WO2016104580A1 - カチオン性脂質 - Google Patents
カチオン性脂質 Download PDFInfo
- Publication number
- WO2016104580A1 WO2016104580A1 PCT/JP2015/085969 JP2015085969W WO2016104580A1 WO 2016104580 A1 WO2016104580 A1 WO 2016104580A1 JP 2015085969 W JP2015085969 W JP 2015085969W WO 2016104580 A1 WO2016104580 A1 WO 2016104580A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- lipid
- composition
- cationic lipid
- same manner
- sirna
- Prior art date
Links
- ZEMNDHMQMYTBIN-UHFFFAOYSA-N CCCCCCCCCCC(CCCCCCCCC(OCCC(CCCSSCCCC)CCCSSCCCC)=O)OC(C1CCN(C)CC1)=O Chemical compound CCCCCCCCCCC(CCCCCCCCC(OCCC(CCCSSCCCC)CCCSSCCCC)=O)OC(C1CCN(C)CC1)=O ZEMNDHMQMYTBIN-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/36—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D211/60—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D211/62—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals attached in position 4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/22—Heterocyclic compounds, e.g. ascorbic acid, tocopherol or pyrrolidones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/28—Steroids, e.g. cholesterol, bile acids or glycyrrhetinic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/02—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C229/04—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C229/06—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/02—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C229/04—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C229/06—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton
- C07C229/10—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings
- C07C229/12—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings to carbon atoms of acyclic carbon skeletons
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1137—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against enzymes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering N.A.
Definitions
- the present invention relates to a novel cationic lipid.
- This application claims priority based on Japanese Patent Application No. 2014-266548 filed in Japan on December 26, 2014, the contents of which are incorporated herein by reference.
- Nucleic acids such as siRNA (small interfering RNA), miRNA (micro RNA), shRNA (short hairpin RNA, or small hairpin RNA) expression vectors, antisense oligonucleotides, and the like induce sequence-specific gene expression suppression in vivo. It is a nucleic acid and is known as a nucleic acid medicine.
- siRNA is particularly attracting attention.
- siRNA is a double-stranded RNA consisting of 19 to 23 base pairs, and induces sequence-specific gene expression suppression called RNA interference (RNAi).
- RNAi RNA interference
- siRNA is chemically stable, it has problems for therapeutic use in that it is easily degraded by RNase (ribonuclease) in plasma and it is difficult to permeate the cell membrane alone.
- RNase ribonuclease
- encapsulating siRNA in microparticles containing cationic lipids protects the encapsulated siRNA from degradation in plasma and allows permeation to a fat-soluble cell membrane.
- encapsulating siRNA in microparticles containing cationic lipids protects the encapsulated siRNA from degradation in plasma and allows permeation to a fat-soluble cell membrane.
- Patent Documents 2 to 4 describe cationic lipids that are used for delivery of nucleic acid drugs such as siRNA and have improved biodegradability.
- fine particles containing cationic lipids have a problem of stability that they tend to aggregate during storage, and a method of suppressing aggregation by containing polyethylene glycol-modified lipid (PEG lipid) in the fine particles is known. ing. Furthermore, a method for improving aggregation efficiency and nucleic acid delivery efficiency by using PEG-DPG, which is a specific PEG lipid, as a constituent of microparticles or a preparation comprising the microparticles and a deionized solvent, It is described in Patent Document 5.
- the present invention relates to the following [1] to [8].
- [1] A compound selected from the group consisting of compounds represented by the following formulas (1) to (11) or a pharmaceutically acceptable salt thereof.
- the compound represented by the above formula (8) is a cationic lipid.
- composition comprising at least one lipid and (III) a nucleic acid.
- the cationic lipid of the present invention nucleic acid can be efficiently released into the cytoplasm. Therefore, the cationic lipid of the present invention has applicability as a lipid for nucleic acid delivery to the cytoplasm.
- the present invention is a compound represented by any one of the above formulas (1) to (11) or a pharmaceutically acceptable salt thereof, and can be used as a cationic lipid.
- the cationic lipid may be a hydrate of the salt or a solvate of the salt.
- a cationic lipid is an amphiphilic molecule having a lipophilic region containing one or more hydrocarbon groups and a hydrophilic region containing a polar group that is protonated at physiological pH. That is, the cationic lipid of the present invention can be protonated to form a cation.
- the anion that can be contained in the cationic lipid of the present embodiment in combination with the cation is not particularly limited as long as it is pharmaceutically acceptable.
- R 3 represents an alkylene group having 4 to 12 carbon atoms
- R 4 represents an alkyl group having 7 to 12 carbon atoms
- R 8 represents a structure represented by the following formula (13).
- R 9 represents a structure represented by the following formula (14).
- R 1 is an alkyl group having 4 to 10 carbon atoms which may have one or more cyclopropane or cyclobutane formed by condensation of a part of the carbon chain, or 4 to 10 carbon atoms.
- R 2 represents a hydrogen atom or an alkyl group having 2 to 8 carbon atoms, and m represents an integer of 0 to 5.
- R 5 , R 6 and R 7 each independently represents a hydrogen atom or an alkyl group having 1 to 10 carbon atoms, and n represents an integer of 0 to 5.
- R 5 and R 6 may be combined to form a monocyclic heterocycle, and R 6 and R 7 may be combined to form a monocyclic heterocycle.
- the cationic lipid (1-7) can be synthesized as follows.
- a dialkyl dicarboxylate represented by the formula (1-1) is hydrolyzed to obtain a monoester represented by the formula (1-2).
- Formula (1-1) shows an example in which the alkyl group of the dialkyl dicarboxylate is a methyl group, but the alkyl group of the dialkyl dicarboxylate is not limited to a methyl group, and an appropriate alkyl group should be selected. Can do.
- the compound represented by the formula (1-2) is acid chlorided to obtain the compound represented by the formula (1-3).
- the compound represented by the formula (1-4) is obtained by reacting the compound represented by the formula (1-3) with a Grignard reagent.
- a compound represented by formula (1-5) is obtained by transesterification, and a compound represented by formula (1-6) is obtained by reduction.
- the cationic lipid (1-7) can be synthesized by condensing the compound represented by the formula (1-6) with a carboxylic acid.
- the present invention provides a lipid complex containing (I) the above-described cationic lipid and (II) at least one lipid selected from the group consisting of a neutral lipid, a polyethylene glycol-modified lipid, and a sterol.
- the lipid complex includes (I) the above-described cationic lipid, and (II) at least one lipid selected from the group consisting of neutral lipid, polyethylene glycol-modified lipid, and sterol, III) a nucleic acid.
- the lipid complex of this embodiment enables nucleic acid to be efficiently released into the cytoplasm.
- the lipid complex of the present embodiment contains, for example, 10 to 100 mol%, such as 20 to 90 mol%, such as 40 to 70 mol%, of the above-described cationic lipid based on the total lipid contained in the lipid complex.
- Cationic lipids can be used singly or in combination of two or more.
- nucleic acid examples include siRNA, miRNA, shRNA expression vector, antisense oligonucleotide, ribozyme and the like.
- the nucleic acid may be siRNA or miRNA.
- the lipid complex of this embodiment contains, for example, 0.01 to 50% by weight of nucleic acid, for example, 0.1 to 30% by weight, for example, 1 to 10% by weight.
- the lipid complex of the present embodiment comprises, as a lipid component, (I) the above-described cationic lipid, and (II) at least one lipid selected from the group consisting of a neutral lipid, a polyethylene glycol-modified lipid, and a sterol. contains.
- Neutral lipids include dioleoylphosphatidylethanolamine (DOPE), palmitoyl oleoylphosphatidylcholine (POPC), egg phosphatidylcholine (EPC), dimyristoylphosphatidylcholine (DMPC), dipalmitoylphosphatidylcholine (DPPC), distearoylphosphatidylcholine (DSPC) , Diarachidoylphosphatidylcholine (DAPC), dibehenoylphosphatidylcholine (DBPC), dilignocelloylphosphatidylcholine (DLPC), dioleoylphosphatidylcholine (DOPC), sphingomyelin, ceramide, dioleoylphosphatidylglycerol (DOPG), dipalmitoyl Phosphatidylglycerol (DPPG), phosphatidylethanolamine POPE), dioleoyl - phosphatid
- the lipid complex of the present embodiment may contain neutral lipid, for example, 0 to 50 mol%, for example 0 to 40 mol%, for example 0 to 30 mol%, based on the total lipid contained in the lipid complex. Good.
- polyethylene glycol-modified lipid examples include PEG2000-DMG (PEG2000-dimyristylglycerol, PEG2000-DPG (PEG2000-dipalmitoylglycerol), PEG2000-DSG (PEG2000-distearoylglycerol), PEG5000-DMG (PEG5000-dimyristylglycerol, PEG5000).
- -DPG PEG5000-dipalmitoylglycerol
- PEG5000-DSG PEG5000-distearoylglycerol
- PEG-cDMA N-[(methoxypoly (ethyleneglycol) 2000) carbamyl] -1,2-dimyristyloxylpropyl-3- Amine
- PEG-C-DOMG R-3-[( ⁇ -methoxy-poly (ethylene glycol) 2 00) carbamoyl)]-1,2-dimyristyloxylpropyl-3-amine
- PEG polyethylene glycol
- PEG -diacylglycerol
- DAA PEG-dialkyloxypropyl
- PEG-phospholipid PEG-ceramide
- PEG-dialkyloxypropyl examples include PEG-dilauryloxypropyl, PEG-dimyristyloxypropyl, PEG-dipalmityloxypropyl, PEG-distearyloxypropyl, and the like.
- Polyethylene glycol-modified lipids can be used alone or in combination of two or more.
- the lipid complex of the present embodiment contains, for example, 0 to 30 mol%, such as 0 to 20 mol%, such as 0 to 10 mol%, of polyethylene glycol-modified lipid, based on the total lipid contained in the lipid complex. Also good.
- sterols examples include cholesterol, dihydrocholesterol, lanosterol, ⁇ -sitosterol, campesterol, stigmasterol, brassicasterol, ergocasterol, fucosterol, and 3 ⁇ - [N- (N ′, N′-dimethylaminoethyl) carbamoyl].
- examples include cholesterol (DC-Chol).
- a sterol can be used individually by 1 type or in mixture of 2 or more types.
- the lipid complex of this embodiment may contain, for example, 0 to 90 mol%, such as 10 to 80 mol%, such as 20 to 50 mol%, of sterol based on the total lipid contained in the lipid complex.
- the combination of lipid components in the lipid complex of the present embodiment is not particularly limited.
- the combination of the above-described cationic lipid, neutral lipid and sterol, the above-described cationic lipid, neutral lipid, polyethylene glycol-modified lipid, and Examples include sterol combinations.
- the present invention provides (I) a cationic lipid as described above, (II) at least one lipid selected from the group consisting of a neutral lipid, a polyethylene glycol-modified lipid, and a sterol, and (III) a nucleic acid.
- the composition containing this is provided.
- the composition of this embodiment makes it possible to efficiently release nucleic acids into the cytoplasm.
- the composition of this embodiment may contain the lipid complex described above, a pharmaceutically acceptable medium, and optionally other additives. Pharmaceutically acceptable media and other additives will be described later.
- composition of the present embodiment contains, for example, 10 to 100 mol%, such as 20 to 90 mol%, such as 40 to 70 mol%, of the above-described cationic lipid based on the total lipid contained in the composition.
- Cationic lipids can be used singly or in combination of two or more.
- Nucleic acids include those similar to those described above.
- the composition of this embodiment contains, for example, 0.01 to 50% by weight, for example, 0.1 to 30% by weight, for example 1 to 10% by weight, of nucleic acid.
- composition of this embodiment contains, as a lipid component, (I) the cationic lipid described above, and (II) at least one lipid selected from the group consisting of neutral lipids, polyethylene glycol-modified lipids, and sterols. To do.
- composition of the present embodiment may contain, for example, 0 to 50 mol%, such as 0 to 40 mol%, such as 0 to 30 mol%, based on the total lipid contained in the composition.
- the composition of the present embodiment may contain, for example, 0 to 30 mol%, such as 0 to 20 mol%, such as 0 to 10 mol%, of polyethylene glycol-modified lipid, based on the total lipid contained in the composition. .
- composition of the present embodiment may contain, for example, 0 to 90 mol%, such as 10 to 80 mol%, such as 20 to 50 mol%, based on the total lipid contained in the composition.
- the combination of lipid components in the composition of the present embodiment is not particularly limited.
- composition of the present embodiment includes, as other additives, sugars such as sucrose, glucose, sorbitol, and lactose; amino acids such as glutamine, glutamic acid, sodium glutamate, and histidine; citric acid, phosphorus Acid salts such as acid, acetic acid, lactic acid, carbonic acid and tartaric acid may be contained.
- sugars such as sucrose, glucose, sorbitol, and lactose
- amino acids such as glutamine, glutamic acid, sodium glutamate, and histidine
- citric acid citric acid
- phosphorus Acid salts such as acid, acetic acid, lactic acid, carbonic acid and tartaric acid may be contained.
- composition of the present embodiment may be formulated as a pharmaceutical composition.
- dosage form of the pharmaceutical composition include injections.
- the composition of the present embodiment may be in a powder state from which the solvent has been removed by freeze drying or the like, or in a liquid state.
- the composition When the composition is in a powder state, it can be suspended or dissolved in a pharmaceutically acceptable medium before use and used as an injection.
- the composition When the composition is in a liquid state, it can be used as an injection as it is or suspended or dissolved in a pharmaceutically acceptable medium.
- compositions of the present embodiment may further contain additives such as solubilizing agents such as ethanol, propylene glycol, polyethylene glycol and the like, stabilizers, antioxidants, preservatives and the like.
- Administration of the composition to a patient can be performed, for example, by intraarterial injection, intravenous injection, subcutaneous injection, or the like.
- the dose of the composition varies depending on the administration subject, target organ, symptom, and administration method.
- the composition of the present embodiment forms a lipid complex in which a nucleic acid is encapsulated in fine particles composed of a lipid containing a cationic lipid.
- the “average particle diameter” of the lipid complex can be calculated by any one of volume average, number average, and Z-average.
- the average particle size (Z-average) of the lipid complex may be, for example, 10 to 1000 nm, such as 30 to 500 nm, such as 30 to 200 nm.
- the composition of the present embodiment preferably has almost no surface charge in an environment of about pH 7.4, such as in blood, from the viewpoint of suppressing non-specific adsorption and immune reaction.
- an environment of about pH 7.4 such as in blood
- the present invention provides a polar organic solvent comprising (I) the above-described cationic lipid and (II) at least one lipid selected from the group consisting of a neutral lipid, a polyethylene glycol-modified lipid, and a sterol.
- a composition comprising: a step (a) of obtaining a mixed solution by mixing an aqueous solution containing (III) an aqueous solution containing a nucleic acid; and a step (b) for reducing the content of a polar organic solvent in the mixed solution.
- a manufacturing method is provided. According to the production method of the present embodiment, a composition capable of efficiently releasing nucleic acid into the cytoplasm can be produced.
- a lipid complex in which nucleic acid is encapsulated in fine particles composed of lipid can be formed by electrostatic interaction between water-soluble nucleic acid and the above-mentioned cationic lipid and hydrophobic interaction between lipids.
- a lipid complex can be formed by changing.
- the polar organic solvent include alcohols such as ethanol.
- step (a) a polar organic in which (I) the above-described cationic lipid and (II) at least one lipid selected from the group consisting of a neutral lipid, a polyethylene glycol-modified lipid, and a sterol are dissolved.
- a solvent-containing aqueous solution and (III) an aqueous solution containing a nucleic acid are mixed to obtain a mixed solution.
- the concentration of the polar organic solvent in the polar organic solvent-containing aqueous solution is not particularly limited as long as it satisfies the conditions for dissolving lipid molecules even after mixing with the nucleic acid aqueous solution.
- the content of the polar organic solvent is reduced by adding water or the like to the above mixed solution.
- a lipid complex can be formed.
- lipid complex in which nucleic acids are efficiently encapsulated inside fine particles can be obtained.
- the composition can be used as a pharmaceutical composition.
- the chemical shift of the proton nuclear magnetic resonance spectrum is recorded in ⁇ units (ppm) relative to tetramethylsilane.
- the abbreviations of the patterns are as follows. s: singlet, d: doublet, t: triplet, q: quartet, dd: double doublet, ddd: double doublet, dt: double triplet, tt: triple triplet, m: multiplet.
- the reaction was confirmed by TLC and quenched with water with the disappearance of the starting material as the end point.
- the precipitated solid was filtered off, and the filtrate was extracted with ethyl acetate, washed with water and brine, and then dried over anhydrous magnesium sulfate.
- the desiccant was removed by filtration, and the filtrate was concentrated under reduced pressure.
- the product was purified by silica gel column chromatography to obtain the fourth step product (1.2 g, 2.7 mmol, 99%).
- Example 2 ((Z) -1- (Non-2-en-1-yloxy) -1-oxononadecan-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-101”) Synthesis)
- YS-101 represented by the following formula (16) was synthesized in the same manner as in Example 1 except that the fifth step was not performed.
- Example 3 Synthesis of 1-oxo-1- (undecan-5-yloxy) nonadecan-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-103”))
- YS-103 2-butyloctane-1-ol is reacted instead of 2-nonenol, and the fifth step is not performed, and is represented by the following formula (2) in the same manner as in Example 1.
- YS-103 was synthesized.
- Example 5 Synthesis of 1-oxo-1- (undecan-5-yloxy) pentadecan-6-yl-1-methylpiperidine 4-carboxylate (hereinafter sometimes referred to as “YS-111”)) Similar to Example 4 except that dimethyl adipate was reacted in place of dimethyl sebacate in the first step and undecan-5-ol was reacted in place of 2-butyloctane-1-ol in the fourth step. Thus, YS-111 represented by the following formula (17) was synthesized.
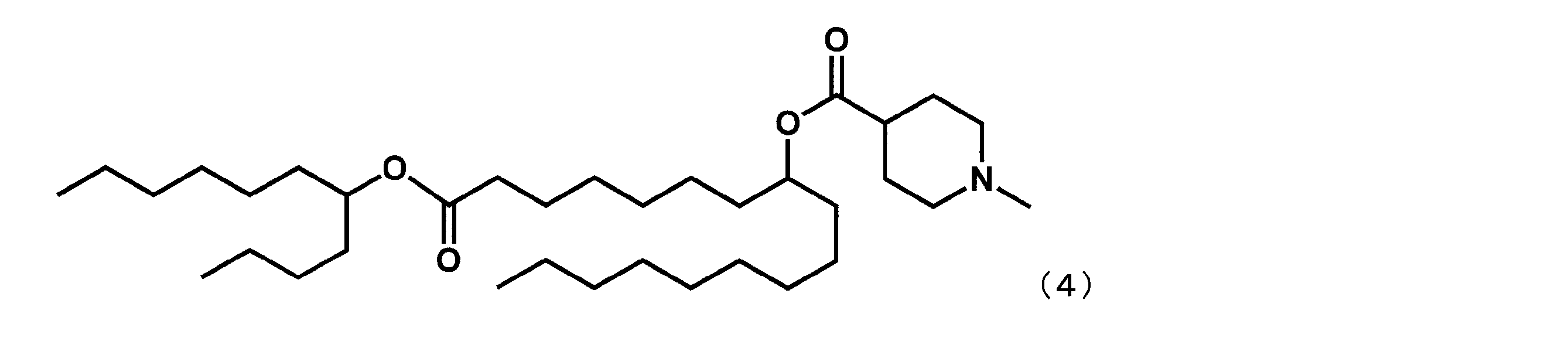
- Example 6 Synthesis of 1-oxo-1- (undecan-5-yloxy) heptadecan-8-yl-1-methylpiperidine 4-carboxylate (hereinafter sometimes referred to as “YS-112”)) Same as Example 4 except that dimethyl suberate was reacted in place of dimethyl sebacate in the first step and undecan-5-ol was reacted in place of 2-butyloctane-1-ol in the fourth step. Thus, YS-112 represented by the following formula (4) was synthesized.
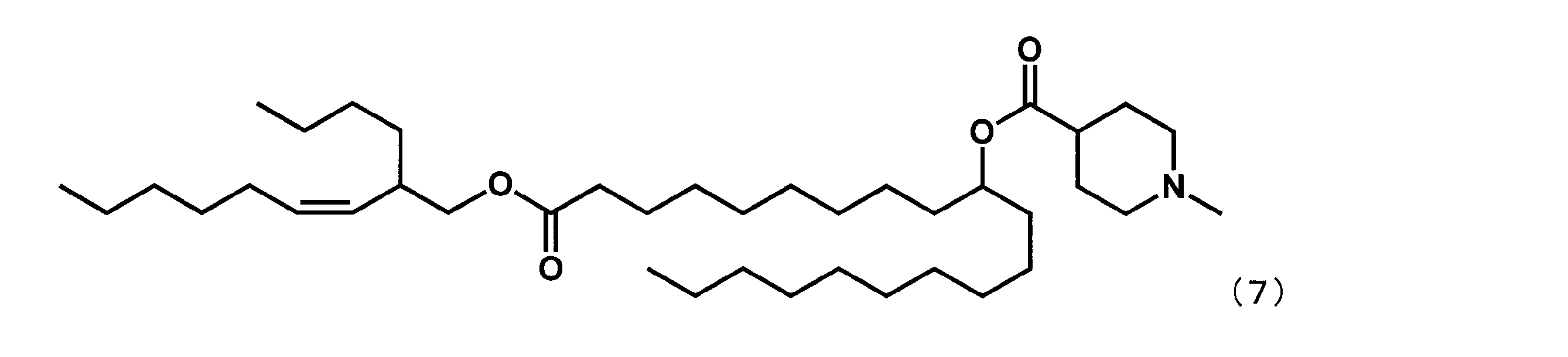
- Example 7 Synthesis of 21-oxo-21- (undecan-5-yloxy) henicosane-10-yl-1-methylpiperidine 4-carboxylate (hereinafter sometimes referred to as “YS-113”))
- Example 4 except that dimethyl dodecanedioate was reacted instead of dimethyl sebacate in the first step, and undecan-5-ol was reacted instead of 2-butyloctane-1-ol in the fourth step.
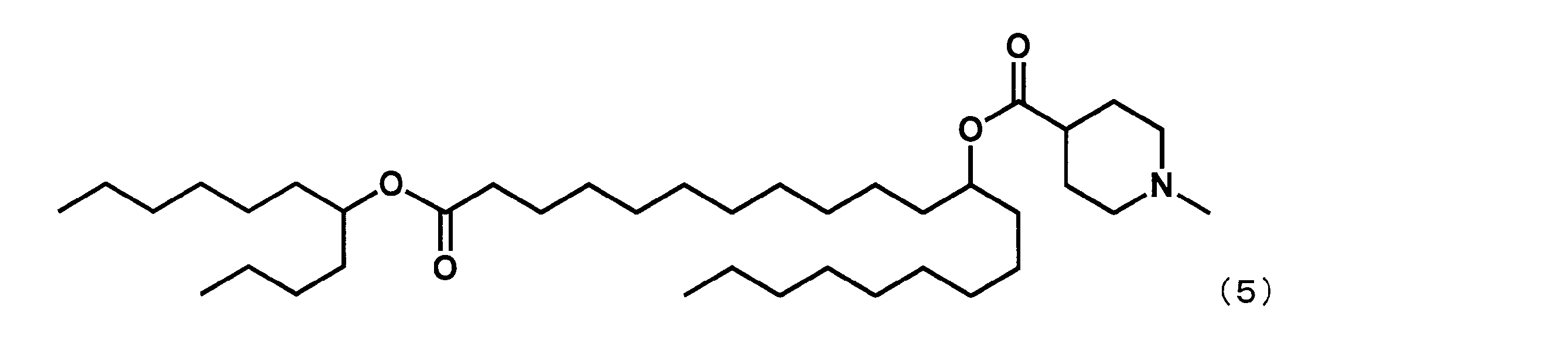
- YS-113 represented by the following formula (5) was synthesized.
- Example 8 Synthesis of 23-oxo-23- (undecan-5-yloxy) tricosan-10-yl-1-methylpiperidine 4-carboxylate (hereinafter sometimes referred to as “YS-114”))
- Example 4 except that dimethyltetradecandioate was reacted in place of dimethyl sebacate in the first step, and undecan-5-ol was reacted in place of 2-butyloctane-1-ol in the fourth step.
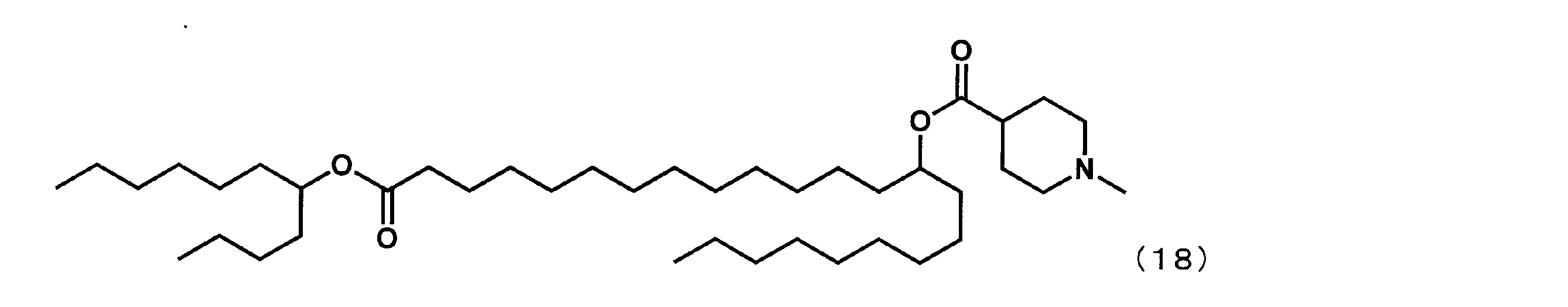
- YS-114 represented by the following formula (18) was synthesized.
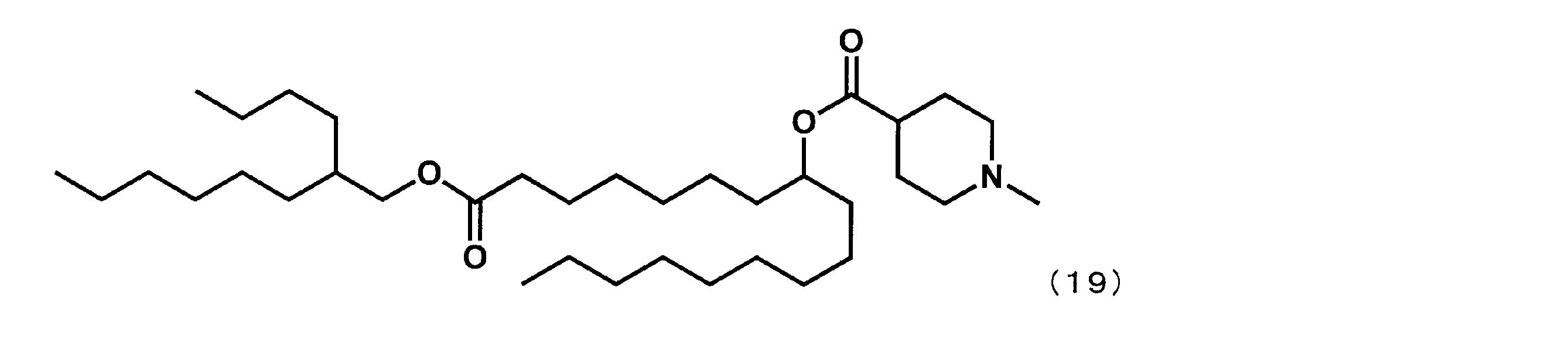
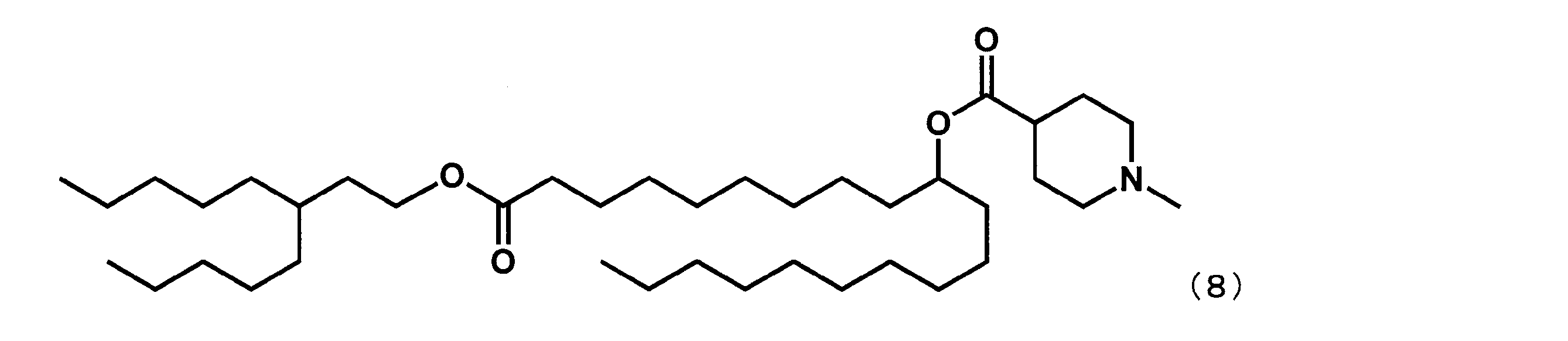
- Example 9 Synthesis of 1-((2-butyloctyl) oxy) -1-oxoheptadecan-8-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-115”)) YS-115 represented by the following formula (19) was synthesized in the same manner as in Example 4 except that dimethyl suberate was reacted instead of dimethyl sebacate in the first step.
- Example 10 Synthesis of 21-((2-butyloctyl) oxy) -21-oxohenicosan-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-116”)) YS-116 represented by the following formula (20) was synthesized in the same manner as in Example 4 except that dimethyl suberate was reacted instead of dimethyl sebacate in the first step.
- Example 11 Synthesis of 1- (octane-3-yloxy) -1-oxoheptadecan-8-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-117”)) Similar to Example 4 except that dimethyl suberate was reacted in place of dimethyl sebacate in the first step and octane-3-ol was reacted in place of 2-butyloctane-1-ol in the fourth step. Thus, YS-117 represented by the following formula (21) was synthesized.
- Example 12 Synthesis of 1-((S) octane-3-yloxy) -1-oxoheptadecan-8-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-117S”)) Performed except that dimethyl suberate was reacted in place of dimethyl sebacate in the first step, and (S) -octane-3-ol was reacted in place of 2-butyloctane-1-ol in the fourth step.
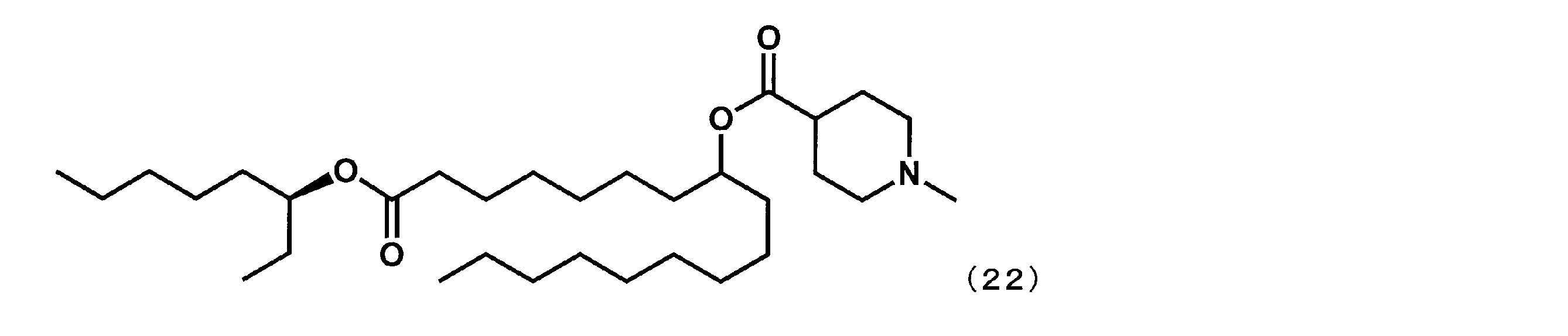
- YS-117S represented by the following formula (22) was synthesized.
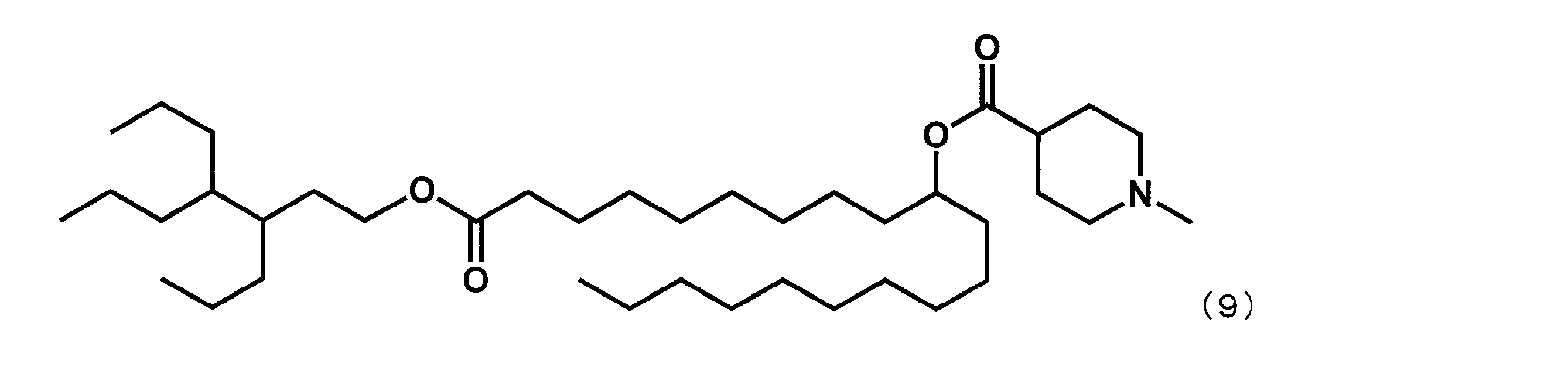
- Example 13 Synthesis of 21- (octane-3-yloxy) -21-oxohenicosan-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-118”))
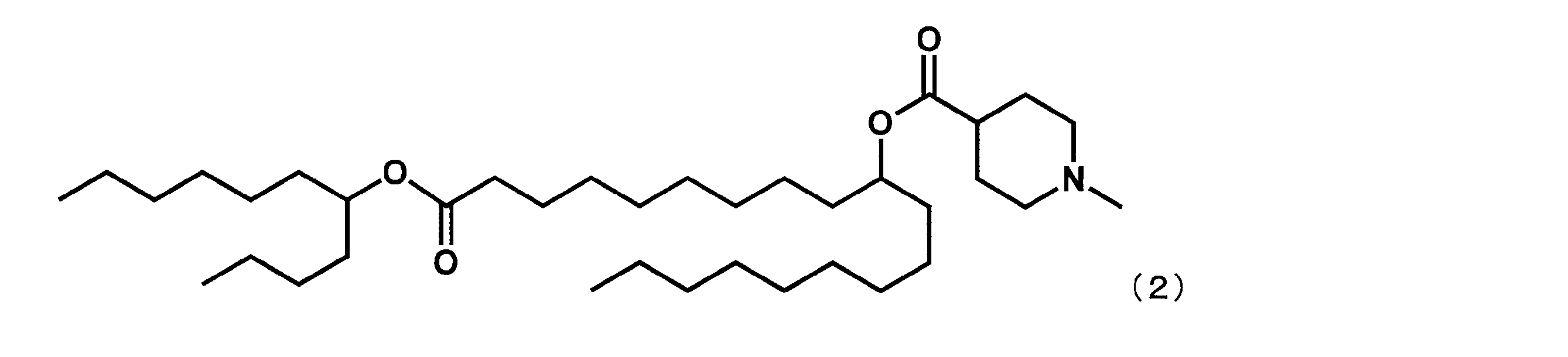
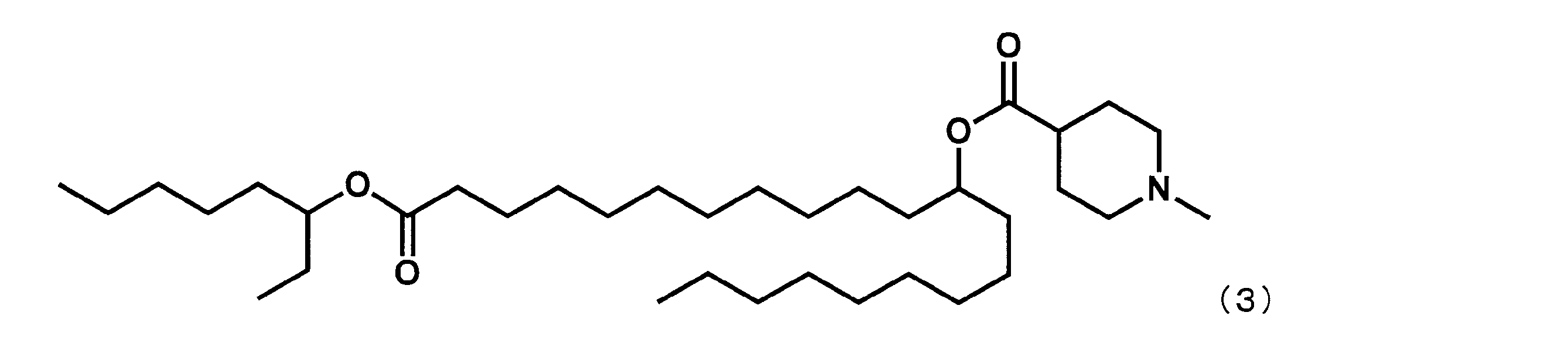
- Example 4 except that dimethyl dodecanedioate was reacted instead of dimethyl sebacate in the first step and octane-3-ol was reacted instead of 2-butyloctane-1-ol in the fourth step.
- YS-118 represented by the following formula (3) was synthesized.
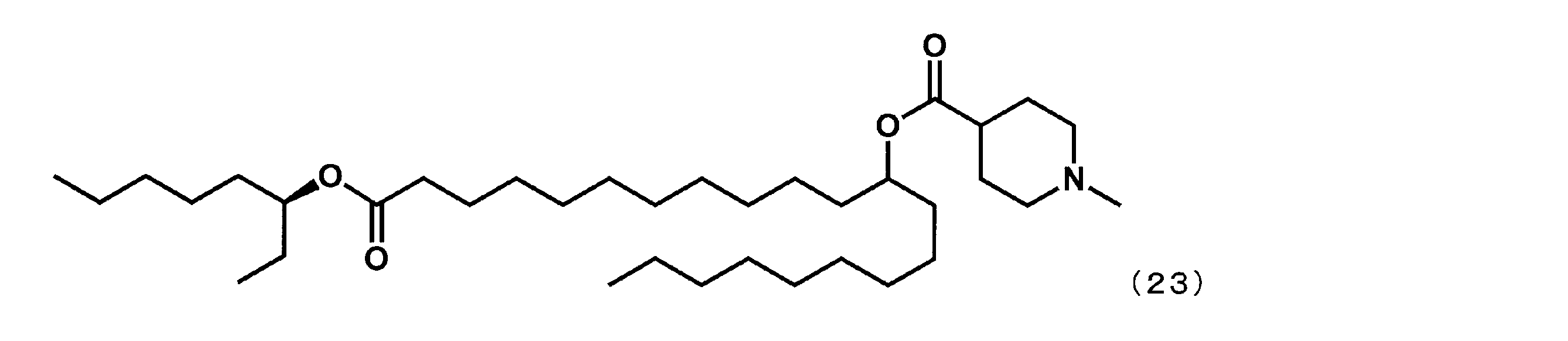
- Example 14 (21-((S) -octane-3-yloxy) -21-oxohenicosan-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-118S”). Synthesis) In the first step, dimethyl dodecanedioate was reacted in place of dimethyl sebacate, and (S) -octane-3-ol was reacted in place of 2-butyloctane-1-ol in the fourth step. In the same manner as in Example 4, YS-118S represented by the following formula (23) was synthesized.
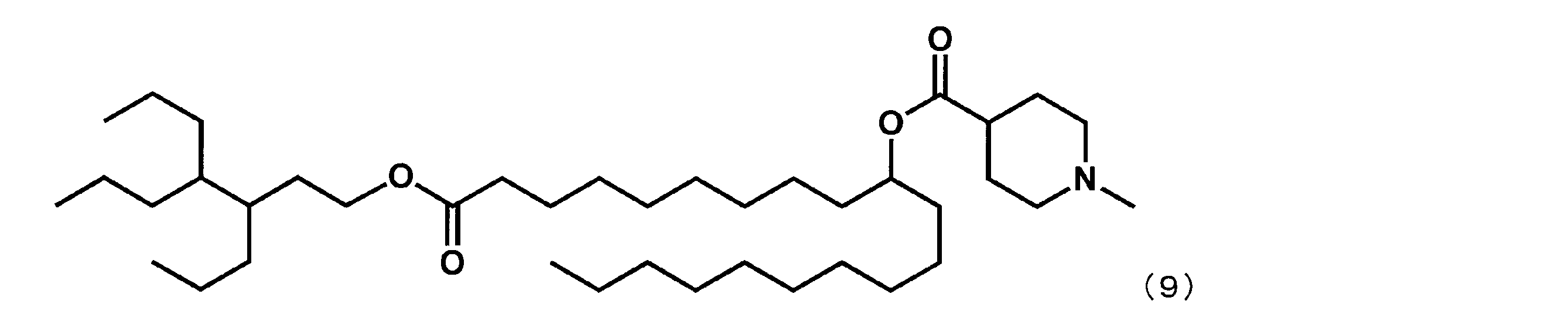
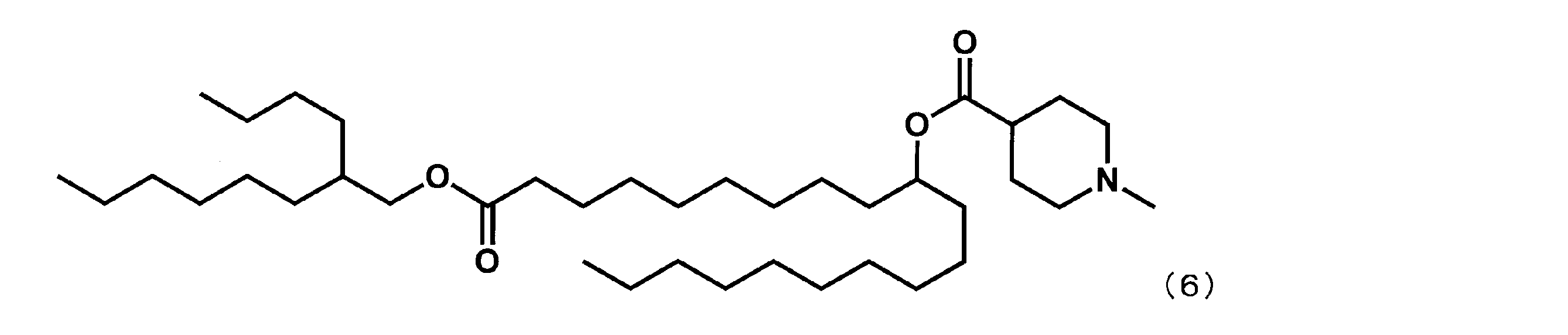
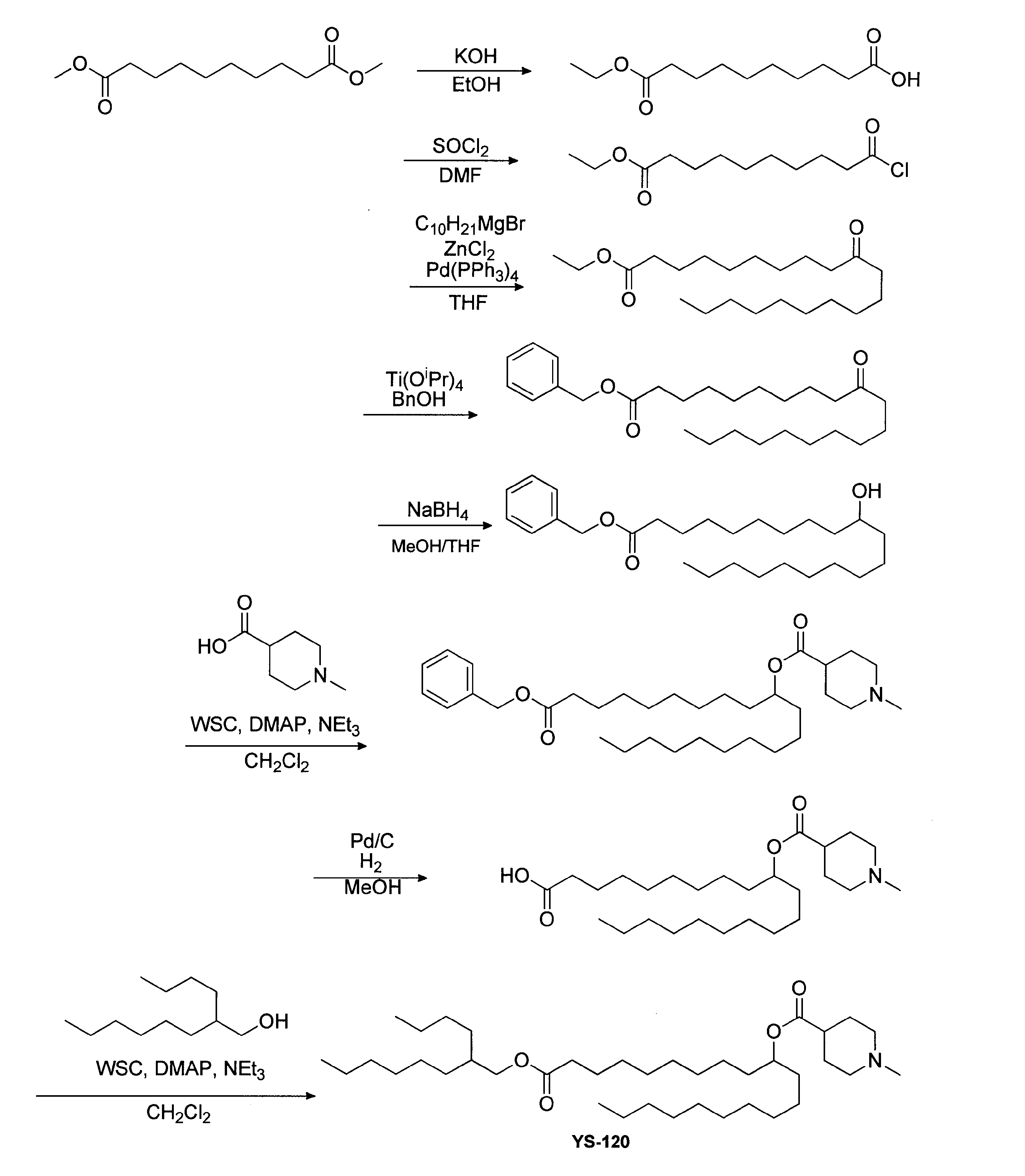
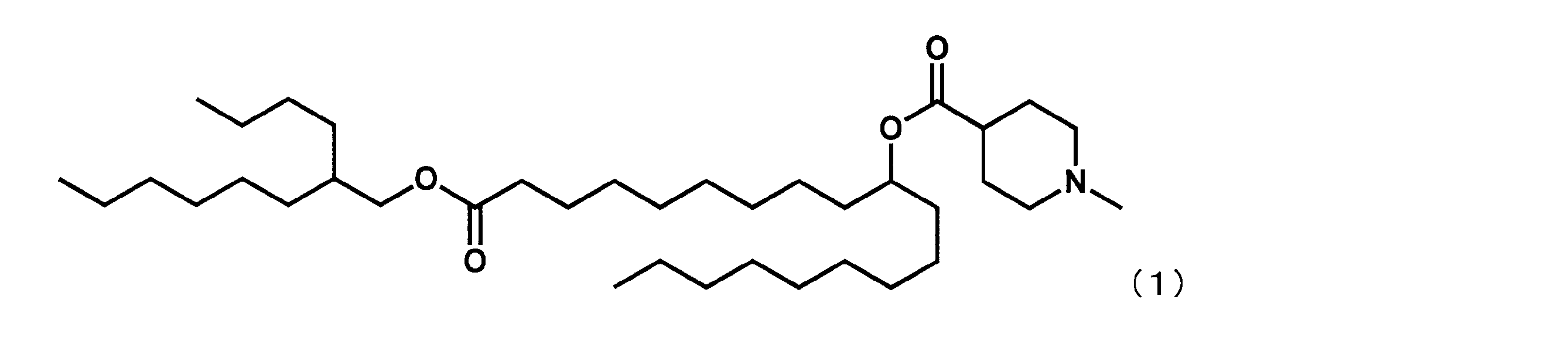
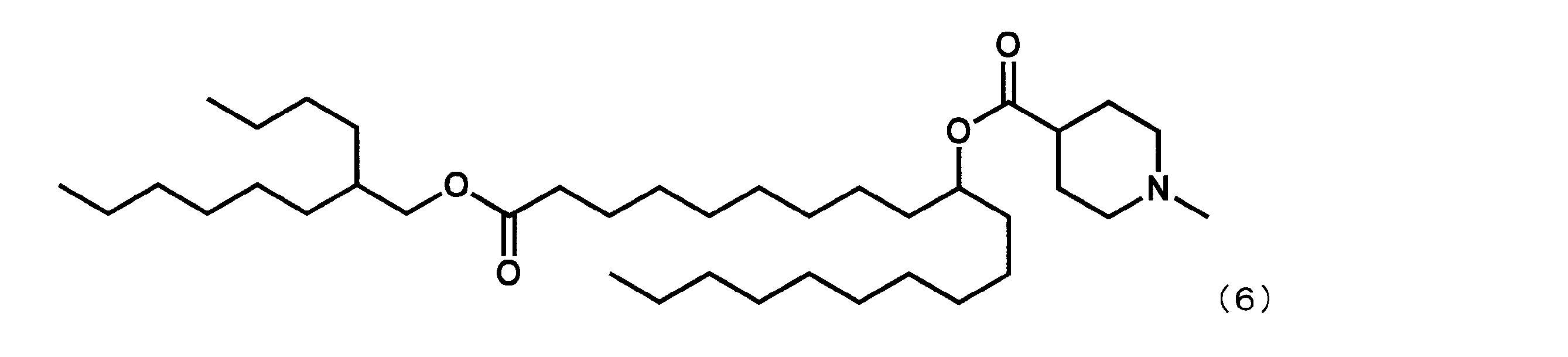
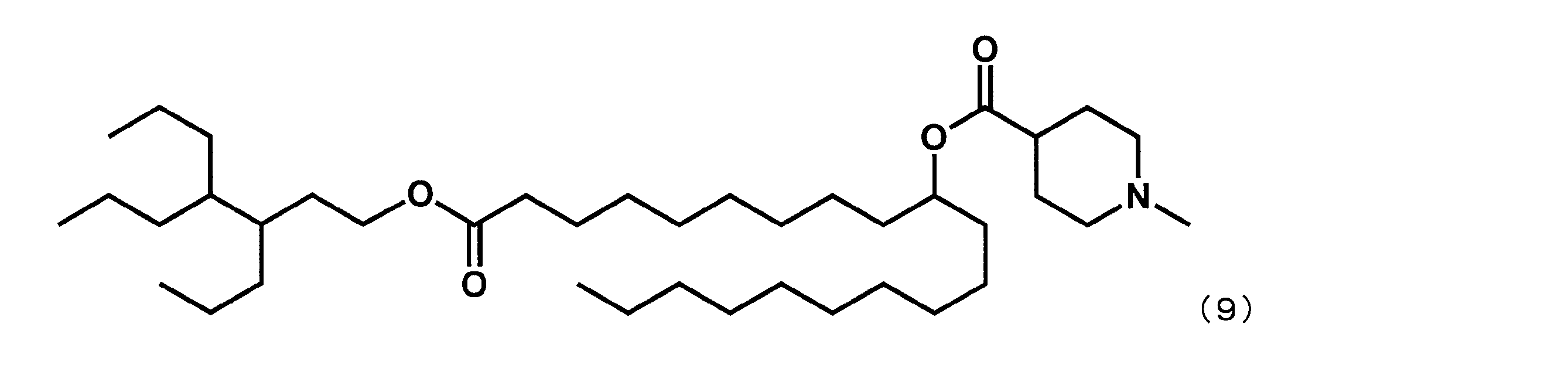
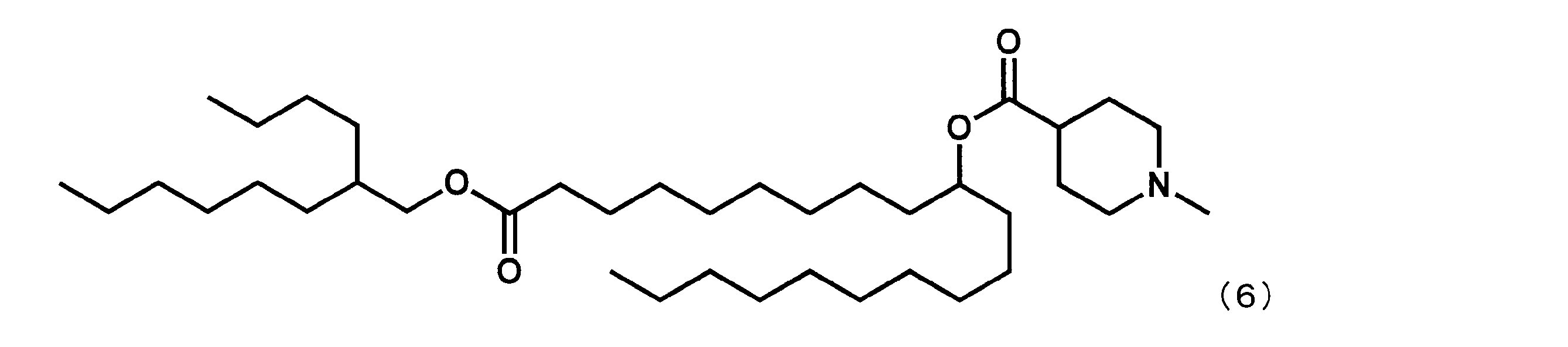
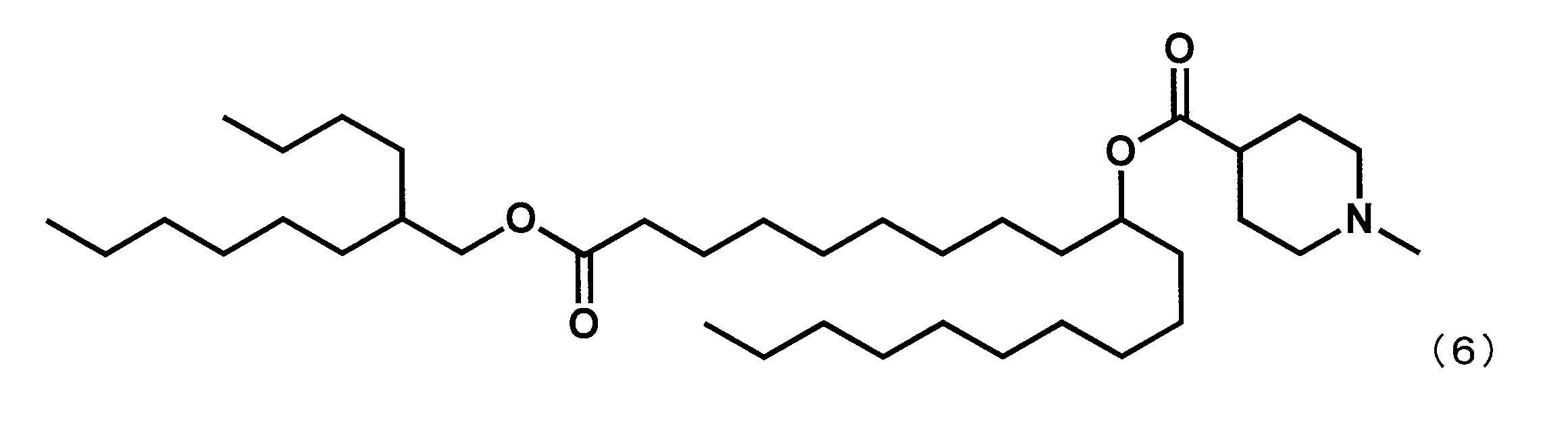
- Example 15 Synthesis of 1-((2-butyloctyl) oxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-120”)) YS-120 represented by the following formula (6) was synthesized in the same manner as in Example 4 except that decanylmagnesium bromide was reacted instead of nonylmagnesium bromide in the third step.
- the sixth step product (37.2 g, 68.4 mmol), palladium / carbon (4.9 mL) was suspended in ethyl acetate (136.8 mL) and stirred overnight under a hydrogen atmosphere. The reaction solution was filtered off and concentrated after removing paradim / carbon. The product was purified by silica gel chromatography to obtain a seventh step product (26.8 g, 59.1 mmol).
- Example 17 (1-((2Z, 5Z) -deca-2,5-dien-1-yloxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter referred to as “YS-121”)
- YS-121 (1-((2Z, 5Z) -deca-2,5-dien-1-yloxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate
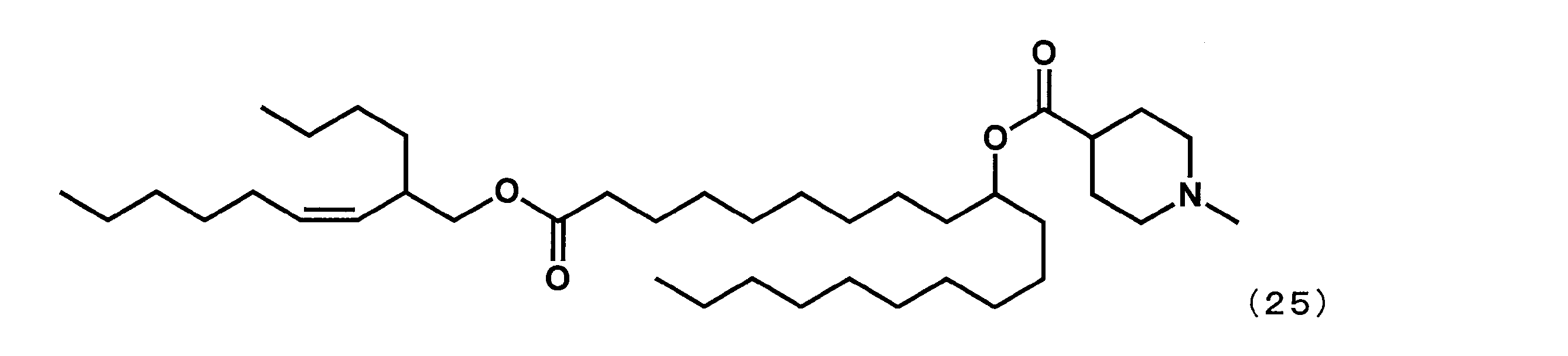
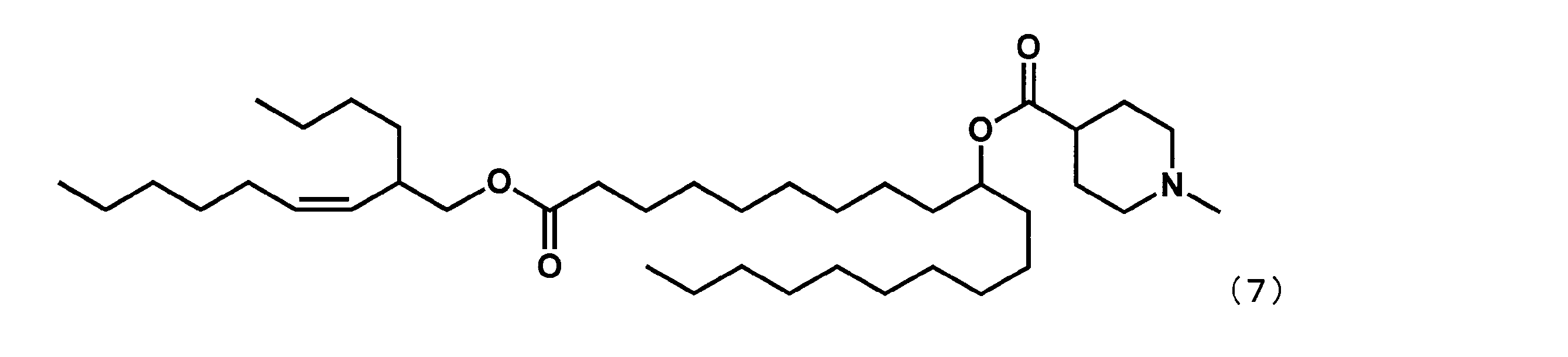
- Example 18 ((Z) -1-((2-Butylnon-3-en-1-yl) oxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter referred to as “YS-122”) May be a combination of))
- YS-122 ((Z) -1-((2-Butylnon-3-en-1-yl) oxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter referred to as “YS-122”) May be a combination of))
- Example 19 ((Z) -1-oxo-1-((5-propylnon-2-en-1-yl) oxy) icosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter referred to as “YS-123 )))))
- YS-123 icosane-10-yl-1-methylpiperidine-4-carboxylate
- the eighth step in the same manner as in Example 16 except that (Z) -5-propylnon-2-en-1-ol was reacted instead of 2-butyloctane-1-ol, the following formula (26 ) was synthesized.
- Example 20 Synthesis of 1-oxo-1-((3-pentyloctyl) oxy) icosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-124”))
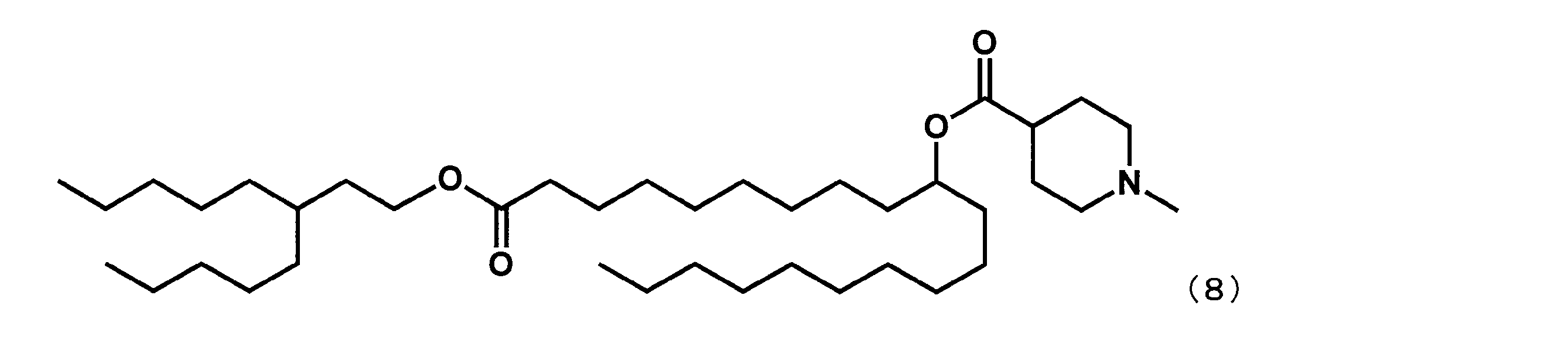
- YS-124 represented by the following formula (27) was obtained in the same manner as in Example 16 except that 3-pentyloctane-1-ol was reacted instead of 2-butyloctane-1-ol. was synthesized.
- Example 21 Synthesis of 1-((2,4-dipropylheptyl) oxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-125”))
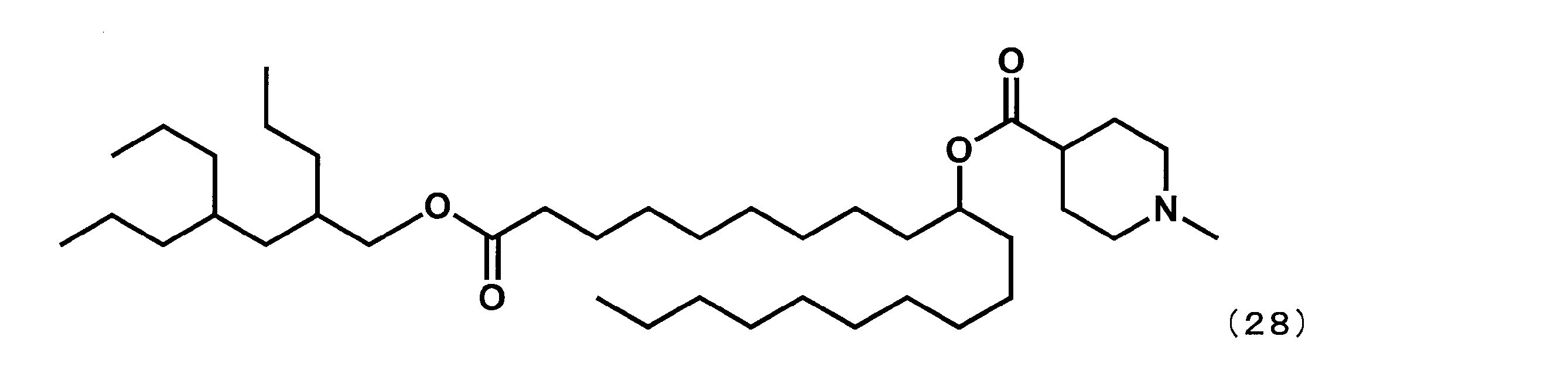
- YS-125 Synthesis of 1-((2,4-dipropylheptyl) oxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-125”))
- YS-125 was synthesized.
- Example 22 Synthesis of 1-((3,4-dipropylheptyl) oxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-126”))
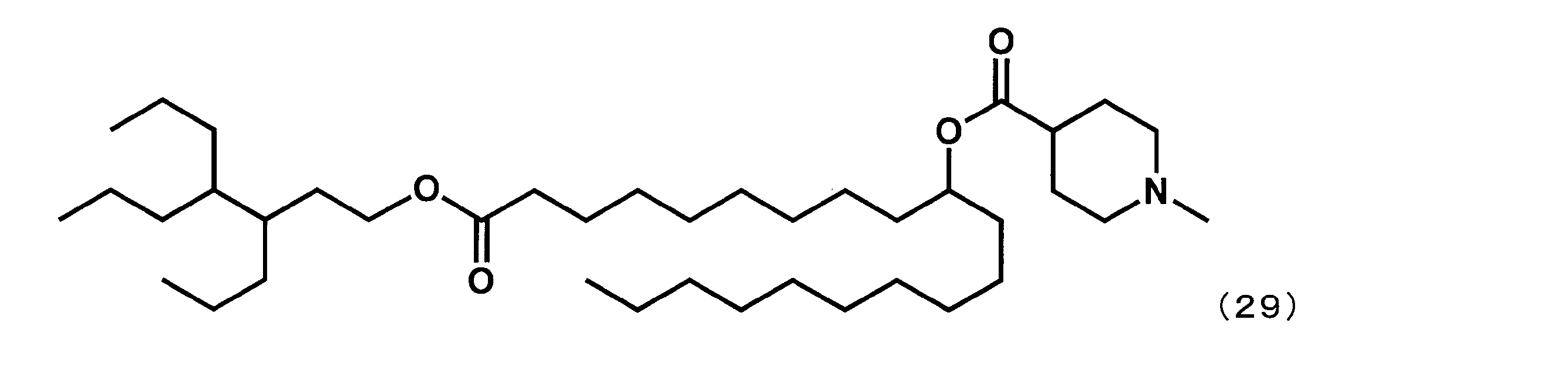
- YS-126 Synthesis of 1-((3,4-dipropylheptyl) oxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-126”))
- YS-126 was synthesized.
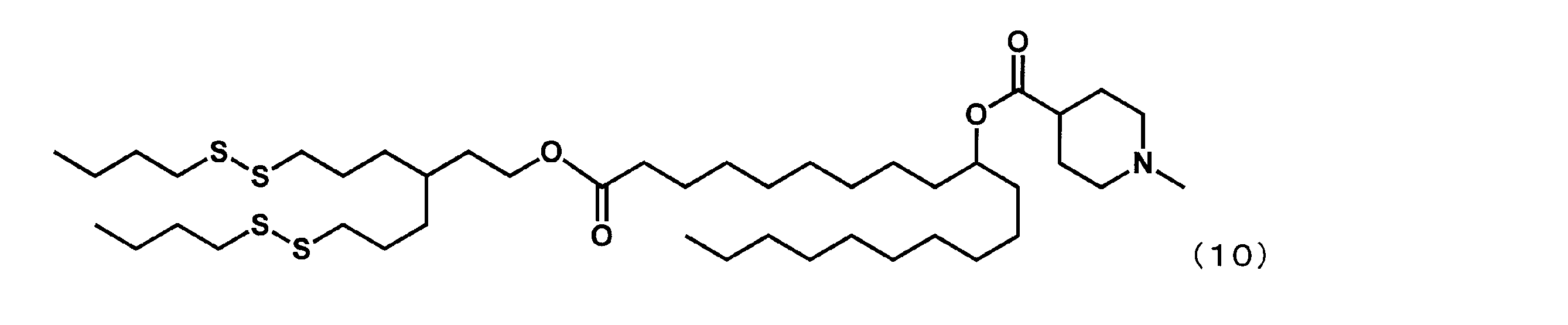
- Example 23 (1- (4- (hexyldisulfanyl) -3-((hexyldisulfanyl) methyl) butoxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate (hereinafter “YS-127”)
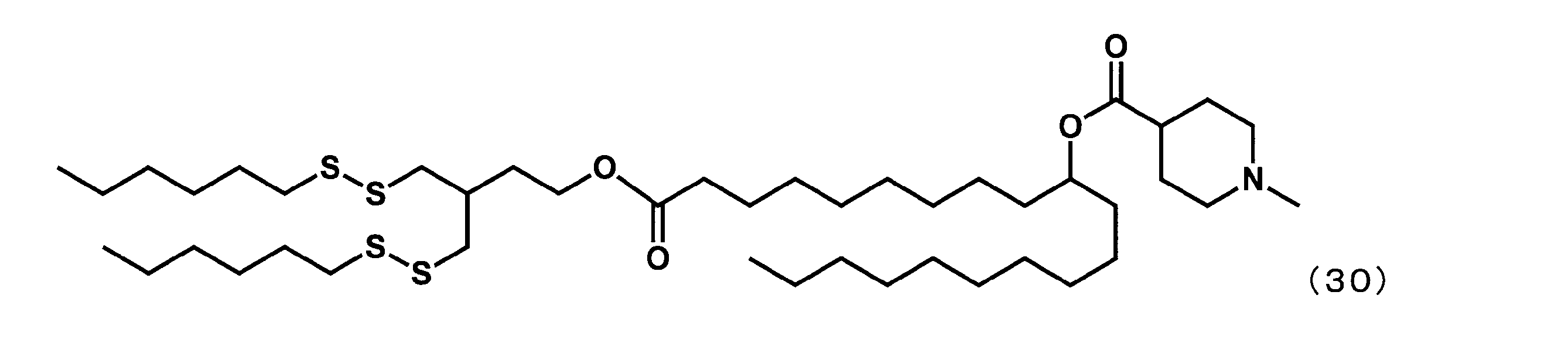
- YS-127 (1- (4- (hexyldisulfanyl) -3-((hexyldisulfanyl) methyl) butoxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate
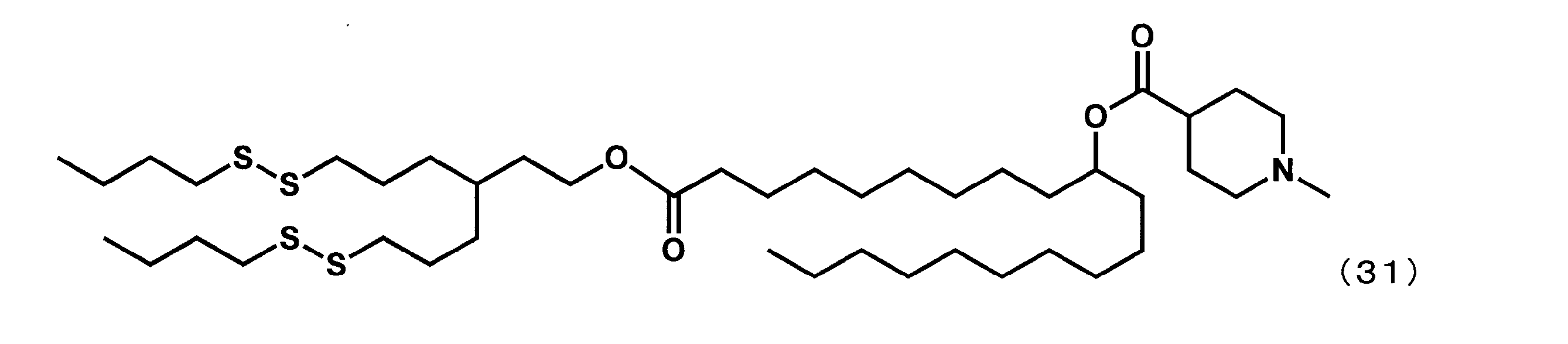
- Example 24 (1-((6- (butyldisulfanyl) -3- (3- (butyldisulfanyl) propyl) hexyl) oxy) -1-oxoicosane-10-yl-1-methylpiperidine-4-carboxylate Sometimes referred to as “YS-128”))
- Example 16 except that in the eighth step, 6- (butyldisulfanyl) -3- (3- (butyldisulfanyl) propyl) hexane-1-ol was reacted instead of 2-butyloctane-1-ol.
