KR20200040407A - 신규 항-c-KIT 항체 - Google Patents
신규 항-c-KIT 항체 Download PDFInfo
- Publication number
- KR20200040407A KR20200040407A KR1020180120233A KR20180120233A KR20200040407A KR 20200040407 A KR20200040407 A KR 20200040407A KR 1020180120233 A KR1020180120233 A KR 1020180120233A KR 20180120233 A KR20180120233 A KR 20180120233A KR 20200040407 A KR20200040407 A KR 20200040407A
- Authority
- KR
- South Korea
- Prior art keywords
- antibody
- cancer
- seq
- kit
- ser
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/575—Immunoassay; Biospecific binding assay; Materials therefor for cancer
- G01N33/57575—Immunoassay; Biospecific binding assay; Materials therefor for cancer involving oncogenic proteins
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- G01N33/574—
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/575—Immunoassay; Biospecific binding assay; Materials therefor for cancer
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6893—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids related to diseases not provided for elsewhere
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
- G01N2333/705—Assays involving receptors, cell surface antigens or cell surface determinants
- G01N2333/70503—Immunoglobulin superfamily, e.g. VCAMs, PECAM, LFA-3
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
- G01N2333/705—Assays involving receptors, cell surface antigens or cell surface determinants
- G01N2333/71—Assays involving receptors, cell surface antigens or cell surface determinants for growth factors; for growth regulators
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Molecular Biology (AREA)
- Hematology (AREA)
- Urology & Nephrology (AREA)
- Biomedical Technology (AREA)
- Biochemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Physics & Mathematics (AREA)
- Pathology (AREA)
- Cell Biology (AREA)
- General Physics & Mathematics (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Food Science & Technology (AREA)
- Analytical Chemistry (AREA)
- Biophysics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Ophthalmology & Optometry (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Hospice & Palliative Care (AREA)
- Oncology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
도 2는 HUVEC 세포를 SCF로 처리하였을 때 2G4 항체의 농도별 튜브 형성 억제 효과를 비-처리 대조군 대비 상대적인 수준으로 나타낸 그래프이다.
도 3은 마우스 유래 내피세포 MS-1을 SCF로 처리하였을 때 2G4 항체의 농도별 튜브 형성 억제 효과를 나타낸 그래프이다.
도 4 및 5는 각각 2G4 항체의 경쇄 가변영역 및 중쇄 가변영역 밴드를 나타낸다.
도 6은 2G4 항체의 경쇄 가변영역의 염기 서열, 아미노산 서열 및 CDR 영역을 나타낸다.
도 7은 2G4 항체의 중쇄 가변영역의 염기 서열, 아미노산 서열 및 CDR 영역을 나타낸다.
도 8은 클로닝, 분리 및 정제를 통해 수득된 2G4 항체의 SDS-PAGE 분석 결과를 나타낸다.
도 9는 2G4 항체의 c-KIT 친화도를 확인하기 위한 SPR 분석 결과를 나타내는 그래프이다.
도 10은 2G4 항체의 c-KIT 결합 도메인을 확인하기 위한 실험결과를 나타낸다.
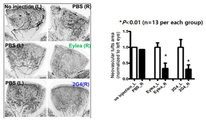
도 11은 마우스 산소 유도 망막증 모델을 이용하여 2G4 항체와 시판 중인 Eylea의 비정상적 혈관형성 억제능을 비교한 결과를 나타낸다.
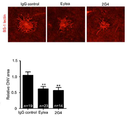
도 12는 브라운 노르웨이 래트 황반변성 모델을 이용하여 2G4 항체와 시판 중인 Eylea의 비정상적 혈관형성 억제능을 비교한 결과를 나타낸다.
도 13은 HUVEC 세포주에서 SCF에 의한 AKT 인산화에 대해 2G4 항체의 억제능을 나타낸다.
도 14는 TF-1 세포주에서 AKT 인산화, c-KIT 인산화, ERK 1/2 인산화 및 β-카테닌에 대해 2G4 항체의 억제능을 나타내며, 이를 통해 백혈병 세포 증식 억제를 보여준다.
도 15는 2G4 항체에 의한 HUVEC과 TF-1 세포 증식 억제능을 나타낸다.
Claims (13)
- c-KIT의 도메인 II에 특이적으로 결합하는 것을 특징으로 하는, 항-c-KIT 항체 또는 이의 항체 단편.
- 제1항에 있어서,
상기 항체는 서열번호 1로 표시되는 경쇄 CDR1, 서열번호 2로 표시되는 경쇄 CDR2 및 서열번호 3으로 표시되는 경쇄 CDR3을 포함하는 경쇄 가변영역; 및 서열번호 4로 표시되는 중쇄 CDR1, 서열번호 5로 표시되는 중쇄 CDR2 및 서열번호 6으로 표시되는 중쇄 CDR3을 포함하는 중쇄 가변영역;을 포함하는 것을 특징으로 하는, 항-c-KIT 항체 또는 이의 항체 단편. - 제1항에 있어서,
상기 항체는 서열번호 7의 아미노산 서열을 포함하는 경쇄 가변영역; 및 서열번호 8의 아미노산 서열을 포함하는 중쇄 가변영역;을 포함하는 것을 특징으로 하는, 항-c-KIT 항체 또는 이의 항체 단편. - 제1항에 있어서,
상기 항체는 인간 IgG1 유래 불변영역을 포함하는 것을 특징으로 하는, 항-c-KIT 항체 또는 이의 항체 단편. - 제1항 내지 제4항 중 어느 한 항의 항-c-KIT 항체 또는 이의 항체 단편을 코딩하는 핵산.
- 제5항에 있어서,
상기 핵산은 (i) 서열번호 9 내지 14, (ii) 서열번호 15 및 16, (iii) 서열번호 17 내지 22, 또는 (iv) 서열번호 23 및 24를 포함하는 것을 특징으로 하는 핵산. - 제5항의 핵산을 포함하는 벡터.
- 제7항의 벡터로 형질전환된 세포.
- 제1항 내지 제4항 중 어느 한 항의 항-c-KIT 항체 또는 이의 항체 단편을 포함하는, 혈관신생 관련 질환의 예방 또는 치료용 약학 조성물.
- 제9항에 있어서,
상기 혈관신생 관련 질환은 암, 백혈병, 안혈관 질환, 류마티스 관절염, 건선, 만성 상처, 만성 염, 혈관종, 혈관섬유종, 혈관기형, 동맥경화, 혈관유착, 혈관염, 화농성 육아종, 수포질환, 폐고혈압증, 천식, 비폴립, 감염성 질환, 염증성 장 질환, 치주 질환, 복막 유착, 자궁내막, 자궁출혈, 난소낭종, 골수염, 골증, 패혈증 및 자가면역질환으로 이루어진 군으로부터 선택된 것을 특징으로 하는 약학 조성물. - 제10항에 있어서,
상기 암은 골암, 폐암, 두부암, 경부암, 갑상선암, 부갑상선암, 비소세포성폐암, 위암, 간암, 췌장암, 피부암, 피부 또는 안구내 흑색종, 직장암, 항문부근암, 결장암, 자궁암, 유방암, 난소암, 나팔관암종, 자궁내막암종, 자궁경부암종, 질암종, 음문암종, 호지킨병, 식도암, 소장암, 내분비선암, 부신암, 연조직 육종, 요도암, 음경암, 전립선암, 방광암, 신장암, 수뇨관암, 신장세포 암종, 신장골반 암종, 중추신경계 종양, 중추신경계 림프종, 척수 종양, 교모세포종, 뇌간 신경교종 및 뇌하수체 선종으로 이루어진 군으로부터 선택된 것을 특징으로 하는 약학 조성물. - 제10항에 있어서,
상기 안혈관 질환은 당뇨성 망막병, 황반변성, 노인성 황반변성, 녹내장, 녹내장성 망막색소변성, 맥락막 혈관신생, 미숙망막증, 각막 이영양증 및 망막층간분리로 이루어진 군으로부터 선택된 것을 특징으로 하는 약학 조성물. - 제1항 내지 제4항 중 어느 한 항의 항-c-KIT 항체 또는 이의 항체 단편을 포함하는 혈관신생 관련 질환 진단용 키트.
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180120233A KR102716514B1 (ko) | 2018-10-10 | 2018-10-10 | 신규 항-c-KIT 항체 |
| BR112021006924-0A BR112021006924A2 (pt) | 2018-10-10 | 2019-10-10 | novo anticorpo anti-c-kit |
| AU2019358647A AU2019358647B2 (en) | 2018-10-10 | 2019-10-10 | Novel anti-c-kit antibody |
| EA202190983A EA202190983A1 (ru) | 2018-10-10 | 2019-10-10 | Новое антитело к с-kit |
| JP2021545343A JP7627045B2 (ja) | 2018-10-10 | 2019-10-10 | 新規抗-c-kit抗体 |
| CN201980079885.XA CN113166256B (zh) | 2018-10-10 | 2019-10-10 | 一种新型抗c-kit抗体 |
| PCT/KR2019/013310 WO2020076105A1 (ko) | 2018-10-10 | 2019-10-10 | 신규 항-c-kit 항체 |
| EP19870797.8A EP3865512A4 (en) | 2018-10-10 | 2019-10-10 | Novel anti-c-kit antibody |
| US17/284,672 US12134646B2 (en) | 2018-10-10 | 2019-10-10 | Anti-C-KIT antibody |
| CA3116154A CA3116154A1 (en) | 2018-10-10 | 2019-10-10 | Novel anti-c-kit antibody |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020180120233A KR102716514B1 (ko) | 2018-10-10 | 2018-10-10 | 신규 항-c-KIT 항체 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20200040407A true KR20200040407A (ko) | 2020-04-20 |
| KR102716514B1 KR102716514B1 (ko) | 2024-10-15 |
Family
ID=70164222
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020180120233A Active KR102716514B1 (ko) | 2018-10-10 | 2018-10-10 | 신규 항-c-KIT 항체 |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US12134646B2 (ko) |
| EP (1) | EP3865512A4 (ko) |
| JP (1) | JP7627045B2 (ko) |
| KR (1) | KR102716514B1 (ko) |
| CN (1) | CN113166256B (ko) |
| AU (1) | AU2019358647B2 (ko) |
| BR (1) | BR112021006924A2 (ko) |
| CA (1) | CA3116154A1 (ko) |
| EA (1) | EA202190983A1 (ko) |
| WO (1) | WO2020076105A1 (ko) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023287251A1 (ko) * | 2021-07-16 | 2023-01-19 | 주식회사 노벨티노빌리티 | Hif-1 알파 저해제 |
| WO2023157989A1 (ko) * | 2022-02-17 | 2023-08-24 | 주식회사 노벨티노빌리티 | 항체-약물 접합체 |
| WO2023204547A1 (ko) * | 2022-04-18 | 2023-10-26 | 주식회사 노벨티노빌리티 | 슈도모나스 외독소 a를 포함하는 c-kit 표적 면역접합체 |
| WO2025151018A1 (ko) * | 2024-01-12 | 2025-07-17 | 주식회사 노벨티노빌리티 | C-kit 및 vegf에 특이적으로 결합하는 이중특이적 항체 및 이의 용도 |
| KR20250111039A (ko) | 2024-01-12 | 2025-07-22 | 주식회사 노벨티노빌리티 | c-Kit 및 VEGF에 특이적으로 결합하는 이중특이적 항체 및 이의 용도 |
Families Citing this family (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2019265019B2 (en) | 2018-05-11 | 2025-11-06 | Beam Therapeutics Inc. | Methods of substituting pathogenic amino acids using programmable base editor systems |
| IL294032A (en) | 2019-12-24 | 2022-08-01 | Carna Biosciences Inc | Compounds that regulate diacylglycerol kinase |
| TWI890283B (zh) | 2020-02-14 | 2025-07-11 | 美商基利科學股份有限公司 | 結合ccr8之抗體及融合蛋白及其用途 |
| JP7651018B2 (ja) | 2021-06-23 | 2025-03-25 | ギリアード サイエンシーズ, インコーポレイテッド | ジアシルグリセロールキナーゼ調節化合物 |
| MX2023014762A (es) | 2021-06-23 | 2024-01-15 | Gilead Sciences Inc | Compuestos moduladores de diacilglicerol quinasa. |
| JP7686091B2 (ja) | 2021-06-23 | 2025-05-30 | ギリアード サイエンシーズ, インコーポレイテッド | ジアシルグリセロールキナーゼ調節化合物 |
| AU2022299051B2 (en) | 2021-06-23 | 2025-03-13 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| US20250230235A1 (en) * | 2021-10-07 | 2025-07-17 | Novelty Nobility Inc. | Treatment of mast cell related disorders |
| AU2022375782B2 (en) | 2021-10-28 | 2026-02-26 | Gilead Sciences, Inc. | Pyridizin-3(2h)-one derivatives |
| JP7787991B2 (ja) | 2021-10-29 | 2025-12-17 | ギリアード サイエンシーズ, インコーポレイテッド | Cd73化合物 |
| US12122764B2 (en) | 2021-12-22 | 2024-10-22 | Gilead Sciences, Inc. | IKAROS zinc finger family degraders and uses thereof |
| KR20240123836A (ko) | 2021-12-22 | 2024-08-14 | 길리애드 사이언시즈, 인코포레이티드 | 이카로스 아연 핑거 패밀리 분해제 및 이의 용도 |
| KR102544135B1 (ko) * | 2022-01-26 | 2023-06-19 | 주식회사 노벨티노빌리티 | c-Kit을 표적으로 하는 면역접합체 |
| TW202340168A (zh) | 2022-01-28 | 2023-10-16 | 美商基利科學股份有限公司 | Parp7抑制劑 |
| WO2023178181A1 (en) | 2022-03-17 | 2023-09-21 | Gilead Sciences, Inc. | Ikaros zinc finger family degraders and uses thereof |
| CN114397453B (zh) * | 2022-03-25 | 2022-06-07 | 江苏美克医学技术有限公司 | 新型冠状病毒突变株的检测试剂盒及其应用 |
| CR20240451A (es) | 2022-04-21 | 2024-12-04 | Gilead Sciences Inc | Compuestos de modulación de kras g12d |
| KR20250028371A (ko) | 2022-07-01 | 2025-02-28 | 길리애드 사이언시즈, 인코포레이티드 | Cd73 화합물 |
| WO2024040194A1 (en) | 2022-08-17 | 2024-02-22 | Capstan Therapeutics, Inc. | Conditioning for in vivo immune cell engineering |
| AU2023409398A1 (en) | 2022-12-22 | 2025-06-05 | Gilead Sciences, Inc. | Prmt5 inhibitors and uses thereof |
| AU2024252725A1 (en) | 2023-04-11 | 2025-11-06 | Gilead Sciences, Inc. | Kras modulating compounds |
| CR20250446A (es) | 2023-04-21 | 2025-12-02 | Gilead Sciences Inc | Inhibidores de prmt5 y usos de los mismos |
| AU2024279278A1 (en) | 2023-05-31 | 2025-12-18 | Capstan Therapeutics, Inc. | Lipid nanoparticle formulations and compositions |
| AU2024306338A1 (en) | 2023-06-30 | 2026-01-08 | Gilead Sciences, Inc. | Kras modulating compounds |
| KR20260046403A (ko) | 2023-07-26 | 2026-04-07 | 길리애드 사이언시즈, 인코포레이티드 | Parp7 저해제 |
| CN121620513A (zh) | 2023-07-26 | 2026-03-06 | 吉利德科学公司 | Parp7抑制剂 |
| EP4516807A1 (en) | 2023-08-28 | 2025-03-05 | Consorcio Centro de Investigación Biomédica en Red | Compositions for use in a method of providing improved hematopoietic stem cell engraftment |
| US20250101042A1 (en) | 2023-09-08 | 2025-03-27 | Gilead Sciences, Inc. | Kras g12d modulating compounds |
| US20250109147A1 (en) | 2023-09-08 | 2025-04-03 | Gilead Sciences, Inc. | Kras g12d modulating compounds |
| US20250127728A1 (en) | 2023-10-05 | 2025-04-24 | Capstan Therapeutics, Inc. | Constrained Ionizable Cationic Lipids and Lipid Nanoparticles |
| WO2025076113A1 (en) | 2023-10-05 | 2025-04-10 | Capstan Therapeutics, Inc. | Ionizable cationic lipids with conserved spacing and lipid nanoparticles |
| US20250154172A1 (en) | 2023-11-03 | 2025-05-15 | Gilead Sciences, Inc. | Prmt5 inhibitors and uses thereof |
| WO2025137640A1 (en) | 2023-12-22 | 2025-06-26 | Gilead Sciences, Inc. | Azaspiro wrn inhibitors |
| WO2025217454A2 (en) | 2024-04-11 | 2025-10-16 | Capstan Therapeutics, Inc. | Ionizable cationic lipids and lipid nanoparticles |
| WO2025217452A1 (en) | 2024-04-11 | 2025-10-16 | Capstan Therapeutics, Inc. | Constrained ionizable cationic lipids and lipid nanoparticles |
| WO2025245003A1 (en) | 2024-05-21 | 2025-11-27 | Gilead Sciences, Inc. | Prmt5 inhibitors and uses thereof |
| US20260098049A1 (en) | 2024-08-12 | 2026-04-09 | Gilead Sciences, Inc. | Kras modulating compounds |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101384360B1 (ko) * | 2012-05-04 | 2014-04-14 | 아주대학교산학협력단 | Scf 또는 이의 수용체를 억제하는 물질을 포함하는 혈관 투과성 관련 질환의 치료 또는 예방용 조성물 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004050850A2 (en) * | 2002-12-02 | 2004-06-17 | Abgenix, Inc. | Antibodies directed to phospholipase a2 and uses thereof |
| BRPI1013177A2 (pt) | 2009-05-28 | 2016-04-12 | Glaxo Group Ltd | construto de ligação de antígeno, polipeptídeo isolado, e, método para tratar doença cardíaca |
| WO2011119948A1 (en) * | 2010-03-26 | 2011-09-29 | Kolltan Pharmaceuticals, Inc. | Anti-kit antibodies and uses thereof |
| AR086044A1 (es) * | 2011-05-12 | 2013-11-13 | Imclone Llc | Anticuerpos que se unen especificamente a un dominio extracelular de c-kit y usos de los mismos |
| NZ630363A (en) * | 2012-07-25 | 2018-09-28 | Celldex Therapeutics Inc | Anti-kit antibodies and uses thereof |
| ES2701051T3 (es) | 2013-03-15 | 2019-02-20 | Novartis Ag | Conjugados de anticuerpo-fármaco |
| WO2016020791A1 (en) * | 2014-08-05 | 2016-02-11 | Novartis Ag | Ckit antibody drug conjugates |
-
2018
- 2018-10-10 KR KR1020180120233A patent/KR102716514B1/ko active Active
-
2019
- 2019-10-10 CA CA3116154A patent/CA3116154A1/en active Pending
- 2019-10-10 JP JP2021545343A patent/JP7627045B2/ja active Active
- 2019-10-10 US US17/284,672 patent/US12134646B2/en active Active
- 2019-10-10 BR BR112021006924-0A patent/BR112021006924A2/pt unknown
- 2019-10-10 WO PCT/KR2019/013310 patent/WO2020076105A1/ko not_active Ceased
- 2019-10-10 AU AU2019358647A patent/AU2019358647B2/en active Active
- 2019-10-10 EP EP19870797.8A patent/EP3865512A4/en active Pending
- 2019-10-10 CN CN201980079885.XA patent/CN113166256B/zh active Active
- 2019-10-10 EA EA202190983A patent/EA202190983A1/ru unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101384360B1 (ko) * | 2012-05-04 | 2014-04-14 | 아주대학교산학협력단 | Scf 또는 이의 수용체를 억제하는 물질을 포함하는 혈관 투과성 관련 질환의 치료 또는 예방용 조성물 |
Non-Patent Citations (1)
| Title |
|---|
| 비특허문헌1* * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023287251A1 (ko) * | 2021-07-16 | 2023-01-19 | 주식회사 노벨티노빌리티 | Hif-1 알파 저해제 |
| WO2023157989A1 (ko) * | 2022-02-17 | 2023-08-24 | 주식회사 노벨티노빌리티 | 항체-약물 접합체 |
| WO2023204547A1 (ko) * | 2022-04-18 | 2023-10-26 | 주식회사 노벨티노빌리티 | 슈도모나스 외독소 a를 포함하는 c-kit 표적 면역접합체 |
| WO2025151018A1 (ko) * | 2024-01-12 | 2025-07-17 | 주식회사 노벨티노빌리티 | C-kit 및 vegf에 특이적으로 결합하는 이중특이적 항체 및 이의 용도 |
| KR20250111039A (ko) | 2024-01-12 | 2025-07-22 | 주식회사 노벨티노빌리티 | c-Kit 및 VEGF에 특이적으로 결합하는 이중특이적 항체 및 이의 용도 |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2019358647B2 (en) | 2024-12-12 |
| EP3865512A4 (en) | 2022-08-03 |
| KR102716514B1 (ko) | 2024-10-15 |
| EA202190983A1 (ru) | 2021-07-23 |
| CN113166256A (zh) | 2021-07-23 |
| US12134646B2 (en) | 2024-11-05 |
| CA3116154A1 (en) | 2020-04-16 |
| JP2022508747A (ja) | 2022-01-19 |
| BR112021006924A2 (pt) | 2021-07-20 |
| EP3865512A1 (en) | 2021-08-18 |
| AU2019358647A1 (en) | 2021-06-03 |
| US20210355212A1 (en) | 2021-11-18 |
| CN113166256B (zh) | 2024-11-29 |
| WO2020076105A1 (ko) | 2020-04-16 |
| JP7627045B2 (ja) | 2025-02-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113166256B (zh) | 一种新型抗c-kit抗体 | |
| KR102924276B1 (ko) | 항-pd-1/vegfa 이기능성 항체, 이의 약제학적 조성물 및 이의 용도 | |
| AU2017317227B2 (en) | Anti-Tim-3 antibodies and use thereof | |
| KR102816742B1 (ko) | 항 SIRPα 항체 | |
| KR20220016974A (ko) | Cd73에 특이적인 결합 분자 및 결합 분자의 용도 | |
| KR102921110B1 (ko) | 항체-약물 접합체 및 이의 용도 | |
| CN107207602A (zh) | 改良的april结合抗体 | |
| CN101291954A (zh) | 具有改变的细胞信号传导活性的修饰的抗原结合分子 | |
| CN111763261B (zh) | 一种融合蛋白及其用途 | |
| CN114057877A (zh) | 抗pd-l1抗体及其应用 | |
| KR102916382B1 (ko) | 항-cldn-18.2 항체 및 그 용도 | |
| EA045024B1 (ru) | Новое антитело к с-kit | |
| KR102572039B1 (ko) | 슈도모나스 외독소 A를 포함하는 c-Kit 표적 면역접합체 | |
| CN102250250A (zh) | 一种人鼠嵌合型抗人CD19抗体Hm2E8b及用途 | |
| RU2829997C1 (ru) | Антитело к cldn-18.2 и его применение | |
| CN116437926B (zh) | 抗pd-1抗体在联合用药中的应用 | |
| RU2811120C2 (ru) | Слитый белок и его применение | |
| HK40069234A (en) | Anti -pd-l1 antibody and use thereof | |
| HK40029004B (en) | A fusion protein and use thereof | |
| HK40029004A (en) | A fusion protein and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
St.27 status event code: A-0-1-A10-A12-nap-PA0109 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| D13-X000 | Search requested |
St.27 status event code: A-1-2-D10-D13-srh-X000 |
|
| D14-X000 | Search report completed |
St.27 status event code: A-1-2-D10-D14-srh-X000 |
|
| PA0302 | Request for accelerated examination |
St.27 status event code: A-1-2-D10-D17-exm-PA0302 St.27 status event code: A-1-2-D10-D16-exm-PA0302 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U11-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| R18 | Changes to party contact information recorded |
Free format text: ST27 STATUS EVENT CODE: A-5-5-R10-R18-OTH-X000 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |