WO1998054970A1 - Fungizide mischungen - Google Patents
Fungizide mischungen Download PDFInfo
- Publication number
- WO1998054970A1 WO1998054970A1 PCT/EP1998/003282 EP9803282W WO9854970A1 WO 1998054970 A1 WO1998054970 A1 WO 1998054970A1 EP 9803282 W EP9803282 W EP 9803282W WO 9854970 A1 WO9854970 A1 WO 9854970A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- iii
- compound
- formula
- alkylamino
- alkyl
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/24—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof containing the groups, or; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/44—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing at least one carboxylic group or a thio analogue, or a derivative thereof, and a nitrogen atom attached to the same carbon skeleton by a single or double bond, this nitrogen atom not being a member of a derivative or of a thio analogue of a carboxylic group, e.g. amino-carboxylic acids
- A01N37/50—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing at least one carboxylic group or a thio analogue, or a derivative thereof, and a nitrogen atom attached to the same carbon skeleton by a single or double bond, this nitrogen atom not being a member of a derivative or of a thio analogue of a carboxylic group, e.g. amino-carboxylic acids the nitrogen atom being doubly bound to the carbon skeleton
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/84—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms six-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,4
Definitions
- the present invention relates to a fungicidal mixture which
- X is CH and N
- n is 0, 1 or 2
- R is halogen, -CC 4 -alkyl and -C-C 4 -haloalkyl, where the radicals R may be different if n is 2, or
- U is CHOCH 3 or N0CH 3
- R a is cyano, halogen, C 1 -C 4 -alkyl or C ⁇ -C 4 -haloalkyl
- x is 1, 2 or 3, where the radicals R a can be different, when x is 2 or 3 and Y is one of the radicals 0CH 2 or 0- (4, 6-pyrimidinyl) -O,
- R 'phenyl which can carry one to three of the following substituents: cyano, halogen, C 1 -C 4 -alkyl,
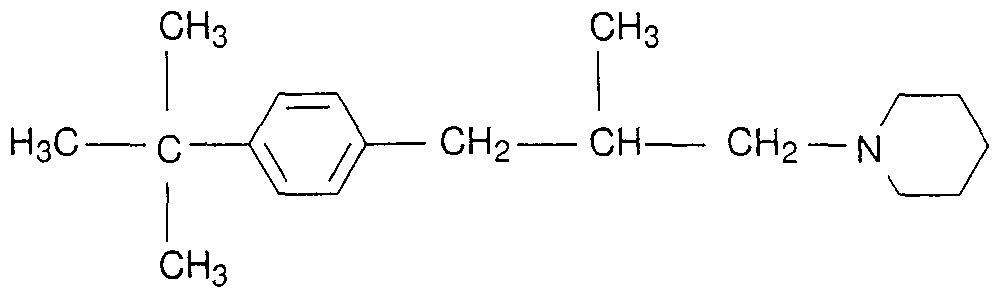
- N-C ⁇ -C4 alkylamino where the phenyl groups may be partially or fully halogenated and / or may carry one to three of the following radicals: cyano, C 4 -alkyl, C 4 haloalkyl, C ⁇ -C 4 -Alkoxy, -CC 4 -haloalkoxy and -CC 4 -alkylthio,
- the invention relates to methods for controlling harmful fungi with mixtures of the compounds I (I.a and I.b), II (II. A to II. C) and III (III.1 and III.2).
- strobilurin fungicides III are described in the literature as active ingredients for combating harmful fungi: Oxime ether ester III.1: EP-A 253 213, EP-A 254 426 and EP-A 382 375;
- EP-A 645 087 discloses synergistic mixtures of the compound I.b with active ingredients II. A to II. C.
- the present invention was based on mixtures which, with a reduced total amount of active compounds applied, have an improved activity against harmful fungi (synergistic mixtures).
- Formula I.a in particular represents carbamates in which the combination of the substituents corresponds to one row of Table 1 below:
- Compounds Ia are particularly preferred. 12, Ia. 23 Ia. 32 and Ia. 38.
- Formula III.1 represents in particular methyl phenylacetate, in which the combination of the substituents corresponds to one row of Table 2 below:
- the compounds III-l. L and III-1.6 are particularly preferred.
- Formula III.2 represents in particular benzyloxybisoximes in which the combination of the substituents corresponds to one row of Table 3 below:
- the compounds I, II and III are able to form salts or adducts with inorganic or organic acids or with metal ions.
- inorganic acids are hydrohalic acids such as hydrogen fluoride, hydrogen chloride, hydrogen bromide and iodine-hydrogen, sulfuric acid, phosphoric acid and nitric acid.
- organic acids are formic acid, carbonic acid and alkanoic acids such as acetic acid, trifluoroacetic acid, trichloroacetic acid and propionic acid as well as glycolic acid, thiocyanic acid, lactic acid, succinic acid, citric acid, benzoic acid, cinnamic acid, oxalic acid, alkyl sulfonic acids (sulfonic acids with branched or branched chain radicals) 20 carbon atoms), arylsulfonic acids or disulfonic acids (aromatic radicals such as phenyl and naphthyl which carry one or two sulfonic acid groups), alkylphosphonic acids (phosphonic acids with straight-chain or branched alkyl radicals with 1 to 20 carbon atoms),
- the ions of the elements of the first to eighth subgroups especially chromium, manganese, iron, cobalt, nickel, copper, zinc and, in addition, of the second main group, especially calcium and magnesium, of the third and fourth main group, come as metal ions Aluminum, tin and lead are considered.
- the metals can, if appropriate, be present in various valencies to which they are assigned.
- the mixtures of the compounds I and II or the compounds I and II applied simultaneously, together or separately, are distinguished by an excellent action against a broad spectrum of phytopathogenic fungi, in particular from the class of the Ascomycetes, Basidiomycetes, Phycomycetes and Deuteromycetes. They are partly systemically effective and can therefore also be used as leaf and soil fungicides.
- Sugar cane Venturia inaequalis (scab) on apples, Helminthos - porium species on cereals, Septoria nodorum on wheat, Botrytis cinera (gray mold) on strawberries, vegetables, ornamental plants and vines, Cercospora arachidicola on peanuts, Pseudocercosporella herpotrichoides on wheat and barley oryzae on rice, Phytophthora infestans on potatoes and tomatoes, Plasmopara viticola on vines, Pseudoperonospora species in hops and cucumbers, Alternaria species on vegetables and fruits, Mycosphaerella species in bananas and Fusarium and Verticillium species.
- the compounds I and II can be applied simultaneously, that is jointly or separately, or in succession, the sequence in the case of separate application generally not having any effect on the success of the control measures.
- the compounds I and II are usually in one
- the compounds I and III are usually used in a weight ratio of 10: 1 to 0.01: 1, preferably 5: 1 to 0.05: 1, in particular 1: 1 to 0.05: 1.
- the application rates of the mixtures according to the invention, especially for agricultural crop areas, are from 0.01 to 8 kg / ha, preferably 0.1 to 5 kg / ha, in particular 0.2 to 3.0 kg / ha, depending on the type of effect desired .
- the application rates for the compounds I are 0.01 to 2.5 kg / ha, preferably 0.05 to 2.5 kg / ha, in particular 0.1 to 1.0 kg / ha.
- the application rates for the compounds II are accordingly from 0.01 to 10 kg / ha, preferably 0.05 to 5 kg / ha, in particular 0.05 to 2.0 kg / ha.
- the application rates for the compounds III are accordingly from 0.01 to 10 kg / ha, preferably 0.05 to 5 kg / ha, in particular 0.05 to 2.0 kg / ha.
- application rates of mixture of 0.001 to 250 g / kg of seed preferably 0.01 to 100 g / kg, in particular 0.01 to 50 g / kg, are generally used.
- the compounds I and II or the mixtures of the compounds I and II are applied separately or jointly by spraying or dusting the seeds, the plants or the soil before or after the plants are sown or before or after emergence of the plants.
- the fungicidal synergistic mixtures according to the invention or the compounds I and II can be used, for example, in the form of directly sprayable solutions, powders and suspensions or in the form of high-strength aqueous, oily or other suspensions, dispersions, emulsions, oil dispersions, pastes, dusts, spreading agents or Granules are processed and applied by spraying, atomizing, dusting, scattering or pouring.
- the form of application depends on the intended use; in any case, it should ensure that the mixture according to the invention is as fine and uniform as possible.
- the formulations are prepared in a manner known per se, e.g. by adding solvents and / or carrier substances.
- Inert additives such as emulsifiers or dispersants are usually added to the formulations.
- alkali, alkaline earth, ammonium salts of aromatic sulfonic acids e.g. Lignin, phenol, naphthalene and dibutylnaphthalenesulfonic acid, as well as of fatty acids, alkyl and alkylarylsulfonates, alkyl, lauryl ether and fatty alcohol sulfates, as well as salts of sulfated hexa-, hepta- and octadecanols or fatty alcohol glycol ethers, condensation products of sulfonated naphthalene and its derivatives - aldehyde, condensation products of naphthalene or
- Naphthalenesulfonic acids with phenol and formaldehyde polyoxyethylene octylphenol ether, ethoxylated isooctyl, octyl or nonylphenol, alkylphenol or tributylphenyl polyglycol ether, alkylaryl polyether alcohols, isotridecyl alcohol, fatty alcohol - ethylene oxide condensates, ethoxylated polyalkylene glycol, ethoxylated polyalkylene glycol oil ether acetate, sorbitol ester, lignin sulfite liquor or methyl cellulose into consideration.
- Powders, dusts and dusts can be prepared by mixing or jointly grinding the compounds I or II or the mixture of the compounds I and II with a solid carrier.
- Granules e.g. coating, impregnation or homogeneous granules
- a solid carrier e.g., a wax, a wax, or a wax.
- Mineral soils such as silica gel, silicas, silica gels, silicates, talc, kaolin, limestone, lime, chalk, boluses, loess, clay, dolomite, diatomaceous earth, calcium and magnesium sulfate, magnesium oxide, ground plastics and fertilizers are used as fillers or solid carriers such as ammonium sulfate, ammonium phosphate, ammonium nitrate, ureas and vegetable products such as cereal flour, tree bark, wood and nutshell flour, cellulose powder or other solid carriers.
- mineral soils such as silica gel, silicas, silica gels, silicates, talc, kaolin, limestone, lime, chalk, boluses, loess, clay, dolomite, diatomaceous earth, calcium and magnesium sulfate, magnesium oxide, ground plastics and fertilizers are used as fillers or solid carriers such as ammonium sulfate, ammonium phosphate, ammonium
- the formulations generally contain 0.1 to 95% by weight, preferably 0.5 to 90% by weight, of one of the compounds I or II or of the mixture of the compounds I and II.
- the active ingredients are in a purity of 90% to 100%, preferably 95% to 100% (according to the NMR or HPLC spectrum) is used.
- the compounds I or II, the mixtures or the corresponding formulations are used in such a way that the harmful fungi, their habitat or the plants, seeds, soils, areas, materials or spaces to be kept free by them are mixed with a fungicidally effective amount of the mixture, or the compounds I and II treated separately.
- the application can take place before or after the infestation by the harmful fungi.
- aqueous active ingredient preparation consisting of a stock solution consisting of 10% active ingredient, 63% cyclo hexanone and 27% emulsifier was added, sprayed to runoff and dusted with spores of powdery mildew (Erysiphe graminis forma specialis tritici) 24 hours after the spray coating had dried on.
- the test plants were then set up in a greenhouse at temperatures between 20 and 24 ° C. and 60 to 90% relative atmospheric humidity. After 7 days, the extent of mildew development was determined visually in% infestation of the entire leaf area.
- the evaluation was carried out by determining the infected
- ß corresponds to the fungal attack on the untreated (control) plants in%
- the infection of the treated plants corresponds to that of the untreated control plants; at an efficiency of 100, the treated plants showed no infection.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002291784A CA2291784A1 (en) | 1997-06-04 | 1998-06-02 | Fungicidal mixtures |
| JP50148399A JP2002508756A (ja) | 1997-06-04 | 1998-06-02 | 殺菌剤混合物 |
| EA199901068A EA002404B1 (ru) | 1997-06-04 | 1998-06-02 | Фунгицидная смесь |
| AU98115/98A AU9811598A (en) | 1997-06-04 | 1998-06-02 | Fungicidal mixtures |
| HU0003909A HUP0003909A3 (en) | 1997-06-04 | 1998-06-02 | Synergetic fungicidal mixture and its use |
| EP98967167A EP1009235A1 (de) | 1997-06-04 | 1998-06-02 | Fungizide mischungen |
| BR9810421-7A BR9810421A (pt) | 1997-06-04 | 1998-06-02 | Mistura fungicida, e, processo para controlar fungos daninhos |
| SK1635-99A SK163599A3 (en) | 1997-06-04 | 1998-06-02 | Fungicidal mixtures |
| IL13293298A IL132932A0 (en) | 1997-06-04 | 1998-06-02 | Fungicidal mixtures |
| KR19997011337A KR20010013341A (ko) | 1997-06-04 | 1998-06-02 | 살진균성 혼합물 |
| PL98337157A PL337157A1 (en) | 1997-06-04 | 1998-06-02 | Fungicidal mixtures |
| US09/424,918 US6303599B1 (en) | 1997-06-04 | 1998-06-02 | Fungicidal mixtures |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19723289.2 | 1997-06-04 | ||
| DE19723289 | 1997-06-04 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1998054970A1 true WO1998054970A1 (de) | 1998-12-10 |
Family
ID=7831293
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP1998/003282 WO1998054970A1 (de) | 1997-06-04 | 1998-06-02 | Fungizide mischungen |
Country Status (18)
| Country | Link |
|---|---|
| US (1) | US6303599B1 (de) |
| EP (1) | EP1009235A1 (de) |
| JP (1) | JP2002508756A (de) |
| KR (1) | KR20010013341A (de) |
| CN (1) | CN1259015A (de) |
| AR (1) | AR015855A1 (de) |
| AU (1) | AU9811598A (de) |
| BR (1) | BR9810421A (de) |
| CA (1) | CA2291784A1 (de) |
| CO (1) | CO5031334A1 (de) |
| EA (1) | EA002404B1 (de) |
| HU (1) | HUP0003909A3 (de) |
| IL (1) | IL132932A0 (de) |
| PL (1) | PL337157A1 (de) |
| SK (1) | SK163599A3 (de) |
| TW (1) | TW505505B (de) |
| WO (1) | WO1998054970A1 (de) |
| ZA (1) | ZA984756B (de) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2773261B1 (fr) * | 1997-12-30 | 2000-01-28 | Commissariat Energie Atomique | Procede pour le transfert d'un film mince comportant une etape de creation d'inclusions |
| US20050032903A1 (en) * | 2003-08-08 | 2005-02-10 | Suarez-Cervieri Miguel Octavio | Method for controlling fungal sieases in legumes |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0645087A1 (de) * | 1993-09-24 | 1995-03-29 | BASF Aktiengesellschaft | Fungizide Mischungen |
| EP0648417A1 (de) * | 1993-09-24 | 1995-04-19 | BASF Aktiengesellschaft | Fungizide Mischungen |
| EP0737421A1 (de) * | 1993-12-27 | 1996-10-16 | Sumitomo Chemical Company Limited | Bakterizide zusammensetzung |
| WO1997006681A1 (de) * | 1995-08-17 | 1997-02-27 | Basf Aktiengesellschaft | Fungizide mischungen eines oximethercarbonsäureamids mit einem morpholin- oder piperidinderivat |
| WO1997006679A1 (de) * | 1995-08-17 | 1997-02-27 | Basf Aktiengesellschaft | Fungizide mischungen |
| FR2742633A1 (fr) * | 1995-12-22 | 1997-06-27 | Rhone Poulenc Agrochimie | Composition fongicide synergique comprenant un compose analogue de la strobilurine |
| WO1997040673A1 (de) * | 1996-04-26 | 1997-11-06 | Basf Aktiengesellschaft | Fungizide mischungen |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3623921A1 (de) | 1986-07-16 | 1988-01-21 | Basf Ag | Oximether und diese enthaltende fungizide |
| GB8617648D0 (en) | 1986-07-18 | 1986-08-28 | Ici Plc | Fungicides |
| GB8903019D0 (en) | 1989-02-10 | 1989-03-30 | Ici Plc | Fungicides |
| DE4030038A1 (de) | 1990-09-22 | 1992-03-26 | Basf Ag | Ortho-substituierte phenylessigsaeureamide |
| DE4124255A1 (de) | 1991-07-22 | 1993-01-28 | Basf Ag | Fungizide mischung |
| WO1995018789A1 (en) | 1994-01-05 | 1995-07-13 | Ciba-Geigy Ag | Pesticides |
| US5889059A (en) | 1994-02-04 | 1999-03-30 | Basf Aktiengesellschaft | Phenylacetic acid derivatives, preparation thereof and intermediates therefor, and compositions containing them |
| JP3307396B2 (ja) | 1994-02-04 | 2002-07-24 | ビーエーエスエフ アクチェンゲゼルシャフト | フェニル酢酸誘導体、これを製造するための方法および中間生成物、並びにこれを含有する薬剤 |
| DE4423613A1 (de) | 1994-07-06 | 1996-01-11 | Basf Ag | 2-[1',2',4'-Triazol-3'yloxymethylen]-anilide, Verfahren zu ihrer Herstellung und ihre Verwendung |
| DE4423612A1 (de) | 1994-07-06 | 1996-01-11 | Basf Ag | 2-[(Dihydro)pyrazolyl-3'-oxymethylen]-anilide, Verfahren zu ihrer Herstelung und ihre Verwendung |
| US5554616A (en) | 1994-12-28 | 1996-09-10 | Basf Aktiengesellschaft | Fungicidal mixtures |
| HU227961B1 (en) | 1996-04-26 | 2012-07-30 | Basf Ag | Fungicide mixture |
-
1998
- 1998-06-02 JP JP50148399A patent/JP2002508756A/ja active Pending
- 1998-06-02 US US09/424,918 patent/US6303599B1/en not_active Expired - Fee Related
- 1998-06-02 SK SK1635-99A patent/SK163599A3/sk unknown
- 1998-06-02 CA CA002291784A patent/CA2291784A1/en not_active Abandoned
- 1998-06-02 WO PCT/EP1998/003282 patent/WO1998054970A1/de not_active Application Discontinuation
- 1998-06-02 IL IL13293298A patent/IL132932A0/xx unknown
- 1998-06-02 HU HU0003909A patent/HUP0003909A3/hu unknown
- 1998-06-02 BR BR9810421-7A patent/BR9810421A/pt not_active IP Right Cessation
- 1998-06-02 EA EA199901068A patent/EA002404B1/ru not_active IP Right Cessation
- 1998-06-02 CN CN98805796A patent/CN1259015A/zh active Pending
- 1998-06-02 AU AU98115/98A patent/AU9811598A/en not_active Abandoned
- 1998-06-02 PL PL98337157A patent/PL337157A1/xx unknown
- 1998-06-02 KR KR19997011337A patent/KR20010013341A/ko not_active Application Discontinuation
- 1998-06-02 EP EP98967167A patent/EP1009235A1/de not_active Withdrawn
- 1998-06-03 ZA ZA9804756A patent/ZA984756B/xx unknown
- 1998-06-03 CO CO98031429A patent/CO5031334A1/es unknown
- 1998-06-04 AR ARP980102639A patent/AR015855A1/es not_active Application Discontinuation
- 1998-06-04 TW TW087108843A patent/TW505505B/zh active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0645087A1 (de) * | 1993-09-24 | 1995-03-29 | BASF Aktiengesellschaft | Fungizide Mischungen |
| EP0648417A1 (de) * | 1993-09-24 | 1995-04-19 | BASF Aktiengesellschaft | Fungizide Mischungen |
| EP0737421A1 (de) * | 1993-12-27 | 1996-10-16 | Sumitomo Chemical Company Limited | Bakterizide zusammensetzung |
| WO1997006681A1 (de) * | 1995-08-17 | 1997-02-27 | Basf Aktiengesellschaft | Fungizide mischungen eines oximethercarbonsäureamids mit einem morpholin- oder piperidinderivat |
| WO1997006679A1 (de) * | 1995-08-17 | 1997-02-27 | Basf Aktiengesellschaft | Fungizide mischungen |
| FR2742633A1 (fr) * | 1995-12-22 | 1997-06-27 | Rhone Poulenc Agrochimie | Composition fongicide synergique comprenant un compose analogue de la strobilurine |
| WO1997040673A1 (de) * | 1996-04-26 | 1997-11-06 | Basf Aktiengesellschaft | Fungizide mischungen |
Also Published As
| Publication number | Publication date |
|---|---|
| ZA984756B (en) | 1999-12-03 |
| CA2291784A1 (en) | 1998-12-10 |
| SK163599A3 (en) | 2000-06-12 |
| US6303599B1 (en) | 2001-10-16 |
| CO5031334A1 (es) | 2001-04-27 |
| EA199901068A1 (ru) | 2000-06-26 |
| KR20010013341A (ko) | 2001-02-26 |
| CN1259015A (zh) | 2000-07-05 |
| AR015855A1 (es) | 2001-05-30 |
| HUP0003909A3 (en) | 2002-01-28 |
| AU9811598A (en) | 1998-12-21 |
| JP2002508756A (ja) | 2002-03-19 |
| IL132932A0 (en) | 2001-03-19 |
| EA002404B1 (ru) | 2002-04-25 |
| HUP0003909A2 (hu) | 2001-04-28 |
| PL337157A1 (en) | 2000-07-31 |
| TW505505B (en) | 2002-10-11 |
| BR9810421A (pt) | 2000-07-25 |
| EP1009235A1 (de) | 2000-06-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0900009B1 (de) | Fungizide mischungen | |
| EP0906017B1 (de) | Fungizide mischungen | |
| EP0901322A1 (de) | Fungizide mischungen | |
| EP1130967A1 (de) | Fungizide mischungen | |
| EP0900019A1 (de) | Fungizide mischungen | |
| EP0984687B1 (de) | Fungizide mischungen | |
| WO1998053688A1 (de) | Fungizide mischungen | |
| EP0912088B1 (de) | Fungizide mischungen | |
| EP1365652A1 (de) | Fungizide mischungen aus benzophenonen und n-biphenylnikotinamiden | |
| WO2002049435A1 (de) | Fungizide mischungen auf der basis von amidverbindungen | |
| WO1998054970A1 (de) | Fungizide mischungen | |
| EP0900013A1 (de) | Fungizide mischungen | |
| WO1998008384A1 (de) | Fungizide mischungen | |
| WO1998053686A1 (de) | Fungizide mischung | |
| WO2003022053A1 (de) | Fungizide mischungen | |
| WO1997040678A1 (de) | Fungizide mischungen | |
| EP0900012A1 (de) | Fungizide mischungen | |
| EP0986305A1 (de) | Fungizide mischungen | |
| WO2002056690A1 (de) | Fungizide mischungen | |
| EP0984690B1 (de) | Fungizide mischung | |
| WO1998053692A1 (de) | Fungizide mischungen | |
| WO1997040684A1 (de) | Fungizide mischungen | |
| WO2002049438A2 (de) | Fungizide mischungen auf der basis von carbamaten | |
| WO1998053693A1 (de) | Fungizide mischungen | |
| WO1998053685A1 (de) | Fungizide mischungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 132932 Country of ref document: IL Ref document number: 98805796.4 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AL AU BG BR BY CA CN CZ GE HU ID IL JP KR KZ LT LV MX NO NZ PL RO RU SG SI SK TR UA US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1998967167 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 501239 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/1999/010819 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 2291784 Country of ref document: CA Ref document number: 2291784 Country of ref document: CA Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 163599 Country of ref document: SK Ref document number: PV1999-4286 Country of ref document: CZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 09424918 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1019997011337 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 98115/98 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 199901068 Country of ref document: EA |
|
| NENP | Non-entry into the national phase |
Ref document number: 1999501483 Country of ref document: JP |
|
| WWP | Wipo information: published in national office |
Ref document number: PV1999-4286 Country of ref document: CZ |
|
| WWP | Wipo information: published in national office |
Ref document number: 1998967167 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1019997011337 Country of ref document: KR |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1998967167 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1019997011337 Country of ref document: KR |
|
| WWR | Wipo information: refused in national office |
Ref document number: PV1999-4286 Country of ref document: CZ |