WO2009112678A2 - Derives de carboxam i d es azabicycliques, leur preparation et leur application en therapeutique. - Google Patents
Derives de carboxam i d es azabicycliques, leur preparation et leur application en therapeutique. Download PDFInfo
- Publication number
- WO2009112678A2 WO2009112678A2 PCT/FR2009/000052 FR2009000052W WO2009112678A2 WO 2009112678 A2 WO2009112678 A2 WO 2009112678A2 FR 2009000052 W FR2009000052 W FR 2009000052W WO 2009112678 A2 WO2009112678 A2 WO 2009112678A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pyridin
- methyl
- pyrrolo
- carboxamide
- cycloalkyl
- Prior art date
Links
- 0 CC(C)NC(C(N(C)C1[*@@](C)C(*)=**)=CC1=C)=NC Chemical compound CC(C)NC(C(N(C)C1[*@@](C)C(*)=**)=CC1=C)=NC 0.000 description 2
- DJCJHFFRHKGOCQ-UHFFFAOYSA-N Cc1cc(cc[nH]2)c2nc1 Chemical compound Cc1cc(cc[nH]2)c2nc1 DJCJHFFRHKGOCQ-UHFFFAOYSA-N 0.000 description 2
- BYYHFYQVWDNVQJ-UHFFFAOYSA-N Cc1cnc2NCCc2c1 Chemical compound Cc1cnc2NCCc2c1 BYYHFYQVWDNVQJ-UHFFFAOYSA-N 0.000 description 2
- GKROJJAWCYEQEA-UHFFFAOYSA-N CC(C=C(C=CN1Cc2cc(cc[n]3C)c3nc2)C1=C)=C[NH2+][O-] Chemical compound CC(C=C(C=CN1Cc2cc(cc[n]3C)c3nc2)C1=C)=C[NH2+][O-] GKROJJAWCYEQEA-UHFFFAOYSA-N 0.000 description 1
- OQUHPMKIHRLWMY-UHFFFAOYSA-N CC(N1c2ncc(C)cc2CC1)=O Chemical compound CC(N1c2ncc(C)cc2CC1)=O OQUHPMKIHRLWMY-UHFFFAOYSA-N 0.000 description 1
- FFVZJALFKFGUJB-UHFFFAOYSA-N CCOC(c([n](Cc1cccc(C)c1)c1c2)cc1ccc2[Si](C)(C)C)=O Chemical compound CCOC(c([n](Cc1cccc(C)c1)c1c2)cc1ccc2[Si](C)(C)C)=O FFVZJALFKFGUJB-UHFFFAOYSA-N 0.000 description 1
- ANDLVYXWZYDHOD-UHFFFAOYSA-N CCOC(c([n](Cc1ncc[s]1)c1c2)cc1ccc2[Si](C)(C)C)=O Chemical compound CCOC(c([n](Cc1ncc[s]1)c1c2)cc1ccc2[Si](C)(C)C)=O ANDLVYXWZYDHOD-UHFFFAOYSA-N 0.000 description 1
- VVBJNUBABXZOCC-UHFFFAOYSA-N CCOC(c1cc(ccc([Si+](C)(C)C)c2)c2[n]1Cc1ccnc(CN=C)c1)=O Chemical compound CCOC(c1cc(ccc([Si+](C)(C)C)c2)c2[n]1Cc1ccnc(CN=C)c1)=O VVBJNUBABXZOCC-UHFFFAOYSA-N 0.000 description 1
- BPENBRBQWYIWMN-UHFFFAOYSA-N CCOC(c1cc2cc([Si](C)(C)C)ccc2[n]1Cc1cccc(C(F)(F)F)c1)=O Chemical compound CCOC(c1cc2cc([Si](C)(C)C)ccc2[n]1Cc1cccc(C(F)(F)F)c1)=O BPENBRBQWYIWMN-UHFFFAOYSA-N 0.000 description 1
- SPWISINYHHHLKC-UHFFFAOYSA-N CCOC(c1cc2cc([Si](C)(C)C)ccc2[n]1Cc1cccc(C)c1)=O Chemical compound CCOC(c1cc2cc([Si](C)(C)C)ccc2[n]1Cc1cccc(C)c1)=O SPWISINYHHHLKC-UHFFFAOYSA-N 0.000 description 1
- QBDAQDGVEMZCCF-UHFFFAOYSA-N CCOC(c1cc2cc([Si](C)(C)C)ccc2[n]1Cc1ccncc1)=O Chemical compound CCOC(c1cc2cc([Si](C)(C)C)ccc2[n]1Cc1ccncc1)=O QBDAQDGVEMZCCF-UHFFFAOYSA-N 0.000 description 1
- JNFKLPXVKSFLPU-UHFFFAOYSA-N CCOC(c1cc2cc([Si](C)(C)CC)ccc2[n]1Cc1ncc[s]1)=O Chemical compound CCOC(c1cc2cc([Si](C)(C)CC)ccc2[n]1Cc1ncc[s]1)=O JNFKLPXVKSFLPU-UHFFFAOYSA-N 0.000 description 1
- WLLXKYBSJUMKII-UHFFFAOYSA-N Cc1cc(CC(N2)=O)c2nc1 Chemical compound Cc1cc(CC(N2)=O)c2nc1 WLLXKYBSJUMKII-UHFFFAOYSA-N 0.000 description 1
- CCGIHXFFRUDGLC-UHFFFAOYSA-N Cc1cc(CCN2C)c2nc1 Chemical compound Cc1cc(CCN2C)c2nc1 CCGIHXFFRUDGLC-UHFFFAOYSA-N 0.000 description 1
- JMFRWFPPCICJCQ-UHFFFAOYSA-N Cc1cc([nH]cc2)c2nc1 Chemical compound Cc1cc([nH]cc2)c2nc1 JMFRWFPPCICJCQ-UHFFFAOYSA-N 0.000 description 1
- YQDMUZFABFBKJN-UHFFFAOYSA-N Cc1ccc(cc[nH]2)c2n1 Chemical compound Cc1ccc(cc[nH]2)c2n1 YQDMUZFABFBKJN-UHFFFAOYSA-N 0.000 description 1
- RFYQKMAGEWSMPV-UHFFFAOYSA-N Cc1ccc(cc[n]2C)c2n1 Chemical compound Cc1ccc(cc[n]2C)c2n1 RFYQKMAGEWSMPV-UHFFFAOYSA-N 0.000 description 1
- ZYGLVDPTWXJGKV-UHFFFAOYSA-N Cc1ncc2[nH]ccc2c1 Chemical compound Cc1ncc2[nH]ccc2c1 ZYGLVDPTWXJGKV-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/04—Antipruritics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/14—Decongestants or antiallergics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/20—Antivirals for DNA viruses
- A61P31/22—Antivirals for DNA viruses for herpes viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- WO2007 / 010138 disclose bicyclic carboxamide derivatives having in vitro and in vivo antagonistic or agonist activity for TRPV1 receptors
- the present invention addresses this need by providing azabicyclic carboxamide derivatives which exhibit antagonistic or agonist activity in vitro and in vivo for TRPV1 (or VR1) type receptors.
- a first subject of the invention relates to the compounds corresponding to the general formula (I) below.
- Another subject of the invention relates to processes for the preparation of the compounds of general formula (I).
- Another subject of the invention relates to the use of compounds of general formula
- Xi, X ⁇ , X 3 and X 4 independently of one another, a nitrogen atom or CR 1; It being understood that when one of X 1 , X 2 , X 3 and X 4 represents a nitrogen atom, the others correspond to a C-Ri group;
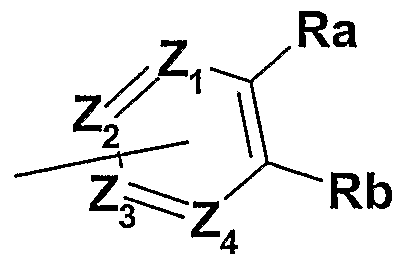
- Z 1 , Z 2 , Z 3 and Z 4 represent, independently of one another, a nitrogen atom, a carbon atom or a CR 2 group, at least one of Z 1 and Z 2 , Z 3 and Z 4 corresponding to a nitrogen atom and one of Z 1 , Z 2 , Z 3 and Z 4 , corresponding to a carbon atom, being bonded to the nitrogen atom of the amide or thioamide of formula (I);
- Ra and Rb form together with the carbon atoms which carry them a five-membered ring , this ring comprising a nitrogen atom and carbon atoms, this ring being partially saturated or unsaturated and being optionally substituted by one or more substituents R 3 ;
- W represents an oxygen or sulfur atom
- n 0, 1, 2 or 3;
- Y represents an optionally substituted aryl or heteroaryl with one or more groups selected from a halogen atom, a CrC ⁇ -alkyl, C 3 -C 7 - cycloalkyl, cycloalkyl-QrCy CrQralkylène, C r C 6 fluoroalkyl, hydroxy , C r C 6 alkoxy, C 3 -C 7 cycloalkyloxy, C 3 -C 7 -cycloalkyl--C 6 -alkylene-O-, -C 6 -fluoroalkoxy, cyano, C (O) NR 4 R 5, nitro, NR 4 R 5, C r C 6 thioalkyl, thiol, -S (O) -C 1 -C 6 -alkyl, -S (O) 2 -C 1 -C 6 - alkyl, SO 2 NR 4 R 5, NR 6 C (O) R 7 , NR 6 SO 2 R
- R 1 is chosen from a hydrogen atom, a halogen atom, CrC 6 -alkyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-C r C 3 -alkylene, CrC 6 -fluoroalkyl, aryloxy -CRC 6 -alkyl, heteroaryloxy-Cl-C 6 -alkyl, aryl-FCP-alkyleneoxy-Ci-C ⁇ -alkyl, heteroaryl-C r C 3 - alkyleneoxy-C r C 6 alkyl, arylthio-C r C 6 - alkyl, heteroarylthio-C-BL-alkyl, aryl-C r C 3 - Alkylene-thio-CrCe-alkyl, heteroaryl-C r -C 3 -alkylene-thio-C -C 6 -alkyl, dC 6 -alk
- R 2 represents a hydrogen atom, a halogen atom, a group -C 6 -alky! E, C 3 -C 7 -cycloaIkyle, C 3 -C 7 -cycloalkyl--C 3 -alkylene, dC 6 fluoroalkyl, Ci-C 6 alkoxy, C 3 -C 7 -cycloalkyloxy, C 3 -C 7 -cycloalkyl-Ci-C 3 -alkylene-O-, hydroxy, thiol, C 1 -C 6 - fluoroalkoxy;
- R 3 represents, when carried by a carbon atom, a hydrogen atom, a hydroxyl, thiol, CrC 6 -alkyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-Ci group; -C 3 -alkylene, C r C 6 fluoroalkyl, Ci-C 6 alkoxy, C 3 -C 7 -cycloalkyloxy, C 3 -C 7 -cycloalkyl-C r C 3 - alkyleneoxy, dC 6 -alkoxy-Ci-C 3 -alkylene, C 3 -C 7 -cycloalkyloxy-Ci-C 3 -alkylene, C 3 -C 7 - cycIoalkyl-CRCS CRCS-alkyleneoxy-alkylene, C (O) NR 4 R 5, C (O) ODC 6 - alkyl, CO 2 H, or an oxo
- alkylene C 3 -C 7 - cycloalkyl-Ci-C 3 -alkylénoxy-C r C 3 -alkylene which can be substituted by hydroxy, C r C 6 alkoxy or NR 4 R 5; or
- R 3 represents, when it is borne by a nitrogen atom, a hydrogen atom, a d-C ⁇ -alkyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-C 1 - C 3 -alkylene, RCJC-fluoroalkyl, aryl-C (O) -, C r C 6 -alkyl-C (O) -, C 3 -C 7 -cycloalkyl-C (O) -, C 3 -C 7 - cycloalkyl-C 1 -C 3 -alkylene-C (O) -) C r -C 6 -fluoroalkyl-C (O) -, aryl-S (O), C r C 6 -aIkyle- S (O) -, dC 6 -fluoroaIkyle-S (0) -, aryl-S (O) 2 -, C r
- R 4 and R 5 independently of one another, a hydrogen atom or a C r C 6 -alkyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-C r C 3 -alkylene, aryl-C 1 -C 5 -alkylene or aryl, or R 4 and R 5 together with the nitrogen atom carrying them, an azetidine, pyrrolidine, piperidine, azepine, morpholine, thiomorpholine, piperazine group, homopiperazine; the NR 4 R 5 group being optionally substituted by a group -C 6 -alkyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-C r C 3 -alkylene, aryl-C r C 6 - alkylene, aryl , heteroaryl, aryl-S (O) 2 -, C 1 -C 6
- R 6 and R 7 represent, independently of one another, a hydrogen atom, a C r C 6 -aIkyle, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-C r C 3 -alkylene, aryl-C r C 6 - alkylene or aryl; the aryl group being optionally substituted by one or more substituents selected from a halogen atom, a CiC 6 -aIkyle group, C 3 -C 7 - cycloalkyl, C 3 -C 7 -cycloalkyl-C r C 3 -alkylene, C r -C 6 fluoroalkyl, C r C 6 alkoxy, C 3 -C 7 - cycloalkyloxy, C 3 -C 7 -cycloalkyl-CrC -alkylénoxy 3, -C 6 -fluoroalkoxy, nitro or cyano
- R 8 is CrC ⁇ -alkyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-Ci-C 3 - alkylene, aryl-C r C 6 -alkylene or aryl; the aryl group being optionally substituted by one or more substituents chosen from a halogen atom, a C 1 -C 6 -alkyl, C 3 -C 7 -cycloalkyl, C 3 -C 7 -cycloalkyl-C 1 -C 3 - alkylene, C 1 -C 6 -fluoroalkyl, C ⁇ C 6 -alkoxy, C 3 -C 7 -cycloalkyloxy, C 3 -C 7 -cycloalkyl-C 1 -C 3 -alkylenoxy, C 1 -C 6 -fluoroalkoxy, nitro or cyano ; or R 6 and R 8 together form a 4- to 7
- sulfur atom or atoms may be in oxidized form (S (O) or S (O) 2 );
- the nitrogen atom or atoms may optionally be in oxidized form (N-oxide).
- the compounds of formula (I) may comprise one or more asymmetric carbon atoms. They can therefore exist as enantiomers or diastereoisomers. These enantiomers, diastereoisomers, as well as their mixtures, including the racemic mixtures, form part of the invention.
- the compounds of formula (I) may exist in the form of bases or addition salts with acids. Such addition salts are part of the invention.
- salts can be prepared with pharmaceutically acceptable acids, but the salts of other acids that are useful, for example, for the purification or the isolation of the compounds of formula (I) are also part of the invention.
- the compounds of formula (I) may also exist in the form of hydrates or solvates, namely in the form of associations or combinations with one or more water molecules or with a solvent. Such hydrates and solvates are also part of the invention.
- halogen atom a fluorine, a chlorine, a bromine or an iodine
- a carbon chain may have from t to z carbon atoms where t and z can take the values of 1 to 7; for example C 1 -C 3 is a carbon chain which may have from 1 to 3 carbon atoms; an alkyl: a saturated linear or branched aliphatic group.
- alkylene a saturated divalent linear or branched alkyl group, for example a C 1-3 -alkylene group, represents a divalent carbon chain of 1 to 3 carbon atoms, linear or branched, more particularly a methylene, ethylene, 1-methylethylene propylene;
- a cycloalkyl a cyclic alkyl group, saturated or partially unsaturated.
- cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl groups and the like;
- a cycloalkyloxy a -O-cycloalkyl radical where the cycloalkyl group is as previously defined;
- fluoroalkyl an alkyl group of which one or more hydrogen atoms have been substituted by a fluorine atom;
- an alkoxy an -O-alkyl radical where the alkyl group is as previously defined;
- a fluoroalkoxy an alkoxy group in which one or more hydrogen atoms have been substituted by a fluorine atom;
- a thioalkyl or alkyl-thio a -S-alkyl radical where the alkyl group is as previously defined;
- aryl a mono- or bicyclic aromatic group comprising between 6 and 10 carbon atoms.
- aryl group mention may be made of phenyl or naphthyl groups; a heteroaryl: a 5- to 12-membered aromatic mono- or bicyclic group containing from 1 to 5 heteroatoms selected from O, S or N.
- Examples of monocyclic heteroaryl include imidazolyl, pyrazolyl, thiazolyl, oxazolyl, isothiazolyl, isoxazolyl, furanyl, thiophenyl, oxadiazolyl, thiadiazolyl, triazolyl, tetrazolyl, pyridinyl, pyrazinyl, pyrimidinyl, pyridazinyl, triazinyl.
- bicyclic heteroaryl there may be mentioned indolyl, isoindolyl, benzofuranyl, benzothiophenyl, benzoxazolyl, benzimidazolyl, indazolyl, benzothiazolyl, isobenzofuranyl, isobenzothiazolyl, pyrrolo [2,3-c] pyridinyl, pyrrolo [2,3 pyridinyl, pyrrolo [3,2-b] pyridinyl, pyrrolo [3,2-c] pyridinyl, pyrrolo [1,2-a] pyridinyl, quinolinyl, isoquinolinyl, cinnolinyl, quinazolinyl, quinoxalinyl, pyrrolo [1, 2] -a] imidazolyl, imidazo [1,2-a] pyridinyl, imidazo [1, 2- a] pyridazinyl, imidazoly
- a first subgroup of compounds is constituted by the compounds for which X 1 , X 2 , X 3 and X 4 represent, independently of one another, a group CR 1 ; R 1 being as defined in the general formula (I).
- a second subgroup of compounds is constituted by the compounds for which one of X 1 , X 2 , X 3 and X 4 represents a nitrogen atom, the others of X 1 , X 2 , X 3 and X 4 represent, independently of one another, a CR 1 group; R 1 being as defined in the general formula (I).
- a third subgroup of compounds consists of the compounds for which X 1, X 2, X 3 and X 4 one of X 3 and X 4 is a nitrogen atom, the others represent, independently of one another, a CRi group; R 1 being as defined in the general formula (I).
- a fourth subgroup of compounds is constituted by the compounds for which R 1 is chosen from a hydrogen atom, a halogen atom, a Cl 2 group, C ⁇ -fluoroalkyl or -Si (C r C 6 -alkyl) 3 .
- a fifth subgroup of compounds consists of the compounds for which R 1 is chosen from a hydrogen atom, a fluorine atom, a CF 3 group or If (CHg) 3 .
- R 1 is chosen from a hydrogen atom, a fluorine atom, a CF 3 group or If (CHg) 3 .
- a sixth subgroup of compounds is constituted by the compounds for which n is equal to 1.
- a seventh subgroup of compounds is constituted by compounds for which
- Y represents an aryl or a heteroaryl optionally substituted with one or more groups selected from a halogen atom, a C ⁇ C 6 -alkyl or CrCe-fluoroalkyl group.
- an eighth subgroup of compounds is constituted by the compounds for which
- Y represents a phenyl, optionally substituted with one or more groups selected from a halogen atom, a C 1 -C 6 -alkyl or C 1 -C 6 -fluoroalkyl group; or Y is pyridinyl or thiazolyl.
- a ninth subgroup of compounds consists of the compounds for which
- Y represents a phenyl, optionally substituted with a fluorine atom, a methyl group or CF 3 ; or Y is pyridinyl or thiazolyl.
- Y represents a phenyl, optionally substituted by a fluorine atom, a methyl group or CF 3 .
- an eleventh subgroup of compounds consists of the compounds for which W represents an oxygen atom.
- a twelfth subgroup of compounds consists of the compounds for which Z 1 , Z 2 , Z 3 and Z 4 represent, independently of one another a nitrogen atom, a carbon atom or a CR 2 group, one of Z 1 , Z 2 , Z 3 and Z 4 corresponding to a nitrogen atom and possibly being in oxidized form; one of Z 1 , Z 2 , Z 3 and Z 4 , corresponding to a carbon atom, being bonded to the nitrogen atom of the amide or thioamide of formula (I); and the other two of Z 1 , Z 2 , Z 3 and Z 4 corresponding to a group CR 2 ;
- R 2 being as defined in the general formula (I).
- a thirteenth subgroup of compounds is constituted by the compounds for which Z 1 , Z 2 , Z 3 and Z 4 represent, independently of one another a nitrogen atom, a carbon atom or a CR 2 group, one of Z 2 , Z 2 , Z 3 and Z 4 corresponding to a nitrogen atom and possibly being in oxidized form; one of Z 1 , Z 2 , Z 3 and Z 4 , corresponding to a carbon atom, being bonded to the nitrogen atom of the amide or thioamide of formula (I); and the other two of Z 1 , Z 2 , Z 3 and Z 4 corresponding to a CH group.
- Ra and Rb form together with the carbon atoms which carry them a five-membered ring, this ring comprising a nitrogen atom and carbon atoms, this ring being partially saturated or unsaturated and being optionally substituted by one or more substituents R 3 ;
- R 3 represents, when carried by a carbon atom, a hydrogen atom or an oxo group
- R 3 represents, when worn by a nitrogen atom, a hydrogen atom, C r C 6 alkyl or Ci-C 6 -alkyl-C (O) -.
- a fifteenth subgroup of compounds consists of the compounds for which Ra and Rb together with the carbon atoms carrying them form a five-membered ring. comprising a nitrogen atom and carbon atoms, this ring being partially saturated or unsaturated and being optionally substituted by one or more substituents R 3 ;
- R 3 represents, when carried by a carbon atom, a hydrogen atom or an oxo group; R 3 represents, when it is borne by a nitrogen atom, a hydrogen atom, a methyl group or CH 3 -C (O) -.
- Z 1 , Z 2 , Z 3 and Z 4 corresponding to a nitrogen atom and possibly being in oxidized form; these groups being optionally substituted with R 2 and R 3 as defined in the general formula (I);
- R 2 represents a hydrogen atom
- R 3 represents, when carried by a carbon atom, a hydrogen atom or an oxo group
- R 3 represents, when worn by a nitrogen atom, a hydrogen atom, a Ci-C 6 alkyl group or C r C 6 -alkyl-C (O) -.
- an eighteenth subgroup of compounds is constituted by the compounds for which the group
- a twentieth subgroup of compounds is constituted by the compounds for which the group
- R 2 represents a hydrogen atom
- R 3 represents, when carried by a carbon atom, a hydrogen atom or an oxo group
- R 3 represents, when worn by a nitrogen atom, a hydrogen atom, a C r C 6 -alkyl or C r C 6 -alkyl-C (O) -.
- a twenty-first subgroup of compounds is constituted by the compounds for which the group
- a twenty-second subgroup of compounds is constituted by the compounds for which the definitions of X 1 , X 2 , X 3 , and X 4 , n, Y , W, Z 1 , Z 2 , Z 3 , Z 4 ; Ra and Rb given above are combined.
- a twenty-third subgroup of compounds is constituted by the compounds for which X 1 , X 2 , X 3 and X 4 represent, independently of one of the other, a CRi group; either of X 1 , X 2 , X 3 and X 4 one of X 3 and X 4 represents a nitrogen atom, the others represent, independently of one another, a CR 1 group;
- R 1 is chosen from a hydrogen atom, a halogen atom, a group
- Y represents a phenyl, optionally substituted by one or more groups selected from a halogen atom, a CrC ⁇ -alkyl or C-1-C ⁇ -fluoroalkyl group; or Y is pyridinyl or thiazolyl;
- W represents an oxygen atom
- Z 1 , Z 2 , Z 3 and Z 4 corresponding to a nitrogen atom and possibly being in oxidized form; these groups being optionally substituted with R 2 and R 3 as defined in the general formula (I);
- R 2 represents a hydrogen atom
- R 3 represents, when carried by a carbon atom, a hydrogen atom or an oxo group
- R 3 represents, when carried by a nitrogen atom, a hydrogen atom or a C-
- a twenty-fourth subgroup of compounds is defined such as the compounds for which
- X 1 , X 2 , X 3 , and X 4 represent, independently of one another, a CR 1 group
- R 1 is selected from a hydrogen atom, a halogen atom, a C 1 -C 6 - alkyl, Ci-C 6 fluoroalkyl, C r C 6 alkoxy, C r -C 6 -fluoroalkoxy, NR 4 R 5, C r C 6 thioalkyl, phenyl or isoxazolyl; the phenyl group being optionally substituted by one or more substituents R 9 , which are identical or different from one another;
- R 4 and R 5 represent, independently of one another, a hydrogen atom or a C 1 -C 6 -alkyl group
- Y represents a phenyl optionally substituted by one or more substituents R 9 , which are identical or different from one another; or Y represents an isoxazole;
- R 9 represents a halogen atom, a C 1 -C 6 alkyl, C 1 -C 6 alkoxy, cyano group; the group
- L represents a hydrogen atom, a halogen atom or a Ci-C 4 alkoxy
- the five-membered ring is partially saturated or unsaturated;
- R ' represents a hydrogen atom or a C 1 -C 4 -alkyl or aryl-C (O) - group, the aryl group being optionally substituted by one or more C 1 -C 6 alkyl group; are excluded.
- a twenty-fifth subgroup of compounds is constituted by compounds for which X 1 , X 2 , X 3 , and X 4 represent, independently of the other a CR 1 group; and R 1 is selected from a hydrogen atom, a halogen atom, more particularly a fluorine atom.
- a twenty-sixth subgroup of compounds is constituted by the compounds for which n is equal to 1 and Y represents an aryl, more particularly a phenyl, optionally substituted by one or more halogen atoms, more particularly fluorine.
- a twenty-seventh subgroup of compounds consists of the compounds for which W represents an oxygen atom.
- a twenty-ninth subgroup of compounds is constituted by the compounds for which X 1 , X 2 , X 3 , and X 4 represent, independently of each other, a CR 1 group; and R 1 is selected from a hydrogen atom, a halogen atom, more particularly a fluorine atom; n is 1;
- Y represents an aryl, more particularly a phenyl, optionally substituted by one or more halogen atoms, more particularly fluorine;
- W represents an oxygen atom
- leaving group is meant, in what follows, a group that can be easily cleaved from a molecule by breaking a heterolytic bond, with departure from an electronic pair. This group can thus be easily replaced by another group during a substitution reaction, for example.
- Such leaving groups are, for example, halogens or an activated hydroxy group such as methanesulfonate, benzenesulfonate, p-toluenesulfonate, triflate, acetate, etc. Examples of leaving groups and references for their preparation are given in "Advances in Organic Chemistry", J. March, 5th Edition, Wiley Interscience, 2001.
- Protective group in the following, a group that can be momentarily incorporated in a chemical structure for the purpose of temporarily inactivating a part of the molecule during a reaction and which can be easily removed at a later stage of the synthesis. Examples of protecting groups as well as references to their properties are given in TW Greene, PGM Wutz, 3rd Edition, Wiley Interscience 1999. According to the invention, the compounds of general formula (I) can be prepared according to the process illustrated by the general scheme 1 which follows:
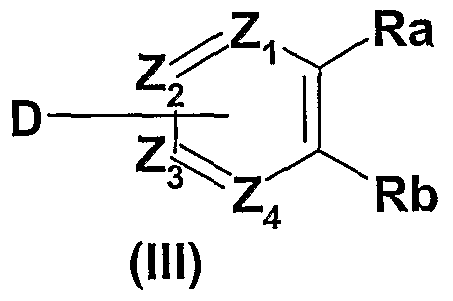
- the compounds of general formula (I) can be obtained by reaction of a compound of general formula (II) in which X 1 , X 2 , X 3 , X 4 , n, Y and W are as defined in the general formula (I) above and B is a hydroxyl group, with an amine of general formula (III), wherein Z 1 , Z 2 , Z 3 , Z 4 , Ra and Rb are as defined in the general formula ( I) above and D is amino, in the presence of a coupling agent such as dialkylcarbodiimide, [(benzotriazol-1-yl) oxy] [tris (pyrrolidino)] phosphonium hexafluorophosphate. , diethylcyanophosphonate or any other coupling agent known to those skilled in the art, optionally in the presence of a base such as triethylamine, in a solvent such as for example dimethylformamide.
- a coupling agent such as dialkylcarbodiimi
- the compound of general formula (II), for which B represents a C 1 -C 6 -alkoxyl group, can be converted into a compound of general formula (II), where B represents a hydroxyl group, by the action of a base such as sodium hydroxide or potassium hydroxide solution in a solvent such as ethanol.
- the compound of general formula (II), in which B represents a hydroxyl group may subsequently be converted into a compound of general formula (II), in which B represents a chlorine atom, by the action of a chlorinating agent such as than thionyl chloride in a solvent such as dichloromethane.
- the compounds of general formula (I) can be obtained by reaction of a compound of general formula (II) in which X 1 , X 2 , X 3 , X 4 , n, Y and W are as defined in the general formula (I) above and B corresponds to a chlorine atom, with an amine of general formula (III), in which Z 1 , Z 2 , Z 3 , Z 4 , Ra and Rb are as defined in the general formula (I) above and D corresponds to a group amino, by reaction in solution in a solvent such as dichloromethane or toluene.
- a solvent such as dichloromethane or toluene.
- the compounds of general formula (I) can also be obtained by reaction of a compound of general formula (II) in which X 1 , X 2 , X 3 , X 4 , n, Y and W are as defined in the formula general (I) above and B corresponds to a C 1 -C 6 -alkoxyl group, with an amide, resulting from (III), in which Z 1 , Z 2 , Z 3 , Z 4 , Ra and Rb are such as defined in the general formula (I) above and D is an amino group, and an organometallic reagent such as trimethylaluminum.
- This reaction can be carried out in a solvent such as toluene.
- the compound of general formula (I) can be obtained by reaction with the compound of general formula (III), wherein Z 1 , Z 2 , Z 3 , Z 4 , Ra and Rb are such as defined in the general formula (I) above and D corresponds to a leaving group as defined above, such as a bromine atom or a triflate group, for example by a method similar to that described in J. Am.Chem.Soc.
- the compounds of general formula (II) or (I), for which one of X 1 , X 2 , X 3 or X 4 , corresponds to a carbon atom substituted by an alkyl group, may be obtained by a reaction of metal-catalyzed coupling such as palladium or iron, carried out on the compounds of general formulas (II) or (I) corresponding, substituted by a halogen atom, such as a chlorine, in the presence for example of an alkylmagnesium halide or an alkylzinc halide, according to the methods described in the literature (A. Furstner et al JA Chem Soc 2002, 124 (46), 13856, G. Quéguiner et al J Org Chem 1998, 63 (9), 2892) for example, or known to those skilled in the art.
- the compounds of general formula (II) or (I), for which one of X 1 , X 2 , X 3 or X 4 , corresponds to a carbon atom substituted with a cyano, aryl or heteroaryl group, may be obtained by a metal-catalyzed coupling reaction, such as palladium, carried out on the corresponding compounds of general formula (II) or (I), substituted, for example, with a bromine atom, in the presence of trimethylsilyl cyanide, arylboronic acid or heteroarylboronic acid, or by any other method described in the literature or known to those skilled in the art.
- the compounds of general formula (I) or (II) substituted with a C (O) NR 4 R 5 group may be obtained from the corresponding compounds of general formula (I) or (II) substituted with a cyano group, according to methods described in the literature or known to those skilled in the art.
- the compounds of general formula (I) or (II) substituted with a -S (O) -alkyl or -S (O) 2 -alkyl group can be obtained by oxidation of the compounds of general formula (II) or (I) corresponding, substituted by a thioalkyl group, according to methods described in the literature or known to those skilled in the art.
- the compounds of general formula (II) or (I), substituted with a group NR 4 R 5 , NR 6 COR 7 or NR 6 SO 2 R 8 may be obtained from compounds of general formula (II) or (I ) corresponding substituted by nitro, for example by ' reduction, and then acylation or sulfonylation, according to methods described in the literature or known to those skilled in the art.
- the compounds of general formula (I) or (II) in which W represents a sulfur atom may, for example, be obtained by reaction of the corresponding compounds of general formula (I) or (II) in which W represents an atom of oxygen, with a reagent such as Lawesson's reagent.
- R 3 corresponds to a protective group borne by a nitrogen, such as an acetyl, ethoxycarbonyl, terbutyloxycarbonyl or a benzyloxycarbonyl group, may be deprotected according to chemical methods known to man. of the art, to lead to compounds of general formula (I) wherein R 3 is a hydrogen atom.
- Z 4 corresponds to an N-oxide group
- Z 4 may, for example, be obtained by reaction of the corresponding compounds of general formula (I) in which at least one of Z 1 , Z 2 , Z 3 and Z 4 corresponds to a nitrogen atom, with a reagent such as metachloroperbenzoic acid.
- the compounds of general formula (II), defined such that n is equal to 1, 2 or 3, are obtained by reaction of the compounds of general formula (VI) corresponding with a reagent of general formula (VIII), wherein GP represents a leaving group such as a chlorine, bromine or iodine atom and n is equal to 1, 2 or 3.
- the compounds of general formula (II) can be obtained by reaction of the compound of general formula (VI ) with a compound of general formula (VIII) in the presence of a phosphine such as, for example, triphenylphosphine and a reagent such as, for example, diethyl azodicarboxylate dissolved in a solvent such as dichloromethane or tetrahydrofuran (O. Mitsonobu, Synthesis, 1981, 1-28).
- a phosphine such as, for example, triphenylphosphine
- a reagent such as, for example, diethyl azodicarboxylate dissolved in a solvent such as dichloromethane or tetrahydrofuran (O. Mitsonobu, Synthesis, 1981, 1-28).
- the compounds of general formula (II) can be obtained by reacting the compound of general formula (VI) with a compound of general formula (VIII) in the presence of a phosphine supported on a resin and a reagent.
- a phosphine supported on a resin and a reagent such as, for example, diisopropyl azodicarboxylate dissolved in a solvent such as dichloromethane or tetrahydrofuran.
- the compounds of general formula (VI) are prepared from aromatic or heteroaromatic aldehydes substituted with a silanyl group of general formula (IV), in which X 1 , X 2 , X 3 and X 4 are as defined in the formula general (I) and one of them corresponding to a silanyl group, by reaction with an alkyl azidoacetate of general formula (VII) in which B represents a C r C 6 -alkoxyl group, such as azidoacetate for example, in the presence of a base such as sodium ethoxide, in a solvent such as ethanol or methanol, to yield the alkyl azido-2-cinnamates of general formula (V).
- the formation of the compounds of general formula (VI) can be obtained by decomposition of the alkyl azido-2-cinnamate of general formula (V), in the presence of a dimeric rhodium complex, in a solvent such as toluene at a temperature of between 25 ° C. and 60 ° C., according to an adaptation of protocols described in the literature (Tom G. Drivers et al J. Am Chem Soc, 2007, 129, 7500-7501; J. Sawyer et al J. Med Chem, 2005, 48, 893-896).
- aromatic or heteroaromatic aldehydes substituted by a silanyl group of general formula (IV), when they are not commercially available, can be obtained from the corresponding aromatic or heteroaromatic aldehydes, preferably masked in the form of an acetal by for example, substituted with a halogen atom such as bromine or iodine, in the position where the silanyl group is to be introduced:
- a disilane such as hexamethyldisilane
- a catalytic amount of a metal complex preferably a palladium complex, such as, for example, tetrakis (triphenylphosphine) palladium
- a base such as potassium carbonate
- HMPT hexamethylphosphoric triamide
- the aromatic or heteroaromatic aldehydes substituted with a silanyl group of the general formula (IV), when they are not commercially available, can also be obtained from the corresponding dihalogenated aromatic or heteroaromatic compounds, such as a dibromo derivative, in the position where the silanyl group is to be introduced, by exchange with an organometallic, such as n-butyllithium.
- the aromatic or heteroaromatic metal thus formed can then react with organohalosilanes or be converted into formylated derivatives by adaptation of the methods described in the literature.
- the reaction is preferably carried out at low temperatures between -110 ° C.
- the invention also relates to the compounds of general formulas (IIa), (Nb), (Ile), (Hd), (lie), (Nf), (Ng) and (Hh ), in which Et represents an ethyl group. These compounds are useful as synthesis intermediates for the compounds of formula (I).
- esters (IIa), (Nb), (Ile), (Hd), (Ile), (Hf), (Hg) and (Hh) are prepared according to the processes described in Examples No. 9, 10, 14, 16, 21, 38, 40 and 41.
- the reaction mixture is stirred for 14 hours at 20 ° C. and is then concentrated under reduced pressure.
- the resulting product is taken up in 100 mL of water.
- the suspension is then extracted with 3 times 30 ml of ethyl acetate.
- the combined organic phases are washed twice with 20 ml of water, dried over sodium sulphate and then concentrated under reduced pressure.
- the product obtained is purified by chromatography on a silica column, eluting with a mixture of dichloromethane and methanol. 290 mg of the expected product are thus isolated.
- PF 186 - 188 0 C
- Example 2 (Compound No. 2)? - [1H-Pyrrolo [2,3-b] pyridin-5-yl] -5-fluoro-1- (3-fluorobenzyl) -1H-indole-2-carboxamide.
- Example 4 (Compound No. 4)? - [1W-pyrrolo [3,2-b] pyridin-6-yl] -5-fluoro-1- (3-fluorobenzyl) -1H-indole-2-carboxamide.
- the crude reaction product is then purified by flash chromatography on a column of silica gel, eluting with a mixture of heptane and ethyl acetate to give 386 mg of 5-trifluoromethyl-1- [(pyridin-4-yl) methyl) )] - 1H-indole-2-carboxylic acid ethyl ester expected in the form of a white solid.
- reaction crude is then diluted with 50 ml of a standard solution of HCl and 100 ml of ethyl acetate.
- the organic phase is separated off, washed with 50 ml of a saturated solution of sodium chloride and then dried over sodium sulphate and concentrated under reduced pressure.
- the resulting product is purified by silica column chromatography. 147 mg of the expected product are thus isolated.
- This compound is prepared, according to a process similar to that described in Example 5.1, by reacting 475 mg (2.459 mmol) of methyl 6-fluoro-1H-indole-2-carboxylate with 0.59 ml (4.918 mmol). 3-methylphenylmethanol in the presence of 0.92 g
- CMBP cyanomethylene tributylphosphorane
- Compound No. 5 was prepared according to a process similar to that described in step 5.2 by reacting 200 mg (0.673 mmol) of 5-fluoro-1 - [(3-methylphenyl) methyl] -1 / - / - methyl indole-2-carboxylate prepared according to the protocol described in step 6.1 with 107 mg (0.807 mmol) of pyrrolo [2,3- ⁇ ] pyridin-5-ylamine in the presence of 0.5 ml (1.01 mmol ) a solution of trimethylaluminum (2M / toluene). The product is collected by filtration, to give 107 mg of expected product
- This compound was prepared according to a process similar to that described in step 5.1. by reacting 390 mg (1.51 mmol) of ethyl 5-trifluoromethyl-1H-pyrrolo [2,3-b] pyridin-2-carboxylate (WO2008107543) with 348 mg (3.02 mmol) of thiazol-2 in the presence of 0.92 g (3.826 mmol) of (cyanomethylene) tributylphosphorane (CMBP). The reaction mixture is then purified by flash column chromatography on silica gel in a mixture of heptane and ethyl acetate to give 446 mg of the expected product in the form of an oil.
- CMBP cyanomethylene tributylphosphorane
- Compound No. 37 was prepared according to a method similar to that described in step 5.2 by reacting 150 mg (0.449 mmol) of 6-trifluoromethyl-1 - [(pyridin-4-yl) methyl)] - 1W- methyl indole-2-carboxylate prepared in the preceding step with 72 mg (0.538 mmol) of pyrrolo [2,3- ⁇ ] pyridin-5-ylamine in the presence of 0.34 ml (0.674 mmol) of a solution of trimethylaluminum (2M / toluene). The product is collected by filtration, to give 99 mg of expected product.
- the reaction medium is hydrolysed by addition, with vigorous stirring, of 100 ml of a solution of ammonium chloride (30% aqueous).
- the product is extracted with three times 50 ml of ethyl acetate.
- the combined organic phases are washed twice with 20 ml of water, dried over sodium sulphate and concentrated under reduced pressure.
- the resulting oil is purified by column chromatography on silica gel eluting with a mixture of heptane and dichloromethane. 4.96 g of the expected product are isolated in the form of a yellow oil.
- This compound was prepared according to a method similar to that described in step 5.1 by reacting 500 mg (1,913 mmol) of ethyl 6-trimethylsilyl-1H-indole-2-carboxylate with 0.52. mL (3.826 mmol) of 3- (trifluoromethyl) phenylmethanol in the presence of 0.92 g (3.826 mmol) of (cyanomethylene) tributylphosphorane (CMBP). The reaction crude is then purified by flash column chromatography on silica gel in a mixture of heptane and ethyl acetate to give 720 mg of the expected product.
- CMBP cyanomethylene tributylphosphorane
- Compound No. 6 was prepared according to a process similar to that described in step 5.2 by reacting 200 mg (0.477 mmol) of 6-trimethylsilyl-1 - [[(3-trifluoromethyl) phenyl] methyl] -1 / ethyl indole-2-carboxylate prepared according to the protocol described in the previous step with 76 mg (0.572 mmol) of pyrrolo [2,3-b] pyridin-5-ylamine in the presence of 0.36 ml ( 0.716 mmol) of a solution of trimethylaluminum (2M / toluene). The product is isolated by purification by flash column chromatography on silica gel in a mixture of heptane and ethyl acetate. 163 mg of expected product is obtained.
- reaction mixture is hydrolyzed at 0 ° C. by successive additions of 10 ml of concentrated HCl solution and 100 ml of water.
- the product is extracted with 3 x 50 mL of CH 2 Cl 2 .
- the combined organic phases are washed with 100 ml of water, dried over Na 2 SO 4 , filtered and evaporated under reduced pressure.
- the crude reaction product is purified by flash chromatography on a column of silica gel, eluting with a gradient of 10 to 20% of CH 2 Cl 2 in heptane. to give 1.82 g of expected 3-trimethylsilyl benzaldehyde as a yellow oil.
- the aqueous phase is extracted with 3 x 50 mL of AcOEt.
- the combined organic phases are washed with water, dried over Na 2 SO 4 and concentrated under reduced pressure.
- the crude reaction product is purified by chromatography on a column of silica gel, eluting with an isocratic mixture of heptane and CH 2 Cl 2 (80/20). 1.7 g of the expected ethyl 2-azido-3- (3-trimethylsilylphenyl) propenoate are thus isolated in the form of a yellow oil.
- reaction mixture After cooling to room temperature, the reaction mixture is filtered through silica gel, eluting with toluene. The filtrate is then concentrated under reduced pressure. The greenish solid obtained is triturated several times in a minimum of heptane until a white powder is obtained. This is dried under pressure reduced to give 0.87 g of the expected ethyl 5-trimethylsilyl-1H-indole-2-carboxylate, in the form of a white powder.
- This compound was prepared according to a process similar to that described in step 5.1 by reacting 0.49 g (1.87 mmol) of ethyl 5-trimethylsilyl-1H-indole-2-carboxylate with 0.51 ml. (3,749 mmol) of 3- (trifluoromethyl) phenylmethanol in the presence of 0.9 g (3.749 mmol) of (cyanomethylene) tributylphosphorane (CMBP). The crude reaction product is then purified by flash column chromatography on silica gel in a mixture of heptane and ethyl acetate to give 730 mg of the expected product.
- CMBP cyanomethylene tributylphosphorane
- Compound No. 7 was prepared according to a process similar to that described in step 5.2 by reacting 200 mg (0.477 mmol) of 5-trimethylsilyl-1 - [[(3-trifluoromethyl) phenyl] methyl] -1H- ethyl indole-2-carboxylate prepared according to the protocol described in step 10.5 with 76 mg (0.572 mmol) of pyrrolo [2,3-f] pyridin-5-ylamine in the presence of 0.36 ml (0.716 mmol) ) a solution of trimethylaluminum (2M / toluene). The product is collected by filtration, to give 107 mg of expected product
- This compound was prepared according to a protocol similar to that described in Example 5.
- Example 13 (Compound No. 20) ⁇ - (1W-Pyrrolo [2,3-b] pyridin-5-yl) -6-trifluoromethyl-1 - [(3-methylphenyl) methyl] -1H-indole-2 carboxamide
- This compound was prepared according to a method similar to that described in step 5.1 by reacting the ethyl 5-trimethylsilyl-1H-indole-2-carboxylate prepared according to a process described in step 10.4 with the (3- methylphenyl) methanol in the presence of (cyanomethylene) tributylphosphorane (CMBP).
- CMBP cyanomethylene tributylphosphorane
- This compound was prepared according to a process similar to that described in step 5.1 by reacting the ethyl 6-trimethylsilyl-1H-indole-2-carboxylate prepared according to a process described in step 9.2 with the (3- methylphenyl) methanol in the presence of (cyanomethylene) tributylphosphorane (CMBP).
- CMBP cyanomethylene tributylphosphorane
- This compound was prepared according to a protocol similar to that described in Example 5.
- This compound was prepared according to a process similar to that described in step 5.1 by reacting the ethyl 6-trimethylsilyl-1H-indole-2-carboxylate prepared according to a process described in step 9.2 with the (thienyl) 2-yl) methanol in the presence of (cyanomethylene) tributylphosphorane (CMBP).
- CMBP cyanomethylene tributylphosphorane
- This compound was prepared according to a protocol similar to that described in Example 5. MP: 203-204 ° C.
- This compound was prepared by stirring for 30 h at 20 ° C., a mixture of 0.5 g (1.24 mol) of compound N O 2 (Example 2) in the presence of 0.66 g (2.74 mmol) of metachloroperbenzoic acid. in 130 ml of dichloromethane. After this time, the mixture is poured into 200 ml of water and 200 ml of dichloromethane. The organic phase is separated, washed once with 100 ml of a saturated solution of sodium hydrogencarbonate, twice with 100 ml of water, dried over magnesium sulphate and then concentrated under reduced pressure. The resulting product is purified by chromatography on a silica column, 0.13 g of expected product is isolated. MP: 260-263 ° C
- Example 29 (Compound No. 14)? - [1-Methyl-2,3-dihydro-1H-pyrrolo [2,3-b] pyridin-5-yl] -5-fluoro-1 - [(3-fluorophenyl) methyl] -1H-indole-2-carboxamide.
- This compound was prepared according to a method similar to that of Example 26.
- This compound was prepared according to a method similar to that of Example 26.
- This compound was prepared according to a method similar to that of Example 26.
- Example 33 (Compound No. 16) N- [1-Methyl-pyrrolo [2,3-b] pyridin-6-yl] -5-fluoro-1 - [(3-fluorophenyl) methyl] -1H-indole-2 carboxamide
- This compound was prepared according to a method similar to that of Example 29.1 from commercially available 6-bromo-1 / - / - pyrrolo [2,3- ⁇ ] pyridine.
- Example 34 (Compound No. 17) N- (1-Methyl-1H-pyrrolo [2,3-b] pyridin-5-yl) -5-fluoro-1 - [[(3-trifluoromethyl) phenyl] methyl] - 1H-indole-2-carboxamide
- This compound was prepared according to a process similar to that described in step 5.1 by reacting the ethyl 5-trimethylsilyl-1H-indole-2-carboxylate prepared according to a process described in step 10.4 with the (thienyl) 2-yl) methanol in the presence of (cyanomethylene) tributylphosphorane (CMBP).
- CMBP cyanomethylene tributylphosphorane
- This compound was prepared according to a process similar to that described in step 5.1 by reacting the ethyl 6-trimethylsilyl-1H-indole-2-carboxylate prepared according to a process described in step 9.2 with the (pyridine) 2-yl) methanol in the presence of (cyanomethylene) tributylphosphorane (CMBP).
- CMBP cyanomethylene tributylphosphorane
- This compound was prepared according to a method similar to that described in step 5.1 by reacting the ethyl 5-trimethylsilyl-1 / - / - indole-2-carboxylate prepared according to a process described in step 10.4 with the (pyridin-2-yl) methanol in the presence of (cyanomethylene) tributylphosphorane (CMBP). The crude reaction product is then purified by flash column chromatography on silica gel to give the expected product.
- CMBP cyanomethylene tributylphosphorane
- This compound was prepared according to a protocol similar to that described in Example 5.
- This compound was prepared according to a protocol similar to that described in the example
- This compound was prepared according to a protocol similar to that described in the example
- This compound was prepared according to a protocol similar to that described in the example
- This compound was prepared according to a protocol similar to that described in Example 5.
- Table 1 which follows illustrates the chemical structures and the physical properties of some examples of compounds according to the invention.
- Z 2 represents a carbon atom bonded to the nitrogen atom of the amide of formula (I);
- n 1;
- R represents a substituent of the phenyl group
- Table 2 which follows illustrates the chemical structures and the physical properties of some examples of compounds according to the invention.
- Z 2 represents a carbon atom bonded to the nitrogen atom of the amide of formula (I); n is 1.
- the compounds according to the invention have been the subject of in vitro and in vivo pharmacological tests which have demonstrated their interest as substances with therapeutic activities. These compounds exhibit an antagonist or agonist activity with respect to TRPV1 (or VR1) receptors.
- DRG dorsal root ganglia
- the measurement chambers (volume 800 ⁇ l) containing the cell preparation are placed on the stage of an inverted microscope (Olympus IMT2) equipped with Hoffman optics (Modulation Contrast, New York) and observed at 400X magnification.
- the chambers are continuously perfused by gravity (2.5 mL / min) using an 8-way solution distributor whose single outlet, consisting of a polyethylene tube (500 ⁇ m opening) is placed within 3 mm of the studied cell.
- the "whole cell" configuration of the patch-clamp technique was used. Borosilicate glass pipettes (5-10 MOhms resistance) are approached from the cell using a 3D piezoelectric micromanipulator (Burleigh, PC1000).
- the application of a 300 nM capsaicin solution causes the DRG cells (voltage set at -70 mV) to produce an incoming cationic current.
- the interval of one minute minimum between two capsaicin applications is respected.
