WO2005075585A1 - Verwendung strahlenhärtbarer harze auf basis hydrierter keton- und phenol-aldehydharze - Google Patents
Verwendung strahlenhärtbarer harze auf basis hydrierter keton- und phenol-aldehydharze Download PDFInfo
- Publication number
- WO2005075585A1 WO2005075585A1 PCT/EP2004/053316 EP2004053316W WO2005075585A1 WO 2005075585 A1 WO2005075585 A1 WO 2005075585A1 EP 2004053316 W EP2004053316 W EP 2004053316W WO 2005075585 A1 WO2005075585 A1 WO 2005075585A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- radiation
- component
- curable resins
- aldehyde
- resins according
- Prior art date
Links
- 0 CCC(CCCC(C)C(*)O)C(*)N Chemical compound CCC(CCCC(C)C(*)O)C(*)N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L61/00—Compositions of condensation polymers of aldehydes or ketones; Compositions of derivatives of such polymers
- C08L61/02—Condensation polymers of aldehydes or ketones only
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F299/00—Macromolecular compounds obtained by interreacting polymers involving only carbon-to-carbon unsaturated bond reactions, in the absence of non-macromolecular monomers
- C08F299/02—Macromolecular compounds obtained by interreacting polymers involving only carbon-to-carbon unsaturated bond reactions, in the absence of non-macromolecular monomers from unsaturated polycondensates
- C08F299/022—Macromolecular compounds obtained by interreacting polymers involving only carbon-to-carbon unsaturated bond reactions, in the absence of non-macromolecular monomers from unsaturated polycondensates from polycondensates with side or terminal unsaturations
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G12/00—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen
- C08G12/02—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes
- C08G12/04—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds
- C08G12/10—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds with acyclic compounds having the moiety X=C(—N<)2 in which X is O, S or —N
- C08G12/12—Ureas; Thioureas
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/54—Polycondensates of aldehydes
- C08G18/548—Polycondensates of aldehydes with ketones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/81—Unsaturated isocyanates or isothiocyanates
- C08G18/8141—Unsaturated isocyanates or isothiocyanates masked
- C08G18/815—Polyisocyanates or polyisothiocyanates masked with unsaturated compounds having active hydrogen
- C08G18/8158—Polyisocyanates or polyisothiocyanates masked with unsaturated compounds having active hydrogen with unsaturated compounds having only one group containing active hydrogen
- C08G18/8175—Polyisocyanates or polyisothiocyanates masked with unsaturated compounds having active hydrogen with unsaturated compounds having only one group containing active hydrogen with esters of acrylic or alkylacrylic acid having only one group containing active hydrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G6/00—Condensation polymers of aldehydes or ketones only
- C08G6/02—Condensation polymers of aldehydes or ketones only of aldehydes with ketones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L61/00—Compositions of condensation polymers of aldehydes or ketones; Compositions of derivatives of such polymers
- C08L61/20—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen
- C08L61/22—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds
- C08L61/24—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds with urea or thiourea
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/02—Printing inks
- C09D11/10—Printing inks based on artificial resins
- C09D11/101—Inks specially adapted for printing processes involving curing by wave energy or particle radiation, e.g. with UV-curing following the printing
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D161/00—Coating compositions based on condensation polymers of aldehydes or ketones; Coating compositions based on derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D161/00—Coating compositions based on condensation polymers of aldehydes or ketones; Coating compositions based on derivatives of such polymers
- C09D161/02—Condensation polymers of aldehydes or ketones only
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D161/00—Coating compositions based on condensation polymers of aldehydes or ketones; Coating compositions based on derivatives of such polymers
- C09D161/20—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen
- C09D161/22—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds
- C09D161/24—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds with urea or thiourea
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D175/00—Coating compositions based on polyureas or polyurethanes; Coating compositions based on derivatives of such polymers
- C09D175/04—Polyurethanes
- C09D175/14—Polyurethanes having carbon-to-carbon unsaturated bonds
- C09D175/16—Polyurethanes having carbon-to-carbon unsaturated bonds having terminal carbon-to-carbon unsaturated bonds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/34—Filling pastes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J161/00—Adhesives based on condensation polymers of aldehydes or ketones; Adhesives based on derivatives of such polymers
- C09J161/02—Condensation polymers of aldehydes or ketones only
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J161/00—Adhesives based on condensation polymers of aldehydes or ketones; Adhesives based on derivatives of such polymers
- C09J161/20—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen
- C09J161/22—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds
- C09J161/24—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds with urea or thiourea
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/02—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2666/00—Composition of polymers characterized by a further compound in the blend, being organic macromolecular compounds, natural resins, waxes or and bituminous materials, non-macromolecular organic substances, inorganic substances or characterized by their function in the composition
- C08L2666/02—Organic macromolecular compounds, natural resins, waxes or and bituminous materials
- C08L2666/14—Macromolecular compounds according to C08L59/00 - C08L87/00; Derivatives thereof
- C08L2666/16—Addition or condensation polymers of aldehydes or ketones according to C08L59/00 - C08L61/00; Derivatives thereof
Definitions
- the invention relates to the use of radiation-curable resins based on carbonyl-hydrogenated ketone-aldehyde and core-hydrogenated phenol-aldehyde resins.
- VOC volatile organic compounds
- the film-forming components in the coating material are relatively low molecular weight and therefore low-viscosity, so that high proportions of organic solvents can be dispensed with.
- Durable coatings are obtained by applying a high molecular polymer network through e.g. B. electron beams or UV light initiated crosslinking reactions.
- Ketone-aldehyde resins are used in coating materials such. As used as an additive resins to certain properties such as initial drying, gloss, hardness or scratch resistance to improve '. Because of their relatively low molecular weight, conventional ketone-aldehyde resins have a low melt and solution viscosity and are therefore used in coating materials, inter alia, as film-forming functional fillers.
- ketone-aldehyde resins have hydroxyl groups and can therefore only be used with e.g. B. polyisocyanates or amine resins. These crosslinking reactions are usually initiated or accelerated thermally.
- the ketone-aldehyde resins are not suitable for radiation-initiated crosslinking reactions based on cationic and / or radical reaction mechanisms.
- the ketone-aldehyde resins are usually used in radiation-curable coating systems such.
- B. as film-forming, passive, ie not cross-linking Additional component used.
- Such coatings often have a low resistance to z.
- EP 0 902 065 describes the use of non-radiation-curable resins made from urea (derivatives), ketones or aldehydes as an additional component in a mixture with radiation-curable resins.
- No. 4,070,500 describes the use of non-radiation-curable ketone-formaldehyde resins as film-forming components in radiation-curable inks.
- this object was achieved by using carbonyl-hydrogenated ketone-aldehyde resins and / or core-hydrogenated phenolic resins with ethylenically unsaturated groups as the main component, base component or additional component in radiation-curing coating materials, adhesives, printing inks and inks, polishes, glazes, pigment pastes and masterbatches, fillers, sealing compounds and insulating materials and / or cosmetic articles were used.
- the use of the radiation-curable resins according to the invention based on carbonyl-hydrogenated ketone-aldehyde and core-hydrogenated phenol-aldehyde resins as the main component, base component or additional component in radiation-curing coating materials, adhesives, printing inks and inks, polishes, glazes, pigment pastes and masterbatches, fillers, cosmetic articles and / or sealants - and Insulating materials result in a higher gloss and a higher hardness as well as abrasion resistance, an improved chemical and solvent resistance with a very high saponification stability and a low tendency to yellowing.
- the adhesion to substrates such as. B. metals, plastics, wood, paper and glass and mineral substrates improved, whereby the protection of these substrates is increased, for. B. by increasing the corrosion resistance.
- the interlayer adhesion is also increased, so that the adhesion of further applied layers is increased.
- the pigment wetting is improved, as is the stabilization of the pigments. It is possible to achieve the same color loci and color strengths with a smaller amount of pigment if the products according to the invention are used. This is particularly interesting, not least for economic reasons, since both high-priced pigments and additive wetting and stabilizing agents can at least be reduced.
- the radiation-curable resins as the main component, base component or additional component in radiation-curing fillers, primers, fillers, basecoats, topcoats and clearcoats, in particular on metals, plastics, wood, paper, textiles and glass and mineral substrates.
- further oligomers and / or polymers selected from the group consisting of polyurethanes, polyesters, polyacrylates, polyolefins, natural resins, epoxy resins, silicone oils and resins, amine resins, fluorine-containing polymers and their derivatives may be contained alone or in combination.
- the amount of further oligomers and / or polymers can be between 98 and 5%.
- the radiation-curable resins can also contain auxiliaries and additives selected from inhibitors, organic solvents, which may contain unsaturated groups, surface-active substances, oxygen and / or radical scavengers, catalysts,
- Light stabilizers color brighteners, photoinitiators, photosensitizers, Thixotropic agents, skin preventing agents, defoamers, dyes, pigments, fillers and matting agents.
- the amount varies greatly depending on the area of application and the type of auxiliary and additive.
- the invention relates to the use of radiation-curable resins, essentially containing
- the radiation-curable resins based on carbonyl-hydrogenated ketone-aldehyde and core-hydrogenated phenol-aldehyde resins are described in more detail below.
- Suitable ketones for the preparation of the carbonyl-hydrogenated ketone-aldehyde resins are all ketones, in particular acetone, acetophenone, methyl ethyl ketone, tert-butyl methyl ketone, heptanone-2, pentanone-3, methyl isobutyl ketone, cyclopentanone, cyclododecanone, mixtures of 2.2, 4- and 2,4,4-trimethylcyclopentanone, cycloheptanone and cyclooctanone, cyclohexanone and all alkyl-substituted cyclohexanones with one or more alkyl radicals which have a total of 1 to 8 hydrocarbon atoms, individually or in a mixture.
- alkyl-substituted cyclohexanones examples include 4-tert-amylcyclohexanone, 2-sec-butylcyclohexanone, 2-tert-butylcyclohexanone, 4-tert-butylcyclohexanone, 2-methylcyclohexanone and 3,3,5-trimethylcyclohexanone.
- ketones mentioned in the literature as suitable for ketone resin syntheses generally all C-H-acidic ketones, can be used.
- Carbonyl-hydrogenated ketone-aldehyde resins based on the ketones acetophenone, cyclohexadone, 4-tert-butylcyclohexanone, 3,3,5-trimethylcyclohexanone and heptanone alone or in a mixture are preferred.
- aldehyde component of the carbonyl-hydrogenated ketone-aldehyde resins are in principle undisclosed or branched aldehydes, such as. B. formaldehyde, acetaldehyde, n-butyraldehyde and / or isobutyraldehyde, valerianaldehyde and dodecanal.
- aldehydes mentioned as suitable in the literature for ketone resin syntheses can be used.
- formaldehyde is preferably used alone or in mixtures.
- the required formaldehyde is usually used as an approx. 20 to 40% by weight aqueous or alcoholic (e.g. methanol or butanol) solution.
- aqueous or alcoholic e.g. methanol or butanol
- Other forms of use of formaldehyde such as. B. the use of para-formaldehyde or trioxane are also possible.
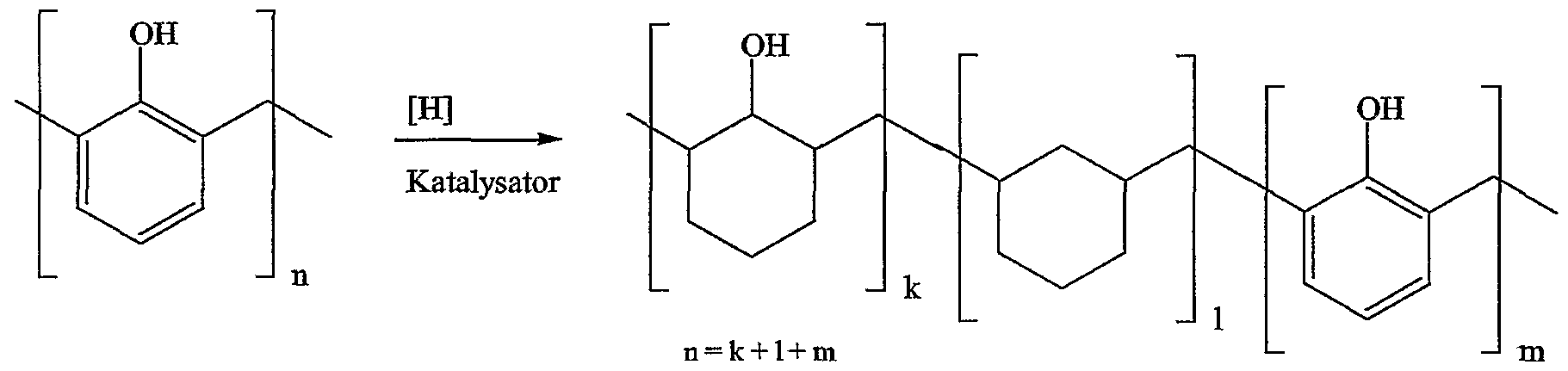
- the ketone and aldehyde resins are hydrogenated in the presence of a catalyst with hydrogen at pressures of up to 300 bar.
- the carbonyl group of the ketone-aldehyde resin is converted into a secondary hydroxy group.
- part of the hydroxyl groups can be split off, so that methylene groups result.
- the following scheme serves as an illustration:
- Nuclear hydrogenated phenol-aldehyde resins of the novolak type are used as component B) using aldehydes such as, for. B. formaldehyde, butyraldehyde or benzaldehyde, preferably formaldehyde used.
- aldehydes such as, for. B. formaldehyde, butyraldehyde or benzaldehyde, preferably formaldehyde used.
- non-hydrogenated novolaks can be used, but then they have lower light fastness.
- Core-hydrogenated resins based on alkyl-substituted phenols are particularly suitable as component B).
- component B Core-hydrogenated resins based on alkyl-substituted phenols.
- all phenols mentioned as suitable in the literature for phenolic resin syntheses can be used.
- Suitable phenols are phenol, 2- and 4-tert-butylphenol, 4-amylphenol, nonylphenol, 2- and 4-tert-octylphenol, dodecylphenol, cresol, xylenols and bisphenols. They can be used alone or in a mixture.
- Nuclear-hydrogenated, alkyl-substituted phenol-formaldehyde resins of the novolak type are very particularly preferably used.
- Preferred phenolic resins are reaction products of formaldehyde and 2- and 4-tert-butylphenol, 4-amylphenol, nonylphenol, 2- and 4-tert-octylphenol as well as dodecylphenol.
- the novolaks are hydrogenated in the presence of a suitable catalyst using hydrogen.
- the choice of catalyst transforms the aromatic ring into a cycloaliphatic one.
- the hydroxyl group is retained by suitable selection of the parameters.
- the hydroxy groups can also be hydrogenated by the choice of hydrogenation conditions, so that cycloaliphatic rings are formed.
- the core-hydrogenated resins have OH numbers of 50 to 450 mg KOH / g, preferably 100 to 350 mg KOH / g, particularly preferably 150 to 300 mg KOH / g.
- the proportion of aromatic groups is below 50% by weight, preferably below 30% by weight, particularly preferably below 10% by weight.
- the radiation-curable resins on which the invention is based are obtained by polymer-analogous reaction of the carbonyl-hydrogenated ketone-aldehyde resins and / or the core-hydrogenated phenolic resins in the melt or in solution of a suitable solvent with component C).
- suitable as component C) are maleic anhydride, (meth) acrylic acid derivatives such as. B. (meth) acryloyl chloride, glycidyl (meth) acrylate, (meth) acrylic acid and / or their low molecular weight alkyl esters and / or anhydrides alone or in a mixture.
- radiation-curable resins can be obtained by reacting the carbonyl-hydrogenated ketone-aldehyde resins and the core-hydrogenated phenolic resins Isocyanates that have an ethylenically unsaturated group, such as.
- B. (Meth) ac ⁇ yloyl isocyanate, ⁇ , ⁇ -dimethyl-3-isopropenylbenzyl isocyanate, (meth) acrylic alkyl isocyanate with splkylspacern, which have one to 12, preferably 2 to 8, particularly preferably 2 to 6 carbon atoms, such as.
- reaction products of hydroxyalkyl (meth) acrylates the alkyl spacers of which have from 1 to 12, preferably 2 to 8, particularly preferably 2 to 6 coblene atoms, and diisocyanates such as, for. B.
- cyclohexane diisocyanate methylcyclohexane, Ethylcyclohexandiisocyanat, Propylcyclohexandiisocyanat, Methyldiethylcyclohexan- diisocyanate, phenylene diisocyanate, toluene diisocyanate, bis (isocyanatophenyl) methane, Propandiisocyanat, butane diisocyanate, pentane diisocyanate, hexane diisocyanate, such as hexamethylene diisocyanate (HDI) or l, 5-diisocyanato-2-methylpentane (MPDI) , Heptane diisocyanate, octane diisocyanate, nonane diisocyanate such as 1,6-diisocyanato-2,4,4-trimethylhexane or 1,6-diisocyanato-2,2,4
- polyisocyanates are the compounds with more than two isocyanate groups per molecule produced by trimerization, allophanatization, biuretization and or urethanization of the simple diisocyanates, for example the reaction products of these simple diisocyanates, such as IPDI, HDI and / or HMDI with polyhydric alcohols (e.g. glycerol, trimethylolpropane, pentaerythritol) or polyvalent polyamines or the triisocyanurates which can be obtained by trimerizing the simple diisocyanates such as IPDI, HDI and HMDI.
- polyhydric alcohols e.g. glycerol, trimethylolpropane, pentaerythritol
- polyvalent polyamines e.g. glycerol, trimethylolpropane, pentaerythritol
- triisocyanurates which can be obtained by trimerizing the simple diisocyanates such as IPD
- a suitable catalyst for the preparation of the invention Resins are used. Suitable are all compounds known in the literature which accelerate an OH-NCO reaction, such as. B. diazäbicyclooctane (DABCO) or dibutyltin dilaurate (DBTL).
- DABCO diazäbicyclooctane
- DBTL dibutyltin dilaurate
- a hard resin such as the hydrogenated acetophenone-formaldehyde resin with ⁇ , ⁇ -dimethyl-3-isopropenylbenzyl isocyanate
- products of higher hardness are obtained than by using (meth) acrylic ethyl isocyanate and / or hydroxyethyl acrylate-isophorone diisocyanate adducts; however, the flexibility is then less.
- the other hy ⁇ -oxy-functional polymers generally have molecular weights Mn between 200 and 10,000 g / mol, preferably between 300 and 5,000 g / mol.
- the resins on which the invention is based are produced in the melt or in solution of a suitable organic solvent of the carbonyl-hydrogenated ketone-aldehyde resin and / or core-hydrogenated phenol-aldehyde resin.
- the organic solvent may also have unsaturated groups and then acts directly as a reactive thinner in later use.
- I to the solution or melt of the carbonyl-hydrogenated ketone-aldehyde resin A) and / or core-hydrogenated phenol-aldehyde resin B) is the compound which on the one hand has at least one ethylenically unsaturated group and at the same time at least one which is reactive towards A) and B) Has grouping, optionally in the presence of a suitable catalyst.
- the temperature of the reaction is selected depending on the reactivity of component C). When isocyanates are used as component C), temperatures between 30 and 150 ° C., preferably between 50 and 140 ° C., have proven successful.
- the solvent present can be separated off after the reaction has ended, in which case a powder of the product according to the invention is generally obtained.
- a preferred embodiment II is to the solution or melt of the carbonyl-hydrogenated ketone-aldehyde resin A) and / or core-hydrogenated phenol-aldehyde resin B) and the hydroxy-functional polymer, such as. B. polyether, polyester and / or polyacrylate, the compound which has at least one ethylenically unsaturated group and at the same time at least one group reactive towards A) and B) and the additional polymer, optionally in the presence of a suitable catalyst.
- the carbonyl-hydrogenated ketone-aldehyde resin A) and / or core-hydrogenated phenol-aldehyde resin B) and the hydroxy-functional polymer such as. B. polyether, polyester and / or polyacrylate, the compound which has at least one ethylenically unsaturated group and at the same time at least one group reactive towards A) and B) and the additional polymer, optionally in the presence of a suitable catalyst.
- the temperature of the reaction is selected depending on the reactivity of component C).
- temperatures have between 30 and 150 ° C, preferably between 50 and 140 ° C proven.
- the solvent contained can be separated off after the reaction has ended, in which case a powder of the product according to the invention is generally obtained. 5 It has proven advantageous to use 1 mol of component A) and / or component B) and / or additional polymers - based on M n - with 0.5 to 15 mol, preferably 1 to 10 mol, particularly 2 to 8 mol to bring the unsaturated compound (component C) to the reaction.
- the solution or melt of the carbonyl-hydrogenated ketone-aldehyde resin A) and / or the core-hydrogenated phenol-aldehyde resin B) and the hydroxy-functional polymer such as, for. B. polyether, polyester and / or polyacrylate a di- and / or trifunctional isocyanate and a hydroxy-functional pre-adduct. Only then is the compound 5, which has at least one ethylenically unsaturated group and at the same time at least one group reactive towards A) and B) and the additional polymer, optionally in the presence of a suitable catalyst.
- the temperature of the reaction is selected depending on the reactivity of component C). When using isocyanates as component C), temperatures between 30 and 150 ° C., preferably between 50 and 140 ° C., have proven successful.
- the solvent contained can be separated off after the reaction has ended, in which case a powder of the product according to the invention is generally obtained.5 It has proven advantageous to use 1 mol of component A) and / or component B) and / or additional polymers - based on M n - with 0.5 to 15 mol, preferably 1 to 10 mol, particularly 2 to 8 mol of the unsaturated compound (component C) to react. 0 In the presence of suitable photoinitiators, if appropriate in the presence of suitable photosensitizers, these resins can be converted by irradiation into polymeric, insoluble networks which, depending on the content of ethylenically unsaturated groups, up to Thermosets result.
- the dynamic viscosity of the formulations decreases with an increasing proportion of the products according to the invention.
- the mixtures were provided with Darocure 1173 (for quantity, see table) and applied to metal sheets using a doctor blade.
- the systems contain solvents; therefore, pre-drying was carried out at 80 ° C for 30 minutes.
- the films were then cured using UV light (mercury medium-pressure lamp, 70 W / optical filter 350 nm) (time in table).
- the physical admixture of the unsubstituted resins improves the hardness, adhesion, Peugeot and MEK tests. However, mechanical properties, as can be determined by impact test and Erichsen deepening, are worsened.
- the hardness and the adhesion are increased by chemical crosslinking of the products according to the invention with the clear lacquer.
- D Super gasoline resistance (Peugeot test) and solvent resistance (MEK test) are also improved. Mechanical properties that have deteriorated with the purely physical admixtures are also improved, which is expressed in good values in the impact test and Erichsen deepening.
- the tendency to yellowing is improved compared to the standard system, especially when exposed to high temperatures.
- MOP acetate methoxypropylacetate nfA .: non-volatile components
- Ferrari test resistance to super gasoline
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002555357A CA2555357A1 (en) | 2004-02-03 | 2004-12-07 | Use of radiation-curable resins based on hydrogenated ketone-aldehyde and phenolaldehyde resins |
| EP04804712A EP1711567A1 (de) | 2004-02-03 | 2004-12-07 | Verwendung strahlenhärtbarer harze auf basis hydrierter keton- und phenol-aldehydharze |
| JP2006551738A JP2007519816A (ja) | 2004-02-03 | 2004-12-07 | 水素化されたケトン−及びフェノール−アルデヒド樹脂系の放射線硬化性樹脂の使用 |
| BRPI0418495-5A BRPI0418495A (pt) | 2004-02-03 | 2004-12-07 | uso de resinas endurecìveis por radiação à base de resinas de cetona- e fenol-aldeìdo hidrogenadas |
| US10/588,193 US20070123661A1 (en) | 2004-02-03 | 2004-12-07 | Utilization of radiohardenable resins based on hydrogenated ketone and phenol aldehyde resins |
| TNP2006000240A TNSN06240A1 (en) | 2004-02-03 | 2006-08-02 | Utilization of radiohardenable resins based on hydrogenated ketone and phenol aldehyde resins |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102004005208A DE102004005208A1 (de) | 2004-02-03 | 2004-02-03 | Verwendung strahlenhärtbarer Harze auf Basis hydrierter Keton- und Phenol-Aldehydharze |
| DE102004005208.5 | 2004-02-03 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2005075585A1 true WO2005075585A1 (de) | 2005-08-18 |
Family
ID=34745218
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2004/053316 WO2005075585A1 (de) | 2004-02-03 | 2004-12-07 | Verwendung strahlenhärtbarer harze auf basis hydrierter keton- und phenol-aldehydharze |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US20070123661A1 (de) |
| EP (1) | EP1711567A1 (de) |
| JP (1) | JP2007519816A (de) |
| KR (1) | KR20060130643A (de) |
| CN (1) | CN1875076A (de) |
| BR (1) | BRPI0418495A (de) |
| CA (1) | CA2555357A1 (de) |
| DE (1) | DE102004005208A1 (de) |
| RU (1) | RU2006131447A (de) |
| TN (1) | TNSN06240A1 (de) |
| WO (1) | WO2005075585A1 (de) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006021479A1 (de) * | 2004-08-26 | 2006-03-02 | Degussa Gmbh | Strahlungsempfindliche masse |
| WO2007098813A1 (de) * | 2006-02-28 | 2007-09-07 | Evonik Degussa Gmbh | Formaldehydfreie, carbonylhydrierte keton-aldehydharze auf basis von formaldehyd und ein verfahren zu ihrer herstellung |
| WO2009047102A1 (de) * | 2007-10-05 | 2009-04-16 | Evonik Degussa Gmbh | Beschichtungsstoffzusammensetzungen |
| CN101343341B (zh) * | 2008-08-12 | 2010-06-23 | 无锡市虎皇漆业有限公司 | 丙烯酸改性聚氨酯水分散体的制备方法 |
| JP2010525088A (ja) * | 2007-04-20 | 2010-07-22 | エヴォニク ゴールドシュミット ゲーエムベーハー | ポリエーテルを含む分散および乳化樹脂 |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE10326893A1 (de) * | 2003-06-14 | 2004-12-30 | Degussa Ag | Harze auf Basis von Ketonen und Aldehyde mit verbesserten Löslichkeitseigenschaften und geringen Farbzahlen |
| DE102004049544A1 (de) * | 2004-10-12 | 2006-04-13 | Degussa Ag | Strahlungshärtbar modifizierte, ungesättigte, amorphe Polyester |
| DE102005002388A1 (de) * | 2005-01-19 | 2006-07-27 | Degussa Ag | Wässrige, strahlungshärtbar modifizierte, ungesättigte, amorphe Polyester |
| DE102005013329A1 (de) * | 2005-03-23 | 2006-11-16 | Degussa Ag | Niedrigviskose uretdiongruppenhaltige Polyadditionsverbindungen, Verfahren zur Herstellung und Verwendung |
| DE102005017200A1 (de) | 2005-04-13 | 2006-10-19 | Degussa Ag | Verwendung eines hochviskosen, weitgehend amorphen Polyolefins zur Herstellung einer Folie |
| DE102005026765A1 (de) * | 2005-06-10 | 2006-12-14 | Degussa Ag | Rückenfixierung von Kunstrasenrohware mit Schmelzklebern auf Basis von amorphen Poly-alpha-Olefinen und/oder modifizierten, amorphen Poly-alpha-Olefinen |
| DE102006000646A1 (de) * | 2006-01-03 | 2007-07-12 | Degussa Gmbh | Zusammensetzung zur Herstellung von Universalpigmentpräparationen |
| DE102006000645A1 (de) * | 2006-01-03 | 2007-07-12 | Degussa Gmbh | Universalpigmentpräparationen |
| DE102006026760A1 (de) * | 2006-06-09 | 2008-01-10 | Evonik Degussa Gmbh | Formaldehydfreie, OH-funktionelle, carbonyl- und kernhydrierte Keton-Aldehydharze auf Basis von Alkylarylketonen und Formaldehyd und ein Verfahren zu ihrer Herstellung |
| DE102006026758A1 (de) * | 2006-06-09 | 2008-01-10 | Evonik Degussa Gmbh | Formaldehydfreie, carbonyl- und kernhydrierte Keton-Aldehydharze auf Basis von Alkylarylketonen und Formaldehyd mit geringer OH-Funktionalität und ein Verfahren zu ihrer Herstellung |
| DE102007063698B4 (de) * | 2007-09-13 | 2010-10-28 | Carl Zeiss Vision Gmbh | Optisches Bauelement mit Oberflächenbeschichtung |
| DE102007045944A1 (de) | 2007-09-25 | 2009-04-09 | Evonik Degussa Gmbh | Verfahren zur lösemittelfreien, kontinuierlichen Herstellung von zinnfreien, strahlungshärtbaren Urethanacrylaten auf Basis von carbonylhydrierten Keton-Aldehydharzen und/oder hydrierten Keton und/oder carbonylhydrierten und kernhydrierten Keton-Aldehydharzen auf Basis von aromatischen Ketonen, die hergestellten Produkte und deren Verwendung |
| DE102007047585A1 (de) * | 2007-10-05 | 2009-04-09 | Evonik Degussa Gmbh | Kugelschreiberpastenzusammensetzungen |
| DE102007047584A1 (de) * | 2007-10-05 | 2009-04-09 | Evonik Degussa Gmbh | Druckfarbenzusammensetzungen |
| CN102260448A (zh) * | 2010-05-30 | 2011-11-30 | 湘潭瑞鑫电气科技有限责任公司 | 表面绝缘漆 |
| CN102504073B (zh) * | 2011-11-11 | 2015-02-11 | 乐凯华光印刷科技有限公司 | 一种氨酯化改性杂化酚醛树脂及其制备方法 |
| IN2014DN10757A (de) * | 2012-05-29 | 2015-09-04 | Mitsubishi Gas Chemical Co | |
| CN104559656A (zh) * | 2014-12-23 | 2015-04-29 | 王新民 | 一种烘干清漆 |
| CN110003411B (zh) * | 2019-04-03 | 2021-05-07 | 北京化工大学 | 缩聚大分子光引发剂的制备方法及制备得到的光引发剂 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0111686A1 (de) * | 1982-11-11 | 1984-06-27 | Hüls Aktiengesellschaft | Benzinlösliche, hydrierte Alkylarylketon/Formaldehyd-Harze sowie deren Herstellung |
| EP0133451A1 (de) * | 1983-07-06 | 1985-02-27 | Hüls Aktiengesellschaft | Kondensationsharze auf Basis von Alkylarylketonen und Formaldehyd |
| WO1999055785A1 (de) * | 1998-04-27 | 1999-11-04 | Herberts Gmbh & Co. Kg | Strahlungshärtbare beschichtungsmittel und deren verwendung |
| WO2003061849A1 (de) * | 2002-01-24 | 2003-07-31 | Basf Coatings Ag | Gehärtete materialien, verfahren zu ihrer herstellung und ihre verwendung |
| EP1508582A1 (de) * | 2003-08-22 | 2005-02-23 | Degussa AG | Strahlenhärtbare Harze auf Basis hydrierter Keton- und Phenol-Aldehydharze und ein Verfahren zu ihrer Herstellung |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE19643703A1 (de) * | 1996-10-23 | 1998-04-30 | Huels Chemische Werke Ag | Hydrophile verseifungsstabil veresterte Kunstharze, besonders geeignet für wäßrige Systeme |
| DE19739620A1 (de) * | 1997-09-10 | 1999-03-11 | Basf Ag | Strahlungshärtbares Bindemittel für Druckfarben |
| DE50005383D1 (de) * | 1999-06-21 | 2004-04-01 | Surface Specialties Austria | Wasserverdünnbare Harze, Verfahren zu ihrer Herstellung und ihre Verwendung |
| DE19944373A1 (de) * | 1999-09-16 | 2001-03-22 | Degussa | Katalysator und Verfahren zur Herstellung von farbreduzierten isocyanuratgruppenhaltigen Polyisocyananten |
| DE10033099A1 (de) * | 2000-07-07 | 2002-01-17 | Degussa | Verfahren zur Herstellung von geruchsarmen und lagerstabilen monomerhaltigen Polyisocyanuraten aus Isophorondiisocyanat |
| DE10047762A1 (de) * | 2000-09-27 | 2002-04-11 | Degussa | Pulverförmige, wasserdispergierbare blockierte Polyisocyanataddukte, ein Verfahren zur Herstellung und ihre Verwendung |
| DE10065176A1 (de) * | 2000-12-23 | 2002-06-27 | Degussa | Katalysator und Verfahren zur Herstellung von niedrigviskosen und farbreduzierten isocyanuratgruppenhaltigen Polyisocyanaten |
| DE10134261A1 (de) * | 2001-07-18 | 2003-01-30 | Basf Ag | Stabilisatoren enthaltende UV-vernetzbare Schmelzhaftklebstoffe |
| DE10163783A1 (de) * | 2001-12-22 | 2003-07-03 | Degussa | Verfahren zur Herstellung epoxidierter Polyalkenylene und Verwendung von Phosphonsäuren und deren Derivaten als Katalysator |
| DE10212706A1 (de) * | 2002-03-21 | 2003-10-02 | Degussa | Ungesättigte, amorphe Polyester auf Basis bestimmter Dicidolisomerer |
| DE10242265A1 (de) * | 2002-09-12 | 2004-03-25 | Degussa Ag | Haftungsverbessernder Zusatz aus einem ungesättigten, amorphen Polyester |
| DE10258573A1 (de) * | 2002-12-14 | 2004-07-01 | Degussa Ag | Polymermodifizierte Harze |
| DE10258574A1 (de) * | 2002-12-14 | 2004-07-01 | Degussa Ag | Polymermodifizierte Harze |
| DE10261006A1 (de) * | 2002-12-24 | 2004-07-08 | Degussa Ag | Dispersionen amorpher, ungesättigter Polyesterharze auf Basis bestimmter Dicidolisomerer |
| DE10261005A1 (de) * | 2002-12-24 | 2004-07-08 | Degussa Ag | Dispersionen amorpher, urethanisierter ungesättigter Polyesterharze auf Basis bestimmter Dicidolisomerer |
| DE10322845A1 (de) * | 2003-05-19 | 2004-12-16 | Degussa Ag | Verzweigte,amorphe Makropolyole auf Polyesterbasis mit enger Molekulargewichtsverteilung |
| DE10326893A1 (de) * | 2003-06-14 | 2004-12-30 | Degussa Ag | Harze auf Basis von Ketonen und Aldehyde mit verbesserten Löslichkeitseigenschaften und geringen Farbzahlen |
| DE10338562A1 (de) * | 2003-08-22 | 2005-03-17 | Degussa Ag | Strahlenhärtbare Harze auf Basis von Keton- und/oder Harnstoff-Aldehydharzen und ein Verfahren zu ihrer Herstellung |
| DE10338559A1 (de) * | 2003-08-22 | 2005-04-14 | Degussa Ag | Verfahren zur Herstellung von Keton-Formaldehydharzen |
| DE10338561A1 (de) * | 2003-08-22 | 2005-04-14 | Degussa Ag | Keton-Aldehydharze, insbesondere Cyclohexanon-Formaldehydharze mit geringem Wassergehalt und hoher thermischer Bestätigkeit und Vergilbungsbeständigkeit sowie ein Verfahren zur Herstellung und Verwendung |
-
2004
- 2004-02-03 DE DE102004005208A patent/DE102004005208A1/de not_active Withdrawn
- 2004-12-07 KR KR1020067015706A patent/KR20060130643A/ko not_active Application Discontinuation
- 2004-12-07 JP JP2006551738A patent/JP2007519816A/ja not_active Withdrawn
- 2004-12-07 CN CNA2004800316969A patent/CN1875076A/zh active Pending
- 2004-12-07 WO PCT/EP2004/053316 patent/WO2005075585A1/de not_active Application Discontinuation
- 2004-12-07 BR BRPI0418495-5A patent/BRPI0418495A/pt not_active IP Right Cessation
- 2004-12-07 EP EP04804712A patent/EP1711567A1/de not_active Withdrawn
- 2004-12-07 RU RU2006131447/04A patent/RU2006131447A/ru unknown
- 2004-12-07 CA CA002555357A patent/CA2555357A1/en not_active Abandoned
- 2004-12-07 US US10/588,193 patent/US20070123661A1/en not_active Abandoned
-
2006
- 2006-08-02 TN TNP2006000240A patent/TNSN06240A1/en unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0111686A1 (de) * | 1982-11-11 | 1984-06-27 | Hüls Aktiengesellschaft | Benzinlösliche, hydrierte Alkylarylketon/Formaldehyd-Harze sowie deren Herstellung |
| EP0133451A1 (de) * | 1983-07-06 | 1985-02-27 | Hüls Aktiengesellschaft | Kondensationsharze auf Basis von Alkylarylketonen und Formaldehyd |
| WO1999055785A1 (de) * | 1998-04-27 | 1999-11-04 | Herberts Gmbh & Co. Kg | Strahlungshärtbare beschichtungsmittel und deren verwendung |
| WO2003061849A1 (de) * | 2002-01-24 | 2003-07-31 | Basf Coatings Ag | Gehärtete materialien, verfahren zu ihrer herstellung und ihre verwendung |
| EP1508582A1 (de) * | 2003-08-22 | 2005-02-23 | Degussa AG | Strahlenhärtbare Harze auf Basis hydrierter Keton- und Phenol-Aldehydharze und ein Verfahren zu ihrer Herstellung |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006021479A1 (de) * | 2004-08-26 | 2006-03-02 | Degussa Gmbh | Strahlungsempfindliche masse |
| WO2007098813A1 (de) * | 2006-02-28 | 2007-09-07 | Evonik Degussa Gmbh | Formaldehydfreie, carbonylhydrierte keton-aldehydharze auf basis von formaldehyd und ein verfahren zu ihrer herstellung |
| JP2009528407A (ja) * | 2006-02-28 | 2009-08-06 | エボニック デグサ ゲーエムベーハー | ホルムアルデヒド不含の、ホルムアルデヒドをベースとするカルボニル水素化されたケトン−アルデヒド樹脂およびその製造法 |
| JP2010525088A (ja) * | 2007-04-20 | 2010-07-22 | エヴォニク ゴールドシュミット ゲーエムベーハー | ポリエーテルを含む分散および乳化樹脂 |
| WO2009047102A1 (de) * | 2007-10-05 | 2009-04-16 | Evonik Degussa Gmbh | Beschichtungsstoffzusammensetzungen |
| CN101343341B (zh) * | 2008-08-12 | 2010-06-23 | 无锡市虎皇漆业有限公司 | 丙烯酸改性聚氨酯水分散体的制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| RU2006131447A (ru) | 2008-03-10 |
| US20070123661A1 (en) | 2007-05-31 |
| JP2007519816A (ja) | 2007-07-19 |
| KR20060130643A (ko) | 2006-12-19 |
| DE102004005208A1 (de) | 2005-08-11 |
| CA2555357A1 (en) | 2005-08-18 |
| TNSN06240A1 (en) | 2007-12-03 |
| CN1875076A (zh) | 2006-12-06 |
| BRPI0418495A (pt) | 2007-06-19 |
| EP1711567A1 (de) | 2006-10-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2005075585A1 (de) | Verwendung strahlenhärtbarer harze auf basis hydrierter keton- und phenol-aldehydharze | |
| EP1508582B1 (de) | Strahlenhärtbare Harze auf Basis hydrierter Keton- und Phenol-Aldehydharze und ein Verfahren zu ihrer Herstellung | |
| EP1740634B1 (de) | Polymerzusammensetzungen von carbonylhydrierten keton-aldehydharzen und polyisocyanaten in reaktiven lösemitteln | |
| EP1508581B1 (de) | Strahlenhärtbare Harze auf Basis von Keton- und/oder Harnstoff-Aldehydharzen und ein Verfahren zu ihrer Herstellung | |
| WO2006042769A1 (de) | Wässrige, strahlenhärtbare harze, ein verfahren zu ihrer herstellung und ihre verwendung | |
| EP1711566B1 (de) | Verwendung strahlenhärtbarer harze auf basis von keton- und/ oder harnstoff-aldehydharzen | |
| DE102005024246A1 (de) | Copolymere, ein Verfahren zu deren Herstellung und die Verwendung als Bindemittel | |
| EP1776402B1 (de) | Zinnfreie, hochschmelzende reaktionsprodukte aus carbonylhydrierten keton-aldehydharzen, hydrierten ketonharzen sowie carbonyl- und kernhydrierten keton-aldehydharzen auf basis von aromatischen ketonen und polyisocyanaten | |
| DE102005010967A1 (de) | Wässrige Lackzusammensetzungen für Holz | |
| WO2006094849A1 (de) | Wässrige beschichtungsstoffzusammensetzungen für effektlacke | |
| WO2006094847A1 (de) | Wässrige druckfarbenzusammensetzungen | |
| WO2009047102A1 (de) | Beschichtungsstoffzusammensetzungen | |
| WO2006094850A1 (de) | Wässrige beschichtungsstoffzusammensetzungen mit verbessertem steinschlagschutz | |
| WO2006094851A1 (de) | Wässrige beschichtungsstoffzusammensetzungen für flexible untergründe | |
| DE102004060285A1 (de) | Strahlenhärtbare Zusammensetzungen, ein Verfahren zu ihrer Herstellung und ihre Verwendung | |
| WO2007036488A1 (de) | Nicht schmelzende reaktionsprodukte mit einer hohen glasübergangstemperatur und geringer lösungsviskosität | |
| MXPA06008814A (en) | Utilization of radiohardenable resins based on hydrogenated ketone and phenol aldehyde resins |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200480031696.9 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2004804712 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007123661 Country of ref document: US Ref document number: 2555357 Country of ref document: CA Ref document number: 10588193 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006551738 Country of ref document: JP Ref document number: 1020067015706 Country of ref document: KR Ref document number: PA/a/2006/008814 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006131447 Country of ref document: RU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004804712 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020067015706 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 10588193 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0418495 Country of ref document: BR |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2004804712 Country of ref document: EP |