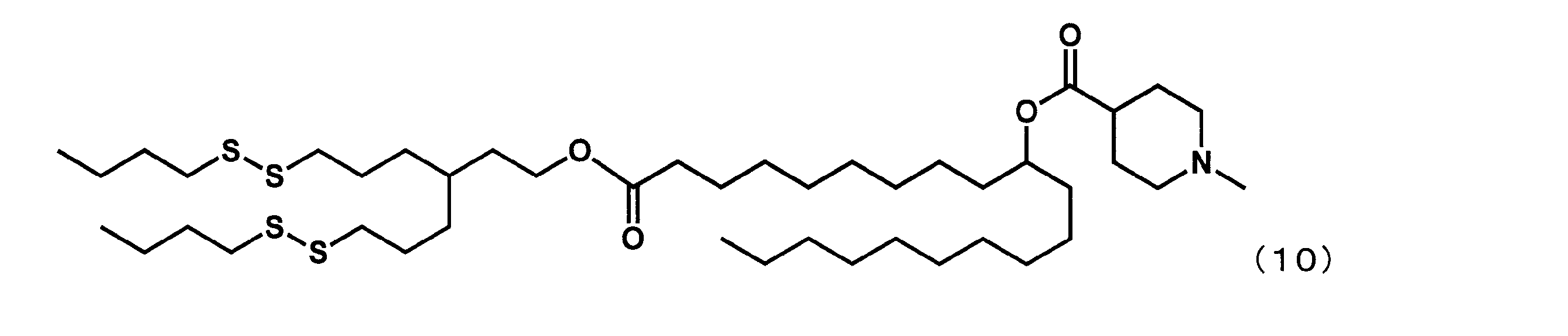
- YS-128 represented by the following formula (31) was synthesized.
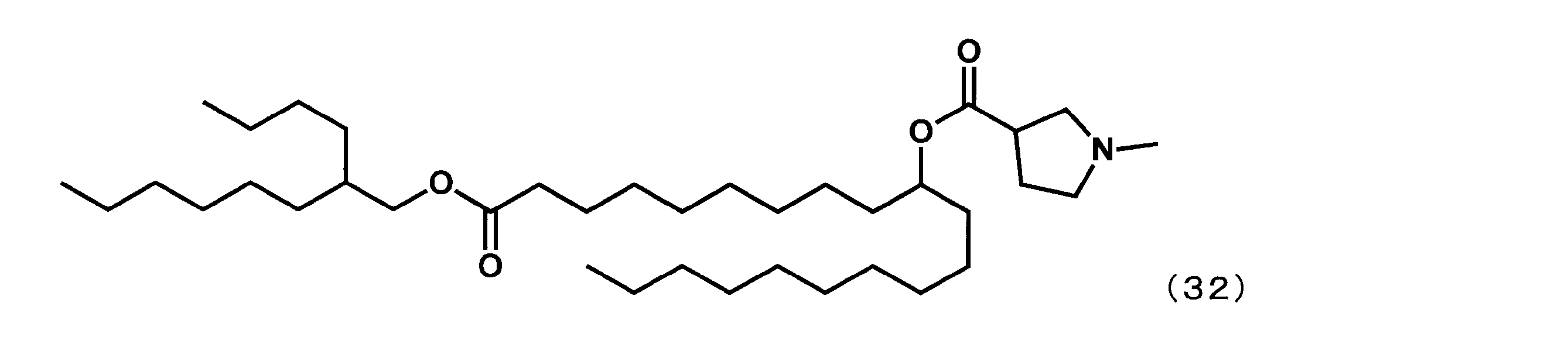
- Example 25 Synthesis of 1-((2-butyloctyl) oxy) -1-oxoicosane-10-yl-1-methylpyrrolidine-3-carboxylate (hereinafter sometimes referred to as “YS-129”))
- YS-129 Synthesis of 1-((2-butyloctyl) oxy) -1-oxoicosane-10-yl-1-methylpyrrolidine-3-carboxylate (hereinafter sometimes referred to as “YS-129”))
- YS-129 was synthesized.
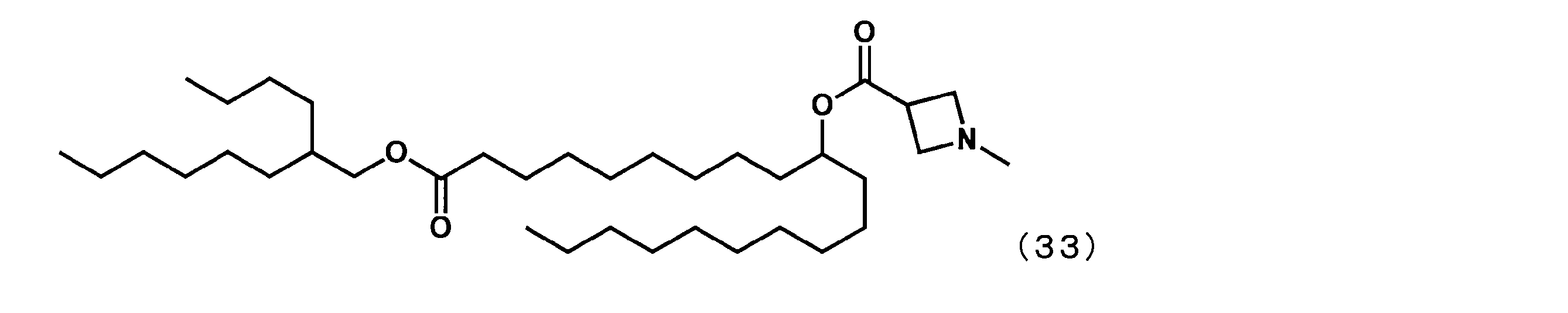
- Example 26 (Synthesis of 1-((2-butyloctyl) oxy) -1-oxoicosane-10-yl-1-methylazetidine-3-carboxylate (hereinafter sometimes referred to as “YS-131”))
- YS-131 is represented by the following formula (33) in the same manner as in Example 16 except that 1-methylazatidine-3-carboxylic acid was reacted instead of 1-methylpiperidine-4-carboxylic acid.
- YS-131 was synthesized.
- Example 27 (Synthesis of 1-((2-butyloctyl) oxy) -1-oxoicosane-10-yl-1-ethylpiperidine-4-carboxylate (hereinafter sometimes referred to as “YS-132”))
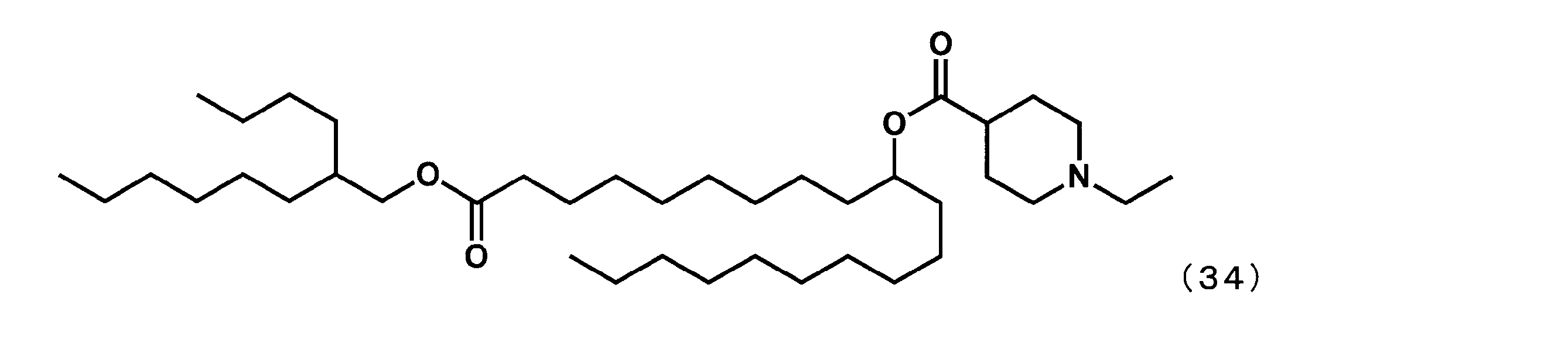
- the sixth step is represented by the following formula (34) in the same manner as in Example 16 except that 1-ethylpiperidine-4-carboxylic acid is reacted instead of 1-methylpiperidine-4-carboxylic acid.
- YS-132 was synthesized.
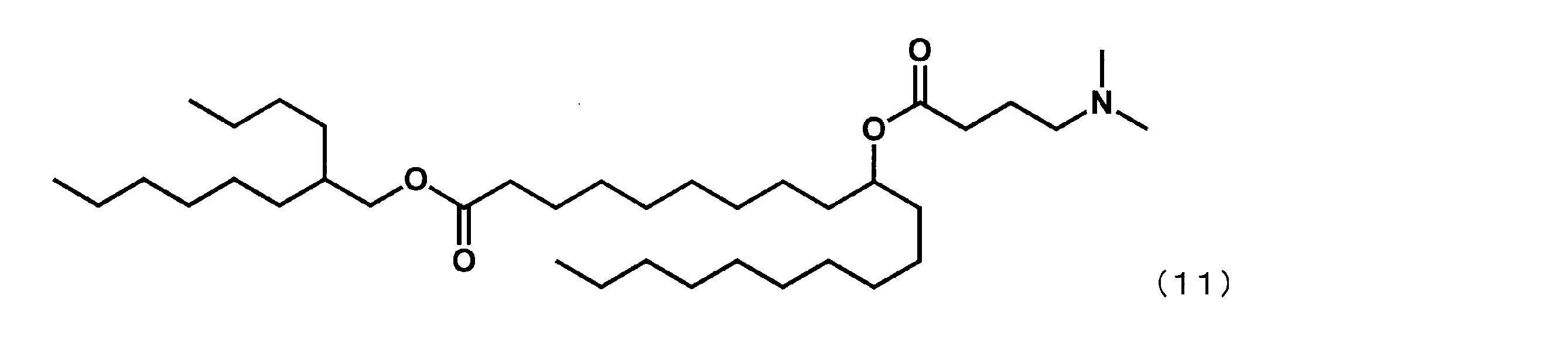
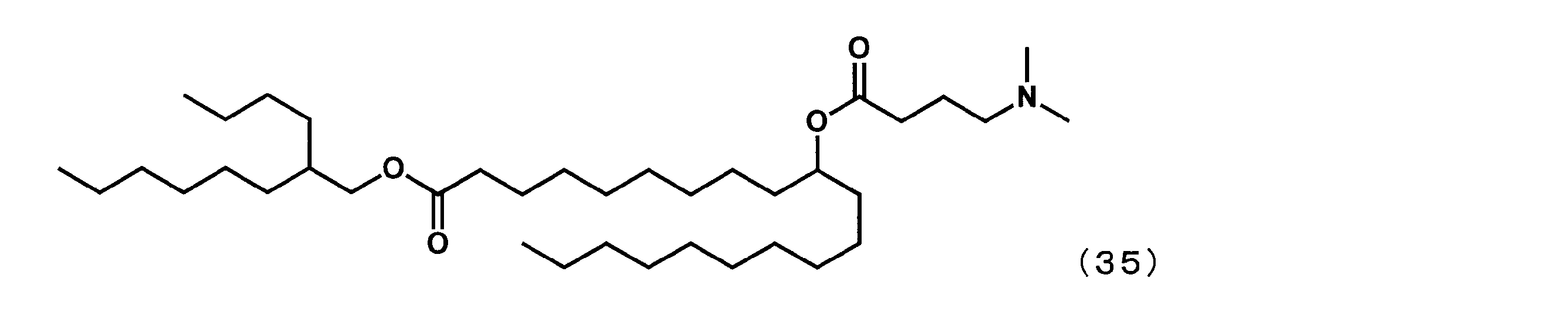
- Example 28 Synthesis of 2-butyloctyl-10-((4- (dimethylamino) butanoyl) oxy) icosanoate (hereinafter sometimes referred to as “YS-133”))
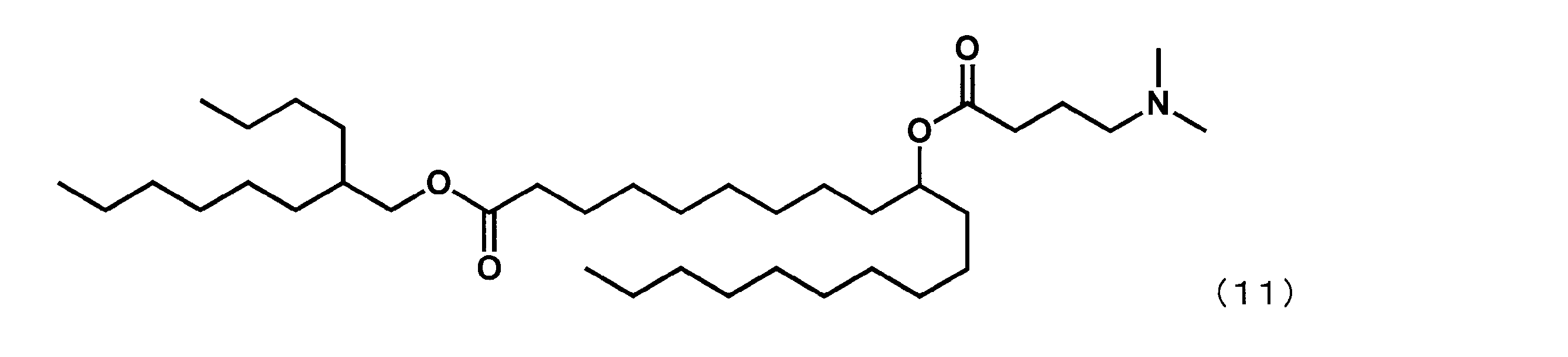
- YS represented by the following formula (35) was obtained in the same manner as in Example 16 except that 4- (dimethylamino) butanoic acid was reacted instead of 1-methylpiperidine-4-carboxylic acid.
- -133 was synthesized.
- composition (1) [Example 29] (YS-102) A composition was prepared using the cationic lipid of Example 1 (YS-102).
- cationic lipid YS-102
- DSPC Nippon Seika Co., Ltd.
- Cholesterol Nippon Seika Co., Ltd.
- MPEG2000-DMG Nippon Oil Co., Ltd.
- Example 29 By mixing siRNA dilution and lipid solution at a flow rate of 2.4 mL / min and 1.29 mL / min, respectively, and further mixing 25 mM sodium acetate (pH 4.0) at a flow rate of 9.25 mL / min, A lipid complex aqueous solution was obtained.
- the obtained lipid complex aqueous solution is subjected to dialysis using a dialysis membrane (trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO), and the external solution is phosphate buffer (PBS, pH 7.5). Replaced with After dialysis, concentration and filter sterilization were performed to obtain the composition of Example 29.
- a dialysis membrane trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO
- PBS phosphate buffer
- Example 30 A composition of Example 30 was obtained in the same manner as in Example 29 except that the cationic lipid (YS-101) of Example 2 was used instead of YS-102 as the cationic lipid.
- Example 31 (YS-103) A composition of Example 31 was obtained in the same manner as in Example 29 except that the cationic lipid (YS-103) of Example 3 was used instead of YS-102 as the cationic lipid.
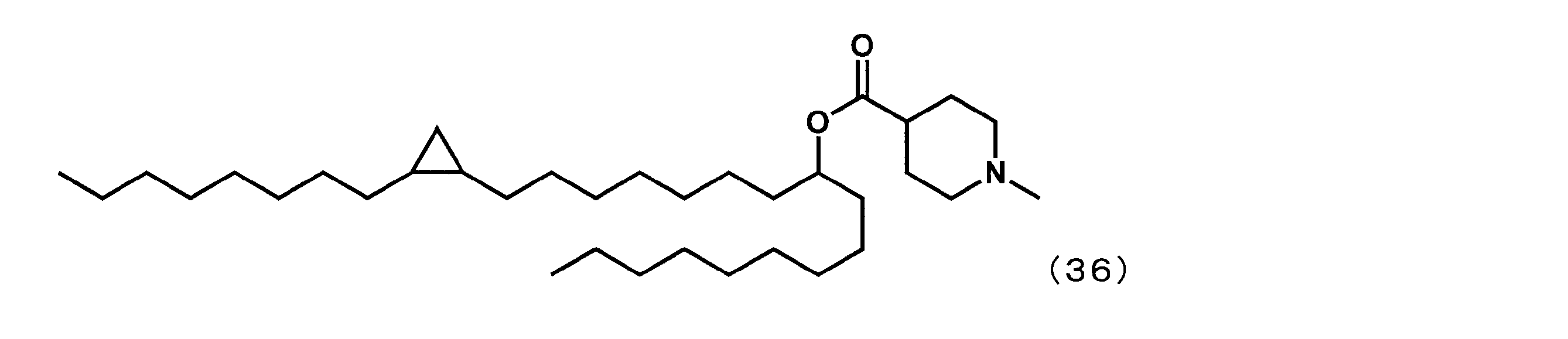
- Reference Example 1 As a cationic lipid, 1- (2-octylcyclopropyl) heptadecan-8-yl-1-methylpiperidine-4-carboxylate (hereinafter referred to as “YS-”) represented by the following formula (36) instead of YS-102:
- YS- 1- (2-octylcyclopropyl) heptadecan-8-yl-1-methylpiperidine-4-carboxylate
- composition (1) ⁇ Analysis of composition (1)>
- the encapsulation rate of siRNA in the lipid complex was measured.
- the composition was diluted with RNase Free Water, and the siRNA concentration (A) measured using Quant-iT RiboGreen RNA Reagent (Invitrogen) was defined as siRNA present in the lipid complex external solution.
- the nucleic acid encapsulation rate was calculated by the following formula (F1).
- Encapsulation rate (%) 100 ⁇ (A / B) ⁇ 100 (F1)
- the average particle size of the lipid complex was measured with a particle size measuring device (trade name “Zetasizer Nano ZS”, manufactured by Malvern).
- Table 1 shows the siRNA encapsulation rate and the average particle size (Z-average) of the lipid complex.
- composition (2) A composition was prepared using the cationic lipid of Example 5 (YS-111). As the nucleic acid, the same Factor VII siRNA as that used in the composition of Example 29 was used.
- RNA diluted solution was prepared by cationic lipid (YS-111), DSPC (Nippon Seika Co., Ltd.), Cholesterol (Nippon Seika Co., Ltd.), MPEG2000-DMG (Nippon Oil Co., Ltd.) can be added to 60 / 8.5 / 30 / 1.5.
- a lipid solution was prepared by dissolving in ethanol so that the total lipid concentration was 10 mM at a molar ratio.
- Example 32 By mixing the siRNA dilution and the lipid solution at a flow rate of 2.4 mL / min and 1.29 mL / min, respectively, and further mixing 25 mM sodium acetate (pH 4.0) at a flow rate of 5.0 mL / min, A lipid complex aqueous solution was obtained.
- the obtained lipid complex aqueous solution is subjected to dialysis using a dialysis membrane (trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO), and the external solution is phosphate buffer (PBS, pH 7.5). Replaced with After dialysis, concentration and filter sterilization were performed to obtain the composition of Example 32.
- a dialysis membrane trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO
- PBS phosphate buffer
- Example 33 (YS-112) A composition of Example 33 was obtained in the same manner as in Example 32 except that the cationic lipid (YS-112) of Example 6 was used instead of YS-111 as the cationic lipid.
- Example 34 (YS-113) A composition of Example 34 was obtained in the same manner as in Example 32 except that the cationic lipid (YS-113) of Example 7 was used instead of YS-111 as the cationic lipid.
- Example 35 (YS-114) A composition of Example 35 was obtained in the same manner as in Example 32 except that the cationic lipid (YS-114) of Example 8 was used instead of YS-111 as the cationic lipid.
- Example 36 (YS-115) A composition of Example 36 was obtained in the same manner as in Example 32 except that the cationic lipid (YS-115) of Example 9 was used instead of YS-111 as the cationic lipid.
- Example 37 (YS-116) A composition of Example 36 was obtained in the same manner as in Example 32 except that the cationic lipid (YS-116) of Example 10 was used instead of YS-111 as the cationic lipid.
- composition (3) was prepared using the cationic lipid of Example 11 (YS-117).
- the same Factor VII siRNA as that used in the composition of Example 29 was used.
- cationic lipid YS-111
- DSPC Nippon Seika Co., Ltd.
- Cholesterol Nippon Seika Co., Ltd.
- MPEG2000-DMG Nippon Oil Co., Ltd.
- lipid complex aqueous solution was obtained.
- the obtained lipid complex aqueous solution is subjected to dialysis using a dialysis membrane (trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO), and the external solution is phosphate buffer (PBS, pH 7.5). Replaced with After dialysis, concentration and filter sterilization were performed to obtain the composition of Example 38.
- Example 39 (YS-117S) A composition of Example 39 was obtained in the same manner as in Example 38 except that the cationic lipid (YS-117S) of Example 12 was used instead of YS-117 as the cationic lipid.
- Example 40 (YS-118) A composition of Example 40 was obtained in the same manner as Example 38, except that the cationic lipid (YS-118) of Example 13 was used instead of YS-117 as the cationic lipid.
- Example 41 (YS-118S) A composition of Example 41 was obtained in the same manner as Example 38 except that the cationic lipid (YS-118S) of Example 14 was used instead of YS-117 as the cationic lipid.
- Example 42 (YS-119) A composition of Example 42 was obtained in the same manner as Example 38 except that the cationic lipid (YS-119) of Example 4 was used instead of YS-117 as the cationic lipid.
- composition (3) ⁇ Analysis of composition (3)> In the same manner as the composition of Example 29, the encapsulation rate of siRNA in the lipid complex and the average particle size of the lipid complex were measured for the compositions of Examples 38 to 42. Table 3 shows the siRNA encapsulation rate and the average particle size (Z-average) of the lipid complexes.
- composition (4) was prepared using the cationic lipid of Example 4 (YS-119).
- the same Factor VII siRNA as that used in the composition of Example 29 was used.
- cationic lipid (YS-119), DSPC (Nippon Seika Co., Ltd.), Cholesterol (Nippon Seika Co., Ltd.), MPEG2000-DMG (Nippon Oil Co., Ltd.) can be added to 60 / 8.5 / 30 / 1.5. (Molar ratio) was dissolved in ethanol so that the total lipid concentration was 6 mM to obtain a lipid solution.
- Example 43 By mixing the siRNA diluted solution and the lipid solution at flow rates of 1.80 mL / min and 0.97 mL / min, respectively, and further mixing 25 mM sodium acetate (pH 4.0) at a flow rate of 6.94 mL / min, A lipid complex aqueous solution was obtained.
- the obtained lipid complex aqueous solution is subjected to dialysis using a dialysis membrane (trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO), and the external solution is phosphate buffer (PBS, pH 7.5). Replaced with After dialysis, concentration and filter sterilization were performed to obtain the composition of Example 43.
- a dialysis membrane trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO
- PBS phosphate buffer
- Comparative Example 2 (ALN-319) A composition of Comparative Example 2 was obtained in the same manner as in Example 43 except that ALN-319 was used instead of YS-119 as the cationic lipid.
- composition (5) A composition was prepared using the cationic lipid of Example 4 (YS-119). As the nucleic acid, the same Factor VII siRNA as that used in the composition of Example 29 was used.
- RNA diluted solution 216 ⁇ g / mL with 25 mM sodium acetate (pH 4.0) to prepare a siRNA diluted solution. Further, cationic lipid (YS-119), Cholesterol (Nippon Seika Co., Ltd.) and MPEG2000-DPG (Nippon Oil Co., Ltd.) were added at a ratio of 60 / 38.5 / 1.5 (molar ratio) to a total lipid concentration of 6 mM. Then, it was dissolved in ethanol to obtain a lipid solution.
- cationic lipid YS-119
- Cholesterol Cholesterol
- MPEG2000-DPG Natural Oil Co., Ltd.
- Example 44 By mixing the siRNA dilution and the lipid solution at flow rates of 3.36 mL / min and 1.81 mL / min, respectively, and further mixing 25 mM sodium acetate (pH 4.0) at a flow rate of 12.95 mL / min, A lipid complex aqueous solution was obtained.
- the obtained lipid complex aqueous solution is subjected to dialysis using a dialysis membrane (trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO), and the external solution is phosphate buffer (PBS, pH 7.5). Replaced with After dialysis, concentration and filter sterilization were performed to obtain the composition of Example 44.
- a dialysis membrane trade name “Float-A-Lyser G2,” SPECTRUM, 50K MWCO
- PBS phosphate buffer
- Example 45 (YS-120) A composition of Example 45 was obtained in the same manner as in Example 44 except that the cationic lipid (YS-120) of Example 15 was used instead of YS-119 as the cationic lipid.
- composition (6) A composition was prepared using the cationic lipid of Example 16 (YS-120). As the nucleic acid, the same Factor VII siRNA as that used in the composition of Example 29 was used.
- RNA diluted solution was diluted in 450 ⁇ g / mL with 25 mM sodium acetate (pH 4.0) to prepare a siRNA diluted solution.
- cationic lipid YS-120
- DSPC Nippon Seika Co., Ltd.
- Cholesterol Nippon Seika Co., Ltd.
- MPEG2000-DMG Nippon Oil Co., Ltd.
- a lipid solution was prepared by dissolving in ethanol so that the total lipid concentration was 40 mM at a molar ratio.
- the ratio of lipid to siRNA was 0.06, and the siRNA dilution and lipid solution were mixed at a flow rate of 4.0 mL / min and 1.3 mL / min, respectively, to obtain a lipid complex aqueous solution.
- the obtained lipid complex aqueous solution is subjected to dialysis using a dialysis membrane (trade name “Float-A-Lyzer G2,” SPECTRUM, 50K MWCO), and the external solution is phosphate buffer (PBS, pH 7.4).
- concentration and filter sterilization were performed to obtain the composition of Example 46.
- Example 47 (YS-121) A composition of Example 47 was obtained in the same manner as in Example 46 except that the cationic lipid (YS-121) of Example 17 was used instead of YS-120 as the cationic lipid.
- Example 48 (YS-122) A composition of Example 48 was obtained in the same manner as in Example 46 except that the cationic lipid (YS-122) of Example 18 was used instead of YS-120 as the cationic lipid.
- Example 49 (YS-123) A composition of Example 49 was obtained in the same manner as in Example 46 except that the cationic lipid (YS-123) of Example 19 was used instead of YS-120 as the cationic lipid.
- Example 50 A composition of Example 50 was obtained in the same manner as in Example 46 except that the cationic lipid (YS-124) of Example 20 was used instead of YS-120 as the cationic lipid.
- Example 51 (YS-125) A composition of Example 51 was obtained in the same manner as in Example 46 except that the cationic lipid (YS-125) of Example 21 was used instead of YS-120 as the cationic lipid.
- Example 52 (YS-126) A composition of Example 52 was obtained in the same manner as in Example 46 except that the cationic lipid (YS-126) of Example 22 was used instead of YS-120 as the cationic lipid.
- compositions of Examples 46 to 52 were stored in sealed vials at 4 ° C., and the particle sizes (Z-average and polydispersity index) before storage, 3 months after storage, and 6 months were measured as Example 29. The measurement was performed in the same manner as the composition of
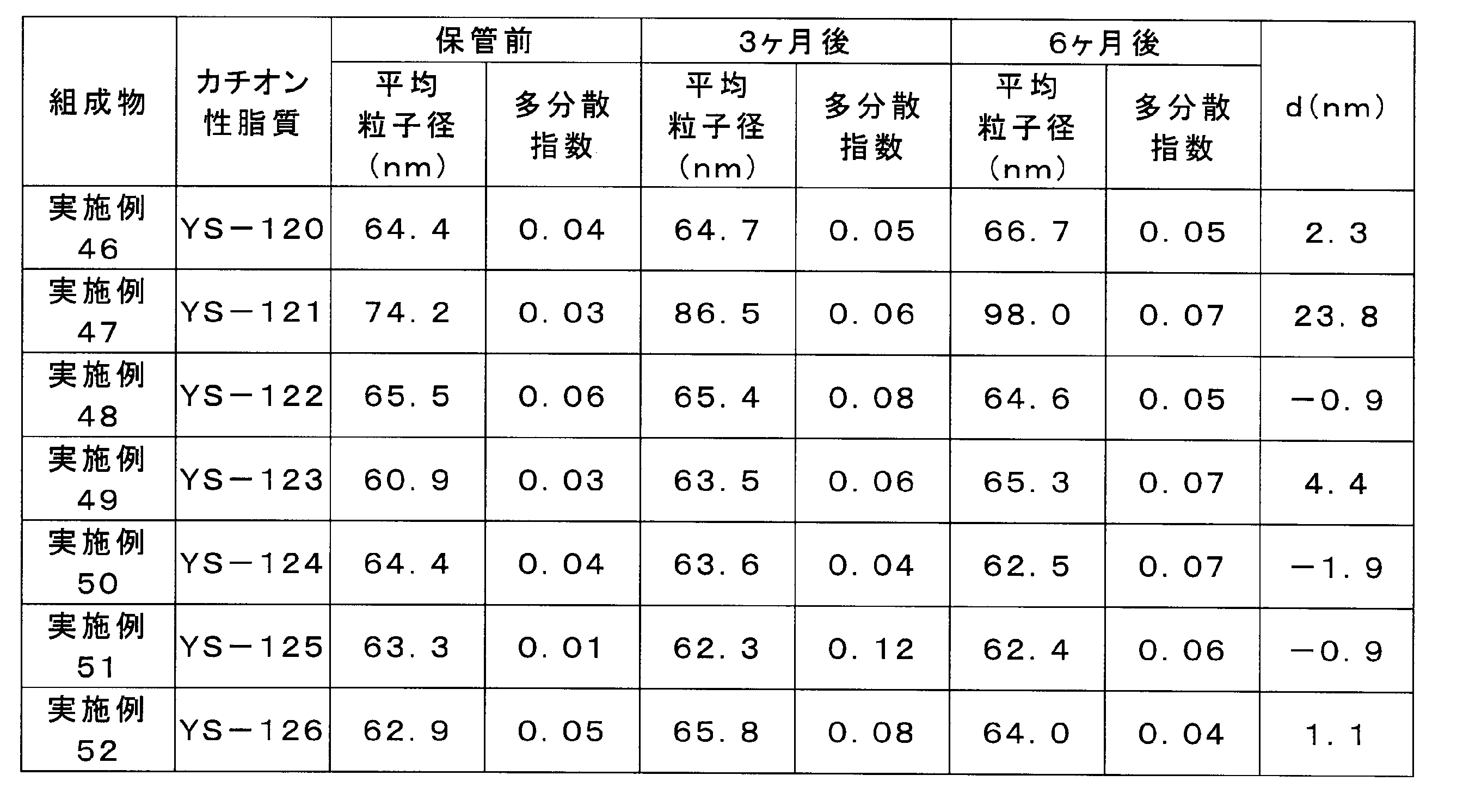
- Table 7 shows the change over time in the average particle diameter of the compositions of Examples 46 to 52, the sixth month of storage, and the change in particle diameter before storage (d).
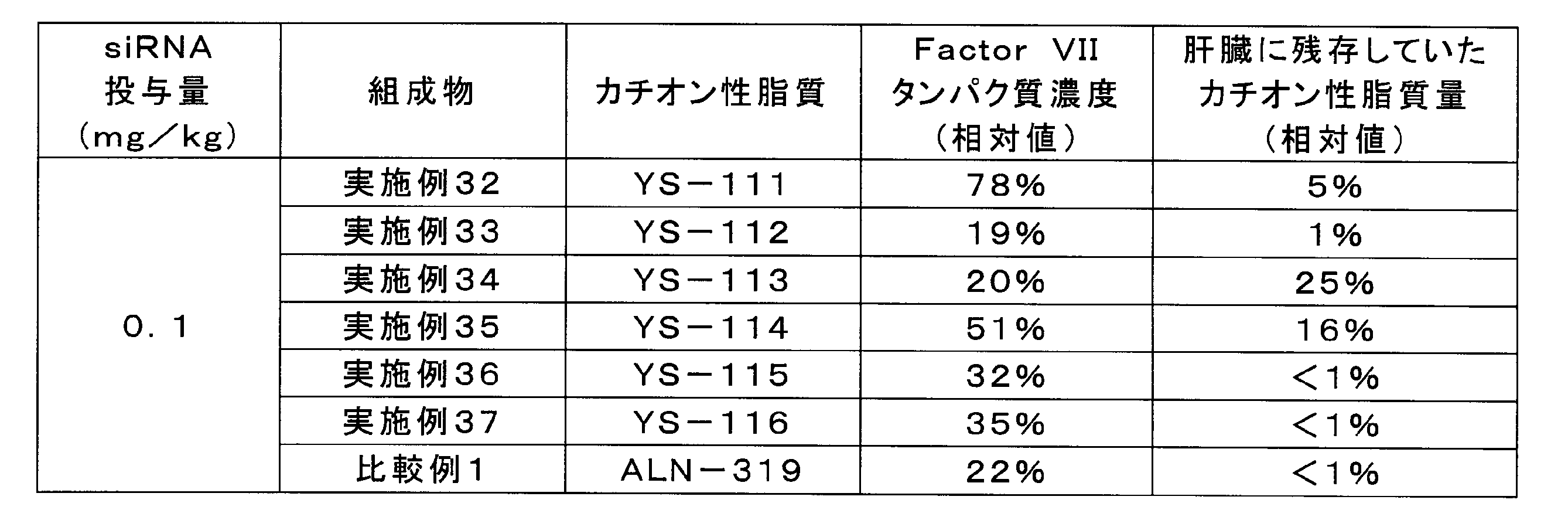
- Compositions containing cationic lipids other than YS-121 were shown to inhibit the increase in particle size of the lipid complex during storage.
- Example 54 A composition of Example 54 was obtained in the same manner as in Example 53 except that the cationic lipid (YS-127) of Example 23 was used instead of YS-120 as the cationic lipid.
- Comparative Example 3 (ALN-319) A composition of Comparative Example 3 was obtained in the same manner as in Example 53 except that ALN-319 was used instead of YS-120 as the cationic lipid.
- composition (9) ⁇ Analysis of composition (9)>
- the lipid complex was dialyzed against PBS, Measure the particle size (Z-average and polydispersity index) at 2 ° C and 6th month after storage at 4 ° C in a sealed vial with a particle size measuring device (trade name “Zetasizer Nano ZS”, manufactured by Malvern) did.
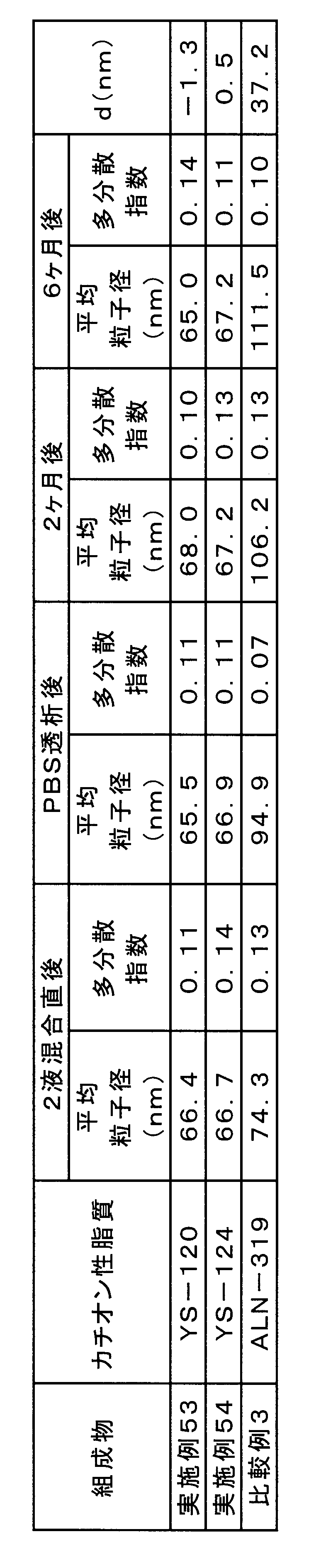
- Table 9 shows the change over time in the average particle size of the compositions of Examples 53 and 54 and Comparative Example 3, and the particle size change (d) immediately after mixing at the sixth month of storage. Compared with the composition of Comparative Example 3, the composition containing YS-120 or YS-124 cationic lipid was shown to suppress the increase in the particle size of the lipid complex during the storage period.
- Example 56 A composition of Example 56 was obtained in the same manner as in Example 55 except that the cationic lipid (YS-127) of Example 23 was used instead of YS-120 as the cationic lipid.
- Example 57 A composition of Example 57 was obtained in the same manner as in Example 55 except that the cationic lipid (YS-128) of Example 24 was used instead of YS-120 as the cationic lipid.
- Example 58 (YS-129) The composition of Example 58 was obtained in the same manner as in Example 55 except that the cationic lipid (YS-129) of Example 25 was used instead of YS-120 as the cationic lipid.
- composition (10) ⁇ Analysis of composition (10)>
- the encapsulation rate of siRNA in the lipid complex and the average particle diameter (Z-average) of the lipid complex were measured for the compositions of Examples 55 to 58.
- Table 10 shows the siRNA encapsulation rate and the average particle size of the lipid complex.
- Example 60 (YS-131) A composition of Example 60 was obtained in the same manner as in Example 59 except that the cationic lipid (YS-131) of Example 26 was used instead of YS-101 as the cationic lipid.
- Example 61 (YS-132) A composition of Example 61 was obtained in the same manner as in Example 59 except that the cationic lipid (YS-132) of Example 27 was used instead of YS-101 as the cationic lipid.
- Example 62 (YS-133) A composition of Example 62 was obtained in the same manner as in Example 59 except that the cationic lipid (YS-133) of Example 28 was used instead of YS-101 as the cationic lipid.
- Example 63 (YS-120) A composition of Example 63 was obtained in the same manner as in Example 59 except that the cationic lipid (YS-120) of Example 16 was used instead of YS-101 as the cationic lipid.
- Comparative Example 4 (ALN-319) A composition of Comparative Example 4 was obtained in the same manner as in Example 59 except that ALN-319 was used instead of YS-101 as the cationic lipid.
- composition (11) ⁇ Analysis of composition (11)>
- the siRNA encapsulation rate in the lipid complex and the average particle size (Z-average) of the lipid complex were determined. It was measured.
- Table 11 shows the siRNA encapsulation rate and the average particle size of the lipid complex.
- the Factor VII protein concentration in the PBS administration group was taken as 100%, and the Factor VII protein concentration in the composition administration group was calculated as a relative value.
- the liver was homogenized, and lipids constituting the composition were extracted with methanol, and then the cationic lipid was quantified by LC-MS.
- the amount of cationic lipid administered was 100%, and the amount of cationic lipid remaining in the liver was calculated as a relative value. The results are shown in Table 12.
- Example 43 and Comparative Example 2 were diluted with PBS so that the concentration of Factor VII siRNA encapsulated in the lipid complex was 1 ⁇ g / mL or 5 ⁇ g / mL.
- Each composition was administered to C57 / BL6 mice (5 weeks old, male) via the tail vein at a dose volume of 10 mL / kg, and blood collection and liver collection were performed under anesthesia 24 hours after administration. Plasma was separated from the blood by centrifugation, and Factor VII protein concentration in the plasma was quantified with a commercially available kit (trade name “BIOPHEN FVII”, HYPHEN BioMed). As a negative control, the group treated with PBS was treated in the same manner.
- the Factor VII protein concentration in the PBS administration group was taken as 100%, and the Factor VII protein concentration in the composition administration group was calculated as a relative value.
- the results are shown in Table 15 and FIG.
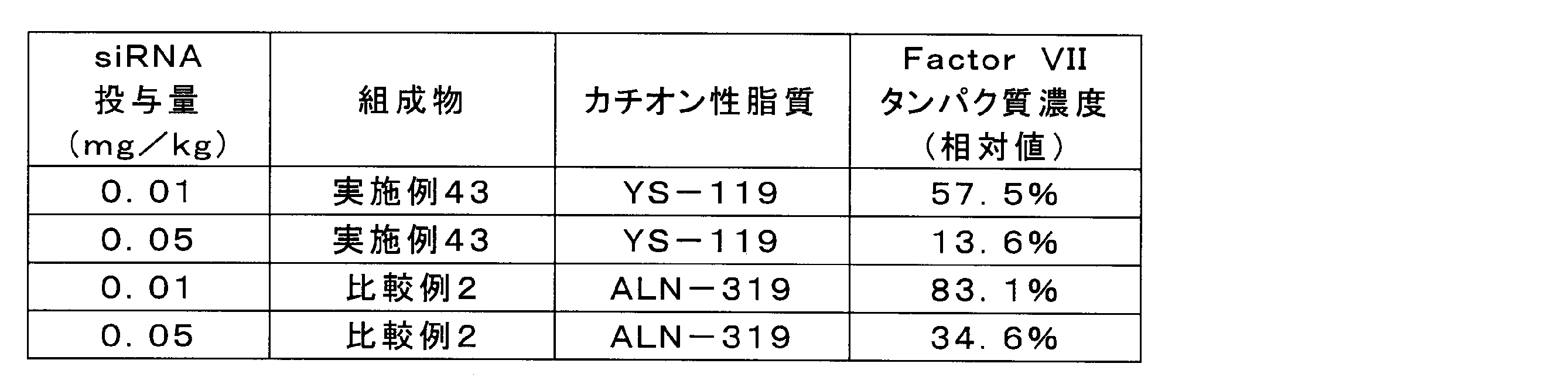
- the composition of Example 43 was shown to have a higher Factor VII protein expression inhibitory effect than the composition of Comparative Example 2.
- compositions of Examples 44 and 45 were diluted with PBS so that the concentration of Factor VII siRNA encapsulated in the lipid complex was 1 ⁇ g / mL or 5 ⁇ g / mL.
- Each composition was administered to C57 / BL6 mice (5 weeks old, male) via the tail vein at a dose volume of 10 mL / kg, and blood collection and liver collection were performed under anesthesia 24 hours after administration. Plasma was separated from the blood by centrifugation, and Factor VII protein concentration in the plasma was quantified with a commercially available kit (trade name “BIOPHEN FVII”, HYPHEN BioMed). As a negative control, the group treated with PBS was treated in the same manner.
- the Factor VII protein concentration in the PBS administration group was taken as 100%, and the Factor VII protein concentration in the composition administration group was calculated as a relative value.
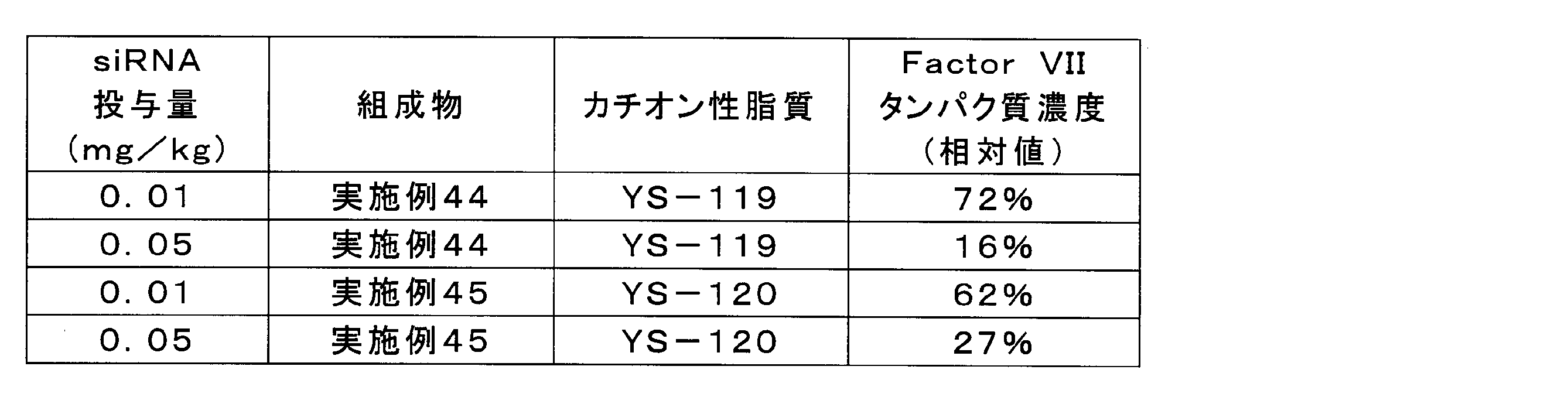
- the results are shown in Table 16 and FIG.
- compositions of Examples 53 and 54 and Comparative Example 3 were diluted with PBS so that the concentration of Factor VII siRNA encapsulated in the lipid complex was 2 ⁇ g / mL or 10 ⁇ g / mL.
- Each composition was administered to ICR mice (6 weeks old, female) via the tail vein at a dose volume of 10 mL / kg, and blood collection and liver collection were performed under anesthesia 24 hours after administration. Plasma was separated from the blood by centrifugation, and Factor VII protein concentration in the plasma was quantified with a commercially available kit (trade name “BIOPHEN FVII”, HYPHEN BioMed). As a negative control, the group treated with PBS was treated in the same manner.
- the Factor VII protein concentration in the PBS administration group was taken as 100%, and the Factor VII protein concentration in the composition administration group was calculated as a relative value.
- the results are shown in Table 18.
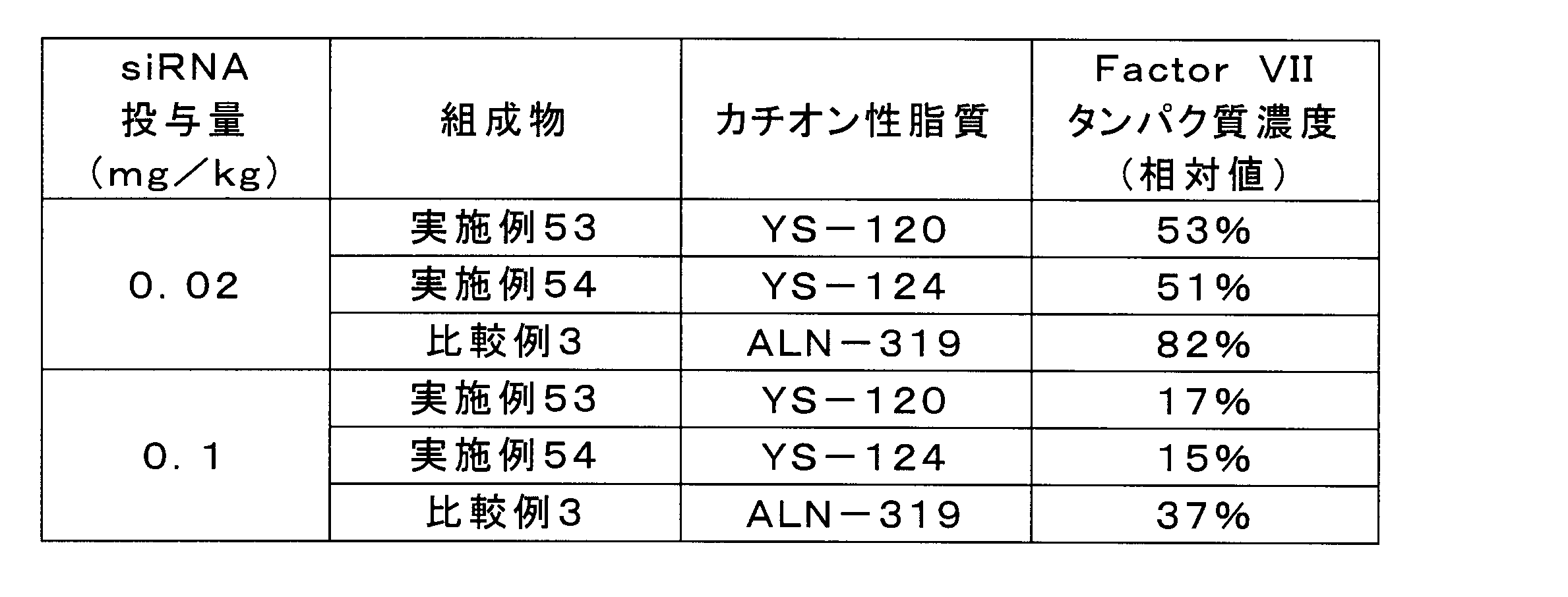
- the compositions of Examples 53 and 54 showed a higher expression inhibitory effect on Factor VII protein than the composition of Comparative Example 3.
- a cationic lipid capable of efficiently releasing nucleic acid into the cytoplasm can be provided.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biomedical Technology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biotechnology (AREA)
- Wood Science & Technology (AREA)
- General Engineering & Computer Science (AREA)
- Zoology (AREA)
- Biophysics (AREA)
- Microbiology (AREA)
- Plant Pathology (AREA)
- Virology (AREA)
- Physics & Mathematics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Hydrogenated Pyridines (AREA)
Abstract
Description
一実施形態において、本発明は、上記式(1)~(11)のいずれかで表される化合物又はその薬剤学的に許容される塩であり、カチオン性脂質として用いることができる。カチオン性脂質は、前記塩の水和物又は前記塩の溶媒和物であってもよい。本明細書において、カチオン性脂質とは、一つ以上の炭化水素基を含む脂質親和性領域と、生理的pHでプロトン化する極性基を含む親水性領域を有する両親媒性分子である。即ち、本発明のカチオン性脂質は、プロトン化し、陽イオンを形成し得る。上記陽イオンと対になって、本実施形態のカチオン性脂質が含有し得る陰イオンとしては、薬剤学的に許容されるものであれば特に限定されず、例えば、塩化物イオン、臭化物イオン、硝酸イオン、硫酸イオン、リン酸イオン等の無機イオン;酢酸イオン、シュウ酸イオン、マレイン酸イオン、フマル酸イオン、クエン酸イオン、安息香酸イオン、メタンスルホン酸イオン等の有機酸イオン等が挙げられる。
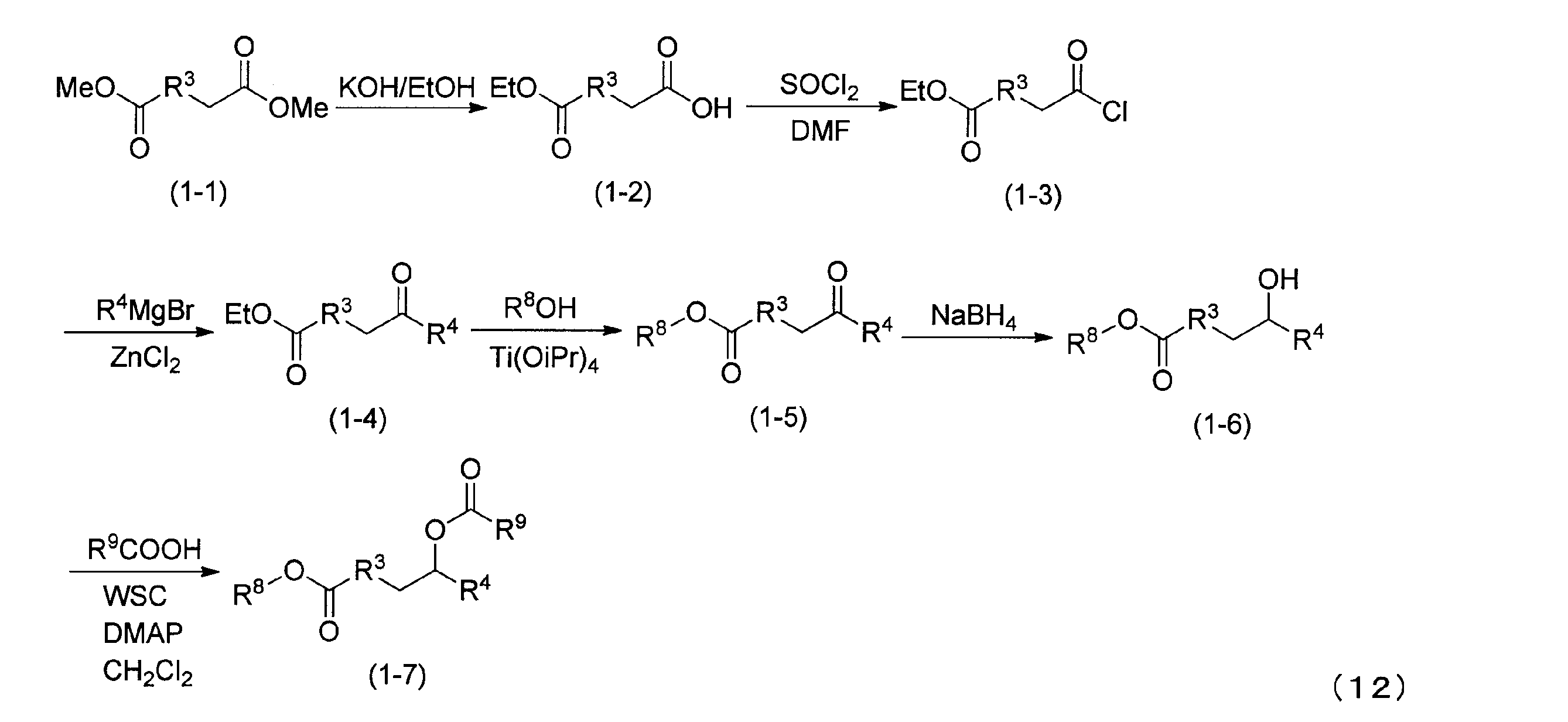
本発明のカチオン性脂質の製造方法について説明する。下記式(12)に、カチオン性脂質の合成スキームの一実施形態を示す。
本発明は、(I)上述したカチオン性脂質と、(II)中性脂質、ポリエチレングリコール修飾脂質及びステロールからなる群より選択される少なくとも一種の脂質と、を含有する脂質複合体を提供する。本発明の一実施形態として、脂質複合体は、(I)上述したカチオン性脂質と、(II)中性脂質、ポリエチレングリコール修飾脂質及びステロールからなる群より選択される少なくとも一種の脂質と、(III)核酸と、を含有する。本実施形態の脂質複合体は、核酸を細胞質に効率よく放出することを可能とする。
一実施形態において、本発明は、(I)上述したカチオン性脂質と、(II)中性脂質、ポリエチレングリコール修飾脂質及びステロールからなる群より選択される少なくとも一種の脂質と、(III)核酸と、を含有する組成物を提供する。本実施形態の組成物は、核酸を細胞質に効率よく放出することを可能とする。本実施形態の組成物は、上述した脂質複合体、薬学的に許容される媒体、及び場合によりその他の添加剤を含有するものであってもよい。薬学的に許容される媒体、その他の添加剤については後述する。
一実施形態において、本発明は、(I)上述したカチオン性脂質と、(II)中性脂質、ポリエチレングリコール修飾脂質及びステロールからなる群より選択される少なくとも一種の脂質とを含有する極性有機溶媒含有水溶液と、(III)核酸を含有する水溶液とを混合して混合液を得る工程(a)と、混合液中の極性有機溶媒の含有量を減少させる工程(b)とを備える、組成物の製造方法を提供する。本実施形態の製造方法によれば、核酸を細胞質に効率よく放出することが可能な組成物を製造することができる。
DMF:N,N-ジメチルホルムアミド
TFA:トリフルオロ酢酸
WSC:1-エチル-3-(3-ジメチルアミノプロピル)カルボジイミド塩酸塩
DMAP:N,N-ジメチル-4-アミノピリジン
THF:テトラヒドロフラン
LC-MS:液体クロマトグラフィー-マススペクトルメトリー
ESI-MS:エレクトロスプレーイオン化質量分析
s:シングレット、d:ダブレット、t:トリプレット、q:カルテット、dd:ダブルダブレット、ddd:ダブルダブルダブレット、dt:ダブルトリプレット、tt:トリプルトリプレット、m:マルチプレット。
[実施例1]
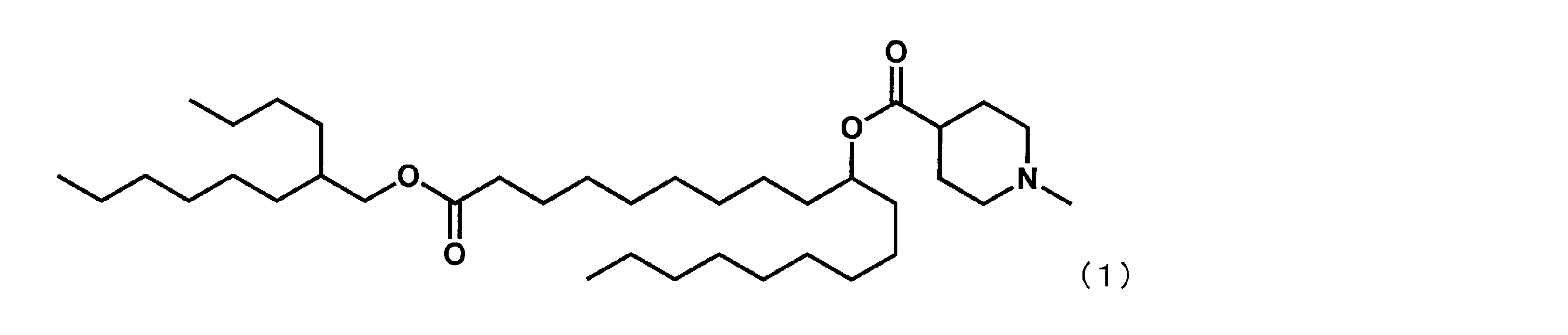
(1-((2-ヘキシルシクロプロピル)メトキシ)-1-オキソノナデカン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-102」という場合がある。)の合成)
以下に、YS-102の合成スキームを示す。
セバシン酸ジメチル(200g、868.4mmol)をエタノール(868mL)に溶かした溶液を0℃まで冷却した。その溶液に水酸化カリウム(48.73g、868.4mmol)のエタノール(300mL)溶液を滴下し、滴下後0℃で12時間撹拌した。反応終了後、酢酸エチルと水を加え、未反応物を有機層に抽出することで除去した。水層を塩酸にて酸性にし、酢酸エチルを加えて目的物を抽出した。有機層を水、食塩水で洗浄し、濃縮した。第1工程品モノエステル体(150.3g、652.6mmol、75%)は精製を行わずに次のステップへ用いた。
第1工程品モノエステル体(50.0g、231.2mmol)と触媒量のDMF(23mL)との懸濁液に、塩化チオニル(41.3g、346.8mmol)を滴下した。反応終了後、塩化チオニルを減圧下留去した後、蒸留精製し、第2工程品(25.8g、109.9mmol、48%)を得た。
塩化亜鉛(2.7g、20.1mmol)をTHF(61mL)に溶解させ、-78℃まで冷却した。この溶液に1Mのノニルマグネシウムブロミド(40.2mL、40.2mmol)を窒素雰囲気下-78℃で滴下した。滴下後0℃まで昇温し、0℃で30分撹拌した後、テトラキストリフェニルホスフィンパラジウム(0.58mg、0.5mmol)を投下し、次いで第2工程品(5.0g、20.1mmol)を滴下した。0℃で更に1時間撹拌した後に1M塩酸水溶液を加えてクエンチした。反応溶液より析出物をろ別し、ろ液を酢酸エチルで抽出し有機層を水、食塩水で洗浄後、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第3工程品(5.0g、14.7mmol、73%)を得た。
第3工程品(2.0g、5.9mmol)、2-ノネノール(4.2g、29.4mmol)、チタニウムテトラプロポキシド(0.2g、0.6mmol)の混合液を撹拌しながら、外浴を110℃まで昇温した。出てくる留出液を除きながら撹拌を続け、留出が見えなくなった時点を反応終点として冷却後水を加えてクエンチした。反応液を酢酸エチルで抽出し水、食塩水で洗浄後、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第4工程品(2.6g、5.8mmol、99%)を得た。
ジエチル亜鉛(8.3mL、8.3mmol)をジクロロメタン(15mL)に溶解させ、0℃まで冷却した。そこへトリフルオロメタンスルホン酸(0.9g、8.3mmol)を滴下し、次いでジヨードメタン(2.2g、8.3mmol)を滴下後、0℃で1時間撹拌した。この溶液に第4工程品(1.2g、2.8mmol)のジクロロメタン(5mL)溶液を滴下し、室温まで昇温した。反応をTLCで確認し、原料の消失を終点として水でクエンチした。析出した固体をろ別し、ろ液を酢酸エチルで抽出、水、食塩水で洗浄後、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第4工程品(1.2g、2.7mmol、99%)を得た。
第5工程品(1.0g、2.2mmol)をエタノール(22mL)に溶かした溶液中に、水素化ホウ素ナトリウム(0.08g、2.2mmol)を添加し、10分間反応させた。反応終了後1N塩酸でクエンチした。酢酸エチルで抽出し、水、食塩水で洗浄後、有機層を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第6工程品(0.3g、0.7mmol、30%)を得た。
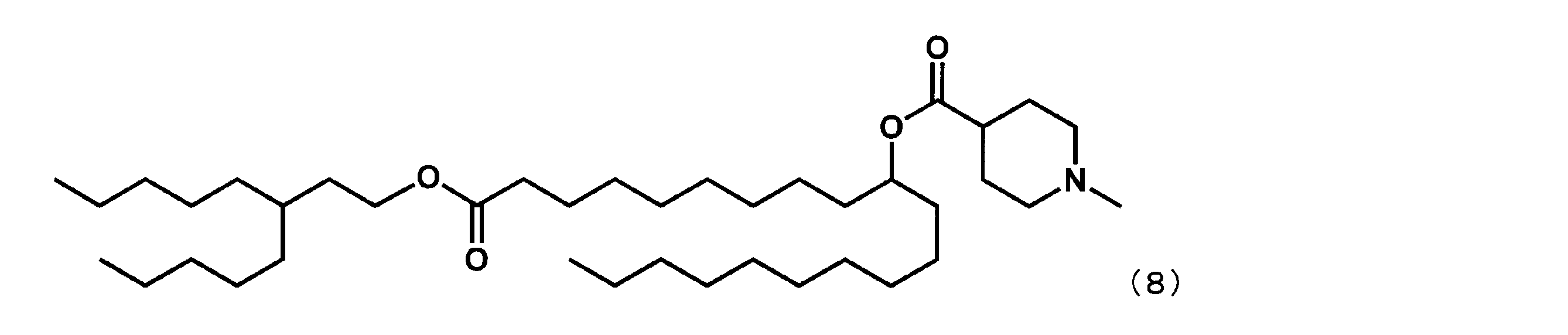
第6工程品(0.8g、1.8mmol)を塩化メチレン(7mL)に溶かした溶液中に、WSC(0.7g、3.7mmol)、ジメチルアミノピリジン(0.04g、0.4mmol)、1-メチルピペリジン-4-カルボン酸(0.5g、3.7mmol)を添加した。室温で翌日まで撹拌した後、水を加え、有機層を分液した。有機層を水で5回洗浄後、1N水酸化ナトリウム水溶液で1度洗浄し、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、下記式(15)で表されるYS-102(0.12g、0.2mmol、12%)を得た。
((Z)-1-(ノン-2-エン-1-イルオキシ)-1-オキソノナデカン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-101」という場合がある。)の合成)
第5工程を行わなかった点以外は実施例1と同様にして、下記式(16)で表されるYS-101を合成した。
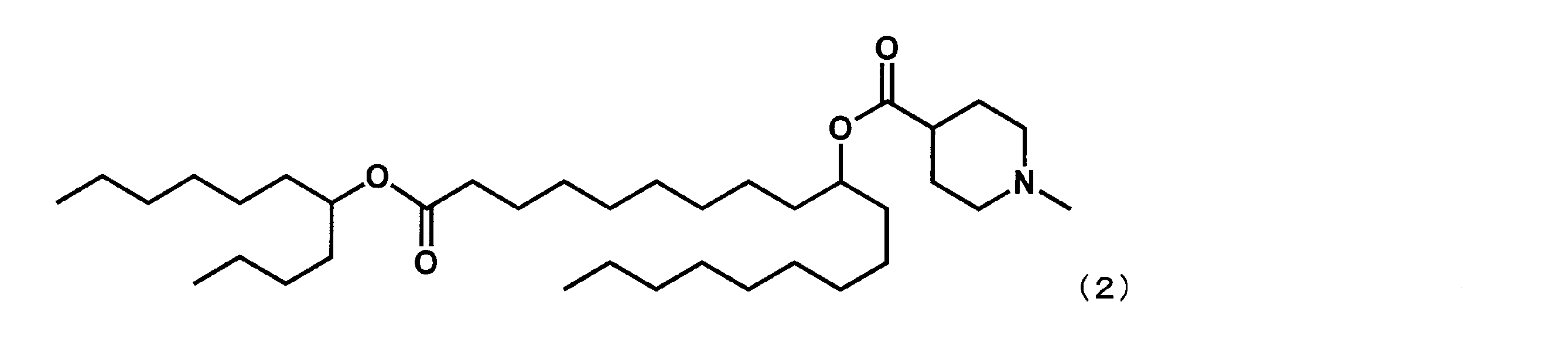
(1-オキソ-1-(ウンデカン-5-イルオキシ)ノナデカン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-103」という場合がある。)の合成)
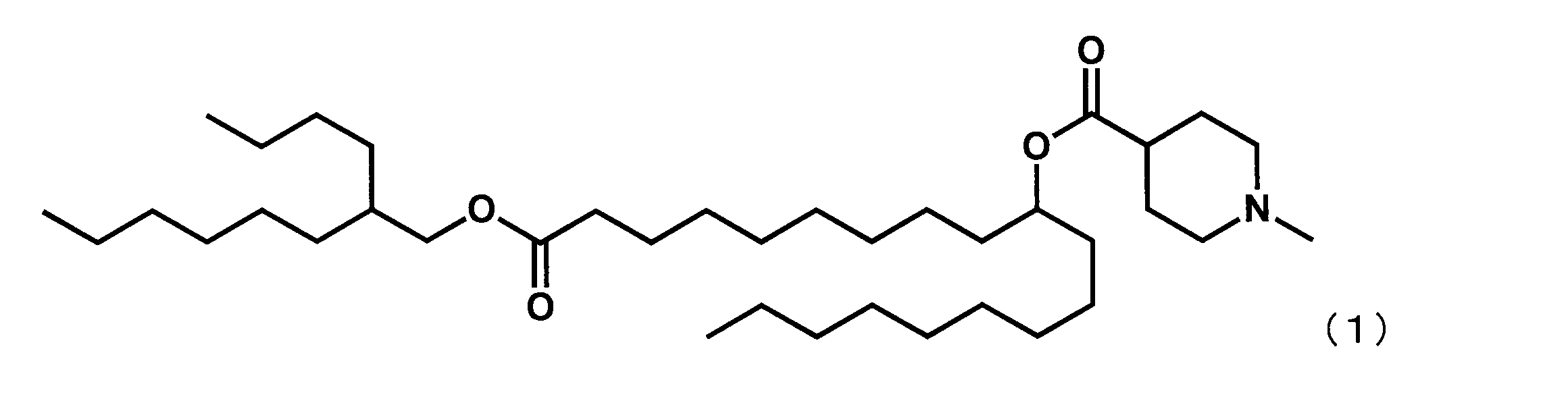
第4工程において、2-ノネノールの代わりに2-ブチルオクタン-1-オールを反応させ、第5工程を行わなかった点以外は実施例1と同様にして、下記式(2)で表されるYS-103を合成した。
(1-((2-ブチルオクチル)オキシ)-1-オキソノナデカン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-119」という場合がある。)の合成)
以下に、YS-119の合成スキームを示す。
第1工程から第3工程は上述したYS-102の合成と同様に行った。
第3工程品(4.1g、12.0mmol)、2-ブチルオクタン-1-オール(6.73g、36.12mmol)、チタニウムテトラプロポキシド(0.34g、1.2mmol)の混合液を撹拌しながら、外浴を110℃まで昇温した。出てくる留出液を除きながら撹拌を続け、留出が見えなくなった時点を反応終点として冷却後水を加えてクエンチした。反応液を酢酸エチルで抽出し、水、食塩水で洗浄後、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第4工程品(4.5g、9.4mmol、78%)を得た。
第4工程品(4.5g、9.4mmol)をTHF(18.7mL)、メタノール(18.7mL)に溶かした溶液中に、水素化ホウ素ナトリウム(1.8g、46.8mmol)を添加し、一時間反応させた。反応終了後1N塩酸でクエンチした。酢酸エチルで抽出し、水、食塩水で洗浄後、有機層を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第5工程品(3.1g、6.42mmol、69%)を得た。
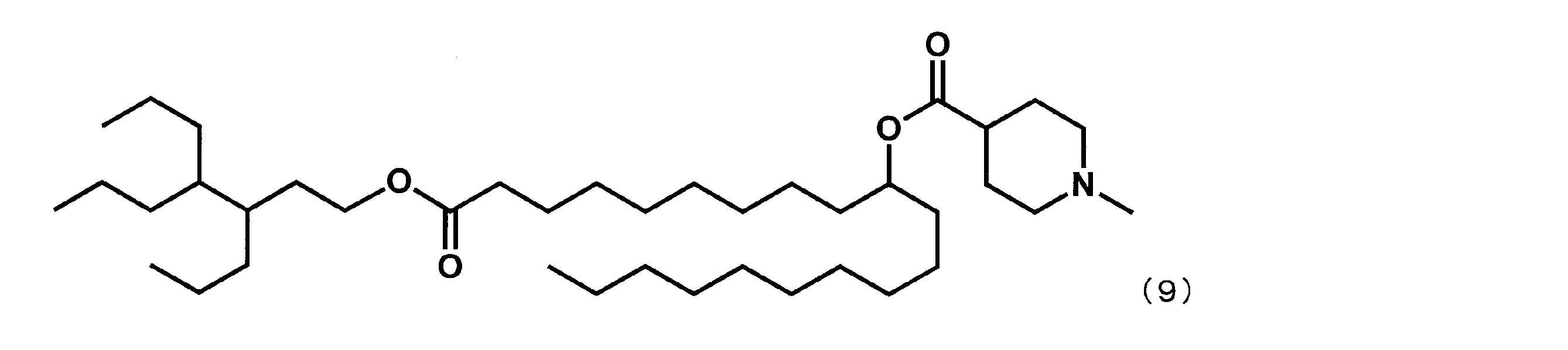
第5工程品(2.0g、4.14mmol)を塩化メチレン(8.28mL)に溶かした溶液中に、WSC(1.59g、8.28mmol)、ジメチルアミノピリジン(0.04g、0.4mmol)、1-メチルピペリジン-4-カルボン酸(1.19g、8.28mmol)を添加した。室温で翌日まで撹拌した後、水を加え、有機層を分液した。有機層を水で5回洗浄後、1N水酸化ナトリウム水溶液で1度洗浄し、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、式(1)で表されるYS-119(1.9g、3.1mmol、74%)を得た。
(1-オキソ-1-(ウンデカン-5-イルオキシ)ペンタデカン-6-イル-1-メチルピペリジン4-カルボキシレート(以下、「YS-111」という場合がある。)の合成)
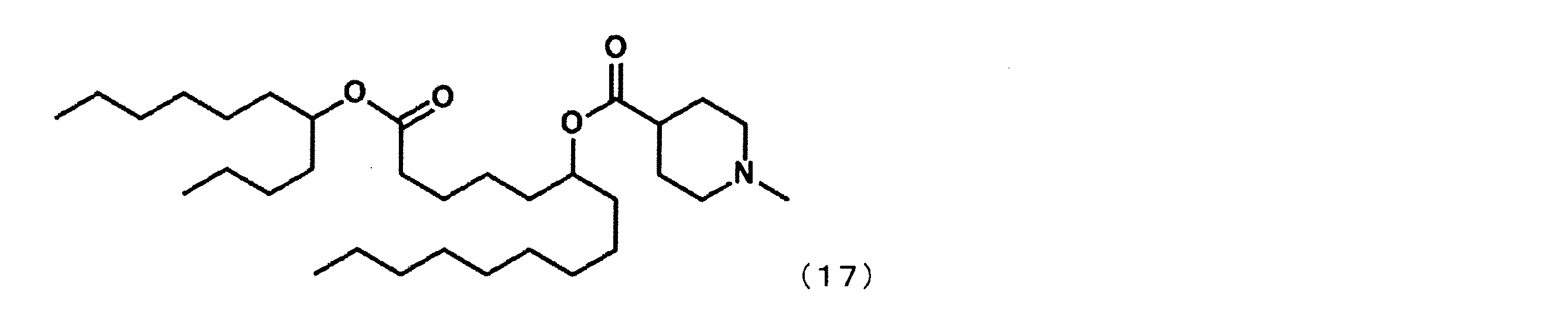
第1工程において、セバシン酸ジメチルの代わりにアジピン酸ジメチルを反応させ、第4工程において、2-ブチルオクタン-1-オールの代わりにウンデカン-5-オールを反応させた以外は実施例4と同様にして、下記式(17)で表されるYS-111を合成した。
(1-オキソ-1-(ウンデカン-5-イルオキシ)ヘプタデカン-8-イル-1-メチルピペリジン4-カルボキシレート(以下、「YS-112」という場合がある。)の合成)
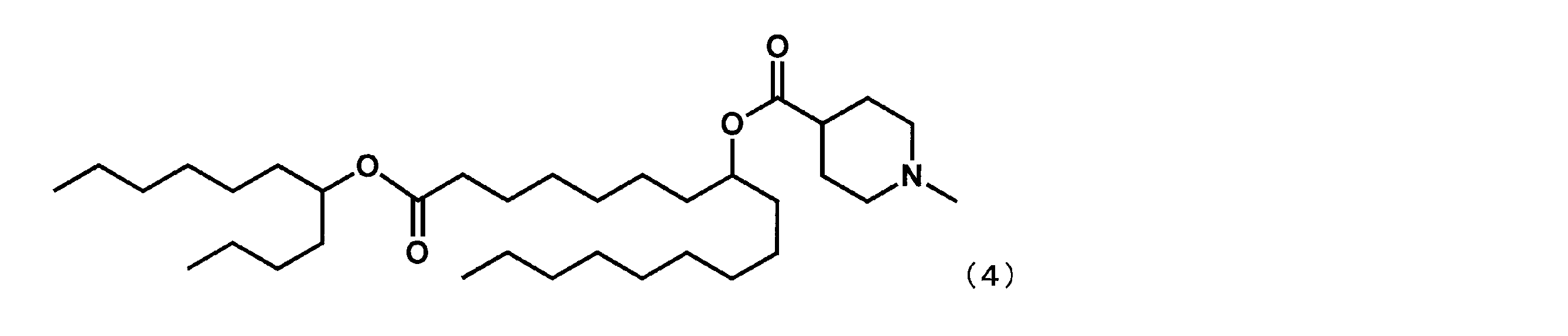
第1工程において、セバシン酸ジメチルの代わりにスベリン酸ジメチルを反応させ、第4工程において、2-ブチルオクタン-1-オールの代わりにウンデカン-5-オールを反応させた以外は実施例4と同様にして、下記式(4)で表されるYS-112を合成した。
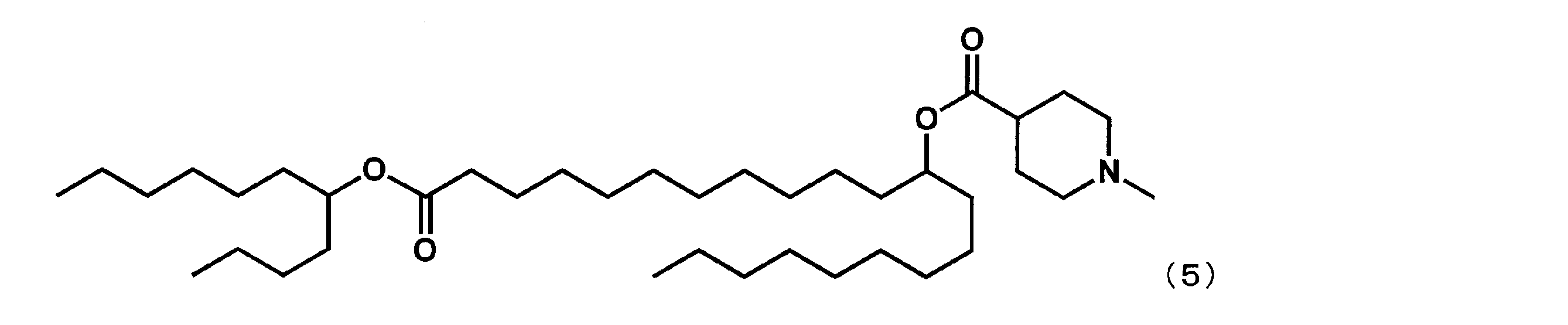
(21-オキソ-21-(ウンデカン-5-イルオキシ)ヘンイコサン-10-イル-1-メチルピペリジン4-カルボキシレート(以下、「YS-113」という場合がある。)の合成)
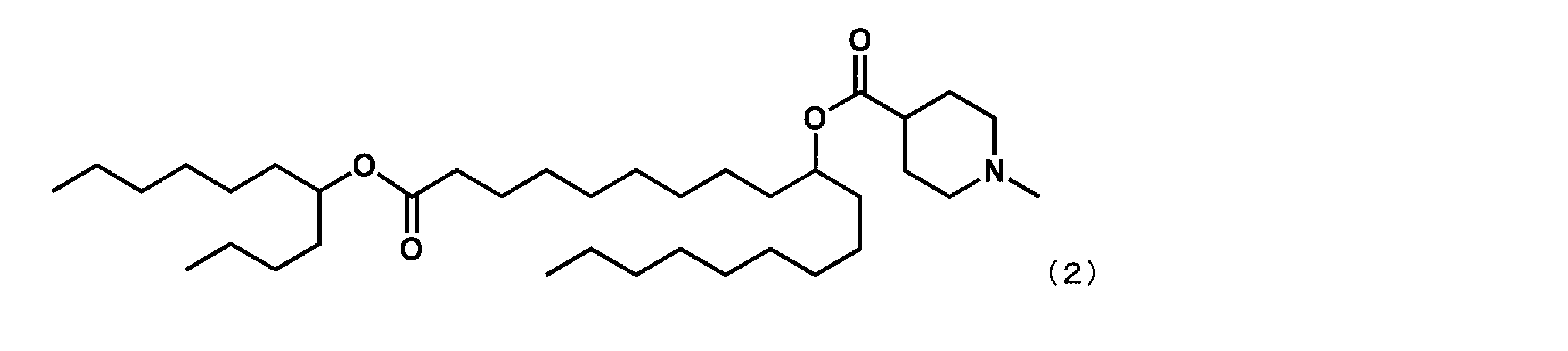
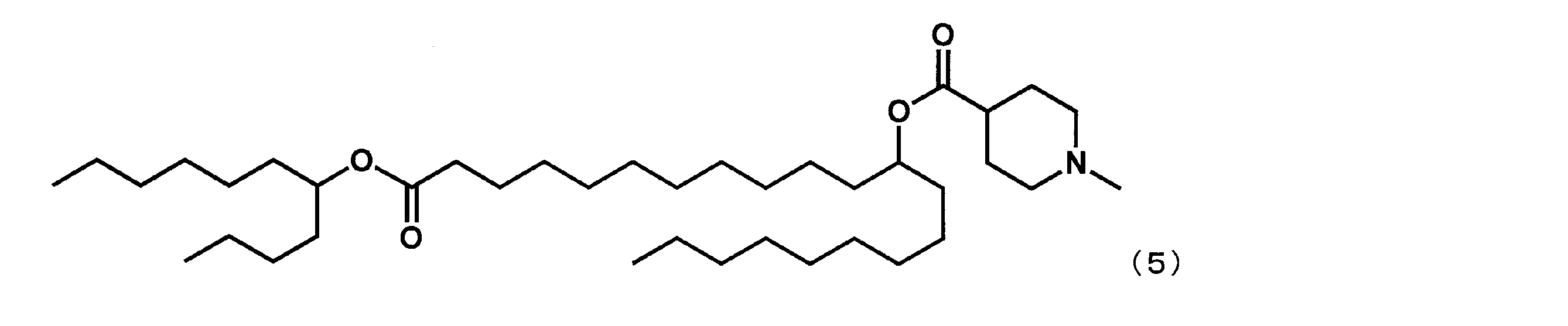
第1工程において、セバシン酸ジメチルの代わりにドデカン二酸ジメチルを反応させ、第4工程において、2-ブチルオクタン-1-オールの代わりにウンデカン-5-オールを反応させた以外は実施例4と同様にして、下記式(5)で表されるYS-113を合成した。
(23-オキソ-23-(ウンデカン-5-イルオキシ)トリコサン-10-イル-1-メチルピペリジン4-カルボキシレート(以下、「YS-114」という場合がある。)の合成)
第1工程において、セバシン酸ジメチルの代わりにテトラデカン二酸ジメチルを反応させ、第4工程において、2-ブチルオクタン-1-オールの代わりにウンデカン-5-オールを反応させた以外は実施例4と同様にして、下記式(18)で表されるYS-114を合成した。
(1-((2-ブチルオクチル)オキシ)-1-オキソヘプタデカン-8-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-115」という場合がある。)の合成)
第1工程において、セバシン酸ジメチルの代わりにスベリン酸ジメチルを反応させた以外は実施例4と同様にして、下記式(19)で表されるYS-115を合成した。
(21-((2-ブチルオクチル)オキシ)-21-オキソヘンイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-116」という場合がある。)の合成)
第1工程において、セバシン酸ジメチルの代わりにスベリン酸ジメチルを反応させた以外は実施例4と同様にして、下記式(20)で表されるYS-116を合成した。
(1-(オクタン-3-イルオキシ)-1-オキソヘプタデカン-8-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-117」という場合がある。)の合成)
第1工程において、セバシン酸ジメチルの代わりにスベリン酸ジメチルを反応させ、第4工程において、2-ブチルオクタン-1-オールの代わりにオクタン-3-オールを反応させた以外は実施例4と同様にして、下記式(21)で表されるYS-117を合成した。
(1-((S)オクタン-3-イルオキシ)-1-オキソヘプタデカン-8-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-117S」という場合がある。)の合成)
第1工程において、セバシン酸ジメチルの代わりにスベリン酸ジメチルを反応させ、第4工程において、2-ブチルオクタン-1-オールの代わりに(S)-オクタン-3-オールを反応させた以外は実施例4と同様にして、下記式(22)で表されるYS-117Sを合成した。
(21-(オクタン-3-イルオキシ)-21-オキソヘンイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-118」という場合がある。)の合成)
第1工程において、セバシン酸ジメチルの代わりにドデカン二酸ジメチルを反応させ、第4工程において、2-ブチルオクタン-1-オールの代わりにオクタン-3-オールを反応させた以外は実施例4と同様にして、下記式(3)で表されるYS-118を合成した。
(21-((S)-オクタン-3-イルオキシ)-21-オキソヘンイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-118S」という場合がある。)の合成)
第1工程において、セバシン酸ジメチルの代わりにドデカン二酸ジメチルを反応させ、第4工程において、2-ブチルオクタン-1-オールの代わりに(S)-オクタン-3-オールを反応させた以外は実施例4と同様にして、下記式(23)で表されるYS-118Sを合成した。
(1-((2-ブチルオクチル)オキシ)-1-オキソイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-120」という場合がある。)の合成)
第3工程において、ノニルマグネシウムブロミドの代わりにデカニルマグネシウムブロミドを反応させた以外は実施例4と同様にして、下記式(6)で表されるYS-120を合成した。
(1-((2-ブチルオクチル)オキシ)-1-オキソイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(「YS-120」)の合成)
以下に、YS-120の別の合成スキームを示す。
第1工程から第3工程は上述したYS-102の合成と同様に行った。
第3工程品(150.0g、440.5mmol)、ベンジルアルコール(142.9g、1321.4mmol)、チタニウムテトラプロポキシド(12.5g、44.5mmol)の混合液を撹拌しながら、外浴を130℃まで昇温した。出てくる留出液を除きながら撹拌を続け、留出が見えなくなった時点を反応終点として冷却後水を加えてクエンチした。反応液を酢酸エチルで抽出し、水、食塩水で洗浄後、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第4工程品(146.2g、350.9mmol)を得た。