- the compounds of the invention to be tested are applied alone at a given concentration (concentration of 10 nM or 1 nM) for a duration of 4 to 5 minutes, during which several "capsaicin + compound" tests are performed (obtaining the maximum inhibition). The results are expressed as percentage inhibition of the control capsaicin response.
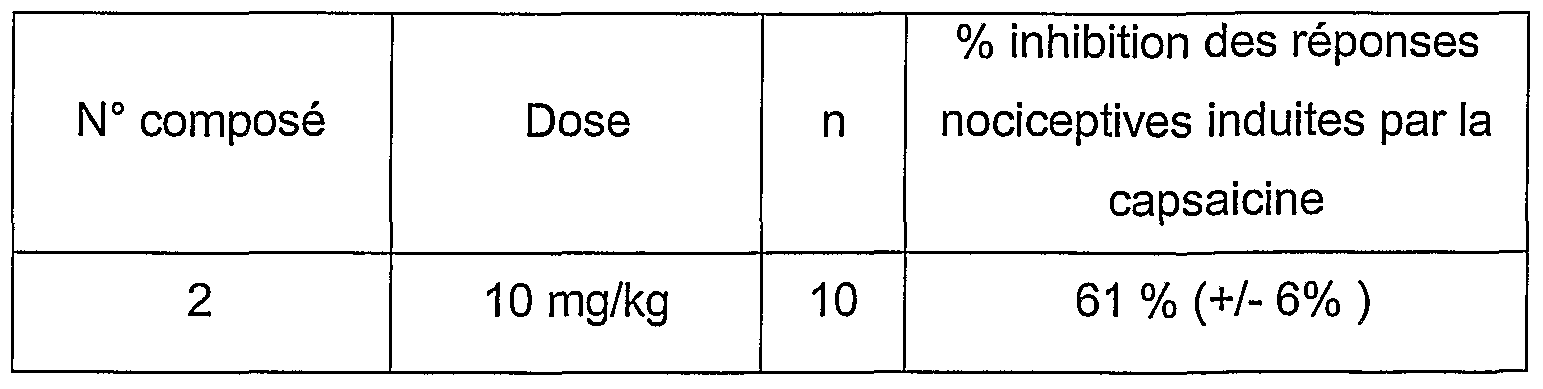
- the percentages of inhibition of the capsaicin response (1 .mu.M) are between 20% and 100% for the most active compounds of the invention tested at concentrations of 0.1 to 100 nM. They are therefore effective antagonists of TRPV1 type receptors. Table 3 gives an example of percentage inhibition obtained with the compounds of the invention.
- (E) -capsaicin is initially diluted to 3 mg / ml in DMSO, and then diluted again for its end use to 1.5 ⁇ g / 20 ⁇ l in saline.
- Solvent administration has no effect on the behavior of the mouse. Capsaicin is injected into one of the rear legs of the animal, at the level of the upper face.
- the compounds to be tested are administered orally 120 minutes before the capsaicin injection. Two hours after the administration of the compounds, the mice are placed in a glass beaker. The nociceptive behavior of the animals is then immediately evaluated by the experimenter and the duration of the behavioral manifestations induced by the capsaicin is timed during a period of 2 minutes (licking and nibbling, total or partial deflection of the injected paw).
- an inhibition corresponding to the average of the nociceptive responses induced by capsaicin is determined, in response to a dose of product studied (expressed in mg / kg) administered orally on a sample of a determined number of mice (not).
- Table 4 gives an example of percentage inhibition obtained with the compounds of the invention.
- the compounds of the invention can therefore be used for the preparation of medicaments, in particular for the preparation of a medicament intended to prevent or treat pathologies in which the TRPV1 type receptors are involved.
- the compounds of the invention may be useful for preventing or treating diseases in which TRPV1 receptors are involved.
- the subject of the invention is medicaments which comprise at least one compound of formula (I), or a pharmaceutically acceptable salt, or a hydrate or a solvate of said compound.
- These drugs may find use in therapy, particularly in the prevention and / or treatment of pain and inflammation, chronic pain, neuropathic (traumatic, diabetic, metabolic, infectious, toxic, induced by cancer treatment or iatrogenic), (osteo-) arthritic, rheumatic, fibromyalgia, back pain, cancer pain, facial neuralgia, headache, migraine, dental pain, burn, stroke sun, bite or sting, postherpetic neuralgia, muscle pain, nerve compression (central and / or peripheral), trauma to the spinal cord and / or brain, ischemia (from marrow and / or brain), neurodegeneration, hemorrhagic (marrow and / or brain) stroke, post-stroke pain.
- the compounds of the invention can also be used to prevent and / or treat metabolic disorders such as diabetes.
- the compounds of the invention can also be used to prevent and / or treat urological disorders such as overactive bladder, bladder hyperflexia, bladder instability, incontinence, emergency urination, urinary incontinence, cystitis, renal colic, pelvic hypersensitivity and pelvic pain.
- urological disorders such as overactive bladder, bladder hyperflexia, bladder instability, incontinence, emergency urination, urinary incontinence, cystitis, renal colic, pelvic hypersensitivity and pelvic pain.
- the compounds of the invention may be useful for preventing and / or treating gynecological disorders such as vulvodynia, pain related to salpingitis, dysmenorrhoea.
- These products can also be used to prevent and / or treat gastrointestinal disorders such as gastroesophageal reflex disorder, stomach ulcer, duodenal ulcer, functional dyspepsia, colitis, IBS, Crohn's disease, pancreatitis, esophagitis, hepatic colic.
- gastrointestinal disorders such as gastroesophageal reflex disorder, stomach ulcer, duodenal ulcer, functional dyspepsia, colitis, IBS, Crohn's disease, pancreatitis, esophagitis, hepatic colic.
- the products of the present invention may be useful in the prevention and / or treatment of respiratory disorders such as asthma, cough, chronic obstructive pulmonary disease (COPD), bronchoconstriction and inflammatory disorders of the sphere. respiratory.
- COPD chronic obstructive pulmonary disease
- These products can also be used to prevent and / or treat psoriasis, pruritis, dermal irritations, eyes or mucous membranes, herpes, shingles.
- the compounds of the invention can also be used to treat depression.
- the compounds of the invention can also be used to treat diseases of the central nervous system such as multiple sclerosis.
- the compounds of the invention could also be used to treat cancers.
- the present invention relates to pharmaceutical compositions comprising, as active ingredient, at least one compound according to the invention.
- These pharmaceutical compositions contain an effective dose of at least one compound according to the invention, or a pharmaceutically acceptable salt, a hydrate or solvate of said compound, as well as at least one pharmaceutically acceptable excipient.
- Said excipients are chosen according to the pharmaceutical form and the desired mode of administration, from the usual excipients which are known to those skilled in the art.
- compositions of the present invention may be administered orally, sublingually, subcutaneously, intramuscularly, intravenously, topically, locally, intratracheally, intranasally, transdermally or rectally.
- These compositions may be administered in unit form, in admixture with conventional pharmaceutical excipients. They are intended to be administered to animals and humans for the prophylaxis or treatment of the disorders or diseases mentioned above.
- Suitable unit dosage forms include oral forms such as tablets, soft or hard capsules, powders, granules and oral solutions or suspensions, sublingual, oral, intratracheal, intraocular, intranasal forms of administration. by inhalation, topical, transdermal, subcutaneous, intramuscular or intravenous administration forms, rectal administration forms and implants.
- oral forms such as tablets, soft or hard capsules, powders, granules and oral solutions or suspensions, sublingual, oral, intratracheal, intraocular, intranasal forms of administration. by inhalation, topical, transdermal, subcutaneous, intramuscular or intravenous administration forms, rectal administration forms and implants.
- the compounds according to the invention can be used in creams, gels, ointments or lotions.
- a unitary form of administration of a compound according to the invention in tablet form may comprise the following components:
- Said unit forms are dosed to allow a daily administration of 0.001 to 30 mg of active ingredient per kg of body weight, according to the dosage form.
- the dosage appropriate to each patient is determined by the physician according to the mode of administration, the weight and the response of said patient.
- the compounds of the invention may also be used for the preparation of medicaments, in particular for the preparation of a medicament intended to prevent or treat pathologies in which the TRPV1 type receptors are involved, as mentioned above.
- the present invention also relates to a method for treating the pathologies indicated above, which comprises the administration to a patient of an effective dose of at least one compound according to the invention, or a pharmaceutically acceptable salt or hydrate or solvate thereof.
Abstract
Description
Claims
Priority Applications (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| UAA201010237A UA102837C2 (ru) | 2008-01-22 | 2009-01-20 | Производные азабициклических карбоксамидов, их получение и их применение в терапии |
| NZ586938A NZ586938A (en) | 2008-01-22 | 2009-01-20 | Azabicyclic carboxamide derivatives, preparation thereof and therapeutic use thereof |
| EP09719730.5A EP2235015B1 (fr) | 2008-01-22 | 2009-01-20 | Derives de carboxam i d es azabicycliques, leur preparation et leur application en therapeutique. |
| BRPI0907381-7A BRPI0907381A2 (pt) | 2008-01-22 | 2009-01-20 | Derivados de carboxamidas azabicíclicos, o respectivo preparo e a respectiva aplicação em terapêutica. |
| AU2009224533A AU2009224533B2 (en) | 2008-01-22 | 2009-01-20 | Azabicyclic carboxamide derivatives, preparation thereof and therapeutic use thereof |
| CN2009801100023A CN101977911A (zh) | 2008-01-22 | 2009-01-20 | 氮杂双环羧酰胺衍生物,及其制备和治疗用途 |
| MX2010007977A MX2010007977A (es) | 2008-01-22 | 2009-01-20 | Derivados de carboxamidas azabiciclicas, su preparacion y su aplicacion en terapeutica. |
| CA2712609A CA2712609C (fr) | 2008-01-22 | 2009-01-20 | Derives de carboxamides azabicycliques, leur preparation et leur application en therapeutique |
| JP2010543539A JP5478512B2 (ja) | 2008-01-22 | 2009-01-20 | アザ二環式カルボキサミド誘導体、これらの調製およびこれらの治療上の使用 |
| EA201070872A EA201070872A1 (ru) | 2008-01-22 | 2009-01-20 | Производные азабициклических карбоксамидов, их получение и их применение в терапии |
| TNP2010000320A TN2010000320A1 (fr) | 2009-01-20 | 2010-07-08 | Derives de carboxamides azabicycliques, leur preparation et leur application en therapeutique |
| EC2010010337A ECSP10010337A (es) | 2008-01-22 | 2010-07-12 | Derivados de carboxamidas azabicíclicas, su preparación y su aplicación en terapéutica |
| IL207078A IL207078A (en) | 2008-01-22 | 2010-07-19 | The history of azabiccal carboxamide, their preparation and their use for treatment |
| US12/840,659 US8354425B2 (en) | 2008-01-22 | 2010-07-21 | Azabicyclic carboxamide derivatives, preparation thereof and therapeutic use thereof |
| ZA2010/05202A ZA201005202B (en) | 2008-01-22 | 2010-07-21 | Azabicyclic carboxamide derivatives,preparation thereof and theapeutic use thereof |
| MA33097A MA32089B1 (fr) | 2008-01-22 | 2010-08-17 | Derives de carboxamides azabicycliques, leur preparation et leur application en therapeutique |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0800308A FR2926554B1 (fr) | 2008-01-22 | 2008-01-22 | Derives de carboxamides azabicycliques, leur preparation et leur application en therapeutique |
| FR08/00308 | 2008-01-22 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/840,659 Continuation US8354425B2 (en) | 2008-01-22 | 2010-07-21 | Azabicyclic carboxamide derivatives, preparation thereof and therapeutic use thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2009112678A2 true WO2009112678A2 (fr) | 2009-09-17 |
| WO2009112678A3 WO2009112678A3 (fr) | 2009-12-30 |
Family
ID=39843820
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2009/000052 WO2009112678A2 (fr) | 2008-01-22 | 2009-01-20 | Derives de carboxam i d es azabicycliques, leur preparation et leur application en therapeutique. |
Country Status (29)
| Country | Link |
|---|---|
| US (1) | US8354425B2 (fr) |
| EP (1) | EP2235015B1 (fr) |
| JP (1) | JP5478512B2 (fr) |
| KR (1) | KR101593711B1 (fr) |
| CN (1) | CN101977911A (fr) |
| AR (1) | AR070206A1 (fr) |
| AU (1) | AU2009224533B2 (fr) |
| BR (1) | BRPI0907381A2 (fr) |
| CA (1) | CA2712609C (fr) |
| CO (1) | CO6280509A2 (fr) |
| CR (1) | CR11546A (fr) |
| DO (1) | DOP2010000213A (fr) |
| EA (1) | EA201070872A1 (fr) |
| EC (1) | ECSP10010337A (fr) |
| FR (1) | FR2926554B1 (fr) |
| HN (1) | HN2010001459A (fr) |
| IL (1) | IL207078A (fr) |
| MA (1) | MA32089B1 (fr) |
| MX (1) | MX2010007977A (fr) |
| MY (1) | MY150576A (fr) |
| NI (1) | NI201000124A (fr) |
| NZ (1) | NZ586938A (fr) |
| PA (1) | PA8812801A1 (fr) |
| PE (1) | PE20091320A1 (fr) |
| TW (1) | TWI428339B (fr) |
| UA (1) | UA102837C2 (fr) |
| UY (1) | UY31606A1 (fr) |
| WO (1) | WO2009112678A2 (fr) |
| ZA (1) | ZA201005202B (fr) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9266886B2 (en) | 2014-02-03 | 2016-02-23 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9481674B1 (en) | 2016-06-10 | 2016-11-01 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9663515B2 (en) | 2014-11-05 | 2017-05-30 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9796710B2 (en) | 2014-10-14 | 2017-10-24 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9845308B2 (en) | 2014-11-05 | 2017-12-19 | Vitae Pharmaceuticals, Inc. | Isoindoline inhibitors of ROR-gamma |
| US10301261B2 (en) | 2015-08-05 | 2019-05-28 | Vitae Pharmaceuticals, Llc | Substituted indoles as modulators of ROR-gamma |
| US10829481B2 (en) | 2016-01-29 | 2020-11-10 | Vitae Pharmaceuticals, Llc | Benzimidazole derivatives as modulators of ROR-gamma |
| US10913739B2 (en) | 2017-07-24 | 2021-02-09 | Vitae Pharmaceuticals, LLC (121374) | Inhibitors of RORγ |
| US11008340B2 (en) | 2015-11-20 | 2021-05-18 | Vitae Pharmaceuticals, Llc | Modulators of ROR-gamma |
| US11186573B2 (en) | 2017-07-24 | 2021-11-30 | Vitae Pharmaceuticals, Llc | Inhibitors of ROR gamma |
Families Citing this family (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EA201070876A1 (ru) * | 2008-01-22 | 2011-04-29 | Такеда Фармасьютикал Компани Лимитед | Трициклические соединения, обладающие антагонистической активностью по отношению к кортикотропин-высвобождающему фактору, и содержащие их фармацевтические композиции |
| FR2926555B1 (fr) | 2008-01-22 | 2010-02-19 | Sanofi Aventis | Derives bicycliques de carboxamides azabicycliques, leur preparation et leur application en therapeutique |
| EA037918B1 (ru) | 2011-12-21 | 2021-06-07 | Новира Терапьютикс, Инк. | Противовирусные агенты против гепатита в |
| SG10201605291WA (en) | 2012-08-28 | 2016-08-30 | Janssen Sciences Ireland Uc | Sulfamoyl-arylamides and the use thereof as medicaments for the treatment of hepatitis b |
| HUE034820T2 (en) | 2013-02-28 | 2018-02-28 | Janssen Sciences Ireland Uc | Sulphamoyl arylamides and their use as medicaments for the treatment of hepatitis B |
| BR112015025052A2 (pt) | 2013-04-03 | 2021-07-06 | Janssen Sciences Ireland Uc | derivados de n-fenil-carboxamida e o seu uso como medicamentos para o tratamento da hepatite b |
| JO3603B1 (ar) | 2013-05-17 | 2020-07-05 | Janssen Sciences Ireland Uc | مشتقات سلفامويل بيرولاميد واستخدامها كادوية لمعالجة التهاب الكبد نوع بي |
| PL2997019T3 (pl) | 2013-05-17 | 2019-03-29 | Janssen Sciences Ireland Uc | Pochodne sulfamoilotiofenamidu i ich zastosowanie jako leków do leczenia wirusowego zapalenia wątroby typu B |
| AP2015008968A0 (en) | 2013-07-25 | 2015-12-31 | Janssen Sciences Ireland Uc | Glyoxamide substituted pyrrolamide derivatives andthe use thereof as medicaments for the treatment of hepatitis b |
| EP3060547B1 (fr) | 2013-10-23 | 2017-10-11 | Janssen Sciences Ireland UC | Dérivés de carboxamide et leur utilisation comme médicaments pour le traitement de l'hépatite b |
| US10392349B2 (en) | 2014-01-16 | 2019-08-27 | Novira Therapeutics, Inc. | Azepane derivatives and methods of treating hepatitis B infections |
| US9169212B2 (en) | 2014-01-16 | 2015-10-27 | Novira Therapeutics, Inc. | Azepane derivatives and methods of treating hepatitis B infections |
| ES2792848T3 (es) | 2014-02-05 | 2020-11-12 | Novira Therapeutics Inc | Politerapia para el tratamiento de infecciones por VHB |
| EA035848B1 (ru) | 2014-02-06 | 2020-08-20 | Янссен Сайенсиз Айрлэнд Юси | Производные сульфамоилпирроламида и их применение в качестве медикаментов для лечения гепатита b |
| EP3271019A1 (fr) | 2015-03-19 | 2018-01-24 | Novira Therapeutics Inc. | Dérivés d'azocane et d'azonane, et méthodes de traitement d'infections provoquées par le virus de l'hépatite b |
| US10875876B2 (en) | 2015-07-02 | 2020-12-29 | Janssen Sciences Ireland Uc | Cyclized sulfamoylarylamide derivatives and the use thereof as medicaments for the treatment of hepatitis B |
| WO2017059059A1 (fr) | 2015-09-29 | 2017-04-06 | Novira Therapeutics, Inc. | Formes cristallines d'un agent antiviral de l'hépatite b |
| SG11201808949SA (en) | 2016-04-15 | 2018-11-29 | Novira Therapeutics Inc | Combinations and methods comprising a capsid assembly inhibitor |
| US10973801B2 (en) | 2018-03-14 | 2021-04-13 | Janssen Sciences Ireland Unlimited Company | Capsid assembly modulator dosing regimen |
| TW202045499A (zh) | 2019-02-22 | 2020-12-16 | 愛爾蘭商健生科學愛爾蘭無限公司 | 用於治療hbv感染或hbv誘發疾病之醯胺衍生物 |
| CA3132554A1 (fr) | 2019-05-06 | 2020-11-12 | Bart Rudolf Romanie Kesteleyn | Derives d'amide utiles dans le traitement d'une infection par le virus de l'hepatite b ou de maladies induites par le virus de l'hepatite b |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2677358B1 (fr) | 1991-06-05 | 1994-09-30 | Rhone Poulenc Chimie | Composes organosilyles et leur procede de synthese. |
| JP2001151771A (ja) | 1999-09-10 | 2001-06-05 | Kyowa Hakko Kogyo Co Ltd | 含窒素芳香族複素環誘導体 |
| US7538121B2 (en) | 2002-02-15 | 2009-05-26 | Glaxo Group Limited | Vanilloid receptor modulators |
| US7338950B2 (en) | 2003-10-07 | 2008-03-04 | Renovis, Inc. | Amide compounds as ion channel ligands and uses thereof |
| CA2553968A1 (fr) | 2004-01-23 | 2005-08-11 | Amgen Inc. | Ligands du recepteur vanilloide et leur application au traitement des douleurs inflammatoires et neuropathiques |
| GB0403780D0 (en) | 2004-02-20 | 2004-03-24 | Astrazeneca Ab | Therapeutic agents |
| BRPI0510980A (pt) * | 2004-05-14 | 2007-11-27 | Pfizer Prod Inc | derivados de pirimidina para o tratamento do crescimento anormal de células |
| FR2874015B1 (fr) * | 2004-08-05 | 2006-09-15 | Sanofi Synthelabo | Derives de n-(1h-indolyl)-1h-indole-2-carboxamides, leur preparation et leur application en therapeutique |
| CA2581516C (fr) | 2004-10-12 | 2013-06-11 | Astrazeneca Ab | Derives de quinazoline |
| FR2880625B1 (fr) * | 2005-01-07 | 2007-03-09 | Sanofi Aventis Sa | Derives de n-(heteroaryl)-1h-indole-2-carboxamides, leur preparation et leur application en therapeutique |
| FR2888847B1 (fr) * | 2005-07-22 | 2007-08-31 | Sanofi Aventis Sa | Derives de n-(heteriaryl)-1-heteorarylalkyl-1h-indole-2- carboxamides, leur preparation et application en therapeutique |
| FR2888848B1 (fr) * | 2005-07-22 | 2007-09-28 | Sanofi Aventis Sa | Derives de n-(arylalkyl)-1h-pyrrrolopyridine-2-carboxamides, leur preparation et leur application en therapeutique |
| FR2897061B1 (fr) | 2006-02-03 | 2010-09-03 | Sanofi Aventis | Derives de n-heteroaryl-carboxamides tricycliques contenant un motif benzimidazole, leur preparation et leur application en therapeutique. |
| FR2904316B1 (fr) * | 2006-07-31 | 2008-09-05 | Sanofi Aventis Sa | Derives de n-(amino-heteroaryl)-1h-indole-2-carboxamides, leur preparation et leur application en therapeutique. |
| FR2910473B1 (fr) * | 2006-12-26 | 2009-02-13 | Sanofi Aventis Sa | Derives de n-(amino-heteroaryl)-1h-pyrrolopyridine-2- carboxamides, leur preparation et leur application en therapeutique. |
| FR2911605B1 (fr) | 2007-01-19 | 2009-04-17 | Sanofi Aventis Sa | Derives de pyrrolopyridine-2-carbowamides, leur preparation et leur application en therapeutique |
| FR2911604B1 (fr) * | 2007-01-19 | 2009-04-17 | Sanofi Aventis Sa | Derives de n-(heteroaryl-1h-indole-2-carboxamides, leur preparation et leur application en therapeutique |
| FR2926555B1 (fr) * | 2008-01-22 | 2010-02-19 | Sanofi Aventis | Derives bicycliques de carboxamides azabicycliques, leur preparation et leur application en therapeutique |
| FR2926556B1 (fr) * | 2008-01-22 | 2010-02-19 | Sanofi Aventis | Derives de carboxamides n-azabicycliques, leur preparation et leur application en therapeutique |
| FR2926553B1 (fr) * | 2008-01-23 | 2010-02-19 | Sanofi Aventis | Derives d'indole-2-carboxamides et d'azaindole-2- carboxamides substitues par un groupe silanyle, leur preparation et leur application en therapeutique |
-
2008
- 2008-01-22 FR FR0800308A patent/FR2926554B1/fr not_active Expired - Fee Related
-
2009
- 2009-01-20 UA UAA201010237A patent/UA102837C2/ru unknown
- 2009-01-20 AU AU2009224533A patent/AU2009224533B2/en not_active Ceased
- 2009-01-20 AR ARP090100166A patent/AR070206A1/es unknown
- 2009-01-20 KR KR1020107018566A patent/KR101593711B1/ko not_active IP Right Cessation
- 2009-01-20 MY MYPI20103439 patent/MY150576A/en unknown
- 2009-01-20 PE PE2009000064A patent/PE20091320A1/es not_active Application Discontinuation
- 2009-01-20 EP EP09719730.5A patent/EP2235015B1/fr not_active Not-in-force
- 2009-01-20 JP JP2010543539A patent/JP5478512B2/ja not_active Expired - Fee Related
- 2009-01-20 EA EA201070872A patent/EA201070872A1/ru unknown
- 2009-01-20 CA CA2712609A patent/CA2712609C/fr not_active Expired - Fee Related
- 2009-01-20 BR BRPI0907381-7A patent/BRPI0907381A2/pt not_active IP Right Cessation
- 2009-01-20 CN CN2009801100023A patent/CN101977911A/zh active Pending
- 2009-01-20 NZ NZ586938A patent/NZ586938A/en not_active IP Right Cessation
- 2009-01-20 WO PCT/FR2009/000052 patent/WO2009112678A2/fr active Application Filing
- 2009-01-20 MX MX2010007977A patent/MX2010007977A/es active IP Right Grant
- 2009-01-21 PA PA20098812801A patent/PA8812801A1/es unknown
- 2009-01-21 TW TW098102268A patent/TWI428339B/zh not_active IP Right Cessation
- 2009-01-22 UY UY031606A patent/UY31606A1/es not_active Application Discontinuation
-
2010
- 2010-06-29 CR CR11546A patent/CR11546A/es not_active Application Discontinuation
- 2010-07-09 DO DO2010000213A patent/DOP2010000213A/es unknown
- 2010-07-12 EC EC2010010337A patent/ECSP10010337A/es unknown
- 2010-07-19 IL IL207078A patent/IL207078A/en not_active IP Right Cessation
- 2010-07-21 US US12/840,659 patent/US8354425B2/en not_active Expired - Fee Related
- 2010-07-21 HN HN2010001459A patent/HN2010001459A/es unknown
- 2010-07-21 NI NI201000124A patent/NI201000124A/es unknown
- 2010-07-21 ZA ZA2010/05202A patent/ZA201005202B/en unknown
- 2010-07-22 CO CO10089487A patent/CO6280509A2/es not_active Application Discontinuation
- 2010-08-17 MA MA33097A patent/MA32089B1/fr unknown
Non-Patent Citations (1)
| Title |
|---|
| None |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11535614B2 (en) | 2014-02-03 | 2022-12-27 | Vitae Pharmaceuticals, Llc | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9624217B2 (en) | 2014-02-03 | 2017-04-18 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10047085B2 (en) | 2014-02-03 | 2018-08-14 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9266886B2 (en) | 2014-02-03 | 2016-02-23 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10399976B2 (en) | 2014-02-03 | 2019-09-03 | Vitae Pharmaceuticals, Llc | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10807980B2 (en) | 2014-02-03 | 2020-10-20 | Vitae Pharmaceuticals, Llc | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9796710B2 (en) | 2014-10-14 | 2017-10-24 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10087184B2 (en) | 2014-10-14 | 2018-10-02 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of RORγ |
| US11001583B2 (en) | 2014-11-05 | 2021-05-11 | Vitae Pharmaceuticals, Llc | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9663515B2 (en) | 2014-11-05 | 2017-05-30 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9845308B2 (en) | 2014-11-05 | 2017-12-19 | Vitae Pharmaceuticals, Inc. | Isoindoline inhibitors of ROR-gamma |
| US10301261B2 (en) | 2015-08-05 | 2019-05-28 | Vitae Pharmaceuticals, Llc | Substituted indoles as modulators of ROR-gamma |
| US10829448B2 (en) | 2015-08-05 | 2020-11-10 | Vitae Pharmaceuticals, Llc | Substituted benzoimidazoles as modulators of ROR-γ |
| US11008340B2 (en) | 2015-11-20 | 2021-05-18 | Vitae Pharmaceuticals, Llc | Modulators of ROR-gamma |
| US10829481B2 (en) | 2016-01-29 | 2020-11-10 | Vitae Pharmaceuticals, Llc | Benzimidazole derivatives as modulators of ROR-gamma |
| US9481674B1 (en) | 2016-06-10 | 2016-11-01 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10913739B2 (en) | 2017-07-24 | 2021-02-09 | Vitae Pharmaceuticals, LLC (121374) | Inhibitors of RORγ |
| US11186573B2 (en) | 2017-07-24 | 2021-11-30 | Vitae Pharmaceuticals, Llc | Inhibitors of ROR gamma |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2235015B1 (fr) | Derives de carboxam i d es azabicycliques, leur preparation et leur application en therapeutique. | |
| CA2712914C (fr) | Derives bicycliques de carboxamides azabicycliques, leur preparation et leur application en therapeutique | |
| EP2235003B1 (fr) | Derives d'indole 2-carboxamides et d'azaindole 2-carboxamides substitues par un groupe silanyle, leur preparation et leur application en therapeutique | |
| CA2712629C (fr) | Derives de carboxamides n-azabicycliques, leur preparation et leur application en therapeutique | |
| FR2897061A1 (fr) | Derives de n-heteroaryl-carboxamides tricycliques contenant un motif benzimidazole, leur preparation et leur application en therapeutique. | |
| EP2125787A1 (fr) | Derives de n-(heteroaryl)-1-heteroaryl-1h-indole-2-carboxamides, leur preparation et leur application en therapeutique | |
| CA2615676C (fr) | Derives de n-(arylalkyl)-1h-pyrrolopyridine-2-carboxamides, leur preparation et leur application en therapeutique | |
| EP2106401B1 (fr) | Derives de pyrrolopyridine-2-carboxamides, leur preparation et leur application en therapeutique | |
| CA2694994C (fr) | Derives de n-heteroaryl-carboxamides tricycliques, leur preparation et leur application en therapeutique |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980110002.3 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09719730 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 201011546 Country of ref document: CR Ref document number: CR2010-011546 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: DZP2010000400 Country of ref document: DZ |
|
| REEP | Request for entry into the european phase |
Ref document number: 2009719730 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009719730 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 207078 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2712609 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 1001114 Country of ref document: KE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010543539 Country of ref document: JP Ref document number: 586938 Country of ref document: NZ Ref document number: 2679/KOLNP/2010 Country of ref document: IN Ref document number: MX/A/2010/007977 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10089487 Country of ref document: CO |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009224533 Country of ref document: AU Ref document number: 12010501676 Country of ref document: PH |
|
| ENP | Entry into the national phase |
Ref document number: 2009224533 Country of ref document: AU Date of ref document: 20090120 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PI 2010003439 Country of ref document: MY |
|
| ENP | Entry into the national phase |
Ref document number: 20107018566 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 201070872 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: A201010237 Country of ref document: UA |
|
| ENP | Entry into the national phase |
Ref document number: PI0907381 Country of ref document: BR Kind code of ref document: A2 Effective date: 20100721 |