KR20100072184A - 지지체에 나노입자를 퇴적시키는 방법 - Google Patents
지지체에 나노입자를 퇴적시키는 방법 Download PDFInfo
- Publication number
- KR20100072184A KR20100072184A KR1020107005411A KR20107005411A KR20100072184A KR 20100072184 A KR20100072184 A KR 20100072184A KR 1020107005411 A KR1020107005411 A KR 1020107005411A KR 20107005411 A KR20107005411 A KR 20107005411A KR 20100072184 A KR20100072184 A KR 20100072184A
- Authority
- KR
- South Korea
- Prior art keywords
- nanoparticles
- support
- plasma
- colloidal solution
- gold
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 239000002105 nanoparticle Substances 0.000 title claims abstract description 116
- 238000000034 method Methods 0.000 title claims abstract description 79
- 238000000151 deposition Methods 0.000 title claims abstract description 43
- 238000005507 spraying Methods 0.000 claims abstract description 17
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 45
- 239000000725 suspension Substances 0.000 claims description 17
- 239000007789 gas Substances 0.000 claims description 11
- 229910052751 metal Inorganic materials 0.000 claims description 11
- 239000002184 metal Substances 0.000 claims description 11
- 239000002041 carbon nanotube Substances 0.000 claims description 9
- 229910021393 carbon nanotube Inorganic materials 0.000 claims description 9
- 230000004913 activation Effects 0.000 claims description 7
- 239000000203 mixture Substances 0.000 claims description 7
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 claims description 6
- 150000002739 metals Chemical class 0.000 claims description 5
- 229920000642 polymer Polymers 0.000 claims description 5
- 238000004140 cleaning Methods 0.000 claims description 4
- 239000011521 glass Substances 0.000 claims description 4
- 239000001257 hydrogen Substances 0.000 claims description 4
- 229910052739 hydrogen Inorganic materials 0.000 claims description 4
- 229910001092 metal group alloy Inorganic materials 0.000 claims description 4
- 229910044991 metal oxide Inorganic materials 0.000 claims description 4
- 150000004706 metal oxides Chemical class 0.000 claims description 4
- 239000002086 nanomaterial Substances 0.000 claims description 4
- 239000007787 solid Substances 0.000 claims description 4
- 230000003213 activating effect Effects 0.000 claims description 3
- 229910052786 argon Inorganic materials 0.000 claims description 3
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 3
- 239000003570 air Substances 0.000 claims description 2
- UBAZGMLMVVQSCD-UHFFFAOYSA-N carbon dioxide;molecular oxygen Chemical compound O=O.O=C=O UBAZGMLMVVQSCD-UHFFFAOYSA-N 0.000 claims description 2
- 239000000919 ceramic Substances 0.000 claims description 2
- 239000001307 helium Substances 0.000 claims description 2
- 229910052734 helium Inorganic materials 0.000 claims description 2
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 claims description 2
- 239000004065 semiconductor Substances 0.000 claims description 2
- 229910052723 transition metal Inorganic materials 0.000 claims description 2
- 229910000687 transition metal group alloy Inorganic materials 0.000 claims description 2
- 150000003624 transition metals Chemical class 0.000 claims description 2
- 239000010457 zeolite Substances 0.000 claims description 2
- 239000010931 gold Substances 0.000 description 58
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical group [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 52
- 229910052737 gold Inorganic materials 0.000 description 50
- 230000008021 deposition Effects 0.000 description 29
- 239000000523 sample Substances 0.000 description 27
- 229910002804 graphite Inorganic materials 0.000 description 26
- 239000010439 graphite Substances 0.000 description 26
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical group [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 23
- 239000002245 particle Substances 0.000 description 22
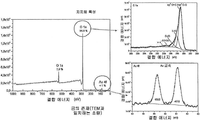
- 238000001228 spectrum Methods 0.000 description 16
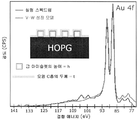
- 238000004833 X-ray photoelectron spectroscopy Methods 0.000 description 13
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 12
- 229910052697 platinum Inorganic materials 0.000 description 11
- 238000004458 analytical method Methods 0.000 description 10
- 229910052799 carbon Inorganic materials 0.000 description 10
- 239000010948 rhodium Substances 0.000 description 10
- 238000004506 ultrasonic cleaning Methods 0.000 description 10
- 229910052703 rhodium Inorganic materials 0.000 description 9
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 9
- 239000000126 substance Substances 0.000 description 9
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 8
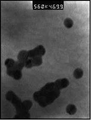
- 238000001239 high-resolution electron microscopy Methods 0.000 description 8
- 229910052760 oxygen Inorganic materials 0.000 description 8
- 239000004094 surface-active agent Substances 0.000 description 8
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 7
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 7
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 7
- 239000010410 layer Substances 0.000 description 7
- 239000001301 oxygen Substances 0.000 description 7
- 229910000831 Steel Inorganic materials 0.000 description 6
- 238000000026 X-ray photoelectron spectrum Methods 0.000 description 6
- 239000010959 steel Substances 0.000 description 6
- 239000000758 substrate Substances 0.000 description 6
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 4
- 125000004429 atom Chemical group 0.000 description 4
- 238000004630 atomic force microscopy Methods 0.000 description 4
- 229920001903 high density polyethylene Polymers 0.000 description 4
- 239000004700 high-density polyethylene Substances 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 239000000377 silicon dioxide Substances 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 3
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 3
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 3
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 3
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 3
- 238000000089 atomic force micrograph Methods 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 239000010949 copper Substances 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 238000000635 electron micrograph Methods 0.000 description 3
- 239000000499 gel Substances 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 239000002048 multi walled nanotube Substances 0.000 description 3
- 230000007935 neutral effect Effects 0.000 description 3
- 238000000527 sonication Methods 0.000 description 3
- 229910052717 sulfur Inorganic materials 0.000 description 3
- 239000011593 sulfur Substances 0.000 description 3
- 239000010936 titanium Substances 0.000 description 3
- 238000004627 transmission electron microscopy Methods 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 2
- 229910045601 alloy Inorganic materials 0.000 description 2
- 239000000956 alloy Substances 0.000 description 2
- 229910052782 aluminium Inorganic materials 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 229910052793 cadmium Inorganic materials 0.000 description 2
- BDOSMKKIYDKNTQ-UHFFFAOYSA-N cadmium atom Chemical compound [Cd] BDOSMKKIYDKNTQ-UHFFFAOYSA-N 0.000 description 2
- CREMABGTGYGIQB-UHFFFAOYSA-N carbon carbon Chemical compound C.C CREMABGTGYGIQB-UHFFFAOYSA-N 0.000 description 2
- 239000011651 chromium Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 239000000084 colloidal system Substances 0.000 description 2
- 238000011109 contamination Methods 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 230000006866 deterioration Effects 0.000 description 2
- HTXDPTMKBJXEOW-UHFFFAOYSA-N dioxoiridium Chemical compound O=[Ir]=O HTXDPTMKBJXEOW-UHFFFAOYSA-N 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 230000005518 electrochemistry Effects 0.000 description 2
- 238000005265 energy consumption Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 239000000446 fuel Substances 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 239000011572 manganese Substances 0.000 description 2
- 239000010955 niobium Substances 0.000 description 2
- 238000009832 plasma treatment Methods 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 229910052707 ruthenium Inorganic materials 0.000 description 2
- 150000003384 small molecules Chemical class 0.000 description 2
- 239000001509 sodium citrate Substances 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- QPLDLSVMHZLSFG-UHFFFAOYSA-N Copper oxide Chemical compound [Cu]=O QPLDLSVMHZLSFG-UHFFFAOYSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- 229910010413 TiO 2 Inorganic materials 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- MCMNRKCIXSYSNV-UHFFFAOYSA-N ZrO2 Inorganic materials O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 1
- 230000001133 acceleration Effects 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000002390 adhesive tape Substances 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 239000002280 amphoteric surfactant Substances 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 230000000845 anti-microbial effect Effects 0.000 description 1
- 239000012300 argon atmosphere Substances 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000003093 cationic surfactant Substances 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 230000005495 cold plasma Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 238000010924 continuous production Methods 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 229910003460 diamond Inorganic materials 0.000 description 1
- 239000010432 diamond Substances 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000002848 electrochemical method Methods 0.000 description 1
- 238000004993 emission spectroscopy Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 150000002343 gold Chemical class 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 229910052741 iridium Inorganic materials 0.000 description 1
- GKOZUEZYRPOHIO-UHFFFAOYSA-N iridium atom Chemical compound [Ir] GKOZUEZYRPOHIO-UHFFFAOYSA-N 0.000 description 1
- 229910052746 lanthanum Inorganic materials 0.000 description 1
- FZLIPJUXYLNCLC-UHFFFAOYSA-N lanthanum atom Chemical compound [La] FZLIPJUXYLNCLC-UHFFFAOYSA-N 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 239000011859 microparticle Substances 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 229910052758 niobium Inorganic materials 0.000 description 1
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 238000004628 non contact atomic force microscopy Methods 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 229910052762 osmium Inorganic materials 0.000 description 1
- SYQBFIAQOQZEGI-UHFFFAOYSA-N osmium atom Chemical compound [Os] SYQBFIAQOQZEGI-UHFFFAOYSA-N 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 1
- NDLPOXTZKUMGOV-UHFFFAOYSA-N oxo(oxoferriooxy)iron hydrate Chemical compound O.O=[Fe]O[Fe]=O NDLPOXTZKUMGOV-UHFFFAOYSA-N 0.000 description 1
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- 238000007747 plating Methods 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 229910052702 rhenium Inorganic materials 0.000 description 1
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 description 1
- 238000004626 scanning electron microscopy Methods 0.000 description 1
- VSZWPYCFIRKVQL-UHFFFAOYSA-N selanylidenegallium;selenium Chemical compound [Se].[Se]=[Ga].[Se]=[Ga] VSZWPYCFIRKVQL-UHFFFAOYSA-N 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical group O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 1
- JBQYATWDVHIOAR-UHFFFAOYSA-N tellanylidenegermanium Chemical compound [Te]=[Ge] JBQYATWDVHIOAR-UHFFFAOYSA-N 0.000 description 1
- 230000008646 thermal stress Effects 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- HRXKRNGNAMMEHJ-UHFFFAOYSA-K trisodium citrate Chemical compound [Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O HRXKRNGNAMMEHJ-UHFFFAOYSA-K 0.000 description 1
- 229940038773 trisodium citrate Drugs 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 238000001771 vacuum deposition Methods 0.000 description 1
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
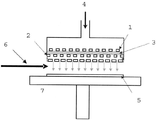
- C23C4/00—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge
- C23C4/02—Pretreatment of the material to be coated, e.g. for coating on selected surface areas
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C24/00—Coating starting from inorganic powder
- C23C24/02—Coating starting from inorganic powder by application of pressure only
- C23C24/04—Impact or kinetic deposition of particles
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C4/00—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge
- C23C4/04—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge characterised by the coating material
- C23C4/06—Metallic material
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C4/00—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge
- C23C4/04—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge characterised by the coating material
- C23C4/06—Metallic material
- C23C4/08—Metallic material containing only metal elements
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C4/00—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge
- C23C4/04—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge characterised by the coating material
- C23C4/10—Oxides, borides, carbides, nitrides or silicides; Mixtures thereof
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C4/00—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge
- C23C4/12—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge characterised by the method of spraying
- C23C4/134—Plasma spraying
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C4/00—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge
- C23C4/12—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge characterised by the method of spraying
- C23C4/14—Coating by spraying the coating material in the molten state, e.g. by flame, plasma or electric discharge characterised by the method of spraying for coating elongate material
- C23C4/16—Wires; Tubes
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Plasma & Fusion (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Manufacture Of Metal Powder And Suspensions Thereof (AREA)
- Powder Metallurgy (AREA)
- Physical Or Chemical Processes And Apparatus (AREA)
- Carbon And Carbon Compounds (AREA)
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP07114344.0 | 2007-08-14 | ||
| EP07114344 | 2007-08-14 | ||
| EP08151463.0 | 2008-02-14 | ||
| EP08151463A EP2093305A1 (fr) | 2008-02-14 | 2008-02-14 | Procédé de dépôt de nanoparticules sur un support |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20100072184A true KR20100072184A (ko) | 2010-06-30 |
Family
ID=39800555
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020107005411A Withdrawn KR20100072184A (ko) | 2007-08-14 | 2008-08-14 | 지지체에 나노입자를 퇴적시키는 방법 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20120003397A1 (enExample) |
| EP (1) | EP2179071B1 (enExample) |
| JP (1) | JP2010535624A (enExample) |
| KR (1) | KR20100072184A (enExample) |
| CN (1) | CN101821421A (enExample) |
| CA (1) | CA2696081A1 (enExample) |
| WO (1) | WO2009021988A1 (enExample) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100983224B1 (ko) * | 2008-04-10 | 2010-09-20 | 김선휘 | 흑연의 미세입자 및 흑연 콜로이드 제조방법 |
| KR20180103525A (ko) * | 2017-03-10 | 2018-09-19 | 경북대학교 산학협력단 | 플라즈마 발생장치 |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101127121B1 (ko) * | 2009-06-12 | 2012-03-20 | 한국과학기술원 | 공기 분사를 이용하여 목표 기판에 금속 나노입자층을 형성시키는 방법 |
| DE102009037846A1 (de) * | 2009-08-18 | 2011-02-24 | Siemens Aktiengesellschaft | Partikelgefüllte Beschichtungen, Verfahren zur Herstellung und Verwendungen dazu |
| CN101840852A (zh) * | 2010-04-02 | 2010-09-22 | 中国科学院半导体研究所 | 在图形化的半导体衬底上制作有序半导体纳米结构的方法 |
| EP2611948A2 (en) * | 2010-09-01 | 2013-07-10 | Facultés Universitaires Notre-Dame de la Paix | Method for depositing nanoparticles on substrates |
| CN102258921A (zh) * | 2011-05-27 | 2011-11-30 | 安徽南风环境工程技术有限公司 | 一种油烟吸附过滤网及其制备方法 |
| DK2736837T3 (da) | 2011-07-26 | 2021-10-25 | Oned Mat Inc | Fremgangsmåde til fremstilling af nanosiliciumtråde |
| US9963345B2 (en) * | 2013-03-15 | 2018-05-08 | The United States Of America As Represented By The Administrator Of Nasa | Nanoparticle hybrid composites by RF plasma spray deposition |
| KR101622033B1 (ko) * | 2013-08-01 | 2016-05-17 | 주식회사 엘지화학 | 탄소 담체 - 금속 나노 입자 복합체의 제조방법 및 이에 의해 제조된 탄소 담체 - 금속 나노 입자 복합체 |
| EP2937890B1 (en) * | 2014-04-22 | 2020-06-03 | Europlasma nv | Plasma coating apparatus with a plasma diffuser and method preventing discolouration of a substrate |
| KR102254644B1 (ko) * | 2014-12-19 | 2021-05-21 | (주)바이오니아 | 바인더 결합형 탄소나노구조체 나노다공막 및 그의 제조방법 |
| CN104711568B (zh) * | 2015-02-27 | 2017-11-14 | 南京邮电大学 | 一种在金属丝上包裹碳纳米材料的制备方法及其装置 |
| US20180248199A1 (en) * | 2015-08-27 | 2018-08-30 | Osaka University | Method for manufacturing metal nanoparticles, method for manufacturing metal nanoparticle-loaded carrier, and metal nanoparticle-loaded carrier |
| CN105369180A (zh) * | 2015-12-02 | 2016-03-02 | 广州有色金属研究院 | 一种致密氧离子-电子混合导体氧化物涂层的制备方法 |
| ES3032418T3 (en) | 2016-07-15 | 2025-07-18 | Oned Mat Inc | Manufacturing apparatus and method for making silicon nanowires on carbon based powders for use in batteries |
| AU2019364543A1 (en) * | 2018-10-24 | 2021-05-27 | Atmospheric Plasma Solutions, Inc. | Plasma source and method for preparing and coating surfaces using atmospheric plasma pressure waves |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5141017B2 (enExample) * | 1972-12-26 | 1976-11-08 | ||
| JPH0941251A (ja) * | 1995-07-28 | 1997-02-10 | Kyodo Kumiai Zou Shudan | 撥水布に合成樹脂立体模様の形成付着方法 |
| JP3511128B2 (ja) * | 1998-03-02 | 2004-03-29 | 日立造船株式会社 | 金属微粒子の製造方法および同微粒子の多孔質担体への担持方法 |
| JP3256741B2 (ja) * | 1998-07-24 | 2002-02-12 | 独立行政法人産業技術総合研究所 | 超微粒子成膜法 |
| JP2002237480A (ja) * | 2000-07-28 | 2002-08-23 | Sekisui Chem Co Ltd | 放電プラズマ処理方法 |
| JP2002121024A (ja) * | 2000-10-12 | 2002-04-23 | Seiko Epson Corp | 酸化チタン膜の製造方法、酸化チタン膜および太陽電池 |
| JP2003007497A (ja) * | 2001-06-19 | 2003-01-10 | Pearl Kogyo Kk | 大気圧プラズマ処理装置 |
| DE10211701A1 (de) * | 2002-03-16 | 2003-09-25 | Studiengesellschaft Kohle Mbh | Verfahren zur in situ Immobilisierung von wasserlöslichen nanodispergierten Metalloxid-Kolloiden |
| GB0208263D0 (en) * | 2002-04-10 | 2002-05-22 | Dow Corning | Protective coating composition |
| WO2004035496A2 (en) * | 2002-07-19 | 2004-04-29 | Ppg Industries Ohio, Inc. | Article having nano-scaled structures and a process for making such article |
| US20050031876A1 (en) * | 2003-07-18 | 2005-02-10 | Songwei Lu | Nanostructured coatings and related methods |
| EA010388B1 (ru) * | 2003-01-31 | 2008-08-29 | Дау Корнинг Айэлэнд Лимитед | Электродный узел для генерации плазмы |
| JP2004356558A (ja) * | 2003-05-30 | 2004-12-16 | Toshio Goto | コーティング装置およびコーティング方法 |
| CH696811A5 (de) * | 2003-09-26 | 2007-12-14 | Michael Dvorak Dr Ing Dipl Phy | Verfahren zur Beschichtung einer Substratoberfläche unter Verwendung eines Plasmastrahles. |
| JP2005163117A (ja) * | 2003-12-03 | 2005-06-23 | Ainobekkusu Kk | 金属コロイドの製造方法及びこの方法によって製造された金属コロイド |
| JP4177244B2 (ja) * | 2003-12-15 | 2008-11-05 | 日信工業株式会社 | 多孔質複合金属材料の製造方法 |
| JP4300105B2 (ja) * | 2003-12-19 | 2009-07-22 | 株式会社コーセー | 抗酸化剤 |
| FR2877015B1 (fr) * | 2004-10-21 | 2007-10-26 | Commissariat Energie Atomique | Revetement nanostructure et procede de revetement. |
| WO2006048649A1 (en) * | 2004-11-05 | 2006-05-11 | Dow Corning Ireland Limited | Plasma system |
| DE102006005775A1 (de) * | 2006-02-07 | 2007-08-09 | Forschungszentrum Jülich GmbH | Thermisches Spritzverfahren mit kolloidaler Suspension |
-
2008
- 2008-08-14 WO PCT/EP2008/060676 patent/WO2009021988A1/fr not_active Ceased
- 2008-08-14 EP EP08787216.4A patent/EP2179071B1/fr not_active Not-in-force
- 2008-08-14 CA CA2696081A patent/CA2696081A1/en not_active Abandoned
- 2008-08-14 JP JP2010520582A patent/JP2010535624A/ja active Pending
- 2008-08-14 US US12/673,437 patent/US20120003397A1/en not_active Abandoned
- 2008-08-14 CN CN200880111576A patent/CN101821421A/zh active Pending
- 2008-08-14 KR KR1020107005411A patent/KR20100072184A/ko not_active Withdrawn
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100983224B1 (ko) * | 2008-04-10 | 2010-09-20 | 김선휘 | 흑연의 미세입자 및 흑연 콜로이드 제조방법 |
| KR20180103525A (ko) * | 2017-03-10 | 2018-09-19 | 경북대학교 산학협력단 | 플라즈마 발생장치 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2010535624A (ja) | 2010-11-25 |
| CN101821421A (zh) | 2010-09-01 |
| US20120003397A1 (en) | 2012-01-05 |
| EP2179071B1 (fr) | 2016-04-13 |
| CA2696081A1 (en) | 2009-02-19 |
| EP2179071A1 (fr) | 2010-04-28 |
| WO2009021988A1 (fr) | 2009-02-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20100072184A (ko) | 지지체에 나노입자를 퇴적시키는 방법 | |
| Achour et al. | Influence of plasma functionalization treatment and gold nanoparticles on surface chemistry and wettability of reactive-sputtered TiO2 thin films | |
| CN103747870B (zh) | 用热稳定金属合金纳米颗粒结构化的基材表面,制备其的方法及其特别是作为催化剂的用途 | |
| US6656339B2 (en) | Method of forming a nano-supported catalyst on a substrate for nanotube growth | |
| KR100831069B1 (ko) | 나노크기의 금속분화 촉매 및 그의 제조방법 | |
| US20060083674A1 (en) | Method for forming catalyst metal particles for production of single-walled carbon nanotube | |
| CN101370734A (zh) | 用富勒烯官能化的碳纳米管 | |
| EP2611948A2 (en) | Method for depositing nanoparticles on substrates | |
| Choi et al. | Significant enhancement of the NO2 sensing capability in networked SnO2 nanowires by Au nanoparticles synthesized via γ-ray radiolysis | |
| WO2005075340A1 (en) | A method for the preparation of y-branched carbon nanotubes | |
| Ehrenburg et al. | Electroreduction of nitrate anions on cubic and polyoriented platinum nanoparticles modified by copper adatoms | |
| JP2011006775A (ja) | 貴金属で表面修飾されたナノポ−ラス金属及びナノポ−ラス金属の貴金属による表面修飾 | |
| JP2004149954A (ja) | 金属/金属化合物被覆カーボンナノファイバー及びその製造方法 | |
| US10828622B2 (en) | Engineering high-performance palladium core magnesium oxide porous shell nanocatalysts via heterogeneous gas-phase synthesis | |
| Lapham et al. | The preparation of NiCo2O4 films by electrostatic spray deposition | |
| Dudin et al. | Electro-oxidation of hydrazine at gold nanoparticle functionalised single walled carbon nanotube network ultramicroelectrodes | |
| US20240010850A1 (en) | Carbon supported surface functionalized silver nanoparticles for ink/electrodes/mea | |
| EP2093305A1 (fr) | Procédé de dépôt de nanoparticules sur un support | |
| JP2024046607A (ja) | ガス拡散電極用の炭素担持カルボキシル官能化銀ナノ粒子 | |
| Su et al. | Pt monolayer deposition onto carbon nanotube mattes with high electrochemical activity | |
| CN111575665B (zh) | 一种纳米羰基金属复合超滑含氢碳薄膜的制备方法 | |
| Savinova et al. | Electrocatalytic properties of platinum anchored to the surface of highly oriented pyrolytic graphite | |
| KR101070870B1 (ko) | 실리콘 옥사이드 나노 와이어에 지지된 금속 나노 입자 촉매 및 이의 제조방법 | |
| Choi et al. | Platinum nanoparticle-functionalized tin dioxide nanowires via radiolysis and their sensing capability | |
| Yoshida et al. | The patterned Au oxide layer formation on Au surfaces by F2 laser irradiation under the atmospheric conditions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20100311 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PC1203 | Withdrawal of no request for examination | ||
| WITN | Application deemed withdrawn, e.g. because no request for examination was filed or no examination fee was paid |