第4工程品(100.0g、240.0mmol)をTHF(480.0mL)、メタノール(480.0mL)に溶かした溶液中に、水素化ホウ素ナトリウム(10.9g、288.0mmol)を添加し、10分間反応させた。反応終了後1N塩酸でクエンチした。酢酸エチルで抽出し、水、食塩水で洗浄後、有機層を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第5工程品(70.0g、167.2mmol)を得た。
第5工程品(50.0g、119.4mmol)をTHF(480.0mL)に溶かした溶液中に、WSC(45.8g、238.9mmol)、ジメチルアミノピリジン(2.92g、23.9mmol)、1-メチルピペリジン-4-カルボン酸(34.2g、238.9mmol)を添加した。室温で翌日まで撹拌した後、水を加え、有機層を分液した。有機層を水で5回洗浄後、1N水酸化ナトリウム水溶液で1度洗浄し、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、第6工程品(37.2g、68.4mmol)を得た。
第6工程品(37.2g、68.4mmol)、パラジウム/炭素(4.9mL)を酢酸エチル(136.8mL)に懸濁させ、水素雰囲気下一晩攪拌した。反応液をろ別し、パラジム/炭素を除いて濃縮した。シリカゲルクロマトグラフィーにて精製し、第7工程品(26.8g、59.1mmol)を得た。
第7工程品(20.0g、44.1mmol)を塩化メチレン(220.0mL)に溶かした溶液中に、WSC(8.9g、46.3mmol)、ジメチルアミノピリジン(1.08g、0.4mmol)、2-ブチルオクタン-1-オール(16.4g、88.2mmol)を添加した。室温で翌日まで撹拌した後、水を加え、有機層を分液した。有機層を水で5回洗浄後、無水硫酸マグネシウムで乾燥した。乾燥剤をろ過で除き、ろ液を減圧下濃縮した。シリカゲルカラムクロマトグラフィーにて精製し、式(6)で表されるYS-120(22.0g、35.4mmol)を得た。
(1-((2Z,5Z)-デカ-2,5-ジエン-1-イルオキシ)-1-オキソイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-121」という場合がある。)の合成)
第8工程において、2-ブチルオクタン-1-オールの代わりに(2Z,5Z)-デカ-2,5-ジエン-1-オールを反応させた以外は実施例16と同様にして、下記式(24)で表されるYS-121を合成した。
((Z)-1-((2-ブチルノン-3-エン-1-イル)オキシ)-1-オキソイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-122」という場合がある。)の合成)
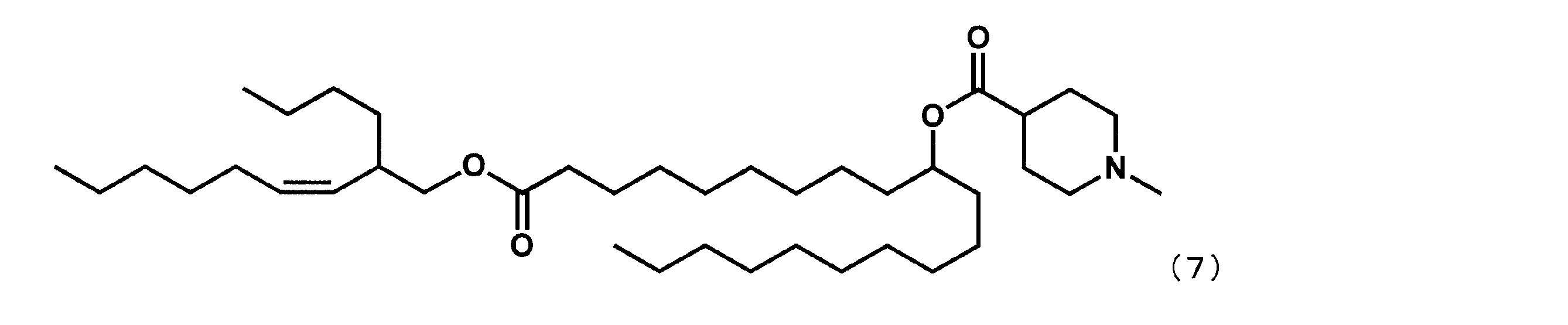
第8工程において、2-ブチルオクタン-1-オールの代わりに(Z)-2-ブチルノン-3-エン-1-オールを反応させた以外は実施例16と同様にして、下記式(25)で表されるYS-122を合成した。
((Z)-1-オキソ-1-((5-プロピルノン-2-エン-1-イル)オキシ)イコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-123」という場合がある。)の合成)
第8工程において、2-ブチルオクタン-1-オールの代わりに(Z)-5-プロピルノン-2-エン-1-オールを反応させた以外は実施例16と同様にして、下記式(26)で表されるYS-123を合成した。
(1-オキソ-1-((3-ペンチルオクチル)オキシ)イコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-124」という場合がある。)の合成)
第8工程において、2-ブチルオクタン-1-オールの代わりに3-ペンチルオクタン-1-オールを反応させた以外は実施例16と同様にして、下記式(27)で表されるYS-124を合成した。
(1-((2,4-ジプロピルヘプチル)オキシ)-1-オキソイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-125」という場合がある。)の合成)
第8工程において、2-ブチルオクタン-1-オールの代わりに2,4-ジプロピルヘプタン-1-オールを反応させた以外は実施例16と同様にして、下記式(28)で表されるYS-125を合成した。
(1-((3,4-ジプロピルヘプチル)オキシ)-1-オキソイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-126」という場合がある。)の合成)
第8工程において、2-ブチルオクタン-1-オールの代わりに3,4-ジプロピルヘプタン-1-オールを反応させた以外は実施例16と同様にして、下記式(29)で表されるYS-126を合成した。
(1-(4-(ヘキシルジスルファニル)-3-((ヘキシルジスルファニル)メチル)ブトキシ)-1-オキソイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-127」という場合がある。)の合成)
第8工程において、2-ブチルオクタン-1-オールの代わりに4-(ヘキシルジスルファニル)-3-((ヘキシルジスルファニル)メチル)ブタン-1-オールを反応させた以外は実施例16と同様にして、下記式(30)で表されるYS-127を合成した。
(1-((6-(ブチルジスルファニル)-3-(3-(ブチルジスルファニル)プロピル)ヘキシル)オキシ)-1-オキソイコサン-10-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-128」という場合がある。)の合成)
第8工程において、2-ブチルオクタン-1-オールの代わりに6-(ブチルジスルファニル)-3-(3-(ブチルジスルファニル)プロピル)ヘキサン-1-オールを反応させた以外は実施例16と同様にして、下記式(31)で表されるYS-128を合成した。
(1-((2-ブチルオクチル)オキシ)-1-オキソイコサン-10-イル-1-メチルピロリジン-3-カルボキシレート(以下、「YS-129」という場合がある。)の合成)
第6工程において、1-メチルピぺリジン-4-カルボン酸の代わりに1-メチルピロリジン-3-カルボン酸を反応させた以外は実施例16と同様にして、下記式(32)で表されるYS-129を合成した。
(1-((2-ブチルオクチル)オキシ)-1-オキソイコサン-10-イル-1-メチルアゼチジン-3-カルボキシレート(以下、「YS-131」という場合がある。)の合成)
第6工程において、1-メチルピぺリジン-4-カルボン酸の代わりに1-メチルアザチジン-3-カルボン酸を反応させた以外は実施例16と同様にして、下記式(33)で表されるYS-131を合成した。
(1-((2-ブチルオクチル)オキシ)-1-オキソイコサン-10-イル-1-エチルピペリジン-4-カルボキシレート(以下、「YS-132」という場合がある。)の合成)
第6工程において、1-メチルピぺリジン-4-カルボン酸の代わりに1-エチルピペリジン-4-カルボン酸を反応させた以外は実施例16と同様にして、下記式(34)で表されるYS-132を合成した。
(2-ブチルオクチル-10-((4-(ジメチルアミノ)ブタノイル)オキシ)イコサノエート(以下、「YS-133」という場合がある。)の合成)
第6工程において、1-メチルピぺリジン-4-カルボン酸の代わりに4-(ジメチルアミノ)ブタン酸を反応させた以外は実施例16と同様にして、下記式(35)で表されるYS-133を合成した。
[実施例29]
(YS-102)
実施例1のカチオン性脂質(YS-102)を用いて組成物を調製した。核酸としては、センス鎖5’-GGAfUfCAfUfCfUfCAAGfUfCfUfUAfCT*T-3’(配列番号1、T:DNA、fU,fC=2’-Fluoro RNA、*=Phosphorothioate linkage)、アンチセンス鎖5’-GfUAAGAfCfUfUGAGAfUGAfUfCfCT*T-3’(配列番号2、T:DNA、fU,fC=2’-Fluoro RNA、*=Phosphorothioate linkage)の塩基配列からなる、Factor VII(血液凝固第VII因子)遺伝子の発現を抑制するsiRNAであり、アニーリング済みのもの(株式会社ジーンデザイン、以下「Factor VII siRNA」という場合がある。)を使用した。
(YS-101)
カチオン性脂質として、YS-102の代わりに実施例2のカチオン性脂質(YS-101)を用いた以外は実施例29と同様にして、実施例30の組成物を得た。
(YS-103)
カチオン性脂質として、YS-102の代わりに実施例3のカチオン性脂質(YS-103)を用いた以外は実施例29と同様にして、実施例31の組成物を得た。
(YS-021)
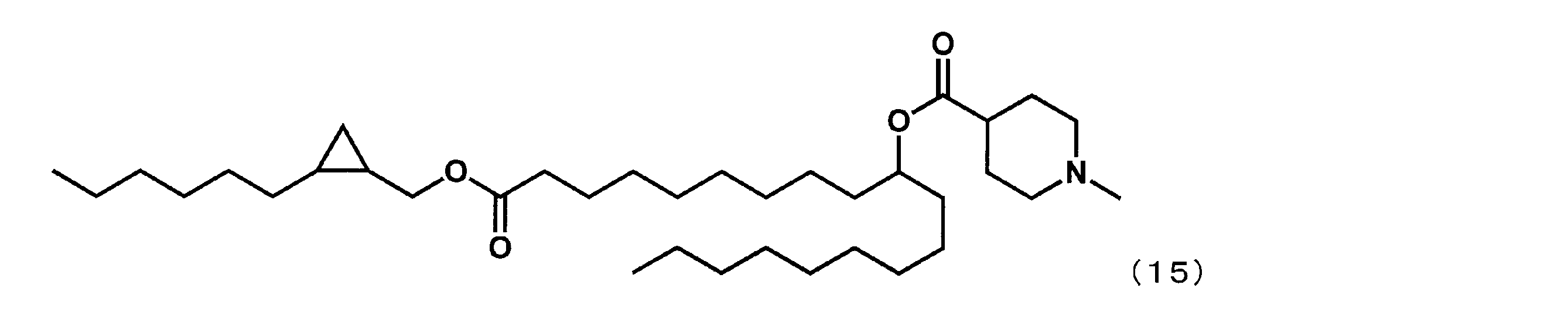
カチオン性脂質として、YS-102の代わりに下記式(36)で表される1-(2-オクチルシクロプロピル)ヘプタデカン-8-イル-1-メチルピペリジン-4-カルボキシレート(以下、「YS-021」という場合がある。)を用いた以外は実施例29と同様にして、参考例1の組成物を得た。
実施例29~31及び参考例1の組成物について、脂質複合体へのsiRNAの封入率を測定した。
封入率(%)=100-(A/B)×100 (F1)
[実施例32]
(YS-111)
実施例5のカチオン性脂質(YS-111)を用いて組成物を調製した。核酸としては、実施例29の組成物で用いたものと同じFactor VII siRNAを使用した。
(YS-112)
カチオン性脂質として、YS-111の代わりに実施例6のカチオン性脂質(YS-112)を用いた以外は実施例32と同様にして、実施例33の組成物を得た。
(YS-113)
カチオン性脂質として、YS-111の代わりに実施例7のカチオン性脂質(YS-113)を用いた以外は実施例32と同様にして、実施例34の組成物を得た。
(YS-114)
カチオン性脂質として、YS-111の代わりに実施例8のカチオン性脂質(YS-114)を用いた以外は実施例32と同様にして、実施例35の組成物を得た。
(YS-115)
カチオン性脂質として、YS-111の代わりに実施例9のカチオン性脂質(YS-115)を用いた以外は実施例32と同様にして、実施例36の組成物を得た。
(YS-116)
カチオン性脂質として、YS-111の代わりに実施例10のカチオン性脂質(YS-116)を用いた以外は実施例32と同様にして、実施例36の組成物を得た。
(ALN-319)
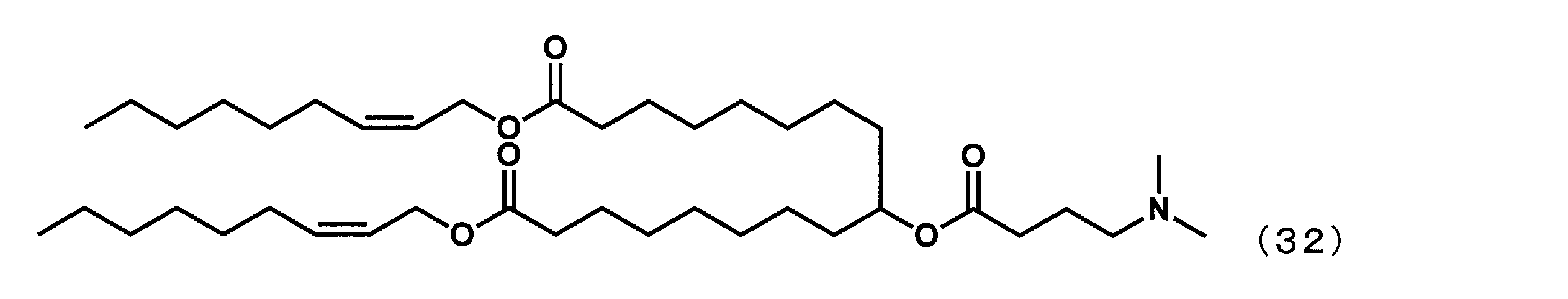
カチオン性脂質として、YS-111の代わりに、特許文献1に記載された下記式(32)で表されるジ((Z)-ノン-2-エン-1-イル)9-((4-(ジメチルアミノ)ブタノイル)オキシ)ヘプタデカンジオエート(以下、「ALN-319」という場合がある。)を、特許文献1に記載された方法にしたがって合成して用いた以外は実施例32と同様にして、比較例1の組成物を得た。
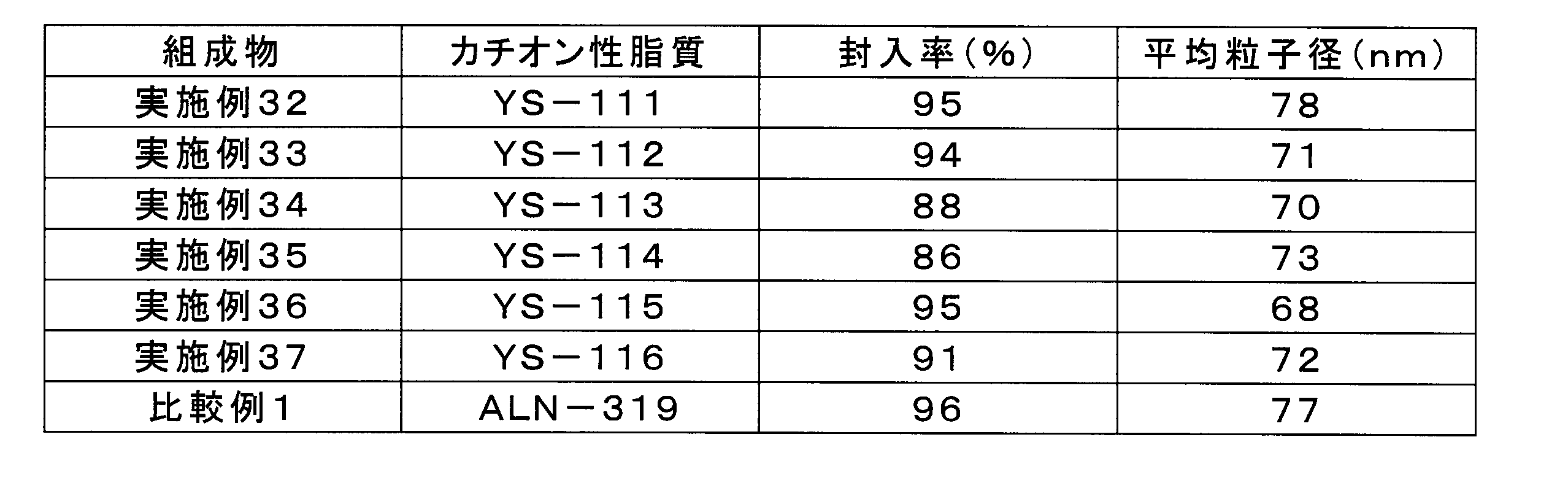
実施例29の組成物と同様にして、実施例32~37及び比較例1の組成物について、脂質複合体へのsiRNAの封入率、及び脂質複合体の平均粒子径を測定した。表2に、siRNAの封入率及び脂質複合体の平均粒子径(Z-平均)を示す。
[実施例38]
(YS-117)
実施例11のカチオン性脂質(YS-117)を用いて組成物を調製した。核酸としては、実施例29の組成物で用いたものと同じFactor VII siRNAを使用した。
(YS-117S)
カチオン性脂質として、YS-117の代わりに実施例12のカチオン性脂質(YS-117S)を用いた以外は実施例38と同様にして、実施例39の組成物を得た。
(YS-118)
カチオン性脂質として、YS-117の代わりに実施例13のカチオン性脂質(YS-118)を用いた以外は実施例38と同様にして、実施例40の組成物を得た。
(YS-118S)
カチオン性脂質として、YS-117の代わりに実施例14のカチオン性脂質(YS-118S)を用いた以外は実施例38と同様にして、実施例41の組成物を得た。
(YS-119)
カチオン性脂質として、YS-117の代わりに実施例4のカチオン性脂質(YS-119)を用いた以外は実施例38と同様にして、実施例42の組成物を得た。
実施例29の組成物と同様にして、実施例38~42の組成物について、脂質複合体へのsiRNAの封入率、及び脂質複合体の平均粒子径を測定した。表3に、siRNAの封入率及び脂質複合体の平均粒子径(Z-平均)を示す。
[実施例43]
(YS-119)
実施例4のカチオン性脂質(YS-119)を用いて組成物を調製した。核酸としては、実施例29の組成物で用いたものと同じFactor VII siRNAを使用した。
(ALN-319)
カチオン性脂質として、YS-119の代わりに、上述したALN-319を用いた以外は実施例43と同様にして、比較例2の組成物を得た。
実施例29の組成物と同様にして、実施例43及び比較例2の組成物について、脂質複合体へのsiRNAの封入率、及び脂質複合体の平均粒子径(Z-平均)を測定した。表4に、siRNAの封入率及び脂質複合体の平均粒子径を示す。
[実施例44]
(YS-119)
実施例4のカチオン性脂質(YS-119)を用いて組成物を調製した。核酸としては、実施例29の組成物で用いたものと同じFactor VII siRNAを使用した。
(YS-120)
カチオン性脂質として、YS-119の代わりに、実施例15のカチオン性脂質(YS-120)を用いた以外は実施例44と同様にして、実施例45の組成物を得た。
実施例29の組成物と同様にして、実施例44及び45の組成物について、脂質複合体へのsiRNAの封入率、及び脂質複合体の平均粒子径(Z-平均)を測定した。表5に、siRNAの封入率及び脂質複合体の平均粒子径を示す。
[実施例46]
(YS-120)
実施例16のカチオン性脂質(YS-120)を用いて組成物を調製した。核酸としては、実施例29の組成物で用いたものと同じFactor VII siRNAを使用した。
(YS-121)
カチオン性脂質として、YS-120の代わりに、実施例17のカチオン性脂質(YS-121)を用いた以外は実施例46と同様にして、実施例47の組成物を得た。
(YS-122)
カチオン性脂質として、YS-120の代わりに、実施例18のカチオン性脂質(YS-122)を用いた以外は実施例46と同様にして、実施例48の組成物を得た。
(YS-123)
カチオン性脂質として、YS-120の代わりに、実施例19のカチオン性脂質(YS-123)を用いた以外は実施例46と同様にして、実施例49の組成物を得た。
(YS-124)
カチオン性脂質として、YS-120の代わりに、実施例20のカチオン性脂質(YS-124)を用いた以外は実施例46と同様にして、実施例50の組成物を得た。
(YS-125)
カチオン性脂質として、YS-120の代わりに、実施例21のカチオン性脂質(YS-125)を用いた以外は実施例46と同様にして、実施例51の組成物を得た。
(YS-126)
カチオン性脂質として、YS-120の代わりに、実施例22のカチオン性脂質(YS-126)を用いた以外は実施例46と同様にして、実施例52の組成物を得た。
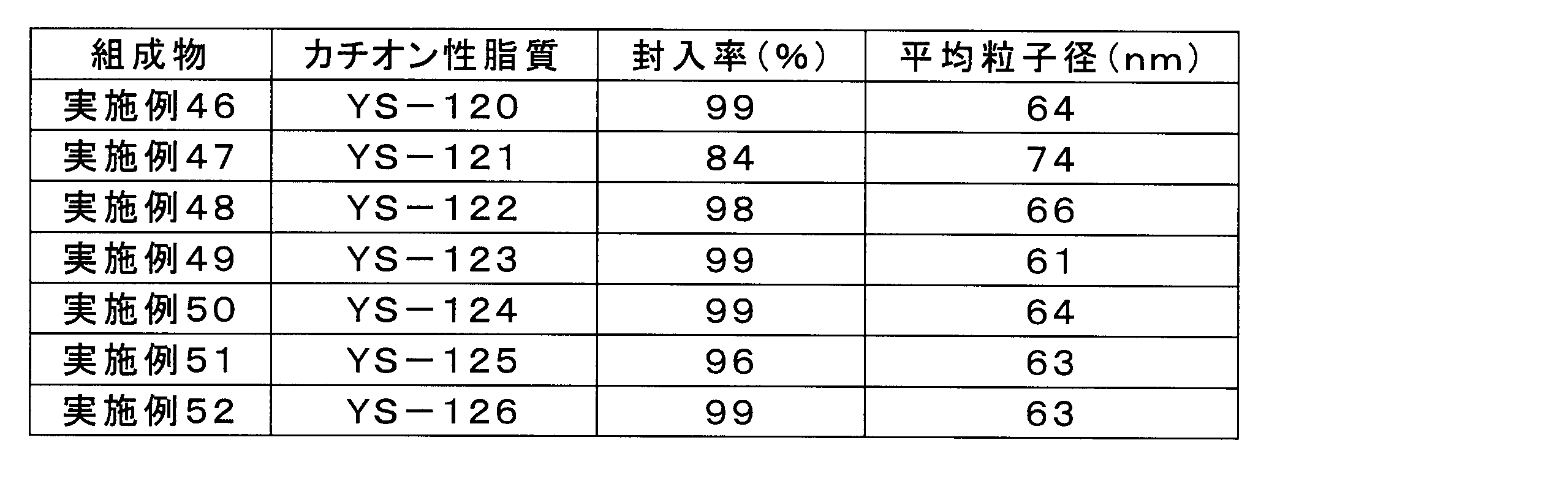
実施例29の組成物と同様にして、実施例46~52の組成物について、脂質複合体へのsiRNAの封入率、及び脂質複合体の平均粒子径(Z-平均)を測定した。表6に、siRNAの封入率及び脂質複合体の平均粒子径を示す。
実施例46~52の組成物について、密閉したバイアルにて4℃で保管し、保管前、保管後3ヶ月目、及び6ヶ月目の粒子径(Z-平均及び多分散指数)を実施例29の組成物と同様にして測定した。
[実施例53]
(YS-120)
「組成物の調製(6)」と同様にして、実施例16のカチオン性脂質(YS-120)を用いて組成物を調製した。
(YS-124)
カチオン性脂質として、YS-120の代わりに、実施例23のカチオン性脂質(YS-127)を用いた以外は実施例53と同様にして、実施例54の組成物を得た。
(ALN-319)
カチオン性脂質として、YS-120の代わりに、上述したALN-319を用いた以外は実施例53と同様にして、比較例3の組成物を得た。
実施例29の組成物と同様にして、実施例53、54、及び比較例3の組成物について、脂質複合体へのsiRNAの封入率、及び脂質複合体の平均粒子径(Z-平均)を測定した。表8に、siRNAの封入率及び脂質複合体の平均粒子径を示す。
実施例53、54、及び比較例3の組成物について、各組成物の調製においてsiRNA希釈液と脂質溶液との混合により脂質複合体が生じた直後、該脂質複合体をPBSに透析した後、密閉したバイアルにて4℃で保管後2ヶ月目、及び6ヶ月目の粒子径(Z-平均及び多分散指数)を粒子径測定装置(商品名「Zetasizer Nano ZS」、Malvern社製)により測定した。
[実施例55]
(YS-120)
「組成物の調製(6)」と同様にして、実施例16のカチオン性脂質(YS-120)を用いて組成物を調製した。
(YS-127)
カチオン性脂質として、YS-120の代わりに、実施例23のカチオン性脂質(YS-127)を用いた以外は実施例55と同様にして、実施例56の組成物を得た。
(YS-128)
カチオン性脂質として、YS-120の代わりに、実施例24のカチオン性脂質(YS-128)を用いた以外は実施例55と同様にして、実施例57の組成物を得た。
(YS-129)
カチオン性脂質として、YS-120の代わりに、実施例25のカチオン性脂質(YS-129)を用いた以外は実施例55と同様にして、実施例58の組成物を得た。
実施例29の組成物と同様にして、実施例55~58の組成物について、脂質複合体へのsiRNAの封入率、及び脂質複合体の平均粒子径(Z-平均)を測定した。表10に、siRNAの封入率及び脂質複合体の平均粒子径を示す。
[実施例59]
(YS-101)
「組成物の調製(6)」と同様にして、実施例2のカチオン性脂質(YS-101)を用いて組成物を調製した。
(YS-131)
カチオン性脂質として、YS-101の代わりに、実施例26のカチオン性脂質(YS-131)を用いた以外は実施例59と同様にして、実施例60の組成物を得た。
(YS-132)
カチオン性脂質として、YS-101の代わりに、実施例27のカチオン性脂質(YS-132)を用いた以外は実施例59と同様にして、実施例61の組成物を得た。
(YS-133)
カチオン性脂質として、YS-101の代わりに、実施例28のカチオン性脂質(YS-133)を用いた以外は実施例59と同様にして、実施例62の組成物を得た。
(YS-120)
カチオン性脂質として、YS-101の代わりに、実施例16のカチオン性脂質(YS-120)を用いた以外は実施例59と同様にして、実施例63の組成物を得た。
(ALN-319)
カチオン性脂質として、YS-101の代わりに、上述したALN-319を用いた以外は実施例59と同様にして、比較例4の組成物を得た。
実施例29の組成物と同様にして、実施例59~63、及び比較例4の組成物について、脂質複合体へのsiRNAの封入率、及び脂質複合体の平均粒子径(Z-平均)を測定した。表11に、siRNAの封入率及び脂質複合体の平均粒子径を示す。
実施例29~31及び参考例1の組成物を、脂質複合体に封入されたFactor VII siRNA濃度が10μg/mLとなるようにPBSで希釈した。各組成物を、C57/BL6マウス(5週齢、オス)に10mL/kgの投与容量で尾静脈内投与し、投与から24時間後に麻酔下で採血及び肝臓の採取を実施した。遠心により血液から血漿を分離し、血漿中のFactor VIIタンパク質濃度を市販のキット(商品名「BIOPHEN FVII」、HYPHEN BioMed社)により定量した。陰性対照として、PBSを投与した群に同様の処理を行った。
実施例32~37及び比較例1の組成物を「動物実験(1)」と同様にしてC57/BL6マウス(5週齢、オス)に投与し、投与から24時間後の血漿中のFactor VIIタンパク質濃度の相対値及び肝臓に残存していたカチオン性脂質量の相対値を算出した。結果を表13に示す。
実施例38~42の組成物を「動物実験(1)」と同様にしてC57/BL6マウス(5週齢、オス)に投与し、投与から24時間後の血漿中のFactor VIIタンパク質濃度の相対値及び肝臓に残存していたカチオン性脂質量の相対値を算出した。結果を表14に示す。
実施例43及び比較例2の組成物を、脂質複合体に封入されたFactor VII siRNA濃度が1μg/mL又は5μg/mLとなるようにPBSで希釈した。各組成物を、C57/BL6マウス(5週齢、オス)に10mL/kgの投与容量で尾静脈内投与し、投与から24時間後に麻酔下で採血及び肝臓の採取を実施した。遠心により血液から血漿を分離し、血漿中のFactor VIIタンパク質濃度を市販のキット(商品名「BIOPHEN FVII」、HYPHEN BioMed社)により定量した。陰性対照として、PBSを投与した群に同様の処理を行った。
実施例44及び45の組成物を、脂質複合体に封入されたFactor VII siRNA濃度が1μg/mL又は5μg/mLとなるようにPBSで希釈した。各組成物を、C57/BL6マウス(5週齢、オス)に10mL/kgの投与容量で尾静脈内投与し、投与から24時間後に麻酔下で採血及び肝臓の採取を実施した。遠心により血液から血漿を分離し、血漿中のFactor VIIタンパク質濃度を市販のキット(商品名「BIOPHEN FVII」、HYPHEN BioMed社)により定量した。陰性対照として、PBSを投与した群に同様の処理を行った。
実施例46~52の組成物を「動物実験(5)」と同様にしてC57/BL6マウス(5週齢、オス)に投与し、投与から24時間後の血漿中のFactor VIIタンパク質濃度の相対値を算出した。結果を表17に示す。
実施例53、54、及び比較例3の組成物を、脂質複合体に封入されたFactor VII siRNA濃度が2μg/mL又は10μg/mLとなるようにPBSで希釈した。各組成物を、ICRマウス(6週齢、メス)に10mL/kgの投与容量で尾静脈内投与し、投与から24時間後に麻酔下で採血及び肝臓の採取を実施した。遠心により血液から血漿を分離し、血漿中のFactor VIIタンパク質濃度を市販のキット(商品名「BIOPHEN FVII」、HYPHEN BioMed社)により定量した。陰性対照として、PBSを投与した群に同様の処理を行った。
実施例55~58の組成物を「動物実験(7)」と同様にしてICRマウス(6週齢、メス)に投与し、投与から24時間後の血漿中のFactor VIIタンパク質濃度の相対値を算出した。結果を表19に示す。
実施例59~63、及び比較例4の組成物を「動物実験(7)」と同様にしてICRマウス(5週齢、メス)に投与し、投与から24時間後の血漿中のFactor VIIタンパク質濃度の相対値を算出した。結果を表20に示す。
Claims (8)
- (I)請求項1~5のいずれか一項に記載の化合物又はその薬学的に許容される塩と、(II)中性脂質、ポリエチレングリコール修飾脂質、及びステロールからなる群から選ばれる少なくとも一種の脂質と、を含有する脂質複合体。
- (I)請求項1~5のいずれか一項に記載の化合物又はその薬学的に許容される塩と、(II)中性脂質、ポリエチレングリコール修飾脂質、及びステロールからなる群から選ばれる少なくとも一種の脂質と、(III)核酸と、を含有する組成物。
- (I)請求項1~5のいずれか一項に記載の化合物又はその薬学的に許容される塩と、(II)中性脂質、ポリエチレングリコール修飾脂質、及びステロールからなる群から選ばれる少なくとも一種の脂質とを含有する極性有機溶媒含有水溶液と、(III)核酸を含有する水溶液とを混合して混合液を得る工程と、混合液中の極性有機溶媒の含有量を減少させる工程と、を含む組成物の製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/533,576 US10081598B2 (en) | 2014-12-26 | 2015-12-24 | Cationic lipid |
| CN201580068712.XA CN107207413B (zh) | 2014-12-26 | 2015-12-24 | 阳离子性脂质 |
| EP15873148.9A EP3239132B1 (en) | 2014-12-26 | 2015-12-24 | Cationic lipid |
| JP2016566429A JP6592458B2 (ja) | 2014-12-26 | 2015-12-24 | カチオン性脂質 |
| CA2969664A CA2969664C (en) | 2014-12-26 | 2015-12-24 | Cationic lipid |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014-266548 | 2014-12-26 | ||
| JP2014266548 | 2014-12-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016104580A1 true WO2016104580A1 (ja) | 2016-06-30 |
Family
ID=56150593
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/085969 WO2016104580A1 (ja) | 2014-12-26 | 2015-12-24 | カチオン性脂質 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US10081598B2 (ja) |
| EP (1) | EP3239132B1 (ja) |
| JP (1) | JP6592458B2 (ja) |
| CN (1) | CN107207413B (ja) |
| CA (1) | CA2969664C (ja) |
| TW (1) | TWI699354B (ja) |
| WO (1) | WO2016104580A1 (ja) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019131580A1 (ja) | 2017-12-27 | 2019-07-04 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | カチオン性脂質 |
| WO2019131770A1 (ja) | 2017-12-27 | 2019-07-04 | 武田薬品工業株式会社 | 核酸含有脂質ナノ粒子及びその用途 |
| US10501416B2 (en) | 2016-06-24 | 2019-12-10 | Eisai R&D Management Co., Ltd. | Cationic lipid |
| WO2020080475A1 (ja) | 2018-10-18 | 2020-04-23 | 武田薬品工業株式会社 | T細胞の活性化/増殖方法 |
| WO2020085456A1 (ja) | 2018-10-26 | 2020-04-30 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | 補体c5の発現を抑制する二本鎖リボ核酸 |
| WO2021132462A1 (ja) | 2019-12-26 | 2021-07-01 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | 補体c5の発現を抑制する二本鎖リボ核酸を含む医薬組成物 |
| WO2022168884A1 (ja) | 2021-02-04 | 2022-08-11 | 塩野義製薬株式会社 | カチオン性脂質 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7273718B2 (ja) | 2017-10-05 | 2023-05-15 | 株式会社クラレ | 変性液状ジエン系ゴム |
| WO2023053017A2 (en) * | 2021-09-28 | 2023-04-06 | Seqirus Inc. | Ionizable cationic compounds for messenger rna delivery |
| AU2022358609A1 (en) * | 2021-09-28 | 2024-05-02 | Seqirus Inc. | Ionizable cationic compound |
| WO2023067560A1 (en) * | 2021-10-22 | 2023-04-27 | Seqirus Inc. | Ionizable cationic compound |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013533224A (ja) * | 2010-06-03 | 2013-08-22 | アルニラム・ファーマシューティカルズ・インコーポレーテッド | 活性剤の送達のための生分解性脂質 |
| WO2013158579A1 (en) * | 2012-04-19 | 2013-10-24 | Merck Sharp & Dohme Corp. | Novel diester and triester based low molecular weight, biodegradable cationic lipids for oligonucleotide delivery |
| WO2015105131A1 (ja) * | 2014-01-09 | 2015-07-16 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | カチオン性脂質 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SI3431076T1 (sl) | 2009-06-10 | 2022-04-29 | Arbutus Biopharma Corporation | Izboljšana lipidna formulacija |
| CA2856742A1 (en) | 2011-12-07 | 2013-06-13 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| AU2013355258A1 (en) | 2012-12-07 | 2015-06-11 | Alnylam Pharmaceuticals, Inc. | Improved nucleic acid lipid particle formulations |
-
2015
- 2015-12-24 TW TW104143612A patent/TWI699354B/zh active
- 2015-12-24 WO PCT/JP2015/085969 patent/WO2016104580A1/ja active Application Filing
- 2015-12-24 CN CN201580068712.XA patent/CN107207413B/zh active Active
- 2015-12-24 EP EP15873148.9A patent/EP3239132B1/en active Active
- 2015-12-24 US US15/533,576 patent/US10081598B2/en active Active
- 2015-12-24 JP JP2016566429A patent/JP6592458B2/ja active Active
- 2015-12-24 CA CA2969664A patent/CA2969664C/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2013533224A (ja) * | 2010-06-03 | 2013-08-22 | アルニラム・ファーマシューティカルズ・インコーポレーテッド | 活性剤の送達のための生分解性脂質 |
| WO2013158579A1 (en) * | 2012-04-19 | 2013-10-24 | Merck Sharp & Dohme Corp. | Novel diester and triester based low molecular weight, biodegradable cationic lipids for oligonucleotide delivery |
| WO2015105131A1 (ja) * | 2014-01-09 | 2015-07-16 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | カチオン性脂質 |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10501416B2 (en) | 2016-06-24 | 2019-12-10 | Eisai R&D Management Co., Ltd. | Cationic lipid |
| WO2019131580A1 (ja) | 2017-12-27 | 2019-07-04 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | カチオン性脂質 |
| WO2019131770A1 (ja) | 2017-12-27 | 2019-07-04 | 武田薬品工業株式会社 | 核酸含有脂質ナノ粒子及びその用途 |
| JPWO2019131580A1 (ja) * | 2017-12-27 | 2020-12-24 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | カチオン性脂質 |
| US10947193B2 (en) | 2017-12-27 | 2021-03-16 | Eisai R&D Management Co., Ltd. | Cationic lipid |
| JP7164547B2 (ja) | 2017-12-27 | 2022-11-01 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | カチオン性脂質 |
| WO2020080475A1 (ja) | 2018-10-18 | 2020-04-23 | 武田薬品工業株式会社 | T細胞の活性化/増殖方法 |
| WO2020085456A1 (ja) | 2018-10-26 | 2020-04-30 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | 補体c5の発現を抑制する二本鎖リボ核酸 |
| WO2021132462A1 (ja) | 2019-12-26 | 2021-07-01 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | 補体c5の発現を抑制する二本鎖リボ核酸を含む医薬組成物 |
| WO2022168884A1 (ja) | 2021-02-04 | 2022-08-11 | 塩野義製薬株式会社 | カチオン性脂質 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP3239132A1 (en) | 2017-11-01 |
| CN107207413A (zh) | 2017-09-26 |
| CN107207413B (zh) | 2019-04-30 |
| JPWO2016104580A1 (ja) | 2017-10-05 |
| TWI699354B (zh) | 2020-07-21 |
| CA2969664C (en) | 2022-06-21 |
| EP3239132A4 (en) | 2018-05-30 |
| US20170334852A1 (en) | 2017-11-23 |
| JP6592458B2 (ja) | 2019-10-16 |
| EP3239132B1 (en) | 2019-03-06 |
| CA2969664A1 (en) | 2016-06-30 |
| TW201630883A (zh) | 2016-09-01 |
| US10081598B2 (en) | 2018-09-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6592458B2 (ja) | カチオン性脂質 | |
| JP6389477B2 (ja) | カチオン性脂質 | |
| JP6409011B2 (ja) | 活性剤の送達のための生分解性脂質 | |
| RU2740921C2 (ru) | Катионный липид | |
| JP7164547B2 (ja) | カチオン性脂質 | |
| JP6240570B2 (ja) | 脂質粒子および核酸送達キャリア | |
| JP6495995B2 (ja) | 脂質粒子および核酸送達キャリア | |
| CN117715886A (zh) | 用于rna递送的可电离阳离子脂质 | |
| JP2008105961A (ja) | アリールメチルアミン誘導体およびその塩ならびにそれを構成成分とする薬物担体 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15873148 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016566429 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2969664 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15533576 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015873148 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |