WO2006100241A2 - Verfahren zur herstellung von mehrfach ungesättigten c20- und c22-fettsäuren mit mindestens vier doppelbindungen in transgenen pflanzen - Google Patents
Verfahren zur herstellung von mehrfach ungesättigten c20- und c22-fettsäuren mit mindestens vier doppelbindungen in transgenen pflanzen Download PDFInfo
- Publication number
- WO2006100241A2 WO2006100241A2 PCT/EP2006/060913 EP2006060913W WO2006100241A2 WO 2006100241 A2 WO2006100241 A2 WO 2006100241A2 EP 2006060913 W EP2006060913 W EP 2006060913W WO 2006100241 A2 WO2006100241 A2 WO 2006100241A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- desaturase
- nucleic acid
- fatty acids
- acid
- Prior art date
Links
- 239000000194 fatty acid Substances 0.000 title claims abstract description 186
- 235000014113 dietary fatty acids Nutrition 0.000 title claims abstract description 169
- 229930195729 fatty acid Natural products 0.000 title claims abstract description 169
- 150000004665 fatty acids Chemical class 0.000 title claims abstract description 146
- 230000009261 transgenic effect Effects 0.000 title claims abstract description 56
- 238000004519 manufacturing process Methods 0.000 title abstract description 32
- 150000007523 nucleic acids Chemical class 0.000 claims abstract description 236
- 238000000034 method Methods 0.000 claims abstract description 154
- 108020004707 nucleic acids Proteins 0.000 claims abstract description 112
- 102000039446 nucleic acids Human genes 0.000 claims abstract description 112
- 239000013598 vector Substances 0.000 claims abstract description 82
- 230000000694 effects Effects 0.000 claims abstract description 69
- 235000020777 polyunsaturated fatty acids Nutrition 0.000 claims abstract description 67
- 102100034542 Acyl-CoA (8-3)-desaturase Human genes 0.000 claims abstract description 65
- 108010073542 Delta-5 Fatty Acid Desaturase Proteins 0.000 claims abstract description 65
- 102100034544 Acyl-CoA 6-desaturase Human genes 0.000 claims abstract description 64
- 108010037138 Linoleoyl-CoA Desaturase Proteins 0.000 claims abstract description 64
- 108010033653 omega-3 fatty acid desaturase Proteins 0.000 claims abstract description 56
- 101000912235 Rebecca salina Acyl-lipid (7-3)-desaturase Proteins 0.000 claims abstract description 48
- 101000877236 Siganus canaliculatus Acyl-CoA Delta-4 desaturase Proteins 0.000 claims abstract description 48
- 229920001184 polypeptide Polymers 0.000 claims abstract description 40
- 108090000765 processed proteins & peptides Proteins 0.000 claims abstract description 40
- 102000004196 processed proteins & peptides Human genes 0.000 claims abstract description 40
- 239000000203 mixture Substances 0.000 claims abstract description 36
- 241000196324 Embryophyta Species 0.000 claims description 204
- 108090000623 proteins and genes Proteins 0.000 claims description 163
- YZXBAPSDXZZRGB-DOFZRALJSA-N arachidonic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YZXBAPSDXZZRGB-DOFZRALJSA-N 0.000 claims description 94
- MBMBGCFOFBJSGT-KUBAVDMBSA-N all-cis-docosa-4,7,10,13,16,19-hexaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCC(O)=O MBMBGCFOFBJSGT-KUBAVDMBSA-N 0.000 claims description 75
- 230000008569 process Effects 0.000 claims description 69
- 108091028043 Nucleic acid sequence Proteins 0.000 claims description 62
- 150000002632 lipids Chemical class 0.000 claims description 62
- 102000004169 proteins and genes Human genes 0.000 claims description 51
- JAZBEHYOTPTENJ-JLNKQSITSA-N all-cis-5,8,11,14,17-icosapentaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O JAZBEHYOTPTENJ-JLNKQSITSA-N 0.000 claims description 49
- 235000020673 eicosapentaenoic acid Nutrition 0.000 claims description 49
- 229960005135 eicosapentaenoic acid Drugs 0.000 claims description 49
- JAZBEHYOTPTENJ-UHFFFAOYSA-N eicosapentaenoic acid Natural products CCC=CCC=CCC=CCC=CCC=CCCCC(O)=O JAZBEHYOTPTENJ-UHFFFAOYSA-N 0.000 claims description 48
- 235000021342 arachidonic acid Nutrition 0.000 claims description 47
- 229940114079 arachidonic acid Drugs 0.000 claims description 47
- 230000015572 biosynthetic process Effects 0.000 claims description 43
- 235000019198 oils Nutrition 0.000 claims description 42
- 239000003921 oil Substances 0.000 claims description 41
- 235000020669 docosahexaenoic acid Nutrition 0.000 claims description 39
- 229940090949 docosahexaenoic acid Drugs 0.000 claims description 37
- 230000001105 regulatory effect Effects 0.000 claims description 29
- 150000003626 triacylglycerols Chemical class 0.000 claims description 26
- 235000010469 Glycine max Nutrition 0.000 claims description 25
- 150000001413 amino acids Chemical class 0.000 claims description 23
- 235000004977 Brassica sinapistrum Nutrition 0.000 claims description 20
- 240000002791 Brassica napus Species 0.000 claims description 19
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 19
- 240000006240 Linum usitatissimum Species 0.000 claims description 18
- 244000005700 microbiome Species 0.000 claims description 17
- 235000006008 Brassica napus var napus Nutrition 0.000 claims description 15
- 235000003222 Helianthus annuus Nutrition 0.000 claims description 15
- 235000004431 Linum usitatissimum Nutrition 0.000 claims description 15
- 240000005979 Hordeum vulgare Species 0.000 claims description 14
- 235000013305 food Nutrition 0.000 claims description 14
- 230000037356 lipid metabolism Effects 0.000 claims description 14
- 230000004129 fatty acid metabolism Effects 0.000 claims description 13
- 244000105624 Arachis hypogaea Species 0.000 claims description 12
- 235000007340 Hordeum vulgare Nutrition 0.000 claims description 12
- 235000021588 free fatty acids Nutrition 0.000 claims description 12
- 241001465754 Metazoa Species 0.000 claims description 11
- 244000098338 Triticum aestivum Species 0.000 claims description 11
- 235000017060 Arachis glabrata Nutrition 0.000 claims description 10
- 235000010777 Arachis hypogaea Nutrition 0.000 claims description 10
- 235000018262 Arachis monticola Nutrition 0.000 claims description 10
- 240000007594 Oryza sativa Species 0.000 claims description 10
- 235000007164 Oryza sativa Nutrition 0.000 claims description 10
- 240000008042 Zea mays Species 0.000 claims description 10
- 235000020232 peanut Nutrition 0.000 claims description 10
- 238000002360 preparation method Methods 0.000 claims description 10
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims description 9
- 235000014698 Brassica juncea var multisecta Nutrition 0.000 claims description 9
- 235000006618 Brassica rapa subsp oleifera Nutrition 0.000 claims description 9
- 240000001432 Calendula officinalis Species 0.000 claims description 9
- 240000003183 Manihot esculenta Species 0.000 claims description 9
- 241000219925 Oenothera Species 0.000 claims description 9
- 235000002017 Zea mays subsp mays Nutrition 0.000 claims description 9
- 235000009566 rice Nutrition 0.000 claims description 9
- 102000057234 Acyl transferases Human genes 0.000 claims description 8
- 108700016155 Acyl transferases Proteins 0.000 claims description 8
- 235000007689 Borago officinalis Nutrition 0.000 claims description 8
- 235000013162 Cocos nucifera Nutrition 0.000 claims description 8
- 244000060011 Cocos nucifera Species 0.000 claims description 8
- 235000004496 Oenothera biennis Nutrition 0.000 claims description 8
- 240000001090 Papaver somniferum Species 0.000 claims description 8
- 235000021307 Triticum Nutrition 0.000 claims description 8
- 244000075850 Avena orientalis Species 0.000 claims description 7
- 240000001980 Cucurbita pepo Species 0.000 claims description 7
- 235000001950 Elaeis guineensis Nutrition 0.000 claims description 7
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 claims description 7
- AJBZENLMTKDAEK-UHFFFAOYSA-N 3a,5a,5b,8,8,11a-hexamethyl-1-prop-1-en-2-yl-1,2,3,4,5,6,7,7a,9,10,11,11b,12,13,13a,13b-hexadecahydrocyclopenta[a]chrysene-4,9-diol Chemical compound CC12CCC(O)C(C)(C)C1CCC(C1(C)CC3O)(C)C2CCC1C1C3(C)CCC1C(=C)C AJBZENLMTKDAEK-UHFFFAOYSA-N 0.000 claims description 6
- 102000000452 Acetyl-CoA carboxylase Human genes 0.000 claims description 6
- 108010016219 Acetyl-CoA carboxylase Proteins 0.000 claims description 6
- 101710146995 Acyl carrier protein Proteins 0.000 claims description 6
- 235000007319 Avena orientalis Nutrition 0.000 claims description 6
- 235000000832 Ayote Nutrition 0.000 claims description 6
- 235000003880 Calendula Nutrition 0.000 claims description 6
- 229920000742 Cotton Polymers 0.000 claims description 6
- 235000009854 Cucurbita moschata Nutrition 0.000 claims description 6
- 108010087894 Fatty acid desaturases Proteins 0.000 claims description 6
- 240000007049 Juglans regia Species 0.000 claims description 6
- 235000016735 Manihot esculenta subsp esculenta Nutrition 0.000 claims description 6
- 240000007817 Olea europaea Species 0.000 claims description 6
- 235000008753 Papaver somniferum Nutrition 0.000 claims description 6
- 235000016761 Piper aduncum Nutrition 0.000 claims description 6
- 235000007238 Secale cereale Nutrition 0.000 claims description 6
- 244000082988 Secale cereale Species 0.000 claims description 6
- 244000000231 Sesamum indicum Species 0.000 claims description 6
- 239000002537 cosmetic Substances 0.000 claims description 6
- 102000018659 1-Acylglycerophosphocholine O-Acyltransferase Human genes 0.000 claims description 5
- 108010052187 1-Acylglycerophosphocholine O-Acyltransferase Proteins 0.000 claims description 5
- 108010001058 Acyl-CoA Dehydrogenase Proteins 0.000 claims description 5
- 102000002735 Acyl-CoA Dehydrogenase Human genes 0.000 claims description 5
- 102000004539 Acyl-CoA Oxidase Human genes 0.000 claims description 5
- 108020001558 Acyl-CoA oxidase Proteins 0.000 claims description 5
- 102100022089 Acyl-[acyl-carrier-protein] hydrolase Human genes 0.000 claims description 5
- 235000002566 Capsicum Nutrition 0.000 claims description 5
- 235000009804 Cucurbita pepo subsp pepo Nutrition 0.000 claims description 5
- 102000036181 Fatty Acid Elongases Human genes 0.000 claims description 5
- 108010058732 Fatty Acid Elongases Proteins 0.000 claims description 5
- 108010039731 Fatty Acid Synthases Proteins 0.000 claims description 5
- 102100027297 Fatty acid 2-hydroxylase Human genes 0.000 claims description 5
- 102000009114 Fatty acid desaturases Human genes 0.000 claims description 5
- 101000937693 Homo sapiens Fatty acid 2-hydroxylase Proteins 0.000 claims description 5
- 235000009496 Juglans regia Nutrition 0.000 claims description 5
- 102000004882 Lipase Human genes 0.000 claims description 5
- 108090001060 Lipase Proteins 0.000 claims description 5
- 102000003820 Lipoxygenases Human genes 0.000 claims description 5
- 108090000128 Lipoxygenases Proteins 0.000 claims description 5
- 241000208467 Macadamia Species 0.000 claims description 5
- 244000025272 Persea americana Species 0.000 claims description 5
- 235000008184 Piper nigrum Nutrition 0.000 claims description 5
- 235000003434 Sesamum indicum Nutrition 0.000 claims description 5
- 235000010599 Verbascum thapsus Nutrition 0.000 claims description 5
- 108010064894 hydroperoxide lyase Proteins 0.000 claims description 5
- 235000015136 pumpkin Nutrition 0.000 claims description 5
- 235000020234 walnut Nutrition 0.000 claims description 5
- 235000003351 Brassica cretica Nutrition 0.000 claims description 4
- 235000003343 Brassica rupestris Nutrition 0.000 claims description 4
- 241000132536 Cirsium Species 0.000 claims description 4
- 240000007582 Corylus avellana Species 0.000 claims description 4
- 239000006002 Pepper Substances 0.000 claims description 4
- 235000008673 Persea americana Nutrition 0.000 claims description 4
- 235000017804 Piper guineense Nutrition 0.000 claims description 4
- 240000006711 Pistacia vera Species 0.000 claims description 4
- 241001083505 Punica Species 0.000 claims description 4
- 108010056748 acetylenase Proteins 0.000 claims description 4
- 108700021044 acyl-ACP thioesterase Proteins 0.000 claims description 4
- 108060000307 allene oxide cyclase Proteins 0.000 claims description 4
- QKSKPIVNLNLAAV-UHFFFAOYSA-N bis(2-chloroethyl) sulfide Chemical compound ClCCSCCCl QKSKPIVNLNLAAV-UHFFFAOYSA-N 0.000 claims description 4
- 108010022240 delta-8 fatty acid desaturase Proteins 0.000 claims description 4
- 239000003814 drug Substances 0.000 claims description 4
- 230000000813 microbial effect Effects 0.000 claims description 4
- 235000010460 mustard Nutrition 0.000 claims description 4
- 240000004355 Borago officinalis Species 0.000 claims description 3
- 235000001543 Corylus americana Nutrition 0.000 claims description 3
- 235000007466 Corylus avellana Nutrition 0.000 claims description 3
- 235000019714 Triticale Nutrition 0.000 claims description 3
- 244000178289 Verbascum thapsus Species 0.000 claims description 3
- 235000004426 flaxseed Nutrition 0.000 claims description 3
- 235000020233 pistachio Nutrition 0.000 claims description 3
- 238000011084 recovery Methods 0.000 claims description 3
- 235000015112 vegetable and seed oil Nutrition 0.000 claims description 3
- 241000228158 x Triticosecale Species 0.000 claims description 3
- 244000144725 Amygdalus communis Species 0.000 claims description 2
- 235000011437 Amygdalus communis Nutrition 0.000 claims description 2
- WLYGSPLCNKYESI-RSUQVHIMSA-N Carthamin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1[C@@]1(O)C(O)=C(C(=O)\C=C\C=2C=CC(O)=CC=2)C(=O)C(\C=C\2C([C@](O)([C@H]3[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O3)O)C(O)=C(C(=O)\C=C\C=3C=CC(O)=CC=3)C/2=O)=O)=C1O WLYGSPLCNKYESI-RSUQVHIMSA-N 0.000 claims description 2
- 241000208809 Carthamus Species 0.000 claims description 2
- 240000003133 Elaeis guineensis Species 0.000 claims description 2
- 244000299507 Gossypium hirsutum Species 0.000 claims description 2
- 244000020551 Helianthus annuus Species 0.000 claims description 2
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 claims description 2
- 235000020224 almond Nutrition 0.000 claims description 2
- 229910052799 carbon Inorganic materials 0.000 claims description 2
- 235000005822 corn Nutrition 0.000 claims description 2
- 238000002156 mixing Methods 0.000 claims description 2
- 239000008158 vegetable oil Substances 0.000 claims description 2
- 244000056139 Brassica cretica Species 0.000 claims 1
- 244000188595 Brassica sinapistrum Species 0.000 claims 1
- 240000003889 Piper guineense Species 0.000 claims 1
- -1 Δ-6-elongase Proteins 0.000 abstract description 100
- 230000014509 gene expression Effects 0.000 description 97
- 210000004027 cell Anatomy 0.000 description 69
- 235000018102 proteins Nutrition 0.000 description 41
- 239000000047 product Substances 0.000 description 28
- 239000002253 acid Substances 0.000 description 25
- 125000003729 nucleotide group Chemical group 0.000 description 25
- 150000001875 compounds Chemical class 0.000 description 24
- 239000002773 nucleotide Substances 0.000 description 24
- 108020004414 DNA Proteins 0.000 description 23
- 238000003786 synthesis reaction Methods 0.000 description 22
- 235000001014 amino acid Nutrition 0.000 description 21
- 244000068988 Glycine max Species 0.000 description 20
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 20
- 239000013612 plasmid Substances 0.000 description 20
- 239000013604 expression vector Substances 0.000 description 19
- 241000233866 Fungi Species 0.000 description 18
- 229940024606 amino acid Drugs 0.000 description 17
- 238000009396 hybridization Methods 0.000 description 17
- 235000020660 omega-3 fatty acid Nutrition 0.000 description 17
- 230000009466 transformation Effects 0.000 description 16
- 235000011684 Sorghum saccharatum Nutrition 0.000 description 15
- 235000003255 Carthamus tinctorius Nutrition 0.000 description 14
- 244000020518 Carthamus tinctorius Species 0.000 description 14
- 241000195493 Cryptophyta Species 0.000 description 14
- 241001148462 Funaria <moss> Species 0.000 description 14
- 241000208818 Helianthus Species 0.000 description 14
- 241001504286 Physcomitrium Species 0.000 description 14
- 230000001965 increasing effect Effects 0.000 description 14
- 239000000758 substrate Substances 0.000 description 14
- 210000001519 tissue Anatomy 0.000 description 14
- 241000196321 Tetraselmis Species 0.000 description 13
- DTOSIQBPPRVQHS-PDBXOOCHSA-N alpha-linolenic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCC(O)=O DTOSIQBPPRVQHS-PDBXOOCHSA-N 0.000 description 13
- 238000010367 cloning Methods 0.000 description 13
- 235000019387 fatty acid methyl ester Nutrition 0.000 description 13
- 150000004670 unsaturated fatty acids Chemical class 0.000 description 13
- 235000021122 unsaturated fatty acids Nutrition 0.000 description 13
- 102000004190 Enzymes Human genes 0.000 description 12
- 108090000790 Enzymes Proteins 0.000 description 12
- 229940088598 enzyme Drugs 0.000 description 12
- KQQKGWQCNNTQJW-UHFFFAOYSA-N linolenic acid Natural products CC=CCCC=CCC=CCCCCCCCC(O)=O KQQKGWQCNNTQJW-UHFFFAOYSA-N 0.000 description 12
- 239000002609 medium Substances 0.000 description 12
- 229940012843 omega-3 fatty acid Drugs 0.000 description 12
- 238000012546 transfer Methods 0.000 description 12
- 244000025254 Cannabis sativa Species 0.000 description 11
- 235000012766 Cannabis sativa ssp. sativa var. sativa Nutrition 0.000 description 11
- 235000012765 Cannabis sativa ssp. sativa var. spontanea Nutrition 0.000 description 11
- OYHQOLUKZRVURQ-HZJYTTRNSA-N Linoleic acid Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC(O)=O OYHQOLUKZRVURQ-HZJYTTRNSA-N 0.000 description 11
- 238000004458 analytical method Methods 0.000 description 11
- 235000009120 camo Nutrition 0.000 description 11
- 235000005607 chanvre indien Nutrition 0.000 description 11
- 238000006243 chemical reaction Methods 0.000 description 11
- 239000011487 hemp Substances 0.000 description 11
- 235000020778 linoleic acid Nutrition 0.000 description 11
- OYHQOLUKZRVURQ-IXWMQOLASA-N linoleic acid Natural products CCCCC\C=C/C\C=C\CCCCCCCC(O)=O OYHQOLUKZRVURQ-IXWMQOLASA-N 0.000 description 11
- 229960004488 linolenic acid Drugs 0.000 description 11
- LWTDZKXXJRRKDG-KXBFYZLASA-N (-)-phaseollin Chemical compound C1OC2=CC(O)=CC=C2[C@H]2[C@@H]1C1=CC=C3OC(C)(C)C=CC3=C1O2 LWTDZKXXJRRKDG-KXBFYZLASA-N 0.000 description 10
- 241000391889 Ditrichum Species 0.000 description 10
- 235000020661 alpha-linolenic acid Nutrition 0.000 description 10
- 230000002255 enzymatic effect Effects 0.000 description 10
- 239000006014 omega-3 oil Substances 0.000 description 10
- 150000003904 phospholipids Chemical class 0.000 description 10
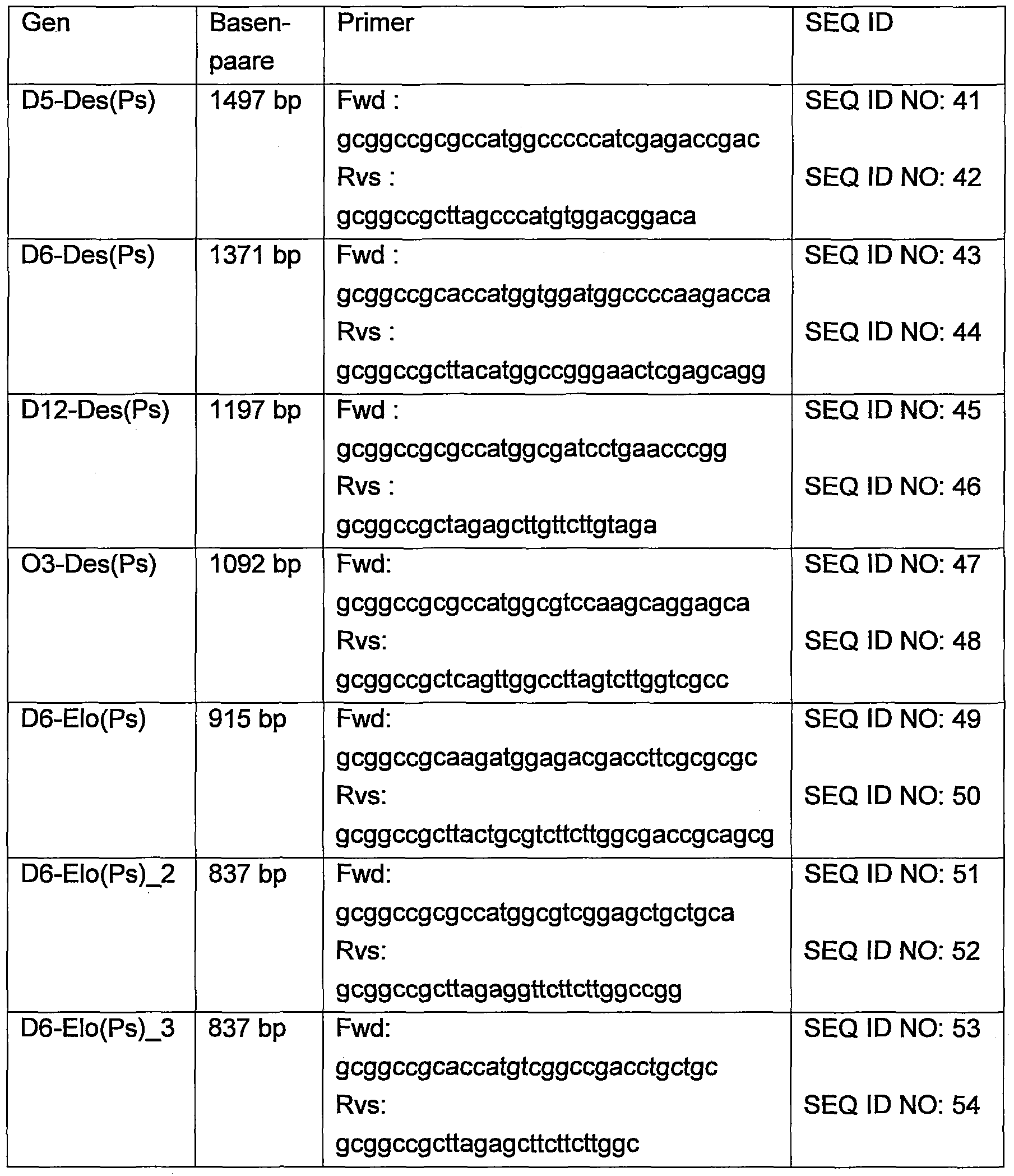
- 239000013615 primer Substances 0.000 description 10
- 241000886188 Entosthodon Species 0.000 description 9
- 239000002299 complementary DNA Substances 0.000 description 9
- 230000001939 inductive effect Effects 0.000 description 9
- YUFFSWGQGVEMMI-JLNKQSITSA-N (7Z,10Z,13Z,16Z,19Z)-docosapentaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCCCCC(O)=O YUFFSWGQGVEMMI-JLNKQSITSA-N 0.000 description 8
- 241001072256 Boraginaceae Species 0.000 description 8
- 241000219198 Brassica Species 0.000 description 8
- 240000000385 Brassica napus var. napus Species 0.000 description 8
- 241000195898 Ceratodon Species 0.000 description 8
- 241001221669 Ostreococcus Species 0.000 description 8
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 8
- 241001530097 Verbascum Species 0.000 description 8
- 125000000539 amino acid group Chemical group 0.000 description 8
- 238000000605 extraction Methods 0.000 description 8
- 239000003925 fat Substances 0.000 description 8
- 235000019197 fats Nutrition 0.000 description 8
- 239000012634 fragment Substances 0.000 description 8
- 125000005456 glyceride group Chemical group 0.000 description 8
- 230000001404 mediated effect Effects 0.000 description 8
- 239000012528 membrane Substances 0.000 description 8
- 230000004048 modification Effects 0.000 description 8
- 238000012986 modification Methods 0.000 description 8
- 235000020665 omega-6 fatty acid Nutrition 0.000 description 8
- 229940033080 omega-6 fatty acid Drugs 0.000 description 8
- 150000004671 saturated fatty acids Chemical class 0.000 description 8
- 241000894007 species Species 0.000 description 8
- 238000013518 transcription Methods 0.000 description 8
- 230000035897 transcription Effects 0.000 description 8
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 7
- 241000589155 Agrobacterium tumefaciens Species 0.000 description 7
- 241000335053 Beta vulgaris Species 0.000 description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 7
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 7
- 240000003768 Solanum lycopersicum Species 0.000 description 7
- 235000016383 Zea mays subsp huehuetenangensis Nutrition 0.000 description 7
- 238000013459 approach Methods 0.000 description 7
- 230000008901 benefit Effects 0.000 description 7
- 150000002148 esters Chemical class 0.000 description 7
- 238000003780 insertion Methods 0.000 description 7
- 230000037431 insertion Effects 0.000 description 7
- 235000009973 maize Nutrition 0.000 description 7
- 239000002243 precursor Substances 0.000 description 7
- 235000003441 saturated fatty acids Nutrition 0.000 description 7
- 241000589158 Agrobacterium Species 0.000 description 6
- 244000099147 Ananas comosus Species 0.000 description 6
- 241000894006 Bacteria Species 0.000 description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- 244000127993 Elaeis melanococca Species 0.000 description 6
- 241000588724 Escherichia coli Species 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 6
- 241000219146 Gossypium Species 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 241000208125 Nicotiana Species 0.000 description 6
- 244000302909 Piper aduncum Species 0.000 description 6
- 235000004443 Ricinus communis Nutrition 0.000 description 6
- 240000006394 Sorghum bicolor Species 0.000 description 6
- 244000138286 Sorghum saccharatum Species 0.000 description 6
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 6
- 244000299461 Theobroma cacao Species 0.000 description 6
- 238000007792 addition Methods 0.000 description 6
- 239000007795 chemical reaction product Substances 0.000 description 6
- 230000006870 function Effects 0.000 description 6
- VZCCETWTMQHEPK-QNEBEIHSSA-N gamma-linolenic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/CCCCC(O)=O VZCCETWTMQHEPK-QNEBEIHSSA-N 0.000 description 6
- 235000020664 gamma-linolenic acid Nutrition 0.000 description 6
- 238000010353 genetic engineering Methods 0.000 description 6
- 150000002327 glycerophospholipids Chemical class 0.000 description 6
- 238000006467 substitution reaction Methods 0.000 description 6
- DCXXMTOCNZCJGO-UHFFFAOYSA-N tristearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(OC(=O)CCCCCCCCCCCCCCCCC)COC(=O)CCCCCCCCCCCCCCCCC DCXXMTOCNZCJGO-UHFFFAOYSA-N 0.000 description 6
- ZUUFLXSNVWQOJW-MBIXAETLSA-N (2e,4e,6e)-octadeca-2,4,6-trienoic acid Chemical compound CCCCCCCCCCC\C=C\C=C\C=C\C(O)=O ZUUFLXSNVWQOJW-MBIXAETLSA-N 0.000 description 5
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 5
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 5
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 5
- 241000220479 Acacia Species 0.000 description 5
- 244000005894 Albizia lebbeck Species 0.000 description 5
- 235000021533 Beta vulgaris Nutrition 0.000 description 5
- 235000011331 Brassica Nutrition 0.000 description 5
- 241000195897 Ceratodon purpureus Species 0.000 description 5
- 241001148463 Funaria hygrometrica Species 0.000 description 5
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 5
- 235000002637 Nicotiana tabacum Nutrition 0.000 description 5
- 239000005642 Oleic acid Substances 0.000 description 5
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 5
- 101710163504 Phaseolin Proteins 0.000 description 5
- 235000010627 Phaseolus vulgaris Nutrition 0.000 description 5
- 244000046052 Phaseolus vulgaris Species 0.000 description 5
- 108700001094 Plant Genes Proteins 0.000 description 5
- 235000002595 Solanum tuberosum Nutrition 0.000 description 5
- 244000061456 Solanum tuberosum Species 0.000 description 5
- 244000062793 Sorghum vulgare Species 0.000 description 5
- 241001491691 Thalassiosira Species 0.000 description 5
- 235000009470 Theobroma cacao Nutrition 0.000 description 5
- 150000007513 acids Chemical class 0.000 description 5
- 230000001580 bacterial effect Effects 0.000 description 5
- 239000002585 base Substances 0.000 description 5
- 238000012217 deletion Methods 0.000 description 5
- 230000037430 deletion Effects 0.000 description 5
- 230000004927 fusion Effects 0.000 description 5
- VZCCETWTMQHEPK-UHFFFAOYSA-N gamma-Linolensaeure Natural products CCCCCC=CCC=CCC=CCCCCC(O)=O VZCCETWTMQHEPK-UHFFFAOYSA-N 0.000 description 5
- 229960002733 gamolenic acid Drugs 0.000 description 5
- 230000002068 genetic effect Effects 0.000 description 5
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 5
- 235000021374 legumes Nutrition 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 230000004060 metabolic process Effects 0.000 description 5
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 5
- 235000021313 oleic acid Nutrition 0.000 description 5
- 210000000056 organ Anatomy 0.000 description 5
- 230000037361 pathway Effects 0.000 description 5
- LWTDZKXXJRRKDG-UHFFFAOYSA-N phaseollin Natural products C1OC2=CC(O)=CC=C2C2C1C1=CC=C3OC(C)(C)C=CC3=C1O2 LWTDZKXXJRRKDG-UHFFFAOYSA-N 0.000 description 5
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 5
- 210000002706 plastid Anatomy 0.000 description 5
- 230000008488 polyadenylation Effects 0.000 description 5
- 238000003259 recombinant expression Methods 0.000 description 5
- 238000012552 review Methods 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- YUFFSWGQGVEMMI-UHFFFAOYSA-N (7Z,10Z,13Z,16Z,19Z)-7,10,13,16,19-docosapentaenoic acid Natural products CCC=CCC=CCC=CCC=CCC=CCCCCCC(O)=O YUFFSWGQGVEMMI-UHFFFAOYSA-N 0.000 description 4
- HOBAELRKJCKHQD-UHFFFAOYSA-N (8Z,11Z,14Z)-8,11,14-eicosatrienoic acid Natural products CCCCCC=CCC=CCC=CCCCCCCC(O)=O HOBAELRKJCKHQD-UHFFFAOYSA-N 0.000 description 4
- 235000011468 Albizia julibrissin Nutrition 0.000 description 4
- 240000007185 Albizia julibrissin Species 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- 241000206761 Bacillariophyta Species 0.000 description 4
- 244000178993 Brassica juncea Species 0.000 description 4
- 235000011332 Brassica juncea Nutrition 0.000 description 4
- 235000014700 Brassica juncea var napiformis Nutrition 0.000 description 4
- 240000007154 Coffea arabica Species 0.000 description 4
- 241000199913 Crypthecodinium Species 0.000 description 4
- 239000003155 DNA primer Substances 0.000 description 4
- 235000021298 Dihomo-γ-linolenic acid Nutrition 0.000 description 4
- 235000021294 Docosapentaenoic acid Nutrition 0.000 description 4
- 241000220485 Fabaceae Species 0.000 description 4
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 4
- 108010068370 Glutens Proteins 0.000 description 4
- 229930186217 Glycolipid Natural products 0.000 description 4
- 241000209219 Hordeum Species 0.000 description 4
- 241000196322 Marchantia Species 0.000 description 4
- 101710202365 Napin Proteins 0.000 description 4
- 241000206731 Phaeodactylum Species 0.000 description 4
- 241000195888 Physcomitrella Species 0.000 description 4
- 241000233614 Phytophthora Species 0.000 description 4
- 240000004713 Pisum sativum Species 0.000 description 4
- 241000591209 Pleuridium Species 0.000 description 4
- 108020004511 Recombinant DNA Proteins 0.000 description 4
- 235000002597 Solanum melongena Nutrition 0.000 description 4
- 244000061458 Solanum melongena Species 0.000 description 4
- 244000202761 Sorghum bicolor subsp verticilliflorum Species 0.000 description 4
- 235000013457 Sorghum bicolor subsp verticilliflorum Nutrition 0.000 description 4
- 235000012308 Tagetes Nutrition 0.000 description 4
- 241000736851 Tagetes Species 0.000 description 4
- 241001122767 Theaceae Species 0.000 description 4
- ISAKRJDGNUQOIC-UHFFFAOYSA-N Uracil Chemical compound O=C1C=CNC(=O)N1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 0.000 description 4
- 230000033228 biological regulation Effects 0.000 description 4
- 230000001851 biosynthetic effect Effects 0.000 description 4
- 230000006696 biosynthetic metabolic pathway Effects 0.000 description 4
- 239000000872 buffer Substances 0.000 description 4
- AZZMGZXNTDTSME-JUZDKLSSSA-M cefotaxime sodium Chemical compound [Na+].N([C@@H]1C(N2C(=C(COC(C)=O)CS[C@@H]21)C([O-])=O)=O)C(=O)\C(=N/OC)C1=CSC(N)=N1 AZZMGZXNTDTSME-JUZDKLSSSA-M 0.000 description 4
- 238000012512 characterization method Methods 0.000 description 4
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 4
- 244000038559 crop plants Species 0.000 description 4
- 238000011161 development Methods 0.000 description 4
- 235000005911 diet Nutrition 0.000 description 4
- HOBAELRKJCKHQD-QNEBEIHSSA-N dihomo-γ-linolenic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/CCCCCCC(O)=O HOBAELRKJCKHQD-QNEBEIHSSA-N 0.000 description 4
- XIVFQYWMMJWUCD-UHFFFAOYSA-N dihydrophaseic acid Natural products C1C(O)CC2(C)OCC1(C)C2(O)C=CC(C)=CC(O)=O XIVFQYWMMJWUCD-UHFFFAOYSA-N 0.000 description 4
- 239000003623 enhancer Substances 0.000 description 4
- 238000004817 gas chromatography Methods 0.000 description 4
- 239000000543 intermediate Substances 0.000 description 4
- 235000005739 manihot Nutrition 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 108020004999 messenger RNA Proteins 0.000 description 4
- 238000010369 molecular cloning Methods 0.000 description 4
- 101150029798 ocs gene Proteins 0.000 description 4
- 241000196307 prasinophytes Species 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 239000013049 sediment Substances 0.000 description 4
- 150000003408 sphingolipids Chemical class 0.000 description 4
- 239000007858 starting material Substances 0.000 description 4
- 238000004809 thin layer chromatography Methods 0.000 description 4
- 101150085703 vir gene Proteins 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- OYHQOLUKZRVURQ-NTGFUMLPSA-N (9Z,12Z)-9,10,12,13-tetratritiooctadeca-9,12-dienoic acid Chemical compound C(CCCCCCC\C(=C(/C\C(=C(/CCCCC)\[3H])\[3H])\[3H])\[3H])(=O)O OYHQOLUKZRVURQ-NTGFUMLPSA-N 0.000 description 3
- TZCPCKNHXULUIY-RGULYWFUSA-N 1,2-distearoyl-sn-glycero-3-phosphoserine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OC[C@H](N)C(O)=O)OC(=O)CCCCCCCCCCCCCCCCC TZCPCKNHXULUIY-RGULYWFUSA-N 0.000 description 3
- HEWZVZIVELJPQZ-UHFFFAOYSA-N 2,2-dimethoxypropane Chemical compound COC(C)(C)OC HEWZVZIVELJPQZ-UHFFFAOYSA-N 0.000 description 3
- 229920001817 Agar Polymers 0.000 description 3
- 241000220433 Albizia Species 0.000 description 3
- 235000007119 Ananas comosus Nutrition 0.000 description 3
- 241000219194 Arabidopsis Species 0.000 description 3
- 241000208838 Asteraceae Species 0.000 description 3
- 101000972350 Bombyx mori Lebocin-4 Proteins 0.000 description 3
- 241000195940 Bryophyta Species 0.000 description 3
- 244000197813 Camelina sativa Species 0.000 description 3
- 240000004160 Capsicum annuum Species 0.000 description 3
- 240000008574 Capsicum frutescens Species 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 108091026890 Coding region Proteins 0.000 description 3
- 102000004163 DNA-directed RNA polymerases Human genes 0.000 description 3
- 241000195899 Ditrichaceae Species 0.000 description 3
- 241000302790 Ditrichum pusillum Species 0.000 description 3
- 241001117772 Elaeagnaceae Species 0.000 description 3
- 102100031780 Endonuclease Human genes 0.000 description 3
- OPGOLNDOMSBSCW-CLNHMMGSSA-N Fursultiamine hydrochloride Chemical compound Cl.C1CCOC1CSSC(\CCO)=C(/C)N(C=O)CC1=CN=C(C)N=C1N OPGOLNDOMSBSCW-CLNHMMGSSA-N 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- ZWZWYGMENQVNFU-UHFFFAOYSA-N Glycerophosphorylserin Natural products OC(=O)C(N)COP(O)(=O)OCC(O)CO ZWZWYGMENQVNFU-UHFFFAOYSA-N 0.000 description 3
- 239000004471 Glycine Substances 0.000 description 3
- 241000238631 Hexapoda Species 0.000 description 3
- 244000017020 Ipomoea batatas Species 0.000 description 3
- 241000627035 Ipomoea tiliacea Species 0.000 description 3
- 241000208822 Lactuca Species 0.000 description 3
- 235000003228 Lactuca sativa Nutrition 0.000 description 3
- 240000008415 Lactuca sativa Species 0.000 description 3
- 235000003127 Lactuca serriola Nutrition 0.000 description 3
- 240000006137 Lactuca serriola Species 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- 241000658379 Manihot esculenta subsp. esculenta Species 0.000 description 3
- 241000736252 Mantoniella Species 0.000 description 3
- 241000736247 Mantoniella squamata Species 0.000 description 3
- 241000235575 Mortierella Species 0.000 description 3
- 244000291473 Musa acuminata Species 0.000 description 3
- 241000128378 Napoleonaea leonensis Species 0.000 description 3
- 241001221668 Ostreococcus tauri Species 0.000 description 3
- 241000206744 Phaeodactylum tricornutum Species 0.000 description 3
- 241000195887 Physcomitrella patens Species 0.000 description 3
- 235000010582 Pisum sativum Nutrition 0.000 description 3
- 241000209504 Poaceae Species 0.000 description 3
- 241000196315 Prasinocladus Species 0.000 description 3
- 108010092799 RNA-directed DNA polymerase Proteins 0.000 description 3
- 238000012300 Sequence Analysis Methods 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- 241000208292 Solanaceae Species 0.000 description 3
- 240000002439 Sorghum halepense Species 0.000 description 3
- 241000233675 Thraustochytrium Species 0.000 description 3
- ATBOMIWRCZXYSZ-XZBBILGWSA-N [1-[2,3-dihydroxypropoxy(hydroxy)phosphoryl]oxy-3-hexadecanoyloxypropan-2-yl] (9e,12e)-octadeca-9,12-dienoate Chemical compound CCCCCCCCCCCCCCCC(=O)OCC(COP(O)(=O)OCC(O)CO)OC(=O)CCCCCCC\C=C\C\C=C\CCCCC ATBOMIWRCZXYSZ-XZBBILGWSA-N 0.000 description 3
- 239000008272 agar Substances 0.000 description 3
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 3
- 230000009286 beneficial effect Effects 0.000 description 3
- 239000006227 byproduct Substances 0.000 description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 3
- 229940088530 claforan Drugs 0.000 description 3
- 238000003776 cleavage reaction Methods 0.000 description 3
- 238000010276 construction Methods 0.000 description 3
- 230000037213 diet Effects 0.000 description 3
- 150000002066 eicosanoids Chemical class 0.000 description 3
- IQLUYYHUNSSHIY-HZUMYPAESA-N eicosatetraenoic acid Chemical compound CCCCCCCCCCC\C=C\C=C\C=C\C=C\C(O)=O IQLUYYHUNSSHIY-HZUMYPAESA-N 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 210000003527 eukaryotic cell Anatomy 0.000 description 3
- 239000013613 expression plasmid Substances 0.000 description 3
- 230000004136 fatty acid synthesis Effects 0.000 description 3
- 238000000855 fermentation Methods 0.000 description 3
- 230000004151 fermentation Effects 0.000 description 3
- 230000002538 fungal effect Effects 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 230000001976 improved effect Effects 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 230000010354 integration Effects 0.000 description 3
- 230000000670 limiting effect Effects 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 235000020978 long-chain polyunsaturated fatty acids Nutrition 0.000 description 3
- 238000002703 mutagenesis Methods 0.000 description 3
- 231100000350 mutagenesis Toxicity 0.000 description 3
- 230000035772 mutation Effects 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 235000015097 nutrients Nutrition 0.000 description 3
- 239000003208 petroleum Substances 0.000 description 3
- 238000003752 polymerase chain reaction Methods 0.000 description 3
- 230000008092 positive effect Effects 0.000 description 3
- 238000003825 pressing Methods 0.000 description 3
- 210000001236 prokaryotic cell Anatomy 0.000 description 3
- 230000006798 recombination Effects 0.000 description 3
- 238000005215 recombination Methods 0.000 description 3
- 230000007017 scission Effects 0.000 description 3
- 238000012163 sequencing technique Methods 0.000 description 3
- 238000010561 standard procedure Methods 0.000 description 3
- JIWBIWFOSCKQMA-UHFFFAOYSA-N stearidonic acid Natural products CCC=CCC=CCC=CCC=CCCCCC(O)=O JIWBIWFOSCKQMA-UHFFFAOYSA-N 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 230000002103 transcriptional effect Effects 0.000 description 3
- 238000001890 transfection Methods 0.000 description 3
- 238000013519 translation Methods 0.000 description 3
- 230000032258 transport Effects 0.000 description 3
- 241000701447 unidentified baculovirus Species 0.000 description 3
- DVSZKTAMJJTWFG-SKCDLICFSA-N (2e,4e,6e,8e,10e,12e)-docosa-2,4,6,8,10,12-hexaenoic acid Chemical compound CCCCCCCCC\C=C\C=C\C=C\C=C\C=C\C=C\C(O)=O DVSZKTAMJJTWFG-SKCDLICFSA-N 0.000 description 2
- DQGMPXYVZZCNDQ-KBPWROHVSA-N (8E,10E,12Z)-octadecatrienoic acid Chemical compound CCCCC\C=C/C=C/C=C/CCCCCCC(O)=O DQGMPXYVZZCNDQ-KBPWROHVSA-N 0.000 description 2
- SAOSKFBYQJLQOS-KTKRTIGZSA-N (9Z)-octadec-9-en-12-ynoic acid Chemical compound CCCCCC#CC\C=C/CCCCCCCC(O)=O SAOSKFBYQJLQOS-KTKRTIGZSA-N 0.000 description 2
- CUXYLFPMQMFGPL-BGDVVUGTSA-N (9Z,11E,13Z)-octadecatrienoic acid Chemical compound CCCC\C=C/C=C/C=C\CCCCCCCC(O)=O CUXYLFPMQMFGPL-BGDVVUGTSA-N 0.000 description 2
- UHPMCKVQTMMPCG-UHFFFAOYSA-N 5,8-dihydroxy-2-methoxy-6-methyl-7-(2-oxopropyl)naphthalene-1,4-dione Chemical compound CC1=C(CC(C)=O)C(O)=C2C(=O)C(OC)=CC(=O)C2=C1O UHPMCKVQTMMPCG-UHFFFAOYSA-N 0.000 description 2
- GZJLLYHBALOKEX-UHFFFAOYSA-N 6-Ketone, O18-Me-Ussuriedine Natural products CC=CCC=CCC=CCC=CCC=CCC=CCCCC(O)=O GZJLLYHBALOKEX-UHFFFAOYSA-N 0.000 description 2
- 241000251468 Actinopterygii Species 0.000 description 2
- 241000734368 Albizia berteroana Species 0.000 description 2
- 235000008060 Albizia julibrissin var julibrissin Nutrition 0.000 description 2
- 244000192797 Albizia julibrissin var. julibrissin Species 0.000 description 2
- 235000011438 Albizia odoratissima Nutrition 0.000 description 2
- 108091093088 Amplicon Proteins 0.000 description 2
- 241000208223 Anacardiaceae Species 0.000 description 2
- 244000226021 Anacardium occidentale Species 0.000 description 2
- 241000208173 Apiaceae Species 0.000 description 2
- 241000219195 Arabidopsis thaliana Species 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- 241000209764 Avena fatua Species 0.000 description 2
- 235000007320 Avena fatua Nutrition 0.000 description 2
- 241000219495 Betulaceae Species 0.000 description 2
- 241000722877 Borago Species 0.000 description 2
- 241000219193 Brassicaceae Species 0.000 description 2
- 241000234670 Bromeliaceae Species 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- 235000005881 Calendula officinalis Nutrition 0.000 description 2
- 235000016401 Camelina Nutrition 0.000 description 2
- 241001181921 Camissonia Species 0.000 description 2
- 241000218235 Cannabaceae Species 0.000 description 2
- 241000218236 Cannabis Species 0.000 description 2
- 235000008534 Capsicum annuum var annuum Nutrition 0.000 description 2
- 235000002283 Capsicum annuum var aviculare Nutrition 0.000 description 2
- 235000002568 Capsicum frutescens Nutrition 0.000 description 2
- 235000009467 Carica papaya Nutrition 0.000 description 2
- 240000006432 Carica papaya Species 0.000 description 2
- 241000219172 Caricaceae Species 0.000 description 2
- 241000978753 Cathormion Species 0.000 description 2
- 235000005940 Centaurea cyanus Nutrition 0.000 description 2
- 240000004385 Centaurea cyanus Species 0.000 description 2
- 241000282994 Cervidae Species 0.000 description 2
- 241000871189 Chenopodiaceae Species 0.000 description 2
- 235000007542 Cichorium intybus Nutrition 0.000 description 2
- 244000298479 Cichorium intybus Species 0.000 description 2
- 241000223782 Ciliophora Species 0.000 description 2
- 241000737241 Cocos Species 0.000 description 2
- 108020004705 Codon Proteins 0.000 description 2
- 101800004637 Communis Proteins 0.000 description 2
- 241001442589 Convoluta Species 0.000 description 2
- 241000207782 Convolvulaceae Species 0.000 description 2
- 241000207892 Convolvulus Species 0.000 description 2
- 241001531245 Crypthecodiniaceae Species 0.000 description 2
- 241000219104 Cucurbitaceae Species 0.000 description 2
- 244000019459 Cynara cardunculus Species 0.000 description 2
- 235000019106 Cynara scolymus Nutrition 0.000 description 2
- 108010066133 D-octopine dehydrogenase Proteins 0.000 description 2
- 102000053602 DNA Human genes 0.000 description 2
- 244000000626 Daucus carota Species 0.000 description 2
- 241000391895 Distichium Species 0.000 description 2
- 241000805646 Ditrichum montanum Species 0.000 description 2
- 241001650498 Eccremidium Species 0.000 description 2
- 241001508399 Elaeagnus Species 0.000 description 2
- 241000208421 Ericaceae Species 0.000 description 2
- 241000221017 Euphorbiaceae Species 0.000 description 2
- 241001134131 Funaria calvescens Species 0.000 description 2
- 241001484413 Funaria microstoma Species 0.000 description 2
- 241000223218 Fusarium Species 0.000 description 2
- 241000208150 Geraniaceae Species 0.000 description 2
- 108010070675 Glutathione transferase Proteins 0.000 description 2
- 102000005720 Glutathione transferase Human genes 0.000 description 2
- JZNWSCPGTDBMEW-UHFFFAOYSA-N Glycerophosphorylethanolamin Natural products NCCOP(O)(=O)OCC(O)CO JZNWSCPGTDBMEW-UHFFFAOYSA-N 0.000 description 2
- 241000053425 Heteromastix Species 0.000 description 2
- 241000250396 Hordeum secalinum Species 0.000 description 2
- 206010061218 Inflammation Diseases 0.000 description 2
- 240000002329 Inga feuillei Species 0.000 description 2
- 241000758791 Juglandaceae Species 0.000 description 2
- 241000758789 Juglans Species 0.000 description 2
- 235000013757 Juglans Nutrition 0.000 description 2
- 241000194328 Kalmia Species 0.000 description 2
- 241000193986 Kalmia latifolia Species 0.000 description 2
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 2
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 2
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 2
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 2
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 2
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 2
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 2
- 241000218195 Lauraceae Species 0.000 description 2
- 244000147568 Laurus nobilis Species 0.000 description 2
- 241000208202 Linaceae Species 0.000 description 2
- 241000208204 Linum Species 0.000 description 2
- 244000039622 Linum bienne Species 0.000 description 2
- 241000551441 Linum grandiflorum Species 0.000 description 2
- 241000254023 Locusta Species 0.000 description 2
- 241000219071 Malvaceae Species 0.000 description 2
- 241001093152 Mangifera Species 0.000 description 2
- 240000004658 Medicago sativa Species 0.000 description 2
- 235000017587 Medicago sativa ssp. sativa Nutrition 0.000 description 2
- 241000234295 Musa Species 0.000 description 2
- 235000018290 Musa x paradisiaca Nutrition 0.000 description 2
- NWBJYWHLCVSVIJ-UHFFFAOYSA-N N-benzyladenine Chemical compound N=1C=NC=2NC=NC=2C=1NCC1=CC=CC=C1 NWBJYWHLCVSVIJ-UHFFFAOYSA-N 0.000 description 2
- 241001442227 Nephroselmis Species 0.000 description 2
- 241001442226 Nephroselmis olivacea Species 0.000 description 2
- 108091092724 Noncoding DNA Proteins 0.000 description 2
- 101710089395 Oleosin Proteins 0.000 description 2
- 241000277275 Oncorhynchus mykiss Species 0.000 description 2
- 108700026244 Open Reading Frames Proteins 0.000 description 2
- 241000209117 Panicum Species 0.000 description 2
- 235000006443 Panicum miliaceum subsp. miliaceum Nutrition 0.000 description 2
- 235000009037 Panicum miliaceum subsp. ruderale Nutrition 0.000 description 2
- 235000011236 Persea americana var americana Nutrition 0.000 description 2
- 108010002747 Pfu DNA polymerase Proteins 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 241000151569 Physcomitrella readeri Species 0.000 description 2
- 241000948155 Phytophthora sojae Species 0.000 description 2
- 235000003447 Pistacia vera Nutrition 0.000 description 2
- 241000596422 Pithecellobium Species 0.000 description 2
- 241001309997 Pleuridium subulatum Species 0.000 description 2
- 241000147095 Prasinococcus Species 0.000 description 2
- 244000294611 Punica granatum Species 0.000 description 2
- 241001509341 Pyramimonas Species 0.000 description 2
- MUPFEKGTMRGPLJ-RMMQSMQOSA-N Raffinose Natural products O(C[C@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@@H](O[C@@]2(CO)[C@H](O)[C@@H](O)[C@@H](CO)O2)O1)[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 MUPFEKGTMRGPLJ-RMMQSMQOSA-N 0.000 description 2
- 241000124033 Salix Species 0.000 description 2
- 241000196303 Scherffelia Species 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 241000534520 Stenocarpus Species 0.000 description 2
- PQRKPYLNZGDCFH-UHFFFAOYSA-N Sterculic-saeure Natural products CCCCCCCCC1=C(CCCCCCCC(O)=O)C1 PQRKPYLNZGDCFH-UHFFFAOYSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 241000894100 Tetraselmis chuii Species 0.000 description 2
- 241000405713 Tetraselmis suecica Species 0.000 description 2
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 2
- 239000004473 Threonine Substances 0.000 description 2
- 241000723873 Tobacco mosaic virus Species 0.000 description 2
- 108700019146 Transgenes Proteins 0.000 description 2
- 241000391886 Trichodon <moss> Species 0.000 description 2
- 241001233593 Trichodon cylindricus Species 0.000 description 2
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 2
- MUPFEKGTMRGPLJ-UHFFFAOYSA-N UNPD196149 Natural products OC1C(O)C(CO)OC1(CO)OC1C(O)C(O)C(O)C(COC2C(C(O)C(O)C(CO)O2)O)O1 MUPFEKGTMRGPLJ-UHFFFAOYSA-N 0.000 description 2
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 2
- 241001661641 Verrucosa Species 0.000 description 2
- 241000219873 Vicia Species 0.000 description 2
- 108020000999 Viral RNA Proteins 0.000 description 2
- 241000700605 Viruses Species 0.000 description 2
- ZSLZBFCDCINBPY-ZSJPKINUSA-N acetyl-CoA Chemical compound O[C@@H]1[C@H](OP(O)(O)=O)[C@@H](COP(O)(=O)OP(O)(=O)OCC(C)(C)[C@@H](O)C(=O)NCCC(=O)NCCSC(=O)C)O[C@H]1N1C2=NC=NC(N)=C2N=C1 ZSLZBFCDCINBPY-ZSJPKINUSA-N 0.000 description 2
- 125000002252 acyl group Chemical group 0.000 description 2
- 238000000246 agarose gel electrophoresis Methods 0.000 description 2
- 230000003321 amplification Effects 0.000 description 2
- 238000000137 annealing Methods 0.000 description 2
- 238000004061 bleaching Methods 0.000 description 2
- 239000001511 capsicum annuum Substances 0.000 description 2
- 230000006652 catabolic pathway Effects 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 238000004113 cell culture Methods 0.000 description 2
- 238000006482 condensation reaction Methods 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000004925 denaturation Methods 0.000 description 2
- 230000036425 denaturation Effects 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- KAUVQQXNCKESLC-UHFFFAOYSA-N docosahexaenoic acid (DHA) Natural products COC(=O)C(C)NOCC1=CC=CC=C1 KAUVQQXNCKESLC-UHFFFAOYSA-N 0.000 description 2
- 238000004520 electroporation Methods 0.000 description 2
- 210000002257 embryonic structure Anatomy 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 239000012467 final product Substances 0.000 description 2
- 235000019688 fish Nutrition 0.000 description 2
- 229940013317 fish oils Drugs 0.000 description 2
- 239000005350 fused silica glass Substances 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 238000012239 gene modification Methods 0.000 description 2
- 230000005017 genetic modification Effects 0.000 description 2
- 235000013617 genetically modified food Nutrition 0.000 description 2
- 239000008103 glucose Substances 0.000 description 2
- 210000004209 hair Anatomy 0.000 description 2
- 208000019622 heart disease Diseases 0.000 description 2
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 2
- 239000005556 hormone Substances 0.000 description 2
- 229940088597 hormone Drugs 0.000 description 2
- VKOBVWXKNCXXDE-UHFFFAOYSA-N icosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCC(O)=O VKOBVWXKNCXXDE-UHFFFAOYSA-N 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 239000000411 inducer Substances 0.000 description 2
- 230000004054 inflammatory process Effects 0.000 description 2
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 2
- 229960000310 isoleucine Drugs 0.000 description 2
- 229930027917 kanamycin Natural products 0.000 description 2
- SBUJHOSQTJFQJX-NOAMYHISSA-N kanamycin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CN)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O2)O)[C@H](N)C[C@@H]1N SBUJHOSQTJFQJX-NOAMYHISSA-N 0.000 description 2
- 229960000318 kanamycin Drugs 0.000 description 2
- 229930182823 kanamycin A Natural products 0.000 description 2
- 150000004668 long chain fatty acids Chemical class 0.000 description 2
- HPSSZFFAYWBIPY-UHFFFAOYSA-N malvalic acid Chemical compound CCCCCCCCC1=C(CCCCCCC(O)=O)C1 HPSSZFFAYWBIPY-UHFFFAOYSA-N 0.000 description 2
- 210000001161 mammalian embryo Anatomy 0.000 description 2
- 238000003801 milling Methods 0.000 description 2
- 150000002759 monoacylglycerols Chemical class 0.000 description 2
- 239000006870 ms-medium Substances 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 238000003199 nucleic acid amplification method Methods 0.000 description 2
- 235000016709 nutrition Nutrition 0.000 description 2
- 150000002894 organic compounds Chemical class 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- IJTNSXPMYKJZPR-UHFFFAOYSA-N parinaric acid Chemical compound CCC=CC=CC=CC=CCCCCCCCC(O)=O IJTNSXPMYKJZPR-UHFFFAOYSA-N 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 2
- 150000008104 phosphatidylethanolamines Chemical class 0.000 description 2
- 150000003905 phosphatidylinositols Chemical class 0.000 description 2
- 102000054765 polymorphisms of proteins Human genes 0.000 description 2
- MUPFEKGTMRGPLJ-ZQSKZDJDSA-N raffinose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO[C@@H]2[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO)O2)O)O1 MUPFEKGTMRGPLJ-ZQSKZDJDSA-N 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 230000001850 reproductive effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 230000028201 sequestering of triglyceride Effects 0.000 description 2
- 239000002689 soil Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- GVZXZHWIIXHZOB-UHFFFAOYSA-N tariric acid Chemical compound CCCCCCCCCCCC#CCCCCC(O)=O GVZXZHWIIXHZOB-UHFFFAOYSA-N 0.000 description 2
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 2
- 238000011144 upstream manufacturing Methods 0.000 description 2
- 229940035893 uracil Drugs 0.000 description 2
- 239000004474 valine Substances 0.000 description 2
- 239000013603 viral vector Substances 0.000 description 2
- 230000003612 virological effect Effects 0.000 description 2
- 210000005253 yeast cell Anatomy 0.000 description 2
- XMVQWNRDPAAMJB-UHFFFAOYSA-N (+)-13-Cyclopent-2-enyl-tridecansaeure Natural products OC(=O)CCCCCCCCCCCCC1CCC=C1 XMVQWNRDPAAMJB-UHFFFAOYSA-N 0.000 description 1
- LGHXTTIAZFVCCU-SSVNFBSYSA-N (2E,4E,6E,8E)-octadeca-2,4,6,8-tetraenoic acid Chemical compound CCCCCCCCC\C=C\C=C\C=C\C=C\C(O)=O LGHXTTIAZFVCCU-SSVNFBSYSA-N 0.000 description 1
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 1
- HPSWUFMMLKGKDS-DNKOKRCQSA-N (2e,4e,6e,8e,10e,12e)-tetracosa-2,4,6,8,10,12-hexaenoic acid Chemical compound CCCCCCCCCCC\C=C\C=C\C=C\C=C\C=C\C=C\C(O)=O HPSWUFMMLKGKDS-DNKOKRCQSA-N 0.000 description 1
- HXQHFNIKBKZGRP-URPRIDOGSA-N (5Z,9Z,12Z)-octadecatrienoic acid Chemical compound CCCCC\C=C/C\C=C/CC\C=C/CCCC(O)=O HXQHFNIKBKZGRP-URPRIDOGSA-N 0.000 description 1
- WBHHMMIMDMUBKC-XFXZXTDPSA-N (9Z)-12-hydroxyoctadec-9-enoic acid Chemical compound CCCCCCC(O)C\C=C/CCCCCCCC(O)=O WBHHMMIMDMUBKC-XFXZXTDPSA-N 0.000 description 1
- 239000001195 (9Z,12Z,15Z)-octadeca-9,12,15-trienoic acid Substances 0.000 description 1
- XMVQWNRDPAAMJB-QGZVFWFLSA-N (S)-chaulmoogric acid Chemical compound OC(=O)CCCCCCCCCCCC[C@H]1CCC=C1 XMVQWNRDPAAMJB-QGZVFWFLSA-N 0.000 description 1
- QHDHNVFIKWGRJR-UHFFFAOYSA-N 1-cyclohexenol Chemical compound OC1=CCCCC1 QHDHNVFIKWGRJR-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- GYLJVPBWGZUNMB-AATRIKPKSA-N 13,14-dihydrooropheic acid Chemical compound OC(=O)CCCCCCCC#CC#C\C=C\CCC=C GYLJVPBWGZUNMB-AATRIKPKSA-N 0.000 description 1
- GUMOJENFFHZAFP-UHFFFAOYSA-N 2-Ethoxynaphthalene Chemical compound C1=CC=CC2=CC(OCC)=CC=C21 GUMOJENFFHZAFP-UHFFFAOYSA-N 0.000 description 1
- AAEZMHSWRQVQEK-UHFFFAOYSA-N 2-methylideneoxirane Chemical compound C=C1CO1 AAEZMHSWRQVQEK-UHFFFAOYSA-N 0.000 description 1
- 101710099475 3'-phosphoadenosine 5'-phosphate phosphatase Proteins 0.000 description 1
- SOWUYFYWTFVKPP-UHFFFAOYSA-N 4,4-dimethoxy-5h-1,3-oxazole Chemical class COC1(OC)COC=N1 SOWUYFYWTFVKPP-UHFFFAOYSA-N 0.000 description 1
- PIFPCDRPHCQLSJ-WYIJOVFWSA-N 4,8,12,15,19-Docosapentaenoic acid Chemical compound CC\C=C\CC\C=C\C\C=C\CC\C=C\CC\C=C\CCC(O)=O PIFPCDRPHCQLSJ-WYIJOVFWSA-N 0.000 description 1
- NMAKJOWVEDTHOA-UHFFFAOYSA-N 4-(chloromethyl)-1,3-thiazol-2-amine;hydron;chloride Chemical compound Cl.NC1=NC(CCl)=CS1 NMAKJOWVEDTHOA-UHFFFAOYSA-N 0.000 description 1
- FBUKMFOXMZRGRB-UHFFFAOYSA-N 8-(3-oct-2-enyloxiran-2-yl)octanoic acid Chemical compound CCCCCC=CCC1OC1CCCCCCCC(O)=O FBUKMFOXMZRGRB-UHFFFAOYSA-N 0.000 description 1
- SZNVIDWMWMWUCY-UHFFFAOYSA-N 8-(5-hexylfuran-2-yl)octanoic acid Chemical compound CCCCCCC1=CC=C(CCCCCCCC(O)=O)O1 SZNVIDWMWMWUCY-UHFFFAOYSA-N 0.000 description 1
- XKRTYIHZFHFKMC-UHFFFAOYSA-N 9-methylheptadec-8-enoic acid Chemical compound CC(=CCCCCCCC(=O)O)CCCCCCCC XKRTYIHZFHFKMC-UHFFFAOYSA-N 0.000 description 1
- 108010013043 Acetylesterase Proteins 0.000 description 1
- 241000712946 Adenolinum Species 0.000 description 1
- 102100039239 Amidophosphoribosyltransferase Human genes 0.000 description 1
- 108010039224 Amidophosphoribosyltransferase Proteins 0.000 description 1
- 241000611184 Amphora Species 0.000 description 1
- 241000693997 Anacardium Species 0.000 description 1
- 235000001271 Anacardium Nutrition 0.000 description 1
- 235000001274 Anacardium occidentale Nutrition 0.000 description 1
- 241000744007 Andropogon Species 0.000 description 1
- 241000886702 Aphanorrhegma Species 0.000 description 1
- 108700021822 Arabidopsis oleosin Proteins 0.000 description 1
- 235000003911 Arachis Nutrition 0.000 description 1
- 241000607305 Arctica Species 0.000 description 1
- 241000233788 Arecaceae Species 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- 241001663937 Astomiopsis Species 0.000 description 1
- 241000972773 Aulopiformes Species 0.000 description 1
- 235000005781 Avena Nutrition 0.000 description 1
- 235000009393 Avena byzantina Nutrition 0.000 description 1
- 240000000372 Avena hybrida Species 0.000 description 1
- 235000009123 Avena hybrida Nutrition 0.000 description 1
- 241000193830 Bacillus <bacterium> Species 0.000 description 1
- 101001074429 Bacillus subtilis (strain 168) Polyketide biosynthesis acyltransferase homolog PksD Proteins 0.000 description 1
- 101000936617 Bacillus velezensis (strain DSM 23117 / BGSC 10A6 / FZB42) Polyketide biosynthesis acyltransferase homolog BaeD Proteins 0.000 description 1
- 235000016068 Berberis vulgaris Nutrition 0.000 description 1
- 241000219310 Beta vulgaris subsp. vulgaris Species 0.000 description 1
- 239000002028 Biomass Substances 0.000 description 1
- 108010018763 Biotin carboxylase Proteins 0.000 description 1
- 244000293889 Bombax malabaricum Species 0.000 description 1
- 235000011303 Brassica alboglabra Nutrition 0.000 description 1
- 235000008427 Brassica arvensis Nutrition 0.000 description 1
- 244000024671 Brassica kaber Species 0.000 description 1
- 235000011291 Brassica nigra Nutrition 0.000 description 1
- 244000180419 Brassica nigra Species 0.000 description 1
- 240000007124 Brassica oleracea Species 0.000 description 1
- 235000011302 Brassica oleracea Nutrition 0.000 description 1
- 240000008100 Brassica rapa Species 0.000 description 1
- 235000011292 Brassica rapa Nutrition 0.000 description 1
- DPUOLQHDNGRHBS-UHFFFAOYSA-N Brassidinsaeure Natural products CCCCCCCCC=CCCCCCCCCCCCC(O)=O DPUOLQHDNGRHBS-UHFFFAOYSA-N 0.000 description 1
- 241000234673 Bromelia Species 0.000 description 1
- 235000004936 Bromus mango Nutrition 0.000 description 1
- IAUSBCHPOLWRET-UHFFFAOYSA-N C1CP(C=C1)CC(=O)C(=O)O Chemical compound C1CP(C=C1)CC(=O)C(=O)O IAUSBCHPOLWRET-UHFFFAOYSA-N 0.000 description 1
- 241000244202 Caenorhabditis Species 0.000 description 1
- 241000244203 Caenorhabditis elegans Species 0.000 description 1
- 241001126923 Calcarea Species 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- 235000014595 Camelina sativa Nutrition 0.000 description 1
- 241000209507 Camellia Species 0.000 description 1
- 235000002567 Capsicum annuum Nutrition 0.000 description 1
- 244000145132 Capsicum annuum var. aviculare Species 0.000 description 1
- 235000013303 Capsicum annuum var. frutescens Nutrition 0.000 description 1
- 235000002284 Capsicum baccatum var baccatum Nutrition 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- 235000014649 Carica monoica Nutrition 0.000 description 1
- 244000132069 Carica monoica Species 0.000 description 1
- 102000014914 Carrier Proteins Human genes 0.000 description 1
- 241000701489 Cauliflower mosaic virus Species 0.000 description 1
- 241000132570 Centaurea Species 0.000 description 1
- 241000894903 Ceratodon heterophyllus Species 0.000 description 1
- 241001414754 Chroothece Species 0.000 description 1
- 241001650501 Chrysoblastella Species 0.000 description 1
- 241000359391 Chrysoblastella chilensis Species 0.000 description 1
- 241000723343 Cichorium Species 0.000 description 1
- 108020004638 Circular DNA Proteins 0.000 description 1
- 235000002548 Cistus Nutrition 0.000 description 1
- 241000984090 Cistus Species 0.000 description 1
- PIFPCDRPHCQLSJ-UHFFFAOYSA-N Clupanodonic acid Natural products CCC=CCCC=CCC=CCCC=CCCC=CCCC(O)=O PIFPCDRPHCQLSJ-UHFFFAOYSA-N 0.000 description 1
- 241000252203 Clupea harengus Species 0.000 description 1
- 241000723377 Coffea Species 0.000 description 1
- 235000007460 Coffea arabica Nutrition 0.000 description 1
- 241000228031 Coffea liberica Species 0.000 description 1
- 244000016593 Coffea robusta Species 0.000 description 1
- 235000002187 Coffea robusta Nutrition 0.000 description 1
- 241000248395 Colpidium Species 0.000 description 1
- 241000723382 Corylus Species 0.000 description 1
- 235000011747 Corylus colurna Nutrition 0.000 description 1
- 244000046464 Corylus colurna Species 0.000 description 1
- 241000186216 Corynebacterium Species 0.000 description 1
- 241000199912 Crypthecodinium cohnii Species 0.000 description 1
- 240000007092 Cucurbita argyrosperma Species 0.000 description 1
- 235000003949 Cucurbita mixta Nutrition 0.000 description 1
- 240000004244 Cucurbita moschata Species 0.000 description 1
- 235000009852 Cucurbita pepo Nutrition 0.000 description 1
- 241001607798 Cymbella Species 0.000 description 1
- 241001607806 Cymbellaceae Species 0.000 description 1
- 241000208947 Cynara Species 0.000 description 1
- 235000003198 Cynara Nutrition 0.000 description 1
- 241000252233 Cyprinus carpio Species 0.000 description 1
- 108090000626 DNA-directed RNA polymerases Proteins 0.000 description 1
- 241000208175 Daucus Species 0.000 description 1
- 235000002767 Daucus carota Nutrition 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 1
- 241000391898 Distichium capillaceum Species 0.000 description 1
- 241000584019 Distichium hagenii Species 0.000 description 1
- 241000735553 Distichium inclinatum Species 0.000 description 1
- 241000626990 Ditrichum ambiguum Species 0.000 description 1
- 241000302791 Ditrichum heteromallum Species 0.000 description 1
- 241000591211 Ditrichum pallidum Species 0.000 description 1
- 241000805644 Ditrichum rhynchostegium Species 0.000 description 1
- 241000219764 Dolichos Species 0.000 description 1
- 241001650582 Eccremidium floridanum Species 0.000 description 1
- 241001480508 Entomophthora Species 0.000 description 1
- 241001134128 Entosthodon attenuatus Species 0.000 description 1
- 241000886190 Entosthodon bonplandii Species 0.000 description 1
- 241001309994 Entosthodon drummondii Species 0.000 description 1
- URXZXNYJPAJJOQ-UHFFFAOYSA-N Erucic acid Natural products CCCCCCC=CCCCCCCCCCCCC(O)=O URXZXNYJPAJJOQ-UHFFFAOYSA-N 0.000 description 1
- 241000620209 Escherichia coli DH5[alpha] Species 0.000 description 1
- 241000195619 Euglena gracilis Species 0.000 description 1
- 241000195623 Euglenida Species 0.000 description 1
- 102100034543 Fatty acid desaturase 3 Human genes 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 241000391892 Flexitrichum flexicaule Species 0.000 description 1
- 241000651647 Flintiella <caddisfly> Species 0.000 description 1
- 101710196411 Fructose-1,6-bisphosphatase Proteins 0.000 description 1
- 101710186733 Fructose-1,6-bisphosphatase, chloroplastic Proteins 0.000 description 1
- 101710109119 Fructose-1,6-bisphosphatase, cytosolic Proteins 0.000 description 1
- 101710198902 Fructose-1,6-bisphosphate aldolase/phosphatase Proteins 0.000 description 1
- 241000384422 Funaria americana Species 0.000 description 1
- 241000072597 Funaria flavicans Species 0.000 description 1
- 241000296058 Funaria polaris Species 0.000 description 1
- 241000195889 Funariaceae Species 0.000 description 1
- 108700028146 Genetic Enhancer Elements Proteins 0.000 description 1
- 108700007698 Genetic Terminator Regions Proteins 0.000 description 1
- 206010071602 Genetic polymorphism Diseases 0.000 description 1
- 108010061711 Gliadin Proteins 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- 102100031181 Glyceraldehyde-3-phosphate dehydrogenase Human genes 0.000 description 1
- PVNIQBQSYATKKL-UHFFFAOYSA-N Glycerol trihexadecanoate Natural products CCCCCCCCCCCCCCCC(=O)OCC(OC(=O)CCCCCCCCCCCCCCC)COC(=O)CCCCCCCCCCCCCCC PVNIQBQSYATKKL-UHFFFAOYSA-N 0.000 description 1
- 235000009438 Gossypium Nutrition 0.000 description 1
- 235000014751 Gossypium arboreum Nutrition 0.000 description 1
- 240000001814 Gossypium arboreum Species 0.000 description 1
- 240000000047 Gossypium barbadense Species 0.000 description 1
- 235000009429 Gossypium barbadense Nutrition 0.000 description 1
- 235000004341 Gossypium herbaceum Nutrition 0.000 description 1
- 240000002024 Gossypium herbaceum Species 0.000 description 1
- 241001479472 Gossypium thurberi Species 0.000 description 1
- 235000004342 Gossypium thurberi Nutrition 0.000 description 1
- 108010051696 Growth Hormone Proteins 0.000 description 1
- 241000744855 Holcus Species 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000927999 Homo sapiens Diacylglycerol O-acyltransferase 2-like protein 6 Proteins 0.000 description 1
- 235000007335 Hordeum jubatum Nutrition 0.000 description 1
- 240000007036 Hordeum jubatum Species 0.000 description 1
- 241000393028 Hordeum murinum Species 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- 241000207783 Ipomoea Species 0.000 description 1
- 235000002678 Ipomoea batatas Nutrition 0.000 description 1
- 235000006350 Ipomoea batatas var. batatas Nutrition 0.000 description 1
- 241000032996 Ipomoea pandurata Species 0.000 description 1
- 244000257782 Ipomoea triloba Species 0.000 description 1
- 241001501885 Isochrysis Species 0.000 description 1
- 241001501873 Isochrysis galbana Species 0.000 description 1
- 108020003285 Isocitrate lyase Proteins 0.000 description 1
- 241000221089 Jatropha Species 0.000 description 1
- 244000001369 Juglans ailantifolia Species 0.000 description 1
- 235000014046 Juglans ailantifolia Nutrition 0.000 description 1
- 235000014053 Juglans californica Nutrition 0.000 description 1
- 244000026839 Juglans californica Species 0.000 description 1
- 235000014056 Juglans cinerea Nutrition 0.000 description 1
- 240000004929 Juglans cinerea Species 0.000 description 1
- 235000014068 Juglans hindsii Nutrition 0.000 description 1
- 240000004869 Juglans hindsii Species 0.000 description 1
- 241000339115 Juglans jamaicensis Species 0.000 description 1
- 235000019099 Juglans jamaicensis Nutrition 0.000 description 1
- 235000014074 Juglans major Nutrition 0.000 description 1
- 244000264668 Juglans major Species 0.000 description 1
- 235000014071 Juglans microcarpa Nutrition 0.000 description 1
- 240000002220 Juglans microcarpa Species 0.000 description 1
- 235000013740 Juglans nigra Nutrition 0.000 description 1
- 244000184861 Juglans nigra Species 0.000 description 1
- 244000083894 Juglans sieboldiana Species 0.000 description 1
- 235000000052 Juglans sieboldiana Nutrition 0.000 description 1
- 244000276162 Juglans x intermedia Species 0.000 description 1
- 241000193988 Kalmia angustifolia Species 0.000 description 1
- 241000193989 Kalmia microphylla Species 0.000 description 1
- 241000193999 Kalmia microphylla var. occidentalis Species 0.000 description 1
- 241000194340 Kalmia polifolia Species 0.000 description 1
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- 241001444143 Lagurus <angiosperm> Species 0.000 description 1
- 241001671311 Laurus Species 0.000 description 1
- 235000017858 Laurus nobilis Nutrition 0.000 description 1
- 101710094902 Legumin Proteins 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 108090000364 Ligases Proteins 0.000 description 1
- 102000003960 Ligases Human genes 0.000 description 1
- 241001632879 Linum austriacum Species 0.000 description 1
- 241001058641 Linum catharticum Species 0.000 description 1
- 241000665629 Linum flavum Species 0.000 description 1
- 240000009043 Linum lewisii Species 0.000 description 1
- 235000001743 Linum lewisii Nutrition 0.000 description 1
- 235000008569 Linum perenne Nutrition 0.000 description 1
- 244000236527 Linum perenne Species 0.000 description 1
- 235000006648 Linum perenne ssp. lewisii Nutrition 0.000 description 1
- 235000018495 Linum perenne var lewisii Nutrition 0.000 description 1
- 244000222932 Linum perenne var. lewisii Species 0.000 description 1
- 241000290473 Linum pratense Species 0.000 description 1
- 241001632858 Linum trigynum Species 0.000 description 1
- 235000019510 Long pepper Nutrition 0.000 description 1
- 241000408521 Lucida Species 0.000 description 1
- 241000227653 Lycopersicon Species 0.000 description 1
- 235000002262 Lycopersicon Nutrition 0.000 description 1
- 244000276497 Lycopersicon esculentum Species 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- LTYOQGRJFJAKNA-KKIMTKSISA-N Malonyl CoA Natural products S(C(=O)CC(=O)O)CCNC(=O)CCNC(=O)[C@@H](O)C(CO[P@](=O)(O[P@](=O)(OC[C@H]1[C@@H](OP(=O)(O)O)[C@@H](O)[C@@H](n2c3ncnc(N)c3nc2)O1)O)O)(C)C LTYOQGRJFJAKNA-KKIMTKSISA-N 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 241001347890 Mamiella gilva Species 0.000 description 1
- 235000014826 Mangifera indica Nutrition 0.000 description 1
- 235000004456 Manihot esculenta Nutrition 0.000 description 1
- 244000149260 Manihot esculenta subsp flabellifolia Species 0.000 description 1
- 235000019091 Manihot esculenta subsp flabellifolia Nutrition 0.000 description 1
- 241000445369 Marchantia foliacea Species 0.000 description 1
- 241001148464 Marchantiaceae Species 0.000 description 1
- 241000219823 Medicago Species 0.000 description 1
- 235000010624 Medicago sativa Nutrition 0.000 description 1
- 244000046070 Medicago sativa subsp falcata Species 0.000 description 1
- 235000010621 Medicago sativa subsp falcata Nutrition 0.000 description 1
- 235000018328 Medicago sativa subsp x varia Nutrition 0.000 description 1
- 241001508791 Medicago sativa subsp. x varia Species 0.000 description 1
- 108090000301 Membrane transport proteins Proteins 0.000 description 1
- 102000003939 Membrane transport proteins Human genes 0.000 description 1
- 241001536503 Micromonas Species 0.000 description 1
- 241001536507 Micromonas pusilla Species 0.000 description 1
- 241000907999 Mortierella alpina Species 0.000 description 1
- 229920000715 Mucilage Polymers 0.000 description 1
- 241000235395 Mucor Species 0.000 description 1
- 240000008790 Musa x paradisiaca Species 0.000 description 1
- 241000234615 Musaceae Species 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 241000013516 Nephroselmis pyriformis Species 0.000 description 1
- 241000710248 Nephroselmis rotunda Species 0.000 description 1
- 244000061322 Nicotiana alata Species 0.000 description 1
- 241000228653 Nicotiana attenuata Species 0.000 description 1
- 241000208128 Nicotiana glauca Species 0.000 description 1
- 241001144493 Nicotiana obtusifolia Species 0.000 description 1
- 241000493375 Nicotiana quadrivalvis Species 0.000 description 1
- 241001290303 Nicotiana repanda Species 0.000 description 1
- 241000208134 Nicotiana rustica Species 0.000 description 1
- 241000208136 Nicotiana sylvestris Species 0.000 description 1
- 244000061176 Nicotiana tabacum Species 0.000 description 1
- 101001068640 Nicotiana tabacum Basic form of pathogenesis-related protein 1 Proteins 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 240000008916 Oenothera biennis Species 0.000 description 1
- 235000002725 Olea europaea Nutrition 0.000 description 1
- 108091034117 Oligonucleotide Proteins 0.000 description 1
- 241000219929 Onagraceae Species 0.000 description 1
- 241000277334 Oncorhynchus Species 0.000 description 1
- 241000209094 Oryza Species 0.000 description 1
- 240000001516 Oryza latifolia Species 0.000 description 1
- 238000012408 PCR amplification Methods 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 101150101414 PRP1 gene Proteins 0.000 description 1
- 235000021314 Palmitic acid Nutrition 0.000 description 1
- 235000011096 Papaver Nutrition 0.000 description 1
- 235000014794 Papaver dubium Nutrition 0.000 description 1
- 241001106558 Papaver dubium Species 0.000 description 1
- 235000007846 Papaver rhoeas Nutrition 0.000 description 1
- 240000004674 Papaver rhoeas Species 0.000 description 1
- 241000218180 Papaveraceae Species 0.000 description 1
- 241000207960 Pedaliaceae Species 0.000 description 1
- 241000208181 Pelargonium Species 0.000 description 1
- 241000611783 Pelargonium grossularioides Species 0.000 description 1
- 241000721490 Peperomia Species 0.000 description 1
- 241000886625 Peperomia elongata Species 0.000 description 1
- 241000269799 Perca fluviatilis Species 0.000 description 1
- 241000218196 Persea Species 0.000 description 1
- 240000002426 Persea americana var. drymifolia Species 0.000 description 1
- CNVZJPUDSLNTQU-UHFFFAOYSA-N Petroselaidic acid Natural products CCCCCCCCCCCC=CCCCCC(O)=O CNVZJPUDSLNTQU-UHFFFAOYSA-N 0.000 description 1
- 241000219833 Phaseolus Species 0.000 description 1
- 241000653411 Physcomitrium collenchymatum Species 0.000 description 1
- 241000653412 Physcomitrium eurystomum Species 0.000 description 1
- 241000976036 Physcomitrium hookeri Species 0.000 description 1
- 241001484414 Physcomitrium immersum Species 0.000 description 1
- 241001504255 Physcomitrium pyriforme Species 0.000 description 1
- 241000976020 Physcomitrium subsphaericum Species 0.000 description 1
- 241000233622 Phytophthora infestans Species 0.000 description 1
- 241000722363 Piper Species 0.000 description 1
- 241000886627 Piper amalago Species 0.000 description 1
- 235000016763 Piper auritum Nutrition 0.000 description 1
- 240000005534 Piper auritum Species 0.000 description 1
- 240000008154 Piper betle Species 0.000 description 1
- 235000002711 Piper cubeba Nutrition 0.000 description 1
- 240000003731 Piper cubeba Species 0.000 description 1
- 240000003455 Piper longum Species 0.000 description 1
- 244000203593 Piper nigrum Species 0.000 description 1
- 241000758706 Piperaceae Species 0.000 description 1
- 241000543704 Pistacia Species 0.000 description 1
- 235000003445 Pistacia Nutrition 0.000 description 1
- 241000219843 Pisum Species 0.000 description 1
- 244000058602 Pisum arvense Species 0.000 description 1
- 235000016815 Pisum sativum var arvense Nutrition 0.000 description 1
- 235000009504 Pisum sativum var pumilio Nutrition 0.000 description 1
- 244000290339 Pisum sativum var. pumilio Species 0.000 description 1
- 241000591220 Pleuridium acuminatum Species 0.000 description 1
- 241000269980 Pleuronectidae Species 0.000 description 1
- 241001655088 Polyblepharides amylifera Species 0.000 description 1
- 241000206618 Porphyridium Species 0.000 description 1
- 241001494715 Porphyridium purpureum Species 0.000 description 1
- 241000013527 Prasinococcus capsulatus Species 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- 241000208465 Proteaceae Species 0.000 description 1
- 235000014360 Punica granatum Nutrition 0.000 description 1
- 241000134217 Pycnococcus Species 0.000 description 1
- 241000134041 Pycnococcus provasolii Species 0.000 description 1
- 241000949276 Pyramimonas disomata Species 0.000 description 1
- 241000724480 Pyramimonas obovata Species 0.000 description 1
- 241001509179 Pyramimonas parkeae Species 0.000 description 1
- 241001509164 Pyramimonas plurioculata Species 0.000 description 1
- 241001509149 Pyramimonas sp. Species 0.000 description 1
- LRDHPEKBQXDTLO-BQYQJAHWSA-N Pyrulic acid Chemical compound CCCCCC\C=C\C#CCCCCCCC(O)=O LRDHPEKBQXDTLO-BQYQJAHWSA-N 0.000 description 1
- 241000233612 Pythiaceae Species 0.000 description 1
- 238000012180 RNAeasy kit Methods 0.000 description 1
- 241001632427 Radiola Species 0.000 description 1
- HXQHFNIKBKZGRP-UHFFFAOYSA-N Ranuncelin-saeure-methylester Natural products CCCCCC=CCC=CCCC=CCCCC(O)=O HXQHFNIKBKZGRP-UHFFFAOYSA-N 0.000 description 1
- 101100368710 Rattus norvegicus Tacstd2 gene Proteins 0.000 description 1
- 108700005075 Regulator Genes Proteins 0.000 description 1
- 241000597892 Reimeria Species 0.000 description 1
- 208000025747 Rheumatic disease Diseases 0.000 description 1
- 241001460404 Rhodosorus Species 0.000 description 1
- 108010003581 Ribulose-bisphosphate carboxylase Proteins 0.000 description 1
- 235000003846 Ricinus Nutrition 0.000 description 1
- 241000322381 Ricinus <louse> Species 0.000 description 1
- 240000000528 Ricinus communis Species 0.000 description 1
- 235000005066 Rosa arkansana Nutrition 0.000 description 1
- 241000109365 Rosa arkansana Species 0.000 description 1
- 241001107098 Rubiaceae Species 0.000 description 1
- 229910003798 SPO2 Inorganic materials 0.000 description 1
- 241000235070 Saccharomyces Species 0.000 description 1
- 101100434411 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) ADH1 gene Proteins 0.000 description 1
- 101100342406 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) PRS1 gene Proteins 0.000 description 1
- 241000209051 Saccharum Species 0.000 description 1
- 240000000111 Saccharum officinarum Species 0.000 description 1
- 241001650571 Saelania Species 0.000 description 1
- 241001650568 Saelania glaucescens Species 0.000 description 1
- 241000607142 Salmonella Species 0.000 description 1
- 241000277331 Salmonidae Species 0.000 description 1
- 241000785681 Sander vitreus Species 0.000 description 1
- 241001125046 Sardina pilchardus Species 0.000 description 1
- 241000196327 Scherffelia dubia Species 0.000 description 1
- 241000233671 Schizochytrium Species 0.000 description 1
- 101100478210 Schizosaccharomyces pombe (strain 972 / ATCC 24843) spo2 gene Proteins 0.000 description 1
- 241000269821 Scombridae Species 0.000 description 1
- 241000207844 Scrophulariaceae Species 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- 235000009367 Sesamum alatum Nutrition 0.000 description 1
- 241000863430 Shewanella Species 0.000 description 1
- 241000220261 Sinapis Species 0.000 description 1
- 108020004682 Single-Stranded DNA Proteins 0.000 description 1
- 235000002634 Solanum Nutrition 0.000 description 1
- 241000207763 Solanum Species 0.000 description 1
- 244000244100 Solanum integrifolium Species 0.000 description 1
- 235000000099 Solanum integrifolium Nutrition 0.000 description 1
- 235000002560 Solanum lycopersicum Nutrition 0.000 description 1
- 102100038803 Somatotropin Human genes 0.000 description 1
- 235000007230 Sorghum bicolor Nutrition 0.000 description 1
- 235000015503 Sorghum bicolor subsp. drummondii Nutrition 0.000 description 1
- 241000694025 Sorghum x drummondii Species 0.000 description 1
- 235000009184 Spondias indica Nutrition 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 241000187747 Streptomyces Species 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- 241000248520 Stylonychia Species 0.000 description 1
- 244000099500 Sudangras Species 0.000 description 1
- 235000021536 Sugar beet Nutrition 0.000 description 1
- 240000000785 Tagetes erecta Species 0.000 description 1
- 235000012311 Tagetes erecta Nutrition 0.000 description 1
- 235000003591 Tagetes lucida Nutrition 0.000 description 1
- 240000002670 Tagetes lucida Species 0.000 description 1
- 235000013492 Tagetes tenuifolia Nutrition 0.000 description 1
- 240000008630 Tagetes tenuifolia Species 0.000 description 1
- 108010006785 Taq Polymerase Proteins 0.000 description 1
- 235000005212 Terminalia tomentosa Nutrition 0.000 description 1
- 241000482316 Tetraselmis apiculata Species 0.000 description 1
- 241001478733 Tetraselmis convolutae Species 0.000 description 1
- 241000482317 Tetraselmis inconspicua Species 0.000 description 1
- 241000134057 Tetraselmis marina Species 0.000 description 1
- 241000196320 Tetraselmis striata Species 0.000 description 1
- 241000196316 Tetraselmis subcordiformis Species 0.000 description 1
- 241000450725 Tetraselmis tetrathele Species 0.000 description 1
- 244000269722 Thea sinensis Species 0.000 description 1
- 241000219161 Theobroma Species 0.000 description 1
- 241001298230 Thraustochytrium sp. Species 0.000 description 1
- 102000004357 Transferases Human genes 0.000 description 1
- 108090000992 Transferases Proteins 0.000 description 1
- 241000209140 Triticum Species 0.000 description 1
- 235000007264 Triticum durum Nutrition 0.000 description 1
- 235000002041 Triticum macha Nutrition 0.000 description 1
- 244000102426 Triticum macha Species 0.000 description 1
- 235000007247 Triticum turgidum Nutrition 0.000 description 1
- 240000002805 Triticum turgidum Species 0.000 description 1
- 241000209143 Triticum turgidum subsp. durum Species 0.000 description 1
- 101150057897 USP gene Proteins 0.000 description 1
- 108090000848 Ubiquitin Proteins 0.000 description 1
- 102000044159 Ubiquitin Human genes 0.000 description 1
- 235000021322 Vaccenic acid Nutrition 0.000 description 1
- UWHZIFQPPBDJPM-FPLPWBNLSA-M Vaccenic acid Natural products CCCCCC\C=C/CCCCCCCCCC([O-])=O UWHZIFQPPBDJPM-FPLPWBNLSA-M 0.000 description 1
- 241000792914 Valeriana Species 0.000 description 1
- 235000003560 Valerianella locusta Nutrition 0.000 description 1
- 240000004668 Valerianella locusta Species 0.000 description 1
- 241000201404 Verbascum blattaria Species 0.000 description 1
- 241001290187 Verbascum chaixii Species 0.000 description 1
- 241001508736 Verbascum lychnitis Species 0.000 description 1
- 241000293860 Verbascum nigrum Species 0.000 description 1
- 241000293859 Verbascum phlomoides Species 0.000 description 1
- 241000293865 Verbascum pulverulentum Species 0.000 description 1
- 241000607284 Vibrio sp. Species 0.000 description 1
- 235000010749 Vicia faba Nutrition 0.000 description 1
- 240000006677 Vicia faba Species 0.000 description 1
- 101100209349 Vicia faba USP gene Proteins 0.000 description 1
- 108700026292 Vicia faba usp Proteins 0.000 description 1
- 101710196023 Vicilin Proteins 0.000 description 1
- 241000209149 Zea Species 0.000 description 1
- 235000007244 Zea mays Nutrition 0.000 description 1
- 108010055615 Zein Proteins 0.000 description 1
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 1
- 230000036579 abiotic stress Effects 0.000 description 1
- 125000000218 acetic acid group Chemical group C(C)(=O)* 0.000 description 1
- 238000005903 acid hydrolysis reaction Methods 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 101150102866 adc1 gene Proteins 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 235000004279 alanine Nutrition 0.000 description 1
- 108010027591 aleurain Proteins 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 108090000637 alpha-Amylases Proteins 0.000 description 1
- 102000004139 alpha-Amylases Human genes 0.000 description 1
- CUXYLFPMQMFGPL-UHFFFAOYSA-N alpha-Elaeostearinsaeure Natural products C(CCCCCCCC=CC=CC=CCCCC)(=O)O CUXYLFPMQMFGPL-UHFFFAOYSA-N 0.000 description 1
- 229940024171 alpha-amylase Drugs 0.000 description 1
- IJTNSXPMYKJZPR-WVRBZULHSA-N alpha-parinaric acid Natural products CCC=C/C=C/C=C/C=CCCCCCCCC(=O)O IJTNSXPMYKJZPR-WVRBZULHSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 238000012435 analytical chromatography Methods 0.000 description 1
- 235000019728 animal nutrition Nutrition 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000012062 aqueous buffer Substances 0.000 description 1
- 101150010487 are gene Proteins 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 235000016520 artichoke thistle Nutrition 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 235000008452 baby food Nutrition 0.000 description 1
- OGBUMNBNEWYMNJ-UHFFFAOYSA-N batilol Chemical class CCCCCCCCCCCCCCCCCCOCC(O)CO OGBUMNBNEWYMNJ-UHFFFAOYSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 229910002056 binary alloy Inorganic materials 0.000 description 1
- 108091008324 binding proteins Proteins 0.000 description 1
- 238000005842 biochemical reaction Methods 0.000 description 1
- 239000012620 biological material Substances 0.000 description 1
- 230000004790 biotic stress Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 230000003925 brain function Effects 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000001728 capsicum frutescens Substances 0.000 description 1
- 239000001390 capsicum minimum Substances 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical group 0.000 description 1
- 235000020226 cashew nut Nutrition 0.000 description 1
- 229960002727 cefotaxime sodium Drugs 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 210000003763 chloroplast Anatomy 0.000 description 1
- 235000012000 cholesterol Nutrition 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 230000002759 chromosomal effect Effects 0.000 description 1
- 230000006020 chronic inflammation Effects 0.000 description 1
- 101150074451 clpP gene Proteins 0.000 description 1
- 101150043719 clpP1 gene Proteins 0.000 description 1
- 101150102296 clpP2 gene Proteins 0.000 description 1
- 238000000975 co-precipitation Methods 0.000 description 1
- 235000018597 common camellia Nutrition 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 230000021615 conjugation Effects 0.000 description 1
- 108091036078 conserved sequence Proteins 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- PQANGXXSEABURG-UHFFFAOYSA-N cyclohexenol Natural products OC1CCCC=C1 PQANGXXSEABURG-UHFFFAOYSA-N 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 108010011713 delta-15 desaturase Proteins 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 150000001982 diacylglycerols Chemical class 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 229910001882 dioxygen Inorganic materials 0.000 description 1
- FRKBLBQTSTUKOV-UHFFFAOYSA-N diphosphatidyl glycerol Natural products OP(O)(=O)OCC(OP(O)(O)=O)COP(O)(O)=O FRKBLBQTSTUKOV-UHFFFAOYSA-N 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- ZGSPNIOCEDOHGS-UHFFFAOYSA-L disodium [3-[2,3-di(octadeca-9,12-dienoyloxy)propoxy-oxidophosphoryl]oxy-2-hydroxypropyl] 2,3-di(octadeca-9,12-dienoyloxy)propyl phosphate Chemical compound [Na+].[Na+].CCCCCC=CCC=CCCCCCCCC(=O)OCC(OC(=O)CCCCCCCC=CCC=CCCCCC)COP([O-])(=O)OCC(O)COP([O-])(=O)OCC(OC(=O)CCCCCCCC=CCC=CCCCCC)COC(=O)CCCCCCCC=CCC=CCCCCC ZGSPNIOCEDOHGS-UHFFFAOYSA-L 0.000 description 1
- NEKNNCABDXGBEN-UHFFFAOYSA-L disodium;4-(4-chloro-2-methylphenoxy)butanoate;4-(2,4-dichlorophenoxy)butanoate Chemical compound [Na+].[Na+].CC1=CC(Cl)=CC=C1OCCCC([O-])=O.[O-]C(=O)CCCOC1=CC=C(Cl)C=C1Cl NEKNNCABDXGBEN-UHFFFAOYSA-L 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 238000011143 downstream manufacturing Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 230000002526 effect on cardiovascular system Effects 0.000 description 1
- 210000002472 endoplasmic reticulum Anatomy 0.000 description 1
- 238000006911 enzymatic reaction Methods 0.000 description 1
- 210000001339 epidermal cell Anatomy 0.000 description 1
- 210000002615 epidermis Anatomy 0.000 description 1
- DPUOLQHDNGRHBS-KTKRTIGZSA-N erucic acid Chemical compound CCCCCCCC\C=C/CCCCCCCCCCCC(O)=O DPUOLQHDNGRHBS-KTKRTIGZSA-N 0.000 description 1
- 235000004626 essential fatty acids Nutrition 0.000 description 1
- 241001233957 eudicotyledons Species 0.000 description 1
- 210000001723 extracellular space Anatomy 0.000 description 1
- 239000010685 fatty oil Substances 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 230000004907 flux Effects 0.000 description 1
- 239000004459 forage Substances 0.000 description 1
- 230000005714 functional activity Effects 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 238000010413 gardening Methods 0.000 description 1
- 238000002290 gas chromatography-mass spectrometry Methods 0.000 description 1
- 238000001030 gas--liquid chromatography Methods 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 238000001502 gel electrophoresis Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 108010050792 glutenin Proteins 0.000 description 1
- 108020004445 glyceraldehyde-3-phosphate dehydrogenase Proteins 0.000 description 1
- 150000002314 glycerols Chemical class 0.000 description 1
- 108010083391 glycinin Proteins 0.000 description 1
- 150000002339 glycosphingolipids Chemical class 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 239000000122 growth hormone Substances 0.000 description 1
- ZJYYHGLJYGJLLN-UHFFFAOYSA-N guanidinium thiocyanate Chemical compound SC#N.NC(N)=N ZJYYHGLJYGJLLN-UHFFFAOYSA-N 0.000 description 1
- 238000003306 harvesting Methods 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 235000019514 herring Nutrition 0.000 description 1
- 235000006486 human diet Nutrition 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 239000003501 hydroponics Substances 0.000 description 1
- 208000026278 immune system disease Diseases 0.000 description 1
- 230000008676 import Effects 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 238000011005 laboratory method Methods 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 150000002617 leukotrienes Chemical class 0.000 description 1
- 238000001638 lipofection Methods 0.000 description 1
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 1
- 235000020667 long-chain omega-3 fatty acid Nutrition 0.000 description 1
- 235000020640 mackerel Nutrition 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- LTYOQGRJFJAKNA-DVVLENMVSA-N malonyl-CoA Chemical compound O[C@@H]1[C@H](OP(O)(O)=O)[C@@H](COP(O)(=O)OP(O)(=O)OCC(C)(C)[C@@H](O)C(=O)NCCC(=O)NCCSC(=O)CC(O)=O)O[C@H]1N1C2=NC=NC(N)=C2N=C1 LTYOQGRJFJAKNA-DVVLENMVSA-N 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000009061 membrane transport Effects 0.000 description 1
- 230000037353 metabolic pathway Effects 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 238000006140 methanolysis reaction Methods 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 150000004702 methyl esters Chemical class 0.000 description 1
- 230000002906 microbiologic effect Effects 0.000 description 1
- 238000013048 microbiological method Methods 0.000 description 1
- 235000019713 millet Nutrition 0.000 description 1
- 210000003470 mitochondria Anatomy 0.000 description 1
- 108091005573 modified proteins Proteins 0.000 description 1
- 102000035118 modified proteins Human genes 0.000 description 1
- 238000001823 molecular biology technique Methods 0.000 description 1
- 235000021281 monounsaturated fatty acids Nutrition 0.000 description 1
- 239000004570 mortar (masonry) Substances 0.000 description 1
- 235000021290 n-3 DPA Nutrition 0.000 description 1
- WQEPLUUGTLDZJY-UHFFFAOYSA-N n-Pentadecanoic acid Natural products CCCCCCCCCCCCCCC(O)=O WQEPLUUGTLDZJY-UHFFFAOYSA-N 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 108091027963 non-coding RNA Proteins 0.000 description 1
- 102000042567 non-coding RNA Human genes 0.000 description 1
- IHVJOQPMHFEHNY-UHFFFAOYSA-N nonadec-6-ynoic acid Chemical compound CCCCCCCCCCCCC#CCCCCC(O)=O IHVJOQPMHFEHNY-UHFFFAOYSA-N 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 108090000021 oryzin Proteins 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 229940094443 oxytocics prostaglandins Drugs 0.000 description 1
- 230000020477 pH reduction Effects 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 210000002824 peroxisome Anatomy 0.000 description 1
- CNVZJPUDSLNTQU-SEYXRHQNSA-N petroselinic acid Chemical compound CCCCCCCCCCC\C=C/CCCCC(O)=O CNVZJPUDSLNTQU-SEYXRHQNSA-N 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 238000005191 phase separation Methods 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 230000035479 physiological effects, processes and functions Effects 0.000 description 1
- 229930001119 polyketide Natural products 0.000 description 1
- 150000003881 polyketide derivatives Chemical class 0.000 description 1
- KWYUFKZDYYNOTN-UHFFFAOYSA-M potassium hydroxide Substances [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 1
- 238000004382 potting Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 238000011027 product recovery Methods 0.000 description 1
- 230000000770 proinflammatory effect Effects 0.000 description 1
- 108060006613 prolamin Proteins 0.000 description 1
- 230000000644 propagated effect Effects 0.000 description 1
- 150000003180 prostaglandins Chemical class 0.000 description 1
- LRDHPEKBQXDTLO-UHFFFAOYSA-N pyrulic acid Natural products CCCCCCC=CC#CCCCCCCC(O)=O LRDHPEKBQXDTLO-UHFFFAOYSA-N 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000006722 reduction reaction Methods 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 230000008844 regulatory mechanism Effects 0.000 description 1
- 230000003252 repetitive effect Effects 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 108091008146 restriction endonucleases Proteins 0.000 description 1
- 238000010839 reverse transcription Methods 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 235000019515 salmon Nutrition 0.000 description 1
- 238000007127 saponification reaction Methods 0.000 description 1
- 235000019512 sardine Nutrition 0.000 description 1
- 238000007086 side reaction Methods 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 238000002741 site-directed mutagenesis Methods 0.000 description 1
- AWUCVROLDVIAJX-GSVOUGTGSA-N sn-glycerol 3-phosphate Chemical compound OC[C@@H](O)COP(O)(O)=O AWUCVROLDVIAJX-GSVOUGTGSA-N 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000000527 sonication Methods 0.000 description 1
- 238000004611 spectroscopical analysis Methods 0.000 description 1
- 238000007447 staining method Methods 0.000 description 1
- 238000012409 standard PCR amplification Methods 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 235000013706 tagetes lucida Nutrition 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- UWHZIFQPPBDJPM-BQYQJAHWSA-N trans-vaccenic acid Chemical compound CCCCCC\C=C\CCCCCCCCCC(O)=O UWHZIFQPPBDJPM-BQYQJAHWSA-N 0.000 description 1
- 238000010361 transduction Methods 0.000 description 1
- 230000026683 transduction Effects 0.000 description 1
- 238000005809 transesterification reaction Methods 0.000 description 1
- 238000000844 transformation Methods 0.000 description 1
- 238000011426 transformation method Methods 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 238000012250 transgenic expression Methods 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- CUXYLFPMQMFGPL-UYWAGRGNSA-N trichosanic acid Natural products CCCCC=C/C=C/C=CCCCCCCCC(=O)O CUXYLFPMQMFGPL-UYWAGRGNSA-N 0.000 description 1
- 210000003934 vacuole Anatomy 0.000 description 1
- 235000017468 valeriana Nutrition 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 150000004669 very long chain fatty acids Chemical class 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 238000010626 work up procedure Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P7/00—Preparation of oxygen-containing organic compounds
- C12P7/64—Fats; Fatty oils; Ester-type waxes; Higher fatty acids, i.e. having at least seven carbon atoms in an unbroken chain bound to a carboxyl group; Oxidised oils or fats
- C12P7/6436—Fatty acid esters
- C12P7/6445—Glycerides
- C12P7/6472—Glycerides containing polyunsaturated fatty acid [PUFA] residues, i.e. having two or more double bonds in their backbone
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/82—Vectors or expression systems specially adapted for eukaryotic hosts for plant cells, e.g. plant artificial chromosomes (PACs)
- C12N15/8241—Phenotypically and genetically modified plants via recombinant DNA technology
- C12N15/8242—Phenotypically and genetically modified plants via recombinant DNA technology with non-agronomic quality (output) traits, e.g. for industrial processing; Value added, non-agronomic traits
- C12N15/8243—Phenotypically and genetically modified plants via recombinant DNA technology with non-agronomic quality (output) traits, e.g. for industrial processing; Value added, non-agronomic traits involving biosynthetic or metabolic pathways, i.e. metabolic engineering, e.g. nicotine, caffeine
- C12N15/8247—Phenotypically and genetically modified plants via recombinant DNA technology with non-agronomic quality (output) traits, e.g. for industrial processing; Value added, non-agronomic traits involving biosynthetic or metabolic pathways, i.e. metabolic engineering, e.g. nicotine, caffeine involving modified lipid metabolism, e.g. seed oil composition
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/0004—Oxidoreductases (1.)
- C12N9/0071—Oxidoreductases (1.) acting on paired donors with incorporation of molecular oxygen (1.14)
- C12N9/0083—Miscellaneous (1.14.99)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/10—Transferases (2.)
- C12N9/1025—Acyltransferases (2.3)
- C12N9/1029—Acyltransferases (2.3) transferring groups other than amino-acyl groups (2.3.1)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P7/00—Preparation of oxygen-containing organic compounds
- C12P7/64—Fats; Fatty oils; Ester-type waxes; Higher fatty acids, i.e. having at least seven carbon atoms in an unbroken chain bound to a carboxyl group; Oxidised oils or fats
- C12P7/6409—Fatty acids
- C12P7/6427—Polyunsaturated fatty acids [PUFA], i.e. having two or more double bonds in their backbone
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P7/00—Preparation of oxygen-containing organic compounds
- C12P7/64—Fats; Fatty oils; Ester-type waxes; Higher fatty acids, i.e. having at least seven carbon atoms in an unbroken chain bound to a carboxyl group; Oxidised oils or fats
- C12P7/6409—Fatty acids
- C12P7/6427—Polyunsaturated fatty acids [PUFA], i.e. having two or more double bonds in their backbone
- C12P7/6432—Eicosapentaenoic acids [EPA]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P7/00—Preparation of oxygen-containing organic compounds
- C12P7/64—Fats; Fatty oils; Ester-type waxes; Higher fatty acids, i.e. having at least seven carbon atoms in an unbroken chain bound to a carboxyl group; Oxidised oils or fats
- C12P7/6409—Fatty acids
- C12P7/6427—Polyunsaturated fatty acids [PUFA], i.e. having two or more double bonds in their backbone
- C12P7/6434—Docosahexenoic acids [DHA]
Definitions
- the present invention relates to a process for the production of polyunsaturated fatty acids in transgenic plants, in which nucleic acids are introduced into the plant, which for polypeptides having ⁇ 6-desaturase, ⁇ 6-elongase, a ⁇ 5-desaturase - encode ⁇ -5-elongase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3-desaturase activity.
- These desaturases and elongases are advantageously derived from Phytophtora sojae.
- the invention further relates to the nucleic acid sequences, nucleic acid constructs, vectors and organisms containing the nucleic acid sequences according to the invention, vectors containing the nucleic acid sequences and / or the nucleic acid constructs and transgenic plants contain the aforementioned nucleic acid sequences, nucleic acid constructs and / or vectors.
- Another part of the invention relates to fatty acid compositions prepared by the process according to the invention and their use.
- Fatty acids and triacylglycerides have a variety of uses in the food, animal nutrition, cosmetics and pharmaceutical industries. Depending on whether they are free saturated and unsaturated fatty acids or triacylglycerides with an increased content of saturated or unsaturated fatty acids, they are suitable for a wide variety of applications.
- Polyunsaturated fatty acids such as linoleic and linolenic acids are essential for mammals because they can not be produced by them. Therefore, polyunsaturated ⁇ -3 fatty acids and ⁇ -6 fatty acids are an important component of animal and human food.
- polyunsaturated long-chain fatty acids Due to the customary composition of human food today, addition of polyunsaturated ⁇ -3 fatty acids, which are preferred in fish oils, is particularly important for food.
- the unsaturated fatty acid DHA is thereby attributed a positive effect on the development and maintenance of brain functions.
- polyunsaturated fatty acids are referred to as PUFA, PUFAs, LCPUFA or LCPUFAs (poly unsaturated fatty acids, PUFA, polyunsaturated fatty acids, long chain polyunsaturated fatty acids, LCPUFA).
- the free fatty acids are advantageously prepared by saponification.
- Common natural sources of these fatty acids are fish such as herring, salmon, sardine, perch, eel, carp, trout, halibut, mackerel, zander or tuna or algae.
- oils with saturated or unsaturated fatty acids are preferred.
- lipids with unsaturated fatty acids especially polyunsaturated fatty acids
- the polyunsaturated ⁇ -3 fatty acids thereby a positive effect on the cholesterol level in the blood and thus the possibility of preventing heart disease is attributed.
- ⁇ -3 fatty acids By adding these ⁇ -3 fatty acids to the diet, the risk of heart disease, stroke or hypertension can be significantly reduced.
- inflammatory especially chronic inflammatory processes in the context of immunological diseases such as rheumatoid arthritis can be positively influenced by ⁇ -3 fatty acids. They are therefore added to foods especially dietary foods or found in medicines application.
- ⁇ -6 fatty acids such as arachidonic acid tend to have a negative effect on these diseases in these rheumatic diseases due to our usual food composition.
- ⁇ -3- and ⁇ -6 fatty acids are precursors of tissue hormones, the so-called eicosanoids such as the prostaglandins derived from dihomo- ⁇ -linolenic acid, arachidonic acid and eicosapentaenoic acid, the thromoxanes and leukotrienes derived from arachidonic acid and Derive the eicosapentaenoic acid.
- Eicosanoids which are formed from ⁇ -6 fatty acids usually promote inflammatory reactions
- eicosanoids (so-called PG 3 series) of ⁇ -3 fatty acids have little or no pro-inflammatory effect. Due to their positive properties, there has been no lack of approaches in the past, genes involved in the synthesis of fatty acids or triglycerides, for to make available the production of oils in various organisms with modified unsaturated fatty acid content. Thus, WO 91/13972 and its US equivalent describe a ⁇ -9-desaturase. In WO 93/11245 a ⁇ -15-desaturase is claimed in WO 94/11516 a ⁇ -12-desaturase.
- microorganisms for the production of PUFAs are microorganisms such as microalgae such as Phaeodactylum tricornutum, Porphiridium species, Thraustochytrien species, Schizochytria species or Crypthecodinium species, ciliates such as Stylonychia or Colpidium, fungi such as Mortierella, Entomophthora or Mucor and / or mosses such as Physcomitrella, Ceratodon and Marchantia (R.
- microalgae such as Phaeodactylum tricornutum, Porphiridium species, Thraustochytrien species, Schizochytria species or Crypthecodinium species
- ciliates such as Stylonychia or Colpidium
- fungi such as Mortierella, Entomophthora or Mucor and / or mosses
- Physcomitrella, Ceratodon and Marchantia R.
- Chain length - instead of the ⁇ 4-desaturation, a further elongation step at 24 C, a further ⁇ 6-desaturation and finally beta-oxidation to the C 22 is carried out here.
- the so-called spokesman synthesis path (see FIG. 1) is not suitable since the regulatory mechanisms are not known.
- the unsaturated fatty acids having double bonds for example in delta-9 , delta-6 or delta-5-position by at least two carbon atoms extend.
- Their function according to two different Elongasetypen be distinguished.
- the type I elongases which are widespread in the animal kingdom, have only a low substrate specificity, that is, they prolong a number of different unsaturated fatty acids.
- Type I I elongases are characterized by a much higher substrate specificity. They convert only a few fatty acids into double bonds in specific positions.
- WO 2005/012316 describes some of the abovementioned desaturases and elongases. Specifically WO 2005/012316 discloses first type II elongases which specifically elongate fatty acids having a double bond in the delta-5 position.
- the polyunsaturated fatty acids can be classified according to their desaturation pattern into two large classes, ⁇ -6 or ⁇ -3 fatty acids, which have metabolically and functionally different activities (Figure 1).
- the starting material for the ⁇ -6 pathway is the fatty acid linoleic acid (18: 2 ⁇ 912 ), while the ⁇ -3 pathway is via linolenic acid (18: 3 ⁇ 9 12 '15 ).
- Linolenic acid is formed by the activity of an ⁇ -3-desaturase (Tocher et al., 1998, Prague Lipid Res., 37, 73-117, Domergue et al., 2002, Eur. J. Biochem., 269, 4105-4113).
- ⁇ -3 fatty acids shows the therapeutic effect as described above in the treatment of cardiovascular diseases (Shimikawa 2001, World Rev. Nutr., Diet, 88, 100-108), inflammations (Calder 2002, Proc. Soc., 61, 345-358) and Arthridis (Cleland and James 2000, J. Rheumatoi 27, 2305-2307).
- Higher plants contain polyunsaturated fatty acids such as linoleic acid (C18: 2) and linolenic acid (C18: 3).
- ARA, EPA and DHA are absent or only found in the seed oil of higher plants (E. Ucciani: Wunsch Dictionnaire des Huiles Vegeches, Technique & Documentation - Lavoisier, 1995. ISBN: 2-7430-0009-0).
- LCPUFAs in higher plants, preferably in oilseeds such as oilseed rape, linseed, sunflower and soybeans, as this will enable large quantities of high quality LCPUFAs to be obtained inexpensively for the food, animal and pharmaceutical industries.
- WO 2005/012316 discloses such an approach.
- a disadvantage of the method described in WO 2005/012316 is that the yield of the desired fatty acids is still too low for industrial use.
- desired fatty acids such as ARA, EPA or DHA
- undesired side reactions also produce fatty acids which further reduce the yield and contaminate the resulting product.
- the inventive method for the preparation of polyunsaturated C 2 o- or C 22 -fatty acids having at least four double bonds in transgenic plants with a content of at least 15 wt .-% based on the total triglyceride content of the transgenic plant characterized in that it comprises the following method steps: a) introduction of a nucleic acid construct into the transgenic plant which contains nucleic acid sequences coding for a ⁇ 6-desaturase, a ⁇ 6-elongase and a ⁇ 5-desaturase, or b) Introducing a nucleic acid construct into the transgenic plant containing nucleic acid sequences encoding a ⁇ 6-desaturase, a ⁇ 6-elongase, a ⁇ 5-desaturase, a ⁇ 12-desaturase and ⁇ 3-desaturase
- nucleic acid construct containing nucleic acid sequences encoding a ⁇ 6-desaturase, a ⁇ 6- ⁇ 5-desaturase, a ⁇ 5-elongase and ⁇ 4-desaturase; -Longase, a ⁇ -5-desaturase, a ⁇ -5 elongase, ⁇ -4-desaturase, a ⁇ -12-desaturase and ⁇ -3-desaturase encoded, and e) recovery of the oils or lipids from the plants ,
- the polyunsaturated C 2 o- or C 22 -fatty acids prepared in the process according to the invention contain at least four, five or six double bonds.
- the fatty acids contain five or six double bonds.
- saturated fatty acids are little or not reacted with the nucleic acids used in the process. Little is to be understood that compared to polyunsaturated fatty acids, the saturated fatty acids with less than 5% of the activity, advantageously less than 3%, particularly advantageous with less than 2%, most preferably less than 1; 0.5; 0.25 or 0.125% are implemented. These produced fatty acids can be produced as the only product in the process or present in a fatty acid mixture.
- nucleic acid sequences used in the method according to the invention are isolated nucleic acid sequences which are suitable for ⁇ -6 polypeptides.
- ARA arachidonic acid
- EPA eicosape ⁇ tenic acid
- DHA docosahexaenoic acid
- the polyunsaturated C 20 - or C 3-8 fatty acids containing at least four double bonds are arachidonic acid.
- eicosapentaenoic acid or docosahexaenoic acid or mixtures thereof are prepared as polyunsaturated C 20 - or C 22 -fatty acids having at least four double bonds.
- Mixtures of ARA, EPA and DHA can also be prepared by the process according to the invention.
- Arachidonic acid or eicosapentaenoic acid having a content of at least 15, 16, 17, 18, 19 or 20% by weight, preferably 25, 30, 35 or 40% by weight, particularly preferably 45.50, are advantageous in the process according to the invention , 55 or 60 wt .-% based on the total triglyceride content produced in the transgenic plant.
- docosahexaenoic acid is produced with a content of at least 4, 5 or 6% by weight, advantageously with a content of 7, 8, 9 or 10% by weight, based on the total triglyceride content in the transgenic plant.
- triglycerides less than 0.5; 0.4; 0.3; 0.2 or 0.1% by weight, advantageously less than 0.09; 0.08; 0.07; 0.06 or 0.05% by weight, more preferably less than 0.04; 0.03; 0.02 or 0.01 wt .-% based on the total fatty acid content of the triglycerides of a polyunsaturated fatty acid selected from the group of c22: 4 ⁇ 7 ' 10 ' 13 '16 -, C22 . 5 ⁇ 4,7,1O, 13, 1 6 _ qThe c2 2: 5 ⁇ 7 '10' 13 '16' 19 fatty acid be present.
- the polyunsaturated fatty acids produced in the process are advantageously bound in triacylglycerides, but may also occur as free fatty acids or bound in the form of other fatty acid esters in the organisms. They may be present as "pure products" or advantageously in the form of mixtures of different fatty acids or mixtures of different glycerides.
- fatty acid esters having polyunsaturated C 2 o- and / or C 22 -fatty acid molecules having at least four double bonds in the fatty acid ester, advantageously having at least five or six double bonds in the fatty acid ester, are advantageously prepared.
- polyunsaturated fatty acids are particularly advantageously selected from the group ⁇ -6-docosatetraenoic acid c22: 4 ⁇ 7 ' 10 ' 13 '16 , ⁇ -6-docosapentaenoic acid c22: 5 ⁇ 4 ' 7 ' i ⁇ 13 ' 16 or ⁇ -3-docosapentaenoic acid c22: 5 ⁇ 7 ' 10 ' 13 ' 16 ' 19 was produced in the triglycerides at less than 0.5% by weight, based on the total fatty acid content of the triglycerides.
- the fatty acid esters with polyunsaturated C 2 o- and / or C 22 -fatty acid molecules can be prepared from the organisms used for the preparation of the fatty acid esters in the form of an oil or lipid, for example in the form of compounds such as sphingolipids, phosphoglycerides, lipids, glycolipids, such as Glycosphingolipids, phospholipids such as phosphatidylethanolamine, phosphatidylcholine, phosphatidylserine, phosphatidylglycerol, phosphatidylinositol or diphosphatidylglycerol, monoacylglycerides, diacylglycerides, triacylglycerides or other fatty acid esters such as the acetyl-coenzymeA esters which contain the polyunsaturated fatty acids having at least four, five or six, preferably five or six double bonds, they are advantageously isolated in the form of their diacylg
- the polyunsaturated fatty acids are also included as free fatty acids or bound in other compounds in the organisms beneficial to the plants.
- the various compounds mentioned above are present in the organisms in an approximate distribution of 80 to 90% by weight of triglycerides, 2 to 5% by weight of diglycerides, 5 to 10% by weight of monoglycerides, 1 to 5 wt .-% of free fatty acids, 2 to 8 wt .-% phospholipids ago, wherein the sum of the various compounds to 100 wt .-% complements.
- the LCPUFAs produced such as arachidonic acid and eicosapentaenoic acid, having a content of at least 15% by weight, advantageously of at least 16 or 17% by weight, preferably of at least 18, 19 or 20% by weight, are particularly preferred of at least 21, 22, 23, 24 or 25 wt .-%, most preferably of at least 26, 27, 28, 29 or 30 wt .-% based on the total fatty acids in the transgenic organisms advantageously produced in a transgenic plant.
- the fatty acids are prepared in bound form.
- these unsaturated fatty acids can be brought to the sn1, sn2 and / or sn3 position of the advantageously prepared triglycerides.
- linoleic acid C18: 2 or linolenic acid (C18: 3) are undergoing several reaction steps, the end products of the method such as arachidonic acid (ARA), eicosapentaenoic acid (EPA) or docosapentaenoic acid (DHA) are not as absolute pure products, there are always small traces of precursors in the final product. If both linoleic acid and linolenic acid are present in the starting organism or in the parent plant, for example, the end products such as ARA, EPA or DHA are generally present as mixtures.
- ARA arachidonic acid
- EPA eicosapentaenoic acid
- DHA docosapentaenoic acid
- the precursors linoleic acid and / or linolenic acid should advantageously not more than 20 wt .-%, preferably not more than 15 wt .-%, more preferably not more than 10 wt .-%, most preferably not more than 5 wt .-% based amount to the amount of each end product.
- ARA, ARA and EPA, EPA and DHA or only DHA are bound in the process according to the invention or prepared as free acids in a transgenic plant as end products.
- the compounds ARA, EPA and DHA are prepared simultaneously, they are advantageously used in a ratio of at least 1: 1: 2 (EPA: ARA: DHA), preferably of at least 1: 1: 3, preferably of 1: 1: 4 preferably prepared from 1: 1: 5. If ARA and EPA are produced in the process, they are advantageously prepared in a ratio of at least 5: 1 to at least 1: 5. If EPA and DHA are produced in the process, they are advantageously prepared in a ratio of at least 5: 1 to at least 1: 5.
- Fatty acid esters or fatty acid mixtures which have been prepared by the process according to the invention advantageously contain 6 to 15% palmitic acid, 1 to 6% stearic acid; 7 - 85% oleic acid; 0.5 to 8% of vaccenic acid, 0.1 to 1% of arachidic acid, 7 to 25% of saturated fatty acids, 8 to 85% of monounsaturated fatty acids and 60 to 85% of polyunsaturated fatty acids in each case based on 100% and on the total fatty acid content of the organisms.
- Fatty acid are preferably at least 0.1 in the fatty acid esters or fatty acid mixtures; 0.2; 0.3; 0.4; 0.5; 0.6; 0.7; 0.8; 0.9 or 1% based on the total fatty acid content of arachidonic acid.
- the fatty acid esters or fatty acid mixtures which have been prepared by the process according to the invention contain advantageous fatty acids selected from the group of the fatty acids erucic acid (13-docosaic acid), sterculic acid (9,10-methylene octadec-9-enoic acid), malvalic acid (8,9-methylheptadec-8-enoic acid), chaulmoogric acid (cyclopentenodecanoic acid), Furan fatty acid (9,12-epoxy-octadeca-9,11-dienoic acid), vernonic acid (9,10-epoxyoctadec-12-enoic acid), taric acid (6-octadecynoic acid), 6-nonadecynoic acid, santalbinic acid (t11-octadecene -9-ynoic acid), 6,9-octadecenynoic acid, pyrulic acid (t10-heptade
- Octadecatrienoic acid Octadecatrienoic acid
- elecetic acid (9c11t13t octadecatrienoic acid), jacric acid (8c10t12c octadecatrienoic acid), punicic acid (9c11t13c octadecatrienoic acid), parinaric acid (9c11t13t15c octadecatetraenoic acid), pinolenic acid (all-cis-5,9,12-octadecatrienoic acid), LaballenLitere (5,6-Octadecadienallen Textre), Ricinolklare (12-Hydroxyölklare) and / or Coriolinklare (13-Hydroxy-9c, 11 t-Octadecadienonklare).
- the abovementioned fatty acids are generally advantageously present only in small amounts in the fatty acid esters or fatty acid mixtures prepared by the process according to the invention, that is to say they come to less than 30%, preferably less than 25%, 24%, 23, based on the total fatty acids %, 22% or 21%, more preferably less than 20%, 15%, 10%, 9%, 8%, 7%, 6% or 5%, most preferably less than 4%, 3%, 2 % or 1% ago.
- An increase in the yield of polyunsaturated fatty acids of at least 50%, advantageously of at least 80%, particularly advantageously of at least 100%, very particularly advantageously of at least 150%, compared with the non-transgenic plant can be achieved by the nucleic acid sequences according to the invention or in the method according to the invention such as Arabidopsis, flax, rapeseed, soy or Camelina can be achieved in the comparison in the GC analysis.
- chemically pure polyunsaturated fatty acids or fatty acid compositions can be prepared by the methods described above.
- the fatty acids or fatty acid compositions are isolated from the plant in a known manner, for example via extraction, distillation, crystallization, chromatography or combinations of these methods.
- These chemically pure fatty acids or fatty acid compositions are advantageous for applications in the food industry, the cosmetics industry and especially the pharmaceutical industry.
- all plants are suitable as organism for the production in the process according to the invention.
- oil crops or crops are used in the process.
- plants in principle, all plants are in question, which are able to synthesize fatty acids as all dicotyledonous or monocotyledonous plants, algae or mosses.
- Advantageous plants are selected from the group of the plant families Adelotheciaceae, Anacardiaceae, Asteraceae, Apiaceae, Betulaceae, Boraginaceae, Brassicadown, Bromeliaceae, Caricaceae, Cannabaceae, Convolvulaceae, Chenopodiaceae, Crypthecodiniaceae, Cucurbitaceae, Ditrichaceae, Elaeagnaceae, Ericaceae, Euphorbiaceae , Fabaceae, Geraniaceae, Gramineae, Juglandaceae, Lauraceae, Leguminose, Linaceae, Prasinophyceae or vegetables or ornamental plants such as Tagetes.
- Examples include the following plants selected from the group: Adelotheciaceae as the genera Physcomitrella eg the genus and species Physcomitrella patens, Anacardiaceae such as the genera Pistacia, Mangifera, Anacardium eg the genus and species Pistacia vera [pistachio], Mangifer indica [Mango ] or Anacardium occidentale [Cashew], Asteraceae such as the genera Calendula, Carthage, Centaurea, Cichorium, Cynara, Helianthus, Lactuca, Locusta, Tagetes, Valeriana eg the genus and species Calendula officinalis [Gardening Marigold], Carthamus tinctorius [Dyer's Thistle , safflower], Centaurea cyanus [cornflower], Cichorium intybus [chicory], Cynara scolymus [Artichoke], Helianthus annus [sunflower],
- Elaeagnaceae such as the genus Elaeagnus eg the genus and Art Olea europaea [Olive]
- Ericaceae as the genus Kalmia eg the genera and species Kalmia latifolia, Kalmia angustifolia, Kalmia microphylla, Kalmia polifolia, Kalmia occidentalis, Cistus chamaerhodendros or Kalmia lucida [Berglorbeer]
- Euphorbiaceae such as the genera Manihot, Janipha, Jatropha Ricinus eg the genera and species Manihot utilissima, Janipha manihot ,, Jatropha manihot, Manihot aipil, Manihot dulcis, Manihot manihot, Manihot melanobasis, Manihot esculenta [Manihot] or Ricinus communis [Castor], Fabaceae
- obtusifolia Funaria muhlenbergii, Funaria orcuttii, Funaria plano convexa, Funaria polaris, Funaria ravenelii, Funaria rubriseta, Funaria serrata, Funaria sonorae, Funaria sublimbatus, Funaria tucsoni Physcomitrella californica, Physcomitrella patens, Physcomitrella readeri, Physcomitrium spp., Physcomitrium californicum, Physcomitrium collenchymatum, Physcomitrium coloradense, Physcomitrium cupuliferum, Physcomitrium drummondii, Physcomitrium eurystomum, Physcomitrium flexifolium, Physcomitrium hookeri, Physcomitrium hooke ⁇ var.
- [Cayenne pepper] such as the genera Hordeum, Seeale, Avena, Sorghum, Andropogon, Holcus, Panicum, Oryza, Zea (maize), Triticum eg the genera and species Hordeum vulgare, Hordeum jubatum, Hordeum murinum, Hordeum secalinum, Hordeum distichon Hordeum aegiceras, Hordeum hexastichonum, Hordeum hexastichum, Hordeum irregular, Hordeum sativum, Hordeum secalinum [barley], Seeale cereale [rye], Avena sativa, Avena fatua, Avena byzantina, Avena fatua var.
- Sativa Avena hybrida [Oats], Sorghum bicolor, Sorghum halepense, Sorghum saccharatum, Sorghum vulgaris, Andropogon drummondii, Holcus bicolor, Holcus sorghum, Sorghum aethiopicum, Sorghum arundinaceum, Sorghum caffrorum, Sorghum cernuum, Sorghum bathna, Sorghum drummondii, Sorghum durra, Sorghum guineense, Sorghum lanceolatum, sorghum nervosa, sorghum saccharatum, sorghum subglabrescens, sorghum verticilliflorum, sorghum vulgaris, holcus halepensis, sorghum miliaceum, panicum militaceum [millet], oryza sativa, oryza latifolia
- Macadamia intergrifolia [Macadamia]
- Prasinophyceae such as the genera Nephroselmis, Prasinococcus, Scherffelia, Tetraselmis, Mantoniella, Ostreococcus e.g. the genera and species Nephroselmis olivacea, Prasinococcus capsulatus, Scherffelia dubia, Tetraselmis chui, Tetraselmis suecica, Mantoniella squamata, Ostreococcus tauri, Rubiaceae such as the genus Coffea e.g.
- Cofea spp. Coffea arabica, Coffea canephora or Coffea liberica [coffee]
- Scrophulariaceae such as the genus Verbascum e.g. the genera and species Verbascum blattaria, Verbascum chaixii, Verbascum densiflorum, Verbascum lagurus, Verbascum longifolium, Verbascum lychnitis, Verbascum nigrum, Verbascum olympicum, Verbascum phlomoides, Verbascum phenicum, Verbascum pulverulentum or Verbascum thapsus [Mullein], Solanaceae such as the genera Capsicum, Nicotiana , Solanum, Lycopersicon eg the genera and species Capsicum annuum, Capsicum annuum var.
- Capsicum frutescens [pepper], Capsicum annuum [paprika], Nicotiana tabacum, Nicotiana alata, Nicotiana attenuata, Nicotiana glauca, Nicotiana slowdorffii, Nicotiana obtusifolia, Nicotiana quadrivalvis, Nicotiana repanda, Nicotiana rustica, Nicotiana sylvestris [tobacco], Solanum tuberosum [potato], Solanum melongena [eggplant] Lycopersicon esculentum, Lycopersicon lycopersicum., Lycopersicon pyriforme, Solanum integrifolium or Solanum lycopersicum [tomato], Sterculiaceae such as the genus Theobroma eg the genus and species Theobroma cacao [cocoa] or Theaceae such as the genus Camellia e.g.
- transgenic plants such as dicotyledonous or monocotyledonous plants are used. It is particularly advantageous in the process of the invention to use oil crop plants which contain large amounts of lipid compounds, such as peanut, rapeseed, canola, sunflower, safflower (Carthamus tinetoria), poppy, mustard, hemp, castor, olive, sesame, calendula, punica, evening primrose, mullein , Thistle, wild roses, hazelnut, almond, macadamia, avocado, bay leaf, pumpkin, flax, soya, pistachio, borage, trees (oil palm, coconut or walnut), or there are useful crops such as maize, wheat, rye, oats, Triticale, rice, barley, cotton, cassava, pepper, tagetes, solanaceae plants, such as Potato, tobacco, eggplant and tomato, Vicia species, pea, alfaifa or bush plants (coffee, cocoa, tea),
- lipid compounds such as
- Preferred plants according to the invention are oil crop plants, such as peanut, rapeseed, canola, sunflower, safflower, poppy, mustard, hemp, castor, olive, calendula, punica, evening primrose, pumpkin, flax, soy, borage, trees (oil palm, coconut).
- Particularly preferred are C18: 2 and / or C18: 3 fatty acid rich plants such as sunflower, safflower, tobacco, mullein, sesame, cotton, pumpkin, poppy, evening primrose, walnut, flax, hemp, thistle or safflower.
- plants such as safflower, sunflower, poppy, evening primrose, walnut, flax or hemp.
- genes of fatty acid or lipid metabolism may advantageously be used in combination with the inventive ⁇ -6-desaturase (s), ⁇ -6 elongase (s), ⁇ -5-desaturase (s), ⁇ -5 Elongase (s), ⁇ -4-desaturase (s), ⁇ -12-desaturase (s) and / or ⁇ -3-desaturase (s) [in the context of this application, the plural shall contain the singular and vice versa] in the process of Production of polyunsaturated fatty acids are used.
- the fatty acids can also be fed from the outside.
- Preferred substrates are the linoleic acid (C18: 2 ⁇ 9 '12 ), and / or the ⁇ -linolenic acid (C18: 3 ⁇ 69 12 ).
- ⁇ 12-desaturase oleic acid C18: 1 ⁇ 9
- C18: 2 ⁇ 9 12 linoleic acid
- Nucleic acids used in the method according to the invention are advantageously derived from plants such as algae, for example algae of the family Prasinophyceae as from the genera Heteromastix, Mammella, Mantoniella, Micromonas, Nephroselmis, Ostreococcus, Prasinocladus, Prasinococcus, Pseudoscourfielda, Pycnococcus, Pyramimonas, Scherffelia or Tetraselmis such as the genera and Species Heteromastix longifillis, Mamiella gilva, Mantoniella squamata, Micromonas pusilla, Nephroselmis olivacea, Nephroselmis pyriformis, Nephroselmis rotunda, Ostreococcus tauri, Ostreococcus sp. Prasinocladus ascus, Prasinocladus lubricus, Pycnoc
- the nucleic acids used are advantageously derived from algae of the genus Ostreococcus. Further advantageous organisms are diatoms such as Thalassiosira, Phaeodactylum or Thraustochytrium or fungi such as Thraustochytrium or Phytophthora.
- nucleic acid sequences or their derivative or homologs which code for polypeptides which still possess the enzymatic activity of the proteins encoded by nucleic acid sequences.
- These sequences are used singly or in combination with those for ⁇ 6-desaturase, ⁇ 6-oligonase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ 12-desaturase and / or ⁇ 3-desaturase-encoding nucleic acid sequences are cloned in expression constructs and used for introduction and expression in the plants. By their construction, these expression constructs enable a favorable optimal synthesis of the polyunsaturated fatty acids produced in the process according to the invention.
- the method further comprises the step of obtaining a cell or whole plant containing the nucleic acid sequences used in the method, wherein the cell and / or the plant is infected with a nucleic acid sequence of the invention which is responsible for the ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ -5-desaturase, ⁇ -5-ionogase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3 Desaturase, a gene construct or a vector as described below, alone or in combination with other nucleic acid sequences that code for proteins of the fatty acid or lipid metabolism is transformed.
- a nucleic acid sequence of the invention which is responsible for the ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ -5-desaturase, ⁇ -5-ionogase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3 Desaturase, a gene construct or
- this method further comprises the step of recovering the oils, lipids or free fatty acids from the cell or plant.
- the culture may be, for example, a fermentation culture, for example in the case of cultivation of plant cells or a greenhouse or field crop of a plant.
- the cell or plant thus produced is advantageously a cell of an oil-producing organism such as an oil crop such as peanut, canola, canola, flax, hemp, peanut, soybean, safflower, hemp, sunflower or borage.
- Cultivation is, for example, culturing in the case of plant cells, tissue or organs on or in a nutrient medium or the whole plant on or in a substrate, for example in hydroponics, potting soil or on arable land.
- Natural genetic environment means the natural genomic or chromosomal locus in the organ of origin or presence in a genomic library.
- the natural genetic environment of the nucleic acid sequence is preferably at least partially conserved.
- the environment flanks the nucleic acid sequence at least on one side and has a sequence length of at least 50 bp, preferably at least 500 bp, more preferably at least 1000 bp, most preferably at least 5000 bp.
- non-natural, synthetic artificial
- transgenic plants within the meaning of the invention are understood to mean that the nucleic acids used in the method are not in their natural position in the genome of the plant, in which case the nucleic acids can be expressed homologously or heterologously.
- transgene also means, as mentioned, that the nucleic acids according to the invention are in their natural place in the genome of the plant, but that the sequence has been changed compared to the natural sequence and / or the regulatory sequences of the natural sequences have been altered.
- Transgenic is preferably understood to mean the expression of the nucleic acids according to the invention at a non-natural site in the genome, that is to say a homologous or preferably heterologous expression of the nucleic acids is present.
- plants which are able to synthesize fatty acids, especially unsaturated fatty acids or are suitable for the expression of recombinant genes are suitable in principle as host organisms for the nucleic acids, the expression cassette or the vector used in the method according to the invention.
- Examples include plants such as Arabidopsis, Asteraceae such as calendula or crops such as soybean, peanut, castor, sunflower, corn, cotton, flax, rapeseed, coconut, oil palm, dyer safflower (Carthamus tinctorius) or cocoa bean.
- the plants according to the invention include plant cells and certain tissues, organs and parts of plants in all their forms, such as anthhers, fibers, root hair, stems, embryos, callosis, kotelydons, petioles, crops, plant tissue, reproductive tissue and Cell cultures derived from the actual transgenic plant and / or used to produce the transgenic plant.
- Transgenic plants containing the polyunsaturated fatty acids synthesized in the process according to the invention can advantageously be marketed directly without the synthesized oils, lipids or fatty acids having to be isolated.
- Plants in the process according to the invention include whole plants and all plant parts, plant organs or plant parts such as leaves, stems, seeds, roots, tubers, anthers, fibers, root hairs, stems, embryos, kaIIi, kotelydons,
- the seed includes all seed parts such as the seed shells, epidermis and sperm cells, endosperm or embryonic tissue.
- the compounds prepared in the process according to the invention can also be advantageous plants from the organisms in the form of their oils, fat, lipids and / or free Be isolated fatty acids. Polyunsaturated fatty acids produced by this process can be harvested by harvesting the organisms either from the culture in which they grow or from the field. This can be done by pressing or extraction of the plant parts, preferably the plant seeds.
- the oils, fats, lipids and / or free fatty acids by so-called cold beat or cold pressing can be obtained without supplying heat by pressing.
- the plant parts, especially the seeds, to be easier to digest they are first crushed, steamed or roasted.
- the thus pretreated seeds can then be pressed or extracted with solvents such as warm hexane.
- the solvent is removed again.
- more than 96% of the compounds prepared in the process can be isolated.
- the products thus obtained are further processed, that is refined.
- the mucilages and turbid matter are removed.
- the so-called desliming can be carried out enzymatically or, for example, chemically / physically by adding acid, such as phosphoric acid.
- the free ones such as phosphoric acid.
- Fatty acids are removed by treatment with a base, for example sodium hydroxide solution.
- a base for example sodium hydroxide solution.
- the product obtained is thoroughly washed with water to remove the lye remaining in the product and dried.
- the products are subjected to bleaching with, for example, bleaching earth or activated carbon. Finally, the product is still deodorized, for example, with steam.
- An embodiment of the invention is the use of the oils, lipids, fatty acids and / or fatty acid composition prepared by the process of the invention or by mixing these oils, lipids and / or fatty acids with animal, microbial or vegetable oils, lipids or fatty acids in feed, food, cosmetics or pharmaceuticals.
- the oils, lipids, fatty acids prepared in the process according to the invention may be mixed with other oils, lipids, fatty acids or fatty acid mixtures of animal origin, such as those known to those skilled in the art, for example. Fish oils are used.
- oil is understood as meaning a fatty acid mixture which contains unsaturated, saturated, preferably esterified fatty acid (s), advantageously bound in triglycerides. It is preferred that the oil, lipid or fat contain a high proportion of polyunsaturated free or advantageously esterified fatty acid (s), in particular linoleic acid, ⁇ -linolenic acid, dihomo- ⁇ -linolenic acid, arachidonic acid, ⁇ -linolenic acid, stearidonic acid, eicosatetraenoic acid, eicosapentaenoic acid, Docosapentaenoic acid or docosahexaenoic acid, particularly advantageously arachidonic acid, eicosapentaenoic acid or docosahexaenoic acid.
- s polyunsaturated free or advantageously esterified fatty acid
- the proportion of unsaturated esterified fatty acids is about 30%, more preferred is a proportion of 50%, even more preferred is a proportion of 60%, 70%, 80% or more.
- the proportion of fatty acid after conversion of the fatty acids into the methyl esters can be determined by transesterification by gas chromatography.
- the oil, lipid or fat may contain various other saturated or unsaturated fatty acids, eg calendulic acid, Palmitin, palmitoleic, stearic, oleic acid, etc., included. In particular, depending on the starting organism, the proportion of the various fatty acids in the oil or fat may vary.
- the polyunsaturated fatty acids having advantageously at least four double bonds prepared in the process are, as described above, for example sphingolipids, phosphoglycerides, lipids, glycolipids, phospholipids, monoacylglycerols, diacylglycerides, triacylglycerides or other fatty acid esters, preferably triacylglycerides.
- the polyunsaturated fatty acids containing, for example, an alkali treatment such as aqueous KOH or NaOH or acid hydrolysis advantageously in the presence of an alcohol such as methanol or ethanol or via an enzymatic cleavage liberate and isolate via, for example, phase separation and subsequent acidification via, for example, H 2 SO 4 .
- an alkali treatment such as aqueous KOH or NaOH or acid hydrolysis
- an alcohol such as methanol or ethanol or via an enzymatic cleavage
- the release of the fatty acids can also be carried out directly without the workup described above.
- the nucleic acids used in the method can advantageously be either a plant cell or plant, either on a separate plasmid or advantageously integrated into the genome of the host cell.
- integration may be at random or by such recombination as to replace the native gene with the incorporated copy, thereby modulating the production of the desired compound by the cell, or by using a gene in trans in that the gene is functionally linked to a functional expression unit which contains at least one sequence ensuring the expression of a gene and at least one sequence ensuring the polyadenylation of a functionally transcribed gene.
- the nucleic acids are brought into the plants via multi-expression cassettes or constructs for multiparallel expression in the organisms, advantageously for multiparallel seed-specific expression of genes. Moose and algae are the only known plant systems that have significant
- PUFAs polyunsaturated fatty acids
- ARA arachidonic acid
- EPA eicosapentaenoic acid
- DHA docosahexaenoic acid
- Moose contain PUFAs in membrane lipids, while algae, algae-related organisms and some fungi also accumulate significant levels of PUFAs in the triacylglycerol fraction. Therefore, nucleic acid molecules isolated from strains which also accumulate PUFAs in the triacylglycerol fraction are particularly advantageous for the process of the invention and thus for modification of the lipid and PUFA production system in a host, in particular plants such as oilseed crops, for example oilseed rape.
- ARA arachidonic acid
- EPA eicosapentaenoic acid
- DHA docosahexaenoic acid
- Synthase (s), hydroperoxide lyase (s) or fatty acid elongase (s) encode advantageously C 16 -, C 18 - or C 2 o fatty acids.
- the fatty acids reacted as substrates in the process are preferably reacted in the form of their acyl-CoA esters and / or their phospholipid esters.
- the polyunsaturated C 18 -fatty acids must first be desaturated by the enzymatic activity of a desaturase and then be extended by at least two carbon atoms via an elongase.
- this enzyme activity leads to C 20 -fatty acids, and after two rounds of elongation to C 22 -fatty acids.
- Desaturases and elongases used in the activity of the method according to the invention lead to C 2O- and / or C 22 -FEW acids with at least four double bonds in the fatty acid molecule, preferably to C 20 -fatty acids having at least four double bonds, particularly preferably to C 20 and / or C 22 fatty acids having at least five or six double bonds, most preferably having six double bonds in the molecule.
- Particularly preferred as products of the method according to the invention are arachidonic acid, eicosapentaenoic acid and / or docosaheic acid.
- the C 20 and / or C 22 fatty acids having at least four double bonds in the fatty acid can be synthesized by the enzymatic activity according to the invention in the form of the free fatty acid or in the form of the esters, such as phospholipids, glycolipids, sphingolipids, phosphoglycerides, monoacylglycerol, diacylglycerol or triacylglycerol. be extended.
- the preferred biosynthesis site of fatty acids, oils, lipids or fats in the advantageously used plants is, for example, generally the seeds or cell layers of the seed, so that a seed-specific expression of the nucleic acids used in the method is useful.
- biosynthesis of fatty acids, oils or lipids need not be limited to the seed tissue, but may also be tissue-specific in all other parts of the plant - for example in epidermal cells or in the tubers.
- the processes can be carried out in the process polyunsaturated fatty acids at least 5%, preferably at least 10%, more preferably at least 20%, most preferably at least 50% compared to the wild-type plant, which does not contain the recombinant nucleic acids increased.
- the polyunsaturated fatty acids produced in the plants used in the process can in principle be increased in two ways.
- the pool of free polyunsaturated fatty acids and / or the proportion of esterified polyunsaturated fatty acids produced by the process can be increased.
- the process according to the invention increases the pool of esterified polyunsaturated fatty acids in the transgenic plant.
- the fatty acids obtained in the process are also suitable as starting material for the chemical synthesis of further products of value. They may be used, for example, in combination with each other or solely for the manufacture of pharmaceuticals, foods, animal feed or cosmetics.
- a further subject of the invention are isolated nucleic acid sequences which code for polypeptides with ⁇ -6-desaturase activity, selected from the group: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 1, b) nucleic acid sequences which are suitable for a protein having the sequence shown in SEQ Or c) derivatives of the nucleic acid sequence shown in SEQ ID NO: 1 which code for polypeptides with at least 70% identity at the amino acid level with SEQ ID NO: 2 and have a ⁇ 6-desaturase activity.
- a further subject of the invention are isolated nucleic acid sequences which code for polypeptides with ⁇ 6-elongase activity, selected from the group: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 3, SEQ ID NO: 5 or SEQ ID NO: 7, b) nucleic acid sequences which code for a protein having the sequence shown in SEQ ID NO: 4, SEQ ID NO: 6 or SEQ ID NO: 8, or c) derivatives of SEQ ID NO: 3, SEQ ID NO: 5 or SEQ ID NO: 7, which code for polypeptides having at least 70% identity at the amino acid level with SEQ ID NO: 4, SEQ ID NO: 6 or SEQ ID NO: 8 and have a ⁇ 6-elongase activity.
- a further subject of the invention are isolated nucleic acid sequences which code for polypeptides with ⁇ 5-desaturase activity, selected from the group: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 9, b) nucleic acid sequences which code for a protein having the sequence shown in SEQ ID NO: 10, or c) derivatives of the nucleic acid sequence shown in SEQ ID NO: 9, which for polypeptides having at least 40% identity on Aminoklareebe ⁇ e with SEQ ID NO: 10 encode and have a ⁇ -5-desaturase activity.
- nucleic acid sequences which code for polypeptides having ⁇ -3-desaturase activity, selected from the group: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 23, b) nucleic acid sequences which are suitable for a protein having the sequence shown in SEQ Or c) derivatives of the nucleic acid sequence shown in SEQ ID NO: 23, which code for polypeptides having at least 40% identity at the amino acid level with SEQ ID NO: 24 and have an ⁇ -3-desaturase activity.
- a further subject of the invention are isolated nucleic acid sequences which code for polypeptides with ⁇ -12-desaturase activity, selected from the group consisting of: a) a nucleic acid sequence with the sequence shown in SEQ ID NO: 25, or b) nucleic acid sequences which are suitable for a protein with the protein disclosed in US Pat Or SEQ ID NO: 26 encode derivatives of the nucleic acid sequence shown in SEQ ID NO: 25 which are suitable for
- a further subject of the invention are gene constructs which contain the nucleic acid sequences according to the invention SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 23 or SEQ ID NO: 25, wherein the nucleic acid is operably linked to one or more regulatory signals.
- acyl-CoA dehydrogenase acy
- biosynthesis genes of the fatty acid or lipid metabolism selected from the group of ⁇ -4-desaturase, ⁇ -8-desaturase, ⁇ -9 elongase or ⁇ -5 elongase are additionally contained.
- all the nucleic acid sequences used in the method according to the invention are derived from an organism such as a plant or a microorganism.
- the nucleic acid sequences are derived from algae such as Ostreococcus, fungi such as Phytophthora or diatoms such as Thalassiosira.
- the nucleic acid sequences used in the method which are useful for ⁇ -6 proteins.
- an expression cassette nucleic acid construct
- a ⁇ 6-desaturase, a ⁇ 6-elongase, a ⁇ 5-desaturase, a ⁇ 5-elongase, a ⁇ 4-desaturase, a ⁇ 12-desaturase and / or an ⁇ -3-desaturase be included.
- the nucleic acids used in the method are advantageously subjected to amplification and ligation in a known manner.
- the procedure is based on the protocol of the Pfu DNA polymerase or of a Pfu / Taq DNA polymerase mixture.
- the primers are selected on the basis of the sequence to be amplified. Conveniently, the primer (s) should be chosen so that the amplicon comprises the entire codogenic sequence from the start to the stop codon.
- the amplificate is conveniently analyzed. For example, the analysis can be carried out after gel electrophoretic separation in terms of quality and quantity. Subsequently, the amplificate can be purified according to a standard protocol (eg Qiagen).
- Suitable cloning vectors are well known to those skilled in the art. These include, in particular, vectors which can be replicated in microbial systems, ie in particular vectors which ensure efficient cloning in yeasts or fungi, and which enable stable transformation of plants. Particular mention should be made of various binary and co-integrated vector systems suitable for T-DNA-mediated transformation. Such vector systems are usually characterized in that they contain at least the vir genes required for the Agrobacterium-mediated transformation as well as the T-DNA limiting sequences (T-DNA border).
- these vector systems also include other cis-regulatory regions such as promoters and terminators and / or selection markers, with which correspondingly transformed organisms can be identified.
- vir genes and T-DNA sequences are located on the same vector
- binary systems are based on at least two vectors, one of them vir genes, but no T-DNA and a second T-DNA, but no carries vir gene.
- the latter vectors are relatively small, easy to manipulate and replicate in both E. coli and Agrobacterium.
- These binary vectors include vectors of the series pBIB-HYG, pPZP, pBecks, pGreen.
- Bin19, pBI101, pBinAR, pGPTV and pCAMBIA are preferably used according to the invention.
- An overview of binary vectors and their use is given by Hellens et al, Trends in Plant Science (2000) 5, 446-151.
- the vectors can first be linearized with restriction endonuclease (s) and then enzymatically modified in a suitable manner. The vector is then purified and an aliquot used for cloning. In cloning, the enzymatically cut and if necessary purified amplicon is cloned with similarly prepared vector fragments using ligase.
- a particular nucleic acid construct or vector or plasmid construct can have one or more codogenic gene segments.
- the codogenic gene segments in these constructs are functionally linked to regulatory sequences.
- the regulatory sequences include, in particular, plant sequences such as the promoters and terminators described above.
- the constructs can advantageously be stably propagated in microorganisms, in particular Escherichia coli and Agrobacterium tumefaciens, under selective conditions and enable a transfer of heterologous DNA into plants or microorganisms.
- nucleic acids used in the method can be introduced into organisms such as microorganisms or advantageously plants and thus used in plant transformation, such as those published in and cited therein Molecular Biology and Biotechnology (CRC Press, Boca Raton, Florida), Chapter 6/7, pp. 71-119 (1993); FF White, Vectors for Gene Transfer to Higher Plants; in: Transgenic Plants, Vol. 1, Engineering and Utilization, eds .: Kung and R. Wu, Academic Press, 1993, 15-38; Genes Transfer, in: Transgenic Plants, Vol. 1, Engineering and Utilization, eds. Kung and R.
- nucleic acids used in the method, the inventive nucleic acids and nucleic acid constructs and / or vectors can thus be used advantageously for the genetic modification of a broad spectrum of organisms to plants, so that they become better and / or more efficient producers of PUFAs.
- the number or activity of ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ 12-desaturase and ⁇ -3 -Desaturase proteins or genes can be increased so that larger amounts of the gene products and thus ultimately larger amounts of the polyunsaturated fatty acids prepared by the process according to the invention are produced with at least four double bonds. Also, a de novo synthesis in a plant lacking the activity and ability to biosynthesize the compounds prior to introduction of the corresponding gene (s) is possible. Correspond- this applies to the combination with other desaturases or elongases or other enzymes from the fatty acid and lipid metabolism.
- the use of different divergent, ie different sequences on DNA sequence level may be advantageous or the use of promoters for gene expression, which allows a different time gene expression, for example, depending on the degree of ripeness of a seed or oil-storing tissue
- a ⁇ 6-desaturase, a ⁇ 6-elongase, a ⁇ 5-desaturase, a ⁇ 5-elongase, a ⁇ 4-desaturase, a ⁇ 12-desaturase and / or an omega-3 desaturase gene in a plant alone or in combination with other genes into a cell can not only increase the biosynthetic flux to the final product, but also increase or de novo create the corresponding triacylglycerol composition.
- the number or activity of other genes necessary for the import of nutrients necessary for the biosynthesis of one or more fatty acids, oils, polar and / or neutral lipids may be increased, such that the concentration of these precursors, cofactors or intermediates within the cells or within the storage compartment, thereby further increasing the ability of the cells to produce PUFAs, as described below.
- ⁇ 6-desaturase By optimizing the activity or increasing the number of one or more ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ 12- Desaturase and / or ⁇ -3-desaturase genes involved in the biosynthesis of these compounds, or by disrupting the activity of one or more genes involved in the degradation of these compounds, may allow the yield, production and / or to increase the efficiency of the production of fatty acid and lipid molecules from plants.
- the isolated nucleic acid molecules used in the method of the invention encode proteins or parts thereof, wherein the proteins or the single protein or parts thereof contain an amino acid sequence sufficiently homologous to an amino acid sequence represented in the sequences SEQ ID NO: 2, SEQ ID NO 4, SEQ ID NO: 6, SEQ ID NO: 8, SEQ ID NO: 10, SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 16, SEQ ID NO: 18, SEQ ID NO: 20 , SEQ ID NO: 22, SEQ ID NO: 24 or SEQ ID NO: 26, so that the proteins or parts thereof still contain a ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase , ⁇ -5 elongase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3-desaturase activity.
- the proteins or portions thereof encoded by the nucleic acid molecule (s) still have their essential enzymatic activity and
- the proteins encoded by the nucleic acid molecules are at least about 40%, preferably at least about 50% or 60%, and more preferably at least about 70%, 80% or 90%, and most preferably at least about 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or more identical to those shown in SEQ ID NO: 2, SEQ ID NO : 4, SEQ ID NO: 6, SEQ ID NO: 8, SEQ ID NO: 10, SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 16, SEQ ID NO: 18, SEQ ID NO: 20, SEQ ID NO: 22, SEQ ID NO: 24 or SEQ ID NO: 26 shown amino acid sequences.
- the homology was calculated over the entire amino acid or nucleic acid sequence range.
- a number of programs that are based on different algorithms are available to the person skilled in the art.
- the algorithms of Needleman and Wunsch or Smith and Waterman provide particularly reliable results.
- the program PiIeUp was used (J. Mol. Evolution., 25, 351-360, 1987, Higgins et al., CABIOS, 5 1989: 151-153) or the programs Gap and BestFit [Needleman and Wunsch (J. Mol. Biol. 48: 443-453 (1970) and Smith and Waterman (Adv. Appl. Math.
- Desaturase is understood to be superior to that represented by the sequence of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 and their derivatives encoded proteins / enzymes in comparison still have at least one enzymatic activity of at least 10%, preferably 20%, more preferably 30% and very particularly 40% and thus the metabolism of compounds necessary for building up fatty acids, fatty acid esters such as diacylglycerides and / or triacylglycerides in a
- Carbon chains in the fatty acid molecule with double bonds at least four, five or six digits are meant.
- Nucleic acids useful in the process are derived from bacteria, fungi, diatoms, animals such as Caenorhabditis or Oncorhynchus or plants such as algae or mosses such as the genera Shewanella, Physcomitrella, Thraustochytrium,
- nucleotide sequences can be used which are suitable for a ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ 12-desaturase and / or encode ⁇ -3 desaturase and which are linked to a nucleotide sequence as in SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 advantageously hybridize under stringent conditions.
- the nucleic acid sequences used in the method are advantageously introduced into an expression cassette which enables expression of the nucleic acids in organisms such as microorganisms or plants.
- nucleic acid sequences which are used for the ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ 12-desaturase and / or ⁇ -3 Desaturase with one or more regulatory signals advantageously functionally linked to increase gene expression.
- These regulatory sequences are intended to allow the targeted expression of genes and protein expression. Depending on the host plant, this may mean, for example, that the gene is expressed and / or overexpressed only after induction, or that it is expressed and / or overexpressed immediately.
- these regulatory sequences are sequences that bind to the inducers or repressors and thus regulate the expression of the nucleic acid.
- the natural regulation of these sequences may still be present before the actual structural genes and may have been genetically altered so that natural regulation is eliminated and expression of genes increased.
- the gene construct may advantageously also contain one or more so-called enhancer sequences functionally linked to the promoter, which allow increased expression of the nucleic acid sequence. Additional advantageous sequences can also be inserted at the 3 'end of the DNA sequences, such as further regulatory elements or terminators.
- This gene construct or gene constructs can be expressed together in the host plant.
- the gene construct or the gene constructs can be inserted in one or more vectors and be present freely in the cell or else be inserted in the genome. It is advantageous for the insertion of further genes in the host genome if the genes to be expressed are present together in a gene construct.
- the regulatory sequences or factors can, as described above, preferably positively influence the gene expression of the introduced genes and thereby increase them.
- enhancement of the regulatory elements can advantageously be done at the transcriptional level by using strong transcription signals such as promoters and / or enhancers.
- an enhancement of the translation is possible by, for example, the stability of the mRNA is improved.
- a further embodiment of the invention is one or more gene constructs which contain one or more sequences represented by SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 or its derivatives and for polypeptides according to SEQ (D NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, SEQ ID NO: 10, SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 16, SEQ ID NO: 18, SEQ ID NO: 20, SEQ ID NO: 22, SEQ ID NO: 24 or SEQ ID NO: 26.
- the said ⁇ 6-desaturase, ⁇ 6-elongase -, ⁇ -5-desaturase, ⁇ -5-elongase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3-desaturase proteins lead advantageously to a desaturation or elongation of fatty acids, wherein the substrate advantageously has one, two, three, four, five or six double bonds and advantageously 18, 20 or 22 carbon atoms in the fatty acid molecule au
- the substrate advantageously has one, two, three, four, five or six double bonds and advantageously 18, 20 or 22 carbon atoms in the fatty acid molecule au
- Advantageous regulatory sequences which can be used for the preparation of the nucleic acid sequences used in the method according to the invention and their subsequent introduction into plants are, for example, in promoters such as the cos, tac, trp, tet, trp tet, Ipp , lac, lpp-lac, laclq, T7, T5, T3, gal, trc-ara, SP6, ⁇ -PR or ⁇ -PL promoter and are advantageously used in Gram-negative bacteria.
- promoters such as the cos, tac, trp, tet, trp tet, Ipp , lac, lpp-lac, laclq, T7, T5, T3, gal, trc-ara, SP6, ⁇ -PR or ⁇ -PL promoter and are advantageously used in Gram-negative bacteria.
- Further advantageous regulatory sequences are, for example, in the Gram-positive promoters amy and SPO2, in the yeast or fungal promoters ADC1, MFa, AC, P-60, CYC1, GAPDH, TEF, rp28, ADH or in the plant promoters CaMV / 35S [Franck et al., Cell 21 (1980) 285-294], PRP1 [Ward et al., Plant. Biol. 22 (1993)], SSU, OCS, Iib4, usp, STLS1, B33, nos or in the ubiquitin or phaseolin promoter.
- inducible promoters such as those which can be induced in EP-AO 388 186 (benzylsulfonamide-inducible), Plant J. 2, 1992: 397-404 (Gatz et al., Tetracycline-inducible), EP-AO 335 528 (US Pat. Abzisinic inducible) or WO 93/21334 (ethanol or cyclohexenol inducible) promoters.
- suitable plant promoters are the cytosolic FBPase promoter or the potato ST-LSI promoter (Stockhaus et al., EMBO J.
- promoters which allow expression in tissues involved in fatty acid biosynthesis.
- Very specific advantages are seed-specific promoters, such as the USP promoter according to the invention, but also other promoters such as the LeB4, DC3, phaseolin or napin promoter.
- Further particularly advantageous promoters are seed-specific promoters which can be used for monocotyledonous or dicotyledonous plants and in US Pat. No.
- promoters are suitable, for example, for monocotylone lpt-2 or Ipt-1 promoter from barley (WO 95/15389 and WO 95/23230), hordein promoter from barley and other suitable promoters described in WO 99/16890 ,
- the PUFA biosynthesis genes should advantageously be expressed seed-specifically in oilseeds.
- seed-specific promoters can be used, or such promoters that are active in the embryo and / or in the endosperm.
- seed-specific promoters can be isolated from both dicotolydone and monocotolydonous plants.
- Plant gene expression can also be facilitated by a chemically inducible promoter (see review in Gatz 1997, Annu Rev. Plant Physiol Plant Mol. Biol., 48: 89-108).
- Chemically inducible promoters are particularly useful when it is desired that gene expression be in a time-specific manner. Examples of such promoters are a salicylic acid-inducible promoter
- each of the nucleic acids used in the method which is responsible for the ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3-desaturase can be expressed under the control of its own preferably a different promoter, since repetitive sequence motifs can lead to instability of the T-DNA or to recombination events.
- the expression cassette is advantageously constructed in such a way that a promoter is followed by a suitable interface for insertion of the nucleic acid to be expressed, advantageously followed by a terminator behind the polylinker in a polylinker.
- This sequence is repeated several times, preferably three, four or five times, so that up to five genes can be brought together in one construct and thus introduced into the transgenic plant for expression.
- the sequence is repeated up to three times or, if necessary, several more times.
- the nucleic acid sequences are inserted for expression via the appropriate interface, for example in the polylinker behind the promoter.
- each nucleic acid sequence has its own promoter and optionally its own terminator.
- Such advantageous constructs are disclosed for example in DE 10102337 or DE 10102338.
- nucleic acid sequences behind a promoter and possibly in front of a terminator.
- the insertion site or the sequence of the inserted nucleic acids in the expression cassette is not of decisive importance, that is to say a nucleic acid sequence can be inserted at the first or last position in the cassette, without this significantly influencing the expression.
- different promoters such as the USP, LegB4 or DC3 promoter and different terminators can be used in the expression cassette. But it is also possible to use only one type of promoter in the cassette. However, this can lead to unwanted recombination events.
- transcription of the introduced genes should be advantageously aborted by suitable terminators at the 3 'end of the introduced biosynthetic genes (beyond the stop codon). It can be used here e.g. the OCS1 terminator. As for the promoters, different terminator sequences should be used for each gene.
- the gene construct may, as described above, also include other genes to be introduced into the organisms. It is possible and advantageous to introduce into the host plants regulatory genes, such as genes for inducers, repressors or enzymes, which intervene by their enzyme activity in the regulation of one or more genes of a biosynthetic pathway, and to express therein. These genes may be of heterologous or homologous origin. Furthermore, in the nucleic acid construct or gene construct, further biosynthesis genes of the fatty acid or lipid metabolism, or else these genes may be on one or more other nucleic acid constructs.
- nucleic acids or genes can be cloned in combination with other elongases and desaturases in expression cassettes, such as those mentioned above, and used to transform plants using Agrobacterium.
- the regulatory sequences or factors can, as described above, preferably positively influence the gene expression of the introduced genes and thereby increase them.
- enhancement of the regulatory elements can advantageously be done at the transcriptional level by using strong transcription signals such as promoters and / or enhancers.
- an enhancement of the translation is possible by, for example, the stability of the mRNA is improved.
- the expression cassettes can in principle be used directly for introduction into the plant or else introduced into a vector.
- These advantageous vectors contain the nucleic acids used in the method which are useful for ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ -12 Or a nucleic acid construct which comprises the nucleic acids used alone or in combination with further biosynthesis genes of the fatty acid or lipid metabolism, such as the acyl-CoA: lysophospholipid acyltransferases, ⁇ -4-desaturases, ⁇ -8-desaturases, ⁇ -5-EIongases and / or ⁇ -9-elongases.
- vector refers to a nucleic acid molecule that can transport another nucleic acid to which it is attached.
- plasmid which is a circular double-stranded DNA loop into which additional DNA segments can be ligated.
- viral vector Another type of vector is a viral vector, where additional DNA segments can be ligated into the viral genome.
- Certain vectors may autonomously replicate in a host cell into which they have been introduced (eg bacterial vectors of bacterial origin of replication). Other vectors are advantageously integrated into the genome of a host cell upon introduction into the host cell and thereby replicated together with the host genome.
- certain vectors are capable of expressing genes with which they function. capable of being steered.
- expression vectors suitable for recombinant DNA techniques are in the form of plasmids.
- plasmid and “vector” can be used interchangeably because the plasmid is the most commonly used vector form.
- the invention is intended to encompass these other forms of expression vectors, such as viral vectors that perform similar functions.
- vector is also intended to encompass other vectors known to those skilled in the art, such as phages, viruses such as SV40, CMV, TMV, transposons, IS elements, phasmids, phagemids, cosmids, linear or circular DNA.
- the recombinant expression vectors advantageously used in the method comprise the below-described nucleic acids or the above-described gene construct in a form suitable for expression of the nucleic acids used in a host cell, which means that the recombinant expression vectors comprise one or more regulatory sequences selected on the basis of for expression to be used host cells, which is operably linked to the nucleic acid sequence to be expressed comprises.
- operably linked means that the nucleotide sequence of interest is bound to the regulatory sequence (s) such that expression of the nucleotide sequence is possible and they are linked to each other such that both sequences fulfill the predicted function ascribed to the sequence (eg, in an in vitro transcription / transcription system or in a host cell when the vector is introduced into the host cell).
- regulatory sequence is intended to include promoters, enhancers, and other expression control elements (eg, polyadenylation signals).
- Regulatory sequences include those that direct the constitutive expression of a nucleotide sequence in many host cell types and those that direct the direct expression of the nucleotide sequence only in certain host cells under certain conditions.
- the design of the expression vector may depend on factors such as the selection of the host cell to be transformed, the level of expression of the desired protein, etc.
- the recombinant expression vectors used can be used for the expression of ⁇ -6-desaturases, ⁇ -6-EIongases, ⁇ -5-desaturases, ⁇ -5-elongases, ⁇ -4-desaturases, ⁇ -12-desaturases and / or ⁇ -3 -Desaturases be designed in prokaryotic or eukaryotic cells. This is advantageous because intermediate steps of the vector construction are often carried out in microorganisms for the sake of simplicity.
- the recombinant expression vector may alternatively be transcribed and translated in vitro using, for example, T7 promoter regulatory sequences and T7 polymerase.
- Typical fusion expression vectors include i.a. pGEX (Pharmacia Biotech Inc., Smith, DB, and Johnson, KS (1988) Gene 67: 31-40), pMAL (New England Biolabs, Beverly, MA) and pRIT5 (Pharmacia, Piscataway, NJ), in which glutathione-S Transferase (GST), maltose E-binding protein or protein A is fused to the recombinant target protein.
- GST glutathione-S Transferase
- pTrc amann et al., (1988) Gene 69: 301-315
- pET 11d a coexpressed viral RNA polymerase
- T7 gn1 a coexpressed viral RNA polymerase
- This viral polymerase is provided by the host strains BL21 (DE3) or HMS174 (DE3) from a resident ⁇ prophage harboring a T7 gn1 gene under the transcriptional control of the lacUV 5 promoter.
- vectors in prokaryotic organisms are known to the person skilled in the art, these vectors are, for example, in E. coli pLG338, pACYC184, the pBR series, such as pBR322, the pUC series, such as pUC18 or pUC19, the M113mp series, pKC30, pRep4, pHS1, pHS2, pPLc236, pMBL24, pLG200, pUR290, plN-HI113-B1, ⁇ gt11 or pBdCI, in Streptomyces plJ101, pIJ364, pIJ702 or pIJ361, in Bacillus pUB110, pC194 or pBD214, in Corynebacterium pSA77 or pAJ667.
- the expression vector is a yeast expression vector.
- yeast expression vectors for expression in the yeast S. cerevisiae include pYeDesaturased (Baldari et al. (1987) Embo J. 6: 229-234), pMFa (Kurjan and
- Vectors and methods for constructing vectors suitable for use in other fungi, such as filamentous fungi include those described in detail in: van den Hondel, C.A.M.J.J., & Punt, PJ. (1991) "Gene transfer systems and vector development for filamentous fungi, in: Applied Molecular Genetics of Fungi, JF Peberdy et al., Eds., Pp.
- yeast vectors are, for example, pAG-1, YEp6, YEpI 3 or pEMBLYe23.
- ⁇ -6-desaturase, ⁇ -6 elongase, ⁇ -5-desaturase, ⁇ -5 elongase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3 desaturase can be used in insect cells using of baculovirus expression vectors.
- Baculovirus vectors available for expression of proteins in cultured insect cells include the pAc series (Smith et al. (1983) Mol. Cell Bio! 3: 2156-2165) and the pVL Series (Lucklow and Summers (1989) Virology 170: 31-39).
- ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ -12-desaturase and / or ⁇ -3-desaturase genes are expressed in plant cells or whole plants (eg spermatophytes, such as crops).
- plant expression vectors include those described in detail in: Becker, D., Kemper, E., Schell, J., and Masterson, R. (1992) "New plant binary vectors with selectable markers located proximal to the left Border ", Plant Mol. Biol.
- a plant expression cassette preferably contains regulatory sequences that can control gene expression in plant cells and are functional so that each sequence can fulfill its function, such as termination of transcription, for example, polyadenylation signals.
- Preferred polyadenylation signals are those derived from Agrobacterium tumefaciens T-DNA, such as the gene 3 of the Ti plasmid pTiACH5 known as octopine synthase (Gielen et al., EMBO J. 3 (1984) 835ff.) Or functional equivalents thereof, but also all other terminators functionally active in plants are suitable.
- a plant expression cassette preferably contains other operably linked sequences, such as translation enhancers, e.g., the overdrive sequence containing the 5'-untranslated tobacco mosaic virus leader sequence, which is the protein / RNA ratio increases (Gallie et al., 1987, Nucl. Acids Research 15: 8693-8711).
- translation enhancers e.g., the overdrive sequence containing the 5'-untranslated tobacco mosaic virus leader sequence, which is the protein / RNA ratio increases (Gallie et al., 1987, Nucl. Acids Research 15: 8693-8711).
- Plant gene expression must be operably linked to a suitable promoter that performs gene expression in a timely, cell or tissue-specific manner.
- useful promoters are constitutive promoters (Benfey et al., EMBO J. 8 (1989) 2195-2202), such as those derived from plant viruses, such as 35S CAMV (Franck et al., Cell 21 (1980) 285-294), 19S CaMV (see also US 5352605 and WO 84/02913) or plant promoters, such as the Rubisco small subunit described in US 4,962,028.
- telomeres are preferred sequences necessary to direct the gene product into its corresponding cell compartment (see review in Kermode, Crit., Plant, 15, 4 (1996) 285) -423 and references cited therein), for example, in the vacuole, the nucleus, all kinds of plastids, such as amyloplasts, chloroplasts, chromoplasts, the extracellular space, the
- Mitochondria the endoplasmic reticulum, oil bodies, peroxisomes and other compartments of plant cells.
- Plant gene expression can also be facilitated by a chemically inducible promoter as described above (see review in Gatz 1997, Annu Rev. Plant Physiol Plant Mol. Biol., 48: 89-108).
- Chemically inducible promoters are particularly useful when it is desired that gene expression be in a time-specific manner. Examples of such promoters are a salicylic acid-inducible promoter (WO 95/19443), a tetracycline-inducible promoter (Gatz et al. (1992) Plant J. 2, 397-404) and an ethanol-inducible promoter.
- Promoters which react to biotic or abiotic stress conditions are also suitable promoters, for example the pathogen-induced PRP 1 gene promoter (Ward et al., Plant Mol. Biol. 22 (1993) 361-366), the heat-inducible hsp ⁇ O promoter from tomato (US Pat. No. 5,187,267), the potato-alpha-amylase-inducible promoter (WO 96/12814) or the wound-inducible pinII promoter (EP-A-0 375 091).
- those promoters which induce gene expression in tissues and organs in which the fatty acid, lipid and oil biosynthesis take place are preferred in sperm cells such as the cells of the endosperm and the developing embryo.
- Suitable promoters are the rapeseed napin promoter (US Pat. No. 5,608,152), the Vicia faba USP promoter (Baeumlein et al., Mol Gen Genet.
- promoters which induce seed-specific expression in monocotyledonous plants such as maize, barley, wheat, rye, rice, and the like.
- Suitable noteworthy promoters are the lpt2 or Ipt1 gene promoter from barley (WO 95/15389 and WO 95/23230) or those described in WO 99/16890 (promoters from the barley hordein gene, the rice glutelin gene , the rice oryzin gene, the rice prolamin gene, the wheat gliadin gene, the wheat glutelin gene, the maize zein gene, the oat glutelin gene, the sorghum kasirin gene, the rye secalin gene).
- multiparallel expression of the ⁇ -6-desaturase, ⁇ -6 elongase, ⁇ -5-desaturase, ⁇ -5 elongase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3 used in the method can be used Desaturase be desired.
- the introduction of such expression cassettes can be carried out via a simultaneous transformation of a plurality of individual expression constructs or preferably by combining a plurality of expression cassettes on a construct. It is also possible to transform a plurality of vectors each having a plurality of expression cassettes and to transfer them to the host cell.
- promoters which induce plastid-specific expression are particularly suitable.
- Suitable promoters such as the viral RNA polymerase promoter, are described in WO 95/16783 and WO 97/06250, and the Arabidopsis clpP promoter described in WO 99/46394.
- Vector DNA can be introduced into prokaryotic or eukaryotic cells via conventional transformation or transfection techniques.
- transformation and “transfection”, conjugation and transduction are intended to encompass a variety of methods known in the art for introducing foreign nucleic acid (eg DNA) into a host cell, including calcium phosphate or calcium chloride coprecipitation, DEAE- Dextran-mediated transfection, lipofection, natural competence, chemically mediated transfer, electroporation or particle bombardment.
- Suitable methods for transforming or transfecting host cells, including plant cells can be found in Sambrook et al. (Molecular Cloning: A Laboratory Manual, 2nd ed., CoId Spring Harbor Laboratory, Col. Spring Harbor Laboratory Press, Col. Spring Harbor, NY, 1989) and others Laboratory Manuals, such as Methods in Molecular Biology, 1995, Vol. 44, Agrobacterium protocols, Eds: Gartland and Davey, Humana Press, Totowa, New Jersey.
- Host cells which are suitable in principle for receiving the nucleic acid according to the invention, the gene product according to the invention or the vector according to the invention are all prokaryotic or eukaryotic organisms.
- the host organisms which are advantageously used as intermediate hosts are microorganisms, such as fungi or yeasts.
- Preferred in the process according to the invention are plants such as oil crop plants which contain large amounts of lipid compounds, such as oilseed rape, evening primrose, hemp, Diestel, peanut, canola, linseed, soybean, safflower, sunflower, borage, or plants such as maize, wheat, rye, Oats, triticale, rice, barley, cotton,
- Cassava, pepper, tagetes, solanaceae plants such as potato, tobacco, eggplant and tomato, vicia species, pea, alfalfa, bush plants (coffee, cocoa, tea), salix species, trees (oil plan, coconut) and forage crops.
- Particularly preferred plants according to the invention are oil crop plants, such as soybean, peanut, rapeseed, canola, flax, hemp, evening primrose, sunflower, safflower, trees (oil palm, coconut).
- nucleic acid sequences enumerated below which code for ⁇ -6-desaturases, ⁇ -6-elongases, ⁇ -5-desaturases, ⁇ -3-desaturases and / or ⁇ -12-desaturases.
- Isolated nucleic acid sequences encoding polypeptides having ⁇ -6 desaturase activity selected from the group consisting of: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 1, b) nucleic acid sequences encoding a protein having the amino acid sequence shown in SEQ ID NO: 2 or c) derivatives of the nucleic acid sequence shown in SEQ ID NO: 1, which code for polypeptides with at least 70% identity at the amino acid level with SEQ ID NO: 2 and have a ⁇ -6-desaturase activity.
- Isolated nucleic acid sequences encoding polypeptides having ⁇ 6-elongase activity selected from the group consisting of: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 3, SEQ ID NO: 5 or SEQ ID NO: 7, b) nucleic acid sequences, which code for a protein having the sequence shown in SEQ ID NO: 4, SEQ ID NO: 6 or SEQ ID NO: 8, or c) derivatives of SEQ ID NO: 3, SEQ ID NO: 5 or SEQ ID NO: 7, which code for polypeptides with at least 70% identity at the amino acid level with SEQ ID NO: 4, SEQ ID NO: 6 or SEQ ID NO: 8 and have a ⁇ 6-elongase activity.
- Isolated nucleic acid sequences encoding polypeptides having ⁇ 5-desaturase activity selected from the group consisting of: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 9, b) nucleic acid sequences encoding a protein having the amino acid sequence shown in SEQ ID NO: 10 or c) derivatives of the nucleic acid sequence shown in SEQ ID NO: 9, which code for polypeptides having at least 40% identity at the amino acid level with SEQ ID NO: 10 and have a ⁇ -5-desaturase activity.
- Isolated nucleic acid sequences encoding polypeptides having ⁇ -3 desaturase activity selected from the group consisting of: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 23, b) nucleic acid sequences encoding a protein having the amino acid sequence shown in SEQ ID NO: 24 or c) derivatives of the nucleic acid sequence shown in SEQ ID NO: 23, which code for polypeptides having at least 40% identity at the amino acid level with SEQ ID NO: 24 and have an ⁇ -3-desaturase activity.
- Isolated nucleic acid sequences which code for polypeptides having ⁇ 12-desaturase activity, selected from the group consisting of: a) a nucleic acid sequence having the sequence shown in SEQ ID NO: 25, or b) nucleic acid sequences encoding a protein having the amino acid sequence shown in SEQ ID NO: Or c) derivatives of the nucleic acid sequence set forth in SEQ ID NO: 25 that encode polypeptides having at least 40% identity at the amino acid level with SEQ ID NO: 26 and have ⁇ -12 desaturase activity.
- nucleic acids according to the invention are derived from organisms such as fungi, plants such as algae or diatoms which can synthesize PUFAs.
- nucleic acid sequences are advantageously derived from the diatome genus Thalassiosira or from the family Prasinophyceae such as the genus Ostreococcus, from the class of Euglenophyceae such as the genus Euglenia or Pythiaceae such as the genus Phytophtora.
- nucleic acid sequences are of the genus and species Phytophtora sojae. These nucleic acid sequences enable in an advantageous Embodiment the synthesis of EPA and / or ARA.
- the following table represents such advantageous nucleic acid sequences.
- the subject of the invention also includes, as described above, isolated nucleic acid sequence useful for polypeptides having ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ -3-desaturase and ⁇ 12- Encode desaturase activity, wherein the encoded by these nucleic acid sequences ⁇ -6-desaturases, ⁇ -6-elongases, ⁇ -5-desaturases, ⁇ -3-desaturases and / or ⁇ -12-desaturases C 18 - or C 2 o fatty acids with one, two, three or four double bonds such as C18: 1 ⁇ 9 , C18: 2 ⁇ 9 12 or C18: 3 ⁇ 9 ' 12 ' 15 polyunsaturated C 2 o fatty acids with three or four double bonds such as
- nucleic acid (molecule) also comprises, in an advantageous embodiment, the untranslated sequence located at the 3 'and 5' end of the coding gene region: at least 500, preferably 200, more preferably 100 nucleotides of the sequence upstream of the 5 'end of the coding region and at least 100, preferably 50, more preferably 20 nucleotides of the sequence downstream of the 3' end of the coding gene region.
- An "isolated" nucleic acid molecule is separated from other nucleic acid molecules present in the natural source of the nucleic acid.
- nucleic acid preferably does not have sequences that naturally flank the nucleic acid in the genomic DNA of the organism from which the nucleic acid is derived (eg, sequences located at the 5 'and 3' ends of the nucleic acid).
- the isolated ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 3-desaturase, and ⁇ 12-desaturase molecules may be less than about 5 kb, 4 kb, 3 kb, 2 kb, 1 kb, 0.5 kb or 0.1 kb to nucleotide sequences that naturally flank the nucleic acid molecule in the genomic DNA of the cell from which the nucleic acid is derived.
- nucleic acid molecules used in the method for example a nucleic acid molecule having a nucleotide sequence of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 or a part thereof can be isolated using standard molecular biology techniques and the sequence information provided herein. For example, comparative algorithms can be used to identify a homologous sequence or homologous, conserved sequence regions at the DNA or amino acid level.
- a nucleic acid molecule comprising a complete sequence of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 or a part thereof, by polymerase chain reaction, wherein Oligonucleotide primers used on the basis of this sequence or parts thereof (eg a nucleic acid molecule comprising the complete sequence or a part thereof can be isolated by polymerase chain reaction using oligonucleotide primers prepared on the basis of this same sequence) ,
- mRNA can be isolated from cells (eg, by the guanidinium thiocyanate extraction method of Chirgwin et al.
- Synthetic oligonucleotide primers for polymerase chain reaction amplification can be prepared on the basis of one of the sequences shown in SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 or using the sequences shown in SEQ ID NO 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, SEQ ID NO: 10, SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 16, SEQ ID NO: 18 , SEQ ID NO: 20, SEQ ID NO: 22, SEQ ID NO: 24 or SEQ ID NO: 26 create amino acid sequences shown.
- a nucleic acid of the invention may be amplified using cDNA or alternatively genomic DNA as a template and suitable oligonucleotide primers according to standard PCR amplification techniques.
- the thus amplified nucleic acid can be cloned into a suitable vector and characterized by DNA sequence analysis.
- Oligonucleotides corresponding to a desaturase nucleotide sequence can be prepared by standard synthetic methods, for example, with an automated DNA synthesizer.
- Nucleic acid sequences having the sequence SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 means, for example, allelic variants with at least about 40 or 50%, preferably at least about 60 or 70%, more preferably at least about 70 or 80%, 90% or 95%, and even more preferably at least about 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92 %, 93%, 94%, 95%, 96%, 97%, 98%, 99% or more identity or homology to one of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ
- nucleic acid molecules of a nucleotide sequence which correspond to one of the amino acid sequences shown in SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 hybridizing nucleotide sequences or a part thereof, eg under stringent Conditions hybridized.
- allelic variants include functional variants which are obtained by deletion, insertion or substitution of nucleotides from / in which is shown in SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 Sequence obtained, but the intention is that the enzyme activity of the resulting synthesized proteins for the insertion of one or more genes is advantageously maintained. Proteins which still retain the enzymatic activity of ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ -5
- Elongase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3-desaturase possess, that is, whose activity is substantially not reduced, means proteins with at least 10%, preferably 20%, more preferably 30%, completely more preferably 40% of the original enzyme activity compared to that represented by SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 encoded protein. Homology was calculated over the entire amino acid or nucleic acid sequence range.
- SEQ ID NO: 1 SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ [D NO: 11, SEQ ID NO: 13, SEQ ID NO: 15
- SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 also mean bacterial, fungal and plant homologs, truncated sequences, single-stranded DNA or RNA of the coding and non-coding DNA sequence.
- Homologues of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 also means derivatives such as promoter variants.
- the promoters upstream of the indicated nucleotide sequences may be modified by one or more nucleotide exchanges, insertions, and / or deletions, without, however, interfering with the functionality or activity of the promoters. It is also possible that the activity of the promoters is increased by modification of their sequence or that they are completely replaced by more active promoters, even from heterologous organisms.
- the lipid synthesis can be divided into two sections: the synthesis of fatty acids and their attachment to sn-glycerol-3-phosphate and the addition or modification of a polar head group.
- Common lipids used in membranes include phospholipids, glycolipids, sphingolipids and phosphoglycerides.
- Fatty acid synthesis begins with the conversion of acetyl-CoA into malonyl-CoA by the acetyl-CoA carboxylase or into acetyl-ACP by the acetyl transacylase.
- these enzymes can transfer the elongated fatty acids again from the CoA esters to the phospholipids. This reaction sequence can optionally be run through several times.
- Precursors for the PUFA biosynthesis are, for example, oleic acid, linoleic acid and linolenic acid. These C18-carbon fatty acids must on C 20 and C 22 are extended, to obtain fatty acids of the eicosa and docosa chain type.
- Arachidonic acid, eicosapentaenoic acid and / or docosahexaenoic acid, advantageously eicosapentaenoic acid and / or docosahexaenoic acid can be produced by means of the desaturases and / or elongases used in the process and subsequently used for various purposes in food, feed, cosmetic or pharmaceutical applications.
- C 20 - and / or C 22 -fatty acids having at least two, advantageously at least four, five or six double bonds in the fatty acid molecule, preferably C 20 - or C 8 -fatty acids with advantageously five or six double bonds in the fatty acid molecule can be prepared with the abovementioned enzymes.
- Substrates of the desaturases and elongases used in the process according to the invention are C 16 , C 18 or C 20 fatty acids, for example linoleic acid, ⁇ -1-linolenic acid, ⁇ -linolenic acid, dihomo- ⁇ -linolenic acid, eicosatetraenoic acid or stearidonic acid.
- Preferred substrates are linoleic acid, ⁇ -linolenic acid and / or ⁇ -linolenic acid, dihomo- ⁇ -iinolenic acid or arachidonic acid, eicosatetraenoic acid or eicosapentaenoic acid.
- the synthesized C 20 - or C 22 -fatty acids having at least four, five or six double bonds in the fatty acid are advantageously added to their triglycerides in the novel process in the form of the free fatty acid or in the form of their esters, for example in the form of their glycerides.
- glycolide is understood to mean a glycerol esterified with one, two or three carboxylic acid residues (mono-, di- or triglyceride).
- glycolide is also meant a mixture of different glycerides.
- the glyceride or glyceride mixture may contain other additives, e.g. contain free fatty acids, antioxidants, proteins, carbohydrates, vitamins and / or other substances.
- a "glyceride” in the sense of the method according to the invention is also understood to mean derivatives derived from glycerol.
- these also include glycerophospholipids and glyceroglycolipids.
- the glycerophospholipids such as lecithin (phosphatidylcholine), cardiolipin, phosphatidylglycerol, phosphatidylserine and alkylacylglycerophospholipids, may be mentioned by way of example here.
- fatty acids must then be transported to various modification sites and incorporated into the triacylglycerol storage lipid.
- Another important step in lipid synthesis is the transfer of fatty acids to the polar head groups, for example, by glycerol-fatty acid acyltransferase (see Frentzen, 1998, Lipid, 100 (4-5): 161-166).
- the PUFAs produced in the process comprise a group of molecules that are no longer able to synthesize, and therefore need to take up, higher animals, or that can no longer sufficiently produce higher animals themselves, and thus have to additionally take up, even though they are readily synthesized by other organisms, such as bacteria For example, cats can no longer synthesize arachidonic acid.
- phospholipids are to be understood as meaning phosphatidylcholine, phosphatidylethanolamine, phosphatidylserine, phosphatidylglycerol and / or phosphatidylinositol, advantageously phosphatidylcholine.
- production or productivity are known in the art and include productivity within a plant cell or plant, that is, the content of the desired fatty acids produced in the process based on the content of all fatty acids in that cell or plant.
- biosynthesis or biosynthetic pathway are known in the art and involve the synthesis of a compound, preferably an organic compound, by a cell from intermediates, for example in a multi-step and highly regulated process.
- degradation or degradation pathway are known in the art and involve the cleavage of a compound, preferably an organic compound, by a cell into degradation products (more generally, smaller or less complex molecules), for example in a multi-step and highly regulated process.
- metabolism is known in the art and includes the entirety of the biochemical reactions that take place in an organism. The metabolism of a particular compound (e.g., the metabolism of a fatty acid) then comprises all of the biosynthetic, modification, and degradation pathways of that compound in the cell that affect that compound.
- derivatives of the nucleic acid molecule of the invention encode again in SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 23 or SEQ ID NO: 25 proteins having at least 40%, preferably about 50 or 60%, preferably at least about 60 or 70%, and more preferably at least about 70 or 80%, 80 to 90%, 90 to 95%, and most preferably at least about 96% , 97%, 98%, 99% or more homology (homology within the meaning of the invention is intended to mean identity) to a complete amino acid sequence of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8 , SEQ ID NO: 10, SEQ ID NO: 24 or SEQ ID NO: 26.
- the invention also encompasses nucleic acid molecules which differ from one of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 23 or SEQ ID NO: 25 nucleotide sequences (and parts thereof) differ due to the degenerate genetic code and thus the same ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ -3-desaturase or ⁇ - 12-desaturase activity encode such as those of the in SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 23 or SEQ ID NO : 25 nucleotide sequences shown is encoded.
- Desaturase and / or ⁇ -12-desaturase gene are the result of natural variation and which do not alter the functional activity of, are intended to be included within the scope of the invention.
- Nucleic acid molecules which are advantageous for the process according to the invention can be prepared on the basis of their homology to the ⁇ 6-desaturase, ⁇ 6 -enongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase , ⁇ -12-desaturase and / or ⁇ -3-desaturase nucleic acids using the sequences or a portion thereof as a hybridization probe according to standard hybridization techniques under stringent hybridization conditions.
- isolated nucleic acid molecules can be used which are at least 15 nucleotides long and under stringent conditions with the nucleic acid molecules having a nucleotide sequence of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 include hybridizing.
- Nucleic acids may also be used at least 25, 50, 100, 250 or more nucleotides are used.
- hybridized under stringent conditions is intended to describe hybridization and washing conditions under which nucleotide sequences that are at least 60% homologous to one another usually remain hybridized to one another.
- the conditions are preferably such that sequences that are at least about 65%, more preferably at least about 70%, and even more preferably at least about 75% or more homologous, are usually hybridized to each other.
- stringent conditions are known to those skilled in the art and can be found in Current Protocols in Molecular Biology, John Wiley & Sons, NY (1989), 6.3.1-6.3.6.
- a preferred, non-limiting example of stringent hybridization conditions are hybridizations in 6x sodium chloride / sodium citrate (SSC) at about 45 ° C, followed by one or more washes in 0.2x SSC, 0.1%. SDS at 50 to 65 ° C. It is known to those skilled in the art that these hybridization conditions differ with respect to the type of nucleic acid and, for example, when organic solvents are present, with respect to the temperature and the concentration of the buffer. The temperature differs, for example under "standard hybridization conditions" depending on the type of nucleic acid between 42 ° C and 58 ° C in aqueous buffer with a concentration of 0.1 to 5 x SSC (pH 7.2).
- the temperature is about 42 ° C under standard conditions.
- the hybridization conditions for DNA: DNA hybrids are, for example, 0.1 x SSC and 20 0 C to 45 ° C, preferably between 30 ° C and 45 ° C.
- the hybridization conditions for DNA: RNA hybrids are, for example, 0.1 x SSC and 30 0 C to 55 ° C, preferably between 45 ° C and 55 ° C.
- sequences are written one below the other for the purpose of optimal comparison (eg, gaps may be inserted into the sequence of a protein or nucleic acid to create an optimal alignment with the other protein or nucleic acid).
- amino acid residues or nucleotides at the corresponding amino acid positions or nucleotide positions are then compared. If a position in one sequence is occupied by the same amino acid residue or nucleotide as the corresponding site in the other sequence, then the molecules are homologous at that position (ie, amino acid or nucleic acid 'homology' as used herein). corresponds to amino acid or nucleic acid "identity".)
- the terms homology and identity are thus to be regarded as synonymous
- the programs or algorithms used are described above.
- An isolated nucleic acid molecule encoding a ⁇ 6-desaturase, ⁇ 6-Elo ⁇ gase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ 12-desaturase and / or ⁇ -3-desaturase which corresponds to a protein sequence of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, SEQ ID NO: 10, SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 16, SEQ ID NO: 18, SEQ ID NO: 20, SEQ ID NO: 22, SEQ ID NO: 24 or SEQ ID NO: 26 may be obtained by introducing one or more nucleotide substitutions, additions or deletions into a nucleotide sequence of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17,
- Mutations can be converted into one of the sequences of SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 15, SEQ ID NO: 17, SEQ ID NO: 19, SEQ ID NO: 21, SEQ ID NO: 23 or SEQ ID NO: 25 are introduced by standard techniques such as site-directed mutagenesis and PCR-mediated mutagenesis.
- conservative amino acid substitutions are made on one or more of the predicted nonessential amino acid residues.
- the amino acid residue is replaced with an amino acid residue having a similar side chain.
- families of amino acid residues have been defined with similar side chains.
- amino acids with basic side chains eg, lysine, arginine, histidine
- acidic side chains eg, aspartic acid, glutamic acid
- uncharged polar side chains eg, glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine
- nonpolar side chains eg Alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan
- beta-branched side chains eg, threonine, valine, isoleucine
- aromatic side chains eg, tyrosine, phenylalanine, tryptophan, histidine
- a predicted nonessential amino acid residue in a ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ -4-desaturase, ⁇ 12-desaturase or ⁇ -3-desaturase thus becomes preferably replaced by another amino acid residue from the same side chain family.
- the mutations may be randomized over all or part of the ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ 12- Desaturase or ⁇ -3-desaturase coding sequence, for example by saturation mutagenesis, and the resulting mutants can be synthesized according to the here described ⁇ 6-desaturase, ⁇ 6-elongase, ⁇ 5-desaturase, ⁇ - 5-elongase, ⁇ -4-desaturase, ⁇ -12-desaturase and / or ⁇ -3-desaturase activity to identify mutants containing the ⁇ -6-desaturase, ⁇ -6 elongase , ⁇ 5-desaturase, ⁇ 5-elongase, ⁇ 4-desaturase, ⁇ 12-desaturase and / or ⁇ -3-desaturase activity to identify mutants
- the encoded protein can be recombinantly expressed, and the activity of the protein can be determined, for example, using of the tests described herein.
- transgenic non-human organism advantageously a transgenic plant, which comprises the nucleic acids according to the invention SEQ ID NO: 1, SEQ ID NO: 3, SEQ ID NO: 5, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 23 or SEQ ID NO: 25 or a gene construct or a vector containing these nucleic acid sequences according to the invention.
- the cloning methods e.g. Restriction cleavage, agarose gel electrophoresis, purification of DNA fragments, transfer of nucleic acids to nitrocellulose and nylon membranes, linkage of DNA fragments, transformation of Escherichia coli cells, culture of bacteria and sequence analysis of recombinant DNA were performed as described in Sambrook et al. (1989) (CoId Spring Harbor Laboratory Press: ISBN 0-87969-309-6).
- RNA of Phytophthora sojae was isolated using the RNAeasy kit from Qiagen (Valencia, CA, US). From the total RNA, poly-A + RNA (mRNA) was isolated using oligo-dT-cellulose (Sambrook et al., 1989). The RNA was reverse-transcribed with Promega's Reverse Transcription System Kit and the synthesized cDNA cloned into the lambda ZAP vector (lambda ZAP Gold, Stratagene). According to the manufacturer's instructions, the cDNA was decompressed to the plasmid DNA.
- mRNA poly-A + RNA
- the plasmid library was then screened for the corresponding putative candidate genes (hybridization of bacterial clones, Sambrook et al., 1989) and positive clones were sequenced. The corresponding cDNA clones were then used for the PCR for the cloning of expression plasmids.
- the corresponding sequences are amplified by PCR using appropriate specific primers and cloned into the yeast expression vector pYES2.1 -TOPO (Invitrogen) according to the manufacturer's instructions. Only the open reading frames of the genes coding for the PUFA proteins are amplified. In addition, a Kozak sequence (Cell 1986, 44: 283-292) is attached to the 5 ':
- Annealing temperature 1 min 55 ° C denaturation temperature: 1 min 94 0 C elongation temperature: 2 min 72 0 C number of cycles: 35
- PCR products are incubated according to the manufacturer's instructions with the plasmid pYES2.1-TOPO (Invitrogen). The incubation reactions are then transformed into E. coli DH5 ⁇ cells (Invitrogen) according to the manufacturer's instructions. Positive clones are identified by PCR (see reaction above) and the plasmid DNA isolated (Qiagen Dneasy). The resulting plasmids are checked by sequencing and transformed into the Saccharomyces strain INVSd (Invitrogen) by electroporation (1500 V). As a control, pYES2.1 (empty vector) is transformed in parallel. The selection of the transfor- Yeasts are plated on complete minimal medium (CMdum) agar plates with 2% glucose but without uracil. After selection, three transformants each are selected for further functional expression.
- Cmum complete minimal medium
- CMdum liquid medium with 2% (w / v) raffinose but without uracil precultures from each 5 ml of CMdum liquid medium with 2% (w / v) raffinose but without uracil are inoculated with the selected transformants and incubated for 2 days at 30 0 C, 200 rpm.
- 5 ml of CMdum liquid medium (without uracif) with 2% raffinose and 300 ⁇ M of different fatty acids are then inoculated with the precultures to an OD 60O of 0.05.
- Expression is induced by the addition of 2% (w / v) galactose.
- the cultures are incubated for a further 96 h at 20 ° C.
- Annealing temperature 1 min 55 0 C denaturation temperature: 1 min 94 0 C elongation temperature: 2 min 72 0 C number of cycles: 35
- the PCR products are incubated for 4 h at 37 0 C with the Restriktio ⁇ senzym Notl.
- the plant expression vector pSUN300-USP is incubated in the same way.
- the PCR products and the 7624 bp vector are separated by agarose gel electrophoresis and the corresponding DNA fragments are excised.
- the DNA is purified by Qiagen gel purification kit according to the manufacturer's instructions.
- vector and PCR products are ligated. To do this, Roche's Rapid Ligation Kit is used. The resulting plasmids are verified by sequencing.
- pSUN300 is a derivative of the plasmid pPZP (Hajdukiewicz P, Svab, Z, Maliga, P., (1994) The small versatile pPZP family of Agrobacterium binary vectors for plant transformation, Plant Mol Biol 25: 989-994).
- pSUN-USP was generated from pSUN300 by inserting into pSUN300 a USP promoter as an EcoRI fragment.
- the polyadenylation signal is that of the OCS gene from the A.
- tumefaciens Ti plasmid (ocs terminator, Genbank Accession V00088) (De Greve.H., Dhaese.P., Seurinck, J., Lemmers.M., Van Montagu M. and Schell, J. Nucleotide sequence and transcript map of the Agrobacterium tumefaciens Ti plasmid-encoded octopine synthase gene J. Mol.
- the USP promoter corresponds nucleotides 1 to 684 (Genbank Accession X56240), wherein part of the non-coding region of the USP gene is contained in the promoter
- the 684 base pair promoter fragment was amplified by means of commercially available T7 standard primer (Stratagene) and with the aid of a synthesized primer. Reaction amplified by standard methods (primer sequence: 5'-
- the PCR fragment was rescored with EcoRI / SalI and inserted into the vector pSUN300 with OCS terminator.
- the result was the plasmid with the name pSUN-USP, which can be used for the plant transformation by means of Agrobacterium tumefaciens.
- the yeast cells from the main cultures are (100 ⁇ g, 5 min, 20 0 C) harvested by centrifugation and washed with 100 mM NaHCO 3, pH 8.0 to remove residual medium and fatty acids to be removed.
- fatty acid methyl esters (FAMEs) are produced by acid methanolysis.
- the cell sediments are incubated with 2 ml of 1N methanolic sulfuric acid and 2% (v / v) dimethoxypropane for 1 h at 80 ° C.
- the extraction of the FAMEs is carried out by extraction twice with petroleum ether (PE).
- the organic phases are each determined once with 2 ml of 100 mM NaHCO 3 , pH 8.0 and 2 ml of distilled water. washed. Subsequently, the PE phases are dried with Na 2 SO 4 , evaporated under argon and taken up in 100 ⁇ l of PE. Samples are placed on a DB-23 capillary column (30 m, 0.25 mm, 0.25 ⁇ m, Agilent) in a Hewlett-Packard 6850 Gas chromatograph with flame ionization detector disconnected.
- the conditions for the GLC analysis are as follows: The oven temperature was programmed from 50 finally 0 C to 250 0 C at a rate of 5 ° C / min and 10 min at 250 ° C (hold). The signals are identified by comparison of the retention times with corresponding fatty acid standards (Sigma). The methodology is described for
- the activity and substrate specificity of the individual genes can be determined after expression and feeding of various fatty acids.
- the fed substrates are present in large quantities in all transgenic yeasts, demonstrating the uptake of these fatty acids into the yeasts.
- the transgenic yeasts show the synthesis of new fatty acids, the products of genes. This means that the genes from Phytophtora sojae can be functionally expressed.
- the substrate specificity of desaturases and elongases can be determined after expression in yeast by feeding using various yeasts. Descriptions for the determination of the individual activities can be found in WO 93/11245 for o3-desaturases, WO 94/11516 for ⁇ 12-desaturases, WO 93/06712, US 5,614,393,
- the activity of the individual desaturases and elongases is calculated from the conversion rate according to the formula [substrate / (substrate + product) * 100].
- Example 8 Generation of Transgenic Plants a) Generation of Transgenic Oilseed Rape Plants (Altered According to Moloney et al., 1992, Plant Cell Reports, 8: 238-242) For generating transgenic rape plants, binary vectors such as those described in US Pat
- Example 5 pSUN plasmids generated with genes from Phytophtora sojae transformed into Agrobacterium tumefaciens C58C1: pGV2260 (Deblaere et al, 1984, Nucl. Acids. Res. 13, 4777-4788).
- oilseed rape plants Var Drakkar, NPZ Nor- wegische Maizucht, Hohenlieth, Germany
- Petioles or hypocotyledons of freshly germinated sterile rape plants were incubated in a Petri dish with a 1:50 agrobacterial dilution for 5-10 minutes. This is followed by a 3-day co-incubation in darkness at 25 ° C on 3MS medium with 0.8% Bacto agar. Cultivation was continued after 3 days at 16 hours light / 8 hours darkness and at weekly intervals on MS medium containing 500 mg / L claforan (Cefotaxime sodium), 50 mg / L kanamycin, 20 microM benzylaminopurine (BAP) and 1, 6 g / l glucose continued.
- MS medium containing 500 mg / L claforan (Cefotaxime sodium), 50 mg / L kanamycin, 20 microM benzylaminopurine (BAP) and 1, 6 g / l glucose continued.
- Regenerated shoots were obtained on 2MS medium with kanamycin and claforan, transferred into soil after rooting and grown in a climatic chamber or greenhouse after cultivation for two weeks, flowered, harvested mature seeds, and expression of the desaturase and elongase genes, respectively investigated by lipid analysis as exemplified in Qiu et al. 2001, J. Biol. Chem. 276, 31561-31566. b) Production of transgenic flax plants
- transgenic flax plants can be carried out, for example, according to the method of Bell et al., 1999, In Vitro Cell. Dev. Biol. Plant. 35 (6): 456-465 by means of particle bombartment.
- Agrobacteria-mediated transformations can be carried out, for example, according to Mlynarova et al. (1994), Plant Cell Report 13: 282-285
- the effect of genetic modification in plants, fungi, algae, ciliates or on the production of a desired compound may be determined by cultivating the modified microorganism or modified plant under suitable conditions (such as those described above), and Medium and / or the cellular components on the increased production of the desired product (ie of lipids or a fatty acid) is examined.
- suitable conditions such as those described above
- These analytical techniques are well known to those skilled in the art and include spectroscopy, thin layer chromatography, staining methods of various types, enzymatic and microbiological methods, and analytical chromatography such as high performance liquid chromatography (see, for example, Ullman, Encyclopedia of Industrial Chemistry, Vol. A2, pp. 89-90 and p.
- Analytical techniques include measurements of nutrient levels in the medium (e.g., sugars, hydrocarbons, nitrogen sources, phosphate and other ions), measurements of biomass composition and growth, analysis of production of common biosynthetic pathway metabolites, and measurements of gases produced during fermentation. Standard methods for these measurements are in Applied Microbial Physiology; A Practical Approach, P.M. Rhodes and P.F. Stanbury, eds., IRL Press, pp.
- fatty acids abbreviations: FAME, fatty acid methyl ester, GC-MS, gas-liquid chromatography-mass spectrometry, TAG, triacylglycerol, TLC, thin-layer chromatography.
- the material to be analyzed may be broken up by sonication, milling in the glass mill, liquid nitrogen and milling or other applicable methods.
- the material must be centrifuged after rupture.
- the sediment is distilled in aqua. re-suspended, heated at 100 ° C. for 10 minutes, cooled on ice and recentrifuged, followed by extraction into 0.5 M sulfuric acid in methanol with 2% dimethoxypropane for 1 hour at 90 ° C., resulting in hydrolyzed oil and lipid compounds, which give transmethylated lipids.
- fatty acid methyl esters are extracted in petroleum ether and finally subjected to GC analysis using a capillary column (Chrompack, WCOT fused silica, CP-Wax-52 CB, 25 microm, 0.32 mm) at a temperature gradient between 170 0 C and 240 0 C for 20 min and 5 min at 240 0 C subjected.
- Chropack, WCOT fused silica, CP-Wax-52 CB, 25 microm, 0.32 mm a temperature gradient between 170 0 C and 240 0 C for 20 min and 5 min at 240 0 C subjected.
- the identity of the resulting fatty acid methyl esters must be defined using standards available from commercial sources (ie Sigma).
- Plant material is first mechanically homogenized by mortars to make it more accessible to extraction.
- the mixture is then heated for 10 min at 100 0 C and sedimented again after cooling on ice.
- the cell sediment is hydrolyzed with 1 M methanolic sulfuric acid and 2% Dimetho- xypropan 1 h at 90 0 C and the lipids are transmethylated.
- the resulting fatty acid methyl esters (FAME) are extracted into petroleum ether.
- the extracted FAMEs are (m Chrompack, WCOT Fused Silica, CP-Wax-52 CB, 25, 0.32 mm) by gas-liquid chromatography with a capillary column and a temperature gradient of 170 0 C to 240 ° C in 20 minutes and 5 minutes at 240 0 C analyzed.
- the identity of the fatty acid methyl esters is confirmed by comparison with corresponding FAME standards (Sigma).
- the identity and position of the double bond can be further analyzed by GC-MS by suitable chemical derivatization of the FAME mixtures, eg to 4,4-dimethoxyoxazoline derivatives (Christie, 1998).
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Plant Pathology (AREA)
- Biophysics (AREA)
- Physics & Mathematics (AREA)
- Cell Biology (AREA)
- Nutrition Science (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Breeding Of Plants And Reproduction By Means Of Culturing (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
- Fats And Perfumes (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ES06725200.7T ES2635013T3 (es) | 2005-03-22 | 2006-03-21 | Procedimiento para la preparación de ácidos grasos poliinsaturados de C20 y C22 con al menos cuatro enlaces dobles en plantas transgénicas |
| CA2600286A CA2600286C (en) | 2005-03-22 | 2006-03-21 | Method for producing polyunsaturated c20 and c22 fatty acids with at least four double bonds in transgenic plants |
| EP06725200.7A EP1866417B1 (de) | 2005-03-22 | 2006-03-21 | Verfahren zur herstellung von mehrfach ungesättigten c20- und c22-fettsäuren mit mindestens vier doppelbindungen in transgenen pflanzen |
| US11/886,857 US8049064B2 (en) | 2005-03-22 | 2006-03-21 | Method for producing polyunsaturated C20- and C22-fatty acids with at least four double bonds in transgenic plants |
| AU2006226338A AU2006226338B9 (en) | 2005-03-22 | 2006-03-21 | Method for producing polyunsaturated C20 and C22 fatty acids with at least four double bonds in transgenic plants |
| DK06725200.7T DK1866417T3 (en) | 2005-03-22 | 2006-03-21 | PROCEDURE FOR MANUFACTURING MULTI-Saturated C20 AND C22 FAT ACIDS WITH AT LEAST FOUR DOUBLE BINDINGS IN TRANSGENIC PLANTS |
| NO20074434A NO20074434L (no) | 2005-03-22 | 2007-09-03 | Fremgangsmate for fremstilling av polyumettede C20 og C22 fettsyrer med minst fire dobbeltbindinger i transgene planter |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102005013779.2 | 2005-03-22 | ||
| DE102005013779A DE102005013779A1 (de) | 2005-03-22 | 2005-03-22 | Verfahren zur Herstellung von mehrfach ungesättigten C20- und C22-Fettsäuren mit mindestens vier Doppelbindungen in transgenen Pflanzen |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2006100241A2 true WO2006100241A2 (de) | 2006-09-28 |
| WO2006100241A3 WO2006100241A3 (de) | 2006-12-07 |
Family
ID=36693120
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2006/060913 WO2006100241A2 (de) | 2005-03-22 | 2006-03-21 | Verfahren zur herstellung von mehrfach ungesättigten c20- und c22-fettsäuren mit mindestens vier doppelbindungen in transgenen pflanzen |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US8049064B2 (de) |
| EP (1) | EP1866417B1 (de) |
| CN (2) | CN101146911A (de) |
| AU (1) | AU2006226338B9 (de) |
| CA (1) | CA2600286C (de) |
| DE (1) | DE102005013779A1 (de) |
| DK (1) | DK1866417T3 (de) |
| ES (1) | ES2635013T3 (de) |
| NO (1) | NO20074434L (de) |
| PL (1) | PL1866417T3 (de) |
| WO (1) | WO2006100241A2 (de) |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007017419A3 (de) * | 2005-08-09 | 2007-05-10 | Basf Plant Science Gmbh | Verfahren zur herstellung von arachidonsäure und/oder eicosapentaensäure in transgenen nutzpflanzen |
| WO2007123999A3 (en) * | 2006-04-20 | 2008-04-24 | Du Pont | Δ17 desaturases and their use in making polyunsaturated fatty acids |
| WO2008054565A1 (en) * | 2006-10-30 | 2008-05-08 | E. I. Du Pont De Nemours And Company | Δ17 desaturase and its use in making polyunsaturated fatty acids |
| WO2009016208A2 (de) * | 2007-07-31 | 2009-02-05 | Basf Plant Science Gmbh | Elongasen und verfahren zur herstellung mehrfach ungesättigter fettsäuren in transgenen organismen |
| WO2010142522A2 (en) | 2009-06-08 | 2010-12-16 | Basf Plant Science Company Gmbh | Novel fatty acid elongation components and uses thereof |
| WO2011006948A1 (en) | 2009-07-17 | 2011-01-20 | Basf Plant Science Company Gmbh | Novel fatty acid desaturases and elongases and uses thereof |
| WO2011023800A1 (en) | 2009-08-31 | 2011-03-03 | Basf Plant Science Company Gmbh | Regulatory nucleic acid molecules for enhancing seed-specific gene expression in plants promoting enhanced polyunsaturated fatty acid synthesis |
| WO2011064183A1 (en) | 2009-11-24 | 2011-06-03 | Basf Plant Science Company Gmbh | Novel fatty acid elongase and uses thereof |
| WO2011064181A1 (en) | 2009-11-24 | 2011-06-03 | Basf Plant Science Company Gmbh | Novel fatty acid desaturase and uses thereof |
| WO2011161093A1 (en) | 2010-06-25 | 2011-12-29 | Basf Plant Science Company Gmbh | Acyltransferases and uses therof in fatty acid production |
| WO2012052468A2 (en) | 2010-10-21 | 2012-04-26 | Basf Plant Science Company Gmbh | Novel fatty acid desaturases, elongases, elongation components and uses therof |
| DE112009003708T5 (de) | 2008-12-12 | 2012-09-13 | Basf Plant Science Gmbh | Desaturasen und Verfahren zur Herstellung mehrfach ungesättigter Fettsäuren in transgenenOrganismen |
| WO2013153404A1 (en) | 2012-04-12 | 2013-10-17 | Rothamsted Research Ltd | Production of omega-3 long chain polyunsaturated fatty acids |
| WO2014020533A2 (en) | 2012-08-03 | 2014-02-06 | Basf Plant Science Company Gmbh | Novel enzymes, enzyme components and uses thereof |
| EP2821492A2 (de) | 2009-05-13 | 2015-01-07 | BASF Plant Science Company GmbH | Acyltransferasen und Verwendungen davon zur Fettsäureherstellung |
| WO2015001505A2 (en) | 2013-07-05 | 2015-01-08 | Basf Plant Science Company Gmbh | Gene expression or activity enhancing elements |
| US9050309B2 (en) | 2012-01-06 | 2015-06-09 | Omthera Pharmaceuticals, Inc. | DPA-enriched compositions of omega-3 polyunsaturated fatty acids in free acid form |
| WO2016075327A2 (en) | 2014-11-14 | 2016-05-19 | Basf Plant Science Company Gmbh | Production of pufas in plants |
| US9492545B2 (en) | 2012-05-07 | 2016-11-15 | Omthera Pharmaceuticals Inc. | Compositions of statins and omega-3 fatty acids |
| WO2020049157A1 (en) | 2018-09-07 | 2020-03-12 | Basf Plant Science Company Gmbh | Improved method for the production of high levels of pufa in plants |
| WO2020049155A1 (en) | 2018-09-07 | 2020-03-12 | Basf Plant Science Company Gmbh | Improved method for the production of high levels of pufa in plants |
| WO2020049159A1 (en) | 2018-09-07 | 2020-03-12 | Basf Plant Science Company Gmbh | Improved method for the production of high levels of pufa in plants |
| WO2020168277A1 (en) | 2019-02-14 | 2020-08-20 | Cargill, Incorporated | Brassica plants producing elevated levels of polyunsaturated fatty acids |
| WO2022098631A1 (en) | 2020-11-04 | 2022-05-12 | Cargill, Incorporated | Harvest management |
| WO2022204454A1 (en) | 2021-03-25 | 2022-09-29 | Cargill, Incorporated | Fertilizer management |
Families Citing this family (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MX347962B (es) | 2003-08-01 | 2017-05-19 | Basf Plant Science Gmbh * | Metodo para la produccion de acidos grasos poli-insaturados en organismos transgenicos. |
| US11952581B2 (en) | 2003-08-01 | 2024-04-09 | Basf Plant Science Gmbh | Process for the production of polyunsaturated fatty acids in transgenic organisms |
| ES2421440T3 (es) | 2004-02-27 | 2013-09-02 | Basf Plant Science Gmbh | Método para preparar ácidos grasos poliinsaturados en plantas transgénicas |
| CA2563875C (en) | 2004-04-22 | 2015-06-30 | Commonwealth Scientific And Industrial Research Organisation | Synthesis of long-chain polyunsaturated fatty acids by recombinant cells |
| ES2529572T3 (es) | 2004-04-22 | 2015-02-23 | Commonwealth Scientific And Industrial Research Organisation | Síntesis de ácidos grasos poliinsaturados de cadena larga por células recombinantes |
| AR059376A1 (es) | 2006-02-21 | 2008-03-26 | Basf Plant Science Gmbh | Procedimiento para la produccion de acidos grasos poliinsaturados |
| AU2013202498B2 (en) * | 2006-08-24 | 2015-04-30 | Basf Plant Science Gmbh | Isolation and characterization of a novel Pythium omega 3 desaturase with specificity to all omega 6 fatty acids longer than 18 carbon chains |
| CN101578363A (zh) | 2006-08-29 | 2009-11-11 | 联邦科学技术研究组织 | 脂肪酸的合成 |
| AU2009286755B2 (en) * | 2008-08-26 | 2015-10-22 | Basf Plant Science Gmbh | Nucleic acids encoding desaturases and modified plant oil |
| US8809559B2 (en) | 2008-11-18 | 2014-08-19 | Commonwelath Scientific And Industrial Research Organisation | Enzymes and methods for producing omega-3 fatty acids |
| CN102220351A (zh) * | 2011-04-04 | 2011-10-19 | 山东农业大学 | 马铃薯致病疫霉△5去饱和酶基因PinD5的功能鉴定与应用 |
| CN102220352A (zh) * | 2011-04-04 | 2011-10-19 | 山东农业大学 | 马铃薯致病疫霉Δ12去饱和酶基因PinD12的功能鉴定与应用 |
| CN102191257A (zh) * | 2011-04-04 | 2011-09-21 | 山东农业大学 | 马铃薯致病疫霉Δ6去饱和酶基因PinD6的克隆及其应用 |
| AU2012362825B2 (en) * | 2011-12-30 | 2016-07-21 | Corteva Agriscience Llc | DHA retention during canola processing |
| PL2861059T3 (pl) | 2012-06-15 | 2017-10-31 | Commw Scient Ind Res Org | Wytwarzanie długołańcuchowych wielonienasyconych kwasów tłuszczowych w komórkach roślinnych |
| CN102978177B (zh) * | 2012-11-09 | 2015-06-03 | 浙江工业大学 | 冬虫夏草合成代谢二十碳五烯酸的△-5-脱氢酶、基因及应用 |
| KR102535223B1 (ko) | 2013-12-18 | 2023-05-30 | 커먼웰쓰 사이언티픽 앤 인더스트리알 리서치 오거니제이션 | 장쇄 다중불포화 지방산을 포함하는 지질 |
| US10045540B2 (en) | 2014-04-01 | 2018-08-14 | Fayetteville State University | Pest control composition |
| CN105219789B (zh) | 2014-06-27 | 2023-04-07 | 联邦科学技术研究组织 | 包含二十二碳五烯酸的提取的植物脂质 |
| CN104450748B (zh) * | 2014-11-18 | 2017-07-18 | 西南大学 | 糙草△6‑脂肪酸脱饱和酶ApD6D基因家族及其重组表达载体和应用 |
| CN108699568A (zh) * | 2015-08-28 | 2018-10-23 | 密苏里大学管理者 | 通过改变乙酰-coa羧化酶的负调控因子来增加植物油含量 |
Citations (60)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1984002913A1 (en) | 1983-01-17 | 1984-08-02 | Monsanto Co | Chimeric genes suitable for expression in plant cells |
| EP0249676A2 (de) | 1986-01-28 | 1987-12-23 | Sandoz Ltd. | Verfahren zur Genexpression in Pflanzen |
| EP0335528A2 (de) | 1988-03-29 | 1989-11-15 | E.I. Du Pont De Nemours And Company | DNS-Promotorfragmente aus Weizen |
| EP0375091A1 (de) | 1988-12-21 | 1990-06-27 | Institut Für Genbiologische Forschung Berlin Gmbh | Wundinduzierbare und kartoffelknollenspezifische transkriptionale Regulation |
| EP0388186A1 (de) | 1989-03-17 | 1990-09-19 | E.I. Du Pont De Nemours And Company | Externe Regulierung der Genexpression |
| US4962028A (en) | 1986-07-09 | 1990-10-09 | Dna Plant Technology Corporation | Plant promotors |
| WO1991013980A1 (en) | 1990-03-16 | 1991-09-19 | Calgene, Inc. | Novel sequences preferentially expressed in early seed development and methods related thereto |
| WO1991013972A1 (en) | 1990-03-16 | 1991-09-19 | Calgene, Inc. | Plant desaturases - compositions and uses |
| WO1992018634A1 (en) | 1991-04-09 | 1992-10-29 | Unilever Plc | Plant promoter involved in controlling lipid biosynthesis in seeds |
| US5187267A (en) | 1990-06-19 | 1993-02-16 | Calgene, Inc. | Plant proteins, promoters, coding sequences and use |
| WO1993006712A1 (en) | 1991-10-10 | 1993-04-15 | Rhone-Poulenc Agrochimie | Production of gamma linolenic acid by a δ6-desaturase |
| WO1993011245A1 (en) | 1991-12-04 | 1993-06-10 | E.I. Du Pont De Nemours And Company | Fatty acid desaturase genes from plants |
| EP0550162A1 (de) | 1991-12-31 | 1993-07-07 | University of Kentucky Research Foundation | Änderung von Fettsäuren durch eine Desaturase in transgenischem Pflanzengewebe |
| WO1993020216A1 (en) | 1991-02-22 | 1993-10-14 | University Technologies International, Inc. | Oil-body protein cis-elements as regulatory signals |
| WO1993021334A1 (en) | 1992-04-13 | 1993-10-28 | Zeneca Limited | Dna constructs and plants incorporating them |
| EP0571741A2 (de) | 1992-03-27 | 1993-12-01 | Sumitomo Chemical Company, Limited | Samen-Expressionsplasmid |
| JPH0662870A (ja) | 1992-08-18 | 1994-03-08 | Mitsui Giyousai Shokubutsu Bio Kenkyusho:Kk | 大豆ホスホエノールピルビン酸カルボキシラーゼ遺伝子のプロモーター領域及び5’非翻訳領域 |
| US5315001A (en) | 1986-07-31 | 1994-05-24 | Calgene Inc. | Acyl carrier protein - DNA sequence and synthesis |
| WO1994011516A1 (en) | 1992-11-17 | 1994-05-26 | E.I. Du Pont De Nemours And Company | Genes for microsomal delta-12 fatty acid desaturases and related enzymes from plants |
| WO1994018337A1 (en) | 1993-02-05 | 1994-08-18 | Monsanto Company | Altered linolenic and linoleic acid content in plants |
| US5352605A (en) | 1983-01-17 | 1994-10-04 | Monsanto Company | Chimeric genes for transforming plant cells using viral promoters |
| WO1995015389A2 (en) | 1993-12-02 | 1995-06-08 | Olsen Odd Arne | Promoter |
| WO1995016783A1 (en) | 1993-12-14 | 1995-06-22 | Calgene Inc. | Controlled expression of transgenic constructs in plant plastids |
| WO1995018222A1 (fr) | 1993-12-28 | 1995-07-06 | Kirin Beer Kabushiki Kaisha | Gene pour acide gras-desaturase, vecteur contenant ledit gene, vegetal contenant ledit gene lui ayant ete transfere, et procede pour creer ledit vegetal |
| WO1995019443A2 (en) | 1994-01-13 | 1995-07-20 | Ciba-Geigy Ag | Chemically regulatable and anti-pathogenic dna sequences and uses thereof |
| WO1995023230A1 (en) | 1994-02-24 | 1995-08-31 | Olsen Odd Arne | Promoter from a lipid transfer protein gene |
| US5504200A (en) | 1983-04-15 | 1996-04-02 | Mycogen Plant Science, Inc. | Plant gene expression |
| WO1996012814A1 (en) | 1994-10-21 | 1996-05-02 | Danisco A/S | Promoter sequence from potato |
| US5530149A (en) | 1992-03-13 | 1996-06-25 | Bayer Aktiengesellschaft | Azolylmethyl-fluorocyclopropyl derivatives |
| WO1996021022A2 (en) | 1994-12-30 | 1996-07-11 | Rhone-Poulenc Agrochimie | Production of gamma linolenic acid by a δ6-desaturase |
| US5565350A (en) | 1993-12-09 | 1996-10-15 | Thomas Jefferson University | Compounds and methods for site directed mutations in eukaryotic cells |
| WO1997006250A1 (en) | 1995-08-10 | 1997-02-20 | Rutgers University | Nuclear-encoded transcription system in plastids of higher plants |
| US5608152A (en) | 1986-07-31 | 1997-03-04 | Calgene, Inc. | Seed-specific transcriptional regulation |
| WO1997021340A1 (en) | 1995-12-14 | 1997-06-19 | Cargill, Incorporated | Plants having mutant sequences that confer altered fatty acid profiles |
| EP0781849A1 (de) | 1995-07-05 | 1997-07-02 | Sapporo Breweries Ltd. | Gewebsspezifischer promotor |
| WO1997030582A1 (en) | 1996-02-06 | 1997-08-28 | Carnegie Institution Of Washington | Production of hydroxylated fatty acids in genetically modified plants |
| EP0794250A1 (de) | 1996-03-04 | 1997-09-10 | Soremartec S.A. | Isolierung und Sequenzierung des FAd2-N Gens der Haselnuss |
| US5677474A (en) | 1988-07-29 | 1997-10-14 | Washington University | Producing commercially valuable polypeptides with genetically transformed endosperm tissue |
| US5689040A (en) | 1995-02-23 | 1997-11-18 | The Regents Of The University Of California | Plant promoter sequences useful for gene expression in seeds and seedlings |
| WO1998001572A1 (de) | 1996-07-03 | 1998-01-15 | Hoechst Research & Technology Deutschland Gmbh & Co. Kg | Genetische transformation von ciliatenzellen durch microcarrier- bombardement mit dna-beladenen goldpartikeln |
| WO1998008962A1 (en) | 1996-08-30 | 1998-03-05 | Monsanto Company | Early seed 5' regulatory sequence |
| WO1998045461A1 (en) | 1997-04-09 | 1998-10-15 | Rhone-Poulenc Agro | An oleosin 5' regulatory region for the modification of plant seed lipid composition |
| WO1998046776A2 (en) | 1997-04-11 | 1998-10-22 | Calgene Llc | Plant fatty acid synthases and use in improved methods for production of medium-chain fatty acids |
| WO1998046763A1 (en) | 1997-04-11 | 1998-10-22 | Calgene Llc | Methods and compositions for synthesis of long chain polyunsaturated fatty acids |
| WO1998046764A1 (en) | 1997-04-11 | 1998-10-22 | Calgene Llc | Methods and compositions for synthesis of long chain polyunsaturated fatty acids in plants |
| WO1998046765A1 (en) | 1997-04-11 | 1998-10-22 | Calgene Llc | Methods and compositions for synthesis of long chain polyunsaturated fatty acids |
| WO1999016890A2 (en) | 1997-09-30 | 1999-04-08 | The Regents Of The University Of California | Production of proteins in plant seeds |
| WO1999027111A1 (en) | 1997-11-24 | 1999-06-03 | University Of Bristol | Desaturase genes and their use |
| WO1999046394A1 (en) | 1998-03-11 | 1999-09-16 | Novartis Ag | Novel plant plastid promoter sequence |
| WO1999064616A2 (en) | 1998-06-12 | 1999-12-16 | Abbott Laboratories | Polyunsaturated fatty acids in plants |
| WO2000012720A2 (en) | 1998-09-02 | 2000-03-09 | Abbott Laboratories | Elongase genes and uses thereof |
| WO2000015815A1 (en) | 1998-09-14 | 2000-03-23 | Pioneer Hi-Bred International, Inc. | Rac-like genes from maize and methods of use |
| WO2000021557A1 (en) | 1998-10-09 | 2000-04-20 | Merck & Co., Inc. | Delta 6 fatty acid desaturase |
| WO2001059128A2 (de) | 2000-02-09 | 2001-08-16 | Basf Aktiengesellschaft | Neues elongasegen und verfahren zur herstellung mehrfach ungesättigter fettsäuren |
| WO2002008401A2 (en) | 2000-07-24 | 2002-01-31 | Abbott Laboratories | Elongase genes and uses thereof |
| WO2002044320A2 (en) | 2000-11-29 | 2002-06-06 | Xenon Genetics Inc. | Human elongase genes and uses thereof |
| DE10102337A1 (de) | 2001-01-19 | 2002-07-25 | Basf Plant Science Gmbh | Verfahren zur Herstellung mehrfach ungesättigter Fettsäuren, neue Biosynthesegene sowie neue pflanzliche Expressionskonstrukte |
| DE10102338A1 (de) | 2001-01-19 | 2002-07-25 | Basf Plant Science Gmbh | Verfahren zur Expression von Biosynthesegenen in pflanzlichen Samen unter Verwendung von neuen multiplen Expressionskonstrukten |
| WO2002077213A2 (en) | 2001-03-26 | 2002-10-03 | University Of Bristol | New elongase gene and production of δ9-polyunsaturated fatty acids |
| WO2005012316A2 (de) | 2003-08-01 | 2005-02-10 | Basf Plant Science Gmbh | Verfahren zur herstellung mehrfach ungesättigter fettsäuren in transgenen |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6075183A (en) * | 1997-04-11 | 2000-06-13 | Abbott Laboratories | Methods and compositions for synthesis of long chain poly-unsaturated fatty acids in plants |
| GB0316629D0 (en) | 2003-07-16 | 2003-08-20 | Univ York | Transgenic cell |
| EP1322752B2 (de) | 2000-09-28 | 2015-02-11 | Bioriginal Food & Science Corp. | Fad4, fad5, fad5-2, and fad6, mitglieder der fettsäuredesaturasefamilie und ihre verwendungen |
| DE10137374A1 (de) | 2001-07-31 | 2003-02-27 | Ipk Inst Fuer Pflanzengenetik | Verfahren zur Herstellung von Arachidonsäure in transgenen Organismen |
| ATE452198T1 (de) | 2002-01-30 | 2010-01-15 | Basf Plant Science Gmbh | Verfahren zur herstellung mehrfach ungesättigter fettsäuren mittels eines neuen elongase-gens |
| DE10219203A1 (de) * | 2002-04-29 | 2003-11-13 | Basf Plant Science Gmbh | Verfahren zur Herstellung mehrfach ungesättigter Fettsäuren in Pflanzen |
-
2005
- 2005-03-22 DE DE102005013779A patent/DE102005013779A1/de not_active Withdrawn
-
2006
- 2006-03-21 US US11/886,857 patent/US8049064B2/en active Active
- 2006-03-21 CA CA2600286A patent/CA2600286C/en active Active
- 2006-03-21 CN CNA200680009188XA patent/CN101146911A/zh active Pending
- 2006-03-21 AU AU2006226338A patent/AU2006226338B9/en active Active
- 2006-03-21 DK DK06725200.7T patent/DK1866417T3/en active
- 2006-03-21 ES ES06725200.7T patent/ES2635013T3/es active Active
- 2006-03-21 WO PCT/EP2006/060913 patent/WO2006100241A2/de not_active Application Discontinuation
- 2006-03-21 PL PL06725200T patent/PL1866417T3/pl unknown
- 2006-03-21 EP EP06725200.7A patent/EP1866417B1/de active Active
- 2006-03-21 CN CN201810649116.6A patent/CN108753857A/zh active Pending
-
2007
- 2007-09-03 NO NO20074434A patent/NO20074434L/no not_active Application Discontinuation
Patent Citations (61)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5352605A (en) | 1983-01-17 | 1994-10-04 | Monsanto Company | Chimeric genes for transforming plant cells using viral promoters |
| WO1984002913A1 (en) | 1983-01-17 | 1984-08-02 | Monsanto Co | Chimeric genes suitable for expression in plant cells |
| US5504200A (en) | 1983-04-15 | 1996-04-02 | Mycogen Plant Science, Inc. | Plant gene expression |
| EP0249676A2 (de) | 1986-01-28 | 1987-12-23 | Sandoz Ltd. | Verfahren zur Genexpression in Pflanzen |
| US4962028A (en) | 1986-07-09 | 1990-10-09 | Dna Plant Technology Corporation | Plant promotors |
| US5315001A (en) | 1986-07-31 | 1994-05-24 | Calgene Inc. | Acyl carrier protein - DNA sequence and synthesis |
| US5608152A (en) | 1986-07-31 | 1997-03-04 | Calgene, Inc. | Seed-specific transcriptional regulation |
| EP0335528A2 (de) | 1988-03-29 | 1989-11-15 | E.I. Du Pont De Nemours And Company | DNS-Promotorfragmente aus Weizen |
| US5677474A (en) | 1988-07-29 | 1997-10-14 | Washington University | Producing commercially valuable polypeptides with genetically transformed endosperm tissue |
| EP0375091A1 (de) | 1988-12-21 | 1990-06-27 | Institut Für Genbiologische Forschung Berlin Gmbh | Wundinduzierbare und kartoffelknollenspezifische transkriptionale Regulation |
| EP0388186A1 (de) | 1989-03-17 | 1990-09-19 | E.I. Du Pont De Nemours And Company | Externe Regulierung der Genexpression |
| WO1991013972A1 (en) | 1990-03-16 | 1991-09-19 | Calgene, Inc. | Plant desaturases - compositions and uses |
| WO1991013980A1 (en) | 1990-03-16 | 1991-09-19 | Calgene, Inc. | Novel sequences preferentially expressed in early seed development and methods related thereto |
| US5187267A (en) | 1990-06-19 | 1993-02-16 | Calgene, Inc. | Plant proteins, promoters, coding sequences and use |
| WO1993020216A1 (en) | 1991-02-22 | 1993-10-14 | University Technologies International, Inc. | Oil-body protein cis-elements as regulatory signals |
| WO1992018634A1 (en) | 1991-04-09 | 1992-10-29 | Unilever Plc | Plant promoter involved in controlling lipid biosynthesis in seeds |
| WO1993006712A1 (en) | 1991-10-10 | 1993-04-15 | Rhone-Poulenc Agrochimie | Production of gamma linolenic acid by a δ6-desaturase |
| US5614393A (en) | 1991-10-10 | 1997-03-25 | Rhone-Poulenc Agrochimie | Production of γ-linolenic acid by a Δ6-desaturase |
| WO1993011245A1 (en) | 1991-12-04 | 1993-06-10 | E.I. Du Pont De Nemours And Company | Fatty acid desaturase genes from plants |
| EP0550162A1 (de) | 1991-12-31 | 1993-07-07 | University of Kentucky Research Foundation | Änderung von Fettsäuren durch eine Desaturase in transgenischem Pflanzengewebe |
| US5530149A (en) | 1992-03-13 | 1996-06-25 | Bayer Aktiengesellschaft | Azolylmethyl-fluorocyclopropyl derivatives |
| EP0571741A2 (de) | 1992-03-27 | 1993-12-01 | Sumitomo Chemical Company, Limited | Samen-Expressionsplasmid |
| WO1993021334A1 (en) | 1992-04-13 | 1993-10-28 | Zeneca Limited | Dna constructs and plants incorporating them |
| JPH0662870A (ja) | 1992-08-18 | 1994-03-08 | Mitsui Giyousai Shokubutsu Bio Kenkyusho:Kk | 大豆ホスホエノールピルビン酸カルボキシラーゼ遺伝子のプロモーター領域及び5’非翻訳領域 |
| WO1994011516A1 (en) | 1992-11-17 | 1994-05-26 | E.I. Du Pont De Nemours And Company | Genes for microsomal delta-12 fatty acid desaturases and related enzymes from plants |
| WO1994018337A1 (en) | 1993-02-05 | 1994-08-18 | Monsanto Company | Altered linolenic and linoleic acid content in plants |
| WO1995015389A2 (en) | 1993-12-02 | 1995-06-08 | Olsen Odd Arne | Promoter |
| US5565350A (en) | 1993-12-09 | 1996-10-15 | Thomas Jefferson University | Compounds and methods for site directed mutations in eukaryotic cells |
| WO1995016783A1 (en) | 1993-12-14 | 1995-06-22 | Calgene Inc. | Controlled expression of transgenic constructs in plant plastids |
| WO1995018222A1 (fr) | 1993-12-28 | 1995-07-06 | Kirin Beer Kabushiki Kaisha | Gene pour acide gras-desaturase, vecteur contenant ledit gene, vegetal contenant ledit gene lui ayant ete transfere, et procede pour creer ledit vegetal |
| WO1995019443A2 (en) | 1994-01-13 | 1995-07-20 | Ciba-Geigy Ag | Chemically regulatable and anti-pathogenic dna sequences and uses thereof |
| WO1995023230A1 (en) | 1994-02-24 | 1995-08-31 | Olsen Odd Arne | Promoter from a lipid transfer protein gene |
| WO1996012814A1 (en) | 1994-10-21 | 1996-05-02 | Danisco A/S | Promoter sequence from potato |
| WO1996021022A2 (en) | 1994-12-30 | 1996-07-11 | Rhone-Poulenc Agrochimie | Production of gamma linolenic acid by a δ6-desaturase |
| US5689040A (en) | 1995-02-23 | 1997-11-18 | The Regents Of The University Of California | Plant promoter sequences useful for gene expression in seeds and seedlings |
| EP0781849A1 (de) | 1995-07-05 | 1997-07-02 | Sapporo Breweries Ltd. | Gewebsspezifischer promotor |
| WO1997006250A1 (en) | 1995-08-10 | 1997-02-20 | Rutgers University | Nuclear-encoded transcription system in plastids of higher plants |
| WO1997021340A1 (en) | 1995-12-14 | 1997-06-19 | Cargill, Incorporated | Plants having mutant sequences that confer altered fatty acid profiles |
| WO1997030582A1 (en) | 1996-02-06 | 1997-08-28 | Carnegie Institution Of Washington | Production of hydroxylated fatty acids in genetically modified plants |
| EP0794250A1 (de) | 1996-03-04 | 1997-09-10 | Soremartec S.A. | Isolierung und Sequenzierung des FAd2-N Gens der Haselnuss |
| WO1998001572A1 (de) | 1996-07-03 | 1998-01-15 | Hoechst Research & Technology Deutschland Gmbh & Co. Kg | Genetische transformation von ciliatenzellen durch microcarrier- bombardement mit dna-beladenen goldpartikeln |
| WO1998008962A1 (en) | 1996-08-30 | 1998-03-05 | Monsanto Company | Early seed 5' regulatory sequence |
| WO1998045461A1 (en) | 1997-04-09 | 1998-10-15 | Rhone-Poulenc Agro | An oleosin 5' regulatory region for the modification of plant seed lipid composition |
| WO1998046765A1 (en) | 1997-04-11 | 1998-10-22 | Calgene Llc | Methods and compositions for synthesis of long chain polyunsaturated fatty acids |
| WO1998046763A1 (en) | 1997-04-11 | 1998-10-22 | Calgene Llc | Methods and compositions for synthesis of long chain polyunsaturated fatty acids |
| WO1998046764A1 (en) | 1997-04-11 | 1998-10-22 | Calgene Llc | Methods and compositions for synthesis of long chain polyunsaturated fatty acids in plants |
| WO1998046776A2 (en) | 1997-04-11 | 1998-10-22 | Calgene Llc | Plant fatty acid synthases and use in improved methods for production of medium-chain fatty acids |
| WO1999016890A2 (en) | 1997-09-30 | 1999-04-08 | The Regents Of The University Of California | Production of proteins in plant seeds |
| WO1999027111A1 (en) | 1997-11-24 | 1999-06-03 | University Of Bristol | Desaturase genes and their use |
| WO1999046394A1 (en) | 1998-03-11 | 1999-09-16 | Novartis Ag | Novel plant plastid promoter sequence |
| WO1999064616A2 (en) | 1998-06-12 | 1999-12-16 | Abbott Laboratories | Polyunsaturated fatty acids in plants |
| WO2000012720A2 (en) | 1998-09-02 | 2000-03-09 | Abbott Laboratories | Elongase genes and uses thereof |
| WO2000015815A1 (en) | 1998-09-14 | 2000-03-23 | Pioneer Hi-Bred International, Inc. | Rac-like genes from maize and methods of use |
| WO2000021557A1 (en) | 1998-10-09 | 2000-04-20 | Merck & Co., Inc. | Delta 6 fatty acid desaturase |
| WO2001059128A2 (de) | 2000-02-09 | 2001-08-16 | Basf Aktiengesellschaft | Neues elongasegen und verfahren zur herstellung mehrfach ungesättigter fettsäuren |
| WO2002008401A2 (en) | 2000-07-24 | 2002-01-31 | Abbott Laboratories | Elongase genes and uses thereof |
| WO2002044320A2 (en) | 2000-11-29 | 2002-06-06 | Xenon Genetics Inc. | Human elongase genes and uses thereof |
| DE10102337A1 (de) | 2001-01-19 | 2002-07-25 | Basf Plant Science Gmbh | Verfahren zur Herstellung mehrfach ungesättigter Fettsäuren, neue Biosynthesegene sowie neue pflanzliche Expressionskonstrukte |
| DE10102338A1 (de) | 2001-01-19 | 2002-07-25 | Basf Plant Science Gmbh | Verfahren zur Expression von Biosynthesegenen in pflanzlichen Samen unter Verwendung von neuen multiplen Expressionskonstrukten |
| WO2002077213A2 (en) | 2001-03-26 | 2002-10-03 | University Of Bristol | New elongase gene and production of δ9-polyunsaturated fatty acids |
| WO2005012316A2 (de) | 2003-08-01 | 2005-02-10 | Basf Plant Science Gmbh | Verfahren zur herstellung mehrfach ungesättigter fettsäuren in transgenen |
Non-Patent Citations (121)
| Title |
|---|
| "Applied Microbial Physiology; A Practical Approach", IRL PRESS, pages: 103 - 129,131- |
| "Current Protocols in Molecular Biology", 1989, JOHN WILEY & SONS, pages: 6.3.1 - 6.3.6 |
| "Gaschromatographie-Massenspektrometrie-Verfahren", LIPIDE, vol. 33, pages 343 - 353 |
| "New plant binary vectors with selectable markers located proximal to the left border", PLANT MOL. BIOL., vol. 20, 1992, pages 1195 - 1197 |
| "Nucleic Acids Hybridization: A Practical Approach", IRL PRESS AT OXFORD UNIVERSITY PRESS |
| "Plant Molecular Biology and Biotechnology", 1993, C PRESS, article "Kapitel 6/7,", pages: 71 - 119 |
| "Plant Molecular Biology and Biotechnology", 1993, CRC PRESS, article "Kapitel 6/7", pages: 71 - 119 |
| "Progress in Lipid Research", vol. 1, 1952, PERGAMON PRESS |
| ALGEN ET AL., MARINE BIOTECHNOLOGY, vol. 1, no. 3, 1999, pages 239 - 251 |
| AMANN ET AL., GENE, vol. 69, 1988, pages 301 - 315 |
| B. JENES ET AL.: "Transgenic Plants, Bd. 1, Engineering and Utilization", 1993, ACADEMIC PRESS, article "Techniques for Gene Transfer", pages: 128 - 143 |
| BAEUMLEIN ET AL., MOL GEN GENET, vol. 225, no. 3, 1991, pages 459 - 67 |
| BAEUMLEIN ET AL., PLANT JOURNAL, vol. 2, no. 2, 1992, pages 233 - 9 |
| BALDARI ET AL., EMBO J., vol. 6, 1987, pages 229 - 234 |
| BELTER, P.A. ET AL.: "Bioseparations: downstream processing for Biotechnology", 1988, JOHN WILEY AND SONS |
| BENFEY ET AL., EMBO J., vol. 8, 1989, pages 2195 - 2202 |
| BEVAN, M.W.: "Binary Agrobacterium vectors for plant transformation", NUCL. ACIDS RES., vol. 12, 1984, pages 8711 - 8721 |
| BROWN: "Essential Molecular Biology: A Practical Approach", 1991, IRL PRESS AT OXFORD UNIVERSITY PRESS |
| BROWSE ET AL., ANALYTIC BIOCHEMISTRY, vol. 152, 1986, pages 141 - 145 |
| CABIOS, vol. 5, 1989, pages 151 - 153 |
| CALDER, PROC. NUTR. SOC., vol. 61, 2002, pages 345 - 358 |
| CELL, vol. 44, 1986, pages 283 - 292 |
| CHIRGWIN ET AL., BIOCHEMISTRY, vol. 18, 1979, pages 5294 - 5299 |
| CHRISTIE, WILLIAM W., ADVANCES IN LIPID METHODOLOGY |
| CHRISTIE, WILLIAM W.: "Gas Chromatography and Lipids. A Practical Guide - Ayr", vol. IX, 1992, OILY PRESS, pages: 307 |
| CHRISTIE: "Advances on Lipid Methodology", 1997, OILY PRESS, pages: 119 - 169 |
| CLELAND; JAMES, J. RHEUMATOL., vol. 27, 2000, pages 2305 - 2307 |
| DE GREVE,H.; DHAESE,P.; SEURINCK,J.; LEMMERS,M.; VAN MONTAGU,M.; SCHE,J.: "Nucleotide sequence and transcript map of the Agrobacterium tumefaciens Ti plasmid-encoded octopine synthase gene", J. MOL. APPL. GENET., vol. 1, no. 6, 1982, pages 499 - 511 |
| DEBLAERE ET AL., NUCL. ACIDS. RES., vol. 13, 1984, pages 4777 - 4788 |
| DECHOW, F.J.: "Separation and purification techniques in biotechnology", 1989, NOYES PUBLICATIONS |
| DOMERGUE ET AL., EUR. J. BIOCHEM., vol. 269, 2002, pages 4105 - 4113 |
| E. UCCIANI: "Nouveau Dictionnaire des Huiles Vegetales", 1995, LAVOISIER, ISBN: 2-7430-0009-0 |
| F.C. NEIDHARDT ET AL.: "E. coli und Salmonella", 1996, ASM PRESS, pages: 612 - 636 |
| F.F. WHITE: "Transgenic Plants, Bd. 1, Engineering and Utilization", 1993, ACADEMIC PRESS, article "Vectors for Gene Transfer in Higher Plants" |
| F.F. WHITE; B. JENES ET AL.: "Transgenic Plants, Bd. 1, Engineering and Utilization", 1993, ACADEMIC PRESS, article "Techniques for Gene Transfer", pages: 128 - 43 |
| FALLON, A. ET AL.: "Applications of HPLC in Biochemistry", LABORATORY TECHNIQUES IN BIOCHEMISTRY AND MOLECULAR BIOLOGY, vol. 17, 1987 |
| FRANCK ET AL., CELL, vol. 21, 1980, pages 285 - 294 |
| FRENTZEN, LIPID, vol. 100, no. 4-5, 1998, pages 161 - 166 |
| GALLIE ET AL., NUCL. ACIDS RESEARCH, vol. 15, 1987, pages 8693 - 8711 |
| GARTLAND UND DAVEY,: "Methods in Molecular Biology", vol. 44, 1995, HUMANA PRESS |
| GATZ ET AL., PLANT J., vol. 2, 1992, pages 397 - 404 |
| GATZ, ANNU. REV. PLANT PHYSIOL. PLANT MOL. BIOL., vol. 48, 1997, pages 89 - 108 |
| GERHARDT, PROG. LIPID R., vol. 31, 1992, pages 397 - 417 |
| GIELEN ET AL., EMBO J., vol. 3, 1984, pages 835 |
| GOEDDEL: "Gene Expression Technology: Methods in Enzymology", vol. 185, 1990, ACADEMIC PRESS |
| GRUBER; CROSBY: "Methods in Plant Molecular Biology and Biotechnolgy", CRC PRESS, pages: 89 - 108 |
| GÜHNEMANN-SCHÄFER; KINDL, BIOCHIM. BIOPHYS ACTA, vol. 1256, 1995, pages 181 - 186 |
| HAJDUKIEWICZ,P; SVAB, Z; MALIGA, P.: "The small versatile pPZP family of Agrobacterium binary vectors for plant transformation", PLANT MOL BIOL, vol. 25, 1994, pages 989 - 994 |
| HELLENS ET AL., TRENDS IN PLANT SCIENCE, vol. 5, 2000, pages 446 - 451 |
| HERSKOWITZ, CELL, vol. 30, 1982, pages 933 - 943 |
| HIGGINS ET AL., CABIOS, vol. 5, 1989, pages 151 - 153 |
| HONG ET AL., LIPIDS, vol. 37, 2002, pages 863 - 868 |
| HORROCKS, LA; YEO YK, PHARMACOL RES, vol. 40, 1999, pages 211 - 225 |
| HUANG ET AL., LIPIDS, vol. 34, 1999, pages 649 - 659 |
| J. MOL. EVOLUTION., vol. 25, 1987, pages 351 - 360 |
| J.W. BENNET & L.L. LASURE,: "More Gene Manipulations in Fungi", ACADEMIC PRESS, pages: 396 - 428 |
| K. TOTANI; K. OBA, LIPIDS, vol. 22, 1987, pages 1060 - 1062 |
| KENNEDY, J.F.; CABRAL, J.M.S.: "Recovery processes for biological Materials", 1992, JOHN WILEY AND SONS |
| KERMODE, CRIT. REV. PLANT SCI., vol. 15, no. 4, 1996, pages 285 - 423 |
| KINNEY: "Genetic Engeneering", vol. 19, 1997, pages: 149 - 166 |
| KUNAU ET AL., PROG. LIPID RES., vol. 34, 1995, pages 267 - 342 |
| KUNG UND R. WU,: "Transgenic Plants, Bd. 1, Engineering and Utilization", 1993, ACADEMIC PRESS, article "Vectors for Gene Transfer in Higher Plants", pages: 15 - 38 |
| LENGELER ET AL.: "Biology of Procaryotes", 1999, THIEME |
| LUCKLOW; SUMMERS, VIROLOGY, vol. 170, 1989, pages 31 - 39 |
| M. AKIMOTO ET AL., APPL. BIOCHEMISTRY AND BIOTECHNOLOGY, vol. 73, 1998, pages 269 - 278 |
| MAGNUSON, K. ET AL., MICROBIOLOGICAL REVIEWS, vol. 57, 1993, pages 522 - 542 |
| MCKEON ET AL., METHODS IN ENZYMOL., vol. 71, 1981, pages 12141 - 12147 |
| MICHAELSON ET AL., FEBS LETTERS., vol. 439, no. 3, 1998, pages 215 - 218 |
| MIKOKLAJCZAK ET AL., JOURNAL OF THE AMERICAN OIL CHEMICAL SOCIETY, vol. 38, 1961, pages 678 - 681 |
| MILLAR ET AL., PLANT CELL, vol. 11, 1999, pages 825 - 838 |
| MILLAR; KUNST, PLANT JOURNAL, vol. 12, 1997, pages 121 - 131 |
| MLYNAROVA ET AL., PLANT CELL REPORT, vol. 13, 1994, pages 282 - 285 |
| MOLONEY ET AL., PLANT CELL REPORTS, vol. 8, 1992, pages 238 - 242 |
| MURASHIGE; SKOOG, PHYSIOL. PLANT., vol. 15, 1962, pages 473 |
| MURPHY; ROSS, PLANT JOURNAL., vol. 13, no. 1, 1998, pages 1 - 16 |
| NAPIER; MICHAELSON, LIPIDS, vol. 36, no. 8, 2001, pages 761 - 766 |
| NEEDLEMAN; WUNSCH, J. MOL. BIOL., vol. 48, 1970, pages 443 - 453 |
| OHLROGGE; BROWSE, PLANT CELL, vol. 7, 1995, pages 957 - 970 |
| ON BELL ET AL., IN VITRO CELL. DEV. BIOL.-PLANT, vol. 35, no. 6, 1999, pages 456 - 465 |
| PLANT J., vol. 2, 1992, pages 397 - 404 |
| POTRYKUS, ANNU. REV. PLANT PHYSIOL. PLANT MOLEC. BIOL., vol. 42, 1991, pages 205 - 225 |
| POULOS, A, LIPIDS, vol. 30, 1995, pages 1 - 14 |
| POUWELS, P.H., ET AL.,: "Cloning Vectors", 1985, ELSEVIER, ISBN: 0 444 904018 |
| QIU ET AL., J. BIOL. CHEM., vol. 276, 2001, pages 31561 - 31566 |
| R. VAZHAPPILLY; F. CHEN, BOTANICA MARINA, vol. 41, 1998, pages 553 - 558 |
| REHM ET AL.: "Biotechnology", vol. 3, 1993, VCH, article "Product recovery and purification", pages: 469 - 714 |
| ROMANOS, M.A. ET AL.: "Foreign gene expression in yeast: a review", YEAST, vol. 8, 1992, pages 423 - 488, XP002939357, DOI: doi:10.1002/yea.320080602 |
| SAKURADANI, E. ET AL., GENE, vol. 238, 1999, pages 445 - 453 |
| SAMBROOK ET AL.: "Molecular Cloning", 1989, COLD SPRING HARBOR LABORATORY |
| SAMBROOK ET AL.: "Molecular Cloning: A Laboratory Manual. 2. Aufl.,", 1989, COLD SPRING HARBOR LABORATORY PRESS |
| SAMBROOK ET AL.: "Molecular Cloning: A Laboratory Manual.., 2. Aufl.,", 1989, COLD SPRING HARBOR LABORATORY |
| SAMBROOK, J.; FRITSCH, E.F.; MANIATIS, T.: "Molecular Cloning: A Laboratory Manual", 1989, COLD SPRING HARBOR LABORATORY PRESS, article "Kapiteln 16 und 17" |
| SANGER ET AL., PROC. NATL. ACAD. SCI. USA, vol. 74, 1977, pages 5463 - 5467 |
| SAYANOVA ET AL., JOURNAL OF EXPERIMENTAL BOTANY., vol. 52, no. 360, 2001, pages 1581 - 1585 |
| SCHMIDT, R.; WILLMITZER, L.: "High efficiency Agrobacterium tumefaciens-mediated transformation of Arabidopsis thaliana leaf and cotyledon explants", PLANT CELL REP., 1988, pages 583 - 586 |
| SCHULTZ ET AL., GENE, vol. 54, 1987, pages 113 - 123 |
| SHAEIWITZ, J.A.; HENRY, J.D.: "Ullmann's Encyclopedia of Industrial Chemistry", vol. B3, 1988, VCH, article "Biochemical Separations", pages: 1 - 27 |
| SHANKLIN; CAHOON, ANNU. REV. PLANT PHYSIOL. PLANT MOL. BIOL., vol. 49, 1998, pages 611 - 641 |
| SHIMIKAWA, WORLD REV. NUTR. DIET., vol. 88, 2001, pages 100 - 108 |
| SMITH ET AL.: "Mol. Cell Biol.", vol. 3, 1983, pages: 2156 - 2165 |
| SMITH, D.B.; JOHNSON, K.S., GENE, vol. 67, 1988, pages 31 - 40 |
| SMITH; WATERMAN, ADV. APPL. MATH., vol. 2, 1981, pages 482 - 489 |
| SPERLING ET AL., ARCH. BIOCHEM. BIOPHYS, vol. 388, no. 2, 2001, pages 293 - 298 |
| SPRECHER, BIOCHIM. BIOPHYS. ACTA, vol. 1486, 2000, pages 219 - 231 |
| STOCKHAUS ET AL., EMBO J., vol. 8, 1989, pages 2445 |
| STUDIER ET AL.: "Gene Expression Technology: Methods in Enzymology", vol. 185, 1990, ACADEMIC PRESS, pages: 60 - 89 |
| STUKEY ET AL., J. BIOL. CHEM., vol. 265, 1990, pages 20144 - 20149 |
| STYMNE ET AL.: "Biochemistry and Molecular Biology of Membrane and Storage Lipids of Plants", 1993, AMERICAN SOCIETY OF PLANT PHYSIOLOGISTS, pages: 150 - 158 |
| TAKEYAMA, H. ET AL., MICROBIOLOGY, vol. 143, 1997, pages 2725 - 2731 |
| TOCHER ET AL., PROG. LIPID RES., vol. 37, 1998, pages 73 - 117 |
| TVRDIK ET AL., JCB, vol. 149, 2000, pages 707 - 717 |
| ULLMAN: "Encyclopedia of Industrial Chemistry", vol. A2, 1985, VCH, pages: 89 - 90,443-6 |
| VAN DEN HONDEL, C.A.M.J.J. ET AL.: "More Gene Manipulations in Fungi", 1991, ACADEMIC PRESS, article "Heterologous gene expression in filamentous fungi", pages: 396 - 428 |
| VAN DEN HONDEL, C.A.M.J.J.; PUNT, P.J. ET AL.: "Applied Molecular Genetics of Fungi", 1991, CAMBRIDGE UNIVERSITY PRESS, article "Gene transfer systems and vector development for filamentous fungi", pages: 1 - 28 |
| VOELKER: "Genetic Engeneering", vol. 18, 1996, pages: 111 - 13 |
| VON CAHOON ET AL., PROC. NATL. ACAD. SCI. USA, vol. 96, no. 22, 1999, pages 12935 - 12940 |
| WADA ET AL., NATURE, vol. 347, 1990, pages 200 - 203 |
| WANG ET AL., PLANT PHYSIOL. BIOCHEM., vol. 26, 1988, pages 777 - 792 |
| WARD ET AL., PLANT. MOL. BIOL., vol. 22, 1993, pages 361 - 366 |
| YU, R. ET AL., LIPIDS, vol. 35, 2000, pages 1061 - 1064 |
| ZANK, T.K. ET AL., PLANT JOURNAL, vol. 31, 2002, pages 255 - 268 |
Cited By (59)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007017419A3 (de) * | 2005-08-09 | 2007-05-10 | Basf Plant Science Gmbh | Verfahren zur herstellung von arachidonsäure und/oder eicosapentaensäure in transgenen nutzpflanzen |
| US8134046B2 (en) | 2005-08-09 | 2012-03-13 | Basf Plant Science Gmbh | Method for producing arachidonic acid and/or eicosapentaenoic acid in useful transgenic plants |
| AU2007240741B2 (en) * | 2006-04-20 | 2013-01-31 | E. I. Du Pont De Nemours And Company | Delta17 desaturases and their use in making polyunsaturated fatty acids |
| WO2007123999A3 (en) * | 2006-04-20 | 2008-04-24 | Du Pont | Δ17 desaturases and their use in making polyunsaturated fatty acids |
| US7465793B2 (en) | 2006-04-20 | 2008-12-16 | E.I. Du Pont De Nemours And Company | Synthetic Δ17 desaturase derived from Phytophthora ramourum and its use in making polyunsaturated fatty acids |
| WO2008054565A1 (en) * | 2006-10-30 | 2008-05-08 | E. I. Du Pont De Nemours And Company | Δ17 desaturase and its use in making polyunsaturated fatty acids |
| US8241884B2 (en) | 2006-10-30 | 2012-08-14 | E I Du Pont De Nemours And Company | Δ17 desaturase and its use in making polyunsaturated fatty acids |
| US7556949B2 (en) | 2006-10-30 | 2009-07-07 | E.I. Du Pont De Nemours And Company | Δ17 desaturase and its use in making polyunsaturated fatty acids |
| AU2007314481B2 (en) * | 2006-10-30 | 2012-11-29 | E. I. Du Pont De Nemours And Company | Delta17 desaturase and its use in making polyunsaturated fatty acids |
| WO2009016208A2 (de) * | 2007-07-31 | 2009-02-05 | Basf Plant Science Gmbh | Elongasen und verfahren zur herstellung mehrfach ungesättigter fettsäuren in transgenen organismen |
| AU2008281760B2 (en) * | 2007-07-31 | 2014-07-17 | Basf Plant Science Gmbh | Elongases and methods for producing polyunsaturated fatty acids in transgenic organisms |
| WO2009016208A3 (de) * | 2007-07-31 | 2009-05-07 | Basf Plant Science Gmbh | Elongasen und verfahren zur herstellung mehrfach ungesättigter fettsäuren in transgenen organismen |
| US8318914B2 (en) | 2007-07-31 | 2012-11-27 | Bioriginal Food & Science Corp. | Elongases and methods for producing polyunsaturated fatty acids in transgenic organisms |
| EP2669380A1 (de) | 2008-12-12 | 2013-12-04 | BASF Plant Science GmbH | Desaturasen und Verfahren zur Herstellung von mehrfach ungesättigten Fettsäuren in transgenen Organismen |
| US8822662B2 (en) | 2008-12-12 | 2014-09-02 | Basf Plant Science Company Gmbh | Desaturases and process for the production of polyunsaturated fatty acids in transgenic organisms |
| US9139854B2 (en) | 2008-12-12 | 2015-09-22 | Basf Plant Science Company Gmbh | Desaturases and process for the production of polyunsaturated fatty acids in transgenic organisms |
| DE112009003708T5 (de) | 2008-12-12 | 2012-09-13 | Basf Plant Science Gmbh | Desaturasen und Verfahren zur Herstellung mehrfach ungesättigter Fettsäuren in transgenenOrganismen |
| EP2821492A2 (de) | 2009-05-13 | 2015-01-07 | BASF Plant Science Company GmbH | Acyltransferasen und Verwendungen davon zur Fettsäureherstellung |
| DE112010002353T5 (de) | 2009-06-08 | 2012-08-09 | Basf Plant Science Company Gmbh | Neue fettsäure-elongations-komponenten und anwenduingen davon |
| WO2010142522A2 (en) | 2009-06-08 | 2010-12-16 | Basf Plant Science Company Gmbh | Novel fatty acid elongation components and uses thereof |
| US8993841B2 (en) | 2009-06-08 | 2015-03-31 | Basf Plant Science Company Gmbh | Fatty acid elongation components and uses thereof |
| WO2011006948A1 (en) | 2009-07-17 | 2011-01-20 | Basf Plant Science Company Gmbh | Novel fatty acid desaturases and elongases and uses thereof |
| DE112010002967T5 (de) | 2009-07-17 | 2012-10-11 | Basf Plant Science Company Gmbh | Neue Fettsäuredesaturasen und -elongasen und Anwendungen davon |
| EP3178937A1 (de) | 2009-08-31 | 2017-06-14 | BASF Plant Science Company GmbH | Regulatorische nukleinsäuremoleküle zur erhöhung der samenspezifischen genexpression bei pflanzen,welche die synthese von mehrfach ungesättigten fettsäuren begünstigen |
| WO2011023800A1 (en) | 2009-08-31 | 2011-03-03 | Basf Plant Science Company Gmbh | Regulatory nucleic acid molecules for enhancing seed-specific gene expression in plants promoting enhanced polyunsaturated fatty acid synthesis |
| EP3121283A1 (de) | 2009-08-31 | 2017-01-25 | BASF Plant Science Company GmbH | Regulatorische nukleinsäuremoleküle für erhöhte samenspezifische genexpression bei pflanzen, welche die erhöhte synthese von mehrfach ungesättigten fettsäuren unterstützen |
| EP3418387A1 (de) | 2009-08-31 | 2018-12-26 | Basf Plant Science Company GmbH | Regulatorische nukleinsäuremoleküle zur erhöhung der samenspezifischen genexpression in pflanzen zur förderung der erhöhten synthese von mehrfach ungesättigten fettsäuren |
| WO2011064181A1 (en) | 2009-11-24 | 2011-06-03 | Basf Plant Science Company Gmbh | Novel fatty acid desaturase and uses thereof |
| WO2011064183A1 (en) | 2009-11-24 | 2011-06-03 | Basf Plant Science Company Gmbh | Novel fatty acid elongase and uses thereof |
| WO2011161093A1 (en) | 2010-06-25 | 2011-12-29 | Basf Plant Science Company Gmbh | Acyltransferases and uses therof in fatty acid production |
| WO2012052468A2 (en) | 2010-10-21 | 2012-04-26 | Basf Plant Science Company Gmbh | Novel fatty acid desaturases, elongases, elongation components and uses therof |
| DE112011103527T5 (de) | 2010-10-21 | 2013-10-17 | Basf Plant Science Company Gmbh | Neue Fettsäure-Desaturasen, -Elongasen, -Elongations-Komponenten und Anwendungen davon |
| EP2695936A1 (de) | 2010-10-21 | 2014-02-12 | BASF Plant Science Company GmbH | Neue Fettsäuredesaturasen und Verwendungen davon |
| US9458477B2 (en) | 2010-10-21 | 2016-10-04 | Basf Plant Science Company Gmbh | Fatty acid desaturases, elongases, elongation components and uses thereof |
| US9050309B2 (en) | 2012-01-06 | 2015-06-09 | Omthera Pharmaceuticals, Inc. | DPA-enriched compositions of omega-3 polyunsaturated fatty acids in free acid form |
| US9050308B2 (en) | 2012-01-06 | 2015-06-09 | Omthera Pharmaceuticals, Inc. | DPA-enriched compositions of omega-3 polyunsaturated fatty acids in free acid form |
| US10117844B2 (en) | 2012-01-06 | 2018-11-06 | Omthera Pharmaceuticals, Inc. | DPA-enriched compositions of omega-3 polyunsaturated fatty acids in free acid form |
| WO2013153404A1 (en) | 2012-04-12 | 2013-10-17 | Rothamsted Research Ltd | Production of omega-3 long chain polyunsaturated fatty acids |
| US10881631B2 (en) | 2012-04-12 | 2021-01-05 | Rothamsted Research Ltd. | Production of omega-3 long chain polyunsaturated fatty acids |
| US9492545B2 (en) | 2012-05-07 | 2016-11-15 | Omthera Pharmaceuticals Inc. | Compositions of statins and omega-3 fatty acids |
| WO2014020533A2 (en) | 2012-08-03 | 2014-02-06 | Basf Plant Science Company Gmbh | Novel enzymes, enzyme components and uses thereof |
| US10731169B2 (en) | 2013-07-05 | 2020-08-04 | Basf Plant Science Company Gmbh | Gene expression or activity enhancing elements |
| WO2015001505A2 (en) | 2013-07-05 | 2015-01-08 | Basf Plant Science Company Gmbh | Gene expression or activity enhancing elements |
| EP3795686A2 (de) | 2013-07-05 | 2021-03-24 | Basf Plant Science Company GmbH | Genexpressions- oder aktivitätsverstärkende elemente |
| US10760089B2 (en) | 2014-11-14 | 2020-09-01 | Basf Plant Science Company Gmbh | Materials and methods for increasing the tocopherol content in seed oil |
| US11033593B2 (en) | 2014-11-14 | 2021-06-15 | Basf Plant Science Company Gmbh | Brassica events LBFLFK and LBFDAU and methods for detection thereof |
| US11813302B2 (en) | 2014-11-14 | 2023-11-14 | Basf Plant Science Company Gmbh | Brassica events LBFLFK and LBFDAU and methods for detection thereof |
| WO2016075327A2 (en) | 2014-11-14 | 2016-05-19 | Basf Plant Science Company Gmbh | Production of pufas in plants |
| US10829775B2 (en) | 2014-11-14 | 2020-11-10 | Basf Plant Science Company Gmbh | Materials and methods for increasing the tocopherol content in seed oil |
| US11771728B2 (en) | 2014-11-14 | 2023-10-03 | Basf Plant Science Company Gmbh | Materials and methods for increasing the tocopherol content in seed oil |
| US11613761B1 (en) | 2014-11-14 | 2023-03-28 | Bioriginal Food & Science Corporation | Materials and methods for PUFA production, and PUFA-containing compositions |
| US11484560B2 (en) | 2014-11-14 | 2022-11-01 | Basf Plant Science Company Gmbh | Stabilising fatty acid compositions |
| US11260095B2 (en) | 2014-11-14 | 2022-03-01 | Basf Plant Science Company Gmbh | Modification of plant lipids containing PUFAs |
| WO2020049159A1 (en) | 2018-09-07 | 2020-03-12 | Basf Plant Science Company Gmbh | Improved method for the production of high levels of pufa in plants |
| WO2020049157A1 (en) | 2018-09-07 | 2020-03-12 | Basf Plant Science Company Gmbh | Improved method for the production of high levels of pufa in plants |
| WO2020049155A1 (en) | 2018-09-07 | 2020-03-12 | Basf Plant Science Company Gmbh | Improved method for the production of high levels of pufa in plants |
| WO2020168277A1 (en) | 2019-02-14 | 2020-08-20 | Cargill, Incorporated | Brassica plants producing elevated levels of polyunsaturated fatty acids |
| WO2022098631A1 (en) | 2020-11-04 | 2022-05-12 | Cargill, Incorporated | Harvest management |
| WO2022204454A1 (en) | 2021-03-25 | 2022-09-29 | Cargill, Incorporated | Fertilizer management |
Also Published As
| Publication number | Publication date |
|---|---|
| CN108753857A (zh) | 2018-11-06 |
| AU2006226338B9 (en) | 2011-09-22 |
| EP1866417B1 (de) | 2017-04-26 |
| CA2600286C (en) | 2017-11-28 |
| NO20074434L (no) | 2007-11-27 |
| AU2006226338B2 (en) | 2011-05-12 |
| US20090158462A1 (en) | 2009-06-18 |
| AU2006226338A1 (en) | 2006-09-28 |
| WO2006100241A3 (de) | 2006-12-07 |
| ES2635013T3 (es) | 2017-10-02 |
| EP1866417A2 (de) | 2007-12-19 |
| DK1866417T3 (en) | 2017-08-21 |
| DE102005013779A1 (de) | 2006-09-28 |
| PL1866417T3 (pl) | 2017-09-29 |
| US8049064B2 (en) | 2011-11-01 |
| CN101146911A (zh) | 2008-03-19 |
| CA2600286A1 (en) | 2006-09-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1866417B1 (de) | Verfahren zur herstellung von mehrfach ungesättigten c20- und c22-fettsäuren mit mindestens vier doppelbindungen in transgenen pflanzen | |
| EP1769074B1 (de) | Verfahren zur erhöhung des gehalts an mehrfach ungesättigten langkettigen fettsäuren in transgenen organismen | |
| EP2623584B1 (de) | Verfahren zur Herstellung mehrfach ungesättigter Fettsäuren in transgenen Pflanzen | |
| EP1723220B1 (de) | Verfahren zur herstellung mehrfach ungesättigter fettsäuren in transgenen pflanzen | |
| EP1991684B1 (de) | Verfahren zur herstellung von mehrfach ungesättigten fettsäuren | |
| EP2169053B1 (de) | Verfahren zur Herstellung mehrfach ungesättigter Fettsäuren in transgenen Organismen | |
| EP1915451B1 (de) | Verfahren zur herstellung von arachidonsäure und/oder eicosapentaensäure in transgenen nutzpflanzen | |
| EP1720988A2 (de) | Verfahren zur herstellung von ungesättigten omega-3-fettsäuren in transgenen organismen | |
| EP1831377A1 (de) | Verfahren zur herstellung mehrfach ungesättigter fettsäuren in transgenen organismen | |
| EP2176433A2 (de) | Desaturasen und verfahren zur herstellung mehrfach ungesättigter fettsäuren in transgenen organismen | |
| DE102006008030A1 (de) | Verfahren zur Herstellung von mehrfach ungesättigten Fettsäuren | |
| DE102004062543A1 (de) | Verfahren zur Herstellung mehrfach ungesättigter Fettsäuren in transgenen Pflanzen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| REEP | Request for entry into the european phase |
Ref document number: 2006725200 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006725200 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2600286 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11886857 Country of ref document: US Ref document number: 2006226338 Country of ref document: AU Ref document number: 200680009188.X Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2006226338 Country of ref document: AU Date of ref document: 20060321 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2006226338 Country of ref document: AU |
|
| NENP | Non-entry into the national phase |
Ref country code: RU |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: RU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2006725200 Country of ref document: EP |