KR20200059773A - Method of preparing fermented product of mulberry leaves with enhanced GABA component and food composition comprising the fermented product thereby - Google Patents
Method of preparing fermented product of mulberry leaves with enhanced GABA component and food composition comprising the fermented product thereby Download PDFInfo
- Publication number
- KR20200059773A KR20200059773A KR1020180144896A KR20180144896A KR20200059773A KR 20200059773 A KR20200059773 A KR 20200059773A KR 1020180144896 A KR1020180144896 A KR 1020180144896A KR 20180144896 A KR20180144896 A KR 20180144896A KR 20200059773 A KR20200059773 A KR 20200059773A
- Authority
- KR
- South Korea
- Prior art keywords
- mulberry leaf
- gaba
- fermented
- mulberry
- fermented product
- Prior art date
Links
- 240000000249 Morus alba Species 0.000 title claims abstract description 121
- 235000008708 Morus alba Nutrition 0.000 title claims abstract description 121
- 239000000203 mixture Substances 0.000 title claims abstract description 17
- 235000013305 food Nutrition 0.000 title claims abstract description 13
- 238000000034 method Methods 0.000 title claims description 20
- BTCSSZJGUNDROE-UHFFFAOYSA-N gamma-aminobutyric acid Chemical compound NCCCC(O)=O BTCSSZJGUNDROE-UHFFFAOYSA-N 0.000 title abstract description 6
- 229960003692 gamma aminobutyric acid Drugs 0.000 title abstract description 5
- 238000004519 manufacturing process Methods 0.000 claims abstract description 25
- 239000004615 ingredient Substances 0.000 claims abstract description 7
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 claims description 74
- 238000000855 fermentation Methods 0.000 claims description 50
- 230000004151 fermentation Effects 0.000 claims description 49
- 241000894006 Bacteria Species 0.000 claims description 37
- 239000004310 lactic acid Substances 0.000 claims description 37
- 235000014655 lactic acid Nutrition 0.000 claims description 37
- 239000000284 extract Substances 0.000 claims description 30
- 240000006024 Lactobacillus plantarum Species 0.000 claims description 23
- 235000013965 Lactobacillus plantarum Nutrition 0.000 claims description 23
- 229940072205 lactobacillus plantarum Drugs 0.000 claims description 23
- 239000002609 medium Substances 0.000 claims description 14
- 235000013923 monosodium glutamate Nutrition 0.000 claims description 11
- 239000007787 solid Substances 0.000 claims description 10
- 235000013311 vegetables Nutrition 0.000 claims description 10
- 235000013373 food additive Nutrition 0.000 claims description 4
- 239000002778 food additive Substances 0.000 claims description 4
- 239000001963 growth medium Substances 0.000 claims description 4
- LPUQAYUQRXPFSQ-DFWYDOINSA-M monosodium L-glutamate Chemical compound [Na+].[O-]C(=O)[C@@H](N)CCC(O)=O LPUQAYUQRXPFSQ-DFWYDOINSA-M 0.000 claims description 4
- 239000004223 monosodium glutamate Substances 0.000 claims description 4
- 238000012258 culturing Methods 0.000 claims description 3
- 238000010298 pulverizing process Methods 0.000 claims description 3
- 235000021109 kimchi Nutrition 0.000 abstract description 11
- 244000164595 Lactobacillus plantarum subsp plantarum Species 0.000 abstract description 5
- 235000012523 Lactobacillus plantarum subsp plantarum Nutrition 0.000 abstract description 5
- 235000013402 health food Nutrition 0.000 abstract description 2
- 230000002708 enhancing effect Effects 0.000 abstract 1
- 239000000047 product Substances 0.000 description 42
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 12
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 12
- 235000015872 dietary supplement Nutrition 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- 241000196324 Embryophyta Species 0.000 description 8
- 239000003531 protein hydrolysate Substances 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 6
- 210000004556 brain Anatomy 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 5
- 241000186660 Lactobacillus Species 0.000 description 5
- 229940039696 lactobacillus Drugs 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 108090000623 proteins and genes Proteins 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 230000036541 health Effects 0.000 description 4
- 230000000813 microbial effect Effects 0.000 description 4
- 239000008213 purified water Substances 0.000 description 4
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- 108090000790 Enzymes Proteins 0.000 description 3
- 102000004190 Enzymes Human genes 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 239000005862 Whey Substances 0.000 description 3
- 102000007544 Whey Proteins Human genes 0.000 description 3
- 108010046377 Whey Proteins Proteins 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 235000013376 functional food Nutrition 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 208000024891 symptom Diseases 0.000 description 3
- 108020004465 16S ribosomal RNA Proteins 0.000 description 2
- 235000019750 Crude protein Nutrition 0.000 description 2
- SRBFZHDQGSBBOR-SOOFDHNKSA-N D-ribopyranose Chemical compound O[C@@H]1COC(O)[C@H](O)[C@@H]1O SRBFZHDQGSBBOR-SOOFDHNKSA-N 0.000 description 2
- 229920001353 Dextrin Polymers 0.000 description 2
- 239000004375 Dextrin Substances 0.000 description 2
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 2
- 240000005979 Hordeum vulgare Species 0.000 description 2
- 235000007340 Hordeum vulgare Nutrition 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 108010009736 Protein Hydrolysates Proteins 0.000 description 2
- 108010073771 Soybean Proteins Proteins 0.000 description 2
- 206010052428 Wound Diseases 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 229940024606 amino acid Drugs 0.000 description 2
- 235000001014 amino acid Nutrition 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- SRBFZHDQGSBBOR-UHFFFAOYSA-N beta-D-Pyranose-Lyxose Natural products OC1COC(O)C(O)C1O SRBFZHDQGSBBOR-UHFFFAOYSA-N 0.000 description 2
- 235000021329 brown rice Nutrition 0.000 description 2
- 229940041514 candida albicans extract Drugs 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 229960002433 cysteine Drugs 0.000 description 2
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 2
- 235000018417 cysteine Nutrition 0.000 description 2
- 235000019425 dextrin Nutrition 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 235000013922 glutamic acid Nutrition 0.000 description 2
- 239000004220 glutamic acid Substances 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 238000011081 inoculation Methods 0.000 description 2
- 229910021432 inorganic complex Inorganic materials 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- ZWLUXSQADUDCSB-UHFFFAOYSA-N phthalaldehyde Chemical compound O=CC1=CC=CC=C1C=O ZWLUXSQADUDCSB-UHFFFAOYSA-N 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 229940001941 soy protein Drugs 0.000 description 2
- 238000001694 spray drying Methods 0.000 description 2
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- 239000012138 yeast extract Substances 0.000 description 2
- OGNSCSPNOLGXSM-UHFFFAOYSA-N (+/-)-DABA Natural products NCCC(N)C(O)=O OGNSCSPNOLGXSM-UHFFFAOYSA-N 0.000 description 1
- QIJRTFXNRTXDIP-UHFFFAOYSA-N (1-carboxy-2-sulfanylethyl)azanium;chloride;hydrate Chemical compound O.Cl.SCC(N)C(O)=O QIJRTFXNRTXDIP-UHFFFAOYSA-N 0.000 description 1
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 206010048909 Boredom Diseases 0.000 description 1
- 240000007124 Brassica oleracea Species 0.000 description 1
- 235000003899 Brassica oleracea var acephala Nutrition 0.000 description 1
- 235000011301 Brassica oleracea var capitata Nutrition 0.000 description 1
- 235000001169 Brassica oleracea var oleracea Nutrition 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- 206010010774 Constipation Diseases 0.000 description 1
- 208000034656 Contusions Diseases 0.000 description 1
- RFSUNEUAIZKAJO-VRPWFDPXSA-N D-Fructose Natural products OC[C@H]1OC(O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-VRPWFDPXSA-N 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- SHZGCJCMOBCMKK-UHFFFAOYSA-N D-mannomethylose Natural products CC1OC(O)C(O)C(O)C1O SHZGCJCMOBCMKK-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 1
- SRBFZHDQGSBBOR-IOVATXLUSA-N D-xylopyranose Chemical compound O[C@@H]1COC(O)[C@H](O)[C@H]1O SRBFZHDQGSBBOR-IOVATXLUSA-N 0.000 description 1
- 206010012289 Dementia Diseases 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 241001440219 Gabara Species 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 208000008454 Hyperhidrosis Diseases 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- LKDRXBCSQODPBY-AMVSKUEXSA-N L-(-)-Sorbose Chemical compound OCC1(O)OC[C@H](O)[C@@H](O)[C@@H]1O LKDRXBCSQODPBY-AMVSKUEXSA-N 0.000 description 1
- IFQSXNOEEPCSLW-DKWTVANSSA-N L-cysteine hydrochloride Chemical compound Cl.SC[C@H](N)C(O)=O IFQSXNOEEPCSLW-DKWTVANSSA-N 0.000 description 1
- SHZGCJCMOBCMKK-JFNONXLTSA-N L-rhamnopyranose Chemical compound C[C@@H]1OC(O)[C@H](O)[C@H](O)[C@H]1O SHZGCJCMOBCMKK-JFNONXLTSA-N 0.000 description 1
- PNNNRSAQSRJVSB-UHFFFAOYSA-N L-rhamnose Natural products CC(O)C(O)C(O)C(O)C=O PNNNRSAQSRJVSB-UHFFFAOYSA-N 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- GXCLVBGFBYZDAG-UHFFFAOYSA-N N-[2-(1H-indol-3-yl)ethyl]-N-methylprop-2-en-1-amine Chemical compound CN(CCC1=CNC2=C1C=CC=C2)CC=C GXCLVBGFBYZDAG-UHFFFAOYSA-N 0.000 description 1
- 208000008589 Obesity Diseases 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 108010084695 Pea Proteins Proteins 0.000 description 1
- 235000010627 Phaseolus vulgaris Nutrition 0.000 description 1
- 244000046052 Phaseolus vulgaris Species 0.000 description 1
- 208000004880 Polyuria Diseases 0.000 description 1
- 235000014443 Pyrus communis Nutrition 0.000 description 1
- 240000001987 Pyrus communis Species 0.000 description 1
- 244000088415 Raphanus sativus Species 0.000 description 1
- 235000006140 Raphanus sativus var sativus Nutrition 0.000 description 1
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 1
- 241000270295 Serpentes Species 0.000 description 1
- 244000269722 Thea sinensis Species 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 238000010564 aerobic fermentation Methods 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 239000012491 analyte Substances 0.000 description 1
- 208000022531 anorexia Diseases 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 235000013405 beer Nutrition 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 229910021538 borax Inorganic materials 0.000 description 1
- 230000008344 brain blood flow Effects 0.000 description 1
- 210000004958 brain cell Anatomy 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229960001305 cysteine hydrochloride Drugs 0.000 description 1
- 235000013365 dairy product Nutrition 0.000 description 1
- 206010061428 decreased appetite Diseases 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- AIUDWMLXCFRVDR-UHFFFAOYSA-N dimethyl 2-(3-ethyl-3-methylpentyl)propanedioate Chemical compound CCC(C)(CC)CCC(C(=O)OC)C(=O)OC AIUDWMLXCFRVDR-UHFFFAOYSA-N 0.000 description 1
- UQGFMSUEHSUPRD-UHFFFAOYSA-N disodium;3,7-dioxido-2,4,6,8,9-pentaoxa-1,3,5,7-tetraborabicyclo[3.3.1]nonane Chemical compound [Na+].[Na+].O1B([O-])OB2OB([O-])OB1O2 UQGFMSUEHSUPRD-UHFFFAOYSA-N 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 230000035619 diuresis Effects 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 210000002615 epidermis Anatomy 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 235000021107 fermented food Nutrition 0.000 description 1
- 108010007119 flavourzyme Proteins 0.000 description 1
- 230000037406 food intake Effects 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 235000020510 functional beverage Nutrition 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 230000035784 germination Effects 0.000 description 1
- 235000009569 green tea Nutrition 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- BAUYGSIQEAFULO-UHFFFAOYSA-L iron(2+) sulfate (anhydrous) Chemical compound [Fe+2].[O-]S([O-])(=O)=O BAUYGSIQEAFULO-UHFFFAOYSA-L 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 210000002429 large intestine Anatomy 0.000 description 1
- 230000003908 liver function Effects 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- SQQMAOCOWKFBNP-UHFFFAOYSA-L manganese(II) sulfate Chemical compound [Mn+2].[O-]S([O-])(=O)=O SQQMAOCOWKFBNP-UHFFFAOYSA-L 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 230000007102 metabolic function Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000006872 mrs medium Substances 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 210000000944 nerve tissue Anatomy 0.000 description 1
- 210000000653 nervous system Anatomy 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 231100000862 numbness Toxicity 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- 235000020824 obesity Nutrition 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 235000019702 pea protein Nutrition 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- HEBKCHPVOIAQTA-ZXFHETKHSA-N ribitol Chemical compound OC[C@H](O)[C@H](O)[C@H](O)CO HEBKCHPVOIAQTA-ZXFHETKHSA-N 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 239000010802 sludge Substances 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- FQENQNTWSFEDLI-UHFFFAOYSA-J sodium diphosphate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]P([O-])(=O)OP([O-])([O-])=O FQENQNTWSFEDLI-UHFFFAOYSA-J 0.000 description 1
- 235000010339 sodium tetraborate Nutrition 0.000 description 1
- 239000004328 sodium tetraborate Substances 0.000 description 1
- UIIMBOGNXHQVGW-UHFFFAOYSA-N sodium;hydron;carbonate Chemical compound [Na+].OC(O)=O UIIMBOGNXHQVGW-UHFFFAOYSA-N 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000008223 sterile water Substances 0.000 description 1
- 230000035900 sweating Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- RYCLIXPGLDDLTM-UHFFFAOYSA-J tetrapotassium;phosphonato phosphate Chemical compound [K+].[K+].[K+].[K+].[O-]P([O-])(=O)OP([O-])([O-])=O RYCLIXPGLDDLTM-UHFFFAOYSA-J 0.000 description 1
- 230000035922 thirst Effects 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/135—Bacteria or derivatives thereof, e.g. probiotics
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/38—Other non-alcoholic beverages
- A23L2/382—Other non-alcoholic beverages fermented
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L29/00—Foods or foodstuffs containing additives; Preparation or treatment thereof
- A23L29/065—Microorganisms
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2250/00—Food ingredients
- A23V2250/20—Natural extracts
- A23V2250/21—Plant extracts
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2300/00—Processes
- A23V2300/14—Extraction
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2400/00—Lactic or propionic acid bacteria
- A23V2400/11—Lactobacillus
- A23V2400/169—Plantarum
-
- A23Y2220/67—
Abstract
Description
본 발명은 김치 유래 신규한 유산균을 이용한 가바 성분이 강화된 뽕잎의 발효방법, 이로부터 제조된 뽕잎 발효물 및 이를 포함하는 식품 조성물에 관한 것으로, 더욱 상세하게는 김치에서 분리한 락토바실러스 플란타룸 GW0709 균주를 이용하여 뽕잎을 발효시켜 가바 성분이 강화된 뽕잎 발효물의 제조방법, 이로부터 제조된 뽕잎 발효물 및 이를 포함하는 식품 조성물에 관한 것이다.The present invention relates to a fermentation method of a mulberry leaf with a reinforced gaba component using a novel lactic acid bacteria derived from kimchi, a mulberry leaf fermentation product prepared therefrom and a food composition comprising the same, and more specifically, Lactobacillus plantarum separated from kimchi. It relates to a method for producing a mulberry leaf fermentation product having enhanced Gaba ingredients by fermenting a mulberry leaf using a strain GW0709, a mulberry leaf fermentation product prepared therefrom, and a food composition comprising the same.
뽕잎에 대하여 우리나라의 전통의학서인 『동의보감(東醫寶鑑)』 에는 "뽕잎은 따뜻하고 독이 없으며, 각기(脚氣)와 수종을 없애주고 대장(大腸), 소장(小腸)을 이롭게 하며, 풍통을 없앤다."라고 기록되어 있고, 『본초강목(本草綱目)』 에는 "뽕잎은 소갈증, 풍, 멍들고 풍으로 아픈 곳, 뱀과 벌레에 물린데, 밤에 땀이 나는 증세, 청맹(靑盲), 머리가 자라지 않는 증세, 정강이가 오그라져 펴지지 않는 증세, 탈항(脫肛), 종기(腫氣)나 등창, 못에 찔린 상처, 데인 상처, 손발이 저리고 감각이 없는 증세에 효험이 있다."고 기록되어 있다. 뽕잎이 상기한 바와 같은 여러 가지 기능성을 가지고 있다는 것이 밝혀지면서 최근 이러한 뽕잎의 기능성 효과를 이용하고자 하는 다양한 노력이 시도되고 있다.About Mulberry Leaves In Korean traditional medical book "Donguibogam (東 醫 寶 鑑)", "Mulberry leaves are warm and non-toxic, removing each and every species, benefiting large intestine and small intestine, and eliminating wind pain. . ", And in" Bonchogangmok (本草綱目) "," Mulberry leaves are bitten by thirst, wind, bruises, and snakes and worms, symptoms of sweating at night, boredom, hair " It is effective for symptoms that don't grow, symptoms that the shank is not stretched, dehydration, boils or backspans, nail puncture wounds, burn wounds, hands and feet, and numbness. " have. As it was found that the mulberry leaf has various functionalities as described above, various efforts have been recently made to utilize the functional effect of the mulberry leaf.
감마아미노부틸산(gamma-aminobutyric acid, GABA, 이후 가바라 칭함.)은 자연계에 널리 분포하는 비단백질성 아미노산의 일종으로, 사람에 있어서는 신경계 및 혈액에서 발견되고, 뇌와 척추에 많이 존재하는 흥분 억제성 신경전달물질로 알려져 있다. 1950년대 상기 가바가 포유동물의 뇌신경조직에 많이 분포되어 있다는 것이 발견되면서 뇌의 신경기능에 영향을 미칠 것으로 추정되었다. 그 후에 가바가 생체내에서 글루탐산으로부터 생합성되어 생성되고, 뇌의 혈류를 활발하게 하여 뇌의 산소 공급량을 증진시킴으로서 뇌세포의 대사기능을 촉진시키는 것으로 밝혀졌다. 현재까지 가바가 뇌 혈류개선, 뇌 대사증진 작용, 중풍 및 치매의 예방, 기억력 증진 등에 효과가 있는 것으로 연구되었고, 고혈압 예방, 이뇨작용, 간 기능개선 및 비만 개선 등의 효과가 연구되면서 다양한 기능성을 바탕으로 의약품 및 건강 기능성 식품의 소재로 응용되고 있다.Gamma-aminobutyric acid (GABA, hereinafter called Gabara) is a type of non-proteinaceous amino acid that is widely distributed in nature. It is found in the nervous system and blood in humans, and has a lot of excitement in the brain and spine. It is known as an inhibitory neurotransmitter. In the 1950s, it was estimated that Gaba was widely distributed in the brain nerve tissues of mammals, and it was estimated that it affects the brain's nerve function. After that, gaba was found to be biosynthesized from glutamic acid in vivo, and it was found that it promotes the metabolic function of brain cells by increasing the oxygen supply to the brain by activating blood flow in the brain. To date, Gaba has been studied to improve brain blood flow, promote brain metabolism, prevent stroke and dementia, and improve memory, and has been studied for effects such as preventing hypertension, diuresis, improving liver function, and improving obesity. Based on this, it is applied as a material for pharmaceuticals and health functional foods.
이러한 가바의 다양한 기능성으로 인하여 의약품 또는 기능성 식품의 소재로서 가바에 대한 관심이 고조되고 있으나 일반적으로 식물체가 함유하고 있는 가바의 양이 많지 않기 때문에 순수한 가바를 회수하는 것이 매우 어려워 그 사용이 제한되고 있는 실정이다. 식물 중에서도 발아현미의 배, 녹차 또는 뽕잎의 섭취를 통하여 섭취될 수 있는 가바의 함량은 약리작용을 발휘하기에 현저히 낮아 자연적인 섭취로 가바의 생리작용을 기대하기는 어려운 실정이다. 또한, 지난 수년 동안 연구되어온 가바의 화학적 합성법에 의해 제조된 합성 가바의 경우 식욕부진, 변비 또는 설사 등의 부작용이 있었다. 이러한 문제점을 해결하기 위한 연구들의 일예로 식물 내 가바 함유량을 높이기 위해서 보리맥아의 제조시 발아된 보리의 혐기적 처리법 또는 현미 발아시 글루탐산(glutamic acid) 첨가를 통하여 식물체 내 가바 함량을 증가시키기 위한 방법이 제안되었으나, 방법이 효율적이지 않고 수율이 낮은 문제점이 있었다.Due to the various functionalities of such gaba, interest in gaba is increasing as a material for pharmaceuticals or functional foods, but in general, it is very difficult to recover pure gaba because the amount of gaba contained in plants is not very large, and its use is limited. This is true. Among plants, the content of gaba that can be consumed through the ingestion of pear, green tea or mulberry leaves of germinated brown rice is remarkably low to exert a pharmacological action, so it is difficult to expect physiological action of gaba by natural intake. In addition, synthetic gaba produced by the chemical synthesis method of gaba that has been studied for the past several years has side effects such as anorexia, constipation or diarrhea. As an example of studies to solve these problems, in order to increase the gaba content in the plant, a method for increasing the gaba content in the plant through anaerobic treatment of barley germinated during the production of barley malt or adding glutamic acid during brown rice germination Although this was proposed, there was a problem that the method was not efficient and the yield was low.
식물을 원재료로 한 가바의 생산에는 한계가 있으므로, 최근 들어 미생물을 이용한 가바의 대량생산에 관한 연구가 수행되고 있다. 상기의 미생물을 이용한 연구와 관련하여, 안전성의 측면에서 유산균을 이용한 연구가 주로 진행되고 있다(Nomura et al., Microbiology 145 : 1375-1480 (1999) 및 Park et al.,J. Food Sci. Nutri. 9 : 324-329 (2004)). 특히, 상기 유산균에 대한 연구는 유제품, 쌀 및 콩 등의 발효식품 및 김치에 대하여 주로 진행되어 왔다. 지금까지 가바의 효능 연구 및 가바가 포함된 식품에 대한 연구에 비하여 가바의 생산에 관한 연구는 아직 미비한 실정이다. 따라서, 식물 내 가바가 존재하는 것으로 알려진 뽕잎을 유산균에 의해 발효시켜 가바를 고함량으로 생산할 수 있는 방법에 대한 개발이 요구된다.Since there is a limit to the production of gaba made from plants as a raw material, research on mass production of gaba using microorganisms has been recently conducted. In relation to the study using the microorganism, studies on lactic acid bacteria have been mainly conducted in terms of safety (Nomura et al., Microbiology 145: 1375-1480 (1999) and Park et al., J. Food Sci. Nutri .9: 324-329 (2004). In particular, research on the lactic acid bacteria has been mainly conducted on fermented foods such as dairy products, rice and beans, and kimchi. Until now, research on the production of gaba has been incomplete compared to studies on the efficacy of gaba and foods containing gaba. Therefore, there is a need to develop a method capable of producing a high content of gaba by fermenting mulberry leaves known to exist in plants with lactic acid bacteria.
본 발명의 발명자들은 뽕잎을 발효하여 고함량의 가바를 생산하는 방법에 대하여 연구하던 중, 뽕잎 추출액에 김치에서 분리한 락토바실러스 플란타룸 GW0709(Lactobacillus plantarum subsp. plantarum GW0709, 수탁번호: KFCC11796P) 균주를 이용하여 무 추출액이 포함된 식물성 유산균 배지에서 발효함으로써 뽕잎 발효물에서의 가바 함량이 현저히 증가할 수 있다는 것을 발견하였다. 또한, 상기 뽕잎 발효물을 포함하는 식품 조성물에서도 가바 함량이 증가할 수 있다는 것을 확인하였다.The inventors of the present invention while trying and by fermenting the mulberry study a method of producing GABA in the content, a Lactobacillus Planta room isolated from kimchi on mulberry leaves extract GW0709 (Lactobacillus plantarum subsp plantarum GW0709, accession number:. KFCC11796P) strains It was found that the gaba content in the fermented mulberry leaf can be significantly increased by fermenting in a vegetable lactic acid bacteria medium containing no extract using. In addition, it was confirmed that the gaba content may increase in the food composition containing the fermented mulberry leaf.
따라서, 본 발명은 김치 유래 신규한 유산균을 이용한 가바 성분이 강화된 뽕잎 발효물의 제조방법을 제공하는 것을 목적으로 한다.Accordingly, an object of the present invention is to provide a method for producing a fermented product of mulberry leaves with enhanced gaba components using new lactic acid bacteria derived from kimchi.
본 발명의 일측면에 따라, (a) 건조된 뽕잎을 분쇄하여 뽕잎 분쇄물을 얻는 단계; (b) 얻어진 뽕잎 분쇄물을 추출하여 뽕잎 추출물을 얻는 단계; 및 (c) 얻어진 뽕잎 추출물에 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 균주를 접종하고 배양하여 발효물을 얻는 단계를 포함하는 가바 성분이 강화된 뽕잎 발효물의 제조방법이 제공된다.According to an aspect of the present invention, (a) pulverizing the dried mulberry leaves to obtain a mulberry pulverized product; (B) extracting the obtained mulberry leaf pulverized to obtain a mulberry leaf extract; And (c) inoculating the obtained mulberry leaf extract with Lactobacillus plantarum GW0709 (Accession No .: KFCC11796P) strain and culturing to obtain a fermentation product, wherein a method for producing a mulberry leaf fermentation product with enhanced gaba components is provided.
일 구현예에서, 단계(a)에서 건조된 뽕잎이 수분함량 5 내지 20 %로 포함될 수 있고, 단계(c)에서 뽕잎 추출물이 뽕잎 고형분을 1 내지 15 중량 %로 포함될 수 있다. 또한, 단계(c)에서 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 균주가 무 추출액이 5 내지 40 중량 %로 포함된 식물성 유산균 배지에서 배양될 수 있고, 상기 균주의 배양액이 1 내지 30 %(v/v)로 포함될 수 있다. 또한, 뽕잎 발효물은 모노소디움 글루타메이트가 0.05 내지 5 %(w/v)로 포함되고, 32 내지 38 ℃의 배양 온도에서 4일 동안 호기 배양한 후 4일 동안 혐기 배양하여 제조될 수 있다.In one embodiment, the dried mulberry leaf in step (a) may be included with a moisture content of 5 to 20%, and the mulberry leaf extract in step (c) may contain 1 to 15% by weight of the solid content of the mulberry leaf. In addition, in step (c), the Lactobacillus plantarum GW0709 (Accession No .: KFCC11796P) strain can be cultured in a vegetable lactic acid bacteria medium containing 5-40% by weight of an extract free, and the culture medium of the strain is 1-30%. (v / v). In addition, the mulberry leaf fermentation product may contain monosodium glutamate in an amount of 0.05 to 5% (w / v), and may be prepared by aerobically incubating for 4 days at an incubation temperature of 32 to 38 ° C. for 4 days.
본 발명의 다른 측면에 따라, 상기 발효방법에 의하여 발효된 가바 성분이 강화된 뽕잎 발효물이 제공된다.According to another aspect of the present invention, a fermentation product of a mulberry leaf in which a gaba component fermented by the fermentation method is strengthened is provided.
본 발명의 또 다른 측면에 따라, 상기 뽕잎 발효물을 포함하는 가바 성분이 강화된 식품 조성물 및 식품 첨가제가 제공된다.According to another aspect of the present invention, a food composition and food additives with enhanced gaba components including the fermented mulberry leaf are provided.
본 발명에 의해, 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 균주를 이용하여 특정 발효 조건에서 발효되는 본 발명의 뽕잎 발효물의 제조방법은 발효 전과 비교하여 가바 함량을 6배 이상 증가시킬 수 있다는 것이 밝혀졌다. 또한, 고함량의 가바를 포함하는 뽕잎 발효물을 이용하여 제조된 식품 조성물을 섭취시 가바의 1일 필요량(500 mg 이상)을 충분히 충족하는 것으로 확인되었다.According to the present invention, the method of manufacturing the fermented mulberry leaf of the present invention that is fermented under a specific fermentation condition using the Lactobacillus plantarum GW0709 (Accession No .: KFCC11796P) strain can increase the gaba content by more than 6 times compared to before fermentation. Turned out. In addition, it was confirmed that when ingesting a food composition prepared using a mulberry leaf fermentation product containing a high content of gaba, the daily amount of gaba (500 mg or more) was sufficiently satisfied.
따라서, 본 발명의 김치 유래 신규한 유산균을 이용한 가바 성분이 강화된 뽕잎의 발효방법은 건강 식품 분야에서 간편하게 고함량의 가바를 섭취할 수 있는 방법으로서 유용하게 사용될 수 있다.Therefore, the fermentation method of the mulberry leaf in which the gaba component is strengthened using the novel lactic acid bacteria derived from kimchi of the present invention can be conveniently used as a method for easily ingesting a high content of gaba in the health food field.
도 1은 4개의 유산균에서의 가바 생성능을 확인한 TLC 사진이다.
도 2는 유산균(GW-0709)의 16S rRNA 유전자 염기서열이다.
도 3은 유산균(GW-0709)의 계통수이다.
도 4는 뽕잎 발효물의 제조공정을 개략적으로 나타난 순서도이다.
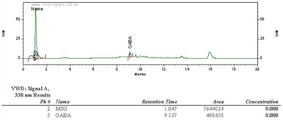
도 5는 뽕잎 비발효물의 가바함량을 분석한 크로마토그래프이다.
도 6은 뽕잎 발효물의 가바함량을 분석한 크로마토그래프이다.
도 7은 맥주 효모 가수분해물의 제조방법을 개략적으로 나타낸 순서도이다.
도 8은 혼합단백 가수분해물의 제조방법을 개략적으로 나타낸 순서도이다.
도 9는 영양보충식품의 제조방법을 개략적으로 나타낸 순서도이다. 1 is a TLC photograph confirming the gaba production capacity in four lactic acid bacteria.
2 is a 16S rRNA gene sequence of lactic acid bacteria (GW-0709).
3 is a phylogenetic tree of lactic acid bacteria (GW-0709).
Figure 4 is a flow chart schematically showing the manufacturing process of the fermented mulberry leaves.
5 is a chromatograph analyzing the gaba content of non-fermented mulberry leaves.
Figure 6 is a chromatographic analysis of the gaba content of the fermented mulberry leaves.
7 is a flow chart schematically showing a method for producing a hydrolyzate of beer yeast.
8 is a flowchart schematically showing a method for producing a mixed protein hydrolyzate.
9 is a flowchart schematically showing a method of manufacturing a nutritional supplement.
본 발명은 (a) 건조된 뽕잎을 분쇄하여 뽕잎 분쇄물을 얻는 단계; (b) 얻어진 뽕잎 분쇄물을 추출하여 뽕잎 추출물을 얻는 단계; 및 (c) 얻어진 뽕잎 추출물에 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 균주를 접종하고 배양하여 발효물을 얻는 단계를 포함하는 가바 성분이 강화된 뽕잎 발효물의 제조방법을 제공한다.The present invention (a) pulverizing the dried mulberry leaves to obtain a mulberry leaf pulverized product; (B) extracting the obtained mulberry leaf pulverized to obtain a mulberry leaf extract; And (c) inoculating the obtained mulberry leaf extract with Lactobacillus plantarum GW0709 (Accession No .: KFCC11796P) strain and obtaining a fermented product by incubating the culture, thereby providing a method for producing a fermented product of mulberry leaf with enhanced gaba components.
본 발명의 제조방법은 건조된 뽕잎을 분쇄하여 뽕잎 분쇄물을 얻는 단계[즉, 단계(a)]를 포함한다. 단계(a)에서, 신선한 뽕잎을 선별하여 채취한 후 별도의 수세공정을 거쳐 세정한 것을 물기 제거기 등의 통상의 방법을 사용하여 표피에 묻어 있는 물기를 제거한다. 물기를 제거한 뽕잎을 건조시켜 수분함량 5 ~ 20 %, 바람직하게는 7 ~ 10 %로 조절하고, 건조된 뽕잎을 2 ~ 7 mm로 조분쇄하여 뽕잎 분쇄물을 얻을 수 있다.The manufacturing method of the present invention includes the step of obtaining a mulberry pulverized product by crushing the dried mulberry leaf (that is, step (a)). In step (a), fresh mulberry leaves are selected and collected, and then washed through a separate water washing process to remove water from the epidermis using a conventional method such as a degasser. The dried mulberry leaves are dried to adjust the moisture content to 5 to 20%, preferably 7 to 10%, and coarsely pulverized dried mulberry leaves to 2 to 7 mm to obtain a mulberry pulverized product.
본 발명의 제조방법은 얻어진 뽕잎 분쇄물을 추출하여 뽕잎 추출물을 얻는 단계[즉, 단계(b)]를 포함한다. 단계(b)에서, 10 ~ 30배의 정수를 첨가한 뽕잎 분쇄물을 추출용기에 투입하고 60 ~ 100 ℃에서 추출한 후 정밀 여과기에서 갑압여과하여 정제시킨 후 농축하여 뽕잎 추출물을 제조할 수 있다.The manufacturing method of the present invention includes the step of obtaining a mulberry leaf extract by extracting the obtained mulberry leaf pulverized product (that is, step (b)). In step (b), a mulberry leaf pulverized product with an added water of 10 to 30 times is added to an extraction container, extracted at 60 to 100 ° C., purified by precision filtration in a fine filter, and then concentrated to prepare a mulberry leaf extract.
본 발명의 제조방법은 얻어진 뽕잎 추출물에 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 균주를 접종하고 배양하여 발효물을 얻는 단계[즉, 단계(c)]를 포함한다. 단계(c)에서, 뽕잎 추출물은 김치로부터 분리한 신규한 락토바실러스 플란타룸 GW0709 균주를 이용하여 발효물을 얻을 수 있다. 상기 락토바실러스 플란타룸 GW0709 균주는 한국미생물보존센터에 2018.10.19일자로 기탁된 수탁번호 KFCC11796P로서 기탁된 균주이고, 김치로부터 분리한 유산균 중에서 가장 많은 함량의 가바를 생산하는 균주로 선정되어 한국종균협회에서 생화학적 특성을 분석한 결과, 락토바실러스 플란타룸으로 99.9% 추정되었다. 락토바실러스 플란타룸 GW0709 균주는 무 추출액을 포함하는 식물성 배지에서 32 내지 38 ℃의 배양 온도에서 배양하고 24시간 후에 유청 대두단백 및 덱스트린을 첨가하여 혼합한 혼합물을 분무건조하여 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 균주 분말을 제조한다.The manufacturing method of the present invention comprises the step of obtaining a fermentation product by inoculating and culturing the Lactobacillus plantarum GW0709 (Accession No .: KFCC11796P) strain on the obtained mulberry leaf extract [ie, step (c)]. In step (c), the mulberry leaf extract can be obtained by using a new Lactobacillus plantarum GW0709 strain isolated from kimchi. The Lactobacillus plantarum GW0709 strain is a strain deposited with accession number KFCC11796P deposited as of October 19, 2018 at the Korea Microbial Conservation Center, and selected as a strain that produces the largest amount of gaba among lactic acid bacteria isolated from kimchi. As a result of analyzing the biochemical properties of the association, it was estimated to be 99.9% with Lactobacillus plantarum. The Lactobacillus plantarum GW0709 strain is cultured at a culture temperature of 32 to 38 ° C in a vegetable medium containing no extract, and after 24 hours, the mixture is added by adding whey soy protein and dextrin to spray drying to obtain a Lactobacillus plantarum GW0709. (Accession No .: KFCC11796P) A strain powder is prepared.
일 구현예에서, 단계(c)에서 뽕잎 추출물을 발효시키기 위해서 플란타룸 GW0709(수탁번호: KFCC11796P) 균주 분말 1 ~ 10 %(w/v)을 접종하여 24시간 동안 배양한 배양액을 이용할 수 있다. 뽕잎 고형분의 함량이 1 ~ 15 중량 %, 바람직하게는 5 ~ 10 중량 %인 뽕잎 추출물의 희석액에 모노소디움 글루타메이트(monosodium glutamate, MSG) 0.05 ~ 5 %(w/v), 바람직하게는 0.5 ~ 2.5 %(w/v), 가장 바람직하게는 1 %(w/v)를 첨가하고 무 추출액이 5 ~ 40 중량 %, 바람직하게는 10 ~ 30 중량 %, 가장 바람직하게는 20 중량 %로 포함된 식물 유산균 배지를 1 ~ 30 %(v/v), 바람직하게는 5 ~ 20 %(v/v), 가장 바람직하게는 10 %(v/v)로 첨가하여 혼합한 뽕잎 혼합액을 멸균한 후 멸균된 뽕잎 혼합액에 락토바실러스 플란타룸 GW0709 균주의 배양액을 1 ~ 30 %(v/v), 바람직하게는 5 ~ 20 %(v/v), 가장 바람직하게는 10 %(v/v)로 접종하고 32 ~ 38 ℃, 바람직하게는 34 ~ 36 ℃, 가장 바람직하게는 35 ℃의 배양 온도에서 4일 동안 호기 배양한 후 3일 동안 혐기 배양할 수 있다.In one embodiment, in order to ferment the mulberry leaf extract in step (c), the plantarum GW0709 (Accession No .: KFCC11796P) strain powder 1-10% (w / v) was inoculated and cultured for 24 hours can be used. . Monosodium glutamate (MSG) 0.05 to 5% (w / v), preferably 0.5 to 2.5, in a diluent of the mulberry leaf extract having a mulberry leaf solids content of 1 to 15% by weight, preferably 5 to 10% by weight Plants containing 5% to 40% by weight, preferably 10% to 30% by weight, and most preferably 20% by weight with the addition of% (w / v), most preferably 1% (w / v) Lactic acid bacteria medium is added to 1 to 30% (v / v), preferably 5 to 20% (v / v), and most preferably 10% (v / v) to sterilize the mixed mulberry leaf mixture and sterilized The mulberry leaf mixture was inoculated with 1 to 30% (v / v), preferably 5 to 20% (v / v), and most preferably 10% (v / v) of the culture solution of the Lactobacillus plantarum GW0709 strain After aerobic culture for 4 days at a culture temperature of 32 to 38 ° C, preferably 34 to 36 ° C, most preferably 35 ° C, anaerobic culture may be performed for 3 days.
또한, 본 발명은 상기 발효방법에 의하여 발효된 가바 성분이 강화된 뽕잎 발효물을 제공한다. 상기 뽕잎 발효물의 가바 함량을 HPLC 분석을 이용하여 분석한 결과, 뽕잎 비발효물에 비하여 가바 함량이 약 6배 이상 증가한 것을 확인하여 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 균주를 이용한 발효방법이 뽕잎의 가바 성분을 강화시킨다는 것을 확인하였다.In addition, the present invention provides a fermented mulberry leaf fermented gaba component fermented by the fermentation method. As a result of analyzing the gaba content of the fermented mulberry leaf using HPLC analysis, it was confirmed that the gaba content increased by about 6 times or more compared to the non-fermented mulberry leaf, and the fermentation method using the Lactobacillus plantarum GW0709 (Accession No .: KFCC11796P) strain It was confirmed that the gaba component of this mulberry leaf was strengthened.
또한, 본 발명은 상기 뽕잎 발효물을 포함하는 가바 성분이 강화된 식품 조성물을 및 식품 첨가제를 제공한다. 상기 뽕잎 발효물은 가바 성분이 강화된 특성이 있으므로, 이를 혼합하여 다양한 건강 제품으로 이용될 수 있다. 구체적으로, 상기 뽕잎 발효물에 혼합단백 가수분해물, 효모 가수분해물 및 비타민-무기질 복합물을 첨가하고 분무건조하여 영양보충제품을 제조할 수 있으며, 이로부터 제조된 영양보충제품은 가바 함량이 식품으로 섭취할 수 있는 1일 필요량(500 mg 이상)을 충분히 충족하는 것을 확인하였다. 또한, 상기 뽕잎 발효물을 포함하는 식품 첨가제는 가바 함량을 증진시키는 기능성 식품 또는 음료의 제조에 첨가제로서 용이하게 활용할 수 있다.In addition, the present invention provides a food composition and food additives are enhanced Gaba component containing the fermented mulberry leaves. The mulberry leaf fermentation product has a characteristic that the gaba component is strengthened, so it can be mixed and used as a variety of health products. Specifically, a nutrient supplement product may be prepared by adding a mixed protein hydrolyzate, yeast hydrolyzate, and vitamin-inorganic complex to the mulberry leaf fermentation product and spray-drying it, and the nutritional supplement product prepared therefrom is consumed as food. It was confirmed that the daily required amount (500 mg or more) could be sufficiently satisfied. In addition, the food additive containing the fermented mulberry leaf can be easily utilized as an additive in the production of a functional food or beverage that enhances the gaba content.
이하, 본 발명을 실시예를 통하여 더욱 상세히 설명한다. 그러나, 하기 실시예는 본 발명을 예시하기 위한 것으로, 본 발명의 범위가 이에 제한되는 것은 아니다.Hereinafter, the present invention will be described in more detail through examples. However, the following examples are intended to illustrate the present invention, and the scope of the present invention is not limited thereto.
<실시예><Example>
1. 유산균의 분리 및 동정1. Isolation and identification of lactic acid bacteria
(1) 분리(1) separation
시중에서 수집한 배추김치, 무김치를 각각 파쇄하여 멸균수로 희석한 후, 희석액을 MRS 고체배지(MRS agar medium : Difco, USA)에 접종하고, 37 ℃에서 48시간 동안 배양하여 콜로니를 형성한 균주(GW-0507, GW-0608, GW-0709 및 GW-40014)를 분리하였다.Strains of cabbage kimchi and radish kimchi collected commercially are diluted with sterile water, and then inoculated with MRS agar medium (Difco, USA), and cultured at 37 ° C for 48 hours to form colonies. (GW-0507, GW-0608, GW-0709 and GW-40014) were isolated.
(2) 가바 생성능 유산균 선별(2) Selection of lactic acid bacteria capable of producing gaba
분리한 균주로부터 가바생성능이 있는 균주를 선별하기 위하여, MRS배지에 모노소디움 글루타메이트(monosodium glutamate, MSG) 1 %를 첨가하고 각 균주를 접종하여 24시간 동안 배양 후 TLC를 실시하여 균주의 GABA 생산성 유무를 측정하였다. 도 1에 나타난 바와 같이, TLC를 이용한 선별 실험에서 가장 많은 함량의 가바를 생산하는 균주(GW-0709)를 본 발명에 따른 균주로서 최종 선정하였다.In order to select strains with Gaba production performance from the isolated strains, monosodium glutamate (MSG) 1% is added to the MRS medium, inoculated with each strain, and cultured for 24 hours, followed by TLC to incubate for strain to determine whether the strain has GABA productivity or not. Was measured. As shown in Figure 1, in the screening experiment using TLC, the strain that produces the largest amount of gaba (GW-0709) was finally selected as the strain according to the present invention.
(3) 분리된 가바생성 균주의 동정(3) Identification of isolated Gaba-producing strains
가바 생성능이 가장 높은 균주로 선정된 유산균(GW-0709)을 한국종균협회에 동정을 의뢰하였다. 상기 유산균의 생화학적 특성을 API 50-CHL 키트를 이용하여 분석하였다. 표 1에서 나타난 바와 같이, L-아라비노스, 리보스, 갈락토스, D-글루코스, D-프럭토스, D-만노스, 만니톨, 소르비톨 등을 분해하였으나, D-자일로스, 아도니톨, L-소르보스, 람노스 등은 분해하지 못한 결과로부터 유산균(GW-0709)은 락토바실러스 플란타룸(Lactobacillus plantarum)으로 99.9 %로 추정되었다.Lactobacillus (GW-0709), which was selected as the strain with the highest gaba production ability, was referred to the Korean Society of Seeds. The biochemical properties of the lactic acid bacteria were analyzed using the API 50-CHL kit. As shown in Table 1, L-arabinose, ribose, galactose, D-glucose, D-fructose, D-mannose, mannitol, sorbitol, etc. were decomposed, but D-xylose, adonitol, and L-sorbose , Rhamnose, etc., Lactobacillus plantarum ( Lactobacillus plantarum ) was estimated to be 99.9% from lactic acid bacteria (GW-0709).
또한, 본 발명의 유산균의 16S rRNA 유전자의 염기서열을 조사한 결과는 도 2에 나타난 바와 같이 약 1,477 bp의 염기서열(서열번호 1)을 조사하였으며, 분자계통학적 염기서열 검색을 이용하여 미생물 동정을 확인하였다. 이로부터 락토바실러스 플란타룸 서브스피시스 플란타룸(Lactobacillus plantarum subsp. plantarum)과 99 %의 상동성(Gene Bank Data homology)을 확인하여, 상기 유산균을 락토바실러스 플란타룸(Lactobacillus plantarum subsp. plantarum)으로 동정하고(도 3 참조), 이를 락토바실러스 플란타룸 GW0709(Lactobacillus plantarum subsp. plantarum GW0709) 균주로 명명하였다. 상기 본 발명에 따른 유산균 균주를 한국미생물보존센터에 2018.10.19일자 수탁번호 KFCC11796P로서 기탁하였다.In addition, as a result of examining the base sequence of the 16S rRNA gene of the lactic acid bacteria of the present invention, a base sequence of about 1,477 bp (SEQ ID NO: 1) was investigated as shown in FIG. 2, and microbial identification was performed using molecular sequence search. Confirmed. From this, Lactobacillus Plantarum subsphysis Plantarum ( Lactobacillus plantarum subsp. Plantarum ) and 99% homology (Gene Bank Data homology) was confirmed to confirm the lactobacillus. The plant was identified as Lactobacillus plantarum subsp. Plantarum (see FIG. 3), and it was lactobacillus. It was named as a plantarum GW0709 ( Lactobacillus plantarum subsp. Plantarum GW0709) strain. The lactic acid bacteria strain according to the present invention was deposited with the Korea Microbial Conservation Center as accession number KFCC11796P on October 19, 2018.
2. 뽕잎 발효2. Mulberry leaf fermentation
(1) 뽕잎 추출물의 제조(1) Preparation of mulberry leaf extract
조분쇄하여 수분함량 약 8 %로 건조된 뽕잎 1 ㎏에 20배의 정수 20 ㎏을 가하여 80 ℃에서 6시간 동안 추출하였다. 이 추출액을 감압여과한 후 농축하여 뽕잎 추출물을 제조하였다.20 kg of purified water was added to 1 kg of mulberry leaves that were coarsely crushed and dried to a water content of about 8%, and extracted at 80 ° C. for 6 hours. The extract was filtered under reduced pressure and concentrated to prepare a mulberry leaf extract.
(2) 유산균의 배양(2) Culture of lactic acid bacteria
무말랭이 중량 대비 20배수의 물을 가하여 100 ℃에서 1시간 동안 가열한 후 여과하여 얻은 무 추출액을 포함하는 식물성 유산균 배지(하기 표 2 참조)를 제조하여 121 ℃에서 15분간 멸균한 후, 가바 생성 균주로 선별된 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P)를 접종하여, 35 ℃에서 24시간 동안 배양한 후, 유청 대두단백과 덱스트린을 각각 15 %(w/w)로 첨가하여 용해한 후, 분무건조하여 유산균 분말을 제조하였다.A 20-fold water-to-weight ratio was added, heated at 100 ° C. for 1 hour, and then filtered to produce a vegetable lactic acid bacteria medium (see Table 2 below) containing the extract, and sterilized at 121 ° C. for 15 minutes, followed by a Gaba-producing strain After inoculation with Lactobacillus plantarum GW0709 (Accession No .: KFCC11796P), which was selected as, and cultured at 35 ° C. for 24 hours, whey soy protein and dextrin were added at 15% (w / w), dissolved, and sprayed. Drying to prepare a lactic acid bacteria powder.
상기 제조된 분말을 멸균한 무 추출액이 포함된 식물성 유산균 배지에 유산균 분말 5 %(w/v)를 접종하여 24시간 동안 발효용 유산균을 배양하였다.Lactic acid bacteria for fermentation were incubated for 24 hours by inoculating 5% (w / v) of lactic acid bacteria powder in the vegetable lactic acid bacteria medium containing the sterile, non-extracted powder.
(3) 뽕잎 발효조건(3) Mulberry leaf fermentation conditions
뽕잎 발효물 제조에 있어서 최적의 조건을 확립하기 위하여 하기 표 3의 뽕잎 추출물의 농도 및 배지의 조성에 따른 3가지 뽕잎 발효물을 제조하였다.In order to establish optimal conditions in the preparation of mulberry leaf fermentation, three mulberry leaf fermentations were prepared according to the concentration of the mulberry leaf extract in Table 3 and the composition of the medium.
뽕잎 발효물 A는 뽕잎 고형분이 5 %(w/w)로 포함되도록 희석하고, 포도당 1.6 %(w/v), 효모 가수분해물 0.8 %(w/v), 모노소디움 글루타메이트(MSG) 1 %(w/v), 시스테인염산 0.08 %(w/v)를 첨가하여 121 ℃에서 15분간 살균하였다. 여기에 유산균 배양액 10 %(v/v)를 접종하였다.Mulberry leaf fermentation product A was diluted so that the mulberry leaf solid content was 5% (w / w), glucose 1.6% (w / v), yeast hydrolyzate 0.8% (w / v), monosodium glutamate (MSG) 1% ( w / v) and cysteine hydrochloric acid 0.08% (w / v) were added and sterilized at 121 ° C for 15 minutes. Here, 10% (v / v) of lactic acid bacteria culture was inoculated.
뽕잎 발효물 B는 뽕잎 고형분이 5 %(w/w)로 포함되도록 희석하고, 식물성 유산균배지GW(무즙배지) 10 %(v/v), 모노소디움 글루타메이트(MSG) 1 %(w/v)를 첨가하여 121 ℃에서 15분간 살균하였다. 여기에 유산균 배양액 10 %(v/v)를 접종하였다. Mulberry leaf fermentation product B is diluted to include 5% (w / w) of mulberry leaf solids, 10% (v / v) of vegetable lactic acid bacteria medium (v / v), 1% of monosodium glutamate (MSG) (w / v) Was added and sterilized at 121 ° C for 15 minutes. Here, 10% (v / v) of lactic acid bacteria culture was inoculated.
뽕잎 발효물 C는 뽕잎 고형분이 10 %(w/w)로 포함되도록 희석하고, 포도당 1.6 %(w/v), 효모 가수분해물 0.8 %(w/v), 모노소디움 글루타메이트(MSG) 1 %(w/v), 시스테인염산 0.08 %(w/v)를 첨가하여 121 ℃에서 15분간 살균하였다. 여기에 유산균 배양액 10 %(v/v)를 접종하였다. Mulberry leaf fermentation product C is diluted to contain 10% (w / w) of mulberry leaf solids, glucose 1.6% (w / v), yeast hydrolyzate 0.8% (w / v), monosodium glutamate (MSG) 1% ( w / v) and cysteine hydrochloric acid 0.08% (w / v) were added and sterilized at 121 ° C for 15 minutes. Here, 10% (v / v) of lactic acid bacteria culture was inoculated.
뽕잎 발효물 A, B 및 C를 35 ℃에서 4일 동안 호기배양한 후 8일 동안 혐기배양하면서 가바함량 및 pH의 변화를 관찰하였다.The mulberry leaf fermentation products A, B and C were aerobically cultured at 35 ° C. for 4 days, followed by anaerobic culture for 8 days to observe changes in gaba content and pH.
표 4에 나타난 바와 같이, 가바함량은 발효초기에서 7 ~ 8일까지 증가하는 경향을 나타내고, 발효 8일 이후부터 감소하는 경향을 나타내었으며, 특히, 뽕잎 발효물 B의 조성에서 가바함량이 가장 높은 것으로 확인되었다. 또한, 표 5에 나타난 바와 같이, 뽕잎 발효물 B에서 pH는 발효 7일까지 감소하는 경향을 나타내고, 가바함량이 최대로 증가하는 발효 8일부터 pH가 높아지는 경향을 나타내었다.As shown in Table 4, the gaba content tends to increase from 7 to 8 days at the beginning of fermentation, and tends to decrease from 8 days after fermentation. In particular, the composition of the mulberry leaf fermentation product B has the highest gaba content. Was confirmed. In addition, as shown in Table 5, the pH of the mulberry leaf fermentation product B tended to decrease until 7 days of fermentation, and the pH increased from 8 days of fermentation, where the gaba content increased to the maximum.
상기 실험의 결과로부터, 고함량의 가바 생산을 위한 뽕잎 발효물 제조를 위하여 뽕잎 추출물은 고형분이 5 %(w/w)로 포함되도록 하고, 식물성 유산균배지GW(무즙배지) 10 %(v/v), 모노소디움 글루타메이트(MSG) 1 %(w/v)를 첨가한 후 유산균 배양액 10 %(v/v)를 접종하여 4일간 호기배양 후 4일간 혐기배양하는 발효조건이 가장 바람직한 것으로 확인되었다.From the results of the above experiment, for the production of fermented mulberry leaves for the production of high-content Gaba, the mulberry leaf extract contains 5% (w / w) of solid content, and 10% (v / v) of vegetable lactic acid bacteria medium GW (juice-free medium) ), Monosodium glutamate (MSG) 1% (w / v) was added, followed by inoculation of 10% (v / v) of lactic acid bacteria culture, followed by aerobic culture for 4 days and anaerobic culture for 4 days.
(4) 뽕잎 발효물의 제조(4) Preparation of fermented mulberry leaves
뽕잎 추출물의 발효를 위하여 락토바실러스 플란타룸 GW0709(Lactobacillus plantarum subsp. plantarum GW0709, 수탁번호: KFCC11796P) 균주의 배양액을 이용하였다.For the entry into force of the mulberry leaf extract of Lactobacillus Planta room GW0709 (Lactobacillus plantarum subsp plantarum GW0709, accession number:. KFCC11796P) was used as the culture medium of the strain.
뽕잎 추출물은 고형분이 5 %(w/w)로 포함되도록 희석하고, 식물성 유산균배지GW(무즙 배지) 10 %(v/v), 모노소디움 글루타메이트(MSG) 1 %(w/v)를 첨가하여 121 ℃에서 15분간 살균하였다. 여기에 유산균 배양액 10 중량 %(v/v)를 접종하고 4일간 호기발효한 후 4일간 혐기발효하여 뽕잎 발효액을 제조하였다. 제조된 뽕잎 발효액 및 유산균 배양액만 제외된 뽕잎 비발효액을 각각 농축한 후 분무건조하여 뽕잎 발효물 및 뽕잎 비발효물(대조군)을 수득하였다(도 4 참조).Mulberry leaf extract is diluted to contain 5% (w / w) of solids, and 10% (v / v) of vegetable lactic acid bacteria medium (v / v) and 1% (w / v) of monosodium glutamate (MSG) are added. Sterilized at 121 ° C for 15 minutes. Here, 10% by weight of lactic acid bacteria culture solution (v / v) was inoculated, followed by aerobic fermentation for 4 days, followed by anaerobic fermentation for 4 days to prepare a fermented mulberry leaf. The prepared mulberry leaf fermentation broth and lactic acid bacteria non-fermentation broth except for the culture medium were concentrated and spray-dried to obtain a fermented mulberry fermentation and a non-fermented mulberry fermentation (control) (see FIG. 4).
(5) 가바의 분석 방법(5) Gaba analysis method
뽕잎 발효액의 가바(GABA)의 분석은 HPLC를 이용하여 하기 조건으로 수행하였다. 가바의 분리를 위하여 Agilent Poroshell HPH-C18(2.1 × 100 ㎜, 4 ㎛) 컬럼을 사용하였고, 가드(guard) 컬럼으로서 Agilent Poroshell HPH-C18(2.1 × 5 ㎜, 4 ㎛) 컬럼을 사용하였으며, 40 ℃로 컬럼 온도를 유지하여 분석하였다. 이동상 A 용매는 10 mM 제2인산나트륨(Na2HPO4) 용액 및 10 mM 테트라붕산나트륨(Na2B4O7·10H2O) 용액을 1 : 1(v/v)로 혼합한 후, 인산(H3PO4)을 이용하여 pH 8.2로 조절된 용액을 제조하였다. 이동상 B 용매는 아세토니트릴(acetonitrile), 메탄올 및 물을 45 : 45 : 10(v : v : v)의 비율로 혼합하여 제조하였다. 이동상은 0분 2 %:B, 5분 2 %:B, 9분 57 %:B, 11분 80 %:B, 13분 80 %:B, 13.1분 2 %:B, 15분 20 %:B, 20분 2 %:B로 변화를 주었으며, 유속은 분당 0.5 ㎖로 흐르도록 하였다. 분석 시료는 10배수로 희석한 후, 0.45 ㎛ 주사기 필터(syringe filter)를 이용하여 여과하였다. 그 다음, 오토 샘플러(auto sampler)를 이용하여 OPA(o-phthaldialdehyde) 유도체화한 후 이 중에서 5 ㎕을 주입하여 UV 검출기 338 ㎚로 결과를 측정하였다(도 5 및 도 6 참조).Analysis of Gaba (GABA) of the mulberry leaf fermentation broth was performed under the following conditions using HPLC. An Agilent Poroshell HPH-C18 (2.1 × 100 mm, 4 μm) column was used for separation of the gaba, and an Agilent Poroshell HPH-C18 (2.1 × 5 mm, 4 μm) column was used as a guard column, 40 It was analyzed by maintaining the column temperature at ℃. The mobile phase A solvent was mixed with a 10 mM sodium diphosphate (Na 2 HPO 4 ) solution and a 10 mM sodium tetraborate (Na 2 B 4 O 7 · 10H 2 O) solution at 1: 1 (v / v), A solution adjusted to pH 8.2 was prepared using phosphoric acid (H 3 PO 4 ). The mobile phase B solvent was prepared by mixing acetonitrile, methanol and water in a ratio of 45:45:10 (v: v: v). Mobile phase is 0
(6) 뽕잎 발효액의 가바 함량 분석(6) Gaba content analysis of fermented mulberry leaves
뽕잎 추출물에 MSG를 첨가한 조건에서 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 배양액으로 발효시킨 뽕잎 발효물의 가바함량(5.78 %)이 뽕잎 비발효물(대조군)의 가바 함량(0.89 %)에 비해 약 6배 이상 증가한 것을 확인하였다(표 6 참조).The gaba content (5.78%) of the fermented mulberry fermentation fermented with Lactobacillus plantarum GW0709 (Accession No .: KFCC11796P) culture under the condition that MSG was added to the mulberry leaf extract was added to the gaba content (0.89%) of the non-fermented mulberry leaf (control). It was confirmed that the increase was about 6 times or more as compared (see Table 6).
3. 영양보충식품의 제조3. Preparation of nutritional supplements
뽕잎 발효물에 효모 가수분해물과 혼합단백 가수분해물, 비타민-무기질 혼합물을 혼합 및 분무건조하여 영양보충식품을 제조하였다.Yeast hydrolysates, mixed protein hydrolysates, and vitamin-inorganic mixtures were mixed with mulberry leaf fermentation products and spray-dried to prepare nutritional supplements.
(1) 효모 가수분해물의 제조(1) Preparation of yeast hydrolyzate
맥주 발효 후 여과하여 얻어진 맥주 효모 슬러지를 정수로 1차 세척하고, 데칸터(decanter)로 여과한 세척된 맥주 효모에 정수를 첨가하여 고형분이 10 %(w/w)로 포함되도록 조정한 후, 컴플렉스 FF 104 효소(Complex enzyme FF 104) 0.3 %(w/v)를 첨가하여 55 ℃에서 15시간 동안 가수분해하였다. 그 다음, 90 ℃에서 30분 동안 열처리하여 효소의 반응을 정지하고, 100 메쉬로 여과하여 효모 가수분해물을 수득하였다(도 7 참조).After the beer fermentation, the beer yeast sludge obtained by filtration is first washed with purified water, and purified water is added to the washed beer yeast filtered with a decanter to adjust the solid content to include 10% (w / w), 0.3% (w / v) of Complex enzyme FF 104 was added and hydrolyzed at 55 ° C. for 15 hours. Then, the reaction of the enzyme was stopped by heat treatment at 90 ° C. for 30 minutes, and filtered with 100 mesh to obtain a yeast hydrolyzate (see FIG. 7).
(2) 혼합단백 가수분해물(2) Mixed protein hydrolyzate
완두단백, 대두분리단백 및 유청분리단백을 혼합한 혼합물 1 kg에 증류수 9L를 첨가하고 혼합하여 혼합액을 얻고, 여기에 프로타맥스(Protamex) 0.3 %(w/v) 및 플라보자임(Flavourzyme) 0.3 %(w/v)를 첨가하여 15시간 동안 가수분해하였다. 그 다음, 90 ℃에서 30분 동안 열처리하여 효소의 반응을 정지하고, 100 메쉬로 여과하여 혼합단백 가수분해물을 수득하였다(도 8 참조).To 1 kg of a mixture of pea protein, soybean isolate protein and whey isolate protein, 9 L of distilled water was added and mixed to obtain a mixed solution, wherein Protamex 0.3% (w / v) and flavozyme (Flavourzyme) Hydrolysis was performed for 15 hours by adding 0.3% (w / v). Then, the reaction of the enzyme was stopped by heat treatment at 90 ° C. for 30 minutes, and filtered with 100 mesh to obtain a mixed protein hydrolyzate (see FIG. 8).
(3) 영양보충제품의 제조(3) Preparation of nutritional supplement products
뽕잎 발효물에 혼합단백 가수분해물, 효모 가수분해물 및 비타민-무기질 복합물을 첨가하여 분무건조하여 영양보충제품을 제조하였다.A mixed protein hydrolyzate, yeast hydrolyzate, and vitamin-inorganic complex were added to the mulberry leaf fermentation product, and spray-dried to prepare a nutritional supplement product.
뽕잎 발효물 1,000 g에 혼합단백 가수분해물 500 g, 효모 가수분해물 1,000 g 및 비타민·무기질 복합물 2 g을 첨가하여 혼합하고, 이를 여과한 후 90 ℃에서 30분 동안 살균한 다음, 이를 분무건조하여 제조하였다(도 9 참조).500 g of mixed protein hydrolyzate, 1,000 g of yeast hydrolyzate and 2 g of vitamin / mineral complex are added to 1,000 g of fermented mulberry leaves, filtered, sterilized at 90 ° C for 30 minutes, and then spray dried to prepare. (See FIG. 9).
(4) 영양보충제품의 성분 분석 결과(4) Results of ingredient analysis of nutritional supplement products
뽕잎 발효물이 포함된 영양보충제품에 대한 성분별 분석 결과를 하기 표 7에 나타내었다. 영양보충제품의 분석 결과 가바는 676 ㎎/100 g을 함유하고, 조단백질 함량은 58.6 g/100 g으로 확인되었다. 이로부터 상기 영양보충제품 100 g을 섭취하여 가바의 1일 필요량(500 mg ~ 3000 mg, BioWave 9(14): 1-18 (2007))을 충족하는 것으로 나타났다.Table 7 shows the results of analysis for each component of the nutritional supplement product containing the fermented mulberry leaves. As a result of analysis of nutritional supplement products, Gaba contained 676 mg / 100 g, and the crude protein content was found to be 58.6 g / 100 g. From this, it was found that 100 g of the nutritional supplement product was ingested to meet the daily requirement of Gaba (500 mg to 3000 mg, BioWave 9 (14): 1-18 (2007)).
또한, 상기 영양보충제품의 성분을 표 8에 나타내었다. 이로부터 상기 영양보충제품 1회 제공량 40 g을 섭취하는 경우 아미노산, 비타민, 무기질의 성인남성(19세 ~ 29세) 1일 권장섭취량(보건복지부·한국영양학회 2015 한국인 영양소 섭취기준)에 대한 비율이 30 % 이상인 것으로 나타났다.In addition, the components of the nutritional supplement products are shown in Table 8. From this, the ratio of the recommended daily intake of adult males (ages 19 to 29) of amino acids, vitamins, and minerals (intake standards for Korean nutrients by the Ministry of Health and Welfare) It was found to be over 30%.
※ 보건복지부·한국영양학회 2015 한국인 영양소 섭취기준※ Ministry of Health and Welfare · Korean Nutrition Society 2015 Korean Nutrients Intake Standards
<110> GEONWOO FP Co., Ltd. <120> Method of preparing fermented product of mulberry leaves with enhanced GABA component and food composition comprising the fermented product thereby <130> P180914 <160> 1 <170> KoPatentIn 3.0 <210> 1 <211> 1477 <212> RNA <213> Lactobacillus plantarum <400> 1 caggacgaac gctggcggcg tgcctaatac atgcaagtcg aacgaactct ggtattgatt 60 ggtgcttgca tcatgattta catttgagtg agtggcgaac tggtgagtaa cacgtgggaa 120 acctgcccag aagcggggga taacacctgg aaacagatgc taataccgca taacaacttg 180 gaccgcatgg tccgagcttg aaagatggct tcggctatca cttttggatg gtcccgcggc 240 gtattagcta gatggtgggg taacggctca ccatggcaat gatacgtagc cgacctgaga 300 gggtaatcgg ccacattggg actgagacac ggcccaaact cctacgggag gcagcagtag 360 ggaatcttcc acaatggacg aaagtctgat ggagcaacgc cgcgtgagtg aagaagggtt 420 tcggctcgta aaactctgtt gttaaagaag aacatatctg agagtaactg ttcaggtatt 480 gacggtattt aaccagaaag ccacggctaa ctacgtgcca gcagccgcgg taatacgtag 540 gtggcaagcg ttgtccggat ttattgggcg taaagcgagc gcaggcggtt ttttaagtct 600 gatgtgaaag ccttcggctc aaccgaagaa gtgcatcgga aactgggaaa cttgagtgca 660 gaagaggaca gtggaactcc atgtgtagcg gtgaaatgcg tagatatatg gaagaacacc 720 agtggcgaag gcggctgtct ggtctgtaac tgacgctgag gctcgaaagt atgggtagca 780 aacaggatta gataccctgg tagtccatac cgtaaacgat gaatgctaag tgttggaggg 840 tttccgccct tcagtgctgc agctaacgca ttaagcattc cgcctgggga gtacggccgc 900 aaggctgaaa ctcaaaggaa ttgacggggg cccgcacaag cggtggagca tgtggtttaa 960 ttcgaagcta cgcgaagaac cttaccaggt cttgacatac tatgcaaatc taagagatta 1020 gacgttccct tcggggacat ggatacaggt ggtgcatggt tgtcgtcagc tcgtgtcgtg 1080 agatgttggg ttaagtcccg caacgagcgc aacccttatt atcagttgcc agcattaagt 1140 tgggcactct ggtgagactg ccggtgacaa accggaggaa ggtggggatg acgtcaaatc 1200 atcatgcccc ttatgacctg ggctacacac gtgctacaat ggatggtaca acgagttgcg 1260 aactcgcgag agtaagctaa tctcttaaag ccattctcag ttcggattgt aggctgcaac 1320 tcgcctacat gaagtcggaa tcgctagtaa tcgcggatca gcatgccgcg gtgaatacgt 1380 tcccgggcct tgtacacacc gcccgtcaca ccatgagagt ttgtaacacc caaagtcggt 1440 ggggtaacct tttaggaacc agccgcctaa gggtgga 1477 <110> GEONWOO FP Co., Ltd. <120> Method of preparing fermented product of mulberry leaves with enhanced GABA component and food composition comprising the fermented product thereby <130> P180914 <160> 1 <170> KoPatentIn 3.0 <210> 1 <211> 1477 <212> RNA <213> Lactobacillus plantarum <400> 1 caggacgaac gctggcggcg tgcctaatac atgcaagtcg aacgaactct ggtattgatt 60 ggtgcttgca tcatgattta catttgagtg agtggcgaac tggtgagtaa cacgtgggaa 120 acctgcccag aagcggggga taacacctgg aaacagatgc taataccgca taacaacttg 180 gaccgcatgg tccgagcttg aaagatggct tcggctatca cttttggatg gtcccgcggc 240 gtattagcta gatggtgggg taacggctca ccatggcaat gatacgtagc cgacctgaga 300 gggtaatcgg ccacattggg actgagacac ggcccaaact cctacgggag gcagcagtag 360 ggaatcttcc acaatggacg aaagtctgat ggagcaacgc cgcgtgagtg aagaagggtt 420 tcggctcgta aaactctgtt gttaaagaag aacatatctg agagtaactg ttcaggtatt 480 gacggtattt aaccagaaag ccacggctaa ctacgtgcca gcagccgcgg taatacgtag 540 gtggcaagcg ttgtccggat ttattgggcg taaagcgagc gcaggcggtt ttttaagtct 600 gatgtgaaag ccttcggctc aaccgaagaa gtgcatcgga aactgggaaa cttgagtgca 660 gaagaggaca gtggaactcc atgtgtagcg gtgaaatgcg tagatatatg gaagaacacc 720 agtggcgaag gcggctgtct ggtctgtaac tgacgctgag gctcgaaagt atgggtagca 780 aacaggatta gataccctgg tagtccatac cgtaaacgat gaatgctaag tgttggaggg 840 tttccgccct tcagtgctgc agctaacgca ttaagcattc cgcctgggga gtacggccgc 900 aaggctgaaa ctcaaaggaa ttgacggggg cccgcacaag cggtggagca tgtggtttaa 960 ttcgaagcta cgcgaagaac cttaccaggt cttgacatac tatgcaaatc taagagatta 1020 gacgttccct tcggggacat ggatacaggt ggtgcatggt tgtcgtcagc tcgtgtcgtg 1080 agatgttggg ttaagtcccg caacgagcgc aacccttatt atcagttgcc agcattaagt 1140 tgggcactct ggtgagactg ccggtgacaa accggaggaa ggtggggatg acgtcaaatc 1200 atcatgcccc ttatgacctg ggctacacac gtgctacaat ggatggtaca acgagttgcg 1260 aactcgcgag agtaagctaa tctcttaaag ccattctcag ttcggattgt aggctgcaac 1320 tcgcctacat gaagtcggaa tcgctagtaa tcgcggatca gcatgccgcg gtgaatacgt 1380 tcccgggcct tgtacacacc gcccgtcaca ccatgagagt ttgtaacacc caaagtcggt 1440 ggggtaacct tttaggaacc agccgcctaa gggtgga 1477
Claims (10)
(b) 얻어진 뽕잎 분쇄물을 추출하여 뽕잎 추출물을 얻는 단계; 및
(c) 얻어진 뽕잎 추출물에 락토바실러스 플란타룸 GW0709(수탁번호: KFCC11796P) 균주를 접종하고 배양하여 발효물을 얻는 단계
를 포함하는 가바 성분이 강화된 뽕잎 발효물의 제조방법.(a) pulverizing the dried mulberry leaf to obtain a mulberry leaf pulverized product;
(B) extracting the obtained mulberry leaf pulverized to obtain a mulberry leaf extract; And
(c) inoculating the obtained mulberry leaf extract with Lactobacillus plantarum GW0709 (Accession number: KFCC11796P) strain and culturing to obtain a fermentation product
A method of manufacturing a fermented mulberry leaf with enhanced gaba ingredients comprising a.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180144896A KR20200059773A (en) | 2018-11-21 | 2018-11-21 | Method of preparing fermented product of mulberry leaves with enhanced GABA component and food composition comprising the fermented product thereby |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180144896A KR20200059773A (en) | 2018-11-21 | 2018-11-21 | Method of preparing fermented product of mulberry leaves with enhanced GABA component and food composition comprising the fermented product thereby |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20200059773A true KR20200059773A (en) | 2020-05-29 |
Family
ID=70911931
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020180144896A KR20200059773A (en) | 2018-11-21 | 2018-11-21 | Method of preparing fermented product of mulberry leaves with enhanced GABA component and food composition comprising the fermented product thereby |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR20200059773A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114933989A (en) * | 2022-05-18 | 2022-08-23 | 四川生力源生物工程有限公司 | Lactobacillus plantarum and fermentation medium thereof, mulberry leaf tea rich in gamma-aminobutyric acid and preparation method of mulberry leaf tea |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20010100061A (en) | 2001-08-18 | 2001-11-14 | -- | Manufacturing method of mulberry extract and liquid beverage containing this |
| KR101067979B1 (en) | 2011-03-17 | 2011-09-26 | 양평군 | Functional beverage using fermentation mulberry leaf / tree and manufacturing method thereof |
-
2018
- 2018-11-21 KR KR1020180144896A patent/KR20200059773A/en not_active Application Discontinuation
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20010100061A (en) | 2001-08-18 | 2001-11-14 | -- | Manufacturing method of mulberry extract and liquid beverage containing this |
| KR101067979B1 (en) | 2011-03-17 | 2011-09-26 | 양평군 | Functional beverage using fermentation mulberry leaf / tree and manufacturing method thereof |
Non-Patent Citations (1)
| Title |
|---|
| Korean J. Medicinal Crop Sci. 19(4) : 276-286 (2011) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114933989A (en) * | 2022-05-18 | 2022-08-23 | 四川生力源生物工程有限公司 | Lactobacillus plantarum and fermentation medium thereof, mulberry leaf tea rich in gamma-aminobutyric acid and preparation method of mulberry leaf tea |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Park et al. | Production of yogurt with enhanced levels of gamma-aminobutyric acid and valuable nutrients using lactic acid bacteria and germinated soybean extract | |
| KR102134209B1 (en) | Novel strains derived from fermented food and having with excellent enzyme activity and method for producing grains-fermented food using the same | |
| KR102361556B1 (en) | Extracellular polysaccharides of lactic acid bacteria and their uses | |
| JP2008061584A (en) | Microorganism having equol-producing ability, food and beverage, medicine, animal feed and method for producing the same equol | |
| WO2014208848A1 (en) | Lactobacillus brevis having high productivity of gamma-amino butyric acid and use thereof | |
| AU2006309588B2 (en) | Method of producing gaba-containing fermented product | |
| KR102044602B1 (en) | Preparation method for soybean-fermented composition having enhanced GABA, Conjugated linoleic acid and Aglycone isoflavones, combined probiotics for its fermentation and functional food of anti-diabetic and anti-obesity effects containing the soybean-fermented composition | |
| KR20070016253A (en) | 1-deoxynojirimycin-producing bacillus subtilis s10 strain and composition comprising same | |
| KR101980450B1 (en) | Composition of active wild-cultivated ginseng having increased ginsenoside Rd and compound K, chlorgenic acid and quercetin, and preparation method thereof | |
| KR101091833B1 (en) | Methods for preparing high quantity of S-allyl-L-cysteine to fermented products of garlic using lactic acid bacteria | |
| KR20130107940A (en) | Method of producing fermented red ginseng | |
| KR101767606B1 (en) | Method for producing rubusoside from stevioside using lactic acid bacteria | |
| KR20190018204A (en) | Lactobacillus brevis WCP02 strain having high gastric acid tolerance and bile acid, and high GABA productivity, and probiotics and fermented food by using the same | |
| KR20200059773A (en) | Method of preparing fermented product of mulberry leaves with enhanced GABA component and food composition comprising the fermented product thereby | |
| KR100631857B1 (en) | -3 Lactobacillus Brevis OPK-3 Having a High Production Ability of Gamma-Aminobutyric Acid | |
| KR101481791B1 (en) | The strain of pediococcus acidilactici DM-9 having ornithine production capacity and the method of cheonggukjang preparation containing orenithine | |
| KR102567790B1 (en) | Lactobacillus gasseri GFC_1220(GFC_EJR_JOY) for alcohol metabolism enzyme activity and food composition comprising the same as an effective ingredient | |
| WO1999062347A1 (en) | Process for producing seasoning liquors with flavor of vegetables pickled in salted rice bran paste | |
| KR102243418B1 (en) | Novel acetic acid bacteria and process for preparing Kombucha beverages using the same | |
| KR102007569B1 (en) | Fermentation product of seomae mugwort with high palatability and functionality, and method for preparing the same | |
| KR101063624B1 (en) | Schizandra fermented product, fermentation method thereof and food composition comprising the fermented product | |
| KR20150088588A (en) | Novel Gamma-Amino Butyric Acid Producing Strain of Lactobacillus Brevis CFM11 and Use of the Same | |
| KR101493919B1 (en) | Bacillus subtilis cbn strain converting isoflavone glycoside to aglycone and direct-fed microorganisms comprising the same | |
| KR102588661B1 (en) | A novel Lactobacillus plantarum KS2020 strain having GABA production activity, and method for preparing red ginseng fermented product or lacquer tree fermented product containing high concentration γ-PGA and GABA using the strain | |
| KR20180044706A (en) | Method for preparing corn silk fermentation product enhanced gamma-aminobutyric acid contents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| E902 | Notification of reason for refusal | ||
| E601 | Decision to refuse application |