WO1999047120A1 - Ophthalmic solution comprising glycogen - Google Patents
Ophthalmic solution comprising glycogen Download PDFInfo
- Publication number
- WO1999047120A1 WO1999047120A1 PCT/EP1999/001624 EP9901624W WO9947120A1 WO 1999047120 A1 WO1999047120 A1 WO 1999047120A1 EP 9901624 W EP9901624 W EP 9901624W WO 9947120 A1 WO9947120 A1 WO 9947120A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- ophthalmic solution
- solution
- solution according
- glycogen
- determined
- Prior art date
Links
- 239000002997 ophthalmic solution Substances 0.000 title claims abstract description 33
- 229940054534 ophthalmic solution Drugs 0.000 title claims abstract description 33
- 229920002527 Glycogen Polymers 0.000 title claims abstract description 22
- 229940096919 glycogen Drugs 0.000 title claims abstract description 22
- 150000004676 glycans Chemical class 0.000 claims abstract description 22
- 229920001282 polysaccharide Polymers 0.000 claims abstract description 22
- 239000005017 polysaccharide Substances 0.000 claims abstract description 22
- 239000007864 aqueous solution Substances 0.000 claims abstract description 11
- XOAAWQZATWQOTB-UHFFFAOYSA-N taurine Chemical group NCCS(O)(=O)=O XOAAWQZATWQOTB-UHFFFAOYSA-N 0.000 claims description 16
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical group OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims description 14
- 235000010355 mannitol Nutrition 0.000 claims description 9
- 229960003080 taurine Drugs 0.000 claims description 8
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 claims description 6
- 239000003963 antioxidant agent Substances 0.000 claims description 5
- 229930195725 Mannitol Natural products 0.000 claims description 4
- 239000000594 mannitol Substances 0.000 claims description 4
- 230000003078 antioxidant effect Effects 0.000 claims description 3
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 2
- 235000010228 ethyl p-hydroxybenzoate Nutrition 0.000 claims description 2
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 claims description 2
- 239000003755 preservative agent Substances 0.000 claims description 2
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 claims description 2
- RTKIYNMVFMVABJ-UHFFFAOYSA-L thimerosal Chemical compound [Na+].CC[Hg]SC1=CC=CC=C1C([O-])=O RTKIYNMVFMVABJ-UHFFFAOYSA-L 0.000 claims description 2
- 229940033663 thimerosal Drugs 0.000 claims description 2
- 239000003795 chemical substances by application Substances 0.000 claims 2
- 230000001105 regulatory effect Effects 0.000 claims 2
- 230000002335 preservative effect Effects 0.000 claims 1
- 239000000243 solution Substances 0.000 description 42
- 230000035807 sensation Effects 0.000 description 16
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 13
- 229910001868 water Inorganic materials 0.000 description 13
- 210000004087 cornea Anatomy 0.000 description 11
- 239000000607 artificial tear Substances 0.000 description 9
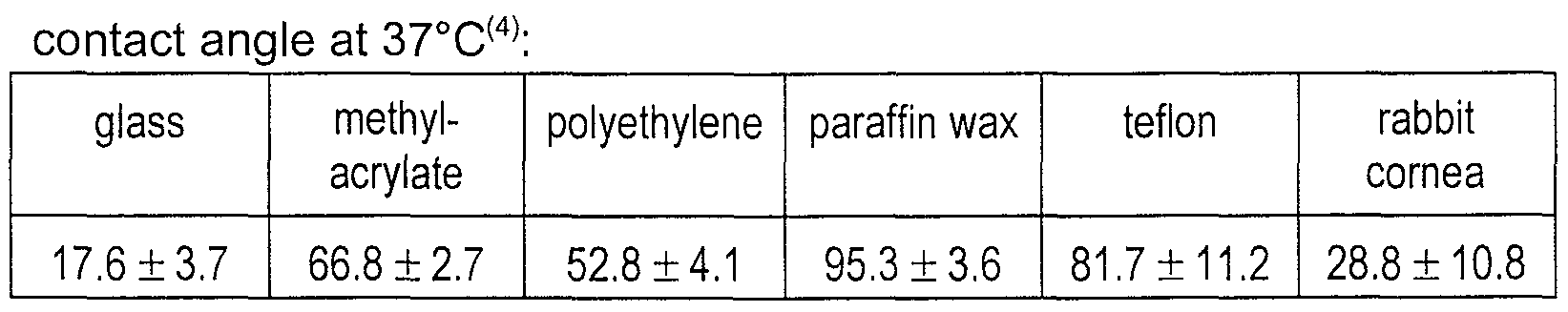
- 239000011521 glass Substances 0.000 description 9
- 238000000034 method Methods 0.000 description 9
- 238000002360 preparation method Methods 0.000 description 9
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 8
- 241000283973 Oryctolagus cuniculus Species 0.000 description 8
- 239000004698 Polyethylene Substances 0.000 description 8
- 239000004809 Teflon Substances 0.000 description 8
- 229920006362 Teflon® Polymers 0.000 description 8
- 239000012188 paraffin wax Substances 0.000 description 8
- -1 polyethylene Polymers 0.000 description 8
- 229920000573 polyethylene Polymers 0.000 description 8
- 239000012153 distilled water Substances 0.000 description 6
- 230000036512 infertility Effects 0.000 description 6
- 230000036642 wellbeing Effects 0.000 description 6
- 229920002307 Dextran Polymers 0.000 description 5
- 229940061607 dibasic sodium phosphate Drugs 0.000 description 5
- BNIILDVGGAEEIG-UHFFFAOYSA-L disodium hydrogen phosphate Chemical compound [Na+].[Na+].OP([O-])([O-])=O BNIILDVGGAEEIG-UHFFFAOYSA-L 0.000 description 5
- 239000012530 fluid Substances 0.000 description 5
- 229940045641 monobasic sodium phosphate Drugs 0.000 description 5
- 229910000403 monosodium phosphate Inorganic materials 0.000 description 5
- 235000019799 monosodium phosphate Nutrition 0.000 description 5
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 5
- 208000002193 Pain Diseases 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 239000003906 humectant Substances 0.000 description 4
- 235000006708 antioxidants Nutrition 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000000314 lubricant Substances 0.000 description 3
- 230000003278 mimic effect Effects 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 239000008363 phosphate buffer Substances 0.000 description 3
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 206010047513 Vision blurred Diseases 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 210000000795 conjunctiva Anatomy 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- SATHPVQTSSUFFW-UHFFFAOYSA-N 4-[6-[(3,5-dihydroxy-4-methoxyoxan-2-yl)oxymethyl]-3,5-dihydroxy-4-methoxyoxan-2-yl]oxy-2-(hydroxymethyl)-6-methyloxane-3,5-diol Chemical compound OC1C(OC)C(O)COC1OCC1C(O)C(OC)C(O)C(OC2C(C(CO)OC(C)C2O)O)O1 SATHPVQTSSUFFW-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 229920000189 Arabinogalactan Polymers 0.000 description 1
- 239000001904 Arabinogalactan Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- 208000003556 Dry Eye Syndromes Diseases 0.000 description 1
- 206010013774 Dry eye Diseases 0.000 description 1
- 239000004150 EU approved colour Substances 0.000 description 1
- 241000287826 Gallus Species 0.000 description 1
- 238000007696 Kjeldahl method Methods 0.000 description 1
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 1
- 241000237524 Mytilus Species 0.000 description 1
- 241000237536 Mytilus edulis Species 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 230000000172 allergic effect Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 235000019312 arabinogalactan Nutrition 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 208000010668 atopic eczema Diseases 0.000 description 1
- JXLHNMVSKXFWAO-UHFFFAOYSA-N azane;7-fluoro-2,1,3-benzoxadiazole-4-sulfonic acid Chemical compound N.OS(=O)(=O)C1=CC=C(F)C2=NON=C12 JXLHNMVSKXFWAO-UHFFFAOYSA-N 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 230000002016 colloidosmotic effect Effects 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 229960002433 cysteine Drugs 0.000 description 1
- 238000009509 drug development Methods 0.000 description 1
- 210000000744 eyelid Anatomy 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 206010023332 keratitis Diseases 0.000 description 1
- 201000010666 keratoconjunctivitis Diseases 0.000 description 1
- 206010023365 keratopathy Diseases 0.000 description 1
- 230000001050 lubricating effect Effects 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 229940006093 opthalmologic coloring agent diagnostic Drugs 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 206010039083 rhinitis Diseases 0.000 description 1
- 235000002639 sodium chloride Nutrition 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008174 sterile solution Substances 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/04—Artificial tears; Irrigation solutions
Definitions
- This invention relates to a humectant and lubricant solution for ophthalmic use based on a glycogen polysaccharide. It is known that the instillation of humectant and lubricant solutions into the eye is indicated in some circumstances. These solutions are all the more useful if they mimic the functions of natural tear fluid.
- Artificial tears are used, for example, to alleviate the symptoms of dry keratoconjunctivitis, exposure-induced keratopathies and other situations resulting in dry eye syndrome. In addition to this artificial tears are useful to wearers of contact lenses, particularly in the case of hard lenses.
- US patent 4 039 662 proposes that this disadvantage should be overcome through a low viscosity ophthalmic solution containing dextran or arabinogalactan together with benzylalkonium chloride.
- the abovementioned patent specifies that the polysaccharide component alone without benzylalkonium chloride is incapable of remaining adsorbed onto the cornea for a sufficiently long period (column 3, lines 9-13).
- the polysaccharide particles described in the aforesaid patent combine in solution with the benzylalkonium chloride by electrostatic attraction. This seems to form a complex having an electron charge which causes the macromolecule to be adsorbed onto the surface of the cornea (column 3, lines 19-37).
- glycogen polysaccharides provide an ophthalmic solution of low viscosity and low oncotic pressure and exert a pleasing refreshing, lubricating and humectant effect on the cornea even in the absence of benzylalkonium chloride (Table I).
- an ophthalmic solution characterized in that it comprises a sterile aqueous solution of a glycogen polysaccharide.
- glycogen polysaccharide is that described in patent EP-B-0 654 048.
- aqueous solutions of glycogen polysaccharides can be filtered to 0.2 microns, and can thus provide sterile solutions up to a concentration of 12% (w/v). In addition to this, these solutions have a low viscosity and a low oncotic pressure.
- the quantity of glycogen polysaccharide in the ophthalmic solution according to this invention is of from 0J to 12% (w/v). Preferably it is of from 1 to 6% (w/v). Even more preferably it is of from 2 to 4% (w/v).
- the ophthalmic solution according to this invention has a viscosity of between 1 and 9 cP. Preferably it has a viscosity between 2 and 7 cP. Even more preferably, it has a viscosity between 2 and 6 cP.
- the ophthalmic solution according to this invention has an oncotic pressure of less than 5 mmHg. Preferably it has an oncotic pressure of less than 3 mmHg.
- the ophthalmic solution according to this invention may also contain other conventional ingredients such as: antioxidants, buffers, compounds to render the solution isotonic with tear fluid, stabilising agents, colouring agents and the like.
- antioxidants are cysteine, ascorbic acid and taurine. The latter is also particularly preferred on account of its anti- free radical properties. 4 -
- the quantity of antioxidant in the ophthalmic solution according to this invention will vary widely according to the preselected composition.
- the quantity is preferably of from OJ to 0.6% (w/v) and, even more preferably, from 0.2 to 0.4% (w/v).
- Typical examples of buffers are borate, bicarbonate, acetate and phosphate buffers, and their quantities will be selected so as to regulate the pH of the ophthalmic solution according to this invention between 5 and 8.
- the buffer will be a phosphate buffer and the pH of the ophthalmic solution according to the invention will be adjusted between 6.5 and 7.5.
- Typical examples of compounds suitable for ensuring that the ophthalmic solution according to this invention is isotonic with natural tear fluid are glycerine, sodium chloride and mannitol.
- the quantities of these in the ophthalmic solution according to this invention will vary widely according to the preselected composition.
- the quantity is preferably of from 0.5 to 3% (w/v). Even more preferably it is of from 1.5% to 2% (w/v).
- composition according to this invention may also contain one or more preservatives selected from the group comprising benzylalkonium chloride, thimerosal, methyl parabenes, ethyl parabenes, propyl parabenes and butyl parabenes.
- glycogen polysaccharide used in the examples below was extracted from Mytilus edulis or Mytilus gallus provincialis using the method described in EP-B-0 654 048 and had the following characteristics:
- the abovementioned ophthalmic solution was prepared by dissolving the abovementioned components in the prescribed quantity of water at 6 -
- the solution prepared in this way was subdivided into 0.4 ml doses in single-use phials having a capacity of 1 ml.
- the solution prepared in this way had the following properties: pH 7.18 osmolarity (5) 272 density (6) (g/ml) at 25°C 1.024 density (6) (g/ml) at 37°C 1.016 viscosity' 7 ' (cP) 5 oncotic pressure (8) (mmHg) 1.7 sterility sterile mean surface tension (3) (m/Ntn) at 25°C 61.80 at 37°C 61.90
- the solution prepared in this way had the following characteristics: sterility sterile oncotic pressure (8) (mmHg) 1.7 mean surface tension (3) m/Nm at 25°C 61.43 at 37°C 60.02
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Ophthalmology & Optometry (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Inorganic Chemistry (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
Description
Claims
Priority Applications (14)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU34115/99A AU754288B2 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
| SI9930607T SI1069885T1 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
| JP2000536360A JP4485054B2 (en) | 1998-03-19 | 1999-03-11 | Glycogen-containing ophthalmic solution |
| IL13818599A IL138185A0 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
| EA200000963A EA003139B1 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
| HU0101177A HU228114B1 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
| SK1370-2000A SK283602B6 (en) | 1998-03-19 | 1999-03-11 | An ophthalmic solution comprising a sterile aqueous solution of a glycogen |
| CA002323810A CA2323810C (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
| AT99915585T ATE267580T1 (en) | 1998-03-19 | 1999-03-11 | OPHTHALMOLOGICAL SOLUTION CONTAINING GLYCOGEN |
| UA2000105872A UA61133C2 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution containing glycogen |
| EP99915585A EP1069885B1 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
| DE69917623T DE69917623T2 (en) | 1998-03-19 | 1999-03-11 | OPHTHALMOLOGICAL SOLUTION CONTAINING GLYCOGEN |
| US09/623,502 US6486139B1 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
| HK01103568A HK1032913A1 (en) | 1998-03-19 | 2001-05-23 | Ophthalmic solution comprising glycogen. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IT98MI000555A IT1298758B1 (en) | 1998-03-19 | 1998-03-19 | WETTING AND LUBRICANT SOLUTION FOR OPHTHALMIC USE |
| ITMI98A000555 | 1998-03-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1999047120A1 true WO1999047120A1 (en) | 1999-09-23 |
Family
ID=11379374
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP1999/001624 WO1999047120A1 (en) | 1998-03-19 | 1999-03-11 | Ophthalmic solution comprising glycogen |
Country Status (26)
| Country | Link |
|---|---|
| US (1) | US6486139B1 (en) |
| EP (1) | EP1069885B1 (en) |
| JP (1) | JP4485054B2 (en) |
| KR (1) | KR100579500B1 (en) |
| CN (1) | CN1146404C (en) |
| AR (1) | AR015543A1 (en) |
| AT (1) | ATE267580T1 (en) |
| AU (1) | AU754288B2 (en) |
| BG (1) | BG64740B1 (en) |
| CA (1) | CA2323810C (en) |
| CZ (1) | CZ298855B6 (en) |
| DE (1) | DE69917623T2 (en) |
| DK (1) | DK1069885T3 (en) |
| EA (1) | EA003139B1 (en) |
| ES (1) | ES2220056T3 (en) |
| GE (1) | GEP20032966B (en) |
| HK (1) | HK1032913A1 (en) |
| HU (1) | HU228114B1 (en) |
| IL (1) | IL138185A0 (en) |
| IT (1) | IT1298758B1 (en) |
| PT (1) | PT1069885E (en) |
| SK (1) | SK283602B6 (en) |
| TR (1) | TR200002687T2 (en) |
| UA (1) | UA61133C2 (en) |
| WO (1) | WO1999047120A1 (en) |
| ZA (1) | ZA992192B (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010084038A1 (en) | 2009-01-23 | 2010-07-29 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Controlled release pharmaceutical or food formulation and process for its preparation |
| WO2011015509A1 (en) | 2009-08-03 | 2011-02-10 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Food formulation comprising glycogen |
| US8840923B2 (en) | 2007-12-28 | 2014-09-23 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Slow-release pharmaceutical formulation and process for its preparation |
| US20160083484A1 (en) * | 2013-04-26 | 2016-03-24 | Mirexus Biotechnologies Inc. | Monodisperse glycogen and phytoglycogen nanoparticles and use thereof as additives in cosmetics, pharmaceuticals, and food products |
| WO2017191041A1 (en) * | 2016-05-05 | 2017-11-09 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Ophthalmic composition comprising a synergistic combination of glycogen and hyaluronic acid or a salt thereof |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002154989A (en) * | 2000-11-14 | 2002-05-28 | Lion Corp | Ophthalmic composition and composition having improved retention of medicine in biological mucosa |
| US20090270345A1 (en) | 2008-04-26 | 2009-10-29 | Alcon Research, Ltd. | Polymeric artificial tear system |
| RU2581025C1 (en) * | 2015-02-09 | 2016-04-10 | Государственное бюджетное образовательное учреждение высшего профессионального образования "Башкирский государственный медицинский университет" Министерства здравоохранения Российской Федерации | Eye medicinal film with moxifloxacin |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4039662A (en) * | 1975-12-04 | 1977-08-02 | Alcon Laboratories, Inc. | Ophthalmic solution |
| JPS5344614A (en) * | 1976-10-04 | 1978-04-21 | Ebios Pharma | Antitumor agent |
| JPS55160719A (en) * | 1979-06-01 | 1980-12-13 | Sankyo Co Ltd | Remedy for microbial infectious disease |
| EP0267015A2 (en) * | 1986-11-05 | 1988-05-11 | Ethicon, Inc. | Stabilized compositions containing epidermal growth factor |
| JPH02111726A (en) * | 1988-10-19 | 1990-04-24 | Dainippon Pharmaceut Co Ltd | Stabilization of human interleukin 1 |
| WO1994014417A1 (en) * | 1992-12-29 | 1994-07-07 | Insite Vision Incorporated | Plasticized bioerodible controlled delivery system |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4039962A (en) * | 1974-08-01 | 1977-08-02 | John Leonard Hughes | System for amplifying laser beams |
| JPS62178505A (en) * | 1986-02-03 | 1987-08-05 | Maruzen Kasei Kk | Cosmetic |
| GB2189800B (en) * | 1986-04-07 | 1990-03-14 | Michael Anthony West | Marking of articles |
| JPS63188365A (en) * | 1987-01-29 | 1988-08-03 | Ajinomoto Co Inc | Novel edible cellulose |
| JPS63290809A (en) * | 1987-05-25 | 1988-11-28 | Noebia:Kk | Cosmetic for lip |
| JPH0611704B2 (en) * | 1989-01-19 | 1994-02-16 | 参天製薬株式会社 | Eye drops for corneal disease treatment |
| JP2749080B2 (en) * | 1988-11-09 | 1998-05-13 | 株式会社日立製作所 | Information recording materials |
| JP3050898B2 (en) * | 1990-07-06 | 2000-06-12 | 千寿製薬株式会社 | Aqueous pharmaceutical preparation |
| ATE154513T1 (en) * | 1992-07-08 | 1997-07-15 | Leiras Oy | EYE MEDICINAL CONTAINING TIMOLOL HEMIHYDRATE |
| IT1255488B (en) * | 1992-08-04 | 1995-11-06 | Angelini Francesco Ist Ricerca | GLYCOGEN POLYSACCHARIDES |
| JP3919240B2 (en) * | 1995-01-19 | 2007-05-23 | 日新製鋼株式会社 | Stainless steel lubricant for hot rolling with excellent nozzle clogging resistance |
| JPH08325143A (en) * | 1995-05-31 | 1996-12-10 | Taisho Pharmaceut Co Ltd | Medicine for curing damage of corneal parenchyma |
| JPH09143064A (en) * | 1995-11-27 | 1997-06-03 | Taisho Pharmaceut Co Ltd | Ophthalmic solution for improving eye fatigue |
| AU703540B2 (en) * | 1996-03-26 | 1999-03-25 | Meddiss, Incorporated | Methods for inducing analgesia or anesthesia and treating or preventing ischemic injury of tissues in general |
| US5673788A (en) * | 1996-07-01 | 1997-10-07 | Mclane; Kevin J. | Collapsible holder |
| JPH1072376A (en) * | 1996-09-02 | 1998-03-17 | Ofutekusu:Kk | Eye drop aqueous solution comprising hyaluronic acid |
-
1998
- 1998-03-19 IT IT98MI000555A patent/IT1298758B1/en active IP Right Grant
-
1999
- 1999-03-11 US US09/623,502 patent/US6486139B1/en not_active Expired - Lifetime
- 1999-03-11 ES ES99915585T patent/ES2220056T3/en not_active Expired - Lifetime
- 1999-03-11 CZ CZ20003407A patent/CZ298855B6/en not_active IP Right Cessation
- 1999-03-11 DK DK99915585T patent/DK1069885T3/en active
- 1999-03-11 JP JP2000536360A patent/JP4485054B2/en not_active Expired - Fee Related
- 1999-03-11 PT PT99915585T patent/PT1069885E/en unknown
- 1999-03-11 UA UA2000105872A patent/UA61133C2/en unknown
- 1999-03-11 IL IL13818599A patent/IL138185A0/en not_active IP Right Cessation
- 1999-03-11 HU HU0101177A patent/HU228114B1/en not_active IP Right Cessation
- 1999-03-11 SK SK1370-2000A patent/SK283602B6/en not_active IP Right Cessation
- 1999-03-11 TR TR2000/02687T patent/TR200002687T2/en unknown
- 1999-03-11 AT AT99915585T patent/ATE267580T1/en active
- 1999-03-11 CA CA002323810A patent/CA2323810C/en not_active Expired - Fee Related
- 1999-03-11 AU AU34115/99A patent/AU754288B2/en not_active Ceased
- 1999-03-11 EA EA200000963A patent/EA003139B1/en not_active IP Right Cessation
- 1999-03-11 CN CNB998041866A patent/CN1146404C/en not_active Expired - Fee Related
- 1999-03-11 KR KR1020007010370A patent/KR100579500B1/en not_active IP Right Cessation
- 1999-03-11 WO PCT/EP1999/001624 patent/WO1999047120A1/en active IP Right Grant
- 1999-03-11 DE DE69917623T patent/DE69917623T2/en not_active Expired - Lifetime
- 1999-03-11 GE GEAP19995600A patent/GEP20032966B/en unknown
- 1999-03-11 EP EP99915585A patent/EP1069885B1/en not_active Expired - Lifetime
- 1999-03-17 AR ARP990101149A patent/AR015543A1/en active IP Right Grant
- 1999-03-18 ZA ZA9902192A patent/ZA992192B/en unknown
-
2000
- 2000-10-04 BG BG104825A patent/BG64740B1/en unknown
-
2001
- 2001-05-23 HK HK01103568A patent/HK1032913A1/en not_active IP Right Cessation
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4039662A (en) * | 1975-12-04 | 1977-08-02 | Alcon Laboratories, Inc. | Ophthalmic solution |
| JPS5344614A (en) * | 1976-10-04 | 1978-04-21 | Ebios Pharma | Antitumor agent |
| JPS55160719A (en) * | 1979-06-01 | 1980-12-13 | Sankyo Co Ltd | Remedy for microbial infectious disease |
| EP0267015A2 (en) * | 1986-11-05 | 1988-05-11 | Ethicon, Inc. | Stabilized compositions containing epidermal growth factor |
| JPH02111726A (en) * | 1988-10-19 | 1990-04-24 | Dainippon Pharmaceut Co Ltd | Stabilization of human interleukin 1 |
| WO1994014417A1 (en) * | 1992-12-29 | 1994-07-07 | Insite Vision Incorporated | Plasticized bioerodible controlled delivery system |
Non-Patent Citations (3)
| Title |
|---|
| DATABASE WPI Section Ch Week 7822, Derwent World Patents Index; Class B04, AN 78-39519A, XP002108975 * |
| DATABASE WPI Section Ch Week 8108, Derwent World Patents Index; Class B04, AN 81-12557D, XP002108974 * |
| DATABASE WPI Section Ch Week 9022, Derwent World Patents Index; Class A96, AN 90-169086, XP002108973 * |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8840923B2 (en) | 2007-12-28 | 2014-09-23 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Slow-release pharmaceutical formulation and process for its preparation |
| WO2010084038A1 (en) | 2009-01-23 | 2010-07-29 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Controlled release pharmaceutical or food formulation and process for its preparation |
| US9125823B2 (en) | 2009-01-23 | 2015-09-08 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Controlled release pharmaceutical or food formulation and process for its preparation |
| WO2011015509A1 (en) | 2009-08-03 | 2011-02-10 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Food formulation comprising glycogen |
| US8889654B2 (en) | 2009-08-03 | 2014-11-18 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Food formulation comprising glycogen |
| US20160083484A1 (en) * | 2013-04-26 | 2016-03-24 | Mirexus Biotechnologies Inc. | Monodisperse glycogen and phytoglycogen nanoparticles and use thereof as additives in cosmetics, pharmaceuticals, and food products |
| US10172946B2 (en) * | 2013-04-26 | 2019-01-08 | Mirexus Biotechnologies Inc. | Monodisperse glycogen and phytoglycogen nanoparticles and use thereof as additives in cosmetics, pharmaceuticals, and food products |
| WO2017191041A1 (en) * | 2016-05-05 | 2017-11-09 | Aziende Chimiche Riunite Angelini Francesco A.C.R.A.F. S.P.A. | Ophthalmic composition comprising a synergistic combination of glycogen and hyaluronic acid or a salt thereof |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2022116007A (en) | Artificial tear liquid, contact lens and medicine carrier composition, and use of the same | |
| RU2545675C2 (en) | Ophthalmological composition, based on polyunsaturated fatty acids omega-3 and omega-6 | |
| US6107347A (en) | Bimodal molecular weight hyaluronate formulations and methods for using same | |
| JP2010065055A (en) | Contact lens loading or preserving agent and agar-containing ophthalmic solution | |
| US6486139B1 (en) | Ophthalmic solution comprising glycogen | |
| JPH08510731A (en) | In situ produced gel for therapeutic use | |
| JP4736540B2 (en) | Ophthalmic thickener | |
| CA3112278A1 (en) | Artificial tear, contact lens and drug vehicle compositions and methods of use thereof | |
| JP2003342197A (en) | Thermosensitive gelling preparation containing hyaluronic acid | |
| MXPA00009197A (en) | Ophthalmic solution comprising glycogen | |
| JP4263418B2 (en) | Thermal gelation artificial tears | |
| WO2016162894A1 (en) | Ophthalmic compositions based on polyunsaturated fatty acids and triacylglycerols | |
| US11779593B2 (en) | Agent for improving ocular subjective symptoms and method thereof | |
| WO2022113970A1 (en) | Therapeutic agent for short tear breakup time-type dry eye and eye drops containing same | |
| KR20080077585A (en) | Ophthalmic composition for preventing and treating ocular diseases | |
| JP2024072717A (en) | Ophthalmic Composition | |
| JP2011084575A (en) | Eye-drop |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 99804186.6 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG US UZ VN YU ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW SD SL SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 138185 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1999915585 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2323810 Country of ref document: CA Ref document number: 2323810 Country of ref document: CA Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13702000 Country of ref document: SK |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PV2000-3407 Country of ref document: CZ Ref document number: 2000/02687 Country of ref document: TR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2000/009197 Country of ref document: MX Ref document number: 1020007010370 Country of ref document: KR Ref document number: 09623502 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 34115/99 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200000963 Country of ref document: EA |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 1999915585 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: PV2000-3407 Country of ref document: CZ |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020007010370 Country of ref document: KR |
|
| NENP | Non-entry into the national phase |
Ref country code: CA |
|
| WWG | Wipo information: grant in national office |
Ref document number: 34115/99 Country of ref document: AU |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1999915585 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1020007010370 Country of ref document: KR |
|
| WWG | Wipo information: grant in national office |
Ref document number: PV2000-3407 Country of ref document: CZ |