USRE49918E1 - Cytotoxic benzodiazepine derivatives - Google Patents
Cytotoxic benzodiazepine derivatives Download PDFInfo
- Publication number
- USRE49918E1 USRE49918E1 US17/316,241 US202117316241A USRE49918E US RE49918 E1 USRE49918 E1 US RE49918E1 US 202117316241 A US202117316241 A US 202117316241A US RE49918 E USRE49918 E US RE49918E
- Authority
- US
- United States
- Prior art keywords
- carbon atoms
- linear
- optionally substituted
- branched
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
- A61K31/5517—1,4-Benzodiazepines, e.g. diazepam or clozapine condensed with five-membered rings having nitrogen as a ring hetero atom, e.g. imidazobenzodiazepines, triazolam
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/545—Heterocyclic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/64—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/68035—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug being a pyrrolobenzodiazepine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6849—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a receptor, a cell surface antigen or a cell surface determinant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6867—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from a cell of a blood cancer
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/94—Stability, e.g. half-life, pH, temperature or enzyme-resistance
Definitions
- the present invention relates to novel cytotoxic compounds, and cytotoxic conjugates comprising these cytotoxic compounds and cell-binding agents. More specifically, this invention relates to novel benzodiazepine compounds derivatives thereof, intermediates thereof, conjugates thereof, and pharmaceutically acceptable salts thereof, which are useful as medicaments, in particular as anti-proliferative agents.
- Benzodiazepine derivatives are useful compounds for treating various disorders, and include medicaments such as, antiepileptics (imidazo [2,1-b][1,3,5]benzothiadiazepines, U.S. Pat. Nos. 4,444,688; 4,062,852), antibacterials (pyrimido[1,2-c][1,3,5]benzothiadiazepines, GB 1476684), diuretics and hypotensives (pyrrolo(1,2-b)[1,2,5]benzothiadiazepine 5,5 dioxide, U.S. Pat. No. 3,506,646), hypolipidemics (WO 03091232), anti-depressants (U.S. Pat. No. 3,453,266); osteoporosis (JP 2138272).
- medicaments such as, antiepileptics (imidazo [2,1-b][1,3,5]benzothiadiazepines, U.S. Pat. Nos. 4,444,688; 4,062,852),
- benzodiazepine derivatives such as pyrrolobenzodiazepines (PBDs)
- PBDs pyrrolobenzodiazepines
- PBDs Physical structure of PBDs is described in US Publication Number 20070072846.
- the PBDs differ in the number, type and position of substituents, in both their aromatic A rings and pyrrolo C rings, and in the degree of saturation of the C ring. Their ability to form an adduct in the minor groove and crosslink DNA enables them to interfere with DNA processing, hence their potential for use as antiproliferative agents.
- SJG-136 (NSC 694501) is a potent cytotoxic agent that causes DNA inter-strand crosslinks (S. G Gregson et al., 2001, J. Med. Chem., 44: 737-748; M. C. Alley et al., 2004, Cancer Res., 64: 6700-6706; J. A. Hartley et al., 2004, Cancer Res., 64: 6693-6699; C. Martin et al., 2005, Biochemistry., 44: 4135-4147; S. Arnould et al., 2006, Mol. Cancer Ther., 5: 1602-1509).
- Cytotoxic benzodiazepine dimers disclosed in the art possess two imine functionalities in their free form or reversibly protected form, such as a hydrate, alkoxylate or sulfonate. The presence of these two imine functionalities results in crosslinking of DNA (S. G. Gregson et al., 2001, J. Med. Chem., 44: 737-748).
- the present invention is partly based on the unexpected finding that cell binding agent conjugates of new cytotoxic benzodiazepine derivatives, such as indolinobenzodiazapene dimers that are devoid of two imine functionalities (e.g., one imine functionality and one amine functionality), and thus incapable of crosslinking DNA, display a much higher therapeutic index (ratio of maximum tolerated dose to minimum effective dose) in vivo compared to benzodiazepine derivatives that can crosslink DNA that are previously disclosed in the art.
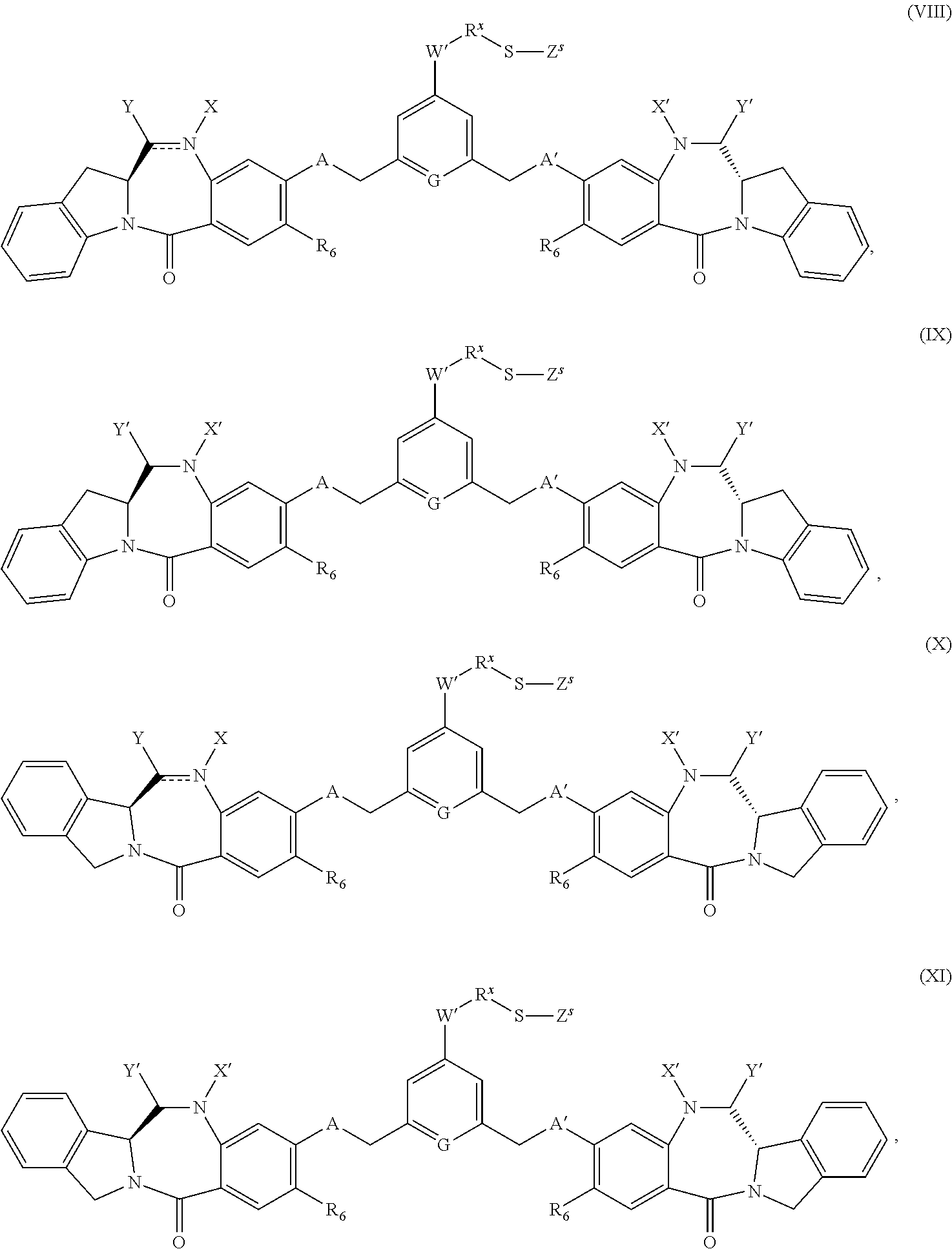
- one object of the invention is to provide cytotoxic compound comprising a linking group with a reactive group bonded thereto capable of covalently linking the cytotoxic compound to a cell binding agent (CBA, see below), wherein the cytotoxic compound is represented by any one of the following formulas:
- X is not the linking group with the reactive group bonded thereto.
- the double line between N and C represents a single bond, Y is not —H.
- the compound is not any one of the following compounds:
- Y is a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R′′, —NR′R′′, —NR′COR′′, —NR′NR′R′′, an optionally substituted 5- or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C ⁇ NH)NR′R′′, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO 2 M, —SO 3 M, —OSO 3 M, halogen, cyano and an azido.
- P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO 2 M
- a second object of the invention is to provide conjugates of cell binding agents with the novel benzodiazepine compounds or derivatives thereof of the present invention. These conjugates are useful as therapeutic agents, which are delivered specifically to target cells and are cytotoxic.
- a conjugate of the invention may comprise: a cytotoxic compound and a cell binding agent (CBA), wherein the cytotoxic compound comprises a linking group which covalently links the cytotoxic compound to the CBA, and wherein the cytotoxic compound is represented by any one of the following formulas:
- X is not the linking group.
- the double line between N and C represents a single bond, Y is not —H.
- Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R′′, —NR′R′′, —NR′COR′′, —NR′NR′R′′, an optionally substituted 5- or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C ⁇ NH)NR′R′′, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO 2 M, —SO 3 M, —OSO 3 M, halogen, cyano and an azido.
- Y is not —H.
- the compound of the conjugate is not any one of the following compounds (the wavy bond represents the bond through which the compound
- the present invention also includes a composition (e.g., a pharmaceutical composition) comprising novel benzodiazepine compounds, derivatives thereof, or conjugates thereof, (and/or solvates, hydrates and/or salts thereof) and a carrier (a pharmaceutically acceptable carrier).
- a composition e.g., a pharmaceutical composition
- the present invention additionally includes a composition (e.g., a pharmaceutical composition) comprising novel benzodiazepine compounds, derivatives thereof, or conjugates thereof (and/or solvates, hydrates and/or salts thereof), and a carrier (a pharmaceutically acceptable carrier), further comprising a second therapeutic agent.
- the present compositions are useful for inhibiting abnormal cell growth or treating a proliferative disorder in a mammal (e.g., human).
- compositions are useful for treating conditions such as cancer, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, lupus, myositis, infection, immune deficiency such as AIDS, and inflammatory diseases in a mammal (e.g., human).
- conditions such as cancer, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, lupus, myositis, infection, immune deficiency such as AIDS, and inflammatory diseases in a mammal (e.g., human).
- the present invention includes a method of inhibiting abnormal cell growth or treating a proliferative disorder in a mammal (e.g., human) comprising administering to said mammal a therapeutically effective amount of novel benzodiazepine compounds, derivatives thereof, or conjugates thereof, (and/or solvates and salts thereof) or a composition thereof, alone or in combination with a second therapeutic agent.
- a mammal e.g., human
- the present invention includes a method of synthesizing and using novel benzodiazepine compounds, derivatives thereof, and conjugates thereof for in vitro, in situ, and in vivo diagnosis or treatment of mammalian cells, organisms, or associated pathological conditions.
- the compounds of this invention, derivatives thereof, or conjugates thereof, and compositions comprising them, are useful for treating or lessening the severity of disorders, such as, characterized by abnormal growth of cells (e.g., cancer).
- Other applications for compounds and conjugates of this invention include, but are not limited to, treating conditions such as cancer, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, lupus, myositis, infection, immune deficiency such as AIDS and inflammatory diseases in a mammal (e.g., human).
- FIGS. 1 - 6 show the schemes for the synthesis of benzodiazepine compounds and the corresponding linkable compounds suitable for conjugation of the present invention.
- FIG. 7 shows the scheme for the synthesis of representative compounds with PEG modified linkers of the present invention.
- FIG. 8 shows the scheme for the synthesis of representative compounds with a methylthio linker of the present invention.
- FIGS. 9 - 10 show the schemes for the synthesis of representative compounds containing a tertiary amine of the present invention.
- FIG. 11 shows the scheme for the synthesis of representative compounds with a peptide linker of the present invention.
- FIGS. 12 A, 12 B, and 13 - 19 show the schemes for the synthesis of representative compounds suitable for one-step conjugation methods of the present invention.
- FIG. 20 shows the scheme for a two-step mono-imine dimer synthesis.
- FIG. 21 shows the scheme for a two-step di-reduced dimer synthesis.
- FIG. 22 A , FIG. 22 B and FIG. 22 C show the scheme for the one-step synthesis of the representative antibody-drug conjugates.
- FIG. 22 A shows the one-step conjugate scheme of Compound 1g to a lysine moiety of an antibody.
- FIG. 22 B shows the one-step conjugate scheme of Compound 10 to a lysine moiety of an antibody.
- FIG. 22 C shows the one-step conjugate scheme of Compound 1g to the antibody huMy9-6.
- FIG. 23 shows the scheme for the two-step synthesis of the representative antibody-drug conjugates.
- FIGS. 24 A, 24 B, and 24 C show the in vitro cytotoxicity of the methyldithio dimer 1d against Namalwa, KB and HL60/QC cell lines.
- FIGS. 25 A, 25 B and 25 C show the in vitro cytotoxicity and specificity of the huMy9-6-SPDB-1f conjugates against various cell lines.
- FIG. 25 A shows the in vitro cytotoxicity and specificity of the huMy9-6-SPDB-1f conjugates against HL60/QC (Ag + ) cells with and without blocking of antigen binding sites NB-4 (Ag + ) cells.
- FIG. 25 B shows the in vitro cytotoxicity and specificity of the huMy9-6-SPDB-1f conjugates against HL60/ATCC (Ag + ) cells.
- FIG. 25 C shows the in vitro cytotoxicity and specificity of the huMy9-6-SPDB-1f conjugates against NB-4 (Ag + ) cells. Note that sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIG. 26 shows the in vitro cytotoxicity and specificity of the huFOLR1-SPDB-1f conjugates.
- FIG. 27 shows conjugation of dimer does not reduce binding affinity of antibody. Note that sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIG. 28 shows the in vivo antitumor activity of huMy9-6 conjugate. Note that sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIGS. 29 A, 29 B, 29 C and 29 D show in vitro cytotoxicity of huMy9-6-SPDB-1f conjugate against antigen positive cells.
- FIG. 29 A shows in vitro cytotoxicity of huMy9-6-SPDB-1f conjugate against antigen positive HL60/QC cells.
- FIG. 29 B shows in vitro cytotoxicity of huMy9-6-SPDB-1f conjugate against antigen positive HL60/ATCC cells.
- FIG. 29 C shows in vitro cytotoxicity of huMy9-6-SPDB-1f conjugate against antigen positive NB-4 cells.
- FIG. 29 D shows in vitro cytotoxicity of huMy9-6-SPDB-1f conjugate against antigen positive HEL 92.1.7 cells. Note that sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIG. 30 shows synthetic scheme for making thioether-containing linker disulfides 27e-h.
- FIG. 31 shows synthetic scheme for making dimers 28c-f.
- FIG. 32 shows synthetic scheme for making phenyl linked dimers 29b-c.
- FIG. 33 shows the scheme for an alternative two-step synthesis for mono-imine dimers.
- FIGS. 34 A, 34 B and 34 C show in vitro cytotoxicity for huMy9-6-SPDB-1f (A), huMy9-6-sulfoSPDB-1f (B) and huMy9-6-BMPS-1f (C) against HL60/QC (Ag + ) cells with and without blocking of antigen binding sites, respectively. Note that in all three experiments ( 34 A, 34 B, and 34 C), sodium bisulfite were added to the conjugation reaction for making the conjugate.
- FIGS. 35 A and 35 B show in vitro cytotoxicity for chB38.1-SPDB-1f (A), and chB38.1-sulfoSPDB-1f (B) against COLO205 (Ag + ) cells, respectively. Note that in both experiments, sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIG. 36 shows in vivo efficacy of huMy9-6-SPDB-1f in HL60/QC bearing mice. Note that sodium bisulfite was added to the conjugation reaction.
- FIG. 37 shows in vivo efficacy of huFOLR1-SPDB-1f in KB tumor bearing mice.
- FIG. 38 shows synthetic scheme of compound 1.
- FIG. 39 shows a synthetic scheme of compound 1d with 5-ethyl-2-methylpyridine borane (PEMB).
- PEMB 5-ethyl-2-methylpyridine borane
- FIG. 40 shows a synthetic scheme of compound 1d with sodium triacetoxyborohydride (STAB).
- FIG. 41 shows a synthetic scheme of compound 31a-c.
- FIG. 42 shows a synthetic scheme of compound 32c,d.
- FIG. 43 shows a synthetic scheme of compounds ii and 12a.
- FIGS. 44 A, 44 B and 44 C show antiproliferative activity by comparing huMy9-6-SPDB-1f, huMy9-6-sulfoSPDB-1f, and huMy9-6-BMPS-1f, respectively, against OCI-AML3 (Ag + ) cells with and without blocking of antigen binding sites. Note that in all three experiments, sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIG. 45 shows an alternate scheme for synthesizing 4-(benzyloxy)-5-methoxy-2-nitrobenzoic acid used in the preparation of IBD monomer.
- FIG. 46 is an alternate synthesis scheme for (5-((2-(2-(2-methoxyethoxy)ethoxy)ethyl)(2-methyl-2-(methyldisulfanyl)propyl)amino)-1,3-phenylene)dimethanol (1b).
- FIG. 47 is an alternate synthesis scheme for (5-((2-(2-(2-methoxyethoxy)ethoxy)ethyl)(2-methyl-2-(methyldisulfanyl)propyl)amino)-1,3-phenylene)dimethanol (1b).
- FIG. 48 is an alternate synthetic scheme for a two-step mono-imine dimer synthesis.
- FIG. 49 shows potency of various conjugates against various cell lines.
- the IC 50 values listed in the table are in the unit of nM.
- FIG. 50 shows in vivo efficacy of huMy9-6-sulfo-SPDB-1f in MOLM-13 tumor bearing mice.
- FIG. 51 shows in vivo efficacy of huMy9-6-sulfo-SPDB-1f in NB4 tumor bearing mice.
- FIG. 52 shows in vivo efficacy of huMy9-6-BMPS-1f in HL60/QC tumor bearing mice.
- FIG. 53 shows in vivo efficacy of huMy9-6-BMPS-1f in MOLM-13 tumor bearing mice. Note that sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIG. 54 shows a representative synthesis scheme for a Sulfonated folate/cytotoxic compound conjugate.
- FIG. 55 shows several representative sulfonated drug-antibody conjugates with different linkers.
- FIG. 56 shows in vivo efficacy of huMy9-6-Drug 2 in HL60/QC tumor bearing mice. Note that sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIG. 57 shows in vivo efficacy of huMy9-6-Drug 2 in MOLM-13 tumor bearing mice. Note that sodium bisulfite was added to the conjugation reaction for making the conjugate.
- FIG. 58 shows similar in vitro cytotoxicity of HuMy9-6-Drug 2 (conjugates prepared without and with sodium bisulfite against CD33-antigen expressing HL60 cells.
- FIG. 59 shows similar in vitro cytotoxicity of anti-CD22 Ab-Drug 2 conjugates prepared without and with sodium bisulfite against CD22-antigen expressing BJAB cells.
- FIG. 60 shows the preparation of huMy9-6-sulfo-SPDB-1d using the highly reactive 4-nitroPy-sulfo-SPDB linker.
- Linear or branched alkyl refers to a saturated linear or branched-chain monovalent hydrocarbon radical of one to twenty carbon atoms.
- alkyl include, but are not limited to, methyl, ethyl, 1-propyl, 2-propyl, 1-butyl, 2-methyl-1-propyl, —CH 2 CH(CH 3 ) 2 ), 2-butyl, 2-methyl-2-propyl, 1-pentyl, 2-pentyl 3-pentyl, 2-methyl-2-butyl, 3-methyl-2-butyl, 3-methyl-1-butyl, 2-methyl-1-butyl, 1-hexyl), 2-hexyl, 3-hexyl, 2-methyl-2-pentyl, 3-methyl-2-pentyl, 4-methyl-2-pentyl, 3-methyl-3-pentyl, 2-methyl-3-pentyl, 2,3-dimethyl-2-butyl, 3,3-dimethyl-2-butyl, 1-heptyl,
- Linear or branched alkenyl refers to linear or branched-chain monovalent hydrocarbon radical of two to twenty carbon atoms with at least one site of unsaturation, i.e., a carbon-carbon, double bond, wherein the alkenyl radical includes radicals having “cis” and “trans” orientations, or alternatively, “E” and “Z” orientations. Examples include, but are not limited to, ethylenyl or vinyl (—CH ⁇ CH 2 ), allyl (—CH 2 CH ⁇ CH 2 ), and the like.
- the alkenyl has two to ten carbon atoms. More preferably, the alkyl has two to four carbon atoms.
- Linear or branched alkynyl refers to a linear or branched monovalent hydrocarbon radical of two to twenty carbon atoms with at least one site of unsaturation, i.e., a carbon-carbon, triple bond. Examples include, but are not limited to, ethynyl, propynyl, 1-butynyl, 2-butynyl, 1-pentynyl, 2-pentynyl, 3-pentynyl, hexynyl, and the like.
- the alkynyl has two to ten carbon atoms. More preferably, the alkynyl has two to four carbon atoms.
- carrier refers to a monovalent non-aromatic, saturated or partially unsaturated ring having 3 to 12 carbon atoms as a monocyclic ring or 7 to 12 carbon atoms as a bicyclic ring.
- Bicyclic carbocycles having 7 to 12 atoms can be arranged, for example, as a bicyclo [4,5], [5,5], [5,6], or [6,6] system, and bicyclic carbocycles having 9 or 10 ring atoms can be arranged as a bicyclo [5,6] or [6,6] system, or as bridged systems such as bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane and bicyclo[3.2.2]nonane.
- monocyclic carbocycles include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, 1-cyclopent-1-enyl, 1-cyclopent-2-enyl, 1-cyclopent-3-enyl, cyclohexyl, 1-cyclohex-1-enyl, 1-cyclohex-2-enyl, 1-cyclohex-3-enyl, cyclohexadienyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, cycloundecyl, cyclododecyl, and the like.
- cyclic alkyl and “cycloalkyl” can be used interchangeably. They refer to a monovalent saturated carbocyclic ring radical. Preferably, the cyclic alkyl is 3 to 7 membered monocyclic ring radical. More preferably, the cyclic alkyl is cyclohexyl.
- cyclic alkenyl refers to a carbocyclic ring radical having at least one double bond in the ring structure.
- cyclic alkynyl refers to a carbocyclic ring radical having at least one triple bond in the ring structure.
- Aryl means a monovalent aromatic hydrocarbon radical of 6-18 carbon atoms derived by the removal of one hydrogen atom from a single carbon atom of a parent aromatic ring system. Some aryl groups are represented in the exemplary structures as “Ar.” Aryl includes bicyclic radicals comprising an aromatic ring fused to a saturated, partially unsaturated ring, or aromatic carbocyclic or heterocyclic ring.
- Typical aryl groups include, but are not limited to, radicals derived from benzene (phenyl), substituted benzenes, naphthalene, anthracene, indenyl, indanyl, 1,2-dihydronapthalene, 1,2,3,4-tetrahydronapthyl, and the like.
- aryl is phenyl group.
- heterocycle refers to a saturated or a partially unsaturated (i.e., having one or more double and/or triple bonds within the ring) carbocyclic radical of 3 to 18 ring atoms in which at least one ring atom is a heteroatom selected from nitrogen, oxygen, phosphorus, and sulfur, the remaining ring atoms being C, where one or more ring atoms is optionally substituted independently with one or more substituents described below.
- a heterocycle may be a monocycle having 3 to 7 ring members (2 to 6 carbon atoms and 1 to 4 heteroatoms selected from N, O, P, and S) or a bicycle having 7 to 10 ring members (4 to 9 carbon atoms and 1 to 6 heteroatoms selected from N, O, P, and S), for example: a bicyclo [4,5], [5,5], [5,6], or [6,6] system.
- Heterocycles are described in Paquette, Leo A.; “Principles of Modern Heterocyclic Chemistry” (W. A.
- Heterocyclyl also includes radicals where heterocycle radicals are fused with a saturated, partially unsaturated ring, or aromatic carbocyclic or heterocyclic ring.
- heterocyclic rings include, but are not limited to, pyrrolidinyl, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothienyl, tetrahydropyranyl, dihydropyranyl, tetrahydrothiopyranyl, piperidino, morpholino, thiomorpholino, thioxanyl, piperazinyl, homopiperazinyl, azetidinyl, oxetanyl, thietanyl, homopiperidinyl, oxepanyl, thiepanyl, oxazepinyl, diazepinyl, thiazepinyl, 2-pyrrolinyl, 3-pyrrolinyl, indolinyl, 2H-pyranyl, 4H-pyranyl, dioxanyl, 1,3-dioxolanyl, pyrazolinyl, dithianyl, dithiolan
- Spiro moieties are also included within the scope of this definition.
- Examples of a heterocyclic group wherein ring atoms are substituted with oxo ( ⁇ O) moieties are pyrimidinonyl and 1,1-dioxo-thiomorpholinyl.
- heteroaryl refers to a monovalent aromatic radical of 5- or 6-membered rings, and includes fused ring systems (at least one of which is aromatic) of 5-18 atoms, containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur.
- heteroaryl groups are pyridinyl (including, for example, 2-hydroxypyridinyl), imidazolyl, imidazopyridinyl, pyrimidinyl (including, for example, 4-hydroxypyrimidinyl), pyrazolyl, triazolyl, pyrazinyl, tetrazolyl, furyl, thienyl, isoxazolyl, thiazolyl, oxazolyl, isothiazolyl, pyrrolyl, quinolinyl, isoquinolinyl, indolyl, benzimidazolyl, benzofuranyl, cinnolinyl, indazolyl, indolizinyl, phthalazinyl, pyridazinyl, triazinyl, isoindolyl, pteridinyl, purinyl, oxadiazolyl, triazolyl, thiadiazolyl, furazanyl,
- the heterocycle or heteroaryl groups may be carbon (carbon-linked) or nitrogen (nitrogen-linked) attached where such is possible.
- carbon bonded heterocycles or heteroaryls are bonded at position 2, 3, 4, 5, or 6 of a pyridine, position 3, 4, 5, or 6 of a pyridazine, position 2, 4, 5, or 6 of a pyrimidine, position 2, 3, 5, or 6 of a pyrazine, position 2, 3, 4, or 5 of a furan, tetrahydrofuran, thiofuran, thiophene, pyrrole or tetrahydropyrrole, position 2, 4, or 5 of an oxazole, imidazole or thiazole, position 3, 4, or 5 of an isoxazole, pyrazole, or isothiazole, position 2 or 3 of an aziridine, position 2, 3, or 4 of an azetidine, position 2, 3, 4, 5, 6, 7, or 8 of a quinoline or position 1, 3, 4, 5, 6, 7,
- nitrogen bonded heterocycles or heteroaryls are bonded at position 1 of an aziridine, azetidine, pyrrole, pyrrolidine, 2-pyrroline, 3-pyrroline, imidazole, imidazolidine, 2-imidazoline, 3-imidazoline, pyrazole, pyrazoline, 2-pyrazoline, 3-pyrazoline, piperidine, piperazine, indole, indoline, 1H-indazole, position 2 of a isoindole, or isoindoline, position 4 of a morpholine, and position 9 of a carbazole, or O-carboline.
- heteroatoms present in heteroaryl or heterocyclcyl include the oxidized forms such as NO, SO, and SO 2 .
- halo or halogen refers to F, Cl, Br or I.
- alkyl, alkenyl, alkynyl, cyclic alkyl, cyclic alkenyl, cyclic alkynyl, carbocyclyl, aryl, heterocyclyl and heteroaryl described above can be optionally substituted with one more (e.g., 2, 3, 4, 5, 6 or more) substituents.
- a non-hydrogen substituent is in the place of a hydrogen substituent on a carbon, oxygen, sulfur or nitrogen of the substituent.
- a substituted alkyl substituent is an alkyl substituent wherein at least one non-hydrogen substituent is in the place of a hydrogen substituent on the alkyl substituent.
- monofluoroalkyl is alkyl substituted with a fluoro substituent
- difluoroalkyl is alkyl substituted with two fluoro substituents. It should be recognized that if there is more than one substitution on a substituent, each non-hydrogen substituent may be identical or different (unless otherwise stated).
- substituent may be either (1) not substituted, or (2) substituted. If a carbon of a substituent is described as being optionally substituted with one or more of a list of substituents, one or more of the hydrogens on the carbon (to the extent there are any) may separately and/or together be replaced with an independently selected optional substituent. If a nitrogen of a substituent is described as being optionally substituted with one or more of a list of substituents, one or more of the hydrogens on the nitrogen (to the extent there are any) may each be replaced with an independently selected optional substituent.
- One exemplary substituent may be depicted as —NR′R′′, wherein R′ and R′′ together with the nitrogen atom to which they are attached, may form a heterocyclic ring.
- the heterocyclic ring formed from R′ and R′′ together with the nitrogen atom to which they are attached may be partially or fully saturated.
- the heterocyclic ring consists of 3 to 7 atoms.
- the heterocyclic ring is selected from the group consisting of pyrrolyl, imidazolyl, pyrazolyl, triazolyl, tetrazolyl, isoxazolyl, pyridyl and thiazolyl.
- a group of substituents are collectively described as being optionally substituted by one or more of a list of substituents, the group may include: (1) unsubstitutable substituents, (2) substitutable substituents that are not substituted by the optional substituents, and/or (3) substitutable substituents that are substituted by one or more of the optional substituents.
- a substituent is described as being optionally substituted with up to a particular number of non-hydrogen substituents, that substituent may be either (1) not substituted; or (2) substituted by up to that particular number of non-hydrogen substituents or by up to the maximum number of substitutable positions on the substituent, whichever is less.
- a substituent is described as a heteroaryl optionally substituted with up to 3 non-hydrogen substituents, then any heteroaryl with less than 3 substitutable positions would be optionally substituted by up to only as many non-hydrogen substituents as the heteroaryl has substitutable positions.
- substituents in non-limiting examples, can be selected from a linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, aryl, heteroaryl, heterocycyclyl, halogen, guanidinium [—NH(C ⁇ NH)NH 2 ], —OR 100 , NR 101 R 102 , —NO 2 , —NR 101 COR 102 , —SR 100 , a sulfoxide represented by —SOR 101 , a sulfone represented by —SO 2 R 101 , a sulfonate —SO 3 M, a sulfate —OSO 3 M, a sulfonamide represented by —SO 2 NR 101 R 102 , cyano, an azido, —COR 101 , —OCOR 101 , —OCONR 101 R 102 and a polyethylene glycol unit (—OCH 2 CH 2 )
- the substituents for the optionally substituted alkyl, alkenyl, alkynyl, cyclic alkyl, cyclic alkenyl, cyclic alkynyl, carbocyclyl, aryl, heterocyclyl and heteroaryl described above include halogen, —CN, —NR 102 R 103 , —CF 3 , —OR 101 , aryl, heteroaryl, heterocycycl, —SR 101 , —SOR 101 , —SO 2 R 101 and —SO 3 M.
- cytotoxic compound or “cytotoxic compound,” “cytotoxic dimer” and “cytotoxic dimer compound” are used interchangeably. They are intended to include compounds for which a structure or formula or any derivative thereof has been disclosed in the present invention or a structure or formula or any derivative thereof that has been incorporated by reference.
- the term also includes, stereoisomers, geometric isomers, tautomers, solvates, metabolites, salts (e.g., pharmaceutically acceptable salts) and prodrugs, and prodrug salts of a compound of all the formulae disclosed in the present invention.
- the term also includes any solvates, hydrates, and polymorphs of any of the foregoing.
- conjugate refers to a compound described herein or a derivative thereof that is linked to a cell binding agent.
- linkable to a cell binding agent refers to the compounds described herein or derivates thereof comprising at least one linking group or a precursor thereof suitable to bond these compounds or derivatives thereof to a cell binding agent.
- precursor of a given group refers to any group which may lead to that group by any deprotection, a chemical modification, or a coupling reaction.
- linked to a cell binding agent refers to a conjugate molecule comprising at least one of the compounds described herein (e.g., compounds of formula (I)-(IV) and (VIII)-(XI) and drug-linker compounds describe herein), or derivative thereof bound to a cell binding agent via a suitable linking group or a precursor thereof.
- chiral refers to molecules which have the property of non-superimposability of the mirror image partner, while the term “achiral” refers to molecules which are superimposable on their mirror image partner.
- stereoisomer refers to compounds which have identical chemical constitution and connectivity, but different orientations of their atoms in space that cannot be interconverted by rotation about single bonds.
- Diastereomer refers to a stereoisomer with two or more centers of chirality and whose molecules are not mirror images of one another. Diastereomers have different physical properties, e.g. melting points, boiling points, spectral properties, and reactivities. Mixtures of diastereomers may separate under high resolution analytical procedures such as crystallization, electrophoresis and chromatography.
- Enantiomers refer to two stereoisomers of a compound which are non-superimposable mirror images of one another.
- the compounds of the invention may contain asymmetric or chiral centers, and therefore exist in different stereoisomeric forms. It is intended that all stereoisomeric forms of the compounds of the invention, including but not limited to, diastereomers, enantiomers and atropisomers, as well as mixtures thereof such as racemic mixtures, form part of the present invention.
- a specific stereoisomer may also be referred to as an enantiomer, and a mixture of such isomers is often called an enantiomeric mixture.
- a 50:50 mixture of enantiomers is referred to as a racemic mixture or a racemate, which may occur where there has been no stereoselection or stereospecificity in a chemical reaction or process.
- the terms “racemic mixture” and “racemate” refer to an equimolar mixture of two enantiomeric species, devoid of optical activity.
- tautomer or “tautomeric form” refers to structural isomers of different energies which are interconvertible via a low energy barrier.
- proton tautomers also known as prototropic tautomers
- Valence tautomers include interconversions by reorganization of some of the bonding electrons.
- prodrug refers to a precursor or derivative form of a compound of the invention that is capable of being enzymatically or hydrolytically activated or converted into the more active parent form. See, e.g., Wilman, “Prodrugs in Cancer Chemotherapy” Biochemical Society Transactions, 14, pp. 375-382, 615th Meeting Harbor (1986) and Stella et al., “Prodrugs: A Chemical Approach to Targeted Drug Delivery,” Directed Drug Delivery, Borchardt et al., (ed.), pp. 247-267, Humana Press (1985).
- the prodrugs of this invention include, but are not limited to, ester-containing prodrugs, phosphate-containing prodrugs, thiophosphate-containing prodrugs, sulfate-containing prodrugs, peptide-containing prodrugs, D-amino acid-modified prodrugs, glycosylated prodrugs, ⁇ -lactam-containing prodrugs, optionally substituted phenoxyacetamide-containing prodrugs, optionally substituted phenylacetamide-containing prodrugs, 5-fluorocytosine and other 5-fluorouridine prodrugs which can be converted into the more active cytotoxic free drug.
- cytotoxic drugs that can be derivatized into a prodrug form for use in this invention include, but are not limited to, compounds of the invention and chemotherapeutic agents such as described above.
- prodrug is also meant to include a derivative of a compound that can hydrolyze, oxidize, or otherwise react under biological conditions (in vitro or in vivo) to provide a compound of this invention.
- Prodrugs may only become active upon such reaction under biological conditions, or they may have activity in their unreacted forms.
- Examples of prodrugs contemplated in this invention include, but are not limited to, analogs or derivatives of compounds of any one of the formulae disclosed herein that comprise biohydrolyzable moieties such as biohydrolyzable amides, biohydrolyzable esters, biohydrolyzable carbamates, biohydrolyzable carbonates, biohydrolyzable ureides, and biohydrolyzable phosphate analogues.
- prodrugs include derivatives of compounds of any one of the formulae disclosed herein that comprise —NO, —NO 2 , —ONO, or —ONO 2 moieties.
- Prodrugs can typically be prepared using well-known methods, such as those described by Burger's Medicinal Chemistry and Drug Discovery (1995) 172-178, 949-982 (Manfred E. Wolff ed., 5th ed); see also Goodman and Gilman's, The Pharmacological basis of Therapeutics, 8th ed., McGraw-Hill, Int. Ed. 1992, “Biotransformation of Drugs.”
- prodrug of the invention includes compounds (with or without any linker groups) and conjugates of the invention comprising an adduct formed between an imine bond of the compounds/conjugates and an imine reactive reagent.
- Another preferred form of prodrug of the invention includes compounds such as those of formula (I)-(IV), wherein when the double line between N and C represents a single bond, X is H or an amine protecting group, and the compound becomes a prodrug.
- a prodrug of the invention may contain one or both forms of prodrugs described herein (e.g., containing an adduct formed between an imine bond of the compounds/conjugates and an imine reactive reagent, and/or containing a Y leaving group when X is —H).
- imine reactive reagent refers to a reagent that is capable of reacting with an imine group.
- imine reactive reagent includes, but is not limited to, sulfites (H 2 SO 3 , H 2 SO 2 or a salt of HSO 3 ⁇ , SO 3 2 ⁇ or HSO 2 formed with a cation), metabisulfite (H 2 S 2 O 5 or a salt of S 2 O 5 2 ⁇ formed with a cation), mono, di, tri, and tetra-thiophosphates (PO 3 SH 3 , PO 2 S 2 H 3 , POS 3 H 3 , PS 4 H 3 or a salt of PO 3 S 3 ⁇ , PO 2 S 2 3- , POS 3 3 ⁇ or PS 4 3 ⁇ formed with a cation), thio phosphate esters ((R i O) 2 PS(OR i ), R i SH, R i SOH, R i SO 2 H, R i SO 3 H), various amines (
- the cation is a monovalent cation, such as Na + or K + .
- the imine reactive reagent is selected from sulfites, hydroxyl amine, urea and hydrazine. More preferably, the imine reactive reagent is NaHSO 3 or KHSO 3 .
- biohydrolyzable amide As used herein and unless otherwise indicated, the terms “biohydrolyzable amide,” “biohydrolyzable ester,” “biohydrolyzable carbamate,” “biohydrolyzable carbonate,” “biohydrolyzable ureide” and “biohydrolyzable phosphate analogue” mean an amide, ester, carbamate, carbonate, ureide, or phosphate analogue, respectively, that either: 1) does not destroy the biological activity of the compound and confers upon that compound advantageous properties in vivo, such as uptake, duration of action, or onset of action; or 2) is itself biologically inactive but is converted in vivo to a biologically active compound.
- biohydrolyzable amides include, but are not limited to, lower alkyl amides, ⁇ -amino acid amides, alkoxyacyl amides, and alkylaminoalkylcarbonyl amides.
- biohydrolyzable esters include, but are not limited to, lower alkyl esters, alkoxyacyloxy esters, alkyl acylamino alkyl esters, and choline esters.
- biohydrolyzable carbamates include, but are not limited to, lower alkylamines, substituted ethylenediamines, amino acids, hydroxyalkylamines, heterocyclic and heteroaromatic amines, and polyether amines.
- Particularly favored prodrugs and prodrug salts are those that increase the bioavailability of the compounds of this invention when such compounds are administered to a mammal.
- phrases “pharmaceutically acceptable salt” as used herein, refers to pharmaceutically acceptable organic or inorganic salts of a compound of the invention.
- Exemplary salts include, but are not limited, to sulfate, citrate, acetate, oxalate, chloride, bromide, iodide, nitrate, bisulfate, phosphate, acid phosphate, isonicotinate, lactate, salicylate, acid citrate, tartrate, oleate, tannate, pantothenate, bitartrate, ascorbate, succinate, maleate, gentisinate, fumarate, gluconate, glucuronate, saccharate, formate, benzoate, glutamate, methanesulfonate “mesylate,” ethanesulfonate, benzenesulfonate, p-toluenesulfonate, pamoate (i.e., 1,1′-methylene-bis-(
- a pharmaceutically acceptable salt may involve the inclusion of another molecule such as an acetate ion, a succinate ion or other counter ion.
- the counter ion may be any organic or inorganic moiety that stabilizes the charge on the parent compound.
- a pharmaceutically acceptable salt may have more than one charged atom in its structure. Instances where multiple charged atoms are part of the pharmaceutically acceptable salt can have multiple counter ions. Hence, a pharmaceutically acceptable salt can have one or more charged atoms and/or one or more counter ion.
- the desired pharmaceutically acceptable salt may be prepared by any suitable method available in the art, for example, treatment of the free base with an inorganic acid, such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, methanesulfonic acid, phosphoric acid and the like, or with an organic acid, such as acetic acid, maleic acid, succinic acid, mandelic acid, fumaric acid, malonic acid, pyruvic acid, oxalic acid, glycolic acid, salicylic acid, a pyranosidyl acid, such as glucuronic acid or galacturonic acid, an alpha hydroxy acid, such as citric acid or tartaric acid, an amino acid, such as aspartic acid or glutamic acid, an aromatic acid, such as benzoic acid or cinnamic acid, a sulfonic acid, such as p-toluenesulfonic acid or ethanesulfonic acid
- an inorganic acid such as hydro
- the desired pharmaceutically acceptable salt may be prepared by any suitable method, for example, treatment of the free acid with an inorganic or organic base, such as an amine (primary, secondary or tertiary), an alkali metal hydroxide or alkaline earth metal hydroxide, or the like.
- suitable salts include, but are not limited to, organic salts derived from amino acids, such as glycine and arginine, ammonia, primary, secondary, and tertiary amines, and cyclic amines, such as piperidine, morpholine and piperazine, and inorganic salts derived from sodium, calcium, potassium, magnesium, manganese, iron, copper, zinc, aluminum and lithium.
- solvate means a compound which further includes a stoichiometric or non-stoichiometric amount of solvent such as water, isopropanol, acetone, ethanol, methanol, DMSO, ethyl acetate, acetic acid, and ethanolamine dichloromethane, 2-propanol, or the like, bound by non-covalent intermolecular forces.
- Solvates or hydrates of the compounds are readily prepared by addition of at least one molar equivalent of a hydroxylic solvent such as methanol, ethanol, 1-propanol, 2-propanol or water to the compound to result in solvation or hydration of the imine moiety.
- abnormal cell growth and “proliferative disorder” are used interchangeably in this application.
- Abnormal cell growth refers to cell growth that is independent of normal regulatory mechanisms (e.g., loss of contact inhibition).
- tumor cells tumor cells
- tumors tumor cells
- any tumors that proliferate by receptor tyrosine kinases any tumors that proliferate by aberrant serine/threonine kinase activation
- benign and malignant cells of other proliferative diseases in which aberrant serine/threonine kinase activation benign and malignant cells of other proliferative diseases in which aberrant serine/threonine kinase activation occurs.
- cancer and “cancerous” refer to or describe the physiological condition in mammals that is typically characterized by unregulated cell growth.
- a “tumor” comprises one or more cancerous cells, and/or benign or pre-cancerous cells. Examples of cancer include, but are not limited to, carcinoma, lymphoma, blastoma, sarcoma, and leukemia or lymphoid malignancies.
- cancers include squamous cell cancer (e.g., epithelial squamous cell cancer), lung cancer including small-cell lung cancer, non-small cell lung cancer (“NSCLC”), adenocarcinoma of the lung and squamous carcinoma of the lung, cancer of the peritoneum, hepatocellular cancer, gastric or stomach cancer including gastrointestinal cancer, pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, hepatoma, breast cancer, colon cancer, rectal cancer, colorectal cancer, endometrial or uterine carcinoma, salivary gland carcinoma, kidney or renal cancer, prostate cancer, vulval cancer, thyroid cancer, hepatic carcinoma, anal carcinoma, penile carcinoma, acute leukemia, head/brain and neck cancer, cancers of lymphatic organs and hematological malignancy including Leukemia (Acute lymphoblastic leukemia (ALL), Acute myelogenous leukemia (AML), Chronic
- a “therapeutic agent” encompasses both a biological agent such as an antibody, a peptide, a protein, an enzyme or a chemotherapeutic agent.
- a “chemotherapeutic agent” is a chemical compound useful in the treatment of cancer.
- chemotherapeutic agents include Erlotinib (TARCEVA®, Genentech/OSI Pharm.), Bortezomib (VELCADE®, Millennium Pharm.), Fulvestrant (FASLODEX®, AstraZeneca), Sutent (SU11248, Pfizer), Letrozole (FEMARA®, Novartis), Imatinib mesylate (GLEEVEC®, Novartis), PTK787/ZK 222584 (Novartis), Oxaliplatin (Eloxatin®, Sanofi), 5-FU (5-fluorouracil), Leucovorin, Rapamycin (Sirolimus, RAPAMUNE®, Wyeth), Lapatinib (TYKERB®, GSK572016, Glaxo Smith Kline), Lonafarnib (SCH 66336), Sorafenib (BAY43-9006, Bayer Labs), and Ge
- dynemicin including dynemicin A; bisphosphonates, such as clodronate; an esperamicin; as well as neocarzinostatin chromophore and related chromoprotein enediyne antibiotic chromophores), aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, caminomycin, carzinophilin, chromomycinis, dactinomycin, daunorubicin, detorubicin, 6-diazo-5-oxo-L-norleucine, ADRIAMYCIN® (doxorubicin), morpholino-doxorubicin, cyanomorpholino-doxorubicin, 2-pyrrolino-doxorubicin and deoxydoxorubicin), epirubicin,
- chemotherapeutic agent include: (i) anti-hormonal agents that act to regulate or inhibit hormone action on tumors such as anti-estrogens and selective estrogen receptor modulators (SERMs), including, for example, tamoxifen (including NOLVADEX®; tamoxifen citrate), raloxifene, droloxifene, 4-hydroxytamoxifen, trioxifene, keoxifene, LY117018, onapristone, and FARESTON® (toremifine citrate); (ii) aromatase inhibitors that inhibit the enzyme aromatase, which regulates estrogen production in the adrenal glands, such as, for example, 4(5)-imidazoles, aminoglutethimide, MEGASE® (megestrol acetate), AROMASIN® (exemestane; Pfizer), formestanie, fadrozole, RIVISOR® (vorozole), FEMARA® (letrozole),
- SERMs
- anti-angiogenic agents include MMP-2 (matrix-metalloproteinase 2) inhibitors, MMP-9 (matrix-metalloproteinase 9) inhibitors, COX-II (cyclooxygenase II) inhibitors, and VEGF receptor tyrosine kinase inhibitors.
- VEGF receptor tyrosine kinase inhibitors include 4-(4-bromo-2-fluoroanilino)-6-methoxy-7-(1-methylpiperidin-4-ylmethoxy)quinazoline (ZD6474; Example 2 within WO 01/32651), 4-(4-fluoro-2-methylindol-5-yloxy)-6-methoxy-7-(3-pyrrolidin-1-ylpropoxy)-quinazoline (AZD2171; Example 240 within WO 00/47212), vatalanib (PTK787; WO 98/35985) and SU11248 (sunitinib; WO 01/60814), and compounds such as those disclosed in PCT Publication Nos. WO 97/22596, WO 97/30035, WO 97/32856, and WO WO 97/32856, and WO
- chemotherapeutic agents that can be used in combination with the present compounds include inhibitors of PI3K (phosphoinositide-3 kinase), such as those reported in Yaguchi et al (2006) Jour. of the Nat. Cancer Inst. 98(8):545-556; U.S. Pat. Nos.
- PI3K phosphoinositide-3 kinase
- PI3K inhibitors include SF-1126 (PI3K inhibitor, Semafore Pharmaceuticals), BEZ-235 (PI3K inhibitor, Novartis), XL-147 (PI3K inhibitor, Exelixis, Inc.).
- Chemotherapeutic agents may also include any of the generic drugs or biosimilars of the brand-name drugs referenced herein, or improvements thereof, including improved formulations, prodrugs, delivery means (sustained release, bioadhesive coating, targeted delivery etc.), and dosage forms.
- a “metabolite” is a product produced through metabolism in the body of a specified compound, a derivative thereof, or a conjugate thereof, or salt thereof. Metabolites of a compound, a derivative thereof, or a conjugate thereof, may be identified using routine techniques known in the art and their activities determined using tests such as those described herein. Such products may result for example from the oxidation, hydroxylation, reduction, hydrolysis, amidation, deamidation, esterification, deesterification, enzymatic cleavage, and the like, of the administered compound.
- the invention includes metabolites of compounds, a derivative thereof, or a conjugate thereof, of the invention, including compounds, a derivative thereof, or a conjugate thereof, produced by a process comprising contacting a compound, a derivative thereof, or a conjugate thereof, of this invention with a mammal for a period of time sufficient to yield a metabolic product thereof.
- phrases “pharmaceutically acceptable” indicates that the substance or composition must be compatible chemically and/or toxicologically, with the other ingredients comprising a formulation, and/or the mammal being treated therewith.
- protecting group refers to a substituent that is commonly employed to block or protect a particular functionality while reacting other functional groups on the compound, a derivative thereof, or a conjugate thereof.
- an “amine-protecting group” or an “amino-protecting moiety” is a substituent attached to an amino group that blocks or protects the amino functionality in the compound.
- Such groups are well known in the art (see for example P. Wuts and T. Greene, 2007, Protective Groups in Organic Synthesis, Chapter 7, J.
- carbamates such as methyl and ethyl carbamate, FMOC, substituted ethyl carbamates, carbamates cleaved by 1,6- ⁇ -elimination (also termed “self immolative”), ureas, amides, peptides, alkyl and aryl derivatives.
- Suitable amino-protecting groups include acetyl, trifluoroacetyl, t-butoxycarbonyl (BOC), benzyloxycarbonyl (CBZ) and 9-fluorenylmethylenoxycarbonyl (Fmoc).
- leaving group refers to an group of charged or uncharged moiety that departs during a substitution or displacement.
- leaving groups include, but not limited to, halogens, esters, alkoxy, hydroxyl, tosylates, triflates, mesylates, nitriles, azide, carbamate, disulfides, thioesters, thioethers and diazonium compounds.
- bifunctional crosslinking agent refers to modifying agents that possess two reactive groups; one of which is capable of reacting with a cell binding agent while the other one reacts with the cytotoxic compound to link the two moieties together.
- bifunctional crosslinkers are well known in the art (see, for example, Isalm and Dent in Bioconjugation chapter 5, p 218-363, Groves Dictionaries Inc. New York, 1999).
- SMCC N-succinimidyl-4-(N-maleimidomethyl)-cyclohexane-1-carboxylate
- SIAB N-succinimidyl-4-(iodoacetyl)-aminobenzoate
- Other bifunctional crosslinking agents that introduce maleimido groups or haloacetyl groups on to a cell binding agent are well known in the art (see US Patent Applications 2008/0050310, 20050169933, available from Pierce Biotechnology Inc. P.O. Box 117, Rockland
- 61105 USA
- BMPEO bis-maleimidopolyethyleneglycol
- BMPS N-(3-maleimidopropyloxy)succinimide ester
- GMBS ⁇ -maleimidobutyric acid N-succinimidyl ester
- EMCS ⁇ -maleimidocaproic acid N-hydroxysuccinimide ester
- NHS 5-maleimidovaleric acid NHS
- HBVS N-succinimidyl-4-(N-maleimidomethyl)-cyclohexane-1-carboxy-(6-amidocaproate
- LC-SMCC m-maleimidobenzoyl-N-hydroxysuccinimide ester
- MCS 4-(4-N-maleimidophenyl)-butyric acid hydrazide or HCl salt
- Heterobifunctional crosslinking agents are bifunctional crosslinking agents having two different reactive groups. Heterobifunctional crosslinking agents containing both an amine-reactive N-hydroxysuccinimide group (NHS group) and a carbonyl-reactive hydrazine group can also be used to link the cytotoxic compounds described herein with a cell-binding agent (e.g., antibody). Examples of such commercially available heterobifunctional crosslinking agents include succinimidyl 6-hydrazinonicotinamide acetone hydrazone (SANH), succinimidyl 4-hydrazidoterephthalate hydrochloride (SHTH) and succinimidyl hydrazinium nicotinate hydrochloride (SHNH).
- SSH succinimidyl 6-hydrazinonicotinamide acetone hydrazone
- SHTH succinimidyl 4-hydrazidoterephthalate hydrochloride
- SHNH succinimidyl hydrazinium nicotinate hydrochlor
- Conjugates bearing an acid-labile linkage can also be prepared using a hydrazine-bearing benzodiazepine derivative of the present invention.
- bifunctional crosslinking agents include succinimidyl-p-formyl benzoate (SFB) and succinimidyl-p-formylphenoxyacetate (SFPA).
- Bifunctional crosslinking agents that enable the linkage of cell binding agent with cytotoxic compounds via disulfide bonds are known in the art and include N-succinimidyl-3-(2-pyridyldithio)propionate (SPDP), N-succinimidyl-4-(2-pyridyldithio)pentanoate (SPP), N-succinimidyl-4-(2-pyridyldithio)butanoate (SPDB), N-succinimidyl-4-(2-pyridyldithio)2-sulfo butanoate (sulfo-SPDB) to introduce dithiopyridyl groups.
- SPDP N-succinimidyl-3-(2-pyridyldithio)propionate
- SPP N-succinimidyl-4-(2-pyridyldithio)pentanoate
- SPDB N-succinimidyl
- crosslinking agents that can be used to introduce disulfide groups are known in the art and are disclosed in U.S. Pat. Nos. 6,913,748, 6,716,821 and US Patent Publications 20090274713 and 20100129314, all of which are incorporated herein by reference.
- crosslinking agents such as 2-iminothiolane, homocysteine thiolactone or S-acetylsuccinic anhydride that introduce thiol groups can also be used.
- a “linker,” “linker moiety,” or “linking group” as defined herein refers to a moiety that connects two groups, such as a cell binding agent and a cytotoxic compound, together. Typically, the linker is substantially inert under conditions for which the two groups it is connecting are linked.
- a bifunctional crosslinking agent may comprise two reactive groups, one at each ends of a linker moiety, such that one reactive group can be first reacted with the cytotoxic compound to provide a compound bearing the linker moiety and a second reactive group, which can then react with a cell binding agent.
- one end of the bifunctional crosslinking agent can be first reacted with the cell binding agent to provide a cell binding agent bearing a linker moiety and a second reactive group, which can then react with a cytotoxic compound.
- the linking moiety may contain a chemical bond that allows for the release of the cytotoxic moiety at a particular site. Suitable chemical bonds are well known in the art and include disulfide bonds, thioether bonds, acid labile bonds, photolabile bonds, peptidase labile bonds and esterase labile bonds (see for example U.S. Pat. Nos.
- linkers that can be used in the present invention include non-cleavable linkers, such as those described in are described in detail in U.S. publication number 20050169933, or charged linkers or hydrophilic linkers and are described in US 2009/0274713, US 2010/01293140 and WO 2009/134976, each of which is expressly incorporated herein by reference, each of which is expressly incorporated herein by reference.
- the linking group with a reactive group attached at one end is selected from the following:
- n, p, q, m′, n′, t′ are integer from 1 to 10, or are optionally 0;
- t, m′′, n′′, and p′′ are 0 or 1;
- Y′′ is absent or is selected from O, S, S—S or NR 32 , wherein R 32 has the same definition as given above for R; or
- X′′ is selected from a maleimido group, a haloacetyl group or SR 37 , wherein R 37 has the same definition as above;
- A′′ is an amino acid selected from glycine, alanine, leucine, valine, lysine, citrulline and glutamate or a polypeptide containing between 2 to 20 amino acid units;
- R 20 , R 21 , R 22 , R 23 , R 24 , R 25 , R 26 , and R 27 are the same or different, and are —H or a linear or branched alkyl having from 1 to 5 carbon atoms;
- R 29 and R 30 are the same or different, and are —H or alkyl from 1 to 5 carbon atoms;
- R 33 is —H or linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 12 carbon atoms, a polyethylene glycol unit R—(OCH 2 CH 2 ) n —, or R 33 is —COR 34 , —CSR 34 , —SOR 34 , or —SO 2 R 34 , wherein R 34 is H or linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 20 carbon atoms or, a polyethylene glycol unit —(OCH 2 CH 2 ); and
- R 40 and R 41 is optionally a negatively or positively charged functional group and the other is H or alkyl, alkenyl, alkynyl having 1 to 4 carbon atoms.
- linking groups may be present in any of the compounds, drug-linker compounds, or conjugates of the invention, including replacing the linking groups of any of the formulas described herein.
- amino acid refers to naturally occurring amino acids or non-naturally occurring amino acid represented by NH 2 —C(R aa′ R aa )—C( ⁇ O)OH, wherein R aa and R aa′ are each independently H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, aryl, heteroaryl or heterocyclyl.
- amino acid also refers to the corresponding residue when one hydrogen atom is removed from the amine and/or carboxy end of the amino acid, such as —NH—C(R aa′ R aa )—C( ⁇ O)O—.
- the term “cation” refers to an ion with positive charge.
- the cation can be monovalent (e.g., Na + , K + , etc.), bi-valent (e.g., Ca 2+ , Mg 2+, etc.) or multi-valent (e.g., Al 3+ etc.).
- the cation is monovalent.
- terapéuticaally effective amount means that amount of active compound or conjugate that elicits the desired biological response in a subject. Such response includes alleviation of the symptoms of the disease or disorder being treated, prevention, inhibition or a delay in the recurrence of symptom of the disease or of the disease itself, an increase in the longevity of the subject compared with the absence of the treatment, or prevention, inhibition or delay in the progression of symptom of the disease or of the disease itself. Determination of the effective amount is well within the capability of those skilled in the art, especially in light of the detailed disclosure provided herein. Toxicity and therapeutic efficacy of compound I can be determined by standard pharmaceutical procedures in cell cultures and in experimental animals.
- the effective amount of compound or conjugate of the present invention or other therapeutic agent to be administered to a subject will depend on the stage, category and status of the multiple myeloma and characteristics of the subject, such as general health, age, sex, body weight and drug tolerance.
- the effective amount of compound or conjugate of the present invention or other therapeutic agent to be administered will also depend on administration route and dosage form. Dosage amount and interval can be adjusted individually to provide plasma levels of the active compound that are sufficient to maintain desired therapeutic effects.
- the present invention is directed to cytotoxic compounds described herein (e.g., compounds of formulas (I), (II), (III), and (IV)).
- the cytotoxic compounds of the present invention do not include any compounds described in US 2010/0203007 (the entire teaching of which is incorporated herein by reference), such as those specifically disclaimed in the proviso below.
- the invention provides a cytotoxic compound comprising a linking group with a reactive group bonded thereto capable of covalently linking the cytotoxic compound to a cell binding agent (CBA), wherein said cytotoxic compound is represented by any one of the following formulas (I), (II), (III) or (IV):
- X is not the linking group with the reactive group bonded thereto.
- the double line between N and C represents a single bond, Y is not —H.
- the cytotoxic compounds of the present invention are not any one of the following compounds:
- Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R′′, —NR′R′′, —NR′COR′′, —NR′NR′R′′, an optionally substituted 5 or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C ⁇ NH)NR′R′′, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO 2 M, —SO 3 M, —OSO 3 M, halogen, cyano and an azido.
- Y is Sodium Bisulfite adduct, Sodium Hydrosulfite adduct, or
- L is absent, or is selected from an optionally substituted phenyl group and an optionally substituted pyridyl group, wherein the phenyl and the pyridyl group bears the linking group with the reactive group bonded thereto, or L is an amine group bearing the linking group with the reactive group bonded thereto (i.e., —N(linking group)-), or L is a linear, branched or cyclic alkyl or alkenyl having from 1 to 6 carbon atoms and bearing the linking group with the reactive group bonded thereto.
- Y is —H
- W is C ⁇ O
- R 1 , R 2 , R 1 ′, R 2 ′, R 4 and R 4 ′ are —H;
- R 3 is optionally a linking group and the other is —H;
- R 6 is —OMe;
- Z and Z′ are —CH 2 —;
- X′ is —H
- Y′ is —H
- a and A′ are —O—; and the remainder of the variables are as described in the first specific embodiment.
- cytotoxic dimers of formula (I), (II), (III) and (IV) are represented by the following formulas:
- X is not the linking group with the reactive group bonded thereto.
- the double line between N and C represents a single bond, Y is not —H.
- a and A′ are both —O—, R 6 is —OMe, and G is —CH—.
- L′ is represented by the formula: —W′—R x —V—R y -J,
- J is —SH, —SSR d , a maleimide, or a N-hydroxysuccinimide ester.
- R e′ is —H or -Me;
- R e is a linear or branched alkyl having 1 to 6 carbon atoms or —(CH 2 —CH 2 —O) n —R k ;
- n is an integer from 2 to 8; preferably R k is —H, -Me or —CH 2 CH 2 —NMe 2 , and the remainder of the variables are as described above in the fourth specific embodiment.
- V is an amino acid or a peptide having 2 to 8 amino acids. In certain embodiments, V is valine-citrulline, gly-gly-gly, or ala-leu-ala-leu.
- L′ is represented by the following formula: —W′—[CR 1′′ R 2′′ ] a —V—[Cy] 0-1 -[CR 3′′ R 4′′ ] b —COE,
- W′ is —N(R e )—.
- R e is —(CH 2 —CH 2 —O) 2-6 —R k , wherein R k is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms.
- V is —S— or —SS—.
- L′ is represented by the following formula: —NR e —[CR 1′′ R 2′′ ] a —S—[CR 3′′ ,R 4′′ ] b —COE.
- the compound is any of the following:
- L′ is represented by the following formula: —NR e —[CR 1′′ R 2′′ ] a —S—Cy—[CR 3′′ ,R 4′′ ] b —COE.
- the compound is any one of the following:
- cytotoxic dimers of formula (I), (II), (III) and (IV) are represented by the following formulas:
- R k is —H or -Me, and n is an integer from 2 to 8.
- R x is a linear or branched alkyl having 1 to 6 carbon atoms; and the remainder of the variables is as described above in the third, fourth, and/or the fifth specific embodiment.
- cytotoxic dimers of formula (I), (II), (III) and (IV) are represented by the following formulas:
- Z s is represented by any one of the following formulas:
- W′ is —N(R e )—.
- R e is —(CH 2 —CH 2 —O) n —R k , wherein R k is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms.
- R k is —H or -Me
- n is 4, and q is 2.
- R x is a linear or branched alkyl having 1 to 6 carbon atoms.
- R x may be —(CH 2 ) p —(CR f R g )—, wherein R f and R g are each independently selected from H or a linear or branched alkyl having 1 to 4 carbon atoms; and p is 0, 1, 2 or 3.
- R f and R g are the same or different, and are selected from —H and -Me; and p is 1.
- Y is —OH or —SO 3 M.
- R k is —H or -Me
- n is an integer from 2 to 8.
- R x is a linear or branched alkyl having 1 to 6 carbon atoms.
- R x is —(CH 2 ) p —(CR f R g )—, wherein R f and R g are each independently selected from H or a linear or branched alkyl having 1 to 4 carbon atoms; p is 0, 1, 2 or 3. More preferably, R f and R g are the same or different, and are selected from —H and -Me; and p is 1.
- the linker is represented by any one of the formula selected from formulas (a1), (a4), (a5), (a10) and (a11) shown above; and the remainder of the variables are as described above in the tenth specific embodiment.
- R x is —(CH 2 ) p —(CR f R g )—, wherein R f and R g are each independently selected from H or a linear or branched alkyl having 1 to 4 carbon atoms; p is 0, 1, 2 or 3. More preferably, R f and R g are the same or different, and are selected from —H and -Me; and p is 1.
- the double line between N and C may represent a double bond.
- the double line between N and C may represent a single bond
- X is —H, the linking group with the reactive group bonded thereto, or an amine protecting group
- Y is selected from —H, —OR, —OCOR′, —SR, —NR′R,′′ an optionally substituted 5- or 6-membered nitrogen-containing heterocycle, —SO 3 M, —SO 2 M and a sulfate —OSO 3 M (e.g., Y is —OR, —OCOR′, —SR, —NR′R,′′ an optionally substituted 5- or 6-membered nitrogen-containing heterocycle, —SO 3 M, —SO 2 M and a sulfate —OSO 3 M).
- Y is selected from —H, —SO 3 M, —OH, —OMe, —OEt or —NHOH (e.g., Y is —SO 3 M, —OH, —OMe, —OEt or —NHOH).
- Y is —H, —SO 3 M or —OH (e.g., Y is —SO 3 M or —OH).
- M is —H, Na + or K + .
- W when present, is C ⁇ O.
- Z and Z′ when present, are —CH 2 .
- X′ is selected from the group consisting of —H, —OH, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, phenyl, the linking group with the reactive group bounded thereto, and an amine-protecting group.
- X′ is —H, —OH, -Me or the linking group with the reactive group bounded thereto.
- X′ is —H.
- Y′ is selected from the group consisting of —H, an oxo group, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms.
- Y′ is —H or oxo.
- Y′ is —H.
- a and A′ are the same or different, and are selected from O, S, NR 5 and oxo (C ⁇ O).
- a and A′ may be same or different and selected from —O— and —S—.
- both A and A′ are —O—.
- D and D′ when present, are the same or different, and are independently selected from a polyethylene glycol unit (—OCH 2 CH 2 ) n , wherein n is an integer from 1 to 24, an amino acid, a peptide bearing 2 to 6 amino acids, or a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, wherein the alkyl, alkenyl and alkynyl are optionally substituted with one or more substituents independently selected from the group consisting of halogen, —OR, —NR′COR′′, —SR and —COR′.
- D and D′ are linear or branched alkyl bearing 1 to 4 carbon atoms.
- the cytotoxic compound of the present invention as described in the first, third, and ninth embodiment is represented by the following:
- a and A′ are —O—.
- the cytotoxic compound of the present invention is:
- compound 29b can used in methods of the present invention described herein. In a preferred embodiment, compound 29b can be used for treating a proliferative disorder, such as cancer.
- compound 29b can be used for screening cell lines to identify cell lines that are sensitive to benzodiazepine compounds, such as benzodiazepine derivatives described herein.
- cytotoxic compounds described above comprise a linking group with a reactive group bonded thereto, which compounds may result from reacting a bifunctional crosslinking reagent with “linker-less” compounds to form the so-called drug-linker compounds.
- linker-less compounds may result from reacting a bifunctional crosslinking reagent with “linker-less” compounds to form the so-called drug-linker compounds.
- drug compounds that are otherwise identical to the drug-linker compounds, but without the linker moiety are also encompassed by the present invention.
- the invention provides a cytotoxic compound without linking group, but may be capable of reacting with a bifunctional crosslinking agent to form a compound of the invention, such as any one of the 1 st to the 12 th specific embodiments described above; or to form a cell-binding agent conjugate of the invention (such as those described below).
- a linkerless cytotoxic compound of the invention includes compound 29b of the 13 th specific embodiment above.
- the linkerless cytotoxic compounds of the invention are represented by any one of the following formulas (I′), (II′), (III′) or (IV′):
- the double line between N and C represents a single bond
- Y is not —H.
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is selected from —H, or an amine protecting group (preferably X is —H); W is C ⁇ O; R 1 , R 2 , R 3 , R 4 , R 1′ , R 2′ , R 3′ , and R 4 ′ are —H; Z and Z′ are —CH 2 —; A and A′ are both —O—; W is —(C ⁇ O)—; G is —CH—; R 6 is —H, or optionally substituted C1-C10 linear, C1-C10 branched, or C3-C7 cyclic alkyl, —O-alkyl, or —O-haloalkyl, such as —OMe; X′ is selected from the group consisting of —H, —OH, a substituted
- Y is selected from —OR, —OCOR′, —SR, —NR′R′′, —SO 3 M, —SO 2 M, or —OSO 3 M, wherein M is —H or a cation such as Na + . K + .
- Y is selected from —H, —OH, —OMe, —OEt, —NHOH or —SO 3 M (e.g., Y is —OH, —OMe, —OEt, —NHOH or —SO 3 M).
- Y is —H, —OH or —SO 3 M (e.g., Y is —OH or —SO 3 M), preferably M is —H or Na + .
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is selected from —H, or an amine protecting group (preferably X is —H); W is C ⁇ O; R 1 , R 2 , R 3 , R 4 , R 1 , R 2′ , R 3′ , R 4′ , X′ and Y′ are —H; Z and Z′ are —CH 2 —; A and A′ are both —O—; W is —(C ⁇ O)—; G is —CH—; R 6 is —H, or optionally substituted C1-C10 linear, C1-C10 branched, or C3-C7 cyclic alkyl, —O-alkyl, or —O-haloalkyl, such as —OMe.
- the bifunctional crosslinking agents can be any bifunctional linker known in the art.
- the bifunctional linkers can be used for making the drug-linker compounds are those that form disulfide bonds, thioether bonds, acid labile bonds, photolabile bonds, peptidase labile bonds and esterase labile bonds with the cytotoxic compounds (see for example, U.S. Pat. Nos. 5,208,020; 5,475,092; 6,441,163; 6,716,821; 6,913,748; 7,276,497; 7,276,499; 7,368,565; 7,388,026 and 7,414,073, all of which are incorporated herein by reference).
- the bifunctional crosslinking agents are those that form disulfide bonds, thioether and peptidase labile bonds with the cytotoxic compounds.
- Other bifunctional crosslinking agents that can be used in the present invention include non-cleavable linkers, such as those described in U.S. publication number US 2005/0169933, or charged linkers or hydrophilic linkers and are described in US 2009/0274713, US 2010/01293140 and WO 2009/134976, each of which is expressly incorporated herein by reference.
- the bifunctional crosslinking agents that can be used for making the (drug-linker) compounds of the present invention also include those described in Thermo Scientific Pierce Crosslinking Technical Handbook, the entire teaching of which is incorporated herein by reference.
- FIGS. 1 - 11 Representative processes for preparing the cytotoxic dimer compounds of the present invention are shown in FIGS. 1 - 11 .
- the dimers were prepared by reacting a monomer with linker compounds which possess two leaving groups such as halogen, triflate, mesylate, or tosylate such as that described for the synthesis of 1c in FIG. 1 .
- Synthesis of representative dimers which bear a thiol or disulfide moiety to enable linkage to cell binding agents via reducible or non-reducible bonds are shown in FIGS. 1 - 5 , 7 , 8 , and 10 .
- a linker containing a short polyethylene glycol moiety and an alkyl disulfide was prepared through reductive amination of 1a.
- FIG. 3 a modified form of IBD monomer was prepared and coupled to give a dimer of the present invention in which the reduced imine was converted to a linker.
- FIG. 4 describes a dimer possessing a short polyethylene glycol moiety and an amide disulfide which was reduced to thiol 4c and converted to a reactive ester.
- FIGS. 1 and 12 - 19 Representative processes for preparing the cytotoxic dimer compounds of the present invention suitable for one-step conjugation with a cell binding agent are shown in FIGS. 1 and 12 - 19 .
- a dimer containing a thiol moiety is reacted with a bifunctional crosslinking reagent possessing a reactive group such as, but not limited to, a thiopyridyl, a maleimide, iodide, bromide, or tosylate on one side and a reactive substituent suitable for reaction with a cell binding agent such as, but not limited to, N-hydroxysuccinimide esters, N-hydroxyphtalimide esters, N-hydroxy sulfo-succinimide esters, paranitrophenyl esters, pentafluorophenyl esters.
- a bifunctional crosslinking reagent possessing a reactive group such as, but not limited to, a thiopyridyl, a
- FIGS. 20 - 21 Alternative synthetic processes for preparing representative cytotoxic dimer compounds of the present invention are shown in FIGS. 20 - 21 .
- the synthesis of the mono reduced dimer i.e., having one imine group
- the synthesis of the mono reduced dimer is accomplished by a two step coupling method, in which a reduced form of monomer is either initially coupled to the linker followed by coupling with the IBD monomer or the dimer is prepared using a mixture of both reduced monomer and the IBD monomer in the coupling with the reactive linker.
- the di-reduced dimer is potentially a byproduct of the second synthetic pathway previously described, a more direct route is shown in FIG. 21 in which the reduced monomer is coupled to both with the linker directly.
- Cell-binding agents may be of any kind presently known, or that become known and includes peptides and non-peptides. Generally, these can be antibodies (especially monoclonal antibodies), lymphokines, hormones, growth factors, vitamins (such as folate etc., which may bind to a cell surface receptor thereof, e.g., a folate receptor), nutrient-transport molecules (such as transferrin), or any other cell-binding molecule or substance.
- the cell-binding agents are proteins or polypeptides, or compounds comprising proteins or polypeptides.
- the proteins or polypeptides comprise one or more Lys residues with side chain —NH 2 groups.
- the proteins or polypeptides comprise one or more Cys residues.
- the side chain —SH groups of the Cys residues may be intact, or may be in a disulfide bond that can be reduced.
- reduction of the disulfide bond(s) does not significantly negatively impact the cell-binding function of the proteins or polypeptides (e.g., in the case of antibody or antigen-binding portion thereof, reduction of the disulfide bonds does not substantially increase the dissociation of light chains/heavy chains).
- the Lys side chain —NH 2 groups and/or the Cys side chain —SH groups may be covalently linked to the linkers, which are in turn linked to the dimer compounds of the invention, thus conjugating the cell-binding agents to the dimer compounds of the invention.
- Each protein-based cell-binding agents may contain multiple Lys side chain —NH 2 groups and/or the Cys side chain —SH groups available for linking the compounds of the invention through the bifunctional crosslinkers.
- cell-binding agents More specific examples of cell-binding agents that can be used include:
- interferons e.g. ⁇ , ⁇ , ⁇
- lymphokines such as IL-2, IL-3, IL-4, IL-6;
- hormones such as insulin, TRH (thyrotropin releasing hormone), MSH (melanocyte-stimulating hormone), steroid hormones, such as androgens and estrogens;
- EGF EGF
- TGF-alpha FGF
- FGF FGF
- VEGF vascular endothelial growth factor
- G-CSF G-CSF
- M-CSF M-CSF
- GM-CSF GM-CSF
- Protein scaffolds based on a consensus sequence of fibronectin type III (FN3) repeats also known as Centyrins; See U.S. Patent Publication 2010/0255056, incorporated herein by reference;
- Fibronectin domain scaffold proteins (Adnectins: US Patent Application Nos. 20070082365; 20080139791, incorporated herein by reference).
- Monoclonal antibody techniques allow for the production of extremely specific cell-binding agents in the form of specific monoclonal antibodies.
- Particularly well known in the art are techniques for creating monoclonal antibodies produced by immunizing mice, rats, hamsters or any other mammal with the antigen of interest such as the intact target cell, antigens isolated from the target cell, whole virus, attenuated whole virus, and viral proteins such as viral coat proteins.
- Sensitized human cells can also be used.
- Another method of creating monoclonal antibodies is the use of phage libraries of scFv (single chain variable region), specifically human scFv (see e.g., Griffiths et al., U.S. Pat. Nos.
- resurfaced antibodies disclosed in U.S. Pat. No. 5,639,641 may also be used, as may chimeric antibodies and humanized antibodies. Selection of the appropriate cell-binding agent is a matter of choice that depends upon the particular cell population that is to be targeted, but in general human monoclonal antibodies are preferred if an appropriate one is available.
- the monoclonal antibody MY9 is a murine IgG antibody that binds specifically to the CD33 Antigen ⁇ J. D. Griffin et al 8 Leukemia Res., 521 (1984) ⁇ and can be used if the target cells express CD33 as in the disease of acute myelogenous leukemia (AML).
- the cell-binding agent may be any compound that can bind a cell, either in a specific or non-specific manner. Generally, these can be antibodies (especially monoclonal antibodies and antibody fragments), interferons, lymphokines, hormones, growth factors, vitamins, nutrient-transport molecules (such as transferrin), or any other cell-binding molecule or substance.
- the cell-binding agent binds to an antigen that is a polypeptide and may be a transmembrane molecule (e.g. receptor) or a ligand such as a growth factor.
- antigens include molecules such as renin; a growth hormone, including human growth hormone and bovine growth hormone; growth hormone releasing factor; parathyroid hormone; thyroid stimulating hormone; lipoproteins; alpha-1-antitrypsin; insulin A-chain; insulin B-chain; proinsulin; follicle stimulating hormone; calcitonin; luteinizing hormone; glucagon; clotting factors such as factor vmc, factor IX, tissue factor (TF), and von Willebrands factor; anti-clotting factors such as Protein C; atrial natriuretic factor; lung surfactant; a plasminogen activator, such as urokinase or human urine or tissue-type plasminogen activator (t-PA); bombesin; thrombin; hemo
- GM-CSF which binds to myeloid cells can be used as a cell-binding agent to diseased cells from acute myelogenous leukemia.
- IL-2 which binds to activated T-cells can be used for prevention of transplant graft rejection, for therapy and prevention of graft-versus-host disease, and for treatment of acute T-cell leukemia.
- MSH which binds to melanocytes, can be used for the treatment of melanoma, as can antibodies directed towards melanomas.
- Folic acid can be used to target the folate receptor expressed on ovarian and other tumors.
- Epidermal growth factor can be used to target squamous cancers such as lung and head and neck.
- Somatostatin can be used to target neuroblastomas and other tumor types.
- Cancers of the breast and testes can be successfully targeted with estrogen (or estrogen analogues) or androgen (or androgen analogues) respectively as cell-binding agents.
- the cell-binding agent is humanized monoclonal antibodies.
- the cell-binding agent is huMy9-6, or other related antibodies, which are described in U.S. Pat. Nos. 7,342,110 and 7,557,189 (incorporated herein by reference).
- the cell-binding agent is an anti-folate receptor antibody described in U.S. Provisional Application Nos. 61/307,797, 61/346,595, 61/413,172 and U.S. application Ser. No. 13/033,723 (published as US 2012-0009181 A1). The teachings of all these applications are incorporated herein by reference in its entirety.
- the cell-binding agent may be a monoclonal antibody or antigen-binding portions thereof sharing sequences critical for antigen-binding with an antibody disclosed herein, such as huMy9-6 or its related antibodies described in U.S. Pat. Nos. 7,342,110 and 7,557,189 (incorporated herein by reference).
- These derivative antibodies may have substantially the same or identical (1) light chain and/or heavy chain CDR3 regions; (2) light chain and/or heavy chain CDR1, CDR2, and CDR3 regions; or (3) light chain and/or heavy chain regions, compared to an antibody described herein. Sequences within these regions may contain conservative amino acid substitutions, including substitutions within the CDR regions. Preferably, there is no more than 1, 2, 3, 4, or 5 conservative substitutions.
- the derivative antibodies have a light chain region and/or a heavy chain region that is at least about 90%, 95%, 99% or 100% identical to an antibody described herein.
- These derivative antibodies may have substantially the same binding specificity and/or affinity to the target antigen compared to an antibody described herein.
- the K d and/or k off values of the derivative antibodies are within 10-fold (either higher or lower), 5-fold (either higher or lower), 3-fold (either higher or lower), or 2-fold (either higher or lower) of an antibody described herein.
- These derivative antibodies may be fully human antibodies, or humanized antibodies, or chimeric antibodies.
- the derivative antibodies may be produced according to any art-recognized methods.
- the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof that specifically binds a human folate receptor 1, wherein the antibody comprises: (a) a heavy chain CDR1 comprising GYFMN (SEQ ID NO: 1); a heavy chain CDR2 comprising RIHPYDGDTFYNQX aa1 FX aa2 X aa3 (SEQ ID NO: 2); and a heavy chain CDR3 comprising YDGSRAMDY (SEQ ID NO: 3); and (b) a light chain CDR1 comprising KASQSVSFAGTSLMH (SEQ ID NO: 4); a light chain CDR2 comprising RASNLEA (SEQ ID NO: 5); and a light chain CDR3 comprising QQSREYPYT (SEQ ID NO: 6); wherein Xaa 1 is selected from K, Q, H, and R; Xaa 2 is selected from Q, H, N, and R; and Xaa 3 is
- the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof that specifically binds the human folate receptor 1 comprising the heavy chain having the amino acid sequence of QVQLVQSGAEVVKPGASVKISCKASGYTFTGYFMNWVKQSPGQSLEWIGRIHPY DGDTFYNQKFQGKATLTVDKSSNTAHMELLSLTSEDFAVYYCTRYDGSRAMDY WGQGTTVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNS GALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKV EPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDP EVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKV SNKALPAPIEKTISKAKGQPREP
- the anti-folate antibody is a humanized antibody or antigen binding fragment thereof encoded by the plasmid DNA deposited with the ATCC on Apr. 7, 2010 and having ATCC deposit nos. PTA-10772 and PTA-10773 or 10774.
- the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof that specifically binds the human folate receptor 1 comprising the light chain having the amino acid sequence of DIVLTQSPLSLAVSLGQPAIISCKASQSVSFAGTSLMHWYHQKPGQQPRLLIYRAS NLEAGVPDRFSGSGSKTDFTLNISPVEAEDAATYYCQQSREYPYTFGGGTKLEIK RTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQE SVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC (SEQ ID NO: 9); or DIVLTQSPLSLAVSLGQPAIISCKASQSVSFAGTSLMHWYHQKPGQQPRLLIYRAS NLEAGVPDRFSGSGSKTDFTLTISPVEAEDAATYYCQQSREYPYTFGGGTKLEI
- the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof that specifically binds the human folate receptor 1 comprising the heavy chain having the amino acid sequence of SEQ ID NO: 8, and the light chain having the amino acid sequence of SEQ ID NO: 9 or SEQ ID NO: 10.
- the antibody comprises the heavy chain having the amino acid sequence of SEQ ID NO: 8 and the light chain having the amino acid sequence of SEQ ID NO: 10 (hu FOLR1).
- the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof encoded by the plasmid DNA deposited with the ATCC on Apr. 7, 2010 and having ATCC deposit nos. PTA-10772 and PTA-10773 or 10774.
- the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof comprising a heavy chain variable domain at least about 90%, 95%, 99% or 100% identical to QVQLVQSGAEVVKPGASVKISCKASGYTFTGYFMNWVKQSPGQSLEWIGRIHPY DGDTFYNQKFQGKATLTVDKSSNTAHMELLSLTSEDFAVYYCTRYDGSRAMDY WGQGTTVTVSS (SEQ ID NO: 11), and a light chain variable domain at least about 90%, 95%, 99% or 100% identical to DIVLTQSPLSLAVSLGQPAIISCKASQSVSFAGTSLMHWYHQKPGQQQPRLLIYRAS NLEAGVPDRFSGSGSKTDFTLNISPVEAEDAATYYCQQSREYPYTFGGGTKLEIK R (SEQ ID NO: 12); orDIVLTQSPLSLAVSLGQPAIISCKASQSVSFAGTSLMHWYHQK
- the present invention also provides cell-binding agent-drug conjugates comprising a cell-binding agent linked to one or more cytotoxic compounds of the present invention via a variety of linkers, including, but not limited to, disulfide linkers, thioether linkers, amide bonded linkers, peptidase-labile linkers, acid-labile linkers, esterase-labile linkers.
- linkers including, but not limited to, disulfide linkers, thioether linkers, amide bonded linkers, peptidase-labile linkers, acid-labile linkers, esterase-labile linkers.
- Representative conjugates of the invention are antibody/cytotoxic compound, antibody fragment/cytotoxic compound, epidermal growth factor (EGF)/cytotoxic compound, melanocyte stimulating hormone (MSH)/cytotoxic compound, thyroid stimulating hormone (TSH)/cytotoxic compound, somatostatin/cytotoxic compound, folate/cytotoxic compound, estrogen/cytotoxic compound, estrogen analogue/cytotoxic compound, androgen/cytotoxic compound, and androgen analogue/cytotoxic compound.
- a representative folate/cytotoxic compound conjugate is depicted below, with the optional —SO 3 —Na adduct on the imine bond of one of the two drug monomers.
- a representative synthesis scheme for this conjugate is shown in FIG. 54 .
- the present invention provides conjugates comprising an indolinobenzodiazepine dimer compound (e.g., compounds of formulas (I)-(IV), (IA)-(IVA) and (IB)-(IVB)) and the cell-binding agent linked through a covalent bond.
- the linker can be cleaved at the site of the tumor/unwanted proliferating cells to deliver the cytotoxic agent to its target in a number of ways.
- the linker can be cleaved, for example, by low pH (hydrazone), reductive environment (disulfide), proteolysis (amide/peptide link), or through an enzymatic reaction (esterase/glycosidase).
- representative cytotoxic conjugates of the invention are antibody/indolinobenzodiazepine dimer compound, antibody fragment/indolinobenzodiazepine dimer compound, epidermal growth factor (EGF)/indolinobenzodiazepine dimer compound, melanocyte stimulating hormone (MSH)/indolinobenzodiazepine dimer compound, thyroid stimulating hormone (TSH)/indolinobenzodiazepine dimer compound, somatostatin/indolinobenzodiazepine dimer compound, folate/indolinobenzodiazepine dimer compound, estrogen/indolinobenzodiazepine dimer compound, estrogen analogue/indolinobenzodiazepine dimer compound, prostate specific membrane antigen (PSMA) inhibitor/indolinobenzodiazepine dimer compound, matriptase inhibitor/indolinobenzodiazepine dimer compound, designed ankyrin repeat proteins (DARPins)/indolinobenzodiazepine dimer compound, and
- the invention provides a conjugate comprising: a cytotoxic compound and a cell binding agent (CBA), wherein the cytotoxic compound comprises a linking group which covalently links the cytotoxic compound to the CBA, and wherein the cytotoxic compound is represented by any one of the following formulas:
- X is not the linking group.
- the double line between N and C represents a single bond, Y is not —H.
- Y is a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R′′, —NR′R′′, —NR′COR′′, —NR′NR′R′′, an optionally substituted 5 or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C ⁇ NH)NR′R′′, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO 2 M, —SO 3 M, —OSO 3 M, halogen, cyano and an azido.
- P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO 2 M
- the compound is not any one of the following compounds:
- conjugates of the invention include the following:
- L is absent, or is selected from an optionally substituted phenyl group and an optionally substituted pyridyl group, wherein the phenyl and the pyridyl group bears the linking group, or L is an amine group bearing the linking group (i.e., —N(linking group)-), or L is a linear, branched or cyclic alkyl or alkenyl having from 1 to 6 carbon atoms and bearing the linking group.
- the compound is represented by any one of the following formulas:
- one of L′, L′′, or L′′′ is the linking group, while the others are —H.
- L′ is the linking group
- L′′ and L′′′ are —H.
- a and A′ are both —O—, R 6 is —OMe, and G is —CH—.
- L′ is represented by the following formula: —W′—R x —V—R y -J,
- J is —S—, —SS—, a succinimide, or —C( ⁇ O)—.
- R e is —H or -Me; R e is a linear or branched alkyl having 1 to 6 carbon atoms or —(CH 2 —CH 2 —O) n —R k ; n is an integer from 2 to 8; and R k is —H, -Me or —CH 2 CH 2 —NMe 2 , and the remainder of the variables are as described above in the fifteenth specific embodiment.
- V is an amino acid or a peptide having 2 to 8 amino acids.
- V is valine-citrulline, gly-gly-gly, or ala-leu-ala-leu.
- L′ in the sixteenth specific embodiment is represented by the following formula: —W′—[CR 1′′ R 2′′ ] a —V—[Cy] 0-1 —[CR 3′′ R 4′′ ] b —C( ⁇ O)—,
- W′ is —N(R e )—.
- R e is —(CH 2 —CH 2 —O) 2-6 —R k , wherein R k is a linear or branched alkyl having 1 to 6 carbon atoms.
- V is —S— or —SS—.
- L′ in the sixteenth or the seventeenth specific embodiment is represented by the following formula: —NR e —[CR 1′′ R 2′′ ] a —S—[CR 3′′ R 4′′ ] b —C( ⁇ O)—.
- the conjugate is:
- the antibody is huMy9-6.
- L′ in the sixteenth or the seventeenth specific embodiment is represented by the following formula: —NR e —[CR 1′′ R 2′′ ] a -S—Cy—[CR 3′′ R 4′′ ] b —C( ⁇ O)—.
- the conjugate is:
- the antibody is huMy9-6.
- the compound is represented by the following formula:
- the compound is represented by the following formula:
- Z s is represented by any one of the following formulas:
- W′ is —N(R e )—.
- R e is —(CH 2 —CH 2 —O) n —R k , wherein R k is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms.
- R k is —H or -Me
- n is 4, and q is 2.
- R x is a linear or branched alkyl having 1 to 6 carbon atoms.
- R x is —(CH 2 ) p —(CR f R g )—, wherein R f and R g are each independently selected from H or a linear or branched alkyl having 1 to 4 carbon atoms; and p is 0, 1, 2 or 3.
- R f and R g are the same or different, and are selected from —H and -Me; and p is 1.
- R x is —(CH 2 ) p —(CR f R g )—, wherein R f and R g are each independently selected from —H or a linear or branched alkyl having 1 to 4 carbon atoms; p is 0, 1, 2 or 3. More preferably, R f and R g are the same or different, and are selected from —H and -Me; and p is 1.
- the double line between N and C may represent a double bond.
- the double line between N and C may represent a single bond
- X is —H, the linking group, or an amine protecting group (e.g., X is —H);
- Y is —H or selected from —OR, —OCOR′, —SR, —NR′R′′, an optionally substituted 5- or 6-membered nitrogen-containing heterocycle, —SO 3 M, —SO 2 M and a sulfate —OSO 3 M.
- Y is not —H.
- Y is selected from —H, —SO 3 M, —OH, —OMe, —OEt or —NHOH (e.g., Y is —SO 3 M, —OH, —OMe, —OEt or —NHOH).
- Y is —H, —SO 3 M or —OH (e.g., Y is —SO 3 M or —OH).
- M is —H, Na + or K + .
- W when present, is C ⁇ O.
- Z and Z′ when present, are —CH 2 —.
- X′ is selected from the group consisting of —H, —OH, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, phenyl, the linking group, and an amine-protecting group.
- X′ is —H, —OH, -Me or the linking group.
- X′ is —H.
- Y′ is selected from the group consisting of —H, an oxo group, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms.
- Y′ is —H or oxo.
- Y′ is —H.
- a and A′ are the same or different, and are selected from —O—, —S—, —N(R 5 )—, and oxo (C ⁇ O).
- a and A′ are the same or different, and are selected from —O— and —S—.
- a and A′ are —O—.
- D and D′ when present, are the same or different, and are independently selected from a polyethylene glycol unit (—OCH 2 CH 2 ) n , wherein n is an integer from 1 to 24, an amino acid, a peptide bearing 2 to 6 amino acids, or a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, wherein the alkyl, alkenyl and alkynyl are optionally substituted with one or more substituents independently selected from the group consisting of halogen, —OR, —NR′COR′′, —SR and —COR′.
- D and D′ are linear or branched alkyl bearing 1 to 4 carbon atoms.
- the conjugate of the present invention as described in the fourteenth, fifteenth, or the twenty-first specific embodiment is represented by the following:
- the conjugate of any one of the described embodiments may comprise 1-10 cytotoxic compounds, 2-9 cytotoxic compounds, 3-8 cytotoxic compounds, 4-7 cytotoxic compounds, or 5-6 cytotoxic compounds, each cytotoxic compound comprising the linking group linking the cytotoxic compound to the CBA, and each cytotoxic compound on the conjugate is the same.
- the cell-binding agent may bind to target cells selected from tumor cells, virus infected cells, microorganism infected cells, parasite infected cells, autoimmune cells, activated cells, myeloid cells, activated T-cells, B cells, or melanocytes; cells expressing the CD4, CD6, CD19, CD20, CD22, CD30, CD33, CD37, CD38, CD40, CD44, CD56, EpCAM, CanAg, CALLA, or Her-2 antigens; Her-3 antigens; or cells expressing insulin growth factor receptor, epidermal growth factor receptor, and folate receptor.
- target cells selected from tumor cells, virus infected cells, microorganism infected cells, parasite infected cells, autoimmune cells, activated cells, myeloid cells, activated T-cells, B cells, or melanocytes
- the cell-binding agent may be an antibody, a single chain antibody, an antibody fragment that specifically binds to the target cell, a monoclonal antibody, a single chain monoclonal antibody, or a monoclonal antibody fragment that specifically binds to a target cell, a chimeric antibody, a chimeric antibody fragment that specifically binds to the target cell, a domain antibody, a domain antibody fragment that specifically binds to the target cell, a lymphokine, a hormone, a vitamin, a growth factor, a colony stimulating factor, or a nutrient-transport molecule.
- the antibody may be a resurfaced antibody, a resurfaced single chain antibody, or a resurfaced antibody fragment.
- the antibody may be a monoclonal antibody, a single chain monoclonal antibody, or a monoclonal antibody fragment thereof.
- the antibody may be a humanized antibody, a humanized single chain antibody, or a humanized antibody fragment.
- the invention further provides a pharmaceutical composition comprising any of the conjugates described herein, and a pharmaceutically acceptable carrier.
- the invention further provides a drug-linker compound comprising any of the subject compound covalently linked to a bifunctional linker.
- the invention additional provides a conjugate comprising any of the subject compounds, or the subject drug-linker compounds, linked to a cell-binding agent.
- the invention further provides a method of inhibiting abnormal cell growth or treating a proliferative disorder, an autoimmune disorder, destructive bone disorder, infectious disease, viral disease, fibrotic disease, neurodegenerative disorder, pancreatitis or kidney disease in a mammal comprising administering to the mammal a therapeutically effective amount of any of the compounds (with or without any linker group) or conjugates of the invention, and, optionally, a second chemotherapeutic agent.
- the compound or the conjugate is:
- the second chemotherapeutic agent is administered to the mammal sequentially or consecutively.
- the method is for treating a condition selected from cancer, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, lupus, myositis, infection, and immune deficiency.
- a condition selected from cancer, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, lupus, myositis, infection, and immune deficiency.
- the method or conjugate is for treating a cancer.
- the cancer is selected from breast cancer, colon cancer, brain cancer, prostate cancer, kidney cancer, pancreatic cancer, ovarian cancer, head and neck cancer, melanoma, colorectal cancer, gastric cancer, squamous cancer, small-cell lung cancer, non small-cell lung cancer, testicular cancer, Merkel cell carcinoma, glioblastoma, neuroblastoma, cancers of lymphatic organs and hematological malignancy including Leukemia (Acute lymphoblastic leukemia (ALL), Acute myelogenous leukemia (AML), Chronic lymphocytic leukemia (CLL), Chronic myelogenous leukemia (CML), Acute monocytic leukemia (AMOL), Hairy cell leukemia (HCL), T-cell prolymphocytic leukemia (T-PLL), Large granular lymphocytic leukemia, Adult T-cell leukemia), Lymphoma (small lymphocytic lymphoma (SLL), Hodg
- ALL
- the cytotoxic compound may comprise a linking moiety with a reactive group bonded thereto.
- a bifunctional crosslinking reagent can be first reacted with the cytotoxic compound to provide the compound bearing a linking moiety with one reactive group bonded thereto (i.e., drug-linker compound), which can then react with a cell binding agent.
- one end of the bifunctional crosslinking reagent can first react with the cell binding agent to provide the cell binding agent bearing a linking moiety with one reactive group bonded thereto, which can then react with a cytotoxic compound.
- the linking moiety may contain a chemical bond that allows for the release of the cytotoxic moiety at a particular site.
- Suitable chemical bonds are well known in the art and include disulfide bonds, thioether bonds, acid labile bonds, photolabile bonds, peptidase labile bonds and esterase labile bonds (see for example U.S. Pat. Nos. 5,208,020; 5,475,092; 6,441,163; 6,716,821; 6,913,748; 7,276,497; 7,276,499; 7,368,565; 7,388,026 and 7,414,073).
- Preferred are disulfide bonds, thioether and peptidase labile bonds.
- the compounds of formula (I)-(IV), (IA)-(IVA), and (IB)-(IVB) can be linked through R 1 , R 2 , R 3 , R 4 , R 1 ′, R 2 ′, R 3 ′, R 4 ′, L′, L′′, L′′′, or X (when present).
- preferred linkable groups are R 2 ′, R 3 ′, R 4 ′, L′, L′′, L′′′ and most preferred linkable groups are R 2 ′, R 3 ′, and L′. Examples of linking groups for compounds of formula (I)-(IV), (IA)-(IVA), and (IB)-(IVB) are described above.
- a solution of an antibody in aqueous buffer may be incubated with a molar excess of an antibody modifying agent such as N-succinimidyl-3-(2-pyridyldithio)propionate (SPDP) or with N-succinimidyl-4-(2-pyridyldithio)butanoate (SPDB) to introduce dithiopyridyl groups.
- an antibody modifying agent such as N-succinimidyl-3-(2-pyridyldithio)propionate (SPDP) or with N-succinimidyl-4-(2-pyridyldithio)butanoate (SPDB) to introduce dithiopyridyl groups.
- SPDP N-succinimidyl-3-(2-pyridyldithio)propionate
- SPDB N-succinimidyl-4-(2-pyridyldithio)butanoate
- the cell binding agent-drug conjugate may then be purified using any purification methods known in the art, such as those described in U.S. Pat. No. 7,811,572 and US Publication No. 2006/0182750, both of which are incorporated herein by reference.
- the cell-binding agent-drug conjugate can be purified using tangential flow filtration, adsorptive chromatography, adsorptive filtration, selective precipitation, non-absorptive filtration or combination thereof.
- tangential flow filtration also known as cross flow filtration, ultrafiltration and diafiltration
- adsorptive chromatography resins are used for the purification of the conjugates.
- the antibody may be incubated with a molar excess of an antibody modifying agent such as 2-iminothiolane, L-homocysteine thiolactone (or derivatives), or N-succinimidyl-S-acetylthioacetate (SATA) to introduce sulfhydryl groups.
- an antibody modifying agent such as 2-iminothiolane, L-homocysteine thiolactone (or derivatives), or N-succinimidyl-S-acetylthioacetate (SATA) to introduce sulfhydryl groups.
- the modified antibody is then reacted with the appropriate disulfide-containing cytotoxic agent, to produce a disulfide-linked antibody-cytotoxic agent conjugate.
- the antibody-cytotoxic agent conjugate may then be purified by methods described above.
- the cell binding may also be engineered to introduce thiol moieties, such as cysteine-engineered antibodies disclosed in U.S. Pat.
- a solution of an antibody in aqueous buffer may be incubated with a molar excess of an antibody-modifying agent such as N-succinimidyl-4-(N-maleimidomethyl)-cyclohexane-1-carboxylate to introduce maleimido groups, or with N-succinimidyl-4-(iodoacetyl)-aminobenzoate (SIAB) to introduce iodoacetyl groups.
- the modified antibody is then reacted with the thiol-containing cytotoxic agent to produce a thioether-linked antibody-cytotoxic conjugate.
- the antibody-cytotoxic conjugate may then be purified by methods described above.
- the number of cytotoxic molecules bound per antibody molecule can be determined spectrophotometrically by measuring the ratio of the absorbance at 280 nm and 330 nm.
- An average of 1-10 cytotoxic compounds/antibody molecule(s) can be linked by the methods described herein.
- the preferred average number of linked cytotoxic compounds per antibody molecule is 2-5, and the most preferred is 2.5-4.0.
- Cytotoxic agents containing linkers terminating in an N-hydroxy succinimidyl (NHS) ester can react with the antibody to produce direct amide linked conjugates such as huMy9-6-SPDB-1f or huMy9-6-BMPS-1f.
- the antibody-cytotoxic agent conjugate may then be purified by gel-filtration by any methods described above.
- FIGS. 22 and 23 Representative processes for preparing the cell-binding agent-drug conjugates of the present invention are shown in FIGS. 22 and 23 .
- a cytotoxic dimer compound of the present invention can be conjugated with a cell binding agent through either a one-step or a two-step conjugation method.
- FIGS. 22 a and 22 b representative examples are described, wherein a dimer compound that possesses a linker such as an N-hydroxysuccinimide ester is reacted directly with a cell binding agent, such as an antibody, generating the desired conjugate.
- linkable dimer 1g was first treated with sodium bisulfate to provide a modified dimer compound 26 before adding antibody to form the conjugate huMy9-6-SBDP-1f of the present invention.
- FIG. 23 A representative example of a two-step conjugation method is described in FIG. 23 , wherein an antibody is first modified with a bifunctional crosslinking agent resulting in an antibody that possesses a desired number of linkers suitable for reaction with a dimer compound having a free thiol moiety.
- the antibody huMy9-6 was first modified with SPDB to give an antibody with linkers containing the dithiopyridyl moiety. The modified antibody was then exposed to a free thiol, such as 2a, generating the desired conjugate huMy9-6-SPDB-2a.
- the cytotoxic compounds and cell-binding agent-drug conjugates of the invention can be evaluated for their ability to suppress proliferation of various cancer cell lines in vitro.
- cell lines such as the human colon carcinoma line COLO 205, the rhabdomyosarcoma cell line RH-30, and the multiple myeloma cell line MOLP-8 can be used for the assessment of cytotoxicity of these compounds and conjugates.
- Cells to be evaluated can be exposed to the compounds or conjugates for 1-5 days and the surviving fractions of cells measured in direct assays by known methods. IC 50 values can then be calculated from the results of the assays.
- an in vitro cell line sensitivity screen such as the one described by the U.S.
- FIGS. 25 - 26 Examples of in vitro potency and target specificity of antibody-cytotoxic agent conjugates of the present invention are shown in FIGS. 25 - 26 . All of the conjugates are extremely cytotoxic on the antigen positive cancer cells with an IC 50 in the low picomolar range. Antigen negative cell lines remained viable when exposed to the same conjugates. The indolinobenzodiazepine dimers showed target specific potency being 160 fold less potent when blocked with unconjugated antibody huMy9-6 (anti-CD33) and 40 less potent when blocked with unconjugated antibody FOLR1 (anti-folate receptor antibody).
- the huMy9-6-SPDB-1f conjugate is also highly potent towards both the HL60/ATCC cell line with an IC 50 value of 21 pM and the NB-4 cell line with an IC 50 value of 190 pM ( FIGS. 25 B and 25 C ).
- the huFOLR1-SPDB-1f conjugate was highly potent, with an IC 50 value of 55 pM for antigen positive KB cells ( FIG. 26 ). Addition of an excess of unconjugated huFOLR1 antibody reduced this cytotoxic effect >40 fold, demonstrating antigen-specificity.
- in vivo efficacy of a cell binding agent/cytotoxic agent conjugate was measured.
- Nude mice bearing human HL60/QC tumors were treated with huMy9-6-SPDB-1f conjugate and significant tumor regression was observed at multiple doses while untreated mice grew tumors rapidly ( FIG. 28 ).
- Activity was observed at doses as low as 20 ⁇ g/kg which is at least 35-fold lower than the maximum tolerated dose.
- the present invention includes a composition (e.g., a pharmaceutical composition) comprising novel benzodiazepine compounds described herein (e.g., indolinobenzodiazepine or oxazolidinobenzodiazepine), derivatives thereof, or conjugates thereof, (and/or solvates, hydrates and/or salts thereof) and a carrier (a pharmaceutically acceptable carrier).
- a composition e.g., a pharmaceutical composition
- a carrier e.g., a pharmaceutical composition
- the present compositions are useful for inhibiting abnormal cell growth or treating a proliferative disorder in a mammal (e.g., human).
- the present compositions are also useful for treating depression, anxiety, stress, phobias, panic, dysphoria, psychiatric disorders, pain, and inflammatory diseases in a mammal (e.g., human).
- the present invention includes a method of inhibiting abnormal cell growth or treating a proliferative disorder in a mammal (e.g., human) comprising administering to said mammal a therapeutically effective amount of novel benzodiazepine compounds described herein (e.g., indolinobenzodiazepine or oxazolidinobenzodiazepine), derivatives thereof, or conjugates thereof, (and/or solvates and salts thereof) or a composition thereof, alone or in combination with a second therapeutic agent.
- novel benzodiazepine compounds described herein e.g., indolinobenzodiazepine or oxazolidinobenzodiazepine
- derivatives thereof or conjugates thereof, (and/or solvates and salts thereof) or a composition thereof, alone or in combination with a second therapeutic agent.
- the present invention provides a method for inducing cell death in selected cell populations comprising contacting target cells or tissue containing target cells with an effective amount of a cytotoxic agent comprising any of the cytotoxic compound-cell-binding agents (e.g., indolinobenzodiazepine or oxazolidinobenzodiazepine dimer linked to a cell binding agent) of the present invention, a salt or solvate thereof.
- a cytotoxic agent comprising any of the cytotoxic compound-cell-binding agents (e.g., indolinobenzodiazepine or oxazolidinobenzodiazepine dimer linked to a cell binding agent) of the present invention, a salt or solvate thereof.
- the target cells are cells to which the cell-binding agent can bind.
- other active agents such as other anti-tumor agents, may be administered along with the conjugate.
- Suitable pharmaceutically acceptable carriers, diluents, and excipients are well known and can be determined by those of ordinary skill in the art as the clinical situation warrants.
- Suitable carriers, diluents and/or excipients include: (1) Dulbecco's phosphate buffered saline, pH about 7.4, containing or not containing about 1 mg/mL to 25 mg/mL human serum albumin, (2) 0.9% saline (0.9% w/v NaCl), and (3) 5% (w/v) dextrose; and may also contain an antioxidant such as tryptamine and a stabilizing agent such as Tween 20.
- the method for inducing cell death in selected cell populations can be practiced in vitro, in vivo, or ex vivo.
- Examples of in vitro uses include treatments of autologous bone marrow prior to their transplant into the same patient in order to kill diseased or malignant cells: treatments of bone marrow prior to their transplantation in order to kill competent T cells and prevent graft-versus-host-disease (GVHD); treatments of cell cultures in order to kill all cells except for desired variants that do not express the target antigen; or to kill variants that express undesired antigen.
- treatments of autologous bone marrow prior to their transplant into the same patient in order to kill diseased or malignant cells treatments of bone marrow prior to their transplantation in order to kill competent T cells and prevent graft-versus-host-disease (GVHD); treatments of cell cultures in order to kill all cells except for desired variants that do not express the target antigen; or to kill variants that express undesired antigen.
- GVHD graft-versus-host-disease
- Examples of clinical ex vivo use are to remove tumor cells or lymphoid cells from bone marrow prior to autologous transplantation in cancer treatment or in treatment of autoimmune disease, or to remove T cells and other lymphoid cells from autologous or allogenic bone marrow or tissue prior to transplant in order to prevent GVHD.
- Treatment can be carried out as follows. Bone marrow is harvested from the patient or other individual and then incubated in medium containing serum to which is added the cytotoxic agent of the invention, concentrations range from about 10 ⁇ M to 1 pM, for about 30 minutes to about 48 hours at about 37° C. The exact conditions of concentration and time of incubation, i.e., the dose, are readily determined by one of ordinary skill in the art.
- the bone marrow cells After incubation the bone marrow cells are washed with medium containing serum and returned to the patient intravenously according to known methods. In circumstances where the patient receives other treatment such as a course of ablative chemotherapy or total-body irradiation between the time of harvest of the marrow and reinfusion of the treated cells, the treated marrow cells are stored frozen in liquid nitrogen using standard medical equipment.
- the cytotoxic agent of the invention will be supplied as a solution or a lyophilized powder that are tested for sterility and for endotoxin levels.
- suitable protocols of conjugate administration are as follows. Conjugates are given weekly for 4 weeks as an intravenous bolus each week. Bolus doses are given in 50 to 1000 mL of normal saline to which 5 to 10 mL of human serum albumin can be added. Dosages will be 10 ⁇ g to 2000 mg per administration, intravenously (range of 100 ng to 20 mg/kg per day). After four weeks of treatment, the patient can continue to receive treatment on a weekly basis. Specific clinical protocols with regard to route of administration, excipients, diluents, dosages, times, etc., can be determined by one of ordinary skill in the art as the clinical situation warrants.
- Examples of medical conditions that can be treated according to the in vivo or ex vivo methods of inducing cell death in selected cell populations include malignancy of any type including, for example, cancer of the lung (small cell and non-small cell), breast, colon, brain, prostate, kidney, pancreas, ovary, head and neck, skin (melanoma), Merkel cell carcinoma, glioblastoma, neuroblastoma, and cancers of lymphatic organs; autoimmune diseases, such as systemic lupus, rheumatoid arthritis, and multiple sclerosis; graft rejections, such as renal transplant rejection, liver transplant rejection, lung transplant rejection, cardiac transplant rejection, and bone marrow transplant rejection; graft versus host disease; viral infections, such as CMV infection, HIV infection, AIDS, etc.; and parasite infections, such as giardiasis, amoebiasis, schistosomiasis, and others as determined by one of ordinary skill in the art.
- PDR Physician's Desk Reference
- the PDR discloses dosages of the agents that have been used in treatment of various cancers.
- the dosing regimen and dosages of these aforementioned chemotherapeutic drugs that are therapeutically effective will depend on the particular cancer being treated, the extent of the disease and other factors familiar to the physician of skill in the art and can be determined by the physician.
- the contents of the PDR are expressly incorporated herein in its entirety by reference.
- One of skill in the art can review the PDR, using one or more of the following parameters, to determine dosing regimen and dosages of the chemotherapeutic agents and conjugates that can be used in accordance with the teachings of this invention. These parameters include:
- cytotoxic agents include analogues and derivatives of the compounds described herein.
- the organic layer was dried over anhydrous sodium sulfate, filtered, concentrated and high vacuumed to give the mesylates as light yellowish oil (340 mg).
- the mesylates was transferred into a 10 mL round-bottomed flask with ethyl acetate/dichloromethane, concentrated and high vacuumed.
- IBD monomer (412 mg, 1.4 mmol) was added followed by the addition of anhydrous dimethylformamide (3 mL) and anhydrous potassium carbonate (232 mg, 1.68 mmol).
- the obtained yellowish mixture was stirred at room temperature overnight. It was diluted with dichloromethane and washed with brine.
- the organic layer was dried over anhydrous sodium sulfate, filtered and concentrated.
- the organic extracts were washed with ice water, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give the dimesylate.
- Compound 1g was pre-treated with 3 molar equivalents of sodium bisulfite (using a freshly prepared NaHSO 3 solution in water) in 96-98% DMA in water for 4-5 hrs at 25° C.
- the humanized antibody at 2 mg/mL was reacted with 5-7 molar equivalents of compound 1g (pre-treated with NaHSO 3 ) for 6 h at 25° C. in 85-90% PBS, pH 7.4, aqueous buffer, or 50 mM HEPES, pH 8.5, aqueous buffer, containing 10-15% N,N-dimethylacetamide (DMA) and then purified over a G25 gel filtration column in PBS, pH 7.4, to remove unreacted or hydrolyzed drug compound.
- DMA N,N-dimethylacetamide
- the humanized antibody-SPDB-drug conjugates were dialyzed in 10 mM Histidine, 250 mM Glycine, 1% sucrose, pH 6.5 buffer.
- the Drug Antibody Ratio (DAR) of the conjugates were measured to be 2.2-2.9 by UV absorbance measurements at 280 and 320 nm and using the extinction coefficients of the drug and antibody at 280 nm (215,000 M ⁇ 1 cm ⁇ 1 ) and 320 nm (9137 M ⁇ 1 cm ⁇ 1 ).
- the percentage of monomer in the conjugates were determined as >90% by SEC (Size Exclusion Chromatography) using TSK-Gel G300SWXL column (7.8 mm ⁇ 300 mm, 5 ⁇ m particle size).
- the monomer conjugate peaks had linked drug molecules.
- the conjugate was acetone extracted to remove protein, dried, and reconstituted in mobile phase and injected onto a VYDAC 208TP C8 reverse phase HPLC column (4.6 ⁇ 250 mm, 7 ⁇ m particle size) and compared to standards. The percentage of free drug compound in the conjugate was determined as ⁇ 0.5% of conjugated drug compound. See FIG. 22 .
- Humanized Ab at 8 mg/mL was derivatized with 4-6 molar equivalents of SPDB hetrobifunctional linker for 1.5 h at 25° C. in 95% PBS, PH 7.4, containing 5% DMA (v/v), and then purified over a G25 desalting column into citrate buffer (35 mM citrate buffer, pH 5.5, containing 2 mM EDTA, 150 mM NaCl) to remove unreacted linker.
- citrate buffer 35 mM citrate buffer, pH 5.5, containing 2 mM EDTA, 150 mM NaCl
- the LAR Linker Antibody Ratio
- the SPDB-modified antibody at 2 mg/mL was reacted with 2 molar equivalents of compound 2a (HCl salt) per linked SPDB for 20 h at ambient temperature in 85% citrate buffer, 15% DMA (v/v) and then purified over a G25 desalting column into PBS, pH 7.4 to remove unconjugated drug compound.
- the DAR of the final humanized Ab-SPDB-2a conjugate was measured by UV spectrophotometry at 280 and 350 nm and calculated to be ⁇ 1.7-2.1 DAR.
- the percentage of monomer and linked drug compound on the monomer in the conjugate was determined by HPLC using an SEC (size exclusion chromatography) column. See FIG. 23 .
- COLO 205 1,000 cells/well
- Namalwa 3,000 cells/well
- HEL 92.1.7 3,000 cells/well
- RH30 1,000 cells/well
- HL60/QC 5,000 cells/well
- Ramos 10,000 cells/well
- KB 2,000 cells/well
- BJAB 2,000 cells/well
- NB4 3,000 cells/well
- RPMI 8226 8,000 cells/well
- cytotoxic potencies were then assessed using a WST-8 based cell viability assay and surviving cells were measured by developing with WST-8 (2-7 hours). The absorbance in each well was measured and the surviving fraction of cells at each concentration was plotted to reveal the cytotoxicity and/or antigen specificity (of the conjugates).
- the cytotoxicity of the unconjugated free drug compounds was measured against seven cell lines: KB, a HeLa cell contaminant, HL60/QC, an acute myeloid leukemia cell line, Namalwa, a Burkitt lymphoma cell line, NB4, an acute promyelocytic leukemia cell line, HEL92.1.7, an erythroleukemia cell line, RPMI8226, a multiple myeloma cell line and BJAB, a B-cell leukemia cell line.
- the results, shown in FIG. 24 and Table 10 demonstrate the high potency of these compounds across a wide range of cell types.
- the potency and specificity of the antibody-drug conjugates were measured against antigen-expressing cells, with and without the additions of an excess amount of blocking unconjugated antibody to show specificity of the killing effect.
- the MY9-6-drug conjugate was extremely potent towards three different antigen-expressing cells: HL60/ATCC, HL60/QC and NB-4, despite the very low antigen expression in NB4 cells.
- the specific potency could be blocked by addition of excess unconjugated antibody, demonstrating that the cell killing effect is antigen-specific.
- the huFOLR1-drug conjugate was effective in killing antigen-expressing KB cells in a specific manner. Results are illustrated in FIGS. 25 and 26 .
- FIG. 34 huMy9-6-SPDB-1f, huMy9-6-sulfoSPDB-1f, and huMy9-6-BMPS-1f against HL60/QC (Ag + ) cells
- FIG. 34 chB38.1-SPDB-1f and chB38.1-sulfoSPDB-1f against COLO205 (Ag + ) cells
- FIG. 35 huMy9-6-SPDB-1f, huMy9-6-sulfoSPDB-1f, and huMy9-6-BMPS-1f against OCI-AML3 (Ag + ) cells
- FIG. 44 Also see FIG. 49 for the potency of the various conjugates against various cell lines, expressed as IC 50 values (nM). Note that in FIGS. 25 , 29 , 34 , 35 , and 44 , conjugagtes were prepared in the presence of sodium bisulfite.
- the binding of huMY9-6-drug conjugate was compared with that of the unmodified huMY9-6 antibody against antigen-expressing HL60/QC cells using flow cytometry. Briefly, the antigen-positive cells were incubated with conjugates or unmodified antibodies at 4° C., then with a secondary antibody-FITC conjugate at 4° C., fixed with formaldehyde (1% in PBS) and analyzed by flow cytometry. No significant difference was observed between the binding of the conjugate versus that of the unmodified antibody. An example is shown in FIG. 27 , where a huMY9-6-drug conjugate bound to antigen-positive cells with a high affinity similar to that of the unmodified antibody.
- Treatment was initiated the day of randomization, and groups included a control group dosed with PBS (200 ⁇ L/injection), or a single treatment at various doses (5 to 100 ⁇ g/kg) of huMY9-6-SPDB-1f (50 ⁇ g/kg 1f dose corresponded to 2.5 mg/kg antibody dose). All treatments were well tolerated with the mean body weight losses comparable to loss seen in PBS control mice. Mean tumor volume vs time is shown ( FIGS. 28 and 36 ) with the data demonstrating a dose-dependent anti-tumor activity of the huMY9-6-SPDB-1f conjugate. The minimum effective dose was estimated to be 20 ⁇ g/kg, which is about 35-fold lower that the maximum tolerated dose.
- huFOLR-1 conjugates were investigated in female CD-1 mice. Animals were observed for seven days prior to study initiation and found to be free of disease or illness. The mice were administered a single i.v. injection of the conjugate and the animals were monitored daily for body weight loss, morbidity or mortality. Table 9 shows that for huFOLR1-drug1 the conjugate was tolerated at only the lowest dose tested of 50 ⁇ g/kg. In contrast, both mono-imine conjugates huFOLR1-Drug 2 and huFOLR1-SPDB-1f were found to be better tolerated with a maximum tolerated dose of ⁇ 198 ⁇ g/kg and >560 ⁇ g/kg respectively.
- the conjugate was purified and buffer exchanged into 250 mM Glycine, 10 mM Histidine, 1% sucrose, 0.01% Tween, 50 ⁇ M sodium bisulfite formulation buffer, using NAP desalting columns (Illustra Sephadex G-25 DNA Grade, GE Healthcare). Dialysis was performed in the same buffer for 4 hours at room temperature utilizing Slide-a-Lyzer dialysis cassettes (ThermoScientific 20,000 MWCO).
- reaction containing 2.0 mg/mL huMy9-6 antibody and 5 molar equivalents of 12 pretreated with 5-fold excess of sodium bisulfite in 90:10 DMA:water
- 50 mM HEPES (4-(2-hydroxyethyl)-1-piperazine ethanesulfonic acid) pH 8.5 buffer and 15% v/v DMA (N,N-dimethylacetamide) cosolvent was allowed to react for 6 hours at 25° C.
- the conjugate was purified and buffer exchanged into 250 mM Glycine, 10 mM Histidine, 1% sucrose, 0.01% Tween, 50 ⁇ M sodium bisulfite formulation buffer, using NAP desalting columns (Illustra Sephadex G-25 DNA Grade, GE Healthcare). Dialysis was performed in the same buffer for 4 hours at room temperature utilizing Slide-a-Lyzer dialysis cassettes (ThermoScientific 20,000 MWCO).
- Treatment was initiated the day after randomization, and groups included a control group dosed with PBS (200 ⁇ L/injection), or a single treatment at various doses (20 to 200 ⁇ g/kg) of hu FOLR1-SPDB-1f (50 mg/kg linked drug dose corresponded to 2.8 mg/kg antibody dose). All treatments were well tolerated with no body weight loss seen in any of the test groups. Mean tumor volume vs time is shown ( FIG. 37 ) with the data demonstrating a dose-dependent anti-tumor activity of the hu FOLR1-SPDB-1f conjugate. The minimum effective dose was estimated to be ⁇ 50 ⁇ g/kg, which is about 14-fold lower than the maximum tolerated dose.
- huMy9-6-Drug 2 were formulated with or without 50 ⁇ M sodium bisulfite, and the conjugates were used to treat mice bearing HL60-QC tumor xenografts.
- the data below shows that conjugate formulated with or without 50 ⁇ M sodium bisulfite showed comparable T/C % at ⁇ 20 ⁇ g/kg drug dose, indicating that the inclusion of sodium bisulfite in the conjugate preparation step does not negatively impact the in vivo potency of the subject conjugate.
- (5-((2-mercapto-2-methylpropylthio)methyl)-1,3-phenylene)dimethanol was dissolved in methanol (3 mL) in a small vial and a stir bar was added. To this solution was added triethylamine (0.016 mL, 0.118 mmol) followed by 2,2-dimethylthiirane (0.058 mL, 0.590 mmol) and the resulting mixture was capped and stirred overnight (16 hrs) at room temperature.
- IBD Monomer (0.060 g, 0.204 mmol) was dissolved in acetone (4 ml) in a small vial, a stir bar was added, followed by 3,5-bis(iodomethyl)-N-(2-(2-(2-methoxyethoxy)ethoxy)ethyl)-N-(2-methyl-2-(methyldisulfanyl)propyl)aniline (0.167 g, 0.255 mmol) and potassium carbonate (0.070 g, 0.510 mmol). The vial was capped and stirred at room temperature overnight. The solids were filtered off and the filtrate was concentrated.
- mice The tolerability of huMy9-6 conjugates was investigated in female CD-1 mice. Animals were observed for seven days prior to study initiation and found to be free of disease or illness. The mice were administered a single i.v. injection of the conjugate and the animals were monitored daily for body weight loss, morbidity or mortality. Table 10 shows that the huMy9-6-SPDB-1c di-imine disulfide containing conjugate was tolerated at a dose of less than 300 ⁇ g/kg.
- the mono-imine disulfide conjugates huMy9-6-SPDB-1f and huMy9-6-sulfo-SPDB-1f were found to be better tolerated with a maximum tolerated dose of >729 ⁇ g/kg and ⁇ 750 ⁇ g/kg respectively.
- HEPES 4-(2-hydroxyethyl)-1-piperazine ethanesulfonic acid
- the conjugate was purified and buffer exchanged into 250 mM Glycine, 10 mM Histidine, 1% sucrose, 0.01% Tween-20, 50 ⁇ M sodium bisulfite formulation buffer, pH 6.2, using NAP desalting columns (Illustra Sephadex G-25 DNA Grade, GE Healthcare). Dialysis was performed in the same buffer for 4 hours at room temperature utilizing Slide-a-Lyzer dialysis cassettes (ThermoScientific 20,000 MWCO).
- nude or SCID immune-compromised mice
- nude rats may be employed.
- the conjugates to be tested include any one or more of the conjugates described herein.
- the various tumor cell lines that can be used for inoculating the nude mice included HL60/QC, MOLM-13, NB4, HEL92.1.7, OCI-AML3, KB, and/or any other cancer cell lines recognized in the art as a proper model for a disease indication (e.g., cancer).
- Some criteria that may be applied for the selection of tumor cell lines suitable for in vivo evaluation include: a) expression of the target antigen on the tumor cell, and, b) sensitivity of tumor cells to the unconjugated drug in vitro.
- an in vitro cell line sensitivity screen such as the 60-cell line screen described by the U.S. National Cancer Institute (see Voskoglou-Nomikos et al., 2003, Clinical Cancer Res. 9; 42227-4239, incorporated herein by reference) can be used as one of the guides to determine the types of cancers that may be suitable for treatment with the compounds of the invention.
- the potency of the various conjugates against the various tumor cell lines, as expressed by IC 50 values (nM) is measured accordingly.
- the various tumor cell lines are inoculated to nude or SCID mice using substantially the same protocol as outlined in Example 15.
- about 1 ⁇ 10 6 -5 ⁇ 10 7 tumor cells (typically 1 ⁇ 10 7 ) cells/mouse are subcutaneously inoculated at a volume of approximately 0.1-0.2 mL/mouse, in the area over the right shoulder of female athymic nude mice, 6 weeks of age.
- Treatment is initiated the day after randomization, and groups includes a control group dosed with the appropriate vehicle (200 ⁇ L/injection), or a single treatment at various doses (5 to 700 ⁇ g/kg) of the above referenced drug conjugates (50 ⁇ g/kg linked drug dose corresponded to about 2 mg/kg antibody dose). Multiple dosing schedules (for example treatment at day 1, 3, 5, or day 1, 4, 7) may also be employed.
- Linker modified huMy9-6 was diluted to 2 mg/mL in 50 mM HEPES buffer at pH 8.5, 10% v/v DMA, and reacted with 2 molar equivalents of compound 1d per linker (5 mM stock in DMA; 4.6 equivalents per antibody) for 30 min at 25° C. Completion of disulfide exchange reaction was determined by monitoring absorbance increase at 394 nm by UV.
- the conjugate was purified and buffer exchanged into 250 mM glycine, 10 mM histidine, 1% sucrose, 0.01% Tween-20, 50 ⁇ M sodium bisulfite at pH 6.2 using a desalting column (G-25 Sephadex, fine grade, GE Healthcare).
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Immunology (AREA)
- Cell Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Oncology (AREA)
- Hematology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Biochemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Indole Compounds (AREA)
Abstract
The invention relates to novel benzodiazepine derivatives with antiproliferative activity and more specifically to novel benzodiazepine compounds of formula (I)-(VII). The invention also provides conjugates of the benzodiazepine compounds linked to a cell-binding agent. The invention further provides compositions and methods useful for inhibiting abnormal cell growth or treating a proliferative disorder in a mammal using the compounds or conjugates of the invention.
Description
This application is a reissue application of U.S. Pat. No. 10,570,212 issued on Feb. 25, 2020, which is a continuation of U.S. patent application Ser. No. 15/645,597, filed Jul. 10, 2017 and now U.S. Pat. No. 10,179,818; which is a continuation of U.S. patent application Ser. No. 15/221,255, filed Jul. 27, 2016 and now U.S. Pat. No. 9,840,564; which is a continuation of U.S. patent application Ser. No. 14/849,270, filed Sep. 9, 2015 and now U.S. Pat. No. 9,434,748; which is a continuation of U.S. patent application Ser. No. 14/512,059, filed Oct. 10, 2014 and now U.S. Pat. No. 9,169,272; which is a continuation of U.S. patent application Ser. No. 13/827,355, filed Mar. 14, 2013 and now U.S. Pat. No. 8,889,669; which is a continuation of U.S. patent application Ser. No. 13/397,195, filed Feb. 15, 2012 and now U.S. Pat. No. 8,765,740; which claims the benefit of priority from U.S. Provisional Application No. 61/483,499, filed on May 6, 2011, U.S. Provisional Application No. 61/443,092, filed on Feb. 15, 2011; and U.S. Provisional Application No. 61/443,022, filed on Feb. 15, 2011. The entire contents of each of the above-referenced applications, including all drawings, formulae, sequence listings, specifications, and claims, are incorporated herein by reference.
The present invention relates to novel cytotoxic compounds, and cytotoxic conjugates comprising these cytotoxic compounds and cell-binding agents. More specifically, this invention relates to novel benzodiazepine compounds derivatives thereof, intermediates thereof, conjugates thereof, and pharmaceutically acceptable salts thereof, which are useful as medicaments, in particular as anti-proliferative agents.
Benzodiazepine derivatives are useful compounds for treating various disorders, and include medicaments such as, antiepileptics (imidazo [2,1-b][1,3,5]benzothiadiazepines, U.S. Pat. Nos. 4,444,688; 4,062,852), antibacterials (pyrimido[1,2-c][1,3,5]benzothiadiazepines, GB 1476684), diuretics and hypotensives (pyrrolo(1,2-b)[1,2,5] benzothiadiazepine 5,5 dioxide, U.S. Pat. No. 3,506,646), hypolipidemics (WO 03091232), anti-depressants (U.S. Pat. No. 3,453,266); osteoporosis (JP 2138272).
Recently, it has been shown in animal tumor models that benzodiazepine derivatives, such as pyrrolobenzodiazepines (PBDs), act as anti-tumor agents (N-2-imidazolyl alkyl substituted 1,2,5-benzothiadiazepine-1,1-dioxide, U.S. Pat. No. 6,156,746), benzo-pyrido or dipyrido thiadiazepine (WO 2004/069843), pyrrolo [1,2-b][1,2,5] benzothiadiazepines and pyrrolo[1,2-b][1,2,5] benzodiazepine derivatives (WO2007/015280), tomaymycin derivatives (e.g., pyrrolo[1,4]benzodiazepines), such as those described in WO 00/12508, WO2005/085260, WO2007/085930, and EP 2019104. Benzodiazepines are also known to affect cell growth and differentiation (Kamal A., et al., Bioorg Med Chem. 2008 Aug. 15; 16(16):7804-10 (and references cited therein); Kumar R, Mini Rev Med Chem. 2003 June; 3(4):323-39 (and references cited therein); Bednarski J J, et al., 2004; Sutter A. P, et al., 2002; Blatt N B, et al., 2002), Kamal A. et al., Current Med. Chem., 2002; 2; 215-254, Wang J-J., J. Med. Chem., 2206; 49:1442-1449, Alley M. C. et al., Cancer Res. 2004; 64:6700-6706, Pepper C. J., Cancer Res 2004; 74:6750-6755, Thurston D. E. and Bose D. S., Chem Rev 1994; 94:433-465; and Tozuka, Z., et al., Journal of Antibiotics, (1983) 36; 1699-1708. General structure of PBDs is described in US Publication Number 20070072846. The PBDs differ in the number, type and position of substituents, in both their aromatic A rings and pyrrolo C rings, and in the degree of saturation of the C ring. Their ability to form an adduct in the minor groove and crosslink DNA enables them to interfere with DNA processing, hence their potential for use as antiproliferative agents.
The first pyrrolobenzodiazepine to enter the clinic, SJG-136 (NSC 694501) is a potent cytotoxic agent that causes DNA inter-strand crosslinks (S. G Gregson et al., 2001, J. Med. Chem., 44: 737-748; M. C. Alley et al., 2004, Cancer Res., 64: 6700-6706; J. A. Hartley et al., 2004, Cancer Res., 64: 6693-6699; C. Martin et al., 2005, Biochemistry., 44: 4135-4147; S. Arnould et al., 2006, Mol. Cancer Ther., 5: 1602-1509). Results from a Phase I clinical evaluation of SJG-136 revealed that this drug was toxic at extremely low doses (maximum tolerated dose of 45 μg/m2, and several adverse effects were noted, including vascular leak syndrome, peripheral edema, liver toxicity and fatigue. DNA damage was noted at all doses in circulating lymphocytes (D. Hochhauser et al., 2009, Clin. Cancer Res., 15: 2140-2147). Thus, there exists a need for improved benzodiazepine derivatives that are less toxic and still therapeutically active for treating a variety of proliferative disease states, such as cancer.
Cytotoxic benzodiazepine dimers disclosed in the art possess two imine functionalities in their free form or reversibly protected form, such as a hydrate, alkoxylate or sulfonate. The presence of these two imine functionalities results in crosslinking of DNA (S. G. Gregson et al., 2001, J. Med. Chem., 44: 737-748). The present invention is partly based on the unexpected finding that cell binding agent conjugates of new cytotoxic benzodiazepine derivatives, such as indolinobenzodiazapene dimers that are devoid of two imine functionalities (e.g., one imine functionality and one amine functionality), and thus incapable of crosslinking DNA, display a much higher therapeutic index (ratio of maximum tolerated dose to minimum effective dose) in vivo compared to benzodiazepine derivatives that can crosslink DNA that are previously disclosed in the art.
Thus one object of the invention is to provide cytotoxic compound comprising a linking group with a reactive group bonded thereto capable of covalently linking the cytotoxic compound to a cell binding agent (CBA, see below), wherein the cytotoxic compound is represented by any one of the following formulas:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, or a linear or branched alkyl having 1 to 4 carbon atoms, and when it is a single bond, X is —H, the linking group with the reactive group bonded thereto, or an amine protecting moiety;
- Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5 or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido; or,
- Y is a sulfite (HSO3, HSO2 or a salt of HSO3 −, SO3 2− or HSO2 − formed with a cation), metabisulfite (H2S2O5 or a salt of S2O5 2− formed with a cation), mono-, di-, tri-, and tetra-thiophosphate (PO3SH3, PO2S2H2, POS3H2, PS4H2 or a salt of PO3S3 −, PO2S2 3−, POS3 3− or PS4 3− formed with a cation), thio phosphate ester (RiO)2PS(ORi), RiS—, RiSO, RiSO2, RiSO3, thiosulfate (HS2O3 or a salt of S2O3 2− formed with a cation), dithionite (HS2O4 or a salt of S2O4 2− formed with a cation), phosphorodithioate (P(═S)(ORk′)(S)(OH) or a salt thereof formed with a cation), hydroxamic acid (Rk′C(═O)NOH or a salt formed with a cation), formaldehyde sulfoxylate (HOCH2SO2 − or a salt of HOCH2SO2 − formed with a cation, such as HOCH2SO2 −Na+) or a mixture thereof, wherein Ri is a linear or branched alkyl having 1 to 10 carbon atoms and is substituted with at least one substituent selected from —N(Rj)2, —CO2H, —SO3H, and —PO3H; Ri can be further optionally substituted with a substituent for an alkyl described herein; Rj is a linear or branched alkyl having 1 to 6 carbon atoms; Rk′ is a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, aryl, heterocyclyl or heteroaryl; preferably, Y is an adduct of a bisulfite, a hydrosulfite, or a metabisulfite, or salts thereof (such as sodium salt);
- M is —H or a cation;
- R, for each occurrence, is independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P;
- R′ and R″ are each independently selected from —H, —OH, —OR, —NHR, —NR2, —COR, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, and an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms independently selected from O, S, N and P;
- Rc is —H or a substituted or unsubstituted linear or branched alkyl having 1 to 4 carbon atoms, or the linking group with the reactive group bonded thereto;
- n is an integer from 1 to 24;
- W is selected from C═O, C═S, CH2, BH, SO and SO2;
- X′ is selected from —H, an amine-protecting group, the linking group with the reactive group bonded thereto, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, and an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P;
- Y′ is selected from —H, an oxo group, the linking group with the reactive group bonded thereto, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, an optionally substituted 6- to 18-membered aryl, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, an optionally substituted 3 to 18-membered heterocyclic ring having 1 to 6 heteroatoms;
- R1, R2, R3, R4, R1′, R2′, R3′ and R4′ are each independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—Rc, halogen, guanidinium [—NH(C═NH)NH2], —OR, —NR′R″, —NO2, —NCO, —NR′COR″, —SR, a sulfoxide represented by —SOR′, a sulfone represented by —SO2R′, a sulfonate —SO3 −M+, a sulfate —OSO3 −M+, a sulfonamide represented by —SO2NR′R″, cyano, an azido, —COR′, —OCOR′, —OCONR′R″ and the linking group with the reactive group bonded thereto;
- R6 is —H, —R, —OR, —SR, —NR′R″, —NO2, halogen or the linking group with the reactive group bonded thereto;
- Z and Z′ are independently selected from —(CH2)n′—, —(CH2)n′—CR7R8—(CH2)na′—, —(CH2)n′—NR9—(CH2)na′—, —(CH2)n′—O—(CH2)na′— and —(CH2)n′—S—(CH2)na′—;
- n′ and na′ are the same or different, and are selected from 0, 1, 2 and 3;
- R7 and R8 are the same or different, and are each independently selected from —H, —OH, —SH, —COOH, —NHR′, a polyethylene glycol unit —(OCH2CH2)n—, an amino acid, a peptide unit bearing 2 to 6 amino acids, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms;
- R9 is independently selected from —H, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—;
- A and A′ are the same or different, and are independently selected from —O—, oxo (—C(═O)—), —CRR′O—, —CRR′—, —S—, —CRR′S—, —NR5 and —CRR′N(R5)—;
- R5 for each occurrence is independently —H or an optionally substituted linear or branched alkyl having 1 to 10 carbon atoms;
- D and D′ are the same or different, and are independently absent or selected from the group consisting of an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, an amino acid, a peptide bearing 2 to 6 amino acids, and a polyethylene glycol unit (—OCH2CH2)n—;
- L is absent, the linking group with the reactive group bonded thereto, a polyethylene glycol unit (—OCH2CH2)n—, a linear, branched or cyclic alkyl or alkenyl having 1 to 10 carbon atoms, a phenyl group, a 3 to 18-membered heterocyclic ring or a 5- to 18-membered heteroaryl ring having 1 to 6 heteroatoms independently selected from O, S, N and P, wherein the alkyl or alkenyl is optionally substituted with the linking group with the reactive group bonded thereto; phenyl or heterocyclic or heteroaryl ring can be optionally substituted, wherein the substituent can be the linking group with the reactive group bonded thereto.
In certain embodiments, X is not the linking group with the reactive group bonded thereto. In certain embodiments, the double line between N and C represents a single bond, Y is not —H.
In certain embodiments, the compound is not any one of the following compounds:
In certain embodiments, Y is a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5- or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido.
A second object of the invention is to provide conjugates of cell binding agents with the novel benzodiazepine compounds or derivatives thereof of the present invention. These conjugates are useful as therapeutic agents, which are delivered specifically to target cells and are cytotoxic.
Specifically, a conjugate of the invention may comprise: a cytotoxic compound and a cell binding agent (CBA), wherein the cytotoxic compound comprises a linking group which covalently links the cytotoxic compound to the CBA, and wherein the cytotoxic compound is represented by any one of the following formulas:
or a pharmaceutically acceptable salt thereof, wherein:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, or a linear or branched alkyl having 1 to 4 carbon atoms, and when it is a single bond, X is —H, the linking group, or an amine protecting moiety;
- Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5 or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido; or,
- Y is a sulfite (HSO3, HSO2 or a salt of HSO3 −, SO3 2− or HSO2 − formed with a cation), metabisulfite (H2S2O5 or a salt of S2O5 2− formed with a cation), mono-, di-, tri-, and tetra-thiophosphate (PO3SH3, PO2S2H2, POS3H2, PS4H2 or a salt of PO3S3−, PO2S2 3−, POS3 3− or PS4 3− formed with a cation), thio phosphate ester (RiO)2PS(ORi), RiS—, RiSO, RiSO2, RiSO3, thiosulfate (HS2O3 or a salt of S2O3 2− formed with a cation), dithionite (HS2O4 or a salt of S2O4 2− formed with a cation), phosphorodithioate (P(═S)(ORk′)(S)(OH) or a salt thereof formed with a cation), hydroxamic acid (Rk′C(═O)NOH or a salt formed with a cation), formaldehyde sulfoxylate (HOCH2SO2 − or a salt of HOCH2SO2 − formed with a cation, such as HOCH2SO2 −Na+) or a mixture thereof, wherein Ri is a linear or branched alkyl having 1 to 10 carbon atoms and is substituted with at least one substituent selected from —N(Rj)2, —CO2H, —SO3H, and —PO3H; Ri can be further optionally substituted with a substituent for an alkyl described herein; Rj is a linear or branched alkyl having 1 to 6 carbon atoms; Rk′ is a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, aryl, heterocyclyl or heteroaryl; preferably, Y is an adduct of a bisulfite, a hydrosulfite, or a metabisulfite, or salts thereof (such as sodium salt);
- M is —H or a cation;
- R, for each occurrence, is independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P;
- R′ and R″ are each independently selected from —H, —OH, —OR, —NHR, —NR2, —COR, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, and an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms independently selected from O, S, N and P;
- Rc is —H or a substituted or unsubstituted linear or branched alkyl having 1 to 4 carbon atoms, or the linking group;
- n is an integer from 1 to 24;
- W is selected from C═O, C═S, CH2, BH, SO and SO2;
- X′ is selected from —H, an amine-protecting group, the linking group, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, and an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P;
- Y′ is selected from —H, an oxo group, the linking group, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, an optionally substituted 6- to 18-membered aryl, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, an optionally substituted 3 to 18-membered heterocyclic ring having 1 to 6 heteroatoms;
- R1, R2, R3, R4, R1′, R2′, R3′ and R4′ are each independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—Rc, halogen, guanidinium [—NH(C═NH)NH2], —OR, —NR′R″, —NO2, —NCO, —NR′COR″, —SR, a sulfoxide represented by —SOR′, a sulfone represented by —SO2R′, a sulfonate —SO3 −M+, a sulfate —OSO3 −M+, a sulfonamide represented by —SO2NR′R″, cyano, an azido, —COR′, —OCOR′, —OCONR′R″ and the linking group;
- R6 is —H, —R, —OR, —SR, —NR′R″, —NO2, halogen or the linking group;
- Z and Z′ are independently selected from —(CH2)n′—, —(CH2)n′—CR7R8—(CH2)na′—, —(CH2)n′—NR9—(CH2)na′—, —(CH2)n′—O—(CH2)na′— and —(CH2)n′—S—(CH2)na′—;
- n′ and na′ are the same or different, and are selected from 0, 1, 2 and 3;
- R7 and R8 are same or different, and are each independently selected from —H, —OH, —SH, —COOH, —NHR′, a polyethylene glycol unit —(OCH2CH2)n—, an amino acid, a peptide unit bearing 2 to 6 amino acids, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms;
- R9 is independently selected from —H, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—;
- A and A′ are the same or different, and are independently selected from —O—, oxo (—C(═O)—), —CRR′O—, —CRR′—, —S—, —CRR′S—, —NR5 and —CRR′N(R5)—,
- R5 for each occurrence is independently —H or an optionally substituted linear or branched alkyl having 1 to 10 carbon atoms;
- D and D′ are the same or different, and are independently absent or selected from the group consisting of an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, an amino acid, a peptide bearing 2 to 6 amino acids, and a polyethylene glycol unit (—OCH2CH2)n—;
- L is absent, the linking group, a polyethylene glycol unit (—OCH2CH2)n—, a linear, branched or cyclic alkyl or alkenyl having 1 to 10 carbon atoms, a phenyl group, a 3- to 18-membered heterocyclic ring or a 5- to 18-membered heteroaryl ring having 1 to 6 heteroatoms independently selected from O, S, N and P, wherein the alkyl or alkenyl is optionally substituted with the linking group; phenyl or heterocyclic or heteroaryl ring can be optionally substituted, wherein the substituent can be the linking group.
In certain embodiments, X is not the linking group. In certain embodiments, the double line between N and C represents a single bond, Y is not —H.
In certain embodiments, Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5- or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido. In certain embodiments, Y is not —H.
In certain embodiments, the compound of the conjugate is not any one of the following compounds (the wavy bond represents the bond through which the compound
The present invention also includes a composition (e.g., a pharmaceutical composition) comprising novel benzodiazepine compounds, derivatives thereof, or conjugates thereof, (and/or solvates, hydrates and/or salts thereof) and a carrier (a pharmaceutically acceptable carrier). The present invention additionally includes a composition (e.g., a pharmaceutical composition) comprising novel benzodiazepine compounds, derivatives thereof, or conjugates thereof (and/or solvates, hydrates and/or salts thereof), and a carrier (a pharmaceutically acceptable carrier), further comprising a second therapeutic agent. The present compositions are useful for inhibiting abnormal cell growth or treating a proliferative disorder in a mammal (e.g., human). The present compositions are useful for treating conditions such as cancer, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, lupus, myositis, infection, immune deficiency such as AIDS, and inflammatory diseases in a mammal (e.g., human).
The present invention includes a method of inhibiting abnormal cell growth or treating a proliferative disorder in a mammal (e.g., human) comprising administering to said mammal a therapeutically effective amount of novel benzodiazepine compounds, derivatives thereof, or conjugates thereof, (and/or solvates and salts thereof) or a composition thereof, alone or in combination with a second therapeutic agent. The present invention includes a method of synthesizing and using novel benzodiazepine compounds, derivatives thereof, and conjugates thereof for in vitro, in situ, and in vivo diagnosis or treatment of mammalian cells, organisms, or associated pathological conditions.
The compounds of this invention, derivatives thereof, or conjugates thereof, and compositions comprising them, are useful for treating or lessening the severity of disorders, such as, characterized by abnormal growth of cells (e.g., cancer). Other applications for compounds and conjugates of this invention include, but are not limited to, treating conditions such as cancer, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, lupus, myositis, infection, immune deficiency such as AIDS and inflammatory diseases in a mammal (e.g., human).
As used herein, when referring to a group (e.g., Rc, L, X′ etc.) “is/be” (or “is not”) the linking group or the linking group with the reactive group bounded thereto, it is meant that the group “comprises” (or “does not comprise”) the linking group or the linking group with the reactive group bounded thereto.
Reference will now be made in detail to certain embodiments of the invention, examples of which are illustrated in the accompanying structures and formulas. While the invention will be described in conjunction with the enumerated embodiments, it will be understood that they are not intended to limit the invention to those embodiments. On the contrary, the invention is intended to cover all alternatives, modifications, and equivalents which may be included within the scope of the present invention as defined by the claims. One skilled in the art will recognize many methods and materials similar or equivalent to those described herein, which could be used in the practice of the present invention.
It should be understood that any of the embodiments described herein, including those described under different aspects of the invention (e.g., compounds, compound-linker molecules, conjugates, compositions, methods of making and using) and different parts of the specification (including embodiments described only in the Examples) can be combined with one or more other embodiments of the invention, unless explicitly disclaimed or improper. Combination of embodiments are not limited to those specific combinations claimed via the multiple dependent claims.
“Linear or branched alkyl” as used herein refers to a saturated linear or branched-chain monovalent hydrocarbon radical of one to twenty carbon atoms. Examples of alkyl include, but are not limited to, methyl, ethyl, 1-propyl, 2-propyl, 1-butyl, 2-methyl-1-propyl, —CH2CH(CH3)2), 2-butyl, 2-methyl-2-propyl, 1-pentyl, 2-pentyl 3-pentyl, 2-methyl-2-butyl, 3-methyl-2-butyl, 3-methyl-1-butyl, 2-methyl-1-butyl, 1-hexyl), 2-hexyl, 3-hexyl, 2-methyl-2-pentyl, 3-methyl-2-pentyl, 4-methyl-2-pentyl, 3-methyl-3-pentyl, 2-methyl-3-pentyl, 2,3-dimethyl-2-butyl, 3,3-dimethyl-2-butyl, 1-heptyl, 1-octyl, and the like. Preferably, the alkyl has one to ten carbon atoms. More preferably, the alkyl has one to four carbon atoms.
“Linear or branched alkenyl” refers to linear or branched-chain monovalent hydrocarbon radical of two to twenty carbon atoms with at least one site of unsaturation, i.e., a carbon-carbon, double bond, wherein the alkenyl radical includes radicals having “cis” and “trans” orientations, or alternatively, “E” and “Z” orientations. Examples include, but are not limited to, ethylenyl or vinyl (—CH═CH2), allyl (—CH2CH═CH2), and the like. Preferably, the alkenyl has two to ten carbon atoms. More preferably, the alkyl has two to four carbon atoms.
“Linear or branched alkynyl” refers to a linear or branched monovalent hydrocarbon radical of two to twenty carbon atoms with at least one site of unsaturation, i.e., a carbon-carbon, triple bond. Examples include, but are not limited to, ethynyl, propynyl, 1-butynyl, 2-butynyl, 1-pentynyl, 2-pentynyl, 3-pentynyl, hexynyl, and the like. Preferably, the alkynyl has two to ten carbon atoms. More preferably, the alkynyl has two to four carbon atoms.
The term “carbocycle,” “carbocyclyl” and “carbocyclic ring” refer to a monovalent non-aromatic, saturated or partially unsaturated ring having 3 to 12 carbon atoms as a monocyclic ring or 7 to 12 carbon atoms as a bicyclic ring. Bicyclic carbocycles having 7 to 12 atoms can be arranged, for example, as a bicyclo [4,5], [5,5], [5,6], or [6,6] system, and bicyclic carbocycles having 9 or 10 ring atoms can be arranged as a bicyclo [5,6] or [6,6] system, or as bridged systems such as bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane and bicyclo[3.2.2]nonane. Examples of monocyclic carbocycles include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, 1-cyclopent-1-enyl, 1-cyclopent-2-enyl, 1-cyclopent-3-enyl, cyclohexyl, 1-cyclohex-1-enyl, 1-cyclohex-2-enyl, 1-cyclohex-3-enyl, cyclohexadienyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, cycloundecyl, cyclododecyl, and the like.
The terms “cyclic alkyl” and “cycloalkyl” can be used interchangeably. They refer to a monovalent saturated carbocyclic ring radical. Preferably, the cyclic alkyl is 3 to 7 membered monocyclic ring radical. More preferably, the cyclic alkyl is cyclohexyl.
The term “cyclic alkenyl” refers to a carbocyclic ring radical having at least one double bond in the ring structure.
The term “cyclic alkynyl” refers to a carbocyclic ring radical having at least one triple bond in the ring structure.
“Aryl” means a monovalent aromatic hydrocarbon radical of 6-18 carbon atoms derived by the removal of one hydrogen atom from a single carbon atom of a parent aromatic ring system. Some aryl groups are represented in the exemplary structures as “Ar.” Aryl includes bicyclic radicals comprising an aromatic ring fused to a saturated, partially unsaturated ring, or aromatic carbocyclic or heterocyclic ring. Typical aryl groups include, but are not limited to, radicals derived from benzene (phenyl), substituted benzenes, naphthalene, anthracene, indenyl, indanyl, 1,2-dihydronapthalene, 1,2,3,4-tetrahydronapthyl, and the like. Preferably, aryl is phenyl group.
The terms “heterocycle,” “heterocyclyl,” and “heterocyclic ring” are used interchangeably herein and refer to a saturated or a partially unsaturated (i.e., having one or more double and/or triple bonds within the ring) carbocyclic radical of 3 to 18 ring atoms in which at least one ring atom is a heteroatom selected from nitrogen, oxygen, phosphorus, and sulfur, the remaining ring atoms being C, where one or more ring atoms is optionally substituted independently with one or more substituents described below. A heterocycle may be a monocycle having 3 to 7 ring members (2 to 6 carbon atoms and 1 to 4 heteroatoms selected from N, O, P, and S) or a bicycle having 7 to 10 ring members (4 to 9 carbon atoms and 1 to 6 heteroatoms selected from N, O, P, and S), for example: a bicyclo [4,5], [5,5], [5,6], or [6,6] system. Heterocycles are described in Paquette, Leo A.; “Principles of Modern Heterocyclic Chemistry” (W. A. Benjamin, New York, 1968), particularly Chapters 1, 3, 4, 6, 7, and 9; “The Chemistry of Heterocyclic Compounds, A series of Monographs” (John Wiley & Sons, New York, 1950 to present), in particular Volumes 13, 14, 16, 19, and 28; and J. Am. Chem. Soc. (1960) 82:5566. “Heterocyclyl” also includes radicals where heterocycle radicals are fused with a saturated, partially unsaturated ring, or aromatic carbocyclic or heterocyclic ring. Examples of heterocyclic rings include, but are not limited to, pyrrolidinyl, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothienyl, tetrahydropyranyl, dihydropyranyl, tetrahydrothiopyranyl, piperidino, morpholino, thiomorpholino, thioxanyl, piperazinyl, homopiperazinyl, azetidinyl, oxetanyl, thietanyl, homopiperidinyl, oxepanyl, thiepanyl, oxazepinyl, diazepinyl, thiazepinyl, 2-pyrrolinyl, 3-pyrrolinyl, indolinyl, 2H-pyranyl, 4H-pyranyl, dioxanyl, 1,3-dioxolanyl, pyrazolinyl, dithianyl, dithiolanyl, dihydropyranyl, dihydrothienyl, dihydrofuranyl, pyrazolidinylimidazolinyl, imidazolidinyl, 3-azabicyco[3.1.0]hexanyl, 3-azabicyclo[4.1.0]heptanyl, and azabicyclo[2.2.2]hexanyl. Spiro moieties are also included within the scope of this definition. Examples of a heterocyclic group wherein ring atoms are substituted with oxo (═O) moieties are pyrimidinonyl and 1,1-dioxo-thiomorpholinyl.
The term “heteroaryl” refers to a monovalent aromatic radical of 5- or 6-membered rings, and includes fused ring systems (at least one of which is aromatic) of 5-18 atoms, containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur. Examples of heteroaryl groups are pyridinyl (including, for example, 2-hydroxypyridinyl), imidazolyl, imidazopyridinyl, pyrimidinyl (including, for example, 4-hydroxypyrimidinyl), pyrazolyl, triazolyl, pyrazinyl, tetrazolyl, furyl, thienyl, isoxazolyl, thiazolyl, oxazolyl, isothiazolyl, pyrrolyl, quinolinyl, isoquinolinyl, indolyl, benzimidazolyl, benzofuranyl, cinnolinyl, indazolyl, indolizinyl, phthalazinyl, pyridazinyl, triazinyl, isoindolyl, pteridinyl, purinyl, oxadiazolyl, triazolyl, thiadiazolyl, furazanyl, benzofurazanyl, benzothiophenyl, benzothiazolyl, benzoxazolyl, quinazolinyl, quinoxalinyl, naphthyridinyl, and furopyridinyl.
The heterocycle or heteroaryl groups may be carbon (carbon-linked) or nitrogen (nitrogen-linked) attached where such is possible. By way of example and not limitation, carbon bonded heterocycles or heteroaryls are bonded at position 2, 3, 4, 5, or 6 of a pyridine, position 3, 4, 5, or 6 of a pyridazine, position 2, 4, 5, or 6 of a pyrimidine, position 2, 3, 5, or 6 of a pyrazine, position 2, 3, 4, or 5 of a furan, tetrahydrofuran, thiofuran, thiophene, pyrrole or tetrahydropyrrole, position 2, 4, or 5 of an oxazole, imidazole or thiazole, position 3, 4, or 5 of an isoxazole, pyrazole, or isothiazole, position 2 or 3 of an aziridine, position 2, 3, or 4 of an azetidine, position 2, 3, 4, 5, 6, 7, or 8 of a quinoline or position 1, 3, 4, 5, 6, 7, or 8 of an isoquinoline.
By way of example and not limitation, nitrogen bonded heterocycles or heteroaryls are bonded at position 1 of an aziridine, azetidine, pyrrole, pyrrolidine, 2-pyrroline, 3-pyrroline, imidazole, imidazolidine, 2-imidazoline, 3-imidazoline, pyrazole, pyrazoline, 2-pyrazoline, 3-pyrazoline, piperidine, piperazine, indole, indoline, 1H-indazole, position 2 of a isoindole, or isoindoline, position 4 of a morpholine, and position 9 of a carbazole, or O-carboline.
The heteroatoms present in heteroaryl or heterocyclcyl include the oxidized forms such as NO, SO, and SO2.
The term “halo” or “halogen” refers to F, Cl, Br or I.
The alkyl, alkenyl, alkynyl, cyclic alkyl, cyclic alkenyl, cyclic alkynyl, carbocyclyl, aryl, heterocyclyl and heteroaryl described above can be optionally substituted with one more (e.g., 2, 3, 4, 5, 6 or more) substituents.
If a substituent is described as being “substituted,” a non-hydrogen substituent is in the place of a hydrogen substituent on a carbon, oxygen, sulfur or nitrogen of the substituent. Thus, for example, a substituted alkyl substituent is an alkyl substituent wherein at least one non-hydrogen substituent is in the place of a hydrogen substituent on the alkyl substituent. To illustrate, monofluoroalkyl is alkyl substituted with a fluoro substituent, and difluoroalkyl is alkyl substituted with two fluoro substituents. It should be recognized that if there is more than one substitution on a substituent, each non-hydrogen substituent may be identical or different (unless otherwise stated).
If a substituent is described as being “optionally substituted,” the substituent may be either (1) not substituted, or (2) substituted. If a carbon of a substituent is described as being optionally substituted with one or more of a list of substituents, one or more of the hydrogens on the carbon (to the extent there are any) may separately and/or together be replaced with an independently selected optional substituent. If a nitrogen of a substituent is described as being optionally substituted with one or more of a list of substituents, one or more of the hydrogens on the nitrogen (to the extent there are any) may each be replaced with an independently selected optional substituent. One exemplary substituent may be depicted as —NR′R″, wherein R′ and R″ together with the nitrogen atom to which they are attached, may form a heterocyclic ring. The heterocyclic ring formed from R′ and R″ together with the nitrogen atom to which they are attached may be partially or fully saturated. In one embodiment, the heterocyclic ring consists of 3 to 7 atoms. In another embodiment, the heterocyclic ring is selected from the group consisting of pyrrolyl, imidazolyl, pyrazolyl, triazolyl, tetrazolyl, isoxazolyl, pyridyl and thiazolyl.
This specification uses the terms “substituent,” “radical,” and “group” interchangeably.
If a group of substituents are collectively described as being optionally substituted by one or more of a list of substituents, the group may include: (1) unsubstitutable substituents, (2) substitutable substituents that are not substituted by the optional substituents, and/or (3) substitutable substituents that are substituted by one or more of the optional substituents.
If a substituent is described as being optionally substituted with up to a particular number of non-hydrogen substituents, that substituent may be either (1) not substituted; or (2) substituted by up to that particular number of non-hydrogen substituents or by up to the maximum number of substitutable positions on the substituent, whichever is less. Thus, for example, if a substituent is described as a heteroaryl optionally substituted with up to 3 non-hydrogen substituents, then any heteroaryl with less than 3 substitutable positions would be optionally substituted by up to only as many non-hydrogen substituents as the heteroaryl has substitutable positions. Such substituents, in non-limiting examples, can be selected from a linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, aryl, heteroaryl, heterocycyclyl, halogen, guanidinium [—NH(C═NH)NH2], —OR100, NR101R102, —NO2, —NR101COR102, —SR100, a sulfoxide represented by —SOR101, a sulfone represented by —SO2R101, a sulfonate —SO3M, a sulfate —OSO3M, a sulfonamide represented by —SO2NR101R102, cyano, an azido, —COR101, —OCOR101, —OCONR101R102 and a polyethylene glycol unit (—OCH2CH2)nR101 wherein M is H or a cation (such as Na+ or K+); R101, R102 and R103 are each independently selected from H, linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit (—OCH2CH2)n—R104, wherein n is an integer from 1 to 24, an aryl having from 6 to 10 carbon atoms, a heterocyclic ring having from 3 to 10 carbon atoms and a heteroaryl having 5 to 10 carbon atoms; and R104 is H or a linear or branched alkyl having 1 to 4 carbon atoms, wherein the alkyl, alkenyl, alkynyl, aryl, heteroaryl and heterocyclcyl in the groups represented by R100, R101, R102, R103 and R104 are optionally substituted with one or more (e.g., 2, 3, 4, 5, 6 or more) substituents independently selected from halogen, —OH, —CN, —NO2 and unsubstituted linear or branched alkyl having 1 to 4 carbon atoms. Preferably, the substituents for the optionally substituted alkyl, alkenyl, alkynyl, cyclic alkyl, cyclic alkenyl, cyclic alkynyl, carbocyclyl, aryl, heterocyclyl and heteroaryl described above include halogen, —CN, —NR102R103, —CF3, —OR101, aryl, heteroaryl, heterocycycl, —SR101, —SOR101, —SO2R101 and —SO3M.
The term “compound” or “cytotoxic compound,” “cytotoxic dimer” and “cytotoxic dimer compound” are used interchangeably. They are intended to include compounds for which a structure or formula or any derivative thereof has been disclosed in the present invention or a structure or formula or any derivative thereof that has been incorporated by reference. The term also includes, stereoisomers, geometric isomers, tautomers, solvates, metabolites, salts (e.g., pharmaceutically acceptable salts) and prodrugs, and prodrug salts of a compound of all the formulae disclosed in the present invention. The term also includes any solvates, hydrates, and polymorphs of any of the foregoing. The specific recitation of “stereoisomers,” “geometric isomers,” “tautomers,” “solvates,” “metabolites,” “salt” “prodrug,” “prodrug salt,” “conjugates,” “conjugates salt,” “solvate,” “hydrate,” or “polymorph” in certain aspects of the invention described in this application shall not be interpreted as an intended omission of these forms in other aspects of the invention where the term “compound” is used without recitation of these other forms.
The term “conjugate” as used herein refers to a compound described herein or a derivative thereof that is linked to a cell binding agent.
The term “linkable to a cell binding agent” as used herein refers to the compounds described herein or derivates thereof comprising at least one linking group or a precursor thereof suitable to bond these compounds or derivatives thereof to a cell binding agent.
The term “precursor” of a given group refers to any group which may lead to that group by any deprotection, a chemical modification, or a coupling reaction.
The term “linked to a cell binding agent” refers to a conjugate molecule comprising at least one of the compounds described herein (e.g., compounds of formula (I)-(IV) and (VIII)-(XI) and drug-linker compounds describe herein), or derivative thereof bound to a cell binding agent via a suitable linking group or a precursor thereof.
The term “chiral” refers to molecules which have the property of non-superimposability of the mirror image partner, while the term “achiral” refers to molecules which are superimposable on their mirror image partner.
The term “stereoisomer” refers to compounds which have identical chemical constitution and connectivity, but different orientations of their atoms in space that cannot be interconverted by rotation about single bonds.
“Diastereomer” refers to a stereoisomer with two or more centers of chirality and whose molecules are not mirror images of one another. Diastereomers have different physical properties, e.g. melting points, boiling points, spectral properties, and reactivities. Mixtures of diastereomers may separate under high resolution analytical procedures such as crystallization, electrophoresis and chromatography.
“Enantiomers” refer to two stereoisomers of a compound which are non-superimposable mirror images of one another.
Stereochemical definitions and conventions used herein generally follow S. P. Parker, Ed., McGraw-Hill Dictionary of Chemical Terms (1984) McGraw-Hill Book Company, New York; and Eliel, E. and Wilen, S., “Stereochemistry of Organic Compounds,” John Wiley & Sons, Inc., New York, 1994. The compounds of the invention may contain asymmetric or chiral centers, and therefore exist in different stereoisomeric forms. It is intended that all stereoisomeric forms of the compounds of the invention, including but not limited to, diastereomers, enantiomers and atropisomers, as well as mixtures thereof such as racemic mixtures, form part of the present invention. Many organic compounds exist in optically active forms, i.e., they have the ability to rotate the plane of plane-polarized light. In describing an optically active compound, the prefixes D and L, or R and S, are used to denote the absolute configuration of the molecule about its chiral center(s). The prefixes d and I or (+) and (−) are employed to designate the sign of rotation of plane-polarized light by the compound, with (−) or 1 meaning that the compound is levorotatory. A compound prefixed with (+) or d is dextrorotatory. For a given chemical structure, these stereoisomers are identical except that they are mirror images of one another. A specific stereoisomer may also be referred to as an enantiomer, and a mixture of such isomers is often called an enantiomeric mixture. A 50:50 mixture of enantiomers is referred to as a racemic mixture or a racemate, which may occur where there has been no stereoselection or stereospecificity in a chemical reaction or process. The terms “racemic mixture” and “racemate” refer to an equimolar mixture of two enantiomeric species, devoid of optical activity.
The term “tautomer” or “tautomeric form” refers to structural isomers of different energies which are interconvertible via a low energy barrier. For example, proton tautomers (also known as prototropic tautomers) include interconversions via migration of a proton, such as keto-enol and imine-enamine isomerizations. Valence tautomers include interconversions by reorganization of some of the bonding electrons.
The term “prodrug” as used in this application refers to a precursor or derivative form of a compound of the invention that is capable of being enzymatically or hydrolytically activated or converted into the more active parent form. See, e.g., Wilman, “Prodrugs in Cancer Chemotherapy” Biochemical Society Transactions, 14, pp. 375-382, 615th Meeting Belfast (1986) and Stella et al., “Prodrugs: A Chemical Approach to Targeted Drug Delivery,” Directed Drug Delivery, Borchardt et al., (ed.), pp. 247-267, Humana Press (1985). The prodrugs of this invention include, but are not limited to, ester-containing prodrugs, phosphate-containing prodrugs, thiophosphate-containing prodrugs, sulfate-containing prodrugs, peptide-containing prodrugs, D-amino acid-modified prodrugs, glycosylated prodrugs, β-lactam-containing prodrugs, optionally substituted phenoxyacetamide-containing prodrugs, optionally substituted phenylacetamide-containing prodrugs, 5-fluorocytosine and other 5-fluorouridine prodrugs which can be converted into the more active cytotoxic free drug. Examples of cytotoxic drugs that can be derivatized into a prodrug form for use in this invention include, but are not limited to, compounds of the invention and chemotherapeutic agents such as described above.
The term “prodrug” is also meant to include a derivative of a compound that can hydrolyze, oxidize, or otherwise react under biological conditions (in vitro or in vivo) to provide a compound of this invention. Prodrugs may only become active upon such reaction under biological conditions, or they may have activity in their unreacted forms. Examples of prodrugs contemplated in this invention include, but are not limited to, analogs or derivatives of compounds of any one of the formulae disclosed herein that comprise biohydrolyzable moieties such as biohydrolyzable amides, biohydrolyzable esters, biohydrolyzable carbamates, biohydrolyzable carbonates, biohydrolyzable ureides, and biohydrolyzable phosphate analogues. Other examples of prodrugs include derivatives of compounds of any one of the formulae disclosed herein that comprise —NO, —NO2, —ONO, or —ONO2 moieties. Prodrugs can typically be prepared using well-known methods, such as those described by Burger's Medicinal Chemistry and Drug Discovery (1995) 172-178, 949-982 (Manfred E. Wolff ed., 5th ed); see also Goodman and Gilman's, The Pharmacological basis of Therapeutics, 8th ed., McGraw-Hill, Int. Ed. 1992, “Biotransformation of Drugs.”
One preferred form of prodrug of the invention includes compounds (with or without any linker groups) and conjugates of the invention comprising an adduct formed between an imine bond of the compounds/conjugates and an imine reactive reagent. Another preferred form of prodrug of the invention includes compounds such as those of formula (I)-(IV), wherein when the double line between N and C represents a single bond, X is H or an amine protecting group, and the compound becomes a prodrug. A prodrug of the invention may contain one or both forms of prodrugs described herein (e.g., containing an adduct formed between an imine bond of the compounds/conjugates and an imine reactive reagent, and/or containing a Y leaving group when X is —H).
The term “imine reactive reagent” refers to a reagent that is capable of reacting with an imine group. Examples of imine reactive reagent includes, but is not limited to, sulfites (H2SO3, H2SO2 or a salt of HSO3 −, SO3 2− or HSO2 formed with a cation), metabisulfite (H2S2O5 or a salt of S2O5 2− formed with a cation), mono, di, tri, and tetra-thiophosphates (PO3SH3, PO2S2H3, POS3H3, PS4H3 or a salt of PO3S3 −, PO2S2 3-, POS3 3− or PS4 3− formed with a cation), thio phosphate esters ((RiO)2PS(ORi), RiSH, RiSOH, RiSO2H, RiSO3H), various amines (hydroxyl amine (e.g., NH2OH), hydrazine (e.g., NH2NH2), NH2O—Ri, Ri′NH—Ri, NH2—Ri), NH2—CO—NH2, NH2—C(═S)—NH2′ thiosulfate (H2S2O3 or a salt of S2O3 2− formed with a cation), dithionite (H2S2O4 or a salt of S2O4 2− formed with a cation), phosphorodithioate (P(═S)(ORk)(SH)(OH) or a salt thereof formed with a cation), hydroxamic acid (RkC(═O)NHOH or a salt formed with a cation), hydrazide (RkCONHNH2), formaldehyde sulfoxylate (HOCH2SO2H or a salt of HOCH2SO2 formed with a cation, such as HOCH2SO2 −—Na+), glycated nucleotide (such as GDP-mannose), fludarabine or a mixture thereof, wherein Ri and Ri′ are each independently a linear or branched alkyl having 1 to 10 carbon atoms and are substituted with at least one substituent selected from —N(Rj)2, —CO2H, —SO3H, and —PO3H; Ri and Ri′ can be further optionally substituted with a substituent for an alkyl described herein; Rj is a linear or branched alkyl having 1 to 6 carbon atoms; and Rk is a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, aryl, heterocyclyl or heteroaryl (preferably, Rk is a linear or branched alkyl having 1 to 4 carbon atoms; more preferably, Rk is methyl, ethyl or propyl). Preferably, the cation is a monovalent cation, such as Na+ or K+. Preferably, the imine reactive reagent is selected from sulfites, hydroxyl amine, urea and hydrazine. More preferably, the imine reactive reagent is NaHSO3 or KHSO3.
As used herein and unless otherwise indicated, the terms “biohydrolyzable amide,” “biohydrolyzable ester,” “biohydrolyzable carbamate,” “biohydrolyzable carbonate,” “biohydrolyzable ureide” and “biohydrolyzable phosphate analogue” mean an amide, ester, carbamate, carbonate, ureide, or phosphate analogue, respectively, that either: 1) does not destroy the biological activity of the compound and confers upon that compound advantageous properties in vivo, such as uptake, duration of action, or onset of action; or 2) is itself biologically inactive but is converted in vivo to a biologically active compound. Examples of biohydrolyzable amides include, but are not limited to, lower alkyl amides, α-amino acid amides, alkoxyacyl amides, and alkylaminoalkylcarbonyl amides. Examples of biohydrolyzable esters include, but are not limited to, lower alkyl esters, alkoxyacyloxy esters, alkyl acylamino alkyl esters, and choline esters. Examples of biohydrolyzable carbamates include, but are not limited to, lower alkylamines, substituted ethylenediamines, amino acids, hydroxyalkylamines, heterocyclic and heteroaromatic amines, and polyether amines. Particularly favored prodrugs and prodrug salts are those that increase the bioavailability of the compounds of this invention when such compounds are administered to a mammal.
The phrase “pharmaceutically acceptable salt” as used herein, refers to pharmaceutically acceptable organic or inorganic salts of a compound of the invention. Exemplary salts include, but are not limited, to sulfate, citrate, acetate, oxalate, chloride, bromide, iodide, nitrate, bisulfate, phosphate, acid phosphate, isonicotinate, lactate, salicylate, acid citrate, tartrate, oleate, tannate, pantothenate, bitartrate, ascorbate, succinate, maleate, gentisinate, fumarate, gluconate, glucuronate, saccharate, formate, benzoate, glutamate, methanesulfonate “mesylate,” ethanesulfonate, benzenesulfonate, p-toluenesulfonate, pamoate (i.e., 1,1′-methylene-bis-(2-hydroxy-3-naphthoate)) salts, alkali metal (e.g., sodium and potassium) salts, alkaline earth metal (e.g., magnesium) salts, and ammonium salts. A pharmaceutically acceptable salt may involve the inclusion of another molecule such as an acetate ion, a succinate ion or other counter ion. The counter ion may be any organic or inorganic moiety that stabilizes the charge on the parent compound. Furthermore, a pharmaceutically acceptable salt may have more than one charged atom in its structure. Instances where multiple charged atoms are part of the pharmaceutically acceptable salt can have multiple counter ions. Hence, a pharmaceutically acceptable salt can have one or more charged atoms and/or one or more counter ion.
If the compound of the invention is a base, the desired pharmaceutically acceptable salt may be prepared by any suitable method available in the art, for example, treatment of the free base with an inorganic acid, such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, methanesulfonic acid, phosphoric acid and the like, or with an organic acid, such as acetic acid, maleic acid, succinic acid, mandelic acid, fumaric acid, malonic acid, pyruvic acid, oxalic acid, glycolic acid, salicylic acid, a pyranosidyl acid, such as glucuronic acid or galacturonic acid, an alpha hydroxy acid, such as citric acid or tartaric acid, an amino acid, such as aspartic acid or glutamic acid, an aromatic acid, such as benzoic acid or cinnamic acid, a sulfonic acid, such as p-toluenesulfonic acid or ethanesulfonic acid, or the like.
If the compound of the invention is an acid, the desired pharmaceutically acceptable salt may be prepared by any suitable method, for example, treatment of the free acid with an inorganic or organic base, such as an amine (primary, secondary or tertiary), an alkali metal hydroxide or alkaline earth metal hydroxide, or the like. Illustrative examples of suitable salts include, but are not limited to, organic salts derived from amino acids, such as glycine and arginine, ammonia, primary, secondary, and tertiary amines, and cyclic amines, such as piperidine, morpholine and piperazine, and inorganic salts derived from sodium, calcium, potassium, magnesium, manganese, iron, copper, zinc, aluminum and lithium.
As used herein, the term “solvate” means a compound which further includes a stoichiometric or non-stoichiometric amount of solvent such as water, isopropanol, acetone, ethanol, methanol, DMSO, ethyl acetate, acetic acid, and ethanolamine dichloromethane, 2-propanol, or the like, bound by non-covalent intermolecular forces. Solvates or hydrates of the compounds are readily prepared by addition of at least one molar equivalent of a hydroxylic solvent such as methanol, ethanol, 1-propanol, 2-propanol or water to the compound to result in solvation or hydration of the imine moiety.
The terms “abnormal cell growth” and “proliferative disorder” are used interchangeably in this application. “Abnormal cell growth,” as used herein, unless otherwise indicated, refers to cell growth that is independent of normal regulatory mechanisms (e.g., loss of contact inhibition). This includes, for example, the abnormal growth of: (1) tumor cells (tumors) that proliferate by expressing a mutated tyrosine kinase or overexpression of a receptor tyrosine kinase; (2) benign and malignant cells of other proliferative diseases in which aberrant tyrosine kinase activation occurs; (3) any tumors that proliferate by receptor tyrosine kinases; (4) any tumors that proliferate by aberrant serine/threonine kinase activation; and (5) benign and malignant cells of other proliferative diseases in which aberrant serine/threonine kinase activation occurs.
The terms “cancer” and “cancerous” refer to or describe the physiological condition in mammals that is typically characterized by unregulated cell growth. A “tumor” comprises one or more cancerous cells, and/or benign or pre-cancerous cells. Examples of cancer include, but are not limited to, carcinoma, lymphoma, blastoma, sarcoma, and leukemia or lymphoid malignancies. More particular examples of such cancers include squamous cell cancer (e.g., epithelial squamous cell cancer), lung cancer including small-cell lung cancer, non-small cell lung cancer (“NSCLC”), adenocarcinoma of the lung and squamous carcinoma of the lung, cancer of the peritoneum, hepatocellular cancer, gastric or stomach cancer including gastrointestinal cancer, pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, hepatoma, breast cancer, colon cancer, rectal cancer, colorectal cancer, endometrial or uterine carcinoma, salivary gland carcinoma, kidney or renal cancer, prostate cancer, vulval cancer, thyroid cancer, hepatic carcinoma, anal carcinoma, penile carcinoma, acute leukemia, head/brain and neck cancer, cancers of lymphatic organs and hematological malignancy including Leukemia (Acute lymphoblastic leukemia (ALL), Acute myelogenous leukemia (AML), Chronic lymphocytic leukemia (CLL), Chronic myelogenous leukemia (CML), Acute monocytic leukemia (AMOL), Hairy cell leukemia (HCL), T-cell prolymphocytic leukemia (T-PLL), Large granular lymphocytic leukemia, Adult T-cell leukemia), Lymphoma (small lymphocytic lymphoma (SLL), Hodgkin's lymphomas (Nodular sclerosis, Mixed cellularity, Lymphocyte-rich, Lymphocyte depleted or not depleted, and Nodular lymphocyte-predominant Hodgkin lymphoma), Non-Hodgkin's lymphomas (all subtypes), Chronic lymphocytic leukemia/Small lymphocytic lymphoma, B-cell prolymphocytic leukemia, Lymphoplasmacytic lymphoma (such as Waldenstrim macroglobulinemia), Splenic marginal zone lymphoma, Plasma cell neoplasms (Plasma cell myeloma, Plasmacytoma, Monoclonal immunoglobulin deposition diseases, Heavy chain diseases), Extranodal marginal zone B cell lymphoma (MALT lymphoma), Nodal marginal zone B cell lymphoma (NMZL), Follicular lymphoma, Mantle cell lymphoma, Diffuse large B cell lymphoma, Mediastinal (thymic) large B cell lymphoma, Intravascular large B cell lymphoma, Primary effusion lymphoma, Burkitt lymphoma/leukemia, T cell prolymphocytic leukemia, T cell large granular lymphocytic leukemia, Aggressive NK cell leukemia, Adult T cell leukemia/lymphoma, Extranodal NK/T cell lymphoma (nasal type), Enteropathy-type T cell lymphoma, Hepatosplenic T cell lymphoma, Blastic NK cell lymphoma, Mycosis fungoides/Sezary syndrome, Primary cutaneous CD30-positive T cell lymphoproliferative disorders, Primary cutaneous anaplastic large cell lymphoma, Lymphomatoid papulosis, Angioimmunoblastic T cell lymphoma, Peripheral T cell lymphoma (unspecified), Anaplastic large cell lymphoma), multiple myeloma (plasma cell myeloma or Kahler's disease).
A “therapeutic agent” encompasses both a biological agent such as an antibody, a peptide, a protein, an enzyme or a chemotherapeutic agent.
A “chemotherapeutic agent” is a chemical compound useful in the treatment of cancer. Examples of chemotherapeutic agents include Erlotinib (TARCEVA®, Genentech/OSI Pharm.), Bortezomib (VELCADE®, Millennium Pharm.), Fulvestrant (FASLODEX®, AstraZeneca), Sutent (SU11248, Pfizer), Letrozole (FEMARA®, Novartis), Imatinib mesylate (GLEEVEC®, Novartis), PTK787/ZK 222584 (Novartis), Oxaliplatin (Eloxatin®, Sanofi), 5-FU (5-fluorouracil), Leucovorin, Rapamycin (Sirolimus, RAPAMUNE®, Wyeth), Lapatinib (TYKERB®, GSK572016, Glaxo Smith Kline), Lonafarnib (SCH 66336), Sorafenib (BAY43-9006, Bayer Labs), and Gefitinib (IRESSA®, AstraZeneca), AG1478, AG1571 (SU 5271; Sugen), alkylating agents such as thiotepa and CYTOXAN® cyclosphosphamide; alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; ethylenimines and methylamelamines including altretamine, triethylenemelamine, triethylenephosphoramide, triethylenethiophosphoramide and trimethylomelamine; acetogenins (especially bullatacin and bullatacinone); a camptothecin (including the synthetic analog topotecan); bryostatin; callystatin; CC-1065 (including its adozelesin, carzelesin and bizelesin synthetic analogs); cryptophycins (particularly cryptophycin 1 and cryptophycin 8); dolastatin; duocarmycin (including the synthetic analogs, KW-2189 and CB 1-TM1); eleutherobin; pancratistatin; a sarcodictyin; spongistatin; nitrogen mustards such as chlorambucil, chlornaphazine, chlorophosphamide, estramustine, ifosfamide, mechlorethamine, mechlorethamine oxide hydrochloride, melphalan, novembichin, phenesterine, prednimustine, trofosfamide, uracil mustard; nitrosureas such as carmustine, chlorozotocin, fotemustine, lomustine, nimustine, and ranimnustine; antibiotics such as the enediyne antibiotics (e.g., calicheamicin, especially calicheamicin gammall and calicheamicin omegall (Angew Chem. Intl. Ed. Engl. (1994) 33:183-186); dynemicin, including dynemicin A; bisphosphonates, such as clodronate; an esperamicin; as well as neocarzinostatin chromophore and related chromoprotein enediyne antibiotic chromophores), aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, caminomycin, carzinophilin, chromomycinis, dactinomycin, daunorubicin, detorubicin, 6-diazo-5-oxo-L-norleucine, ADRIAMYCIN® (doxorubicin), morpholino-doxorubicin, cyanomorpholino-doxorubicin, 2-pyrrolino-doxorubicin and deoxydoxorubicin), epirubicin, esorubicin, idarubicin, marcellomycin, mitomycins such as mitomycin C, mycophenolic acid, nogalamycin, olivomycins, peplomycin, porfiromycin, puromycin, quelamycin, rodorubicin, streptonigrin, streptozocin, tubercidin, ubenimex, zinostatin, zorubicin; anti-metabolites such as methotrexate and 5-fluorouracil (5-FU); folic acid analogs such as denopterin, methotrexate, pteropterin, trimetrexate; purine analogs such as fludarabine, 6-mercaptopurine, thiamniprine, thioguanine; pyrimidine analogs such as ancitabine, azacitidine, 6-azauridine, carmofur, cytarabine, dideoxyuridine, doxifluridine, enocitabine, floxuridine; androgens such as calusterone, dromostanolone propionate, epitiostanol, mepitiostane, testolactone; anti-adrenals such as aminoglutethimide, mitotane, trilostane; folic acid replenisher such as frolinic acid; aceglatone; aldophosphamide glycoside; aminolevulinic acid; eniluracil; amsacrine; bestrabucil; bisantrene; edatraxate; defofamine; demecolcine; diaziquone; elformithine; elliptinium acetate; an epothilone; etoglucid; gallium nitrate; hydroxyurea; lentinan; lonidainine; maytansinoids such as maytansine and ansamitocins; mitoguazone; mitoxantrone; mopidanmol; nitraerine; pentostatin; phenamet; pirarubicin; losoxantrone; podophyllinic acid; 2-ethylhydrazide; procarbazine; PSK® polysaccharide complex (JHS Natural Products, Eugene, Oreg.); razoxane; rhizoxin; sizofuran; spirogermanium; tenuazonic acid; triaziquone; 2,2′,2″-trichlorotriethylamine; trichothecenes (especially T-2 toxin, verracurin A, roridin A and anguidine); urethan; vindesine; dacarbazine; mannomustine; mitobronitol; mitolactol; pipobroman; gacytosine; arabinoside (“Ara-C”); cyclophosphamide; thiotepa; taxoids, e.g., TAXOL® (paclitaxel; Bristol-Myers Squibb Oncology, Princeton, N.J.), ABRAXANE® (Cremophor-free), albumin-engineered nanoparticle formulations of paclitaxel (American Pharmaceutical Partners, Schaumberg, Ill.), and TAXOTERE® (doxetaxel; Rhone-Poulenc Rorer, Antony, France); chloranmbucil; GEMZAR® (gemcitabine); 6-thioguanine; mercaptopurine; methotrexate; platinum analogs such as cisplatin and carboplatin; vinblastine; etoposide (VP-16); ifosfamide; mitoxantrone; vincristine; NAVELBINE® (vinorelbine); novantrone; teniposide; edatrexate; daunomycin; aminopterin; capecitabine (XELODA®); ibandronate; CPT-11; topoisomerase inhibitor RFS 2000; difluoromethylornithine (DMFO); retinoids such as retinoic acid; and pharmaceutically acceptable salts, acids and derivatives of any of the above.
Also included in the definition of “chemotherapeutic agent” are: (i) anti-hormonal agents that act to regulate or inhibit hormone action on tumors such as anti-estrogens and selective estrogen receptor modulators (SERMs), including, for example, tamoxifen (including NOLVADEX®; tamoxifen citrate), raloxifene, droloxifene, 4-hydroxytamoxifen, trioxifene, keoxifene, LY117018, onapristone, and FARESTON® (toremifine citrate); (ii) aromatase inhibitors that inhibit the enzyme aromatase, which regulates estrogen production in the adrenal glands, such as, for example, 4(5)-imidazoles, aminoglutethimide, MEGASE® (megestrol acetate), AROMASIN® (exemestane; Pfizer), formestanie, fadrozole, RIVISOR® (vorozole), FEMARA® (letrozole; Novartis), and ARIMIDEX® (anastrozole; AstraZeneca); (iii) anti-androgens such as flutamide, nilutamide, bicalutamide, leuprolide, and goserelin; as well as troxacitabine (a 1,3-dioxolane nucleoside cytosine analog); (iv) protein kinase inhibitors; (v) lipid kinase inhibitors; (vi) antisense oligonucleotides, particularly those which inhibit expression of genes in signaling pathways implicated in aberrant cell proliferation, such as, for example, PKC-alpha, Ralf and H-Ras; (vii) ribozymes such as VEGF expression inhibitors (e.g., ANGIOZYME®) and HER2 expression inhibitors; (viii) vaccines such as gene therapy vaccines, for example, ALLOVECTIN®, LEUVECTIN®, and VAXID®; PROLEUKIN® rIL-2; a topoisomerase 1 inhibitor such as LURTOTECAN®; ABARELIX® rmRH; (ix) anti-angiogenic agents such as bevacizumab (AVASTIN®, Genentech); and (x) pharmaceutically acceptable salts, acids and derivatives of any of the above. Other anti-angiogenic agents include MMP-2 (matrix-metalloproteinase 2) inhibitors, MMP-9 (matrix-metalloproteinase 9) inhibitors, COX-II (cyclooxygenase II) inhibitors, and VEGF receptor tyrosine kinase inhibitors. Examples of such useful matrix metalloproteinase inhibitors that can be used in combination with the present compounds/compositions are described in WO 96/33172, WO 96/27583, EP 818442, EP 1004578, WO 98/07697, WO 98/03516, WO 98/34918, WO 98/34915, WO 98/33768, WO 98/30566, EP 606,046, EP 931,788, WO 90/05719, WO 99/52910, WO 99/52889, WO 99/29667, WO 99/07675, EP 945864, U.S. Pat. Nos. 5,863,949, 5,861,510, and EP 780,386, all of which are incorporated herein in their entireties by reference. Examples of VEGF receptor tyrosine kinase inhibitors include 4-(4-bromo-2-fluoroanilino)-6-methoxy-7-(1-methylpiperidin-4-ylmethoxy)quinazoline (ZD6474; Example 2 within WO 01/32651), 4-(4-fluoro-2-methylindol-5-yloxy)-6-methoxy-7-(3-pyrrolidin-1-ylpropoxy)-quinazoline (AZD2171; Example 240 within WO 00/47212), vatalanib (PTK787; WO 98/35985) and SU11248 (sunitinib; WO 01/60814), and compounds such as those disclosed in PCT Publication Nos. WO 97/22596, WO 97/30035, WO 97/32856, and WO 98/13354).
Other examples of chemotherapeutic agents that can be used in combination with the present compounds include inhibitors of PI3K (phosphoinositide-3 kinase), such as those reported in Yaguchi et al (2006) Jour. of the Nat. Cancer Inst. 98(8):545-556; U.S. Pat. Nos. 7,173,029; 7,037,915; 6,608,056; 6,608,053; 6,838,457; 6,770,641; 6,653,320; 6,403,588; WO 2006/046031; WO 2006/046035; WO 2006/046040; WO 2007/042806; WO 2007/042810; WO 2004/017950; US 2004/092561; WO 2004/007491; WO 2004/006916; WO 2003/037886; US 2003/149074; WO 2003/035618; WO 2003/034997; US 2003/158212; EP 1417976; US 2004/053946; JP 2001247477; JP 08175990; JP 08176070; U.S. Pat. No. 6,703,414; and WO 97/15658, all of which are incorporated herein in their entireties by reference. Specific examples of such PI3K inhibitors include SF-1126 (PI3K inhibitor, Semafore Pharmaceuticals), BEZ-235 (PI3K inhibitor, Novartis), XL-147 (PI3K inhibitor, Exelixis, Inc.).
Chemotherapeutic agents may also include any of the generic drugs or biosimilars of the brand-name drugs referenced herein, or improvements thereof, including improved formulations, prodrugs, delivery means (sustained release, bioadhesive coating, targeted delivery etc.), and dosage forms.
A “metabolite” is a product produced through metabolism in the body of a specified compound, a derivative thereof, or a conjugate thereof, or salt thereof. Metabolites of a compound, a derivative thereof, or a conjugate thereof, may be identified using routine techniques known in the art and their activities determined using tests such as those described herein. Such products may result for example from the oxidation, hydroxylation, reduction, hydrolysis, amidation, deamidation, esterification, deesterification, enzymatic cleavage, and the like, of the administered compound. Accordingly, the invention includes metabolites of compounds, a derivative thereof, or a conjugate thereof, of the invention, including compounds, a derivative thereof, or a conjugate thereof, produced by a process comprising contacting a compound, a derivative thereof, or a conjugate thereof, of this invention with a mammal for a period of time sufficient to yield a metabolic product thereof.
The phrase “pharmaceutically acceptable” indicates that the substance or composition must be compatible chemically and/or toxicologically, with the other ingredients comprising a formulation, and/or the mammal being treated therewith.
The term “protecting group” or “protecting moiety” refers to a substituent that is commonly employed to block or protect a particular functionality while reacting other functional groups on the compound, a derivative thereof, or a conjugate thereof. For example, an “amine-protecting group” or an “amino-protecting moiety” is a substituent attached to an amino group that blocks or protects the amino functionality in the compound. Such groups are well known in the art (see for example P. Wuts and T. Greene, 2007, Protective Groups in Organic Synthesis, Chapter 7, J. Wiley & Sons, NJ) and exemplified by carbamates such as methyl and ethyl carbamate, FMOC, substituted ethyl carbamates, carbamates cleaved by 1,6-β-elimination (also termed “self immolative”), ureas, amides, peptides, alkyl and aryl derivatives. Suitable amino-protecting groups include acetyl, trifluoroacetyl, t-butoxycarbonyl (BOC), benzyloxycarbonyl (CBZ) and 9-fluorenylmethylenoxycarbonyl (Fmoc). For a general description of protecting groups and their use, see P. G. M. Wuts & T. W. Greene, Protective Groups in Organic Synthesis, John Wiley & Sons, New York, 2007.
The term “leaving group” refers to an group of charged or uncharged moiety that departs during a substitution or displacement. Such leaving groups are well known in the art and include, but not limited to, halogens, esters, alkoxy, hydroxyl, tosylates, triflates, mesylates, nitriles, azide, carbamate, disulfides, thioesters, thioethers and diazonium compounds.
The term “bifunctional crosslinking agent,” “bifunctional linker” or “crosslinking agents” refers to modifying agents that possess two reactive groups; one of which is capable of reacting with a cell binding agent while the other one reacts with the cytotoxic compound to link the two moieties together. Such bifunctional crosslinkers are well known in the art (see, for example, Isalm and Dent in Bioconjugation chapter 5, p 218-363, Groves Dictionaries Inc. New York, 1999). For example, bifunctional crosslinking agents that enable linkage via a thioether bond include N-succinimidyl-4-(N-maleimidomethyl)-cyclohexane-1-carboxylate (SMCC) to introduce maleimido groups, or with N-succinimidyl-4-(iodoacetyl)-aminobenzoate (SIAB) to introduce iodoacetyl groups. Other bifunctional crosslinking agents that introduce maleimido groups or haloacetyl groups on to a cell binding agent are well known in the art (see US Patent Applications 2008/0050310, 20050169933, available from Pierce Biotechnology Inc. P.O. Box 117, Rockland, Ill. 61105, USA) and include, but not limited to, bis-maleimidopolyethyleneglycol (BMPEO), BM(PEO)2, BM(PEO)3, N-(3-maleimidopropyloxy)succinimide ester (BMPS), γ-maleimidobutyric acid N-succinimidyl ester (GMBS), ε-maleimidocaproic acid N-hydroxysuccinimide ester (EMCS), 5-maleimidovaleric acid NHS, HBVS, N-succinimidyl-4-(N-maleimidomethyl)-cyclohexane-1-carboxy-(6-amidocaproate), which is a “long chain” analog of SMCC (LC-SMCC), m-maleimidobenzoyl-N-hydroxysuccinimide ester (MBS), 4-(4-N-maleimidophenyl)-butyric acid hydrazide or HCl salt (MPBH), N-succinimidyl 3-(bromoacetamido)propionate (SBAP), N-succinimidyl iodoacetate (SIA), κ-maleimidoundecanoic acid N-succinimidyl ester (KMUA), N-succinimidyl 4-(p-maleimidophenyl)-butyrate (SMPB), succinimidyl-6-(3-maleimidopropionamido)hexanoate (SMPH), succinimidyl-(4-vinylsulfonyl)benzoate (SVSB), dithiobis-maleimidoethane (DTME), 1,4-bis-maleimidobutane (BMB), 1,4 bismaleimidyl-2,3-dihydroxybutane (BMDB), bis-maleimidohexane (BMH), bis-maleimidoethane (BMOE), sulfosuccinimidyl 4-(N-maleimido-methyl)cyclohexane-1-carboxylate (sulfo-SMCC), sulfosuccinimidyl(4-iodo-acetyl)aminobenzoate (sulfo-SIAB), m-maleimidobenzoyl-N-hydroxysulfosuccinimide ester (sulfo-MBS), N-(γ-maleimidobutryloxy)sulfosuccinimde ester (sulfo-GMBS), N-(ε-maleimidocaproyloxy)sulfosuccimido ester (sulfo-EMCS), N-(κ-maleimidoundecanoyloxy)sulfosuccinimide ester (sulfo-KMUS), and sulfosuccinimidyl 4-(p-maleimidophenyl)butyrate (sulfo-SMPB).
Heterobifunctional crosslinking agents are bifunctional crosslinking agents having two different reactive groups. Heterobifunctional crosslinking agents containing both an amine-reactive N-hydroxysuccinimide group (NHS group) and a carbonyl-reactive hydrazine group can also be used to link the cytotoxic compounds described herein with a cell-binding agent (e.g., antibody). Examples of such commercially available heterobifunctional crosslinking agents include succinimidyl 6-hydrazinonicotinamide acetone hydrazone (SANH), succinimidyl 4-hydrazidoterephthalate hydrochloride (SHTH) and succinimidyl hydrazinium nicotinate hydrochloride (SHNH). Conjugates bearing an acid-labile linkage can also be prepared using a hydrazine-bearing benzodiazepine derivative of the present invention. Examples of bifunctional crosslinking agents that can be used include succinimidyl-p-formyl benzoate (SFB) and succinimidyl-p-formylphenoxyacetate (SFPA).
Bifunctional crosslinking agents that enable the linkage of cell binding agent with cytotoxic compounds via disulfide bonds are known in the art and include N-succinimidyl-3-(2-pyridyldithio)propionate (SPDP), N-succinimidyl-4-(2-pyridyldithio)pentanoate (SPP), N-succinimidyl-4-(2-pyridyldithio)butanoate (SPDB), N-succinimidyl-4-(2-pyridyldithio)2-sulfo butanoate (sulfo-SPDB) to introduce dithiopyridyl groups. Other bifunctional crosslinking agents that can be used to introduce disulfide groups are known in the art and are disclosed in U.S. Pat. Nos. 6,913,748, 6,716,821 and US Patent Publications 20090274713 and 20100129314, all of which are incorporated herein by reference. Alternatively, crosslinking agents such as 2-iminothiolane, homocysteine thiolactone or S-acetylsuccinic anhydride that introduce thiol groups can also be used.
A “linker,” “linker moiety,” or “linking group” as defined herein refers to a moiety that connects two groups, such as a cell binding agent and a cytotoxic compound, together. Typically, the linker is substantially inert under conditions for which the two groups it is connecting are linked. A bifunctional crosslinking agent may comprise two reactive groups, one at each ends of a linker moiety, such that one reactive group can be first reacted with the cytotoxic compound to provide a compound bearing the linker moiety and a second reactive group, which can then react with a cell binding agent. Alternatively, one end of the bifunctional crosslinking agent can be first reacted with the cell binding agent to provide a cell binding agent bearing a linker moiety and a second reactive group, which can then react with a cytotoxic compound. The linking moiety may contain a chemical bond that allows for the release of the cytotoxic moiety at a particular site. Suitable chemical bonds are well known in the art and include disulfide bonds, thioether bonds, acid labile bonds, photolabile bonds, peptidase labile bonds and esterase labile bonds (see for example U.S. Pat. Nos. 5,208,020; 5,475,092; 6,441,163; 6,716,821; 6,913,748; 7,276,497; 7,276,499; 7,368,565; 7,388,026 and 7,414,073). Preferred are disulfide bonds, thioether and peptidase labile bonds. Other linkers that can be used in the present invention include non-cleavable linkers, such as those described in are described in detail in U.S. publication number 20050169933, or charged linkers or hydrophilic linkers and are described in US 2009/0274713, US 2010/01293140 and WO 2009/134976, each of which is expressly incorporated herein by reference, each of which is expressly incorporated herein by reference.
In one embodiment, the linking group with a reactive group attached at one end, such as a reactive ester, is selected from the following:
- —O(CR20R21)m(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —O(CR20R21)m(CR26═CR27)m′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —O(CR20R21)m(alkynyl)n′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″ (CR24R25)q(CO)tX″,
- —O(CR20R21)m(piperazino)t′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —O(CR20R21)m(pyrrolo)t′(CR22R23)n(OCH2H2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —O(CR20R21)mA″m″(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —S(CR20R21)m(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —S(CR20R21)m(CR26═CR27)m′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —S(CR20R21)m(alkynlyl)n′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —S(CR20R21)m(piperazino)t′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —S(CR20R21)m(pyrrolo)t′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —S(CR20R21)mA″m″(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —NR33(C═O)p″(CR20R21)m(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —NR33(C═O)p″(CR20R21)m(CR26═CR27)m′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —NR33(C═O)p″(CR20R21)m(alkynlyl)n′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q (CO)tX″,
- —NR33(C═O)p″(CR20R21)m(piperazino)t′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —NR33(C═O)p″(CR20R21)m(pyrrolo)t′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —NR33(C═O)p″(CR20R21)mA″m″(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —(CR20R21)m(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —(CR20R21)m(CR26═CR27)m′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —(CR20R21)m(alkynlyl)n′(CR22R23)n(OCH2CH2)p(CR40R41)p″CR24R25)q(CO)tX″,
- —(CR20R21)m(piperazino)t′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —(CR20R21)mA″m″(CR22R23)n(OCH2CH2)p(CR40R41)p″(CR24R25)q(CO)tX″,
- —(CR20R21)m(CR29═N—NR30)n″(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
- —(CR20R21)m(CR29═N—NR30)n″(CR26═CR27)m′(CR22R23)n(OCH2CH2)p(CR40R41) Y″, (CR24R25)q(CO)tX″,
- —(CR20R21)m(CR29═N—NR30)n″(alkynyl)n′(CR22R23)n(OCH2CH2)p(CR40R41)p″Y, (CR24R25)q(CO)tX″,
- —(CR20R21)m(CR29═N—NR30)n″A″m″(CR22R23)n(OCH2CH2)p(CR40R41)p″Y″(CR24R25)q(CO)tX″,
wherein:
m, n, p, q, m′, n′, t′ are integer from 1 to 10, or are optionally 0;
t, m″, n″, and p″ are 0 or 1;
X″ is selected from OR36, SR37, NR38R39, wherein R36, R37, R38, R39 are H, or linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 20 carbon atoms and, or, a polyethylene glycol unit —(OCH2CH2)n, R37, optionally, is a thiol protecting group when t=1, COX″ forms a reactive ester selected from N-hydroxysuccinimide esters, N-hydroxyphthalimide esters, N-hydroxy sulfo-succinimide esters, para-nitrophenyl esters, dinitrophenyl esters, pentafluorophenyl esters and their derivatives, wherein said derivatives facilitate amide bond formation;
Y″ is absent or is selected from O, S, S—S or NR32, wherein R32 has the same definition as given above for R; or
when Y″ is not S—S and t=0, X″ is selected from a maleimido group, a haloacetyl group or SR37, wherein R37 has the same definition as above;
A″ is an amino acid selected from glycine, alanine, leucine, valine, lysine, citrulline and glutamate or a polypeptide containing between 2 to 20 amino acid units;
R20, R21, R22, R23, R24, R25, R26, and R27 are the same or different, and are —H or a linear or branched alkyl having from 1 to 5 carbon atoms;
R29 and R30 are the same or different, and are —H or alkyl from 1 to 5 carbon atoms;
R33 is —H or linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 12 carbon atoms, a polyethylene glycol unit R—(OCH2CH2)n—, or R33 is —COR34, —CSR34, —SOR34, or —SO2R34, wherein R34 is H or linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 20 carbon atoms or, a polyethylene glycol unit —(OCH2CH2); and
one of R40 and R41 is optionally a negatively or positively charged functional group and the other is H or alkyl, alkenyl, alkynyl having 1 to 4 carbon atoms.
Any of the above linking groups may be present in any of the compounds, drug-linker compounds, or conjugates of the invention, including replacing the linking groups of any of the formulas described herein.
The term “amino acid” refers to naturally occurring amino acids or non-naturally occurring amino acid represented by NH2—C(Raa′Raa)—C(═O)OH, wherein Raa and Raa′ are each independently H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, aryl, heteroaryl or heterocyclyl. The term “amino acid” also refers to the corresponding residue when one hydrogen atom is removed from the amine and/or carboxy end of the amino acid, such as —NH—C(Raa′Raa)—C(═O)O—.
The term “cation” refers to an ion with positive charge. The cation can be monovalent (e.g., Na+, K+, etc.), bi-valent (e.g., Ca2+, Mg2+, etc.) or multi-valent (e.g., Al3+ etc.). Preferably, the cation is monovalent.
The term “therapeutically effective amount” means that amount of active compound or conjugate that elicits the desired biological response in a subject. Such response includes alleviation of the symptoms of the disease or disorder being treated, prevention, inhibition or a delay in the recurrence of symptom of the disease or of the disease itself, an increase in the longevity of the subject compared with the absence of the treatment, or prevention, inhibition or delay in the progression of symptom of the disease or of the disease itself. Determination of the effective amount is well within the capability of those skilled in the art, especially in light of the detailed disclosure provided herein. Toxicity and therapeutic efficacy of compound I can be determined by standard pharmaceutical procedures in cell cultures and in experimental animals. The effective amount of compound or conjugate of the present invention or other therapeutic agent to be administered to a subject will depend on the stage, category and status of the multiple myeloma and characteristics of the subject, such as general health, age, sex, body weight and drug tolerance. The effective amount of compound or conjugate of the present invention or other therapeutic agent to be administered will also depend on administration route and dosage form. Dosage amount and interval can be adjusted individually to provide plasma levels of the active compound that are sufficient to maintain desired therapeutic effects.
Cytotoxic Compounds
The present invention is directed to cytotoxic compounds described herein (e.g., compounds of formulas (I), (II), (III), and (IV)). In one embodiment, the cytotoxic compounds of the present invention do not include any compounds described in US 2010/0203007 (the entire teaching of which is incorporated herein by reference), such as those specifically disclaimed in the proviso below.
In a first specific embodiment, the invention provides a cytotoxic compound comprising a linking group with a reactive group bonded thereto capable of covalently linking the cytotoxic compound to a cell binding agent (CBA), wherein said cytotoxic compound is represented by any one of the following formulas (I), (II), (III) or (IV):
or a pharmaceutically acceptable salt thereof, wherein:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond, X is absent and Y is —H, or a linear or branched alkyl having 1 to 4 carbon atoms, and when it is a single bond, X is —H, the linking group with the reactive group bonded thereto, or an amine protecting moiety; preferably, the double line between N and C represents a double bond;
- Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5- or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido, wherein M is —H or a cation; such as Na+ or K+. Preferably, M is —H or Na+. Preferably, Y is selected from —SO3M, —OH, —OMe, —OEt or —NHOH. More preferably, Y is —SO3M or —OH; or,
- Y is a sulfite (HSO3, HSO2 or a salt of HSO3 −, SO3 2− or HSO2 − formed with a cation), metabisulfite (H2S2O5 or a salt of S2O5 2− formed with a cation), mono-, di-, tri-, and tetra-thiophosphate (PO3SH3, PO2S2H2, POS3H2, PS4H2 or a salt of PO3S3 −, PO2S2 3−, POS3 3− or PS4 3− formed with a cation), thio phosphate ester (RiO)2PS(ORi), RiS—, RiSO, RiSO2, RiSO3, thiosulfate (HS2O3 or a salt of S2O3 2− formed with a cation), dithionite (HS2O4 or a salt of S2O4 2− formed with a cation), phosphorodithioate (P(═S)(ORk′)(S)(OH) or a salt thereof formed with a cation), hydroxamic acid (Rk′C(═O)NOH or a salt formed with a cation), formaldehyde sulfoxylate (HOCH2SO2 or a salt of HOCH2SO2 formed with a cation, such as HOCH2SO2Na+) or a mixture thereof, wherein Ri is a linear or branched alkyl having 1 to 10 carbon atoms and is substituted with at least one substituent selected from —N(Rj)2, —CO2H, —SO3H, and —PO3H; Ri can be further optionally substituted with a substituent for an alkyl described herein; Rj is a linear or branched alkyl having 1 to 6 carbon atoms; Rk′ is a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, aryl, heterocyclyl or heteroaryl; preferably, Y is an adduct of a bisulfite, a hydrosulfite, or a metabisulfite, or salts thereof (such as sodium salt);
- R, for each occurrence, is independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P;
- R′ and R″ are the same or different, and are independently selected from —H, —OH, —OR, —NHR, —NR2, —COR, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, and an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms independently selected from O, S, N and P;
- Rc is —H or a substituted or unsubstituted linear or branched alkyl having 1 to 4 carbon atoms, or the linking group with the reactive group bonded thereto;
- n is an integer from 1 to 24;
- W is selected from C═O, C═S, CH2, BH, SO, and SO2;
- X′ is selected from the group consisting of —H, —OH, an amine-protecting group, the linking group with the reactive group bonded thereto, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms (e.g., phenyl), an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, and an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P. Preferably, X′ is —H, —OH, -Me or the linking group with the reactive group bonded thereto. More preferably, X′ is —H;
- Y′ is selected from the group consisting of —H, an oxo group, the linking group with the reactive group bonded thereto, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, an optionally substituted 6- to 18-membered aryl, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms. Preferably, Y′ is selected from —H or oxo. More preferably, Y′ is —H;
- R1, R2, R3, R4, R1′. R2′. R3′ and R4′ are each independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—Rc, halogen, guanidinium [—NH(C═NH)NH2], —OR, —NR′R″, —NO2, —NCO, —NR′COR″, —SR, a sulfoxide represented by —SOR′, a sulfone represented by —SO2R′, a sulfonate —SO3 −M+, a sulfate —OSO3 −M+, a sulfonamide represented by —SO2NR′R″, cyano, an azido, —COR′, —OCOR′, —OCONR′R″ and the linking group with the reactive group bonded thereto. Preferably, one of R2, R3, R2′ and R3′ is the linking group with the reactive group bonded thereto and the rest are —H;
- R6 is —H, —R, —OR, —SR, —NR′R″, —NO2, halogen, the linking group with the reactive group bonded thereto, —ORc or —SRc, wherein Rc is —H, a linear or branched alkyl having 1 to 4 carbon atoms. Preferably, R6 is —OMe or —SMe. Even more preferably, R6 is —OMe;
- Z and Z′ are independently selected from —(CH2)n′—, —(CH2)n′—CR7R8—(CH2)na′—, —(CH2)n′—NR9—(CH2)na′—, —(CH2)n′—O—(CH2)na′— and —(CH2)n′—S—(CH2)na′—;
- n′ and na′ are same or different, and are selected from 0, 1, 2 and 3;
- R7 and R8 are the same or different, and are each independently selected from —H, —OH, —SH, —COOH, —NHR′, a polyethylene glycol unit —(OCH2CH2)n—, an amino acid, a peptide unit bearing 2 to 6 amino acids, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms;
- R9 is independently selected from —H, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—;
- A and A′ are the same or different, and are independently selected from —O—, oxo (—C(═O)—), —CRR′O—, —CRR′—, —S—, —CRR′S—, —N(R5)— and —CRR′N(R5)—. Preferably, A and A′ are the same or different, and are selected from —O— and —S—. More preferably, A and A′ are —O—;
- R5 for each occurrence is independently —H or an optionally substituted linear or branched alkyl having 1 to 10 carbon atoms;
- D and D′ are the same or different, and are independently absent or selected from the group consisting of an amino acid, a peptide bearing 2 to 6 amino acids, and a polyethylene glycol unit (—OCH2CH2)n—, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, wherein the alkyl, alkenyl and alkynyl are optionally substituted with one or more (e.g., 2, 3, 4, 5, 6 or more) substituents independently selected from the group consisting of halogen, —OR, —NR′COR″, —SR and —COR′;
- Preferably, D and D′ are the same or different, and are independently selected from linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms. More preferably, D and D′ are linear or branched alkyl bearing 1 to 4 carbon atoms. Still more preferably, D and D′ are the same or different, and are selected from a linear alkyl having 1 to 4 carbon atoms;
- L is absent, the linking group with the reactive group bonded thereto, a polyethylene glycol unit (—OCH2CH2)n—, a linear, branched or cyclic alkyl or alkenyl having 1 to 10 carbon atoms (e.g., 1-6 carbon atoms), a phenyl group, a 3- to 18-membered heterocyclic ring or a 5- to 18-membered heteroaryl ring having 1 to 6 heteroatoms independently selected from O, S, N and P, wherein the alkyl or alkenyl is optionally substituted with the linking group with the reactive group bonded thereto; phenyl or heterocyclic or heteroaryl ring can be optionally substituted, wherein the substituent can be the linking group with the reactive group bonded thereto.
In certain embodiments, X is not the linking group with the reactive group bonded thereto. In certain embodiments, the double line between N and C represents a single bond, Y is not —H.
In certain embodiments, the cytotoxic compounds of the present invention are not any one of the following compounds:
In certain embodiments, Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5 or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido. Preferably, Y is Sodium Bisulfite adduct, Sodium Hydrosulfite adduct, or Sodium Metabisulfite adduct. In certain embodiments, Y is not —H.
In certain embodiments, L is absent, or is selected from an optionally substituted phenyl group and an optionally substituted pyridyl group, wherein the phenyl and the pyridyl group bears the linking group with the reactive group bonded thereto, or L is an amine group bearing the linking group with the reactive group bonded thereto (i.e., —N(linking group)-), or L is a linear, branched or cyclic alkyl or alkenyl having from 1 to 6 carbon atoms and bearing the linking group with the reactive group bonded thereto.
In a second specific embodiment, for cytotoxic dimers (I), (II), (III) and (IV), the variables are as described below:
Y is —H;
W is C═O;
R1, R2, R1′, R2′, R4 and R4′ are —H;
one of R3, or R3′ is optionally a linking group and the other is —H; R6 is —OMe;
Z and Z′ are —CH2—;
X′ is —H;
Y′ is —H;
A and A′ are —O—; and the remainder of the variables are as described in the first specific embodiment.
In a third specific embodiment, the cytotoxic dimers of formula (I), (II), (III) and (IV) are represented by the following formulas:
wherein:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is selected from —H, the linking group with the reactive group bonded thereto, or an amine protecting group (preferably X is —H);
- Y is selected from —H, —OR, —OCOR′, —SR, —NR′R″, —SO3M, —SO2M, or —OSO3M, wherein M is —H or a cation such as N+. K+. Preferably, Y is selected from —OH, —OMe, —OEt, —NHOH or —SO3M. Even more preferably, Y is —OH or —SO3M, preferably M is —H or Na+;
- R is —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms or a PEG group (CH2CH2O)n—Rc, wherein n is an integer from 1 to 24 and Rc is a linear or branched alkyl having 1 to 4 carbon atoms;
- R′ and R″ are the same or different, and are selected from —H, —OH, —OR, —NRRg′, —COR, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, an optionally substituted aryl having from 6 to 18 carbon atoms, an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms selected from O, S, N and P, a PEG group —(CH2CH2O)n—Rc, wherein n is an integer from 1 to 24, preferably n is 2, 4 or 8; and Rg′ is —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms or a PEG group —(CH2CH2O)n—Rc;
- X′ is selected from the group consisting of —H, —OH, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, phenyl, and an amine-protecting group. Preferably, X′ is —H, —OH or -Me. More preferably, X′ is —H;
- Y′ is selected from the group consisting of —H, an oxo group, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms. Preferably, Y′ is selected from —H or -Me. More preferably Y′ is —H;
- R6 is —ORc or —SRc, wherein Rc is a linear or branched alkyl having 1 to 4 carbon atoms. Preferably, R6 is —OMe or —SMe. Even more preferably, R6 is —OMe;
- A and A′ are selected from —O— and —S—. Preferably, A and A′ are —O—;
- L′, L″, and L′″ are the same or different, and are independently selected from —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—Rc, halogen, guanidinium [—NH(C═NH)NH2], —OR, —NR′R″, —NO2, —NR′COR″, —SR, a sulfoxide represented by —SOR′, a sulfone represented by —SO2R′, a sulfonate —SO3 −M+, a sulfate —OSO3+M−, a sulfonamide represented by SO2NR′R″, cyano, an azido, —COR′, —OCOR′, —OCONR′R″ and the linking group with the reactive group bonded thereto, provided only one of L′, L″, and L′″ is the linking group with the reactive group bonded thereto. Preferably, L′ is the linking group with the reactive group bonded thereto. Alternatively, one of L′, L″ or L′″ is the linking group with the reactive group bonded thereto, while the others are —H. More preferably, L′ is the linking group with the reactive group bonded thereto, and L″ and L′″ are —H;
- G is selected from —CH— or —N—; and the remainder of the variables are as described in the first specific embodiment.
In certain embodiments, X is not the linking group with the reactive group bonded thereto. In certain embodiments, the double line between N and C represents a single bond, Y is not —H.
In certain embodiments, A and A′ are both —O—, R6 is —OMe, and G is —CH—.
In a fourth specific embodiment, for the cytotoxic dimers of formula (IA), (IIA), (IIIA) or (IVA), L′ is represented by the formula:
—W′—Rx—V—Ry-J,
—W′—Rx—V—Ry-J,
wherein:
-
- W′ and V are the same or different, and are each independently absent, or selected from —CReRe′—, —O—, —O—C(═O)—, —C(═O)—O—, —S—, —SO—, —SO2—, —CH2—S—, —CH2O—, —CH2NRe—, —O—(C═O)O—, —O—(C═O)N(Re)—, —N(Re)—, —N(Re)—C(═O)—, —C(═O)—N(Re)—, —N(Re)—C(═O)O—, —N(C(═O)Re)C(═O)—, —N(C(═O)Re)—, —(O—CH2—CH2)n—, —SS—, or —C(═O)—, or an amino acid, or a peptide having 2 to 8 amino acids;
- Rx and Ry are the same or different, and are each independently absent or an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, an
aryl bearing 6 to 10 carbon atoms or a 3- to 8-membered hetereocyclic ring bearing 1 to 3 heteroatoms selected from O, N or S; Re and Re′ are the same or different, and are selected from —H, a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms or —(CH2—CH2—O)n—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms, optionally bearing a secondary amino (e.g., —NHR101) or tertiary amino (—NR101R102) group or a 5 or 6-membered nitrogen containing heterocycle, such as piperidine or morpholine, wherein R101 and R102 are each independently a linear, branched, or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms. Preferably, R101 and R102 are each independently a linear or branched alkyl having 1 to 6 carbon atoms; - n is an integer from 1 to 24; and
- J comprises the reactive group bonded thereto, and is selected from a maleimide, a haloacetamido, —SH, —SSRd, —CH2SH, —CH(Me)SH, —C(Me)2SH, —NHRc1, —CH2NHRc1, —NRc1NH2, —COOH, and —COE, wherein —COE represents a reactive ester selected from, but not limited to, N-hydroxysuccinimde ester, N-hydroxy sulfosuccinimide ester, nitrophenyl (e.g., 2 or 4-nitrophenyl) ester, dinitrophenyl (e.g., 2,4-dinitrophenyl) ester, sulfo-tetraflurophenyl (e.g., 4-sulfo-2,3,5,6-tetrafluorophenyl) ester, and pentafluorophenyl ester, and wherein Rc1 is —H or a substituted or unsubstituted linear or branched alkyl having 1 to 4 carbon atoms, and
- Rd is selected from phenyl, nitrophenyl (e.g., 2 or 4-nitrophenyl), dinitrophenyl (e.g., 2 or 4-nitrophenyl), carboxynitrophenyl (e.g., 3-carboxy-4-nitrophenyl), pyridyl or nitropyridyl (e.g., 4-nitropyridyl).
In certain embodiments, J is —SH, —SSRd, a maleimide, or a N-hydroxysuccinimide ester.
In certain embodiments, Re′ is —H or -Me; Re is a linear or branched alkyl having 1 to 6 carbon atoms or —(CH2—CH2—O)n—Rk; n is an integer from 2 to 8; preferably Rk is —H, -Me or —CH2CH2—NMe2, and the remainder of the variables are as described above in the fourth specific embodiment.
In certain embodiments, V is an amino acid or a peptide having 2 to 8 amino acids. In certain embodiments, V is valine-citrulline, gly-gly-gly, or ala-leu-ala-leu.
In certain embodiments,
-
- W′ is —O—, —N(Re)— or —N(Re)—C(═O)—;
- Re is H, a linear or branched alkyl having 1 to 4 carbon atoms, or —(CH2—CH2—O)n—Rk;
- Rx is a linear or branched alkyl having 1 to 6 carbon atoms;
- V is absent, —(O—CH2—CH2)n—, —C(═O)—NH—, —S—, —NH—C(═O)—;
- Ry is absent or a linear or branched alkyl having 1 to 4 carbon atoms; and
- J is —SH, —SSRd or —COE (preferably, N-hydroxysuccinimide ester). The remainder of the variables is as described in the fourth specific embodiment.
In certain embodiments,
-
- W′ is —O—, —N(Re)— or —N(Re)—C(═O)—;
- Re is H, Me, or —(CH2—CH2—O)n-Me;
- n is an integer from 2 to 6;
- Rx is linear or branched
alkyl bearing 1 to 6 carbon atoms; - V and Ry are absent; and
- J is —COE, preferably N-hydroxysuccinimide ester. The remainder of the variables is as described in the fourth specific embodiment.
In a fifth specific embodiment, L′ is represented by the following formula:
—W′—[CR1″R2″]a—V—[Cy]0-1-[CR3″R4″]b—COE,
—W′—[CR1″R2″]a—V—[Cy]0-1-[CR3″R4″]b—COE,
wherein:
-
- R1″, R2″, and R3″ are each independently —H or a linear or branched
alkyl bearing 1 to 4 carbon atoms, preferably -Me; - R4″ is —H, a linear or branched
alkyl bearing 1 to 4 carbon atoms (preferably -Me), —SO3H, or —SO3 −M+, wherein M+ is a pharmaceutically acceptable cation; - a is an integers from 0-5 (e.g., from 0 to 2, 3, 4, or 5), and b is an integer from 0-6 (e.g., from 0 to 3, 4, 5, or 6); and,
- Cy is an optionally substituted 5-membered heterocyclic ring bearing an N heteroatom, preferably Cy is
- R1″, R2″, and R3″ are each independently —H or a linear or branched
In certain embodiments, W′ is —N(Re)—.
In certain embodiments, such as in the fourth and/or the fifth specific embodiment, Re is —(CH2—CH2—O)2-6—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms.
In certain embodiments, such as in the fourth and/or the fifth specific embodiment, V is —S— or —SS—.
In a sixth specific embodiments, such as in the fourth and/or the fifth specific embodiment, L′ is represented by the following formula:
—NRe—[CR1″R2″]a—S—[CR3″,R4″]b—COE.
—NRe—[CR1″R2″]a—S—[CR3″,R4″]b—COE.
In certain embodiments, the compound is any of the following:
-
- wherein Y is —H or —SO3M, and M is —H or a pharmaceutically acceptable cation. In certain embodiments, Y is —SO3M.
In a seventh specific embodiments, such as in the fourth and/or the fifth specific embodiment, L′ is represented by the following formula:
—NRe—[CR1″R2″]a—S—Cy—[CR3″,R4″]b—COE.
—NRe—[CR1″R2″]a—S—Cy—[CR3″,R4″]b—COE.
In certain embodiments, the compound is any one of the following:
-
- wherein Y is —H or —SO3M, and M is —H or a pharmaceutically acceptable cation. In certain embodiments, Y is —SO3M.
In a eighth specific embodiment, the cytotoxic dimers of formula (I), (II), (III) and (IV) are represented by the following formulas:
wherein:
-
- W′ is absent, or selected from —O—, —N(Re)—C(═O)—, —N(C(═O)Re)—, —S— or —CH2—S—, —CH2NRe—;
- Rx is absent or selected from a linear, branched or cyclic alkyl having 1 to 10 carbon atoms;
- Re is —H, a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms or —(CH2—CH2—O)n—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms, optionally bearing a secondary amino (e.g., —NHR101) or tertiary amino (—NR101R102) group or a 5- or 6-membered nitrogen containing heterocycle, such as piperidine or morpholine, wherein R101 and R102 are each independently a linear, branched, or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms. Preferably, R101 and R102 are each independently a linear or branched alkyl having 1 to 6 carbon atoms;
- Zs is —H, —SRm;
- Rm is Rd or a substituted or unsubstituted linear or branched alkyl having 1 to 4 carbon atoms bearing a reactive ester, selected from N-hydroxysuccinimide esters, N-hydroxyphtalimide esters, N-hydroxy sulfo-succinimide esters, para-nitrophenyl esters, dinitrophenyl esters, pentafluorophenyl esters;
- Rd is selected from phenyl, nitrophenyl, dinitrophenyl, carboxynitrophenyl, pyridyl or nitropyridyl; and,
- n is an integer from 1 to 24; and the remainder of the variables are as described above in the third specific embodiment.
Preferably, Rk is —H or -Me, and n is an integer from 2 to 8. Preferably, Rx is a linear or branched alkyl having 1 to 6 carbon atoms; and the remainder of the variables is as described above in the third, fourth, and/or the fifth specific embodiment.
In a ninth specific embodiment, the cytotoxic dimers of formula (I), (II), (III) and (IV) are represented by the following formulas:
wherein:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond, X is absent and Y is —H, and when it is a single bond, X is selected from —H, the linking group with the reactive group bonded thereto, or an amine protecting group (preferably X is —H or an amine protecting group; more preferably, X is —H);
- Y is selected from —H, —OR, —OCOR′, —SR, —NR′R,″ —SO3M, —SO2M or —OSO3M (e.g., Y is —OR, —OCOR′, —SR, —NR′R,″ —SO3M, —SO2M or —OSO3M), wherein M is —H or a cation such as Na+ or K+;
- R is —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms or a PEG group —(CH2CH2O)n—Rc, wherein n is an integer from 1 to 24, and Rc is a linear or branched alkyl having 1 to 4 carbon atoms;
- R′ and R″ are the same or different, and are selected from —H, —OH, —OR, —NRRg′, —COR, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, an optionally substituted aryl having from 6 to 18 carbon atoms, an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms selected from O, S, N and P, a PEG group —(CH2CH2O)n—Rc, wherein n is an integer from 1 to 24, preferably n is 2, 4 or 8; and Rg′ is —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms or a PEG group —(CH2CH2O)n—Rc;
- X′ is selected from the group consisting of —H, —OH, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, phenyl, and an amine-protecting group;
- Y′ is selected from the group consisting of —H, an oxo group, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms;
- A and A′ are selected from —O— and —S—;
- W′ is absent, or selected from —O—, —N(Re)—, —N(Re)—C(═O)—, —N(C(═O)Re)—, —S— or —CH2—S—, —CH2NRe—;
- Rx is absent or selected from a linear, branched or cyclic alkyl having 1 to 10 carbon atoms;
- Re is —H, a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms or —(CH2—CH2—O)n—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms, optionally bearing a secondary amino (e.g., —NHR101) or tertiary amino (—NR101R102) group or a 5 or 6-membered nitrogen containing heterocycle, such as piperidine or morpholine, wherein R101 and R102 are each independently a linear, branched, or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms;
- G is selected from —CH— or —N—;
- Zs is —H, or is selected from any one of the following formulas:
wherein:
-
- q is an integer from 1 to 5;
- n is an integer from 2 to 6;
- D is —H or —SO3M;
- M is —H or a cation, such as Na+ or K+.
In certain embodiments, Zs is represented by any one of the following formulas:
In certain embodiments, W′ is —N(Re)—.
In certain embodiments, Re is —(CH2—CH2—O)n—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms.
In certain embodiments, Rk is —H or -Me, n is 4, and q is 2.
In certain embodiments, Rx is a linear or branched alkyl having 1 to 6 carbon atoms.
In certain embodiments, Rx may be —(CH2)p—(CRfRg)—, wherein Rf and Rg are each independently selected from H or a linear or branched alkyl having 1 to 4 carbon atoms; and p is 0, 1, 2 or 3.
In certain embodiments, Rf and Rg are the same or different, and are selected from —H and -Me; and p is 1.
In a tenth specific embodiment, the compounds of formula (VIII), (IX), (X) and (XI) described in the ninth specific embodiment, the variables are as described below:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is —H; Y is —H, —OH or —SO3M;
- M is —H or a pharmaceutically acceptable cation (e.g., Na+);
- X′ and Y′ are both —H;
- A and A′ are both —O—;
- R6 is —OMe; and
- Rx is a linear or branched alkyl having 1 to 6 carbon atoms.
In a related embodiment, Y is —OH or —SO3M.
In another embodiment, the compounds of formula (VIII), (IX), (X) and (XI) described in the ninth specific embodiment, the variables are as described below:
-
- W′ is —O—, —N(Re)—, —N(Re)—C(═O)—, —N(CORe)—, —S— or —CH2—S—;
- Rx is absent or selected from a linear, branched or cyclic alkyl having 1 to 6 carbon atoms;
- Re is —H, a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms or —(CH2—CH2—O)n—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms, optionally bearing a primary, secondary or tertiary amino group or a 5- or 6-membered Nitrogen containing heterocycle, such as piperidine or morpholine;
- n is an integer from 1 to 24; and the remainder of the variables are as described above in the ninth specific embodiment.
Preferably, Rk is —H or -Me, and n is an integer from 2 to 8. Preferably, Rx is a linear or branched alkyl having 1 to 6 carbon atoms.
Preferably, Rx is —(CH2)p—(CRfRg)—, wherein Rf and Rg are each independently selected from H or a linear or branched alkyl having 1 to 4 carbon atoms; p is 0, 1, 2 or 3. More preferably, Rf and Rg are the same or different, and are selected from —H and -Me; and p is 1.
In another preferred embodiment, the linker is represented by any one of the formula selected from formulas (a1), (a4), (a5), (a10) and (a11) shown above; and the remainder of the variables are as described above in the tenth specific embodiment.
In a eleventh specific embodiment, for compounds of formula (IB), (IIB), (IIIB) and (IVB) described in the eighth specific embodiment, the variables are as described below:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is —H; Y is —H, —OH or —SO3M (e.g., Y is —OH or —SO3M);
- M is —H or Na+;
- X′ and Y′ are both —H;
- A and A′ are both —O—;
- R6 is —OMe;
- Rx is a linear or branched alkyl having 1 to 6 carbon atoms; and the remainder of the variables is as described above in the third, fourth, or the fifth specific embodiment.
Preferably, Rx is —(CH2)p—(CRfRg)—, wherein Rf and Rg are each independently selected from H or a linear or branched alkyl having 1 to 4 carbon atoms; p is 0, 1, 2 or 3. More preferably, Rf and Rg are the same or different, and are selected from —H and -Me; and p is 1.
In any of the specific embodiments above (e.g., the first to the 11th specific embodiments), the double line between N and C may represent a double bond.
In any of the specific embodiments above (e.g., the first to the 11th specific embodiments), the double line between N and C may represent a single bond, X is —H, the linking group with the reactive group bonded thereto, or an amine protecting group (e.g., X is —H or or an amine protecting group); and Y is selected from —H, —OR, —OCOR′, —SR, —NR′R,″ an optionally substituted 5- or 6-membered nitrogen-containing heterocycle, —SO3M, —SO2M and a sulfate —OSO3M (e.g., Y is —OR, —OCOR′, —SR, —NR′R,″ an optionally substituted 5- or 6-membered nitrogen-containing heterocycle, —SO3M, —SO2M and a sulfate —OSO3M).
In certain embodiments, Y is selected from —H, —SO3M, —OH, —OMe, —OEt or —NHOH (e.g., Y is —SO3M, —OH, —OMe, —OEt or —NHOH).
In certain embodiments, Y is —H, —SO3M or —OH (e.g., Y is —SO3M or —OH).
In certain embodiments, M is —H, Na+ or K+.
In any of the specific embodiments above (e.g., the first to the 11th specific embodiments), W, when present, is C═O.
In any of the specific embodiments above (e.g., the first to the 11th specific embodiments), Z and Z′, when present, are —CH2.
In any of the specific embodiments above (e.g., the first to the 11th specific embodiments), X′ is selected from the group consisting of —H, —OH, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, phenyl, the linking group with the reactive group bounded thereto, and an amine-protecting group.
In certain embodiments, X′ is —H, —OH, -Me or the linking group with the reactive group bounded thereto.
In certain embodiments, X′ is —H.
In any of the specific embodiments above (e.g., the first to the 11th specific embodiments), Y′ is selected from the group consisting of —H, an oxo group, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms.
In certain embodiments, Y′ is —H or oxo.
In certain embodiments, Y′ is —H.
In any of the specific embodiments above (e.g., the first to the 11th specific embodiments), A and A′ are the same or different, and are selected from O, S, NR5 and oxo (C═O). A and A′ may be same or different and selected from —O— and —S—. Preferably, both A and A′ are —O—.
In any of the specific embodiments above (e.g., the first to the 11th specific embodiments), D and D′, when present, are the same or different, and are independently selected from a polyethylene glycol unit (—OCH2CH2)n, wherein n is an integer from 1 to 24, an amino acid, a peptide bearing 2 to 6 amino acids, or a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, wherein the alkyl, alkenyl and alkynyl are optionally substituted with one or more substituents independently selected from the group consisting of halogen, —OR, —NR′COR″, —SR and —COR′. Preferably, D and D′ are linear or branched alkyl bearing 1 to 4 carbon atoms.
In a twelfth embodiment, the cytotoxic compound of the present invention as described in the first, third, and ninth embodiment is represented by the following:
-
- the double line between N and C represents a double bond;
- Y is —H;
- W is C═O;
- R1, R2, R1′, R2′, R4 and R4′ are —H;
- one of R3, or R3′ is optionally the linking group with the reactive group bounded thereto and the other is —H;
- R6 is —OMe;
- Z and Z′ are —CH2
- X′ is —H; Y′ is —H; and
A and A′ are —O—.
In a thirteenth embodiment, the cytotoxic compound of the present invention is:
or a pharmaceutically acceptable salt thereof.
In one embodiment, compound 29b can used in methods of the present invention described herein. In a preferred embodiment, compound 29b can be used for treating a proliferative disorder, such as cancer.
In another embodiment, compound 29b can be used for screening cell lines to identify cell lines that are sensitive to benzodiazepine compounds, such as benzodiazepine derivatives described herein.
Drug Compounds & Drug-Linker Compounds
The cytotoxic compounds described above comprise a linking group with a reactive group bonded thereto, which compounds may result from reacting a bifunctional crosslinking reagent with “linker-less” compounds to form the so-called drug-linker compounds. Alternatively, drug compounds that are otherwise identical to the drug-linker compounds, but without the linker moiety are also encompassed by the present invention.
Thus in certain embodiments, the invention provides a cytotoxic compound without linking group, but may be capable of reacting with a bifunctional crosslinking agent to form a compound of the invention, such as any one of the 1st to the 12th specific embodiments described above; or to form a cell-binding agent conjugate of the invention (such as those described below). An exemplary linkerless cytotoxic compound of the invention includes compound 29b of the 13th specific embodiment above. The linkerless cytotoxic compounds of the invention are represented by any one of the following formulas (I′), (II′), (III′) or (IV′):
or a pharmaceutically acceptable salt thereof, wherein:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond, X is absent and Y is —H, or a linear or branched alkyl having 1 to 4 carbon atoms, and when it is a single bond, X is —H, or an amine protecting moiety; preferably, the double line between N and C represents a double bond;
- Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5- or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido, wherein M is —H or a cation; such as Na′ or K+. Preferably, M is —H or Na′. Preferably, Y is selected from —SO3M, —OH, —OMe, —OEt or —NHOH. More preferably, Y is —SO3M or —OH; or,
- Y is a sulfite (HSO3, HSO2 or a salt of HSO3 −, SO3 2− or HSO2 formed with a cation), metabisulfite (H2S2O5 or a salt of S2O5 2− formed with a cation), mono-, di-, tri-, and tetra-thiophosphate (PO3SH3, PO2S2H2, POS3H2, PS4H2 or a salt of PO3S3 −, PO2S2 3−, POS3 3− or PS4 3− formed with a cation), thio phosphate ester (RiO)2PS(ORi), RiS—, RiSO, RiSO2, RiSO3, thiosulfate (HS2O3 or a salt of S2O3 2− formed with a cation), dithionite (HS2O4 or a salt of S2O4 2− formed with a cation), phosphorodithioate (P(═S)(ORk′)(S)(OH) or a salt thereof formed with a cation), hydroxamic acid (Rk′C(═O)NOH or a salt formed with a cation), formaldehyde sulfoxylate (HOCH2SO2 or a salt of HOCH2SO2 formed with a cation, such as HOCH2SO2 −Na+) or a mixture thereof, wherein Ri is a linear or branched alkyl having 1 to 10 carbon atoms and is substituted with at least one substituent selected from —N(Rj)2, —CO2H, —SO3H, and —PO3H; Ri can be further optionally substituted with a substituent for an alkyl described herein; Rj is a linear or branched alkyl having 1 to 6 carbon atoms; Rk′ is a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, aryl, heterocyclyl or heteroaryl; preferably, Y is an adduct of a bisulfite, a hydrosulfite, or a metabisulfite, or salts thereof (such as sodium salt);
- R, for each occurrence, is independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P;
- R′ and R″ are the same or different, and are independently selected from —H, —OH, —OR, —NHR, —NR2, —COR, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, and an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms independently selected from O, S, N and P;
- Rc is —H or a substituted or unsubstituted linear or branched alkyl having 1 to 4 carbon atoms;
- n is an integer from 1 to 24;
- W is selected from C═O, C═S, CH2, BH, SO, and SO2;
- X′ is selected from the group consisting of —H, —OH, an amine-protecting group, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms (e.g., phenyl), an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, and an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P. Preferably, X′ is —H, —OH, or -Me. More preferably, X′ is —H;
- Y′ is selected from the group consisting of —H, an oxo group, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, an optionally substituted 6- to 18-membered aryl, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms. Preferably, Y′ is selected from —H or oxo. More preferably, Y′ is —H;
- R1, R2, R3, R4, R1′. R2′. R3′ and R4′ are each independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—Rc, halogen, guanidinium [—NH(C═NH)NH2], —OR, —NR′R″, —NO2, —NCO, —NR′COR″, —SR, a sulfoxide represented by —SOR′, a sulfone represented by —SO2R′, a sulfonate —SO3 −M+, a sulfate —OSO3 −M+, a sulfonamide represented by —SO2NR′R″, cyano, an azido, —COR′, —OCOR′, and —OCONR′R″. Preferably, 1, 2, 3, or all of R2, R3, R2′ and R3′ is —H;
- R6 is —H, —R, —OR, —SR, —NR′R″, —NO2, halogen, —ORc or —SRc, wherein Rc is —H, a linear or branched alkyl having 1 to 4 carbon atoms. Preferably, R6 is —OMe or —SMe. Even more preferably, R6 is —OMe;
- Z and Z′ are independently selected from —(CH2)n′—, —(CH2)n′—CR7R8—(CH2)na′—, —(CH2)n′—NR9—(CH2)na′—, —(CH2)n′—O—(CH2)na′— and —(CH2)n′—S—(CH2)na′—;
- n′ and na′ are same or different, and are selected from 0, 1, 2 and 3;
- R7 and R8 are the same or different, and are each independently selected from —H, —OH, —SH, —COOH, —NHR′, a polyethylene glycol unit —(OCH2CH2)n—, an amino acid, a peptide unit bearing 2 to 6 amino acids, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms;
- R9 is independently selected from —H, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—;
- A and A′ are the same or different, and are independently selected from —O—, oxo (—C(═O)—), —CRR′O—, —CRR′—, —S—, —CRR′S—, —N(R5)— and —CRR′N(R5)—. Preferably, A and A′ are the same or different, and are selected from —O— and —S—. More preferably, A and A′ are —O—;
- R5 for each occurrence is independently —H or an optionally substituted linear or branched alkyl having 1 to 10 carbon atoms;
- L′, L″, and L′″ are the same or different, and are independently selected from —H, halogen, an optionally substituted linear, branched or cyclic alkyl, haloalkyl, alkoxy, haloalkoxy, —NO2, or —CN;
- G is selected from —CH— or —N—.
In certain embodiments, the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is selected from —H, or an amine protecting group (preferably X is —H); W is C═O; R1, R2, R3, R4, R1′, R2′, R3′, and R4′ are —H; Z and Z′ are —CH2—; A and A′ are both —O—; W is —(C═O)—; G is —CH—; R6 is —H, or optionally substituted C1-C10 linear, C1-C10 branched, or C3-C7 cyclic alkyl, —O-alkyl, or —O-haloalkyl, such as —OMe; X′ is selected from the group consisting of —H, —OH, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, phenyl, and an amine-protecting group; and Y′ is selected from the group consisting of —H, an oxo group, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms.
Preferably, when Y is not —H, Y is selected from —OR, —OCOR′, —SR, —NR′R″, —SO3M, —SO2M, or —OSO3M, wherein M is —H or a cation such as Na+. K+. Preferably, Y is selected from —H, —OH, —OMe, —OEt, —NHOH or —SO3M (e.g., Y is —OH, —OMe, —OEt, —NHOH or —SO3M). Even more preferably, Y is —H, —OH or —SO3M (e.g., Y is —OH or —SO3M), preferably M is —H or Na+.
In certain embodiments, the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is selected from —H, or an amine protecting group (preferably X is —H); W is C═O; R1, R2, R3, R4, R1, R2′, R3′, R4′, X′ and Y′ are —H; Z and Z′ are —CH2—; A and A′ are both —O—; W is —(C═O)—; G is —CH—; R6 is —H, or optionally substituted C1-C10 linear, C1-C10 branched, or C3-C7 cyclic alkyl, —O-alkyl, or —O-haloalkyl, such as —OMe.
The bifunctional crosslinking agents can be any bifunctional linker known in the art. For example, the bifunctional linkers can be used for making the drug-linker compounds are those that form disulfide bonds, thioether bonds, acid labile bonds, photolabile bonds, peptidase labile bonds and esterase labile bonds with the cytotoxic compounds (see for example, U.S. Pat. Nos. 5,208,020; 5,475,092; 6,441,163; 6,716,821; 6,913,748; 7,276,497; 7,276,499; 7,368,565; 7,388,026 and 7,414,073, all of which are incorporated herein by reference). Preferably, the bifunctional crosslinking agents are those that form disulfide bonds, thioether and peptidase labile bonds with the cytotoxic compounds. Other bifunctional crosslinking agents that can be used in the present invention include non-cleavable linkers, such as those described in U.S. publication number US 2005/0169933, or charged linkers or hydrophilic linkers and are described in US 2009/0274713, US 2010/01293140 and WO 2009/134976, each of which is expressly incorporated herein by reference. The bifunctional crosslinking agents that can be used for making the (drug-linker) compounds of the present invention also include those described in Thermo Scientific Pierce Crosslinking Technical Handbook, the entire teaching of which is incorporated herein by reference.
Synthesis of Cytotoxic Compounds
Representative processes for preparing the cytotoxic dimer compounds of the present invention are shown in FIGS. 1-11 . The dimers were prepared by reacting a monomer with linker compounds which possess two leaving groups such as halogen, triflate, mesylate, or tosylate such as that described for the synthesis of 1c in FIG. 1 . Synthesis of representative dimers which bear a thiol or disulfide moiety to enable linkage to cell binding agents via reducible or non-reducible bonds are shown in FIGS. 1-5, 7, 8, and 10 . In FIG. 1 a linker containing a short polyethylene glycol moiety and an alkyl disulfide was prepared through reductive amination of 1a. Conversion of 1b to its corresponding mesylate and coupling with the IBD (indolinobenzodiazepine) monomer unit gave dimer 1c which was reduced to the mono-imine, converted to the free thiol, and coupled with 2 to give compound 1g of the present invention. In FIG. 3 , a modified form of IBD monomer was prepared and coupled to give a dimer of the present invention in which the reduced imine was converted to a linker. FIG. 4 describes a dimer possessing a short polyethylene glycol moiety and an amide disulfide which was reduced to thiol 4c and converted to a reactive ester. FIG. 5 describes the synthesis of pyridyl disulfide containing linker 5e which was converted to the mono-imine thiol 5i of the present invention before being converted to a reactive ester. Synthesis of representative dimers which possess linkers that can react with cell binding agents are prepared by converting the methyl esters to the corresponding reactive esters of a leaving group such as, but not limited to, N-hydroxysuccinimide esters, N-hydroxyphthalimide esters, N-hydroxy sulfo-succinimide esters, para-nitrophenyl esters, pentafluorophenyl esters are shown in FIGS. 6, 9, and 11 .
Representative processes for preparing the cytotoxic dimer compounds of the present invention suitable for one-step conjugation with a cell binding agent are shown in FIGS. 1 and 12-19 . In all of these examples a dimer containing a thiol moiety is reacted with a bifunctional crosslinking reagent possessing a reactive group such as, but not limited to, a thiopyridyl, a maleimide, iodide, bromide, or tosylate on one side and a reactive substituent suitable for reaction with a cell binding agent such as, but not limited to, N-hydroxysuccinimide esters, N-hydroxyphtalimide esters, N-hydroxy sulfo-succinimide esters, paranitrophenyl esters, pentafluorophenyl esters.
Alternative synthetic processes for preparing representative cytotoxic dimer compounds of the present invention are shown in FIGS. 20-21 . In FIG. 20 , the synthesis of the mono reduced dimer (i.e., having one imine group) is accomplished by a two step coupling method, in which a reduced form of monomer is either initially coupled to the linker followed by coupling with the IBD monomer or the dimer is prepared using a mixture of both reduced monomer and the IBD monomer in the coupling with the reactive linker. While the di-reduced dimer is potentially a byproduct of the second synthetic pathway previously described, a more direct route is shown in FIG. 21 in which the reduced monomer is coupled to both with the linker directly.
Cell-Binding Agents
The effectiveness of the conjugates of the invention as therapeutic agents depends on the careful selection of an appropriate cell-binding agent. Cell-binding agents may be of any kind presently known, or that become known and includes peptides and non-peptides. Generally, these can be antibodies (especially monoclonal antibodies), lymphokines, hormones, growth factors, vitamins (such as folate etc., which may bind to a cell surface receptor thereof, e.g., a folate receptor), nutrient-transport molecules (such as transferrin), or any other cell-binding molecule or substance.
In certain embodiments, the cell-binding agents are proteins or polypeptides, or compounds comprising proteins or polypeptides. Preferably, the proteins or polypeptides comprise one or more Lys residues with side chain —NH2 groups. Alternatively or in addition, the proteins or polypeptides comprise one or more Cys residues. The side chain —SH groups of the Cys residues may be intact, or may be in a disulfide bond that can be reduced. Preferably, reduction of the disulfide bond(s) does not significantly negatively impact the cell-binding function of the proteins or polypeptides (e.g., in the case of antibody or antigen-binding portion thereof, reduction of the disulfide bonds does not substantially increase the dissociation of light chains/heavy chains).
The Lys side chain —NH2 groups and/or the Cys side chain —SH groups may be covalently linked to the linkers, which are in turn linked to the dimer compounds of the invention, thus conjugating the cell-binding agents to the dimer compounds of the invention. Each protein-based cell-binding agents may contain multiple Lys side chain —NH2 groups and/or the Cys side chain —SH groups available for linking the compounds of the invention through the bifunctional crosslinkers.
More specific examples of cell-binding agents that can be used include:
polyclonal antibodies;
monoclonal antibodies;
fragments of antibodies such as Fab, Fab′, and F(ab′)2, Fv, minibodies, diabodies, tribodies, tetrabodies (Parham, J. Immunol. 131:2895-2902 (1983); Spring et al. J. Immunol. 113:470-478 (1974); Nisonoff et al. Arch. Biochem. Biophys. 89:230-244 (1960), Kim et al., Mol, Cancer Ther., 7: 2486-2497 (2008), Carter, Nature Revs., 6: 343-357 (2006));
interferons (e.g. α, β, γ);
lymphokines such as IL-2, IL-3, IL-4, IL-6;
hormones such as insulin, TRH (thyrotropin releasing hormone), MSH (melanocyte-stimulating hormone), steroid hormones, such as androgens and estrogens;
growth factors and colony-stimulating factors such as EGF, TGF-alpha, FGF, VEGF, G-CSF, M-CSF and GM-CSF (Burgess, Immunology Today 5:155-158 (1984)); transferrin (O'Keefe et al. J. Biol. Chem. 260:932-937 (1985)); vitamins, such as folate;
Protein scaffolds based on a consensus sequence of fibronectin type III (FN3) repeats (also known as Centyrins; See U.S. Patent Publication 2010/0255056, incorporated herein by reference);
Designer Ankyrin Repeat Proteins (DARPins; U.S. Patent Application Nos. 20040132028; 20090082274; 20110118146; 20110224100, incorporated herein by reference), C. Zahnd et al. 2010, Cancer Res., 70; 1595-1605, incorporated herein by reference); and,
Fibronectin domain scaffold proteins (Adnectins: US Patent Application Nos. 20070082365; 20080139791, incorporated herein by reference).
Monoclonal antibody techniques allow for the production of extremely specific cell-binding agents in the form of specific monoclonal antibodies. Particularly well known in the art are techniques for creating monoclonal antibodies produced by immunizing mice, rats, hamsters or any other mammal with the antigen of interest such as the intact target cell, antigens isolated from the target cell, whole virus, attenuated whole virus, and viral proteins such as viral coat proteins. Sensitized human cells can also be used. Another method of creating monoclonal antibodies is the use of phage libraries of scFv (single chain variable region), specifically human scFv (see e.g., Griffiths et al., U.S. Pat. Nos. 5,885,793 and 5,969,108; McCafferty et al., WO 92/01047; Liming et al., WO 99/06587). In addition, resurfaced antibodies disclosed in U.S. Pat. No. 5,639,641 may also be used, as may chimeric antibodies and humanized antibodies. Selection of the appropriate cell-binding agent is a matter of choice that depends upon the particular cell population that is to be targeted, but in general human monoclonal antibodies are preferred if an appropriate one is available.
For example, the monoclonal antibody MY9 is a murine IgG antibody that binds specifically to the CD33 Antigen {J. D. Griffin et al 8 Leukemia Res., 521 (1984)} and can be used if the target cells express CD33 as in the disease of acute myelogenous leukemia (AML). The cell-binding agent may be any compound that can bind a cell, either in a specific or non-specific manner. Generally, these can be antibodies (especially monoclonal antibodies and antibody fragments), interferons, lymphokines, hormones, growth factors, vitamins, nutrient-transport molecules (such as transferrin), or any other cell-binding molecule or substance.
Where the cell-binding agent is an antibody, it binds to an antigen that is a polypeptide and may be a transmembrane molecule (e.g. receptor) or a ligand such as a growth factor. Exemplary antigens include molecules such as renin; a growth hormone, including human growth hormone and bovine growth hormone; growth hormone releasing factor; parathyroid hormone; thyroid stimulating hormone; lipoproteins; alpha-1-antitrypsin; insulin A-chain; insulin B-chain; proinsulin; follicle stimulating hormone; calcitonin; luteinizing hormone; glucagon; clotting factors such as factor vmc, factor IX, tissue factor (TF), and von Willebrands factor; anti-clotting factors such as Protein C; atrial natriuretic factor; lung surfactant; a plasminogen activator, such as urokinase or human urine or tissue-type plasminogen activator (t-PA); bombesin; thrombin; hemopoietic growth factor; tumor necrosis factor-alpha and -beta; enkephalinase; RANTES (regulated on activation normally T-cell expressed and secreted); human macrophage inflammatory protein (MIP-1-alpha); a serum albumin, such as human serum albumin; Muellerian-inhibiting substance; relaxin A-chain; relaxin B-chain; prorelaxin; mouse gonadotropin-associated peptide; a microbial protein, such as beta-lactamase; DNase; IgE; a cytotoxic T-lymphocyte associated antigen (CTLA), such as CTLA-4; inhibin; activin; vascular endothelial growth factor (VEGF); receptors for hormones or growth factors; protein A or D; rheumatoid factors; a neurotrophic factor such as bone-derived neurotrophic factor (BDNF), neurotrophin-3, -4, -5, or -6 (NT-3, NT4, NT-5, or NT-6), or a nerve growth factor such as NGF-β; platelet-derived growth factor (PDGF); fibroblast growth factor such as aFGF and bFGF; fibroblast growth factor receptor 2 (FGFR2), epidermal growth factor (EGF); transforming growth factor (TGF) such as TGF-alpha and TGF-beta, including TGF-β1, TGF-β2, TGF-β3, TGF-β4, or TGF-β5; insulin-like growth factor-I and -II (IGF-I and IGF-II); des(1-3)-IGF-I (brain IGF-I), insulin-like growth factor binding proteins, melanotransferrin, EpCAM, GD3, FLT3, PSMA, PSCA, MUC1, MUC16, STEAP, CEA, TENB2, EphA receptors, EphB receptors, folate receptor, FOLR1, mesothelin, cripto, alphavbeta6, integrins, VEGF, VEGFR, EGFR, tarnsferrin receptor, IRTA1, IRTA2, IRTA3, IRTA4, IRTA5; CD proteins such as CD2, CD3, CD4, CD5, CD6, CD8, CD11, CD14, CD19, CD20, CD21, CD22, CD25, CD26, CD28, CD30, CD33, CD36, CD37, CD38, CD40, CD44, CD52, CD55, CD56, CD59, CD70, CD79, CD80. CD81, CD103, CD105, CD134, CD137, CD138, CD152 or an antibody which binds to one or more tumor-associated antigens or cell-surface receptors disclosed in US Publication No. 20080171040 or US Publication No. 20080305044 and are incorporated in their entirety by reference; erythropoietin; osteoinductive factors; immunotoxins; a bone morphogenetic protein (BMP); an interferon, such as interferon-alpha, -beta, and -gamma; colony stimulating factors (CSFs), e.g., M-CSF, GM-CSF, and G-CSF; interleukins (ILs), e.g., IL-1 to IL-10; superoxide dismutase; T-cell receptors; surface membrane proteins; decay accelerating factor; viral antigen such as, for example, a portion of the HIV envelope; transport proteins; homing receptors; addressins; regulatory proteins; integrins, such as CD11a, CD11b, CD11c, CD18, an ICAM, VLA-4 and VCAM; a tumor associated antigen such as HER2, HER3 or HER4 receptor; endoglin, c-Met, c-kit, 1GF1R, PSGR, NGEP, PSMA, PSCA, LGR5, B7H4, and fragments of any of the above-listed polypeptides.
Additionally, GM-CSF, which binds to myeloid cells can be used as a cell-binding agent to diseased cells from acute myelogenous leukemia. IL-2 which binds to activated T-cells can be used for prevention of transplant graft rejection, for therapy and prevention of graft-versus-host disease, and for treatment of acute T-cell leukemia. MSH, which binds to melanocytes, can be used for the treatment of melanoma, as can antibodies directed towards melanomas. Folic acid can be used to target the folate receptor expressed on ovarian and other tumors. Epidermal growth factor can be used to target squamous cancers such as lung and head and neck. Somatostatin can be used to target neuroblastomas and other tumor types.
Cancers of the breast and testes can be successfully targeted with estrogen (or estrogen analogues) or androgen (or androgen analogues) respectively as cell-binding agents.
In one embodiment, the cell-binding agent is humanized monoclonal antibodies. In another embodiment, the cell-binding agent is huMy9-6, or other related antibodies, which are described in U.S. Pat. Nos. 7,342,110 and 7,557,189 (incorporated herein by reference). In another embodiment, the cell-binding agent is an anti-folate receptor antibody described in U.S. Provisional Application Nos. 61/307,797, 61/346,595, 61/413,172 and U.S. application Ser. No. 13/033,723 (published as US 2012-0009181 A1). The teachings of all these applications are incorporated herein by reference in its entirety.
In certain embodiments, the cell-binding agent may be a monoclonal antibody or antigen-binding portions thereof sharing sequences critical for antigen-binding with an antibody disclosed herein, such as huMy9-6 or its related antibodies described in U.S. Pat. Nos. 7,342,110 and 7,557,189 (incorporated herein by reference). These derivative antibodies may have substantially the same or identical (1) light chain and/or heavy chain CDR3 regions; (2) light chain and/or heavy chain CDR1, CDR2, and CDR3 regions; or (3) light chain and/or heavy chain regions, compared to an antibody described herein. Sequences within these regions may contain conservative amino acid substitutions, including substitutions within the CDR regions. Preferably, there is no more than 1, 2, 3, 4, or 5 conservative substitutions. In certain embodiments, the derivative antibodies have a light chain region and/or a heavy chain region that is at least about 90%, 95%, 99% or 100% identical to an antibody described herein. These derivative antibodies may have substantially the same binding specificity and/or affinity to the target antigen compared to an antibody described herein. Preferably, the Kd and/or koff values of the derivative antibodies are within 10-fold (either higher or lower), 5-fold (either higher or lower), 3-fold (either higher or lower), or 2-fold (either higher or lower) of an antibody described herein. These derivative antibodies may be fully human antibodies, or humanized antibodies, or chimeric antibodies. The derivative antibodies may be produced according to any art-recognized methods.
In one embodiment, the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof that specifically binds a human folate receptor 1, wherein the antibody comprises: (a) a heavy chain CDR1 comprising GYFMN (SEQ ID NO: 1); a heavy chain CDR2 comprising RIHPYDGDTFYNQXaa1FXaa2Xaa3 (SEQ ID NO: 2); and a heavy chain CDR3 comprising YDGSRAMDY (SEQ ID NO: 3); and (b) a light chain CDR1 comprising KASQSVSFAGTSLMH (SEQ ID NO: 4); a light chain CDR2 comprising RASNLEA (SEQ ID NO: 5); and a light chain CDR3 comprising QQSREYPYT (SEQ ID NO: 6); wherein Xaa1 is selected from K, Q, H, and R; Xaa2 is selected from Q, H, N, and R; and Xaa3 is selected from G, E, T, S, A, and V. Preferably, the heavy chain CDR2 sequence comprises RIHPYDGDTFYNQKFQG (SEQ ID NO: 7).
In another embodiment, the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof that specifically binds the human folate receptor 1 comprising the heavy chain having the amino acid sequence of QVQLVQSGAEVVKPGASVKISCKASGYTFTGYFMNWVKQSPGQSLEWIGRIHPY DGDTFYNQKFQGKATLTVDKSSNTAHMELLSLTSEDFAVYYCTRYDGSRAMDY WGQGTTVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNS GALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKV EPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDP EVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKV SNKALPAPIEKTISKAKGQPREPQVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAV EWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEAL HNHYTQKSLSLSPGK (SEQ ID NO: 8).
In another embodiment, the anti-folate antibody is a humanized antibody or antigen binding fragment thereof encoded by the plasmid DNA deposited with the ATCC on Apr. 7, 2010 and having ATCC deposit nos. PTA-10772 and PTA-10773 or 10774.
In another embodiment, the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof that specifically binds the human folate receptor 1 comprising the light chain having the amino acid sequence of DIVLTQSPLSLAVSLGQPAIISCKASQSVSFAGTSLMHWYHQKPGQQPRLLIYRAS NLEAGVPDRFSGSGSKTDFTLNISPVEAEDAATYYCQQSREYPYTFGGGTKLEIK RTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQE SVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC (SEQ ID NO: 9); or DIVLTQSPLSLAVSLGQPAIISCKASQSVSFAGTSLMHWYHQKPGQQPRLLIYRAS NLEAGVPDRFSGSGSKTDFTLTISPVEAEDAATYYCQQSREYPYTFGGGTKLEIK RTVAAPSVFIFPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQE SVTEQDSKDSTYSLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC (SEQ ID NO: 10).
In another embodiment the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof that specifically binds the human folate receptor 1 comprising the heavy chain having the amino acid sequence of SEQ ID NO: 8, and the light chain having the amino acid sequence of SEQ ID NO: 9 or SEQ ID NO: 10. Preferably, the antibody comprises the heavy chain having the amino acid sequence of SEQ ID NO: 8 and the light chain having the amino acid sequence of SEQ ID NO: 10 (hu FOLR1).
In another embodiment, the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof encoded by the plasmid DNA deposited with the ATCC on Apr. 7, 2010 and having ATCC deposit nos. PTA-10772 and PTA-10773 or 10774.
In another embodiment, the anti-folate receptor antibody is a humanized antibody or antigen binding fragment thereof comprising a heavy chain variable domain at least about 90%, 95%, 99% or 100% identical to QVQLVQSGAEVVKPGASVKISCKASGYTFTGYFMNWVKQSPGQSLEWIGRIHPY DGDTFYNQKFQGKATLTVDKSSNTAHMELLSLTSEDFAVYYCTRYDGSRAMDY WGQGTTVTVSS (SEQ ID NO: 11), and a light chain variable domain at least about 90%, 95%, 99% or 100% identical to DIVLTQSPLSLAVSLGQPAIISCKASQSVSFAGTSLMHWYHQKPGQQPRLLIYRAS NLEAGVPDRFSGSGSKTDFTLNISPVEAEDAATYYCQQSREYPYTFGGGTKLEIK R (SEQ ID NO: 12); orDIVLTQSPLSLAVSLGQPAIISCKASQSVSFAGTSLMHWYHQKPGQQPRLLIYR ASNLEAGVPDRFSGSGSKTDFTLTISPVEAEDAATYYCQQSREYPYTFGGGTKLEI KR (SEQ ID NO: 13).
Cell-Binding Agent-Drug Conjugates
The present invention also provides cell-binding agent-drug conjugates comprising a cell-binding agent linked to one or more cytotoxic compounds of the present invention via a variety of linkers, including, but not limited to, disulfide linkers, thioether linkers, amide bonded linkers, peptidase-labile linkers, acid-labile linkers, esterase-labile linkers.
Representative conjugates of the invention are antibody/cytotoxic compound, antibody fragment/cytotoxic compound, epidermal growth factor (EGF)/cytotoxic compound, melanocyte stimulating hormone (MSH)/cytotoxic compound, thyroid stimulating hormone (TSH)/cytotoxic compound, somatostatin/cytotoxic compound, folate/cytotoxic compound, estrogen/cytotoxic compound, estrogen analogue/cytotoxic compound, androgen/cytotoxic compound, and androgen analogue/cytotoxic compound. A representative folate/cytotoxic compound conjugate is depicted below, with the optional —SO3—Na adduct on the imine bond of one of the two drug monomers. A representative synthesis scheme for this conjugate is shown in FIG. 54 .
In a preferred embodiment, the present invention provides conjugates comprising an indolinobenzodiazepine dimer compound (e.g., compounds of formulas (I)-(IV), (IA)-(IVA) and (IB)-(IVB)) and the cell-binding agent linked through a covalent bond. The linker can be cleaved at the site of the tumor/unwanted proliferating cells to deliver the cytotoxic agent to its target in a number of ways. The linker can be cleaved, for example, by low pH (hydrazone), reductive environment (disulfide), proteolysis (amide/peptide link), or through an enzymatic reaction (esterase/glycosidase).
In a preferred aspect, representative cytotoxic conjugates of the invention are antibody/indolinobenzodiazepine dimer compound, antibody fragment/indolinobenzodiazepine dimer compound, epidermal growth factor (EGF)/indolinobenzodiazepine dimer compound, melanocyte stimulating hormone (MSH)/indolinobenzodiazepine dimer compound, thyroid stimulating hormone (TSH)/indolinobenzodiazepine dimer compound, somatostatin/indolinobenzodiazepine dimer compound, folate/indolinobenzodiazepine dimer compound, estrogen/indolinobenzodiazepine dimer compound, estrogen analogue/indolinobenzodiazepine dimer compound, prostate specific membrane antigen (PSMA) inhibitor/indolinobenzodiazepine dimer compound, matriptase inhibitor/indolinobenzodiazepine dimer compound, designed ankyrin repeat proteins (DARPins)/indolinobenzodiazepine dimer compound, androgen/indolinobenzodiazepine dimer compound, and androgen analogue/indolinobenzodiazepine dimer compound.
Thus in the fourteenth specific embodiment, the invention provides a conjugate comprising: a cytotoxic compound and a cell binding agent (CBA), wherein the cytotoxic compound comprises a linking group which covalently links the cytotoxic compound to the CBA, and wherein the cytotoxic compound is represented by any one of the following formulas:
or a pharmaceutically acceptable salt thereof, wherein:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, or a linear or branched alkyl having 1 to 4 carbon atoms, and when it is a single bond, X is —H, the linking group, or an amine protecting moiety;
- Y is —H or a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5 or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido; or,
- Y is a sulfite (HSO3, HSO2 or a salt of HSO3 −, SO3 2− or HSO2 − formed with a cation), metabisulfite (H2S2O5 or a salt of S2O5 2− formed with a cation), mono-, di-, tri-, and tetra-thiophosphate (PO3SH3, PO2S2H2, POS3H2, PS4H2 or a salt of PO3S3 −, PO2S2 3−, POS3 3− or PS4 3− formed with a cation), thio phosphate ester (RiO)2PS(ORi), RiS—, RiSO, RiSO2, RiSO3, thiosulfate (HS2O3 or a salt of S2O3 2− formed with a cation), dithionite (HS2O4 or a salt of S2O4 2− formed with a cation), phosphorodithioate (P(═S)(ORk′)(S)(OH) or a salt thereof formed with a cation), hydroxamic acid (Rk′C(═O)NOH or a salt formed with a cation), formaldehyde sulfoxylate (HOCH2SO2 − or a salt of HOCH2SO2 − formed with a cation, such as HOCH2SO2Na+) or a mixture thereof, wherein Ri is a linear or branched alkyl having 1 to 10 carbon atoms and is substituted with at least one substituent selected from —N(R)2, —CO2H, —SO3H, and —PO3H; Ri can be further optionally substituted with a substituent for an alkyl described herein; R3 is a linear or branched alkyl having 1 to 6 carbon atoms; Rk′ is a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, aryl, heterocyclyl or heteroaryl; preferably, Y is an adduct of a bisulfite, a hydrosulfite, or a metabisulfite, or salts thereof (such as sodium salt);
- M is —H or a cation;
- R, for each occurrence, is independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, or an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P;
- R′ and R″ are each independently selected from —H, —OH, —OR, —NHR, —NR2, —COR, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, and an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms independently selected from O, S, N and P;
- Rc is —H or a substituted or unsubstituted linear or branched alkyl having 1 to 4 carbon atoms, or the linking group;
- n is an integer from 1 to 24;
- W is selected from C═O, C═S, CH2, BH, SO and SO2;
- X′ is selected from —H, an amine-protecting group, the linking group, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(CH2CH2O)n—Rc, an optionally substituted aryl having 6 to 18 carbon atoms, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, and an optionally substituted 3- to 18-membered heterocyclic ring containing 1 to 6 heteroatoms independently selected from O, S, N and P;
- Y′ is selected from —H, an oxo group, the linking group, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, an optionally substituted 6- to 18-membered aryl, an optionally substituted 5- to 18-membered heteroaryl ring containing one or more heteroatoms independently selected from nitrogen, oxygen, and sulfur, an optionally substituted 3- to 18-membered heterocyclic ring having 1 to 6 heteroatoms;
- R1, R2, R3, R4, R1′, R2′, R3′ and R4′ are each independently selected from the group consisting of —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—Rc, halogen, guanidinium [—NH(C═NH)NH2], —OR, —NR′R″, —NO2, —NCO, —NR′COR″, —SR, a sulfoxide represented by —SOR′, a sulfone represented by —SO2R′, a sulfonate —SO3 −M+, a sulfate —OSO3 −M+, a sulfonamide represented by —SO2NR′R″, cyano, an azido, —COR′, —OCOR′, —OCONR′R″ and the linking group;
- R6 is —H, —R, —OR, —SR, —NR′R″, —NO2, halogen or the linking group;
- Z and Z′ are independently selected from —(CH2)n′—, —(CH2)n′—CR7R8—(CH2)na′—, —(CH2)n′—NR9—(CH2)na′—, —(CH2)n′—O—(CH2)na′— and —(CH2)n′—S—(CH2)na′—;
- n′ and na′ are the same or different, and are selected from 0, 1, 2 and 3;
- R7 and R8 are the same or different, and are each independently selected from —H, —OH, —SH, —COOH, —NHR′, a polyethylene glycol unit —(OCH2CH2)n—, an amino acid, a peptide unit bearing 2 to 6 amino acids, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms;
- R9 is independently selected from —H, an optionally substituted linear, branched or cyclic alkyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)n—;
- A and A′ are the same or different, and are independently selected from —O—, oxo (—C(═O)—), —CRR′O—, —CRR′—, —S—, —CRR′S—, —NR5 and —CRR′N(R5)—;
- R5 for each occurrence is independently —H or an optionally substituted linear or branched alkyl having 1 to 10 carbon atoms;
- D and D′ are the same or different, and are independently absent or selected from the group consisting of an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, an amino acid, a peptide bearing 2 to 6 amino acids, and a polyethylene glycol unit (—OCH2CH2)n—;
- L is absent, the linking group, a polyethylene glycol unit (—OCH2CH2)n—, a linear, branched or cyclic alkyl or alkenyl having 1 to 10 carbon atoms, a phenyl group, a 3- to 18-membered heterocyclic ring or a 5- to 18-membered heteroaryl ring having 1 to 6 heteroatoms independently selected from O, S, N and P, wherein the alkyl or alkenyl is optionally substituted with the linking group; phenyl or heterocyclic or heteroaryl ring can be optionally substituted, wherein the substituent can be the linking group.
In certain embodiments, X is not the linking group. In certain embodiments, the double line between N and C represents a single bond, Y is not —H.
In certain embodiments, Y is a leaving group selected from —OR, —OCOR′, —OCOOR′, —OCONR′R″, —NR′R″, —NR′COR″, —NR′NR′R″, an optionally substituted 5 or 6-membered nitrogen-containing heterocycle (e.g., piperidine, tetrahydropyrrole, pyrazole, morpholine, etc.), a guanidinum represented by —NR′(C═NH)NR′R″, an amino acid, or a peptide represented by —NRCOP′, wherein P′ is an amino acid or a polypeptide containing between 2 to 20 amino acid units, —SR, —SOR′, —SO2M, —SO3M, —OSO3M, halogen, cyano and an azido.
In certain embodiments, the compound is not any one of the following compounds:
In certain embodiments, the conjugates of the invention include the following:
wherein:
-
- CBA is the cell-binding agent, r is an integer from 1 to 10, Y is —H, an adduct of a bisulfite, a hydrosulfite, or a metabisulfite, or salts thereof, or —SO3M, and M is —H or a pharmaceutically acceptable cation.
In certain embodiments, L is absent, or is selected from an optionally substituted phenyl group and an optionally substituted pyridyl group, wherein the phenyl and the pyridyl group bears the linking group, or L is an amine group bearing the linking group (i.e., —N(linking group)-), or L is a linear, branched or cyclic alkyl or alkenyl having from 1 to 6 carbon atoms and bearing the linking group.
In the fifteenth specific embodiment, the compound is represented by any one of the following formulas:
wherein:
-
- L′, L″, and L′″ are the same or different, and are independently selected from —H, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, a polyethylene glycol unit —(OCH2CH2)—Rc, halogen, guanidinium [—NH(C═NH)NH2], —OR, —NR′R″, —NO2, —NR′COR″, —SR, a sulfoxide represented by —SOR′, a sulfone represented by —SO2R′, a sulfonate —SO3M, a sulfate —OSO3M, a sulfonamide represented by —SO2NR′R″, cyano, an azido, —COR′, —OCOR′, —OCONR′R″ and the linking group, provided only one of L′, L″, and L′″ is the linking group; and
- G is selected from —CH— or —N—. The remaining groups are as described in the fourteenth specific embodiment above.
In certain embodiments, one of L′, L″, or L′″ is the linking group, while the others are —H. Preferably, L′ is the linking group, and L″ and L′″ are —H.
In certain embodiments, A and A′ are both —O—, R6 is —OMe, and G is —CH—.
In a sixteenth specific embodiment, L′ is represented by the following formula:
—W′—Rx—V—Ry-J,
—W′—Rx—V—Ry-J,
wherein:
-
- W′ and V are the same or different, and are each independently absent, or selected from —CReRe′—, —O—, —O—C(═O)—, —C(═O)—O—, —S—, —SO—, —SO2—, —CH2—S—, —CH2O—, —CH2NRe—, —O—(C═O)O—, —O—(C═O)N(Re)—, —N(Re)—, —N(Re)—C(═O)—, —C(═O)—N(Re)—, —N(Re)—C(═O)O—, —N(C(═O)Re)C(═O)—, —N(C(═O)Re)—, —(O—CH2—CH2)n—, —SS—, or —C(═O)—, or an amino acid, or a peptide having 2 to 8 amino acids;
- Rx and Ry are the same or different, and are each independently absent or an optionally substituted linear, branched or cyclic alkyl, alkenyl, or alkynyl having 1 to 10 carbon atoms, an
aryl bearing 6 to 10 carbon atoms or a 3- to 8-membered hetereocyclic ring bearing 1 to 3 heteroatoms selected from O, N or S; - Re and Re′ are the same or different, and are selected from —H, a linear, branched or cyclic alkyl, alkenyl, or alkynyl having 1 to 10 carbon atoms or —(CH2—CH2—O)—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms, optionally bearing a secondary amino (e.g., —NHR101) or tertiary amino (—NR101R102) group or a 5- or 6-membered nitrogen containing heterocycle, such as piperidine or morpholine, wherein R101 and R102 are each independently a linear, branched, or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms; preferably, R101 and R102 are each independently a linear or branched alkyl having 1 to 6 carbon atoms;
- n is an integer from 1 to 24; and
- J is covalently linked to the CBA, and is selected from a succinimide, a acetamido, —S—, —SS—, —CH2S—, —CH(Me)S—, —C(Me)2S—, —NRc1—, —CH2NRc1—, —NRc1N—, and —C(═O)—, wherein Rc1 is —H or a substituted or unsubstituted linear or branched alkyl having 1 to 4 carbon atoms.
In certain embodiments, J is —S—, —SS—, a succinimide, or —C(═O)—.
In certain embodiments, Re is —H or -Me; Re is a linear or branched alkyl having 1 to 6 carbon atoms or —(CH2—CH2—O)n—Rk; n is an integer from 2 to 8; and Rk is —H, -Me or —CH2CH2—NMe2, and the remainder of the variables are as described above in the fifteenth specific embodiment.
In certain embodiments, V is an amino acid or a peptide having 2 to 8 amino acids.
In certain embodiments, V is valine-citrulline, gly-gly-gly, or ala-leu-ala-leu.
In certain embodiments,
-
- W′ is —O—, —N(Re)— or —N(Re)—C(═O)—;
- Re is H, a linear or branched alkyl having 1 to 4 carbon atoms, or —(CH2—CH2—O)n—Rk;
- Rx is a linear or branched alkyl having 1 to 6 carbon atoms;
- V is absent, —(O—CH2—CH2)n—, —C(═O)—NH—, —S—, —NH—C(═O)—;
- Ry is absent or a linear or branched alkyl having 1 to 4 carbon atoms; and
- J is —S—, —SS—, or —C(═O)—, and the remaining groups are as defined in the sixteenth specific embodiment.
In certain embodiments,
-
- W′ is —O—, —N(Re)— or —N(Re)—C(═O)—;
- Re is —H, -Me, or —(CH2—CH2—O)n-Me;
- n is an integer from 2 to 6;
- Rx is linear or branched
alkyl bearing 1 to 6 carbon atoms; - V and Ry are absent; and
- J is —C(═O)—. The remaining groups are as defined in the sixteenth specific embodiment.
In a seventeenth specific embodiment, L′ in the sixteenth specific embodiment is represented by the following formula:
—W′—[CR1″R2″]a—V—[Cy]0-1—[CR3″R4″]b—C(═O)—,
—W′—[CR1″R2″]a—V—[Cy]0-1—[CR3″R4″]b—C(═O)—,
wherein:
-
- R1″, R2″, and R3″ are each independently —H or a linear or branched
alkyl bearing 1 to 4 carbon atoms, preferably -Me; - R4″ is —H, a linear or branched
alkyl bearing 1 to 4 carbon atoms (preferably -Me), —SO3H, or —SO3 −M+, wherein M+ is a pharmaceutically acceptable cation; - a is an integers from 0-5 (e.g., from 0 to 2, 3, 4, or 5), and b is an integer from 0-6 (e.g., from 0 to 3, 4, 5, or 6); and,
- Cy is an optionally substituted 5-membered heterocyclic ring bearing an N heteroatom, preferably Cy is
- R1″, R2″, and R3″ are each independently —H or a linear or branched
In certain embodiments, such as in the sixteenth or the seventeenth specific embodiment, W′ is —N(Re)—.
In certain embodiments, such as in the sixteenth or the seventeenth specific embodiment, Re is —(CH2—CH2—O)2-6—Rk, wherein Rk is a linear or branched alkyl having 1 to 6 carbon atoms.
In certain embodiments, such as in the sixteenth or the seventeenth specific embodiment, V is —S— or —SS—.
In an eighteenth specific embodiment, L′ in the sixteenth or the seventeenth specific embodiment is represented by the following formula:
—NRe—[CR1″R2″]a—S—[CR3″R4″]b—C(═O)—.
—NRe—[CR1″R2″]a—S—[CR3″R4″]b—C(═O)—.
In certain embodiments, such as in the sixteenth to eighteenth specific embodiments, the conjugate is:
-
- wherein r is an integer from 1 to 10, Y is —H or —SO3M (e.g., Y is —SO3M is —H or a pharmaceutically acceptable cation.
In certain embodiments, such as in the sixteenth to eighteenth specific embodiments, the antibody is huMy9-6.
In a nineteenth specific embodiment, L′ in the sixteenth or the seventeenth specific embodiment is represented by the following formula:
—NRe—[CR1″R2″]a-S—Cy—[CR3″R4″]b—C(═O)—.
—NRe—[CR1″R2″]a-S—Cy—[CR3″R4″]b—C(═O)—.
In certain embodiments, such as in the sixteenth, seventeenth, and the nineteenth specific embodiments, the conjugate is:
-
- wherein r is an integer from 1 to 10, Y is —H or —SO3M (e.g., Y is —SO3M), and M is —H or a pharmaceutically acceptable cation.
In certain embodiments, such as in the sixteenth, seventeenth, and the nineteenth specific embodiments, the antibody is huMy9-6.
In a twentieth specific embodiment, the compound is represented by the following formula:
wherein:
-
- W′ is absent, or selected from —O—, —N(Re)—, —N(Re)—C(═O)−, —N(C(═O)Re)—, —S—, —CH2—S—, or —CH2NRe—;
- Rx is absent or selected from a linear, branched or cyclic alkyl having 1 to 10 carbon atoms;
- Re is —H, a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms or —(CH2—CH2—O)n—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms, optionally bearing a secondary amino (e.g., —NHR101) or tertiary amino (—NR101R102) group or a 5 or 6-membered nitrogen containing heterocycle, such as piperidine or morpholine, wherein R101 and R102 are each independently a linear, branched, or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms;
- Zs is linked to the CBA, and is either a bond, or —SRm—;
- Rm is Rd or a substituted linear or branched alkyl having 1 to 4 carbon atoms bearing a reactive ester, selected from N-hydroxysuccinimide esters, N-hydroxyphtalimide esters, N-hydroxy sulfo-succinimide esters, para-nitrophenyl esters, dinitrophenyl esters, and pentafluorophenyl esters;
- Rd is selected from phenyl, nitrophenyl, dinitrophenyl, carboxynitrophenyl, pyridyl or nitropyridyl; and
- n is an integer from 1 to 24; and the remainder of the variables are as described above in the eighth or the fifteenth specific embodiment.
In a twenty-first specific embodiment, the compound is represented by the following formula:
wherein:
-
- W′ is absent, or selected from —O—, —N(Re)—, —N(Re)—C(═O)—, —N(C(═O)Re)—, —S—, —CH2—S—, or —CH2NRe—;
- Rx is absent or selected from a linear, branched or cyclic alkyl having 1 to 10 carbon atoms;
- Re is —H, a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms or —(CH2—CH2—O)n—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms, optionally bearing a secondary amino (e.g., —NHR101) or tertiary amino (—NR101R102) group or a 5 or 6-membered nitrogen containing heterocycle, such as piperidine or morpholine, wherein R101 and R102 are each independently a linear, branched, or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms;
- n is an integer from 2 to 6;
- Zs is linked to the CBA, and is selected from:
- a bond;
wherein:
-
- q is an integer from 1 to 5; and,
- M is —H or a cation, such as Na+ or K+.
In certain embodiments, Zs is represented by any one of the following formulas:
In certain embodiments, W′ is —N(Re)—.
In certain embodiments, Re is —(CH2—CH2—O)n—Rk, wherein Rk is a —H, a linear, branched cyclic alkyl having 1 to 6 carbon atoms.
In certain embodiments, Rk is —H or -Me, n is 4, and q is 2.
In certain embodiments, Rx is a linear or branched alkyl having 1 to 6 carbon atoms.
In certain embodiments, Rx is —(CH2)p—(CRfRg)—, wherein Rf and Rg are each independently selected from H or a linear or branched alkyl having 1 to 4 carbon atoms; and p is 0, 1, 2 or 3.
In certain embodiments, Rf and Rg are the same or different, and are selected from —H and -Me; and p is 1.
In a twenty-second specific embodiment, the conjugate of formula (VIII), (IX), (X) and (XI) described in the twenty-first specific embodiment, the variables are as described below:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is —H; Y is —H, —OH or —SO3M (e.g., Y is —OH or —SO3M);
- M is —H or a pharmaceutically acceptable cation (e.g., Na+);
- X′ and Y′ are both —H;
- A and A′ are both —O—;
- R6 is —OMe; and
- Rx is a linear or branched alkyl having 1 to 6 carbon atoms.
In a twenty-third specific embodiment, for compounds of formula (IB), (IIB), (IIIB) and (IVB) described in the twentieth specific embodiment, the variables are as described below:
-
- the double line between N and C represents a single bond or a double bond, provided that when it is a double bond X is absent and Y is —H, and when it is a single bond, X is —H; Y is —H, —OH or —SO3M (e.g., Y is —OH or —SO3M);
- M is —H or Na+;
- X′ and Y′ are both —H;
- A and A′ are both —O—;
- R6 is —OMe;
- Rx is a linear or branched alkyl having 1 to 6 carbon atoms.
Preferably, Rx is —(CH2)p—(CRfRg)—, wherein Rf and Rg are each independently selected from —H or a linear or branched alkyl having 1 to 4 carbon atoms; p is 0, 1, 2 or 3. More preferably, Rf and Rg are the same or different, and are selected from —H and -Me; and p is 1.
In any of the specific embodiments for the conjugate of the invention above, such as the fourteenth to the twenty-third specific embodiments, the double line between N and C may represent a double bond.
In any of the specific embodiments for the conjugate of the invention above, such as the fourteenth to the twenty-third specific embodiments, the double line between N and C may represent a single bond, X is —H, the linking group, or an amine protecting group (e.g., X is —H); and Y is —H or selected from —OR, —OCOR′, —SR, —NR′R″, an optionally substituted 5- or 6-membered nitrogen-containing heterocycle, —SO3M, —SO2M and a sulfate —OSO3M. In certain embodiments, Y is not —H.
In certain embodiments, Y is selected from —H, —SO3M, —OH, —OMe, —OEt or —NHOH (e.g., Y is —SO3M, —OH, —OMe, —OEt or —NHOH).
In certain embodiments, Y is —H, —SO3M or —OH (e.g., Y is —SO3M or —OH).
In certain embodiments, M is —H, Na+ or K+.
In any of the specific embodiments for the conjugate of the invention above, such as the fourteenth to the twenty-third specific embodiments, W, when present, is C═O.
In any of the specific embodiments for the conjugate of the invention above, such as the fourteenth to the twenty-third specific embodiments, Z and Z′, when present, are —CH2—.
In any of the specific embodiments for the conjugate of the invention above, such as the fourteenth to the twenty-third specific embodiments, X′ is selected from the group consisting of —H, —OH, an optionally substituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms, phenyl, the linking group, and an amine-protecting group.
In certain embodiments, X′ is —H, —OH, -Me or the linking group.
In certain embodiments, X′ is —H.
In any of the specific embodiments for the conjugate of the invention above, such as the fourteenth to the twenty-third specific embodiments, Y′ is selected from the group consisting of —H, an oxo group, a substituted or unsubstituted linear, branched or cyclic alkyl, alkenyl or alkynyl having from 1 to 10 carbon atoms.
In certain embodiments, Y′ is —H or oxo.
In certain embodiments, Y′ is —H.
In any of the specific embodiments for the conjugate of the invention above, such as the fourteenth to the twenty-third specific embodiments, A and A′ are the same or different, and are selected from —O—, —S—, —N(R5)—, and oxo (C═O).
In certain embodiments, A and A′ are the same or different, and are selected from —O— and —S—.
In certain embodiments, A and A′ are —O—.
In any of the specific embodiments for the conjugate of the invention above, such as the fourteenth to the twenty-third specific embodiments, D and D′, when present, are the same or different, and are independently selected from a polyethylene glycol unit (—OCH2CH2)n, wherein n is an integer from 1 to 24, an amino acid, a peptide bearing 2 to 6 amino acids, or a linear, branched or cyclic alkyl, alkenyl or alkynyl having 1 to 10 carbon atoms, wherein the alkyl, alkenyl and alkynyl are optionally substituted with one or more substituents independently selected from the group consisting of halogen, —OR, —NR′COR″, —SR and —COR′.
In certain embodiments, D and D′ are linear or branched alkyl bearing 1 to 4 carbon atoms.
In a twenty-fourth specific embodiment, the conjugate of the present invention as described in the fourteenth, fifteenth, or the twenty-first specific embodiment is represented by the following:
-
- the double line between N and C represents a double bond;
- Y is —H;
- W is C═O;
- R1, R2, R1′, R2′, R4 and R4′ are —H;
- one of R3, or R3′ is optionally the linking group and the other is —H;
- R6 is —OMe;
- Z and Z′ are —CH2
- X′ is —H;
- Y′ is —H; and
- A and A′ are —O—.
In certain embodiments, the conjugate of any one of the described embodiments, such as the fourteenth to the twenty-fourth specific embodiments, may comprise 1-10 cytotoxic compounds, 2-9 cytotoxic compounds, 3-8 cytotoxic compounds, 4-7 cytotoxic compounds, or 5-6 cytotoxic compounds, each cytotoxic compound comprising the linking group linking the cytotoxic compound to the CBA, and each cytotoxic compound on the conjugate is the same.
In any of the conjugates embodiments, such as the fourteenth to the twenty-fourth specific embodiments, the cell-binding agent may bind to target cells selected from tumor cells, virus infected cells, microorganism infected cells, parasite infected cells, autoimmune cells, activated cells, myeloid cells, activated T-cells, B cells, or melanocytes; cells expressing the CD4, CD6, CD19, CD20, CD22, CD30, CD33, CD37, CD38, CD40, CD44, CD56, EpCAM, CanAg, CALLA, or Her-2 antigens; Her-3 antigens; or cells expressing insulin growth factor receptor, epidermal growth factor receptor, and folate receptor.
In any of the conjugates embodiments, such as the fourteenth to the twenty-fourth specific embodiments, the cell-binding agent may be an antibody, a single chain antibody, an antibody fragment that specifically binds to the target cell, a monoclonal antibody, a single chain monoclonal antibody, or a monoclonal antibody fragment that specifically binds to a target cell, a chimeric antibody, a chimeric antibody fragment that specifically binds to the target cell, a domain antibody, a domain antibody fragment that specifically binds to the target cell, a lymphokine, a hormone, a vitamin, a growth factor, a colony stimulating factor, or a nutrient-transport molecule.
The antibody may be a resurfaced antibody, a resurfaced single chain antibody, or a resurfaced antibody fragment.
The antibody may be a monoclonal antibody, a single chain monoclonal antibody, or a monoclonal antibody fragment thereof.
The antibody may be a humanized antibody, a humanized single chain antibody, or a humanized antibody fragment.
The invention further provides a pharmaceutical composition comprising any of the conjugates described herein, and a pharmaceutically acceptable carrier.
The invention further provides a drug-linker compound comprising any of the subject compound covalently linked to a bifunctional linker.
The invention additional provides a conjugate comprising any of the subject compounds, or the subject drug-linker compounds, linked to a cell-binding agent.
The invention further provides a method of inhibiting abnormal cell growth or treating a proliferative disorder, an autoimmune disorder, destructive bone disorder, infectious disease, viral disease, fibrotic disease, neurodegenerative disorder, pancreatitis or kidney disease in a mammal comprising administering to the mammal a therapeutically effective amount of any of the compounds (with or without any linker group) or conjugates of the invention, and, optionally, a second chemotherapeutic agent.
In certain embodiments, the compound or the conjugate is:
-
- wherein r is an integer from 1 to 10, Y is —H or —SO3M (e.g., Y is —SO3M), and M is —H or a pharmaceutically acceptable cation.
In certain embodiments, the second chemotherapeutic agent is administered to the mammal sequentially or consecutively.
In certain embodiments, the method is for treating a condition selected from cancer, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, lupus, myositis, infection, and immune deficiency.
In certain embodiments, the method or conjugate is for treating a cancer.
In certain embodiments, the cancer is selected from breast cancer, colon cancer, brain cancer, prostate cancer, kidney cancer, pancreatic cancer, ovarian cancer, head and neck cancer, melanoma, colorectal cancer, gastric cancer, squamous cancer, small-cell lung cancer, non small-cell lung cancer, testicular cancer, Merkel cell carcinoma, glioblastoma, neuroblastoma, cancers of lymphatic organs and hematological malignancy including Leukemia (Acute lymphoblastic leukemia (ALL), Acute myelogenous leukemia (AML), Chronic lymphocytic leukemia (CLL), Chronic myelogenous leukemia (CML), Acute monocytic leukemia (AMOL), Hairy cell leukemia (HCL), T-cell prolymphocytic leukemia (T-PLL), Large granular lymphocytic leukemia, Adult T-cell leukemia), Lymphoma (small lymphocytic lymphoma (SLL), Hodgkin's lymphomas (Nodular sclerosis, Mixed cellularity, Lymphocyte-rich, Lymphocyte depleted or not depleted, and Nodular lymphocyte-predominant Hodgkin lymphoma), Non-Hodgkin's lymphomas (all subtypes), Chronic lymphocytic leukemia/Small lymphocytic lymphoma, B-cell prolymphocytic leukemia, Lymphoplasmacytic lymphoma (such as Waldenstrim macroglobulinemia), Splenic marginal zone lymphoma, Plasma cell neoplasms (Plasma cell myeloma, Plasmacytoma, Monoclonal immunoglobulin deposition diseases, Heavy chain diseases), Extranodal marginal zone B cell lymphoma (MALT lymphoma), Nodal marginal zone B cell lymphoma (NMZL), Follicular lymphoma, Mantle cell lymphoma, Diffuse large B cell lymphoma, Mediastinal (thymic) large B cell lymphoma, Intravascular large B cell lymphoma, Primary effusion lymphoma, Burkitt lymphoma/leukemia, T cell prolymphocytic leukemia, T cell large granular lymphocytic leukemia, Aggressive NK cell leukemia, Adult T cell leukemia/lymphoma, Extranodal NK/T cell lymphoma (nasal type), Enteropathy-type T cell lymphoma, Hepatosplenic T cell lymphoma, Blastic NK cell lymphoma, Mycosis fungoides/Sezary syndrome, Primary cutaneous CD30-positive T cell lymphoproliferative disorders, Primary cutaneous anaplastic large cell lymphoma, Lymphomatoid papulosis, Angioimmunoblastic T cell lymphoma, Peripheral T cell lymphoma (unspecified), Anaplastic large cell lymphoma), multiple myeloma (plasma cell myeloma or Kahler's disease).
Production of Cell-Binding Agent-Drug Conjugates
In order to link the cytotoxic compounds or derivative thereof of the present invention to the cell-binding agent, the cytotoxic compound may comprise a linking moiety with a reactive group bonded thereto. In one embodiment, a bifunctional crosslinking reagent can be first reacted with the cytotoxic compound to provide the compound bearing a linking moiety with one reactive group bonded thereto (i.e., drug-linker compound), which can then react with a cell binding agent. Alternatively, one end of the bifunctional crosslinking reagent can first react with the cell binding agent to provide the cell binding agent bearing a linking moiety with one reactive group bonded thereto, which can then react with a cytotoxic compound. The linking moiety may contain a chemical bond that allows for the release of the cytotoxic moiety at a particular site. Suitable chemical bonds are well known in the art and include disulfide bonds, thioether bonds, acid labile bonds, photolabile bonds, peptidase labile bonds and esterase labile bonds (see for example U.S. Pat. Nos. 5,208,020; 5,475,092; 6,441,163; 6,716,821; 6,913,748; 7,276,497; 7,276,499; 7,368,565; 7,388,026 and 7,414,073). Preferred are disulfide bonds, thioether and peptidase labile bonds. Other linkers that can be used in the present invention include non-cleavable linkers, such as those described in are described in detail in U.S. publication number 2005/0169933, or charged linkers or hydrophilic linkers and are described in US 2009/0274713, US 2010/01293140 and WO 2009/134976, each of which is expressly incorporated herein by reference, each of which is expressly incorporated herein by reference.
The compounds of formula (I)-(IV), (IA)-(IVA), and (IB)-(IVB) can be linked through R1, R2, R3, R4, R1′, R2′, R3′, R4′, L′, L″, L′″, or X (when present). Of these, preferred linkable groups are R2′, R3′, R4′, L′, L″, L′″ and most preferred linkable groups are R2′, R3′, and L′. Examples of linking groups for compounds of formula (I)-(IV), (IA)-(IVA), and (IB)-(IVB) are described above.
In one embodiment, a solution of an antibody in aqueous buffer may be incubated with a molar excess of an antibody modifying agent such as N-succinimidyl-3-(2-pyridyldithio)propionate (SPDP) or with N-succinimidyl-4-(2-pyridyldithio)butanoate (SPDB) to introduce dithiopyridyl groups. The modified antibody is then reacted with the thiol-containing cytotoxic compound, such as compound 2a, to produce a disulfide-linked antibody-indolinobenzodiazepine dimer conjugate. The cell binding agent-drug conjugate may then be purified using any purification methods known in the art, such as those described in U.S. Pat. No. 7,811,572 and US Publication No. 2006/0182750, both of which are incorporated herein by reference. For example, the cell-binding agent-drug conjugate can be purified using tangential flow filtration, adsorptive chromatography, adsorptive filtration, selective precipitation, non-absorptive filtration or combination thereof. Preferably, tangential flow filtration (TFF, also known as cross flow filtration, ultrafiltration and diafiltration) and/or adsorptive chromatography resins are used for the purification of the conjugates.
Alternatively, the antibody may be incubated with a molar excess of an antibody modifying agent such as 2-iminothiolane, L-homocysteine thiolactone (or derivatives), or N-succinimidyl-S-acetylthioacetate (SATA) to introduce sulfhydryl groups. The modified antibody is then reacted with the appropriate disulfide-containing cytotoxic agent, to produce a disulfide-linked antibody-cytotoxic agent conjugate. The antibody-cytotoxic agent conjugate may then be purified by methods described above. The cell binding may also be engineered to introduce thiol moieties, such as cysteine-engineered antibodies disclosed in U.S. Pat. Nos. 7,772,485 and 7,855,275.
In another embodiment, a solution of an antibody in aqueous buffer may be incubated with a molar excess of an antibody-modifying agent such as N-succinimidyl-4-(N-maleimidomethyl)-cyclohexane-1-carboxylate to introduce maleimido groups, or with N-succinimidyl-4-(iodoacetyl)-aminobenzoate (SIAB) to introduce iodoacetyl groups. The modified antibody is then reacted with the thiol-containing cytotoxic agent to produce a thioether-linked antibody-cytotoxic conjugate. The antibody-cytotoxic conjugate may then be purified by methods described above.
The number of cytotoxic molecules bound per antibody molecule can be determined spectrophotometrically by measuring the ratio of the absorbance at 280 nm and 330 nm. An average of 1-10 cytotoxic compounds/antibody molecule(s) can be linked by the methods described herein. The preferred average number of linked cytotoxic compounds per antibody molecule is 2-5, and the most preferred is 2.5-4.0.
Cytotoxic agents containing linkers terminating in an N-hydroxy succinimidyl (NHS) ester, such as compounds 1g and 10, can react with the antibody to produce direct amide linked conjugates such as huMy9-6-SPDB-1f or huMy9-6-BMPS-1f. The antibody-cytotoxic agent conjugate may then be purified by gel-filtration by any methods described above.
Representative processes for preparing the cell-binding agent-drug conjugates of the present invention are shown in FIGS. 22 and 23 . A cytotoxic dimer compound of the present invention can be conjugated with a cell binding agent through either a one-step or a two-step conjugation method. In FIGS. 22a and 22b , representative examples are described, wherein a dimer compound that possesses a linker such as an N-hydroxysuccinimide ester is reacted directly with a cell binding agent, such as an antibody, generating the desired conjugate. In FIG. 22c linkable dimer 1g was first treated with sodium bisulfate to provide a modified dimer compound 26 before adding antibody to form the conjugate huMy9-6-SBDP-1f of the present invention.
A representative example of a two-step conjugation method is described in FIG. 23 , wherein an antibody is first modified with a bifunctional crosslinking agent resulting in an antibody that possesses a desired number of linkers suitable for reaction with a dimer compound having a free thiol moiety. In this example the antibody huMy9-6 was first modified with SPDB to give an antibody with linkers containing the dithiopyridyl moiety. The modified antibody was then exposed to a free thiol, such as 2a, generating the desired conjugate huMy9-6-SPDB-2a.
Processes for synthesizing the drug-linker compounds and conjugates of the invention are also described in U.S. provisional patent application No. 61/443,092, filed on Feb. 15, 2011, and a U.S. utility application claiming the benefit of filing date thereof and filed on the same day of the instant application, entitled “METHODS OF PREPARATION OF CONJUGATES,” the entire contents of which applications, including all drawings, formulae, synthesis schemes, specifications, and claims, are incorporated herein by reference.
The structures of representative compounds and conjugates of the present invention are shown in Tables 1-8. These compounds and conjugates can be prepared according to the methods described herein.
| TABLE 1 |
| Structures of representative compounds in the present invention. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Notes: |
| n = 1 or 3; m = 3 or 4 |
| W = OH, OMe, ONHS, NHNH2, H, Me, Ph, Peptide |
| X = CH2, O, S, NH or NMe |
| Y = CH2 or absent |
| Z″ = H, Me, SMe, S(CH2)3C(O)NHS or CH2C(O)NHS or BMPS or SMCC or SPy or SPy—NO2 |
| TABLE 2 |
| Structures of representative compounds in the present invention (Continued). |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Note: |
| n = 1, 2 or 3 |
| m = 3 or 4 |
| W = OH, OMe, ONHS, NHNH2, H, Me, Ph, Peptide |
| X = CH2, O, S, NH, NMe |
| Y = absent or CH2 |
| Z = CH or N |
| Z″ = H, Me, SMe, S(CH2)3C(O)NHS or CH2C(O)NHS or BMPS or SMCC or SPy or SPy—NO2 |
| TABLE 3 |
| Structures of representative compounds in the present invention (Continued). |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Note: |
| n = 1, 2 or 3 |
| m = 3 or 4 |
| W = OH, OMe, ONHS, NHNH2, H, Me, Ph, Peptide |
| X = CH2, O, S, NH, NMe |
| Z = CH or N |
| Z″ = H, Me, SMe, S(CH2)3C(O)NHS or CH2C(O)NHS or BMPS or SMCC or SPy or SPy—NO2 |
| TABLE 4 |
| Structures of representative compounds in the present invention (Continued). |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Note: |
| n = 1, 2 or 3 |
| m = 3 or 4 |
| W = OH, OMe, ONHS, NHNH2, H, Me, Ph, Peptide |
| X = CH2, O, S, NH, NMe |
| Z = CH or N |
| Z″ = H, Me, SMe, S(CH2)3C(O)NHS or CH2C(O)NHS or BMPS or SMCC or SPy or SPy—NO2 |
| TABLE 8 |
| Structures of representative conjugates of the present invention. |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
In Vitro Cytotoxicity of Compounds and Conjugates
The cytotoxic compounds and cell-binding agent-drug conjugates of the invention can be evaluated for their ability to suppress proliferation of various cancer cell lines in vitro. For example, cell lines such as the human colon carcinoma line COLO 205, the rhabdomyosarcoma cell line RH-30, and the multiple myeloma cell line MOLP-8 can be used for the assessment of cytotoxicity of these compounds and conjugates. Cells to be evaluated can be exposed to the compounds or conjugates for 1-5 days and the surviving fractions of cells measured in direct assays by known methods. IC50 values can then be calculated from the results of the assays. Alternatively or in addition, an in vitro cell line sensitivity screen, such as the one described by the U.S. National Cancer Institute (see Voskoglou-Nomikos et al., 2003, Clinical Cancer Res. 9: 42227-4239, incorporated herein by reference) can be used as one of the guides to determine the types of cancers that may be sensitive to treatment with the compounds or conjugates of the invention.
Examples of in vitro potency and target specificity of antibody-cytotoxic agent conjugates of the present invention are shown in FIGS. 25-26 . All of the conjugates are extremely cytotoxic on the antigen positive cancer cells with an IC50 in the low picomolar range. Antigen negative cell lines remained viable when exposed to the same conjugates. The indolinobenzodiazepine dimers showed target specific potency being 160 fold less potent when blocked with unconjugated antibody huMy9-6 (anti-CD33) and 40 less potent when blocked with unconjugated antibody FOLR1 (anti-folate receptor antibody). For example, the huMy9-6-SPDB-1f conjugate killed antigen positive HL60/QC cells with an IC50 value of 10.5 pM, while the addition of an excess of unconjugated huMy9-6 antibody reduced this cytotoxic effect (IC50=1.69 nM), demonstrating antigen specificity (FIG. 25A ). In addition, the huMy9-6-SPDB-1f conjugate is also highly potent towards both the HL60/ATCC cell line with an IC50 value of 21 pM and the NB-4 cell line with an IC50 value of 190 pM (FIGS. 25B and 25C ).
Similarly, the huFOLR1-SPDB-1f conjugate was highly potent, with an IC50 value of 55 pM for antigen positive KB cells (FIG. 26 ). Addition of an excess of unconjugated huFOLR1 antibody reduced this cytotoxic effect >40 fold, demonstrating antigen-specificity.
The effect of conjugation on antibody binding was measured by comparing the binding of both unconjugated huMy9-6 antibody and the huMy9-6-SPDB-1f conjugate towards the HL60/QC cell line (FIG. 27 ). FACS analysis revealed that there is no change in binding capability of the conjugate to naked antibody indicating that there is no compromise in binding due to conjugation of the cytotoxic agent to the antibody.
In one example, in vivo efficacy of a cell binding agent/cytotoxic agent conjugate was measured. Nude mice bearing human HL60/QC tumors were treated with huMy9-6-SPDB-1f conjugate and significant tumor regression was observed at multiple doses while untreated mice grew tumors rapidly (FIG. 28 ). Activity was observed at doses as low as 20 μg/kg which is at least 35-fold lower than the maximum tolerated dose.
The effect of imine saturation towards tolerability is shown in Table 9. Di-imine huFOLR1-drug1 was tested at multiple doses all of which were found to be highly toxic leaving only survivors in the lowest group tested at 50 μg/kg. In contrast the partially reduced mono-imine huFOLR1-Drug 2 and huFOLR1-SPDB-IGN (huFOLR1-SPDB-1f) conjugates were found to have significantly improved tolerability with the huFOLR1-SPDB-IGN (huFOLR1-SPDB-1f) conjugate showing 100% animal survival at the highest doses tested of 560 μg/kg.
Compositions and Methods of Use
The present invention includes a composition (e.g., a pharmaceutical composition) comprising novel benzodiazepine compounds described herein (e.g., indolinobenzodiazepine or oxazolidinobenzodiazepine), derivatives thereof, or conjugates thereof, (and/or solvates, hydrates and/or salts thereof) and a carrier (a pharmaceutically acceptable carrier). The present invention also includes a composition (e.g., a pharmaceutical composition) comprising novel benzodiazepine compounds described herein, derivatives thereof, or conjugates thereof, (and/or solvates, hydrates and/or salts thereof) and a carrier (a pharmaceutically acceptable carrier), further comprising a second therapeutic agent. The present compositions are useful for inhibiting abnormal cell growth or treating a proliferative disorder in a mammal (e.g., human). The present compositions are also useful for treating depression, anxiety, stress, phobias, panic, dysphoria, psychiatric disorders, pain, and inflammatory diseases in a mammal (e.g., human).
The present invention includes a method of inhibiting abnormal cell growth or treating a proliferative disorder in a mammal (e.g., human) comprising administering to said mammal a therapeutically effective amount of novel benzodiazepine compounds described herein (e.g., indolinobenzodiazepine or oxazolidinobenzodiazepine), derivatives thereof, or conjugates thereof, (and/or solvates and salts thereof) or a composition thereof, alone or in combination with a second therapeutic agent.
The present invention also provides methods of treatment comprising administering to a subject in need of treatment an effective amount of any of the conjugates described above.
Similarly, the present invention provides a method for inducing cell death in selected cell populations comprising contacting target cells or tissue containing target cells with an effective amount of a cytotoxic agent comprising any of the cytotoxic compound-cell-binding agents (e.g., indolinobenzodiazepine or oxazolidinobenzodiazepine dimer linked to a cell binding agent) of the present invention, a salt or solvate thereof. The target cells are cells to which the cell-binding agent can bind.
If desired, other active agents, such as other anti-tumor agents, may be administered along with the conjugate.
Suitable pharmaceutically acceptable carriers, diluents, and excipients are well known and can be determined by those of ordinary skill in the art as the clinical situation warrants.
Examples of suitable carriers, diluents and/or excipients include: (1) Dulbecco's phosphate buffered saline, pH about 7.4, containing or not containing about 1 mg/mL to 25 mg/mL human serum albumin, (2) 0.9% saline (0.9% w/v NaCl), and (3) 5% (w/v) dextrose; and may also contain an antioxidant such as tryptamine and a stabilizing agent such as Tween 20.
The method for inducing cell death in selected cell populations can be practiced in vitro, in vivo, or ex vivo.
Examples of in vitro uses include treatments of autologous bone marrow prior to their transplant into the same patient in order to kill diseased or malignant cells: treatments of bone marrow prior to their transplantation in order to kill competent T cells and prevent graft-versus-host-disease (GVHD); treatments of cell cultures in order to kill all cells except for desired variants that do not express the target antigen; or to kill variants that express undesired antigen.
The conditions of non-clinical in vitro use are readily determined by one of ordinary skill in the art.
Examples of clinical ex vivo use are to remove tumor cells or lymphoid cells from bone marrow prior to autologous transplantation in cancer treatment or in treatment of autoimmune disease, or to remove T cells and other lymphoid cells from autologous or allogenic bone marrow or tissue prior to transplant in order to prevent GVHD. Treatment can be carried out as follows. Bone marrow is harvested from the patient or other individual and then incubated in medium containing serum to which is added the cytotoxic agent of the invention, concentrations range from about 10 μM to 1 pM, for about 30 minutes to about 48 hours at about 37° C. The exact conditions of concentration and time of incubation, i.e., the dose, are readily determined by one of ordinary skill in the art. After incubation the bone marrow cells are washed with medium containing serum and returned to the patient intravenously according to known methods. In circumstances where the patient receives other treatment such as a course of ablative chemotherapy or total-body irradiation between the time of harvest of the marrow and reinfusion of the treated cells, the treated marrow cells are stored frozen in liquid nitrogen using standard medical equipment.
For clinical in vivo use, the cytotoxic agent of the invention will be supplied as a solution or a lyophilized powder that are tested for sterility and for endotoxin levels. Examples of suitable protocols of conjugate administration are as follows. Conjugates are given weekly for 4 weeks as an intravenous bolus each week. Bolus doses are given in 50 to 1000 mL of normal saline to which 5 to 10 mL of human serum albumin can be added. Dosages will be 10 μg to 2000 mg per administration, intravenously (range of 100 ng to 20 mg/kg per day). After four weeks of treatment, the patient can continue to receive treatment on a weekly basis. Specific clinical protocols with regard to route of administration, excipients, diluents, dosages, times, etc., can be determined by one of ordinary skill in the art as the clinical situation warrants.
Examples of medical conditions that can be treated according to the in vivo or ex vivo methods of inducing cell death in selected cell populations include malignancy of any type including, for example, cancer of the lung (small cell and non-small cell), breast, colon, brain, prostate, kidney, pancreas, ovary, head and neck, skin (melanoma), Merkel cell carcinoma, glioblastoma, neuroblastoma, and cancers of lymphatic organs; autoimmune diseases, such as systemic lupus, rheumatoid arthritis, and multiple sclerosis; graft rejections, such as renal transplant rejection, liver transplant rejection, lung transplant rejection, cardiac transplant rejection, and bone marrow transplant rejection; graft versus host disease; viral infections, such as CMV infection, HIV infection, AIDS, etc.; and parasite infections, such as giardiasis, amoebiasis, schistosomiasis, and others as determined by one of ordinary skill in the art.
Cancer therapies and their dosages, routes of administration and recommended usage are known in the art and have been described in such literature as the Physician's Desk Reference (PDR). The PDR discloses dosages of the agents that have been used in treatment of various cancers. The dosing regimen and dosages of these aforementioned chemotherapeutic drugs that are therapeutically effective will depend on the particular cancer being treated, the extent of the disease and other factors familiar to the physician of skill in the art and can be determined by the physician. The contents of the PDR are expressly incorporated herein in its entirety by reference. One of skill in the art can review the PDR, using one or more of the following parameters, to determine dosing regimen and dosages of the chemotherapeutic agents and conjugates that can be used in accordance with the teachings of this invention. These parameters include:
Comprehensive index
By Manufacturer
Products (by company's or trademarked drug name)
Category index
Generic/chemical index (non-trademark common drug names)
Color images of medications
Product information, consistent with FDA labeling
Chemical information
Function/action
Indications & Contraindications
Trial research, side effects, warnings
Analogues and Derivatives
One skilled in the art of cytotoxic agents will readily understand that each of the cytotoxic agents described herein can be modified in such a manner that the resulting compound still retains the specificity and/or activity of the starting compound. The skilled artisan will also understand that many of these compounds can be used in place of the cytotoxic agents described herein. Thus, the cytotoxic agents of the present invention include analogues and derivatives of the compounds described herein.
All references cited herein and in the examples that follow are expressly incorporated by reference in their entireties.
The invention will now be illustrated by reference to non-limiting examples. Unless otherwise stated, all percents, ratios, parts, etc. are by weight. All reagents were purchased from the Aldrich Chemical Co., New Jersey, or other commercial sources. Nuclear Magnetic Resonance (1H NMR) spectra were acquired on a Bruker 400 MHz instrument and mass spectra were acquired on a Bruker Daltonics Esquire 3000 instrument using electrospray ionization.
To a stirred solution of the aniline 1a (1.55 g, 5.18 mmol) and 2-(methyldithio)-isobutyraldehyde (0.7 mL, 5.18 mmol) in anhydrous 1,2-dichloromethane (20 mL) was added sodium triacetoxyborohydride (1.1 g, 5.18 mmol) and zinc chloride powder (353 mg, 2.59 mmol) followed by the addition of anhydrous magnesium sulfate (800 mg). The mixture was stirred at room temperature (rt) for 6 hours then a second portion of 2-(methyldithio)-isobutyraldehyde (0.7 mL, 5.18 mmol) and sodium triacetoxyborohydride (1.1 g, 5.18 mmol) were added. It continued to be stirred at rt overnight. The reaction mixture was filtered through celite and washed with dichloromethane. The filtrate was concentrated and the remainder was purified by silica gel chromatography (Combiflash, 40 g column, dichloromethane/MeOH) to give compound 1b (487 mg y=22%) as colorless oil. Unreacted starting material aniline 1a (1.02 g) was also recovered in 65% yield. 1H NMR (400 Hz, CDCl3): δ 6.76 (s, 2H), 6.63 (s, 1H), 4.55 (s, 4H), 3.65-3.51 (m, 14H), 3.35 (s, 3H), 2.44 (s, 3H), 1.33 (s, 6H); 13C NMR (400 Hz, CDCl3): δ 149.0, 142.35, 114.0, 111.1, 71.98, 70.7, 70.6, 70.5, 67.6, 65.5, 59.75, 59.1, 53.9, 51.9, 26.6, 25.7, 20.75; MS (m/z): found 456.2 (M+Na)+. See FIG. 1 .
To a stirred solution of 1b (243 mg, 0.56 mmol) in anhydrous dichloromethane (3.5 mL) was added triethylamine (234 μl, 1.68 mmol). The mixture was cooled to −10° C. and methanesulfonyl chloride (113 μl, 1.46 mmol) was added slowly over 15 minutes via a syringe. The solution continued to be stirred for 60 minutes at −10˜−7° C. and quenched by addition of ice/water. It was diluted with ethyl acetate and washed with cold water. The organic layer was dried over anhydrous sodium sulfate, filtered, concentrated and high vacuumed to give the mesylates as light yellowish oil (340 mg). The mesylates was transferred into a 10 mL round-bottomed flask with ethyl acetate/dichloromethane, concentrated and high vacuumed. IBD monomer (412 mg, 1.4 mmol) was added followed by the addition of anhydrous dimethylformamide (3 mL) and anhydrous potassium carbonate (232 mg, 1.68 mmol). The obtained yellowish mixture was stirred at room temperature overnight. It was diluted with dichloromethane and washed with brine. The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated. The residue was dissolved in dichloromethane and loaded on silica gel column and eluted with dichloromethane/methanol (15:1 then 10:1). The fractions that contained compound 1c were combined and concentrated to give 705 mg of crude product which was further purified by preparative reverse phase HPLC (C18 column, eluted with acetonitrile/water) to give compound 1c as a yellowish fluffy solid (181 mg, y=33%). 1H NMR (400 Hz, CDCl3): δ 8.28 (d, J=8.0 Hz, 2H), 7.86 (d, J=3.6 Hz, 2H), 7.59 (s, 2H), 7.31-7.26 (m, 4H), 7.12 (t, J=7.6 Hz, 2H), 6.87-6.80 (m, 5H), 5.18 (dd, J1=20.8 Hz, J2=12.4 Hz, 4H), 4.50-4.47 (m, 2H), 3.99 (s, 6H), 3.75-3.48 (m, 18H), 3.37 (s, 3H), 2.44 (s, 3H), 1.32 (s, 6H); MS (m/z): found 1025.9 (M+H2O+Na)+, 1043.9 (M+2H2O+Na)+, 983.8 (M−H)−, 1055.8 (M+4H2O—H)−. See FIG. 1 .
To a stirred solution of compound 1c (112 mg, 0.114 mmol) in anhydrous dichloromethane (0.3 mL) and absolute ethanol (0.6 mL) was added sodium borohydride (0.9 mg, 0.023 mmol) at 0° C. The ice bath was removed after 5 minutes and the mixture was stirred at room temperature for 3 hours and then cooled to 0° C. and quenched with saturated ammonium chloride, diluted with dichloromethane, separated and the organic layer was washed with brine, dried over anhydrous Na2SO4 and filtered through celite and concentrated. The residue was purified by reverse phase HPLC (C18 column, acetonitrile/water). The corresponding fractions were extracted with dichloromethane and concentrated to obtain the products 1d, 1e and the unreacted starting material 1c. Compound 1d: 37.1 mg (y=33%), MS (m/z): found 1010.4 (M+Na)+, 1028.4 (M+H2O+Na)+, 1040.3 (M+3H2O—H)−; compound 1e: 6.4 mg (y=5.7%), MS (m/z): found 1012.4 (M+Na)+; compound 1c: 44.1 mg (y=39%). See FIG. 1 .
To a stirred solution of 1d (23.6 mg, 0.024 mmol) in acetonitrile (3 mL) and methanol (3 mL) was added freshly prepared TCEP solution (17 mg of TCEP HCl salt was neutralized with saturated sodium bicarbonate to pH 6-6.5 then diluted with 0.5 mL of pH 6.5 phosphate buffer) at room temperature. The mixture was stirred at room temperature for 3 hours and then diluted with dichloromethane and deionized water, separated and the organic layer was washed with brine, dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated and high vacuumed to give 22 mg of 1f as light yellowish foam. Another 18 mg of 1f was prepared from 19 mg of 1d following the same procedure. The combined 40 mg (0.042 mmol) of 1f was dissolved in anhydrous dichloromethane (0.5 mL) and stirred. To this stirred solution was added SPDB NHS ester 2 (34.6 mg, 80% purity, 0.085 mmol) and diisopropylethylamine (15 μl, 0.085 mmol). It continued to be stirred at room temperature overnight, quenched with saturated ammonium chloride and diluted with dichloromethane, separated and washed with brine, dried over sodium sulfate, filtered and concentrated. The residue was purified by preparative reverse phase HPLC (C18 column, acetonitrile/water). The fractions containing product were combined, extracted with dichloromethane and concentrated to give compound 1g as white solid (29.7 mg, y=60%). 1H NMR (400 Hz, CD3CN): δ 8.28-8.25 (m, 1H), 8.20-8.17 (m, 1H), 7.87-7.84 (m, 1H), 7.49 (d, J=4.4 Hz, 1H), 7.39 (d, J=4.4 Hz, 1H), 7.31-7.19 (m, 4H), 7.13-7.01 (m, 2H), 6.92-6.87 (m, 3H), 6.77 (bs, 1H), 6.31-6.29 (m, 1H), 5.16-5.09 (m, 2H), 5.00 (d, J=4.4 Hz, 2H), 4.94 (bs, —NH), 4.48-4.43 (m, 1H), 4.40-4.34 (m, 1H), 3.90 (d, J=4.4 Hz, 3H), 3.77 (d, J=4.4 Hz, 3H), 3.64-3.39 (m, 18H), 3.26 (d, J=4.4 Hz, 3H), 2.82-2.70 (m, 8H), 2.17 (d, J=4.4 Hz, 1H), 2.08-2.01 (m, 3H), 1.30 (d, J=4.4 Hz, 6H); MS (m/z): found 1025.9 (M+H2O+Na)+, 1043.9 (M+2H2O+Na)+, 983.8 (M−H)−, 1055.8 (M+4H2O—H)−; MS (m/z), found 1179.5 (M+Na)+. See FIG. 1 .
To a stirred solution of 1c (8 mg, 0.0081 mmol) in anhydrous 1,2-dichloromethane (0.2 mL) was added sodium triacetoxyborohydride (3.8 mg, 0.018 mmol). The mixture was stirred at room temperature for 1.5 hours, then the mixture was diluted with dichloromethane and quenched with saturated sodium bicarbonate, separated and the organic layer was washed with brine, dried over sodium sulfate, and filtered. The filtrate was concentrated and the remainder was purified by reverse phase HPLC (C18 column, acetonitrile/water) to give compound 1e as a white solid (4.7 mg, y=58%). MS (m/z), found 1012.4 (M+Na)+, 1024.2 (M+2H2O—H)−. See FIG. 2 .
To a stirred solution of compound 1e (12 mg, 0.012 mmol) in acetonitrile (1 mL) and methanol (3 mL) was added freshly prepared TCEP solution (11 mg of TCEP HCl salt was neutralized with saturated sodium bicarbonate to pH ˜6.5 then diluted with 0.4 mL of pH 6.5 phosphate buffer) at room temperature. The mixture was stirred at room temperature for 3.5 hours and then diluted with dichloromethane and deionized water, separated and the organic layer was washed with brine, dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated and the remainder was purified by reverse phase HPLC (C18 column, acetonitrile/water) to give compound 2a as a white solid (4.9 mg, y=43%). MS (m/z), found 966.4 (M+Na)+, 978.2 (M+2H2O—H)−. See FIG. 2 .
To a solution of compound 3a (830 mg, 1.9 mmol) in methanol (15 mL) was added Pd/C (10%, 204 mg, 0.19 mmol). The air in the flask was removed by vacuum and then replaced with hydrogen in a balloon. The mixture was stirred at room temperature overnight. The mixture was filtered through celite and washed the celite/Pd/C with dichloromethane and methanol. The filtrate was concentrated and the residue diluted with dichloromethane and evaporated for a few cycles and then was purified by silica gel chromatography (dichloromethane/methanol) to give compound 3b as a light yellowish solid (558 mg, y=98%). 1H NMR (400 Hz, CDCl3): δ 8.34 (d, J=8.0 Hz, 1H), 7.40 (s, 1H), 7.22 (dd, J1=8.0 Hz, J2=7.6 Hz, 1H), 7.17 (d, J=7.2 Hz, 1H), 7.02 (dd, J1=7.2 Hz, J2=7.6 Hz, 1H), 6.16 (s, 1H), 4.37 (tt, J1=10.4 Hz, J2=7.2 Hz, 1H), 3.76 (s, 3H), 3.49-3.36 (m, 3H), 2.73 (dd, J1=16.8 Hz, J2=3.6 Hz, 1H); 13C NMR (400 Hz, CDCl3): δ 167.0, 150.4, 142.6, 141.2, 140.8, 129.9, 127.7, 124.8, 123.96, 117.4, 113.7, 112.5, 104.7, 57.3, 56.3, 54.7, 33.0; MS (m/z), found 295.1 (M−H)−. See FIG. 3 .
To a solution of the 2-(methyldithio)-isobutyraldehyde (113 mg, 0.75 mmol) and compound 3b (148 mg, 0.5 mmol) in anhydrous 1,2-dichloroethane (2 mL) was added sodium triacetoxyborohydride (212 mg, 1.0 mmol). The mixture was stirred at room temperature for 2 days. During the time, another two portions (0.05 mL, 0.5 mmol/portion) of 2-(methyldithio)-isobutyraldehyde along with one portion of sodium triacetoxyborohydride (106 mg, 0.5 mmol) were added. The reaction was quenched with saturated sodium bicarbonate, diluted with dichloromethane and water. The organic layer was washed with brine, dried over sodium sulfate and filtered. The filtrate was concentrated under reduced pressure and the residue was purified by silica gel chromatography (Combiflash, 24 g column, hexanes/ethyl acetate) to give compound 3c as a white fluffy solid (92.5 mg, y=43%). Unreacted starting material 3b was also recovered (49.3 mg, y=33%). 1H NMR (400 Hz, CDCl3): δ 8.30 (d, J=8.0 Hz, 1H), 7.28 (dd, J1=6.8 Hz, J2=7.6 Hz, 1H), 7.25-7.20 (m, 2H), 7.07 (t, J=7.6 Hz, 1H), 6.80 (s, 1H), 6.17 (s, 1H), 4.36-4.28 (m, 1H), 3.89 (s, 3H), 3.78 (d, J=14.4 Hz, 1H), 3.46-3.34 (m, 3H), 2.90 (d, J=14.4 Hz, 1H), 2.73 (dd, J1=16.4 Hz, J2=2.8 Hz, 1H), 2.34 (s, 3H), 1.17 (s, 3H), 1.05 (s, 3H); 13C NMR (400 Hz, CDCl3): δ 167.2, 149.0, 142.5, 142.2, 141.9, 129.9, 128.0, 125.3, 124.5, 124.1, 117.1, 112.0, 108.5, 64.8, 61.4, 58.1, 56.3, 53.4, 32.0, 26.3, 25.7, 25.4; MS (m/z), found 453.3 (M+Na)+, 429.2 (M−H)−. See FIG. 3 .
To a stirred solution of IBD monomer (125 mg, 0.425 mmol) and 1,5-diiodopentane (0.63 mL, 4.25 mmol) in anhydrous dimethylformamide (3 mL) was added potassium carbonate (59 mg, 0.425 mmol) and the mixture was stirred at room temperature overnight. The reaction solution was diluted with dichloromethane, washed with brine and dried over anhydrous sodium sulfate. It was filtered and concentrated. The residue was purified by silica gel chromatography (hexanes/ethyl acetate) to give compound 3d as yellowish foam (94 mg, y=45%). 1H NMR (400 Hz, CDCl3): δ 8.27 (d, J=8.0 Hz, 1H), 7.86 (d, J=4.8 Hz, 1H), 7.56 (s, 1H), 7.27 (dd, J1=8.4 Hz, J2=7.6 Hz, 2H), 7.10 (dd, J1=7.6 Hz, J2=7.2 Hz, 1H), 6.82 (s, 1H), 4.48 (dt, J1=10.8 Hz, J2=4.4 Hz, 1H), 4.15-4.07 (m, 2H), 3.96 (s, 3H), 3.70 (dd, J1=16.8 Hz, J2=10.8 Hz, 1H), 3.49 (dd, J1=16.8 Hz, J2=4.0 Hz, 1H), 3.22 (t, J=7.2 Hz, 2H), 1.96-1.87 (m, 4H), 1.64-1.57 (m, 2H); 13C NMR (400 Hz, CDCl3): δ 164.0, 163.2, 151.4, 148.3, 142.2, 140.3, 129.6, 128.3, 124.9, 120.5, 117.0, 112.0, 110.6, 68.8, 56.4, 55.1, 33.3, 32.7, 28.0, 27.2, 6.6; MS (m/z), found 513.3 (M+Na)+, 543.2 (M+3H2O—H)−. See FIG. 3 .
To a stirred solution of the starting materials 3c (91 mg, 0.21 mmol) and 3d (94 mg, 0.19 mmol) in anhydrous dimethylformamide (1 mL) was added potassium carbonate (29 mg, 0.21 mmol) and the mixture was stirred at room temperature overnight. The reaction solution was diluted with dichloromethane, washed with brine and dried over anhydrous sodium sulfate. It was filtered, concentrated and the residue was purified by silica gel chromatography (hexanes/ethyl acetate) to give compound 3e as yellowish foam (89.1 mg, y=58%). 1H NMR (400 Hz, CDCl3): δ 8.32-8.28 (m, 2H), 7.91 (bs, 1H), 7.57 (s, 1H), 7.36-7.21 (m, 5H), 7.15-7.05 (m, 2H), 6.85 (s, 1H), 6.74 (s, 1H), 4.53-4.48 (m, 1H), 4.37-4.31 (m, 1H), 4.21-4.03 (m, 4H), 3.98 (s, 3H), 3.88 (s, 3H), 3.86-3.70 (m, 2H), 3.55-3.35 (m, 4H), 2.93 (d, J=4.0 Hz, 1H), 2.73 (dd, J1=16.4 Hz, J2=2.4 Hz, 1H), 2.36 (s, 3H), 2.03-1.96 (m, 3H), 1.77-1.67 (m, 3H), 1.21 (s, 3H), 1.06 (s, 3H); MS (m/z), found 815.3 (M+Na)+. See FIG. 3 .
To a stirred solution of compound 3e (33.1 mg, 0.042 mmol) in acetonitrile (2 mL) and methanol (4 mL) was added freshly prepared TCEP solution (36 mg of TCEP HCl salt was neutralized with saturated sodium bicarbonate to pH ˜6.5 then diluted with 0.4 mL of pH 6.5 phosphate buffer) at room temperature. The mixture was stirred at room temperature for 3 hours and then diluted with dichloromethane and deionized water, separated and the organic layer was washed with brine, dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated and high vacuumed to give 31 mg of compound 3f as yellowish solid. It was dissolved in anhydrous dichloromethane (0.5 mL). SPDB NHS ester 2 (26 mg, 80% purity, 0.063 mmol) and diisopropylethylamine (11 μl, 0.063 mmol) were added subsequently. The mixture continued to be stirred at room temperature overnight, quenched with saturated ammonium chloride and diluted with dichloromethane, separated and washed with brine, dried over sodium sulfate, filtered and concentrated. The residue was purified by preparative reverse phase HPLC (C18 column, acetonitrile/water). The fractions containing product were combined, extracted with dichloromethane and concentrated to give compound 3g as yellowish solid (15.2 mg, y=38%). MS (m/z), found 984.3 (M+Na)+, 1014.2 (M+3H2O—H)−. See FIG. 3 .
A stirred solution of compound 4a (111 mg, 0.108 mmol) in absolute ethanol (720 μL) and anhydrous dichloromethane (360 μL) was cooled to 0° C. in an ice bath. Sodium borohydride (0.817 mg, 0.022 mmol) in 50 uL absolute ethanol was added at 0° C. The reaction stirred at ambient temperature for two hours. The mixture was cooled to 0° C. in an ice bath, quenched with saturated ammonium chloride and extracted with dichloromethane. The organic extracts were washed with brine, dried over anhydrous sodium sulfate and filtered through Celite. The filtrate was concentrated under reduced pressure and the crude material was purified by RP-HPLC (C18 DI water/acetonitrile) to yield compound 4b (43 mg, 38%). 1H NMR (400 Hz, CDCl3): δ 8.26 (d, 1H, J=8.0 Hz), 8.18 (d, 1H, J=8.0 Hz), 7.77 (d, 1H, J=4.4 Hz), 7.51 (s, 1H), 7.41 (s, 2H), 7.17 (m, 6H), 7.03 (t, 1H, J=7.2 Hz), 6.96 (t, 1H, J=7.2 Hz), 6.76 (s, 1H), 6.04 (s, 1H), 5.13 (m, 4H), 4.38 (m, 2H), 3.90 (s, 3H), 3.81 (s, 3H), 3.79 (m, 2H), 3.63 (m, 1H), 3.51 (m, 8H), 3.43 (m, 6H), 3.25 (s, 3H), 2.73 (dd, 1H, J=3.6, 16.4 Hz), 2.22 (s, 3H), 2.04 (m, 2H), 1.81 (m, 2H), 1.18 (s, 6H); MS (m/z) found, 1051.9 (M+Na), 1069.9 (M+Na+H2O). See FIG. 4 .
To a stirred solution of compound 4b (40 mg, 0.039 mmol) in methanol (4.45 mL) and acetonitrile (2.225 mL) was added TCEP.HCl (39.0 mg, 0.136 mmol) in sodium phosphate buffer (0.89 mL, pH 6.5). The mixture stirred at ambient temperature for 18 hours. The mixture was diluted with dichloromethane and washed with water and brine. The organic extracts were dried over anhydrous sodium sulfate, filtered and concentrated. Purification by RP-hPLC (C18, DI water/acetonitrile) and extraction with dichloromethane yielded compound 4c (26.5 mg, 64%); MS (m/z) found, 1006.0 (M+Na). See FIG. 4 .
To a stirred solution of compound 4c (24 mg, 0.024 mmol) in anhydrous dichloromethane (800 μL) was added PBA (11.18 mg, 0.049 mmol) and diisopropylethylamine (20.18 μL, 0.116 mmol). After stirring for 18 hours at ambient temperature the reaction was diluted with dichloromethane and quenched with saturated ammonium chloride. The layers were separated and the organic was washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. Purification by PTLC (5% methanol/dichloromethane) yielded compound 4d (17 mg, 63%); MS (m/z) found, 1123.9 (M+Na) 1139.9 (M+K); 1099.8 (M−H) 117.9 (M−H+H2O). See FIG. 4 .
To a mixture of compound 4d (15 mg, 0.014 mmol) and N-hydroxy succinimide (4.70 mg, 0.041 mmol) in anhydrous dichloromethane (1.0 mL) was added EDC.HCl (7.83 mg, 0.041 mmol). After stirring 18 hours at ambient temperature the reaction was diluted with dichloromethane and quenched with saturated ammonium chloride. The mixture was washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crude material was purified by RP-HPLC (C18, DI water/acetonitrile). Fractions containing product were pooled and extracted with dichloromethane, dried over anhydrous magnesium sulfate, filtered and concentrated to give compound 4e (13 mg, 80%); MS (m/z) found, 1220.8 (M+Na) 1238.8 (M+Na+H2O), 1254.8 (M+K+H2O). See FIG. 4 .
A mixture of chelidamic acid hydrate (3.0 g, 15.56 mmol) and sulfuric acid (0.6 mL, 11.26 mmol) in absolute ethanol (40 mL) was refluxed for 20 hours. The reaction was cooled to ambient temperature, neutralized with aqueous sodium carbonate, and then acidified with concentrated HCl. Water was added and the mixture was extracted with dichloromethane. The extracts were dried with anhydrous magnesium sulfate, filtered and concentrated. The crude material was purified by silica gel chromatography (5% methanol/dichloromethane) to yield diethyl 4-hydroxypyridine-2,6-dicarboxylate (5a) (2.5 g, 68%) as a white solid. See FIG. 5 .
A solution of 4-methyl-4-(methyldisulfanyl)pentan-1-ol (5b) (2.0 g, 11.09 mmol) in anhydrous dichloromethane (55.5 mL) was cooled to 0° C. in an ice bath. Triethylamine (5.41 mL, 38.8 mmol) and toluene sulfonyl chloride (3.17 g, 16.64 mmol) were added at 0° C. The reaction stirred for three hours at ambient temperature. The mixture was extracted with ethyl acetate and washed with brine. The organic extracts were dried with anhydrous sodium sulfate, filtered and concentrated. Purification by silica gel chromatography (5% ethyl actetate/hexanes) resulted in 4-methyl-4-(methyldisulfanyl)pentyl 4-methylbenzenesulfonate (5c) (1.5 g, 40%). 5b: 1H NMR (400 Hz, CDCl3): δ 3.42 (m, 2H), 2.19 (s, 3H), 1.77 (bs, 1H), 1.43 (m, 4H), 1.09 (s, 6H). 5c: 1H NMR (400 Hz, CDCl3): δ 7.66 (d, 2H, J=7.6 Hz), 7.22 (d, 2H, J=8.0 Hz), 3.90 (t, 2H, J=6.4 Hz), 2.32 (s, 3H), 2.23 (s, 3H), 1.60 (m, 2H), 1.44 (m, 2H), 1.11 (s, 6H). See FIG. 5 .
To a stirred solution of 4-methyl-4-(methyldisulfanyl)pentyl 4-methylbenzenesulfonate (5c) (0.48 g, 1.435 mmol) and diethyl 4-hydroxypyridine-2,6-dicarboxylate (5a)(0.343 g, 1.435 mmol) in anhydrous dimethylformamide (6.5 mL) was added Potassium carbonate (0.297 g, 2.152 mmol). The reaction was stirred at 90° C. for 18 hours. Then allowed to cool to ambient temperature and quenched with saturated ammonium chloride. The mixture was extracted three times with ethyl acetate. The extracts were dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure. Purification by silica gel chromatography (30% hexanes/ethyl acetate) yielded diethyl 4-(4-methyl-4-(methyldisulfanyl)pentyloxy)pyridine-2,6-dicarboxylate (5d)(300 mg, 52%); 1H NMR (400 Hz, CDCl3): δ 7.70 (s, 2H), 4.40 (q, 4H, J=7.2, 14.4 Hz), 4.07 (t, 2H, J=6. Hz), 2.35 (s, 3H), 1.86 (m, 2H), 1.70 (m, 2H), 1.38 (t, 6H, J=7.2 Hz), 1.27 (s, 6H); MS (m/z), found 424.1 (M+Na), 440.1 (M+K). See FIG. 5 .
To a stirred solution of diethyl 4-(4-methyl-4-(methyldisulfanyl)pentyloxy)pyridine-2,6-dicarboxylate (5d) (270 mg, 0.672 mmol) in absolute ethanol (7.0 mL) was added calcium chloride (224 mg, 2.017 mmol) and sodium borohydride (76 mg, 2.017 mmol). The reaction was allowed to stir at ambient temperature for 90 minutes after which it was quenched with water and concentrated in vacuo to remove the ethanol. The mixture was then extracted twice with dichloromethane. The organic extracts were combined, washed with water, dried with anhydrous magnesium sulfate and filtered through celite. The filtrate was concentrated under reduced pressure and the crude material was purified by silica gel chromatography eluting with 10% methanol/dichloromethane to yield (4-(4-methyl-4-(methyldisulfanyl)pentyloxy)pyridine-2,6-diyl)dimethanol (5e)(75 mg, 35%); 1H NMR (400 Hz, CDCl3): δ 6.63 (s, 2H), 4.60 (s, 4H), 3.95 (t, 2H, J=6.2 Hz), 3.54 (bs, 2H), 2.35 (s, 3H), 1.82 (m, 2H), 1.66 (m, 2H), 1.26 (s, 6H); MS (m/z), found 340.1 (M+Na). See FIG. 5 .
A stirred solution of (4-(4-methyl-4-(methyldisulfanyl)pentyloxy)pyridine-2,6-diyl)dimethanol (5e) (51 mg, 0.161 mmol) in anhydrous dichloromethane (1.6 mL) was cooled to −5° C. in an acetone/ice bath. Triethylamine (0.112 mL, 0.803 mmol) and methanesulfonyl chloride (0.031 mL, 0.402 mmol) were added. The mixture was stirred for 60 minutes at −5° C. The reaction was quenched with ice water and extracted with cold ethyl acetate. The organic extracts were washed with ice water, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give the dimesylate. To a stirred mixture of the dimesylate intermediate (179 mg, 0.378 mmol) and IBD monomer (256 mg, 0.869 mmol) in anhydrous dimethylformamide (3.8 mL) was added potassium carbonate (261 mg, 1.890 mmol) and potassium iodide (31.4 mg, 0.189 mmol). The reaction was allowed to stir at ambient temperature for 18 hours. The mixture was quenched with water and extracted three times with dichloromethane. The organic extracts were dried with sodium sulfate, filtered and concentrated. The crude material was redissolved in acetonitrile and purified by RP-HPLC (C18, deionized water/acetonitrile). Fractions containing product were combined and extracted with dichloromethane, dried with anhydrous magnesium sulfate, filtered and concentrated under reduced pressure to yield compound 5f (65 mg, 20%); 1H NMR (400 Hz, CDCl3): δ 8.20 (d, 2H, J=8.0 Hz), 7.78 (m, 2H), 7.53 (s, 2H), 7.20 (m, 4H), 7.04 (t, 2H, J=7.4 Hz), 6.91 (m, 2H), 6.80 (s, 2H), 5.22 (s, 4H), 4.40 (m, 2H), 3.94 (s, 6H), 3.93 (m, 2H), 3.63 (m, 2H), 3.42 (dd, 2H, J=Hz), 2.32 (s, 3H), 1.80 (m, 2H), 1.64 (m, 2H), 1.24 (s, 6H); MS (m/z), found 892.3 (M+Na) 910.3 (M+Na+H2O) 928.3 (M+Na+2H2O). See FIG. 5 .
A solution of compound 5f (74 mg, 0.085 mmol) in absolute ethanol (600 μL) and anhydrous dichloromethane (300 μL) was cooled 0° C. in an ice bath. Sodium borohydride (0.644 mg, 0.017 mmol) in 50 μL absolute ethanol was added at 0° C. The mixture was allowed to stir at ambient temperature for two hours and was then cooled to 0° C. The reaction was quenched with saturated ammonium chloride and extracted with dichloromethane. The organic extracts were washed with brine, dried over anhydrous sodium sulfate, filtered through Celite and concentrated under reduced pressure. The crude material was redissolved in dimethylformamide and purified by RP-HPLC (C18 deionized water/acetonitrile). Fractions containing compounds 5g and 5h were combined separately and extracted with dichloromethane, dried with anhydrous magnesium sulfate, filtered and concentrated to yield compound 5g (20 mg, 27%) and compound 5h. 5g: 1H NMR (400 Hz, CDCl3): δ 8.25 (m, 1H), 8.18 (m, 1H), 7.77 (m, 1H), 7.51 (ss, 1H), 7.40 (ss, 1H), 7.18 (m, 4H), 7.08 (m, 1H), 7.03 (m, 1H), 6.92 (m, 2H), 6.86 (ss, 1H) 5.98/6.06 (ss, 1H), 5.24 (m, 4H), 4.40 (m, 1H), 4.30 (m, 1H), 3.94 (s, 3H), 3.92 (m, 2H), 3.84 (s, 3H), 3.62 (m, 1H), 3.37 (m, 4H), 2.65 (m or dd, 1H), 2.32 (ss, 3H), 1.77 (m, 2H), 1.64 (m, 2H), 1.24 (s, 6H). 5h: 1H NMR (400 Hz, CDCl3): δ 8.24 (d, 2H, J=8.0 Hz), 7.39 (s, 2H), 7.14 (m, 4H), 6.97 (m, 2H), 6.93 (m, 2H), 6.15 (ss, 2H), 5.25 (s, 4H), 4.37 (m or t, 2H, J=9.8 Hz), 4.2 (bs, 2H), 3.94 (m, 2H), 3.83 (s, 6H), 3.40 (m, 6H), 2.72 (dd, 2H, J=Hz), 2.32 (s, 3H), 1.79 (m, 2H), 1.64 (m, 2H), 1.24 (s, 6H). See FIG. 5 .
Compound 5i:
To a stirred solution of compound 5g (20 mg, 0.023 mmol) in methanol (5.25 mL) and acetonitrile (1.750 mL) was added TCEP.HCl (19.72 mg, 0.069 mmol) in sodium phosphate buffer (0.7 mL, pH 6.5). The mixture was stirred for 3 hours at ambient temperature and then diluted with dichloromethane and water. The layers were separated and the organic was washed with brine. The crude product was purified by RP-HPLC (C18, deionized water/acetonitrile). Fractions containing product were combined, extracted with dichloromethane and evaporated to yield compound 5i (7 mg, 37%). MS (m/z), found 848.3 (M+Na) 866.3 (M+Na+H2O) 880.3 (M+Na+MeOH). See FIG. 5 .
Compound 5j:
To a stirred solution of compound 5i (7 mg, 8.47 μmol) and 2,5-dioxopyrrolidin-1-yl 4-(pyridin-2-yldisulfanyl)butanoate (8.64 mg, 0.021 mmol) in anhydrous dichloromethane (113 μL) was added diisopropylethylamine (3.69 μL, 0.021 mmol). After stirring for 18 hours at ambient temperature the reaction was quenched with saturated ammonium chloride solution and extracted with dichloromethane. The organic extracts were washed with brine, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crude material was purified by preparative RP-HPLC (C18, deionized water/acetonitrile). Fractions containing product were extracted with dichloromethane, filtered and evaporated to yield compound 5j (3 mg, 34%). MS (m/z), found 1063.3 (M+Na) 1081.3 (M+Na+H2O). See FIG. 5 .
Preparation of Antibody-SPDB-Drug Conjugate:
Preparation of Humanized Ab-SPDB-2a Conjugate:
Humanized Ab at 8 mg/mL was derivatized with 4-6 molar equivalents of SPDB hetrobifunctional linker for 1.5 h at 25° C. in 95% PBS, PH 7.4, containing 5% DMA (v/v), and then purified over a G25 desalting column into citrate buffer (35 mM citrate buffer, pH 5.5, containing 2 mM EDTA, 150 mM NaCl) to remove unreacted linker. The LAR (Linker Antibody Ratio) were measured using UV absorbance at 280 and 343 nm without and with 50 mM dithiothreitol addition (to measure total antibody and dithiothreitol-released SPy) and were determined to be 2.7-4.1 LAR. The SPDB-modified antibody at 2 mg/mL was reacted with 2 molar equivalents of compound 2a (HCl salt) per linked SPDB for 20 h at ambient temperature in 85% citrate buffer, 15% DMA (v/v) and then purified over a G25 desalting column into PBS, pH 7.4 to remove unconjugated drug compound. The DAR of the final humanized Ab-SPDB-2a conjugate was measured by UV spectrophotometry at 280 and 350 nm and calculated to be ˜1.7-2.1 DAR. The percentage of monomer and linked drug compound on the monomer in the conjugate was determined by HPLC using an SEC (size exclusion chromatography) column. See FIG. 23 .
In Vitro Potency of Free Drugs and Conjugates:
General Procedure Used: Samples of unconjugated free drug compounds or drug conjugates were added to 96-well flat bottomed tissue culture plates and titrated using serial dilutions to cover the desired molar range. Antigen positive (Ag+) or Antigen negative (Ag−) cells were added to the wells in specific cell densities in such a way that there were triplicate samples for each drug concentration for each corresponding cell line. The plates were then incubated at 37° C. in an atmosphere of 5% CO2 for 4-5 days depending on the cell line. COLO 205 (1,000 cells/well), Namalwa (3,000 cells/well), HEL 92.1.7 (3,000 cells/well)—4 days; RH30 (1,000 cells/well), HL60/QC (5,000 cells/well), Ramos (10,000 cells/well), KB (2,000 cells/well), BJAB (2,000 cells/well), NB4 (3,000 cells/well)—5 days, RPMI 8226 (8,000 cells/well)—6 days.
At the end of the incubation period cytotoxic potencies were then assessed using a WST-8 based cell viability assay and surviving cells were measured by developing with WST-8 (2-7 hours). The absorbance in each well was measured and the surviving fraction of cells at each concentration was plotted to reveal the cytotoxicity and/or antigen specificity (of the conjugates).
Using the general procedure described above, the cytotoxicity of the unconjugated free drug compounds was measured against seven cell lines: KB, a HeLa cell contaminant, HL60/QC, an acute myeloid leukemia cell line, Namalwa, a Burkitt lymphoma cell line, NB4, an acute promyelocytic leukemia cell line, HEL92.1.7, an erythroleukemia cell line, RPMI8226, a multiple myeloma cell line and BJAB, a B-cell leukemia cell line. The results, shown in FIG. 24 and Table 10 demonstrate the high potency of these compounds across a wide range of cell types. The potency and specificity of the antibody-drug conjugates were measured against antigen-expressing cells, with and without the additions of an excess amount of blocking unconjugated antibody to show specificity of the killing effect. The MY9-6-drug conjugate was extremely potent towards three different antigen-expressing cells: HL60/ATCC, HL60/QC and NB-4, despite the very low antigen expression in NB4 cells. The specific potency could be blocked by addition of excess unconjugated antibody, demonstrating that the cell killing effect is antigen-specific. Similarly, the huFOLR1-drug conjugate was effective in killing antigen-expressing KB cells in a specific manner. Results are illustrated in FIGS. 25 and 26 .
| TABLE 10 |
| Potency of free drugs against various cell lines. The IC50 values listed in the |
| table are in the unit of nM. |
| Namalwa | KB | HL60/QC | NB4 | HEL92.1.7 | | BJAB | ||
| 1c | 0.056 | 0.16 | 0.023 | ||||
| 1d | 0.069 | 0.18 | 0.032 | ||||
| 1e | 2.4 | >3.0 | 0.67 | ||||
| 27d | 0.23 | 0.05 | 0.039 | 0.14 | 0.07 | 0.04 | |
| 27e | 0.39 | 0.09 | 0.13 | 0.2 | 0.24 | 0.12 | |
| 27f | 4.4 | 1.7 | 1.1 | 1.8 | >3.0 | 1 | |
| 29a | 0.002 | 0.004 | 0.001 | 0.0023 | 0.0031 | 0.011 | 0.001 |
| 29b | 0.003 | 0.007 | 0.006 | 0.007 | 0.007 | 0.005 | 0.003 |
| 29c | 0.013 | 0.057 | 0.03 | 0.023 | 0.027 | 0.16 | 0.015 |
Similar results have also been obtained using different cell lines and different conjugates of the invention, including: huMY9-6-SPDB-1f against HL60/QC (Ag+) cells, HL60/ATCC (Ag+) cells, and NB-4 (Ag+) cells (FIG. 25 ); huFOLR1-SPDB-1f against KB (Ag+) cells (FIG. 26 ); huMY9-6-SPDB-1f against antigen positive HL60/QC cells, HL60/ATCC cells, NB-4 cells, and HEL 92.1.7 cells (FIG. 29 ); huMy9-6-SPDB-1f, huMy9-6-sulfoSPDB-1f, and huMy9-6-BMPS-1f against HL60/QC (Ag+) cells (FIG. 34 ); chB38.1-SPDB-1f and chB38.1-sulfoSPDB-1f against COLO205 (Ag+) cells (FIG. 35 ); huMy9-6-SPDB-1f, huMy9-6-sulfoSPDB-1f, and huMy9-6-BMPS-1f against OCI-AML3 (Ag+) cells (FIG. 44 ). Also see FIG. 49 for the potency of the various conjugates against various cell lines, expressed as IC50 values (nM). Note that in FIGS. 25, 29, 34, 35, and 44 , conjugagtes were prepared in the presence of sodium bisulfite.
To compare in vitro potency measurements for the subject conjugates prepared with and without imine reactive reagent, such as sodium bisulfite, huMy9-6-BMPS-1f, huMy9-6-sulfo-SPDB-1f, and huMy9-6-Drug 2 were prepared with and without sodium bisulfite using the in situ sulfonation method (wherein the respective compounds of the invention was first mixed with sodium bisulfite and a bifunctional crosslinker bearing a reactive group, then the reaction mixture, without further purification, was reacted with the huMy9-6 monoclonal antibody as the cell-binding agent). IC50s for the conjugates on HL60-QC cells are shown below. The data indicates that the inclusion of imine reactive group (such as sodium bisulfite) in the conjugate preparation step does not negatively impact the in vitro potency of the subject conjugates.
| NaHSO3 | IC50 | IC50 (pM) huMy9-6 | |
| Conjugate | treatment | (pM) | blocking |
| huMy9-BMPS-1f | − | 2 | 130 |
| + | 1.5 | 55 | |
| huMy9-6-sulfo-SPDB-1f | − | 5.6 | 1200 |
| + | 7.1 | 610 | |
| huMy9-6- |
− | 16 | >3000 |
| + | 6.8 | >3000 | |
It is apparent that pre-treatment of the drug compounds with sodium bisulfite (5 molar equivalents, 22 h, 4° C., 90:10 DMA:pH 5.5 water) prior to conjugation with huMy9-6 had no significant effect on the antigen dependent or antigen independent (antigen blocking with 1 μM unconjugated huMy9-6) in vitro potency of the conjugates.
Binding of Antibody-Drug Conjugate is Similar to that of Unmodified Antibody:
The binding of huMY9-6-drug conjugate was compared with that of the unmodified huMY9-6 antibody against antigen-expressing HL60/QC cells using flow cytometry. Briefly, the antigen-positive cells were incubated with conjugates or unmodified antibodies at 4° C., then with a secondary antibody-FITC conjugate at 4° C., fixed with formaldehyde (1% in PBS) and analyzed by flow cytometry. No significant difference was observed between the binding of the conjugate versus that of the unmodified antibody. An example is shown in FIG. 27 , where a huMY9-6-drug conjugate bound to antigen-positive cells with a high affinity similar to that of the unmodified antibody.
In Vivo Efficacy of huMY9-6-SPDB-1f Conjugate in HL60/QC Tumor Bearing Nude Mice:
In this study, the anti-tumor activity of huMY9-6-SPDB-1f was investigated in female nude mice bearing HL60/QC tumors, a human acute myeloid leukemia model. HL60/QC tumor cells, 2×106 cells/mouse were subcutaneously inoculated at a volume of 0.1 mL/mouse in the area over the right shoulder of female athymic nude mice, 5 weeks of age. Eight days after tumor cell inoculation mice were randomized into groups (n=6 per group) by tumor volume. Treatment was initiated the day of randomization, and groups included a control group dosed with PBS (200 μL/injection), or a single treatment at various doses (5 to 100 μg/kg) of huMY9-6-SPDB-1f (50 μg/kg 1f dose corresponded to 2.5 mg/kg antibody dose). All treatments were well tolerated with the mean body weight losses comparable to loss seen in PBS control mice. Mean tumor volume vs time is shown (FIGS. 28 and 36 ) with the data demonstrating a dose-dependent anti-tumor activity of the huMY9-6-SPDB-1f conjugate. The minimum effective dose was estimated to be 20 μg/kg, which is about 35-fold lower that the maximum tolerated dose.
The tolerability of huFOLR-1 conjugates was investigated in female CD-1 mice. Animals were observed for seven days prior to study initiation and found to be free of disease or illness. The mice were administered a single i.v. injection of the conjugate and the animals were monitored daily for body weight loss, morbidity or mortality. Table 9 shows that for huFOLR1-drug1 the conjugate was tolerated at only the lowest dose tested of 50 μg/kg. In contrast, both mono-imine conjugates huFOLR1-Drug 2 and huFOLR1-SPDB-1f were found to be better tolerated with a maximum tolerated dose of <198 μg/kg and >560 μg/kg respectively.
| TABLE 9 |
| Tolerability comparison data for (A) huFOLR1-drug1, (B) huFOLR1- |
| and (C) huFOLR1-SPDB-1f conjugates. |
| Dose | % |
| (μg/kg) | Survival |
| A) |
|
|
| 50 | 100 |
| 100 | 0 |
| 200 | 0 |
| 300 | 0 |
| 400 | 0 |
| B) |
|
|
| 66 | 100 |
| 132 | 100 |
| 198 | 50 |
| 264 | 25 |
| C) |
|
|
| 120 | 100 |
| 160 | 100 |
| 200 | 100 |
| 320 | 100 |
| 560 | 100 |
Compound 10:
To a stirred solution of 1f (18 mg, 0.019 mmol) and N—(β-maleimidopropyloxy)succinimide (BMPS) ester (9.2 mg, 0.034 mmol) in anhydrous dichloromethane (0.3 mL) was added anhydrous diisopropylethylamine (5 μL, 0.029 mmol). The mixture was stirred at room temperature for 27 hours, quenched with saturated ammonium chloride and diluted with dichloromethane. The organic layer was washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by preparative reverse phase HPLC (C18 column, CH3CN/H2O). The fractions containing product were combined, extracted with dichloromethane and evaporated to give compound 10 as a white solid (7.6 mg, y=33%). MS (m/z): found 1208.3 (M+H)+. See FIG. 13 .
Compound 12:
To a stirred solution of 1f (16.5 mg, 0.018 mmol) and sulfo-SPDB (14.2 mg, 0.036 mmol) in anhydrous dichloromethane (0.3 mL) was added anhydrous diisopropylethylamine (9 μL, 0.054 mmol). The mixture was stirred at room temperature overnight and concentrated under reduced pressure. The residue was purified by preparative reverse phase HPLC (C18 column, CH3CN/H2O). The fractions containing product were combined, extracted with dichloromethane and evaporated to give 6.6 mg of compound 12 as yellowish foam. The aqueous layer was lyophilized to give another 0.5 mg of compound 12 as white solid. MS (m/z): found 1235.0 (M−H)−. See FIG. 15 .
Preparation of Humanized Antibody-sulfoSPDB-1f Conjugate
A reaction containing 2.5 mg/mL huMy9-6 antibody and 10 molar equivalents of 10 (pretreated with 5-fold excess of sodium bisulfite in 90:10 DMA:water) in 50 mM HEPES (4-(2-hydroxyethyl)-1-piperazine ethanesulfonic acid) pH 8.5 buffer and 15% v/v DMA (N,N-dimethylacetamide) cosolvent was allowed to conjugate for 6 hours at 25° C. Post-reaction, the conjugate was purified and buffer exchanged into 250 mM Glycine, 10 mM Histidine, 1% sucrose, 0.01% Tween, 50 μM sodium bisulfite formulation buffer, using NAP desalting columns (Illustra Sephadex G-25 DNA Grade, GE Healthcare). Dialysis was performed in the same buffer for 4 hours at room temperature utilizing Slide-a-Lyzer dialysis cassettes (ThermoScientific 20,000 MWCO). The purified conjugate was found to have a DAR of 2.4 (by UV-Vis using molar extinction coefficients ε330 nm=15,484 cm−1 M− and ε280 nm=30, 115 cm−M−1 for 1f, and ε280 nm=146,000 cm−1M−1 for My9-6 antibody), 96.7% monomer (by size exclusion chromatography), <1% unconjugated free drug compound (by acetone extraction/reverse-phase HPLC) and a final protein concentration of 1.4 mg/mL.
In vitro potency of antibody-sulfoSPDB-1f conjugates were measured according to general procedure described in Example 7 and the data are shown in FIGS. 34 and 35 . The antibody-sulfoSPDB-1f conjugates have comparable or higher potency than the antibody-SPDB-1f conjugates.
Use of covalent imine reactants, such as sodium bisulfite, improves Ab-compound conjugate specifications (e.g., % monomer and drug load). In one experiment, adduct formation was carried out with 5 molar equivalents of imine reactant over NHS-BMPS-1f in 90% DMSO/10% PBS pH 7.4 for 4 h at 25° C. The reaction mixture was then added to huMy9-6 antibody (4 molar equivalents IGN, 2 mg/ml, 10% v/v DMSO, 50 mM HEPES buffer, pH 8.5, 5 h, 25° C.). Conjugates made using sodium hydrosulfite, sodium bisulfite, or sodium metabisulfite had similar IGN/Ab ratios and % monomer, while conjugates made with no additive treatment led to very low drug incorporation. See table below.
| IGN/Ab | | % | 1f on | ||
| Reactant | (UV) | (SEC) | monomer | ||
| Sodium | 2.6 | 88 | 82 | |
| Hydrosulfite | ||||
| Sodium Bisulfite | 2.6 | 88 | 83 | |
| Sodium | 2.7 | 88 | 82 | |
| Metabisulfite | ||||
| No additive | 0.1 | 98 | 94 | |
Preparation of Humanized Antibody-BMPS-1f Conjugate
A reaction containing 2.0 mg/mL huMy9-6 antibody and 5 molar equivalents of 12 (pretreated with 5-fold excess of sodium bisulfite in 90:10 DMA:water) in 50 mM HEPES (4-(2-hydroxyethyl)-1-piperazine ethanesulfonic acid) pH 8.5 buffer and 15% v/v DMA (N,N-dimethylacetamide) cosolvent was allowed to react for 6 hours at 25° C. Post-reaction, the conjugate was purified and buffer exchanged into 250 mM Glycine, 10 mM Histidine, 1% sucrose, 0.01% Tween, 50 μM sodium bisulfite formulation buffer, using NAP desalting columns (Illustra Sephadex G-25 DNA Grade, GE Healthcare). Dialysis was performed in the same buffer for 4 hours at room temperature utilizing Slide-a-Lyzer dialysis cassettes (ThermoScientific 20,000 MWCO). The purified conjugate was found to have a DAR of 2.8 (by UV-Vis using molar extinction coefficients ε330 nm=15,484 cm−1M−1 and F280 nm=30, 115 cm−1M−1 for 1f, and ε280 nm=146,000 cm−1M−1 for My9-6 antibody), 91.7% monomer (by size exclusion chromatography), <1% unconjugated free drug compound (by acetone extraction/reverse-phase HPLC) and a final protein concentration of 1.2 mg/mL.
In vitro potency of antibody-BMPS-1f conjugates were measured according to general procedure described in Example 7 and the data are shown in FIGS. 34 and 35 . The antibody-BMPS-1f conjugates have comparable potency to the antibody-SPDB-1f conjugates.
In Vivo Efficacy of Hu FOLR1-SPDB-1f Conjugate in KB Tumor Bearing Nude Mice:
In this study, the anti-tumor activity of hu FOLR1-SPDB-1f was investigated in female nude mice bearing KB tumors, a human cervical carcinoma model. KB, 1×107 cells/mouse were subcutaneously inoculated at a volume of 0.1 mL/mouse in the area over the right shoulder of female athymic nude mice, 6 weeks of age. Six days after tumor cell inoculation mice were randomized into groups (n=6 per group) by tumor volume. Treatment was initiated the day after randomization, and groups included a control group dosed with PBS (200 μL/injection), or a single treatment at various doses (20 to 200 μg/kg) of hu FOLR1-SPDB-1f (50 mg/kg linked drug dose corresponded to 2.8 mg/kg antibody dose). All treatments were well tolerated with no body weight loss seen in any of the test groups. Mean tumor volume vs time is shown (FIG. 37 ) with the data demonstrating a dose-dependent anti-tumor activity of the hu FOLR1-SPDB-1f conjugate. The minimum effective dose was estimated to be <50 μg/kg, which is about 14-fold lower than the maximum tolerated dose.
Similar in vivo results have also been obtained using other conjugates of the invention against various other cancer models, including huMy9-6-sulfo-SPDB-1f in MOLM-13 tumor bearing mice (FIG. 50 ); huMy9-6-sulfo-SPDB-1f in NB4 tumor bearing mice (FIG. 51 ); huMy9-6-BMPS-1f in HL60/QC tumor bearing mice (FIG. 52 ); huMy9-6-BMPS-1f in MOLM-13 tumor bearing mice (FIG. 53 ); huMy9-6-Drug 2 in HL60/QC tumor bearing mice (FIG. 56 ); and huMy9-6-Drug 2 in MOLM-13 tumor bearing mice (FIG. 57 ). Note that in FIGS. 53, 54, 56, and 57 , conjugagtes were prepared in the presence of sodium bisulfite.
To compare in vivo efficacy of the subject conjugates prepared with or without an imine reactive group, huMy9-6-Drug 2 were formulated with or without 50 μM sodium bisulfite, and the conjugates were used to treat mice bearing HL60-QC tumor xenografts. The data below shows that conjugate formulated with or without 50 μM sodium bisulfite showed comparable T/C % at ˜20 μg/kg drug dose, indicating that the inclusion of sodium bisulfite in the conjugate preparation step does not negatively impact the in vivo potency of the subject conjugate.
| Minimum effective dose | ||
| NaHSO3 | (μg/kg drug) | T/C % |
| − | 18 | 20 |
| + | 19 | 16 |
(5-((2-mercapto-2-methylpropylthio)methyl)-1,3-phenylene)dimethanol: (5-(mercaptomethyl)-1,3-phenylene)dimethanol (0.163 g, 0.885 mmol) was dissolved in methanol (3 mL) in a small vial and a stir bar was added. To this solution was added triethylamine (0.016 mL, 0.118 mmol) followed by 2,2-dimethylthiirane (0.058 mL, 0.590 mmol) and the resulting mixture was capped and stirred overnight (16 hrs) at room temperature. The reaction was then concentrated, redissolved in dichloromethane, loaded onto a silica ptlc plate (1000 micron) and the plate was developed using 10% methanol in dichloromethane. The band corresponding to the product was scraped, filtered with neat ethyl acetate, and concentrated to give (5-((2-mercapto-2-methylpropylthio)methyl)-1,3-phenylene)dimethanol (0.095 g, 0.349 mmol, 59.1% yield). 1H NMR (400 Hz, CDCl3): δ 7.26 (s, 3H), 4.69 (s, 4H), 3.82 (s, 2H), 2.74 (s, 2H), 2.17 (s, 1H), 2.12 (br s, 2H), 1.43 (s, 6H); 13C NMR (400 Hz, CDCl3): δ 141.6, 138.9, 126.7, 124.3, 65.0, 49.0, 45.4, 38.4, 31.5; MS (m/z), expected: 272.4, found 295.0 (M+Na). See FIG. 30 .
(5-((2-methyl-2-(methyldisulfanyl)propylthio)methyl)-1,3-phenylene)dimethanol: (5-((2-mercapto-2-methylpropylthio)methyl)-1,3-phenylene)dimethanol (0.120 g, 0.440 mmol) was dissolved in ethanol (5 mL) and 1.0 M potassium phosphate buffer (pH 7) (5.00 mL) and cooled in an ice bath (a ppt formed but it was ignored). S-methyl methanesulfonothioate (0.083 mL, 0.881 mmol) was added and the mixture stirred overnight with gradual (over 30 minutes) warming to room temperature. The reaction was diluted with dichloromethane and the organic layer was removed, washed with water, dried over anhydrous sodium sulfate, and concentrated in vacuo. The residue was dissolved in dichloromethane and loaded onto a 500 micron ptlc plate and developed with 66% ethyl acetate in hexane. The band corresponding to the product was scraped, filtered using ethyl acetate, and concentrated to give (5-((2-methyl-2-(methyldisulfanyl)propylthio)methyl)-1,3-phenylene)dimethanol (0.091 g, 0.286 mmol, 64.9% yield). 1H NMR (400 Hz, CDCl3): δ 7.27 (s, 3H), 4.71 (s, 4H), 3.78 (s, 2H), 2.77 (s, 2H), 2.41 (s, 3H), 1.94 (br s, 2H), 1.38 (s, 6H); 13C NMR (400 Hz, CDCl3): δ 141.6, 139.0, 126.7, 124.2, 65.0, 51.8, 44.0, 38.2, 26.7, 25.3; MS (m/z), expected: 341.5, found 341.1 (M+Na). See FIG. 30 .
(5-((2-methyl-2-(methyldisulfanyl)propylthio)methyl)-1,3-phenylene)dimethanol (80 mg, 0.251 mmol) in anhydrous dichloromethane (1.75 mL) was cooled to −5° C. in a brine/ice bath. Triethylamine (105 μL, 0.753 mmol) was added followed by the addition of methanesulfonyl chloride (50.7 μL, 0.653 mmol) at −5° C. The reaction was stirred at −5° C. for one hour after which it was diluted with cold ethyl acetate and ice was added. The mixture was transferred to a separatory funnel and extracted with cold ethyl acetate. The organic extracts were washed with ice water and then dried with anhydrous magnesium and sodium sulfate, filtered and concentrated under reduced pressure. The resulting (5-((2-methyl-2-(methyldisulfanyl)propylthio)methyl)-1,3-phenylene)bis(methylene)dimethanesulfonate was used without further purification.
IBD monomer (177 mg, 0.602 mmol) in anhydrous N, N-dimethylformamide (1.75 mL) was added to (5-((2-methyl-2-(methyldisulfanyl)propylthio)methyl)-1,3-phenylene)bis(methylene) dimethanesulfonate (119 mg, 0.251 mmol) at ambient temperature. Potassium carbonate (173 mg, 1.253 mmol) was added and the reaction was allowed to stir at ambient temperature for 20 hours. The reaction mixture was quenched with water and extracted with dichloromethane. The extracts were washed with brine and then dried with anhydrous sodium sulfate, filtered and concentrated on high vacuum. The crude product was purified by flash silica gel chromatography (neat DCM→2% MeOH/DCM). Fractions containing product were combined, concentrated and purified by semi-prep RP-HPLC (C18, A=DI water B=ACN, 20 mL/min). Fractions containing desired product were combined, extracted with dichloromethane, dried with anhydrous magnesium sulfate, filtered and concentrated to yield the desired product (46 mg, 21%). 1H NMR (400 Hz, CDCl3): δ 8.19 (d, J=8.0 Hz, 2H), 7.77 (m, d, J=4.4 Hz, 2H), 7.50 (s, 2H), 7.34 (s, 1H), 7.31 (s, 2H), 7.19 (m, 4H), 7.03 (t, J=7.2, 7.6 Hz, 2H), 6.77 (s, 2H), 5.14 (m, 4H), 4.40 (m, 2H), 3.91 (s, 6H), 3.70 (m, 2H), 3.63 (m, 2H), 3.41 (m, 2H), 2.65 (s, 2H), 2.29 (s, 3H), 1.26 (s, 6H). MS (m/z), Calcd. 893.2 (M+Na)+; found 893.2 (M+Na)+, 911.2 (M+H2O+Na)+, 929.2 (M+2H2O+Na)+, 945.1 (M+2H2O+K)+. See FIG. 30 .
To a cooled solution (0° C.) of 27d (50 mg, 0.057 mmol) in anhydrous dichloromethane (225 μL) and ethanol (450 μL) was added sodium borohydride (0.651 mg, 0.017 mmol). The reaction was stirred for five minutes at 0° C. and then at ambient temperature for 2.5 hrs. The reaction mixture was cooled to 0° C., quenched with saturated ammonium chloride, and extracted with dichloromethane. The organic extracts were washed with brine, dried over anhydrous sodium sulfate, filtered through Celite and concentrated. The crude material was purified by semi-prep RP-HPLC (C18, A=DI water B=ACN, 20 mL/min). Fractions containing desired product were combined, extracted with dichloromethane, dried with anhydrous magnesium sulfate, filtered and concentrated to yield the mono reduced amine 27e (11 mg, 22%) MS (m/z), Calcd. 895.3 (M+Na)+ found 895.2 (M+Na)+, 913.2 (M+H2O+Na)+, 929.2 (M+H2O+K)+ and the di-reduced amine 27f (5 mg, 10%) MS (m/z), Calcd. 897.3 (M+Na)+, found 897.3 (M+Na)+. See FIG. 30 .
Compound 27g:
To a stirred solution of 27e (10 mg, 0.011 mmol) in methanol (733 μL) and acetonitrile (880 μL) was added tris(2-Carboxyethyl) phosphine hydrochloride (9.85 mg, 0.034 mmol) in buffer 6.5 (147 μL). The mixture stirred at 3 hours at ambient temperature. The reaction was diluted with dichloromethane. Water was added and the layers were separated. The organic layer was washed with brine, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give compound 27g (9 mg, 95%). MS (m/z), Calcd. 849.3 (M+Na)+; found 849.2 (M+Na)+, 867.2 (M+K)+. See FIG. 30 .
To a stirred solution of 27g (9 mg, 10.88 μmol) and 2,5-dioxopyrrolidin-1-yl 4-(pyridin-2-yldisulfanyl)butanoate (9.3 mg, 0.023 mmol) in anhydrous dichloromethane (0.4 mL) was added anhydrous diisopropylethylamine (9 μl, 0.054 mmol) and the reaction was stirred at room temperature overnight. The mixture was quenched with saturated ammonium chloride solution and extracted with dichloromethane. The extracts were washed with brine, dried with anhydrous sodium sulfate and concentrated under reduced pressure. The residue was purified by preparative reverse phase HPLC (C18 column, CH3CN/H2O). The fractions containing product were combined, extracted with dichloromethane and evaporated to give compound 27h (5 mg, 44%). MS (m/z), Calcd. 1064.3 (M+Na)+; found 1064.1 (M+Na)+, 1082.1 (M+H2O+Na)+, 1098.1 (M+H2O+K)+. See FIG. 30 .
(5-(methyl(2-methyl-2-(methyldisulfanyl)propyl)amino)-1,3-phenylene)dimethanol (52 mg, 0.172 mmol) was dissolved in anhydrous dichloromethane (1.7 mL) and cooled to −5 in an acetone/ice bath. First, triethylamine (0.120 mL, 0.862 mmol) was added followed by methanesulfonyl chloride (0.040 mL, 0.517 mmol). The mixture was stirred in the bath for 1 hour. The reaction was then diluted with cold ethyl acetate and washed with cold water three times and then dried over anhydrous magnesium sulfate. The dimesylate was filtered, concentrated in vacuo, and placed under high vacuum until completely dry. The product was used as is directly in the next step.
IBD monomer (115 mg, 0.39 mmol) in anhydrous N,N-dimethylformamide (1.5 mL) was added to (5-(methyl(2-methyl-2-(methyldisulfanyl)propyl)amino)-1,3-phenylene)bis(methylene) dimethanesulfonate (72 mg, 0.156 mmol) at ambient temperature. Potassium carbonate (108 mg, 0.780 mmol) was added and the reaction was allowed to stir at ambient temperature for 20 hours. Water (10 mL) was added directly to the mixture with stirring resulting in the formation of a white precipitate. The mixture was filtered and the solids were washed with additional portions of water. The solid was then dissolved in dichloromethane, extracted with water, the organic layer was then dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo to give compound 28b (104 mg, 78%) which was used in the next step without further treatment. MS (m/z), found 912.1 (M+2H2O+Na). See FIG. 31 .
(5-((2-(2-(2-methoxyethoxy)ethoxy)ethyl)(2-methyl-2-(methyldisulfanyl)propyl)amino)-1,3-phenylene)bis(methylene) dimethanesulfonate (0.566 g, 0.960 mmol) was dissolved in acetone (30 mL) and a solution of sodium iodide (0.544 g, 3.63 mmol) dissolved in acetone (2 mL) was added with vigorous stirring. The reaction was monitored by tlc (50% ethyl acetate in hexane) and after 2 hours the reaction was filtered, concentrated in vacuo and dichloromethane was added to the residue. The solid salt left behind was filtered, the filtrate was concentrated and the resulting residue was purified on silica gel using a 3:5:2 mixture of ethyl acetate:hexane:dichloromethane to give 3,5-bis(iodomethyl)-N-(2-(2-(2-methoxyethoxy)ethoxy)ethyl)-N-(2-methyl-2-(methyldisulfanyl)propyl)aniline (0.505 g, 0.773 mmol, 74.5% yield) as a yellow oil. 1H NMR (400 Hz, CDCl3): δ 6.75 (s, 2H), 6.73 (s, 1H), 4.38 (s, 4H), 3.63 (m, 14H), 3.40 (s, 3H), 2.50 (s, 3H), 1.38 (s, 6H); 13C NMR (400 Hz, CDCl3): δ 148.7, 140.3, 117.3, 113.4, 71.9, 70.7, 70.6, 67.2, 59.8, 59.1, 53.5, 53.4, 51.8, 26.5, 25.6, 6.11; MS (m/z), Calcd 676.0 (M+Na)+; found 675.8 (M+Na)+. See FIG. 33 .
IBD Monomer (0.060 g, 0.204 mmol) was dissolved in acetone (4 ml) in a small vial, a stir bar was added, followed by 3,5-bis(iodomethyl)-N-(2-(2-(2-methoxyethoxy)ethoxy)ethyl)-N-(2-methyl-2-(methyldisulfanyl)propyl)aniline (0.167 g, 0.255 mmol) and potassium carbonate (0.070 g, 0.510 mmol). The vial was capped and stirred at room temperature overnight. The solids were filtered off and the filtrate was concentrated. The residue was dissolved in dichloromethane, extracted with water, and the organic layer was dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo to give 108 mg of crude material. The crude material was purified on silica gel using 30% ethyl acetate to remove the di-iodo starting material followed by 10% methanol in dichloromethane to give the desired product 30b (21 mg, 0.026 mmol, 13%). MS (m/z), expected: 819.1, found: 858.0 (M+K)+, 890.0 (M+CH3OH+K)+. See FIG. 33 .
The reduced monomer 3b (4.16 mg, 0.014 mmol) was dissolved in acetone (2 ml) in a small vial, a stir bar was added, followed by 30b (10 mg, 0.012 mmol) and potassium carbonate (4.21 mg, 0.030 mmol). The vial was capped and stirred at room temperature overnight. The reaction was concentrated to remove the acetone and then redissolved in dichloromethane, extracted with water, dried over anhydrous sodium sulfate, filtered, and concenrated in vacuo. The residue was purified by reverse phase C18 HPLC to get 1d (2.1 mg, 2.125 μmol, 17.42% yield). MS (m/z): found 1010.4 (M+Na)+, 1028.4 (M+H2O+Na)+. See FIG. 33 .
Compound 1:
To a stirred suspension of 1f (226 mg, 0.24 mmol) in IPA (20 mL) and deionized water (10 mL) was added sodium bisulphite (50 mg, 0.48 mmol). The mixture was stirred vigorously at rt for 2 hours. It was frozen with dry ice/aceton and lyophilized. The obtained white fluffy solid was dissolved in CH3CN/H2O and purified by reverse phase HPLC (C18 column, CH3CN/H2O). The fractions containing the desired product were combined and frozen with dry ice/acetone and lyophilized to give the desired compound 1 as white fluffy solid (179.6 mg, 5=71.6%). MS (m/z): found 1022.0 (M−H)−. See FIG. 38 .
To a stirred solution of 1c (60 mg, 0.061 mmol) in CH3CN (3 mL) was added freshly prepared TCEP solution (49 mg, 0.17 mmol of TCEP HCl salt was neutralized with saturated sodium bicarbonate to pH ˜6.5 then diluted with 0.5 mL of pH 6.5 phosphate buffer) at room temperature. MeOH (2.5 mL) was added and the mixture was stirred at room temperature for 3 hours. The reaction mixture was diluted with dichloromethane and deionized water, separated and the organic layer was washed with brine, dried over anhydrous Na2SO4 and filtered. The filtrate was stripped and high vacuumed to give 60 mg of 1h as light yellowish foam. MS (m/z): found 940.1 (M+H)+. It was dissolved in methanol (1.0 mL) and CH3CN (1.4 mL) followed by addition of iodoacetic acid (24 mg, 0.13 mmol), deionized water (0.1 mL) and potassium carbonate (27 mg, 0.19 mmol). The mixture was stirred at rt overnight (monitored by LCMS). It was quenched with saturated ammonium chloride to make the solution acidic then diluted with dichloromethane, separated and washed with brine, dried over anhydrous Na2SO4, filtered and stripped to give compound 9c (57.8 mg, y=91%) which was directly used for next step without purification. MS (m/z): found 998.1 (M+H)+. See FIG. 12A .
To a stirred solution of compound 9c (57.8 mg, 0.058 mmol) in anhydrous dichloromethane (0.2 mL) and absolute ethanol (0.6 mL) was added NaBH4 (2.5 mg, 0.066 mmol) at 0° C. The ice bath was removed and the mixture was stirred at room temperature for 3 hours and then quenched with saturated ammonium chloride, diluted with dichloromethane, separated and the organic layer was washed with brine, dried over anhydrous Na2SO4 and filtered through celite and stripped. The residue was purified by reverse phase HPLC (C18 column, CH3CN/H2O). The product fractions were extracted with dichloromethane and stripped to give compound 9a (13.0 mg, y=22%). MS (m/z): found 1000.0 (M+H)+, 1015.9 (M+H2O−H)−. See FIG. 12A .
To a solution of the free thiol 1f (45 mg, 0.048 mmol) and iodoacetic acid (18 mg, 0.096 mmol) in methanol (1.0 mL) and CH3CN (1.4 mL) was added deionized water (0.1 mL) and potassium carbonate (20 mg, 0.14 mmol). The mixture was stirred at rt overnight (monitored by LCMS). It was quenched with saturated ammonium chloride to make the solution acidic then diluted with dichloromethane, separated and washed with brine, dried over anhydrous Na2SO4, filtered and stripped. The residue was purified by preparative reverse phase HPLC (C18, CH3CN/H2O). The pure product fractions (based on MS) were extracted with dichloromethane, stripped to give the desired acid 9a (18 mg, y=38%). MS (m/z): found 1000.1 (M+H)+. See FIG. 12B .
To a stirred solution of compound 1c (178 mg, 0.18 mmol) in anhydrous dichloromethane (1.2 mL) and absolute ethanol or anhydrous methanol (0.1 mL) was added 5-ethyl-2-methylpyridine borane (PEMB, 0.017 mL, 0.11 mmol) dropwise. The mixture was stirred at rt for 1 hour and quenched with 88% formic acid. It was basified with saturated NaHCO3 and diluted with dichloromethane, separated and the organic layer was washed with brine, dried over anhydrous Na2SO4 and filtered through celite and stripped. The residue was dissolved in CH3CN/H2O/88% HCOOH (5:1:0.05) and purified by reverse phase HPLC (C18, CH3CN/H2O). The fractions that contained pure product were extracted with dichloromethane and stripped to give compound 1d (56 mg, y=31%). MS (m/z): found 988.1 (M+H)+. See FIG. 39 .
To a stirred solution of compound 1c (71 mg, 0.072 mmol) in anhydrous 1,2-dichloroethane (0.8 mL) was added sodium triacetoxyborohydride (14 mg, 0.65 mmol). The mixture was stirred at rt for 2 hours and quenched with saturated NaHCO3 and diluted with dichloromethane, separated and the organic layer was washed with brine, dried over anhydrous Na2SO4 and filtered through celite and stripped. The residue was dissolved in CH3CN/H2O/88% HCOOH (5:1:0.05) and purified by reverse phase HPLC (C18, CH3CN/H2O). The fractions that contained pure product were extracted with dichlormethane and stripped to give compound 1d (17 mg, y=24%). MS (m/z): found 988.1 (M+H)+. Unreacted starting material 1c was also recovered (24 mg, y=34%). See FIG. 40 .
To a solution of compound 1f (57.8 mg, 0.061 mmol) and methyl 4-bromobutyrate (22 mg, 0.12 mmol) in methanol (1.0 mL) and CH3CN (1.0 mL) was added deionized water (0.1 mL) and potassium carbonate (17 mg, 0.12 mmol). The mixture was stirred at rt overnight then quenched with saturated ammonium chloride and diluted with dichloromethane, separated and washed with brine, dried over anhydrous Na2SO4, filtered and stripped. The residue was purified by preparative reverse phase HPLC (C18, CH3CN/H2O) to give the desired product 31a (14 mg, y=22%) as yellowish foam. MS (m/z): found 1042.1 (M+H)+. See FIG. 41 .
To a solution of the methyl ester 31a (14 mg, 0.013 mmol) in anhydrous 1,2-dichloroethane (1.5 mL) was added trimethyltin hydroxide (36 mg, 0.2 mmol). The mixture was stirred overnight in a 80° C. oil bath until starting material was completely consumed. It was cooled to room temperature, diluted with dichloromethane, washed with brine/drops 5% HCl and brine, dried and filtered. The filtrate was stripped and purified with silica gel chromatography (dichloromethane/MeOH) to give acid 31b as yellowish solid (10.2 mg, y=74%). MS (m/z): found 1028.2 (M+H)+, 1044.1 (M+H2O—H)−. See FIG. 41 .
To a solution of acid 31b (10.2 mg, 0.0099 mmol) in anhydrous dichloromethane (0.5 mL) was added N-hydoxysuccinimide (3.4 mg, 0.03 mmol) and PL-DCC (26 mg, 0.04 mmol, 1.55 mmol/g). The mixture was stirred at room temperature overnight and filtered to remove the resin. The resin was washed with dichloromethane then ethyl acetate. The filtrate was stripped and the residue was purified by reverse phase HPLC (C18, CH3CN/H2O). The fractions containing product were combined and lyophilized to give NHS ester 31c as white solid (3.6 mg, y=32%). MS (m/z): found 1125.1 (M+H)+. See FIG. 41 .
To a stirred solution of the aniline 1a (339 mg, 1.1 mmol) in anhydrous tetrahydrofuran (4.0 mL) was added Boc anhydride (272 mg, 1.2 mmol). The mixture was continued to be stirred at room temperature for three days. The reaction mixture was concentrated under reduced pressure and the residue was purified by silica gel chromatography (CH2C12/MeOH) to give compound 32a (405 mg, y=90%) as colorless oil. 1H NMR (400 Hz, CDCl3): δ 7.00 (s, 2H), 6.97 (s, 1H), 4.38 (s, 4H), 4.12 (s, 2h), 3.64 (t, J=5.6 Hz, 2H), 3.48-3.44 (m, 8H), 3.40-3.38 (m, 2H), 3.21 (s, 3H), 1.31 (s, 9H); 13C NMR (400 Hz, CDCl3): δ 154.65, 142.3, 142.1, 124.1, 122.7, 80.2, 71.6, 70.3, 70.1, 69.9, 68.5, 63.9, 58.65, 49.4, 28.1. See FIG. 42 .
To a stirred solution of compound 32a (51 mg, 0.128 mmol) in anhydrous dichloromethane was added triethylamine (0.053 mL, 0.383 mmol) at −5˜−10° C. Methansulfonyl chloride (0.026 mL, 0.332 mmol) was then added slowly in 15 minutes with a syringe. The mixture was stirred at −5-10° C. for 1 hours (TLC, DCM/MeOH 10:1). The reaction was quenched with ice/water, diluted with cold AcOEt, separated and the organic layer was washed with cold water, dried over anhydrous Na2SO4/MgSO4, filtered and stripped. The residue was transferred into a small reaction flask with dichloromethane, stripped and high vacuumed. It was dissolved in anhydrous DMF (0.8 mL) followed by addition of IBD monomer (90 mg, 0.31 mmol) and potassium (53 mg, 0.38 mmol). The mixture was stirred at rt overnight. It was diluted with dichloromethane, washed with brine, dried over anhydrous sodium sulfate, filtered and stripped. The residue was purified by reverse phase HPLC (C18, CH3CN/H2O) to give compound 32b (56 mg, 46%) as yellowish solid. %). 1H NMR (400 Hz, CDCl3): δ 8.29 (d, J=8.0 Hz, 2H), 7.87 (d, J=4.8 Hz, 2H), 7.60 (s, 2H), 7.38-7.36 (m, 3H), 7.33-7.27 (m, 4H), 7.13 (t, J=7.6 Hz, 2H), 6.88 (s, 2H), 5.21 (dd, J1=20.0 Hz, J2=12.4 Hz, 4H), 4.49 (dt, J1=11.2 Hz, J2=4.0 Hz, 2H), 3.99 (s, 6H), 3.83 (t, J=6.0 Hz, 2H), 3.76-3.48 (m, 14H), 3.35 (s, 3H), 1.43 (s, 9H); MS (m/z): found 992.2 (M+H2O+Na)+, 1010.2 (M+2H2O+Na)+. See FIG. 42 .
To a stirred solution of compound 32b (56 mg, 0.059 mmol) in anhydrous dichloromethane (0.3 mL) and absolute ethanol (0.9 mL) was added NaBH4 (2.7 mg, 0.07 mmol) at 0° C. The ice bath was removed and the mixture was stirred at room temperature for 3 hours and then quenched with saturated ammonium chloride, diluted with dichloromethane, separated and the organic layer was washed with brine, dried over anhydrous Na2SO4 and filtered through celite and stripped. The residue was purified by reverse phase HPLC (C18 column, CH3CN/H2O). Recovered starting material 32b weighed 12 mg which was re-subjected to the reduction conditions and purified by reverse phase HPLC. All the fractions that contained pure product were extracted with dichloromethane and stripped to give compound 32c (20.7 mg, y=37%) as a light yellowish solid. MS (m/z): found 954.2 (M+H)+. See FIG. 42 .
The tolerability of huMy9-6 conjugates was investigated in female CD-1 mice. Animals were observed for seven days prior to study initiation and found to be free of disease or illness. The mice were administered a single i.v. injection of the conjugate and the animals were monitored daily for body weight loss, morbidity or mortality. Table 10 shows that the huMy9-6-SPDB-1c di-imine disulfide containing conjugate was tolerated at a dose of less than 300 μg/kg. In contrast, the mono-imine disulfide conjugates huMy9-6-SPDB-1f and huMy9-6-sulfo-SPDB-1f were found to be better tolerated with a maximum tolerated dose of >729 μg/kg and <750 μg/kg respectively.
| TABLE 10 |
| Tolerability comparison data for (A) huMy9-6-SPDB-1c, (B) huMy9-6-SPDB- |
| 1f, (C) huMy9-6-sulfo-SPDB-1f, and (D) huMy9-6-BMPS-1f conjugates. |
| Dose | % |
| (μg/kg) | Survival |
| A) |
|
|
| 100 | 100 |
| 300 | 50 |
| 500 | 0 |
| 700 | 0 |
| B) |
|
|
| 405 | 100 |
| 567 | 100 |
| 729 | 100 |
| C) |
|
|
| 450 | 100 |
| 600 | 100 |
| 750 | 88 |
| 900 | 50 |
| D) |
|
|
| 100 | 100 |
| 200 | 100 |
| 284 | 100 |
| 324 | 83 |
| 405 | 50 |
Compound 33b:
A mixture of (5-amino-1,3-phenylene)dimethanol (11.78 g, 77 mmol), 2-(2-(2-methoxyethoxy)ethoxy)ethyl 4-methylbenzenesulfonate (15.3 g, 48.1 mmol), and potassium carbonate (13.28 g, 96 mmol) in DMF (96 ml) was refluxed for 20 hours. The reaction was cooled to ambient temperature and diluted with dichloromethane. The mixture was filtered through celite and concentrated in vacuo. The resulting orange oil was dissolved in dichloromethane (240 ml) and t-butyldimethylsilyl chloride (18.09 g, 120 mmol) and imidazole (9.80 g, 144 mmol) were added. The reaction was stirred at ambient temperature for 20 hours upon which it was diluted with dichloromethane and filtered through celite. Purification by silica gel chromatography (EtOAc/Hex) yielded 3,5-bis(((tert-butyldimethylsilyl)oxy)methyl)-N-(2-(2-(2-methoxyethoxy)ethoxy)ethyl)aniline (13 g, 52%). 1H NMR (400 Hz, CDCl3): δ 6.52 (s, 1H), 6.40 (s, 2H), 4.56 (s, 4H), 3.60 (t, 2H, J=5.2 Hz), 3.56 (m, 6H), 3.46 (m, 2H), 3.29 (s, 3H), 3.20 (t, 2H, J=5.2 Hz), 0.84 (s, 18H), 0.00 (s, 12H). MS (m/z): found 550.1 (M+Na)+. See FIG. 46 .
To a solution of 3,5-bis(((tert-butyldimethylsilyl)oxy)methyl)-N-(2-(2-(2-methoxyethoxy)ethoxy)ethyl)aniline (6.7 g, 12.69 mmol) in anhydrous 1,2-dichloroethane (50 ml) was added 2-(methyldithio)isobutyraldehyde (2.74 ml, 19.04 mmol), sodium triacetoxyborohydride (2.8 g, 1 eq), zinc(II) chloride (0.865 g, 6.35 mmol) and magnesium sulfate (2.292 g, 19.04 mmol). The mixture was stirred for five hours at ambient temperature. Sodium triacetoxyborohydride (2.8 g, 1 eq) was added. The reaction continued to stir at ambient temperature for 20 hours. The mixture was filtered through celite rinsing with dichloromethane and concentrated under reduced pressure then extracted with ethyl acetate and water. The organic extracts were washed with brine, dried over magnesium sulfate, filtered, concentrated and purified by combiflash (EtOAc/Hex) to give 3,5-bis(((tert-butyldimethylsilyl)oxy)methyl)-N-(2-(2-(2-methoxyethoxy)ethoxy)ethyl)-N-(2-methyl-2-(methyldisulfanyl)propyl)aniline (3.5 g, 40%). 1H NMR (400 Hz, CDCl3): δ 6.73 (s, 2H), 6.59 (s, 1H), 4.56 (s, 4H), 3.65-3.51 (m, 14H), 3.30 (s, 3H), 2.38 (s, 3H), 1.28 (s, 6H), 0.84 (s, 18H), 0.00 (s, 12H). MS (m/z): found 684.2 (M+Na)+. See FIG. 46 .
Tetrabutylammonium fluoride (1M in THF) (10.57 ml, 10.57 mmol) was added dropwise to stirring solution of 3,5-bis(((tert-butyldimethylsilyl)oxy)methyl)-N-(2-(2-(2-methoxyethoxy)ethoxy)ethyl)-N-(2-methyl-2-(methyldisulfanyl)propyl)aniline (3.5 g, 5.29 mmol) in anhydrous THF (65 ml) at 0° C. in an ice bath. Following addition the mixture was stirred at ambient temperature for two hours. The mixture was quenched with saturated ammonium chloride and extracted with ethyl acetate. The extracts were washed with water and brine, dried with magnesium sulfate, filtered and concentrated under reduced pressure. Purification by silica gel chromatography (MeOH/DCM) yielded (5-((2-(2-(2-methoxyethoxy)ethoxy)ethyl)(2-methyl-2-(methyldisulfanyl)propyl)amino)-1,3-phenylene)dimethanol (2 g, 87%). 1H NMR (400 Hz, CDCl3): δ 6.76 (s, 2H), 6.63 (s, 1H), 4.55 (s, 4H), 3.65-3.51 (m, 14H), 3.35 (s, 3H), 2.44 (s, 3H), 1.33 (s, 6H); 13C NMR (400 Hz, CDCl3): δ 149.0, 142.35, 114.0, 111.1, 71.98, 70.7, 70.6, 70.5, 67.6, 65.5, 59.75, 59.1, 53.9, 51.9, 26.6, 25.7, 20.75; MS (m/z): found 456.2 (M+Na)+. See FIG. 46 .
(5-amino-1,3-phenylene)dimethanol (2.5 g, 16.32 mmol) and 2-(methyldithio)isobutyraldehyde (2.347 ml, 16.32 mmol) were stirred at ambient temperature in absolute ethanol (82 ml) until completely dissolved (3 hours). The mixture was cooled to 0° C. in an ice bath and sodium borohydride (0.741 g, 19.59 mmol) was added. The reaction was stirred for 1 hour at 0° C., and was then quenched slowly with cold 5% HCl solution. The mixture was diluted with dichloromethane and the pH was adjusted to pH=8 with saturated sodium bicarbonate solution then extracted with dichloromethane and then washed with brine. The organic extracts were dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. Purification by silica gel chromatography (MeOH/DCM) yielded (5-(2-methyl-2-(methyldisulfanyl)propylamino)-1,3-phenylene)dimethanol (3 g, 65%) as a white solid. 1H NMR (400 Hz, CDCl3): δ 6.62 (s, 1H), 6.54 (s, 2H), 4.53 (s, 4H), 3.13 (s, 2H), 2.30 (s, 3H), 1.32 (s, 6H). See FIG. 47 .
To a solution of 9-hydroxy-8-methoxy-11,12,12a,13-tetrahydro-6H-benzo[5,6][1,4]diazepino[1,2-a]indol-6-one 3b (0.3 g, 1.012 mmol) in methanol (5.06 ml) were added di-tert-butyl dicarbonate (0.265 g, 1.215 mmol), triethylamine (0.212 ml, 1.519 mmol) and DMAP (6.18 mg, 0.051 mmol). After 5 hours of stirring at ambient temperature the reaction mixture was concentrated in vacuo. The residue was redissolved in dichloromethane and filtered through celite. Purification by silica gel chromatography (20% EtOAc/DCM) yielded tert-butyl 9-hydroxy-8-methoxy-6-oxo-12a,13-dihydro-6H-benzo[5,6][1,4]diazepino[1,2-a]indole-11(12H)-carboxylate (0.21 g, 52%) as a white solid. 1H NMR (400 Hz, CDCl3): δ 8.25 (d, J=8.0 Hz, 1H), 7.44 (s, 1H), 7.18 (t, J=7.2 Hz, 1H), 7.11 (d, J=7.2 Hz, 1H), 6.98 (t, J=7.2 Hz, 1H), 6.39 (s, 1H), 4.37 (m, 1H), 3.75 (s, 3H), 3.42 (m, 3H), 2.74 (dd, J=3.6, 16.4 Hz, 1H), 1.47 (s, 9H). See FIG. 48 .
Preparation and Testing of huMy9-6-31c
A reaction containing 2.0 mg/mL huMy9-6 antibody and 5 molar equivalents of compound 31c (pretreated with 5-fold excess of sodium bisulfite in 90:10 DMA:water) in 50 mM HEPES (4-(2-hydroxyethyl)-1-piperazine ethanesulfonic acid) pH 8.5 buffer and 10% v/v DMA (N,N-Dimethylacetamide) cosolvent was allowed to conjugate for 6 hours at 25° C. Post-reaction, the conjugate was purified and buffer exchanged into 250 mM Glycine, 10 mM Histidine, 1% sucrose, 0.01% Tween-20, 50 μM sodium bisulfite formulation buffer, pH 6.2, using NAP desalting columns (Illustra Sephadex G-25 DNA Grade, GE Healthcare). Dialysis was performed in the same buffer for 4 hours at room temperature utilizing Slide-a-Lyzer dialysis cassettes (ThermoScientific 20,000 MWCO).
The purified conjugate was found to have an average of 3.1 IGN molecules linked per antibody (by UV-Vis using molar extinction coefficients ε330 nm=15,484 cm−1M−1 and ε280 nm=30, 115 cm−1M−1 for 1, and ε280 nm=207,000 cm−1M−1 for My9-6 antibody), 98% monomer (by size exclusion chromatography), <0.2% unconjugated drug (by dual-column reverse-phase HPLC analysis) and a final protein concentration of 0.4 mg/ml.
In vitro potency measurements for conjugates of huMy9-6 with 31c at two different drug loads were shown below. Both conjugates were highly potent towards antigen-positive HL60-QC cells, with IC50 values between 1.3-1.8 pM. Antigen blocking with 1 μM unconjugated huMy9-6 significantly diminished the potency, demonstrating the antigen specificity of the cytotoxic effect.
| IC50 (pM) | ||||
| Conjugate | huMy9-6 | Specificity | ||
| huMy9-6-31c | IC50 (pM) | blocking | window | |
| 3.1 IGN/Ab | 1.8 | 940 | 522 | |
| 3.9 IGN/Ab | 1.3 | 790 | 608 | |
In Vivo Efficacy of Various Conjugates in Tumor Bearing Nude Mice
In this study, the anti-tumor activity of several conjugates of the invention are investigated in immune-compromised mice (nude or SCID), preferably female nude mice, bearing various tumors. In some cases, in addition or as an alternative, nude rats may be employed. The conjugates to be tested include any one or more of the conjugates described herein. The various tumor cell lines that can be used for inoculating the nude mice included HL60/QC, MOLM-13, NB4, HEL92.1.7, OCI-AML3, KB, and/or any other cancer cell lines recognized in the art as a proper model for a disease indication (e.g., cancer). Some criteria that may be applied for the selection of tumor cell lines suitable for in vivo evaluation include: a) expression of the target antigen on the tumor cell, and, b) sensitivity of tumor cells to the unconjugated drug in vitro. For example, an in vitro cell line sensitivity screen, such as the 60-cell line screen described by the U.S. National Cancer Institute (see Voskoglou-Nomikos et al., 2003, Clinical Cancer Res. 9; 42227-4239, incorporated herein by reference) can be used as one of the guides to determine the types of cancers that may be suitable for treatment with the compounds of the invention. The potency of the various conjugates against the various tumor cell lines, as expressed by IC50 values (nM), is measured accordingly.
The various tumor cell lines are inoculated to nude or SCID mice using substantially the same protocol as outlined in Example 15. For example, about 1×106-5×107 tumor cells (typically 1×107) cells/mouse are subcutaneously inoculated at a volume of approximately 0.1-0.2 mL/mouse, in the area over the right shoulder of female athymic nude mice, 6 weeks of age. When the tumor has reached an average size of ˜100 mm3 (typically 6 to 8 days after tumor cell inoculation), mice are randomized into groups (e.g., n=5-8 per group) by tumor volume. Treatment is initiated the day after randomization, and groups includes a control group dosed with the appropriate vehicle (200 μL/injection), or a single treatment at various doses (5 to 700 μg/kg) of the above referenced drug conjugates (50 μg/kg linked drug dose corresponded to about 2 mg/kg antibody dose). Multiple dosing schedules (for example treatment at day 1, 3, 5, or day 1, 4, 7) may also be employed.
Median and mean tumor volume vs time is measured, with the data demonstrating a dose-dependent anti-tumor activity of the subject conjugates. The minimum effective dose is then calculated and compared to the maximum tolerated dose.
A reaction containing 6 mg/mL huMy9-6 antibody and 5 molar equivalents of the highly reactive N-succinimidyl-4-(4-nitropyridyl-2-dithio)butanoate linker (20 mM stock in ethanol) was incubated for 3 h at 25° C. in 50 mM EPPS buffer at pH 8. Unreacted linker was removed using a NAP desalting column (Illustra Sephadex G-25 DNA Grade, GE Healthcare). The linker to antibody ratio (LAR) was determined to be about 2.3 based on antibody concentration and DTT-released nitropyridine-2-thione concentration by UV-Vis (ε394 nm=14205 cm−1M−1 for 2-thio-4-nitropyridone).
Linker modified huMy9-6 was diluted to 2 mg/mL in 50 mM HEPES buffer at pH 8.5, 10% v/v DMA, and reacted with 2 molar equivalents of compound 1d per linker (5 mM stock in DMA; 4.6 equivalents per antibody) for 30 min at 25° C. Completion of disulfide exchange reaction was determined by monitoring absorbance increase at 394 nm by UV.
Post-reaction, the conjugate was purified and buffer exchanged into 250 mM glycine, 10 mM histidine, 1% sucrose, 0.01% Tween-20, 50 μM sodium bisulfite at pH 6.2 using a desalting column (G-25 Sephadex, fine grade, GE Healthcare).
The purified conjugate was found to have an average of 2.1 molecules of 1d linked per antibody (by UV-Vis using molar extinction coefficients ε330 nm=15,484 cm−1M−1 and ε280 nm=30, 115 cm−1M−1 for 1d, and ε280 nm=207,000 cm−1M−1 for huMy9-6), 98% monomer (by size exclusion chromatography), <1% unconjugated 1d (by acetone extraction/reverse-phase HPLC), a 70% protein yield, and a 32% overall 1d yield. See FIG. 60 .
Claims (14)
3. The method of claim 2 , wherein the palladium catalyst is palladium on carbon (Pd/C).
4. The method of claim 2 , wherein the reaction is carried out in methanol.
5. The method of claim 2 , wherein the reaction is carried out at room temperature.
6. The method of claim 5 , wherein the reaction is carried out at room temperature overnight.
7. The method of claim 2 , wherein the method further comprises purifying compound 3b by silica gel chromatography.
8. The method of claim 3 , wherein the reaction is carried out at room temperature.
9. The method of claim 8 , wherein the reaction is carried out at room temperature overnight.
10. The method of claim 3 , wherein the method further comprises purifying compound 3b by silica gel chromatography.
11. The method of claim 2 , wherein the palladium catalyst is palladium on carbon (Pd/C) and the reaction is carried out in methanol.
12. The method of claim 11 , wherein the reaction is carried out at room temperature.
13. The method of claim 12 , wherein the reaction is carried out at room temperature overnight.
14. The method of claim 11 , wherein the method further comprises purifying compound 3b by silica gel chromatography.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17/316,241 USRE49918E1 (en) | 2011-02-15 | 2021-05-10 | Cytotoxic benzodiazepine derivatives |
Applications Claiming Priority (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201161443092P | 2011-02-15 | 2011-02-15 | |
| US201161443062P | 2011-02-15 | 2011-02-15 | |
| US201161483499P | 2011-05-06 | 2011-05-06 | |
| US13/397,195 US8765740B2 (en) | 2011-02-15 | 2012-02-15 | Cytotoxic benzodiazepine derivatives |
| US13/827,355 US8889669B2 (en) | 2011-02-15 | 2013-03-14 | Cytotoxic benzodiazepine derivatives |
| US14/512,059 US9169272B2 (en) | 2011-02-15 | 2014-10-10 | Cytotoxic benzodiazepine derivatives |
| US14/849,270 US9434748B2 (en) | 2011-02-15 | 2015-09-09 | Cytotoxic benzodiazepine derivatives |
| US15/221,255 US9840564B2 (en) | 2011-02-15 | 2016-07-27 | Cytotoxic benzodiazepine derivatives |
| US15/645,597 US10179818B2 (en) | 2011-02-15 | 2017-07-10 | Cytotoxic benzodiazepine derivatives |
| US16/205,609 US10570212B2 (en) | 2011-02-15 | 2018-11-30 | Cytotoxic benzodiazepine derivatives |
| US17/316,241 USRE49918E1 (en) | 2011-02-15 | 2021-05-10 | Cytotoxic benzodiazepine derivatives |
Related Parent Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/645,597 Continuation US10179818B2 (en) | 2011-02-15 | 2017-07-10 | Cytotoxic benzodiazepine derivatives |
| US16/205,609 Reissue US10570212B2 (en) | 2011-02-15 | 2018-11-30 | Cytotoxic benzodiazepine derivatives |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE49918E1 true USRE49918E1 (en) | 2024-04-16 |
Family
ID=45755579
Family Applications (12)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/397,195 Active 2032-08-30 US8765740B2 (en) | 2011-02-15 | 2012-02-15 | Cytotoxic benzodiazepine derivatives |
| US13/984,762 Expired - Fee Related US9534000B2 (en) | 2011-02-15 | 2012-02-15 | Cytotoxic benzodiazepine derivatives and methods of preparation |
| US13/397,205 Active 2034-06-28 US9353127B2 (en) | 2011-02-15 | 2012-02-15 | Methods of preparation of conjugates |
| US13/827,355 Active US8889669B2 (en) | 2011-02-15 | 2013-03-14 | Cytotoxic benzodiazepine derivatives |
| US14/512,059 Active US9169272B2 (en) | 2011-02-15 | 2014-10-10 | Cytotoxic benzodiazepine derivatives |
| US14/849,270 Expired - Fee Related US9434748B2 (en) | 2011-02-15 | 2015-09-09 | Cytotoxic benzodiazepine derivatives |
| US15/097,979 Expired - Fee Related US9868791B2 (en) | 2011-02-15 | 2016-04-13 | Methods of preparation of conjugates |
| US15/221,255 Active US9840564B2 (en) | 2011-02-15 | 2016-07-27 | Cytotoxic benzodiazepine derivatives |
| US15/645,597 Active US10179818B2 (en) | 2011-02-15 | 2017-07-10 | Cytotoxic benzodiazepine derivatives |
| US15/677,846 Expired - Fee Related US10364294B2 (en) | 2011-02-15 | 2017-08-15 | Methods of preparation of conjugates |
| US16/205,609 Ceased US10570212B2 (en) | 2011-02-15 | 2018-11-30 | Cytotoxic benzodiazepine derivatives |
| US17/316,241 Active USRE49918E1 (en) | 2011-02-15 | 2021-05-10 | Cytotoxic benzodiazepine derivatives |
Family Applications Before (11)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/397,195 Active 2032-08-30 US8765740B2 (en) | 2011-02-15 | 2012-02-15 | Cytotoxic benzodiazepine derivatives |
| US13/984,762 Expired - Fee Related US9534000B2 (en) | 2011-02-15 | 2012-02-15 | Cytotoxic benzodiazepine derivatives and methods of preparation |
| US13/397,205 Active 2034-06-28 US9353127B2 (en) | 2011-02-15 | 2012-02-15 | Methods of preparation of conjugates |
| US13/827,355 Active US8889669B2 (en) | 2011-02-15 | 2013-03-14 | Cytotoxic benzodiazepine derivatives |
| US14/512,059 Active US9169272B2 (en) | 2011-02-15 | 2014-10-10 | Cytotoxic benzodiazepine derivatives |
| US14/849,270 Expired - Fee Related US9434748B2 (en) | 2011-02-15 | 2015-09-09 | Cytotoxic benzodiazepine derivatives |
| US15/097,979 Expired - Fee Related US9868791B2 (en) | 2011-02-15 | 2016-04-13 | Methods of preparation of conjugates |
| US15/221,255 Active US9840564B2 (en) | 2011-02-15 | 2016-07-27 | Cytotoxic benzodiazepine derivatives |
| US15/645,597 Active US10179818B2 (en) | 2011-02-15 | 2017-07-10 | Cytotoxic benzodiazepine derivatives |
| US15/677,846 Expired - Fee Related US10364294B2 (en) | 2011-02-15 | 2017-08-15 | Methods of preparation of conjugates |
| US16/205,609 Ceased US10570212B2 (en) | 2011-02-15 | 2018-11-30 | Cytotoxic benzodiazepine derivatives |
Country Status (29)
| Country | Link |
|---|---|
| US (12) | US8765740B2 (en) |
| EP (6) | EP3498303A1 (en) |
| JP (8) | JP5826863B2 (en) |
| KR (6) | KR20190089048A (en) |
| CN (5) | CN113896731A (en) |
| AU (5) | AU2012217719B2 (en) |
| BR (2) | BR112013019913B1 (en) |
| CA (4) | CA3110544A1 (en) |
| CY (2) | CY1117340T1 (en) |
| DK (2) | DK2675479T3 (en) |
| ES (2) | ES2717657T3 (en) |
| HR (2) | HRP20160164T1 (en) |
| HU (2) | HUE028736T2 (en) |
| IL (6) | IL279304B (en) |
| LT (1) | LT2675480T (en) |
| ME (1) | ME02381B (en) |
| MX (3) | MX384514B (en) |
| MY (2) | MY169448A (en) |
| PL (2) | PL2675479T3 (en) |
| PT (1) | PT2675480T (en) |
| RS (2) | RS58620B1 (en) |
| RU (4) | RU2631498C2 (en) |
| SG (4) | SG10202007640VA (en) |
| SI (2) | SI2675479T1 (en) |
| SM (2) | SMT201900181T1 (en) |
| TW (1) | TWI617555B (en) |
| UA (1) | UA120696C2 (en) |
| WO (3) | WO2012112687A1 (en) |
| ZA (2) | ZA201305386B (en) |
Families Citing this family (167)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101098854B (en) | 2004-07-23 | 2012-12-05 | 恩多塞特公司 | Bivalent linkers and conjugates thereof |
| CA3000520C (en) | 2005-08-24 | 2023-04-04 | Immunogen, Inc. | Process for preparing antibody maytansinoid conjugates |
| CN101784565B (en) | 2007-06-25 | 2014-12-10 | 恩多塞特公司 | Conjugates containing a hydrophilic spacer linker |
| US9877965B2 (en) | 2007-06-25 | 2018-01-30 | Endocyte, Inc. | Vitamin receptor drug delivery conjugates for treating inflammation |
| RU2683325C2 (en) | 2009-02-05 | 2019-03-28 | Иммьюноджен, Инк. | New benzodiazepine derivatives |
| SG10201810743WA (en) | 2009-06-03 | 2018-12-28 | Immunogen Inc | Conjugation methods |
| CA2795349C (en) | 2010-04-15 | 2016-11-29 | Seattle Genetics, Inc. | Targeted pyrrolobenzodiazepine conjugates |
| JP5875083B2 (en) | 2010-04-15 | 2016-03-02 | メディミューン リミテッド | Pyrrolobenzodiazepine for the treatment of proliferative diseases |
| JP5826863B2 (en) * | 2011-02-15 | 2015-12-02 | イミュノジェン・インコーポレーテッド | Cytotoxic benzodiazepine derivatives |
| ME03353B (en) * | 2011-03-29 | 2019-10-20 | Immunogen Inc | MAYTANSINOID ANTIBODY CONJUGATE PREPARATION BY ONE-STEP PROCESS |
| SG11201400770SA (en) | 2011-09-20 | 2014-04-28 | Spirogen Sarl | Pyrrolobenzodiazepines as unsymmetrical dimeric pbd compounds for inclusion in targeted conjugates |
| CA2850373C (en) | 2011-10-14 | 2019-07-16 | Seattle Genetics, Inc. | Pyrrolobenzodiazepines and targeted conjugates |
| CN103997893B (en) | 2011-10-14 | 2019-04-12 | 西雅图基因公司 | Pyrrolobenzodiazepines and targeted conjugates |
| AU2012322932B2 (en) | 2011-10-14 | 2016-11-10 | Medimmune Limited | Pyrrolobenzodiazepines |
| EP2751111B1 (en) | 2011-10-14 | 2017-04-26 | MedImmune Limited | Asymmetrical bis-(5H-Pyrrolo[2,1-c][1,4]benzodiazepin-5-one) derivatives for the treatment of proliferative or autoimmune diseases |
| EP2793947B1 (en) | 2011-12-23 | 2021-02-03 | Innate Pharma | Enzymatic conjugation of polypeptides |
| CA2869704A1 (en) * | 2012-04-04 | 2013-10-10 | Perseus Proteomics Inc. | Drug conjugate comprising anti-cdh3 (pcadherin) antibody |
| US9504756B2 (en) | 2012-05-15 | 2016-11-29 | Seattle Genetics, Inc. | Self-stabilizing linker conjugates |
| WO2013173337A2 (en) | 2012-05-15 | 2013-11-21 | Seattle Genetics, Inc. | Self-stabilizing linker conjugates |
| US10132799B2 (en) | 2012-07-13 | 2018-11-20 | Innate Pharma | Screening of conjugated antibodies |
| HK1211208A1 (en) | 2012-08-22 | 2016-05-20 | Immunogen, Inc. | Cytotoxic benzodiazepine derivative |
| RU2661083C2 (en) | 2012-10-04 | 2018-07-11 | Иммуноджен, Инк. | Use of pvdf membrane to purify cell-binding agent - cytotoxic agent conjugates |
| EP2906296B1 (en) * | 2012-10-12 | 2018-03-21 | ADC Therapeutics SA | Pyrrolobenzodiazepine-antibody conjugates |
| EP2906249B1 (en) | 2012-10-12 | 2018-06-27 | MedImmune Limited | Synthesis and intermediates of pyrrolobenzodiazepine derivatives for conjugation |
| EP2906297B1 (en) * | 2012-10-12 | 2017-12-06 | ADC Therapeutics SA | Pyrrolobenzodiazepine-antibody conjugates |
| JP6392765B2 (en) | 2012-10-12 | 2018-09-19 | エイディーシー・セラピューティクス・エス・アーAdc Therapeutics Sa | Pyrrolobenzodiazepine-antibody conjugate |
| HRP20190366T1 (en) | 2012-10-12 | 2019-04-19 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| RS57694B1 (en) | 2012-10-12 | 2018-11-30 | Adc Therapeutics Sa | Pyrrolobenzodiazepine - anti-psma antibody conjugates |
| HRP20182129T1 (en) * | 2012-10-12 | 2019-02-08 | Adc Therapeutics Sa | CONJUGATES ANTIBODY - PIROLOBENZODIAZEPINE |
| ES2680153T3 (en) | 2012-10-12 | 2018-09-04 | Adc Therapeutics Sa | Anti-PSMA-pyrrolobenzodiazepine antibody conjugates |
| WO2014057118A1 (en) * | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sarl | Pyrrolobenzodiazepine-anti-cd22 antibody conjugates |
| MX338711B (en) | 2012-10-12 | 2016-04-28 | Medimmune Ltd | Pyrrolobenzodiazepines and conjugates thereof. |
| EP3564259A3 (en) | 2012-11-09 | 2020-02-12 | Innate Pharma | Recognition tags for tgase-mediated conjugation |
| AU2013341711A1 (en) * | 2012-11-12 | 2015-05-21 | Redwood Bioscience, Inc. | Compounds and methods for producing a conjugate |
| WO2014089177A2 (en) | 2012-12-04 | 2014-06-12 | Massachusetts Institute Of Technology | Compounds, conjugates and compositions of epipolythiodiketopiperazines and polythiodiketopiperazines |
| US9089614B2 (en) | 2012-12-21 | 2015-07-28 | Bioalliance C.V. | Hydrophilic self-immolative linkers and conjugates thereof |
| CN105189507A (en) | 2012-12-21 | 2015-12-23 | 斯皮罗根有限公司 | Pyrrolobenzodiazepines and conjugates thereof |
| CN110627797A (en) * | 2012-12-21 | 2019-12-31 | 麦迪穆有限责任公司 | Asymmetric pyrrolobenzodiazepine dimers for the treatment of proliferative and autoimmune diseases |
| AU2014248640B2 (en) | 2013-03-13 | 2018-03-01 | Seagen Inc. | Cyclodextrin and antibody-drug conjugate formulations |
| DK2968494T3 (en) | 2013-03-13 | 2019-03-04 | Seattle Genetics Inc | ACTIVATED CARBON FILTERING FOR CLEANING BENZODIAZEPINE ADCs |
| AU2014229529B2 (en) | 2013-03-13 | 2018-02-15 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| TWI680766B (en) * | 2013-03-13 | 2020-01-01 | 英商梅迪繆思有限公司 | Pyrrolobenzodiazepines and conjugates thereof |
| US10611824B2 (en) | 2013-03-15 | 2020-04-07 | Innate Pharma | Solid phase TGase-mediated conjugation of antibodies |
| EP3010547B1 (en) | 2013-06-20 | 2021-04-21 | Innate Pharma | Enzymatic conjugation of polypeptides |
| CN105517577A (en) * | 2013-06-21 | 2016-04-20 | 先天制药公司 | Enzymatic conjugation of polypeptides |
| US10208125B2 (en) | 2013-07-15 | 2019-02-19 | University of Pittsburgh—of the Commonwealth System of Higher Education | Anti-mucin 1 binding agents and uses thereof |
| SG11201601424PA (en) | 2013-08-28 | 2016-03-30 | Stemcentrx Inc | Site-specific antibody conjugation methods and compositions |
| CN105792836A (en) | 2013-08-28 | 2016-07-20 | 施特姆森特克斯股份有限公司 | Novel SEZ6 modulators and methods of use |
| KR20160047567A (en) * | 2013-08-28 | 2016-05-02 | 스템센트알엑스 인코포레이티드 | Engineered anti-dll3 conjugates and methods of use |
| EP3041846B1 (en) * | 2013-09-02 | 2018-11-07 | Hangzhou Dac Biotech Co., Ltd | Novel cytotoxic agents for conjugation of drugs to cell binding molecule |
| EP3054983B1 (en) | 2013-10-11 | 2019-03-20 | Medimmune Limited | Pyrrolobenzodiazepine-antibody conjugates |
| GB201317981D0 (en) | 2013-10-11 | 2013-11-27 | Spirogen Sarl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2015052535A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052534A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| NZ758049A (en) | 2013-10-15 | 2024-03-22 | Seagen Inc | Pegylated drug-linkers for improved ligand-drug conjugate pharmacokinetics |
| KR20230088389A (en) | 2014-02-11 | 2023-06-19 | 씨젠 인크. | Selective reduction of proteins |
| CA2939941A1 (en) | 2014-02-21 | 2015-08-27 | Abbvie Stemcentrx Llc | Anti-dll3 antibodies and drug conjugates for use in melanoma |
| US10550190B2 (en) * | 2014-04-04 | 2020-02-04 | Merck Sharp & Dohme Corp. | Phosphate based linkers for intracellular delivery of drug conjugates |
| EP3145542A4 (en) * | 2014-05-20 | 2018-01-17 | ImmunoGen, Inc. | Methods for characterizing and treating acute myeloid leukemia |
| US9950077B2 (en) * | 2014-06-20 | 2018-04-24 | Bioalliance C.V. | Anti-folate receptor alpha (FRA) antibody-drug conjugates and methods of using thereof |
| WO2016008112A1 (en) | 2014-07-16 | 2016-01-21 | Medshine Discovery Inc. | Linkers and application towards adc thereof |
| SG11201701328XA (en) * | 2014-09-02 | 2017-03-30 | Immunogen Inc | Methods for formulating antibody drug conjugate compositions |
| WO2016036804A1 (en) | 2014-09-03 | 2016-03-10 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| AU2015311987B2 (en) * | 2014-09-03 | 2020-06-04 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| KR102508023B1 (en) * | 2014-09-03 | 2023-03-10 | 이뮤노젠 아이엔씨 | Conjugates comprising cell-binding agents and cytotoxic agents |
| GB201416112D0 (en) * | 2014-09-12 | 2014-10-29 | Medimmune Ltd | Pyrrolobenzodiazepines and conjugates thereof |
| LT3191135T (en) | 2014-09-12 | 2020-11-25 | Genentech, Inc. | ANTI-HER2 ANTIBODIES AND IMMUNOCONJUGATES |
| MX2017003472A (en) * | 2014-09-17 | 2017-10-31 | Genentech Inc | Immunoconjugates comprising anti-her2 antibodies and pyrrolobenzodiazepines. |
| GB201419108D0 (en) | 2014-10-27 | 2014-12-10 | Glythera Ltd | Materials and methods relating to linkers for use in antibody drug conjugates |
| US10413615B2 (en) * | 2014-11-19 | 2019-09-17 | Immunogen, Inc. | Process for preparing cell-binding agent-cytotoxic agent conjugates |
| CN107148285B (en) | 2014-11-25 | 2022-01-04 | Adc治疗股份有限公司 | Pyrrolobenzodiazepine-antibody conjugates |
| CN113209306A (en) * | 2014-12-09 | 2021-08-06 | 艾伯维公司 | Antibody drug conjugates with cell permeable BCL-XL inhibitors |
| JP6800853B2 (en) | 2014-12-11 | 2020-12-16 | ピエール、ファーブル、メディカマン | Anti-C10ORF54 antibody and its use |
| BR112017014937A2 (en) | 2015-01-14 | 2018-03-13 | Bristol-Myers Squibb Company | heteroarylene bridged benzodiazepine dimers, conjugates thereof, and methods of preparation and use |
| MX2017009145A (en) | 2015-01-14 | 2017-11-22 | Bristol Myers Squibb Co | Benzodiazepine dimers, conjugates thereof, and methods of making and using. |
| WO2016148674A1 (en) * | 2015-03-13 | 2016-09-22 | Endocyte, Inc. | Conjugates for treating diseases |
| EP3270903B1 (en) | 2015-03-17 | 2020-05-06 | Arrowhead Pharmaceuticals, Inc. | Disulfide-containing alkyne linking agents |
| US9504694B2 (en) | 2015-03-19 | 2016-11-29 | Cellerant Therapeutics, Inc. | Isoquinolidinobenzodiazepines |
| GB201506411D0 (en) | 2015-04-15 | 2015-05-27 | Bergenbio As | Humanized anti-axl antibodies |
| GB201506402D0 (en) | 2015-04-15 | 2015-05-27 | Berkel Patricius H C Van And Howard Philip W | Site-specific antibody-drug conjugates |
| EA039619B1 (en) * | 2015-05-20 | 2022-02-17 | Иммуноджен, Инк. | Method for producing indolinobenzodiazepine compounds |
| GB201510010D0 (en) | 2015-06-09 | 2015-07-22 | King S College London | PDD and BPD compounds |
| CA2990030A1 (en) | 2015-06-23 | 2016-12-29 | Bristol-Myers Squibb Company | Macrocyclic benzodiazepine dimers, conjugates thereof, preparation and uses |
| SMT202000614T1 (en) * | 2015-06-29 | 2021-01-05 | Immunogen Inc | Conjugates of cysteine engineered antibodies |
| AU2016285552B2 (en) * | 2015-06-29 | 2022-10-13 | Immunogen, Inc. | Anti-CD123 antibodies and conjugates and derivatives thereof |
| EP3325482B1 (en) * | 2015-07-21 | 2020-06-24 | ImmunoGen, Inc. | Methods of preparing cytotoxic benzodiazepine derivatives |
| GB201514928D0 (en) | 2015-08-21 | 2015-10-07 | King S College London | PDD compounds |
| US20180339985A1 (en) | 2015-08-21 | 2018-11-29 | Femtogenix Limited | Pdd compounds |
| CR20180243A (en) | 2015-10-02 | 2018-07-31 | Genentech Inc | PIRROLOBENZODIAZEPIN ANTIBODY-DRUG CONJUGATES AND METHODS OF USE |
| US20170189548A1 (en) * | 2015-11-25 | 2017-07-06 | Immunogen, Inc. | Pharmaceutical formulations and methods of use thereof |
| IL260289B (en) | 2016-01-08 | 2022-08-01 | Bioalliance Cv | Quadrivalent anti-psgl-1 antibodies and uses thereof |
| GB201601431D0 (en) | 2016-01-26 | 2016-03-09 | Medimmune Ltd | Pyrrolobenzodiazepines |
| TW201731532A (en) * | 2016-02-05 | 2017-09-16 | 伊繆諾金公司 | Efficient process for preparing cell-binding agent-cytotoxic agent conjugates |
| EA201891723A1 (en) * | 2016-02-05 | 2018-12-28 | Милленниум Фармасьютикалз, Инк. | GCC-directed anti-body drug conjugates |
| GB201602356D0 (en) | 2016-02-10 | 2016-03-23 | Medimmune Ltd | Pyrrolobenzodiazepine Conjugates |
| GB201602359D0 (en) | 2016-02-10 | 2016-03-23 | Medimmune Ltd | Pyrrolobenzodiazepine Conjugates |
| CN109843919A (en) | 2016-03-25 | 2019-06-04 | 西雅图基因公司 | The method for being used to prepare the agent-linker and its intermediate of Pegylation |
| WO2017172930A1 (en) * | 2016-03-29 | 2017-10-05 | Endocyte, Inc. | Pbd conjugates for treating diseases |
| BR112018071465A2 (en) * | 2016-04-20 | 2019-02-05 | Hangzhou Dac Biotech Co Ltd | amanita toxin derivatives and their conjugation to a cell binding molecule |
| JP2019522960A (en) | 2016-04-21 | 2019-08-22 | アッヴィ・ステムセントルクス・エル・エル・シー | Novel anti-BMPR1B antibody and method of use |
| GB201607478D0 (en) | 2016-04-29 | 2016-06-15 | Medimmune Ltd | Pyrrolobenzodiazepine Conjugates |
| WO2017197045A1 (en) | 2016-05-11 | 2017-11-16 | Movassaghi Mohammad | Convergent and enantioselective total synthesis of communesin analogs |
| TW201808936A (en) | 2016-05-18 | 2018-03-16 | 美商梅爾莎納醫療公司 | Pyrrolobenzodiazepines and conjugates thereof |
| CN109641910A (en) | 2016-06-24 | 2019-04-16 | 梅尔莎纳医疗公司 | Pyrroles acene phenodiazine * and its conjugate |
| TWI851531B (en) | 2016-08-09 | 2024-08-11 | 美商思進公司 | Drug conjugates with self-stabilizing linkers having improved physiochemical properties |
| GB201617466D0 (en) | 2016-10-14 | 2016-11-30 | Medimmune Ltd | Pyrrolobenzodiazepine conjugates |
| RU2019114863A (en) | 2016-11-02 | 2020-12-03 | Иммуноджен, Инк. | COMBINED TREATMENT WITH ANTIBODY-DRUG CONJUGATES AND PARP INHIBITORS |
| MY200695A (en) * | 2016-11-03 | 2024-01-11 | Bristol Myers Squibb Co | Activatable anti-ctla-4 antibodies and uses thereof |
| WO2018098258A2 (en) | 2016-11-23 | 2018-05-31 | Immunogen, Inc. | Selective sulfonation of benzodiazepine derivatives |
| JP7244987B2 (en) | 2016-12-14 | 2023-03-23 | シージェン インコーポレイテッド | Multidrug Antibody Drug Conjugates |
| EP3554544A4 (en) | 2016-12-16 | 2020-07-29 | Bluefin Biomedicine, Inc. | ANTI-CUB-CONTAINING PROTEIN 1 (CDCP1) ANTIBODIES, ANTIBODY-ACTIVE SUBSTANCE CONJUGATES AND METHOD FOR USE THEREOF |
| PL3558391T3 (en) | 2016-12-23 | 2022-05-16 | Immunogen, Inc. | ADAM9 PROTEIN IMMUNOCONIUGATES AND THEIR APPLICATION |
| US20180230218A1 (en) | 2017-01-04 | 2018-08-16 | Immunogen, Inc. | Met antibodies and immunoconjugates and uses thereof |
| CN110300765B (en) | 2017-01-24 | 2024-08-23 | 依奈特制药公司 | Nkp46 binders |
| EP4148044A1 (en) | 2017-01-25 | 2023-03-15 | ImmunoGen, Inc. | Methods of preparing cytotoxic benzodiazepine derivatives |
| EP3577138A1 (en) | 2017-02-06 | 2019-12-11 | Innate Pharma | Immunomodulatory antibody drug conjugates binding to a human mica polypeptide |
| RS61795B1 (en) | 2017-02-08 | 2021-06-30 | Adc Therapeutics Sa | Pyrrolobenzodiazepine-antibody conjugates |
| GB201702031D0 (en) | 2017-02-08 | 2017-03-22 | Medlmmune Ltd | Pyrrolobenzodiazepine-antibody conjugates |
| KR102648564B1 (en) | 2017-03-24 | 2024-03-19 | 씨젠 인크. | Manufacturing process of glucuronide drug-linker and its intermediates |
| WO2018183494A1 (en) | 2017-03-31 | 2018-10-04 | Immunogen, Inc. | Cd19-targeting antibody-drug conjugates |
| CN106841192B (en) * | 2017-04-18 | 2019-05-03 | 乐凯(沈阳)科技产业有限责任公司 | A kind of diazepine structure directing agent detection carbaldehyde Method and its application |
| CN110582505B (en) | 2017-04-18 | 2021-04-02 | 免疫医疗有限公司 | Pyrrolobenzodiazepine* conjugates |
| IL305224B2 (en) * | 2017-04-20 | 2024-09-01 | Immunogen Inc | Methods for the preparation of indolinobenzodiazepine derivatives |
| WO2018195243A1 (en) | 2017-04-20 | 2018-10-25 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives and conjugates thereof |
| WO2018193102A1 (en) | 2017-04-20 | 2018-10-25 | Adc Therapeutics Sa | Combination therapy with an anti-axl antibody-drug conjugate |
| US11932650B2 (en) | 2017-05-11 | 2024-03-19 | Massachusetts Institute Of Technology | Potent agelastatin derivatives as modulators for cancer invasion and metastasis |
| CA3064804A1 (en) | 2017-06-14 | 2018-12-20 | Adc Therapeutics Sa | Dosage regimes for the administration of an anti-cd19 adc |
| US11566044B2 (en) | 2017-07-26 | 2023-01-31 | The University Of Queensland | Disulfide bond containing compounds and uses thereof |
| KR102270107B1 (en) | 2017-08-18 | 2021-06-30 | 메디뮨 리미티드 | pyrrolobenzodiazepine conjugate |
| NZ762505A (en) | 2017-09-29 | 2026-03-27 | Daiichi Sankyo Co Ltd | Antibody-pyrrolobenzodiazepine derivative conjugate |
| US10640508B2 (en) | 2017-10-13 | 2020-05-05 | Massachusetts Institute Of Technology | Diazene directed modular synthesis of compounds with quaternary carbon centers |
| US20190160089A1 (en) | 2017-10-31 | 2019-05-30 | Immunogen, Inc. | Combination treatment with antibody-drug conjugates and cytarabine |
| WO2019092148A1 (en) | 2017-11-10 | 2019-05-16 | Innate Pharma | Antibodies with functionalized glutamine residues |
| WO2019104289A1 (en) | 2017-11-27 | 2019-05-31 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepine antibody conjugates |
| US11793885B2 (en) | 2017-12-28 | 2023-10-24 | Immunogen, Inc. | Substituted benzo[5,6][1,4]diazepino[1,2-a]indoles for the treatment of proliferative disorders |
| US11274121B2 (en) | 2018-01-12 | 2022-03-15 | Immunogen, Inc. | Methods for antibody drug conjugation, purification, and formulation |
| GB201803342D0 (en) | 2018-03-01 | 2018-04-18 | Medimmune Ltd | Methods |
| EP3774923A4 (en) | 2018-03-28 | 2021-12-29 | Mitsubishi Tanabe Pharma Corporation | Drug conjugates of cmet monoclonal binding agents, and uses thereof |
| GB201806022D0 (en) | 2018-04-12 | 2018-05-30 | Medimmune Ltd | Pyrrolobenzodiazepines and conjugates thereof |
| WO2019222130A1 (en) | 2018-05-15 | 2019-11-21 | Immunogen, Inc. | Combination treatment with antibody-drug conjugates and flt3 inhibitors |
| TWI897855B (en) * | 2018-10-26 | 2025-09-21 | 美商免疫遺傳股份有限公司 | Epcam antibodies, activatable antibodies, and immunoconjugates, and uses thereof |
| CN113166053A (en) | 2018-11-12 | 2021-07-23 | 伊缪诺金公司 | Method for preparing cytotoxic benzodiazepine derivatives |
| MA54228A (en) | 2018-11-12 | 2021-09-22 | Immunogen Inc | METHODS FOR PREPARING CYTOTOXIC BENZODIAZEPINE DERIVATIVES |
| CN111393502B (en) * | 2019-01-03 | 2022-04-29 | 北京京东方技术开发有限公司 | A kind of peptoid and its preparation method and use |
| GB201901197D0 (en) | 2019-01-29 | 2019-03-20 | Femtogenix Ltd | G-A Crosslinking cytotoxic agents |
| MY206596A (en) | 2019-02-08 | 2024-12-25 | Janssen Pharmaceuticals Inc | Method of safe administration of phosphorylated tau peptide vaccine |
| CN113631560B (en) | 2019-03-15 | 2025-02-18 | 麦迪穆有限责任公司 | Azetidine benzodiazepine dimers and conjugates containing them for treating cancer |
| CN113661172A (en) * | 2019-03-29 | 2021-11-16 | 伊缪诺金公司 | Cytotoxic bisbenzodiazepine derivatives and their conjugates with cell-binding agents for inhibiting abnormal cell growth or treating proliferative diseases |
| SG11202110922QA (en) | 2019-04-26 | 2021-10-28 | Immunogen Inc | Camptothecin derivatives |
| US11535634B2 (en) | 2019-06-05 | 2022-12-27 | Massachusetts Institute Of Technology | Compounds, conjugates, and compositions of epipolythiodiketopiperazines and polythiodiketopiperazines and uses thereof |
| AU2021251875A1 (en) | 2020-04-10 | 2022-11-03 | Seagen Inc. | Charge variant linkers |
| MX2023005873A (en) | 2020-11-25 | 2023-06-05 | Innate Pharma | Treatment of cancer. |
| WO2022182415A1 (en) | 2021-02-24 | 2022-09-01 | Massachusetts Institute Of Technology | Himastatin derivatives, and processes of preparation thereof, and uses thereof |
| BR112023018676A2 (en) | 2021-03-18 | 2023-10-10 | Seagen Inc | ANTIBODY-DRUG CONJUGATE, PHARMACEUTICAL COMPOSITION, METHODS OF TREATMENT OF A DISEASE OR CONDITION AND A CANCER, AND, LINDER-DRUG CONJUGATE COMPOSITION |
| CA3213625A1 (en) | 2021-03-18 | 2022-09-22 | Seagen Inc. | Selective drug release from internalized conjugates of biologically active compounds |
| CA3228268A1 (en) | 2021-08-13 | 2023-02-16 | Andrew Brian Hague | Improved methods for preparing cytotoxic benzodiazepine derivatives |
| JP7707943B2 (en) * | 2022-01-25 | 2025-07-15 | 株式会社三洋物産 | Gaming Machines |
| JP7707941B2 (en) * | 2022-01-25 | 2025-07-15 | 株式会社三洋物産 | Gaming Machines |
| JP7707942B2 (en) * | 2022-01-25 | 2025-07-15 | 株式会社三洋物産 | Gaming Machines |
| JP7707940B2 (en) * | 2022-01-25 | 2025-07-15 | 株式会社三洋物産 | Gaming Machines |
| JP2025517476A (en) | 2022-05-25 | 2025-06-05 | イナート・ファルマ・ソシエテ・アノニム | Nectin-4 binding agent |
| EP4548932A1 (en) | 2022-06-30 | 2025-05-07 | Toray Industries, Inc. | Pharmaceutical composition for treating and/or preventing cancer |
| JP2025524628A (en) | 2022-07-12 | 2025-07-30 | シートムエックス セラピューティクス,インコーポレイテッド | EPCAM immunoconjugates and uses thereof |
| TW202409080A (en) * | 2022-07-26 | 2024-03-01 | 美商沙塔克實驗室股份有限公司 | Combination therapy for treatment of ovarian cancer |
| WO2024076973A1 (en) * | 2022-10-04 | 2024-04-11 | Brown University | Compositions and methods of delivering large proteins |
| WO2024129756A1 (en) | 2022-12-13 | 2024-06-20 | Seagen Inc. | Site-specific engineered cysteine antibody drug conjugates |
| WO2025149947A1 (en) | 2024-01-12 | 2025-07-17 | Seagen Inc. | Antibody-drug conjugates |
Citations (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3763183A (en) | 1972-07-10 | 1973-10-02 | Sterling Drug Inc | 1,2,3,10,11,11a-hexahydro-5h-pyrrolo (2,1-c)(1,4)benzodiazepines |
| US3860600A (en) | 1972-07-10 | 1975-01-14 | Sterling Drug Inc | Octahydropyrrido{8 2,1-c{9 {8 1,4{9 benzodiazepines |
| DE2435382A1 (en) | 1973-07-26 | 1975-02-13 | Squibb & Sons Inc | PYRIMIDOBENZOTHIADIAZEPINE AND PYRIMIDOTHIADIAZOCINE AND THEIR DIOXIDES, METHOD FOR THEIR MANUFACTURING AND MEDICINAL PRODUCTS |
| JPS57131791A (en) | 1980-12-31 | 1982-08-14 | Fujisawa Pharmaceut Co Ltd | Benzodiazepine derivative and its preparation |
| EP0219292A2 (en) | 1985-10-07 | 1987-04-22 | McNeilab, Inc. | Use of indolobenzodiazepines for antagonizing luteinizing hormone releasing hormone |
| WO1993018045A1 (en) | 1992-03-09 | 1993-09-16 | Cancer Research Campaign Technology Limited | Anti-cancer pyrrolobenzodiazepine derivatives |
| WO2000012507A2 (en) | 1998-08-27 | 2000-03-09 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2000012508A2 (en) | 1998-08-27 | 2000-03-09 | Spirogen Limited | Pyrrolbenzodiazepines |
| CN1452492A (en) | 2000-05-26 | 2003-10-29 | 奥索-麦克尼尔药品公司 | Neuroprotective peptides |
| WO2004087716A1 (en) | 2003-03-31 | 2004-10-14 | Council Of Scientific And Industrial Research | Pyrrolo[(2,1-c)(1,4) benzodiazepines dimers as antitumour agents and process thereof |
| WO2004087717A1 (en) | 2003-03-31 | 2004-10-14 | Council Of Scientific And Industrial Research | Non-cross-linking pyrrolo[2,1-c][1,4]benzodiazepines as potential antitumour agents and process thereof |
| WO2005040170A2 (en) | 2003-10-22 | 2005-05-06 | Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Pyrrolobenzodiazepine derivatives, compositions comprising the same and methods related thereto |
| WO2005085250A1 (en) | 2004-03-01 | 2005-09-15 | Spirogen Limited | C8, c8' linked 5-oxo-1,2,3,11a-tetrahydro-5h-pyrrolo[2,1-c][1,4]benzodiazepine dimers with 1h-pyrrole-dicarboxylic acid amide linkers and oligomeric analogs therof as well as related compounds for the treatment of proliferative diseases |
| WO2005110423A2 (en) | 2004-05-13 | 2005-11-24 | Spirogen Limited | Pyrrolobenzodiazepine therapeutic agents useful in the treatment of leukaemias |
| RU2005133443A (en) | 2003-03-31 | 2006-04-27 | Каунсил Оф Сайентифик Энд Индастриал Рисерч (In) | NOT crosslinked pyrrolo [2.1-C] [1.4] ENZODIAZEPINES AS POTENTIAL ANTITUMOR AGENTS AND METHOD FOR PRODUCING THEM |
| RU2005133442A (en) | 2003-03-31 | 2006-05-10 | Каунсил Оф Сайентифик Энд Индастриал Рисерч (In) | PYRROLO DIMERS [2, 1-C] [1, 4] BENZODIAZEPINE AS ANTITUMOR AGENTS AND METHOD FOR PRODUCING THEM |
| WO2006078809A2 (en) | 2005-01-21 | 2006-07-27 | Immunogen, Inc. | Method for the preparation of maytansinoid esters |
| WO2007021674A2 (en) | 2005-08-09 | 2007-02-22 | Millennium Pharmaceuticals, Inc. | Method of acylating maytansinol with chiral amino acids |
| WO2007039752A1 (en) | 2005-10-05 | 2007-04-12 | Spirogen Limited | Alkyl 4- [4- (5-oxo-2, 3, 5, 11a-tetrahyd0-5h-pyrr0l0 [2, 1-c] [1, 4] benzodiazepine-8-yloxy) -butyrylamino]-1h-pyrrole-2-carboxylate derivatives and related compounds for the treatment of a proliferative disease |
| WO2007085930A1 (en) | 2006-01-25 | 2007-08-02 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| WO2007093873A1 (en) | 2006-02-13 | 2007-08-23 | Council Of Scientific And Industrial Research | Bis-pyrr0l0[2,l-c] [1, 4] benzodiazepine- anthraquinone conjugates as antitumour agents and a process for the preparation thereof |
| WO2007144709A2 (en) | 2006-06-09 | 2007-12-21 | Sanofi-Aventis | Leptomycin derivatives |
| EP2019104A1 (en) | 2007-07-19 | 2009-01-28 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| WO2009016647A1 (en) | 2007-08-01 | 2009-02-05 | Council Of Scientific & Industrial Research | Pyrrolo [2,1-c][1, 4] benzodiazepine-glycoside prodrugs useful as a selective anti tumor agent |
| CN101693742A (en) | 2002-06-14 | 2010-04-14 | 伊缪诺金公司 | anti-igf-i receptor antibody |
| WO2010043880A1 (en) | 2008-10-17 | 2010-04-22 | Spirogen Limited | Unsymmetrical pyrrolobenzodiazepine-dimers for treatment of proliferative diseases |
| WO2010091150A1 (en) | 2009-02-05 | 2010-08-12 | Immunogen, Inc. | Novel benzodiazepine derivatives |
| WO2010122038A1 (en) | 2009-04-24 | 2010-10-28 | F. Hoffmann-La Roche Ag | Inhibitors of bruton's tyrosine kinase |
| WO2010126551A1 (en) | 2009-04-30 | 2010-11-04 | Immunogen, Inc. | Potent conjugates and hydrophilic linkers |
| WO2011106528A1 (en) | 2010-02-24 | 2011-09-01 | Immunogen, Inc. | Folate receptor 1 antibodies and immunoconjugates and uses thereof |
| US20110256157A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines and conjugates thereof |
| WO2011130616A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines used to treat proliferative diseases |
| WO2011130613A1 (en) | 2010-04-15 | 2011-10-20 | Seattle Genetics, Inc. | Targeted pyrrolobenzodiazapine conjugates |
| WO2012074757A1 (en) | 2010-11-17 | 2012-06-07 | Genentech, Inc. | Alaninyl maytansinol antibody conjugates |
| WO2012112687A1 (en) | 2011-02-15 | 2012-08-23 | Immunogen, Inc. | Methods of preparation of conjugates |
| WO2014031566A1 (en) | 2012-08-22 | 2014-02-27 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
Family Cites Families (92)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SU11248A1 (en) | 1927-03-29 | 1929-09-30 | В.С. Григорьев | Anthracene cleaning method |
| US3453266A (en) | 1966-03-14 | 1969-07-01 | American Home Prod | 1,2,5-benzothiadiazepine 1,1-dioxides |
| US3506646A (en) | 1966-06-27 | 1970-04-14 | American Home Prod | Process for the preparation of 7h-pyrido (1,2-b)(1,2,5)benzothiadiazepine 5,5 - dioxides and pyrrolo(1,2-b)(1,2,5)benzothiadiazepine 5,5-dioxides |
| US4003905A (en) | 1974-12-11 | 1977-01-18 | E. R. Squibb & Sons, Inc. | Diels-alder adducts of benzdiazepines |
| US4342771A (en) | 1981-01-02 | 1982-08-03 | Pfizer Inc. | Hypoglycemic 5-substituted oxazolidine-2,4-diones |
| US4444688A (en) | 1981-05-11 | 1984-04-24 | Ciba-Geigy Corporation | Imidazobenzothiadiazepines |
| JP2920385B2 (en) | 1988-08-18 | 1999-07-19 | 武田薬品工業株式会社 | 1,2,5-benzothiadiazepine derivatives, their production and use |
| GB8827305D0 (en) | 1988-11-23 | 1988-12-29 | British Bio Technology | Compounds |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| GB9015198D0 (en) | 1990-07-10 | 1990-08-29 | Brien Caroline J O | Binding substance |
| PT1024191E (en) | 1991-12-02 | 2008-12-22 | Medical Res Council | Production of anti-self antibodies from antibody segment repertoires and displayed on phage |
| EP0563475B1 (en) | 1992-03-25 | 2000-05-31 | Immunogen Inc | Cell binding agent conjugates of derivatives of CC-1065 |
| US5639641A (en) | 1992-09-09 | 1997-06-17 | Immunogen Inc. | Resurfacing of rodent antibodies |
| US5455258A (en) | 1993-01-06 | 1995-10-03 | Ciba-Geigy Corporation | Arylsulfonamido-substituted hydroxamic acids |
| JPH08176070A (en) | 1994-12-19 | 1996-07-09 | Mitsubishi Chem Corp | Didepside derivative and PI3 kinase inhibitor |
| JPH08175990A (en) | 1994-12-19 | 1996-07-09 | Mitsubishi Chem Corp | PI3 kinase inhibitor and method for producing the same |
| US5863949A (en) | 1995-03-08 | 1999-01-26 | Pfizer Inc | Arylsulfonylamino hydroxamic acid derivatives |
| ES2153031T4 (en) | 1995-04-20 | 2001-05-16 | Pfizer | ARILSULFONIL HYDROXAMIC ACID DERIVATIVES AS INHIBITORS OF MMP AND TNF. |
| GB9521987D0 (en) | 1995-10-26 | 1996-01-03 | Ludwig Inst Cancer Res | Phosphoinositide 3-kinase modulators |
| GB9624482D0 (en) | 1995-12-18 | 1997-01-15 | Zeneca Phaema S A | Chemical compounds |
| ATE225343T1 (en) | 1995-12-20 | 2002-10-15 | Hoffmann La Roche | MATRIX METALLOPROTEASE INHIBITORS |
| DE69720965T2 (en) | 1996-02-13 | 2004-02-05 | Astrazeneca Ab | CHINAZOLE DERIVATIVES AND THEIR USE AS VEGF INHIBITORS |
| NZ331191A (en) | 1996-03-05 | 2000-03-27 | Zeneca Ltd | 4-anilinoquinazoline derivatives and pharmaceutical compositions thereof |
| EP0818442A3 (en) | 1996-07-12 | 1998-12-30 | Pfizer Inc. | Cyclic sulphone derivatives as inhibitors of metalloproteinases and of the production of tumour necrosis factor |
| KR20000067904A (en) | 1996-07-18 | 2000-11-25 | 디. 제이. 우드, 스피겔 알렌 제이 | Phosphinate Based Inhibitors of Matrix Metalloproteases |
| KR20000068248A (en) | 1996-08-23 | 2000-11-25 | 디. 제이. 우드, 스피겔 알렌 제이 | Arylsulfonylamino Hydroxamic Acid Derivatives |
| GB9718972D0 (en) | 1996-09-25 | 1997-11-12 | Zeneca Ltd | Chemical compounds |
| JP3338064B2 (en) | 1997-01-06 | 2002-10-28 | ファイザー・インク | Cyclic sulfone derivative |
| NZ336840A (en) | 1997-02-03 | 2001-01-26 | Pfizer Prod Inc | Arylsulfonylamino hydroxamic acid derivatives useful in the treatment of tumor necrosis factor and matrix metalloproteinase mediated diseases |
| JP2000507975A (en) | 1997-02-07 | 2000-06-27 | ファイザー・インク | N-hydroxy-β-sulfonylpropionamide derivatives and their use as matrix metalloproteinase inhibitors |
| EP0960098A1 (en) | 1997-02-11 | 1999-12-01 | Pfizer Inc. | Arylsulfonyl hydroxamic acid derivatives |
| CA2280930C (en) | 1997-02-12 | 2010-04-06 | The Regents Of The University Of Michigan | Protein markers for lung cancer and use thereof |
| WO1999006587A2 (en) | 1997-08-01 | 1999-02-11 | Morphosys Ag | Novel method and phage for the identification of nucleic acid sequences encoding members of a multimeric (poly)peptide complex |
| CA2299355C (en) | 1997-08-08 | 2005-09-27 | Pfizer Products Inc. | Aryloxyarylsulfonylamino hydroxamic acid derivatives |
| GB9725782D0 (en) | 1997-12-05 | 1998-02-04 | Pfizer Ltd | Therapeutic agents |
| GB9801690D0 (en) | 1998-01-27 | 1998-03-25 | Pfizer Ltd | Therapeutic agents |
| JP4462654B2 (en) | 1998-03-26 | 2010-05-12 | ソニー株式会社 | Video material selection device and video material selection method |
| PA8469501A1 (en) | 1998-04-10 | 2000-09-29 | Pfizer Prod Inc | HYDROXAMIDES OF THE ACID (4-ARILSULFONILAMINO) -TETRAHIDROPIRAN-4-CARBOXILICO |
| PA8469401A1 (en) | 1998-04-10 | 2000-05-24 | Pfizer Prod Inc | BICYCLE DERIVATIVES OF HYDROXAMIC ACID |
| US6156746A (en) | 1998-08-25 | 2000-12-05 | Bristol-Myers Squibb Company | 1,2,5-benzothiadiazepine-1,1-dioxides with n-2 imidazolylalkyl substituents |
| EP1004578B1 (en) | 1998-11-05 | 2004-02-25 | Pfizer Products Inc. | 5-oxo-pyrrolidine-2-carboxylic acid hydroxamide derivatives |
| US7115396B2 (en) | 1998-12-10 | 2006-10-03 | Compound Therapeutics, Inc. | Protein scaffolds for antibody mimics and other binding proteins |
| PT1154774E (en) | 1999-02-10 | 2005-10-31 | Astrazeneca Ab | QUINAZOLINE DERIVATIVES AS ANGIOGENESE INHIBITORS |
| FR2800507B1 (en) | 1999-10-27 | 2002-01-04 | Valeo Systemes Dessuyage | ROTARY ELECTRIC SWITCH, PARTICULARLY FOR A MOTOR VEHICLE WINDSCREEN WIPER MOTOR |
| JP3287409B2 (en) | 1999-10-28 | 2002-06-04 | 日本精工株式会社 | Seat belt device with seat belt retractor with locking mechanism |
| KR100881105B1 (en) | 1999-11-05 | 2009-02-02 | 아스트라제네카 아베 | Quinazolin Derivatives as VEGF Inhibitors |
| CA2388063C (en) | 1999-11-24 | 2010-06-08 | Immunogen, Inc. | Cytotoxic agents comprising taxanes and their therapeutic use |
| JP3663382B2 (en) | 2000-02-15 | 2005-06-22 | スージェン・インコーポレーテッド | Pyrrole-substituted 2-indolinone protein kinase inhibitor |
| JP2001247477A (en) | 2000-03-03 | 2001-09-11 | Teikoku Hormone Mfg Co Ltd | Antitumor agent |
| US6608053B2 (en) | 2000-04-27 | 2003-08-19 | Yamanouchi Pharmaceutical Co., Ltd. | Fused heteroaryl derivatives |
| US6403588B1 (en) | 2000-04-27 | 2002-06-11 | Yamanouchi Pharmaceutical Co., Ltd. | Imidazopyridine derivatives |
| EP1332209B1 (en) | 2000-09-08 | 2009-11-11 | Universität Zürich | Collections of repeat proteins comprising repeat modules |
| DE60203260T2 (en) | 2001-01-16 | 2006-02-02 | Glaxo Group Ltd., Greenford | PHARMACEUTICAL COMBINATION CONTAINING A 4-CHINA-ZININAMINE AND PACLITAXEL, CARBOPLATIN OR VINORELBINE FOR THE TREATMENT OF CANCER |
| US6441163B1 (en) | 2001-05-31 | 2002-08-27 | Immunogen, Inc. | Methods for preparation of cytotoxic conjugates of maytansinoids and cell binding agents |
| CA2454976C (en) | 2001-07-26 | 2011-05-10 | Santen Pharmaceutical Co., Ltd. | Therapeutic agent for glaucoma comprising compound having pi3 kinase inhibitory action as active ingredient |
| US6703414B2 (en) | 2001-09-14 | 2004-03-09 | Arizona Board Of Regents On Behalf Of The University Of Arizona | Device and method for treating restenosis |
| AU2002357667A1 (en) | 2001-10-24 | 2003-05-06 | Iconix Pharmaceuticals, Inc. | Modulators of phosphoinositide 3-kinase |
| WO2003035618A2 (en) | 2001-10-24 | 2003-05-01 | Iconix Pharmaceuticals, Inc. | Modulators of phosphoinositide 3-kinase |
| JP2005509645A (en) | 2001-10-30 | 2005-04-14 | ファルマシア・コーポレーション | Heteroaromatic carboxamide derivatives for the treatment of inflammation |
| US6716821B2 (en) | 2001-12-21 | 2004-04-06 | Immunogen Inc. | Cytotoxic agents bearing a reactive polyethylene glycol moiety, cytotoxic conjugates comprising polyethylene glycol linking groups, and methods of making and using the same |
| US6756397B2 (en) | 2002-04-05 | 2004-06-29 | Immunogen, Inc. | Prodrugs of CC-1065 analogs |
| GB0209467D0 (en) | 2002-04-25 | 2002-06-05 | Astrazeneca Ab | Chemical compounds |
| US20040092561A1 (en) | 2002-11-07 | 2004-05-13 | Thomas Ruckle | Azolidinone-vinyl fused -benzene derivatives |
| JP4782564B2 (en) | 2002-07-10 | 2011-09-28 | メルク セローノ ソシエテ アノニム | Azolidinone-vinyl condensation-benzene derivative |
| AU2003255529B2 (en) | 2002-07-10 | 2008-11-20 | Laboratoires Serono Sa | Use of compounds for increasing spermatozoa motility |
| BR0313197A (en) | 2002-08-02 | 2005-08-09 | Immunogen Inc | Cytotoxic agents containing potent taxanes and their therapeutic use |
| DE60336149D1 (en) | 2002-08-16 | 2011-04-07 | Immunogen Inc | Highly reactive and soluble crosslinkers and their use in the preparation of conjugates for the targeted delivery of small-molecule drugs |
| AU2003255845A1 (en) | 2002-08-22 | 2004-03-11 | Piramed Limited | Phosphadidylinositol 3,5-biphosphate inhibitors as anti-viral agents |
| ZA200503075B (en) | 2002-11-07 | 2006-09-27 | Immunogen Inc | Anti-CD33 antibodies and method for treatment of acute myeloid leukemia using the same |
| FR2850654A1 (en) | 2003-02-03 | 2004-08-06 | Servier Lab | NOVEL TRICYCLIC AZEPINE DERIVATIVES, PROCESS FOR PREPARING THEM AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM |
| US8088387B2 (en) | 2003-10-10 | 2012-01-03 | Immunogen Inc. | Method of targeting specific cell populations using cell-binding agent maytansinoid conjugates linked via a non-cleavable linker, said conjugates, and methods of making said conjugates |
| US7276497B2 (en) | 2003-05-20 | 2007-10-02 | Immunogen Inc. | Cytotoxic agents comprising new maytansinoids |
| WO2005085260A1 (en) | 2004-03-09 | 2005-09-15 | Spirogen Limited | Pyrrolobenzodiazepines |
| NZ579482A (en) | 2004-06-01 | 2011-02-25 | Genentech Inc | Antibody drug conjugates and methods |
| DK1791565T3 (en) | 2004-09-23 | 2016-08-01 | Genentech Inc | Cysteingensplejsede antibodies and conjugates |
| GB0423653D0 (en) | 2004-10-25 | 2004-11-24 | Piramed Ltd | Pharmaceutical compounds |
| JP2008521828A (en) | 2004-11-29 | 2008-06-26 | シアトル ジェネティックス, インコーポレイテッド | Engineered antibodies and immunoconjugates |
| ES2503719T3 (en) | 2005-02-11 | 2014-10-07 | Immunogen, Inc. | Procedure for preparing antibody and maitansinoid conjugates |
| BRPI0613593A2 (en) | 2005-07-08 | 2011-01-18 | Univ Zuerich | filamentous phage display test method, phage or phagemid vector and phage or phagemid vector library |
| US7772485B2 (en) | 2005-07-14 | 2010-08-10 | Konarka Technologies, Inc. | Polymers with low band gaps and high charge mobility |
| ITRM20050416A1 (en) | 2005-08-03 | 2007-02-04 | Uni Degli Studi Di Roma Tor Vergata | BENZODIAZEPINE DERIVATIVES AND THEIR USE IN MEDICAL FIELD. |
| CA3000520C (en) | 2005-08-24 | 2023-04-04 | Immunogen, Inc. | Process for preparing antibody maytansinoid conjugates |
| GB0520657D0 (en) | 2005-10-11 | 2005-11-16 | Ludwig Inst Cancer Res | Pharmaceutical compounds |
| EP2032606B1 (en) | 2006-05-30 | 2013-11-27 | Genentech, Inc. | Antibodies and immunoconjugates and uses therefor |
| CL2008001334A1 (en) | 2007-05-08 | 2008-09-22 | Genentech Inc | ANTI-MUC16 ANTIBODY DESIGNED WITH CISTEINE; CONJUGADO THAT UNDERSTANDS IT; METHOD OF PRODUCTION; PHARMACEUTICAL FORMULATION THAT UNDERSTANDS IT; AND ITS USE TO TREAT CANCER. |
| US20100003641A1 (en) | 2007-08-15 | 2010-01-07 | Hansen John P | Dental crown system |
| US9365629B2 (en) | 2007-09-24 | 2016-06-14 | University Of Zurich | Designed armadillo repeat proteins |
| SG189817A1 (en) | 2008-04-30 | 2013-05-31 | Immunogen Inc | Potent conjugates and hydrophilic linkers |
| NZ610239A (en) | 2008-04-30 | 2014-11-28 | Immunogen Inc | Cross-linkers and their uses |
| BRPI1008532B1 (en) | 2009-02-12 | 2021-12-14 | Janssen Biotech, Inc | ISOLATED PROTEIN FRAMEWORK, METHOD FOR BUILDING AN ISOLATED PROTEIN FRAMEWORK LIBRARY, ISOLATED NUCLEIC ACID MOLECULE, ISOLATED NUCLEIC ACID VECTOR, BACTERIAL OR FUNGUS HOST CELL, COMPOSITION, MEDICAL DEVICE AND MANUFACTURING ARTICLE FOR PRODUCT USE IN HUMAN BEINGS |
| WO2010134437A1 (en) | 2009-05-18 | 2010-11-25 | Nishiyama Shuhei | Meta-information sharing distributed database system in virtual single memory storage |
| JP2015202619A (en) * | 2014-04-14 | 2015-11-16 | 独立行政法人 国立印刷局 | Intaglio |
-
2012
- 2012-02-15 JP JP2013554574A patent/JP5826863B2/en not_active Expired - Fee Related
- 2012-02-15 RS RS20190394A patent/RS58620B1/en unknown
- 2012-02-15 RS RS20160186A patent/RS54667B1/en unknown
- 2012-02-15 AU AU2012217719A patent/AU2012217719B2/en not_active Ceased
- 2012-02-15 CA CA3110544A patent/CA3110544A1/en not_active Abandoned
- 2012-02-15 EP EP18213740.6A patent/EP3498303A1/en not_active Withdrawn
- 2012-02-15 TW TW101104846A patent/TWI617555B/en not_active IP Right Cessation
- 2012-02-15 RU RU2013141829A patent/RU2631498C2/en active
- 2012-02-15 MX MX2017004016A patent/MX384514B/en unknown
- 2012-02-15 SM SM20190181T patent/SMT201900181T1/en unknown
- 2012-02-15 KR KR1020197018885A patent/KR20190089048A/en not_active Ceased
- 2012-02-15 EP EP16150802.3A patent/EP3053600B1/en active Active
- 2012-02-15 KR KR1020207035392A patent/KR20200141534A/en not_active Ceased
- 2012-02-15 BR BR112013019913-0A patent/BR112013019913B1/en not_active IP Right Cessation
- 2012-02-15 KR KR1020207035369A patent/KR20200140939A/en not_active Ceased
- 2012-02-15 LT LTEP12705761.0T patent/LT2675480T/en unknown
- 2012-02-15 CN CN202110553208.6A patent/CN113896731A/en active Pending
- 2012-02-15 WO PCT/US2012/025257 patent/WO2012112687A1/en not_active Ceased
- 2012-02-15 HR HRP20160164TT patent/HRP20160164T1/en unknown
- 2012-02-15 KR KR1020197012764A patent/KR20190051081A/en not_active Ceased
- 2012-02-15 PT PT12705761T patent/PT2675480T/en unknown
- 2012-02-15 BR BR112013020540-7A patent/BR112013020540B1/en not_active IP Right Cessation
- 2012-02-15 SG SG10202007640VA patent/SG10202007640VA/en unknown
- 2012-02-15 ME MEP-2016-59A patent/ME02381B/en unknown
- 2012-02-15 EP EP12705600.0A patent/EP2675479B1/en active Active
- 2012-02-15 UA UAA201310945A patent/UA120696C2/en unknown
- 2012-02-15 AU AU2012231640A patent/AU2012231640B2/en not_active Ceased
- 2012-02-15 SG SG2013053632A patent/SG191955A1/en unknown
- 2012-02-15 IL IL279304A patent/IL279304B/en unknown
- 2012-02-15 RU RU2017131902A patent/RU2748733C2/en active
- 2012-02-15 EP EP12707186.8A patent/EP2675481A1/en not_active Withdrawn
- 2012-02-15 WO PCT/US2012/025252 patent/WO2012128868A1/en not_active Ceased
- 2012-02-15 IL IL293365A patent/IL293365A/en unknown
- 2012-02-15 KR KR1020137023868A patent/KR20140010067A/en not_active Ceased
- 2012-02-15 EP EP20153273.6A patent/EP3666289A1/en not_active Withdrawn
- 2012-02-15 SG SG2013053780A patent/SG191965A1/en unknown
- 2012-02-15 MY MYPI2013002959A patent/MY169448A/en unknown
- 2012-02-15 DK DK12705600.0T patent/DK2675479T3/en active
- 2012-02-15 ES ES12705761T patent/ES2717657T3/en active Active
- 2012-02-15 MX MX2013009417A patent/MX343337B/en active IP Right Grant
- 2012-02-15 RU RU2017117634A patent/RU2017117634A/en unknown
- 2012-02-15 EP EP12705761.0A patent/EP2675480B1/en active Active
- 2012-02-15 KR KR1020137024312A patent/KR20140010076A/en not_active Ceased
- 2012-02-15 US US13/397,195 patent/US8765740B2/en active Active
- 2012-02-15 SI SI201230460T patent/SI2675479T1/en unknown
- 2012-02-15 HU HUE12705600A patent/HUE028736T2/en unknown
- 2012-02-15 CA CA3034596A patent/CA3034596C/en active Active
- 2012-02-15 US US13/984,762 patent/US9534000B2/en not_active Expired - Fee Related
- 2012-02-15 SG SG10201601046QA patent/SG10201601046QA/en unknown
- 2012-02-15 CN CN201280014480.6A patent/CN103687623B/en not_active Expired - Fee Related
- 2012-02-15 CA CA2824864A patent/CA2824864C/en active Active
- 2012-02-15 MX MX2013009420A patent/MX346635B/en active IP Right Grant
- 2012-02-15 CN CN201280018499.8A patent/CN103702686B/en not_active Expired - Fee Related
- 2012-02-15 WO PCT/US2012/025292 patent/WO2012112708A1/en not_active Ceased
- 2012-02-15 PL PL12705600.0T patent/PL2675479T3/en unknown
- 2012-02-15 JP JP2013554575A patent/JP6049642B2/en not_active Expired - Fee Related
- 2012-02-15 CA CA2825919A patent/CA2825919C/en active Active
- 2012-02-15 PL PL12705761T patent/PL2675480T3/en unknown
- 2012-02-15 RU RU2013142179A patent/RU2621035C2/en active
- 2012-02-15 CN CN201710188948.8A patent/CN107335061B/en not_active Expired - Fee Related
- 2012-02-15 US US13/397,205 patent/US9353127B2/en active Active
- 2012-02-15 DK DK12705761.0T patent/DK2675480T3/en active
- 2012-02-15 HU HUE12705761A patent/HUE043976T2/en unknown
- 2012-02-15 MY MYPI2013002960A patent/MY183977A/en unknown
- 2012-02-15 SI SI201231578T patent/SI2675480T1/en unknown
- 2012-02-15 ES ES12705600.0T patent/ES2567418T3/en active Active
- 2012-02-15 CN CN201610950438.5A patent/CN107050468A/en active Pending
- 2012-02-15 HR HRP20190576TT patent/HRP20190576T1/en unknown
-
2013
- 2013-03-14 US US13/827,355 patent/US8889669B2/en active Active
- 2013-07-17 ZA ZA2013/05386A patent/ZA201305386B/en unknown
- 2013-08-14 IL IL227963A patent/IL227963A/en active IP Right Grant
- 2013-08-14 IL IL227964A patent/IL227964A/en active IP Right Grant
-
2014
- 2014-10-10 US US14/512,059 patent/US9169272B2/en active Active
-
2015
- 2015-09-09 US US14/849,270 patent/US9434748B2/en not_active Expired - Fee Related
- 2015-10-14 JP JP2015202619A patent/JP6216356B2/en not_active Expired - Fee Related
-
2016
- 2016-02-05 AU AU2016200752A patent/AU2016200752B2/en not_active Ceased
- 2016-03-24 CY CY20161100252T patent/CY1117340T1/en unknown
- 2016-04-08 SM SM201600099T patent/SMT201600099B/en unknown
- 2016-04-13 US US15/097,979 patent/US9868791B2/en not_active Expired - Fee Related
- 2016-07-27 US US15/221,255 patent/US9840564B2/en active Active
-
2017
- 2017-07-10 US US15/645,597 patent/US10179818B2/en active Active
- 2017-07-14 ZA ZA2017/04808A patent/ZA201704808B/en unknown
- 2017-08-15 US US15/677,846 patent/US10364294B2/en not_active Expired - Fee Related
- 2017-08-17 IL IL254055A patent/IL254055A0/en active IP Right Grant
- 2017-08-23 IL IL254128A patent/IL254128A0/en unknown
- 2017-09-22 JP JP2017182007A patent/JP6526138B2/en not_active Expired - Fee Related
- 2017-11-28 AU AU2017268554A patent/AU2017268554B2/en not_active Ceased
-
2018
- 2018-11-30 US US16/205,609 patent/US10570212B2/en not_active Ceased
-
2019
- 2019-03-22 CY CY20191100338T patent/CY1121532T1/en unknown
- 2019-04-24 JP JP2019082629A patent/JP6703632B2/en not_active Expired - Fee Related
- 2019-11-13 AU AU2019264595A patent/AU2019264595A1/en not_active Abandoned
-
2020
- 2020-05-08 JP JP2020082654A patent/JP2020143102A/en active Pending
-
2021
- 2021-05-10 US US17/316,241 patent/USRE49918E1/en active Active
- 2021-10-13 JP JP2021167884A patent/JP2022009073A/en active Pending
-
2023
- 2023-11-29 JP JP2023201247A patent/JP2024026207A/en active Pending
Patent Citations (74)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3763183A (en) | 1972-07-10 | 1973-10-02 | Sterling Drug Inc | 1,2,3,10,11,11a-hexahydro-5h-pyrrolo (2,1-c)(1,4)benzodiazepines |
| US3860600A (en) | 1972-07-10 | 1975-01-14 | Sterling Drug Inc | Octahydropyrrido{8 2,1-c{9 {8 1,4{9 benzodiazepines |
| DE2435382A1 (en) | 1973-07-26 | 1975-02-13 | Squibb & Sons Inc | PYRIMIDOBENZOTHIADIAZEPINE AND PYRIMIDOTHIADIAZOCINE AND THEIR DIOXIDES, METHOD FOR THEIR MANUFACTURING AND MEDICINAL PRODUCTS |
| US3875162A (en) | 1973-07-26 | 1975-04-01 | Squibb & Sons Inc | Certain 6H-pyrimido{8 1,2-c{9 {8 1,3,5{9 benzothiadiaza compounds |
| JPS57131791A (en) | 1980-12-31 | 1982-08-14 | Fujisawa Pharmaceut Co Ltd | Benzodiazepine derivative and its preparation |
| EP0219292A2 (en) | 1985-10-07 | 1987-04-22 | McNeilab, Inc. | Use of indolobenzodiazepines for antagonizing luteinizing hormone releasing hormone |
| WO1993018045A1 (en) | 1992-03-09 | 1993-09-16 | Cancer Research Campaign Technology Limited | Anti-cancer pyrrolobenzodiazepine derivatives |
| WO2000012507A2 (en) | 1998-08-27 | 2000-03-09 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2000012508A2 (en) | 1998-08-27 | 2000-03-09 | Spirogen Limited | Pyrrolbenzodiazepines |
| US6562806B1 (en) | 1998-08-27 | 2003-05-13 | Spirogen Limited | Pyrrolobenzodiazepines |
| US20030120069A1 (en) | 1998-08-27 | 2003-06-26 | Spirogen Limited | Pyrrolobenzodiazepines |
| CN1452492A (en) | 2000-05-26 | 2003-10-29 | 奥索-麦克尼尔药品公司 | Neuroprotective peptides |
| CN101693742A (en) | 2002-06-14 | 2010-04-14 | 伊缪诺金公司 | anti-igf-i receptor antibody |
| WO2004087716A1 (en) | 2003-03-31 | 2004-10-14 | Council Of Scientific And Industrial Research | Pyrrolo[(2,1-c)(1,4) benzodiazepines dimers as antitumour agents and process thereof |
| RU2005133443A (en) | 2003-03-31 | 2006-04-27 | Каунсил Оф Сайентифик Энд Индастриал Рисерч (In) | NOT crosslinked pyrrolo [2.1-C] [1.4] ENZODIAZEPINES AS POTENTIAL ANTITUMOR AGENTS AND METHOD FOR PRODUCING THEM |
| RU2005133442A (en) | 2003-03-31 | 2006-05-10 | Каунсил Оф Сайентифик Энд Индастриал Рисерч (In) | PYRROLO DIMERS [2, 1-C] [1, 4] BENZODIAZEPINE AS ANTITUMOR AGENTS AND METHOD FOR PRODUCING THEM |
| WO2004087717A1 (en) | 2003-03-31 | 2004-10-14 | Council Of Scientific And Industrial Research | Non-cross-linking pyrrolo[2,1-c][1,4]benzodiazepines as potential antitumour agents and process thereof |
| WO2005040170A2 (en) | 2003-10-22 | 2005-05-06 | Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Pyrrolobenzodiazepine derivatives, compositions comprising the same and methods related thereto |
| WO2005085250A1 (en) | 2004-03-01 | 2005-09-15 | Spirogen Limited | C8, c8' linked 5-oxo-1,2,3,11a-tetrahydro-5h-pyrrolo[2,1-c][1,4]benzodiazepine dimers with 1h-pyrrole-dicarboxylic acid amide linkers and oligomeric analogs therof as well as related compounds for the treatment of proliferative diseases |
| WO2005110423A2 (en) | 2004-05-13 | 2005-11-24 | Spirogen Limited | Pyrrolobenzodiazepine therapeutic agents useful in the treatment of leukaemias |
| WO2006078809A2 (en) | 2005-01-21 | 2006-07-27 | Immunogen, Inc. | Method for the preparation of maytansinoid esters |
| WO2007021674A2 (en) | 2005-08-09 | 2007-02-22 | Millennium Pharmaceuticals, Inc. | Method of acylating maytansinol with chiral amino acids |
| WO2007039752A1 (en) | 2005-10-05 | 2007-04-12 | Spirogen Limited | Alkyl 4- [4- (5-oxo-2, 3, 5, 11a-tetrahyd0-5h-pyrr0l0 [2, 1-c] [1, 4] benzodiazepine-8-yloxy) -butyrylamino]-1h-pyrrole-2-carboxylate derivatives and related compounds for the treatment of a proliferative disease |
| WO2007085930A1 (en) | 2006-01-25 | 2007-08-02 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| US20090036431A1 (en) | 2006-01-25 | 2009-02-05 | Sanofi-Aventis | Cytotoxic Agents Comprising New Tomaymycin Derivatives |
| WO2007093873A1 (en) | 2006-02-13 | 2007-08-23 | Council Of Scientific And Industrial Research | Bis-pyrr0l0[2,l-c] [1, 4] benzodiazepine- anthraquinone conjugates as antitumour agents and a process for the preparation thereof |
| WO2007144709A2 (en) | 2006-06-09 | 2007-12-21 | Sanofi-Aventis | Leptomycin derivatives |
| KR20090018103A (en) | 2006-06-09 | 2009-02-19 | 사노피-아벤티스 | Leptomycin Derivatives |
| US20100316656A1 (en) | 2007-07-19 | 2010-12-16 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| WO2009016516A2 (en) | 2007-07-19 | 2009-02-05 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| EP2019104A1 (en) | 2007-07-19 | 2009-01-28 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| EA201070163A1 (en) | 2007-07-19 | 2010-08-30 | Санофи-Авентис | CYTOTOXIC MEANS, INCLUDING NEW TOMAYMIC DERIVATIVES, AND THEIR THERAPEUTIC APPLICATION |
| WO2009016647A1 (en) | 2007-08-01 | 2009-02-05 | Council Of Scientific & Industrial Research | Pyrrolo [2,1-c][1, 4] benzodiazepine-glycoside prodrugs useful as a selective anti tumor agent |
| WO2010043880A1 (en) | 2008-10-17 | 2010-04-22 | Spirogen Limited | Unsymmetrical pyrrolobenzodiazepine-dimers for treatment of proliferative diseases |
| US20210238301A1 (en) | 2009-02-05 | 2021-08-05 | Immunogen, Inc. | Novel benzodiazepine derivatives |
| JP6204294B2 (en) | 2009-02-05 | 2017-09-27 | イミュノジェン・インコーポレーテッド | New benzodiazepine derivatives |
| US8809320B2 (en) | 2009-02-05 | 2014-08-19 | Immunogen, Inc. | Benzodiazepine derivatives |
| WO2010091150A1 (en) | 2009-02-05 | 2010-08-12 | Immunogen, Inc. | Novel benzodiazepine derivatives |
| US10947315B2 (en) | 2009-02-05 | 2021-03-16 | Immunogen, Inc. | Benzodiazepine derivatives |
| US10208127B2 (en) | 2009-02-05 | 2019-02-19 | Immunogen, Inc. | Benzodiazepine derivatives |
| US20180079823A1 (en) | 2009-02-05 | 2018-03-22 | Immunogen, Inc. | Novel benzodiazepine derivatives |
| US8802667B2 (en) | 2009-02-05 | 2014-08-12 | Immunogen, Inc. | Benzodiazepine derivatives |
| US20170183419A1 (en) | 2009-02-05 | 2017-06-29 | Immunogen, Inc. | Novel benzodiazepine derivatives |
| US9550787B2 (en) | 2009-02-05 | 2017-01-24 | Immunogen, Inc. | Benzodiazepine derivatives |
| JP5977522B2 (en) | 2009-02-05 | 2016-08-24 | イミュノジェン・インコーポレーテッド | New benzodiazepine derivatives |
| US8426402B2 (en) | 2009-02-05 | 2013-04-23 | Immunogen, Inc. | Benzodiazepine derivatives |
| US9265841B2 (en) | 2009-02-05 | 2016-02-23 | Immunogen, Inc. | Benzodiazepine derivatives |
| WO2010122038A1 (en) | 2009-04-24 | 2010-10-28 | F. Hoffmann-La Roche Ag | Inhibitors of bruton's tyrosine kinase |
| WO2010126551A1 (en) | 2009-04-30 | 2010-11-04 | Immunogen, Inc. | Potent conjugates and hydrophilic linkers |
| WO2011106528A1 (en) | 2010-02-24 | 2011-09-01 | Immunogen, Inc. | Folate receptor 1 antibodies and immunoconjugates and uses thereof |
| US20110256157A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines and conjugates thereof |
| WO2011130616A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines used to treat proliferative diseases |
| WO2011130613A1 (en) | 2010-04-15 | 2011-10-20 | Seattle Genetics, Inc. | Targeted pyrrolobenzodiazapine conjugates |
| WO2012074757A1 (en) | 2010-11-17 | 2012-06-07 | Genentech, Inc. | Alaninyl maytansinol antibody conjugates |
| US20160108129A1 (en) | 2011-02-15 | 2016-04-21 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| US20130302359A1 (en) | 2011-02-15 | 2013-11-14 | Wei Li | Cytotoxic benzodiazepine derivatives |
| US9353127B2 (en) | 2011-02-15 | 2016-05-31 | Immunogen, Inc. | Methods of preparation of conjugates |
| WO2012128868A1 (en) | 2011-02-15 | 2012-09-27 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| US9434748B2 (en) | 2011-02-15 | 2016-09-06 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| US20160324980A1 (en) | 2011-02-15 | 2016-11-10 | Immunogen, Inc. | Methods of preparation of conjugates |
| US9534000B2 (en) | 2011-02-15 | 2017-01-03 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives and methods of preparation |
| US20120238731A1 (en) | 2011-02-15 | 2012-09-20 | Immunogen, Inc. | Methods of preparation of conjugates |
| WO2012112687A1 (en) | 2011-02-15 | 2012-08-23 | Immunogen, Inc. | Methods of preparation of conjugates |
| US8765740B2 (en) | 2011-02-15 | 2014-07-01 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| US9840564B2 (en) | 2011-02-15 | 2017-12-12 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| US9868791B2 (en) | 2011-02-15 | 2018-01-16 | Immunogen, Inc. | Methods of preparation of conjugates |
| US20180037659A1 (en) | 2011-02-15 | 2018-02-08 | Immunogen, Inc. | Methods of preparation of conjugates |
| JP5826863B2 (en) | 2011-02-15 | 2015-12-02 | イミュノジェン・インコーポレーテッド | Cytotoxic benzodiazepine derivatives |
| US10179818B2 (en) | 2011-02-15 | 2019-01-15 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| US9169272B2 (en) | 2011-02-15 | 2015-10-27 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| US10364294B2 (en) | 2011-02-15 | 2019-07-30 | Immunogen, Inc. | Methods of preparation of conjugates |
| US10570212B2 (en) | 2011-02-15 | 2020-02-25 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| US8889669B2 (en) | 2011-02-15 | 2014-11-18 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| WO2014031566A1 (en) | 2012-08-22 | 2014-02-27 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
Non-Patent Citations (27)
| Title |
|---|
| Greene et al., Protective Groups in Organic Synthesis, Second Edition. John Wiley & Sons, Inc., New York. pp. 156-157, (1991). |
| Guo et al., Synthesis and evaluation of a cyclic imine derivative conjugated to a fluorescent molecule for labeling of proteins. Bioorg Med Chem Lett. Feb. 15, 2009;19(4):1210-3. |
| Guo et al., Synthesis and evaluation of a cyclic imine derivative conjugated to a fluorescent molecule for labeling of proteins. Bioorg Med Chem Lett. Feb. 2009;19(4):1210-3. |
| Henne et al., Synthesis and activity of a folate peptide camptothecin prodrug. Bioorg Med Chem Lett. Oct. 15, 2006;16(20):5350-5. |
| Kamal et al., Conversion of Amines to Imines Employing Polymer-Supported Sulfoxide (PSS) and Polymer-Supported Perruthenate (PSP): Synthesis of Pyrrolo[2,1-c][1,4]benzodiazepines. Advanced Synthesis & Catalysis. Jan. 2006;348(1-2):249-254. |
| Kamal et al., Design, synthesis, and evaluation of mixed imineamine pyrrolobenzodiazepine dimers with efficient DNA binding affinity and potent cytotoxicity. Bioorg Med Chem. Oct. 15, 2004;12(20):5427-36. |
| Kamal et al., Development of Pyrrolo[2,1-c][1,4]benzodiazepine beta-Galactoside Prodrugs for Selective Therapy of Cancer by ADEPT and PMT. ChemMedChem. 2008;3:794-802. |
| Kamal et al., Development of pyrrolo[2,1-c][1,4]benzodiazepine beta-galactoside prodrugs for selective therapy of cancer by ADEPT and PMT. ChemMedChem. May 2008;3(5):794-802. |
| Kamal et al., Synthesis and DNA binding affinity of novel A-C8/C-C2-exo unsaturated alkoxyamido-linked pyrrolo[2,1-c][1,4]benzodiazepine dimers. Bioorg Med Chem. Aug. 15, 2004;12(16):4337-50. |
| Kamal et al., Synthesis of pyrrolo[2,1-c][1,4]benzodiazepine antibiotics: Oxidation of cyclic secondary amine with TPAP. Tetrahedron. Mar. 1997;53(9):3223-30. |
| Li et al,, "Design, synthesis and evaluation of a novel DNA-interactive agent: a promising new class of cytotoxic molecules for use in antibody-drug conjugates", 239th ACS National Meeting. Mar. 21-25, 2010, 1 page. (Year: 2010). * |
| Li et al., Design, Synthesis and Evaluation of a Novel DNA-Interactive Agent: A Promising New Class of Cytotoxic Molecules for Use in Antibody-Drug Conjugates. 239th ACS National Meeting, San Francisco, CA 2010 [MEDI 251]. |
| Li et al., Design, synthesis and evaluation of a novel DNA-interactive agent: a promising new class of cytotoxic molecules for use in antibody-drug conjugates. 239th ACS National Meeting. Mar. 21-25, 2010, 1 page. |
| Masterson et al., Synthesis and biological evaluation of novel pyrrolo[2,1-c][1,4]benzodiazepine prodrugs for use in antibody-directed enzyme prodrug therapy. Bioorg Med Chem Lett. Jan. 15, 2006;16(2):252-6. |
| Miller et al., Potent antigen-specific anti-tumor activity observed with antibody-drug conjugates (ADCs) made using a new class of DNA-crosslinking agents. Mol Cancer Ther. 2009;8(12) Suppl 1, Abstract B126. |
| Miller et al., Potent antigen-specific anti-tumor activity observed with antibody-drug conjugates (ADCs) made using a new class of DNA-crosslinking agents. Poster presentation AACR-NCI-EORTC, Abstract B126 (Nov. 2009). |
| Mori et al., Structure and Syntheses of Sen-215 and Oxotomaymycin. Hetercycles. 1986;24(5):1257-1260. |
| STN Accession No. 1983:72145, Benzodiazepine derivatives. 1 page, (2020). |
| Thurston et al., Synthesis and reactivity of a novel oxazolo[2,3-c] [1,4]benziodiazepine ring system with DNA recognition potential: a new class of anthramycins. J Chem. Soc Commun. 1990;12:874-876. |
| Thurston et al., Synthesis and reactivity of a novel oxazolo[2,3-c][1,4]benziodiazepine ring system with DNA recognition potential: a new class of anthramycins. J Chem Soc. 1990;12:874-876. |
| Tozuka et al., Studies on tomaymycin. III. Syntheses and antitumor activity of tomaymycin analogs. J Antibiot. Jun. 29, 1983;36(12):1699-1708. |
| Tozuka et al., Studies on tomaymycin. III. Synthesis and antitumor activity of tomaymycin analogs. J Antibiot (Tokyo). Dec. 1983;36(12):1699-708. |
| U.S. Appl. No. 13/826,181, filed Mar. 14, 2013, now U.S. Pat. No. 8,802,667, Issued. |
| U.S. Appl. No. 14/990,569, filed Jan. 7, 2016, now U.S. Pat. No. 9,550,787, Issued. |
| U.S. Appl. No. 15/221,255, filed Jul. 27, 2016, now U.S. Pat. No. 9,840,564, Issued. |
| U.S. Appl. No. 15/677,846, filed Aug. 15, 2017, now U.S. Pat. No. 10,364,294, Issued. |
| U.S. Appl. No. 16/205,609, filed Nov. 30, 2018, now U.S. Pat. No. 10,570,212, Issued. |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| USRE49918E1 (en) | Cytotoxic benzodiazepine derivatives | |
| AU2019206132B9 (en) | Cytotoxic benzodiazepine derivatives | |
| HK1226944A (en) | Cytotoxic benzodiazepine derivatives | |
| HK40030962A (en) | Cytotoxic benzodiazepine derivatives | |
| HK1226944A1 (en) | Cytotoxic benzodiazepine derivatives | |
| HK1186686B (en) | Cytotoxic benzodiazepine derivatives | |
| NZ613121B2 (en) | Cytotoxic benzodiazepine derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FEPP | Fee payment procedure |
Free format text: ENTITY STATUS SET TO UNDISCOUNTED (ORIGINAL EVENT CODE: BIG.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |