1]。
1H NMR(400MHz,CD
3OD):δ7.77(m,1H),7.54(m,1H),7.38-7.40(m,1H),6.96-7.00(m,2H),6.90-6.92(m,1H),6.82-6.86(m,1H),6.61-6.63(m,1H),4.21(s,2H),2.55-2.61(m,2H),1.11-1.14(m,3H)。
实施例5
2-(4-氟-2-甲基苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺5
第一步
2-((4-氟-2-甲基苯基)(羟基)甲基)-4-(三氟甲基)苯甲酸5c
将化合物2-溴-4-(三氟甲基)苯甲酸5a(1g,3.72mmol,韶远科技(上海)有限公司)溶于四氢呋喃(50mL),冷却至-78℃,滴加正丁基锂(2.5M,6mL,14.9mmol,百灵威科技有限公司)。保持-78℃反应1小时,将4-氟-2-甲基苯甲醛5b(771mg,5.58mmol,韶远科技(上海)有限公司)溶于四氢呋喃(10mL),滴入上述反应液中。升温至-20℃,反应1小时。少量甲醇反应,反应液减压浓缩,得到粗品标题化合物5c(1g),产物不经纯化,直接用于下一步反应。
第二步
3-(4-氟-2-甲基苯基)-5-(三氟甲基)异苯并呋喃-1(3H)-酮5d
将化合物5c(1g)溶于乙酸(50mL),加入氢溴酸(2mL,国药集团化学试剂有限公司),90℃反应过夜。反应液减压浓缩,用硅胶柱色谱法以展开剂体系B纯化, 得到标题化合物5d(600mg),产率52%。
第三步
2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯甲酸5e
将醋酸钯(44mg,0.2mmol,百灵威科技有限公司)和钠氢(78mg,3.4mmol,国药集团化学试剂有限公司)置于N,N-二甲基乙酰胺(1mL),氩气保护下,反应10分钟。将化合物5d(310mg,1mmol)溶于N,N-二甲基乙酰胺(1mL),滴入上述的反应液中,室温反应2.5小时。将稀盐酸加入到反应液中,滤去残渣,滤液用乙酸乙酯萃取(20mL×3)。合并有机相,用无水硫酸钠干燥,过滤,滤液减压浓缩,用硅胶柱色谱法以展开剂体系B纯化,得到标题化合物5e(200mg),产率67%。
第四步
2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯甲酰氯5f
将化合物5e(100mg,0.32mmol)溶于氯化亚砜(2mL),80℃反应2小时。反应液减压浓缩,得到标题化合物5f(105mg),直接用于下一步反应。
第五步
2-(4-氟-2-甲基苄基)-N-(2-甲氧基吡啶-4-基)-4-(三氟甲基)苯甲酰胺5g
将化合物5f(105mg,0.32mmol),4-氨基-4-甲氧基吡啶(79mg,0.64mmol,韶远科技(上海)有限公司),吡啶(1mL)加入二氯甲烷(5mL)。室温反应过夜,反应液减压浓缩,用硅胶柱色谱法以展开剂体系A纯化,得到标题化合物5g(130mg),产率97%。
第六步
2-(4-氟-2-甲基苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺5
将化合物5g(130mg,0.31mmol)溶于N,N-二甲基甲酰胺(3mL),加入溴化氢吡啶(150mg,0.94mmol,韶远科技(上海)有限公司),100℃反应1小时。反应液减压浓缩,用硅胶柱色谱法以展开剂体系A纯化,得到标题化合物5(30mg),产率24%。
MS m/z(ESI):405.0[M+1]。
1H NMR(400MHz,CD
3OD)δ7.66(s,2H),7.54-7.56(m,1H),7.46(s,1H),7.22(s,1H),6.95-6.97(m,1H),6.84-6.87(m,1H),6.76-6.79(m,2H),4.21(s,2H),2.19(s,3H)。
实施例6
5-氯-2-(4-氟-2-(甲氧基-d
3)苄基)-N-(2-氧代-1,2-二氢吡啶-4-基)-4-(三氟甲基)苯甲酰胺6
采用实施例1的合成路线,将第一步原料1-溴-4-氟-2-甲基苯替换为1-溴-4-氟-2-(甲氧基-d3)苯,制得标题化合物6(20mg)。
MS m/z(ESI):458.1[M+1]。
实施例7
(4-(5-氯-2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯甲酰基)-2-氧代吡啶-1(2H)-基)甲基磷酸二氢酯7
第一步
5-氯-N-(1-(氯甲基)-2-氧代-1,2-二氢吡啶-4-基)-2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯甲酰胺7a
将化合物1(50mg,0.11mmol)和1,4-二氮杂双环[2.2.2]辛烷(6.40mg,0.06mmol,韶远科技(上海)有限公司)置于N,N-二甲基甲酰胺(1mL)和二氯甲烷(15mL)中。氩气保护下,滴加氯甲酸氯甲酯(22mg,0.17mmol,韶远科技(上海)有限公司),60℃反应3小时,室温搅拌过夜。加入水,用乙酸乙酯萃取(5mL×3)。合并有机相,用无水硫酸钠干燥,过滤,减压浓缩,得到粗品标题化合物7a(55mg),产物不经纯化,直接用于下一步反应。
MS m/z(ESI):487.0[M+1]。
第二步
((4-(5-氯-2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯甲酰氨基)-2-氧代吡啶-1(2H)-基)甲 基)磷酸二叔丁酯7b
化合物7a(55mg,0.11mmol)溶于N,N-二甲基甲酰胺(3mL),加入磷酸二叔丁酯钾盐(56mg,0.22mmol,国药集团化学试剂有限公司),四丁基碘化铵(3.76mg,0.011mmol,韶远科技(上海)有限公司),70℃反应3小时。加入水,用乙酸乙酯萃取(5mL×3)。合并有机相,用无水硫酸钠干燥,过滤,减压浓缩,得到标题化合物7b粗品(70mg),产物不经纯化,直接用于下一步反应。
MS m/z(ESI):659.2[M-1]。
第三步
(4-(5-氯-2-(4-氟-2-甲基苄基)-4-(三氟甲基)苯甲酰氨基)-2-氧代吡啶-1(2H)-基)甲基磷酸二氢酯7
化合物7b(70mg,0.1mmol)溶于乙腈(1mL),乙酸(1mL)和水(1mL)的混合溶剂中,70℃反应3小时。反应液减压浓缩,用高效液相色谱制备(Waters 2767-SQDetecor2,洗脱体系:碳酸氢铵,水,乙腈)纯化,得到标题化合物7(15mg)。
MS m/z(ESI):548.8[M+1]
1H NMR(400MHz,DMSO-d
6)δ10.64(s,1H),7.88(s,1H),7.67(d,1H),7.49(s,1H),7.18(br,2H),6.76-6.96(m,3H),6.28(d,1H),5.31(d,2H),4.05(s,2H),2.09(s,3H)。
生物学评价
以下结合测试例进一步描述解释本公开,但这些实施例并非意味着限制本公开的范围。
测试例1、本公开化合物对Nav1.8抑制活性的测定
实验的目的是为了调查化合物在离体实验中对Na
V1.8离子通道的影响,Na
V1.8离子通道稳定地表达在HEK293细胞上。在Na
V1.8电流稳定后,比较化合物应用前后Na
V1.8电流的大小,可以得到化合物对Na
V1.8离子通道的影响。
1实验材料及仪器
1)膜片钳放大器:patch clamp PC-505B(WARNER instruments)/MultiClamp 700A(Axon instrument)
2)数模转换器:Digidata 1440A(Axon CNS)/Digidata1550A(Axon instruments)
3)微操控仪:MP-225(SUTTER instrument)
4)倒置显微镜:TL4(Olympus)
5)玻璃微电极拉制仪:PC-10(NARISHIGE)
6)微电极玻璃毛细管:B12024F(武汉微探科学仪器有限公司)
7)二甲基亚砜(DMSO):D2650(Sigma-Aldrich)
8)河豚毒素(TTX):AF3014(Affix Scientific)
2实验步骤
2.1化合物配制
配制细胞内外液的化合物除用于酸碱滴定的NaOH和KOH外,均从Sigma(St.Louis,MO)公司购买。细胞外液(mM)为:NaCl,137;KCl,4;CaCl
2,1.8;MgCl
2,1;HEPES,10;葡萄糖10;pH7.4(NaOH滴定)。细胞内液(mM)为天冬氨酸,140;MgCl2,2;EGTA 11;HEPES,10;pH 7.2(CsOH滴定)。所有测试化合物和对照化合物溶液均含1μM TTX。
测试化合物的保存浓度为9mM,溶于二甲基亚砜(DMSO)。测试当天再溶于细胞外液,配制成要求浓度。
2.2手动膜片钳测试过程
1)化合物配制成指定浓度的溶液后,按浓度从从低到高顺序将药液依次加入各个管道,并对各个管道进行标记。
2)将细胞转移到灌流槽中,电极内施加正压,将电极尖端接触到细胞,抽气装置三通阀调成三通状态,然后对电极施加负压,使得电极与细胞形成高阻封接。继续施加负压,使得细胞膜破裂,形成电流通路。
3)待细胞破膜电流稳定后,依次进行不同的浓度的灌注。若电流稳定至少一分钟即可换下一个浓度进行灌流。每个浓度灌流时间不超过五分钟。
4)清洗灌流槽。按药液浓度从高到低进行冲洗,每个浓度药液冲洗20s。最后用细胞外液冲洗1min。
2.3测试电压方程(resting)及结果
将细胞钳制在–80mV,然后用持续10毫秒方波去极化到10mV,以得到Na
V1.8电流。这一程序每5秒重复一次。检测方波引发的最大电流,待其稳定后,灌流测试化合物,当反应稳定后,计算阻断的强度。
3数据分析
资料将存于计算机系统做分析。资料采集和分析将用pCLAMP 10(Molecular Devices,Union City,CA),管理人员将审查分析结果。电流稳定指的是电流随时间变化在有限的范围内。电流稳定后的大小说用来计算化合物在此溶度的作用。
本公开化合物对Nav1.8的抑制活性通过以上的试验进行测定,测得的IC
50值见表1。
表1本公开化合物对Nav1.8通道活性抑制的IC
50
| 实施例编号 | IC 50(nM) |
| 1 | 1.6 |
| 2 | 0.36 |
| 3 | 0.41 |
| 4 | 1.31 |
| 5 | 43.9 |
结论:本公开中的化合物对Nav1.8通道活性具有明显的抑制效果。
药代动力学评价
测试例2、本公开化合物的药代动力学测试
1、摘要
以大鼠为受试动物,应用LC/MS/MS法测定了大鼠灌胃给予实施例3化合物和实施例7化合物后不同时刻血浆中的药物浓度。研究本公开化合物在大鼠体内的药代动力学行为,评价其药动学特征。
2、试验方案
2.1试验药品
实施例3化合物和实施例7化合物。
2.2试验动物
健康成年SD大鼠8只,雌雄各半,平均分成2组,每组4只,购自上海杰思捷实验动物有限公司。
2.3药物配制
称取一定量药物,加5%DMSO、5%tween80和90%生理盐水配制成0.2mg/mL无色澄明溶液。
2.4给药
SD大鼠禁食过夜后灌胃给药,给药剂量均为2.0mg/kg,给药体积均为10.0mL/kg。
3.操作
大鼠灌胃给药实施例3化合物和实施例7化合物,于给药前及给药后0.5,1.0,2.0,4.0,6.0,8.0,11.0,24.0小时由眼眶采血0.2mL,置于肝素化试管中,4℃、3500转/分钟离心10分钟分离血浆,于-20℃保存,给药后2小时进食。
测定不同浓度的药物灌胃给药后大鼠血浆中的待测化合物含量:取给药后各时刻的大鼠血浆25μL,加入内标溶液30μL,乙腈200μL,涡旋混合5分钟,离心10分钟(3600转/分钟),血浆样品取上清液2μL进行LC/MS/MS分析。
4、药代动力学参数结果
本公开化合物的药代动力学参数如下:
结论:上述研究结果证实,在大鼠中,实施例7化合物在体内转化为实施例1化合物。并且,本公开化合物的药代吸收良好,具有明显的药代动力学优势。
Claims (24)
- 一种通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中:R 0为氢原子或-CH 2O-R x;M为CR 7R 8或S原子;环A为芳基或杂芳基;R 1选自氢原子、卤素、烷基、烷氧基、卤代烷基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;R 2选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;R 3、R 4、R 5和R 6相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;R 7和R 8相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;或者,所述R 7和R 8与相连接的碳原子一起形成羰基、环烷基和杂环基,其中所述的环烷基和杂环基任选被选自烷基、烷氧基、氧代基、卤素、氨基、氰基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;R x选自氢原子、-S(O) 2OH、-S(O) 2O -Q +、-PO(OR y) 2、-PO(OH)O -Q +、-PO(O -) 22Q +和-PO(O -) 2W 2+;Q +为药学上可接受的单价阳离子;W 2+为药学上可接受的二价阳 离子;R y为氢原子或烷基;n为0、1、2、3、4或5;且t为0、1或2。
- 根据权利要求1所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其为通式(I)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中:M为CR 7R 8或S原子;环A为芳基或杂芳基;R 1选自氢原子、卤素、烷基、烷氧基、卤代烷基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;R 2选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;R 3、R 4、R 5和R 6相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;R 7和R 8相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基;或者,所述R 7和R 8与相连接的碳原子一起形成羰基、环烷基和杂环基,其中所述的环烷基和杂环基任选被选自烷基、烷氧基、氧代基、卤素、氨基、氰基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;n为0、1、2、3、4或5;且t为0、1或2。
- 根据权利要求1或2所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中环A为苯基或吡啶基。
- 根据权利要求1至3中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其为通式(II)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中:R 9、R 10、R 11、R 12和R 13相同或不同,且各自独立地选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;且M、R 1、R 3~R 6和t如权利要求1中所定义。
- 根据权利要求1所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其为通式(IIG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中:R 9、R 10、R 11、R 12和R 13相同或不同,且各自独立地选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、卤代烷基、卤代烷氧基、氰基、氨基、硝基、羟基、羟烷基、环烷基、杂环基、芳基和杂芳基,其中所述的烷基、环烷基、杂环基、芳基和杂芳基任选被选自烷基、卤代烷基、卤素、氨基、硝基、氰基、羟基、烷氧基、卤代烷氧基、羟烷基、环烷基、杂环基、芳基和杂芳基中的一个或多个取代基所取代;且R x、M、R 1、R 3~R 6和t如权利要求1中所定义。
- 根据权利要求1至5中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R 3、R 4、R 5和R 6相同或不同,且各自独立地选自氢原子、卤素、烷基、烷氧基、卤代烷基和卤代烷氧基。
- 根据权利要求4至6中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R 9、R 10、R 11、R 12和R 13相同或不同,且各自独立地选自氢原子、卤素、烷基、氘代烷基、烷氧基、氘代烷氧基、环烷基、卤代烷基和卤代烷氧基。
- 根据权利要求1至8中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中M为CR 7R 8;R 7和R 8如权利要求1中所定义。
- 根据权利要求1至11中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R 1为氢原子。
- 根据权利要求1至12中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R 4为卤素;且R 5为卤代烷基。
- 根据权利要求1、5和11中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,其中R x为-PO(OH) 2。
- 一种药物组合物,其含有根据权利要求1至15中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,以及一种或多种药学上可接受的载体、稀释剂或赋形剂。
- 根据权利要求1至15中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,或根据权利要求22所述的药物组合物在制备抑制受试者电压门控钠离子通道的药物中的用途,优选地,所述的电压门控钠离子通道为Na V1.8。
- 根据权利要求1至15中任一项所述的通式(IG)所示的化合物,或其互变异构体、内消旋体、外消旋体、对映异构体、非对映异构体或其混合物形式,或其可药用的盐,或根据权利要求22所述的药物组合物在制备治疗和/或减轻疼痛和疼痛相关疾病、多发性硬化症、夏-马-图三氏综合症、失禁或心律失常的药物中的用途,优选地,所述的疼痛选自慢性疼痛、急性疼痛、炎性疼痛、癌症疼痛、神经性疼痛、肌肉骨骼痛、手术后疼痛、内脏痛、肠痛和特发性疼痛。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202080005989.9A CN112996774B (zh) | 2019-01-25 | 2020-01-22 | 2-氧代-1,2-二氢吡啶类衍生物、其制备方法及其在医药上的应用 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910072432 | 2019-01-25 | ||
| CN201910072432.6 | 2019-01-25 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020151728A1 true WO2020151728A1 (zh) | 2020-07-30 |
Family
ID=71735671
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2020/073668 Ceased WO2020151728A1 (zh) | 2019-01-25 | 2020-01-22 | 2-氧代-1,2-二氢吡啶类衍生物、其制备方法及其在医药上的应用 |
Country Status (3)
| Country | Link |
|---|---|
| CN (1) | CN112996774B (zh) |
| TW (1) | TW202043200A (zh) |
| WO (1) | WO2020151728A1 (zh) |
Cited By (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022256702A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | Substituted tetrahydrofuran-2-carboxamides as modulators of sodium channels |
| WO2022256676A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | Substituted tetrahydrofuran analogs as modulators of sodium channels |
| WO2022256842A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | Hydroxy and (halo)alkoxy substituted tetrahydrofurans as modulators of sodium channels |
| WO2022256708A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | Solid dosage forms and dosing regimens comprising (2r,3s,4s,5r)-4-[[3-(3,4-difluoro-2-methoxy-phenyl)-4,5-dimethyl-5-(trifluoromethyl) tetrahydrofuran-2-carbonyl]amino]pyridine-2-carboxamide |
| WO2022256679A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | N-(hydroxyalkyl (hetero)aryl) tetrahydrofuran carboxamide analogs as modulators of sodium channels |
| WO2022256622A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | N-(hydroxyalkyl (hetero)aryl) tetrahydrofuran carboxamides as modulators of sodium channels |
| WO2023138599A1 (zh) | 2022-01-18 | 2023-07-27 | 成都康弘药业集团股份有限公司 | 芳香并环类Nav1.8抑制剂及其用途 |
| WO2023205465A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205463A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205468A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205778A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2024123815A1 (en) | 2022-12-06 | 2024-06-13 | Vertex Pharmaceuticals Incorporated | Process for the synthesis of substituted tetrahydrofuran modulators of sodium channels |
| WO2025090511A1 (en) | 2023-10-23 | 2025-05-01 | Vertex Pharmaceuticals Incorporated | Methods of preparing modulators of sodium channels and solid forms of the same for treating pain |
| WO2025090480A1 (en) | 2023-10-23 | 2025-05-01 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2025090516A1 (en) | 2023-10-23 | 2025-05-01 | Vertex Pharmaceuticals Incorporated | Methods of preparing compounds for treating pain and solid forms thereof |
| WO2025090465A1 (en) | 2023-10-23 | 2025-05-01 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2025122953A1 (en) | 2023-12-07 | 2025-06-12 | Vertex Pharmaceuticals Incorporated | Dosing regimens and formulations of suzetrigine for use in the treatment of acute and chronic pain |
| WO2026006674A1 (en) | 2024-06-28 | 2026-01-02 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2026008013A1 (zh) * | 2024-07-03 | 2026-01-08 | 江苏恒瑞医药股份有限公司 | 氧代杂芳基类化合物、其制备方法及其在医药上的应用 |
| WO2026013449A2 (en) | 2024-07-11 | 2026-01-15 | Sea4Us - Biotecnologia E Recursos Marinhos, Sa | Oxazolidone-derived compounds and their use in the treatment of chronic and acute pain |
| WO2026030525A1 (en) | 2024-07-31 | 2026-02-05 | Vertex Pharmaceuticals Incorporated | Zilvetrigine dosage forms and dosing regimens for treating pain |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105026373A (zh) * | 2013-01-31 | 2015-11-04 | 沃泰克斯药物股份有限公司 | 作为钠通道调节剂的吡啶酮酰胺 |
| CN105814067A (zh) * | 2013-12-13 | 2016-07-27 | 沃泰克斯药物股份有限公司 | 作为钠通道调节剂的吡啶酮酰胺的前药 |
| WO2018213426A1 (en) * | 2017-05-16 | 2018-11-22 | Vertex Pharmaceuticals Incorporated | Deuterated pyridone amides and prodrugs thereof as modulators of sodium channels |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BR112020000553A2 (pt) * | 2017-07-11 | 2020-07-21 | Vertex Pharmaceuticals Incorporated | carboxamidas como moduladores de canal de sódio |
-
2020
- 2020-01-22 TW TW109102572A patent/TW202043200A/zh unknown
- 2020-01-22 CN CN202080005989.9A patent/CN112996774B/zh active Active
- 2020-01-22 WO PCT/CN2020/073668 patent/WO2020151728A1/zh not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105026373A (zh) * | 2013-01-31 | 2015-11-04 | 沃泰克斯药物股份有限公司 | 作为钠通道调节剂的吡啶酮酰胺 |
| CN105814067A (zh) * | 2013-12-13 | 2016-07-27 | 沃泰克斯药物股份有限公司 | 作为钠通道调节剂的吡啶酮酰胺的前药 |
| WO2018213426A1 (en) * | 2017-05-16 | 2018-11-22 | Vertex Pharmaceuticals Incorporated | Deuterated pyridone amides and prodrugs thereof as modulators of sodium channels |
Cited By (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022256702A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | Substituted tetrahydrofuran-2-carboxamides as modulators of sodium channels |
| US11827627B2 (en) | 2021-06-04 | 2023-11-28 | Vertex Pharmaceuticals Incorporated | N-(hydroxyalkyl (hetero)aryl) tetrahydrofuran carboxamides as modulators of sodium channels |
| WO2022256842A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | Hydroxy and (halo)alkoxy substituted tetrahydrofurans as modulators of sodium channels |
| WO2022256708A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | Solid dosage forms and dosing regimens comprising (2r,3s,4s,5r)-4-[[3-(3,4-difluoro-2-methoxy-phenyl)-4,5-dimethyl-5-(trifluoromethyl) tetrahydrofuran-2-carbonyl]amino]pyridine-2-carboxamide |
| WO2022256679A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | N-(hydroxyalkyl (hetero)aryl) tetrahydrofuran carboxamide analogs as modulators of sodium channels |
| WO2022256622A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | N-(hydroxyalkyl (hetero)aryl) tetrahydrofuran carboxamides as modulators of sodium channels |
| WO2022256676A1 (en) | 2021-06-04 | 2022-12-08 | Vertex Pharmaceuticals Incorporated | Substituted tetrahydrofuran analogs as modulators of sodium channels |
| EP4699607A2 (en) | 2021-06-04 | 2026-02-25 | Vertex Pharmaceuticals Incorporated | N-(hydroxyalkyl (hetero)aryl) tetrahydrofuran carboxamides as modulators of sodium channels |
| EP4714435A2 (en) | 2021-06-04 | 2026-03-25 | Vertex Pharmaceuticals Incorporated | Solid dosage forms and dosing regimens comprising (2r,3s,4s,5r)-4-[[3-(3,4-difluoro-2-methoxy-phenyl)-4,5-dimethyl-5-(trifluoromethyl) tetrahydrofuran-2-carbonyl]amino]pyridine-2-carboxamide |
| US12258333B2 (en) | 2021-06-04 | 2025-03-25 | Vertex Pharmaceuticals Incorporated | N-(hydroxyalkyl (hetero)aryl) tetrahydrofuran carboxamides as modulators of sodium channels |
| WO2023138599A1 (zh) | 2022-01-18 | 2023-07-27 | 成都康弘药业集团股份有限公司 | 芳香并环类Nav1.8抑制剂及其用途 |
| WO2023205468A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| US12503439B2 (en) | 2022-04-22 | 2025-12-23 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205778A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205465A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205463A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2024123815A1 (en) | 2022-12-06 | 2024-06-13 | Vertex Pharmaceuticals Incorporated | Process for the synthesis of substituted tetrahydrofuran modulators of sodium channels |
| WO2025090465A1 (en) | 2023-10-23 | 2025-05-01 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2025090516A1 (en) | 2023-10-23 | 2025-05-01 | Vertex Pharmaceuticals Incorporated | Methods of preparing compounds for treating pain and solid forms thereof |
| WO2025090480A1 (en) | 2023-10-23 | 2025-05-01 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2025090511A1 (en) | 2023-10-23 | 2025-05-01 | Vertex Pharmaceuticals Incorporated | Methods of preparing modulators of sodium channels and solid forms of the same for treating pain |
| WO2025122953A1 (en) | 2023-12-07 | 2025-06-12 | Vertex Pharmaceuticals Incorporated | Dosing regimens and formulations of suzetrigine for use in the treatment of acute and chronic pain |
| WO2026006674A1 (en) | 2024-06-28 | 2026-01-02 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2026008013A1 (zh) * | 2024-07-03 | 2026-01-08 | 江苏恒瑞医药股份有限公司 | 氧代杂芳基类化合物、其制备方法及其在医药上的应用 |
| WO2026013449A2 (en) | 2024-07-11 | 2026-01-15 | Sea4Us - Biotecnologia E Recursos Marinhos, Sa | Oxazolidone-derived compounds and their use in the treatment of chronic and acute pain |
| WO2026030525A1 (en) | 2024-07-31 | 2026-02-05 | Vertex Pharmaceuticals Incorporated | Zilvetrigine dosage forms and dosing regimens for treating pain |
Also Published As
| Publication number | Publication date |
|---|---|
| CN112996774A (zh) | 2021-06-18 |
| CN112996774B (zh) | 2022-11-22 |
| TW202043200A (zh) | 2020-12-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112996774B (zh) | 2-氧代-1,2-二氢吡啶类衍生物、其制备方法及其在医药上的应用 | |
| CN112390745B (zh) | 吡啶烟酰胺类衍生物、其制备方法及其在医药上的应用 | |
| TW202115038A (zh) | 苯甲醯胺稠芳環類衍生物、其製備方法及其在醫藥上的應用 | |
| CN112955436B (zh) | 6-氧代-1,6-二氢哒嗪类衍生物、其制备方法及其在医药上的应用 | |
| JP7813970B2 (ja) | 縮合イミダゾール誘導体、その調製方法及びその医薬的応用 | |
| WO2021254470A1 (zh) | 6-氧代-3,6-二氢吡啶类衍生物、其制备方法及其在医药上的应用 | |
| WO2021121210A1 (zh) | 并环类衍生物、其制备方法及其在医药上的应用 | |
| CN112996776B (zh) | 6-氧代-1,6-二氢哒嗪类前药衍生物、其制备方法及其在医药上的应用 | |
| WO2025067219A1 (zh) | 杂环类化合物、其制备方法及其在医药上的应用 | |
| WO2021238834A1 (zh) | 芳甲酰胺类化合物及其制备方法和医药用途 | |
| WO2021018165A1 (zh) | 吡啶苯甲酰胺类衍生物、其制备方法及其在医药上的应用 | |
| HK40051454A (zh) | 6-氧代-1,6-二氢哒嗪类前药衍生物、其制备方法及其在医药上的应用 | |
| WO2020192553A1 (zh) | 磺酰基取代的苯并杂环甲酰胺衍生物、其制法与医药上的用途 | |
| RU2791534C2 (ru) | Производное 6-оксо-1,6-дигидропиридазина, способ его получения и его применение в медицине | |
| CN121627648A (zh) | 芳香环类化合物、其制备方法及其在医药上的应用 | |
| WO2026008013A1 (zh) | 氧代杂芳基类化合物、其制备方法及其在医药上的应用 | |
| WO2026001999A1 (zh) | 芳香环类化合物、其制备方法及其在医药上的应用 | |
| WO2026057012A1 (zh) | 芳香环类化合物、其制备方法及其在医药上的应用 | |
| HK40051450A (zh) | 6-氧代-1,6-二氢哒嗪类衍生物、其制备方法及其在医药上的应用 | |
| CN115785094A (zh) | 苄基取代α-咔啉化合物或其药用盐、其药物组合物及其制备方法和用途 | |
| TW202547515A (zh) | 芳香環類化合物、其製備方法及其在醫藥上的應用 | |
| CN120774886A (zh) | 芳香环类化合物、其制备方法及其在医药上的应用 | |
| HK40082497B (zh) | 稠合咪唑类衍生物、其制备方法及其在医药上的应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20745602 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 20745602 Country of ref document: EP Kind code of ref document: A1 |