KR20170140407A - 올레핀 중합용 촉매 성분 - Google Patents
올레핀 중합용 촉매 성분 Download PDFInfo
- Publication number
- KR20170140407A KR20170140407A KR1020177034969A KR20177034969A KR20170140407A KR 20170140407 A KR20170140407 A KR 20170140407A KR 1020177034969 A KR1020177034969 A KR 1020177034969A KR 20177034969 A KR20177034969 A KR 20177034969A KR 20170140407 A KR20170140407 A KR 20170140407A
- Authority
- KR
- South Korea
- Prior art keywords
- group
- ethoxycarbonyl
- amino
- catalyst component
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/65912—Component covered by group C08F4/64 containing a transition metal-carbon bond in combination with an organoaluminium compound
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/65—Pretreating the metal or compound covered by group C08F4/64 before the final contacting with the metal or compound covered by group C08F4/44
- C08F4/651—Pretreating with non-metals or metal-free compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F110/00—Homopolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F110/04—Monomers containing three or four carbon atoms
- C08F110/06—Propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2/00—Processes of polymerisation
- C08F2/38—Polymerisation using regulators, e.g. chain terminating agents, e.g. telomerisation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/643—Component covered by group C08F4/64 with a metal or compound covered by group C08F4/44 other than an organo-aluminium compound
- C08F4/6432—Component of C08F4/64 containing at least two different metals
- C08F4/6435—Component of C08F4/64 containing at least two different metals containing magnesium
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/65—Pretreating the metal or compound covered by group C08F4/64 before the final contacting with the metal or compound covered by group C08F4/44
- C08F4/652—Pretreating with metals or metal-containing compounds
- C08F4/654—Pretreating with metals or metal-containing compounds with magnesium or compounds thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2500/00—Characteristics or properties of obtained polyolefins; Use thereof
- C08F2500/12—Melt flow index or melt flow ratio
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2500/00—Characteristics or properties of obtained polyolefins; Use thereof
- C08F2500/15—Isotactic
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Crystallography & Structural Chemistry (AREA)
- Transition And Organic Metals Composition Catalysts For Addition Polymerization (AREA)
Abstract
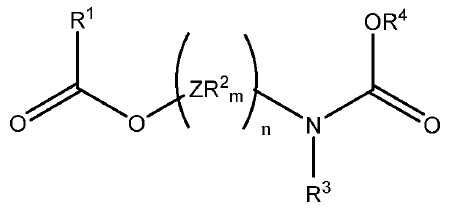
[화학식 1]
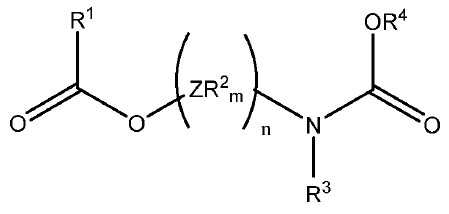
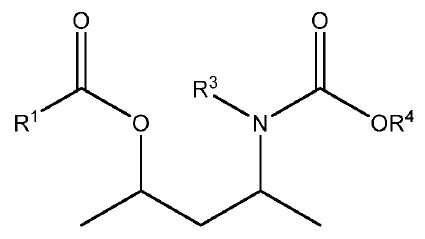
상기 식에서, 독립적으로 Z는 가교기이고, 상기 R2기는 서로 동일하거나 상이하며, 수소 또는 C1-C20 탄화수소 라디칼이며, m은 Z의 원자가를 만족시키는 수이고, n은 1 내지 10의 범위의 정수이고, R1 및 R4는 선택적으로 할로겐, P, S, N 및 O로부터 선택된 헤테로 원자를 함유하는 C1-C15 탄화수소기로부터 선택되고; R3기는 수소 또는 R4기이다. 상기 고체 촉매성분을 기본으로 하는 촉매 시스템은 높은 활성 및 입체 특이성을 제공한다.
Description
Claims (15)
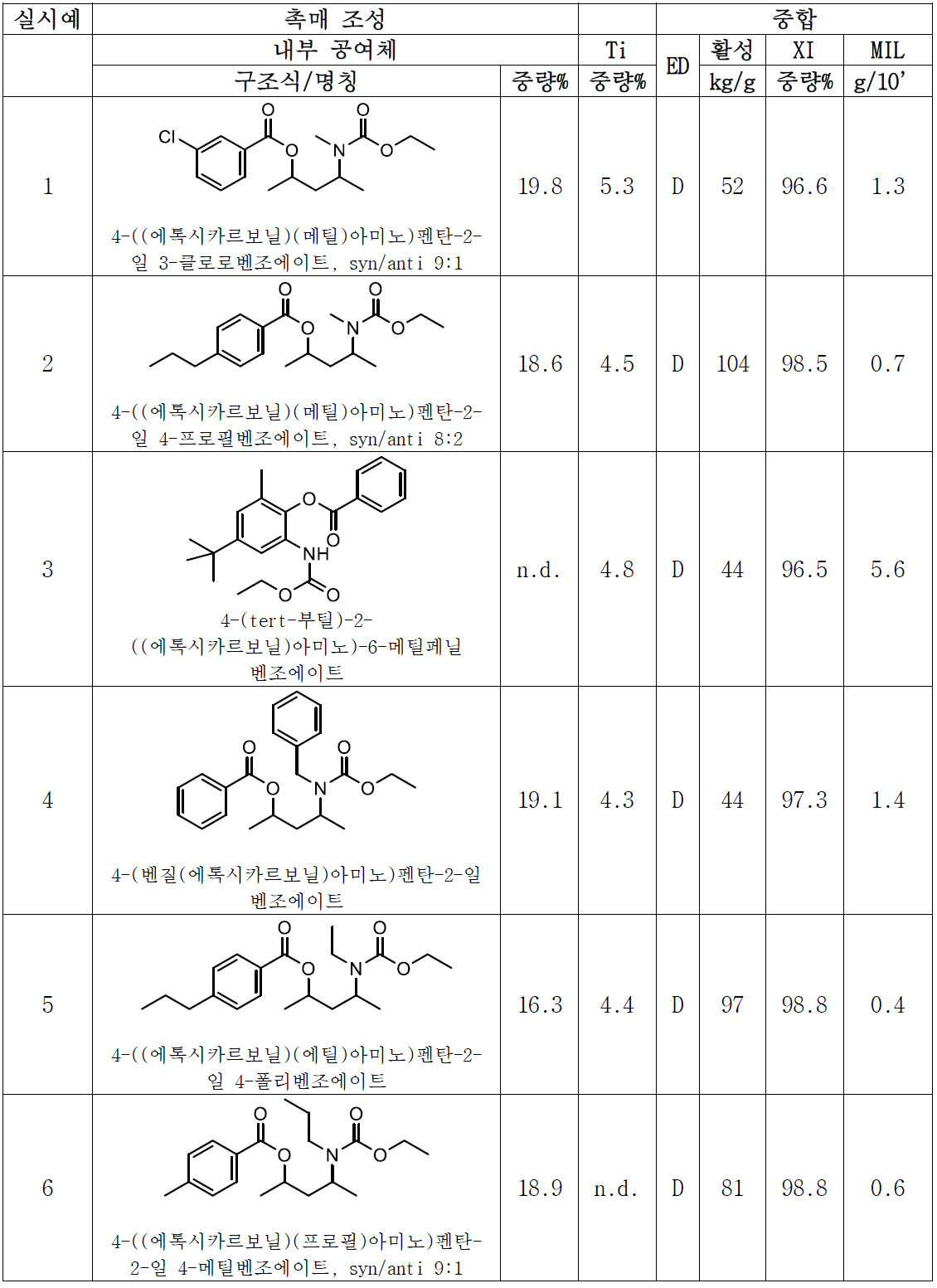
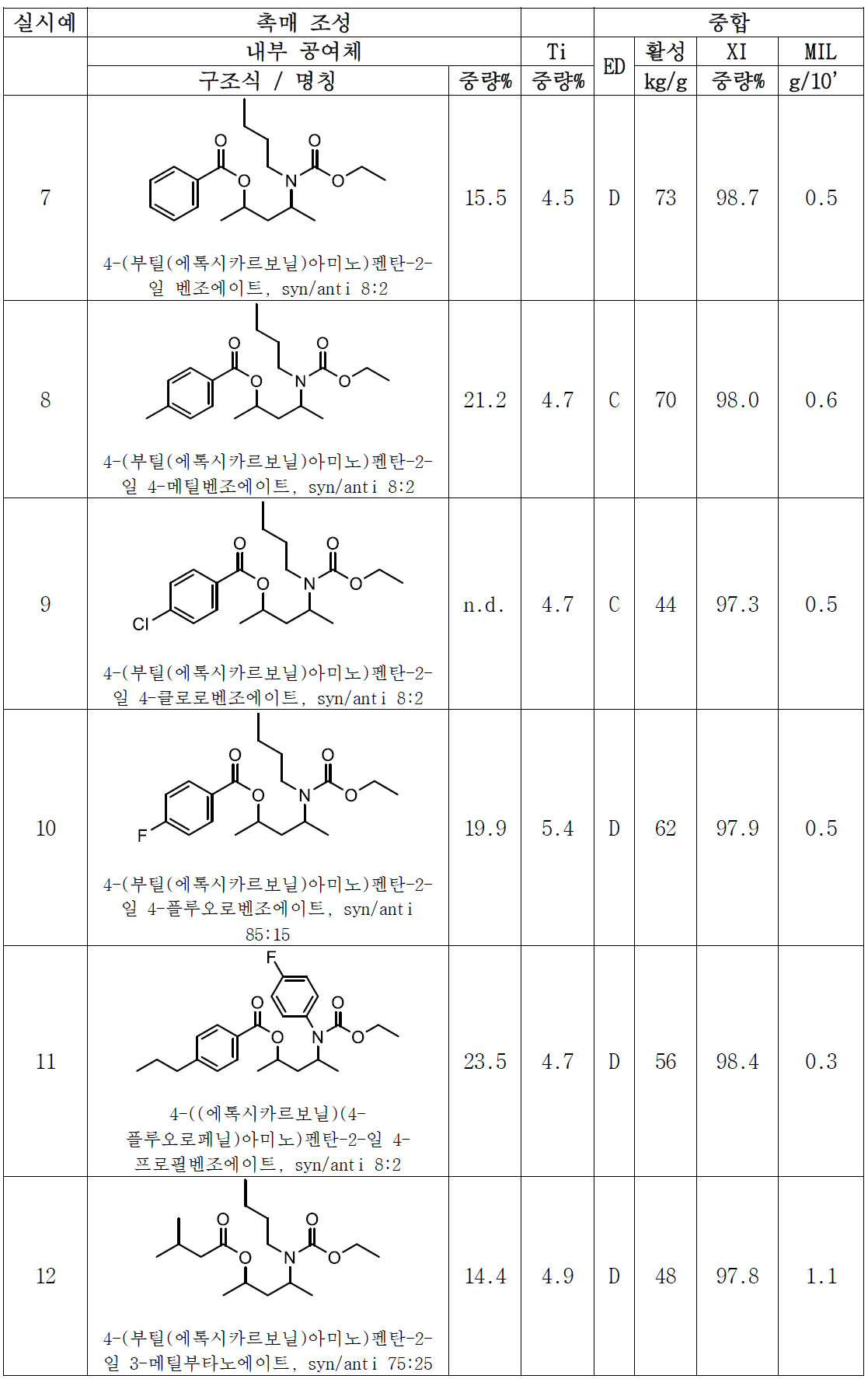
- 올레핀의 중합용 고체 촉매 성분으로서, Mg, Ti 및 하기 화학식 (I)의 전자 공여체를 포함하고,
[화학식 1]
상기 식에서, 독립적으로, Z는 C, Si, Ge, O, N, S 또는 P로부터 선택되며, 단, 원자 O, S 및 N은 화학식 (I)의 에스테르 산소 또는 카르밤산 질소 중 어느 하나에 직접 결합하지 않으며, 상기 R2기는 서로 동일하거나 상이하며, 수소 또는 필요에 따라 할로겐, P, S, N, O 및 Si로부터 선택된 헤테로 원자를 함유하는 C1-C20 탄화수소 라디칼이며, 이들은 함께 융합되어 하나 이상의 환을 형성할 수 있으며, m은 Z의 원자가를 만족시키는 수이고, n은 1 내지 10의 범위의 정수이고, R1 및 R4는 선택적으로 할로겐, P, S, N 및 O로부터 선택된 헤테로 원자를 함유하는 C1-C15 탄화수소기로부터 선택되고; R3 기는 수소 또는 R4 기인, 촉매 성분. - 제1항에 있어서, 화학식 (I)의 (ZR2m)n 기는 C1-C15 탄화수소기로 및/또는 선택적으로 할로겐, P, S, N, O 및 Si로부터 선택된 헤테로 원자로 치환되는, 지방족, 지환족 및 방향족 2가 라디칼로 이루어진 군으로부터 선택되며, 여기서 n은 1 내지 6개의 범위의 원자인, 촉매 성분.
- 제2항에 있어서, 상기 (ZR2m)n기는 n이 1 내지 6개의 범위의 탄소 원자인 지방족 또는 지환족 가교기인, 촉매 성분.
- 제1항에 있어서, R1기는 아릴기 및 알킬아릴기로부터 선택되는, 촉매 성분.
- 제4항에 있어서, R1기는 페닐기로부터 선택되는, 촉매 성분.
- 제5항에 있어서, 상기 페닐기가 할로겐 및/또는 C1-C5 알킬기로 치환되는, 촉매 성분.
- 제1항에 있어서, R3기가 독립적으로 할로겐 또는 C1-C10 알킬기로부터 선택되는, 촉매 성분.
- 제7항에 있어서, R3기가 독립적으로 C1-C5 알킬기로부터 선택되는, 촉매 성분.
- 제1항에 있어서, R4기가 독립적으로 C1-C10 알킬기로부터 선택되는, 촉매 성분.
- 제9항에 있어서, R4기가 C1-C5 알킬기로부터 선택되는, 촉매 성분.
- 제11항에 있어서, R3은 수소 또는 메틸이고, R4는 C1-C15 알킬기인, 촉매 성분.
- 올레핀 중합용 촉매로서:
(i) 제1항 내지 제12항 중 어느 한 항에 따른 고체 촉매 성분 및
(ii) 알킬알루미늄 화합물 및 선택적으로,
(iii) 외부 전자 공여 화합물 사이의 반응 생성물을 포함하는, 올레핀 중합용 촉매. - 제13항에 있어서, 외부 전자 공여체 화합물을 추가로 포함하는, 촉매.
- 촉매계의 존재 하에 수행되는 올레핀 CH2=CHR의 (공)중합의 방법으로서, 여기서 R은 수소 또는 1 내지 12개의 탄소 원자를 갖는 히드로카르빌 라디칼기고, 상기 촉매계가:
i. 제1항 내지 제14항 중 어느 한 항에 따른 고체 촉매 성분;
ii. 알킬알루미늄 화합물 및,
iii. 선택적으로 외부 전자 공여체 화합물 사이의 반응 생성물을 포함하는, 방법.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15168208 | 2015-05-19 | ||
| EP15168208.5 | 2015-05-19 | ||
| PCT/EP2016/061078 WO2016184884A1 (en) | 2015-05-19 | 2016-05-18 | Catalyst components for the polymerization of olefins |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20170140407A true KR20170140407A (ko) | 2017-12-20 |
| KR101895565B1 KR101895565B1 (ko) | 2018-09-06 |
Family
ID=53180640
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020177034969A Active KR101895565B1 (ko) | 2015-05-19 | 2016-05-18 | 올레핀 중합용 촉매 성분 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US10221256B2 (ko) |
| EP (1) | EP3298048B1 (ko) |
| JP (1) | JP6404497B2 (ko) |
| KR (1) | KR101895565B1 (ko) |
| CN (1) | CN107580607B (ko) |
| BR (1) | BR112017023776B1 (ko) |
| ES (1) | ES2746290T3 (ko) |
| RU (1) | RU2667539C1 (ko) |
| WO (1) | WO2016184884A1 (ko) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EA038463B1 (ru) * | 2016-09-29 | 2021-08-31 | Сабик Глоубл Текнолоджиз Б.В. | Прокатализатор для полимеризации олефинов |
| WO2018161854A1 (zh) | 2017-03-10 | 2018-09-13 | 北京利和知信科技有限公司 | 用于烯烃聚合的固体催化剂组分、催化剂及其应用 |
| CN108517021B (zh) * | 2017-03-10 | 2020-10-27 | 北京利和知信科技有限公司 | 一种适用于生产宽分子量分布聚合物的固体催化剂组分、催化剂及其应用 |
| CN108517022B (zh) * | 2017-03-10 | 2020-10-27 | 北京利和知信科技有限公司 | 用于烯烃聚合的固体催化剂组分、及其催化剂和应用 |
| JP7100775B2 (ja) * | 2019-07-03 | 2022-07-13 | バーゼル・ポリオレフィン・イタリア・ソチエタ・ア・レスポンサビリタ・リミタータ | オレフィン重合のための触媒成分 |
| JP2026506029A (ja) * | 2023-03-17 | 2026-02-20 | バーゼル・ポリオレフィン・イタリア・ソチエタ・ア・レスポンサビリタ・リミタータ | オレフィン重合用触媒成分 |
| EP4692050A1 (en) | 2023-03-31 | 2026-02-11 | Mitsui Chemicals, Inc. | Carbamate compound |
| EP4713375A1 (en) * | 2023-05-15 | 2026-03-25 | Basell Poliolefine Italia S.r.l. | Catalyst components for the polymerization of olefins |
| WO2025144069A1 (en) * | 2023-12-28 | 2025-07-03 | Public Joint Stock Company "Sibur Holding" (Pjsc "Sibur Holding") | Procatalyst and catalyst system for olefin polymerization, and method for olefin (co)polymerization |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014001257A1 (en) * | 2012-06-29 | 2014-01-03 | Saudi Basic Industries Corporation | Catalyst composition for polymerization of olefins |
| WO2015032939A1 (en) * | 2013-09-09 | 2015-03-12 | Basell Poliolefine Italia S.R.L. | Catalyst components for the polymerization of olefins |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SE363977B (ko) | 1968-11-21 | 1974-02-11 | Montedison Spa | |
| YU35844B (en) | 1968-11-25 | 1981-08-31 | Montedison Spa | Process for obtaining catalysts for the polymerization of olefines |
| JPS4817672B1 (ko) * | 1970-01-30 | 1973-05-31 | ||
| US3661844A (en) | 1970-12-11 | 1972-05-09 | Cincinnati Milacron Chem | Organic acyl thioacetal and thioketal stabilizers for halogenated resins |
| JPS5615406B2 (ko) * | 1973-08-02 | 1981-04-10 | ||
| GB1603724A (en) | 1977-05-25 | 1981-11-25 | Montedison Spa | Components and catalysts for the polymerisation of alpha-olefins |
| IT1096661B (it) | 1978-06-13 | 1985-08-26 | Montedison Spa | Procedimento per la preparazione di prodotti in forma sferoidale solidi a temperatura ambiente |
| IT1098272B (it) | 1978-08-22 | 1985-09-07 | Montedison Spa | Componenti,di catalizzatori e catalizzatori per la polimerizzazione delle alfa-olefine |
| IT1230134B (it) | 1989-04-28 | 1991-10-14 | Himont Inc | Componenti e catalizzatori per la polimerizzazione di olefine. |
| IT1262934B (it) | 1992-01-31 | 1996-07-22 | Montecatini Tecnologie Srl | Componenti e catalizzatori per la polimerizzazione di olefine |
| IT1262935B (it) | 1992-01-31 | 1996-07-22 | Montecatini Tecnologie Srl | Componenti e catalizzatori per la polimerizzazione di olefine |
| IT1256648B (it) | 1992-12-11 | 1995-12-12 | Montecatini Tecnologie Srl | Componenti e catalizzatori per la polimerizzazione delle olefine |
| IL127230A (en) | 1997-03-29 | 2004-07-25 | Montell Technology Company Bv | Magnesium dichloride-alcohol adducts, process for their preparation and catalyst components obtained therefrom |
| CN1169845C (zh) | 2002-02-07 | 2004-10-06 | 中国石油化工股份有限公司 | 用于烯烃聚合的固体催化剂组分和含该催化剂组分的催化剂及其应用 |
| US8288606B2 (en) | 2005-03-08 | 2012-10-16 | Ineos Usa Llc | Propylene polymer catalyst donor component |
| EP2545087B1 (en) * | 2010-02-26 | 2016-06-22 | W.R. Grace & Co.-Conn. | Procatalyst composition with substituted amide ester internal electron donor |
| US9790291B2 (en) * | 2013-03-14 | 2017-10-17 | Formosa Plastics Corporation, Usa | Non-phthalate compounds as electron donors for polyolefin catalysts |
| EP2803679A1 (en) * | 2013-05-17 | 2014-11-19 | Basell Poliolefine Italia S.r.l. | Catalyst components for the polymerization of olefins |
-
2016
- 2016-05-18 JP JP2017558544A patent/JP6404497B2/ja active Active
- 2016-05-18 BR BR112017023776-8A patent/BR112017023776B1/pt active IP Right Grant
- 2016-05-18 EP EP16725438.2A patent/EP3298048B1/en active Active
- 2016-05-18 WO PCT/EP2016/061078 patent/WO2016184884A1/en not_active Ceased
- 2016-05-18 KR KR1020177034969A patent/KR101895565B1/ko active Active
- 2016-05-18 RU RU2017140819A patent/RU2667539C1/ru active
- 2016-05-18 ES ES16725438T patent/ES2746290T3/es active Active
- 2016-05-18 CN CN201680026686.9A patent/CN107580607B/zh active Active
- 2016-05-18 US US15/574,807 patent/US10221256B2/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014001257A1 (en) * | 2012-06-29 | 2014-01-03 | Saudi Basic Industries Corporation | Catalyst composition for polymerization of olefins |
| WO2015032939A1 (en) * | 2013-09-09 | 2015-03-12 | Basell Poliolefine Italia S.R.L. | Catalyst components for the polymerization of olefins |
Also Published As
| Publication number | Publication date |
|---|---|
| BR112017023776A2 (pt) | 2018-07-31 |
| US10221256B2 (en) | 2019-03-05 |
| JP2018514633A (ja) | 2018-06-07 |
| KR101895565B1 (ko) | 2018-09-06 |
| WO2016184884A1 (en) | 2016-11-24 |
| ES2746290T3 (es) | 2020-03-05 |
| BR112017023776B1 (pt) | 2021-11-16 |
| CN107580607A (zh) | 2018-01-12 |
| JP6404497B2 (ja) | 2018-10-10 |
| RU2667539C1 (ru) | 2018-09-27 |
| US20180142044A1 (en) | 2018-05-24 |
| EP3298048A1 (en) | 2018-03-28 |
| EP3298048B1 (en) | 2019-07-03 |
| CN107580607B (zh) | 2019-09-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101895565B1 (ko) | 올레핀 중합용 촉매 성분 | |
| US9670298B2 (en) | Magnesium dichloride-ethanol adducts and catalyst component obtained therefrom | |
| JP6112593B2 (ja) | オレフィン重合用触媒成分 | |
| US10654946B2 (en) | Catalyst components for the polymerization of olefins | |
| US11873365B2 (en) | Catatlyst components for the polymerization of olefins | |
| KR101872195B1 (ko) | 올레핀 중합용 촉매 성분 | |
| US20130131293A1 (en) | Catalyst components for the polymerization of olefins | |
| US10246532B2 (en) | Catalyst components for the polymerization of olefins | |
| US10113013B2 (en) | Catalyst components for the polymerization of olefins | |
| WO2024194125A1 (en) | Catalyst components for the polymerization of olefins |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| A302 | Request for accelerated examination | ||
| PA0105 | International application |
Patent event date: 20171204 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20171204 Comment text: Request for Examination of Application |
|
| PA0302 | Request for accelerated examination |
Patent event date: 20171204 Patent event code: PA03022R01D Comment text: Request for Accelerated Examination |
|
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20171228 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20180531 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20180831 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20180903 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration | ||
| FPAY | Annual fee payment |
Payment date: 20210702 Year of fee payment: 4 |
|
| PR1001 | Payment of annual fee |
Payment date: 20210702 Start annual number: 4 End annual number: 4 |
|
| FPAY | Annual fee payment |
Payment date: 20220622 Year of fee payment: 5 |
|
| PR1001 | Payment of annual fee |
Payment date: 20220622 Start annual number: 5 End annual number: 5 |
|
| PR1001 | Payment of annual fee |
Payment date: 20230626 Start annual number: 6 End annual number: 6 |
|
| PR1001 | Payment of annual fee |
Payment date: 20240624 Start annual number: 7 End annual number: 7 |